Realities of Diagnosing Helicobacter pylon Infection in Clinical Practice: A Case for Non-invasive Indirect Methodologies
|
|
|
- Ami Turner
- 5 years ago
- Views:
Transcription
1 YALE JOURNAL OF BIOLOGY AND MEDICINE 71 (1998), pp Copyright All rights reserved. Realities of Diagnosing Helicobacter pylon Infection in Clinical Practice: A Case for Non-invasive Indirect Methodologies David C. Metz, Emma E. Furth, Douglas 0. Faigel, Joyann A. Kroser, Abass Alavi, David M. Barrett and Kathleen Montone Gastroenterology Division, Department ofpathology and Laboratory Medicine, and Division of Nuclear Medicine, University ofpennsylvania Medical Center, Philadelphia, Pennsylvania Background: The current, arbitrarily defined gold standard for the diagnosis of H. pylori infection requires histologic examination of two specially stained antral biopsy specimens. However, routine histology is potentially limited in general clinical practice by both sampling and observer error. The current study was designed to examine the diagnostic performance of invasive and non-invasive H. pylori detection methods that would likely be available in general clinical practice. Methods: The diagnostic performance of rotating clinical pathology faculty using thiazine staining was compared with that of an expert gastrointestinal pathologist in 38 patients. In situ hybridization stains of adjacent biopsy cuts were also examined by the expert pathologist for further comparison. Receiver operator characteristic (ROC) analysis was performed to evaluate whether the diagnostic performance of the expert pathologist differed depending upon the histologic method employed. A similar analysis was made to evaluate the diagnostic performance of pathology trainees relative to the expert. In the absence of an established invasive gold standard, non-invasive testing methods (rapid serum antibodies, formal Elisa antibodies and carbon-14 urea breath testing) were evaluated in 74 patients by comparison with a gold standard defined using a combination of diagnostic tests. Results: Using either rapid urease testing of biopsy specimens or urea breath testing as the gold standard for comparison, the diagnostic performance of the rotating clinical pathology faculty was inferior to that of the expert gastrointestinal pathologist especially with regard to specificity (e.g., 69 percent for the former versus 88 percent, with the latter relative to rapid urease testing). Although interpretation of in situ hybridization staining by the expert appeared to have an even higher specificity, ROC analysis failed to show a difference. The mean ROC areas for thiazine and in situ hybridization staining for trainee pathologists relative to the expert were 0.88 and 0.94, respectively. In untreated patients, urea breath testing had a sensitivity and specificity of 100 percent as compared with thiazine staining with a sensitivity of 83 percent and a specificity of 97 percent. Post-therapy, breath testing had a sensitivity of 100 percent but a specificity of only 86 percent as compared with invasive testing with a sensitivity and specificity of 100 percent. Rapid serum antibody testing and formal Elisa antibody testing agreed in 93 percent of cases (Kappa 0.78) with the rapid test being correct in three of the four disagreements Conclusions: The current study illustrates a number of realities regarding H. pylori diagnosis. There is no diagnostic gold standard in general clinical practice. Accurate interpretation of specially stained slides is a learned activity with a tendency towards overdiagnosis early on. Urea breath testing is likely to be the ato whom all correspondence should be addressed: Dr. David C. Metz, Assistant Professor of Medicine, Gastroenterology Division - 3 Ravdin, University of Pennsylvania Medical Center, 3400 Spruce Street, Philadelphia, PA Tel.: (215) ; Fax: (215) ; Metzda@mail.med.upenn.edu. babbreviations: PCR, polymerase chain reaction; ROC, receiver operator characteristic; DPM, disintergrations per minute. 81
2 82 Metz et al.: H. pylori diagnosis diagnostic method of choice for untreated patients in general clinical practice although antibody testing is almost as accurate. Rapid antibody tests are at least as accurate as formal Elisa antibody tests. Urea breath testing is useful for confirming cure after therapy, but false-positive results may occur in some patients. INTRODUCTION The current arbitrarily defined gold standard for the diagnosis of Helicobacterpylori infection consists of histologic examination of two specially-stained antral biopsy specimens [1, 2]. Studies with the Giemsa, Warthin Starry or Genta stains have revealed an accuracy that may be as high as 98 percent in expert hands [2, 3]. However, histology may be less accurate in general clinical practice for a variety of reasons. Sampling error is commonly cited as a potential cause of inaccurate histological diagnosis so that certain authorities have recommended that additional specimens be taken from the lesser curve and/or the gastric body to increase the diagnostic yield [4]. Additional biopsy specimens may be especially important in patients taking antisecretory therapy. Histology is also potentially limited in clinical practice by inter- and intra-observer variability depending on the expertise of the examining pathologist [5, 6]. Moreover, none of the currently accepted special stains used to identify H. pylori infection are specific for the organism so that diagnosis is made upon morphologic characteristics alone [1, 2]. The most specific method for diagnosing H. pylori infection is culture of biopsy specimens, but this method is highly insensitive because of the fastidiousness of the organism [1, 2]. Consequently, routine culture cannot be considered an acceptable gold standard for general clinical practice. Other specific invasive methodologies such as polymerase chain reaction (PCR)b or immunohistochemistry are also limited in clinical practice because of the expertise required to perform these studies [7, 8]. A variety of highly accurate, non-invasive diagnostic tests for H. pylori infection have recently become available [1, 2]. These include rapid office-based antibody tests, formal Elisa antibody testing, and urea breath testing (both carbon 13 and carbon 14 tests) [1, 2, 6, 9-12]. In the absence of a true gold standard for diagnosis, these newer methodologies have been compared against a battery of other methods to demonstrate their accuracy [11, 12]. From a statistical point of view, this approach is limited by the inclusion of the test method under consideration in the arbitrarily defined gold standard itself, so that it can be argued that the method under consideration is actually being compared with itself. An alternative approach is to compare the test methodology with a gold standard that is defined by concordance of two or more other methodologies (for example, histology, rapid urease testing or breath testing) [9]. An obvious limitation of this approach is that patients with discordant gold standard component test results have to be excluded from analysis. Little is known about the accuracy of these newer, non-invasive methodologies in general clinical practice since they too may be limited by interpretation bias (for example, where to define the transition from positive to negative in terms of the magnitude of the test response). The aims of the current study were to determine the accuracy of invasive and noninvasive H. pylori detection methods in general clinical practice. We chose to examine methods that would likely be available in most diagnostic centers and that would require minimal expertise for accurate interpretation of results.
3 Metz et al.: H. pylori diagnosis 83 METHODS General All patients presenting to the Gastroenterology Division of the University of Pennsylvania for upper endoscopy were invited to participate in the current study. Patients who were pregnant, breast feeding, had used antibiotics or bismuth preparations within four weeks or proton pump inhibitors within one week, or had previously undergone gastric surgery were excluded. Patients who had received specific anti-h. pylori therapy in the past were included providing that this treatment had been completed at least four weeks prior to testing. Three antral biopsy specimens (within 3 cm of the pylorus) were taken in all patients at the time of their endoscopy. Two specimens were submitted for histological analysis, and one was used for rapid urease testing (CLO test, Delta West, Bentley, Australia). To determine the intra-observer reproducibility of histology for the diagnosis of H. pylori infection, the expert gastrointestinal pathologist read 16 randomly selected antral biopsy slides on three separate days over a four-week period in a different random order at each session. There was disagreement as to the presence or absence of H. pylori organisms in two of 16 cases. In each case, the slides were read as negative twice but positive once with an organism burden of one on a scale of 0 to three on both occasions. There was complete agreement amongst all three readings in the remaining 14 cases. The intra-class correlation coefficient was 0.82 with a P value for a difference between replicates of 0.62 [6]. All studies had the approval of the committee on studies involving humans and the radiation safety committee of the University of Pennsylvania and were only performed after the patients provided informed consent. Invasive testing In order to assess the diagnostic performance of standard histological methods for H. pylori diagnosis in general clinical practice, we compared the sensitivity and specificity of thiazine staining for H. pylori diagnosis (relative to rapid urease testing and urea breath testing) as assessed by rotating clinical pathology faculty with the results obtained by the expert gastrointestinal pathologist in 38 Bouin's-fixed paraffin-imbedded antral biopsies. To further evaluate the diagnostic performance of the expert gastrointestinal pathologist using standard histology, the latter's sensitivity and specificity with thiazine staining were compared with in situ hybridization staining of adjacent cuts of all 38 biopsy specimens [13]. In situ hybridization was performed using a 22 base biotin-labeled oligonucleotide probe complementary to a portion of the H. pylori 16 s ribosomal RNA at a final concentration of 1 pg/ml using manual capillary action technology on the Microprobe Staining System (Fisher Scientific, Boston, Massachussetts) with modifications as previously described. Receiver operator characteristic (ROC) analysis of thiazine staining and in situ hybridization for H. pylori detection relative to rapid urease and urea breath testing was determined for the experienced gastrointestinal pathologist for a more detailed comparison of standard histological methods with our in situ hybridization technique for H. pylori detection [14]. ROC analysis is independent of disease prevalence making it possible to determine sensitivity and specificity over all "cut-off' points. In the absence of a "true" gold standard for H. pylori diagnosis, we chose to keep the rapid urease and urea breath tests as invariant tests for these comparisons since it is generally easy to distinguish positive from negative tests with these methods. This in no way implies that either the rapid urease or urea breath test is the "true" gold standard for diagnosis. Instead, one test must be kept constant such that relative comparisons can be made. ROC analysis was also performed on five resident pathologists in training relative to the expert gastrointestinal pathologist in order to determine how much experience was required for accurate H.
4 84 Metz et al.: H. pylori diagnosis pylori detection. For each ROC analysis, the observers graded on a scale of 1 through 5 their surety of the presence of H. pylori organisms (one being definitely not present, five being definitely present). The area under the ROC curve was calculated for each analysis. This value is a measure of diagnostic performance with a value of 0.5 representing chance and 1.0 representing perfect test performance [14]. The grading of thiazine and in situ stains were done on separate occasions. Non-invasive testing The diagnostic performance of non-invasive testing methodologies was assessed first by defining the gold standard using a combination of results from all tests in 74 study subjects in a manner similar to those described by Cutler [11] and the Division of Anti-infective Drug Products Center for Drug Evaluation and Research of the US Food and Drug Administration (Dr. A. Hopkins, personal communication). These tests included evaluation of two antral biopsy specimens stained with thiazine for the presence of organisms, two antral biopsies stained with hematoxyllin and eosin for the presence of chronic inflammation on routine histology [12] and one antral biopsy specimen, which was used for rapid urease testing. Histological interpretation was performed by the same expert gastrointestinal pathologist who performed the invasive testing studies (see above). In addition, all patients underwent urea breath testing after ingestion of a capsule containing 1 pci (37 IEq) carbon 14 urea (Trimed specialties, Charlottesville, Virginia) with subsequent collection of breath samples at five-minute intervals for 30 minutes [6]. Radioactivity of exhaled breath was measured in a scintillation counter in auto-dpm mode with a calculated efficiency of greater than 98 percent and expressed as disintegrations per minute (DPM). A positive test with breath testing was defined as an increase over baseline of more than 100 DPMs. Antibody testing was performed on all patients using two methodologies, the Flexure HP rapid serum test (SmithKline Diagnostics, San Jose, California), which was performed in the office, and the HM CAP Elisa antibody test (Enteric Products, Inc., Stony Brook, New York), which was performed on serum that was mailed to a reference laboratory. For the former test, supernatant was removed after centrifugation at 3200 RPM for 20 minutes and placed on Flexure HP cards [15]. A positive test result was defined as the appearance of a pink colored test line in addition to a visible control line. The latter test was performed after additional serum samples were coded and transferred to SmithKline Diagnostics for quantitative Elisa assay using the HM CAP antigen [16]. Elisa values greater than 2.2 were considered positive for H. pylori infection. Using a combination of these testing methods, a pre-therapy true-positive diagnosis was defined as the presence of four or more positive tests while a true-negative required one or none positive. Patients with either two or three positive tests were considered nonevaluable and were excluded from analysis. For post-therapy diagnosis, the second antibody test was removed from the above scheme, and a positive diagnosis post-therapy required three or more of the four remaining tests to be positive. A negative diagnosis required one or none of the four tests to be positive, and patients with two positive tests were excluded from analysis. Statistical analysis were performed using Statistica 4.0 (Statsoft Inc., Tulsa, Oklahoma). Estimates of sensitivity and specificity were computed in the usual manner, and comparisons between the two serological tests were performed using percentage agreement and the chance adjusted agreement statistic, Kappa. RESULTS Rapid urease testing and urea breath testing detected H. pylori infection in 15/38 (39 percent) and 19/38 patients (50 percent), respectively. Using the rapid urease test as the
5 Metz et al.: H. pylori diagnosis 85 Table 1. Sensitivity and specificity of histology for H. pylori diagnosis (n = 38). Relative to rapid urease test Relative to urea breath test Sensitivity (%) Specificity (%) Sensitivity (%) Specificity (%) Routine pathology (Thiazine stain) GI pathologist (Thiazine stain) GI pathologist (In situ hybridization) gold standard, thiazine staining of antral biopsy specimens had a sensitivity of 87 percent and a specificity of 69 percent for H. pylori detection when evaluated by the rotating clinical pathology faculty (Table 1). When the same slides were examined by the expert gastrointestinal pathologist, the sensitivity of thiazine was unchanged but the specificity increased to 88 percent. The sensitivity and specificity of in situ hybridization in the expert pathologist's hands was 80 percent and 100 percent, respectively, as compared with the rapid urease test results (Table 1). Using urea breath testing as the gold standard, a similar trend occurred, although the sensitivity of histology was somewhat lower for all calculations because urea breath testing identified more patients with H. pylori gastritis than any other modality. In order to determine whether the relative diagnostic performance of thiazine and in situ hybridization staining for H. pylori diagnosis was truly different in expert gastrointestinal pathologists' hands, ROC analysis was performed. ROC values did not differ significantly between the two histological methods indicating equivalent test performance (Table 2). The relative diagnostic performance of thiazine staining and in situ hybridization for five pathologists in training, with the expert gastrointestinal pathologist serving as the gold standard, is shown in Table 3. The mean ROC areas for thiazine staining and in situ hybridization were 0.88 and 0.94, respectively. In addition, when asked which of the two diagnostic methods was easier to interpret, four of the five trainees felt the in situ hybridization stain to be superior, and the one trainee who felt that the thiazine stain was superior actually performed better relative to the expert gastrointestinal pathologist with the in situ stain (data not shown). Table 2. Relative diagnostic performance of thiazine and in situ hybridization staining for H. pyloni diagnosis by the expert gastrointestinal pathologist (n = 38). Relative to rapid urease test (ROCI values) Relative to urea breath test (ROCI values) Thiazine stain In situ hybridization ROC = receiver operator characteristic
6 86 Metz et al.: H. pylori diagnosis Table 3. Relative diagnostic performance of two histological methods for H. pyloni diagnosis by five pathologists in training (n = 38). Observer Thiazine stain relative to In situ hybridization relative to expert GI pathologist expert GI pathologist (ROC1 values) (ROCI values) Average l ROC = receiver operator characteristic. Table 4. Sensitivity and specificity of various testing methods H. pylori diagnosis (n = 74).1 Elisa Rapid serum Pre-therapy Thiazine Rapid urease Chronic Urea breath antibody antibody (n = 54) stain test gastritis test test test Sensitivity (%) Specificity (%) Post-therapy (n= 15) Sensitivity (%) N/A2 N/A2 Specificity (%) N/A2 N/A2 I The gold standard for diagnosis is defined according to the scheme outlined in Methods. 2 N/A, not applicable. Table 5. Analysis of discordant antibody tests. Rapid serum Rapid Urea Patient antibody Elisal urease Thiazine breath Correct number test value test stain test test 7006 Positive 1.6 Positive Positive Positive Rapid antibody 7026 Negative 2.5 Negative Negative Negative Rapid antibody 7029 Negative 2.5 Positive Positive Positive Elisa test 7051 Negative 2.5 Negative Negative Negative Rapid antibody I A value greater than 2.2 is considered positive.
7 Metz et al.: H. pylori diagnosis 87 Negative Positive 0 0f M.).. w Cl) X LLJ >v J * ELISA VALUE (Units) Figure 1. Correlation between office-based rapid serum antibody testing and laboratorybased formal Elisa antibody testing for H. pyloyi diagnosis in 58 patients not previously treated for H. pyloni infection. Elisa test results are shown along the x-axis (an Elisa value of > 2. 2 units corresponds with a positive result). Rapid serum antibody test results are shown along the y-axis. There was a 93 percent agreement between the two H. pylori antibody test methods (Kappa = 0.78). Of 74 patients evaluated with all non-invasive diagnostic tests, five were excluded because they had indeterminate gold standard results. Fifty-four of the remaining patients had not previously been treated for H. pylori infection, and 15 were post therapy. Twentythree of the 54 untreated patients were H. pylori positive by the gold standard, and one of the 15 post-therapy patients was still infected. The sensitivity and specificity of each diagnostic test in the diagnosis of H. pylori infection both pre- and post-therapy are shown in Table 4. In untreated patients, invasive thiazine staining (the currently accepted gold standard for diagnosis) had a sensitivity of 83 percent and a specificity of 97 percent. By comparison, the sensitivity and specificity of urea breath testing in this population was 100 percent. In evaluating H. pylori status post-therapy, the sensitivity and specificity of invasive testing with thiazine (for organisms) or hematoxyllin and eosin staining (for chronic inflammation), or rapid urease testing (for urease detection) was 100 percent. Urea breath testing had a sensitivity of 100 percent, but its specificity was only 86 percent. Figure 1 illustrates the agreement between the two serologic tests in patients not previously treated for H. pylori infection, including four additional patients with indeterminate gold standard results. Test results agreed in 54 of 58 patients (93 percent) with a Kappa statistic of Test results disagreed in four patients (7 percent), three with negative rapid antibody results and positive Elisa tests and one with a positive rapid antibody test and a negative Elisa test. The discordant antibody test results are shown in more detail in Table 5. In three of the four disagreements, the rapid antibody test was the correct test when compared with other diagnostic modalities including rapid urease testing, thiazine testing and urea breath testing.
8 88 Metz et al.: H. pylori diagnosis DISCUSSION The current study makes clear a number of realities regarding H. pylori diagnosis. First, there is no true diagnostic gold standard in general clinical practice. This is brought out by the fact that the correlation between standard thiazine staining for H. pylori diagnosis and other non-invasive diagnostic methods (urea breath testing or rapid urease testing) is closer when interpreted by an expert gastrointestinal pathologist as opposed to rotating pathology faculty in a large teaching hospital. The major shortcoming appears to be one of overdiagnosis since specificity is unacceptably low when slides are interpreted by rotating pathology faculty. In keeping with our previous report [6], the initial specificity of in situ hybridization relative to thiazine staining suggested that the tendency for over-diagnosis exists with expert pathologists as well. However, the current ROC analysis suggested that this was not the case. The ROC analyses of the pathologists in training suggest that the learning curve for diagnosing H. pylori infection both by thiazine stain and by in situ hybridization is short. ROC values for the trainees were all acceptable, but their diagnostic performance was more favorable with the in situ method (area under the ROC curve of 0.94) than with standard thiazine staining (0. 88). We interpret this data to suggest that a trained gastrointestinal pathologist accurately diagnoses H. pylori infection using standard special stains most of the time. In comparison, trainee pathologists are less accurate with standard stains, but their accuracy approaches that of an expert if specialized methods such as in situ hybridization are utilized. However, such methods are not routinely available and are, thus, clearly beyond the scope of routine clinical practice. The results of the current study do suggest that in situ hybridization may be useful for training pathologists in interpreting H. pylori slides. Thus, the second reality of the current study regarding H. pylori diagnosis is that accurate interpretation of thiazine-stained slides is a learned activity. The current study did not directly address the potential effect of sampling error on invasive diagnostic results. Other investigators have suggested that this is a real consideration affecting diagnostic yield [4]. In this regard, it is interesting to note that in the current study, urea breath testing (which samples a much larger area of the gastric mucosa than any of the biopsy-based tests) detected more potential infections than either rapid urease testing or histology (thiazine staining and in situ hybridization). Thus, sampling error may be a factor affecting test accuracy with diagnostic methods that depend upon small pinch biopsies of the gastric mucosa. Given the limitations of invasive testing in clinical practice, we chose to evaluate the diagnostic performance of non-invasive diagnostic methods in previously untreated and post therapy patients as possible clinical practice alternatives. The gold standard for comparison in these studies was devised using a combination of tests in order to limit the likelihood that the test under consideration appeared less accurate than it actually was because of a gold standard inaccuracy. The limitations of this approach have been described above. This methodology was chosen over a concordance method using invasive testing because we feel that invasive testing has limitations even in the best hands. For diagnosis in patients not previously treated for H. pylori infection, urea breath testing appears to be the most accurate (i.e., the gold standard for clinical practice). While the current study addresses only carbon 14 urea breath testing, it is likely that results with carbon 13 urea breath testing would be similar. Previous studies on the diagnostic accuracy of urea breath testing have shown it to be an excellent test [6, 9, 11]. For post-therapy H. pylori diagnosis, the current data would suggest that invasive testing is most accurate. However, in evaluating patients post-therapy in general clinical practice, it is most important that the test under consideration always correctly identify non-cured patients. Urea breath testing was positive in all the non-cured patients in the current study. Its apparent shortcoming, in
9 Metz et al.: H. pylori diagnosis 89 keeping with other investigators [11], was that it was occasionally positive in post-therapy patients who appeared to be cured by all other testing methods. This data could also be interpreted to suggest that urea breath testing is the most sensitive of all methods for determining failure to eradicate an infection. In order to determine which of these two possibilities is true, patients with apparent false-positive urea breath tests will need to be reevaluated on a second occasion some time later to see whether the breath test becomes negative (i.e., a false-positive initial breath test) or the other tests became positive (i.e., a true-positive initial breath test). Further studies will be required to address this issue. Thus the third reality of the current study regarding H. pylori diagnosis is that urea breath testing is likely to be the diagnostic method of choice in general clinical practice for previously untreated patients. For post-therapy confirmation of H. pylori eradication, urea breath testing is accurate, although there is a question regarding false-positive testing in some patients. In the absence of urea breath testing, the next best diagnostic modalities in untreated patients appear to be rapid antibody testing or rapid urease testing. Both these modalities have specificities of 100 percent and sensitivities of 91 percent, but the rapid antibody test is likely to be more useful in routine clinical practice because it is non-invasive and less costly. Rapid antibody testing is at least as accurate as formal Elisa testing [10, 17]. In the current study, rapid antibody testing had similar sensitivity (91 percent) but a better specificity (100 percent versus 94 percent). In three of the four cases in which there was discordance between rapid antibody testing and formal Elisa testing, the rapid test was the correct test. Thus, the fourth diagnostic reality of the current study is the fact that rapid antibody testing is at least as accurate as formal Elisa testing and almost as accurate as breath testing. There is no longer a need to send blood to a reference laboratory in order to test for H. pylori antibodies, and antibody testing should remain the screening method of choice for H. pylori diagnosis because of ease of performance and low cost. Whole blood antibody testing is now being developed for routine office use, and it is likely that whole blood antibody testing will ultimately replace serum antibody testing because of ease of use, providing that it can be shown to be as accurate [18]. REFERENCES 1. Marshall, B.J. Helicobacter pylori. Am. J. Gastroenterol. 89:SI 16-S 128, Brown, K.E. and Peura, D.A. Diagnosis of Helicobacter pylori infection. Gastroenterol. Clin. N. Am. 22: , Genta, R.M., Robason, G.R., and Graham, D.Y. Simultaneous visualization of Helicobacter pylori and gastric morphology: a new stain. Hum. Pathol.. 25: , Genta, R.M. and Graham, D.Y. Comparison of biopsy sites for the histopathologic diagnosis of Helicobacter pylori: a topographic study of H. pylori density and distribution. Gastrointest. Endosc. 40: , Genta, R.M., El-Zimaity, H.M.T., Al-Assi, M.T., Malaty, H.M., Kartunen, T.J., Graham, D.P., Huberman, R.M. and Graham, D.Y. Interobserver variation in the histologic evaluation of Helicobacter pylori gastritis. Gastroenterology 108:A100, (Abstract). 6. Faigel, D.O., Childs, M., Furth, E.E., Alavi, A., and Metz, D.C. New noninvasive tests for Helicobacterpylori gastritis: comparison with tissue-based gold standard. Dig. Dis. Sci. 41: , van Zwet, A.A., Thijs, J.C., Kooistra-Smid, A.M.D., Schirm, J., and Snijder, J.A.M. Sensitivity of culture compared to that of polymerase chain reaction for detection of Helicobacter pylori from antral biopsy samples. J. Clin. Microbiol. 31: , Vandenberg, F.M., Ziglmans, H., Langenberg, W., Ruows, E., and Schipper, M. Detection of Campylobacter pylori in stomach tissue by DNA in-situ hybridization. J. Clin. Pathol. 42: , Klein, P.D., Malaty, H.M., Martin, R.F., Graham, K.S., Genta, R.M., and Graham, D.Y. Noninvasive detection of Helicobacter pylori infection in clinical practice: the C- 13 urea breath test. Am. J. Gastroenterol. 91: , 1996.
10 90 Metz et al.: H. pylori diagnosis 10. Graham, D.Y, Evans, D.J., Jr., Peacock, J., Baker, J.T., and Schrier, W.H. Comparison of rapid serological tests (FlexSure HP and QuickVue) with conventional ELISA for detection of Helicobacter pylori infection. Am. J. Gastroenterol. 91: , Cutler, A.F., Havstad, S., Ma, C.K., Blaser, M.J., Perez-Perez, G.I., and Schubert, T.T. Accuracy of invasive and non-invasive tests to diagnose Helicobacter pylori infection. Gastroenterology 109: , Faigel, D.O., Furth, E.E., Childs, M., Goin, J., and Metz, D.C. Histological predictors of active Helicobacterpylori infection. Dig. Dis. Sci. 41: , Barrett, D.M., Faigel, D., Metz, D., Montone, K., and Furth, E.E. In situ hybridization for Helicobacter pylori in gastric mucosal biopsy specimens: Quantitative evaluation of test performance in comparison with the CLOtest and thiazine stain. Lab. Invest. 74:A303, Metz, C.E. Basic principles of ROC analysis. Semin. Nucl. Med. 8: , Anonymous. Product instructions for FlexSure HP test for serum IgG antibodies to Helicobacter pylori. SmithKline Diagnostics, Inc.; Anonymous. Product instructions for HM-CAP enzyme immunoassay for the detection of IgG antibodies to Helicobacter pylori in human serum. Enteric Products, Inc.; Kroser, J.A., Faigel, D.O., Childs, M., Furth, E.E., and Metz, D.C. Faster is better: comparison of rapid versus formal antibody testing for the diagnosis of Helicobacter pylori (Hp) gastritis. Gastroenterology 110: A163, (Abstract). 18. Harrison, J.R., Bevan, J., Furth, E.E., and Metz, D.C. Rapid whole blood antibody testing for untreated H. pylori infection: the diagnostic modality of choice. Gastroenterology. (Abstract, in press).
Type of intervention Diagnosis. Economic study type Cost-effectiveness analysis.
 Noninvasive tests as a substitute for histology in the diagnosis of Helicobacter pylori infection Hahn M, Fennerty M B, Corless C L, Magaret N, Lieberman D A, Faigel D O Record Status This is a critical
Noninvasive tests as a substitute for histology in the diagnosis of Helicobacter pylori infection Hahn M, Fennerty M B, Corless C L, Magaret N, Lieberman D A, Faigel D O Record Status This is a critical
Comparative study of invasive methods for diagnosis of Helicobacter pylori in humans
 ISSN: 2319-7706 Volume 2 Number 7 (2013) pp. 63-68 http://www.ijcmas.com Original Research Article Comparative study of invasive methods for diagnosis of Helicobacter pylori in humans V.Subbukesavaraja
ISSN: 2319-7706 Volume 2 Number 7 (2013) pp. 63-68 http://www.ijcmas.com Original Research Article Comparative study of invasive methods for diagnosis of Helicobacter pylori in humans V.Subbukesavaraja
594 Lewin, Weinstein, and Riddell s Gastrointestinal Pathology and Its Clinical Implications
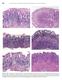 594 Lewin, Weinstein, and Riddell s Gastrointestinal Pathology and Its Clinical Implications Figure 13-20. Stages in the natural history of H. pylori. Biopsies from the antrum are on the left and the oxyntic
594 Lewin, Weinstein, and Riddell s Gastrointestinal Pathology and Its Clinical Implications Figure 13-20. Stages in the natural history of H. pylori. Biopsies from the antrum are on the left and the oxyntic
Urea Breath Test for Diagnosis of Helicobactor pylori. Original Policy Date 12:2013
 MP 2.04.04 Urea Breath Test for Diagnosis of Helicobactor pylori Medical Policy Section Medicine Issue 12:2013 Original Policy Date 12:2013 Last Review Status/Date 12:2013 Return to Medical Policy Index
MP 2.04.04 Urea Breath Test for Diagnosis of Helicobactor pylori Medical Policy Section Medicine Issue 12:2013 Original Policy Date 12:2013 Last Review Status/Date 12:2013 Return to Medical Policy Index
Breath Test Diagnosis of Helicobacter pylori in Peptic Ulcer Disease: A Noninvasive Primary Care Option
 ORIGINAL ARTICLES Breath Test Diagnosis of Helicobacter pylori in Peptic Ulcer Disease: A Noninvasive Primary Care Option Michael W Felz, MD, George] Burke, MD, and Bernard M. Schuman, MD Background: Hellcobacter
ORIGINAL ARTICLES Breath Test Diagnosis of Helicobacter pylori in Peptic Ulcer Disease: A Noninvasive Primary Care Option Michael W Felz, MD, George] Burke, MD, and Bernard M. Schuman, MD Background: Hellcobacter
PDF hosted at the Radboud Repository of the Radboud University Nijmegen
 PDF hosted at the Radboud Repository of the Radboud University Nijmegen The following full text is a publisher's version. For additional information about this publication click this link. http://hdl.handle.net/2066/48400
PDF hosted at the Radboud Repository of the Radboud University Nijmegen The following full text is a publisher's version. For additional information about this publication click this link. http://hdl.handle.net/2066/48400
H. pylori Antigen ELISA Kit
 H. pylori Antigen ELISA Kit Catalog Number KA3142 96 assays Version: 04 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
H. pylori Antigen ELISA Kit Catalog Number KA3142 96 assays Version: 04 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
Comparison of the Accuracy of Two Commercial Rapid Urase Tests, CLOtest and Pronto Dry, in Detecting Helicobacter pylori Infection ABSTRACT
 Original Article Rojborwonwitaya J, Vijitjunyakul N THAI J GASTROENTEROL 2005 Vol. 6 No. 2 May - Aug. 2005 55 Comparison of the Accuracy of Two Commercial Rapid Urase Tests, CLOtest and Pronto Dry, in
Original Article Rojborwonwitaya J, Vijitjunyakul N THAI J GASTROENTEROL 2005 Vol. 6 No. 2 May - Aug. 2005 55 Comparison of the Accuracy of Two Commercial Rapid Urase Tests, CLOtest and Pronto Dry, in
Utility of In House made Rapid Urease Broth Test for Detection of Helicobacter pylori Infection in Resource Constraint Settings
 Original article: Utility of In House made Rapid Urease Broth Test for Detection of Helicobacter pylori Infection in Resource Constraint Settings 1.Dr. Swati Rahul Dhope, 2. Dr. Sachinkumar Vasantrao Wankhede
Original article: Utility of In House made Rapid Urease Broth Test for Detection of Helicobacter pylori Infection in Resource Constraint Settings 1.Dr. Swati Rahul Dhope, 2. Dr. Sachinkumar Vasantrao Wankhede
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: helicobacter_pylori_testing 01/01/2019 N/A 01/01/2020 01/01/2019 Policy Effective April 1, 2019 Description
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: helicobacter_pylori_testing 01/01/2019 N/A 01/01/2020 01/01/2019 Policy Effective April 1, 2019 Description
Helicobacter pylori Improved Detection of Helicobacter pylori
 DOI:http://dx.doi.org/10.7314/APJCP.2016.17.4.2099 RESEARCH ARTICLE Improved Detection of Helicobacter pylori Infection and Premalignant Gastric Mucosa Using Conventional White Light Source Gastroscopy
DOI:http://dx.doi.org/10.7314/APJCP.2016.17.4.2099 RESEARCH ARTICLE Improved Detection of Helicobacter pylori Infection and Premalignant Gastric Mucosa Using Conventional White Light Source Gastroscopy
Helicobacter pylori: drowning in a pool of blood?
 Helicobacter pylori: drowning in a pool of blood? Diagnosis of Helicobacter pylori infection is crucial in the short-term and long-term management of patients with bleeding ulcers. If a patient with ulcer
Helicobacter pylori: drowning in a pool of blood? Diagnosis of Helicobacter pylori infection is crucial in the short-term and long-term management of patients with bleeding ulcers. If a patient with ulcer
H.Pylori IgG
 DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
Imprint cytology: a simple, cost effectiveness analysis for diagnosing Helicobacter pylori, in west of Iran
 Original Research Medical Journal of Islamic Republic of Iran, Vol. 26, No. 1, Feb. 2012, pp. 12-16 Imprint cytology: a simple, cost effectiveness analysis for diagnosing Helicobacter pylori, in west of
Original Research Medical Journal of Islamic Republic of Iran, Vol. 26, No. 1, Feb. 2012, pp. 12-16 Imprint cytology: a simple, cost effectiveness analysis for diagnosing Helicobacter pylori, in west of
- Helicobacter - THE EASE AND DIFFICULTY OF A NEW DISCOVERY. Robin Warren
 - Helicobacter - THE EASE AND DIFFICULTY OF A NEW DISCOVERY Robin Warren EARLY DAYS First reports 100 years ago considered spirochaetes 1940 Freedburg saw curved organisms in the stomach 1954 Palmer: Freedburg
- Helicobacter - THE EASE AND DIFFICULTY OF A NEW DISCOVERY Robin Warren EARLY DAYS First reports 100 years ago considered spirochaetes 1940 Freedburg saw curved organisms in the stomach 1954 Palmer: Freedburg
Helicobacter and gastritis
 1 Helicobacter and gastritis Dr. Hala Al Daghistani Helicobacter pylori is a spiral-shaped gram-negative rod. H. pylori is associated with antral gastritis, duodenal (peptic) ulcer disease, gastric ulcers,
1 Helicobacter and gastritis Dr. Hala Al Daghistani Helicobacter pylori is a spiral-shaped gram-negative rod. H. pylori is associated with antral gastritis, duodenal (peptic) ulcer disease, gastric ulcers,
H.Pylori IgG Cat # 1503Z
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Correlation Between Endoscopic and Histological Findings in Different Gastroduodenal Lesion and its Association with Helicobacter Pylori
 ORIGINAL ARTICLE Correlation Between Endoscopic and Histological Findings in Different Gastroduodenal Lesion and its Association with Helicobacter Pylori *A. Sultana 1, SM Badruddoza 2, F Rahman 3 1 Dr.
ORIGINAL ARTICLE Correlation Between Endoscopic and Histological Findings in Different Gastroduodenal Lesion and its Association with Helicobacter Pylori *A. Sultana 1, SM Badruddoza 2, F Rahman 3 1 Dr.
Research Article Performance of Routine Helicobacter pylori Invasive Tests in Patients with Dyspepsia
 Gastroenterology Research and Practice Volume 2013, Article ID 184806, 5 pages http://dx.doi.org/10.1155/2013/184806 Research Article Performance of Routine Helicobacter pylori Invasive Tests in Patients
Gastroenterology Research and Practice Volume 2013, Article ID 184806, 5 pages http://dx.doi.org/10.1155/2013/184806 Research Article Performance of Routine Helicobacter pylori Invasive Tests in Patients
Gastric atrophy: use of OLGA staging system in practice
 Gastroenterology and Hepatology From Bed to Bench. 2016 RIGLD, Research Institute for Gastroenterology and Liver Diseases ORIGINAL ARTICLE Gastric atrophy: use of OLGA staging system in practice Mahsa
Gastroenterology and Hepatology From Bed to Bench. 2016 RIGLD, Research Institute for Gastroenterology and Liver Diseases ORIGINAL ARTICLE Gastric atrophy: use of OLGA staging system in practice Mahsa
Value of Serology as a Noninvasive Method for Evaluating the Efficacy of Treatment of Helicobacter pylori Infection
 1038 Value of Serology as a Noninvasive Method for Evaluating the Efficacy of Treatment of Helicobacter pylori Infection Guillermo I. Pérez-Pérez, Alan F. Cutler, and Martin J. Blaser From the Division
1038 Value of Serology as a Noninvasive Method for Evaluating the Efficacy of Treatment of Helicobacter pylori Infection Guillermo I. Pérez-Pérez, Alan F. Cutler, and Martin J. Blaser From the Division
Disclosures. Co-founder and Chief Science Officer, TechLab
 H. pylori testing Disclosures Co-founder and Chief Science Officer, TechLab Learning Objectives Evaluate the appropriate testing methodology by balancing performance, economics, and workflow. Discuss the
H. pylori testing Disclosures Co-founder and Chief Science Officer, TechLab Learning Objectives Evaluate the appropriate testing methodology by balancing performance, economics, and workflow. Discuss the
Intragastric Acidification Reduces the Occurrence of False-Negative Urea Breath Test Results in Patients Taking a Proton Pump Inhibitor
 THE AMERICAN JOURNAL OF GASTROENTEROLOGY Vol. 96, No. 4, 2001 2001 by Am. Coll. of Gastroenterology ISSN 0002-9270/01/$20.00 Published by Elsevier Science Inc. PII S0002-9270(01)02250-X Intragastric Acidification
THE AMERICAN JOURNAL OF GASTROENTEROLOGY Vol. 96, No. 4, 2001 2001 by Am. Coll. of Gastroenterology ISSN 0002-9270/01/$20.00 Published by Elsevier Science Inc. PII S0002-9270(01)02250-X Intragastric Acidification
The Association of CagA + Helicobacter pylori Infection and Gastric Carcinoma
 The Association of CagA + Helicobacter pylori Infection and Gastric Carcinoma PRESENTER: Dr. Md. Khalilur Rahman Student of M.D.(Internal Medicine) Sylhet MAG Osmani Medical College Gastric Cancer- ranked
The Association of CagA + Helicobacter pylori Infection and Gastric Carcinoma PRESENTER: Dr. Md. Khalilur Rahman Student of M.D.(Internal Medicine) Sylhet MAG Osmani Medical College Gastric Cancer- ranked
Case Report Features of the Atrophic Corpus Mucosa in Three Cases of Autoimmune Gastritis Revealed by Magnifying Endoscopy
 Volume 2012, Article ID 368160, 4 pages doi:10.1155/2012/368160 Case Report Features of the Atrophic Corpus Mucosa in Three Cases of Autoimmune Gastritis Revealed by Magnifying Endoscopy Kazuyoshi Yagi,
Volume 2012, Article ID 368160, 4 pages doi:10.1155/2012/368160 Case Report Features of the Atrophic Corpus Mucosa in Three Cases of Autoimmune Gastritis Revealed by Magnifying Endoscopy Kazuyoshi Yagi,
Correlation between Gastric Mucosal Morphologic Patterns and Histopathological Severity of
 Hindawi Publishing Corporation BioMed Research International Volume 2015, Article ID 808505, 7 pages http://dx.doi.org/10.1155/2015/808505 Research Article Correlation between Gastric Mucosal Morphologic
Hindawi Publishing Corporation BioMed Research International Volume 2015, Article ID 808505, 7 pages http://dx.doi.org/10.1155/2015/808505 Research Article Correlation between Gastric Mucosal Morphologic
Research Article Reliability of Diagnostic Tests for Helicobacterpylori Infection
 Gastroenterology Research and Practice Volume 211, Article ID 946, 6 pages doi:1.1155/211/946 Research Article Reliability of Diagnostic Tests for Helicobacterpylori Infection S. Redéen, 1, 2 F. Petersson,
Gastroenterology Research and Practice Volume 211, Article ID 946, 6 pages doi:1.1155/211/946 Research Article Reliability of Diagnostic Tests for Helicobacterpylori Infection S. Redéen, 1, 2 F. Petersson,
H. pylori IgM CLIA kit
 H. pylori IgM CLIA kit Cat. No.:DEEL0251 Pkg.Size:96 tests Intended use Helicobacter pylori IgM Chemiluminescence ELISA is intended for use in evaluating the serologic status to H. pylori infection in
H. pylori IgM CLIA kit Cat. No.:DEEL0251 Pkg.Size:96 tests Intended use Helicobacter pylori IgM Chemiluminescence ELISA is intended for use in evaluating the serologic status to H. pylori infection in
See external label 2 C-8 C Σ=96 tests Cat # 1505Z. MICROWELL ELISA H.Pylori IgA Cat # 1505Z
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
H.pylori IgA Cat #
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
H. pylori IgM. Cat # H. pylori IgM ELISA. ELISA: Enzyme Linked Immunosorbent Assay. ELISA - Indirect; Antigen Coated Plate
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com H. pylori
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com H. pylori
For the convenience of storage, Cassette and Sample Tubes can be stored separately. OneStep. H.Pylori Antigen. RapiCard InstaTest.
 CORTEZ DIAGNOSTICS, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 USA Tel: (818) 591-3030 Fax: (818) 591-8383 E-mail: onestep@rapidtest.com Web site: www.rapidtest.com See external label
CORTEZ DIAGNOSTICS, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 USA Tel: (818) 591-3030 Fax: (818) 591-8383 E-mail: onestep@rapidtest.com Web site: www.rapidtest.com See external label
H.Pylori IgM Cat # 1504Z
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Screening (Diagnostic Tests) Shaker Salarilak
 Screening (Diagnostic Tests) Shaker Salarilak Outline Screening basics Evaluation of screening programs Where we are? Definition of screening? Whether it is always beneficial? Types of bias in screening?
Screening (Diagnostic Tests) Shaker Salarilak Outline Screening basics Evaluation of screening programs Where we are? Definition of screening? Whether it is always beneficial? Types of bias in screening?
Helicobacter pylori 幽門螺旋桿菌 馬偕紀念醫院新竹分院一般內科, 肝膽腸胃科陳重助醫師
 Helicobacter pylori 幽門螺旋桿菌 馬偕紀念醫院新竹分院一般內科, 肝膽腸胃科陳重助醫師 Hp : Helicobacter pylori Part 1. Pathophysiology and immune response Pathogenesis of Hp infection Part 2. Clinical manifestation Part 3. Dx tests for
Helicobacter pylori 幽門螺旋桿菌 馬偕紀念醫院新竹分院一般內科, 肝膽腸胃科陳重助醫師 Hp : Helicobacter pylori Part 1. Pathophysiology and immune response Pathogenesis of Hp infection Part 2. Clinical manifestation Part 3. Dx tests for
Table 2.9. Case control studies of helicobacter pylori infection and oesophageal adenocarcinoma
 Characteristics of Characteristics of controls Detection Chow et al (1998) 1993-1995 129 of newly diagnosed oesophageal/gastric cardia (OGC) adenocarcinoma. 224 population controls selected by random digit
Characteristics of Characteristics of controls Detection Chow et al (1998) 1993-1995 129 of newly diagnosed oesophageal/gastric cardia (OGC) adenocarcinoma. 224 population controls selected by random digit
ORIGINAL INVESTIGATION. Effectiveness of Helicobacter pylori Therapies in a Clinical Practice Setting
 ORIGINAL INVESTIGATION Effectiveness of Helicobacter pylori Therapies in a Clinical Practice Setting M. Brian Fennerty, MD; David A. Lieberman, MD; Nimish Vakil, MD; Nathan Magaret; Douglas O. Faigel,
ORIGINAL INVESTIGATION Effectiveness of Helicobacter pylori Therapies in a Clinical Practice Setting M. Brian Fennerty, MD; David A. Lieberman, MD; Nimish Vakil, MD; Nathan Magaret; Douglas O. Faigel,
Evaluation of a new rapid immunoassay for the detection of Helicobacter pylori in faeces: a prospective pilot study
 Aliment Pharmacol Ther 2005; 21: 485 489. doi: 10.1111/j.1365-2036.2005.02355.x Evaluation of a new rapid immunoassay for the detection of Helicobacter pylori in faeces: a prospective pilot study L. TREVISANI*,
Aliment Pharmacol Ther 2005; 21: 485 489. doi: 10.1111/j.1365-2036.2005.02355.x Evaluation of a new rapid immunoassay for the detection of Helicobacter pylori in faeces: a prospective pilot study L. TREVISANI*,
TRANSPARENCY COMMITTEE OPINION. 13 December 2006
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 13 December 2006 HELIKIT 75 mg, powder for oral solution CIP : 343 132-1 Applicant : MAYOLY SPINDLER 13 Curea anhydrous
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 13 December 2006 HELIKIT 75 mg, powder for oral solution CIP : 343 132-1 Applicant : MAYOLY SPINDLER 13 Curea anhydrous
Fecoprevalence and determinants of Helicobacter pylor infection among asymptomatic children in Myanmar
 International Journal of Gastroenterology, Hepatology, Transplant & Nutrition Original Article Fecoprevalence and determinants of Helicobacter pylor infection among asymptomatic children in Myanmar Hnin
International Journal of Gastroenterology, Hepatology, Transplant & Nutrition Original Article Fecoprevalence and determinants of Helicobacter pylor infection among asymptomatic children in Myanmar Hnin
The PYtest The Cutting Edge Urea Breath Test to detect Helicobacter pylori
 The PYtest The Cutting Edge Urea Breath Test to detect Helicobacter pylori Barry Marshall The Gold-Standard Non-invasive Diagnostic Test for The Detection of H.pylori As a Healthcare Provider... Do you
The PYtest The Cutting Edge Urea Breath Test to detect Helicobacter pylori Barry Marshall The Gold-Standard Non-invasive Diagnostic Test for The Detection of H.pylori As a Healthcare Provider... Do you
RAPID DIAGNOSIS OF HELICOBACTER PYLORI INFECTION IN GASTRIC IMPRINT SMEARS
 RAPID DIAGNOSIS OF HELICOBACTER PYLORI INFECTION IN GASTRIC IMPRINT SMEARS Gurjeet Kaur 1, Manoharan Madhavan 1, Amir Hakim Basri 2, Abdul Hamid Mat Sain 3, Mohd Shaiful Bahrun Hussain 3, Mohd Kamal Yatiban
RAPID DIAGNOSIS OF HELICOBACTER PYLORI INFECTION IN GASTRIC IMPRINT SMEARS Gurjeet Kaur 1, Manoharan Madhavan 1, Amir Hakim Basri 2, Abdul Hamid Mat Sain 3, Mohd Shaiful Bahrun Hussain 3, Mohd Kamal Yatiban
Helicobacter Pylori Testing HELICOBACTER PYLORI TESTING HS-131. Policy Number: HS-131. Original Effective Date: 9/17/2009
 Easy Choice Health Plan, Inc. Harmony Health Plan of Illinois, Inc. Missouri Care, Inc. Ohana Health Plan, a plan offered by WellCare Health Insurance of Arizona, Inc. WellCare Health Insurance of Illinois,
Easy Choice Health Plan, Inc. Harmony Health Plan of Illinois, Inc. Missouri Care, Inc. Ohana Health Plan, a plan offered by WellCare Health Insurance of Arizona, Inc. WellCare Health Insurance of Illinois,
Index. Note: Page numbers of article titles are in boldface type.
 Note: Page numbers of article titles are in boldface type. A Adherence, to bismuth quadruple therapy, 543 546 Adjuvant therapy, probiotics as, 567 569 Age factors, in gastric cancer, 611 612, 616 AID protein,
Note: Page numbers of article titles are in boldface type. A Adherence, to bismuth quadruple therapy, 543 546 Adjuvant therapy, probiotics as, 567 569 Age factors, in gastric cancer, 611 612, 616 AID protein,
Helicobacter Connections. Barry Marshall
 Helicobacter Connections Barry Marshall The greatest obstacle to knowledge is not ignorance, it is the illusion of knowledge. Daniel Boorstein - Historian Peptic Ulcers Duodenal Ulcer (DU) Gastric Ulcer
Helicobacter Connections Barry Marshall The greatest obstacle to knowledge is not ignorance, it is the illusion of knowledge. Daniel Boorstein - Historian Peptic Ulcers Duodenal Ulcer (DU) Gastric Ulcer
H. pylori Stool Rapid Test (Cassette)
 H. pylori Stool Rapid Test (Cassette) Cat. No.:DTS590 Pkg.Size: Intended use The H. pylori Stool Cassette is an immunochromatographic screening assay for the qualitative detectionof Helicobacter pylori
H. pylori Stool Rapid Test (Cassette) Cat. No.:DTS590 Pkg.Size: Intended use The H. pylori Stool Cassette is an immunochromatographic screening assay for the qualitative detectionof Helicobacter pylori
Key words : low-grade MALT lymphoma, epithelial change, empty lamina propria, B-cell clonality, H. pylori, eradication, long-term follow-up
 Department of Pathology, The University of Tokushima School of Medicine, Tokushima, Japan ; Department of Oral and Maxillofacial Surgery, The University of Tokushima School of Dentistry, Tokushima, Japan
Department of Pathology, The University of Tokushima School of Medicine, Tokushima, Japan ; Department of Oral and Maxillofacial Surgery, The University of Tokushima School of Dentistry, Tokushima, Japan
Cases requiring intra-departmental or extra-departmental consultation
 CMS ID/CMS QCDR ID: CAP 28 Title: Helicobacter pylori Status and Turnaround Time Specifications Description CAP QCDR Percentage of stomach biopsy cases with gastritis that addresses presence or absence
CMS ID/CMS QCDR ID: CAP 28 Title: Helicobacter pylori Status and Turnaround Time Specifications Description CAP QCDR Percentage of stomach biopsy cases with gastritis that addresses presence or absence
PYTEST - urea c-14 capsule Avent, Inc Description
 PYTEST - urea c-4 capsule Avent, Inc. ---------- PYtest ( 4 Description 4 C-Urea Capsules) PYtest ( C-urea capsules) is intended for use in the detection of gastric urease as an aid in the diagnosis of
PYTEST - urea c-4 capsule Avent, Inc. ---------- PYtest ( 4 Description 4 C-Urea Capsules) PYtest ( C-urea capsules) is intended for use in the detection of gastric urease as an aid in the diagnosis of
MOSAIC APPEARANCE OF GASTRIC MUCOSA AS A PREDICTOR FOR HELICOBACTER PYLORI INFECTION
 Basrah Journal Original Article Of Surgery MOSAIC APPEARANCE OF GASTRIC MUCOSA AS A PREDICTOR FOR HELICOBACTER PYLORI INFECTION Ali Adnan Mohsin* & Sarkis K Strak @ *MBChB Registrar, @ MBChB, MRCP, FRCP,
Basrah Journal Original Article Of Surgery MOSAIC APPEARANCE OF GASTRIC MUCOSA AS A PREDICTOR FOR HELICOBACTER PYLORI INFECTION Ali Adnan Mohsin* & Sarkis K Strak @ *MBChB Registrar, @ MBChB, MRCP, FRCP,
Introduction. Original articles. Nicolás Rocha, 1 Sandra Huertas, 2 Rosario Albis, 3 Diego Aponte, 4 Luis Carlos Sabbagh. 5
 Original articles Correlation of endoscopic and histological findings in diagnosis of gastrointestinal metaplasia in patients referred to the Clinica Colombia for upper endoscopies Nicolás Rocha, 1 Sandra
Original articles Correlation of endoscopic and histological findings in diagnosis of gastrointestinal metaplasia in patients referred to the Clinica Colombia for upper endoscopies Nicolás Rocha, 1 Sandra
ÐÑÏÓÊÅÊËÇÌÅÍÅÓ ÎÅÍÏÃËÙÓÓÅÓ ÁÍÁÊÏÉÍÙÓÅÉÓ ÅËËÇÍÙÍ ÅÑÅÕÍÇÔÙÍ
 ÐÑÏÓÊÅÊËÇÌÅÍÅÓ ÎÅÍÏÃËÙÓÓÅÓ ÁÍÁÊÏÉÍÙÓÅÉÓ ÅËËÇÍÙÍ ÅÑÅÕÍÇÔÙÍ 205 206 ÐÑÏÓÊÅÊËÇÌÅÍÅÓ ÎÅÍÏÃËÙÓÓÅÓ ÁÍÁÊÏÉÍÙÓÅÉÓ ÅËËÇÍÙÍ ÅÑÅÕÍÇÔÙÍ EPITHELIAL CELL TURNOVER IN NON-DYSPLASTIC GASTRIC MUCOSA ADJACENT TO EARLY AND
ÐÑÏÓÊÅÊËÇÌÅÍÅÓ ÎÅÍÏÃËÙÓÓÅÓ ÁÍÁÊÏÉÍÙÓÅÉÓ ÅËËÇÍÙÍ ÅÑÅÕÍÇÔÙÍ 205 206 ÐÑÏÓÊÅÊËÇÌÅÍÅÓ ÎÅÍÏÃËÙÓÓÅÓ ÁÍÁÊÏÉÍÙÓÅÉÓ ÅËËÇÍÙÍ ÅÑÅÕÍÇÔÙÍ EPITHELIAL CELL TURNOVER IN NON-DYSPLASTIC GASTRIC MUCOSA ADJACENT TO EARLY AND
The significance of Helicobacter pylori in the approach of dyspepsia in primary care Arents, Nicolaas Lodevikus Augustinus
 University of Groningen The significance of Helicobacter pylori in the approach of dyspepsia in primary care Arents, Nicolaas Lodevikus Augustinus IMPORTANT NOTE: You are advised to consult the publisher's
University of Groningen The significance of Helicobacter pylori in the approach of dyspepsia in primary care Arents, Nicolaas Lodevikus Augustinus IMPORTANT NOTE: You are advised to consult the publisher's
Novel diagnostic tests to detect Helicobacter pylori infection: A pediatric perspective
 CANADIAN HELICOBACTER CONSENSUS CONFERENCE ON PEDIATRIC ISSUES Novel diagnostic tests to detect Helicobacter pylori infection: A pediatric perspective John D Snyder MD 1, Sander Veldhuyzen van Zanten MD
CANADIAN HELICOBACTER CONSENSUS CONFERENCE ON PEDIATRIC ISSUES Novel diagnostic tests to detect Helicobacter pylori infection: A pediatric perspective John D Snyder MD 1, Sander Veldhuyzen van Zanten MD
Rapid-VIDITEST. Helicobacter pylori. One step Helicobacter pylori Blister test. Instruction manual
 Rapid-VIDITEST Helicobacter pylori One step Helicobacter pylori Blister test. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, 252 50 Vestec, Czech Republic, Tel.: +420 261 090 565,
Rapid-VIDITEST Helicobacter pylori One step Helicobacter pylori Blister test. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, 252 50 Vestec, Czech Republic, Tel.: +420 261 090 565,
MEDICAL POLICY EFFECTIVE DATE: 05/19/11 REVISED DATE: 05/24/12, 05/23/13 ARCHIVED DATE: 05/22/14 EDITED DATE: 05/28/15, 05/25/16, 05/18/17, 05/17/18
 MEDICAL POLICY SUBJECT: NON-INVASIVE HELICOBACTER PAGE: 1 OF: 6 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including
MEDICAL POLICY SUBJECT: NON-INVASIVE HELICOBACTER PAGE: 1 OF: 6 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including
Immunoglobulin G Antibody against Helicobacter pylori: Clinical Implications of Levels Found in Serum
 CLINICAL AND DIAGNOSTIC LABORATORY IMMUNOLOGY, Sept. 2002, p. 1044 1048 Vol. 9, No. 5 1071-412X/02/$04.00 0 DOI: 10.1128/CDLI.9.5.1044 1048.2002 Copyright 2002, American Society for Microbiology. All Rights
CLINICAL AND DIAGNOSTIC LABORATORY IMMUNOLOGY, Sept. 2002, p. 1044 1048 Vol. 9, No. 5 1071-412X/02/$04.00 0 DOI: 10.1128/CDLI.9.5.1044 1048.2002 Copyright 2002, American Society for Microbiology. All Rights
instrument. When 13C-UBT positive value is greater than or equal to / - 0.4, the the subject can be 1. Data and methods details are as follows:
 Application of 13 C-urea breath test in screening helicobacter pylori infection during health examination in Chengdu, Sichuan YANG Yan-hua. LIU Yu-ping. CHENG You-fu, SHUAI Ping. LU Qiao. ZHENG Xiao-xia,
Application of 13 C-urea breath test in screening helicobacter pylori infection during health examination in Chengdu, Sichuan YANG Yan-hua. LIU Yu-ping. CHENG You-fu, SHUAI Ping. LU Qiao. ZHENG Xiao-xia,
RESEARCH COMMUNICATION. Comparison of Helicobacter pylori Antibody Detection in Stool with other Diagnostic Tests for Infection
 Comparison of Helicobacter pylori Antibody Detection in Stool with other Diagnostic Tests RESEARCH COMMUNICATION Comparison of Helicobacter pylori Antibody Detection in Stool with other Diagnostic Tests
Comparison of Helicobacter pylori Antibody Detection in Stool with other Diagnostic Tests RESEARCH COMMUNICATION Comparison of Helicobacter pylori Antibody Detection in Stool with other Diagnostic Tests
THE PREVALENCE OF HELICBACTER PYLORI AMONG PATIENTS COMPLAINING FROM ABDOMINAL PAIN
 THE PREVALENCE OF HELICBACTER PYLORI AMONG PATIENTS COMPLAINING FROM ABDOMINAL PAIN Ahed J. Al-Khatib Jordan University of Science and Technology, Jordan Ahmed Saber Abu-zaiton Al-albayt University Abstract
THE PREVALENCE OF HELICBACTER PYLORI AMONG PATIENTS COMPLAINING FROM ABDOMINAL PAIN Ahed J. Al-Khatib Jordan University of Science and Technology, Jordan Ahmed Saber Abu-zaiton Al-albayt University Abstract
Detection of Helicobacter pylori Gastritis by PCR Correlation With Inflammation Scores and Immunohistochemical and CLOtest Findings
 Anatomic Pathology / HELICOBACTER PYLORI GASTRITIS Detection of Helicobacter pylori Gastritis by PCR Correlation With Inflammation Scores and Immunohistochemical and CLOtest Findings Jason Weiss, DO, 1,2
Anatomic Pathology / HELICOBACTER PYLORI GASTRITIS Detection of Helicobacter pylori Gastritis by PCR Correlation With Inflammation Scores and Immunohistochemical and CLOtest Findings Jason Weiss, DO, 1,2
Helicobacter 2008;13:1-6. Am J Gastroent 2007;102: Am J of Med 2004;117:31-35.
 An Update on Helicobacter pylori and Its Treatment Trenika Mitchell, PharmD, BCPS Clinical Assistant Professor University of Kentucky College of Pharmacy October 18, 2008 Objectives Review the epidemiology
An Update on Helicobacter pylori and Its Treatment Trenika Mitchell, PharmD, BCPS Clinical Assistant Professor University of Kentucky College of Pharmacy October 18, 2008 Objectives Review the epidemiology
Association of Helicobacter pylori infection with Atrophic gastritis in patients with Dyspepsia
 ADVANCES IN BIORESEARCH Adv. Biores., Vol 8 [3] May 2017: 137-141 2017 Society of Education, India Print ISSN 0976-4585; Online ISSN 2277-1573 Journal s URL:http://www.soeagra.com/abr.html CODEN: ABRDC3
ADVANCES IN BIORESEARCH Adv. Biores., Vol 8 [3] May 2017: 137-141 2017 Society of Education, India Print ISSN 0976-4585; Online ISSN 2277-1573 Journal s URL:http://www.soeagra.com/abr.html CODEN: ABRDC3
Quantitative Image Analysis of HER2 Immunohistochemistry for Breast Cancer
 Quantitative Image Analysis of HER2 Immunohistochemistry for Breast Cancer Guideline from the College of American Pathologists Early Online Release Publication: Archives of Pathology & Laboratory Medicine
Quantitative Image Analysis of HER2 Immunohistochemistry for Breast Cancer Guideline from the College of American Pathologists Early Online Release Publication: Archives of Pathology & Laboratory Medicine
Helicobacter pylori:an Emerging Pathogen
 Bacteriology at UW-Madison Bacteriology 330 Home Page Helicobacter pylori:an Emerging Pathogen by Karrie Holston, Department of Bacteriology University of Wisconsin-Madison Description of Helicobacter
Bacteriology at UW-Madison Bacteriology 330 Home Page Helicobacter pylori:an Emerging Pathogen by Karrie Holston, Department of Bacteriology University of Wisconsin-Madison Description of Helicobacter
Validation of serological tests for Helicobacter pylori infection in an Irish population
 original paper Validation of serological tests for Helicobacter pylori infection in an Irish population NP Breslin, JM Lee, MJ Buckley, E Balbirnie 1, D Rice, 1 CA O'Morain Tallaght Regional Hospital,
original paper Validation of serological tests for Helicobacter pylori infection in an Irish population NP Breslin, JM Lee, MJ Buckley, E Balbirnie 1, D Rice, 1 CA O'Morain Tallaght Regional Hospital,
Health technology Serological testing and endoscopy with biopsy for suspected peptic ulcer disease.
 Immediate endoscopy or initial Helicobacter pylori serological testing for suspected peptic ulcer disease: estimating cost-effectiveness using decision analysis Fendrick A M, Chernew M E, Hirth R A, Bloom
Immediate endoscopy or initial Helicobacter pylori serological testing for suspected peptic ulcer disease: estimating cost-effectiveness using decision analysis Fendrick A M, Chernew M E, Hirth R A, Bloom
Helicobacter pylori Antigen Test
 Helicobacter pylori Antigen Test Instructions For Use Format: Cassette For: Catalog Number: VEL-001-HP(s) Specimen: Fecal Specimen * Please read the instructions carefully before use INTENDED USE Helicobacter
Helicobacter pylori Antigen Test Instructions For Use Format: Cassette For: Catalog Number: VEL-001-HP(s) Specimen: Fecal Specimen * Please read the instructions carefully before use INTENDED USE Helicobacter
JMSCR Vol 05 Issue 08 Page August 2017
 www.jmscr.igmpublication.org Impact Factor 5.84 Index Copernicus Value: 83.27 ISSN (e)-2347-176x ISSN (p) 2455-0450 DOI: https://dx.doi.org/10.18535/jmscr/v5i8.83 Role of Special Stain Giemsa in Demonstration
www.jmscr.igmpublication.org Impact Factor 5.84 Index Copernicus Value: 83.27 ISSN (e)-2347-176x ISSN (p) 2455-0450 DOI: https://dx.doi.org/10.18535/jmscr/v5i8.83 Role of Special Stain Giemsa in Demonstration
Campylobacter like organisms on the gastric mucosa:
 J Clin Pathol 1984;37:1002-1006 Campylobacter like organisms on the gastric mucosa: culture, histological, and serological studies DM JONES,* AM LESSELLS,t JOAN ELDRIDGE* From the *Public Health Laboratory
J Clin Pathol 1984;37:1002-1006 Campylobacter like organisms on the gastric mucosa: culture, histological, and serological studies DM JONES,* AM LESSELLS,t JOAN ELDRIDGE* From the *Public Health Laboratory
Questionnaire design. Questionnaire Design: Content. Questionnaire Design. Questionnaire Design: Wording. Questionnaire Design: Wording OUTLINE
 Questionnaire design OUTLINE Questionnaire design tests Reliability Validity POINTS TO CONSIDER Identify your research objectives. Identify your population or study sample Decide how to collect the information
Questionnaire design OUTLINE Questionnaire design tests Reliability Validity POINTS TO CONSIDER Identify your research objectives. Identify your population or study sample Decide how to collect the information
The long-term management of patients with bleeding duodenal ulcers
 Aliment Pharmacol Ther (1997); 11: 505±510. The long-term management of patients with bleeding duodenal ulcers M. E. MCALINDON, J. S. W. TAYLOR & S. D. RYDER Department of Medicine, University Hospital,
Aliment Pharmacol Ther (1997); 11: 505±510. The long-term management of patients with bleeding duodenal ulcers M. E. MCALINDON, J. S. W. TAYLOR & S. D. RYDER Department of Medicine, University Hospital,
Ethnic Distribution of Atrophic Autoimmune Gastritis in the United States
 Ethnic Distribution of Atrophic Autoimmune Gastritis in the United States Robert M. Genta, Regan Allen, Massimo Rugge Miraca Life Sciences Research Institute, Miraca Life Sciences, Irving, Texas UTSW University
Ethnic Distribution of Atrophic Autoimmune Gastritis in the United States Robert M. Genta, Regan Allen, Massimo Rugge Miraca Life Sciences Research Institute, Miraca Life Sciences, Irving, Texas UTSW University
~Helicobacter pylori, gastritis, and peptic ulceration
 17 Department of Pathology J I Wyatt Department of Medicine T M Shallcross J E Crabtree RV Heatley St James University Hospital, Leeds LS9 7TF Correspondence to: Dr J I Wyatt. Accepted for publication
17 Department of Pathology J I Wyatt Department of Medicine T M Shallcross J E Crabtree RV Heatley St James University Hospital, Leeds LS9 7TF Correspondence to: Dr J I Wyatt. Accepted for publication
SCIENTIFIC DISCUSSION
 SCIENTIFIC DISCUSSION This module reflects the initial scientific discussion and scientific discussion on procedures, which have been finalised before 1 October 2001. For scientific information on procedures
SCIENTIFIC DISCUSSION This module reflects the initial scientific discussion and scientific discussion on procedures, which have been finalised before 1 October 2001. For scientific information on procedures
Evaluation of Three Commercial Enzyme Immunoassays Compared with the 13 C Urea Breath Test for Detection of Helicobacter pylori Infection
 JOURNAL OF CLINICAL MICROBIOLOGY, May 1996, p. 1147 1152 Vol. 34, No. 5 0095-1137/96/$04.00 0 Copyright 1996, American Society for Microbiology Evaluation of Three Commercial Enzyme Immunoassays Compared
JOURNAL OF CLINICAL MICROBIOLOGY, May 1996, p. 1147 1152 Vol. 34, No. 5 0095-1137/96/$04.00 0 Copyright 1996, American Society for Microbiology Evaluation of Three Commercial Enzyme Immunoassays Compared
PYTEST- urea, c-14 capsule Avent, Inc Des cription
 PYTEST- urea, c-4 capsule Avent, Inc. ---------- 4 PYtes t ( C-Urea Caps ules) Des cription 4 PYtest ( C-urea capsules) is intended for use in the detection of gastric urease as an aid in the diagnosis
PYTEST- urea, c-4 capsule Avent, Inc. ---------- 4 PYtes t ( C-Urea Caps ules) Des cription 4 PYtest ( C-urea capsules) is intended for use in the detection of gastric urease as an aid in the diagnosis
CLIA Complexity for Whole Blood: WAIVED. Intended Use. Summary and Explanation
 CLIA Complexity for Whole Blood: WAIVED Intended Use The is a lateral-flow immunoassay intended for the rapid, qualitative detection of IgG antibodies specific to Helicobacter pylori in whole blood. The
CLIA Complexity for Whole Blood: WAIVED Intended Use The is a lateral-flow immunoassay intended for the rapid, qualitative detection of IgG antibodies specific to Helicobacter pylori in whole blood. The
Catalog # 0W009 0W010 Test Cassettes, murine monoclonal antibody to human IgG (Test Line) and rabbit polyclonal antibody (Control Line)
 For in vitro diagnostic use. CLIA Complexity for Whole Blood: WAIVED INTENDED USE The QuickVue H. pylori Test is a lateral-flow immunoassay intended for the rapid, qualitative detection of IgG antibodies
For in vitro diagnostic use. CLIA Complexity for Whole Blood: WAIVED INTENDED USE The QuickVue H. pylori Test is a lateral-flow immunoassay intended for the rapid, qualitative detection of IgG antibodies
pylori positive patients with duodenal ulcer
 Gut 1996; 39: 629-633 629 PAPERS Prevalence of Helicobacter pylori infection and related gastroduodenal lesions in spouses of Helicobacter pylori positive patients with duodenal ulcer F Parente, G Maconi,
Gut 1996; 39: 629-633 629 PAPERS Prevalence of Helicobacter pylori infection and related gastroduodenal lesions in spouses of Helicobacter pylori positive patients with duodenal ulcer F Parente, G Maconi,
Laboratory Considerations
 Laboratory Considerations In Support of HPV Vaccine Programs Elizabeth R. Unger PhD, MD Team Leader HPV Laboratory Centers for Disease Prevention and Control Towards Comprehensive Cervical Cancer Control:
Laboratory Considerations In Support of HPV Vaccine Programs Elizabeth R. Unger PhD, MD Team Leader HPV Laboratory Centers for Disease Prevention and Control Towards Comprehensive Cervical Cancer Control:
Korean gastric cancer screening program, algorithms and experience.
 Korean gastric cancer screening program, algorithms and experience. Jun Haeng Lee, MD. Department of Medicine, Sungkyunkwan University School of Medicine, Seoul, Korea Today s topics Korean cancer screening
Korean gastric cancer screening program, algorithms and experience. Jun Haeng Lee, MD. Department of Medicine, Sungkyunkwan University School of Medicine, Seoul, Korea Today s topics Korean cancer screening
Assessment of symptomatic response as predictor of Helicobacter pylori status following eradication therapy in patients with ulcer
 618 University Department of Medicine and Therapeutics, Western Infirmary, Glasgow G11 6NT, UK K E L McColl A El-Nujumi L S Murray E M El-Omar A Dickson A W Kelman T E Hilditch Correspondence to: Professor
618 University Department of Medicine and Therapeutics, Western Infirmary, Glasgow G11 6NT, UK K E L McColl A El-Nujumi L S Murray E M El-Omar A Dickson A W Kelman T E Hilditch Correspondence to: Professor
UK National Screening Committee. Screening for Stomach Cancer. 12 February 2016
 14/397 UK National Screening Committee Screening for Stomach Cancer 12 February 2016 Aim 1. To ask the UK National Screening Committee to make a recommendation, based upon the evidence presented in this
14/397 UK National Screening Committee Screening for Stomach Cancer 12 February 2016 Aim 1. To ask the UK National Screening Committee to make a recommendation, based upon the evidence presented in this
Quality Indicators - Anatomic Pathology- HSC/STC Jul-Sep 2 nd Qtr. Apr-Jun 1 st Qtr
 Eastern Health Volume 86 Page 001 CIHRT Exhibit P-3595 Page 1 INDICATOR Financial Overtime Hours / FTE Workload Increase - FTE equivalent Workload Quality Indicators - Anatomic Pathology- HSC/STC TOTAL
Eastern Health Volume 86 Page 001 CIHRT Exhibit P-3595 Page 1 INDICATOR Financial Overtime Hours / FTE Workload Increase - FTE equivalent Workload Quality Indicators - Anatomic Pathology- HSC/STC TOTAL
6/25/ % 20% 50% 19% Functional Dyspepsia Peptic Ulcer GERD Cancer Other
 Peptic Ulcer Disease and Dyspepsia John M. Inadomi, MD Professor of Medicine UCSF Chief, Clinical Gastroenterology San Francisco General Hospital Case History 49 y/o woman complains of several months of
Peptic Ulcer Disease and Dyspepsia John M. Inadomi, MD Professor of Medicine UCSF Chief, Clinical Gastroenterology San Francisco General Hospital Case History 49 y/o woman complains of several months of
KEYWORDS Dyspepsia, Acid Peptic Disease, Helicobacter Pylori, Urease, Giemsa, Peptic Ulcer, Non-Ulcer Dyspepsia.
 INCIDENCE OF HELICOBACTER PYLORI WITH ACID PEPTIC DISEASE AND MALIGNANT CONDITIONS OF UPPER GASTROINTESTINAL TRACT IN A TERTIARY CENTRE - A PROSPECTIVE STUDY Karunamoorthy Rajachidambaram 1, Dinkaran Kaarthesan
INCIDENCE OF HELICOBACTER PYLORI WITH ACID PEPTIC DISEASE AND MALIGNANT CONDITIONS OF UPPER GASTROINTESTINAL TRACT IN A TERTIARY CENTRE - A PROSPECTIVE STUDY Karunamoorthy Rajachidambaram 1, Dinkaran Kaarthesan
COMPARISON OF ONCE-A-DAY VERSUS TWICE-A-DAY CLARITHROMYCIN IN TRIPLE THERAPY FOR HELICOBACTER PYLORI ERADICATION
 Phil J Gastroenterol 2006; 2: 25-29 COMPARISON OF ONCE-A-DAY VERSUS TWICE-A-DAY CLARITHROMYCIN IN TRIPLE THERAPY FOR HELICOBACTER PYLORI ERADICATION Marianne P Collado, Ma Fatima P Calida, Peter P Sy,
Phil J Gastroenterol 2006; 2: 25-29 COMPARISON OF ONCE-A-DAY VERSUS TWICE-A-DAY CLARITHROMYCIN IN TRIPLE THERAPY FOR HELICOBACTER PYLORI ERADICATION Marianne P Collado, Ma Fatima P Calida, Peter P Sy,
Received: 11 Feb. 2013; Accepted: 7 Jun. 2013
 ORIGINAL REPORT Inter-Observer Reproducibility before and after Web-Based Education in the Gleason Grading of the Prostate Adenocarcinoma among the Iranian Pathologists Alireza Abdollahi 1*, Sara Sheikhbahaei
ORIGINAL REPORT Inter-Observer Reproducibility before and after Web-Based Education in the Gleason Grading of the Prostate Adenocarcinoma among the Iranian Pathologists Alireza Abdollahi 1*, Sara Sheikhbahaei
What is the status of Sequential Therapy Versus Standard Triple- Drug Therapy in peptic ulcer disease in eradicating H pylori?
 What is the status of Sequential Therapy Versus Standard Triple- Drug Therapy in peptic ulcer disease in eradicating H pylori? Sequential Therapy Versus Standard Triple- Drug Therapy for Helicobacter pylori
What is the status of Sequential Therapy Versus Standard Triple- Drug Therapy in peptic ulcer disease in eradicating H pylori? Sequential Therapy Versus Standard Triple- Drug Therapy for Helicobacter pylori
Management of dyspepsia and of Helicobacter pylori infection
 Management of dyspepsia and of Helicobacter pylori infection The University of Nottingham John Atherton Wolfson Digestive Diseases Centre University of Nottingham, UK Community management of dyspepsia
Management of dyspepsia and of Helicobacter pylori infection The University of Nottingham John Atherton Wolfson Digestive Diseases Centre University of Nottingham, UK Community management of dyspepsia
Helicobacter pylori Eradication Therapy Success Regarding Different Treatment Period Based on Clarithromycin or Metronidazole Triple-Therapy Regimens
 Helicobacter ISSN 1523-5378 Filipec Blackwell Oxford, HEL 1083-4389 1523-5378 Journal XXX Original H. 2008 pylori Kanizaj compilation The UK Eradication Publishing Article Authors et al. Ltd 2008 Therapy
Helicobacter ISSN 1523-5378 Filipec Blackwell Oxford, HEL 1083-4389 1523-5378 Journal XXX Original H. 2008 pylori Kanizaj compilation The UK Eradication Publishing Article Authors et al. Ltd 2008 Therapy
The role of antisecretory drugs in the treatment of Helicobacter pylori infection
 Aliment Pharmacol Ther 1997; 11 (Suppl. 1): 21 25. The role of antisecretory drugs in the treatment of Helicobacter pylori infection W. L. PETERSON Department of Internal Medicine, University of Texas
Aliment Pharmacol Ther 1997; 11 (Suppl. 1): 21 25. The role of antisecretory drugs in the treatment of Helicobacter pylori infection W. L. PETERSON Department of Internal Medicine, University of Texas
Microbiological and serological diagnostic tests for Helicobacter pylori: an overview
 Microbiological and serological diagnostic tests for Helicobacter pylori: an overview Youri Glupczynski Service de Biologie Clinique, Cliniques Universitaires UCL de Mont-Godinne, Yvoir, Belgium Different
Microbiological and serological diagnostic tests for Helicobacter pylori: an overview Youri Glupczynski Service de Biologie Clinique, Cliniques Universitaires UCL de Mont-Godinne, Yvoir, Belgium Different
Catalog # 0W009 0W010 Test Cassettes, murine monoclonal antibody to human IgG (Test Line) and rabbit polyclonal antibody (Control Line) Reagents
 For in vitro diagnostic use. CLIA Complexity: MODERATE INTENDED USE The QuickVue H. pylori Test is a lateral-flow immunoassay intended for the rapid, qualitative detection of IgG antibodies specific to
For in vitro diagnostic use. CLIA Complexity: MODERATE INTENDED USE The QuickVue H. pylori Test is a lateral-flow immunoassay intended for the rapid, qualitative detection of IgG antibodies specific to
Clinical Policy: Helicobacter Pylori Serology Testing Reference Number: CP.MP.153
 Clinical Policy: Reference Number: CP.MP.153 Effective Date: 12/17 Last Review Date: 12/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory and
Clinical Policy: Reference Number: CP.MP.153 Effective Date: 12/17 Last Review Date: 12/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory and
Campylobacter pylori in non-specific abdominal pain in childhood
 Gut, 1989, 3, 912-916 Serum pepsinogen I and IgG antibody to Campylobacter pylori in non-specific abdominal pain in childhood GIUSEPPINA ODERDA, D VAIRA, J HOLTON, J F DOWSETT, AND NICOLETTA ANSALDI From
Gut, 1989, 3, 912-916 Serum pepsinogen I and IgG antibody to Campylobacter pylori in non-specific abdominal pain in childhood GIUSEPPINA ODERDA, D VAIRA, J HOLTON, J F DOWSETT, AND NICOLETTA ANSALDI From
A PLACEBO CONTROLLED TRIAL OF BISMUTH SALICYLATE IN HELICOBACTER PYLORI ASSOCIATED GASTRITIS
 A PLACEBO CONTROLLED TRIAL OF BISMUTH SALICYLATE IN HELICOBACTER PYLORI ASSOCIATED GASTRITIS Pages with reference to book, From 154 To 156 Javed Iqbal Kazi, Naeem Aon Jafarey, Syed Mahmood Alam ( Department
A PLACEBO CONTROLLED TRIAL OF BISMUTH SALICYLATE IN HELICOBACTER PYLORI ASSOCIATED GASTRITIS Pages with reference to book, From 154 To 156 Javed Iqbal Kazi, Naeem Aon Jafarey, Syed Mahmood Alam ( Department
Summary HTA. Efficacy and cost-effectiveness of the
 Summary HTA HTA-Report Summary Efficacy and cost-effectiveness of the 13 C-urea breath test as the primary diagnostic investigation for the detection of Helicobacter pylori infection compared to invasive
Summary HTA HTA-Report Summary Efficacy and cost-effectiveness of the 13 C-urea breath test as the primary diagnostic investigation for the detection of Helicobacter pylori infection compared to invasive
