F rectal cancer have been almost constant over the
|
|
|
- Jade Carpenter
- 5 years ago
- Views:
Transcription
1 Screening and Rescreening for Colorectal Cancer A Controlled Trial of Fecal Occult Blood Testing in 27,700 Subjects J. KEWENTER, MD, PHD; s. BJORK, MD, PHD,* EVA HAGLIND, MD, PHD," L. SMITH, MD, PHD,' J. SVANVIK, MD, PHD; AND C. AHREN, MD, PHDt All inhabitants of the city of Goteborg who in 1982 were between 60 and 64 years of age (27,700) were randomly divided into a test and a control group. The 13,759 subjects in the test group were invited to perform Hemoccult I1 (Smith Kline Diagnostic, Sunnyvale, CA) fecal occult blood testing over 3 days and to repeat the testing after 16 to 22 months. At the first screening 9,040 (66%) completed the test, and 7,770 (58%) completed the test at the second screening. In the first screening the test group was divided into two subgroups in which the tests were rehydrated and unhydrated before development. All tests were rehydrated in the second screening; 1.9% and 5.8% of the tests were positive in the unhydrated and rehydrated subgroups, respectively. The number of diagnosed neoplasms in the first screening was significantly larger (P < 0.01) in the rehydrated group compared to the unhydrated group, 50 and 24 neoplasms, respectively. Sixteen of 61 carcinomas in the test group were found in the interval between the two screenings, 19 of the carcinomas at the second screening, and ten among the nonresponders. Rehydration of the Hemoccult I1 test is a necessity. Significantly more carcinomas (61) were found in the test group compared to the control group (20). There was a trend toward favorable tumor staging in the test group compared to the control group. Cancer 62: ,1988. IVE-YEAR SURVIVAL RATES in patients with COlO- F rectal cancer have been almost constant over the last couple of decades. This is the main reason for the interest in mass screening programs for early detection of colorectal neoplasms. Such mass screening is, for practical reasons, only possible by the detection of occult blood in feces. The test most often used for this purpose is the guaiac-impregnated filter paper test, which is not ideal as the test is not specific for hemoglobin. However, there are indications from uncontrolled studies that colorectal carcinomas diagnosed by means of guaiac screening are often localized.' A drawback of this method of screening is that other nonneoplastic bleeding lesions will also give a positive result. The aim of occult blood testing is to reduce the mortality from colorectal cancer in the screened population. From the *Departments of Surgery and tpathology, Sahlgrenska Hospital, Goteborg, Sweden. This work was supported by The Swedish Cancer Society (Grant no Bg5-04X) and Assar Gabrielsson Foundation. The authors acknowledge the considerable assistance provided by Christina Svensson, RN, and the staff of the Departments of Radiology, Pathology, the outpatient clinic, the Operation of Surgery I, and Professor Kock's laboratory. Address for reprints: Jan Kewenter, MD, Department of Surgery, Sahlgrenska Sjukhuset, S Goteborg, Sweden. Accepted for publication December 24, Whether this can be achieved can only be answered by the results of prospective controlled studies. Five such studies are underway, two in the United States and three in Europe. At the Memorial Sloan-Kettering Cancer Center the study is focused on the large bowel as the subjects in both the test group and the control group are sigmoidoscoped. Occult blood testing is offered to the subjects in the test group.* In another study performed in Minnesota 45,000 subjects 50 years or older were randomly divided into a control and two experimental groups, which are tested for occult bleeding yearly or every other year.3 The participants were selected from among health-conscious volunteers, which means that it is doubtful that the results will be representative of the population as a whole. The results will be presented in In England a study underway includes 26,975 patients from general practioners lists. The compliance in this study is low, 38.59'0.~ In Denmark a recently started study has a representative population from 45 to 74 years of age.5 Most of the ongoing studies thus focus on a special group of individuals. The need for more representative, randomized, controlled studies that reflect the general population is obvious. A controlled study in a sample that represents the general population was, therefore, started in Goteborg in The purpose of this study is to evaluate several factors 645
2 646 CANCER Augusr I 1988 Vol. 62 associated with this type of screening such as (I) the sensitivity and specificity of the test and of the methods of workup, (2) the compliance rate, (3) the distribution of the carcinomas according to the Dukes classification, (4) the effect on survival, and (5) the appropriate interval between repeated screenings. The answers to some of these questions require several more years of follow-up. First Screening Materials and Methods The first screening study was performed between August 1982 and December All 27,700 inhabitants of the city of Goteborg born between 1918 and 1922 (60 to 64 years of age in 1982) were randomly allocated to a control or a test group. The control group consisted of 13,744 people, and the test group included 13,759 people. One hundred and ninety-seven people in the test group did not participate; 145 had died before the test slides were sent out, and 52 could not be located. The subjects in the control group were never contacted. Three Hemoccult I1 tests (Smith Nine Diagnostic, Sunnyvale, CA), a letter of instruction, a questionnaire, and a postage-paid reply envelope were mailed to the subjects in the test group. The subjects were asked to perform Hemoccult I1 occult blood testing with two samples from each of three consecutive stools and to mail the slides back to the hospital immediately after the last test. Two reminder letters were sent out to subjects who did not answer. The last letter included another set of Hemoccult I1 slides. In order to test the effect of rehydration, the test group was divided into two subgroups. Slides from persons born between January 1, I9 18 and June 3 1, 1920 were not rehydrated before development, while slides from subjects born during the second half of the period were rehydrated before development. The people in the latter group were asked to maintain a low fruit and vegetable intake for two days before sample collection. The subjects in both groups were also asked to avoid food made of blood and vitamin C tablets. The time between preparation of smears and testing was less than 6 days in 96% of the subjects. The majority of the slides were smeared wet. The vast majority of the slides were developed and read by the same technician. Individuals with one or more positive tests were called to the hospital for a workup, including a personal interview during which the doctor decided whether the subject had abdominal symptoms or not (asymptomatic and symptomatic, respectively). It was noted whether the subject had observed blood in the feces during the previous six months. A digital rectal examination, proctoscopy, and rectosigmoidoscopy with a flexible instru- ment (60 cm) were performed after the interview. All subjects were then referred for a double-contrast enema (DCE) examination of the colon. Subjects with a negative workup for neoplasm or inflammatory bowel disease were asked to perform a second test series with three new Hemoccult I1 slides. Those with one or more positive tests were further examined by radiography of the stomach and by colonoscopy. Second Screening All subjects in the test group were offered rescreening 16 to 22 months (mean, 20) after the first study. The rescreening was started in April 1984 and completed in December Seven hundred forty-nine subjects died between the first and second screening, and the addresses of 22 subjects could not be found. The control and test groups consisted of 13,375 and 13,357 subjects, respectively at the start of the second screening. This screening was performed in the same way as the first one, but all slides were rehydrated before development. The subjects were asked to avoid fruit and vegetables 2 days before and during the sample collection as well as food made of blood and vitamin C tablets. The workup of subjects with a positive test was the same as in the first screening. Those who had been positive in the first screening and participated in the workup then, and were again positive in the second screening were called for colonoscopy without an ordinary workup in the second screening. No repeat Hemoccult tests, after a negative workup of an initial positive test, were done in the second screening. Follow- Up Follow-up of the whole study population is achieved by continuous quarterly contact with the local Cancer and Death Register, weekly contact with the three Departments of Surgery in the city of Goteborg, and continuous contact with the register at the Department of Pathology. Individuals leaving the area are continuously observed through the National Cancer Register. By these routines working through four different administrative channels, it should be virtually impossible for a person in our study with any diagnosed colorectal neoplasm to slip away from us. All removed neoplasms have been examined by the same pathologist. The adenomas were measured before fixation. All new batches of Hemoccult I1 were evaluated at the Reference Laboratory for Fecal Occult Blood Testing, Memorial Sloan-Kettering Cancer Center, New York City, NY. The statistical calculations were performed using Fisher s exact test significance: P <
3 No. 3 SCREENING FOR COLORECTAL CANCER - Kewenter et al. 647 TABLE 1. Comparative Data on Hydrated Versus Nonhydrated Slides in the First Screening Period No. of Complete Neoplasms Positive predict subjects Positive tests workup (true positive) value Nonhydrated group % % Hvdrated erouu % % First Screening Results Sixty-six percent of the 13,759 subjects completed both the test and the questionnaire, and 12% answered only the questionnaire. The compliance in terms of completed tests in the two subgroups with and without rehydration was 64 and 67%, respectively. Seventy-eight percent (7063) of all responders returned the completed tests after the first mailing, 15% (1393) after the first reminder letter, and 7% (584) after the second letter. Of the subjects in the unrehydrated test group, 1.9% (84 of 4436) had positive tests compared to 5.8% (265 of 4604) of the subjects in the group where the slides were rehydrated before development. The rate of true positives and the positive predictive value in the two groups is shown in Table 1. Three hundred four of the 349 subjects with a positive test series had a complete workup, 18 had a limited workup with procotoscopy and rectosigmoidoscopy, and 27 subjects refused workup. A new series of Hemoccult I1 tests was done by 206 of the 224 subjects in whom the workup had not revealed any colorectal neoplasm or inflammatory bowel disease. The new Hemoccult I1 test series was positive in 50 of these subjects (24%). Forty of these subjects underwent colonoscopy, and 43 had radiography of the stomach. One subject with carcinoma and five with adenoma were found at colonoscopy to be among those with a positive Hemoccult I1 at the repeat investigation. One polyp was found in a subject with unhydrated tests, and the other neoplasms were found in subjects with hydrated tests. Only one subject with a polyp has been diagnosed among the 156 subjects with a negative Hemoccult I1 test. No neoplasm was found in those who refused to repeat the test. All radiographs of the stomach were normal. The investigation of the 322 subjects with positive Hemoccult I1 tests revealed neoplastic disease in 74 individuals, 16 with infiltrating carcinoma and 58 with altogether 75 adenomas, 43 of which were 1 cm or larger in diameter. Four of the subjects with carcinomas and 20 with adenomas belonged to the group where the slides were not rehydrated (Table 2). Second Screening The compliance rate in the second screening is shown in Table 3. Ninety-three percent of those who submitted the slides in the second screening had also performed the test on the first occasion, and 7% (524) were new in the second screening. One thousand seven hundred eightyfive of the subjects who had performed the test in the first screening and were alive did not participate in the second screening. Eight percent (307 of 3820) positive Hemoccult I1 tests were found in those who had belonged ta the subgroup in which the slides were not rehydrated in the first TABLE 2. The Number of Subjects With Diagnosed Neoplasms in the Test and Control Groups in the First and Second Screenings and During the Interval Between the Two Screenings First screening period Second screening period* Interval No. of carcinomas No. of Total subjects Carcinomas (mean, 20 mo) subjects Carcinomas carcinomas Unhydrated group Rehydrated group Nonresponders Control group Adenomas Unhydrated group Rehydrated group Nonresponders Control group * The slides from all subjects in the second screening were rehydrated.
4 ~~ ~~ ~ 648 CANCER August I 1988 Vol. 62 TABLE 3. Compliance in the First and Second Screenings Performed Hemoccult 11 plus questionnaire Questionnaire only Nonresponders No. of subjects No. Percent No. Percent No. Percent First screening 13, Second screening 13, screening. Four point eight percent positive tests were found in those who belonged to the subgroup in which the Hemoccult slides were rehydrated in the first screening. Four hundred thirty-five (88%) of those with a positive test had a full workup, 20 had a limited workup with proctoscopy and rectosigmoidoscopy, and 42 subjects refused workup. Twenty-five subjects with a positive test in the second screening also had a positive test in the first screening. All these subjects were offered and underwent a colonoscopy. The investigation of the 455 subjects with a complete or limited workup during the second screening revealed neoplastic disease in 110 individuals (Table 2). Sixtyeight of 126 adenomas were one centimeter or larger in diameter. Combined Results of First and Second Screening Colorectal carcinomas were diagnosed in the interval between the two screening periods in 16 subjects with negative tests, in 14 in the unhydrated subgroup, add in two individuals in the rehydrated subgroup. Carcinomas were found during the same period in ten of the refusers who did not participate at all (Table 2). Altogether, 6 1 subjects with colorectal carcinomas were found in the screening group, and 20 subjects with colorectal carcinomas were found in the control group from the start of the first screening to the end of the second screening (P < 0.001, Table 2). The mean duration of this period was 27 months. The distribution of the carcinomas according to the Dukes classification in the two groups and among those in the test group who were TABLE 4. Distribution of Carcinomas According to the Dukes Classification Diagnosed with Hemoccult 11* Screening group Dukes No. Percent No. Percent A I B C D 4 11 I1 18 Total Control group No. Percent * Diagnosed with Hemoccult I1 are those cases (35/61) in the screening group that were found with the aid of the Hemoccult I1 test. diagnosed with the aid of the Hemoccult I1 test is shown in Table 4. Carcinomas were overlooked in five subjects at the initial workup of a positive Hemoccult I1 test. Three of them had a positive Hemoccult I1 at the repeat test, and Carcinomas were diagnosed in two subjects at the subsequent colonoscopy (Dukes A and C). The third repeatedly positive subject refused colonoscopy and returned due to symptoms caused by carcinoma 8 months later (Dukes D). The fourth subject had a negative Hemoccult at the repeat test, and the fifth was not offered another test. In these two subjects the carcinomas in the cecum and the transverse colon, respectively, were overlooked at the radiographic examination. The subjects returned with symptoms caused by the tumor 27 and 28 months later (Dukes C and D), respectively. The occurrence of abdominal symptoms that might be due to the diagnosed neoplasm and the presence of blood from the anus during a 6-month period before diagnosis are shown in Table 5. Only one of seven subjects with a carcinoma and abdominal symptoms had seen a doctor for these symptoms. This happened just before she was called for workup of the positive Hemoccult I1 test. Colorectal adenomas were diagnosed in 162 subjects in the test group and in 24 subjects in the control group (P < 0.01, Table 2). One hundred four and eleven, respectively, of these adenomas were one centimeter or larger in diameter (P < 0.0 1). The number of diagnosed neoplasms in the rehydrated group at the first screening was significantly (P < 0.01) larger than in the unhydrated group. The number of subjects with a diagnosed neoplasm in the interval between the two screening periods as well as the number of diagnosed neoplasms in the second screening was, conversely, significantly greater (P < 0.0 1) in the unhydrated group (Table 6). The diagnostic findings from the two screenings are shown in Table 7. Hemorrhoids were diagnosed at these times in 23 1 and 284 subjects, respectively. The hemorrhoids were considered to be a possible source of bleeding in 88 and 77 individuals, respectively, and a decision about possible bleeding in these subjects could not been made in 79 and 98 subjects, respectively. A probable
5 No. 3 SCREENING FOR COLORECTAL CANCER Kewenter et al. 649 TABLE 5. Number of Clinically Asymptomatic Subjects With Neoplasm and the Number of Subjects Who Had Not Noticed Blood From the Anus Once or More During 6 Months Before Diagnosis No abdominal No abdominal symptoms and no symptoms No history of bleeding bleeding No. of Tumor type subjects No. Percent No. Percent No. Percent All adenomas Adenomas z I cm Carcinomas source of bleeding was found in at least 48% (1 54) and 43% (197) of the worked-up subjects in the first and second screening, respectively. Diverticula were not looked upon as a source of bleeding. Discussion Rehydration of the Hemoccult 11 slide before development increases the sensitivity but decreases the specificity of the test. This means that rehydration considerably increases the work load as more subjects have to be investigated because of a false-positive test. However, if too many carcinomas are overlooked due to false-negative tests, screening will be worthless. The proportion of negative tests in subjects who were subsequently found to have a colorectal carcinoma was 72% in the subgroup with unhydrated tests. This figure is much higher than those previously reported but similar to that postulated by Simon.' In the rehydrated group the number of subjects with carcinoma and a false-negative Hemoccult test was much lower (7%), as could be expected. Our results are thus in agreement with those obtained in the Minnesota study; i.e. Hemoccult I1 slides must be rehydrated before de~elopment.~ We could not confirm the claim that rehydration does not increase the diagnostic yield.8 If the slides are not rehydrated, the incidence of diagnosed colorectal carcinomas will be about the same as among the nonresponders. We therefore disagree with those who do not recommend rehydration of the Hemoccult I1 before de~elopment.~.~ The increased sensitivity after rehydration means that the number of false-positive tests increases strongly as the specificity decreases. In order to reduce the number of false-positive tests after rehydration, it has been suggested that the subjects should keep to a low meat- and peroxidase-containing diet before and during the test period as such food can cause a false-positive test."." However, Norfelt et al. could not show any effect on sensitivity or specificity of a red meat-free diet.i2 We do not know whether the dietary restrictions in our program had an impact on the number of false-positive tests. The rate of false-positive tests, here defined as the absence of neoplasms, was 71% and 83% in the unhy- drated and rehydrated groups, respectively. Another way to reduce the number of false-positive tests might be to reexamine those with a positive rehydrated test and only investigate the subjects who are positive at the second testing.i3 Strictly speaking, false-positive tests are only those that are positive due to other causes than blood as the guaiac test measures the presence of blood and not the presence of neoplasm. Only about half of the positive tests were found to be due to a possible source of bleeding when some of the diagnosed hemorrhoids, inflammatory bowel disease, and neoplasms were looked upon as possible sources. Diverticula in the colon were not considered a source.14 A blood-specific test like the HemoquantI5 (Smith Mine Diagnostic, Sunnyvale, CA) might, therefore, possibly reduce the number of all positive tests to about half without decreasing the high sensitivity obtained with rehydrated Hemoccult I1 tests. Screening for the early detection of colorectal cancer requires a considerable effort in order to reach a compliance of 68%. A great deal of effort is also needed to achieve a high compliance rate in the workup of those with a positive test. The population to be screened, therefore, has to be carefully selected in order to get the most benefit from the available resources. Screening with the guaiac test will always result in a number of false-positive tests, which means that the positive predictive value will decrease with decreasing incidence of neoplasms in the screened population.16 The incidence TABLE 6. Number of Diagnosed Neoplasms in the Unhydrated and Rehydrated Groups in the First and Second Screening and During the Intervening Interval No. of First Second subjects screening Interval screening* Unhydrated group Rehydrated group P < P < 0.00 I P < * The slides from all subjects in the second screening were rehydrated before development.
6 650 CANCER August I 1988 Vol. 62 TABLE 7. Diagnostic Findings in the Two Screenings in Subjects With a Positive Hemoccult I1 Test No. of subjects examined for Percent a positive Percent inflammatory Percent Percent Hemoccult neoplasms bowel disease hemorrhoids diverticula First screening Second screening of colorectal cancer in individuals between 45 and 49 years of age is only 30% of that in individuals between 55 and 59 years of age and only 20% of that in individuals between 65 and 69 years of age.17 Forty-five years has generally been chosen as the age at which screening shall start, but no scientific evidence for this decision seems to exi~t.~.~ In our mind screening for colorectal cancer should start at the age of 55 as it seems likely that the diagnostic yield will thereby increase. The diagnostic methods used in the workup of positive tests are important for the outcome of the screening program.i8 Rectoscopy with a rigid instrument and barium enema is too unsatisfactory to be used. Colonoscopy is considered to be a necessity in the Minnesota study, while we consider double-contrast barium enema and rectosigmoidoscopy (60 cm) to be satisfactory for the e~amination.~. ~ To our knowledge, there is no controlled study that has shown better results with colonoscopy. In the present study five carcinomas were overlooked. One of the overlooked carcinomas was diagnosed at colonoscopy performed as a consequence of a positive test at a repeat fecal examination and another was diagnosed at colonoscopy performed to remove a diagnosed adenoma. The other three carcinomas were diagnosed only as a result of the fact that they caused symptoms reputed by individuals. We do not know how often and at what intervals the screening should be repeated in order to get the best results. According to our results, at least one rescreening within 12 to 20 months seems to be necessary when the slides are rehydrated as 30% of the carcinomas and about 50% of the adenomas were found at rescreening in this group. Whether a second rescreening is of any benefit is still unknown, but it might be possible to determine its value from the results of continued observation of our population. The number of asymptomatic subjects with colorectal neoplasms vanes with different screening materials. The relationship between individuals with and without gastrointestinal symptoms and a colorectal neoplasm in a screening material is important in the evaluation of the screening. This relationship most likely reflects the possible admixture of subjects who have been included as they have consulted for symptoms caused by a colorec- tal neoplasm and not found by the screening procedure. In the present material 80% of the subjects with a diagnosed carcinoma and 91% of those with an adenoma had not noticed any abdominal symptoms. Some of these subjects had noticed bleeding from the anus within 6 months before the screening. However, bleeding in this age group is a rather unspecific symptom because no less than 12% reported that they had noticed blood from the anus one or more times during a 6-month period before the screening. Forty-three percent of the subjects with carcinomas and 60% of the subjects with an adenoma were clinically asymptomatic and had never noticed any bleeding. The distribution of the diagnosed colorectal carcinomas according to the Dukes classification varies with different materials. Twelve of twenty diagnosed carcinomas in Hardcastle s material belonged to Dukes A.4 In a study in Germany about 70% of the carcinomas were of Dukes A or B type, and in the Minnesota study 61% of the carcinomas were classified as Dukes A or B. In this study there was no significant difference in the Dukes classification between the diagnosed carcinomas in the test and control group, although three times more carcinomas were diagnosed in the test group compared to the control group (6 1 and 20 carcinomas, respectively). If the staging of the carcinoma diagnosed with the aid of Hemoccult I1 was compared with the carcinomas in the control group, there was a clear trend toward more favorable tumor staging in the test group. The reason for the discrepancy between this and other materials could be that only a little more than half of the carcinomas in the test group were diagnosed with the aid of the Hemoccult 11. Thus, 14 subjects with carcinomas had a false-negative test (1 3 in the unhydrated subgroup), the carcinomas were overlooked during the double-contrast enema in two individuals, and ten carcinomas were found among the nonresponders. All these carcinomas were diagnosed due to the subjects symptoms. Another reason for the difference in Dukes classification between this and some other materials might be that all subjects but one with malignant adenomas had a large bowel resection. Surprisingly, two of these subjects had metastases to the lymph nodes. If these subjects had not been operated on, they would have been classified as
7 No. 3 SCREENING FOR COLORECTAL CANCER - Kewenter et al. 651 Dukes A instead of Dukes C. One must remember that Dukes classification is impossible to perform if the lymph nodes are not microscopically examined. In order to get the best possible results with this method of screening, special attention must be paid to three factors: the sensitivity of the test, the compliance, and the sensitivity of the methods used for workup of a positive test. For the purpose of increasing the possibilities of obtaining an answer to these questions, the screening study has been increased to twice as many subjects (51,477). The second half of this study is in progress. REFERENCES 1. Gnauck R. Praktische Erfahrungen mit der Stuhltestung auf okkultes Blut. Schweiz Med wschr 1983; Winawer SJ, Andrews M, Flehinger B et al. Progress report on controlled trial of fecal occult blood testing for the detection of colorectal neoplasia. Cancer 1980; 45: Gilbertsen VA, Church TR, Grewe FJ. The design of a study to assess occult-blood screening for colon cancer. J Chron Dis 1980; 33: Hardcastle JD, Armitage NC, Camberlain J, Amar SS, James PD, Balfour TW. Fecal occult blood screening for colorectal cancer in the general population. Cancer 1986; 58: Kronborg 0, Fenger C, Sodergaard 0 et a!. Initial mass screening for colorectal cancer fecal occult blood test: A prospective randomized study at Funen in Denmark (in press). 6. Kewenter J, Svensson C, Bjorck S el al. A randomized controlled trial of fecal occult blood testing for the detection of colorectal cancer: A preliminary report of a study including 27,503 persons between 60 and 64 years of age. In: Hardcastle JD, ed. Hemoccult Screening for the Early Detection of Colorectal Cancer, 1984; Simon BS. Occult blood screening for colorectal carcinoma: A critical review. Gastroenterology 1985; 88: Winawer SJ, Andrews M, Miller CH ef al. Review of screening for colorectal cancer using fecal occult blood testing. In: Winawer SJ, Schottenfeld D, Sherlock P, eds. Colorectal Cancer: Prevention, Epidemiology, and Screening. New York Raven Press, 1980; Hammes PH, Gnauck R. Verbessert Rehydrieren das Haemoccult-Screening nach Darmkrebs? Z. Gastroenterologie 1985; 23~ Macrae FA, St. John DJB, Caligiore P et al. Optimal dietary conditions for Hemoccult testing. Gastroenterology 1982; 82: Winawer SJ, Fleisher M. Sensitivity and specificity of the fecal occult blood test for colorectal neoplasia. Gastroenterology 1982; 82: Nortleet RG. Effect of diet on fecal occult blood testing in patients with colorectal polyps. Dig Dis Sci 1986; 31: Elliot MS, Levenstein JH, Wright JP. Fecal occult blood testing in the detection of colorectal cancer. Br JSurg 1984; 71: Kewenter J, Hellzen-Ingermarsson A, Kewenter G et al. Diverticular disease and minor rectal bleeding. Scand J Gastroenterot 1985; 20: Schwartz S, Dahl J, Ellefson M et al. The Hemoquant test: A specific and quantitative determination of heme (hemoglobin) in feces and other materials. Clin Chern 1983; 29: Fletcher H, Fletcher S, Wagner E. Diagnostic Test in Clinical Epidemiology. Baltimore: Williams and Witkins, 1982; Cancer incidence in Sweden National Board of Health and Welfare. The Cancer Registry, Stockholm. Sweden. 18. Jensen J, Kewenter J, Haglind E et al. Diagnostic accuracy of double-contrast enema and rectosigmoidoscopy in connection with fecal occult blood testing for the detection of rectosigmoid neoplasms. Br J Surg 1986; 73: Mandel JS. The University of Minnesota s Colon Cancer Control Study: Design and Progress to Date, 1986 (to be published).
Randomised study of screening for colorectal cancer with faecaloccult-blood
 Articles Randomised study of screening for colorectal cancer with faecaloccult-blood test Ole Kronborg, Claus Fenger, Jørn Olsen, Ole Dan Jørgensen, Ole Søndergaard Summary Background Case-control studies
Articles Randomised study of screening for colorectal cancer with faecaloccult-blood test Ole Kronborg, Claus Fenger, Jørn Olsen, Ole Dan Jørgensen, Ole Søndergaard Summary Background Case-control studies
Colorectal Cancer Screening What are my options?
 069-Colorectal cancer (Rosen) 1/23/04 12:59 PM Page 69 What are my options? Wayne Rosen, MD, FRCSC As presented at the 37th Annual Mackid Symposium: Cancer Care in the Community (May 22, 2003) There are
069-Colorectal cancer (Rosen) 1/23/04 12:59 PM Page 69 What are my options? Wayne Rosen, MD, FRCSC As presented at the 37th Annual Mackid Symposium: Cancer Care in the Community (May 22, 2003) There are
SCREENING FOR COLON AND RECTAL CANCER
 Review Article SCREENING FOR COLON AND RECTAL CANCER Jack S. Mandel, PhD, MPH School of Public Health, University of Minnesota Introduction Approximately 138,200 new cases and 55,300 deaths occured in
Review Article SCREENING FOR COLON AND RECTAL CANCER Jack S. Mandel, PhD, MPH School of Public Health, University of Minnesota Introduction Approximately 138,200 new cases and 55,300 deaths occured in
Frequency of Diagnosis of Colorectal Cancer with Double Contrast Barium Enema
 Bahrain Medical Bulletin, Vol.24, No.3, September 2002 Frequency of Diagnosis of Colorectal Cancer with Double Contrast Barium Enema Najeeb S Jamsheer, MD, FRCR* Neelam. Malik, MD, MNAMS** Objective: To
Bahrain Medical Bulletin, Vol.24, No.3, September 2002 Frequency of Diagnosis of Colorectal Cancer with Double Contrast Barium Enema Najeeb S Jamsheer, MD, FRCR* Neelam. Malik, MD, MNAMS** Objective: To
treated successfully. The predictive value of a positive test varied
 Detecting Colorectal Cancer with a Large Scale Fecal Occult Blood Testing Program DAVID 1. GREGORIO, PhD, MS PERI LOLACHI, MPH HOLGER HANSEN, MD, DrPH The authors are with the University of Connecticut,
Detecting Colorectal Cancer with a Large Scale Fecal Occult Blood Testing Program DAVID 1. GREGORIO, PhD, MS PERI LOLACHI, MPH HOLGER HANSEN, MD, DrPH The authors are with the University of Connecticut,
GUAIAC tests for fecal occult blood detect the pseudoperoxidase
 Vol. 334 No. 3 COMPARISON OF FECAL OCCULT-BLOOD TESTS FOR COLORECTAL-CANCER SCREENING 155 A COMPARISON OF FECAL OCCULT-BLOOD TESTS FOR COLORECTAL-CANCER SCREENING JAMES E. ALLISON, M.D., IRENE S. TEKAWA,
Vol. 334 No. 3 COMPARISON OF FECAL OCCULT-BLOOD TESTS FOR COLORECTAL-CANCER SCREENING 155 A COMPARISON OF FECAL OCCULT-BLOOD TESTS FOR COLORECTAL-CANCER SCREENING JAMES E. ALLISON, M.D., IRENE S. TEKAWA,
patients over the age of 40
 Postgraduate Medical Journal (1988) 64, 364-368 Frank rectal bleeding: a prospective study of causes in patients over the age of 40 P.S.Y. Cheung, S.K.C. Wong, J. Boey and C.K. Lai Department of Surgery,
Postgraduate Medical Journal (1988) 64, 364-368 Frank rectal bleeding: a prospective study of causes in patients over the age of 40 P.S.Y. Cheung, S.K.C. Wong, J. Boey and C.K. Lai Department of Surgery,
Updates in Colorectal Cancer Screening & Prevention
 Updates in Colorectal Cancer Screening & Prevention Swati G. Patel, MD MS Assistant Professor of Medicine Division of Gastroenterology & Hepatology Gastrointestinal Cancer Risk and Prevention Clinic University
Updates in Colorectal Cancer Screening & Prevention Swati G. Patel, MD MS Assistant Professor of Medicine Division of Gastroenterology & Hepatology Gastrointestinal Cancer Risk and Prevention Clinic University
Screening for colorectal cancer
 Postgrad Med J (1994) 70, 469-474 A) The Fellowship of Postgraduate Medicine, 1994 Review Article Screening for colorectal cancer D.H. Bennett and J.D. Hardcastle Department ofsurgery, E Floor, West Block,
Postgrad Med J (1994) 70, 469-474 A) The Fellowship of Postgraduate Medicine, 1994 Review Article Screening for colorectal cancer D.H. Bennett and J.D. Hardcastle Department ofsurgery, E Floor, West Block,
The Canadian Cancer Society estimates that in
 How Do I Screen For Colorectal Cancer? By Ted M. Ross, MD, FRCS(C); and Naomi Ross, RD, BSc To be presented at the University of Toronto s Primary Care Today sessions (October 3, 2003) The Canadian Cancer
How Do I Screen For Colorectal Cancer? By Ted M. Ross, MD, FRCS(C); and Naomi Ross, RD, BSc To be presented at the University of Toronto s Primary Care Today sessions (October 3, 2003) The Canadian Cancer
Screening for colorectal cancer in a factory-based population
 Br. J. Cancer (1983), 48, 843-847 Screening for colorectal cancer in a factory-based population with Fecatest F.I. Lee Department of Gastroenterology, Victoria Hospital, Blackpool. Summary This report
Br. J. Cancer (1983), 48, 843-847 Screening for colorectal cancer in a factory-based population with Fecatest F.I. Lee Department of Gastroenterology, Victoria Hospital, Blackpool. Summary This report
C cancer and the second leading cause of cancer
 Screening for Colorectal Cancer Using the Hemoccult /I Stool Guaiac Slide Test K. MICHAEL CUMMINGS, PHD, MPH,' ARTHUR MICHALEK, PHD,' CURTIS METTLIN, PHD,' AND ARNOLD MITTELMAN, MDt Following a series
Screening for Colorectal Cancer Using the Hemoccult /I Stool Guaiac Slide Test K. MICHAEL CUMMINGS, PHD, MPH,' ARTHUR MICHALEK, PHD,' CURTIS METTLIN, PHD,' AND ARNOLD MITTELMAN, MDt Following a series
Colorectal cancer screening
 26 Colorectal cancer screening BETHAN GRAF AND JOHN MARTIN Colorectal cancer is theoretically a preventable disease and is ideally suited to a population screening programme, as there is a long premalignant
26 Colorectal cancer screening BETHAN GRAF AND JOHN MARTIN Colorectal cancer is theoretically a preventable disease and is ideally suited to a population screening programme, as there is a long premalignant
Written By: Joann O Connor Date: 01/27/2017 Effective Date: 02/22/2017 1
 Department Of Pathology Point of Care Testing POC.516.12 Hemoccult-LBH Version #12 Printed copies are for reference only. Please refer to the electronic copy for the latest version. A. PURPOSE: The Hemoccult
Department Of Pathology Point of Care Testing POC.516.12 Hemoccult-LBH Version #12 Printed copies are for reference only. Please refer to the electronic copy for the latest version. A. PURPOSE: The Hemoccult
A Clinician's Guide: Tips on
 The following reprint is provided with the compliments of Cenogenics Corporation, 620 Route 520, Morganvile, NJ 07751 (908) 536-6457. A Clinician's Guide: Tips on Fecal Occult Blood Testing Questions from
The following reprint is provided with the compliments of Cenogenics Corporation, 620 Route 520, Morganvile, NJ 07751 (908) 536-6457. A Clinician's Guide: Tips on Fecal Occult Blood Testing Questions from
FECAL OCCULT BLOOD TEST
 MEDICAL POLICY For use with the UnitedHealthcare Laboratory Benefit Management Program, administered by BeaconLBS FECAL OCCULT BLOOD TEST Policy Number: CMP - 023 Effective Date: January 1, 2018 Table
MEDICAL POLICY For use with the UnitedHealthcare Laboratory Benefit Management Program, administered by BeaconLBS FECAL OCCULT BLOOD TEST Policy Number: CMP - 023 Effective Date: January 1, 2018 Table
THE EFFECT OF FECAL OCCULT-BLOOD SCREENING ON THE INCIDENCE OF COLORECTAL CANCER
 THE EFFECT OF FECAL OCCULT-BLOOD SCREENING ON THE INCIDENCE OF COLORECTAL CANCER JACK S. MANDEL, PH.D., M.P.H., TIMOTHY R. CHURCH, PH.D., JOHN H. BOND, M.D., FRED EDERER, M.A., MINDY S. GEISSER, M.S.,
THE EFFECT OF FECAL OCCULT-BLOOD SCREENING ON THE INCIDENCE OF COLORECTAL CANCER JACK S. MANDEL, PH.D., M.P.H., TIMOTHY R. CHURCH, PH.D., JOHN H. BOND, M.D., FRED EDERER, M.A., MINDY S. GEISSER, M.S.,
Performance Characteristics and Comparison of Two Fecal Occult Blood Tests in Patients Undergoing Colonoscopy
 Dig Dis Sci (2007) 52:1009 1013 DOI 10.1007/s10620-006-9383-y ORIGINAL ARTICLE Performance Characteristics and Comparison of Two Fecal Occult Blood Tests in Patients Undergoing Colonoscopy Marcia Cruz-Correa
Dig Dis Sci (2007) 52:1009 1013 DOI 10.1007/s10620-006-9383-y ORIGINAL ARTICLE Performance Characteristics and Comparison of Two Fecal Occult Blood Tests in Patients Undergoing Colonoscopy Marcia Cruz-Correa
Structured Follow-Up after Colorectal Cancer Resection: Overrated. R. Taylor Ripley University of Colorado Grand Rounds April 23, 2007
 Structured Follow-Up after Colorectal Cancer Resection: Overrated R. Taylor Ripley University of Colorado Grand Rounds April 23, 2007 Guidelines for Colonoscopy Production: Surveillance US Multi-Society
Structured Follow-Up after Colorectal Cancer Resection: Overrated R. Taylor Ripley University of Colorado Grand Rounds April 23, 2007 Guidelines for Colonoscopy Production: Surveillance US Multi-Society
Papers. A systematic review of the effects of screening for colorectal cancer using the faecal occult blood test, Hemoccult. Abstract.
 A systematic review of the effects of screening for colorectal cancer using the faecal occult blood test, Hemoccult Bernie Towler, Les Irwig, Paul Glasziou, Jan Kewenter, David Weller, Chris Silagy Abstract
A systematic review of the effects of screening for colorectal cancer using the faecal occult blood test, Hemoccult Bernie Towler, Les Irwig, Paul Glasziou, Jan Kewenter, David Weller, Chris Silagy Abstract
Prognosis after Treatment of Villous Adenomas
 Prognosis after Treatment of Villous Adenomas of the Colon and Rectum JOHN CHRISTIANSEN, M.D., PREBEN KIRKEGAARD, M.D., JYTTE IBSEN, M.D. With the existing evidence of neoplastic polyps of the colon and
Prognosis after Treatment of Villous Adenomas of the Colon and Rectum JOHN CHRISTIANSEN, M.D., PREBEN KIRKEGAARD, M.D., JYTTE IBSEN, M.D. With the existing evidence of neoplastic polyps of the colon and
COLORECTAL CANCER FAISALGHANISIDDIQUI MBBS; FCPS; PGDIP-BIOETHICS; MCPS-HPE
 COLORECTAL CANCER FAISALGHANISIDDIQUI MBBS; FCPS; PGDIP-BIOETHICS; MCPS-HPE PROFESSOR OF SURGERY & DIRECTOR, PROFESSIONAL DEVELOPMENT CENTRE J I N N A H S I N D H M E D I C A L U N I V E R S I T Y faisal.siddiqui@jsmu.edu.pk
COLORECTAL CANCER FAISALGHANISIDDIQUI MBBS; FCPS; PGDIP-BIOETHICS; MCPS-HPE PROFESSOR OF SURGERY & DIRECTOR, PROFESSIONAL DEVELOPMENT CENTRE J I N N A H S I N D H M E D I C A L U N I V E R S I T Y faisal.siddiqui@jsmu.edu.pk
Fecal occult blood tests: a cost-effectiveness analysis Gyrd-Hansen D
 Fecal occult blood tests: a cost-effectiveness analysis Gyrd-Hansen D Record Status This is a critical abstract of an economic evaluation that meets the criteria for inclusion on NHS EED. Each abstract
Fecal occult blood tests: a cost-effectiveness analysis Gyrd-Hansen D Record Status This is a critical abstract of an economic evaluation that meets the criteria for inclusion on NHS EED. Each abstract
removal of adenomatous polyps detects important effectively as follow-up colonoscopy after both constitute a low-risk Patients with 1 or 2
 Supplementary Table 1. Study Characteristics Author, yr Design Winawer et al., 6 1993 National Polyp Study Jorgensen et al., 9 1995 Funen Adenoma Follow-up Study USA Multi-center, RCT for timing of surveillance
Supplementary Table 1. Study Characteristics Author, yr Design Winawer et al., 6 1993 National Polyp Study Jorgensen et al., 9 1995 Funen Adenoma Follow-up Study USA Multi-center, RCT for timing of surveillance
CLINICAL PRACTICE GUIDELINE FOR COLORECTAL CANCER SCREENING
 CLINICAL PRACTICE GUIDELINE FOR COLORECTAL CANCER SCREENING This guideline is designed to assist practitioners by providing the framework for colorectal cancer (CRC) screening, and is not intended to replace
CLINICAL PRACTICE GUIDELINE FOR COLORECTAL CANCER SCREENING This guideline is designed to assist practitioners by providing the framework for colorectal cancer (CRC) screening, and is not intended to replace
Colorectal Cancer Screening
 Colorectal Cancer Screening An Integrated Care Pathway of the Collaborative Care Network Subject Matter Expert: Kevin Wolov, DO Pathway Custodian: Pat Czapp, MD First, a Friendly Reminder... This Integrated
Colorectal Cancer Screening An Integrated Care Pathway of the Collaborative Care Network Subject Matter Expert: Kevin Wolov, DO Pathway Custodian: Pat Czapp, MD First, a Friendly Reminder... This Integrated
C olorectal adenomas are reputed to be precancerous
 568 COLORECTAL CANCER Incidence and recurrence rates of colorectal adenomas estimated by annually repeated colonoscopies on asymptomatic Japanese Y Yamaji, T Mitsushima, H Ikuma, H Watabe, M Okamoto, T
568 COLORECTAL CANCER Incidence and recurrence rates of colorectal adenomas estimated by annually repeated colonoscopies on asymptomatic Japanese Y Yamaji, T Mitsushima, H Ikuma, H Watabe, M Okamoto, T
Faecal Immunochemical Testing (FIT) for Screening and Symptomatic Patients
 Faecal Immunochemical Testing (FIT) for Screening and Symptomatic Patients Caroline Addison NE BCSP Hub Director and Consultant Clinical Scientist What is FIT Type of Faecal Occult Blood test Designed
Faecal Immunochemical Testing (FIT) for Screening and Symptomatic Patients Caroline Addison NE BCSP Hub Director and Consultant Clinical Scientist What is FIT Type of Faecal Occult Blood test Designed
Increasing Trend in the Incidence of Colorectal Cancer in Japan
 Cancer Increasing Trend in the Incidence of Colorectal Cancer in Japan JMAJ 46(6): 251 256, 2003 Susumu KODAIRA Professor, Department of Surgery, Teikyo University School of Medicine Abstract: Malignant
Cancer Increasing Trend in the Incidence of Colorectal Cancer in Japan JMAJ 46(6): 251 256, 2003 Susumu KODAIRA Professor, Department of Surgery, Teikyo University School of Medicine Abstract: Malignant
Cost-effectiveness Model for Colon Cancer Screening
 GASTROENTEROLOGY 1995;109:1781-1790 Cost-effectiveness Model for Colon Cancer Screening DAVID A. LIEBERMAN Division of Gastroenterology, Department of Medicine, Oregon Health Sciences University, and Portland
GASTROENTEROLOGY 1995;109:1781-1790 Cost-effectiveness Model for Colon Cancer Screening DAVID A. LIEBERMAN Division of Gastroenterology, Department of Medicine, Oregon Health Sciences University, and Portland
Screening for Colorectal Cancer in British Columbia.
 Screening for Colorectal Cancer in British Columbia. Iain G.M.Cleator, Robert Parson, Josefina Baker, Andrew Rae, Gregory McGregor, Gregory Hislop, David Klaassen, Walter MacDonald, Brenda Morrison, Andrew
Screening for Colorectal Cancer in British Columbia. Iain G.M.Cleator, Robert Parson, Josefina Baker, Andrew Rae, Gregory McGregor, Gregory Hislop, David Klaassen, Walter MacDonald, Brenda Morrison, Andrew
CRC Risk Factors. U.S. Adherence Rates Cancer Screening. Genetic Model of Colorectal Cancer. Epidemiology and Clinical Consequences of CRC
 10:45 11:45 am Guide to Colorectal Cancer Screening SPEAKER Howard Manten M.D. Presenter Disclosure Information The following relationships exist related to this presentation: Howard Manten MD: No financial
10:45 11:45 am Guide to Colorectal Cancer Screening SPEAKER Howard Manten M.D. Presenter Disclosure Information The following relationships exist related to this presentation: Howard Manten MD: No financial
FECAL OCCULT BLOOD TEST (FOBT) Common Guaiac versus Immunochemical Test
 FECAL OCCULT BLOOD TEST (FOBT) Common Guaiac versus Immunochemical Test LIMBACH-LABORATORY H E I D E L B E R G H J Roth H Schmidt-Gayk Estimated incidence of cancer in Europe and European Union, 2006 Limbach
FECAL OCCULT BLOOD TEST (FOBT) Common Guaiac versus Immunochemical Test LIMBACH-LABORATORY H E I D E L B E R G H J Roth H Schmidt-Gayk Estimated incidence of cancer in Europe and European Union, 2006 Limbach
Citation for published version (APA): Wijkerslooth de Weerdesteyn, T. R. (2013). Population screening for colorectal cancer by colonoscopy
 UvA-DARE (Digital Academic Repository) Population screening for colorectal cancer by colonoscopy de Wijkerslooth, T.R. Link to publication Citation for published version (APA): Wijkerslooth de Weerdesteyn,
UvA-DARE (Digital Academic Repository) Population screening for colorectal cancer by colonoscopy de Wijkerslooth, T.R. Link to publication Citation for published version (APA): Wijkerslooth de Weerdesteyn,
COLON CANCER SCREENING: AN UPDATE
 Overview COLON CANCER SCREENING: AN UPDATE Siddharth Verma, DO, JD Rutgers New Jersey Medical School Background Screening Updates in Specific Populations African Americans CRC in the younger age USPSTF
Overview COLON CANCER SCREENING: AN UPDATE Siddharth Verma, DO, JD Rutgers New Jersey Medical School Background Screening Updates in Specific Populations African Americans CRC in the younger age USPSTF
Colorectal Cancer Screening. Dr Kishor Muniyappa 2626 Care Drive, Suite 101 Tallahassee, FL Ph:
 Colorectal Cancer Screening Dr Kishor Muniyappa 2626 Care Drive, Suite 101 Tallahassee, FL 32308 Ph: 850-297-0351 What we ll be talking about How common is colorectal cancer? What is colorectal cancer?
Colorectal Cancer Screening Dr Kishor Muniyappa 2626 Care Drive, Suite 101 Tallahassee, FL 32308 Ph: 850-297-0351 What we ll be talking about How common is colorectal cancer? What is colorectal cancer?
JOHN H. STROGER HOSPITAL OF COOK COUNTY ANNUAL TRAINING HEMOCCULT
 JOHN H. STROGER HOSPITAL OF COOK COUNTY ANNUAL TRAINING HEMOCCULT Syn: Fecal Occult Blood Test, Guiac Test PURPOSE: Hemoccult Test is a qualitative screening method for detecting fecal occult blood which
JOHN H. STROGER HOSPITAL OF COOK COUNTY ANNUAL TRAINING HEMOCCULT Syn: Fecal Occult Blood Test, Guiac Test PURPOSE: Hemoccult Test is a qualitative screening method for detecting fecal occult blood which
Global colorectal cancer screening appropriate or practical? Graeme P Young, Flinders University WCC, Melbourne
 Global colorectal cancer screening appropriate or practical? Graeme P Young, Flinders University. 2014 WCC, Melbourne Outline WHO criteria to justify screening Appropriateness: Global variation in incidence
Global colorectal cancer screening appropriate or practical? Graeme P Young, Flinders University. 2014 WCC, Melbourne Outline WHO criteria to justify screening Appropriateness: Global variation in incidence
Joint Session with ACOFP and Cancer Treatment Centers of America (CTCA): Cancer Screening: Consensus & Controversies. Ashish Sangal, M.D.
 Joint Session with ACOFP and Cancer Treatment Centers of America (CTCA): Cancer Screening: Consensus & Controversies Ashish Sangal, M.D. Cancer Screening: Consensus & Controversies Ashish Sangal, MD Director,
Joint Session with ACOFP and Cancer Treatment Centers of America (CTCA): Cancer Screening: Consensus & Controversies Ashish Sangal, M.D. Cancer Screening: Consensus & Controversies Ashish Sangal, MD Director,
Alberta Colorectal Cancer Screening Program (ACRCSP) Post Polypectomy Surveillance Guidelines
 Alberta Colorectal Cancer Screening Program (ACRCSP) Post Polypectomy Surveillance Guidelines June 2013 ACRCSP Post Polypectomy Surveillance Guidelines - 2 TABLE OF CONTENTS Background... 3 Terms, Definitions
Alberta Colorectal Cancer Screening Program (ACRCSP) Post Polypectomy Surveillance Guidelines June 2013 ACRCSP Post Polypectomy Surveillance Guidelines - 2 TABLE OF CONTENTS Background... 3 Terms, Definitions
Estimation of screening test (Hemoccult ) sensitivity in colorectal cancer mass screening
 doi: 1.154/ bjoc.21.1752, available online at http://www.idealibrary.com on http://www.bjcancer.com Estimation of screening test (Hemoccult ) sensitivity in colorectal cancer mass screening JL Jouve 1,
doi: 1.154/ bjoc.21.1752, available online at http://www.idealibrary.com on http://www.bjcancer.com Estimation of screening test (Hemoccult ) sensitivity in colorectal cancer mass screening JL Jouve 1,
Get tested for. Colorectal cancer. Doctors know how to prevent colon or rectal cancer- and you can, too. Take a look inside.
 Get tested for Colorectal cancer Doctors know how to prevent colon or rectal cancer- and you can, too. Take a look inside. 1 If you re 50 or older, you need to get tested for colorectal cancer. It s one
Get tested for Colorectal cancer Doctors know how to prevent colon or rectal cancer- and you can, too. Take a look inside. 1 If you re 50 or older, you need to get tested for colorectal cancer. It s one
HARBOR-UCLA MEDICAL CENTER Torrance, California DEPARTMENT OF EMERGENCY MEDICINE POLICY AND PROCEDURE MANUAL
 25.5 Point of Care Testing Occult Blood Testing by Physicians POLICY The Hemoccult test is a rapid convenient and virtually odorless qualitative method for detecting fecal occult blood. It is used as a
25.5 Point of Care Testing Occult Blood Testing by Physicians POLICY The Hemoccult test is a rapid convenient and virtually odorless qualitative method for detecting fecal occult blood. It is used as a
C olorectal cancer (CRC) is the second most common
 CANCER Effect of faecal occult blood screening on mortality from colorectal cancer: results from a randomised controlled trial J H Scholefield, S Moss, F Sufi, C M Mangham, J D Hardcastle... See end of
CANCER Effect of faecal occult blood screening on mortality from colorectal cancer: results from a randomised controlled trial J H Scholefield, S Moss, F Sufi, C M Mangham, J D Hardcastle... See end of
Incidence and Management of Hemorrhage after Endoscopic Removal of Colorectal Lesions
 Showa Univ J Med Sci 12(3), 253-258, September 2000 Original Incidence and Management of Hemorrhage after Endoscopic Removal of Colorectal Lesions Masaaki MATSUKAWA, Mototsugu FUJIMORI, Takahiko KOUDA,
Showa Univ J Med Sci 12(3), 253-258, September 2000 Original Incidence and Management of Hemorrhage after Endoscopic Removal of Colorectal Lesions Masaaki MATSUKAWA, Mototsugu FUJIMORI, Takahiko KOUDA,
Colorectal Cancer Screening. Paul Berg MD
 Colorectal Cancer Screening Paul Berg MD What is clinical integration? AMA Definition The means to facilitate the coordination of patient care across conditions, providers, settings, and time in order
Colorectal Cancer Screening Paul Berg MD What is clinical integration? AMA Definition The means to facilitate the coordination of patient care across conditions, providers, settings, and time in order
Cologuard Screening for Colorectal Cancer
 Pending Policies - Medicine Cologuard Screening for Colorectal Cancer Print Number: MED208.056 Effective Date: 08-15-2016 Coverage: I.Cologuard stool DNA testing may be considered medically necessary for
Pending Policies - Medicine Cologuard Screening for Colorectal Cancer Print Number: MED208.056 Effective Date: 08-15-2016 Coverage: I.Cologuard stool DNA testing may be considered medically necessary for
Colon Cancer Screening & Surveillance. Amit Patel, MD PGY-4 GI Fellow
 Colon Cancer Screening & Surveillance Amit Patel, MD PGY-4 GI Fellow Epidemiology CRC incidence and mortality rates vary markedly around the world. Globally, CRC is the third most commonly diagnosed cancer
Colon Cancer Screening & Surveillance Amit Patel, MD PGY-4 GI Fellow Epidemiology CRC incidence and mortality rates vary markedly around the world. Globally, CRC is the third most commonly diagnosed cancer
Colorectal Cancer Screening: A Clinical Update
 11:05 11:45am Colorectal Cancer Screening: A Clinical Update SPEAKER Kevin A. Ghassemi, MD Presenter Disclosure Information The following relationships exist related to this presentation: Kevin A. Ghassemi,
11:05 11:45am Colorectal Cancer Screening: A Clinical Update SPEAKER Kevin A. Ghassemi, MD Presenter Disclosure Information The following relationships exist related to this presentation: Kevin A. Ghassemi,
The choice of methods for Colorectal Cancer Screening; The Dutch experience
 The choice of methods for Colorectal Cancer Screening; The Dutch experience Monique van Leerdam, Gastroenterologist, NKI-AVL, Amsterdam The Netherlands Colorectal cancer CRC 2 nd cause of cancer related
The choice of methods for Colorectal Cancer Screening; The Dutch experience Monique van Leerdam, Gastroenterologist, NKI-AVL, Amsterdam The Netherlands Colorectal cancer CRC 2 nd cause of cancer related
The New Grade A: USPSTF Updated Colorectal Cancer Screening Guidelines, What does it all mean?
 The New Grade A: USPSTF Updated Colorectal Cancer Screening Guidelines, What does it all mean? Robert A. Smith, PhD Cancer Control, Department of Prevention and Early Detection American Cancer Society
The New Grade A: USPSTF Updated Colorectal Cancer Screening Guidelines, What does it all mean? Robert A. Smith, PhD Cancer Control, Department of Prevention and Early Detection American Cancer Society
R. J. L. F. Loffeld, 1 P. E. P. Dekkers, 2 and M. Flens Introduction
 ISRN Gastroenterology Volume 213, Article ID 87138, 5 pages http://dx.doi.org/1.1155/213/87138 Research Article The Incidence of Colorectal Cancer Is Decreasing in the Older Age Cohorts in the Zaanstreek
ISRN Gastroenterology Volume 213, Article ID 87138, 5 pages http://dx.doi.org/1.1155/213/87138 Research Article The Incidence of Colorectal Cancer Is Decreasing in the Older Age Cohorts in the Zaanstreek
Discussion. Points: 20. Objectives:
 EXERCISE 15: FECAL OCCULT BLOOD Points: 20 Objectives: 1. State the purpose for performing the occult blood test. 2. Define occult blood. 3. Describe the diet and state the forbidden foods/medications
EXERCISE 15: FECAL OCCULT BLOOD Points: 20 Objectives: 1. State the purpose for performing the occult blood test. 2. Define occult blood. 3. Describe the diet and state the forbidden foods/medications
A: PARTICIPANT INFORMATION
 A: PARTICIPANT INFMATION 1. What is your age today? Years of age 2. What is the date of your birth? Month: Day: Most of the questions we will be asking you in this follow-up questionnaire are about the
A: PARTICIPANT INFMATION 1. What is your age today? Years of age 2. What is the date of your birth? Month: Day: Most of the questions we will be asking you in this follow-up questionnaire are about the
Quality ID #343: Screening Colonoscopy Adenoma Detection Rate National Quality Strategy Domain: Effective Clinical Care
 Quality ID #343: Screening Colonoscopy Adenoma Detection Rate National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE TYPE: Outcome DESCRIPTION:
Quality ID #343: Screening Colonoscopy Adenoma Detection Rate National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE TYPE: Outcome DESCRIPTION:
THE NEW ZEALAND MEDICAL JOURNAL
 THE NEW ZEALAND MEDICAL JOURNAL Vol 120 No 1258 ISSN 1175 8716 A survey of colonoscopy capacity in New Zealand s public hospitals Andrew Yeoman, Susan Parry Abstract Aims Population screening for colorectal
THE NEW ZEALAND MEDICAL JOURNAL Vol 120 No 1258 ISSN 1175 8716 A survey of colonoscopy capacity in New Zealand s public hospitals Andrew Yeoman, Susan Parry Abstract Aims Population screening for colorectal
The most common methods currently used to investigate the colon (alone or in combination) include:
 CCO No.9 Oct 2002 Before CCO decides to undertake a health technology assessment, a pre-assessment of the literature is performed. Pre-assessments are based on a limited literature search; they are not
CCO No.9 Oct 2002 Before CCO decides to undertake a health technology assessment, a pre-assessment of the literature is performed. Pre-assessments are based on a limited literature search; they are not
BACKGROUND. Fecal immunochemical tests (FIT) are an advanced fecal occult. METHODS. Individuals sampled consecutive stools, at home, with both FIT and
 2152 Comparison of a Brush-Sampling Fecal Immunochemical Test for Hemoglobin With a Sensitive Guaiac-Based Fecal Occult Blood Test in Detection of Colorectal Neoplasia Alicia Smith 1,2 Graeme P. Young,
2152 Comparison of a Brush-Sampling Fecal Immunochemical Test for Hemoglobin With a Sensitive Guaiac-Based Fecal Occult Blood Test in Detection of Colorectal Neoplasia Alicia Smith 1,2 Graeme P. Young,
UPPER GASTROINTESTINAL AND COLONIC LESIONS IN PATIENTS WITH POSITIVE FECAL OCCULT-BLOOD TESTS
 UPPER GASTROINTESTINAL AND COLONIC LESIONS IN WITH POSITIVE FECAL OCCULT-BLOOD TESTS RELATIVE FREQUENCY OF UPPER GASTROINTESTINAL AND COLONIC LESIONS IN WITH POSITIVE FECAL OCCULT-BLOOD TESTS DON C. ROCKEY,
UPPER GASTROINTESTINAL AND COLONIC LESIONS IN WITH POSITIVE FECAL OCCULT-BLOOD TESTS RELATIVE FREQUENCY OF UPPER GASTROINTESTINAL AND COLONIC LESIONS IN WITH POSITIVE FECAL OCCULT-BLOOD TESTS DON C. ROCKEY,
Feasibility Of SigIlloidoscopic Screening For Bowel Cancer In A PriInary Care Setting
 Feasibility Of SigIlloidoscopic Screening For Bowel Cancer In A PriInary Care Setting David L. Hahn, M.D. Abstract: Sigmoidoscopic screening for bowel cancer is controversial because of its debatable efficacy,
Feasibility Of SigIlloidoscopic Screening For Bowel Cancer In A PriInary Care Setting David L. Hahn, M.D. Abstract: Sigmoidoscopic screening for bowel cancer is controversial because of its debatable efficacy,
Wellness Along the Cancer Journey: Healthy Habits and Cancer Screening Revised October 2015 Chapter 7: Cancer Screening and Early Detection of Cancer
 Wellness Along the Cancer Journey: Healthy Habits and Cancer Screening Revised October 2015 Chapter 7: Cancer Screening and Early Detection of Cancer Healthy Habits and Cancer Screening Rev 10.20.15 Page
Wellness Along the Cancer Journey: Healthy Habits and Cancer Screening Revised October 2015 Chapter 7: Cancer Screening and Early Detection of Cancer Healthy Habits and Cancer Screening Rev 10.20.15 Page
Colorectal cancer screening in asymptomatic populations
 590 Colorectal cancer screening in asymptomatic populations Gut 1995; 36: 590-598 Gastroenterology Research Unit, Leicester General Hospital, Leicester A R Hart A C B Wicks J F Mayberry Correspondence
590 Colorectal cancer screening in asymptomatic populations Gut 1995; 36: 590-598 Gastroenterology Research Unit, Leicester General Hospital, Leicester A R Hart A C B Wicks J F Mayberry Correspondence
Guidelines for the Early Detection of Cancer
 Guidelines for the Early Detection of Cancer The American Cancer Society recommends these cancer screening guidelines for most adults. Screening tests are used to find cancer before a person has any symptoms.
Guidelines for the Early Detection of Cancer The American Cancer Society recommends these cancer screening guidelines for most adults. Screening tests are used to find cancer before a person has any symptoms.
Colon Screening in 2014 Offering Patients a Choice. Clark A Harrison MD The Nevada Colon Cancer Partnership
 Colon Screening in 2014 Offering Patients a Choice Clark A Harrison MD The Nevada Colon Cancer Partnership Objectives 1. Understand the incidence and mortality rates for CRC in the US. 2. Understand risk
Colon Screening in 2014 Offering Patients a Choice Clark A Harrison MD The Nevada Colon Cancer Partnership Objectives 1. Understand the incidence and mortality rates for CRC in the US. 2. Understand risk
CENTERS FOR DISEASE CONTROL AND PREVENTION CENTERS FOR DISEASE CONTROL AND PREVENTION. Incidence Male. Incidence Female.
 A Call to Action: Prevention and Early Detection of Colorectal Cancer (CRC) 5 Key Messages Screening reduces mortality from CRC All persons aged 50 years and older should begin regular screening High-risk
A Call to Action: Prevention and Early Detection of Colorectal Cancer (CRC) 5 Key Messages Screening reduces mortality from CRC All persons aged 50 years and older should begin regular screening High-risk
C olorectal cancer (CRC) is one of the leading causes of
 1329 COLON Testing for faecal calprotectin (PhiCal) in the Norwegian Colorectal Cancer Prevention trial on flexible sigmoidoscopy screening: comparison with an immunochemical test for occult blood (FlexSure
1329 COLON Testing for faecal calprotectin (PhiCal) in the Norwegian Colorectal Cancer Prevention trial on flexible sigmoidoscopy screening: comparison with an immunochemical test for occult blood (FlexSure
Screening & Surveillance Guidelines
 Chapter 2 Screening & Surveillance Guidelines I. Eligibility Coloradans ages 50 and older (average risk) or under 50 at elevated risk for colon cancer (personal or family history) that meet the following
Chapter 2 Screening & Surveillance Guidelines I. Eligibility Coloradans ages 50 and older (average risk) or under 50 at elevated risk for colon cancer (personal or family history) that meet the following
The Cost-Effectiveness of Colorectal Cancer Screening in Average-Risk Adults. April 1995 OTA-BP-H-146
 The Cost-Effectiveness of Colorectal Cancer Screening in Average-Risk Adults April 1995 OTA-BP-H-146 ii Recommended Citation: U.S. Congress, Office of Technology Assessment, Cost-Effectiveness of Colorectal
The Cost-Effectiveness of Colorectal Cancer Screening in Average-Risk Adults April 1995 OTA-BP-H-146 ii Recommended Citation: U.S. Congress, Office of Technology Assessment, Cost-Effectiveness of Colorectal
UNDERSTANDING X-RAYS: ABDOMINAL IMAGING THE ABDOMEN
 UNDERSTANDING X-RAYS: ABDOMINAL IMAGING THE ABDOMEN Radiology Enterprises radiologyenterprises@gmail.com www.radiologyenterprises.com STOMACH AND SMALL BOWEL STOMACH AND SMALL BOWEL Swallowed air is a
UNDERSTANDING X-RAYS: ABDOMINAL IMAGING THE ABDOMEN Radiology Enterprises radiologyenterprises@gmail.com www.radiologyenterprises.com STOMACH AND SMALL BOWEL STOMACH AND SMALL BOWEL Swallowed air is a
Grand Rounds. Des Moines University. May 5, Durado Brooks, MD, MPH Director, Cancer Control Intervention American Cancer Society
 Grand Rounds Des Moines University May 5, 2016 Durado Brooks, MD, MPH Director, Cancer Control Intervention American Cancer Society Case Summary Mrs. J is a 56 y o w female complaining of always tired;
Grand Rounds Des Moines University May 5, 2016 Durado Brooks, MD, MPH Director, Cancer Control Intervention American Cancer Society Case Summary Mrs. J is a 56 y o w female complaining of always tired;
Patologia sistematica V Gastroenterologia Prof. Stefano Fiorucci. Colon polyps. Colorectal cancer
 Patologia sistematica V Gastroenterologia Prof. Stefano Fiorucci Colon polyps Colorectal cancer Harrison s Principles of Internal Medicine 18 Ed. 2012 Colorectal cancer 70% Colorectal cancer CRC and colon
Patologia sistematica V Gastroenterologia Prof. Stefano Fiorucci Colon polyps Colorectal cancer Harrison s Principles of Internal Medicine 18 Ed. 2012 Colorectal cancer 70% Colorectal cancer CRC and colon
Colorectal Cancer Screening
 Scan for mobile link. Colorectal Cancer Screening What is colorectal cancer screening? Screening examinations are tests performed to identify disease in individuals who lack any signs or symptoms. The
Scan for mobile link. Colorectal Cancer Screening What is colorectal cancer screening? Screening examinations are tests performed to identify disease in individuals who lack any signs or symptoms. The
Local Coverage Determination for Colorectal Cancer Screening (L29796)
 Page 1 of 15 Home Medicare Medicaid CHIP About CMS Regulations & Guidance Research, Statistics, Data & Systems Outreach & E People with Medicare & Medicaid Questions Careers Newsroom Contact CMS Acronyms
Page 1 of 15 Home Medicare Medicaid CHIP About CMS Regulations & Guidance Research, Statistics, Data & Systems Outreach & E People with Medicare & Medicaid Questions Careers Newsroom Contact CMS Acronyms
Cite this article as: BMJ, doi: /bmj c (published 5 July 2004)
 Cite this article as: BMJ, doi:10.1136/bmj.38153.491887.7c (published 5 July 2004) Results of the first round of a demonstration pilot of screening for colorectal cancer in the United Kingdom UK Colorectal
Cite this article as: BMJ, doi:10.1136/bmj.38153.491887.7c (published 5 July 2004) Results of the first round of a demonstration pilot of screening for colorectal cancer in the United Kingdom UK Colorectal
The New England Journal of Medicine
 The New England Journal of Medicine Copyright 2001 by the Massachusetts Medical Society VOLUME 345 A UGUST 23, 2001 NUMBER 8 ONE-TIME SCREENING FOR COLORECTAL CANCER WITH COMBINED FECAL OCCULT-BLOOD TESTING
The New England Journal of Medicine Copyright 2001 by the Massachusetts Medical Society VOLUME 345 A UGUST 23, 2001 NUMBER 8 ONE-TIME SCREENING FOR COLORECTAL CANCER WITH COMBINED FECAL OCCULT-BLOOD TESTING
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Virtual Colonoscopy / CT Colonography Page 1 of 19 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Virtual Colonoscopy / CT Colonography Professional Institutional
Virtual Colonoscopy / CT Colonography Page 1 of 19 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Virtual Colonoscopy / CT Colonography Professional Institutional
Cancer Screening Programmes BOWEL CANCER SCREENING. The Facts
 Cancer Screening Programmes BOWEL CANCER SCREENING The Facts What is the aim of this leaflet? This leaflet gives you information about bowel cancer, and the benefits and risks of bowel cancer screening.
Cancer Screening Programmes BOWEL CANCER SCREENING The Facts What is the aim of this leaflet? This leaflet gives you information about bowel cancer, and the benefits and risks of bowel cancer screening.
Financial Disclosers
 Slide 1 Colorectal Cancer Screening Jason Hemming, MD NESGNA November 15, 2014 Slide 2 Bio Slide 3 Financial Disclosers I have no actual or potential conflict of interest relation to this presentation
Slide 1 Colorectal Cancer Screening Jason Hemming, MD NESGNA November 15, 2014 Slide 2 Bio Slide 3 Financial Disclosers I have no actual or potential conflict of interest relation to this presentation
Objectives. Definitions. Colorectal Cancer Screening 5/8/2018. Payam Afshar, MS, MD Kaiser Permanente, San Diego. Colorectal cancer background
 Colorectal Cancer Screening Payam Afshar, MS, MD Kaiser Permanente, San Diego Objectives Colorectal cancer background Colorectal cancer screening populations Colorectal cancer screening modalities Colonoscopy
Colorectal Cancer Screening Payam Afshar, MS, MD Kaiser Permanente, San Diego Objectives Colorectal cancer background Colorectal cancer screening populations Colorectal cancer screening modalities Colonoscopy
Colon Cancer Screening. A Provider Opinion Survey
 Colon Cancer Screening A Provider Opinion Survey 1. Background Information What is colon cancer? Who needs to be screened? Colorectal Cancer» Presence of abnormal cells in the colon or rectum that divide
Colon Cancer Screening A Provider Opinion Survey 1. Background Information What is colon cancer? Who needs to be screened? Colorectal Cancer» Presence of abnormal cells in the colon or rectum that divide
Colon Cancer , The Patient Education Institute, Inc. oc Last reviewed: 05/17/2017 1
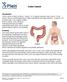 Colon Cancer Introduction Colon cancer is fairly common. About 1 in 15 people develop colon cancer. Colon cancer can be a life threatening condition that affects the large intestine. However, if it is
Colon Cancer Introduction Colon cancer is fairly common. About 1 in 15 people develop colon cancer. Colon cancer can be a life threatening condition that affects the large intestine. However, if it is
Bowel Cancer Information Leaflet THE DIGESTIVE SYSTEM
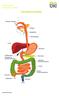 THE DIGESTIVE SYSTEM This factsheet is about bowel cancer Throughout our lives, the lining of the bowel constantly renews itself. This lining contains many millions of tiny cells, which grow, serve their
THE DIGESTIVE SYSTEM This factsheet is about bowel cancer Throughout our lives, the lining of the bowel constantly renews itself. This lining contains many millions of tiny cells, which grow, serve their
1101 First Colonial Road, Suite 300, Virginia Beach, VA Phone (757) Fax (757)
 1101 First Colonial Road, Suite 300, Virginia Beach, VA 23454 www.vbgastro.com Phone (757) 481-4817 Fax (757) 481-7138 1150 Glen Mitchell Drive, Suite 208 Virginia Beach, VA 23456 www.vbgastro.com Phone
1101 First Colonial Road, Suite 300, Virginia Beach, VA 23454 www.vbgastro.com Phone (757) 481-4817 Fax (757) 481-7138 1150 Glen Mitchell Drive, Suite 208 Virginia Beach, VA 23456 www.vbgastro.com Phone
Guidelines for Breast, Cervical and Colorectal Cancer Screening
 Guidelines for Breast, Cervical and Colorectal Cancer Screening Your recommendation counts. Talk to your patients about screening for cancer. CancerCare Manitoba provides organized, population-based screening
Guidelines for Breast, Cervical and Colorectal Cancer Screening Your recommendation counts. Talk to your patients about screening for cancer. CancerCare Manitoba provides organized, population-based screening
Razvan I. Arsenescu, MD Assistant Professor of Medicine Division of Digestive Diseases EARLY DETECTION OF COLORECTAL CANCER
 Razvan I. Arsenescu, MD Assistant Professor of Medicine Division of Digestive Diseases EARLY DETECTION OF COLORECTAL CANCER Epidemiology of CRC Colorectal cancer (CRC) is a common and lethal disease Environmental
Razvan I. Arsenescu, MD Assistant Professor of Medicine Division of Digestive Diseases EARLY DETECTION OF COLORECTAL CANCER Epidemiology of CRC Colorectal cancer (CRC) is a common and lethal disease Environmental
2015 Gastroenterology Survival Guide
 2015 Gastroenterology Survival Guide Chapter 11: ICD-9 Coding Your first line of ICD-9 coding is to attach signs or symptoms to your claim, but even after your gastroenterologist performs a diagnostic
2015 Gastroenterology Survival Guide Chapter 11: ICD-9 Coding Your first line of ICD-9 coding is to attach signs or symptoms to your claim, but even after your gastroenterologist performs a diagnostic
Alison Douglass Gillian Lieberman, MD. November. Colon Cancer. Alison Douglass, Harvard Medical School Year III Gillian Lieberman, MD
 November Colon Cancer Alison Douglass, Harvard Medical School Year III Our Patient Mr. K. is a 67 year old man with no prior medical problems other than hemorrhoids which have caused occasional rectal
November Colon Cancer Alison Douglass, Harvard Medical School Year III Our Patient Mr. K. is a 67 year old man with no prior medical problems other than hemorrhoids which have caused occasional rectal
Guidelines for Colonoscopy Surveillance After Screening and Polypectomy: A Consensus Update by the US Multi-Society Task Force on Colorectal Cancer
 Guidelines for Colonoscopy Surveillance After Screening and Polypectomy: A Consensus Update by the US Multi-Society Task Force on Colorectal Cancer David A. Lieberman, 1 Douglas K. Rex, 2 Sidney J. Winawer,
Guidelines for Colonoscopy Surveillance After Screening and Polypectomy: A Consensus Update by the US Multi-Society Task Force on Colorectal Cancer David A. Lieberman, 1 Douglas K. Rex, 2 Sidney J. Winawer,
EARLY DETECTION OF COLORECTAL CANCER. Epidemiology of CRC
 Razvan I. Arsenescu, MD Assistant Professor of Medicine Division of Digestive Diseases EARLY DETECTION OF COLORECTAL CANCER Epidemiology of CRC Colorectal cancer (CRC) is a common and lethal disease Environmental
Razvan I. Arsenescu, MD Assistant Professor of Medicine Division of Digestive Diseases EARLY DETECTION OF COLORECTAL CANCER Epidemiology of CRC Colorectal cancer (CRC) is a common and lethal disease Environmental
Health Professional Information
 Bowel Screening Wales Health Professional Information 2011 www.bowelscreeningwales.org.uk Version 2.0 Acknowledgements Bowel Screening Wales would like to express their sincere thanks to the English and
Bowel Screening Wales Health Professional Information 2011 www.bowelscreeningwales.org.uk Version 2.0 Acknowledgements Bowel Screening Wales would like to express their sincere thanks to the English and
CurrentStatusof FecalOccuR BloodTestingin Screening for ColorectalCancer
 CurrentStatusof FecalOccuR BloodTestingin Screening for ColorectalCancer Sidney J. Winawer, M.D. Martin Fleisher, Ph.D. Margaret Baldwin, M.P.A. Paul Sherlock, M.D. Introduction Colorectal cancer is one
CurrentStatusof FecalOccuR BloodTestingin Screening for ColorectalCancer Sidney J. Winawer, M.D. Martin Fleisher, Ph.D. Margaret Baldwin, M.P.A. Paul Sherlock, M.D. Introduction Colorectal cancer is one
PATIENT BROCHURE. 441 Charmany Dr 1 Madison WI, RX Only
 PATIENT BROCHURE 441 Charmany Dr 1 Madison WI, 53719 844-870- 8870 Cologuard colorectal cancer screening test is a registered trademark of Exact Sciences Corporation. Indications for Use Cologuard is intended
PATIENT BROCHURE 441 Charmany Dr 1 Madison WI, 53719 844-870- 8870 Cologuard colorectal cancer screening test is a registered trademark of Exact Sciences Corporation. Indications for Use Cologuard is intended
COLORECTAL SCREENING PROGRAMME: IMPACT ON THE HOSPITAL S PATHOLOGY SERVICES SINCE ITS INTRODUCTION.
 The West London Medical Journal 2009 Vol No 1 pp 23-31 COLORECTAL SCREENING PROGRAMME: IMPACT ON THE HOSPITAL S PATHOLOGY SERVICES SINCE ITS INTRODUCTION. Competing interests: None declared ABSTRACT Sarah
The West London Medical Journal 2009 Vol No 1 pp 23-31 COLORECTAL SCREENING PROGRAMME: IMPACT ON THE HOSPITAL S PATHOLOGY SERVICES SINCE ITS INTRODUCTION. Competing interests: None declared ABSTRACT Sarah
A Trip Through the GI Tract: Common GI Diseases and Complaints. Jennifer Curtis, MD
 A Trip Through the GI Tract: Common GI Diseases and Complaints Jennifer Curtis, MD Colon Cancer How does it develop? Most cancers arise from polyps Over time these can turn into cancer Combination of genetic
A Trip Through the GI Tract: Common GI Diseases and Complaints Jennifer Curtis, MD Colon Cancer How does it develop? Most cancers arise from polyps Over time these can turn into cancer Combination of genetic
Colorectal Cancer. Mark Chapman. MA MS FRCS EBSQ(coloproct) 21 st March 2018 Consultant Coloproctologist
 Colorectal Cancer Mark Chapman MA MS FRCS EBSQ(coloproct) 21 st March 2018 Consultant Coloproctologist Overview Epidemiology of colorectal cancer Adenoma carcinoma sequence Tumour diagnosis & staging Treatment
Colorectal Cancer Mark Chapman MA MS FRCS EBSQ(coloproct) 21 st March 2018 Consultant Coloproctologist Overview Epidemiology of colorectal cancer Adenoma carcinoma sequence Tumour diagnosis & staging Treatment
DR.RUPNATHJI( DR.RUPAK NATH )
 8. Screening for Colorectal Cancer Burden of Suffering RECOMMENDATION Screening for colorectal cancer is recommended for all persons aged 50 and older with annual fecal occult blood testing (FOBT), or
8. Screening for Colorectal Cancer Burden of Suffering RECOMMENDATION Screening for colorectal cancer is recommended for all persons aged 50 and older with annual fecal occult blood testing (FOBT), or
