Interview with Prof. Guido Costamagna
|
|
|
- Morgan Floyd
- 5 years ago
- Views:
Transcription
1 Interview with Prof. Guido Costamagna Extraxts of his curriculum vitae: Full Professor of Surgery, Università Cattolica del Sacro Cuore, Rome, Italy Director, Digestive Endoscopy Unit, Policlinico A. Gemelli, Università Cattolica S. Cuore, Rome, Italy. Scientific Director, European Endoscopy Training Centre, Università Cattolica del Sacro Cuore, Rome, Italy. Director of the Institute of Clinical Surgery, Università Cattolica del Sacro Cuore, Rome, Italy. His main areas of interest amongst many others: Endoscopy and Gastroenterology Clinical Trials Research and development in Digestive Endoscopy Questions: Professor Dr. J. F. Riemann (JFR), Executive Chairman of LebensBlicke, Foundation for the Prevention of Colorectal Cancer ( JFR: Dear Guido, you are a well know specialist in endoscopy and also very much engaged in the evaluation of colon capsule endoscopy. Colon cancer is one of the most common cancers in Europe. With g-fobt and colonoscopy there are two well investigated procedures for detection and even prevention of this cancer at hand, which in many countries are even paid by insurance companies. What is the rationale to introduce colon capsule endoscopy (CCE) in this context? GC: Colon capsule endoscopy (CCE) is not intended to replace colonoscopy, but it should be considered a complementary test to colonoscopy in selected patients. Colonoscopy is the gold standard for colon evaluation and it will remain the reference endoscopic procedure for the next years. However, there are challenges associated with colonoscopy being perceived as an invasive procedure, usually associated by patients fear of discomfort and inconvenience as well as psychological inhibition. For these reasons, colorectal cancer screening uptake is still low, especially when compared with the high rate of attendance of other cancer screening programs (breast, cervical and prostate). In order to overcome the limitations related to the invasiveness of colonoscopy, a non-invasive test may be proposed in patients who deserve colonoscopy for CRC prevention and refuse colonoscopy. Among non-invasive tests, however, imaging tests might be preferred over nonimaging tests (i.e. fecal tests), because of the ability to detect pre-neoplastic and non-neoplastic conditions that may be regarded as clinically useful (e.g. vascular malformations). For these reasons, in my opinion colonoscopy is the
2 - 2 - procedure of choice for CRC prevention, however, when a non-invasive imaging test is required, CCE should be considered as one of the preferable options, which was also the opinion by almost 30 worldwide GI experts who did the voting during the ESGE guideline meeting for CCE. JFR: Can you tell us a little more in detail how the non invasive CCE works? Is the preparation similar to that for colonoscopy? GC: Now the second generation of colon capsule system is available. The new system includes new developments of colon capsule (Colon Capsule 2 CCE-2), data recorder and software for video processing and viewing. The new CCE-2 is 11.6 x 31.5 mm in size. It has 2 imagers with a much wider angle of view that has been increased to 172 degrees for each imager, allowing nearly degrees coverage of the colon. Furthermore, in order to enhance colon visualization and to save battery energy, the capsule is equipped with an adaptive frame rate. This represents a major progress over the previous version. CCE-2 captures 35 images per second when in motion and 4 images per second when it is virtually stationary. This advanced system for the control of capsule image rate is the result of a bidirectional communication between CCE-2 and the new data recorder that, besides storing the images transmitted from the capsule, also controls the capsule image rate in real time, analysing the capsule images. To further save battery energy, as well as to allow automatic identification of the small bowel, CCE-2, instead of going into a sleep mode, continues to work at a low rate of only 14 images per minute until small bowel images are automatically detected, then it switches into the adaptive frame rate. At this point, the data recorder buzzes and vibrates and displays instructions on its liquid crystal diode (LCD) screen to alert the patient to continue the preparation protocol, assisting and guiding the physician and the patient through the procedure. The software (Rapid 8 software) for advanced video processing and viewing has also been implemented. Regarding the regimen of preparation, a bowel preparation is specifically designed for colon capsule endoscopy. Compared to colonoscopy, the cleansing procedure is the same. Similarly to colonoscopy, a clean colon is necessary to accurately explore the colonic mucosa, CCE being unable to suck or wash the mucosa. However, differently from conventional endoscopy, an additional bowel preparation after swallowing of the capsule is required to promote capsule propulsion, since the colon has only few spontaneous longitudinal contractions per day. For this reason, one or two boosters of sodium phosphate (NaP) have been added to the usual preparation of polyethylene glycol solution (PEG) adopted for colonoscopy (4L of splitdose).
3 - 3 - The main role of NaP booster is to accelerate CCE transit through both the small and large bowel within the limited operating-time of the CCE battery (i.e. 10 hours). Differently from initial studies in which high doses of NaP were used, low doses of NaP are now included in the regimen of preparation for CCE-2, in order to reduce the risk of adverse events. In detail, one booster of 30mL with 1 L of water is required when the capsule had entered the small bowel, and one of ml with 0.5L water 3 hours later if the capsule had not been egested by that time which is the case in about 50% of patients. JFR: Meanwhile quite a few data are available concerning the efficacy of this technique. What are the most important results? GC: To date, two studies evaluated the CCE-2 in comparison to colonoscopy that was considered the gold standard: an Israeli multicenter trial and a European multicenter trial. Both trials have used a similar methodology: colonoscopy was independently performed within 8-10 hours after capsule ingestion and colon capsule endoscopy was prospectively compared with colonoscopy for the detection of colorectal polyps and masses in a cohort of patients scheduled for colonoscopy and having known or suspected colonic disease. Overall more than 200 patients were enrolled. Patients were considered having a significant finding when polyps 6mm and 10 mm or masses were detected. Perpatient CCE-2 sensitivity for polyps 6 mm ranged between 84-89% and 10 mm was 88% in both trials. Specificity for polyps 6mm ranged between 64-76% and 89-94% for polyps 10 mm. The relatively low specificity for 6-mm polyps observed in these series was explained by a substantial rate of falsepositive polyps because of size mismatch. In these series no missed cancers were reported: overall 4 cancers were detected by CCE-2 and were confirmed at colonoscopy. During the last DDW, the results from a large US multicenter trial were presented. In this study, the second generation CCE system was compared in a sixteen-center study with colonoscopy in a cohort of patients classified as average risk for colorectal cancer screening. Differently from the Israeli and European trials, the recorded capsule video was reviewed by central readers and blinded colonoscopy was performed 4-6 weeks post CCE procedure. The colonoscopist unblinded the capsule report at the end of the procedure and repeated the procedure when the capsule reported false positive lesion 6 mm. A total of 884 subjects were enrolled. The capsule sensitivity for detecting subjects with adenomas 6 mm and 10 mm was 88% and 92%, and the specificity was 82% and 95%, respectively.
4 - 4 - Then we have the interesting chapter of incomplete colonoscopy. In such cases several radiologic methods have traditionally been used, but more recently, capsule endoscopy was also shown to be accurate. A French study published by Pioche in Endoscopy in 2012 and more recently papers published by Alarcon in Clin Gastroenterol & Hepatol (2013) and Triantafyllou in GIE (2013), homogeneously demonstrated that CCE is a feasible and safe tool for colon mucosa visualization in patients with incomplete colonoscopy being able to complete the colon visualization in the majority of the patients. However no comparative trials between CCE and radiology are available, to date. For this reason, in my Unit in Rome, we designed a study with the aim to compare CCE and CTC in a prospective cohort of patients with incomplete colonoscopy. 100 patients were enrolled. CCE and CTC were both able to achieve complete colonic evaluation in 98% of cases. In a per-patient analysis for polyps 6 mm, the CCE diagnostic yield was 2-folds higher than CTC (CCE detected 24 patients with at least 1 polyp 6 mm and CTC 12 patients); and this difference was statistically significant (p=0.02). Also when considering larger polyps ( 10 mm), CCE diagnostic yield was higher (5% for CCE and 3% for CTC), but the difference was not statistically significant (p=0.549). Both the tests show a high positive predictive value for polyps 6 mm and 10 mm being 96% and 86%, and 83% and 100% for CCE and CTC, respectively. The results of this trial confirmed that both colon capsule endoscopy and CT colonography were of comparable efficacy in completing colon evaluation after incomplete colonoscopy; however, the overall diagnostic yield of CCE was superior to CT colonography. JFR: Is there any decision making so far at this point how to proceed with diminutive polyps? GC: Most colon polyps discovered at screening are diminutive, with negligible risk of harboring advanced features (high grade dysplasia, villous component or malignancy). Moreover, 40% of diminutive colon polyps are hyperplastic rather than adenomatous. Diminutive lesions identified by a non-invasive test may also be missed by the post-test colonoscopy, because of the imperfect sensitivity of the latter for diminutive lesions. For this reason, diminutive polyps do not represent a primary target for non-invasive imaging tests. By extrapolating data from CT colonography studies that modelled the impact of colonoscopy or continued surveillance for diminutive polyps discovered at CT colonography, it can be concluded that referral for removal of diminutive lesions found at CCE might carry an unjustified burden of costs and complications relative to minimal gain in clinical efficacy. Moreover, studies on the second generation of CCE only provide accuracy data for >6 mm lesions, its specificity for diminutive lesions being largely unknown.
5 - 5 - The only exception for post-cce referral for diminutive polyps is the simultaneous presence of at least 3 of these lesions. Polyp multiplicity has appeared as a strong predictive factor of subsequent advanced neoplasia development in post-polypectomy follow-up studies. JFR: What evidence level is necessary in order to be recommended as an important tool for colon cancer detection and its precursor lesions? Are the current data sufficient (German S3 guidelines for Colorectal Cancer do not recommend the procedure due to missing RCT`s)? GC: To date, there is data available with average risk patients (FDA registration file) showing good accuracy, however, we do not have trials that evaluated the potential role of the capsule in colorectal cancer screening programs. This means that we do not know if the capsule might have a role in CRC screening programs and, if yes, which could be its role. Only few and preliminary data are available on the possible adherence to CCE in a screening setting. We only have a cost-effectiveness analysis published by Hassan in Endoscopy that compared the CCE with colonoscopy in a screening setting. Although CCE was not a cost-effective alternative when assuming an equal adherence, it became an efficient option when assuming that adherence to CCE was higher compared to colonoscopy for CRC screening, a feature which has to be evaluated to support the assumption that CCE may play a role in CRC screening. JFR: To get evidence-based data more large clinical trials are necessary. Would a prospective randomized controlled trial i(immunological)-fobt versus CCE makes sense? Would you recommend other trials? GC: Two big trials are planned in Europe. Both will enroll patients attending in organized screening programs. It is an Italian multicenter trial (CCANDY trial- Colon Capsule Advanced Neoplasia Diagnostic Yield) aimed to assess the accuracy (sensitivity and specificity) and the positive and negative predictive value (PPV and NPV) of CCE, compared to conventional colonoscopy, in detecting CRC and advanced adenomas, among subjects with a positive FIT, attending in an organized population screening program. We started with the enrollment of patients a couple of weeks ago and at the end 400 patients will be enrolled. In the Netherlands, in Rotterdam, Prof. Kuipers designed the so-
6 - 6 - called ORCA (Population COlon cancer screening by Capsule endoscopy) trial aimed to determine the uptake and diagnostic yield of primary population screening for CRC by means of CCE. They will enroll asymptomatic volunteers aged years by means an active invitation process until 1000 participants have been included. Soon, an Irish prospective comparison study will be published in Endoscopy. CCE was compared to colonoscopy in patients attending within the second round of FIT bowel screening. The Authors aimed to assess the sensitivity, specificity, and negative and positive predictive value of CCE compared to colonoscopy in an FIT positive colorectal cancer screening cohort. CCE was demonstrated to be a safe and effective means of detecting cancer and polyps in a positive FIT screening cohort, suggesting that CCE would be a useful filter test in this situation, reducing the number of colonoscopies performed by 71%. I think that the results of this trial and the forthcoming results of the Italian and Dutch trials will clarify the potential role of colon capsule in the screening programs. JFR: Are there anyhow established indications for CCE now? Could CCE be seen as a possible filter for those who do not need colonoscopy? GC: The ESGE guidelines recommend the use of CCE in average risk subjects, while patients at high risk for CRC, because of alarm symptoms or signs, a family or a personal history of CRC should be referred to colonoscopy. However, in patients not compliant or unable to undergo colonoscopy, the use of CCE could be discussed with the patient. CCE is a feasible and safe tool for colon mucosa visualization in patients with a previous incomplete colonoscopy. To date, there is insufficient data to support the use of CCE in the diagnostic work-up or in the surveillance of patients with suspected or known inflammatory bowel disease. Based on preliminary data, CCE may be useful to monitor inflammation in ulcerative colitis, which may help to guide therapy JFR: What are the limitations, what kind of complications are possible and how often do they occur? In brief: how safe is CCE? GC: Concerning the limitations, one of the most relevant is the fact that the colon capsule is only diagnostic. The other problem is that we cannot control the movements of colon
7 - 7 - capsule: this means that we cannot control the transit of the capsule in the colon and make it homogeneous, but also we cannot ensure that the capsule will complete the colonic exploration. In my opinion these are limitations that will be overcome in the next few years with some technological developments. Contraindications for CCE are similar to those of small-bowel capsule endoscopy. The use of sodium phosphate (NaP) as a booster should be avoided in patients at increased risk of NaP toxicity. The risk of capsule retention with CCE is very low. CCE has consistently been shown to be a safe procedure: no major complication has been reported in about 3000 procedures. Therefore, we can say that CCE is a safe procedure. JFR: Are there data about the acceptance of the procedure available? GC: The group of Prof. Rösch published an interesting trial in BMC Gastroenterol in It is a prospective study performed to evaluate whether adding capsule colonoscopy to the endoscopic screening options increases uptake. Invitation letters were sent to 2150 persons above the age of 55 insured with a German medical insurance company in an area in Germany (Rinteln, Lower Saxony) with a baseline spontaneous annual screening colonoscopy uptake of 1 %. Both capsule or conventional colonoscopy were offered. Interested persons were given information about the two screening options by local gastroenterologists and examinations were then performed according to screenees' final choice. Interestingly, the option of capsule endoscopy led to a fourfold increase of screening uptake (4.2 % vs. 1 %, p < 0.001). Despite similar age distribution in both sex groups, uptake in men was significantly higher (5.6 % vs. 2.8 %, p = 002). The present study suggests that offering the option of capsule colonoscopy increases uptake of endoscopic colorectal cancer screening. On the other hand, the new system offers the advantage of performing the colon capsule endoscopy out-of the clinic. No colorectal imaging test may be performed on an out-of-clinic basis. This represents a major drawback compared with fecal tests. Because colon capsule endoscopy (CCE) automatically detects small bowel mucosa, it has the potential to become the first colorectal imaging test to be performed out-of-clinic. Sam Adler from Jerusalem last year published in Surgic Endosc a study aimed to evaluate the feasibility and efficiency of CCE when offered as an out-of-clinic procedure. He demonstrated that as an out-of-clinic procedure, CCE is
8 - 8 - feasible and easily performed. This was a feasibility study and it did not demonstrate that an out-of the clinic colon capsule endoscopy increases the acceptability. However, it might be argued that a home-based procedure may be associated with better acceptability and potentially with increased adherence to Colorectal cancer screening. JFR: Where do you see the place of CCE in the near future having in mind that this technique will not be paid by insurance companies and it is very expensive? GC: I think that the place of CCE comes out from the evidence in the literature. The main evidence relates to incomplete colonoscopy and CCE seems to be a good test in this group of patients, being able to complete the vast majority of the previous incomplete colonoscopy and showing a higher diagnostic yield when compared to CTC. This is also good for the Endoscopist who fails to complete a colonoscopy. In fact these patients can be managed internally in the Endoscopy Unit, without the need of referring them to Radiologists JFR: Your personal opinion: will CCE become a major player as a noninvasive and presumably safe technique in the broad range of low threshold offers for colon cancer detection? And what further requirements are absolutely necessary for it? GC: In my opinion CCE will play a role in the future. One of the most intriguing aspect is its potential role as a filter to select patients who need colonoscopy. In this vision, the filter test if effective would select patients with neoplasia only for invasive standard colonoscopy and reduce the number of negative colonoscopies and therefore the burden on endoscopy services, staff and patients. Before this, our priority is to simplify the regimen of preparation in terms of amount of liquids and type of agents used; and to increase the evidence supporting this new endoscopic technology. On the other hand, in order to spread the use of this technology, costs are still an issue. The reduction of costs is out of our business and should be one of the priorities of the producer company. JFR: Dear Guido, thank you very much for this exciting and interesting interview.
Capsule endoscopy screening. Carlo SENORE
 Capsule endoscopy screening Carlo SENORE Possible conflicts of interest Medtronics provides CCE-2 devices to conduct a multicenter independent, non profit study, aimed to assess CCE-2 diagnostic accuracy
Capsule endoscopy screening Carlo SENORE Possible conflicts of interest Medtronics provides CCE-2 devices to conduct a multicenter independent, non profit study, aimed to assess CCE-2 diagnostic accuracy
Guidelines for Colonoscopy Surveillance After Screening and Polypectomy: A Consensus Update by the US Multi-Society Task Force on Colorectal Cancer
 Guidelines for Colonoscopy Surveillance After Screening and Polypectomy: A Consensus Update by the US Multi-Society Task Force on Colorectal Cancer David A. Lieberman, 1 Douglas K. Rex, 2 Sidney J. Winawer,
Guidelines for Colonoscopy Surveillance After Screening and Polypectomy: A Consensus Update by the US Multi-Society Task Force on Colorectal Cancer David A. Lieberman, 1 Douglas K. Rex, 2 Sidney J. Winawer,
Colorectal Cancer Screening: A Clinical Update
 11:05 11:45am Colorectal Cancer Screening: A Clinical Update SPEAKER Kevin A. Ghassemi, MD Presenter Disclosure Information The following relationships exist related to this presentation: Kevin A. Ghassemi,
11:05 11:45am Colorectal Cancer Screening: A Clinical Update SPEAKER Kevin A. Ghassemi, MD Presenter Disclosure Information The following relationships exist related to this presentation: Kevin A. Ghassemi,
Alberta Colorectal Cancer Screening Program (ACRCSP) Post Polypectomy Surveillance Guidelines
 Alberta Colorectal Cancer Screening Program (ACRCSP) Post Polypectomy Surveillance Guidelines June 2013 ACRCSP Post Polypectomy Surveillance Guidelines - 2 TABLE OF CONTENTS Background... 3 Terms, Definitions
Alberta Colorectal Cancer Screening Program (ACRCSP) Post Polypectomy Surveillance Guidelines June 2013 ACRCSP Post Polypectomy Surveillance Guidelines - 2 TABLE OF CONTENTS Background... 3 Terms, Definitions
Colon capsule endoscopy as possible filter test for colonoscopy selection in a screening population with positive fecal immunology
 Original article 473 Colon capsule endoscopy as possible filter test for colonoscopy selection in a screening population with positive fecal immunology Authors Grainne Holleran 1, 2, Ronan Leen 1,2, Colm
Original article 473 Colon capsule endoscopy as possible filter test for colonoscopy selection in a screening population with positive fecal immunology Authors Grainne Holleran 1, 2, Ronan Leen 1,2, Colm
Colon Cancer Screening. Layth Al-Jashaami, MD GI Fellow, PGY 4
 Colon Cancer Screening Layth Al-Jashaami, MD GI Fellow, PGY 4 -Colorectal cancer (CRC) is a common and lethal cancer. -It has the highest incidence among GI cancers in the US, estimated to be newly diagnosed
Colon Cancer Screening Layth Al-Jashaami, MD GI Fellow, PGY 4 -Colorectal cancer (CRC) is a common and lethal cancer. -It has the highest incidence among GI cancers in the US, estimated to be newly diagnosed
Advice Statement. Advice Statement November Advice for NHSScotland. Why is SHTG looking at this topic?
 Advice Statement 014-18 November 2018 Advice Statement Colon capsule endoscopy (CCE-2) for the detection of colorectal polyps and cancer in adults with signs or symptoms of colorectal cancer or at increased
Advice Statement 014-18 November 2018 Advice Statement Colon capsule endoscopy (CCE-2) for the detection of colorectal polyps and cancer in adults with signs or symptoms of colorectal cancer or at increased
Early detection and screening for colorectal neoplasia
 Early detection and screening for colorectal neoplasia Robert S. Bresalier Department of Gastroenterology, Hepatology and Nutrition. The University of Texas. MD Anderson Cancer Center. Houston, Texas U.S.A.
Early detection and screening for colorectal neoplasia Robert S. Bresalier Department of Gastroenterology, Hepatology and Nutrition. The University of Texas. MD Anderson Cancer Center. Houston, Texas U.S.A.
Quality ID #343: Screening Colonoscopy Adenoma Detection Rate National Quality Strategy Domain: Effective Clinical Care
 Quality ID #343: Screening Colonoscopy Adenoma Detection Rate National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE TYPE: Outcome DESCRIPTION:
Quality ID #343: Screening Colonoscopy Adenoma Detection Rate National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE TYPE: Outcome DESCRIPTION:
CRC Risk Factors. U.S. Adherence Rates Cancer Screening. Genetic Model of Colorectal Cancer. Epidemiology and Clinical Consequences of CRC
 10:45 11:45 am Guide to Colorectal Cancer Screening SPEAKER Howard Manten M.D. Presenter Disclosure Information The following relationships exist related to this presentation: Howard Manten MD: No financial
10:45 11:45 am Guide to Colorectal Cancer Screening SPEAKER Howard Manten M.D. Presenter Disclosure Information The following relationships exist related to this presentation: Howard Manten MD: No financial
The Dutch bowel cancer screening program Relevant lessions for Ontario
 The Dutch bowel cancer screening program Relevant lessions for Ontario Ernst J Kuipers Erasmus MC University Medical Center Rotterdam - The Netherlands 1 Ismar Boas (1858 1938) Colorectal cancer screening
The Dutch bowel cancer screening program Relevant lessions for Ontario Ernst J Kuipers Erasmus MC University Medical Center Rotterdam - The Netherlands 1 Ismar Boas (1858 1938) Colorectal cancer screening
Citation for published version (APA): Wijkerslooth de Weerdesteyn, T. R. (2013). Population screening for colorectal cancer by colonoscopy
 UvA-DARE (Digital Academic Repository) Population screening for colorectal cancer by colonoscopy de Wijkerslooth, T.R. Link to publication Citation for published version (APA): Wijkerslooth de Weerdesteyn,
UvA-DARE (Digital Academic Repository) Population screening for colorectal cancer by colonoscopy de Wijkerslooth, T.R. Link to publication Citation for published version (APA): Wijkerslooth de Weerdesteyn,
Colorectal cancer screening
 26 Colorectal cancer screening BETHAN GRAF AND JOHN MARTIN Colorectal cancer is theoretically a preventable disease and is ideally suited to a population screening programme, as there is a long premalignant
26 Colorectal cancer screening BETHAN GRAF AND JOHN MARTIN Colorectal cancer is theoretically a preventable disease and is ideally suited to a population screening programme, as there is a long premalignant
Screening & Surveillance Guidelines
 Chapter 2 Screening & Surveillance Guidelines I. Eligibility Coloradans ages 50 and older (average risk) or under 50 at elevated risk for colon cancer (personal or family history) that meet the following
Chapter 2 Screening & Surveillance Guidelines I. Eligibility Coloradans ages 50 and older (average risk) or under 50 at elevated risk for colon cancer (personal or family history) that meet the following
The New Grade A: USPSTF Updated Colorectal Cancer Screening Guidelines, What does it all mean?
 The New Grade A: USPSTF Updated Colorectal Cancer Screening Guidelines, What does it all mean? Robert A. Smith, PhD Cancer Control, Department of Prevention and Early Detection American Cancer Society
The New Grade A: USPSTF Updated Colorectal Cancer Screening Guidelines, What does it all mean? Robert A. Smith, PhD Cancer Control, Department of Prevention and Early Detection American Cancer Society
SCREENING FOR BOWEL CANCER USING FLEXIBLE SIGMOIDOSCOPY REVIEW APPRAISAL CRITERIA FOR THE UK NATIONAL SCREENING COMMITTEE
 SCREENING FOR BOWEL CANCER USING FLEXIBLE SIGMOIDOSCOPY REVIEW APPRAISAL CRITERIA FOR THE UK NATIONAL SCREENING COMMITTEE The Condition 1. The condition should be an important health problem Colorectal
SCREENING FOR BOWEL CANCER USING FLEXIBLE SIGMOIDOSCOPY REVIEW APPRAISAL CRITERIA FOR THE UK NATIONAL SCREENING COMMITTEE The Condition 1. The condition should be an important health problem Colorectal
Colon Cancer Screening and Surveillance. Louis V. Antignano, M.D. Wilson Gastroenterology October 11, 2011
 Colon Cancer Screening and Surveillance Louis V. Antignano, M.D. Wilson Gastroenterology October 11, 2011 Colorectal Cancer Preventable cancer Number 2 cancer killer in the USA Often curable if detected
Colon Cancer Screening and Surveillance Louis V. Antignano, M.D. Wilson Gastroenterology October 11, 2011 Colorectal Cancer Preventable cancer Number 2 cancer killer in the USA Often curable if detected
removal of adenomatous polyps detects important effectively as follow-up colonoscopy after both constitute a low-risk Patients with 1 or 2
 Supplementary Table 1. Study Characteristics Author, yr Design Winawer et al., 6 1993 National Polyp Study Jorgensen et al., 9 1995 Funen Adenoma Follow-up Study USA Multi-center, RCT for timing of surveillance
Supplementary Table 1. Study Characteristics Author, yr Design Winawer et al., 6 1993 National Polyp Study Jorgensen et al., 9 1995 Funen Adenoma Follow-up Study USA Multi-center, RCT for timing of surveillance
Digestive Health Southwest Endoscopy 2016 Quality Report
 Digestive Health 2016 Quality Report Our 2016 our quality and value management program focused on one primary area of interest: Performing high quality colonoscopy High quality Colonoscopy We selected
Digestive Health 2016 Quality Report Our 2016 our quality and value management program focused on one primary area of interest: Performing high quality colonoscopy High quality Colonoscopy We selected
Dr Alasdair Patrick Gastroenterologist
 Dr Alasdair Patrick Gastroenterologist Bowel Cancer screening Dr Alasdair Patrick Gastroenterologist MacMurray Gastroenterology Case- Patient for Screening? 62 year old lady Father diagnosed with advanced
Dr Alasdair Patrick Gastroenterologist Bowel Cancer screening Dr Alasdair Patrick Gastroenterologist MacMurray Gastroenterology Case- Patient for Screening? 62 year old lady Father diagnosed with advanced
Japan Journal of Medicine 2018; 1(4): doi: /jjm
 Jpn J Med 2018,1:4 191 Review Colon capsule endoscopy for colorectal cancer screening in patients with the positive fecal occult blood test (FOBT) in Japan Konosuke Nakaji * Endoscopy Center, Aishinkai
Jpn J Med 2018,1:4 191 Review Colon capsule endoscopy for colorectal cancer screening in patients with the positive fecal occult blood test (FOBT) in Japan Konosuke Nakaji * Endoscopy Center, Aishinkai
2019 COLLECTION TYPE: MIPS CLINICAL QUALITY MEASURES (CQMS) MEASURE TYPE: Outcome High Priority
 Quality ID #343: Screening Colonoscopy Adenoma Detection Rate National Quality Strategy Domain: Effective Clinical Care Meaningful Measure Area: Preventive Care 2019 COLLECTION TYPE: MIPS CLINICAL QUALITY
Quality ID #343: Screening Colonoscopy Adenoma Detection Rate National Quality Strategy Domain: Effective Clinical Care Meaningful Measure Area: Preventive Care 2019 COLLECTION TYPE: MIPS CLINICAL QUALITY
The choice of methods for Colorectal Cancer Screening; The Dutch experience
 The choice of methods for Colorectal Cancer Screening; The Dutch experience Monique van Leerdam, Gastroenterologist, NKI-AVL, Amsterdam The Netherlands Colorectal cancer CRC 2 nd cause of cancer related
The choice of methods for Colorectal Cancer Screening; The Dutch experience Monique van Leerdam, Gastroenterologist, NKI-AVL, Amsterdam The Netherlands Colorectal cancer CRC 2 nd cause of cancer related
What Questions Should You Ask?
 ? Your Doctor Has Ordered a Colonoscopy. What Questions Should You sk? From the merican College of Gastroenterology www.acg.gi.org Normal colon Is the doctor performing your colonoscopy a Gastroenterologist?
? Your Doctor Has Ordered a Colonoscopy. What Questions Should You sk? From the merican College of Gastroenterology www.acg.gi.org Normal colon Is the doctor performing your colonoscopy a Gastroenterologist?
CLINICAL PRACTICE GUIDELINE FOR COLORECTAL CANCER SCREENING
 CLINICAL PRACTICE GUIDELINE FOR COLORECTAL CANCER SCREENING This guideline is designed to assist practitioners by providing the framework for colorectal cancer (CRC) screening, and is not intended to replace
CLINICAL PRACTICE GUIDELINE FOR COLORECTAL CANCER SCREENING This guideline is designed to assist practitioners by providing the framework for colorectal cancer (CRC) screening, and is not intended to replace
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE SCOPE
 NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE 1 Guideline title SCOPE Colorectal cancer: colonoscopic surveillance for prevention of colorectal cancer in patients with ulcerative colitis, Crohn
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE 1 Guideline title SCOPE Colorectal cancer: colonoscopic surveillance for prevention of colorectal cancer in patients with ulcerative colitis, Crohn
Study population The study population comprised a hypothetical cohort of 50-year-olds at average risk of CRC.
 Colon cancer prevention in Italy: cost-effectiveness analysis with CT colonography and endoscopy Hassan C, Zullo A, Laghi A, Reitano I, Taggi F, Cerro P, Iafrate F, Giustini M, Winn S, Morini S Record
Colon cancer prevention in Italy: cost-effectiveness analysis with CT colonography and endoscopy Hassan C, Zullo A, Laghi A, Reitano I, Taggi F, Cerro P, Iafrate F, Giustini M, Winn S, Morini S Record
Endoscopic Corner CASE 1. Kimtrakool S Aniwan S Linlawan S Muangpaisarn P Sallapant S Rerknimitr R
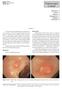 170 Endoscopic Corner Kimtrakool S Aniwan S Linlawan S Muangpaisarn P Sallapant S Rerknimitr R CASE 1 A 54-year-old woman underwent a colorectal cancer screening. Her fecal immunochemical test was positive.
170 Endoscopic Corner Kimtrakool S Aniwan S Linlawan S Muangpaisarn P Sallapant S Rerknimitr R CASE 1 A 54-year-old woman underwent a colorectal cancer screening. Her fecal immunochemical test was positive.
Accepted Manuscript. En bloc resection for mm polyps to reduce post-colonoscopy cancer and surveillance. C. Hassan, M. Rutter, A.
 Accepted Manuscript En bloc resection for 10-20 mm polyps to reduce post-colonoscopy cancer and surveillance C. Hassan, M. Rutter, A. Repici PII: S1542-3565(19)30412-4 DOI: https://doi.org/10.1016/j.cgh.2019.04.022
Accepted Manuscript En bloc resection for 10-20 mm polyps to reduce post-colonoscopy cancer and surveillance C. Hassan, M. Rutter, A. Repici PII: S1542-3565(19)30412-4 DOI: https://doi.org/10.1016/j.cgh.2019.04.022
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: capsule_endoscopy_wireless 5/2002 5/2016 5/2017 11/2016 Description of Procedure or Service Wireless capsule
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: capsule_endoscopy_wireless 5/2002 5/2016 5/2017 11/2016 Description of Procedure or Service Wireless capsule
Quality ID #439: Age Appropriate Screening Colonoscopy National Quality Strategy Domain: Efficiency and Cost Reduction
 Quality ID #439: Age Appropriate Screening Colonoscopy National Quality Strategy Domain: Efficiency and Cost Reduction 2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE TYPE: Efficiency DESCRIPTION:
Quality ID #439: Age Appropriate Screening Colonoscopy National Quality Strategy Domain: Efficiency and Cost Reduction 2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE TYPE: Efficiency DESCRIPTION:
Dysplasia 4/19/2017. How do I practice Chromoendoscopy for Surveillance of Colitis? SCENIC: Polypoid Dysplasia in UC. Background
 SCENIC: Polypoid in UC Definition How do I practice for Surveillance of Colitis? Themos Dassopoulos, M.D. Director, BSW Center for IBD Themistocles.Dassopoulos@BSWHealth.org Tel: 469-800-7189 Cell: 314-686-2623
SCENIC: Polypoid in UC Definition How do I practice for Surveillance of Colitis? Themos Dassopoulos, M.D. Director, BSW Center for IBD Themistocles.Dassopoulos@BSWHealth.org Tel: 469-800-7189 Cell: 314-686-2623
Improving Outcomes in Colorectal Cancer: The Science of Screening. Colorectal Cancer (CRC)
 Improving Outcomes in Colorectal Cancer: The Science of Screening Tennessee Primary Care Association October 23, 2014 Durado Brooks, MD, MPH Director, Prostate and Colorectal Cancers Colorectal Cancer
Improving Outcomes in Colorectal Cancer: The Science of Screening Tennessee Primary Care Association October 23, 2014 Durado Brooks, MD, MPH Director, Prostate and Colorectal Cancers Colorectal Cancer
Colon Screening in 2014 Offering Patients a Choice. Clark A Harrison MD The Nevada Colon Cancer Partnership
 Colon Screening in 2014 Offering Patients a Choice Clark A Harrison MD The Nevada Colon Cancer Partnership Objectives 1. Understand the incidence and mortality rates for CRC in the US. 2. Understand risk
Colon Screening in 2014 Offering Patients a Choice Clark A Harrison MD The Nevada Colon Cancer Partnership Objectives 1. Understand the incidence and mortality rates for CRC in the US. 2. Understand risk
Colorectal Cancer Screening
 Recommendations from the U.S. Multi-Society Task Force on Colorectal Cancer Colorectal Cancer Screening Rex DK, Boland CR, Dominitz JA, Giardiello FM, Johnson DA, Kaltenbach T, Levin TR, Lieberman D, Robertson
Recommendations from the U.S. Multi-Society Task Force on Colorectal Cancer Colorectal Cancer Screening Rex DK, Boland CR, Dominitz JA, Giardiello FM, Johnson DA, Kaltenbach T, Levin TR, Lieberman D, Robertson
Colorectal Cancer Screening and Surveillance
 1 Colorectal Cancer Screening and Surveillance Jeffrey Lee MD, MAS Assistant Clinical Professor of Medicine University of California, San Francisco jeff.lee@ucsf.edu Objectives Review the various colorectal
1 Colorectal Cancer Screening and Surveillance Jeffrey Lee MD, MAS Assistant Clinical Professor of Medicine University of California, San Francisco jeff.lee@ucsf.edu Objectives Review the various colorectal
11/9/2015 OUTLINE. Quality Indicators for the Doctor Performing Screening Colonoscopy: What you should expect from your Endoscopist
 Quality Indicators for the Doctor Performing Screening Colonoscopy: What you should expect from your Endoscopist Anil K Sharma MD FACP Professor of Clinical Medicine, University of Rochester Chief of Gastroenterology,
Quality Indicators for the Doctor Performing Screening Colonoscopy: What you should expect from your Endoscopist Anil K Sharma MD FACP Professor of Clinical Medicine, University of Rochester Chief of Gastroenterology,
Corporate Medical Policy
 Corporate Medical Policy Chromoendoscopy as an Adjunct to Colonoscopy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: chromoendoscopy_as_an_adjunct_to_colonoscopy 7/2012 11/2017
Corporate Medical Policy Chromoendoscopy as an Adjunct to Colonoscopy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: chromoendoscopy_as_an_adjunct_to_colonoscopy 7/2012 11/2017
Colorectal Cancer Screening
 Colorectal Cancer Screening An Integrated Care Pathway of the Collaborative Care Network Subject Matter Expert: Kevin Wolov, DO Pathway Custodian: Pat Czapp, MD First, a Friendly Reminder... This Integrated
Colorectal Cancer Screening An Integrated Care Pathway of the Collaborative Care Network Subject Matter Expert: Kevin Wolov, DO Pathway Custodian: Pat Czapp, MD First, a Friendly Reminder... This Integrated
Improving you ADR. Robert Enns Colonoscopy Education Day October 2018
 Improving you ADR Robert Enns Colonoscopy Education Day October 2018 ADR Applying to CSP Assume 50% ADR in FIT positive patients Out of 40 patients only 20 will have polyps Out of 20 likely 15 will be
Improving you ADR Robert Enns Colonoscopy Education Day October 2018 ADR Applying to CSP Assume 50% ADR in FIT positive patients Out of 40 patients only 20 will have polyps Out of 20 likely 15 will be
Colon capsule endoscopy: European Society of Gastrointestinal Endoscopy (ESGE) Guideline
 Colon capsule endoscopy: European Society of Gastrointestinal Endoscopy (ESGE) Guideline Authors C. Spada 1, C. Hassan 1, J. P. Galmiche 2, H. Neuhaus 3, J. M. Dumonceau 4, S. Adler 5, O. Epstein 6,G.Gay
Colon capsule endoscopy: European Society of Gastrointestinal Endoscopy (ESGE) Guideline Authors C. Spada 1, C. Hassan 1, J. P. Galmiche 2, H. Neuhaus 3, J. M. Dumonceau 4, S. Adler 5, O. Epstein 6,G.Gay
Page 1. Is the Risk This High? Dysplasia in the IBD Patient. Dysplasia in the Non IBD Patient. Increased Risk of CRC in Ulcerative Colitis
 Screening for Colorectal Neoplasia in Inflammatory Bowel Disease Francis A. Farraye MD, MSc Clinical Director, Section of Gastroenterology Co-Director, Center for Digestive Disorders Boston Medical Center
Screening for Colorectal Neoplasia in Inflammatory Bowel Disease Francis A. Farraye MD, MSc Clinical Director, Section of Gastroenterology Co-Director, Center for Digestive Disorders Boston Medical Center
Objectives. Definitions. Colorectal Cancer Screening 5/8/2018. Payam Afshar, MS, MD Kaiser Permanente, San Diego. Colorectal cancer background
 Colorectal Cancer Screening Payam Afshar, MS, MD Kaiser Permanente, San Diego Objectives Colorectal cancer background Colorectal cancer screening populations Colorectal cancer screening modalities Colonoscopy
Colorectal Cancer Screening Payam Afshar, MS, MD Kaiser Permanente, San Diego Objectives Colorectal cancer background Colorectal cancer screening populations Colorectal cancer screening modalities Colonoscopy
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Virtual Colonoscopy / CT Colonography Page 1 of 19 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Virtual Colonoscopy / CT Colonography Professional Institutional
Virtual Colonoscopy / CT Colonography Page 1 of 19 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Virtual Colonoscopy / CT Colonography Professional Institutional
Latest Endoscopic Guidelines for FAP, HNPCC, IBD, and the General Population
 Latest Endoscopic Guidelines for FAP, HNPCC, IBD, and the General Population David T. Rubin, M.D. Assistant Professor of Medicine Inflammatory Bowel Disease Center MacLean Center for Clinical Medical Ethics
Latest Endoscopic Guidelines for FAP, HNPCC, IBD, and the General Population David T. Rubin, M.D. Assistant Professor of Medicine Inflammatory Bowel Disease Center MacLean Center for Clinical Medical Ethics
Kenneth D. Chi, MD Medical Director, GI Lab Advocate Lutheran General Hospital Center for Digestive Health May 7, 2016
 Kenneth D. Chi, MD Medical Director, GI Lab Advocate Lutheran General Hospital Center for Digestive Health May 7, 2016 Why have Quality Indicators? Pre-procedure Quality Indicators Intra-procedure Quality
Kenneth D. Chi, MD Medical Director, GI Lab Advocate Lutheran General Hospital Center for Digestive Health May 7, 2016 Why have Quality Indicators? Pre-procedure Quality Indicators Intra-procedure Quality
Same-day colon capsule endoscopy is a viable means to assess unexplored colonic segments after incomplete colonoscopy in selected patients
 Original Article Same-day colon capsule endoscopy is a viable means to assess unexplored colonic segments after incomplete colonoscopy in selected patients United European Gastroenterology Journal 2018,
Original Article Same-day colon capsule endoscopy is a viable means to assess unexplored colonic segments after incomplete colonoscopy in selected patients United European Gastroenterology Journal 2018,
Screening di Popolazione. del Cancro Colorettale. C. Hassan
 Screening di Popolazione del Cancro Colorettale C. Hassan The Present 1. Organized screening program - Active invitation (less inequality) - Rapid coverage of the target population (2 yrs) - Quality Assurance
Screening di Popolazione del Cancro Colorettale C. Hassan The Present 1. Organized screening program - Active invitation (less inequality) - Rapid coverage of the target population (2 yrs) - Quality Assurance
Colorectal cancer screening A puzzle of tests and strategies
 Colorectal cancer screening A puzzle of tests and strategies A. Van Gossum, MD, PhD Head of the Clinic of Intestinal Diseases and Nutritional Support Department of Gastroenterology Hôpital Erasme ULB -
Colorectal cancer screening A puzzle of tests and strategies A. Van Gossum, MD, PhD Head of the Clinic of Intestinal Diseases and Nutritional Support Department of Gastroenterology Hôpital Erasme ULB -
Colon Cancer Detection by Rendezvous Colonoscopy : Successful Removal of Stuck Colon Capsule by Conventional Colonoscopy
 19 Colon Cancer Detection by Rendezvous Colonoscopy : Successful Removal of Stuck Colon Capsule by Conventional Colonoscopy István Rácz Márta Jánoki Hussam Saleh Department of Gastroenterology, Petz Aladár
19 Colon Cancer Detection by Rendezvous Colonoscopy : Successful Removal of Stuck Colon Capsule by Conventional Colonoscopy István Rácz Márta Jánoki Hussam Saleh Department of Gastroenterology, Petz Aladár
French National Colon Capsule Endoscopy
 French National Colon Capsule Endoscopy Observatory (ONECC) Evaluation and first lessons Jean-Christophe Saurin On behalf of the comité scientifique de l Oservatoire national de l endoscopie par capsule
French National Colon Capsule Endoscopy Observatory (ONECC) Evaluation and first lessons Jean-Christophe Saurin On behalf of the comité scientifique de l Oservatoire national de l endoscopie par capsule
Measure #343: Screening Colonoscopy Adenoma Detection Rate National Quality Strategy Domain: Effective Clincal Care
 Measure #343: Screening Colonoscopy Adenoma Detection Rate National Quality Strategy Domain: Effective Clincal Care 2016 PQRS OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY DESCRIPTION: The percentage
Measure #343: Screening Colonoscopy Adenoma Detection Rate National Quality Strategy Domain: Effective Clincal Care 2016 PQRS OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY DESCRIPTION: The percentage
[Type here] CG [Type here]
![[Type here] CG [Type here] [Type here] CG [Type here]](/thumbs/78/77374363.jpg) TRTs: - Physicians: 5:20 - Patients & Advocacy: 11:52 - Company: 4:32 Exact Sciences FDA Broll SUGGESTED B- ROLL & GRAPHICS Opening (white background, black lettering) SOUNDBITES FDA Approves Cologuard
TRTs: - Physicians: 5:20 - Patients & Advocacy: 11:52 - Company: 4:32 Exact Sciences FDA Broll SUGGESTED B- ROLL & GRAPHICS Opening (white background, black lettering) SOUNDBITES FDA Approves Cologuard
Colon Capsule Endoscopy: Where Are We and Where Are We Going
 REVIEW Clin Endosc 2016;49:449-453 http://dx.doi.org/10.5946/ce.2016.095 Print ISSN 2234-2400 On-line ISSN 2234-2443 Open Access Colon Capsule Endoscopy: Where Are We and Where Are We Going Yoo Min Han
REVIEW Clin Endosc 2016;49:449-453 http://dx.doi.org/10.5946/ce.2016.095 Print ISSN 2234-2400 On-line ISSN 2234-2443 Open Access Colon Capsule Endoscopy: Where Are We and Where Are We Going Yoo Min Han
Cancer Control in the Workplace: A Corporate Standard
 Healthy Outcomes Conference, Whistler, BC March 31 April 2, 2009 Cancer Control in the Workplace: A Corporate Standard Dr. Alain Sotto, Hon.BSc, MD, CCFP(EM), FCBOM Chief Physician, Ontario Power Generation
Healthy Outcomes Conference, Whistler, BC March 31 April 2, 2009 Cancer Control in the Workplace: A Corporate Standard Dr. Alain Sotto, Hon.BSc, MD, CCFP(EM), FCBOM Chief Physician, Ontario Power Generation
Second-generation colon capsule endoscopy compared with colonoscopy
 Second-generation colon capsule endoscopy compared with colonoscopy Cristiano Spada, MD, Cesare Hassan, MD, PhD, Miguel Munoz-Navas, MD, PhD, Horst Neuhaus, MD, Jacques Deviere, MD, PhD, Paul Fockens,
Second-generation colon capsule endoscopy compared with colonoscopy Cristiano Spada, MD, Cesare Hassan, MD, PhD, Miguel Munoz-Navas, MD, PhD, Horst Neuhaus, MD, Jacques Deviere, MD, PhD, Paul Fockens,
Measuring performance and quality indicators of CRC screening
 Measuring performance and quality indicators of CRC screening Ondřej MÁJEK Institute of Biostatistics and Analyses, Masaryk University Institute of Biostatistics and Analyses, Masaryk University, Brno
Measuring performance and quality indicators of CRC screening Ondřej MÁJEK Institute of Biostatistics and Analyses, Masaryk University Institute of Biostatistics and Analyses, Masaryk University, Brno
ASGE and AGA Issue Consensus Statement on Surveillance and Management of Dysplasia in Patients With Inflammatory Bowel Disease
 ASGE and AGA Issue Consensus Statement on Surveillance and Management of Dysplasia in Patients With Inflammatory Bowel Disease DOWNERS GROVE, Ill., (March 5, 2015) The American Society for Gastrointestinal
ASGE and AGA Issue Consensus Statement on Surveillance and Management of Dysplasia in Patients With Inflammatory Bowel Disease DOWNERS GROVE, Ill., (March 5, 2015) The American Society for Gastrointestinal
Chromoendoscopy as an Adjunct to Colonoscopy
 Chromoendoscopy as an Adjunct to Colonoscopy Policy Number: 2.01.84 Last Review: 1/2018 Origination: 7/2017 Next Review: 7/2018 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will not provide
Chromoendoscopy as an Adjunct to Colonoscopy Policy Number: 2.01.84 Last Review: 1/2018 Origination: 7/2017 Next Review: 7/2018 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will not provide
Technology and Interventions to Improve ADR
 Technology and Interventions to Improve ADR Aasma Shaukat, MD MPH, FACG GI Section Chief, Minneapolis VAMC Associate Professor, University of Minnesota Outline Why is quality important? Fundamentals of
Technology and Interventions to Improve ADR Aasma Shaukat, MD MPH, FACG GI Section Chief, Minneapolis VAMC Associate Professor, University of Minnesota Outline Why is quality important? Fundamentals of
Cologuard Screening for Colorectal Cancer
 Pending Policies - Medicine Cologuard Screening for Colorectal Cancer Print Number: MED208.056 Effective Date: 08-15-2016 Coverage: I.Cologuard stool DNA testing may be considered medically necessary for
Pending Policies - Medicine Cologuard Screening for Colorectal Cancer Print Number: MED208.056 Effective Date: 08-15-2016 Coverage: I.Cologuard stool DNA testing may be considered medically necessary for
The Usefulness of Capsule Endoscopy
 The Usefulness of Capsule Endoscopy David J. Hass, MD, FACG Associate Clinical Professor of Medicine Yale University School of Medicine Gastroenterology Center of Connecticut INDICATIONS FOR USE PillCam
The Usefulness of Capsule Endoscopy David J. Hass, MD, FACG Associate Clinical Professor of Medicine Yale University School of Medicine Gastroenterology Center of Connecticut INDICATIONS FOR USE PillCam
2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY. MEASURE TYPE: Process
 Quality ID #185 (NQF 0659): Colonoscopy Interval for Patients with a History of Adenomatous Polyps Avoidance of Inappropriate Use National Quality Strategy Domain: Communication and Care Coordination 2018
Quality ID #185 (NQF 0659): Colonoscopy Interval for Patients with a History of Adenomatous Polyps Avoidance of Inappropriate Use National Quality Strategy Domain: Communication and Care Coordination 2018
Corporate Presentation Fourth Quarter 2017
 Corporate Presentation Fourth Quarter 2017 November 2017 1 Safe harbor statement This presentation contains forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as
Corporate Presentation Fourth Quarter 2017 November 2017 1 Safe harbor statement This presentation contains forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as
Predict, Resect and discard : Yes we can! (at least in some hands)
 Diminutive polyps : Real time endoscopic histology Predict, Resect and discard : Yes we can! (at least in some hands) Robert Benamouzig Hôpital Avicenne AP-HP & Paris 13 University France Why it is important?
Diminutive polyps : Real time endoscopic histology Predict, Resect and discard : Yes we can! (at least in some hands) Robert Benamouzig Hôpital Avicenne AP-HP & Paris 13 University France Why it is important?
2017 OPTIONS FOR INDIVIDUAL MEASURES: CLAIMS ONLY. MEASURE TYPE: Process
 Measure. #185 (NQF 0659): Colonoscopy Interval for Patients with a History of Adenomatous Polyps Avoidance of Inappropriate Use National Quality Strategy Domain: Communication and Care Coordination 2017
Measure. #185 (NQF 0659): Colonoscopy Interval for Patients with a History of Adenomatous Polyps Avoidance of Inappropriate Use National Quality Strategy Domain: Communication and Care Coordination 2017
Colorectal Cancer Screening Guideline Issue Brief Updated May 30 th, 2018
 Colorectal Cancer Screening Guideline Issue Brief Updated May 30 th, 2018 Issue Summary The American Cancer Society has updated its colorectal screening guideline, which have been published in CA: A Journal
Colorectal Cancer Screening Guideline Issue Brief Updated May 30 th, 2018 Issue Summary The American Cancer Society has updated its colorectal screening guideline, which have been published in CA: A Journal
Pathology in Slovenian CRC screening programme:
 Pathology in Slovenian CRC screening programme: Findings, organisation and quality assurance Snježana Frković Grazio University Medical Center Ljubljana, Slovenia Slovenia s population: 2 million Incidence
Pathology in Slovenian CRC screening programme: Findings, organisation and quality assurance Snježana Frković Grazio University Medical Center Ljubljana, Slovenia Slovenia s population: 2 million Incidence
Northern Ireland Bowel Cancer Screening Programme. Pathways. Version 4 1 st October 2013
 Northern Ireland Bowel Cancer Screening Programme Pathways These changes will be version controlled, led by the Quality Assurance Director for the Programme. Any updated versions will be circulated and
Northern Ireland Bowel Cancer Screening Programme Pathways These changes will be version controlled, led by the Quality Assurance Director for the Programme. Any updated versions will be circulated and
COMPUTED TOMOGRAPHIC COLONOGRAPHY
 MEDICAL POLICY COMPUTED TOMOGRAPHIC COLONOGRAPHY Policy Number: 2013T0320M Effective Date: November 1, 2013 Table of Contents COVERAGE RATIONALE... BACKGROUND... CLINICAL EVIDENCE... U.S. FOOD AND DRUG
MEDICAL POLICY COMPUTED TOMOGRAPHIC COLONOGRAPHY Policy Number: 2013T0320M Effective Date: November 1, 2013 Table of Contents COVERAGE RATIONALE... BACKGROUND... CLINICAL EVIDENCE... U.S. FOOD AND DRUG
2. Describe pros/cons of screening interventions (including colonoscopy, CT colography, fecal tests)
 Learning Objectives 1. Review principles of colon adenoma/cancer biology that permit successful prevention regimes 2. Describe pros/cons of screening interventions (including colonoscopy, CT colography,
Learning Objectives 1. Review principles of colon adenoma/cancer biology that permit successful prevention regimes 2. Describe pros/cons of screening interventions (including colonoscopy, CT colography,
CASE DISCUSSION: The Patient with Dysplasia: Surgery or Active Surveillance? Noa Krugliak Cleveland, MD David T. Rubin, MD
 CASE DISCUSSION: The Patient with Dysplasia: Surgery or Active Surveillance? Noa Krugliak Cleveland, MD David T. Rubin, MD Disclosure Statement NKC: No relevant conflicts to disclose. DTR: No relevant
CASE DISCUSSION: The Patient with Dysplasia: Surgery or Active Surveillance? Noa Krugliak Cleveland, MD David T. Rubin, MD Disclosure Statement NKC: No relevant conflicts to disclose. DTR: No relevant
COLON: Innovations 3 steps, 3 parts..
 COLON: Innovations 3 steps, 3 parts.. Detection: I see an abnormality (usually a polyp) Characterization: Is this abnormality neoplastic? (for example: an adenoma) Treatment: it is neoplastic. Can I treat
COLON: Innovations 3 steps, 3 parts.. Detection: I see an abnormality (usually a polyp) Characterization: Is this abnormality neoplastic? (for example: an adenoma) Treatment: it is neoplastic. Can I treat
Rx Only. Detecting Cancer In Blood.
 Epi procolon is an FDA-approved blood test for colorectal cancer screening for patients who are unwilling or unable to be screened by recommended methods. Rx Only Intended Use, Contraindications, Warnings,
Epi procolon is an FDA-approved blood test for colorectal cancer screening for patients who are unwilling or unable to be screened by recommended methods. Rx Only Intended Use, Contraindications, Warnings,
효과적인대장정결법 김태준 삼성서울병원소화기내과
 효과적인대장정결법 김태준 삼성서울병원소화기내과 부적절한장정결 Efficacy blurring - 긴검사시간 - 낮은맹장도달율 & 선종발견율 Risk intensification - 시술관련합병증증가 Waste of cost - 검사반복 - 내시경의사의 workload 증가 Patient dissatisfaction 장정결에따른선종발견율차이 Multi-centers
효과적인대장정결법 김태준 삼성서울병원소화기내과 부적절한장정결 Efficacy blurring - 긴검사시간 - 낮은맹장도달율 & 선종발견율 Risk intensification - 시술관련합병증증가 Waste of cost - 검사반복 - 내시경의사의 workload 증가 Patient dissatisfaction 장정결에따른선종발견율차이 Multi-centers
FREQUENTLY ASKED QUESTIONS
 FREQUENTLY ASKED QUESTIONS What is CRC? CRC (CRC) is cancer of the large intestine (colon), the lower part of the digestive system. Rectal cancer is cancer of the last several inches of the colon. Together,
FREQUENTLY ASKED QUESTIONS What is CRC? CRC (CRC) is cancer of the large intestine (colon), the lower part of the digestive system. Rectal cancer is cancer of the last several inches of the colon. Together,
NHS Bowel Cancer Screening Programme
 NHS Bowel Cancer Screening Programme Wolverhampton Bowel Cancer Screening Centre Annual Report April 2015 to March 2016 Introduction Bowel cancer is the fourth most common cancer in the UK (2012) accounting
NHS Bowel Cancer Screening Programme Wolverhampton Bowel Cancer Screening Centre Annual Report April 2015 to March 2016 Introduction Bowel cancer is the fourth most common cancer in the UK (2012) accounting
Colorectal Cancer Screening and Risk Assessment Workflow. Documentation Guide for Health Center NextGen Users
 Colorectal Cancer Screening and Risk Assessment Workflow Documentation Guide for Health Center NextGen Users Colorectal Cancer Screening and Risk Assessment Workflow and Documentation Guide for Health
Colorectal Cancer Screening and Risk Assessment Workflow Documentation Guide for Health Center NextGen Users Colorectal Cancer Screening and Risk Assessment Workflow and Documentation Guide for Health
BENEFIT APPLICATION BLUE CARD/NATIONAL ACCOUNT ISSUES
 Medical Policy BCBSA Ref. Policy: 2.01.84 Last Review: 11/15/2018 Effective Date: 11/15/2018 Section: Medicine Related Policies 2.01.87 Confocal Laser Endomicroscopy 6.01.32 Virtual Colonoscopy/Computed
Medical Policy BCBSA Ref. Policy: 2.01.84 Last Review: 11/15/2018 Effective Date: 11/15/2018 Section: Medicine Related Policies 2.01.87 Confocal Laser Endomicroscopy 6.01.32 Virtual Colonoscopy/Computed
Financial Disclosers
 Slide 1 Colorectal Cancer Screening Jason Hemming, MD NESGNA November 15, 2014 Slide 2 Bio Slide 3 Financial Disclosers I have no actual or potential conflict of interest relation to this presentation
Slide 1 Colorectal Cancer Screening Jason Hemming, MD NESGNA November 15, 2014 Slide 2 Bio Slide 3 Financial Disclosers I have no actual or potential conflict of interest relation to this presentation
CTC vs. Colonoscopy for Surveillance After CRC? David Weinberg, MD, MSc June 1, 2018
 CTC vs. Colonoscopy for Surveillance After CRC? David Weinberg, MD, MSc June 1, 2018 Goals of Post-operative CRC surveillance: Prevention or early detection of metachronous disease 30%-50% will suffer
CTC vs. Colonoscopy for Surveillance After CRC? David Weinberg, MD, MSc June 1, 2018 Goals of Post-operative CRC surveillance: Prevention or early detection of metachronous disease 30%-50% will suffer
How to characterize dysplastic lesions in IBD?
 How to characterize dysplastic lesions in IBD? Name: Institution: Helmut Neumann, MD, PhD, FASGE University Medical Center Mainz What do we know? Patients with IBD carry an increased risk of developing
How to characterize dysplastic lesions in IBD? Name: Institution: Helmut Neumann, MD, PhD, FASGE University Medical Center Mainz What do we know? Patients with IBD carry an increased risk of developing
Guidelines for Breast, Cervical and Colorectal Cancer Screening
 Guidelines for Breast, Cervical and Colorectal Cancer Screening Your recommendation counts. Talk to your patients about screening for cancer. CancerCare Manitoba provides organized, population-based screening
Guidelines for Breast, Cervical and Colorectal Cancer Screening Your recommendation counts. Talk to your patients about screening for cancer. CancerCare Manitoba provides organized, population-based screening
Improving Access to Endoscopy at Safety-Net Hospitals. Lukejohn W. Day MD Assistant Professor of Medicine
 Improving Access to Endoscopy at Safety-Net Hospitals Lukejohn W. Day MD Assistant Professor of Medicine Goals Background Improving Access to Endoscopic Care Electronic referral: ereferral Direct Access
Improving Access to Endoscopy at Safety-Net Hospitals Lukejohn W. Day MD Assistant Professor of Medicine Goals Background Improving Access to Endoscopic Care Electronic referral: ereferral Direct Access
Supplementary Appendix
 Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Kaminski MF, Regula J, Kraszewska E, et al. Quality indicators
Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Kaminski MF, Regula J, Kraszewska E, et al. Quality indicators
Epi procolon 2.0 CE is a blood test for colorectal cancer screening.
 Epi procolon 2.0 CE is a blood test for colorectal cancer screening. About Epi procolon 2.0 CE... 2 Screening Recommendations... 3 What You Should Know About Epi procolon 2.0 CE and the Septin 9 DNA Biomarker...
Epi procolon 2.0 CE is a blood test for colorectal cancer screening. About Epi procolon 2.0 CE... 2 Screening Recommendations... 3 What You Should Know About Epi procolon 2.0 CE and the Septin 9 DNA Biomarker...
11/11/2015. Colon Cancer Screening in Underserved Communities The Road to 80% by Colonoscopic Findings. Eighty by Cancer Screening Rates
 Colonoscopic Findings Polyps Polyp encircled with wire loop and cut from wall using electrical current Post Polypectomy Colon Cancer Screening in Underserved Communities The Road to 80% by 2018 Suzanne
Colonoscopic Findings Polyps Polyp encircled with wire loop and cut from wall using electrical current Post Polypectomy Colon Cancer Screening in Underserved Communities The Road to 80% by 2018 Suzanne
Screening for GI Cancer Past Present and Future. Prof. Bob Steele University of Dundee
 Screening for GI Cancer Past Present and Future Prof. Bob Steele University of Dundee Worldwide Cancer Incidence Rates UK Cancer Incidence Rates Screening The detection of disease in asymptomatic subjects
Screening for GI Cancer Past Present and Future Prof. Bob Steele University of Dundee Worldwide Cancer Incidence Rates UK Cancer Incidence Rates Screening The detection of disease in asymptomatic subjects
Experience and challenges of implementing optical diagnosis into clinical practice UK and European Perspective
 Experience and challenges of implementing optical diagnosis into clinical practice UK and European Perspective WEO Image Enhanced Endoscopy San Diego, USA Dr James East Consultant Gastroenterologist Honorary
Experience and challenges of implementing optical diagnosis into clinical practice UK and European Perspective WEO Image Enhanced Endoscopy San Diego, USA Dr James East Consultant Gastroenterologist Honorary
Razvan I. Arsenescu, MD Assistant Professor of Medicine Division of Digestive Diseases EARLY DETECTION OF COLORECTAL CANCER
 Razvan I. Arsenescu, MD Assistant Professor of Medicine Division of Digestive Diseases EARLY DETECTION OF COLORECTAL CANCER Epidemiology of CRC Colorectal cancer (CRC) is a common and lethal disease Environmental
Razvan I. Arsenescu, MD Assistant Professor of Medicine Division of Digestive Diseases EARLY DETECTION OF COLORECTAL CANCER Epidemiology of CRC Colorectal cancer (CRC) is a common and lethal disease Environmental
WEO CRC SC Meeting. Barcelona, Spain October 23, 2015
 WEO CRC SC Meeting Barcelona, Spain October 23, 2015 Identification of serrated polyposis syndrome in the context of population-based CRC screening programs Evelien Dekker Academic Medical Center Amsterdam,
WEO CRC SC Meeting Barcelona, Spain October 23, 2015 Identification of serrated polyposis syndrome in the context of population-based CRC screening programs Evelien Dekker Academic Medical Center Amsterdam,
Improving the Adenoma Detection Rate. ADR is a (the) priority quality indicator
 Improving the Adenoma Detection Rate Douglas K. Rex, MD, MACG Indiana University School of Medicine Indianapolis, IN ADR is a (the) priority quality indicator Endorsed Adenoma Detection Rate Cecal intubation
Improving the Adenoma Detection Rate Douglas K. Rex, MD, MACG Indiana University School of Medicine Indianapolis, IN ADR is a (the) priority quality indicator Endorsed Adenoma Detection Rate Cecal intubation
Chromoendoscopy or Narrow Band Imaging with Targeted biopsies Should be the Cancer Surveillance Endoscopy Procedure of Choice in Ulcerative Colitis
 Chromoendoscopy or Narrow Band Imaging with Targeted biopsies Should be the Cancer Surveillance Endoscopy Procedure of Choice in Ulcerative Colitis Bret A. Lashner, M.D. Professor of Medicine Director,
Chromoendoscopy or Narrow Band Imaging with Targeted biopsies Should be the Cancer Surveillance Endoscopy Procedure of Choice in Ulcerative Colitis Bret A. Lashner, M.D. Professor of Medicine Director,
EARLY DETECTION OF COLORECTAL CANCER. Epidemiology of CRC
 Razvan I. Arsenescu, MD Assistant Professor of Medicine Division of Digestive Diseases EARLY DETECTION OF COLORECTAL CANCER Epidemiology of CRC Colorectal cancer (CRC) is a common and lethal disease Environmental
Razvan I. Arsenescu, MD Assistant Professor of Medicine Division of Digestive Diseases EARLY DETECTION OF COLORECTAL CANCER Epidemiology of CRC Colorectal cancer (CRC) is a common and lethal disease Environmental
Natural history of adenomas by CT colonography Evelien Dekker Charlotte Tutein Nolthenius, Jaap Stoker
 Natural history of adenomas by CT colonography Charlotte Tutein Nolthenius, Jaap Stoker Academic Medical Center Amsterdam, the Netherlands Possible conflicts of interest None Colonoscopy.. plus polypectomy
Natural history of adenomas by CT colonography Charlotte Tutein Nolthenius, Jaap Stoker Academic Medical Center Amsterdam, the Netherlands Possible conflicts of interest None Colonoscopy.. plus polypectomy
Current trends in virtual colonoscopy
 Current trends in virtual colonoscopy Zarina I Lockhat, FFRad(D)SA Department of Radiology, Pretoria Academic Hospital and Irma van de Werke, FRCR Department of Radiology, Kalafong Hospital and André du
Current trends in virtual colonoscopy Zarina I Lockhat, FFRad(D)SA Department of Radiology, Pretoria Academic Hospital and Irma van de Werke, FRCR Department of Radiology, Kalafong Hospital and André du
PATIENT BROCHURE. 441 Charmany Dr 1 Madison WI, RX Only
 PATIENT BROCHURE 441 Charmany Dr 1 Madison WI, 53719 844-870- 8870 Cologuard colorectal cancer screening test is a registered trademark of Exact Sciences Corporation. Indications for Use Cologuard is intended
PATIENT BROCHURE 441 Charmany Dr 1 Madison WI, 53719 844-870- 8870 Cologuard colorectal cancer screening test is a registered trademark of Exact Sciences Corporation. Indications for Use Cologuard is intended
Prof Rupert Leong, Director of Endoscopy, Head of IBD Professor of Medicine UNSW, University of Sydney, Concord Hospital Australia IBDSydney
 10:30-10:50 25 Sept 2017 Monday A-PSDE / WEO Colorectal Cancer Screening Committee (CRCSC) Meeting Room S228 (2/F) Prof Rupert Leong, Director of Endoscopy, Head of IBD Professor of Medicine UNSW, University
10:30-10:50 25 Sept 2017 Monday A-PSDE / WEO Colorectal Cancer Screening Committee (CRCSC) Meeting Room S228 (2/F) Prof Rupert Leong, Director of Endoscopy, Head of IBD Professor of Medicine UNSW, University
COLON CANCER SCREENING: AN UPDATE
 Overview COLON CANCER SCREENING: AN UPDATE Siddharth Verma, DO, JD Rutgers New Jersey Medical School Background Screening Updates in Specific Populations African Americans CRC in the younger age USPSTF
Overview COLON CANCER SCREENING: AN UPDATE Siddharth Verma, DO, JD Rutgers New Jersey Medical School Background Screening Updates in Specific Populations African Americans CRC in the younger age USPSTF
