What is New in CML Jorge Cortes, MD Chief, CML and AML Sections Department of Leukemia MD Anderson Cancer Center Houston, Texas
|
|
|
- Allyson Butler
- 5 years ago
- Views:
Transcription
1 What is New in CML 2018 Jorge Cortes, MD Chief, CML and AML Sections Department of Leukemia MD Anderson Cancer Center Houston, Texas
2 Final Results CML-IV Molecular Response with Imatinib 1538 pts newly diagnosed CML-CP randomized to imatinib 400, imatinib 800, imatinib + IFN, imatinib + ara-c, or imatinib after IFN Median age 53 yrs, 12% HR EURO score 62% still on imatinib at final evaluation 92% 89% 81% 72% 59% Patients at risk Kalmanti L, et al. Leukemia. 2015;29(5): Hehlmann R, et al. Blood. 2017;130(suppl): Abstract 897.
3 Final Results CML-IV Overall Survival By Treatment Arm By 6 mo Predictors of survival (MVA): EURO score (low v high), setting (academic v community or office-based), comorbidity score, smoking (non v any), ACA at diagnosis (no major route v major route) Hehlmann R, et al. Blood. 2017;130(suppl): Abstract 897.
4 BFORE: First-line Bosutinib vs Imatinib in CML Molecular Response at 12 and 18 Months Randomized frontline pivotal study of bosutinib 400 mg (n=268) v imatinib 400 mg (n=268) Stratified by Sokal and geographic area Molecular response (%) P= P= P= P= P=0.01 Month: P= Bosutinib ITT: n=268 mitt: n= months 18 months 18.3 P=0.02 MMR (ITT) MMR (mitt) MR 4 (mitt) MR 4.5 (mitt) MR 4 (ITT), % Bosutinib Imatinib P 12 mo < mo Imatinib ITT: n=268 mitt: n= months 18 months 3.3 P= MR 4.5 (ITT), % Bosutinib Imatinib P 12 mo mo Gambacorti-Passerini C, et al. Blood. 2017;130(suppl): Abstract 896.
5 BFORE: First-line Bosutinib vs Imatinib in CML EFS and OS at 18 Months (mitt Population) Bosutinib (n=246) Imatinib (n=241) Patients with on-treatment transformation to AP/BP, n (%)* 6 (2.4) 7 (2.9) Cumulative incidence of ontreatment progression or 4.5 death, % (95% CI) ( ) 6.7 ( ) Overall survival, % (95% CI) 99.6 ( ) 96.6 ( ) * 6 pts (n=3 each arm) met AP criteria within 2 wks based on basophils; all 6 continued on study drug with 4 achieving MMR and 1 CCyR. Includes death, transformation to AP/BP, doubling of WBC without CHR, loss of CCyR, or loss of CHR. Gambacorti-Passerini C, et al. Blood. 2017;130(suppl): Abstract 896.
6 BFORE: First-line Bosutinib vs Imatinib in CML Grade 3 TEAEs* (Safety Population) System organ class preferred term Blood and lymphatic system disorders, n (%) Thrombocytopenia Neutropenia Anemia Leukopenia Leukocytosis Bosutinib (n=268) 48 (17.9) 37 (13.8) 19 (7.1) 10 (3.7) 3 (1.1) 2 (0.7) Imatinib (n=265) 58 (21.9) 16 (6.0) 34 (12.8) 13 (4.9) 10 (3.8) 7 (2.6) Gastrointestinal disorders, n (%) Diarrhea Laboratory investigations, n (%) Alanine aminotransferase increased Lipase increased Aspartate aminotransferase increased Metabolism and nutrition disorders, n (%) Hypophosphatemia 31 (11.6) 22 (8.2) 91 (34.0) 56 (20.9) 28 (10.4) 27 (10.1) 14 (5.2) 3 (1.1) 9 (3.4) 2 (0.8) 35 (13.2) 4 (1.5) 12 (4.5) 5 (1.9) 12 (4.5) 9 (3.4) * Select all-causality adverse events occurring in 2% pts in either treatment arm. Medical Dictionary for Regulatory Activities (MEDRA) v20.0; pts may report >1 AE at the level of preferred term within each system organ class. Gambacorti-Passerini C, et al. Blood. 2017;130(suppl): Abstract 896.
7 Matched Cohort Study of Generic v Brand Imatinib 732 pts frontline imatinib since 2001 (prospective data 2009) Endpoints: Primary: switch to another TKI; secondary: discontinuation Probability of persistence Switch Branded 88.2 ( ) Generic 72.8 ( ) Log-rank P = 0.03 Adjusted HR = 2.13 ( ) Probability of persistence Discontinuation Branded Generic Log-rank P = 0.12 Adjusted HR = 2.85 ( ) 92.0 ( ) 85.3 ( ) Duration of follow-up (months) Duration of follow-up (months) Switched from generic: 36/167 (22%); toxicity 25 (15%), suboptimal response 12 (7%) To branded 64%, dasatinib 28%, nilotinib 8% Switched from branded: 17/167 (10%); toxicity 9 (5%), suboptimal response 9 (5%) To dasatinib 76%, nilotinib 18%, bosutinib 6% Discontinued generic: 13/167 (8%); 9 for susmr5 (4 re-treated) Discontinued branded: 4/167 (2%); 2 for susmr5 (1 re-treated) 7 Klil-Drori A, et al. Blood. 2017;130(suppl): abstract 315.
8 Generic Imatinib in US 27 pts switched to generic imatinib (19 frontline, 8 post-ifn) Median time on innovator 11.9 yrs ( ); all MMR, 20 MR4.5 After switch: CBC, SMA every 1 mo, PCR every 3 mo BCR-ABL PCR (IS) on innovator imatinib before the switch. BCR-ABL PCR (IS) on generic imatinib at last follow-up. Sustained complete molecular response. Aboudalle I, et al. Blood. 2017;130(suppl): Abstract 2906.
9 Total N = 27 Generic Imatinib in US Number (Percent) Innovator Imatinib Generic Imatinib Non hematologic AE (any grade) Edema 17 (63) 7 (26) Muscle cramps 15 (56) 8 (30) Diarrhea 11 (41) 5 (19) Fatigue 9 (33) 5 (19) Nausea 9 (33) 7 (26) Rash 4 (15) 0 (0) Weight gain 3 (11) 0 (0) Increased Creatinine 2 (7) 3 (11) Increased Bilirubin 2 (7) 0 (0) Increased LDH 2 (7) 2 (7) Insomnia 1 (4) 2 (7) Pruritus 1 (4) 0 (0) Hematologic AE (any grade) Anemia 9 (33) 2 (7) Thrombocytopenia 2 (7) 0 (0) Neutropenia 1 (4) 2 (7) 5 pts discontinued: 2 elective, 2 due to diarrhea (back to innovator), 1 due to renal toxicity (pre-existing, changed to dasatinib) 1 dose reduction (fatigue, diarrhea), 1 interruption (edema) No deaths or transformations (median f/u 7 mo [3-28]) Aboudalle I, et al. Blood. 2017;130(suppl): Abstract 2906.
10 n= PETAL: Nilotinib ± PEG-IFN Overall molecular response rates 200 pts newly diagnosed CML randomized to nilotinib 300 mg BID ± PEG-IFN (lead in 30 mcg/wk x1 mo, then add nilotinib + PEG-IFN 30 mcg/wk x2, then 45 mcg/wk x 2yr) p=0.019 p=0.91 p= Predictors MR4.5 at 12 mo: female, Sokal low + intermediate, PEG-IFN cumulative 540 mcg (by 9 mo) Nicolini F, et al. Blood. 2017;130(suppl): Abstract 899.
11 Lower-Dose Dasatinib Newly Diagnosed CML-CP 56 pts, median age 46 y (range, y) Dasatinib 50 mg/d Response, n/n (%) 3 mo 6 mo 9 mo 12 mo BCR-ABL/ABL <10% IS 40/43 (93) 26/27 (96) 19/20 (95) 9/9 (100) CCyR 23/43 (51) 25/27 (93) 18/20 (90) 9/9 (100) MMR 12/43 (28) 19/27 (70) 16/20 (80) 9/9/ (100) MR4 1/43 (2) 8/27 (30) 14/20 (70) 9/9 (100) MR4.5 0/43 (0) 2/27 (7) 4/20 (20) 3/9 (33) Treatment interruption 14 (25); no dose modifications Naqvi K, et al. Blood. 2017;130(suppl): Abstract 1611.
12 Radotinib v Imatinib in Frontline CML Molecular Response 2 nd generation TKI approved in Korea Active against BCR-ABL and most mutants (not T315I) MMR MR4.5 By 36 months Radotinib 300mg BID Patients with MMR, % By 12 months 52%, P =.0044 Δ 22% 30% By 24 months 66%, P =.0536 Δ 15% 51% 75%, P =.0076 Δ 21% 54% Patients with MR 4.5, % Imatinib 400mg QD By 24 months 33%, P =.1331 By 12 months Δ 11% 15%, P = % By 36 months 43%, P =.0538 Δ 13% 28% Δ 6% 9% Months since randomization Months since randomization 3 mo EMR: radotinib 86%, imatinib 71% 6 mo EMR: radotinib 77%, imatinib 58% Do YR, et al. Blood. 2017;130(suppl): Abstract 317.
13 Radotinib v Imatinib in Frontline CML Cardiovascular Events (All Grades) Adverse Event, n (%) Radotinib 300mg BID (n=79) Imatinib 400mg QD (n=81) Ischemic Heart Disease 0 (0) 0 (0) Acute MI 1 (1) 0 (0) Unstable Angina 1 (1) 0 (0) Angina Pectoris 2 (3) 0 (0) PAOD a 0 (0) 0 (0) a PAOD: Peripheral Arterial Occlusive Disease Regardless of causalities Do YR, et al. Blood. 2017;130(suppl): Abstract 317.
14 Frontline Imatinib in Children and Adolescents with CML 148 pts, mean age 12.3 y (range, y) Imatinib 300 mg/m 2 (max dose 400 mg) Response At time (mo) Percentage CCyR MMR MR MR Failure* 27 PFS TFR 2/7 pts * Inadequate response 12%, relapse 6%, intolerance 6%, poor adherence 1%, unknown 2% Median growth impairment: ~-0.25 SD/y Suttorp M, et al. Blood. 2017;130(suppl): Abstract 898.
15 2 nd -line Bosutinib in CP CML: 8-Year Update Efficacy Summary Phase 1/2 bosutinib 500 mg/d 284 pts: imatinib resistant 195, intolerant 89 Median age 53 y (18-91 y), prior IFN 35%, SCT 3% n (%) Imatinibresistant Imatinibintolerant Total Cytogenetic responses Evaluable patients MCyR 110 (60) 48 (60) 158 (60) CCyR 89 (49) 41 (51) 130 (50) Survival outcomes Cumulative incidence of progression or death 57 (29) 10 (11) 67 (24) Deaths 40 (21) 11 (12) 51 (18) New toxicities year 5-8: renal (14%), diarrhea 1 (0.8%), liver 7 (6%) Vascular events (per 100 pt/year): cardiovascular 0.008, cerebrovascular 0.005, peripheral vascular Brümmendorf TH, et al. Blood. 2017;130(suppl): Abstract 900.
16 2 nd -line Bosutinib in CP CML: 8-Year Update MCyR Duration & Overall Survival MCyR Duration Overall Survival Probability of retaining response Evaluable patients n (%) Sustained MCyR* at Year 8, % (95% CI) Imatinib-resistant (n=182) 110 (60.4) 60.4 ( ) Imatinib-intolerant (n=80) 48 (60.0) 74.5 ( ) Total (n=262) 158 (60.3) 64.5 ( ) Probability of overall survival Patients Deaths n (%) OS at Year 8, % (95% CI) Imatinib-resistant (n=195) 40 (20.5) 74.5 ( ) Imatinib-intolerant (n=89) 11 (12.4) 87.2 ( ) Total (N=284) 51 (18.0) 78.5 ( ) Duration of MCyR, weeks Months No deaths assessed as treatment-related; 6 new deaths after the 5-year F/U * Includes responses newly attained or maintained from baseline; 29.7% of patients had loss of response, PD, or death (imatinib-resistant: 34.5%; imatinib-intolerant: 18.8%). Received 1 dose of bosutinib and had a valid baseline cytogenetic assessment. 13 deaths occurred within 30 days of the last bosutinib dose (imatinib-resistant patients: n=11; imatinib-intolerant patients: n=2). Most deaths were due to progressive disease (n=29 [10.2%]; imatinib-resistant patients: n=23 [11.8%]; imatinib-intolerant patients: n=6 [6.7%]) or AEs unrelated to study drug (n=16 [5.6%]; imatinib-resistant patients: n=14 [7.2%]; imatinib-intolerant patients: n=2 [2.2%]). Brümmendorf TH, et al. Blood. 2017;130(suppl): Abstract 900.
17 2 nd Line TKI in CML CP 621 pts treated with 2 nd TKI: dasatinib 55%, nilotinib 31%, bosutinib 6%, other (7%) 1 st TKI: imatinib (85%), ponatinib (7%), nilotinib (5%), dasatinib (3%) or bosutinib (<1%) Reason to switch: resistance 55%, intolerance 45% Median F/U: 50 mo ( mo) Response: CCyR 50%; Best molecular: MMR 13%, MR4.5 38% MVA: specific TKI no impact in OS or TFS; nilotinib or other inferior EFS and FFS Chamoun K, et al. Blood. 2017;130(suppl): Abstract 1594.
18 2 nd Line TKI in CML CP MR4.5 Overall Survival N Events Transformation-Free Survival Event-Free Survival N Events N Events Chamoun K, et al. Blood. 2017;130(suppl): Abstract 1594.
19 5-Year Outcome with Ponatinib in CML-CP by Prior TKI 270 pts CML-CP treated with ponatinib 45 mg/d post- TKI failure Median F/U 56.8 mo ( mo) Percent 5-yr Probability MCyR MMR MR4.5 MCyR MMR PFS OS 1 TKI NA TKI TKI TKI NA NA NA Hochhaus A, et al. Blood. 2017;130(suppl): Abstract 1617.
20 3 rd Line TKI in CML 185 pts treated with 3 rd TKI: nilotinib 36%, dasatinib 35%, ponatinib 12%, imatinib 10%, bosutinib 7% Median time from Dx: 58 mo (3 199 mo) 1 st TKI: intolerance 44%, resistance 67%; 2 nd TKI: intolerance 60%, resistance 49% Response % CCyR 38 MMR 41 MR4 33 MR MVA predictors of CCyR: ponatinib, Hgb, intolerance (same + nilotinib for MMR) Khan M, et al. Blood. 2017;130(suppl): Abstract 2882.
21 3 rd Line TKI in CML OS TFS EFS MVA for survival: ponatinib, bosutinib; age; female; intolerance (v resistance) Khan M, et al. Blood. 2017;130(suppl): Abstract 2882.
22 PF rd Generation Inhibitor of Bcr-Abl 3 rd generation Abl inhibitor, structural analog of ponatinib Rationally designed to avoid inhibition of off-target kinases Orthotopic murine BaF3 cell model of T315I+ CML Ponatinib NSG Mice iv xenograft of human T315I Ph+ALL Turkina AG, et al. Blood. 2017;130(suppl): Abstract 895.
23 Phase 1 of PF-114 in CML Structural analog of ponatinib modified to avoid inhibition of key kinases 24 pts with CML any phase with failure to 2 TKI or with T315I CML Phase Chronic Accelerated Mutation N CHR MCyR % n * /N ** % n * /N *** T315I /5 80 4/5 All / /10 T315I 1 0 0/1 0 0/1 All /2 0 0/2 Blast T315I /1 0 0/1 No DLT at doses up to 500 mg Main AE rash: 13 grade 2, 4 grade 3 (1 DLT) No cardiac events (54% high- to fatal-riak per European SCORE) n*- number of patients who achieved response during treatment N** - number of patients evaluable for hematologic response assessment: were not in CHR at enrollment N*** - number of patients evaluable for cytogenetic response assessment: were not in MCyR at enrollment and completed at least 3 cycles Turkina AG, et al. Blood. 2017;130(suppl): Abstract 895.
24 New TKI Under Development TKI Features Current status ABL-001 Allosteric inhibitor Completed phase 1, single agent and combination Pivotal phase 3 3 rd line v bosutinib started Radotinib 2 nd generation Approved in South Korea 1 st and 2 nd line Pending studies elsewhere PF-114 Ponatinib analog, not binding VEGFR Nearing MTD Starting phase 2 K rd generation Phase 1 started
25 EURO-Ski: The Effect of Duration of TKI Treatment and MR4 755 pts with sustained MR4 for 1 yr Molecular relapse = loss MMR Overall MRFS 47% at 36 months Predictors of outcome: Sokal, depth of response, history of resistance, prior IFN, treatment duration, molecular response duration Duration of TKI Therapy Duration of MR4 Saussele S, et al. Blood. 2017;130(suppl): Abstract 313.
26 EURO-Ski: Probability of Molecular Relapse by Duration of TKI Treatment and Duration of MR4 Duration of MR4 TKI Treatment Duration Before MR4 4 years MR 4 duration: Probability = 0.53 (95%-CI: ) 6 years MR 4 duration: Probability = 0.59 (95%-CI: ) 0.2 years duration: Probability = years duration: Probability = 0.62 absolute increase of probability of about 3% / year MR 4 duration: Odds ratio: 1.13 (95%-CI ) relative increase of probability to stay in MMR about 0.86% / year TKI before MR 4 : Odds ratio: 1.04 (95%-CI: ) TFR is mostly dependent on duration of response Duration of TKI independent of duration of response has minimal benefit as time on TKI prior to MR4 has minimal benefit Saussele S, et al. Blood. 2017;130(suppl): Abstract 313.
27 Outcome of CML Patients After TKI Discontinuation - MDACC Initial Report Updated Report 1.00 CMR before TKI discontinuation 64 months 75% 75% 65% Cumulative survival < 64 months Rate of relapse 6% 15% p = % Month to molecular relapse < >120 MR4.5 duration before discontinuation (months) Benjamini O, et al. Leuk Lymphoma. 2014;55(12): Chamoun K, et al. Blood. 2016;128(suppl): Abstract 1923.
28 DASFREE - Molecular Relapse-Free Survival 84 pts with sustained MR4.5 for 1 yr (confirmed in central lab) Relapse = loss of MMR Frontline 44%, subsequent 56% (resistant 53%, intolerant 38%) 100 Estimated survival Molecular Relapse-free Survival (%) % (38.0, 59.4) 95% confidence band Pts on 1 st -line dasatinib (n = 37) Pts on subsequent lines of dasatinib (n = 47) Resistant (n = 25) Intolerant (n = 18) MRFS, % (95% CI) 54 (38.0, 70.1) 45 (30.2, 58.7) 44 (24.5, 63.5) 50 (26.9, 73.1) 0 Patients at risk Months Since Dasatinib Discontinuation No patients lost CCyR or CHR, transformed or died All evaluable pts regained MMR a median of 1.9 mo after restarting dasatinib Factors associated with RFS: age (p = 0.04), 1 st line (p = 0.07) 8 withdrawal events Shah N, et al. Blood. 2017;130(suppl): Abstract 314.
29 Nilotinib Schedule Change to QD Observational study of nilotinib schedule change to QD: 71 pts to 450 mg QD 89%, 400 mg QD 9%, 300 mg QD 2% Loss of MMR not observed in pts with DMR* before change 2 pts with MMR only lost MMR spontaneously re-gained Most patients improved response after schedule change Survival without MMR loss 100% 80% 60% 40% 20% Survival without MMR loss by molecular response at baseline Molecular response category: MR3 only (n=10) MR4 only (detectable) (n=19) At least MR4.5 (detectable+ undetectable) (n=42) Logrank: % *DMR: deep molecular response Months since nilotinib dose reduction Proportion of patients % Molecular response after change 80% 60% 40% 20% 0% 14.5% 8.8% MR3 only 27.5% 20.6% MR4 only Month 0 Month 12 58% 70.6% At least MR4.5 Rea D, et al. Blood. 2017;130(suppl): Abstract 318.
30 2 nd -line Bosutinib in CP CML: 8-Year Update Cytogenetic Responses After Dose Reduction Imatinibresistant (n=195) Imatinibintolerant (n=89) Total (N=284) Patients reducing bosutinib to 400 mg QD, n MCyR, % 1 st response following dose reduction Response maintained after dose reduction Response lost after dose reduction CCyR, % 1 st response following dose reduction Response maintained after dose reduction Response lost after dose reduction Brümmendorf TH, et al. Blood. 2017;130(suppl): Abstract 900.
31 Significance of Dose Adjustments of 2 nd Generation TKI in Frontline Therapy 98 pts treated with nilotinib 400mg BID (n=50) or dasatinib 50 mg BID or 100 mg QD (n=48) as initial therapy fro CML CP Dose reductions in 50 (51%) (nilotinib 42%, dasatinib 60%) Percentage Dose reduction No dose reduction MCyR CCyR yr FFS yr OS Santos FP, et al. Br J Haematol. 2010;150(3):
32 Management of Pregnancy and CML Assi R, et al. Blood. 2017;130(suppl): Abstract 2881.
33 Management of Pregnancy and CML 20 unplanned pregnancies Status at pregnancy: CHR 2, CCyR 3, MMR 6, MR4.5 9 Treatment during pregnancy: observation 18, hydroxyurea 1, leukapheresis 1 Pregnancy outcome: live birth 11, miscarriage 5, elective termination 3, ongoing 1 Response last F/U: MR4.5 10, MMR 7, CCyR 3 14 CML diagnosis during pregnancy Treatment: None 5, hydorxyurea 5, IFN 2, leukapheresis 1, TKI 1 (@ 3 rd trimester) Pregnancy outcomes: live birth 5, miscarriage 2, elective termination 1 Response: MR4.5 = 6, MMR = 7, 1 NR (had received IFN, nor response to dasatinib BP) 9 planned pregnancies (6 CMR pre-conception) 2 transcripts observation, 1 lost CHR hydrea dasatinib after delivery (MR4) Assi R, et al. Blood. 2017;130(suppl): Abstract 2881.
34 NGS for Mutations Analysis in CML 214 pts with failure (n=132) or warning (n=82) to TKI sequenced by Sanger and NGS 4 labs: concordance 81% (discordant samples with mutations <3%) Sanger: 42 (20%) with mutations NGS: 74 (36%) new mutations: 52 negative by Sanger, 24 additional mutations T315I 8, resistant to TKI (non-t315i) 43, unknown profile 25 Median mutation burden 11% (3-19%) 1-4 low-burden mutation per patient Compound mutations 2 pts (CML-BP) Soverini S, et al. Blood. 2017;130(suppl): Abstract 248.
35 Genetic Complexity of CML Integrative genomics: multiple mutations and fusions at diagnosis and BP BP: ABL1 mutations usually with other mutations Mutations frequently associated with progression (most commonly RUNX1, ASXL1, TET2, BCOR) Also identified in pts with CCA/Ph- Novel gene mutations: SETD1B, PTPR; novel fusions: MLL, RUNX1, IKZF1, MECOM) Branford S, et al. Blood. 2017;130(suppl): Abstract 251. Awad SA, et al. Blood. 2017;130(suppl): Abstract 250. Jain P, et al. Blood. 2017;130(suppl): Abstract 4172.
36 CML in 2018 Frontline therapy is good (and getting better, safer and cheaper?) 2 nd line options grossly equivalent; 3 rd line ponatinib better (new ones safer?) Dose reductions safe in most instances Treatment discontinuation feasible if done right (better to wait for long MR4.5) Minimal intervention during pregnancy CML more molecularly complex than we thought
37 Questions?
CML: Living with a Chronic Disease
 CML: Living with a Chronic Disease Jorge Cortes, MD Chief, CML and AML Sections Department of Leukemia M. D. Anderson Cancer Center Houston, Texas Survival in Early Chronic Phase CML TKI Interferon Chemotherapy
CML: Living with a Chronic Disease Jorge Cortes, MD Chief, CML and AML Sections Department of Leukemia M. D. Anderson Cancer Center Houston, Texas Survival in Early Chronic Phase CML TKI Interferon Chemotherapy
BMS Satellite Symposium
 ICKSH 2018 BMS Satellite Symposium Emerging Trends in CML Management CHAIRMAN The Head of Catholic Hematology Hospital The Director of the Catholic Leukemia Research Institute at the Catholic University
ICKSH 2018 BMS Satellite Symposium Emerging Trends in CML Management CHAIRMAN The Head of Catholic Hematology Hospital The Director of the Catholic Leukemia Research Institute at the Catholic University
2 nd Generation TKI Frontline Therapy in CML
 2 nd Generation TKI Frontline Therapy in CML Elias Jabbour, M.D. April 212 New York Frontline Therapy of CML in 212 - imatinib 4 mg daily - nilotinib 3 mg BID - dasatinib 1 mg daily Second / third line
2 nd Generation TKI Frontline Therapy in CML Elias Jabbour, M.D. April 212 New York Frontline Therapy of CML in 212 - imatinib 4 mg daily - nilotinib 3 mg BID - dasatinib 1 mg daily Second / third line
The concept of TFR (Treatment Free Remission) in CML
 The concept of TFR (Treatment Free Remission) in CML Giuseppe Saglio University of Turin, Italy What can we expect today on long-term therapy with TKIs in CML? German CML study IV Relative and overall
The concept of TFR (Treatment Free Remission) in CML Giuseppe Saglio University of Turin, Italy What can we expect today on long-term therapy with TKIs in CML? German CML study IV Relative and overall
Role of Second Generation Tyrosine Kinase Inhibitors in Newly Diagnosed CML. GIUSEPPE SAGLIO, MD University of Torino, Italy
 Role of Second Generation Tyrosine Kinase Inhibitors in Newly Diagnosed CML GIUSEPPE SAGLIO, MD University of Torino, Italy Outcome in 282 Patients Treated with Imatinib First Line in Hammersmith Hospital
Role of Second Generation Tyrosine Kinase Inhibitors in Newly Diagnosed CML GIUSEPPE SAGLIO, MD University of Torino, Italy Outcome in 282 Patients Treated with Imatinib First Line in Hammersmith Hospital
CML: Yesterday, Today and Tomorrow. Jorge Cortes, MD Chief CML Section Department of Leukemia The University of Texas, M.D. Anderson Cancer Center
 CML: Yesterday, Today and Tomorrow Jorge Cortes, MD Chief CML Section Department of Leukemia The University of Texas, M.D. Anderson Cancer Center Five Years of Signal Transduction Inhibition The Beginning
CML: Yesterday, Today and Tomorrow Jorge Cortes, MD Chief CML Section Department of Leukemia The University of Texas, M.D. Anderson Cancer Center Five Years of Signal Transduction Inhibition The Beginning
IRIS 8-Year Update. Management of TKI Resistance Will KD mutations matter? Sustained CCyR on study. 37% Unacceptable Outcome 17% 53% 15%
 Management of TKI Resistance Will KD mutations matter? IRIS 8-Year Update 17% 53% 5% 15% 37% Unacceptable Outcome No CCyR Lost CCyR CCyR Other 3% 7% Safety Lost-regained CCyR Sustained CCyR on study Deininger
Management of TKI Resistance Will KD mutations matter? IRIS 8-Year Update 17% 53% 5% 15% 37% Unacceptable Outcome No CCyR Lost CCyR CCyR Other 3% 7% Safety Lost-regained CCyR Sustained CCyR on study Deininger
2nd generation TKIs to first line therapy
 New Horizons 2011 Newly diagnosed CML moving 2nd generation TKIs to first line therapy Gianantonio Rosti Dept. Of Hematology and Oncology St. Orsola-Malpighi University Hospital Bologna (Italy) GIMEMA
New Horizons 2011 Newly diagnosed CML moving 2nd generation TKIs to first line therapy Gianantonio Rosti Dept. Of Hematology and Oncology St. Orsola-Malpighi University Hospital Bologna (Italy) GIMEMA
What is New in CML in Hagop Kantarjian, M.D. February 2011
 What is New in CML in 2011 Hagop Kantarjian, M.D. February 2011 1 CML. Historical vs. Modern Perspective Parameter Historical Modern Course Fatal Indolent Prognosis Poor Excellent 10-yr survival 10% 84-90%
What is New in CML in 2011 Hagop Kantarjian, M.D. February 2011 1 CML. Historical vs. Modern Perspective Parameter Historical Modern Course Fatal Indolent Prognosis Poor Excellent 10-yr survival 10% 84-90%
ELN Recommendations on treatment choice and response. Gianantonio Rosti, MD, Department of Hematology, University of Bologna, Italy
 ELN Recommendations on treatment choice and response Gianantonio Rosti, MD, Department of Hematology, University of Bologna, Italy ELN 2013 Response to Front-line Treatment Baseline 3 months 6 months OPTIMAL
ELN Recommendations on treatment choice and response Gianantonio Rosti, MD, Department of Hematology, University of Bologna, Italy ELN 2013 Response to Front-line Treatment Baseline 3 months 6 months OPTIMAL
Contemporary and Future Approaches in CML. Emory Meeting; Sea Island August 2014 Hagop Kantarjian, M.D.
 Contemporary and Future Approaches in CML Emory Meeting; Sea Island August 2014 Hagop Kantarjian, M.D. 1 CML. Historical vs. Modern Perspective Parameter Historical Modern Course Fatal Indolent Prognosis
Contemporary and Future Approaches in CML Emory Meeting; Sea Island August 2014 Hagop Kantarjian, M.D. 1 CML. Historical vs. Modern Perspective Parameter Historical Modern Course Fatal Indolent Prognosis
Guidelines and real World: Management of CML in chronic and advanced phases. Carolina Pavlovsky. FUNDALEU May 2017 Frankfurt
 Guidelines and real World: Management of CML in chronic and advanced phases Carolina Pavlovsky. FUNDALEU 26-28 May 217 Frankfurt Some Issues in CML 217 First Line treatment: Imatinib vs 2nd generation
Guidelines and real World: Management of CML in chronic and advanced phases Carolina Pavlovsky. FUNDALEU 26-28 May 217 Frankfurt Some Issues in CML 217 First Line treatment: Imatinib vs 2nd generation
Stopping Treatment in CML and dose reduction in clinical practice: Can we do it safely? YES WE CAN!
 Stopping Treatment in CML and dose reduction in clinical practice: Can we do it safely? YES WE CAN! Dragana Milojković The Hammersmith Hospital, London, UK Leukemic burden Current Aim of TKI therapy Molecular
Stopping Treatment in CML and dose reduction in clinical practice: Can we do it safely? YES WE CAN! Dragana Milojković The Hammersmith Hospital, London, UK Leukemic burden Current Aim of TKI therapy Molecular
HOW I TREAT CML. 4. KONGRES HEMATOLOGOV IN TRANSFUZIOLOGOV SLOVENIJE Z MEDNARODNO UDELEŽBO Terme Olimia, Podčetrtek,
 HOW I TREAT CML 4. KONGRES HEMATOLOGOV IN TRANSFUZIOLOGOV SLOVENIJE Z MEDNARODNO UDELEŽBO Terme Olimia, Podčetrtek, 12. - 14. april, 2012 Gianantonio Rosti Dpt of Hematology and Oncological Sciences S.
HOW I TREAT CML 4. KONGRES HEMATOLOGOV IN TRANSFUZIOLOGOV SLOVENIJE Z MEDNARODNO UDELEŽBO Terme Olimia, Podčetrtek, 12. - 14. april, 2012 Gianantonio Rosti Dpt of Hematology and Oncological Sciences S.
SESSION III: Chronic myeloid leukemia PONATINIB. Gianantonio Rosti, MD, Department of Hematology, University of Bologna, Italy
 SESSION III: Chronic myeloid leukemia PONATINIB Gianantonio Rosti, MD, Department of Hematology, University of Bologna, Italy Ponatinib A Pan-BCR-ABL Inhibitor Rationally designed inhibitor of BCR- ABL
SESSION III: Chronic myeloid leukemia PONATINIB Gianantonio Rosti, MD, Department of Hematology, University of Bologna, Italy Ponatinib A Pan-BCR-ABL Inhibitor Rationally designed inhibitor of BCR- ABL
New drugs and trials. Andreas Hochhaus
 New drugs and trials. Andreas Hochhaus Hadera I Oct 2018 Introduction ABL001 is a potent, specific inhibitor of BCR-ABL1 with a distinct allosteric mechanism of action BCR-ABL1 Protein Binds a distinct
New drugs and trials. Andreas Hochhaus Hadera I Oct 2018 Introduction ABL001 is a potent, specific inhibitor of BCR-ABL1 with a distinct allosteric mechanism of action BCR-ABL1 Protein Binds a distinct
Contemporary and Future Approaches in Management of CML. Disclosures
 Winship Cancer Institute of Emory University Contemporary and Future Approaches in Management of CML Hagop Kantarjian, MD Chairman and Professor, Department of Leukemia University of Texas M. D. Anderson
Winship Cancer Institute of Emory University Contemporary and Future Approaches in Management of CML Hagop Kantarjian, MD Chairman and Professor, Department of Leukemia University of Texas M. D. Anderson
CML David L Porter, MD University of Pennsylvania Medical Center Abramson Cancer Center CML Current treatment options for CML
 1 CML 2012 LLS Jan 26, 2012 David L Porter, MD University of Pennsylvania Medical Center Abramson Cancer Center CML 2012 Current treatment options for CML patients Emerging therapies for CML treatment
1 CML 2012 LLS Jan 26, 2012 David L Porter, MD University of Pennsylvania Medical Center Abramson Cancer Center CML 2012 Current treatment options for CML patients Emerging therapies for CML treatment
New drugs in first-line therapy
 New drugs in first-line therapy Gianantonio Rosti Dept of Hematology and Oncology Seràgnoli, Bologna University (Italy) GIMEMA (Gruppo Italiano Malattie Ematologiche dell Adulto) CML WORKING PARTY IRIS
New drugs in first-line therapy Gianantonio Rosti Dept of Hematology and Oncology Seràgnoli, Bologna University (Italy) GIMEMA (Gruppo Italiano Malattie Ematologiche dell Adulto) CML WORKING PARTY IRIS
Oxford Style Debate on STOPPING Treatment.
 Oxford Style Debate on STOPPING Treatment. This house believes that there are good reasons NOT to stop CML treatment. It should be done within clinical trials, OR only in expert centers where frequent,
Oxford Style Debate on STOPPING Treatment. This house believes that there are good reasons NOT to stop CML treatment. It should be done within clinical trials, OR only in expert centers where frequent,
Chronic Myeloid Leukaemia
 Chronic Myeloid Leukaemia Molecular Response: What is really important? Jeff Szer The Royal Melbourne Hospital PROBABILITY, % PROBABILITY OF SURVIVAL AFTER MYELOABLATIVE TRANSPLANTS FOR CML IN CHRONIC
Chronic Myeloid Leukaemia Molecular Response: What is really important? Jeff Szer The Royal Melbourne Hospital PROBABILITY, % PROBABILITY OF SURVIVAL AFTER MYELOABLATIVE TRANSPLANTS FOR CML IN CHRONIC
Starting & stopping therapy in Chronic Myeloid Leukemia: What more is needed? Richard A. Larson, MD University of Chicago March 2019
 Starting & stopping therapy in Chronic Myeloid Leukemia: What more is needed? Richard A. Larson, MD University of Chicago March 2019 Disclosures Richard A. Larson, MD Research funding to the University
Starting & stopping therapy in Chronic Myeloid Leukemia: What more is needed? Richard A. Larson, MD University of Chicago March 2019 Disclosures Richard A. Larson, MD Research funding to the University
The BCR-ABL1 fusion. Epidemiology. At the center of advances in hematology and molecular medicine
 At the center of advances in hematology and molecular medicine Philadelphia chromosome-positive chronic myeloid leukemia Robert E. Richard MD PhD rrichard@uw.edu robert.richard@va.gov Philadelphia chromosome
At the center of advances in hematology and molecular medicine Philadelphia chromosome-positive chronic myeloid leukemia Robert E. Richard MD PhD rrichard@uw.edu robert.richard@va.gov Philadelphia chromosome
Low doses of tyrosine kinase inhibitors in CML
 CML Horizons Conference Warsaw 4-6 May 2018 Low doses of tyrosine kinase inhibitors in CML Delphine Rea, MD, PhD Pôle Hématologie Oncologie Radiothérapie INSERM UMR-1160 Centre Hospitalo-Universitaire
CML Horizons Conference Warsaw 4-6 May 2018 Low doses of tyrosine kinase inhibitors in CML Delphine Rea, MD, PhD Pôle Hématologie Oncologie Radiothérapie INSERM UMR-1160 Centre Hospitalo-Universitaire
Stopping TKI s in CML- Are we There Yet? Joseph O. Moore, MD Duke Cancer Institute
 Stopping TKI s in CML- Are we There Yet? Joseph O. Moore, MD Duke Cancer Institute Natural History of CML Accumulation of immature myeloid cells New cytogenetic changes Chronic Phase Accelerated Phase
Stopping TKI s in CML- Are we There Yet? Joseph O. Moore, MD Duke Cancer Institute Natural History of CML Accumulation of immature myeloid cells New cytogenetic changes Chronic Phase Accelerated Phase
CML and Future Perspective. Hani Al-Hashmi, MD
 CML and Future Perspective Hani Al-Hashmi, MD Objectives Learning from CML history Outcome of interest to clinician Patient and community interest!! Learning from CML history Survival Probability (All
CML and Future Perspective Hani Al-Hashmi, MD Objectives Learning from CML history Outcome of interest to clinician Patient and community interest!! Learning from CML history Survival Probability (All
Juan Luis Steegmann Hospital de la Princesa. Madrid. JL Steegmann
 Juan Luis Steegmann Hospital de la Princesa. Madrid. Juan Luis Steegmann Hospital de la Princesa. Madrid No rush,at least in Chronic Phase Blast Phase*: SCT asap, after restablishing CP with TKI Accelerated
Juan Luis Steegmann Hospital de la Princesa. Madrid. Juan Luis Steegmann Hospital de la Princesa. Madrid No rush,at least in Chronic Phase Blast Phase*: SCT asap, after restablishing CP with TKI Accelerated
Dose reduction. What do we know and how we do it in clinical practice. Andreas Hochhaus
 Dose reduction. What do we know and how we do it in clinical practice. Andreas Hochhaus Hadera I Oct 2018 Front-line Randomized Trials in CML-CP Trial Drugs References IRIS IM 400 vs IFN/AraC TOPS IM
Dose reduction. What do we know and how we do it in clinical practice. Andreas Hochhaus Hadera I Oct 2018 Front-line Randomized Trials in CML-CP Trial Drugs References IRIS IM 400 vs IFN/AraC TOPS IM
Imatinib & Ponatinib. Two ends of the spectrum in 2016s reality
 Imatinib & Ponatinib Two ends of the spectrum in 2016s reality CML 2016 Benefits & risks Steve O Brien CML Horizons, May 2016 Disclosures Research funding, participation in company trial, speaker, consultant,
Imatinib & Ponatinib Two ends of the spectrum in 2016s reality CML 2016 Benefits & risks Steve O Brien CML Horizons, May 2016 Disclosures Research funding, participation in company trial, speaker, consultant,
What is the optimal management strategy for younger CP-CML patients with matched, related donors who fail to achieve CCyR
 What is the optimal management strategy for younger CP-CML patients with matched, related donors who fail to achieve CCyR after 18 months of imatinib? Second generation TKIs as a bridge to allogeneic SCT
What is the optimal management strategy for younger CP-CML patients with matched, related donors who fail to achieve CCyR after 18 months of imatinib? Second generation TKIs as a bridge to allogeneic SCT
Treatment free remission in CML: from the concept to practice. François-Xavier Mahon. Cancer Center Bordeaux Université Bordeaux, France
 Treatment free remission in CML: from the concept to practice François-Xavier Mahon Cancer Center Bordeaux Université Bordeaux, France CML is a model 1960 1970 1980 1990 2000 2010 Philadelphia chromosome
Treatment free remission in CML: from the concept to practice François-Xavier Mahon Cancer Center Bordeaux Université Bordeaux, France CML is a model 1960 1970 1980 1990 2000 2010 Philadelphia chromosome
Stopping treatment how much we understand about mechanisms to stop successfully today, and where are the limits? Andreas Hochhaus
 Stopping treatment how much we understand about mechanisms to stop successfully today, and where are the limits? Andreas Hochhaus Frankfurt I 27.5.2017 Aims of CML Therapy Leukemia cells >10 12 CHR 10
Stopping treatment how much we understand about mechanisms to stop successfully today, and where are the limits? Andreas Hochhaus Frankfurt I 27.5.2017 Aims of CML Therapy Leukemia cells >10 12 CHR 10
Chronic myeloid leukemia (CML) Warunsuda Sripakdee, BCOP,BCP Prince of Songkla University
 Chronic myeloid leukemia (CML) 1 Warunsuda Sripakdee, BCOP,BCP Prince of Songkla University Hematologic malignancies CML ALL AML 2 CML CD34+ results from an acquired mutation that affects hematopoietic
Chronic myeloid leukemia (CML) 1 Warunsuda Sripakdee, BCOP,BCP Prince of Songkla University Hematologic malignancies CML ALL AML 2 CML CD34+ results from an acquired mutation that affects hematopoietic
Management of CML in blast crisis. Lymphoma Tumor Board November 27, 2015
 Management of CML in blast crisis Lymphoma Tumor Board November 27, 2015 Chronic Phase CML - 2. Peter Maslak, ASH Image Bank 2011; 2011-2455 Copyright 2011 American Society of Hematology. Copyright restrictions
Management of CML in blast crisis Lymphoma Tumor Board November 27, 2015 Chronic Phase CML - 2. Peter Maslak, ASH Image Bank 2011; 2011-2455 Copyright 2011 American Society of Hematology. Copyright restrictions
State of the Art Therapy and Monitoring of CML Hagop Kantarjian, M.D. Grand Rounds Hackensack, New Jersey. September 22, 2010
 State of the Art Therapy and Monitoring of CML - 2010 Hagop Kantarjian, M.D. Grand Rounds Hackensack, ew Jersey September 22, 2010 1 CML. Historical vs. Modern Perspective Parameter Historical Modern Course
State of the Art Therapy and Monitoring of CML - 2010 Hagop Kantarjian, M.D. Grand Rounds Hackensack, ew Jersey September 22, 2010 1 CML. Historical vs. Modern Perspective Parameter Historical Modern Course
CML UPDATE 2018 DAVID S. SNYDER, M.D. MARCH
 CML UPDATE 2018 DAVID S. SNYDER, M.D. MARCH 15, 2018 Click to edit Master Presentation Date DISCLOSURES I have nothing to disclose 2001 2016 Loss in expectation of life of patients with chronic myeloid
CML UPDATE 2018 DAVID S. SNYDER, M.D. MARCH 15, 2018 Click to edit Master Presentation Date DISCLOSURES I have nothing to disclose 2001 2016 Loss in expectation of life of patients with chronic myeloid
pan-canadian Oncology Drug Review Final Clinical Guidance Report Bosutinib (Bosulif) for Chronic Myelogenous Leukemia April 21, 2015
 pan-canadian Oncology Drug Review Final Clinical Guidance Report Bosutinib (Bosulif) for Chronic Myelogenous Leukemia April 21, 2015 DISCLAIMER Not a Substitute for Professional Advice This report is primarily
pan-canadian Oncology Drug Review Final Clinical Guidance Report Bosutinib (Bosulif) for Chronic Myelogenous Leukemia April 21, 2015 DISCLAIMER Not a Substitute for Professional Advice This report is primarily
I nuovi guariti? La malattia minima residua nella leucemia mieloide cronica. Fabrizio Pane
 I nuovi guariti? La malattia minima residua nella leucemia mieloide cronica Fabrizio Pane What could be the concept of Cure in CML? Sustained DMR with or without TKI therapy And 100% CML-related survival
I nuovi guariti? La malattia minima residua nella leucemia mieloide cronica Fabrizio Pane What could be the concept of Cure in CML? Sustained DMR with or without TKI therapy And 100% CML-related survival
AGGIORNAMENTI IN EMATOLOGIA Faenza, 7 Giugno 2018 LMC: ALGORITMI TERAPEUTICI ATTUALI E IL PROBLEMA DELLA RESISTENZA.
 AGGIORNAMENTI IN EMATOLOGIA Faenza, 7 Giugno 2018 LMC: ALGORITMI TERAPEUTICI ATTUALI E IL PROBLEMA DELLA RESISTENZA Michele.Baccarani@unibo.it EUROPEAN LEUKEMIANET 2013 (Blood 2013;122:885 892). RESPONSE
AGGIORNAMENTI IN EMATOLOGIA Faenza, 7 Giugno 2018 LMC: ALGORITMI TERAPEUTICI ATTUALI E IL PROBLEMA DELLA RESISTENZA Michele.Baccarani@unibo.it EUROPEAN LEUKEMIANET 2013 (Blood 2013;122:885 892). RESPONSE
Does Generic Imatinib Change the Treatment Approach in CML?
 Does Generic Imatinib Change the Treatment Approach in CML? Jerald P. Radich, MD Fred Hutchinson Cancer Research Center/ Seattle Cancer Care Alliance NCCN.org For Clinicians NCCN.org/patients For Patients
Does Generic Imatinib Change the Treatment Approach in CML? Jerald P. Radich, MD Fred Hutchinson Cancer Research Center/ Seattle Cancer Care Alliance NCCN.org For Clinicians NCCN.org/patients For Patients
CML TREATMENT GUIDELINES
 CML TREATMENT GUIDELINES INITIAL INVESTIGATION Propose enrolment in the CML Registry of the CML-MPN Quebec Research Group. Medical history : Question for cardio-respiratory disorders, diabetes, pancreatitis,
CML TREATMENT GUIDELINES INITIAL INVESTIGATION Propose enrolment in the CML Registry of the CML-MPN Quebec Research Group. Medical history : Question for cardio-respiratory disorders, diabetes, pancreatitis,
La terapia della LMC: è possibile guarire senza trapianto? Fabrizio Pane
 La terapia della LMC: è possibile guarire senza trapianto? Fabrizio Pane What could be the concept of Cure in CML? Sustained DMR with or without TKI therapy And 100% CML-related survival And QoL comparable
La terapia della LMC: è possibile guarire senza trapianto? Fabrizio Pane What could be the concept of Cure in CML? Sustained DMR with or without TKI therapy And 100% CML-related survival And QoL comparable
How I treat high risck CML
 Torino, September 14, 2018 How I treat high risck CML Patrizia Pregno Hematology Dept. Citta della Salute e della Scienza Torino Disclosures Advisory Board: Novartis, Pfizer, Incyte Speaker Honoraria:
Torino, September 14, 2018 How I treat high risck CML Patrizia Pregno Hematology Dept. Citta della Salute e della Scienza Torino Disclosures Advisory Board: Novartis, Pfizer, Incyte Speaker Honoraria:
Accepted Manuscript. Improving Outcomes in Chronic Myeloid Leukemia Over Time in the Era of Tyrosine Kinase Inhibitors. Pradnya Chopade, Luke P.
 Accepted Manuscript Improving Outcomes in Chronic Myeloid Leukemia Over Time in the Era of Tyrosine Kinase Inhibitors Pradnya Chopade, Luke P. Akard PII: S2152-2650(18)30343-4 DOI: 10.1016/j.clml.2018.06.029
Accepted Manuscript Improving Outcomes in Chronic Myeloid Leukemia Over Time in the Era of Tyrosine Kinase Inhibitors Pradnya Chopade, Luke P. Akard PII: S2152-2650(18)30343-4 DOI: 10.1016/j.clml.2018.06.029
DAVID S. SNYDER, M.D.
 CML UPDATE 2019 DAVID S. SNYDER, M.D. MARCH 21,2019 Click to edit Master Presentation Date Disclosures I do not have anything to disclose 2001 2016 Loss in expectation of life of patients with chronic
CML UPDATE 2019 DAVID S. SNYDER, M.D. MARCH 21,2019 Click to edit Master Presentation Date Disclosures I do not have anything to disclose 2001 2016 Loss in expectation of life of patients with chronic
State of the Art Therapy and Monitoring of CML Hagop Kantarjian, M.D. Grand Rounds UT Southwestern. October 28, 2010
 State of the Art Therapy and Monitoring of CML - 2010 Hagop Kantarjian, M.D. Grand Rounds UT Southwestern October 28, 2010 1 CML. Historical vs. Modern Perspective Parameter Historical Modern Course Fatal
State of the Art Therapy and Monitoring of CML - 2010 Hagop Kantarjian, M.D. Grand Rounds UT Southwestern October 28, 2010 1 CML. Historical vs. Modern Perspective Parameter Historical Modern Course Fatal
Allogeneic SCT for. 1st TKI. Vienna Austria. Dr. Eduardo Olavarría Complejo Hospitalario de Navarra
 The International Congress on Controversies in Stem Cell Transplantation and Cellular Therapies (COSTEM) Berlin, Germany September 8-11, 2011 Vienna Austria Allogeneic SCT for CML Allogeneic after failure
The International Congress on Controversies in Stem Cell Transplantation and Cellular Therapies (COSTEM) Berlin, Germany September 8-11, 2011 Vienna Austria Allogeneic SCT for CML Allogeneic after failure
A 34-year old women came because of abdominal discomfort. Vital sign was stable. Spleen tip was palpable.
 1 Case 1 A 34-year old women came because of abdominal discomfort. Vital sign was stable. Spleen tip was palpable. CBC and bone marrow aspiration and biopsy were done. Chromosome study showed she had t(9;22)
1 Case 1 A 34-year old women came because of abdominal discomfort. Vital sign was stable. Spleen tip was palpable. CBC and bone marrow aspiration and biopsy were done. Chromosome study showed she had t(9;22)
Welcome and Introductions
 Living with Chronic Myeloid Leukemia Welcome and Introductions Living with Chronic Myeloid Leukemia Living with Chronic Myeloid Leukemia (CML) Neil P. Shah, MD, PhD Edward S. Ageno Distinguished Professor
Living with Chronic Myeloid Leukemia Welcome and Introductions Living with Chronic Myeloid Leukemia Living with Chronic Myeloid Leukemia (CML) Neil P. Shah, MD, PhD Edward S. Ageno Distinguished Professor
Oncology Highlights: Leukemia & Myelodysplastic Syndromes
 Oncology Highlights: Leukemia & Myelodysplastic Syndromes Jorge Cortes, MD Department of Leukemia The University of Texas, M.D. Anderson Cancer Center Highlights of the Day Leukemia & MDS AML: The field
Oncology Highlights: Leukemia & Myelodysplastic Syndromes Jorge Cortes, MD Department of Leukemia The University of Texas, M.D. Anderson Cancer Center Highlights of the Day Leukemia & MDS AML: The field
Current Monitoring for CML: Goals and. Jorge Cortes, MD Chief, CML & AML Section Department of Leukemia MD Anderson Cancer Center
 Current Monitoring for CML: Goals and Principles Jorge Cortes, MD Chief, CML & AML Section Department of Leukemia MD Anderson Cancer Center Survival in Early Chronic Phase CML MDACC 2009 The Philadelphia
Current Monitoring for CML: Goals and Principles Jorge Cortes, MD Chief, CML & AML Section Department of Leukemia MD Anderson Cancer Center Survival in Early Chronic Phase CML MDACC 2009 The Philadelphia
EUROPEAN LEUKEMIANET RECOMMENDATIONS FOR CHRONIC MYELOID LEUKEMIA
 EUROPEAN LEUKEMIANET RECOMMENDATIONS FOR CHRONIC MYELOID LEUKEMIA SAN DIEGO, 11 DECEMBER 2011 AMSTERDAM, 14 JUNE 2012 BALTIMORE, 20 SEPTEMBER 2012 ATLANTA, 6 DECEMBER 2012 ELN, CML Panel Jane Apperley
EUROPEAN LEUKEMIANET RECOMMENDATIONS FOR CHRONIC MYELOID LEUKEMIA SAN DIEGO, 11 DECEMBER 2011 AMSTERDAM, 14 JUNE 2012 BALTIMORE, 20 SEPTEMBER 2012 ATLANTA, 6 DECEMBER 2012 ELN, CML Panel Jane Apperley
10 YEARS EXPERIENCE OF TYROSINE KINASE INHIBITOR THERAPY FOR CML IN OXFORD
 10 YEARS EXPERIENCE OF TYROSINE KINASE INHIBITOR THERAPY FOR CML IN OXFORD Dalia Khan 1, Noemi Roy 1, Vasha Bari 1, Grant Vallance 1, Helene Dreau 1, Timothy Littlewood 1, Andrew Peniket 1, Paresh Vyas
10 YEARS EXPERIENCE OF TYROSINE KINASE INHIBITOR THERAPY FOR CML IN OXFORD Dalia Khan 1, Noemi Roy 1, Vasha Bari 1, Grant Vallance 1, Helene Dreau 1, Timothy Littlewood 1, Andrew Peniket 1, Paresh Vyas
CML Clinical Case Scenario
 CML Clinical Case Scenario Neil Shah, MD, PhD Edward S. Ageno Distinguished Professor in Hematology/Oncology Leader, Hematopoietic Malignancies Program Helen Diller Family Comprehensive Cancer Center at
CML Clinical Case Scenario Neil Shah, MD, PhD Edward S. Ageno Distinguished Professor in Hematology/Oncology Leader, Hematopoietic Malignancies Program Helen Diller Family Comprehensive Cancer Center at
Imatinib dose intensification, combination therapies. Andreas Hochhaus Universitätsklinikum Jena, Germany
 Imatinib dose intensification, combination therapies Andreas Hochhaus Universitätsklinikum Jena, Germany Apperley JF. Lancet Oncol. 2007 High OCT-1 activity is associated with faster MMR in imatinib treated
Imatinib dose intensification, combination therapies Andreas Hochhaus Universitätsklinikum Jena, Germany Apperley JF. Lancet Oncol. 2007 High OCT-1 activity is associated with faster MMR in imatinib treated
NEW DRUGS IN HEMATOLOGY Bologna, 9-11 May 2016 CHRONIC MYELOID LEUKEMIA STATUS OF THE ART OF TREATMENT.
 NEW DRUGS IN HEMATOLOGY Bologna, 9-11 May 2016 CHRONIC MYELOID LEUKEMIA STATUS OF THE ART OF TREATMENT Michele.baccarani@unibo.it Michele BACCARANI, MD Professor of Hematology at the Universities of Trieste,
NEW DRUGS IN HEMATOLOGY Bologna, 9-11 May 2016 CHRONIC MYELOID LEUKEMIA STATUS OF THE ART OF TREATMENT Michele.baccarani@unibo.it Michele BACCARANI, MD Professor of Hematology at the Universities of Trieste,
Supplementary Online Content
 Supplementary Online Content Douxfils J, Haguet H, Mullier F, Chatelain C, Graux C, Dogné J-M. Use of BCR-ABL tyrosine kinase inhibitors in chronic myeloid leukemia: a systematic review and metaanalysis
Supplementary Online Content Douxfils J, Haguet H, Mullier F, Chatelain C, Graux C, Dogné J-M. Use of BCR-ABL tyrosine kinase inhibitors in chronic myeloid leukemia: a systematic review and metaanalysis
Study Design and Endpoints
 Complete Molecular Response (CMR) Rate With Nilotinib in Patients With CML-CP Without CMR After 2 Years on Imatinib: Preliminary Results From the Randomized ENESTcmr Trial Timothy P. Hughes, Jeffrey H.
Complete Molecular Response (CMR) Rate With Nilotinib in Patients With CML-CP Without CMR After 2 Years on Imatinib: Preliminary Results From the Randomized ENESTcmr Trial Timothy P. Hughes, Jeffrey H.
History of CML Treatment
 History of CML Treatment Eduardo Olavarria No conflict of interest Lisbon, 20th March 2018 #EBMT18 www.ebmt.or What is CML? The mystery of chronic myeloid leukaemia Chronic myeloid leukaemia Often diagnosed
History of CML Treatment Eduardo Olavarria No conflict of interest Lisbon, 20th March 2018 #EBMT18 www.ebmt.or What is CML? The mystery of chronic myeloid leukaemia Chronic myeloid leukaemia Often diagnosed
CML Update 2016 Arthur 2016
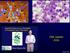 CML Update 2016 Chronic Myeloid Leukemia Splenomegaly CML (3 phase disease) Increased white cells Malignant proliferation of myeloid white cells Initially mature cells a) chronic phase of disease Evolution
CML Update 2016 Chronic Myeloid Leukemia Splenomegaly CML (3 phase disease) Increased white cells Malignant proliferation of myeloid white cells Initially mature cells a) chronic phase of disease Evolution
Venice Meeting Highlights: Key lessons. Conclusions Michele Baccarani Rüdiger Hehlmann
 Venice Meeting Highlights: Key lessons Conclusions Michele Baccarani Rüdiger Hehlmann CML therapy in the imatinib era CML prognosis has improved dramatically Cellular and molecular biology studies help
Venice Meeting Highlights: Key lessons Conclusions Michele Baccarani Rüdiger Hehlmann CML therapy in the imatinib era CML prognosis has improved dramatically Cellular and molecular biology studies help
ARIAD Pharmaceuticals, Inc.
 ARIAD Pharmaceuticals, Inc. June 8, 2016 David Sachs Non-small cell lung cancer 1 ARIAD clinical trial patient Some of the statements in this presentation constitute forward looking statements under the
ARIAD Pharmaceuticals, Inc. June 8, 2016 David Sachs Non-small cell lung cancer 1 ARIAD clinical trial patient Some of the statements in this presentation constitute forward looking statements under the
What Can We Expect from Imatinib? CML Case Presentation. Presenter Disclosure Information. CML Case Presentation (cont)? Session 2: 8:15 AM - 9:00 AM
 Welcome to Master Class for Oncologists Session 2: 8:15 AM - 9: AM Miami, FL December 18, 29 Chronic Myelocytic Leukemia: Imatinib and Beyond Speaker: Daniel J. DeAngelo, MD, PhD Dana-Farber Cancer Institute
Welcome to Master Class for Oncologists Session 2: 8:15 AM - 9: AM Miami, FL December 18, 29 Chronic Myelocytic Leukemia: Imatinib and Beyond Speaker: Daniel J. DeAngelo, MD, PhD Dana-Farber Cancer Institute
The speaker has no financial relationships with a commercial interest to disclose and no conflicts of interest to resolve.
 Hana Safah MD Professor of Medicine Tulane University School of Medicine Director of the SCT program, Tulane Medical Center The speaker has no financial relationships with a commercial interest to disclose
Hana Safah MD Professor of Medicine Tulane University School of Medicine Director of the SCT program, Tulane Medical Center The speaker has no financial relationships with a commercial interest to disclose
517 Spirit 2: An NCRI Randomised Study Comparing Dasatinib with Imatinib in Patients with Newly Diagnosed CML
 517 Spirit 2: An NCRI Randomised Study Comparing Dasatinib with Imatinib in Patients with Newly Diagnosed CML https://ash.confex.com/ash/2014/webprogram/paper66809.html Objective. SPIRIT 2 is the largest
517 Spirit 2: An NCRI Randomised Study Comparing Dasatinib with Imatinib in Patients with Newly Diagnosed CML https://ash.confex.com/ash/2014/webprogram/paper66809.html Objective. SPIRIT 2 is the largest
What is New in Leukemia & MPN in 2011?
 Leukemia Update What Did We Learn from ASCO 211? Jorge Cortes, MD Department of Leukemia MD Anderson Cancer Center Houston, Texas What is New in Leukemia & MPN in 211? Cool things happening in MPN The
Leukemia Update What Did We Learn from ASCO 211? Jorge Cortes, MD Department of Leukemia MD Anderson Cancer Center Houston, Texas What is New in Leukemia & MPN in 211? Cool things happening in MPN The
Discontinuation of Tyrosine Kinase Inhibitors in Chronic Myeloid Leukemia: What s Stopping us from Stopping?
 Discontinuation of Tyrosine Kinase Inhibitors in Chronic Myeloid Leukemia: What s Stopping us from Stopping? David Pham, PharmD PGY2 Hematology/Oncology Pharmacy Resident South Texas VA Health Care System
Discontinuation of Tyrosine Kinase Inhibitors in Chronic Myeloid Leukemia: What s Stopping us from Stopping? David Pham, PharmD PGY2 Hematology/Oncology Pharmacy Resident South Texas VA Health Care System
pan-canadian Oncology Drug Review Final Clinical Guidance Report Ponatinib (Iclusig) for Chronic Myeloid Leukemia / Acute Lymphoblastic Leukemia
 pan-canadian Oncology Drug Review Final Clinical Guidance Report Ponatinib (Iclusig) for Chronic Myeloid Leukemia / Acute Lymphoblastic Leukemia October 1, 2015 DISCLAIMER Not a Substitute for Professional
pan-canadian Oncology Drug Review Final Clinical Guidance Report Ponatinib (Iclusig) for Chronic Myeloid Leukemia / Acute Lymphoblastic Leukemia October 1, 2015 DISCLAIMER Not a Substitute for Professional
Approval based on the successful BFORE Phase 3 study conducted by Avillion under a collaborative development agreement with Pfizer
 Avillion Announces US Approval of Pfizer s BOSULIF (bosutinib) for the Treatment of Patients with Newly-Diagnosed Ph+ Chronic Myelogenous Leukemia (CML) Approval based on the successful BFORE Phase 3 study
Avillion Announces US Approval of Pfizer s BOSULIF (bosutinib) for the Treatment of Patients with Newly-Diagnosed Ph+ Chronic Myelogenous Leukemia (CML) Approval based on the successful BFORE Phase 3 study
Second-generation BCR-ABL inhibitors for frontline treatment of chronic myeloid leukemia in chronic phase
 Critical Reviews in Oncology/Hematology 82 (2012) 159 170 Second-generation BCR-ABL inhibitors for frontline treatment of chronic myeloid leukemia in chronic phase Gianantonio Rosti, Fausto Castagnetti,
Critical Reviews in Oncology/Hematology 82 (2012) 159 170 Second-generation BCR-ABL inhibitors for frontline treatment of chronic myeloid leukemia in chronic phase Gianantonio Rosti, Fausto Castagnetti,
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert.
 Public Disclosure Synopsis Protocol 316A4-223 (B18717) 11 October 216 Final PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved
Public Disclosure Synopsis Protocol 316A4-223 (B18717) 11 October 216 Final PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved
Post ASH Actualités LMC
 Post ASH 2014 - Actualités LMC Actualités de première ligne LMC PC ENESTnd (6 ans) Dasision (5 ans) EPIC Spirit France (5 ans) Spirit 2 UK (5 ans) ENESTnd: Mise à jour à 6 ans Design = 846 ults with wly
Post ASH 2014 - Actualités LMC Actualités de première ligne LMC PC ENESTnd (6 ans) Dasision (5 ans) EPIC Spirit France (5 ans) Spirit 2 UK (5 ans) ENESTnd: Mise à jour à 6 ans Design = 846 ults with wly
CML EHA: what s new? Novità dall EHA >> [ Leucemia mieloide cronica ] Relatore: G. MARTINELLI. Borgo S. Luigi Monteriggioni (Siena) ottobre 2008
![CML EHA: what s new? Novità dall EHA >> [ Leucemia mieloide cronica ] Relatore: G. MARTINELLI. Borgo S. Luigi Monteriggioni (Siena) ottobre 2008 CML EHA: what s new? Novità dall EHA >> [ Leucemia mieloide cronica ] Relatore: G. MARTINELLI. Borgo S. Luigi Monteriggioni (Siena) ottobre 2008](/thumbs/75/71485931.jpg) Novità dall EHA >> [ Leucemia mieloide cronica ] CML EHA: what s new? Relatore: G. MARTINELLI 27-28 ottobre 2008 Borgo S. Luigi Monteriggioni (Siena) Leucemia mieloide cronica - Copyright FSE 1 CML EHA:
Novità dall EHA >> [ Leucemia mieloide cronica ] CML EHA: what s new? Relatore: G. MARTINELLI 27-28 ottobre 2008 Borgo S. Luigi Monteriggioni (Siena) Leucemia mieloide cronica - Copyright FSE 1 CML EHA:
Updated review of nilotinib as frontline treatment for newly diagnosed Philadelphia chromosome-positive chronic myeloid leukemia
 For reprint orders, please contact: reprints@futuremedicine.com Updated review of nilotinib as frontline treatment for newly diagnosed Philadelphia chromosome-positive chronic myeloid leukemia Nilotinib,
For reprint orders, please contact: reprints@futuremedicine.com Updated review of nilotinib as frontline treatment for newly diagnosed Philadelphia chromosome-positive chronic myeloid leukemia Nilotinib,
Practical Guidance for the Management of CML in 2016
 Practical Guidance for the Management of CML in 2016 Neil Shah, MD, PhD Edward S. Ageno Distinguished Professor in Hematology/Oncology Leader, Hematopoietic Malignancies Program Helen Diller Family Comprehensive
Practical Guidance for the Management of CML in 2016 Neil Shah, MD, PhD Edward S. Ageno Distinguished Professor in Hematology/Oncology Leader, Hematopoietic Malignancies Program Helen Diller Family Comprehensive
RESEARCH ARTICLE. Introduction. Methods Wiley Periodicals, Inc.
 BCR/ABL level at 6 months identifies good risk CML subgroup after failing early molecular response at 3 months following imatinib therapy for CML in chronic phase AJH Dennis (Dong Hwan) Kim, 1 * Nada Hamad,
BCR/ABL level at 6 months identifies good risk CML subgroup after failing early molecular response at 3 months following imatinib therapy for CML in chronic phase AJH Dennis (Dong Hwan) Kim, 1 * Nada Hamad,
CML: definition. CML epidemiology. CML diagnosis. CML: peripheralbloodsmear. Cytogenetic abnormality of CML
 MolecularDiagnostic.be Third Scientific Meeting Molecular Diagnostics.be t(9;22) CML: definition Management of CML patients treated with TKI: the place of molecular monitoring Antwerp, December 13 th 11
MolecularDiagnostic.be Third Scientific Meeting Molecular Diagnostics.be t(9;22) CML: definition Management of CML patients treated with TKI: the place of molecular monitoring Antwerp, December 13 th 11
NCCP Chemotherapy Protocol. Bosutinib Monotherapy
 Bosutinib Monotherapy INDICATIONS FOR USE: INDICATION Treatment of adult patients with chronic phase (CP), accelerated phase (AP), and blast phase (BP) Philadelphia chromosome positive chronic myelogenous
Bosutinib Monotherapy INDICATIONS FOR USE: INDICATION Treatment of adult patients with chronic phase (CP), accelerated phase (AP), and blast phase (BP) Philadelphia chromosome positive chronic myelogenous
Chronic Myeloid Leukemia A Disease of Young at Heart but Not of Body
 Chronic Myeloid Leukemia A Disease of Young at Heart but Not of Body Jeffrey H Lipton, PhD MD FRCPC Staff Physician, Princess Margaret Cancer Centre Professor of Medicine University of Toronto POGO November,
Chronic Myeloid Leukemia A Disease of Young at Heart but Not of Body Jeffrey H Lipton, PhD MD FRCPC Staff Physician, Princess Margaret Cancer Centre Professor of Medicine University of Toronto POGO November,
Milestones and Monitoring
 Curr Hematol Malig Rep (2015) 10:167 172 DOI 10.1007/s11899-015-0258-1 CHRONIC MYELOID LEUKEMIAS (E JABBOUR, SECTION EDITOR) Milestones and Monitoring Alessandro Morotti 1 & Carmen Fava 1 & Giuseppe Saglio
Curr Hematol Malig Rep (2015) 10:167 172 DOI 10.1007/s11899-015-0258-1 CHRONIC MYELOID LEUKEMIAS (E JABBOUR, SECTION EDITOR) Milestones and Monitoring Alessandro Morotti 1 & Carmen Fava 1 & Giuseppe Saglio
Blast Phase Chronic Myelogenous Leukemia
 Blast Phase Chronic Myelogenous Leukemia Benjamin Powers, MD; and Suman Kambhampati, MD The dramatic improvement in survival with tyrosine kinase inhibitors has not been demonstrated in the advanced blast
Blast Phase Chronic Myelogenous Leukemia Benjamin Powers, MD; and Suman Kambhampati, MD The dramatic improvement in survival with tyrosine kinase inhibitors has not been demonstrated in the advanced blast
Dati sulla sospensione della terapia
 Leucemia Mieloide Cronica Dati sulla sospensione della terapia Gianantonio Rosti, Bologna BCR-ABL Loading in CML Patients 100% 10% 1% MMR MR 4 MR 4.5 STIM study design N=100 Start Imatinib CMR Sustained
Leucemia Mieloide Cronica Dati sulla sospensione della terapia Gianantonio Rosti, Bologna BCR-ABL Loading in CML Patients 100% 10% 1% MMR MR 4 MR 4.5 STIM study design N=100 Start Imatinib CMR Sustained
15 th Annual Miami Cancer Meeting
 15 th Annual Miami Cancer Meeting CLL and CML Mohamed A. Kharfan-Dabaja, MD, MBA, FACP Director, Blood and Marrow Transplantation and Cellular Therapies Mayo Clinic Jacksonville, FL 15 th Annual Miami
15 th Annual Miami Cancer Meeting CLL and CML Mohamed A. Kharfan-Dabaja, MD, MBA, FACP Director, Blood and Marrow Transplantation and Cellular Therapies Mayo Clinic Jacksonville, FL 15 th Annual Miami
Summary 1. Comparative effectiveness of ponatinib
 Cost-effectiveness of ponatinib (Iclusig ) as indicated for adult patients with: (i) CP-, AP-, or BP- chronic myeloid leukaemia (CML) who are resistant to dasatinib or nilotinib; who are intolerant to
Cost-effectiveness of ponatinib (Iclusig ) as indicated for adult patients with: (i) CP-, AP-, or BP- chronic myeloid leukaemia (CML) who are resistant to dasatinib or nilotinib; who are intolerant to
1794 Updating Long-Term Outcome of Intermittent Imatinib. (INTERIM) Treatment in Elderly Patients with Ph+-CML
 738 What Is the Most Cost-Effective Strategy for Treating Newly Diagnosed Chronic Phase Chronic Myeloid Leukaemia (CML) after Imatinib Loses Patent Exclusivity? https://ash.confex.com/ash/2014/webprogram/paper71699.html
738 What Is the Most Cost-Effective Strategy for Treating Newly Diagnosed Chronic Phase Chronic Myeloid Leukaemia (CML) after Imatinib Loses Patent Exclusivity? https://ash.confex.com/ash/2014/webprogram/paper71699.html
NEW DRUGS IN HEMATOLOGY
 NEW DRUGS IN HEMATOLOGY BOLOGNA, 15-17 April 2013 TYROSINE KINASE INHIBITORS IN CHRONIC MYELOID LEUKEMIA MICHELE BACCARANI michele.baccarani@unibo.it Historic Development of CML Therapy Palliative Therapy
NEW DRUGS IN HEMATOLOGY BOLOGNA, 15-17 April 2013 TYROSINE KINASE INHIBITORS IN CHRONIC MYELOID LEUKEMIA MICHELE BACCARANI michele.baccarani@unibo.it Historic Development of CML Therapy Palliative Therapy
Impact of Age on Efficacy and Toxicity of Nilotinib in Patients With Chronic Myeloid Leukemia in Chronic Phase (CML-CP): ENEST1st Sub-Analysis
 Impact of Age on Efficacy and Toxicity of Nilotinib in Patients With Chronic Myeloid Leukemia in Chronic Phase (CML-CP): ENEST1st Sub-Analysis Abstract 479 Giles FJ, Rea D, Baccarani M, Cross NCP, Steegmann
Impact of Age on Efficacy and Toxicity of Nilotinib in Patients With Chronic Myeloid Leukemia in Chronic Phase (CML-CP): ENEST1st Sub-Analysis Abstract 479 Giles FJ, Rea D, Baccarani M, Cross NCP, Steegmann
C Longer follow up on IRIS data
 hronic Myeloid Leukemia Drs. Rena Buckstein, Mervat Mahrous & Eugenia Piliotis with input from Dr. J. Lipton (PMH) Updated August 2008* Updates: C Longer follow up on IRIS data Guidelines for monitoring
hronic Myeloid Leukemia Drs. Rena Buckstein, Mervat Mahrous & Eugenia Piliotis with input from Dr. J. Lipton (PMH) Updated August 2008* Updates: C Longer follow up on IRIS data Guidelines for monitoring
Abstracts and notes on CML presentations 1 ASH 2012 Atlanta
 Abstracts and notes on CML presentations 1 ASH 2012 Atlanta Steve O Brien, Newcastle University 1 Highlights... 1 2 Plenary session [3]... 4 3 CML Therapy I [163-168]... 4 4 CML Therapy II [913-918]...
Abstracts and notes on CML presentations 1 ASH 2012 Atlanta Steve O Brien, Newcastle University 1 Highlights... 1 2 Plenary session [3]... 4 3 CML Therapy I [163-168]... 4 4 CML Therapy II [913-918]...
Is there a best TKI for chronic phase CML?
 MANAGING TYPICAL AND ATYPICAL CHRONIC MYELOID LEUKEMIA Is there a best TKI for chronic phase CML? Richard A. Larson 1 1 Section of Hematology/Oncology, Department of Medicine, and Comprehensive Cancer
MANAGING TYPICAL AND ATYPICAL CHRONIC MYELOID LEUKEMIA Is there a best TKI for chronic phase CML? Richard A. Larson 1 1 Section of Hematology/Oncology, Department of Medicine, and Comprehensive Cancer
The current standard of care in CML. Gianantonio Rosti, MD University of Bologna Bologna, Italy
 The current standard of care in CML Gianantonio Rosti, MD University of Bologna Bologna, Italy CML: Overall Survival 1898-1977 just to remember Clinical Landmarks in CML 1845 1865 1879 1903 1953 1965 1968
The current standard of care in CML Gianantonio Rosti, MD University of Bologna Bologna, Italy CML: Overall Survival 1898-1977 just to remember Clinical Landmarks in CML 1845 1865 1879 1903 1953 1965 1968
Executive summary Overview
 Executive summary Overview In this appraisal, we have demonstrated that dasatinib is clinically more effective, as well as more cost effective, than imatinib, the current standard of care. In the pivotal
Executive summary Overview In this appraisal, we have demonstrated that dasatinib is clinically more effective, as well as more cost effective, than imatinib, the current standard of care. In the pivotal
Chronic myeloid leukemia: 2018 update on diagnosis, therapy and monitoring
 Received: 13 December 2017 Accepted: 17 December 2017 DOI: 10.1002/ajh.25011 ANNUAL CLINICAL UPDATES IN HEMATOLOGICAL MALIGNANCIES Chronic myeloid leukemia: 2018 update on diagnosis, therapy and monitoring
Received: 13 December 2017 Accepted: 17 December 2017 DOI: 10.1002/ajh.25011 ANNUAL CLINICAL UPDATES IN HEMATOLOGICAL MALIGNANCIES Chronic myeloid leukemia: 2018 update on diagnosis, therapy and monitoring
MRD in CML (BCR-ABL1)
 MRD in CML (BCR-ABL1) Moleculaire Biologie en Cytometrie cursus Barbara Denys LAbo Hematologie UZ Gent 6 mei 2011 2008 Universitair Ziekenhuis Gent 1 Myeloproliferative Neoplasms o WHO classification 2008:
MRD in CML (BCR-ABL1) Moleculaire Biologie en Cytometrie cursus Barbara Denys LAbo Hematologie UZ Gent 6 mei 2011 2008 Universitair Ziekenhuis Gent 1 Myeloproliferative Neoplasms o WHO classification 2008:
When to change therapy? Andreas Hochhaus Universitätsklinikum Jena, Germany
 When to change therapy? Andreas Hochhaus Universitätsklinikum Jena, Germany Chromosome 22 Chromosome 9 e1 1b m-bcr M-bcr e1 e2 b1 b5 5 3 BCR ABL 5 3 1a a2 a3 μ -bcr e19 a11 e1a2 b2a2 b3a2 e19a2 p190 bcr-abl
When to change therapy? Andreas Hochhaus Universitätsklinikum Jena, Germany Chromosome 22 Chromosome 9 e1 1b m-bcr M-bcr e1 e2 b1 b5 5 3 BCR ABL 5 3 1a a2 a3 μ -bcr e19 a11 e1a2 b2a2 b3a2 e19a2 p190 bcr-abl
Molecular monitoring of CML patients
 EHA, Education Session, CML Stockholm, 14 June 2013 Molecular monitoring of CML patients Martin C. Müller Medical Faculty Mannheim Ruprecht-Karls-University Heidelberg Mannheim, Germany Disclosures Research
EHA, Education Session, CML Stockholm, 14 June 2013 Molecular monitoring of CML patients Martin C. Müller Medical Faculty Mannheim Ruprecht-Karls-University Heidelberg Mannheim, Germany Disclosures Research
Welcome & Introductions
 What s on the Horizon for Chronic Myeloid Leukemia? Welcome & Introductions Dr. Mauro s slides are available for download at www.lls.org/programs Wednesday, September 27, 2017 1 What s on the Horizon for
What s on the Horizon for Chronic Myeloid Leukemia? Welcome & Introductions Dr. Mauro s slides are available for download at www.lls.org/programs Wednesday, September 27, 2017 1 What s on the Horizon for
9/26/2018. Learning Objectives
 ADVANCES IN CHRONIC MYELOID LEUKEMIA Alison Wakoff Loren, MD, MSCE Associate Professor of Medicine Director, Blood & Marrow Transplantation Vice Chair, Faculty Development Department of Medicine Perelman
ADVANCES IN CHRONIC MYELOID LEUKEMIA Alison Wakoff Loren, MD, MSCE Associate Professor of Medicine Director, Blood & Marrow Transplantation Vice Chair, Faculty Development Department of Medicine Perelman
IS MUTATION ANALYSIS OF BCR-ABL OF ANY VALUE IN CLINICAL MANAGEMENT OF CML PATIENTS? David Marin, Imperial College London
 IS MUTATION ANALYSIS OF BCR-ABL OF ANY VALUE IN CLINICAL MANAGEMENT OF CML PATIENTS? David Marin, Imperial College London Tell me generals, are we politicians necessary? I have to admit defeat before starting
IS MUTATION ANALYSIS OF BCR-ABL OF ANY VALUE IN CLINICAL MANAGEMENT OF CML PATIENTS? David Marin, Imperial College London Tell me generals, are we politicians necessary? I have to admit defeat before starting
Outlook CML 2016: What is being done on the way to cure
 New Horizons 2011 Outlook CML 2016: What is being done on the way to cure Gianantonio Rosti Dept. Of Hematology and Oncology St. Orsola-Malpighi University Hospital Bologna (Italy) GIMEMA CML Working Party
New Horizons 2011 Outlook CML 2016: What is being done on the way to cure Gianantonio Rosti Dept. Of Hematology and Oncology St. Orsola-Malpighi University Hospital Bologna (Italy) GIMEMA CML Working Party
