Factors associated with delayed time to adjuvant chemotherapy in stage iii colon cancer
|
|
|
- Alisha Jefferson
- 5 years ago
- Views:
Transcription
1 Curr Oncol, Vol. 21, pp doi: DELAYED TIME TO ADJUVANT CHEMOTHERAPY ORIGINAL ARTICLE Factors associated with delayed time to adjuvant chemotherapy in stage iii colon cancer A. Chan md,* R. Woods ms,* H. Kennecke md,* and S. Gill md mph* ABSTRACT Background Adjuvant chemotherapy started more than 56 days after colon cancer resection has been associated with lesser overall survival among patients with stage iii colon cancer. The objective of the present population-based study was to determine, in referred patients with resected stage iii colon cancer, factors associated with delayed time to adjuvant chemotherapy (ttac), defined as more than 56 days from the date of surgery. Methods Eligible patients had been diagnosed with stage iii colon cancer and had received at least 1 cycle of adjuvant chemotherapy at one of the four regional cancer treatment sites during Prognostic and treatment information was prospectively collected through the BC Cancer Agency s GI Cancers Outcomes Unit, and Charlson comorbidity score was retrospectively determined by chart review. Chi-square and Wilcoxon rank-sum tests were used to measure associations between the timing of adjuvant chemotherapy and select prognostic and treatment variables. Results Median ttac from surgery for the 395 included patients was 58 days, with 54% of the patients receiving adjuvant chemotherapy beyond the recommended 56 days. On multivariate analysis, only treatment at the highest-volume site was independently associated with delayed ttac. Comorbidity index, age, performance status, T stage, tumour location, and oral chemotherapy (compared with intravenous) were not independently associated with delayed ttac. Delays were observed during each interval associated with the patient s transition from surgery to first cycle of adjuvant chemotherapy. Conclusions More than half the patients failed to receive adjuvant chemotherapy within the recommended ttac of 56 days. Delayed ttac was associated with processrelated delays rather than with patient- or diseaserelated factors. Efforts to improve timely referral, triage of consultations, and chemotherapy wait lists are required. KEY WORDS Colon cancer, stage iii, delay, adjuvant therapy 1. INTRODUCTION Colon cancer is responsible for 8900 deaths annually in Canada and represents the 2nd most common cause of cancer-related morbidity and mortality 1. The use of adjuvant chemotherapy for resected stage iii colon cancer has been shown to significantly lower recurrence rates and to improve overall survival 2. The current standard of treatment includes 5-fluorouracil (5fu) based chemotherapy with any of 5fu leucovorin 3,4, 5fu oxaliplatin 5,6, and capecitabine 7. In clinical trials, adjuvant chemotherapy is typically initiated within 6 8 weeks after surgery, but it has been shown that, outside of the clinical trial setting, up to 19% of patients do not receive adjuvant therapy within that period 8. Several recent metaanalyses have confirmed that delayed administration of adjuvant chemotherapy after curative surgery is associated with significantly lesser overall survival 9,10. In the present study, we used a contemporary population-based patient cohort in the province of British Columbia to ascertain the temporal factors and patient characteristics associated with delayed delivery of adjuvant chemotherapy in referred patients with resected stage iii colon cancer. 181
2 CHAN et al. 2. METHODS 2.1 Data Source Patients with resected stage iii colon cancer who were referred to the BC Cancer Agency (bcca) were identified in the GI Cancers Outcomes Unit database. The bcca is a provincial cancer agency responsible for treatment policy and funding of all systemic therapy in a province of 4.4 million. Of all patients with a diagnosis of colon cancer, 65% are referred to one of five treatment centres. Data relating to receipt of chemotherapy is captured in the provincial pharmacy database. For the purposes of the present analysis, two of the cancer centres were considered together ( centre C ) because of low patient volumes at the most newly opened cancer centre. Provincial treatment guidelines during the study period recommended that adjuvant chemotherapy be initiated within 8 weeks of definitive surgery. For eligible patients with stage iii colon cancer, the recommended regimen is 6 months of modified folfox6 (leucovorin, 5-fluorouracil, oxaliplatin); adjuvant capecitabine is recommended for patients deemed to be ineligible for folfox Patient Selection Patients with resected stage iii colon cancer referred to the bcca between January 1, 2008, and December 31, 2009, and treated with at least 1 cycle of adjuvant chemotherapy were included. Patients were excluded if they had previously been diagnosed with a colon cancer, if any portion of their colon cancer treatment (including surgery or chemotherapy) was performed outside of British Columbia, or if the referral was for a second opinion. The study was approved by the University of British Columbia Research Ethics Board. 2.3 Data Collection Patient data collected included age, sex, Eastern Cooperative Oncology Group (ecog) performance status (as reported by the treating physician at consultation), type of chemotherapy, and regional treatment centre. Disease factor data including tumour location, T stage, N stage, histology, and grade were abstracted from the GI Cancers Outcomes Unit database. Comorbidities, scored using the Charlson comorbidity index, were determined from a retrospective chart review. The time from curative surgery to initiation of adjuvant chemotherapy (ttac) was recorded, as were the intervals between the times of surgery, hospital discharge, referral to bcca, first medical oncology consultation, and first cycle of adjuvant chemotherapy. Delayed ttac was defined as initiation of chemotherapy more than 56 days after surgery. 2.4 Statistical Analysis Chi-square and Wilcoxon rank-sum tests were used to assess differences in characteristics and time intervals for patients who received delayed and timely adjuvant chemotherapy. Multivariate analysis with logistic regression was used to investigate independent patient and tumour characteristics predictive of delayed chemotherapy. 3. RESULTS Of 395 referred patients with resected stage iii colon cancer who met the inclusion criteria over the 2-year study period, 54% also met the definition for delayed ttac. Median time from surgery to adjuvant chemotherapy was 58 days. Table i presents the characteristics of the full cohort and of the timely and delayed therapy groups. Median age at diagnosis was 65 years, and 52% of patients were men. By entry criteria, all patients had node-positive disease (63%, N1 disease; the remainder, N2 disease). Most patients had T3 tumours located in the proximal colon. No significant differences in age or sex were observed between the timely and delayed chemotherapy groups. Also, no significant differences were observed in nodal status, tumour stage, or location of the primary tumour. Oral adjuvant capecitabine was given to 40% of the patients, but the proportion of patients receiving oral capecitabine compared with intravenous folfox6 was not different between the delayed and timely groups. Most patients had a Charlson comorbidity index of 0 1 and an ecog performance of 0 1. On univariate analysis (Table i), an ecog performance status of 2 or more was associated with a significant delay in adjuvant chemotherapy; a higher score on the Charlson comorbidity index was not. However, in multivariate analysis (Table ii), performance status did not retain a significant association with delayed chemotherapy (p = 0.079). Patients were well distributed across health regions, with the most urban centre (centre A) receiving the highest percentage of referrals overall. Compared with the other centres, centre A had a significantly higher proportion of patients who received delayed chemotherapy, and centre was the only factor to retain significance in the multivariate analysis ( p = ). The most common sequence of events was surgery, hospital discharge, referral to bcca, medical oncology consultation, and initiation of adjuvant chemotherapy. That sequence occurred in 69% of cases. As seen in Table iii, significant temporal differences between the timely and delayed chemotherapy groups were identified in all intervals. Overall, the average time from surgery to referral was 15 days; from referral to medical oncology consultation, 21 days; and from medical oncology consultation to initiation of chemotherapy, 20 days. All of those intervals were 182
3 DELAYED TIME TO ADJUVANT CHEMOTHERAPY table i Patient and tumour characteristics by time to adjuvant chemotherapy Variable Time to adjuvant chemotherapy p Value a Overall 56 Days >56 Days Patients (n) Age at diagnosis (years) Median Interquartile range Sex [n (%)] Women 188 (48) 92 (50) 96 (45) Men 207 (52) 90 (50) 117 (55) N Stage [n (%)] N1 249 (63) 119 (65) 130 (61) N2 146 (37) 63 (35) 83 (39) Pathologic T stage [n (%)] T1/2 49 (12) 24 (13) 25 (12) T3 276 (70) 132 (73) 144 (68) T4 68 (17) 25 (14) 43 (20) TX 2 (1) 1 (1) 1 (1) Tumour location [n (%)] Proximal 226 (57) 103 (57) 123 (58) Distal 168 (43) 79 (43) 89 (42) Colon nos 1 (0) 0 (0) 1 (1) cci score [n (%)] (73) 128 (70) 159 (75) (18) 35 (19) 37 (17) 2 29 (7) 16 (9) 13 (6) 3 5 (1) 2 (1) 3 (1) 4 2 (1) 1 (1) 1 (1) Unknown 0 (0) 0 (0) 0 (0) ecog ps [n (%)] (26) 44 (24) 57 (27) (24) 49 (27) 44 (21) 2 22 (6) 5 (3) 17 (8) 3 1 (0) 0 (0) 1 (1) Unknown 178 (45) 84 (46) 94 (44) Centre [n (%)] A 124 (31) 42 (23) 82 (39) B 79 (20) 31 (17) 48 (23) C 110 (28) 58 (32) 52 (24) D 82 (21) 51 (28) 31 (15) Chemotherapy type [n (%)] Intravenous 5fu ± oxaliplatin 235 (60) 104 (57) 131 (62) Oral capecitabine monotherapy 160 (40) 78 (43) 82 (39) a Unknown, TX, and colon nos were removed before statistical testing was performed. Scores of 2 or 3 on the ecog ps and scores 2, 3, or 4 on the cci were pooled for statistical testing. nos = not otherwise specified; cci = Charlson comorbidity index; ecog ps = Eastern Cooperative Oncology Group performance status; 5fu = 5-fluorouracil. 183
4 CHAN et al. table ii Multivariate model for predictors of delayed adjuvant chemotherapy Variable Type iii p value a Comparison or 95% cl p Value a Age at diagnosis Per year , Sex Men vs. women , N Stage N2 vs. N , Pathologic T stage T3 vs. T1/T , T4 vs. T1/T , Tumour location Distal vs. proximal , cci score vs , vs , ecog ps vs , and 3 vs , Unknown vs , Centre B vs. A , C vs. A , D vs. A , Chemotherapy type Capecitabine vs. folfox , a The type iii p value column represents the variable s overall contribution to the model; the p value column represents the specific comparison ( Comparison column) involving the variable. or = odds ratio; cl = confidence limits; cci = Charlson comorbidity index; ecog ps = Eastern Cooperative Oncology Group performance status; folfox = leucovorin 5-fluorouracil oxaliplatin. table iii Differences in process time intervals between timely and delayed adjuvant chemotherapy Variable Time to adjuvant chemotherapy p Value Overall 56 Days >56 Days Surgery to referral (days) (n=370) (n=164) (n=206) Median < Interquartile range Range Referral to medical oncology consultation (days) (n=349) (n=156) (n=193) Median < Interquartile range Range Medical oncology consultation to chemotherapy (days) (n=395) (n=182) (n=213) Median < Interquartile range Range significantly increased in the delayed ttac cohort. On average, the delayed cohort commenced adjuvant chemotherapy 67 days after surgery; the timely cohort started adjuvant chemotherapy an average of 43 days after surgery. 4. DISCUSSION In this population-based analysis examining the factors associated with ttac, more than half the patients referred with resected stage iii colon cancer (54%) received delayed adjuvant chemotherapy. This real-world pattern has previously been reported in the published literature; however, the rates in British Columbia are higher than those reported in other provinces in Canada 8,9. Comparing retrospective studies is difficult, but the proportion of patients with delayed chemotherapy ranges from 26% in Alberta 11 to 32% in Winnipeg 12 and 35% in Saskatchewan 13. Although the definition of delayed chemotherapy varies, most studies have accepted fewer than 56 days as the standard against which to judge delay. In univariate analysis in the present study, poor ecog performance status and treatment at the most 184
5 DELAYED TIME TO ADJUVANT CHEMOTHERAPY urban cancer centre in the province were the only characteristics significantly associated with delayed chemotherapy. It is not surprising that ecog status might result in delayed initiation of chemotherapy: it represents a functional status that might change, especially in the postoperative period. In contrast to previously published meta-analyses, in which the effect of timing was based on clinical trials, our study had a referred population with relatively poorer ecog scores, highlighting some of the phenotypic differences between trial-eligible and real-world patients. However, a higher score on the Charlson comorbidity index was not associated with delayed treatment. That finding might be explained by the strict criteria defining comorbidities in the Charlson index, which might not significantly change in the perioperative period. On multivariate analysis, treatment at the most urban health centre remained the only significant independent factor associated with delayed chemotherapy. It might be postulated that the difference is a function of centre-specific referral and triage processes, workload and patient volumes, and chemotherapy administration resources. Interestingly, oral compared with intravenous chemotherapy was not associated with timeliness of adjuvant therapy. The reasons for that lack of an association are unclear, but might be related to a confounding effect of patient selection. For example, patients selected to receive capecitabine monotherapy as adjuvant chemotherapy for stage iii disease might have had factors related to patient preference, personal supports, or frailty that were associated with delayed initiation of chemotherapy and yet were not captured in our retrospective analysis. Ultimately, delayed ttac appears to be more process-related than patient-related, with all intervals (surgery to referral, referral to medical oncology consultation, and medical oncology consultation to chemotherapy) being significantly prolonged. Given that logistics factors rather than intrinsic patient factors are driving the delay in chemotherapy, we are encouraged that those factors can be remedied. Our findings highlight the need to address process-related delays at each step, including timely referral, medical oncology consultation triage, and management of chemotherapy wait lists. The referring community has to be educated about the importance of timely referral for adjuvant chemotherapy, which typically can be initiated in the immediate postoperative period. Attempts to reduce chemotherapy wait times also need to be considered, including prioritization for patients requiring curative-intent adjuvant chemotherapy and greater utilization of oral fluoropyrimidine oxaliplatin combinations (xelox, capox), which might mitigate delays related to infusional 5fu therapy and implantation of vascular access devices 14. We acknowledge the limitations associated with the retrospective nature of our study. First, although most patient demographics were prospectively recorded, comorbidities and ecog performance status were retrospectively abstracted in a chart review. We were therefore unable to confirm the ecog performance status in 45% of cases. Whether the presence of those performance status data would have remained a significant patient factor between the timely and delayed chemotherapy groups is unclear. Second, specific dates were not available to calculate time from surgery to medical oncology referral for 20 patients or time from bcca referral to medical oncology consultation appointment for 46 patients. However, those patients represent a very small proportion of the 395 in the cohort, and their missing data are unlikely to have significantly altered the results. 5. SUMMARY More than half the referred patients with stage iii colon cancer attending four B.C. cancer centres experienced delayed initiation of adjuvant chemotherapy after surgical resection. Factors associated with delayed chemotherapy included being treated at the most urban provincial centre. Temporal delays occurred in all intervals (surgery to time of medical oncology consultation to time of chemotherapy initiation). Recognizing that delayed ttac is associated with a detrimental effect on survival, efforts must be made to address the delays, potentially including improved education, referral and triage guidelines, and expanded resources. 6. CONFLICT OF INTEREST DISCLOSURES The authors report that there are no financial conflicts related to this paper. 7. REFERENCES 1. Canadian Cancer Society s Steering Committee on Cancer Statistics. Canadian Cancer Statistics Toronto, ON: Canadian Cancer Society; Moertel CG, Fleming TR, Macdonald JS, et al. Levamisole and fluorouracil for adjuvant therapy of resected colon carcinoma. N Engl J Med 1990;322: Wolmark N, Rockette H, Fisher B, et al. The benefit of leucovorin-modulated fluorouracil as postoperative adjuvant therapy for primary colon cancer: results from National Surgical Adjuvant Breast and Bowel Project protocol C-03. J Clin Oncol 1993;11: O Connell MJ, Mailliard JA, Kahn MJ, et al. Controlled trial of fluorouracil and low-dose leucovorin given for 6 months as postoperative adjuvant therapy for colon cancer. J Clin Oncol 1997;15: Andre T, Boni C, Mounedji Boudiaf L, et al. on behalf of the Multicenter International Study of Oxaliplatin/5- Fluorouracil/Leucovorin in the Adjuvant Treatment of Colon Cancer (mosaic) Investigators. Oxaliplatin, fluorouracil, and 185
6 CHAN et al. leucovorin as adjuvant treatment for colon cancer. N Engl J Med 2004;350: Wolmark N, Wieand S, Kuebler PJ, Colangelo L, O Connell MJ, Yothers G. A phase iii trial comparing fulv to fulv + oxaliplatin in stage ii or iii carcinoma of the colon: survival results of nsabp Protocol C-07 [abstract LBA4005]. J Clin Oncol 2008;26:. [Available online at: ascopubs.org/cgi/content/short/26/15_suppl/lba4005; cited May 25, 2014] 7. Twelves C, Wong A, Nowacki MP, et al. Capecitabine as adjuvant treatment for stage iii colon cancer. N Engl J Med 2005;352: Hershman D, Hall MJ, Wang X, et al. Timing of adjuvant chemotherapy initiation after surgery for stage iii colon cancer. Cancer 2006;107: Des Guetz G, Nicolas P, Perret GY, Morere JF, Uzzan B. Does delaying adjuvant chemotherapy after curative surgery for colorectal cancer impair survival? A meta-analysis. Eur J Cancer 2010;46: Biagi JJ, Raphael MJ, Mackillop WJ, Kong W, King WD, Booth CM. Association between time to initiation of adjuvant chemotherapy and survival in colorectal cancer: a systematic review and meta-analysis. JAMA 2011;305: Lima IS, Yasui Y, Scarfe A, Winget M. Association between receipt and timing of adjuvant chemotherapy and survival for patients with stage iii colon cancer in Alberta, Canada. Cancer 2011;117: Cyaykowski PM, Gill S, Kennecke HF, Gordon VL, Turner D. Adjuvant chemotherapy for stage iii colon cancer: does timing matter? Dis Colon Rectum 2011;54: Ahmed S, Ahmad I, Zhu T, et al. Early discontinuation but not the timing of adjuvant therapy affects survival of patients with high-risk colorectal cancer: a population-based study. Dis Colon Rectum 2010;53: Haller DG, Tabenero J, Maroun J, et al. Capecitabine plus oxaliplatin compared with fluorouracil and folinic acid as adjuvant therapy for stage iii colon cancer. J Clin Oncol 2011;29: Correspondence to: Sharlene Gill, BC Cancer Agency, 600 West 10th Avenue, Vancouver, British Columbia V5Z 4E6 sgill@bccancer.bc.ca * BC Cancer Agency, Vancouver, BC. 186
Retrospective analysis of the effect of CAPOX and mfolfox6 dose intensity on survival in colorectal patients in the adjuvant setting
 ORIGINAL ARTICLE CAPOX AND mfolfox6 DOSE INTENSITY AND CLINICAL OUTCOMES IN STAGE III CRC, Mamo et al. Retrospective analysis of the effect of CAPOX and mfolfox6 dose intensity on survival in colorectal
ORIGINAL ARTICLE CAPOX AND mfolfox6 DOSE INTENSITY AND CLINICAL OUTCOMES IN STAGE III CRC, Mamo et al. Retrospective analysis of the effect of CAPOX and mfolfox6 dose intensity on survival in colorectal
Adjuvant therapies for large bowel cancer Wasantha Rathnayake, MD
 LEADING ARTICLE Adjuvant therapies for large bowel cancer Wasantha Rathnayake, MD Consultant Clinical Oncologist, National Cancer Institute, Maharagama, Sri Lanka. Key words: Large bowel; Cancer; Adjuvant
LEADING ARTICLE Adjuvant therapies for large bowel cancer Wasantha Rathnayake, MD Consultant Clinical Oncologist, National Cancer Institute, Maharagama, Sri Lanka. Key words: Large bowel; Cancer; Adjuvant
Current Status of Adjuvant Therapy for Colorectal Cancer
 Review Article [1] May 01, 2004 By Michael J. O connell, MD [2] Adjuvant therapy with chemotherapy and/or radiation therapy in addition to surgery improves outcome for patients with high-risk carcinomas
Review Article [1] May 01, 2004 By Michael J. O connell, MD [2] Adjuvant therapy with chemotherapy and/or radiation therapy in addition to surgery improves outcome for patients with high-risk carcinomas
Age and factors associated with access and time to postoperative adjuvant chemotherapy in colon cancer: a French epidemiological study
 Original Article Age and factors associated with access and time to postoperative adjuvant chemotherapy in colon cancer: a French epidemiological study Jean Capsec 1, Carole Lefebvre 1, Fabienne Chupé
Original Article Age and factors associated with access and time to postoperative adjuvant chemotherapy in colon cancer: a French epidemiological study Jean Capsec 1, Carole Lefebvre 1, Fabienne Chupé
Adjuvant Chemotherapy for Rectal Cancer: Are we making progress?
 Adjuvant Chemotherapy for Rectal Cancer: Are we making progress? Hagen Kennecke, MD, MHA, FRCPC Division Of Medical Oncology British Columbia Cancer Agency October 25, 2008 Objectives Review milestones
Adjuvant Chemotherapy for Rectal Cancer: Are we making progress? Hagen Kennecke, MD, MHA, FRCPC Division Of Medical Oncology British Columbia Cancer Agency October 25, 2008 Objectives Review milestones
The International Duration Evaluation of Adjuvant Chemotherapy study: implications for clinical practice
 Editorial The International Duration Evaluation of Adjuvant Chemotherapy study: implications for clinical practice Marwan Fakih Department of Medical Oncology and Therapeutics Research, City of Hope Comprehensive
Editorial The International Duration Evaluation of Adjuvant Chemotherapy study: implications for clinical practice Marwan Fakih Department of Medical Oncology and Therapeutics Research, City of Hope Comprehensive
/m 2 Oxaliplatin 85 1 Q2W 1-3 Leucovorin Q2W 5-FU Q2W 5-FU Q2W
 癌症診療指引33 Adjuvant therapy of colon cancer mfolfox6 Oxaliplatin 85 1 Q2W 1-3 FOLFOX4 Oxaliplatin 85 1 Q2W 9 Leucovorin 200 1-2 Q2W 5-FU 400 1-2 Q2W 5-FU 600 1-2 Q2W FLOX Oxaliplatin 85 1,15,29 Q8W 4 Leucovorin
癌症診療指引33 Adjuvant therapy of colon cancer mfolfox6 Oxaliplatin 85 1 Q2W 1-3 FOLFOX4 Oxaliplatin 85 1 Q2W 9 Leucovorin 200 1-2 Q2W 5-FU 400 1-2 Q2W 5-FU 600 1-2 Q2W FLOX Oxaliplatin 85 1,15,29 Q8W 4 Leucovorin
Stage III Colon Cancer Susquehanna Cancer Center Warren L Robinson, MD, FACP May 9, 2007
 Stage III Colon Cancer Susquehanna Cancer Center 1997-21 Warren L Robinson, MD, FACP May 9, 27 Stage III Colon Cancer Susquehanna Cancer Center 1997-21 Colorectal cancer is the third most common cancer
Stage III Colon Cancer Susquehanna Cancer Center 1997-21 Warren L Robinson, MD, FACP May 9, 27 Stage III Colon Cancer Susquehanna Cancer Center 1997-21 Colorectal cancer is the third most common cancer
PRINCESS MARGARET CANCER CENTRE CLINICAL PRACTICE GUIDELINES
 PRINCESS MARGARET CANCER CENTRE CLINICAL PRACTICE GUIDELINES GASTROINTESTINAL RECTAL CANCER GI Site Group Rectal Cancer Authors: Dr. Jennifer Knox, Dr. Mairead McNamara 1. INTRODUCTION 3 2. SCREENING AND
PRINCESS MARGARET CANCER CENTRE CLINICAL PRACTICE GUIDELINES GASTROINTESTINAL RECTAL CANCER GI Site Group Rectal Cancer Authors: Dr. Jennifer Knox, Dr. Mairead McNamara 1. INTRODUCTION 3 2. SCREENING AND
Is There a New Standard of Care for Adjuvant Therapy in Colon Cancer? When is 3 Months Enough?
 Is There a New Standard of Care for Adjuvant Therapy in Colon Cancer? When is 3 Months Enough? Jeffrey Meyerhardt, MD, MPH Dana-Farber Cancer Institute Boston, MA 1 Disclosure Ad Board: Genentech Honorarium:
Is There a New Standard of Care for Adjuvant Therapy in Colon Cancer? When is 3 Months Enough? Jeffrey Meyerhardt, MD, MPH Dana-Farber Cancer Institute Boston, MA 1 Disclosure Ad Board: Genentech Honorarium:
Northwestern University, Division of Hematology/Oncology, Chicago, Illinois, USA. Key Words. Colon cancer Stage II Adjuvant chemotherapy
 The Oncologist Dialogues in Oncology Adjuvant Therapy in Stage II Colon Cancer: Current Approaches LISA BADDI, AL BENSON III Northwestern University, Division of Hematology/Oncology, Chicago, Illinois,
The Oncologist Dialogues in Oncology Adjuvant Therapy in Stage II Colon Cancer: Current Approaches LISA BADDI, AL BENSON III Northwestern University, Division of Hematology/Oncology, Chicago, Illinois,
療指引 34 Adjuvant Therapy of Colon Cancer
 療指引 34 Adjuvant Therapy of Colon Cancer mfolfox6 Oxaliplatin 85 1 Q2W 1~3, 10 FLOX Oxaliplatin 85 1,15,29 Q8W 4 Leucovorin 500 1,8,15,22,29,35 Q8W 5-FU 500 1,8,15,22,29,35 Q8W Capecitabine Capecitabine
療指引 34 Adjuvant Therapy of Colon Cancer mfolfox6 Oxaliplatin 85 1 Q2W 1~3, 10 FLOX Oxaliplatin 85 1,15,29 Q8W 4 Leucovorin 500 1,8,15,22,29,35 Q8W 5-FU 500 1,8,15,22,29,35 Q8W Capecitabine Capecitabine
Pharmaco-economic analysis of adjuvant chemotherapy for stage II and III colorectal cancer
 794 Pharmaco-economic analysis of adjuvant chemotherapy for stage II and III colorectal cancer MINA IWAI 1, MICHIO KIMURA 1, EISEKI USAMI 1, TOMOAKI YOSHIMURA 1 and HITOMI TERAMACHI 2 1 Department of Pharmacy,
794 Pharmaco-economic analysis of adjuvant chemotherapy for stage II and III colorectal cancer MINA IWAI 1, MICHIO KIMURA 1, EISEKI USAMI 1, TOMOAKI YOSHIMURA 1 and HITOMI TERAMACHI 2 1 Department of Pharmacy,
S u p p o r t e d b y a n i n d e p e n d e n t E d u c a t i o n a l G r a n t f r o m B a y e r
 EXPERTS KNOWLEDGE SHARE with Prof. Köhne, Dr. Modest and Dr. Vecchione Madrid (Spain) Sunday September 10 th 2017 S u p p o r t e d b y a n i n d e p e n d e n t E d u c a t i o n a l G r a n t f r o m
EXPERTS KNOWLEDGE SHARE with Prof. Köhne, Dr. Modest and Dr. Vecchione Madrid (Spain) Sunday September 10 th 2017 S u p p o r t e d b y a n i n d e p e n d e n t E d u c a t i o n a l G r a n t f r o m
Adjuvant chemotherapy for high-risk stage II and stage III colon cancer: timing of initiation and optimal duration
 JBUON 2018; 23(3): 568-573 ISSN: 1107-0625, online ISSN: 2241-6293 www.jbuon.com E-mail: editorial_office@jbuon.com REVIEW ARTICLE Adjuvant chemotherapy for high-risk stage II and stage III colon cancer:
JBUON 2018; 23(3): 568-573 ISSN: 1107-0625, online ISSN: 2241-6293 www.jbuon.com E-mail: editorial_office@jbuon.com REVIEW ARTICLE Adjuvant chemotherapy for high-risk stage II and stage III colon cancer:
Comparative Efficacy of Adjuvant Chemotherapy in Patients With Dukes B Versus Dukes C Colon Cancer: Results From
 Comparative Efficacy of Adjuvant Chemotherapy in Patients With Dukes B Versus Dukes C Colon Cancer: Results From Four National Surgical Adjuvant Breast and Bowel Project Adjuvant Studies (C-01, C-02, C-03,
Comparative Efficacy of Adjuvant Chemotherapy in Patients With Dukes B Versus Dukes C Colon Cancer: Results From Four National Surgical Adjuvant Breast and Bowel Project Adjuvant Studies (C-01, C-02, C-03,
Adjuvant chemotherapy outcomes in patients over 65 years with early stage colorectal carcinoma
 JBUON 2014; 19(4): 906-912 ISSN: 1107-0625, online ISSN: 2241-6293 www.jbuon.com E-mail: editorial_office@jbuon.com ORIGINAL ARTICLE Adjuvant chemotherapy outcomes in patients over 65 years with early
JBUON 2014; 19(4): 906-912 ISSN: 1107-0625, online ISSN: 2241-6293 www.jbuon.com E-mail: editorial_office@jbuon.com ORIGINAL ARTICLE Adjuvant chemotherapy outcomes in patients over 65 years with early
Cover Page. The handle holds various files of this Leiden University dissertation
 Cover Page The handle http://hdl.handle.net/1887/24307 holds various files of this Leiden University dissertation Author: Broek, Colette van den Title: Optimisation of colorectal cancer treatment Issue
Cover Page The handle http://hdl.handle.net/1887/24307 holds various files of this Leiden University dissertation Author: Broek, Colette van den Title: Optimisation of colorectal cancer treatment Issue
Efficacy and Toxicity of Adjuvant Chemotherapy in Elderly Patients with Colon Carcinoma
 1931 Efficacy and Toxicity of Adjuvant Chemotherapy in Elderly Patients with Colon Carcinoma A 10-Year Experience of the Geisinger Medical Center Farid Fata, M.D. 1 Ayoub Mirza, M.D. 2 G. Craig Wood, M.S.
1931 Efficacy and Toxicity of Adjuvant Chemotherapy in Elderly Patients with Colon Carcinoma A 10-Year Experience of the Geisinger Medical Center Farid Fata, M.D. 1 Ayoub Mirza, M.D. 2 G. Craig Wood, M.S.
Targeted Therapies in Metastatic Colorectal Cancer: An Update
 Targeted Therapies in Metastatic Colorectal Cancer: An Update ASCO 2007: Targeted Therapies in Metastatic Colorectal Cancer: An Update Bevacizumab is effective in combination with XELOX or FOLFOX-4 Bevacizumab
Targeted Therapies in Metastatic Colorectal Cancer: An Update ASCO 2007: Targeted Therapies in Metastatic Colorectal Cancer: An Update Bevacizumab is effective in combination with XELOX or FOLFOX-4 Bevacizumab
Radiotherapy and Conservative Surgery For Merkel Cell Carcinoma - The British Columbia Cancer Agency Experience
 Radiotherapy and Conservative Surgery For Merkel Cell Carcinoma - The British Columbia Cancer Agency Experience Poster No.: RO-0003 Congress: RANZCR FRO 2012 Type: Scientific Exhibit Authors: C. Harrington,
Radiotherapy and Conservative Surgery For Merkel Cell Carcinoma - The British Columbia Cancer Agency Experience Poster No.: RO-0003 Congress: RANZCR FRO 2012 Type: Scientific Exhibit Authors: C. Harrington,
Surgical Management of Advanced Stage Colon Cancer. Nathan Huber, MD 6/11/14
 Surgical Management of Advanced Stage Colon Cancer Nathan Huber, MD 6/11/14 Colon Cancer Overview Approximately 50,000 attributable deaths per year Colorectal cancer is the 3 rd most common cause of cancer-related
Surgical Management of Advanced Stage Colon Cancer Nathan Huber, MD 6/11/14 Colon Cancer Overview Approximately 50,000 attributable deaths per year Colorectal cancer is the 3 rd most common cause of cancer-related
pan-canadian Oncology Drug Review Final Economic Guidance Report Irinotecan liposome (Onivyde) for Metastatic Pancreatic Cancer January 5, 2018
 pan-canadian Oncology Drug Review Final Economic Guidance Report Irinotecan liposome (Onivyde) for Metastatic Pancreatic Cancer January 5, 2018 DISCLAIMER Not a Substitute for Professional Advice This
pan-canadian Oncology Drug Review Final Economic Guidance Report Irinotecan liposome (Onivyde) for Metastatic Pancreatic Cancer January 5, 2018 DISCLAIMER Not a Substitute for Professional Advice This
EARLY STAGE COLON CANCER
 EARLY STAGE COLON CANCER Effective Date: December 2017 Copyright (2017) Alberta Health Services This material is protected by Canadian and other international copyright laws. All rights reserved. This
EARLY STAGE COLON CANCER Effective Date: December 2017 Copyright (2017) Alberta Health Services This material is protected by Canadian and other international copyright laws. All rights reserved. This
Comparative Effectiveness of Adjuvant Chemotherapy in Elderly Colon Cancer Patients
 Cancer and Clinical Oncology; Vol. 2, No. 1; 2013 ISSN 1927-4858 E-ISSN 1927-4866 Published by Canadian Center of Science and Education Comparative Effectiveness of Adjuvant Chemotherapy in Elderly Colon
Cancer and Clinical Oncology; Vol. 2, No. 1; 2013 ISSN 1927-4858 E-ISSN 1927-4866 Published by Canadian Center of Science and Education Comparative Effectiveness of Adjuvant Chemotherapy in Elderly Colon
Adjuvant Chemotherapy for Patients with Resected Dukes C and High-risk B2 Colon Cancer with Fluorouracil and Levamisole
 733 Adjuvant Chemotherapy for Patients with Resected Dukes C and High-risk B2 Colon Cancer with Fluorouracil and Levamisole E Au,*FAMS, M Med (Int Med), MRCP, P T Ang,**FAMS, FACP, FRCP (Edin), F Seow-Choen,***FAMS,
733 Adjuvant Chemotherapy for Patients with Resected Dukes C and High-risk B2 Colon Cancer with Fluorouracil and Levamisole E Au,*FAMS, M Med (Int Med), MRCP, P T Ang,**FAMS, FACP, FRCP (Edin), F Seow-Choen,***FAMS,
Evaluation of the Efficacy of Modified De Gramont and Modified FOLFOX4 Regimens for Adjuvant Therapy of Locally Advanced Rectal Cancer
 Efficacy of Modified De Gramont and FOLFOX4 Regimens for Locally Advanced Rectal Cancer RESEARCH COMMUNICATION Evaluation of the Efficacy of Modified De Gramont and Modified FOLFOX4 Regimens for Adjuvant
Efficacy of Modified De Gramont and FOLFOX4 Regimens for Locally Advanced Rectal Cancer RESEARCH COMMUNICATION Evaluation of the Efficacy of Modified De Gramont and Modified FOLFOX4 Regimens for Adjuvant
大腸直腸癌 抗癌藥物治療指引 討論日期 團隊討論 : 105 年 10 月 19 日 三院討論 : 105 年 12 月 7 日 團隊確認 : 106 年 1 月 25 日 核備日期 : 106 年 4 月 18 日
 大腸直腸癌 抗癌藥物治療指 討論日期 團隊討論 : 05 年 0 月 9 日 三院討論 : 05 年 2 月 7 日 團隊確認 : 06 年 月 25 日 核備日期 : 06 年 4 月 8 日 Adjuvant Therapy of Colon Cancer mfolfox6 參考文獻 -3 Oxaliplatin 85 Q2W 2 Leucovorin 400 Q2W 2 5-FU 400 Q2W
大腸直腸癌 抗癌藥物治療指 討論日期 團隊討論 : 05 年 0 月 9 日 三院討論 : 05 年 2 月 7 日 團隊確認 : 06 年 月 25 日 核備日期 : 06 年 4 月 8 日 Adjuvant Therapy of Colon Cancer mfolfox6 參考文獻 -3 Oxaliplatin 85 Q2W 2 Leucovorin 400 Q2W 2 5-FU 400 Q2W
Randomized phase III trial of treatment duration for oral uracil and tegafur plus
 Annals of Oncology Advance Access published September 7, 2015 1 Randomized phase III trial of treatment duration for oral uracil and tegafur plus leucovorin as adjuvant chemotherapy for patients with stage
Annals of Oncology Advance Access published September 7, 2015 1 Randomized phase III trial of treatment duration for oral uracil and tegafur plus leucovorin as adjuvant chemotherapy for patients with stage
Adjuvant Chemotherapy for Stage II Colon Cancer
 March 01, 2008 By Scott Kopetz, MD [1], Daniela Freitas, MD [2], Aknar F. C. Calabrich, MD [3], and Paulo M. Hoff, MD, FACP [4] Adjuvant therapy is defined as any treatment administered after surgical
March 01, 2008 By Scott Kopetz, MD [1], Daniela Freitas, MD [2], Aknar F. C. Calabrich, MD [3], and Paulo M. Hoff, MD, FACP [4] Adjuvant therapy is defined as any treatment administered after surgical
RESEARCH ARTICLE. Joanne Chiu 1, Vikki Tang 1, Roland Leung 1, Hilda Wong 1, Kin Wah Chu 2, Jensen Poon 3, Richard J Epstein 4, Thomas Yau 1,3,5 *
 DOI:http://dx.doi.org/10.7314/APJCP.2013.14.11.6585 Adjuvant XELOX in Asian Patients with Colorectal Cancer RESEARCH ARTICLE Efficacy and Tolerability of Adjuvant Oral Capecitabine plus Intravenous Oxaliplatin
DOI:http://dx.doi.org/10.7314/APJCP.2013.14.11.6585 Adjuvant XELOX in Asian Patients with Colorectal Cancer RESEARCH ARTICLE Efficacy and Tolerability of Adjuvant Oral Capecitabine plus Intravenous Oxaliplatin
Preoperative or Postoperative Therapy for the Management of Patients with Stage II or III Rectal Cancer
 Evidence-Based Series 2-4 Version 2 A Quality Initiative of the Program in Evidence-Based Care (PEBC), Cancer Care Ontario (CCO) Preoperative or Postoperative Therapy for the Management of Patients with
Evidence-Based Series 2-4 Version 2 A Quality Initiative of the Program in Evidence-Based Care (PEBC), Cancer Care Ontario (CCO) Preoperative or Postoperative Therapy for the Management of Patients with
Research Article The Aetiology of Delay to Commencement of Adjuvant Chemotherapy following Colorectal Resection
 International Surgical Oncology, Article ID 670212, 7 pages http://dx.doi.org/10.1155/2014/670212 Research Article The Aetiology of Delay to Commencement of Adjuvant Chemotherapy following Colorectal Resection
International Surgical Oncology, Article ID 670212, 7 pages http://dx.doi.org/10.1155/2014/670212 Research Article The Aetiology of Delay to Commencement of Adjuvant Chemotherapy following Colorectal Resection
Reference No: Author(s) Approval date: 12/05/16. Committee. June Operational Date: Review:
 Reference No: Title: Author(s) Systemic Anti-Cancer Therapy (SACT) Guidelines for Biliary Tract Cancer (BTC) Dr Colin Purcell, Consultant Medical Oncologist on behalf of the GI Oncologists Group, Cancer
Reference No: Title: Author(s) Systemic Anti-Cancer Therapy (SACT) Guidelines for Biliary Tract Cancer (BTC) Dr Colin Purcell, Consultant Medical Oncologist on behalf of the GI Oncologists Group, Cancer
Disclosures. Clinical and molecular features to guide adjuvant therapy. Personalized Medicine - Decision Tools -
 Disclosures Clinical and molecular features to guide adjuvant therapy Daniel Sargent Professor of Biostatistics & Oncology Mayo Clinic Consulting activities Amgen Pfizer Roche/Genentech Sanofi-Aventis
Disclosures Clinical and molecular features to guide adjuvant therapy Daniel Sargent Professor of Biostatistics & Oncology Mayo Clinic Consulting activities Amgen Pfizer Roche/Genentech Sanofi-Aventis
Efficiency and tolerability of 5- fluorouracil-based adjuvant chemotherapy in elderly patients with colorectal carcinoma
 Turkish Journal of Cancer Volume 34, No.4, 2004 139 Efficiency and tolerability of 5- fluorouracil-based adjuvant chemotherapy in elderly patients with colorectal carcinoma LHAN ÖZTOP 1, ARZU YAREN 1,
Turkish Journal of Cancer Volume 34, No.4, 2004 139 Efficiency and tolerability of 5- fluorouracil-based adjuvant chemotherapy in elderly patients with colorectal carcinoma LHAN ÖZTOP 1, ARZU YAREN 1,
Impact of Adjuvant Chemotherapy in Patients With Curatively Resected Stage IV Colorectal Cancer
 Impact of in Patients With Curatively Resected Stage IV Colorectal Cancer Hirotoshi Kobayashi, MD, FACS, Kenjiro Kotake, MD, and Kenichi Sugihara, MD, Study Group for Peritoneal Metastasis from Colorectal
Impact of in Patients With Curatively Resected Stage IV Colorectal Cancer Hirotoshi Kobayashi, MD, FACS, Kenjiro Kotake, MD, and Kenichi Sugihara, MD, Study Group for Peritoneal Metastasis from Colorectal
Management of Advanced Colorectal Cancer in Older Patients
 Review Article [1] April 15, 2005 By Stuart M. Lichtman, MD, FACP [2] Many elderly individuals have substantial life expectancy, even in the setting of significant illness. There is evidence to indicate
Review Article [1] April 15, 2005 By Stuart M. Lichtman, MD, FACP [2] Many elderly individuals have substantial life expectancy, even in the setting of significant illness. There is evidence to indicate
Long term survival study of de-novo metastatic breast cancers with or without primary tumor resection
 Long term survival study of de-novo metastatic breast cancers with or without primary tumor resection Dr. Michael Co Division of Breast Surgery Queen Mary Hospital The University of Hong Kong Conflicts
Long term survival study of de-novo metastatic breast cancers with or without primary tumor resection Dr. Michael Co Division of Breast Surgery Queen Mary Hospital The University of Hong Kong Conflicts
Radiation Therapy for Resectable Colon Cancer
 Review Article [1] February 01, 2006 By Brian G. Czito, MD [2], Johanna C. Bendell, MD [3], and Christopher G. Willett, MD [4] Colon cancer is a major public health problem. The primary treatment is resection.
Review Article [1] February 01, 2006 By Brian G. Czito, MD [2], Johanna C. Bendell, MD [3], and Christopher G. Willett, MD [4] Colon cancer is a major public health problem. The primary treatment is resection.
Study population The study population comprised patients with completely resected Stage III colon cancer.
 Cost-effectiveness of oxaliplatin and capecitabine in the adjuvant treatment of stage III colon cancer Eggington S, Tappenden P, Pandor A, Paisley S, Saunders M, Seymour M, Sutcliffe P, Chilcott J Record
Cost-effectiveness of oxaliplatin and capecitabine in the adjuvant treatment of stage III colon cancer Eggington S, Tappenden P, Pandor A, Paisley S, Saunders M, Seymour M, Sutcliffe P, Chilcott J Record
A clinical study of adjuvant chemotherapy in younger and elder rectal cancer patientsa
 A clinical study of adjuvant chemotherapy in younger and elder rectal cancer patientsa The role of postoperative chemotherapy (CT) is still unclear and the evidence for recommendations of adjuvant therapy
A clinical study of adjuvant chemotherapy in younger and elder rectal cancer patientsa The role of postoperative chemotherapy (CT) is still unclear and the evidence for recommendations of adjuvant therapy
Jonathan Dickinson, LCL Xeloda
 Xeloda A blockbuster in the making Jonathan Dickinson, LCL Xeloda Xeloda unique tumor-activated mechanism Delivering more cancer-killing agent straight into cancer Highly effective comparable efficacy
Xeloda A blockbuster in the making Jonathan Dickinson, LCL Xeloda Xeloda unique tumor-activated mechanism Delivering more cancer-killing agent straight into cancer Highly effective comparable efficacy
Oncologic Outcomes of Stage IIIA Colon Cancer for Different Chemotherapeutic Regimens
 Original Article Journal of the Korean Society of J Korean Soc Coloproctol 2012;28(5):259-264 http://dx.doi.org/10.3393/jksc.2012.28.5.259 pissn 2093-7822 eissn 2093-7830 Oncologic Outcomes of Stage IIIA
Original Article Journal of the Korean Society of J Korean Soc Coloproctol 2012;28(5):259-264 http://dx.doi.org/10.3393/jksc.2012.28.5.259 pissn 2093-7822 eissn 2093-7830 Oncologic Outcomes of Stage IIIA
DETERMINATION OF OXALIPLATIN DOSE IN ADJUVANT CHEMOTHERAPY : VALIDATION OF A DEDICATED SOFTWARE BASED ON INITIAL CT SCAN
 DETERMINATION OF OXALIPLATIN DOSE IN ADJUVANT CHEMOTHERAPY : VALIDATION OF A DEDICATED SOFTWARE BASED ON INITIAL CT SCAN R Tetreau, M Theillac, F Castan, P Senesse, V Baracos 07 APRIL 2016 BACKGROUND 1
DETERMINATION OF OXALIPLATIN DOSE IN ADJUVANT CHEMOTHERAPY : VALIDATION OF A DEDICATED SOFTWARE BASED ON INITIAL CT SCAN R Tetreau, M Theillac, F Castan, P Senesse, V Baracos 07 APRIL 2016 BACKGROUND 1
Chemotherapy for resectable liver mets: Options and Issues. Herbert Hurwitz Duke University Medical Center Durham, North Carolina, USA
 Chemotherapy for resectable liver mets: Options and Issues Herbert Hurwitz Duke University Medical Center Durham, North Carolina, USA Chemotherapy regimens in 1 st line mcrc Standard FOLFOX-Bev FOLFIRI-Bev
Chemotherapy for resectable liver mets: Options and Issues Herbert Hurwitz Duke University Medical Center Durham, North Carolina, USA Chemotherapy regimens in 1 st line mcrc Standard FOLFOX-Bev FOLFIRI-Bev
Case Conference. Craig Morgenthal Department of Surgery Long Island College Hospital
 Case Conference Craig Morgenthal Department of Surgery Long Island College Hospital Neoadjuvant versus Adjuvant Radiation Therapy in Rectal Carcinoma Epidemiology American Cancer Society statistics for
Case Conference Craig Morgenthal Department of Surgery Long Island College Hospital Neoadjuvant versus Adjuvant Radiation Therapy in Rectal Carcinoma Epidemiology American Cancer Society statistics for
MEETING SUMMARY ESMO 2018, Munich, Germany. Dr. Jenny Seligmann University of Leeds, UK HIGHLIGHTS ON COLORECTAL CANCER
 MEETING SUMMARY ESMO 2018, Munich, Germany Dr. Jenny Seligmann University of Leeds, UK HIGHLIGHTS ON COLORECTAL CANCER DISCLAIMER Please note: The views expressed within this presentation are the personal
MEETING SUMMARY ESMO 2018, Munich, Germany Dr. Jenny Seligmann University of Leeds, UK HIGHLIGHTS ON COLORECTAL CANCER DISCLAIMER Please note: The views expressed within this presentation are the personal
Carcinoma del retto: Highlights
 Carcinoma del retto: Highlights Stefano Cordio Struttura Complessa di Oncologia Medica ARNAS Garibaldi Catania Roma 17 Febbraio 2018 Disclosures Advisory Committee, research funding and speakers bureau
Carcinoma del retto: Highlights Stefano Cordio Struttura Complessa di Oncologia Medica ARNAS Garibaldi Catania Roma 17 Febbraio 2018 Disclosures Advisory Committee, research funding and speakers bureau
ADJUVANT CHEMOTHERAPY...
 Colorectal Pathway Board: Non-Surgical Oncology Guidelines October 2015 Organization» Table of Contents ADJUVANT CHEMOTHERAPY... 2 DUKES C/ TNM STAGE 3... 2 DUKES B/ TNM STAGE 2... 3 LOCALLY ADVANCED
Colorectal Pathway Board: Non-Surgical Oncology Guidelines October 2015 Organization» Table of Contents ADJUVANT CHEMOTHERAPY... 2 DUKES C/ TNM STAGE 3... 2 DUKES B/ TNM STAGE 2... 3 LOCALLY ADVANCED
COLON CANCER FOLLOW UP GUIDELINES
 COLON CANCER FOLLOW UP GUIDELINES CAGPO Dr. Raziya Mia October 1, 2016 1 DISCLOSURES NONE 2 MITIGATING POTENTIAL BIAS Recommendations are consistent with published guidelines American Society of Clinical
COLON CANCER FOLLOW UP GUIDELINES CAGPO Dr. Raziya Mia October 1, 2016 1 DISCLOSURES NONE 2 MITIGATING POTENTIAL BIAS Recommendations are consistent with published guidelines American Society of Clinical
Adjuvant therapy in colon cancer: which treatment in 2005?
 Annals of Oncology 16 (Supplement 4): iv69 iv73, 2005 doi:10.1093/annonc/mdi911 Adjuvant therapy in colon cancer: which treatment in 2005? F. Di Costanzo* & L. Doni Medical Oncology Unit, Department of
Annals of Oncology 16 (Supplement 4): iv69 iv73, 2005 doi:10.1093/annonc/mdi911 Adjuvant therapy in colon cancer: which treatment in 2005? F. Di Costanzo* & L. Doni Medical Oncology Unit, Department of
A Population-Based Study on the Uptake and Utilization of Stereotactic Radiosurgery (SRS) for Brain Metastasis in Nova Scotia
 A Population-Based Study on the Uptake and Utilization of Stereotactic Radiosurgery (SRS) for Brain Metastasis in Nova Scotia Gaurav Bahl, Karl Tennessen, Ashraf Mahmoud-Ahmed, Dorianne Rheaume, Ian Fleetwood,
A Population-Based Study on the Uptake and Utilization of Stereotactic Radiosurgery (SRS) for Brain Metastasis in Nova Scotia Gaurav Bahl, Karl Tennessen, Ashraf Mahmoud-Ahmed, Dorianne Rheaume, Ian Fleetwood,
Advances in gastric cancer: How to approach localised disease?
 Advances in gastric cancer: How to approach localised disease? Andrés Cervantes Professor of Medicine Classical approach to localised gastric cancer Surgical resection Pathology assessment and estimation
Advances in gastric cancer: How to approach localised disease? Andrés Cervantes Professor of Medicine Classical approach to localised gastric cancer Surgical resection Pathology assessment and estimation
Adjuvant therapy in older adults: controversies and challenges - Colorectal cancer -
 International Society of Geriatric Oncology Lisbon October 23 rd 25t h 2014 Adjuvant therapy in older adults: controversies and challenges - Colorectal cancer - Claus-Henning Köhne Klinik für Onkologie
International Society of Geriatric Oncology Lisbon October 23 rd 25t h 2014 Adjuvant therapy in older adults: controversies and challenges - Colorectal cancer - Claus-Henning Köhne Klinik für Onkologie
Follow this and additional works at: Part of the Neoplasms Commons
 Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 2015 Will the Addition of Oxaliplatin to 5-Fluorouracil
Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 2015 Will the Addition of Oxaliplatin to 5-Fluorouracil
Inadequate lymph node sampling as a risk factor in stage II colon cancer
 Inadequate lymph node sampling as a risk factor in stage II colon cancer F. Zakaria and N. EL- Mashad Clinical Oncology Department. Faculty of Medicine, Tanta University Hospital, Tanta, Egypt Abstract
Inadequate lymph node sampling as a risk factor in stage II colon cancer F. Zakaria and N. EL- Mashad Clinical Oncology Department. Faculty of Medicine, Tanta University Hospital, Tanta, Egypt Abstract
Peritoneal Involvement in Stage II Colon Cancer
 Anatomic Pathology / PERITONEAL INVOLVEMENT IN STAGE II COLON CANCER Peritoneal Involvement in Stage II Colon Cancer A.M. Lennon, MB, MRCPI, H.E. Mulcahy, MD, MRCPI, J.M.P. Hyland, MCh, FRCS, FRCSI, C.
Anatomic Pathology / PERITONEAL INVOLVEMENT IN STAGE II COLON CANCER Peritoneal Involvement in Stage II Colon Cancer A.M. Lennon, MB, MRCPI, H.E. Mulcahy, MD, MRCPI, J.M.P. Hyland, MCh, FRCS, FRCSI, C.
Chemotherapy of colon cancers
 Chemotherapy of colon cancers Stage distribution Stage I : 15% T 1,2 NO Stage IV: 20 25% M+ Stage II : 20 30% T3,4 NO Stage III N+: 30 40% clinical stages I, II, or III colon cancer are at risk for having
Chemotherapy of colon cancers Stage distribution Stage I : 15% T 1,2 NO Stage IV: 20 25% M+ Stage II : 20 30% T3,4 NO Stage III N+: 30 40% clinical stages I, II, or III colon cancer are at risk for having
Weekly 5-fluorouracil and leucovorin: achieving lower toxicity with higher dose-intensity in adjuvant chemotherapy after colorectal cancer resection
 Original article Annals of Oncology 15: 568 573, 2004 DOI: 10.1093/annonc/mdh134 Weekly 5-fluorouracil and leucovorin: achieving lower toxicity with higher dose-intensity in adjuvant chemotherapy after
Original article Annals of Oncology 15: 568 573, 2004 DOI: 10.1093/annonc/mdh134 Weekly 5-fluorouracil and leucovorin: achieving lower toxicity with higher dose-intensity in adjuvant chemotherapy after
Adjuvant treatment Colon Cancer
 ESMO Preceptorship Colorectal Cancer, October 2016 Singapore Adjuvant treatment Colon Cancer Claus-Henning Köhne University Clinic for Onkology und Haematology Oldenburg, Germany Aim of the lecture Adjuvant
ESMO Preceptorship Colorectal Cancer, October 2016 Singapore Adjuvant treatment Colon Cancer Claus-Henning Köhne University Clinic for Onkology und Haematology Oldenburg, Germany Aim of the lecture Adjuvant
Adjuvant/neoadjuvant systemic treatment of colorectal cancer
 5th ESO-ESMO Eastern Europe and Balkan Region Masterclass in Medical Oncology Belgrade, June 19 th 2018 Adjuvant/neoadjuvant systemic treatment of colorectal cancer Carlotta Antoniotti Polo Oncologico
5th ESO-ESMO Eastern Europe and Balkan Region Masterclass in Medical Oncology Belgrade, June 19 th 2018 Adjuvant/neoadjuvant systemic treatment of colorectal cancer Carlotta Antoniotti Polo Oncologico
Reference No: Author(s) 12/05/16. Approval date: committee. June Operational Date: Review:
 Reference No: Title: Author(s) Systemic Anti-Cancer Therapy (SACT) Guidelines for Pancreatic Adenocarcinoma Dr Colin Purcell, Consultant Medical Oncologist & on behalf of the GI Oncologists Group, Cancer
Reference No: Title: Author(s) Systemic Anti-Cancer Therapy (SACT) Guidelines for Pancreatic Adenocarcinoma Dr Colin Purcell, Consultant Medical Oncologist & on behalf of the GI Oncologists Group, Cancer
THE VALIDITY OF ADMINISTRATIVE DATA AND PATTERNS OF CHEMOTHERAPY USE AMONG ELDERLY COLORECTAL CANCER PATIENTS. Jennifer L.
 THE VALIDITY OF ADMINISTRATIVE DATA AND PATTERNS OF CHEMOTHERAPY USE AMONG ELDERLY COLORECTAL CANCER PATIENTS Jennifer L. Lund, MSPH A dissertation submitted to the faculty of the University of North Carolina
THE VALIDITY OF ADMINISTRATIVE DATA AND PATTERNS OF CHEMOTHERAPY USE AMONG ELDERLY COLORECTAL CANCER PATIENTS Jennifer L. Lund, MSPH A dissertation submitted to the faculty of the University of North Carolina
ESMO 2017, Madrid, Spain Dr. Loredana Vecchione Charite Comprehensive Cancer Center, Berlin HIGHLIGHTS ON CANCERS OF THE UPPER GI TRACT
 ESMO 2017, Madrid, Spain Dr. Loredana Vecchione Charite Comprehensive Cancer Center, Berlin HIGHLIGHTS ON CANCERS OF THE UPPER GI TRACT DOCETAXEL, OXALIPLATIN AND FLUOROURACIL/LEUCOVORIN (FLOT) FOR RESECTABLE
ESMO 2017, Madrid, Spain Dr. Loredana Vecchione Charite Comprehensive Cancer Center, Berlin HIGHLIGHTS ON CANCERS OF THE UPPER GI TRACT DOCETAXEL, OXALIPLATIN AND FLUOROURACIL/LEUCOVORIN (FLOT) FOR RESECTABLE
Cover Page. The handle holds various files of this Leiden University dissertation.
 Cover Page The handle http://hdl.handle.net/1887/38705 holds various files of this Leiden University dissertation. Author: Gijn, Willem van Title: Rectal cancer : developments in multidisciplinary treatment,
Cover Page The handle http://hdl.handle.net/1887/38705 holds various files of this Leiden University dissertation. Author: Gijn, Willem van Title: Rectal cancer : developments in multidisciplinary treatment,
Van Cutsem E et al. Proc ASCO 2009;Abstract LBA4509.
 Efficacy Results from the ToGA Trial: A Phase III Study of Trastuzumab Added to Standard Chemotherapy in First-Line HER2- Positive Advanced Gastric Cancer Van Cutsem E et al. Proc ASCO 2009;Abstract LBA4509.
Efficacy Results from the ToGA Trial: A Phase III Study of Trastuzumab Added to Standard Chemotherapy in First-Line HER2- Positive Advanced Gastric Cancer Van Cutsem E et al. Proc ASCO 2009;Abstract LBA4509.
A Systematic Review on Economic Evaluation of Oxaliplatin Added Regimens as the Adjuvant Chemotherapy in Stage III Colon Cancer
 Original Article Mahidol University Journal of Pharmaceutical Science 2010; 37 3-4, 29-36 A Systematic Review on Economic Evaluation of Oxaliplatin Added Regimens as the Adjuvant Chemotherapy in Stage
Original Article Mahidol University Journal of Pharmaceutical Science 2010; 37 3-4, 29-36 A Systematic Review on Economic Evaluation of Oxaliplatin Added Regimens as the Adjuvant Chemotherapy in Stage
RECTAL CANCER CLINICAL CASE PRESENTATION
 RECTAL CANCER CLINICAL CASE PRESENTATION Francesco Sclafani Medical Oncologist, Clinical Research Fellow The Royal Marsden NHS Foundation Trust, London, UK esmo.org Disclosure I have nothing to declare
RECTAL CANCER CLINICAL CASE PRESENTATION Francesco Sclafani Medical Oncologist, Clinical Research Fellow The Royal Marsden NHS Foundation Trust, London, UK esmo.org Disclosure I have nothing to declare
Systemic Therapy for Metastatic Colorectal Cancer: Patterns of Chemotherapy and Biologic Therapy Use in US Medical Oncology Practice
 Original Contribution Systemic Therapy for Metastatic Colorectal Cancer: Patterns of Chemotherapy and Biologic Therapy Use in US Medical Oncology Practice By Gregory P. Hess, MD, MSc, Peter Feng Wang,
Original Contribution Systemic Therapy for Metastatic Colorectal Cancer: Patterns of Chemotherapy and Biologic Therapy Use in US Medical Oncology Practice By Gregory P. Hess, MD, MSc, Peter Feng Wang,
We are IntechOpen, the world s leading publisher of Open Access books Built by scientists, for scientists. International authors and editors
 We are IntechOpen, the world s leading publisher of Open Access books Built by scientists, for scientists 3,500 108,000 1.7 M Open access books available International authors and editors Downloads Our
We are IntechOpen, the world s leading publisher of Open Access books Built by scientists, for scientists 3,500 108,000 1.7 M Open access books available International authors and editors Downloads Our
Medicinae Doctoris. One university. Many futures.
 Medicinae Doctoris The Before and The After: Can chemotherapy revise the trajectory of gastric and esophageal cancers? Dr. David Dawe MD, FRCPC Medical Oncologist Assistant Professor Disclosures None All
Medicinae Doctoris The Before and The After: Can chemotherapy revise the trajectory of gastric and esophageal cancers? Dr. David Dawe MD, FRCPC Medical Oncologist Assistant Professor Disclosures None All
Cetuximab plus 5-FU/FA/oxaliplatin (FOLFOX-4) in the first-line treatment of metastatic colorectal cancer: a large-scale Phase II study (OPUS)
 Cetuximab plus 5-FU/FA/oxaliplatin (FOLFOX-4) in the first-line treatment of metastatic colorectal cancer: a large-scale Phase II study (OPUS) C Bokemeyer, E Staroslawska, A Makhson, I Bondarenko, JT Hartmann,
Cetuximab plus 5-FU/FA/oxaliplatin (FOLFOX-4) in the first-line treatment of metastatic colorectal cancer: a large-scale Phase II study (OPUS) C Bokemeyer, E Staroslawska, A Makhson, I Bondarenko, JT Hartmann,
GSK Medicine: Study Number: Title: Rationale: Study Period: Objectives: Indication: Study Investigators/Centers: Research Methods: Data Source
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The impact of operation center and the prognostic factors on the outcome of patients with stage II and stage III colorectal cancer
 Turkish Journal of Cancer Volume 38, No. 4, 28 175 The impact of operation center and the prognostic factors on the outcome of patients with stage II and stage III colorectal cancer ABDULLAH BÜYÜKÇELİK
Turkish Journal of Cancer Volume 38, No. 4, 28 175 The impact of operation center and the prognostic factors on the outcome of patients with stage II and stage III colorectal cancer ABDULLAH BÜYÜKÇELİK
How Old is Too Old for Chemotherapy in Early C olon Colon Cancer: Role of Geriatric Assessments Winson Y. Cheung, MD, MPH, FRCPC
 How Old is Too Old for Chemotherapy in Early Colon Cancer: Role of Geriatric Assessments Winson Y. Cheung, MD, MPH, FRCPC British Columbia Cancer Agency - Vancouver CAGPO - October 20, 2013 1 About Me
How Old is Too Old for Chemotherapy in Early Colon Cancer: Role of Geriatric Assessments Winson Y. Cheung, MD, MPH, FRCPC British Columbia Cancer Agency - Vancouver CAGPO - October 20, 2013 1 About Me
trial update clinical
 clinical trial update by John W. Mucenski, BS, PharmD, Director of Pharmacy Operations, UMPC Cancer Centers In order to provide the most up-to-date and efficacious care to their patients, oncologists must
clinical trial update by John W. Mucenski, BS, PharmD, Director of Pharmacy Operations, UMPC Cancer Centers In order to provide the most up-to-date and efficacious care to their patients, oncologists must
Adjuvant Treatment of Pancreatic Cancer in 2009: Where Are We? Highlights from the 45 th ASCO Annual Meeting. Orlando, FL, USA. May 29 - June 2, 2009
 HIGHLIGHT ARTICLE - Slide Show Adjuvant Treatment of Pancreatic Cancer in 2009: Where Are We? Highlights from the 45 th ASCO Annual Meeting. Orlando, FL, USA. May 29 - June 2, 2009 Muhammad Wasif Saif
HIGHLIGHT ARTICLE - Slide Show Adjuvant Treatment of Pancreatic Cancer in 2009: Where Are We? Highlights from the 45 th ASCO Annual Meeting. Orlando, FL, USA. May 29 - June 2, 2009 Muhammad Wasif Saif
Long Term Outcomes of Preoperative versus
 RESEARCH ARTICLE Long Term Outcomes of Preoperative versus Postoperative Concurrent Chemoradiation for Locally Advanced Rectal Cancer: Experience from Ramathibodi Medical School in Thailand Pichayada Darunikorn
RESEARCH ARTICLE Long Term Outcomes of Preoperative versus Postoperative Concurrent Chemoradiation for Locally Advanced Rectal Cancer: Experience from Ramathibodi Medical School in Thailand Pichayada Darunikorn
Disclosures. Colorectal Cancer Update GAFP November Risk Assessment. Colon and Rectal Cancer The Challenge. Issues in Colon and Rectal Cancer
 Disclosures Colorectal Cancer Update GAFP November 2006 Robert C. Hermann, MD Georgia Center for Oncology Research and Education Northwest Georgia Oncology Centers, PC WellStar Health System Marietta,
Disclosures Colorectal Cancer Update GAFP November 2006 Robert C. Hermann, MD Georgia Center for Oncology Research and Education Northwest Georgia Oncology Centers, PC WellStar Health System Marietta,
Oxaliplatin, Fluorouracil, and Leucovorin as Adjuvant Treatment for Colon Cancer
 original article Oxaliplatin, Fluorouracil, and Leucovorin as Adjuvant Treatment for Colon Cancer Thierry André, M.D., Corrado Boni, M.D., Lamia Mounedji-Boudiaf, M.D., Matilde Navarro, M.D., Josep Tabernero,
original article Oxaliplatin, Fluorouracil, and Leucovorin as Adjuvant Treatment for Colon Cancer Thierry André, M.D., Corrado Boni, M.D., Lamia Mounedji-Boudiaf, M.D., Matilde Navarro, M.D., Josep Tabernero,
Pharmacologyonline 1: (2010)
 THE EFFECT OF USING COMBINATION CHEMOTHERAPY IN COLORECTAL CANCER IN INDIA: A SINGLE INSTITUTE SURVEY Adiga Sachidananda*, Meena Kumari K**, Bairy KL***, Mohan Babu A**, Vadiraja BM+, Vidyasagar MS++ *Associate
THE EFFECT OF USING COMBINATION CHEMOTHERAPY IN COLORECTAL CANCER IN INDIA: A SINGLE INSTITUTE SURVEY Adiga Sachidananda*, Meena Kumari K**, Bairy KL***, Mohan Babu A**, Vadiraja BM+, Vidyasagar MS++ *Associate
4.3 Input parameters Patient
 Table of Content 1 Introduction -------------------------------------------------------------------------------------------------- 7 Summary -----------------------------------------------------------------------------------------------------------
Table of Content 1 Introduction -------------------------------------------------------------------------------------------------- 7 Summary -----------------------------------------------------------------------------------------------------------
Terapia neoadyuvante en cáncer de recto Estado del arte Mauricio Lema Medina MD Clínica de Oncología Astorga / Clínica SOMA - Medellín, Colombia
 Terapia neoadyuvante en cáncer de recto Estado del arte Mauricio Lema Medina MD Clínica de Oncología Astorga / Clínica SOMA - Medellín, Colombia Temario Generalidades Adyuvancia en colon y recto FU / Capecitabina
Terapia neoadyuvante en cáncer de recto Estado del arte Mauricio Lema Medina MD Clínica de Oncología Astorga / Clínica SOMA - Medellín, Colombia Temario Generalidades Adyuvancia en colon y recto FU / Capecitabina
Adjuvant treatment for stage III colon cancer
 ESMO Preceptorship Programme Rectal cancer Singapur November 2017 Adjuvant treatment for stage III colon cancer Andrés Cervantes Disclosures Consulting and advisory services, speaking or writing engagements,
ESMO Preceptorship Programme Rectal cancer Singapur November 2017 Adjuvant treatment for stage III colon cancer Andrés Cervantes Disclosures Consulting and advisory services, speaking or writing engagements,
ADJUVANT AND NEOADJUVANT MANAGEMENT OF COLORECTAL CANCER
 gastrointestinal tract and abdomen ADJUVANT AND NEOADJUVANT MANAGEMENT OF COLORECTAL CANCER Christina E. Bailey, MD, MSCI, Eduardo Vilar, MD, PhD, and Y. Nancy You, MD, MHSc In 2013, colorectal cancer
gastrointestinal tract and abdomen ADJUVANT AND NEOADJUVANT MANAGEMENT OF COLORECTAL CANCER Christina E. Bailey, MD, MSCI, Eduardo Vilar, MD, PhD, and Y. Nancy You, MD, MHSc In 2013, colorectal cancer
Outcome of rectal cancer after radiotherapy with a long or short waiting period before surgery, a descriptive clinical study
 Original Article Outcome of rectal cancer after radiotherapy with a long or short waiting period before surgery, a descriptive clinical study Elmer E. van Eeghen 1, Frank den Boer 2, Sandra D. Bakker 1,
Original Article Outcome of rectal cancer after radiotherapy with a long or short waiting period before surgery, a descriptive clinical study Elmer E. van Eeghen 1, Frank den Boer 2, Sandra D. Bakker 1,
Adjuvant Therapy for Stage II Colon Cancer: Prognostic and Predictive Markers
 927 Original Article Adjuvant Therapy for Stage II Colon Cancer: Prognostic and Predictive Markers Brian Vicuna, MD, and Al B. Benson III, MD, Chicago, Illinois Key Words Stage II, colon cancer, treatment,
927 Original Article Adjuvant Therapy for Stage II Colon Cancer: Prognostic and Predictive Markers Brian Vicuna, MD, and Al B. Benson III, MD, Chicago, Illinois Key Words Stage II, colon cancer, treatment,
Inequity in access to guideline-recommended colorectal cancer treatment in Nova Scotia, Canada
 Inequity in access to guideline-recommended colorectal cancer treatment in Nova Scotia, Canada André Maddison MSc, Yukiko Asada PhD, Robin Urquhart PhD(c), Grace Johnston PhD, Fred Burge MD MSc CAHSPR
Inequity in access to guideline-recommended colorectal cancer treatment in Nova Scotia, Canada André Maddison MSc, Yukiko Asada PhD, Robin Urquhart PhD(c), Grace Johnston PhD, Fred Burge MD MSc CAHSPR
JK Coller, S Korver, IA Ball, RJ Gibson, J Tuke, RM Logan, AM Richards, KR Mead, CS Karapetis, DM Keefe and JM Bowen
 JK Coller, S Korver, IA Ball, RJ Gibson, J Tuke, RM Logan, AM Richards, KR Mead, CS Karapetis, DM Keefe and JM Bowen Incidence of other toxicities in patients with severe gastrointestinal toxicity and
JK Coller, S Korver, IA Ball, RJ Gibson, J Tuke, RM Logan, AM Richards, KR Mead, CS Karapetis, DM Keefe and JM Bowen Incidence of other toxicities in patients with severe gastrointestinal toxicity and
Exploring and Validating Surrogate Endpoints in Colorectal Cancer. Center, Pittsburgh, USA
 Page 1 Exploring and Validating Surrogate Endpoints in Colorectal Cancer Tomasz Burzykowski, PhD 1,2, Marc Buyse, ScD 1,3, Greg Yothers PhD 4, Junichi Sakamoto, PhD 5, Dan Sargent, PhD 6 1 Center for Statistics,
Page 1 Exploring and Validating Surrogate Endpoints in Colorectal Cancer Tomasz Burzykowski, PhD 1,2, Marc Buyse, ScD 1,3, Greg Yothers PhD 4, Junichi Sakamoto, PhD 5, Dan Sargent, PhD 6 1 Center for Statistics,
Resectable locally advanced oesophagogastric cancer
 Resectable locally advanced oesophagogastric cancer Clinical Case Discussion Florian Lordick University Cancer Center Leipzig University Clinic Leipzig Leipzig, Germany esmo.org DISCLOSURES Honoraria for
Resectable locally advanced oesophagogastric cancer Clinical Case Discussion Florian Lordick University Cancer Center Leipzig University Clinic Leipzig Leipzig, Germany esmo.org DISCLOSURES Honoraria for
The effect of surgeon volume on procedure selection in non-small cell lung cancer surgeries. Dr. Christian Finley MD MPH FRCSC McMaster University
 The effect of surgeon volume on procedure selection in non-small cell lung cancer surgeries Dr. Christian Finley MD MPH FRCSC McMaster University Disclosures I have no conflict of interest disclosures
The effect of surgeon volume on procedure selection in non-small cell lung cancer surgeries Dr. Christian Finley MD MPH FRCSC McMaster University Disclosures I have no conflict of interest disclosures
EVIDENCE IN BRIEF OVERALL CLINICAL BENEFIT
 information aligned with the expected toxicity profile of panitumumab, which is well-known as panitumumab is already used as a third-line therapy for patients with mcrc. perc also noted QoL did not deteriorate
information aligned with the expected toxicity profile of panitumumab, which is well-known as panitumumab is already used as a third-line therapy for patients with mcrc. perc also noted QoL did not deteriorate
* * * * Supplementary Figure 1. DS Lv CK HSA CK HSA. CK Col-3. CK Col-3. See overleaf for figure legend. Cancer cells
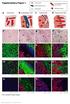 Supplementary Figure 1 Cancer cells Desmoplastic stroma Hepatocytes Pre-existing sinusoidal blood vessel New blood vessel a Normal liver b Desmoplastic HGP c Pushing HGP d Replacement HGP e f g h i DS
Supplementary Figure 1 Cancer cells Desmoplastic stroma Hepatocytes Pre-existing sinusoidal blood vessel New blood vessel a Normal liver b Desmoplastic HGP c Pushing HGP d Replacement HGP e f g h i DS
Patterns of HIV testing among Ontario physicians, 2006
 Patterns of HIV testing among Ontario physicians, 2006 Claudia Rank, Robert S. Remis, Carol Swantee and Keyi Wu HIV Laboratory, Ontario Agency for Health Protection and Promotion Canadian Association of
Patterns of HIV testing among Ontario physicians, 2006 Claudia Rank, Robert S. Remis, Carol Swantee and Keyi Wu HIV Laboratory, Ontario Agency for Health Protection and Promotion Canadian Association of
Appendix E - Summary form Oxaliplatin and capecitabine for the adjuvant treatment of colon cancer table of consultee comments
 Oxaliplatin and capecitabine for the adjuvant treatment of colon cancer table of consultee comments Section Consultees Comments Action Objective Roche RCP RCP As far as capecitabine is concerned, the objective
Oxaliplatin and capecitabine for the adjuvant treatment of colon cancer table of consultee comments Section Consultees Comments Action Objective Roche RCP RCP As far as capecitabine is concerned, the objective
The effect of immune conditions on pesticide use and the risk of non- Hodgkin lymphoma. Manisha Pahwa, OCRC CARWH Conference June 2, 2012
 The effect of immune conditions on pesticide use and the risk of non- Hodgkin lymphoma Manisha Pahwa, OCRC CARWH Conference June 2, 2012 Authors Shelley A. Harris, University of Toronto, Occupational Cancer
The effect of immune conditions on pesticide use and the risk of non- Hodgkin lymphoma Manisha Pahwa, OCRC CARWH Conference June 2, 2012 Authors Shelley A. Harris, University of Toronto, Occupational Cancer
A DISSERTATION SUBMITTED TO THE FACULTY OF THE GRADUATE SCHOOL OF THE UNIVERSITY OF MINNESOTA BY. Helen Mari Parsons
 A Culture of Quality? Lymph Node Evaluation for Colon Cancer Care A DISSERTATION SUBMITTED TO THE FACULTY OF THE GRADUATE SCHOOL OF THE UNIVERSITY OF MINNESOTA BY Helen Mari Parsons IN PARTIAL FULFILLMENT
A Culture of Quality? Lymph Node Evaluation for Colon Cancer Care A DISSERTATION SUBMITTED TO THE FACULTY OF THE GRADUATE SCHOOL OF THE UNIVERSITY OF MINNESOTA BY Helen Mari Parsons IN PARTIAL FULFILLMENT
This clinical study synopsis is provided in line with Boehringer Ingelheim s Policy on Transparency and Publication of Clinical Study Data.
 abcd Clinical Study for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis which is part of the clinical
abcd Clinical Study for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis which is part of the clinical
