Guidance for the prevention and treatment of venous thromboembolism in cancer patients
|
|
|
- Cynthia Fowler
- 6 years ago
- Views:
Transcription
1 Guidance for the prevention and treatment of venous thromboembolism in cancer patients A. Awada 1, J-F. Baurain 2, P. Clement 3, P. Hainaut 2, S. Holbrechts 4, J-M. Hougardy 5, K. Jochmans 6, V. Mathieux, J. Mebis 8, M. Strijbos 9, C. Vulsteke 10, P. Verhamme 3 Venous thrombosis is a common complication in cancer patients and thromboembolism is the second most common cause of death. Several practice guidelines provide recommendations for the management of cancer-associated thrombosis. However, these guidelines do not sufficiently cover commonly encountered clinical challenges. With this expert panel, consisting of medical oncologists, haematologists, internists and thrombosis specialists, we aimed to develop a practical Belgian guidance for adequate prevention and treatment of cancer-associated thrombosis that covered several challenging situations encountered in daily clinic. This paper discusses the following topics: type and treatment duration of anticoagulant therapy, recurrent VTE despite anticoagulation, anticoagulation in case of renal impairment, liver disease and thrombocytopenia, the role of anti-xa monitoring, central venous catheter-associated thrombosis, the position of direct oral anticoagulants and thromboprophylaxis, both in ambulatory and hospitalised patients. For an overview of the recommendations formulated by the expert panel, we refer to the key messages for clinical practice in this article. (Belg J Med Oncol 2016;10(): ) Introduction It is well established that cancer patients are at increased risk of venous thromboembolism (VTE), which includes both pulmonary embolism (PE) and deep vein thrombosis (DVT). The presence of malignancy increases the risk of VTE by a factor of four. 1,2 Importantly, VTE is strongly associated with short- and long-term mortality. In fact, in cancer patients, thromboembolism represents the second most common cause of death after cancer progression. 2,3 In addition to this, VTE associated with cancer is also associated with a higher rate of recurrent thrombosis as well as bleeding. 3,4 Finally, VTE also leads to a threefold increase in hospitalisations and higher total health care costs. 5 As such, adequate prevention and treatment of cancer-associated thrombosis (CAT) is vital to reduce its burden on patients with malignancy and on the health care system as a whole. Numerous international clinical guidelines provide recommendations to oncologists for the management of cancer-associated thrombosis However, some challenging clinical situations are frequently encountered by physicians caring for this patient population (e.g. thrombocytopenia, recurrent VTE, catheter-related thrombosis, renal impairment, etc.), which are not sufficiently 1 Institut Jules Bordet, Université libre de Bruxelles, Brussels, 2 Cliniques Universitaires Saint-Luc, Université catholique de Louvain, Brussels, 3 University Hospital Leuven, Leuven, 4 CHU Ambroise Paré, Mons, 5 Hôpital Erasme, Brussels, 6 Universitair Ziekenhuis Brussel, Brussels, CHU Namur, Sainte-Elisabeth, Namur, 8 Jessa Ziekenhuis, Hasselt, 9 AZ KLINA, Brasschaat, 10 Department of Molecular Imaging, Pathology, Radiotherapy & Oncology (MIPRO), Center for Oncological Research (CORE), Antwerp University, Antwerp. Please send all correspondence to: A. Awada, MD, PhD, Institut Jules Bordet, Waterloolaan 121, 1000 Brussels, Belgium, tel: , ahmad.awada@bordet.be. Conflict of interest: This publication is based on the Belgian expert consensus meeting logistically organised by Leo Pharma. Leo Pharma also provided fi nancial support for editorial assistance. Keywords: anti-xa monitoring, cancer-associated thrombosis, central venous catheter, direct oral anticoagulant (DOAC), liver disease, prophylaxis, recurrent venous thromboembolism, renal impairment, thrombocytopenia, thrombosis, VTE. 249
2 addressed in these guidelines. In addition to this, there are also geographical differences in how CAT is managed. With this expert panel, consisting of medical oncologists, haematologists, internists and thrombosis specialists, we aimed to develop a practical Belgian guidance for adequate prevention and treatment of CAT and covering the challenging situations encountered in daily clinic Methods The purpose of this consensus paper was to provide expert guidance for challenging VTE scenarios. The specific topics to be addressed were identified by the chairmen of the working group (A. Awada and P. Verhamme). For each of the identified challenges, two experts of the panel performed an in depth literature search after which their findings were reviewed and discussed by the entire expert panel. The following eight topics were selected: 1. Type and treatment duration of anticoagulant therapy (including treatment beyond 6 months) 2. Recurrent VTE despite anticoagulation (including role of inferior vena cava [IVC] filters) 3. Anticoagulation in case of renal impairment and liver disease 4. Anticoagulation in case of thrombocytopenia 5. Anti-Xa monitoring 6. Prophylaxis of VTE in cancer patients with a Central venous catheter (CVC) and treatment of CVCassociated thrombosis. Direct oral anticoagulants (DOACs) and CAT 8. Prophylaxis a. Indication and type of prophylaxis in ambulatory patients b. Indication and type of prophylaxis in hospitalised patients After discussion of these different topics reviewed and presented by two experts during a first consensus meeting (June 15 th 2016), a paper was drafted. This paper was then reviewed by the members of the expert panel and finalised during a second consensus meeting (September th 2016). These meetings were supported by Leo Pharma at the logistical level. Type and treatment duration of anticoagulant therapy The standard initial treatment of an acute episode of VTE in cancer patients should consist of the administration of three to six months of subcutaneous low molecular weight heparin (LMWH) at a dose adjusted to the body weight (Table 1). This recommendation is based on the outcome of large randomised controlled trials. 11 Oral anticoagulation with vitamin K antagonists (VKA) can be problematic in patients with cancer as these patients often have several co-morbidities and co-medications. Drug interactions, malnutrition and liver dysfunction can lead to wide fluctuations in the International Normalised Ratio (INR). The results of randomised clinical trials indicate that treatment with a LMWH for six months is safe and more effective than treatment with a VKA. 12,13 Studies with dalteparin implemented a dose reduction from a full-therapeutic to a subtherapeutic dose (200 to 150 anti-xa units/kg) after four weeks, whereas studies with tinzaparin and enoxaparin were performed without dose change (15 and 150 anti-xa units/kg, respectively) in the first three months. There are no published studies addressing optimal anticoagulation beyond six months in patients with cancer. However, it is the consensus of the panel that continuing anticoagulation beyond six months should be considered in patients with a persistent high-risk of recurrence in patients with active cancer. However, there is much debate on a firm definition of active disease. In the context of this paper, the experts defined active disease as the presence of progressive tumour or ongoing cancer therapy. From six months onwards it is warranted to individualise the dosing of the LMWH to find an optimal balance between safety, the risk of recurrence and the quality of life (QoL). Of note, in patients with cancer with a first deep vein thrombosis (DVT) who were treated for 6 months with a LMWH, the absence of residual vein thrombosis identified a population at low risk for recurrent thrombotic events. 14 A special case was made for patients that are treated in a curative setting (adjuvant therapy). This includes breast cancer patients receiving tamoxifen or chemotherapy in the adjuvant setting. In line with the ESMO guidelines, it was concluded that these patients should be treated with a LMWH for six months, though oral treatment was considered to be an alternative option for selected patients. 9 Recurrent VTE despite anticoagulation The rate of recurrent VTE despite anticoagulation remains high in cancer patients and is associated with a poor prognosis. A systematic review of 1,292 patients with cancer showed that VTE recurrence rates with LM- WH and VKA were 6.5% and 1.9% respectively. 15 The risk of recurrence is highest during the 1 st month of anticoagulation. 250
3 Table 1. LMWH doses for the treatment of acute VTE in Belgium. LMWH Recommended dose Once Daily Tinzaparin (Innohep ) Dalteparin (Fragmin ) Nadroparin (Fraxodi ) Enoxaparin (Clexane ) 15 U/kg/24h 200 U/kg/24h 0.1 ml/10kg/24h 1.5 mg/kg/24h Twice Daily Dalteparin (Fragmin ) Nadroparin (Fraxiparine ) Enoxaparin (Clexane ) 100 U/kg/12h 0.1 ml/10kg/12h 1 mg/kg/12h Patients with recurrent VTE, despite standard doses of anticoagulant therapy, should be assessed for treatment compliance, heparin-induced thrombocytopenia (HIT), or any evidence of mechanical compression resulting from malignancy. If a patient is treated with VKA, it is recommended to switch to a LMWH. However, the reverse does not hold true: switching from a LMWH to VKA in case of VTE recurrence should not be considered. If a patient is treated with a sub-therapeutic LMWH dose, it is recommended to return to the therapeutic dose. If a therapeutic dose is already given, the dose can be escalated by 20% to 25%. 16 In case of supra-therapeutic dosing, twice daily dosing of the LM- WH was recommended by the expert panel. If moving to supra-therapeutic levels, anti-xa monitoring should be considered when feasible. In some cases, the addition of low-dose aspirin can be considered, although firm evidence for this is lacking. The expert group advises against the routine use of an IVC filters in patients who present with recurrent VTE despite therapeutic anticoagulation. This should only be considered in selected cases. Anticoagulation in case of renal impairment Renal impairment is a common problem in cancer and is associated with a higher bleeding risk. The safe use of LMWHs implies a preserved renal function (GFR >50 ml/min). In patients with lower GFR (30-50 ml/min), LMWHs remain the first choice but bioaccumulation can already occur. The most important risk of bleeding concerns patients with severe renal impairment (GFR <30 ml/min). LMWHs with little bioaccumulation (e.g. dalteparin, tinzaparin) are preferred in patients with severe renal impairment. 1 Dose adjustment based on anti-xa monitoring was suggested in patients with CrCl <30 ml/min, however evidence for a clear benefit of this approach is lacking. For severely renally impaired patients with acute VTE, intravenous unfractionated heparin with aptt monitoring is recommended for the initial treatment. In fact, patients with solid tumours and such a low creatinine clearance are usually in a very bad shape and are better managed in the hospital. For a more complete overview on the management of cancer-associated thrombosis (CAT) in renally impaired patients we also refer to the letter to the editor of professor Hougardy and professor Motte in the same issue of this journal. Anticoagulation in case of liver disease The data on the treatment of CAT in patients with liver disease are limited. In a preventive study, it was shown that twelve months of enoxaparin was safe and effective in preventing portal vein thrombosis in patients with cirrhosis and a Child-Pugh score of This effect was associated with improved survival. LMWHs are believed to be safe in patients with comorbid liver disease. However, the clinician needs to assess the indication of LMWH case by case in patients with liver disease. Of note, the anti-xa assay is difficult in patients with liver disease and might underestimate the true degree of anticoagulation, which could lead to incorrect dose adaptation and subsequent morbidity and mortality. 251
4 Anticoagulation in case of thrombocytopenia There are no trial data on the anticoagulation of cancer patients with thrombocytopenia. Before moving to treatment, one must first search for the aetiology of the thrombocytopenia (treatment-related, bone marrow involvement of the cancer, idiopathic thrombocytopenic purpura [ITP], or heparin-induced thrombocytopenia [HIT]). In this light one must exclude a reversible, or treatable cause. For the treatment of CAT with thrombocytopenia, the expert group endorses the recommendation of the International Society on Thrombosis and Haemostasis (ISTH) on this matter. 19 Full therapeutic doses of anticoagulation with LMWHs are recommended without platelet transfusion in patients with CAT and a platelet count of 50 x109/l or higher and no clinical signs of bleeding. In case of acute CAT and thrombocytopenia (platelet count below 50 x109/l) full therapeutic doses of anticoagulation are recommended; in these patients platelet transfusion to maintain a platelet level of 50 x109/l or higher should be considered. For subacute or chronic CAT and thrombocytopenia (platelet count below 50 x109/l) it is recommended to reduce the LMWH dose to 50% of the therapeutic dose or to use a prophylactic dose of LMWH in patients with a platelet count between 25 and 50 x109/l. In patients with a platelet count below 25 x109 discontinuing anticoagulation is suggested. For thrombopenic patients who need LMWH prophylaxis, consider case-by-case approach between 20 and 50 x109/l. Below 20 x109/l platelets, no prophylaxis is recommended (except for some hematologic patients). Anti-Xa monitoring LMWHs have predictable pharmacodynamic profiles and wide therapeutic windows that do not require routine coagulation monitoring in clinically stable and uncomplicated patients. Anti-Xa is a surrogate marker that measures the anticoagulant effect of LMWH and is assumed to correlate with haemorrhagic and thromboembolic events. From a practical point of view, there are large variations between reagents used in anti-xa assays and between laboratories, limiting their utility. Moreover, target anti-xa levels are not clinically validated and there are no standardised methods to adjust LMWH doses based on anti-xa levels. In fact, there are no data suggesting that adjusting the LMWH dose based on peak anti-xa levels is correlated with improved safety and efficacy. Peak anti-xa levels (blood sampling four hours after injection) should not be utilised to evaluate prophylactic or therapeutic LMWH dosing regimens in routine clinical practice. In selected patient populations (e.g. severe renal impairment), trough anti-xa levels (blood sampling before the next injection) may have a role in evaluating LMWH accumulation and the need to adjust the dose or prolong the dosing interval (trough target <0.4 IU/mL). Only limited data are available on the use of anti-xa levels to monitor and adjust LMWH daily doses in patients with severe renal impairment (<30 ml/min). 20 Anti-Xa levels remain of limited value in patients with renal impairment because they are poorly correlated with bleeding, especially in patients with high risk of bleeding complications. 21 Prophylaxis of VTE in cancer patients with a CVC and treatment of CVCassociated thrombosis Central venous catheters (CVC) are associated with upper extremity DVT and PE and are independent risk factors for VTE. Recent studies and a meta-analysis of randomised controlled trials on this matter did not show clinically meaningful degrees of protection against catheter-induced VTE using either low-dose warfarin or LM- WH in patients with cancer. 22,23,19 As such, the expert group does not recommend primary prophylaxis for cancer patients with a CVC, unless in patients with a very high VTE risk (e.g. patients with a femoral catheter). A diagnosis of a CVC-related thrombosis should be confirmed by ultrasonography. If there is a high suspicion for a CVC-related thrombosis despite a negative ultrasonography, a venography, or CT venography is preferred over a repeated ultrasonography. For the treatment of CVC-related VTE, LMWH is the preferred treatment. Local thrombolysis is not routinely recommended, but may be occasionally considered. The catheter should be left in place if the symptoms improve upon anticoagulation and if the catheter is still needed, functions properly and is not infected. Patients with CVC-associated thrombosis should be treated for 3 months. When the catheter remains in place and is still used for chemotherapy and the bleeding risk is low, the panel recommends prolonging the thromboprophylaxis for as long as the chemotherapy is given, and/or in cases where venous access is precious. If the catheter is removed one should not treat longer than three months. A shorter period can be considered if there is complete resolution of the thrombus. LMWH is the preferred treatment for long-term pre- 252
5 vention, but the dose can be adjusted based on the assessment of the bleeding risk. Oral treatment can be considered if subcutaneous administration is poorly tolerated. 23 Direct oral anticoagulants (DOACs) and CAT None of the recently introduced direct oral anticoagulants (DOAC) (rivaroxaban, dabigatran, apixaban and edoxaban) was specifically tested in cancer patients, and in all of the studies with these agents the control arm received VKA instead of LMWH (the current standard of care). Therefore, the safety and efficacy of DO- ACs in patients with CAT remains uncertain. As such, the expert panel concluded that LMWH should remain the preferred treatment option for both the initial and long-term treatment of CAT, awaiting the result of ongoing studies. Nevertheless, in some cases DOACs could form an alternative for non-progressive cancer patients (e.g. breast cancer patients with thrombosis while under adjuvant therapy with tamoxifen). DOACs could also be an alternative for patients who can no longer tolerate parenteral therapy. In these cases, the ease of use of DOACs may be preferred over the disadvantages of VKA (e.g. small window of efficacy). However, when opting for a DOAC one should take into account the specific metabolic pathways and the route of elimination of these agents (to avoid drug-drug interactions) and ask for advice from a haemostasis specialist. Prophylaxis Indication and type of prophylaxis in ambulatory patients The incidence of VTE is highly heterogeneous in the cancer population ranging from 0.6 to 26 %. 24 The Khorana score is the only validated score to assess the risk of VTE, but with limitations of not being representative of all tumour categories and not prospectively tested in clinical trials. 25 Several studies have assessed thromboprophylaxis of ambulatory cancer patients under chemotherapy Despite the fact that these studies consistently demonstrate a benefit for thromboprophylaxis, the difference in absolute risk was very small (to avoid 1 event, patients should be treated). Combined with the important cost (and risk) of LMWHs, these findings did not convince the expert group to recommend routine thromboprophylaxis in ambulatory cancer patients. A special exception is made for patients with multiple myeloma receiving thalidomide- or lenalidomide-based therapy. The risk for VTE in these patients is very high and ranges from 12% to 28%. Two randomised trials have been published comparing VTE prophylaxis and observation. 29,30 In these studies, both aspirin and LM- WH were shown to be acceptable thromboprophylaxis options (with slightly greater efficacy of LMWH). Based on these findings, the expert panel recommends thromboprophylaxis with aspirin for low-risk multiple myeloma patients and with LMWH for patients with a high thrombosis risk. In addition to this, the expert panel indicated that thromboprophylaxis with LMWH could be considered for selected patients at particularly high risk, such as pancreatic cancer patients receiving chemotherapy with a low risk of bleeding. Indication and type of prophylaxis in hospitalised patients Three large randomised phase III studies reported a significant reduction in VTE following prophylactic treatment with LMWH, or fondaparinux in medically ill hospitalised patients However, no specific studies on the use of thromboprophylaxis in hospitalised, nonsurgical cancer patients have been conducted. A recent meta-analysis of the cancer population in three placebocontrolled trials on thromboprophylaxis failed to show a significant reduction in the incidence of VTE when pharmacological anticoagulation was used. 34 The expert panel supports the use of thromboprophylaxis with LMWHs in all cancer patients with progressive disease hospitalised for an acute medical complication and who are bedridden. For all other cases (e.g. paucisymptomatic disease), other risk factors should be taken into account (e.g. restricted mobility, obesity, history of VTE, stage and histology of the cancer, race, platelet count, presence of CVC, use of anti-angiogenic agents, etc.) For cancer patients who are hospitalised in a surgical setting, the situation is easier. In patients with malignant disease undergoing a major surgical intervention, thromboprophylaxis with LMWH for at least -10 days is recommended. In patients with high-risk features, the prophylaxis can be extended up to four weeks. Of note, mechanical methods of thromboprophylaxis (graduated compression stockings [GCS], intermittent pneumatic compression [IPC]) can be used in high-risk surgical patients with a contra-indication for pharmacological thromboprophylaxis. In this respect, IPC should be preferred over GCS when available. References 1. Horsted F, West J, Grainge M, et al. Risk of venous thromboembolism in patients with cancer: a systematic review and meta-analyis. PLoS Med 2012;9:e
6 Key messages for clinical practice 1. Type and treatment duration of anticoagulant therapy 3 to 6 months of LMWH is the preferred initial treatment for CAT. In case of active disease, or when risk of recurrence remains high, treatment can be prolonged beyond 6 months. 2. Recurrent VTE despite anticoagulation Assess for treatment compliance, HIT or any evidence of mechanical compression resulting from malignancy. If treated with VKA, switch to LMWH. If LMWH dosing is sub-therapeutic, move towards therapeutic dose. If therapeutic dosing, increase dose with 20%-25%. Twice daily dosing is preferred. IVC filters to be considered in very selected patients. 3. Anticoagulation in case of renal impairment and liver disease Caution is warranted in the anticoagulation of cancer patients with renal impairment given the increased bleeding risk resulting from bio-accumulation. In patients with a severe renal impairment, the use LMWHs that are less depending on clearance by the kidney (i.e. dalteparin, tinzaparin) is preferred. LMWHs appear to be safe in patients with comorbid liver disease. In cases with severe liver disease the clinician needs to assess the indication of LMWH on a case-by-case basis. 4. Anticoagulation in case of thrombocytopenia Platelet count of 50 x109/l or higher and no clinical sign of bleeding: full dose LMWH without platelet transfusion. Platelet count below 50 x109/l and acute CAT: full dose LMWH and consider platelet transfusion to maintain a platelet level of 50 x109/l or higher. Platelet count between 25 and 50 x109/l and subacute or chronic CAT: reduce LMWH dose by 50%, or use a prophylactic dose of LMWH. Platelet count below 25 x109: discontinue anticoagulation. 5. Anti-Xa monitoring Routine anti-xa monitoring is not needed, only to be considered in special cases (recurrence during optimal anticoagulation, renal impairment). If anti-xa monitoring is used to exclude accumulation, measurement of trough levels is recommended. 6. Prophylaxis of VTE in cancer patients with a CVC and treatment of CVC-associated thrombosis Primary prophylaxis for cancer patients with a CVC is not recommended. Diagnosis of a CVC-related thrombosis should be confirmed by ultrasonography. LMWH for at least 3 months is the preferred treatment option. Extend therapy for as long as catheter is in place can be considered. Catheter can be left in place if the symptoms improve upon anticoagulation and if the catheter is still needed, functions properly and is not infected.. Direct oral anticoagulants (DOACs) and CAT LMWHs remain the preferred treatment option for both the initial and long-term treatment of CAT. The safety and efficacy of DOACs in patients with CAT remains uncertain. Results of clinical trials are awaited. 8. Prophylaxis Indication and type of prophylaxis in ambulatory patients. - Routine thromboprophylaxis in ambulatory cancer patients is not recommended. - Thromboprophylaxis can be considered in selected patients: o Multiple myeloma patients treated with immunomodulatory agents, or carfilzomib. o Pancreatic cancer patients receiving chemotherapy with a low risk of bleeding. Indication and type of prophylaxis in hospitalised patients. - In patients with malignant disease undergoing a major surgical intervention, thromboprophylaxis with LMWH for at least -10 days is recommended. - It is recommended to extend prophylaxis to 4 weeks in high-risk situations. - Thromboprophylaxis with LMWHs can be considered in all active cancer patients hospitalised for a medical illness. - For all other cases, specific risk factors for VTE should be taken into account. 254
7 2. Khorana A, Francis C, Culakova E, et al. Frequency, risk factors and trends for venous thromboembolism among hospitalized cancer patients. Cancer 200;110: Khorana A, Francis C, Culakova E, et al. Thromboembolism is a leading cause of death in cancer patients receiving outpatient chemotherapy. J Thromb Haemost 200;5(3): Prandoni P, Lensing A, Piccioli A, et al. Recurrent venous thromboembolism and bleeding complications during anticoagulant treatment in patients with cancer and venous thrombosis. Blood 2002;100(10): Khorana A, Dalal M, Lin J, et al. Health care costs associated with venous thromboembolism in selected high-risk ambulatory patients with solid tumours undergoing chemotherapy in the United States. Clinicecon Outcomes Res 2013;5: Kearon C, Akl E, Ornelas J, et al. Antithrombotic therapy for VTE disease. Chest 2016;149(2): Lyman G, Bohlke K, Khorana A, et al. Venous thromboembolism prophylaxis and treatment in patients with cancer: American society of clinical oncology clinical practice guideline update J Clin Oncol 2015;33(6): Streiff MB, Holmstrom B, Ashrani A, et al. Cancer-Associated Venous Thromboembolic Disease, Version J Natl Compr Canc Netw 2015;13(9): Mandalà M, Falanga A, Roila F, et al. Management of venous thromboembolism (VTE) in cancer patients: ESMO Clinical Practice Guidelines. Ann Oncol 2011;22 Suppl 6:vi Khorana A, Carrier M, Garcia D, et al. Guidance for the prevention and treatment of cancer-associated venous thromboembolism. J Thromb Thrombolysis 2016;41(1): Akl E, Labedi N, Barba M, et al. Anticoagulation for the initial treatment of venous thromboembolism in patients with cancer. Cochrane database Syst Rev 2011;6:CD Lee A, Levine M, Baker R, et al. Low-molecular weight heparin versus a coumarin for the prevention of recurrent venous thromboembolism in patients with cancer. N Engl J Med 2003;349: Meyer G, Marjanovic Z, Valcke J, et al. Comparison of low-molecular weight heparin and warfarin for the secondary prevention or recurrent venous thromboembolism in patients with cancer: a randomized controlled trial. Arch Intern Med 2002;162: Romera-Villegas A, Cairols-Castellote M, Vila-Coll R, et al. Long-term use of different doses of low-molecular-weight heparin versus vitamin K antagonists in the treatment of venous thromboembolism. Ann Vasc Surg 2010;24: Carrier M, Le Gal G, Cho R, et al. Dose escalation of low-molecular-weight heparin to manage recurrent venous thromboembolic events despite systemic anticoagulation in cancer patients. J Thromb Haemost 2009;: Di Nisio M, Carrier M, Lyman G, et al. Prevention of venous thromboembolism in hospitalized medical cancer patients: guidance from the SSC of the ISTH. J Thromb Haemost 2014;12(10): Easaw J, Shea-Budgell M, Wu C, et al. Canadian consensus recommendations on the management of venous thromboembolism in patients with cancer. Part 2: treatment. Curr Oncol 2015;22(2): Villa E, Cammà C, Marietta M, et al. Enoxaparin prevents portal vein thrombosis and liver decompensation in patients with advanced cirrhosis. Gastroenterology 2012;143(5): e Carrier M, Kohrana A, Zwicker J, et al. Management of challenging cases of patients with cancer-associated thrombosis including recurrent thrombosis and bleeding: guidance from the SSC of the ISTH. J Thromb Haemost 2013;11(9): Nutescu E, Spinler S, Wittkowsky A, et al. Low-molecular-weight heparins in renal impairment and obesity: available evidence and clinical practice recommendations across medical and surgical settings. Ann Pharmacother 2009;43(6): Brophy D, Martin E, Gehr T, et al. Enhanced anticoagulant activity of enoxaparin in patients with ESRD as measured by thrombin generation time. Am J Kidney Dis 2004;44(2): Brophy D, Martin E, Gehr T, et al. Thrombin generation time is a novel parameter for monitoring enoxaparin therapy in patients with end-stage renal disease. J Thromb Haemost 2006;4(2): Debourdeau P, Farge D, Beckers M, et al. International clinical practice guidelines for the treatment and prophylaxis of thrombosis associated with central venous catheters in patients with cancer. J Thromb Haemost 2013;11(1): Carrier M, Lazo-Langner A, Shivakumar S, et al. Clinical challenges in patients with cancer-associated thrombosis: Canadian expert consensus recommendations. Curr Oncol 2015;22(1): Khorana A, Connoly G, et al. Assessing risk of venous thromboembolism in patient with cancer. J Clin Oncol 2009;2(29): Khorana A, Kuderer N, Culakova E, et al. Development and validation of a predictive model for chemotherapy-associated thrombosis. Blood 2008;111(10): Akl E, Gunukula S, Barba M, et al. Parenteral anticoagulation in patients with cancer who have no therapeutic or prophylactic indication for anticoagulation. Cochrane Database Syst Rev 2011;(4):CD Di Nisio M, Porreca E, Ferrante N, et al. Primary prophylaxis for venous thromboembolism in ambulatory cancer patients receiving chemotherapy. Cochrane Database Syst Rev 2012;(2):CD Akl E, Schünemann H. Routine heparin for patients with cancer? One answer, more questions. N Engl J Med 2012;366(): Palumbo A, Cavo M, Bringhen S, et al. Aspirin, warfarin, or enoxaparin thromboprophylaxis in patients with multiple myeloma treated with thalidomide: a phase III, open-label, randomized trial. J Clin Oncol 2011;29(8): Larocca A, Cavallo F, Bringhen S, et al. Aspirin or enoxaparin thromboprophylaxis for patients with newly diagnosed multiple myeloma treated with lenalidomide. Blood 2012;119(4): Cohen A, Davidson B, Gallus A, et al. Efficacy and safety of fondaparinux for the prevention of venous thromboembolism in older acute medical patients: randomised placebo controlled trial. BMJ 2006;332(53): Samama M, Cohen A, Darmon J, et al. comparison of enoxaparin with placebo for the prevention of venous thromboembolism in acutely ill medical patients. Prophylaxis in Medical Patients with Enoxaparin Study Group. N Engl J Med 1999;341(11): Leizorovic A, Cohen A, Turpie A, et al. Efficacy and safety of fixed low-dose dalteparin in preventing venous thromboembolism among obese or elderly hospitalized patients: a subgroup analysis of the PREVENT trial. Arch Intern Med 2005;165(3): Carrier M, Khorana A, Moretto P, et al. Lack of evidence to support thromboprophylaxis in hospitalized medical patients with cancer. Am J Med 2014;12(1):82-6.e Napolitano M, Saccullo G, Malato A, et al. Optimal duration lof low molecular weight heparin for the treatment of cancer-related deep vein thrombosis: The Cancer- DACUS study. J Clin Oncol 2014;32(32):
DEEP VEIN THROMBOSIS (DVT): TREATMENT
 DEEP VEIN THROMBOSIS (DVT): TREATMENT OBJECTIVE: To provide an evidence-based approach to treatment of patients presenting with deep vein thrombosis (DVT). BACKGROUND: An estimated 45,000 patients in Canada
DEEP VEIN THROMBOSIS (DVT): TREATMENT OBJECTIVE: To provide an evidence-based approach to treatment of patients presenting with deep vein thrombosis (DVT). BACKGROUND: An estimated 45,000 patients in Canada
incidence of cancer-associated thrombosis (CAT) is further increased by additional risk factors such as chemotherapeutic 2
 CANCER ASSOCIATED THROMBOSIS TREATMENT Patients with cancer are at a greater risk of developing venous thromboembolism than non-cancer patients, partly due to the ability of tumour cells to activate the
CANCER ASSOCIATED THROMBOSIS TREATMENT Patients with cancer are at a greater risk of developing venous thromboembolism than non-cancer patients, partly due to the ability of tumour cells to activate the
Patients with cancer are at a greater risk of developing venous thromboembolism than non-cancer patients, partly due to the 1
 CANCER ASSOCIATED THROMBOSIS TREATMENT Patients with cancer are at a greater risk of developing venous thromboembolism than non-cancer patients, partly due to the 1 ability of tumour cells to activate
CANCER ASSOCIATED THROMBOSIS TREATMENT Patients with cancer are at a greater risk of developing venous thromboembolism than non-cancer patients, partly due to the 1 ability of tumour cells to activate
Management of Cancer Associated Thrombosis (CAT) where data is lacking. Tim Nokes Haematologist, Derriford Hospital, Plymouth
 Management of Cancer Associated Thrombosis (CAT) where data is lacking Tim Nokes Haematologist, Derriford Hospital, Plymouth Contents Overview of the statistics and aetiology for Cancer Associated Thrombosis
Management of Cancer Associated Thrombosis (CAT) where data is lacking Tim Nokes Haematologist, Derriford Hospital, Plymouth Contents Overview of the statistics and aetiology for Cancer Associated Thrombosis
Are guidelines for anticoagulation useful in cancer patients?
 Session 3 Striking a Balance Between Bleeding and the Risk of Thrombosis in Cancer Patients Are guidelines for anticoagulation useful in cancer patients? Sebastian Szmit Department of Pulmonary Circulation
Session 3 Striking a Balance Between Bleeding and the Risk of Thrombosis in Cancer Patients Are guidelines for anticoagulation useful in cancer patients? Sebastian Szmit Department of Pulmonary Circulation
Venous Thrombo-Embolism. John de Vos Consultant Haematologist RSCH
 Venous Thrombo-Embolism John de Vos Consultant Haematologist RSCH overview The statistics Pathogenesis Prophylaxis Treatment Agent Duration Incidental VTE Recurrence of VTE IVC filters CVC related thrombosis
Venous Thrombo-Embolism John de Vos Consultant Haematologist RSCH overview The statistics Pathogenesis Prophylaxis Treatment Agent Duration Incidental VTE Recurrence of VTE IVC filters CVC related thrombosis
PRIMARY THROMBOPROPHYLAXIS IN AMBULATORY CANCER PATIENTS: CURRENT GUIDELINES
 PRIMARY THROMBOPROPHYLAXIS IN AMBULATORY CANCER PATIENTS: CURRENT GUIDELINES Mario Mandalà, MD Unit of Clinical Research Department of Oncology and Haematology Papa Giovanni XXIII Hospital Cancer Center
PRIMARY THROMBOPROPHYLAXIS IN AMBULATORY CANCER PATIENTS: CURRENT GUIDELINES Mario Mandalà, MD Unit of Clinical Research Department of Oncology and Haematology Papa Giovanni XXIII Hospital Cancer Center
THROMBOPROPHYLAXIS IN CANCER PATIENTS
 CANCER ASSOCIATED THROMBOSIS THROMBOPROPHYLAXIS IN CANCER PATIENTS Cancer is an important risk factor for venous thromboembolism (VTE). Research has shown that 4-20% of 1 patients with cancer experience
CANCER ASSOCIATED THROMBOSIS THROMBOPROPHYLAXIS IN CANCER PATIENTS Cancer is an important risk factor for venous thromboembolism (VTE). Research has shown that 4-20% of 1 patients with cancer experience
Venous Thromboembolism (VTE) in Myeloma. Christine Chen May 2017
 Venous Thromboembolism (VTE) in Myeloma Christine Chen May 2017 Objectives 1. Review the magnitude of the problem and why myeloma patients are at risk of VTE 2. Discuss thromboprophylaxis approaches in
Venous Thromboembolism (VTE) in Myeloma Christine Chen May 2017 Objectives 1. Review the magnitude of the problem and why myeloma patients are at risk of VTE 2. Discuss thromboprophylaxis approaches in
VENOUS THROMBOEMBOLISM, CANCER AND CKD ANOTHER TRIAD TO MANAGE
 VENOUS THROMBOEMBOLISM, CANCER AND CKD ANOTHER TRIAD TO MANAGE I. ELALAMY Service d Hématologie Biologique HOPITAL TENON PARIS INSERM UMR U938 DISCLOSURES Conferences Clinical Studies Board Sanofi X X
VENOUS THROMBOEMBOLISM, CANCER AND CKD ANOTHER TRIAD TO MANAGE I. ELALAMY Service d Hématologie Biologique HOPITAL TENON PARIS INSERM UMR U938 DISCLOSURES Conferences Clinical Studies Board Sanofi X X
CANCER ASSOCIATED THROMBOSIS. Pankaj Handa Department of General Medicine Tan Tock Seng Hospital
 CANCER ASSOCIATED THROMBOSIS Pankaj Handa Department of General Medicine Tan Tock Seng Hospital My Talk Today 1.Introduction 2. Are All Cancer Patients at Risk of VTE? 3. Should All VTE Patients Be Screened
CANCER ASSOCIATED THROMBOSIS Pankaj Handa Department of General Medicine Tan Tock Seng Hospital My Talk Today 1.Introduction 2. Are All Cancer Patients at Risk of VTE? 3. Should All VTE Patients Be Screened
Cancer Associated Thrombosis
 Cancer Associated Thrombosis Can we use DOACs? D R. C Y N T H I A W U MD F R C P ( C ) D I V I S I O N O F H E M A T O L O G Y F A M I L Y P H Y S I C I A N S A N D C A N C E R C O N T R O L A P R I L
Cancer Associated Thrombosis Can we use DOACs? D R. C Y N T H I A W U MD F R C P ( C ) D I V I S I O N O F H E M A T O L O G Y F A M I L Y P H Y S I C I A N S A N D C A N C E R C O N T R O L A P R I L
Anticoagulation in Special populations. Ng Heng Joo Department of Haematology Singapore General Hospital
 Anticoagulation in Special populations Ng Heng Joo Department of Haematology Singapore General Hospital roymatheson.com Objectives Safer anticoagulation for The elderly Chronic kidney disease Obese patients
Anticoagulation in Special populations Ng Heng Joo Department of Haematology Singapore General Hospital roymatheson.com Objectives Safer anticoagulation for The elderly Chronic kidney disease Obese patients
La terapia del TEV nel paziente oncologico nell'era dei DOAC
 XXVI CONGRESSO NAZIONALE FCSA Bologna, 5-7 Novembre 2015 Tromboembolismo venoso La terapia del TEV nel paziente oncologico nell'era dei DOAC ANNA FALANGA Immunoematologia e Medicina Trasfusionale e Centro
XXVI CONGRESSO NAZIONALE FCSA Bologna, 5-7 Novembre 2015 Tromboembolismo venoso La terapia del TEV nel paziente oncologico nell'era dei DOAC ANNA FALANGA Immunoematologia e Medicina Trasfusionale e Centro
Cancer Associated Thrombosis An update.
 Cancer Associated Thrombosis An update. Simon Noble Marie Curie Professor of Supportive and Palliative Medicine Marie Curie Palliative Care Research Centre Cardiff University The coagulation pathway LIQUID
Cancer Associated Thrombosis An update. Simon Noble Marie Curie Professor of Supportive and Palliative Medicine Marie Curie Palliative Care Research Centre Cardiff University The coagulation pathway LIQUID
Cancer and Thrombosis
 Cancer and Thrombosis The close relationship between venous thromboembolism and cancer has been known since at least the 19th century by Armand Trousseau. Thrombosis is a major cause of morbidity and mortality
Cancer and Thrombosis The close relationship between venous thromboembolism and cancer has been known since at least the 19th century by Armand Trousseau. Thrombosis is a major cause of morbidity and mortality
Medical Patients: A Population at Risk
 Case Vignette A 68-year-old woman with obesity was admitted to the Medical Service with COPD and pneumonia and was treated with oral corticosteroids, bronchodilators, and antibiotics. She responded well
Case Vignette A 68-year-old woman with obesity was admitted to the Medical Service with COPD and pneumonia and was treated with oral corticosteroids, bronchodilators, and antibiotics. She responded well
In the Clinic: Annals Sweta Kakaraparthi 1/23/15
 In the Clinic: Annals Sweta Kakaraparthi 1/23/15 Case Scenerio 56 year old female with breast cancer presents to the clinic for her 3 month followup! She is concerned about blood clots and asks you about
In the Clinic: Annals Sweta Kakaraparthi 1/23/15 Case Scenerio 56 year old female with breast cancer presents to the clinic for her 3 month followup! She is concerned about blood clots and asks you about
THROMBOPROPHYLAXIS: NON-ORTHOPEDIC SURGERY
 THROMBOPROPHYLAXIS: NON-ORTHOPEDIC SURGERY OBJECTIVE: To outline a practical approach for the prevention of venous thromboembolism (VTE) in patients undergoing non-orthopedic surgery. BACKGROUND: VTE is
THROMBOPROPHYLAXIS: NON-ORTHOPEDIC SURGERY OBJECTIVE: To outline a practical approach for the prevention of venous thromboembolism (VTE) in patients undergoing non-orthopedic surgery. BACKGROUND: VTE is
Epidemiology of Thrombosis in Patients with Malignancy. Cancer and Venous Thromboembolism. Chew HK, Arch Int Med, Feb Blom et al, JAMA, Feb 2005
 Cancer and Venous Thromboembolism Objectives 1. Epidemiology of thrombosis in patients with malignancy 2. Anticancer agents and thrombosis 3. Current treatment protocols at UHN 4. Prevention of DVT 5.
Cancer and Venous Thromboembolism Objectives 1. Epidemiology of thrombosis in patients with malignancy 2. Anticancer agents and thrombosis 3. Current treatment protocols at UHN 4. Prevention of DVT 5.
Mabel Labrada, MD Miami VA Medical Center
 Mabel Labrada, MD Miami VA Medical Center *1-Treatment for acute DVT with underlying malignancy is for 3 months. *2-Treatment of provoked acute proximal DVT can be stopped after 3months of treatment and
Mabel Labrada, MD Miami VA Medical Center *1-Treatment for acute DVT with underlying malignancy is for 3 months. *2-Treatment of provoked acute proximal DVT can be stopped after 3months of treatment and
Once-Daily, Oral LIXIANA (edoxaban) Met Primary Endpoint in Investigational Hokusai-VTE CANCER Study
 Once-Daily, Oral LIXIANA (edoxaban) Met Primary Endpoint in Investigational Hokusai-VTE CANCER Study Hokusai-VTE CANCER study is a phase 3b, prospective, randomised, open-label, blind end-point (PROBE)
Once-Daily, Oral LIXIANA (edoxaban) Met Primary Endpoint in Investigational Hokusai-VTE CANCER Study Hokusai-VTE CANCER study is a phase 3b, prospective, randomised, open-label, blind end-point (PROBE)
DVT PROPHYLAXIS IN HOSPITALIZED MEDICAL PATIENTS SAURABH MAJI SR (PULMONARY,MEDICINE)
 DVT PROPHYLAXIS IN HOSPITALIZED MEDICAL PATIENTS SAURABH MAJI SR (PULMONARY,MEDICINE) Introduction VTE (DVT/PE) is an important complication in hospitalized patients Hospitalization for acute medical illness
DVT PROPHYLAXIS IN HOSPITALIZED MEDICAL PATIENTS SAURABH MAJI SR (PULMONARY,MEDICINE) Introduction VTE (DVT/PE) is an important complication in hospitalized patients Hospitalization for acute medical illness
Outpatient Treatment of Deep Vein Thrombosis with Low Molecular Weight Heparin (LMWH) Clinical Practice Guideline August 2015
 Outpatient Treatment of Deep Vein Thrombosis with Low Molecular Weight Heparin (LMWH) Clinical Practice Guideline August 2015 General Principles: There is compelling data in the medical literature to support
Outpatient Treatment of Deep Vein Thrombosis with Low Molecular Weight Heparin (LMWH) Clinical Practice Guideline August 2015 General Principles: There is compelling data in the medical literature to support
New oral anticoagulants and Palliative Care.
 New oral anticoagulants and Palliative Care. Simon Noble Marie Curie Professor of Supportive and Palliative Medicine Marie Curie Palliative Care Research Centre Cardiff University The coagulation pathway
New oral anticoagulants and Palliative Care. Simon Noble Marie Curie Professor of Supportive and Palliative Medicine Marie Curie Palliative Care Research Centre Cardiff University The coagulation pathway
Treatment Options and How They Work
 Treatment Options and How They Work Robin Offord Director of Clinical Pharmacy UCL Hospitals NHS Foundation Trust robin.offord@uclh.nhs.uk Introducing the term anticoagulant... What they do Inhibit the
Treatment Options and How They Work Robin Offord Director of Clinical Pharmacy UCL Hospitals NHS Foundation Trust robin.offord@uclh.nhs.uk Introducing the term anticoagulant... What they do Inhibit the
Clinical Guideline for Anticoagulation in VTE
 Clinical Guideline for Anticoagulation in VTE These clinical guidelines are intended to provide evidence-based recommendations regarding the anticoagulation in patients with DVT and PE. Please note that
Clinical Guideline for Anticoagulation in VTE These clinical guidelines are intended to provide evidence-based recommendations regarding the anticoagulation in patients with DVT and PE. Please note that
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE. Single Technology Appraisal (STA)
 Thank you for agreeing to give us a statement on your organisation s view of the technology and the way it should be used in the NHS. Healthcare professionals can provide a unique perspective on the technology
Thank you for agreeing to give us a statement on your organisation s view of the technology and the way it should be used in the NHS. Healthcare professionals can provide a unique perspective on the technology
DVT - initial management NSCCG
 Background information Information resources for patients and carers Updates to this care map Synonyms Below knee DVT and bleeding risks Patient with confirmed DVT Scan confirms superficial thrombophlebitis
Background information Information resources for patients and carers Updates to this care map Synonyms Below knee DVT and bleeding risks Patient with confirmed DVT Scan confirms superficial thrombophlebitis
Cancer Associated Thrombosis: six months and beyond. Farzana Haque Hull York Medical School
 Cancer Associated Thrombosis: six months and beyond Farzana Haque Hull York Medical School Disclosure I have no disclosure The Challenge of Anticoagulation in Patients with Venous Thromboembolism and Cancer
Cancer Associated Thrombosis: six months and beyond Farzana Haque Hull York Medical School Disclosure I have no disclosure The Challenge of Anticoagulation in Patients with Venous Thromboembolism and Cancer
Venous Thromboembolism. Prevention
 Venous Thromboembolism Prevention August 2010 Venous Thromboembloism Prevention 1 1 Expected Practice Assess all patients upon admission to the ICU for risk factors of venous thromboembolism (VTE) and
Venous Thromboembolism Prevention August 2010 Venous Thromboembloism Prevention 1 1 Expected Practice Assess all patients upon admission to the ICU for risk factors of venous thromboembolism (VTE) and
Non commercial use only. The treatment of venous thromboembolism with new oral anticoagulants. Background
 Italian Journal of Medicine 2013; volume 7(s8):29-35 The treatment of venous thromboembolism with new oral anticoagulants Davide Imberti AUSL Piacenza, Italy ABSTRACT Traditional anticoagulants, such as
Italian Journal of Medicine 2013; volume 7(s8):29-35 The treatment of venous thromboembolism with new oral anticoagulants Davide Imberti AUSL Piacenza, Italy ABSTRACT Traditional anticoagulants, such as
Results from RE-COVER RE-COVER II RE-MEDY RE-SONATE EXECUTIVE SUMMARY
 Assessment of the safety and efficacy of dabigatran etexilate (Pradaxa ) in the treatment of deep vein thrombosis (DVT) and pulmonary embolism (PE) and the prevention of recurrent DVT and PE Results from
Assessment of the safety and efficacy of dabigatran etexilate (Pradaxa ) in the treatment of deep vein thrombosis (DVT) and pulmonary embolism (PE) and the prevention of recurrent DVT and PE Results from
Thromboembolism and cancer: New practices. Marc Carrier
 Thromboembolism and cancer: New practices Marc Carrier Marc Carrier Research Support/P.I. Employee Consultant Major Stockholder Speakers Bureau Honoraria Scientific Advisory Board Leo Pharma, BMS No relevant
Thromboembolism and cancer: New practices Marc Carrier Marc Carrier Research Support/P.I. Employee Consultant Major Stockholder Speakers Bureau Honoraria Scientific Advisory Board Leo Pharma, BMS No relevant
Prophylaxie primaire sur le patient ambulatoire. Marc Carrier
 Prophylaxie primaire sur le patient ambulatoire Marc Carrier Marc Carrier In compliance with COI policy, SSVQ requires the following disclosures to the session audience: Research Support/P.I. Employee
Prophylaxie primaire sur le patient ambulatoire Marc Carrier Marc Carrier In compliance with COI policy, SSVQ requires the following disclosures to the session audience: Research Support/P.I. Employee
Obesity, renal failure, HIT: which anticoagulant to use?
 Obesity, renal failure, HIT: which anticoagulant to use? Mark Crowther with thanks to Dr David Garcia and others. This Photo by Unknown Author is licensed under CC BY-SA 1 2 Drug choices The DOACs have
Obesity, renal failure, HIT: which anticoagulant to use? Mark Crowther with thanks to Dr David Garcia and others. This Photo by Unknown Author is licensed under CC BY-SA 1 2 Drug choices The DOACs have
Clinical practice guidelines for prophylaxis of venous thomboembolism in cancer patients
 618 Theme Issue Article Clinical practice guidelines for prophylaxis of venous thomboembolism in cancer patients Corinne Frere 1 ; Dominique Farge 2 1 Assistance Publique Hôpitaux de Marseille, Timone
618 Theme Issue Article Clinical practice guidelines for prophylaxis of venous thomboembolism in cancer patients Corinne Frere 1 ; Dominique Farge 2 1 Assistance Publique Hôpitaux de Marseille, Timone
Prevention and treatment of venous thromboembolic disease
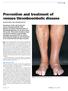 REVIEW Prevention and treatment of venous thromboembolic disease SUSAN McNEILL AND CATHERINE BAGOT Awareness of the risk factors for venous thromboembolic (VTE) disease and timely administration of thromboprophylaxis
REVIEW Prevention and treatment of venous thromboembolic disease SUSAN McNEILL AND CATHERINE BAGOT Awareness of the risk factors for venous thromboembolic (VTE) disease and timely administration of thromboprophylaxis
Objectives. Venous Thromboembolism (VTE) Prophylaxis. Case VTE WHY DO IT? Question: Who Is At Risk?
 Objectives Venous Thromboembolism (VTE) Prophylaxis Rishi Garg, MD Department of Medicine Identify patients at risk for VTE Options for VTE prophylaxis Current Recommendations (based on The Seventh ACCP
Objectives Venous Thromboembolism (VTE) Prophylaxis Rishi Garg, MD Department of Medicine Identify patients at risk for VTE Options for VTE prophylaxis Current Recommendations (based on The Seventh ACCP
Canadian Society of Internal Medicine Annual Meeting 2016 Montreal, QC
 Canadian Society of Internal Medicine Annual Meeting 2016 Montreal, QC 1 st workshop: update to VTE guidelines in 2016 2 nd workshop: VTE controversies + new horizons André Roussin MD, FRCP, CSPQ CHUM
Canadian Society of Internal Medicine Annual Meeting 2016 Montreal, QC 1 st workshop: update to VTE guidelines in 2016 2 nd workshop: VTE controversies + new horizons André Roussin MD, FRCP, CSPQ CHUM
Prevention and management of deep venous thrombosis (DVT) John Fletcher Wound Care Association of New South Wales
 Prevention and management of deep venous thrombosis (DVT) John Fletcher Wound Care Association of New South Wales Merimbula, 6 th November 2010 University of Sydney Department of Surgery Westmead Hospital
Prevention and management of deep venous thrombosis (DVT) John Fletcher Wound Care Association of New South Wales Merimbula, 6 th November 2010 University of Sydney Department of Surgery Westmead Hospital
PULMONARY EMBOLISM (PE): DIAGNOSIS AND TREATMENT
 PULMONARY EMBOLISM (PE): DIAGNOSIS AND TREATMENT OBJECTIVE: To provide a diagnostic algorithm and treatment options for patients with acute pulmonary embolism (PE). BACKGROUND: Venous thromboembolism (VTE)
PULMONARY EMBOLISM (PE): DIAGNOSIS AND TREATMENT OBJECTIVE: To provide a diagnostic algorithm and treatment options for patients with acute pulmonary embolism (PE). BACKGROUND: Venous thromboembolism (VTE)
NOTE: The first appearance of terms in bold in the body of this document (except titles) are defined terms please refer to the Definitions section.
 TITLE VENOUS THROMBOEMBOLISM PROPHYLAXIS SCOPE Provincial Acute and Sub-Acute Care Facilities APPROVAL AUTHORITY Alberta Health Services Executive Committee SPONSOR Vice President, Quality and Chief Medical
TITLE VENOUS THROMBOEMBOLISM PROPHYLAXIS SCOPE Provincial Acute and Sub-Acute Care Facilities APPROVAL AUTHORITY Alberta Health Services Executive Committee SPONSOR Vice President, Quality and Chief Medical
RISK FACTORS. Cancer type. Cancer stage
 CANCER ASSOCIATED THROMBOSIS RISK FACTORS The link between cancer and thrombosis is well established, with malignancy recognised as the most important individual risk factor for venous thromboembolism
CANCER ASSOCIATED THROMBOSIS RISK FACTORS The link between cancer and thrombosis is well established, with malignancy recognised as the most important individual risk factor for venous thromboembolism
Updates in Anticoagulation for Atrial Fibrillation and Venous Thromboembolism
 Disclosures Updates in Anticoagulation for Atrial Fibrillation and Venous Thromboembolism No financial conflicts of interest Member of the ABIM Focused- Practice in Hospital Medicine Self Examination Process
Disclosures Updates in Anticoagulation for Atrial Fibrillation and Venous Thromboembolism No financial conflicts of interest Member of the ABIM Focused- Practice in Hospital Medicine Self Examination Process
Is There a Role for Prophylaxis in Cancer Patients During Therapy?
 Victor F. Tapson, MD, FCCP, FRCP Professor of Medicine Director, Center for Pulmonary Vascular Disease Division of Pulmonary and Critical Care Duke University Medical Center Durham, N.C. USA Is There a
Victor F. Tapson, MD, FCCP, FRCP Professor of Medicine Director, Center for Pulmonary Vascular Disease Division of Pulmonary and Critical Care Duke University Medical Center Durham, N.C. USA Is There a
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE. Single Technology Appraisal (STA)
 Thank you for agreeing to give us a statement on your organisation s view of the technology and the way it should be used in the NHS. Healthcare professionals can provide a unique perspective on the technology
Thank you for agreeing to give us a statement on your organisation s view of the technology and the way it should be used in the NHS. Healthcare professionals can provide a unique perspective on the technology
Fatal P.E. Historic 1-2% Current %
 Dr. (Prof.) Anil Arora MS (Ortho) DNB (Ortho) Dip SIROT (USA) FAPOA (Korea), FIGOF (Germany), FJOA (Japan) Commonwealth Fellow Joint Replacement (Royal National Orthopaedic Hospital, London, UK) Senior
Dr. (Prof.) Anil Arora MS (Ortho) DNB (Ortho) Dip SIROT (USA) FAPOA (Korea), FIGOF (Germany), FJOA (Japan) Commonwealth Fellow Joint Replacement (Royal National Orthopaedic Hospital, London, UK) Senior
VTE Prevention After Hip or Knee Replacement
 This Clinical Resource gives subscribers additional insight related to the Recommendations published in May 2018 ~ Resource #340506 VTE Prevention After Hip or Knee Replacement The American College of
This Clinical Resource gives subscribers additional insight related to the Recommendations published in May 2018 ~ Resource #340506 VTE Prevention After Hip or Knee Replacement The American College of
New Oral Anticoagulants in treatment of VTE, PE DR.AMR HANAFY (LECTURER OF CARDIOLOGY ) ASWAN UNIVERSITY
 New Oral Anticoagulants in treatment of VTE, PE DR.AMR HANAFY (LECTURER OF CARDIOLOGY ) ASWAN UNIVERSITY Fact VTE is deadly! It nibbles after it bites! The 30-day mortality rates for first-time DVT or
New Oral Anticoagulants in treatment of VTE, PE DR.AMR HANAFY (LECTURER OF CARDIOLOGY ) ASWAN UNIVERSITY Fact VTE is deadly! It nibbles after it bites! The 30-day mortality rates for first-time DVT or
UvA-DARE (Digital Academic Repository) Cancer, thrombosis and low-molecular-weight heparins Piccioli, A. Link to publication
 UvA-DARE (Digital Academic Repository) Cancer, thrombosis and low-molecular-weight heparins Piccioli, A. Link to publication Citation for published version (APA): Piccioli, A. (2015). Cancer, thrombosis
UvA-DARE (Digital Academic Repository) Cancer, thrombosis and low-molecular-weight heparins Piccioli, A. Link to publication Citation for published version (APA): Piccioli, A. (2015). Cancer, thrombosis
The risk of venous thromboembolism is four to seven times as
 review article Dan L. Longo, M.D., Editor Prophylaxis against Venous Thromboembolism in Ambulatory Patients with Cancer Jean M. Connors, M.D. The risk of venous thromboembolism is four to seven times as
review article Dan L. Longo, M.D., Editor Prophylaxis against Venous Thromboembolism in Ambulatory Patients with Cancer Jean M. Connors, M.D. The risk of venous thromboembolism is four to seven times as
PREVENTION AND TREATMENT OF VENOUS THROMBOEMBOLISM
 PREVENTION AND TREATMENT OF VENOUS THROMBOEMBOLISM International Consensus Statement 2013 Guidelines According to Scientific Evidence Developed under the auspices of the: Cardiovascular Disease Educational
PREVENTION AND TREATMENT OF VENOUS THROMBOEMBOLISM International Consensus Statement 2013 Guidelines According to Scientific Evidence Developed under the auspices of the: Cardiovascular Disease Educational
DOACs in CAT. Fellow: Shweta Jain, MD Faculty Discussant: David Garcia, MD
 DOACs in CAT Fellow: Shweta Jain, MD Faculty Discussant: David Garcia, MD Case 65 year old post menopausal female Left breast lesion Oct 2015 Biopsy Invasive ductal carcinoma Lumpectomy with SNB- pt1cno
DOACs in CAT Fellow: Shweta Jain, MD Faculty Discussant: David Garcia, MD Case 65 year old post menopausal female Left breast lesion Oct 2015 Biopsy Invasive ductal carcinoma Lumpectomy with SNB- pt1cno
Duration of Anticoagulant Therapy. Linda R. Kelly PharmD, PhC, CACP September 17, 2016
 Duration of Anticoagulant Therapy Linda R. Kelly PharmD, PhC, CACP September 17, 2016 Conflicts of Interest No conflicts of interest to report Objectives At the end of the program participants will be
Duration of Anticoagulant Therapy Linda R. Kelly PharmD, PhC, CACP September 17, 2016 Conflicts of Interest No conflicts of interest to report Objectives At the end of the program participants will be
Venous thromboembolic diseases: diagnosis, management and thrombophilia testing (2012) NICE guideline CG144
 Venous thromboembolic diseases: diagnosis, management and thrombophilia testing (2012) NICE guideline CG144 Appendix A: Summary of new evidence from Summary of evidence from previous year Diagnosis Diagnostic
Venous thromboembolic diseases: diagnosis, management and thrombophilia testing (2012) NICE guideline CG144 Appendix A: Summary of new evidence from Summary of evidence from previous year Diagnosis Diagnostic
Misunderstandings of Venous thromboembolism prophylaxis
 Misunderstandings of Venous thromboembolism prophylaxis Veerendra Chadachan Senior Consultant Dept of General Medicine (Vascular Medicine and Hypertension) Tan Tock Seng Hospital, Singapore Case scenario
Misunderstandings of Venous thromboembolism prophylaxis Veerendra Chadachan Senior Consultant Dept of General Medicine (Vascular Medicine and Hypertension) Tan Tock Seng Hospital, Singapore Case scenario
Individualizing VTE Treatment and Prevention of Recurrence: The Place for Direct Oral Anticoagulants in VTE
 Transcript Details This is a transcript of a continuing medical education (CME) activity accessible on the ReachMD network. Additional media formats for the activity and full activity details (including
Transcript Details This is a transcript of a continuing medical education (CME) activity accessible on the ReachMD network. Additional media formats for the activity and full activity details (including
ADVANCES IN HEMATOLOGY
 ADVANCES IN HEMATOLOGY Current Developments in the Management of Hematologic Disorders Section Editor: Craig M. Kessler, MD Are Direct Acting Oral Anticoagulants Ready for Prime-Time Use in Cancer-Related
ADVANCES IN HEMATOLOGY Current Developments in the Management of Hematologic Disorders Section Editor: Craig M. Kessler, MD Are Direct Acting Oral Anticoagulants Ready for Prime-Time Use in Cancer-Related
Venous Thromboembolism Prophylaxis
 Approved by: Venous Thromboembolism Prophylaxis Vice President and Chief Medical Officer; and Vice President and Chief Operating Officer Corporate Policy & Procedures Manual Number: Date Approved January
Approved by: Venous Thromboembolism Prophylaxis Vice President and Chief Medical Officer; and Vice President and Chief Operating Officer Corporate Policy & Procedures Manual Number: Date Approved January
10/8/2012. Disclosures. Making Sense of AT9: Review of the 2012 ACCP Antithrombotic Guidelines. Goals and Objectives. Outline
 Disclosures Making Sense of AT9: Review of the 2012 ACCP Antithrombotic Guidelines No relevant conflicts of interest related to the topic presented. Cyndy Brocklebank, PharmD, CDE Chronic Disease Management
Disclosures Making Sense of AT9: Review of the 2012 ACCP Antithrombotic Guidelines No relevant conflicts of interest related to the topic presented. Cyndy Brocklebank, PharmD, CDE Chronic Disease Management
Abstract. Background. Methods
 Abstract Background Malignancy is a major risk factor for developing venous thromboembolism (VTE), and thus cancer patients are at high risk for deep vein thrombosis (DVT) and/or pulmonary embolism (PE).
Abstract Background Malignancy is a major risk factor for developing venous thromboembolism (VTE), and thus cancer patients are at high risk for deep vein thrombosis (DVT) and/or pulmonary embolism (PE).
Management of Cancer Associated VTE
 Management of Cancer Associated VTE Jean M. Connors, MD 2017 Master Class Course Anticoagulation Management Services BWH/DFCI Hemostatic Antithrombotic Stewardship BWH Assistant Professor of Medicine HMS
Management of Cancer Associated VTE Jean M. Connors, MD 2017 Master Class Course Anticoagulation Management Services BWH/DFCI Hemostatic Antithrombotic Stewardship BWH Assistant Professor of Medicine HMS
What s new with DOACs? Defining place in therapy for edoxaban &
 What s new with DOACs? Defining place in therapy for edoxaban & Use of DOACs in cardioversion Caitlin M. Gibson, PharmD, BCPS Assistant Professor, Department of Pharmacotherapy University of North Texas
What s new with DOACs? Defining place in therapy for edoxaban & Use of DOACs in cardioversion Caitlin M. Gibson, PharmD, BCPS Assistant Professor, Department of Pharmacotherapy University of North Texas
With All the New Drugs, This is How I Treat Acute DVT and Superficial Phlebitis
 BRIGHAM AND WOMEN S HOSPITAL With All the New Drugs, This is How I Treat Acute DVT and Superficial Phlebitis Gregory Piazza, MD, MS Division of Cardiovascular Medicine Brigham and Women s Hospital April
BRIGHAM AND WOMEN S HOSPITAL With All the New Drugs, This is How I Treat Acute DVT and Superficial Phlebitis Gregory Piazza, MD, MS Division of Cardiovascular Medicine Brigham and Women s Hospital April
4.7 Algorithm for the Peri-operative Management of Anticoagulants and Antiplatelet agents in Adult patients
 4.7 Algorithm for the Peri-operative Management of Anticoagulants and Antiplatelet agents in Adult patients Assess Thrombosis risk: baseline risk in an individual patient plus additional thrombotic risk
4.7 Algorithm for the Peri-operative Management of Anticoagulants and Antiplatelet agents in Adult patients Assess Thrombosis risk: baseline risk in an individual patient plus additional thrombotic risk
Anticoagulation in Special populations. Ng Heng Joo Department of Haematology Singapore General Hospital
 Anticoagulation in Special populations Ng Heng Joo Department of Haematology Singapore General Hospital roymatheson.com Objectives Safer anticoagulation for The elderly Chronic kidney disease Obese patients
Anticoagulation in Special populations Ng Heng Joo Department of Haematology Singapore General Hospital roymatheson.com Objectives Safer anticoagulation for The elderly Chronic kidney disease Obese patients
EXTENDING VTE PROPHYLAXIS IN ACUTELY ILL MEDICAL PATIENTS
 EXTENDING VTE PROPHYLAXIS IN ACUTELY ILL MEDICAL PATIENTS Samuel Z. Goldhaber, MD Director, VTE Research Group Cardiovascular Division Brigham and Women s Hospital Professor of Medicine Harvard Medical
EXTENDING VTE PROPHYLAXIS IN ACUTELY ILL MEDICAL PATIENTS Samuel Z. Goldhaber, MD Director, VTE Research Group Cardiovascular Division Brigham and Women s Hospital Professor of Medicine Harvard Medical
Update on the Management of Cancer Associated VTE
 Update on the Management of Cancer Associated VTE Jean M. Connors, MD 2018 Master Class Course Anticoagulation Management Services BWH/DFCI Hemostatic Antithrombotic Stewardship BWH Associate Professor
Update on the Management of Cancer Associated VTE Jean M. Connors, MD 2018 Master Class Course Anticoagulation Management Services BWH/DFCI Hemostatic Antithrombotic Stewardship BWH Associate Professor
PREVENTION AND TREATMENT OF VENOUS THROMBOEMBOLISM
 PREVENTION AND TREATMENT OF VENOUS THROMBOEMBOLISM International Consensus Statement 2013 Guidelines According to Scientific Evidence Developed under the auspices of the: Cardiovascular Disease Educational
PREVENTION AND TREATMENT OF VENOUS THROMBOEMBOLISM International Consensus Statement 2013 Guidelines According to Scientific Evidence Developed under the auspices of the: Cardiovascular Disease Educational
USE OF DIRECT ORAL ANTICOAGULANTS IN OBESITY
 SDSHP ANNUAL MEETING CLINICAL PEARLS APRIL 7 TH, 2017 USE OF DIRECT ORAL ANTICOAGULANTS IN OBESITY STEFFANIE DANLEY, PHARM D, BCPS, CACP DISCLOSURE I have had no financial relationship over the past 12
SDSHP ANNUAL MEETING CLINICAL PEARLS APRIL 7 TH, 2017 USE OF DIRECT ORAL ANTICOAGULANTS IN OBESITY STEFFANIE DANLEY, PHARM D, BCPS, CACP DISCLOSURE I have had no financial relationship over the past 12
A Review of the Role of Non-Vitamin K Oral Anticoagulants in the Acute and Long-Term Treatment of Venous Thromboembolism
 Cardiol Ther (2018) 7:1 13 https://doi.org/10.1007/s40119-018-0107-0 REVIEW A Review of the Role of Non-Vitamin K Oral Anticoagulants in the Acute and Long-Term Treatment of Venous Thromboembolism Andrew
Cardiol Ther (2018) 7:1 13 https://doi.org/10.1007/s40119-018-0107-0 REVIEW A Review of the Role of Non-Vitamin K Oral Anticoagulants in the Acute and Long-Term Treatment of Venous Thromboembolism Andrew
DOAC for VTE. Direct Oral Anticoagulants Clint Shedd DNP, FNP-BC Emory University
 DOAC for VTE Direct Oral Anticoagulants Clint Shedd DNP, FNP-BC Emory University No disclosures Direct Oral Anticoagulants Understand VTE and the ACCP s position on it List the DOACs available in the US
DOAC for VTE Direct Oral Anticoagulants Clint Shedd DNP, FNP-BC Emory University No disclosures Direct Oral Anticoagulants Understand VTE and the ACCP s position on it List the DOACs available in the US
HEPARIN-INDUCED THROMBOCYTOPENIA (HIT)
 HEPARIN-INDUCED THROMBOCYTOPENIA (HIT) OBJECTIVE: To assist clinicians with the investigation and management of suspected and documented heparin-induced thrombocytopenia (HIT). BACKGROUND: HIT is a transient,
HEPARIN-INDUCED THROMBOCYTOPENIA (HIT) OBJECTIVE: To assist clinicians with the investigation and management of suspected and documented heparin-induced thrombocytopenia (HIT). BACKGROUND: HIT is a transient,
Prevention and management of venous thromboembolism M. AAPRO
 Prevention and management of venous thromboembolism M. AAPRO Thromboprophylaxisof DVT and PE in AmbulatoryCancerPatients Zurich, February 2017 M. AAPRO Based on a lesson in April 2016 by M. DICATO M.D.,
Prevention and management of venous thromboembolism M. AAPRO Thromboprophylaxisof DVT and PE in AmbulatoryCancerPatients Zurich, February 2017 M. AAPRO Based on a lesson in April 2016 by M. DICATO M.D.,
Rapid Fire-Top Articles You Need to Know
 Rapid Fire-Top Articles You Need to Know TRACY MINICHIELLO, MD CHIEF, ANTICOAGULATION& THROMBOSIS SERVICE- SAN FRANCISCO VAMC PROFESSOR OF MEDICINE UNIVERSITY OF CALIFORNIA, SAN FRANCISCO Financial Disclosures-NONE
Rapid Fire-Top Articles You Need to Know TRACY MINICHIELLO, MD CHIEF, ANTICOAGULATION& THROMBOSIS SERVICE- SAN FRANCISCO VAMC PROFESSOR OF MEDICINE UNIVERSITY OF CALIFORNIA, SAN FRANCISCO Financial Disclosures-NONE
NEW/NOVEL ORAL ANTICOAGULANTS (NOACS): COMPARISON AND FREQUENTLY ASKED QUESTIONS
 NEW/NOVEL ORAL ANTICOAGULANTS (NOACS): COMPARISON AND FREQUENTLY ASKED QUESTIONS OBJECTIVES: To provide a comparison of the new/novel oral anticoagulants (NOACs) currently available in Canada. To address
NEW/NOVEL ORAL ANTICOAGULANTS (NOACS): COMPARISON AND FREQUENTLY ASKED QUESTIONS OBJECTIVES: To provide a comparison of the new/novel oral anticoagulants (NOACs) currently available in Canada. To address
Direct Oral Anticoagulants (DOACs). Dr GM Benson Director NI Haemophilia Comprehensive Care Centre and Thrombosis Unit BHSCT
 Direct Oral Anticoagulants (DOACs). Dr GM Benson Director NI Haemophilia Comprehensive Care Centre and Thrombosis Unit BHSCT OAC WARFARIN Gold standard DABIGATRAN RIVAROXABAN APIXABAN EDOXABAN BETRIXABAN
Direct Oral Anticoagulants (DOACs). Dr GM Benson Director NI Haemophilia Comprehensive Care Centre and Thrombosis Unit BHSCT OAC WARFARIN Gold standard DABIGATRAN RIVAROXABAN APIXABAN EDOXABAN BETRIXABAN
Disclosures. DVT: Diagnosis and Treatment. Questions To Ask. Dr. Susanna Shin - DVT: Diagnosis and Treatment. Acute Venous Thromboembolism (VTE) None
 Disclosures DVT: Diagnosis and Treatment None Susanna Shin, MD, FACS Assistant Professor University of Washington Acute Venous Thromboembolism (VTE) Deep Venous Thrombosis (DVT) Pulmonary Embolism (PE)
Disclosures DVT: Diagnosis and Treatment None Susanna Shin, MD, FACS Assistant Professor University of Washington Acute Venous Thromboembolism (VTE) Deep Venous Thrombosis (DVT) Pulmonary Embolism (PE)
Low Molecular Weight Heparin for Prevention and Treatment of Venous Thromboembolic Disorders
 SURGICAL GRAND ROUNDS March 17 th, 2007 Low Molecular Weight Heparin for Prevention and Treatment of Venous Thromboembolic Disorders Guillermo Escobar, M.D. LMWH vs UFH Jayer s sales pitch: FALSE LMW is
SURGICAL GRAND ROUNDS March 17 th, 2007 Low Molecular Weight Heparin for Prevention and Treatment of Venous Thromboembolic Disorders Guillermo Escobar, M.D. LMWH vs UFH Jayer s sales pitch: FALSE LMW is
Updates in venous thromboembolism. Cecilia Becattini University of Perugia
 Updates in venous thromboembolism Cecilia Becattini University of Perugia News for VTE Diagnosis Treatment the acute phase the agents Pulmonary embolism: diagnosis Vein ultrasonography Meta-analysis 15
Updates in venous thromboembolism Cecilia Becattini University of Perugia News for VTE Diagnosis Treatment the acute phase the agents Pulmonary embolism: diagnosis Vein ultrasonography Meta-analysis 15
Treatment of cancer-associated venous thromboembolism by new oral anticoagulants: a meta-analysis
 Original Article Page of 9 Treatment of cancer-associated venous thromboembolism by new oral anticoagulants: a meta-analysis Satyanarayana R. Vaidya, Sonu Gupta, Santhosh R. Devarapally, Department of
Original Article Page of 9 Treatment of cancer-associated venous thromboembolism by new oral anticoagulants: a meta-analysis Satyanarayana R. Vaidya, Sonu Gupta, Santhosh R. Devarapally, Department of
Direct Oral Anticoagulant Use in Older Adults Brian Skinner, PharmD
 Direct Oral Anticoagulant Use in Older Adults Brian Skinner, PharmD Dr. Skinner serves as an Assistant Professor of Pharmacy Practice at Manchester University, and he is one of three Internal Medicine
Direct Oral Anticoagulant Use in Older Adults Brian Skinner, PharmD Dr. Skinner serves as an Assistant Professor of Pharmacy Practice at Manchester University, and he is one of three Internal Medicine
Review Guideline recommendations for the treatment of established VTE Initial treatment (fi rst 10 days) Panel 1:
 International clinical practice guidelines including guidance for direct oral anticoagulants in the treatment and prophylaxis of venous thromboembolism in patients with cancer Dominique Farge, Henri Bounameaux,
International clinical practice guidelines including guidance for direct oral anticoagulants in the treatment and prophylaxis of venous thromboembolism in patients with cancer Dominique Farge, Henri Bounameaux,
GLIOMA - VENOUS THROMBOEMBOLISM. Miguel Navarro. Hospital Universitario de Salamanca-IBSAL
 GLIOMA - VENOUS THROMBOEMBOLISM Miguel Navarro. Hospital Universitario de Salamanca-IBSAL GLIOMA - VTE GLIOMA - VTE The two string problem Substantial risk for developing VTE Concern antithrombotic agents
GLIOMA - VENOUS THROMBOEMBOLISM Miguel Navarro. Hospital Universitario de Salamanca-IBSAL GLIOMA - VTE GLIOMA - VTE The two string problem Substantial risk for developing VTE Concern antithrombotic agents
Canadian consensus recommendations on the management of venous thromboembolism in patients with cancer. Part 1: prophylaxis
 Curr Oncol, Vol. 22, pp. 133-143; doi: http://dx.doi.org/10.3747/co.22.2586 CANADIAN RECOMMENDATIONS FOR VTE PROPHYLAXIS IN CANCER PRACTICE GUIDELINE Canadian consensus recommendations on the management
Curr Oncol, Vol. 22, pp. 133-143; doi: http://dx.doi.org/10.3747/co.22.2586 CANADIAN RECOMMENDATIONS FOR VTE PROPHYLAXIS IN CANCER PRACTICE GUIDELINE Canadian consensus recommendations on the management
10 Key Things the Vascular Community Should Know about the DOACs Heather Gornik, MD, RVT, RPVI
 10 Key Things the Vascular Community Should Know about the DOACs Heather Gornik, MD, RVT, RPVI Cleveland Clinic Heart and Vascular Institute Heather L. Gornik, MD has the following relationships to disclose:
10 Key Things the Vascular Community Should Know about the DOACs Heather Gornik, MD, RVT, RPVI Cleveland Clinic Heart and Vascular Institute Heather L. Gornik, MD has the following relationships to disclose:
General. Recommendations. Guideline Title. Bibliographic Source(s) Guideline Status. Major Recommendations
 General Guideline Title Prevention of deep vein thrombosis and pulmonary embolism. Bibliographic Source(s) American College of Obstetricians and Gynecologists (ACOG). Prevention of deep vein thrombosis
General Guideline Title Prevention of deep vein thrombosis and pulmonary embolism. Bibliographic Source(s) American College of Obstetricians and Gynecologists (ACOG). Prevention of deep vein thrombosis
Bath, Philip M.W. and England, Timothy J. (2009) Thighlength compression stockings and DVT after stroke. Lancet. ISSN (In Press)
 Bath, Philip M.W. and England, Timothy J. (2009) Thighlength compression stockings and DVT after stroke. Lancet. ISSN 0140-6736 (In Press) Access from the University of Nottingham repository: http://eprints.nottingham.ac.uk/1087/1/lancet_clots_1_20090522_4.pdf
Bath, Philip M.W. and England, Timothy J. (2009) Thighlength compression stockings and DVT after stroke. Lancet. ISSN 0140-6736 (In Press) Access from the University of Nottingham repository: http://eprints.nottingham.ac.uk/1087/1/lancet_clots_1_20090522_4.pdf
Clinical challenges in patients with cancer-associated thrombosis: Canadian expert consensus recommendations
 ABSTRACT Venous thromboembolism is a common complication in cancer patients, and thromboembolism is the second most common cause of death after cancer progression. A number of clinical practice guidelines
ABSTRACT Venous thromboembolism is a common complication in cancer patients, and thromboembolism is the second most common cause of death after cancer progression. A number of clinical practice guidelines
Cancer associated thrombosis. 17 th November 2016 Simon Noble Clinical Professor Palliative Medicine Cardiff University Wales, UK
 Cancer associated thrombosis 17 th November 2016 Simon Noble Clinical Professor Palliative Medicine Cardiff University Wales, UK Today What is VTE? How does CAT differ? Initial anticoagulation Anticoagulation
Cancer associated thrombosis 17 th November 2016 Simon Noble Clinical Professor Palliative Medicine Cardiff University Wales, UK Today What is VTE? How does CAT differ? Initial anticoagulation Anticoagulation
ROLE OF LOW MOLECULAR WEIGHT HEPARIN IN THE AGE OF DIRECT ORAL ANTICOAGULANTS
 ROLE OF LOW MOLECULAR WEIGHT HEPARIN IN THE AGE OF DIRECT ORAL ANTICOAGULANTS Chee Yen Lin Consultant Haematologist NCIS Haematology National University Hospital Singapore Nomenclature direct oral anticoagulant
ROLE OF LOW MOLECULAR WEIGHT HEPARIN IN THE AGE OF DIRECT ORAL ANTICOAGULANTS Chee Yen Lin Consultant Haematologist NCIS Haematology National University Hospital Singapore Nomenclature direct oral anticoagulant
Thromboprophylaxis for medical patients with cancer: what do the guidelines say?
 ith Thromboprophylaxis for medical patients with cancer: what do the guidelines say? Practice Points Hospitalized medically ill cancer patients should receive parenteral thromboprophylaxis for the duration
ith Thromboprophylaxis for medical patients with cancer: what do the guidelines say? Practice Points Hospitalized medically ill cancer patients should receive parenteral thromboprophylaxis for the duration
Venous Thromboembolism National Hospital Inpatient Quality Measures
 Venous Thromboembolism National Hospital Inpatient Quality Measures Presentation Overview Review venous thromboembolism as a new mandatory measure set Outline measures with exclusions and documentation
Venous Thromboembolism National Hospital Inpatient Quality Measures Presentation Overview Review venous thromboembolism as a new mandatory measure set Outline measures with exclusions and documentation
Handbook for Venous Thromboembolism
 Handbook for Venous Thromboembolism Gregory Piazza Benjamin Hohlfelder Samuel Z. Goldhaber Handbook for Venous Thromboembolism Gregory Piazza Cardiovascular Division Harvard Medical School Brigham and
Handbook for Venous Thromboembolism Gregory Piazza Benjamin Hohlfelder Samuel Z. Goldhaber Handbook for Venous Thromboembolism Gregory Piazza Cardiovascular Division Harvard Medical School Brigham and
Novel oral anticoagulants in the treatment of cancer patients
 Novel oral anticoagulants in the treatment of cancer patients Renata Biernacka, MD 1, Tomasz Lewandowski, MD PhD 1, Jolanta Andrzejuk, MD 2, Marek Szmyd, MD 1 1 Department of Chemotherapy, Oncology Clinic,
Novel oral anticoagulants in the treatment of cancer patients Renata Biernacka, MD 1, Tomasz Lewandowski, MD PhD 1, Jolanta Andrzejuk, MD 2, Marek Szmyd, MD 1 1 Department of Chemotherapy, Oncology Clinic,
Updates in Diagnosis & Management of VTE
 Updates in Diagnosis & Management of VTE Financial Disclosures-NONE TRACY MINICHIELLO, MD CHIEF, ANTICOAGULATION& THROMBOSIS SERVICE- SAN FRANCISCO VAMC PROFESSOR OF MEDICINE UNIVERSITY OF CALIFORNIA,
Updates in Diagnosis & Management of VTE Financial Disclosures-NONE TRACY MINICHIELLO, MD CHIEF, ANTICOAGULATION& THROMBOSIS SERVICE- SAN FRANCISCO VAMC PROFESSOR OF MEDICINE UNIVERSITY OF CALIFORNIA,
DVT Pathophysiology and Prophylaxis in Medically Hospitalized Patients. David Liff MD Oklahoma Heart Institute Vascular Center
 DVT Pathophysiology and Prophylaxis in Medically Hospitalized Patients David Liff MD Oklahoma Heart Institute Vascular Center Overview Pathophysiology of DVT Epidemiology and risk factors for DVT in the
DVT Pathophysiology and Prophylaxis in Medically Hospitalized Patients David Liff MD Oklahoma Heart Institute Vascular Center Overview Pathophysiology of DVT Epidemiology and risk factors for DVT in the
Consensus Statement for Management of Anticoagulants and Antiplatelet drugs in Patients with Hip Fracture
 Consensus Statement for Management of Anticoagulants and Antiplatelet drugs in Patients with Hip Fracture Patients with hip fractures should be operated on within 36 hours of presentation wherever possible.
Consensus Statement for Management of Anticoagulants and Antiplatelet drugs in Patients with Hip Fracture Patients with hip fractures should be operated on within 36 hours of presentation wherever possible.
A Review of Direct-Acting Oral Anticoagulants (DOACs) and Their Use in Special Populations
 A Review of Direct-Acting Oral Anticoagulants (DOACs) and Their Use in Special Populations Allison Bernard, PharmD PGY2 Ambulatory Care Resident University of Iowa Hospitals and Clinics October 25 th,
A Review of Direct-Acting Oral Anticoagulants (DOACs) and Their Use in Special Populations Allison Bernard, PharmD PGY2 Ambulatory Care Resident University of Iowa Hospitals and Clinics October 25 th,
