YEARLY SUMMARY PATIENT INFORMATION
|
|
|
- Louisa Skinner
- 5 years ago
- Views:
Transcription
1 From: / / To: / / For RRC use only Form : Status: Person completing form: (please print) REFERRING PHYSICIAN Name: Institution Name: Institution Address: City/Village: State/Province: Zip/Postal Code: Telephone Number: ( ) ( ) Fax Number: ( ) ( ) Address PATIENT DETAILS Complete only if change from last information provided. Patient: Address: City/Village: State/Province: Zip/Postal Code: Country: Telephone Number: ( )( ) For RRC Sent: Data Review: use only Received: Entered: Clinical Review: Verified: 30Jun
2 From: / / To / / EXAMINATIONS AND SIGNIFICANT NON-INFECTIOUS CLINICAL EVENTS Bone marrow evaluation done AML/MDS Cytogenetics evaluation done Cytogenetic abnormality detected Bone density evaluation done Abnormal bone density/osteopenia/osteoporosis In vitro research testing done Glomerulonephritis Vasculitis Arthritis Splenectomy Date: Other significant non-infectious events including all hospitalizations (specify): Height assessed: or Date: cm in Weight assessed: or Date: kg lbs CBCs done during this time period (Please attach all reports) Patient pregnant during this time period or currently pregnant Patient died during this time period Date: Cause of death: _ 16Jan
3 From: / / To / / Mouth ulcers Skin abscesses SIGNIFICANT INFECTIOUS EPISODES 1-3 per 4-12 per > 12 per Year, Frequency ne Year Year Continuous Unknown Other significant infections (specify): IV Antibiotics administered ( if yes) TREATMENT Cytokine (growth factor, e.g., G-CSF) treatment during this time period Type: G-CSF GM-CSF EPO Other (specify): Current cytokine dose: Units*: Freq**: Brand Name: Units*: Freq**: Brand Name: Indicate typical dose range for this year: Was cytokine discontinued during this time period: *Units **Freq If yes, date discontinued: mcg qd Reason: Ineffective Pt. chose to withdraw mcg/kg bid Toxicity Neutrophil recovery ml qod n-compliant qtd Other, specify qwk Other treatments for neutropenia: Steroids Gammaglobulin Other, specify Bone marrow transplant BONE MARROW CELL BANK Next bone marrow exam planned? If yes: / mo yr Please plan to send bone marrow sample to cell bank every year. 30Jun
4 Patient Id Number / / / Patient Initials REPORT GLYCOGENOSIS TYPE IB BIRTH TYPE Single Identical twin Fraternal twin, gender: same different Other multiple: GSD-IB RELATED SYMPTOMS Unknown Asymptomatic Low birth weight Skeletal abnormalities : Muscular hypotonia Xanthomas or lipomas Malignant adenoma Epilepsy Dental problems : Growth retardation Failure to thrive Hematological abnormalities : Malignancy : Other dysfunction : Supplementary feeding : tube feeding percutaneous endoscopic gastrostomy (PEG) SERUM PARAMETERS C-Gluc/GD/e Lactate Alanine Creatinine Urea Cholesterol Triglycerides µmol/l Date [DD/MON/YY]: µmol/l Date [DD/MON/YY]: RADIOLOGY RESULTS (please attach reports) Pancreas CT U/S rmal Abnormal: Liver CT U/S rmal Abnormal: Ribs rmal Abnormal: Long bones rmal Abnormal: Dental radiology rmal Abnormal:
5 PSYCHOLOGY Overall functioning rmal Concerns: Concentration power rmal Concerns: Mental development rmal Concerns: General behaviour rmal Concerns: Social competence rmal Concerns: Other issues Genotype t tested Tested GENOTYPE (if not reported previously) Mutation of G6P-Transporter Gene:
REGISTRATION PATIENT DETAILS
 PATIENT DETAILS Person completing form: (please print) FOR REGIONAL REFERRAL CENTER USE ONLY Registration: Approved Date: / / Not approved RRC telephone contact with primary MD: Date: / / Reviewers Signature:
PATIENT DETAILS Person completing form: (please print) FOR REGIONAL REFERRAL CENTER USE ONLY Registration: Approved Date: / / Not approved RRC telephone contact with primary MD: Date: / / Reviewers Signature:
REGISTRATION PATIENT DETAILS
 PATIENT DETAILS Person completing form: (please print) FOR REGIONAL REFERRAL CENTER USE ONLY Registration: Approved Date: / / Not approved RRC telephone contact with primary MD: Date: / / Reviewers Signature:
PATIENT DETAILS Person completing form: (please print) FOR REGIONAL REFERRAL CENTER USE ONLY Registration: Approved Date: / / Not approved RRC telephone contact with primary MD: Date: / / Reviewers Signature:
SCNIR 1107 NE 45 th St, Suite #345 Seattle, WA REGISTRATION FORM. Reviewer s Signature: PHYSICIAN CONTACT INFORMATION
 Physician Name: Institution Name: Institution Address: FOR OFFICE USE ONLY Registration: Approved Date: Not approved Reviewer s Signature: PHYSICIAN CONTACT INFORMATION Specialty: City: State/Province:
Physician Name: Institution Name: Institution Address: FOR OFFICE USE ONLY Registration: Approved Date: Not approved Reviewer s Signature: PHYSICIAN CONTACT INFORMATION Specialty: City: State/Province:
Division of AIDS Safety Office EXPEDITED ADVERSE EVENT (EAE) Form
 To: DAIDS SAFETY OFFICE Division of AIDS Safety Office EXPEDITED ADVERSE EVENT (EAE) Form Sent by: Please type or print in English Fax: 1-800-275-7619 (USA) or + 1-301-897-1710 (International) Phone: 1-800-537-9979
To: DAIDS SAFETY OFFICE Division of AIDS Safety Office EXPEDITED ADVERSE EVENT (EAE) Form Sent by: Please type or print in English Fax: 1-800-275-7619 (USA) or + 1-301-897-1710 (International) Phone: 1-800-537-9979
Different Types of Cancer
 Different Types of Cancer Cancer can originate almost anywhere in the body. Sarcomas (connective tissue) Ø arise from cells found in the supporting tissues of the body such as bone, cartilage, fat, connective
Different Types of Cancer Cancer can originate almost anywhere in the body. Sarcomas (connective tissue) Ø arise from cells found in the supporting tissues of the body such as bone, cartilage, fat, connective
2. Is therapy prescribed by, or in consultation with, a hematologist and/or oncologist?
 Pharmacy Prior Authorization AETA BETTER HEALTH EW JERSE (MEDICAID) Colony Stimulating Factors (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review
Pharmacy Prior Authorization AETA BETTER HEALTH EW JERSE (MEDICAID) Colony Stimulating Factors (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review
Form 2033 R3.0: Wiskott-Aldrich Syndrome Pre-HSCT Data
 Key Fields Sequence Number: Date Received: - - CIBMTR Center Number: CIBMTR Recipient ID: Has this patient's data been previously reported to USIDNET? USIDNET ID: Today's Date: - - Date of HSCT for which
Key Fields Sequence Number: Date Received: - - CIBMTR Center Number: CIBMTR Recipient ID: Has this patient's data been previously reported to USIDNET? USIDNET ID: Today's Date: - - Date of HSCT for which
11/5/2011. Disclosures. Key References. JAK Inhibitors. Tofacitinib(CP ): Phase 2 Studies
 Disclosures KINASE INHIBITORS IN RA Michael E. Weinblatt, M.D. Dr. Weinblatt has been a consultant to the following companies involved in the JAK and SYK pathways in RA Pfizer Vertex Astellas Rigel Astra-Zeneca
Disclosures KINASE INHIBITORS IN RA Michael E. Weinblatt, M.D. Dr. Weinblatt has been a consultant to the following companies involved in the JAK and SYK pathways in RA Pfizer Vertex Astellas Rigel Astra-Zeneca
Pharmacy Prior Authorization
 Pharmacy Prior Authorization MERC CARE (MEDICAID) Colony Stimulating Factors (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign
Pharmacy Prior Authorization MERC CARE (MEDICAID) Colony Stimulating Factors (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign
Tocilizumab Guided Questionnaire Gastrointestinal Perforation and Related Events
 Tocilizumab Guided Questionnaire Gastrointestinal Perforation and Related Events AER: Site : Local Case ID: Patient Date of Birth (dd-mmm-yyyy): Patient ID/Initials: Patient Gender: M F Patient Weight
Tocilizumab Guided Questionnaire Gastrointestinal Perforation and Related Events AER: Site : Local Case ID: Patient Date of Birth (dd-mmm-yyyy): Patient ID/Initials: Patient Gender: M F Patient Weight
UC DavisTB Screening Requirement: How to submit your TB Health Assessment Form
 UC DavisTB Screening Requirement: How to submit your TB Health Assessment Form Step 1 If after completing the TB Risk Screening you are found to be at high risk, you must complete the Tuberculosis (TB)
UC DavisTB Screening Requirement: How to submit your TB Health Assessment Form Step 1 If after completing the TB Risk Screening you are found to be at high risk, you must complete the Tuberculosis (TB)
Wiskott-Aldrich Registry Data Collection Form Patient Identification: Patient Name (first, middle, last)
 Patient Identification: Patient Name (first, middle, last) Patient s USIDNET Registry Number assigned after online enrollment Date of Birth / / (mm/dd/yyyy) or Year of Birth Gender: male [ ], female [
Patient Identification: Patient Name (first, middle, last) Patient s USIDNET Registry Number assigned after online enrollment Date of Birth / / (mm/dd/yyyy) or Year of Birth Gender: male [ ], female [
REQUISITION FOR DIAGNOSTIC SERVICES
 2300 McPhillips Street, Winnipeg, Manitoba R2V 3M3 REQUISITION FOR DIAGNOSTIC SERVICES PATIENT NAME ADDRESS BIRTH M.H.# P.H.I.N.# TELEPHONE HOME BUSINESS MALE FEMALE POSTAL CODE HOSPITAL USE ONLY ACCOUNT
2300 McPhillips Street, Winnipeg, Manitoba R2V 3M3 REQUISITION FOR DIAGNOSTIC SERVICES PATIENT NAME ADDRESS BIRTH M.H.# P.H.I.N.# TELEPHONE HOME BUSINESS MALE FEMALE POSTAL CODE HOSPITAL USE ONLY ACCOUNT
Subject ID: I N D # # U A * Consent Date: Day Month Year
 IND Study # Eligibility Checklist Pg 1 of 15 Instructions: Check the appropriate box for each Inclusion and Exclusion Criterion below. Each criterion must be marked and all protocol criteria have to be
IND Study # Eligibility Checklist Pg 1 of 15 Instructions: Check the appropriate box for each Inclusion and Exclusion Criterion below. Each criterion must be marked and all protocol criteria have to be
Institution INSTRUCTIONS (I6) 1. This form is to be completed by a DESIGNATED STUDY NUCLEAR MEDICINE SPECIALIST
 I6 ACRIN 6660 Whole Body MRI in the Evaluation of Pediatric Malignancies Conventional Scintigraphy Imaging Form If this is a revised or corrected form, indicate by checking box and fax to 215-717 - 0936.
I6 ACRIN 6660 Whole Body MRI in the Evaluation of Pediatric Malignancies Conventional Scintigraphy Imaging Form If this is a revised or corrected form, indicate by checking box and fax to 215-717 - 0936.
PATIENT INFORMATION. First
 GUIDE DOGS OF AMERICA General Physician s Report This General Physician s Report is being requested in connection with an application for a guide dog. We require a recent physical exam and complete medical
GUIDE DOGS OF AMERICA General Physician s Report This General Physician s Report is being requested in connection with an application for a guide dog. We require a recent physical exam and complete medical
EGD Data Collection Form
 Sociodemographic Information Type Zip Code Gender Height (in inches) Race Ethnicity Inpatient Outpatient Male Female Birth Date Weight (in pounds) American Indian (Native American) or Alaska Native Asian
Sociodemographic Information Type Zip Code Gender Height (in inches) Race Ethnicity Inpatient Outpatient Male Female Birth Date Weight (in pounds) American Indian (Native American) or Alaska Native Asian
Patient Information. First Name Middle Last Preferred Name. Street Address City State Postal Code
 Ms. Patient Information First Name Middle Last Preferred Name Street Address City State Postal Code Work Phone ( ) Home Phone ( ) Cell Phone ( ) Email Preferred Contact Email Cell Home Work Emergency Contact
Ms. Patient Information First Name Middle Last Preferred Name Street Address City State Postal Code Work Phone ( ) Home Phone ( ) Cell Phone ( ) Email Preferred Contact Email Cell Home Work Emergency Contact
Case Discussion. Nutrition in IBD. Rémy Meier MD. Ulcerative colitis. Crohn s disease
 26.08.2017 Case Discussion Nutrition in IBD Crohn s disease Ulcerative colitis Rémy Meier MD Case Presentation 30 years old female, with diarrhea for 3 months Shool frequency 3-4 loose stools/day with
26.08.2017 Case Discussion Nutrition in IBD Crohn s disease Ulcerative colitis Rémy Meier MD Case Presentation 30 years old female, with diarrhea for 3 months Shool frequency 3-4 loose stools/day with
Glycogen Storage Disease
 Glycogen Storage Disease 1 Introduction The food we eat is usually used for growth, tissue repair and energy. The body stores what it does not use. Excess sugar, or glucose, is stored as glycogen in the
Glycogen Storage Disease 1 Introduction The food we eat is usually used for growth, tissue repair and energy. The body stores what it does not use. Excess sugar, or glucose, is stored as glycogen in the
34th St. and Civic Center Blvd, Philadelphia, PA 19104, phone
 34th St. and Civic Center Blvd, Philadelphia, PA 19104, phone 215-590-3630 www.chop.edu/gastroenterology Please complete this form prior to your child s visit. Please fax to (215) 590-7224 or e-mail it
34th St. and Civic Center Blvd, Philadelphia, PA 19104, phone 215-590-3630 www.chop.edu/gastroenterology Please complete this form prior to your child s visit. Please fax to (215) 590-7224 or e-mail it
Qualification Form Instructions
 Qualification Form Instructions Congratulations on taking steps toward maintaining or improving your health! The Blue Cross Blue Shield of Michigan qualification form is enclosed for you and your physician
Qualification Form Instructions Congratulations on taking steps toward maintaining or improving your health! The Blue Cross Blue Shield of Michigan qualification form is enclosed for you and your physician
E096. Essential Shared Care Agreement: Melatonin (Circadin ) in Children, adolescents and adults only with Learning Disability
 E096 Essential Shared Care Agreement: Melatonin (Circadin ) in Children, adolescents and adults only with Learning Disability Please complete the following details: Patient s name, address, date of birth,
E096 Essential Shared Care Agreement: Melatonin (Circadin ) in Children, adolescents and adults only with Learning Disability Please complete the following details: Patient s name, address, date of birth,
SUSPECT ADVERSE REACTION REPORT
 CIOMS FORM SUSPECT ADVERSE REACTION REPORT DE-BFARM-17359392 I. REACTION INFORMATION 1. PATIENT INITIALS 1a. COUNTRY 2. DATE OF BIRTH 2a. AGE 3. SEX 4-6 REACTION ONSET privacy DE [ MedDRA 20.0 LLT (10013968):
CIOMS FORM SUSPECT ADVERSE REACTION REPORT DE-BFARM-17359392 I. REACTION INFORMATION 1. PATIENT INITIALS 1a. COUNTRY 2. DATE OF BIRTH 2a. AGE 3. SEX 4-6 REACTION ONSET privacy DE [ MedDRA 20.0 LLT (10013968):
BC Cancer Protocol Summary for Therapy of Acute Myeloid Leukemia Using azacitidine and SORAfenib
 BC Cancer Protocol Summary for Therapy of Acute Myeloid Leukemia Using azacitidine and SORAfenib Protocol Code Tumour Group Contact Physician ULKAMLAS Leukemia/BMT Dr. Donna Hogge ELIGIBILITY: Acute myeloid
BC Cancer Protocol Summary for Therapy of Acute Myeloid Leukemia Using azacitidine and SORAfenib Protocol Code Tumour Group Contact Physician ULKAMLAS Leukemia/BMT Dr. Donna Hogge ELIGIBILITY: Acute myeloid
The Clinical Information Data Entry Screen is the main screen in the DQCMS application.
 DATA ENTRY Clinical Information The Clinical Information Data Entry Screen is the main screen in the DQCMS application. To enter data, a patient must first be selected from the Patient pull-down list.
DATA ENTRY Clinical Information The Clinical Information Data Entry Screen is the main screen in the DQCMS application. To enter data, a patient must first be selected from the Patient pull-down list.
PLACE LABEL HERE. Radiation Therapy Oncology Group Phase II Nasopharyngeal Cancer Follow-Up Form
 F1 AMENDED DATA Radiation Therapy Oncology Group Phase II Nasopharyngeal Cancer Follow-Up Form YES No INSTRUCTIONS: Submit this form at the appropriate follow-up interval and at death Dates are recorded
F1 AMENDED DATA Radiation Therapy Oncology Group Phase II Nasopharyngeal Cancer Follow-Up Form YES No INSTRUCTIONS: Submit this form at the appropriate follow-up interval and at death Dates are recorded
IMPACT #: Local Inventory #: form 04. Age at admission: d. mo yr. Postal code:
 - Date of birth: birth: Date of admission: year month day year month day Age at admission: d mo yr Postal code: Ethnic code: Hospital: Gender: 1 = male 2 = female 1 = Impact 2 = Other local, specify: Code
- Date of birth: birth: Date of admission: year month day year month day Age at admission: d mo yr Postal code: Ethnic code: Hospital: Gender: 1 = male 2 = female 1 = Impact 2 = Other local, specify: Code
Northwestern Polytechnic University
 Clinical Tuberculosis Assessment by Health Care Provider Clinicians should review and verify the information in the Tuberculosis (TB) Screening Questionnaire (attached). Persons answering YES to any questions
Clinical Tuberculosis Assessment by Health Care Provider Clinicians should review and verify the information in the Tuberculosis (TB) Screening Questionnaire (attached). Persons answering YES to any questions
2. Does the patient have a diagnosis of chronic idiopathic thrombocytopenic purpura (ITP)?
 Pharmacy Prior Authorization MERC CARE (MEDICAID) Promacta (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign and date. Fax
Pharmacy Prior Authorization MERC CARE (MEDICAID) Promacta (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign and date. Fax
Invasive Bacterial Disease
 Invasive Bacterial Disease Neisseria meningitidis, Haemophilus influenzae, and Group B Streptococcus (Streptococcus agalactiae) Case Report Form Electronic Disease Surveillance System Division of Surveillance
Invasive Bacterial Disease Neisseria meningitidis, Haemophilus influenzae, and Group B Streptococcus (Streptococcus agalactiae) Case Report Form Electronic Disease Surveillance System Division of Surveillance
The Bone Wellness Centre - Specialists in Dexa Total Body 855 Broadview Avenue Suite # 305 Toronto, Ontario M4K 3Z1
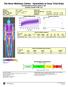 Patient: Obese, Sample Birth Date: 0/Jan/966 44.4 years Height / Weight: 72.0 cm 95.0 kg Sex / Ethnic: Male Patient ID: Referring Physician: DR. SMITH Measured: 07/Jun/200 7:0:52 PM (.40) Analyzed: 02/Apr/203
Patient: Obese, Sample Birth Date: 0/Jan/966 44.4 years Height / Weight: 72.0 cm 95.0 kg Sex / Ethnic: Male Patient ID: Referring Physician: DR. SMITH Measured: 07/Jun/200 7:0:52 PM (.40) Analyzed: 02/Apr/203
Removal of gastrostomy tube icd 10
 Removal of gastrostomy tube icd 10 The Borg System is 100 % Removal of gastrostomy tube icd 10 14-10-2008 Would I use code 43247 for the removal of PEG tube with dx V55.1? I am fairly new to gastro and
Removal of gastrostomy tube icd 10 The Borg System is 100 % Removal of gastrostomy tube icd 10 14-10-2008 Would I use code 43247 for the removal of PEG tube with dx V55.1? I am fairly new to gastro and
HEALTH SCREENING QUESTIONNAIRE CT Work-up. Donor ID EdgeCell #:
 HEALTH SCREENING QUESTIONNAIRE CT Work-up Héma-Québec Registre des Donneurs de Cellules Souches 4045 Côte-vertu, St-Laurent, QC, Canada, H4R 2W7 Tél : + 514-832-1031 Fax : + 514-832-0266 www.hema-quebec.qc.ca
HEALTH SCREENING QUESTIONNAIRE CT Work-up Héma-Québec Registre des Donneurs de Cellules Souches 4045 Côte-vertu, St-Laurent, QC, Canada, H4R 2W7 Tél : + 514-832-1031 Fax : + 514-832-0266 www.hema-quebec.qc.ca
What to do and not do before seeking surgical consultation for a patient with suspected pancreatic cancer
 What to do and not do before seeking surgical consultation for a patient with suspected pancreatic cancer 9 Th Annual Symposium on Gastrointestinal Cancers, St. Louis University School of Medicine Carlos
What to do and not do before seeking surgical consultation for a patient with suspected pancreatic cancer 9 Th Annual Symposium on Gastrointestinal Cancers, St. Louis University School of Medicine Carlos
APPLICATION FORM PILOT STUDY OF OMEGA-3 AND VITAMIN D IN T1D
 Page 1 APPLICATION FORM PILOT STUDY OF OMEGA-3 AND VITAMIN D IN T1D INSTRUCTIONS: This application and the information you provide will be used to determine if you qualify to participate in a study of
Page 1 APPLICATION FORM PILOT STUDY OF OMEGA-3 AND VITAMIN D IN T1D INSTRUCTIONS: This application and the information you provide will be used to determine if you qualify to participate in a study of
Myelodysplasia/Myeloproliferative Neoplasms (MDS/MPN) Post-HCT Data
 Instructions for Myelodysplasia/Myeloproliferative Neoplasms (MDS/MPN) Post-HCT Data (Form 2114) This section of the CIBMTR Forms Instruction Manual is intended to be a resource for completing the Myelodysplasia/Myeloproliferative
Instructions for Myelodysplasia/Myeloproliferative Neoplasms (MDS/MPN) Post-HCT Data (Form 2114) This section of the CIBMTR Forms Instruction Manual is intended to be a resource for completing the Myelodysplasia/Myeloproliferative
Drug Prescription and Administration Record
 Chart No. this admission Drug Prescription and Administration Record Patient s Name: of Admission: Chart Start : Chart Finish : NHS No: Consultant Code: Ward: ALERT: of Birth: ADDITIONAL CAUTIONS FOR PRESCRIBING
Chart No. this admission Drug Prescription and Administration Record Patient s Name: of Admission: Chart Start : Chart Finish : NHS No: Consultant Code: Ward: ALERT: of Birth: ADDITIONAL CAUTIONS FOR PRESCRIBING
IN-CENTER HEMODIALYSIS (HD) CLINICAL PERFORMANCE MEASURES DATA COLLECTION FORM 2006
 IN-CENTER HEMODIALYSIS (HD) CLINICAL PERFORMANCE MEASURES DATA COLLECTION FORM 2006 PATIENT IDENTIFICATION [Before completing please read instructions at the bottom of this page and on pages 5 and 6] MAKE
IN-CENTER HEMODIALYSIS (HD) CLINICAL PERFORMANCE MEASURES DATA COLLECTION FORM 2006 PATIENT IDENTIFICATION [Before completing please read instructions at the bottom of this page and on pages 5 and 6] MAKE
Etiology based definitions for adult malnutrition: Role of inflammation A systematic approach to nutrition assessment
 Etiology based definitions for adult malnutrition: Role of inflammation A systematic approach to nutrition assessment Gordon L Jensen, MD, PhD Penn State University University Park, PA Objectives Review
Etiology based definitions for adult malnutrition: Role of inflammation A systematic approach to nutrition assessment Gordon L Jensen, MD, PhD Penn State University University Park, PA Objectives Review
WT1 peptide cancer vaccine for solid tumors Phase I/II Clinical Trials Registration Form
 WT1 peptide cancer vaccine for solid tumors Phase I/II Clinical Trials Registration Form Hospital Department Attending doctor Country Address Phone number FAX E mail address Patient Information Name of
WT1 peptide cancer vaccine for solid tumors Phase I/II Clinical Trials Registration Form Hospital Department Attending doctor Country Address Phone number FAX E mail address Patient Information Name of
Micafungin, a new Echinocandin: Pediatric Development
 Micafungin, a new Echinocandin: Pediatric Development Andreas H. Groll, M.D. Infectious Disease Research Program Center for Bone Marrow Transplantation and Department of Pediatric Hematology/Oncology University
Micafungin, a new Echinocandin: Pediatric Development Andreas H. Groll, M.D. Infectious Disease Research Program Center for Bone Marrow Transplantation and Department of Pediatric Hematology/Oncology University
Approaching Neutropenia in Children. SW Florida Osteopathic Medical Society: 39 th Annual Seminars in Family Practice
 Approaching Neutropenia in Children SW Florida Osteopathic Medical Society: 39 th Annual Seminars in Family Practice Approaching Neutropenia in Children Emad Salman M.D Golisano Children s Hospital of
Approaching Neutropenia in Children SW Florida Osteopathic Medical Society: 39 th Annual Seminars in Family Practice Approaching Neutropenia in Children Emad Salman M.D Golisano Children s Hospital of
Pharmacy Prior Authorization
 Pharmacy Prior Authorization AETA BETTER HEALTH PESLVAIA & AETA BETTER HEALTH KIDS Promacta (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review
Pharmacy Prior Authorization AETA BETTER HEALTH PESLVAIA & AETA BETTER HEALTH KIDS Promacta (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review
Synopsis. Study Phase and Title: Study Objectives: Overall Study Design
 Synopsis Study Phase and Title: Study Objectives: Overall Study Design Phase III randomized sequential open-label study to evaluate the efficacy and safety of sorafenib followed by pazopanib versus pazopanib
Synopsis Study Phase and Title: Study Objectives: Overall Study Design Phase III randomized sequential open-label study to evaluate the efficacy and safety of sorafenib followed by pazopanib versus pazopanib
Fever. National Pediatric Nighttime Curriculum Written by Debbie Sakai, M.D. Institution: Lucile Packard Children s Hospital
 Fever National Pediatric Nighttime Curriculum Written by Debbie Sakai, M.D. Institution: Lucile Packard Children s Hospital Case 1 4-month-old well-appearing girl admitted for croup and respiratory distress.
Fever National Pediatric Nighttime Curriculum Written by Debbie Sakai, M.D. Institution: Lucile Packard Children s Hospital Case 1 4-month-old well-appearing girl admitted for croup and respiratory distress.
Case Workshop of Society for Hematopathology and European Association for Haematopathology
 Case 148 2007 Workshop of Society for Hematopathology and European Association for Haematopathology Robert P Hasserjian Department of Pathology Massachusetts General Hospital Boston, MA Clinical history
Case 148 2007 Workshop of Society for Hematopathology and European Association for Haematopathology Robert P Hasserjian Department of Pathology Massachusetts General Hospital Boston, MA Clinical history
Blood and Marrow Transplant (BMT) for Sickle Cell Disease
 Blood and Marrow Transplant (BMT) for Sickle Cell Disease Rhiannon is now cured of sickle cell disease after BMT. Blood and marrow transplant (BMT) is a proven cure for sickle cell disease. This handbook
Blood and Marrow Transplant (BMT) for Sickle Cell Disease Rhiannon is now cured of sickle cell disease after BMT. Blood and marrow transplant (BMT) is a proven cure for sickle cell disease. This handbook
A new model for prescribing varenicline
 Pharmacist Independent Prescribers in partnership with A new model for prescribing varenicline Dear Stop Smoking Advisor You will be aware of the stop smoking drug varenicline that goes under the brand
Pharmacist Independent Prescribers in partnership with A new model for prescribing varenicline Dear Stop Smoking Advisor You will be aware of the stop smoking drug varenicline that goes under the brand
PERITONEAL DIALYSIS CLINICAL PERFORMANCE MEASURES DATA COLLECTION FORM 2006
 PERITONEAL DIALYSIS CLINICAL PERFORMANCE MEASURES DATA COLLECTION FORM 2006 PATIENT IDENTIFICATION [Before completing please read instructions at the bottom of this page and on pages 5 and 6] MAKE CORRECTIONS
PERITONEAL DIALYSIS CLINICAL PERFORMANCE MEASURES DATA COLLECTION FORM 2006 PATIENT IDENTIFICATION [Before completing please read instructions at the bottom of this page and on pages 5 and 6] MAKE CORRECTIONS
International IBD Genetics Consortium
 International IBD Genetics Consortium PRED4 Proton Pump Inhibitor Induced Nephritis Case Report FormS Please stick study label here On completion, please return to: IBD Pharmacogenetics Research Office
International IBD Genetics Consortium PRED4 Proton Pump Inhibitor Induced Nephritis Case Report FormS Please stick study label here On completion, please return to: IBD Pharmacogenetics Research Office
Mount Mystics MSVU Athletics & Recreation
 Mount Mystics 2015-2016 MSVU Athletics & Recreation Student Athlete Medical History Card Please complete the first 3 pages and bring to entire document to the doctor s office. Athlete Information Sport:
Mount Mystics 2015-2016 MSVU Athletics & Recreation Student Athlete Medical History Card Please complete the first 3 pages and bring to entire document to the doctor s office. Athlete Information Sport:
Patient enrolment details CKDOD registry
 Patient enrolment details CKDOD registry Personal Information Date of enrolment D D Date of birth D D Gender Descent Height cm m History of smoking Y N U Date of first diagnosis of CKD D D Primary Cause
Patient enrolment details CKDOD registry Personal Information Date of enrolment D D Date of birth D D Gender Descent Height cm m History of smoking Y N U Date of first diagnosis of CKD D D Primary Cause
Influenza-Associated Pediatric Deaths Case Report Form
 STATE USE ONLY DO NOT SEND INFORMATION IN THIS SECTION TO CDC Form approved OMB No. 0920-0007 Last Name: First Name: County: Address: City: State, Zip: Patient Demographics 1. State: 2. County: 3. State
STATE USE ONLY DO NOT SEND INFORMATION IN THIS SECTION TO CDC Form approved OMB No. 0920-0007 Last Name: First Name: County: Address: City: State, Zip: Patient Demographics 1. State: 2. County: 3. State
PARTICIPANT DIARY TREATMENT ALLOCATION: LACTIC ACID GEL
 PARTICIPANT DIARY TREATMENT ALLOCATION: LACTIC ACID GEL For site staff completion only: Participating Site: Participant Initials: Participant ID: Date of Randomisation: Allocated Treatment: Lactic Acid
PARTICIPANT DIARY TREATMENT ALLOCATION: LACTIC ACID GEL For site staff completion only: Participating Site: Participant Initials: Participant ID: Date of Randomisation: Allocated Treatment: Lactic Acid
New Patient Information Form
 New Patient Information Form Patient Identification Prenatal Alcohol & Drug Exposure Clinic FASD CLINIC Patient s OHIP N. Female Male Race Patient s Name Birth Date Age First Middle Last Patient s Address
New Patient Information Form Patient Identification Prenatal Alcohol & Drug Exposure Clinic FASD CLINIC Patient s OHIP N. Female Male Race Patient s Name Birth Date Age First Middle Last Patient s Address
POST-OP CARDIAC SURGERY PHYSICIAN S ORDER SHEET USE BALLPOINT PEN ONLY. CARDIAC INTENSIVE CARE UNIT
 PHYSICIAN S SHEET Automatically Activate, if not in agreement, cross out and initial Activated by Checking Box ALLERGIES: None known YES Patient s Height: Patient s Weight: ALL MEDICATION and INTRAVENOUS
PHYSICIAN S SHEET Automatically Activate, if not in agreement, cross out and initial Activated by Checking Box ALLERGIES: None known YES Patient s Height: Patient s Weight: ALL MEDICATION and INTRAVENOUS
ADPedKD: detailed description of data which will be collected in this registry
 ADPedKD: detailed description of data which will be collected in this registry I. Basic data 1. Patient ID: will be given automatically 2. Personal information - Date of informed consent: DD/MM/YYYY -
ADPedKD: detailed description of data which will be collected in this registry I. Basic data 1. Patient ID: will be given automatically 2. Personal information - Date of informed consent: DD/MM/YYYY -
International IBD Genetics Consortium
 International IBD Genetics Consortium PRED4 Thiopurine Induced Pancreatitis Case Report Form Please stick study label here On completion, please return to: Claire Bewshea IBD Pharmacogenetics Research
International IBD Genetics Consortium PRED4 Thiopurine Induced Pancreatitis Case Report Form Please stick study label here On completion, please return to: Claire Bewshea IBD Pharmacogenetics Research
Company/Group Name: Business Telephone: Fax: Option 2:
 Application Form Please read through the following before completing this application form in BLOCK CAPITALS. You must disclose all material facts. Failure to do so may invalidate the Cover. A material
Application Form Please read through the following before completing this application form in BLOCK CAPITALS. You must disclose all material facts. Failure to do so may invalidate the Cover. A material
What s Inside. Influenza and Pneumococcal Pneumonia
 Aug. 21, 2006 06-064 Special Flu Bulletin - Texas What s Inside Influenza Virus Vaccine Claims Filing Requirements...2 Electronic Billing...2 CMS-1500 Claim Form Sample...5 Completion of the Influenza
Aug. 21, 2006 06-064 Special Flu Bulletin - Texas What s Inside Influenza Virus Vaccine Claims Filing Requirements...2 Electronic Billing...2 CMS-1500 Claim Form Sample...5 Completion of the Influenza
Mountain Area Health Education Center 121 Hendersonville Road. Asheville, NC PHONE: (828) FAX: (828)
 Coronal Polishing Clinical Participation Consent Form Coronal Polishing for the DA II MAHEC I,, participant in the Coronal Polishing for the DA II Program to be held on November 3, 2018, do hereby consent
Coronal Polishing Clinical Participation Consent Form Coronal Polishing for the DA II MAHEC I,, participant in the Coronal Polishing for the DA II Program to be held on November 3, 2018, do hereby consent
Sarcoidosis Case. Robert P. Baughman Interstitial Lung Disease and Sarcoidosis Clinic University of Cincinnati, USA. WASOG: educational material
 Sarcoidosis Case Robert P. Baughman Interstitial Lung Disease and Sarcoidosis Clinic University of Cincinnati, USA WASOG: educational material Sarcoidosis Case patient is a Caucasian male age 46 was diagnosed
Sarcoidosis Case Robert P. Baughman Interstitial Lung Disease and Sarcoidosis Clinic University of Cincinnati, USA WASOG: educational material Sarcoidosis Case patient is a Caucasian male age 46 was diagnosed
Lymphoma Case Scenario 1
 Lymphoma Case Scenario 1 HISTORY: A 23-year-old healthy female presented with a month-long history of persistent headache of increasing severity. She noted episodic nausea and vomiting in association with
Lymphoma Case Scenario 1 HISTORY: A 23-year-old healthy female presented with a month-long history of persistent headache of increasing severity. She noted episodic nausea and vomiting in association with
Interferon Side Effects and The Future of Interferon Sparing Regimens. Todd Wills, MD ETAC Infectious Disease Specialist
 Interferon Side Effects and The Future of Interferon Sparing Regimens Todd Wills, MD ETAC Infectious Disease Specialist HEPATITIS C TREATMENT EXPANSION INITIATIVE MULTISITE CONFERENCE CALL FEBRUARY 15,
Interferon Side Effects and The Future of Interferon Sparing Regimens Todd Wills, MD ETAC Infectious Disease Specialist HEPATITIS C TREATMENT EXPANSION INITIATIVE MULTISITE CONFERENCE CALL FEBRUARY 15,
for healthcare professionals
 Fertility toolkit for healthcare professionals We ve developed this toolkit for setting up a fertility referral pathway for young women with breast cancer at the point of diagnosis. We want to help improve
Fertility toolkit for healthcare professionals We ve developed this toolkit for setting up a fertility referral pathway for young women with breast cancer at the point of diagnosis. We want to help improve
AXA MANSARD PERSONAL GOLD PLAN Cover & Exclusions
 This plan covers the following services; AXA MANSARD PERSONAL GOLD PLAN Cover & Exclusions EMERGENCY CARE: Initial stabilization and care, it also covers intensive care if required INTENSIVE CARE (covered
This plan covers the following services; AXA MANSARD PERSONAL GOLD PLAN Cover & Exclusions EMERGENCY CARE: Initial stabilization and care, it also covers intensive care if required INTENSIVE CARE (covered
Acute Stroke with Alteplase Administration Order Set
 Review Due Date: 2017 October PATIENT CARE DERS Weight: Adverse Reactions or Intolerances Drug No Yes (list) Food No Yes (list) _ Latex No Yes Admission Admit to Neurology service: Dr. Critical Care Diagnosis:
Review Due Date: 2017 October PATIENT CARE DERS Weight: Adverse Reactions or Intolerances Drug No Yes (list) Food No Yes (list) _ Latex No Yes Admission Admit to Neurology service: Dr. Critical Care Diagnosis:
SUPPLEMENTAL APPENDIX
 SUPPLEMENTAL APPENDIX Table of Contents 1. Supplemental Tables 2. Supplemental Figures Nature Medicine: doi:1.138/nm.4441 Supplemental Table 1. Baseline Characteristics of Patients Treated with anti-cd22
SUPPLEMENTAL APPENDIX Table of Contents 1. Supplemental Tables 2. Supplemental Figures Nature Medicine: doi:1.138/nm.4441 Supplemental Table 1. Baseline Characteristics of Patients Treated with anti-cd22
Drug Prior Authorization Form Opdivo (nivolumab)
 This document contains both information and form fields. To read information, use the Down Arrow from a form field. Drug Prior Authorization Form The purpose of this form is to obtain information required
This document contains both information and form fields. To read information, use the Down Arrow from a form field. Drug Prior Authorization Form The purpose of this form is to obtain information required
Objectives. Types of HIV Tests. Age Appropriateness of Tests. Breastfeeding and HIV Testing. Why are there different tests for different ages?
 Objectives At the end of the lesson participants will be able to: Identify the types of HIV tests available in Botswana State who should be tested Identify which tests are used for infants and children
Objectives At the end of the lesson participants will be able to: Identify the types of HIV tests available in Botswana State who should be tested Identify which tests are used for infants and children
Personal Information Protection Act Consent Form
 Personal Information Protection Act Consent Form Lloydminster Denture Clinic Inc. In our office, we are dedicated to ensuring the protection of our patients personal information and insuring that this
Personal Information Protection Act Consent Form Lloydminster Denture Clinic Inc. In our office, we are dedicated to ensuring the protection of our patients personal information and insuring that this
CIBMTR Center Number: CIBMTR Recipient ID: Today s Date: Date of HSCT for which this form is being completed:
 Chronic Myelogenous Leukemia (CML) Post-HSCT Data Sequence Number: Date Received: Registry Use Only Today s Date: Date of HSCT for which this form is being completed: HSCT type: autologous allogeneic,
Chronic Myelogenous Leukemia (CML) Post-HSCT Data Sequence Number: Date Received: Registry Use Only Today s Date: Date of HSCT for which this form is being completed: HSCT type: autologous allogeneic,
Influenza-Associated Pediatric Mortality Case Report Form Form Approved OMB No
 Influenza-Associated Pediatric Mortality Case Report Form Form Approved OMB No. 0920-0004 STATE USE ONLY DO NOT SEND INFORMATION IN THIS SECTION TO CDC Last Name: First Name: County: Address: City: State,
Influenza-Associated Pediatric Mortality Case Report Form Form Approved OMB No. 0920-0004 STATE USE ONLY DO NOT SEND INFORMATION IN THIS SECTION TO CDC Last Name: First Name: County: Address: City: State,
Ebola Virus Disease Interim Public health follow-up form
 Ebola Virus Disease Interim Public health follow-up For asymptomatic returning travellers from an affected country/area or close contacts of an Ebola virus disease case October 22, 2014 (1) Case Ination
Ebola Virus Disease Interim Public health follow-up For asymptomatic returning travellers from an affected country/area or close contacts of an Ebola virus disease case October 22, 2014 (1) Case Ination
MEDICAL OFFICE PROCEDURES (250)
 Page 1 of 7 MEDICAL OFFICE PROCEDURES (250) REGIONAL 2016 Part I Multiple Choice (20 @ 5 points each) Part II Matching (5 @ 3 points each) Part III Abbreviations (15 @ 4 points each) Part IV Telephone
Page 1 of 7 MEDICAL OFFICE PROCEDURES (250) REGIONAL 2016 Part I Multiple Choice (20 @ 5 points each) Part II Matching (5 @ 3 points each) Part III Abbreviations (15 @ 4 points each) Part IV Telephone
Qualification Form Instructions
 Qualification Form Instructions Congratulations on taking steps toward maintaining or improving your health! The Blue Cross Blue Shield of Michigan Qualification Form is enclosed for you and your physician
Qualification Form Instructions Congratulations on taking steps toward maintaining or improving your health! The Blue Cross Blue Shield of Michigan Qualification Form is enclosed for you and your physician
Essential Shared Care Agreement: Melatonin (Circadin, Bio-Melatonin)
 Ref No: E056 Essential Shared Care Agreement: Melatonin (Circadin, Bio-Melatonin) Please complete the following details: Patient s name, address, date of birth, and either complete when the consultant
Ref No: E056 Essential Shared Care Agreement: Melatonin (Circadin, Bio-Melatonin) Please complete the following details: Patient s name, address, date of birth, and either complete when the consultant
New Zealand Bone Marrow Donor Registry
 Title: SECTION 4.0 NZBMDR STANDARDS DONOR RECRUITMENT, ENROLMENT & INFORMATION Authorised by: Executive Officer Contributing Authors: ABMDR Dr Hilary Blacklock Raewyn Fisher NZBMDR-Guidelines-004-13 Date
Title: SECTION 4.0 NZBMDR STANDARDS DONOR RECRUITMENT, ENROLMENT & INFORMATION Authorised by: Executive Officer Contributing Authors: ABMDR Dr Hilary Blacklock Raewyn Fisher NZBMDR-Guidelines-004-13 Date
Welcome to the Rubin Institute for Advanced Orthopedics!
 Welcome to the Rubin Institute for Advanced Orthopedics! Dear New Patient, Welcome to the Rubin Institute for Advanced Orthopedics! Our goal is to provide you with caring, compassionate and professional
Welcome to the Rubin Institute for Advanced Orthopedics! Dear New Patient, Welcome to the Rubin Institute for Advanced Orthopedics! Our goal is to provide you with caring, compassionate and professional
Hepatitis C Medications Hawaii PRIOR AUTHORIZATION REQUEST FORM
 Please complete this entire form and fax it to: 866-940-7328. If you have questions, please call 800-310-6826. This form contains multiple pages. Please complete all pages to avoid a delay in our decision.
Please complete this entire form and fax it to: 866-940-7328. If you have questions, please call 800-310-6826. This form contains multiple pages. Please complete all pages to avoid a delay in our decision.
Granix. Granix (tbo-filgrastim) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.85.16 Subject: Granix 1 of 7 Last Review Date: December 2, 2016 Granix Description Granix (tbo-filgrastim)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.85.16 Subject: Granix 1 of 7 Last Review Date: December 2, 2016 Granix Description Granix (tbo-filgrastim)
Stem cell transplantation. Dr Mohammed Karodia NHLS & UP
 Stem cell transplantation Dr Mohammed Karodia NHLS & UP The use of haemopoeitic stem cells from a donor harvested from peripheral blood or bone marrow, to repopulate recipient bone marrow. Allogeneic From
Stem cell transplantation Dr Mohammed Karodia NHLS & UP The use of haemopoeitic stem cells from a donor harvested from peripheral blood or bone marrow, to repopulate recipient bone marrow. Allogeneic From
Granix. Granix (tbo-filgrastim) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.10.16 Section: Prescription Drugs Effective Date: April 1, 2014 Subject: Granix 1 of 7 Last Review Date:
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.10.16 Section: Prescription Drugs Effective Date: April 1, 2014 Subject: Granix 1 of 7 Last Review Date:
ROTUNDA HOSPITAL DEPARTMENT OF LABORATORY MEDICINE
 This active test table informs the user of Biochemistry tests available in house. s referred to other sites are recorded in the Referred Table. Issue date: 4 TH April 2016 Contact Phone Number ext.1345/2522
This active test table informs the user of Biochemistry tests available in house. s referred to other sites are recorded in the Referred Table. Issue date: 4 TH April 2016 Contact Phone Number ext.1345/2522
Drug Niraparib Olaparib
 Dear NCCN Value Pathway Committee, We are making this submission to provide information that we believe is relevant for developing NCCN Categories of Preference for the use of PARP inhibitors in recurrent
Dear NCCN Value Pathway Committee, We are making this submission to provide information that we believe is relevant for developing NCCN Categories of Preference for the use of PARP inhibitors in recurrent
68 Ga-DOTAXXX Template Case Report Forms (CRFs) Table of Contents
 68 Ga-DOTAXXX Template Case Report Forms (CRFs) Table of Contents I. Screening Visit II. Imaging Baseline Visit III. Adverse Event (Baseline) IV. Image Interpretation Initial V. 24-hour Follow-Up Call
68 Ga-DOTAXXX Template Case Report Forms (CRFs) Table of Contents I. Screening Visit II. Imaging Baseline Visit III. Adverse Event (Baseline) IV. Image Interpretation Initial V. 24-hour Follow-Up Call
Exercise Referral Form
 Exercise Referral Form To be completed by the referring Health Professional All patient data will be kept securely and in accordance with Data Protection guidelines Patient Details: Title: Mr/Mrs/Ms/Miss/Other:
Exercise Referral Form To be completed by the referring Health Professional All patient data will be kept securely and in accordance with Data Protection guidelines Patient Details: Title: Mr/Mrs/Ms/Miss/Other:
Granix. Granix (tbo-filgrastim) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.10.16 Subject: Granix 1 of 7 Last Review Date: September 18, 2015 Granix Description Granix (tbo-filgrastim)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.10.16 Subject: Granix 1 of 7 Last Review Date: September 18, 2015 Granix Description Granix (tbo-filgrastim)
SAMPLE. Survivorship Care Plan for Lymphoma (Diffuse Large B-Cell) General Information. Care team
 SAMPLE This Survivorship Care Plan will facilitate cancer care following active treatment. It may include important contact information, a treatment summary, recommendations for follow-up care testing,
SAMPLE This Survivorship Care Plan will facilitate cancer care following active treatment. It may include important contact information, a treatment summary, recommendations for follow-up care testing,
Serious Adverse Event (SAE) Form Clinical Trials
 Protocol Code number: LP0162-1325 1. Trial information: Subject ID: Site No.: Country: If blinded trial, was the study treatment unblinded? Yes No Is this the initial report of an SAE or a follow-up? Initial
Protocol Code number: LP0162-1325 1. Trial information: Subject ID: Site No.: Country: If blinded trial, was the study treatment unblinded? Yes No Is this the initial report of an SAE or a follow-up? Initial
ISTITUTO DI RICERCHE FARMACOLOGICHE MARIO NEGRI CLINICAL RESEARCH CENTER ALDO E FOR CELE RARE DACCO DISEASES ALDO E CELE DACCO
 ISTITUTO DI RICERCHE FARMACOLOGICHE MARIO NEGRI CENTRO MARIO DI NEGRI RICERCHE INSTITUTE CLINICHE FOR PHARMACOLOGICAL PER LE MALATTIE RESEARCH RARE CLINICAL RESEARCH CENTER ALDO E FOR CELE RARE DACCO DISEASES
ISTITUTO DI RICERCHE FARMACOLOGICHE MARIO NEGRI CENTRO MARIO DI NEGRI RICERCHE INSTITUTE CLINICHE FOR PHARMACOLOGICAL PER LE MALATTIE RESEARCH RARE CLINICAL RESEARCH CENTER ALDO E FOR CELE RARE DACCO DISEASES
HEALTH SCREENING QUESTIONNAIRE. Work-up. Donor ID EdgeCell #:
 HEALTH SCREENING QUESTIONNAIRE Héma-Québec Registre des Donneurs de Cellules Souches 4045 Côte-vertu, St-Laurent, QC, Canada, H4R 2W7 Tél : + 514-832-1031 Fax : + 514-832-0266 www.hema-quebec.qc.ca CT
HEALTH SCREENING QUESTIONNAIRE Héma-Québec Registre des Donneurs de Cellules Souches 4045 Côte-vertu, St-Laurent, QC, Canada, H4R 2W7 Tél : + 514-832-1031 Fax : + 514-832-0266 www.hema-quebec.qc.ca CT
Circle Yes or Y N. [Note: requests without this information will not be accepted.] [If no, then no further questions.
![Circle Yes or Y N. [Note: requests without this information will not be accepted.] [If no, then no further questions. Circle Yes or Y N. [Note: requests without this information will not be accepted.] [If no, then no further questions.](/thumbs/96/126589073.jpg) 10/01/2016 Prior Authorization Aetna Better Health of West Virginia COLO STIMULATIG FACTORS (WV88) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information,
10/01/2016 Prior Authorization Aetna Better Health of West Virginia COLO STIMULATIG FACTORS (WV88) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information,
CHECK LIST FORM-MONTH 27 (Please note Month 27 is from enrolment not randomisation)
 CHECK LIST FORM-MONTH 27 (Please note Month 27 is from enrolment not randomisation) Participant Initials: Date of Birth: Were the following forms completed for this visit? Follow Up Form Done t Done BVASWG
CHECK LIST FORM-MONTH 27 (Please note Month 27 is from enrolment not randomisation) Participant Initials: Date of Birth: Were the following forms completed for this visit? Follow Up Form Done t Done BVASWG
Patient Registration Please fill out and bring to your first visit. (Please Print) PATIENT INFORMATION. P.O. Box: City: State: ZIP Code:
 Nutrition Works LLC 805 Stevens Avenue Portland, Maine 04103 (207) 772-6279 Fax (207) 347-4281 Susan Quimby, R.D., L.D. Judy Donnelly, R.D., L.D. Kim Norbert, M.S., R.D., L.D. Patsy Catsos, M.S., R.D.,
Nutrition Works LLC 805 Stevens Avenue Portland, Maine 04103 (207) 772-6279 Fax (207) 347-4281 Susan Quimby, R.D., L.D. Judy Donnelly, R.D., L.D. Kim Norbert, M.S., R.D., L.D. Patsy Catsos, M.S., R.D.,
LCD for Sargramostim (GM-CSF, Leukine ) (L29275)
 LCD for Sargramostim (GM-CSF, Leukine ) (L29275) Contractor Information Contractor Name First Coast Service Options, Inc. Contractor Number 09102 Contractor Type MAC - Part B LCD ID Number L29275 LCD Information
LCD for Sargramostim (GM-CSF, Leukine ) (L29275) Contractor Information Contractor Name First Coast Service Options, Inc. Contractor Number 09102 Contractor Type MAC - Part B LCD ID Number L29275 LCD Information
Granix. Granix (tbo-filgrastim) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.85.16 Subject: Granix 1 of 6 Last Review Date: September 15, 2017 Granix Description Granix (tbo-filgrastim)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.85.16 Subject: Granix 1 of 6 Last Review Date: September 15, 2017 Granix Description Granix (tbo-filgrastim)
Date of Birth. Black/African American. What is your occupation? Retired? Yes No
 Health Risk Assessment Today s Date: Name Date of Birth GENERAL INFORMATION What is your race? American Indian or Alaskan Native Native Hawaiian or Other Pacific Islander Asian, Chinese, Japanese, Korean
Health Risk Assessment Today s Date: Name Date of Birth GENERAL INFORMATION What is your race? American Indian or Alaskan Native Native Hawaiian or Other Pacific Islander Asian, Chinese, Japanese, Korean
