DIAGNOSIS OF INTESTINAL AMEBIASIS USING SALIVARY IgA ANTIBODY DETECTION
|
|
|
- Silvester Nicholson
- 5 years ago
- Views:
Transcription
1 DIAGNOSIS OF INTESTINAL AMEBIASIS USING SALIVARY IgA ANTIBODY DETECTION Paibul Punthuprapasa 1, Nitaya Thammapalerd 2, Udomporn Chularerk 3, Kravee Charoenlarp 4 and Manoon Bhaibulaya 1 1 Department of Parasitology and 4 Department of Medicine, Faculty of Medicine Siriraj Hospital; 2 Department of Microbiology and Immunology, Faculty of Tropical Medicine; 3 Department of Parasitology, Faculty of Public Health, Mahidol University, Bangkok, Thailand Abstract. Attempts were made to use soluble antigen extract of strain HK-9 of Entamoeba histolytica to detect salivary IgA antibodies in intestinal amebiasis patients by using ELISA. Total salivary samples of 109 individuals were divided into four groups. Group I comprised 32 patients whose stools were positive only for E. histolytica cysts and/or trophozoites. Group II comprised 12 individuals whose stools were positive for E. histolytica and other intestinal parasites. Group III comprised 36 individuals whose stools were negative for E. histolytica but contained other intestinal parasites such as E. coli, E. nana, Blastocystis hominis, Trichomonas hominis, Giardia lamblia, Opisthorchis viverrini, and hookworm. Group IV comprised 29 healthy individuals whose stools were free from any intestinal parasitic infections. Based on the mean optical density, OD + 2SD of the results from 29 parasitologically negative healthy individuals, the cut-off OD value for salivary IgA antibodies was Therefore, the assays were positive in 14 out of 32 (43.75%) of group I and 2 out of 12 (16.6%) of group II. The assays were positive in 16 out of 36 (44.44%) for group III whereas 2 out of 29 (6.90%) for group IV were positive. The overall sensitivity and specificity of the assays were 36% and 72%, respectively. The false positive rate was 28% and the false negative rate was 64%. The predictive values of positive and negative results were 47% and 63%, respectively. The diagnostic accuracy of ELISA for the presence of salivary IgA antibodies was 58%. INTRODUCTION Entamoeba histolytica is a protozoan parasite that infects the large intestine of man. The disease caused by this protozoan is called amebiasis and has a worldwide distribution. The prevalence rate is high in some tropical regions and is a public health problem in many developing countries such as Mexico, India, countries of northern and southern Africa, and Southeast Asia (Elsdon-Dew, 1968). It has been estimated that 10% of the world s population is infected with E. histolytica and that about 36 million suffering from amebic colitis and extraintestinal amebiasis. At least 40,000 to 100,000 deaths are attributable to amebiasis every year (Walsh, 1986). Amebic abscesses are found in the liver, lung and brain (Merritt et al, 1982). Many individuals habor the parasites and pass cysts without any signs of illness, while others manifest symptoms ranging from mild diarrhea to fulminant, bloody mucous dysentery and amebic liver abscess. Correspondence: Assoc Prof Nitaya Thammapalerd, Department of Microbiology and Immunology, Faculty of Tropical Medicine, Mahidol University, 420/6 Rajvithi Road, Bangkok 10400, Thailand. Tel: +66 (0) Ext 1591, 1592 Fax: +66 (0) tmntm@mahidol.ac.th Thailand, one of the tropical countries, is regarded as an endemic area in which the survey study of Department of Parasitology, Faculty of Public Health, Mahidol University during found the parasite all over the country. The prevalence rate was between % depending on areas and socioeconomic status (Harinasuta and Charoenlarp, 1971; Chullaruk, 1977; Bunnag et al, 1982). Due to lacking a sanitary standard system and health education during that time period, the people have opportunity to acquire this parasite from contaminated food and drinking water. Although the rate of infection is quite low but it is the cause of potential lost of life. Metronidazole, a good effective drug can treat amebiasis efficiently (Maltz and Knauer, 1991). However, the treatment is always done after the obvious symptom have been shown which indicated that the patient is in an invasive stage. One of the traditional methods in circulating antibody determination is serological assay techniques. Serological assays use many methods such as immunoelectrophoresis (IEP) (Savanat and Chaicumpa, 1969) where circulating antibody in patients with liver abscess and acute diarrhea could be detected for 85-97% and 66-80%, respectively, complement fixation (CF) (Jatinandana and Tharavanij, 1975), latex agglutination (LA) (Savanat et al, 1974), indirect hemagglutination (IHA) Vol 32 (Suppl 2)
2 SOUTHEAST ASIAN J TROP MED PUBLIC HEALTH (Soavana et al, 1981) and enzyme-linked immunosorbent assay (ELISA) (Samrejrongroj and Tharavanij, 1985), cellulose acetate precipitating (CAP) test (Thammapalerd et al, 1981). However, there are differences with each method (Tharavanij, 1975; Voller and De Savigny, 1981). Account for the fact that most of the circulating antibody type is immunoglobulin-g (IgG), the result of the examination may be false positive although the disease has been eradicated at that time, IgG still exist for a long time. The examination result, also, could be false negative if the antibody system of patient is deficient. (Healy, 1986). The weak points of the serological assay technique mentioned above can mislead in treatment. Detection of amebic antigen is the method of choice to assess recent infection and test of cure after successful treatment as has been shown clearly in feces of all 13 patients haboring E. histolytica trophozoites by MAb-PAb-based ELISA (Thammapalerd and Thararanij, 1991; Wonsit et al, 1992), circulating amebic antigens in infected hamsters (Mesocricetus auratus) (Thammapalerd et al, 1996) and intrahepatic localization of trophozoite antigens by MAb-based IFA and MAb-based immunoperoxidase (Ipx) tests (Sherchand et al, 1994). The precision examination for treatment should be the methods that can show the real causes of sickness. Another one method, antiamebic antibody examination of secretory IgA (siga) type to detect intestinal amebiasis caused by E. histolytica, has been reported by del Muro et al (1990). The siga antibody is local secretory immune response as an indicator of infectious diseases affecting mucosal sites. It can prevent the attachment of E. histolytica in trophozoites stage on intestinal wall which is the first step before invasive process and it is the important cause of intestinal amebiasis. (Leyva et al, 1992). It is, therefore, the objective of this study to attempt to use soluble antigen of E. histolytica for detecting secretory IgA antibody in saliva of patients with intestinal amebiasis. MATERIALS AND METHODS Subjects There are four groups of subjects whose stools were examined for parasites, when positive for E. histolytica either trophozoites and/or cysts, or negative as control group, their saliva were then collected. Group I. Clinical specimens from amebiasis patients: Stool samples from patients with symptomatic and/or asymptomatic amebiasis were obtained from wards of Siriraj Hospital; laboratory unit of the Bangkok Hospital for Tropical Diseases, Faculty of Tropical Medicine, Mahidol University and from stool surveying at Kog Sabang village, Thai- Cambodian border, Sakaew Province. These stool samples were examined by a single fresh smear technique. The examination revealed cysts and/or hemophagous trophozoites of E. histolytica, Thirtytwo stool samples positive for only E. histolytica were classified in this group and their corresponding saliva were collected. Group II. The specimens from patients infected with E. histolytica and other parasites: This group comprised 12 patients whose stools were positive with E. histolytica and other parasites, and their saliva were collected accordingly. Group III. The specimens from people infected by parasites other than E. histolytica: Thirty-six saliva were collected from people who lived at Kog Sabang village, Thai-Cambodian border, whose stool examinations were negative for E. histolytica but contained other intestinal parasites. Group IV. Healthy controls: Twenty-nine saliva were obtained from volunteers whose stool examinations were free from any parasites. Collection and preparation of saliva specimens Five ml of nonstimulated saliva was obtained from each group of the subjects. The saliva was collected into the cylindrical plastic box, 3x5 cm in size, and centrifuged at 13,000 rpm 10 minutes and the supernatant was frozen at -20 C until used. Preparation of E. histolytica antigen Antigen was prepared from axenically grown of E. histolytica strain HK-9. The amebae were maintained axenically in TPS-1 medium according to the technique described by Diamond (1968). The medium consists of a nutrient broth supplemented with 2.5% vitamin mixture and 10% heat inactivated horse serum. Subculture was done at hours of incubation at 37 C. The amebae were washed 3 times in physiological saline by centrifugation at 1,000 rpm for 5 minutes at room temperature. The ameba suspension was chilled and disrupted by an ultrasonic disintegrator (M and SE Ltd, London.) for 3-5 minutes. Total disruption of the ameba was ascertained by examination under a light microscope. The disrupted ameba suspension was centrifuged at 3,000 rpm for 10 minutes. The opalescent supernatant was distributed in small aliquots, lyophilized and kept at -20 C for being used as the antigen. The protein content of antigen was determined by the method of Lowry et al (1951) using bovine serum albumin as a standard. 160 Vol 32 (Suppl 2) 2001
3 DIAGNOSIS OF AMEBIASIS Enzyme-linked immunosorbent assay (ELISA) for anti-amebic salivary IgA detection The indirect ELISA was done on 96-wells flat bottomed polyvinyl microtiter plate (Nunc-Immuno Plate) as described previously (Tharavanij, 1975). The conjugates were horseradish peroxidase lebeled goatantihuman IgA conjugated (A-07032, Sigma). The wells of the Nunc polyvinyl microtiter plate were precoated with 100 μl of amoebic antigen in a dilution of 5 μg/ml in carbonate buffer 0.1M, ph 9.6 and incubated at 37 C for 1 hour and overnight at 4 C in refrigerator, usually 18 to 20 hours. The unbound antigens were washed away excessively three times with phosphate buffer saline (PBS), ph 7.2, containing 0.05% Tween 20 (PBS-T). After washing the unbound surfaces were saturated with PBS-T containing 5% non-fat dry milk. The plate was incubated at 37 C for 1 hour and washed as above. After three additonal washing, 100 μl of undiluted clarified saliva was added into the wells. The plate was further incubated for 2 hours at 37 C, and then washed. One hundred μl of diluted horseradish peroxidase labeled goat anti-human IgA conjugate (1:1,000 dilution in PBST-0.1% BSA), was added to each well and the plate incubated at 37 C for 1 hour. The wells were washed and 100 μl of the freshly prepared substrate (orthophenylenediamine dihydrochloride : OPD in phosphate-critrate buffer, ph 5.0) were added, the reaction was allowed to occur at 37 C. After 30 minutes, the enzyme reaction was stopped by adding 50 μl of 12.5% H 2 SO 4 into each well. The optical density (OD), represented the substrate conversion by the enzyme was measured by ELISA reader (Titertek Multiskan, MCC/340, Flow Laboratories) at 492 nm. RESULTS ELISA for salivary IgA anti-amebic antibody detection The cut-off OD value was obtained by taking 2SD (2 x 0.588) above mean value (0.677) of healthy subjects. Thus, the test was considered positive with OD of or above. Therefore, with the cut off value of OD (Mean + 2SD), the assay was positive only 16 out of 44 cases (36%) in groups I and II. In 65 of stools negative examination for E. histolytica, 20 out of 36 cases (56%) in group III and 27 out of 29 (93%) in group IV were negative by ELISA, respectively (Fig 1). There were other parasites present in three E. histolytica positive stool samples of group II, one each of E. coli, B. hominis and H. nana, and 6 samples of O. viverrini. There were mixed parasitic infections in 2 samples with A. lumbricoides, T. trichiura ova and O.viverrini, S. stercoralis, and hookworm respectively. In group III, 36 stool samples were negative for E. histolytica but contained other parasites as follow: E. coli, E. nana, B. hominis, T. hominis, G. lamblia, O. viverrini, hookworm and intestinal fluke. Comparison of salivary IgA ELISA and microscopic stool examination for diagnosis of intestinal amebiasis The salivary IgA detection was compared to microscopic stool examination. It was found that IgA antibodies to E. histolytica were present in saliva samples from 36% (16 of 44) of cases infected with this parasite. The overall sensitivity of IgA ELISA was 36% (16 of 44) and specificity 72% (47 of 65). The false positive rate was 28% (18 of 65) and the false negative 64% (28 of 44). The predictive value of a positive ELISA was 47% (16 of 34) and the predictive value of a negative ELISA was 63% (47 of 75). The diagnostic accuracy for the presence of salivary IgA antibodies to E. histolytica was 58% (63 of 109) (Table 1). Optical Density (492 nm) E.h (gr I) (32) E.h+OP (gr II) (12) OP (gr III) (36) NP (gr IV) (29) Cut-off Fig 1- Scatter diagram of the optical density (OD) and cutoff OD from indirect ELISA for detecting siga antibodies in saliva samples. gr I : Stool positive only for E. histolytica trophozoites. gr II : Stool positive for E. histolytica and other intestinal parasites. gr III : Stool negative for E. histolytica but containing other intestinal parasites. gr IV : Stool negative for all parasites. ( ) : Total subjects in each group. Vol 32 (Suppl 2)
4 SOUTHEAST ASIAN J TROP MED PUBLIC HEALTH Table 1 Comparison of salivary IgA ELISA and microscopic stool examination for diagnosis of E. histolytica Tested by IgA ELISA Positive for Negative for Total E. histolytica by E. histolytica by microscopic stool microscopic stool examination examination Positive for E.histolytica by siga ELISA Negative for E.histolytica by siga ELISA Total DISCUSSION Traditionally, the diagnosis of amebic infection is based on stool examination or specific antibodies detection. Both methods have several inherent problems. Microscopic examination of stool samples is tedious, time-consuming and dependent on a high degree of skill. The cystic stages could not be used to distinguish between E. histolytica and E. dispar. The most accurate method for diagnosis of invasive amebiasis is microscopic examination, showing hematophagous E. histolytica trophozoites. Another group of individuals, so-called asymptomatic cyst passers may be E. dispar which are an important group in transmission of the parasite, are not detected by such methods. There have been cases of misidentification of E. histolytica cysts, confusing them not only with cysts from other, non-pathogenic ameba, but also with lymphocytes and other cells found in stool samples (Neal, 1988). There are many serological tests for amebic diagnosis, including the indirect hemagglutination (IHA), latex agglutination (LA), counterimmunoelectrophoresis (CIEP), gel diffusion (GD) and ELISA. These serological tests are particularly useful in the detection of extraintestinal amebiasis such as amebic liver abscess, since in such cases stool examination is often negative for E. histolytica. Since positive antibody titers may persist for months or years after successful treatment, it was therefore, difficult to differentiate between present and past infection (Krupp and Powell, 1971). The detection of salivary IgA antibodices to E. histolytica for diagnosis of intestinal amebiasis has been described (del Muro et al, 1990). The membrane antigen of E. histolytica strain HM1:IMSS was used for plate coating. The sensitivity and specificity of the assays were 85% and 98% respectively. The diagnostic accuracy of salivary IgA antibodies was 91%. Therefore, salivary sampling may be a useful diagnosis of intestinal amebiasis. ELISA established in this study demonstrated the possibility of using soluble antigen preparation from trophozoite extracted of axenic culture of E. histolytica strain HK-9, and peroxidase-labeled goat anti-human IgA conjugate (A-7032, Sigma) to detect anti-amebic IgA antibody in saliva from amebiasis patients. The positive results are those greater than absorbance units (see Fig 1). Positive ELISA results in which microscopic stool examination were positive for E. histolytica, were 36% (16 of 44). The failure of the ELISA to detect the IgA in other specimens (28/44) may be due to either the amount of antibody below the sensitivity of the assay or recently acquired infection in which the antibody response has not yet appreared. In addition, the detecting antigens might not recognize the antibody in these specimens because of their loss in the antigenicity for specific siga to E. histolytica after long-term storage and repeated freezing and thawing (Root et al, 1978). Moreover, it was possible that the false-positive of microscopic examination may be due to misidentification between E. histolytica, E. dispar and E. hartmanni or other nonpathogenic amebae such as Iodamoeba butschlii as well as white blood cells (Neal, 1988). The siga ELISA results were positive in 18 out of 65 subjects without demonstrable E. histolytica in stools. Therefore, the false positive rate was as high as 28%. The problem might be due to the assays detecting residual IgA antibodies. In addition, the subjects studied were mostly from the Thai-Cambodian border where a high parasitic load and a high risk of infection were expected (Kataitong, 1994). Under these conditions, intestinal parasites in constant contact with the host s tissue may elicit a secretory immune response even in the absence of invasiveness (Mestecky et al, 1978). In human, IgA antibodies appeared in external secretion about two weeks after presentation of bacterial antigens to the gut associated lymphoid tissue (Czerkinsky et al, 1987) and that will decrease over 45 days after treatment. Furthermore, it was possible that the examination of stool samples might miss the 162 Vol 32 (Suppl 2) 2001
5 DIAGNOSIS OF AMEBIASIS parasites from collection since the parasites might be variably excreted or unevenly ditributed in the stool. Marsden and Smith (1946) reported the probability of diagnosis based on one direct microscopic stool examination as 50% in a person excreting 100,000 cysts daily or 0.45% in a person excreting only 1,000 cysts a day. The IgA ELISA was found in a salivary sample from one child (OD 1.636) whose stool was negative for all parasites. The false-positive results could be attributed to missing diagnosis of E. histolytica in his stool. Unfortunately, we could not follow up this case for further study. The assay was less satisfactory because of the overall sensitivity and specificity of the IgA ELISA in this study was 36% and 72% respectively. The false positive rate was 28% and the false negative rate was 64%. The predictive value of a positive ELISA was 47% and the predictive value of a negative ELISA was 63%. The diagnostic accuracy for the presence of salivary IgA antibodies to E. histolytica was 58%. Therefore, the membrane antigens were more appropriate than those of soluble antigens for the diagnosis of salivary IgA antibodies in intestinal amebiasis. The presence of secretory immunoglobulin A (siga) anti-e. histolytica antibodies in the saliva of patients with intestinal amebiasis was demonstrated by immunoblot assay, and the capacity of these antibodies to inhibit amebic adherence to a monolayer of MDCK cells was analyzed. (Carrero et al, 1994). Inhibition was due to IgA antiamebic antibodies and in part to anti-gal-binding-lectin antibodies, as demonstrated by absorption experiments with total amebic extract and with the fraction of Gal-binding lectin (Carrero et al, 1994). These results emphasize the relevance of secretory IgA antibodies in the phenomenon of E. histolytica adherence to epithelial cells. The E. histolytica surface antigens capable of inducing secretory IgA (siga) responses in humans have been cloned and sequenced (Carrero et al, 2000). A cdna library from the strain HM1:IMSS was immuno-screened with saliva from patients with intestinal amebiasis or amebic liver abscess. Clones isolated with siga antibodies from patients with intestinal amebiasis corresponded to the known serinerich protein isoform, a 29 kda cysteine-rich protein and 1-alpha elongation factor. Clones corresponding to enolase, cyclophilin, ribosomal protein L23a, and an Hsp70 family protein were isolated with siga from a patient with amebic liver abscess. A glutamic acidrich peptide (EhGARP) positive with siga from a patient with amebic liver abscess was also isolated; for EhGARP, no homologs were found in the protein databases (Carrero et al, 2000). The antigens isolated are potentially useful in the development of an oral vaccine or new diagnostic tools for amebiasis. ACKNOWLEDGEMENTS This study was financially supported by grants from the Faculty of Graduate Studies, Mahidol University Bangkok, Thailand. The authors would like to thank all the personnel who helped in collecting some clinical specimens used in this study. REFERENCES Bunnag T, Klongkamnuankarn K, Thirachandra S, et al. Seroepidemiology of amoebiasis in the villagers in Pichit Province and urban slum dwellers in Bangkok, Thailand. Southeast Asain J Trop Med Public Health 1982;13: Carrero JC, Diaz MY, Vivoros M, et al. Human secretory immunoglobulin A anti-entamoeba histolytica antibodies inhibit adherence to MDCK cells. Infect Immun 1994;62: Carrero JC, Petrossian P, Acosta E, et al. Cloning and characterization of Entamoeba histolytica antigens recognized by human secretory IgA antibodies. Parasitol Res 2000;86: Chullaruk U. Socio-economic status and parasitic diseases. J Trop Med Assoc Thai 1977;1: Czerkinsky C, Prince SJ, Michalek SM, et al. IgA antibody producing cells in peripheral blood after antigen ingestion : evidence for a common mucosal immune system in humans. Proc Natl Acad Sci USA 1987;84: Del Muro R, Acosta E, Merino E, et al. Diagnosis of intestinal amebiasis using salivary IgA antibody detection. J Infect Dis 1990;162: Diamond LS. Techniques of axenic cultivation of Entamoeba histolytica Schaudinn, 1903 and E. histolytica like amoebae. J Parasitol 1968;54: Elsdon-Dew R. The epidemiology of amoebiasis. Adv Parasitol 1968;6:1-62. Harinasuta T, Charoenlarp P. Parasitic infections of the gastrointestinal tract in Thailand, A Review. In: Cross JM, ed. Proceedings of the 7th Southeast Asian Regional Seminar on Tropical Medicine and Public Health. Infectious Disease of the Gastrointestinal System in Southeast Asian and the Far East, Taipei 1971: Vol 32 (Suppl 2)
6 SOUTHEAST ASIAN J TROP MED PUBLIC HEALTH Healy GR. Immunologic tools in the diagnosis of amebiasis, epidemiology in the United States. Rev Infect Dis 1986;8: Jatinandana V, Tharavanij S. Antigenicity of subcellular components of Entamoeba histolytica. Southeast Asian J Trop Med Public Health 1975;6: Kataitong K. Opisthorchis, comparative study on the efficiency of the fecal examination techniques. Bangkok: Faculty of Graduate Studies, Mahidol University, 1994: Msc Thesis. Krupp I, Powell SJ. Comparative study of the antibody response in amoebiasis: Persistence after successful treatment. Am J Trop Med Hyg 1971;20: Leyva O, Rico G, Ramos F, et al. Inhibittion of adhesion process mediated by anti-entamoeba histolytica specific monoclonal IgA antibodies. Arch Med Res 1992;23: Lowry OH, Rosebrough NJ, Farr AL, et al. Protein measurement with Folin phenol reagent. J Biol Chem 1951;193: Maltz G, Knauer CM. Amebic liver abscess: a 15 year experience. Am J Gastroenterol 1991;86: Marsden APH, Smith HF. The detection of the cysts of E. histolytica in the feces by microbiology examination. Med J Aust 1946;1: Merritt R, Coughlin E, Thomas D, et al. Spectrum of amoebiasis in children. Am J Dis Child 1982;136: 785. Mestecky J, McGhee JR, Arnold RR, et al. Selective induction of an immune response in human external secretions by ingestion of bacteria antigen. J Clin Invest 1978;61: Neal RA. Phylogeny: the relationship of Entamoeba histolytica to morphologically similar amebae of the four-nucleated cyst group. In: Ravdin JI, ed. Amoebiasis: human infection by Entamoeba histolytica. New York: Churchill Livingstone, 1988: Root DM, Cole FX, Williamson JA. The development and standardization of an ELISA method for the detection of Entamoeba histolytica antigens in fecal samples. Arch Invest Med (Mex) 1978;9: Samrejrongroj P, Tharavanij S. Assessment of validity of counter immunoelectrophoresis and ELISA in the routine diagnosis of amoebiasis. Southeast Asian J Trop Med Public Health 1985;16: Savanat T, Chaicumpa W. Immunoelectrophoresis test for amoebiasis. Bull WHO 1969;40: Savanat T, Tantivanich S, Klonglamnuankarn K. Latex agglutinations test for amoebiasis. J Med Assoc Thai 1974;57: Sherchand JB, Thammapalerd N, Riganti M, et al. Monoclonal antibodies-based immunohistochemical demonstration of Entamoeba histolytica in liver tissues of experimentally infected hamster (Mesocricetus auratus). Int J Parasitol 1994;24: Soavana T, Tharavanij S, Thammapalerd N, et al. Leucocyte migration agarose test to study cellmediated immunity in amoebiasis. Southeast Asian J Trop Med Public Health 1981;12: Thammapalerd N, Sherchand JB, Kotimanusvanij D, et al. Monoclonal antibody-based ELISA for the detection of circulating Entamoeba histolytica antigen in hepatic amebiasis in hamsters (Mesocricetus auratus). Southeast Asian J Trop Med Public Health 1996;27: Thammapalerd N, Tharavanij S. Biomedical application of monoclonal antibodies (McAb) against Entamoeba histolytica. In: Maramorosch K, ed. Biotechnology for Biological Control of Pests and Vectors. Florida: CRC Press. 1991; Thammapalerd N, Tharavanij S, Samrejrongroj P. Cellulose acetate membrane preciptin (CAP) test in amoebiasis using antigens from four different strains of Entamoeba histolytica. Southeast Asian J Trop Med Public Health 1981;12: Tharavanij S. Immunological tests in amoebiasis. In: Harinasuta C, Reynolds DC, eds. SEAMEO- TROPMED Technical Meeting : Diagnostic methods for important helminthiasis and amoebiasis in Southeast Asia and the Far East. Tokyo; SEAMEO-TROPMED Project. Bangkok: Thai Watana Punich Press, 1975:1-25. Voller A, De Savigny D. Diagnostic serology of tropical parasitic diseases. J Immunol Methods 1981;46:1-29. Walsh J. Problems in the recognition and diagnosis of amoebiasis : estimation of the global magnitude of morbidity and mortality. Rev Infect Dis 1986; 8: Wonsit R, Thammapalerd N, Tharavanij S, et al. Enzyme-linked immunosorbent assay based on monoclonal and polyclonal antibodies for the detection of Entamoeba histolytica antigens in faecal specimens. Trans R Soc Trop Med Hyg 1992;86: Vol 32 (Suppl 2) 2001
E. Histolytica IgG (Amebiasis)
 DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and 116, Woodland hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and 116, Woodland hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
PARASITOLOGY CASE HISTORY 15 (HISTOLOGY) (Lynne S. Garcia)
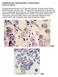 PARASITOLOGY CASE HISTORY 15 (HISTOLOGY) (Lynne S. Garcia) A biopsy was performed on a 27-year-old man with no known travel history, presenting with a perianal ulcer. The specimen was preserved in formalin
PARASITOLOGY CASE HISTORY 15 (HISTOLOGY) (Lynne S. Garcia) A biopsy was performed on a 27-year-old man with no known travel history, presenting with a perianal ulcer. The specimen was preserved in formalin
E. Histolytica IgG ELISA Kit
 E. Histolytica IgG ELISA Kit Catalog Number KA3193 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
E. Histolytica IgG ELISA Kit Catalog Number KA3193 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
Entamoeba histolytica
 Entamoeba histolytica cosmopolitan distribution no animal reservoirs facultative pathogen most clear the infection spontaneous in 6-12 months with mild or no symptoms can cause a serious invasive disease
Entamoeba histolytica cosmopolitan distribution no animal reservoirs facultative pathogen most clear the infection spontaneous in 6-12 months with mild or no symptoms can cause a serious invasive disease
Parasitology. Lab. Amoeba
 Parasitology. Lab. Kingdom : Protista Subkingdom : Protozoa Phylum : Sacromastigophora Subphylum : Sarcodina Superclass : Rhizopoda Class : Lobosea Order : Amoebida Amoeba Protozoa Amoebae geneus Entamoeba
Parasitology. Lab. Kingdom : Protista Subkingdom : Protozoa Phylum : Sacromastigophora Subphylum : Sarcodina Superclass : Rhizopoda Class : Lobosea Order : Amoebida Amoeba Protozoa Amoebae geneus Entamoeba
A CROSS-SECTIONAL STUDY OF INTESTINAL PARASITIC INFECTIONS AMONG SCHOOLCHILDREN IN NAN PROVINCE, NORTHERN THAILAND
 SOUTHEAST ASIAN J TROP MED PUBLIC HEALTH A CROSS-SECTIONAL STUDY OF INTESTINAL PARASITIC INFECTIONS AMONG SCHOOLCHILDREN IN NAN PROVINCE, NORTHERN THAILAND J Waikagul 1, S Krudsood 2, P Radomyos 3, B Radomyos
SOUTHEAST ASIAN J TROP MED PUBLIC HEALTH A CROSS-SECTIONAL STUDY OF INTESTINAL PARASITIC INFECTIONS AMONG SCHOOLCHILDREN IN NAN PROVINCE, NORTHERN THAILAND J Waikagul 1, S Krudsood 2, P Radomyos 3, B Radomyos
Catalog # OKDA00123 SUMMARY AND EXPLANATION
 Aviva Systems Biology 5754 Pacific Center Blvd., Suite 201, San Diego, CA 92121, USA Tel: 858-552-6979 Fax: 858-552-6975 www.avivasysbio.com Email: info@avivasysbio.com Giardia lamblia ELISA Kit Catalog
Aviva Systems Biology 5754 Pacific Center Blvd., Suite 201, San Diego, CA 92121, USA Tel: 858-552-6979 Fax: 858-552-6975 www.avivasysbio.com Email: info@avivasysbio.com Giardia lamblia ELISA Kit Catalog
Entamoeba histolytica/e. dispar. A. Haghighi,
 Entamoeba histolytica/e. dispar A. Haghighi, Wednesday, February 14, 2018 Classification of Protozoa? The protozoa are generally unicellular and may be divided for convenience, into four distinct groups
Entamoeba histolytica/e. dispar A. Haghighi, Wednesday, February 14, 2018 Classification of Protozoa? The protozoa are generally unicellular and may be divided for convenience, into four distinct groups
Bacillary Dysentery (Shigellosis)
 Bacillary Dysentery (Shigellosis) An acute bacterial disease involving the large and distal small intestine, caused by the bacteria of the genus shigella. Infectious agent Shigella is comprised of four
Bacillary Dysentery (Shigellosis) An acute bacterial disease involving the large and distal small intestine, caused by the bacteria of the genus shigella. Infectious agent Shigella is comprised of four
Giardia lamblia ELISA Kit
 Giardia lamblia ELISA Kit Catalog Number KA2093 96 assay Version: 01 Intend for research use only www.abnova.com Introduction and Background A. Intended Use Giardia lamblia ELISA Kit is an Enzyme-Linked
Giardia lamblia ELISA Kit Catalog Number KA2093 96 assay Version: 01 Intend for research use only www.abnova.com Introduction and Background A. Intended Use Giardia lamblia ELISA Kit is an Enzyme-Linked
CHAPTER 4 IMMUNOLOGICAL TECHNIQUES
 CHAPTER 4 IMMUNOLOGICAL TECHNIQUES Nitroblue Tetrazolium Chloride (NBT) Reduction test NBT reduction test was evaluated by employing the method described by Hudson and Hay,1989 based upon principle that
CHAPTER 4 IMMUNOLOGICAL TECHNIQUES Nitroblue Tetrazolium Chloride (NBT) Reduction test NBT reduction test was evaluated by employing the method described by Hudson and Hay,1989 based upon principle that
Stitaya Sirisinha, Runglawan Chawengkirttikul.and. Department of Microbiology, Faculty of Science, Mahidol University, Bangkok 10400, Thailand.
 Stitaya Sirisinha, Runglawan Chawengkirttikul.and Rasana Sennswan Department of Microbiology, Faculty of Science, Mahidol University, Bangkok 10400, Thailand. Abstract. Monoclonal antibody-based enzyme-linked
Stitaya Sirisinha, Runglawan Chawengkirttikul.and Rasana Sennswan Department of Microbiology, Faculty of Science, Mahidol University, Bangkok 10400, Thailand. Abstract. Monoclonal antibody-based enzyme-linked
RIDA QUICK Entamoeba. Article no: N1702
 RIDA QUICK Entamoeba Article no: N1702 R-Biopharm AG, An der neuen Bergstraße 17, D-64297 Darmstadt, Germany Phone: +49 (0) 61 51 81 02-0 / Fax: +49 (0) 61 51 81 02-20 1. Intended use For in vitro diagnostic
RIDA QUICK Entamoeba Article no: N1702 R-Biopharm AG, An der neuen Bergstraße 17, D-64297 Darmstadt, Germany Phone: +49 (0) 61 51 81 02-0 / Fax: +49 (0) 61 51 81 02-20 1. Intended use For in vitro diagnostic
ccess safe drinking wa r is everyone s right Protozoans that cause diarrheal disease
 ccess safe drinking wa r is everyone s right Protozoa: Protozoans that cause diarrheal disease 1. Giardia lamblia 2. Entameba histolytica 3. Cryptosporidium parvum 4. Cyclospora cayetanensis 1 Giardia
ccess safe drinking wa r is everyone s right Protozoa: Protozoans that cause diarrheal disease 1. Giardia lamblia 2. Entameba histolytica 3. Cryptosporidium parvum 4. Cyclospora cayetanensis 1 Giardia
Cryptosporidium spp. ELISA Kit
 Cryptosporidium spp. ELISA Kit Catalog Number KA2094 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Cryptosporidium spp. ELISA Kit Catalog Number KA2094 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Trichinella Cat #
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Ameba has two stages of development: cyst and trophozoite
 Amebiasis A parasitic disease of worldwide public health importance Second to malaria in mortality due to protozoan parasites Invasive amebiasis results in up to 100,000 deaths / year Amebiasis is infection
Amebiasis A parasitic disease of worldwide public health importance Second to malaria in mortality due to protozoan parasites Invasive amebiasis results in up to 100,000 deaths / year Amebiasis is infection
RIDA QUICK Entamoeba. Article no: N 1703
 RIDA QUICK Entamoeba Article no: N 1703 R-Biopharm AG, Landwehrstr. 54, D-64293 Darmstadt, Germany Tel: +49 (0) 61 51 81 02-0 / Fax: +49 (0) 61 51 81 02-20 1. Intended use For in vitro diagnostic use.
RIDA QUICK Entamoeba Article no: N 1703 R-Biopharm AG, Landwehrstr. 54, D-64293 Darmstadt, Germany Tel: +49 (0) 61 51 81 02-0 / Fax: +49 (0) 61 51 81 02-20 1. Intended use For in vitro diagnostic use.
Amebiasis Ab E. histolytica IgG
 Code: HP003 96 Test distribuito da: Amebiasis Ab E. histolytica IgG Amebiasis E. histolytica lgg test is an enzyme immunoassay for the detection of specific lgg antibodies against Anti - Entamoeba histolytica
Code: HP003 96 Test distribuito da: Amebiasis Ab E. histolytica IgG Amebiasis E. histolytica lgg test is an enzyme immunoassay for the detection of specific lgg antibodies against Anti - Entamoeba histolytica
Alberta Health and Wellness Public Health Notifiable Disease Management Guidelines August 2011
 August 2011 Amoebiasis Revision Dates Case Definition Reporting Requirements Remainder of the Guideline (i.e., Etiology to References sections inclusive) Case Definition August 2011 August 2011 October
August 2011 Amoebiasis Revision Dates Case Definition Reporting Requirements Remainder of the Guideline (i.e., Etiology to References sections inclusive) Case Definition August 2011 August 2011 October
Crypto / Giardia Ag Combo (Fecal) ELISA
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Please use only the valid version of the package insert provided with the kit.
 Please use only the valid version of the package insert provided with the kit. Intended Use For the qualitative screening of serum IgG antibodies to Trichinella using an Enzyme Linked Immunoabsorbant Assay
Please use only the valid version of the package insert provided with the kit. Intended Use For the qualitative screening of serum IgG antibodies to Trichinella using an Enzyme Linked Immunoabsorbant Assay
Taenia solium IgG ELISA Kit
 Taenia solium IgG ELISA Kit Catalog Number KA3191 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
Taenia solium IgG ELISA Kit Catalog Number KA3191 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
Toxocara. Cat # Toxocara ELISA. Enzyme Linked Immunosorbent Assay. ELISA-Indirect; Antigen Coated Plate
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Influenza A IgG ELISA
 Influenza A IgG ELISA For the qualitative determination of IgG-class antibodies against Influenza A virus in human serum or plasma (citrate, heparin). For Research Use Only. Not For Use In Diagnostic Procedures.
Influenza A IgG ELISA For the qualitative determination of IgG-class antibodies against Influenza A virus in human serum or plasma (citrate, heparin). For Research Use Only. Not For Use In Diagnostic Procedures.
Giardia lamblia/ Cryptosporidium spp. ELISA Kit
 Giardia lamblia/ Cryptosporidium spp. ELISA Kit Catalog Number KA2095 96 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background...
Giardia lamblia/ Cryptosporidium spp. ELISA Kit Catalog Number KA2095 96 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background...
Enzyme-Linked Immunosorbent Assay for Detection of Antibody to Gnathostoma Antigen in Patients with Intermittent Cutaneous Migratory Swelling
 JOURNAL OF CLINICAL MICROBIOLOGY, May 1986, p. 847-851 95-1137/86/5847-5$2./ Copyright D 1986, American Society for Microbiology Vol. 23, No. 5 Enzyme-Linked Immunosorbent Assay for Detection of Antibody
JOURNAL OF CLINICAL MICROBIOLOGY, May 1986, p. 847-851 95-1137/86/5847-5$2./ Copyright D 1986, American Society for Microbiology Vol. 23, No. 5 Enzyme-Linked Immunosorbent Assay for Detection of Antibody
Infection of Blastocystis hominis in primary schoolchildren from Nakhon Pathom province, Thailand
 Tropical Biomedicine 23(1): 117 122 (2006) Infection of Blastocystis hominis in primary schoolchildren from Nakhon Pathom province, Thailand Rapeeporn Yaicharoen, Warunee Ngrenngarmlert, Nuttapong Wongjindanon,
Tropical Biomedicine 23(1): 117 122 (2006) Infection of Blastocystis hominis in primary schoolchildren from Nakhon Pathom province, Thailand Rapeeporn Yaicharoen, Warunee Ngrenngarmlert, Nuttapong Wongjindanon,
Amoebiasis. (Amoebic dysentery)
 Amoebiasis (Amoebic dysentery) Causative agent: Entamoeba histolytica Amoebiasis Harbouring of protozoa E. histolytica inside the body with or without disease only 10% of infected develop disease two types
Amoebiasis (Amoebic dysentery) Causative agent: Entamoeba histolytica Amoebiasis Harbouring of protozoa E. histolytica inside the body with or without disease only 10% of infected develop disease two types
IV2-113E Use by. Invitron Glargine ELISA Kit REF LOT IVD. Definitions. English. For in-vitro diagnostic use. Instructions for use.
 Definitions Instructions for use REF Catalogue number IV2-113E Use by English Invitron Glargine ELISA Kit For in-vitro diagnostic use Σ 96 LOT IVD Lot/Batch Code Storage temperature limitations In vitro
Definitions Instructions for use REF Catalogue number IV2-113E Use by English Invitron Glargine ELISA Kit For in-vitro diagnostic use Σ 96 LOT IVD Lot/Batch Code Storage temperature limitations In vitro
Cryptosporidium Veterinary
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
H.pylori IgA Cat #
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Non_ pathogenic Amoeba of humans:
 The parasite protozoa Phylum: Sarcomastigophora Sarcodina االميبات Amoebae Non_ pathogenic Amoeba of humans: 1. Entamoeba hartmanii, contain trophozoite and cyst 2. Entamoeba coli, cyst and trophozoite
The parasite protozoa Phylum: Sarcomastigophora Sarcodina االميبات Amoebae Non_ pathogenic Amoeba of humans: 1. Entamoeba hartmanii, contain trophozoite and cyst 2. Entamoeba coli, cyst and trophozoite
Diagnosis of Amebic Liver Abscess and Intestinal Infection with the TechLab Entamoeba histolytica II Antigen Detection and Antibody Tests
 JOURNAL OF CLINICAL MICROBIOLOGY, Sept. 2000, p. 3235 3239 Vol. 38, No. 9 0095-1137/00/$04.00 0 Copyright 2000, American Society for Microbiology. All Rights Reserved. Diagnosis of Amebic Liver Abscess
JOURNAL OF CLINICAL MICROBIOLOGY, Sept. 2000, p. 3235 3239 Vol. 38, No. 9 0095-1137/00/$04.00 0 Copyright 2000, American Society for Microbiology. All Rights Reserved. Diagnosis of Amebic Liver Abscess
INTERNATIONAL JOURNAL OF PHARMACEUTICAL RESEARCH AND BIO-SCIENCE
 INTERNATIONAL JOURNAL OF PHARMACEUTICAL RESEARCH AND BIO-SCIENCE SEROPREVALENCE OF AMOEBIASIS IN A TERTIARY CARE HOSPITAL DEEPINDER CHHINA, VEENU GUPTA, JASDEEP SINGH, POOJA SURI Department of Microbiology,
INTERNATIONAL JOURNAL OF PHARMACEUTICAL RESEARCH AND BIO-SCIENCE SEROPREVALENCE OF AMOEBIASIS IN A TERTIARY CARE HOSPITAL DEEPINDER CHHINA, VEENU GUPTA, JASDEEP SINGH, POOJA SURI Department of Microbiology,
H.Pylori IgG Cat # 1503Z
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Outline EP1201. NEHA 2012 AEC June 2012
 Food and Water Borne Enteric Protozoa: Environmental Health Perspectives Stephanie M. Fletcher, PhD (c) NEHA AEC & Exhibition San Diego June 30, 2012 Outline Introduction Overview of the Epidemiology of
Food and Water Borne Enteric Protozoa: Environmental Health Perspectives Stephanie M. Fletcher, PhD (c) NEHA AEC & Exhibition San Diego June 30, 2012 Outline Introduction Overview of the Epidemiology of
See external label 2 C-8 C Σ=96 tests Cat # 1505Z. MICROWELL ELISA H.Pylori IgA Cat # 1505Z
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Toxoplasma gondii IgM (Toxo IgM)
 DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
H.Pylori IgG
 DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
H. pylori IgM. Cat # H. pylori IgM ELISA. ELISA: Enzyme Linked Immunosorbent Assay. ELISA - Indirect; Antigen Coated Plate
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com H. pylori
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com H. pylori
Diagnosis of amoebic colitis by antigen capture ELISA in patients presenting with acute diarrhoea in Cairo, Egypt
 Tropical Medicine and International Health volume 7 no 4 pp 365 370 april 2002 Diagnosis of amoebic colitis by antigen capture ELISA in patients presenting with acute diarrhoea in Cairo, Egypt Mohamed
Tropical Medicine and International Health volume 7 no 4 pp 365 370 april 2002 Diagnosis of amoebic colitis by antigen capture ELISA in patients presenting with acute diarrhoea in Cairo, Egypt Mohamed
Laboratory diagnosis of parasitic diseases. (Amoebiasis)
 Laboratory diagnosis of parasitic diseases (Amoebiasis) Sarah Alharbi Clinical Laboratory department Collage of Applied Medical Sciences King Saud University This document contains materials modified or
Laboratory diagnosis of parasitic diseases (Amoebiasis) Sarah Alharbi Clinical Laboratory department Collage of Applied Medical Sciences King Saud University This document contains materials modified or
POLYPEPTIDES ASSOCIATED WITH IN VITRO CYST FORMATION OF BLASTOCYSTIS HOMINIS
 IN VITRO CYST FORMATION OF B. HOMINIS POLYPEPTIDES ASSOCIATED WITH IN VITRO CYST FORMATION OF BLASTOCYSTIS HOMINIS I Init 1, JW Mak 2, S Top 3, Z Zulhainan 4, S Prummongkol 5, V Nissapatorn 1, WS Wan-Yusoff
IN VITRO CYST FORMATION OF B. HOMINIS POLYPEPTIDES ASSOCIATED WITH IN VITRO CYST FORMATION OF BLASTOCYSTIS HOMINIS I Init 1, JW Mak 2, S Top 3, Z Zulhainan 4, S Prummongkol 5, V Nissapatorn 1, WS Wan-Yusoff
IVD. DRG Toxocara canis ELISA (EIA-3518) Revised 12 Feb rm (Vers. 6.1)
 Please use only the valid version of the package insert provided with the kit. Intended Use For the qualitative screening of serum IgG antibodies to Toxocara using an Enzyme-Linked Immunosorbent Assay
Please use only the valid version of the package insert provided with the kit. Intended Use For the qualitative screening of serum IgG antibodies to Toxocara using an Enzyme-Linked Immunosorbent Assay
Human Apolipoprotein A1 EIA Kit
 A helping hand for your research Product Manual Human Apolipoprotein A1 EIA Kit Catalog Number: 83901 96 assays 1 Table of Content Product Description 3 Assay Principle 3 Kit Components 3 Storage 4 Reagent
A helping hand for your research Product Manual Human Apolipoprotein A1 EIA Kit Catalog Number: 83901 96 assays 1 Table of Content Product Description 3 Assay Principle 3 Kit Components 3 Storage 4 Reagent
Giardiasis. Table of Contents
 Table of Contents Case Definition... Error! Bookmark not defined. Reporting Requirements... 2 Etiology... Error! Bookmark not defined. Clinical Presentation... Error! Bookmark not defined. Diagnosis...
Table of Contents Case Definition... Error! Bookmark not defined. Reporting Requirements... 2 Etiology... Error! Bookmark not defined. Clinical Presentation... Error! Bookmark not defined. Diagnosis...
Mouse HBsAg(Hepatitis B Virus Surface Antigen) ELISA Kit
 Mouse HBsAg(Hepatitis B Virus Surface Antigen) ELISA Kit Catalogue No.: EM0002 Size: 96T Reactivity: Mouse Application: This immunoassay kit allows for the qualitative determination of HBsAg in Mouse serum
Mouse HBsAg(Hepatitis B Virus Surface Antigen) ELISA Kit Catalogue No.: EM0002 Size: 96T Reactivity: Mouse Application: This immunoassay kit allows for the qualitative determination of HBsAg in Mouse serum
See external label 96 tests HSV 2 IgA. Cat #
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
CYTOMEGALOVIRUS (CMV) IgM ELISA Kit Protocol
 CYTOMEGALOVIRUS (CMV) IgM ELISA Kit Protocol (Cat. No.:EK-310-91) 330 Beach Road, Burlingame CA Tel: 650-558-8898 Fax: 650-558-1686 E-Mail: info@phoenixpeptide.com www.phoenixpeptide.com INTENDED USE The
CYTOMEGALOVIRUS (CMV) IgM ELISA Kit Protocol (Cat. No.:EK-310-91) 330 Beach Road, Burlingame CA Tel: 650-558-8898 Fax: 650-558-1686 E-Mail: info@phoenixpeptide.com www.phoenixpeptide.com INTENDED USE The
GLP-2 (Rat) ELISA. For the quantitative determination of glucagon-like peptide 2 (GLP-2) in rat serum and plasma
 GLP-2 (Rat) ELISA For the quantitative determination of glucagon-like peptide 2 (GLP-2) in rat serum and plasma For Research Use Only. Not For Use In Diagnostic Procedures. Catalog Number: 48-GP2RT-E01
GLP-2 (Rat) ELISA For the quantitative determination of glucagon-like peptide 2 (GLP-2) in rat serum and plasma For Research Use Only. Not For Use In Diagnostic Procedures. Catalog Number: 48-GP2RT-E01
Hompes Method. Practitioner Training Level II. Lesson Seven Part A DRG Pathogen Plus Interpretation
 Hompes Method Practitioner Training Level II Lesson Seven Part A DRG Pathogen Plus Interpretation Health for the People Ltd not for reuse without expressed permission Hompes Method is a trading name of
Hompes Method Practitioner Training Level II Lesson Seven Part A DRG Pathogen Plus Interpretation Health for the People Ltd not for reuse without expressed permission Hompes Method is a trading name of
Helicobacter pylori IgA ELISA Kit
 Helicobacter pylori IgA ELISA Kit Catalog Number KA0964 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Helicobacter pylori IgA ELISA Kit Catalog Number KA0964 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Mycoplasma pneumoniae IgG ELISA Kit
 Mycoplasma pneumoniae IgG ELISA Kit Catalog Number KA2260 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Mycoplasma pneumoniae IgG ELISA Kit Catalog Number KA2260 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Toxoplasma gondii IgM ELISA Kit
 Toxoplasma gondii IgM ELISA Kit Catalog Number KA0226 96 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Toxoplasma gondii IgM ELISA Kit Catalog Number KA0226 96 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
ELISA TEST FOR DETECTION OF BLASTOCYSTIS SPP. IN HUMAN FAECES: COMPARISON OF THREE METHODS
 ELISA TEST FOR DETECTION OF BLASTOCYSTIS SPP. IN HUMAN FAECES: COMPARISON OF THREE METHODS István Kucsera, Mónika Molnár, Eszter Gályász, József Danka, Erika Orosz National Center for Epidemiology, Department
ELISA TEST FOR DETECTION OF BLASTOCYSTIS SPP. IN HUMAN FAECES: COMPARISON OF THREE METHODS István Kucsera, Mónika Molnár, Eszter Gályász, József Danka, Erika Orosz National Center for Epidemiology, Department
American Association of Bioanalysts 5615 Kirby Drive, Suite 870 Houston, TX
 Q3 2018 Parasitology American Association of Bioanalysts 5615 Kirby Drive, Suite 870 Houston, TX 77005 800-234-5315 281-436-5357 Specimen 1 Referees Extent 1 Extent 2 Total Few to 534 Giardia lamblia Many
Q3 2018 Parasitology American Association of Bioanalysts 5615 Kirby Drive, Suite 870 Houston, TX 77005 800-234-5315 281-436-5357 Specimen 1 Referees Extent 1 Extent 2 Total Few to 534 Giardia lamblia Many
CHAPTER FOUR. Intestinal Amebae
 PART II Protozoa CHAPTER FOUR Intestinal Amebae OUTLINE INTESTINAL AMEBAE Entamoeba histolytica/entamoeba dispar (EN-ta-MEE-buh HIS-toe-LIT-I-ka/EN-ta-MEEbuh DIS-par) Entamoeba hartmanni (EN-ta-MEE-buh
PART II Protozoa CHAPTER FOUR Intestinal Amebae OUTLINE INTESTINAL AMEBAE Entamoeba histolytica/entamoeba dispar (EN-ta-MEE-buh HIS-toe-LIT-I-ka/EN-ta-MEEbuh DIS-par) Entamoeba hartmanni (EN-ta-MEE-buh
SIV p27 Antigen ELISA Catalog Number:
 INTENDED USE The RETRO-TEK SIV p27 Antigen ELISA is for research use only and is not intended for in vitro diagnostic use. The RETRO-TEK SIV p27 Antigen ELISA is an enzyme linked immunoassay used to detect
INTENDED USE The RETRO-TEK SIV p27 Antigen ELISA is for research use only and is not intended for in vitro diagnostic use. The RETRO-TEK SIV p27 Antigen ELISA is an enzyme linked immunoassay used to detect
News and Notes. Parasitology Comprehensive 2 October Sample Preparation and Quality Control. 12 K (All Parasites)
 NEW YORK STATE Parasitology Proficiency Testing Program News and Notes Recent reports in the literature have indicated a high rate of Cryptosporidium sp. false positive associated with Rapid Cartridge
NEW YORK STATE Parasitology Proficiency Testing Program News and Notes Recent reports in the literature have indicated a high rate of Cryptosporidium sp. false positive associated with Rapid Cartridge
PARATEST STABILITY STUDY, SYSTEM USED FOR DIAGNOSIS OF INTESTINAL PARASITES
 PARATEST STABILITY STUDY, SYSTEM USED FOR DIAGNOSIS OF INTESTINAL PARASITES Rev.00 January 10, 2011 1. Introduction Intestinal parasitic diseases represent a serious public health issue in underdeveloped
PARATEST STABILITY STUDY, SYSTEM USED FOR DIAGNOSIS OF INTESTINAL PARASITES Rev.00 January 10, 2011 1. Introduction Intestinal parasitic diseases represent a serious public health issue in underdeveloped
Human HIV (1+2) antigen&antibody ELISA Kit
 Human HIV (1+2) antigen&antibody ELISA Kit Catalog Number. CSB-E18042h For the qualitative determination of human HIV (1+2) antibody and P24 antigen concentrations in serum, plasma. This package insert
Human HIV (1+2) antigen&antibody ELISA Kit Catalog Number. CSB-E18042h For the qualitative determination of human HIV (1+2) antibody and P24 antigen concentrations in serum, plasma. This package insert
Anton van Leeuwenhoek. Protozoa: This is what he saw in his own stool sample. Morphology 10/14/2009. Protozoans that cause diarrheal disease
 Access to safe drinking water is everyone s right Anton van Leeuwenhoek Protozoa: Protozoans that cause diarrheal disease This is what he saw in his own stool sample 1. Giardia lamblia 2. Entameba histolytica
Access to safe drinking water is everyone s right Anton van Leeuwenhoek Protozoa: Protozoans that cause diarrheal disease This is what he saw in his own stool sample 1. Giardia lamblia 2. Entameba histolytica
Influenza A H1N1 HA ELISA Pair Set
 Influenza A H1N1 HA ELISA Pair Set for H1N1 ( A/Puerto Rico/8/1934 ) HA Catalog Number : SEK11684 To achieve the best assay results, this manual must be read carefully before using this product and the
Influenza A H1N1 HA ELISA Pair Set for H1N1 ( A/Puerto Rico/8/1934 ) HA Catalog Number : SEK11684 To achieve the best assay results, this manual must be read carefully before using this product and the
Access to safe drinking water is everyone s right. Protozoans that cause diarrheal disease
 Access to safe drinking water is everyone s right Protozoa: Protozoans that cause diarrheal disease 1. Giardia lamblia 2. Entameba histolytica 3. Cryptosporidium parvum 4. Cyclospora cayetanensis 1 Giardia
Access to safe drinking water is everyone s right Protozoa: Protozoans that cause diarrheal disease 1. Giardia lamblia 2. Entameba histolytica 3. Cryptosporidium parvum 4. Cyclospora cayetanensis 1 Giardia
HSV-1 IgM ELISA. Catalog No (96 Tests) For Research Use Only. Not for use in Diagnostic Procedures.
 For Research Use Only. Not for use in Diagnostic Procedures. INTENDED USE The GenWay, Inc. HSV-1 IgM ELISA Kit is intended for the detection of IgM antibody to HSV-1 in human serum or plasma. SUMMARY AND
For Research Use Only. Not for use in Diagnostic Procedures. INTENDED USE The GenWay, Inc. HSV-1 IgM ELISA Kit is intended for the detection of IgM antibody to HSV-1 in human serum or plasma. SUMMARY AND
GLP-2 ELISA. For the quantitative determination of GLP-2 in human serum and plasma samples.
 GLP-2 ELISA For the quantitative determination of GLP-2 in human serum and plasma samples. For Research Use Only. Not For Use In Diagnostic Procedures. Catalog Number: 48-GP2HU-E01.1 Size: 96 wells Version:
GLP-2 ELISA For the quantitative determination of GLP-2 in human serum and plasma samples. For Research Use Only. Not For Use In Diagnostic Procedures. Catalog Number: 48-GP2HU-E01.1 Size: 96 wells Version:
TYPES OF ORGANISM RELATIONSHIPS
 TYPES OF ORGANISM RELATIONSHIPS Normal Flora. Normal flora consists of microorganisms that are normally and consistently found in or on the body in the absence of disease. Symbiosis. This is the close
TYPES OF ORGANISM RELATIONSHIPS Normal Flora. Normal flora consists of microorganisms that are normally and consistently found in or on the body in the absence of disease. Symbiosis. This is the close
Campylobacter Antigen ELISA Kit
 Campylobacter Antigen ELISA Kit Catalog Number KA3204 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Campylobacter Antigen ELISA Kit Catalog Number KA3204 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
HBeAg and HBeAg Ab ELISA Kit
 HBeAg and HBeAg Ab ELISA Kit Catalog Number KA0290 96 assays Version: 17 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Principle of the Assay... 3
HBeAg and HBeAg Ab ELISA Kit Catalog Number KA0290 96 assays Version: 17 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Principle of the Assay... 3
CONTENTS. STUDY DESIGN METHODS ELISA protocol for quantitation of mite (Dermatophagoides spp.) Der p 1 or Der f 1
 CONTENTS STUDY DESIGN METHODS ELISA protocol for quantitation of mite (Dermatophagoides spp.) Der p 1 or Der f 1 ELISA protocol for mite (Dermatophagoides spp.) Group 2 ALLERGENS RESULTS (SUMMARY) TABLE
CONTENTS STUDY DESIGN METHODS ELISA protocol for quantitation of mite (Dermatophagoides spp.) Der p 1 or Der f 1 ELISA protocol for mite (Dermatophagoides spp.) Group 2 ALLERGENS RESULTS (SUMMARY) TABLE
Serological Techniques for Laboratory Diagnosis
 JOURNAL OF CLINICAL MICROBIOLOGY, Aug. 1979, p. 128-133 0095-1 137/79/08-0128/06$02.00/0 Vol. 10, No. 2 Entamoeba histolytica: Efficacy of Microscopic, Cultural, and Serological Techniques for Laboratory
JOURNAL OF CLINICAL MICROBIOLOGY, Aug. 1979, p. 128-133 0095-1 137/79/08-0128/06$02.00/0 Vol. 10, No. 2 Entamoeba histolytica: Efficacy of Microscopic, Cultural, and Serological Techniques for Laboratory
Ph. Eur. Reference Standard - LEAFLET
 European Directorate for the Quality of Medicines & HealthCare European Pharmacopoeia (Ph. Eur.) 7, Allée Kastner CS 30026, F-67081 Strasbourg (France) Tel. +33 (0)3 88 41 20 35 Fax. + 33 (0)3 88 41 27
European Directorate for the Quality of Medicines & HealthCare European Pharmacopoeia (Ph. Eur.) 7, Allée Kastner CS 30026, F-67081 Strasbourg (France) Tel. +33 (0)3 88 41 20 35 Fax. + 33 (0)3 88 41 27
Mouse GLP-2 EIA FOR LABORATORY USE ONLY
 YK142 Mouse GLP-2 EIA FOR LABORATORY USE ONLY Kasumigaseki place, 3-6-7, Kasumigaseki, Chiyoda-ku, Tokyo 100-0013 Japan http://www.sceti.co.jp/english/export e-mail exp-pet@sceti.co.jp
YK142 Mouse GLP-2 EIA FOR LABORATORY USE ONLY Kasumigaseki place, 3-6-7, Kasumigaseki, Chiyoda-ku, Tokyo 100-0013 Japan http://www.sceti.co.jp/english/export e-mail exp-pet@sceti.co.jp
Anthrax protective antigen IgG ELISA Kit
 Anthrax protective antigen IgG ELISA Kit Catalog Number KA0953 96 assays Version: 04 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3
Anthrax protective antigen IgG ELISA Kit Catalog Number KA0953 96 assays Version: 04 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3
Toxoplasma gondii IgM ELISA Kit
 Toxoplasma gondii IgM ELISA Kit Catalog Number KA0226 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Toxoplasma gondii IgM ELISA Kit Catalog Number KA0226 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
RIDA QUICK Cryptosporidium/Giardia/Entamoeba Combi
 RIDA QUICK Cryptosporidium/Giardia/Entamoeba Combi Article no: N1723 R-Biopharm AG, An der neuen Bergstraße 17, D-64297 Darmstadt, Germany Phone: +49 (0) 61 51 81 02-0 / Fax: +49 (0) 61 51 81 02-20 1.
RIDA QUICK Cryptosporidium/Giardia/Entamoeba Combi Article no: N1723 R-Biopharm AG, An der neuen Bergstraße 17, D-64297 Darmstadt, Germany Phone: +49 (0) 61 51 81 02-0 / Fax: +49 (0) 61 51 81 02-20 1.
PREVALENCE OF INTESTINAL PARASITES IN HIV-POSITIVE/AIDS PATIENTS. O.O Oguntibeju
 Malaysian Journal of Medical Sciences, Vol. 13, No. 1, January 2006 (68-73) ORIGINAL ARTICLE PREVALENCE OF INTESTINAL PARASITES IN HIV-POSITIVE/AIDS PATIENTS O.O Oguntibeju School of Health Technology,
Malaysian Journal of Medical Sciences, Vol. 13, No. 1, January 2006 (68-73) ORIGINAL ARTICLE PREVALENCE OF INTESTINAL PARASITES IN HIV-POSITIVE/AIDS PATIENTS O.O Oguntibeju School of Health Technology,
DIAGNOSTIC AUTOMATION, INC.
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
IgG Antibodies To Toxoplasma Gondii ELISA Kit Protocol
 IgG Antibodies To Toxoplasma Gondii ELISA Kit Protocol (Cat. No.:EK-310-85) 330 Beach Road, Burlingame CA Tel: 650-558-8898 Fax: 650-558-1686 E-Mail: info@phoenixpeptide.com www.phoenixpeptide.com INTENDED
IgG Antibodies To Toxoplasma Gondii ELISA Kit Protocol (Cat. No.:EK-310-85) 330 Beach Road, Burlingame CA Tel: 650-558-8898 Fax: 650-558-1686 E-Mail: info@phoenixpeptide.com www.phoenixpeptide.com INTENDED
Thyroid Stimulating Hormone (S-TSH) Thyroid Stimulating
 ab108659 Thyroid Stimulating Hormone (S-TSH) Human ELISA Kit Instructions for Use For the quantitative measurement of Human Thyroid Stimulating Hormone (S-TSH) concentrations in serum. This product is
ab108659 Thyroid Stimulating Hormone (S-TSH) Human ELISA Kit Instructions for Use For the quantitative measurement of Human Thyroid Stimulating Hormone (S-TSH) concentrations in serum. This product is
25(OH) Vitamin D ELISA (BD-220BA), 192 Tests
 INTENDED USE The 25-hydroxy (25-OH) Vitamin D ELISA is intended for the quantitative determination of total 25-OH Vitamin D in human serum and Plasma. SUMMARY AND EXPLANATION Vitamin D is a steroid hormone
INTENDED USE The 25-hydroxy (25-OH) Vitamin D ELISA is intended for the quantitative determination of total 25-OH Vitamin D in human serum and Plasma. SUMMARY AND EXPLANATION Vitamin D is a steroid hormone
Dr. Jabar Etaby Lecture GIARDIASIS(lambliasis) Etiology: Giardia lamblia (flagellate)
 Dr. Jabar Etaby Lecture Two GIARDIASIS(lambliasis) Etiology: Giardia lamblia (flagellate) Epidemiology: It has worldwide distribution and is not uncommon in South Carolina. It is the most frequent protozoan
Dr. Jabar Etaby Lecture Two GIARDIASIS(lambliasis) Etiology: Giardia lamblia (flagellate) Epidemiology: It has worldwide distribution and is not uncommon in South Carolina. It is the most frequent protozoan
Chymotrypsin ELISA Kit
 Chymotrypsin ELISA Kit Cat. No.:DEIA10041 Pkg.Size:96T Intended use The Chymotrypsin ELISA Kit is a sandwich Enzyme Immuno Assay intended for the quantitative determination of Chymotrypsin in stool. General
Chymotrypsin ELISA Kit Cat. No.:DEIA10041 Pkg.Size:96T Intended use The Chymotrypsin ELISA Kit is a sandwich Enzyme Immuno Assay intended for the quantitative determination of Chymotrypsin in stool. General
NF-κB p65 (Phospho-Thr254)
 Assay Biotechnology Company www.assaybiotech.com Tel: 1-877-883-7988 Fax: 1-877-610-9758 NF-κB p65 (Phospho-Thr254) Colorimetric Cell-Based ELISA Kit Catalog #: OKAG02015 Please read the provided manual
Assay Biotechnology Company www.assaybiotech.com Tel: 1-877-883-7988 Fax: 1-877-610-9758 NF-κB p65 (Phospho-Thr254) Colorimetric Cell-Based ELISA Kit Catalog #: OKAG02015 Please read the provided manual
H.Pylori IgM Cat # 1504Z
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Chapter 1 The Public Health Role of Clinical Laboratories
 Chapter 1 The Public Health Role of Clinical Laboratories A. Epidemic Diarrhea The two most common types of epidemic diarrhea in developing countries are watery diarrhea caused by Vibrio cholerae serogroup
Chapter 1 The Public Health Role of Clinical Laboratories A. Epidemic Diarrhea The two most common types of epidemic diarrhea in developing countries are watery diarrhea caused by Vibrio cholerae serogroup
This kit is intended for Research Use Only. Not for use in diagnostic procedures.
 This kit is intended for Research Use Only. Not for use in diagnostic procedures. Introduction The DRG Epstein-Barr Virus (VCA) IgM Enzyme Immunoassay Kit provides materials for determination of IgM-class
This kit is intended for Research Use Only. Not for use in diagnostic procedures. Introduction The DRG Epstein-Barr Virus (VCA) IgM Enzyme Immunoassay Kit provides materials for determination of IgM-class
Rubella Latex Agglutination Test
 Rubella Latex Agglutination Test Cat. No.:DLAT1088 Pkg.Size:30T Intended use The Rubella Latex Agglutination Test is a rapid latex particle agglutination test for the qualitative and semi-quantitative
Rubella Latex Agglutination Test Cat. No.:DLAT1088 Pkg.Size:30T Intended use The Rubella Latex Agglutination Test is a rapid latex particle agglutination test for the qualitative and semi-quantitative
Triiodothyronine (T3) ELISA
 For Research Use Only. Not for use in Diagnostic Procedures. INTENDED USE The GenWay, Inc. Triiodothyronine (T3) ELISA Kit is intended for the detection of total T3 in human serum or plasma. For research
For Research Use Only. Not for use in Diagnostic Procedures. INTENDED USE The GenWay, Inc. Triiodothyronine (T3) ELISA Kit is intended for the detection of total T3 in human serum or plasma. For research
CoproELISA Giardia/Cryptosporidium
 CoproELISA Giardia/Cryptosporidium Enzyme-Linked Immunosorbent Assay (ELISA) for the detection of Giardia lamblia and Cryptosporidium spp. antigens in human feces Instruction Manual Test kit for 96 determinations
CoproELISA Giardia/Cryptosporidium Enzyme-Linked Immunosorbent Assay (ELISA) for the detection of Giardia lamblia and Cryptosporidium spp. antigens in human feces Instruction Manual Test kit for 96 determinations
Mouse GLP-2 EIA. Cat. No. KT-374. For the quantitative determination of GLP-2 in mouse serum or plasma. For Research Use Only. 1 Rev.
 Mouse GLP-2 EIA For the quantitative determination of GLP-2 in mouse serum or plasma. Cat. No. KT-374 For Research Use Only. 1 Rev. 11357374 PRODUCT INFORMATION Mouse GLP-2 EIA Cat. No. KT-374 INTENDED
Mouse GLP-2 EIA For the quantitative determination of GLP-2 in mouse serum or plasma. Cat. No. KT-374 For Research Use Only. 1 Rev. 11357374 PRODUCT INFORMATION Mouse GLP-2 EIA Cat. No. KT-374 INTENDED
Rat Leptin-HS ELISA FOR LABORATORY USE ONLY YANAIHARA INSTITUTE INC AWAKURA, FUJINOMIYA - SHI SHIZUOKA, JAPAN
 YK051 Rat Leptin-HS ELISA FOR LABORATORY USE ONLY YANAIHARA INSTITUTE INC. 2480-1 AWAKURA, FUJINOMIYA - SHI SHIZUOKA, JAPAN 418 0011 Contents Introduction 2 Characteristics 3 Composition 4 Method 5-6 Notes
YK051 Rat Leptin-HS ELISA FOR LABORATORY USE ONLY YANAIHARA INSTITUTE INC. 2480-1 AWAKURA, FUJINOMIYA - SHI SHIZUOKA, JAPAN 418 0011 Contents Introduction 2 Characteristics 3 Composition 4 Method 5-6 Notes
Rubella virus IgG ELISA Kit
 Rubella virus IgG ELISA Kit Catalog Number KA0223 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
Rubella virus IgG ELISA Kit Catalog Number KA0223 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
however, and the present communication is concerned with some of
 THE AGGLUTINATION OF HUMAN ERYTHROCYTES MODIFIED BY TREATMENT WITH NEWCASTLE DISEASE AND INFLUENZA VIRUS' ALFRED L. FLORMAN' Pediatric Service and Division of Bacteriology, The Mount Sinai Hospital, New
THE AGGLUTINATION OF HUMAN ERYTHROCYTES MODIFIED BY TREATMENT WITH NEWCASTLE DISEASE AND INFLUENZA VIRUS' ALFRED L. FLORMAN' Pediatric Service and Division of Bacteriology, The Mount Sinai Hospital, New
Giardia lamblia (flagellates)
 Giardia lamblia (flagellates) Dr. Hala Al Daghistani Giardia lamblia (Giardia duodenalis or Giardia intestinalis) is the causative agent of giardiasis and is the only common pathogenic protozoan found
Giardia lamblia (flagellates) Dr. Hala Al Daghistani Giardia lamblia (Giardia duodenalis or Giardia intestinalis) is the causative agent of giardiasis and is the only common pathogenic protozoan found
