Issue Date: December 29, 1982 Authority: 32 CFR 199.4(b)(2)(v), (b)(3)(iii), (b)(5)(v), (d)(3)(vi), and (e)(11)(i)
|
|
|
- Hector Casey
- 6 years ago
- Views:
Transcription
1 TRICARE/CHAMPUS POLICY MANUAL M DEC 1998 OTHER SERVICES CHAPTER 7 SECTION 7.1 Issue Date: December 29, 1982 Authority: 32 CFR 199.4(b)(2)(v), (b)(3)(iii), (b)(5)(v), (d)(3)(vi), and (e)(11)(i) I. POLICY A. Drugs and medications, including "unlabeled or off-label use", whether administered by a physician or obtained by prescription, are covered when all of the following conditions are met: 1. The drug is medically necessary for the treatment of the condition for which it is administered, according to accepted standards of medical practice. Approval for "unlabeled or off-label use" requires medical review for medical necessity. The review for medical necessity must include demonstrations from medical literature, national organizations, or national technology assessment bodies, that the "off-label or unlabeled" use of the drug is safe, effective, and in accordance with nationally accepted standards of practice in the medical community. If the "off-label or unlabeled" use of the drug does not meet all the above requirements, cost-sharing of the drug will be denied. 2. The drug is approved by the U.S. Food and Drug Administration for commercial marketing. Drugs grandfathered by the Federal Food, Drug and Cosmetic Act of 1938 may be cost-shared as if FDA approved. A drug that is approved for testing in humans is not covered. Information concerning FDA approved drugs may be obtained by calling (301) By law of the United States, the drug requires a physician's or other authorized individual professional provider's prescription (acting within the scope of their license) and are ordered or prescribed by a physician or other authorized individual professional provider. 4. The drug is prescribed and dispensed in accordance with state law and licensing requirements. 5. The drug is not prescribed or furnished by a member of the patient's immediate family. For information concerning immunizations, see Chapter 1, Section 9.1. For information concerning processing of prescription drugs and medicine claims, see the OPM Part Two, Chapter 1, Section V.C.5.h.(4). B. Insulin is covered for diabetic patients, regardless of whether a prescription is required under state law. 1
2 CHAPTER 7, SECTION 7.1 TRICARE/CHAMPUS POLICY MANUAL M DEC 1998 C. Vitamins may be covered only when used in accordance with nationally accepted standards of practice in the medical community as a specific treatment of a medical condition. D. Drugs which previously required a prescription but which are now deemed safe by the FDA for use without a doctor's prescription, and are available over-the-counter, can not be cost-shared. E. The current FDA-approved label supersedes the information contained in this policy. II. POLICY CONSIDERATIONS A. The following is a list of drugs that will or will not be cost-shared for "unlabeled or off-labeled" uses as specifically stated. 1. ACTH (adrenocorticotropic hormone) is covered when used in the treatment of multiple sclerosis and rheumatoid arthritis. 2. Adrenal cortex extract injections are not covered. 3. Anti-inhibitor coagulant complex (Autoplex ) is covered only when administered to hemophiliacs who have developed antibodies to antihemophilic factor. It is usually used in the treatment of massive bleeding or when surgery is necessary. All claims require a determination that the drug was medically necessary and that other appropriate means of dealing with the situation were not available. 4. Autogenous vaccines are not covered. 5. Azathioprine (Imuran ) is covered: a. For prophylaxis of organ rejections in patients receiving liver, liver-kidney, heart, lung, and heart-lung transplants, effective January 1, b. When used to induce and maintain remission of the symptoms of active inflammatory bowel disease, including Crohn s disease. Because of potential adverse effects, use of this drug requires thorough patient education and close physician supervision, effective July 15, Botulinum Toxin Type A (Botox ), see Chapter 1, Section Calcitriol (Rocaltrol ) is covered for the treatment of osteoporosis, effective June 1, Chymopapain is covered only for the treatment of herniated lumbar intervertebral discs in accordance with FDA requirements. See Chapter 3, Section Cladribine, 2CdA (Leustatin ) and Cyclophosphamide (Cytoxan ) are covered for the treatment of Waldenstrom s macroglobulinemia, effective February 26, These drugs may be administered subcutaneously or intravenously. 2
3 TRICARE/CHAMPUS POLICY MANUAL M DEC 1998 CHAPTER 7, SECTION Cyclophosphamide (Cytoxan, Neosar ) is covered for the treatment of lupus nephritis, effective March 1, Cyclosporine (Sandimmune ) is covered for the prophylaxis of rejection in patients receiving bone marrow transplantation. 12. Disodium edetate (EDTA), see Chapter 1, Section Dimethyl sulfoxide (DMSO), is covered when used in the treatment of interstitial cystitis. 14. Epogen (EPO, eythropoietin, Eprix, Procrit, Epoetin Alfa) may be covered for patients diagnosed with orthostatic hypotension (dysautonomia). 15. Epoprostenol Sodium (FLOLAN ) is covered for the management of severe secondary pulmonary hypertension for patients who have been authorized for lung transplant and are awaiting transplant, effective May 1, Gemcitabine (Gemzar ) is not covered for the treatment of non-small cell lung cancer. 17. Group C Drugs are not eligible for cost-sharing. 18. Heparin therapy in the treatment of pregnant patients who have systemic lupus erythematosus (SLE) or who have lupus anticoagulant (LA) is not covered. 19. High dose calcitriol and/or interferon gamma in the treatment of malignant osteopetrosis is not covered. 20. High dose chemotherapy (HDC), see Chapter 3, Section Human chorionic gonadotropin (HCG), or any other drug, administered for purposes of weight control is not covered. 22. Hycamtin (Topotecan ) is covered for treatment of glioblastoma multiforme (brain tumors), effective February 1, Interferon alfa-2a (Roferon -A) is covered for the treatment of metastatic renal cell carcinoma, effective March 1, Interferon alfa-2b (Intron A) is covered for the treatment of: a. Chronic myelogenous leukemia (CML), effective March 1, b. Children with malignant melanoma when prescribed as adjuvant to surgical treatment, effective December 5, Interferon alfa-2b for treatment of bladder cancer is not covered. 26. Interferon Gamma for the treatment of scleroderma is not covered. 3
4 CHAPTER 7, SECTION 7.1 TRICARE/CHAMPUS POLICY MANUAL M DEC Intravenous injections of Gamma Globulin for pregnancy rejection and polycystic ovarian disease is not covered. 28. Intravenous Immune Globulin (IVIG) (Venoglobulin ): a. Is covered when used with aspirin to decrease coronary artery aneurysm formation and other manifestations of Kawasaki syndrome, b. Is not covered in the treatment of multiple sclerosis. 29. Irinotecan (Camptosar ) may be covered for the treatment of glioblastoma multiforme, effective June 14, Isotretinoin (Accutane ) is covered when used in combination with other drugs that are FDA-approved for chemotherapy to treat squamous cell carcinoma of the skin. It is not covered as a single-agent treatment nor for maintenance of remission of squamous cell carcinoma of the skin. 31. Laetrile (amygdalin, sarcarcinase, vitamin B-17) and all other drugs characterized as a "nitriloside," are not recognized to be safe and effective for any therapeutic use and are not covered. 32. Leuprolide acetate (Lupron ) for uterine leiomyomata is covered when any of the following conditions are present in women of reproductive age: infertility; nearing menopause; patients scheduled for myomectomy, or who might be candidates for hysterectomy, upon medical determination that the total uterine volume could be sufficiently reduced with use of the drug, effective December NOTE: No medication has been shown to safely cause complete regression or disappearance of leiomyoma uteri; however, studies have shown that approximately 50% of patients experience a decrease in total uterine volume by about 50% following treatment with leuprolide acetate. Response to treatment is usually achieved by the 12th week of treatment. Since use of leuprolide acetate beyond six months increases the risk of hypogonadism, and leads to osteoporosis (thus appearing to outweigh the beneficial effects), in no event will reimbursement extend beyond six months for treatment with leuprolide acetate. 33. Methotrexate for systemic lupus erythematosus is not covered. 34. Methotrexate for unruptured ectopic pregnancies is covered, effective December 1, Methylprednisolone acetate suspension (Depo-Medrol ) is covered for the treatment of multiple sclerosis. 36. Mitomycin-C (Mutamycin ) is not covered when used in trabeculectomy (glaucoma filtering surgery). 37. Mitoxantrone (Novantrone ) is covered when used in the treatment of intermediate and high grade non-hodgkin's lymphomas. 4
5 TRICARE/CHAMPUS POLICY MANUAL M DEC 1998 CHAPTER 7, SECTION Myocophenolate Mofetil (Cellcept ) for the prophylaxis of organ rejection in patients receiving heart transplantation or liver transplantation is not covered. 39. Paclitaxel (Taxol ) is covered for the following treatments: a. Metastatic ovarian cancer after failure of first-line or subsequent chemotherapy, effective December 29, b. Non-small cell lung cancer as primary treatment choice when the tumor is inoperable or as adjuvant treatment in patients following surgical removal of a lung to impede regional or distant metastases, effective March 3, c. Breast cancer in patients who have not responded to first-line metastatic chemotherapy or who have relapsed after chemotherapy, effective April 21, Paclitaxel (Taxol ) for the treatment of malignant melanoma is not covered. 41. Pamidronate (Aredia ) for the treatment of osteoporosis is not covered. 42. Potassium preparations ordered by a physician and obtained by prescription are covered when the medical necessity is documented and verified by medical review. 43. Tacrolimus (Prograf ), formerly known as FK506, is covered for prophylaxis of organ rejection in patients receiving allogeneic liver transplants, lung transplants, heart transplants, and kidney transplants, effective April 8, Terbutaline Sulfate to induce and maintain tocolysis for the management of preterm labor is covered in the inpatient setting, see Chapter 3, Section Transdermal nicotine patch for the treatment of ulcerative colitis is not covered. 46. Treatment Investigational New Drugs (INDs) are not approved by the FDA for commercial marketing and therefore are not eligible for coverage. Medical care related to the use of Treatment INDs may be covered when the care is provided in accordance with nationally accepted standards of practice in the medical community. 47. Vinorelbine Tartrate (Navelbine ) is covered for the treatment of metastatic breast cancer, effective April 1, B. The above list is not all-inclusive and will be updated as required. III. EXCLUSIONS Placebo injections and drugs are not covered under any circumstances. 5
6 CHAPTER 7, SECTION 7.1 TRICARE/CHAMPUS POLICY MANUAL M DEC 1998 IV. EFFECTIVE DATE Cost-sharing of drugs is as follows: A. Labeled uses. The effective date of FDA approval for the specific labeled indication/ use. B. Off-labeled uses. The effective date of the FDA approval for the latest specific labeled indication/use; or if medical literature supports prior safety and efficacy, the first effective date of FDA approval of the drug for general use in humans may be used. C. Orphan drugs. The effective date of FDA's marketing approval (MA) for the proposed use. Orphan drugs are defined as a drug or biological product which are used for the diagnosis, treatment or prevention of a rare disease or condition. A rare disease is one which affects fewer than one in 200,000 persons in the U.S. - END - 6
CHAPTER 7 SECTION 7.1 DRUGS AND MEDICINES TRICARE/CHAMPUS POLICY MANUAL M DEC 1998 OTHER SERVICES
 TRICARE/CHAMPUS POLICY MANUAL 6010.47-M DEC 1998 OTHER SERVICES CHAPTER 7 SECTION 7.1 Issue Date: December 29, 1982 Authority: 32 CFR 199.4(b)(2)(v), (b)(3)(iii), (b)(5)(v), (d)(3)(vi), and (e)(11)(i)
TRICARE/CHAMPUS POLICY MANUAL 6010.47-M DEC 1998 OTHER SERVICES CHAPTER 7 SECTION 7.1 Issue Date: December 29, 1982 Authority: 32 CFR 199.4(b)(2)(v), (b)(3)(iii), (b)(5)(v), (d)(3)(vi), and (e)(11)(i)
HMO: Medical (provider setting); Rx (out patient) PPO/CDHP: Rx
 BENEFIT DESCRIPTION AND LIMITATIONS OF COVERAGE ITEM: PRODUCT LINES: COVERED UNDER: DESCRIPTION: CPT/HCPCS Code: Company Supplying: Setting: Epogen, Procrit (epoetin alfa, injection) Commercial HMO/PPO/CDHP
BENEFIT DESCRIPTION AND LIMITATIONS OF COVERAGE ITEM: PRODUCT LINES: COVERED UNDER: DESCRIPTION: CPT/HCPCS Code: Company Supplying: Setting: Epogen, Procrit (epoetin alfa, injection) Commercial HMO/PPO/CDHP
Acthar Gel. H. P. Acthar Gel (corticotropin; ACTH) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.30.10 Subject: Acthar Gel Page: 1 of 7 Last Review Date: November 30, 2018 Acthar Gel Description H.
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.30.10 Subject: Acthar Gel Page: 1 of 7 Last Review Date: November 30, 2018 Acthar Gel Description H.
Medication Prior Authorization Form
 Policy Number: 1047 Eligard (leuprolide acetate) 7.5mg Eligard (leuprolide acetate) 22.5mg Eligard (leuprolide acetate) 30mg Eligard (leuprolide acetate)45mg Firmagon (degarelix) Lupaneta Pack (leuprolide
Policy Number: 1047 Eligard (leuprolide acetate) 7.5mg Eligard (leuprolide acetate) 22.5mg Eligard (leuprolide acetate) 30mg Eligard (leuprolide acetate)45mg Firmagon (degarelix) Lupaneta Pack (leuprolide
VI.2 Elements for a Public Summary VI.2.1 Overview of Disease Epidemiology Acute Nausea and Vomiting (N&V) Etiologies:
 VI.2 Elements for a Public Summary VI.2.1 Overview of Disease Epidemiology Acute Nausea and Vomiting (N&V) Incidence: The incidence of acute and delayed N&V was investigated in highly and moderately emetogenic
VI.2 Elements for a Public Summary VI.2.1 Overview of Disease Epidemiology Acute Nausea and Vomiting (N&V) Incidence: The incidence of acute and delayed N&V was investigated in highly and moderately emetogenic
CHAPTER 3 SECTION 1.6G SIMULTANEOUS PANCREAS-KIDNEY, PANCREAS-AFTER-KIDNEY, AND PANCREAS-TRANSPLANT-ALONE
 TRICARE POLICY MANUAL 6010.47-M, MARCH 15, 2002 SURGERY AND RELATED SERVICES CHAPTER 3 SECTION 1.6G SIMULTANEOUS PANCREAS-KIDNEY, PANCREAS-AFTER-KIDNEY, AND PANCREAS-TRANSPLANT-ALONE ISSUE DATE: February
TRICARE POLICY MANUAL 6010.47-M, MARCH 15, 2002 SURGERY AND RELATED SERVICES CHAPTER 3 SECTION 1.6G SIMULTANEOUS PANCREAS-KIDNEY, PANCREAS-AFTER-KIDNEY, AND PANCREAS-TRANSPLANT-ALONE ISSUE DATE: February
Chapter 4 Section Simultaneous Pancreas-Kidney (SPK), Pancreas-After-Kidney (PAK), And Pancreas-Transplant-Alone (PTA)
 Surgery Chapter 4 Section 24.7 Simultaneous Pancreas-Kidney (SPK), Pancreas-After-Kidney (PAK), And Pancreas-Transplant-Alone Issue Date: February 5, 1996 Authority: 32 CFR 199.4(e)(5) 1.0 CPT 1 PROCEDURE
Surgery Chapter 4 Section 24.7 Simultaneous Pancreas-Kidney (SPK), Pancreas-After-Kidney (PAK), And Pancreas-Transplant-Alone Issue Date: February 5, 1996 Authority: 32 CFR 199.4(e)(5) 1.0 CPT 1 PROCEDURE
Chapter 4 Section 24.7
 Surgery Chapter 4 Section 24.7 Simultaneous Pancreas-Kidney (SPK), Pancreas-After-Kidney (PAK), And Pancreas-Transplant-Alone (PTA), And Pancreatic Islet Cell Transplantation Issue Date: February 5, 1996
Surgery Chapter 4 Section 24.7 Simultaneous Pancreas-Kidney (SPK), Pancreas-After-Kidney (PAK), And Pancreas-Transplant-Alone (PTA), And Pancreatic Islet Cell Transplantation Issue Date: February 5, 1996
Summary of Strategic Competitive Analysis and Publication Planning
 Summary of Strategic Competitive Analysis and Publication Planning Carole Alison Chrvala, PhD 919.545.2149 (Work) 919.951.5230 (Mobile) cchrvala@centurylink.net www.healthmattersmedwriting.com 1 Briefing
Summary of Strategic Competitive Analysis and Publication Planning Carole Alison Chrvala, PhD 919.545.2149 (Work) 919.951.5230 (Mobile) cchrvala@centurylink.net www.healthmattersmedwriting.com 1 Briefing
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Lynparza) Reference Number: CP.PHAR.360 Effective Date: 10.03.17 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this
Clinical Policy: (Lynparza) Reference Number: CP.PHAR.360 Effective Date: 10.03.17 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this
Form 2023 R2.0: Ovarian Cancer Pre-HSCT Data
 Key Fields Sequence Number Date Received: - - CIBMTR Center Number: CIBMTR Recipient ID: Today's Date: - - Date of HSCT for which this form is being completed: - - HSCT type: (check all that apply) Autologous
Key Fields Sequence Number Date Received: - - CIBMTR Center Number: CIBMTR Recipient ID: Today's Date: - - Date of HSCT for which this form is being completed: - - HSCT type: (check all that apply) Autologous
Intron A. Intron A (interferon alfa-2b) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Intron A Page: 1 of 6 Last Review Date: June 19, 2015 Intron A Description Intron A (interferon
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Intron A Page: 1 of 6 Last Review Date: June 19, 2015 Intron A Description Intron A (interferon
Annual General Meeting Roche Holding Ltd 5 March Art Levinson Chairman and CEO Genentech
 Annual General Meeting Roche Holding Ltd 5 March 2007 Art Levinson Chairman and CEO Genentech Agenda Company Overview/Approach Accomplishments & Performance Research & Development Roche Partnership Patient
Annual General Meeting Roche Holding Ltd 5 March 2007 Art Levinson Chairman and CEO Genentech Agenda Company Overview/Approach Accomplishments & Performance Research & Development Roche Partnership Patient
Multiple Sclerosis , The Patient Education Institute, Inc. nr Last reviewed: 04/17/2017 1
 Multiple Sclerosis Introduction Multiple sclerosis, or MS, is a disease of the brain and spinal cord. Most patients with multiple sclerosis are able to lead a very functional life. However, MS causes a
Multiple Sclerosis Introduction Multiple sclerosis, or MS, is a disease of the brain and spinal cord. Most patients with multiple sclerosis are able to lead a very functional life. However, MS causes a
Evolve180 / Ideal Northwest Health Profile
 Evolve180 / Ideal Northwest Health Profile ABOUT YOU First Name: Last Name: Address: City: State: Zip: Phone: Email: Date of Birth: Age: Height: Occupation: How did you find out about our program? Marital
Evolve180 / Ideal Northwest Health Profile ABOUT YOU First Name: Last Name: Address: City: State: Zip: Phone: Email: Date of Birth: Age: Height: Occupation: How did you find out about our program? Marital
Directorate General of Health Services Office of Drugs Controller General (India) (Biological Division)
 Directorate General of Health Services Office of Drugs Controller General (India) (Biological Division) List of r-dna drug products approved in the country (Form 45 and 45A) from 1 st Jan 2015 to 30 th
Directorate General of Health Services Office of Drugs Controller General (India) (Biological Division) List of r-dna drug products approved in the country (Form 45 and 45A) from 1 st Jan 2015 to 30 th
CHRONIC MEDICINE PROGRAMME: GENERAL INFORMATION LETTER
 CHRONIC MEDICINE PROGRAMME: GENERAL INFORMATION LETTER The Prescribed Minimum Benefit Chronic Disease List In terms of the Medical Scheme Act Regulations that came into effect on 1 January 2004, Medical
CHRONIC MEDICINE PROGRAMME: GENERAL INFORMATION LETTER The Prescribed Minimum Benefit Chronic Disease List In terms of the Medical Scheme Act Regulations that came into effect on 1 January 2004, Medical
TRANSPARENCY COMMITTEE OPINION. 29 April 2009
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 29 April 2009 NAVELBINE 20 mg, soft capsules B/1 (CIP: 365 948-4) NAVELBINE 30 mg, soft capsules B/1 (CIP: 365 949-0)
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 29 April 2009 NAVELBINE 20 mg, soft capsules B/1 (CIP: 365 948-4) NAVELBINE 30 mg, soft capsules B/1 (CIP: 365 949-0)
Avastin Sample Coding
 First- and Second-line Metastatic Colorectal Cancer C18.0 Malignant neoplasm of the cecum C18.1 Malignant neoplasm of appendix C18.2-C18.9 C19 C20 C21.8 Malignant neoplasm of the colon, various sites Malignant
First- and Second-line Metastatic Colorectal Cancer C18.0 Malignant neoplasm of the cecum C18.1 Malignant neoplasm of appendix C18.2-C18.9 C19 C20 C21.8 Malignant neoplasm of the colon, various sites Malignant
WARNING LETTER. The Indications and Usage section of the approved product labeling (PI) for Prograf states:
 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration Rockville, MD 20857 TRANSMITTED BY FACSIMILE Hideo Fukumoto CEO Three Parkway North Deerfield, IL 60015-2548 RE:
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration Rockville, MD 20857 TRANSMITTED BY FACSIMILE Hideo Fukumoto CEO Three Parkway North Deerfield, IL 60015-2548 RE:
Name of Policy: Boniva (Ibandronate Sodium) Infusion
 Name of Policy: Boniva (Ibandronate Sodium) Infusion Policy #: 266 Latest Review Date: April 2010 Category: Pharmacology Policy Grade: Active Policy but no longer scheduled for regular literature reviews
Name of Policy: Boniva (Ibandronate Sodium) Infusion Policy #: 266 Latest Review Date: April 2010 Category: Pharmacology Policy Grade: Active Policy but no longer scheduled for regular literature reviews
CHRONIC TREATMENT GUIDELINES
 CHRONIC TREATMENT GUIDELINES REGISTRATION OF CHRONIC CONDITIONS You can only access benefits for chronic medication, as listed below, if your prescribing/treating doctor or pharmacist registers your chronic
CHRONIC TREATMENT GUIDELINES REGISTRATION OF CHRONIC CONDITIONS You can only access benefits for chronic medication, as listed below, if your prescribing/treating doctor or pharmacist registers your chronic
Multiple Sclerosis. Brain Spinal Cord. Peripheral Nerves
 Multiple Sclerosis Introduction Multiple sclerosis, or MS, is a disease of the brain and spinal cord. It affects about 400,000 Americans at any one time. Most patients with multiple sclerosis are able
Multiple Sclerosis Introduction Multiple sclerosis, or MS, is a disease of the brain and spinal cord. It affects about 400,000 Americans at any one time. Most patients with multiple sclerosis are able
CHAPTER 3 SECTION 1.6E COMBINED LIVER-KIDNEY TRANSPLANTATION. TRICARE/CHAMPUS POLICY MANUAL M DEC 1998 Surgery And Related Services
 TRICARE/CHAMPUS POLICY MANUAL 6010.47-M DEC 1998 Surgery And Related Services CHAPTER 3 SECTION 1.6E Issue Date: October 26, 1994 Authority: 32 CFR 199.4(e)(5) I. PROCEDURE CODE RANGE 47150 II. POLICY
TRICARE/CHAMPUS POLICY MANUAL 6010.47-M DEC 1998 Surgery And Related Services CHAPTER 3 SECTION 1.6E Issue Date: October 26, 1994 Authority: 32 CFR 199.4(e)(5) I. PROCEDURE CODE RANGE 47150 II. POLICY
Committee Approval Date: May 9, 2014 Next Review Date: May 2015
 Medication Policy Manual Policy No: dru248 Topic: Benlysta, belimumab Date of Origin: May 13, 2011 Committee Approval Date: May 9, 2014 Next Review Date: May 2015 Effective Date: June 1, 2014 IMPORTANT
Medication Policy Manual Policy No: dru248 Topic: Benlysta, belimumab Date of Origin: May 13, 2011 Committee Approval Date: May 9, 2014 Next Review Date: May 2015 Effective Date: June 1, 2014 IMPORTANT
Do you or did you have any family members who have been diagnosed with MS? Yes No I Don t Know. Yes identical Yes not identical No
 The following survey will ask you questions about your family history of MS, your MS diagnosis and history, your symptoms, and your treatments. You may find it easier to fully answer these questions if
The following survey will ask you questions about your family history of MS, your MS diagnosis and history, your symptoms, and your treatments. You may find it easier to fully answer these questions if
Chapter 4 Section Combined Heart-Kidney Transplantation (CHKT)
 Surgery Chapter 4 Section 24.3 Issue Date: May 7, 1999 Authority: 32 CFR 199.4(e)(5) 1.0 POLICY 1.1 is a TRICARE benefit that requires preauthorization. 1.1.1 A TRICARE Prime enrollee must have a referral
Surgery Chapter 4 Section 24.3 Issue Date: May 7, 1999 Authority: 32 CFR 199.4(e)(5) 1.0 POLICY 1.1 is a TRICARE benefit that requires preauthorization. 1.1.1 A TRICARE Prime enrollee must have a referral
Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document.
 ERYTHROPOIESIS-STIMULATING AGENTS (ESAs) Epoetin alfa (Epogen, Procrit ) Darbepoetin alfa (Aranesp ) Methoxy polyethylene glycol (PEG) epoetin-beta (Mircera ) Non-Discrimination Statement and Multi-Language
ERYTHROPOIESIS-STIMULATING AGENTS (ESAs) Epoetin alfa (Epogen, Procrit ) Darbepoetin alfa (Aranesp ) Methoxy polyethylene glycol (PEG) epoetin-beta (Mircera ) Non-Discrimination Statement and Multi-Language
CHRONIC MEDICINE PROGRAMME: PICK N PAY PLUS OPTION - GENERAL INFORMATION LETTER
 CHRONIC MEDICINE PROGRAMME: PICK N PAY PLUS OPTION - GENERAL INFORMATION LETTER Prescribed Minimum Benefits The prescribed minimum benefits (PMBs) comprise a list of 270 conditions or group of conditions
CHRONIC MEDICINE PROGRAMME: PICK N PAY PLUS OPTION - GENERAL INFORMATION LETTER Prescribed Minimum Benefits The prescribed minimum benefits (PMBs) comprise a list of 270 conditions or group of conditions
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 H.P. Acthar Gel (repository corticotropin) Page 1 of 7 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: H.P. Acthar Gel (repository corticotropin) Prime Therapeutics
H.P. Acthar Gel (repository corticotropin) Page 1 of 7 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: H.P. Acthar Gel (repository corticotropin) Prime Therapeutics
Integumentary system pertains to the skin, subcutaneous tissue and areolar tissue.
 TRICARE/CHAMPUS POLICY MANUAL 6010.47-M DEC 1998 Surgery And Related Services CHAPTER 3 SECTION 2.1 Issue Date: August 26, 1985 Authority: 32 CFR 199.4(c)(2) and (c)(3) I. PROCEDURE CODE RANGE 10040-19499
TRICARE/CHAMPUS POLICY MANUAL 6010.47-M DEC 1998 Surgery And Related Services CHAPTER 3 SECTION 2.1 Issue Date: August 26, 1985 Authority: 32 CFR 199.4(c)(2) and (c)(3) I. PROCEDURE CODE RANGE 10040-19499
trial update clinical
 trial update clinical by John W. Mucenski, BS, PharmD, Director of Pharmacy Operations, UPMC Cancer Centers The treatment outcome for patients with relapsed or refractory cervical carcinoma remains dismal.
trial update clinical by John W. Mucenski, BS, PharmD, Director of Pharmacy Operations, UPMC Cancer Centers The treatment outcome for patients with relapsed or refractory cervical carcinoma remains dismal.
Magellan Rx. A smarter approach to pharmacy benefits management
 Magellan Rx A smarter approach to pharmacy benefits management Presented by: Cheri Caruso, VP of Sales, Magellan Rx Management Bryce Canfield, VP, Client Development, GoodRx A unique vision of care We
Magellan Rx A smarter approach to pharmacy benefits management Presented by: Cheri Caruso, VP of Sales, Magellan Rx Management Bryce Canfield, VP, Client Development, GoodRx A unique vision of care We
KYMRIAH (tisagenlecleucel)
 KYMRIAH (tisagenlecleucel) Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and
KYMRIAH (tisagenlecleucel) Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and
Medication Review. Cancer Chemotherapy Drugs. Pharmacy Technician Training Systems Passassured, LLC
 Medication Review Cancer Chemotherapy Drugs Pharmacy Technician Training Systems Passassured, LLC Medication Review, Cancer Chemotherapy Drugs PassAssured's Pharmacy Technician Training Program Medication
Medication Review Cancer Chemotherapy Drugs Pharmacy Technician Training Systems Passassured, LLC Medication Review, Cancer Chemotherapy Drugs PassAssured's Pharmacy Technician Training Program Medication
Drugs That Require Prior Authorization (PA) Before Being Approved for Coverage
 Drugs That Require Prior Authorization (PA) Before Being Approved for Coverage You will need authorization by your UA Medicare Part D Prescription Drug Plan before filling prescriptions for the drugs shown
Drugs That Require Prior Authorization (PA) Before Being Approved for Coverage You will need authorization by your UA Medicare Part D Prescription Drug Plan before filling prescriptions for the drugs shown
Acthar Gel. H. P. Acthar Gel (corticotropin; ACTH) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.30.10 Subject: Acthar Gel Page: 1 of 6 Last Review Date: December 8, 2017 Acthar Gel Description H. P.
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.30.10 Subject: Acthar Gel Page: 1 of 6 Last Review Date: December 8, 2017 Acthar Gel Description H. P.
Intron A. Intron A (interferon alfa-2b) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.21.07 Subject: Intron A Page: 1 of 6 Last Review Date: June 22, 2018 Intron A Description Intron A (interferon
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.21.07 Subject: Intron A Page: 1 of 6 Last Review Date: June 22, 2018 Intron A Description Intron A (interferon
WITBANK COALFIELDS MEDICAL AID SCHEME (WCMAS) CHRONIC MEDICINE PROGRAMME GENERAL INFORMATION LETTER
 WITBANK COALFIELDS MEDICAL AID SCHEME (WCMAS) CHRONIC MEDICINE PROGRAMME GENERAL INFORMATION LETTER The Prescribed Minimum Benefit Chronic Disease List In terms of the Medical Scheme Act Regulations that
WITBANK COALFIELDS MEDICAL AID SCHEME (WCMAS) CHRONIC MEDICINE PROGRAMME GENERAL INFORMATION LETTER The Prescribed Minimum Benefit Chronic Disease List In terms of the Medical Scheme Act Regulations that
Update from the 25th Annual San Antonio Breast Cancer Symposium December 11 14, 2002
 Update from the 25th Annual San Antonio Breast Cancer Symposium December 11 14, 2002 Nearly 5,000 physicians, oncologists, radiologists, epidemiologists, basic scientists, and breast cancer advocates from
Update from the 25th Annual San Antonio Breast Cancer Symposium December 11 14, 2002 Nearly 5,000 physicians, oncologists, radiologists, epidemiologists, basic scientists, and breast cancer advocates from
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Abraxane) Reference Number: CP.PHAR.176 Effective Date: 07.01.15 Last Review Date: 05.18 Line of Business: HIM, Medicaid Coding Implications Revision Log See Important Reminder at the
Clinical Policy: (Abraxane) Reference Number: CP.PHAR.176 Effective Date: 07.01.15 Last Review Date: 05.18 Line of Business: HIM, Medicaid Coding Implications Revision Log See Important Reminder at the
Acthar Gel. H. P. Acthar Gel (corticotropin; ACTH) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Acthar Gel Page: 1 of 5 Last Review Date: September 18, 2015 Acthar Gel Description H. P. Acthar
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Acthar Gel Page: 1 of 5 Last Review Date: September 18, 2015 Acthar Gel Description H. P. Acthar
9/25/2013 SYSTEMIC LUPUS ERYTHEMATOSUS (SLE)
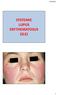 SYSTEMIC LUPUS ERYTHEMATOSUS (SLE) 1 Other Types of Lupus Discoid Lupus Erythematosus Lupus Pernio --- Sarcoidosis Lupus Vulgaris --- Tuberculosis of the face Manifestations of SLE Fever Rashes Arthritis
SYSTEMIC LUPUS ERYTHEMATOSUS (SLE) 1 Other Types of Lupus Discoid Lupus Erythematosus Lupus Pernio --- Sarcoidosis Lupus Vulgaris --- Tuberculosis of the face Manifestations of SLE Fever Rashes Arthritis
Acthar Gel. H. P. Acthar Gel (corticotropin; ACTH) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.30.10 Subject: Acthar Gel Page: 1 of 5 Last Review Date: December 2, 2016 Acthar Gel Description H. P.
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.30.10 Subject: Acthar Gel Page: 1 of 5 Last Review Date: December 2, 2016 Acthar Gel Description H. P.
Understanding Myositis Medications
 Understanding Myositis Medications 2015 TMA Annual Patient Conference Orlando, Florida Chester V. Oddis, MD University of Pittsburgh Director, Myositis Center Disclosures Mallinckrodt: Research Grant Genentech:
Understanding Myositis Medications 2015 TMA Annual Patient Conference Orlando, Florida Chester V. Oddis, MD University of Pittsburgh Director, Myositis Center Disclosures Mallinckrodt: Research Grant Genentech:
Coverage Period: 01/01/ /31/2018 Coverage for: Individual and/or Family Plan: Healthy Rewards HSA
 This is only a summary of your GatorCare pharmacy benefits. If you would like detail about your coverage and costs, you can get the complete terms in the policy or plan document at gatorcare.magellanrx.com/member
This is only a summary of your GatorCare pharmacy benefits. If you would like detail about your coverage and costs, you can get the complete terms in the policy or plan document at gatorcare.magellanrx.com/member
H.P. ACTHAR GEL (repository corticotropin injection)
 H.P. ACTHAR GEL (repository corticotropin injection) Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures,
H.P. ACTHAR GEL (repository corticotropin injection) Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures,
Stelara. Stelara (ustekinumab) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.90.04 Subject: Stelara Page: 1 of 9 Last Review Date: September 20, 2018 Stelara Description Stelara
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.90.04 Subject: Stelara Page: 1 of 9 Last Review Date: September 20, 2018 Stelara Description Stelara
CLINICAL MEDICAL POLICY
 CLINICAL MEDICAL POLICY Policy Name: Avastin (bevacizumab) Policy Number: MP-030-MD-DE Responsible Department(s): Medical Management; Clinical Pharmacy Provider Notice Date: 10/01/2017 Original Effective
CLINICAL MEDICAL POLICY Policy Name: Avastin (bevacizumab) Policy Number: MP-030-MD-DE Responsible Department(s): Medical Management; Clinical Pharmacy Provider Notice Date: 10/01/2017 Original Effective
Chapter 4 Section 24.1
 Surgery Chapter 4 Section 24.1 Issue Date: October 27, 1995 Authority: 32 CFR 199.4(e)(5) 1.0 CPT 1 PROCEDURE CODES 32850-32854, 33930-33935 2.0 DIAGNOSTIC RELATED GROUPS (DRGs) 495 for lung transplant.
Surgery Chapter 4 Section 24.1 Issue Date: October 27, 1995 Authority: 32 CFR 199.4(e)(5) 1.0 CPT 1 PROCEDURE CODES 32850-32854, 33930-33935 2.0 DIAGNOSTIC RELATED GROUPS (DRGs) 495 for lung transplant.
CHAPTER 3 SECTION 1.6B HEART-LUNG AND LUNG TRANSPLANTATION TRICARE POLICY MANUAL M, MARCH 15, 2002 SURGERY AND RELATED SERVICES
 TRICARE POLICY MANUAL 6010.47-M, MARCH 15, 2002 SURGERY AND RELATED SERVICES CHAPTER 3 SECTION 1.6B ISSUE DATE: October 27, 1995 AUTHORITY: 32 CFR 199.4(e)(5) I. CODES A. CPT 1 Procedure Codes 33930, 33935,
TRICARE POLICY MANUAL 6010.47-M, MARCH 15, 2002 SURGERY AND RELATED SERVICES CHAPTER 3 SECTION 1.6B ISSUE DATE: October 27, 1995 AUTHORITY: 32 CFR 199.4(e)(5) I. CODES A. CPT 1 Procedure Codes 33930, 33935,
Epogen / Procrit. Epogen / Procrit (epoetin alfa) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.10.06 Section: Prescription Drugs Effective Date: April1, 2014 Subject: Epogen / Procrit Page: 1 of 7
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.10.06 Section: Prescription Drugs Effective Date: April1, 2014 Subject: Epogen / Procrit Page: 1 of 7
List of Qualifying Conditions
 List of Qualifying Conditions Cancer Conditions 1) Adrenal cancer 2) Bladder cancer 3) Bone cancer all forms 4) Brain cancer 5) Breast cancer 6) Cervical cancer 7) Colon cancer 8) Colorectal cancer 9)
List of Qualifying Conditions Cancer Conditions 1) Adrenal cancer 2) Bladder cancer 3) Bone cancer all forms 4) Brain cancer 5) Breast cancer 6) Cervical cancer 7) Colon cancer 8) Colorectal cancer 9)
2011 Physician Quality Reporting System Measures for Consideration by Oncology Providers: Cancer Care Measures
 2011 Physician Quality Reporting System Measures for Consideration by Oncology Providers: Cancer Care Measures The table below includes measures directly relevant to oncology providers as well as general
2011 Physician Quality Reporting System Measures for Consideration by Oncology Providers: Cancer Care Measures The table below includes measures directly relevant to oncology providers as well as general
Corporate Medical Policy
 Corporate Medical Policy Hematopoietic Stem-Cell Transplantation for Autoimmune Diseases File Name: Origination: Last CAP Review: Next CAP Review: Last Review: hematopoietic_stem-cell_transplantation_for_autoimmune_diseases
Corporate Medical Policy Hematopoietic Stem-Cell Transplantation for Autoimmune Diseases File Name: Origination: Last CAP Review: Next CAP Review: Last Review: hematopoietic_stem-cell_transplantation_for_autoimmune_diseases
Opinion 23 July 2014
 The legally binding text is the original French version TRANSPARENCY COMMITTEE Opinion 23 July 2014 IMUREL 50 mg, film-coated tablet (B/100) (CIP: 34009 364 149 0 7) IMUREL 25 mg, film-coated tablet (B/50)
The legally binding text is the original French version TRANSPARENCY COMMITTEE Opinion 23 July 2014 IMUREL 50 mg, film-coated tablet (B/100) (CIP: 34009 364 149 0 7) IMUREL 25 mg, film-coated tablet (B/50)
Arthritis & Rheumatology Clinics of Kansas PATIENT EDUCATION SYSTEMIC LUPUS ERYTHEMATOSUS
 Arthritis & Rheumatology Clinics of Kansas PATIENT EDUCATION SYSTEMIC LUPUS ERYTHEMATOSUS Introduction: There is perhaps no rheumatic disease that evokes so much fear and confusion among both patients
Arthritis & Rheumatology Clinics of Kansas PATIENT EDUCATION SYSTEMIC LUPUS ERYTHEMATOSUS Introduction: There is perhaps no rheumatic disease that evokes so much fear and confusion among both patients
NCIC CLINICAL TRIALS GROUP DATA SAFETY MONITORING COMMITTEE Fall Conference Call 23 November 2009 SUMMARY REPORT
 NCIC CLINICAL TRIALS GROUP DATA SAFETY MONITORING COMMITTEE Fall Conference Call 23 November 2009 SUMMARY REPORT The NCIC CTG DSMC reviewed the following trials with respect to safety, trial conduct, including
NCIC CLINICAL TRIALS GROUP DATA SAFETY MONITORING COMMITTEE Fall Conference Call 23 November 2009 SUMMARY REPORT The NCIC CTG DSMC reviewed the following trials with respect to safety, trial conduct, including
Lupus and Your Kidneys. Michael P. Madaio, MD Professor of Medicine Chairman of Medicine Medical College of Georgia Augusta, Georgia
 Lupus and Your Kidneys Michael P. Madaio, MD Professor of Medicine Chairman of Medicine Medical College of Georgia Augusta, Georgia Kidney Inflammation and Abnormal Function as a Result of Lupus (Lupus
Lupus and Your Kidneys Michael P. Madaio, MD Professor of Medicine Chairman of Medicine Medical College of Georgia Augusta, Georgia Kidney Inflammation and Abnormal Function as a Result of Lupus (Lupus
Clinical Policy: Goserelin Acetate (Zoladex) Reference Number: CP.PHAR.171 Effective Date: Last Review Date: Line of Business: Medicaid
 Clinical Policy: (Zoladex) Reference Number: CP.PHAR.171 Effective Date: 10.01.16 Last Review Date: 11.17 Line of Business: Medicaid Coding Implications Revision Log See Important Reminder at the end of
Clinical Policy: (Zoladex) Reference Number: CP.PHAR.171 Effective Date: 10.01.16 Last Review Date: 11.17 Line of Business: Medicaid Coding Implications Revision Log See Important Reminder at the end of
SCHEDULE OF CONTINUING EDUCATION COURSES FOR RN s and CNAs. January 2018 WALK INS ARE ACCEPTED BUT WE ADVISE STUDENTS TO PRE-REGISTER BEFORE THURSDAY
 SCHEDULE OF CONTINUING EDUCATION COURSES FOR RN s and CNAs January 201 DATE DAY TIME TOPICS January 04 January 11 January 1 January 25 9:00AM 9:00AM 9:00AM 9:00AM 1. Understanding Fibromyalagia 2. Diabetes
SCHEDULE OF CONTINUING EDUCATION COURSES FOR RN s and CNAs January 201 DATE DAY TIME TOPICS January 04 January 11 January 1 January 25 9:00AM 9:00AM 9:00AM 9:00AM 1. Understanding Fibromyalagia 2. Diabetes
Opdivo. Opdivo (nivolumab) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.21.53 Subsection: Antineoplastic Agents Original Policy Date: January 16, 2015 Subject: Opdivo Page:
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.21.53 Subsection: Antineoplastic Agents Original Policy Date: January 16, 2015 Subject: Opdivo Page:
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Verzenio) Reference Number: CP.PHAR.355 Effective Date: 10.24.17 Last Review Date: 02.19 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this
Clinical Policy: (Verzenio) Reference Number: CP.PHAR.355 Effective Date: 10.24.17 Last Review Date: 02.19 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this
SCHEDULE OF CONTINUING EDUCATION COURSES FOR RN s and CNAs. January 2018
 January 201 DATE DAY TIME TOPICS TOTAL January 04 1. Understanding Fibromyalagia 2. Diabetes and Cardiovascular Disease 3. Prostate Cancer 4. Hepatitis C 5. Understanding Hepatitis B January 11 1. Dysphagia
January 201 DATE DAY TIME TOPICS TOTAL January 04 1. Understanding Fibromyalagia 2. Diabetes and Cardiovascular Disease 3. Prostate Cancer 4. Hepatitis C 5. Understanding Hepatitis B January 11 1. Dysphagia
Intron A. Intron A (interferon alfa-2b) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.21.07 Subject: Intron A Page: 1 of 6 Last Review Date: June 24, 2016 Intron A Description Intron A (interferon
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.21.07 Subject: Intron A Page: 1 of 6 Last Review Date: June 24, 2016 Intron A Description Intron A (interferon
SCHEDULE OF CONTINUING EDUCATION COURSES FOR RN s and CNAs. January 2019
 January 2019 DATE DAY TIME TOPICS TOTAL January 03 1. Alcoholism 2. Nutrition for the Elderly 3. Uterine Fibroids 4. HIPAA 5.Arthritis 6. Childhood Obesity January 10 1. Understanding Epilepsy: Latest
January 2019 DATE DAY TIME TOPICS TOTAL January 03 1. Alcoholism 2. Nutrition for the Elderly 3. Uterine Fibroids 4. HIPAA 5.Arthritis 6. Childhood Obesity January 10 1. Understanding Epilepsy: Latest
SOURCES: U.S. Department of Labor, Bureau of Labor Statistics; the Federal Reserve Bank of Cleveland; the Commodity Research Bureau; and U.S.
 SOURCES: U.S. Department of Labor, Bureau of Labor Statistics; the Federal Reserve Bank of Cleveland; the Commodity Research Bureau; and U.S. Department of Commerce, Bureau of Economic Analysis. Identified
SOURCES: U.S. Department of Labor, Bureau of Labor Statistics; the Federal Reserve Bank of Cleveland; the Commodity Research Bureau; and U.S. Department of Commerce, Bureau of Economic Analysis. Identified
MEDICAL COVERAGE GUIDELINES ORIGINAL EFFECTIVE DATE: 08/19/14 SECTION: DRUGS LAST REVIEW DATE: LAST CRITERIA REVISION DATE: ARCHIVE DATE:
 RITUXAN (rituximab) Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Medical Coverage Guideline
RITUXAN (rituximab) Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Medical Coverage Guideline
H.P. Acthar Gel (repository corticotropin) Prior Authorization Criteria Program Summary
 OBJECTIVE The intent of the H.P. Acthar Gel (repository corticotropin) Prior Authorization (PA) Criteria is to appropriately select patients for therapy according to product labeling and/or clinical studies
OBJECTIVE The intent of the H.P. Acthar Gel (repository corticotropin) Prior Authorization (PA) Criteria is to appropriately select patients for therapy according to product labeling and/or clinical studies
An Overview of Blood and Marrow Transplantation
 An Overview of Blood and Marrow Transplantation October 24, 2009 Stephen Couban Department of Medicine Dalhousie University Objectives What are the types of blood and marrow transplantation? Who may benefit
An Overview of Blood and Marrow Transplantation October 24, 2009 Stephen Couban Department of Medicine Dalhousie University Objectives What are the types of blood and marrow transplantation? Who may benefit
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Imbruvica) Reference Number: HIM.PA.SP48 Effective Date: 01.01.18 Last Review Date: Line of Business: Health Insurance Marketplace Revision Log See Important Reminder at the end of this
Clinical Policy: (Imbruvica) Reference Number: HIM.PA.SP48 Effective Date: 01.01.18 Last Review Date: Line of Business: Health Insurance Marketplace Revision Log See Important Reminder at the end of this
Coverage Period: Coverage for: Plans: This is only a summary of your GatorCare pharmacy benefits. Coinsurance: you your Dependent Copayment: you
 This is only a summary of your GatorCare pharmacy benefits. If you would like detail about your coverage and costs, you can get the complete terms in the policy or plan document at gatorcare.magellanrx.com/member
This is only a summary of your GatorCare pharmacy benefits. If you would like detail about your coverage and costs, you can get the complete terms in the policy or plan document at gatorcare.magellanrx.com/member
Manipulation of the Immune Response - Immunomodulation -
 Manipulation of the Immune Response - Immunomodulation - Janeway s Immunobiology, 9 th ed., 2017. Ivana Novak Nakir Immune system Immune system make components, cells and organs that act together to defend
Manipulation of the Immune Response - Immunomodulation - Janeway s Immunobiology, 9 th ed., 2017. Ivana Novak Nakir Immune system Immune system make components, cells and organs that act together to defend
Clinical Policy: Pemetrexed (Alimta) Reference Number: CP.PHAR.368 Effective Date: Last Review Date: Line of Business: Medicaid
 Clinical Policy: (Alimta) Reference Number: CP.PHAR.368 Effective Date: 10.31.17 Last Review Date: 02.18 Line of Business: Medicaid Coding Implications Revision Log See Important Reminder at the end of
Clinical Policy: (Alimta) Reference Number: CP.PHAR.368 Effective Date: 10.31.17 Last Review Date: 02.18 Line of Business: Medicaid Coding Implications Revision Log See Important Reminder at the end of
Treatment Options for Breast Cancer in Low- and Middle-Income Countries: Adjuvant and Metastatic Systemic Therapy
 Women s Empowerment Cancer Advocacy Network (WE CAN) Conference Bucharest, Romania October 2015 Treatment Options for Breast Cancer in Low- and Middle-Income Countries: Adjuvant and Metastatic Systemic
Women s Empowerment Cancer Advocacy Network (WE CAN) Conference Bucharest, Romania October 2015 Treatment Options for Breast Cancer in Low- and Middle-Income Countries: Adjuvant and Metastatic Systemic
ASSESSMENT OF THE PAEDIATRIC NEEDS IMMUNOLOGY DISCLAIMER
 European Medicines Agency Evaluation of Medicines for Human Use London, September 2006 Doc. Ref.: EMEA/381922/2006 ASSESSMENT OF THE PAEDIATRIC NEEDS IMMUNOLOGY DISCLAIMER The Paediatric Working Party
European Medicines Agency Evaluation of Medicines for Human Use London, September 2006 Doc. Ref.: EMEA/381922/2006 ASSESSMENT OF THE PAEDIATRIC NEEDS IMMUNOLOGY DISCLAIMER The Paediatric Working Party
MEDICATIONS: THE GOOD, THE BAD, THE UGLY
 MEDICATIONS: THE GOOD, THE BAD, THE UGLY July 13, 2013 Dr. Tanaz Kermani Assistant Clinical Professor of Medicine, Division of Rheumatology, David Geffen School of Medicine UCLA Dr. Robert Spiera Professor
MEDICATIONS: THE GOOD, THE BAD, THE UGLY July 13, 2013 Dr. Tanaz Kermani Assistant Clinical Professor of Medicine, Division of Rheumatology, David Geffen School of Medicine UCLA Dr. Robert Spiera Professor
Lupus. Fast facts. What is lupus? What causes lupus? Who gets lupus?
 Lupus Systemic lupus erythematosus, referred to as SLE or lupus, is sometimes called the "great imitator." Why? Because of its wide range of symptoms, people often confuse lupus with other health problems.
Lupus Systemic lupus erythematosus, referred to as SLE or lupus, is sometimes called the "great imitator." Why? Because of its wide range of symptoms, people often confuse lupus with other health problems.
CHRONIC MEDICINE PROGRAMME GENERAL INFORMATION LETTER
 RAND WATER MEDICAL SCHEME RAND WATER MEDICAL SCHEME CHRONIC MEDICINE PROGRAMME GENERAL INFORMATION LETTER LIST OF CHRONIC CONDITIONS Conditions covered under s chronic medication benefit are detailed below.
RAND WATER MEDICAL SCHEME RAND WATER MEDICAL SCHEME CHRONIC MEDICINE PROGRAMME GENERAL INFORMATION LETTER LIST OF CHRONIC CONDITIONS Conditions covered under s chronic medication benefit are detailed below.
Adjuvant Systemic Therapy in Early Stage Breast Cancer
 Adjuvant Systemic Therapy in Early Stage Breast Cancer Julie R. Gralow, M.D. Director, Breast Medical Oncology Jill Bennett Endowed Professor of Breast Cancer Professor, Global Health University of Washington
Adjuvant Systemic Therapy in Early Stage Breast Cancer Julie R. Gralow, M.D. Director, Breast Medical Oncology Jill Bennett Endowed Professor of Breast Cancer Professor, Global Health University of Washington
LUPUS. and Medication LUPUSUK 2015
 9 LUPUS and Medication LUPUSUK 2015 LUPUS and Medication The types of drugs used in lupus can be broadly divided into those that treat the disease itself (e.g. hydroxychloroquine and prednisolone) and
9 LUPUS and Medication LUPUSUK 2015 LUPUS and Medication The types of drugs used in lupus can be broadly divided into those that treat the disease itself (e.g. hydroxychloroquine and prednisolone) and
Active Cancer Studies by Approval Date For additional information on any one of these studies contact the Lancaster General Cancer Center
 Active Cancer Studies by Approval Date For additional information on any one of these studies contact the Lancaster General Cancer Center 717-544-3113 PROTOCOL NO STUDY TITLE PRINCIPAL INVESTIGATOR ECOG
Active Cancer Studies by Approval Date For additional information on any one of these studies contact the Lancaster General Cancer Center 717-544-3113 PROTOCOL NO STUDY TITLE PRINCIPAL INVESTIGATOR ECOG
 Exhibit B United States Patent Application 20020012663 Kind Code A1 Waksal, Harlan W. January 31, 2002 Treatment of refractory human tumors with epidermal growth factor receptor antagonists Abstract A
Exhibit B United States Patent Application 20020012663 Kind Code A1 Waksal, Harlan W. January 31, 2002 Treatment of refractory human tumors with epidermal growth factor receptor antagonists Abstract A
Clinical Policy: Nivolumab (Opdivo) Reference Number: CP.PHAR.121 Effective Date: Last Review Date: Line of Business: Medicaid
 Clinical Policy: (Opdivo) Reference Number: CP.PHAR.121 Effective Date: 07.15 Last Review Date: 01.18 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: (Opdivo) Reference Number: CP.PHAR.121 Effective Date: 07.15 Last Review Date: 01.18 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
acting aggressive, being angry or violent yourself, or feeling hopeless)
 MEDICATION GUIDE INTRON A (In-tron-aye) (Interferon alfa-2b, recombinant) If you are taking INTRON A with REBETOL, also read the Medication Guide for REBETOL (ribavirin) Capsules and Oral Solution. INTRON
MEDICATION GUIDE INTRON A (In-tron-aye) (Interferon alfa-2b, recombinant) If you are taking INTRON A with REBETOL, also read the Medication Guide for REBETOL (ribavirin) Capsules and Oral Solution. INTRON
Report on Investigation Results
 Pharmaceuticals and Medical Devices Agency This English version is intended to be a reference material for the convenience of users. In the event of inconsistency between the Japanese original and this
Pharmaceuticals and Medical Devices Agency This English version is intended to be a reference material for the convenience of users. In the event of inconsistency between the Japanese original and this
CHRONIC MEDICATION PROGRAMME INCLUDES PRESCRIBED MINIMUM BENEFIT CHRONIC DISEASE LIST (CDL)
 CHRONIC MEDICATION PROGRAMME INCLUDES PRESCRIBED MINIMUM BENEFIT CHRONIC DISEASE LIST (CDL) A. GENERAL INFORMATION LIST OF CHRONIC CONDITIONS Conditions covered under KeyHealth s chronic medication benefit
CHRONIC MEDICATION PROGRAMME INCLUDES PRESCRIBED MINIMUM BENEFIT CHRONIC DISEASE LIST (CDL) A. GENERAL INFORMATION LIST OF CHRONIC CONDITIONS Conditions covered under KeyHealth s chronic medication benefit
DENOSUMAB (PROLIA & XGEVA )
 DENOSUMAB (PROLIA & XGEVA ) UnitedHealthcare Oxford Clinical Policy Policy Number: PHARMACY 306.3 T2 Effective Date: July 2, 2018 Table of Contents Page INSTRUCTIONS FOR USE... 1 CONDITIONS OF COVERAGE...
DENOSUMAB (PROLIA & XGEVA ) UnitedHealthcare Oxford Clinical Policy Policy Number: PHARMACY 306.3 T2 Effective Date: July 2, 2018 Table of Contents Page INSTRUCTIONS FOR USE... 1 CONDITIONS OF COVERAGE...
Avastin (bevacizumab)
 Avastin (bevacizumab) Policy Number: 5.02.502 Last Review: 04/2018 Origination: 03/2017 Next Review: 04/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will provide coverage for Avastin
Avastin (bevacizumab) Policy Number: 5.02.502 Last Review: 04/2018 Origination: 03/2017 Next Review: 04/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will provide coverage for Avastin
Prior treatment with non-biologic Disease- Modifying Antirheumatic. Not to be used in combination with another biologic DMARD
 Abatacept (Orencia) 1, 2, 7, 11, 13, 14, 18, 24, 31, 44, 48, 49, 51, 53, 55, 57 J0129 Alpha 1 - Proteinase inhibitor (Prolastin-C) 5, 6, 10, 12, 40 Medically Necessary (if all the following criteria apply):
Abatacept (Orencia) 1, 2, 7, 11, 13, 14, 18, 24, 31, 44, 48, 49, 51, 53, 55, 57 J0129 Alpha 1 - Proteinase inhibitor (Prolastin-C) 5, 6, 10, 12, 40 Medically Necessary (if all the following criteria apply):
Hodgkin and Non-Hodgkin Lymphoma (LYM) Post-Infusion Data
 Hodgkin and Non-Hodgkin Lymphoma (LYM) Post-Infusion Data Registry Use Only Sequence Number: Date Received: CIBMTR Center Number: CIBMTR Research ID: Event date: / / Visit 100 day 6 months 1 year 2 years
Hodgkin and Non-Hodgkin Lymphoma (LYM) Post-Infusion Data Registry Use Only Sequence Number: Date Received: CIBMTR Center Number: CIBMTR Research ID: Event date: / / Visit 100 day 6 months 1 year 2 years
H&HD ANTINEOPLASTIC DRUG CARD ASSEMBLY INSTRUCTIONS
 H&HD ANTINEOPLASTIC DRUG CARD ASSEMBLY INSTRUCTIONS Each of you should have 37 new cards: 7 orange cards for antimetabolites 11 white cards for miscellaneous drugs (2 DNA synthesis inhibitors, 1 enzyme,
H&HD ANTINEOPLASTIC DRUG CARD ASSEMBLY INSTRUCTIONS Each of you should have 37 new cards: 7 orange cards for antimetabolites 11 white cards for miscellaneous drugs (2 DNA synthesis inhibitors, 1 enzyme,
Liver Health: Do you have liver problems? Yes No If so, please specify:
 Medical History General Last Name: First Name: Date of Birth: Age: Contact Number: Are you in good health to the best of your knowledge Medical Information: Please list any physicians you see and their
Medical History General Last Name: First Name: Date of Birth: Age: Contact Number: Are you in good health to the best of your knowledge Medical Information: Please list any physicians you see and their
PROFMED MEDICAL SCHEME CHRONIC MEDICINE BENEFIT GENERAL INFORMATION
 PROFMED MEDICAL SCHEME CHRONIC MEDICINE BENEFIT GENERAL INFORMATION The Prescribed Minimum Benefit Chronic Disease List In terms of the Medical Schemes Act Regulations that came into effect on 1 January
PROFMED MEDICAL SCHEME CHRONIC MEDICINE BENEFIT GENERAL INFORMATION The Prescribed Minimum Benefit Chronic Disease List In terms of the Medical Schemes Act Regulations that came into effect on 1 January
Bevacizumab (Avastin)
 Bevacizumab (Avastin) Policy Number: Original Effective Date: MM.04.001 09/14/2004 Line(s) of Business: Current Effective Date: HMO; PPO; QUEST 03/01/2014 Section: Prescription Drugs Place(s) of Service:
Bevacizumab (Avastin) Policy Number: Original Effective Date: MM.04.001 09/14/2004 Line(s) of Business: Current Effective Date: HMO; PPO; QUEST 03/01/2014 Section: Prescription Drugs Place(s) of Service:
Avastin. Avastin (bevacizumab) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.21.04 Subject: Avastin Page: 1 of 9 Last Review Date: September 20, 2018 Avastin Description Avastin
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.21.04 Subject: Avastin Page: 1 of 9 Last Review Date: September 20, 2018 Avastin Description Avastin
* Note: please reference the Highmark Health Options Gender Transition Services (MP- 033-MD-DE) policy for all gender dysphoria requests.
 Request for Prior Authorization for Lupron, Lupron Depot (leuprolide acetate) Website Form www.highmarkhealthoptions.com Submit request via: Fax - 1-855-476-4158 All requests for Lupron (leuprolide acetate)
Request for Prior Authorization for Lupron, Lupron Depot (leuprolide acetate) Website Form www.highmarkhealthoptions.com Submit request via: Fax - 1-855-476-4158 All requests for Lupron (leuprolide acetate)
