Table S1: ICD-9-CM codes for patient enrollment from HIS (main or secondary diagnoses)
|
|
|
- Helena Shepherd
- 5 years ago
- Views:
Transcription
1 Appendix (Table S1-S16) Table S1: ICD-9-CM codes for patient enrollment from HIS (main or secondary diagnoses) ICD-9-CM CODES DISEASE 555 Crohn s disease 556 (EXCLUDING: 556.0, 566.1, 556.4,556.8) Ulcerative colitis Table S2: Codes for patient enrollment from copayment exemption register CODES DISEASE Crohn s disease Ulcerative colitis Table S3: ICD-9-CM codes for exclusion criteria from diagnosis reported in discharge abstract (main or secondary) ICD-9-CM CODES DISEASE 714 Rheumatoid arthritis Psoriatic arthritis Ankylosing spondylitis Table S4: ATC codes for exclusion criteria from drug utilization in the year before index date: ATC Active agent Therapeutic area L01XC02 Rituximab Rheumatoid arthritis, Leukemia, Lymphocytic Chronic B- Cell,Non-Hodgkin lymphoma L04AA13 Leflunomide Psoriatic arthritis, Rheumatoid arthritis L04AA24 Abatacept Juvenile rheumatoid arthritis, Rheumatoid arthritis, L04AB01 Etanercept Juvenile rheumatoid arthritis, Psoriatic arthritis, Rheumatoid arthritis, Psoriasis, Ankylosing spondylitis, L04AB05 Certolizumab pegol Rheumatoid arthritis, L04AC03 Anakinra Rheumatoid arthritis, L04AC07 Tocilizumab Juvenile rheumatoid arthritis, Rheumatoid arthritis
2 Table S5: ICD-9-CM codes for comorbid conditions, registered in the 12 months preceding the index date ICD-9-cm codes Co-morbidity Ischemic disease 428 Congestive Heart failure Cerebrovascular Diseases Dementia Peripheral vascular disease Diabetes , 311, , 300.4, 309.0, Anxiety 334.1,336.2,348.1,3483,784.3,333.4,333.5, , Neurological disease 331,332,334,335, 341, 345, , , 311, , 293.8, 299.1, 331.0, Psychiatric 294.1, 300.4, 309.0, , V42.0, V45.1, procedures Renal disease (38.95,39.95,54.98,55.6) Liver Disease Hypertension
3 Table S6: ICD-9-CM codes used to characterize disease course, registered in the 12 months preceding the index date Condition ICD9cm code Description Anemia / malabsorption Secondary to blood loss (chronic) Iron deficiency anemia, unspecified Unspecified intestinal malabsorption Intestinal stenosis / obstruction Stenosis of rectum and anus Acute pseudo-obstruction of intestine Mural thickening causing obstruction Unspecified intestinal obstruction Stenosis of rectum and anus Other obstruction of duodenum Atresia and stenosis of large intestine, rectum, and anal canal Intestinal fistula / abscess/ ulcer Fistula of stomach or duodenum Fistula of intestine, excluding rectum and anus Fistula of bile duct Fistula of gallbladder Intestinoureteral fistula Urethral fistula Anal fistula 619 Fistula involving female genital tract Intestinovesical fistula 566 Abscess of anal and rectal regions Abscess of intestine Ulceration of intestine Ulcer of anus and rectum Perforation of intestine Haemorrhage 578 Gastrointestinal hemorrhage Hemorrhage of gastrointestinal tract, unspecified Hemorrhage of rectum and anus Blood in stool
4 Table S7: ATC codes for drug prescription patterns, registered in the year preceding the index date Drug Class Generic Name ATC code Antimetabolites Mercaptopurine L01BB02 Methotrexate L01BA01 Immunosuppressants Azathioprine L04AX01 Intestinal Antiinfectives Rifaximin A07AA11 Steroids Corticosteroids acting locally A07EA Corticosteroids for systemic use H02 Aminosalicylic acid and similar agents Mesalamine A07EC02 Sulfasalazine Balsalazide A07EC01 A07EC04 Antibacterials Metronidazole J01XD01 Ciprofloxacin J01MA02
5 Table S8: ICD-9-CM procedure codes for IBD-related bowel surgery used as effectiveness outcomes ICD-9-CM codes Procedure 458 Tot Intra-Abd Colectomy 485 Abd-Perineal Rect Resect 4561 Mult Seg Sm Bowel Excis 4562 Part Sm Bowel Resect Nec 4563 Total Removal Sm Bowel 4571 Mult Seg Lg Bowel Excis 4572 Cecectomy 4573 Right Hemicolectomy 4574 Transverse Colon Resect 4575 Left Hemicolectomy 4576 Sigmoidectomy 4579 Part Lg Bowel ExcisNec 4590 Intestinal Anastom Nos 4591 Sm-To-Sm Bowel Anastom 4592 Sm Bowel-Rect Stump Anas 4593 Small-To-Large Bowel Nec 4594 Lg-To-Lg Bowel Anastom 4595 Anal Anastomosis 4610 Colostomy Nos 4611 Temporary Colostomy 4613 Permanent Colostomy 4614 Delay Opening Colostomy 4620 Ileostomy Nos 4621 Temporary Ileostomy 4622 Continent Ileostomy 4623 Permanent Ileostomy Nec 4624 Delay Opening Ileostomy 4841 Soave SubmucRect Resect 4849 Pull-Thru RectResecNec 4861 TranssacRectosigmoidect 4862 Ant Rect Resect W Colost 4863 Anterior Rect Resect Nec 4864 Posterior Rect Resection 4865 Duhamel Rectal Resection 4869 Rectal Resection Nec
6 Table S9: List of serious and opportunistic infections used as safety outcome, registered in discharge abstract (principal or secondary diagnosis) after index date and throughout followup. ICD-9-CM codes Description Tuberculosis 31 Diseases due to other mycobacteria 36 Meningococcal infection 38 Septicemia 42 Human immunodeficiency virus [HIV] disease Subacute sclerosing panencephalitis Progressive multifocal leukoencephalopathy 53 Herpes zoster 54 Herpes simplex 75 Infectious mononucleosis Cytomegaloviral disease convert to ICD-10-CM Retrovirus in conditions classified elsewhere and of unspecified site Respiratory syncytial virus (RSV) convert to ICD-10-CM Other specified viral and chlamydial infections Mycoses 130 Toxoplasmosis 131 Trichomoniasis
7 Table S10: Patients discharged from hospital after index date and throughout follow-up with diagnosis (principal or secondary) of any of the following conditions. Disease CD CU Condition Infusion reaction (ICD-9-CM codes: 995.0, 995.2, 995.3) Dermomiosytis (ICD-9-CM codes: 710.3) Brain neoplasm (ICD-9-CM codes: 191.9) Acquired hemolytic anemias (ICD-9-CM codes: 283.0, 283.1, , , , 283.2, 283.9) IFX (N=367) ADA (N=505) IFX (N=469) ADA (N=91) Sum % Sum % Sum % Sum %
8 Figure 1: Unadjusted time to event analysis, Kaplan-Meier survivor curves for drug discontinuation or switch*, among CD or UC patients. As treated study design. Drug discontinuation or switch Crohn's disease Proportion of patients free of events Log-rank test Pr>chi2 < ADA IFX Time (years) Number at risk (events) 505 (35) 470 (70) 400 (44) 319 (41) (33) 334 (30) 304 (21) 259 (14) 216 exp = ADA exp = IFX Drug discontinuation or switch Ulcerative colitis Proportion of patients free of events Log-rank test Pr>chi2 < ADA IFX Time (years) Number at risk (events) 90 (12) 78 (18) 60 (10) 43 (11) (28) 439 (24) 415 (23) 362 (12) 310 exp = ADA exp = IFX * Whichever comes first. IFX: Infliximab, ADA: Adalimumab.
9 Table S11: Patient characteristics included in the predictive model (Intention to treat study design): Results of stepwise Cox regression models. Hazard ratios for abdominal surgery and serious and opportunistic infections among Crohn s disease patients Characteristic HR (Abdominal surgery) LCL UCL P-Value Gender Male Female Age class (years) > Intestinal obstruction/stenosis No Yes Previous surgery No Yes Number of comorbidities None or more Characteristic HR (Infections) LCL UCL P-Value Intestinal fistola/ulcera/abscess No Yes Number of comorbidities None or more
10 Table S12: Patient characteristics included in the predictive model (Intention to treat study design): Results of stepwise Cox regression models. Hazard ratios for corticosteroids utilization (60 days after index date) among Crohn s disease patients Characteristic Remote or no users Previous steroid utilization (within 365 days before index date) Recent users HR* LCL UCL P-Value HR* LCL UCL P-Value Time from last evidence of IBD from HIS to index date More than 5 years before index date Within 5 years before index date Number of comorbidities None or more Anemia/hemorrage No Yes Intestinal obstruction/stenosis No Yes Age class (years) > Previous aminosalicylic acid prescription Remote or no user Recent user * HR of corticosteroids utilization (60 days after index date) Previous drugs utilization (within 365 days before index date) - Recent: last prescription <= 6 months before index date - Remote or no users: last prescription > 6 months before index date or no prescriptions
11 Table S13: Patient characteristics included in the predictive model (Intention to treat study design): Results of stepwise Cox regression models. Hazard ratios for corticosteroids utilization (60 days after index date) among Ulcerative colitis patients Remote or no users Previous steroid utilization (within 365 days before index date) Recent users Characteristic HR* LCL UCL P-Value HR* LCL UCL P-Value Place of residence Rome municipality Rome province Other Lazio provinces Age class (years) > Number of comorbidities None or more Previous immunosuppressant prescription Remote or no user Recent user Anemia/hemorrage No Yes * HR of corticosteroids utilization (60 days after index date) Previous drugs utilization (within 365 days before index date) - Recent: last prescription <= 6 months before index date - Remote or no users: last prescription > 6 months before index date or no prescriptions
12 Table S14: Patient characteristics included in the propensity score. Characteristics of Crohn s Disease patients according to exposure before and after adjustment for propensity score quintile. Infliximab Adalimumab n=367 n=505 p-value After adjustement for propensity score quintile N % N % p-value Gender Male Female Age class (years) > Residence Rome municipality Rome province Other Lazio provinces CD disease form (ICD9-CM code) Regional enteritis of small intestine (555.0) Regional enteritis of large intestine (555.1) Regional enteritis of small intestine with large intestine (555.2) Regional enteritis of unspecified site (555.9) CU diagnosis in CD patients Patients not discharge within 5 years before index date Time from last evidence of IBD from HIS to index date Within 5 years before index date More than 5 years before index date Risk factors for surgery Intestinal fistola/ulcera/abscess Drug exposure Corticosteroid (local/systemic) No Recent (<= 6 months) Remote (>6 months)
13 Table S15: Patient characteristics included in the propensity score. Characteristics of Ulcerative colitis patients according to exposure before and after adjustment for propensity quintile score Infliximab Adalimumab n=467 n=90 p-value After adjustement for propensity score quintile N % N % p-value Gender Male Female Age class > Residence Rome municipality Rome province Other Lazio provinces UC disease form (ICD9-CM code) Ulcerative enterocolitis/ulcerative ileocolitis/pseudopolyposis of colon (556.0/ 556.1/ 556.4) Ulcerative proctitis/ulcerative proctosigmoiditis (556.2/ 556.3) Left-sided ulcerative colitis (556.5) Pancolitis (556.6) Other ulcerative colitis/ulcerative colitis, unspecified (556.8) CD diagnosis in CU patients Patients not discharged within 5 year before index date Risk factors for surgery Anemia/hemorrage Drug exposure Aminosalicylic acid No Remote (>6 months) Recent (<= 6 months) Immunosuppressant No Remote (>6 months) Recent (<= 6 months)
14 Table S16: Rates and HR of abdominal surgery and infections among patients of UC treated with Infliximab versus those treated with Adalimumab. Intention to treat approach. Disease Outcome UC Events IFX ADA Crude HR Adjusted HR Rate (per 100 PY) Events Rate (per 100 PY) Estimate ICL ICU P Value Estimate ICL ICU P Value Surgery Infections IFX: Infliximab, ADA: Adalimumab, CD: Crohn sdisease, UC: Ulcerative colitis.py 100 person years. ICL / ICU: 95% lower / upper confidence limits.
INFLIXIMAB Remicade (infliximab), Inflectra (infliximab-dyyb), Ixifi* (infliximabqbtx), Renflexis (infliximab-abda)
 Pre - PA Allowance None Prior-Approval Requirements Diagnoses Patient must have ONE of the following: 6 years of age or older 1. Moderate to severe Crohn s disease (CD) a. Patient has fistulizing disease
Pre - PA Allowance None Prior-Approval Requirements Diagnoses Patient must have ONE of the following: 6 years of age or older 1. Moderate to severe Crohn s disease (CD) a. Patient has fistulizing disease
SUPPLEMENTAL MATERIAL
 SUPPLEMENTAL MATERIAL efigures efigure: Standardised differences between TNF-α inhibitor users and non-users before and after matching on fixed factors and propensity scores. etables etable 1: All diagnoses
SUPPLEMENTAL MATERIAL efigures efigure: Standardised differences between TNF-α inhibitor users and non-users before and after matching on fixed factors and propensity scores. etables etable 1: All diagnoses
What prescribers need to know
 HUMIRA Citrate-free presentations in an Electronic Medical Record (EMR) What prescribers need to know 2 / This is your guide to identifying HUMIRA Citrate-free presentations in your Electronic Medical
HUMIRA Citrate-free presentations in an Electronic Medical Record (EMR) What prescribers need to know 2 / This is your guide to identifying HUMIRA Citrate-free presentations in your Electronic Medical
Surgical Management of IBD. Val Jefford Grand Rounds October 14, 2003
 Surgical Management of IBD Val Jefford Grand Rounds October 14, 2003 Introduction Important Features Clinical Presentation Evaluation Medical Treatment Surgical Treatment Cases Overview Introduction Two
Surgical Management of IBD Val Jefford Grand Rounds October 14, 2003 Introduction Important Features Clinical Presentation Evaluation Medical Treatment Surgical Treatment Cases Overview Introduction Two
Crohn s Disease. Resident Lecture 1/17/19
 Crohn s Disease Resident Lecture 1/17/19 Objectives Features/Classification of Crohn s Disease Medical Treatment Surgical Indications Surgical Considerations 2 Case 25 yo F presents to your office with
Crohn s Disease Resident Lecture 1/17/19 Objectives Features/Classification of Crohn s Disease Medical Treatment Surgical Indications Surgical Considerations 2 Case 25 yo F presents to your office with
C. Assess clinical response after the first three months of treatment.
 Government Health Plan (GHP) of Puerto Rico Authorization Criteria Tumor Necrosis Factor Alpha (TNFα) Adalimumab (Humira ) Managed by MCO Section I. Prior Authorization Criteria A. Physician must submit
Government Health Plan (GHP) of Puerto Rico Authorization Criteria Tumor Necrosis Factor Alpha (TNFα) Adalimumab (Humira ) Managed by MCO Section I. Prior Authorization Criteria A. Physician must submit
Index. Surg Clin N Am 87 (2007) Note: Page numbers of article titles are in boldface type.
 Surg Clin N Am 87 (2007) 787 796 Index Note: Page numbers of article titles are in boldface type. A Abscesses in anorectal Crohn s disease, 622 intra-abdominal, in Crohn s disease, 590 591 perirectal,
Surg Clin N Am 87 (2007) 787 796 Index Note: Page numbers of article titles are in boldface type. A Abscesses in anorectal Crohn s disease, 622 intra-abdominal, in Crohn s disease, 590 591 perirectal,
1 P a g e. Systemic Juvenile Idiopathic Arthritis (SJIA) (1.3) Patients 2 years of age and older with active systemic juvenile idiopathic arthritis.
 LENGTH OF AUTHORIZATION: Initial: 3 months for Crohn s or Ulcerative Colitis; 1 year for all other indications. Renewal: 1 year dependent upon medical records supporting response to therapy and review
LENGTH OF AUTHORIZATION: Initial: 3 months for Crohn s or Ulcerative Colitis; 1 year for all other indications. Renewal: 1 year dependent upon medical records supporting response to therapy and review
Garrick Brown, MD. Digestive Health Specialists Tacoma Gig Harbor
 Garrick Brown, MD Digestive Health Specialists Tacoma Gig Harbor Today s Objectives Define IBD, its potential causes and diagnosis Discuss management and treatment Discuss complementary and alternative
Garrick Brown, MD Digestive Health Specialists Tacoma Gig Harbor Today s Objectives Define IBD, its potential causes and diagnosis Discuss management and treatment Discuss complementary and alternative
Understanding Inflammatory Bowel Diseases (IBD):
 Understanding Inflammatory Bowel Diseases (IBD): What Every Patient Needs to Know William H Holderman, MD Digestive Health Specialists Tacoma, WA Today s Objectives Define IBD, its potential causes and
Understanding Inflammatory Bowel Diseases (IBD): What Every Patient Needs to Know William H Holderman, MD Digestive Health Specialists Tacoma, WA Today s Objectives Define IBD, its potential causes and
CIMZIA (certolizumab pegol)
 Pre - PA Allowance None Prior-Approval Requirements Age Diagnoses 18 years of age or older Patient must have ONE of the following: 1. Moderate to severe Crohn s Disease (CD) a. Inadequate response, intolerance
Pre - PA Allowance None Prior-Approval Requirements Age Diagnoses 18 years of age or older Patient must have ONE of the following: 1. Moderate to severe Crohn s Disease (CD) a. Inadequate response, intolerance
2. Has the patient had a response to treatment? Y N. 3. Does the patient have a diagnosis of rheumatoid arthritis (RA)? Y N
 12/21/2016 Prior Authorization Aetna Better Health of West Virginia Humira (WV88) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign and
12/21/2016 Prior Authorization Aetna Better Health of West Virginia Humira (WV88) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign and
Circle Yes or No Y N. [If no, skip to question 7.] 2. Does the patient have a diagnosis of ulcerative colitis? Y N. [If no, skip to question 4.
![Circle Yes or No Y N. [If no, skip to question 7.] 2. Does the patient have a diagnosis of ulcerative colitis? Y N. [If no, skip to question 4. Circle Yes or No Y N. [If no, skip to question 7.] 2. Does the patient have a diagnosis of ulcerative colitis? Y N. [If no, skip to question 4.](/thumbs/85/91758188.jpg) 06/01/2016 Prior Authorization AETA BETTER HEALTH OF MICHIGA (MEDICAID) Humira (MI88) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign
06/01/2016 Prior Authorization AETA BETTER HEALTH OF MICHIGA (MEDICAID) Humira (MI88) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign
Immune Modulating Drugs Prior Authorization Request Form
 Patient: HPHC member ID #: Requesting provider: Phone: Servicing provider: Diagnosis: Contact for questions (name and phone #): Projected start and end date for requested Requesting provider NPI: Fax:
Patient: HPHC member ID #: Requesting provider: Phone: Servicing provider: Diagnosis: Contact for questions (name and phone #): Projected start and end date for requested Requesting provider NPI: Fax:
Biologics for Autoimmune Diseases
 Biologics for Autoimmune Diseases Goal(s): Restrict use of biologics to OHP funded conditions and according to OHP guidelines for use. Promote use that is consistent with national clinical practice guidelines
Biologics for Autoimmune Diseases Goal(s): Restrict use of biologics to OHP funded conditions and according to OHP guidelines for use. Promote use that is consistent with national clinical practice guidelines
FY 2016 MCRCEDP Approved ICD-10 Code List
 Approved List C18.0 Malignant neoplasm of cecum C18.1 Malignant neoplasm of appendix C18.2 Malignant neoplasm of ascending colon C18.3 Malignant neoplasm of hepatic flexure C18.4 Malignant neoplasm of
Approved List C18.0 Malignant neoplasm of cecum C18.1 Malignant neoplasm of appendix C18.2 Malignant neoplasm of ascending colon C18.3 Malignant neoplasm of hepatic flexure C18.4 Malignant neoplasm of
The Spectrum of IBD. Inflammatory Bowel Disease. Symptoms. Epidemiology. Tests for IBD. CD or UC? Inflamatory Bowel Disease. Fernando Vega, M.D.
 The Spectrum of IBD Inflammatory Bowel Disease Fernando Vega, M.D. Epidemiology CD and UC together 1:400 UC Prevalence 1:500 UC Incidence 6-12K/annum CD Prevalence 1:1000 CD Incidence 3-6K/annum Symptoms
The Spectrum of IBD Inflammatory Bowel Disease Fernando Vega, M.D. Epidemiology CD and UC together 1:400 UC Prevalence 1:500 UC Incidence 6-12K/annum CD Prevalence 1:1000 CD Incidence 3-6K/annum Symptoms
Adult organisational audit
 Adult organisational audit Health Information Coding Responses for this section require data for the period between 1/12/13-30/11/14. [Infliximab was introduced for the treatment of ulcerative colitis
Adult organisational audit Health Information Coding Responses for this section require data for the period between 1/12/13-30/11/14. [Infliximab was introduced for the treatment of ulcerative colitis
Hu J, Gonsahn MD, Nerenz DR. Socioeconomic status and readmissions: evidence from an urban teaching hospital. Health Aff (Millwood). 2014;33(5).
 Appendix Definitions of Index Admission and Readmission Definitions of index admission and readmission follow CMS hospital-wide all-cause unplanned readmission (HWR) measure as far as data are available.
Appendix Definitions of Index Admission and Readmission Definitions of index admission and readmission follow CMS hospital-wide all-cause unplanned readmission (HWR) measure as far as data are available.
NCD for Fecal Occult Blood Test
 NCD for Fecal Occult Blood Test Applicable CPT Code(s): 82272 Blood, occult, by peroxidase activity (e.g., guaiac), qualitative, feces, 1-3 simultaneous determinations, performed for other than colorectal
NCD for Fecal Occult Blood Test Applicable CPT Code(s): 82272 Blood, occult, by peroxidase activity (e.g., guaiac), qualitative, feces, 1-3 simultaneous determinations, performed for other than colorectal
Inflammatory Bowel Disease and Surgery: What You Should Know
 Inflammatory Bowel Disease and Surgery: What You Should Know Ask the Experts March 9, 2019 Kristen Blaker, MD Colon and Rectal Surgery MetroHealth Medical Center Disclosures None Outline Who undergoes
Inflammatory Bowel Disease and Surgery: What You Should Know Ask the Experts March 9, 2019 Kristen Blaker, MD Colon and Rectal Surgery MetroHealth Medical Center Disclosures None Outline Who undergoes
Treatment of Inflammatory Bowel Disease. Michael Weiss MD, FACG
 Treatment of Inflammatory Bowel Disease Michael Weiss MD, FACG What is IBD? IBD is an immune-mediated chronic intestinal disorder, characterized by chronic or relapsing inflammation within the GI tract.
Treatment of Inflammatory Bowel Disease Michael Weiss MD, FACG What is IBD? IBD is an immune-mediated chronic intestinal disorder, characterized by chronic or relapsing inflammation within the GI tract.
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION. 3 October 2012
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 3 October 2012 REMICADE 100 mg, powder for concentrate for solution for infusion B/1 vial (CIP code: 562 070-1) Applicant:
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 3 October 2012 REMICADE 100 mg, powder for concentrate for solution for infusion B/1 vial (CIP code: 562 070-1) Applicant:
Remicade (infliximab) DRUG.00002
 Applicability/Effective Date *- Florida Healthy Kids Remicade (infliximab) DRUG.00002 Override(s) Prior Authorization Step Therapy Medications Remicade (infliximab) Approval Duration 1 year Comment Intravenous
Applicability/Effective Date *- Florida Healthy Kids Remicade (infliximab) DRUG.00002 Override(s) Prior Authorization Step Therapy Medications Remicade (infliximab) Approval Duration 1 year Comment Intravenous
Welcome to Week 2 of the Crohn s & Colitis Foundation of America (CCFA) Online Support Group.
 Welcome to Week 2 of the Crohn s & Colitis Foundation of America (CCFA) Online. Last week s material consisted of an overview of inflammatory bowel diseases (IBD), specifically Crohn s disease and ulcerative
Welcome to Week 2 of the Crohn s & Colitis Foundation of America (CCFA) Online. Last week s material consisted of an overview of inflammatory bowel diseases (IBD), specifically Crohn s disease and ulcerative
Regulatory Status FDA-approved indications: Entyvio is an α4β7integrin receptor antagonist indicated for: (1)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.12 Subject: Entyvio Page: 1 of 7 Last Review Date: September 20, 2018 Entyvio Description Entyvio
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.12 Subject: Entyvio Page: 1 of 7 Last Review Date: September 20, 2018 Entyvio Description Entyvio
Ileal Pouch Anal Anastomosis: The Preferred Method of Reconstruction after Proctocolectomy in Children
 Ileal Pouch Anal Anastomosis: The Preferred Method of Reconstruction after Proctocolectomy in Children Stephanie Jones, D.O. Surgical Fellow March 21, 2011 Ulcerative Colitis Spectrum of inflammatory bowel
Ileal Pouch Anal Anastomosis: The Preferred Method of Reconstruction after Proctocolectomy in Children Stephanie Jones, D.O. Surgical Fellow March 21, 2011 Ulcerative Colitis Spectrum of inflammatory bowel
3. Did the patient show evidence of remission by week 8 of Humira Y N therapy?
 09/23/2015 Prior Authorization AETA BETTER HEALTH OF MICHIGA (MEDICAID) Humira (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information,
09/23/2015 Prior Authorization AETA BETTER HEALTH OF MICHIGA (MEDICAID) Humira (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information,
Crohn's disease CAUSES COURSE OF CROHN'S DISEASE TREATMENT. Sulfasalazine
 Crohn's disease Crohn's disease is an inflammatory condition of the digestive tract that affects children and adults. Common features of Crohn's disease include mouth sores, diarrhea, abdominal pain, weight
Crohn's disease Crohn's disease is an inflammatory condition of the digestive tract that affects children and adults. Common features of Crohn's disease include mouth sores, diarrhea, abdominal pain, weight
ULCERATIVE COLITIS. Sean Lynch, MD and Richard Bloomfeld, MD Wake Forest University School of Medicine Winston-Salem, NC
 ULCERATIVE COLITIS Sean Lynch, MD and Richard Bloomfeld, MD Wake Forest University School of Medicine Winston-Salem, NC What is Ulcerative Colitis? Ulcerative colitis (UC) is a disease marked by inflammation
ULCERATIVE COLITIS Sean Lynch, MD and Richard Bloomfeld, MD Wake Forest University School of Medicine Winston-Salem, NC What is Ulcerative Colitis? Ulcerative colitis (UC) is a disease marked by inflammation
Medical therapies and IBD
 Medical therapies and IBD Although there is no cure for IBD, there are many treatment options available. There is no standard treatment for IBD that is effective in all situations or for all patients,
Medical therapies and IBD Although there is no cure for IBD, there are many treatment options available. There is no standard treatment for IBD that is effective in all situations or for all patients,
Infliximab/Infliximab-dyyb DRUG.00002
 Infliximab/Infliximab-dyyb DRUG.00002 Override(s) Prior Authorization Step Therapy Medications Remicade (infliximab) Inflectra (inflectra-dyyb) Approval Duration 1 year Comment Intravenous administration
Infliximab/Infliximab-dyyb DRUG.00002 Override(s) Prior Authorization Step Therapy Medications Remicade (infliximab) Inflectra (inflectra-dyyb) Approval Duration 1 year Comment Intravenous administration
HCPCS Codes (Alphanumeric, CPT AMA) ICD-9-CM Codes Covered by Medicare Program
 HCPCS s (Alphanumeric, CPT AMA) 82272 Blood, occult, by peroxidase activity (e.g., guaiac), qualitative, feces, 1-3 simultaneous determinations, performed for other than colorectal neoplasm screening ICD-9-CM
HCPCS s (Alphanumeric, CPT AMA) 82272 Blood, occult, by peroxidase activity (e.g., guaiac), qualitative, feces, 1-3 simultaneous determinations, performed for other than colorectal neoplasm screening ICD-9-CM
Cimzia. Cimzia (certolizumab pegol) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Cimzia Page: 1 of 5 Last Review Date: March 17, 2017 Cimzia Description Cimzia (certolizumab pegol)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Cimzia Page: 1 of 5 Last Review Date: March 17, 2017 Cimzia Description Cimzia (certolizumab pegol)
CROHN S DISEASE. The term "inflammatory bowel disease" includes Crohn's disease and the other related condition called ulcerative colitis.
 CROHN S DISEASE What does it consist of? Crohn s disease is an inflammatory process that affects mostly to the intestinal tract, although it can affect any other part of the digestive apparatus from the
CROHN S DISEASE What does it consist of? Crohn s disease is an inflammatory process that affects mostly to the intestinal tract, although it can affect any other part of the digestive apparatus from the
Risk of serious infections associated with use of immunosuppressive agents in pregnant women with autoimmune inflammatory conditions: cohor t study
 Risk of serious infections associated with use of immunosuppressive agents in pregnant women with autoimmune inflammatory conditions: cohor t study BMJ 2017; 356 doi: https://doi.org/10.1136/bmj.j895 (Published
Risk of serious infections associated with use of immunosuppressive agents in pregnant women with autoimmune inflammatory conditions: cohor t study BMJ 2017; 356 doi: https://doi.org/10.1136/bmj.j895 (Published
Drug Class Prior Authorization Criteria Therapeutic Agents in Rheumatic and Inflammatory Diseases
 Drug Class Prior Authorization Criteria Therapeutic Agents in Rheumatic and Inflammatory Diseases Line of Business: Medicaid P & T Approval Date: August 16, 2017 Effective Date: August 16, 2017 This policy
Drug Class Prior Authorization Criteria Therapeutic Agents in Rheumatic and Inflammatory Diseases Line of Business: Medicaid P & T Approval Date: August 16, 2017 Effective Date: August 16, 2017 This policy
IBD Understanding Your Medications. Thomas V. Aguirre, MD Santa Barbara GI Consultants
 IBD Understanding Your Medications Thomas V. Aguirre, MD Santa Barbara GI Consultants IBD Understanding Your Medications (& Your Doctor) Thomas V. Aguirre, MD Santa Barbara GI Consultants Disclosure I
IBD Understanding Your Medications Thomas V. Aguirre, MD Santa Barbara GI Consultants IBD Understanding Your Medications (& Your Doctor) Thomas V. Aguirre, MD Santa Barbara GI Consultants Disclosure I
Drug Effectiveness Review Project Summary Report Biologics (Targeted Immune Modulators)
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
How do I choose amongst medicines for inflammatory bowel disease. Maria T. Abreu, MD
 How do I choose amongst medicines for inflammatory bowel disease Maria T. Abreu, MD Overview of IBD Pathogenesis Bacterial Products Moderately Acutely Inflamed Chronic Inflammation = IBD Normal Gut Mildly
How do I choose amongst medicines for inflammatory bowel disease Maria T. Abreu, MD Overview of IBD Pathogenesis Bacterial Products Moderately Acutely Inflamed Chronic Inflammation = IBD Normal Gut Mildly
First Name. Specialty: Fax. First Name DOB: Duration:
 Prescriber Information Last ame: First ame DEA/PI: Specialty: Phone - - Fax - - Member Information Last ame: First ame Member ID umber DOB: - - Medication Information: Drug ame and Strength: Diagnosis:
Prescriber Information Last ame: First ame DEA/PI: Specialty: Phone - - Fax - - Member Information Last ame: First ame Member ID umber DOB: - - Medication Information: Drug ame and Strength: Diagnosis:
INFLAMMATORY BOWEL DISEASE (IBD): CROHN S DISEASE
 INFLAMMATORY BOWEL DISEASE (IBD): CROHN S DISEASE Symptoms The symptoms of Crohn s disease can vary from person to person, based on where the disease is in the body and how bad the inflammation is. The
INFLAMMATORY BOWEL DISEASE (IBD): CROHN S DISEASE Symptoms The symptoms of Crohn s disease can vary from person to person, based on where the disease is in the body and how bad the inflammation is. The
Paediatric Organisational Audit
 Paediatric Organisational Audit Health Information Coding Responses for this section require data for the period between 1/12/13-30/11/14. [Infliximab was introduced for the treatment of ulcerative colitis
Paediatric Organisational Audit Health Information Coding Responses for this section require data for the period between 1/12/13-30/11/14. [Infliximab was introduced for the treatment of ulcerative colitis
Center for Evidence-based Policy
 P&T Committee Brief Targeted Immune Modulators: Comparative Drug Class Review Alison Little, MD Center for Evidence-based Policy Oregon Health & Science University 3455 SW US Veterans Hospital Road, SN-4N
P&T Committee Brief Targeted Immune Modulators: Comparative Drug Class Review Alison Little, MD Center for Evidence-based Policy Oregon Health & Science University 3455 SW US Veterans Hospital Road, SN-4N
What is Crohn's disease?
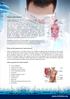 What is Crohn's disease? Crohn's disease is a chronic inflammatory disorder that causes inflammation of the digestive tract. It can affect any area of the GI tract, from the mouth to the anus, but it most
What is Crohn's disease? Crohn's disease is a chronic inflammatory disorder that causes inflammation of the digestive tract. It can affect any area of the GI tract, from the mouth to the anus, but it most
HARVARD PILGRIM HEALTH CARE RECOMMENDED MEDICATION REQUEST GUIDELINES HUMIRA PEDIATRIC
 Generic Brand HICL GCN Exception/Other ADALIMUMAB HUMIRA 24800 HUMIRA PEDIATRIC GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Is the patient currently taking Humira? If
Generic Brand HICL GCN Exception/Other ADALIMUMAB HUMIRA 24800 HUMIRA PEDIATRIC GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Is the patient currently taking Humira? If
HELPING YOU AND YOUR PATIENTS TALK OPENLY ABOUT MODERATELY TO SEVERELY ACTIVE RA
 SIMPONI ARIA (golimumab) is indicated for the treatment of adults with moderately to severely active rheumatoid arthritis (RA) in combination with MTX, active psoriatic arthritis, and active ankylosing
SIMPONI ARIA (golimumab) is indicated for the treatment of adults with moderately to severely active rheumatoid arthritis (RA) in combination with MTX, active psoriatic arthritis, and active ankylosing
CLINICAL MEDICAL POLICY
 Policy Name: Policy Number: Approved By: CLINICAL MEDICAL POLICY Remicade (Infliximab) MP-026-MD-DE Provider Notice Date: 11/1/2016 Original Effective Date: 12/1/2016 Annual Approval Date: 9/17/2017 Revision
Policy Name: Policy Number: Approved By: CLINICAL MEDICAL POLICY Remicade (Infliximab) MP-026-MD-DE Provider Notice Date: 11/1/2016 Original Effective Date: 12/1/2016 Annual Approval Date: 9/17/2017 Revision
Cimzia. Cimzia (certolizumab pegol) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.11 Section: Prescription Drugs Effective Date: April 1, 2018 Subject: Cimzia Page: 1 of 5 Last Review
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.11 Section: Prescription Drugs Effective Date: April 1, 2018 Subject: Cimzia Page: 1 of 5 Last Review
2. Is the patient responding to Remicade therapy? Y N
 09/29/2015 Prior Authorization AETA BETTER HEALTH OF MICHIGA (MEDICAID) Remicade (MI88) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign
09/29/2015 Prior Authorization AETA BETTER HEALTH OF MICHIGA (MEDICAID) Remicade (MI88) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign
Regulatory Status FDA- approved indication: Simponi and Simponi ARIA are tumor necrosis factor (TNF) blockers indicated for the treatment of: (2-3)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.51 Subject: Simponi / Simponi ARIA Page: 1 of 9 Last Review Date: March 16, 2018 Simponi / Simponi
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.51 Subject: Simponi / Simponi ARIA Page: 1 of 9 Last Review Date: March 16, 2018 Simponi / Simponi
Colorectal Laparoscopic Standards and Coding Protocols July 2015 v2.0
 Laparoscopic Standards and Coding Protocols July 2015 v2.0 COLORECTAL LAPAROSCOPIC STANDARDS AND CODING PROTOCOLS Contents 1 Context... 3 2 Laparoscopic Standards... 3 3 Coding Protocols... 3 Appendix
Laparoscopic Standards and Coding Protocols July 2015 v2.0 COLORECTAL LAPAROSCOPIC STANDARDS AND CODING PROTOCOLS Contents 1 Context... 3 2 Laparoscopic Standards... 3 3 Coding Protocols... 3 Appendix
Inflectra (infliximab-dyyb), Remicade (infliximab), Renflexis (infliximab-abda) DRUG CG-DRUG-64
 Inflectra (infliximab-dyyb), Remicade (infliximab), Renflexis (infliximab-abda) DRUG.00002 CG-DRUG-64 Override(s) Prior Authorization *Washington Medicaid See State Specific Mandates Medications Inflectra
Inflectra (infliximab-dyyb), Remicade (infliximab), Renflexis (infliximab-abda) DRUG.00002 CG-DRUG-64 Override(s) Prior Authorization *Washington Medicaid See State Specific Mandates Medications Inflectra
Indications for use of Infliximab
 Indications for use of Infliximab Moscow, June 10 th 2006 Prof. Dr. Dr. Gerhard Rogler Klinik und Poliklinik für Innere Medizin I Universität Regensburg Case report 1989: Diagnosis of Crohn s disease of
Indications for use of Infliximab Moscow, June 10 th 2006 Prof. Dr. Dr. Gerhard Rogler Klinik und Poliklinik für Innere Medizin I Universität Regensburg Case report 1989: Diagnosis of Crohn s disease of
1. Background: Infliximab is administered parenterally; therefore, it is not covered under retail pharmacy benefits.
 Subject: Infliximab (Remicade ) Original Original Committee Approval: October 13, 2006 Revised Last Committee Approval: December 3, 2008 Last Review: October 19, 2007 1. Background: Infliximab is a genetically
Subject: Infliximab (Remicade ) Original Original Committee Approval: October 13, 2006 Revised Last Committee Approval: December 3, 2008 Last Review: October 19, 2007 1. Background: Infliximab is a genetically
Cimzia. Cimzia (certolizumab pegol) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.11 Subject: Cimzia Page: 1 of 5 Last Review Date: December 8, 2017 Cimzia Description Cimzia (certolizumab
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.11 Subject: Cimzia Page: 1 of 5 Last Review Date: December 8, 2017 Cimzia Description Cimzia (certolizumab
Surgery for Inflammatory Bowel Disease
 Surgery for Inflammatory Bowel Disease Emily Steinhagen, MD Assistant Professor Department of Surgery, Division of Colorectal Surgery University Hospitals Cleveland Medical Center Common Questions Why
Surgery for Inflammatory Bowel Disease Emily Steinhagen, MD Assistant Professor Department of Surgery, Division of Colorectal Surgery University Hospitals Cleveland Medical Center Common Questions Why
Pharmacy Prior Authorization
 Pharmacy Prior Authorization AETA BETTER HEALTH PESLVAIA & AETA BETTER HEALTH KIDS Humira (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information,
Pharmacy Prior Authorization AETA BETTER HEALTH PESLVAIA & AETA BETTER HEALTH KIDS Humira (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information,
Optimal care during pregnancy and delivery
 Oxford Inflammatory Bowel Disease MasterClass Optimal care during pregnancy and delivery Professor Catherine Nelson-Piercy, London, UK Oxford Inflammatory Bowel Disease MasterClass Therapeutic goals in
Oxford Inflammatory Bowel Disease MasterClass Optimal care during pregnancy and delivery Professor Catherine Nelson-Piercy, London, UK Oxford Inflammatory Bowel Disease MasterClass Therapeutic goals in
Infections and Biologics
 Overview Infections and Biologics James Galloway What is the risk of infection with biologics? Are some patients at greater risk? Are some drugs safer? Case scenario You recently commenced Judith, a 54
Overview Infections and Biologics James Galloway What is the risk of infection with biologics? Are some patients at greater risk? Are some drugs safer? Case scenario You recently commenced Judith, a 54
Supplementary Online Content
 Supplementary Online Content Tran AH, Ngor EWM, Wu BU. Surveillance colonoscopy in elderly patients: a retrospective cohort study. JAMA Intern Med. Published online August 11, 2014. doi:10.1001/jamainternmed.2014.3746
Supplementary Online Content Tran AH, Ngor EWM, Wu BU. Surveillance colonoscopy in elderly patients: a retrospective cohort study. JAMA Intern Med. Published online August 11, 2014. doi:10.1001/jamainternmed.2014.3746
INFLAMMATORY BOWEL DISEASE. Jean-Paul Achkar, MD Center for Inflammatory Bowel Disease Cleveland Clinic
 INFLAMMATORY BOWEL DISEASE Jean-Paul Achkar, MD Center for Inflammatory Bowel Disease Cleveland Clinic WHAT IS INFLAMMATORY BOWEL DISEASE (IBD)? Chronic inflammation of the intestinal tract Two related
INFLAMMATORY BOWEL DISEASE Jean-Paul Achkar, MD Center for Inflammatory Bowel Disease Cleveland Clinic WHAT IS INFLAMMATORY BOWEL DISEASE (IBD)? Chronic inflammation of the intestinal tract Two related
MEDICATION GUIDE XELJANZ (ZEL JANS ) (tofacitinib)
 MEDICATION GUIDE XELJANZ (ZEL JANS ) (tofacitinib) Read this Medication Guide before you start taking XELJANZ and each time you get a refill. There may be new information. This Medication Guide does not
MEDICATION GUIDE XELJANZ (ZEL JANS ) (tofacitinib) Read this Medication Guide before you start taking XELJANZ and each time you get a refill. There may be new information. This Medication Guide does not
UnitedHealthcare Pharmacy Clinical Pharmacy Programs
 UnitedHealthcare Pharmacy Clinical Pharmacy Programs Program Number 2017 P 1041-8 Program Prior Authorization/Notification Medication Humira (adalimumab) P&T Approval Date 1/2007, 6/2008, 4/2009, 6/2009,
UnitedHealthcare Pharmacy Clinical Pharmacy Programs Program Number 2017 P 1041-8 Program Prior Authorization/Notification Medication Humira (adalimumab) P&T Approval Date 1/2007, 6/2008, 4/2009, 6/2009,
HARVARD PILGRIM HEALTH CARE RECOMMENDED MEDICATION REQUEST GUIDELINES
 Generic Brand HICL GCN Exception/Other CERTOLIZUMAB PEGOL CIMZIA 35554 GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Is the request for a patient with a diagnosis of moderate
Generic Brand HICL GCN Exception/Other CERTOLIZUMAB PEGOL CIMZIA 35554 GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Is the request for a patient with a diagnosis of moderate
11/13/11. Biologics for CD and CUC: The Impact on Surgical Outcomes. Principles of Successful Intestinal Surgery
 Biologics for CD and CUC: The Impact on Surgical Outcomes Robert R. Cima, M.D., M.A. Associate Professor of Surgery Division of Colon and Rectal Surgery Overview Antibody based medications (biologics)
Biologics for CD and CUC: The Impact on Surgical Outcomes Robert R. Cima, M.D., M.A. Associate Professor of Surgery Division of Colon and Rectal Surgery Overview Antibody based medications (biologics)
Drugs and Applicable Coding: J-code: Enbrel-J1438; Humira-J0135; Remicade-J1745; Inflectra-Q5102; Cimzia-J0718; Simponi-J1602 Renflexis - pending
 Policy Subject: Anti-TNF Agents Policy Number: SHS PBD16 Category: Rheumatology & Autoimmune Policy Type: Medical Pharmacy Department: Pharmacy Product (check all that apply): Group HMO/POS Individual
Policy Subject: Anti-TNF Agents Policy Number: SHS PBD16 Category: Rheumatology & Autoimmune Policy Type: Medical Pharmacy Department: Pharmacy Product (check all that apply): Group HMO/POS Individual
SURGICAL MANAGEMENT OF ULCERATIVE COLITIS
 SURGICAL MANAGEMENT OF ULCERATIVE COLITIS Cary B. Aarons, MD Associate Professor of Surgery Division of Colon & Rectal Surgery University of Pennsylvania AGENDA Background Diagnosis/Work-up Medical Management
SURGICAL MANAGEMENT OF ULCERATIVE COLITIS Cary B. Aarons, MD Associate Professor of Surgery Division of Colon & Rectal Surgery University of Pennsylvania AGENDA Background Diagnosis/Work-up Medical Management
Prior Authorization Conditions for Approval of Humira (adalimumab) Website Form Submit request via: Fax
 Prior Authorization Conditions for Approval of Humira (adalimumab) Website Form www.highmarkhealthoptions.com Submit request via: Fax - 1-855-476-4158 All requests for Humira (adalimumab) require a prior
Prior Authorization Conditions for Approval of Humira (adalimumab) Website Form www.highmarkhealthoptions.com Submit request via: Fax - 1-855-476-4158 All requests for Humira (adalimumab) require a prior
Understanding Learning is the first step to getting help.
 Understanding Inflammatory Bowel Disease (IBD) Learning is the first step to getting help. This booklet has been created through an educational grant provided by Fulford India Ltd., A subsidiary of Schering-Plough
Understanding Inflammatory Bowel Disease (IBD) Learning is the first step to getting help. This booklet has been created through an educational grant provided by Fulford India Ltd., A subsidiary of Schering-Plough
Inflammatory Bowel Disease
 Inflammatory Bowel Disease Inflammatory bowel disease (IBD) is a group of inflammatory conditions of the colon and small intestine. Crohn's disease andulcerative colitis are the principal types of inflammatory
Inflammatory Bowel Disease Inflammatory bowel disease (IBD) is a group of inflammatory conditions of the colon and small intestine. Crohn's disease andulcerative colitis are the principal types of inflammatory
Infliximab may be considered medically necessary as first-line therapy (ie, initial treatment) for the following condition:
 Medical Policy MP 5.01.15 Original Policy Date: February 2002 Last Review: 01/30/2018 Effective Date: 04/30/2018 Section: Prescription Related Policies 2.04.84 Measurement of Serum Antibodies to and Adalimumab
Medical Policy MP 5.01.15 Original Policy Date: February 2002 Last Review: 01/30/2018 Effective Date: 04/30/2018 Section: Prescription Related Policies 2.04.84 Measurement of Serum Antibodies to and Adalimumab
TABLE C-1 RESIDENT DEATHS, LIVE BIRTHS, FETAL, INFANT, NEONATAL, AND MATERNAL DEATHS: PENNSYLVANIA,
 TABLE C-1 RESIDENT DEATHS, LIVE BIRTHS, FETAL, INFANT, NEONATAL, AND MATERNAL DEATHS: PENNSYLVANIA, 1950-1997 - TOTAL LIVE FETAL INFANT NEONATAL MATERNAL DEATHS BIRTHS DEATHS DEATHS DEATHS DEATHS ----------------------------------------------------------------------------------
TABLE C-1 RESIDENT DEATHS, LIVE BIRTHS, FETAL, INFANT, NEONATAL, AND MATERNAL DEATHS: PENNSYLVANIA, 1950-1997 - TOTAL LIVE FETAL INFANT NEONATAL MATERNAL DEATHS BIRTHS DEATHS DEATHS DEATHS DEATHS ----------------------------------------------------------------------------------
Pharmacy Prior Authorization
 Pharmacy Prior Authorization MERC CARE PLA (MEDICAID) Humira (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign and date. Fax
Pharmacy Prior Authorization MERC CARE PLA (MEDICAID) Humira (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign and date. Fax
PRIOR AUTHORIZATION REQUEST GUIDE
 PRIOR AUTHORIZATION REQUEST GUIDE Drafting a Prior Authorization Request The following information is presented for informational purposes only and is not intended to provide reimbursement or legal advice.
PRIOR AUTHORIZATION REQUEST GUIDE Drafting a Prior Authorization Request The following information is presented for informational purposes only and is not intended to provide reimbursement or legal advice.
Inflectra Frequently Asked Questions
 Inflectra Frequently Asked Questions 1. What is the funding status of Inflectra (infliximab)? Earlier in 2016, Inflectra (infliximab) was added to the Ontario Drug Benefit (ODB) Formulary as a Limited
Inflectra Frequently Asked Questions 1. What is the funding status of Inflectra (infliximab)? Earlier in 2016, Inflectra (infliximab) was added to the Ontario Drug Benefit (ODB) Formulary as a Limited
Slide 1 Medications in inflammatory bowel disease a primer for health care providers. Slide 2. Slide 3 Theory of pathogenesis. IBD - epidemiology
 Slide 1 Medications in inflammatory bowel disease a primer for health care providers Athos Bousvaros, MD Associate director Inflammatory Bowel Disease Center Boston Children s Hospital 617 355 2962 Slide
Slide 1 Medications in inflammatory bowel disease a primer for health care providers Athos Bousvaros, MD Associate director Inflammatory Bowel Disease Center Boston Children s Hospital 617 355 2962 Slide
Subject: Remicade (Page 1 of 5)
 Subject: Remicade (Page 1 of 5) Objective: I. To ensure that Health Share/Tuality Health Alliance (THA) has a process by which the appropriate utilization of Remicade (Infliximab) for members whose diagnosis
Subject: Remicade (Page 1 of 5) Objective: I. To ensure that Health Share/Tuality Health Alliance (THA) has a process by which the appropriate utilization of Remicade (Infliximab) for members whose diagnosis
Novel anti-inflammatory compounds for ulcerative colitis and rheumatoid arthritis therapy
 Novel anti-inflammatory compounds for ulcerative colitis and rheumatoid arthritis therapy JANSA, P. 1, HOLÝ, A. 1, ZÍDEK, Z. 2, JANEBA, Z. 1, KOLMAN, V. 1 1 Institute of Organic Chemistry and Biochemistry
Novel anti-inflammatory compounds for ulcerative colitis and rheumatoid arthritis therapy JANSA, P. 1, HOLÝ, A. 1, ZÍDEK, Z. 2, JANEBA, Z. 1, KOLMAN, V. 1 1 Institute of Organic Chemistry and Biochemistry
Humira. (adalimumab) Drug Update Slideshow NEW INDICATION
 Humira (adalimumab) NEW INDICATION Drug Update Slideshow Introduction Brand name: Humira Generic name: Adalimumab Pharmacological class: Tumor necrosis factor (TNF) blocker Strength and Formulation: 10mg/0.2mL,
Humira (adalimumab) NEW INDICATION Drug Update Slideshow Introduction Brand name: Humira Generic name: Adalimumab Pharmacological class: Tumor necrosis factor (TNF) blocker Strength and Formulation: 10mg/0.2mL,
Surgical Management of IBD in the Age of Biologics
 Surgical Management of IBD in the Age of Biologics Lisa S. Poritz, M.D Associate Professor of Surgery Division of Colon and Rectal Surgery Objectives Discuss surgical management of IBD When to operate
Surgical Management of IBD in the Age of Biologics Lisa S. Poritz, M.D Associate Professor of Surgery Division of Colon and Rectal Surgery Objectives Discuss surgical management of IBD When to operate
Pediatric Inflammatory Bowel Diseases
 Pediatric Inflammatory Bowel Diseases Michael Stephens, M.D. Denny Sanford Pediatric Symposium November 21 2014, Sioux Falls, SD 2014 slide-1 Disclosures I have nothing to disclose 2014 slide-2 Objectives
Pediatric Inflammatory Bowel Diseases Michael Stephens, M.D. Denny Sanford Pediatric Symposium November 21 2014, Sioux Falls, SD 2014 slide-1 Disclosures I have nothing to disclose 2014 slide-2 Objectives
Opinion 24 July 2013
 The legally binding text is the original French version TRANSPARENCY COMMITTEE Opinion 24 July 2013 HUMIRA 40 mg, solution for injection in pre-filled syringe B/2 x 0.8 ml pre-filled glass syringes with
The legally binding text is the original French version TRANSPARENCY COMMITTEE Opinion 24 July 2013 HUMIRA 40 mg, solution for injection in pre-filled syringe B/2 x 0.8 ml pre-filled glass syringes with
Cyltezo (adalimumab-adbm) CG-DRUG-64, CG-DRUG-65
 Market DC Cyltezo (adalimumab-adbm) CG-DRUG-64, CG-DRUG-65 Override(s) Prior Authorization Quantity Limit Medications Cyltezo (adalimumab-adbm) 40 mg/0.8 ml prefilled syringe #* ^ Approval Duration 1 year
Market DC Cyltezo (adalimumab-adbm) CG-DRUG-64, CG-DRUG-65 Override(s) Prior Authorization Quantity Limit Medications Cyltezo (adalimumab-adbm) 40 mg/0.8 ml prefilled syringe #* ^ Approval Duration 1 year
Efficacy and Safety of Treatment for Pediatric IBD
 Efficacy and Safety of Treatment for Pediatric IBD Andrew B. Grossman MD Co-Director, Center for Pediatric Inflammatory Bowel Disease Associate Professor of Clinical Pediatrics Division of Gastroenterology,
Efficacy and Safety of Treatment for Pediatric IBD Andrew B. Grossman MD Co-Director, Center for Pediatric Inflammatory Bowel Disease Associate Professor of Clinical Pediatrics Division of Gastroenterology,
Humira (adalimumab) DRUG.00002
 Humira (adalimumab) DRUG.00002 Override(s) Prior Authorization Quantity Limit Approval Duration 1 year Medications Humira 10 mg/0.2 ml syringe Humira pediatric Crohn s Disease starter pack 40 mg/0.8 ml
Humira (adalimumab) DRUG.00002 Override(s) Prior Authorization Quantity Limit Approval Duration 1 year Medications Humira 10 mg/0.2 ml syringe Humira pediatric Crohn s Disease starter pack 40 mg/0.8 ml
1.0 Abstract. Title. HUMIRA 40 mg syringe 0.8 ml for Subcutaneous Injection. Special investigation (All-case survey) in patients with Crohn's disease
 1.0 Abstract Title HUMIRA 40 mg syringe 0.8 ml for Subcutaneous Injection Special investigation (All-case survey) in patients with Crohn's disease Keywords HUMIRA, adalimumab, All-case, PMOS, Crohn's Disease,
1.0 Abstract Title HUMIRA 40 mg syringe 0.8 ml for Subcutaneous Injection Special investigation (All-case survey) in patients with Crohn's disease Keywords HUMIRA, adalimumab, All-case, PMOS, Crohn's Disease,
Regulatory Status FDA-approved indication: Humira and Amjevita are tumor necrosis factor (TNF) blockers indicated for the treatment of: (2-3)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.29 Subject: Humira Page: 1 of 10 Last Review Date: June 22, 2017 Humira Description Humira (adalimumab),
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.29 Subject: Humira Page: 1 of 10 Last Review Date: June 22, 2017 Humira Description Humira (adalimumab),
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: golimumab_simponi 8/2013 2/2018 2/2019 3/2018 Description of Procedure or Service Golimumab (Simponi and
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: golimumab_simponi 8/2013 2/2018 2/2019 3/2018 Description of Procedure or Service Golimumab (Simponi and
Temporary Compliance Waiver Notice
 Temporary Compliance Waiver Notice At the time of initial posting on August 24th, 2012, the attached PDF document may not be fully accessible to readers using assistive technology. A fully accessible version
Temporary Compliance Waiver Notice At the time of initial posting on August 24th, 2012, the attached PDF document may not be fully accessible to readers using assistive technology. A fully accessible version
ADALIMUMAB Generic Brand HICL GCN Exception/Other ADALIMUMAB HUMIRA GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW)
 Generic Brand HICL GCN Exception/Other ADALIMUMAB HUMIRA 24800 GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Does the patient have a diagnosis of moderate to severe rheumatoid
Generic Brand HICL GCN Exception/Other ADALIMUMAB HUMIRA 24800 GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Does the patient have a diagnosis of moderate to severe rheumatoid
Small Bowel and Colon Surgery
 Small Bowel and Colon Surgery Why Do I Need a Small Bowel Resection? A variety of conditions can damage your small bowel. In severe cases, your doctor may recommend removing part of your small bowel. Conditions
Small Bowel and Colon Surgery Why Do I Need a Small Bowel Resection? A variety of conditions can damage your small bowel. In severe cases, your doctor may recommend removing part of your small bowel. Conditions
ExtraintestinalManifestations of IBD
 ExtraintestinalManifestations of IBD Hyun Kim, M.D. San Diego Digestive Disease Consultants Associate Professor, UCSD School of Medicine Why Other Organs Involved in IBD? Organ Involvement Bones, Joints
ExtraintestinalManifestations of IBD Hyun Kim, M.D. San Diego Digestive Disease Consultants Associate Professor, UCSD School of Medicine Why Other Organs Involved in IBD? Organ Involvement Bones, Joints
2. Does the patient have a diagnosis of ulcerative colitis or Crohn s? Y N
 Pharmacy Prior Authorization AETA BETTER HEALTH LOUISIAA (MEDICAID) Remicade (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign
Pharmacy Prior Authorization AETA BETTER HEALTH LOUISIAA (MEDICAID) Remicade (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign
Proposed Retirement for HEDIS : Disease-Modifying Anti-Rheumatic Drug Therapy for Rheumatoid Arthritis (ART)
 Proposed Retirement for HEDIS 1 2020 2 : Disease-Modifying Anti-Rheumatic Drug Therapy for Rheumatoid Arthritis (ART) NCQA seeks public comment on the proposed retirement of the Disease-Modifying Anti-Rheumatic
Proposed Retirement for HEDIS 1 2020 2 : Disease-Modifying Anti-Rheumatic Drug Therapy for Rheumatoid Arthritis (ART) NCQA seeks public comment on the proposed retirement of the Disease-Modifying Anti-Rheumatic
3. Has the patient shown improvement in signs and symptoms of the disease? Y N
 Pharmacy Prior Authorization MERC CARE (MEDICAID) Renflexis (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign and date. Fax
Pharmacy Prior Authorization MERC CARE (MEDICAID) Renflexis (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign and date. Fax
Crohn's Disease. The What, When, and Why of Treatment
 Crohn's Disease The What, When, and Why of Treatment Brian Feagan, MD, FACG Professor of Medicine and Epidemiology and Biostatistics Director, Robarts Clinical Trials Robarts Research Institute University
Crohn's Disease The What, When, and Why of Treatment Brian Feagan, MD, FACG Professor of Medicine and Epidemiology and Biostatistics Director, Robarts Clinical Trials Robarts Research Institute University
Colostomy & Ileostomy
 Colostomy & Ileostomy Indications, problems and preference By Waleed Omar Professor of Colorectal surgery, Mansoura University. Disclosure I have no disclosures. Presentation outline Stoma: Definition
Colostomy & Ileostomy Indications, problems and preference By Waleed Omar Professor of Colorectal surgery, Mansoura University. Disclosure I have no disclosures. Presentation outline Stoma: Definition
