Contemporary Views of the Impact of Biologic Therapy on Infection Risk
|
|
|
- Rosamund Arnold
- 6 years ago
- Views:
Transcription
1 Contemporary Views of the Impact of Biologic Therapy on Infection Risk Cezarina Mindru, M.D. Assistant Professor of Medicine Baylor College of Medicine VA Michel E. Debakey, Houston, TX Faculty Disclosures Dr. Mindru has listed an affiliation with: Consultant Gilead Sciences, Inc. However, no conflict of interest exists for this conference. Special thanks to Dr. John W. Baddley, UAB. 1
2 Objectives Describe the frequency of infectious complications associated with TNF alpha inhibitor use Measures we can take to mitigate those risks Discuss few cases TNF alpha: the good and the bad Tsiodras et al. Mayo Clin Proc 2008;83:
3 Nature Reviews: Immunology 3
4 Traditional TNF alpha inhibitors Drug Brand Mechanism Approval Indications infliximab Remicade Chimeric anti-tnf mab etanercept Enbrel Soluble TNF- receptor fusion protein adalimumab Humira Human anti-tnf mab golimumab Simponi Human anti-tnf mab certolizumab Cimzia Pegylated Fab frag humanized mab IBD, RA, AS, PsA, plaque Ps 1998 RA, AS, JIA, PsA, plaque ps 2008 IBD, RA, AS, JIL, PsA, plaque ps, HS, uv UC, RA, AS, JIA, PsA 2008 CD, RA, AS, PsA Other agents Drug Brand Mechanism Approval Indications rituximab Rituxan Anti CD RA, lymphoma, CLL, polyangiitis anakira Kineret IL-1 rec.antagonist 2001 RA abatacept Orancia CTLA-4 ligand, select 2005 RA Tcell costim. ustekinumab Stelara Ab anti IL-12,IL psoriasis tocilizumab Acterma Ab anti-il-6 reeptor 2010 RA, JIA belimumab Benlista Ab anti B-cell act. fact 2011 SLE tofacinib Xelianj JK inhibitor 2012 RA 4
5 - Risk of infections with use of TNF alpha inhibitors increased risk for granulomatous inf. TB (reactivation) theoretically increase susceptibility to intracellular pathogen- bacteria (Listeria, Salmonella spp.) viruses (HBV, VZV, JC) may cause neutropenia- increase IFI dependent on agent Clinical trials not powered to detect adverse events such as rare infections high rate of co-morbidities in patients immunosuppressant use associated with disease severity (confounding) patients receiving many immuno-supressants (impact of prednisone?) Case 38 yo man with severe IBD, currently on po steroids, is being evaluated for biologic therapy. A TST was 11 mm and a Quantiferon test is borderline. Patient is a healthcare worker and states he has been always allergic to PPD and CXR yearly was negative. He was told that BCG he received in his country of origin would make PPD positive for ever. What do you recommend? 5
6 LTBI and TNF alpha inhibitor use What is the best strategy for LTBI screening? Single step TST followed by ELISA based IGRA Is the systematic retesting needed while on anti TNF? Not if risk is low Unclear in highly endemic area- yes in RA, annually, if ongoing or future risk (21-29% converts at least one LTBI screening assay during the first year of therapy - but no active TB noted) When do we start biologics after initiation of LTBI treatment? At least a month Sing JA et al, ACR, Arthritis care 2012 Hatzara C et al, AnnRheum Dis 2015 Cantini F, et al, Autoimmun Rev (2015) LTBI and TNF alpha inhibitor use LTBI- 4x increase in risk of developing active TB Screening and treatment LTBI reduced but not completely eliminated risk of reactivation -age, comorbidities, previous/concurrent DMARDs -cumulative steroid use* -disease itself (RA 2-8.9x; PsAx 3.1, AS x3.9) -defective/incomplete compliance with guidelines 7x -etarnecept has the lowest risk -IBD 2-2.5xrisk of developing active TB 6
7 Case 56 y.o. male with severe seropositive RF, nodular RA with vasculitis 2009 Abnormal CT lung small caseating granuloma in his lung by bx; no cultures sent!; not felt to be caused by TB On Humira since 2012 with dramatic improvement in symptoms; 1/2017: Weight loss, poor appetite, night sweats, cough, dyspnea 2/2017: T spot positive. Sputum sent, BAL; 1/3 +M.fortuitum. 3 small nodules RUL on CT chest 2/2017: Candida esophagitis -responded splendidly to fluconazole 3/16/2017: 2/3 M. fortuitum Case points? Do we treat LTBI (positive T-spot)? Do we consider M. fortuitum pathogenic? Do we restart biologic (poor QOL)? 7
8 Case (con t) 3/30/2017: seen, prescribed Bactrim, moxifloxacin and tedizolid ; INH/B6 6/22/2017: doing well, completely quit smoking; minimal cough, no hemoptysis; main issue is lower back pain; wants surgery for QOL. 6/30/2017: AFB + in broth week 1, smear negative, result reported 7/19/2017 M. fortuitum 9/2017 :AFB+ 1/3 M. fortuitum, growth week 2- broth. Smear negative 7/24/2017: CT chest: 2/3 nodules stable, one mildly improved; quit smoking 8/23/2017: sputum 3/3 growth, smear negative but 3/3 MAC! 9/18/2017: had surgery of the lumbar spine, with improvement in QOL, pain Other case points? Changed to azithromycin/ethambutol/rifampin M. fortuitum?... Had spine surgery- when do we restart the biologic? For how long do we treat? 8
9 The Nontuberculous Mycobacteria and TNF alpha inhibitors Incidence of TB (per 100,000 p-years) Incidence of NTM (per 100,000 p-years) Relative Risk (NTM/TB) All Anti-TNF Users 49 (95% CI 18-79) 74 (95% CI ) 1.5 Anti-TNF >59 y/o 64 (95% CI ) 118 (95% CI ) 1.8 RA Anti-TNF Users 56 (95% CI ) 112 (95% CI ) patients Winthrop et al., Ann Rheum Dis 2013;72:37-42 Other serious bacterial infections Occur early (<6 months) after staring anti-tnf 2-14/100 person years Probably an increase of 2-3 infections/100 person-years with a biologic (relative increase of %) compared to non-biologic DMARDS monoclonals (ada/inflix)- more risky versus etanercept/abatacept underlying disease confers an increased risk for specific infection (septic arthritis in RA, intra-abdominal abscess in IBD) 2015: 2x risk of serious infections 2016: in IBD no increased risk; it seems CD activity index and steroids/dmadrs are more important! Steroids and co-morbidities important contributors Singh JA et al, Lancet 2015 Bonovas et al, Clin. Gastroenterology 2016 Osterman et al, Am J. Gastroenterology 2016 Baddley et al,
10 Fungal infections and TNF alpha inhibitors Numerous case reports Meta-analysis (RA): -only 9 cases of non-pjp IFI (5 IA)*; no increased risk of IFI with biologics Infections occur within 1-6 months of starting the biologic Advanced disease; high morbidity and mortality (FDA black box warning) Pneumocystosis (0.5/100,000) Histoplasmosis (2-19/100,000) Coccidioidomycosis (1-5/100,000) French study : /100,000 p-yrs (PJP, crypto, Aspergillus) 41 non-tb OIs were identified Risk factors for OIs: steroid therapy (>10mg/d) and monoclonal TNF antibodies Kourbeti et al., Clin Infect Dis 2014 Salmon-Ceron et al., Ann Rheum Dis ,309 pt screened 33, 324 TNF 80 OIs 16 PJP Crude rate (/1000 p-yrs) Adjusted Rate Comparator 1.7 (1.2, 2.5) ref TNF users 2.7 (2.2, 3.4) 1.6 (1.0, 2.6) Baseline steroids 2.5 (1.5, 4.0) Baddley et al., Ann Rheum Dis
11 Management of IFIs Stop the biologic: beware of IRIS (42% in Histoplasmosis study) Standard course of antifungal therapy Document clinical resolution of infection Re-start the biologic? Chronic suppressive therapy/secondary prophylaxis? Histoplasmosis: Restarted in 4/15; 1 had recurrence Coccidioidomycosis : 44 cases (9 disseminated), treated x9mo avg., restarted after 10 months, no recativation Candida : Candida infections in 4.7% on secukinimab (IL-17 pathway), greater than etanercept 1.2% (no need to stop therapy) Need more data regarding re-starting biologics and duration of antifungal therapy Hage, C. et al. Clin Infect Dis 2010;50:85-92 Olson et al BMC ID 2011 Taroumian et al., Arthritis Care and Research, 2012 Langley et al, NEJM 2014 Viral Infections and TNF alpha inhibitor use Reactivation of varicella-zoster virus Reactivation of hepatitis B HSV (severe cases) JC virus (PML)-rituximab WNV (1 case) Risk of EBV, CMV controversial (steroids*) HCV infection is not worsening on therapy Baddley, JW et al., CMI
12 Case 62 y.o woman with RA on tofacinib presents to her rheumatologist with 2 days of low grade fever and a rash. Did she need VZV vaccination? VZV infection and TNF inhibitors Year Published Source HR for VZV 2009 German, RABBIT registry US, Veterans Affairs French, RATIO registry US, 4 databases British, BSRBR database US, 4 databases, tofacinib (JAK i) 4.4 (if max +GC) Novasad and Winthrop, Clin Infect Dis 2014 Winthrop el al, Arthritis Rheumatol
13 Case 32 y.o man born in Vietnam is being treated for IBD. He failed few courses of DMARDs and is planned to start infliximab. Hepatitis B serology shows positive HBsAg, positive HBcAb, negative HBsAb. ALT, AST, bilirubin, INR, albumin are normal. Abdominal US is unremarkable. What is the next test you perform? If deciding to treat, what would you choose? HBV reactivation and biologics all rheumatic disease patients who are scheduled to start treatment with biological and/or non-biological DMARDs should receive screening for HBV infection. :HBsAg, anti-hbc, and anti-hbs If HBsAg + and DNA high- start antiviral If only isolated HBc Ab+- the data is not clear except rituximab 13
14 Risk of HVB reactivation Bessone F. et al, World Journal of Gatroenterorogy 2016 Conclusions Biologics are here to stay Biologics are associated with increased incidence of infections, and often with increased severity of disease Mycobacterial diseases, bacterial infections and zoster are the most important No benefit from antibacterial/antifungal/antiviral prophylaxis Age appropriate use of vaccines (inactive, some live vaccines) important Newer drugs will bring new challenges Baddley, JW, et al., ESCMID Study Group, Clin Microbiology and Infection
15 Thanks! if any questions 15
DISCLOSURES. Online A. Infectious Complications of Monoclonal Antibody Therapies 6/22/2012. Cytokine blocking. Lymphocyte depleting.
 Online A Infectious Complications of Monoclonal Antibody Therapies Steven M. Holland, M.D. Off-Label Usage None DISCLOSURES Financial Relationships with Relevant Commercial Interests None Resolution: N/A
Online A Infectious Complications of Monoclonal Antibody Therapies Steven M. Holland, M.D. Off-Label Usage None DISCLOSURES Financial Relationships with Relevant Commercial Interests None Resolution: N/A
Tuberculosis and Biologic Therapies: Risk and Prevention
 Tuberculosis and Biologic Therapies: Risk and Prevention Kevin L. Winthrop, MD, MPH Associate Professor, Divisions of Infectious Diseases, Public Health and Preventive Medicine Oregon Health & Science
Tuberculosis and Biologic Therapies: Risk and Prevention Kevin L. Winthrop, MD, MPH Associate Professor, Divisions of Infectious Diseases, Public Health and Preventive Medicine Oregon Health & Science
Center for Evidence-based Policy
 P&T Committee Brief Targeted Immune Modulators: Comparative Drug Class Review Alison Little, MD Center for Evidence-based Policy Oregon Health & Science University 3455 SW US Veterans Hospital Road, SN-4N
P&T Committee Brief Targeted Immune Modulators: Comparative Drug Class Review Alison Little, MD Center for Evidence-based Policy Oregon Health & Science University 3455 SW US Veterans Hospital Road, SN-4N
Cimzia. Cimzia (certolizumab pegol) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.11 Section: Prescription Drugs Effective Date: April 1, 2018 Subject: Cimzia Page: 1 of 5 Last Review
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.11 Section: Prescription Drugs Effective Date: April 1, 2018 Subject: Cimzia Page: 1 of 5 Last Review
Regulatory Status FDA-approved indication: Orencia is a selective T cell costimulation modulator indicated for: (1)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.18 Subject: Orencia Page: 1 of 8 Last Review Date: March 16, 2018 Orencia Description Orencia (abatacept)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.18 Subject: Orencia Page: 1 of 8 Last Review Date: March 16, 2018 Orencia Description Orencia (abatacept)
Tuberculosis and TNF Inhibitors
 Tuberculosis and TNF Inhibitors Sundari Mase MD, MPH Medical Team Lead CDC/DTBE/FSEB January 18, 2011 Objectives Discuss the association between and epidemiology of TNF inhibitors and TB Discuss the challenges
Tuberculosis and TNF Inhibitors Sundari Mase MD, MPH Medical Team Lead CDC/DTBE/FSEB January 18, 2011 Objectives Discuss the association between and epidemiology of TNF inhibitors and TB Discuss the challenges
Cimzia. Cimzia (certolizumab pegol) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Cimzia Page: 1 of 5 Last Review Date: March 17, 2017 Cimzia Description Cimzia (certolizumab pegol)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Cimzia Page: 1 of 5 Last Review Date: March 17, 2017 Cimzia Description Cimzia (certolizumab pegol)
Lecture: Rheumatology Update on DMARDs in Common Rheumatologic Diseases for the Family Physician. Sco Jay Harris, DO, FACFR
 Lecture: Rheumatology Update on DMARDs in Common Rheumatologic Diseases for the Family Physician Sco Jay Harris, DO, FACFR UPDATE ON DMARDS, BIOLOGICALS, CYTOKINE INHIBITORS/B CELL DEPLETIONS/CO-STIMULATION
Lecture: Rheumatology Update on DMARDs in Common Rheumatologic Diseases for the Family Physician Sco Jay Harris, DO, FACFR UPDATE ON DMARDS, BIOLOGICALS, CYTOKINE INHIBITORS/B CELL DEPLETIONS/CO-STIMULATION
Remicade and Friends What You Need to Know Treating the Patient on TNF-alpha Inhibitors and Related Meds
 Remicade and Friends What You Need to Know Treating the Patient on TNF-alpha Inhibitors and Related Meds Scott Stienecker MD FACP FSHEA Medical Director for Epidemiology and Infection Prevention Parkview
Remicade and Friends What You Need to Know Treating the Patient on TNF-alpha Inhibitors and Related Meds Scott Stienecker MD FACP FSHEA Medical Director for Epidemiology and Infection Prevention Parkview
5/9/2015. Disclosures. Infection in the (non-hiv) Immunocompromised Patient. Outline. Case #1. None. Critical Care Medicine May 2015
 Disclosures None Infection in the (non-hiv) Immunocompromised Patient Critical Care Medicine May 2015 Jennifer Babik, MD, PhD Assistant Clinical Professor Division of Infectious Diseases, UCSF Outline
Disclosures None Infection in the (non-hiv) Immunocompromised Patient Critical Care Medicine May 2015 Jennifer Babik, MD, PhD Assistant Clinical Professor Division of Infectious Diseases, UCSF Outline
Cimzia. Cimzia (certolizumab pegol) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.11 Subject: Cimzia Page: 1 of 5 Last Review Date: December 8, 2017 Cimzia Description Cimzia (certolizumab
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.11 Subject: Cimzia Page: 1 of 5 Last Review Date: December 8, 2017 Cimzia Description Cimzia (certolizumab
Regulatory Status FDA- approved indication: Simponi and Simponi ARIA are tumor necrosis factor (TNF) blockers indicated for the treatment of: (2-3)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.51 Subject: Simponi / Simponi ARIA Page: 1 of 9 Last Review Date: March 16, 2018 Simponi / Simponi
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.51 Subject: Simponi / Simponi ARIA Page: 1 of 9 Last Review Date: March 16, 2018 Simponi / Simponi
MEDICATION GUIDE XELJANZ (ZEL JANS ) (tofacitinib)
 MEDICATION GUIDE XELJANZ (ZEL JANS ) (tofacitinib) Read this Medication Guide before you start taking XELJANZ and each time you get a refill. There may be new information. This Medication Guide does not
MEDICATION GUIDE XELJANZ (ZEL JANS ) (tofacitinib) Read this Medication Guide before you start taking XELJANZ and each time you get a refill. There may be new information. This Medication Guide does not
8/11/2015. Febrile neutropenia Bone marrow transplant Immunosuppressant medications
 Dean Van Loo Pharm.D. Febrile neutropenia Bone marrow transplant Immunosuppressant medications Steroids Biologics Antineoplastic Most data from cancer chemotherapy Bone marrow suppression Fever is the
Dean Van Loo Pharm.D. Febrile neutropenia Bone marrow transplant Immunosuppressant medications Steroids Biologics Antineoplastic Most data from cancer chemotherapy Bone marrow suppression Fever is the
MEDICATION GUIDE. (tocilizumab)
 MEDICATION GUIDE ACTEMRA (AC-TEM-RA) (tocilizumab) Read this Medication Guide before you start ACTEMRA and before each infusion. There may be new information. This Medication Guide does not take the place
MEDICATION GUIDE ACTEMRA (AC-TEM-RA) (tocilizumab) Read this Medication Guide before you start ACTEMRA and before each infusion. There may be new information. This Medication Guide does not take the place
ESCMID Online Lecture Library. by author
 Therapies or diseases predisposing to infection Congenital immunodeficiences Acquired conditions Common variable immunedeficiency particular diseases (e.g. HIV, cancer,leukemia, lymphoma, diabetes, cystic
Therapies or diseases predisposing to infection Congenital immunodeficiences Acquired conditions Common variable immunedeficiency particular diseases (e.g. HIV, cancer,leukemia, lymphoma, diabetes, cystic
Infectious and Other Complications of New Immunobiologic Agents Used by HIV-Infected Individuals
 Infectious and Other Complications of New Immunobiologic Agents Used by HIV-Infected Individuals Peter Chin-Hong, MD Professor of Medicine University of California San Francisco San Francisco, CA Learning
Infectious and Other Complications of New Immunobiologic Agents Used by HIV-Infected Individuals Peter Chin-Hong, MD Professor of Medicine University of California San Francisco San Francisco, CA Learning
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: golimumab_simponi 8/2013 2/2018 2/2019 3/2018 Description of Procedure or Service Golimumab (Simponi and
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: golimumab_simponi 8/2013 2/2018 2/2019 3/2018 Description of Procedure or Service Golimumab (Simponi and
Infectious Complications of. Biologic Therapy
 Infectious Complications of Biologic Therapy Nicholas Haddad, MD, FIDSA, CIC Friday November 29, 2013 12th Annual Congress of the Lebanese Society of Gastroenterology Generic Issues Overall, quite safe
Infectious Complications of Biologic Therapy Nicholas Haddad, MD, FIDSA, CIC Friday November 29, 2013 12th Annual Congress of the Lebanese Society of Gastroenterology Generic Issues Overall, quite safe
abstract CLINICAL REPORT Guidance for the Clinician in Rendering Pediatric Care
 CLINICAL REPORT Guidance for the Clinician in Rendering Pediatric Care Infectious Complications With the Use of Biologic Response Modifiers in Infants and Children H. Dele Davies, MD, FAAP, COMMITTEE ON
CLINICAL REPORT Guidance for the Clinician in Rendering Pediatric Care Infectious Complications With the Use of Biologic Response Modifiers in Infants and Children H. Dele Davies, MD, FAAP, COMMITTEE ON
Infectious and Other Complications of Immunobiologic Agents Used by Individuals With HIV Infection
 Infectious and Other Complications of Immunobiologic Agents Used by Individuals With HIV Infection Peter Chin-Hong, MD Professor of Medicine University of California San Francisco San Francisco, California
Infectious and Other Complications of Immunobiologic Agents Used by Individuals With HIV Infection Peter Chin-Hong, MD Professor of Medicine University of California San Francisco San Francisco, California
Opportunistic Infections and Tuberculosis Risk Management
 Opportunistic Infections and Tuberculosis Risk Management Kevin L. Winthrop, MD, MPH Associate Professor, Divisions of Infectious Diseases, Public Health, and Preventive Medicine Oregon Health & Science
Opportunistic Infections and Tuberculosis Risk Management Kevin L. Winthrop, MD, MPH Associate Professor, Divisions of Infectious Diseases, Public Health, and Preventive Medicine Oregon Health & Science
Anti-TNF medication and tuberculosis
 Anti-TNF medication and tuberculosis Marleen Bakker Amsterdam 23-09-14 Contents Why? Who? How? How? - 1 - testing + interpretation - 2 treatment Why? Blocking TNF has major effect on immune-mediated inflammatory
Anti-TNF medication and tuberculosis Marleen Bakker Amsterdam 23-09-14 Contents Why? Who? How? How? - 1 - testing + interpretation - 2 treatment Why? Blocking TNF has major effect on immune-mediated inflammatory
TUBERCULOSIS AND THE TNF-α INHIBITORS. Lloyd Friedman, M.D. Yale University Milford Hospital
 TUBERCULOSIS AND THE TNF-α INHIBITORS Lloyd Friedman, M.D. Yale University Milford Hospital Outline TNF-α Anti-TNF-α medications Rates of tuberculosis Lower rates with etanercept Screening for latent tuberculosis
TUBERCULOSIS AND THE TNF-α INHIBITORS Lloyd Friedman, M.D. Yale University Milford Hospital Outline TNF-α Anti-TNF-α medications Rates of tuberculosis Lower rates with etanercept Screening for latent tuberculosis
INFLIXIMAB Remicade (infliximab), Inflectra (infliximab-dyyb), Ixifi* (infliximabqbtx), Renflexis (infliximab-abda)
 Pre - PA Allowance None Prior-Approval Requirements Diagnoses Patient must have ONE of the following: 6 years of age or older 1. Moderate to severe Crohn s disease (CD) a. Patient has fistulizing disease
Pre - PA Allowance None Prior-Approval Requirements Diagnoses Patient must have ONE of the following: 6 years of age or older 1. Moderate to severe Crohn s disease (CD) a. Patient has fistulizing disease
HIV medicine in the era of immune modulators
 HIV medicine in the era of immune modulators Peter Chin-Hong MD Professor of Medicine UCSF Disclosures Research support from Karius December 2018 Learning objectives HIV-infected patients living longer
HIV medicine in the era of immune modulators Peter Chin-Hong MD Professor of Medicine UCSF Disclosures Research support from Karius December 2018 Learning objectives HIV-infected patients living longer
Stelara. Stelara (ustekinumab) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.90.04 Subject: Stelara Page: 1 of 9 Last Review Date: September 20, 2018 Stelara Description Stelara
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.90.04 Subject: Stelara Page: 1 of 9 Last Review Date: September 20, 2018 Stelara Description Stelara
2019 COLLECTION TYPE: MIPS CLINICAL QUALITY MEASURES (CQMS) MEASURE TYPE: Process
 Quality ID #337: Psoriasis: Tuberculosis (TB) Prevention for Patients with Psoriasis, Psoriatic Arthritis and Rheumatoid Arthritis on a Biological Immune Response Modifier National Quality Strategy Domain:
Quality ID #337: Psoriasis: Tuberculosis (TB) Prevention for Patients with Psoriasis, Psoriatic Arthritis and Rheumatoid Arthritis on a Biological Immune Response Modifier National Quality Strategy Domain:
Xeljanz. Xeljanz, Xeljanz XR (tofacitinib) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.24 Subject: Xeljanz Page: 1 of 6 Last Review Date: March 16, 2018 Xeljanz Description Xeljanz, Xeljanz
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.24 Subject: Xeljanz Page: 1 of 6 Last Review Date: March 16, 2018 Xeljanz Description Xeljanz, Xeljanz
Proliferation of Medications
 Proliferation of Medications Novel Biologic Therapies for Rheumatic Diseases: An Overview Jonathan Graf, MD Professor of Clinical Medicine, UCSF Division of Rheumatology San Francisco General Hospital
Proliferation of Medications Novel Biologic Therapies for Rheumatic Diseases: An Overview Jonathan Graf, MD Professor of Clinical Medicine, UCSF Division of Rheumatology San Francisco General Hospital
Simponi / Simponi ARIA (golimumab)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.51 Subject: Simponi / Simponi ARIA Page: 1 of 6 Last Review Date: September 15, 2016 Simponi / Simponi
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.51 Subject: Simponi / Simponi ARIA Page: 1 of 6 Last Review Date: September 15, 2016 Simponi / Simponi
2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY. MEASURE TYPE: Process
 Quality ID #337: Psoriasis: Tuberculosis (TB) Prevention for Patients with Psoriasis, Psoriatic Arthritis and Rheumatoid Arthritis Patients on a Biological Immune Response Modifier National Quality Strategy
Quality ID #337: Psoriasis: Tuberculosis (TB) Prevention for Patients with Psoriasis, Psoriatic Arthritis and Rheumatoid Arthritis Patients on a Biological Immune Response Modifier National Quality Strategy
MEDICATION GUIDE HUMIRA
 MEDICATION GUIDE HUMIRA (Hu-MARE-ah) (adalimumab) injection Read the Medication Guide that comes with HUMIRA before you start taking it and each time you get a refill. There may be new information. This
MEDICATION GUIDE HUMIRA (Hu-MARE-ah) (adalimumab) injection Read the Medication Guide that comes with HUMIRA before you start taking it and each time you get a refill. There may be new information. This
Cosentyx. Cosentyx (secukinumab) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.90.11 Subject: Cosentyx Page: 1 of 7 Last Review Date: September 20, 2018 Cosentyx Description Cosentyx
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.90.11 Subject: Cosentyx Page: 1 of 7 Last Review Date: September 20, 2018 Cosentyx Description Cosentyx
Remicade. Remicade (infliximab), Inflectra (infliximab-dyyb) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.02 Subsection: Gastrointestinal nts Original Policy Date: May 20, 2011 Subject: Remicade Page: 1 of
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.02 Subsection: Gastrointestinal nts Original Policy Date: May 20, 2011 Subject: Remicade Page: 1 of
o Your healthcare provider should test you for TB before starting CIMZIA.
 Medication Guide CIMZIA (CIM-zee-uh) (certolizumab pegol) lyophilized powder or solution for subcutaneous use Read the Medication Guide that comes with CIMZIA before you start using it, and before each
Medication Guide CIMZIA (CIM-zee-uh) (certolizumab pegol) lyophilized powder or solution for subcutaneous use Read the Medication Guide that comes with CIMZIA before you start using it, and before each
Infections and Biologics
 Overview Infections and Biologics James Galloway What is the risk of infection with biologics? Are some patients at greater risk? Are some drugs safer? Case scenario You recently commenced Judith, a 54
Overview Infections and Biologics James Galloway What is the risk of infection with biologics? Are some patients at greater risk? Are some drugs safer? Case scenario You recently commenced Judith, a 54
Regulatory Status FDA- approved indication: Simponi and Simponi ARIA are tumor necrosis factor (TNF) blockers indicated for the treatment of:
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.51 Subject: Simponi / Simponi ARIA Page: 1 of 8 Last Review Date: March 17, 2017 Simponi / Simponi
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.51 Subject: Simponi / Simponi ARIA Page: 1 of 8 Last Review Date: March 17, 2017 Simponi / Simponi
Regulatory Status FDA-approved indication: Humira and Amjevita are tumor necrosis factor (TNF) blockers indicated for the treatment of: (2-3)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.29 Subject: Humira Page: 1 of 10 Last Review Date: June 22, 2017 Humira Description Humira (adalimumab),
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.29 Subject: Humira Page: 1 of 10 Last Review Date: June 22, 2017 Humira Description Humira (adalimumab),
Annual Rheumatology & Therapeutics Review for Organizations & Societies
 Annual Rheumatology & Therapeutics Review for Organizations & Societies Opportunistic Infections and Tuberculosis Risk Management Learning Objectives Define the risks of TB and non-tb opportunistic infections,
Annual Rheumatology & Therapeutics Review for Organizations & Societies Opportunistic Infections and Tuberculosis Risk Management Learning Objectives Define the risks of TB and non-tb opportunistic infections,
Synoviocytes. Macrophage. B cell C H R O N I C. Neutrophil. Mast cell I N F L A M M A T I O N. Tissue cell. Endothelial cell. Th1/Th17 IL17 IL22
 DC IL12, IL23 chemokines, ECM, Co-stimulation IL17, IL22 IFNγ ± Macrophage peptidoglycan lipopolysaccharide heat shock proteins Th1/Th17 IL17 IL22 Cell contact, co-stimulation immune complexes acute phase
DC IL12, IL23 chemokines, ECM, Co-stimulation IL17, IL22 IFNγ ± Macrophage peptidoglycan lipopolysaccharide heat shock proteins Th1/Th17 IL17 IL22 Cell contact, co-stimulation immune complexes acute phase
Tuberculosis Tools: A Clinical Update
 Tuberculosis Tools: A Clinical Update CAPA Conference 2014 JoAnn Deasy, PA-C. MPH, DFAAPA jadeasy@sbcglobal.net Adjunct Faculty Touro PA Program Learning Objectives Outline the pathogenesis of active pulmonary
Tuberculosis Tools: A Clinical Update CAPA Conference 2014 JoAnn Deasy, PA-C. MPH, DFAAPA jadeasy@sbcglobal.net Adjunct Faculty Touro PA Program Learning Objectives Outline the pathogenesis of active pulmonary
The Medical Letter. on Drugs and Therapeutics
 The Medical Letter publications are protected by US and international copyright laws. Forwarding, copying or any other distribution of this material is strictly prohibited. For further information call:
The Medical Letter publications are protected by US and international copyright laws. Forwarding, copying or any other distribution of this material is strictly prohibited. For further information call:
Remicade. Remicade (infliximab), Inflectra (infliximab-dyyb) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Remicade Page: 1 of 9 Last Review Date: June 22, 2017 Remicade Description Remicade (infliximab),
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Remicade Page: 1 of 9 Last Review Date: June 22, 2017 Remicade Description Remicade (infliximab),
What prescribers need to know
 HUMIRA Citrate-free presentations in an Electronic Medical Record (EMR) What prescribers need to know 2 / This is your guide to identifying HUMIRA Citrate-free presentations in your Electronic Medical
HUMIRA Citrate-free presentations in an Electronic Medical Record (EMR) What prescribers need to know 2 / This is your guide to identifying HUMIRA Citrate-free presentations in your Electronic Medical
ORENCIA (oh-ren-see-ah) (abatacept)
 17.7 FDA-Approved Patient Labeling PATIENT INFORMATION ORENCIA (oh-ren-see-ah) (abatacept) Read this Patient Information before you start receiving ORENCIA and each time before you are scheduled to receive
17.7 FDA-Approved Patient Labeling PATIENT INFORMATION ORENCIA (oh-ren-see-ah) (abatacept) Read this Patient Information before you start receiving ORENCIA and each time before you are scheduled to receive
Drugs and Applicable Coding: J-code: Enbrel-J1438; Humira-J0135; Remicade-J1745; Inflectra-Q5102; Cimzia-J0718; Simponi-J1602 Renflexis - pending
 Policy Subject: Anti-TNF Agents Policy Number: SHS PBD16 Category: Rheumatology & Autoimmune Policy Type: Medical Pharmacy Department: Pharmacy Product (check all that apply): Group HMO/POS Individual
Policy Subject: Anti-TNF Agents Policy Number: SHS PBD16 Category: Rheumatology & Autoimmune Policy Type: Medical Pharmacy Department: Pharmacy Product (check all that apply): Group HMO/POS Individual
Coccidioidomycosis in Rheumatologic Patients. Susan E. Hoover, MD
 Coccidioidomycosis in Rheumatologic Patients Susan E. Hoover, MD The problem Prevalence of rheumatoid arthritis is 1% Population of Arizona is 6 million At least 150,000 new cocci infections yearly DMARDs
Coccidioidomycosis in Rheumatologic Patients Susan E. Hoover, MD The problem Prevalence of rheumatoid arthritis is 1% Population of Arizona is 6 million At least 150,000 new cocci infections yearly DMARDs
2017 PERIOPERATIVE MEDICINE SYMPOSIUM Peri-operative use of immunosuppression in rheumatology patients
 2017 PERIOPERATIVE MEDICINE SYMPOSIUM Peri-operative use of immunosuppression in rheumatology patients Dr Alberta Hoi Rheumatologist MBBS, FRACP, PhD NEW ERA IN MUSCULOSKELETAL MEDICINE New drugs - Biologics,
2017 PERIOPERATIVE MEDICINE SYMPOSIUM Peri-operative use of immunosuppression in rheumatology patients Dr Alberta Hoi Rheumatologist MBBS, FRACP, PhD NEW ERA IN MUSCULOSKELETAL MEDICINE New drugs - Biologics,
Practical RA Treatment: James R. O Dell, M.D. University of Nebraska Medical Center May 24, 2014
 Practical RA Treatment: 2014 James R. O Dell, M.D. University of Nebraska Medical Center May 24, 2014 Disclosures James R. O Dell PI of Multinational RA trial supported by VA and NIH (NIAMS) that receives
Practical RA Treatment: 2014 James R. O Dell, M.D. University of Nebraska Medical Center May 24, 2014 Disclosures James R. O Dell PI of Multinational RA trial supported by VA and NIH (NIAMS) that receives
HELPING YOU AND YOUR PATIENTS TALK OPENLY ABOUT MODERATELY TO SEVERELY ACTIVE RA
 SIMPONI ARIA (golimumab) is indicated for the treatment of adults with moderately to severely active rheumatoid arthritis (RA) in combination with MTX, active psoriatic arthritis, and active ankylosing
SIMPONI ARIA (golimumab) is indicated for the treatment of adults with moderately to severely active rheumatoid arthritis (RA) in combination with MTX, active psoriatic arthritis, and active ankylosing
Northwestern Polytechnic University
 Clinical Tuberculosis Assessment by Health Care Provider Clinicians should review and verify the information in the Tuberculosis (TB) Screening Questionnaire (attached). Persons answering YES to any questions
Clinical Tuberculosis Assessment by Health Care Provider Clinicians should review and verify the information in the Tuberculosis (TB) Screening Questionnaire (attached). Persons answering YES to any questions
TB: Management in an era of multiple drug resistance. Bob Belknap M.D. Denver Public Health November 2012
 TB: Management in an era of multiple drug resistance Bob Belknap M.D. Denver Public Health November 2012 Objectives: 1. Explain the steps for diagnosing latent and active TB role of interferon-gamma release
TB: Management in an era of multiple drug resistance Bob Belknap M.D. Denver Public Health November 2012 Objectives: 1. Explain the steps for diagnosing latent and active TB role of interferon-gamma release
Stelara. Stelara (ustekinumab) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.90.04 Subject: Stelara Page: 1 of 6 Last Review Date: December 8, 2017 Stelara Description Stelara (ustekinumab)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.90.04 Subject: Stelara Page: 1 of 6 Last Review Date: December 8, 2017 Stelara Description Stelara (ustekinumab)
Get an Insurance Benefits Review for ORENCIA (abatacept)
 Get an Insurance Benefits Review for ORENCIA (abatacept) If you and your doctor decide that ORENCIA is right for you, you can call 1-800-ORENCIA (1-800-673-6242) to do an Insurance Benefits Review. Our
Get an Insurance Benefits Review for ORENCIA (abatacept) If you and your doctor decide that ORENCIA is right for you, you can call 1-800-ORENCIA (1-800-673-6242) to do an Insurance Benefits Review. Our
Specialty Pharmacies: What they are. Why they are different.
 Specialty Pharmacies: What they are. Why they are different. A changing environment of medicines and pharmacies. Filling a prescription used to be as simple as going to the corner drug store. Today, the
Specialty Pharmacies: What they are. Why they are different. A changing environment of medicines and pharmacies. Filling a prescription used to be as simple as going to the corner drug store. Today, the
LTBI Treatment and Anti TNF alpha
 LTBI Treatment and Anti TNF alpha Therapy Julie Higashi, MD PhD Director, TB Control Section San Francisco Department of Public Health TNF alpha is important for the immune response against TB Macrophages
LTBI Treatment and Anti TNF alpha Therapy Julie Higashi, MD PhD Director, TB Control Section San Francisco Department of Public Health TNF alpha is important for the immune response against TB Macrophages
RHEUMATOID ARTHRITIS DRUGS
 Rheumatology Biologics Criteria from the Exceptional Access Program RHEUMATOID ARTHRITIS DRUGS DRUG NAME BRS REIMBURSED DOSAGE FORM/ STRENGTH Adalimumab Humira 40 mg/0.8 syringe and 40mg/0.8 pen for Anakinra
Rheumatology Biologics Criteria from the Exceptional Access Program RHEUMATOID ARTHRITIS DRUGS DRUG NAME BRS REIMBURSED DOSAGE FORM/ STRENGTH Adalimumab Humira 40 mg/0.8 syringe and 40mg/0.8 pen for Anakinra
CIMZIA (certolizumab pegol)
 Pre - PA Allowance None Prior-Approval Requirements Age Diagnoses 18 years of age or older Patient must have ONE of the following: 1. Moderate to severe Crohn s Disease (CD) a. Inadequate response, intolerance
Pre - PA Allowance None Prior-Approval Requirements Age Diagnoses 18 years of age or older Patient must have ONE of the following: 1. Moderate to severe Crohn s Disease (CD) a. Inadequate response, intolerance
Tuberculosis Update. Topics to be Addressed
 Tuberculosis Update Robert M. Jasmer, M.D. University of California, San Francisco TB Control Section, San Francisco Department of Public Health Topics to be Addressed TB in the USA Screening recommendations
Tuberculosis Update Robert M. Jasmer, M.D. University of California, San Francisco TB Control Section, San Francisco Department of Public Health Topics to be Addressed TB in the USA Screening recommendations
Infection in the (non HIV) Immunocompromised Host
 Infection in the (non HIV) Immunocompromised Host No financial relationships to disclose Brian S. Schwartz, MD UCSF, Division of Infectious Diseases Lecture outline Background/why is this topic important?
Infection in the (non HIV) Immunocompromised Host No financial relationships to disclose Brian S. Schwartz, MD UCSF, Division of Infectious Diseases Lecture outline Background/why is this topic important?
Regulatory Status FDA-approved indication: Orencia is a selective T cell co-stimulation modulator indicated for: (1)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Orencia Page: 1 of 9 Last Review Date: September 20, 2018 Orencia Description Orencia (abatacept)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Orencia Page: 1 of 9 Last Review Date: September 20, 2018 Orencia Description Orencia (abatacept)
Cyltezo (adalimumab-adbm) CG-DRUG-64, CG-DRUG-65
 Market DC Cyltezo (adalimumab-adbm) CG-DRUG-64, CG-DRUG-65 Override(s) Prior Authorization Quantity Limit Medications Cyltezo (adalimumab-adbm) 40 mg/0.8 ml prefilled syringe #* ^ Approval Duration 1 year
Market DC Cyltezo (adalimumab-adbm) CG-DRUG-64, CG-DRUG-65 Override(s) Prior Authorization Quantity Limit Medications Cyltezo (adalimumab-adbm) 40 mg/0.8 ml prefilled syringe #* ^ Approval Duration 1 year
Otezla. Otezla (apremilast) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Otezla Page: 1 of 5 Last Review Date: March 16, 2018 Otezla Description Otezla (apremilast) Background
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Otezla Page: 1 of 5 Last Review Date: March 16, 2018 Otezla Description Otezla (apremilast) Background
Drug Class Prior Authorization Criteria Therapeutic Agents in Rheumatic and Inflammatory Diseases
 Drug Class Prior Authorization Criteria Therapeutic Agents in Rheumatic and Inflammatory Diseases Line of Business: Medicaid P & T Approval Date: August 16, 2017 Effective Date: August 16, 2017 This policy
Drug Class Prior Authorization Criteria Therapeutic Agents in Rheumatic and Inflammatory Diseases Line of Business: Medicaid P & T Approval Date: August 16, 2017 Effective Date: August 16, 2017 This policy
The ORENCIA (abatacept) JIA Observational Registry
 Moderate to Severe Juvenile Idiopathic Arthritis (JIA) Injection for Intravenous Use Injection for Subcutaneous Use The ORENCIA (abatacept) JIA Observational Registry Injection for Intravenous Use Injection
Moderate to Severe Juvenile Idiopathic Arthritis (JIA) Injection for Intravenous Use Injection for Subcutaneous Use The ORENCIA (abatacept) JIA Observational Registry Injection for Intravenous Use Injection
Steps TO HELP YOU TALK WITH YOUR DOCTOR ABOUT YOUR TREATMENT OPTIONS
 Moderate to Severe Rheumatoid Arthritis (RA) 3 Steps TO HELP YOU TALK WITH YOUR DOCTOR ABOUT YOUR TREATMENT OPTIONS INDICATION/USAGE Adult Rheumatoid Arthritis (RA): ORENCIA (abatacept) is a prescription
Moderate to Severe Rheumatoid Arthritis (RA) 3 Steps TO HELP YOU TALK WITH YOUR DOCTOR ABOUT YOUR TREATMENT OPTIONS INDICATION/USAGE Adult Rheumatoid Arthritis (RA): ORENCIA (abatacept) is a prescription
Inflectra (infliximab-dyyb), Remicade (infliximab), Renflexis (infliximab-abda) DRUG CG-DRUG-64
 Inflectra (infliximab-dyyb), Remicade (infliximab), Renflexis (infliximab-abda) DRUG.00002 CG-DRUG-64 Override(s) Prior Authorization *Washington Medicaid See State Specific Mandates Medications Inflectra
Inflectra (infliximab-dyyb), Remicade (infliximab), Renflexis (infliximab-abda) DRUG.00002 CG-DRUG-64 Override(s) Prior Authorization *Washington Medicaid See State Specific Mandates Medications Inflectra
Medication Guide Enbrel (en-brel) (etanercept)
 Medication Guide Enbrel (en-brel) (etanercept) Read the Medication Guide that comes with Enbrel before you start using it and each time you get a refill. There may be new information. This Medication Guide
Medication Guide Enbrel (en-brel) (etanercept) Read the Medication Guide that comes with Enbrel before you start using it and each time you get a refill. There may be new information. This Medication Guide
Pathogenesis of Rheumatoid Arthritis. Smolen, J. S. et al. (2012) Nat. Rev. Rheumatol. doi: /nrrheum
 1 Pathogenesis of Rheumatoid Arthritis 2 Smolen, J. S. et al. (2012) Nat. Rev. Rheumatol. doi:10.1038/nrrheum 2012.23 Biologic therapies used in the treatment of rheumatoid arthritis 3 Etanercept Infliximab
1 Pathogenesis of Rheumatoid Arthritis 2 Smolen, J. S. et al. (2012) Nat. Rev. Rheumatol. doi:10.1038/nrrheum 2012.23 Biologic therapies used in the treatment of rheumatoid arthritis 3 Etanercept Infliximab
Cell-Mediated Immunity and T Lymphocytes
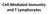 Cell-Mediated Immunity and T Lymphocytes T helper (Th) Cells Peripheral lymhoid tissue thymus Lymphoid stem cell CD8+ CD4+ CD4+ Treg CD8+ CTL + antigen Cytotoxic T lymphocyte CD4+ Th Helper T cell CD4+
Cell-Mediated Immunity and T Lymphocytes T helper (Th) Cells Peripheral lymhoid tissue thymus Lymphoid stem cell CD8+ CD4+ CD4+ Treg CD8+ CTL + antigen Cytotoxic T lymphocyte CD4+ Th Helper T cell CD4+
KEEP UP THE FIGHT WITH. Little Victories
 FOR MODERATE TO SEVERE RHEUMATOID ARTHRITIS (RA) KEEP UP THE FIGHT WITH Little Victories Small improvements in your daily activities may be a sign that ORENCIA (abatacept) is working for you. Use this
FOR MODERATE TO SEVERE RHEUMATOID ARTHRITIS (RA) KEEP UP THE FIGHT WITH Little Victories Small improvements in your daily activities may be a sign that ORENCIA (abatacept) is working for you. Use this
Remicade (infliximab) DRUG.00002
 Applicability/Effective Date *- Florida Healthy Kids Remicade (infliximab) DRUG.00002 Override(s) Prior Authorization Step Therapy Medications Remicade (infliximab) Approval Duration 1 year Comment Intravenous
Applicability/Effective Date *- Florida Healthy Kids Remicade (infliximab) DRUG.00002 Override(s) Prior Authorization Step Therapy Medications Remicade (infliximab) Approval Duration 1 year Comment Intravenous
Rheumatoid Arthritis. Improving Outcomes in RA: Three Pillars. RA: Chronic Joint Destruction and Disability What We Try to Prevent
 Rheumatoid Arthritis Modern Management of Common Problems in Rheumatology: Rheumatoid Arthritis Jonathan Graf, M.D. Associate Professor of Medicine, UCSF Division of Rheumatology, SFGH Director, UCSF Rheumatoid
Rheumatoid Arthritis Modern Management of Common Problems in Rheumatology: Rheumatoid Arthritis Jonathan Graf, M.D. Associate Professor of Medicine, UCSF Division of Rheumatology, SFGH Director, UCSF Rheumatoid
Rheumatoid Arthritis. Update in Rheumatoid Arthritis Diagnosis and Therapy. RA: Chronic Joint Destruction and Disability What We Try to Prevent
 Rheumatoid Arthritis Update in Rheumatoid Arthritis Diagnosis and Therapy Jonathan Graf, M.D. Associate Professor of Medicine, UCSF Division of Rheumatology, SFGH Director, UCSF Rheumatoid Arthritis Clinic
Rheumatoid Arthritis Update in Rheumatoid Arthritis Diagnosis and Therapy Jonathan Graf, M.D. Associate Professor of Medicine, UCSF Division of Rheumatology, SFGH Director, UCSF Rheumatoid Arthritis Clinic
Tuberculosis: update 2013
 Tuberculosis: update 2013 William R. Bishai, MD, PhD Center for TB Research Division of Infectious Diseases Department of Medicine Johns Hopkins School of Medicine Question 1 A TB speaker at a major conference
Tuberculosis: update 2013 William R. Bishai, MD, PhD Center for TB Research Division of Infectious Diseases Department of Medicine Johns Hopkins School of Medicine Question 1 A TB speaker at a major conference
Perioperative Medicine:
 Perioperative Medicine: Management of rheumatologic agents Divya Gollapudi, MD May 2016 Medical Operative Consult Clinic Harborview Medical Center Your patient Ms. L is a 55 year-old F w/ h/o RA who presents
Perioperative Medicine: Management of rheumatologic agents Divya Gollapudi, MD May 2016 Medical Operative Consult Clinic Harborview Medical Center Your patient Ms. L is a 55 year-old F w/ h/o RA who presents
Announcing HUMIRA. Psoriasis Starter Package
 Announcing HUMIRA (adalimumab) Psoriasis Starter Package HUMIRA is indicated for the treatment of adult patients with moderate to severe chronic plaque psoriasis who are candidates for systemic therapy
Announcing HUMIRA (adalimumab) Psoriasis Starter Package HUMIRA is indicated for the treatment of adult patients with moderate to severe chronic plaque psoriasis who are candidates for systemic therapy
The GPs role with biologics for immune-mediated inflammatory diseases David Gardner Rheumatologist GPCME 2015
 The GPs role with biologics for immune-mediated inflammatory diseases David Gardner Rheumatologist GPCME 2015 The views and opinions expressed in the following presentation are those of the presenter and
The GPs role with biologics for immune-mediated inflammatory diseases David Gardner Rheumatologist GPCME 2015 The views and opinions expressed in the following presentation are those of the presenter and
1 P a g e. Systemic Juvenile Idiopathic Arthritis (SJIA) (1.3) Patients 2 years of age and older with active systemic juvenile idiopathic arthritis.
 LENGTH OF AUTHORIZATION: Initial: 3 months for Crohn s or Ulcerative Colitis; 1 year for all other indications. Renewal: 1 year dependent upon medical records supporting response to therapy and review
LENGTH OF AUTHORIZATION: Initial: 3 months for Crohn s or Ulcerative Colitis; 1 year for all other indications. Renewal: 1 year dependent upon medical records supporting response to therapy and review
10/3/2017. Updates in Tuberculosis. Global Tuberculosis, WHO 2015 report. Objectives. Disclosures. I have nothing to disclose
 Disclosures Updates in Tuberculosis I have nothing to disclose Chris Keh, MD Assistant Clinical Professor, Division of Infectious Diseases, UCSF TB Controller, TB Prevention and Control Program, Population
Disclosures Updates in Tuberculosis I have nothing to disclose Chris Keh, MD Assistant Clinical Professor, Division of Infectious Diseases, UCSF TB Controller, TB Prevention and Control Program, Population
JUST FOR KIDS SELECTED IMPORTANT SAFETY INFORMATION
 JUST FOR KIDS For children ages 6-17 with moderately to severely active Crohn s disease or ulcerative colitis (UC) who haven t responded well to other therapies SELECTED IMPORTANT SAFETY INFORMATION REMICADE
JUST FOR KIDS For children ages 6-17 with moderately to severely active Crohn s disease or ulcerative colitis (UC) who haven t responded well to other therapies SELECTED IMPORTANT SAFETY INFORMATION REMICADE
Tuberculosis in Non-HIV Infected Immunosuppresed Hosts. TNFα inhibitors and beyond
 Tuberculosis in Non-HIV Infected Immunosuppresed Hosts TNFα inhibitors and beyond David E. Griffith, MD Assistant Medical Director Heartland National TB Center Professor of Medicine University of Texas
Tuberculosis in Non-HIV Infected Immunosuppresed Hosts TNFα inhibitors and beyond David E. Griffith, MD Assistant Medical Director Heartland National TB Center Professor of Medicine University of Texas
Canadian Society of Internal Medicine Annual Meeting 2016 Montreal, QC
 Canadian Society of Internal Medicine Annual Meeting 2016 Montreal, QC Update on the Treatment of Rheumatoid Arthritis Sabrina Fallavollita MDCM McGill University Canadian Society of Internal Medicine
Canadian Society of Internal Medicine Annual Meeting 2016 Montreal, QC Update on the Treatment of Rheumatoid Arthritis Sabrina Fallavollita MDCM McGill University Canadian Society of Internal Medicine
Humira (adalimumab) DRUG.00002
 Humira (adalimumab) DRUG.00002 Override(s) Prior Authorization Quantity Limit Approval Duration 1 year Medications Humira 10 mg/0.2 ml syringe Humira pediatric Crohn s Disease starter pack 40 mg/0.8 ml
Humira (adalimumab) DRUG.00002 Override(s) Prior Authorization Quantity Limit Approval Duration 1 year Medications Humira 10 mg/0.2 ml syringe Humira pediatric Crohn s Disease starter pack 40 mg/0.8 ml
Jennifer Lam MPH candidate 2009 Johns Hopkins Bloomberg School of Public Health. Preceptors: Wendy Cronin, PhD MT(ASCP), Cathy Goldsborough, RN
 Jennifer Lam MPH candidate 2009 Johns Hopkins Bloomberg School of Public Health Preceptors: Wendy Cronin, PhD MT(ASCP), Cathy Goldsborough, RN Phase Symposium: May 6, 2009 Background & Rationale Maryland
Jennifer Lam MPH candidate 2009 Johns Hopkins Bloomberg School of Public Health Preceptors: Wendy Cronin, PhD MT(ASCP), Cathy Goldsborough, RN Phase Symposium: May 6, 2009 Background & Rationale Maryland
Basic Approach/Goals. Medical Management of Rheumatic Diseases. Acetaminophen. Anti-inflammatories (NSAIDs) Steroids.
 Basic Approach/Goals Medical Management of Rheumatic Diseases Diane Lacaille, Rheumatologist ACE Workshop April 23, 2018 1) Symptom control: Acetaminophen, NSAIDs, steroids (intraart, po) 2) Prevention
Basic Approach/Goals Medical Management of Rheumatic Diseases Diane Lacaille, Rheumatologist ACE Workshop April 23, 2018 1) Symptom control: Acetaminophen, NSAIDs, steroids (intraart, po) 2) Prevention
Summary of Risk Minimization Measures
 Table 6.1.4-1: Summary of Risk Minimization Measures Safety Concern Vaccination Hepatic and renal impairment Combination therapy Elderly Routine Risk Minimization Measures Specific subsection on vaccination
Table 6.1.4-1: Summary of Risk Minimization Measures Safety Concern Vaccination Hepatic and renal impairment Combination therapy Elderly Routine Risk Minimization Measures Specific subsection on vaccination
Biologics for Autoimmune Diseases
 Biologics for Autoimmune Diseases Goal(s): Restrict use of biologics to OHP funded conditions and according to OHP guidelines for use. Promote use that is consistent with national clinical practice guidelines
Biologics for Autoimmune Diseases Goal(s): Restrict use of biologics to OHP funded conditions and according to OHP guidelines for use. Promote use that is consistent with national clinical practice guidelines
Fml Limits. Azathioprine (Imuran) 50mg, 75mg, 100mg - $26.85 Cyclosporine, 25mg, 100mg. $ Leflunomide (Arava) 10mg Tablet - $144.
 MEDICATION COVERAGE POLICY PHARMACY AND THERAPEUTICS ADVISORY COMMITTEE POLICY: Rheumatoid Arthritis (RA) P&T DATE: 2/15/2017 CLASS: Rheumatology/Anti-inflammatory Disorders REVIEW HISTORY 2/16, 5/15,
MEDICATION COVERAGE POLICY PHARMACY AND THERAPEUTICS ADVISORY COMMITTEE POLICY: Rheumatoid Arthritis (RA) P&T DATE: 2/15/2017 CLASS: Rheumatology/Anti-inflammatory Disorders REVIEW HISTORY 2/16, 5/15,
Regulatory Status FDA-approved indication: Orencia is a selective T cell costimulation modulator indicated for: (1)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.18 Subject: Orencia Page: 1 of 6 Last Review Date: December 8, 2017 Orencia Description Orencia (abatacept)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.18 Subject: Orencia Page: 1 of 6 Last Review Date: December 8, 2017 Orencia Description Orencia (abatacept)
Rheumatoid Arthritis Update
 Rheumatoid Arthritis Update Beth Valashinas, DO, FACOI, FACR Disclosures Speaker for AbbVie Pharmaceuticals Learning Objectives Upon completion of this session, participants should be able to discuss:
Rheumatoid Arthritis Update Beth Valashinas, DO, FACOI, FACR Disclosures Speaker for AbbVie Pharmaceuticals Learning Objectives Upon completion of this session, participants should be able to discuss:
Cimzia (certolizumab pegol) Data Showed Broad and Rapid Relief From Burden of Symptoms In Rheumatoid Arthritis Patients
 Cimzia (certolizumab pegol) Data Showed Broad and Rapid Relief From Burden of Symptoms In Rheumatoid Arthritis Patients Rapid, sustained and clinically meaningful improvement in wideranging patient-reported
Cimzia (certolizumab pegol) Data Showed Broad and Rapid Relief From Burden of Symptoms In Rheumatoid Arthritis Patients Rapid, sustained and clinically meaningful improvement in wideranging patient-reported
Amjevita (adalimumab-atto) CG-DRUG-64, CG-DRUG-65
 Market DC Amjevita (adalimumab-atto) CG-DRUG-64, CG-DRUG-65 Override(s) Prior Authorization Quantity Limit Medications Amjevita 20 mg/0.4 ml prefilled syringe Amjevita (adalimumab-atto) 40 mg/0.8 ml 2
Market DC Amjevita (adalimumab-atto) CG-DRUG-64, CG-DRUG-65 Override(s) Prior Authorization Quantity Limit Medications Amjevita 20 mg/0.4 ml prefilled syringe Amjevita (adalimumab-atto) 40 mg/0.8 ml 2
Tuberculosis in Primary Care COC GTA Spring Symposium Dr Elizabeth Rea April 2013
 Tuberculosis in Primary Care COC GTA Spring Symposium Dr Elizabeth Rea April 2013 1 TB in Toronto - risk groups Diagnosis of active TB LTBI diagnosis and management Infection control 2 TB in Toronto Case
Tuberculosis in Primary Care COC GTA Spring Symposium Dr Elizabeth Rea April 2013 1 TB in Toronto - risk groups Diagnosis of active TB LTBI diagnosis and management Infection control 2 TB in Toronto Case
Infliximab/Infliximab-dyyb DRUG.00002
 Infliximab/Infliximab-dyyb DRUG.00002 Override(s) Prior Authorization Step Therapy Medications Remicade (infliximab) Inflectra (inflectra-dyyb) Approval Duration 1 year Comment Intravenous administration
Infliximab/Infliximab-dyyb DRUG.00002 Override(s) Prior Authorization Step Therapy Medications Remicade (infliximab) Inflectra (inflectra-dyyb) Approval Duration 1 year Comment Intravenous administration
Medication Policy Manual. Topic: Xeljanz, tofacitinib Date of Origin: January 21, 2013
 Medication Policy Manual Policy No: dru289 Topic: Xeljanz, tofacitinib Date of Origin: January 21, 2013 Committee Approval Date: January 19, 2015 Next Review Date: January 2016 Effective Date: April 1,
Medication Policy Manual Policy No: dru289 Topic: Xeljanz, tofacitinib Date of Origin: January 21, 2013 Committee Approval Date: January 19, 2015 Next Review Date: January 2016 Effective Date: April 1,
Corporate Medical Policy
 Corporate Medical Policy Rituximab for the Treatment of Rheumatoid Arthritis File Name: Origination: Last CAP Review: Next CAP Review: Last Review: rituximab_for_the_treatment_of_rheumatoid_arthritis 4/2008
Corporate Medical Policy Rituximab for the Treatment of Rheumatoid Arthritis File Name: Origination: Last CAP Review: Next CAP Review: Last Review: rituximab_for_the_treatment_of_rheumatoid_arthritis 4/2008
DENOMINATOR: All patients aged 18 and older with a diagnosis of inflammatory bowel disease
 Measure #274: Inflammatory Bowel Disease (IBD): Testing for Latent Tuberculosis (TB) Before Initiating Anti-TNF (Tumor Necrosis Factor) Therapy National Quality Strategy Domain: Effective Clinical Care
Measure #274: Inflammatory Bowel Disease (IBD): Testing for Latent Tuberculosis (TB) Before Initiating Anti-TNF (Tumor Necrosis Factor) Therapy National Quality Strategy Domain: Effective Clinical Care
