Value of Serology as a Noninvasive Method for Evaluating the Efficacy of Treatment of Helicobacter pylori Infection
|
|
|
- Jessica Chase
- 5 years ago
- Views:
Transcription
1 1038 Value of Serology as a Noninvasive Method for Evaluating the Efficacy of Treatment of Helicobacter pylori Infection Guillermo I. Pérez-Pérez, Alan F. Cutler, and Martin J. Blaser From the Division of Infectious Diseases, Vanderbilt University School of Medicine, and the Infectious Disease Section, Department of Veterans Affairs Medical Center, Nashville, Tennessee, and the Section of Gastroenterology, Sinai Hospital, Detroit, Michigan The systemic humoral response to Helicobacter pylori was studied in 86 infected adult patients before antimicrobial therapy and at intervals following therapy. Endoscopy with collection of biopsy specimens was performed immediately before treatment; a 13 C-labeled urea breath test was performed, and blood specimens were collected before treatment and at 1, 3, 6, 9, and 12 months after treatment. Serum samples from three patient groups (eradication success [n Å 50], eradication failure [n Å 16], and no treatment [n Å 20]) were assayed for IgA and IgG antibodies to H. pylori by enzyme-linked immunosorbent assay. Levels of antibody to H. pylori before treatment were similar in all three groups. As expected, the no treatment and eradication failure groups had no significant changes in antibody levels during the study period. In contrast, for the eradication success group, the specific IgA and IgG antibody levels decreased progressively and significantly. We conclude that serology is a potentially useful way to monitor the success of treatment of H. pylori infection without using invasive or more expensive methods. clinically useful for other chronic infectious diseases, including syphilis, brucellosis, and coccidioidomycosis, although the de- cline in antibody level is slow (often requiring 6 12 months for optimal accuracy). The aim of this study was to use a highly sensitive and specific serological assay to monitor the antibody response before and after antimicrobial therapy for eradication of H. pylori infection. We sought to determine the accuracy of serology for determining bacterial eradication in relation to time after therapy. It now is well established that gastric colonization with Helicobacter pylori induces chronic gastritis, which may lead to peptic ulcer disease [1 3]. Once acquired, H. pylori infection persists in the gastric mucosa for years or decades in the absence of treatment [4, 5], thus eliciting a stable systemic antibody response [6]. Eradication of H. pylori infection results in reduction of gastritis and in diminished recurrence of duodenal ulcers [7]. Until now, the monitoring of treatment usually involved either repeated gastroscopy or the use of urea breath tests (UBTs) [8]. Endoscopy is invasive and relatively expensive [9]. Although the UBT is noninvasive, less expensive, and quite accurate, it requires special equipment, trained personnel, and relative expense (especially for developing countries [8]); may involve radioisotopes; and is still relatively unavailable in most geographic areas. Moreover, for both techniques, sensi- tivity for detecting treatment failure is not 100% [10]. Serology has proven to be an accurate noninvasive test for detecting H. pylori infection [4, 11, 12]. Several studies have suggested that a decline in antibody levels might be useful for evaluation of eradication of H. pylori infection [13 15]. Previous studies [16 18] showing declining IgG antibody levels after confirmed eradication did not include an untreated control group. Serological assessment of treatment efficacy is This article is part of a series of papers presented at the Second International Workshop on Helicobacter pylori Infections in the Developing World held in Lima, Peru, in January Financial support: This study was supported in part by the Iris and Homer Akers Fellowship in Infectious Diseases. Reprints or correspondence: Dr. Guillermo I. Pérez-Pérez, Vanderbilt University School of Medicine, Division of Infectious Diseases, A-3310 Medical Center North, st Avenue South, Nashville, Tennessee Clinical Infectious Diseases 1997;25: This article is in the public domain. Materials and Methods Patients and specimens. Eighty-six patients with H. pylori infection who received care in the Division of Gastroenterology at Henry Ford Hospital (Detroit) between 1990 and 1992 were included in this study. Of these patients, 66 (76.7%) had partici- pated in an investigation evaluating specific treatments for the eradication of H. pylori infection. For 63 of the 66 patients, the treatment regimen was based on triple-antimicrobial therapy (2- week course of bismuth subsalicylate [525 mg q.i.d.], tetracy- cline [500 mg q.i.d.], and metronidazole [500 mg t.i.d.]), and the three other patients were treated with only colloidal bismuth subcitrate (Denol, Gist-Brocades, West Byfleet, England). There were 20 patients involved in other protocols who were not treated for their H. pylori infection. For all patients, baseline studies included endoscopy with collection of two antral biopsy specimens for histological examination with Warthin-Starry silver stain and one biopsy specimen for Campylobacter-like organism (CLO) (rapid urease) testing as previously reported [19]. Serum samples for examination of antibodies to H. pylori and for the 13 C-labeled UBT (Cambridge Isotope Laboratories, Boston) were obtained before treatment, 1 month after the
2 CID 1997;25 (November) Serological Evaluation of Treatment of H. pylori Infection 1039 termination of treatment, and then at 3-month intervals. One, Table 1. Sensitivity of diagnostic methods in the pretreatment eval- two, three, or four follow-up specimens were obtained from uation of 86 Helicobacter pylori infected patients. 100%, 84%, 50%, and 27% of the patients, respectively. No. of No. of Study definitions. For all patients, confirmation of H. pylori patients positive Sensitivity infection by the 13 C-labeled UBT was required for entry into Test studied tests (%) the study; the criteria used were previously reported [19]. In addition, H. pylori infection was diagnosed by positive War- CLO (rapid urease) test* Histological staining thin-starry silver staining of gastric mucosa specimens ob- ELISA for IgA tained by antral biopsy. In cases in which Warthin-Starry silver ELISA for IgG staining was negative, diagnosis of H. pylori infection was established on the basis of the CLO test. Status of H. pylori NOTE. All 86 patients had positive 13 C-labeled urea breath tests, as re- quired for entry into the study. CLO Å Campylobacter-like organism. infection during follow-up visits was established by the 13 C- * A positive test was defined as a change of color within the first 24 hours. labeled UBT. Eradication of H. pylori infection was defined A positive test was defined as the demonstration of an organism with the as a negative 13 C-labeled UBT 1 month after cessation of ther- typical appearance of H. pylori. The patient was considered seropositive when an optical density ratio of apy, as is now considered to be standard [20]. 13 C-labeled 1.0 was obtained. UBTs repeated 3 months after cessation of therapy, which are more accurate and eliminate false-negative results due to suppression but not elimination of infection [21], verified no the three patients treated solely with colloidal bismuth subcitrate. further changes in status of H. pylori infection. None of those patients whose infection was eradicated relapsed, and none of The enrollment characteristics of the three patient groups those patients who were not treated or for whom initial treatment (eradication success, eradication failure, and no treatment) are failed to eradicate the infection had spontaneous clearance summarized in table 2. The control (no treatment) group had of the organism. a higher average age and proportion of other pathologies associ- Serological analysis. Serological assays for IgA and IgG ated with the upper gastrointestinal tract (55% vs. 1.5%, respectively) antibodies to H. pylori were performed by ELISAs according than did the treatment groups. These patients repreantibodies to previously reported methods, and values were expressed as sented less severe clinical cases. This clear disproportion optical density ratios (ODRs) [4, 5, 11, 12]. The antigen was between the groups was due to specific recommendations of a pool of five H. pylori strains, and the sensitivity and specific- the Human Subjects Committee who approved the study in ity of testing with this antigen were ú90% [11, 12]. All followup The proportions of males and females were similar be- serum specimens from one patient were tested in the same tween the groups. None of the enrollment values could be used run with the original serum sample to evaluate the percentage to predict the results of treatment. change from baseline. By using the 13 C-labeled UBT to establish the presence of Statistical analysis. The results were expressed as means H. pylori infection, none of the treated patients in whom { SEM, and statistical analyses included comparison of means H. pylori infection was eradicated relapsed during the observation between groups by the t test. x 2 and Fisher s exact tests were period. Conversely, and as expected, in none of the patients also performed for two-by-two tables. A P value of.05 was who were not treated or for whom treatment failed was there considered statistically significant. spontaneous elimination of H. pylori infection. Since antibody kinetics were very similar between persons who were never treated or for whom treatment failed, we combined Results these two groups in all subsequent analyses. Levels of IgG and IgA antibodies specific to H. pylori decreased significantly We determined the sensitivity of each of the methods used only in the eradication success group (figure 1). In that for the diagnosis of H. pylori infection (table 1) and compared group, by the third month of follow-up, the mean levels of these sensitivities with that of the 13 C-labeled UBT, which was specific antibodies decreased ú30% from the original values. used for study entry. Although the 13 C-labeled UBT and CLO A persistent drop in IgG and IgA antibody values was observed test are based on the same enzymatic principle (urease activity), until the end of the study at 12 months of follow-up. In the the sensitivity of the CLO test was only 82.7%. As expected, eradication success group, the mean reduction in IgG antibody the sensitivity of the ELISAs was similar to that of histological ODR was greater than 40% of the pretreatment values by 6 examination. months. H. pylori infection was found to be eradicated in 50 (75.8%) Although the mean changes in antibody levels between of the 66 treated patients at 1 month after cessation of therapy. pretreatment and follow-up samples are of interest, the observations Sixteen of the treated patients remained infected with H. pylori; in figure 1 represent changes for all groups studied. these patients were included in the eradication failure group. A more important analysis for clinical purposes is to compare As expected, H. pylori infection was not eliminated in any of the change in antibody level for an individual patient in rela-
3 1040 Pérez-Pérez, Cutler, and Blaser CID 1997;25 (November) Table 2. Enrollment characteristics of Helicobacter pylori infected adult patients. Treatment group Characteristic Eradication success Eradication failure No treatment group No. of patients Mean age { SEM in y 58.2 { { { 2.9 Male patients (%) No. of patients in diagnostic category Duodenal ulcer Gastric ulcer NUD Other* Mean IgA ODR at baseline { SEM 2.48 { { { 0.3 Mean IgG ODR at baseline { SEM 3.85 { { { 0.7 Mean 13 C-labeled UBT value (%) at baseline { SEM 24.7 { { { 5.5 NOTE. NUD Å nonulcer dyspepsia; ODR Å optical density ratio [11]; UBT Å urea breath test. * Including 10 patients with NUD associated with nonsteroidal antiinflammatory drug use and one patient with atrophic gastritis. The increase in the mole fraction of tracer 13 CO 2 at 60 minutes compared with that at baseline, with ú6% considered positive. tion to the pretreatment serum sample. A summary of this analysisisshownintable3,inwhichthecriterionofchanging from seropositive to seronegative was used to define serological evidence of treatment success. Although seroreversion to IgG antibodies was not found in any sample from a patient for whom treatment failed, the sensitivity for ascer- taining treatment success was low (only 25.6% at 6 months and 47% at 12 months). We next sought to determine whether comparison between pretreatment and posttreatment antibody levels can more quickly and accurately reflect treatment success. As working guidelines, we defined seroreversion in a patient as a decrease Figure 1. Effect of specific antimicrobial therapy on the mean level { SEM of IgA antibody (A) and IgG antibody (B) tohelicobacter pylori antigens in 50 patients for whom therapy was successful ( ), 16 patients for whom therapy failed ( ), and 20 controls who received no specific treatment ( ). * Å P value of õ.005; /ÅP value of õ.05.
4 CID 1997;25 (November) Serological Evaluation of Treatment of H. pylori Infection 1041 Table 3. Seroreversion rates among patients after specific treatment observed as early as 3 months after cessation of treatment for Helicobacter pylori infection. (table 4). Percent of patients seroreverting* No. of patients studied at Discussion specified time Eradication Eradication For optimal posttreatment follow-up of H. pylori infections, failure success accurate, rapid, and inexpensive assays are needed. Because Mo. of Eradication Eradication techniques based on either endoscopy or UBTs have important follow-up failure success IgA IgG IgA IgG limitations [10, 22], we addressed whether serological techniques may be useful. Our study is limited in that we were not able to obtain specimens from each patient at each follow-up, and thus, comparisons of the different times involved different patients. We are not aware of any systematic selection that would bias the results obtained. To address the issue of bias * Seroreversion was defined as an optical density ratio of õ1.0. from follow-up differences, we divided the patients into those Includes patients in eradication failure and no treatment groups. who had peptic ulcer disease (n Å 58) and those with other milder conditions (n Å 28). There was no substantial difference in the mean total number of visits for each group (2.7 { 0.12 in IgG and IgA antibody levels from the pretreatment serum vs. 2.4 { 0.22, respectively; P Å.22). However, because the value of ú30% or ú50%. We also used a decrease of 50% therapy used was not as effective as treatments presently being in either IgA or IgG antibody value from the pretreatment advocated [23], we were able to follow a reasonable number serum sample as a criterion of seroreversion. In the eradication of patients for whom therapy failed as well as a control group failure group, the seroreversions that occurred were present of patients who did not receive any treatment. mainly for IgA antibodies and during the early months after All studies of follow-up after treatment are limited by the therapy (table 4). Three patients with positive 13 C-labeled lack of an absolute gold standard because both biopsy-based UBTs had a decrease of ú30% in IgG antibody values 6 and breath test based assays may falsely indicate treatment months after the cessation of therapy. Unfortunately, there success [10]. Despite these limitations, the results of our studies was not another follow-up sample from these patients, and are consistent with previous observations that serological techniques whether this drop in IgG antibody levels was an artifact or can be used to determine treatment efficacy [14 16]. the 13 C-labeled UBT results were false-positive due to the One limitation of the serological techniques is that they have sensitivity of the test could not be confirmed. In contrast, not been subjected to standardization involving different labo- among the patients for whom treatment was successful, rates ratories. of seroreversion to both IgG and IgA antibodies increased Biopsy-based tests examine samples from only a small progressively during the entire study period. Statistically sig- (õ1%) portion of the stomach, whereas the noninvasive UBT nificant differences in seroreversion rates between patients in and serology are global (in effect testing samples from the the eradication success and eradication failure groups were entire stomach). This phenomenon may help explain why the Table 4. Seroreversion rates among patients after specific treatment for Helicobacter pylori infection. Percent of patients seroreverting* No. of patients IgG antibody IgA antibody studied at specified time ú30% ú50% ú30% ú50% Mo. of follow-up F S F S F S F S F S NOTE. F Å eradication failure group includes patients who were not treated; S Å eradication success group. * Criteria for seroreversion are a ú30% or ú50% decrease in antibody level from the pretreatment value at the specific time during follow-up. P õ.05 compared with rate among persons in eradication failure group.
5 1042 Pérez-Pérez, Cutler, and Blaser CID 1997;25 (November) UBT was more sensitive than the CLO test in the initial diagno- obtain follow-up specimens as early as 3 months after the end sis of H. pylori infection. The presence of other urease-positive of therapy. In contrast, the criterion of using a ú30% decrease organisms (e.g., in the mouth) has little impact on UBT results in IgG antibody level is more sensitive for detecting treatment when tests are performed according to now standard criteria. success, and if used at an appropriate time (e.g., 9 months), If appropriate diagnostic criteria can be selected, serology and it is highly specific. For persons in whom there is no particular UBT have the double advantage of being both noninvasive and urgency to detect treatment efficacy, follow-up at 9 12 months globally based [24, 25]. A 13 C-labeled UBT recently has been might be ideal. On occasion, patients for whom therapy ultimately licensed by the U.S. Food and Drug Administration, and such fails have a transient and small decrease in antibody tests should become widely available in the near future. levels [16, 18, 26], presumably due to suppression of the infection. Because levels of antibody specific to H. pylori are often Examining specimens obtained beyond 6 months after the very high before treatment, the definition used for seroreversion cessation of therapy should overcome this problem. is critical to its clinical utility. For example, as shown in table In conclusion, we confirm that serology can be used for 3, the standard criterion of changing from seropositive to sero- the individual patient to noninvasively determine efficacy of negative is relatively insensitive and slow to develop for detecting therapy for H. pylori infection. Further investigations should treatment success. In contrast, the data in figure 1 sug- help better define optimal diagnostic criteria and timing of gest that using a comparison between pretreatment and follow-up examinations. posttreatment antibody levels in a particular patient will be more useful. The results of using several different definitions of seroreversion that are shown in table 4 confirm that this is a superior approach to that shown in table 3. However, even Acknowledgments so, this comparative method has several limitations. The authors thank T. T. Schubert and A. B. Schubert for their First, the comparative method requires that the pretreatment contributions during the study. serum sample be saved and then assayed simultaneously with the posttreatment serum sample. Although these are logistic handicaps, they can be easily overcome in a dedicated labora- References tory in which pretreatment samples are stored and are easily 1. Warren JR, Marshall B. Unidentified curved bacilli on gastric epithelium accessible. The use of appropriate definitions can minimize the in active chronic gastritis. Lancet 1983;1:1273. variability inherent in freeze-thawing or long-term storage of 2. Blaser MJ. Helicobacter pylori and the pathogenesis of gastroduodenal specimens. Examination of the paired specimens together limits inflammation. J Infect Dis 1990;161: day-to-day and plate-to-plate variation, which could affect inology 3. Graham DY. Campylobacter pylori and peptic ulcer disease. Gastroenter- 1989;96: terpretation of results. Second, the assay for IgA antibody adds 4. Pérez-Pérez GI, Dworkin BM, Chodos JE, Blaser MJ. Campylobacter little to the assay for IgG antibody and, in our hands, is both pylori antibodies in humans. Ann Intern Med 1988;109:11 7. less specific and less sensitive than the assay for IgG antibody. 5. Parsonnet J, Blaser MJ, Pérez-Pérez GI, Hargrett-Bean N, Tauxe RV. Our results suggest that this assay adds no clinical utility and Symptoms and risk factors of Helicobacter pylori infection in a cohort should not be done. of epidemiologists. Gastroenterology 1992;102: Langenberg W, Rauws EAJ, Houthoff HJ, et al. Follow-up study of indi- Third, the loss of antibodies is a slow process, thus reflecting viduals with untreated Campylobacter pylori associated gastritis and the intensive impact of H. pylori infection on the responof noninfected persons with non-ulcer dyspepsia. J Infect Dis 1988; siveness human B cells to H. pylori antigens [24]. Using a 157: relatively stringent definition (ú50% decrease in IgG antibody 7. Graham DY, Lew GM, Klein PD, et al. Effect of treatment of Helicobacter level) at 6 months after therapy was not very sensitive for pylori infection on the long-term recurrence of gastric or duodenal ulcer. A randomized, controlled study. Ann Intern Med 1992;116: detecting treatment success in that specimens from only 50% 8. Graham DY, Klein PD, Evans DJ, et al. Campylobacter pylori detected of the successfully treated persons met this criterion. Even after non-invasively by the 13 C-urea breath test. Lancet 1987;1: months of follow-up, not all successfully treated persons 9. Kolts BE, Joseph B, Achem SR, Bianchi T, Monteiro C. Helicobacter had a ú50% decrease in specific IgG antibody levels. This pylori detection: a quality and cost analysis. Am J Gastroenterol 1993; phenomenon reflects both the delay in loss of antibodies and 88: Peterson WL, Graham DY, Marshall B, et al. Clarithromycin as monotherthe extensive biological diversity in the H. pylori infected apy for eradication of Helicobacter pylori: a randomized, double-blind population. trial. Am J Gastroenterol 1993;88: Our results indicate that using a ú50% decrease in IgG 11. Dooley CP, Fitzgibbons PL, Cohen H, Appleman MD, Pérez-Pérez GI, antibody levels as the criterion is a highly specific method Blaser MJ. Prevalence of Helicobacter pylori infection and histologic of determining treatment success. None of the 74 specimens gastritis in asymptomatic persons. N Engl J Med 1989;321: Drumm B, Pérez-Pérez GI, Blaser MJ, Sherman P. Intrafamilial clustering obtained during follow-up from patients in whom H. pylori of Helicobacter pylori infection. N Engl J Med 1990;322: infection was not eradicated met this criterion. As discussed 13. Morris A, Ali G, Nicholson G, Pérez-Pérez GI, Blaser MJ. Long-term previously, its sensitivity is suboptimal. However, use of a follow-up of voluntary ingestion of Helicobacter pylori. Ann Intern method with such high specificity could allow clinicians to Med 1991;114:662 3.
6 CID 1997;25 (November) Serological Evaluation of Treatment of H. pylori Infection Oderda G, Vaira D, Ainley C, et al. Eighteen month follow up of Helico- 21. Logan RPH, Gummett PA, Misiewicz JJ, Karim QN, Walker MM, Baron bacter pylori-positive children treated with amoxycillin and tinidazole. JH. One week eradication regimen for Helicobacter pylori. Lancet 1991; Gut 1992;33: : Truesdale R, Chamberlain C, Martin D, Maydonovitch C, Peura D. Long- 22. Cutler AF, Havstad S, Ma CK, Blaser MJ, Pérez-Pérez GI, Schubert TT. term follow-up and antibody response to treatment of patients Helico- Accuracy of invasive and noninvasive tests to diagnose Helicobacter bacter pylori. Gastroenterology 1990;98:A140. pylori infection. Gastroenterology 1995;109: Kosunen TU, Seppala K, Sarna S, Sipponen P. Diagnostic value of decreas- 23. de Boer WA, Driessen WMM, Jansz AR, Tytgat GNJ. Effect of acid ing IgG, IgA, and IgM antibody titres after eradication of Helicobacter suppression on efficacy of treatment for Helicobacter pylori infection. pylori. Lancet 1992;339: Lancet 1995;345: Wang WM, Chen CY, Jan CM, et al. Long-term follow-up and serological 24. Jaskiewicz K, Louw JA, Marks IN. Local cellular and immune response study after triple therapy of Helicobacter pylori-associated duodenal by antral mucosa in patients undergoing treatment for eradication of ulcer. Am J Gastroenterol 1994;89: Helicobacter pylori. Dig Dis Sc 1993;38: Glupczynski Y, Burette A, Goossens H, Deprez C, Butzler JP. Effect of 25. Nensey YM, Schubert TT, Schubert AB, Ma CK. Simplified 13 C urea antimicrobial therapy on the specific serological response to Helicobacter breath test as a method of detection of Helicobacter pylori in both pylori infection. Eur J Clin Microbiol Infect Dis 1992;11: treated and untreated patients. Gastroenterology 1991;100:A Cutler A, Schubert A, Schubert T. Role of Helicobacter pylori serology in evaluating treatment success. Dig Dis Sci 1993;38: Shimoyama T, Fukuda Y, Fukuda S, Munakata A, Yoshida Y, Shimoyama 20. Atherton JC, Spiller RC. The urea breath test for Helicobacter pylori. Gut T. Validity of various diagnostic tests to evaluate cure of Helicobacter 1994;35: pylori infection. J Gastroenterol 1996;31:171 4.
Urea Breath Test for Diagnosis of Helicobactor pylori. Original Policy Date 12:2013
 MP 2.04.04 Urea Breath Test for Diagnosis of Helicobactor pylori Medical Policy Section Medicine Issue 12:2013 Original Policy Date 12:2013 Last Review Status/Date 12:2013 Return to Medical Policy Index
MP 2.04.04 Urea Breath Test for Diagnosis of Helicobactor pylori Medical Policy Section Medicine Issue 12:2013 Original Policy Date 12:2013 Last Review Status/Date 12:2013 Return to Medical Policy Index
H. pylori IgM. Cat # H. pylori IgM ELISA. ELISA: Enzyme Linked Immunosorbent Assay. ELISA - Indirect; Antigen Coated Plate
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com H. pylori
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com H. pylori
H.Pylori IgG
 DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
H.Pylori IgG Cat # 1503Z
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
H.pylori IgA Cat #
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
H. pylori Antigen ELISA Kit
 H. pylori Antigen ELISA Kit Catalog Number KA3142 96 assays Version: 04 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
H. pylori Antigen ELISA Kit Catalog Number KA3142 96 assays Version: 04 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
H.Pylori IgM Cat # 1504Z
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
See external label 2 C-8 C Σ=96 tests Cat # 1505Z. MICROWELL ELISA H.Pylori IgA Cat # 1505Z
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Research Article Performance of Routine Helicobacter pylori Invasive Tests in Patients with Dyspepsia
 Gastroenterology Research and Practice Volume 2013, Article ID 184806, 5 pages http://dx.doi.org/10.1155/2013/184806 Research Article Performance of Routine Helicobacter pylori Invasive Tests in Patients
Gastroenterology Research and Practice Volume 2013, Article ID 184806, 5 pages http://dx.doi.org/10.1155/2013/184806 Research Article Performance of Routine Helicobacter pylori Invasive Tests in Patients
H. pylori IgM CLIA kit
 H. pylori IgM CLIA kit Cat. No.:DEEL0251 Pkg.Size:96 tests Intended use Helicobacter pylori IgM Chemiluminescence ELISA is intended for use in evaluating the serologic status to H. pylori infection in
H. pylori IgM CLIA kit Cat. No.:DEEL0251 Pkg.Size:96 tests Intended use Helicobacter pylori IgM Chemiluminescence ELISA is intended for use in evaluating the serologic status to H. pylori infection in
Comparative study of invasive methods for diagnosis of Helicobacter pylori in humans
 ISSN: 2319-7706 Volume 2 Number 7 (2013) pp. 63-68 http://www.ijcmas.com Original Research Article Comparative study of invasive methods for diagnosis of Helicobacter pylori in humans V.Subbukesavaraja
ISSN: 2319-7706 Volume 2 Number 7 (2013) pp. 63-68 http://www.ijcmas.com Original Research Article Comparative study of invasive methods for diagnosis of Helicobacter pylori in humans V.Subbukesavaraja
COMPARISON OF ONCE-A-DAY VERSUS TWICE-A-DAY CLARITHROMYCIN IN TRIPLE THERAPY FOR HELICOBACTER PYLORI ERADICATION
 Phil J Gastroenterol 2006; 2: 25-29 COMPARISON OF ONCE-A-DAY VERSUS TWICE-A-DAY CLARITHROMYCIN IN TRIPLE THERAPY FOR HELICOBACTER PYLORI ERADICATION Marianne P Collado, Ma Fatima P Calida, Peter P Sy,
Phil J Gastroenterol 2006; 2: 25-29 COMPARISON OF ONCE-A-DAY VERSUS TWICE-A-DAY CLARITHROMYCIN IN TRIPLE THERAPY FOR HELICOBACTER PYLORI ERADICATION Marianne P Collado, Ma Fatima P Calida, Peter P Sy,
What is the status of Sequential Therapy Versus Standard Triple- Drug Therapy in peptic ulcer disease in eradicating H pylori?
 What is the status of Sequential Therapy Versus Standard Triple- Drug Therapy in peptic ulcer disease in eradicating H pylori? Sequential Therapy Versus Standard Triple- Drug Therapy for Helicobacter pylori
What is the status of Sequential Therapy Versus Standard Triple- Drug Therapy in peptic ulcer disease in eradicating H pylori? Sequential Therapy Versus Standard Triple- Drug Therapy for Helicobacter pylori
- Helicobacter - THE EASE AND DIFFICULTY OF A NEW DISCOVERY. Robin Warren
 - Helicobacter - THE EASE AND DIFFICULTY OF A NEW DISCOVERY Robin Warren EARLY DAYS First reports 100 years ago considered spirochaetes 1940 Freedburg saw curved organisms in the stomach 1954 Palmer: Freedburg
- Helicobacter - THE EASE AND DIFFICULTY OF A NEW DISCOVERY Robin Warren EARLY DAYS First reports 100 years ago considered spirochaetes 1940 Freedburg saw curved organisms in the stomach 1954 Palmer: Freedburg
Comparison of the Accuracy of Two Commercial Rapid Urase Tests, CLOtest and Pronto Dry, in Detecting Helicobacter pylori Infection ABSTRACT
 Original Article Rojborwonwitaya J, Vijitjunyakul N THAI J GASTROENTEROL 2005 Vol. 6 No. 2 May - Aug. 2005 55 Comparison of the Accuracy of Two Commercial Rapid Urase Tests, CLOtest and Pronto Dry, in
Original Article Rojborwonwitaya J, Vijitjunyakul N THAI J GASTROENTEROL 2005 Vol. 6 No. 2 May - Aug. 2005 55 Comparison of the Accuracy of Two Commercial Rapid Urase Tests, CLOtest and Pronto Dry, in
Clinical significance of Helicobacter infection in children
 Clinical significance of Helicobacter infection in children Marion Rowland and Brendan Drumm Department of Paediatrics, University College Dublin, Children's Research Centre, Our Lady's Hospital for Sick
Clinical significance of Helicobacter infection in children Marion Rowland and Brendan Drumm Department of Paediatrics, University College Dublin, Children's Research Centre, Our Lady's Hospital for Sick
Correlation Between Endoscopic and Histological Findings in Different Gastroduodenal Lesion and its Association with Helicobacter Pylori
 ORIGINAL ARTICLE Correlation Between Endoscopic and Histological Findings in Different Gastroduodenal Lesion and its Association with Helicobacter Pylori *A. Sultana 1, SM Badruddoza 2, F Rahman 3 1 Dr.
ORIGINAL ARTICLE Correlation Between Endoscopic and Histological Findings in Different Gastroduodenal Lesion and its Association with Helicobacter Pylori *A. Sultana 1, SM Badruddoza 2, F Rahman 3 1 Dr.
Helicobacter 2008;13:1-6. Am J Gastroent 2007;102: Am J of Med 2004;117:31-35.
 An Update on Helicobacter pylori and Its Treatment Trenika Mitchell, PharmD, BCPS Clinical Assistant Professor University of Kentucky College of Pharmacy October 18, 2008 Objectives Review the epidemiology
An Update on Helicobacter pylori and Its Treatment Trenika Mitchell, PharmD, BCPS Clinical Assistant Professor University of Kentucky College of Pharmacy October 18, 2008 Objectives Review the epidemiology
Helicobacter Pylori Testing HELICOBACTER PYLORI TESTING HS-131. Policy Number: HS-131. Original Effective Date: 9/17/2009
 Easy Choice Health Plan, Inc. Harmony Health Plan of Illinois, Inc. Missouri Care, Inc. Ohana Health Plan, a plan offered by WellCare Health Insurance of Arizona, Inc. WellCare Health Insurance of Illinois,
Easy Choice Health Plan, Inc. Harmony Health Plan of Illinois, Inc. Missouri Care, Inc. Ohana Health Plan, a plan offered by WellCare Health Insurance of Arizona, Inc. WellCare Health Insurance of Illinois,
Helicobacter and gastritis
 1 Helicobacter and gastritis Dr. Hala Al Daghistani Helicobacter pylori is a spiral-shaped gram-negative rod. H. pylori is associated with antral gastritis, duodenal (peptic) ulcer disease, gastric ulcers,
1 Helicobacter and gastritis Dr. Hala Al Daghistani Helicobacter pylori is a spiral-shaped gram-negative rod. H. pylori is associated with antral gastritis, duodenal (peptic) ulcer disease, gastric ulcers,
PDF hosted at the Radboud Repository of the Radboud University Nijmegen
 PDF hosted at the Radboud Repository of the Radboud University Nijmegen The following full text is a publisher's version. For additional information about this publication click this link. http://hdl.handle.net/2066/48400
PDF hosted at the Radboud Repository of the Radboud University Nijmegen The following full text is a publisher's version. For additional information about this publication click this link. http://hdl.handle.net/2066/48400
The long-term management of patients with bleeding duodenal ulcers
 Aliment Pharmacol Ther (1997); 11: 505±510. The long-term management of patients with bleeding duodenal ulcers M. E. MCALINDON, J. S. W. TAYLOR & S. D. RYDER Department of Medicine, University Hospital,
Aliment Pharmacol Ther (1997); 11: 505±510. The long-term management of patients with bleeding duodenal ulcers M. E. MCALINDON, J. S. W. TAYLOR & S. D. RYDER Department of Medicine, University Hospital,
Helicobacter Connections. Barry Marshall
 Helicobacter Connections Barry Marshall The greatest obstacle to knowledge is not ignorance, it is the illusion of knowledge. Daniel Boorstein - Historian Peptic Ulcers Duodenal Ulcer (DU) Gastric Ulcer
Helicobacter Connections Barry Marshall The greatest obstacle to knowledge is not ignorance, it is the illusion of knowledge. Daniel Boorstein - Historian Peptic Ulcers Duodenal Ulcer (DU) Gastric Ulcer
Type of intervention Diagnosis. Economic study type Cost-effectiveness analysis.
 Noninvasive tests as a substitute for histology in the diagnosis of Helicobacter pylori infection Hahn M, Fennerty M B, Corless C L, Magaret N, Lieberman D A, Faigel D O Record Status This is a critical
Noninvasive tests as a substitute for histology in the diagnosis of Helicobacter pylori infection Hahn M, Fennerty M B, Corless C L, Magaret N, Lieberman D A, Faigel D O Record Status This is a critical
ACG Clinical Guideline: Treatment of Helicobacter pylori Infection
 ACG Clinical Guideline: Treatment of Helicobacter pylori Infection William D. Chey, MD, FACG 1, Grigorios I. Leontiadis, MD, PhD 2, Colin W. Howden, MD, FACG 3 and Steven F. Moss, MD, FACG 4 1 Division
ACG Clinical Guideline: Treatment of Helicobacter pylori Infection William D. Chey, MD, FACG 1, Grigorios I. Leontiadis, MD, PhD 2, Colin W. Howden, MD, FACG 3 and Steven F. Moss, MD, FACG 4 1 Division
Immunoglobulin G Antibody against Helicobacter pylori: Clinical Implications of Levels Found in Serum
 CLINICAL AND DIAGNOSTIC LABORATORY IMMUNOLOGY, Sept. 2002, p. 1044 1048 Vol. 9, No. 5 1071-412X/02/$04.00 0 DOI: 10.1128/CDLI.9.5.1044 1048.2002 Copyright 2002, American Society for Microbiology. All Rights
CLINICAL AND DIAGNOSTIC LABORATORY IMMUNOLOGY, Sept. 2002, p. 1044 1048 Vol. 9, No. 5 1071-412X/02/$04.00 0 DOI: 10.1128/CDLI.9.5.1044 1048.2002 Copyright 2002, American Society for Microbiology. All Rights
Disclosures. Co-founder and Chief Science Officer, TechLab
 H. pylori testing Disclosures Co-founder and Chief Science Officer, TechLab Learning Objectives Evaluate the appropriate testing methodology by balancing performance, economics, and workflow. Discuss the
H. pylori testing Disclosures Co-founder and Chief Science Officer, TechLab Learning Objectives Evaluate the appropriate testing methodology by balancing performance, economics, and workflow. Discuss the
Helicobacter pylori:an Emerging Pathogen
 Bacteriology at UW-Madison Bacteriology 330 Home Page Helicobacter pylori:an Emerging Pathogen by Karrie Holston, Department of Bacteriology University of Wisconsin-Madison Description of Helicobacter
Bacteriology at UW-Madison Bacteriology 330 Home Page Helicobacter pylori:an Emerging Pathogen by Karrie Holston, Department of Bacteriology University of Wisconsin-Madison Description of Helicobacter
Helicobacter pylori: Diagnosis, treatment and risks of untreated infection
 Helicobacter pylori: Diagnosis, treatment and risks of untreated infection Klaus Mönkemüller Department of Gastroenterology, Hepatology und Infectius Diseases Otto-von-Guericke University, Magdeburg bb
Helicobacter pylori: Diagnosis, treatment and risks of untreated infection Klaus Mönkemüller Department of Gastroenterology, Hepatology und Infectius Diseases Otto-von-Guericke University, Magdeburg bb
Evaluation of Three Commercial Enzyme Immunoassays Compared with the 13 C Urea Breath Test for Detection of Helicobacter pylori Infection
 JOURNAL OF CLINICAL MICROBIOLOGY, May 1996, p. 1147 1152 Vol. 34, No. 5 0095-1137/96/$04.00 0 Copyright 1996, American Society for Microbiology Evaluation of Three Commercial Enzyme Immunoassays Compared
JOURNAL OF CLINICAL MICROBIOLOGY, May 1996, p. 1147 1152 Vol. 34, No. 5 0095-1137/96/$04.00 0 Copyright 1996, American Society for Microbiology Evaluation of Three Commercial Enzyme Immunoassays Compared
Management of dyspepsia and of Helicobacter pylori infection
 Management of dyspepsia and of Helicobacter pylori infection The University of Nottingham John Atherton Wolfson Digestive Diseases Centre University of Nottingham, UK Community management of dyspepsia
Management of dyspepsia and of Helicobacter pylori infection The University of Nottingham John Atherton Wolfson Digestive Diseases Centre University of Nottingham, UK Community management of dyspepsia
Utility of In House made Rapid Urease Broth Test for Detection of Helicobacter pylori Infection in Resource Constraint Settings
 Original article: Utility of In House made Rapid Urease Broth Test for Detection of Helicobacter pylori Infection in Resource Constraint Settings 1.Dr. Swati Rahul Dhope, 2. Dr. Sachinkumar Vasantrao Wankhede
Original article: Utility of In House made Rapid Urease Broth Test for Detection of Helicobacter pylori Infection in Resource Constraint Settings 1.Dr. Swati Rahul Dhope, 2. Dr. Sachinkumar Vasantrao Wankhede
A PLACEBO CONTROLLED TRIAL OF BISMUTH SALICYLATE IN HELICOBACTER PYLORI ASSOCIATED GASTRITIS
 A PLACEBO CONTROLLED TRIAL OF BISMUTH SALICYLATE IN HELICOBACTER PYLORI ASSOCIATED GASTRITIS Pages with reference to book, From 154 To 156 Javed Iqbal Kazi, Naeem Aon Jafarey, Syed Mahmood Alam ( Department
A PLACEBO CONTROLLED TRIAL OF BISMUTH SALICYLATE IN HELICOBACTER PYLORI ASSOCIATED GASTRITIS Pages with reference to book, From 154 To 156 Javed Iqbal Kazi, Naeem Aon Jafarey, Syed Mahmood Alam ( Department
For the convenience of storage, Cassette and Sample Tubes can be stored separately. OneStep. H.Pylori Antigen. RapiCard InstaTest.
 CORTEZ DIAGNOSTICS, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 USA Tel: (818) 591-3030 Fax: (818) 591-8383 E-mail: onestep@rapidtest.com Web site: www.rapidtest.com See external label
CORTEZ DIAGNOSTICS, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 USA Tel: (818) 591-3030 Fax: (818) 591-8383 E-mail: onestep@rapidtest.com Web site: www.rapidtest.com See external label
Intragastric Acidification Reduces the Occurrence of False-Negative Urea Breath Test Results in Patients Taking a Proton Pump Inhibitor
 THE AMERICAN JOURNAL OF GASTROENTEROLOGY Vol. 96, No. 4, 2001 2001 by Am. Coll. of Gastroenterology ISSN 0002-9270/01/$20.00 Published by Elsevier Science Inc. PII S0002-9270(01)02250-X Intragastric Acidification
THE AMERICAN JOURNAL OF GASTROENTEROLOGY Vol. 96, No. 4, 2001 2001 by Am. Coll. of Gastroenterology ISSN 0002-9270/01/$20.00 Published by Elsevier Science Inc. PII S0002-9270(01)02250-X Intragastric Acidification
Rapid increase in the prevalence of metronidazole resistant Helicobacter pylori in the Netherlands.
 Chapter 2 Rapid increase in the prevalence of metronidazole resistant Helicobacter pylori in the Netherlands. E.J.van der Wouden 1, A.A.van Zwet 2, J.C.Thijs 1, G.D.C.Vosmaer 3, J.A.J.Oom 4, A.de Jong
Chapter 2 Rapid increase in the prevalence of metronidazole resistant Helicobacter pylori in the Netherlands. E.J.van der Wouden 1, A.A.van Zwet 2, J.C.Thijs 1, G.D.C.Vosmaer 3, J.A.J.Oom 4, A.de Jong
The significance of Helicobacter pylori in the approach of dyspepsia in primary care Arents, Nicolaas Lodevikus Augustinus
 University of Groningen The significance of Helicobacter pylori in the approach of dyspepsia in primary care Arents, Nicolaas Lodevikus Augustinus IMPORTANT NOTE: You are advised to consult the publisher's
University of Groningen The significance of Helicobacter pylori in the approach of dyspepsia in primary care Arents, Nicolaas Lodevikus Augustinus IMPORTANT NOTE: You are advised to consult the publisher's
TRANSPARENCY COMMITTEE OPINION. 13 December 2006
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 13 December 2006 HELIKIT 75 mg, powder for oral solution CIP : 343 132-1 Applicant : MAYOLY SPINDLER 13 Curea anhydrous
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 13 December 2006 HELIKIT 75 mg, powder for oral solution CIP : 343 132-1 Applicant : MAYOLY SPINDLER 13 Curea anhydrous
NCCLS. Accutest H. pylori WB Test. (Catalog. No. ID-92A225) For Professional and In Vitro Diagnostic Use Only
 NCCLS Accutest H. pylori WB Test (Catalog. No. ID-92A225) For Professional and In Vitro Diagnostic Use Only A Rapid Visual Test for the Qualitative Detection of IgG Antibodies Specific to Helicobacter
NCCLS Accutest H. pylori WB Test (Catalog. No. ID-92A225) For Professional and In Vitro Diagnostic Use Only A Rapid Visual Test for the Qualitative Detection of IgG Antibodies Specific to Helicobacter
Acid-Peptic Diseases of the Stomach and Duodenum Including Helicobacter pylori and NSAIDs Prof. Sheila Crowe
 Acid-Peptic Diseases of the Stomach and Duodenum Including Helicobacter pylori and NSAIDs 1 Division of Gastroenterology UC San Diego School of Medicine Clinical presentations of Helicobacter pylori infection
Acid-Peptic Diseases of the Stomach and Duodenum Including Helicobacter pylori and NSAIDs 1 Division of Gastroenterology UC San Diego School of Medicine Clinical presentations of Helicobacter pylori infection
Realities of Diagnosing Helicobacter pylon Infection in Clinical Practice: A Case for Non-invasive Indirect Methodologies
 YALE JOURNAL OF BIOLOGY AND MEDICINE 71 (1998), pp. 81-90. Copyright 1999. All rights reserved. Realities of Diagnosing Helicobacter pylon Infection in Clinical Practice: A Case for Non-invasive Indirect
YALE JOURNAL OF BIOLOGY AND MEDICINE 71 (1998), pp. 81-90. Copyright 1999. All rights reserved. Realities of Diagnosing Helicobacter pylon Infection in Clinical Practice: A Case for Non-invasive Indirect
New immunoassay for the detection of Helicobacter pylori infection compared with urease test,
 Chapter V New immunoassay for the detection of Helicobacter pylori infection compared with urease test, 13C breath test and histology: validation in the primary care setting Catherine F Weijnen 1, Henriëtte
Chapter V New immunoassay for the detection of Helicobacter pylori infection compared with urease test, 13C breath test and histology: validation in the primary care setting Catherine F Weijnen 1, Henriëtte
The role of antisecretory drugs in the treatment of Helicobacter pylori infection
 Aliment Pharmacol Ther 1997; 11 (Suppl. 1): 21 25. The role of antisecretory drugs in the treatment of Helicobacter pylori infection W. L. PETERSON Department of Internal Medicine, University of Texas
Aliment Pharmacol Ther 1997; 11 (Suppl. 1): 21 25. The role of antisecretory drugs in the treatment of Helicobacter pylori infection W. L. PETERSON Department of Internal Medicine, University of Texas
Peptic Ulcer Disease Update
 Peptic Ulcer Disease Update Col Pat Storms RAM 2005 Disclosure Information 84th Annual AsMA Scientific Meeting Col Patrick Storms I have no financial relationships to disclose. I will discuss the following
Peptic Ulcer Disease Update Col Pat Storms RAM 2005 Disclosure Information 84th Annual AsMA Scientific Meeting Col Patrick Storms I have no financial relationships to disclose. I will discuss the following
Breath Test Diagnosis of Helicobacter pylori in Peptic Ulcer Disease: A Noninvasive Primary Care Option
 ORIGINAL ARTICLES Breath Test Diagnosis of Helicobacter pylori in Peptic Ulcer Disease: A Noninvasive Primary Care Option Michael W Felz, MD, George] Burke, MD, and Bernard M. Schuman, MD Background: Hellcobacter
ORIGINAL ARTICLES Breath Test Diagnosis of Helicobacter pylori in Peptic Ulcer Disease: A Noninvasive Primary Care Option Michael W Felz, MD, George] Burke, MD, and Bernard M. Schuman, MD Background: Hellcobacter
H. pylori Stool Rapid Test (Cassette)
 H. pylori Stool Rapid Test (Cassette) Cat. No.:DTS590 Pkg.Size: Intended use The H. pylori Stool Cassette is an immunochromatographic screening assay for the qualitative detectionof Helicobacter pylori
H. pylori Stool Rapid Test (Cassette) Cat. No.:DTS590 Pkg.Size: Intended use The H. pylori Stool Cassette is an immunochromatographic screening assay for the qualitative detectionof Helicobacter pylori
Helicobacter pylori REGULAR REVIEW. epithelium.3 This adherence is associated with. Spiral organisms have been noted on the gastric
 1278 Archives ofdisease in Childhood 1990; 65: 1278-1282 REGULAR REVIEW Department of Paediatrics, University College Dublin Correspondence to: Professor B Drumm, Our Lady's Hospital for Sick Children,
1278 Archives ofdisease in Childhood 1990; 65: 1278-1282 REGULAR REVIEW Department of Paediatrics, University College Dublin Correspondence to: Professor B Drumm, Our Lady's Hospital for Sick Children,
ORIGINAL INVESTIGATION. Effectiveness of Helicobacter pylori Therapies in a Clinical Practice Setting
 ORIGINAL INVESTIGATION Effectiveness of Helicobacter pylori Therapies in a Clinical Practice Setting M. Brian Fennerty, MD; David A. Lieberman, MD; Nimish Vakil, MD; Nathan Magaret; Douglas O. Faigel,
ORIGINAL INVESTIGATION Effectiveness of Helicobacter pylori Therapies in a Clinical Practice Setting M. Brian Fennerty, MD; David A. Lieberman, MD; Nimish Vakil, MD; Nathan Magaret; Douglas O. Faigel,
Helicobacter pylori Reinfection Is Virtually Absent after Successful Eradication
 196 Helicobacter pylori Reinfection Is Virtually Absent after Successful Eradication René W. M. van der Hulst, Erik A. J. Rauws, Bircan Köycü, Josbert J. Keller, Fibo J. W. ten Kate, Jacob Dankert, Guido
196 Helicobacter pylori Reinfection Is Virtually Absent after Successful Eradication René W. M. van der Hulst, Erik A. J. Rauws, Bircan Köycü, Josbert J. Keller, Fibo J. W. ten Kate, Jacob Dankert, Guido
The Nobel Prize in Physiology or Medicine for 2005
 The Nobel Prize in Physiology or Medicine for 2005 jointly to Barry J. Marshall and J. Robin Warren for their discovery of "the bacterium Helicobacter pylori and its role in gastritis and peptic ulcer
The Nobel Prize in Physiology or Medicine for 2005 jointly to Barry J. Marshall and J. Robin Warren for their discovery of "the bacterium Helicobacter pylori and its role in gastritis and peptic ulcer
pylori positive patients with duodenal ulcer
 Gut 1996; 39: 629-633 629 PAPERS Prevalence of Helicobacter pylori infection and related gastroduodenal lesions in spouses of Helicobacter pylori positive patients with duodenal ulcer F Parente, G Maconi,
Gut 1996; 39: 629-633 629 PAPERS Prevalence of Helicobacter pylori infection and related gastroduodenal lesions in spouses of Helicobacter pylori positive patients with duodenal ulcer F Parente, G Maconi,
Helicobacter pylori: drowning in a pool of blood?
 Helicobacter pylori: drowning in a pool of blood? Diagnosis of Helicobacter pylori infection is crucial in the short-term and long-term management of patients with bleeding ulcers. If a patient with ulcer
Helicobacter pylori: drowning in a pool of blood? Diagnosis of Helicobacter pylori infection is crucial in the short-term and long-term management of patients with bleeding ulcers. If a patient with ulcer
EDUCATION PRACTICE. Persistent Helicobacter pylori Infection After a Course of Antimicrobial Therapy What s Next? Clinical Scenario.
 CLINICAL GASTROENTEROLOGY AND HEPATOLOGY 2008;6:1086 1090 EDUCATION PRACTICE Persistent Helicobacter pylori Infection After a Course of Antimicrobial Therapy What s Next? RICHARD J. SAAD* and WILLIAM D.
CLINICAL GASTROENTEROLOGY AND HEPATOLOGY 2008;6:1086 1090 EDUCATION PRACTICE Persistent Helicobacter pylori Infection After a Course of Antimicrobial Therapy What s Next? RICHARD J. SAAD* and WILLIAM D.
Helicobacter pylori Eradication Therapy Success Regarding Different Treatment Period Based on Clarithromycin or Metronidazole Triple-Therapy Regimens
 Helicobacter ISSN 1523-5378 Filipec Blackwell Oxford, HEL 1083-4389 1523-5378 Journal XXX Original H. 2008 pylori Kanizaj compilation The UK Eradication Publishing Article Authors et al. Ltd 2008 Therapy
Helicobacter ISSN 1523-5378 Filipec Blackwell Oxford, HEL 1083-4389 1523-5378 Journal XXX Original H. 2008 pylori Kanizaj compilation The UK Eradication Publishing Article Authors et al. Ltd 2008 Therapy
Correlation between Serological and Mucosal Inflammatory
 CLINICAL AND DIAGNOSTIC LABORATORY IMMUNOLOGY, May 1994, p. 325-329 Vol. 1, No. 3 1071-412X/94/$04.00+0 Copyright C) 1994, American Society for Microbiology Correlation between Serological and Mucosal
CLINICAL AND DIAGNOSTIC LABORATORY IMMUNOLOGY, May 1994, p. 325-329 Vol. 1, No. 3 1071-412X/94/$04.00+0 Copyright C) 1994, American Society for Microbiology Correlation between Serological and Mucosal
~Helicobacter pylori, gastritis, and peptic ulceration
 17 Department of Pathology J I Wyatt Department of Medicine T M Shallcross J E Crabtree RV Heatley St James University Hospital, Leeds LS9 7TF Correspondence to: Dr J I Wyatt. Accepted for publication
17 Department of Pathology J I Wyatt Department of Medicine T M Shallcross J E Crabtree RV Heatley St James University Hospital, Leeds LS9 7TF Correspondence to: Dr J I Wyatt. Accepted for publication
"4C-urea breath test in Cpylori gastritis
 Gut, 1989, 3,798-83 "4C-urea breath test in Cpylori gastritis E A J RAUWS, E A V ROYEN, W LANGENBERG, J V WOENSEL, A A VRIJ, AND G N TYTGAT From the Departments of Gastroenterology-Hepatology, Microbiology
Gut, 1989, 3,798-83 "4C-urea breath test in Cpylori gastritis E A J RAUWS, E A V ROYEN, W LANGENBERG, J V WOENSEL, A A VRIJ, AND G N TYTGAT From the Departments of Gastroenterology-Hepatology, Microbiology
Seroconversion and seroreversion in IgG antibodies to Helicobacter pylori: a serology based prospective cohort study
 444 Copenhagen County Centre of Preventive Medicine, Glostrup University Hospital, Glostrup, Denmark S Rosenstock T Jørgensen Department of Clinical Microbiology, Rigshospitalet, Copenhagen, Denmark L
444 Copenhagen County Centre of Preventive Medicine, Glostrup University Hospital, Glostrup, Denmark S Rosenstock T Jørgensen Department of Clinical Microbiology, Rigshospitalet, Copenhagen, Denmark L
Helicobacter pylori infection: several studies on epidemiology, eradication and gastric epithelial cell turnover Liu, W.
 UvA-DARE (Digital Academic Repository) Helicobacter pylori infection: several studies on epidemiology, eradication and gastric epithelial cell turnover Liu, W. Link to publication Citation for published
UvA-DARE (Digital Academic Repository) Helicobacter pylori infection: several studies on epidemiology, eradication and gastric epithelial cell turnover Liu, W. Link to publication Citation for published
Gilles Jequier. Commercial Director Organobalance, a Novozymes Company
 "Latest clinical Evidences showing that a proprietary Lactobacillus reuteri Strain can reduce the Symptoms associated with a Helicobacter pylori Infection" Gilles Jequier Commercial Director Organobalance,
"Latest clinical Evidences showing that a proprietary Lactobacillus reuteri Strain can reduce the Symptoms associated with a Helicobacter pylori Infection" Gilles Jequier Commercial Director Organobalance,
ORIGINAL INVESTIGATION. Maintenance Treatment Is Not Necessary After Helicobacter pylori Eradication and Healing of Bleeding Peptic Ulcer
 ORIGINAL INVESTIGATION Maintenance Treatment Is Not Necessary After Helicobacter pylori Eradication and Healing of Bleeding Peptic Ulcer A 5-Year Prospective, Randomized, Controlled Study Chen-Chiung Liu,
ORIGINAL INVESTIGATION Maintenance Treatment Is Not Necessary After Helicobacter pylori Eradication and Healing of Bleeding Peptic Ulcer A 5-Year Prospective, Randomized, Controlled Study Chen-Chiung Liu,
Helicobacter pylori IgA ELISA Kit
 Helicobacter pylori IgA ELISA Kit Catalog Number KA0964 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Helicobacter pylori IgA ELISA Kit Catalog Number KA0964 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Clinical practice - breath tests
 Clinical practice - breath tests G D Bell Gastroenterology Department, Sunderland District General Hospital, Sunderland, UK The underlying principle of the two non-invasive radio-labelled urea breath tests
Clinical practice - breath tests G D Bell Gastroenterology Department, Sunderland District General Hospital, Sunderland, UK The underlying principle of the two non-invasive radio-labelled urea breath tests
Helicobacter pylori infection: several studies on epidemiology, eradication and gastric epithelial cell turnover Liu, W.
 UvA-DARE (Digital Academic Repository) Helicobacter pylori infection: several studies on epidemiology, eradication and gastric epithelial cell turnover Liu, W. Link to publication Citation for published
UvA-DARE (Digital Academic Repository) Helicobacter pylori infection: several studies on epidemiology, eradication and gastric epithelial cell turnover Liu, W. Link to publication Citation for published
Treating H. pylori in 2016
 Treating H. pylori in 2016 William D. Chey, MD, FACG Professor of Medicine University of Michigan The Case: A 38 yo Russian man presents with recurrent epigastric pain which occurs after meals and sometimes
Treating H. pylori in 2016 William D. Chey, MD, FACG Professor of Medicine University of Michigan The Case: A 38 yo Russian man presents with recurrent epigastric pain which occurs after meals and sometimes
Helicobacter pylori 幽門螺旋桿菌 馬偕紀念醫院新竹分院一般內科, 肝膽腸胃科陳重助醫師
 Helicobacter pylori 幽門螺旋桿菌 馬偕紀念醫院新竹分院一般內科, 肝膽腸胃科陳重助醫師 Hp : Helicobacter pylori Part 1. Pathophysiology and immune response Pathogenesis of Hp infection Part 2. Clinical manifestation Part 3. Dx tests for
Helicobacter pylori 幽門螺旋桿菌 馬偕紀念醫院新竹分院一般內科, 肝膽腸胃科陳重助醫師 Hp : Helicobacter pylori Part 1. Pathophysiology and immune response Pathogenesis of Hp infection Part 2. Clinical manifestation Part 3. Dx tests for
Helicobacter pyloni and upper gastrointestinal
 314 Helicobacter pyloni and upper gastrointestinal disease: a survey of gastroenterologists in the United Kingdom Gut 1995; 37: 314-318 R Milne, R P H Logan, D Harwood, J J Misiewicz, D Forman Health Services
314 Helicobacter pyloni and upper gastrointestinal disease: a survey of gastroenterologists in the United Kingdom Gut 1995; 37: 314-318 R Milne, R P H Logan, D Harwood, J J Misiewicz, D Forman Health Services
A Heaney, J S A Collins, RGPWatson, R J McFarland, K B Bamford, T C K Tham
 186 Royal Victoria Hospital, Belfast, A Heaney J S A Collins Department of Medicine, Queen s University, Belfast, RGPWatson Ulster Hospital, Dundonald, R J McFarland T C K Tham Department of Microbiology
186 Royal Victoria Hospital, Belfast, A Heaney J S A Collins Department of Medicine, Queen s University, Belfast, RGPWatson Ulster Hospital, Dundonald, R J McFarland T C K Tham Department of Microbiology
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: helicobacter_pylori_testing 01/01/2019 N/A 01/01/2020 01/01/2019 Policy Effective April 1, 2019 Description
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: helicobacter_pylori_testing 01/01/2019 N/A 01/01/2020 01/01/2019 Policy Effective April 1, 2019 Description
THE PREVALENCE OF HELICBACTER PYLORI AMONG PATIENTS COMPLAINING FROM ABDOMINAL PAIN
 THE PREVALENCE OF HELICBACTER PYLORI AMONG PATIENTS COMPLAINING FROM ABDOMINAL PAIN Ahed J. Al-Khatib Jordan University of Science and Technology, Jordan Ahmed Saber Abu-zaiton Al-albayt University Abstract
THE PREVALENCE OF HELICBACTER PYLORI AMONG PATIENTS COMPLAINING FROM ABDOMINAL PAIN Ahed J. Al-Khatib Jordan University of Science and Technology, Jordan Ahmed Saber Abu-zaiton Al-albayt University Abstract
Title. Author(s)Kato, Mototsugu. Issue Date Doc URL. Type. Note. File Information.
 Title Effects of lansoprazole plus amoxycillin on the cure Author(s)Kato, Mototsugu Issue Date 1996-12-25 Doc URL http://hdl.handle.net/2115/32629 Type theses (doctoral) Note 共著者あり 共著者名 :Asaka Masahiro,
Title Effects of lansoprazole plus amoxycillin on the cure Author(s)Kato, Mototsugu Issue Date 1996-12-25 Doc URL http://hdl.handle.net/2115/32629 Type theses (doctoral) Note 共著者あり 共著者名 :Asaka Masahiro,
KEYWORDS Dyspepsia, Acid Peptic Disease, Helicobacter Pylori, Urease, Giemsa, Peptic Ulcer, Non-Ulcer Dyspepsia.
 INCIDENCE OF HELICOBACTER PYLORI WITH ACID PEPTIC DISEASE AND MALIGNANT CONDITIONS OF UPPER GASTROINTESTINAL TRACT IN A TERTIARY CENTRE - A PROSPECTIVE STUDY Karunamoorthy Rajachidambaram 1, Dinkaran Kaarthesan
INCIDENCE OF HELICOBACTER PYLORI WITH ACID PEPTIC DISEASE AND MALIGNANT CONDITIONS OF UPPER GASTROINTESTINAL TRACT IN A TERTIARY CENTRE - A PROSPECTIVE STUDY Karunamoorthy Rajachidambaram 1, Dinkaran Kaarthesan
American College of Gastroenterology Guideline on the Management of Helicobacter pylori Infection
 American Journal of Gastroenterology ISSN 0002-9270 C 2007 by Am. Coll. of Gastroenterology doi: 10.1111/j.1572-0241.2007.01393.x Published by Blackwell Publishing American College of Gastroenterology
American Journal of Gastroenterology ISSN 0002-9270 C 2007 by Am. Coll. of Gastroenterology doi: 10.1111/j.1572-0241.2007.01393.x Published by Blackwell Publishing American College of Gastroenterology
High frequency of helicobacter negative gastritis
 Gut 1996; 38: 379-383 High frequency of helicobacter negative gastritis in patients with Crohn's disease 379 L Halme, P Karkkainen, H Rautelin, T U Kosunen, P Sipponen Fourth Department of Surgery, Helsinki
Gut 1996; 38: 379-383 High frequency of helicobacter negative gastritis in patients with Crohn's disease 379 L Halme, P Karkkainen, H Rautelin, T U Kosunen, P Sipponen Fourth Department of Surgery, Helsinki
Helicobacter pylori causes antral gastritis and is associated. Low Rates of Helicobacter pylori Reinfection in Children. Materials and Methods
 GASTROENTEROLOGY 1999;117:336 341 Low Rates of Helicobacter pylori Reinfection in Children MARION ROWLAND,* DEEPAK KUMAR,* LESLIE DALY, PAMELA O CONNOR, DAVID VAUGHAN, and BRENDAN DRUMM*, *Department of
GASTROENTEROLOGY 1999;117:336 341 Low Rates of Helicobacter pylori Reinfection in Children MARION ROWLAND,* DEEPAK KUMAR,* LESLIE DALY, PAMELA O CONNOR, DAVID VAUGHAN, and BRENDAN DRUMM*, *Department of
Effect of Helicobacter pylori infection and its eradication on nutrition
 Aliment Pharmacol Ther 2002; 16: 799 806. Effect of Helicobacter pylori infection and its eradication on nutrition T. FURUTA*, N. SHIRAI*, F. XIAO*, M. TAKASHIMA* & H. HANAI *First Department of Medicine
Aliment Pharmacol Ther 2002; 16: 799 806. Effect of Helicobacter pylori infection and its eradication on nutrition T. FURUTA*, N. SHIRAI*, F. XIAO*, M. TAKASHIMA* & H. HANAI *First Department of Medicine
Research Article Reliability of Diagnostic Tests for Helicobacterpylori Infection
 Gastroenterology Research and Practice Volume 211, Article ID 946, 6 pages doi:1.1155/211/946 Research Article Reliability of Diagnostic Tests for Helicobacterpylori Infection S. Redéen, 1, 2 F. Petersson,
Gastroenterology Research and Practice Volume 211, Article ID 946, 6 pages doi:1.1155/211/946 Research Article Reliability of Diagnostic Tests for Helicobacterpylori Infection S. Redéen, 1, 2 F. Petersson,
Characteristics of Helicobacter pylorinegative and -positive peptic ulcer disease
 Age and Ageing 1998; 27: 427-43 I 1998, British Geriatrics Society Characteristics of Helicobacter pylorinegative and -positive peptic ulcer disease HELENA KEMPPAINEN, ISMO MIHA, HARRY KUJARI 1, LEIF SOURANDER
Age and Ageing 1998; 27: 427-43 I 1998, British Geriatrics Society Characteristics of Helicobacter pylorinegative and -positive peptic ulcer disease HELENA KEMPPAINEN, ISMO MIHA, HARRY KUJARI 1, LEIF SOURANDER
594 Lewin, Weinstein, and Riddell s Gastrointestinal Pathology and Its Clinical Implications
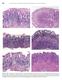 594 Lewin, Weinstein, and Riddell s Gastrointestinal Pathology and Its Clinical Implications Figure 13-20. Stages in the natural history of H. pylori. Biopsies from the antrum are on the left and the oxyntic
594 Lewin, Weinstein, and Riddell s Gastrointestinal Pathology and Its Clinical Implications Figure 13-20. Stages in the natural history of H. pylori. Biopsies from the antrum are on the left and the oxyntic
Duodenal gastric metaplasia and Helicobacter pylori infection in patients with diffuse nodular duodenitis
 Brazilian Journal of Medical and Biological Research (2007) Online Ahead of Print Duodenal gastric metaplasia and Helicobacter pylori infection ISSN 0100-879X 1 Duodenal gastric metaplasia and Helicobacter
Brazilian Journal of Medical and Biological Research (2007) Online Ahead of Print Duodenal gastric metaplasia and Helicobacter pylori infection ISSN 0100-879X 1 Duodenal gastric metaplasia and Helicobacter
PrevalenceofHelicobacterPyloriInfectionamongtheWholeSpectrumofAgeandthePerformanceoftheDifferentDiagnosticTests
 : F Diseases Volume 16 Issue 4 Version 1.0 Year 2016 Type: Double Blind Peer Reviewed International Research Journal Publisher: Global Journals Inc. (USA) Online ISSN: 2249-4618 & Print ISSN: 0975-5888
: F Diseases Volume 16 Issue 4 Version 1.0 Year 2016 Type: Double Blind Peer Reviewed International Research Journal Publisher: Global Journals Inc. (USA) Online ISSN: 2249-4618 & Print ISSN: 0975-5888
One-week low-dose triple therapy for Helicobacter pylori is sufficient for relief from symptoms and healing of duodenal ulcers
 Aliment Pharmacol Ther 1997; 11: 89 93. One-week low-dose triple therapy for Helicobacter pylori is sufficient for relief from symptoms and healing of duodenal ulcers J. LABENZ*, J.-P. IDSTRO M, B. TILLENBURG*,
Aliment Pharmacol Ther 1997; 11: 89 93. One-week low-dose triple therapy for Helicobacter pylori is sufficient for relief from symptoms and healing of duodenal ulcers J. LABENZ*, J.-P. IDSTRO M, B. TILLENBURG*,
Gastric metaplasia and duodenal ulcer disease in children infected by Helicobacter pylori
 Gut 1996;38: 513-517 Gastric metaplasia and duodenal ulcer disease in children infected by Helicobacter pylori 513 Department of Paediatrics S M Gormally B Bourke B Drumm and Public Health Medicine and
Gut 1996;38: 513-517 Gastric metaplasia and duodenal ulcer disease in children infected by Helicobacter pylori 513 Department of Paediatrics S M Gormally B Bourke B Drumm and Public Health Medicine and
Prevpac Pylera Omeclamox-Pak
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.21 Subject: -Pak Page: 1 of 5 Last Review Date: September 20, 2018 -Pak Description (lansoprazole,
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.21 Subject: -Pak Page: 1 of 5 Last Review Date: September 20, 2018 -Pak Description (lansoprazole,
Treatment of Helicobacter pylori in Patients With Duodenal Ulcer Hemorrhage A Long-Term Randomized, Controlled Study
 THE AMERICAN JOURNAL OF GASTROENTEROLOGY Vol. 95, No. 9, 2000 2000 by Am. Coll. of Gastroenterology ISSN 0002-9270/00/$20.00 Published by Elsevier Science Inc. PII S0002-9270(00)01041-8 Treatment of Helicobacter
THE AMERICAN JOURNAL OF GASTROENTEROLOGY Vol. 95, No. 9, 2000 2000 by Am. Coll. of Gastroenterology ISSN 0002-9270/00/$20.00 Published by Elsevier Science Inc. PII S0002-9270(00)01041-8 Treatment of Helicobacter
QUANTA Lite TM H. pylori IgA ELISA For In Vitro Diagnostic Use CLIA Complexity: High
 QUANTA Lite TM H. pylori IgA ELISA 708720 For In Vitro Diagnostic Use CLIA Complexity: High Intended Use The QUANTA Lite TM H. pylori IgA kit is an enzyme-linked immunosorbent assay (ELISA) for the qualitative
QUANTA Lite TM H. pylori IgA ELISA 708720 For In Vitro Diagnostic Use CLIA Complexity: High Intended Use The QUANTA Lite TM H. pylori IgA kit is an enzyme-linked immunosorbent assay (ELISA) for the qualitative
CLIA Complexity for Whole Blood: WAIVED. Intended Use. Summary and Explanation
 CLIA Complexity for Whole Blood: WAIVED Intended Use The is a lateral-flow immunoassay intended for the rapid, qualitative detection of IgG antibodies specific to Helicobacter pylori in whole blood. The
CLIA Complexity for Whole Blood: WAIVED Intended Use The is a lateral-flow immunoassay intended for the rapid, qualitative detection of IgG antibodies specific to Helicobacter pylori in whole blood. The
Om epr azole, am ox icillin, clarithr omycin H elicobacter p y lori
 : 58 6 2000 Om epr azole, am ox icillin, clarithr omycin H elicobacter p y lori, =A b s t r a c t s = T he e f f e ct of He lic obac te r p y lo ri eradic ation of triple therapy w ith ome prazole, am
: 58 6 2000 Om epr azole, am ox icillin, clarithr omycin H elicobacter p y lori, =A b s t r a c t s = T he e f f e ct of He lic obac te r p y lo ri eradic ation of triple therapy w ith ome prazole, am
PEPTIC ULCER DISEASE JOHN R SALTZMAN, MD. Director of Endoscopy Brigham and Women s Hospital Professor of Medicine Harvard Medical School
 PEPTIC ULCER DISEASE JOHN R SALTZMAN, MD Director of Endoscopy Brigham and Women s Hospital Professor of Medicine Harvard Medical School No disclosures Disclosures Overview Causes of peptic ulcer disease
PEPTIC ULCER DISEASE JOHN R SALTZMAN, MD Director of Endoscopy Brigham and Women s Hospital Professor of Medicine Harvard Medical School No disclosures Disclosures Overview Causes of peptic ulcer disease
Prevalence of Helicobacter pylori in Patients with End Stage Renal Disease
 2000;20:97-102 Helicobacter pylori Prevalence of Helicobacter pylori in Patients with End Stage Renal Disease Do Ha Kim, M.D., Hwoon-Yong Jung, M.D., Suk-Kyun Yang, M.D. Weon-Seon Hong, M.D. and Young
2000;20:97-102 Helicobacter pylori Prevalence of Helicobacter pylori in Patients with End Stage Renal Disease Do Ha Kim, M.D., Hwoon-Yong Jung, M.D., Suk-Kyun Yang, M.D. Weon-Seon Hong, M.D. and Young
Catalog # 0W009 0W010 Test Cassettes, murine monoclonal antibody to human IgG (Test Line) and rabbit polyclonal antibody (Control Line)
 For in vitro diagnostic use. CLIA Complexity for Whole Blood: WAIVED INTENDED USE The QuickVue H. pylori Test is a lateral-flow immunoassay intended for the rapid, qualitative detection of IgG antibodies
For in vitro diagnostic use. CLIA Complexity for Whole Blood: WAIVED INTENDED USE The QuickVue H. pylori Test is a lateral-flow immunoassay intended for the rapid, qualitative detection of IgG antibodies
Campylobacter like organisms on the gastric mucosa:
 J Clin Pathol 1984;37:1002-1006 Campylobacter like organisms on the gastric mucosa: culture, histological, and serological studies DM JONES,* AM LESSELLS,t JOAN ELDRIDGE* From the *Public Health Laboratory
J Clin Pathol 1984;37:1002-1006 Campylobacter like organisms on the gastric mucosa: culture, histological, and serological studies DM JONES,* AM LESSELLS,t JOAN ELDRIDGE* From the *Public Health Laboratory
Clinical Policy: Helicobacter Pylori Serology Testing Reference Number: CP.MP.153
 Clinical Policy: Reference Number: CP.MP.153 Effective Date: 12/17 Last Review Date: 12/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory and
Clinical Policy: Reference Number: CP.MP.153 Effective Date: 12/17 Last Review Date: 12/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory and
ReQuest H. Pylori IgG
 Intended use 01-550H 96 Test Set Enzyme Immunoassay for the Detection of Anti- Helicobacter pylori IgG Antibodies in Human Serum The SeraQuest H. Pylori IgG is a qualitative enzyme immunoassay (EIA) kit
Intended use 01-550H 96 Test Set Enzyme Immunoassay for the Detection of Anti- Helicobacter pylori IgG Antibodies in Human Serum The SeraQuest H. Pylori IgG is a qualitative enzyme immunoassay (EIA) kit
ÐÑÏÓÊÅÊËÇÌÅÍÅÓ ÎÅÍÏÃËÙÓÓÅÓ ÁÍÁÊÏÉÍÙÓÅÉÓ ÅËËÇÍÙÍ ÅÑÅÕÍÇÔÙÍ
 ÐÑÏÓÊÅÊËÇÌÅÍÅÓ ÎÅÍÏÃËÙÓÓÅÓ ÁÍÁÊÏÉÍÙÓÅÉÓ ÅËËÇÍÙÍ ÅÑÅÕÍÇÔÙÍ 205 206 ÐÑÏÓÊÅÊËÇÌÅÍÅÓ ÎÅÍÏÃËÙÓÓÅÓ ÁÍÁÊÏÉÍÙÓÅÉÓ ÅËËÇÍÙÍ ÅÑÅÕÍÇÔÙÍ EPITHELIAL CELL TURNOVER IN NON-DYSPLASTIC GASTRIC MUCOSA ADJACENT TO EARLY AND
ÐÑÏÓÊÅÊËÇÌÅÍÅÓ ÎÅÍÏÃËÙÓÓÅÓ ÁÍÁÊÏÉÍÙÓÅÉÓ ÅËËÇÍÙÍ ÅÑÅÕÍÇÔÙÍ 205 206 ÐÑÏÓÊÅÊËÇÌÅÍÅÓ ÎÅÍÏÃËÙÓÓÅÓ ÁÍÁÊÏÉÍÙÓÅÉÓ ÅËËÇÍÙÍ ÅÑÅÕÍÇÔÙÍ EPITHELIAL CELL TURNOVER IN NON-DYSPLASTIC GASTRIC MUCOSA ADJACENT TO EARLY AND
instrument. When 13C-UBT positive value is greater than or equal to / - 0.4, the the subject can be 1. Data and methods details are as follows:
 Application of 13 C-urea breath test in screening helicobacter pylori infection during health examination in Chengdu, Sichuan YANG Yan-hua. LIU Yu-ping. CHENG You-fu, SHUAI Ping. LU Qiao. ZHENG Xiao-xia,
Application of 13 C-urea breath test in screening helicobacter pylori infection during health examination in Chengdu, Sichuan YANG Yan-hua. LIU Yu-ping. CHENG You-fu, SHUAI Ping. LU Qiao. ZHENG Xiao-xia,
SystemicandLocalImmuneResponsetoHPyloriInfectionandtheirCorrelationwiththeDegreeofAntralandDuodenalInflammation
 : F Diseases Volume 16 Issue 3 Version 1.0 Year 2016 Type: Double Blind Peer Reviewed International Research Journal Publisher: Global Journals Inc. (USA) Online ISSN: 2249-4618 & Print ISSN: 0975-5888
: F Diseases Volume 16 Issue 3 Version 1.0 Year 2016 Type: Double Blind Peer Reviewed International Research Journal Publisher: Global Journals Inc. (USA) Online ISSN: 2249-4618 & Print ISSN: 0975-5888
