SENSITIVITY AND SPECIFICITY OF AN IN-HOUSE RAPID UREASE TEST FOR DETECTING HELICOBACTER PYLORI INFECTION ON GASTRIC BIOPSY
|
|
|
- Lesley Carter
- 6 years ago
- Views:
Transcription
1 SOUTHEAST ASIAN J TROP MED PUBLIC HEALTH SENSITIVITY AND SPECIFICITY OF AN IN-HOUSE RAPID UREASE TEST FOR DETECTING HELICOBACTER PYLORI INFECTION ON GASTRIC BIOPSY Chariya Chomvarin 1, Yingrit Chantarasuk 1, Pisaln Mairiang 2, Churairat Kularbkaew 3, Apichat Sangchan 2, Kitti Chanlertrith 2 and Wises Namwat 1 1 Department of Microbiology, 2 Department of Medicine, 3 Department of Pathology, Faculty of Medicine, Khon Kaen University, Khon Kaen, Thailand Abstract. We developed an in-house rapid urease test (irut) and evaluated the efficacy and the agreement of the irut and the crut compared with culture and histology for the detection of H. pylori infection. Five irut media were tested with H. pylori isolates and other bacteria. The most suitable irut medium was further evaluated for detection of H. pylori infection. Gastric biopsies from 120 patients were diagnosed by culture, irut, crut and histology. The results of the irut and crut were read at 30 minutes, 1 hour and up to 24 hours. A true positive result was either the culture or both the RUT (crut or irut) and the histological examination being positive. The sensitivity and specificity of the irut result at 30 minutes, 1 hour and up to 24 hours were 77.1% and 100%, 77.6% and 100%, and 94.1% and 94.2%, respectively. Values for the same parameters of crut were 87.5% and 100%, 89.8% and 100%, and 100% and 94.2%, respectively. The agreement between the irut and crut was very good (kappa values 0.82). Our results indicate that the irut is a sensitive, specific and cost effective test. It can be appropriately applied for detecting H. pylori infection in gastric biopsy specimens. INTRODUCTION Helicobacter pylori is a gram-negative curved-to-spiral microaerobic bacteria, considered an important etiological agent in the development of gastritis, peptic ulcers and gastric carcinoma (Ansorg et al, 1991; Parsonnet, 1994; Logan and Walker, 2001). At present, there are several techniques available for the detection of H. pylori, including bacterial culture, histological examination, serological testing, a rapid urease test (RUT), a urea breath test (UBT) and polymerase chain reaction (Fabre et al, 1994; Heatley, 1995; Kisa et al, 2002). RUT is an attractive diagnostic method because it is rapid, sensitive, specific and requires only visual interpretation (Kawanishi et al, 1995; Onders, 1997; Said et al, 2004). It can give a presumptive result which expedites therapeutic decision making (Onders, 1997; Kuo et al, 2002; Lim et al, Correspondence: Chariya Chomvarin, Department of Microbiology, Faculty of Medicine, Khon Kaen University, Khon Kaen 40002, Thailand. Tel: ; Fax: chariya@kku.ac.th 2004; Morio et al, 2004). However, the unit cost for crut is relatively expensive for routine work in developing countries, such as Thailand. The development of an irut has been reported (Goldie et al, 1989; Forman et al, 1994; Pajares-Garcia, 1998; Adesanya et al, 2002), but some iruts have had disadvantages such as containing sodium azide, which is potentially toxic (Hazell et al, 1987). Some iruts have been insensitive, time consuming and have false positive results, especially when long incubation times were used to increase the sensitivity (Das et al, 1987; Ho et al, 1996). There are several types of irut including buffered and unbuffered urea media, and liquid and semisolid media (Vaira et al, 1988; Thillainayagam et al, 1991; Kuo et al, 2002; Montes et al, 2003). The results for each irut are different (Goldie et al, 1989; Forman et al, 1994; Pajares-Garcia, 1998; Adesanya et al, 2002). We developed and tested (in vitro) five irut formulae and sought one that would allow easy interpretation after a short incubation period without false positive results due to other urease positive organisms. We tested the sen- 312 Vol 37 No. 2 March 2006
2 IN-HOUSE RAPID UREASE TEST FOR H. PYLORI DETECTION sitivity and specificity of irut on gastric biopsies from dyspeptic patients by comparing with standard culture, histological examination and crut. The stability of the medium and unit cost were also determined. METHODS Patients and endoscopy One hundred twenty consecutive patients with dyspeptic symptoms who underwent upper gastrointestinal endoscopy were included in this study. They were recruited from the Endoscopy Unit of Srinagarind Hospital, Faculty of Medicine, Khon Kaen University between February 2002 and February The subjects were diagnosed as non-ulcer dyspepsia (NUD), peptic ulcer dyspepsia (PUD), gastric carcinoma (GCA) and other gastrointestinal diseases (GERD, duodenitis, etc). We excluded patients who had antibiotic therapy, bismuth treatment, proton pump inhibitors, or H2-blockers within the previous month. Informed consent was obtained from each patient before being included in the study. Biopsy specimens Four antral and four corpus biopsy specimens were obtained from each patient and divided into four parts. Both antral and corpus specimens were used for cultures, a commercial rapid urease test (crut), an in-house rapid urease test (irut) and histological examination. Culture The culture was performed according to Hazell et al (1989) with modification. Briefly, each antral and corpus specimen was immediately placed into transport media and brought to the laboratory within 2 hours, and stored under cold conditions. The biopsy specimens were homogenized in 200 µl of normal saline and cultured on 7% human blood agar (Difco, Detroit, Michigan, USA) containing the supplement SR147 (5 mg/l trimethoprim +10 mg/l vancomycin + 5 mg/ l amphotericin B + 5 mg/l cefsulodin, SR147, OXOID). The plates were incubated at 37ºC under microaerophilic conditions (5% O 2, 10% CO 2, 85% N 2 ) and were examined after 4 and 7 days of incubation. Characteristic colonies of H. pylori were confirmed by Gram staining, oxidase, catalase and urease tests. Commercial rapid urease test (RUT, Pronto Dry test) The RUT was performed according to the manufacturer s instructions (Medical Instruments Corporation, Solothurn, Switzerland). Briefly, one antral and one corpus specimen were directly inoculated onto the crut agar gel. The results were observed and recorded at 24 hours. a positive was indicated when the color changed from yellow to pink. Histological examination One antral and one corpus biopsy were fixed in 10% buffered formalin, processed, then embedded in paraffin. Four slices 3-4 µm thick were stained with modified Warthin-Starry stain for identification of H. pylori (Cohen and Laine, 1997; Li et al, 2004). The presence of spiral organisms on any of the slides was considered positive for H. pylori. In-house rapid urease test (irut) Organisms. Proteus mirabilis (a strong urease producer), Pseudomonas aeruginosa (a weak urease producer), and Escherichia coli (a urease negative organism) were grown on blood agar under atmospheric conditions for 24 hours. Four isolates of H. pylori were grown on blood agar under microaerophilic conditions for 4 days. The concentration of H.pylori were adjusted to 10 8 organisms/ml in 1 % proteose peptone water at McFarland standard no. 3 (Xia et al, 1994) and the other bacteria at McFarland standard no. 0.5 (National Committee for Clinical-Laboratory Standards, 2002). Ten fold serial dilutions were prepared ranging from 10 7 to 10 2 cells/ml. Ten microliters of each dilution was then used for the irut. Media. The following 5 media were tested: Formula I was comprised of urea 20 g/l, KH 2 PO 4 2 g/l, phenol red g/l, NaCl 5 g/l, peptone 1 g/l, glucose 10 g/l and agar 4 g/l (Modified Christensen urease test). Formula II was comprised of urea 20 g/l, NaH 2 PO 4.H 2 O 1.4 g/l, phenol red g/l and agar 4 g/l (Buffered medium). Formula III was comprised of urea 20 g/l, phenol red g/l and agar 4 g/l.(unbuffered medium). Formula IV was comprised of urea 20 g/l, NaH 2 PO 4.H 2 O 1.4 g/l, phenol red 0.04 g/l Vol 37 No. 2 March
3 SOUTHEAST ASIAN J TROP MED PUBLIC HEALTH and agar 4 g/l (Buffered medium). Formula V was comprised of urea 20 g/l, phenol red 0.04 g/l and agar 4 g/l (Unbuffered medium). After preparation, 2 ml of each medium was aseptically dispensed into a steriled 24 well plate, wrapping with aluminium foil and stored at 4 P C until used. Test for optimal medium. The five formula media were tested for the optimal irut medium used. One hundred microliters of each bacterial suspension was inoculated into each well of medium. The results were observed and recorded after 30 minutes, and at 1, 2, 4 and 24 hours. The grading criteria were interpreted according to the following criteria: 1) negative, when the medium had no color (yellow); 2) positive 1+, when the color changed from yellow to pale pink; 3) positive 2+, when the color changed from yellow to pink; 4) positive 3+, when the color changed from yellow to red and; 5) positive 4+, when the color changed from yellow to deep red or purple. Test for specimens. One antral and one corpus specimen were each directly inoculated onto an irut. The results were observed and recorded, the same as the crut. Statistical analysis Sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV), and accuracy of the irut were evaluated by comparing with culture, histological examination and the crut. While the same parameters for the crut were evaluated by comparing with culture, histological examination and the irut. The criteria for a true positive H. pylori result was considered as having a positive result Table 1 In intro IRUT testing for H. pylori, P. mirabilis, Ps. aerginosa and E. coli. Reaction a with the following organisms/ml Organisms and Reaction time H. pylori (isolate 1) 30 minutes hour hours hours H. pylori (isolate 2) 30 minutes hour hours hours H. pylori (isolate 3) 30 minutes hour hours hours H. pylori (isolate 4) 30 minutes hour hours hours P. mirabilis, E. coli, Ps. aeruginosa 24 hours a Grading of reaction: - = no color change, 1+ = pale pink, 2+ = pink, 3+ = red, 4+ = deep red to purple 314 Vol 37 No. 2 March 2006
4 IN-HOUSE RAPID UREASE TEST FOR H. PYLORI DETECTION Table 2 Numbers and percentages of 120 H. pylori infections detected by culture, histology, crut and irut. Culture Histology crut irut Total No. (%) Evaluation of infection a (32.5) TP (2.5) TP (7.5) TP (3.3) FP (20.8) FP (33.3) TN Total 120 (100) 51 (42.5%) TP = True positive, TN = True negative, FP = False positive a = TP, culture positive on both urease and histological examination positive Table 3 Sensitivity, specificity, positive and negative predictive values and accuracy of the irut compared with the gold standard c. Time Sensitivity (%) Specificity (%) PPV a (%) NPV b (%) Accuracy (%) 30 minutes 77.1 (37/48) 100 (72/72) 100 (37/37) 86.7 (72/83) 90.8 (109/120) 1 hour 77.6 (38/49) 100 (71/71) 100 (38/38) 86.6 (71/82) 90.8 (109/120) 2 hours 79.6 (39/49) 100 (71/71) 100 (39/39) 87.6 (71/81) 91.6 (110/120) 24 hours 94.1 (48/51) 94.2 (65/69) 92.3 (48/52) 95.6 (65/68) 94.1 (113/120) a Positive predictive value; b Negative predictive value; c The criteria for the gold standard were positive culture and/ or positive histology and crut. on either the culture or RUT and histological examination (Pajares-Garcia, 1998; Liao et al, 2003). The degree of agreement between the commercial and irut was also analyzed by a Kappa statistic (Landis et al, 1977). RESULTS We selected the medium that gave a positive reaction in the shortest time, at the lowest concentration of H. pylori, while giving a negative reaction with other organisms. The buffered medium formula II (urea 20 g/l, NaH 2 PO g/l, phenol red g/l and agar 4 g/l) was the optimal medium, since the positive results were observed within 30 minutes when tested with 4 H. pylori isolates at organisms/ml or cells when loaded in 10 µl (Table 1). Moreover, it was easy to differentiate between a weakly positive or a weakly negative result compared with the other formulae. The medium was further applied to the gastric biopsies from 120 dyspeptic patients. H. pylori was detected by culture, histological examination, crut and irut in 42 (35%), 76 (63.3%), 55 (66%) and 52 (43.3%), respectively. Regarding the true positive test criteria, H. pylori infection was found in 42.5% (Table 2). The sensitivity and specificity for the irut were 77.1% and 100%, 77.6 and 100%, 79.6% and 100%, and 94.1% and 94.2 % when tested at 30 minutes, 1 hour, 2 hours and 24 hours, respectively (Table 3). Values of the same parameters of crut have been shown in Table 4. A false positive reaction was not found for either the irut or the crut at 30 minutes or 1 hour but was found in 4 of 120 specimens (3.33 %) at 24 hours. Four of them were positive by both the irut and the crut, but nega- Vol 37 No. 2 March
5 SOUTHEAST ASIAN J TROP MED PUBLIC HEALTH Table 4 Sensitivity, specificity, positive and negative predictive values, and accuracy of crut with the gold standard c. Time Sensitivity (%) Specificity (%) PPV a (%) NPV b (%) Accuracy (%) 30 minutes 87.5 (42/48) 100 (72/72) 100 (42/42) 92.3 (72/78) 95 (114/120) 1 hour 89.8 (44/49) 100 (71/71) 100 (44/44) 93.4 (71/76) 95.8 (115/120) 2 hours 92 (46/50) 100 (70/70) 100 (46/46) 94.6 (70/74) 96.7 (116/120) 24 hours 100 (51/51) 94.2 (65/69) 92.7 (51/55) 100 (65/65) 96.7 (116/120) a Positive predictive value; b Negative predictive value; c The criteria for the gold standard must be positive culture and/or positive histology and the irut. Table 5 Percentages of positive reactions by time interval for irut and crut. RUT Percentage of true positives by time interval c 30 min > 30 min - 1 hr > 1 hr - 2 hr > 2 hr - 24 hr In house (N = 48) 77.1 (37/48) 2.1 (1/48) 2.1 (1/48) 18.7 (9/48) Commercial (N = 51) 82.4 (42/51) 3.9 (2/51) 3.9 (2/51) 9.8 (5/51) c True positives for both crut and irut compared with gold standard criteria Table 6 Agreement between irut and crut, culture and histological examination. Time Kappa value a crut Culture Histology 30 min hr hr hrs Kappa value a < 0.0, poor; , slight; , fair; , moderate; , substantial; , almost perfect tive by culture and histology. The positive reactions according to time for irut and crut are shown in Table 5. The agreement between irut and crut, culture and histology analyzed by the kappa statistic is shown in Table 6. The agreement was very good (kappa > 0.81) between irut and crut. The medium was stable for up to 5 months after being stored in a refrigerator and protected from light (data not shown). DISCUSSION Dyspeptic patients infected with Helicobacter pylori are generally investigated by the physician during endoscopy. Detection of H. pylori infection on gastric biopsy specimens commonly uses RUT because the results can be interpreted easily, rapidly and can give a result before patient is discharged from the endoscope room (Kuo et al, 2002; Lim et al, 2004). Some results for crut, such as Pronto Dry and CLO, were compared. The findings show that the Pronto Dry test has a quicker positive reaction time and the positive color change is more distinct (Said et al, 2004). Although highly sensitivie and specific, the cost is relatively expensive. A rapid and economical RUT is needed to give results before the patient leaves the endoscope unit. Some researchers developed an irut with a sensitivity and specificity comparable to histological examination of between 65% and 100% at 4 hours, and 83% to 100% at 24 hours, respectively (Cifuentes et al, 2002). Our study was designed to evaluate the performance of an irut for the diagnosis of H. pylori infection, using culture, histology and crut (Pronto Dry) as 316 Vol 37 No. 2 March 2006
6 IN-HOUSE RAPID UREASE TEST FOR H. PYLORI DETECTION the gold standard. For the best medium for the irut, we compared buffered medium, unbuffered medium and modified Christensen urease with different amounts of phenol red. The unbuffered mediums immediately changed color when tested with a high concentration of bacteria, but it was difficult to differentiate between a negative and a weakly positive reaction. There was also poor specificity, with false positives occuring in less than 24 hours (data not shown). Of the three buffered media, we chose formula II as the most suitable medium because it was easy to determine a positive result within 1 hour. Color change could be observed when the organisms were at a low concentration of approximately cells ( organisms/ml). This is comparable to other investigations where a positive RUT required the presence of approximately10 5 to 10 7 organisms/ml (Vaira et al, 1988; Goldie et al, 1989). Some researchers have used liquid urease for the detection of H. pylori. The results showed sensitivity and specificity of 91% and 88%, respectively (Montes et al, 2003). A previous report showed RUT broth was unstable, required the addition of phenol red indicator before use and required many more organisms ( 1 x 10 6 ) to show a color change (Thillainayagam et al, 1991). There were 3 (2.5%) false negative specimens on the irut (Table 2). However, the specificity of the irut was excellent, there were no false positive results found in 1 hour and only 3.3% (4/120) were seen on irut and cruts at 24 hours (Table 2). Most irut and crut showed positive results within 30 minutes in 77.1% and 87.5%, respectively. The sensitivity and specificity of irut increased to 94.1% and 100% at 24 hours, similar to previous studies. Previous researchers have reported that RUT increased in sensitivity and reduced in specificity with longer incubation times (Adesanya et al, 2002; Cifuentes et al, 2002; Viiala et al, 2002; Lim et al, 2004). Several factors may affect the results on RUT, such as the amount of urea, the incubation temperature, the indicator used in the medium and the number of biopsies used in a RUT. In our study, we modified the formula of irut based on the Christensen urease test that used 2% urea and 0.12% phenol red as an indicator. It has been suggested that increasing the concentration of urea to 3% may increase the sensitivity of the irut. Mobley et al (1988) reported the optimal temperature of urease activity for H. pylori is 45ºC. Laine et al (1996) reported incubation of CLO test at 37ºC hastened the time to a positive test and specificity was not influenced by warming. Thus, increasing the incubation temperature of irut from 37ºC to 45ºC may accelerate the color change. The number of biopsies used in a RUT affects the results because some reseachers have shown that taking two biopsies instead of one has resulted in an earlier positive RUT result (Lim et al, 2004). Therefore, we used one antral and one corpus biopsy specimen in our RUT test. McNulty et al (1989) suggested that adjustment of the ph of the medium and the use of an indicator with a ph range closer to ideal ph of urease activity (ph 8.2) may increase the sensitivity and decrease the time to develop a positive result. The use of an indicator with a higher ph range than phenol red (ph range ), such as m-cresol purple (ph range ) or thymol blue (ph range ), and a buffer with a higher pk should be evaluated further (McNulty et al, 1989). However, phenol red at a ph of 6.8 is generally used as an indicator (Adesanya et al, 2002). We found the agreement between the irut and cruts was very good or almost perfect (kappa > 0.82) whereas it was moderate compared to histology (kappa = ) and substantial compared to culture (kappa = ). The irut was slightly less sensitive than the crut, however, the irut was easy to prepare, had a shelf-life of up to 5 months and the unit cost was approximately 20 times cheaper than the crut. The results indicate that the irut may replace the crut in our Endoscopy Unit for routine and rapid diagnosis of H. pylori infection. ACKNOWLEDGEMENTS This study was supported by a research grant from the Faculty of Medicine, Khon Kaen University, Thailand. We would like to thank the Vol 37 No. 2 March
7 SOUTHEAST ASIAN J TROP MED PUBLIC HEALTH staff of the Endoscopy Unit for their kind help with specimen collection and Mr Bryan Roderick Hamman for his assistance in the English language of the manuscript. REFERENCES Adesanya AA, Oluwatowoju IO, Oyedeji KS, et al. Evaluation of a locally-made urease test for detecting Helicobacter pylori infection. Niger Postgrad Med J 2002; 9: Ansorg R, Von Recklinghausen G, Pomarius R, et al. Evaluation of techniques for isolation, subcultivation, and preservation of Helicobacter pylori. J Clin Microbiol 1991; 29: Cifuentes P, Topor J, Avagnina A, et al. [Validation of a rapid urease test for the diagnosis of Helicobacter pylori infection]. Acta Gastroenterol Latinoam 2002; 32: Cohen H, Laine L. Endoscopic methods for the diagnosis of Helicobacter pylori. Aliment Pharmacol Ther 1997; 11 (suppl 1): 3-9. Das SS, Bain LA, Karim QN, et al. Rapid diagnosis of Campylobacter pyloridis infection. J Clin Pathol 1987; 40: Fabre R, Sobhani I, Laurent-Puig P, et al. Polymerase chain reaction assay for the detection of Helicobacter pylori in gastric biopsy specimens: comparison with culture, rapid urease test, and histopathological tests. Gut 1994; 35: Forman D, Webb P, Parsonnet J. H. pylori and gastric cancer. Lancet 1994; 343: Goldie J, Veldhuyzen van Zanten SJ, Jalali S, et al. Optimization of a medium for the rapid urease test for detection of Campylobacter pylori in gastric antral biopsies. J Clin Microbiol 1989; 27: Hazell SL, Borody TJ, Gal A, et al. Campylobacter pyloridis gastritis I: Detection of urease as a marker of bacterial colonization and gastritis. Am J Gastroenterol 1987; 82: Hazell SL, Markesich DC, Evans DJ, et al. Influence of media supplements on growth and survival of Campylobacter pylori. Eur J Clin Microbiol Infect Dis 1989; 8: Heatley RV. The Helicobacter pylori handbook. Leeds: Blackwell Science, Ho AS, Young TH, Shyu RY, et al. The accuracy of the rapid urease test and 13C-urea breath test in the diagnosis of Helicobacter pylori infection. Zhonghua Yi Xue Za Zhi (Taipei) 1996; 58: Kawanishi M, Fukuda S, Kawaguchi H, et al. Significance of rapid urease test for identification of Helicobacter pylori in comparison with histological and culture studies. J Gastroenterol 1995; 30: Kisa O, Albay A, Mas MR, et al. The evaluation of diagnostic methods for the detection of Helicobacter pylori in gastric biopsy specimens. Diagn Microbiol Infect Dis 2002; 43: Kuo CH, Wu DC, Lu CY, et al. The media of rapid urease test influence the diagnosis of Helicobacter pylori. Hepatogastroenterology 2002; 49: Laine L, Estrada R, Lewin DN, et al. The influence of warming on rapid urease test results: a prospective evaluation. Gastrointest Endosc 1996; 44: Landis JR, Koch GG. The measurement of observer agreement for categorical data. Biometrics 1977; 33: Li YH, Guo H, Zhang PB, et al. Clinical value of Helicobacter pylori stool antigen test, Immuno Card STAT HpSA, for detecting H. pylori infection. World J Gastroenterol 2004; 10: Liao CC, Lee CL, Lai YC, et al. Accuracy of three diagnostic tests used alone and in combination for detecting Helicobacter pylori infection in patients with bleeding gastric ulcers. Chin Med J 2003; 116: Lim LL, Ho KY, Ho B, et al. Effect of biopsies on sensitivity and specificity of ultra-rapid urease test for detection of Helicobacter pylori infection: a prospective evaluation. World J Gastroenterol 2004; 10: Logan RP, Walker MM. ABC of the upper gastrointestinal tract: Epidemiology and diagnosis of Helicobacter pylori infection. Br Med J 2001; 323: McNulty CA, Dent JC, Uff JS, et al. Detection of Campylobacter pylori by the biopsy urease test: an assessment in 1445 patients. Gut 1989; 30: Mobley HL, Cortesia MJ, Rosenthal LE, et al. Characterization of urease from Campylobacter pylori. J Clin Microbiol 1988; 26: Montes H, Salmen S, Dolfo W, et al. Evaluation of a liquid urease test (LUT) for detection of Helicobacter pylori. Acta Gastroenterol Latinoam 2003; 33: Morio O, Rioux-Leclercq N, Pagenault M, et al. Prospective evaluation of a new rapid urease test 318 Vol 37 No. 2 March 2006
8 IN-HOUSE RAPID UREASE TEST FOR H. PYLORI DETECTION (Pronto Dry) for the diagnosis of Helicobacter pylori infection. Gastroenterol Clin Biol 2004; 28: National Committee for Clinical Laboratory Standards., Performance standards for antimicrobial disk and dilution susceptibility tests for bacteria isolated from animals, approved standard. 2 nd ed. M31- A2. Wayne(PA): The committee, Onders RP. Detection methods of Helicobacter pylori: accuracy and costs. Am Surg 1997; 63: Pajares-Garcia JM. Diagnosis of Helicobacter pylori: invasive methods. Ital J Gastroenterol Hepatol 1998; 30 (suppl 3): S Parsonnet J. Gastric adenocarcinoma and Helicobacter pylori infection. West J Med 1994; 161: 60. Said RM, Cheah PL, Chin SC, et al. Evaluation of a new biopsy urease test: Pronto Dry, for the diagnosis of Helicobacter pylori infection. Eur J Gastroenterol Hepatol 2004; 16: Thillainayagam AV, Arvind AS, Cook RS, et al. Diagnostic efficiency of an ultrarapid endoscopy room test for Helicobacter pylori. Gut 1991; 32: Vaira D, Holton J, Cairns S, et al. Urease tests for Campylobacter pylori: care in interpretation. J Clin Pathol 1988; 41: Viiala CH, Windsor HM, Forbes GM, et al. Evaluation of a new formulation CLOtest. J Gastroenterol Hepatol 2002; 17: Xia H, Keane CT, Beattie S, et al. Standardization of disk diffusion test and its clinical significance for susceptibility testing of metronidazole against Helicobacter pylori. Antimicrob Agents Chemother 1994; 38: Vol 37 No. 2 March
Utility of In House made Rapid Urease Broth Test for Detection of Helicobacter pylori Infection in Resource Constraint Settings
 Original article: Utility of In House made Rapid Urease Broth Test for Detection of Helicobacter pylori Infection in Resource Constraint Settings 1.Dr. Swati Rahul Dhope, 2. Dr. Sachinkumar Vasantrao Wankhede
Original article: Utility of In House made Rapid Urease Broth Test for Detection of Helicobacter pylori Infection in Resource Constraint Settings 1.Dr. Swati Rahul Dhope, 2. Dr. Sachinkumar Vasantrao Wankhede
Comparison of the Accuracy of Two Commercial Rapid Urase Tests, CLOtest and Pronto Dry, in Detecting Helicobacter pylori Infection ABSTRACT
 Original Article Rojborwonwitaya J, Vijitjunyakul N THAI J GASTROENTEROL 2005 Vol. 6 No. 2 May - Aug. 2005 55 Comparison of the Accuracy of Two Commercial Rapid Urase Tests, CLOtest and Pronto Dry, in
Original Article Rojborwonwitaya J, Vijitjunyakul N THAI J GASTROENTEROL 2005 Vol. 6 No. 2 May - Aug. 2005 55 Comparison of the Accuracy of Two Commercial Rapid Urase Tests, CLOtest and Pronto Dry, in
Research Article Performance of Routine Helicobacter pylori Invasive Tests in Patients with Dyspepsia
 Gastroenterology Research and Practice Volume 2013, Article ID 184806, 5 pages http://dx.doi.org/10.1155/2013/184806 Research Article Performance of Routine Helicobacter pylori Invasive Tests in Patients
Gastroenterology Research and Practice Volume 2013, Article ID 184806, 5 pages http://dx.doi.org/10.1155/2013/184806 Research Article Performance of Routine Helicobacter pylori Invasive Tests in Patients
H.pylori IgA Cat #
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Evaluation of a new rapid immunoassay for the detection of Helicobacter pylori in faeces: a prospective pilot study
 Aliment Pharmacol Ther 2005; 21: 485 489. doi: 10.1111/j.1365-2036.2005.02355.x Evaluation of a new rapid immunoassay for the detection of Helicobacter pylori in faeces: a prospective pilot study L. TREVISANI*,
Aliment Pharmacol Ther 2005; 21: 485 489. doi: 10.1111/j.1365-2036.2005.02355.x Evaluation of a new rapid immunoassay for the detection of Helicobacter pylori in faeces: a prospective pilot study L. TREVISANI*,
Type of intervention Diagnosis. Economic study type Cost-effectiveness analysis.
 Noninvasive tests as a substitute for histology in the diagnosis of Helicobacter pylori infection Hahn M, Fennerty M B, Corless C L, Magaret N, Lieberman D A, Faigel D O Record Status This is a critical
Noninvasive tests as a substitute for histology in the diagnosis of Helicobacter pylori infection Hahn M, Fennerty M B, Corless C L, Magaret N, Lieberman D A, Faigel D O Record Status This is a critical
See external label 2 C-8 C Σ=96 tests Cat # 1505Z. MICROWELL ELISA H.Pylori IgA Cat # 1505Z
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
H.Pylori IgG
 DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
H.Pylori IgG Cat # 1503Z
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Helicobacter and gastritis
 1 Helicobacter and gastritis Dr. Hala Al Daghistani Helicobacter pylori is a spiral-shaped gram-negative rod. H. pylori is associated with antral gastritis, duodenal (peptic) ulcer disease, gastric ulcers,
1 Helicobacter and gastritis Dr. Hala Al Daghistani Helicobacter pylori is a spiral-shaped gram-negative rod. H. pylori is associated with antral gastritis, duodenal (peptic) ulcer disease, gastric ulcers,
(Received September 12, Accepted April 23, 1997) Jpn. J. Med. Sci. Biol., 50, 55-62, 1997.
 Jpn. J. Med. Sci. Biol., 50, 55-62, 1997. EVALUATION OF CULTURE, HISTOLOGICAL EXAMINATION, SEROLOGY AND THE RAPID UREASE TEST FOR DIAGNOSIS OF HELICOBACTER PYLORI IN PATIENTS WITH DYSPEPSIA IN BANGLADESH
Jpn. J. Med. Sci. Biol., 50, 55-62, 1997. EVALUATION OF CULTURE, HISTOLOGICAL EXAMINATION, SEROLOGY AND THE RAPID UREASE TEST FOR DIAGNOSIS OF HELICOBACTER PYLORI IN PATIENTS WITH DYSPEPSIA IN BANGLADESH
H. pylori IgM. Cat # H. pylori IgM ELISA. ELISA: Enzyme Linked Immunosorbent Assay. ELISA - Indirect; Antigen Coated Plate
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com H. pylori
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com H. pylori
Comparative study of invasive methods for diagnosis of Helicobacter pylori in humans
 ISSN: 2319-7706 Volume 2 Number 7 (2013) pp. 63-68 http://www.ijcmas.com Original Research Article Comparative study of invasive methods for diagnosis of Helicobacter pylori in humans V.Subbukesavaraja
ISSN: 2319-7706 Volume 2 Number 7 (2013) pp. 63-68 http://www.ijcmas.com Original Research Article Comparative study of invasive methods for diagnosis of Helicobacter pylori in humans V.Subbukesavaraja
H.Pylori IgM Cat # 1504Z
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
PCR-RFLP AND ANTIMICROBIAL SUSCEPTIBILITY PROFILES OF HELICOBACTER PYLORI ISOLATED FROM ANTRUM AND CORPUS OF DYSPEPTIC PATIENTS IN THAILAND
 PCR-RFLP and Susceptibility Profiles of H. pylori PCR-RFLP AND ANTIMICROBIAL SUSCEPTIBILITY PROFILES OF HELICOBACTER PYLORI ISOLATED FROM ANTRUM AND CORPUS OF DYSPEPTIC PATIENTS IN THAILAND Panthong Kulsantiwong
PCR-RFLP and Susceptibility Profiles of H. pylori PCR-RFLP AND ANTIMICROBIAL SUSCEPTIBILITY PROFILES OF HELICOBACTER PYLORI ISOLATED FROM ANTRUM AND CORPUS OF DYSPEPTIC PATIENTS IN THAILAND Panthong Kulsantiwong
H. pylori Antigen ELISA Kit
 H. pylori Antigen ELISA Kit Catalog Number KA3142 96 assays Version: 04 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
H. pylori Antigen ELISA Kit Catalog Number KA3142 96 assays Version: 04 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
H. pylori IgM CLIA kit
 H. pylori IgM CLIA kit Cat. No.:DEEL0251 Pkg.Size:96 tests Intended use Helicobacter pylori IgM Chemiluminescence ELISA is intended for use in evaluating the serologic status to H. pylori infection in
H. pylori IgM CLIA kit Cat. No.:DEEL0251 Pkg.Size:96 tests Intended use Helicobacter pylori IgM Chemiluminescence ELISA is intended for use in evaluating the serologic status to H. pylori infection in
PDF hosted at the Radboud Repository of the Radboud University Nijmegen
 PDF hosted at the Radboud Repository of the Radboud University Nijmegen The following full text is a publisher's version. For additional information about this publication click this link. http://hdl.handle.net/2066/48400
PDF hosted at the Radboud Repository of the Radboud University Nijmegen The following full text is a publisher's version. For additional information about this publication click this link. http://hdl.handle.net/2066/48400
Helicobacter Connections. Barry Marshall
 Helicobacter Connections Barry Marshall The greatest obstacle to knowledge is not ignorance, it is the illusion of knowledge. Daniel Boorstein - Historian Peptic Ulcers Duodenal Ulcer (DU) Gastric Ulcer
Helicobacter Connections Barry Marshall The greatest obstacle to knowledge is not ignorance, it is the illusion of knowledge. Daniel Boorstein - Historian Peptic Ulcers Duodenal Ulcer (DU) Gastric Ulcer
Correlation Between Endoscopic and Histological Findings in Different Gastroduodenal Lesion and its Association with Helicobacter Pylori
 ORIGINAL ARTICLE Correlation Between Endoscopic and Histological Findings in Different Gastroduodenal Lesion and its Association with Helicobacter Pylori *A. Sultana 1, SM Badruddoza 2, F Rahman 3 1 Dr.
ORIGINAL ARTICLE Correlation Between Endoscopic and Histological Findings in Different Gastroduodenal Lesion and its Association with Helicobacter Pylori *A. Sultana 1, SM Badruddoza 2, F Rahman 3 1 Dr.
Campylobacter like organisms on the gastric mucosa:
 J Clin Pathol 1984;37:1002-1006 Campylobacter like organisms on the gastric mucosa: culture, histological, and serological studies DM JONES,* AM LESSELLS,t JOAN ELDRIDGE* From the *Public Health Laboratory
J Clin Pathol 1984;37:1002-1006 Campylobacter like organisms on the gastric mucosa: culture, histological, and serological studies DM JONES,* AM LESSELLS,t JOAN ELDRIDGE* From the *Public Health Laboratory
Helicobacter Pylori Testing HELICOBACTER PYLORI TESTING HS-131. Policy Number: HS-131. Original Effective Date: 9/17/2009
 Easy Choice Health Plan, Inc. Harmony Health Plan of Illinois, Inc. Missouri Care, Inc. Ohana Health Plan, a plan offered by WellCare Health Insurance of Arizona, Inc. WellCare Health Insurance of Illinois,
Easy Choice Health Plan, Inc. Harmony Health Plan of Illinois, Inc. Missouri Care, Inc. Ohana Health Plan, a plan offered by WellCare Health Insurance of Arizona, Inc. WellCare Health Insurance of Illinois,
New immunoassay for the detection of Helicobacter pylori infection compared with urease test,
 Chapter V New immunoassay for the detection of Helicobacter pylori infection compared with urease test, 13C breath test and histology: validation in the primary care setting Catherine F Weijnen 1, Henriëtte
Chapter V New immunoassay for the detection of Helicobacter pylori infection compared with urease test, 13C breath test and histology: validation in the primary care setting Catherine F Weijnen 1, Henriëtte
Urea Breath Test for Diagnosis of Helicobactor pylori. Original Policy Date 12:2013
 MP 2.04.04 Urea Breath Test for Diagnosis of Helicobactor pylori Medical Policy Section Medicine Issue 12:2013 Original Policy Date 12:2013 Last Review Status/Date 12:2013 Return to Medical Policy Index
MP 2.04.04 Urea Breath Test for Diagnosis of Helicobactor pylori Medical Policy Section Medicine Issue 12:2013 Original Policy Date 12:2013 Last Review Status/Date 12:2013 Return to Medical Policy Index
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: helicobacter_pylori_testing 01/01/2019 N/A 01/01/2020 01/01/2019 Policy Effective April 1, 2019 Description
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: helicobacter_pylori_testing 01/01/2019 N/A 01/01/2020 01/01/2019 Policy Effective April 1, 2019 Description
Catalog # 0W009 0W010 Test Cassettes, murine monoclonal antibody to human IgG (Test Line) and rabbit polyclonal antibody (Control Line)
 For in vitro diagnostic use. CLIA Complexity for Whole Blood: WAIVED INTENDED USE The QuickVue H. pylori Test is a lateral-flow immunoassay intended for the rapid, qualitative detection of IgG antibodies
For in vitro diagnostic use. CLIA Complexity for Whole Blood: WAIVED INTENDED USE The QuickVue H. pylori Test is a lateral-flow immunoassay intended for the rapid, qualitative detection of IgG antibodies
Helicobacter pylori: Diagnosis, treatment and risks of untreated infection
 Helicobacter pylori: Diagnosis, treatment and risks of untreated infection Klaus Mönkemüller Department of Gastroenterology, Hepatology und Infectius Diseases Otto-von-Guericke University, Magdeburg bb
Helicobacter pylori: Diagnosis, treatment and risks of untreated infection Klaus Mönkemüller Department of Gastroenterology, Hepatology und Infectius Diseases Otto-von-Guericke University, Magdeburg bb
CLIA Complexity for Whole Blood: WAIVED. Intended Use. Summary and Explanation
 CLIA Complexity for Whole Blood: WAIVED Intended Use The is a lateral-flow immunoassay intended for the rapid, qualitative detection of IgG antibodies specific to Helicobacter pylori in whole blood. The
CLIA Complexity for Whole Blood: WAIVED Intended Use The is a lateral-flow immunoassay intended for the rapid, qualitative detection of IgG antibodies specific to Helicobacter pylori in whole blood. The
Helicobacter pylori Improved Detection of Helicobacter pylori
 DOI:http://dx.doi.org/10.7314/APJCP.2016.17.4.2099 RESEARCH ARTICLE Improved Detection of Helicobacter pylori Infection and Premalignant Gastric Mucosa Using Conventional White Light Source Gastroscopy
DOI:http://dx.doi.org/10.7314/APJCP.2016.17.4.2099 RESEARCH ARTICLE Improved Detection of Helicobacter pylori Infection and Premalignant Gastric Mucosa Using Conventional White Light Source Gastroscopy
Helicobacter pylori Eradication Therapy Success Regarding Different Treatment Period Based on Clarithromycin or Metronidazole Triple-Therapy Regimens
 Helicobacter ISSN 1523-5378 Filipec Blackwell Oxford, HEL 1083-4389 1523-5378 Journal XXX Original H. 2008 pylori Kanizaj compilation The UK Eradication Publishing Article Authors et al. Ltd 2008 Therapy
Helicobacter ISSN 1523-5378 Filipec Blackwell Oxford, HEL 1083-4389 1523-5378 Journal XXX Original H. 2008 pylori Kanizaj compilation The UK Eradication Publishing Article Authors et al. Ltd 2008 Therapy
Catalog # 0W009 0W010 Test Cassettes, murine monoclonal antibody to human IgG (Test Line) and rabbit polyclonal antibody (Control Line) Reagents
 For in vitro diagnostic use. CLIA Complexity: MODERATE INTENDED USE The QuickVue H. pylori Test is a lateral-flow immunoassay intended for the rapid, qualitative detection of IgG antibodies specific to
For in vitro diagnostic use. CLIA Complexity: MODERATE INTENDED USE The QuickVue H. pylori Test is a lateral-flow immunoassay intended for the rapid, qualitative detection of IgG antibodies specific to
Disclosures. Co-founder and Chief Science Officer, TechLab
 H. pylori testing Disclosures Co-founder and Chief Science Officer, TechLab Learning Objectives Evaluate the appropriate testing methodology by balancing performance, economics, and workflow. Discuss the
H. pylori testing Disclosures Co-founder and Chief Science Officer, TechLab Learning Objectives Evaluate the appropriate testing methodology by balancing performance, economics, and workflow. Discuss the
A PLACEBO CONTROLLED TRIAL OF BISMUTH SALICYLATE IN HELICOBACTER PYLORI ASSOCIATED GASTRITIS
 A PLACEBO CONTROLLED TRIAL OF BISMUTH SALICYLATE IN HELICOBACTER PYLORI ASSOCIATED GASTRITIS Pages with reference to book, From 154 To 156 Javed Iqbal Kazi, Naeem Aon Jafarey, Syed Mahmood Alam ( Department
A PLACEBO CONTROLLED TRIAL OF BISMUTH SALICYLATE IN HELICOBACTER PYLORI ASSOCIATED GASTRITIS Pages with reference to book, From 154 To 156 Javed Iqbal Kazi, Naeem Aon Jafarey, Syed Mahmood Alam ( Department
Fecal H. pylori Antigen Rapid Test (Strip)
 Fecal H. pylori Antigen Rapid Test (Strip) Cat. No.:DTS526 Pkg.Size: Intended use This H. pylori Antigen Rapid Test is intended for the direct qualitative detection of the presence of H. pylori antigen
Fecal H. pylori Antigen Rapid Test (Strip) Cat. No.:DTS526 Pkg.Size: Intended use This H. pylori Antigen Rapid Test is intended for the direct qualitative detection of the presence of H. pylori antigen
KEYWORDS Dyspepsia, Acid Peptic Disease, Helicobacter Pylori, Urease, Giemsa, Peptic Ulcer, Non-Ulcer Dyspepsia.
 INCIDENCE OF HELICOBACTER PYLORI WITH ACID PEPTIC DISEASE AND MALIGNANT CONDITIONS OF UPPER GASTROINTESTINAL TRACT IN A TERTIARY CENTRE - A PROSPECTIVE STUDY Karunamoorthy Rajachidambaram 1, Dinkaran Kaarthesan
INCIDENCE OF HELICOBACTER PYLORI WITH ACID PEPTIC DISEASE AND MALIGNANT CONDITIONS OF UPPER GASTROINTESTINAL TRACT IN A TERTIARY CENTRE - A PROSPECTIVE STUDY Karunamoorthy Rajachidambaram 1, Dinkaran Kaarthesan
Helicobacter 2008;13:1-6. Am J Gastroent 2007;102: Am J of Med 2004;117:31-35.
 An Update on Helicobacter pylori and Its Treatment Trenika Mitchell, PharmD, BCPS Clinical Assistant Professor University of Kentucky College of Pharmacy October 18, 2008 Objectives Review the epidemiology
An Update on Helicobacter pylori and Its Treatment Trenika Mitchell, PharmD, BCPS Clinical Assistant Professor University of Kentucky College of Pharmacy October 18, 2008 Objectives Review the epidemiology
Helicobacter pylori: drowning in a pool of blood?
 Helicobacter pylori: drowning in a pool of blood? Diagnosis of Helicobacter pylori infection is crucial in the short-term and long-term management of patients with bleeding ulcers. If a patient with ulcer
Helicobacter pylori: drowning in a pool of blood? Diagnosis of Helicobacter pylori infection is crucial in the short-term and long-term management of patients with bleeding ulcers. If a patient with ulcer
Low Recovery Rate of Helicobacter Pylori from Positive CLO Test Patients Suffering from Dyspepsia
 Bahrain Medical Bulletin, Vol. 37, No. 2, June 2015 Low Recovery Rate of Helicobacter Pylori from Positive CLO Test Patients Suffering from Dyspepsia Suliman Al Humayed, MBBS, SBIM, ABIM, MRCP* Martin
Bahrain Medical Bulletin, Vol. 37, No. 2, June 2015 Low Recovery Rate of Helicobacter Pylori from Positive CLO Test Patients Suffering from Dyspepsia Suliman Al Humayed, MBBS, SBIM, ABIM, MRCP* Martin
Rapid increase in the prevalence of metronidazole resistant Helicobacter pylori in the Netherlands.
 Chapter 2 Rapid increase in the prevalence of metronidazole resistant Helicobacter pylori in the Netherlands. E.J.van der Wouden 1, A.A.van Zwet 2, J.C.Thijs 1, G.D.C.Vosmaer 3, J.A.J.Oom 4, A.de Jong
Chapter 2 Rapid increase in the prevalence of metronidazole resistant Helicobacter pylori in the Netherlands. E.J.van der Wouden 1, A.A.van Zwet 2, J.C.Thijs 1, G.D.C.Vosmaer 3, J.A.J.Oom 4, A.de Jong
Prevalence of Helicobacter pylori in Patients with End Stage Renal Disease
 2000;20:97-102 Helicobacter pylori Prevalence of Helicobacter pylori in Patients with End Stage Renal Disease Do Ha Kim, M.D., Hwoon-Yong Jung, M.D., Suk-Kyun Yang, M.D. Weon-Seon Hong, M.D. and Young
2000;20:97-102 Helicobacter pylori Prevalence of Helicobacter pylori in Patients with End Stage Renal Disease Do Ha Kim, M.D., Hwoon-Yong Jung, M.D., Suk-Kyun Yang, M.D. Weon-Seon Hong, M.D. and Young
Diagnostic accuracy of reused Pronto Dry test and CLOtest in the detection of Helicobacter pylori infection
 Jamaludin et al. BMC Gastroenterology (2015) 15:101 DOI 10.1186/s12876-015-0332-0 RESEARCH ARTICLE Diagnostic accuracy of reused Pronto Dry test and CLOtest in the detection of Helicobacter pylori infection
Jamaludin et al. BMC Gastroenterology (2015) 15:101 DOI 10.1186/s12876-015-0332-0 RESEARCH ARTICLE Diagnostic accuracy of reused Pronto Dry test and CLOtest in the detection of Helicobacter pylori infection
RESEARCH ARTICLE. Abstract. Introduction
 DOI:10.22034/APJCP.2017.18.4.927 Outcomes of a Randomized Controlled Trial Comparing Modified High Dose Omeprazole RESEARCH ARTICLE Outcomes of a Randomized Controlled Trial Comparing Modified High Dose
DOI:10.22034/APJCP.2017.18.4.927 Outcomes of a Randomized Controlled Trial Comparing Modified High Dose Omeprazole RESEARCH ARTICLE Outcomes of a Randomized Controlled Trial Comparing Modified High Dose
Rapid-VIDITEST. Helicobacter pylori. One step Helicobacter pylori Blister test. Instruction manual
 Rapid-VIDITEST Helicobacter pylori One step Helicobacter pylori Blister test. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, 252 50 Vestec, Czech Republic, Tel.: +420 261 090 565,
Rapid-VIDITEST Helicobacter pylori One step Helicobacter pylori Blister test. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, 252 50 Vestec, Czech Republic, Tel.: +420 261 090 565,
Peptic Ulcer Disease Update
 Peptic Ulcer Disease Update Col Pat Storms RAM 2005 Disclosure Information 84th Annual AsMA Scientific Meeting Col Patrick Storms I have no financial relationships to disclose. I will discuss the following
Peptic Ulcer Disease Update Col Pat Storms RAM 2005 Disclosure Information 84th Annual AsMA Scientific Meeting Col Patrick Storms I have no financial relationships to disclose. I will discuss the following
EXERCISE. Proteins,Amino Acids, and Enzymes VII: Oxidase Test. Suggested Reading in Textbook. Pronunciation Guide. Materials per Student
 EXERCISE 30 Proteins,Amino Acids, SAFETY CONSIDERATIONS Be careful with the Bunsen burner flame. No mouth pipetting. The oxidase reagent is caustic. Avoid contact with eyes and skin. In case of contact,
EXERCISE 30 Proteins,Amino Acids, SAFETY CONSIDERATIONS Be careful with the Bunsen burner flame. No mouth pipetting. The oxidase reagent is caustic. Avoid contact with eyes and skin. In case of contact,
S. aureus NCTC 6571, E. coli NCTC (antibiotic
 ISO Sensitivity Test Agar Code: KM1204 A semi-defined nutritionally rich sensitivity medium. It is composed of specially selected peptones with a small amount of glucose, solidified with a very pure agar
ISO Sensitivity Test Agar Code: KM1204 A semi-defined nutritionally rich sensitivity medium. It is composed of specially selected peptones with a small amount of glucose, solidified with a very pure agar
NCCLS. Accutest H. pylori WB Test. (Catalog. No. ID-92A225) For Professional and In Vitro Diagnostic Use Only
 NCCLS Accutest H. pylori WB Test (Catalog. No. ID-92A225) For Professional and In Vitro Diagnostic Use Only A Rapid Visual Test for the Qualitative Detection of IgG Antibodies Specific to Helicobacter
NCCLS Accutest H. pylori WB Test (Catalog. No. ID-92A225) For Professional and In Vitro Diagnostic Use Only A Rapid Visual Test for the Qualitative Detection of IgG Antibodies Specific to Helicobacter
The significance of Helicobacter pylori in the approach of dyspepsia in primary care Arents, Nicolaas Lodevikus Augustinus
 University of Groningen The significance of Helicobacter pylori in the approach of dyspepsia in primary care Arents, Nicolaas Lodevikus Augustinus IMPORTANT NOTE: You are advised to consult the publisher's
University of Groningen The significance of Helicobacter pylori in the approach of dyspepsia in primary care Arents, Nicolaas Lodevikus Augustinus IMPORTANT NOTE: You are advised to consult the publisher's
Fecoprevalence and determinants of Helicobacter pylor infection among asymptomatic children in Myanmar
 International Journal of Gastroenterology, Hepatology, Transplant & Nutrition Original Article Fecoprevalence and determinants of Helicobacter pylor infection among asymptomatic children in Myanmar Hnin
International Journal of Gastroenterology, Hepatology, Transplant & Nutrition Original Article Fecoprevalence and determinants of Helicobacter pylor infection among asymptomatic children in Myanmar Hnin
instrument. When 13C-UBT positive value is greater than or equal to / - 0.4, the the subject can be 1. Data and methods details are as follows:
 Application of 13 C-urea breath test in screening helicobacter pylori infection during health examination in Chengdu, Sichuan YANG Yan-hua. LIU Yu-ping. CHENG You-fu, SHUAI Ping. LU Qiao. ZHENG Xiao-xia,
Application of 13 C-urea breath test in screening helicobacter pylori infection during health examination in Chengdu, Sichuan YANG Yan-hua. LIU Yu-ping. CHENG You-fu, SHUAI Ping. LU Qiao. ZHENG Xiao-xia,
Table 2.9. Case control studies of helicobacter pylori infection and oesophageal adenocarcinoma
 Characteristics of Characteristics of controls Detection Chow et al (1998) 1993-1995 129 of newly diagnosed oesophageal/gastric cardia (OGC) adenocarcinoma. 224 population controls selected by random digit
Characteristics of Characteristics of controls Detection Chow et al (1998) 1993-1995 129 of newly diagnosed oesophageal/gastric cardia (OGC) adenocarcinoma. 224 population controls selected by random digit
H. pylori Stool Rapid Test (Cassette)
 H. pylori Stool Rapid Test (Cassette) Cat. No.:DTS590 Pkg.Size: Intended use The H. pylori Stool Cassette is an immunochromatographic screening assay for the qualitative detectionof Helicobacter pylori
H. pylori Stool Rapid Test (Cassette) Cat. No.:DTS590 Pkg.Size: Intended use The H. pylori Stool Cassette is an immunochromatographic screening assay for the qualitative detectionof Helicobacter pylori
COMPARISON OF ONCE-A-DAY VERSUS TWICE-A-DAY CLARITHROMYCIN IN TRIPLE THERAPY FOR HELICOBACTER PYLORI ERADICATION
 Phil J Gastroenterol 2006; 2: 25-29 COMPARISON OF ONCE-A-DAY VERSUS TWICE-A-DAY CLARITHROMYCIN IN TRIPLE THERAPY FOR HELICOBACTER PYLORI ERADICATION Marianne P Collado, Ma Fatima P Calida, Peter P Sy,
Phil J Gastroenterol 2006; 2: 25-29 COMPARISON OF ONCE-A-DAY VERSUS TWICE-A-DAY CLARITHROMYCIN IN TRIPLE THERAPY FOR HELICOBACTER PYLORI ERADICATION Marianne P Collado, Ma Fatima P Calida, Peter P Sy,
JMSCR Vol 05 Issue 08 Page August 2017
 www.jmscr.igmpublication.org Impact Factor 5.84 Index Copernicus Value: 83.27 ISSN (e)-2347-176x ISSN (p) 2455-0450 DOI: https://dx.doi.org/10.18535/jmscr/v5i8.83 Role of Special Stain Giemsa in Demonstration
www.jmscr.igmpublication.org Impact Factor 5.84 Index Copernicus Value: 83.27 ISSN (e)-2347-176x ISSN (p) 2455-0450 DOI: https://dx.doi.org/10.18535/jmscr/v5i8.83 Role of Special Stain Giemsa in Demonstration
Imprint cytology: a simple, cost effectiveness analysis for diagnosing Helicobacter pylori, in west of Iran
 Original Research Medical Journal of Islamic Republic of Iran, Vol. 26, No. 1, Feb. 2012, pp. 12-16 Imprint cytology: a simple, cost effectiveness analysis for diagnosing Helicobacter pylori, in west of
Original Research Medical Journal of Islamic Republic of Iran, Vol. 26, No. 1, Feb. 2012, pp. 12-16 Imprint cytology: a simple, cost effectiveness analysis for diagnosing Helicobacter pylori, in west of
RESEARCH COMMUNICATION. Comparison of Helicobacter pylori Antibody Detection in Stool with other Diagnostic Tests for Infection
 Comparison of Helicobacter pylori Antibody Detection in Stool with other Diagnostic Tests RESEARCH COMMUNICATION Comparison of Helicobacter pylori Antibody Detection in Stool with other Diagnostic Tests
Comparison of Helicobacter pylori Antibody Detection in Stool with other Diagnostic Tests RESEARCH COMMUNICATION Comparison of Helicobacter pylori Antibody Detection in Stool with other Diagnostic Tests
Intragastric Acidification Reduces the Occurrence of False-Negative Urea Breath Test Results in Patients Taking a Proton Pump Inhibitor
 THE AMERICAN JOURNAL OF GASTROENTEROLOGY Vol. 96, No. 4, 2001 2001 by Am. Coll. of Gastroenterology ISSN 0002-9270/01/$20.00 Published by Elsevier Science Inc. PII S0002-9270(01)02250-X Intragastric Acidification
THE AMERICAN JOURNAL OF GASTROENTEROLOGY Vol. 96, No. 4, 2001 2001 by Am. Coll. of Gastroenterology ISSN 0002-9270/01/$20.00 Published by Elsevier Science Inc. PII S0002-9270(01)02250-X Intragastric Acidification
Acid-Peptic Diseases of the Stomach and Duodenum Including Helicobacter pylori and NSAIDs Prof. Sheila Crowe
 Acid-Peptic Diseases of the Stomach and Duodenum Including Helicobacter pylori and NSAIDs 1 Division of Gastroenterology UC San Diego School of Medicine Clinical presentations of Helicobacter pylori infection
Acid-Peptic Diseases of the Stomach and Duodenum Including Helicobacter pylori and NSAIDs 1 Division of Gastroenterology UC San Diego School of Medicine Clinical presentations of Helicobacter pylori infection
Assessment of symptomatic response as predictor of Helicobacter pylori status following eradication therapy in patients with ulcer
 618 University Department of Medicine and Therapeutics, Western Infirmary, Glasgow G11 6NT, UK K E L McColl A El-Nujumi L S Murray E M El-Omar A Dickson A W Kelman T E Hilditch Correspondence to: Professor
618 University Department of Medicine and Therapeutics, Western Infirmary, Glasgow G11 6NT, UK K E L McColl A El-Nujumi L S Murray E M El-Omar A Dickson A W Kelman T E Hilditch Correspondence to: Professor
- Helicobacter - THE EASE AND DIFFICULTY OF A NEW DISCOVERY. Robin Warren
 - Helicobacter - THE EASE AND DIFFICULTY OF A NEW DISCOVERY Robin Warren EARLY DAYS First reports 100 years ago considered spirochaetes 1940 Freedburg saw curved organisms in the stomach 1954 Palmer: Freedburg
- Helicobacter - THE EASE AND DIFFICULTY OF A NEW DISCOVERY Robin Warren EARLY DAYS First reports 100 years ago considered spirochaetes 1940 Freedburg saw curved organisms in the stomach 1954 Palmer: Freedburg
Sequioa Education Systems, Inc. 1
 Functional Diagnostic Medicine Training Program Module 2 The Functional Diagnostic Medicine Approach in the Treatment of Gastrointestinal Dysfunction and Disease Dr. Wayne L. Sodano, D.C., D.A.B.C.I. &
Functional Diagnostic Medicine Training Program Module 2 The Functional Diagnostic Medicine Approach in the Treatment of Gastrointestinal Dysfunction and Disease Dr. Wayne L. Sodano, D.C., D.A.B.C.I. &
594 Lewin, Weinstein, and Riddell s Gastrointestinal Pathology and Its Clinical Implications
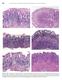 594 Lewin, Weinstein, and Riddell s Gastrointestinal Pathology and Its Clinical Implications Figure 13-20. Stages in the natural history of H. pylori. Biopsies from the antrum are on the left and the oxyntic
594 Lewin, Weinstein, and Riddell s Gastrointestinal Pathology and Its Clinical Implications Figure 13-20. Stages in the natural history of H. pylori. Biopsies from the antrum are on the left and the oxyntic
American College of Gastroenterology Guideline on the Management of Helicobacter pylori Infection
 American Journal of Gastroenterology ISSN 0002-9270 C 2007 by Am. Coll. of Gastroenterology doi: 10.1111/j.1572-0241.2007.01393.x Published by Blackwell Publishing American College of Gastroenterology
American Journal of Gastroenterology ISSN 0002-9270 C 2007 by Am. Coll. of Gastroenterology doi: 10.1111/j.1572-0241.2007.01393.x Published by Blackwell Publishing American College of Gastroenterology
MEDICAL POLICY EFFECTIVE DATE: 05/19/11 REVISED DATE: 05/24/12, 05/23/13 ARCHIVED DATE: 05/22/14 EDITED DATE: 05/28/15, 05/25/16, 05/18/17, 05/17/18
 MEDICAL POLICY SUBJECT: NON-INVASIVE HELICOBACTER PAGE: 1 OF: 6 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including
MEDICAL POLICY SUBJECT: NON-INVASIVE HELICOBACTER PAGE: 1 OF: 6 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including
PEPTIC ULCER DISEASE JOHN R SALTZMAN, MD. Director of Endoscopy Brigham and Women s Hospital Professor of Medicine Harvard Medical School
 PEPTIC ULCER DISEASE JOHN R SALTZMAN, MD Director of Endoscopy Brigham and Women s Hospital Professor of Medicine Harvard Medical School No disclosures Disclosures Overview Causes of peptic ulcer disease
PEPTIC ULCER DISEASE JOHN R SALTZMAN, MD Director of Endoscopy Brigham and Women s Hospital Professor of Medicine Harvard Medical School No disclosures Disclosures Overview Causes of peptic ulcer disease
Helicobacter pylori 幽門螺旋桿菌 馬偕紀念醫院新竹分院一般內科, 肝膽腸胃科陳重助醫師
 Helicobacter pylori 幽門螺旋桿菌 馬偕紀念醫院新竹分院一般內科, 肝膽腸胃科陳重助醫師 Hp : Helicobacter pylori Part 1. Pathophysiology and immune response Pathogenesis of Hp infection Part 2. Clinical manifestation Part 3. Dx tests for
Helicobacter pylori 幽門螺旋桿菌 馬偕紀念醫院新竹分院一般內科, 肝膽腸胃科陳重助醫師 Hp : Helicobacter pylori Part 1. Pathophysiology and immune response Pathogenesis of Hp infection Part 2. Clinical manifestation Part 3. Dx tests for
Management of dyspepsia and of Helicobacter pylori infection
 Management of dyspepsia and of Helicobacter pylori infection The University of Nottingham John Atherton Wolfson Digestive Diseases Centre University of Nottingham, UK Community management of dyspepsia
Management of dyspepsia and of Helicobacter pylori infection The University of Nottingham John Atherton Wolfson Digestive Diseases Centre University of Nottingham, UK Community management of dyspepsia
Research Article Reliability of Diagnostic Tests for Helicobacterpylori Infection
 Gastroenterology Research and Practice Volume 211, Article ID 946, 6 pages doi:1.1155/211/946 Research Article Reliability of Diagnostic Tests for Helicobacterpylori Infection S. Redéen, 1, 2 F. Petersson,
Gastroenterology Research and Practice Volume 211, Article ID 946, 6 pages doi:1.1155/211/946 Research Article Reliability of Diagnostic Tests for Helicobacterpylori Infection S. Redéen, 1, 2 F. Petersson,
JJBS Jordan Journal of Biological Sciences
 JJBS Jordan Journal of Biological Sciences Volume 4, Number 2, June 2011 ISSN 1995-6673 Pages 71-76 Prevalence of Helicobacter Pylori Gastritis at the North of Jordan N. M. Abu-Ahmad, A. Odeh and A-K.
JJBS Jordan Journal of Biological Sciences Volume 4, Number 2, June 2011 ISSN 1995-6673 Pages 71-76 Prevalence of Helicobacter Pylori Gastritis at the North of Jordan N. M. Abu-Ahmad, A. Odeh and A-K.
Realities of Diagnosing Helicobacter pylon Infection in Clinical Practice: A Case for Non-invasive Indirect Methodologies
 YALE JOURNAL OF BIOLOGY AND MEDICINE 71 (1998), pp. 81-90. Copyright 1999. All rights reserved. Realities of Diagnosing Helicobacter pylon Infection in Clinical Practice: A Case for Non-invasive Indirect
YALE JOURNAL OF BIOLOGY AND MEDICINE 71 (1998), pp. 81-90. Copyright 1999. All rights reserved. Realities of Diagnosing Helicobacter pylon Infection in Clinical Practice: A Case for Non-invasive Indirect
Campylobacter pylori in non-specific abdominal pain in childhood
 Gut, 1989, 3, 912-916 Serum pepsinogen I and IgG antibody to Campylobacter pylori in non-specific abdominal pain in childhood GIUSEPPINA ODERDA, D VAIRA, J HOLTON, J F DOWSETT, AND NICOLETTA ANSALDI From
Gut, 1989, 3, 912-916 Serum pepsinogen I and IgG antibody to Campylobacter pylori in non-specific abdominal pain in childhood GIUSEPPINA ODERDA, D VAIRA, J HOLTON, J F DOWSETT, AND NICOLETTA ANSALDI From
An evaluation of whole blood testing for Helicobacter pylori in general practice
 Aliment Pharmacol Ther 1998; 12: 641±645. An evaluation of whole blood testing for Helicobacter pylori in general practice N. J. TALLEY, J. R. LAMBERT*, S. HOWELL, H. H.-X. XIA, S. K. LIN* & L. AGREUS
Aliment Pharmacol Ther 1998; 12: 641±645. An evaluation of whole blood testing for Helicobacter pylori in general practice N. J. TALLEY, J. R. LAMBERT*, S. HOWELL, H. H.-X. XIA, S. K. LIN* & L. AGREUS
An evaluation of 2.0 McFarland Etest method for detection of heterogeneous vancomycin-intermediate Staphylococcus aureus
 Asian Biomedicine Vol. 4 No. 1 February 2010; 141-145 Brief communication (Original) An evaluation of 2.0 McFarland Etest method for detection of heterogeneous vancomycin-intermediate Staphylococcus aureus
Asian Biomedicine Vol. 4 No. 1 February 2010; 141-145 Brief communication (Original) An evaluation of 2.0 McFarland Etest method for detection of heterogeneous vancomycin-intermediate Staphylococcus aureus
TRANSPARENCY COMMITTEE OPINION. 13 December 2006
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 13 December 2006 HELIKIT 75 mg, powder for oral solution CIP : 343 132-1 Applicant : MAYOLY SPINDLER 13 Curea anhydrous
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 13 December 2006 HELIKIT 75 mg, powder for oral solution CIP : 343 132-1 Applicant : MAYOLY SPINDLER 13 Curea anhydrous
Failure of first-line eradication treatment significantly increases prevalence of antimicrobial-resistant Helicobacter pylori clinical isolates
 cp60392 Module 1 Journal of Clinical Pathology 28/8/08 12:38:20 Topics: ; Failure of first-line eradication treatment significantly increases prevalence of antimicrobial-resistant Helicobacter pylori clinical
cp60392 Module 1 Journal of Clinical Pathology 28/8/08 12:38:20 Topics: ; Failure of first-line eradication treatment significantly increases prevalence of antimicrobial-resistant Helicobacter pylori clinical
ENG MYCO WELL D- ONE REV. 1.UN 29/09/2016 REF. MS01283 REF. MS01321 (COMPLETE KIT)
 ENG MYCO WELL D- ONE MYCO WELL D-ONE System for the presumptive identification and antimicrobial susceptibility test of urogenital mycoplasmas, Gardnerella vaginalis, Trichomonas vaginalis, Candida albicans
ENG MYCO WELL D- ONE MYCO WELL D-ONE System for the presumptive identification and antimicrobial susceptibility test of urogenital mycoplasmas, Gardnerella vaginalis, Trichomonas vaginalis, Candida albicans
EFFECT OF SOME CHEMICAL AND PHYSICAL FACTORS ON THE COCCI SHAPE FORMATION OF HELICOBACTER PYLORI ISOLATED FROM PATIENTS WITH DUODENUM ULCER
 . Helicobacter pylori. Helicobacterpylori (Duodenal ulcer) (Ski row medium) H. pylori (Nalidixic Acid).(Cephalothin) H. pylori (%). ( ) ().(86.9) (PH) (%) EFFECT OF SOME CHEMICAL AND PHYSICAL FACTORS ON
. Helicobacter pylori. Helicobacterpylori (Duodenal ulcer) (Ski row medium) H. pylori (Nalidixic Acid).(Cephalothin) H. pylori (%). ( ) ().(86.9) (PH) (%) EFFECT OF SOME CHEMICAL AND PHYSICAL FACTORS ON
MOSAIC APPEARANCE OF GASTRIC MUCOSA AS A PREDICTOR FOR HELICOBACTER PYLORI INFECTION
 Basrah Journal Original Article Of Surgery MOSAIC APPEARANCE OF GASTRIC MUCOSA AS A PREDICTOR FOR HELICOBACTER PYLORI INFECTION Ali Adnan Mohsin* & Sarkis K Strak @ *MBChB Registrar, @ MBChB, MRCP, FRCP,
Basrah Journal Original Article Of Surgery MOSAIC APPEARANCE OF GASTRIC MUCOSA AS A PREDICTOR FOR HELICOBACTER PYLORI INFECTION Ali Adnan Mohsin* & Sarkis K Strak @ *MBChB Registrar, @ MBChB, MRCP, FRCP,
RAPID DIAGNOSIS OF HELICOBACTER PYLORI INFECTION IN GASTRIC IMPRINT SMEARS
 RAPID DIAGNOSIS OF HELICOBACTER PYLORI INFECTION IN GASTRIC IMPRINT SMEARS Gurjeet Kaur 1, Manoharan Madhavan 1, Amir Hakim Basri 2, Abdul Hamid Mat Sain 3, Mohd Shaiful Bahrun Hussain 3, Mohd Kamal Yatiban
RAPID DIAGNOSIS OF HELICOBACTER PYLORI INFECTION IN GASTRIC IMPRINT SMEARS Gurjeet Kaur 1, Manoharan Madhavan 1, Amir Hakim Basri 2, Abdul Hamid Mat Sain 3, Mohd Shaiful Bahrun Hussain 3, Mohd Kamal Yatiban
A Heaney, J S A Collins, RGPWatson, R J McFarland, K B Bamford, T C K Tham
 186 Royal Victoria Hospital, Belfast, A Heaney J S A Collins Department of Medicine, Queen s University, Belfast, RGPWatson Ulster Hospital, Dundonald, R J McFarland T C K Tham Department of Microbiology
186 Royal Victoria Hospital, Belfast, A Heaney J S A Collins Department of Medicine, Queen s University, Belfast, RGPWatson Ulster Hospital, Dundonald, R J McFarland T C K Tham Department of Microbiology
F Mirshahi, G Fowler, A Patel, G Shaw
 22 The School of Health and Sports Science, The University of North London, Holloway Road, London N7 8DB, UK F Mirshahi G Fowler A Patel G Shaw Correspondence to: Dr Shaw. Accepted for publication 11 November
22 The School of Health and Sports Science, The University of North London, Holloway Road, London N7 8DB, UK F Mirshahi G Fowler A Patel G Shaw Correspondence to: Dr Shaw. Accepted for publication 11 November
What is the status of Sequential Therapy Versus Standard Triple- Drug Therapy in peptic ulcer disease in eradicating H pylori?
 What is the status of Sequential Therapy Versus Standard Triple- Drug Therapy in peptic ulcer disease in eradicating H pylori? Sequential Therapy Versus Standard Triple- Drug Therapy for Helicobacter pylori
What is the status of Sequential Therapy Versus Standard Triple- Drug Therapy in peptic ulcer disease in eradicating H pylori? Sequential Therapy Versus Standard Triple- Drug Therapy for Helicobacter pylori
Immunoglobulin G Antibody against Helicobacter pylori: Clinical Implications of Levels Found in Serum
 CLINICAL AND DIAGNOSTIC LABORATORY IMMUNOLOGY, Sept. 2002, p. 1044 1048 Vol. 9, No. 5 1071-412X/02/$04.00 0 DOI: 10.1128/CDLI.9.5.1044 1048.2002 Copyright 2002, American Society for Microbiology. All Rights
CLINICAL AND DIAGNOSTIC LABORATORY IMMUNOLOGY, Sept. 2002, p. 1044 1048 Vol. 9, No. 5 1071-412X/02/$04.00 0 DOI: 10.1128/CDLI.9.5.1044 1048.2002 Copyright 2002, American Society for Microbiology. All Rights
Helicobacter pylori resistance in the Netherlands: a growing problem?
 ORIGINAL ARTICLE Helicobacter pylori resistance in the Netherlands: a growing problem? R. Ruiter 1,2 *, H.F. Wunderink 3, R.A. Veenendaal 5, L.G. Visser 4, M.G.J. de Boer 4 Departments of 1 Internal Medicine,
ORIGINAL ARTICLE Helicobacter pylori resistance in the Netherlands: a growing problem? R. Ruiter 1,2 *, H.F. Wunderink 3, R.A. Veenendaal 5, L.G. Visser 4, M.G.J. de Boer 4 Departments of 1 Internal Medicine,
Campylobacter. Helicobacter. Arcobacter 10/14/2011. General. Difference from Vibrio. Difference from Vibrio. Vibrio. Campylobacter
 Helicobacter Gram-negative Short, curved (comma shaped), spiral or S forms May turn coccoid in old cultures Oxidase + Urease negative cf. Helicobacter mostly urease positive Microaerophilic (3% - 15% O
Helicobacter Gram-negative Short, curved (comma shaped), spiral or S forms May turn coccoid in old cultures Oxidase + Urease negative cf. Helicobacter mostly urease positive Microaerophilic (3% - 15% O
SUMMARY INTRODUCTION. Accepted for publication 25 May 2005
 Aliment Pharmacol Ther 2005; 22: 243 249. doi: 10.1111/j.1365-2036.2005.02554.x Trends in the prevalence of peptic ulcer disease and Helicobacter pylori infection in family physician-referred uninvestigated
Aliment Pharmacol Ther 2005; 22: 243 249. doi: 10.1111/j.1365-2036.2005.02554.x Trends in the prevalence of peptic ulcer disease and Helicobacter pylori infection in family physician-referred uninvestigated
Three-day lansoprazole quadruple therapy for Helicobacter pylori-positive duodenal ulcers: a randomized controlled study
 Aliment Pharmacol Ther 2001; 15: 843±849. Three-day lansoprazole quadruple therapy for Helicobacter pylori-positive duodenal ulcers: a randomized controlled study B. C. Y. WONG*, W. H. WANG*, W.M.WONG*,G.K.K.LAU*,F.M.Y.FUNG*,N.N.S.KUNGà,
Aliment Pharmacol Ther 2001; 15: 843±849. Three-day lansoprazole quadruple therapy for Helicobacter pylori-positive duodenal ulcers: a randomized controlled study B. C. Y. WONG*, W. H. WANG*, W.M.WONG*,G.K.K.LAU*,F.M.Y.FUNG*,N.N.S.KUNGà,
Gastric atrophy: use of OLGA staging system in practice
 Gastroenterology and Hepatology From Bed to Bench. 2016 RIGLD, Research Institute for Gastroenterology and Liver Diseases ORIGINAL ARTICLE Gastric atrophy: use of OLGA staging system in practice Mahsa
Gastroenterology and Hepatology From Bed to Bench. 2016 RIGLD, Research Institute for Gastroenterology and Liver Diseases ORIGINAL ARTICLE Gastric atrophy: use of OLGA staging system in practice Mahsa
Microbiological Methods V-A- 1 SALMONELLA SPECIES PRESUMPTIVE AND CONFIRMATION TESTS
 Microbiological Methods V-A- 1 PRESUMPTIVE AND CONFIRMATION TESTS PRINCIPLE SCOPE Enrichment and selective procedures are used to provide a reasonably sensitive, definitive and versatile means of qualitatively
Microbiological Methods V-A- 1 PRESUMPTIVE AND CONFIRMATION TESTS PRINCIPLE SCOPE Enrichment and selective procedures are used to provide a reasonably sensitive, definitive and versatile means of qualitatively
PREVALENCE OF caga EPIYA MOTIFS IN HELICOBACTER PYLORI AMONG DYSPEPTIC PATIENTS IN NORTHEAST THAILAND
 PREVALENCE OF caga EPIYA MOTIFS IN HELICOBACTER PYLORI AMONG DYSPEPTIC PATIENTS IN NORTHEAST THAILAND Chariya Chomvarin 1, Karnchanawadee Phusri 1, Kookwan Sawadpanich 2, Pisaln Mairiang 2, Wises Namwat
PREVALENCE OF caga EPIYA MOTIFS IN HELICOBACTER PYLORI AMONG DYSPEPTIC PATIENTS IN NORTHEAST THAILAND Chariya Chomvarin 1, Karnchanawadee Phusri 1, Kookwan Sawadpanich 2, Pisaln Mairiang 2, Wises Namwat
The Nobel Prize in Physiology or Medicine for 2005
 The Nobel Prize in Physiology or Medicine for 2005 jointly to Barry J. Marshall and J. Robin Warren for their discovery of "the bacterium Helicobacter pylori and its role in gastritis and peptic ulcer
The Nobel Prize in Physiology or Medicine for 2005 jointly to Barry J. Marshall and J. Robin Warren for their discovery of "the bacterium Helicobacter pylori and its role in gastritis and peptic ulcer
Campylobacter Antigen ELISA Kit
 Campylobacter Antigen ELISA Kit Catalog Number KA3204 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Campylobacter Antigen ELISA Kit Catalog Number KA3204 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Detection of Helicobacter pylori in Gastroduodenal Diseases by Real Time PCR
 Detection of Helicobacter pylori in Gastroduodenal Diseases by Real Time PCR Abstract: We have investigated in the present study that H. pylori as causative agent of GC remain controversial; therefore,
Detection of Helicobacter pylori in Gastroduodenal Diseases by Real Time PCR Abstract: We have investigated in the present study that H. pylori as causative agent of GC remain controversial; therefore,
THE PREVALENCE OF HELICBACTER PYLORI AMONG PATIENTS COMPLAINING FROM ABDOMINAL PAIN
 THE PREVALENCE OF HELICBACTER PYLORI AMONG PATIENTS COMPLAINING FROM ABDOMINAL PAIN Ahed J. Al-Khatib Jordan University of Science and Technology, Jordan Ahmed Saber Abu-zaiton Al-albayt University Abstract
THE PREVALENCE OF HELICBACTER PYLORI AMONG PATIENTS COMPLAINING FROM ABDOMINAL PAIN Ahed J. Al-Khatib Jordan University of Science and Technology, Jordan Ahmed Saber Abu-zaiton Al-albayt University Abstract
CerTest Turbilatex. A quantitative immunological latex method. FOB Calprotectin Transferrin H. pylori
 CerTest Turbilatex A quantitative immunological latex method FOB Calprotectin Transferrin H. pylori Turbidimetric technique. A latex turbidimetric assay The turbidimetric assay is based on the agglutination
CerTest Turbilatex A quantitative immunological latex method FOB Calprotectin Transferrin H. pylori Turbidimetric technique. A latex turbidimetric assay The turbidimetric assay is based on the agglutination
Treatment of H. pylori Infection: The Reality
 YALE JOURNAL OF BIOLOGY AND MEDICINE 71 (1998), pp. 119-124. Copyright 1999. All rights reserved. Treatment of H. pylori Infection: The Reality Nimish Vakil University of Wisconsin Medical School, Milwaukee
YALE JOURNAL OF BIOLOGY AND MEDICINE 71 (1998), pp. 119-124. Copyright 1999. All rights reserved. Treatment of H. pylori Infection: The Reality Nimish Vakil University of Wisconsin Medical School, Milwaukee
Comparative study of rapid urease test and histopathological examination for detection of H. pylori infection
 International Surgery Journal Athavale VS et al. Int Surg J. 2017 Dec;4(12):4071-4075 http://www.ijsurgery.com pissn 2349-3305 eissn 2349-2902 Original Research Article DOI: http://dx.doi.org/10.18203/2349-2902.isj20175413
International Surgery Journal Athavale VS et al. Int Surg J. 2017 Dec;4(12):4071-4075 http://www.ijsurgery.com pissn 2349-3305 eissn 2349-2902 Original Research Article DOI: http://dx.doi.org/10.18203/2349-2902.isj20175413
CHANGING PATTERNS OF THE PREVALENCE OF HELICOBACTER PYLORI AMONG PATIENTS AT A CORPORATE HOSPITAL IN GHANA
 CHANGING PATTERNS OF THE PREVALENCE OF HELICOBACTER PYLORI AMONG PATIENTS AT A CORPORATE HOSPITAL IN GHANA R. DARKO 1, A. E. YAWSON 2, V. OSEI 3, J. OWUSU-ANSAH 3 and S. ALUZE-ELE 2 1 Department of Surgery,
CHANGING PATTERNS OF THE PREVALENCE OF HELICOBACTER PYLORI AMONG PATIENTS AT A CORPORATE HOSPITAL IN GHANA R. DARKO 1, A. E. YAWSON 2, V. OSEI 3, J. OWUSU-ANSAH 3 and S. ALUZE-ELE 2 1 Department of Surgery,
