ORIGINAL INVESTIGATION. Treatment for Hepatitis C Virus in Human Immunodeficiency Virus Infected Patients
|
|
|
- Charlotte Payne
- 5 years ago
- Views:
Transcription
1 Treatment for Hepatitis C Virus in Human Immunodeficiency Virus Infected Patients Clinical Benefits and Cost-effectiveness ORIGINAL INVESTIGATION Felicitas C. Kuehne, MSc; Ullrich Bethe, MD, MSc; Kenneth Freedberg, MD, MSc; Sue J. Goldie, MD, MPH Background: Hepatitis C virus (HCV) is an important cause of liver disease in human immunodeficiency virus (HIV) infected patients. Objective: To assess the cost-effectiveness of alternative management strategies for chronic HCV in coinfected patients with moderate hepatitis. Methods: A state-transition model was used to simulate a cohort of HIV-infected patients with a mean CD4 cell count of 350 cells/µl and moderate chronic hepatitis C stratified by genotype. Strategies included interferon alfa (48 weeks), pegylated interferon alfa (48 weeks), interferon alfa and ribavirin (24 and 48 weeks), pegylated interferon alfa and ribavirin (48 weeks), and no treatment. Outcomes included life expectancy, qualityadjusted life years (QALYs), and incremental costeffectiveness ratios. Results: Treatment for moderate chronic HCV with combination therapy using an interferon-based regimen reduced the incidence of cirrhosis and provided gains in quality-adjusted life expectancy ranging from 6.2 to 13.9 months, depending on genotype. Regardless of genotype, the cost-effectiveness of interferon alfa and ribavirin for patients with moderate hepatitis was lower than $50000 per QALY vs the next best strategy. With genotype 1, pegylated interferon alfa (vs interferon alfa) and ribavirin therapy provided an additional 1.6 qualityadjusted life-months for $40000 per QALY. Because treatment is more effective with non-1 genotypes, pegylated interferon (vs interferon alfa) and ribavirin provided only 3 additional quality-adjusted life-months for $ per QALY. For patients who were intolerant of ribavirin, monotherapy with pegylated interferon was always the most cost-effective option. Conclusions: Combination therapy for moderate hepatitis in coinfected patients will increase quality-adjusted life expectancy and have a cost-effectiveness ratio comparable to that of other well-accepted clinical interventions. Arch Intern Med. 2002;162: From the Center for Risk Analysis, Department of Health Policy and Management, Harvard School of Public Health, Boston, Mass (Ms Kuehne and Drs Freedberg and Goldie); the Department of Internal Medicine, University of Cologne, Cologne, Germany (Dr Bethe); and the Divisions of General Medicine and Infectious Diseases and the Partner s AIDS Research Center, Massachusetts General Hospital, Harvard Medical School, Boston (Dr Freedberg). APPROXIMATELY ONE third of all human immunodeficiency virus (HIV) infected persons in the United States are coinfected with hepatitis C virus (HCV). 1-3 Although the prognosis for HIV infection has improved dramatically with the availability of highly active antiretroviral therapy (HAART), 4,5 coinfected patients are now vulnerable to the complications associated with longterm HCV infection (eg, cirrhosis). 6-9 Numerous studies report an increase in hospital admissions and deaths due to liver disease in HIV-infected patients. Compared with HIV-uninfected patients, coinfected patients have higher levels of HCV RNA and a greater risk of progression to severe liver disease. 2,6,7,19,24-39 Hepatitis C virus may also have a negative impact on HIV disease progression, although this issue remains controversial. 2,19,38,40-45 Clinical trials have documented the enhanced antiviral efficacy of combination therapy with ribavirin and interferon alfa, with sustained treatment responses ranging from 33% to 49%. Moreover, multiple studies focusing on HIV-uninfected patients have shown that the cost-effectiveness of treatment for chronic HCV is comparable to that of other well-accepted standard preventive interventions. Recently, treatment with pegylated interferon alfa (with and without ribavirin), a long-acting variant of interferon alfa that requires less frequent dosing, has been shown to have higher efficacy than use of interferon alfa alone The cost-effectiveness of pegylated interferon alfa therapy has not yet been explored in either HIV-infected or HIVuninfected patients. The longer expected survival of HIV-infected patients receiving HAART, coupled with the potentially accelerated progression of HCV-related liver disease, has resulted in a set of complex clinical and policy questions. Treatment recommen- 2545
2 Moderate Chronic Hepatitis C Mild Chronic Hepatitis C Compensated Cirrhosis Death HIV HCV Other dations for HIV-uninfected patients may not be directly applicable to coinfected patients Although information from ongoing clinical studies in coinfected patients is anticipated in the next several years, the ideal source of data long-term placebo-controlled trials of all potential treatment strategies is not likely to be feasible. For many coinfected patients, treatment decisions must be made now, in the setting of considerable uncertainty and despite the absence of perfect information. To inform these decisions, we incorporated the best available data to assess the health and economic consequences of a variety of strategies to manage HCV-related liver disease in HIV-infected patients. METHODS A computer-based mathematical model was used to compare the following strategies for the management of moderate chronic HCV in coinfected patients with CD4 cell counts greater than 350 cells/µl: (1) no HCV treatment, (2) monotherapy with interferon alfa (48 weeks), (3) monotherapy with pegylated interferon alfa (48 weeks), (4) combination therapy with interferon alfa and ribavirin (24 and 48 weeks), and (5) combination therapy with pegylated interferon alfa and ribavirin (48 weeks). Analyses were stratified by HCV genotype because distributions of genotypes vary in different patient subgroups and affect response to therapy. 64,65,76 We adopted a societal perspective and followed the recommendations of the Panel on Cost-effectiveness in Health and Medicine. 77 Model outcomes included qualityadjusted life expectancy (QALE) and total lifetime costs. Comparative performance of alternative strategies was measured by the incremental cost-effectiveness ratio, defined as the additional cost of a specific treatment strategy divided by its additional clinical benefit compared with the next least-expensive strategy. We conducted univariate and multivariate sensitivity analyses to assess variable uncertainty, and we explored the effect of alternative assumptions compared with those made in the base case. THE MODEL Decompensated Cirrhosis Hepatocellular Carcinoma Figure 1. Overview of the model. The Markov model has 6 general categories of health states that reflect the stage of hepatitis C virus (HCV) associated liver disease. Each health state is stratified by CD4 cell count and treatment status with highly active antiretroviral therapy. Death may be caused by a human immunodeficiency virus (HIV) related illness, HCV-related liver failure or cancer, or other causes. See the Methods section for details. A deterministic state-transition Markov model (DATA 3.5; Tree- Age Software Inc, Williamstown, Mass) was used to simulate the natural history of HCV-induced liver disease in coinfected patients (Figure 1). Health states were defined to reflect 5 broad categories of HCV-related liver disease (mild chronic HCV, moderate chronic HCV, compensated cirrhosis, decompensated cirrhosis, and hepatocellular carcinoma [HCC]), stage of HIV disease (CD4 cell count 500, , and 200 cells/µl), and treatment status with HAART. Decompensated cirrhosis was defined as the presence of complications of portal hypertension, such as ascites, variceal bleeding, or hepatic encephalopathy. In the base case, a cohort of coinfected individuals entered the model with a mean CD4 cell count of 350 cells/µl and histologically moderate chronic hepatitis. Transition probabilities from the literature were used to move individuals through different health states over time. The time horizon of the analysis was divided into equal increments during which patients could progress to later stages of HCV or HIV. Each month, patients could progress from mild to moderate hepatitis or from moderate hepatitis to cirrhosis. Patients with cirrhosis could develop complications (ie, decompensated cirrhosis) or primary HCC. In the absence of HAART, patients could progress from higher to lower CD4 cell strata. We conservatively assumed that HIV progression was independent of HCV but explored the implications of a more rapid progression to acquired immunodeficiency syndrome. Each month, patients faced competing mortality risks from advanced liver disease, acquired immunodeficiency syndrome, and other causes. We made the following assumptions: (1) HCV treatment was discontinued in patients not responding to monotherapy after 3 months or combination therapy after 6 months 71,72,74 ; (2) HCV treatment resulted in 1 of 3 potential outcomes no treatment response, a partial but nonsustained response, or a sustained response; (3) patients with no treatment response received no clinical benefit and were subject to their annual pretreatment risk of HCV-related liver disease progression; (4) patients with a partial but nonsustained response did not progress in their HCV-related liver disease during treatment but were subject to pretreatment risks of liver disease once treatment was stopped; (5) patients with a sustained response (ie, undetectable HCV RNA for 6 months after treatment) did not experience a future risk of HCV-related liver disease, although the implications of relapse were evaluated in sensitivity analysis; (6) response to treatment was conditional on genotype; (7) the rate of liver disease progression in the absence of treatment was independent of genotype; (8) minor adverse effects of treatment resulted in additional costs and a temporary decrease in quality of life, whereas major toxicity due to treatment resulted in treatment discontinuation; (9) patients were eligible to receive 4 sequential regimens of HAART for HIV 78,79 ; and (10) coinfected patients with decompensated cirrhosis or HCC were not eligible for liver transplantation. Alternative assumptions were evaluated in sensitivity analysis. CLINICAL DATA The variable estimates and plausible ranges used in the base case analysis are shown in Table A summary of selected clinical studies used to establish the plausible ranges for variables related to the relationship between HIV and HCV, and the efficacy of treatment for HCV, is available from the authors In HIVuninfected patients, progression from chronic HCV to cirrhosis and HCC may take up to several decades. 8,9 The risk of cirrhosis implied by cohort studies of chronically infected patients ranges from 8% to 50% after 1 to 2 decades, although these studies are subject to referral bias because most were performed at tertiary referral centers. A much slower rate of progression has been implied from prospective cohort studies 80,88, of acutely infected patients after transfusion or during defined periods of exposure. 2546
3 Table 1. Values Used for the Base Case and Plausible Ranges Used for Sensitivity Analysis* Variable Base Case (Range ) Source Reference Numbers Natural history of chronic HCV, annual probability 8, 56, Mild to moderate chronic HCV 4.1 ( ) Moderate chronic HCV to compensated cirrhosis 7.3 ( ) Moderate chronic HCV to hepatocellular carcinoma 0.1 ( ) Natural history of cirrhosis, annual probability Compensated to decompensated cirrhosis 4.2 ( ) Cirrhosis to hepatocellular carcinoma 1.5 ( ) Mortality attributable to decompensated cirrhosis 12.9 ( ) Mortality attributable to hepatocellular carcinoma 78.7 ( ) Relative risk of progression in HIV-infected vs HIV-uninfected patients 2, 6, 7, 19, Mild to moderate chronic HCV 2 (1-5) Moderate HCV to cirrhosis 2 (1-5) Natural history of HIV (without antiretroviral treatment) , 138 Mean duration in HIV stage, y 100, 104 CD4 cell count 500 cells/µl 5.5 CD4 cell count cells/µl 3.6 CD4 cell count 200 cells/µl 3.2 Mortality rate, % 100, 104 CD4 cell count 500 cells/µl CD4 cell count cells/µl CD4 cell count 200 cells/µl HCV treatment efficacy, % Genotype 1 Genotype non , Partial response Interferon alfa, 48 wk 9 (7-15) 21 (17-28) Pegylated interferon alfa, 48 wk 11 (9-25) 26 (21-31) Interferon alfa and ribavirin, 24 wk 25 (16-29) 11 (9-26) Interferon alfa and ribavirin, 48 wk 8 (5-14) 5 (3-17) Pegylated interferon alfa and ribavirin, 48 wk 13 (± 25%) 6 (± 25%) Sustained response Interferon alfa, 48 wk 6 (2-8) 27 (15-28) Pegylated interferon alfa, 48 wk 14 (12-31) 46 (40-67) Interferon alfa and ribavirin, 24 wk 16 (14-28) 69 (62-73) Interferon and ribavirin, 48 wk 33 (28-40) 75 (61-85) Pegylated interferon alfa and ribavirin, 48 wk 42 (34-45) 79 (76-88) Toxic effects, % Minor Major 49, 69, 70 Interferon alfa, 48 wk 31 6 Pegylated interferon alfa, 48 wk Interferon alfa and ribavirin, 24 wk 24 8 Interferon alfa and ribavirin, 48 wk Pegylated interferon alfa and ribavirin, 48 wk Health state quality weights (CD4 cell count 200 cells/µl) 53, 54, 56, Mild chronic HCV 0.87 Moderate chronic HCV 0.81 Compensated cirrhosis 0.68 Decompensated cirrhosis 0.47 Hepatocellular carcinoma 0.23 Health state quality weights (CD4 cell count 200 cells/µl) , 138 Mild chronic HCV 0.80 Moderate chronic HCV 0.74 Compensated cirrhosis 0.62 Decompensated cirrhosis 0.43 Hepatocellular carcinoma 0.21 (continued) To estimate the probabilities of progression from compensated to decompensated cirrhosis as well as the risk of HCC and death, a series of calibration exercises was conducted using one of the largest published studies of cirrhotic patients with chronic HCV. 91 First, we used the model to simulate the course of chronic HCV in an HIV-uninfected cohort and projected several clinical outcomes, including decompensated cirrhosis, HCC, and death. Second, the probabilities of disease progression were adjusted until the model outcome projections closely matched the observed data. 91 Finally, these disease progression probabilities were adjusted to reflect the increased risk of advanced liver disease reported in coinfected patients. 2,21-25,27,29-39, In the base case, we conservatively assumed a relative risk (RR) of progression to cirrhosis of 2 compared with HIV-uninfected patients, but varied it from 1 (ie, no increased risk compared with HIV-uninfected patients) to 5 in sensitivity analysis. The natural history of HIV disease was modeled by stratifying health states by CD4 cell counts, as was done in previous studies. 138,165 To incorporate the impact of HAART, we first 2547
4 Table 1. Values Used for the Base Case and Plausible Ranges Used for Sensitivity Analysis* (cont) Variable Base Case (Range ) Source Reference Numbers Cost of HCV-related disease, $ 20, 55, 56, 125, Direct medical costs, monthly Chronic hepatitis 28 Compensated cirrhosis 37 Decompensated cirrhosis 1906 Hepatocellular carcinoma 1243 Treatment costs, $ Monthly drug costs Interferon alfa 426 Pegylated interferon alfa 965 Ribavirin (compounded) 225 Ribavirin (separately marketed) 1653 Physician visits 48 wk wk 384 Laboratory services 48 wk wk 450 HIV disease costs, $ , 108, Monthly care (excluding antiretroviral therapy and resistance testing) CD4 cell count 500 cells/µl 379 CD4 cell count cells/µl 577 CD4 cell count 200 cells/µl 2919 CD4 test 83 HIV RNA test 110 Antiretroviral therapy 706 *HCV indicates hepatitis C virus; HIV, human immunodeficiency virus. Rates are converted into monthly probabilities. If no range is specified, the variable was varied ± 50% in sensitivity analysis. Sources are for the base case estimate and establishment of the plausible range. Data on antiretroviral efficacy from the Dupont 006 trial (mean CD4 cell count, 350 cells/µl; median log HIV RNA, 4.8), zidovudine, lamivudine, and efavirenz arm, with 70% suppressed at 48 weeks; the Johns Hopkins Clinical Cohort trial and the AIDS Clinical Trial Protocol 320 trial, zidovudine, lamivudine, and indinavir arm, with 44% and 60% suppressed at 24 weeks, respectively; and the Community Program for AIDS Research Clinical Trial 046, with 34% suppressed at 12 weeks. See the Methods section for details on the use of calibration to an independent model to incorporate highly active antiretroviral therapy efficacy Represents all costs that occur during 1 treatment episode, including visits, drugs, and laboratory tests. extrapolated average probabilities of CD4 cell decline and HIVrelated mortality using published data Because health states were not stratified by HIV RNA level, we calibrated our simplified model to results obtained with a previously described computer-based simulation model of HIV disease that includes both HIV RNA and CD4 cell count as predictors of disease progression Assuming a hypothetical cohort of 1 million HIV-infected adults with a mean CD4 cell count of 350 cells/µl, the probabilities of CD4 decline and HIV-related mortality were adjusted such that projected life expectancy, QALE, and costs for both models converged. Details of this more comprehensive HIV policy model have been published elsewhere Briefly, it is a state-transition model programmed in C and compiled with Visual C (Microsoft Corp, Redmond, Wash) in which the risks of opportunistic infections and death are stratified by CD4 cell count and the rate of CD4 cell decline is determined by level of HIV RNA. Methods used to derive the transition probabilities for antiretroviral treatment efficacy in this model are described elsewhere and are based on data from several clinical trials Data have also been incorporated regarding the initiation, discontinuation, efficacy, and toxic effects of prophylaxis against major opportunistic infections We assumed that the efficacy of combination regimens for chronic HCV in coinfected patients was the same as in HIVuninfected patients, although we explored lower rates of response in sensitivity analyses ,163 We used data from several studies to estimate a plausible range for the risk of minor and major toxic effects from HCV treatment or HAART. HEALTH-RELATED QUALITY OF LIFE The consequences of mortality and morbidity were incorporated into a single outcome measure by adjusting life expectancy for quality of life on a scale from 0 (death) to 1 (perfect health). Data were not available for the specific health states in our model, which reflect coinfection with HCV and HIV. Therefore, we assumed a multiplicative relationship using the quality weights derived for each disease separately. 185,186 The quality weights for the HCV-specific health states were from published studies using the visual analog scale. We calculated utilities from these values using a rating scale transformation derived by Torrance. 187 The quality weights for the HIVspecific health states were derived from recent works based on data from the HIV Cost and Services Utilization Study. An increasing number of studies 106, ,188,189 document the impact of chronic HCV on health-related quality of life. We conservatively assumed that the quality of life benefit associated with treating moderate hepatitis was captured by averting progression to cirrhosis. We explored the effect of temporary decrements in quality of life associated with treatment (due to adverse effects and medication toxic effects) for chronic HCV. 172,173 COSTS We used previously published estimates for the direct medical costs associated with chronic HCV that were based on a cost accounting approach and combined unit cost estimates for of- 2548
5 fice visits, hospitalizations, laboratory tests, and medications, with resource utilization frequency estimated by an expert panel. 55 Drug costs were based on average wholesale prices. 128,132 Because of a wide discrepancy in costs based on compounded vs separately marketed drugs, a wide plausible range was used in sensitivity analyses. 63,125,130,131 To account for inflation, all costs were converted to constant dollars using the Medical Care Component of the Consumer Price Index. 129 Costs of monthly HIV care were based on previously published data and reflected routine HIV care, antiretroviral drug costs, HIV RNA testing every 3 months, and resistance testing after virologic failure after the first HAART regimen The costs of genotypic resistance tests, CD4 tests, and HIV RNA and HCV RNA assays were obtained from the public use Clinical Diagnostic Laboratory Fee Schedule. 134 Estimates were similar to other sources of costs attributable to HIV and acquired immunodeficiency syndrome. 135 PREDICTIVE VALIDITY OF THE NATURAL HISTORY MODEL We compared the model s predictions of cirrhosis and mortality with data from published studies that were not used for variable estimation. For an HIV-infected cohort with mild chronic HCV, our model predicted a cumulative risk of cirrhosis after 10 years of 16.9% compared with 14.9% reported by Soto et al. 37 The model predicted that 90% of deaths were attributable to HIV disease and that 5% were attributable to HCV compared with the 89% and 5%, respectively, observed by Puoti et al. 17 RESULTS BASE CASE For a cohort of 35-year-old coinfected patients (mean CD4 cell count of 350 cells/µl) with moderate HCV, the average discounted QALE was 83.8 months, and lifetime costs were $ (Table 2). In patients with genotype 1, treatment for HCV provided incremental gains ranging from 6.2 to 7.8 quality-adjusted life-months compared with no therapy. Combination therapy with interferon alfa and ribavirin for 48 weeks dominated combination therapy for 24 weeks and interferon alfa alone because it was more effective and had a lower (more attractive) cost-effectiveness ratio. Compared with no therapy, its incremental cost-effectiveness ratio was $11600 per quality-adjusted life-year (QALY). In comparison, combination therapy with pegylated interferon alfa and ribavirin for 48 weeks provided an additional 1.6 quality-adjusted months of life for a cost of $40000 per QALY. Treatment provided even greater QALE gains in individuals with non-1 genotypes. Combination therapy with interferon alfa and ribavirin for 24 weeks provided 12.3 quality-adjusted life-months and had an incremental cost-effectiveness ratio of $2900 per QALY compared with no therapy. Combination therapy for 48 weeks provided 1.0 additional month of life expectancy and cost $38800 per QALY compared with a 24-week course. In comparison, pegylated interferon alfa and ribavirin for 48 weeks provided an additional 3 weeks and cost $ per QALY gained. For an otherwise identical cohort of coinfected patients with mild chronic HCV, QALE gains were lower, reflecting their overall lower risk of progression to cirrhosis. In those with genotype 1, combination therapy with interferon alfa and ribavirin for 48 weeks increased QALE by 2.2 months and cost $35900 per QALY compared with no therapy. In comparison, pegylated interferon alfa and ribavirin therapy for 48 weeks increased QALE by 3 weeks and cost $ per QALY. In those with genotypes other than 1, interferon alfa and ribavirin administration for 24 weeks increased the QALE by 4.5 months and cost $11900 per QALY compared with no treatment. The additional clinical benefits achieved with 48 weeks of combination therapy with either regular interferon alfa or pegylated interferon alfa ranged from 2 to 3 weeks and cost $ and $ per QALY, respectively. SENSITIVITY ANALYSES Results were most sensitive to the RR of progression to cirrhosis compared with HIV-negative patients, the ability to tolerate HAART, and the discount rate (Figure 2). Results were moderately sensitive to the long-term effectiveness and cost of treatment for HCV. General results were minimally sensitive to the direct medical costs associated with HIV disease, a potential treatment effect in nonresponders, and minor toxic effects. The impact of major toxic effects depended on assumptions made about the necessity for long-term treatment interruptions with HAART. Figure 3 shows the relationship between the RR of progression to cirrhosis in coinfected patients with mild and moderate chronic HCV compared with HIVuninfected patients and the incremental cost-effectiveness ratios for combination therapy compared with the next best treatment strategy. General results for patients with genotype 1 (Figure 3A) and non-1 genotypes (Figure 3B) are similar, although the optimal strategies for treatment differ. For patients with genotype 1, even with an RR of 1 the gains in QALE ranged from 4.04 to 5.12 months. For this conservative scenario, the incremental cost-effectiveness ratio for combination therapy with interferon alfa and ribavirin was $18800 per QALY compared with no therapy. In comparison, treatment with pegylated interferon alfa and ribavirin for 48 weeks was $62000 per QALY. As the RR was increased from 1 (ie, risk is no different than in HIV-uninfected patients) to 5, the incremental benefits associated with treatment increased and the cost-effectiveness ratios became more attractive. The impact on the cost-effectiveness ratios associated with treatment was most pronounced between an RR of 1 and 2, with less effect observed at RRs higher than 3. Changes in the RR had a greater effect on patients with mild HCV than with moderate HCV because the former have a lower baseline probability of disease progression. We explored the potential implications of major toxicity resulting from the use of interferon alfa and ribavirin if such toxic effects resulted in a 50% reduction in the effectiveness of HAART (eg, secondary to a lower rate of adherence or necessity for discontinuation of HAART). Under these exploratory and hypothetical conditions, if 20% of patients experienced major toxicity resulting from 2549
6 Table 2. Costs, Quality-Adjusted Life Expectancy, and Cost-effectiveness of Treatment for HCV* Treatment Strategy Costs, $ Undiscounted Life Expectancy, mo Discounted Life Expectancy, mo C/E Ratio, $/YLS QALM C/E Ratio, $/QALY Coinfected Patients With CD4 Cell Counts of 350 Cells/µL and Moderate Chronic HCV, Genotype 1 No treatment Interferon alfa, 48 wk Dominated Dominated Interferon alfa and ribavirin, 24 wk Dominated Dominated Interferon alfa and ribavirin, 48 wk Pegylated interferon alfa, 48 wk Dominated Dominated Pegylated interferon alfa and ribavirin, 48 wk Coinfected Patients With CD4 Cell Counts of 350 Cells/µL and Moderate Chronic HCV, Non-1 Genotype No treatment Interferon alfa and ribavirin, 24 wk Interferon alfa, 48 wk Dominated Dominated Interferon alfa and ribavirin, 48 wk Pegylated interferon alfa, 48 wk Dominated Dominated Pegylated interferon alfa and ribavirin, 48 wk Coinfected Patients With CD4 Cell Counts of 350 Cells/µL and Mild Chronic HCV, Genotype 1 No treatment Interferon alfa, 48 wk Dominated Dominated Interferon alfa and ribavirin, 24 wk Dominated Dominated Pegylated interferon alfa, 48 wk Dominated Dominated Interferon alfa and ribavirin, 48 wk Pegylated interferon alfa and ribavirin, 48 wk Coinfected Patients With CD4 Cell Counts of 350 Cells/µL and Mild Chronic HCV, Non-1 Genotype No treatment Interferon alfa, 48 wk Dominated Dominated Interferon alfa and ribavirin, 24 wk Interferon alfa and ribavirin, 48 wk Pegylated interferon alfa, 48 wk Dominated Dominated Pegylated interferon alfa and ribavirin, 48 wk Coinfected Patients With CD4 Cell Counts of 200 Cells/µL and Moderate Chronic HCV, Genotype 1 No treatment Interferon alfa, 48 wk Dominated Dominated Interferon alfa and ribavirin, 24 wk Dominated Dominated Pegylated interferon alfa, 48 wk Dominated Dominated Interferon alfa and ribavirin, 48 wk Pegylated interferon alfa and ribavirin, 48 wk Coinfected Patients With CD4 Cell Counts of 200 Cells/µL and Moderate Chronic HCV, Non-1 Genotype No treatment Interferon alfa, 48 wk Dominated Dominated Interferon alfa and ribavirin, 24 wk Interferon alfa and ribavirin, 48 wk Pegylated interferon alfa, 48 wk Dominated Dominated Pegylated interferon alfa and ribavirin, 48 wk Coinfected Patients With CD4 Cell Counts of 200 Cells/µL and Mild Chronic HCV, Genotype 1 No treatment Interferon alfa, 48 wk Dominated Dominated Interferon alfa and ribavirin, 24 wk Dominated Dominated Pegylated interferon alfa, 48 wk Dominated Dominated Interferon alfa and ribavirin, 48 wk Pegylated interferon alfa and ribavirin, 48 wk Coinfected Patients With CD4 Cell Counts of 200 Cells/µL and Mild Chronic HCV, Non-1 Genotype No treatment Interferon alfa, 48 wk Dominated Dominated Interferon alfa and ribavirin, 24 wk Interferon alfa and ribavirin, 48 wk Pegylated interferon alfa, 48 wk Dominated Dominated Pegylated interferon alfa and ribavirin, 48 wk *HCV indicates hepatitis C virus; HIV, human immunodeficiency virus; QALM, quality-adjusted life-month; C/E ratio, incremental cost-effectiveness ratio; YLS, years of life saved; and QALY, quality-adjusted life-year. Interferon alfa was given as 3 MIU 3 times weekly; pegylated interferon alfa, 1.5 µg/kg once weekly; and ribavirin, mg/d (in combination with interferon alfa) or 800 mg/d (in combination with pegylated interferon alfa). The difference in cost divided by the difference in life expectancy or quality-adjusted life expectancy for each strategy compared with the next best strategy. All strategies are assumed to be equally available. This strategy is strongly dominated because it is more costly and less effective than an available alternative strategy. This strategy is weakly dominated (ie, eliminated by extended dominance) because it is less effective and is associated with a less attractive C/E ratio than an available alternative strategy. 2550
7 A Discount Rate (0%-10%) Cost of Interferon Alfa (75%-125%) Cost of Pegylated Interferon (75%-125%) Cost of Ribavirin (25%-500%) HCV Treatment Efficacy (0%-30%) Transient HCV Treatment Effect in Nonresponders Relapse After a Sustained Response (0%-20% per Year) A Cost-effectiveness Ratio, $/QALY Mild Chronic Hepatitis, Genotype 1 Moderate Chronic Hepatitis, Genotype 1 Peg + Rib (48 wk) vs IFN + Rib (48 wk) IFN + Rib (48 wk) vs No Treatment HAART Efficacy (±50%) Disutility of Minor Toxicity (±10%) B Discount Rate (0%-10%) Cost of Interferon Alfa (75%-125%) Cost of Pegylated Interferon (75%-125%) Cost of Ribavirin (25%-500%) HCV Treatment Efficacy (0%-30%) Transient HCV Treatment Effect in Nonresponders Relapse After a Sustained Response (0%-20% per Year) B Cost-effectiveness Ratio, $/QALY Mild Chronic Hepatitis, Non-1 Genotype Moderate Chronic Hepatitis, Non-1 Genotype IFN + Rib (48 wk) vs IFN + Rib (24 wk) IFN + Rib (24 wk) vs No Treatment HAART Efficacy (±50%) Disutility of Minor Toxicity (±10%) Incremental Cost-effectivenss Ratio, $ Relative Risk of Progression to Cirrhosis in Coinfected vs HIV-Uninfected Patients Figure 2. One-way sensitivity analyses of selected model input variables. The impact on the incremental cost-effectiveness ratio of 48 weeks of combination therapy with pegylated interferon alfa and ribavirin in coinfected patients with genotype 1 (compared with 48 weeks of interferon alfa and ribavirin therapy) (A) and of 48 weeks of interferon alfa and ribavirin therapy in coinfected patients with non-1 genotypes (compared with 24 weeks of combination therapy) (B). The dashed lines show the base case values, and the horizontal bars represent the range of incremental cost-effectiveness ratios resulting from varying each variable over its plausible range. HCV indicates hepatitis C infection; HAART, highly active antiretroviral therapy. 48 weeks of combination therapy with pegylated interferon alfa and ribavirin, the incremental cost-effectiveness ratio increased from $40600 to $69000 per QALY with genotype 1 compared with the next best strategy. In contrast, minor adverse affects from treatment had little effect on the results. In coinfected patients with mild chronic hepatitis, results were sensitive to assumptions about the impact of treatment on quality of life. The life expectancy benefits from treating mild HCV ranged from 1 to 2 weeks, whereas the QALE benefits ranged from 2 weeks to nearly 2 months (Table 2). These results demonstrate that a substantial proportion of the clinical benefit associated with treating mild chronic HCV results from averting progression to moderate HCV, which is associated with a poorer quality of life. For patients with moderate HCV and genotype 1, the cost-effectiveness of pegylated interferon alfa and ribavirin therapy was sensitive to assumptions about the potential quality of life benefits associated with a single weekly injection compared with the 3 weekly injections Figure 3. One-way sensitivity analyses of the relative risk of progression to cirrhosis in coinfected patients compared with human immunodeficiency virus (HIV) uninfected patients. A, The effect of varying the relative risk between 1 and 5 in patients with genotype 1 on the incremental cost-effectiveness ratio of different treatment strategies for mild and moderate chronic hepatitis C virus (HCV). B, The effect of varying the relative risk between 1 and 5 in patients with non-1 genotypes on the incremental cost-effectiveness ratio of different treatment strategies for mild and moderate chronic HCV. Peg indicates pegylated interferon alfa; IFN, interferon alfa; Rib, ribavirin; and QALY, quality-adjusted life-year. needed for regular interferon alfa. We conducted a threshold analysis to identify the quality of life decrement associated with 3 weekly subcutaneous injections that would make pegylated interferon alfa plus ribavirin the preferred strategy. We found that if the quality of life during 48 weeks of treatment was reduced by more than 10%, combination therapy with pegylated interferon alfa and ribavirin dominated conventional interferon alfa based strategies. OTHER ANALYSES We conducted an exploratory analysis for patients with CD4 cell counts below 200 cells/µl. For coinfected patients in later stages of HIV with CD4 cell counts below 200 cells/µl and with moderate HCV, the general results were similar, although all cost-effectiveness ratios were higher, reflecting the increased competing morbidity and mortality associated with HIV disease (Table 2). In sensitivity analysis, when the efficacy of HAART was increased such that life expectancy approached that of 2551
8 the average patient in the base case cohort, the costeffectiveness ratios associated with HCV treatment consistently declined below $50000 per QALY. We also evaluated the cost-effectiveness of interferon alfa based monotherapy in coinfected patients intolerant of ribavirin. For these patients, monotherapy with pegylated interferon alfa was the most effective option. Compared with regular interferon alfa therapy, the costeffectiveness ratio associated with use of pegylated interferon alfa was $29400 per QALY in patients with genotype 1 and $20300 per QALY in patients with non-1 genotypes. COMMENT We found that for HIV-infected patients with moderate chronic HCV and a mean CD4 cell count of 350 cells/ µl, treatment with combination therapy using an interferon alfa based regimen reduced the incidence of cirrhosis and provided substantial gains in QALE. Regardless of genotype, the cost-effectiveness of interferon alfa and ribavirin therapy for patients with moderate HCV was less than $50000 per QALY. The incremental benefits and cost-effectiveness of pegylated interferon alfa and ribavirin therapy for 48 weeks (compared with use of regular interferon alfa and ribavirin for 24 and 48 weeks) differed between individuals with genotype 1 and non-1 genotypes. Combination therapy (pegylated interferon alfa and ribavirin) in patients with moderate HCV and genotype 1 cost less than $50000 per QALY. In contrast, because treatment is relatively more effective in patients with non-1 genotypes, the incremental gain in QALE associated with the more costly pegylated interferon alfa, compared with regular interferon alfa and ribavirin, was relatively small. Accordingly, although the cost-effectiveness ratio of interferon alfa and ribavirin therapy for non-1 genotypes was less than $50000 per QALY compared with no treatment, combination therapy with pegylated interferon alfa exceeded $ per QALY. For all patients, regardless of genotype, if ribavirin was not tolerated, monotherapy with pegylated interferon alfa was the best option. There is considerable uncertainty in the natural history of chronic HCV, although recent data indicate that the RR of liver disease progression seems to be increased in coinfected patients compared with HIVuninfected patients. 2,6,17,19,24-37 We assumed an RR of 2 for progression of liver disease compared with HIVuninfected patients, but all analyses were repeated using the more conservative assumption of an RR of 1. When we assumed that the risk of liver disease was no different than in HIV-uninfected patients, the life expectancy gains and cost-effectiveness ratios associated with treatment for moderate HCV were comparable or superior to those provided by prophylaxis for opportunistic infections. In contrast, the cost-effectiveness of treatment for mild HCV was more sensitive to assumptions about the RR of HCV progression in HIV-infected vs HIVuninfected patients. Unless this RR was 2 or greater, the incremental cost-effectiveness ratio for combination therapy exceeded $50000 per QALY. These results highlight the importance of obtaining better data to inform the attributable excess risk associated with HIV coinfection. The results of this analysis indicate that the information with the greatest potential value will be that providing detail on whether the RR is closer to 1 or 2 in coinfected compared with HIV-uninfected patients. Detailed data on the interactions between the tolerance and effectiveness of HAART and treatment for HCV are not yet available, to our knowledge. Therefore, the goal of our exploratory analyses examining the implications of these interactions was to provide qualitative insight into the importance of certain variables but not to answer questions for which data are not yet available. We found that changes in HCV treatment efficacy and minor toxic effects, when varied over a wide plausible range, had a minimal effect on our major results. In contrast, there was a greater effect of major toxic effects when they resulted in a lower efficacy of HAART. Without the clinical benefits associated with HAART, patients experienced HIV disease progression before the development HCV-related end-stage liver disease; thus, they received minimal life-extending benefit from HCV treatment despite accruing its treatment costs. On the other hand, it is equally plausible that treatment for HCV could improve the probability of tolerance to HAART by reducing the risk of hepatotoxicity. If this turns out to be true, HCV treatment will provide even greater clinical benefits and be even more cost-effective. These results indicate that better information on whether HCV treatment limits or enhances tolerability to HAART should be a high priority. This study has several additional limitations. The natural history of HCV is uncertain in HIV-uninfected patients and in HIV-infected patients undergoing HAART. The efficacy and toxic effects of HCV therapy in HIVinfected patients have only been assessed in relatively small clinical studies. There are no published data, to our knowledge, on the quality of life associated with coinfection, stratified by stage of HIV disease, that are suitable for weighting the specific health states in our model. We assumed a multiplicative relationship between the quality weights for HIV and HCV, but empiric data to inform the validity of this assumption are lacking. The quality of life benefits associated with use of pegylated interferon alfa, recently approved by the Food and Drug Association for treatment of chronic HCV, remain unclear. Our results indicate that, in theory, if HCV could be treated before an individual s need for HAART (thereby eliminating any potential negative consequences of HCV treatment on tolerance of HAART or preventing hepatotoxicity of HAART), such a strategy would likely be effective and cost-effective. It is possible that temporary interruptions in HAART due to HCV treatment toxic effects will be less detrimental than we assumed; in this case, the negative impact of toxic effects was overestimated. Our results do not address the question of whether treatment for HCV should be administered much earlier in the disease progression (eg, CD4 cell counts 500 cells/µl). Data in these patients are lacking but will be important to incorporate when available. A key attribute of a model such as this one is that as better data become available, the impact of new information can be rapidly evaluated. 2552
9 In the meantime, however, clinical decisions for individuals and broader public policy decisions must proceed before all uncertainties are resolved. Evaluating the effectiveness of HCV treatment in coinfected patients requires specification of the natural history of both diseases (HIV and HCV), consideration of the heterogeneity of risk, treatment efficacy and toxic effects, and accessibility, feasibility, and affordability of medication and health care. Data are not available for all of this information, and our analysis therefore required multiple assumptions. However, even in the future, a single study will not be able to simultaneously consider all of these components and assess all possible strategies for all relevant populations. This analysis was conducted to provide qualitative and quantitative insight into the relative importance of different components of the treatment process and to investigate how results would change when values of key variables were changed. By identifying the most influential variables, these results may be used to help prioritize and guide data collection efforts. Our results suggest that the most critical variables for future research include the RR of progression in chronic HCV, the health-related quality of life in coinfected patients with mild HCV, and the ability to tolerate treatment for HIV with HAART. Based on conservative assumptions about the natural history of HCV and treatment efficacy, coinfected patients with a CD4 cell count of at least 350 cells/µl and moderate chronic HCV should be treated with combination therapy. In patients able to tolerate combination therapy, the choice of pegylated vs standard interferon alfa (in combination with ribavirin) depends on the genotype (and likelihood of sustained treatment response to traditional interferon alfa and ribavirin) and assumptions about the quality of life decrement associated with 3 subcutaneous injections per week. Patients who are intolerant of ribavirin therapy should be treated with pegylated interferon alfa regardless of genotype. Accepted for publication April 24, This study was presented in part at the meeting of the Society of Medical Decision Making, Cincinnati, Ohio, September 25, Corresponding author and reprints: Sue J. Goldie, MD, MPH, Harvard Program on the Economic Evaluation of Medical Technology, 718 Huntington Ave, Second Floor, Boston, MA ( sgoldie@hsph.harvard.edu). REFERENCES 1. Bonacini M, Puoti M. Hepatitis C in patients with human immunodeficiency virus infection: diagnosis, natural history, meta-analysis of sexual and vertical transmission, and therapeutic issues. Arch Intern Med. 2000;160: Dieterich DT. Hepatitis C virus and human immunodeficiency virus: clinical issues in coinfection. Am J Med. 1999;107(suppl):79S-84S. 3. Soriano V, Rodriguez-Rosado R, Garcia-Samaniego J. Management of chronic hepatitis C in HIV-infected patients. AIDS. 1999;13: Palella FJ Jr, Delaney KM, Moorman AC, et al. Declining morbidity and mortality among patients with advanced human immunodeficiency virus infection. N Engl J Med. 1998;338: Kaplan JE, Hanson D, Dworkin MS, et al. Epidemiology of human immunodeficiency virus associated opportunistic infections in the United States in the era of highly active antiretroviral therapy. Clin Infect Dis. 2000;30(suppl 1):S5- S Di Martino V, Ezenfis J, Tainturier MH, et al. Impact of HIV coinfection on the long-term outcome of HCV cirrhosis. Paper presented at: 8th Conference on Retroviruses and Opportunistic Infections; February 4-8, 2001; Chicago, Ill. Available at: htm. Accessed April 23, Abstract Soriano V, Garcia-Samaniego J, Rodriguez-Rosado R, Gonzalez J, Pedreira J. Hepatitis C and HIV infection: biological, clinical, and therapeutic implications. J Hepatol. 1999;31: Seeff LB. Natural history of hepatitis C. Hepatology. 1997;26(suppl 1):21S- 28S. 9. Alter HJ, Seeff LB. Recovery, persistence, and sequelae in hepatitis C virus infection: a perspective on long-term outcome. Semin Liver Dis. 2000;20: Bica I, McGovern B, Dhar R, et al. Increasing mortality due to end-stage liver disease in patients with human immunodeficiency virus infection. Clin Infect Dis. 2001;32: Valdez H, Chowdhry TK, Asaad R, et al. Changing spectrum of mortality due to human immunodeficiency virus: analysis of 260 deaths during Clin Infect Dis. 2001;32: Soriano V, Martin-Carbonero L, Garcia-Samaniego J, Puoti M. Mortality due to chronic viral liver disease among patients infected with human immunodeficiency virus. Clin Infect Dis. 2001;33: Bonnet F, Morlat P, Chene G, et al. Causes of death among HIV-infected patients in the era of highly active antiretroviral therapy: Aquitane Cohort France, Paper presented at: 8th Conference on Retroviruses and Opportunistic Infections; February 4-8, 2001; Chicago, Ill. Available at: Accessed April 23, Abstract Di Perri G, Raiteri R, Bonora S, et al. Liver failure from HCV as the current leading cause of death in HIV-infected patients in Northern Italy. Paper presented at: 8th Conference on Retroviruses and Opportunistic Infections; February 4-8, 2001; Chicago, Ill. Available at: abstracts/abstracts/573.htm. Accessed April 23, Abstract Ahmad S, Pulvirenti J, Shastri P, Irfan M, Ariga D. Death in HIV-infected inpatients in the HAART era: an evaluation of mortality in an inner-city hospital. Paper presented at: 8th Conference on Retroviruses and Opportunistic Infections; February 4-8,2001;Chicago,Ill.Availableat: abstracts/abstracts/300.htm. Accessed April 23, Abstract Justice A, Chang C, Fusco J, et al. Extrapolating long-term HIV/AIDS survival in the post-haart era. In: Program and abstracts of the 39th International Conference on Antimicrobial Agents and Chemotherapy; September 26-29, 1999; San Francisco, Calif. Abstract Puoti M, Spinetti A, Ghezzi A, et al. Mortality for liver disease in patients with HIV infection: a cohort study. J Acquir Immune Defic Syndr. 2000;24: Martin-Carbonero L, Soriano V, Valencia M, Lopez M, Gonzalez-Lahoz J. Impact of chronic viral hepatitis on hospital admission and mortality in HIVinfected patients. Paper presented at: 8th Conference on Retroviruses and Opportunistic Infections; February 4-8, 2001; Chicago, Ill. Available at: Accessed April 23, 2001). Abstract Klein M, Lalonde R, Suissa S. HCV coinfection is associated with increased morbidity and mortality among HIV-infected patients. Paper presented at: 8th Conference on Retroviruses and Opportunistic Infections; February 4-8, 2001; Chicago, Ill. Available at: abstracts/569.htm. Accessed April 23, Abstract Kim WR, Gross JB Jr, Poterucha JJ, Locke GR, Dickson ER. Outcome of hospital care of liver disease associated with hepatitis C in the United States. Hepatology. 2001;33: Telfer P, Sabin C, Devereux H, Scott F, Dusheiko G, Lee C. The progression of HCV-associated liver disease in a cohort of haemophilic patients. Br J Haematol. 1994;87: Cribier B, Rey D, Schmitt C, et al. High hepatitis C viraemia and impaired antibody response in patients coinfected with HIV. AIDS. 1995;1995: Beld M, Penning M, Lukashov V, et al. Evidence that both HIV and HIV-induced immunodeficiency enhance HCV replication among HCV seroconverters. Virology. 1998;244: Thomas DL, Shih JW, Alter HJ, et al. Effect of human immunodeficiency virus on hepatitis C virus infection among injecting drug users. J Infect Dis. 1996; 174: Eyster ME, Diamondstone LS, Lien JM, Ehmann WC, Quan S, Goedert JJ. Natural history of hepatitis C virus infection in multitransfused hemophiliacs: effect of coinfection with human immunodeficiency virus: the Multicenter Hemophilia Cohort Study. J Acquir Immune Defic Syndr. 1993;6: Graham C, Baden LR, Yu E, et al. Influence of human immunodeficiency virus infection on the course of hepatitis C virus infection: a meta-analysis. Clin Infect Dis. 2001;33: Allory Y, Charlotte F, Benhamou Y, Opolon P, Le Charpentier Y, Poynard T. Impact of human immunodeficiency virus infection on the histological features of chronic hepatitis C: a case-control study. Hum Pathol. 2000;31: Pol S, Lamorthe B, Thi NT, et al. Retrospective analysis of the impact of HIV infection and alcohol use on chronic hepatitis C in a large cohort of drug users. J Hepatol. 1998;28: Ragni MV, Belle SH. Impact of human immunodeficiency virus infection on progression to end-stage liver disease in individuals with hemophilia and hepatitis C virus infection. J Infect Dis. 2001;183: Benhamou Y, Bochet M, Di Martino V, et al, for the Multivirc Group. Liver fi- 2553
Technology appraisal guidance Published: 22 September 2010 nice.org.uk/guidance/ta200
 Peginterferon alfa and ribavirin for the treatment of chronic hepatitis C Technology appraisal guidance Published: 22 September 2010 nice.org.uk/guidance/ta200 NICE 2018. All rights reserved. Subject to
Peginterferon alfa and ribavirin for the treatment of chronic hepatitis C Technology appraisal guidance Published: 22 September 2010 nice.org.uk/guidance/ta200 NICE 2018. All rights reserved. Subject to
Boceprevir for previously untreated patients with chronic hepatitis C Genotype 1 infection: a US-based cost-effectiveness modeling study
 Ferrante et al. BMC Infectious Diseases 2013, 13:190 RESEARCH ARTICLE Open Access Boceprevir for previously untreated patients with chronic hepatitis C Genotype 1 infection: a US-based cost-effectiveness
Ferrante et al. BMC Infectious Diseases 2013, 13:190 RESEARCH ARTICLE Open Access Boceprevir for previously untreated patients with chronic hepatitis C Genotype 1 infection: a US-based cost-effectiveness
Peginterferon alfa and ribavirin for the treatment of chronic hepatitis C. Part review of NICE technology appraisal guidance 75 and 106
 Issue date: September 2010 Peginterferon alfa and ribavirin for the treatment of chronic hepatitis C Part review of NICE technology appraisal guidance 75 and 106 National Institute for Health and Clinical
Issue date: September 2010 Peginterferon alfa and ribavirin for the treatment of chronic hepatitis C Part review of NICE technology appraisal guidance 75 and 106 National Institute for Health and Clinical
Epidemiological and economic impact of potential increased hepatitis C treatment uptake in Australia
 Epidemiological and economic impact of potential increased hepatitis C treatment uptake in Australia 2010 2010 ISBN 978 0 7334 2892-0 This publication is available at Internet address http://www.nchecr.unsw.edu.au
Epidemiological and economic impact of potential increased hepatitis C treatment uptake in Australia 2010 2010 ISBN 978 0 7334 2892-0 This publication is available at Internet address http://www.nchecr.unsw.edu.au
Update on HIV-HCV Epidemiology and Natural History
 Update on HIV-HCV Epidemiology and Natural History Jennifer Price, MD Assistant Clinical Professor of Medicine University of California, San Francisco Learning Objectives Upon completion of this presentation,
Update on HIV-HCV Epidemiology and Natural History Jennifer Price, MD Assistant Clinical Professor of Medicine University of California, San Francisco Learning Objectives Upon completion of this presentation,
Scottish Medicines Consortium
 Scottish Medicines Consortium pegylated interferon α 2b (ViraferonPeg ), 50, 80, 100, 120 or 150 micrograms powder for solution for injection in pre-filled pen, in combination with ribavirin (Rebetol ),
Scottish Medicines Consortium pegylated interferon α 2b (ViraferonPeg ), 50, 80, 100, 120 or 150 micrograms powder for solution for injection in pre-filled pen, in combination with ribavirin (Rebetol ),
Why make this statement?
 HCV Council 2014 10 clinical practice statements were evaluated by the Council A review of the available literature was conducted The level of support and level of evidence for the statements were discussed
HCV Council 2014 10 clinical practice statements were evaluated by the Council A review of the available literature was conducted The level of support and level of evidence for the statements were discussed
ةي : لآا ةرقبلا ةروس
 سورة البقرة: اآلية HCV RELAPSERS AND NONRESPONDERS: How to deal with them? BY Prof. Mohamed Sharaf-Eldin Prof. of Hepatology and Gastroenterology Tanta University Achieving SVR The ability to achieve a
سورة البقرة: اآلية HCV RELAPSERS AND NONRESPONDERS: How to deal with them? BY Prof. Mohamed Sharaf-Eldin Prof. of Hepatology and Gastroenterology Tanta University Achieving SVR The ability to achieve a
The cost-effectiveness of expanded testing for primary HIV infection Coco A
 The cost-effectiveness of expanded testing for primary HIV infection Coco A Record Status This is a critical abstract of an economic evaluation that meets the criteria for inclusion on NHS EED. Each abstract
The cost-effectiveness of expanded testing for primary HIV infection Coco A Record Status This is a critical abstract of an economic evaluation that meets the criteria for inclusion on NHS EED. Each abstract
Technology appraisal guidance Published: 27 November 2013 nice.org.uk/guidance/ta300
 Peginterferon alfa and ribavirin for treating chronic hepatitis C in children and young people Technology appraisal guidance Published: 27 November 2013 nice.org.uk/guidance/ta300 NICE 2018. All rights
Peginterferon alfa and ribavirin for treating chronic hepatitis C in children and young people Technology appraisal guidance Published: 27 November 2013 nice.org.uk/guidance/ta300 NICE 2018. All rights
Cost effectiveness of sofosbuvir and ledipasvir (Harvoni ) in combination with other medicinal products for the treatment of hepatitis C infection
 Cost effectiveness of sofosbuvir and ledipasvir (Harvoni ) in combination with other medicinal products for the treatment of hepatitis C infection The NCPE has issued a recommendation regarding the cost
Cost effectiveness of sofosbuvir and ledipasvir (Harvoni ) in combination with other medicinal products for the treatment of hepatitis C infection The NCPE has issued a recommendation regarding the cost
Over the past decade, the introduction of
 MANAGEMENT OF CHRONIC HEPATITIS C IN HIV-INFECTED PATIENTS: CLINICAL EXPERIENCE WITH PEGYLATED INTERFERON α PLUS RIBAVIRIN Raymond T. Chung, MD* ABSTRACT Coinfection with hepatitis C virus (HCV) is common
MANAGEMENT OF CHRONIC HEPATITIS C IN HIV-INFECTED PATIENTS: CLINICAL EXPERIENCE WITH PEGYLATED INTERFERON α PLUS RIBAVIRIN Raymond T. Chung, MD* ABSTRACT Coinfection with hepatitis C virus (HCV) is common
Supplementary Online Content
 Supplementary Online Content Chahal HS, Marseille EA, Tice JA, et al. Cost-effectiveness of early treatment of hepatitis C virus genotype 1 by stage of liver fibrosis in a US treatment-naive population.
Supplementary Online Content Chahal HS, Marseille EA, Tice JA, et al. Cost-effectiveness of early treatment of hepatitis C virus genotype 1 by stage of liver fibrosis in a US treatment-naive population.
Economic issues in Hepatitis C. Richard Grieve London School of Hygiene and Tropical Medicine
 Economic issues in Hepatitis C Richard Grieve London School of Hygiene and Tropical Medicine Acknowledgements Mild Hepatitis C study Principal Investigators: Howard Thomas, Janice Main Centre co-ordinators
Economic issues in Hepatitis C Richard Grieve London School of Hygiene and Tropical Medicine Acknowledgements Mild Hepatitis C study Principal Investigators: Howard Thomas, Janice Main Centre co-ordinators
Treatment Options in HCV Relapsers and Nonresponders. Raymond T. Chung, M.D.
 Session IV Treatment Options in HCV Relapsers and Nonresponders Raymond T. Chung, M.D. Director of Hepatology, Massachusetts General Hospital, Associate Professor of Medicine, Harvard Medical School, Boston,
Session IV Treatment Options in HCV Relapsers and Nonresponders Raymond T. Chung, M.D. Director of Hepatology, Massachusetts General Hospital, Associate Professor of Medicine, Harvard Medical School, Boston,
Entecavir for the treatment of chronic hepatitis B infection
 DOI: 10.3310/hta13suppl3/05 Health Technology Assessment 2009; Vol. 13: Suppl. 3 Entecavir for the treatment of chronic hepatitis B infection J Shepherd,* E Gospodarevskaya, G Frampton and K Cooper Southampton
DOI: 10.3310/hta13suppl3/05 Health Technology Assessment 2009; Vol. 13: Suppl. 3 Entecavir for the treatment of chronic hepatitis B infection J Shepherd,* E Gospodarevskaya, G Frampton and K Cooper Southampton
Cost-effectiveness of hepatic venous pressure gradient measurements for prophylaxis of variceal re-bleeding Raines D L, Dupont A W, Arguedas M R
 Cost-effectiveness of hepatic venous pressure gradient measurements for prophylaxis of variceal re-bleeding Raines D L, Dupont A W, Arguedas M R Record Status This is a critical abstract of an economic
Cost-effectiveness of hepatic venous pressure gradient measurements for prophylaxis of variceal re-bleeding Raines D L, Dupont A W, Arguedas M R Record Status This is a critical abstract of an economic
The medical management of hepatitis C
 CLINICAL EXPERIENCE WITH PEGYLATED INTERFERON α-2a PLUS RIBAVIRIN FOR CHRONIC HEPATITIS C VIRUS INFECTION IN PATIENTS INFECTED WITH HIV: THE APRICOT STUDY Douglas T. Dieterich, MD* ABSTRACT Currently,
CLINICAL EXPERIENCE WITH PEGYLATED INTERFERON α-2a PLUS RIBAVIRIN FOR CHRONIC HEPATITIS C VIRUS INFECTION IN PATIENTS INFECTED WITH HIV: THE APRICOT STUDY Douglas T. Dieterich, MD* ABSTRACT Currently,
Technology appraisal guidance Published: 25 November 2015 nice.org.uk/guidance/ta364
 Daclatasvir for treating chronic hepatitis C Technology appraisal guidance Published: 25 November 2015 nice.org.uk/guidance/ta364 NICE 2018. All rights reserved. Subject to Notice of rights (https://www.nice.org.uk/terms-and-conditions#notice-ofrights).
Daclatasvir for treating chronic hepatitis C Technology appraisal guidance Published: 25 November 2015 nice.org.uk/guidance/ta364 NICE 2018. All rights reserved. Subject to Notice of rights (https://www.nice.org.uk/terms-and-conditions#notice-ofrights).
Pegasys Pegintron Ribavirin
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.01.47 Subsection: Anti-infective nts Original Policy Date: January 1, 2019 Subject: Pegasys Pegintron
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.01.47 Subsection: Anti-infective nts Original Policy Date: January 1, 2019 Subject: Pegasys Pegintron
Antiviral therapy guidelines for the general population
 Discussion 10 Chapter 10 Hepatitis C a worldwide problem More than 170 million people worldwide suffer from chronic hepatitis C. Its prevalence is 2% in industrialized countries. 1 Approximately 20% of
Discussion 10 Chapter 10 Hepatitis C a worldwide problem More than 170 million people worldwide suffer from chronic hepatitis C. Its prevalence is 2% in industrialized countries. 1 Approximately 20% of
HIV coinfection and HCC
 HIV coinfection and HCC 3 rd APASL STC on HCC 21 st -23 rd Nov 2013 Cebu, Phillippines George KK Lau MBBS (HK), MRCP(UK), FHKCP, FHKAM (GI), MD(HK), FRCP (Edin, Lond) Consultant, Humanity and Health GI
HIV coinfection and HCC 3 rd APASL STC on HCC 21 st -23 rd Nov 2013 Cebu, Phillippines George KK Lau MBBS (HK), MRCP(UK), FHKCP, FHKAM (GI), MD(HK), FRCP (Edin, Lond) Consultant, Humanity and Health GI
Technology appraisal guidance Published: 25 November 2015 nice.org.uk/guidance/ta363
 Ledipasvir sofosbuvir for treating chronic hepatitis C Technology appraisal guidance Published: 25 November 2015 nice.org.uk/guidance/ta363 NICE 2018. All rights reserved. Subject to Notice of rights (https://www.nice.org.uk/terms-and-conditions#notice-ofrights).
Ledipasvir sofosbuvir for treating chronic hepatitis C Technology appraisal guidance Published: 25 November 2015 nice.org.uk/guidance/ta363 NICE 2018. All rights reserved. Subject to Notice of rights (https://www.nice.org.uk/terms-and-conditions#notice-ofrights).
Economic analysis of initial HIV treatment: efavirenz- versus indinavir-containing triple therapy Caro J J, O'Brien J A, Miglaccio-Walle K, Raggio G
 Economic analysis of initial HIV treatment: efavirenz- versus indinavir-containing triple therapy Caro J J, O'Brien J A, Miglaccio-Walle K, Raggio G Record Status This is a critical abstract of an economic
Economic analysis of initial HIV treatment: efavirenz- versus indinavir-containing triple therapy Caro J J, O'Brien J A, Miglaccio-Walle K, Raggio G Record Status This is a critical abstract of an economic
Anemia in the Treatment of Hepatitis C Virus Infection
 SUPPLEMENT ARTICLE Anemia in the Treatment of Hepatitis C Virus Infection Mark S. Sulkowski Center for Viral Hepatitis, Johns Hopkins University, Baltimore, Maryland Hepatitis C virus (HCV) infection is
SUPPLEMENT ARTICLE Anemia in the Treatment of Hepatitis C Virus Infection Mark S. Sulkowski Center for Viral Hepatitis, Johns Hopkins University, Baltimore, Maryland Hepatitis C virus (HCV) infection is
Setting The setting was the community. The economic study was carried out in the USA.
 Cost-effectiveness analysis of NSAIDs, NSAIDs with concomitant therapy to prevent gastrointestinal toxicity, and COX-2 specific inhibitors in the treatment of rheumatoid arthritis Yun H R, Bae S C Record
Cost-effectiveness analysis of NSAIDs, NSAIDs with concomitant therapy to prevent gastrointestinal toxicity, and COX-2 specific inhibitors in the treatment of rheumatoid arthritis Yun H R, Bae S C Record
Management of CHC G1 patients who are relapsers or non-responders to Peg IFN and RBV therapy: Wait or Triple Therapy?
 Management of CHC G1 patients who are relapsers or non-responders to Peg IFN and RBV therapy: Wait or Triple Therapy? Prof. Teerha Piratvisuth NKC Institute of Gastroenterology and Hepatology Prince of
Management of CHC G1 patients who are relapsers or non-responders to Peg IFN and RBV therapy: Wait or Triple Therapy? Prof. Teerha Piratvisuth NKC Institute of Gastroenterology and Hepatology Prince of
HCV care after cure. This program is supported by educational grants from
 HCV care after cure This program is supported by educational grants from Raffaele Bruno,MD Department of Infectious Diseases, Hepatology Outpatients Unit University of Pavia Fondazione IRCCS Policlinico
HCV care after cure This program is supported by educational grants from Raffaele Bruno,MD Department of Infectious Diseases, Hepatology Outpatients Unit University of Pavia Fondazione IRCCS Policlinico
Viral Hepatitis The Preventive Potential of Antiviral Therapy. Thomas Berg
 Viral Hepatitis The Preventive Potential of Antiviral Therapy Thomas Berg Therapeutic and preventive strategies in patients with hepatitis virus infection Treatment of acute infection Treatment of chronic
Viral Hepatitis The Preventive Potential of Antiviral Therapy Thomas Berg Therapeutic and preventive strategies in patients with hepatitis virus infection Treatment of acute infection Treatment of chronic
Technology appraisal guidance Published: 21 February 2018 nice.org.uk/guidance/ta507
 Sofosbuvir velpatasvir voxilaprevir for treating chronic hepatitis C Technology appraisal guidance Published: 21 February 2018 nice.org.uk/guidance/ta507 NICE 2018. All rights reserved. Subject to Notice
Sofosbuvir velpatasvir voxilaprevir for treating chronic hepatitis C Technology appraisal guidance Published: 21 February 2018 nice.org.uk/guidance/ta507 NICE 2018. All rights reserved. Subject to Notice
HIV. KEY WORDS: HIV, esophageal varices, endoscopic injection sclerotherapy (EIS), endoscopic variceal ligation (EVL)
 2013; 19: 25 32 HIV 1, 2 1 1 1 1 HIV 7 3 HCV, 2 HBV B C 2 HCC 2 F 2 RC 1 2.1 F 1 RC 0 30.4 4 HCV 9 HCC 13.5 3 HCC 1 1 AIDS B C HIV HIV HCV HIV KEY WORDS: HIV, esophageal varices, endoscopic injection sclerotherapy
2013; 19: 25 32 HIV 1, 2 1 1 1 1 HIV 7 3 HCV, 2 HBV B C 2 HCC 2 F 2 RC 1 2.1 F 1 RC 0 30.4 4 HCV 9 HCC 13.5 3 HCC 1 1 AIDS B C HIV HIV HCV HIV KEY WORDS: HIV, esophageal varices, endoscopic injection sclerotherapy
About the National Centre for Pharmacoeconomics
 Cost effectiveness of sofosbuvir (in combination with either ribavirin or pegylated interferon + ribavirin) (Sovaldi ) for the treatment of hepatitis C infection The NCPE has issued a recommendation regarding
Cost effectiveness of sofosbuvir (in combination with either ribavirin or pegylated interferon + ribavirin) (Sovaldi ) for the treatment of hepatitis C infection The NCPE has issued a recommendation regarding
Assessment of cost-effectiveness of universal hepatitis B immunization in a low-income country with intermediate endemicity using a Markov model
 Assessment of cost-effectiveness of universal hepatitis B immunization in a low-income country with intermediate endemicity using a Markov model Aggarwal R, Ghoshal U C, Naik S R Record Status This is
Assessment of cost-effectiveness of universal hepatitis B immunization in a low-income country with intermediate endemicity using a Markov model Aggarwal R, Ghoshal U C, Naik S R Record Status This is
Health and economic consequences of HCV lookback Pereira A
 Health and economic consequences of HCV lookback Pereira A Record Status This is a critical abstract of an economic evaluation that meets the criteria for inclusion on NHS EED. Each abstract contains a
Health and economic consequences of HCV lookback Pereira A Record Status This is a critical abstract of an economic evaluation that meets the criteria for inclusion on NHS EED. Each abstract contains a
British HIV Association Guidelines for the Management of Hepatitis Viruses in Adults Infected with HIV 2013 Appendix 2
 British HIV Association Guidelines for the Management of Hepatitis Viruses in Adults Infected with HIV 2013 Appendix 2 Systematic literature search 2.1 Questions and PICO criteria Data bases: Medline,
British HIV Association Guidelines for the Management of Hepatitis Viruses in Adults Infected with HIV 2013 Appendix 2 Systematic literature search 2.1 Questions and PICO criteria Data bases: Medline,
Intron A Hepatitis B. Intron A (interferon alfa-2b) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.01.01 Subject: Intron A Hepatitis B Page: 1 of 7 Last Review Date: November 30, 2018 Intron A Hepatitis
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.01.01 Subject: Intron A Hepatitis B Page: 1 of 7 Last Review Date: November 30, 2018 Intron A Hepatitis
Pegasys Ribavirin
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.01.08 Subsection: Anti-infective Agents Original Policy Date: January 1, 2006 Subject: Pegasys Ribavirin
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.01.08 Subsection: Anti-infective Agents Original Policy Date: January 1, 2006 Subject: Pegasys Ribavirin
HTA: Models, Costs & Benefits.
 HTA: Models, Costs & Benefits John.Cairns@lshtm.ac.uk Explicit evaluation of costs and benefits Advantages Potential for more consistent decision making Potential for more transparent decision making Facilitates
HTA: Models, Costs & Benefits John.Cairns@lshtm.ac.uk Explicit evaluation of costs and benefits Advantages Potential for more consistent decision making Potential for more transparent decision making Facilitates
Hepatitis C Update on New Treatments
 Hepatitis C Update on New Treatments Kevork M. Peltekian, MD, FRCPC 44th Annual Dalhousie Spring Refresher Course - Therapeutics April 5 - April 7, 2018 Halifax Convention Centre Disclosures Conflicts
Hepatitis C Update on New Treatments Kevork M. Peltekian, MD, FRCPC 44th Annual Dalhousie Spring Refresher Course - Therapeutics April 5 - April 7, 2018 Halifax Convention Centre Disclosures Conflicts
Hepatitis C Management and Treatment
 Hepatitis C Management and Treatment Kaya Süer Near East University Faculty of Medicine Infectious Diseases and Clinical Microbiology 1 Discovery of Hepatitis C Key facts Hepatitis C: the virus can cause
Hepatitis C Management and Treatment Kaya Süer Near East University Faculty of Medicine Infectious Diseases and Clinical Microbiology 1 Discovery of Hepatitis C Key facts Hepatitis C: the virus can cause
Hepatitis C Treatment in Oregon
 The Hepatitis C Advisory Group, 12/21/2014 Hepatitis C Treatment in Oregon Introduction The rising health care burden of HCV infection in Oregon is occurring at this time of growing interest in containing
The Hepatitis C Advisory Group, 12/21/2014 Hepatitis C Treatment in Oregon Introduction The rising health care burden of HCV infection in Oregon is occurring at this time of growing interest in containing
A cost effectiveness analysis of treatment options for methotrexate-naive rheumatoid arthritis Choi H K, Seeger J D, Kuntz K M
 A cost effectiveness analysis of treatment options for methotrexate-naive rheumatoid arthritis Choi H K, Seeger J D, Kuntz K M Record Status This is a critical abstract of an economic evaluation that meets
A cost effectiveness analysis of treatment options for methotrexate-naive rheumatoid arthritis Choi H K, Seeger J D, Kuntz K M Record Status This is a critical abstract of an economic evaluation that meets
Cost-effectiveness of tolvaptan (Jinarc ) for the treatment of autosomal dominant polycystic kidney disease (ADPKD)
 Cost-effectiveness of tolvaptan (Jinarc ) for the treatment of autosomal dominant polycystic kidney disease (ADPKD) The NCPE has issued a recommendation regarding the cost-effectiveness of tolvaptan (Jinarc
Cost-effectiveness of tolvaptan (Jinarc ) for the treatment of autosomal dominant polycystic kidney disease (ADPKD) The NCPE has issued a recommendation regarding the cost-effectiveness of tolvaptan (Jinarc
Hepatitis C: Management of Previous Non-responders with First Line Protease Inhibitors
 Hepatitis C: Management of Previous Non-responders with First Line Protease Inhibitors Fred Poordad, MD The Texas Liver Institute Clinical Professor of Medicine University of Texas Health Science Center
Hepatitis C: Management of Previous Non-responders with First Line Protease Inhibitors Fred Poordad, MD The Texas Liver Institute Clinical Professor of Medicine University of Texas Health Science Center
Clopidogrel versus aspirin for secondary prophylaxis of vascular events: a cost-effectiveness analysis Schleinitz M D, Weiss J P, Owens D K
 Clopidogrel versus aspirin for secondary prophylaxis of vascular events: a cost-effectiveness analysis Schleinitz M D, Weiss J P, Owens D K Record Status This is a critical abstract of an economic evaluation
Clopidogrel versus aspirin for secondary prophylaxis of vascular events: a cost-effectiveness analysis Schleinitz M D, Weiss J P, Owens D K Record Status This is a critical abstract of an economic evaluation
Treatment of HCV and HIV coinfection
 Annals of Hepatology 2006; 5(Suppl. 1): S42-S48 Annals of Hepatology Treatment of HCV and HIV coinfection J. Alberto Juárez Navarro 1 Chronic infection with hepatitis C virus (HCV) affects 170 million
Annals of Hepatology 2006; 5(Suppl. 1): S42-S48 Annals of Hepatology Treatment of HCV and HIV coinfection J. Alberto Juárez Navarro 1 Chronic infection with hepatitis C virus (HCV) affects 170 million
Considerations for Antiretroviral Use in Patients with Hepatitis B Virus & Human Immunodeficiency Syndrome Coinfection
 Considerations for Antiretroviral Use in Patients with Hepatitis B Virus & Human Immunodeficiency Syndrome Coinfection Mahnaz Arian, MD Assistant Professor in infectious Disease Mashhad university of Medical
Considerations for Antiretroviral Use in Patients with Hepatitis B Virus & Human Immunodeficiency Syndrome Coinfection Mahnaz Arian, MD Assistant Professor in infectious Disease Mashhad university of Medical
Preventing Mycobacterium avium complex in patients who are using protease inhibitors: a cost-effectiveness analysis Bayoumi A M, Redelmeier D A
 Preventing Mycobacterium avium complex in patients who are using protease inhibitors: a cost-effectiveness analysis Bayoumi A M, Redelmeier D A Record Status This is a critical abstract of an economic
Preventing Mycobacterium avium complex in patients who are using protease inhibitors: a cost-effectiveness analysis Bayoumi A M, Redelmeier D A Record Status This is a critical abstract of an economic
HIV and Hepatitis C Virus Coinfection: Bad Bedfellows
 Perspective HIV and Hepatitis C Virus Coinfection: Bad Bedfellows Hepatitis C virus (HCV) infection is common in HIV-infected individuals and is responsible for increasing morbidity in these patients.
Perspective HIV and Hepatitis C Virus Coinfection: Bad Bedfellows Hepatitis C virus (HCV) infection is common in HIV-infected individuals and is responsible for increasing morbidity in these patients.
The Disease Burden of Hepatitis C in Belgium : development of a realistic disease control strategy
 280 POSITION PAPER : BELGIAN WORKING GROUP FOR HEPATITIS C The Disease Burden of Hepatitis C in Belgium : development of a realistic disease control strategy P. Stärkel 1, D. Vandijck 2,3, W. Laleman 4,
280 POSITION PAPER : BELGIAN WORKING GROUP FOR HEPATITIS C The Disease Burden of Hepatitis C in Belgium : development of a realistic disease control strategy P. Stärkel 1, D. Vandijck 2,3, W. Laleman 4,
Cost-effectiveness of HIV post-exposure prophylaxis in France Herida M, Larsen C, Lot F, Laporte A, Desenclos J C, Hamers F F
 Cost-effectiveness of HIV post-exposure prophylaxis in France Herida M, Larsen C, Lot F, Laporte A, Desenclos J C, Hamers F F Record Status This is a critical abstract of an economic evaluation that meets
Cost-effectiveness of HIV post-exposure prophylaxis in France Herida M, Larsen C, Lot F, Laporte A, Desenclos J C, Hamers F F Record Status This is a critical abstract of an economic evaluation that meets
Clinical Infectious Diseases Advance Access published September 3, 2014
 Clinical Infectious Diseases Advance Access published September 3, 2014 1 Acute hepatitis C virus infection in HIV+ MSM: Should we change our screening practice? Reiberger T. Division of Gastroenterology
Clinical Infectious Diseases Advance Access published September 3, 2014 1 Acute hepatitis C virus infection in HIV+ MSM: Should we change our screening practice? Reiberger T. Division of Gastroenterology
Testing for factor V Leiden in patients with pulmonary or venous thromboembolism: a costeffectiveness
 Testing for factor V Leiden in patients with pulmonary or venous thromboembolism: a costeffectiveness analysis Eckman M H, Singh S K, Erban J K, Kao G Record Status This is a critical abstract of an economic
Testing for factor V Leiden in patients with pulmonary or venous thromboembolism: a costeffectiveness analysis Eckman M H, Singh S K, Erban J K, Kao G Record Status This is a critical abstract of an economic
Common Drug Review Pharmacoeconomic Review Report
 Common Drug Review Pharmacoeconomic Review Report October 2014 Drug Sofosbuvir (Sovaldi) (400 mg/tablet) Indication Sofosbuvir is indicated for the treatment of chronic hepatitis C virus (CHC) infection
Common Drug Review Pharmacoeconomic Review Report October 2014 Drug Sofosbuvir (Sovaldi) (400 mg/tablet) Indication Sofosbuvir is indicated for the treatment of chronic hepatitis C virus (CHC) infection
Viral Hepatitis. Dr Melissa Haines Gastroenterologist Waikato Hospital
 Viral Hepatitis Dr Melissa Haines Gastroenterologist Waikato Hospital Viral Hepatitis HAV HBV HCV HDV HEV Other viral: CMV, EBV, HSV Unknown Hepatitis A Hepatitis A Transmitted via the faecal-oral route
Viral Hepatitis Dr Melissa Haines Gastroenterologist Waikato Hospital Viral Hepatitis HAV HBV HCV HDV HEV Other viral: CMV, EBV, HSV Unknown Hepatitis A Hepatitis A Transmitted via the faecal-oral route
Setting The setting was secondary care. The economic study was carried out in the USA.
 Cost effectiveness of once-daily oral chelation therapy with deferasirox versus infusional deferoxamine in transfusion-dependent thalassaemia patients: US healthcare system perspective Delea T E, Sofrygin
Cost effectiveness of once-daily oral chelation therapy with deferasirox versus infusional deferoxamine in transfusion-dependent thalassaemia patients: US healthcare system perspective Delea T E, Sofrygin
Treatment of Hepatitis C in HIV-Coinfected Patients. Vincent Soriano Department of Infectious Diseases Hospital Carlos III Madrid, Spain
 Treatment of Hepatitis C in HIV-Coinfected Patients Vincent Soriano Department of Infectious Diseases Hospital Carlos III Madrid, Spain Estimated no. of persons infected with HIV and hepatitis viruses
Treatment of Hepatitis C in HIV-Coinfected Patients Vincent Soriano Department of Infectious Diseases Hospital Carlos III Madrid, Spain Estimated no. of persons infected with HIV and hepatitis viruses
The cost-effectiveness of raising the legal smoking age in California Ahmad S
 The cost-effectiveness of raising the legal smoking age in California Ahmad S Record Status This is a critical abstract of an economic evaluation that meets the criteria for inclusion on NHS EED. Each
The cost-effectiveness of raising the legal smoking age in California Ahmad S Record Status This is a critical abstract of an economic evaluation that meets the criteria for inclusion on NHS EED. Each
5/12/2016. Learning Objectives. Management of Hepatitis C Virus Genotype 2 or 3 Infected Treatment-Naive or Experienced Patients
 5/12/216 Management of Hepatitis C Virus Genotype 2 or 3 Infected Treatment-Naive or Experienced Patients Alexander Monto, MD Professor of Clinical Medicine University of California San Francisco San Francisco,
5/12/216 Management of Hepatitis C Virus Genotype 2 or 3 Infected Treatment-Naive or Experienced Patients Alexander Monto, MD Professor of Clinical Medicine University of California San Francisco San Francisco,
SECTION 1: OLYSIO with (PEGASYS) AND RIBAVIRIN SECTION 2: OLYSIO with (PEGINTRON) AND RIBAVIRIN RATIONALE FOR INCLUSION IN PA PROGRAM
 SECTION 1: OLYSIO with (PEGASYS) AND RIBAVIRIN SECTION 2: OLYSIO with (PEGINTRON) AND RIBAVIRIN RATIONALE FOR INCLUSION IN PA PROGRAM SECTION 1: OLYSIO with (PEGASYS) AND RIBAVIRIN Background Hepatitis
SECTION 1: OLYSIO with (PEGASYS) AND RIBAVIRIN SECTION 2: OLYSIO with (PEGINTRON) AND RIBAVIRIN RATIONALE FOR INCLUSION IN PA PROGRAM SECTION 1: OLYSIO with (PEGASYS) AND RIBAVIRIN Background Hepatitis
2016: The State of HIV & Hepatitis C in the District
 2016: The State of HIV & Hepatitis C in the District Travis A. Gayles, MD, PhD Chief Medical Officer, HIV/AIDS, Hepatitis, STD, and TB Administration Division Chief, STD and TB Control February 29, 2016
2016: The State of HIV & Hepatitis C in the District Travis A. Gayles, MD, PhD Chief Medical Officer, HIV/AIDS, Hepatitis, STD, and TB Administration Division Chief, STD and TB Control February 29, 2016
Liver Transplantation: The End of the Road in Chronic Hepatitis C Infection
 University of Massachusetts Medical School escholarship@umms UMass Center for Clinical and Translational Science Research Retreat 2012 UMass Center for Clinical and Translational Science Research Retreat
University of Massachusetts Medical School escholarship@umms UMass Center for Clinical and Translational Science Research Retreat 2012 UMass Center for Clinical and Translational Science Research Retreat
ORIGINAL INVESTIGATION. Cirrhosis and Hepatocellular Carcinoma in HIV-Infected Veterans With and Without the Hepatitis C Virus
 ORIGINAL INVESTIGATION Cirrhosis and Hepatocellular Carcinoma in HIV-Infected Veterans With and Without the Hepatitis C Virus A Cohort Study, 1992-2001 Thomas P. Giordano, MD, MPH; Jennifer R. Kramer,
ORIGINAL INVESTIGATION Cirrhosis and Hepatocellular Carcinoma in HIV-Infected Veterans With and Without the Hepatitis C Virus A Cohort Study, 1992-2001 Thomas P. Giordano, MD, MPH; Jennifer R. Kramer,
Determinants of Response to Pegylated Interferon and Ribavirin for Acute Hepatitis C Infection in Patients with Human Immunodeficiency Virus
 Determinants of Response to Pegylated Interferon and Ribavirin for Acute Hepatitis C Infection in Patients with Human Immunodeficiency Virus Leah Burke, M.D. 1, Daniel Fierer, M.D. 2, David Cassagnol,
Determinants of Response to Pegylated Interferon and Ribavirin for Acute Hepatitis C Infection in Patients with Human Immunodeficiency Virus Leah Burke, M.D. 1, Daniel Fierer, M.D. 2, David Cassagnol,
AASLD, Boston, USA, 10 th November 2014 [oral presentation]
![AASLD, Boston, USA, 10 th November 2014 [oral presentation] AASLD, Boston, USA, 10 th November 2014 [oral presentation]](/thumbs/89/98795522.jpg) Effects of Sustained Virological Response on the of liver transplant, hepatocellular carcinoma, death and re-infection: meta-analysis of 129 studies in 34,563 patients with Hepatitis C infection. Andrew
Effects of Sustained Virological Response on the of liver transplant, hepatocellular carcinoma, death and re-infection: meta-analysis of 129 studies in 34,563 patients with Hepatitis C infection. Andrew
Source of effectiveness data The effectiveness data were derived from a review of completed studies and authors' assumptions.
 Cost-effectiveness of hepatitis A-B vaccine versus hepatitis B vaccine for healthcare and public safety workers in the western United States Jacobs R J, Gibson G A, Meyerhoff A S Record Status This is
Cost-effectiveness of hepatitis A-B vaccine versus hepatitis B vaccine for healthcare and public safety workers in the western United States Jacobs R J, Gibson G A, Meyerhoff A S Record Status This is
Topic: Sovaldi, sofosbuvir Date of Origin: March 14, Committee Approval Date: August 15, 2014 Next Review Date: March 2015
 Medication Policy Manual Policy No: dru332 Topic: Sovaldi, sofosbuvir Date of Origin: March 14, 2014 Committee Approval Date: August 15, 2014 Next Review Date: March 2015 Effective Date: October 1, 2014
Medication Policy Manual Policy No: dru332 Topic: Sovaldi, sofosbuvir Date of Origin: March 14, 2014 Committee Approval Date: August 15, 2014 Next Review Date: March 2015 Effective Date: October 1, 2014
Novel Combination Therapies for the Treatment of Patients with Genotype 1 Hepatitis C Public Meeting
 Novel Combination Therapies for the Treatment of Patients with Genotype 1 Hepatitis C Public Meeting December 18, 2014 1 CTAF Overview Core program of the Institute for Clinical and Economic Review (ICER),
Novel Combination Therapies for the Treatment of Patients with Genotype 1 Hepatitis C Public Meeting December 18, 2014 1 CTAF Overview Core program of the Institute for Clinical and Economic Review (ICER),
Healthy Liver Cirrhosis
 Gioacchino Angarano Clinica delle Malattie Infettive Università degli Studi di Foggia Healthy Liver Cirrhosis Storia naturale dell epatite HCVcorrelata in assenza di terapia Paestum 13-15 Maggio 24 The
Gioacchino Angarano Clinica delle Malattie Infettive Università degli Studi di Foggia Healthy Liver Cirrhosis Storia naturale dell epatite HCVcorrelata in assenza di terapia Paestum 13-15 Maggio 24 The
Delta hepatitis: How to manage and optimize therapy? Dominique ROULOT Unité d Hépatologie, Hôpital Avicenne
 Delta hepatitis: How to manage and optimize therapy? Dominique ROULOT Unité d Hépatologie, Hôpital Avicenne Delta hepatitis treatment in 2018 Currently, Peg-IFNα 180 µg/wk is the only effective treatment
Delta hepatitis: How to manage and optimize therapy? Dominique ROULOT Unité d Hépatologie, Hôpital Avicenne Delta hepatitis treatment in 2018 Currently, Peg-IFNα 180 µg/wk is the only effective treatment
Ari Bunim, M.D. Director of Hepatology New York Hospital Queens Assistant Professor of Clinical Medicine Weill Cornell Medical College
 Ari Bunim, M.D. Director of Hepatology New York Hospital Queens Assistant Professor of Clinical Medicine Weill Cornell Medical College New York State Law Goes into Effect January 1, 2014 Hepatitis C Virus
Ari Bunim, M.D. Director of Hepatology New York Hospital Queens Assistant Professor of Clinical Medicine Weill Cornell Medical College New York State Law Goes into Effect January 1, 2014 Hepatitis C Virus
Scottish Medicines Consortium
 Scottish Medicines Consortium raltegravir, 400mg film-coated tablet (Isentress) No. (461/08) Merck, Sharp and Dohme Limited 04 April 2008 The Scottish Medicines Consortium has completed its assessment
Scottish Medicines Consortium raltegravir, 400mg film-coated tablet (Isentress) No. (461/08) Merck, Sharp and Dohme Limited 04 April 2008 The Scottish Medicines Consortium has completed its assessment
Pertussis in adolescents and adults: should we vaccinate Lee G M, LeBaron C, Murphy T V, Lett S, Schauer S, Lieu T A
 Pertussis in adolescents and adults: should we vaccinate Lee G M, LeBaron C, Murphy T V, Lett S, Schauer S, Lieu T A Record Status This is a critical abstract of an economic evaluation that meets the criteria
Pertussis in adolescents and adults: should we vaccinate Lee G M, LeBaron C, Murphy T V, Lett S, Schauer S, Lieu T A Record Status This is a critical abstract of an economic evaluation that meets the criteria
Update on Real-World Experience With HARVONI
 Update on Real-World Experience With A RESOURCE FOR PAYERS This information is intended for payers only. The HCV-TARGET and TRIO studies were supported by Gilead Sciences, Inc. Real-world experience data
Update on Real-World Experience With A RESOURCE FOR PAYERS This information is intended for payers only. The HCV-TARGET and TRIO studies were supported by Gilead Sciences, Inc. Real-world experience data
Resource impact report: Sofosbuvir velpatasvir for treating chronic hepatitis C TA430
 Putting NICE guidance into practice Resource impact report: Sofosbuvir velpatasvir for treating chronic hepatitis C TA430 Published: January 2017 Summary Sofosbuvir velpatasvir is recommended as an option
Putting NICE guidance into practice Resource impact report: Sofosbuvir velpatasvir for treating chronic hepatitis C TA430 Published: January 2017 Summary Sofosbuvir velpatasvir is recommended as an option
Treatment guidance for Chronic Hepatitis C Infection
 Treatment guidance for Chronic Hepatitis C Infection A meeting of the network was held on 11 April 2008 at which the current evidence base for hepatitis C was reviewed. These guidelines have been produced
Treatment guidance for Chronic Hepatitis C Infection A meeting of the network was held on 11 April 2008 at which the current evidence base for hepatitis C was reviewed. These guidelines have been produced
When to start: guidelines comparison
 The editorial staff When to start: guidelines comparison The optimal time to begin antiretroviral therapy remains a critical question for the HIV field, and consensus about the appropriate CD4+ cell count
The editorial staff When to start: guidelines comparison The optimal time to begin antiretroviral therapy remains a critical question for the HIV field, and consensus about the appropriate CD4+ cell count
Technology appraisal guidance Published: 25 November 2015 nice.org.uk/guidance/ta365
 Ombitasvir paritaprevir ritonavir with or without dasabuvir for treating chronic hepatitis C Technology appraisal guidance Published: 25 November 2015 nice.org.uk/guidance/ta365 NICE 2017. All rights reserved.
Ombitasvir paritaprevir ritonavir with or without dasabuvir for treating chronic hepatitis C Technology appraisal guidance Published: 25 November 2015 nice.org.uk/guidance/ta365 NICE 2017. All rights reserved.
PEGINTRON (peginterferon alfa-2b) SECTION 1: Pegintron - Hepatitis C Monotherapy
 SECTION 1: Hepatitis C Pegintron SECTION 2: Hepatitis C Pegintron & Ribavirin SECTION 3: Hepatitis C Pegintron, Ribavirin, & Sovaldi SECTION 4: Hepatitis C Pegintron, Ribavirin, & Olysio SECTION 1: Pegintron
SECTION 1: Hepatitis C Pegintron SECTION 2: Hepatitis C Pegintron & Ribavirin SECTION 3: Hepatitis C Pegintron, Ribavirin, & Sovaldi SECTION 4: Hepatitis C Pegintron, Ribavirin, & Olysio SECTION 1: Pegintron
Intron A Hepatitis C. Intron A (interferon alfa-2b) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.01.05 Subject: Intron A Hepatitis C Page: 1 of 5 Last Review Date: November 30, 2018 Intron A Hepatitis
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.01.05 Subject: Intron A Hepatitis C Page: 1 of 5 Last Review Date: November 30, 2018 Intron A Hepatitis
Intron A (interferon alfa-2b) with ribavirin, (Moderiba, Rebetol, Ribasphere, RibaTab, ribavirin tablets/capsules - all strengths)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.01.06 Subject: Intron A Ribavirin Page: 1 of 6 Last Review Date: March 18, 2016 Intron A Ribavirin Description
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.01.06 Subject: Intron A Ribavirin Page: 1 of 6 Last Review Date: March 18, 2016 Intron A Ribavirin Description
Dr. Siddharth Srivastava
 Dr. Siddharth Srivastava MD, DM (Gastroenterology) Associate Professor GIPMER, New Delhi Rashtriya Gaurav Award 2013 for work on hepatitis B and C Set up Liver clinic at GIPMER and in charge EUS laboratory.
Dr. Siddharth Srivastava MD, DM (Gastroenterology) Associate Professor GIPMER, New Delhi Rashtriya Gaurav Award 2013 for work on hepatitis B and C Set up Liver clinic at GIPMER and in charge EUS laboratory.
entecavir, 0.5mg and 1mg film-coated tablets and 0.05 mg/ml oral solution, Baraclude SMC No. (747/11) Bristol-Myers Squibb Pharmaceuticals Ltd
 entecavir, 0.5mg and 1mg film-coated tablets and 0.05 mg/ml oral solution, Baraclude SMC No. (747/11) Bristol-Myers Squibb Pharmaceuticals Ltd 09 December 2011 The Scottish Medicines Consortium (SMC) has
entecavir, 0.5mg and 1mg film-coated tablets and 0.05 mg/ml oral solution, Baraclude SMC No. (747/11) Bristol-Myers Squibb Pharmaceuticals Ltd 09 December 2011 The Scottish Medicines Consortium (SMC) has
WHAT CAUSES CERVICAL AND ANAL CANCER?
 Harvard Center for Risk Analysis FEBRUARY 2000 Volume 8 Issue 2 Risk in Perspective Preventing Cervical and Anal Cancer in HIV-Positive Women and Men: Using Disease- Specific Models to Develop Clinical
Harvard Center for Risk Analysis FEBRUARY 2000 Volume 8 Issue 2 Risk in Perspective Preventing Cervical and Anal Cancer in HIV-Positive Women and Men: Using Disease- Specific Models to Develop Clinical
Cost-effectiveness of cesarean section delivery to prevent mother-to-child transmission of HIV-1 Halpern M T, Read J S, Ganoczy D A, Harris D R
 Cost-effectiveness of cesarean section delivery to prevent mother-to-child transmission of HIV-1 Halpern M T, Read J S, Ganoczy D A, Harris D R Record Status This is a critical abstract of an economic
Cost-effectiveness of cesarean section delivery to prevent mother-to-child transmission of HIV-1 Halpern M T, Read J S, Ganoczy D A, Harris D R Record Status This is a critical abstract of an economic
Cost-effectiveness of antiviral drug therapy to reduce mother-to-child HIV transmission in sub-saharan Africa Marseille E, Kahn J G, Saba J
 Cost-effectiveness of antiviral drug therapy to reduce mother-to-child HIV transmission in sub-saharan Africa Marseille E, Kahn J G, Saba J Record Status This is a critical abstract of an economic evaluation
Cost-effectiveness of antiviral drug therapy to reduce mother-to-child HIV transmission in sub-saharan Africa Marseille E, Kahn J G, Saba J Record Status This is a critical abstract of an economic evaluation
Scottish Medicines Consortium
 Scottish Medicines Consortium tenofovir disoproxil (as fumarate), 245 mg film-coated tablet (Viread ) No. (479/08) Gilead Sciences 06 June 2008 The Scottish Medicines Consortium has completed its assessment
Scottish Medicines Consortium tenofovir disoproxil (as fumarate), 245 mg film-coated tablet (Viread ) No. (479/08) Gilead Sciences 06 June 2008 The Scottish Medicines Consortium has completed its assessment
Potential health and economic impact of adding a human papillomavirus vaccine to screening programs Kulasingam S L, Myers E R
 Potential health and economic impact of adding a human papillomavirus vaccine to screening programs Kulasingam S L, Myers E R Record Status This is a critical abstract of an economic evaluation that meets
Potential health and economic impact of adding a human papillomavirus vaccine to screening programs Kulasingam S L, Myers E R Record Status This is a critical abstract of an economic evaluation that meets
Crucial factors that influence cost-effectiveness of universal hepatitis B immunization in India Prakash C
 Crucial factors that influence cost-effectiveness of universal hepatitis B immunization in India Prakash C Record Status This is a critical abstract of an economic evaluation that meets the criteria for
Crucial factors that influence cost-effectiveness of universal hepatitis B immunization in India Prakash C Record Status This is a critical abstract of an economic evaluation that meets the criteria for
07-Nov HCV infection in India. Cost-effectiveness of hepatitis C treatment: The Indian Scenario. Incremental cost-effectiveness plane
 Cost-effectiveness of hepatitis C : The Indian Scenario HCV infection in India Disease burden: 6 to 7 million viremic cases (0.5-0.6% of population), mainly genotype 3 Generic DAAs available at low cost
Cost-effectiveness of hepatitis C : The Indian Scenario HCV infection in India Disease burden: 6 to 7 million viremic cases (0.5-0.6% of population), mainly genotype 3 Generic DAAs available at low cost
The cost-effectiveness of NAT for HIV, HCV, and HBV in whole-blood donations Jackson B R, Busch M P, Stramer S L, AuBuchon J P
 The cost-effectiveness of NAT for HIV, HCV, and HBV in whole-blood donations Jackson B R, Busch M P, Stramer S L, AuBuchon J P Record Status This is a critical abstract of an economic evaluation that meets
The cost-effectiveness of NAT for HIV, HCV, and HBV in whole-blood donations Jackson B R, Busch M P, Stramer S L, AuBuchon J P Record Status This is a critical abstract of an economic evaluation that meets
ANTIVIRAL THERAPY FOR HCV. Alfredo Alberti
 CLINICAL IMPACT OF SVR AFTER ANTIVIRAL THERAPY FOR HCV Alfredo Alberti Department of Histology,Microbiology and Medical Biotechnologies Molecular Hepatology Unit Venetian Institute of Molecular Medicine
CLINICAL IMPACT OF SVR AFTER ANTIVIRAL THERAPY FOR HCV Alfredo Alberti Department of Histology,Microbiology and Medical Biotechnologies Molecular Hepatology Unit Venetian Institute of Molecular Medicine
Cost-effectiveness of radiofrequency catheter ablation for atrial fibrillation Chan P S, Vijan S, Morady F, Oral H
 Cost-effectiveness of radiofrequency catheter ablation for atrial fibrillation Chan P S, Vijan S, Morady F, Oral H Record Status This is a critical abstract of an economic evaluation that meets the criteria
Cost-effectiveness of radiofrequency catheter ablation for atrial fibrillation Chan P S, Vijan S, Morady F, Oral H Record Status This is a critical abstract of an economic evaluation that meets the criteria
47 th Annual Meeting AISF
 47 th Annual Meeting AISF Rome, 21 February 2014 Present and future treatment strategies for patients with HCV infection: chronic hepatitis and special populations (HCV/HIV coinfection, advanced cirrhosis,
47 th Annual Meeting AISF Rome, 21 February 2014 Present and future treatment strategies for patients with HCV infection: chronic hepatitis and special populations (HCV/HIV coinfection, advanced cirrhosis,
Clinical Infectious Diseases Advance Access published June 16, Age-Old Questions: When to Start Antiretroviral Therapy and in Whom?
 Clinical Infectious Diseases Advance Access published June 16, 2015 1 Age-Old Questions: When to Start Antiretroviral Therapy and in Whom? Rochelle P. Walensky, Martin S. Hirsch From the Division of Infectious
Clinical Infectious Diseases Advance Access published June 16, 2015 1 Age-Old Questions: When to Start Antiretroviral Therapy and in Whom? Rochelle P. Walensky, Martin S. Hirsch From the Division of Infectious
Cost-effectiveness of Hepatitis A and Hepatitis B Vaccination for Jail Inmates
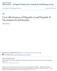 Yale University EliScholar A Digital Platform for Scholarly Publishing at Yale Yale Medicine Thesis Digital Library School of Medicine 2007 Cost-effectiveness of Hepatitis A and Hepatitis B Vaccination
Yale University EliScholar A Digital Platform for Scholarly Publishing at Yale Yale Medicine Thesis Digital Library School of Medicine 2007 Cost-effectiveness of Hepatitis A and Hepatitis B Vaccination
Authors Natasha K. Martin 1,2 Alec Miners 1 Peter Vickerman 1,2. Tropical Medicine, Tavistock Place, London, UK. Whatley Road, Bristol, UK
 Assessing the cost-effectiveness of interventions aimed at promoting and offering hepatitis C testing to injecting drug users: An economic modelling report Authors Natasha K. Martin 1,2 Alec Miners 1 Peter
Assessing the cost-effectiveness of interventions aimed at promoting and offering hepatitis C testing to injecting drug users: An economic modelling report Authors Natasha K. Martin 1,2 Alec Miners 1 Peter
Antiviral Therapy 2015; 20:65 72 (doi: /IMP2827)
 Antiviral Therapy 2015; 20:65 72 (doi: 10.3851/IMP2827) Original article Long-term survival and liver-related events after pegylated interferon/ribavirin therapy in HIV infected patients with chronic hepatitis
Antiviral Therapy 2015; 20:65 72 (doi: 10.3851/IMP2827) Original article Long-term survival and liver-related events after pegylated interferon/ribavirin therapy in HIV infected patients with chronic hepatitis
