BRONCHOALVEOLAR LAVAGE FOR THE EVALUATION OF INTERSTITIAL LUNG DISEASE: IS IT CLINICALLY USEFUL?
|
|
|
- Sibyl Welch
- 5 years ago
- Views:
Transcription
1 ERJ Express. Published on May 3, 2011 as doi: / BRONCHOALVEOLAR LAVAGE FOR THE EVALUATION OF INTERSTITIAL LUNG DISEASE: IS IT CLINICALLY USEFUL? Keith C. Meyer, MD, MS, FACP, FCCP Professor of Medicine Medical Director of Lung Transplantation Director, Interstitial Lung Disease Clinic Co-Director, Adult Cystic Fibrosis Program Division of Allergy, Pulmonary and Critical Care Medicine Department of Medicine University of Wisconsin School of Medicine and Public Health K4/910 CSC 600 Highland Avenue Madison, WI, , USA Tel: (office); (secretary) FAX: Ganesh Raghu, MD, FCCP, FACP Professor of Medicine & Lab Medicine (Adjunct) Division of Pulmonary & Critical Care Medicine Campus Box Director, Interstitial Lung Disease, Sarcoid and Pulmonary Fibrosis Program Medical Director Lung Transplant Program, UW Seattle, WA Telephone: (206) : UWMC Operator: (206) FAX: (206) / (206) Digital Pager: (206) Key Words: interstitial lung disease, bronchoalveolar lavage Word Count: 5115 Copyright 2011 by the European Respiratory Society.
2 Introduction Bronchoalveolar lavage (BAL) has gained widespread acceptance as a procedure that can be performed safely to retrieve respiratory secretions for the examination of cellular and acellular components for both diagnostic and research purposes (1-6). When BAL was initially developed as a tool to sample respiratory secretions in animal models of lung disease and subsequently adapted as a clinical tool to study interstitial lung disease (ILD), it was perceived as holding considerable promise for the diagnosis and management of various forms of ILD such as sarcoidosis, idiopathic pulmonary fibrosis (IPF), and hypersensitivity pneumonitis (HP). Indeed, hundreds of manuscripts have been published in the medical literature over the ensuing decades as various centers around the world began to use BAL to identify agents of respiratory infections as well as examine correlates of changes in the composition of the airspace milieu associated with the presence of non-infectious parenchymal lung diseases. BAL is now routinely used as a diagnostic tool to diagnose respiratory infections, evaluate patients with acute respiratory failure or evidence of diffuse parenchymal lung diseases, or monitor the status of transplanted lung allografts (7). Despite the widespread use of BAL by pulmonologists, BAL cellular analysis, especially nucleated immune BAL cell differential counts, may be underutilized in ILD diagnosis. Information derived from BAL appearance and differential cell counts can provide useful diagnostic clues if recognized and interpreted appropriately with an adequate awareness of the potential diagnoses that are associated with specific BAL cellular patterns. In the early years following its introduction into clinical practice, bronchoscopy with BAL was perceived to hold great potential for diagnosis and management of ILD. It eventually became clear, however, that although BAL nucleated immune cell patterns often had characteristics that were highly consistent with various forms of ILD such as sarcoidosis, BAL cell counts and differentials, lymphocyte subsets, or soluble components could not be relied upon to make a confident diagnosis for many specific forms of ILD if used as a stand-alone diagnostic test. As clinical pulmonary medicine evolved during the 1990s, high-resolution computed tomography (HRCT) of the chest came into widespread clinical use. As radiologists and clinicians became increasingly familiar with imaging patterns on HRCT that were consistent with specific forms of ILD, the likelihood of making a confident diagnosis of specific ILD such as IPF or sarcoidosis is high when characteristic patterns are present and the technical quality of the HRCT is good (8,9). Despite the increasing acceptance of obtaining a HRCT during the initial stages of evaluation of patients with ILD, many patients with new onset ILD may not have the characteristic patterns on HRCT that allow a diagnosis to be made with high level of confidence by HRCT imaging alone. However, when clinical information and HRCT findings are combined with BAL fluid analysis and/or transbronchial lung biopsy, confident diagnoses may emerge that obviate the need for surgical lung
3 biopsy. Although the BAL cell pattern can provide useful information that facilitates the establishment of a specific ILD diagnosis, this is only possible if the bronchoscopist uses appropriate technique to obtain the fluid and if the differential cell count is performed according to good clinical laboratory practice by personnel with adequate experience in BAL cytologic analysis and interpreted by an expert familiar with the diverse spectrum of specific forms of ILD. This article provides a perspective on the current and future use of BAL analysis in achieving a confident diagnosis of specific forms of ILD and the potential of BAL to assist in the management of various forms of ILD. Reaching a consensus on BAL technique and interpretive methods can bridge potential gaps that exist among pulmonologists worldwide in the application of BAL technique and analytic results to clinical practice. Technical aspects of performing BAL BAL technique is of paramount importance in obtaining a useful specimen (10,11). To retrieve a specimen that is likely to reflect the types of inflammatory cells that are present in distal airspaces, a sufficient amount of isotonic saline should be instilled to adequately sample bronchoalveolar secretions from distal airspaces. The bronchoscopist should avoid contamination of the sample with proximal large airway secretions by maintaining the distal end of the bronchoscope in a wedged position in a segmental or subsegmental bronchus throughout the period of time required for the instillation and retrieval of saline aliquots. Additionally, dwell time should be minimized (aliquots should be aspirated immediately once the entire aliquot volume has been instilled). Many different BAL protocols have been published and consist of multiple aliquots that usually range from five or six aliquots of 20 ml each to four aliquots of 60 ml each. Some investigators consider the first aliquot to represent predominantly airway cells and secretions (12) and have recommended that this aliquot be kept separate and used for microbiological analysis, while subsequent sequential aliquots, usually 3 to 4 in number, should be pooled and used for cellular analysis because these are more representative of distal airspaces (alveolar sampling). Others combine all aliquots and do not differentiate between bronchial and alveolar sampling. There are no data that support one approach over the other, and many centers pool all retrieved BAL aliquots prior to submitting BAL for laboratory analysis. Additionally, there is no consensus on the optimal magnitude of negative pressure to retrieve BAL fluid once an aliquot has been instilled. The right middle lobe (RML) and lingula of the left upper lobe are the areas of the lung that are more easily accessible and likely to allow good return of BAL fluid from patients when lavaged while in a supine position (11). These areas have traditionally been used for lavage based on the perception that ILD is a diffuse parenchymal lung disease, as demonstrated on routine chest radiographs. Because
4 patients with ILD are now routinely evaluated with HRCT images of the chest, HRCT images may be used to target areas of the lung that may yield cellular findings more reflective of the disease process, such as areas of ground glass attenuation, more prominent nodular profusion, or fine reticulation. Since HRCT images is sensitive in detecting patterns of otherwise occult segmental and/or patchy alveolar opacifications or ground glass attenuation in patients with ILD, the HRCT images should be used to guide to determine the site of BAL to include sampling from involved segments as indicated by the HRCT scan. However, no studies have been performed that demonstrate that the use of HRCT to target regions that show more prominent parenchymal change in certain geographic lung regions as compared to traditionally utilized sites such as the RML or lingula improve the diagnostic yield of BAL. Ideally, the percentage of BAL fluid that is retrieved would be in the range of 30% or greater for a patient with ILD. However, if ILD is superimposed on emphysema, airways may readily collapse with negative pressure and compromise fluid retrieval. Post-bronchoscopy fever (fever, chills, and extreme malaise) may occur within hours of the BAL procedure as a consequence of proinflammatory mediator release, and these symptoms are more likely to occur if larger volumes of fluid are used for BAL (13). Processing of BAL fluid for cellular analyses An accurate and statistically meaningful enumeration of the BAL nucleated immune cell profiles requires examining at least 300 of these BAL cells randomly on a single slide with accuracy further enhanced by analyzing 400 to 500 cells (especially if more than one slide is analyzed with the differential counts from each slide averaged). The relative numbers of bronchial and squamous epithelial cells should also be noted. Although many investigators have published BAL cell profiles for cohorts of healthy volunteers, laboratories that perform BAL analysis would ideally study a sizable group of normal volunteers and establish normal values in their own laboratories that correspond to their subject population and analytical methods. BAL slide preparations can be analyzed in clinical hematopathology laboratories, but it is of key importance that personnel who analyze the BAL slide preparations and determine differential counts should be adequately trained in the proper identification of BAL cells and in recognizing various atypia that may be present on such preparations (11). However, pulmonologists familiar with BAL cell patterns and ILD should interpret the BAL cell patterns and other findings.
5 BAL fluid obtained from healthy, never-smoking individuals should contain, on average, a majority of alveolar macrophages (80-90%), some lymphocytes (5-15%), and very few neutrophils ( 3%) or eosinophils (<1%). While it is preferential to have laboratories establish normal values (including T lymphocyte subsets), normal values reported by others may be used, especially if the BAL procedure that is utilized conforms to published techniques and values obtained for normal subjects. The presence of squamous epithelial cells suggests that the BAL fluid has been contaminated by oropharyngeal secretions, which may be a consequence of poor technique in performing the BAL or because the subject aspirated upper airway secretions. BAL cell differentials and total cell counts for ex-smokers should be similar to that of never-smokers. Smokers, however, generally have a significantly increased total BAL cell count as well as total macrophages and neutrophils per μl BAL fluid, but the BAL differential cell count for smokers appears to vary little from that for never-smokers or ex-smokers except for a somewhat lower percentage of lymphocytes (5,14). Elderly subjects have been reported to have increases in lymphocytes and neutrophils in the differential cell counts, suggesting that advanced age may affect BAL cell differentials (15,16). Additionally, total volume of retrieved fluid declines with advanced age due to loss of elastic recoil and increased compliance of the aged lung. Some investigators have shown that total volumes of instilled saline that range from 100 ml to 250 ml appear to give similar cell differentials in individual patients with ILD (17,18). Infection can cause the subacute onset of diffuse lung infiltrates or coexist with noninfectious ILD. Therefore, BAL should be examined and screened for mycobacterial or fungal infection if clinical suspicion for an infectious etiology warrants such testing. If bacterial infection is a possibility, an uncentrifuged BAL fluid specimen can be sent for quantitative bacterial culture. Uncentrifuged BAL fluid can also be sent for viral culture, although a centrifuged specimen may enhance virus detection via stains or viral nucleic acid probes. Centrifuged BAL specimens can also be used to culture Legionella, Mycoplasma, and Chlamydiae in addition to mycobacteria and fungi, and cytocentrifuged specimens can be stained to detect the presence of intracellular bacteria, Pneumocystis jiroveci, mycobacteria, or fungi (7). BAL characteristics and the diagnosis of specific types of ILD When performed with a standardized technique, expertly examined, and combined with clinical and imaging data, BAL differential cell counts and other characteristics can provide important information that contributes significantly to the diagnosis of specific ILD (Table 1) (1-3,10,19-22). Additionally, cytopathologic examination may be useful to detect malignancies (e.g. lymphangitic
6 carcinoma, lymphomas) that can present as diffuse lung disease with similar appearance to non-malignant ILD on thoracic imaging (23). Other observations including the gross appearance of BAL fluid and identification of unusual cells (e.g. plasma cells, mast cells, foamy alveolar macrophages) or foreign material can yield important information that may lead to a specific diagnosis. In the appropriate clinical setting, certain gross and cellular findings in BAL are highly suggestive or even virtually diagnostic of specific ILD entities (Table 2), but these observations must be interpreted in the context of the patient s clinical presentation and radiologic findings. Additionally, clinicians should recognize that radiographic imaging, including HRCT, may not appear particularly abnormal when certain forms of ILD (e.g. non-ipf interstitial pneumonias or HP) are present. If BAL is performed on a symptomatic patient who has radiographic imaging that is not particularly suspicious for the presence of ILD, an abnormal cell profile consistent with the presence of an alveolitis can indicate the need for additional investigation including lung tissue biopsy. When BAL fluid is obtained and analyzed from patients with ILD, differential cell counts obtained from slides prepared via cytocentrifugation usually show variations in the BAL cell differential that differ from patterns found in normal subjects. These patterns tend to reflect inflammatory cell profiles in affected lung tissues (1), but cell patterns may be obtained that are not typical for a specific ILD diagnosis, that show little or no change from normal profiles, or that show mixed patterns with significant changes in the relative percentage of more than one type of nucleated immune cell (2). Additionally, altered BAL cell profiles can be observed with various airway disorders (e.g. bronchiolitis, bronchitis, asthma, eosinophilic bronchitis, bronchiectasis, and allergic bronchopulmonary aspergillosis). Nonetheless, certain cell patterns with significant changes in one or more BAL cell type can be highly consistent with or strongly suggest the presence of specific types of ILD. A recently published study of the predictive value of BAL for ILD diagnosis for a large cohort of patients (N=3,118) suggested that BAL cell counts are most useful for relatively common ILD diagnoses such as sarcoidosis, in contrast to relatively rare forms of ILD where BAL differential cell counts tend to have less diagnostic utility (24). A BAL lymphocyte differential count that exceeds 25% is quite likely to be caused by ILD associated with granuloma formation (e.g. sarcoidosis and hypersensitivity pneumonitis) or drug toxicity, if other possibilities, such as mycobacterial or fungal infection, are excluded. However, prominent BAL lymphocytosis can also be observed with other entities such as cryptogenic organizing pneumonia (COP), cellular non-specific interstitial pneumonia (NSIP), lymphoid interstitial pneumonia (LIP), or non-lip lymphoproliferative disorders. Extreme increases in BAL neutrophils are likely caused by infection or relatively acute and diffuse lung injury. Eosinophil differential cell counts 25% are highly likely to be
7 caused by eosinophilic lung disease, especially eosinophilic pneumonia (EP) if the presentation is acute (25,26). Increased numbers of mast cells have been associated with HP, drug reactions, sarcoidosis, ILD associated with collagen vascular disease (CVD), IPF, COP, eosinophilic pneumonia, or malignancy. Plasma cells have been observed in BAL in HP, drug reactions, EP, malignancy, or infection (27). Alveolar macrophages (AM) may also display certain morphologic changes such as a foamy appearance in HP, markedly vacuolated cytoplasm with positive staining of vacuoles for fat in chronic aspiration pneumonitis, cytoplasmic inclusions associated with viral infection (e.g. cytomegalovirus pneumonia), ingested RBCs and RBC fragments plus hemosiderin with diffuse alveolar hemorrhage, ingested asbestos bodies, or other dust particles. Grossly bloody lavage fluid, which may range from pink to red with acute hemorrhage or to orange-brown if subacute, is characteristic of diffuse alveolar hemorrhage (DAH), especially if sequential retrieved BAL fluid aliquots do not show any decrease in the amount of bloody discoloration of the lavage fluid or (especially) if the discoloration increases (28). If DAH is present, RBCs should be identifiable on the cytospin, and AM will stain positively for hemosiderin when stained with an iron stain if the onset of hemorrhage has preceded the time of BAL by 24 to 48 hrs. Freshly retrieved BAL fluid that has a milky or light brown to whitish, cloudy appearance with flocculent debris that settles out of the fluid to the bottom of its container prior to centrifugation suggests PAP as the likely diagnosis (29). The diagnosis can be confirmed if the sediment, which is surfactant-derived lipoproteinaceous material, stains positive with periodic acid-schiff (PAS) staining. Flow cytometric analyses of BAL cells have been reported by numerous investigators (2,24,27), and alterations in BAL lymphocyte subsets have been examined extensively, especially for sarcoidosis (1,2,30-32). The presence of a high CD4+/CD8+ T lymphocyte ratio increases the likelihood of diagnosis of sarcoidosis when combined with BAL lymphocytosis, although the clinician must consider age as a factor in the elevated CD4+/CD8+ ratio if the patient is elderly (16). Nonetheless, it is debatable whether BAL lymphocyte subset data provides a clinical tool that contributes significantly to ILD diagnosis. The BAL CD4+ T lymphocyte subset and CD4+/CD8+ T lymphocyte ratio tend to be elevated in clinically active pulmonary sarcoidosis (31). CD4+/CD8+ lymphocyte ratios in BAL from clinically healthy, younger adults usually range from 1.0 to 3.5 with average value between 1.5 and 2.0. An elevated CD4+/CD8+ ratio of 3.5 is relatively specific for sarcoidosis (32,33). However, the sensitivity of an increased CD4+/CD8+ ratio is relatively low for sarcoidosis (32,33), and many patients do not have an elevated ratio or may even have a low ratio (34). A depressed CD4+/CD8+ ratio has been observed in HP, drug-induced lung disease, COP, EP, and IPF (27), but this ratio has not been shown to contribute
8 meaningfully to the diagnosis of non-sarcoid ILD. Use of the CD4+/CD8+ ratio as a routine component of BAL analysis is questionable and may increase the cost of the procedure considerably. Flow cytometry may be useful to support a diagnosis of pulmonary Langerhans cell histiocytosis (PLCH) if clinical data and imaging suggest this entity. PLCH is associated with increased (>4-5% of total cells) CD1a-positive cells in BAL, although this may no longer be the case in later stages of disease (35,36). However, macrophage autofluorescence may present insurmountable difficulties in obtaining reliable data, and immunohistochemistry on prepared slides is more likely to provide reliable enumeration of the percentage of positively stained Langerhans cells (36). Flow cytometry and/or immunocytochemistry can be useful to identify monoclonal lymphocyte populations to support the diagnosis of a lymphoid malignancy and should be obtained if clinical data suggest the possibility of pulmonary lymphoma and the clinical laboratory can perform appropriate analyses. Although flow cytometric analysis of BAL cells may provide useful information in certain situations, the routine use of flow cytometry to analyze BAL cell subpopulations to assist in the diagnosis of specific forms of ILD is of unclear benefit. However, flow cytometry of BAL cells can be obtained if it is likely to provide important diagnostic information. One application of BAL that evolved over the past decade was the suggested incorporation of the BAL cellular findings into the major and minor criteria for the clinical diagnosis of IPF when surgical lung biopsy is not performed (37). However, since the positive predictive value of the HRCT findings of usual interstitial pneumonia (UIP)pattern for a diagnosis of IPF/UIP is at least 85%, and a confident diagnosis of UIP by experienced radiologists can be made in 95% of cases with characteristic pattern of UIP on HRCT images (38-41), invasive procedures including bronchoscopy, BAL and transbronchial lung biopsy may not be required to make a confident diagnosis of IPF in the appropriate clinical setting (9). In fact, the features of the criteria for the UIP pattern has been precisely described in the new joint ATS-ERS-JRS-ALAT statement evidence -based guideline for the diagnosis and management of IPF ; the new guideline states that the presence of the described UIP pattern on HRCT imaging is sufficient to make the diagnosis of IPF in the appropriate clinical setting and eliminates the need for BAL cellular analyses (42,43). However, an increased lymphocyte BAL differential cell count in BAL from patients whose HRCT demonstrates a UIP pattern may raise the possibility of chronic HP or other diagnoses associated with BAL lymphocytosis. In a recent study, 8 % of patients with a seemingly confident clinical and HRCT diagnosis of IPF had >30 % lymphocytes in BAL, an atypical finding for the diagnosis of UIP/IPF that suggested HP or another disorder as the actual diagnosis (44). The new evidence -based guideline for diagnosis and management of IPF remarks on this point and defers the decision for the need of BAL cellular analyses to individual experts with established BAL laboratories (that provide reliable cell differential counts ) who may opt to use BAL cellular analyses in patients undergoing evaluation for the diagnosis of IPF at their centers to ensure the lack of findings that suggest a non-ipf form of ILD as the diagnosis. (42).With the increasing utilization of HRCT to evaluate patients during early stages of ILD, it is likely for patients with ILD not to manifest the precise pattern of l UIP on HRCT.In this setting many clinicians may routinely perform BAL for cellular analyses as a part of the initial evaluation of patients presenting with early and new onset ILD.Thus, the clinical utility for BAL cellular analyses as an useful intervention in narrowing the differential diagnoses of ILD in this setting is evident. It must
9 however be recognized that for an accurate diagnosis, biopsy of the tissue with adequate sampling may still be required. The performance of transbronchial biopsy in the peripheral lesions of UIP, NSIP, DIP is not advised. Bronchoalveolar lavage can also assist in the diagnosis of acute onset ILD (defined as a combination of illness 4 weeks duration, shortness of breath, hypoxemia, and diffuse radiographic infiltrates in a patient with no history of prior lung disease and no obvious risk factors for ARDS such as sepsis or trauma). Diagnostic considerations in acute onset ILD include infection, non-infectious ILD (acute interstitial pneumonia, acute EP, DAH, acute HP, acute COP, drug toxicity, or acute exacerbation of previously undiagnosed IPF). Examination of BAL fluid can detect infection or hemorrhage, and the BAL cell profile may show large numbers of eosinophils, which is strongly supportive of a diagnosis of acute EP. Additionally, large numbers of lymphocytes would suggest entities such as acute HP or drug toxicity, especially if accompanied by plasma cells and an appropriate exposure history. Bronchoscopy with BAL at the time of acute presentation may facilitate diagnosis and minimize procedural risk if a diagnosis can be made and obviate the need to progress to a surgical lung biopsy. BAL and the Management of ILD A role for BAL cellular analyses in the treatment of ILD (e.g. to initiate, direct and/or monitor pharmacologic therapies) has not been established. One application of BAL to disease management, however, is its use to evaluate acute changes in symptoms and lung function. Several adverse events may occur in the course of interstitial diseases including respiratory infection, drug reactions, hemorrhage, or an acute exacerbation of the disease process, and BAL may help to identify the cause of clinical deterioration and facilitate timely intervention. Many investigators have examined the role of BAL cellular profiles for monitoring disease activity and response to therapies in specific ILD, and BAL cellular findings at the time of diagnosis have been reported by some investigators to reflect disease severity and predict the likelihood of disease progression. The degree of increase in BAL neutrophils has been correlated with disease severity and prognosis for both HP (45,46) and IPF (37,47-49), and BAL eosinophilia has been linked to more severe disease and worse prognosis in IPF (51,51). Similarly, the presence of increased neutrophils in BAL from patients with sarcoidosis has been associated with more progressive disease that is less likely to respond
10 to immunosuppressive therapy (52). BAL lymphocytosis has been linked to prognosis and response to therapy for some forms of IIP. BAL findings from patients diagnosed with IPF when this diagnostic term did not necessarily exclude other IIP such as NSIP suggested that higher BAL lymphocyte differential cell counts correlated with better prognosis and/or response to therapy (37,48-50,53). A more recent study, although retrospective, demonstrated that BAL lymphocytosis in patients who appear to have fibrotic IIP suggests that the diagnosis is not IPF but rather NSIP, and BAL lymphocytosis was found to correlate with a better prognosis (54). Lung lavage was initially used as a therapy for septic lung disease and PAP (55,56). Although its therapeutic application is quite limited at this time, whole lung lavage (WLL) is considered standard of care for PAP (57). However, standardization of technique and controlled clinical trials to determine the efficacy and durability of WLL as a therapy for PAP are lacking (57). Future directions and research questions BAL holds the potential to play an increasingly useful role in both the diagnosis and monitoring of patients with ILD, but many important issues have not been adequately resolved. These unresolved questions include: What is the best technique for performing BAL? Aliquot volume and number of aliquots Type and magnitude of negative pressure to retrieve instilled fluid Pooling of aliquots for subsequent analysis When and how should HRCT be used as a guide to determine the site of BAL in patients demonstrating diffusely distributed lesions in routine chest radiographs? Should microbiologic testing be performed whenever BAL is performed for cytologic analysis to determine BAL cell total and differential cell count? Should cytologic analysis to detect malignant cells be performed whenever BAL is performed to evaluate diffuse lung disease? Who should interpret BAL cellular preparations? Can BAL combined with transbronchial lung biopsy synergistically increase diagnostic power and diminish need for surgical lung biopsy?
11 BAL may prove particularly helpful as treatments become available that may alter the profiles of BAL cells and/or non-cellular components of lower respiratory tract bronchoalveolar surface liquid if patients have a favorable response to such therapies, and detection of these changes may support continued, ongoing treatment with specific agents. Additionally, multi-center studies that prospectively compare BAL with HRCT and histological patterns in large groups of patients will be facilitated by improvements in BAL standardization (11). Such studies have the potential to better define the underlying inflammatory and histological events associated with different HRCT appearances, which may reduce the need for biopsies plus provide information that can guide effective therapies. New tools that utilize genomic and proteomic characterization of BAL cells and soluble components may eventually prove extremely useful in making an accurate ILD diagnosis, choosing and implementing potentially effective therapies, monitoring disease activity, and assessing the effect of therapeutic pharmacologic interventions. The recent development of DNA microarray and other technologies that identify and monitor expression patterns of vast numbers of genes promise to provide important information about the pathobiology of various types of ILD (58,59). Similarly, recent advances in proteomics that allow mapping and identification of multiple protein expression patterns by 2D gel electophoresis, image analysis, protein spot transfer and mass spectrometry now raise the real hope that the products of genes linked to disease pathogenesis can be identified and quantitated (60). Determining gene and protein expression patterns over time as reflected in BAL can potentially identify the key molecules involved in the initiation and progression of the different ILDs, provide an accurate clinical diagnosis of specific ILD without resorting to lung biopsy, and indicate targets for new and effective therapies. A global analysis approach has identified many genes that display increased expression in IPF (58). Additionally, certain gene products that may play an important role in IPF pathogenesis including pigment epithelium-derived factor (61), matrilysin (62), and osteopontin (63) have been identified via these methods and may serve as disease biomarkers. Thonhofer et al (64) demonstrated that stimulated BAL cells from patients with sarcoidosis displayed up- or down-regulation of over a thousand genes, including selective up-regulation of B-MYB, a potent growth factor for lymphocytes and regulator of apoptosis, and FABP4, a regulator of lipid metabolism and arachidonic acid uptake by macrophages. Not only can microarray genetic analysis provide characteristic gene expression patterns for specific ILD, but distinct expression patterns may differentiate one ILD from another. Selman et al. (65) recently validated the concept that gene expression patterns can be characteristic of
12 specific ILD by demonstrating that distinct gene expression patterns in lung tissue differed for patients with IPF versus those with HP. A proteomics approach may also revolutionize the utility of BAL analysis for ILD diagnosis and management (60). Characterizing and quantitating proteins in biological specimens allow one to circumvent modifications of RNA that affect protein production and expression, and proteomic analytic techniques can be applied to acellular compartments such as epithelial surface liquid retrieved via BAL or peripheral blood plasma. Early investigations that utilized electrophoretic techniques to examine protein profiles in BAL were able to show some differences between IPF and sarcoidosis (66,67), and later studies using two-dimensional electrophoretic techniques allowed enhanced fingerprinting of digested proteins from BAL supernatant fluids and demonstrated different profiles for IPF, sarcoidosis, and HP (68,69). More recently Rottoli et al. (70) used 2-dimensional electrophoresis to construct protein maps from BAL fluid that were able to demonstrate differences among IPF, systemic sclerosis, and sarcoidosis. The development of techniques that employ (1) mass spectrometry combined with ionization of peptides via matrix-assisted laser-desorption ionization (MALDI) or electrospray ionization (ESI) or (2) mass spectrometry combined with the use of surface chromatography to capture proteins on a chip surface (SELDI) coupled with greatly enhanced computational abilities and proteomics databases hold considerable promise for the study of ILD via this emerging technology, and these techniques have recently been applied to the study of markers in BAL fluid in patients with lung inflammation (71). Summary and Conclusions. Although investigators have recognized the importance of the BAL procedure in clinical pulmonary medicine, unresolved questions concerning the BAL procedure and the clinical utility of BAL cellular analyses for the evaluation and management of ILD in the present era need to be addressed by consensus reached among international experts. The technical aspects of performing BAL and analyzing cellular features of BAL fluid need standardization, and the status of the clinical utilization of BAL findings in day to day clinical practice needs to be clarified so that principles of performing, analyzing, and interpretating BAL can be applied globally and avoid wide variations in BAL technique and specimen analysis. BAL cellular patterns and other BAL findings can play an important role in the diagnosis of many forms of ILD, especially when these findings are obtained via a properly performed BAL procedure and analysis and then combined with clinical data and HRCT imaging. In many instances the combination of clinical information (history, examination,
13 and pulmonary function testing), HRCT imaging, and BAL analysis can provide enough information to allow the clinician to be assured of a confident diagnosis and avoid more invasive testing to obtain lung tissue for histopathologic diagnostic examination. BAL may be particularly beneficial for the fragile patient or patients at high risk for significant complications if subjected to surgical lung biopsy. Universal adoption of a standardized BAL procedure will undoubtedly improve the chance of identifying clinically relevant differences between specific disorders and provide results that can be compared from center to center. A standardized procedure will enable centers to pool information to achieve the large patient groups necessary to more adequately investigate the value of BAL markers for the identification of specific disease entities, determining clinical disease activity, detecting disease progression, providing a prognosis, and guiding treatment strategies. Identifying specific and sensitive markers that can address these problems remains an elusive but vitally important goal. Future application of BAL to the evaluation of ILD in concert with an evolving, improved understanding of the pathogenesis of specific ILD and the development of novel testing that utilizes genomic and proteomic characterization of BAL fluids holds the potential to provide accurate diagnostic and prognostic information for patients with ILD and potentially play a role in choosing novel therapies for specific forms of ILD. Despite this significant diagnostic potential, very few patients have been investigated so far using gene and protein mapping approaches, and applications of the powerful techniques of gene and protein mapping to the study of ILD are still at a very early stage but have the potential to evolve rapidly in the coming years. Access to large collections of well characterized clinical samples (BAL, lung biopsies, and peripheral blood) to which these techniques can be applied combined with HRCT or newer forms of thoracic imaging will greatly facilitate our understanding of basic and clinical aspects of ILD pathobiology and facilitate the identification of clinically useful biomarkers. References: 1. Costabel U, Guzman J. Bronchoalveolar lavage in interstitial lung disease. Curr Opin Pulm Med 2001;7: Costabel U, Guzman J. Bronchoalveolar lavage. In: Schwartz MI, King TE Jr, eds. Interstitial Lung Disease. Hamilton, Ont: BC Decker; 2003:
14 3. Drent M, Meyer KC, Baughman RP. General aspects: bronchoalveolar lavage. In Diffuse Parenchymal Lung Diseases Progress in Respiratory Research, Volume Editors Costabel U, du Bois R, Egan J, Series Editor Bollinger CT Klech H, Hutter C, eds. Clinical guidelines and indications for bronchoalveolar lavage (BAL): report of the European Society of Pneumology Task Group on BAL. Eur Respir J 1990;3: The BAL Co-operative Group Steering Committee. Bronchoalveolar lavage constituents in healthy individuals, idiopathic pulmonary fibrosis, and selected comparison groups. Am Rev Respir Dis 1990;141(Suppl 5):S169-S Haslam PL, Baughmann RP, eds. Guidelines for the measurement of acellular components and recommendations for standardization of bronchoalveolar lavage (BAL). Eur Respir Rev 1999;9: Meyer KC. Bronchoalveolar lavage as a diagnostic tool. Semin Respir Crit Care Med 2007;28: Kanne JP. Interstitial lung disease (ILD): imaging finding, and the role of imaging in the evaluating the patient with known or suspected ILD. Semin Roentgenol 2010;45:3. 9. Schmidt SL, Sundaram B, Flaherty KR. Diagnosing fibrotic lung disease: when is high-resolution computed tomography sufficient to make a diagnosis of idiopathic pulmonary fibrosis? Respirology 2009;14: Haslam PL, Baughman RP. Report of ERS Task Force: guidelines for measurement of acellular components and standardization of BAL. Eur Respir J 1999;14: Baughman RP. Technical aspects of bronchoalveolar lavage: recommendations for a standard procedure. Semin Respir Crit Care Med 2007;28: Martin TR, Raghu G, Maunder RJ, et al. The effects of chronic bronchitis and chronic air-flow obstruction on lung cell populations recovered by bronchoalveolar lavage. Am Rev Respir Dis 1985;132: Dhillon DP, Haslam PL, Townsend PJ, et al. Bronchoalveolar lavage in patients with interstitial lung diseases: side effects and factors affecting fluid recovery. Eur J Respir Dis 1986;68: Costabel U, Guzman J. Effect of smoking on bronchoalveolar lavage constituents. Eur Respir J 1992;5: Meyer KC. Neutrophils and low-grade inflammation in the seemingly normal aging human lung. Mech Aging Develop 1998;104: Meyer KC, Soergel P. Bronchoalveolar lymphocyte phenotypes change in the normal aging human lung. Thorax 1999;54:
15 17. Dohn MN, Baughman RP. Effect of changing instilled volume for bronchoalveolar lavage in patients with interstitial lung disease. Am Rev Respir Dis 1985;132: Helmers RA, Dayton CS. Floerchinger C, et al. Bronchoalveolar lavage in interstitial lung disease: effect of volume of fluid infused. J Appl Physiol 1989;67: Baughman RP, Drent M. Role of bronchoalveolar lavage in interstitial lung disease. Clin Chest Med 2001;22: Meyer KC. The role of bronchoalveolar lavage in interstitial lung disease. Clin Chest Med 2004;25: Baughman RP. Is bronchoalveolar lavage clinically useful for everyday practice in interstitial lung disease? Con: bronchoalveolar lavage. J Bronchology 1999;6: Raghu G. Is bronchoalveolar lavage clinically useful for everyday practice in interstitial lung disease? Con: bronchoalveolar lavage. J Bronchology 1999;6: Poletti V, Poletti G, Murer B, Saragoni L, Chilosi M. Bronchoalveolar lavage in malignancy. Semin Respir Crit Care Med 2007;28: Welker L, Jorres RA, Costabel U, Magnussen H. Predictive value of BAL cell differentials in the diagnosis of intnerstitial lung diseases. Eur Respir J 2004;24: Allen JN, Davis WB. Eosinophilic lung diseases. Am J Respir Crit Care Med 1994;150: Pope-Harman AL, Davis WB, Allen ED, et al. Acute eosinophilic pneumonia. A summary of 15 cases and review of the literature. Medicine (Baltimore) 1996;75: Drent M, Jacobs JA. Bronchoalveolar lavage. In: Baughman RP, Du Bois RM, Lynch JP III, Wells AU, eds. Diffuse Lung Disease: A Practical Approach. Eds. New York, NY: Oxford University Press; 2004: Fontenot AP, Schwarz MI. Diffuse alveolar hemorrhage. In: Schwartz MI, King TE Jr, eds. Interstitial Lung Disease. Hamilton, Ont: BC Decker; 2003: Martin RJ, Coalson JJ, Rogers RM, et al. Pulmonary alveolar proteinosis: the diagnosis by segmental lavage. Am Rev Respir Dis 1980;121: Drent M, Mansour K, Linssen C. Bronchoalveolar lavage in sarcoidosis. Semin Respir Crit Care Med 2007;28: Hunninghake GW, Crystal RG. Pulmonary sarcoidosis: a disorder mediated by excess helper T- lymphocyte activity at sites of disease activity. NEJM 1981;305: Costabel U, Zaiss AW, Guzman J. Sensitivity and specificity of BAL findings in sarcoidosis. Sarcoidosis 1992;9 Suppl 1: Winterbauer RH, Lammert J, Selland M, et al. Bronchoalveolar lavage cell populations in the diagnosis of sarcoidosis. Chest 1993;104:
16 34. Kantrow SP, Meyer KC, Kidd P, et al. The CD4/CD8 ratio in BAL fluid is highly variable in sarcoidosis. Eur Respir J 1997;10: Auerswald U, Barth J, Magnussen H. Value of CD1 positive cells in bronchoalveolar lavage fluid for the diagnosis of pulmonary histiocytosis X. Lung 1990;169: Costabel U, Guzman J, Bonella F, Oshimo S. Bronchoalveolar lavage in other interstitial lung diseases. Semin Respir Crit Care Med 2007;28: American Thoracic Society. Idiopathic pulmonary fibrosis: diagnosis and treatment. International consensus statement. American Thoracic Society (ATS) and the European Respiratory Society (ERS). Am J Respir Crit Care Med 2000;161: Hunninghake GW, Lynch DA, Galvin JR, et al. Radiologic findings are strongly associated with a pathologic diagnosis of usual interstitial pneumonia. Chest 2003;124: Lynch DA, Godwin D Jr, Safrin S, et al. High-resolution computed tomography in idiopathic pulmonary fibrosis: diagnosis and prognosis. Am J Respir Crit Care Med 2005;172: Swensen SJ, Aughenbaugh GL, Myers JL. Diffuse lung disease: diagnostic accuracy of CT in patients undergoing surgical biopsy of the lung. Radiology 1997;205: Mathieson JR, Mayo JR, Staples CA, et al. Chronic diffuse infiltrative lung disease: comparison of diagnostic accuracy of CT and chest radiography. Radiology 1989;171: Raghu G et al.idiopathic pulmonary fibrosis: evidence based guidelines for diagnosis and management a joint ATS/ERS/JRS/ALAT statement Am J Respir Crit Care Med 2011;183 : Raghu G. Idiopathic pulmonary fibrosis guidelines on clinical management has advanced from consensus based in 2000 to evidence based -based in Eur Respir J 2011; 37 : Ohshimo S, Bonella F, Cui A, Beume M, Kohno N, Guzman J, Costabel U. Significance of bronchoalveolar lavage for the diagnosis of idiopathic pulmonary fibrosis. Am J Respir Crit Care Med 2009;179: Haslam PL, Dewar A, Butchers P, et al. Mast cells, atypical lymphocytes, and neutrophils in bronchoalveolar lavage in extrinsic allergic alveoliltis. Comparison with other interstitial lung diseases. Am Rev Respir Dis 1987;135: Drent M, van Velzen Blad H, Diamant M, et al. Bronchoalveolar lavage in extrinsic allergic alveolitis: effect of time elapsed since antigen exposure. Eur Respir J 1993;6: Lynch JPD, Standiford TJ, Rolfe MW, et al. Neutrophilic alveolitis in idiopathic pulmonary fibrosis. The role of initerleukin-8. Am Rev Respir Dis 1992;145: Haslam PL, Turton CW, Lukoszek A, et al. Bronchoalveolar lavage fluid cell counts in cryptogenic fibrosing alveolitis and their relation to therapy. Thorax 1980;35:
17 49. Rudd RM, Haslam PL, Turner-Warwick M. Cryptogenic fibrosing alveolitis. Relationships of pulmonary physiology and bronchoalveolar lavage to response to treatment and prognosis. Am Rev Respir Dis 1981;124: Watters LC, Schwarz MI, Cherniack RM, et al. Idiopathic pulmonary fibrosis: pretreatment bronchoalveolar lavage cellular constituents and their relationships with lung histopathology and clinical response to therapy. Am Rev Respir Dis 1987;135: Peterson WMW, Monick M, Hunniknghake GW. Prognostic role of eosinophils in pulmonary fibrosis. Chest 1987;92: Drent M, Macobs JA, de Vries J, et al. Does the cellular bronchoalveolar lavage fluid profile reflect the severity of sarcoidosis? Eur Respir J 1999;13: Schwartz DA, Helmers RA, Galvin JR, et al. Determinants of survival in idiopathic pulmonary fibrosis. Am J Respir Crit Care Med 1994;149: Ryu YJ, Chung MP, Han J, Kim TS, Lee KS, Chun E, Kyung SY, Jeong SH, Colby TV, Kim H, Jung Kwon O. Bronchoalveolar lavage in fibrotic idiopathic interstitial pneumonias. Respir Med 2007;101: Kylstra JA, Rausch DC, Hall KD, Spock A. Volume-controlled lung lavage in the treatment of asthma. bronchiectasis, and mucoviscidosis. Am Rev Respir Dis 1971;103: Ramirez J, Schultz RB, Dutton RE. Pulmonary alveolar proteinosis: a new technique and rationale for treatment. Arch Intern Med 1963;112: Huizar I, Kavuru MS. Alveolar proteinosis syndrome: pathogenesis, diagnosis, and management. Curr Opin Pulm Med 2009;15: Agostini C, Miorin M, and Semenzato G. Gene expression profile analysis by DNA microarrays: a new approach to assess functional genomics in diseases. Sarcoidosis Vasc Diffuse Lung Dis 2002; 19: Kaminski N and Rosas IO. Gene expression profiling as a window into idiopathic pulmonary fibrosis pathogenesis: can we identify the right target genes? Proc Am Thorac Soc 2006; 3: Bowler RP, Ellison MC, Reisdorph N. Proteomics in pulmonary medicine. Chest 2006;130: Cosgrove GP, Brown KK, Schiemann WP, Serls AE, Parr JE, Geraci MW, Schwartz ME, Cool CD, Worthen GS. Pigment epithelium-derived factor in idiopathic pulmonary fibrosis: a role in aberrant angiogenesis. Am J Respir Crit Care Med 2004;170: Zuo F, Kaminski N, Eugui E, Allard J, Yakhini Z, et al. Gene expression analysis reveals matrilysin as a key regulator of pulmonary fibrosis in mice and humans. Proc Natl Acad Sci USA 2002;99:
18 63. Pardo A, Gibson K, Cisneros J, Richards TJ, Yang Y, et al. Up-regulation and profibrotic role of osteopontin in human idiopathic pulmonary fibrosis. PloS Med 2005;2:e Thonhofer R, Maercker C, Popper HH. Expression of sarcoidosis related genes in lung lavage cells. Sarcoidosis Vasc Diffuse Lung Dis 2002;19: Selman M, Pardo A, Barrera L, Estrada A, Watson SR, Wilson K, Aziz N, Kaminski N, and Zlotnik A. Gene expression profiles distinguish idiopathic pulmonary fibrosis from hypersensitivity pneumonitis. Am J Respir Crit Care Med 2006; 173: Lenz AG, Meyer B, Costabel U, Maier K. Bronchoalveolar lavage fluid proteins in human lung disease: analysis by two-dimensional electrophoresis. Electrophoresis 1993;14: Uebelhoer M, Bewig B, Oldigs M, Nowak D, Magnussen H, Petermann W, Barth J. Scand J Clin Lab Invest 1993;53: Wattiez R, Hermans C, Cruyt C, Bernard A, and Falmagne P. Human bronchoalveolar lavage fluid protein two-dimensional database: study of interstitial lung diseases. Electrophoresis 2000; 21: Magi B, Bini L, Perari MG, Fossi A, Sanchez J, Hochstrasser D, Paesano S, Raggiaschi R, Santucci A, Pallini V, Rottoli P. Bronchoalveolar lavage fluid protein composition in patients with sarcoidosis and idiopathic pulmonary fibrosis: a two-dimensional electrophoretic study. Electrophoresis 2002;23: Rottoli P, Magi B, Perari MG, Liberatori S, Nikiforakis N, Bargagly E, Cianti R, Bini L, Pallini V. Cytokine profile and proteome analysis in bronchoalveolar lavage of patients with sarcoidosis, pulmonary fibrosis associated with systemic sclerosis and idiopathic pulmonary fibrosis. Proteomics 2005;5: de Torre C, Ying S, Munson PJ, Meduri GU, Suffredini AF. Proteomic analysis of inflammatory biomarkers in bronchoalveolar lavage. Proteomics 2006;6:
19 Table 1. Bronchoalveolar lavage findings that are useful in ILD diagnosis. BAL Finding Eosinophils 25% Lymphocytes 25% Neutrophils 50% Bloody fluid High hemosiderin score Cd1a+ cells >4% Milky BAL fluid with PASpositive amorphous debris Monotypic lymphocytes Malignant cells Squamous epithelial cells >5% Bronchial epithelial cells >5% Consistent Interpretation/Suggested Diagnosis Eosinophilic pneumonia Sarcoidosis, HP, cellular NSIP, drug reaction, CBD, LIP, lymphoproliferative disorder AIP, DAD, AEIPF, pulmonary infection Pulmonary hemorrhage, DAH DAH, DAD Langerhans cell histiocytosis Pulmonary alveolar proteinosis Pulmonary lymphomatous malignancy Pulmonary malignancy Unsuitable sample due to upper airway secretion contamination BAL sample may be unsuitable for cell analysis
20 Table 2. Bronchoalveolar lavage findings: diagnostic value and disease correlations. ILD Type Suspected BAL Useful BAL Diagnostic Value of BAL* Helpful? Findings Sarcoidosis Yes Lymph CD4/CD8 T-cell ratio** Highly consistent if CXR and/or HRCT shows typical changes of BHL, nodules along bronchovascular structures* Hypersensitivity Pneumonitis Yes Lymph Plasma cells Foamy macrophages Highly consistent if significant exposure to known antigen combined with typical HRCT pattern (ground glass nodules in mid/upper lung regions) * Cellular NSIP Yes Lymph Highly consistent if mid/lower lung region Fibrotic NSIP +/- Variable Lymph, Eos, Neut diffuse GGO present* Rule out infection/malignancy; absence of inconsistent BAL findings plus typical/highly consistent HRCT imaging is supportive of diagnosis Drug Reaction Yes Lymph, Eos Highly consistent if patchy/diffuse infiltrates plus drug ingestion known to be pneumotoxic and cause interstitial change* Acute Interstitial Pneumonia Yes / Neut Exclude infection Diffuse Alveolar Damage Yes Neut Reactive/desquamated Type II pneumocytes Rule out infection/malignancy Type II pneumocyte changes are highly suggestive in appropriate clinical setting with patchy/diffuse infiltrates Lymphangitic Carcinoma Yes Malignant cells Diagnostic Lymphoid Malignancies Yes Monoclonal Lymph Diagnostic Langerhans Cell Histiocytosis Yes >4% CD1a+ cells Highly consistent/diagnostic if typical HRCT changes present Lymphangioleiomyomatosis +/- Exclude infection Rule out infection/malignancy; absence of inconsistent BAL findings plus typical/highly consistent HRCT imaging is supportive of diagnosis Alveolar Proteinosis Yes Milky fluid White/tan sediment PAS+ sediment Diagnostic if typical/consistent HRCT changes present Radiation Pneumonitis +/- Lymph Highly consistent if thoracic irradiation occurred within window* Eosinophilic Pneumonia Yes Eos Eos >25% are diagnostic if correlated with typical/highly consistent HRCT imaging (or typical/classic CXR pattern) Cryptogenic Organizing Pneumonia +/- Variable alveolitis Lymph, Eos, Neut Scleroderma +/- Variable Lymph, Eos, Neut Other CTD +/- Variable alveolitis Lymph, Eos, Neut Rule out infection/malignancy; evidence of inflammation with typical/highly consistent HRCT imaging is supportive of diagnosis Rule out infection/malignancy; evidence of inflammation with typical/highly consistent HRCT imaging plus typical clinical findings plus positive serologic testing is supportive of diagnosis Rule out infection/malignancy; evidence of inflammation with typical/highly consistent HRCT imaging plus typical clinical findings plus positive serologic testing is supportive of diagnosis Chronic Beryllium Disease Yes Lymph Rule out infection/malignancy; BAL Be-LPT plus typical/highly consistent HRCT imaging
BRONCHO ALVEOLAR LAVAGE IN INTERSTITIAL LUNG DISEASES A USEFUL TOOL OR AN OUT DATED CONCEPT?
 1 BRONCHO ALVEOLAR LAVAGE IN INTERSTITIAL LUNG DISEASES A USEFUL TOOL OR AN OUT DATED CONCEPT? Dr. M. V. Nagarjuna AIMS 1. Technical Considerations in performing BAL 2. Role of BAL in ILD 1. Diagnosis
1 BRONCHO ALVEOLAR LAVAGE IN INTERSTITIAL LUNG DISEASES A USEFUL TOOL OR AN OUT DATED CONCEPT? Dr. M. V. Nagarjuna AIMS 1. Technical Considerations in performing BAL 2. Role of BAL in ILD 1. Diagnosis
Differential diagnosis
 Differential diagnosis Idiopathic pulmonary fibrosis (IPF) is part of a large family of idiopathic interstitial pneumonias (IIP), one of four subgroups of interstitial lung disease (ILD). Differential
Differential diagnosis Idiopathic pulmonary fibrosis (IPF) is part of a large family of idiopathic interstitial pneumonias (IIP), one of four subgroups of interstitial lung disease (ILD). Differential
Outline Definition of Terms: Lexicon. Traction Bronchiectasis
 HRCT OF IDIOPATHIC INTERSTITIAL PNEUMONIAS Disclosures Genentech, Inc. Speakers Bureau Tadashi Allen, MD University of Minnesota Assistant Professor Diagnostic Radiology 10/29/2016 Outline Definition of
HRCT OF IDIOPATHIC INTERSTITIAL PNEUMONIAS Disclosures Genentech, Inc. Speakers Bureau Tadashi Allen, MD University of Minnesota Assistant Professor Diagnostic Radiology 10/29/2016 Outline Definition of
Diffuse Interstitial Lung Diseases: Is There Really Anything New?
 : Is There Really Anything New? Sujal R. Desai, MBBS, MD ESTI SPEAKER SUNDAY Society of Thoracic Radiology San Antonio, Texas March 2014 Diffuse Interstitial Lung Disease The State of Play DILDs Is There
: Is There Really Anything New? Sujal R. Desai, MBBS, MD ESTI SPEAKER SUNDAY Society of Thoracic Radiology San Antonio, Texas March 2014 Diffuse Interstitial Lung Disease The State of Play DILDs Is There
The diagnostic and prognostic utility of
 Eur Respir Rev 2010; 19: 117, 237 241 DOI: 10.1183/09059180.00005510 CopyrightßERS 2010 REVIEW: ENDOSCOPY The clinical utility of bronchoalveolar lavage in diffuse parenchymal lung disease A.U. Wells ABSTRACT:
Eur Respir Rev 2010; 19: 117, 237 241 DOI: 10.1183/09059180.00005510 CopyrightßERS 2010 REVIEW: ENDOSCOPY The clinical utility of bronchoalveolar lavage in diffuse parenchymal lung disease A.U. Wells ABSTRACT:
11/10/2014. Multi-disciplinary Approach to Diffuse Lung Disease: The Imager s Perspective. Radiology
 Multi-disciplinary Approach to Diffuse Lung Disease: The Imager s Perspective Radiology Pathology Clinical 1 Role of HRCT Diagnosis Fibrosis vs. inflammation Next step in management Response to treatment
Multi-disciplinary Approach to Diffuse Lung Disease: The Imager s Perspective Radiology Pathology Clinical 1 Role of HRCT Diagnosis Fibrosis vs. inflammation Next step in management Response to treatment
5/9/2015. Multi-disciplinary Approach to Diffuse Lung Disease: The Imager s Perspective. No, I am not a pulmonologist! Radiology
 Multi-disciplinary Approach to Diffuse Lung Disease: The Imager s Perspective No, I am not a pulmonologist! Radiology Pathology Clinical 1 Everyone needs a CT Confidence in diagnosis Definitive HRCT +
Multi-disciplinary Approach to Diffuse Lung Disease: The Imager s Perspective No, I am not a pulmonologist! Radiology Pathology Clinical 1 Everyone needs a CT Confidence in diagnosis Definitive HRCT +
Bronchoalveolar Lavage and Histopathologic Diagnosis Based on Biopsy
 Idiopathic Pulmonary Fibrosis Bronchoalveolar Lavage and Histopathologic Diagnosis Based on Biopsy JMAJ 46(11): 469 474, 2003 Yukihiko SUGIYAMA Professor, Division of Pulmonary Medicine, Department of
Idiopathic Pulmonary Fibrosis Bronchoalveolar Lavage and Histopathologic Diagnosis Based on Biopsy JMAJ 46(11): 469 474, 2003 Yukihiko SUGIYAMA Professor, Division of Pulmonary Medicine, Department of
4/17/2010 C ini n ca c l a Ev E a v l a ua u t a ion o n of o ILD U dat a e t e i n I LDs
 Update in ILDs Diagnosis 101: Clinical Evaluation April 17, 2010 Jay H. Ryu, MD Mayo Clinic, Rochester MN Clinical Evaluation of ILD Outline General aspects of ILDs Classification of ILDs Clinical evaluation
Update in ILDs Diagnosis 101: Clinical Evaluation April 17, 2010 Jay H. Ryu, MD Mayo Clinic, Rochester MN Clinical Evaluation of ILD Outline General aspects of ILDs Classification of ILDs Clinical evaluation
Manish Powari Regional Training Day 10/12/2014
 Manish Powari Regional Training Day 10/12/2014 Large number of different types of Interstitial Lung Disease (ILD). Most are very rare Most patients present with one of a smaller number of commoner diseases
Manish Powari Regional Training Day 10/12/2014 Large number of different types of Interstitial Lung Disease (ILD). Most are very rare Most patients present with one of a smaller number of commoner diseases
Progress in Idiopathic Pulmonary Fibrosis
 Progress in Idiopathic Pulmonary Fibrosis David A. Lynch, MB Disclosures Progress in Idiopathic Pulmonary Fibrosis David A Lynch, MB Consultant: t Research support: Perceptive Imaging Boehringer Ingelheim
Progress in Idiopathic Pulmonary Fibrosis David A. Lynch, MB Disclosures Progress in Idiopathic Pulmonary Fibrosis David A Lynch, MB Consultant: t Research support: Perceptive Imaging Boehringer Ingelheim
CTD-related Lung Disease
 13 th Cambridge Chest Meeting King s College, Cambridge April 2015 Imaging of CTD-related Lung Disease Dr Sujal R Desai King s College Hospital, London Disclosure Statement No Disclosures / Conflicts of
13 th Cambridge Chest Meeting King s College, Cambridge April 2015 Imaging of CTD-related Lung Disease Dr Sujal R Desai King s College Hospital, London Disclosure Statement No Disclosures / Conflicts of
Case Presentations in ILD. Harold R. Collard, MD Department of Medicine University of California San Francisco
 Case Presentations in ILD Harold R. Collard, MD Department of Medicine University of California San Francisco Outline Overview of diagnosis in ILD Definition/Classification High-resolution CT scan Multidisciplinary
Case Presentations in ILD Harold R. Collard, MD Department of Medicine University of California San Francisco Outline Overview of diagnosis in ILD Definition/Classification High-resolution CT scan Multidisciplinary
Epidemiology and classification of smoking related interstitial lung diseases
 Epidemiology and classification of smoking related interstitial lung diseases Šterclová M. Department of Respiratory Diseases, Thomayer Hospital, Prague, Czech Republic Supported by an IGA Grant No G 1207
Epidemiology and classification of smoking related interstitial lung diseases Šterclová M. Department of Respiratory Diseases, Thomayer Hospital, Prague, Czech Republic Supported by an IGA Grant No G 1207
Financial disclosure COMMON DIAGNOSES IN HRCT. High Res Chest HRCT. HRCT Pre test. I have no financial relationships to disclose. Anatomy Nomenclature
 Financial disclosure I have no financial relationships to disclose. Douglas Johnson D.O. Cardiothoracic Imaging Gaston Radiology COMMON DIAGNOSES IN HRCT High Res Chest Anatomy Nomenclature HRCT Sampling
Financial disclosure I have no financial relationships to disclose. Douglas Johnson D.O. Cardiothoracic Imaging Gaston Radiology COMMON DIAGNOSES IN HRCT High Res Chest Anatomy Nomenclature HRCT Sampling
Clinical Usefulness of Bronchoalveolar Lavage Cellular Analysis and Lymphocyte Subsets in Diffuse Interstitial Lung Diseases
 Original Article Diagnostic Immunology Ann Lab Med 215;35:22-225 http://dx.doi.org/1.3343/alm.215.35.2.22 ISSN 2234-386 eissn 2234-3814 Clinical Usefulness of Bronchoalveolar Lavage Cellular Analysis and
Original Article Diagnostic Immunology Ann Lab Med 215;35:22-225 http://dx.doi.org/1.3343/alm.215.35.2.22 ISSN 2234-386 eissn 2234-3814 Clinical Usefulness of Bronchoalveolar Lavage Cellular Analysis and
INTERSTITIAL LUNG DISEASE. Radhika Reddy MD Pulmonary/Critical Care Long Beach VA Medical Center January 5, 2018
 INTERSTITIAL LUNG DISEASE Radhika Reddy MD Pulmonary/Critical Care Long Beach VA Medical Center January 5, 2018 Interstitial Lung Disease Interstitial Lung Disease Prevalence by Diagnosis: Idiopathic Interstitial
INTERSTITIAL LUNG DISEASE Radhika Reddy MD Pulmonary/Critical Care Long Beach VA Medical Center January 5, 2018 Interstitial Lung Disease Interstitial Lung Disease Prevalence by Diagnosis: Idiopathic Interstitial
International consensus statement on idiopathic pulmonary fibrosis
 Eur Respir J 2001; 17: 163 167 Printed in UK all rights reserved Copyright #ERS Journals Ltd 2001 European Respiratory Journal ISSN 0903-1936 PERSPECTIVE International consensus statement on idiopathic
Eur Respir J 2001; 17: 163 167 Printed in UK all rights reserved Copyright #ERS Journals Ltd 2001 European Respiratory Journal ISSN 0903-1936 PERSPECTIVE International consensus statement on idiopathic
Non-neoplastic Lung Disease II
 Pathobasic Non-neoplastic Lung Disease II Spasenija Savic Prince Pathology Program Systematic approach to surgical lung biopsies with ILD Examples (chronic ILD): Idiopathic interstitial pneumonias: UIP,
Pathobasic Non-neoplastic Lung Disease II Spasenija Savic Prince Pathology Program Systematic approach to surgical lung biopsies with ILD Examples (chronic ILD): Idiopathic interstitial pneumonias: UIP,
Liebow and Carrington's original classification of IIP
 Liebow and Carrington's original classification of IIP-- 1969 Eric J. Stern MD University of Washington UIP Usual interstitial pneumonia DIP Desquamative interstitial pneumonia BIP Bronchiolitis obliterans
Liebow and Carrington's original classification of IIP-- 1969 Eric J. Stern MD University of Washington UIP Usual interstitial pneumonia DIP Desquamative interstitial pneumonia BIP Bronchiolitis obliterans
Diffuse interstitial lung diseases (DILDs) are a heterogeneous group of non-neoplastic, noninfectious
 Focused Issue of This Month Diagnostic Approaches to Diffuse Interstitial Lung Diseases Dong Soon Kim, MD Department of Pulmonary and Critical Care Medicine, University of Ulsan College of Medicine E -
Focused Issue of This Month Diagnostic Approaches to Diffuse Interstitial Lung Diseases Dong Soon Kim, MD Department of Pulmonary and Critical Care Medicine, University of Ulsan College of Medicine E -
Disclosures. Fibrotic lung diseases: Basic Principles, Common Problems, and Reporting. Relevant financial relationships: None. Off-label usage: None
 Fibrotic lung diseases: Basic Principles, Common Problems, and Reporting Brandon T. Larsen, MD, PhD Senior Associate Consultant Department of Laboratory Medicine and Pathology Mayo Clinic Arizona Arizona
Fibrotic lung diseases: Basic Principles, Common Problems, and Reporting Brandon T. Larsen, MD, PhD Senior Associate Consultant Department of Laboratory Medicine and Pathology Mayo Clinic Arizona Arizona
Case 1 : Question. 1.1 What is the intralobular distribution? 1. Centrilobular 2. Perilymphatic 3. Random
 Interesting case Case 1 Case 1 : Question 1.1 What is the intralobular distribution? 1. Centrilobular 2. Perilymphatic 3. Random Case 1: Answer 1.1 What is the intralobular distribution? 1. Centrilobular
Interesting case Case 1 Case 1 : Question 1.1 What is the intralobular distribution? 1. Centrilobular 2. Perilymphatic 3. Random Case 1: Answer 1.1 What is the intralobular distribution? 1. Centrilobular
Acute and Chronic Lung Disease
 KATHOLIEKE UNIVERSITEIT LEUVEN Faculty of Medicine Acute and Chronic Lung Disease W De Wever, JA Verschakelen Department of Radiology, University Hospitals Leuven, Belgium Clinical utility of HRCT To detect
KATHOLIEKE UNIVERSITEIT LEUVEN Faculty of Medicine Acute and Chronic Lung Disease W De Wever, JA Verschakelen Department of Radiology, University Hospitals Leuven, Belgium Clinical utility of HRCT To detect
A Review of Interstitial Lung Diseases. Paul J. Wolters, MD Associate Professor Department of Medicine University of California San Francisco
 A Review of Interstitial Lung Diseases Paul J. Wolters, MD Associate Professor Department of Medicine University of California San Francisco Outline Overview of diagnosis in ILD Why it is important Definition/Classification
A Review of Interstitial Lung Diseases Paul J. Wolters, MD Associate Professor Department of Medicine University of California San Francisco Outline Overview of diagnosis in ILD Why it is important Definition/Classification
NONE OVERVIEW FINANCIAL DISCLOSURES UPDATE ON IDIOPATHIC PULMONARY FIBROSIS/IPF (UIP) FOR PATHOLOGISTS. IPF = Idiopathic UIP Radiologic UIP Path UIP
 UPDATE ON IDIOPATHIC PULMONARY FIBROSIS/IPF () FOR PATHOLOGISTS Thomas V. Colby, M.D. Professor of Pathology (Emeritus) Mayo Clinic Arizona FINANCIAL DISCLOSURES NONE OVERVIEW IPF Radiologic Dx Pathologic
UPDATE ON IDIOPATHIC PULMONARY FIBROSIS/IPF () FOR PATHOLOGISTS Thomas V. Colby, M.D. Professor of Pathology (Emeritus) Mayo Clinic Arizona FINANCIAL DISCLOSURES NONE OVERVIEW IPF Radiologic Dx Pathologic
A Review of Interstitial Lung Diseases
 Outline A Review of Interstitial Lung Diseases Paul J. Wolters, MD Associate Professor Department of Medicine University of California San Francisco Overview of diagnosis in ILD Why it is important Definition/Classification
Outline A Review of Interstitial Lung Diseases Paul J. Wolters, MD Associate Professor Department of Medicine University of California San Francisco Overview of diagnosis in ILD Why it is important Definition/Classification
The radiological differential diagnosis of the UIP pattern
 5th International Conference on Idiopathic Pulmonary Fibrosis, Modena, 2015, June 12th The radiological differential diagnosis of the UIP pattern Simon Walsh King s College Hospital Foundation Trust London,
5th International Conference on Idiopathic Pulmonary Fibrosis, Modena, 2015, June 12th The radiological differential diagnosis of the UIP pattern Simon Walsh King s College Hospital Foundation Trust London,
DIFFERENCES IN FIBROPROLIFERATIVE HEALING IN EXOGENEOUS AND IDIOPATHIC ILDs. ARE THERE ANY?
 NIHR Southampton Respiratory Biomedical Research Unit DIFFERENCES IN FIBROPROLIFERATIVE HEALING IN EXOGENEOUS AND IDIOPATHIC ILDs. ARE THERE ANY? Fibrosing Interstitial Lung Diseases CPFS/WASOG/AIPO/ERS
NIHR Southampton Respiratory Biomedical Research Unit DIFFERENCES IN FIBROPROLIFERATIVE HEALING IN EXOGENEOUS AND IDIOPATHIC ILDs. ARE THERE ANY? Fibrosing Interstitial Lung Diseases CPFS/WASOG/AIPO/ERS
Difficulties Diagnosing Idiopathic Pulmonary Fibrosis
 1. er Encuentro Entre Neumólogos y Radiólogos, Madrid, Spain, 2016, October 14th Difficulties Diagnosing Idiopathic Pulmonary Fibrosis Simon Walsh King s College Hospital Foundation Trust London, United
1. er Encuentro Entre Neumólogos y Radiólogos, Madrid, Spain, 2016, October 14th Difficulties Diagnosing Idiopathic Pulmonary Fibrosis Simon Walsh King s College Hospital Foundation Trust London, United
IPF AND OTHER FIBROSING LUNG DISEASE: WHAT DRUGS MIGHT WORK AND ON WHOM DO THEY W ORK?
 IPF AND OTHER FIBROSING LUNG DISEASE: WHAT DRUGS MIGHT WORK AND ON WHOM DO THEY W ORK? KEVIN K. BROWN, MD PROFESSOR AND VICE CHAIRMAN, DEPARTMENT OF MEDICINE NATIONAL JEWISH HEALTH DENVER, CO Kevin K.
IPF AND OTHER FIBROSING LUNG DISEASE: WHAT DRUGS MIGHT WORK AND ON WHOM DO THEY W ORK? KEVIN K. BROWN, MD PROFESSOR AND VICE CHAIRMAN, DEPARTMENT OF MEDICINE NATIONAL JEWISH HEALTH DENVER, CO Kevin K.
Pulmonary manifestations of CTDs Diagnosis, differential diagnosis and treatment
 Prague, June 2014 Pulmonary manifestations of CTDs Diagnosis, differential diagnosis and treatment Katerina M. Antoniou, MD, PhD As. Professor in Thoracic Medicine ERS ILD Group Secretary Medical School,
Prague, June 2014 Pulmonary manifestations of CTDs Diagnosis, differential diagnosis and treatment Katerina M. Antoniou, MD, PhD As. Professor in Thoracic Medicine ERS ILD Group Secretary Medical School,
Usual Interstitial pneumonia and Nonspecific Interstitial Pneumonia. Nitra and the Gangs.
 Usual Interstitial pneumonia and Nonspecific Interstitial Pneumonia Nitra and the Gangs. บทน ำและบทท ๓, ๑๐, ๑๒, ๑๓, ๑๔, ๑๕, ๑๗ Usual Interstitial Pneumonia (UIP) Most common & basic pathologic pattern
Usual Interstitial pneumonia and Nonspecific Interstitial Pneumonia Nitra and the Gangs. บทน ำและบทท ๓, ๑๐, ๑๒, ๑๓, ๑๔, ๑๕, ๑๗ Usual Interstitial Pneumonia (UIP) Most common & basic pathologic pattern
Diagnosing Idiopathic Pulmonary Fibrosis on Evidence-Based Guidelines
 Diagnosing Idiopathic Pulmonary Fibrosis on Evidence-Based Guidelines Rebecca Keith, MD Assistant Professor, Division of Pulmonary and Critical Care Medicine National Jewish Health, Denver, CO Objectives
Diagnosing Idiopathic Pulmonary Fibrosis on Evidence-Based Guidelines Rebecca Keith, MD Assistant Professor, Division of Pulmonary and Critical Care Medicine National Jewish Health, Denver, CO Objectives
Imaging: how to recognise idiopathic pulmonary fibrosis
 REVIEW IDIOPATHIC PULMONARY FIBROSIS Imaging: how to recognise idiopathic pulmonary fibrosis Anand Devaraj Affiliations: Dept of Radiology, St George s Hospital, London, UK. Correspondence: Anand Devaraj,
REVIEW IDIOPATHIC PULMONARY FIBROSIS Imaging: how to recognise idiopathic pulmonary fibrosis Anand Devaraj Affiliations: Dept of Radiology, St George s Hospital, London, UK. Correspondence: Anand Devaraj,
Bronchoalveolar lavage in fibrotic idiopathic interstitial pneumonias
 Respiratory Medicine (2007) 101, 655 660 Bronchoalveolar lavage in fibrotic idiopathic interstitial pneumonias Yon Ju Ryu a, Man Pyo Chung b,, Joungho Han c, Tae Sung Kim d, Kyung Soo Lee d, Eun-Mi Chun
Respiratory Medicine (2007) 101, 655 660 Bronchoalveolar lavage in fibrotic idiopathic interstitial pneumonias Yon Ju Ryu a, Man Pyo Chung b,, Joungho Han c, Tae Sung Kim d, Kyung Soo Lee d, Eun-Mi Chun
IPF: Epidemiologia e stato dell arte
 IPF: Epidemiologia e stato dell arte Clinical Classification Diffuse parenchimal lung diseases Exposure-related: - occupational - environmental - medication Desquamative interstitial pneumonia Idiopathic
IPF: Epidemiologia e stato dell arte Clinical Classification Diffuse parenchimal lung diseases Exposure-related: - occupational - environmental - medication Desquamative interstitial pneumonia Idiopathic
HYPERSENSITIVITY PNEUMONITIS
 HYPERSENSITIVITY PNEUMONITIS A preventable fibrosis MOSAVIR ANSARIE MB., FCCP INTERSTITIAL LUNG DISEASES A heterogeneous group of non infectious, non malignant diffuse parenchymal disorders of the lower
HYPERSENSITIVITY PNEUMONITIS A preventable fibrosis MOSAVIR ANSARIE MB., FCCP INTERSTITIAL LUNG DISEASES A heterogeneous group of non infectious, non malignant diffuse parenchymal disorders of the lower
Overview of Idiopathic Pulmonary Fibrosis: Diagnosis and Therapy
 Overview of Idiopathic Pulmonary Fibrosis: Diagnosis and Therapy Jeff Swigris, DO, MS Director, ILD Program National Jewish Health Disclosures Speaker - Boehringer Ingelheim and Genentech Objectives Describe
Overview of Idiopathic Pulmonary Fibrosis: Diagnosis and Therapy Jeff Swigris, DO, MS Director, ILD Program National Jewish Health Disclosures Speaker - Boehringer Ingelheim and Genentech Objectives Describe
TBLB is not recommended as the initial biopsy option in cases of suspected IPF and is unreliable in the diagnosis of rare lung disease (other than
 TBLB is not recommended as the initial biopsy option in cases of suspected IPF and is unreliable in the diagnosis of rare lung disease (other than PAP) BAL is not required as a diagnostic tool in patients
TBLB is not recommended as the initial biopsy option in cases of suspected IPF and is unreliable in the diagnosis of rare lung disease (other than PAP) BAL is not required as a diagnostic tool in patients
Immunocompromised patients. Immunocompromised patients. Immunocompromised patients
 Value of CT in Early Pneumonia in Immunocompromised Patients Nantaka Kiranantawat, PSU Preventative Factors Phagocyts Cellular immunity Humoral immunity Predisposing Factors Infection, Stress, Poor nutrition,
Value of CT in Early Pneumonia in Immunocompromised Patients Nantaka Kiranantawat, PSU Preventative Factors Phagocyts Cellular immunity Humoral immunity Predisposing Factors Infection, Stress, Poor nutrition,
Lines and crackles. Making sense of ILD
 Lines and crackles Making sense of ILD Case JM 65 year old male Gradual shortness of breath, going on over a year Some dry cough Ex-smoker, quit 10 years ago Crackles in the bases CXR presented Sent to
Lines and crackles Making sense of ILD Case JM 65 year old male Gradual shortness of breath, going on over a year Some dry cough Ex-smoker, quit 10 years ago Crackles in the bases CXR presented Sent to
Respiratory Pathology. Kristine Krafts, M.D.
 Respiratory Pathology Kristine Krafts, M.D. Normal lung: alveolar spaces Respiratory Pathology Outline Acute respiratory distress syndrome Obstructive lung diseases Restrictive lung diseases Vascular
Respiratory Pathology Kristine Krafts, M.D. Normal lung: alveolar spaces Respiratory Pathology Outline Acute respiratory distress syndrome Obstructive lung diseases Restrictive lung diseases Vascular
The Egyptian Journal of Hospital Medicine (July 2017) Vol.68 (2), Page
 The Egyptian Journal of Hospital Medicine (July 2017) Vol.68 (2), Page 1135-1140 Role of High Resolution Computed Tomography in Diagnosis of Interstitial Lung Diseases in Patients with Collagen Diseases
The Egyptian Journal of Hospital Medicine (July 2017) Vol.68 (2), Page 1135-1140 Role of High Resolution Computed Tomography in Diagnosis of Interstitial Lung Diseases in Patients with Collagen Diseases
HRCT in Diffuse Interstitial Lung Disease Steps in High Resolution CT Diagnosis. Where are the lymphatics? Anatomic distribution
 Steps in High Resolution CT Diagnosis Pattern of abnormality Distribution of disease Associated findings Clinical history Tomás Franquet MD What is the diagnosis? Hospital de Sant Pau. Barcelona Secondary
Steps in High Resolution CT Diagnosis Pattern of abnormality Distribution of disease Associated findings Clinical history Tomás Franquet MD What is the diagnosis? Hospital de Sant Pau. Barcelona Secondary
Diagnostic challenges in IPF
 Medicine, Nursing and Health Sciences Diagnostic challenges in IPF Dr Ian Glaspole Central and Eastern Clinical School, Alfred Hospital and Monash University March 2015 Disclosures Consultancy fees from
Medicine, Nursing and Health Sciences Diagnostic challenges in IPF Dr Ian Glaspole Central and Eastern Clinical School, Alfred Hospital and Monash University March 2015 Disclosures Consultancy fees from
Daria Manos RSNA 2016 RC 401. https://medicine.dal.ca/departments/depar tment-sites/radiology/contact/faculty/dariamanos.html
 Daria Manos RSNA 2016 RC 401 https://medicine.dal.ca/departments/depar tment-sites/radiology/contact/faculty/dariamanos.html STEP1: Is this fibrotic lung disease? STEP 2: Is this a UIP pattern? If yes:
Daria Manos RSNA 2016 RC 401 https://medicine.dal.ca/departments/depar tment-sites/radiology/contact/faculty/dariamanos.html STEP1: Is this fibrotic lung disease? STEP 2: Is this a UIP pattern? If yes:
Bronchoscopic lung cryobiopsy increases diagnostic confidence. in the multidisciplinary diagnosis of idiopathic pulmonary
 Bronchoscopic lung cryobiopsy increases diagnostic confidence in the multidisciplinary diagnosis of idiopathic pulmonary fibrosis. Sara Tomassetti, Athol U Wells, Ulrich Costabel, Alberto Cavazza, Thomas
Bronchoscopic lung cryobiopsy increases diagnostic confidence in the multidisciplinary diagnosis of idiopathic pulmonary fibrosis. Sara Tomassetti, Athol U Wells, Ulrich Costabel, Alberto Cavazza, Thomas
Radiologic Approach to Smoking Related Interstitial Lung Disease
 Radiologic Approach to Smoking Related Interstitial Lung Disease Poster No.: C-1854 Congress: ECR 2013 Type: Educational Exhibit Authors: K.-N. Lee, J.-Y. Han, E.-J. Kang, J. Kang; Busan/KR Keywords: Toxicity,
Radiologic Approach to Smoking Related Interstitial Lung Disease Poster No.: C-1854 Congress: ECR 2013 Type: Educational Exhibit Authors: K.-N. Lee, J.-Y. Han, E.-J. Kang, J. Kang; Busan/KR Keywords: Toxicity,
Restrictive lung diseases
 Restrictive lung diseases Restrictive lung diseases are diseases that affect the interstitium of the lung. Interstitium of the lung is the very thin walls surrounding the alveoli, it s formed of epithelium
Restrictive lung diseases Restrictive lung diseases are diseases that affect the interstitium of the lung. Interstitium of the lung is the very thin walls surrounding the alveoli, it s formed of epithelium
Cryptogenic Organizing Pneumonia Diagnosis Approach Based on a Clinical-Radiologic-Pathologic Consensus
 Cryptogenic Organizing Pneumonia Diagnosis Approach Based on a Clinical-Radiologic-Pathologic Consensus Poster No.: C-1622 Congress: ECR 2012 Type: Scientific Exhibit Authors: C. Cordero Lares, E. Zorita
Cryptogenic Organizing Pneumonia Diagnosis Approach Based on a Clinical-Radiologic-Pathologic Consensus Poster No.: C-1622 Congress: ECR 2012 Type: Scientific Exhibit Authors: C. Cordero Lares, E. Zorita
Bronkhorst colloquium Interstitiële longziekten. Katrien Grünberg, klinisch patholoog
 Bronkhorst colloquium 2013-2014 Interstitiële longziekten De pathologie achter de CT Katrien Grünberg, klinisch patholoog K.grunberg@vumc.nl Preparing: introduction and 3 cases The introduction on microscopic
Bronkhorst colloquium 2013-2014 Interstitiële longziekten De pathologie achter de CT Katrien Grünberg, klinisch patholoog K.grunberg@vumc.nl Preparing: introduction and 3 cases The introduction on microscopic
* * APPROACH TO NON- NEOPLASTIC LUNG DISEASE IN TRANSBRONCHIAL AND SURGICAL BIOPSIES. Financial Disclosures: NONE. BIOPSY TECHNIQUES Bronchoscopic
 APPROACH TO NON- NEOPLASTIC LUNG DISEASE IN TRANSBRONCHIAL AND SURGICAL BIOPSIES Thomas V. Colby, M.D. Mayo Clinic Arizona Geraldine C. Zeiler Professor of Cytopathology Mayo Clinic Arizona Financial Disclosures:
APPROACH TO NON- NEOPLASTIC LUNG DISEASE IN TRANSBRONCHIAL AND SURGICAL BIOPSIES Thomas V. Colby, M.D. Mayo Clinic Arizona Geraldine C. Zeiler Professor of Cytopathology Mayo Clinic Arizona Financial Disclosures:
Katerina M. Antoniou, MD, PhD As. Professor in Thoracic Medicine ERS ILD Group Secretary Medical School, University of Crete Prague, June 2014
 Hypersensitivity pneumonitis: Causes, clinical course, diagnosis and differential diagnosis, treatment Katerina M. Antoniou, MD, PhD As. Professor in Thoracic Medicine ERS ILD Group Secretary Medical School,
Hypersensitivity pneumonitis: Causes, clinical course, diagnosis and differential diagnosis, treatment Katerina M. Antoniou, MD, PhD As. Professor in Thoracic Medicine ERS ILD Group Secretary Medical School,
IPF - Inquadramento clinico
 IPF - Inquadramento clinico Sergio Harari Unità Operativa di Pneumologia UTIR Servizio di Fisiopat. Resp. e Emodinamica Polmonare Ospedale S. Giuseppe, Milano Clinical Classification Diffuse parenchimal
IPF - Inquadramento clinico Sergio Harari Unità Operativa di Pneumologia UTIR Servizio di Fisiopat. Resp. e Emodinamica Polmonare Ospedale S. Giuseppe, Milano Clinical Classification Diffuse parenchimal
New respiratory symptoms and lung imaging findings in a woman with polymyositis
 Maria Bolaki 1, Konstantinos Karagiannis 1, George Bertsias 2, Ioanna Mitrouska 1, Nikolaos Tzanakis 1, Katerina M. Antoniou 1 kantoniou@uoc.gr 1 Dept of Thoracic Medicine, Heraklion University Hospital,
Maria Bolaki 1, Konstantinos Karagiannis 1, George Bertsias 2, Ioanna Mitrouska 1, Nikolaos Tzanakis 1, Katerina M. Antoniou 1 kantoniou@uoc.gr 1 Dept of Thoracic Medicine, Heraklion University Hospital,
Organizing Pneumonia And Diffuse Alveolar Damage: An Incidental Finding In An Immunocompromised Patient By EBUS-FNA
 ISPUB.COM The Internet Journal of Pathology Volume 17 Number 1 Organizing Pneumonia And Diffuse Alveolar Damage: An Incidental Finding In An Immunocompromised Patient By EBUS-FNA B Lowenthal, F Hasteh
ISPUB.COM The Internet Journal of Pathology Volume 17 Number 1 Organizing Pneumonia And Diffuse Alveolar Damage: An Incidental Finding In An Immunocompromised Patient By EBUS-FNA B Lowenthal, F Hasteh
Disclosures. Traditional Paradigm. Overview 4/17/2010. I have relationships with the following organizations and companies:
 Disclosures Pharmacological Therapy for ILD What to Use and How to Use It Harold R Collard MD Interstitial Lung Disease Program University of California San Francisco (UCSF) I have relationships with the
Disclosures Pharmacological Therapy for ILD What to Use and How to Use It Harold R Collard MD Interstitial Lung Disease Program University of California San Francisco (UCSF) I have relationships with the
Unpaid scientific collaborator & advisor with Veracyte, Inc.
 Diagnosis and Classification of Idiopathic Interstitial Pneumonias: Role of Histopathology in the Golden Age of Consensus Jeffrey L. Myers, M.D. A. James French Professor of Diagnostic Pathology Vice Chair
Diagnosis and Classification of Idiopathic Interstitial Pneumonias: Role of Histopathology in the Golden Age of Consensus Jeffrey L. Myers, M.D. A. James French Professor of Diagnostic Pathology Vice Chair
Case 4 History. 58 yo man presented with prox IP joint swelling 2 months later pain and swelling in multiple joints Chest radiograph: bi-basilar
 Case 4 History 58 yo man presented with prox IP joint swelling 2 months later pain and swelling in multiple joints Chest radiograph: bi-basilar basilar infiltrates suggestive of pulmonary fibrosis Open
Case 4 History 58 yo man presented with prox IP joint swelling 2 months later pain and swelling in multiple joints Chest radiograph: bi-basilar basilar infiltrates suggestive of pulmonary fibrosis Open
Idiopathic Pulmonary Fibrosis: The Importance of Qualitative and Quantitative Phenotyping
 Idiopathic Pulmonary Fibrosis: The Importance of Qualitative and Quantitative Phenotyping K. R. Flaherty Division of Pulmonary and Critical Care Medicine, University of Michigan Health System, Ann Arbor,
Idiopathic Pulmonary Fibrosis: The Importance of Qualitative and Quantitative Phenotyping K. R. Flaherty Division of Pulmonary and Critical Care Medicine, University of Michigan Health System, Ann Arbor,
BRONCHOALVEOLAR LAVAGE (BAL) Venerino Poletti Osp GB Morgagni, Forlì (I)
 BRONCHOALVEOLAR LAVAGE (BAL) Venerino Poletti Osp GB Morgagni, Forlì (I) BAL-OTELLI Typical microliths (calcospherites) Acute Respiratory Failure. Clinical history. Treated for GI cancer Bollèe G, et
BRONCHOALVEOLAR LAVAGE (BAL) Venerino Poletti Osp GB Morgagni, Forlì (I) BAL-OTELLI Typical microliths (calcospherites) Acute Respiratory Failure. Clinical history. Treated for GI cancer Bollèe G, et
Hypersensitivity Pneumonitis Common Diagnostic and Treatment Dilemmas
 Hypersensitivity Pneumonitis Common Diagnostic and Treatment Dilemmas Rishi Raj MD Director, Interstitial Lung Diseases Program Clinical Professor of Pulmonary and Critical Care Medicine Stanford University
Hypersensitivity Pneumonitis Common Diagnostic and Treatment Dilemmas Rishi Raj MD Director, Interstitial Lung Diseases Program Clinical Professor of Pulmonary and Critical Care Medicine Stanford University
Imaging findings in Hypersensitivity Pneumonitis - a pictorical review.
 Imaging findings in Hypersensitivity Pneumonitis - a pictorical review. Poster No.: C-1655 Congress: ECR 2014 Type: Educational Exhibit Authors: B. M. Araujo, A. F. S. Simões, M. S. C. Rodrigues, J. Pereira;
Imaging findings in Hypersensitivity Pneumonitis - a pictorical review. Poster No.: C-1655 Congress: ECR 2014 Type: Educational Exhibit Authors: B. M. Araujo, A. F. S. Simões, M. S. C. Rodrigues, J. Pereira;
Smoking-related Interstitial Lung Diseases: High-Resolution CT Findings
 Smoking-related Interstitial Lung Diseases: High-Resolution CT Findings Poster No.: C-2358 Congress: ECR 2013 Type: Educational Exhibit Authors: V. Cuartero Revilla, M. Nogueras Carrasco, P. Olmedilla
Smoking-related Interstitial Lung Diseases: High-Resolution CT Findings Poster No.: C-2358 Congress: ECR 2013 Type: Educational Exhibit Authors: V. Cuartero Revilla, M. Nogueras Carrasco, P. Olmedilla
Lung Allograft Dysfunction
 Lung Allograft Dysfunction Carlos S. Restrepo M.D. Ameya Baxi M.D. Department of Radiology University of Texas Health San Antonio Disclaimer: We do not have any conflict of interest or financial gain to
Lung Allograft Dysfunction Carlos S. Restrepo M.D. Ameya Baxi M.D. Department of Radiology University of Texas Health San Antonio Disclaimer: We do not have any conflict of interest or financial gain to
Criteria for confident HRCT diagnosis of usual interstitial pneumonia (UIP)
 Criteria for confident HRCT diagnosis of usual interstitial pneumonia (UIP) Assem El Essawy (1) & Amr A. Nassef (٢) Abstract Identification of interstitial pneumonia (IP) was mainly based on histological
Criteria for confident HRCT diagnosis of usual interstitial pneumonia (UIP) Assem El Essawy (1) & Amr A. Nassef (٢) Abstract Identification of interstitial pneumonia (IP) was mainly based on histological
Pathologic Assessment of Interstitial Lung Disease
 Pathologic Assessment of Interstitial Lung Disease Dry and itchy? It could be eczema or fungal infection. We don t need to worry, the drugs aren t that dangerous. Kirk D. Jones, MD UCSF Dept. of Pathology
Pathologic Assessment of Interstitial Lung Disease Dry and itchy? It could be eczema or fungal infection. We don t need to worry, the drugs aren t that dangerous. Kirk D. Jones, MD UCSF Dept. of Pathology
Idiopathic interstitial pneumonias (IIPs) are a group of
 SYMPOSIA C. Isabela S. Silva, MD, PhD and Nestor L. Müller, MD, PhD Abstract: The idiopathic interstitial pneumonias (IIPs) are a group of diffuse parenchymal lung diseases of unknown etiology characterized
SYMPOSIA C. Isabela S. Silva, MD, PhD and Nestor L. Müller, MD, PhD Abstract: The idiopathic interstitial pneumonias (IIPs) are a group of diffuse parenchymal lung diseases of unknown etiology characterized
Nitrofurantoin-Induced Lung Toxicity
 Severe Nitrofurantoin-Induced Lung Toxicity Rami Jambeih, M.D. 1, John Flesher, M.D. 1,3, Joe J. Lin, M.D. 2,4 University of Kansas School of Medicine Wichita 1 Department of Internal Medicine 2 Department
Severe Nitrofurantoin-Induced Lung Toxicity Rami Jambeih, M.D. 1, John Flesher, M.D. 1,3, Joe J. Lin, M.D. 2,4 University of Kansas School of Medicine Wichita 1 Department of Internal Medicine 2 Department
DIAGNOSTIC NOTE TEMPLATE
 DIAGNOSTIC NOTE TEMPLATE SOAP NOTE TEMPLATE WHEN CONSIDERING A DIAGNOSIS OF IDIOPATHIC PULMONARY FIBROSIS (IPF) CHIEF COMPLAINT HISTORY OF PRESENT ILLNESS Consider IPF as possible diagnosis if any of the
DIAGNOSTIC NOTE TEMPLATE SOAP NOTE TEMPLATE WHEN CONSIDERING A DIAGNOSIS OF IDIOPATHIC PULMONARY FIBROSIS (IPF) CHIEF COMPLAINT HISTORY OF PRESENT ILLNESS Consider IPF as possible diagnosis if any of the
Connective Tissue Disorder- Associated Interstitial Lung Disease (CTD-ILD) and Updates
 Connective Tissue Disorder- Associated Interstitial Lung Disease (CTD-ILD) and Updates Maria Elena Vega, M.D Assistant Professor of Medicine Lewis Katz School of Medicine at Temple University Nothing to
Connective Tissue Disorder- Associated Interstitial Lung Disease (CTD-ILD) and Updates Maria Elena Vega, M.D Assistant Professor of Medicine Lewis Katz School of Medicine at Temple University Nothing to
T he diagnostic evaluation of a patient with
 546 REVIEW SERIES Challenges in pulmonary fibrosis? 1: Use of high resolution CT scanning of the lung for the evaluation of patients with idiopathic interstitial pneumonias Michael B Gotway, Michelle M
546 REVIEW SERIES Challenges in pulmonary fibrosis? 1: Use of high resolution CT scanning of the lung for the evaluation of patients with idiopathic interstitial pneumonias Michael B Gotway, Michelle M
UIP Possibile e Probabile
 UIP Possibile e Probabile Sergio Harari U.O. di Pneumologia e UTIR Servizio di Emodinamica e Fisiopatologia Respiratoria Ospedale San Giuseppe - Milano Current definition of IPF IPF is a distinct type
UIP Possibile e Probabile Sergio Harari U.O. di Pneumologia e UTIR Servizio di Emodinamica e Fisiopatologia Respiratoria Ospedale San Giuseppe - Milano Current definition of IPF IPF is a distinct type
Atopic Pulmonary Disease: Findings on Thoracic Imaging
 July 2003 Atopic Pulmonary Disease: Findings on Thoracic Imaging Rebecca G. Breslow Harvard Medical School Year IV Churg-Strauss Syndrome Hypersensitivity Pneumonitis Asthma Atopic Pulmonary Disease Allergic
July 2003 Atopic Pulmonary Disease: Findings on Thoracic Imaging Rebecca G. Breslow Harvard Medical School Year IV Churg-Strauss Syndrome Hypersensitivity Pneumonitis Asthma Atopic Pulmonary Disease Allergic
June 2013 Pulmonary Case of the Month: Diagnosis Makes a Difference. Lewis J. Wesselius, MD 1 Henry D. Tazelaar, MD 2
 June 2013 Pulmonary Case of the Month: Diagnosis Makes a Difference Lewis J. Wesselius, MD 1 Henry D. Tazelaar, MD 2 Departments of Pulmonary Medicine 1 and Laboratory Medicine and Pathology 2 Mayo Clinic
June 2013 Pulmonary Case of the Month: Diagnosis Makes a Difference Lewis J. Wesselius, MD 1 Henry D. Tazelaar, MD 2 Departments of Pulmonary Medicine 1 and Laboratory Medicine and Pathology 2 Mayo Clinic
an inflammation of the bronchial tubes
 BRONCHITIS DEFINITION Bronchitis is an inflammation of the bronchial tubes (or bronchi), which are the air passages that extend from the trachea into the small airways and alveoli. Triggers may be infectious
BRONCHITIS DEFINITION Bronchitis is an inflammation of the bronchial tubes (or bronchi), which are the air passages that extend from the trachea into the small airways and alveoli. Triggers may be infectious
Case 1: Question. 1.1 What is the main pattern of this HRCT? 1. Intralobular line 2. Groundglass opacity 3. Perilymphatic nodule
 HRCT WORK SHOP Case 1 Case 1: Question 1.1 What is the main pattern of this HRCT? 1. Intralobular line 2. Groundglass opacity 3. Perilymphatic nodule Case 1: Question 1.2 What is the diagnosis? 1. Hypersensitivity
HRCT WORK SHOP Case 1 Case 1: Question 1.1 What is the main pattern of this HRCT? 1. Intralobular line 2. Groundglass opacity 3. Perilymphatic nodule Case 1: Question 1.2 What is the diagnosis? 1. Hypersensitivity
Pathobasic Non-neoplastic Lung Disease I. Spasenija Savic Prince Pathology
 Pathobasic Non-neoplastic Lung Disease I Spasenija Savic Prince Pathology Program Introduction to interstitial lung disease (ILD)/ diffuse parenchymal lung disease (DLD) Respiratory specimens Overview
Pathobasic Non-neoplastic Lung Disease I Spasenija Savic Prince Pathology Program Introduction to interstitial lung disease (ILD)/ diffuse parenchymal lung disease (DLD) Respiratory specimens Overview
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Idiopathic Pulmonary Fibrosis Page 1 of 10 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Idiopathic Pulmonary Fibrosis (Esbriet /pirfenidone, Ofev /nintedanib)
Idiopathic Pulmonary Fibrosis Page 1 of 10 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Idiopathic Pulmonary Fibrosis (Esbriet /pirfenidone, Ofev /nintedanib)
ARTICLE IN PRESS. Ahuva Grubstein a, Daniele Bendayan b, Ithak Schactman c, Maya Cohen a, David Shitrit b, Mordechai R. Kramer b,
 Respiratory Medicine (2005) 99, 948 954 Concomitant upper-lobe bullous emphysema, lower-lobe interstitial fibrosis and pulmonary hypertension in heavy smokers: report of eight cases and review of the literature
Respiratory Medicine (2005) 99, 948 954 Concomitant upper-lobe bullous emphysema, lower-lobe interstitial fibrosis and pulmonary hypertension in heavy smokers: report of eight cases and review of the literature
ERS 2016 Congress Highlights Interstitial Lung Disease (ILD)
 ERS 216 Congress Highlights Interstitial Lung Disease (ILD) London, UK September 3 rd 7 th 216 The 26 th European Respiratory Society International Congress, (ERS) the largest respiratory meeting in the
ERS 216 Congress Highlights Interstitial Lung Disease (ILD) London, UK September 3 rd 7 th 216 The 26 th European Respiratory Society International Congress, (ERS) the largest respiratory meeting in the
Chronic Cough Due to Chronic Interstitial Pulmonary Diseases. ACCP Evidence-Based Clinical Practice Guidelines
 Chronic Cough Due to Chronic Interstitial Pulmonary Diseases ACCP Evidence-Based Clinical Practice Guidelines Kevin K. Brown, MD, FCCP Objectives: To review the role of chronic interstitial pulmonary disease
Chronic Cough Due to Chronic Interstitial Pulmonary Diseases ACCP Evidence-Based Clinical Practice Guidelines Kevin K. Brown, MD, FCCP Objectives: To review the role of chronic interstitial pulmonary disease
An earlier and more confident diagnosis of idiopathic pulmonary fibrosis
 Eur Respir Rev 2012; 21: 124, 141 146 DOI: 10.1183/09059180.00000812 CopyrightßERS 2012 REVIEW: IPF An earlier and more confident diagnosis of idiopathic pulmonary fibrosis Roland M. du Bois ABSTRACT:
Eur Respir Rev 2012; 21: 124, 141 146 DOI: 10.1183/09059180.00000812 CopyrightßERS 2012 REVIEW: IPF An earlier and more confident diagnosis of idiopathic pulmonary fibrosis Roland M. du Bois ABSTRACT:
RADIOLOGIC EVALUATION OF PULMONARY NTM INFECTION. Tilman Koelsch, MD National Jewish Health - Department of Radiology
 Pr N op ot er fo ty r R of ep Pr ro es du en ct te io r n RADIOLOGIC EVALUATION OF PULMONARY NTM INFECTION Tilman Koelsch, MD National Jewish Health - Department of Radiology Disclosures No relevant financial
Pr N op ot er fo ty r R of ep Pr ro es du en ct te io r n RADIOLOGIC EVALUATION OF PULMONARY NTM INFECTION Tilman Koelsch, MD National Jewish Health - Department of Radiology Disclosures No relevant financial
INVITED REVIEW SERIES: PULMONARY FIBROSIS SERIES EDITORS: MARTIN KOLB AND GERARD COX
 INVITED REVIEW SERIES: PULMONARY FIBROSIS SERIES EDITORS: MARTIN KOLB AND GERARD COX Diagnosing fibrotic lung disease: When is high-resolution computed tomography sufficient to make a diagnosis of idiopathic
INVITED REVIEW SERIES: PULMONARY FIBROSIS SERIES EDITORS: MARTIN KOLB AND GERARD COX Diagnosing fibrotic lung disease: When is high-resolution computed tomography sufficient to make a diagnosis of idiopathic
Wedge Biopsy for Diffuse Lung Diseases
 Chapter VI Wedge Biopsy for Diffuse Lung Diseases Wedge biopsy via thoracoscopic biopsy or open lung biopsy is occasionally performed to obtain tissue for the diagnosis of a diffuse lung disease. A wedge
Chapter VI Wedge Biopsy for Diffuse Lung Diseases Wedge biopsy via thoracoscopic biopsy or open lung biopsy is occasionally performed to obtain tissue for the diagnosis of a diffuse lung disease. A wedge
INTERSTITIAL LUNG DISEASES: FOCUS ON IDIOPATHIC PULMONARY FIBROSIS (IPF)
 INTERSTITIAL LUNG DISEASES: FOCUS ON IDIOPATHIC PULMONARY FIBROSIS (IPF) Marilyn K. Glassberg Csete, M.D. Professor of Medicine, Surgery, and Pediatrics Director, Interstitial and Rare Lung Disease Program
INTERSTITIAL LUNG DISEASES: FOCUS ON IDIOPATHIC PULMONARY FIBROSIS (IPF) Marilyn K. Glassberg Csete, M.D. Professor of Medicine, Surgery, and Pediatrics Director, Interstitial and Rare Lung Disease Program
Hypersensitivity Pneumonitis: Epidemiology and Classification
 Hypersensitivity Pneumonitis: Epidemiology and Classification Ulrich Costabel, MD University of Duisburg-Essen, Ruhrlandklinik Department of Pneumology/Allergy Objectives Definitions, Etiology Epidemiology
Hypersensitivity Pneumonitis: Epidemiology and Classification Ulrich Costabel, MD University of Duisburg-Essen, Ruhrlandklinik Department of Pneumology/Allergy Objectives Definitions, Etiology Epidemiology
Unit II Problem 2 Pathology: Pneumonia
 Unit II Problem 2 Pathology: Pneumonia - Definition: pneumonia is the infection of lung parenchyma which occurs especially when normal defenses are impaired such as: Cough reflex. Damage of cilia in respiratory
Unit II Problem 2 Pathology: Pneumonia - Definition: pneumonia is the infection of lung parenchyma which occurs especially when normal defenses are impaired such as: Cough reflex. Damage of cilia in respiratory
Imaging Small Airways Diseases: Not Just Air trapping. Eric J. Stern MD University of Washington
 Imaging Small Airways Diseases: Not Just Air trapping Eric J. Stern MD University of Washington What we are discussing SAD classification SAD imaging with MDCT emphasis What is a small airway? Airway with
Imaging Small Airways Diseases: Not Just Air trapping Eric J. Stern MD University of Washington What we are discussing SAD classification SAD imaging with MDCT emphasis What is a small airway? Airway with
Diagnosing ILD. What is important in 2016? Chris Grainge
 Diagnosing ILD What is important in 2016? Chris Grainge Senior Staff Specialist Respiratory Medicine John Hunter Hospital Conjoint A/Prof University of Newcastle Conflict of interest I have acted as a
Diagnosing ILD What is important in 2016? Chris Grainge Senior Staff Specialist Respiratory Medicine John Hunter Hospital Conjoint A/Prof University of Newcastle Conflict of interest I have acted as a
Idiopathic Pulmonary of Care
 Chapter 6.1 Living Medical etextbook A Digital Tool at the Point of Care From Projects In Knowledge Pulmonology Idiopathic Pulmonary Fibrosis @Point of Care IPF Case Study: Typical Presentation, Role of
Chapter 6.1 Living Medical etextbook A Digital Tool at the Point of Care From Projects In Knowledge Pulmonology Idiopathic Pulmonary Fibrosis @Point of Care IPF Case Study: Typical Presentation, Role of
Guidelines for Diagnosis and Treatment of IPF
 Guidelines for Diagnosis and Treatment of IPF Katerina Antoniou, MD, PhD Lecturer in Thoracic Medicine ERS ILD Group Secretary Medical School, University of Crete Classification of Interstitial Lung Disease
Guidelines for Diagnosis and Treatment of IPF Katerina Antoniou, MD, PhD Lecturer in Thoracic Medicine ERS ILD Group Secretary Medical School, University of Crete Classification of Interstitial Lung Disease
Electromagnetic navigational bronchoscopy in patients with solitary pulmonary nodules
 Original article Electromagnetic navigational bronchoscopy in patients with solitary pulmonary nodules Samuel Copeland MD, Shrinivas Kambali MD, Gilbert Berdine MD, Raed Alalawi MD Abstract Background:
Original article Electromagnetic navigational bronchoscopy in patients with solitary pulmonary nodules Samuel Copeland MD, Shrinivas Kambali MD, Gilbert Berdine MD, Raed Alalawi MD Abstract Background:
Monday 10 September Interstitial lung disease 15:10 15:35. The uncommon interstitial lung diseases (ILD)
 Interstitial lung disease 15:10 15:35 The uncommon interstitial lung diseases (ILD) Dr Grant Griffiths, Cwm Taf University Health Board, Cardiff Be familiar with the Diagnostic criteria for idiopathic
Interstitial lung disease 15:10 15:35 The uncommon interstitial lung diseases (ILD) Dr Grant Griffiths, Cwm Taf University Health Board, Cardiff Be familiar with the Diagnostic criteria for idiopathic
I have no relevant conflicts of interest to disclose
 I have no relevant conflicts of interest to disclose Diffuse parenchymal lung disease (DPLD) and its associations Secondary lobular anatomy DPLD History, clinical findings, temporal evolution, and exposures
I have no relevant conflicts of interest to disclose Diffuse parenchymal lung disease (DPLD) and its associations Secondary lobular anatomy DPLD History, clinical findings, temporal evolution, and exposures
Eosinophils and effusion: a clinical conundrum
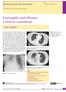 Ruth Sobala, Kevin Conroy, Hilary Tedd, Salem Elarbi kevin.peter.conroy@gmail.com Respiratory Dept, Queen Elizabeth Hospital, Gateshead, UK. Eosinophils and effusion: a clinical conundrum Case report A
Ruth Sobala, Kevin Conroy, Hilary Tedd, Salem Elarbi kevin.peter.conroy@gmail.com Respiratory Dept, Queen Elizabeth Hospital, Gateshead, UK. Eosinophils and effusion: a clinical conundrum Case report A
Influence of Smoking in Interstitial Pneumonia Presenting with a Non-Specific Interstitial Pneumonia Pattern
 ORIGINAL ARTICLE Influence of Smoking in Interstitial Pneumonia Presenting with a Non-Specific Interstitial Pneumonia Pattern Tetsuro Sawata, Masashi Bando, Masayuki Nakayama, Naoko Mato, Hideaki Yamasawa
ORIGINAL ARTICLE Influence of Smoking in Interstitial Pneumonia Presenting with a Non-Specific Interstitial Pneumonia Pattern Tetsuro Sawata, Masashi Bando, Masayuki Nakayama, Naoko Mato, Hideaki Yamasawa
