Are Our Screening Algorithms Appropriate? Prospects for the Next 10 Years
|
|
|
- Loreen Spencer
- 5 years ago
- Views:
Transcription
1 Are Our Screening Algorithms Appropriate? Prospects for the Next 10 Years IPFA/PEI 18 th International Workshop on Surveillance and Screening of Blood Borne Pathogens May 2011, Dublin, Ireland Jay Epstein, M.D. CBER, FDA
2 Are Our Screening Algorithms Appropriate? Value of risk factor screening Value of laboratory testing Limits of bacterial culture Challenges in parasitic detection New technology opportunities Diagnostic methods Pathogen reduction Stem cell based product manufacturing The hospital perspective on blood safety Personal speculations on the future
3 The Current Screening Paradigm Donor deferral based on epidemiologically determined risk factors for TTIDs (medical, geographical and behavioral risks) Laboratory testing for specific agents Serologic testing for antibodies and antigens NAT Bacterial detection in platelets
4 Historical Transfusion Risks of HIV, HCV, and HBV* Risk of Transmission per Unit of Transfused Blood Component HBV HIV HCV *Peterman TA, et al., Transfusion 1987;27: Busch, MP. Transfusion 2006;46: Zou, et al. Transfusion 2009; 49: and
5 Value of Risk Factor Based Deferrals Avoids collection of test positive units that could be released in error Reduces risk to recipients from window period collections Behavioral exclusions foster donation by persons with healthier lifestyles, possibly reducing some EID risk
6 HIV Related Behavioral Deferrals Reduce HIV Marker Prevalence in Donors 0.47% confirmed HIV+ in donor-age general population (McQuillan, 1994) 0.03% confirmed HIV+ in First Time Donors (REDS survey circa 1995) Estimated 93.6% (16 fold) reduction in HIV seroprevalence
7 MSM History Deferral Reduces MSM Behavior Prevalence in Accepted Blood Donors 4.1% prevalence of MSM - past 5 yrs in male general population (Laumann 1994) 0.57% MSM-77 risk in accepted male donors (Williams 1997) Estimated 86.1% (7.2 fold) reduction
8 IDU History Deferral Reduces IDU History Prevalence in Accepted Blood Donors 3.9% IDU since 1978 in general population (Dallas Household Survey 1994) 0.51% IDU-ever among accepted donors (Williams 1997) Estimated 86.9% (7.6 fold) reduction
9 Prevalence of Reported Behavioral Risks in Accepted Blood Donors* IDU-ever 0.5% Sex with IDU (12 mos) 0.4% MSM Ever (males) 2.4% MSM (males) 0.6% 1.2% (n=280) Sex with CSW (males) 0.5% Any deferrable risk 1.9% *REDS Donor Surveys
10 Value of Laboratory Testing Avoids transfusion of marker positive units But, Requires knowledge about the TTID agent Is limited by the sensitivity of the test technology Can be falsely negative (e.g. with viral variants) Enables monitoring of risk in donors Calculation of prevalence, incidence and residual risk yield knowledge of blood safety Studies of risk factors in positive donors permit improvements in donor selection
11 Safety Impact of Laboratory Testing (Based on ARC data for a,b ) Marker Positive Rate (#/100K) Residual Risk (#/100K) Risk Reduction Factor HBV a HCV b 37.43; est d 85% viremic HIV b a From Zou, et al. Transfusion 2010;50: and 2009;49: b From Zou, et al. Transfusion 2010;50: and 2010;50:
12 Value of NAT in Viral Disease Testing Reduction in Window Periods HIV: days vs. antibody tests HCV: days vs. antibody tests HBV: 7-12 days vs. HBsAg tests Reduction in Transmissions Approximately 5 HIV and 56 HCV transmissions per year were prevented by NAT (Stramer, et al., 2004) Interdiction of over 2000 WNV infected units since implementation of WNV NAT in 2003
13 Limits of Early Bacterial Culture (1) ARC experience 1 ( ) with BacT/ALERT: Sampling volume 8mL, 24hrs, with diversion pouch 781,936 collections, ~ 1.3 million components True positive (TP) rate ~ 167 per million collections 11 septic reactions: ~ 1 per 122,000 components 1 death: ~ 1 per 1.3 million components Sensitivity of BacT/ALERT culture at 24 hours based on reculture at expiration: 22-40% 2,3,4 1. Eder A, et al, Transfusion 2009;49: and Richard Benjamin AABB 2008 presentation 2. Dumont LJ, et al. PASSPORT study results. Transfusion 2010; 50: Murphy WG et al. Screening platelet concentrates for bacterial contamination. Vox Sanguinis 2008;95: Pearce S et al. Screening of platelets for bacterial contamination at the Welsh Blood Service. Transfusion Medicine 2011;21:25-32
14 Limits of Early Bacterial Culture (2) Verax PGD test post marketing studies* 18,449 apheresis platelet units, negative by early bacterial culture, were retested on day of transfusion by PGD test with positive results confirmed by culture A subset of 3,627 units had both PGD positive and negative results confirmed by culture 8 true positives picked up days 3 through day 5 (5 Coag Neg Staph; 2 Bacillus cereus; 1 Enterococcus faecalis) for a TP rate of ~ 1/2,306 = 433 per million 1 false negative tested on day 5 (concurrent culture grew Strep oralis-viridans group) for a FN rate of 1/3,627 False positive rate: 1/113 ~ 0.85% *Jacobs et al. Abstract and Oral presentation at AABB Baltimore 2010.Transfusion 2010;50: s2, 30A.
15 Challenges in the Detection of Plasmodia and Babesia Infections with Plasmodia and Babesia can be transmitted by transfusion of both RBCs and Platelets Plasmodia and Babesia cause chronic, asymptomatic infections with very low level parasitemias Plasmodia and Babesia infections can be transmitted by very small numbers of parasites Even the most sensitive nucleic acid based tests may fail to detect parasites in donors with low-grade, asymptomatic infections due to limitations of sampling
16 Limitations of Antibody Testing for Detection of Infections with Plasmodia or Babesia Antibody testing is highly effective to identify historic exposures to Plasmodia and Babesia parasites But, Infections with Plasmodia and Babesia result in highly specific antibodies detectable by IFA or ELISA For Plasmodia the window period from appearance of blood stage parasites to detectable antibodies is about 14 days (not known for Babesia) IgG titers may persist for months or years and cannot distinguish current from past infections. Antibody testing would then cause deferral of recovered donors who would not transmit malaria or babesiosis.
17 New Technology Opportunities Testing Multiplex NAT for arboviruses (WNV, Dengue viruses, Chikungunya) Nanoparticle based assays Microfluidics Alternative manufacturing Pathogen Reduction Extracorporeal Blood Cell Cultivation
18 Nanoparticle Based Diagnostics Nanotechnology is engineering at the atomic, molecular, or macromolecular scale, leading to controlled creation and use of structures with a length scale of nanometers (nm) At the nanoscale, physical, chemical, and biological properties of materials differ fundamentally from those of the corresponding bulk material e.g. color, melting point etc. opening new opportunities for process design Nanotechnology-based techniques could potentially provide a new generation of rapid, sensitive assays for simultaneous detection of multiple analytes in a miniaturized format.
19 PCR-free Nanoparticle-based Whole Genomic Assay for Rapid Detection of Nucleic Acids AAAAAA AAAAAA AAAAAA AAAAAA 15nm Au NP AAAAAA TTTTTT AAAAAA TTTTTT 15nm Au NP DNA/RNA target Hybridization Au-probe Hybridization Silver staining
20 Genomic Oligo-Array for Detection of HIV Subtypes, CRFs and Drug Resistant Mutations Subtype A B C D H F J K CRF01_AE CRF02_AG Gag Pol Env Predicted genotype: CRF01_AE / 02_AG / B
21 Europium NP-based Immunoassay (ENIA) Using Time-Resolved Fluorescence SA-Eu NP r = R 2 = p < primary antibody target 2 nd antibody biotin anti-2 nd antibody SA-Eu NP SA Eu + NP LOD: 0.5 pg/ml or 0.5 ng/l 21 fmol/l or 2 attomoles of p24 (24KDa) Tang S and Hewlett IK. J. Infect. Dis. 2010; 201 (suppl 1):S59-S64
22 On-Chip Detection of HIV p24 Proteins Using Europium Nanoparticles Perkin Elmer Wallac Plate Reader
23 Microfluidic Chip-Based Rapid, Multiplexed Pathogen Detection Fully integrated microchip for rapid HIV diagnosis in resourcelimited settings; up to 4 pathogens can be multiplexed in above format Manual pumping and on chip reagent storage Silver Cluster Porous polymer detection elements Label free SERS detection using monolith detector containing silver clusters
24 Improving Sensitivity of NAT for Plasmodia and Babesia Sample concentration and removal of the interfering substances from the red cell (e.g., hemoglobin) Targeting high copy number genes for amplification
25 P. falciparum NAT: Sample Concentration and Hemoglobin Removal by Saponin Lysis of RBC 100 μl 500 μl 5 ml Normal blood parasite 3-1 parasites 4-10 parasites parasites parasites parasites parasites 1 - Normal blood parasite 3-5 parasites 4-50 parasites 5 5x10 2 parasites 6 5x10 3 parasites 7 5x10 4 parasites 8 5x10 5 parasites 1 - Normal blood parasite 3-1 parasites 4-10 parasites parasites parasites parasites parasites PCR sensitivity: 2-10 P. falciparum parasites/ml of blood ,000 fold more sensitive than thick-film microscopy S. Kumar et al., unpublished)
26 Merits of Pathogen Reduction Technology as an Alternative to Donor Screening and Testing Advantages Shown effective against many organisms including some emerging pathogens May prevent GVHD and other wbc related adverse events Disadvantages May not be effective against all organisms May not be 100% effective even against sensitive pathogens Current technologies are not applicable to all types of transfusion products May have toxicity due to residual compounds May damage the transfusion product May lead to alloimmunization by neoantigens May cause unexpected adverse events
27 Pathogen Reduced Platelets Have Lower Corrected Count Increments (CCI) Clinical Trial Patients in study % of plasma stored platelets CCI at 1 hr P value SPRINT 1, a % < HOVON 1, b % < MIRASOL 2, c % < = UVA/psoralen 2 = UVB/riboflavin a = McCullough, J et al Blood Sep 1;104(5): b = Kerkhoffs JL et al. Br J Haematol Jul;150(2): c = Goodrich et al. Transfusion, May 2010
28 Hemostatic Efficacy for UV A/psoralen (Intercept) Treated Platelets SPRINT study Control platelets Pathogen reduced platelets Proportion of pts with Grade 2 bleeding Days of Grade 2 bleeding % patients with Grade 2-4 bleeding 58.5% 57.5% NS for inferiority HOVON study Control platelets Pathogen reduced platelets % of patients with Grade 1-3 bleeding p p
29 Hemostatic Efficacy for UVB/riboflavin (Mirasol) Treated Platelets MIRASOL study % of patients with Grade 2-4 bleeding Control platelets Pathogen reduced platelets NS p
30 FDA Perspective on Pathogen Reduction Pathogen Reduction of labile blood products could improve blood product safety, especially for platelets, but should not add greater risks Clinical trials with Pathogen Reduced red cells have demonstrated antibody generation Clinical trials with Pathogen Reduced platelets have demonstrated decreased efficacy and associated adverse events including acute lung injury in the SPRINT trial. Further clinical trials of current technologies are needed to resolve FDA s concerns over decreased efficacy and increased adverse events seen with Pathogen Reduced platelets
31 In Vitro Production of Stem Cell- Derived Transfusion products -1 Small scale production of red cells and platelets from stem cells is already possible Benefits of this technology might include generation of universal red cells that would not need typing Continued supply Pathogen reduced during processing Extended storage due to cell synchronization
32 In Vitro Production of Stem Cell- Derived Transfusion products -2 Major hurdles that remain are scale up of process that produces uniform product with appropriate quality control automation High cost per transfusion product due to cytokine cocktail requirements concerns about transfusion of undifferentiated pluripotent stem cells Efforts are continuing in the hope that this technology will transform transfusion medicine
33 The Hospital Side of Transfusion Focused on patient care Requires good transfusion practices Staff education Blood management
34 The Biggest Risks Are Not Infections HIV HCV HBV Death: General anesthesia Death: Medical error Death: Hosp. infection vcjd Bacteria in platelets Mistransfusion TRALI GVHD Cardiac Risk per unit. Modified from S. Dzik, MGH, Boston Metabolic risk in neonates Under transfusion?
35 Transfusion Safety Matters In the Hospital Issue Recruit Screen donor Collect & Prepare Infectious disease tests Pre-transfusion testing Medical decision to transfuse Administer (bedside) Monitor & evaluate Product Hospital-based steps Process While products are safer than ever before, a lack of progress exists for risk reduction in the hospital-based steps of transfusion care. After W.H. Dzik
36 Categories of Events Reported by the SHOT Program in England, TRALI Infections GVHD PTP Delayed reaction Incorrect blood transfused Acute reaction
37 Current Dilemmas in Transfusion How best to address EID threats How to reduce hospital transfusion errors When to transfuse red cells and platelets Safety of older stored red cells Optimal management of severe bleeding Better prevention of TRALI
38 Personal 10 Year Speculations -1 Better testing > Pathogen Reduction > Novel Manufacturing In the next five years: Additional sensitive bacterial testing will be performed at the time of platelet release HBV NAT will replace HBsAg Donor blood will be tested for plasmodia, babesia and dengue viruses (plus CHKV in some regions) B19 and HAV testing will be elevated to donor screens There still will not be donor testing for vcjd New infectious threats will continue to arise!
39 Personal 10 Year Speculations - 2 In the next ten years: Pathogen reduction of Whole Blood and components will be established, and then Testing will be discontinued for syphilis, WNV, CMV, Chagas disease and anti-hbc Deferrals for malaria will be discontinued Some behavior-based deferrals will be discontinued (e.g. for MSM)
40 Additional long term complications of transfusion will be discovered! Personal 10 Year Speculations - 3 In the next ten years (continued) We will have better understanding when blood products should be transfused Management of severe bleeding will become evidence based; HBOCs will still be under study We will know whether older stored blood is safe Blood for emergency use will be manipulated to remove ABO and Rh antigens reducing the need for Type O, Rh negative donations Antigen null and rare red cells types will be generated in extracorporeal bioreactors
Babesia from a donor perspective
 Babesia from a donor perspective American Red Cross, Massachusetts Region Bryan Spencer, MPH Research Scientist American Society for Apheresis Annual Meeting May 8, 2015 San Antonio, TX The need is constant.
Babesia from a donor perspective American Red Cross, Massachusetts Region Bryan Spencer, MPH Research Scientist American Society for Apheresis Annual Meeting May 8, 2015 San Antonio, TX The need is constant.
Keeping the Blood Supply Safe Current and Future Strategies. Marissa Li, MD
 Keeping the Blood Supply Safe Current and Future Strategies Marissa Li, MD Overview Pre-collection Donor Screening Collection Visual Inspection Aseptic Processing Leukoreduction of Apheresis Collections
Keeping the Blood Supply Safe Current and Future Strategies Marissa Li, MD Overview Pre-collection Donor Screening Collection Visual Inspection Aseptic Processing Leukoreduction of Apheresis Collections
Be ready. The INTERCEPT Blood System for Platelets and Plasma pathogen reduction system
 Be ready. The INTERCEPT Blood System for Platelets and Plasma pathogen reduction system INTERCEPT Blood System for Platelets an Blood safety is changing Safe donated blood is critical to improving certain
Be ready. The INTERCEPT Blood System for Platelets and Plasma pathogen reduction system INTERCEPT Blood System for Platelets an Blood safety is changing Safe donated blood is critical to improving certain
Screening donors and donations for transfusion transmissible infectious agents. Alan Kitchen
 Screening donors and donations for transfusion transmissible infectious agents Alan Kitchen Aim Not to teach you microbiology To provide and awareness of the big picture To provide an understanding of
Screening donors and donations for transfusion transmissible infectious agents Alan Kitchen Aim Not to teach you microbiology To provide and awareness of the big picture To provide an understanding of
TRANSFUSION ASSOCIATED DISEASE, RECALL, OR COMPLICATION INVESTIGATION POLICY I. FATALITIES AND COMPLICATIONS ASSOCIATED WITH TRANSFUSION:
 I. FATALITIES AND COMPLICATIONS ASSOCIATED WITH TRANSFUSION: A. TRANSFUSION RELATED FATALITY: FDA and MEDIC must be notified immediately, and subsequently in writing, when a possible transfusion related
I. FATALITIES AND COMPLICATIONS ASSOCIATED WITH TRANSFUSION: A. TRANSFUSION RELATED FATALITY: FDA and MEDIC must be notified immediately, and subsequently in writing, when a possible transfusion related
Surveillance Report 2014
 Surveillance Report 2014 Executive summary We are pleased to present the third report for our stakeholders describing infectious disease surveillance. High quality and timely surveillance is key to the
Surveillance Report 2014 Executive summary We are pleased to present the third report for our stakeholders describing infectious disease surveillance. High quality and timely surveillance is key to the
Transfusion transmitted infections in National Haemovigilance Systems: the Greek experience
 Transfusion transmitted infections in National Haemovigilance Systems: the Greek experience Global blood product safety 10 April 2019, Rome, Italy Constantina Politis National Coordinating Haemovigilance
Transfusion transmitted infections in National Haemovigilance Systems: the Greek experience Global blood product safety 10 April 2019, Rome, Italy Constantina Politis National Coordinating Haemovigilance
EDUCATIONAL COMMENTARY EMERGING INFECTIOUS DISEASE AGENTS
 EDUCATIONAL COMMENTARY EMERGING INFECTIOUS DISEASE AGENTS Educational commentary is provided through our affiliation with the American Society for Clinical Pathology (ASCP). To obtain FREE CME/CMLE credits
EDUCATIONAL COMMENTARY EMERGING INFECTIOUS DISEASE AGENTS Educational commentary is provided through our affiliation with the American Society for Clinical Pathology (ASCP). To obtain FREE CME/CMLE credits
Mary Berg, M.D. Medical Director, Transfusion Services Associate Professor of Pathology University of Colorado Hospital
 Transfusion Reactions/Complications Mary Berg, M.D. Medical Director, Transfusion Services Associate Professor of Pathology University of Colorado Hospital Acute Transfusion Reactions Can be seen with
Transfusion Reactions/Complications Mary Berg, M.D. Medical Director, Transfusion Services Associate Professor of Pathology University of Colorado Hospital Acute Transfusion Reactions Can be seen with
Keeping blood transfusions safe:
 ISP-WIV Seminar Diagnosis and surveillance of infectious diseases May 18 th, 2017 Keeping blood transfusions safe: the challenge of emerging infectious diseases Presented by Roland HÜBNER Superior Health
ISP-WIV Seminar Diagnosis and surveillance of infectious diseases May 18 th, 2017 Keeping blood transfusions safe: the challenge of emerging infectious diseases Presented by Roland HÜBNER Superior Health
The testing of Donated Blood and Components at NHSBT
 The testing of Donated Blood and Components at NHSBT NHSBT is responsible for collecting all donated blood and platelets in England, then processing into components before issuing to client hospitals.
The testing of Donated Blood and Components at NHSBT NHSBT is responsible for collecting all donated blood and platelets in England, then processing into components before issuing to client hospitals.
Changing eligibility criteria for MSM: The Canadian Perspective
 Changing eligibility criteria for MSM: The Canadian Perspective Mindy Goldman MD Canadian Blood Services ASFA & WAA Joint Meeting, San Francisco April 4, 2014 Outline - Historical background - International
Changing eligibility criteria for MSM: The Canadian Perspective Mindy Goldman MD Canadian Blood Services ASFA & WAA Joint Meeting, San Francisco April 4, 2014 Outline - Historical background - International
The cost-effectiveness of NAT for HIV, HCV, and HBV in whole-blood donations Jackson B R, Busch M P, Stramer S L, AuBuchon J P
 The cost-effectiveness of NAT for HIV, HCV, and HBV in whole-blood donations Jackson B R, Busch M P, Stramer S L, AuBuchon J P Record Status This is a critical abstract of an economic evaluation that meets
The cost-effectiveness of NAT for HIV, HCV, and HBV in whole-blood donations Jackson B R, Busch M P, Stramer S L, AuBuchon J P Record Status This is a critical abstract of an economic evaluation that meets
Babesia and Blood Safety
 Babesia and Blood Safety American Red Cross, New England Region Bryan Spencer, MPH Manager of Blood Research / REDS-III Coordinator ASM 46 th Annual Region I Meeting 27 October, 2011 Randolph, MA The need
Babesia and Blood Safety American Red Cross, New England Region Bryan Spencer, MPH Manager of Blood Research / REDS-III Coordinator ASM 46 th Annual Region I Meeting 27 October, 2011 Randolph, MA The need
Assays to Address Emerging Threats to Blood Safety
 Assays to Address Emerging Threats to Blood Safety Jeffrey M. Linnen, Ph.D. Director, Product Development Gen-Probe Incorporated San Diego, CA The IPFA/PEI 17th Workshop on Surveillance and Screening of
Assays to Address Emerging Threats to Blood Safety Jeffrey M. Linnen, Ph.D. Director, Product Development Gen-Probe Incorporated San Diego, CA The IPFA/PEI 17th Workshop on Surveillance and Screening of
Optimal / Evidence-based Method to Prevent Transmission of Cytomegalovirus (CMV) by Transfusion
 ILABB Fall Meeting 2015 Optimal / Evidence-based Method to Prevent Transmission of Cytomegalovirus (CMV) by Transfusion Presenter Ronald G Strauss, MD Associate Medical Director, LifeSource/ITxM Terms
ILABB Fall Meeting 2015 Optimal / Evidence-based Method to Prevent Transmission of Cytomegalovirus (CMV) by Transfusion Presenter Ronald G Strauss, MD Associate Medical Director, LifeSource/ITxM Terms
Surveillance Report 2015
 Surveillance Report 2015 Executive summary We are pleased to present the fourth report for our stakeholders describing infectious disease surveillance. High quality and timely surveillance is key to the
Surveillance Report 2015 Executive summary We are pleased to present the fourth report for our stakeholders describing infectious disease surveillance. High quality and timely surveillance is key to the
For more information about how to cite these materials visit
 Author(s): Robertson Davenport, M.D., 2009 License: Unless otherwise noted, this material is made available under the terms of the Creative Commons Attribution Noncommercial Share Alike 3.0 License: http://creativecommons.org/licenses/by-nc-sa/3.0/
Author(s): Robertson Davenport, M.D., 2009 License: Unless otherwise noted, this material is made available under the terms of the Creative Commons Attribution Noncommercial Share Alike 3.0 License: http://creativecommons.org/licenses/by-nc-sa/3.0/
Pathogen Reduction/Inactivation KABB Annual Meeting Elpidio Pena, MD, MA Norton Healthcare Transfusion Services
 Pathogen Reduction/Inactivation KABB Annual Meeting 2017 Elpidio Pena, MD, MA Norton Healthcare Transfusion Services Goals Discuss the approved methods for pathogen reduction/inactivation of blood products
Pathogen Reduction/Inactivation KABB Annual Meeting 2017 Elpidio Pena, MD, MA Norton Healthcare Transfusion Services Goals Discuss the approved methods for pathogen reduction/inactivation of blood products
Update on Transfusion- Transmitted Infectious Diseases
 Update on Transfusion- Transmitted Infectious Diseases Scott Koepsell MD PhD Associate Professor University of Nebraska Medical Center I have no conflicts of interest to disclose. Global changes and the
Update on Transfusion- Transmitted Infectious Diseases Scott Koepsell MD PhD Associate Professor University of Nebraska Medical Center I have no conflicts of interest to disclose. Global changes and the
Surveillance Report 2016
 Surveillance Report 2016 Executive summary We are pleased to present the fifth report for our stakeholders describing infectious disease surveillance. High quality and timely surveillance is key to the
Surveillance Report 2016 Executive summary We are pleased to present the fifth report for our stakeholders describing infectious disease surveillance. High quality and timely surveillance is key to the
Anticipating (re)emerging infections to ensure blood safety
 Anticipating (re)emerging infections to ensure blood safety Pierre Tiberghien Etablissement Français du Sang, St-Denis and Université de Franche-Comté, Besançon, France IPFA/PEI 23rd International Workshop
Anticipating (re)emerging infections to ensure blood safety Pierre Tiberghien Etablissement Français du Sang, St-Denis and Université de Franche-Comté, Besançon, France IPFA/PEI 23rd International Workshop
Khalid Alquthami (Correspondence) Regional Lab, Makkah. Saudi Arabia
 1 Regional Lab, Makkah. Saudi Arabia stract: Clinical specificity and genotype/subtype detection of viruses using the Cobas TaqScreen MPX system V 2.0, which is a nucleic acid test (NAT) that uses multiples
1 Regional Lab, Makkah. Saudi Arabia stract: Clinical specificity and genotype/subtype detection of viruses using the Cobas TaqScreen MPX system V 2.0, which is a nucleic acid test (NAT) that uses multiples
Source of platelet concentrates FACHBEREICH MEDIZIN
 FACHBEREICH MEDIZIN Efficacy and safety of platelet concentrates from platelet apheresis donations or whole blood donations Gregor Bein Institute for Clinical Immunology and Transfusion Medicine Justus-Liebig-University
FACHBEREICH MEDIZIN Efficacy and safety of platelet concentrates from platelet apheresis donations or whole blood donations Gregor Bein Institute for Clinical Immunology and Transfusion Medicine Justus-Liebig-University
New Advances in Transfusion EM I LY CO BERLY, M D
 New Advances in Transfusion EM I LY CO BERLY, M D TRANSFUSI ON M EDI CI NE FELLO W VANDERBI LT UNI VERSITY Objectives To discuss the terminology, components, transfusion risks, and dosing guidelines for
New Advances in Transfusion EM I LY CO BERLY, M D TRANSFUSI ON M EDI CI NE FELLO W VANDERBI LT UNI VERSITY Objectives To discuss the terminology, components, transfusion risks, and dosing guidelines for
Transfusion therapy: what is new with respect to safety and control of iron loading? Yesim AYDINOK
 Transfusion therapy: what is new with respect to safety and control of iron loading? Yesim AYDINOK Following the Policy of the National Regulation 3.3, page 17, on CME disclosures, dated 5 November 2009,
Transfusion therapy: what is new with respect to safety and control of iron loading? Yesim AYDINOK Following the Policy of the National Regulation 3.3, page 17, on CME disclosures, dated 5 November 2009,
Interventions for Babesia (and Plasmodium)
 Interventions for Babesia (and Plasmodium) Susan L. Stramer, Ph.D. May 16 2017 www.aabb.org Babesiosis Malaria-like illness caused by Babesia spp. Asymptomatic fatal Non-specific symptoms (malaise, fever,
Interventions for Babesia (and Plasmodium) Susan L. Stramer, Ph.D. May 16 2017 www.aabb.org Babesiosis Malaria-like illness caused by Babesia spp. Asymptomatic fatal Non-specific symptoms (malaise, fever,
The Alphabet Soup of Viral Hepatitis Testing
 The Alphabet Soup of Viral Hepatitis Testing August 18, 2011 Patricia Slev, PhD, DABCC Medical Director, Serologic Hepatitis and Retrovirus Laboratory, ARUP Laboratories Assistant Professor of Pathology,
The Alphabet Soup of Viral Hepatitis Testing August 18, 2011 Patricia Slev, PhD, DABCC Medical Director, Serologic Hepatitis and Retrovirus Laboratory, ARUP Laboratories Assistant Professor of Pathology,
Non-reproductive tissues and cells
 Colour key Tested pathogen VIRAL Minimum requirements as set out in Directive 2004/23/EC More stringent testing - legy binding on national level More stringent testing - recommended on national level Not
Colour key Tested pathogen VIRAL Minimum requirements as set out in Directive 2004/23/EC More stringent testing - legy binding on national level More stringent testing - recommended on national level Not
Trends in molecular diagnostics
 Trends in molecular diagnostics Detection of target genes of interest Quantification Infectious diseases HIV Hepatitis C & B TB / MAC Cytomegalovirus Herpes simplex Varicella zoster CT/GC HPV Profiling
Trends in molecular diagnostics Detection of target genes of interest Quantification Infectious diseases HIV Hepatitis C & B TB / MAC Cytomegalovirus Herpes simplex Varicella zoster CT/GC HPV Profiling
Babesia spp. Emerging Transfusion Dilemmas
 Babesia spp. Emerging Transfusion Dilemmas David A. Leiby, PhD Transmissible Diseases Department American Red Cross Holland Laboratory Department of Microbiology and Tropical Medicine George Washington
Babesia spp. Emerging Transfusion Dilemmas David A. Leiby, PhD Transmissible Diseases Department American Red Cross Holland Laboratory Department of Microbiology and Tropical Medicine George Washington
Impact of Testing Strategies to Reduce Transmission Risk for HBV. Ravi Reddy, M Vermeulen South African National Blood Service (SANBS) 29 July 2013
 Impact of Testing Strategies to Reduce Transmission Risk for HBV Ravi Reddy, M Vermeulen South African National Blood Service (SANBS) 29 July 2013 Overview of SANBS SANBS is a private not for profit company
Impact of Testing Strategies to Reduce Transmission Risk for HBV Ravi Reddy, M Vermeulen South African National Blood Service (SANBS) 29 July 2013 Overview of SANBS SANBS is a private not for profit company
Emerging Pathogens that Impact the Canadian Blood Supply Alberta Vein to Vein Conference March 18-19, 2016
 Emerging Pathogens that Impact the Canadian Blood Supply Alberta Vein to Vein Conference March 18-19, 2016 Dr. Margaret Fearon Medical Director, Medical Microbiology, Canadian Blood Services March 18,
Emerging Pathogens that Impact the Canadian Blood Supply Alberta Vein to Vein Conference March 18-19, 2016 Dr. Margaret Fearon Medical Director, Medical Microbiology, Canadian Blood Services March 18,
Non-reproductive tissues and cells
 Colour key Minimum requirements as set out in Directive 2004/23/EC and its technical Directives (particularly 2006/17/EC) More stringent -legally binding, applies for all donations and all donor profiles
Colour key Minimum requirements as set out in Directive 2004/23/EC and its technical Directives (particularly 2006/17/EC) More stringent -legally binding, applies for all donations and all donor profiles
Non-reproductive tissues and cells
 Ministry of Health: Institute for Transplantation and Biomedicine / Colour key VIRAL HIV 1 and HIV 2 Hepatitis B Minimum requirements as set out in Directive 2004/23/EC More stringent - legy binding More
Ministry of Health: Institute for Transplantation and Biomedicine / Colour key VIRAL HIV 1 and HIV 2 Hepatitis B Minimum requirements as set out in Directive 2004/23/EC More stringent - legy binding More
Reducing Babesia and Malaria Risks: Testing, Donor Selection, Inactivation. Susan L. Stramer PhD IPFA 23 rd Meeting May
 Reducing Babesia and Malaria Risks: Testing, Donor Selection, Inactivation Susan L. Stramer PhD IPFA 23 rd Meeting May 26 2016 Babesiosis Malaria-like illness caused by babesia spp. Asymptomatic fatal
Reducing Babesia and Malaria Risks: Testing, Donor Selection, Inactivation Susan L. Stramer PhD IPFA 23 rd Meeting May 26 2016 Babesiosis Malaria-like illness caused by babesia spp. Asymptomatic fatal
Reporting from Council of Europe member states on the collection, testing and use of blood and blood components in Europe The 2006 Survey
 Reporting from Council of Europe member states on the collection, testing and use of blood and blood components in Europe The 2006 Survey This questionnaire consists of three sections: A. Collection and
Reporting from Council of Europe member states on the collection, testing and use of blood and blood components in Europe The 2006 Survey This questionnaire consists of three sections: A. Collection and
Infectious Disease Diagnostics
 Infectious Disease Diagnostics Tackling Infectious Diseases Uppsala Health Summit - October10 th, 2017 Stephan Jäger, Roche Diagnostics GmbH When to tackle infectious diseases Prevention & action contribution
Infectious Disease Diagnostics Tackling Infectious Diseases Uppsala Health Summit - October10 th, 2017 Stephan Jäger, Roche Diagnostics GmbH When to tackle infectious diseases Prevention & action contribution
Viral Hepatitis Diagnosis and Management
 Viral Hepatitis Diagnosis and Management CLINICAL BACKGROUND Viral hepatitis is a relatively common disease (25 per 100,000 individuals in the United States) caused by a diverse group of hepatotropic agents
Viral Hepatitis Diagnosis and Management CLINICAL BACKGROUND Viral hepatitis is a relatively common disease (25 per 100,000 individuals in the United States) caused by a diverse group of hepatotropic agents
Transfusion Medicine (Comprehensive, Limited) J, J1
 www.cap.org Transfusion Medicine Analytes/procedures in bold type are regulated for proficiency testing by the Centers for Medicare & Medicaid Services (CMS). Transfusion Medicine (Comprehensive, Limited)
www.cap.org Transfusion Medicine Analytes/procedures in bold type are regulated for proficiency testing by the Centers for Medicare & Medicaid Services (CMS). Transfusion Medicine (Comprehensive, Limited)
Donation Criteria for Men who have Sex with Men (MSM): Update from Canadian Blood Services
 Donation Criteria for Men who have Sex with Men (MSM): Update from Canadian Blood Services Outline 1 Current criteria Proposed criteria Stakeholder outreach Outline 2 Safety Assessment Results since implementation
Donation Criteria for Men who have Sex with Men (MSM): Update from Canadian Blood Services Outline 1 Current criteria Proposed criteria Stakeholder outreach Outline 2 Safety Assessment Results since implementation
Risk-Reduction Strategies for Platelet Transfusion in the United States
 Review TheScientificWorldJOURNAL (2011) 11, 624 640 ISSN 1537-744X; DOI 10.1100/tsw.2011.60 Risk-Reduction Strategies for Platelet Transfusion in the United States Eleftherios C. Vamvakas Department of
Review TheScientificWorldJOURNAL (2011) 11, 624 640 ISSN 1537-744X; DOI 10.1100/tsw.2011.60 Risk-Reduction Strategies for Platelet Transfusion in the United States Eleftherios C. Vamvakas Department of
2014/LSIF/PD/025 Malaysia s Approach to Testing Strategies
 2014/LSIF/PD/025 Malaysia s Approach to Testing Strategies Submitted by: Malaysia Policy Dialogue and Workshop on Attaining a Safe and Sustainable Blood Supply Chain Manila, Philippines 30 September 1
2014/LSIF/PD/025 Malaysia s Approach to Testing Strategies Submitted by: Malaysia Policy Dialogue and Workshop on Attaining a Safe and Sustainable Blood Supply Chain Manila, Philippines 30 September 1
2017 CST-Astellas Canadian Transplant Fellows Symposium. Optimizing use of organs from Increased Risk Donors
 2017 CST-Astellas Canadian Transplant Fellows Symposium Optimizing use of organs from Increased Risk Donors Atual Humar, MD Atul Humar is a Professor in the Department of Medicine, University of Toronto.
2017 CST-Astellas Canadian Transplant Fellows Symposium Optimizing use of organs from Increased Risk Donors Atual Humar, MD Atul Humar is a Professor in the Department of Medicine, University of Toronto.
Guide to the preparation, use and quality assurance of blood components
 Contents Foreword...3 Members of the European Committee (Partial Agreement) on Blood Transfusion... 8 Members of the GTS working group... 22 Members of the TS066 working group... 30 Recommendation No.
Contents Foreword...3 Members of the European Committee (Partial Agreement) on Blood Transfusion... 8 Members of the GTS working group... 22 Members of the TS066 working group... 30 Recommendation No.
Diagnostic Methods of HBV infection. Zohreh Sharifi,ph.D of Virology Research center, Iranian Blood Transfusion Organization (IBTO)
 Diagnostic Methods of HBV infection Zohreh Sharifi,ph.D of Virology Research center, Iranian Blood Transfusion Organization (IBTO) Hepatitis B-laboratory diagnosis Detection of HBV infection involves
Diagnostic Methods of HBV infection Zohreh Sharifi,ph.D of Virology Research center, Iranian Blood Transfusion Organization (IBTO) Hepatitis B-laboratory diagnosis Detection of HBV infection involves
Non-reproductive tissues and cells Recommending authority/ association
 Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent testing - legy binding on national level More stringent testing - recomd on national level Not legy binding and not recomd
Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent testing - legy binding on national level More stringent testing - recomd on national level Not legy binding and not recomd
HEV Assay Development Update
 HEV Assay Development Update Jeffrey M. Linnen, Ph.D. Senior Director, Research & Development Hologic Gen-Probe, San Diego, California USA IPFA/PEI 20th International Workshop on Surveillance and Screening
HEV Assay Development Update Jeffrey M. Linnen, Ph.D. Senior Director, Research & Development Hologic Gen-Probe, San Diego, California USA IPFA/PEI 20th International Workshop on Surveillance and Screening
Impact of donor epidemiology and screening strategies on the safety of blood and plasma for fractionation
 IPFA 2 nd Asia Workshop in Yogyakarta Impact of donor epidemiology and screening strategies on the safety of blood and plasma for fractionation Yoshihiko Tani, MD, PhD Japanese Red Cross Osaka Blood Center
IPFA 2 nd Asia Workshop in Yogyakarta Impact of donor epidemiology and screening strategies on the safety of blood and plasma for fractionation Yoshihiko Tani, MD, PhD Japanese Red Cross Osaka Blood Center
Non-reproductive tissues and cells Recommending authority/ association
 Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent - legy binding More stringent - recommended Not legy binding and not recommended Tested pathogen Donor test/ technique
Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent - legy binding More stringent - recommended Not legy binding and not recommended Tested pathogen Donor test/ technique
Complicated viral infections
 Complicated viral infections Clinical case discussion Diagnostic dilemmas NSW State Reference Laboratory for HIV St Vincent s Hospital Sydney Diagnostic dilemmas Indeterminate or discordant serology (western
Complicated viral infections Clinical case discussion Diagnostic dilemmas NSW State Reference Laboratory for HIV St Vincent s Hospital Sydney Diagnostic dilemmas Indeterminate or discordant serology (western
A. Anti-HIV-1/2, HIV NAT Lookback: for those identified blood and blood components collected:
 PURPOSE: MATERIALS: To outline a procedure for removing, recalling and/or correcting blood products that do not meet the requirements of the FDA, AABB, or the blood center. Recall/Market Withdrawal Form
PURPOSE: MATERIALS: To outline a procedure for removing, recalling and/or correcting blood products that do not meet the requirements of the FDA, AABB, or the blood center. Recall/Market Withdrawal Form
Non-competitive Internal Control Concept for PCR-based Qualitative Assays
 Christian O. Simon Roche Molecular Diagnostics Rotkreuz, CH Non-competitive Internal Control Concept for PCR-based Qualitative Assays 23 rd SoGAT Meeting, 16 th 17 th April, 2012, Vilnius, Lithuania Design
Christian O. Simon Roche Molecular Diagnostics Rotkreuz, CH Non-competitive Internal Control Concept for PCR-based Qualitative Assays 23 rd SoGAT Meeting, 16 th 17 th April, 2012, Vilnius, Lithuania Design
WHO Parvovirus B19 Genotype Panel
 WHO Parvovirus B19 Genotype Panel Mei-ying W Yu, PhD SoGAT XXII Rome, 14-15 April 2011 1 st WHO International Reference Panel for Parvovirus B19 Genotypes In Oct 2009, a plasma-derived parvovirus B19 (B19V)
WHO Parvovirus B19 Genotype Panel Mei-ying W Yu, PhD SoGAT XXII Rome, 14-15 April 2011 1 st WHO International Reference Panel for Parvovirus B19 Genotypes In Oct 2009, a plasma-derived parvovirus B19 (B19V)
Table of Contents (continued)
 Emerging Molecular and Immunohematology Blood Typing, Grouping And Infectious Disease NAT Screening Assays And Companies Developing New Technologies and Products Table of Contents 1. Blood Typing and Grouping
Emerging Molecular and Immunohematology Blood Typing, Grouping And Infectious Disease NAT Screening Assays And Companies Developing New Technologies and Products Table of Contents 1. Blood Typing and Grouping
Answering your daily challenges. in the ELISA technology I N FECTI OUS DISEASES. bioelisa
 Answering your daily challenges in the ELISA technology I N FECTI OUS DISEASES bioelisa RETROVIRUS HIV is the cause of the most important global pandemic. Due to the lack of vaccination, early diagnosis
Answering your daily challenges in the ELISA technology I N FECTI OUS DISEASES bioelisa RETROVIRUS HIV is the cause of the most important global pandemic. Due to the lack of vaccination, early diagnosis
MSM DEFERRAL POLICY ISSUE DOCUMENT: MSM DEFERRAL ISSUE BACKGROUND
 MSM DEFERRAL POLICY ISSUE DOCUMENT: MSM DEFERRAL ISSUE Countries around the world are reviewing the policy of deferring from blood donation men who have had sex with men (MSM). Both Canadian Blood Services
MSM DEFERRAL POLICY ISSUE DOCUMENT: MSM DEFERRAL ISSUE Countries around the world are reviewing the policy of deferring from blood donation men who have had sex with men (MSM). Both Canadian Blood Services
Situation of XMRV and Blood Transfusion. Celso Bianco, MD ISBT Working Party on TTID Lisbon, June 19, 2011
 Situation of XMRV and Blood Transfusion Celso Bianco, MD ISBT Working Party on TTID Lisbon, June 19, 2011 Lombardi et al. Science 326, 585 (2009) Conclusions: CFS and XMRV XMRV found in 67% of CFS patients
Situation of XMRV and Blood Transfusion Celso Bianco, MD ISBT Working Party on TTID Lisbon, June 19, 2011 Lombardi et al. Science 326, 585 (2009) Conclusions: CFS and XMRV XMRV found in 67% of CFS patients
Impact of multi-dye multiplex technology on testing algorithm
 Impact of multi-dye multiplex technology on testing algorithm Lydia Blanco. Mª Isabel Gonzalez-Fraile Centro de Hemoterapia y Hemodonación de Castilla y León, España TTI EPIDEMIOLOGICAL DATA IN SPAIN NAT
Impact of multi-dye multiplex technology on testing algorithm Lydia Blanco. Mª Isabel Gonzalez-Fraile Centro de Hemoterapia y Hemodonación de Castilla y León, España TTI EPIDEMIOLOGICAL DATA IN SPAIN NAT
Hepa%%s E Virus Is it a Concern?
 IPFA/PEI 17 th Workshop on Surveillance and Screening of Blood Borne Pathogens 26-27 May 2010, The Regent Esplanade Zagreb Hotel, Zagreb, Croa%a Hepa%%s E Virus Is it a Concern? Keiji Matsubayashi Hokkaido
IPFA/PEI 17 th Workshop on Surveillance and Screening of Blood Borne Pathogens 26-27 May 2010, The Regent Esplanade Zagreb Hotel, Zagreb, Croa%a Hepa%%s E Virus Is it a Concern? Keiji Matsubayashi Hokkaido
Chapter 17: Transfusion-Transmitted Infections (TTI)
 Annual SHOT Report 2014 Supplementary Information Chapter 17: Transfusion-Transmitted s (TTI) The table below is an excerpt from the full Table 17.2 which can be viewed in the main report. Case reports
Annual SHOT Report 2014 Supplementary Information Chapter 17: Transfusion-Transmitted s (TTI) The table below is an excerpt from the full Table 17.2 which can be viewed in the main report. Case reports
PATHOGEN INACTIVATION:
 PATHOGEN INACTIVATION: She s worth it. Be SURE. With INTERCEPT you know that you are doing everything in your power to deliver safe and effective blood products to patients. Transfusion-transmitted infection
PATHOGEN INACTIVATION: She s worth it. Be SURE. With INTERCEPT you know that you are doing everything in your power to deliver safe and effective blood products to patients. Transfusion-transmitted infection
Blood Transfusion. What is blood transfusion? What are blood banks? When is a blood transfusion needed? Who can donate blood?
 What is blood transfusion? A blood transfusion is a safe, common procedure in which blood is given through an intravenous (IV) line in one of the blood vessels. A blood transfusion usually takes two to
What is blood transfusion? A blood transfusion is a safe, common procedure in which blood is given through an intravenous (IV) line in one of the blood vessels. A blood transfusion usually takes two to
Frequency of occult hepatitis B in HBsAg seronegative blood donors in a tertiary care hospital in kerala,south India.
 Frequency of occult hepatitis B in HBsAg seronegative blood donors in a tertiary care hospital in kerala,south India. Cinzia Keechilot, Veena Shenoy 1,V Anil kumar 2,Lalita Biswas 3. MBBS student * Transfusion
Frequency of occult hepatitis B in HBsAg seronegative blood donors in a tertiary care hospital in kerala,south India. Cinzia Keechilot, Veena Shenoy 1,V Anil kumar 2,Lalita Biswas 3. MBBS student * Transfusion
REASONS FOR MOVING TOWARD A PATIENT-CENTRIC PARADIGM OF CLINICAL TRANSFUSION MEDICINE PRACTICE
 REASONS FOR MOVING TOWARD A PATIENT-CENTRIC PARADIGM OF CLINICAL TRANSFUSION MEDICINE PRACTICE Eleftherios C. Vamvakas Swisstransfusion 2012 Congress of the Swiss Association for Transfusion Medicine Basel,
REASONS FOR MOVING TOWARD A PATIENT-CENTRIC PARADIGM OF CLINICAL TRANSFUSION MEDICINE PRACTICE Eleftherios C. Vamvakas Swisstransfusion 2012 Congress of the Swiss Association for Transfusion Medicine Basel,
Center for Biologics Evaluation and Research
 Center for Biologics Evaluation and Research Current Activities Future Directions Washington, DC January 25, 2010 Karen Midthun, MD Acting Director, CBER CBER Our Mission To ensure the safety, purity,
Center for Biologics Evaluation and Research Current Activities Future Directions Washington, DC January 25, 2010 Karen Midthun, MD Acting Director, CBER CBER Our Mission To ensure the safety, purity,
TRANSFUSION REACTIONS
 14 TRANSFUSION REACTIONS 14.1 INTRODUCTION Transfusion of blood and blood products are reported to cause reactions during or after procedure specially in patients who receive multiple transfusions. These
14 TRANSFUSION REACTIONS 14.1 INTRODUCTION Transfusion of blood and blood products are reported to cause reactions during or after procedure specially in patients who receive multiple transfusions. These
NAT Screening of Blood Donations in NBC, TRCS
 IPFA/PEI, Italy NAT Screening of Blood Donations in NBC, TRCS 2000-2001 Clinical evaluation using COBAS AmpliiScreen HIV & HCV assays, (MP24) window period rate - 1 : 108,768 for HIV-1 and HCV 2002 Routine
IPFA/PEI, Italy NAT Screening of Blood Donations in NBC, TRCS 2000-2001 Clinical evaluation using COBAS AmpliiScreen HIV & HCV assays, (MP24) window period rate - 1 : 108,768 for HIV-1 and HCV 2002 Routine
Annual SHOT Report 2016 Supplementary Information. Chapter 17: Transfusion-Transmitted Infections (TTI)
 Annual SHOT Report 2016 Supplementary Information Chapter 17: Transfusion-Transmitted s (TTI) The table below is an excerpt from the full Table 17.3 which can be viewed in the main report. Case reports
Annual SHOT Report 2016 Supplementary Information Chapter 17: Transfusion-Transmitted s (TTI) The table below is an excerpt from the full Table 17.3 which can be viewed in the main report. Case reports
Non-reproductive tissues and cells Recommending authority/ association
 Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent - legally binding More stringent - recommended Not legally binding and not recommended Non-reproductive tissues and cells
Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent - legally binding More stringent - recommended Not legally binding and not recommended Non-reproductive tissues and cells
Routine Use of Mini-Pool Nucleic Acid Testing (MP-NAT) Multiplex Assay for Sero-Negative Blood Donors
 Journal of the Egyptian Society of Haematology & Research, Vol. 7, No. 2, September: 1-5, 2011 Routine Use of Mini-Pool Nucleic Acid Testing (MP-NAT) Multiplex Assay for Sero-Negative Blood Donors HISHAM
Journal of the Egyptian Society of Haematology & Research, Vol. 7, No. 2, September: 1-5, 2011 Routine Use of Mini-Pool Nucleic Acid Testing (MP-NAT) Multiplex Assay for Sero-Negative Blood Donors HISHAM
Traditional laboratory-based screening methods
 DONOR INFECTIOUS DISEASE TESTING Description of 15 DNA-positive and antibody-negative window-period blood donations identified during prospective screening for Babesia microti Erin D. Moritz, 1 Laura Tonnetti,
DONOR INFECTIOUS DISEASE TESTING Description of 15 DNA-positive and antibody-negative window-period blood donations identified during prospective screening for Babesia microti Erin D. Moritz, 1 Laura Tonnetti,
Guidance for Industry
 Guidance for Industry Use of Nucleic Acid Tests on Pooled and Individual Samples from Donors of Whole Blood and Blood Components (including Source Plasma and Source Leukocytes) to Adequately and Appropriately
Guidance for Industry Use of Nucleic Acid Tests on Pooled and Individual Samples from Donors of Whole Blood and Blood Components (including Source Plasma and Source Leukocytes) to Adequately and Appropriately
Seroprevalence of Babesia microti in Individuals with Lyme Disease. Sabino R. Curcio, M.S, MLS(ASCP)
 Seroprevalence of Babesia microti in Individuals with Lyme Disease Sabino R. Curcio, M.S, MLS(ASCP) Lyme Disease Most common vectorborne illness in the United States Caused by the tick-transmitted spirochete
Seroprevalence of Babesia microti in Individuals with Lyme Disease Sabino R. Curcio, M.S, MLS(ASCP) Lyme Disease Most common vectorborne illness in the United States Caused by the tick-transmitted spirochete
Transfusion-transmitted Cytomegalovirus
 Transfusion-transmitted Cytomegalovirus Can you confidently abandon CMV seronegative products in the modern era of pre-storage leukoreduction? Jeannie Callum, BA, MD, FRCPC Really? Are we still talking
Transfusion-transmitted Cytomegalovirus Can you confidently abandon CMV seronegative products in the modern era of pre-storage leukoreduction? Jeannie Callum, BA, MD, FRCPC Really? Are we still talking
Transfusion Medicine Potpourri. BUMC - Phoenix Internal Medicine Residents September 29, 2015
 Transfusion Medicine Potpourri BUMC - Phoenix Internal Medicine Residents September 29, 2015 Clinical case A 24 year old female with sickle cell anemia has just moved to the area and presents as a new
Transfusion Medicine Potpourri BUMC - Phoenix Internal Medicine Residents September 29, 2015 Clinical case A 24 year old female with sickle cell anemia has just moved to the area and presents as a new
Thinking Twice About Transfusions: When TACOS and TRALIs Turn Treatment Into Tragedy. Megan Boysen Osborn, MD, MHPE
 Thinking Twice About Transfusions: When TACOS and TRALIs Turn Treatment Into Tragedy Megan Boysen Osborn, MD, MHPE Let s TACO bout Transfusion Reactions Megan Boysen Osborn, MD, MHPE Let s TACO bout Transfusion
Thinking Twice About Transfusions: When TACOS and TRALIs Turn Treatment Into Tragedy Megan Boysen Osborn, MD, MHPE Let s TACO bout Transfusion Reactions Megan Boysen Osborn, MD, MHPE Let s TACO bout Transfusion
Diagnostic Methods of HBV and HDV infections
 Diagnostic Methods of HBV and HDV infections Zohreh Sharifi,ph.D Blood Transfusion Research Center, High Institute for Research and Education in Transfusion Medicine Hepatitis B-laboratory diagnosis Detection
Diagnostic Methods of HBV and HDV infections Zohreh Sharifi,ph.D Blood Transfusion Research Center, High Institute for Research and Education in Transfusion Medicine Hepatitis B-laboratory diagnosis Detection
Learning Objectives: Hepatitis Update. Primary Causes of Chronic Liver Disease in the U.S. Hepatitis Definition. Hepatitis Viruses.
 Learning Objectives: Hepatitis Update ASCLS-Michigan March 31, 2016 Dr. Kathleen Hoag Upon attendance of this seminar and review of material provided, the attendees will be able to: 1. List hepatitis viruses
Learning Objectives: Hepatitis Update ASCLS-Michigan March 31, 2016 Dr. Kathleen Hoag Upon attendance of this seminar and review of material provided, the attendees will be able to: 1. List hepatitis viruses
HIV Update in Laboratory Testing. Patricia Slev, PhD, D(ABCC)
 HIV Update in Laboratory Testing Patricia Slev, PhD, D(ABCC) Objectives Explain the advances in HIV diagnostics, including fourth generation Ag/Ab combination HIV screening assays Describe the new CDC
HIV Update in Laboratory Testing Patricia Slev, PhD, D(ABCC) Objectives Explain the advances in HIV diagnostics, including fourth generation Ag/Ab combination HIV screening assays Describe the new CDC
Hira L. Nakhasi, Ph.D. CBER/USFDA July 3 rd 2015
 Strategies for Implementation of Antibody and Nucleic Acid-based Testing for Babesia microti in Blood Donations: Summary of May 13 th 2015 Blood Product Advisory Committee Meeting Hira L. Nakhasi, Ph.D.
Strategies for Implementation of Antibody and Nucleic Acid-based Testing for Babesia microti in Blood Donations: Summary of May 13 th 2015 Blood Product Advisory Committee Meeting Hira L. Nakhasi, Ph.D.
HIV-1 Subtypes: An Overview. Anna Maria Geretti Royal Free Hospital
 HIV-1 Subtypes: An Overview Anna Maria Geretti Royal Free Hospital Group M Subtypes A (1, 2, 3) B C D F (1, 2) G H J K Mechanisms of HIV-1 genetic diversification Point mutations RT error rate: ~1 per
HIV-1 Subtypes: An Overview Anna Maria Geretti Royal Free Hospital Group M Subtypes A (1, 2, 3) B C D F (1, 2) G H J K Mechanisms of HIV-1 genetic diversification Point mutations RT error rate: ~1 per
APPROACH TO RISK MANAGEMENT IN MEDICAL PRACTICE: STANDPOINT OF THE BLOOD TRASFUSION*
 11 APPROACH TO RISK MANAGEMENT IN MEDICAL PRACTICE: STANDPOINT OF THE BLOOD TRASFUSION* Hisami IKEDA** Asian Med. J. 44(1): 11 21, 2000 Abstract: In the field of blood transfusion, risk management is defined
11 APPROACH TO RISK MANAGEMENT IN MEDICAL PRACTICE: STANDPOINT OF THE BLOOD TRASFUSION* Hisami IKEDA** Asian Med. J. 44(1): 11 21, 2000 Abstract: In the field of blood transfusion, risk management is defined
ISBT WP-TTID Annual Report for Subgroup on Virology Drs. Michael Busch, Kurt Roth and Susan Stramer
 ISBT WP-TTID Annual Report for Subgroup on Virology Drs. Michael Busch, Kurt Roth and Susan Stramer Questionnaire on NAT Screening of Blood Donations for an International Forum on 10 years of NAT Screening
ISBT WP-TTID Annual Report for Subgroup on Virology Drs. Michael Busch, Kurt Roth and Susan Stramer Questionnaire on NAT Screening of Blood Donations for an International Forum on 10 years of NAT Screening
Non-reproductive tissues and cells
 Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent testing - legally binding on national level More stringent testing - recommended on national level Not legally binding
Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent testing - legally binding on national level More stringent testing - recommended on national level Not legally binding
Increased Risk Donors. November 1, 2018 BC Kidney Days. Jagbir Gill MD MPH Associate Professor of Medicine UBC
 Increased Risk Donors November 1, 2018 BC Kidney Days Jagbir Gill MD MPH Associate Professor of Medicine UBC Median dialysis exposure prior to DDKT Case scenarios Which donors should we accept kidneys
Increased Risk Donors November 1, 2018 BC Kidney Days Jagbir Gill MD MPH Associate Professor of Medicine UBC Median dialysis exposure prior to DDKT Case scenarios Which donors should we accept kidneys
Non-reproductive tissues and cells
 Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent - legy binding on national level More stringent - recommended on national level Not legy binding and not recommended on
Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent - legy binding on national level More stringent - recommended on national level Not legy binding and not recommended on
A Transfusion Reaction What Do I Do Now? Judith A. Sullivan, MS, MT(ASCP)SBB, CQA(ASQ) ASCLS Region III Triennial Meeting Birmingham AL
 A Transfusion Reaction What Do I Do Now? Judith A. Sullivan, MS, MT(ASCP)SBB, CQA(ASQ) ASCLS Region III Triennial Meeting Birmingham AL This promotional educational activity is brought to you by Ortho
A Transfusion Reaction What Do I Do Now? Judith A. Sullivan, MS, MT(ASCP)SBB, CQA(ASQ) ASCLS Region III Triennial Meeting Birmingham AL This promotional educational activity is brought to you by Ortho
Boot Camp Transfusion Reactions
 Boot Camp Transfusion Reactions Dr. Kristine Roland Regional Medical Lead for Transfusion Medicine, VCH Objectives By the end of this session, you should be able to: Describe in common language the potential
Boot Camp Transfusion Reactions Dr. Kristine Roland Regional Medical Lead for Transfusion Medicine, VCH Objectives By the end of this session, you should be able to: Describe in common language the potential
Update on THERAFLEX UV-Platelets: a novel system for pathogen reduction of platelets
 Update on THERAFLEX UV-Platelets: a novel system for pathogen reduction of platelets Axel Seltsam, MD, MHBA German Red Cross Blood Service NSTOB, Springe Fribourg 2011 THERAFLEX UV-Platelets Efficient
Update on THERAFLEX UV-Platelets: a novel system for pathogen reduction of platelets Axel Seltsam, MD, MHBA German Red Cross Blood Service NSTOB, Springe Fribourg 2011 THERAFLEX UV-Platelets Efficient
Robert G. Gish MD UC San Diego
 Robert G. Gish MD UC San Diego The Pipeline of HIV-Related Point-Of- Care Tests is Growing Viral detection Examples: 2012 2013 2014 2015 2016 2017 CD4 test Examples: 2009 2010 2011 2012 2013 0 2014 Evidence
Robert G. Gish MD UC San Diego The Pipeline of HIV-Related Point-Of- Care Tests is Growing Viral detection Examples: 2012 2013 2014 2015 2016 2017 CD4 test Examples: 2009 2010 2011 2012 2013 0 2014 Evidence
Confirmatory Testing Algorithm. Routine Donor Testing Algorithm
 Confirmatory Testing Algorithm Blood Confirmatory ABO/Rh Beckman Coulter PK-ABO/Rh Immucor NEO ABO/Rh Immucor NEO ABO/Rh Immucor Manual ABO/Rh Typing Immucor Manual ABO/Rh Typing Discrepancy Resolution
Confirmatory Testing Algorithm Blood Confirmatory ABO/Rh Beckman Coulter PK-ABO/Rh Immucor NEO ABO/Rh Immucor NEO ABO/Rh Immucor Manual ABO/Rh Typing Immucor Manual ABO/Rh Typing Discrepancy Resolution
Armed Services Blood Program
 Armed Services Blood Program Defense Health Board Concerns Regarding the Collection and Transfusion of Non-FDA Compliant Blood Products in Theater Information Brief Defense Health Board 17 August 2009
Armed Services Blood Program Defense Health Board Concerns Regarding the Collection and Transfusion of Non-FDA Compliant Blood Products in Theater Information Brief Defense Health Board 17 August 2009
Donor Screening in The Region. Vincini GA NRL, Melbourne, Australia
 Donor Screening in The Region Vincini GA NRL, Melbourne, Australia NRL Established in 1985 Not-for-profit organisation that exists to support laboratories that perform testing for the diagnosis and management
Donor Screening in The Region Vincini GA NRL, Melbourne, Australia NRL Established in 1985 Not-for-profit organisation that exists to support laboratories that perform testing for the diagnosis and management
Panther has new prey
 Raising the Bar for Performance Testing Panther has new prey The Aptima HIV-1 Quant Dx assay leads the hunt for HIV-1 diagnosis and viral load monitoring. Freedom to work the way you choose Run what assays
Raising the Bar for Performance Testing Panther has new prey The Aptima HIV-1 Quant Dx assay leads the hunt for HIV-1 diagnosis and viral load monitoring. Freedom to work the way you choose Run what assays
JOURNAL CLUB INDICATIONS FOR AND ADVERSE EFFECTS OF RED CELL TRANSFUSION. Maggie Woods PGY-3
 JOURNAL CLUB INDICATIONS FOR AND ADVERSE EFFECTS OF RED CELL TRANSFUSION Maggie Woods PGY-3 BACKGROUND Objective: To describe evidence for current guidelines, review trends, examine the risks of transfusion
JOURNAL CLUB INDICATIONS FOR AND ADVERSE EFFECTS OF RED CELL TRANSFUSION Maggie Woods PGY-3 BACKGROUND Objective: To describe evidence for current guidelines, review trends, examine the risks of transfusion
DHQ Flowcharts v2.0 eff. February 2016
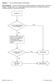 Question: 1. Are you feeling healthy and well today? Donor Eligibility: A person should be free of infectious diseases, including colds, on the day of donation. A person who is not in good health should
Question: 1. Are you feeling healthy and well today? Donor Eligibility: A person should be free of infectious diseases, including colds, on the day of donation. A person who is not in good health should
Adverse Reactions Associated with Methylene Blue Inactivated Fresh Frozen Plasma (MB FFP)
 Adverse Reactions Associated with Methylene Blue Inactivated Fresh Frozen Plasma (MB FFP) Constantina Politis Hellenic Coordinating Haemovigilance Centre (SKAE) Background Pathogen inactivation is expected
Adverse Reactions Associated with Methylene Blue Inactivated Fresh Frozen Plasma (MB FFP) Constantina Politis Hellenic Coordinating Haemovigilance Centre (SKAE) Background Pathogen inactivation is expected
HEV Update Blood Components. Dragoslav Domanović, European Centre for Disease Prevention and Control (Sweden)
 HEV Update Blood Components Dragoslav Domanović, European Centre for Disease Prevention and Control (Sweden) Outline ECDC activities started in 2015 to understand the epidemiology and burden of HEV infection
HEV Update Blood Components Dragoslav Domanović, European Centre for Disease Prevention and Control (Sweden) Outline ECDC activities started in 2015 to understand the epidemiology and burden of HEV infection
