Comparison of Various Biological Agents in the Treatment of Psoriasis
|
|
|
- Oswald Lee
- 6 years ago
- Views:
Transcription
1 VOL.11NO.5 NO.9 MAY SEPTEMBER Comparison of Various Biological Agents in the Treatment of Psoriasis MBBS(HK), FRCP, FHKCP, FHKAM(Med) Yaumatei Dermatology Clinic, Social Hygiene Service. This article has been selected by the Editorial Board of the Hong Kong Medical Diary for participants in the CME programme of the Medical Council of Hong Kong (MCHK) to complete the following self-assessment questions in order to be awarded one CME credit under the programme upon returning the completed answer sheet to the Federation Secretariat on or before 30 September Introduction In recent years, there has been considerable breakthrough in understanding the pathogenesis of psoriasis as a T cell mediated autoimmune disease. In genetically predisposed individuals, a triggering antigen, the nature of which is still not clear, is captured by Langerhans' cells in the epidermis. The Langerhans' cell migrates into the regional lymph node and presents the antigen to the T lymphocyte. When the antigen- Langerhans' cell complex interacts with the T cell in the lymph node, the T lymphocyte is activated by two costimulatory signals 1 and 2. In predisposed individuals, these activated T cells undergo TH1 differentiation into effector memory T cells and selectively traffics back to the skin and releases inflammatory cytokines such as tumour necrosis factor (TNF) alpha. These cytokines act on the epidermal keratinocytes, dermal blood vessels and other inflammatory cells, producing inflammatory responses that result in clinical lesion of psoriasis. Biologic agents are synthetic proteins designed to block one or more of these specific steps in the pathogenetic pathway. Four targets are aimed: Target 1: Inhibiting T cell activation by blocking the co-stimulatory signal 2 (e.g. by efalizumab, alefacept); Target 2: Inducing immune deviation from TH1 differentiation to TH2 differentiation in the activated T cell; Target 3:Eliminating activated T cells selectively (e.g. by alefacept); Target 4: Inhibiting the action of cytokines, especially tumour necrosis factor (TNF) alpha (e.g. by infliximab, etanercept) Four of these new biologic agents, namely alefacept, efalizumab, infliximab and etanercept are discussed below. Comparison of their modes of actions, clinical uses, efficacy, side effects and precautions are summarized in Table 1. Alefacept In psoriasis, the lymphocyte function-associated antigen (LFA)-3 presents on the Langerhans' cell binds to the CD2 expressed on activated T cell, producing costimulating interaction (signal 2) and leads to T cell activation and a cascade of inflammatory response resulting in psoriasis. Alefacept is a recombinant human LFA-3/IgG1 dimeric fusion protein that binds to CD2 receptor expressed on these activated T cells, thus inhibiting T cell activation (Target 1) and proliferation. In addition, it also causes apoptosis and selective elimination of these pathogenic T cells (Target 3) while preserving the naive T cells. In January 2003, Alefacept was approved by FDA of USA for the treatment of moderate to severe plaque psoriasis in adult patients who are candidates for systemic therapy or phototherapy. It is administered weekly either by intramuscular injection of 15mg or by intravenous bolus injection of 7.5mg. A course of 12 weeks is usually recommended. In most clinical trials on efficacy of treatment for psoriasis, the widely adopted parameter is the PASI 75 which measures the percentage of patients achieving at least 75% reduction of their Psoriasis Area and Severity Index (PASI) score from the pretreatment baseline. In alefacept, PASI 75 was achieved in 33% of patients after weekly intramuscular injection for 12-week. 1 Similarly, 28% of patients achieved PASI 75 after a 12 week course of once weekly intravenous alefacept injection while a second course further enhanced clinical efficacy up to 40%. 2 Significant clinical response occurred at around eight weeks after starting treatment. 3 The best clinical efficacy occurred approximately six weeks after the last dose. 1 Comparing with other biologics, remission in the responders was long-lasting and at week 24, 24% of patients still remained clear or almost clear without additional therapy and 71% maintained at least PASI 50 ( i.e. 50% reduction of PASI as compared with baseline). 1 For those who needed retreatment, a median interval of 10 months (range: 6 to 18) between the last dose to subsequent retreatment was noted. 4 The most common side effects were pharyngitis, headache, rhinitis and dizziness. 1,2,3,4 Mild injection site reaction occurred more often in the intramuscular administration 1,2 while transient chill during the first day of treatment occurred more often in the intravenous route. One of the important side effects is the reduction of circulating T cell counts. Weekly CD4 T lymphocyte 13
2 count monitoring is recommended during treatment. Alefacept should be withheld if the CD4 T lymphocyte count is below 250 cells/ml and discontinued if it remains below 250 cells/ml for four weeks. Ten percent and 2% of patients discontinued intravenous alefacept temporarily and permanently because of low CD4 T lymphocyte count 2 while 4% and none when alefacept was given intramuscularly. 1 The rate of infection requiring hospitalization was 0.9%. Up to date, alefacept has a satisfactory safety profile. Efalizumab Efalizumab is a recombinant humanized monoclonal antibody against the alpha subunit of LFA-1(i.e. the CD11a) expressed on T lymphocytes. It inhibits the interaction between CD11a and the intercellular adhesion molecule (ICAM)-1 expressed on antigen presenting cells hence stopping the co-stimulatory signal 2 (Target 1) that activates the T cells which lead to a sequence of inflammatory events. Efalizumab was approved by FDA in October 2003 to treat moderate to severe plaque psoriasis in adult patients who are candidates for systemic therapy or phototherapy. It is usually given by subcutaneous injection of single first conditioning dose at 0.7mg/kg followed by weekly dose at 1mg/kg for a course of 12 weeks. Twenty-two to 29.2% versus 28 to 41% of patients achieved PASI 75 after 12 week treatment with dose of 1mg/kg versus 2mg/kg respectively. 5,6 Seventy-seven percent of these good responders maintained their improvement when additional 12 week treatment were given. 6 The improvement occurred as early as four weeks. 6 Study showed that additional clinical improvement could be achieved by prolonged treatment beyond 12 weeks. 5 Treatment up to 24 and 60 weeks, which were well tolerated, increased the percentage of patients achieving PASI 75 to 51% and 64% respectively. 5 However, when stopping treatment, significant clinical relapse is one of the major disadvantages and 0.7% of patients experienced rebound after discontinuation, suggesting long term maintenance therapy with efalizumab is necessary. 7 Common minor side effects such as flu-like symptoms occurred in 37% of patients during the first few injections. 8 It can be minimized by the use of reduced conditioning dose in the first injection. More serious side effect of thrombocytopenia was rarely reported. 7 Monitoring of platelet count is recommended every month initially and then three-monthly during the treatment period and treatment should be stopped if significant thrombocytopenia occurs. Infliximab Infliximab is a chimeric monoclonal antibody which binds to transmembrane and soluble TNF alpha. It binds to TNF specifically and blocks its interaction with cell surface receptors (Target 4). Thus, TNF alpha, which is an important mediator secreted by the activated pathogenic T cells, cannot bind with its natural receptors in various sites and the inflammatory response leading to psoriasis ceases. Infliximab is approved for the treatment of Crohn's disease and rheumatoid arthritis by FDA and is currently under review by the authority for other indications including psoriasis. The drug is usually given 3mg/kg to 5mg/kg VOL.11 NO.9 SEPTEMBER 2006 by intravenous infusion over two hours at week 0, 2, 6 then every 8 week if maintenance is necessary. Infliximab is highly efficacious and 72% versus 88% of patients achieved PASI 75 at week 10 after infusion at 3mg/kg versus 5mg/kg respectively. 9 Almost half of the responders achieved PASI 75 at 4 weeks. 9 Other studies showed that 40% and 73% of patients receiving 5mg/kg and 10mg/kg infliximab respectively were able to maintain at PASI 50 at week Hence, infliximab could produce a rapid, effective and sustainable effect in the treatment of psoriasis. Common side effects are mild including headache, pruritus, fatigue and myalgia. Five percent of patients may develop infusion reactions ranging from mild hypotension and allergic reactions to severe anaphylaxis and seizures which usually occur during or within two hours of infusion. 11 Serious opportunistic infections such as disseminated or extrapulmonary tuberculosis were reported. 11,12 Reactivation of latent tuberculosis may develop within the first three to six months after starting treatment. Pretreatment tuberculin test, chest x ray and anti-tuberculosis prophylaxis are recommended before the commencement of infliximab. 11,12 Infliximab is also contraindicated in patients with moderate to severe heart failure. Etanercept Etanercept is a dimeric fusion protein consisting of two TNF receptor components linked to the Fc portion of human IgG. Similar to infliximab, etanercept binds to TNF specifically and blocks its interaction with cell surface receptors (Target 4). In April, 2004, FDA has approved the use of etanercept for the treatment of moderate to severe chronic plaque psoriasis who are candidates for systemic therapy or phototherapy. It is also approved for treating psoriatic arthritis, rheumatoid arthritis and ankylosing spondylitis. It is given subcutaneously 50mg twice per week usually for a course of 12 weeks, then 50mg once per week for maintenance if necessary. PASI 75 was achieved in 49% of patients at week 12 and increased to 59% when twice weekly treatment was extended to 24 weeks. 13 Clinical improvement was evident as early as 2 weeks. The main side effects were upper respiratory infection (57%), injection site reactions (20%), and headache (13%). Eleven percent and 15% reported induction of antinuclear antibodies and anti-double-stranded DNA but full cases of lupus erythematosus are rare. 14 Conclusion These new biologic agents are exciting additions of armamentarium for the treatment of psoriasis. As compared with the conventional immunosuppressive therapy (e.g. cyclosporin), they seem to have an improved risk-efficacy ratio. Among them, infliximab seems to be highly efficacious while alefacept apparently offers longer remission to responders. Furthermore, some of them (e.g. efalizumab and etanercept) can conveniently be administered by the patients themselves at home. The downside of these new treatments are the need of laboratory monitoring in some agents (e.g. platelet count in efalizumab and CD4 count in alefacept) as well as the potential serious side 14
3 VOL.11NO.5 NO.9 MAY SEPTEMBER effects such as infusion reaction and opportunistic infections (e.g. tuberculosis in infliximab). The cost of all these new therapeutic agents are very high. Further studies are needed to compare the cost-effectiveness of these new agents with the current systemic anti- psoriatic therapies. References Lebwohl M, Christophers E, Langley R, Ortonne JP, Roberts J, Griffiths CE; Alefacept Clinical Study Group. An international, randomized, double-blind, placebo controlled phase 3 trial of intramuscular alefacept in patients with chronic plaque psoriasis. Arch Dermatol 2003;139: Krueger GG, Papp KA, Stough DB, Loven KH, Gulliver WP, Ellis CN; Alefacept Clinical Study Group. A randomized, double-blind, placebocontrolled phase III study evaluating efficacy and tolerability of 2 courses of alefacept in patients with chronic plaque psoriasis. J Am Acad Dermatol 2002;47: Ellis CN, Krueger GG; Alefacept Clinical Study Group. Treatment of chronic plaque psoriasis by selective targeting of memory effector T lymphocytes. NEnglJMed2001;345: Krueger GG, Ellis CN. Alefacept therapy produces remission for patients with chronic plaque psoriasis. BrJDermatol2003;148: Gottlieb AB, Blacker K, Duvic M, Leonardi CL. Efficacy and safety of efalizumab long term treatment: preliminary findings from an open label trial. Program of the 61st Annual Meeting of the American Academy of Dermatology; March 21-26, 2003; San Francisco, California. Poster 11. Table 1 summary of four biological agents for the treatment of psoriasis Biological agent Alefacept(Amevive TM ) Efalizumab(Raptiva TM ) Lebwohl M, Tyring SK, Hamilton TK, Toth D, Glazer S, Tawfik NH, et al. A novel targeted T-cell modulator, efalizumab, for plaque psoriasis. NEnglJMed2003; 349: Genentech. Full prescribing information. com/gene/products/information/immunological/ raptiva/insert.jsp Gottlieb A, Miller B, Chaudhari U, Oh C, Sherr A, Solodkina G, et al. Clinical and histologic effects of subcutaneously administered anti- CD11a (hu1124) in patients with psoriasis. J Invest Dermatol 2000;114:840. Gottlieb AB, Li S, Evans R, Menter A. Infliximab in the treatment of psoriasis: results from the first 10 weeks of the phase II trial. Program of the 61st Annual Meeting of the American Academy of Dermatology; March 21-26, 2003; San Francisco, California. Poster 596. Gottlieb AB, Chaudhari U, Mulcahy LD, Li S, Dooley LT, Baker DG. Infliximab monotherapy provides rapid and sustained benefit for plaque-type psoriasis. J Am Acad Dermatol 2003;48: Centocor. Full prescribing information. com/pi/interactive_pi.jsp. 12. Keane J, Gershon S, Wise RP, Mirabile-Levens E, Kasznica J, Schwieterman WD, et al. Tuberculosis associated with infliximab, a tumor necrosis factor alpha-neutralizing agent. N Engl J Med 2001;345: Leonardi CL, Powers JL, Matheson RT, Goffe BS, Zitnik R, Wang A, et al. Etanercept as monotherapy in patients with psoriasis. NEnglJ Med 2003;349: Shakoor N, Michalska M, Harris CA, Block JA. Drug induced systemic lupus erythematosus associated with etanercept therapy. Lancet 2002;359: Infliximab(Remicade TM ) Etanercept (Enbrel TM ) Description Fusion protein of human LFA-3 and Fc portion of Ig G1 Humanized form of murine antibody against CD11a Chimeric antibody against TNF alpha with a human IgG1 constant domain and murine variable regions Fusion protein of the Fc of human IgG1 and the extracellular TNF receptor Mechanism Block LFA-3/CD2 interaction and selective eliminate activated T cells Block LFA-1/ICAM-1 interaction and inhibit T cell activation and inflammatory events Binds soluble and bound TNF alpha and blocks its activities Binds soluble and bound TNF alpha and blocks its activities Target of action Target1&3 Target 1 Target 4 Target 4 FDA indication Mod to severe plaque psoriasis in adults who are candidates for systemic therapy or phototherapy (approved Jan 2003) Mod to severe plaque psoriasis in adults who are candidates for systemic therapy or phototherapy (approved Oct 2003) Crohn's disease and RA Mod to severe plaque psoriasis in adults (approved Apr 2004); psoriatic, rheumatoid and juvenile rheumatoid arthritis, ankylosing spondylitis Route of administration Intramuscular or intravenous bolus Subcutaneous (by patients at home after training) Slow intravenous infusion over 2 hours Subcutaneous (by patients at home after training) Dosing for one course 15mg imi or 7.5mg ivi weekly for 12 weeks Begin with single dose of 0.7mg/kg sc, then 1mg/kg sc weekly for 12 weeks 3 to 5 mg/kg iv infusion at week 0,2,6. 50mg sc twice a week for 12 week (50mg sc weekly if maintenance) Time of onset of improvement ~8weeks ~4 weeks ~4weeks ~2 weeks Relative efficacy: % of patients achieving PASI 75 after a course of 12 week Px ~28-40% ~22-41% (Increase to 64% if prolonged treatment) ~72-88% ~49% (Increase to 59% if prolonged treatment) FDA baseline & monitoring CD4 count (baseline and weekly) Platelet count monthly, then every 3 months Screening for latent TB(tuberculin test, CXR) at baseline None Side effect Flu-like symptoms, injection site reaction, transient chill Flu-like symptoms Headache, myalgia, infusion reaction, opportunistic infections Upper respiratory infection, injection site reaction, headache Contraindication Hypersensitivity to alefacept; discontinue if CD4<250cells/uL for 4 weeks Hypersensitivity to efalizumab or any murine or humanized monoclonal antibody Hypersensitivity to infliximab or murine products; congestive heart failure Hypersensitivity to etanercept; active infections or sepsis; congestive heart failure; poorly controlled diabetes Advantage Long lasting remission Self administered by patients possible Highly efficacious Also effective against psoriatic arthritis; self administered by patient possible Disadvantage Need for CD4 count monitoring, withhold if CD4<250cells/uL, discontinue if persists for 4 weeks Significant relapse after treatment; long term maintenance necessary Manpower for iv infusion; risk of anaphylactic infusion reaction; risk of serious opportunistic infection Serious opportunistic infection reported, prior tuberculin test and CXR advisable in TB endemic area Estimated drug cost (US$) ~$10,000 per course depending on dosing ~$15,000 per year for maintenance treatment ~12,000 per year depending on dosage ~12,000 per year 15
4 VOL.11 NO.9 SEPTEMBER 2006 MCHK CME Programme Self-assessment Questions Please read the article entitled "Comparison of Various Biological Agents in the Treatment of Psoriasis" by Dr.KH Lau and complete the following self-assessment questions. Participants in the MCHK CME Programme will be awarded 1 CME credit under the Programme for returning completed answer sheet via fax ( ) or by mail to the Federation Secretariat on or before 30 September Answers to questions will be provided in the next issue of The Hong Kong Medical Diary. Questions 1-10: Please choose the best answer. 1. The following statements concerning treatment of psoriasis by etanercept are correct except: a. Comparing with other biological agents, the onset of clinical improvement is slow. b Etanercept has a similar mode of action as infliximab. c. It is approved by FDA for the treatment of moderate to severe chronic plaque psoriasis patients who are candidates for systemic therapy or phototherapy. d. The main side effects are upper respiratory tract infection and injection site reactions. e. Induction of antinuclear antibodies and anti-double stranded DNA were reported after treatment with etanercept. 2. The following are relevant side effects of alefacept except: a. Pharyngitis b. Headache c. Rhinitis d. Lymphoctyosis e. Injection site reaction and transient chill during the first day of treatment 3. FDA recommends monitoring of the following parameter(s) during treatment with efalizumab: a. Chest X ray b. White cell count c. Tuberculin test d. Platelet count e. All of the above 4.. The following are correct modes of action of the biological agents designed to block the specific steps in the pathogenetic pathway of psoriasis except: a. Alefacept inhibits T cell activation by blocking the co-stimulatory signal 2. b. Etanercept binds soluble and bound tumour necrosis factor (TNF) alpha and blocks its action. c. Alefacept eliminates activated T cells selectively. d. Efalizumab induces immune deviation from TH1 to TH2 differentiation in the activated T cell. e. Infliximab inhibits the action of cytokines, especially tumour necrosis factor (TNF) alpha. 5 The following is/are disadvantage of treatment with infliximab in psoriasis: a. High treatment cost. b. Reactivation of latent tuberculosis. c. Potential serious anaphylaxis during drug administration. d. a and c. e. All of the above 6. The following statements concerning the use of alefacept in the treatment of psoriasis are correct except: a. Comparing with other biologics, the remission induced by alefacept is transient and significant clinical relapse is one of the major disadvantages. b. It was already approved by FDA of USA for the treatment of moderate to severe plaque psoriasis in adult patients. c Alefacept is a recombinant human LFA-3/IgG1 dimeric fusion protein. d. Alefacept is administered weekly either by intramuscular or by intravenous bolus injection. e. The best clinical efficacy usually occurs few weeks after completing the course of treatment. 7. The following biological agent(s) can be administered at home by trained patients themselves: a. Efalizumab b. Infliximab c. Etanercept d. a and c e. all of the above 8. The following statements concerning the use of efalizumab in the treatment of psoriasis are correct except: a. The usual dosage is 1mg/kg weekly for a course of 12 week. b Long term treatment of efalizumab up to 60 weeks was proven to be safe and well tolerated. c. Comparing with other biologics, remission in the responders was long-lasting and a significant number of patients still remained clear even after stopping treatment. d. It is usually given by subcutaneous injection. e. Additional clinical improvement could be achieved by prolonged treatment beyond 12 weeks with enhanced clinical efficacy. 9. In treating patients with psoriatic arthropathy complicating extensive stable plaque psoriasis, the following biological agent is an FDA approved treatment option: a. Alefacept b. Efalizumab c. Infliximab d. Etanercept e. None of the above 10. Which of the following statement(s) concerning the use of infliximab in the treatment of psoriasis is/are correct: a. It is administered subcutaneously. b. It is given in 50mg twice a week for a course of 12 week. c. It is approved by FDA for the treatment of Crohn's disease and rheumatoid arthritis. d. No laboratory monitoring is required by FDA e. All of the above. 16
5 VOL.11 NO.5 MAY 2006 VOL.11 NO.9 SEPTEMBER 2006 ANSWER SHEET FOR SEPTEMBER 2006 Please return the completed answer sheet to the Federation Secretariat on or before 30 September 2006 for documentation. 1 CME point will be awarded for answering the MCHK CME programme (for non-specialists) self-assessment questions. Comparison of Various Biological Agents in the Treatment of Psoriasis MBBS(HK), FRCP, FHKCP, FHKAM(Med) Yaumatei Dermatology Clinic, Social Hygiene Service Name: HKID No. - X X (x) Signature: Contact TelNo.: Answers to August 2006 issue Aesthetic Dentistry and Orthodontics 1.T 2.T 3.F 4.T 5. F 6.F 7.T 8.F 9.T 10. T 17
Biologic Therapies for Psoriasis. A Systematic Review
 Biologic Therapies for Psoriasis. A Systematic Review WOLF-HENNING BOEHNCKE, JÖRG PRINZ, and ALICE B. GOTTLIEB ABSTRACT. Alefacept, efalizumab, etanercept, and infliximab are currently approved for the
Biologic Therapies for Psoriasis. A Systematic Review WOLF-HENNING BOEHNCKE, JÖRG PRINZ, and ALICE B. GOTTLIEB ABSTRACT. Alefacept, efalizumab, etanercept, and infliximab are currently approved for the
The implication of an immunologic
 Evidence-Based Review of Biologic Therapy for Psoriasis: Infliximab, Etanercept, Adalimumab, Efalizumab, and Alefacept Jeffrey M. Weinberg, MD; Robin Buchholz, MD; Noah Scheinfeld, MD DERMATOLOGY ABSTRACT
Evidence-Based Review of Biologic Therapy for Psoriasis: Infliximab, Etanercept, Adalimumab, Efalizumab, and Alefacept Jeffrey M. Weinberg, MD; Robin Buchholz, MD; Noah Scheinfeld, MD DERMATOLOGY ABSTRACT
Ustekinumab for severe treatment-resistant psoriasis: a 24-week pilot study in Hong Kong Chinese
 Hong Kong J. Dermatol. Venereol. (2011) 19, 59-64 Original Article Ustekinumab for severe treatment-resistant psoriasis: a 24-week pilot study in Hong Kong Chinese Ustekinumab SKF Loo, KH Lau, KM Ho Introduction:
Hong Kong J. Dermatol. Venereol. (2011) 19, 59-64 Original Article Ustekinumab for severe treatment-resistant psoriasis: a 24-week pilot study in Hong Kong Chinese Ustekinumab SKF Loo, KH Lau, KM Ho Introduction:
Off-Label Biologic Regimens in Psoriasis: A Systematic Review of Efficacy and Safety of Dose Escalation, Reduction, and Interrupted Biologic Therapy
 : A Systematic Review of Efficacy and Safety of Dose Escalation, Reduction, and Interrupted Biologic Therapy Elizabeth A. Brezinski 2, April W. Armstrong 1 * 1 Department of Dermatology, University of
: A Systematic Review of Efficacy and Safety of Dose Escalation, Reduction, and Interrupted Biologic Therapy Elizabeth A. Brezinski 2, April W. Armstrong 1 * 1 Department of Dermatology, University of
THE THERAPY OF THE REBEL SEVERE PSORIAZIS WITH BIOLOGICAL PREPARATS
 Bulletin of the Transilvania University of Braşov Series VI: Medical Sciences Vol. 5 (54) No. 2-2012 THE THERAPY OF THE REBEL SEVERE PSORIAZIS WITH BIOLOGICAL PREPARATS Mădălina FRÎNCU 1 Abstract: Biological
Bulletin of the Transilvania University of Braşov Series VI: Medical Sciences Vol. 5 (54) No. 2-2012 THE THERAPY OF THE REBEL SEVERE PSORIAZIS WITH BIOLOGICAL PREPARATS Mădălina FRÎNCU 1 Abstract: Biological
Effects of biological response modifiers in psoriasis and psoriatic arthritis Goedkoop, A.Y.
 UvA-DARE (Digital Academic Repository) Effects of biological response modifiers in psoriasis and psoriatic arthritis Goedkoop, A.Y. Link to publication Citation for published version (APA): Goedkoop, A.
UvA-DARE (Digital Academic Repository) Effects of biological response modifiers in psoriasis and psoriatic arthritis Goedkoop, A.Y. Link to publication Citation for published version (APA): Goedkoop, A.
What is Enbrel? Key features
 What is Enbrel? Enbrel (also known by its generic name etanercept) is a biologic medication approved in April 2004 by the U.S. Food and Drug Administration (FDA) for the treatment of moderate to severe
What is Enbrel? Enbrel (also known by its generic name etanercept) is a biologic medication approved in April 2004 by the U.S. Food and Drug Administration (FDA) for the treatment of moderate to severe
Anti-TNF biologic agents Dr Lluís Puig
 Department of Dermatology Hospital de la Santa Creu i Sant Pau IPC NOVARTIS PSORIASIS PRECEPTORSHIP Anti-TNF biologic agents Dr Lluís Puig Barcelona, July 9th-10th, 2013 Anti-TNF therapy in the pathophysiology
Department of Dermatology Hospital de la Santa Creu i Sant Pau IPC NOVARTIS PSORIASIS PRECEPTORSHIP Anti-TNF biologic agents Dr Lluís Puig Barcelona, July 9th-10th, 2013 Anti-TNF therapy in the pathophysiology
Scottish Medicines Consortium
 Scottish Medicines Consortium ustekinumab, 45mg solution for injection (Stelara ) No. (572/09) Janssen-Cilag Ltd 15 January 2010 The Scottish Medicines Consortium (SMC) has completed its assessment of
Scottish Medicines Consortium ustekinumab, 45mg solution for injection (Stelara ) No. (572/09) Janssen-Cilag Ltd 15 January 2010 The Scottish Medicines Consortium (SMC) has completed its assessment of
USTEKINUMAB and BRIAKINUMAB
 USTEKINUMAB and BRIAKINUMAB Andrew Blauvelt, M.D. Professor, Dept. of Dermatology and Dept. of Molecular Microbiology & Immunology Oregon Health & Science University Chief, Dermatology Service Veteran
USTEKINUMAB and BRIAKINUMAB Andrew Blauvelt, M.D. Professor, Dept. of Dermatology and Dept. of Molecular Microbiology & Immunology Oregon Health & Science University Chief, Dermatology Service Veteran
The Cosentyx clinical trial programme 1-11
 The Cosentyx clinical trial programme 1-11 There are eight pivotal trials (four in psoriasis, two in psoriatic arthritis, two in ankylosing spondylitis) There are two head-to-head trials in psoriasis showing
The Cosentyx clinical trial programme 1-11 There are eight pivotal trials (four in psoriasis, two in psoriatic arthritis, two in ankylosing spondylitis) There are two head-to-head trials in psoriasis showing
Announcing HUMIRA. Psoriasis Starter Package
 Announcing HUMIRA (adalimumab) Psoriasis Starter Package HUMIRA is indicated for the treatment of adult patients with moderate to severe chronic plaque psoriasis who are candidates for systemic therapy
Announcing HUMIRA (adalimumab) Psoriasis Starter Package HUMIRA is indicated for the treatment of adult patients with moderate to severe chronic plaque psoriasis who are candidates for systemic therapy
TUBERCULOSIS AND THE TNF-α INHIBITORS. Lloyd Friedman, M.D. Yale University Milford Hospital
 TUBERCULOSIS AND THE TNF-α INHIBITORS Lloyd Friedman, M.D. Yale University Milford Hospital Outline TNF-α Anti-TNF-α medications Rates of tuberculosis Lower rates with etanercept Screening for latent tuberculosis
TUBERCULOSIS AND THE TNF-α INHIBITORS Lloyd Friedman, M.D. Yale University Milford Hospital Outline TNF-α Anti-TNF-α medications Rates of tuberculosis Lower rates with etanercept Screening for latent tuberculosis
The role of current biologic therapies in psoriasis
 : An Update on and IL-17 Inhibitors Joanna Dong, BA; Gary Goldenberg, MD PRACTICE POINTS The newest biologics for treatment of moderate to severe plaque psoriasis are and IL-17 inhibitors with unprecedented
: An Update on and IL-17 Inhibitors Joanna Dong, BA; Gary Goldenberg, MD PRACTICE POINTS The newest biologics for treatment of moderate to severe plaque psoriasis are and IL-17 inhibitors with unprecedented
Medical Coverage Guidelines are subject to change as new information becomes available.
 ENBREL (etanercept) Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Medical Coverage Guideline
ENBREL (etanercept) Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Medical Coverage Guideline
Thank you for agreeing to give us a statement on your organisation s view of the technology and the way it should be used in the NHS.
 Thank you for agreeing to give us a statement on your organisation s view of the technology and the way it should be used in the NHS. Healthcare professionals can provide a unique perspective on the technology
Thank you for agreeing to give us a statement on your organisation s view of the technology and the way it should be used in the NHS. Healthcare professionals can provide a unique perspective on the technology
1. Background: Infliximab is administered parenterally; therefore, it is not covered under retail pharmacy benefits.
 Subject: Infliximab (Remicade ) Original Original Committee Approval: October 13, 2006 Revised Last Committee Approval: December 3, 2008 Last Review: October 19, 2007 1. Background: Infliximab is a genetically
Subject: Infliximab (Remicade ) Original Original Committee Approval: October 13, 2006 Revised Last Committee Approval: December 3, 2008 Last Review: October 19, 2007 1. Background: Infliximab is a genetically
Monitoring Patients on Biologic Therapies Murlidhar Rajagopalan, MBBS, MD
 Monitoring Patients on Biologic Therapies Murlidhar Rajagopalan, MBBS, MD Biologics available for psoriasis 1) tumor necrosis factor alpha (TNF-α) inhibitors, 2) interleukin (IL)-12/23 inhibitors, and
Monitoring Patients on Biologic Therapies Murlidhar Rajagopalan, MBBS, MD Biologics available for psoriasis 1) tumor necrosis factor alpha (TNF-α) inhibitors, 2) interleukin (IL)-12/23 inhibitors, and
2.0 Synopsis. Adalimumab R&D/04/118. (For National Authority Use Only) Referring to Part of Dossier: Volume:
 2.0 Synopsis Abbott Laboratories Name of Study Drug: Adalimumab Name of Active Ingredient: Adalimumab Title of Study: Individual Study Table Referring to Part of Dossier: Volume: Page: (For National Authority
2.0 Synopsis Abbott Laboratories Name of Study Drug: Adalimumab Name of Active Ingredient: Adalimumab Title of Study: Individual Study Table Referring to Part of Dossier: Volume: Page: (For National Authority
Cimzia. Cimzia (certolizumab pegol) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.11 Subject: Cimzia Page: 1 of 5 Last Review Date: December 8, 2017 Cimzia Description Cimzia (certolizumab
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.11 Subject: Cimzia Page: 1 of 5 Last Review Date: December 8, 2017 Cimzia Description Cimzia (certolizumab
Ustekinumab (Stelara) for psoriatic arthritis second line after disease modifying anti rheumatic drugs (DMARDs)
 Ustekinumab (Stelara) for psoriatic arthritis second line after disease modifying anti rheumatic drugs (DMARDs) January 2010 This technology summary is based on information available at the time of research
Ustekinumab (Stelara) for psoriatic arthritis second line after disease modifying anti rheumatic drugs (DMARDs) January 2010 This technology summary is based on information available at the time of research
Primary Results Citation 2
 Table S1. Adalimumab clinical trials 1 ClinicalTrials.gov Rheumatoid Arthritis 3 NCT00195663 Breedveld FC, Weisman MH, Kavanaugh AF, et al. The PREMIER study. A multicenter, randomized, double-blind clinical
Table S1. Adalimumab clinical trials 1 ClinicalTrials.gov Rheumatoid Arthritis 3 NCT00195663 Breedveld FC, Weisman MH, Kavanaugh AF, et al. The PREMIER study. A multicenter, randomized, double-blind clinical
Alka Dogra, Silonie Sachdeva Department of Dermatology, Venereology and Leprology, Dayanand Medical College and Hospital, Ludhiana, Punjab, India.
 Review Article Biologic therapy in psoriasis Alka Dogra, Silonie Sachdeva Department of Dermatology, Venereology and Leprology, Dayanand Medical College and Hospital, Ludhiana, Punjab, India. Address for
Review Article Biologic therapy in psoriasis Alka Dogra, Silonie Sachdeva Department of Dermatology, Venereology and Leprology, Dayanand Medical College and Hospital, Ludhiana, Punjab, India. Address for
Bioavailability 55% - 77%
 Brand Name: Cosentyx Generic Name: secukinumab Manufacturer 1 : Novartis Pharmaceuticals Corporation Drug Class 2,3 : Antipsoriatic Agent, Interleukin-17A Receptor Antagonist Uses: Labeled Uses 1,2,3 :
Brand Name: Cosentyx Generic Name: secukinumab Manufacturer 1 : Novartis Pharmaceuticals Corporation Drug Class 2,3 : Antipsoriatic Agent, Interleukin-17A Receptor Antagonist Uses: Labeled Uses 1,2,3 :
REVIEW Targeting psoriasis with new therapies
 REVIEW Targeting psoriasis with new therapies A. Prohić, H. Helppikangas, J. Muhović Department of Dermatovenerology, Sarajevo University Clinical Centre, Sarajevo, Bosnia and Herzegovina ABSTRACT The
REVIEW Targeting psoriasis with new therapies A. Prohić, H. Helppikangas, J. Muhović Department of Dermatovenerology, Sarajevo University Clinical Centre, Sarajevo, Bosnia and Herzegovina ABSTRACT The
C. Assess clinical response after the first three months of treatment.
 Government Health Plan (GHP) of Puerto Rico Authorization Criteria Tumor Necrosis Factor Alpha (TNFα) Adalimumab (Humira ) Managed by MCO Section I. Prior Authorization Criteria A. Physician must submit
Government Health Plan (GHP) of Puerto Rico Authorization Criteria Tumor Necrosis Factor Alpha (TNFα) Adalimumab (Humira ) Managed by MCO Section I. Prior Authorization Criteria A. Physician must submit
What prescribers need to know
 HUMIRA Citrate-free presentations in an Electronic Medical Record (EMR) What prescribers need to know 2 / This is your guide to identifying HUMIRA Citrate-free presentations in your Electronic Medical
HUMIRA Citrate-free presentations in an Electronic Medical Record (EMR) What prescribers need to know 2 / This is your guide to identifying HUMIRA Citrate-free presentations in your Electronic Medical
ASSESSMENT OF IMMUNOLOGICAL THERAPY WITH INFLIXIMAB AND ETANERCEPT IN THE TREATMENT OF PSORIASIS ARTHROPATHY
 Vol XIV, Number 2, June 2010 Pages 117-121 Copyright reserved 2010 ORIGINAL PAPERS ASSESSMENT OF IMMUNOLOGICAL THERAPY WITH INFLIXIMAB AND ETANERCEPT IN THE TREATMENT OF PSORIASIS ARTHROPATHY Liana Suciu,
Vol XIV, Number 2, June 2010 Pages 117-121 Copyright reserved 2010 ORIGINAL PAPERS ASSESSMENT OF IMMUNOLOGICAL THERAPY WITH INFLIXIMAB AND ETANERCEPT IN THE TREATMENT OF PSORIASIS ARTHROPATHY Liana Suciu,
Cimzia. Cimzia (certolizumab pegol) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Cimzia Page: 1 of 5 Last Review Date: March 17, 2017 Cimzia Description Cimzia (certolizumab pegol)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Cimzia Page: 1 of 5 Last Review Date: March 17, 2017 Cimzia Description Cimzia (certolizumab pegol)
Ustekinumab Treatment of Erythrodermic Psoriasis Occurring after Physical Stress: A Report of Two Cases
 Published online: September 26, 2013 1662 6567/13/0053 0254$38.00/0 This is an Open Access article licensed under the terms of the Creative Commons Attribution-NonCommercial 3.0 Unported license (CC BY-NC)
Published online: September 26, 2013 1662 6567/13/0053 0254$38.00/0 This is an Open Access article licensed under the terms of the Creative Commons Attribution-NonCommercial 3.0 Unported license (CC BY-NC)
Scottish Medicines Consortium
 Scottish Medicines Consortium etanercept 25mg vial of powder for subcutaneous injection (Enbrel ) (No. 212/05) Wyeth New indication: severe active ankylosing spondylitis inadequately controlled by conventional
Scottish Medicines Consortium etanercept 25mg vial of powder for subcutaneous injection (Enbrel ) (No. 212/05) Wyeth New indication: severe active ankylosing spondylitis inadequately controlled by conventional
An otherwise healthy 12-year-old
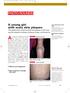 1105 Photo Rounds.finalREV 10/19/05 2:05 PM Page 947 A young girl with scaly skin plaques The patient had numerous thick red plaques on her back and the extensor surfaces of elbows, knees, and forearms
1105 Photo Rounds.finalREV 10/19/05 2:05 PM Page 947 A young girl with scaly skin plaques The patient had numerous thick red plaques on her back and the extensor surfaces of elbows, knees, and forearms
STELARA (ustekinumab)
 STELARA (ustekinumab) Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs
STELARA (ustekinumab) Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs
Humira. (adalimumab) Drug Update Slideshow NEW INDICATION
 Humira (adalimumab) NEW INDICATION Drug Update Slideshow Introduction Brand name: Humira Generic name: Adalimumab Pharmacological class: Tumor necrosis factor (TNF) blocker Strength and Formulation: 10mg/0.2mL,
Humira (adalimumab) NEW INDICATION Drug Update Slideshow Introduction Brand name: Humira Generic name: Adalimumab Pharmacological class: Tumor necrosis factor (TNF) blocker Strength and Formulation: 10mg/0.2mL,
Prior Authorization Conditions for Approval of Enbrel (etanercept) Website Form Submit request via: Fax
 Prior Authorization Conditions for Approval of Enbrel (etanercept) Website Form www.highmarkhealthoptions.com Submit request via: Fax - 1-855-476-4158 All requests for Enbrel (etanercept) require a prior
Prior Authorization Conditions for Approval of Enbrel (etanercept) Website Form www.highmarkhealthoptions.com Submit request via: Fax - 1-855-476-4158 All requests for Enbrel (etanercept) require a prior
Regulatory Status FDA-approved indication: Orencia is a selective T cell costimulation modulator indicated for: (1)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.18 Subject: Orencia Page: 1 of 6 Last Review Date: December 8, 2017 Orencia Description Orencia (abatacept)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.18 Subject: Orencia Page: 1 of 6 Last Review Date: December 8, 2017 Orencia Description Orencia (abatacept)
HELPING YOU AND YOUR PATIENTS TALK OPENLY ABOUT MODERATELY TO SEVERELY ACTIVE RA
 SIMPONI ARIA (golimumab) is indicated for the treatment of adults with moderately to severely active rheumatoid arthritis (RA) in combination with MTX, active psoriatic arthritis, and active ankylosing
SIMPONI ARIA (golimumab) is indicated for the treatment of adults with moderately to severely active rheumatoid arthritis (RA) in combination with MTX, active psoriatic arthritis, and active ankylosing
Using ENBREL to Treat Rheumatoid and Psoriatic Arthritis
 Using ENBREL to Treat Rheumatoid and Psoriatic Arthritis Writing White Papers class Bellevue Community College TABLE OF CONTENTS TABLE OF CONTENTS...2 OVERVIEW...3 RHEUMATOID ARTHRITIS... 3 JUVENILE RHEUMATOID
Using ENBREL to Treat Rheumatoid and Psoriatic Arthritis Writing White Papers class Bellevue Community College TABLE OF CONTENTS TABLE OF CONTENTS...2 OVERVIEW...3 RHEUMATOID ARTHRITIS... 3 JUVENILE RHEUMATOID
Pharmacy Medical Necessity Guidelines: Stelara (ustekinumab)
 Pharmacy Medical Necessity Guidelines: Effective: January 1, 2018 Type of Review Care Prior Authorization Required Management Not Covered Type of Review Clinical Review SQ: RX/ Pharmacy (RX) or Medical
Pharmacy Medical Necessity Guidelines: Effective: January 1, 2018 Type of Review Care Prior Authorization Required Management Not Covered Type of Review Clinical Review SQ: RX/ Pharmacy (RX) or Medical
PACKAGE INSERT. Each vial of the REVELLEX product contains 100 mg of infliximab. After reconstitution, each vial
 PACKAGE INSERT SCHEDULING STATUS S4 PROPRIETARY NAME AND DOSAGE FORM REVELLEX 100 mg COMPOSITION Each vial of the REVELLEX product contains 100 mg of infliximab. After reconstitution, each vial of REVELLEX
PACKAGE INSERT SCHEDULING STATUS S4 PROPRIETARY NAME AND DOSAGE FORM REVELLEX 100 mg COMPOSITION Each vial of the REVELLEX product contains 100 mg of infliximab. After reconstitution, each vial of REVELLEX
Pharmacy Medical Necessity Guidelines: Stelara (ustekinumab)
 Pharmacy Medical Necessity Guidelines: Effective: November 14, 2017 Prior Authorization Required Type of Review Care Management Not Covered Type of Review Clinical Review SQ: RXUM/ RX / Pharmacy (RX) or
Pharmacy Medical Necessity Guidelines: Effective: November 14, 2017 Prior Authorization Required Type of Review Care Management Not Covered Type of Review Clinical Review SQ: RXUM/ RX / Pharmacy (RX) or
2.0 Synopsis. Adalimumab M Clinical Study Report R&D/04/900. (For National Authority Use Only) Referring to Part of Dossier: Volume:
 2. Synopsis Abbott Laboratories Name of Study Drug: Name of Active Ingredient: Title of Study: Individual Study Table Referring to Part of Dossier: Volume: Page: (For National Authority Use Only) Phase
2. Synopsis Abbott Laboratories Name of Study Drug: Name of Active Ingredient: Title of Study: Individual Study Table Referring to Part of Dossier: Volume: Page: (For National Authority Use Only) Phase
Antirheumatic drugs. Rheumatic Arthritis (RA)
 Antirheumatic drugs Rheumatic Arthritis (RA) Disease Modifying Antirheumatic drugs (DMARDs) DMARDs are used in the treatment of rheumatic arthritis RA and have been shown to slow the course of the disease,
Antirheumatic drugs Rheumatic Arthritis (RA) Disease Modifying Antirheumatic drugs (DMARDs) DMARDs are used in the treatment of rheumatic arthritis RA and have been shown to slow the course of the disease,
Research Article Clinical Outcome of a Novel Anti-CD6 Biologic Itolizumab in Patients of Psoriasis with Comorbid Conditions
 Dermatology Research and Practice Volume 2, Article ID 13326, 4 pages http://dx.doi.org/.1155/2/13326 Research Article Clinical Outcome of a Novel Anti-CD6 Biologic Itolizumab in Patients of Psoriasis
Dermatology Research and Practice Volume 2, Article ID 13326, 4 pages http://dx.doi.org/.1155/2/13326 Research Article Clinical Outcome of a Novel Anti-CD6 Biologic Itolizumab in Patients of Psoriasis
London, 1 June 2006 Product name: REMICADE Procedure number: Remicade-H-240-II-73-AR SCIENTIFIC DISCUSSION 1/8
 London, 1 June 2006 Product name: REMICADE Procedure number: Remicade-H-240-II-73-AR SCIENTIFIC DISCUSSION 1/8 1. Introduction Infliximab is a chimeric human-murine IgG1κ monoclonal antibody, which binds
London, 1 June 2006 Product name: REMICADE Procedure number: Remicade-H-240-II-73-AR SCIENTIFIC DISCUSSION 1/8 1. Introduction Infliximab is a chimeric human-murine IgG1κ monoclonal antibody, which binds
IMO 3100, an antagonist of Toll like receptor (TLR) 7 and TLR9, demonstrates clinical activity in psoriasis patients
 IMO 3100, an antagonist of Toll like receptor (TLR) 7 and TLR9, demonstrates clinical activity in psoriasis patients with 4 weeks of treatment in a Phase 2a trial A. B. Kimball 1, J. Krueger 2, T. Sullivan
IMO 3100, an antagonist of Toll like receptor (TLR) 7 and TLR9, demonstrates clinical activity in psoriasis patients with 4 weeks of treatment in a Phase 2a trial A. B. Kimball 1, J. Krueger 2, T. Sullivan
BJD. Summary. British Journal of Dermatology THERAPEUTICS
 THERAPEUTICS BJD British Journal of Dermatology Efficacy of psoralen plus ultraviolet A therapy vs. biologics in moderate to severe chronic plaque psoriasis: retrospective data analysis of a patient registry
THERAPEUTICS BJD British Journal of Dermatology Efficacy of psoralen plus ultraviolet A therapy vs. biologics in moderate to severe chronic plaque psoriasis: retrospective data analysis of a patient registry
ENBREL (Etanercept) 25 mg and 50 mg* powder for injection and water for injections
 PRODUCT INFORMATION ENBREL Etanercept (rch) NAME OF THE MEDICINE ENBREL (Etanercept) 25 mg and 50 mg* powder for injection and water for injections ENBREL (Etanercept) 25 mg* and 50 mg solution for injection
PRODUCT INFORMATION ENBREL Etanercept (rch) NAME OF THE MEDICINE ENBREL (Etanercept) 25 mg and 50 mg* powder for injection and water for injections ENBREL (Etanercept) 25 mg* and 50 mg solution for injection
Cimzia. Cimzia (certolizumab pegol) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.11 Section: Prescription Drugs Effective Date: April 1, 2018 Subject: Cimzia Page: 1 of 5 Last Review
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.11 Section: Prescription Drugs Effective Date: April 1, 2018 Subject: Cimzia Page: 1 of 5 Last Review
Remicade. Remicade (infliximab), Inflectra (infliximab-dyyb) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Remicade Page: 1 of 9 Last Review Date: June 22, 2017 Remicade Description Remicade (infliximab),
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Remicade Page: 1 of 9 Last Review Date: June 22, 2017 Remicade Description Remicade (infliximab),
Actemra (tocilizumab) CG-DRUG-81
 Market DC Actemra (tocilizumab) CG-DRUG-81 Override(s) Prior Authorization Approval Duration 1 year Medications Line of Business Quantity Limit Actemra (tocilizumab) vials VA MCD and All L-AGP May be subject
Market DC Actemra (tocilizumab) CG-DRUG-81 Override(s) Prior Authorization Approval Duration 1 year Medications Line of Business Quantity Limit Actemra (tocilizumab) vials VA MCD and All L-AGP May be subject
Biologics in Psoriasis. Peter CM van de Kerkhof Department of Dermatology Radboud University Nijmegen Medical Centre
 Biologics in Psoriasis Peter CM van de Kerkhof Department of Dermatology Radboud University Nijmegen Medical Centre Disclosures Consultancy services for Celgene, Centocor, Almirall, Amgen, Pfizer, Philips,
Biologics in Psoriasis Peter CM van de Kerkhof Department of Dermatology Radboud University Nijmegen Medical Centre Disclosures Consultancy services for Celgene, Centocor, Almirall, Amgen, Pfizer, Philips,
Remicade. Remicade (infliximab), Inflectra (infliximab-dyyb) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.02 Subsection: Gastrointestinal nts Original Policy Date: May 20, 2011 Subject: Remicade Page: 1 of
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.02 Subsection: Gastrointestinal nts Original Policy Date: May 20, 2011 Subject: Remicade Page: 1 of
Regulatory Status FDA-approved indication: Orencia is a selective T cell costimulation modulator indicated for: (1)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.18 Subject: Orencia Page: 1 of 8 Last Review Date: March 16, 2018 Orencia Description Orencia (abatacept)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.18 Subject: Orencia Page: 1 of 8 Last Review Date: March 16, 2018 Orencia Description Orencia (abatacept)
DRAFT. Remission rates, calculated using observed case (OC) analyses were as follows: Year 1 Year 2 Year 3 Year 4 All patients 62.
 DRAFT New Efficacy Data Shows Cimzia (certolizumab pegol) Provides Long-Term Remission of Moderate to Severe Crohn s Disease Regardless of Prior Anti-TNF Exposure, According to Data Presented at DDW Oral
DRAFT New Efficacy Data Shows Cimzia (certolizumab pegol) Provides Long-Term Remission of Moderate to Severe Crohn s Disease Regardless of Prior Anti-TNF Exposure, According to Data Presented at DDW Oral
Skin & Allergy News. Practical Considerations in the Treatment of Psoriasis with Biologics
 A Supplement to Skin & Allergy News Practical Considerations in the Treatment of Psoriasis with Biologics Introduction 3 David M. Pariser, MD, Chair Professor, Department of Dermatology Eastern Virginia
A Supplement to Skin & Allergy News Practical Considerations in the Treatment of Psoriasis with Biologics Introduction 3 David M. Pariser, MD, Chair Professor, Department of Dermatology Eastern Virginia
New Cimzia data, with sites in the US, demonstrate safety and efficacy, increased participation in social activities for adult RA patients
 Long-term Cimzia (certolizumab pegol) data demonstrated rapid sustained improvements in clinical outcomes and quality of life in moderate to severe rheumatoid arthritis (RA) patients New Cimzia data, with
Long-term Cimzia (certolizumab pegol) data demonstrated rapid sustained improvements in clinical outcomes and quality of life in moderate to severe rheumatoid arthritis (RA) patients New Cimzia data, with
Drug Class Review Targeted Immune Modulators
 Drug Class Review Targeted Immune Modulators Final Update 3 Report March 2012 The Agency for Healthcare Research and Quality has not yet seen or approved this report The purpose of the is to summarize
Drug Class Review Targeted Immune Modulators Final Update 3 Report March 2012 The Agency for Healthcare Research and Quality has not yet seen or approved this report The purpose of the is to summarize
Biologic agents in psoriasis
 Blackwell Publishing AsiaMelbourne, AustraliaAJDAustralasian Journal of Dermatology0004-8380? 2006474217230MiscellaneousBiologic agents in psoriasismr Lee and AJ Cooper Australasian Journal of Dermatology
Blackwell Publishing AsiaMelbourne, AustraliaAJDAustralasian Journal of Dermatology0004-8380? 2006474217230MiscellaneousBiologic agents in psoriasismr Lee and AJ Cooper Australasian Journal of Dermatology
3. Has the patient shown improvement in signs and symptoms of the disease? Y N
 Pharmacy Prior Authorization MERC CARE (MEDICAID) Renflexis (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign and date. Fax
Pharmacy Prior Authorization MERC CARE (MEDICAID) Renflexis (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign and date. Fax
Cigna Drug and Biologic Coverage Policy
 Cigna Drug and Biologic Coverage Policy Subject Apremilast Table of Contents Coverage Policy... 1 General Background... 2 Coding/Billing Information... 4 References... 4 Effective Date... 1/1/2018 Next
Cigna Drug and Biologic Coverage Policy Subject Apremilast Table of Contents Coverage Policy... 1 General Background... 2 Coding/Billing Information... 4 References... 4 Effective Date... 1/1/2018 Next
Psoriasis: Therapeutic goals
 Psoriasis: Therapeutic goals I want to die 50 45 impetiginization infliximab 600 40 35 30 400 25 20 15 200 10 5 0 22-ene 21-feb 23-mar 22-abr 22- may Efalizumab 6 doses: flare + REBOUND CSA 3 21-jun 21-jul
Psoriasis: Therapeutic goals I want to die 50 45 impetiginization infliximab 600 40 35 30 400 25 20 15 200 10 5 0 22-ene 21-feb 23-mar 22-abr 22- may Efalizumab 6 doses: flare + REBOUND CSA 3 21-jun 21-jul
ACTEMRA (tocilizumab)
 ACTEMRA (tocilizumab) Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs
ACTEMRA (tocilizumab) Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs
Clinical Policy: Ixekizumab (Taltz) Reference Number: ERX.SPA.122 Effective Date:
 Clinical Policy: (Taltz) Reference Number: ERX.SPA.122 Effective Date: 10.01.16 Last Review Date: 11.18 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: (Taltz) Reference Number: ERX.SPA.122 Effective Date: 10.01.16 Last Review Date: 11.18 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: Etanercept (Enbrel) Reference Number: PA.CP.PHAR.250 Effective Date: 01/18 Last Review Date: 08/17 Line of Business: Medicaid
 Clinical Policy: (Enbrel) Reference Number: PA.CP.PHAR.250 Effective Date: 01/18 Last Review Date: 08/17 Line of Business: Medicaid Coding Implications Revision Log Description (Enbrel ) is tumor necrosis
Clinical Policy: (Enbrel) Reference Number: PA.CP.PHAR.250 Effective Date: 01/18 Last Review Date: 08/17 Line of Business: Medicaid Coding Implications Revision Log Description (Enbrel ) is tumor necrosis
Stelara. Stelara (ustekinumab) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.90.04 Subject: Stelara Page: 1 of 9 Last Review Date: September 20, 2018 Stelara Description Stelara
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.90.04 Subject: Stelara Page: 1 of 9 Last Review Date: September 20, 2018 Stelara Description Stelara
UvA-DARE (Digital Academic Repository) Innovative therapies and new targets in psoriasis de Groot, M. Link to publication
 UvA-DARE (Digital Academic Repository) Innovative therapies and new targets in psoriasis de Groot, M. Link to publication Citation for published version (APA): de Groot, M. (2011). Innovative therapies
UvA-DARE (Digital Academic Repository) Innovative therapies and new targets in psoriasis de Groot, M. Link to publication Citation for published version (APA): de Groot, M. (2011). Innovative therapies
TRANSPARENCY COMMITTEE OPINION. 26 April 2006
 TRANSPARENCY COMMITTEE OPINION 26 April 2006 REMICADE 100 mg powder for concentrate for solution for infusion Box of 1 (CIP code: 562 070.1) Applicant : laboratoires Schering Plough List I Drug for hospital
TRANSPARENCY COMMITTEE OPINION 26 April 2006 REMICADE 100 mg powder for concentrate for solution for infusion Box of 1 (CIP code: 562 070.1) Applicant : laboratoires Schering Plough List I Drug for hospital
The New and Emerging Agents: Dermatology
 Psoriasis Treatment 2013: What is New and on the Horizon? Neil J Korman, MD, PhD Professor of Dermatology University Hospitals Case Medical Center Clinical Director Murdough Family Center for Psoriasis
Psoriasis Treatment 2013: What is New and on the Horizon? Neil J Korman, MD, PhD Professor of Dermatology University Hospitals Case Medical Center Clinical Director Murdough Family Center for Psoriasis
USTEKINUMAB. London New Drugs Group APC/DTC Briefing Document. May 2009
 Page 1 London New Drugs Group APC/DTC Briefing Document USTEKINUMAB Contents Summary 1 Background 4 Clinical efficacy 5 Cost implications 9 Reference list 10 Appendices 11 Produced for the London New Drugs
Page 1 London New Drugs Group APC/DTC Briefing Document USTEKINUMAB Contents Summary 1 Background 4 Clinical efficacy 5 Cost implications 9 Reference list 10 Appendices 11 Produced for the London New Drugs
Center for Evidence-based Policy
 P&T Committee Brief Targeted Immune Modulators: Comparative Drug Class Review Alison Little, MD Center for Evidence-based Policy Oregon Health & Science University 3455 SW US Veterans Hospital Road, SN-4N
P&T Committee Brief Targeted Immune Modulators: Comparative Drug Class Review Alison Little, MD Center for Evidence-based Policy Oregon Health & Science University 3455 SW US Veterans Hospital Road, SN-4N
Case Example: Exposure Response to Support Extrapolation of Efficacy in Pediatric Ulcerative Colitis
 Case Example: Exposure Response to Support Extrapolation of Efficacy in Pediatric Ulcerative Colitis Kerry Jo Lee, MD Medical Officer Division of Gastroenterology and Inborn Error Products Office of New
Case Example: Exposure Response to Support Extrapolation of Efficacy in Pediatric Ulcerative Colitis Kerry Jo Lee, MD Medical Officer Division of Gastroenterology and Inborn Error Products Office of New
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION. 13 May 2009
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 13 May 2009 STELARA 45 mg, solution for injection B/1 x 0.5 ml vial (CIP code: 392 586-2) JANSSEN-CILAG Ustekinumab
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 13 May 2009 STELARA 45 mg, solution for injection B/1 x 0.5 ml vial (CIP code: 392 586-2) JANSSEN-CILAG Ustekinumab
Scottish Medicines Consortium
 Scottish Medicines Consortium infliximab 100mg powder for intravenous infusion (Remicade ) No. (364/07) Schering-Plough UK Ltd 6 April 2007 The Scottish Medicines Consortium (SMC) has completed its assessment
Scottish Medicines Consortium infliximab 100mg powder for intravenous infusion (Remicade ) No. (364/07) Schering-Plough UK Ltd 6 April 2007 The Scottish Medicines Consortium (SMC) has completed its assessment
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 This medicinal product is subject to additional monitoring. This will allow quick identification of new safety information. Healthcare professionals are asked
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 This medicinal product is subject to additional monitoring. This will allow quick identification of new safety information. Healthcare professionals are asked
ADDITIONAL RESOURCES
 ADDITIONAL RESOURCES CURRENT MODEL OF PSORIASIS IMMUNOPATHOGENESIS IL-17 A/FIL-22 GM-CSF Stressed keratinocytes TNF-α Myeloid dendritic cell Keratinocytes Activation IL-23 IL-12 T cells IL-1 BIL-6 TNF-α
ADDITIONAL RESOURCES CURRENT MODEL OF PSORIASIS IMMUNOPATHOGENESIS IL-17 A/FIL-22 GM-CSF Stressed keratinocytes TNF-α Myeloid dendritic cell Keratinocytes Activation IL-23 IL-12 T cells IL-1 BIL-6 TNF-α
Cyltezo (adalimumab-adbm) CG-DRUG-64, CG-DRUG-65
 Market DC Cyltezo (adalimumab-adbm) CG-DRUG-64, CG-DRUG-65 Override(s) Prior Authorization Quantity Limit Medications Cyltezo (adalimumab-adbm) 40 mg/0.8 ml prefilled syringe #* ^ Approval Duration 1 year
Market DC Cyltezo (adalimumab-adbm) CG-DRUG-64, CG-DRUG-65 Override(s) Prior Authorization Quantity Limit Medications Cyltezo (adalimumab-adbm) 40 mg/0.8 ml prefilled syringe #* ^ Approval Duration 1 year
Regulatory Status FDA-approved indication: Humira and Amjevita are tumor necrosis factor (TNF) blockers indicated for the treatment of: (2-3)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.29 Subject: Humira Page: 1 of 10 Last Review Date: June 22, 2017 Humira Description Humira (adalimumab),
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.29 Subject: Humira Page: 1 of 10 Last Review Date: June 22, 2017 Humira Description Humira (adalimumab),
Clinical Policy: Ustekinumab (Stelara) Reference Number: ERX.SPMN.167
 Clinical Policy: (Stelara) Reference Number: ERX.SPMN.167 Effective Date: 10/16 Last Review Date: 12/16 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
Clinical Policy: (Stelara) Reference Number: ERX.SPMN.167 Effective Date: 10/16 Last Review Date: 12/16 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
Clinical Policy: Secukinumab (Cosentyx) Reference Number: CP.PHAR.261 Effective Date: 08/16 Last Review Date: 08/17
 Clinical Policy: (Cosentyx) Reference Number: CP.PHAR.261 Effective Date: 08/16 Last Review Date: 08/17 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: (Cosentyx) Reference Number: CP.PHAR.261 Effective Date: 08/16 Last Review Date: 08/17 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
HIGHLIGHTS OF PRESCRIBING INFORMATION
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use INFLECTRA safely and effectively. See full prescribing information for INFLECTRA. INFLECTRA (infliximab-dyyb)
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use INFLECTRA safely and effectively. See full prescribing information for INFLECTRA. INFLECTRA (infliximab-dyyb)
A Novel Targeted T-Cell Modulator, Efalizumab, for Plaque Psoriasis
 The new england journal of medicine original article A Novel Targeted T-Cell Modulator, Efalizumab, for laque soriasis Mark Lebwohl, M.D., Stephen K. Tyring, M.D., h.d., Tiffani K. Hamilton, M.D., Darryl
The new england journal of medicine original article A Novel Targeted T-Cell Modulator, Efalizumab, for laque soriasis Mark Lebwohl, M.D., Stephen K. Tyring, M.D., h.d., Tiffani K. Hamilton, M.D., Darryl
Regulatory Status FDA-approved indication: Enbrel is a tumor necrosis factor (TNF) blocker indicated for the treatment of:
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Section: Prescription Drugs Effective Date: Oct 1, 2016 Subject: Enbrel Page: 1 of 7 Last Review Date:
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Section: Prescription Drugs Effective Date: Oct 1, 2016 Subject: Enbrel Page: 1 of 7 Last Review Date:
2017 Blue Cross and Blue Shield of Louisiana
 Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Biological treatments for psoriasis
 REVIEW Biological treatments for psoriasis Sanjay M Rajpara, Anthony D Ormerod & Pick N Woo Author for correspondence Aberdeen Royal Infirmary, Foresterhill, Aberdeen, AB25 2ZN, UK Tel.: +44 122 455 3955
REVIEW Biological treatments for psoriasis Sanjay M Rajpara, Anthony D Ormerod & Pick N Woo Author for correspondence Aberdeen Royal Infirmary, Foresterhill, Aberdeen, AB25 2ZN, UK Tel.: +44 122 455 3955
Cosentyx. Cosentyx (secukinumab) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.90.11 Subject: Cosentyx Page: 1 of 7 Last Review Date: September 20, 2018 Cosentyx Description Cosentyx
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.90.11 Subject: Cosentyx Page: 1 of 7 Last Review Date: September 20, 2018 Cosentyx Description Cosentyx
Etanercept: a new option in paediatric plaque psoriasis
 : a new option in paediatric plaque psoriasis Steve Chaplin MSc, MRPharmS, Medical Writer, Dr David Atherton MA, MB, BChir, FRCP, Honorary Consultant in Paediatric Dermatology, Great Ormond Street Hospital
: a new option in paediatric plaque psoriasis Steve Chaplin MSc, MRPharmS, Medical Writer, Dr David Atherton MA, MB, BChir, FRCP, Honorary Consultant in Paediatric Dermatology, Great Ormond Street Hospital
PHARMACY POLICY STATEMENT Ohio Medicaid
 DRUG NAME BILLING CODE BENEFIT TYPE SITE OF SERVICE ALLOWED COVERAGE REQUIREMENTS LIST OF DIAGNOSES CONSIDERED NOT MEDICALLY NECESSARY PHARMACY POLICY STATEMENT Ohio Medicaid Enbrel (etanercept) Must use
DRUG NAME BILLING CODE BENEFIT TYPE SITE OF SERVICE ALLOWED COVERAGE REQUIREMENTS LIST OF DIAGNOSES CONSIDERED NOT MEDICALLY NECESSARY PHARMACY POLICY STATEMENT Ohio Medicaid Enbrel (etanercept) Must use
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: golimumab_simponi 8/2013 2/2018 2/2019 3/2018 Description of Procedure or Service Golimumab (Simponi and
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: golimumab_simponi 8/2013 2/2018 2/2019 3/2018 Description of Procedure or Service Golimumab (Simponi and
2. Does the patient have a diagnosis of ulcerative colitis or Crohn s? Y N
 Pharmacy Prior Authorization AETA BETTER HEALTH LOUISIAA (MEDICAID) Remicade (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign
Pharmacy Prior Authorization AETA BETTER HEALTH LOUISIAA (MEDICAID) Remicade (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign
Use of extrapolation in small clinical trials:
 Use of extrapolation in small clinical trials: Infliximab for pediatric ulcerative colitis Jessica J. Lee, MD, MMSc Medical Officer Division of Gastroenterology and Inborn Errors Products CDER/ FDA 1 Learning
Use of extrapolation in small clinical trials: Infliximab for pediatric ulcerative colitis Jessica J. Lee, MD, MMSc Medical Officer Division of Gastroenterology and Inborn Errors Products CDER/ FDA 1 Learning
Stelara. Stelara (ustekinumab) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.90.04 Subject: Stelara Page: 1 of 6 Last Review Date: December 8, 2017 Stelara Description Stelara (ustekinumab)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.90.04 Subject: Stelara Page: 1 of 6 Last Review Date: December 8, 2017 Stelara Description Stelara (ustekinumab)
Targeted Immune Modulators
 Oregon Health Resources Commission Targeted Immune Modulators Subcommittee Report Update #1, April 2007 TIMs Subcommittee Report of March 2006. All revisions are highlighted. Produced by: Health Resources
Oregon Health Resources Commission Targeted Immune Modulators Subcommittee Report Update #1, April 2007 TIMs Subcommittee Report of March 2006. All revisions are highlighted. Produced by: Health Resources
Drugs and Applicable Coding: J-code: Enbrel-J1438; Humira-J0135; Remicade-J1745; Inflectra-Q5102; Cimzia-J0718; Simponi-J1602 Renflexis - pending
 Policy Subject: Anti-TNF Agents Policy Number: SHS PBD16 Category: Rheumatology & Autoimmune Policy Type: Medical Pharmacy Department: Pharmacy Product (check all that apply): Group HMO/POS Individual
Policy Subject: Anti-TNF Agents Policy Number: SHS PBD16 Category: Rheumatology & Autoimmune Policy Type: Medical Pharmacy Department: Pharmacy Product (check all that apply): Group HMO/POS Individual
STUDY. Extent and Clinical Consequences of Antibody Formation Against Adalimumab in Patients With Plaque Psoriasis
 STUDY Extent and Clinical Consequences of Antibody Formation Against Adalimumab in Patients With Plaque Psoriasis Lidian L. A. Lecluse, MD; Rieke J. B. Driessen, MD; Phyllis I. Spuls, MD, PhD; Elke M.
STUDY Extent and Clinical Consequences of Antibody Formation Against Adalimumab in Patients With Plaque Psoriasis Lidian L. A. Lecluse, MD; Rieke J. B. Driessen, MD; Phyllis I. Spuls, MD, PhD; Elke M.
Are Biologicals Safe Enough?
 Are Biologicals Safe Enough? Dr Anne Duggan Director of Gastroenterology, John Hunter Hospital, Hunter New England Area Health Service (HNEAHS) Conjoint A/Professor, University of Newcastle, Australia
Are Biologicals Safe Enough? Dr Anne Duggan Director of Gastroenterology, John Hunter Hospital, Hunter New England Area Health Service (HNEAHS) Conjoint A/Professor, University of Newcastle, Australia
SCIENTIFIC DISCUSSION
 European Medicines Agency London, 20 September 2007 Product name: Remicade Procedure number: EMEA/H/C/240/II/95 SCIENTIFIC DISCUSSION 7 Westferry Circus, Canary Wharf, London, E14 4HB, UK Tel. (44-20)
European Medicines Agency London, 20 September 2007 Product name: Remicade Procedure number: EMEA/H/C/240/II/95 SCIENTIFIC DISCUSSION 7 Westferry Circus, Canary Wharf, London, E14 4HB, UK Tel. (44-20)
Clinical Policy: Ustekinumab (Stelara) Reference Number: CP.PHAR.264
 Clinical Policy: (Stelara) Reference Number: CP.PHAR.264 Effective Date: 08/16 Last Review Date: 05/16 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
Clinical Policy: (Stelara) Reference Number: CP.PHAR.264 Effective Date: 08/16 Last Review Date: 05/16 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
STELARA (USTEKINUMAB)
 Oxford STELARA (USTEKINUMAB) UnitedHealthcare Oxford Clinical Policy Policy Number: PHARMACY 218.13 T2 Effective Date: February 1, 2017 Table of Contents Page INSTRUCTIONS FOR USE... 1 CONDITIONS OF COVERAGE...
Oxford STELARA (USTEKINUMAB) UnitedHealthcare Oxford Clinical Policy Policy Number: PHARMACY 218.13 T2 Effective Date: February 1, 2017 Table of Contents Page INSTRUCTIONS FOR USE... 1 CONDITIONS OF COVERAGE...
