SMC briefing note. The following medicines were accepted for use: The following medicine has not been recommended for use: About SMC.
|
|
|
- Annabella Foster
- 5 years ago
- Views:
Transcription
1 Monthly briefings are produced in order to help members of the media and other interested people understand the work and advice of the Scottish Medicines Consortium. The detailed advice for each medicine that we have assessed in full can be found at Number 64 March 2013 SMC briefing note Scottish Medicines Consortium advice to NHSScotland The following medicines were accepted for use: infliximab (Remicade ) ingenol mebutate gel (Picato ) rivaroxaban (Xarelto ) sugammadex (Bridion ) The following medicine has not been recommended for use: bevacizumab (Avastin ) About SMC The purpose of the Scottish Medicines Consortium (SMC) is to accept for use those newly licensed medicines that clearly represent good value for money to NHSScotland. SMC analyses information supplied by the medicine's submitting company on the health benefits of the medicine and justification of its price. Because the NHS has limited resources, SMC works to make sure that those medicines which represent good value for money are accepted for routine use as quickly as possible so that they can benefit patients. The Consortium is made up of lead clinicians, pharmacists and health economists together with representatives of health boards, the pharmaceutical industry, the public and the Scottish Government. SMC is part of Healthcare Improvement Scotland. Contact details If you are interested in the work of SMC you can visit our website at: or contact us at: Scottish Medicines Consortium Delta House (8th floor) 50 West Nile Street Glasgow G1 2NP smcsecretariat@nhshealthquality.org Media contact Members of the media should contact Stephen Ferguson on
2 SMC looks at how well medicines work in relation to how much they cost this is to make sure that they provide value for money for the NHS by providing extra benefits over current treatments at an additional cost that is reasonable. Medicines which represent good value for money are accepted for routine use to benefit patients. The following medicines were accepted for use in NHSScotland: infliximab (Remicade ) What is infliximab used for? Infliximab is an anti-inflammatory medicine that can be used in the treatment of several conditions. In this submission it has been proposed as a treatment for ulcerative colitis in patients aged between 6 and 17 years with severely active ulcerative colitis, when they have not responded to or cannot take other medicines or treatments. Ulcerative colitis is a form of inflammatory bowel disease affecting the large intestine (an organ that absorbs water from material passing from the small intestine). The condition causes the large intestine to become inflamed, and ulcers (large sores) develop on the lining of the intestine which can bleed and produce pus. Symptoms of active ulcerative colitis include bloody diarrhoea, stomach pain, increased frequency of bowel movements and weight loss. It is a long-term condition, with flareups of symptoms and then periods of remission (symptom-free periods). SMC briefing note Infliximab is licensed for use in both induction and maintenance treatment of severely active ulcerative colitis. However, the submitting company requested that SMC considers infliximab when positioned for use as a rescue therapy in acutely ill patients as an alternative to ciclosporin in patients who do not respond to treatment with steroids. Induction treatment involves the use of three doses only. Infliximab is an immunomodulator (a substance that modifies the immune system) and is one of a group of drugs called monoclonal antibodies. It is used when standard treatments do not control symptoms and acts by blocking TNF-alpha, one of the substances that cause the inflammation. Infliximab is an infusion given by a drip which is repeated 2 and 6 weeks after the first dose, then every 8 weeks. SMC accepted infliximab for the treatment of severely active ulcerative colitis in children and adolescents aged 6 to 17 years who have had an inadequate response to conventional therapy including corticosteroids and 6-mercaptopurine or azathioprine, or when there are reasons why the conventional therapy may not be used. It is restricted to use as an alternative to one of the commonly used medicines, ciclosporin, in patients with acute, severe paediatric ulcerative colitis (rescue therapy) who do not respond to treatment with steroids. One small study in 60 children showed that 73% of patients responded to treatment with infliximab at week eight and 40% achieved clinical remission. Some weaknesses in the clinical evidence presented lead to uncertainty about the extent of treatment benefits. On balance, however, infliximab was considered to offer value for money as an acute treatment. SMC accepted infliximab for restricted use because it provides the first licensed treatment for this group of patients and as a medical rescue therapy it may be a valuable alternative to surgery in some patients.
3 SMC briefing note ingenol mebutate gel (Picato ) What is ingenol mebutate used for? Ingenol mebutate is proposed as a treatment for actinic keratoses, which are small crusty, scaly or crumbly patches of skin which commonly occur in people that have had a lot of sun exposure. The lesions may be red, light brown or skin-coloured, and dry or rough to touch. Ingenol mebutate is a gel used to treat actinic keratoses on the face and scalp. It is not yet fully understood how the medicine works. It is thought to have a toxic effect on the skin lesions as well as promoting an inflammatory response. Together, these actions lead to the death of the cells affected by actinic keratosis. It is applied to the affected area once daily. The duration of treatment is limited to 2 or 3 consecutive days depending on the dose. SMC accepted ingenol mebutate for cutaneous treatment of non-hyperkeratotic, non-hypertrophic actinic keratosis in adults. Four studies showed that a higher proportion of patients treated with ingenol mebutate achieved complete clearance of actinic keratosis compared with a dummy gel containing no active treatment. An economic analysis compared ingenol mebutate with a range of other treatments. The length of treatment required with ingenol mebutate is much shorter than for other treatments which is beneficial to patients and may improve compliance. SMC accepted ingenol mebutate for use because the balance of costs and benefits meant that it was considered to offer value for money.
4 SMC briefing note rivaroxaban (Xarelto ) What is rivaroxaban used for? Rivaroxaban can be used to prevent and treat a range of conditions associated with abnormal blood clotting including deep vein thrombosis (DVT), which is a blood clot that forms in a vein in the legs. Blood flow can be restricted around the clot and may cause pain and swelling. There is a risk that all or part of the clot breaks off and is transported through the venous system to become lodged in the lungs where it forms a clot known as a pulmonary embolism (PE), which can be fatal. Rivaroxaban can also be used to treat and prevent recurrence of PE. SMC has previously accepted rivaroxaban for the treatment of venous thromboembolism (VTE) in hip and knee surgery; the prevention of stroke and systemic embolism in non-valvular atrial fibrillation in adults with one or more risk factors; and the treatment of DVT and prevention of recurrent DVT and pulmonary embolism (PE). This submission relates to the use of rivaroxaban to treat PE and to prevent recurrent DVT and PE in adults. Rivaroxaban is a blood-thinning medicine (anticoagulant) used to prevent harmful blood clots. It works by blocking Factor Xa, which is an important component of blood clotting. In this indication it is given as a 15 mg tablet twice daily for the first 3 weeks, then as 20 mg once daily for the duration of therapy. SMC accepted rivaroxaban for treatment of PE, and prevention of recurrent DVT and PE in adults. Two large studies showed that rivaroxaban was as effective as a regimen for the treatment of PE and the prevention of recurrence of DVT or PE. An economic analysis compared rivaroxaban with other anticoagulants. Although there were some limitations with the analysis, it suggested that rivaroxaban could be as effective as other anticoagulants at a reduced cost. SMC accepted rivaroxaban for use because the balance of costs and benefits meant that it was considered to offer value for money.
5 SMC briefing note sugammadex (Bridion ) What is sugammadex used for? During surgery, patients are often given neuromuscular blocking drugs (NMDs), or muscle relaxants, to relax their muscles, including the muscles that help the patient to breathe. This makes it easier for the surgeon to do the operation. This effect usually requires to be reversed at the end of surgery. Sugammadex is a medicine which is proposed for use to speed up the recovery from two muscle relaxants, rocuronium and vecuronium. In this submission it has been proposed for use in the routine reversal of profound neuromuscular blockade. SMC previously accepted sugammadex for restricted use for immediate reversal of neuromuscular block by rocuronium in adults. Sugammadex binds to the muscle relaxant, forming a complex that inactivates the muscle relaxants and stops them having an effect. As a result, the effect of blockade of the muscles due to rocuronium and vecuronium is reversed, and the muscles begin to work normally again. It is given by injection and should only be administered by, or under the supervision of, an anaesthetist. SMC accepted sugammadex for restricted use in the routine reversal of neuromuscular blockade induced by rocuronium or vecuronium in high-risk patients (eg those with morbid obesity, respiratory problems, significant heart disease, or following major abdominal/chest surgery) or where prompt reversal of neuromuscular block is required. Three studies have shown that sugammadex reverses the shallow and deep neuromuscular blocking effects of rocuronium and vecuronium more quickly than another medicine commonly used for this purpose. An economic analysis compared sugammadex with another medicine commonly used to reverse neuromuscular block. The company proposed that sugammadex could lead to cost savings because the more rapid action of sugammadex would lead to a quicker recovery time for the patient and decrease time in surgery. SMC clinical experts were uncertain about this assumption as other factors can influence the time that patients spend in surgery, but acknowledged that it could be useful in high-risk cases. SMC accepted sugammadex for restricted use because the balance of costs and benefits meant that it was considered to offer value for money.
6 SMC looks at how well medicines work in relation to how much they cost. Sometimes, when SMC considers the information submitted for a medicine, the committee is not convinced by the health benefit claims made by the company. On other occasions, the health benefits do not justify the cost of the medicine, or the company does not provide appropriate evidence to give SMC assurance that the medicine offers value for money. In any of these circumstances, SMC is unable to recommend that medicine for routine use in NHSScotland. SMC has not recommended the following medicine for use in NHSScotland: bevacizumab (Avastin ) What is bevacizumab used for? Bevacizumab is used to treat a range of different types of cancer. In this submission it has been proposed as a treatment, when given in combination with carboplatin and gemcitabine, for women with a first recurrence of platinum-sensitive epithelian ovarian, fallopian tube or primary peritoneal cancer. These women should not have received prior therapy with bevacizumab or other VEGF inhibitors or VEGF receptor targeted agents. Ovarian cancer is the fifth most common cause of death from cancer in women. Cancer of the ovaries (female reproductive organs) commonly starts in cells of the surface lining the ovaries called the epithelium and this type is known as epithelial ovarian cancer. Cancer can also affect other parts of the female reproductive system such as the fallopian tubes (tubes that link the ovaries to the womb). Cancer occurring in the lining of the abdomen (peritoneum) is known as primary peritoneal cancer and this rare cancer behaves in a similar way to epithelial ovarian cancer. SMC briefing note Cancer chemotherapy is given to try and kill cancer cells or stop them spreading further. Bevacizumab is given in combination with other cancer chemotherapy medicines. This medicine helps block the growth of new blood vessels that supply the cancer cells with nutrients. As a result, the cancer cells cannot develop their own blood supply and are starved of oxygen and nutrients, helping to slow down the growth of tumours. Bevacizumab is given once every 3 weeks as an infusion (via a drip). SMC did not recommend bevacizumab, in combination with carboplatin and gemcitabine, for treatment of adult patients with first recurrence of platinum-sensitive epithelian ovarian, fallopian tube or primary peritoneal cancer who have not received prior therapy with bevacizumab or other vascular endothelial growth factor (VEGF) inhibitors or VEGF receptor-targeted agents. The indication for recurrent platinum-sensitive epithelian ovarian, fallopian tube or primary peritoneal cancer, under consideration in this submission, is an extension to the marketing authorisation for bevacizumab. In a study, patients with recurrent ovarian cancer receiving bevacizumab plus carboplatin and gemcitabine survived without cancer progression for 4 months longer compared with those receiving only carboplatin and gemcitabine. However, there is uncertainty around the difference that bevacizumab makes to overall survival. An economic analysis compared carboplatin and gemcitabine with the same combination plus bevacizumab. The main limitation of the analysis was that the cost in relation to the health benefit was significantly above the threshold normally accepted by SMC. SMC did not recommend bevacizumab for use because the balance of costs and benefits meant that it was not considered to offer value for money. For medicines that have not been recommended by SMC, all NHS boards have procedures in place to consider individual requests when a doctor feels the medicine would be right for a particular patient. SMC has told the submitting company why the medicine was not recommended and would be pleased to receive any resubmission. For further information and to view the complete advice for the medicines listed above, visit our website at:
SMC briefing note. The following medicines were accepted for use: The following medicine has not been recommended for use: About SMC.
 Monthly briefings are produced in order to help members of the media and other interested people understand the work and advice of the Scottish Medicines Consortium. The detailed advice for each medicine
Monthly briefings are produced in order to help members of the media and other interested people understand the work and advice of the Scottish Medicines Consortium. The detailed advice for each medicine
SMC briefing note. The following medicines were accepted for use: Contact details. About SMC. Media contact. Number 98 January 2016
 Monthly briefings are produced in order to help members of the media and other interested people understand the work and advice of the Scottish Medicines Consortium. The detailed advice for each medicine
Monthly briefings are produced in order to help members of the media and other interested people understand the work and advice of the Scottish Medicines Consortium. The detailed advice for each medicine
A Guide to the Scottish Medicines Consortium
 A Guide to the Scottish Medicines Consortium Providing advice about newly licensed medicines www.scottishmedicines.org About the Scottish Medicines Consortium The Scottish Medicines Consortium (SMC) provides
A Guide to the Scottish Medicines Consortium Providing advice about newly licensed medicines www.scottishmedicines.org About the Scottish Medicines Consortium The Scottish Medicines Consortium (SMC) provides
SMC briefing note. About SMC. cetuximab (Erbitux ) Scottish Medicines Consortium advice to NHSScotland. Number 27 February 2010
 Scottish Medicines Consortium advice to NHSScotland Monthly briefings are produced in order to help members of the media and other interested groups understand the work and advice of the Scottish Medicines
Scottish Medicines Consortium advice to NHSScotland Monthly briefings are produced in order to help members of the media and other interested groups understand the work and advice of the Scottish Medicines
Supplemental New Drug Application for Rivaroxaban to reduce the risk of stent thrombosis in patients with Acute Coronary Syndrome also resubmitted
 Investor News Not intended for U.S. and UK Media Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com Complete Response Submitted to U.S. FDA on Bayer s Rivaroxaban for the Reduction
Investor News Not intended for U.S. and UK Media Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com Complete Response Submitted to U.S. FDA on Bayer s Rivaroxaban for the Reduction
To prevent blood clots after hip or knee replacement surgery This booklet contains information for those who have been prescribed ELIQUIS (apixaban)
 To prevent blood clots after hip or knee replacement surgery This booklet contains information for those who have been prescribed ELIQUIS (apixaban) after hip or knee replacement surgery Always read the
To prevent blood clots after hip or knee replacement surgery This booklet contains information for those who have been prescribed ELIQUIS (apixaban) after hip or knee replacement surgery Always read the
Deep Vein Thrombosis
 Deep Vein Thrombosis from NHS (UK) guidelines Introduction Deep vein thrombosis (DVT) is a blood clot in one of the deep veins in the body. Blood clots that develop in a vein are also known as venous thrombosis.
Deep Vein Thrombosis from NHS (UK) guidelines Introduction Deep vein thrombosis (DVT) is a blood clot in one of the deep veins in the body. Blood clots that develop in a vein are also known as venous thrombosis.
Abdominal surgery for Crohn's disease. Brought to you in association with EIDO Healthcare and endorsed by the Royal College of Surgeons England.
 Abdominal surgery for Crohn's disease Brought to you in association with EIDO Healthcare and endorsed by the Royal College of Surgeons England. Discovery has made every effort to ensure that we obtained
Abdominal surgery for Crohn's disease Brought to you in association with EIDO Healthcare and endorsed by the Royal College of Surgeons England. Discovery has made every effort to ensure that we obtained
Bayer AG Investor Relations Leverkusen Germany Investor News. Not intended for U.S. and UK Media
 Investor News Not intended for U.S. and UK Media Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com Bayer Initiates Xarelto (Rivaroxaban) Study in Patients with Non- Valvular Atrial
Investor News Not intended for U.S. and UK Media Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com Bayer Initiates Xarelto (Rivaroxaban) Study in Patients with Non- Valvular Atrial
SAFE study A-fib ED Anticoagulation Package
 SAFE study A-fib ED Anticoagulation Package In this package you will find documents to help you make decision making easy for the Anticoagulation of New AFib patients being discharged from the ED: 1. Anticoagulation
SAFE study A-fib ED Anticoagulation Package In this package you will find documents to help you make decision making easy for the Anticoagulation of New AFib patients being discharged from the ED: 1. Anticoagulation
Preventing Blood Clots in Adult Patients
 Manchester Royal Eye Hospital Surgical Services Information for Patients Preventing Blood Clots in Adult Patients This leaflet will give you information on how to reduce the risk of developing blood clots
Manchester Royal Eye Hospital Surgical Services Information for Patients Preventing Blood Clots in Adult Patients This leaflet will give you information on how to reduce the risk of developing blood clots
Reducing the risk of venous thrombo-embolism (VTE) in hospital and after discharge
 Reducing the risk of venous thrombo-embolism (VTE) in hospital and after discharge What is a venous thromboembolism (VTE)? This is a medical term that describes a blood clot that develops in a deep vein
Reducing the risk of venous thrombo-embolism (VTE) in hospital and after discharge What is a venous thromboembolism (VTE)? This is a medical term that describes a blood clot that develops in a deep vein
Deep vein thrombosis (DVT) and pulmonary embolism (PE) advice for ophthalmic surgery patients
 Deep vein thrombosis (DVT) and pulmonary embolism (PE) advice for ophthalmic surgery patients What is a deep vein thrombosis (DVT)? A DVT is a blood clot that forms within a vein deep in the leg but can
Deep vein thrombosis (DVT) and pulmonary embolism (PE) advice for ophthalmic surgery patients What is a deep vein thrombosis (DVT)? A DVT is a blood clot that forms within a vein deep in the leg but can
Date of preparation: September 2014 L.GB d
 Date of preparation: September 2014 L.GB.12.2013.4666d This medicine is subject to additional monitoring. This will allow quick identification of new safety information. If you get any side effects, talk
Date of preparation: September 2014 L.GB.12.2013.4666d This medicine is subject to additional monitoring. This will allow quick identification of new safety information. If you get any side effects, talk
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Oral Anticoagulants Page 1 of 7 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Oral Anticoagulant - Bevyxxa (betrixaban), Eliquis (apixaban), Pradaxa (dabigatran),
Oral Anticoagulants Page 1 of 7 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Oral Anticoagulant - Bevyxxa (betrixaban), Eliquis (apixaban), Pradaxa (dabigatran),
Rivaroxaban 10 mg Once Daily from Bayer Submitted to U.S. FDA as Additional Dose Option to Reduce the Risk of Recurrent Venous Thromboembolism
 Investor News Not intended for U.S. and UK Media Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com Rivaroxaban 10 mg Once Daily from Bayer Submitted to U.S. FDA as Additional
Investor News Not intended for U.S. and UK Media Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com Rivaroxaban 10 mg Once Daily from Bayer Submitted to U.S. FDA as Additional
Scottish Medicines Consortium
 Scottish Medicines Consortium infliximab 100mg powder for intravenous infusion (Remicade ) No. (364/07) Schering-Plough UK Ltd 6 April 2007 The Scottish Medicines Consortium (SMC) has completed its assessment
Scottish Medicines Consortium infliximab 100mg powder for intravenous infusion (Remicade ) No. (364/07) Schering-Plough UK Ltd 6 April 2007 The Scottish Medicines Consortium (SMC) has completed its assessment
Cancer and the Risk of Blood Clots
 Patient & Family Guide 2018 Cancer and the Risk of Blood Clots www.nscancercare.ca Table of Contents Terms Used in this Booklet 1 What are Blood Clots? 2 When are Cancer Patients at the Highest Risk for
Patient & Family Guide 2018 Cancer and the Risk of Blood Clots www.nscancercare.ca Table of Contents Terms Used in this Booklet 1 What are Blood Clots? 2 When are Cancer Patients at the Highest Risk for
Drug Class Review Newer Oral Anticoagulant Drugs
 Drug Class Review Newer Oral Anticoagulant Drugs Final Original Report May 2016 The purpose of reports is to make available information regarding the comparative clinical effectiveness and harms of different
Drug Class Review Newer Oral Anticoagulant Drugs Final Original Report May 2016 The purpose of reports is to make available information regarding the comparative clinical effectiveness and harms of different
Bayer Pharma AG Berlin Germany Tel News Release. Not intended for U.S. and UK Media
 News Release Not intended for U.S. and UK Media Bayer Pharma AG 13342 Berlin Germany Tel. +49 30 468-1111 www.bayerpharma.com Landmark Phase III Study of Bayer s Xarelto (Rivaroxaban) Initiated for the
News Release Not intended for U.S. and UK Media Bayer Pharma AG 13342 Berlin Germany Tel. +49 30 468-1111 www.bayerpharma.com Landmark Phase III Study of Bayer s Xarelto (Rivaroxaban) Initiated for the
Rivaroxaban. medication do? Xarelto. is a blood. Medication. Emboli (PE).
 Medication Information (Ri va rox xa - ban) Other names for this medication: Xarelto What does this medication do? This medication is a blood thinner, also called an anticoagulant. It prevents a blood
Medication Information (Ri va rox xa - ban) Other names for this medication: Xarelto What does this medication do? This medication is a blood thinner, also called an anticoagulant. It prevents a blood
Landmark Phase III Study of Bayer s Xarelto (Rivaroxaban) Initiated for the Secondary Prevention of Myo
 Xarelto (Rivaroxaban) Landmark Phase III Study of Bayer s Xarelto (Rivaroxaban) Initiated for the Secondary Prevention of Myocardial Infarction and Death in Patients with Coronary or Peripheral Artery
Xarelto (Rivaroxaban) Landmark Phase III Study of Bayer s Xarelto (Rivaroxaban) Initiated for the Secondary Prevention of Myocardial Infarction and Death in Patients with Coronary or Peripheral Artery
Area Drug and Therapeutics Committee Prescribing Supplement No 3 November 2003
 Area Drug and Therapeutics Committee Prescribing Supplement No 3 In this issue Drugs currently being considered by the SMC advice due on 8 December 2003. SMC Notice of meeting: Patient power Making a Difference,
Area Drug and Therapeutics Committee Prescribing Supplement No 3 In this issue Drugs currently being considered by the SMC advice due on 8 December 2003. SMC Notice of meeting: Patient power Making a Difference,
Venous Thromboembolism (VTE)
 Venous Thromboembolism (VTE) Nursing A guide for patients and carers Contents Why do blood clots form in veins?... 1 How common is a deep vein thrombosis (DVT) or pulmonary embolus (PE)?... 2 How are DVTs/
Venous Thromboembolism (VTE) Nursing A guide for patients and carers Contents Why do blood clots form in veins?... 1 How common is a deep vein thrombosis (DVT) or pulmonary embolus (PE)?... 2 How are DVTs/
Cancer and blood clots
 n The Leeds Teaching Hospitals NHS Trust Cancer and blood clots What you should know whilst you are in source isolation Information for thyroid patients Please read this leaflet carefully. It will give
n The Leeds Teaching Hospitals NHS Trust Cancer and blood clots What you should know whilst you are in source isolation Information for thyroid patients Please read this leaflet carefully. It will give
Patient consent form for liposuction Part 2 of 3
 Patient consent form for liposuction Part 2 of 3 This is an informed consent document. It explains the risks of and alternatives to liposuction. It is important that you read this information carefully
Patient consent form for liposuction Part 2 of 3 This is an informed consent document. It explains the risks of and alternatives to liposuction. It is important that you read this information carefully
Bayer submits application for marketing approval of rivaroxaban for patients with coronary or peripheral artery disease to European Medicines Agency
 Investor News Not intended for U.S. and UK Media Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com Bayer submits application for marketing approval of rivaroxaban for patients
Investor News Not intended for U.S. and UK Media Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com Bayer submits application for marketing approval of rivaroxaban for patients
Genentech Statement on Counterfeit Drug Labeled as Avastin (bevacizumab) in the United States
 Genentech Statement on Counterfeit Drug Labeled as Avastin (bevacizumab) in the United States SOUTH SAN FRANCISCO, Calif. February 14, 2012 Roche and Genentech have been informed that a counterfeit product,
Genentech Statement on Counterfeit Drug Labeled as Avastin (bevacizumab) in the United States SOUTH SAN FRANCISCO, Calif. February 14, 2012 Roche and Genentech have been informed that a counterfeit product,
Is Avastin right for me? Make an informed decision about Avastin
 Is Avastin right for me? Make an informed decision about Avastin Contents Page What is this handbook about? 2 About Avastin 3 What can Avastin do for me? 5 What else do I need to know? 7 Funded and unfunded
Is Avastin right for me? Make an informed decision about Avastin Contents Page What is this handbook about? 2 About Avastin 3 What can Avastin do for me? 5 What else do I need to know? 7 Funded and unfunded
Edoxaban Switch Programme - Frequently Asked Questions
 Edoxaban Switch Programme - Frequently Asked Questions What should I tell patients? NHS Tayside is reviewing all patients currently receiving a Direct Oral Anticoagulant (DOAC) for stroke prevention in
Edoxaban Switch Programme - Frequently Asked Questions What should I tell patients? NHS Tayside is reviewing all patients currently receiving a Direct Oral Anticoagulant (DOAC) for stroke prevention in
Understanding our report and advice: Antimicrobial wound dressings (AWDs) for chronic wounds
 Understanding our report and advice: Antimicrobial wound dressings (AWDs) for chronic wounds Health technology assessment report 13 December 2015 Healthcare Improvement Scotland 2015 Published December
Understanding our report and advice: Antimicrobial wound dressings (AWDs) for chronic wounds Health technology assessment report 13 December 2015 Healthcare Improvement Scotland 2015 Published December
pat hways Key therapeutic topic Published: 26 February 2016 nice.org.uk/guidance/ktt16
 pat hways Anticoagulants, including non-vitamin K antagonist oral anticoagulants (NOACs) Key therapeutic topic Published: 26 February 2016 nice.org.uk/guidance/ktt16 Options for local implementation NICE
pat hways Anticoagulants, including non-vitamin K antagonist oral anticoagulants (NOACs) Key therapeutic topic Published: 26 February 2016 nice.org.uk/guidance/ktt16 Options for local implementation NICE
This information explains the advice about Crohn's disease that is set out in NICE guideline CG152.
 Information for the public Published: 1 October 2012 nice.org.uk About this information NICE guidelines provide advice on the care and support that should be offered to people who use health and care services.
Information for the public Published: 1 October 2012 nice.org.uk About this information NICE guidelines provide advice on the care and support that should be offered to people who use health and care services.
Hemiarthroplasty (half hip replacement)
 Hemiarthroplasty (half hip replacement) Trauma and Orthopaedics Patient Information Leaflet Introduction This leaflet is about an operation called a half hip replacement. It gives information about the
Hemiarthroplasty (half hip replacement) Trauma and Orthopaedics Patient Information Leaflet Introduction This leaflet is about an operation called a half hip replacement. It gives information about the
Pancreas transplants. What you need to know. Information for patients Sheffield Kidney Institute (Renal Unit)
 Pancreas transplants What you need to know Information for patients Sheffield Kidney Institute (Renal Unit) page 2 of 16 What is the pancreas and why is it transplanted? The pancreas is part of the digestive
Pancreas transplants What you need to know Information for patients Sheffield Kidney Institute (Renal Unit) page 2 of 16 What is the pancreas and why is it transplanted? The pancreas is part of the digestive
How to prevent blood clots whilst in hospital and after your return home
 How to prevent blood clots whilst in hospital and after your return home Patient information WHAT What IS is DEEP deep VEIN vein THROMBOSIS? thrombosis? Deep Vein Thrombosis DVT is a blood clot within
How to prevent blood clots whilst in hospital and after your return home Patient information WHAT What IS is DEEP deep VEIN vein THROMBOSIS? thrombosis? Deep Vein Thrombosis DVT is a blood clot within
Bayer s Rivaroxaban Demonstrated Superior Protection Against Recurrent Venous Thromboembolism Compared with Aspirin in EINSTEIN CHOICE Study
 News Release Not intended for U.S. and UK Media Bayer AG Communications and Public Affairs 51368 Leverkusen Germany Tel. +49 214 30-0 www.news.bayer.com New Late-Breaking Study Data Presented at ACC.17:
News Release Not intended for U.S. and UK Media Bayer AG Communications and Public Affairs 51368 Leverkusen Germany Tel. +49 214 30-0 www.news.bayer.com New Late-Breaking Study Data Presented at ACC.17:
Total ankle replacement. Brought to you in association with EIDO Healthcare and endorsed by the Royal College of Surgeons England.
 Total ankle replacement Brought to you in association with EIDO Healthcare and endorsed by the Royal College of Surgeons England. Discovery has made every effort to ensure that we obtained the information
Total ankle replacement Brought to you in association with EIDO Healthcare and endorsed by the Royal College of Surgeons England. Discovery has made every effort to ensure that we obtained the information
PIONEER AF-PCI Study with Bayer s Xarelto Accepted as Late- Breaking Clinical Trial Presentation at AHA 2016
 News Release Not intended for U.S. and UK Media Bayer AG Communications, Government Relations & Corporate Brand 51368 Leverkusen Germany Tel. +49 214 30-0 www.news.bayer.com American Heart Association
News Release Not intended for U.S. and UK Media Bayer AG Communications, Government Relations & Corporate Brand 51368 Leverkusen Germany Tel. +49 214 30-0 www.news.bayer.com American Heart Association
Diagnosed with DVT/PE blood clots? Your journey with XARELTO starts here.
 THERE S MORE TO KNOW Diagnosed with DVT/PE blood clots? Your journey with XARELTO starts here. Do not stop taking XARELTO without talking to your doctor. Stopping XARELTO increases your risk of having
THERE S MORE TO KNOW Diagnosed with DVT/PE blood clots? Your journey with XARELTO starts here. Do not stop taking XARELTO without talking to your doctor. Stopping XARELTO increases your risk of having
Rivaroxaban to prevent blood clots for patients who have a lower limb plaster cast. Information for patients Pharmacy
 Rivaroxaban to prevent blood clots for patients who have a lower limb plaster cast Information for patients Pharmacy Your doctor has prescribed a tablet called rivaroxaban. This leaflet tells you about
Rivaroxaban to prevent blood clots for patients who have a lower limb plaster cast Information for patients Pharmacy Your doctor has prescribed a tablet called rivaroxaban. This leaflet tells you about
UW MEDICINE PATIENT EDUCATION. Treating Blood Clots. What is a blood clot? DRAFT
 UW MEDICINE PATIENT EDUCATION Treating Blood Clots About deep vein thrombosis (DVT) and pulmonary embolism (PE) and how they are treated This handout explains blood clots, their symptoms, and how they
UW MEDICINE PATIENT EDUCATION Treating Blood Clots About deep vein thrombosis (DVT) and pulmonary embolism (PE) and how they are treated This handout explains blood clots, their symptoms, and how they
Drugs reviewed by the SMC in September 2012 ADTC UPDATES ON DRUGS REVIEWED BY THE SMC
 Area Drug and Therapeutics Committee Prescribing Supplement No 62 In this issue Drugs reviewed by the SMC in September 2012 ADTC UPDATES ON DRUGS REVIEWED BY THE SMC The following new drugs have been reviewed
Area Drug and Therapeutics Committee Prescribing Supplement No 62 In this issue Drugs reviewed by the SMC in September 2012 ADTC UPDATES ON DRUGS REVIEWED BY THE SMC The following new drugs have been reviewed
MEDICAL ASSISTANCE HANDBOOK PRIOR AUTHORIZATION OF PHARMACEUTICAL SERVICES. A. Prescriptions That Require Prior Authorization
 MEDICAL ASSISTANCE HBOOK PRI AUTHIZATION OF PHARMACEUTICAL SERVICES I. Requirements for Prior Authorization of Anticoagulants A. Prescriptions That Require Prior Authorization Prescriptions for Anticoagulants
MEDICAL ASSISTANCE HBOOK PRI AUTHIZATION OF PHARMACEUTICAL SERVICES I. Requirements for Prior Authorization of Anticoagulants A. Prescriptions That Require Prior Authorization Prescriptions for Anticoagulants
APPLICATION TO CONSIDER A CHANGE TO A SIGN GUIDELINE
 APPLICATION TO CONSIDER A CHANGE TO A SIGN GUIDELINE 1. Contact person(s) proposing change to the guideline Roberta James on behalf of the SIGN/SMC/SDTC working group 2. Title and number of SIGN guideline
APPLICATION TO CONSIDER A CHANGE TO A SIGN GUIDELINE 1. Contact person(s) proposing change to the guideline Roberta James on behalf of the SIGN/SMC/SDTC working group 2. Title and number of SIGN guideline
diclofenac, 75mg/2ml of solution for intravenous injection (Dyloject ) No. (446/08) Javelin Pharmaceuticals UK Ltd
 Scottish Medicines Consortium diclofenac, 75mg/2ml of solution for intravenous injection (Dyloject ) No. (446/08) Javelin Pharmaceuticals UK Ltd 11 February 2008 The Scottish Medicines Consortium has completed
Scottish Medicines Consortium diclofenac, 75mg/2ml of solution for intravenous injection (Dyloject ) No. (446/08) Javelin Pharmaceuticals UK Ltd 11 February 2008 The Scottish Medicines Consortium has completed
Area Drug and Therapeutics Committee Prescribing Supplement No 59 July 2012
 Area Drug and Therapeutics Committee Prescribing Supplement No 59 In this issue Drugs reviewed by the SMC in June 2012 ADTC UPDATES ON DRUGS REVIEWED BY THE SMC The following new drugs have been reviewed
Area Drug and Therapeutics Committee Prescribing Supplement No 59 In this issue Drugs reviewed by the SMC in June 2012 ADTC UPDATES ON DRUGS REVIEWED BY THE SMC The following new drugs have been reviewed
Appendectomy. Brought to you in association with EIDO Healthcare and endorsed by the Royal College of Surgeons England.
 Appendectomy Brought to you in association with EIDO Healthcare and endorsed by the Royal College of Surgeons England. Discovery has made every effort to ensure that we obtained the information in this
Appendectomy Brought to you in association with EIDO Healthcare and endorsed by the Royal College of Surgeons England. Discovery has made every effort to ensure that we obtained the information in this
Understand. your risk of. blood clot
 Going into hospital? Understand how to reduce your risk of developing a blood clot Printing of this leaflet has been supported by a donation from Bayer HealthCare. Bayer HealthCare has had no editorial
Going into hospital? Understand how to reduce your risk of developing a blood clot Printing of this leaflet has been supported by a donation from Bayer HealthCare. Bayer HealthCare has had no editorial
Patient Information for Consent
 Patient Information for Consent ER OS01 Total Hip Replacement Enhanced Recovery Expires end of February 2017 Issued June 2016 Local information If you need any more information please contact your BMI
Patient Information for Consent ER OS01 Total Hip Replacement Enhanced Recovery Expires end of February 2017 Issued June 2016 Local information If you need any more information please contact your BMI
Discover the facts about
 Avastin is approved to treat metastatic colorectal cancer (mcrc) for: First- or second-line treatment in combination with intravenous 5-fluorouracil based chemotherapy Second-line treatment when used with
Avastin is approved to treat metastatic colorectal cancer (mcrc) for: First- or second-line treatment in combination with intravenous 5-fluorouracil based chemotherapy Second-line treatment when used with
Rivaroxaban film coated tablets are available in 2 strengths for this indication: 15mg and 20mg.
 Primary Care Prescriber Information RIVAROXABAN (XARELTO ) Treatment of acute venous thromboembolism and prevention of recurrent venous thromboembolism INDICATION Rivaroxaban is a non-vitamin K antagonist
Primary Care Prescriber Information RIVAROXABAN (XARELTO ) Treatment of acute venous thromboembolism and prevention of recurrent venous thromboembolism INDICATION Rivaroxaban is a non-vitamin K antagonist
Technology appraisal guidance Published: 28 November 2018 nice.org.uk/guidance/ta547
 Tofacitinib for moderately to severelyerely active ulcerative colitis Technology appraisal guidance Published: 28 November 2018 nice.org.uk/guidance/ta547 NICE 2019. All rights reserved. Subject to Notice
Tofacitinib for moderately to severelyerely active ulcerative colitis Technology appraisal guidance Published: 28 November 2018 nice.org.uk/guidance/ta547 NICE 2019. All rights reserved. Subject to Notice
National Institute for Health and Clinical Excellence Health Technology Appraisal
 National Institute for Health and Clinical Excellence Health Technology Appraisal Rivaroxaban for the prevention of venous thromboembolism after elective orthopaedic surgery of the lower limbs Comment
National Institute for Health and Clinical Excellence Health Technology Appraisal Rivaroxaban for the prevention of venous thromboembolism after elective orthopaedic surgery of the lower limbs Comment
New Study Presented at American Heart Association (AHA) Scientific Sessions 2016:
 Investor News Not intended for U.S. and UK Media Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com New Study Presented at American Heart Association (AHA) Scientific Sessions
Investor News Not intended for U.S. and UK Media Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com New Study Presented at American Heart Association (AHA) Scientific Sessions
Unicompartmental knee replacement. Brought to you in association with EIDO Healthcare and endorsed by the Royal College of Surgeons England.
 Unicompartmental knee replacement Brought to you in association with EIDO Healthcare and endorsed by the Royal College of Surgeons England. Discovery has made every effort to ensure that we obtained the
Unicompartmental knee replacement Brought to you in association with EIDO Healthcare and endorsed by the Royal College of Surgeons England. Discovery has made every effort to ensure that we obtained the
Femoral shaft fracture surgery (femoral nailing)
 Femoral shaft fracture surgery (femoral nailing) Brought to you in association with EIDO Healthcare and endorsed by the Royal College of Surgeons England. Discovery has made every effort to ensure that
Femoral shaft fracture surgery (femoral nailing) Brought to you in association with EIDO Healthcare and endorsed by the Royal College of Surgeons England. Discovery has made every effort to ensure that
Patient Group Submission Form
 Patient Group Submission Form The Scottish Medicines Consortium (SMC) is committed to working in partnership with patient groups to capture patient and carer experiences, and use them to inform decision-making.
Patient Group Submission Form The Scottish Medicines Consortium (SMC) is committed to working in partnership with patient groups to capture patient and carer experiences, and use them to inform decision-making.
Updates in Medical Management of Pulmonary Embolism and Deep Vein Thrombosis. By: Justin Youtsey, Elliott Reiff, William Montgomery, Grant Finlan
 Updates in Medical Management of Pulmonary Embolism and Deep Vein Thrombosis By: Justin Youtsey, Elliott Reiff, William Montgomery, Grant Finlan Objectives Describe the prevalence of PE and DVT as it relates
Updates in Medical Management of Pulmonary Embolism and Deep Vein Thrombosis By: Justin Youtsey, Elliott Reiff, William Montgomery, Grant Finlan Objectives Describe the prevalence of PE and DVT as it relates
Cardiovascular & Metabolism James List, M.D., Ph.D. Global Therapeutic Area Head, Cardiovascular & Metabolism
 Cardiovascular & Metabolism James List, M.D., Ph.D. Global Therapeutic Area Head, Cardiovascular & Metabolism Cardiovascular & Metabolism 1 Our Vision for the Future To improve the lives of the millions
Cardiovascular & Metabolism James List, M.D., Ph.D. Global Therapeutic Area Head, Cardiovascular & Metabolism Cardiovascular & Metabolism 1 Our Vision for the Future To improve the lives of the millions
Results from RE-COVER RE-COVER II RE-MEDY RE-SONATE EXECUTIVE SUMMARY
 Assessment of the safety and efficacy of dabigatran etexilate (Pradaxa ) in the treatment of deep vein thrombosis (DVT) and pulmonary embolism (PE) and the prevention of recurrent DVT and PE Results from
Assessment of the safety and efficacy of dabigatran etexilate (Pradaxa ) in the treatment of deep vein thrombosis (DVT) and pulmonary embolism (PE) and the prevention of recurrent DVT and PE Results from
Bayer s rivaroxaban submitted to U.S. FDA for approval in patients with coronary and/or peripheral artery disease
 Investor News Not intended for U.S. and UK Media Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com Bayer s rivaroxaban submitted to U.S. FDA for approval in patients with coronary
Investor News Not intended for U.S. and UK Media Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com Bayer s rivaroxaban submitted to U.S. FDA for approval in patients with coronary
Femoro-popliteal bypass surgery. Brought to you in association with EIDO Healthcare and endorsed by the Royal College of Surgeons England.
 Femoro-popliteal bypass surgery Brought to you in association with EIDO Healthcare and endorsed by the Royal College of Surgeons England. Discovery has made every effort to ensure that we obtained the
Femoro-popliteal bypass surgery Brought to you in association with EIDO Healthcare and endorsed by the Royal College of Surgeons England. Discovery has made every effort to ensure that we obtained the
Evidence review for Surrey Prescribing Clinical Network SUMMARY
 East Surrey CCG, Guildford & Waverley CCG, North West Surrey CCG, Surrey Downs CCG, Surrey Heath CCG, Crawley CCG, Horsham & Mid-Sussex CCG Evidence review for Surrey Prescribing Clinical Network Medicine
East Surrey CCG, Guildford & Waverley CCG, North West Surrey CCG, Surrey Downs CCG, Surrey Heath CCG, Crawley CCG, Horsham & Mid-Sussex CCG Evidence review for Surrey Prescribing Clinical Network Medicine
Pa#ent Informa#on for Consent
 Pa#ent Informa#on for Consent ER_OS02 Total Knee Replacement Enhanced Recovery Expires end of November 2018 Local Informa#on For further informa0on locally you can contact the Pa0ent Advice & Liaison Service
Pa#ent Informa#on for Consent ER_OS02 Total Knee Replacement Enhanced Recovery Expires end of November 2018 Local Informa#on For further informa0on locally you can contact the Pa0ent Advice & Liaison Service
Confirmed blood clot
 n The Leeds Teaching Hospitals NHS Trust Confirmed blood clot (Deep vein thrombosis and pulmonary embolism) Information for patients Please read this leaflet carefully. It will give you information about
n The Leeds Teaching Hospitals NHS Trust Confirmed blood clot (Deep vein thrombosis and pulmonary embolism) Information for patients Please read this leaflet carefully. It will give you information about
HELPING TO TREAT DEEP VEIN THROMBOSIS AND PULMONARY EMBOLISM
 HELPING TO TREAT DEEP VEIN THROMBOSIS AND PULMONARY EMBOLISM AND PREVENT THEIR RECURRENCE PATIENT INFORMATION BOOKLET IF FOUND, PLEASE RETURN TO: If found, please return to: Name Address Phone number It
HELPING TO TREAT DEEP VEIN THROMBOSIS AND PULMONARY EMBOLISM AND PREVENT THEIR RECURRENCE PATIENT INFORMATION BOOKLET IF FOUND, PLEASE RETURN TO: If found, please return to: Name Address Phone number It
Consultation on the role of the Scottish Health Council: Strengthening people s voices in health and social care
 Consultation on the role of the Scottish Health Council: Strengthening people s voices in health and social care July 2017 Contents Page Introduction 3 The Scottish Health Council 5 Our Voice 6 Why change?
Consultation on the role of the Scottish Health Council: Strengthening people s voices in health and social care July 2017 Contents Page Introduction 3 The Scottish Health Council 5 Our Voice 6 Why change?
Bayer s Xarelto (Rivaroxaban) Meets Primary Efficacy Endpoint and Shows Significant Reduction in Mortality in Major ACS Study
 Investor News Not intended for U.S. and UK Media Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com Secondary Prevention of Acute Coronary Syndrome (ACS): Bayer s Xarelto (Rivaroxaban)
Investor News Not intended for U.S. and UK Media Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com Secondary Prevention of Acute Coronary Syndrome (ACS): Bayer s Xarelto (Rivaroxaban)
Bevacizumab (Avastin ) treatment for Neurofibromatosis Type 2 (NF2) Information for patients
 Bevacizumab (Avastin ) treatment for Neurofibromatosis Type 2 (NF2) Information for patients This booklet has been written to give you more information about bevacizumab (commonly known as Avastin). This
Bevacizumab (Avastin ) treatment for Neurofibromatosis Type 2 (NF2) Information for patients This booklet has been written to give you more information about bevacizumab (commonly known as Avastin). This
Venous Thromboembolism National Hospital Inpatient Quality Measures
 Venous Thromboembolism National Hospital Inpatient Quality Measures Presentation Overview Review venous thromboembolism as a new mandatory measure set Outline measures with exclusions and documentation
Venous Thromboembolism National Hospital Inpatient Quality Measures Presentation Overview Review venous thromboembolism as a new mandatory measure set Outline measures with exclusions and documentation
NEW/NOVEL ORAL ANTICOAGULANTS (NOACS): COMPARISON AND FREQUENTLY ASKED QUESTIONS
 NEW/NOVEL ORAL ANTICOAGULANTS (NOACS): COMPARISON AND FREQUENTLY ASKED QUESTIONS OBJECTIVES: To provide a comparison of the new/novel oral anticoagulants (NOACs) currently available in Canada. To address
NEW/NOVEL ORAL ANTICOAGULANTS (NOACS): COMPARISON AND FREQUENTLY ASKED QUESTIONS OBJECTIVES: To provide a comparison of the new/novel oral anticoagulants (NOACs) currently available in Canada. To address
The London Gastroenterology Partnership CROHN S DISEASE
 CROHN S DISEASE What is Crohn s disease? Crohn s disease is a condition, in which inflammation develops in parts of the gut leading to symptoms such as diarrhoea, abdominal pain and tiredness. The inflammation
CROHN S DISEASE What is Crohn s disease? Crohn s disease is a condition, in which inflammation develops in parts of the gut leading to symptoms such as diarrhoea, abdominal pain and tiredness. The inflammation
Preventing Venous Thromboembolism (VTE)
 INTERNET INFORMATION SOURCES North American Thrombosis Forum www.natfonline.org Clot Care www.clotcare.com MANITOUWADGE GENERAL HOSPITAL National Alliance for Thrombosis/Thrombophilia www.stoptheclot.org
INTERNET INFORMATION SOURCES North American Thrombosis Forum www.natfonline.org Clot Care www.clotcare.com MANITOUWADGE GENERAL HOSPITAL National Alliance for Thrombosis/Thrombophilia www.stoptheclot.org
Arthroscopy of the ankle. Brought to you in association with EIDO Healthcare and endorsed by the Royal College of Surgeons England.
 Arthroscopy of the ankle Brought to you in association with EIDO Healthcare and endorsed by the Royal College of Surgeons England. Discovery has made every effort to ensure that we obtained the information
Arthroscopy of the ankle Brought to you in association with EIDO Healthcare and endorsed by the Royal College of Surgeons England. Discovery has made every effort to ensure that we obtained the information
News Release. For UK Media
 News Release For UK Media Bayer plc 400 South Oak Way Reading RG2 6AD www.bayer.co.uk Bayer s Xarelto (rivaroxaban) in combination with single antiplatelet therapy receives positive CHMP opinion for treatment
News Release For UK Media Bayer plc 400 South Oak Way Reading RG2 6AD www.bayer.co.uk Bayer s Xarelto (rivaroxaban) in combination with single antiplatelet therapy receives positive CHMP opinion for treatment
Deep Vein Thrombosis and Pulmonary Embolism: Risks, Prevention & Treatment
 Deep Vein Thrombosis and Pulmonary Embolism: Risks, Prevention & Treatment Other formats If you need this information in another format such as audio tape or computer disk, Braille, large print, high contrast,
Deep Vein Thrombosis and Pulmonary Embolism: Risks, Prevention & Treatment Other formats If you need this information in another format such as audio tape or computer disk, Braille, large print, high contrast,
Diagnostic laparoscopy. Brought to you in association with EIDO Healthcare and endorsed by the Royal College of Surgeons England.
 Diagnostic laparoscopy Brought to you in association with EIDO Healthcare and endorsed by the Royal College of Surgeons England. Discovery has made every effort to ensure that we obtained the information
Diagnostic laparoscopy Brought to you in association with EIDO Healthcare and endorsed by the Royal College of Surgeons England. Discovery has made every effort to ensure that we obtained the information
The Johns Hopkins Hospital Patient Information. How Do I Prevent Blood Clots? Venous Thromboembolism (VTE) Deep Vein Thrombosis (DVT)
 Page 1 of 11 Venous Thromboembolism () What is a clot or Venous Thromboembolism ()? Blood clots are called Venous Thromboembolism (). There are 2 main types: is a clot in a deep vein, usually an arm or
Page 1 of 11 Venous Thromboembolism () What is a clot or Venous Thromboembolism ()? Blood clots are called Venous Thromboembolism (). There are 2 main types: is a clot in a deep vein, usually an arm or
Summary of the risk management plan (RMP) for Senshio (ospemifene)
 EMA/736568/2014 Summary of the risk management plan (RMP) for Senshio (ospemifene) This is a summary of the risk management plan (RMP) for Senshio, which details the measures to be taken in order to ensure
EMA/736568/2014 Summary of the risk management plan (RMP) for Senshio (ospemifene) This is a summary of the risk management plan (RMP) for Senshio, which details the measures to be taken in order to ensure
Deep Vein Thrombosis and Pulmonary Embolism: Patient Information
 Deep Vein Thrombosis and Pulmonary Embolism: Patient Information A Deep Vein Thrombosis (DVT) and a Pulmonary Embolism (PE) are both disorders of unwanted blood clotting. Unwanted blood clots can occur
Deep Vein Thrombosis and Pulmonary Embolism: Patient Information A Deep Vein Thrombosis (DVT) and a Pulmonary Embolism (PE) are both disorders of unwanted blood clotting. Unwanted blood clots can occur
Laparoscopic excision of a gastric gist. Brought to you in association with EIDO Healthcare and endorsed by the Royal College of Surgeons England.
 Laparoscopic excision of a gastric gist Brought to you in association with EIDO Healthcare and endorsed by the Royal College of Surgeons England. Discovery has made every effort to ensure that we obtained
Laparoscopic excision of a gastric gist Brought to you in association with EIDO Healthcare and endorsed by the Royal College of Surgeons England. Discovery has made every effort to ensure that we obtained
Preventing blood clots while you are in hospital and after you leave. Information for patients Pharmacy
 Preventing blood clots while you are in hospital and after you leave Information for patients Pharmacy Why does blood clot? When we cut ourselves, we bleed. To stop us from bleeding too much, chemicals
Preventing blood clots while you are in hospital and after you leave Information for patients Pharmacy Why does blood clot? When we cut ourselves, we bleed. To stop us from bleeding too much, chemicals
Inferior Vena Cava Filter for DVT
 Inferior Vena Cava Filter for DVT Deep Vein Thrombosis A deep vein thrombosis (DVT) is a blood clot that forms in a deep vein. This is a serious condition that occurs more often than you might think. If
Inferior Vena Cava Filter for DVT Deep Vein Thrombosis A deep vein thrombosis (DVT) is a blood clot that forms in a deep vein. This is a serious condition that occurs more often than you might think. If
Dynamic hip screw (sliding hip screw)
 Dynamic hip screw (sliding hip screw) Trauma and Orthopaedics Patient Information Leaflet Introduction This leaflet is about an operation called a dynamic hip screw, sometimes also known as a sliding hip
Dynamic hip screw (sliding hip screw) Trauma and Orthopaedics Patient Information Leaflet Introduction This leaflet is about an operation called a dynamic hip screw, sometimes also known as a sliding hip
Subcapital hip fracture surgery. Brought to you in association with EIDO Healthcare and endorsed by the Royal College of Surgeons England.
 Subcapital hip fracture surgery Brought to you in association with EIDO Healthcare and endorsed by the Royal College of Surgeons England. Discovery has made every effort to ensure that we obtained the
Subcapital hip fracture surgery Brought to you in association with EIDO Healthcare and endorsed by the Royal College of Surgeons England. Discovery has made every effort to ensure that we obtained the
Arthroscopy of the knee. Brought to you in association with EIDO Healthcare and endorsed by the Royal College of Surgeons England.
 Arthroscopy of the knee Brought to you in association with EIDO Healthcare and endorsed by the Royal College of Surgeons England. Discovery has made every effort to ensure that we obtained the information
Arthroscopy of the knee Brought to you in association with EIDO Healthcare and endorsed by the Royal College of Surgeons England. Discovery has made every effort to ensure that we obtained the information
Warfarin. Information for patients taking warfarin
 Warfarin Information for patients taking warfarin Read this booklet to learn: what warfarin is and why you are taking it what blood tests you need to use warfarin safely how other medicines and diet can
Warfarin Information for patients taking warfarin Read this booklet to learn: what warfarin is and why you are taking it what blood tests you need to use warfarin safely how other medicines and diet can
Toe and forefoot amputation. Brought to you in association with EIDO Healthcare and endorsed by the Royal College of Surgeons England.
 Toe and forefoot amputation Brought to you in association with EIDO Healthcare and endorsed by the Royal College of Surgeons England. Discovery has made every effort to ensure that we obtained the information
Toe and forefoot amputation Brought to you in association with EIDO Healthcare and endorsed by the Royal College of Surgeons England. Discovery has made every effort to ensure that we obtained the information
1. Comparative effectiveness of vedolizumab
 Cost-effectiveness of vedolizumab (Entyvio ) for the treatment of adult patients with moderately to severely active ulcerative colitis who have had an inadequate response with, lost response to, or were
Cost-effectiveness of vedolizumab (Entyvio ) for the treatment of adult patients with moderately to severely active ulcerative colitis who have had an inadequate response with, lost response to, or were
Internal fixation of a hip fracture. Brought to you in association with EIDO Healthcare and endorsed by the Royal College of Surgeons England.
 Internal fixation of a hip fracture Brought to you in association with EIDO Healthcare and endorsed by the Royal College of Surgeons England. Discovery has made every effort to ensure that we obtained
Internal fixation of a hip fracture Brought to you in association with EIDO Healthcare and endorsed by the Royal College of Surgeons England. Discovery has made every effort to ensure that we obtained
Putting NICE guidance into practice
 Putting NICE guidance into practice Resource impact report: Olaparib for maintenance treatment of relapsed, platinum-sensitive, BRCA mutation-positive ovarian, fallopian tube and peritoneal cancer after
Putting NICE guidance into practice Resource impact report: Olaparib for maintenance treatment of relapsed, platinum-sensitive, BRCA mutation-positive ovarian, fallopian tube and peritoneal cancer after
Hip hemi-arthroplasty
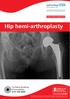 Hip hemi-arthroplasty Turnberg Building Orthopaedics 0161 206 4898 All Rights Reserved 2017. Document for issue as handout. Procedure The hip joint is a ball and socket joint. It is a very important joint
Hip hemi-arthroplasty Turnberg Building Orthopaedics 0161 206 4898 All Rights Reserved 2017. Document for issue as handout. Procedure The hip joint is a ball and socket joint. It is a very important joint
Treatment with Apixaban Eliquis
 Treatment with Apixaban Eliquis Anticoagulation Clinic 416-530-6000 ext 3477 This handout explains the medicine rivaroxaban, a drug that helps prevent blood clots. What is apixaban? Apixaban (brand name
Treatment with Apixaban Eliquis Anticoagulation Clinic 416-530-6000 ext 3477 This handout explains the medicine rivaroxaban, a drug that helps prevent blood clots. What is apixaban? Apixaban (brand name
Understanding your risk of blood clots from hospital to home and how Bevyxxa may help
 Understanding your risk of blood clots from hospital to home and how Bevyxxa may help People who take Bevyxxa and have medicine injected into their spinal and epidural area, or have a spinal puncture,
Understanding your risk of blood clots from hospital to home and how Bevyxxa may help People who take Bevyxxa and have medicine injected into their spinal and epidural area, or have a spinal puncture,
Inflectra for Crohn s disease
 Inflectra for Crohn s disease Some important information to get you started with your treatment This booklet is intended only for use by patients who have been prescribed Inflectra. Introduction When you
Inflectra for Crohn s disease Some important information to get you started with your treatment This booklet is intended only for use by patients who have been prescribed Inflectra. Introduction When you
Treatment with Rivaroxaban Xarelto
 Treatment with Rivaroxaban Xarelto Anticoagulation Clinic This handout explains the medicine rivaroxaban, a drug that helps prevent blood clots. What is rivaroxaban? Rivaroxaban (brand name Xarelto) is
Treatment with Rivaroxaban Xarelto Anticoagulation Clinic This handout explains the medicine rivaroxaban, a drug that helps prevent blood clots. What is rivaroxaban? Rivaroxaban (brand name Xarelto) is
Reducing the Risk of Deep Vein Thrombosis (DVT) whilst in hospital and following your discharge home
 Reducing the Risk of Deep Vein Thrombosis (DVT) whilst in hospital and following your discharge home 1 Part of: South Tyneside and Sunderland Healthcare Group Introduction This leaflet explains what Deep
Reducing the Risk of Deep Vein Thrombosis (DVT) whilst in hospital and following your discharge home 1 Part of: South Tyneside and Sunderland Healthcare Group Introduction This leaflet explains what Deep
Patient Information for Consent
 Patient Information for Consent OS01 Total Hip Replacement Expires end of February 2017 Issued June 2016 Local information If you need any more information please contact your BMI hospital on:...... Get
Patient Information for Consent OS01 Total Hip Replacement Expires end of February 2017 Issued June 2016 Local information If you need any more information please contact your BMI hospital on:...... Get
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE
 NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE Centre for Clinical Practice SCOPE Clinical guideline title: Ulcerative colitis: the management of ulcerative colitis Quality standard title: Ulcerative
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE Centre for Clinical Practice SCOPE Clinical guideline title: Ulcerative colitis: the management of ulcerative colitis Quality standard title: Ulcerative
