Situation of XMRV and Blood Transfusion. Celso Bianco, MD ISBT Working Party on TTID Lisbon, June 19, 2011
|
|
|
- Brooke Moody
- 6 years ago
- Views:
Transcription
1 Situation of XMRV and Blood Transfusion Celso Bianco, MD ISBT Working Party on TTID Lisbon, June 19, 2011
2 Lombardi et al. Science 326, 585 (2009)
3 Conclusions: CFS and XMRV XMRV found in 67% of CFS patients An immune response to the virus was detected in some CFS patients Data suggest that the human population is at risk from infection with XMRV (3.7% of controls are DNA positive) Given that infectious virus is present in plasma and in blood cells, blood-borne transmission is a possibility. Coffin and Stoye. Science
4 Common Names Chronic Fatigue Syndrome (CFS, U.S. Health and Human Services Committee) Myalgic encephalomyelitis (ME, EU) Chronic fatigue and immune dysfunction syndrome (CFIDS, US Association) Post-viral fatigue syndrome Post-infectious fatigue syndrome X-associated neuroimmune disease (XAND) Worldwide prevalence 0.4-1%; US million individual
5 CFIDS Association (U.S.) Chronic Fatigue and Immune Dysfunction Syndrome CFIDS CFIDS Research 1 st Position on blood donation There are numerous medical reasons why people with CFS should not donate blood. The CFIDS Association of America has long advised against CFS patients donating blood [or organs].
6 AABB Task Force Report
7 Association Bulletin # Chronic Fatigue Syndrome & Blood Donation ( ) AABB recommends that blood collecting organizations make educational information available regarding the reasons why an individual diagnosed with CFS should not donate blood.
8 Resources from AABB AABB Bulletin, June 18, 2010 recommends educational materials discouraging individuals with CFS from donating blood Fact Sheet (March 2011) ents/xmrvfactsheet.pdf Table of Published Studies (April 27, 2011) ents/xmrvtable.pdf
9 U.S. National Heart Lung and Blood Institute (NHLBI) Scientific Working Group Analytical NAT panel development: analysis of labs with assays (CDC, FDA, NCI, WPI, BSRI) abilities to find/quantify XMRV from productive cell line in blinded positive and control panel in spiked whole blood and plasma samples Clinical NAT panel development: Plasma and whole blood panels for XMRV prevalence in 400 blood donors from BSRI (Reno), 25 XMRV pos. CFS patients from WPI, 25 controls (+ and -) each Serology validation after nucleic acid Epidemiology study design
10 Phase I Analytical Panel - Results Whole Blood Panel Plasma Panel Proviral copies/ml ,220 3,670 11,000 33,000 99,000 CDC FDA(H) FDA(L) GP NCI WPI Viral RNA copies/ml CDC FDA(H) FDA(L) GP NCI WPI ,000 10,000 50, ,000 0/3 (for dilution series) or 0/6 (for negatives) replicates called positive 1/12 negative controls called positive (panels run twice - once each by two different operators) 1/6 negative controls called positive (identified as non-specific band of human genomic origin by sequencing subsequent to decoding of results) 1/3 replicates called positive 2/3 replicates called positive 3/3 replicates called positive
11 Summary of Phase IIb NAT and Antibody Results Subject ID (description) NCI G-P CDC WPI NCI* Ab CDC Ab Subject 1 - Plasma - day 0 Negative Non reactive Negative Negative Positive Negative Subject 1 - Plasma - day 2 Negative Non reactive Negative Negative Negative Negative Subject 1 - PBMC - day 0 Negative Non reactive Negative Positive Subject 1 - PBMC - day 2 Negative Non reactive Negative Negative Subject 2 - Plasma - day 0 Negative Non reactive Negative Negative Negative Negative Subject 2 - Plasma - day 2 Negative Non reactive Negative Negative Negative Negative Subject 2 - PBMC - day 0 Negative Non reactive Negative Negative Subject 2 - PBMC - day 2 Negative Non reactive Negative Positive Subject 3 - Plasma - day 0 Negative Non reactive Negative Negative Positive Negative Subject 3 - Plasma - day 2 Negative Non reactive Negative Negative Positive Negative Subject 3 - PBMC - day 0 Negative Non reactive Negative Negative Subject 3 - PBMC - day 2 Negative Non reactive Negative Positive Subject 4 - Plasma - day 0 Negative Non reactive Negative Negative Positive Negative Subject 4 - Plasma - day 2 Negative Non reactive Negative Negative Positive Negative Subject 4 - PBMC - day 0 Negative Non reactive Negative Negative Subject 4 - PBMC - day 2 Negative Non reactive Negative Negative Pedigreed Negative - Plasma - day 0 Negative Non reactive Negative Negative Negative Negative Pedigreed Negative - Plasma - day 2 Negative Non reactive Negative Negative Positive Negative Pedigreed Negative - PBMC - day 0 Negative Non reactive Negative Positive Pedigreed Negative - PBMC - day 2 Negative Non reactive Negative Negative * Ruscetti
12 Blood XMRV Scientific Research Working Group Activities Oct Nov Dec Jan Feb Mar Apr May Jun Jul Aug Sep Oct Nov Dec Jan Presentation of findings BPAC XMRV AABB BPAC Conf.
13 Blood XMRV Scientific Research Working Group
14 Question for the Blood Products Advisory Committee of FDA (December 14, 2010): Do the scientific data support asking donors about a medical history &/or diagnosis of CFS as a basis for indefinite deferral? 9 yes, 4 no, no abstentions
15
16 - qpcr and PCR on buffy coat and whole blood - ELISA and Western blot on plasma - Virus culture CFS and 200 matched controls - 14 CFS samples from Reno study
17
18 On the origin of XMRV 1992 Patient CWR22 CaP (No DNA/RNA) CWR22 xenograft in nude mice 1992 CWR22 3 rd pass. (gdna) CWR22 7 th pass. (gdna) CWR22 unk. pass. (No DNA/RNA) prexmrv No XMRV prexmrv recombination 1996 CWR22 (total nucleic acid) 2152, 2524, 2272, Cell line 22Rv1 (gdna) 2001 Cell line CWR R1 (gdna) Late xenografts + cell lines XMRV positive Pathak et al. CROI 2011.
19 Published online 31 May 2011; /science In the issue of 23 October 2009, Science published the Report Detection of an infectious retrovirus, XMRV, in blood cell of patients with chronic fatigue syndrome, a study by Lombardi et al. purporting to show that a retrovirus called XMRV (xenotropic murine leukemia virus-related virus) was present in the blood of 67% of patients with chronic fatigue syndrome (CFS) compared with 3.7% of healthy controls (1). Since then, at least 10 studies conducted by other investigators and published elsewhere have reported a failure to detect XMRV in independent populations of CFS patients. In this week s edition of Science Express, we are publishing two Reports that strongly support the growing view that the association between XMRV and CFS described by Lombardi et al. likely reflects contamination of laboratories and research reagents with the virus. The authors of the Lombardi study believe that it is premature to conclude that the negative studies are accurate or change the conclusions of the original studies and we fully agree, said Annette Whittemore, President of the Whittemore Peterson Institute. (WPI Press release, May 31, 2011)
20 Ongoing Studies NHLBI will continue to support the XMRV blood study. Results are expected in the Fall of 2011., The Lipkin laboratory-based study is designed to rigorously evaluate whether the presence of XMRV/MLV nucleic acids in the blood is associated with CFS. Researchers, working with clinicians in six regions across the United States, will compare blood and plasma samples from patients diagnosed with CFS to samples from healthy people who have not been diagnosed with CFS and who are matched to the CFS patients by age, sex, and geography. Study results are anticipated later this year.
21 Proposed REDS-III XMRV Study Supported by NHLBI Blood donor prevalence over four decades using total of 10,000 samples from 4 NIH repositories: Transfusion Transmitted Viruses Study (TTVS) Transfusion Safety Study (TSS) Retrovirus Epidemiology in Donors Study (REDS) General Leukocyte and Plasma Repository (GLPR) Viral Activation by Transfusions Study (VATS) REDS Allogeneic Donor and Recipient Repository (RADAR) Transfusion-Transmission using TTVS, VATS and RADAR repositories Rates of transfusion-transmission and correlations of transmission with viral and serologic findings in XMRV+ donations Effect of routine blood filtration (leukoreduction) and blood component storage period on transmission Limited data on mortality and morbidity Utilize Abbott and Gen-Probe high-throughput screening assays for serology and NAT
22 Thank you!
Assays to Address Emerging Threats to Blood Safety
 Assays to Address Emerging Threats to Blood Safety Jeffrey M. Linnen, Ph.D. Director, Product Development Gen-Probe Incorporated San Diego, CA The IPFA/PEI 17th Workshop on Surveillance and Screening of
Assays to Address Emerging Threats to Blood Safety Jeffrey M. Linnen, Ph.D. Director, Product Development Gen-Probe Incorporated San Diego, CA The IPFA/PEI 17th Workshop on Surveillance and Screening of
Expression of HIV-1 Markers During Progression of Infection E E E Days Since 1st Bleed
 Seroconversion OVERVIEW HIV-1 AccuVert TM Seroconversion PRB970 is a 4 member, 1.0 ml per vial panel of undiluted, naturally-occurring plasma samples. members represent serial bleeds collected from a single
Seroconversion OVERVIEW HIV-1 AccuVert TM Seroconversion PRB970 is a 4 member, 1.0 ml per vial panel of undiluted, naturally-occurring plasma samples. members represent serial bleeds collected from a single
June 8, Division of Dockets Management (HFA 305) Food and Drug Administration 5630 Fishers Lane, Rm Rockville, MD 20852
 June 8, 2018 Division of Dockets Management (HFA 305) Food and Drug Administration 5630 Fishers Lane, Rm. 1061 Rockville, MD 20852 Submitted via http://www.regulations.gov Re: Docket No. FDA 2016 D 0545,
June 8, 2018 Division of Dockets Management (HFA 305) Food and Drug Administration 5630 Fishers Lane, Rm. 1061 Rockville, MD 20852 Submitted via http://www.regulations.gov Re: Docket No. FDA 2016 D 0545,
*To whom correspondence should be addressed. This PDF file includes:
 www.sciencemag.org/cgi/content/full/science.1212182/dc1 Supporting Online Material for Partial Retraction to Detection of an Infectious Retrovirus, XMRV, in Blood Cells of Patients with Chronic Fatigue
www.sciencemag.org/cgi/content/full/science.1212182/dc1 Supporting Online Material for Partial Retraction to Detection of an Infectious Retrovirus, XMRV, in Blood Cells of Patients with Chronic Fatigue
HIV-1 AccuVert Seroconversion Panel (PRB966) / Batch #
 s/co c/ml DATA SHEET OVERVIEW 0600-0248 (PRB966) is a 10-member panel of undiluted, naturally occurring plasma samples. Panel members represent serial bleeds collected from a single patient over the course
s/co c/ml DATA SHEET OVERVIEW 0600-0248 (PRB966) is a 10-member panel of undiluted, naturally occurring plasma samples. Panel members represent serial bleeds collected from a single patient over the course
Research Article Lack of Detection of Xenotropic Murine Leukemia Virus-Related Virus in HIV-1 Lymphoma Patients
 Advances in Virology Volume 2011, Article ID 797820, 4 pages doi:10.1155/2011/797820 Research Article Lack of Detection of Xenotropic Murine Leukemia Virus-Related Virus in HIV-1 Lymphoma Patients Krista
Advances in Virology Volume 2011, Article ID 797820, 4 pages doi:10.1155/2011/797820 Research Article Lack of Detection of Xenotropic Murine Leukemia Virus-Related Virus in HIV-1 Lymphoma Patients Krista
AccuSet HIV-1/2 Performance Panel
 OVERVIEW AccuSet HIV-1/2 (0800-0380) is a 13-member validation panel of undiluted, naturally occurring plasma samples (1 vial per member, 1.0 ml per vial). Panel members represent bleeds from multiple
OVERVIEW AccuSet HIV-1/2 (0800-0380) is a 13-member validation panel of undiluted, naturally occurring plasma samples (1 vial per member, 1.0 ml per vial). Panel members represent bleeds from multiple
Complicated viral infections
 Complicated viral infections Clinical case discussion Diagnostic dilemmas NSW State Reference Laboratory for HIV St Vincent s Hospital Sydney Diagnostic dilemmas Indeterminate or discordant serology (western
Complicated viral infections Clinical case discussion Diagnostic dilemmas NSW State Reference Laboratory for HIV St Vincent s Hospital Sydney Diagnostic dilemmas Indeterminate or discordant serology (western
HIV-1 Seroconversion Panel PRB964
 A SERACARE PANEL PRODUCT Seroconversion INTENDED USE The is a group of serial bleeds from an individual plasma donor during seroconversion. This is intended for use by diagnostics manufacturers and clinical
A SERACARE PANEL PRODUCT Seroconversion INTENDED USE The is a group of serial bleeds from an individual plasma donor during seroconversion. This is intended for use by diagnostics manufacturers and clinical
HIV-1 Seroconversion Panel
 A SERACARE PANEL PRODUCT Seroconversion INTENDED USE The Seroconversion is a group of serial bleeds from an individual plasma donor during seroconversion. This is intended for use by diagnostics manufacturers
A SERACARE PANEL PRODUCT Seroconversion INTENDED USE The Seroconversion is a group of serial bleeds from an individual plasma donor during seroconversion. This is intended for use by diagnostics manufacturers
HIV-1 AccuVert TM Seroconversion Panel
 PACKAGE INSERT PRB954 (0600-0238) INTENDED USE The is intended for use by diagnostic manufacturers, researchers, and clinical laboratories to develop, evaluate, or troubleshoot test methods. Characterized
PACKAGE INSERT PRB954 (0600-0238) INTENDED USE The is intended for use by diagnostic manufacturers, researchers, and clinical laboratories to develop, evaluate, or troubleshoot test methods. Characterized
Dealing with Post-market Issues: PCV Case Study
 Dealing with Post-market Issues: PCV Case Study CASE STUDY: Adventitious agent in raw material ISSUE: Presence of porcine circovirus (PCV-1) DNA detected in marketed rotavirus vaccine by an independent
Dealing with Post-market Issues: PCV Case Study CASE STUDY: Adventitious agent in raw material ISSUE: Presence of porcine circovirus (PCV-1) DNA detected in marketed rotavirus vaccine by an independent
Trends in molecular diagnostics
 Trends in molecular diagnostics Detection of target genes of interest Quantification Infectious diseases HIV Hepatitis C & B TB / MAC Cytomegalovirus Herpes simplex Varicella zoster CT/GC HPV Profiling
Trends in molecular diagnostics Detection of target genes of interest Quantification Infectious diseases HIV Hepatitis C & B TB / MAC Cytomegalovirus Herpes simplex Varicella zoster CT/GC HPV Profiling
HIV-1 Seroconversion Panel
 OVERVIEW This Data Sheet contains test results specific for HIV-1. s are undiluted aliquots from plasma units collected from a single donor. No preservatives were added. CAUTION: Potentially infectious
OVERVIEW This Data Sheet contains test results specific for HIV-1. s are undiluted aliquots from plasma units collected from a single donor. No preservatives were added. CAUTION: Potentially infectious
AccuVert HIV-1 Seroconversion Panel PRB974 ( )
 PACKAGE INSERT PRB974 (0600-0258) INTENDED USE PRB974 (0600-0258) is a group of serial bleeds from an individual plasma donor during seroconversion. This panel is intended for use by diagnostics manufacturers
PACKAGE INSERT PRB974 (0600-0258) INTENDED USE PRB974 (0600-0258) is a group of serial bleeds from an individual plasma donor during seroconversion. This panel is intended for use by diagnostics manufacturers
Sam: Annette, can we just start out with a brief simple explanation of what neuro-immune diseases are, including ME and CFS?
 Sam: Annette, can we just start out with a brief simple explanation of what neuro-immune diseases are, including ME and CFS? Annette: They are systemic diseases that impact the nervous system and immune
Sam: Annette, can we just start out with a brief simple explanation of what neuro-immune diseases are, including ME and CFS? Annette: They are systemic diseases that impact the nervous system and immune
Picture of cell art Picture of cell under a microscope Daniel L. Peterson, MD Whittemore Peterson Institute
 XMRV, A New Human Pathogenic Retrovirus: Detection In Chronic Diseases Picture of cell art Picture of cell under a microscope Daniel L. Peterson, MD Whittemore Peterson Institute Daniel L. Peterson, MD
XMRV, A New Human Pathogenic Retrovirus: Detection In Chronic Diseases Picture of cell art Picture of cell under a microscope Daniel L. Peterson, MD Whittemore Peterson Institute Daniel L. Peterson, MD
AccuVert HBV Seroconversion Panel PHM941(M) ( )
 PACKAGE INSERT PHM941(M) (0605-0061) INTENDED USE PHM941(M) (0605-0061) is a group of serial bleeds from an individual plasma donor during HBV seroconversion. This panel is intended for use by diagnostics
PACKAGE INSERT PHM941(M) (0605-0061) INTENDED USE PHM941(M) (0605-0061) is a group of serial bleeds from an individual plasma donor during HBV seroconversion. This panel is intended for use by diagnostics
Development of sero-assays to screen for XMRV antibodies in clinical samples
 National Cancer Institute Development of sero-assays to screen for XMRV antibodies in clinical samples Rachel K. Bagni, Ph.D. September 8, 2010 Xentropic murine leukaemia virus-related virus - XMRV The
National Cancer Institute Development of sero-assays to screen for XMRV antibodies in clinical samples Rachel K. Bagni, Ph.D. September 8, 2010 Xentropic murine leukaemia virus-related virus - XMRV The
Parvovirus B19 Mixed Titer Performance Panel PVP201
 A SERACARE PANEL PRODUCT INTENDED USE The is a panel of naturally occurring plasma specimens. This panel is intended for use by diagnostic manufacturers and clinical laboratories to evaluate their Parvovirus
A SERACARE PANEL PRODUCT INTENDED USE The is a panel of naturally occurring plasma specimens. This panel is intended for use by diagnostic manufacturers and clinical laboratories to evaluate their Parvovirus
No Evidence of Murine-Like Gammaretroviruses in CFS Patients Previously Identified as XMRV-Infected
 No Evidence of Murine-Like Gammaretroviruses in CFS Patients Previously Identified as XMRV-Infected Konstance Knox, 1,2 Donald Carrigan, 1,2 Graham Simmons, 3,4 Fernando Teque, 5 Yanchen Zhou, 3,4 John
No Evidence of Murine-Like Gammaretroviruses in CFS Patients Previously Identified as XMRV-Infected Konstance Knox, 1,2 Donald Carrigan, 1,2 Graham Simmons, 3,4 Fernando Teque, 5 Yanchen Zhou, 3,4 John
AccuVert HIV-1 Seroconversion Panel PRB955 ( ) / Batch #
 Signal to cutoff (s/co) Copies per ml (c/ml) OVERVIEW AccuVert is a 5-member validation panel of undiluted, naturally occurring plasma samples (1 vial per member, 1.0 ml per vial). Panel members represent
Signal to cutoff (s/co) Copies per ml (c/ml) OVERVIEW AccuVert is a 5-member validation panel of undiluted, naturally occurring plasma samples (1 vial per member, 1.0 ml per vial). Panel members represent
AccuSet Anti-HIV-1 Mixed Titer Performance Panel
 signal to cut-off (s/co) DATA SHEET OVERVIEW PRB205(M3) is a modified 16-member panel originating from Anti- HIV-1 Mixed Titer PRB205(M2). Panel members 3, 5, 6, 17, 21, 22, 23, 24, and 25 from the original
signal to cut-off (s/co) DATA SHEET OVERVIEW PRB205(M3) is a modified 16-member panel originating from Anti- HIV-1 Mixed Titer PRB205(M2). Panel members 3, 5, 6, 17, 21, 22, 23, 24, and 25 from the original
HIV-1 Seroconversion Panel PRB975
 Seroconversion OVERVIEW is a five member HIV Seroconversion collected from a single donor over a 14 day period in 2002, prior to antibody seroconversion. -03 is a challenging sample for HIV RNA assays,
Seroconversion OVERVIEW is a five member HIV Seroconversion collected from a single donor over a 14 day period in 2002, prior to antibody seroconversion. -03 is a challenging sample for HIV RNA assays,
HIV-1 Seroconversion Panel PRB973
 A SERACARE PANEL PRODUCT INTENDED USE The is a group of serial bleeds from an individual plasma donor during seroconversion. This panel is intended for use by diagnostics manufacturers and clinical laboratorians
A SERACARE PANEL PRODUCT INTENDED USE The is a group of serial bleeds from an individual plasma donor during seroconversion. This panel is intended for use by diagnostics manufacturers and clinical laboratorians
Impact of Testing Strategies to Reduce Transmission Risk for HBV. Ravi Reddy, M Vermeulen South African National Blood Service (SANBS) 29 July 2013
 Impact of Testing Strategies to Reduce Transmission Risk for HBV Ravi Reddy, M Vermeulen South African National Blood Service (SANBS) 29 July 2013 Overview of SANBS SANBS is a private not for profit company
Impact of Testing Strategies to Reduce Transmission Risk for HBV Ravi Reddy, M Vermeulen South African National Blood Service (SANBS) 29 July 2013 Overview of SANBS SANBS is a private not for profit company
AccuSet Anti-HIV-1 Mixed Titer Performance Panel
 PACKAGE INSERT PRB205(M3) / (0800-0379) INTENDED USE The Performance Panel PRB205(M3) / (0800-0379) is intended for use by diagnostics manufacturers and clinical laboratorians to evaluate their HIV-1 test
PACKAGE INSERT PRB205(M3) / (0800-0379) INTENDED USE The Performance Panel PRB205(M3) / (0800-0379) is intended for use by diagnostics manufacturers and clinical laboratorians to evaluate their HIV-1 test
Opportunities Created by Diagnostic HCV and HIV Nucleic Acid Tests
 Opportunities Created by Diagnostic HCV and HIV Nucleic Acid Tests Ann Winters, MD Medical Director, Viral Hepatitis Program, Bureau of Communicable Disease, New York City Department of Health and Mental
Opportunities Created by Diagnostic HCV and HIV Nucleic Acid Tests Ann Winters, MD Medical Director, Viral Hepatitis Program, Bureau of Communicable Disease, New York City Department of Health and Mental
ISBT WP-TTID Annual Report for Subgroup on Virology Drs. Michael Busch, Kurt Roth and Susan Stramer
 ISBT WP-TTID Annual Report for Subgroup on Virology Drs. Michael Busch, Kurt Roth and Susan Stramer Questionnaire on NAT Screening of Blood Donations for an International Forum on 10 years of NAT Screening
ISBT WP-TTID Annual Report for Subgroup on Virology Drs. Michael Busch, Kurt Roth and Susan Stramer Questionnaire on NAT Screening of Blood Donations for an International Forum on 10 years of NAT Screening
HIV Testing. Susan Tusher, LMSW Program Coordinator The Kansas AIDS Education and Training Center
 HIV Testing Susan Tusher, LMSW Program Coordinator The Kansas AIDS Education and Training Center The Number of Persons in the US Living with HIV Continues to Increase Over 1 Million people are estimated
HIV Testing Susan Tusher, LMSW Program Coordinator The Kansas AIDS Education and Training Center The Number of Persons in the US Living with HIV Continues to Increase Over 1 Million people are estimated
Guidance for Industry
 Guidance for Industry Adequate and Appropriate Donor Screening Tests for Hepatitis B; Hepatitis B Surface Antigen (HBsAg) Assays Used to Test Donors of Whole Blood and Blood Components, Including Source
Guidance for Industry Adequate and Appropriate Donor Screening Tests for Hepatitis B; Hepatitis B Surface Antigen (HBsAg) Assays Used to Test Donors of Whole Blood and Blood Components, Including Source
European Centre for Disease Prevention and Control. Zika virus disease
 European Centre for Disease Prevention and Control Zika virus disease Stockholm, 19 February 2016 Background information Zika virus is a member of the Flaviviridae family and transmitted by mosquitoes
European Centre for Disease Prevention and Control Zika virus disease Stockholm, 19 February 2016 Background information Zika virus is a member of the Flaviviridae family and transmitted by mosquitoes
Dengue Fever & Dengue Hemorrhagic Fever Annual Reports to WHO
 Dengue Virus Member of the genus Flavivirus Transmitted by the Aedes mosquito; mosquito => human cycle 4 serotypes: DENV1, 2, 3, and 4 Homotypic immunity is long lasting Heterotypic immunity is short lived
Dengue Virus Member of the genus Flavivirus Transmitted by the Aedes mosquito; mosquito => human cycle 4 serotypes: DENV1, 2, 3, and 4 Homotypic immunity is long lasting Heterotypic immunity is short lived
Center for Biologics Evaluation and Research
 Center for Biologics Evaluation and Research Current Activities Future Directions Washington, DC January 25, 2010 Karen Midthun, MD Acting Director, CBER CBER Our Mission To ensure the safety, purity,
Center for Biologics Evaluation and Research Current Activities Future Directions Washington, DC January 25, 2010 Karen Midthun, MD Acting Director, CBER CBER Our Mission To ensure the safety, purity,
The Alphabet Soup of Viral Hepatitis Testing
 The Alphabet Soup of Viral Hepatitis Testing August 18, 2011 Patricia Slev, PhD, DABCC Medical Director, Serologic Hepatitis and Retrovirus Laboratory, ARUP Laboratories Assistant Professor of Pathology,
The Alphabet Soup of Viral Hepatitis Testing August 18, 2011 Patricia Slev, PhD, DABCC Medical Director, Serologic Hepatitis and Retrovirus Laboratory, ARUP Laboratories Assistant Professor of Pathology,
Khalid Alquthami (Correspondence) Regional Lab, Makkah. Saudi Arabia
 1 Regional Lab, Makkah. Saudi Arabia stract: Clinical specificity and genotype/subtype detection of viruses using the Cobas TaqScreen MPX system V 2.0, which is a nucleic acid test (NAT) that uses multiples
1 Regional Lab, Makkah. Saudi Arabia stract: Clinical specificity and genotype/subtype detection of viruses using the Cobas TaqScreen MPX system V 2.0, which is a nucleic acid test (NAT) that uses multiples
ARCHITECT HIV Ag/Ab Combo: Moving HIV Diagnostics Forward in the U.S.
 ARCHITECT HIV Ag/Ab Combo: Moving HIV Diagnostics Forward in the U.S. Catherine Brennan, Ph.D. Research Fellow Infectious Diseases Research Abbott Diagnostics 1 Agenda ARCHITECT HIV Ag/Ab Combo Assay What
ARCHITECT HIV Ag/Ab Combo: Moving HIV Diagnostics Forward in the U.S. Catherine Brennan, Ph.D. Research Fellow Infectious Diseases Research Abbott Diagnostics 1 Agenda ARCHITECT HIV Ag/Ab Combo Assay What
-738 proteins were found only in the ME subjects proteins were only found in PTLS samples proteins were only found in the normal controls.
 RECENT RESEARCH Currently there is much exciting research being published including the Schutzer et al. study that compared cerebrospinal fluid proteomes to differentiate ME and Post Treatment Lyme Syndrome
RECENT RESEARCH Currently there is much exciting research being published including the Schutzer et al. study that compared cerebrospinal fluid proteomes to differentiate ME and Post Treatment Lyme Syndrome
HIV Diagnostic Testing
 In The name of God HIV Diagnostic Testing By : Dr. Shahzamani PhD of Medical virology Purpose of HIV Testing To identify asymptomatic individuals To diagnose HIV infection in those who practice high risk
In The name of God HIV Diagnostic Testing By : Dr. Shahzamani PhD of Medical virology Purpose of HIV Testing To identify asymptomatic individuals To diagnose HIV infection in those who practice high risk
Reducing Babesia and Malaria Risks: Testing, Donor Selection, Inactivation. Susan L. Stramer PhD IPFA 23 rd Meeting May
 Reducing Babesia and Malaria Risks: Testing, Donor Selection, Inactivation Susan L. Stramer PhD IPFA 23 rd Meeting May 26 2016 Babesiosis Malaria-like illness caused by babesia spp. Asymptomatic fatal
Reducing Babesia and Malaria Risks: Testing, Donor Selection, Inactivation Susan L. Stramer PhD IPFA 23 rd Meeting May 26 2016 Babesiosis Malaria-like illness caused by babesia spp. Asymptomatic fatal
Guidance for Industry
 Guidance for Industry Lookback for Hepatitis C Virus (HCV): Product Quarantine, Consignee Notification, Further Testing, Product Disposition, and Notification of Transfusion Recipients Based on Donor Test
Guidance for Industry Lookback for Hepatitis C Virus (HCV): Product Quarantine, Consignee Notification, Further Testing, Product Disposition, and Notification of Transfusion Recipients Based on Donor Test
The Cat with FIV: The Vaccine and Diagnostic Testing
 The Cat with FIV: The Vaccine and Diagnostic Testing Richard B. Ford, DVM, MS Diplomate ACVIM Diplomate ACVPM (Hon) Professor of Medicine North Carolina State University In July 2002, the first licensed
The Cat with FIV: The Vaccine and Diagnostic Testing Richard B. Ford, DVM, MS Diplomate ACVIM Diplomate ACVPM (Hon) Professor of Medicine North Carolina State University In July 2002, the first licensed
AccuSet HBV Worldwide Performance Panel
 PACKAGE INSERT 0805-033 INTENDED USE The is intended for use by diagnostic manufacturers, researchers, and clinical laboratories to develop, evaluate, or troubleshoot HBV test methods. Characterized samples
PACKAGE INSERT 0805-033 INTENDED USE The is intended for use by diagnostic manufacturers, researchers, and clinical laboratories to develop, evaluate, or troubleshoot HBV test methods. Characterized samples
Blood Supply and Wastage
 Blood Supply and Wastage Inga Willett Lab Matters, Oake Manor, 21 st June 2017 Blood supply UK supplied by 4 blood services: SNBTS NIBTS NHSBT WBS http://commons.wikimedia.org/wiki/file:uk_map_home_nations.png
Blood Supply and Wastage Inga Willett Lab Matters, Oake Manor, 21 st June 2017 Blood supply UK supplied by 4 blood services: SNBTS NIBTS NHSBT WBS http://commons.wikimedia.org/wiki/file:uk_map_home_nations.png
HAEMOPHILUS INFLUENZAE INVASIVE DISEASE
 23 Annual Morbidity Report HAEMOPHILUS INFLUENZAE INVASIVE DISEASE CRUDE DATA 35 Annual Incidence a LA County.37 California b. United States c.2 Age at Diagnosis Mean 4. years Median 36. years Range Birth
23 Annual Morbidity Report HAEMOPHILUS INFLUENZAE INVASIVE DISEASE CRUDE DATA 35 Annual Incidence a LA County.37 California b. United States c.2 Age at Diagnosis Mean 4. years Median 36. years Range Birth
Learning Objectives. New HIV Testing Algorithm from CDC. Overview of HIV infection and disease 3/15/2016
 New HIV Testing Algorithm from CDC ASCLS-Michigan March 31, 2016 Dr. Kathleen Hoag Learning Objectives Following attendance and review of material provided, attendees will be able to: 1. Describe the new
New HIV Testing Algorithm from CDC ASCLS-Michigan March 31, 2016 Dr. Kathleen Hoag Learning Objectives Following attendance and review of material provided, attendees will be able to: 1. Describe the new
HIV Update in Laboratory Testing. Patricia Slev, PhD, D(ABCC)
 HIV Update in Laboratory Testing Patricia Slev, PhD, D(ABCC) Objectives Explain the advances in HIV diagnostics, including fourth generation Ag/Ab combination HIV screening assays Describe the new CDC
HIV Update in Laboratory Testing Patricia Slev, PhD, D(ABCC) Objectives Explain the advances in HIV diagnostics, including fourth generation Ag/Ab combination HIV screening assays Describe the new CDC
Donation Criteria for Men who have Sex with Men (MSM): Update from Canadian Blood Services
 Donation Criteria for Men who have Sex with Men (MSM): Update from Canadian Blood Services Outline 1 Current criteria Proposed criteria Stakeholder outreach Outline 2 Safety Assessment Results since implementation
Donation Criteria for Men who have Sex with Men (MSM): Update from Canadian Blood Services Outline 1 Current criteria Proposed criteria Stakeholder outreach Outline 2 Safety Assessment Results since implementation
McLean ebasis plus TM
 McLean ebasis plus TM Sample Hospital (0000) Report For Qtr HBIPS Core Measures McLean Hospital 115 Mill Street Belmont, MA 02478 1 2012 Department of Mental Health Services Evaluation Tel: 617-855-3797
McLean ebasis plus TM Sample Hospital (0000) Report For Qtr HBIPS Core Measures McLean Hospital 115 Mill Street Belmont, MA 02478 1 2012 Department of Mental Health Services Evaluation Tel: 617-855-3797
HIV Testing Technology and the Latest Algorithm
 HIV Testing Technology and the Latest Algorithm David Warshauer, PhD, D(ABMM) Deputy Director, Communicable Diseases Wisconsin State Laboratory of Hygiene HIV Testing has changed over time Patients with
HIV Testing Technology and the Latest Algorithm David Warshauer, PhD, D(ABMM) Deputy Director, Communicable Diseases Wisconsin State Laboratory of Hygiene HIV Testing has changed over time Patients with
This Week in Virology
 This Week in Virology with Vincent Racaniello, Ph.D. Special Episode: A Paradigm for Pathogen De-Discovery Interview with Dr. W. Ian Lipkin Aired 18 September 2012 http://www.twiv.tv/2012/09/18/twiv-special-a-paradigm-for-pathogen-de-discovery/
This Week in Virology with Vincent Racaniello, Ph.D. Special Episode: A Paradigm for Pathogen De-Discovery Interview with Dr. W. Ian Lipkin Aired 18 September 2012 http://www.twiv.tv/2012/09/18/twiv-special-a-paradigm-for-pathogen-de-discovery/
STRENGTHENING THE COORDINATION, DELIVERY AND MONITORING OF HIV AND AIDS SERVICES IN MALAWI THROUGH FAITH-BASED INSTITUTIONS.
 STRENGTHENING THE COORDINATION, DELIVERY AND MONITORING OF HIV AND AIDS SERVICES IN MALAWI THROUGH FAITH-BASED INSTITUTIONS. Acknowledgements This project was fully funded by Center For Disease Control
STRENGTHENING THE COORDINATION, DELIVERY AND MONITORING OF HIV AND AIDS SERVICES IN MALAWI THROUGH FAITH-BASED INSTITUTIONS. Acknowledgements This project was fully funded by Center For Disease Control
Influenza Surveillance Animal and Public Health Partnership. Jennifer Koeman Director, Producer and Public Health National Pork Board
 Influenza Surveillance Animal and Public Health Partnership Jennifer Koeman Director, Producer and Public Health National Pork Board Outline Background on influenza surveillance in swine Case example animal
Influenza Surveillance Animal and Public Health Partnership Jennifer Koeman Director, Producer and Public Health National Pork Board Outline Background on influenza surveillance in swine Case example animal
A Report from the 9th Hemispherx Biopharma Investigators Meeting
 XMRV & AMPLIGEN A Report from the 9th Hemispherx Biopharma Investigators Meeting HUNTER HOPKINS CENTER Hunter-Hopkins ME-letter March 2011 http://www.drlapp.net/melettermar2011.htm Hunter-Hopkins Center,
XMRV & AMPLIGEN A Report from the 9th Hemispherx Biopharma Investigators Meeting HUNTER HOPKINS CENTER Hunter-Hopkins ME-letter March 2011 http://www.drlapp.net/melettermar2011.htm Hunter-Hopkins Center,
ZIKA VIRUS AND SAFETY OF THE BLOOD SUPPLY. Michael P Busch, MD, PhD Blood Systems Research Institute University of California, San Francisco
 ZIKA VIRUS AND SAFETY OF THE BLOOD SUPPLY Michael P Busch, MD, PhD Blood Systems Research Institute University of California, San Francisco ZIKV Risks of major TTVs linked to interventions, and accelerating
ZIKA VIRUS AND SAFETY OF THE BLOOD SUPPLY Michael P Busch, MD, PhD Blood Systems Research Institute University of California, San Francisco ZIKV Risks of major TTVs linked to interventions, and accelerating
Measles: United States, January 1 through June 10, 2011
 Measles: United States, January 1 through June 10, 2011 Preeta K. Kutty, MD, MPH Measles, Mumps, Rubella and Polio Team Division of Viral Diseases Centers for Disease Control and Prevention Atlanta, GA
Measles: United States, January 1 through June 10, 2011 Preeta K. Kutty, MD, MPH Measles, Mumps, Rubella and Polio Team Division of Viral Diseases Centers for Disease Control and Prevention Atlanta, GA
Quantification of HBV, HCV genotype and HIV subtype panels
 Quantification of HBV, HCV genotype and HIV subtype panels Harry van Drimmelen 1,2, Wim Quint 2, Nico Lelie 3 and the international NAT study group 1. Biologicals Quality Control, 2. DDL Diagnostic Laboratory,
Quantification of HBV, HCV genotype and HIV subtype panels Harry van Drimmelen 1,2, Wim Quint 2, Nico Lelie 3 and the international NAT study group 1. Biologicals Quality Control, 2. DDL Diagnostic Laboratory,
Transfusion-transmitted Cytomegalovirus
 Transfusion-transmitted Cytomegalovirus Can you confidently abandon CMV seronegative products in the modern era of pre-storage leukoreduction? Jeannie Callum, BA, MD, FRCPC Really? Are we still talking
Transfusion-transmitted Cytomegalovirus Can you confidently abandon CMV seronegative products in the modern era of pre-storage leukoreduction? Jeannie Callum, BA, MD, FRCPC Really? Are we still talking
Interventions for Babesia (and Plasmodium)
 Interventions for Babesia (and Plasmodium) Susan L. Stramer, Ph.D. May 16 2017 www.aabb.org Babesiosis Malaria-like illness caused by Babesia spp. Asymptomatic fatal Non-specific symptoms (malaise, fever,
Interventions for Babesia (and Plasmodium) Susan L. Stramer, Ph.D. May 16 2017 www.aabb.org Babesiosis Malaria-like illness caused by Babesia spp. Asymptomatic fatal Non-specific symptoms (malaise, fever,
AccuSet HCV Performance Panel
 Signal to cutoff (s/co) DATA SHEET OVERVIEW AccuSet HCV (0810-0204) is a 25-member validation panel of undiluted, naturally occurring plasma samples (1 vial per member, 1.0 ml per vial). Panel members
Signal to cutoff (s/co) DATA SHEET OVERVIEW AccuSet HCV (0810-0204) is a 25-member validation panel of undiluted, naturally occurring plasma samples (1 vial per member, 1.0 ml per vial). Panel members
An Updated Approach to Colon Cancer Screening and Prevention
 An Updated Approach to Colon Cancer Screening and Prevention Kevin Liebovich, MD Director for Quality for Gastrointestinal diseases Advocate Condell Medical Center Colon Cancer Screening and Prevention
An Updated Approach to Colon Cancer Screening and Prevention Kevin Liebovich, MD Director for Quality for Gastrointestinal diseases Advocate Condell Medical Center Colon Cancer Screening and Prevention
Influenza 2009: Not Yet The Perfect Storm
 Influenza 2009: Not Yet The Perfect Storm What s needed for a pandemic strain? Novel virus (little to no immunity) Capable of causing disease in humans Highly pathogenic / virulent Capable of sustained
Influenza 2009: Not Yet The Perfect Storm What s needed for a pandemic strain? Novel virus (little to no immunity) Capable of causing disease in humans Highly pathogenic / virulent Capable of sustained
Clinical HIV-2 Viral Load Testing of a Large Population of HIV-2 Infected Individuals
 Clinical HIV-2 Viral Load Testing of a Large Population of HIV-2 Infected Individuals Linda M. Styer Wadsworth Center, NYSDOH ** The authors have no conflicts of interest to disclose ** HIV-2 viral load
Clinical HIV-2 Viral Load Testing of a Large Population of HIV-2 Infected Individuals Linda M. Styer Wadsworth Center, NYSDOH ** The authors have no conflicts of interest to disclose ** HIV-2 viral load
Seasonality of influenza activity in Hong Kong and its association with meteorological variations
 Seasonality of influenza activity in Hong Kong and its association with meteorological variations Prof. Paul Chan Department of Microbiology The Chinese University of Hong Kong Mr. HY Mok Senior Scientific
Seasonality of influenza activity in Hong Kong and its association with meteorological variations Prof. Paul Chan Department of Microbiology The Chinese University of Hong Kong Mr. HY Mok Senior Scientific
Armed Services Blood Program
 Armed Services Blood Program Defense Health Board Concerns Regarding the Collection and Transfusion of Non-FDA Compliant Blood Products in Theater Information Brief Defense Health Board 17 August 2009
Armed Services Blood Program Defense Health Board Concerns Regarding the Collection and Transfusion of Non-FDA Compliant Blood Products in Theater Information Brief Defense Health Board 17 August 2009
The CIDRZ Experience: use of data to understand patient outcomes and guide program implementation 07 January 2010
 The CIDRZ Experience: use of data to understand patient outcomes and guide program implementation 07 January 2010 Dr C Bolton Moore Deputy Medical Director CIDRZ Ministry of Health Zambia University of
The CIDRZ Experience: use of data to understand patient outcomes and guide program implementation 07 January 2010 Dr C Bolton Moore Deputy Medical Director CIDRZ Ministry of Health Zambia University of
HIV Serology Quality Assessment Program Summary for Panel HIVSER 2017Apr19
 National Laboratory for HIV Reference Services National HIV and Retrovirology Laboratories National Microbiology Laboratory Public Health Agency of Canada HIV Serology Quality Assessment Program Summary
National Laboratory for HIV Reference Services National HIV and Retrovirology Laboratories National Microbiology Laboratory Public Health Agency of Canada HIV Serology Quality Assessment Program Summary
Multiple Sources of Contamination in Samples from Patients Reported to Have XMRV Infection
 Multiple Sources of Contamination in Samples from Patients Reported to Have XMRV Infection Mary F. Kearney 1 *, Jonathan Spindler 1, Ann Wiegand 1, Wei Shao 2, Elizabeth M. Anderson 1, Frank Maldarelli
Multiple Sources of Contamination in Samples from Patients Reported to Have XMRV Infection Mary F. Kearney 1 *, Jonathan Spindler 1, Ann Wiegand 1, Wei Shao 2, Elizabeth M. Anderson 1, Frank Maldarelli
Diagnosis and Initial Management of HIV/AIDS: What the Primary Care Provider Should Know
 Diagnosis and Initial Management of HIV/AIDS: What the Primary Care Provider Should Know Carolyn K. Burr, EdD, RN Co-Clinical Director Deputy Director François-Xavier Bagnoud Center December 17 th, 2013
Diagnosis and Initial Management of HIV/AIDS: What the Primary Care Provider Should Know Carolyn K. Burr, EdD, RN Co-Clinical Director Deputy Director François-Xavier Bagnoud Center December 17 th, 2013
RNA PCR, Proviral DNA and Emerging Trends in Infant HIV Diagnosis
 RNA PCR, Proviral DNA and Emerging Trends in Infant HIV Diagnosis 1 B R E N D A N M C M U L L A N I N F E C T I O U S D I S E A S E S, S Y D N E Y C H I L D R E N S H O S P I T A L S C H O O L O F W O
RNA PCR, Proviral DNA and Emerging Trends in Infant HIV Diagnosis 1 B R E N D A N M C M U L L A N I N F E C T I O U S D I S E A S E S, S Y D N E Y C H I L D R E N S H O S P I T A L S C H O O L O F W O
DENGUE AND BLOOD SAFETY. Ester C Sabino, MD, PhD Dep. of Infectious Disease/Institute of Tropical Medicine University of São Paulo
 DENGUE AND BLOOD SAFETY Ester C Sabino, MD, PhD Dep. of Infectious Disease/Institute of Tropical Medicine University of São Paulo Dengue virus Arbovirus (arthropod-borne virus) virus transmitted by mosquitoes:
DENGUE AND BLOOD SAFETY Ester C Sabino, MD, PhD Dep. of Infectious Disease/Institute of Tropical Medicine University of São Paulo Dengue virus Arbovirus (arthropod-borne virus) virus transmitted by mosquitoes:
Frequency of occult hepatitis B in HBsAg seronegative blood donors in a tertiary care hospital in kerala,south India.
 Frequency of occult hepatitis B in HBsAg seronegative blood donors in a tertiary care hospital in kerala,south India. Cinzia Keechilot, Veena Shenoy 1,V Anil kumar 2,Lalita Biswas 3. MBBS student * Transfusion
Frequency of occult hepatitis B in HBsAg seronegative blood donors in a tertiary care hospital in kerala,south India. Cinzia Keechilot, Veena Shenoy 1,V Anil kumar 2,Lalita Biswas 3. MBBS student * Transfusion
McHenry County Norovirus Outbreaks November McHenry County Department of Health November 29,2010
 McHenry County Norovirus Outbreaks November 2010 McHenry County Department of Health November 29,2010 What is Norovirus The most common cause of gastrointestinal illness resulting from an inflammation
McHenry County Norovirus Outbreaks November 2010 McHenry County Department of Health November 29,2010 What is Norovirus The most common cause of gastrointestinal illness resulting from an inflammation
General Terms: Appendix B. National Marrow Donor Program and The Medical College of Wisconsin
 Glossary of Terms This appendix is divided into two sections. The first section, General Terms, defines terms used throughout the CIBMTR data collection forms. The second section, FormsNet TM 2 Terms,
Glossary of Terms This appendix is divided into two sections. The first section, General Terms, defines terms used throughout the CIBMTR data collection forms. The second section, FormsNet TM 2 Terms,
BACTERIAL INTERVENTIONS TO INCREASE BLOOD SAFETY
 BACTERIAL INTERVENTIONS TO INCREASE BLOOD SAFETY Jennifer Allen National Bacteriology Laboratory on behalf of Dr. C P McDonald Head of Bacteriology National Bacteriology Laboratory NHSBT Klebsiella oxytoca
BACTERIAL INTERVENTIONS TO INCREASE BLOOD SAFETY Jennifer Allen National Bacteriology Laboratory on behalf of Dr. C P McDonald Head of Bacteriology National Bacteriology Laboratory NHSBT Klebsiella oxytoca
Update on Transfusion- Transmitted Infectious Diseases
 Update on Transfusion- Transmitted Infectious Diseases Scott Koepsell MD PhD Associate Professor University of Nebraska Medical Center I have no conflicts of interest to disclose. Global changes and the
Update on Transfusion- Transmitted Infectious Diseases Scott Koepsell MD PhD Associate Professor University of Nebraska Medical Center I have no conflicts of interest to disclose. Global changes and the
Global and National Trends in Vaccine Preventable Diseases. Dr Brenda Corcoran National Immunisation Office.
 Global and National Trends in Vaccine Preventable Diseases Dr Brenda Corcoran National Immunisation Office Global mortality 2008 Children under 5 years of age 1.5 million deaths due to vaccine preventable
Global and National Trends in Vaccine Preventable Diseases Dr Brenda Corcoran National Immunisation Office Global mortality 2008 Children under 5 years of age 1.5 million deaths due to vaccine preventable
Research Article Decision on conducting HCV Immunoblot and HCV Viral Load Tests Dependent upon the Result of the Screening Tests
 IBIMA Publishing Journal of Virology & Microbiology http://www.ibimapublishing.com/journals/jvm/jvm.html Vol. 2013 (2013), Article ID 332501, 7 pages DOI: 10.5171/2013.332501 Research Article Decision
IBIMA Publishing Journal of Virology & Microbiology http://www.ibimapublishing.com/journals/jvm/jvm.html Vol. 2013 (2013), Article ID 332501, 7 pages DOI: 10.5171/2013.332501 Research Article Decision
FGSZ Zrt. from 28 February 2019 till 29 February 2020 AUCTION CALENDAR: YEARLY YEARLY BUNDLED AT CROSS BORDER POINTS
 AUCTION CALENDAR: YEARLY YEARLY BUNDLED AT CROSS BORDER POINTS FIRM CAPACITY - First Monday of July 01.06.2019* 01.07.2019 07:00 01.10.2019 04:00 01.10.2020 04:00 2019/2020 01.10.2020 04:00 01.10.2021
AUCTION CALENDAR: YEARLY YEARLY BUNDLED AT CROSS BORDER POINTS FIRM CAPACITY - First Monday of July 01.06.2019* 01.07.2019 07:00 01.10.2019 04:00 01.10.2020 04:00 2019/2020 01.10.2020 04:00 01.10.2021
Malaria screening of donors and donations - issues and outcomes
 Malaria screening of donors and donations - issues and outcomes lan Kitchen National Transfusion Microbiology NHS Blood & Transplant London IPF/PEI, Rome, May 2014 Blood and Transplant Key elements Nature
Malaria screening of donors and donations - issues and outcomes lan Kitchen National Transfusion Microbiology NHS Blood & Transplant London IPF/PEI, Rome, May 2014 Blood and Transplant Key elements Nature
Hepatitis E Vaccine Clinical Experience. Mrigendra P. Shrestha
 Hepatitis E Vaccine Clinical Experience Mrigendra P. Shrestha 1 Hepatitis E in Nepal 2 Hepatitis E in Nepal Most common type of acute viral hepatitis Occurs in annual rainy season outbreaks Responsible
Hepatitis E Vaccine Clinical Experience Mrigendra P. Shrestha 1 Hepatitis E in Nepal 2 Hepatitis E in Nepal Most common type of acute viral hepatitis Occurs in annual rainy season outbreaks Responsible
Source Plasma Safety
 Source Plasma Safety Plasma May 15-19, 2017 George Schreiber, ScD, PPTA Director, Epidemiology Three components: Plasma Product Safety Donor Quality Manufacturing Pool Quality Fractionation Process Safe
Source Plasma Safety Plasma May 15-19, 2017 George Schreiber, ScD, PPTA Director, Epidemiology Three components: Plasma Product Safety Donor Quality Manufacturing Pool Quality Fractionation Process Safe
WHO Parvovirus B19 Genotype Panel
 WHO Parvovirus B19 Genotype Panel Mei-ying W Yu, PhD SoGAT XXII Rome, 14-15 April 2011 1 st WHO International Reference Panel for Parvovirus B19 Genotypes In Oct 2009, a plasma-derived parvovirus B19 (B19V)
WHO Parvovirus B19 Genotype Panel Mei-ying W Yu, PhD SoGAT XXII Rome, 14-15 April 2011 1 st WHO International Reference Panel for Parvovirus B19 Genotypes In Oct 2009, a plasma-derived parvovirus B19 (B19V)
Technical Bulletin No. 161
 CPAL Central Pennsylvania Alliance Laboratory Technical Bulletin No. 161 cobas 6800 HIV-1 Viral Load Assay - New Platform - June 1, 2017 Contact: Heather Habig, MLS (ASCP) CM, MB CM, 717-851-1422 Operations
CPAL Central Pennsylvania Alliance Laboratory Technical Bulletin No. 161 cobas 6800 HIV-1 Viral Load Assay - New Platform - June 1, 2017 Contact: Heather Habig, MLS (ASCP) CM, MB CM, 717-851-1422 Operations
AccuSet Autoimmune Performance Panel
 PACKAGE INSERT 0840-0001 INTENDED USE The 0840-0001 is intended for use by diagnostic manufacturers, researchers, and clinical laboratories to develop, evaluate, or troubleshoot autoimmune related test
PACKAGE INSERT 0840-0001 INTENDED USE The 0840-0001 is intended for use by diagnostic manufacturers, researchers, and clinical laboratories to develop, evaluate, or troubleshoot autoimmune related test
Impact of multi-dye multiplex technology on testing algorithm
 Impact of multi-dye multiplex technology on testing algorithm Lydia Blanco. Mª Isabel Gonzalez-Fraile Centro de Hemoterapia y Hemodonación de Castilla y León, España TTI EPIDEMIOLOGICAL DATA IN SPAIN NAT
Impact of multi-dye multiplex technology on testing algorithm Lydia Blanco. Mª Isabel Gonzalez-Fraile Centro de Hemoterapia y Hemodonación de Castilla y León, España TTI EPIDEMIOLOGICAL DATA IN SPAIN NAT
Preventing CMV Transmission through Leukodepletion
 Preventing CMV Transmission through Leukodepletion Possibility & Facts Prof.S.B.Rajadhyaksha, MD,DTM,PGDMLS Head, Dept. of Transfusion Medicine Tata Memorial Hospital, Mumbai 1 Donor Leukocytes Linked
Preventing CMV Transmission through Leukodepletion Possibility & Facts Prof.S.B.Rajadhyaksha, MD,DTM,PGDMLS Head, Dept. of Transfusion Medicine Tata Memorial Hospital, Mumbai 1 Donor Leukocytes Linked
The scientist who put the nail in XMRV's coffin
 Nature News: Q&A IACFS/ME Newsletter December 2012 Attachment 5 The scientist who put the nail in XMRV's coffin W. Ian Lipkin tells Nature about his efforts to validate the link between retroviruses and
Nature News: Q&A IACFS/ME Newsletter December 2012 Attachment 5 The scientist who put the nail in XMRV's coffin W. Ian Lipkin tells Nature about his efforts to validate the link between retroviruses and
Wild Poliovirus*, 03 Aug 2004 to 02 Aug 2005
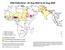 Wild Poliovirus*, 03 Aug 2004 to 02 Aug 2005 Wild virus type 1 Wild virus type 3 Wild virus type 1 & 3 Endemic countries Re-established transmission countries Case or outbreak following importation *Excludes
Wild Poliovirus*, 03 Aug 2004 to 02 Aug 2005 Wild virus type 1 Wild virus type 3 Wild virus type 1 & 3 Endemic countries Re-established transmission countries Case or outbreak following importation *Excludes
HEV Assay Development Update
 HEV Assay Development Update Jeffrey M. Linnen, Ph.D. Senior Director, Research & Development Hologic Gen-Probe, San Diego, California USA IPFA/PEI 20th International Workshop on Surveillance and Screening
HEV Assay Development Update Jeffrey M. Linnen, Ph.D. Senior Director, Research & Development Hologic Gen-Probe, San Diego, California USA IPFA/PEI 20th International Workshop on Surveillance and Screening
Routine NAT-screening for West Nile Virus Infections in Germany: Being prepared
 Routine NAT-screening for West Nile Virus Infections in Germany: Being prepared R. Thermann, W. K. Roth IPFA / PEI 18 th international workshop on Surveillance and screening of blood borne pathogens Budapest,
Routine NAT-screening for West Nile Virus Infections in Germany: Being prepared R. Thermann, W. K. Roth IPFA / PEI 18 th international workshop on Surveillance and screening of blood borne pathogens Budapest,
Flu Watch. MMWR Week 3: January 14 to January 20, and Deaths. Virologic Surveillance. Influenza-Like Illness Surveillance
 Flu Watch MMWR Week 3: January 14 to January 2, 218 All data are provisional and subject to change as more reports are received. Geographic Spread South Carolina reported widespread activity this week.
Flu Watch MMWR Week 3: January 14 to January 2, 218 All data are provisional and subject to change as more reports are received. Geographic Spread South Carolina reported widespread activity this week.
West Nile Virus in Maricopa County
 West Nile Virus in Maricopa County Maricopa County Department of Public Health Office of Epidemiology July 21 January 1, 29 December 31, 29 Commentary West Nile virus (WNV) is a mosquito-borne virus that
West Nile Virus in Maricopa County Maricopa County Department of Public Health Office of Epidemiology July 21 January 1, 29 December 31, 29 Commentary West Nile virus (WNV) is a mosquito-borne virus that
IMPLEMENTING RECOVERY ORIENTED CLINICAL SERVICES IN OPIOID TREATMENT PROGRAMS PILOT UPDATE. A Clinical Quality Improvement Program
 IMPLEMENTING RECOVERY ORIENTED CLINICAL SERVICES IN OPIOID TREATMENT PROGRAMS PILOT UPDATE A Clinical Quality Improvement Program Today: National completion rates for OTP s hover between 11 14% Retention
IMPLEMENTING RECOVERY ORIENTED CLINICAL SERVICES IN OPIOID TREATMENT PROGRAMS PILOT UPDATE A Clinical Quality Improvement Program Today: National completion rates for OTP s hover between 11 14% Retention
Adult Immunizations. Business Health Care Group (BHCG) April 25, Cathy Edwards. Immunization Program Advisor
 Adult Immunizations Business Health Care Group (BHCG) April 25, 2012 Cathy Edwards Immunization Program Advisor Wisconsin Department of Health Services Division of Public Health 1 Adult Immunizations WHY
Adult Immunizations Business Health Care Group (BHCG) April 25, 2012 Cathy Edwards Immunization Program Advisor Wisconsin Department of Health Services Division of Public Health 1 Adult Immunizations WHY
Cost-Effectiveness of Lung Volume Reduction Surgery
 Cost-Effectiveness of Lung Volume Reduction Surgery The Health Industry Forum October 24, 2007 - Washington DC The National Emphysema Treatment Trial National Emphysema Treatment Trial (NETT) Multicenter,
Cost-Effectiveness of Lung Volume Reduction Surgery The Health Industry Forum October 24, 2007 - Washington DC The National Emphysema Treatment Trial National Emphysema Treatment Trial (NETT) Multicenter,
West Nile Virus in Maricopa County
 West Nile Virus in Maricopa County A Culex quinquefasciatus mosquito on a human finger. Image by James Gathany/ CDC gov/ public domain Maricopa County Department of Public Health Office of Epidemiology
West Nile Virus in Maricopa County A Culex quinquefasciatus mosquito on a human finger. Image by James Gathany/ CDC gov/ public domain Maricopa County Department of Public Health Office of Epidemiology
Durham Region Influenza Bulletin: 2017/18 Influenza Season
 Durham Region Influenza Bulletin: 2017/18 Influenza Season Surveillance Week 21 (May 20, 2018 to May 26, 2018) Table 1: Assessment of influenza activity in Durham Region Measure Laboratory confirmed cases
Durham Region Influenza Bulletin: 2017/18 Influenza Season Surveillance Week 21 (May 20, 2018 to May 26, 2018) Table 1: Assessment of influenza activity in Durham Region Measure Laboratory confirmed cases
