REGULATING MAMMOGRAMS
|
|
|
- Abigayle Morton
- 5 years ago
- Views:
Transcription
1 REGULATING MAMMOGRAMS The Harvard community has made this article openly available. Please share how this access benefits you. Your story matters Citation REGULATING MAMMOGRAMS (1998 Third Year Paper) Citable link Terms of Use This article was downloaded from Harvard University s DASH repository, and is made available under the terms and conditions applicable to Other Posted Material, as set forth at nrs.harvard.edu/urn-3:hul.instrepos:dash.current.terms-ofuse#laa
2 # Food and Drug Law Professor Hutt Winter 1998 REGULATING MAMMOGRAMS Breast cancer is perhaps the most feared disease for women. Although other illnesses might be more deadly from a statistic point of view, women seem more concerned about breast cancer (in fact, breast cancer is the second leading cause of cancer mortality in women). This fear has resulted in an enormous preoccupation with the disease, and in particular, with diagnosing and treating the cancer. And [s]ince currently there is no cure for breast cancer or method to prevent it from occurring, the key to saving women s lives is the early detection of the disease, when treatment is the most effective and survival rates are best. 1 Mammography is widely used as a cancer detection device; unfortunately, it is often misused or performed inadequately. The need for proper supervision and regulation is of a concern to every woman. I. BREAST CANCER AND MAMMOGRAPHY In 1997 approximately 180,00 new cases of breast cancer were detected among women in the United States, and around 45,000 women died from the disease. 2 Breast cancer develops in stages; the ability to detect cancer in those first stages is vitally important to a woman s prognosis Nih Recommendations on Mammograms: 1-learings Belore the Subcomm. on Labor, Health and Human Services and Education of the Senate Comm. on Appropriations, in 1997 WL (1997) (statement of Susan J. Blumenthal, Deputy Asst. Sec. for Health, Asst. Surgeon General, U.S. Public Health Service) [hereinafter Hearings 1. 2 See American Cancer Society. Breast Cancer information (as of Jan. 23, 1998), available in html. 1
3 and tbture. Mammography is one of the most important detection tools currently available to women and doctors. In fact, many physicians and experts have agreed that mammography is the best method of detecting breast cancer at its earliest stages. 3 4 There are two types of mammograms. A screening mammogram (which is the type discussed in this paper) is an x-ray of the breast used to detect breast cancer in women who have no signs of cancer. It usually involves two x-rays of each breast. Diagnostic mammograms, by contrast, are used to diagnose unusual breast changes and used to evaluate abnormalities detected on a screening mammogram. About 23.5 million mammograms were performed in the U.S. in 1994, costing approximately $2.5 billion. 5 Mammography is a particularly complex examination. It is one of the hardest radiological examinations to do properly because the quality of the image is more dependent on accurate equipment performance and correct processing of film than other types of x-ray exams.,, 6 See Judith Willis, Why Women Don 1 (Jet Mammograms (And Why They Should), FDA CON5U R, May 1987, at 5. Mammography can detect a breast tumor as much as two years before lump can be felt. John Schwartz, FDA Seeks to Improve Quality of Mammography; Rules Set Mandatory Verification System, WASHINGTON PosT, Dec. 2, 1993, at A3. At the time of the act s enactment, the National Cancer Institute estimated that annual screening of women over 50 can reduce breast cancer deaths by about a third. See Leslie Miller, Mammogram Centers Must Meet Standards, USA TODAY, Sept. 30, 1994, at D6 (Life section). See generally, National Cancer Institute, Cancer Facts: Detection (as of Jan. 23, 1998), available in nih.gov/clinpdq/detectioniscreening Mammograms html. Rich Weiss, Mammography Centers Flunking Inspections, WASH- INGTON POST, Nov. 28, 1995, at 5 (Health section). 6 HHS Issues Mammogram Guidelines That Seek Better Tests, Faster Results, WASHINGTON POST, Oct. 20, 1994, at AS. 2
4 Robert Schmidt, the radiologist in charge of breast imaging at the University of Chicago Hospital, estimates that probably more than ten percent and as high as thirty percent of a mammogram s 7 findings may be wrong - due to human error. Because mammography is so important, the federal government has felt an obligation to ensure as many women have one as possible. Despite the fact that the exam is vital, many women do not have the exam. The National Cancer institute estimates that only half of the women over 50 (the group at greatest risk of contracting cancer) receive mammograms on a regular basis. 8 Ii. DEVICE APPROVAL AND REGULATION There are two primary areas in which mammography has been heavily regulated by the FDA. One such area is the FDA s regulation of radiological/mammography devices under the medical device program of its Center for Devices and Radiological Health. 9 Until recently, the regulation of mammography as a medical device received little attention. Because the machines used are the same as the traditional x-ray machines that are used in a wide variety of medical settings, regulation of the actual equipment seemed to be an afterthought. Yet, regular mammography is not full-proof It misses 15-20% of cancers and 80% Michael Unger, Digital Mammography Lifts Ilopes]br Better Detection, NEWS- DAY, Dec. 19, 1995, at B28. S Sandy Rovner, Standards Set/br Mammograms, WASHINGTON POST, Nov , at 18 (Health section). This section primarily deals with diagnostic devices sold to and used by physicians. There is also a new device on the market, which just recently received FDA approval, which helps a woman perform breast self examinations. The Sensor Pad (designed to increase sensation during breast self examinations) can be sold over the counter originally, in 1995, it was approved by the FDA as a prescription product. See generally, Bruce Ingersoll, FDA Clears Device for Breast Exams Alter Long Wait, WALl. STREET JOURNAL, Nov. 18, 1997, at B16. 3
5 of the lesions found by mammography turn out to be benign. Thus, new technologies and methods are needed to enhance cancer detection. Advances in science and technology have created more complex and novel machines. These new machines must be approved for mammography use by the FDA before they can be marketed as mammography equipment. And the FDA does enforce its rules. In 1997, for example, Biopsys Medical Inc. was forced to stop marketing its product as a cancer detection device after receiving a warning letter from the FDA. In fact, these machines are not truly new. Some employ technology that has been utilized in other medical settings. Thus, these devices have already been approved for other uses by the FDA. Yet, equipment that is marketed specifically as a mammography device must first be approved by the agency. However, this does not preclude manufacturers from creating these devices. Furthermore, many physicians use radiological equipment to perform a mammogram that was originally designed for another use. What we regulate is the marketing of the product by the manufacturer. We don t regulate how doctors use the product once it s out on the market said 12 an FDA spokeswoman. This practice is similar to doctors prescribing drugs that were originally designed for one use to treat another completely different ailment, even though the manufacturer never marketed the drug for that purpose. Thus, radiological devices do not necessarily need to 10 Hearings, supra note 1. See Barbara Marsh, l3iopsys Medical Agrees to Alter Promotion of Its Main Product, Los ANGELES TIMEs, Feb. 25, 1997, at D Dolores Kong, Better Imaging SecAs to Limit Breast Biopsies, BOSTON GLOBE, Apr. 22, 1996, at 25. 4
6 be approved as mammography devices by the FDA if the manufacturer has no intention of marketing its devices as mammography devices. However, some manufacturers do want to be able to market their devices to more physicians and hospitals for use as a mammographic machine. As mentioned above, these new devices first need to be approved by the FDA. Approval of such devices has recently received much attention. The new advances are celebrated by physicians and patients alike because they improve the quality of the image taken. This improvement in quality can both save lives (by detecting cancer earlier) and money (by eliminating costly further tests when a mammogram picture is unclear or inconclusive). And of course, manufacturers are asking for approval to be able to market the device to a wider range of customers. Ultrasound is being used as a possible backup to mammography. In this procedure, the machine builds an image of the breast by sending high frequency sound waves through the body. 3 The FDA approved the application of Advanced Technology Laboratories (ATL) to market and use ultrasound as a breast cancer diagnostic tool. 4 In its 18 month study of 1,000 women, ATL found that using ultrasound to supplement regular mammograms reduced the number of biopsies 13 See John Schwartz, Feds OK Ultrasound Test of Breast Lumps, PORT- LAND OREGoNIAN, Apr. 13, 1996, at Al. Id. ATL was the first company to actually submit clinical data to show the effectiveness and safety of the procedure, in order to market the device as a breast cancer diagnostic tool. See Kong, supra note 12. Although most responses have been positive, ultrasound has its critics. Daniel Kopans, director of the breast imaging division of the Massachusetts General Hospital, simply stated that the approval of the ultrasound machine was a great marketing ploy. Kong. supra note 12. Other doctors are more optimistic about magnetic resonance imaging and digital mammography. See id 5
7 by a third. 15 ATL s machines give an image sharp enough to allow doctors to distinguish between benign and cancerous solid lumps. 6 The device could also reduce the rate of false positives from 75% to ATL was forced to seek pre-market approval, rather than conform to a less stringent standard for existing technology. 8 Magnetic resonance imaging creates images from signals generated by the excitation of nuclear particles in a magnetic field. 19 MIRI has emerged as an exciting development because it offers an important advantage: higher definition images without radiation. 20 It also is more successful in taking images of younger women, whose breast tissue is usually too thick to produce a readable mammogram. Although the device received FDA approval in 1996, the procedure currently is extremely costly. 21 Advanced Mammography Systems, Inc. is currently selling this device. However, due to its high cost, the FDA will only allow the company to market the device for diagnosis, not for screening, purposes See Warren King, Study: Ultrasound Boosts Accuracy of Mammogram, NEW ORu ANs TIMES-Picayune, Feb. 4, 1996, at AIO. The elimination of unnecessary biopsies is important. In biopsies, a portion of the breast is removed and analyzed. Although a biopsy is the only 100% certain way to determine the status of an abnormality in the breast (see Kong, supra note 12), many biopsies are performed needlessly. About 70-80% of tissue from these biopsies turns out to be benign. Biopsies are also painful and costly for the patient. See generally, King, supra. 16 Schwartz, supra note 13. Id. 18 Kong,,supra.note Hearings, supra note I. 20 Eric Convey, Device to Diagnose Breast Cancer OK d, BOSTON HERAI.D, Mar. 1, 1996, at See generally, id 22 See id. 6
8 Digital mammography is perhaps the most advanced improvement to be developed. Prior to 1995, full breast digital imaging was expensive and inadequate. However, the machines have been refined. 23 The new machines use no film. They take an image of the full breast and display that on a high definition computer screen. That image can then be saved or transmitted elsewhere is necessary. Digital enhancement of the image allows a physician to detect abnormalities that are normally difficult to recognize on a film screen. 24 Newer technology has also enabled manufacturers to drop the price of this type of machine. Full breast digital imaging machines were being tested in Northeast areas. A New York company had, as of December 1997, applied for FDA approval to begin marketing the first full-breast digital imaging system. 25 Finally, positron-emission tomography that produces molecular images is an experimental method that seeks to distinguish cancer cells from normal tissue based on differences in their metabolism and other characteristics. 26 Currently, many of these new mammography machines are being studied or marketed. However, truly accurate and long term studies will ultimately determine the safety and 23 See generally, Michael Unger, Digital Mammography Li/Is hopes for Better Detection, NEWSDAY, Dec. 19, 1995, at B See id 25 See Michael Unger, led Approval Sought/br Digital lmaging, NEWSDAY, Dec. 3, 1997, at A60. In fact, the FDA has delineated guidelines for such manufacturers seeking marketing clearance. See Information for Manufacturers Seeking Marketing Clearance of Digital Mammography Systems (updated Aug. 13, 1996), available in In general, manufacturers must have a system of quality assurance tests in place to ensure that the system is operating properly. See id 26 Susan Okie, More Women are Getting Mammograms, WASHINGTON POST, Jan. 21, 1997, at 7 (Health section). 7
9 effectiveness of these methods. In fact, one study will compare various breast imaging devices over a period of five years. 27 III. PERFORMANCE REGULATION The FDA has also regulated mammography by attempting to regulate how well mammograms are performed - by regulating the facilities, doctors, and equipment that are utilized when mammograms are given. Although recent events and laws have focused attention on the FDA s role in regulating mammograms, the FDA s involvement spans more than just a decade or two. The agency first become interested in mammography in 1974, after a report came out measuring the radiation exposures to women from mammography techniques. 28 Thus, the following year the Center for Devices and Radiological Health began the Breast exposure Nationwide Trends program, its objective being to locate facilities giving excessively high radiation exposures and to assist them in reducing the exposures. 29 Due to the government s and the manufacturers involvement, radiation exposure decreased. Attention then focused on the quality of the mammogram s 27 See Helen Bond, New Technology Makes Breast Cancer Easier to Find, DALLAS MORNING NEws, Oct. 27, 1997, at C3. 28 The report was done by Henry Bicehouse, a Pennsylvania state inspector. The report showed that in a few cases, the level of radiation the woman was exposed to was extremely high. See Marian Segel, Mammography Facilities Must Meet Quality Standards, 28 FDA CoNSUMER 8 (1994). 29 ld. 8
10 images. In 1985, the FDA began using a new phantom 30 to conduct a nationwide mammographic survey. This study demonstrated the poor quality of some mammograms, due more to human error than to inaccurate mammographic devices. 31 The problem was handed over to the American College of Radiology (ACR). This prompted ACR to establish its own voluntary accreditation program. At the time, the federal government was also establishing criteria for facilities that wanted to receive Medicare reimbursement. In developing a new system, the government in a sense merged these two programs to create the governing law in place today. Before the early I 990s, FDA regulation consisted primarily of medical device regulation. Yet there remained a problem - human error. All too often, mammograms do not reflect their pull [sic] potential as preventative tools because equipment, physicist, technologist, or radiologist error(s) compromise quality and accuracy., 32 Thus, at the urging of Senator Barbara Mikulski and Representative John Dingell, in 1992 Congress passed a law that would allow the FDA to regulate mammography facilities. The regulations created the first such mandatory national program,. 33 In general, the act, named the Mammography Quality Standards Act 34 (MQSA), 30 A phantom is a plastic device embedded with objects of varying materials and size that is used to evaluate image quality. The device is x-rayed as though it were a breast, and the image score is determined by the number of objects picked up by the x-ray film. Id. 31 See id 32 Suzanne V. Cocea, Who s Monitoring the Quality of Mammograms? The Mammography Quality Standards Act of 1992 ( 7 ould Finally Provide theasnwer, 19 AM. J.L. & MED. 313, 315 (1993). 33 John Schwartz, FDA Seeks to Improve Quality a/mammography; Rules Set Mandatory Verification System, WASHINGTON POST, Dec. 2, 1993, at A3. Mammogaphy quality has also been reguklated at the provate and sta5e level. See Cocca, supra note 32, at
11 creates quality control standards and a certification system for most of the nation s mammography facilities. The Act s enactment made mammography the only diagnostic imaging test to be federally regulated for quality. 35 The final rules were announced at the end of 1997 to much publicity. 36 The implementation of MQSA is delineated into two main areas: accreditation and quality 37 standards/certification. The first part of this regulation discusses the procedures whereby an entity can apply to become a FDA-approved accreditation body. The second part established minimum standards to which a mammography facility must adhere of the regulation lays out specific procedures by which an entity can become accredited. Most importantly, the entity s application submitted to the FDA must discuss the applicant s accreditation review and decision-making process. The following section ( 900.4) delineates certain codes of conduct and responsibilities to which the accreditation body must adhere. The FDA is given the authority to annually evaluate the performance of the accreditation 42 U.S.C. 263(b). It is estimated that the FDA will spend about $26 million in fiscal year 1997 to implement the act. See Hearings, supra note I. Fran Jordan, Quality Mammograms, WASHINGTON POST, Jan. 17, 1995, (Health section) at See Clintons Use Radio to Urge Women to Get Mammograms; New Rules are Announced On Breast-Cancer Testing, BALTIMORE SUN, Oct. 26, 1997, at A3. Implementation is set forth in 21 C.F.R. 900 et seq. (1997). 38 5ee2l C.F.R
12 body. 39 As of 1995, there were four approved accreditation bodies: the ACR, Iowa, California, and Arkansas. 40 The rules governing quality standards can be divided into three specific areas: personnel, practices, and equipment. All mammography facilities must obtain a certificate issued by the FDA to operate lawfully in the U.S. 4 To receive a certificate, trhe facility must demonstrate that it is accredited by an approved body, meets the quality standards for equipment, personnel, procedures, and quality control practices, and have a survey by a quakified medical physicist lists the quality standards applicable to pesonnel,equipment, medical records, and quality assurance. Interpreting physicians, radiologic technologists, and medical physicists all must have adequate schooling and experience. 43 In addition, the equipment used muist be specifically designed for mammography. 44 Facilities also need to prepare written reports of the results of each mammography examination performed. 45 And finally, mammography facilities will be required to set up quality assurance programs - to ensure that mammograms are clear and to properly follow up cases. 46 In order to enforce these rules, the FDA (or an authorized inspector) See 21 C.F.R Florence Houn et al., The Mammography Quality Standard Act of 1992: History and Process, 50 FooD & DRUG L.J. 485, (1995). 41 See 21 C.F.R See Cocea, supra note 32, at 331 See 21 C.F.R (a). 2l C.F.R (b). 43 5ee 21 C.F.R (c). 46 5ee 21 C.F.R (d-f). 11
13 will annually inspect the facilities. The FDA can suspend or revoke a facility s certificate is it finds that the facility has violated these rules. 47 Facilities had until October 1994 to cpmply with the regulations. The government has taken the MQSA and its rules very seriously. At its inception, a government hotline telephone number was set up to provide the names and locations of certified facilities to interested women. 48 And the FDA has even published a publication to help mammography facilities comply with the requirements of MQSA. 49 There were high hopes when the standards were first passed. Others feared that small facilities and businesses would be forced to close due to the new rules. 50 The success of the standards seemed to be apparent in the years after the law was passed. In a very influential report, the General Accounting Office released a study that indicated that the program was a success. 5 According to this report, about four percent of the 10,000-11,000 clinics closed rather than comply with the new rules, and in 95% of those situations another clinic was within ee21 C.F.R See Mammogram Hot Line Starts Monday, CHICAGO TRIBUNE, May 14, 1995, Mammography Matters is a quarterly publication of the Division of Mammography Quality and Radiation Programs, Center for Devices and Radiological Health. so For some additional detailed information, refer to Economic Impact Analysis of Regulations Under the Mammography Quality Standards Act of 1992 (Oct. 7, 1997), available in html. Senator Nancy Kassebaum had requested the report in response to concerns that the FDA rules would put many clinics out of business and would cost too much. tinder Examination, 12 MED. MALPRACTICE L. & STRATEGY 8 (1995). The GAO report was released in November of 1995, 12
14 miles. 52 Furthermore, although 3500 of the facilities that were investigated failed to comply with one of the rules, most of them passed on subsequent inspections. 53 After this report was released, everyone viewed the law as a success: We have delivered to American women better quality mammograms and we have ensured that no matter whether a woman lives in a big city or a small town, she has access to this lifesaving service, Donna Shalala, the Secretary of Health and Human Services, stated. 54 There have been some people who have felt otherwise the report also states that one-third of the facilities originally certified failed the subsequent investigation. 55 As of June 1995, 1,843 facilities had been inspected. 601 facilities had violations, and 119 of these were serious violations - usually involving unqualified personnel. 56 In the latest GAO report, the agency found that although some problem areas do exist, overall there is growing compliance with the mammography standards See Under Examination, 13 MED. MAI.PRACTICE L. & STRATEGY 8 (1995). 53 5ee id US Reports a Healthier Mammogram Picture, WASHINGTON POST, Nov. 2, 1995, at A3. The report was also used for larger, political reasons. Representative Dingell seized the report as evidence that Congress should be cautious in heeding calls by FDA critics to revamp - or even privatize - the agency. US Reports a Healthier Mammogram Picture, WASHINGTON POST, Nov. 2, 1995, at A3. 55 See Rich Weiss, Mammography Centers Flunking Inspections, WASHING- TON POST, Nov. 28, 1995, at 5 (Health section). 56 See id See U.S. General Accounting Office, Report to Congressional Committees, FDA s Mammography Inspections: While Some Problems Need Attention, Facility Compliance Is Growing, GAO/HEHS (Jan. 1997). GAO found the following problems: more consistent reporting is needed, the procedures for assessing image quality need strengthening, and the enforcement process does not ensure timely correction of deficiencies. See id. 13
15 IV. PROBLEMATIC ISSUES Although regulation of mammography has largely proved to be beneficial, some problems still remain. In enforcing the MQSA, the FDA faced a (all too familiar) problem of implementation. With limited resources and labor, the FDA was required to inspect and certify thousands of facilities. At the time, FDA Commissioner David Kessler was optimistic: It is an all-out effort. I have no doubt the goal is achievable. 58 To help achieve its goal, the FDA invited the help of other entities. The FDA has relied on its decades-long alliance with state departments. 59 The FDA does do some of its own investigations, however it primarily trains and provides technical expertise to state inspectors. In addition, the standards adopted under the MQSA were very similar to the voluntary accreditation standards of the American College of Radiology in the 1980s. Because nearly 60% of the facilities adhered to those standards, the FDA s undertaking seemed less daunting. A further interesting problem is how the issue of scientific certainty is dealt with in the regulation of mammography. To date, the FDA has not issued any guidelines recommending who should take annual mammograms or when women should start getting mammograms. The FDA has (wisely?) stayed out of this debate. The problem is simple: when should women start having mammograms? The solution is far from clear. The National Cancer Institute (NCI) and the American Cancer Society have been debating the issue for some time. Initially, the NCI recommended that women start having mammograms every year or two at age 40. However, around 1993 it changed its position, stating that women under 50 should look at all the facts and Schwartz, supra note 13. John Henkel, FDA, States Collaborate For Sa ty s Sake, 30 FDA CONSUMER 27 (1996). 14
16 decide for themselves. This new position conflicts with the views held both by the American Cancer Society and the American Medical Association. 60 This problem is indicative of the inevitable uncertainty of science. Thus far there is no way to prove definitely that women under 50 should or should not have annual mammograms. Recommendations are, at best, made out of educated guesses. Further complicating matters, there is some debate regarding the safety of mammograms and their effectiveness in detecting cancer in young women. 61 And in a telling policy choice, the FDA has decided to not address the (real) issue. A final point to mention is the amazing coverage and attention mammography has received recently. As we all know, the FDA has a limited budget. Devoting resources to one problem siphons funds away from other worthy causes. Yet, the FDA has decided that mammography in particular, and breasts in general, should be a priority item. Even the Clinton administration addressed the issue as part of its focus on women and health. The broad regulation of mammography, together with the immense attention given to breast implants, suggests that the FDA just might be breast obsessed. Has pressure from the public and from women s groups forced the FDA to make choices it normally would have shied away from? Or is the FDA simply responding to a killer disease that has struck down many women and that can be preventable/treatable if caught early enough? 60 See John C. Goodman, Choice Not Qffered]br Mammograms, WALL STREET JOURNAL, Feb. 3, 1997, at A14; American Cancer Society, Workshop on Guidelines for Breast Cancer Detection, Mar. 7-9, 1997, available in html. 61 The link between mammograms and the potential adverse effects of radiation exposure is still being studied and debated. 15
Screening Mammograms: Questions and Answers
 CANCER FACTS N a t i o n a l C a n c e r I n s t i t u t e N a t i o n a l I n s t i t u t e s o f H e a l t h D e p a r t m e n t o f H e a l t h a n d H u m a n S e r v i c e s Screening Mammograms:
CANCER FACTS N a t i o n a l C a n c e r I n s t i t u t e N a t i o n a l I n s t i t u t e s o f H e a l t h D e p a r t m e n t o f H e a l t h a n d H u m a n S e r v i c e s Screening Mammograms:
GAO MAMMOGRAPHY. Current Nationwide Capacity Is Adequate, but Access Problems May Exist in Certain Locations. Report to Congressional Requesters
 GAO United States Government Accountability Office Report to Congressional Requesters July 2006 MAMMOGRAPHY Current Nationwide Capacity Is Adequate, but Access Problems May Exist in Certain Locations GAO-06-724
GAO United States Government Accountability Office Report to Congressional Requesters July 2006 MAMMOGRAPHY Current Nationwide Capacity Is Adequate, but Access Problems May Exist in Certain Locations GAO-06-724
Mammography. What is Mammography? What are some common uses of the procedure?
 Mammography What is Mammography? Mammography is a specific type of imaging that uses a low-dose x-ray system to examine breasts. A mammography exam, called a mammogram, is used to aid in the early detection
Mammography What is Mammography? Mammography is a specific type of imaging that uses a low-dose x-ray system to examine breasts. A mammography exam, called a mammogram, is used to aid in the early detection
HISTORY OF MQSA AND ACR
 HISTORY OF MQSA AND ACR DEBORAH THAMES R.T. (R)(M)(QM) WHY MQSA? In the United States, there was a lack of standards in mammography imaging. Reporting Imaging Type of imaging screening/diagnostic Equipment
HISTORY OF MQSA AND ACR DEBORAH THAMES R.T. (R)(M)(QM) WHY MQSA? In the United States, there was a lack of standards in mammography imaging. Reporting Imaging Type of imaging screening/diagnostic Equipment
F r e q u e n t l y A s k e d Q u e s t i o n s. Mammograms
 Mammograms Q: What is a mammogram? A: A mammogram is a safe, low-dose x-ray exam of the breasts to look for changes that are not normal. The results are recorded on x-ray film or directly into a computer
Mammograms Q: What is a mammogram? A: A mammogram is a safe, low-dose x-ray exam of the breasts to look for changes that are not normal. The results are recorded on x-ray film or directly into a computer
Guidance for States considering legislation
 ACOG STATE LEGISLATIVE TOOLKIT: Breast Density and Screening Mandates Guidance for States considering legislation These legislative mandates are problematic: No reliable, standardized method for assessing
ACOG STATE LEGISLATIVE TOOLKIT: Breast Density and Screening Mandates Guidance for States considering legislation These legislative mandates are problematic: No reliable, standardized method for assessing
The undersigned submit this petition under 42 U.S.C. 263b of the Mammogram Quality Standards Advisory Act of 1992 as amended and renewed in the
 The undersigned submit this petition under 42 U.S.C. 263b of the Mammogram Quality Standards Advisory Act of 1992 as amended and renewed in the Mammogram Quality Standards Reauthorization Act of 1998 and
The undersigned submit this petition under 42 U.S.C. 263b of the Mammogram Quality Standards Advisory Act of 1992 as amended and renewed in the Mammogram Quality Standards Reauthorization Act of 1998 and
Mammography. What is Mammography?
 Scan for mobile link. Mammography Mammography is a specific type of breast imaging that uses low-dose x-rays to detect cancer early before women experience symptoms when it is most treatable. Tell your
Scan for mobile link. Mammography Mammography is a specific type of breast imaging that uses low-dose x-rays to detect cancer early before women experience symptoms when it is most treatable. Tell your
Fact Sheet Flu Vaccine Crisis: The Administration s Response to Recommendations and Warnings
 REP. HENRY A. WAXMAN RANKING MINORITY MEMBER COMMITTEE ON GOVERNMENT REFORM U.S. HOUSE OF REPRESENTATIVES OCTOBER 18, 2004 Fact Sheet Flu Vaccine Crisis: The Administration s Response to Recommendations
REP. HENRY A. WAXMAN RANKING MINORITY MEMBER COMMITTEE ON GOVERNMENT REFORM U.S. HOUSE OF REPRESENTATIVES OCTOBER 18, 2004 Fact Sheet Flu Vaccine Crisis: The Administration s Response to Recommendations
Improving Methods for Breast Cancer Detection and Diagnosis. The National Cancer Institute (NCI) is funding numerous research projects to improve
 CANCER FACTS N a t i o n a l C a n c e r I n s t i t u t e N a t i o n a l I n s t i t u t e s o f H e a l t h D e p a r t m e n t o f H e a l t h a n d H u m a n S e r v i c e s Improving Methods for
CANCER FACTS N a t i o n a l C a n c e r I n s t i t u t e N a t i o n a l I n s t i t u t e s o f H e a l t h D e p a r t m e n t o f H e a l t h a n d H u m a n S e r v i c e s Improving Methods for
Session 2: The Role of Specialist Radiology Technologists
 Session 2: The Role of Specialist Radiology Technologists Louise M. Henderson, MSPH PhD Assistant Professor, Department of Radiology The University of North Carolina, Chapel Hill Overview Role of the technologist
Session 2: The Role of Specialist Radiology Technologists Louise M. Henderson, MSPH PhD Assistant Professor, Department of Radiology The University of North Carolina, Chapel Hill Overview Role of the technologist
Breast Tomosynthesis. What is breast tomosynthesis?
 Scan for mobile link. Breast Tomosynthesis Breast tomosynthesis is an advanced form of mammography, a specific type of breast imaging that uses low-dose x-rays to detect cancer early when it is most treatable.
Scan for mobile link. Breast Tomosynthesis Breast tomosynthesis is an advanced form of mammography, a specific type of breast imaging that uses low-dose x-rays to detect cancer early when it is most treatable.
Q: Why is breast cancer a big deal?
 I hate breast cancer. As a radiologist who specializes in breast imaging, my career is devoted to the detection and diagnosis of breast cancer. I am passionate about women s health and my goal is to find
I hate breast cancer. As a radiologist who specializes in breast imaging, my career is devoted to the detection and diagnosis of breast cancer. I am passionate about women s health and my goal is to find
now a part of Electronic Mammography Exchange: Improving Patient Callback Rates
 now a part of Electronic Mammography Exchange: Improving Patient Callback Rates Overview This case study explores the impact of a mammography-specific electronic exchange network on patient callback rates
now a part of Electronic Mammography Exchange: Improving Patient Callback Rates Overview This case study explores the impact of a mammography-specific electronic exchange network on patient callback rates
created by high-voltage devices Examples include medical and dental x-rays, light, microwaves and nuclear energy
 What is radiation? Radiation is energy emitted from a source, that travels through space and can penetrate matter. Listed below are two types that we are exposed to and contribute to our overall radiation
What is radiation? Radiation is energy emitted from a source, that travels through space and can penetrate matter. Listed below are two types that we are exposed to and contribute to our overall radiation
3 D. Digital. Mobile Mammography. Palmetto Health Baptist Breast Center Columbia, South Carolina
 3 D Digital Mobile Mammography Palmetto Health Baptist Breast Center Columbia, South Carolina METHOD OF PAYMENT Insurance will be filed at the time of the procedure if you have mammography coverage. If
3 D Digital Mobile Mammography Palmetto Health Baptist Breast Center Columbia, South Carolina METHOD OF PAYMENT Insurance will be filed at the time of the procedure if you have mammography coverage. If
The AAO- HNS s position statement on Point- of- Care Imaging in Otolaryngology states that the AAO- HNS,
 AAO- HNS Statement on Diagnostic Imaging Reimbursement for Otolaryngologist Head and Neck Surgeons (September 2014) The American Academy of Otolaryngology Head and Neck Surgery (AAO- HNS), with approximately
AAO- HNS Statement on Diagnostic Imaging Reimbursement for Otolaryngologist Head and Neck Surgeons (September 2014) The American Academy of Otolaryngology Head and Neck Surgery (AAO- HNS), with approximately
History of MQSA ACR creates testing procedures Oct Mammography Quality Standards Act Dec 1993 Interim Regulations Oct All mammog
 15 Years of MQSA, What works, what doesn t Jerry Soen, MS DABR History of MQSA 1990-9191 ACR creates testing procedures Oct. 1992 Mammography Quality Standards Act Dec 1993 Interim Regulations Oct. 1994
15 Years of MQSA, What works, what doesn t Jerry Soen, MS DABR History of MQSA 1990-9191 ACR creates testing procedures Oct. 1992 Mammography Quality Standards Act Dec 1993 Interim Regulations Oct. 1994
Mammography. The Lebanese Society of Obstetrics and Gynecology. Women s health promotion series
 The Lebanese Society of Obstetrics and Gynecology Women s health promotion series Mammography When breast cancer is diagnosed at an early stage it could be treated and the patient would have a high chance
The Lebanese Society of Obstetrics and Gynecology Women s health promotion series Mammography When breast cancer is diagnosed at an early stage it could be treated and the patient would have a high chance
Breast Cancer Screening
 Scan for mobile link. Breast Cancer Screening What is breast cancer screening? Screening examinations are tests performed to find disease before symptoms begin. The goal of screening is to detect disease
Scan for mobile link. Breast Cancer Screening What is breast cancer screening? Screening examinations are tests performed to find disease before symptoms begin. The goal of screening is to detect disease
Comments of the Patient, Consumer, and Public Health Coalition. Strengthening the Center for Devices and Radiological Health s 510(k) Review Process
 March 19, 2010 Division of Dockets Management (HFA-305) Food and Drug Administration 5630 Fishers Lane, Room 1061 Rockville, MD 20852 Comments of the Patient, Consumer, and Public Health Coalition on Strengthening
March 19, 2010 Division of Dockets Management (HFA-305) Food and Drug Administration 5630 Fishers Lane, Room 1061 Rockville, MD 20852 Comments of the Patient, Consumer, and Public Health Coalition on Strengthening
Henda s Law. Supplemental screening for women with dense breast tissue and increased risk
 . Henda s Law Supplemental screening for women with dense breast tissue and increased risk The 2011 Texas Legislature passed House Bill 2102 which is effective 1st September 2011. The law is informally
. Henda s Law Supplemental screening for women with dense breast tissue and increased risk The 2011 Texas Legislature passed House Bill 2102 which is effective 1st September 2011. The law is informally
AAPM Annual Meeting. ACR Accreditation Update in CT
 AAPM Annual Meeting ACR Accreditation Updates in CT, Ultrasound, Mammography and MRI: ACR Accreditation Update in CT Michael McNitt-Gray, PhD, DABR, FAAPM Professor, Department of Radiological Sciences
AAPM Annual Meeting ACR Accreditation Updates in CT, Ultrasound, Mammography and MRI: ACR Accreditation Update in CT Michael McNitt-Gray, PhD, DABR, FAAPM Professor, Department of Radiological Sciences
The American College of Radiology Breast Ultrasound Accreditation Program: Frequently Asked Questions (Revised: July 31, 2017)
 The American College of Radiology Breast Ultrasound Accreditation Program: Frequently Asked Questions (Revised: July 31, 2017) Table of Contents APPLICATION - GENERAL... 1 MOVED FACILITIES AND UNITS...
The American College of Radiology Breast Ultrasound Accreditation Program: Frequently Asked Questions (Revised: July 31, 2017) Table of Contents APPLICATION - GENERAL... 1 MOVED FACILITIES AND UNITS...
Mammography Services Guidebook
 Mammography Services Guidebook Learn about early detection for breast cancer. At Florida Hospital for Women, we re dedicated to helping women protect themselves against breast cancer. Breast cancer is
Mammography Services Guidebook Learn about early detection for breast cancer. At Florida Hospital for Women, we re dedicated to helping women protect themselves against breast cancer. Breast cancer is
Introduction. access site at:
 Radiology is changing. Referring physicians are looking for more value from imaging services. How do radiology practices achieve this? While cutting charges for imaging procedures provides some immediate
Radiology is changing. Referring physicians are looking for more value from imaging services. How do radiology practices achieve this? While cutting charges for imaging procedures provides some immediate
STATE OF MINNESOTA BOARD OF ANIMAL HEALTH
 This document is made available electronically by the Minnesota Legislative Reference Library as part of an ongoing digital archiving project. http://www.leg.state.mn.us/lrl/sonar/sonar.asp STATE OF MINNESOTA
This document is made available electronically by the Minnesota Legislative Reference Library as part of an ongoing digital archiving project. http://www.leg.state.mn.us/lrl/sonar/sonar.asp STATE OF MINNESOTA
NONE. Disclosures. Accreditation Update
 ACR Ultrasound Accreditation: Requirements and Pitfalls Presented to: American Association of Physicists in Medicine Presented by: Jennifer Walter RDMS,RVT, RT(R) ACR Quality & Safety August 03, 2016 Disclosures
ACR Ultrasound Accreditation: Requirements and Pitfalls Presented to: American Association of Physicists in Medicine Presented by: Jennifer Walter RDMS,RVT, RT(R) ACR Quality & Safety August 03, 2016 Disclosures
4/26/2013. Libia Lugo Drug Specialist San Juan District Office Investigations Branch
 Libia Lugo Drug Specialist San Juan District Office Investigations Branch The Food and Drug Administration (FDA) responsibilities extend to the 50 United States, the District of Columbia, Puerto Rico,
Libia Lugo Drug Specialist San Juan District Office Investigations Branch The Food and Drug Administration (FDA) responsibilities extend to the 50 United States, the District of Columbia, Puerto Rico,
Scintimammography. What is scintimammography?
 Scan for mobile link. Scintimammography Scintimammography uses small amounts of radioactive materials called radiotracers, a special camera and a computer to help investigate an abnormality discovered
Scan for mobile link. Scintimammography Scintimammography uses small amounts of radioactive materials called radiotracers, a special camera and a computer to help investigate an abnormality discovered
March 14, The Food and Drug Administration (FDA) is facing a serious crisis that has significant implications for the nation s health.
 March 14, 2007 An Open Letter to: Chairman Edward Kennedy and Senator Mike Enzi, Chairman John Dingell and Representative Joe Barton Members of the Senate Health, Education, Labor and Pensions Committee
March 14, 2007 An Open Letter to: Chairman Edward Kennedy and Senator Mike Enzi, Chairman John Dingell and Representative Joe Barton Members of the Senate Health, Education, Labor and Pensions Committee
The Radiology Aspects
 REQUIREMENTS FOR INTERNATIONAL ACCREDITATION OF BREAST CENTERS/UNITS The Radiology Aspects Miri Sklair-Levy, Israel RADIOLOGY GUIDELINES FOR QUALITY ASSURANCE IN BREAST CANCER SCREENING AND DIAGNOSIS Radiologists
REQUIREMENTS FOR INTERNATIONAL ACCREDITATION OF BREAST CENTERS/UNITS The Radiology Aspects Miri Sklair-Levy, Israel RADIOLOGY GUIDELINES FOR QUALITY ASSURANCE IN BREAST CANCER SCREENING AND DIAGNOSIS Radiologists
As Introduced. 132nd General Assembly Regular Session S. B. No
 132nd General Assembly Regular Session S. B. No. 121 2017-2018 Senator Eklund Cosponsors: Senators Yuko, Schiavoni A B I L L To amend sections 1751.62, 3923.52, and 3923.54 of the Revised Code to include
132nd General Assembly Regular Session S. B. No. 121 2017-2018 Senator Eklund Cosponsors: Senators Yuko, Schiavoni A B I L L To amend sections 1751.62, 3923.52, and 3923.54 of the Revised Code to include
Wardrobe by Paul Simon 0 IS THE NEW PINK. The Facts about Screening Mammography
 Wardrobe by Paul Simon 0 IS THE NEW PINK The Facts about Screening Mammography American Cancer Society Guidelines for Early Detection Screening Mammography Annually starting at age 40 CBE (Clinical Breast
Wardrobe by Paul Simon 0 IS THE NEW PINK The Facts about Screening Mammography American Cancer Society Guidelines for Early Detection Screening Mammography Annually starting at age 40 CBE (Clinical Breast
Breast cancer screening programme: France. Dr. Rosemary Ancelle-Park Department of health Office for chronic diseases
 Breast cancer screening programme: France Dr. Rosemary Ancelle-Park Department of health Office for chronic diseases French health care system General population of 65 Million GP clinics = 32 200 Radiology
Breast cancer screening programme: France Dr. Rosemary Ancelle-Park Department of health Office for chronic diseases French health care system General population of 65 Million GP clinics = 32 200 Radiology
College of American Pathologists
 College of American Pathologists Comments to the Food and Drug Administration on the draft guidance In Vitro Companion Diagnostics Devices October 12, 2011 College of American Pathologists 1350 I Street,
College of American Pathologists Comments to the Food and Drug Administration on the draft guidance In Vitro Companion Diagnostics Devices October 12, 2011 College of American Pathologists 1350 I Street,
August Monthly Call Agenda. Attendees: Teerith, Shahil, Steve, Carol, Emily, Abha, Jan, Marisa, Nancy, Dottie, and Melissa
 August Monthly Call Agenda Attendees: Teerith, Shahil, Steve, Carol, Emily, Abha, Jan, Marisa, Nancy, Dottie, and Melissa Federal Update: How Do You Measure Up? The latest edition of ACS CAN s annual report,
August Monthly Call Agenda Attendees: Teerith, Shahil, Steve, Carol, Emily, Abha, Jan, Marisa, Nancy, Dottie, and Melissa Federal Update: How Do You Measure Up? The latest edition of ACS CAN s annual report,
Clinical Applications of Emerging Tactile-Sensing Technologies
 Transcript Details This is a transcript of an educational program accessible on the ReachMD network. Details about the program and additional media formats for the program are accessible by visiting: https://reachmd.com/programs/clinicians-roundtable/clinical-applications-of-emerging-tactile-sensingtechnologies/2257/
Transcript Details This is a transcript of an educational program accessible on the ReachMD network. Details about the program and additional media formats for the program are accessible by visiting: https://reachmd.com/programs/clinicians-roundtable/clinical-applications-of-emerging-tactile-sensingtechnologies/2257/
Breast Imaging & You
 Breast Imaging & You What s Inside: Breast Imaging... 2 Digital Breast Tomosynthesis (DBT) mammograms... 4 Breast cancer screening... 6 Dense breast tissue... 8 Automated breast ultrasound (ABUS)... 9
Breast Imaging & You What s Inside: Breast Imaging... 2 Digital Breast Tomosynthesis (DBT) mammograms... 4 Breast cancer screening... 6 Dense breast tissue... 8 Automated breast ultrasound (ABUS)... 9
25 TEXAS ADMINSTRATIVE CODE (TAC) (x) Certification of Mammography Systems and Mammography Machines Used for Interventional Breast Radiography
 25 TEXAS ADMINSTRATIVE CODE (TAC) 289.230 Certification of Mammography Systems and Mammography Machines Used for Interventional Breast Radiography Texas Regulations for Control of Radiation (effective
25 TEXAS ADMINSTRATIVE CODE (TAC) 289.230 Certification of Mammography Systems and Mammography Machines Used for Interventional Breast Radiography Texas Regulations for Control of Radiation (effective
Screening Mammography in Primary Care Settings: Implications for Cost Access and Quality. October NTIS order #PB
 Screening Mammography in Primary Care Settings: Implications for Cost Access and Quality October 1991 NTIS order #PB92-182617 Screening Mammography in Primary Care Settings: Implications for Cost, Access
Screening Mammography in Primary Care Settings: Implications for Cost Access and Quality October 1991 NTIS order #PB92-182617 Screening Mammography in Primary Care Settings: Implications for Cost, Access
Mammography Quality Control: A Refresher
 Mammography Quality Control: A Refresher MATT WAIT, MS, DABR Objectives Attendees will re-familiarize themselves with the purpose of quality assurance and quality control Attendees will re-familiarize
Mammography Quality Control: A Refresher MATT WAIT, MS, DABR Objectives Attendees will re-familiarize themselves with the purpose of quality assurance and quality control Attendees will re-familiarize
Breast Cancer. What is breast cancer?
 Scan for mobile link. Breast Cancer Breast cancer is a malignant tumor in or around breast tissue. It usually begins as a lump or calcium deposit that develops from abnormal cell growth. Most breast lumps
Scan for mobile link. Breast Cancer Breast cancer is a malignant tumor in or around breast tissue. It usually begins as a lump or calcium deposit that develops from abnormal cell growth. Most breast lumps
Breast Imaging & You
 Breast Imaging & You What s Inside: Breast Imaging... 2 Digital Breast Tomosynthesis (DBT) mammograms... 4 Breast cancer screening... 6 Dense breast tissue... 8 Automated Breast Ultrasound (ABUS)... 9
Breast Imaging & You What s Inside: Breast Imaging... 2 Digital Breast Tomosynthesis (DBT) mammograms... 4 Breast cancer screening... 6 Dense breast tissue... 8 Automated Breast Ultrasound (ABUS)... 9
Survey of Mammography Facilities
 Survey of Mammography Facilities Summary: Survey completed? Yes Date completed: / / No Comments: Instructions: This survey is part of a study being conducted by [insert surveillance institution] breast
Survey of Mammography Facilities Summary: Survey completed? Yes Date completed: / / No Comments: Instructions: This survey is part of a study being conducted by [insert surveillance institution] breast
Breast Cancer Imaging Webcast October 21, 2009 Peter Eby, M.D. Introduction
 Breast Cancer Imaging Webcast October 21, 2009 Peter Eby, M.D. Please remember the opinions expressed on Patient Power are not necessarily the views of Seattle Cancer Care Alliance, its medical staff or
Breast Cancer Imaging Webcast October 21, 2009 Peter Eby, M.D. Please remember the opinions expressed on Patient Power are not necessarily the views of Seattle Cancer Care Alliance, its medical staff or
Submitted to the House Energy and Commerce Committee. Federal Efforts to Combat the Opioid Crisis
 STATEMENT FOR THE RECORD Submitted to the House Energy and Commerce Committee Federal Efforts to Combat the Opioid Crisis October 25, 2017 America s Health Insurance Plans 601 Pennsylvania Avenue, NW Suite
STATEMENT FOR THE RECORD Submitted to the House Energy and Commerce Committee Federal Efforts to Combat the Opioid Crisis October 25, 2017 America s Health Insurance Plans 601 Pennsylvania Avenue, NW Suite
WOMENCARE A Healthy Woman is a Powerful Woman (407) Mammography
 Mammography WOMENCARE A Healthy Woman is a Powerful Woman (407) 898-1500 Mammography is an X-ray technique used to study the breasts. It can help doctors find breast cancer at an early stage (when treatment
Mammography WOMENCARE A Healthy Woman is a Powerful Woman (407) 898-1500 Mammography is an X-ray technique used to study the breasts. It can help doctors find breast cancer at an early stage (when treatment
Highmark Privileging Application. 1. Complete this application if you provide any diagnostic
 Highmark Privileging Application INSTRUCTIONS 1. Complete this application if you provide any diagnostic imaging services. 2. Please complete a separate application for each physical location where imaging
Highmark Privileging Application INSTRUCTIONS 1. Complete this application if you provide any diagnostic imaging services. 2. Please complete a separate application for each physical location where imaging
Public Health Service Food and Drug Administration San Francisco District 1431 Harbor Bay Parkway Alameda, CA Telephone: 510/
 Department of Health and Human Services Public Health Service Food and Drug Administration San Francisco District 1431 Harbor Bay Parkway Alameda, CA 94502-7070 Telephone: 510/337-6700 WARNING LETTER December
Department of Health and Human Services Public Health Service Food and Drug Administration San Francisco District 1431 Harbor Bay Parkway Alameda, CA 94502-7070 Telephone: 510/337-6700 WARNING LETTER December
RE: Coverage of low-dose Computed Tomography (LDCT) lung cancer screening in Independent Diagnostic Testing Facilities (IDTFs)
 May 11, 2018 Tamara Syrek Jensen, JD Director, Coverage and Analysis Group Department of Health and Human Services Center for Medicare and Medicaid Services 7500 Security Boulevard, Mail Stop S3-02-01
May 11, 2018 Tamara Syrek Jensen, JD Director, Coverage and Analysis Group Department of Health and Human Services Center for Medicare and Medicaid Services 7500 Security Boulevard, Mail Stop S3-02-01
e-cigarette Regulation
 e-cigarette Regulation The Act prohibits the sale of electronic smoking devices and alternative nicotine products to minors, and requires child-resistant packaging for liquid nicotine containers. The Act
e-cigarette Regulation The Act prohibits the sale of electronic smoking devices and alternative nicotine products to minors, and requires child-resistant packaging for liquid nicotine containers. The Act
Via Electronic Submission. March 13, 2017
 APTQI 20 F Street, NW Suite #700 Washington, DC 20001 Phone: 202-507-6354 www.aptqi.com Via Electronic Submission Centers for Medicare & Medicaid Services Department of Health & Human Services Attention:
APTQI 20 F Street, NW Suite #700 Washington, DC 20001 Phone: 202-507-6354 www.aptqi.com Via Electronic Submission Centers for Medicare & Medicaid Services Department of Health & Human Services Attention:
CLINICAL GUIDELINES. Screening Mammography Guidelines
 CLINICAL GUIDELINES Screening Mammography Guidelines Paula George, M.D. and C. Todd Cunningham, M.D., Karen F. Goodhope, M.D., Valerie C. Reichert, M.D. Hayley Sheldon, M.D., Michelle Walters, D.O. 2/17/2016
CLINICAL GUIDELINES Screening Mammography Guidelines Paula George, M.D. and C. Todd Cunningham, M.D., Karen F. Goodhope, M.D., Valerie C. Reichert, M.D. Hayley Sheldon, M.D., Michelle Walters, D.O. 2/17/2016
Like others here today, we are very conflicted on the FDA s proposal on behind-thecounter medications, but thank you for raising the issue.
 Statement of Consumers Union William Vaughan, Senior Policy Analyst before the US Food and Drug Administration Public Meeting on Behind the Counter Availability of Drugs November 14, 2007 Consumers Union
Statement of Consumers Union William Vaughan, Senior Policy Analyst before the US Food and Drug Administration Public Meeting on Behind the Counter Availability of Drugs November 14, 2007 Consumers Union
PUBLIC ATTITUDES, BELIEFS AND KNOWLEDGE ABOUT SMALLPOX
 Working Papers Project on the Public and Biological Security Harvard School of Public Health 5. PUBLIC ATTITUDES, BELIEFS AND KNOWLEDGE ABOUT SMALLPOX Robert J. Blendon, Harvard School of Public Health,
Working Papers Project on the Public and Biological Security Harvard School of Public Health 5. PUBLIC ATTITUDES, BELIEFS AND KNOWLEDGE ABOUT SMALLPOX Robert J. Blendon, Harvard School of Public Health,
Overview of the 2005 IOM Report Improving Breast Imaging Quality Standards
 Overview of the 2005 IOM Report Improving Breast Imaging Quality Standards Goals of 2005 Report Study commissioned by US Congress in preparation for reauthorization of MQSA in 2007. to determine whether
Overview of the 2005 IOM Report Improving Breast Imaging Quality Standards Goals of 2005 Report Study commissioned by US Congress in preparation for reauthorization of MQSA in 2007. to determine whether
This is an edited transcript of a telephone interview recorded in March 2010.
 Sound Advice This is an edited transcript of a telephone interview recorded in March 2010. Dr. Patricia Manning-Courtney is a developmental pediatrician and is director of the Kelly O Leary Center for
Sound Advice This is an edited transcript of a telephone interview recorded in March 2010. Dr. Patricia Manning-Courtney is a developmental pediatrician and is director of the Kelly O Leary Center for
ASRT Position Statements
 ASRT Position Statements Effective June 25, 2017 Contents Collective Bargaining Units... 1 Conjoint Evaluation of Educational Programs... 1 Degree Requirements for Medical Imaging and Radiation Therapy
ASRT Position Statements Effective June 25, 2017 Contents Collective Bargaining Units... 1 Conjoint Evaluation of Educational Programs... 1 Degree Requirements for Medical Imaging and Radiation Therapy
SURVEY OF MAMMOGRAPHY PRACTICE
 Study ID SURVEY OF MAMMOGRAPHY PRACTICE This survey takes about 10-15 minutes to complete. We realize how busy you are and greatly appreciate your time. Your input can help make a difference in mammography.
Study ID SURVEY OF MAMMOGRAPHY PRACTICE This survey takes about 10-15 minutes to complete. We realize how busy you are and greatly appreciate your time. Your input can help make a difference in mammography.
Kish chakrabarti, Ph.D. Senior Physicist CDRH/FDA
 Facility Certification Extension Requirements, Quality Assurance and Medical Physicists role for Hologic Selenia Dimensions Digital Breast Tomosynthesis (DBT) System Kish chakrabarti, Ph.D. Senior Physicist
Facility Certification Extension Requirements, Quality Assurance and Medical Physicists role for Hologic Selenia Dimensions Digital Breast Tomosynthesis (DBT) System Kish chakrabarti, Ph.D. Senior Physicist
Mammography Services. Resources Guidebook
 Services Resources Guidebook Learn about early detection for breast cancer. At, we re dedicated to helping women protect themselves against breast cancer. Breast cancer is the second deadliest cancer among
Services Resources Guidebook Learn about early detection for breast cancer. At, we re dedicated to helping women protect themselves against breast cancer. Breast cancer is the second deadliest cancer among
HEALTHCARE AI DEVELOPMENT CYCLE
 Dr. Keith Dreyer Chief Science Officer, ACR Data Science Institute ACR Board of Chancellors, Chairman Informatics Commission Chief Data Science Officer, MGH, BWH, Partners Healthcare Associate Professor
Dr. Keith Dreyer Chief Science Officer, ACR Data Science Institute ACR Board of Chancellors, Chairman Informatics Commission Chief Data Science Officer, MGH, BWH, Partners Healthcare Associate Professor
Breast Care Unit. 1. The triple assessment means that your breast will be examined by a doctor trained in breast disease.
 Breast Care Unit Information for patients visiting a symptomatic breast clinic at the Medway NHS Foundation Trust Breast Care Unit, with breast symptoms. Your doctor has referred you to our symptomatic
Breast Care Unit Information for patients visiting a symptomatic breast clinic at the Medway NHS Foundation Trust Breast Care Unit, with breast symptoms. Your doctor has referred you to our symptomatic
Chairwoman Bono Mack, Vice-Chairwoman Blackburn, Ranking Member Butterfield and
 United States House of Representatives Committee on Energy and Commerce Subcommittee on Commerce, Manufacturing and Trade Hearing on Chairwoman Bono Mack, Vice-Chairwoman Blackburn, Ranking Member Butterfield
United States House of Representatives Committee on Energy and Commerce Subcommittee on Commerce, Manufacturing and Trade Hearing on Chairwoman Bono Mack, Vice-Chairwoman Blackburn, Ranking Member Butterfield
Breast Cancer. What is breast cancer?
 Scan for mobile link. Breast Cancer Breast cancer is a malignant tumor in or around breast tissue. It usually begins as a lump or calcium deposit that develops from abnormal cell growth. Most breast lumps
Scan for mobile link. Breast Cancer Breast cancer is a malignant tumor in or around breast tissue. It usually begins as a lump or calcium deposit that develops from abnormal cell growth. Most breast lumps
BREAST CANCER SCREENING IS A CHOICE
 BREAST CANCER SCREENING IS A CHOICE by ELAINE SCHATTNER, MD no financial disclosures (ES) American Association for Cancer Research Typical headlines focus on controversy 2 Data: Breast Cancer Incidence
BREAST CANCER SCREENING IS A CHOICE by ELAINE SCHATTNER, MD no financial disclosures (ES) American Association for Cancer Research Typical headlines focus on controversy 2 Data: Breast Cancer Incidence
Supplemental Screening for Women with Dense Breast Tissue. Public Meeting December 13, 2013
 Supplemental Screening for Women with Dense Breast Tissue Public Meeting December 13, 2013 Agenda Meeting Convened 10am-10:15am Presentation of the Evidence and Voting Questions, Q&A 10:15am 11:15am Discussion
Supplemental Screening for Women with Dense Breast Tissue Public Meeting December 13, 2013 Agenda Meeting Convened 10am-10:15am Presentation of the Evidence and Voting Questions, Q&A 10:15am 11:15am Discussion
Update on MQSA and Mammography Accreditation
 MQSA - Who s Who Update on MQSA and Mammography Accreditation The Law: Mammography Quality Standards Act (MQSA) The Regulator: US Food and Drug Administration (FDA) Priscilla F. Butler, M.S. Senior Director,
MQSA - Who s Who Update on MQSA and Mammography Accreditation The Law: Mammography Quality Standards Act (MQSA) The Regulator: US Food and Drug Administration (FDA) Priscilla F. Butler, M.S. Senior Director,
What Does Mammography Follow Up Involve?
 IOWA RADIOLOGY 1 What Does Mammography Follow Up Involve? 515-226-9810 Ankeny Clive Downtown Des Moines Lakeview IOWA RADIOLOGY 2 Table of Contents Introduction... 1 Imaging... 2 Mammography and Ultrasound...
IOWA RADIOLOGY 1 What Does Mammography Follow Up Involve? 515-226-9810 Ankeny Clive Downtown Des Moines Lakeview IOWA RADIOLOGY 2 Table of Contents Introduction... 1 Imaging... 2 Mammography and Ultrasound...
Benign Intraparenchymal Scarring in the DBT Era
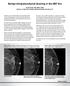 Benign Intraparenchymal Scarring in the DBT Era David Gruen, MD, MBA, FACR Director of Women s Imaging, Stamford Health, Stamford, CT With the advent of Digital Breast Tomosynthesis (DBT or 3D mammography),
Benign Intraparenchymal Scarring in the DBT Era David Gruen, MD, MBA, FACR Director of Women s Imaging, Stamford Health, Stamford, CT With the advent of Digital Breast Tomosynthesis (DBT or 3D mammography),
2018 Candidate Guide. Leading in the fight to end Alzheimer's
 2018 Candidate Guide Leading in the fight to end Alzheimer's Table of Contents Candidate Letter...2 2018 Alzheimer's Disease Facts and Figures...3 Federal Policies to Lead on Alzheimer's...4 State Policies
2018 Candidate Guide Leading in the fight to end Alzheimer's Table of Contents Candidate Letter...2 2018 Alzheimer's Disease Facts and Figures...3 Federal Policies to Lead on Alzheimer's...4 State Policies
Breast Tomosynthesis An additional screening tool in the fight against breast cancer
 What to Expect Breast Tomosynthesis An additional screening tool in the fight against breast cancer Every woman over 40 should be examined for breast cancer once a year. American Cancer Society What to
What to Expect Breast Tomosynthesis An additional screening tool in the fight against breast cancer Every woman over 40 should be examined for breast cancer once a year. American Cancer Society What to
Running head: BREAST CANCER SCREENING 1
 Running head: BREAST CANCER SCREENING 1 Breast Cancer Screening Carol A. Lamoureux-Lewallen Briar Cliff University BREAST CANCER SCREENING 2 Breast Cancer Screening Policy Problem The federally appointed
Running head: BREAST CANCER SCREENING 1 Breast Cancer Screening Carol A. Lamoureux-Lewallen Briar Cliff University BREAST CANCER SCREENING 2 Breast Cancer Screening Policy Problem The federally appointed
Advances in Breast Cancer Diagnosis and Treatment. Heidi Memmel, MD FACS Surgical Director of Caldwell Breast Center September 26, 2015
 Advances in Breast Cancer Diagnosis and Treatment Heidi Memmel, MD FACS Surgical Director of Caldwell Breast Center September 26, 2015 Advances in Breast Cancer Diagnosis and Treatment Recommendations
Advances in Breast Cancer Diagnosis and Treatment Heidi Memmel, MD FACS Surgical Director of Caldwell Breast Center September 26, 2015 Advances in Breast Cancer Diagnosis and Treatment Recommendations
POSITIONING ACR REQUIREMENTS IMAGE REVIEW CATEGORIES DEFICIENCIES IN POSITIONING ACR REQUIREMENTS 3/28/2016 NUMBER 1 REASON FOR ACR FAILURE
 CERTIFYING AGENCIES MAMMOGRAPHY CLINICAL IMAGE EVALUATION Pam Fulmer, BA RT (R)(M)(QM) FDA approved certifying states States can only certify facilities within their state borders Illinois Iowa South Carolina
CERTIFYING AGENCIES MAMMOGRAPHY CLINICAL IMAGE EVALUATION Pam Fulmer, BA RT (R)(M)(QM) FDA approved certifying states States can only certify facilities within their state borders Illinois Iowa South Carolina
Hologic Digital Technology Forms Cornerstone For Growing Women s Health Center
 Hologic Digital Technology Forms Cornerstone For Growing Women s Health Center By Steven K. Wagner During her 30 years in radiology, Peggy Reed has developed a number of guiding principles. One has served
Hologic Digital Technology Forms Cornerstone For Growing Women s Health Center By Steven K. Wagner During her 30 years in radiology, Peggy Reed has developed a number of guiding principles. One has served
Subtitle E--National Bioengineered Food Disclosure Standard
 GMO Labeling Bill S.764 as of July 14, 2016* [Congressional Bills 114th Congress] [From the U.S. Government Publishing Office]* [S. 764 Enrolled Bill (ENR)] S.764 One Hundred Fourteenth Congress of the
GMO Labeling Bill S.764 as of July 14, 2016* [Congressional Bills 114th Congress] [From the U.S. Government Publishing Office]* [S. 764 Enrolled Bill (ENR)] S.764 One Hundred Fourteenth Congress of the
April 30, By Electronic Mail
 April 30, 2018 By Electronic Mail Dr. Scott Gottlieb, Commissioner Office of the Commissioner Food and Drug Administration 10903 New Hampshire Avenue Silver Spring, MD 20993 CommissionerFDA@fda.hhs.gov
April 30, 2018 By Electronic Mail Dr. Scott Gottlieb, Commissioner Office of the Commissioner Food and Drug Administration 10903 New Hampshire Avenue Silver Spring, MD 20993 CommissionerFDA@fda.hhs.gov
The American College of Radiology Digital Mammography QC Manual: Frequently Asked Questions (Revised 12/12/2018; new and updated items in red)
 The American College of Radiology Digital Mammography QC Manual: Frequently Asked Questions (Revised 12/12/2018; new and updated items in red) Table of Contents General... 1 Applicability... 3 Transitioning
The American College of Radiology Digital Mammography QC Manual: Frequently Asked Questions (Revised 12/12/2018; new and updated items in red) Table of Contents General... 1 Applicability... 3 Transitioning
CT Lung Screening Implementation Challenges: ALA/ATS Implementation Microsite
 CT Lung Screening Implementation Challenges: ALA/ATS Implementation Microsite Andrea McKee, MD Chair Radiation Oncology Lahey Hospital and Medical Center November 2018 2016 data, 3 years after ACS recommendation
CT Lung Screening Implementation Challenges: ALA/ATS Implementation Microsite Andrea McKee, MD Chair Radiation Oncology Lahey Hospital and Medical Center November 2018 2016 data, 3 years after ACS recommendation
Alex Azar Secretary, Department of Health and Human Services
 February 28, 2018 Alex Azar Secretary, Department of Health and Human Services Dear Secretary Azar, On behalf of the Endocrine Society members and leaders, I write to offer our assistance as you lead the
February 28, 2018 Alex Azar Secretary, Department of Health and Human Services Dear Secretary Azar, On behalf of the Endocrine Society members and leaders, I write to offer our assistance as you lead the
The Telecommunications Act of 1996
 The Telecommunications Act of 1996 [Editor s note: This article was originally written in 2004 and may have outdated content. Please refer to the DCMP s Clearinghouse of Information for updated information
The Telecommunications Act of 1996 [Editor s note: This article was originally written in 2004 and may have outdated content. Please refer to the DCMP s Clearinghouse of Information for updated information
KEY MESSAGES: GET SCREENED
 KEY MESSAGES: GET SCREENED Get Screened Learning Objectives After your discussion, the listeners or audience should be able to: Name the two most common risk factors for breast cancer, Name at least two
KEY MESSAGES: GET SCREENED Get Screened Learning Objectives After your discussion, the listeners or audience should be able to: Name the two most common risk factors for breast cancer, Name at least two
Experience with Tomosynthesis (3-D) Mammography in a Mobile Setting and Current Controversies
 Experience with Tomosynthesis (3-D) Mammography in a Mobile Setting and Current Controversies Jennifer Lance, MHSA, MS Jerry McLarty, PhD LSU Feist-Weiller Cancer Center Shreveport LA Sept 15, 2008 A brief
Experience with Tomosynthesis (3-D) Mammography in a Mobile Setting and Current Controversies Jennifer Lance, MHSA, MS Jerry McLarty, PhD LSU Feist-Weiller Cancer Center Shreveport LA Sept 15, 2008 A brief
The Harvard Women s Health Watch is committed to helping women un cover helpful
 The Harvard Women s Health Watch is committed to helping women un cover helpful information for staying healthy. This report discusses some of the latest breakthroughs in women s health. You ll read about
The Harvard Women s Health Watch is committed to helping women un cover helpful information for staying healthy. This report discusses some of the latest breakthroughs in women s health. You ll read about
Galactography (Ductography)
 Scan for mobile link. Galactography (Ductography) Galactography uses mammography and an injection of contrast material to create pictures of the inside of the breast s milk ducts. It is most commonly used
Scan for mobile link. Galactography (Ductography) Galactography uses mammography and an injection of contrast material to create pictures of the inside of the breast s milk ducts. It is most commonly used
Screening for Breast Cancer
 Understanding Task Force Recommendations Screening for Breast Cancer U.S. Preventive Services Task Force (Task Force) has issued a final recommendation statement on Screening for Breast Cancer. se final
Understanding Task Force Recommendations Screening for Breast Cancer U.S. Preventive Services Task Force (Task Force) has issued a final recommendation statement on Screening for Breast Cancer. se final
Breast Density It's the Law
 Last year Iowa became the 30th state in the last 12 years to require that density information be added to the written mammogram report to the patient. This report is sent directly from the interpreting
Last year Iowa became the 30th state in the last 12 years to require that density information be added to the written mammogram report to the patient. This report is sent directly from the interpreting
10.2 Summary of the Votes and Considerations for Policy
 CEPAC Voting and Policy Implications Summary Supplemental Screening for Women with Dense Breast Tissue December 13, 2013 The last CEPAC meeting addressed the comparative clinical effectiveness and value
CEPAC Voting and Policy Implications Summary Supplemental Screening for Women with Dense Breast Tissue December 13, 2013 The last CEPAC meeting addressed the comparative clinical effectiveness and value
C. No employee shall report to work or remain on duty while having a detectable blood alcohol concentration.
 1 Series 3000 Personnel Section 3100 General Provisions Policy 3101 Drug Free Work Place File: 3101 3101.1 STATEMENT OF PURPOSE: The WV Board of Education and the Harrison County Board of Education recognize
1 Series 3000 Personnel Section 3100 General Provisions Policy 3101 Drug Free Work Place File: 3101 3101.1 STATEMENT OF PURPOSE: The WV Board of Education and the Harrison County Board of Education recognize
Texas Vendor Drug Program Specialty Drug List Process. February 2019
 Texas Vendor Drug Program Specialty Drug List Process February 2019 Table of Contents Table of Contents...1 1 About the Specialty Drug List...2 1.1 Information for Pharmacies... 2 1.2 Information for Managed
Texas Vendor Drug Program Specialty Drug List Process February 2019 Table of Contents Table of Contents...1 1 About the Specialty Drug List...2 1.1 Information for Pharmacies... 2 1.2 Information for Managed
Association of American Cancer Institutes
 Association of American Cancer Institutes Statement by the Association of American Cancer Institutes on Fiscal Year (FY) 2016 Appropriations for the Department of Health and Human Services Subcommittee
Association of American Cancer Institutes Statement by the Association of American Cancer Institutes on Fiscal Year (FY) 2016 Appropriations for the Department of Health and Human Services Subcommittee
House Committee on Energy and Commerce House Committee on Energy and Commerce. Washington, DC Washington, DC 20515
 February 28, 2018 The Honorable Michael Burgess, M.D. The Honorable Gene Green Chairman Ranking Member Subcommittee on Health Subcommittee on Health House Committee on Energy and Commerce House Committee
February 28, 2018 The Honorable Michael Burgess, M.D. The Honorable Gene Green Chairman Ranking Member Subcommittee on Health Subcommittee on Health House Committee on Energy and Commerce House Committee
FACT SHEET: Federal Parity Task Force Takes Steps to Strengthen Insurance Coverage for Mental Health and Substance Use Disorders
 The White House Office of the Press Secretary For Immediate Release October 27, 2016 FACT SHEET: Federal Parity Task Force Takes Steps to Strengthen Insurance Coverage for Mental Health and Substance Use
The White House Office of the Press Secretary For Immediate Release October 27, 2016 FACT SHEET: Federal Parity Task Force Takes Steps to Strengthen Insurance Coverage for Mental Health and Substance Use
GENERAL COMMENTS. The Task Force Process Should be Fully Open, Balanced and Transparent
 December 9, 2013 Submitted Electronically United States Preventive Services Task Force c/o Dr. Robert Cosby Agency for Healthcare Research and Quality 540 Gaither Road Rockville, MD 20850 RE: USPSTF Draft
December 9, 2013 Submitted Electronically United States Preventive Services Task Force c/o Dr. Robert Cosby Agency for Healthcare Research and Quality 540 Gaither Road Rockville, MD 20850 RE: USPSTF Draft
2015 Reimbursement Information for Mammography, CAD and Digital Breast Tomosynthesis 1
 GE Healthcare 2015 Reimbursement Information for Mammography, CAD and Digital Breast Tomosynthesis 1 April, 2015 www.gehealthcare.com/reimbursement This advisory addresses Medicare coding, coverage and
GE Healthcare 2015 Reimbursement Information for Mammography, CAD and Digital Breast Tomosynthesis 1 April, 2015 www.gehealthcare.com/reimbursement This advisory addresses Medicare coding, coverage and
61 Recommendations for better use of medications
 Chapter 61 Recommendations for better use of medications As stated elsewhere in this book, we are fortunate to have a number of effective medications for treatment of many medical conditions. The drug
Chapter 61 Recommendations for better use of medications As stated elsewhere in this book, we are fortunate to have a number of effective medications for treatment of many medical conditions. The drug
2. Please complete a separate application for each physical location. 3. Professional or Read-Only Groups interpreting imaging
 Highmark Professional Provider Privileging Application INSTRUCTIONS 1. Complete this application if you provide any diagnostic imaging services. 2. Please complete a separate application for each physical
Highmark Professional Provider Privileging Application INSTRUCTIONS 1. Complete this application if you provide any diagnostic imaging services. 2. Please complete a separate application for each physical
Scope of Practice for the Diagnostic Ultrasound Professional
 Scope of Practice for the Diagnostic Ultrasound Professional Copyright 1993-2000 Society of Diagnostic Medical Sonographers, Dallas, Texas USA: All Rights Reserved Worldwide. Organizations which endorse
Scope of Practice for the Diagnostic Ultrasound Professional Copyright 1993-2000 Society of Diagnostic Medical Sonographers, Dallas, Texas USA: All Rights Reserved Worldwide. Organizations which endorse
