Systemic Agents in Psoriasis. Dr SushilTahiliani
|
|
|
- Gabriella Randall
- 5 years ago
- Views:
Transcription
1 Agents in Psoriasis Dr SushilTahiliani
2 Introduction Pso is a systemic disease 30% pts need systemic drugs* Many pts feel: Dissatisfied with treatment Feel under-treated Desire more complete control of disease 2 LiemWH,Mccullough JL,Weinstein GD.Effectiveness of topical therapy for psoriasis:results of a national survey.cutis1995;95:306-10
3 Indications Severe plaque disease(bsa>10 or PASI>10) Gen pustular PSO Erythroderma Significant PSA Cases refractory to topicals/phototherapy Limited disease with significant physical /psychosocial disability 3 *Kanwar AJ,Yadav S,Dogra S.Psoriasis:What s new in non biologic systemic therapy in era of biologics?.indian J DermatolVeneorol Leprol 2010;76:
4 Conventional systemic agents: Advantages Ease of administration Lower cost Vast experience in use with longer term follow-up 4
5 agents Commonly used Methotrexate(MTX) Cyclosporin(CsA) Acitretin Uncommonly used Fumaric Acid esters Hydroxyurea Azathioprine Leflunomide Mycophenolate mofetil 5
6 MTX Gold standard systemic drug First used in PSO > 50yrs ago Approved by FDA in1972 for PSO and in late 80s for RA Monotherapy / as one of molecules in combination/ sequential therapy Clinical experience >> efficacy thro clinical studies Available as tab &Injection for SC/IM/IV use 8
7 Controversies & confusions Criteria for selection of patients Method of lab evaluation Need for liver biopsies in surveillance 9
8 MTX:MOA Inhibits dihydrofolate reductase within 1 hour Also inhibits thymidylate synthetase less rapidly Decreased synthesis of tetrahydrofolate needed for DNA & RNA synth Works on S phase of cell cycle Significant inhibition of lymphoid cell proliferation & CLA & e-selectin Little or no effect on proliferation of epidermal cells Effect on lymphoid cells 1000 times more 10
9 MTX:MOA-New Concepts Inhibits AICAR transformylase Accumulation of AICAR* tissue accumulation of Adenosine Anti-inflammatory action *5-aminoimidazole-4- carbonucleotide riboxamide 12
10 MTX- dosing Test dose: +/ mg/kg/wk Oral vs. IM vs. SC Build up vs. fulldose initially Folic acid supplementation 14
11 Subcutaneous Methotrexate Higher absorption rates & bioavailabilit y Fewer adverse events Higher intracellular level of Mtx-PG Increased efficacy of Subcutaneous Methotrexate 15
12 MTX has been the standard systemic therapy for psoriasis for more than five decades and still continues to be gold standard in era of biologics. Single weekly oral dose of MTX has a favourable side-effect profile compared to intermittent oral schedule Dose of mg/kg/week (25-30 mg), continue till PASI 75 is achieved, then gradually taper it. Good and rapid control of disease in majority of the patients. 16 Concomitant administration of folic acid (1 to 5 mg/day) reduces side effects
13 Methotrexate 25mg is an effective dose as monotherapy for the treatment of severe psoriasis, whereas the 10mg dose is slow to act and less effective, but has a less severe sideeffect profile. 17
14 Monitoring Baseline: History & clinical examn CBC, Platelet count BUN,Sr Creatinine,LFT Pregnancy test* Test for HIV* PPD,X-ray chest* Liver biopsy* 18 *in select cases
15 Monitoring Follow up: 1. CBC,Platelet count, LFT(espTranaminases): 5-6 days after test dose 1-2 wks after each dose escalation Every 3 mths long term(while on treatment) 2. Renal function tests: Twice yearly 19
16 DIs Drugs that increase MTX levels Drugs that increase hematologic toxicity Drugs that may increase hepatotoxicity Salicylates NSAID Sulfonamides Dipyrimadole Phenothiazines Chloramphenicol, Tetracyclines Phenytoin Probenecid Sulfonamides, DDS Trimethoprim Retinoids Alcohol 20
17 Contra-indications Absolute Pregnancy Lactation Alcoholic liver disease Other chr liver diseases Immunodeficiency syndr Bone marrow hypoplasia Hypersensitivity to MTX Relative Abnormal LFT, RFT Active infection Obesity DM Concomitant use of hepatotoxic drug Recent administration of live vaccine Unreliable pt 21 SIG Menter Psoriasis A,Korman N,Elmets C,Feldman S et al. Section 4. Guidelines of care for the management and treatment of psoriasis with traditional systemic agents. J Am Acad Dermatol 2009;61:451-85
18 Indications for liver biopsy Low risk group No baseline biopsy Done after cumulative dose of 3.5-4g Earlier if >5 SGPT values (out of 9 tests done in 12 mths ) elevated If Sr albumin values fall to less than normal High risk group After 2-6 mths of initiation of therapy Repeated after every additional g of MTX 22
19 PIIINP Measures fibrogenic activity in liver If levels remain stable, liver biopsy can be avoided Not organ specific Measures ongoing fibrogenic activity only 23
20 Pso pts on MTX: Diagnostic accuracy of non-invasive markers of liver fibrosis Maybury CM et al British Journal of Dermatology (2014) 170, pp Systematic Review 24
21 MTX induced liver fibrosis MTX is the primary systemic drug for Pso It is also effective in recalcitrant LP, AD etc. There is a risk of liver fibrosis in pts on MTX Liver biopsy is a reference standard for diagnosis Evaluation of non-invasive methods of diagnosis is done (17 studies included) 25
22 Comparison Investigation Sensitivity Specificity LFT 38% 83% P3NP 74% 77% Fibroscan 60% 80% USG 55% 49% Clinical utility limited Low quality data Small sample size If done in isolation, significant number of cases may remain unidentified 26
23 ADR Minor Nausea Anorexia Stomatitis Fatigue Major Myelosuppression Hepatotoxicity Pulmonary fibrosis Reactivation of TB Lymphoma 27
24 Mechanisms of Methotrexate Resistance Di-hydrofolate reductase gene polymorphism Impaired transport of MTX into cells Decreased ability to synthesize MTX polyglutamate Increased expression of drug efflux transporters of the multidrug resistance protein class 28
25 29
26 agents Cyclosporine 30
27 Cyclosporine Syn.-CyclosporinA,Ciclosporin,CsA History: Isolated from soil fungus Tolypocladium inflatum gams in 1970 Also isolated from Cyclindocarpon lucidum Identified as immunosuppressive in discovered to be effective in psoriasis 31
28 Formulation Soft gelatin cap.-25,50,100 mg. Oral soln.-100 mg/ml Inj.-5 ml vial ( 50 mg/ml ) Drug is dissolved in ethanol & olive oil. It forms a microemulsion in contact with aqueous fluids. Oral soln. should be diluted with orange or apple juice just before intake Dose: 3-5mg/kg initial 2-4mg/kg maintenance 32
29 Mechanism of action Ag on APCàT cellsà transcription of IL2à mitogenesis of Ag specific helper Tcellsàsecretion of cytokinesàactivation of cellular & immune response CsA binds to cyclophilins. This complex binds to & inactivates calcineurin. This leads to blockage of signal transduction of events leading to IL2 gene transcription 33
30 Mechanism of Action of Cyclosporine 34
31 CsA in psoriasis 4 regimens: 1)Crisis intervention 2)Short intermittent(as a bridge to other therapies) 3)Six month block 4)Continuous long-term(in unresponsive cases)- should not be used for more than 2 yrs 35
32 CsA ADR- Renal Hypertension GI Neurologic Metabolic Hematologic Neoplastic Musculoskeletal Mucocutaneous 36
33 CsA-C.I. Renal insufficiency Uncontrolled HTN Hypersensitivity to CsA h/o systemic malignancy Prior treatment with PUVA Uncontrolled infections 37
34 CsA-usage guidelines Benefit-to-risk analysis: efficacy,toxicity, economic effect, acceptability,alternative options Thorough history & clinical examn Lab.:Sr creatinine,bun,urinalysis CBC,LFT Lipid profile Sr magnesium, K, uric acid 38
35 CsA Advantages Selective action on T helper cells Rapid therapeutic action Weak myelotoxicity Safety in pregnancy (category C) Drawbacks Nephrotoxicity Risk of hypertension High rates of relapse High cost Not very effective in PSA 39
36 agents Acitretin 40
37 Historical aspects 1972-Bollag discovered Acitretin,Etretinate 1986-Etretinate introduced for treating Pso 1988-Etretinate replaced by Acitretin 2005-Acitretin introduced in India 41
38 Mechanism of Action Decreases the thickness of stratum corneum and the inflammation Promotes epidermal accumulation of mucus-like material Inhibition of neutrophil migration from dermal capillaries Reduction in epidermal concentrations of polyamines which regulate cell growth, proliferation, and differentiation. 42
39 MOA Affects pathways involved in- Inflammation Cellular differentiation Apoptosis Sebaceous gland activity Broad effect on multiple tissues apart from skin 43
40 Mechanism of Action of Retinoid(Acitretin) Normalization of abnormal keratinocyte differentiation Reduction in keratinocyte proliferation Reduction in inflammation 44
41 Retinoids: Channeling to Cell Nucleus Retinoid Epidermal Cell Nucleus CRABP-II RARα,β,g Retinoid CRABP-II 45
42 Dose mg / kg body weight Usually mg / day Maintenance dose 10 mg / day or 25mg on alternate day 0.3 mg / kg in combination therapy 46
43 Indications Pustular psoriasis- gen./ local. Erythrodermic psoriasis Moderate to severe plaque psoriasis In immunosuppressed patients 47
44 Use in chr. plaque psoriasis Combination therapy Sequential therapy Rotational therapy Monotherapy 48
45 Dose-ranging studies - Acitretin Author (Double blind rand. studies) Number of patients(n), disease type Study design Results Lassus et al (placebo controlled) n = 80 Plaque (70), pustular (4), erythrodermic(6) 10,25 or 50 mg /d x 8 weeks Average in PASI 61%, 79%, & 86% in 10, 25 & 50 mg group respectively, side effects >50 mg/d Goldfarb et al (placebo controlled) Gollnick et al (multicentric) 38 10,25,50 or 75 mg/d x 8 weeks followed by open label phase with 50 mg/d x 16 weeks ,25 or 50 mg /d of acitretin 8 weeks 51-75% in PASI score. side effects at higher doses(>50 mg/d) Average in PASI 46% (10 mg/d), 43% (25 mg/d), & 49% (50 mg/d)
46 Acitretin 35 mg/day was observed to be more efficacious compared to 25 mg/day and 50 mg/day dosing, whereas its safety profile is better than 50 mg/day dosing in the management of severe plaque type psoriasis in adult patients.
47 Contraindications Pregnancy-category X drug Severely impaired renal/ liver function Significant chr. abnormality of lipid metabolism Hypersensitivity to retinoids Nursing mothers & children less than 12yr 51
48 Baseline: CBC LFT Lipid profile Pregnancy test Lab. evaluations 52 To be repeated monthly x 2mths & if normal, less often thereafter
49 ADR 53 Teratogenicity Mucocutaneous toxicity Myalgia, arthralgia Hyperlipidemia Hepatotoxicity Skeletal Pseudotumour cerebri Drug interactions Ophthalmologic Psychiatric effects
50 Limitations Slow & unsatisfactory effect as monotherapy in commonest types of PSO Risk of teratogenicity Restrictions for use in children Restrictions for use in men wanting to father a child Long duration of precautions Repeated investigations 54
51 Expert opinion Combination therapy with acitretin will become increasingly important in management of recalcitrant psoriasis Only FDA approved systemic therapy that is not immunosuppressant With/without phototherapy, it is a very important treatment option for pts with chronic infections 55
52 56
53 Combination therapy Accelerator Maintainer Cyclosporine Acitretin Acitretin PUVA UVB Acitretin 59
54 Sequential therapy Involves use of agents in a deliberate sequence to maximize rate of initial improvement & minimize long term toxicity to improve overall outcome 60
55 Second tier systemic agents 65
56 Azathioprine Purine analogue Slow onset of action(6-8wks for significant results) Level of evidence for efficacy weaker May be cosidered in cases of Pso + autoimmune bullous diseases TPMT levels should be done to know risk of myelosuppression 66
57 Fumaric acid esters Used commonly in northern Europe Combination of dimethyl fumarate & monoethyl fumarate salts MOA:InhibitT cells & shift from Th1 to Th2 PASI improvement between 50 & 80% after wks ADR:GI symptoms, flushing, lymphopenia 67
58 Hydroxyurea Synthesized by Dressler & Stein(1869) May work by itself or through conversion to metabolites like acetohydroxamic acid Impairs DNA synthesis though inhibition of ribonucleotide diphosphate reductase Dose:1-1.5 g / day Acts slowly & is preferred for use in combination or as maintenance therapy ADR: Myelosuppression, cutaneous effects 68
59 Int J Dermatol 2001; 40: Hydroxyurea gm/day 75 % patients - about 35% PASI score 8 weeks 50 % had > 70% PASI score All adverse effects were mild and reversible Effective, cheap, safe but relatively slower acting Combination Acitretin
60 Mycophenolate mofetil Prodrug of mycophenolic acid Works by inhibiting proliferation of both B & T cells Specially useful in cases with Pso + immunobullous diseases & in organ transplant recipients Common side effects include GI effects, leucopenia, GU effects 70
61 Leflunomide DMARD agent that inhibits pyrimidine synthesis Approved for use in RA May be used in Pso + PSA More effective on arthritis signs & symptoms ADR: GI, increase liver enzymes, cutaneous, bone marrow, teratogenicity 71
62 Thank you 72
Combination Nonbiologic Therapy in Psoriasis. Sushil Tahiliani, MBBS, MD
 Combination Nonbiologic Therapy in Psoriasis Sushil Tahiliani, MBBS, MD Agenda Rationale Preferred and less preferred combination Morphology-specific preferred combinations Doses used in combinations Potential
Combination Nonbiologic Therapy in Psoriasis Sushil Tahiliani, MBBS, MD Agenda Rationale Preferred and less preferred combination Morphology-specific preferred combinations Doses used in combinations Potential
Update on systemic therapies and emerging treatments How do I choose a systemic agent?
 Update on systemic therapies and emerging treatments How do I choose a systemic agent? Amy S. Paller, M.D. Walter J. Hamlin Professor and Chair of Dermatology Professor of Pediatrics Northwestern University
Update on systemic therapies and emerging treatments How do I choose a systemic agent? Amy S. Paller, M.D. Walter J. Hamlin Professor and Chair of Dermatology Professor of Pediatrics Northwestern University
A systematic review of treatments for severe psoriasis Griffiths C E, Clark C M, Chalmers R J, Li Wan Po A, Williams H C
 A systematic review of treatments for severe psoriasis Griffiths C E, Clark C M, Chalmers R J, Li Wan Po A, Williams H C Authors' objectives To compare the effectiveness of currently available treatments
A systematic review of treatments for severe psoriasis Griffiths C E, Clark C M, Chalmers R J, Li Wan Po A, Williams H C Authors' objectives To compare the effectiveness of currently available treatments
Carolyn Bangert, MD Associated Dermatologists, PC
 Carolyn Bangert, MD Associated Dermatologists, PC Carolyn Bangert, MD Systemic Medications for the Dermatology Toolbox: Cyclosporine I have no relevant conflicts of interest with any companies. I will
Carolyn Bangert, MD Associated Dermatologists, PC Carolyn Bangert, MD Systemic Medications for the Dermatology Toolbox: Cyclosporine I have no relevant conflicts of interest with any companies. I will
Elements of Successful PBS Applications. Barbara Radulski RN. Copyright
 Elements of Successful PBS Applications Barbara Radulski RN PBS Requirements April 1 2006 THE RULES PBS Requirements 18 years and over Psoriasis x 6 months Failed to achieve an adequate response to 3 systemic
Elements of Successful PBS Applications Barbara Radulski RN PBS Requirements April 1 2006 THE RULES PBS Requirements 18 years and over Psoriasis x 6 months Failed to achieve an adequate response to 3 systemic
Psoriasis: Therapeutic goals
 Psoriasis: Therapeutic goals I want to die 50 45 impetiginization infliximab 600 40 35 30 400 25 20 15 200 10 5 0 22-ene 21-feb 23-mar 22-abr 22- may Efalizumab 6 doses: flare + REBOUND CSA 3 21-jun 21-jul
Psoriasis: Therapeutic goals I want to die 50 45 impetiginization infliximab 600 40 35 30 400 25 20 15 200 10 5 0 22-ene 21-feb 23-mar 22-abr 22- may Efalizumab 6 doses: flare + REBOUND CSA 3 21-jun 21-jul
Psoriasis management. A/Prof Amanda Oakley Dermatologist, Waikato
 Psoriasis management A/Prof Amanda Oakley Dermatologist, Waikato AbbVie Breakfast Session, 14 June 2014 Disclosure This breakfast session is sponsored by Abbvie Autoimmune skin disorders Psoriasis Eczema
Psoriasis management A/Prof Amanda Oakley Dermatologist, Waikato AbbVie Breakfast Session, 14 June 2014 Disclosure This breakfast session is sponsored by Abbvie Autoimmune skin disorders Psoriasis Eczema
Kevzara (sarilumab) NEW PRODUCT SLIDESHOW
 Kevzara (sarilumab) NEW PRODUCT SLIDESHOW Introduction Brand name: Kevzara Generic name: Sarilumab Pharmacological class: Interleukin-6 antagonist Strength and Formulation: 150mg/1.14mL, 200mg/1.14mL;
Kevzara (sarilumab) NEW PRODUCT SLIDESHOW Introduction Brand name: Kevzara Generic name: Sarilumab Pharmacological class: Interleukin-6 antagonist Strength and Formulation: 150mg/1.14mL, 200mg/1.14mL;
Efficacy of Concomitant Use of PUVA and Methotrexate in Disease Clearance Time in Plaque Type Psoriasis
 Efficacy of Concomitant Use of PUVA and Methotrexate in Disease Clearance Time in Plaque Type Psoriasis T. Shehzad ( Departments of Dermatology Naval Hospital PNS Shifa, Karachi. ) N. R. Dar ( Departments
Efficacy of Concomitant Use of PUVA and Methotrexate in Disease Clearance Time in Plaque Type Psoriasis T. Shehzad ( Departments of Dermatology Naval Hospital PNS Shifa, Karachi. ) N. R. Dar ( Departments
INITIATING ORAL AUBAGIO (teriflunomide) THERAPY
 FOR YOUR PATIENTS WITH RELAPSING FORMS OF MS INITIATING ORAL AUBAGIO (teriflunomide) THERAPY INDICATION AUBAGIO (teriflunomide) is indicated for the treatment of patients with relapsing forms of multiple
FOR YOUR PATIENTS WITH RELAPSING FORMS OF MS INITIATING ORAL AUBAGIO (teriflunomide) THERAPY INDICATION AUBAGIO (teriflunomide) is indicated for the treatment of patients with relapsing forms of multiple
Mycophenolate Mofetil
 Mycophenolate Mofetil Cynthia L. Chen, MD The Permanente Medical Group, Northern California Diablo Service Area February 17, 2018 January 23, 2018 2011 Kaiser Foundation Health Plan, Inc. For internal
Mycophenolate Mofetil Cynthia L. Chen, MD The Permanente Medical Group, Northern California Diablo Service Area February 17, 2018 January 23, 2018 2011 Kaiser Foundation Health Plan, Inc. For internal
Public Assessment Report Scientific discussion. Metoject Metojectpen (methotrexate disodium) SE/H/643/01/DC SE/H/643/02-11/DC
 Public Assessment Report Scientific discussion Metoject Metojectpen (methotrexate disodium) SE/H/643/01/DC SE/H/643/02-11/DC This module reflects the scientific discussion for the approval of Metoject
Public Assessment Report Scientific discussion Metoject Metojectpen (methotrexate disodium) SE/H/643/01/DC SE/H/643/02-11/DC This module reflects the scientific discussion for the approval of Metoject
Clinical Policy: Secukinumab (Cosentyx) Reference Number: ERX.SPA.165 Effective Date:
 Clinical Policy: (Cosentyx) Reference Number: ERX.SPA.165 Effective Date: 10.01.16 Last Review Date: 11.17 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: (Cosentyx) Reference Number: ERX.SPA.165 Effective Date: 10.01.16 Last Review Date: 11.17 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Prescribing Framework for Methotrexate for Immunosuppression in ADULTS
 Hull & East Riding Prescribing Committee Prescribing Framework for Methotrexate for Immunosuppression in ADULTS Patient s Name:.. NHS Number: Patient s Address:... (Use addressograph sticker) GP s Name:...
Hull & East Riding Prescribing Committee Prescribing Framework for Methotrexate for Immunosuppression in ADULTS Patient s Name:.. NHS Number: Patient s Address:... (Use addressograph sticker) GP s Name:...
Systemic Medications for the Dermatology Toolbox: Azathioprine
 Systemic Medications for the Dermatology Toolbox: Azathioprine Wynnis Tom, M.D. Associate Clinical Professor University of California, San Diego and Rady Children s Hospital Faculty Disclosure Information
Systemic Medications for the Dermatology Toolbox: Azathioprine Wynnis Tom, M.D. Associate Clinical Professor University of California, San Diego and Rady Children s Hospital Faculty Disclosure Information
SHARED CARE AGREEMENT: METHOTREXATE S/C
 NB: This document should be read in conjunction with the current Summary of Product Characteristics (SPC) and BSR Guideline for disease-modifying anti-rheumatic drug (DMARD) therapy (available at www.rheumatology.org.uk/resources/guidelines/bsr_guidelines.aspx
NB: This document should be read in conjunction with the current Summary of Product Characteristics (SPC) and BSR Guideline for disease-modifying anti-rheumatic drug (DMARD) therapy (available at www.rheumatology.org.uk/resources/guidelines/bsr_guidelines.aspx
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE SCOPE
 NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE 1 Guideline title SCOPE Psoriasis: the management of psoriasis 1.1 Short title Psoriasis 2 The remit The Department of Health has asked NICE: 'to produce
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE 1 Guideline title SCOPE Psoriasis: the management of psoriasis 1.1 Short title Psoriasis 2 The remit The Department of Health has asked NICE: 'to produce
PUBLIC SUMMARY OF RISK MANAGEMENT PLAN (RMP) METHOTREXATE ORION 2.5 MG & 10 MG TABLET (METHOTREXATE DISODIUM) DATE: , VERSION 1.
 PUBLIC SUMMARY OF RISK MANAGEMENT PLAN (RMP) METHOTREXATE ORION 2.5 MG & 10 MG TABLET (METHOTREXATE DISODIUM) DATE: 23-6-2016, VERSION 1.2 VI.2 Elements for a Public Summary Methotrexate is indicated for:
PUBLIC SUMMARY OF RISK MANAGEMENT PLAN (RMP) METHOTREXATE ORION 2.5 MG & 10 MG TABLET (METHOTREXATE DISODIUM) DATE: 23-6-2016, VERSION 1.2 VI.2 Elements for a Public Summary Methotrexate is indicated for:
TRANSPARENCY COMMITTEE OPINION. 26 April 2006
 TRANSPARENCY COMMITTEE OPINION 26 April 2006 REMICADE 100 mg powder for concentrate for solution for infusion Box of 1 (CIP code: 562 070.1) Applicant : laboratoires Schering Plough List I Drug for hospital
TRANSPARENCY COMMITTEE OPINION 26 April 2006 REMICADE 100 mg powder for concentrate for solution for infusion Box of 1 (CIP code: 562 070.1) Applicant : laboratoires Schering Plough List I Drug for hospital
Clinical Policy: Secukinumab (Cosentyx) Reference Number: ERX.SPA.165 Effective Date:
 Clinical Policy: (Cosentyx) Reference Number: ERX.SPA.165 Effective Date: 10.01.16 Last Review Date: 11.18 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: (Cosentyx) Reference Number: ERX.SPA.165 Effective Date: 10.01.16 Last Review Date: 11.18 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Drugs and Applicable Coding: J-code: Enbrel-J1438; Humira-J0135; Remicade-J1745; Inflectra-Q5102; Cimzia-J0718; Simponi-J1602 Renflexis - pending
 Policy Subject: Anti-TNF Agents Policy Number: SHS PBD16 Category: Rheumatology & Autoimmune Policy Type: Medical Pharmacy Department: Pharmacy Product (check all that apply): Group HMO/POS Individual
Policy Subject: Anti-TNF Agents Policy Number: SHS PBD16 Category: Rheumatology & Autoimmune Policy Type: Medical Pharmacy Department: Pharmacy Product (check all that apply): Group HMO/POS Individual
Monitoring of adverse drug reactions caused by methotrexate at the dermatology department of a tertiary care centre
 International Journal of Research in Dermatology Sivakumar S et al. Int J Res Dermatol. 2017 Sep;3(3):389-394 http://www.ijord.com Original Research Article DOI: http://dx.doi.org/10.18203/issn.2455-4529.intjresdermatol20173078
International Journal of Research in Dermatology Sivakumar S et al. Int J Res Dermatol. 2017 Sep;3(3):389-394 http://www.ijord.com Original Research Article DOI: http://dx.doi.org/10.18203/issn.2455-4529.intjresdermatol20173078
Clinical Policy: Secukinumab (Cosentyx) Reference Number: CP.PHAR.261 Effective Date: Last Review Date: Line of Business: HIM, Medicaid
 Clinical Policy: (Cosentyx) Reference Number: CP.PHAR.261 Effective Date: 08.16 Last Review Date: 11.18 Line of Business: HIM, Medicaid Coding Implications Revision Log See Important Reminder at the end
Clinical Policy: (Cosentyx) Reference Number: CP.PHAR.261 Effective Date: 08.16 Last Review Date: 11.18 Line of Business: HIM, Medicaid Coding Implications Revision Log See Important Reminder at the end
Clinical Policy: Ixekizumab (Taltz) Reference Number: ERX.SPA.122 Effective Date:
 Clinical Policy: (Taltz) Reference Number: ERX.SPA.122 Effective Date: 10.01.16 Last Review Date: 11.18 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: (Taltz) Reference Number: ERX.SPA.122 Effective Date: 10.01.16 Last Review Date: 11.18 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
The Treatment Toolbox for Severe Pediatric Psoriasis
 The Treatment Toolbox for Severe Pediatric Psoriasis Dr. Kim A. Papp, MD, PhD, FRCPC, FAAD K Papp Clinical Research and Probity Medical Research Objectives: Treating severe pediatric psoriasis 1. Challenges
The Treatment Toolbox for Severe Pediatric Psoriasis Dr. Kim A. Papp, MD, PhD, FRCPC, FAAD K Papp Clinical Research and Probity Medical Research Objectives: Treating severe pediatric psoriasis 1. Challenges
SYSTEMIC THERAPY OF MODERATE AND SEVERE PSORIASIS WITH METHOTREXATE
 Bulletin of the Transilvania University of Braşov Series VI: Medical Sciences Vol. 5 (54) No. 2-2012 SYSTEMIC THERAPY OF MODERATE AND SEVERE PSORIASIS WITH METHOTREXATE M. FRÎNCU 1 A. OANŢĂ 1 Abstract:
Bulletin of the Transilvania University of Braşov Series VI: Medical Sciences Vol. 5 (54) No. 2-2012 SYSTEMIC THERAPY OF MODERATE AND SEVERE PSORIASIS WITH METHOTREXATE M. FRÎNCU 1 A. OANŢĂ 1 Abstract:
Olumiant (baricitinib) NEW PRODUCT SLIDESHOW
 Olumiant (baricitinib) NEW PRODUCT SLIDESHOW Introduction Brand name: Olumiant Generic name: Baricitinib Pharmacological class: Janus kinase (JAK) inhibitor Strength and Formulation: 2mg; tabs Manufacturer:
Olumiant (baricitinib) NEW PRODUCT SLIDESHOW Introduction Brand name: Olumiant Generic name: Baricitinib Pharmacological class: Janus kinase (JAK) inhibitor Strength and Formulation: 2mg; tabs Manufacturer:
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION. 13 May 2009
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 13 May 2009 STELARA 45 mg, solution for injection B/1 x 0.5 ml vial (CIP code: 392 586-2) JANSSEN-CILAG Ustekinumab
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 13 May 2009 STELARA 45 mg, solution for injection B/1 x 0.5 ml vial (CIP code: 392 586-2) JANSSEN-CILAG Ustekinumab
Clinical Policy: Apremilast (Otezla) Reference Number: CP.PHAR.245 Effective Date: Last Review Date: Line of Business: HIM, Medicaid
 Clinical Policy: (Otezla) Reference Number: CP.PHAR.245 Effective Date: 08.16 Last Review Date: 11.18 Line of Business: HIM, Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: (Otezla) Reference Number: CP.PHAR.245 Effective Date: 08.16 Last Review Date: 11.18 Line of Business: HIM, Medicaid Revision Log See Important Reminder at the end of this policy for important
Sarcoidosis Case. Robert P. Baughman Interstitial Lung Disease and Sarcoidosis Clinic University of Cincinnati, USA. WASOG: educational material
 Sarcoidosis Case Robert P. Baughman Interstitial Lung Disease and Sarcoidosis Clinic University of Cincinnati, USA WASOG: educational material Sarcoidosis Case patient is a Caucasian male age 46 was diagnosed
Sarcoidosis Case Robert P. Baughman Interstitial Lung Disease and Sarcoidosis Clinic University of Cincinnati, USA WASOG: educational material Sarcoidosis Case patient is a Caucasian male age 46 was diagnosed
Figure 1. Stepwise approach of treating patients with rheumatoid arthritis.
 Establish diagnosis early Document baseline disease activity and damage Estimate prognosis Initiate therapy Begin patient education Start DMARD therapy within 3 months Consider NSAID Consider local or
Establish diagnosis early Document baseline disease activity and damage Estimate prognosis Initiate therapy Begin patient education Start DMARD therapy within 3 months Consider NSAID Consider local or
ETANERCEPT Generic Brand HICL GCN Exception/Other ETANERCEPT ENBREL GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW)
 Generic Brand HICL GCN Exception/Other ETANERCEPT ENBREL 18830 GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Does the patient have a diagnosis of moderate to severe rheumatoid
Generic Brand HICL GCN Exception/Other ETANERCEPT ENBREL 18830 GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Does the patient have a diagnosis of moderate to severe rheumatoid
Tavalisse (fostamatinib disodium hexahydrate) NEW PRODUCT SLIDESHOW
 Tavalisse (fostamatinib disodium hexahydrate) NEW PRODUCT SLIDESHOW Introduction Brand name: Tavalisse Generic name: Fostamatinib disodium hexahydrate Pharmacological class: Tyrosine kinase inhibitor Strength
Tavalisse (fostamatinib disodium hexahydrate) NEW PRODUCT SLIDESHOW Introduction Brand name: Tavalisse Generic name: Fostamatinib disodium hexahydrate Pharmacological class: Tyrosine kinase inhibitor Strength
SHARED CARE GUIDELINE
 SHARED CARE GUIDELINE Drug: Methotrexate Introduction Indication: Licensed: Rheumatoid arthritis, severe psoriasis, severe active juvenile idiopathic arthritis, severe psoriatic arthritis, mild to moderate
SHARED CARE GUIDELINE Drug: Methotrexate Introduction Indication: Licensed: Rheumatoid arthritis, severe psoriasis, severe active juvenile idiopathic arthritis, severe psoriatic arthritis, mild to moderate
Extreme dermatoheliosis: How to approach the severely sun damaged patient
 Extreme dermatoheliosis: How to approach the severely sun damaged patient Anokhi Jambusaria MD, MSCE Staff Dermatologist Baylor Scott and White Health Round Rock, TX I have no relevant conflicts of interest
Extreme dermatoheliosis: How to approach the severely sun damaged patient Anokhi Jambusaria MD, MSCE Staff Dermatologist Baylor Scott and White Health Round Rock, TX I have no relevant conflicts of interest
chemotherapeutic agents in
 Use of biologics and chemotherapeutic agents in cutaneous emergencies: Focus on lifethreatening forms of psoriasis Alice Bendix Gottlieb MD, PhD Professor of Dermatology New York Medical College Metropolitan
Use of biologics and chemotherapeutic agents in cutaneous emergencies: Focus on lifethreatening forms of psoriasis Alice Bendix Gottlieb MD, PhD Professor of Dermatology New York Medical College Metropolitan
DOSING AND ADMINISTRATION GUIDE
 DOSING AND ADMINISTRATION GUIDE Indication TAVALISSE is a kinase inhibitor indicated for the treatment of thrombocytopenia in adult patients with chronic immune thrombocytopenia (ITP) who have had an insufficient
DOSING AND ADMINISTRATION GUIDE Indication TAVALISSE is a kinase inhibitor indicated for the treatment of thrombocytopenia in adult patients with chronic immune thrombocytopenia (ITP) who have had an insufficient
Clinical Policy: Ixekizumab (Taltz) Reference Number: CP.PHAR.257 Effective Date: Last Review Date: 05.18
 Clinical Policy: (Taltz) Reference Number: CP.PHAR.257 Effective Date: 08.01.16 Last Review Date: 05.18 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: (Taltz) Reference Number: CP.PHAR.257 Effective Date: 08.01.16 Last Review Date: 05.18 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: Ustekinumab (Stelara) Reference Number: ERX.SPA.01 Effective Date:
 Clinical Policy: (Stelara) Reference Number: ERX.SPA.01 Effective Date: 04.01.17 Last Review Date: 11.17 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: (Stelara) Reference Number: ERX.SPA.01 Effective Date: 04.01.17 Last Review Date: 11.17 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Immunosuppressants. Assistant Prof. Dr. Najlaa Saadi PhD Pharmacology Faculty of Pharmacy University of Philadelphia
 Immunosuppressants Assistant Prof. Dr. Najlaa Saadi PhD Pharmacology Faculty of Pharmacy University of Philadelphia Immunosuppressive Agents Very useful in minimizing the occurrence of exaggerated or inappropriate
Immunosuppressants Assistant Prof. Dr. Najlaa Saadi PhD Pharmacology Faculty of Pharmacy University of Philadelphia Immunosuppressive Agents Very useful in minimizing the occurrence of exaggerated or inappropriate
Clinical Policy: Ustekinumab (Stelara) Reference Number: CP.PHAR.264 Effective Date: Last Review Date: Line of Business: HIM, Medicaid
 Clinical Policy: (Stelara) Reference Number: CP.PHAR.264 Effective Date: 08.16 Last Review Date: 05.18 Line of Business: HIM, Medicaid Coding Implications Revision Log See Important Reminder at the end
Clinical Policy: (Stelara) Reference Number: CP.PHAR.264 Effective Date: 08.16 Last Review Date: 05.18 Line of Business: HIM, Medicaid Coding Implications Revision Log See Important Reminder at the end
Serologic Markers CONVENTIONAL ANTIBODIES ANTIBODIES UNCONVENTIONAL. AIH Type I
 Autoimmune Hepatitis By Thomas Frazier Objective What we need to know about AIH Diagnosis Treatment Difficulties in both Liver transplantation concerns AASLD Guidelines: Hepatology. 2010 Jun;51(6):2193-213.
Autoimmune Hepatitis By Thomas Frazier Objective What we need to know about AIH Diagnosis Treatment Difficulties in both Liver transplantation concerns AASLD Guidelines: Hepatology. 2010 Jun;51(6):2193-213.
Shared Care Protocol Oral methotrexate 2.5mg tablets in dermatology/gastroenterology/rheumatology patients
 Shared Care Protocol Oral methotrexate 2.5mg tablets in dermatology/gastroenterology/rheumatology patients Specialist Details Name: Patient Identifier (please include NHS number as minimum) Location: Telephone
Shared Care Protocol Oral methotrexate 2.5mg tablets in dermatology/gastroenterology/rheumatology patients Specialist Details Name: Patient Identifier (please include NHS number as minimum) Location: Telephone
Systemic therapy of psoriasis: methotrexate
 390 394 CLINICAL STUDY Systemic therapy of psoriasis: methotrexate Kozub P, Simaljakova M Department of Dermatology, Faculty of Medicine, University Hospital, Bratislava, Slovakia. peter.kozub@laposte.net
390 394 CLINICAL STUDY Systemic therapy of psoriasis: methotrexate Kozub P, Simaljakova M Department of Dermatology, Faculty of Medicine, University Hospital, Bratislava, Slovakia. peter.kozub@laposte.net
Clinical Policy: Etanercept (Enbrel) Reference Number: PA.CP.PHAR.250 Effective Date: 01/18 Last Review Date: 08/17 Line of Business: Medicaid
 Clinical Policy: (Enbrel) Reference Number: PA.CP.PHAR.250 Effective Date: 01/18 Last Review Date: 08/17 Line of Business: Medicaid Coding Implications Revision Log Description (Enbrel ) is tumor necrosis
Clinical Policy: (Enbrel) Reference Number: PA.CP.PHAR.250 Effective Date: 01/18 Last Review Date: 08/17 Line of Business: Medicaid Coding Implications Revision Log Description (Enbrel ) is tumor necrosis
Psoriasis. Andrei Metelitsa, MD, FRCPC, FAAD Clinical Associate Professor, Dermatology, U of C Co-Director, Institute for Skin Advancement
 Psoriasis Andrei Metelitsa, MD, FRCPC, FAAD Clinical Associate Professor, Dermatology, U of C Co-Director, Institute for Skin Advancement Copyright 2017 by Sea Courses Inc. All rights reserved. No part
Psoriasis Andrei Metelitsa, MD, FRCPC, FAAD Clinical Associate Professor, Dermatology, U of C Co-Director, Institute for Skin Advancement Copyright 2017 by Sea Courses Inc. All rights reserved. No part
Weekly oral and subcutaneous methotrexate
 This guideline is currently under review. In the interim, the guideline remains valid; if GPs have any specific concerns or questions, they should seek advice from the specialist with whom they have agreed
This guideline is currently under review. In the interim, the guideline remains valid; if GPs have any specific concerns or questions, they should seek advice from the specialist with whom they have agreed
Humira. (adalimumab) Drug Update Slideshow NEW INDICATION
 Humira (adalimumab) NEW INDICATION Drug Update Slideshow Introduction Brand name: Humira Generic name: Adalimumab Pharmacological class: Tumor necrosis factor (TNF) blocker Strength and Formulation: 10mg/0.2mL,
Humira (adalimumab) NEW INDICATION Drug Update Slideshow Introduction Brand name: Humira Generic name: Adalimumab Pharmacological class: Tumor necrosis factor (TNF) blocker Strength and Formulation: 10mg/0.2mL,
Aubagio. Aubagio (teriflunomide) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.60.09 Subject: Aubagio Page: 1 of 6 Last Review Date: June 22, 2017 Aubagio Description Aubagio (teriflunomide)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.60.09 Subject: Aubagio Page: 1 of 6 Last Review Date: June 22, 2017 Aubagio Description Aubagio (teriflunomide)
EMEA PUBLIC STATEMENT ON LEFLUNOMIDE (ARAVA) - PANCYTOPENIA AND SERIOUS SKIN REACTIONS
 The European Agency for the Evaluation of Medicinal Products Evaluation of Medicines for Human Use London, 25 October 1999 EMEA/31637/99 EMEA PUBLIC STATEMENT ON LEFLUNOMIDE (ARAVA) - PANCYTOPENIA AND
The European Agency for the Evaluation of Medicinal Products Evaluation of Medicines for Human Use London, 25 October 1999 EMEA/31637/99 EMEA PUBLIC STATEMENT ON LEFLUNOMIDE (ARAVA) - PANCYTOPENIA AND
Methotrexate Injectable Step Therapy Program Summary
 Methotrexate Injectable Step Therapy Program Summary This prior authorization applies to Commercial, SourceRx and Health Insurance Marketplace formularies. OBJECTIVE The intent of the methotrexate injectable
Methotrexate Injectable Step Therapy Program Summary This prior authorization applies to Commercial, SourceRx and Health Insurance Marketplace formularies. OBJECTIVE The intent of the methotrexate injectable
FAMILY PLANNING AND AUBAGIO (teriflunomide)
 FAMILY PLANNING AND AUBAGIO (teriflunomide) INDICATION AUBAGIO (teriflunomide) is indicated for the treatment of patients with relapsing forms of multiple sclerosis. IMPORTANT SAFETY INFORMATION WARNING:
FAMILY PLANNING AND AUBAGIO (teriflunomide) INDICATION AUBAGIO (teriflunomide) is indicated for the treatment of patients with relapsing forms of multiple sclerosis. IMPORTANT SAFETY INFORMATION WARNING:
ACTEMRA (tocilizumab)
 RATIONALE FOR INCLUSION IN PA PROGRAM Background Actemra is an agent in the class of drugs known as biologic disease modifiers. It is used to treat adult onset rheumatoid (RA) arthritis, polyarticular
RATIONALE FOR INCLUSION IN PA PROGRAM Background Actemra is an agent in the class of drugs known as biologic disease modifiers. It is used to treat adult onset rheumatoid (RA) arthritis, polyarticular
Aubagio. Aubagio (teriflunomide) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.07.09 Subsection: Endocrine and Metabolic Drugs Original Policy Date: April 1, 2013 Subject: Aubagio
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.07.09 Subsection: Endocrine and Metabolic Drugs Original Policy Date: April 1, 2013 Subject: Aubagio
PRESCRIBING GUIDANCE METHOTREXATE for the treatment of vasculitis
 PRESCRIBING GUIDANCE METHOTREXATE for the treatment of vasculitis For the latest information on interactions and adverse effects, always consult the latest version of the Summary of Product Characteristics
PRESCRIBING GUIDANCE METHOTREXATE for the treatment of vasculitis For the latest information on interactions and adverse effects, always consult the latest version of the Summary of Product Characteristics
Clinical Policy: Secukinumab (Cosentyx) Reference Number: CP.PHAR.261 Effective Date: 08/16 Last Review Date: 08/17
 Clinical Policy: (Cosentyx) Reference Number: CP.PHAR.261 Effective Date: 08/16 Last Review Date: 08/17 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: (Cosentyx) Reference Number: CP.PHAR.261 Effective Date: 08/16 Last Review Date: 08/17 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
The use of methotrexate in treatment of moderate to severe psoriasis
 Hong Kong J. Dermatol. Venereol. (2009) 17, 13-19 Review Article The use of methotrexate in treatment of moderate to severe psoriasis WY Leung and KH Lau Psoriasis is a common, chronic and non-infectious
Hong Kong J. Dermatol. Venereol. (2009) 17, 13-19 Review Article The use of methotrexate in treatment of moderate to severe psoriasis WY Leung and KH Lau Psoriasis is a common, chronic and non-infectious
THE THERAPY OF THE REBEL SEVERE PSORIAZIS WITH BIOLOGICAL PREPARATS
 Bulletin of the Transilvania University of Braşov Series VI: Medical Sciences Vol. 5 (54) No. 2-2012 THE THERAPY OF THE REBEL SEVERE PSORIAZIS WITH BIOLOGICAL PREPARATS Mădălina FRÎNCU 1 Abstract: Biological
Bulletin of the Transilvania University of Braşov Series VI: Medical Sciences Vol. 5 (54) No. 2-2012 THE THERAPY OF THE REBEL SEVERE PSORIAZIS WITH BIOLOGICAL PREPARATS Mădălina FRÎNCU 1 Abstract: Biological
2. Does the patient have a diagnosis of ulcerative colitis or Crohn s? Y N
 Pharmacy Prior Authorization AETA BETTER HEALTH LOUISIAA (MEDICAID) Remicade (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign
Pharmacy Prior Authorization AETA BETTER HEALTH LOUISIAA (MEDICAID) Remicade (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign
Research Article Clinical Outcome of a Novel Anti-CD6 Biologic Itolizumab in Patients of Psoriasis with Comorbid Conditions
 Dermatology Research and Practice Volume 2, Article ID 13326, 4 pages http://dx.doi.org/.1155/2/13326 Research Article Clinical Outcome of a Novel Anti-CD6 Biologic Itolizumab in Patients of Psoriasis
Dermatology Research and Practice Volume 2, Article ID 13326, 4 pages http://dx.doi.org/.1155/2/13326 Research Article Clinical Outcome of a Novel Anti-CD6 Biologic Itolizumab in Patients of Psoriasis
Clinical Policy: Ixekizumab (Taltz) Reference Number: CP.PHAR.257 Effective Date: Last Review Date: 11.18
 Clinical Policy: (Taltz) Reference Number: CP.PHAR.257 Effective Date: 08.01.16 Last Review Date: 11.18 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: (Taltz) Reference Number: CP.PHAR.257 Effective Date: 08.01.16 Last Review Date: 11.18 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
3. Has the patient shown improvement in signs and symptoms of the disease? Y N
 Pharmacy Prior Authorization MERC CARE (MEDICAID) Renflexis (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign and date. Fax
Pharmacy Prior Authorization MERC CARE (MEDICAID) Renflexis (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign and date. Fax
Ciclosporin 25mg, 50mg, 100mg capsules Ciclosporin oral solution 100mg/ml
 Shared Care Protocol Ciclosporin for the treatment of rheumatoid arthritis Name of drug, form and strength Background Ciclosporin 25mg, 50mg, 100mg capsules Ciclosporin oral solution 100mg/ml Ciclosporin
Shared Care Protocol Ciclosporin for the treatment of rheumatoid arthritis Name of drug, form and strength Background Ciclosporin 25mg, 50mg, 100mg capsules Ciclosporin oral solution 100mg/ml Ciclosporin
Products available Methotrexate tablets 2.5mg ONLY (Methotrexate tablets 10mg are NOT recommended as per NPSA guidance 5 ).
 Methotrexate Traffic light classification- Amber 1 Information sheet for Primary Care Prescribers Part of the Shared Care Protocol: Management of Rheumatological Conditions with Disease-Modifying Anti
Methotrexate Traffic light classification- Amber 1 Information sheet for Primary Care Prescribers Part of the Shared Care Protocol: Management of Rheumatological Conditions with Disease-Modifying Anti
Rheumatoid arthritis
 Rheumatoid arthritis 1 Definition Rheumatoid arthritis is one of the most common inflammatory disorders affecting the population worldwide. It is a systemic inflammatory disease which affects not only
Rheumatoid arthritis 1 Definition Rheumatoid arthritis is one of the most common inflammatory disorders affecting the population worldwide. It is a systemic inflammatory disease which affects not only
Anti-cancer drugs. Introduction : Body : 1) Alkylating Agents
 Anti-cancer drugs Introduction : In this journal I will try to explain what is anti-cancer agents, how they work, how can they inhibit the growth of tumor and what is the advantages and disadvantages of
Anti-cancer drugs Introduction : In this journal I will try to explain what is anti-cancer agents, how they work, how can they inhibit the growth of tumor and what is the advantages and disadvantages of
Significance. Outline and Objectives. S007 Systemic Therapies for Medical Oncology
 S007 Systemic Therapies for Medical Oncology Anisha B. Patel, M.D. Assistant Professor, Dermatology UT MD Anderson Cancer Center UT Health Science Center Houston Safety considerations Outline and Objectives
S007 Systemic Therapies for Medical Oncology Anisha B. Patel, M.D. Assistant Professor, Dermatology UT MD Anderson Cancer Center UT Health Science Center Houston Safety considerations Outline and Objectives
Ustekinumab (Stelara) for psoriatic arthritis second line after disease modifying anti rheumatic drugs (DMARDs)
 Ustekinumab (Stelara) for psoriatic arthritis second line after disease modifying anti rheumatic drugs (DMARDs) January 2010 This technology summary is based on information available at the time of research
Ustekinumab (Stelara) for psoriatic arthritis second line after disease modifying anti rheumatic drugs (DMARDs) January 2010 This technology summary is based on information available at the time of research
Duration of treatment All DMARDs are long term treatments. Clinical benefit may take up to 6 months. 1
 Leflunomide Traffic light classification- Amber 1 Information sheet for Primary Care Prescribers Part of the Shared Care Protocol: Management of Rheumatological Conditions with Disease-Modifying Anti Rheumatic
Leflunomide Traffic light classification- Amber 1 Information sheet for Primary Care Prescribers Part of the Shared Care Protocol: Management of Rheumatological Conditions with Disease-Modifying Anti Rheumatic
USTEKINUMAB Generic Brand HICL GCN Exception/Other USTEKINUMAB STELARA GUIDELINES FOR USE
 Generic Brand HICL GCN Exception/Other USTEKINUMAB STELARA 36187 GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Does the patient have a diagnosis of psoriatic arthritis (PsA)
Generic Brand HICL GCN Exception/Other USTEKINUMAB STELARA 36187 GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Does the patient have a diagnosis of psoriatic arthritis (PsA)
Break the Liver Biopsy Habit
 Break the Liver Biopsy Habit Dirk Elston MD Professor and Chairman Department of Dermatology and Dermatologic Surgery Medical University of South Carolina No conflict of interest Father of one of my nurses:
Break the Liver Biopsy Habit Dirk Elston MD Professor and Chairman Department of Dermatology and Dermatologic Surgery Medical University of South Carolina No conflict of interest Father of one of my nurses:
Understanding Myositis Medications
 Understanding Myositis Medications 2015 TMA Annual Patient Conference Orlando, Florida Chester V. Oddis, MD University of Pittsburgh Director, Myositis Center Disclosures Mallinckrodt: Research Grant Genentech:
Understanding Myositis Medications 2015 TMA Annual Patient Conference Orlando, Florida Chester V. Oddis, MD University of Pittsburgh Director, Myositis Center Disclosures Mallinckrodt: Research Grant Genentech:
Canadian Society of Internal Medicine Annual Meeting 2016 Montreal, QC
 Canadian Society of Internal Medicine Annual Meeting 2016 Montreal, QC Update on the Treatment of Rheumatoid Arthritis Sabrina Fallavollita MDCM McGill University Canadian Society of Internal Medicine
Canadian Society of Internal Medicine Annual Meeting 2016 Montreal, QC Update on the Treatment of Rheumatoid Arthritis Sabrina Fallavollita MDCM McGill University Canadian Society of Internal Medicine
Pravit Asawanonda, MD, DSc, and Yaowalak Nateetongrungsak, MD Bangkok, Thailand
 Methotrexate plus narrowband UVB phototherapy versus narrowband UVB phototherapy alone in the treatment of plaque-type psoriasis: A randomized, placebo-controlled study Pravit Asawanonda, MD, DSc, and
Methotrexate plus narrowband UVB phototherapy versus narrowband UVB phototherapy alone in the treatment of plaque-type psoriasis: A randomized, placebo-controlled study Pravit Asawanonda, MD, DSc, and
2017 Blue Cross and Blue Shield of Louisiana
 Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Prescribing Guidelines Prescribing arrangement for the management of patients transferring from Secondary Care to Primary Care
 Berkshire West Integrated Care System Representing Berkshire West Clinical Commisioning Group Royal Berkshire NHS Foundation Trust Berkshire Healthcare NHS Foundation Trust Berkshire West Primary Care
Berkshire West Integrated Care System Representing Berkshire West Clinical Commisioning Group Royal Berkshire NHS Foundation Trust Berkshire Healthcare NHS Foundation Trust Berkshire West Primary Care
Treatment with steroids and immunosuppressants
 Treatment with steroids and immunosuppressants Donna Culton, MD, PhD University of North Carolina IPPF 2017 Annual Patient Conference Newport Beach, CA September 16, 2017 Factors that will influence therapy
Treatment with steroids and immunosuppressants Donna Culton, MD, PhD University of North Carolina IPPF 2017 Annual Patient Conference Newport Beach, CA September 16, 2017 Factors that will influence therapy
Antineoplastic Drugs. Antineoplastic Drugs. Herbal Alert. Pharmacology Chapter 51. Green tea
 Antineoplastic Drugs Pharmacology Chapter 51 Carolyn Wright, RN, BSN Ford, S. & Roach, S. (2010). Roach's introductory clinical pharmacology (9 th ed.). Philadelphia: Wolters Kluwer/Lippincott Williams
Antineoplastic Drugs Pharmacology Chapter 51 Carolyn Wright, RN, BSN Ford, S. & Roach, S. (2010). Roach's introductory clinical pharmacology (9 th ed.). Philadelphia: Wolters Kluwer/Lippincott Williams
Psoriasis. What is Psoriasis? What causes psoriasis? Medical Topics Psoriasis
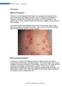 1 Psoriasis What is Psoriasis? Psoriasis is a long standing inflammatory non-contagious skin disease which waxes and wanes with triggering factors. There is a genetic predisposition in psoriasis. Internationally,
1 Psoriasis What is Psoriasis? Psoriasis is a long standing inflammatory non-contagious skin disease which waxes and wanes with triggering factors. There is a genetic predisposition in psoriasis. Internationally,
UNDERSTANDING HAIR THINNING/HAIR LOSS
 UNDERSTANDING HAIR THINNING/HAIR LOSS INDICATION AUBAGIO (teriflunomide) is indicated for the treatment of patients with relapsing forms of multiple sclerosis. IMPORTANT SAFETY INFORMATION WARNING: HEPATOTOXICITY
UNDERSTANDING HAIR THINNING/HAIR LOSS INDICATION AUBAGIO (teriflunomide) is indicated for the treatment of patients with relapsing forms of multiple sclerosis. IMPORTANT SAFETY INFORMATION WARNING: HEPATOTOXICITY
Supplementary appendix
 Supplementary appendix This appendix formed part of the original submission and has been peer reviewed. We post it as supplied by the authors. Supplement to: Warren RB, Mrowietz U, von Kiedrowski R, et
Supplementary appendix This appendix formed part of the original submission and has been peer reviewed. We post it as supplied by the authors. Supplement to: Warren RB, Mrowietz U, von Kiedrowski R, et
Phototherapy for Psoriasis. Henry W. Lim, MD Chairman and C.S. Livingood Chair Department of Dermatology Henry Ford Hospital, Detroit, MI, USA
 Phototherapy for Psoriasis Henry W. Lim, MD Chairman and C.S. Livingood Chair Department of Dermatology Henry Ford Hospital, Detroit, MI, USA Disclosure Investigator: Clinuvel Estée Lauder Ferndale Incyte
Phototherapy for Psoriasis Henry W. Lim, MD Chairman and C.S. Livingood Chair Department of Dermatology Henry Ford Hospital, Detroit, MI, USA Disclosure Investigator: Clinuvel Estée Lauder Ferndale Incyte
ACTEMRA Risk Mitigation Strategy Presenter Name, Degree
 ACTEMRA Risk Mitigation Strategy Presenter Name, Degree Medical Science Liaison Genentech, Inc. 1 Indications and Dosage Rheumatoid Arthritis (RA) (1 of 2) Indication in RA ACTEMRA (tocilizumab) is indicated
ACTEMRA Risk Mitigation Strategy Presenter Name, Degree Medical Science Liaison Genentech, Inc. 1 Indications and Dosage Rheumatoid Arthritis (RA) (1 of 2) Indication in RA ACTEMRA (tocilizumab) is indicated
London, 1 June 2006 Product name: REMICADE Procedure number: Remicade-H-240-II-73-AR SCIENTIFIC DISCUSSION 1/8
 London, 1 June 2006 Product name: REMICADE Procedure number: Remicade-H-240-II-73-AR SCIENTIFIC DISCUSSION 1/8 1. Introduction Infliximab is a chimeric human-murine IgG1κ monoclonal antibody, which binds
London, 1 June 2006 Product name: REMICADE Procedure number: Remicade-H-240-II-73-AR SCIENTIFIC DISCUSSION 1/8 1. Introduction Infliximab is a chimeric human-murine IgG1κ monoclonal antibody, which binds
Greater Manchester Interface Prescribing Group Shared Care Template
 Greater Manchester Interface Prescribing Group Shared Care Template Shared Care Guideline for Oral Methotrexate for Rheumatological Conditions Author(s)/Originator(s): (please state author name and department)
Greater Manchester Interface Prescribing Group Shared Care Template Shared Care Guideline for Oral Methotrexate for Rheumatological Conditions Author(s)/Originator(s): (please state author name and department)
Horizon Scanning Centre January Apremilast for psoriasis SUMMARY NIHR HSC ID: 2652
 Horizon Scanning Centre January 2013 Apremilast for psoriasis SUMMARY NIHR HSC ID: 2652 This briefing is based on information available at the time of research and a limited literature search. It is not
Horizon Scanning Centre January 2013 Apremilast for psoriasis SUMMARY NIHR HSC ID: 2652 This briefing is based on information available at the time of research and a limited literature search. It is not
Clinical Policy: Apremilast (Otezla) Reference Number: CP.PHAR.245 Effective Date: 08/16 Last Review Date 08/17
 Clinical Policy: (Otezla) Reference Number: CP.PHAR.245 Effective Date: 08/16 Last Review Date 08/17 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: (Otezla) Reference Number: CP.PHAR.245 Effective Date: 08/16 Last Review Date 08/17 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
ACTEMRA IV Dosing & Administration Pocket Guide
 IMPORTANT SAFETY INFORMATION BOXED WARNING Serious Infections Serious infections leading to hospitalization or death, including tuberculosis (TB), bacterial, invasive fungal, viral, and other opportunistic
IMPORTANT SAFETY INFORMATION BOXED WARNING Serious Infections Serious infections leading to hospitalization or death, including tuberculosis (TB), bacterial, invasive fungal, viral, and other opportunistic
Clinical Policy: Ustekinumab (Stelara) Reference Number: CP.PHAR.264
 Clinical Policy: (Stelara) Reference Number: CP.PHAR.264 Effective Date: 08/16 Last Review Date: 05/16 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
Clinical Policy: (Stelara) Reference Number: CP.PHAR.264 Effective Date: 08/16 Last Review Date: 05/16 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
Pharmacy Prior Authorization
 Pharmacy Prior Authorization AETA BETTER HEALTH PESLVAIA & AETA BETTER HEALTH KIDS Humira (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information,
Pharmacy Prior Authorization AETA BETTER HEALTH PESLVAIA & AETA BETTER HEALTH KIDS Humira (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information,
Weekly oral and subcutaneous methotrexate
 This guideline is currently under review. In the interim, the guideline remains valid; if GPs have any specific concerns or questions, they should seek advice from the specialist with whom they have agreed
This guideline is currently under review. In the interim, the guideline remains valid; if GPs have any specific concerns or questions, they should seek advice from the specialist with whom they have agreed
Treatment monitoring protocol for Dimethyl fumarate therapy in active Relapsing Remitting Multiple Sclerosis
 Treatment monitoring protocol for Dimethyl fumarate therapy in active Relapsing Remitting Multiple Sclerosis This protocol provides monitoring guidance for adult patients requiring Dimethyl fumarate therapy
Treatment monitoring protocol for Dimethyl fumarate therapy in active Relapsing Remitting Multiple Sclerosis This protocol provides monitoring guidance for adult patients requiring Dimethyl fumarate therapy
Autoimmune Hepatitis. What Drug and for How Long? Hepatology Day May 30 th, 2015
 Autoimmune Hepatitis What Drug and for How Long? Rajaa Chatila, MD Associate Professor of Medicine Director, Internal Medicine Residency Program Lebanese American University Hepatology Day May 30 th, 2015
Autoimmune Hepatitis What Drug and for How Long? Rajaa Chatila, MD Associate Professor of Medicine Director, Internal Medicine Residency Program Lebanese American University Hepatology Day May 30 th, 2015
STELARA. ustekinumab NEW ZEALAND DATA SHEET
 1. PRODUCT NAME ustekinumab NEW ZEALAND DATA SHEET 45 mg solution for injection in vial. 90 mg solution for injection in vial. 45 mg solution for injection in pre-filled syringe. 90 mg solution for injection
1. PRODUCT NAME ustekinumab NEW ZEALAND DATA SHEET 45 mg solution for injection in vial. 90 mg solution for injection in vial. 45 mg solution for injection in pre-filled syringe. 90 mg solution for injection
A study of treatment modalities in psoriasis in dermatology outpatient department of a tertiary care teaching hospital
 Original article A study of treatment modalities in psoriasis in dermatology outpatient department of a tertiary care teaching hospital 1Y Roja Ramani, 2 Benu Panigrahy, 3 Sailenkumar Mishra, 4 BTPS Singh
Original article A study of treatment modalities in psoriasis in dermatology outpatient department of a tertiary care teaching hospital 1Y Roja Ramani, 2 Benu Panigrahy, 3 Sailenkumar Mishra, 4 BTPS Singh
HARVARD PILGRIM HEALTH CARE RECOMMENDED MEDICATION REQUEST GUIDELINES
 Generic Brand HICL GCN Exception/Other CERTOLIZUMAB PEGOL CIMZIA 35554 GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Is the request for a patient with a diagnosis of moderate
Generic Brand HICL GCN Exception/Other CERTOLIZUMAB PEGOL CIMZIA 35554 GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Is the request for a patient with a diagnosis of moderate
Vemlidy. (tenofovir alafenamide) New Product Slideshow
 Vemlidy (tenofovir alafenamide) New Product Slideshow Introduction Brand name: Vemlidy Generic name: Tenofovir alafenamide Pharmacological class: Nucleoside analogue (reverse transcriptase inhibitor) Strength
Vemlidy (tenofovir alafenamide) New Product Slideshow Introduction Brand name: Vemlidy Generic name: Tenofovir alafenamide Pharmacological class: Nucleoside analogue (reverse transcriptase inhibitor) Strength
Drugs Used in Anemia
 Drugs Used in Anemia Drugs of Anemia Anemia is defined as a below-normal plasma hemoglobin concentration resulting from: a decreased number of circulating red blood cells or an abnormally low total hemoglobin
Drugs Used in Anemia Drugs of Anemia Anemia is defined as a below-normal plasma hemoglobin concentration resulting from: a decreased number of circulating red blood cells or an abnormally low total hemoglobin
