Intranasal Vaccination against Cutaneous Leishmaniasis with a Particulated Leishmanial Antigen or DNA Encoding LACK
|
|
|
- Christiana Byrd
- 5 years ago
- Views:
Transcription
1 INFECTION AND IMMUNITY, Aug. 2004, p Vol. 72, No /04/$ DOI: /IAI Copyright 2004, American Society for Microbiology. All Rights Reserved. Intranasal Vaccination against Cutaneous Leishmaniasis with a Particulated Leishmanial Antigen or DNA Encoding LACK Eduardo Fonseca Pinto, Roberta Olmo Pinheiro, 1 Alice Rayol, 1 Vicente Larraga, 2 and Bartira Rossi-Bergmann 1 * Instituto de Biofísica Carlos Chagas Filho, Universidade Federal do Rio de Janeiro, Rio de Janeiro, Brazil, 1 and Centro de Investigaciones Biologicas, Madrid, Spain 2 Received 10 March 2004/Returned for modification 9 April 2004/Accepted 2 May 2004 We have previously demonstrated that oral delivery of a disease-promoting particulated antigen of Leishmania amazonensis (LaAg) partially protects mice against cutaneous leishmaniasis. In the present work, we sought to optimize a mucosal vaccine by using the intranasal route for delivery of different antigen preparations, including (i) LaAg, (ii) soluble recombinant p36/lack leishmanial antigen (LACK), and (iii) plasmid DNA encoding LACK (LACK DNA). BALB/c mice that received two intranasal doses of 10 g of LaAg and were challenged 1 week postvaccination with L. amazonensis developed delayed but effective control of lesion growth. A diminished parasite burden was accompanied by enhancement of both gamma interferon (IFN- ) and interleukin-10 levels in the lesion-draining lymph nodes. The vaccine efficacy improved with time. At 4 months postvaccination, when a strong parasite-specific TH1-type response was present in vivo, the infection was controlled for at least 5 months after challenge. In contrast to the particulated LaAg, soluble LACK (10 g/dose) had no effect. Interestingly, LACK DNA (30 g/dose), but not empty DNA, promoted rapid and durable protective immunity. Parasite growth was effectively controlled, and at 5 months after challenge LACK-reactive cells in both the mucosal and lesion-draining lymph nodes produced high levels of IFN-. These results demonstrate for the first time the feasibility of using the intranasal route for long-lived memory vaccination against cutaneous leishmaniasis with adjuvant-free crude antigens or DNA. * Corresponding author. Mailing address: Instituto de Biofísica Carlos Chagas Filho, Universidade Federal do Rio de Janeiro, Rio de Janeiro-RJ, Brazil. Phone: 55 (21) Fax: 55 (21) bartira@biof.ufrj.br. Leishmaniasis is a disease caused by intracellular protozoan parasites belonging to the genus Leishmania, and there is a wide spectrum of clinical manifestations ranging from chronic cutaneous ulcers to fatal visceral infection. Leishmaniasis is broadly distributed in most tropical and Mediterranean countries, and the estimated incidence is 2 million cases/year (38). Despite the recent advances in new antileishmanial compounds (6), the first-line therapy for all forms of the disease is still based on painful multiple injections of pentavalent antimonials which invariably produce serious toxicity. The problem is further aggravated by the surge of antimonial resistance in some areas where the disease is endemic (22). The development of a vaccine against leishmaniasis is a long-term goal in both human and veterinary medicine. Various subunit and DNA antigens have been identified as potential vaccine candidates (14). However, the development of subunit vaccines for human vaccination against cutaneous leishmaniasis is hampered by the requirement for potentially toxic TH1-inducing adjuvants, which include BCG and interleukin-12 (IL-12). A great number of studies of experimental vaccines against cutaneous leishmaniasis have been based on the murine model of infection with Leishmania major, and these studies have demonstrated the relationship between genetically determined resistance and susceptibility to infection with the expansion of TH1 and TH2 cells, respectively (35). The extreme susceptibility of BALB/c mice to L. major infection has been correlated with early production of IL-4 by LACK (Leishmania homolog of mammalian receptors for activated C kinase)-reactive V 4-V 8 CD4 T cells that drive the differentiation of other responding T cells to the TH2 phenotype (20). BALB/c mice made tolerant to LACK by transgenic expression of the antigen in the thymus exhibit both a diminished TH2 response and a healing phenotype, demonstrating the association of LACK reactivity with disease (16). LACK is a well-conserved antigen that is present in many Leishmania species (31), and it may well account at least in part for the increased susceptibility of mice and monkeys vaccinated by intradermal, intramuscular, and subcutaneous routes with whole promastigote antigens of Leishmania amazonensis or L. major in the absence of adjuvants (19, 21, 32). Mucosal vaccination with immunopathology-related antigens has been used to induce systemic tolerance and protection against TH2-related disorders, such as allergies and some autoimmune diseases (7). In this context, the effect of oral vaccination of susceptible BALB/c mice and the more resistant C57BL/6 mouse strain with a total of 200 g ofl. amazonensis antigen (LaAg) on subsequent parasite challenge was recently evaluated (32). Vaccination rendered both mouse strains more resistant to L. amazonensis and L. major infections. Protectiveness required functional / T-cell receptor-positive T cells and was accompanied by selective systemic tolerance of TH2- type responses. The naturally particulated form of LaAg may have been critical for protectiveness, as up to 8 mg of soluble LACK administered by mucosal routes (oral and nasal) did not affect the susceptibility of BALB/c mice to L. major (26). In that study, only when LACK was conjugated with the adjuvant cholera toxin beta subunit (CTB) was partial protection 4521
2 4522 PINTO ET AL. INFECT. IMMUN. achieved. However, no mucosal adjuvant, including the cholera toxin beta subunit, has been approved for use in humans yet (10). Therefore, identification of a mucosal vaccine that is intrinsically immunogenic and effective is highly desirable. CpG DNA is a potent enhancer of systemic and mucosal immune responses (25), and intranasal DNA vaccines are emerging as a successful noninvasive alternative to DNA injected intramuscularly (2). In leishmaniasis, parenteral injections with LACK-encoding plasmid DNA (LACK DNA) proved to effectively protect BALB/c mice against L. major infection, in contrast to the disease-enhancing effect of adjuvant-free soluble LACK (12, 37). On the other hand, leishmaniasis caused by Leishmania donovani or Leishmania mexicana seems to be more refractory to parenteral LACK DNA (9, 27). The noninvasive nasal route of vaccination is more advantageous than the oral route, because less acidic and enzymatic antigen digestion allows a more controlled and reduced vaccine dosage along with consequent reduced costs. In the present work we attempted to optimize mucosal vaccination against cutaneous leishmaniasis by using the intranasal route for evaluation of the effectiveness of various forms of leishmanial antigens. MATERIALS AND METHODS Mice. BALB/c mice were originally purchased from Jackson Laboratory (Bar Harbor, Maine). They were bred and maintained at our facilities by using sterilized bedding, filtered water, and pelleted food. Female animals were used when they were 4 to 6 weeks old. Experimental protocols were approved by the Animal Use Committee of the Institute of Biophysics/Federal University of Rio de Janeiro (Brazil). Parasites. L. amazonensis (strain MHOM/BR/75/Josefa) was used at the early stationary growth phase for both vaccine preparation and infection. For the vaccine, wild-type parasites were used. For infection, parasites that were rendered fluorescent by transfection with green fluorescent protein (GFP) (36) were used. Both wild-type and GFP-expressing parasites were routinely isolated from mouse lesions and maintained at 26 C as promastigotes in Dulbecco modified minimum essential medium containing 10% heat-inactivated fetal calf serum and antibiotics (50 U of penicillin per ml and 50 g of streptomycin per ml). Transfected parasites were periodically cultured with 100 g of Geneticin per ml for GFP selection. Vaccines. (i) LaAg. LaAg was prepared as previously described (32). Briefly, wild-type L. amazonensis promastigotes were washed three times by centrifugation, and the pellet was resuspended at a concentration of parasites/ml in phosphate-buffered saline (PBS) and subjected to three cycles of freezing and thawing. The resulting particulated cell lysate was termed LaAg. One milliliter of LaAg contained 970 g of protein, as measured by the Lowry assay. Sample aliquots were kept at 20 C until they were required. (ii) LACK DNA. The gene encoding the Leishmania infantum LACK protein was obtained from a genomic library as described previously (11) and was inserted downstream of the cytomegalovirus promoter in the EcoRI/XbaI site of the pci-neo expression vector (Promega), giving plasmid pci-neo-lack (LACK DNA) (34). Empty plasmid pci-neo was used as a DNA control. (iii) LACK. L. infantum p36 recombinant protein was purified from an Escherichia coli BL21 strain transformed with a prset expression plasmid as previously described (11). Nasal immunization. Mice held upward received by instillation 10 g of LaAg, 10 g of LACK, 30 g of LACK DNA, or 30 g of control DNA in 20 l ofpbs (10 l in each nostril) with a fine tip attached to a micropipette. The animals were boosted 7 days later by using the same vaccine dosage. The controls received PBS. Infection. Mice were infected in a hind footpad at 1 week or at 17 weeks (4 months) after the second vaccination dose with 10 5 GFP-labeled promastigotes of L. amazonensis. Lesion sizes were measured with a dial caliper (Mitutoyo, Sao Paolo, Brazil) every 4 to 5 days, and the results were expressed as the difference between the thickness of infected footpads and the thickness of noninfected footpads. For determination of parasite loads in the lesions, infected feet were cut off and individually homogenized in 2 ml of PBS by using a tissue grinder. FIG. 1. Protective effect of nasal immunization with LaAg against subsequent infection with L. amazonensis. BALB/c mice were vaccinated intranasally with 10 g of LaAg and received a booster vaccination 7 days later (F and solid bar). The controls received PBS alone (E and open bar). One week after the booster vaccination each of the animals was infected with 10 5 fluorescent L. amazonensis cells in the footpad. (A) Lesion sizes on different days. (B) On day 110 of infection, the parasite loads in individual feet were expressed in fluorescence units. The values are means and standard deviations (n 5). One asterisk, P 0.05; two asterisks, P After removal of tissue debris by gravity sedimentation for 10 min, 200- l portions of twofold dilutions of the cell suspensions were transferred in triplicate to black microplates, and fluorescence was read with a plate reader fluorometer (Fluoroskan, Ashford, United Kingdom) by using excitation at 435 nm and emission at 538 nm (32, 36). Cytokines. The nose-draining cervical lymph nodes and the lesion-draining popliteal lymph nodes were excised, and single-cell suspensions were prepared in Dulbecco modified minimum essential medium containing 10% heat-inactivated fetal calf serum and antibiotics. Cells were plated in triplicate in 24-well culture plates at a concentration of cells/ml and were stimulated with 2.5 g of Concanavalin A (ConA) (Sigma Aldrich, St. Louis, Mo.) per ml or with 10 g of LACK per ml for 48 h at 37 C with 4% CO 2 in air. ConA was used for stimulation of cells from LaAg-vaccinated animals since crude antigens of L. amazonensis are anergenic to T cells in vitro (32). The levels of gamma interferon (IFN- ) and IL-10 were measured in twofold dilutions of the supernatants by an enzyme-linked immunosorbent assay (ELISA). The levels of cytokines were determined with standard curves by using recombinant murine cytokines and paired antibodies according to the instructions of the manufacturer (R&D Systems, Minneapolis, Minn.). Hypersensitivity reaction. Four months after boosting, each animal was injected in a hind footpad with 20 g of LaAg in 20 l of PBS. Footpad swelling was measured with a dial caliper (Mitutoyo) at various times, and the results were expressed as the difference between the thickness of the footpads inoculated with the antigen and the thickness of the footpads inoculated with 20 l of PBS. Statistical analysis. The statistical significance of differences between vaccinated and control groups of mice was determined by Student s t test. RESULTS Intranasal vaccination with LaAg confers protection against L. amazonensis infection. To evaluate whether the nasal mucosa could be used for controlled administration of vaccine doses lower than those previously shown to be required for oral immunization ( 100 g) (32), BALB/c mice were doubly immunized with 10 g of LaAg prior to subcutaneous infection with GFP-expressing L. amazonensis. Figure 1A shows that following a lag period of 80 days, the vaccinated animals were able to significantly control lesion growth (P 0.02), unlike the nonvaccinated animals, in which the lesions continued to grow steadily. The protection conferred by nasally administered LaAg was confirmed by a significantly lower (P 0.05) parasite burden, as determined by decreased fluorescence in the
3 VOL. 72, 2004 INTRANASAL VACCINATION AGAINST LEISHMANIASIS 4523 FIG. 2. Cytokine production by mice infected 1 week after LaAg vaccination. Mice (n 5) were vaccinated intranasally with LaAg (solid bars) or with PBS alone (open bars) prior to infection, as described in the legend to Fig 1. On day 110 of infection, the levels of IFN- and IL-10 produced by ConA (2.5 g/ml)-stimulated peripheral lymph node cells (PL) and cervical lymph node cells (CL) were measured in the culture supernatants by ELISA. The values are means and standard deviations for triplicate samples. An asterisk indicates that the P value was 0.05 for a comparison with the PBS controls. nd, not detectable. infected feet (Fig. 1B). This finding shows that the protection against homologous infection after mucosal immunization with LaAg is not restricted to the gastrointestinal system but extends to the nasal mucosa when a lower dosage is used. Intranasally administered LaAg induces mixed cytokine responses during infection. To more specifically determine the modulation of the immune responses during control of leishmanial infection in vaccinated animals, cytokine responses were measured in the peripheral popliteal (lesion-draining) lymph nodes and in the cervical (nose-draining) lymph nodes. As shown in Fig. 2, nonvaccinated animals produced little or no IFN- in the late stages of infection, but they produced relatively high levels of IL-10 in both sites, which is compatible with a progressive disease. Nasal vaccination resulted in a 7.4-fold increase in the production of IFN- in the peripheral lymph nodes, whereas production of IL-10 was increased 1.4- fold compared to production in nonvaccinated mice, which was indicative of a preferential increase in the IFN- response during protection. The IFN- and IL-10 levels measured just prior to infection (day 0) in the peripheral lymph nodes were enhanced 2.5- and 1.5-fold (P 0.05 for both), whereas on day 30 they were enhanced 2-fold (P 0.01) and 5-fold (P 0.05), respectively (data not shown). The IL-4 level measured on day 30 showed eightfold enhancement compared to the level in nonvaccinated mice (P 0.01) (data not shown). Together, these findings indicate that short-term vaccination did not prevent the development of TH2 responses in the early weeks of infection, which may have been critical to the lack of protection in the earlier stages of infection, but the increased IFN- response that followed may have contributed to the effective control of lesion growth at the later stages. The absolute levels of IL-10 and IL-4 were generally higher than the IFN- levels, but it is worth emphasizing that the measurements were obtained by using a quantitative method, not a qualitative method, which detected relative variations for a given cytokine but not variations for different cytokines. Intranasally administered LaAg induces a long-lasting immunological memory. Surrogate markers of protective immunity in murine cutaneous leishmaniasis include TH1 responses, FIG. 3. Hypersensitivity response and cytokine production 4 months after nasal vaccination. Mice (n 9) received LaAg (F and solid bars) or PBS alone (E and open bars), as described in the legend to Fig 1. (A) Four months after the booster vaccination, some of the animals (n 5) were challenged in the footpads with 20 g of LaAg, and the local swelling response was scored at different times. (B) The levels of IFN- and IL-10 produced by popliteal lymph node cells (PL) and cervical lymph node cells (CL) of unchallenged animals (n 4) were measured in the culture supernatants of ConA-stimulated cells by ELISA. The values are means and standard deviations. One asterisk, P 0.05 for a comparison with the corresponding PBS controls; two asterisks, P 0.02 for a comparison with the corresponding PBS controls. such as IFN- production and delayed-type hypersensitivity (DTH) responses in vivo (8, 29). These recall responses were evaluated 4 months after vaccination with LaAg. Figure 3 shows that vaccinated animals, but not nonvaccinated animals, displayed a strong DTH response after antigen challenge. The swelling kinetics showed that there was a peak response at 24 h, which was maintained for at least 48 h. A significant (P 0.05) increase in the production of IFN- in both popliteal and cervical lymph nodes was seen in vaccinated animals (Fig. 3, lower left panel), which was compatible with the in vivo DTH response, even though the IL-10 level was also augmented in the popliteal lymph nodes (Fig. 3, lower right panel). Together, these findings indicate that nasal vaccination with adjuvantfree LaAg induced a long-lasting mixed immune response in peripheral lymphoid tissues, which resulted in an inflammatory TH1-type immunity. Intranasally administered LaAg confers long-lasting protective immunity. To evaluate whether the long-lasting immunogenicity of nasally administered LaAg remained protective, animals were challenged 4 months after vaccination, and the course of infection was then monitored for a further 5 months (160 days). As shown in Fig. 4A, vaccinated animals managed to control lesion growth throughout the infection. Protection was confirmed by a significantly (P 0.02) lower parasite burden compared with that of nonvaccinated animals (Fig. 4B). It is worth noting that the efficacy of vaccination increased with time. The delayed control seen in animals challenged only 1 week after vaccination (Fig. 1A) was suppressed 4 months
4 4524 PINTO ET AL. INFECT. IMMUN. FIG. 4. Long-lasting protective effect of nasal LaAg vaccination. Mice were immunized intranasally twice with LaAg (F and solid bar) or with PBS alone (E and open bar) as described in the legend to Fig. 1. Animals were infected 4 months later with 10 5 fluorescent L. amazonensis cells in a footpad. (A) Lesion growth on different days. (B) On day 160 of infection, the parasite loads in the infected feet were expressed in fluorescence units. The values are means and standard deviations (n 5). One asterisk, P 0.01; two asterisks, P later (Fig. 4A), when the animals were able to control the infection since the early days. The cytokine levels measured after 5 months of infection were increased (Fig. 5) compared with the levels measured just prior to infection (Fig. 3), probably due to active stimulation by live parasitism. Interestingly, the levels of IFN- but not the levels of IL-10 were further increased in the lesion-draining popliteal lymph nodes of vaccinated animals, suggesting that the IFN- skewed response was related to infection control. Although the effect was not as pronounced, the levels of IFN- were still elevated in the cervical lymph nodes (P 0.05), indicating that after such a long time after vaccination the mucosa-associated sites were still sensitized. Together, these findings demonstrate that the long-term memory produced by nasal vaccination with LaAg prompted effective control of infection. Intranasally administered LACK DNA is also effective in conferring protection against infection. To verify the efficacy of a soluble defined leishmanial antigen, we used recombinant LACK (rlack) to vaccinate BALB/c mice by the nasal route FIG. 5. Cytokine production by mice infected 4 months after vaccination. Mice (n 5) were immunized intranasally with LaAg (solid bars) or received PBS alone (open bars) and were infected as described in the legend to Fig 4. On day 160 of infection, the levels of IFN- and IL-10 produced by popliteal lymph node cells (PL) and cervical lymph node cells (CL) were measured in the culture supernatants of ConA-stimulated cells by ELISA. The values are means and standard deviations for triplicate samples. An asterisk indicates that the P value is using the same protocol that was used for LaAg, in which animals were challenged 7 days after the booster dose. We found that administration of 10 g of rlack/dose had no effect (Fig. 6A) compared with administration of LaAg. The possibility of a suboptimal dosage was discarded since we used the same dose that we used for LaAg, in which LACK was present in a fraction. We then evaluated whether plasmid DNA expressing LACK could be effective. LACK DNA (30 g/dose) significantly delayed lesion growth (Fig. 6A) and prevented parasite growth (Fig. 6B) for at least 5 months. This effect was not due to an adjuvant effect of CpG sequences in the bacterial DNA, as empty control DNA did not affect lesion growth. Interestingly, the delayed onset of infection was accompanied by increased LACK recall production of IFN- not only in the peripheral lymph nodes but also in the cervical lymph nodes (Fig. 7). The same cell populations restimulated in vitro with ConA showed a similar pattern of responses (data not shown). These results indicate that the DNA induced longterm sensitization in the mucosa-draining lymph nodes. DISCUSSION Previous studies showed that injecting killed promastigotes of L. major or L. amazonensis into the skin rendered mice and rhesus monkeys more susceptible to cutaneous leishmaniasis (19, 21). Interestingly, an opposing protective effect can be produced if killed promastigotes of L. amazonensis (LaAg) are administered orally to mice. There is an association between increased resistance in animals orally vaccinated with LaAg and increased production of IFN- in the lesion-draining lymph nodes, and there is concomitant suppression of parasitespecific TH2-type responses in vivo (32). Mucosal membranes of the various tissues are linked together in a common mucosal immune system network. This is due to the expression of specific lymphocyte homing receptors, such as 4 7, that recognize the mucosal addressin cell adhesion molecule MadCAM-1 (13). Thus, in the present work we attempted to optimize mucosal immunization against cutaneous leishmaniasis by using the nasal route of vaccination for smaller vaccine doses and testing different leishmanial antigen types. Here we show for the first time that intranasal immunization with an adjuvant-free antigen could protect mice against cutaneous leishmaniasis. Passive inhalation of aerosol LaAg was also effective in delaying the onset of infection and reducing the parasite burden, but an amount of LaAg that was 30 times larger than the amount used for nasal instillation was necessary (unpublished observations). When we used a protocol similar to the protocol previously used for oral vaccination, in which animals were challenged 1 week postvaccination (32), intranasal vaccination with a 10-fold-lower LaAg dose also promoted increased resistance in BALB/c mice against L. amazonensis infection (Fig. 1). Interestingly, unlike orally vaccinated BALB/c mice, which responded more rapidly although at a lower intensity (32), nasally vaccinated mice were able to effectively control an infection only late in the infection, as shown by the significantly reduced parasite burden measured on day 110 of infection (Fig. 1B). It is possible that the late control of infection after short-term vaccination was due to the time lag required for an effective IFN- response. A very slow IFN- response has also been observed in humans, and some
5 VOL. 72, 2004 INTRANASAL VACCINATION AGAINST LEISHMANIASIS 4525 FIG. 6. Effect of nasally administered rlack and LACK DNA on subsequent infection with L. amazonensis. Mice were vaccinated intranasally with 10 g of rlack ( and solid bar), 30 g of LACK DNA (F and solid bar), 10 mg of LaAg (ƒ and solid bar), or 30 g of DNA ( ) and received a booster vaccination 7 days later. The controls received PBS alone (E and open bar). One week after the booster vaccination animals were infected with 10 5 fluorescent L. amazonensis cells in a footpad. (A) Lesion sizes on different days. (B) On day 160 of infection, the parasite loads in the infected feet were expressed in fluorescence units. The values are means and standard deviations (n 5). An asterisk indicates that the P value is individuals may take up to 6 months following intramuscular vaccination with L. amazonensis antigens to optimally respond with IFN- production (33). The time lag requirement for an effective immune response may be further substantiated by the observation that the vaccinating effect is better 4 months after immunization (Fig. 4) than 1 week after immunization (Fig. 1). The long-term effectiveness of intranasal vaccination against intracellular pathogens has a corollary in tuberculosis, in which intranasal vaccination with BCG was recently shown to induce IFN- production in the lungs and in the spleen (5). In that study, similar to the results observed with Leishmania, intranasal vaccination was more effective at 6 months than at 3 months postvaccination for preventing Mycobacterium tuberculosis growth at those sites. FIG. 7. IFN- production by mice infected 1 week after LACK vaccination. Mice (n 5) were vaccinated intranasally with rlack (striped bars), LACK DNA (solid bars), or PBS alone (open bars) and infected as described in the legend to Fig 6. On day 160 of infection, the levels of IFN- produced by rlack (10 g/ml)-stimulated popliteal lymph node cells (PL) and cervical lymph node cells (CL) in the culture supernatants were measured by ELISA. The values are means and standard deviations for triplicate samples. One asterisk, P 0.05 for a comparison with PBS controls; two asterisks, P 0.01 for a comparison with PBS controls. We showed here that intranasal vaccination with LaAg promoted a balanced TH1-TH2 response in the periphery, as demonstrated by upregulation of both IFN- and IL-10 production in the popliteal lymph nodes at either 1 week or 4 months postvaccination. In spite of this, subsequent infection with L. amazonensis resulted in controlled parasite growth. The following mechanisms were proposed to explain the effective protection achieved in the presence of upregulated IL-10: (i) the eventual predominant TH1 response (IFN- ) in later stages of infection was sufficient for protection; (ii) upregulated TH2 cytokines (IL-10 and IL-4) are not sufficient to downregulate the TH1 responses during L. amazonensis infection of vaccinated mice (this possibility is supported by the finding that susceptibility of mice to L. amazonensis is not controlled by TH2 responses to the same extent as L. major infection [15]); and (iii) immunomodulatory factors other than the classical TH1 and TH2 cytokines may be favorably modulated by nasal vaccination (this possibility is supported by the finding that upon vaccination mice were capable of mounting a stronger DTH response to parasite antigens in vivo despite the higher level of IL-10 production in vitro [Fig. 3]; in this sense, tumor growth factor is a good candidate as this cytokine is upregulated in aggravated murine cutaneous leishmaniasis [3]). The protection achieved by intranasal immunization was accompanied by long-lasting immunological memory and adaptive immunity, as if LaAg possessed some kind of selfadjuvanticity. Intestinal or nasal antigen uptake may result in peripheral tolerance, and this was the starting point of the present work with disease-promoting parasite antigens. However, systemic immunity may also be achieved after antigen administration through the mucosa (1), and the balance between tolerance and immunity is a function of the nature of the antigen, the antigen dosage, the antigen form (soluble or particulated), the site of antigen administration, and the associa-
6 4526 PINTO ET AL. INFECT. IMMUN. tion with adjuvants (23). Thus, it seems that although both the nasal and oral mucosa may be sites for effective vaccination with LaAg, the antigen may be differentially presented at the two sites, as indicated by the partial tolerance and the active systemic immunity, respectively. The advantage of a crude vaccine, such as LaAg, for mucosal vaccination is that the vaccine is already considered safe for parenteral use in humans (24). However, a defined vaccine may be more appropriate for standardization reasons. We found that soluble rlack, at doses similar to those used for LaAg, had no effect against infection with L. amazonensis in short-term immunized animals (Fig. 6), which is consistent with the finding of McSorley et al. showing that 10 g to1mg of nasally administered rlack failed to protect BALB/c mice against infection with L. major (26). However, the possibility that the effectiveness of rlack becomes apparent in longterm immunized mice should not be discarded. The slight increase in IFN- production after rlack immunization was insufficient to induce protection and was not due to upregulated TH2 cytokines, as both the IL-10 and IL-4 levels were comparable with those of PBS controls (data not shown). It is possible that the soluble nature and the low glycosylation level of the recombinant protein impaired its uptake by M cells in the nasal mucosal epithelium (18), thus explaining the absence of activity. Use of LACK entrapped in biodegradable microspheres would better clarify this issue. On the other hand, LACK DNA effectively delayed the onset of infection and induced strong IFN- production in both peripheral and noseassociated cervical lymph nodes (Fig. 6 and 7). Kinetic studies with intranasally administered -galactosidase-encoding plasmid DNA revealed an absorption rate of 14% and active expression of mrna in mucosa-associated and peripheral lymph nodes 24 h later (30), but the exact mechanism by which plasmid DNA vaccines enter the nasal mucosa and tissue cells remains unclear (2). Susceptibility of BALB/c mice to L. major infection has been correlated with early production of IL-4 by LACK-reactive CD4 T cells that drive the TH2 responses (20). It is worth noting that the presence of LACK-reactive memory T cells in animals not previously exposed to the parasites has been explained by their cross-reactivity with antigens from microbes of the normal intestinal flora (17). In the study of Julia et al., antigen-presenting cells from mesenteric lymph nodes of naive BALB/c mice induced the proliferation of LACK-specific T cells in vitro. Thus, it is possible that LACKspecific mucosal system-associated cells respond differently than peripheral T cells to challenge by mucosally administered LACK antigen as present in LaAg or as DNA transcripts, promoting the development of systemic protective immune responses. Previous studies have demonstrated the efficacy of parenteral DNA vaccines against murine leishmaniasis (4, 12, 28, 34, 39), but this is the first study to report the efficacy of a mucosal DNA vaccine for this disease. Altogether, the work presented here demonstrates the feasibility of using the nasal mucosa for effective delivery of both crude antigens and DNA against cutaneous leishmaniasis. The demonstration of long-lasting and strong effects of nasally administered LaAg and LACK DNA may open new frontiers for noninvasive human vaccination against leishmaniasis. ACKNOWLEDGMENTS This work was supported by Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Brazil. V.L. thanks the Spanish Ministry of Science and Technology for grant BIO P4-04. REFERENCES 1. Bakke, H., K. Lie, I. L. Haugen, G. E. Korsvold, E. A. Høiby, L. M. Næss, J. Holst, I. S. Aaberge, F. Oftung, and B. Haneberg Meningococcal outer membrane vesicle vaccine given intranasally can induce immunological memory and booster responses without evidence of tolerance. Infect. Immun. 69: Barnes, A. G. C., C. Barnfield, R. Brew, and L. S. Klavinskis Recent developments in mucosal delivery of pdna vaccines. Curr. Opin. Mol. Ther. 2: Belkaid, Y The role of CD4 CD25 regulatory T cells in Leishmania infection. Expert Opin. Biol. Ther. 3: Campbell, M., H. Diao, J. X. Ji, and L. Soong DNA immunization with the gene encoding P4 nuclease of Leishmania amazonensis protects mice against cutaneous leishmaniasis. Infect. Immun. 71: Chen, L. H., J. Wang, A. Zganiacz, and Z. Xing Single intranasal mucosal Mycobactetium bovis BCG vaccination confers improved protection compared to subcutaneous vaccination against pulmonary tuberculosis. Infect. Immun. 72: Croft, S. L., and G. H. Coombs Leishmaniasis current chemotherapy and recent advances in the search for novel drugs. Trends Parasitol. 19: Czerkinsky, C., F. Anjuere, J. R. McGhee, A. George-Chandy, J. Holmgren, M. Kieny, K. Fujiyashi, J. F. Mestecky, V. Pierrefite-Carle, C. Rask, and J. Sun Mucosal immunity and tolerance: relevance to vaccine development. Immunol. Rev. 170: Dhaliwal, J. S., and F. Y. Liew Induction of delayed-type hypersensitivity to Leishmania major and the concomitant acceleration of disease development in progressive murine cutaneous leishmaniasis. Infect. Immun. 55: Dumonteil, E., R. S. M. Jesus, E. O. Javier, and G. M. M. del Rosario DNA vaccines induce partial protection against Leishmania mexicana. Vaccine 21: Fujihashi, K., T. Koga, F. W. van Ginkel, Y. Hagiwara, and J. R. McGhee A dilemma for mucosal vaccination: efficacy versus toxicity using enterotoxin-based adjuvants. Vaccine 20: Gonzalez-Aseguinolaza, G., S. Taladriz, A. Marquet, and V. Larraga Molecular cloning, cell localization and binding affinity to DNA replication proteins of the p36/lack protective antigen from Leishmania infantum. Eur. J. Biochem. 259: Gurunathan, S., D. L. Sacks, D. R. Brown, S. L. Reiner, H. Charest, N. Glaichenhaus, and R. A. Seder Vaccination with DNA encoding the immunodominant LACK parasite antigen confers protective immunity to mice infected with Leishmania major. J. Exp. Med. 186: Hamann, A., D. P. Andrew, D. Jablonskiwestrich, B. Holzmann, and E. C. Butcher Role of alpha (4)-integrins in lymphocyte homing to mucosal tissues in-vivo. J. Immunol. 152: Handman, E Leishmaniasis: current status of vaccine development. Clin. Microbiol. Rev. 14: Jones, D. E., M. R. Ackermann, U. Wille, C. A. Hunter, and P. Scott Early enhanced Th1 response after Leishmania amazonensis infection of C57BL/6 interleukin-10-deficient mice does not lead to resolution of infection. Infect. Immun. 70: Julia, V., M. Rassoulzadegan, and N. Glaichenhaus Resistance to Leishmania major induced by tolerance to a single antigen. Science 274: Julia, V., S. S. McSorley, L. Malherbe, J. P. Breittmayer, F. Girard-Pipau, A. Beck, and N. Glaichenhaus Priming by microbial antigens from the intestinal flora determines the ability of CD4 T cells to rapidly secrete IL-4 in BALB/c mice infected with Leishmania major. J. Immunol. 165: Kaiserlian, D., and N. Etchart Entry sites for oral vaccines and drugs: a role for M cells, enterocytes and dendritic cells? Semin. Immunol. 11: Kenney, R. T., D. L. Sacks, J. P. Sypek, L. Vilela, A. A. Gam, and K. Evans-Davis Protective immunity using recombinant human IL-12 and alum as adjuvants in a primate model of cutaneous leishmaniasis. J. Immunol. 163: Launois, P., I. Maillard, S. Pingel, K. G. Swihart, I. Xenarios, H. Acha- Orbea, H. Diggelmann, R. M. Locksley, H. R. MacDonald, and J. A. Louis IL-4 rapidly produced by V beta 4 V alpha 8 CD4 T cells instructs Th2 development and susceptibility to Leishmania major in BALB/c mice. Immunity 6: Liew, F. Y., C. Hale, and J. G. Howard Prophylactic immunization against experimental leishmaniasis. IV. Subcutaneous immunization prevents the induction of protective immunity against fatal Leishmania major infection. J. Immunol. 135:
7 VOL. 72, 2004 INTRANASAL VACCINATION AGAINST LEISHMANIASIS Lira, R., S. Sundar, A. Makharia, R. Kenney, A. Gam, E. Saraiva, and D. Sacks Evidence that the high incidence of treatment failures in Indian kala-azar is due to the emergence of antimony-resistant strains of Leishmania donovani. J. Infect. Dis. 180: Makala, L. H. C., Y. Nishikawa, N. Suzuki, and H. Nagasawa Immunology antigen-presenting cells in the gut. J. Biomed. Sci. 11: Marzochi, K. B. F., M. C. A. Marzochi, A. F. Silva, N. Grativol, R. Duarte, E. M. Conort, and F. Modabber Phase 1 study of an inactivated vaccine against American tegumentary leishmaniasis in normal volunteers in Brazil. Mem. Inst. Oswaldo Cruz 93: McCluskie, M. J., and H. L. Davis CpG DNA is a potent enhancer of systemic and mucosal immune responses against hepatitis B surface antigen with intranasal administration to mice. J. Immunol. 161: McSorley, S. J., C. Rask, R. Pichot, V. Julia, C. Czerkinsky, and N. Glaichenhaus Selective tolerization of Th1-like cells after nasal administration of a cholera toxoid-lack conjugate. Eur. J. Immunol. 28: Melby, P. C., J. Yang, W. Zhao, L. E. Perez, and J. Cheng Leishmania donovani p36(lack) DNA vaccine is highly immunogenic but not protective against experimental visceral leishmaniasis. Infect. Immun. 69: Mendez, A., Y. Belkaid, R. A. Seder, and D. Sacks Optimisation of DNA vaccination against cutaneous leishmaniasis. Vaccine 20: Mosmann, T. R., and S. Sad The expanding universe of T-cell subsets: Th1, Th2 and more. Immunol. Today 17: Oh, Y. K., J. P. Kim, T. S. Hwang, J. J. Ko, J. M. Kim, J. S. Yang, and C. K. Kim Nasal absorption and biodistribution of plasmid DNA: an alternative route of DNA vaccine delivery. Vaccine 19: Okuno, T., M. Takeuchi, Y. Matsumoto, H. Otsuka, and Y. Matsumoto Pretreatment of Leishmania homologue of receptors for activated C kinase (LACK) promotes disease progression caused by Leishmania amazonensis. Exp. Anim. 51: Editor: W. A. Petri, Jr. 32. Pinto, E. F., M. M. Cortezia, and B. Rossi-Bergmann Interferongamma-inducing oral vaccination with Leishmania amazonensis antigens protects BALB/c and C57BL/6 mice against cutaneous leishmaniasis. Vaccine 21: Pompeu, M. M. L., C. Brodskyn, M. J. Teixeira, J. Clarencio, J. Van Weyenberg, I. C. B. Coelho, S. A. Cardoso, A. Barral, and M. Barral-Netto Differences in gamma interferon production in vitro predict the pace of the in vivo response to Leishmania amazonensis in healthy volunteers. Infect. Immun. 69: Ramiro M. J., J. J. Zarate, T. Hanke, D. Rodriguez, J. R. Rodriguez, M. Esteban, J. Lucientes, J. A. Castillo, and V. Larraga Protection in dogs against visceral leishmaniasis caused by Leishmania infantum is achieved by immunization with a heterologous prime-boost regime using DNA and vaccinia recombinant vectors expressing LACK. Vaccine 21: Reiner, S. L., and R. M. Locksley The regulation of immunity to Leishmania major. Annu. Rev. Immunol. 13: Rossi-Bergmann B., A. Lenglet, C. R. Bezerra-Santos, D. Costa-Pinto, and Y. M. Traub-Czeko Use of fluorescent Leishmania for faster quantitation of parasite growth in vitro and in vivo. Mem. Inst. Oswaldo Cruz 94(Suppl. II): Shah, J. A., P. A. Darrah, D. R. Ambrozak, T. N. Turon, S. Mendez, J. Kirman, C. Y. Wu, N. Glaichenhaus, and R. A. Seder Dendritic cells are responsible for the capacity of CpG oligodeoxynucleotides to act as an adjuvant for protective vaccine immunity against Leishmania major in mice. J. Exp. Med. 198: World Health Organization/TDR Tropical diseases research leishmaniasis. Fourteen Programme Report 22. World Health Organization, Geneva, Switzerland. 39. Xu, D., and F. Y. Liew Genetic vaccination against leishmaniasis. Vaccine 12: Downloaded from on October 1, 2018 by guest
Resolution of an Infection with Leishmania braziliensis Confers Complete Protection to a Subsequent Challenge with Leishmania major in BALB/c Mice
 Mem Inst Oswaldo Cruz, Rio de Janeiro, Vol. 94(1): 71-76, Jan./Feb. 1999 Resolution of an Infection with Leishmania braziliensis Confers Complete Protection to a Subsequent Challenge with Leishmania major
Mem Inst Oswaldo Cruz, Rio de Janeiro, Vol. 94(1): 71-76, Jan./Feb. 1999 Resolution of an Infection with Leishmania braziliensis Confers Complete Protection to a Subsequent Challenge with Leishmania major
New insights on leishmaniasis in immunosuppressive conditions
 New insights on leishmaniasis in immunosuppressive conditions Javier Moreno Immunoparasitology Unit WHO Collaborative Center for Leishmaniasis Centro Nacional de Microbiología INSTITUTO DE SALUD CARLOS
New insights on leishmaniasis in immunosuppressive conditions Javier Moreno Immunoparasitology Unit WHO Collaborative Center for Leishmaniasis Centro Nacional de Microbiología INSTITUTO DE SALUD CARLOS
Title: Oral administration of PPC enhances antigen-specific CD8+ T cell responses while reducing IgE levels in sensitized mice.
 Author's response to reviews Title: Oral administration of PPC enhances antigen-specific CD8+ T cell responses while reducing IgE levels in sensitized mice. Authors: Mike Burrows (mburrows@tampabayresearch.org)
Author's response to reviews Title: Oral administration of PPC enhances antigen-specific CD8+ T cell responses while reducing IgE levels in sensitized mice. Authors: Mike Burrows (mburrows@tampabayresearch.org)
Trends in vaccinology
 Trends in vaccinology Mathieu Peeters, MD Joint Conference of European Human Pharmacological Societies and Joint Conference of European Human Pharmacological Societies and 20th Anniversary of AGAH March
Trends in vaccinology Mathieu Peeters, MD Joint Conference of European Human Pharmacological Societies and Joint Conference of European Human Pharmacological Societies and 20th Anniversary of AGAH March
Medical Virology Immunology. Dr. Sameer Naji, MB, BCh, PhD (UK) Head of Basic Medical Sciences Dept. Faculty of Medicine The Hashemite University
 Medical Virology Immunology Dr. Sameer Naji, MB, BCh, PhD (UK) Head of Basic Medical Sciences Dept. Faculty of Medicine The Hashemite University Human blood cells Phases of immune responses Microbe Naïve
Medical Virology Immunology Dr. Sameer Naji, MB, BCh, PhD (UK) Head of Basic Medical Sciences Dept. Faculty of Medicine The Hashemite University Human blood cells Phases of immune responses Microbe Naïve
Effector mechanisms of cell-mediated immunity: Properties of effector, memory and regulatory T cells
 ICI Basic Immunology course Effector mechanisms of cell-mediated immunity: Properties of effector, memory and regulatory T cells Abul K. Abbas, MD UCSF Stages in the development of T cell responses: induction
ICI Basic Immunology course Effector mechanisms of cell-mediated immunity: Properties of effector, memory and regulatory T cells Abul K. Abbas, MD UCSF Stages in the development of T cell responses: induction
IOM Immunization Safety Review 11/12/2001. Immunological Competition and the Infant Immune Response to Vaccines
 IOM Immunization Safety Review 11/12/2001 Immunological Competition and the Infant Immune Response to Vaccines Richard Insel University of Rochester Goals Neonatal and Infant Immune System Broad Effects
IOM Immunization Safety Review 11/12/2001 Immunological Competition and the Infant Immune Response to Vaccines Richard Insel University of Rochester Goals Neonatal and Infant Immune System Broad Effects
T-cell activation T cells migrate to secondary lymphoid tissues where they interact with antigen, antigen-presenting cells, and other lymphocytes:
 Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
T-cell activation T cells migrate to secondary lymphoid tissues where they interact with antigen, antigen-presenting cells, and other lymphocytes:
 Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
Analysis of Enhancing Effect of Sand Fly Saliva on Leishmania Infection in Mice
 INFCTION AND IMMUNITY, May 1991, p. 1592-1598 19-9567/91/51592-7$2./ Copyright X) 1991, American Society for Microbiology Vol. 59, No. 5 Analysis of nhancing ffect of Sand Fly Saliva on Leishmania Infection
INFCTION AND IMMUNITY, May 1991, p. 1592-1598 19-9567/91/51592-7$2./ Copyright X) 1991, American Society for Microbiology Vol. 59, No. 5 Analysis of nhancing ffect of Sand Fly Saliva on Leishmania Infection
Leishmaniasis: Challenges for Vaccine Development
 Leishmaniasis: Challenges for Vaccine Development Steven G. Reed Infectious Disease Research Institute Seattle IDRI Goals: Leishmaniasis To improve existing vaccines for use in therapy and prevention.
Leishmaniasis: Challenges for Vaccine Development Steven G. Reed Infectious Disease Research Institute Seattle IDRI Goals: Leishmaniasis To improve existing vaccines for use in therapy and prevention.
Molecular and Cellular Basis of Immune Protection of Mucosal Surfaces
 Molecular and Cellular Basis of Immune Protection of Mucosal Surfaces Department of Biologic & Materials Sciences School of Dentistry University of Michigan Ann Arbor, Michigan 48109-1078 1 Image quality
Molecular and Cellular Basis of Immune Protection of Mucosal Surfaces Department of Biologic & Materials Sciences School of Dentistry University of Michigan Ann Arbor, Michigan 48109-1078 1 Image quality
TITLE: MODULATION OF T CELL TOLERANCE IN A MURINE MODEL FOR IMMUNOTHERAPY OF PROSTATIC ADENOCARCINOMA
 AD Award Number: DAMD17-01-1-0085 TITLE: MODULATION OF T CELL TOLERANCE IN A MURINE MODEL FOR IMMUNOTHERAPY OF PROSTATIC ADENOCARCINOMA PRINCIPAL INVESTIGATOR: ARTHUR A HURWITZ, Ph.d. CONTRACTING ORGANIZATION:
AD Award Number: DAMD17-01-1-0085 TITLE: MODULATION OF T CELL TOLERANCE IN A MURINE MODEL FOR IMMUNOTHERAPY OF PROSTATIC ADENOCARCINOMA PRINCIPAL INVESTIGATOR: ARTHUR A HURWITZ, Ph.d. CONTRACTING ORGANIZATION:
Leishmaniasis is an endemic disease occurring in several
 Vaccination with Phosphoglycan-Deficient Leishmania major Protects Highly Susceptible Mice from Virulent Challenge without Inducing a Strong Th1 Response 1 Jude E. Uzonna,* Gerald F. Späth, 2 Stephen M.
Vaccination with Phosphoglycan-Deficient Leishmania major Protects Highly Susceptible Mice from Virulent Challenge without Inducing a Strong Th1 Response 1 Jude E. Uzonna,* Gerald F. Späth, 2 Stephen M.
A Multicistronic DNA Vaccine Induces Significant Protection against Tuberculosis in Mice and Offers Flexibility in the Expressed Antigen Repertoire.
 Company LOGO A Multicistronic DNA Vaccine Induces Significant Protection against Tuberculosis in Mice and Offers Flexibility in the Expressed Antigen Repertoire. Fayaz Ahmad Mir, Stefan H. E. Kaufmann,
Company LOGO A Multicistronic DNA Vaccine Induces Significant Protection against Tuberculosis in Mice and Offers Flexibility in the Expressed Antigen Repertoire. Fayaz Ahmad Mir, Stefan H. E. Kaufmann,
Genetics. Environment. You Are Only 10% Human. Pathogenesis of IBD. Advances in the Pathogenesis of IBD: Genetics Leads to Function IBD
 Advances in the Pathogenesis of IBD: Genetics Leads to Function Pathogenesis of IBD Environmental Factors Microbes Scott Plevy, MD Associate Professor of Medicine, Microbiology & Immunology UNC School
Advances in the Pathogenesis of IBD: Genetics Leads to Function Pathogenesis of IBD Environmental Factors Microbes Scott Plevy, MD Associate Professor of Medicine, Microbiology & Immunology UNC School
Immunization with a Toll-Like Receptor 7 and/or 8 Agonist Vaccine Adjuvant Increases Protective Immunity against Leishmania major in BALB/c Mice
 INFECTION AND IMMUNITY, Aug. 2008, p. 3777 3783 Vol. 76, No. 8 0019-9567/08/$08.00 0 doi:10.1128/iai.01527-07 Copyright 2008, American Society for Microbiology. All Rights Reserved. Immunization with a
INFECTION AND IMMUNITY, Aug. 2008, p. 3777 3783 Vol. 76, No. 8 0019-9567/08/$08.00 0 doi:10.1128/iai.01527-07 Copyright 2008, American Society for Microbiology. All Rights Reserved. Immunization with a
Chapter 24 The Immune System
 Chapter 24 The Immune System The Immune System Layered defense system The skin and chemical barriers The innate and adaptive immune systems Immunity The body s ability to recognize and destroy specific
Chapter 24 The Immune System The Immune System Layered defense system The skin and chemical barriers The innate and adaptive immune systems Immunity The body s ability to recognize and destroy specific
Gene Vaccine Dr. Sina Soleimani
 Gene Vaccine Dr. Sina Soleimani Human Viral Vaccines Quality Control Laboratory (HVVQC) Titles 1. A short Introduction of Vaccine History 2. First Lineage of Vaccines 3. Second Lineage of Vaccines 3. New
Gene Vaccine Dr. Sina Soleimani Human Viral Vaccines Quality Control Laboratory (HVVQC) Titles 1. A short Introduction of Vaccine History 2. First Lineage of Vaccines 3. Second Lineage of Vaccines 3. New
1. Overview of Adaptive Immunity
 Chapter 17A: Adaptive Immunity Part I 1. Overview of Adaptive Immunity 2. T and B Cell Production 3. Antigens & Antigen Presentation 4. Helper T cells 1. Overview of Adaptive Immunity The Nature of Adaptive
Chapter 17A: Adaptive Immunity Part I 1. Overview of Adaptive Immunity 2. T and B Cell Production 3. Antigens & Antigen Presentation 4. Helper T cells 1. Overview of Adaptive Immunity The Nature of Adaptive
Differences in Gamma Interferon Production In Vitro Predict the Pace of the In Vivo Response to Leishmania amazonensis in Healthy Volunteers
 INFECTION AND IMMUNITY, Dec. 2001, p. 7453 7460 Vol. 69, No. 12 0019-9567/01/$04.00 0 DOI: 10.1128/IAI.69.12.7453 7460.2001 Copyright 2001, American Society for Microbiology. All Rights Reserved. Differences
INFECTION AND IMMUNITY, Dec. 2001, p. 7453 7460 Vol. 69, No. 12 0019-9567/01/$04.00 0 DOI: 10.1128/IAI.69.12.7453 7460.2001 Copyright 2001, American Society for Microbiology. All Rights Reserved. Differences
Chapter 22: The Lymphatic System and Immunity
 Bio40C schedule Lecture Immune system Lab Quiz 2 this week; bring a scantron! Study guide on my website (see lab assignments) Extra credit Critical thinking questions at end of chapters 5 pts/chapter Due
Bio40C schedule Lecture Immune system Lab Quiz 2 this week; bring a scantron! Study guide on my website (see lab assignments) Extra credit Critical thinking questions at end of chapters 5 pts/chapter Due
Determinants of Immunogenicity and Tolerance. Abul K. Abbas, MD Department of Pathology University of California San Francisco
 Determinants of Immunogenicity and Tolerance Abul K. Abbas, MD Department of Pathology University of California San Francisco EIP Symposium Feb 2016 Why do some people respond to therapeutic proteins?
Determinants of Immunogenicity and Tolerance Abul K. Abbas, MD Department of Pathology University of California San Francisco EIP Symposium Feb 2016 Why do some people respond to therapeutic proteins?
VACCINATION. DR.FATIMA ALKHALEDY M.B.Ch.B;F.I.C.M.S/C.M.
 VACCINATION DR.FATIMA ALKHALEDY M.B.Ch.B;F.I.C.M.S/C.M. IMMUNIZATION Immunization is defined as the procedure by which the body is prepared to fight against a specific disease. It is used to induce the
VACCINATION DR.FATIMA ALKHALEDY M.B.Ch.B;F.I.C.M.S/C.M. IMMUNIZATION Immunization is defined as the procedure by which the body is prepared to fight against a specific disease. It is used to induce the
ECM1 controls T H 2 cell egress from lymph nodes through re-expression of S1P 1
 ZH, Li et al, page 1 ECM1 controls T H 2 cell egress from lymph nodes through re-expression of S1P 1 Zhenhu Li 1,4,Yuan Zhang 1,4, Zhiduo Liu 1, Xiaodong Wu 1, Yuhan Zheng 1, Zhiyun Tao 1, Kairui Mao 1,
ZH, Li et al, page 1 ECM1 controls T H 2 cell egress from lymph nodes through re-expression of S1P 1 Zhenhu Li 1,4,Yuan Zhang 1,4, Zhiduo Liu 1, Xiaodong Wu 1, Yuhan Zheng 1, Zhiyun Tao 1, Kairui Mao 1,
Mitochondrial Trifunctional Protein (TFP) Protein Quantity Microplate Assay Kit
 PROTOCOL Mitochondrial Trifunctional Protein (TFP) Protein Quantity Microplate Assay Kit DESCRIPTION Mitochondrial Trifunctional Protein (TFP) Protein Quantity Microplate Assay Kit Sufficient materials
PROTOCOL Mitochondrial Trifunctional Protein (TFP) Protein Quantity Microplate Assay Kit DESCRIPTION Mitochondrial Trifunctional Protein (TFP) Protein Quantity Microplate Assay Kit Sufficient materials
Acquired Immunity 2. - Vaccines & Immunological Memory - Wataru Ise. WPI Immunology Frontier Research Center (IFReC) Osaka University.
 Acquired Immunity 2 - Vaccines & Immunological Memory - Wataru Ise WPI Immunology Frontier Research Center (IFReC) Osaka University Outline 1. What is vaccine (vaccination)? 2. What is immunological memory?
Acquired Immunity 2 - Vaccines & Immunological Memory - Wataru Ise WPI Immunology Frontier Research Center (IFReC) Osaka University Outline 1. What is vaccine (vaccination)? 2. What is immunological memory?
Kinetics of Local and Systemic Immune Responses after Vaginal Immunization with Recombinant Cholera Toxin B Subunit in Humans
 CLINICAL AND DIAGNOSTIC LABORATORY IMMUNOLOGY, Mar. 2005, p. 447 452 Vol. 12, No. 3 1071-412X/05/$08.00 0 doi:10.1128/cdli.12.3.447 452.2005 Copyright 2005, American Society for Microbiology. All Rights
CLINICAL AND DIAGNOSTIC LABORATORY IMMUNOLOGY, Mar. 2005, p. 447 452 Vol. 12, No. 3 1071-412X/05/$08.00 0 doi:10.1128/cdli.12.3.447 452.2005 Copyright 2005, American Society for Microbiology. All Rights
Vaccination Recommendations Practice and Shelter-Housed Dogs
 Vaccination Recommendations Practice and Shelter-Housed Dogs 1. MIXING VACCINES. Can different types of vaccines be mixed in the same syringe? No. Unless specifically stated on the product label (package
Vaccination Recommendations Practice and Shelter-Housed Dogs 1. MIXING VACCINES. Can different types of vaccines be mixed in the same syringe? No. Unless specifically stated on the product label (package
Mechanistic Studies of Pentamidine Analogs on Leishmania donovani Promastigotes
 Mechanistic Studies of Pentamidine Analogs on Leishmania donovani Promastigotes Undergraduate Honors Thesis The Ohio State University, College of Pharmacy Division of Medicinal Chemistry and Pharmacognosy
Mechanistic Studies of Pentamidine Analogs on Leishmania donovani Promastigotes Undergraduate Honors Thesis The Ohio State University, College of Pharmacy Division of Medicinal Chemistry and Pharmacognosy
Antigen Presentation and T Lymphocyte Activation. Abul K. Abbas UCSF. FOCiS
 1 Antigen Presentation and T Lymphocyte Activation Abul K. Abbas UCSF FOCiS 2 Lecture outline Dendritic cells and antigen presentation The role of the MHC T cell activation Costimulation, the B7:CD28 family
1 Antigen Presentation and T Lymphocyte Activation Abul K. Abbas UCSF FOCiS 2 Lecture outline Dendritic cells and antigen presentation The role of the MHC T cell activation Costimulation, the B7:CD28 family
Nanomedicine & Drug Delivery Systems Group Research Institute for Medicines and Pharmaceutical Sciences (imed.ul)
 Faculty of Pharmacy of the University of Lisbon Nanomedicine & Drug Delivery Systems Group Research Institute for Medicines and Pharmaceutical Sciences (imed.ul) Leishmania in southern Europe. (Duiardin,
Faculty of Pharmacy of the University of Lisbon Nanomedicine & Drug Delivery Systems Group Research Institute for Medicines and Pharmaceutical Sciences (imed.ul) Leishmania in southern Europe. (Duiardin,
Designing a DNA Vaccine-based Leishmania major Polytope (Preliminary Report)
 Iran J Parasitol Tehran University of Medical Sciences Publication http:// tums.ac.ir Open access Journal at http:// ijpa.tums.ac.ir Iranian Society of Parasitology http:// isp.tums.ac.ir Short Communication
Iran J Parasitol Tehran University of Medical Sciences Publication http:// tums.ac.ir Open access Journal at http:// ijpa.tums.ac.ir Iranian Society of Parasitology http:// isp.tums.ac.ir Short Communication
Unit 5 The Human Immune Response to Infection
 Unit 5 The Human Immune Response to Infection Unit 5-page 1 FOM Chapter 21 Resistance and the Immune System: Innate Immunity Preview: In Chapter 21, we will learn about the branch of the immune system
Unit 5 The Human Immune Response to Infection Unit 5-page 1 FOM Chapter 21 Resistance and the Immune System: Innate Immunity Preview: In Chapter 21, we will learn about the branch of the immune system
LESSON 2: THE ADAPTIVE IMMUNITY
 Introduction to immunology. LESSON 2: THE ADAPTIVE IMMUNITY Today we will get to know: The adaptive immunity T- and B-cells Antigens and their recognition How T-cells work 1 The adaptive immunity Unlike
Introduction to immunology. LESSON 2: THE ADAPTIVE IMMUNITY Today we will get to know: The adaptive immunity T- and B-cells Antigens and their recognition How T-cells work 1 The adaptive immunity Unlike
Third line of Defense
 Chapter 15 Specific Immunity and Immunization Topics -3 rd of Defense - B cells - T cells - Specific Immunities Third line of Defense Specific immunity is a complex interaction of immune cells (leukocytes)
Chapter 15 Specific Immunity and Immunization Topics -3 rd of Defense - B cells - T cells - Specific Immunities Third line of Defense Specific immunity is a complex interaction of immune cells (leukocytes)
Adaptive immune responses: T cell-mediated immunity
 MICR2209 Adaptive immune responses: T cell-mediated immunity Dr Allison Imrie allison.imrie@uwa.edu.au 1 Synopsis: In this lecture we will discuss the T-cell mediated immune response, how it is activated,
MICR2209 Adaptive immune responses: T cell-mediated immunity Dr Allison Imrie allison.imrie@uwa.edu.au 1 Synopsis: In this lecture we will discuss the T-cell mediated immune response, how it is activated,
Vaccination-Strategies
 Vaccination-Strategies Active immunity produced by vaccine Immunity and immunologic memory similar to natural infection but without risk of disease. General Rule: The more similar a vaccine is to the disease-causing
Vaccination-Strategies Active immunity produced by vaccine Immunity and immunologic memory similar to natural infection but without risk of disease. General Rule: The more similar a vaccine is to the disease-causing
Immunology 2011 Lecture 2 Immune Reactions, Ag/Ab 20 September
 Immunology 2011 Lecture 2 Immune Reactions, Ag/Ab 20 September HANDOUTS #1 Roadmap TODAY Immune Reactions, Antigen/Antibody, Core Notes Chapters 1 & 2 and Appendix 1,2 (Precipitin Curve, Ab Labeling) Ag/Ab
Immunology 2011 Lecture 2 Immune Reactions, Ag/Ab 20 September HANDOUTS #1 Roadmap TODAY Immune Reactions, Antigen/Antibody, Core Notes Chapters 1 & 2 and Appendix 1,2 (Precipitin Curve, Ab Labeling) Ag/Ab
Monitoring tuberculosis progression using MRI and stereology
 Monitoring tuberculosis progression using MRI and stereology TB the problem Estimated number of new cases in 2007 2 million deaths; 9 million new cases p.a. TB kills someone every 15 secs, 9,153 cases
Monitoring tuberculosis progression using MRI and stereology TB the problem Estimated number of new cases in 2007 2 million deaths; 9 million new cases p.a. TB kills someone every 15 secs, 9,153 cases
Study Guide 23, 24 & 47
 Study Guide 23, 24 & 47 STUDY GUIDE SECTION 23-3 Bacteria and Humans Name Period Date 1. One bacterial disease that is transmitted by contaminated drinking water is a. Lyme disease b. gonorrhea c. tuberculosis
Study Guide 23, 24 & 47 STUDY GUIDE SECTION 23-3 Bacteria and Humans Name Period Date 1. One bacterial disease that is transmitted by contaminated drinking water is a. Lyme disease b. gonorrhea c. tuberculosis
Introduction. In the past 15 years, several technological advancements have open new perspectives and applications in the field of vaccinology.
 Introduction In the past 15 years, several technological advancements have open new perspectives and applications in the field of vaccinology. - Genomics: fasten antigen discovery for complex pathogens
Introduction In the past 15 years, several technological advancements have open new perspectives and applications in the field of vaccinology. - Genomics: fasten antigen discovery for complex pathogens
Introduction and overview of the immune System:
 MOLECULAR IMMUNOLOGY AND IMMUNOINFORMATICS STUDY NOTES UNIT-1 INTRODUCTION TO IMMUNE SYSTEM Introduction and overview of the immune System - Lymphatic System, Cells and Organs of the immune System - Types
MOLECULAR IMMUNOLOGY AND IMMUNOINFORMATICS STUDY NOTES UNIT-1 INTRODUCTION TO IMMUNE SYSTEM Introduction and overview of the immune System - Lymphatic System, Cells and Organs of the immune System - Types
Abstract. Introduction. M.O. Lásaro 1, A.M.B. Alves 1,2, H.C.R. Guillobel 3, D.F. Almeida 1 and L.C.S. Ferreira 1
 Brazilian Journal of Medical and Biological Research (1999) 32: 241-246 DNA and Salmonella vaccines against CFA/I ISSN 0100-879X 241 New vaccine strategies against enterotoxigenic Escherichia coli. II:
Brazilian Journal of Medical and Biological Research (1999) 32: 241-246 DNA and Salmonella vaccines against CFA/I ISSN 0100-879X 241 New vaccine strategies against enterotoxigenic Escherichia coli. II:
General Overview of Immunology. Kimberly S. Schluns, Ph.D. Associate Professor Department of Immunology UT MD Anderson Cancer Center
 General Overview of Immunology Kimberly S. Schluns, Ph.D. Associate Professor Department of Immunology UT MD Anderson Cancer Center Objectives Describe differences between innate and adaptive immune responses
General Overview of Immunology Kimberly S. Schluns, Ph.D. Associate Professor Department of Immunology UT MD Anderson Cancer Center Objectives Describe differences between innate and adaptive immune responses
Fluid movement in capillaries. Not all fluid is reclaimed at the venous end of the capillaries; that is the job of the lymphatic system
 Capillary exchange Fluid movement in capillaries Not all fluid is reclaimed at the venous end of the capillaries; that is the job of the lymphatic system Lymphatic vessels Lymphatic capillaries permeate
Capillary exchange Fluid movement in capillaries Not all fluid is reclaimed at the venous end of the capillaries; that is the job of the lymphatic system Lymphatic vessels Lymphatic capillaries permeate
Chapter 7 Conclusions
 VII-1 Chapter 7 Conclusions VII-2 The development of cell-based therapies ranging from well-established practices such as bone marrow transplant to next-generation strategies such as adoptive T-cell therapy
VII-1 Chapter 7 Conclusions VII-2 The development of cell-based therapies ranging from well-established practices such as bone marrow transplant to next-generation strategies such as adoptive T-cell therapy
Leishmania major Infection
 Immunotherapy with Imiquimod Increases the Efficacy of Glucantime Therapy of Leishmania major Infection Ghader Khalili 1, Faramarz Dobakhti 2, Hamid Mahmoudzadeh Niknam 1, Vahid Khaze 1, Fatemeh Partovi
Immunotherapy with Imiquimod Increases the Efficacy of Glucantime Therapy of Leishmania major Infection Ghader Khalili 1, Faramarz Dobakhti 2, Hamid Mahmoudzadeh Niknam 1, Vahid Khaze 1, Fatemeh Partovi
Immunization. Immunization procedure called vaccination and the immunizing agent called vaccine (or serum in historical references)
 Vaccines BIT 120 Immunization Immunization: a procedure designed to increase concentrations of antibodies and/or effector T-cells which are reactive against infection (or cancer). Immunization procedure
Vaccines BIT 120 Immunization Immunization: a procedure designed to increase concentrations of antibodies and/or effector T-cells which are reactive against infection (or cancer). Immunization procedure
Principles of Vaccination
 Immunology and Vaccine-Preventable Diseases Immunology is a complicated subject, and a detailed discussion of it is beyond the scope of this text. However, an understanding of the basic function of the
Immunology and Vaccine-Preventable Diseases Immunology is a complicated subject, and a detailed discussion of it is beyond the scope of this text. However, an understanding of the basic function of the
ACTIVATION OF T LYMPHOCYTES AND CELL MEDIATED IMMUNITY
 ACTIVATION OF T LYMPHOCYTES AND CELL MEDIATED IMMUNITY The recognition of specific antigen by naïve T cell induces its own activation and effector phases. T helper cells recognize peptide antigens through
ACTIVATION OF T LYMPHOCYTES AND CELL MEDIATED IMMUNITY The recognition of specific antigen by naïve T cell induces its own activation and effector phases. T helper cells recognize peptide antigens through
The Immune System. These are classified as the Innate and Adaptive Immune Responses. Innate Immunity
 The Immune System Biological mechanisms that defend an organism must be 1. triggered by a stimulus upon injury or pathogen attack 2. able to counteract the injury or invasion 3. able to recognise foreign
The Immune System Biological mechanisms that defend an organism must be 1. triggered by a stimulus upon injury or pathogen attack 2. able to counteract the injury or invasion 3. able to recognise foreign
LECTURE 12: MUCOSAL IMMUNITY GUT STRUCTURE
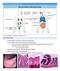 LECTURE 12: MUCOSAL IMMUNITY GUT STRUCTURE - Small intestine in humans is around 3-4 metres long - Internal surface of the small intestines are lined by villi o Villi are composed of absorptive cells (epithelial/enterocytes)
LECTURE 12: MUCOSAL IMMUNITY GUT STRUCTURE - Small intestine in humans is around 3-4 metres long - Internal surface of the small intestines are lined by villi o Villi are composed of absorptive cells (epithelial/enterocytes)
INFLAMMATORY CELL INFILTRATION AND HIGH ANTIBODY PRODUCTION IN BALB/c MICE CAUSED BY NATURAL EXPOSURE TO LUTZOMYIA LONGIPALPIS BITES
 Am. J. Trop. Med. Hyg., 72(1), 2005, pp. 94 98 Copyright 2005 by The American Society of Tropical Medicine and Hygiene INFLAMMATORY CELL INFILTRATION AND HIGH ANTIBODY PRODUCTION IN BALB/c MICE CAUSED
Am. J. Trop. Med. Hyg., 72(1), 2005, pp. 94 98 Copyright 2005 by The American Society of Tropical Medicine and Hygiene INFLAMMATORY CELL INFILTRATION AND HIGH ANTIBODY PRODUCTION IN BALB/c MICE CAUSED
PATHOGENICITY OF MICROORGANISMS
 PATHOGENICITY OF MICROORGANISMS Some microorganisms are : 1- Harmless microorganism, as normal flora 2- Harmfull microorganism, as pathogenic. A pathogenic microorganism is defined as one that causes or
PATHOGENICITY OF MICROORGANISMS Some microorganisms are : 1- Harmless microorganism, as normal flora 2- Harmfull microorganism, as pathogenic. A pathogenic microorganism is defined as one that causes or
The humoral immune responses to IBV proteins.
 The humoral immune responses to IBV proteins. E. Dan Heller and Rosa Meir The Hebrew University of Jerusalem, Israel COST FA1207 meeting WG2 + WG3, Budapest, Jan. 2015 1 IBV encodes four major structural
The humoral immune responses to IBV proteins. E. Dan Heller and Rosa Meir The Hebrew University of Jerusalem, Israel COST FA1207 meeting WG2 + WG3, Budapest, Jan. 2015 1 IBV encodes four major structural
T Cell-Derived IL-10 Determines Leishmaniasis Disease Outcome and Is Suppressed by a Dendritic Cell Based Vaccine
 T Cell-Derived IL-10 Determines Leishmaniasis Disease Outcome and Is Suppressed by a Dendritic Cell Based Vaccine Tobias Schwarz 1 a, Katharina A. Remer 1, Wiebke Nahrendorf 1, Anita Masic 1, Lisa Siewe
T Cell-Derived IL-10 Determines Leishmaniasis Disease Outcome and Is Suppressed by a Dendritic Cell Based Vaccine Tobias Schwarz 1 a, Katharina A. Remer 1, Wiebke Nahrendorf 1, Anita Masic 1, Lisa Siewe
Immunity and how vaccines work
 Immunity and how vaccines work Dr Mary O Meara National Immunisation Office Objectives of session An understanding of the following principles Overview of immunity Different types of vaccines and vaccine
Immunity and how vaccines work Dr Mary O Meara National Immunisation Office Objectives of session An understanding of the following principles Overview of immunity Different types of vaccines and vaccine
Current Strategies in HIV-1 Vaccine Development Using Replication-Defective Adenovirus as a Case Study
 Note: I have added some clarifying comments to the slides -- please click on Comments under View to see them. Current Strategies in HIV-1 Vaccine Development Using Replication-Defective Adenovirus as a
Note: I have added some clarifying comments to the slides -- please click on Comments under View to see them. Current Strategies in HIV-1 Vaccine Development Using Replication-Defective Adenovirus as a
Zheng, BJ; Du, LY; Zhao, GY; Lin, YP; Sui, HY; Chan, C; Ma, S; Guan, Y; Yuen, KY. Citation Hong Kong Medical Journal, 2008, v. 14 suppl. 4, p.
 Title Studies of SARS virus vaccines Author(s) Zheng, BJ; Du, LY; Zhao, GY; Lin, YP; Sui, HY; Chan, C; Ma, S; Guan, Y; Yuen, KY Citation Hong Kong Medical Journal, 2008, v. 14 suppl. 4, p. 39-43 Issued
Title Studies of SARS virus vaccines Author(s) Zheng, BJ; Du, LY; Zhao, GY; Lin, YP; Sui, HY; Chan, C; Ma, S; Guan, Y; Yuen, KY Citation Hong Kong Medical Journal, 2008, v. 14 suppl. 4, p. 39-43 Issued
Lines of Defense. Immunology, Immune Response, and Immunological Testing. Immunology Terminology
 Immunology, Immune Response, and Immunological Testing Lines of Defense If the First and Second lines of defense fail, then the Third line of defense is activated. B and T lymphocytes undergo a selective
Immunology, Immune Response, and Immunological Testing Lines of Defense If the First and Second lines of defense fail, then the Third line of defense is activated. B and T lymphocytes undergo a selective
The Mycobacterium tuberculosis MmpL11 cell wall lipid transporter is important for
 Supplemental materials The Mycobacterium tuberculosis MmpL11 cell wall lipid transporter is important for biofilm formation, intracellular growth and non-replicating persistence Catherine C. Wright, 1
Supplemental materials The Mycobacterium tuberculosis MmpL11 cell wall lipid transporter is important for biofilm formation, intracellular growth and non-replicating persistence Catherine C. Wright, 1
EFFECT OF AN IMMUNOMODULATING DIET ON THE IMMUNE SYSTEM OF DOGS INFECTED WITH Leishmania infantum
 EFFECT OF AN IMMUNOMODULATING DIET ON THE IMMUNE SYSTEM OF DOGS INFECTED WITH Leishmania infantum Summary The experiment conducted on the effects of the nutraceutical food Immuno Active on dogs suffering
EFFECT OF AN IMMUNOMODULATING DIET ON THE IMMUNE SYSTEM OF DOGS INFECTED WITH Leishmania infantum Summary The experiment conducted on the effects of the nutraceutical food Immuno Active on dogs suffering
CHAPTER 4 IMMUNOLOGICAL TECHNIQUES
 CHAPTER 4 IMMUNOLOGICAL TECHNIQUES Nitroblue Tetrazolium Chloride (NBT) Reduction test NBT reduction test was evaluated by employing the method described by Hudson and Hay,1989 based upon principle that
CHAPTER 4 IMMUNOLOGICAL TECHNIQUES Nitroblue Tetrazolium Chloride (NBT) Reduction test NBT reduction test was evaluated by employing the method described by Hudson and Hay,1989 based upon principle that
SUPPLEMENTARY INFORMATION
 Complete but curtailed T-cell response to very-low-affinity antigen Dietmar Zehn, Sarah Y. Lee & Michael J. Bevan Supp. Fig. 1: TCR chain usage among endogenous K b /Ova reactive T cells. C57BL/6 mice
Complete but curtailed T-cell response to very-low-affinity antigen Dietmar Zehn, Sarah Y. Lee & Michael J. Bevan Supp. Fig. 1: TCR chain usage among endogenous K b /Ova reactive T cells. C57BL/6 mice
Animal models for infectious diseases caused by parasites: Leishmaniasis
 Drug Discovery Today: Disease Models Vol. 1, No. 1 2004 DRUG DISCOVERY TODAY DISEASE MODELS Editors-in-Chief Jan Tornell AstraZeneca, Sweden Denis Noble University of Oxford, UK Infectious diseases Animal
Drug Discovery Today: Disease Models Vol. 1, No. 1 2004 DRUG DISCOVERY TODAY DISEASE MODELS Editors-in-Chief Jan Tornell AstraZeneca, Sweden Denis Noble University of Oxford, UK Infectious diseases Animal
By:Reham Alahmadi NOV The production of antibodies and vaccination technology
 By:Reham Alahmadi NOV 2018 The production of antibodies and vaccination technology Antibody Production The blood contains two types of white blood cell or leukocyte Phagocytes ingest bacteria by endocytosis
By:Reham Alahmadi NOV 2018 The production of antibodies and vaccination technology Antibody Production The blood contains two types of white blood cell or leukocyte Phagocytes ingest bacteria by endocytosis
Lymphoid tissue. 1. Central Lymphoid tissue. - The central lymphoid tissue (also known as primary) is composed of bone morrow and thymus.
 1. Central Lymphoid tissue Lymphoid tissue - The central lymphoid tissue (also known as primary) is composed of bone morrow and thymus. Bone Morrow - The major site of hematopoiesis in humans. - Hematopoiesis
1. Central Lymphoid tissue Lymphoid tissue - The central lymphoid tissue (also known as primary) is composed of bone morrow and thymus. Bone Morrow - The major site of hematopoiesis in humans. - Hematopoiesis
Chapter 3, Part A (Pages 37-45): Leukocyte Migration into Tissues
 Allergy and Immunology Review Corner: Chapter 3, Part A (pages 37-45) of Cellular and Molecular Immunology (Seventh Edition), by Abul K. Abbas, Andrew H. Lichtman and Shiv Pillai. Chapter 3, Part A (Pages
Allergy and Immunology Review Corner: Chapter 3, Part A (pages 37-45) of Cellular and Molecular Immunology (Seventh Edition), by Abul K. Abbas, Andrew H. Lichtman and Shiv Pillai. Chapter 3, Part A (Pages
Prof. Ibtesam Kamel Afifi Professor of Medical Microbiology & Immunology
 By Prof. Ibtesam Kamel Afifi Professor of Medical Microbiology & Immunology Lecture objectives: At the end of the lecture you should be able to: Enumerate features that characterize acquired immune response
By Prof. Ibtesam Kamel Afifi Professor of Medical Microbiology & Immunology Lecture objectives: At the end of the lecture you should be able to: Enumerate features that characterize acquired immune response
SPECIFIC ANTIINFECTIOUS IMMUNITY. colostral immunity. administration of antibodies VIRULENT VACCINE INACTIVATED VACCINE
 INDUCTION OF IMMUNITY AGAINST INFECTION SPECIFIC ANTIINFECTIOUS IMMUNITY active passive S AND VACCINATION infection vaccination colostral immunity administration of antibodies VIRULENT MODIFIED LIVE INACTIVATED
INDUCTION OF IMMUNITY AGAINST INFECTION SPECIFIC ANTIINFECTIOUS IMMUNITY active passive S AND VACCINATION infection vaccination colostral immunity administration of antibodies VIRULENT MODIFIED LIVE INACTIVATED
Blocking Interhost Transmission of Influenza Virus by Vaccination in the Guinea Pig Model
 JOURNAL OF VIROLOGY, Apr. 2009, p. 2803 2818 Vol. 83, No. 7 0022-538X/09/$08.00 0 doi:10.1128/jvi.02424-08 Copyright 2009, American Society for Microbiology. All Rights Reserved. Blocking Interhost Transmission
JOURNAL OF VIROLOGY, Apr. 2009, p. 2803 2818 Vol. 83, No. 7 0022-538X/09/$08.00 0 doi:10.1128/jvi.02424-08 Copyright 2009, American Society for Microbiology. All Rights Reserved. Blocking Interhost Transmission
Priming by Microbial Antigens from the. T Cells to Rapidly Secrete IL-4 in. CD4 + Flora Determines the Ability of. major
 This information is current as of April 16, 2018. References Subscription Permissions Email Alerts Priming by Microbial Antigens from the Intestinal CD4 + Flora Determines the Ability of T Cells to Rapidly
This information is current as of April 16, 2018. References Subscription Permissions Email Alerts Priming by Microbial Antigens from the Intestinal CD4 + Flora Determines the Ability of T Cells to Rapidly
FOR OPTIMAL GUT HEALTH KEMIN.COM/GUTHEALTH
 FOR OPTIMAL GUT HEALTH KEMIN.COM/GUTHEALTH ALETA A SOURCE OF 1,3-BETA GLUCANS Aleta is highly bioavailable, offering a concentration greater than 5% of 1,3-beta glucans. Aleta provides a consistent response
FOR OPTIMAL GUT HEALTH KEMIN.COM/GUTHEALTH ALETA A SOURCE OF 1,3-BETA GLUCANS Aleta is highly bioavailable, offering a concentration greater than 5% of 1,3-beta glucans. Aleta provides a consistent response
Mouse Anti-OVA IgM Antibody Assay Kit
 Mouse Anti-OVA IgM Antibody Assay Kit Catalog # 3017 For Research Use Only - Not Human or Therapeutic Use INTRODUCTION Ovalbumin (OVA) is a widely used antigen for inducing allergic reactions in experimental
Mouse Anti-OVA IgM Antibody Assay Kit Catalog # 3017 For Research Use Only - Not Human or Therapeutic Use INTRODUCTION Ovalbumin (OVA) is a widely used antigen for inducing allergic reactions in experimental
General information. Cell mediated immunity. 455 LSA, Tuesday 11 to noon. Anytime after class.
 General information Cell mediated immunity 455 LSA, Tuesday 11 to noon Anytime after class T-cell precursors Thymus Naive T-cells (CD8 or CD4) email: lcoscoy@berkeley.edu edu Use MCB150 as subject line
General information Cell mediated immunity 455 LSA, Tuesday 11 to noon Anytime after class T-cell precursors Thymus Naive T-cells (CD8 or CD4) email: lcoscoy@berkeley.edu edu Use MCB150 as subject line
Page 1 of 2. Standard curve of Human IFN gamma ELISA Ready- SET-Go! Product Information Contents: Human IFN gamma ELISA Ready- SET-Go!
 Page 1 of 2 Human IFN gamma ELISA Ready-SET-Go! Catalog Number: 88-7316 Also known as: Interferon-gamma, IFN-g, IFNg RUO: For. Not for use in diagnostic procedures. Standard curve of Human IFN gamma ELISA
Page 1 of 2 Human IFN gamma ELISA Ready-SET-Go! Catalog Number: 88-7316 Also known as: Interferon-gamma, IFN-g, IFNg RUO: For. Not for use in diagnostic procedures. Standard curve of Human IFN gamma ELISA
COURSE: Medical Microbiology, PAMB 650/720 - Fall 2008 Lecture 16
 COURSE: Medical Microbiology, PAMB 650/720 - Fall 2008 Lecture 16 Tumor Immunology M. Nagarkatti Teaching Objectives: Introduction to Cancer Immunology Know the antigens expressed by cancer cells Understand
COURSE: Medical Microbiology, PAMB 650/720 - Fall 2008 Lecture 16 Tumor Immunology M. Nagarkatti Teaching Objectives: Introduction to Cancer Immunology Know the antigens expressed by cancer cells Understand
9/10/2018. Principles of Vaccination. Immunity. Antigen. September 2018
 Centers for Disease Control and Prevention National Center for Immunization and Respiratory Diseases Principles of Vaccination September 2018 Chapter 1 September 2018 Photographs and images included in
Centers for Disease Control and Prevention National Center for Immunization and Respiratory Diseases Principles of Vaccination September 2018 Chapter 1 September 2018 Photographs and images included in
Cell isolation. Spleen and lymph nodes (axillary, inguinal) were removed from mice
 Supplementary Methods: Cell isolation. Spleen and lymph nodes (axillary, inguinal) were removed from mice and gently meshed in DMEM containing 10% FBS to prepare for single cell suspensions. CD4 + CD25
Supplementary Methods: Cell isolation. Spleen and lymph nodes (axillary, inguinal) were removed from mice and gently meshed in DMEM containing 10% FBS to prepare for single cell suspensions. CD4 + CD25
What is the immune system? Types of Immunity. Pasteur and rabies vaccine. Historical Role of smallpox. Recognition Response
 Recognition Response Effector memory What is the immune system? Types of Immunity Innate Adaptive Anergy: : no response Harmful response: Autoimmunity Historical Role of smallpox Pasteur and rabies vaccine
Recognition Response Effector memory What is the immune system? Types of Immunity Innate Adaptive Anergy: : no response Harmful response: Autoimmunity Historical Role of smallpox Pasteur and rabies vaccine
The Adaptive Immune Response: T lymphocytes and Their Functional Types *
 OpenStax-CNX module: m46560 1 The Adaptive Immune Response: T lymphocytes and Their Functional Types * OpenStax This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution
OpenStax-CNX module: m46560 1 The Adaptive Immune Response: T lymphocytes and Their Functional Types * OpenStax This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution
Kit for assay of thioredoxin
 FkTRX-02-V2 Kit for assay of thioredoxin The thioredoxin system is the major protein disulfide reductase in cells and comprises thioredoxin, thioredoxin reductase and NADPH (1). Thioredoxin systems are
FkTRX-02-V2 Kit for assay of thioredoxin The thioredoxin system is the major protein disulfide reductase in cells and comprises thioredoxin, thioredoxin reductase and NADPH (1). Thioredoxin systems are
Review Cutaneous Manifestations of Human and Murine Leishmaniasis
 Review Cutaneous Manifestations of Human and Murine Leishmaniasis Breanna M. Scorza 1, Edgar M. Carvalho 2,3 and Mary E. Wilson 1,4,5,6, * 1 Interdisciplinary Graduate Program in Immunology, University
Review Cutaneous Manifestations of Human and Murine Leishmaniasis Breanna M. Scorza 1, Edgar M. Carvalho 2,3 and Mary E. Wilson 1,4,5,6, * 1 Interdisciplinary Graduate Program in Immunology, University
Early History of Vaccination Vaccines
 6/23/13 Early History of Vaccination Vaccines The tradition of vaccination may have originated in India in AD 1000 Pioneered India and China in the 17th Powdered scabs from people infected with smallpox
6/23/13 Early History of Vaccination Vaccines The tradition of vaccination may have originated in India in AD 1000 Pioneered India and China in the 17th Powdered scabs from people infected with smallpox
CELL MEDIATED IMMUNE RESPONSE
 CELL MEDIATED IMMUNE RESPONSE Chapter IV - CELL MEDIATED IMMUNE RESPONSE Sujatha, M. 2013. Evaluation of Immunological changes in Fish, Catla catla administered with bacterial pathogen, Aeromonas hydrophila,
CELL MEDIATED IMMUNE RESPONSE Chapter IV - CELL MEDIATED IMMUNE RESPONSE Sujatha, M. 2013. Evaluation of Immunological changes in Fish, Catla catla administered with bacterial pathogen, Aeromonas hydrophila,
Clinical Trials of Pandemic Vaccines: Key Issues. John Treanor University of Rochester Rochester, NY
 Clinical Trials of Pandemic Vaccines: Key Issues John Treanor University of Rochester Rochester, NY Inactivated vaccine approach Proven technology Used successfully in 1957 and 1968 Abundant efficacy data
Clinical Trials of Pandemic Vaccines: Key Issues John Treanor University of Rochester Rochester, NY Inactivated vaccine approach Proven technology Used successfully in 1957 and 1968 Abundant efficacy data
Memory CD4 T Cells Enhance Primary CD8 T-Cell Responses
 INFECTION AND IMMUNITY, July 2007, p. 3556 3560 Vol. 75, No. 7 0019-9567/07/$08.00 0 doi:10.1128/iai.00086-07 Copyright 2007, American Society for Microbiology. All Rights Reserved. Memory CD4 T Cells
INFECTION AND IMMUNITY, July 2007, p. 3556 3560 Vol. 75, No. 7 0019-9567/07/$08.00 0 doi:10.1128/iai.00086-07 Copyright 2007, American Society for Microbiology. All Rights Reserved. Memory CD4 T Cells
LYMPHOCYTES & IMMUNOGLOBULINS. Dr Mere Kende, Lecturer SMHS
 LYMPHOCYTES & IMMUNOGLOBULINS Dr Mere Kende, Lecturer SMHS Immunity Immune- protection against dangers of non-self/invader eg organism 3 components of immune system 1 st line: skin/mucosa/cilia/hair/saliva/fatty
LYMPHOCYTES & IMMUNOGLOBULINS Dr Mere Kende, Lecturer SMHS Immunity Immune- protection against dangers of non-self/invader eg organism 3 components of immune system 1 st line: skin/mucosa/cilia/hair/saliva/fatty
Nori Rabbit IL-2 ELISA Kit DataSheet
 Nori Rabbit IL-2 ELISA Kit DataSheet IL-2 is an interleukin, a type of cytokine immune system signaling molecule. IL-2 is a T cell stimulatory cytokine best known for inducing T cell proliferation, the
Nori Rabbit IL-2 ELISA Kit DataSheet IL-2 is an interleukin, a type of cytokine immune system signaling molecule. IL-2 is a T cell stimulatory cytokine best known for inducing T cell proliferation, the
Hua Tang, Weiping Cao, Sudhir Pai Kasturi, Rajesh Ravindran, Helder I Nakaya, Kousik
 SUPPLEMENTARY FIGURES 1-19 T H 2 response to cysteine-proteases requires dendritic cell-basophil cooperation via ROS mediated signaling Hua Tang, Weiping Cao, Sudhir Pai Kasturi, Rajesh Ravindran, Helder
SUPPLEMENTARY FIGURES 1-19 T H 2 response to cysteine-proteases requires dendritic cell-basophil cooperation via ROS mediated signaling Hua Tang, Weiping Cao, Sudhir Pai Kasturi, Rajesh Ravindran, Helder
Supplementary Figure 1. Characterization of basophils after reconstitution of SCID mice
 Supplementary figure legends Supplementary Figure 1. Characterization of after reconstitution of SCID mice with CD4 + CD62L + T cells. (A-C) SCID mice (n = 6 / group) were reconstituted with 2 x 1 6 CD4
Supplementary figure legends Supplementary Figure 1. Characterization of after reconstitution of SCID mice with CD4 + CD62L + T cells. (A-C) SCID mice (n = 6 / group) were reconstituted with 2 x 1 6 CD4
The Lymphatic System and Immunity. Chapters 20 & 21
 The Lymphatic System and Immunity Chapters 20 & 21 Objectives 1. SC.912.L.14.52 - Explain the basic functions of the human immune system, including specific and nonspecific immune response, vaccines, and
The Lymphatic System and Immunity Chapters 20 & 21 Objectives 1. SC.912.L.14.52 - Explain the basic functions of the human immune system, including specific and nonspecific immune response, vaccines, and
Oncolytic Immunotherapy: A Local and Systemic Antitumor Approach
 Oncolytic Immunotherapy: A Local and Systemic Antitumor Approach Oncolytic immunotherapy Oncolytic immunotherapy the use of a genetically modified virus to attack tumors and induce a systemic immune response
Oncolytic Immunotherapy: A Local and Systemic Antitumor Approach Oncolytic immunotherapy Oncolytic immunotherapy the use of a genetically modified virus to attack tumors and induce a systemic immune response
(From the Hospital of The Rockefeller Institute far Medical Research) (Received for publication, May 13, 1936)
 Published Online: 1 October, 1936 Supp Info: http://doi.org/10.1084/jem.64.4.553 Downloaded from jem.rupress.org on October 30, 2018 A SKIN TEST FOR DETECTING GROUP C HEMOLYTIC STREPTOCOCCAL INFECTION
Published Online: 1 October, 1936 Supp Info: http://doi.org/10.1084/jem.64.4.553 Downloaded from jem.rupress.org on October 30, 2018 A SKIN TEST FOR DETECTING GROUP C HEMOLYTIC STREPTOCOCCAL INFECTION
Atypical cutaneous leishmaniasis cases display elevated antigen-induced interleukin-10
 Parasite Immunology, 2007, 29, 277 282 DOI: 10.1111/j.1365-3024.2007.00944.x Blackwell ORIGINAL Immune response Publishing ARTICLE in atypical Ltd cutaneous leishmaniasis Atypical cutaneous leishmaniasis
Parasite Immunology, 2007, 29, 277 282 DOI: 10.1111/j.1365-3024.2007.00944.x Blackwell ORIGINAL Immune response Publishing ARTICLE in atypical Ltd cutaneous leishmaniasis Atypical cutaneous leishmaniasis
DENDRITIC CELLS ACTIVATED BY CpG MOTIFS ARE POTENT INDUCERS OF A TH1 IMMUNE RESPONSE THAT PROTECTS MICE AGAINST LEISHMANIASIS
 DENDRITIC CELLS ACTIVATED BY CpG MOTIFS ARE POTENT INDUCERS OF A TH1 IMMUNE RESPONSE THAT PROTECTS MICE AGAINST LEISHMANIASIS Dissertation zur Erlangung des naturwissenschaftlichen Doktorgrades der Bayerischen
DENDRITIC CELLS ACTIVATED BY CpG MOTIFS ARE POTENT INDUCERS OF A TH1 IMMUNE RESPONSE THAT PROTECTS MICE AGAINST LEISHMANIASIS Dissertation zur Erlangung des naturwissenschaftlichen Doktorgrades der Bayerischen
Post Kala-azar Dermal Leishmaniasis (PKDL) from the field to the cellular and the subcellular levels
 Post Kala-azar Dermal Leishmaniasis (PKDL) from the field to the cellular and the subcellular levels A M EL Hassan Institute of Endemic Diseases University of Khartoum Introduction PKDL is a VL related
Post Kala-azar Dermal Leishmaniasis (PKDL) from the field to the cellular and the subcellular levels A M EL Hassan Institute of Endemic Diseases University of Khartoum Introduction PKDL is a VL related
Shigella Pathogenesis and Vaccine Development
 Shigella Pathogenesis and Vaccine Development Ryan Ranallo, Ph.D. Department of Enteric Infections Division of Communicable Diseases and Immunology Walter Reed Army Institute of Research Causes of Travelers
Shigella Pathogenesis and Vaccine Development Ryan Ranallo, Ph.D. Department of Enteric Infections Division of Communicable Diseases and Immunology Walter Reed Army Institute of Research Causes of Travelers
Examples of questions for Cellular Immunology/Cellular Biology and Immunology
 Examples of questions for Cellular Immunology/Cellular Biology and Immunology Each student gets a set of 6 questions, so that each set contains different types of questions and that the set of questions
Examples of questions for Cellular Immunology/Cellular Biology and Immunology Each student gets a set of 6 questions, so that each set contains different types of questions and that the set of questions
