Uhl et al. Radiation Oncology 2014, 9:100
|
|
|
- Buddy Ball
- 5 years ago
- Views:
Transcription
1 Uhl et al. Radiation Oncology 2014, 9:100 STUDY PROTOCOL Open Access Randomized phase II trial of hypofractionated proton versus carbon ion radiation therapy in patients with sacrococcygeal chordoma-the ISAC trial protocol Matthias Uhl 1,2*, Lutz Edler 3, Alexandra D Jensen 1,2, Gregor Habl 1, Jan Oelmann 1, Falk Röder 1, Oliver Jäckel 2,3, Jürgen Debus 1,2,3 and Klaus Herfarth 1,2 Abstract Background: Chordomas are relatively rare lesions of the bones. About 30% occur in the sacrococcygeal region. Surgical resection is still the standard treatment. Due to the size, proximity to neurovascular structures and the complex anatomy of the pelvis, a complete resection with adequate safety margin is difficult to perform. A radical resection with safety margins often leads to the loss of bladder and rectal function as well as motoric/sensoric dysfunction. The recurrence rate after surgery alone is comparatively high, such that adjuvant radiation therapy is very important for improving local control rates. Proton therapy is still the international standard in the treatment of chordomas. High-LET beams such as carbon ions theoretically offer biologic advantages in slow-growing tumors. Data of a Japanese study of patients with unresectable sacral chordoma showed comparable high control rates after hypofractionated carbon ion therapy only. Methods and design: This clinical study is a prospective randomized, monocentric phase II trial. Patients with histologically confirmed sacrococcygeal chordoma will be randomized to either proton or carbon ion radiation therapy stratified regarding the clinical target volume. Target volume delineation will be carried out based on CT and MRI data. In each arm the PTV will receive 64 GyE in 16 fractions. The primary objective of this trial is safety and feasibility of hypofractionated irradiation in patients with sacrococygeal chordoma using protons or carbon ions in raster scan technique for primary or additive treatment after R2 resection. The evaluation is therefore based on the proportion of treatments without Grade 3 5 toxicity (CTCAE, version 4.0) up to 12 months after treatment and/or discontinuation of the treatment for any reason as primary endpoint. Local-progression free survival, overall survival and quality of life will be analyzed as secondary end points. Discussion: The aim of this study is to confirm the toxicity results of the Japanese data in raster scan technique and to compare it with the toxicity analysis of proton therapy given in the same fractionation. Using this data, a further randomized phase III trial is planned, comparing hypofractionated proton and carbon ion irradiation. Trial registration: ClinicalTrials.gov Identifier: NCT Keywords: Sacral chordoma, Chordoma, Carbon ion therapy, Proton therapy, Irradiation, Randomized trial, Hypofractionation, Heavy ion therapy * Correspondence: Matthias.Uhl@med.uni-heidelberg.de 1 Department of Radiation Oncology, University of Heidelberg, Im Neuenheimer Feld 400, Heidelberg, Germany 2 Heidelberg Ion Therapy Center (HIT), Im Neuenheimer Feld 450, Heidelberg, Germany Full list of author information is available at the end of the article 2014 Uhl et al.; licensee BioMed Central Ltd. This is an Open Access article distributed under the terms of the Creative Commons Attribution License ( which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly credited. The Creative Commons Public Domain Dedication waiver ( applies to the data made available in this article, unless otherwise stated.
2 Uhl et al. Radiation Oncology 2014, 9:100 Page 2 of 8 Introduction The advantage of radiation therapy with particles is based on the particular physical and biological characteristics of ions. Protons and carbon ions as compared to photons lead to an improved conformal dose distribution, which allows a better sparing of surrounding tissue and concurrent dose escalation in the tumor. Carbon ions seem to have an additional biological advantage due to their higher biological effectiveness in contrast to protons [1-3]. Complex double-strand breaks of the DNA seem to be the reason [4,5]. Therefore, heavy ions provide a promising treatment option for tumors with lower radiosensitivity and critical location. Increased cure rates and low toxicity of a particle therapy are the expected results in tumors with low alpha/beta values. Chordomas, as a typical example, are rare malignant tumors (1-4% of all malignant bone tumors). This slow growing tumor arises from embryonic remnants of the chorda dorsalis. The incidence rate is 0.1/1,000,000 [6]. Historically, a higher incidence of sacral chordomas than skull base chordomas was assumed [7]. However, published SEER data of 400 patients with chordoma show an equal distribution with 32% on the skull base, 32.8% spinal and 29.2% in the sacral region [6]. The histological classification is divided into three types: conventional (most common type), chondroid and dedifferentiated [7-9]. The quality of surgical margin is the most important factor for local control and survival [10-12]. An adequate safety margin can only be achieved in about 50% of the patients with sacrococcygeal chordomas [11-13]. Therefore, after R1/R2 resection the local recurrence rate is about 100% without any additional therapy. Fuchs et al. could show a significantly different time of local control between patients with radical resection and subtotal resection [11]. Nevertheless the data of Park et al. indicate a large difference in local failure rate between patients irradiated for primary versus recurrent sacral chordomas after surgery. Hence, an adjuvant high-dose radiotherapy for acceptable control rates is required after complete resection [14]. High conformal techniques help to achieve a safe dose escalation. As mentioned above, due to their physical properties ions provide the best conditions. Proton therapy is the current international standard in irradiation of skull base chordomas. The results of the Loma Linda University Medical Centre, the Massachusetts General Hospital in Boston, and the PSI in Villingen (Switzerland) show excellent control rates for proton therapy [15-17]. In the GSI in Darmstadt, we irradiated with carbon ions by now about 300 patients with skull base chordoma/ chondrosarcoma. Published data of the first 96 patients that were treated at the GSI displayed 3- and 5-year local tumor control rates (LC) of 81% and 70% with overall survival (OS) rates of 92% and 89%, respectively [18]. Currently, there is a randomized phase III trial under recruitment, testing protons against heavy ions in patients with skull base chordoma in our department [19]. In addition, over 70 patients with sacral chordoma were treated with carbon ion in our heavy ion therapy center (HIT) between 2010 and Furthermore, in chordomas of the sacral region surgical resection is still the standard therapy and the extent of surgical resection is important for the local disease free interval [11]. Due to the size, proximity to neurovascular structures and the complex anatomy of the pelvis a complete resection with adequate safety margin is often difficult to realize. A radical resection, especially above S2, often leads to urinary and bowel incontinence as well as motoric/sensoric dysfunctions [20,21]. An adjuvant/additive treatment with charged particles can increase the local control rate after surgery [14,22,23]. Radiobiological considerations lead to low alpha/beta value for chordoma (the tissue-specific constant that indicates the sensitivity of the tissue for the probability of late toxicity by increasing the single dose). The alpha/beta value for chordoma cells (2 Gy) [24] is significantly lower for other organs at risk (bladder alpha/beta 4.0 Gy; rectum alpha/beta 3.9 Gy). Thereby, a higher single dose can be used, treating the chordoma cells more effectively without increasing the risk of side effects. A Japanese trial with primary carbon ions irradiation without previous surgery confirmed this radiobiological model [25]. The data show an excellent control rate after primary radiotherapy with carbon ions in patients with inoperable sacral chordoma. 95 patients were treated with 16 fractions carbon ions over 4 weeks with a total dose of 52.8 GyE up to 73.6 GyE (70.4 Gy median TD). The fraction dose therefore was 3.3 GyE up to 4.6 GyE (4.4 Gy median SD) applied four times a week. At 5 years, the OS rate and LC rate were 86% and 88%, respectively. The rate of acute/late skin toxicity grade 3 and higher was 6.3%. Only one patient (1%) showed transient rectal bleeding 20 months after therapy (Grade II). The published study with ion irradiation of sacral chordoma was conducted using a passive beam modulation. A further development of the ion irradiation is the active beam delivery using raster-scanning method. The advantage of this method is especially the lower neutron production when used in our department. As a result of the higher neutron production the passive beam modulation shows no advantage over IMRT regarding secondary cancer risk [26]. Until now there have been no published data of hypofractionated irradiation with protons in sacral chordoma. Therefore, this is the first randomized Phase II trial protocol with hypofractionated carbon ion versus proton treatment in patients with sacrococcygeal chordoma in a single institution open for recruitment since January 2013.
3 Uhl et al. Radiation Oncology 2014, 9:100 Page 3 of 8 Methods and design Primary objectives/endpoints of this study The primary objective of this trial is the evaluation of safety and feasibility of hypofractionated irradiation in patients with sacrococygeal chordoma using ions (protons or carbon ions) in raster scan technique for primary or additive treatment after R2 resection. The evaluation is based on as primary endpoint defined as the proportion of patients treated without Grade 3 5 toxicity (NCI-CTC-AE) up to 12 months after treatment and/or without discontinuation of the treatment for any reason. Secondary objectives Assessment of local progression free survival (LPFS) is determined from start of treatment until local progression in imaging (>10% size increase). Further objectives are overall survival (OS) from start of treatment until death or censoring and Quality of life (QoL) using the EORTC-QLQ30 questionnaire. Study design The study is a parallel group prospective clinical phase II trial of patients with sacrococcygeal chordoma, randomized to one of the two treatment arms (arm A: proton therapy, arm B: carbon ion therapy). A total dose of 64 GyE in 16 fractions to the PTV (see target definition below) will be given in arm A using protons and in arm B using carbon ions. The accrual period of this trial will take approximately two years with a follow up time of 12 months for each patient. Patients matching the eligibility criteria and willing to participate with informed consent are registered (Figure 1). Inclusion criteria Histological confirmation of sacrococcygeal chordoma. Karnofsky performance status 70%. Patients age years. Macroscopic tumor (MRI). Written informed consent. Exclusion criteria Distant metastasis (M+). Lack of macroscopic tumor. Tumor extension in craniocaudal direction >16 cm due to technical limitation. Metal implants at the level of the tumor that could influence the treatment planning. Inability of the patient to lie quiet for at least 20 minutes (e.g. due to pain). Prior radiotherapy of the pelvic region. Simultaneous participation in another trial that could influence the results of the study. Active medical implants without treatment approval at the time of ion irradiation (e.g., cardiac pacemaker, defibrillator). Randomization Randomization will be performed regarding treatment arms A (protons) and B (carbon ions) stratified by the volume of CTV (>or 1 L). Figure 1 Workflow.
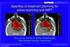
4 Uhl et al. Radiation Oncology 2014, 9:100 Page 4 of 8 Treatment planning Examinations for treatment planning consist of a CT scan (3 mm slice thickness) in treatment position and a MRI for 3D image correlation. The delineation of the chordoma requires a T1 weighted post gadolinium and T2 stir MRI. Target volume Gross Tumor Volume (GTV) includes the gross tumor based on CT and MRI imaging. The Clinical Target Volume (CTV) is defined as GTV plus surrounding areas at risk for containing microscopic disease. The CTV includes the GTV and, after partial resection, the tumour bed and surgical access path with a margin of 2 cm in bone and soft tissue. The CTV margins may be smaller if the GTV is adjacent to the critical normal organs like small bowel or rectum. The Planning Target Volume (PTV) includes the CTV with an additional margin of 5 mm in anterior-posterior direction and 7 mm in lateral direction to compensate set-up variability. The overlap of PTV and rectum is defined as PTV-Rectum. Proton/carbon ion therapy Treatment planning is realized using a treatment planning system (TPS Siemens) that enables conventional and biological optimization. After inverse planning proton and carbon ion treatment is given in active beam application (raster scanning method). Dose prescription 95% of the PTV should obtain the dose of 64 GyE in 16 fractions (5 6 fractions per week). The equivalent photon dose in 2 Gy/fraction (ED2) is 96 Gy, calculated for 2 Gy single dose (SD) and α/β value of 2. Critical normal structure constraints Rectum, bladder and bowel are defined as organs at risk. Rectum is defined therefore ranging from the anus to the recto sigmoid junction, the bowel as the remaining intestine in the pelvis without rectum. The maximum dose to the PTV-rectum is 57.6 GyE (90% isodose). The ED2(2GySD,α/β = 3.9 Gy) is 73.2 Gy. One third of the circumference should be below the 35% isodose (ED2: 20 Gy). The maximum dose for the other parts of the intestine is 51.2 GyE (80% isodose, ED2: 61.4 Gy). This dose may only appear in a small volume. The QUANTEC criteria for the rectum and the other parts of the intestine have to be observed (V50 < 50%, V60 < 35%, V65 < 25%, V70 < 20% and V75 < 15%) (Figure 2). The maximum dose to the cauda equina is 60 Gy (ED2). Assessment of efficacy parameters Toxicity and safety To evaluate the toxicity, this study will use the International Common Terminology Criteria for Adverse Events (CTCAE) version 4.0 for toxicity and adverse event reporting. Safety and toxicity of the study treatment will Figure 2 Dose distribution and DVH of a patient treated with 64GyE carbon ion therapy within the ISAC trial. a) dose distribution and b) dose volume histogram (DVH) of a patient within the trial.
5 Uhl et al. Radiation Oncology 2014, 9:100 Page 5 of 8 be evaluated by clinical examination as well as imaging studies (MRI or CT). At enrollment, each patient will be asked for his medical history and pass through a basic clinical examination with regard to already existing problems. During radiation therapy the patient is monitored continuously. Once a week, a meeting with the investigator will take place in order to record the adverse effects. At the end of the treatment and in the follow up visits the clinical symptoms and toxicity will be documented again. Follow up visits are planned for 6 8 weeks,4months, 8 months and 12 months after irradiation. A safe feasibility exists, if no grade 3 or higher toxicity has occurred (including toxicity-related death, Grade 5) from start of radiation until 12 months after the end of therapy, and if the therapy was not canceled due to any toxicity (grade 1 4). It will be evaluated be the respective portion of patients denoted SFR (safe feasibility rate). Quality of life The quality of life (QoL) is detected using the EORTC QLQ-C30 questionnaires that are filled in by the patient before treatment, at the end of treatment and 12 months after irradiation. The evaluation of the questionnaires is carried out for the individual patients after the complete collection of all data. To determine a change in QoL it will be compared with the pretherapeutic QoL for each patient in both arms. QoL will be compared between the two arms of the study after end of radiation and at end of study for the patients. For the analysis of the EORTC questionnaires the EORTC QLQ-C30 Scoring Manual will be used. Local progression free survival (LPFS) The effectiveness of treatment is examined through MRI follow-ups. A progressive disease (PD) is achieved if the tumor size increases 10% or more, measured as the longest dimension in cranial-caudal, anterior-posterior and lateral direction. This definition of PD is based on the observation of a temporary tumor swelling after the particle therapy. A new tumor nodule and a tumor growth >10% in one direction (for example toward the gluteal muscles) will also be evaluated as PD. Since chordomas usually have a very low proliferation rate (Ki 67: <5%), an assessment by RECIST criteria is too insensitive for detecting a PD. Overall survival (OS) Overall survival is a secondary endpoint of the study. The duration of survival is the time interval between beginning of radiotherapy and the date of death due to any cause or to loss to follow-up (censored observation). Patients not reported dead or lost to follow-up will be censored at the date of the last follow-up examination (Figure 3). Statistical consideration and analysis With the present study, basic data are obtained for hypofractionated carbon ion radiation and proton radiation in sacral chordoma patients to be used for planning a confirmative randomized phase III trial comparing proton and carbon ion irradiation. At present, hypofractionated carbon ion radiation is considered as the standard, supported by data already existing in Japan. However, the treatment in Japan was performed without using the raster-scanning method. Nevertheless, these data can serve as historical control for the planning of this pilot study and provide the framework for the assessment of safety and feasibility of the proposed study treatments using the SFR as endpoint. Each of the planned study treatments (Arm A: protons or arm B: carbon ions) will be tested separately for this endpoint in a non-inferiority study design in comparison with that (historical) control. The comparative evaluation of favorable toxicity is assessed in an exploratory comparison by comparing the SFR of both arms. With these results, a further trial is then planned to evaluate the efficacy and safety of hypofractionated irradiation with carbon ions or protons with active beam delivery of sacral chordoma. Consequently, the following two questions will be considered in preparation for the subsequent confirmatory study: a) Is the toxicity of carbon ion irradiation (Arm B) non-inferior to the current standard? b) Is the toxicity of the proton irradiation (arm A) non-inferior to the current standard? The number of patients to be recruited is calculated separately for each arm with an assumed standard toxicity rate of 7% (Grade 3 5 NCI-CTC AE Tox, including premature termination). Thus, the safety rate is set to 93% accounting for the Japanese results, where a toxicity of grade 3 or higher in 6.3% of patients was found [25]. Identical non-inferiority limits for toxicity and statistical error probabilities (type I error = type II error = 10%) are selected for each of the two arms. Randomization is used to obtain a balanced patient population in both groups and based on permuted block randomization stratified for the CTV size (> or 1 L). Comparison between the two arms will be carried out as a secondary comparison. The non-inferiority of each of the two arms compared to the standard is assessed in a one-sided hypothesis test: H0: SDR <93% versus H1: SDR > = 93% - DELTA. For the proposed pilot trial a non-inferiority limit (DELTA) of 13% is assumed. To test the efficacy and safety of the treatment, 45 evaluable patients per arm are required to reach a power of 90% (type II error = 10%) at a significance level 10% (type I error). Non-inferiority is denied, if safe feasibility is possible in less than 40 patients (i.e. if more than 5 of the
6 Uhl et al. Radiation Oncology 2014, 9:100 Page 6 of 8 Figure 3 Examinations during the trial in tabular view. RT = radiothrapy. 45 (90%) evaluable patients show toxicity. Each of the two tests is to be tested with a power of 90% (power = 90%, beta = 0.1) in a one-sample binomial test. Confidence intervals of the SFR are calculated as describing parameters. With an estimated drop out rate of approximately 10%, 50 patients should be included in each arm in order to obtain 45 evaluable patients. This sample size calculation is based on the PASS software of 2005 and the method of Blackwelder (1982) for non-inferiority trials [27]. The secondary endpoints OS and LPFS will be analyzed via descriptive methods of censored survival times (Kaplan Meier method). Regular study end The estimated accrual period is 24 months. The regular end of the treatment period for each patient is three weeks after initiation of radiation therapy (after 16 fractions). The regular end of study participation for each patient is after a follow up period of 12 months. Prematurely study termination Reasons for premature termination of the entire study are unacceptable risks and toxicities as specified by the Safety Board as occurrence of a toxicity of grade 5, of 2 consecutive grade 4 toxicities or 5 consecutive grade 3 toxicities judged to be definitively associated with study therapy. Other reasons are new scientific findings during the period of study that require a different treatment. Individual reasons for premature study termination are serious events or a patient s request. Data safety monitoring board (DSMB) An independent Data and Safety Monitoring Board (DSMB) will monitor the recruitment, the reported adverse events and the data quality. Based on its review thedsmbwillprovidetheprincipalinvestigator(pi) with recommendations regarding trial modification, continuation or termination. Data collection and management All patient related data are collected pseudonymously. An individual patient number characterizes each patient. The data collection is based on case report forms (documentation forms/case report forms). The originals of all documents are kept in the study center. According to the 13 of the German GCP-Regulation, all important trial documents will be archived for at least 15 years after the end of the ISAC trial. According to the 28c of the German X-Ray Regulation (RöV) and the 87 of the German Radiation Protection Regulation (StrlSchV) the informed consent forms including the patients consent for trial participation and the application of irradiation will be archived for at least 30 years after the end of the trial. The Study Center at the Department of Radiation Oncology will be responsible for archiving all relevant data. Declaration of Helsinki and good clinical practice The trial is conducted in accordance with the Declaration of Helsinki (2008 Version of the Declaration of Helsinki, adopted at the 59th WMA General Assembly, Seoul, October 2008) as well as with the guidelines of Good Clinical Practice (s. ICH-GCP: International Conference on Harmonization - Good Clinical Practice; ) in their current versions. Ethics Committee and Bundesamt für Strahlenschutz The study protocol, patient information and consent are approved by the ethics committee of the university of Heidelberg (S-165/2012). Furthermore, the trial is approved by the Bundesamt für Strahlenschutz (Z / /13). Discussion Currently, radical resection with/without adjuvant radiation treatment is the most performed treatment in patients with sacral chordoma. Hence, the evidence based only on small retrospective series [11,14]. De Lany et al. published data of 29 patients with chordomas of the mobile spine
7 Uhl et al. Radiation Oncology 2014, 9:100 Page 7 of 8 and sacrum that were treated by surgery and high dose proton-/photon irradiation in a phase II trial. In this trial no significant difference between R0 and R1/R2/biopsy could be shown regarding local control [28]. According to these results it can be asked whether an extended surgery with high morbidity is still necessary. Japanese retrospective data compared surgery only with carbon ion therapy only and found out a 5-year local-recurrence free survival rate of 62.5% for surgery and 100% for carbon ion therapy [22]. Further retrospective data of 95 patients after hypofractionated carbon ion treatment at NIRS seems to indicate a new therapeutic option in patients with sacral chordoma, which displays in contrast to the surgery lower morbidity with very good local control [25]. Imai et al. reported about three patients with grade 3 skin toxicity and two patients with grade 4 (73.6 GyE) [25]. A decided evaluation of skin toxicity after carbon ion treatment at NIRS showed grade 3 and 4 skin toxicity only in patients which received doses 70 GyE in 16 fractions. It was indicated that severe skin reaction may not develop if the prescribed dose is 64 GyE or less like in our trial [29]. Furthermore, we limit the maximum dose of the skin to 90-95% of the prescribed dose. Imai et al. reported about one patient with rectal bleeding grade 1, but no patient received a colostomy or urinary diversion after carbon ion treatment. 15 patients (16%) required medication due to sciatic neuropathy after carbon ion treatment. However, five of them received with 73.6 GyE a much higher dose than in our trial. More than 80% of the chordomas were localized above S2 [25]. Thus, the expected toxicity after radical resection would have been a complete urinary and bowel incontinence in at least 80% of the patients [20,21]. Furthermore chronic neuropathic pain, wound complications and walking difficulties are also possible side effects of sacrectomy [30,31]. Against this background, the published side effects after hypofractionated carbon ion therapy should be evaluated. Due to missing data for a hypofractionated carbon ion therapy in raster scan technique, this trial will confirm the historical data from Japan that were developed by using passive beam application. In the Japanese trial patients received a total dose of 52.8 to 73.6 GyE in 16 fractions. Most of the patients received 70.4 GyE. In contrast to our fractionation with up to 6 fractions per week (Monday- Saturday), the patients in Japan received only four fractions per week. Therefore, the total dose in our trial is limited to 64 GyE. Only patients with inoperable, residual or recurrent tumor will be included in our trial. Patients with resectable tumor are included only after refusal of surgery treatment. Moreover, no data of hypofractionated proton therapy in patients with sacral chordoma have been published so far. However, these data are necessary to perform a randomized prospective phase III study in the future. Competing interests The authors declare that they have no competing interests. Authors contributions MU, LE, ADJ, JD und KH have developed the study concept. MU, LE, KH and JD wrote the study protocol and obtained ethics approval. LE was responsible for statistical considerations. MU, ADJ, GH, JO, MBFR, JD and KH will provide patient care. MU, LE, ADJ, GH, JO, MBFR, OJ, JD and KH will implement the protocol and oversee collection of the data. All authors contributed to and approved the final manuscript. Acknowledgement We acknowledge financial support by Deutsche Forschungsgemeinschaft and Ruprecht-Karls-Universität Heidelberg within the funding programme Open Access Publishing. Author details 1 Department of Radiation Oncology, University of Heidelberg, Im Neuenheimer Feld 400, Heidelberg, Germany. 2 Heidelberg Ion Therapy Center (HIT), Im Neuenheimer Feld 450, Heidelberg, Germany. 3 German Cancer Research Center (dkfz), Im Neuenheimer Feld 280, Heidelberg, Germany. Received: 27 February 2014 Accepted: 24 April 2014 Published: 29 April 2014 References 1. Friedrich T, Scholz U, Elsasser T, Durante M, Scholz M: Calculation of the biological effects of ion beams based on the microscopic spatial damage distribution pattern. Int J Radiat Biol 2012, 88: Kramer M, Weyrather WK, Scholz M: The increased biological effectiveness of heavy charged particles: from radiobiology to treatment planning. Technol Cancer Res Treat 2003, 2: Scholz M, Kraft G: Track structure and the calculation of biological effects of heavy charged particles. Adv Space Res 1996, 18: Murakami M, Eguchi-Kasai K, Sato K, Minohara S, Yatagai F, Kanai T: Differences in heavy-ion-induced DNA double-strand breaks in a mouse DNA repair-deficient mutant cell line (SL3-147) before and after chromatin proteolysis. J Radiat Res 1995, 36: Heilmann J, Taucher-Scholz G, Haberer T, Scholz M, Kraft G: Measurement of intracellular dna double-strand break induction and rejoining along the track of carbon and neon particle beams in water. Int J Radiat Oncol Biol Phys 1996, 34: McMaster ML, Goldstein AM, Bromley CM, Ishibe N, Parry DM: Chordoma: incidence and survival patterns in the United States, Cancer Causes Control 2001, 12: Bjornsson J, Wold LE, Ebersold MJ, Laws ER: Chordoma of the mobile spine. A clinicopathologic analysis of 40 patients. Cancer 1993, 71: Rosenberg AE, Brown GA, Bhan AK, Lee JM: Chondroid chordoma a variant of chordoma. A morphologic and immunohistochemical study. Am J Clin Pathol 1994, 101: Chugh R, Tawbi H, Lucas DR, Biermann JS, Schuetze SM, Baker LH: Chordoma: the nonsarcoma primary bone tumor. Oncologist 2007, 12: Chen KW, Yang HL, Lu J, Liu JY, Chen XQ: Prognostic factors of sacral chordoma after surgical therapy: a study of 36 patients. Spinal Cord 2010, 48: Fuchs B, Dickey ID, Yaszemski MJ, Inwards CY, Sim FH: Operative management of sacral chordoma. J Bone Joint Surg Am 2005, 87: Osaka S, Kodoh O, Sugita H, Osaka E, Yoshida Y, Ryu J: Clinical significance of a wide excision policy for sacrococcygeal chordoma. J Cancer Res Clin Oncol 2006, 132: York JE, Kaczaraj A, Abi-Said D, Fuller GN, Skibber JM, Janjan NA, York JE, Kaczaraj A, Abi-Said D, Fuller GN, Skibber JM, Janjan NA, Gokaslan ZL: Sacral chordoma: 40-year experience at a major cancer center. Neurosurgery 1999, 44: discussion Park L, Delaney TF, Liebsch NJ, Hornicek FJ, Goldberg S, Mankin H, Rosenberg AE, Rosenthal DI, Suit HD: Sacral chordomas: Impact of high-dose proton/photon-beam radiation therapy combined with or without surgery for primary versus recurrent tumor. Int J Radiat Oncol Biol Phys 2006, 65:
8 Uhl et al. Radiation Oncology 2014, 9:100 Page 8 of Munzenrider JE, Liebsch NJ: Proton therapy for tumors of the skull base. Strahlenther Onkol 1999, 175(Suppl 2): Hug EB, Loredo LN, Slater JD, DeVries A, Grove RI, Schaefer RA, Rosenberg AE, Slater JM: Proton radiation therapy for chordomas and chondrosarcomas of the skull base. JNeurosurg1999, 91: Ares C, Hug EB, Lomax AJ, Bolsi A, Timmermann B, Rutz HP, Schuller JC, Pedroni E, Goitein G: Effectiveness and safety of spot scanning proton radiation therapy for chordomas and chondrosarcomas of the skull base: first long-term report. Int J Radiat Oncol Biol Phys 2009, 75: Schulz-Ertner D, Karger CP, Feuerhake A, Nikoghosyan A, Combs SE, Jäkel O, Edler L, Scholz M, Debus J: Effectiveness of carbon ion radiotherapy in the treatment of skull-base chordomas. Int J Radiat Oncol Biol Phys 2007, 68: Nikoghosyan AV, Karapanagiotou-Schenkel I, Munter MW, Jensen AD, Combs SE, Debus J: Randomised trial of proton vs. carbon ion radiation therapy in patients with chordoma of the skull base, clinical phase III study HIT-1-Study. BMC Cancer 2010, 10: Devin C, Chong PY, Holt GE, Feurer I, Gonzalez A, Merchant N, Schwartz HS: Level-adjusted perioperative risk of sacral amputations. J Surg Oncol 2006, 94: Cheng EY, Ozerdemoglu RA, Transfeldt EE, Thompson RC Jr: Lumbosacral chordoma. Prognostic factors and treatment. Spine (Phila Pa 1976) 1999, 24: Nishida Y, Kamada T, Imai R, Tsukushi S, Yamada Y, Sugiura H, Shido Y, Wasa J, Ishiguro N: Clinical outcome of sacral chordoma with carbon ion radiotherapy compared with surgery. Int J Radiat Oncol Biol Phys 2011, 79: Imai R, Kamada T, Tsuji H, Sugawara S, Serizawa I, Tsujii H, Tatezaki S, Working Group for Bone and Soft Tissue Sarcomas: Effect of carbon ion radiotherapy for sacral chordoma: results of Phase I-II and Phase II clinical trials. Int J Radiat Oncol Biol Phys 2010, 77: Elsasser T, Kramer M, Scholz M: Accuracy of the local effect model for the prediction of biologic effects of carbon ion beams in vitro and in vivo. Int J Radiat Oncol Biol Phys 2008, 71: Imai R, Kamada T, Sugahara S, Tsuji H, Tsujii H: Carbon ion radiotherapy for sacral chordoma. Br J Radiol 2011, 84(Spec No 1):S48 S Hall EJ: Intensity-modulated radiation therapy, protons, and the risk of second cancers. Int J Radiat Oncol Biol Phys 2006, 65: Blackwelder WC: Proving the null hypothesis in clinical trials. Control Clin Trials 1982, 3: DeLaney TF, Liebsch NJ, Pedlow FX, Adams J, Dean S, Yeap BY, McManus P, Rosenberg AE, Nielsen GP, Harmon DC, Spiro IJ, Raskin KA, Suit HD, Yoon SS, Hornicek FJ: Phase II study of high-dose photon/proton radiotherapy in the management of spine sarcomas. Int J Radiat Oncol Biol Phys 2009, 74: Yanagi T, Kamada T, Tsuji H, Imai R, Serizawa I, Tsujii H: Dose-volume histogram and dose-surface histogram analysis for skin reactions to carbon ion radiotherapy for bone and soft tissue sarcoma. Radiother Oncol 2010, 95: Davidge KM, Eskicioglu C, Lipa J, Ferguson P, Swallow CJ, Wright FC: Qualitative assessment of patient experiences following sacrectomy. J Surg Oncol 2010, 101: Hulen CA, Temple HT, Fox WP, Sama AA, Green BA, Eismont FJ: Oncologic and functional outcome following sacrectomy for sacral chordoma. J Bone Joint Surg Am 2006, 88: doi: / x Cite this article as: Uhl et al.: Randomized phase II trial of hypofractionated proton versus carbon ion radiation therapy in patients with sacrococcygeal chordoma-the ISAC trial protocol. Radiation Oncology :100. Submit your next manuscript to BioMed Central and take full advantage of: Convenient online submission Thorough peer review No space constraints or color figure charges Immediate publication on acceptance Inclusion in PubMed, CAS, Scopus and Google Scholar Research which is freely available for redistribution Submit your manuscript at
Sacral Chordoma: The Loma Linda University Radiation Medicine Experience. Kevin Yiee MD, MPH Resident Physician
 Sacral Chordoma: The Loma Linda University Radiation Medicine Experience Kevin Yiee MD, MPH Resident Physician What is a chordoma? 1 st chordoma discovered in clivus by Virchow and Luschka 1856 Rare tumor
Sacral Chordoma: The Loma Linda University Radiation Medicine Experience Kevin Yiee MD, MPH Resident Physician What is a chordoma? 1 st chordoma discovered in clivus by Virchow and Luschka 1856 Rare tumor
Clinical Results of Carbon Ion Radiotherapy: The Heidelberg Experience
 Clinical Results of Carbon Ion Radiotherapy: The Heidelberg Experience Stephanie E. Combs, MD Department of Radiation Oncology University of Heidelberg, Germany Carbon ion RT at GSI Active beam delivery
Clinical Results of Carbon Ion Radiotherapy: The Heidelberg Experience Stephanie E. Combs, MD Department of Radiation Oncology University of Heidelberg, Germany Carbon ion RT at GSI Active beam delivery
Carbon Ion Radiotherapy for Skull Base and Paracervical Chordomas
 Carbon Ion Radiotherapy for Skull Base and Paracervical Chordomas Azusa Hasegawa, Jun-etsu Mizoe and Hirohiko Tsujii Research Center Hospital for Charged Particle Therapy National Institute of Radiological
Carbon Ion Radiotherapy for Skull Base and Paracervical Chordomas Azusa Hasegawa, Jun-etsu Mizoe and Hirohiko Tsujii Research Center Hospital for Charged Particle Therapy National Institute of Radiological
Proton Radiation Therapy for Osteosarcomas, Chondrogenic Tumors and Soft Tissue Sarcomas
 Proton Radiation Therapy for Osteosarcomas, Chondrogenic Tumors and Soft Tissue Sarcomas Eugen B. Hug Center for Proton Radiation Therapy Paul Scherrer Institute Histologies Osteogenic Tumors Osteogenic
Proton Radiation Therapy for Osteosarcomas, Chondrogenic Tumors and Soft Tissue Sarcomas Eugen B. Hug Center for Proton Radiation Therapy Paul Scherrer Institute Histologies Osteogenic Tumors Osteogenic
Proton Therapy for tumors of the skull base - RESULTS. Eugen B. Hug, MD Medical Director, ProCure Proton Therapy Centers, NY
 Proton Therapy for tumors of the skull base - RESULTS Eugen B. Hug, MD Medical Director, ProCure Proton Therapy Centers, NY Petroclival Chondrosarcoma: 68 72 Gy(RBE) at 1.8 or 2.0 Gy(RBE) GTV: 70.2 Gy(RBE)
Proton Therapy for tumors of the skull base - RESULTS Eugen B. Hug, MD Medical Director, ProCure Proton Therapy Centers, NY Petroclival Chondrosarcoma: 68 72 Gy(RBE) at 1.8 or 2.0 Gy(RBE) GTV: 70.2 Gy(RBE)
Proton Radiation Therapy for Osteosarcomas, Chondrogenic Tumors and Soft Tissue Sarcomas
 Proton Radiation Therapy for Osteosarcomas, Chondrogenic Tumors and Soft Tissue Sarcomas Eugen B. Hug Center for Proton Radiation Therapy Paul Scherrer Institute Is there a place for Proton/Particle Radiotherapy
Proton Radiation Therapy for Osteosarcomas, Chondrogenic Tumors and Soft Tissue Sarcomas Eugen B. Hug Center for Proton Radiation Therapy Paul Scherrer Institute Is there a place for Proton/Particle Radiotherapy
Dosimetric Analysis of 3DCRT or IMRT with Vaginal-cuff Brachytherapy (VCB) for Gynaecological Cancer
 Dosimetric Analysis of 3DCRT or IMRT with Vaginal-cuff Brachytherapy (VCB) for Gynaecological Cancer Tan Chek Wee 15 06 2016 National University Cancer Institute, Singapore Clinical Care Education Research
Dosimetric Analysis of 3DCRT or IMRT with Vaginal-cuff Brachytherapy (VCB) for Gynaecological Cancer Tan Chek Wee 15 06 2016 National University Cancer Institute, Singapore Clinical Care Education Research
Treatment Planning (Protons vs. Photons)
 Treatment Planning Treatment Planning (Protons vs. Photons) Acquisition of imaging data Delineation of regions of interest Selection of beam directions Dose calculation Optimization of the plan Hounsfield
Treatment Planning Treatment Planning (Protons vs. Photons) Acquisition of imaging data Delineation of regions of interest Selection of beam directions Dose calculation Optimization of the plan Hounsfield
Proton Radiotherapy for Skull Base and Para-spinal Tumors
 Proton Radiotherapy for Skull Base and Para-spinal Tumors Carmen Ares Primary tumors Tumors of the Skull Base - Chordomas, Chondrosarcomas Secondary infiltration or involvement by intracranial tumors -
Proton Radiotherapy for Skull Base and Para-spinal Tumors Carmen Ares Primary tumors Tumors of the Skull Base - Chordomas, Chondrosarcomas Secondary infiltration or involvement by intracranial tumors -
Effectiveness and Safety of Spot Scanning Proton Radiation Therapy for Skull Base Tumors: First Long Term Report of the PSI Experience
 Effectiveness and Safety of Spot Scanning Proton Radiation Therapy for Skull Base Tumors: First Long Term Report of the PSI Experience Carmen Ares, Antony J Lomax, Eugen B Hug, Alessandra Bolsi, Beate
Effectiveness and Safety of Spot Scanning Proton Radiation Therapy for Skull Base Tumors: First Long Term Report of the PSI Experience Carmen Ares, Antony J Lomax, Eugen B Hug, Alessandra Bolsi, Beate
A new score predicting the survival of patients with spinal cord compression from myeloma
 Douglas et al. BMC Cancer 2012, 12:425 RESEARCH ARTICLE Open Access A new score predicting the survival of patients with spinal cord compression from myeloma Sarah Douglas 1, Steven E Schild 2 and Dirk
Douglas et al. BMC Cancer 2012, 12:425 RESEARCH ARTICLE Open Access A new score predicting the survival of patients with spinal cord compression from myeloma Sarah Douglas 1, Steven E Schild 2 and Dirk
Radiation Therapy for Soft Tissue Sarcomas
 Radiation Therapy for Soft Tissue Sarcomas Alexander R. Gottschalk, MD, PhD Assistant Professor, Radiation Oncology University of California, San Francisco 1/25/08 NCI: limb salvage vs. amputation 43 patients
Radiation Therapy for Soft Tissue Sarcomas Alexander R. Gottschalk, MD, PhD Assistant Professor, Radiation Oncology University of California, San Francisco 1/25/08 NCI: limb salvage vs. amputation 43 patients
IMRT - the physician s eye-view. Cinzia Iotti Department of Radiation Oncology S.Maria Nuova Hospital Reggio Emilia
 IMRT - the physician s eye-view Cinzia Iotti Department of Radiation Oncology S.Maria Nuova Hospital Reggio Emilia The goals of cancer therapy Local control Survival Functional status Quality of life Causes
IMRT - the physician s eye-view Cinzia Iotti Department of Radiation Oncology S.Maria Nuova Hospital Reggio Emilia The goals of cancer therapy Local control Survival Functional status Quality of life Causes
Case Report Carbon Ion Beam Radiotherapy for Sinonasal Malignant Tumors Invading Skull Base
 Case Reports in Otolaryngology, Article ID 241856, 4 pages http://dx.doi.org/10.1155/2014/241856 Case Report Carbon Ion Beam Radiotherapy for Sinonasal Malignant Tumors Invading Skull Base Nobuo Ohta,
Case Reports in Otolaryngology, Article ID 241856, 4 pages http://dx.doi.org/10.1155/2014/241856 Case Report Carbon Ion Beam Radiotherapy for Sinonasal Malignant Tumors Invading Skull Base Nobuo Ohta,
14. Background. Sarcoma. Resectable extremity soft tissue sarcomas
 96 14. Sarcoma Background Radiotherapy is widely used as an adjunct to surgery in the management of soft tissue sarcomas as the risk of failure in the surgical bed can be high. For bone sarcomas, radiotherapy
96 14. Sarcoma Background Radiotherapy is widely used as an adjunct to surgery in the management of soft tissue sarcomas as the risk of failure in the surgical bed can be high. For bone sarcomas, radiotherapy
Dr. Robert R. Wilson, a physicist who had worked on
 Radiation Therapy With Charged Particles Daniela Schulz-Ertner, MD,* Oliver Jäkel, PhD, and Wolfgang Schlegel, PhD Charged particle beams can offer an improved dose conformation to the target volume as
Radiation Therapy With Charged Particles Daniela Schulz-Ertner, MD,* Oliver Jäkel, PhD, and Wolfgang Schlegel, PhD Charged particle beams can offer an improved dose conformation to the target volume as
EVALUATION OF THERAPEUTIC POTENTIAL OF HEAVY ION THERAPY FOR PATIENTS WITH LOCALLY ADVANCED PROSTATE CANCER
 doi:10.1016/s0360-3016(03)01439-1 Int. J. Radiation Oncology Biol. Phys., Vol. 58, No. 1, pp. 89 97, 2004 Copyright 2004 Elsevier Inc. Printed in the USA. All rights reserved 0360-3016/04/$ see front matter
doi:10.1016/s0360-3016(03)01439-1 Int. J. Radiation Oncology Biol. Phys., Vol. 58, No. 1, pp. 89 97, 2004 Copyright 2004 Elsevier Inc. Printed in the USA. All rights reserved 0360-3016/04/$ see front matter
Proton and heavy ion radiotherapy: Effect of LET
 Proton and heavy ion radiotherapy: Effect of LET As a low LET particle traverses a DNA molecule, ionizations are far apart and double strand breaks are rare With high LET particles, ionizations are closer
Proton and heavy ion radiotherapy: Effect of LET As a low LET particle traverses a DNA molecule, ionizations are far apart and double strand breaks are rare With high LET particles, ionizations are closer
A Comparison of IMRT and VMAT Technique for the Treatment of Rectal Cancer
 A Comparison of IMRT and VMAT Technique for the Treatment of Rectal Cancer Tony Kin Ming Lam Radiation Planner Dr Patricia Lindsay, Radiation Physicist Dr John Kim, Radiation Oncologist Dr Kim Ann Ung,
A Comparison of IMRT and VMAT Technique for the Treatment of Rectal Cancer Tony Kin Ming Lam Radiation Planner Dr Patricia Lindsay, Radiation Physicist Dr John Kim, Radiation Oncologist Dr Kim Ann Ung,
PTCOG 46. Educational Workshop Session IV. Head & Neck CLINICAL. J. Mizoe (NIRS, Japan)
 PTCOG 46 Educational Workshop Session IV CLINICAL Head & Neck J. Mizoe (NIRS, Japan) Photon X-Ray γ-ray Fast Neutron Non-Charged Radiation Electron Proton Helium Light Ion Heavy Particle Carbon Neon Argon
PTCOG 46 Educational Workshop Session IV CLINICAL Head & Neck J. Mizoe (NIRS, Japan) Photon X-Ray γ-ray Fast Neutron Non-Charged Radiation Electron Proton Helium Light Ion Heavy Particle Carbon Neon Argon
Status of H 1 and C 12
 Status of H 1 and C 12 Herman Suit No Conflict of Interest 1 Goal of a New Treatment Modality Tumor Control Probability or No in Complication Rate 2 Truism No Advantage to: any Patient for any Radiation
Status of H 1 and C 12 Herman Suit No Conflict of Interest 1 Goal of a New Treatment Modality Tumor Control Probability or No in Complication Rate 2 Truism No Advantage to: any Patient for any Radiation
Proton-Radiotherapy for Tumors of the Skull Base: Indications, Advantages, Limitations
 Proton-Radiotherapy for Tumors of the Skull Base: Indications, Advantages, Limitations Eugen B. Hug, MD Director, Center for Proton-Radiotherapy, Paul Scherrer Institut, Villigen and Chair of Proton-Radiotherapy.
Proton-Radiotherapy for Tumors of the Skull Base: Indications, Advantages, Limitations Eugen B. Hug, MD Director, Center for Proton-Radiotherapy, Paul Scherrer Institut, Villigen and Chair of Proton-Radiotherapy.
BLADDER RADIOTHERAPY PLANNING DOCUMENT
 A 2X2 FACTORIAL RANDOMISED PHASE III STUDY COMPARING STANDARD VERSUS REDUCED VOLUME RADIOTHERAPY WITH AND WITHOUT SYNCHRONOUS CHEMOTHERAPY IN MUSCLE INVASIVE BLADDER CANCER (ISRCTN 68324339) BLADDER RADIOTHERAPY
A 2X2 FACTORIAL RANDOMISED PHASE III STUDY COMPARING STANDARD VERSUS REDUCED VOLUME RADIOTHERAPY WITH AND WITHOUT SYNCHRONOUS CHEMOTHERAPY IN MUSCLE INVASIVE BLADDER CANCER (ISRCTN 68324339) BLADDER RADIOTHERAPY
Nordic Society for Gynecological Oncology Advisory Board of Radiotherapy
 Nordic Society for Gynecological Oncology Advisory Board of Radiotherapy Guidelines for postoperative irradiation of cervical cancer Contents: 1. Treatment planning for EBRT. 2 2. Target definition for
Nordic Society for Gynecological Oncology Advisory Board of Radiotherapy Guidelines for postoperative irradiation of cervical cancer Contents: 1. Treatment planning for EBRT. 2 2. Target definition for
REVISITING ICRU VOLUME DEFINITIONS. Eduardo Rosenblatt Vienna, Austria
 REVISITING ICRU VOLUME DEFINITIONS Eduardo Rosenblatt Vienna, Austria Objective: To introduce target volumes and organ at risk concepts as defined by ICRU. 3D-CRT is the standard There was a need for a
REVISITING ICRU VOLUME DEFINITIONS Eduardo Rosenblatt Vienna, Austria Objective: To introduce target volumes and organ at risk concepts as defined by ICRU. 3D-CRT is the standard There was a need for a
Particle Therapy for Tumors of the Skull Base. Eugen B. Hug, MD Medical Director, ProCure Proton Therapy Centers, NY
 Particle Therapy for Tumors of the Skull Base Eugen B. Hug, MD Medical Director, ProCure Proton Therapy Centers, NY Particle Radiation Therapy for Tumors of the Skull Base Primary skull base tumors: Chordoma,
Particle Therapy for Tumors of the Skull Base Eugen B. Hug, MD Medical Director, ProCure Proton Therapy Centers, NY Particle Radiation Therapy for Tumors of the Skull Base Primary skull base tumors: Chordoma,
New Technologies for the Radiotherapy of Prostate Cancer
 Prostate Cancer Meyer JL (ed): IMRT, IGRT, SBRT Advances in the Treatment Planning and Delivery of Radiotherapy. Front Radiat Ther Oncol. Basel, Karger, 27, vol. 4, pp 315 337 New Technologies for the
Prostate Cancer Meyer JL (ed): IMRT, IGRT, SBRT Advances in the Treatment Planning and Delivery of Radiotherapy. Front Radiat Ther Oncol. Basel, Karger, 27, vol. 4, pp 315 337 New Technologies for the
Where are we with radiotherapy for biliary tract cancers?
 Where are we with radiotherapy for biliary tract cancers? Professor Maria A. Hawkins Associate Professor in Clinical Oncology MRC Group Leader/Honorary Consultant Clinical Oncologist CRUK MRC Oxford Institute
Where are we with radiotherapy for biliary tract cancers? Professor Maria A. Hawkins Associate Professor in Clinical Oncology MRC Group Leader/Honorary Consultant Clinical Oncologist CRUK MRC Oxford Institute
Linac Based SBRT for Low-intermediate Risk Prostate Cancer in 5 Fractions: Preliminary Report of a Phase II Study with FFF Delivery
 Linac Based SBRT for Low-intermediate Risk Prostate Cancer in 5 Fractions: Preliminary Report of a Phase II Study with FFF Delivery FILIPPO ALONGI MD Radiation Oncology & Radiosurgery Istituto Clinico
Linac Based SBRT for Low-intermediate Risk Prostate Cancer in 5 Fractions: Preliminary Report of a Phase II Study with FFF Delivery FILIPPO ALONGI MD Radiation Oncology & Radiosurgery Istituto Clinico
Review of Workflow NRG (RTOG) 1308: Phase III Randomized Trial Comparing Overall Survival after Photon versus Proton Chemoradiation Therapy for
 Review of Workflow NRG (RTOG) 1308: Phase III Randomized Trial Comparing Overall Survival after Photon versus Proton Chemoradiation Therapy for Inoperable Stage II-IIIB NSCLC 1 Co-Chairs Study Chair: Zhongxing
Review of Workflow NRG (RTOG) 1308: Phase III Randomized Trial Comparing Overall Survival after Photon versus Proton Chemoradiation Therapy for Inoperable Stage II-IIIB NSCLC 1 Co-Chairs Study Chair: Zhongxing
Changing Paradigms in Radiotherapy
 Changing Paradigms in Radiotherapy Marco van Vulpen, MD, PhD Mouldroomdag-2015 Towards the elimination of invasion 1 NIH opinion on the future of oncology Twenty-five years from now,i hope that we won
Changing Paradigms in Radiotherapy Marco van Vulpen, MD, PhD Mouldroomdag-2015 Towards the elimination of invasion 1 NIH opinion on the future of oncology Twenty-five years from now,i hope that we won
IMPT with Carbon Ions
 IMPT with Carbon Ions PTCOG 48, Heidelberg, 28.09.-03.10.2009 Malte Ellerbrock Medical Physics Expert Heidelberg Ion-Beam Therapy Center HIT Betriebs GmbH am Universitätsklinikum Heidelberg http://www.hit-centrum.de
IMPT with Carbon Ions PTCOG 48, Heidelberg, 28.09.-03.10.2009 Malte Ellerbrock Medical Physics Expert Heidelberg Ion-Beam Therapy Center HIT Betriebs GmbH am Universitätsklinikum Heidelberg http://www.hit-centrum.de
Radiotherapy Planning (Contouring Lung Cancer for Radiotherapy dose prescription) Dr Raj K Shrimali
 Radiotherapy Planning (Contouring Lung Cancer for Radiotherapy dose prescription) Dr Raj K Shrimali Let us keep this simple and stick to some basic rules Patient positioning Must be reproducible Must be
Radiotherapy Planning (Contouring Lung Cancer for Radiotherapy dose prescription) Dr Raj K Shrimali Let us keep this simple and stick to some basic rules Patient positioning Must be reproducible Must be
8/3/2016. Clinical Significance of RBE Variations in Proton Therapy. Why RBE (relative biological effectiveness)?
 8//06 Clinical Significance of Variations in Proton Therapy H. Paganetti PhD Professor, Harvard Medical School Director of Physics Research, Massachusetts General Hospital, Radiation Oncology Introduction
8//06 Clinical Significance of Variations in Proton Therapy H. Paganetti PhD Professor, Harvard Medical School Director of Physics Research, Massachusetts General Hospital, Radiation Oncology Introduction
Advances in external beam radiotherapy
 International Conference on Modern Radiotherapy: Advances and Challenges in Radiation Protection of Patients Advances in external beam radiotherapy New techniques, new benefits and new risks Michael Brada
International Conference on Modern Radiotherapy: Advances and Challenges in Radiation Protection of Patients Advances in external beam radiotherapy New techniques, new benefits and new risks Michael Brada
CyberKnife SBRT for Prostate Cancer
 CyberKnife SBRT for Prostate Cancer Robert Meier, MD Swedish Radiosurgery Center Swedish Cancer Institute Seattle, WA 2017 ESTRO Meeting, Vienna Austria 5-year safety, efficacy & quality of life outcomes
CyberKnife SBRT for Prostate Cancer Robert Meier, MD Swedish Radiosurgery Center Swedish Cancer Institute Seattle, WA 2017 ESTRO Meeting, Vienna Austria 5-year safety, efficacy & quality of life outcomes
The Evolution of SBRT and Hypofractionation in Thoracic Radiation Oncology
 The Evolution of SBRT and Hypofractionation in Thoracic Radiation Oncology (specifically, lung cancer) 2/10/18 Jeffrey Kittel, MD Radiation Oncology, Aurora St. Luke s Medical Center Outline The history
The Evolution of SBRT and Hypofractionation in Thoracic Radiation Oncology (specifically, lung cancer) 2/10/18 Jeffrey Kittel, MD Radiation Oncology, Aurora St. Luke s Medical Center Outline The history
Radiation Technology, Hyogo Ion Beam Medical Center, Tatsuno, Hyogo, JAPAN
 Analysis of Visual Loss Due to Radiation- Induced Optic Neuropathy After Particle Therapy for Head and Neck and Skull Base Tumors Adjacent to Optic Nerves Y. Demizu 1, M. Murakami 1, D. Miyawaki 1, Y.
Analysis of Visual Loss Due to Radiation- Induced Optic Neuropathy After Particle Therapy for Head and Neck and Skull Base Tumors Adjacent to Optic Nerves Y. Demizu 1, M. Murakami 1, D. Miyawaki 1, Y.
Defining Target Volumes and Organs at Risk: a common language
 Defining Target Volumes and Organs at Risk: a common language Eduardo Rosenblatt Section Head Applied Radiation Biology and Radiotherapy (ARBR) Section Division of Human Health IAEA Objective: To introduce
Defining Target Volumes and Organs at Risk: a common language Eduardo Rosenblatt Section Head Applied Radiation Biology and Radiotherapy (ARBR) Section Division of Human Health IAEA Objective: To introduce
What s new in bone and soft tissue sarcoma Treatment and Guidelines 2012? Rob Grimer
 What s new in bone and soft tissue sarcoma Treatment and Guidelines 2012? Rob Grimer ESMO conference 2012 Top Oncologists in world (~ 400) Lots of sarcoma basic science key messages: 40% of STS diagnoses
What s new in bone and soft tissue sarcoma Treatment and Guidelines 2012? Rob Grimer ESMO conference 2012 Top Oncologists in world (~ 400) Lots of sarcoma basic science key messages: 40% of STS diagnoses
Biological Optimization of Hadrontherapy. Uwe Oelfke
 4/2/2012 page 1 Biological Optimization of Hadrontherapy Uwe Oelfke DKFZ Heidelberg (E040) Im Neuenheimer Feld 280 69120 Heidelberg, Germany u.oelfke@dkfz.de 4/2/2012 page 2 Contents Introduction and General
4/2/2012 page 1 Biological Optimization of Hadrontherapy Uwe Oelfke DKFZ Heidelberg (E040) Im Neuenheimer Feld 280 69120 Heidelberg, Germany u.oelfke@dkfz.de 4/2/2012 page 2 Contents Introduction and General
Evaluation of Whole-Field and Split-Field Intensity Modulation Radiation Therapy (IMRT) Techniques in Head and Neck Cancer
 1 Charles Poole April Case Study April 30, 2012 Evaluation of Whole-Field and Split-Field Intensity Modulation Radiation Therapy (IMRT) Techniques in Head and Neck Cancer Abstract: Introduction: This study
1 Charles Poole April Case Study April 30, 2012 Evaluation of Whole-Field and Split-Field Intensity Modulation Radiation Therapy (IMRT) Techniques in Head and Neck Cancer Abstract: Introduction: This study
Radiation Therapy for Liver Malignancies
 Outline Radiation Therapy for Liver Malignancies Albert J. Chang, M.D., Ph.D. Department of Radiation Oncology, UCSF March 23, 2014 Rationale for developing liver directed therapies Liver directed therapies
Outline Radiation Therapy for Liver Malignancies Albert J. Chang, M.D., Ph.D. Department of Radiation Oncology, UCSF March 23, 2014 Rationale for developing liver directed therapies Liver directed therapies
Questions may be submitted anytime during the presentation.
 Understanding Radiation Therapy and its Role in Treating Patients with Pancreatic Cancer Presented by Pancreatic Cancer Action Network www.pancan.org August 18, 2014 If you experience technical difficulty
Understanding Radiation Therapy and its Role in Treating Patients with Pancreatic Cancer Presented by Pancreatic Cancer Action Network www.pancan.org August 18, 2014 If you experience technical difficulty
Clinical considerations of RBE in proton therapy
 Clinical considerations of RBE in proton therapy H. Paganetti PhD Professor, Harvard Medical School Director of Physics Research, Massachusetts General Hospital, Radiation Oncology Why do we need the RBE
Clinical considerations of RBE in proton therapy H. Paganetti PhD Professor, Harvard Medical School Director of Physics Research, Massachusetts General Hospital, Radiation Oncology Why do we need the RBE
Re-growth of an incomplete discoid lateral meniscus after arthroscopic partial resection in an 11 year-old boy: a case report
 Bisicchia and Tudisco BMC Musculoskeletal Disorders 2013, 14:285 CASE REPORT Open Access Re-growth of an incomplete discoid lateral meniscus after arthroscopic partial resection in an 11 year-old boy:
Bisicchia and Tudisco BMC Musculoskeletal Disorders 2013, 14:285 CASE REPORT Open Access Re-growth of an incomplete discoid lateral meniscus after arthroscopic partial resection in an 11 year-old boy:
The Heidelberg Ion Therapy Center and PARTNER. Thomas Haberer Heidelberg Ion Therapy Center
 The Heidelberg Ion Therapy Center and PARTNER Thomas Haberer Heidelberg Ion Therapy Center Goal The key element to improve the clinical outcome is local control! entrance channel: low physical dose low
The Heidelberg Ion Therapy Center and PARTNER Thomas Haberer Heidelberg Ion Therapy Center Goal The key element to improve the clinical outcome is local control! entrance channel: low physical dose low
Chemoradiation (CRT) Safety Analysis of ACOSOG Z6041: A Phase II Trial of Neoadjuvant CRT followed by Local Excision in ut2 Rectal Cancer
 Chemoradiation (CRT) Safety Analysis of ACOSOG Z6041: A Phase II Trial of Neoadjuvant CRT followed by Local Excision in ut2 Rectal Cancer Emily Chan, Qian Shi, Julio Garcia-Aguilar, Peter Cataldo, Jorge
Chemoradiation (CRT) Safety Analysis of ACOSOG Z6041: A Phase II Trial of Neoadjuvant CRT followed by Local Excision in ut2 Rectal Cancer Emily Chan, Qian Shi, Julio Garcia-Aguilar, Peter Cataldo, Jorge
Treatment Planning Evaluation of Volumetric Modulated Arc Therapy (VMAT) for Craniospinal Irradiation (CSI)
 Treatment Planning Evaluation of Volumetric Modulated Arc Therapy (VMAT) for Craniospinal Irradiation (CSI) Tagreed AL-ALAWI Medical Physicist King Abdullah Medical City- Jeddah Aim 1. Simplify and standardize
Treatment Planning Evaluation of Volumetric Modulated Arc Therapy (VMAT) for Craniospinal Irradiation (CSI) Tagreed AL-ALAWI Medical Physicist King Abdullah Medical City- Jeddah Aim 1. Simplify and standardize
Clinical Commissioning Policy Proposition: Proton Beam Therapy for Cancer of the Prostate
 Clinical Commissioning Policy Proposition: Proton Beam Therapy for Cancer of the Prostate Reference: NHS England B01X09 First published: March 2016 Prepared by NHS England Specialised Services Clinical
Clinical Commissioning Policy Proposition: Proton Beam Therapy for Cancer of the Prostate Reference: NHS England B01X09 First published: March 2016 Prepared by NHS England Specialised Services Clinical
Institute of Oncology & Radiobiology. Havana, Cuba. INOR
 Institute of Oncology & Radiobiology. Havana, Cuba. INOR 1 Transition from 2-D 2 D to 3-D 3 D conformal radiotherapy in high grade gliomas: : our experience in Cuba Chon. I, MD - Chi. D, MD - Alert.J,
Institute of Oncology & Radiobiology. Havana, Cuba. INOR 1 Transition from 2-D 2 D to 3-D 3 D conformal radiotherapy in high grade gliomas: : our experience in Cuba Chon. I, MD - Chi. D, MD - Alert.J,
Citation Key for more information see:
 Citation Key for more information see: http://open.umich.edu/wiki/citationpolicy Use + Share + Adapt { Content the copyright holder, author, or law permits you to use, share and adapt. } Public Domain
Citation Key for more information see: http://open.umich.edu/wiki/citationpolicy Use + Share + Adapt { Content the copyright holder, author, or law permits you to use, share and adapt. } Public Domain
INTRAOPERATIVE RADIATION THERAPY FOR RETROPERITONEAL SARCOMA
 INTRAOPERATIVE RADIATION THERAPY FOR RETROPERITONEAL SARCOMA ISIORT 2014 Ivy A Petersen, MD Mayo Clinic Rochester, MN NOTHING TO DISCLOSE SOFT TISSUE SARCOMAS 2014 Estimated cases in the USA 12,020 diagnosed
INTRAOPERATIVE RADIATION THERAPY FOR RETROPERITONEAL SARCOMA ISIORT 2014 Ivy A Petersen, MD Mayo Clinic Rochester, MN NOTHING TO DISCLOSE SOFT TISSUE SARCOMAS 2014 Estimated cases in the USA 12,020 diagnosed
Martin Stuschke 1,2*, Andreas Kaiser 1,2, Jehad Abu Jawad 1, Christoph Pöttgen 1, Sabine Levegrün 1 and Jonathan Farr 2,3
 Stuschke et al. Radiation Oncology 2013, 8:145 SHORT REPORT Open Access Multi-scenario based robust intensity-modulated proton therapy (IMPT) plans can account for set-up errors more effectively in terms
Stuschke et al. Radiation Oncology 2013, 8:145 SHORT REPORT Open Access Multi-scenario based robust intensity-modulated proton therapy (IMPT) plans can account for set-up errors more effectively in terms
Specification of Tumor Dose. Prescription dose. Purpose
 Specification of Tumor Dose George Starkschall, Ph.D. Department of Radiation Physics U.T. M.D. Anderson Cancer Center Prescription dose What do we mean by a dose prescription of 63 Gy? Isocenter dose
Specification of Tumor Dose George Starkschall, Ph.D. Department of Radiation Physics U.T. M.D. Anderson Cancer Center Prescription dose What do we mean by a dose prescription of 63 Gy? Isocenter dose
Phase I/II trial evaluating carbon ion radiotherapy for the treatment of recurrent rectal cancer: the PANDORA-01 trial
 STUDY PROTOCOL Open Access Phase I/II trial evaluating carbon ion radiotherapy for the treatment of recurrent rectal cancer: the PANDORA-01 trial Stephanie E Combs 1*, Meinhard Kieser 2, Daniel Habermehl
STUDY PROTOCOL Open Access Phase I/II trial evaluating carbon ion radiotherapy for the treatment of recurrent rectal cancer: the PANDORA-01 trial Stephanie E Combs 1*, Meinhard Kieser 2, Daniel Habermehl
The Physics of Oesophageal Cancer Radiotherapy
 The Physics of Oesophageal Cancer Radiotherapy Dr. Philip Wai Radiotherapy Physics Royal Marsden Hospital 1 Contents Brief clinical introduction Imaging and Target definition Dose prescription & patient
The Physics of Oesophageal Cancer Radiotherapy Dr. Philip Wai Radiotherapy Physics Royal Marsden Hospital 1 Contents Brief clinical introduction Imaging and Target definition Dose prescription & patient
Post-Lumpectomy Radiation Techniques and Toxicities
 Post-Lumpectomy Radiation Techniques and Toxicities Laura Willson, MD Abbott Northwestern Hospital Dept. of Radiation Oncology February 2, 2019 Learning Objectives How radiation therapy works Standard
Post-Lumpectomy Radiation Techniques and Toxicities Laura Willson, MD Abbott Northwestern Hospital Dept. of Radiation Oncology February 2, 2019 Learning Objectives How radiation therapy works Standard
Corporate Medical Policy
 Corporate Medical Policy Intensity-Modulated Radiation Therapy (IMRT) of the Prostate File Name: Origination: Last CAP Review: Next CAP Review: Last Review: intensity_modulated_radiation_therapy_imrt_of_the_prostate
Corporate Medical Policy Intensity-Modulated Radiation Therapy (IMRT) of the Prostate File Name: Origination: Last CAP Review: Next CAP Review: Last Review: intensity_modulated_radiation_therapy_imrt_of_the_prostate
Protons for Head and Neck Cancer. William M Mendenhall, M.D.
 Protons for Head and Neck Cancer William M Mendenhall, M.D. Protons for Head and Neck Cancer Potential Advantages: Reduce late complications via more conformal dose distributions Likely to be the major
Protons for Head and Neck Cancer William M Mendenhall, M.D. Protons for Head and Neck Cancer Potential Advantages: Reduce late complications via more conformal dose distributions Likely to be the major
DOES RADIOTHERAPY TECHNIQUE / DOSE / FRACTIONATION REALLY MATTER? YES
 DOES RADIOTHERAPY TECHNIQUE / DOSE / FRACTIONATION REALLY MATTER? YES Marco Krengli Radiotherapy, Department of Translational Medicine, University of Piemonte Orientale A. Avogadro THE STANDARD OF CARE
DOES RADIOTHERAPY TECHNIQUE / DOSE / FRACTIONATION REALLY MATTER? YES Marco Krengli Radiotherapy, Department of Translational Medicine, University of Piemonte Orientale A. Avogadro THE STANDARD OF CARE
biij Initial experience in treating lung cancer with helical tomotherapy
 Available online at http://www.biij.org/2007/1/e2 doi: 10.2349/biij.3.1.e2 biij Biomedical Imaging and Intervention Journal CASE REPORT Initial experience in treating lung cancer with helical tomotherapy
Available online at http://www.biij.org/2007/1/e2 doi: 10.2349/biij.3.1.e2 biij Biomedical Imaging and Intervention Journal CASE REPORT Initial experience in treating lung cancer with helical tomotherapy
Collection of Recorded Radiotherapy Seminars
 IAEA Human Health Campus Collection of Recorded Radiotherapy Seminars http://humanhealth.iaea.org The Role of Radiosurgery in the Treatment of Gliomas Luis Souhami, MD Professor Department of Radiation
IAEA Human Health Campus Collection of Recorded Radiotherapy Seminars http://humanhealth.iaea.org The Role of Radiosurgery in the Treatment of Gliomas Luis Souhami, MD Professor Department of Radiation
NCCN GUIDELINES ON PROTON THERAPY (AS OF 4/23/18) BONE (Version , 03/28/18)
 BONE (Version 2.2018, 03/28/18) NCCN GUIDELINES ON PROTON THERAPY (AS OF 4/23/18) Radiation Therapy Specialized techniques such as intensity-modulated RT (IMRT); particle beam RT with protons, carbon ions,
BONE (Version 2.2018, 03/28/18) NCCN GUIDELINES ON PROTON THERAPY (AS OF 4/23/18) Radiation Therapy Specialized techniques such as intensity-modulated RT (IMRT); particle beam RT with protons, carbon ions,
An Update on Radiation Therapy for Prostate Cancer
 An Update on Radiation Therapy for Prostate Cancer David C. Beyer, MD, FACR, FACRO, FASTRO Arizona Oncology Services Phoenix, Arizona Objectives Review significant new data Identify leading trends in PCa
An Update on Radiation Therapy for Prostate Cancer David C. Beyer, MD, FACR, FACRO, FASTRO Arizona Oncology Services Phoenix, Arizona Objectives Review significant new data Identify leading trends in PCa
Feasibility of 4D IMRT Delivery for Hypofractionated High Dose Partial Prostate Treatments
 Feasibility of 4D IMRT Delivery for Hypofractionated High Dose Partial Prostate Treatments R.A. Price Jr., Ph.D., J. Li, Ph.D., A. Pollack, M.D., Ph.D.*, L. Jin, Ph.D., E. Horwitz, M.D., M. Buyyounouski,
Feasibility of 4D IMRT Delivery for Hypofractionated High Dose Partial Prostate Treatments R.A. Price Jr., Ph.D., J. Li, Ph.D., A. Pollack, M.D., Ph.D.*, L. Jin, Ph.D., E. Horwitz, M.D., M. Buyyounouski,
Disclosures. Proton therapy advantages. Why are comparing therapies difficult? Proton Therapy for Low Risk Prostate Cancer
 Proton Therapy for Low Risk Prostate Cancer Disclosures No relevant financial disclosures This presentation will not discuss off-label or investigational treatments Andrew K. Lee, MD, MPH Associate Professor
Proton Therapy for Low Risk Prostate Cancer Disclosures No relevant financial disclosures This presentation will not discuss off-label or investigational treatments Andrew K. Lee, MD, MPH Associate Professor
Dose escalation for NSCLC using conformal RT: 3D and IMRT. Hasan Murshed
 Dose escalation for NSCLC using conformal RT: 3D and IMRT. Hasan Murshed Take home message Preliminary data shows CRT technique in NSCLC allows dose escalation to an unprecedented level maintaining cancer
Dose escalation for NSCLC using conformal RT: 3D and IMRT. Hasan Murshed Take home message Preliminary data shows CRT technique in NSCLC allows dose escalation to an unprecedented level maintaining cancer
Protocol of Radiotherapy for Head and Neck Cancer
 106 年 12 月修訂 Protocol of Radiotherapy for Head and Neck Cancer Indication of radiotherapy Indication of definitive radiotherapy with or without chemotherapy (1) Resectable, but medically unfit, or high
106 年 12 月修訂 Protocol of Radiotherapy for Head and Neck Cancer Indication of radiotherapy Indication of definitive radiotherapy with or without chemotherapy (1) Resectable, but medically unfit, or high
Stereotactic radiotherapy
 Stereotactic radiotherapy Influence of patient positioning and fixation on treatment planning - clinical results Frank Zimmermann Institut für Radioonkologie Universitätsspital Basel Petersgraben 4 CH
Stereotactic radiotherapy Influence of patient positioning and fixation on treatment planning - clinical results Frank Zimmermann Institut für Radioonkologie Universitätsspital Basel Petersgraben 4 CH
Meyerhof et al. Trials (2017) 18:98 DOI /s
 Meyerhof et al. Trials (2017) 18:98 DOI 10.1186/s13063-017-1847-1 STUDY PROTOCOL Open Access Radiation-induced toxicity after imageguided and intensity-modulated radiotherapy versus external beam radiotherapy
Meyerhof et al. Trials (2017) 18:98 DOI 10.1186/s13063-017-1847-1 STUDY PROTOCOL Open Access Radiation-induced toxicity after imageguided and intensity-modulated radiotherapy versus external beam radiotherapy
Utility of 18 F-FDG PET/CT in metabolic response assessment after CyberKnife radiosurgery for early stage non-small cell lung cancer
 Utility of F-FDG PET/CT in metabolic response assessment after CyberKnife radiosurgery for early stage non-small cell lung cancer Ngoc Ha Le 1*, Hong Son Mai 1, Van Nguyen Le 2, Quang Bieu Bui 2 1 Department
Utility of F-FDG PET/CT in metabolic response assessment after CyberKnife radiosurgery for early stage non-small cell lung cancer Ngoc Ha Le 1*, Hong Son Mai 1, Van Nguyen Le 2, Quang Bieu Bui 2 1 Department
Hypofractionation in particle therapy. Marco Durante
 Hypofractionation in particle therapy Marco Durante 29.04.2014 Radiosurgery (SBRT): the new frontier in stereotactic imageguided radiotherapy Stage I (T1N0M0) NSCLC Oligometastases Hepatocellular carcinoma
Hypofractionation in particle therapy Marco Durante 29.04.2014 Radiosurgery (SBRT): the new frontier in stereotactic imageguided radiotherapy Stage I (T1N0M0) NSCLC Oligometastases Hepatocellular carcinoma
First, how does radiation work?
 Hello, I am Prajnan Das, Faculty Member in the Department of Radiation Oncology at The University of Texas MD Anderson Cancer Center. We are going to talk today about some of the basic principles regarding
Hello, I am Prajnan Das, Faculty Member in the Department of Radiation Oncology at The University of Texas MD Anderson Cancer Center. We are going to talk today about some of the basic principles regarding
Analysis of prognostic factors for survival in patients with primary spinal chordoma using the SEER Registry from 1973 to 2014
 Pan et al. Journal of Orthopaedic Surgery and Research (2018) 13:76 https://doi.org/10.1186/s13018-018-0784-3 RESEARCH ARTICLE Analysis of prognostic factors for survival in patients with primary spinal
Pan et al. Journal of Orthopaedic Surgery and Research (2018) 13:76 https://doi.org/10.1186/s13018-018-0784-3 RESEARCH ARTICLE Analysis of prognostic factors for survival in patients with primary spinal
The New ICRU/GEC ESTRO Report in Clinical Practice. Disclosures
 The New ICRU/GEC ESTRO Report in Clinical Practice Christian Kirisits, MSc, PhD; Richard Pötter, MD Medical University of Vienna, Vienna, Austria On behalf of the Committee: B. Erickson, C. Haie Meder,
The New ICRU/GEC ESTRO Report in Clinical Practice Christian Kirisits, MSc, PhD; Richard Pötter, MD Medical University of Vienna, Vienna, Austria On behalf of the Committee: B. Erickson, C. Haie Meder,
Feasibility of Proton Beam Therapy for Chordoma and Chondrosarcoma of the Skull Base
 ORIGINAL ARTICLE Feasibility of Proton Beam Therapy for Chordoma and Chondrosarcoma of the Skull Base Hiroshi Fuji, M.D., Ph.D., 1 Yoko Nakasu, M.D., Ph.D., 2 Yuji Ishida, M.D., Ph.D., 3 Satoshi Horiguchi,
ORIGINAL ARTICLE Feasibility of Proton Beam Therapy for Chordoma and Chondrosarcoma of the Skull Base Hiroshi Fuji, M.D., Ph.D., 1 Yoko Nakasu, M.D., Ph.D., 2 Yuji Ishida, M.D., Ph.D., 3 Satoshi Horiguchi,
Particle Radiation Therapy: CurrentStatus Indications -Results
 Particle Radiation Therapy: CurrentStatus Indications -Results Eugen B. Hug Center for Proton Therapy Paul Scherrer Institute and University of Zürich Switzerland Particle Radiation Therapy: Selection
Particle Radiation Therapy: CurrentStatus Indications -Results Eugen B. Hug Center for Proton Therapy Paul Scherrer Institute and University of Zürich Switzerland Particle Radiation Therapy: Selection
Specifics of treatment planning for active scanning and IMPT
 Specifics of treatment planning for active scanning and IMPT SFUD IMPT Tony Lomax, Centre for Proton Radiotherapy, Paul Scherrer Institute, Switzerland Treatment planning for scanning 1. Single Field,
Specifics of treatment planning for active scanning and IMPT SFUD IMPT Tony Lomax, Centre for Proton Radiotherapy, Paul Scherrer Institute, Switzerland Treatment planning for scanning 1. Single Field,
Treatment Planning for Skull Base Tumors PTCOG 52, June 2013
 Treatment Planning for Skull Base Tumors PTCOG 52, June 2013 Judy Adams CMD Hanne Kooy Ph.D, Norbert Liebsch MD Department of Radiation Oncology Massachusetts General Hospital Chordomas and Chondrosarcomas
Treatment Planning for Skull Base Tumors PTCOG 52, June 2013 Judy Adams CMD Hanne Kooy Ph.D, Norbert Liebsch MD Department of Radiation Oncology Massachusetts General Hospital Chordomas and Chondrosarcomas
Role of Belly Board Device in the Age of Intensity Modulated Radiotherapy for Pelvic Irradiation
 Role of Belly Board Device in the Age of Intensity Modulated Radiotherapy for Pelvic Irradiation 2017 AAMD 42 nd Annual Meeting Neil C. Estabrook, MD 6 / 14 / 2017 7/5/2017 1 Conflicts of Interest None
Role of Belly Board Device in the Age of Intensity Modulated Radiotherapy for Pelvic Irradiation 2017 AAMD 42 nd Annual Meeting Neil C. Estabrook, MD 6 / 14 / 2017 7/5/2017 1 Conflicts of Interest None
Clinical Appropriateness Guidelines: Radiation Oncology
 Clinical Appropriateness Guidelines: Radiation Oncology Proton Beam Treatment Guidelines Effective Date: September 5, 2017 Proprietary Date of Origin: 05/14/2014 Last revised: 01/08/2015 Last reviewed:
Clinical Appropriateness Guidelines: Radiation Oncology Proton Beam Treatment Guidelines Effective Date: September 5, 2017 Proprietary Date of Origin: 05/14/2014 Last revised: 01/08/2015 Last reviewed:
Mini J.Elnaggar M.D. Radiation Oncology Ochsner Medical Center 9/23/2016. Background
 Mini J.Elnaggar M.D. Radiation Oncology Ochsner Medical Center 9/23/2016 Background Mostly adenocarcinoma (scc possible, but treated like anal cancer) 39, 220 cases annually Primary treatment: surgery
Mini J.Elnaggar M.D. Radiation Oncology Ochsner Medical Center 9/23/2016 Background Mostly adenocarcinoma (scc possible, but treated like anal cancer) 39, 220 cases annually Primary treatment: surgery
Andrew K. Lee, MD, MPH Associate Professor Department tof fradiation Oncology M.D. Anderson Cancer Center
 Proton Therapy for Prostate Cancer Andrew K. Lee, MD, MPH Associate Professor Department tof fradiation Oncology M.D. Anderson Cancer Center Seungtaek Choi, MD Assistant Professor Department tof fradiation
Proton Therapy for Prostate Cancer Andrew K. Lee, MD, MPH Associate Professor Department tof fradiation Oncology M.D. Anderson Cancer Center Seungtaek Choi, MD Assistant Professor Department tof fradiation
Basic Concepts in Image Based Brachytherapy (GEC-ESTRO Target Concept & Contouring)
 Basic Concepts in Image Based Brachytherapy (GEC-ESTRO Target Concept & Contouring) Dr Umesh Mahantshetty, Professor, Radiation Oncology GYN & Urology Disease Management Group (DMG) Member Tata Memorial
Basic Concepts in Image Based Brachytherapy (GEC-ESTRO Target Concept & Contouring) Dr Umesh Mahantshetty, Professor, Radiation Oncology GYN & Urology Disease Management Group (DMG) Member Tata Memorial
-Proton Beam Therapy in Paediatric Radiation Oncology -
 -Proton Beam Therapy in Paediatric Radiation Oncology - Beate Timmermann, M.D. West German Proton Therapy Centre Essen Germany Preview Survival Toxicity Why protons? (theoretically) Experiences so far
-Proton Beam Therapy in Paediatric Radiation Oncology - Beate Timmermann, M.D. West German Proton Therapy Centre Essen Germany Preview Survival Toxicity Why protons? (theoretically) Experiences so far
Update on Sarcomas of the Head and Neck. Kevin Harrington
 Update on Sarcomas of the Head and Neck Kevin Harrington Overview Classification and incidence of sarcomas Clinical presentation Challenges to treatment Management approaches Prognostic factors Radiation-induced
Update on Sarcomas of the Head and Neck Kevin Harrington Overview Classification and incidence of sarcomas Clinical presentation Challenges to treatment Management approaches Prognostic factors Radiation-induced
Heavy Ion Therapy: Status and Perspectives
 Technology in Cancer Research & Treatment ISSN 1533-0346 Volume 2, Number 5, October (2003) Adenine Press (2003) Heavy Ion Therapy: Status and Perspectives www.tcrt.org Starting with the pioneering work
Technology in Cancer Research & Treatment ISSN 1533-0346 Volume 2, Number 5, October (2003) Adenine Press (2003) Heavy Ion Therapy: Status and Perspectives www.tcrt.org Starting with the pioneering work
Clinically Proven Metabolically-Guided TomoTherapy SM Treatments Advancing Cancer Care
 Clinically Proven Metabolically-Guided TomoTherapy SM Treatments Advancing Cancer Care Institution: San Raffaele Hospital Milan, Italy By Nadia Di Muzio, M.D., Radiotherapy Department (collaborators: Berardi
Clinically Proven Metabolically-Guided TomoTherapy SM Treatments Advancing Cancer Care Institution: San Raffaele Hospital Milan, Italy By Nadia Di Muzio, M.D., Radiotherapy Department (collaborators: Berardi
Current Status and Future Medical Perspectives at MedAustron. U. Mock EBG MedAustron GmbH
 Current Status and Future Medical Perspectives at MedAustron U. Mock EBG MedAustron GmbH Cancer treatment facility Ion beam therapy with protons and carbon ions Research facility Medical physics Radiobiology
Current Status and Future Medical Perspectives at MedAustron U. Mock EBG MedAustron GmbH Cancer treatment facility Ion beam therapy with protons and carbon ions Research facility Medical physics Radiobiology
HDR Brachytherapy: Results and Future Studies in Monotherapy
 HDR Brachytherapy: Results and Future Studies in Monotherapy Nikolaos Zamboglou and Nikolaos Tselis Strahlenklinik Klinikum Offenbach - Germany Prostate Brachytherapy UK & Ireland Conference 2013 Comparison
HDR Brachytherapy: Results and Future Studies in Monotherapy Nikolaos Zamboglou and Nikolaos Tselis Strahlenklinik Klinikum Offenbach - Germany Prostate Brachytherapy UK & Ireland Conference 2013 Comparison
CASE REPORT PLEOMORPHIC LIPOSARCOMA OF PECTORALIS MAJOR MUSCLE IN ELDERLY MAN- CASE REPORT & REVIEW OF LITERATURE.
 PLEOMORPHIC LIPOSARCOMA OF PECTORALIS MAJOR MUSCLE IN ELDERLY MAN- CASE REPORT & REVIEW OF LITERATURE. M. Madan 1, K. Nischal 2, Sharan Basavaraj. C. J 3. HOW TO CITE THIS ARTICLE: M. Madan, K. Nischal,
PLEOMORPHIC LIPOSARCOMA OF PECTORALIS MAJOR MUSCLE IN ELDERLY MAN- CASE REPORT & REVIEW OF LITERATURE. M. Madan 1, K. Nischal 2, Sharan Basavaraj. C. J 3. HOW TO CITE THIS ARTICLE: M. Madan, K. Nischal,
Cancer situation as presented by (EC 1991)
 3.1 Proton and Heavy Ion Beam Therapy G. Kraft, GSI, Biophysik, Darmstadt and J. Debus, DKFZ, Heidelberg Motivation Cancer is the second most frequent cause for disease in the developed countries. Every
3.1 Proton and Heavy Ion Beam Therapy G. Kraft, GSI, Biophysik, Darmstadt and J. Debus, DKFZ, Heidelberg Motivation Cancer is the second most frequent cause for disease in the developed countries. Every
Charged-Particle (Proton or Helium Ion) Radiation Therapy. Original Policy Date
 MP 8.01.08 Charged-Particle (Proton or Helium Ion) Radiation Therapy Medical Policy Section Therapy Issue 12/2013 Original Policy Date 12/2013 Last Review Status/Date Reviewed with literature search/12/2013
MP 8.01.08 Charged-Particle (Proton or Helium Ion) Radiation Therapy Medical Policy Section Therapy Issue 12/2013 Original Policy Date 12/2013 Last Review Status/Date Reviewed with literature search/12/2013
Opportunity for palliative care Research
 Opportunity for palliative care Research Role of Radiotherapy in Multidisciplinary Management of Rectal Cancers Dr Sushmita Pathy Associate Professor Department of Radiation Oncology Dr BRA Institute Rotary
Opportunity for palliative care Research Role of Radiotherapy in Multidisciplinary Management of Rectal Cancers Dr Sushmita Pathy Associate Professor Department of Radiation Oncology Dr BRA Institute Rotary
Proton therapy for prostate cancer
 Proton therapy for prostate cancer Shafak Aluwini Radiation oncologist UMCG Protons versus photons Superior beam properties 200 180 Advantage of protons 160 Photons 140 120 100 Spread out Bragg Peak 80
Proton therapy for prostate cancer Shafak Aluwini Radiation oncologist UMCG Protons versus photons Superior beam properties 200 180 Advantage of protons 160 Photons 140 120 100 Spread out Bragg Peak 80
LA TOMOTERAPIA IN ITALIA: ESPERIENZE A CONFRONTO
 LA TOMOTERAPIA IN ITALIA: ESPERIENZE A CONFRONTO BARD 20 NOVEMBRE 2010 DI MUZIO NADIA H. S. RAFFAELE MILANO PHASE I-II STUDY OF HYPOFRACTIONATED SIMULTANEOUS INTEGRATED BOOST WITH TOMOTHERAPY FOR PROSTATE
LA TOMOTERAPIA IN ITALIA: ESPERIENZE A CONFRONTO BARD 20 NOVEMBRE 2010 DI MUZIO NADIA H. S. RAFFAELE MILANO PHASE I-II STUDY OF HYPOFRACTIONATED SIMULTANEOUS INTEGRATED BOOST WITH TOMOTHERAPY FOR PROSTATE
Ependymoma Programme Synopsis
 Ependymoma Programme Synopsis TITLE SPONSOR PROTOCOL NUMBER EUDRACT NUMBER NATIONAL INVESTIGATOR- COORDINATOR SIOP Ependymoma program II: An International Clinical Program for the diagnosis and treatment
Ependymoma Programme Synopsis TITLE SPONSOR PROTOCOL NUMBER EUDRACT NUMBER NATIONAL INVESTIGATOR- COORDINATOR SIOP Ependymoma program II: An International Clinical Program for the diagnosis and treatment
Giant schwannoma with extensive scalloping of the lumbar vertebral body treated with one-stage posterior surgery: a case report
 Iizuka et al. Journal of Medical Case Reports 2014, 8:421 JOURNAL OF MEDICAL CASE REPORTS CASE REPORT Open Access Giant schwannoma with extensive scalloping of the lumbar vertebral body treated with one-stage
Iizuka et al. Journal of Medical Case Reports 2014, 8:421 JOURNAL OF MEDICAL CASE REPORTS CASE REPORT Open Access Giant schwannoma with extensive scalloping of the lumbar vertebral body treated with one-stage
Modelling the induction of cell death and chromosome damage by therapeutic protons
 Modelling the induction of cell death and chromosome damage by therapeutic protons M.P. Carante 1,2 and F. Ballarini 1,2, * 1 University of Pavia, Physics Department, Pavia, Italy 2 INFN, Sezione di Pavia,
Modelling the induction of cell death and chromosome damage by therapeutic protons M.P. Carante 1,2 and F. Ballarini 1,2, * 1 University of Pavia, Physics Department, Pavia, Italy 2 INFN, Sezione di Pavia,
