HCT Medical Policy. Transcranial Magnetic Stimulation (TMS) for Major Depression Policy # 121 Current Effective Date: 05/24/2016.
|
|
|
- Claude Bradley
- 6 years ago
- Views:
Transcription
1 HCT Medical Policy Transcranial Magnetic Stimulation (TMS) for Major Depression Policy # 121 Current Effective Date: 05/24/2016 Medical Policies are developed by HealthyCT to assist in administering plan benefits and constitute neither offers of coverage nor medical advice. This Medical Policy contains only a partial, general description of plan or program benefits and does not constitute a contract. HealthyCT does not provide health care services and, therefore, cannot guarantee any results or outcomes. Participating providers are independent contractors in private practice and are neither employees nor agents of HealthyCT or its affiliates. Treating providers are solely responsible for medical advice and treatment of members. This Medical Policy may be updated and therefore is subject to change. Policy Statement Transcranial magnetic stimulation (TMS) with a U.S. Food and Drug Administration (FDA) approved device may be considered medically necessary in adult patients > 22 years of age with a diagnosis of major depressive disorder when all of the following criteria are met: 1. Documentation that psychotherapy was attempted without any significant improvement in depressive symptoms as measured by standard rating scales; and 2. Documentation that the patient s condition is medically stable and/or there are no co-morbid medical conditions that are contraindications for TMS such as any of the following: History of a psychotic disorder, including schizoaffective disorder, bipolar disease, or major depression with psychotic features in the current depressive episode; or History of increased intracranial pressure or head trauma; or History of suicide plan or recent attempt Neurological disorders, including cerebrovascular disease, primary or secondary tumors in CNS, cerebral aneurysm, dementia, or movement disorders; or Pregnancy and/or breast feeding; or Presence of an implanted magnetic-sensitive medical device or other implanted metal items in or near the head such as cochlear implant, implanted cardioverter defibrillator (ICD), pacemaker, vagus nerve stimulator (VNS), medication pumps, or metal aneurysm clips or coils, staples, or stents Seizure disorder, epilepsy and history of seizure Page 1 of 11
2 3. Electroconvulsive therapy (ECT) is contraindicated or the patient refuses ECT; and 4. Professional and/or social supports are in place after recovery from the procedure; the patient can be reasonably expected to comply with post-procedure recommendations and appropriate consent is documented for the procedure 5. One or more of the following: Documentation of medication treatment-resistance during the current depressive episode demonstrated by a lack of clinically significant response to four trials of psychopharmacologic agents from at least two different agent classes. At least one of the treatment trials must have been administered as a standard course of mono- or poly-drug therapy; or Documentation of intolerance to psychopharmacologic agents as evidenced by four trials of psychopharmacologic agents from at least two different agent classes, with distinct side effects; or Documented history of good response to TMS during an earlier episode of the treatment-resistant major depressive disorder 6. Transcranial magnetic stimulation is considered investigational and not medically necessary for all other indications Overview Transcranial magnetic stimulation (TMS) is a noninvasive technique that may be used as a treatment for major depression. TMS is described as brief repetitive pulses of magnetic energy that are applied to the scalp via a large electromagnetic coil that generates low levels of electrical current in the underlying brain tissue. The goal of TMS is to stimulate areas of the brain involved in mood regulation to lessen the duration or severity of depressive episodes. TMS may be used to augment pharmacotherapy or in place of a new medication in patients who have not responded to previous trials of antidepressant medication. TMS is also being investigated as a treatment for migraine headaches, neuropathic pain, Parkinson s, fibromyalgia, PTSD, schizophrenia and alcohol dependence among other conditions. TMS can be performed on an outpatient basis and does not require anesthesia. Ear plugs are worn to diminish the noise from the discharging coil. Magnetic resonance imaging can be used to facilitate precise targeting of selected brain regions, which can be unilateral or bilateral. For treatment of depression, a variant of TMS called repetitive TMS (rtms) is used that delivers rhythmic pulses of electromagnetism (instead of a single pulse), using a cut-off of 1 Hz; high or low frequency can be used. The intensity of rtms is usually set as a percentage of the patient s motor threshold (MT), defined as the minimum stimulus strength required to induce involuntary muscle movements (usually in the hand) five times out of ten. Depending on intensity parameters, the patient may experience involuntary spasms of Page 2 of 11
3 scalp muscles. Treatment with rtms usually involves daily sessions lasting about 30 minutes for 2 4 weeks although treatment can extend longer. Scientific Rationale The peer reviewed medical evidence from a number of double-blind, randomized, sham-controlled trials has shown TMS to be effective in modestly reducing depression symptoms in patients with moderate to severe depression that is resistant to antidepressant medication. Randomized, Double-Blind, Sham-Controlled Studies Evaluating TMS In a sham-controlled randomized trial by George et al. (2010) active and sham HFL-TMS at 10 Hz and 120% MT were compared in 199 patients with MDD who were anti-depression medication-free. In a 2- week lead in phase, no treatment or drugs were allowed other than minimal use of sedatives, hypnotics, or anxiolytics. In phase 1, active or sham TMS was delivered daily for three weeks, and patients achieving a reduction of 30% in the HAMD-24 score could continue assigned treatment for an additional three weeks. The study applied the standard definition for response for the HAMD-21 but defined remission as a HAMD-24 score of 3 on two consecutive evaluations. Compared with patients receiving sham TMS, patients receiving active TMS demonstrating significantly greater improvement in mean scores for the MADRS, CGI-S, and IDS-SR but not the HAMD-24. Despite this, active TMS led to higher rates of response (15% versus 5%) and remission (14% versus 5%) than sham TMS. Logistic regression analysis revealed that the treatment approach was the only variable with a significant effect on response or remission, whether analyzing data for the ITT sample (P=0.009 for response; P=0.02 for remission) or analyzing data for the so-called completer sample, 154 patients with 4 rescheduled, missed, or partially completed TMS sessions (P=0.02 for response; P=0.02 for remission). There was no significant treatment effect in the fully adherent sample, 120 patients with < 2 rescheduled, missed, or partially completed TMS sessions. In a randomized controlled sham trial Janicak et al. (2010) assessed the durability of antidepressant effect after acute response to TMS in patients with major depressive disorder (MDD) using protocolspecified maintenance antidepressant monotherapy. Three hundred one patients were randomly assigned to active or sham TMS in a 6-week, controlled trial. Nonresponders could enroll in a second, 6- week, open-label study. Patients who met criteria for partial response (i.e., >25% decrease from the baseline HAMD 17) during either the sham-controlled or open-label study (n = 142) were tapered off TMS over 3 weeks, while simultaneously starting maintenance antidepressant monotherapy. Patients were then followed for 24 weeks in a naturalistic follow-up study examining the long-term durability of TMS. During this durability study, TMS was readministered if patients met prespecified criteria for symptom worsening (i.e., a change of at least one point on the CGI-S scale for 2 consecutive weeks). Relapse was the primary outcome measure. Ten of 99 (10%; Kaplan-Meier survival estimate = 12.9%) Page 3 of 11
4 patients relapsed. Thirty-eight (38.4%) patients met criteria for symptom worsening and 32/38 (84.2%) reachieved symptomatic benefit with adjunctive TMS. Safety and tolerability were similar to acute TMS monotherapy. Slotema et al. (2010) performed a meta-analysis of the efficacy of rtms in psychiatric disorders. Data were obtained from randomized, sham-controlled studies of rtms treatment for depression (34 studies), auditory verbal hallucinations (AVH, 7 studies), negative symptoms in schizophrenia (7 studies), and obsessive-compulsive disorder (OCD, 3 studies). Studies of rtms versus electroconvulsive treatment (ECT, 6 studies) for depression were meta-analyzed. The mean weighted effect size of rtms versus sham for depression was 0.55 (P<.001). Monotherapy with rtms was more effective than rtms as adjunctive to antidepressant medication. ECT was superior to rtms in the treatment of depression (mean weighted effect size -0.47, P =.004). In the treatment of AVH, rtms was superior to sham treatment, with a mean weighted effect size of 0.54 (P<.001). The mean weighted effect size for rtms versus sham in the treatment of negative symptoms in schizophrenia was 0.39 (P =.11) and for OCD, 0.15 (P =.52). Side effects were mild, yet more prevalent with high-frequency rtms at frontal locations. The authors concluded that rtms may be used as a clinical treatment method for depression, for auditory verbal hallucinations, and possibly for negative symptoms. rtms is not recommend for the treatment of OCD. Schutter (2009) conducted another large meta-analysis that included 30 double-blind sham-controlled trials with 1,164 patients. Studies enrolling patients with major depression, employing high frequency (>5 Hz) TMS over the left DLPFC, a minimum of 5 treatment sessions, and measuring pretreatment and post-treatment depression scores using either the HAMD or MADRS were selected. In addition to calculating an overall treatment effect, they performed additional analyses comparing effects between studies that treated patients with medication-resistant depression only (n=17) and patients with nonmedication resistant depression (n=8), and studies that used lower-intensity MT intensity (<100% MT, n=14) versus studies that used higher intensity ( % MT, n=16). The overall weighted mean effect size for treatment was 0.39 (95% CI: , p<0.0001). An analysis comparing studies with patients with treatment-resistant depression versus nontreatment-resistant depression found no difference in effect size. Treatment intensity, comparing studies using high versus low MT intensity, also did not show a difference in effect size. Unilateral vs. Bilateral TMS A randomized double blind study by Blumberger et al. (2012) evaluated the efficacy of high frequency left-sided (HFL) and sequential bilateral repetitive transcranial magnetic stimulation (rtms) compared to sham in treatment-resistant major depression (TRD). Seventy-four subjects with TRD and a 17-item Hamilton Depression Rating Scale (HDRS) greater than 21 were randomized to receive unilateral, bilateral, or sham rtms. The rates of remission were compared among the three treatment groups. The remission rates differed significantly among the three treatment groups using a modified intention to Page 4 of 11
5 treat analysis that excluded subjects who did not respond to electroconvulsive therapy (ECT) during the current episode. The remission rate was significantly higher in the bilateral group than the sham group. The remission rate in the unilateral group did not differ from either group. Another double blind randomized controlled trial by Fitzgerald et al. (2012) investigated whether there is an advantage in efficacy of sequential bilateral rtms compared to standard high-frequency left sided rtms. Sixty seven patients with treatment resistant depression were included in a randomised doubleblind sham controlled trial of sequential bilateral rtms compared to standard high-frequency left sided rtms and sham rtms over a three-week period. The study also included a further three week comparison of the two active treatment conditions. The primary outcome variable was scores on the 17- item Hamilton Depression Rating Scale (HAMD). In the three-week double-blind phase of the trial there was a greater antidepressant response to unilateral left sided rtms compared with sham or bilateral rtms. Across the full six weeks of active rtms, there was also a consistent pattern of improved response in unilateral left compared to bilateral treatment. Response rates were low in both active groups. Randomized Controlled Trials Comparing TMS and Other Treatments for Depression Keshtkar et al. (2011) compared the efficacy of rtms and ECT in adult patients with refractory major depressive disorder (MDD). 73 patients with MDD were randomized to ECT-controlled or parallel-group clinical trial to analyze the antidepressant effects of ECT and rtms. The Beck Depression Inventory and Hamilton Depression Rating Scale were used to measure depression. The results showed that both ECT and rtms significantly improved depression and suicidal behavior scores. However, ECT reduced depression and suicidal behavior scores more than rtms. There were no significant adverse effects in the rtms group. The investigators concluded that both ECT and rtms improved MDD in the short term, but the antidepressant efficacy of ECT was greater than rtms. Additionally, ECT led to greater reductions in suicidal behavior than rtms. Until strong evidence for the safety and efficacy of rtms is available, further studies are needed to compare ECT and rtms in terms of the long-term relapse rate and quality of life. Bares et al. (2009) randomized 60 patients with major depression to active LFR-TMS at 1 Hz and 100% MT + placebo drug (TMS group) or sham TMS + venlafaxine (Drug group). Active and sham TMS were delivered for 20 sessions over four weeks, and placebo and active medication were administered daily during that time. Other antidepressant drugs were withdrawn during a drug wash-out phase 5 to 9 days before beginning study treatment. The study was double-blind, defined response as a decrease of 50% in the MADRS score, and defined remission as a MADRS score of 10. At the end of treatment, mean scores for the three depression measures used (MADRS, CGI, BDI) were similar between groups. There also were no significant differences between groups in the rates of response or the rates of remission, whether analyzed for all patients (ITT, sample) or only patients completing treatment (completer sample). In the ITT sample and the completer sample, respectively, the rate of response was 33.3% and Page 5 of 11
6 34.6% in the TMS group and 38.7% and 42.3% in the Drug group, and the rate of remission was 18.5% and 19.2% in the TMS group and 22.6% and 26.9% in the Drug group. The study by O Reardon et al. (2007) is the largest clinical trial of TMS, and was the clinical trial on which the FDA decision to clear TMS for marketing was based. The study was a multicenter study conducted at 23 sites. Patients were enrolled who had uncomplicated major depression meeting severity criteria. They were required to have failed at least one but no more than four adequate antidepressant treatments in the current or most recent episode of depression. After a 1-week washout, patients were scheduled to have 6 weeks of TMS, 5 sessions per week. Characteristics of the treatment given were left DLPFC treatment location, 120% motor threshold field intensity, 10 Hz pulse frequency, and 3000 pulses per treatment. Patients were not given antidepressant medication during the treatment period. Threehundred twenty-five patients were randomized, and the analysis presented in the published study is based on 301 patients who had at least one post-baseline assessment. The results of the trial s 4-week outcomes reported that the mean difference in 4-week change MADSRS was declared a priori to be the primary outcome of the study. The TMS patients improved 5.6 points on the MADRS, and the sham group improved 3.5 points, leading to a mean difference of 2.1 points which was not statistically significant (p=0.057). On other outcomes measured by mean changes from baseline, the HAMD 24 had a mean difference 2.4 (p=0.012), the HAMD 17 had a mean difference of 1.9 (p=0.006) and the CGI-S had a mean difference of 0.4 (p=0.009). Results were also presented in terms of response and remission rates. Response rates were generally low, but depending on the scale used, the differences in response rates ranged from 7.1 to 9%, and were statistically significant. The remission rates were even lower, between 7.1% and 9.0%, and were not significant when compared to sham treated patients. Professional Society Recommendations: American Psychiatric Association (APA): 1 In the Practice Guideline for the Treatment of Major Depressive Disorder published 2010, transcranial magnetic stimulation is mentioned as a treatment option for patients who do not respond to pharmacotherapy. The guideline outlines the following key points: In comparisons of actual TMS versus sham TMS, recent meta-analyses have found relatively small to moderate benefits of TMS in terms of clinical response. These meta-analyses also support the use of high-frequency TMS over the left dorsolateral prefrontal cortex. In comparison with ECT, TMS has been found in randomized studies to be either less effective than ECT or comparable in efficacy to ECT. Across all studies, TMS was well tolerated and was associated with low rates of treatment dropout Transient scalp discomfort and headaches were the most commonly reported side effects In clinical practice, the need for daily TMS could produce logistical barriers for some patients Page 6 of 11
7 Institute for Clinical Systems Improvement (ICSI): ICSI recommended interventions for treatment resistant depression include: medication augmentation strategies, hospitalization, ECT, and phototherapy. ECT may be indicated in cases of geriatric depression, intolerance of AD medication, unsuccessful trials of AD medications, catatonia, previous successful treatment with ECT, need for rapid response, depression with psychoses, and predominant melancholic symptoms. National Institute for Health and Care Excellence (NICE): NICE recommends combination therapy with AD medication and cognitive-behavioral therapy (CBT) for patients who have not responded to AD medication or any form of psychotherapy. ECT is recommended for patients with moderate-to-severe depression that has not resolved after trials of multiple (number unspecified) AD medications and psychological treatments. NICE recommends against the routine use of ECT for moderate depression. NICE suggests that the use of TMS be reserved for investigational use only as there is uncertainty about the procedure's clinical efficacy but there are no major safety concerns associated with TMS. Related Codes 90867: Therapeutic repetitive transcranial magnetic stimulation (TMS) treatment; initial, including cortical mapping, motor threshold determination, delivery and management 90868: Therapeutic repetitive transcranial magnetic stimulation (TMS) treatment; subsequent delivery and management, per session 90869: Therapeutic repetitive transcranial magnetic stimulation (TMS) treatment; subsequent motor threshold re-determination with delivery and management References 1. American Psychiatric Association (APA). Practice Guideline for the Treatment of Major Depressive Disorder. Published Bares M, Kopecek M, Novak T, et al. Low frequency (1-Hz), right prefrontal repetitive transcranial magnetic stimulation (rtms) compared with venlafaxine ER in the treatment of resistant depression: a double-blind, single-centre, randomized study. J Affect Disord. 2009;118(1-3): Blue Cross Blue Shield Association (BCBSA). Technology Evaluation Center (TEC). Transcranial magnetic stimulation for depression. TEC Assessment Program. Chicago, IL Accessed at: Page 7 of 11
8 4. Blumberger DM, Mulsant BH, Fitzgerald PB, et al. A randomized double-blind sham-controlled comparison of unilateral and bilateral repetitive transcranial magnetic stimulation for treatment-resistant major depression. World J Biol Psychiatry. 2012;13(6): Center for Devices and Radiological Health (CDRH). 510(k) Premarket Notification Database [search: Product Codes OBP; GWF]. Food and Drug Administration (FDA) [website]. Accessed at: 6. Conforto AB, Amaro E Jr et al. Randomized, proof-of-principle clinical trial of active transcranial magnetic stimulation in chronic migraine. Cephalalgia Dec Eranti S, Mogg A, Pluck G, et al. A randomized, controlled trial with 6-month follow-up of repetitive transcranial magnetic stimulation and electroconvulsive therapy for severe depression. Am J Psychiatry. 2007;164(1): Fitzgerald PB, Hoy KE, Herring SE, et al. A double blind randomized trial of unilateral left and bilateral prefrontal cortex transcranial magnetic stimulation in treatment resistant major depression. J Affect Disord. 2012; 139(2): George MS, Lisanby SH, Avery D, et al. Daily left prefrontal transcranial magnetic stimulation therapy for major depressive disorder. a sham-controlled randomized trial. Arch Gen Psychiatry. 2010; 67(5): Institute for Clinical Systems Improvement Guideline: Mitchell J, Trangle M, Degnan B, et al. Institute for Clinical Systems Improvement. Adult Depression in Primary Care. Updated March Available at: Janicak PG, Nahas Z, et al. Durability of clinical benefit with transcranial magnetic stimulation (TMS) in the treatment of pharmacoresistant major depression: assessment of relapse during a 6-month, multisite, open-label study. Brain Stimul Oct;3(4): doi: /j.brs Epub 2010 Aug Jorge RE, Moser DJ, Acion L, Robinson RG. Treatment of vascular depression using repetitive transcranial magnetic stimulation. Arch Gen Psychiatry. 2008;65(3): Kennedy SH, Milev R, Giacobbe P, et al.; Canadian Network for Mood and Anxiety Treatments (CANMAT). Canadian Network for Mood and Anxiety Treatments (CANMAT) Clinical guidelines for the management of major depressive disorder in adults. IV. Neurostimulation therapies. J Affect Disord. 2009;117(Suppl 1):S44-S Keshtkar M, Ghanizadeh A, Firoozabadi A. Repetitive transcranial magnetic stimulation versus electroconvulsive therapy for the treatment of major depressive disorder, a randomized controlled clinical trial. J ECT Dec;27(4): Lam RW, Chan P, Wilkins-Ho M, Yatham LN. Repetitive transcranial magnetic stimulation for treatment-resistant depression: a systematic review and metaanalysis. Can J Psychiatry. 2008;53(9): Lipton RB, Dodick DW, Silberstein SD et al. Single-pulse transcranial magnetic stimulation for acute treatment of migraine with aura: a randomised, double-blind, parallel-group, sham- Page 8 of 11
9 controlled trial. Lancet Neurol Apr;9(4): doi: /S (10) Epub 2010 Mar Mantovani A, Pavlicova M, Avery D, et al. Long-term efficacy of repeated daily prefrontal transcranial magnetic stimulation (TMS) in treatment-resistant depression. Depress Anxiety. 2012;29(10): Mogg A, Pluck G, Eranti SV, et al. A randomized controlled trial with 4-month follow-up of adjunctive repetitive transcranial magnetic stimulation of the left prefrontal cortex for depression. Psychol Med. 2008;38(3): National Institute for Health and Clinical Excellence (NICE). Depression in adults. The treatment and management of depression in adults. London, UK: National Institute for Health and Clinical Excellence; NICE Care Guideline No. 90. Available at: O Reardon JP, Solvason HB, Janicak PG, et al. Efficacy and safety of transcranial magnetic stimulation in the acute treatment of major depression: a multisite randomized controlled trial. Biol Psychiatry. 2007;62(11): Rosa MA, Gattaz WF, Pascual-Leone A, et al. Comparison of repetitive transcranial magnetic stimulation and electroconvulsive therapy in unipolar non-psychotic refractory depression: a randomized, single-blind study. Int J Neuropsychopharmacol. 2006;9(6): Schutter DJ. Antidepressant efficacy of high frequency transcranial magnetic stimulation over the left dorsolateral prefrontal cortex in double-blind sham controlled designs: a meta-analysis. Psychol Med, 2009;39: Slotema CW, Blom JD, Hoek HW, Sommer IE. Should we expand the toolbox of psychiatric treatment methods to include Repetitive Transcranial Magnetic Stimulation (rtms)? A metaanalysis of the efficacy of rtms in psychiatric disorders. J Clin Psychiatry Jul;71(7): Epub 2010 Mar U.S. Food & Drug Administration (FDA) [website]: FDA News Release. FDA allows marketing of first device to relieve migraine headache pain. Accessed at: U.S. Food & Drug Administration (FDA) [website]: Guidance for Industry and FDA Staff - Class II Special Controls Guidance Document: Repetitive Transcranial Magnetic Stimulation (rtms) Systems. July 26, Accessed at: htm 26. U.S. Food and Drug Administration (FDA). 510(k) Premarket Notification Database. Brainsway Deep TMS System. No. K Rockville, MD: FDA. January 7, Accessed at: U.S. Food and Drug Administration (FDA). 510(k) Premarket Notification Database. NeuroStar TMS Therapy System. No. K and K Rockville, MD: FDA. December 16, Accessed at: Page 9 of 11
10 28. Berlim MT, van den Eynde F, Tovar-Perdomo S, Daskalakis ZJ. Response, remission and drop-out rates following high-frequency repetitive transcranial magnetic stimulation (rtms) for treating major depression: a systematic review and meta-analysis of randomized, double-blind and sham-controlled trials. Psychol Med 2014;44: Cheng CM(1), Juan CH(2), Chen MH(3) et al. Different forms of prefrontal theta burst stimulation for executive function of medication- resistant depression: Evidence from a randomized shamcontrolled study. Prog Neuropsychopharmacol Biol Psychiatry Apr 3;66: doi: /j.pnpbp Epub 2015 Nov Chou YH, Hickey PT, Sundman M, et al. Effects of repetitive transcranial magnetic stimulation on motor symptoms in Parkinson disease: a systematic review and meta-analysis. JAMA Neurol. 2015; 72(4): Dlabac-de Lange JJ, Bais L, van Es FD, et al. Efficacy of bilateral repetitive transcranial magnetic stimulation for negative symptoms of schizophrenia: results of a multicenter double-blind randomized controlled trial. Psychol Med. 2015; 45(6): Kedzior KK, Reitz SK, Azorina V, et al. Durability of the antidepressant effect of the highfrequency repetitive transcranial magnetic stimulation (rtms) in the absence of maintenance treatment in major depression: a systematic review and meta-analysis of 16 double-blind, randomized, sham-controlled trials. Depress Anxiety. 2015; 32(3): Kedzior KK, Gellersen HM, Brachetti AK et al. Deep transcranial magnetic stimulation (DTMS) in the treatment of major depression: An exploratory systematic review and meta-analysis. J Affect Disord Nov 15;187: doi: /j.jad Epub 2015 Aug Levkovitz Y, Isserles M, Padberg F, et al. Efficacy and safety of deep transcranial magnetic stimulation for major depression: a prospective multicenter randomized controlled trial. World Psychiatry. 2015; 14(1): Liu B, Zhang Y, Zhang L, Li L. Repetitive transcranial magnetic stimulation as an augmentative strategy for treatment-resistant depression, a meta-analysis of randomized, double-blind and sham-controlled study. BMC Psychiatry Nov 30;14:342. doi: /s Noda Y, Silverstein WK, Barr MS, et al. Neurobiological mechanisms of repetitive transcranial magnetic stimulation of the dorsolateral prefrontal cortex in depression: a systematic review. Psychol Med Dec;45(16): doi: /S Epub 2015 Sep Prasser J, Schecklmann M, Poeppl TB et al. Bilateral prefrontal rtms and theta burst TMS as an add-on treatment for depression: a randomized placebo controlled trial. World J Biol Psychiatry Jan;16(1): doi: / Epub 2014 Nov 28. Document History Date Action 4/1/2015 Original Version 4/15/2016 Annual Review. Reference #10 updated. References #28-37 added to policy. Page 10 of 11
11 05/24/2016 Updates reviewed and approved at UMC Page 11 of 11
Original Effective Date: 8/28/2013. Subject: Transcranial Magnetic Stimulation for the Treatment of Major Depression
 Subject: Transcranial Magnetic Stimulation for the Treatment of Major Depression Guidance Number: MCG-104 Revision Date(s): Original Effective Date: 8/28/2013 Medical Coverage Guidance Approval Date: 8/28/2013
Subject: Transcranial Magnetic Stimulation for the Treatment of Major Depression Guidance Number: MCG-104 Revision Date(s): Original Effective Date: 8/28/2013 Medical Coverage Guidance Approval Date: 8/28/2013
Transcranial Magnetic Stimulation
 Transcranial Magnetic Stimulation Date of Origin: 7/24/2018 Last Review Date: 7/24/2018 Effective Date: 08/01/18 Dates Reviewed: 7/24/2018 Developed By: Medical Necessity Criteria Committee I. Description
Transcranial Magnetic Stimulation Date of Origin: 7/24/2018 Last Review Date: 7/24/2018 Effective Date: 08/01/18 Dates Reviewed: 7/24/2018 Developed By: Medical Necessity Criteria Committee I. Description
Statement on Repetitive Transcranial Magnetic Stimulation for Depression. Position statement CERT03/17
 Statement on Repetitive Transcranial Magnetic Stimulation for Depression Position statement CERT03/17 Approved by the Royal College of Psychiatrists, Committee on ECT and Related Treatments: February 2017
Statement on Repetitive Transcranial Magnetic Stimulation for Depression Position statement CERT03/17 Approved by the Royal College of Psychiatrists, Committee on ECT and Related Treatments: February 2017
Patient Manual Brainsway Deep Transcranial Magnetic Stimulation (Deep TMS) System for Treatment of Major Depressive Disorder
 Dr. Zahida Tayyib www.mvptms.com Patient Manual Brainsway Deep Transcranial Magnetic Stimulation (Deep TMS) System for Treatment of Major Depressive Disorder If you are considering Brainsway Deep treatment
Dr. Zahida Tayyib www.mvptms.com Patient Manual Brainsway Deep Transcranial Magnetic Stimulation (Deep TMS) System for Treatment of Major Depressive Disorder If you are considering Brainsway Deep treatment
Repetitive Transcranial Magnetic Stimulation (rtms)
 Page 1 of 33 Medical Policies - Mental Health Repetitive Transcranial Magnetic Stimulation (rtms) Print Number: PSY301.015 Effective Date: 07-01-2014 Coverage: Initial rtms Treatment Repetitive transcranial
Page 1 of 33 Medical Policies - Mental Health Repetitive Transcranial Magnetic Stimulation (rtms) Print Number: PSY301.015 Effective Date: 07-01-2014 Coverage: Initial rtms Treatment Repetitive transcranial
Case 2:15-cv MWF-JPR Document Filed 03/20/17 Page 1 of 36 Page ID #:388 EXHIBIT 1
 Case 2:15-cv-07074-MWF-JPR Document 65-10 Filed 03/20/17 Page 1 of 36 Page ID #:388 EXHIBIT 1 Case 2:15-cv-07074-MWF-JPR Document 65-10 Filed 03/20/17 Page 2 of 36 Page ID #:389 Cigna Medical Coverage
Case 2:15-cv-07074-MWF-JPR Document 65-10 Filed 03/20/17 Page 1 of 36 Page ID #:388 EXHIBIT 1 Case 2:15-cv-07074-MWF-JPR Document 65-10 Filed 03/20/17 Page 2 of 36 Page ID #:389 Cigna Medical Coverage
Repetitive Transcranial Magnetic Stimulation as a for Treatment of Refractory Depression and other Psychiatric/Neurologic Disorders
 Repetitive Transcranial Magnetic Stimulation as a for Treatment of Refractory Depression and other Psychiatric/Neurologic Disorders Policy Number: Original Effective Date: MM.12.015 08/01/2014 Line(s)
Repetitive Transcranial Magnetic Stimulation as a for Treatment of Refractory Depression and other Psychiatric/Neurologic Disorders Policy Number: Original Effective Date: MM.12.015 08/01/2014 Line(s)
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Transcranial Magnetic Stimulation (TMS) Page 1 of 24 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: See also: Transcranial Magnetic Stimulation (TMS) Vagus Nerve
Transcranial Magnetic Stimulation (TMS) Page 1 of 24 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: See also: Transcranial Magnetic Stimulation (TMS) Vagus Nerve
Transcranial Magnetic Stimulation and Cranial Electrical Stimulation (CES) as a Treatment of Depression and Other Psychiatric/Neurologic Disorders
 Transcranial Magnetic Stimulation and Cranial Electrical Stimulation (CES) as a Treatment of Depression and Other Psychiatric/Neurologic Disorders Policy Number: 2.01.50 Last Review: 10/2017 Origination:
Transcranial Magnetic Stimulation and Cranial Electrical Stimulation (CES) as a Treatment of Depression and Other Psychiatric/Neurologic Disorders Policy Number: 2.01.50 Last Review: 10/2017 Origination:
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Transcranial Magnetic Stimulation (TMS) Page 1 of 21 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: See also: Transcranial Magnetic Stimulation (TMS) Vagus Nerve
Transcranial Magnetic Stimulation (TMS) Page 1 of 21 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: See also: Transcranial Magnetic Stimulation (TMS) Vagus Nerve
Repetitive transcranial magnetic stimulation for depression
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Interventional procedure consultation document Repetitive transcranial magnetic stimulation for depression Depression causes low mood or sadness that can
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Interventional procedure consultation document Repetitive transcranial magnetic stimulation for depression Depression causes low mood or sadness that can
Transcranial Magnetic Stimulation for the Treatment of Depression
 Transcranial Magnetic Stimulation for the Treatment of Depression Paul E. Holtzheimer, MD Associate Professor Departments of Psychiatry and Surgery Geisel School of Medicine at Dartmouth Dartmouth-Hitchcock
Transcranial Magnetic Stimulation for the Treatment of Depression Paul E. Holtzheimer, MD Associate Professor Departments of Psychiatry and Surgery Geisel School of Medicine at Dartmouth Dartmouth-Hitchcock
Medical Policy An Independent Licensee of the Blue Cross and Blue Shield Association
 of Depression and Other Psychiatric Disorders Page 1 of 26 Medical Policy An Independent Licensee of the Blue Cross and Blue Shield Association Title: See also: Transcranial Magnetic Stimulation (TMS)
of Depression and Other Psychiatric Disorders Page 1 of 26 Medical Policy An Independent Licensee of the Blue Cross and Blue Shield Association Title: See also: Transcranial Magnetic Stimulation (TMS)
TRANSCRANIAL MAGNETIC STIMULATION
 MEDICAL POLICY TRANSCRANIAL MAGNETIC STIMULATION Policy Number: 2013T0536F Effective Date: December 1, 2013 Table of Contents COVERAGE RATIONALE... BACKGROUND... CLINICAL EVIDENCE... U.S. FOOD AND DRUG
MEDICAL POLICY TRANSCRANIAL MAGNETIC STIMULATION Policy Number: 2013T0536F Effective Date: December 1, 2013 Table of Contents COVERAGE RATIONALE... BACKGROUND... CLINICAL EVIDENCE... U.S. FOOD AND DRUG
Neuromodulation Approaches to Treatment Resistant Depression
 1 Alternative Treatments: Neuromodulation Approaches to Treatment Resistant Depression Audrey R. Tyrka, MD, PhD Assistant Professor Brown University Department of Psychiatry Associate Chief, Mood Disorders
1 Alternative Treatments: Neuromodulation Approaches to Treatment Resistant Depression Audrey R. Tyrka, MD, PhD Assistant Professor Brown University Department of Psychiatry Associate Chief, Mood Disorders
What is Repetitive Transcranial Magnetic Stimulation?
 rtms for Refractory Depression: Findings and Future Jonathan Downar, MD PhD Asst Professor, Dept of Psychiatry University of Toronto, Canada Co-Director, rtms Clinic Toronto Western Hospital University
rtms for Refractory Depression: Findings and Future Jonathan Downar, MD PhD Asst Professor, Dept of Psychiatry University of Toronto, Canada Co-Director, rtms Clinic Toronto Western Hospital University
Effective Date: July 27, 2016
 Medical Review Criteria Transcranial Magnetic Stimulation Effective Date: July 27, 2016 Subject: Transcranial Magnetic Stimulation Policy: An initial course of left prefrontal TMS 1 is covered for: Patients
Medical Review Criteria Transcranial Magnetic Stimulation Effective Date: July 27, 2016 Subject: Transcranial Magnetic Stimulation Policy: An initial course of left prefrontal TMS 1 is covered for: Patients
Transcranial Magnetic Stimulation as a Treatment of Depression and Other Psychiatric/Neurologic Disorders Corporate Medical Policy
 Transcranial Magnetic Stimulation as a Treatment of Depression and Other Psychiatric/Neurologic Disorders Corporate Medical Policy File name: Transcranial Magnetic Stimulation as a Treatment of Depression
Transcranial Magnetic Stimulation as a Treatment of Depression and Other Psychiatric/Neurologic Disorders Corporate Medical Policy File name: Transcranial Magnetic Stimulation as a Treatment of Depression
Transcranial Magnetic Stimulation and Cranial Electrical Stimulation (CES) as a Treatment of Depression and Other Psychiatric/Neurologic Disorders
 Transcranial Magnetic Stimulation and Cranial Electrical Stimulation (CES) as a Treatment of Depression and Other Psychiatric/Neurologic Disorders Policy Number: 2.01.50 Last Review: 10/2014 Origination:
Transcranial Magnetic Stimulation and Cranial Electrical Stimulation (CES) as a Treatment of Depression and Other Psychiatric/Neurologic Disorders Policy Number: 2.01.50 Last Review: 10/2014 Origination:
Clinical Policy: Transcranial Magnetic Stimulation
 Clinical Policy: Reference Number: CP.MP.172 Last Review Date: 12/18 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory and legal information. Description
Clinical Policy: Reference Number: CP.MP.172 Last Review Date: 12/18 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory and legal information. Description
Todd Hutton, M.D. Karl Lanocha, M.D Richard Bermudes, M.D. Kimberly Cress, M.D.
 Transcranial Magnetic Stimulation: What You Need To Know Todd Hutton, M.D. Karl Lanocha, M.D Richard Bermudes, M.D. Kimberly Cress, M.D. Disclosures Todd Hutton, M.D. Speaker for Neuronetics Board of Directors,
Transcranial Magnetic Stimulation: What You Need To Know Todd Hutton, M.D. Karl Lanocha, M.D Richard Bermudes, M.D. Kimberly Cress, M.D. Disclosures Todd Hutton, M.D. Speaker for Neuronetics Board of Directors,
TRANSCRANIAL MAGNETIC STIMULATION
 TRANSCRANIAL MAGNETIC STIMULATION Optum Behavioral Clinical Policy Policy Number: BH727BCPTMS_112017 Effective Date: November, 2017 Table of Contents Page BENEFIT CONSIDERATIONS... 1 COVERAGE RATIONALE...
TRANSCRANIAL MAGNETIC STIMULATION Optum Behavioral Clinical Policy Policy Number: BH727BCPTMS_112017 Effective Date: November, 2017 Table of Contents Page BENEFIT CONSIDERATIONS... 1 COVERAGE RATIONALE...
FDA CLEARS NEUROSTAR TMS THERAPY FOR THE TREATMENT OF DEPRESSION
 FOR IMMEDIATE RELEASE FDA CLEARS NEUROSTAR TMS THERAPY FOR THE TREATMENT OF DEPRESSION First and Only Non-systemic and Non-invasive Treatment Cleared for Patients Who Have Not Benefited From Prior Antidepressant
FOR IMMEDIATE RELEASE FDA CLEARS NEUROSTAR TMS THERAPY FOR THE TREATMENT OF DEPRESSION First and Only Non-systemic and Non-invasive Treatment Cleared for Patients Who Have Not Benefited From Prior Antidepressant
Subject: Transcranial Magnetic Stimulation
 01-93875-18 Original Effective Date: 06/15/02 Reviewed: 12/06/18 Revised: 12/15/18 Subject: Transcranial Magnetic Stimulation THIS MEDICAL COVERAGE GUIDELINE IS NOT AN AUTHORIZATION, CERTIFICATION, EXPLANATION
01-93875-18 Original Effective Date: 06/15/02 Reviewed: 12/06/18 Revised: 12/15/18 Subject: Transcranial Magnetic Stimulation THIS MEDICAL COVERAGE GUIDELINE IS NOT AN AUTHORIZATION, CERTIFICATION, EXPLANATION
Administration of repetitive transcranial magnetic stimulation (rtms)
 Professional Practice Guideline 16 Administration of repetitive transcranial magnetic stimulation (rtms) November 2018 Authorising Body: Responsible Committee/Department: Responsible Department: Document
Professional Practice Guideline 16 Administration of repetitive transcranial magnetic stimulation (rtms) November 2018 Authorising Body: Responsible Committee/Department: Responsible Department: Document
Treatment of Depression: A Brief History
 2500 82nd Place Urbandale, Ia Treatment of Depression: A Brief History 1960 s to Present Community Based Services SSRI Antidepressants SNRI Antidepressants 1970 s to present 20% of patients do not respond
2500 82nd Place Urbandale, Ia Treatment of Depression: A Brief History 1960 s to Present Community Based Services SSRI Antidepressants SNRI Antidepressants 1970 s to present 20% of patients do not respond
Frequently Asked Questions FAQS. NeuroStar TMS Therapies
 Frequently Asked Questions FAQS NeuroStar TMS Therapies Provided by Dr Terrence A. Boyadjis MD 790 E Market Street Suite 245 West Chester, PA 19382 610.738.9576 FAQS About TMS Therapies Page 1 NeuroStar
Frequently Asked Questions FAQS NeuroStar TMS Therapies Provided by Dr Terrence A. Boyadjis MD 790 E Market Street Suite 245 West Chester, PA 19382 610.738.9576 FAQS About TMS Therapies Page 1 NeuroStar
MEDICAL POLICY SUBJECT: TRANSCRANIAL MAGNETIC STIMULATION
 MEDICAL POLICY SUBJECT: TRANSCRANIAL MAGNETIC CATEGORY: Technology Assessment 12/18/14, 10/15/15, 12/15/16, 12/21/17 PAGE: 1 OF: 14 If a product excludes coverage for a service, it is not covered, and
MEDICAL POLICY SUBJECT: TRANSCRANIAL MAGNETIC CATEGORY: Technology Assessment 12/18/14, 10/15/15, 12/15/16, 12/21/17 PAGE: 1 OF: 14 If a product excludes coverage for a service, it is not covered, and
The Next Chapter in Brain Stimulation Therapy
 The Next Chapter in Brain Stimulation Therapy Outline I. Depression and Treatment Resistant Depression Barriers to Treatment Treatment Options Cost and Impact of Depression II. Electroconvulsive Therapy
The Next Chapter in Brain Stimulation Therapy Outline I. Depression and Treatment Resistant Depression Barriers to Treatment Treatment Options Cost and Impact of Depression II. Electroconvulsive Therapy
Transcranial Magnetic Stimulation (TMS)
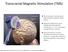 Transcranial Magnetic Stimulation (TMS) The treatment coil produces MRI-strength magnetic field pulses. Magnetic field pulses pass unimpeded through the cranium for 2-3 cm. and induce a small electric
Transcranial Magnetic Stimulation (TMS) The treatment coil produces MRI-strength magnetic field pulses. Magnetic field pulses pass unimpeded through the cranium for 2-3 cm. and induce a small electric
Transcranial Magnetic Stimulation as a Treatment of Depression and Other Psychiatric/Neurologic Disorders Corporate Medical Policy
 Transcranial Magnetic Stimulation as a Treatment of Depression and Other Psychiatric/Neurologic Disorders Corporate Medical Policy File Name: Transcranial Magnetic Stimulation as a Treatment of Depression
Transcranial Magnetic Stimulation as a Treatment of Depression and Other Psychiatric/Neurologic Disorders Corporate Medical Policy File Name: Transcranial Magnetic Stimulation as a Treatment of Depression
BRAIN STIMULATION AN ALTERNATIVE TO DRUG THERAPY IN MATERNAL DEPRESSION?
 BRAIN STIMULATION AN ALTERNATIVE TO DRUG THERAPY IN MATERNAL DEPRESSION? Kira Stein, MD Medical Director West Coast Life Center Sherman Oaks, California CA Maternal Mental Health Initiative - 2013 2013
BRAIN STIMULATION AN ALTERNATIVE TO DRUG THERAPY IN MATERNAL DEPRESSION? Kira Stein, MD Medical Director West Coast Life Center Sherman Oaks, California CA Maternal Mental Health Initiative - 2013 2013
Transcranial Magnetic Stimulation
 Transcranial Magnetic Stimulation Scientific evidence in major depression and schizophrenia C.W. Slotema Parnassia Bavo Group The Hague, the Netherlands Faraday s law (1831) Electrical current magnetic
Transcranial Magnetic Stimulation Scientific evidence in major depression and schizophrenia C.W. Slotema Parnassia Bavo Group The Hague, the Netherlands Faraday s law (1831) Electrical current magnetic
2018 Otsuka Pharmaceutical Development & Commercialization, Inc., Rockville, MD Lundbeck, LLC.
 Neuromodulation Techniques: State of the Science Philip G. Janicak, MD Adjunct Professor Department of Psychiatry and Behavioral Sciences, Northwestern University Feinberg School of Medicine Consultant
Neuromodulation Techniques: State of the Science Philip G. Janicak, MD Adjunct Professor Department of Psychiatry and Behavioral Sciences, Northwestern University Feinberg School of Medicine Consultant
The New Clinical Science of ECT
 The New Clinical Science of ECT C. Edward Coffey, MD Professor of Psychiatry & Behavioral Sciences, and of Neurology Baylor College of Medicine Houston, Texas Fellow and Past President, International Society
The New Clinical Science of ECT C. Edward Coffey, MD Professor of Psychiatry & Behavioral Sciences, and of Neurology Baylor College of Medicine Houston, Texas Fellow and Past President, International Society
Setting up a TMS Treatment Program
 Setting up a TMS Treatment Program Alvaro Pascual-Leone, M.D., Ph.D. Professor in Neurology Harvard Medical School Beth Israel Deaconess Medical Center Daniel Cohen, M.D., M.M.Sc. Instructor in Neurology
Setting up a TMS Treatment Program Alvaro Pascual-Leone, M.D., Ph.D. Professor in Neurology Harvard Medical School Beth Israel Deaconess Medical Center Daniel Cohen, M.D., M.M.Sc. Instructor in Neurology
Nonpharmacologic Interventions for Treatment-Resistant Depression. Public Meeting December 9, 2011
 Nonpharmacologic Interventions for Treatment-Resistant Depression Public Meeting December 9, 2011 New England CEPAC Goal: To improve the application of evidence to guide practice and policy in New England
Nonpharmacologic Interventions for Treatment-Resistant Depression Public Meeting December 9, 2011 New England CEPAC Goal: To improve the application of evidence to guide practice and policy in New England
DEEP TMS TREATMENT CONSENT FORM
 DEEP TMS TREATMENT CONSENT FORM My doctor has recommended that I receive treatment with Brainsway Deep TMS. The nature of this treatment, including the benefits and risks that I may experience, has been
DEEP TMS TREATMENT CONSENT FORM My doctor has recommended that I receive treatment with Brainsway Deep TMS. The nature of this treatment, including the benefits and risks that I may experience, has been
Patient Education Brief. NeuroStar TMS Therapies
 Patient Education Brief NeuroStar TMS Therapies Provided by Dr Terrence A. Boyadjis MD 790 E Market Street Suite 245 West Chester, PA 19382 610.738.9576 About Depression Depression is a serious medical
Patient Education Brief NeuroStar TMS Therapies Provided by Dr Terrence A. Boyadjis MD 790 E Market Street Suite 245 West Chester, PA 19382 610.738.9576 About Depression Depression is a serious medical
Supplementary materials
 Supplementary materials Kedzior, K.K., Gellersen, H., Brachetti, A., Berlim, M.T., 2015. Deep transcranial magnetic stimulation (DTMS) in the treatment of major depression: an exploratory systematic review
Supplementary materials Kedzior, K.K., Gellersen, H., Brachetti, A., Berlim, M.T., 2015. Deep transcranial magnetic stimulation (DTMS) in the treatment of major depression: an exploratory systematic review
Transcranial magnetic stimulation in clinical practice
 BJPsych Advances (2016), vol. 22, 373 379 doi: 10.1192/apt.bp.115.015206 Transcranial magnetic stimulation in clinical practice Sheila Hardy, Lorraine Bastick, Alex O Neill-Kerr, Priyadharshini Sabesan,
BJPsych Advances (2016), vol. 22, 373 379 doi: 10.1192/apt.bp.115.015206 Transcranial magnetic stimulation in clinical practice Sheila Hardy, Lorraine Bastick, Alex O Neill-Kerr, Priyadharshini Sabesan,
Efficacy and Safety of Transcranial Magnetic Stimulation in Patients with Depression
 Showa Univ J Med Sci 30 1, 97 106, March 2018 Original Efficacy and Safety of Transcranial Magnetic Stimulation in Patients with Depression Yu KAWAGUCHI 1 and Akira IWANAMI 2 Abstract : Transcranial magnetic
Showa Univ J Med Sci 30 1, 97 106, March 2018 Original Efficacy and Safety of Transcranial Magnetic Stimulation in Patients with Depression Yu KAWAGUCHI 1 and Akira IWANAMI 2 Abstract : Transcranial magnetic
Setting up atms Clinic. Daniel Press, M.D. Assistant Professor in Neurology, Harvard Medical School and Beth Israel Deaconess Medical Center
 Setting up atms Clinic Daniel Press, M.D. Assistant Professor in Neurology, Harvard Medical School and Beth Israel Deaconess Medical Center Contents Safety and training of personnel Equipment Certification
Setting up atms Clinic Daniel Press, M.D. Assistant Professor in Neurology, Harvard Medical School and Beth Israel Deaconess Medical Center Contents Safety and training of personnel Equipment Certification
Are they still doing that?
 Are they still doing that? Why we still give ECT and when to refer Nicol Ferrier BSc (Hons), MD, FRCP(Ed), FRCPsych Emeritus Professor of Psychiatry Newcastle University Rates of prescribing ECT in the
Are they still doing that? Why we still give ECT and when to refer Nicol Ferrier BSc (Hons), MD, FRCP(Ed), FRCPsych Emeritus Professor of Psychiatry Newcastle University Rates of prescribing ECT in the
rtms Versus ECT The Future of Neuromodulation & Brain Stimulation Therapies
 rtms Versus ECT The Future of Neuromodulation & Brain Stimulation Therapies rtms Treatment Equipment rtms Equipment Positioning of coil ECT & rtms Innovative Treatment Options for Patients Only a fraction
rtms Versus ECT The Future of Neuromodulation & Brain Stimulation Therapies rtms Treatment Equipment rtms Equipment Positioning of coil ECT & rtms Innovative Treatment Options for Patients Only a fraction
Transcranial Magnetic Stimulation Therapy for Patients with Refractory Depression: Clinical-Effectiveness and Guidelines
 TITLE: Transcranial Magnetic Stimulation Therapy for Patients with Refractory Depression: Clinical-Effectiveness and Guidelines DATE: 14 April 2009 RESEARCH QUESTIONS: 1. What is the clinical-effectiveness
TITLE: Transcranial Magnetic Stimulation Therapy for Patients with Refractory Depression: Clinical-Effectiveness and Guidelines DATE: 14 April 2009 RESEARCH QUESTIONS: 1. What is the clinical-effectiveness
ª The Author(s) The Canadian Journal of Psychiatry / La Revue Canadienne de Psychiatrie 1-15
 Canadian Psychiatric Association Association des psychiatres du Canada Canadian Network for Mood and Anxiety Treatments (CANMAT) 2016 Clinical Guidelines for the Management of Adults with Major Depressive
Canadian Psychiatric Association Association des psychiatres du Canada Canadian Network for Mood and Anxiety Treatments (CANMAT) 2016 Clinical Guidelines for the Management of Adults with Major Depressive
Objectives. Objectives. A practice review. 02-Nov-16 MAJOR DEPRESSIVE DISORDER: NEW DEVELOPMENTS AND PRACTICAL IMPLICATIONS
 MAJOR DEPRESSIVE DISORDER: NEW DEVELOPMENTS AND PRACTICAL IMPLICATIONS Jon-Paul Khoo What is treatment resistance really? Database review 328 consecutive non-remitted MDD patients referred for private
MAJOR DEPRESSIVE DISORDER: NEW DEVELOPMENTS AND PRACTICAL IMPLICATIONS Jon-Paul Khoo What is treatment resistance really? Database review 328 consecutive non-remitted MDD patients referred for private
Transcranial Magnetic Stimulation for Depression
 Technology Evaluation Center Transcranial Magnetic Stimulation for Depression Assessment Program Volume 26, No. 3 July 2011 Executive Summary Background Depression is a serious psychiatric condition that
Technology Evaluation Center Transcranial Magnetic Stimulation for Depression Assessment Program Volume 26, No. 3 July 2011 Executive Summary Background Depression is a serious psychiatric condition that
Clinical Perspective on Conducting TRD Studies. Hans Eriksson, M.D., Ph.D., M.B.A. Chief Medical Specialist, H. Lundbeck A/S Valby, Denmark
 Clinical Perspective on Conducting TRD Studies Hans Eriksson, M.D., Ph.D., M.B.A. Chief Medical Specialist, H. Lundbeck A/S Valby, Denmark Overview of Presentation Treatment-Resistant Depression (TRD)
Clinical Perspective on Conducting TRD Studies Hans Eriksson, M.D., Ph.D., M.B.A. Chief Medical Specialist, H. Lundbeck A/S Valby, Denmark Overview of Presentation Treatment-Resistant Depression (TRD)
LEASE DO NOT COPY. Setting up atms Clinic
 Setting up atms Clinic Daniel Press, M.D. Assistant Professor in Neurology, Harvard Medical School and Beth Israel Deaconess Medical Center Contents Safety and training of personnel Equipment Certification
Setting up atms Clinic Daniel Press, M.D. Assistant Professor in Neurology, Harvard Medical School and Beth Israel Deaconess Medical Center Contents Safety and training of personnel Equipment Certification
1. Introduction Overview Organization of Executive Summary
 Executive Summary and Discussion of the Vagus Nerve Stimulation (VNS) Therapy Depression Indication Clinical Data (Updated to Include Information from Deficiency Letter Response) Prepared By: Richard L.
Executive Summary and Discussion of the Vagus Nerve Stimulation (VNS) Therapy Depression Indication Clinical Data (Updated to Include Information from Deficiency Letter Response) Prepared By: Richard L.
Transcranial Magnetic Stimulation: Scientific Underpinnings and Practical Applications
 Transcranial Magnetic Stimulation: Scientific Underpinnings and Practical Applications Jon Draud, MS, MD Clinical Professor of Psychiatry University of Tennessee College of Medicine Memphis, Tennessee
Transcranial Magnetic Stimulation: Scientific Underpinnings and Practical Applications Jon Draud, MS, MD Clinical Professor of Psychiatry University of Tennessee College of Medicine Memphis, Tennessee
Brain Stimulation. Berry S. Anderson, PhD, RN Mary Rosedale, PhD, PMHNP-BC, NEA-BC Theresa Kormos, PMHCNS-BC Cindy Brown, BSN, RN
 Brain Stimulation American Psychiatric Nurses Association 24th Annual Conference October 16, 2010 Kentucky International Convention Center Louisville, Kentucky 1 Berry S. Anderson, PhD, RN Mary Rosedale,
Brain Stimulation American Psychiatric Nurses Association 24th Annual Conference October 16, 2010 Kentucky International Convention Center Louisville, Kentucky 1 Berry S. Anderson, PhD, RN Mary Rosedale,
Are you not responding to antidepressants?
 PATIENTS & RELATIVES Are you not responding to antidepressants? TMS therapy might be the solution for you. Does not affect cognitive function No anesthesia Covered by most private providers Outpatient
PATIENTS & RELATIVES Are you not responding to antidepressants? TMS therapy might be the solution for you. Does not affect cognitive function No anesthesia Covered by most private providers Outpatient
Transcranial Magnetic Stimulation:
 Transcranial Magnetic Stimulation: What has experience told us? Jonathan Downar MD PhD FRCPC MRI-Guided rtms Clinic University Health Network www.rtmsclinic.ca Disclosures Research funding: Canadian Institutes
Transcranial Magnetic Stimulation: What has experience told us? Jonathan Downar MD PhD FRCPC MRI-Guided rtms Clinic University Health Network www.rtmsclinic.ca Disclosures Research funding: Canadian Institutes
Charles H. Kellner, MD
 ECT: Learning From Recent Trials Charles H. Kellner, MD Professor, Department of Psychiatry Icahn School of Medicine at Mount Sinai ISCTM Philadelphia, PA 27 September, 2016 Charles H. Kellner, MD Disclosures
ECT: Learning From Recent Trials Charles H. Kellner, MD Professor, Department of Psychiatry Icahn School of Medicine at Mount Sinai ISCTM Philadelphia, PA 27 September, 2016 Charles H. Kellner, MD Disclosures
Review Paper. Introduction
 Review Paper Unilateral and bilateral repetitive transcranial magnetic stimulation for treatment-resistant depression: a meta-analysis of randomized controlled trials over decades Shayan Sehatzadeh, MD,
Review Paper Unilateral and bilateral repetitive transcranial magnetic stimulation for treatment-resistant depression: a meta-analysis of randomized controlled trials over decades Shayan Sehatzadeh, MD,
(U) USSOCOM. (U) Magnetic qeeg guided Resonance Therapy (MeRT)
 United States Special Operations Command (U) USSOCOM MeRT Clinical Trial for Treatment of TBI COL Mark Baggett, Ph.D., US Army USSOCOM Command Psychologist Date: 14 Feb 2018 1 (U) Magnetic qeeg guided
United States Special Operations Command (U) USSOCOM MeRT Clinical Trial for Treatment of TBI COL Mark Baggett, Ph.D., US Army USSOCOM Command Psychologist Date: 14 Feb 2018 1 (U) Magnetic qeeg guided
TRANSCRANIAL MAGNETIC STIMULATION: CLINICAL UPDATE FOR PSYCHIATRIC APPLICATIONS ANNA MAZUR, PH.D. OSU DEPT. OF PSYCHIATRY AND BEHAVIORAL SCIENCES
 TRANSCRANIAL MAGNETIC STIMULATION: CLINICAL UPDATE FOR PSYCHIATRIC APPLICATIONS ANNA MAZUR, PH.D. OSU DEPT. OF PSYCHIATRY AND BEHAVIORAL SCIENCES FINANCIAL DISCLOSURE I have financial relationships to
TRANSCRANIAL MAGNETIC STIMULATION: CLINICAL UPDATE FOR PSYCHIATRIC APPLICATIONS ANNA MAZUR, PH.D. OSU DEPT. OF PSYCHIATRY AND BEHAVIORAL SCIENCES FINANCIAL DISCLOSURE I have financial relationships to
BRAINSWAY DEEP TMS SYSTEM
 510(K) SUMMARY JAN 0 72013 BRAINSWAY DEEP TMS SYSTEM Applicant Name: Company Name: Address: 510(k) Number K122288 Brainsway Ltd. 19 Hartumn St. (Bynet Bldg) Har Hotzvim, Jerusalem, ISRAEL 91451 Tel: +972-2-5813
510(K) SUMMARY JAN 0 72013 BRAINSWAY DEEP TMS SYSTEM Applicant Name: Company Name: Address: 510(k) Number K122288 Brainsway Ltd. 19 Hartumn St. (Bynet Bldg) Har Hotzvim, Jerusalem, ISRAEL 91451 Tel: +972-2-5813
Interventions for Relapsing Depression: TMS, ECT, and Ketamine
 Interventions for Relapsing Depression: TMS, ECT, and Ketamine MDD top cause of disability; suicide only top-10 cause of death currently increasing. 10-15% lifetime prevalence A third of patients do not
Interventions for Relapsing Depression: TMS, ECT, and Ketamine MDD top cause of disability; suicide only top-10 cause of death currently increasing. 10-15% lifetime prevalence A third of patients do not
Page 1 of 18 Volume 01 Page 1
 Response to Vagus Nerve Stimulation (VNS) Therapy Treatment-Resistant Depression (TRD) Indication Not Approvable Letter from the United States Food and Drug Administration (FDA), Dated August 11, 2004
Response to Vagus Nerve Stimulation (VNS) Therapy Treatment-Resistant Depression (TRD) Indication Not Approvable Letter from the United States Food and Drug Administration (FDA), Dated August 11, 2004
Interventions for Relapsing Depression: TMS, ECT, and Ketamine
 Interventions for Relapsing Depression: TMS, ECT, and Ketamine MDD top cause of disability; suicide only top-10 cause of death currently increasing. 10-15% lifetime prevalence A third of patients do not
Interventions for Relapsing Depression: TMS, ECT, and Ketamine MDD top cause of disability; suicide only top-10 cause of death currently increasing. 10-15% lifetime prevalence A third of patients do not
TMS: Full Board or Expedited?
 TMS: Full Board or Expedited? Transcranial Magnetic Stimulation: - Neurostimulation or neuromodulation technique based on the principle of electro-magnetic induction of an electric field in the brain.
TMS: Full Board or Expedited? Transcranial Magnetic Stimulation: - Neurostimulation or neuromodulation technique based on the principle of electro-magnetic induction of an electric field in the brain.
MEDICAL POLICY SUBJECT: PHOTOTHERAPY FOR THE TREATMENT OF SEASONAL AFFECTIVE DISORDER. POLICY NUMBER: CATEGORY: Behavioral Health
 MEDICAL POLICY SUBJECT: PHOTOTHERAPY FOR THE ARCHIVED: 09/16/04-12/07/06 PAGE: 1 OF: 5 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial
MEDICAL POLICY SUBJECT: PHOTOTHERAPY FOR THE ARCHIVED: 09/16/04-12/07/06 PAGE: 1 OF: 5 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial
NOW I M A NEUROSTAR. Let Your Best Self Shine
 NOW I M A NEUROSTAR. Let Your Best Self Shine It s Time for a New You NeuroStar Advanced Therapy is an in-office treatment that requires no anesthesia or sedation. You are awake and alert during treatment
NOW I M A NEUROSTAR. Let Your Best Self Shine It s Time for a New You NeuroStar Advanced Therapy is an in-office treatment that requires no anesthesia or sedation. You are awake and alert during treatment
Short-term efficacy of repetitive transcranial magnetic stimulation (rtms) in depressionreanalysis of data from meta-analyses up to 2010
 Kedzior and Reitz BMC Psychology 2014, 2:39 RESEARCH ARTICLE Open Access Short-term efficacy of repetitive transcranial magnetic stimulation (rtms) in depressionreanalysis of data from meta-analyses up
Kedzior and Reitz BMC Psychology 2014, 2:39 RESEARCH ARTICLE Open Access Short-term efficacy of repetitive transcranial magnetic stimulation (rtms) in depressionreanalysis of data from meta-analyses up
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: vagus_nerve_stimulation 6/1998 5/2017 5/2018 5/2017 Description of Procedure or Service Stimulation of the
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: vagus_nerve_stimulation 6/1998 5/2017 5/2018 5/2017 Description of Procedure or Service Stimulation of the
IEHP UM Subcommittee Approved Authorization Guidelines Electroconvulsive Therapy- ECT
 Electroconvulsive Therapy- ECT Policy: IEHP considers ECT medically necessary for members with the following disorders: 1. Unipolar and bipolar depression. 2. Bipolar mania. 3. Psychotic disorders including
Electroconvulsive Therapy- ECT Policy: IEHP considers ECT medically necessary for members with the following disorders: 1. Unipolar and bipolar depression. 2. Bipolar mania. 3. Psychotic disorders including
Study No.: Title: Rationale: Phase: Study Period: Study Design: Centers: Indication: Treatment: Objectives: Primary Outcome/Efficacy Variable:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Is There Evidence for Effectiveness of Transcranial Magnetic Stimulation in the Treatment of Psychiatric Disorders?
 [EVIDENCE-BASED PHARMACOLOGY] by BIJU BASIL, MD, DPM; JAMAL MAHMUD, MD, DPM; MAJU MATHEWS, MD, MRCPsych, DIP PSYCH; CARLOS RODRIGUEZ, MD; and BABATUNDE ADETUNJI, MD, DPM Dr. Basil is a resident, Dr. Mahmud
[EVIDENCE-BASED PHARMACOLOGY] by BIJU BASIL, MD, DPM; JAMAL MAHMUD, MD, DPM; MAJU MATHEWS, MD, MRCPsych, DIP PSYCH; CARLOS RODRIGUEZ, MD; and BABATUNDE ADETUNJI, MD, DPM Dr. Basil is a resident, Dr. Mahmud
Transcranial magnetic stimulation for the treatment of major depression
 Neuropsychiatric Disease and Treatment open access to scientific and medical research Open Access Full Text Article Review Transcranial magnetic stimulation for the treatment of major depression Philip
Neuropsychiatric Disease and Treatment open access to scientific and medical research Open Access Full Text Article Review Transcranial magnetic stimulation for the treatment of major depression Philip
HCT Medical Policy. Bronchial Thermoplasty. Policy # HCT113 Current Effective Date: 05/24/2016. Policy Statement. Overview
 HCT Medical Policy Bronchial Thermoplasty Policy # HCT113 Current Effective Date: 05/24/2016 Medical Policies are developed by HealthyCT to assist in administering plan benefits and constitute neither
HCT Medical Policy Bronchial Thermoplasty Policy # HCT113 Current Effective Date: 05/24/2016 Medical Policies are developed by HealthyCT to assist in administering plan benefits and constitute neither
 http://web.uct.ac.za/depts/psychology/postgraduate/hons2008projects/sarah.drowley.pdf ftp://ftp.psy.gla.ac.uk/pub/gregor/tmseeg4marine/ort%20clinneurophysiol%202009.pdf J Nerv Ment Dis. 2010 Sep;198(9):679-81.
http://web.uct.ac.za/depts/psychology/postgraduate/hons2008projects/sarah.drowley.pdf ftp://ftp.psy.gla.ac.uk/pub/gregor/tmseeg4marine/ort%20clinneurophysiol%202009.pdf J Nerv Ment Dis. 2010 Sep;198(9):679-81.
Review. M. Gross 1, L. Nakamura 1, A. Pascual-Leone 2 F. Fregni 2 1 Department of Psychiatry, University of S¼o Paulo, S¼o
 Acta Psychiatr Scand 2007: 116: 165 173 All rights reserved DOI: 10.1111/j.1600-0447.2007.01049.x Copyright Ó 2007 The Authors Journal Compilation Ó 2007 Blackwell Munksgaard ACTA PSYCHIATRICA SCANDINAVICA
Acta Psychiatr Scand 2007: 116: 165 173 All rights reserved DOI: 10.1111/j.1600-0447.2007.01049.x Copyright Ó 2007 The Authors Journal Compilation Ó 2007 Blackwell Munksgaard ACTA PSYCHIATRICA SCANDINAVICA
Curriculum Vitae, Michael J. Downing, M.D.
 CONTACT INFORMATION: Site Selection and Information: Bobbie Theodore, Alliance Director Tel. (916) 939-6696 Fax (208) 575-3169 Email: clinicaltrials@alliancesites.com PROFESSIONAL AFFILIATIONS: Michael
CONTACT INFORMATION: Site Selection and Information: Bobbie Theodore, Alliance Director Tel. (916) 939-6696 Fax (208) 575-3169 Email: clinicaltrials@alliancesites.com PROFESSIONAL AFFILIATIONS: Michael
Therapeutic Neuromodulation: Overview of a Novel Treatment Platform
 Therapeutic Neuromodulation: Overview of a Novel Treatment Platform David G. Brock, MD; and Mark A. Demitrack, MD Shutterstock David G. Brock, MD, is Medical Director, Neuronetics, Inc. Mark A. Demitrack,
Therapeutic Neuromodulation: Overview of a Novel Treatment Platform David G. Brock, MD; and Mark A. Demitrack, MD Shutterstock David G. Brock, MD, is Medical Director, Neuronetics, Inc. Mark A. Demitrack,
Daniel Michael Blumberger
 A Randomized Double-Blind Sham-Controlled Comparison of Unilateral and Bilateral Repetitive Transcranial Magnetic Stimulation for Treatment- Resistant Major Depression By Daniel Michael Blumberger A thesis
A Randomized Double-Blind Sham-Controlled Comparison of Unilateral and Bilateral Repetitive Transcranial Magnetic Stimulation for Treatment- Resistant Major Depression By Daniel Michael Blumberger A thesis
Clinical Policy Title: Ketamine for treatment-resistant depression
 Clinical Policy Title: Ketamine for treatment-resistant depression Clinical Policy Number: 00.02.13 Effective Date: January 1, 2016 Initial Review Date: August 19, 2015 Most Recent Review Date: January
Clinical Policy Title: Ketamine for treatment-resistant depression Clinical Policy Number: 00.02.13 Effective Date: January 1, 2016 Initial Review Date: August 19, 2015 Most Recent Review Date: January
Transcranial magnetic stimulation has become an increasingly
 Within-Session Mood Changes From TMS in Depressed Patients Tobias Dang, M.D. David H. Avery, M.D. Joan Russo, Ph.D. Transcranial magnetic stimulation has become an increasingly well studied tool in neuropsychiatry.
Within-Session Mood Changes From TMS in Depressed Patients Tobias Dang, M.D. David H. Avery, M.D. Joan Russo, Ph.D. Transcranial magnetic stimulation has become an increasingly well studied tool in neuropsychiatry.
Biological treatments: Electroconvulsive Therapy, Transcranial Magnetic Stimulation, Light therapy
 Biological treatments: Electroconvulsive Therapy, Transcranial Magnetic Stimulation, Light therapy Basic trainee talk 23 May 2007 ECT When is ECT used. Depression. Severe, melancholic. Resistant to treatment.
Biological treatments: Electroconvulsive Therapy, Transcranial Magnetic Stimulation, Light therapy Basic trainee talk 23 May 2007 ECT When is ECT used. Depression. Severe, melancholic. Resistant to treatment.
re-emerging role The Recent developments have revived interest in brain stimulation for difficult-to-treat patients
 The re-emerging role Recent developments have revived interest in brain stimulation for difficult-to-treat patients GOMAN MEDICAL ILLUSTRATIONS FOR CURRENT PSYCHIATRY of therapeutic Philip G. Janicak,
The re-emerging role Recent developments have revived interest in brain stimulation for difficult-to-treat patients GOMAN MEDICAL ILLUSTRATIONS FOR CURRENT PSYCHIATRY of therapeutic Philip G. Janicak,
PSYCHOPHARMACOLOGY BULLETIN: Vol. 42 No. 2 5
 DEMITRACK_MWM_549.QXP 7/14/9 1:46 PM Page 5 ORIGINAL RESEARCH Key Words: major depression, treatment resistance, transcranial magnetic stimulation, TMS, antidepressant, efficacy, safety, randomized clinical
DEMITRACK_MWM_549.QXP 7/14/9 1:46 PM Page 5 ORIGINAL RESEARCH Key Words: major depression, treatment resistance, transcranial magnetic stimulation, TMS, antidepressant, efficacy, safety, randomized clinical
Some newer, investigational approaches to treating refractory major depression are being used.
 CREATED EXCLUSIVELY FOR FINANCIAL PROFESSIONALS Rx FOR SUCCESS Depression and Anxiety Disorders Mood and anxiety disorders are common, and the mortality risk is due primarily to suicide, cardiovascular
CREATED EXCLUSIVELY FOR FINANCIAL PROFESSIONALS Rx FOR SUCCESS Depression and Anxiety Disorders Mood and anxiety disorders are common, and the mortality risk is due primarily to suicide, cardiovascular
NEW DIRECTIONS BEHAVIORAL HEALTH, L.L.C.
 NEW DIRECTIONS BEHAVIORAL HEALTH, L.L.C. Medical Policy Repetitive Transcranial Magnetic Stimulation for Treatment of Major Depression Original Effective date: 10/13/2014 Reviewed: 3/2015, 11/2015, 11/2016,
NEW DIRECTIONS BEHAVIORAL HEALTH, L.L.C. Medical Policy Repetitive Transcranial Magnetic Stimulation for Treatment of Major Depression Original Effective date: 10/13/2014 Reviewed: 3/2015, 11/2015, 11/2016,
TITLE: Delivery of Electroconvulsive Therapy in Non-Hospital Settings: A Review of the Safety and Guidelines
 TITLE: Delivery of Electroconvulsive Therapy in Non-Hospital Settings: A Review of the Safety and Guidelines DATE: 08 May 2014 CONTEXT AND POLICY ISSUES Electroconvulsive therapy (ECT) is a treatment that
TITLE: Delivery of Electroconvulsive Therapy in Non-Hospital Settings: A Review of the Safety and Guidelines DATE: 08 May 2014 CONTEXT AND POLICY ISSUES Electroconvulsive therapy (ECT) is a treatment that
Bringing. to patients with depression
 Bringing to patients with depression Hope is knowing remission from depression is possible Depression is a serious illness that affects approximately 350 million people worldwide. 1 While medications may
Bringing to patients with depression Hope is knowing remission from depression is possible Depression is a serious illness that affects approximately 350 million people worldwide. 1 While medications may
Copyright 2002 American Academy of Neurology. Volume 58(8) 23 April 2002 pp
 Copyright 2002 American Academy of Neurology Volume 58(8) 23 April 2002 pp 1288-1290 Improved executive functioning following repetitive transcranial magnetic stimulation [Brief Communications] Moser,
Copyright 2002 American Academy of Neurology Volume 58(8) 23 April 2002 pp 1288-1290 Improved executive functioning following repetitive transcranial magnetic stimulation [Brief Communications] Moser,
obsessive-compulsive disorder; OCD
 9(1), 50 56, 2017 OCD OCD DBSECT tdcsrtms DBS ECT 6 tdcs rtms obsessive-compulsive disorder; OCD 2013 OCD 2012 deep brain stimulation; DBS electroconvulsive therapy; ECT transcranial direct current stimulation;
9(1), 50 56, 2017 OCD OCD DBSECT tdcsrtms DBS ECT 6 tdcs rtms obsessive-compulsive disorder; OCD 2013 OCD 2012 deep brain stimulation; DBS electroconvulsive therapy; ECT transcranial direct current stimulation;
Accepted Manuscript. Peter Fettes, Sarah Peters, Peter Giacobbe, Jonathan Downar
 Accepted Manuscript Neural correlates of successful orbitofrontal 1 Hz rtms following unsuccessful dorsolateral and dorsomedial prefrontal rtms in major depression: A case report Peter Fettes, Sarah Peters,
Accepted Manuscript Neural correlates of successful orbitofrontal 1 Hz rtms following unsuccessful dorsolateral and dorsomedial prefrontal rtms in major depression: A case report Peter Fettes, Sarah Peters,
HHS Public Access Author manuscript J Neuropsychiatry Clin Neurosci. Author manuscript; available in PMC 2017 September 11.
 HHS Public Access Author manuscript Published in final edited form as: J Neuropsychiatry Clin Neurosci. 2017 ; 29(2): 179 182. doi:10.1176/appi.neuropsych.16100181. Initial Response to Transcranial Magnetic
HHS Public Access Author manuscript Published in final edited form as: J Neuropsychiatry Clin Neurosci. 2017 ; 29(2): 179 182. doi:10.1176/appi.neuropsych.16100181. Initial Response to Transcranial Magnetic
Transcranial Magnetic Stimulation
 Medical Coverage Policy Transcranial Magnetic Stimulation Table of Contents Effective Date... 2/15/2018 Next Review Date... 2/15/2019 Coverage Policy Number... 0383 Related Coverage Resources Coverage
Medical Coverage Policy Transcranial Magnetic Stimulation Table of Contents Effective Date... 2/15/2018 Next Review Date... 2/15/2019 Coverage Policy Number... 0383 Related Coverage Resources Coverage
Introduction to TMS Transcranial Magnetic Stimulation
 Introduction to TMS Transcranial Magnetic Stimulation Lisa Koski, PhD, Clin Psy TMS Neurorehabilitation Lab Royal Victoria Hospital 2009-12-14 BIC Seminar, MNI Overview History, basic principles, instrumentation
Introduction to TMS Transcranial Magnetic Stimulation Lisa Koski, PhD, Clin Psy TMS Neurorehabilitation Lab Royal Victoria Hospital 2009-12-14 BIC Seminar, MNI Overview History, basic principles, instrumentation
TITLE: Transcranial Magnetic Stimulation for the Treatment of Adults with PTSD, GAD, or Depression: A Review of Clinical Effectiveness and Guidelines
 TITLE: Transcranial Magnetic Stimulation for the Treatment of Adults with PTSD, GAD, or Depression: A Review of Clinical Effectiveness and Guidelines DATE: 31 October 2014 CONTEXT AND POLICY ISSUES Post-traumatic
TITLE: Transcranial Magnetic Stimulation for the Treatment of Adults with PTSD, GAD, or Depression: A Review of Clinical Effectiveness and Guidelines DATE: 31 October 2014 CONTEXT AND POLICY ISSUES Post-traumatic
Treating treatment resistant depression
 Treating treatment resistant depression These slides are the intellectual property of Ian Anderson and must not be reproduced Ian Anderson Neuroscience and Psychiatry Unit University of Manchester and
Treating treatment resistant depression These slides are the intellectual property of Ian Anderson and must not be reproduced Ian Anderson Neuroscience and Psychiatry Unit University of Manchester and
Biofeedback as a Treatment of Headache
 Biofeedback as a Treatment of Headache Policy Number: 2.01.29 Last Review: 7/2018 Origination: 7/2008 Next Review: 7/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) may provide coverage
Biofeedback as a Treatment of Headache Policy Number: 2.01.29 Last Review: 7/2018 Origination: 7/2008 Next Review: 7/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) may provide coverage
Transcranial Direct-Current Stimulation
 Introduction (tdcs) is a non-invasive form of brain stimulation similar to transcranial magnetic stimulation, but instead of using magnets, it uses a lowintensity, constant current applied through scalp
Introduction (tdcs) is a non-invasive form of brain stimulation similar to transcranial magnetic stimulation, but instead of using magnets, it uses a lowintensity, constant current applied through scalp
