Breathtaking science. Developing respiratory drugs to improve health and quality of life
|
|
|
- Stephany Harvey
- 5 years ago
- Views:
Transcription
1 Breathtaking science Developing respiratory drugs to improve health and quality of life June 2017
2 Forward-Looking Statements This presentation contains forward looking statements that are based on the beliefs and assumptions and on information currently available to management of Verona Pharma plc (together with its consolidated subsidiaries, the Company ). All statements other than statements of historical fact contained in this presentation are forward-looking statements. Forward looking statements include information concerning the initiation, timing, progress and results of clinical trials of the Company s product candidate, the timing or likelihood of regulatory filings and approvals for any of its product candidates, and estimates regarding the Company s expenses, future revenues and future capital requirements. In some cases, you can identify forward-looking statements by terminology such as may, will, should, expects, plans, anticipates, believes, estimates, predicts, potential or continue or the negative of these terms or other comparable terminology. Forward-looking statements involve known and unknown risks, uncertainties and other factors that may cause the Company s actual results, performance or achievements to be materially different from any future results, performance or achievements expressed or implied by the forward-looking statements. These risks, uncertainties and other factors include those under risk Factors in the final prospectus filed with the Securities and Exchange Commission (the SEC ) on April 28, 2017 relating to the Company s Registration Statement on Form F-1 and in its other reports filed with the SEC. Forward-looking statements represent the Company s beliefs and assumptions only as of the date of this presentation. Although the Company believes that the expectations reflected in the forwardlooking statements are reasonable, it cannot guarantee future results, levels of activity, performance or achievements. Except as required by law, the Company assumes no obligation to publicly update any forward looking statements for any reason after the date of this presentation, to conform any of the forward-looking statements to actual results or to changes in its expectations. 2
3 Clinical-stage biopharma focused on developing & commercializing innovative therapeutics for treatment of respiratory diseases with significant unmet need Inhaled dual inhibitor of enzymes PDE3 and PDE4 RPL554 Current Focus: COPD and CF Potential first novel class of bronchodilator in decades Bronchodilator + anti-inflammatory agent in single compound 3
4 RPL554: Rich Product Pipeline Indication Delivery Pre- Clinical Phase 1 Phase 2 Phase 3 Addressable Market Opportunity COPD Maintenance (Home) COPD Acute (Hospital) Cystic Fibrosis Nebulizer COPD Maintenance (Home) Asthma Inhaler 4
5 Experienced Team Management Jan-Anders Karlsson, PhD Chief Executive Officer Piers Morgan, MA, ACA Chief Financial Officer Kenneth Newman, MD, MBA Chief Medical Officer Richard Hennings, BSc Commercial Director Peter Spargo, PhD SVP CMC Claire Poll, LLB Legal Counsel In Management and Board s prior lives involved in successful development / commercialization of many of the drugs used to treat COPD including 5
6 Financial Overview Listed on AiM (VRP.L) and NASDAQ (VRNA) Cash and Cash Equivalents (as of December 31, 2016) April 2017 NASDAQ IPO (including over-allotment option) Operating Expenses Market Cap (fully diluted) $51.1M 2 $90M NASDAQ IPO $9.5M 1 (12 Months Ended 12/31/16) $242M 2,3 ADS ratio 1:8 ordinary shares Exchange rates used (U.S. dollars per pound sterling): 1 Average for May 30, 2017 $ Share price and shares used: m shares fully diluted (16.1m ADSs); share price 150p ($15.40 / ADS) 6
7 Clear Success Drivers Large COPD patient population with significant unmet medical need and little treatment innovation in decades Significant Market opportunity Robust clinical data and validated endpoints FEV 1 in 8 P1 and P2a trials P2b planned 4 weeks; 400 subjects Clear, efficient pathway to Approval Significant potential demand from patients and physicians for new medications Strong potential for Adoption Broad application: pipeline in a product Global commercialization rights Significant outlook for Growth 7
8 COPD: Devastating Disease, Affecting Many CAUSES ALVEOLI BRONCHIOLE PROGRESSIVE DISEASE Smoking Air pollution No cure Inflames and narrows airways (bronchioles) and can damage alveoli Take a deep breath, blow out 20% now walk around, holding the rest forever. COPD Sufferer $$$ Sources: IMS, CDC Affects Many 3 rd leading cause of death 210M global sufferers 24M U.S. sufferers Very Costly Many exacerbations and hospitalizations ~$50B projected annual medical costs by 2020 (U.S. alone) $10B+ annual global sales of COPD drugs 8
9 COPD Sufferers Require Maintenance and Acute Treatment Maintenance (Home) Acute (Hospital) 1.5M ER visits U.S. alone: 24M living with COPD 15M diagnosed and under treatment Approximately 2M severe/very severe Treatment goals: Improved lung function Improved quality of life Prevent exacerbations Note: U.S. only data 20% Rehospitalized within 30 days of discharge 20% Die within 1 year of hospitalization 129K Deaths annually 687K Hospitalization s 9
10 Current Therapies: Little Innovation and Many Limitations Bronchodilators (First Line Treatment) Existing drug class New drug class Anti-Inflammatories Anti-Muscarinics SAMA (e.g. Ipratropium) LAMA (e.g. Spiriva) Beta2-Agonists SABA (e.g. Albuterol) LABA (e.g. Serevent) Inhaled CorticoSteroids ICS PDE4 Inhibitors PDE4 (e.g. Roflumilast) Bronchodilator Anti-Inflammatory Rapid Onset Black box warning - Well tolerated Not indicated as monotherapy Pneumonia Broad Patient Population Many patients treated with approved COPD drugs/combinations do not experience significant improvements in quality of life and continue to suffer from significant symptoms? 10
11 RPL554: Potential to Address Limitations of Current Therapies Bronchodilators (First Line Treatment) Existing drug class New drug class Anti-Inflammatories Anti-Muscarinics SAMA (e.g. Ipratropium) Beta2-Agonists RPL554 Bronchodilator Anti-Inflammatory Rapid Onset - Well tolerated* LAMA (e.g. Spiriva) SABA (e.g. Albuterol) LABA (e.g. Serevent) Black box warning Potentially Broad Patient Population*? Inhaled CorticoSteroids ICS Not indicated as monotherapy Pneumonia PDE4 Inhibitors PDE-4 (e.g. Roflumilast) *In clinical trials to date (RPL554 not yet approved) 11
12 RPL554 First-in-Class Candidate: Bronchodilator and Anti-inflammatory in a Single Compound Bronchodilation Anti-Inflammatory SAMA LAMA SABA LABA PDE3 ICS RPL554 Dual-enzyme inhibitor PDE4 Bronchodilator AND anti-inflammatory in a single compound Airway Smooth Muscle Inflammatory Cells Epithelial Cells Impacts 3 Key Mechanisms in Respiratory Disease: PDE3, PDE4 Relaxation Eosinophils PDE4 Neutrophils PDE4 Epithelial cells PDE3, PDE4 Lymphocytes PDE3, PDE4 Macrophages PDE3, PDE4 Fibroblasts PDE4 PDE3, PDE4 CFTR Activation Increased bronchodilation Increased anti-inflammatory effects Increased mucociliary clearance 12
13 Clear Success Drivers Large COPD patient population with significant unmet medical need and little treatment innovation in decades Significant Market opportunity Robust clinical data and validated endpoints FEV 1 from 8 P1 and P2a trials P2b planned 4 weeks; 400 subjects Clear, efficient pathway to Approval Significant potential demand from patients and physicians for new medications Strong potential for Adoption Broad application: pipeline in a product Global commercialization rights Significant outlook for Growth 13
14 RPL554: Significantly De-Risked Key Findings Phase 1 and 2a: 8 Clinical Trials, 282 Subjects RPL554 ALONE RPL554 IN ADDITION TO: Bronchodilation 51% greater Air Trapping either 14% Time of Onset of Effect Well tolerated Similar effect observed to albuterol or ipratropium alone albuterol reduction 20% more rapid ipratropium 66% greater 52% reduction 75% more rapid Improves FEV1 Improves residual lung volume Faster relief No dose-limiting adverse events * *in completed clinical trials 14
15 ml RPL554: Significantly Improves Lung Function in COPD Patients Peak Change from Baseline in FEV 1 (L) N= *** *** % % RPL554 & Albuterol Albuterol RPL554 & Ipratropium RPL554 Placebo Ipratropium Source: RPL p<0.001 vs placebo *** p<0.001 vs. albuterol or ipratropium alone 15
16 ml RPL554: Reduces Residual Lung Volume (Air Trapping) in COPD Patients Change from Baseline in Residual Volume at 1 Hour N= RPL554+ Albuterol RPL554+ Albuterol Ipratropium Ipratropium RPL554 Placebo ** 14% + 52% Reduction in Hyperinflation Is Typically Correlated with Improvement in Shortness of Breath Source: RPL p<0.001 vs. placebo ** p=0.016 vs. Albuterol alone at 1 hour timepoint *** p=0.001 vs. Ipratropium alone at 1 hour timepoint *** 16
17 RPL554: Faster Onset of Bronchodilation in COPD Patients Time to Onset (10% improvement in FEV 1 ) (mins) N=36 20 Mins 15 Mins 75% 4 Min ** 5 Min 5 Min *** RPL554+ Albuterol Albuterol RPL554+ Ipratropium Ipratropium RPL554 Reinforces Potential of RPL554 in Treating Acute Exacerbations of COPD Source: RPL ** p<0.01 vs. albuterol alone *** p<0.001 vs. ipratropium alone 17
18 Clear Success Drivers Large COPD patient population with significant unmet medical need and little treatment innovation in decades Significant Market opportunity Robust clinical data and validated endpoints FEV 1 from 8 P1 and P2a trials P2b planned 4 weeks; 400 subjects Clear, efficient pathway to Approval Significant potential demand from patients and physicians for new medications Strong potential for Adoption Broad application: pipeline in a product Global commercialization rights Significant outlook for Growth 18
19 RPL554: Potential to Improve Standard of Care Treatment for More Severe Patients Based on GOLD COPD Guidelines LAMA + LABA C RPL554 add-on Further exacerbation(s) LAMA LABA + ICS Consider Daliresp Further exacerbation(s) Further exacerbation(s) D LAMA + LABA + ICS LAMA + LABA RPL554 add-on More 2 Exacerbations/ Year 2 Bronchodilator LAMA + LABA RPL554 add-on 1 1 A long-acting bronchodilator (LABA or LAMA) Fewer A Symptoms B More Fewer 19
20 RPL554 Second Indication: Cystic Fibrosis (CF) Indication Delivery Pre- Clinical Phase 1 Phase 2 Phase 3 Addressable Market Opportunity COPD Maintenance (Home) COPD Acute (Hospital) Cystic Fibrosis Nebulizer COPD Maintenance (Home) Asthma Inhaler 20
21 CF: A Devastating Orphan Disease Population: Most common fatal inherited disease in U.S. Incidence: ~70K globally Cause: Mutations in gene that encodes CFTR protein Symptoms: Inability to clear thickened mucus, impaired lung function and persistent lung infection Consequences: Frequent exacerbations and hospitalization No cure Median age of death 37 years 21
22 RPL554 for CF: Pre-Clinical Studies Stimulation of CFTR Additive Effect When Administered with Kalydeco * p<0.05 ** p<0.01 *** p<0.001 * *** ** *** 10 8 * * p<0.01 vs. Kalydeco alone (3) ** Ion Current Increase in I SC After Fsk Prestimulation (µa cm -2 ) (17) (4) RPL554 (µm) 0 Kalydeco RPL554 RPL554 + Kalydeco Increase Stimulation of Ciliary Beat Frequency 15 DMSO roflumilast RPL554 % Increase in Ciliary Beat Frequency *** *** *** *** *** ** * p<0.05 vs. DMSO (control) measured 1, 5 and 30 minutes post dose *** p<0.001 vs. DMSO (control) measured 1, 5 and 30 minutes post dose -5 Time (Minutes) Post Dose Turner et al, Am J Physiol Lung Cell Mol Physiol 310: L59 L70,
23 RPL554: Potential to provide treatment independent of CF mutation status SAMA (e.g. Ipratropium) LAMA U.S. alone: (e.g. Spiriva) ~30K patients with CF RPL554 Anti-Inflammatory Treatment goals: Improved lung function Reduced exacerbations; antiinflammatory therapy Improved quality of life CFTR potentiator with different mechanism of action Bronchodilator Rapid Onset Well tolerated* *In clinical trials to date (RPL554 not yet approved) 23
24 2017 Achievements and Upcoming Milestones RPL554 addition to LAMA: study start U.S. IND open Listing on NASDAQ PK bioavailability: study start CF PK/PD Study: top-line data expected January March May 2H2017 February April June 1H2018 DPI and MDI commercial formulation: development start CF PK/PD study: study start Closing of IPO RPL554 4 week Ph2b: study start RPL554 addition to LAMA: top-line data expected PK bioavailability: top-line data expected 24
25 Breathtaking science Developing respiratory drugs to improve health and quality of life June 2017
Breathtaking science. Developing respiratory drugs to improve health and quality of life. H.C. Wainwright Global Life Sciences Conference April 2018
 Breathtaking science Developing respiratory drugs to improve health and quality of life H.C. Wainwright Global Life Sciences Conference April 2018 www.veronapharma.com Forward-Looking Statements This presentation
Breathtaking science Developing respiratory drugs to improve health and quality of life H.C. Wainwright Global Life Sciences Conference April 2018 www.veronapharma.com Forward-Looking Statements This presentation
Breathtaking science. Developing respiratory drugs to improve health and quality of life.
 Breathtaking science Developing respiratory drugs to improve health and quality of life www.veronapharma.com February 2018 Forward-Looking Statements This presentation contains forward looking statements
Breathtaking science Developing respiratory drugs to improve health and quality of life www.veronapharma.com February 2018 Forward-Looking Statements This presentation contains forward looking statements
Breathtaking science. Developing respiratory drugs to improve health and quality of life
 Breathtaking science Developing respiratory drugs to improve health and quality of life Wedbush PacGrow Healthcare Conference New York City, August 2018 NASDAQ VRNA www.veronapharma.com Forward-Looking
Breathtaking science Developing respiratory drugs to improve health and quality of life Wedbush PacGrow Healthcare Conference New York City, August 2018 NASDAQ VRNA www.veronapharma.com Forward-Looking
Verona Pharma Announces Positive Top-Line Data from Phase 2a Clinical Trial in COPD with RPL554 Dosed in Addition to Tiotropium (Spiriva )
 Press release 7 September 2017 Verona Pharma Announces Positive Top-Line Data from Phase 2a Clinical Trial in COPD with RPL554 Dosed in Addition to Tiotropium (Spiriva ) Achieved significant and clinically
Press release 7 September 2017 Verona Pharma Announces Positive Top-Line Data from Phase 2a Clinical Trial in COPD with RPL554 Dosed in Addition to Tiotropium (Spiriva ) Achieved significant and clinically
Revefenacin (TD-4208) Phase 3 Efficacy Results
 Revefenacin (TD-4208) Phase 3 Efficacy Results Once-daily, Nebulized Long-Acting Muscarinic Antagonist (LAMA) October 20, 2016 THERAVANCE, the Cross/Star logo and MEDICINES THAT MAKE A DIFFERENCE are registered
Revefenacin (TD-4208) Phase 3 Efficacy Results Once-daily, Nebulized Long-Acting Muscarinic Antagonist (LAMA) October 20, 2016 THERAVANCE, the Cross/Star logo and MEDICINES THAT MAKE A DIFFERENCE are registered
Targeted Therapeutics for Inflammatory Disease
 Targeted Therapeutics for Inflammatory Disease Forward Looking Statements / Safe Harbor This presentation and the accompanying oral commentary contain forward-looking statements that involve substantial
Targeted Therapeutics for Inflammatory Disease Forward Looking Statements / Safe Harbor This presentation and the accompanying oral commentary contain forward-looking statements that involve substantial
Verona Pharma plc AIM: VRP First-in-Class Drugs to Treat Unmet Needs in Respiratory Diseases
 Verona Pharma plc AIM: VRP First-in-Class Drugs to Treat Unmet Needs in Respiratory Diseases Presentation 28 April 2014 Legal disclaimer The information contained in this presentation is being supplied
Verona Pharma plc AIM: VRP First-in-Class Drugs to Treat Unmet Needs in Respiratory Diseases Presentation 28 April 2014 Legal disclaimer The information contained in this presentation is being supplied
Theravance Biopharma, Inc. (NASDAQ: TBPH)
 Theravance Biopharma, Inc. (NASDAQ: TBPH) FDA Approval of YUPELRI TM (revefenacin) Inhalation Solution November 9, 2018 THERAVANCE and the Cross/Star logo are registered trademarks of the Theravance Biopharma
Theravance Biopharma, Inc. (NASDAQ: TBPH) FDA Approval of YUPELRI TM (revefenacin) Inhalation Solution November 9, 2018 THERAVANCE and the Cross/Star logo are registered trademarks of the Theravance Biopharma
Aradigm Corporation. A respiratory specialty pharmaceutical company fulfilling unmet needs in pulmonary medicine (ARDM) February 2008
 Aradigm Corporation A respiratory specialty pharmaceutical company fulfilling unmet needs in pulmonary medicine (ARDM) February 2008 Safe Harbor Statement This presentation contains forward-looking statements
Aradigm Corporation A respiratory specialty pharmaceutical company fulfilling unmet needs in pulmonary medicine (ARDM) February 2008 Safe Harbor Statement This presentation contains forward-looking statements
COPD: A Renewed Focus. Disclosures
 COPD: A Renewed Focus Heath Latham, MD Assistant Professor Division of Pulmonary and Critical Care Medicine Disclosures No Business Interests No Consulting No Speakers Bureau No Off Label Use to Discuss
COPD: A Renewed Focus Heath Latham, MD Assistant Professor Division of Pulmonary and Critical Care Medicine Disclosures No Business Interests No Consulting No Speakers Bureau No Off Label Use to Discuss
SAVARA CORPORATE PRESENTATION (NASDAQ: SVRA) NOVEMBER 2018
 SAVARA CORPORATE PRESENTATION (NASDAQ: SVRA) NOVEMBER 2018 SAFE HARBOR STATEMENT Savara Inc. ( Savara or the Company ) cautions you that statements in this presentation that are not a description of historical
SAVARA CORPORATE PRESENTATION (NASDAQ: SVRA) NOVEMBER 2018 SAFE HARBOR STATEMENT Savara Inc. ( Savara or the Company ) cautions you that statements in this presentation that are not a description of historical
Update on Pulmonary Diseases. Jeffrey Lessar, MD
 Update on Pulmonary Diseases Jeffrey Lessar, MD 1 No disclosures to make No conflicts 2 Goals Update on key changes in Pulmonary Therapy 3 Spirometry Spirometry FEV1- forced expiratory Volume in 1 Sec
Update on Pulmonary Diseases Jeffrey Lessar, MD 1 No disclosures to make No conflicts 2 Goals Update on key changes in Pulmonary Therapy 3 Spirometry Spirometry FEV1- forced expiratory Volume in 1 Sec
Lead team presentation: Roflumilast for treating chronic obstructive pulmonary disease [ID984]
![Lead team presentation: Roflumilast for treating chronic obstructive pulmonary disease [ID984] Lead team presentation: Roflumilast for treating chronic obstructive pulmonary disease [ID984]](/thumbs/85/91889043.jpg) Lead team presentation: Roflumilast for treating chronic obstructive pulmonary disease [ID984] 1 st Appraisal Committee meeting Background & Clinical Effectiveness John McMurray 11 th January 2016 For
Lead team presentation: Roflumilast for treating chronic obstructive pulmonary disease [ID984] 1 st Appraisal Committee meeting Background & Clinical Effectiveness John McMurray 11 th January 2016 For
Corporate Overview June 2014 Jefferies Healthcare Conference NASDAQ: GLYC
 Corporate Overview June 2014 Jefferies Healthcare Conference NASDAQ: GLYC Forward-Looking Statements To the extent that statements contained in this presentation are not descriptions of historical facts
Corporate Overview June 2014 Jefferies Healthcare Conference NASDAQ: GLYC Forward-Looking Statements To the extent that statements contained in this presentation are not descriptions of historical facts
Anti-IL-33 (ANB020) Program
 Anti-IL-33 (ANB020) Program Phase 2a Peanut Allergy Clinical Trial Interim Data Update March 26 th 2018 NASDAQ: ANAB Safe Harbor Statement This presentation and the accompanying oral presentation contain
Anti-IL-33 (ANB020) Program Phase 2a Peanut Allergy Clinical Trial Interim Data Update March 26 th 2018 NASDAQ: ANAB Safe Harbor Statement This presentation and the accompanying oral presentation contain
ERS Investor & Analyst Event. Munich Tuesday 9 th September 2014
 ERS Investor & Analyst Event Munich Tuesday 9 th September 2014 Darrell Baker SVP, Global Head of Respiratory Agenda GSK s Respiratory Portfolio Eosinophils Research in COPD Eosinophils Clinical Experience
ERS Investor & Analyst Event Munich Tuesday 9 th September 2014 Darrell Baker SVP, Global Head of Respiratory Agenda GSK s Respiratory Portfolio Eosinophils Research in COPD Eosinophils Clinical Experience
AM-125 : Intranasal Betahistine
 AM-125 : Intranasal Betahistine February 3, 2017 NASDAQ: EARS Forward-looking Statements This presentation and the accompanying oral commentary contain forward-looking statements that involve substantial
AM-125 : Intranasal Betahistine February 3, 2017 NASDAQ: EARS Forward-looking Statements This presentation and the accompanying oral commentary contain forward-looking statements that involve substantial
27 th Annual J.P. Morgan Healthcare Conference. January 13, 2009
 27 th Annual J.P. Morgan Healthcare Conference January 13, 2009 1 Forward Looking Statements This presentation and any subsequent Q&A may contain forward-looking statements, including with respect to:»
27 th Annual J.P. Morgan Healthcare Conference January 13, 2009 1 Forward Looking Statements This presentation and any subsequent Q&A may contain forward-looking statements, including with respect to:»
PATENCY-1 Top-Line Results
 PATENCY-1 Top-Line Results December 13, 2016 2016 Proteon Therapeutics, Inc. Cautionary Note Regarding Forward-Looking Statements This presentation contains statements that are, or may be deemed to be,
PATENCY-1 Top-Line Results December 13, 2016 2016 Proteon Therapeutics, Inc. Cautionary Note Regarding Forward-Looking Statements This presentation contains statements that are, or may be deemed to be,
Engineering the Future for Inhaled Therapeutics. July 2016 NASDAQ: PULM
 Engineering the Future for Inhaled Therapeutics July 2016 NASDAQ: PULM Disclaimers & Forward Looking Statements This presentation contains forward-looking statements. Forward-looking statements may be
Engineering the Future for Inhaled Therapeutics July 2016 NASDAQ: PULM Disclaimers & Forward Looking Statements This presentation contains forward-looking statements. Forward-looking statements may be
Aquinox Q2 /2015 Conference Call: LEADERSHIP Secondary Endpoint Update - AQX-1125 in BPS/IC. August 6, 2015
 Aquinox Q2 /2015 Conference Call: LEADERSHIP Secondary Endpoint Update - AQX-1125 in BPS/IC August 6, 2015 Forward Looking Statements / Safe Harbor This presentation and the accompanying oral commentary
Aquinox Q2 /2015 Conference Call: LEADERSHIP Secondary Endpoint Update - AQX-1125 in BPS/IC August 6, 2015 Forward Looking Statements / Safe Harbor This presentation and the accompanying oral commentary
Targeted Therapeutics for Inflammatory Disease
 Targeted Therapeutics for Inflammatory Disease Jefferies Global Healthcare Conference June 2015 Forward Looking Statements / Safe Harbor This presentation and the accompanying oral commentary contain forward-looking
Targeted Therapeutics for Inflammatory Disease Jefferies Global Healthcare Conference June 2015 Forward Looking Statements / Safe Harbor This presentation and the accompanying oral commentary contain forward-looking
Targeted Therapeutics for Inflammatory Disease Jefferies Healthcare Conference
 Targeted Therapeutics for Inflammatory Disease 2016 Jefferies Healthcare Conference Forward Looking Statements / Safe Harbor This presentation and the accompanying oral commentary contain forward-looking
Targeted Therapeutics for Inflammatory Disease 2016 Jefferies Healthcare Conference Forward Looking Statements / Safe Harbor This presentation and the accompanying oral commentary contain forward-looking
Creating a Leading Global HBV Therapeutics Company. ARB-1467 Update Call December 12, 2016
 Creating a Leading Global HBV Therapeutics Company ARB-1467 Update Call December 12, 2016 NASDAQ: ABUS www.arbutusbio.com Forward Looking Statements This presentation contains forward-looking statements
Creating a Leading Global HBV Therapeutics Company ARB-1467 Update Call December 12, 2016 NASDAQ: ABUS www.arbutusbio.com Forward Looking Statements This presentation contains forward-looking statements
VERONA PHARMA PLC INTERIM REPORT FOR THE SIX MONTHS ENDED 30 JUNE 2014
 Company Number 05375156 INTERIM REPORT CONTENTS Page Contents 2 Directors, secretary and advisers 3 Corporate statement 4 Chairman and Chief Executive Officer s joint statement 58 Group statement of comprehensive
Company Number 05375156 INTERIM REPORT CONTENTS Page Contents 2 Directors, secretary and advisers 3 Corporate statement 4 Chairman and Chief Executive Officer s joint statement 58 Group statement of comprehensive
COPD: Current Medical Therapy
 COPD: Current Medical Therapy Angela Golden, DNP, FNP-C, FAANP Owner, NP from Home, LLC Outcomes As a result of this activity, learners will be able to: 1. List the appropriate classes of medications for
COPD: Current Medical Therapy Angela Golden, DNP, FNP-C, FAANP Owner, NP from Home, LLC Outcomes As a result of this activity, learners will be able to: 1. List the appropriate classes of medications for
Committed to Transforming the Treatment Paradigm for Migraine Prevention
 June 14, 2018 Committed to Transforming the Treatment Paradigm for Migraine Prevention September 6, 2018 Forward-Looking Statements This presentation and the accompanying commentary contains certain forward-looking
June 14, 2018 Committed to Transforming the Treatment Paradigm for Migraine Prevention September 6, 2018 Forward-Looking Statements This presentation and the accompanying commentary contains certain forward-looking
Defining COPD. Georgina Grantham Community Respiratory Team Leader/ Respiratory Nurse Specialist
 Defining COPD Georgina Grantham Community Respiratory Team Leader/ Respiratory Nurse Specialist Defining COPD Chronic Obstructive Pulmonary Disease (COPD) is a common, preventable and treatable disease
Defining COPD Georgina Grantham Community Respiratory Team Leader/ Respiratory Nurse Specialist Defining COPD Chronic Obstructive Pulmonary Disease (COPD) is a common, preventable and treatable disease
Treatment. Assessing the outcome of interventions Traditionally, the effects of interventions have been assessed by measuring changes in the FEV 1
 58 COPD 59 The treatment of COPD includes drug therapy, surgery, exercise and counselling/psychological support. When managing COPD patients, it is particularly important to evaluate the social and family
58 COPD 59 The treatment of COPD includes drug therapy, surgery, exercise and counselling/psychological support. When managing COPD patients, it is particularly important to evaluate the social and family
aclidinium 322 micrograms inhalation powder (Eklira Genuair ) SMC No. (810/12) Almirall S.A.
 aclidinium 322 micrograms inhalation powder (Eklira Genuair ) SMC No. (810/12) Almirall S.A. 05 October 2012 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product and
aclidinium 322 micrograms inhalation powder (Eklira Genuair ) SMC No. (810/12) Almirall S.A. 05 October 2012 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product and
PROMISE 1 Top-Line Data Results. June 27, 2017
 PROMISE 1 Top-Line Data Results dd June 27, 2017 Forward Looking Statements This presentation and the accompanying commentary contains certain forward-looking statements within the meaning of Section 27A
PROMISE 1 Top-Line Data Results dd June 27, 2017 Forward Looking Statements This presentation and the accompanying commentary contains certain forward-looking statements within the meaning of Section 27A
Merrill Lynch Pharmaceutical, Biotechnology & Medical Device Conference New York, February 4, 2004
 Merrill Lynch Pharmaceutical, Biotechnology & Medical Device Conference New York, February 4, 2004 Disclaimer 2 This presentation contains forward-looking statements, i.e., current estimates or expectations
Merrill Lynch Pharmaceutical, Biotechnology & Medical Device Conference New York, February 4, 2004 Disclaimer 2 This presentation contains forward-looking statements, i.e., current estimates or expectations
Forward Looking Statements
 Therapeutic products for respiratory diseases October 2011 Forward Looking Statements This presentation may contain forward-looking statements that are based on management s current expectations and beliefs
Therapeutic products for respiratory diseases October 2011 Forward Looking Statements This presentation may contain forward-looking statements that are based on management s current expectations and beliefs
Three s Company - The role of triple therapy in chronic obstructive pulmonary disease (COPD)
 Three s Company - The role of triple therapy in chronic obstructive pulmonary disease (COPD) Zahava Picado, PharmD PGY1 Pharmacy Practice Resident Central Texas Veterans Healthcare System Temple, TX October
Three s Company - The role of triple therapy in chronic obstructive pulmonary disease (COPD) Zahava Picado, PharmD PGY1 Pharmacy Practice Resident Central Texas Veterans Healthcare System Temple, TX October
Inarigivir ACHIEVE Trial Results and HBV Clinical Program Update. August 2, 2018
 Inarigivir ACHIEVE Trial Results and HBV Clinical Program Update August 2, 2018 FORWARD LOOKING STATEMENT This presentation includes forward-looking statements within the meaning of the Private Securities
Inarigivir ACHIEVE Trial Results and HBV Clinical Program Update August 2, 2018 FORWARD LOOKING STATEMENT This presentation includes forward-looking statements within the meaning of the Private Securities
NYSE AMER: MTNB. MAT9001 OVERVIEW. September 2018
 NYSE AMER: MTNB www.matinasbiopharma.com MAT9001 OVERVIEW September 2018 1 Forward Looking Statement This presentation contains "forward-looking statements" within the meaning of the Private Securities
NYSE AMER: MTNB www.matinasbiopharma.com MAT9001 OVERVIEW September 2018 1 Forward Looking Statement This presentation contains "forward-looking statements" within the meaning of the Private Securities
Treatment Responses. Ronald Dahl, Aarhus University Hospital, Denmark
 Asthma and COPD: Are They a Spectrum Treatment Responses Ronald Dahl, Aarhus University Hospital, Denmark Pharmacological Treatments Bronchodilators Inhaled short-acting β -Agonist (rescue) Inhaled short-acting
Asthma and COPD: Are They a Spectrum Treatment Responses Ronald Dahl, Aarhus University Hospital, Denmark Pharmacological Treatments Bronchodilators Inhaled short-acting β -Agonist (rescue) Inhaled short-acting
COPD: GOLD guidelines Ijlal Babar, MD Medical Director Pulmonary CCM, Pulmonary Hypertension Center SRHS
 COPD: GOLD guidelines 2017 Ijlal Babar, MD Medical Director Pulmonary CCM, Pulmonary Hypertension Center SRHS Introduction The Global Initiative for Chronic Obstructive Lung Disease (GOLD) program was
COPD: GOLD guidelines 2017 Ijlal Babar, MD Medical Director Pulmonary CCM, Pulmonary Hypertension Center SRHS Introduction The Global Initiative for Chronic Obstructive Lung Disease (GOLD) program was
Santhera Pharmaceuticals Company Presentation. September 2018
 Santhera Pharmaceuticals Company Presentation September 2018 Disclaimer This presentation is not and under no circumstances to be construed as a solicitation, offer, or recommendation, to buy or sell securities
Santhera Pharmaceuticals Company Presentation September 2018 Disclaimer This presentation is not and under no circumstances to be construed as a solicitation, offer, or recommendation, to buy or sell securities
We strive to develop innovative solutions that improve and extend the lives of people with cystic fibrosis
 Nivalis Therapeutics, Inc., October 2015 Analyst Breakfast at NACFC We strive to develop innovative solutions that improve and extend the lives of people with cystic fibrosis Disclaimer Regarding Forward
Nivalis Therapeutics, Inc., October 2015 Analyst Breakfast at NACFC We strive to develop innovative solutions that improve and extend the lives of people with cystic fibrosis Disclaimer Regarding Forward
Forward Looking Statements
 Therapeutic products for respiratory diseases September 2008 Forward Looking Statements This presentation may contain forward-looking statements that are based on management s current expectations and
Therapeutic products for respiratory diseases September 2008 Forward Looking Statements This presentation may contain forward-looking statements that are based on management s current expectations and
Q1 Results 2018 Webcast presentation 26 April 2018
 Q1 Results 2018 Webcast presentation 26 April 2018 Disclaimer This presentation contains forward-looking statements, including (without limitation) statements concerning the progress of our clinical pipeline,
Q1 Results 2018 Webcast presentation 26 April 2018 Disclaimer This presentation contains forward-looking statements, including (without limitation) statements concerning the progress of our clinical pipeline,
Aviragen Therapeutics, Inc. and Vaxart Inc. Joint Conference Call UNLOCKING THE FULL POTENTIAL OF ORAL VACCINES
 Aviragen Therapeutics, Inc. and Vaxart Inc. Joint Conference Call UNLOCKING THE FULL POTENTIAL OF ORAL VACCINES October 30, 2017 Safe Harbor This presentation contains forward-looking statements about
Aviragen Therapeutics, Inc. and Vaxart Inc. Joint Conference Call UNLOCKING THE FULL POTENTIAL OF ORAL VACCINES October 30, 2017 Safe Harbor This presentation contains forward-looking statements about
Air Flow Limitation. In most serious respiratory disease, a key feature causing morbidity and functional disruption is air flow imitation.
 Asthma Air Flow Limitation In most serious respiratory disease, a key feature causing morbidity and functional disruption is air flow imitation. True whether reversible, asthma and exercise-induced bronchospasm,
Asthma Air Flow Limitation In most serious respiratory disease, a key feature causing morbidity and functional disruption is air flow imitation. True whether reversible, asthma and exercise-induced bronchospasm,
Capricor Therapeutics
 Therapeutics Conference Call to Discuss the HOPE-2 Clinical Trial NASDAQ: CAPR November 29, 2017 Forward-Looking Statements Statements in this presentation regarding the efficacy, safety, and intended
Therapeutics Conference Call to Discuss the HOPE-2 Clinical Trial NASDAQ: CAPR November 29, 2017 Forward-Looking Statements Statements in this presentation regarding the efficacy, safety, and intended
Potential risks of ICS use
 Potential risks of ICS use Randomised controlled trial Observational study Systematic review Pneumonia Tuberculosis Bone fracture Skin thinning/easy bruising Cataract Diabetes No effect on fracture risk
Potential risks of ICS use Randomised controlled trial Observational study Systematic review Pneumonia Tuberculosis Bone fracture Skin thinning/easy bruising Cataract Diabetes No effect on fracture risk
MANAGING ASTHMA. Nancy Davis, RRT, AE-C
 MANAGING ASTHMA Nancy Davis, RRT, AE-C What is asthma? Asthma is a chronic respiratory disease characterized by episodes or attacks of inflammation and narrowing of small airways in response to asthma
MANAGING ASTHMA Nancy Davis, RRT, AE-C What is asthma? Asthma is a chronic respiratory disease characterized by episodes or attacks of inflammation and narrowing of small airways in response to asthma
BUDESONIDE AND FORMOTEROL (SYMBICORT ): Α A REVIEW
 Volume 23, Issue 3 December 2007 BUDESONIDE AND FORMOTEROL (SYMBICORT ): A REVIEW Donna L. Smith, Pharm. D. Candidate More than 22 million people in the United States have asthma according to the Centers
Volume 23, Issue 3 December 2007 BUDESONIDE AND FORMOTEROL (SYMBICORT ): A REVIEW Donna L. Smith, Pharm. D. Candidate More than 22 million people in the United States have asthma according to the Centers
N a s d a q : I N S Y
 N a s d a q : I N S Y 0 Safe Harbor Statement This presentation contains both historical information and forward-looking statements. Forward-looking statements are based on management's current expectations
N a s d a q : I N S Y 0 Safe Harbor Statement This presentation contains both historical information and forward-looking statements. Forward-looking statements are based on management's current expectations
Committed to the research, development and commercialisation of human therapeutic products for chronic respiratory and autoimmune diseases
 ABN 75 082 811 630 Committed to the research, development and commercialisation of human therapeutic products for chronic respiratory and autoimmune diseases November 2003 Overview Our Objective to build
ABN 75 082 811 630 Committed to the research, development and commercialisation of human therapeutic products for chronic respiratory and autoimmune diseases November 2003 Overview Our Objective to build
COPD Update. Plus New and Improved Products for Inhaled Therapy. Catherine Bourg Rebitch, PharmD, BCACP Clinical Associate Professor
 COPD Update Plus New and Improved Products for Inhaled Therapy Catherine Bourg Rebitch, PharmD, BCACP Clinical Associate Professor Disclosure The presenter has nothing to disclose concerning possible financial
COPD Update Plus New and Improved Products for Inhaled Therapy Catherine Bourg Rebitch, PharmD, BCACP Clinical Associate Professor Disclosure The presenter has nothing to disclose concerning possible financial
Putting NICE guidance into practice. Resource impact report: Asthma: diagnosis, monitoring and chronic asthma management (NG80)
 Putting NICE guidance into practice Resource impact report: Asthma: diagnosis, monitoring and chronic asthma management (NG80) Published: November 2017 Summary This report focuses on the recommendations
Putting NICE guidance into practice Resource impact report: Asthma: diagnosis, monitoring and chronic asthma management (NG80) Published: November 2017 Summary This report focuses on the recommendations
Innovative products for r espiratory rrespiratory d iseases diseases April 2012
 Innovative products for respiratory diseases April 2012 Pharmaxis - company overview Objectives Employees Development of products for respiratory diseases 153 involved in research and development, clinical
Innovative products for respiratory diseases April 2012 Pharmaxis - company overview Objectives Employees Development of products for respiratory diseases 153 involved in research and development, clinical
COPD: Treatment Update Property of Presenter. Not for Reproduction. Barry Make, MD Professor of Medicine National Jewish Health
 COPD: Treatment Update Barry Make, MD Professor of Medicine National Jewish Health Disclosures Advisory board, consultant, multi-center trial, research funding, Data Safety Monitoring Board (DSMB), or
COPD: Treatment Update Barry Make, MD Professor of Medicine National Jewish Health Disclosures Advisory board, consultant, multi-center trial, research funding, Data Safety Monitoring Board (DSMB), or
CHRONIC OBSTRUCTIVE PULMONARY DISEASE
 CHRONIC OBSTRUCTIVE PULMONARY DISEASE INCIDENCE UP TO 380,000 PEOPLE IN IRELAND HSE FIGURES 110,000 DIAGNOSED AND 200,000 UNDIAGNOSED. AFFECTS MORE MEN THAN WOMEN BUT RATES ARE RISING 1500 DEATHS PER YEAR
CHRONIC OBSTRUCTIVE PULMONARY DISEASE INCIDENCE UP TO 380,000 PEOPLE IN IRELAND HSE FIGURES 110,000 DIAGNOSED AND 200,000 UNDIAGNOSED. AFFECTS MORE MEN THAN WOMEN BUT RATES ARE RISING 1500 DEATHS PER YEAR
37 th Annual J.P. Morgan Healthcare Conference. January 9, 2019
 37 th Annual J.P. Morgan Healthcare Conference January 9, 2019 Forward Looking Statement This presentation includes forward-looking statements. All statements, other than statements of historical facts,
37 th Annual J.P. Morgan Healthcare Conference January 9, 2019 Forward Looking Statement This presentation includes forward-looking statements. All statements, other than statements of historical facts,
Therapeutic products. for. respiratory and autoimmune diseases
 Therapeutic products for respiratory and autoimmune diseases Annual General Meeting November 2005 Highlights Bronchitol Aridol Autoimmune disease Manufacturing Bronchitol: Entering Phase III Successful
Therapeutic products for respiratory and autoimmune diseases Annual General Meeting November 2005 Highlights Bronchitol Aridol Autoimmune disease Manufacturing Bronchitol: Entering Phase III Successful
ObsEva Reports Third Quarter 2017 Financial Results and Provides Business Update
 ObsEva Reports Third Quarter 2017 Financial Results and Provides Business Update - All Three Development Compounds Progressing with Key Clinical Milestones Over the Next 12 Months - Geneva, Switzerland
ObsEva Reports Third Quarter 2017 Financial Results and Provides Business Update - All Three Development Compounds Progressing with Key Clinical Milestones Over the Next 12 Months - Geneva, Switzerland
Summary A pharmaceutical company which develops therapeutic products for
 Innovative products for respiratory diseases Annual General Meeting October 2012 Pharmaxis - company overview Summary A pharmaceutical company which develops therapeutic products for human chronic respiratory
Innovative products for respiratory diseases Annual General Meeting October 2012 Pharmaxis - company overview Summary A pharmaceutical company which develops therapeutic products for human chronic respiratory
Medicines that make a difference
 Medicines that make a difference 11 th Annual Needham Healthcare Conference Mathai Mammen, M.D., Ph.D. Senior Vice President, Research and Early Clinical Development April 4, 2012 Theravance, Theravance
Medicines that make a difference 11 th Annual Needham Healthcare Conference Mathai Mammen, M.D., Ph.D. Senior Vice President, Research and Early Clinical Development April 4, 2012 Theravance, Theravance
Calliditas Therapeutics Q2 Report Webcast August 16, 2018, 10:00 Presenters: Renée Aguiar-Lucander, CEO Fredrik Johansson, CFO
 Calliditas Therapeutics Q2 Report 2018 Webcast August 16, 2018, 10:00 Presenters: Renée Aguiar-Lucander, CEO Fredrik Johansson, CFO Disclaimer Important information This presentation may contain certain
Calliditas Therapeutics Q2 Report 2018 Webcast August 16, 2018, 10:00 Presenters: Renée Aguiar-Lucander, CEO Fredrik Johansson, CFO Disclaimer Important information This presentation may contain certain
Up in FLAMES: Stable Chronic Obstructive Pulmonary Disease (COPD) Management. Colleen Sakon, PharmD BCPS September 27, 2018
 Up in FLAMES: Stable Chronic Obstructive Pulmonary Disease (COPD) Management Colleen Sakon, PharmD BCPS September 27, 2018 Disclosures I have no actual or potential conflicts of interest 2 Objectives Summarize
Up in FLAMES: Stable Chronic Obstructive Pulmonary Disease (COPD) Management Colleen Sakon, PharmD BCPS September 27, 2018 Disclosures I have no actual or potential conflicts of interest 2 Objectives Summarize
Changing Landscapes in COPD New Zealand Respiratory Conference
 Changing Landscapes in COPD New Zealand Respiratory Conference Dr Robert Young BMedSc MBChB DPhil (Oxon) FRACP FRCP Associate Professor Consultant Physician Changing Landscapes in COPD: Summary 1. Overview
Changing Landscapes in COPD New Zealand Respiratory Conference Dr Robert Young BMedSc MBChB DPhil (Oxon) FRACP FRCP Associate Professor Consultant Physician Changing Landscapes in COPD: Summary 1. Overview
Optum Research Database Proportion of asthma patients with prescription fills for rescue and controller medications by year,
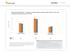 ASTHMA CHART 1 Optum Research Database Proportion of asthma patients with prescription fills for rescue and controller medications by year, 215-216 7 6 5 4 3 2 1 1 Controller medication 1 Rescue medication
ASTHMA CHART 1 Optum Research Database Proportion of asthma patients with prescription fills for rescue and controller medications by year, 215-216 7 6 5 4 3 2 1 1 Controller medication 1 Rescue medication
Surveillance report Published: 6 April 2016 nice.org.uk. NICE All rights reserved.
 Surveillance report 2016 Chronic obstructive pulmonary disease in over 16s: diagnosis and management (2010) NICE guideline CG101 Surveillance report Published: 6 April 2016 nice.org.uk NICE 2016. All rights
Surveillance report 2016 Chronic obstructive pulmonary disease in over 16s: diagnosis and management (2010) NICE guideline CG101 Surveillance report Published: 6 April 2016 nice.org.uk NICE 2016. All rights
Roflumilast (Daxas) for chronic obstructive pulmonary disease
 Roflumilast (Daxas) for chronic obstructive pulmonary disease August 2009 This technology summary is based on information available at the time of research and a limited literature search. It is not intended
Roflumilast (Daxas) for chronic obstructive pulmonary disease August 2009 This technology summary is based on information available at the time of research and a limited literature search. It is not intended
Presented by UIC College of Nursing
 Presented by UIC College of Nursing Describe COPD. Identify red flags for a COPD exacerbation. Identify COPD triggers or risk factors. Differentiate between long-acting inhalers and emergency use inhalers.
Presented by UIC College of Nursing Describe COPD. Identify red flags for a COPD exacerbation. Identify COPD triggers or risk factors. Differentiate between long-acting inhalers and emergency use inhalers.
Chronic Obstructive Pulmonary Disease 1/18/2018
 Presented by UIC College of Nursing Describe COPD. Identify red flags for a COPD exacerbation. Identify COPD triggers or risk factors. Differentiate between long acting inhalers and emergency use inhalers.
Presented by UIC College of Nursing Describe COPD. Identify red flags for a COPD exacerbation. Identify COPD triggers or risk factors. Differentiate between long acting inhalers and emergency use inhalers.
Dynavax Corporate Presentation
 Dynavax Corporate Presentation Forward-Looking Statements This presentation contains forward-looking statements, including statements regarding our HEPLISAV-B TM regulatory submissions, product profile,
Dynavax Corporate Presentation Forward-Looking Statements This presentation contains forward-looking statements, including statements regarding our HEPLISAV-B TM regulatory submissions, product profile,
CHRONIC OBSTRUCTIVE PULMONARY DISEASE (COPD) TREATMENT GUIDELINES
 CHRONIC OBSTRUCTIVE PULMONARY DISEASE (COPD) TREATMENT GUIDELINES Document Description Document Type Service Application Version Guidelines All healthcare professionals(hcps) caring for patients with asthma
CHRONIC OBSTRUCTIVE PULMONARY DISEASE (COPD) TREATMENT GUIDELINES Document Description Document Type Service Application Version Guidelines All healthcare professionals(hcps) caring for patients with asthma
Supplementary appendix
 Supplementary appendix This appendix formed part of the original submission and has been peer reviewed. We post it as supplied by the authors. Supplement to: Calverley P M A, Anzueto A R, Carter K, et
Supplementary appendix This appendix formed part of the original submission and has been peer reviewed. We post it as supplied by the authors. Supplement to: Calverley P M A, Anzueto A R, Carter K, et
Targeted Therapeutics for Inflammatory Disease. Investor Presentation April 2014
 Targeted Therapeutics for Inflammatory Disease Investor Presentation April 2014 Forward Looking Statements / Safe Harbor This presentation and the accompanying oral commentary contain forward-looking statements
Targeted Therapeutics for Inflammatory Disease Investor Presentation April 2014 Forward Looking Statements / Safe Harbor This presentation and the accompanying oral commentary contain forward-looking statements
LJPC-401 Phase 1 Results and Development Update. September 7, 2016
 LJPC-401 Phase 1 Results and Development Update September 7, 2016 Forward-Looking Statements These slides contain "forward-looking" statements within the meaning of the Private Securities Litigation Reform
LJPC-401 Phase 1 Results and Development Update September 7, 2016 Forward-Looking Statements These slides contain "forward-looking" statements within the meaning of the Private Securities Litigation Reform
Verona Pharma plc. First-in-Class Drugs to Treat Unmet Needs in Respiratory Diseases A UK-based drug development company. Preliminary Results 2012
 Verona Pharma plc First-in-Class Drugs to Treat Unmet Needs in Respiratory Diseases A UK-based drug development company Preliminary Results 2012 10 th April 2013 Legal Disclaimer The information contained
Verona Pharma plc First-in-Class Drugs to Treat Unmet Needs in Respiratory Diseases A UK-based drug development company Preliminary Results 2012 10 th April 2013 Legal Disclaimer The information contained
New Ideas. Better Medicines. Third Quarter Financial Results Conference Call
 New Ideas. Better Medicines. Third Quarter 2018 Financial Results Conference Call Forward-Looking Statements 2 This presentation contains forward-looking statements that involve substantial risks and uncertainties.
New Ideas. Better Medicines. Third Quarter 2018 Financial Results Conference Call Forward-Looking Statements 2 This presentation contains forward-looking statements that involve substantial risks and uncertainties.
Chronic Obstructive Pulmonary Disease (COPD) Treatment Guidelines
 Chronic Obstructive Pulmonary Disease (COPD) Treatment Guidelines Where appropriate the following should be offered before commencing inhaled treatment: Offer treatment and support to stop smoking. Smoking
Chronic Obstructive Pulmonary Disease (COPD) Treatment Guidelines Where appropriate the following should be offered before commencing inhaled treatment: Offer treatment and support to stop smoking. Smoking
Committed to Transforming the Treatment Paradigm for Migraine Prevention
 Committed to Transforming the Treatment Paradigm for Migraine Prevention 36th Annual J.P. Morgan Healthcare Conference January 8, 2018 Forward-Looking Statements This presentation and the accompanying
Committed to Transforming the Treatment Paradigm for Migraine Prevention 36th Annual J.P. Morgan Healthcare Conference January 8, 2018 Forward-Looking Statements This presentation and the accompanying
May 10, 2016 Q & Business Update
 May 10, 2016 Q1 2016 Thank Financial you Results & Business Update Q1 2016 Financial Results Prepared Remarks Q1 Update Tom Hughes, Ph.D., Chief Executive Officer Clinical Update Dennis Kim, M.D., Chief
May 10, 2016 Q1 2016 Thank Financial you Results & Business Update Q1 2016 Financial Results Prepared Remarks Q1 Update Tom Hughes, Ph.D., Chief Executive Officer Clinical Update Dennis Kim, M.D., Chief
Provider Respiratory Inservice
 Provider Respiratory Inservice 2 Welcome Opening Remarks We will cover: Definition of Asthma & COPD Evidence based guidelines for diagnosis, evaluation, and management of asthma Evidence based guidelines
Provider Respiratory Inservice 2 Welcome Opening Remarks We will cover: Definition of Asthma & COPD Evidence based guidelines for diagnosis, evaluation, and management of asthma Evidence based guidelines
REATA PHARMACEUTICALS, INC. ANNOUNCES SECOND QUARTER 2018 FINANCIAL RESULTS AND AN UPDATE ON DEVELOPMENT PROGRAMS
 REATA PHARMACEUTICALS, INC. ANNOUNCES SECOND QUARTER 2018 FINANCIAL RESULTS AND AN UPDATE ON DEVELOPMENT PROGRAMS IRVING, Texas August 8, 2018 Reata Pharmaceuticals, Inc. (Nasdaq: RETA), a clinical-stage
REATA PHARMACEUTICALS, INC. ANNOUNCES SECOND QUARTER 2018 FINANCIAL RESULTS AND AN UPDATE ON DEVELOPMENT PROGRAMS IRVING, Texas August 8, 2018 Reata Pharmaceuticals, Inc. (Nasdaq: RETA), a clinical-stage
AGM Presentation For the year to 30 September February 2016
 AGM Presentation For the year to 30 September 2015 25 February 2016 11 Forward Looking Statements This presentation includes forward-looking statements. These forward-looking statements involve known and
AGM Presentation For the year to 30 September 2015 25 February 2016 11 Forward Looking Statements This presentation includes forward-looking statements. These forward-looking statements involve known and
David Zaccardelli, PharmD Chief Executive Officer. J.P. Morgan 37 th Annual Healthcare Conference January 8, 2019
 David Zaccardelli, PharmD Chief Executive Officer J.P. Morgan 37 th Annual Healthcare Conference January 8, 2019 Disclaimer Certain information contained in this presentation relates to or is based on
David Zaccardelli, PharmD Chief Executive Officer J.P. Morgan 37 th Annual Healthcare Conference January 8, 2019 Disclaimer Certain information contained in this presentation relates to or is based on
GW Pharmaceuticals plc. Investor Presentation August 2014
 GW Pharmaceuticals plc Investor Presentation August 2014 Forward Looking Statements and Disclaimer This presentation contains forward-looking statements. Some of the matters discussed concerning our operations
GW Pharmaceuticals plc Investor Presentation August 2014 Forward Looking Statements and Disclaimer This presentation contains forward-looking statements. Some of the matters discussed concerning our operations
Determined to realize a future in which people with cancer live longer and better than ever before
 Determined to realize a future in which people with cancer live longer and better than ever before 4Q 2016 EARNINGS PRESENTATION MARCH 2017 1 Forward-looking statements disclosure This presentation contains
Determined to realize a future in which people with cancer live longer and better than ever before 4Q 2016 EARNINGS PRESENTATION MARCH 2017 1 Forward-looking statements disclosure This presentation contains
Investor Presentation
 Investor Presentation June 2017 Joe Turgeon President and Chief Operating Officer Safe Harbor Statement This presentation contains forward-looking statements regarding future events and the future performance
Investor Presentation June 2017 Joe Turgeon President and Chief Operating Officer Safe Harbor Statement This presentation contains forward-looking statements regarding future events and the future performance
Bayer and Regeneron start additional Phase 3 Study for VEGF Trap-Eye in Wet Age-related Macular Degeneration
 Investor News Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com Bayer and Regeneron start additional Phase 3 Study for VEGF Trap-Eye in Wet Age-related Macular Degeneration International
Investor News Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com Bayer and Regeneron start additional Phase 3 Study for VEGF Trap-Eye in Wet Age-related Macular Degeneration International
ASTHMA-COPD OVERLAP SYNDROME 2018: What s All the Fuss?
 ASTHMA-COPD OVERLAP SYNDROME 2018: What s All the Fuss? Randall W. Brown, MD MPH AE-C Association of Asthma Educators Annual Conference July 20, 2018 Phoenix, Arizona FACULTY/DISCLOSURES Randall Brown,
ASTHMA-COPD OVERLAP SYNDROME 2018: What s All the Fuss? Randall W. Brown, MD MPH AE-C Association of Asthma Educators Annual Conference July 20, 2018 Phoenix, Arizona FACULTY/DISCLOSURES Randall Brown,
ObsEva Reports First Quarter 2018 Financial Results and Provides Business Update
 ObsEva Reports First Quarter 2018 Financial Results and Provides Business Update -Key First Quarter 2018 Clinical Milestones Achieved Patient randomization completed in Phase 2b EDELWEISS trial of OBE2109
ObsEva Reports First Quarter 2018 Financial Results and Provides Business Update -Key First Quarter 2018 Clinical Milestones Achieved Patient randomization completed in Phase 2b EDELWEISS trial of OBE2109
The FDA Critical Path Initiative
 The FDA Critical Path Initiative Clinical Considerations for Demonstration of Dose-response for Inhaled Corticosteroids - Exhaled Nitric Oxide Model Badrul A. Chowdhury, MD, PhD Director Division of Pulmonary
The FDA Critical Path Initiative Clinical Considerations for Demonstration of Dose-response for Inhaled Corticosteroids - Exhaled Nitric Oxide Model Badrul A. Chowdhury, MD, PhD Director Division of Pulmonary
Respiratory Health. Asthma and COPD
 Respiratory Health Asthma and COPD Definition of asthma Working definition by AAH 2014: Chronic lung disease Can be controlled not cured Large variation in lung function Large variation in respiratory
Respiratory Health Asthma and COPD Definition of asthma Working definition by AAH 2014: Chronic lung disease Can be controlled not cured Large variation in lung function Large variation in respiratory
GINA. At-A-Glance Asthma Management Reference. for adults, adolescents and children 6 11 years. Updated 2017
 GINA At-A-Glance Asthma Management Reference for adults, adolescents and children 6 11 years Updated 2017 This resource should be used in conjunction with the Global Strategy for Asthma Management and
GINA At-A-Glance Asthma Management Reference for adults, adolescents and children 6 11 years Updated 2017 This resource should be used in conjunction with the Global Strategy for Asthma Management and
Merck Announces FDA Approval of KEYTRUDA. Provided to Investors as a Reference
 Merck Announces FDA Approval of KEYTRUDA Provided to Investors as a Reference Forward-Looking Statement This presentation includes forward-looking statements within the meaning of the safe harbor provisions
Merck Announces FDA Approval of KEYTRUDA Provided to Investors as a Reference Forward-Looking Statement This presentation includes forward-looking statements within the meaning of the safe harbor provisions
Q3 18 Earnings Supplemental Slides
 (Nasdaq: INSY) Q3 18 Earnings Supplemental Slides November 5, 2018 Safe-Harbor Statement This presentation contains both historical information and forward-looking statements. Forward-looking statements
(Nasdaq: INSY) Q3 18 Earnings Supplemental Slides November 5, 2018 Safe-Harbor Statement This presentation contains both historical information and forward-looking statements. Forward-looking statements
(Asthma) Diagnosis, monitoring and chronic asthma management
 Dubai Standards of Care 2018 (Asthma) Diagnosis, monitoring and chronic asthma management Preface Asthma is one of the most common problem dealt with in daily practice. In Dubai, the management of chronic
Dubai Standards of Care 2018 (Asthma) Diagnosis, monitoring and chronic asthma management Preface Asthma is one of the most common problem dealt with in daily practice. In Dubai, the management of chronic
AVENUE THERAPEUTICS, INC. NASDAQ: ATXI JANUARY 2018
 AVENUE THERAPEUTICS, INC. NASDAQ: ATXI JANUARY 2018 Forward Looking Statements Statements in this presentation that are not descriptions of historical facts are forward-looking statements within the meaning
AVENUE THERAPEUTICS, INC. NASDAQ: ATXI JANUARY 2018 Forward Looking Statements Statements in this presentation that are not descriptions of historical facts are forward-looking statements within the meaning
Therapeutic products for respiratory and autoimmune diseases. Annual General Meeting
 Therapeutic products for respiratory and autoimmune diseases Annual General Meeting November 2007 Summary Objective Lead products Discovery Listings Location Facility Employees Cash (30/6/07) Shares outstanding
Therapeutic products for respiratory and autoimmune diseases Annual General Meeting November 2007 Summary Objective Lead products Discovery Listings Location Facility Employees Cash (30/6/07) Shares outstanding
Oppenheimer 29th Annual Healthcare Conference Mark Timney, Chief Executive Officer
 Oppenheimer 29th Annual Healthcare Conference Mark Timney, Chief Executive Officer New York City 20th March 2019 1 Safe Harbor Forward-looking statements Statements contained in this presentation about
Oppenheimer 29th Annual Healthcare Conference Mark Timney, Chief Executive Officer New York City 20th March 2019 1 Safe Harbor Forward-looking statements Statements contained in this presentation about
Medications for Managing COPD in Hospice Patients. Jim Joyner, PharmD, CGP Director of Clinical Operations Outcome Resources
 Medications for Managing COPD in Hospice Patients Jim Joyner, PharmD, CGP Director of Clinical Operations Outcome Resources Goal of medications in COPD Decrease symptoms and/or complications Reduce frequency
Medications for Managing COPD in Hospice Patients Jim Joyner, PharmD, CGP Director of Clinical Operations Outcome Resources Goal of medications in COPD Decrease symptoms and/or complications Reduce frequency
February 23, Q4 and Year-End 2016 Financial Results
 February 23, 2017 Q4 and Year-End 2016 Financial Results 2 RETHINKING CNS Agenda Today s Speakers Paul Cox, Senior Director, Investor Relations Jeff Jonas, M.D., Chief Executive Officer Jim Doherty, Ph.D.,
February 23, 2017 Q4 and Year-End 2016 Financial Results 2 RETHINKING CNS Agenda Today s Speakers Paul Cox, Senior Director, Investor Relations Jeff Jonas, M.D., Chief Executive Officer Jim Doherty, Ph.D.,
