SUPPLEMENTAL DATA. Cytochrome P450-type hydroxylation and epoxidation in a tyrosine-liganded hemoprotein, catalase-related allene oxide synthase
|
|
|
- Ann Perkins
- 6 years ago
- Views:
Transcription
1 SUPPLEMENTAL DATA Cytochrome P450-type hydroxylation and epoxidation in a tyrosine-liganded hemoprotein, catalase-related allene oxide synthase William E. Boeglin & Alan R. Brash Fig. S1 SP-HPLC separation of the methyl esters of erythro and threo 8R-hydroxy-9,10- epoxy-eicosatrienoates (Products 1 and 2). Fig. S2 Illustrating the identity of the 1 H-NMR spectra (2.5 6 ppm) of the methyl ester of caos/phio- and mcpba-derived Product 1. Fig. S3 RP-HPLC analysis of the products formed from 8S-HETE by caos/phio. Fig. S4 Identification of 8,13-diHETE, product of 8S-HETE with caos/phio. Fig. S5 Assignment of the 13-hydroxyl configuration in caos-derived 8S,13-diHETE as 13R. Fig. S6 Identification of 8,16-diHETE, product of 8S-HETE with caos/phio. Fig. S7 Chiral analysis of 5,6-EET, product of Arachidonic Acid with caos/phio. Fig. S8 Chiral analysis of 14,15-EET, product of Arachidonic Acid with caos/phio. Fig. S9 Chiral analysis of 10-HETE, product of Arachidonic Acid with caos/phio. Pages 10-12, List of NMR data. Page 12, SI references. 1
2 Supplement Fig. S1: SP- HPLC separa-on of the methyl esters of erythro and threo 8R- hydroxy- 9,10- epoxy- eicosatrienoates (Products 1 and 2) Column: Thomson Advantage 5µ silica (25 x 0.46 cm); Solvent: of hexane/isopropanol (100:1, v/v), using a flow rate of 1 ml/min, with UV deteckon at 205 nm. Product 1 (erythro), from caos- PhIO- 8R- HETE incubakon Product 2 (threo), from mcpba reackon with 8R- HETE Product 1 (arrow) from caos- PhIO, co- injected with a 50:50 mix of erythro/threo standards (mcpba products) 2
3 Supplement Fig. S2 Fig. S2. Illustra/ng the iden/ty of the 1 H- NMR spectra (2.5 6 ppm) of the methyl ester of caos/phio- and mcpba- derived Product 1. The spectra were recorded in d6- benzene at 283 K. Top: caos/phio product 1 Below: mcpba product 1 Product 1 8R-hydroxy, 9R,10R-epoxy-20:3 H6 H12 H14 H15 H 3 CO 2 C H5 H O erythro caos/phio product H10 H8 - OCH3 H13 H9 mcpba product The enantiomers of 10RS-HETE are well resolved by chiral HPLC as the PFB ester. Assignment of the enantiomers was established by rearrangement of the earlier-eluting chiral column peak to 8S- HETE and 12R-HETE on exposure to 0.1% acetic acid. The rearrangement occurs with partial retention of the hydroxyl oxygen and is therefore assumed to be suprafacial, hence the assignment as 10S-HETE, cf. SI-ref. 2). This was confirmed by comparison with 10S-HETE prepared by total chemical synthesis (cf. SI-ref 2). 3
4 A 200 Absorbance mau Supplement Fig. S3 8S,13-diOH, 14,15-epoxy 8S,13-diOH 8S-OH,14,15-epoxy 8S-OH,11,12-epoxy 8S-OH,9,10-epoxy 235 nm retention time of 8S-HETE nm Retention time (min) B major products: (UV at 235 nm) 8 2 C 8 2 C OH 13 O OH 13 8S,13-diOH, 14,15-epoxy-20:3 8S,13-diHETE 2 C major products: (UV at 205 nm) O 8S-hydroxy, 14,15-epoxy-20:3 2 C O 8S-hydroxy, 11,12-epoxy-20:3 minor products: 2 C 8 OH 16 8S,16-diHETE 2 C O threo 8S-hydroxy, 9R,10R-epoxy-20:3 Fig. S3. RP-HPLC analysis of the products products formed from 8S-HETE by caos/phio (A) 8S-HETE (150 µm) was reacted with caos in the presence of 300 µm PhIO for 3 min at room temperature. Following extraction and formation of the methyl esters using diazomethane, an aliquot was analyzed on a Thomson Instrument Co. Advantage C18 column (25 x 0.46 cm) using a solvent system of MeOH/H 2 O/HAc (80/20/0.01 by volume) and a flow rate of 1 ml/min. As in Fig. 2A and 5A (main text), the UV traces of 205 nm and 235 nm are at the same sensitivity and are offset for clarity. The black dot at ~7.5 min marks a small peak identified by GC-MS, after hydrogenation, as 8R,16-diHETE (Fig S6). (B) Structures of the products identified (NMR and GC-MS data included in SI). 4
5 Supplement Fig. S4 Fig. S4. Identification of 8,13-diHETE, product of 8S-HETE with caos/phio 8,13-diHETE methyl ester was hydrogenated, converted to the trimethylsilyl ether (TMS) derivative and analyzed by GC-MS. Top: GC profile at m/z 73 (equivalent to any compounds containing TMS). Lower panel: Electron impact mass spectrum. Diagnostic ions are listed in the lower panel and illustrated on the structure. 5
6 Assignment of the 13- hydroxyl configura9on in 8S,13- dihete, product of 8S- HETE/cAOS/PhIO Chiral 13- HETE enan9omers were prepared by: (i) Vitamin E- controlled autoxida9on of arachidonate PFB ester, using 500% excess of vitamin E, and purifica9on of 13- HETE PFB ester. (ii) Resolu9on of 13- HETE enan9omers as the PFB esters using a Chiralpak AD column (solvent hexane/meoh, 100:2 v/v). (iii) Assignment of the 13R/13S configura9ons by mild acid (0.1% ace9c acid) treatment of the individual 13- HETE free acid enan9omers followed by purifica9on and chiral analysis of the 11- HETE and 15- HETE products (cf. SI- ref 2). Then standards of 8,13- dihete were prepared by: (iv) Reac9on of 13S- HETE with 8R- LOX to produce 8R,13S- dihete as standard. (v) Vitamin E- controlled autoxida9on of 13- HETE PFB ester followed by isola9on the two diastereomers of 8,13- dihete as standards: structures confirmed by GC- MS. (vi) Comparison of the caos/phio/8s- HETE- derived 8S,13- dihete with the 8R,13S- dihete and the pair of diastereomers (Fig. S7). Fig. S5: Assignment of the 13-hydroxyl A 8S,13- dihete (methyl ester) from caos- PhIO- 8S- HETE incuba9on configuration in caos-derived 8S,13-diHETE as 13R. Column: Thomson Advantage 5µ silica (25 x 0.46 cm), solvent, hexane/ipa (95:5 v/v), flow rate 1 ml/min, UV detection at 235 nm. Panel A: SP-HPLC of the 8S,13-diHETE product from caos/phio/8s-hete (methyl ester derivative), which elutes at a retention time of 11.2 min. B Co- injec9on of 8S,13- dihete (from caos- PhIO- 8S- HETE incuba9on) with 8,13- dihete diastereomer standards Panel B: as shown by co-injection, the 8S-HETEderived 8,13-diHETE co-chromatographed with the second of the two diastereomeric standards. C Co- injec9on of 8R,13S- dihete with 8,13- dihete diastereomer standards Panel C: The standard of 8R,13S-diHETE also co-chromatographed with the second diastereomer. Co-chromatography of the caos/phio/8s- HETE product and the 8R,13S-diHETE standard indicates that the two are enantiomers. Thus, the 8S-HETE-derived 8,13- dihete is 8S,13R-diHETE. MeO 2 C 8S,13R-diHETE OH S 13R C 5 H 11 6
7 Supplement Fig. S6 Fig. S6. Identification of 8,16-diHETE, product of 8S-HETE with caos/phio The peak marked with a dot in Fig S1A, was hydrogenated, converted to the trimethylsilyl ether (TMS) derivative and analyzed by GC-MS. Top: GC profile at m/z 73 (equivalent to any compounds containing TMS). Middle and lower panels: Electron impact mass spectrum. Diagnostic ions are listed in the middle panel and illustrated on the structure. 7
8 Fig. S7. Chiral analysis of 5,6- EET (top), product of Arachidonic Acid with caos/phio 5,6- EET was chromatographed on a Chiralcel OJ column (25 x 0.46 cm) as described (SI ref 1) using a solvent of hexane/isopropanol/glacial acemc acid (100:0.5:0.1, by vol.) at a flow rate of 1 ml/min, with UV detecmon at 205 nm. Chiral assignment was established under these condimons using synthemc standards (SI ref 1). 5,6- EET, enzyme product 5R,6S 5R,6S Racemic Standard 5S,6R Fig. S8. Chiral analysis of 14,15- EET (top), product of Arachidonic Acid with caos/phio 14,15- EET was chromatographed on a Chiralcel OJ column (25 x 0.46 cm) as described (SI ref 1) using a solvent of hexane/isopropanol/glacial acemc acid (100:0.5:0.1, by vol.) at a flow rate of 1 ml/ min, with UV detecmon at 205 nm. Chiral assignment was established under these condimons using synthemc standards (SI ref 1). 14,15- EET, enzyme product 14S,15R 14R,15S Racemic Standard 14S,15R 14R,15S 8
9 Fig. S9. Chiral analysis of 10- HETE, product of Arachidonic Acid with caos/phio 10- HETE was converted to the pentafluorobenzyl ester (PFB) and chromatographed on a Chiralpak AD column (25 x 0.46 cm) using a solvent of hexane/methanol (100:2, v/v) at a flow rate of 1 ml/ min, with UV detecson at 205 nm. The panel sizes below are adjusted to match retenson Smes on the x- axis. 10- HETE, enzyme product 10S, 83% 10R, 17% 10S Racemic Standard: 10RS- HETE PFB ester 10R The enantiomers of 10RS-HETE are well resolved by chiral HPLC as the PFB ester, (Supplemental Fig S9, above). Assignment of the enantiomers was established by rearrangement of the earliereluting chiral column peak to 8S-HETE and 12R-HETE, and the later peak to 8R-HETE and 12S- HETE, on exposure to 0.1% acetic acid. The rearrangement occurs with partial retention of the hydroxyl oxygen (SI ref 2) and is therefore assumed to be suprafacial, hence the assignment of the early eluting peak as 10S-HETE. The assignment using this method was confirmed by comparison to 10S-HETE prepared by total chemical synthesis (cf. SI ref 2). 9
10 NMR data Product 1, 8R-hydroxy-9R,10R-epoxy-eicosa-5Z,11Z,14Z-trienoate (methyl ester) (The erythro 8,9 diastereomer). 1 H-NMR, 600 MHz, C 6 D 6, 283 K, δ 5.61, dt, 1H, H12, J 11,12 = 10.9 Hz, J 12,13 = 7.7 Hz; 5.49, 1H, H6; , m, 2H, H14, H15; 5.36, m, 1H, H6; 5.155, dd, 1H, H11, J 11,12 = 10.8 Hz, J 10,11 = 9.9 Hz ; 3.81, dd, 1H, H10, J 9,10 = 2 Hz, J 10,11 = 8.9 Hz; 3.59, m, 1H, H8; 3.33, s, 3H, OCH3; 2.905, m, 2H, H13; 2.75, dd, 1H, H9, J 8,9 = 3.4 Hz, J 9,10 = 2 Hz; 2.24, m, 2H, H7; 2.08, t, 2H, H2; 2.00, q, 2H, H16; 1.91, m, 2H, H4; 1.57, p, 2H, H3; , m, 6H, H17, H18, H19; 0.89, t, 3H, H20. (In spectra recorded at a temperature of 288 K compared to 283 K, the H10 epoxide signal shifts upfield by ~ ppm at the higher temperature, from 3.81 to ~3.78 ppm). Product 2, 8R-hydroxy-9S,10S-epoxy-eicosa-5Z,11Z,14Z-trienoate (methyl ester) (The threo 8,9 diastereomer). 1 H-NMR, 600 MHz, C 6 D 6, 285 K, δ 5.95, dt, 1H, H12, J 11,12 = 10.9 Hz, J 12,13 = 7.7 Hz; ~ , 3H, H6, H14, H15; 5.34, m, 1H, H5; 5.13, t, 1H, H11, J = 10 Hz; 3.72, dd, 1H, H10, J 9,10 = 2 Hz, J 10,11 = 8.9 Hz; 3.405, m, 1H, H8; 3.34, s, 3H, OCH3; 2.88, m, 2H, H13; 2.745, dd, 1H, H9, J 8,9 = 4.0 Hz, J 9,10 = 2 Hz; 2.23, m, 2H, H7; 2.06, dt (!), 2H, H2; 1.99, q, 2H, H16; 1.91, m, 2H, H4; 1.70, d, 1H, OH (H8) J = 2 Hz; 1.55, p, 2H, H3; , m, 6H, H17, H18, H19; 0.89, t, 3H, H20. 8S-HETE product: 8S,13-dihydroxy-14,15-epoxy-eicosa-5Z,9E,11Z-trienoate (methyl ester) 1 H-NMR, 600 MHz, CDCl 3, 285 K, δ 6.55, dd, 1H, H10, J 10,11 = 11.3, J 9,10 = 15.1 Hz; 6.185, t, 1H, H11, J = 11.1 Hz; 5.84, dd, 1H, H9, J 8,9 = 5.8, J 9,10 = 15.1 Hz; 5.54, m, 1H, H5; 5.51, (dd?/m), 1H, H12; 5.42, m, 1H, H6; 4.44, t, 1H, H13, J = 8.1 Hz ; 4.23, m 1H, H8; 3.65, s, 3H, OCH3; 3.035, m, 2H, H14, H15; , m 2H, H7,a,b; 2.32, t, 2H, H2; 2.095, q, 2H, H4; 2.065, d, 1H, OH (H13) J = 2 Hz; 1.825, d, 1H, OH (H8) J = 2 Hz; 1.70, p, 2H, H3; ~ , m, H16; , m, 6H, H17, H18, H19; 0.87, t, 3H, H20. 8S-HETE product: 8S,13-dihydroxy-eicosa-5Z,9E,11Z,14Z-tetraenoate (methyl ester) 1 H-NMR, 600 MHz, CDCl 3, 285 K, δ 6.62, dd, 1H, H10, J 10,11 = 11.3, J 9,10 = 15.1 Hz; 6.04, t, 1H, H11, J = 11.1 Hz; 5.79, dd, 1H, H9, J 8,9 = 6.1, J 9,10 = 15.1 Hz; ~ , m, 6H, H5, H6, H12, H13, H14, H15; 4.24, m, 1H, H8; 3.67, s, 3H, OCH3; , m 2H, H7,a,b; 2.32, t, 2H, H2; ~ , m, 4H, H4, H16; 1.80, d, 1H, OH (H8) J = 4.3 Hz; 1.70, p, 2H, H3; 1.51, d, 1H, OH (H13) J = 2.4 Hz; 1.37, m, 2H, H16; , m, 6H, H17, H18, H19; 0.87, t, 3H, H20. 8S-HETE product: 8S-hydroxy-14,15-epoxy-eicosa-5Z,9E,11Z-trienoate (methyl ester) 1 H-NMR, 600 MHz, C 6 D 6, 285 K, δ 6.60, dd, 1H, H10, J 10,11 = 11.2, J 9,10 = 15.1 Hz; 6.11, t, 1H, H11, J = 11 Hz; 5.62, dd, 1H, H9, J 8,9 = 5.8, J 9,10 = 15.2 Hz; 5.46, dt, 1H, H12, J 11,12 = 10.9 Hz, J 12,13 = 7.6 Hz; 5.41, m, 1H, H6; 5.35, m, 1H, H5; 4.00, m, 1H, H8; 3.32, s, 3H, OCH3; 2.785, dt, 1H, H14, J 13,14 = 6.3, J 14,15 = 4. 1 Hz; 2.675, m, 1H, H15; 2.41, m, 1H, H13a; 2.28, m, 1H, H13b; 2.21, m, 1H, H7a; 2.16, m, 1H, H7b; 2.05, t, 2H, H2; 1.905, q, 2H, H4; 1.55, p, 2H, H3; 1.34, d, 1H, OH (H8), J = 4.4 Hz; ~ , m, 2H, H16; ~ , m, 6H, H17, H18, H19; 0.86, t, 3H, H20. 8S-HETE + caos + PhIO product (8S-hydroxy-11,12-epoxy-eicosa-5Z,9E,14Z-trienoate (methyl ester) 1 H-NMR, 600 MHz, C 6 D 6, 285 K, δ 5.805, dd, 1H, H9, J 8,9 = 5.3 Hz, J 9,10 = 15.5 Hz; 5.67, 1H, H10, J 9,10 = 15.5 Hz, J 10,11 = 7.2 Hz; 5.49, m, 2H, H14, H15; 5.35, m, 2H, H5, H6; 3.93, m, 1H, H8; 3.32, s, 3H, OCH3; 3.24, dd, 1H, H11, J 10,11 = 7 Hz, J 11,12 = 4 Hz; 2.94, dt, 1H, H12, J 11,12 = 4 Hz, J 12,13 = 6.3 Hz; 2.42, m, 1H, H13a; 2.195, m, 1H, H13b; ~ , m, 2H, H7a,7b; 2.05, 7, 2H, H2; 1.97, q, 2H, H16; 1.89, q, 2H, H4; 1.55, p, 2H, H3; , m, 6H, H17, H18, H19; 0.89, t, 3H, H20. 10
11 8S-HETE + caos + PhIO product (8S-hydroxy-9R,10R-epoxy-eicosa-5Z,9E,14Z-trienoate (methyl ester) This 8,9-threo diastereomer was the only 9,10-epoxide formed by reaction of 8S-HETE with caos+phio (Fig 5, main text). It is the enantiomer of the 8R-HETE threo product, with an identical NMR spectrum. mcpba products from 8R-HETE As the methyl esters, these eluted on RP-HPLC (MeOH/H 2 O, ) as: RP1 (products a 1 and a 2 ) - a mixture of 14,15-epoxy diastereomers RP2/RP3 (products b 1 and b 2 ) - two 11,12-epoxy diastereomers, partly resolved by RP-HPLC RP4/RP5 (products c 1 and c 2 ) - two 5,6-epoxy diastereomers, partly resolved by RP-HPLC RP6 (Products 1 and 2) - the erythro and threo 9,10-epoxy diastereomers The pairs of diastereomers were well resolved using a Chiralpak AD column (25 x 0.46 cm) with a solvent system of Hexane/EtOH/MeOH (100:5:5, v/v/v), flow rate 1 ml/min. Retention volumes: RP ml and 42.6 ml (8-hydroxy, 14,15-epoxy) RP ml (8-hydroxy, 11,12-epoxy) RP3 6.6 ml (8-hydroxy, 11,12-epoxy) RP ml (8-hydroxy, 5,6-epoxy) RP ml (8-hydroxy, 5,6-epoxy) RP6 9.4 ml (threo), 12.8 ml (erythro) 8-hydroxy, 9,10-epoxy Fig. 2B (main text) products a 1.2 : two mcpba products, diastereomers of 8R-hydroxy- 14,15-epoxy-eicosa-5Z,9E,11Z-trienoate methyl ester (NMR data not recorded but see 8S- HETE product, a 14,15-epoxide) Fig. 2B (main text) products b 1.2 : two mcpba products, diastereomers of 8R-hydroxy- 11,12-epoxy-eicosa-5Z,9E,14Z-trienoate methyl ester These had indistinguishable NMR spectra from each other and from the 11,12-epoxy product from 8S-HETE + caos + PhIO. Fig. 2B (main text) product c 1 : mcpba product, 5,6-epoxy-8R-hydroxy-eicosa-9E,11Z,14Ztrienoate methyl ester (the first diastereomer to elute, as the methyl ester, on RP-HPLC using MeOH/H 2 O solvent) 1 H-NMR, 600 MHz, C 6 D 6, δ 6.765, dd, 1H, H10, J 9,10 = 15.1 Hz, J 10,11 = 11.2 Hz; 6.085, t, 1H, H11, J = 11.1 Hz; 5.625, dd, 1H, H9, J 9,10 = 15.1 Hz, J 8,9 = 6.1; , m, 6H, H12, H14, H15; 4.285, q, 1H, H8, J = 6 Hz; 3.30, s, 3H, OCH3; 2.98, t, 2H, H13, J = 6 Hz; 2.80, dt, 1H, H6, J 5,6 = 4.2 Hz, J 6,7 = 6.2 Hz ; 2.52, dt, 1H, H5, J 4,5 = 6.2 Hz, J 5,6 = 4.2 Hz; ~ , m, 4H, H2, H16; , m, 4H, H3, H7; 1.31, m, H4; ~ , m, 6H, H17, H18, H19; 0.89, t, 3H, H20. Fig. 2B (main text) product c 2 : mcpba product, 5,6-epoxy-8R-hydroxy-eicosa-9E,11Z,14Ztrienoate methyl ester (the second diastereomer to elute, as the methyl ester, on RP-HPLC using MeOH/H 2 O solvent). There are significant differences in the chemical shifts compared to the first diastereomer, e.g. on the protons in the conjugated diene, and especially noticeable is the more downfield position of H6. 1 H-NMR, 600 MHz, C 6 D 6, 285 K, δ 6.65, dd, 1H, H10, J 9,10 = 15 Hz, J 10,11 = 11 Hz; 6.04, t, 1H, H11, J = 10.9 Hz; 5.585, dd, 1H, H9, J 9,10 = 15.1 Hz, J 8,9 = 6.0; 5.46, m, 6H, H12, H14, H15; 4.21, m, 1H, H8; 3.30, s, 3H, OCH3; 3.02, m (ddd?), 1H, H6; 2.97, t, 2H, H13, J = 7.5 Hz; 2.61, dt, 1H, H5, J 4,5 = 6.2 Hz, J 5,6 = 4.2 Hz; ~ , m, 4H, H2, H16; 1.595, m, 3H, H3, H7a; 1.505, ddd, H7b; ~ , m, 8H, H4, H17, H18, H19; 0.89, t, 3H, H20. 11
12 Fig. 2B (main text) products 1 and 2: two mcpba products, diastereomers of 8Rhydroxy-9,10-epoxy-eicosa-5Z,9E,14Z-trienoate methyl ester NMR data given above as Products 1 and 2. SI References 1. Wei S, et al. (2006) Chiral resolution of the epoxyeicosatrienoic acids, arachidonic acid epoxygenase metabolites. Anal. Biochem. 352: Brash AR, Boeglin WE, Capdevila JH, Suresh Y, Blair IA (1995) 7-HETE, 10-HETE, and 13-HETE are major products of NADPH-dependent metabolism of arachidonic acid in rat liver microsomes. Analysis of their stereochemistry and the stereochemistry of their acidcatalyzed rearrangement. Arch. Biochem. Biophys. 321:
Supporting Information
 Supporting Information Zheng, Y. et al, Lipoxygenases mediate the effect of essential fatty acid in skin barrier formation: A proposed role in releasing omega-hydroxyceramide for construction of the corneocyte
Supporting Information Zheng, Y. et al, Lipoxygenases mediate the effect of essential fatty acid in skin barrier formation: A proposed role in releasing omega-hydroxyceramide for construction of the corneocyte
* This work was supported, in whole or in part, by National Institutes of Health Grant GM (to A. R. B.). S
 THE JOURNAL OF BIOLOGICAL CHEMISTRY VOL. 287, NO. 29, pp. 24139 24147, July 13, 2012 2012 by The American Society for Biochemistry and Molecular Biology, Inc. Published in the U.S.A. Cytochrome P450-type
THE JOURNAL OF BIOLOGICAL CHEMISTRY VOL. 287, NO. 29, pp. 24139 24147, July 13, 2012 2012 by The American Society for Biochemistry and Molecular Biology, Inc. Published in the U.S.A. Cytochrome P450-type
Copyright Wiley-VCH Verlag GmbH, D Weinheim, Angew. Chem
 Copyright Wiley-VCH Verlag GmbH, D-69451 Weinheim, 2000. Angew. Chem. 2000. Supporting Information for Salen as Chiral Activator : Anti vs Syn Switchable Diastereoselection in the Enantioselective Addition
Copyright Wiley-VCH Verlag GmbH, D-69451 Weinheim, 2000. Angew. Chem. 2000. Supporting Information for Salen as Chiral Activator : Anti vs Syn Switchable Diastereoselection in the Enantioselective Addition
SUPPLEMENTARY DATA. Materials and Methods
 SUPPLEMENTARY DATA Materials and Methods HPLC-UV of phospholipid classes and HETE isomer determination. Fractionation of platelet lipid classes was undertaken on a Spherisorb S5W 150 x 4.6 mm column (Waters
SUPPLEMENTARY DATA Materials and Methods HPLC-UV of phospholipid classes and HETE isomer determination. Fractionation of platelet lipid classes was undertaken on a Spherisorb S5W 150 x 4.6 mm column (Waters
Electronic Supplementary Information. Quinine/Selectfluor Combination Induced Asymmetric Semipinacol Rearrangement of
 Electronic Supplementary Information Quinine/Selectfluor Combination Induced Asymmetric Semipinacol Rearrangement of Allylic Alcohols: An Effective and Enantioselective Approach to α Quaternary β Fluoro
Electronic Supplementary Information Quinine/Selectfluor Combination Induced Asymmetric Semipinacol Rearrangement of Allylic Alcohols: An Effective and Enantioselective Approach to α Quaternary β Fluoro
Student Handout. This experiment allows you to explore the properties of chiral molecules. You have
 Student Handout This experiment allows you to explore the properties of chiral molecules. You have learned that some compounds exist as enantiomers non-identical mirror images, such as your left and right
Student Handout This experiment allows you to explore the properties of chiral molecules. You have learned that some compounds exist as enantiomers non-identical mirror images, such as your left and right
Two chiral centres (diastereoisomers)
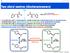 1 Two chiral centres (diastereoisomers) Mirror N S N Two enantiomers differ by absolute configuration N N 2 N 2 N A molecule with 1 stereogenic centre exists as 2 stereoisomers or enantiomers Enantiomers
1 Two chiral centres (diastereoisomers) Mirror N S N Two enantiomers differ by absolute configuration N N 2 N 2 N A molecule with 1 stereogenic centre exists as 2 stereoisomers or enantiomers Enantiomers
Supplementary Figure 1. ESI/MS/MS analyses of native and de-acetylated S2A Supplementary Figure 2. Partial 1D 1H NMR spectrum of S2A
 Supplementary Figure 1. ESI/MS/MS analyses of native and de-acetylated S2A. Panel A, Positive ESI mass spectra of native (N) and de-acetylated (DA) non-active S2A were obtained, and demonstrated a shift
Supplementary Figure 1. ESI/MS/MS analyses of native and de-acetylated S2A. Panel A, Positive ESI mass spectra of native (N) and de-acetylated (DA) non-active S2A were obtained, and demonstrated a shift
SUPPORTING INFORMATION for. Identification of key structural characteristics of Schisandra chinensis lignans
 SUPPORTING INFORMATION for Identification of key structural characteristics of Schisandra chinensis lignans involved in P-Glycoprotein inhibition Jiří Slanina #, Gabriela Páchniková #, Martina Čarnecká,
SUPPORTING INFORMATION for Identification of key structural characteristics of Schisandra chinensis lignans involved in P-Glycoprotein inhibition Jiří Slanina #, Gabriela Páchniková #, Martina Čarnecká,
Supporting information
 Supporting information Diversity Oriented Asymmetric Catalysis (DOAC): Stereochemically Divergent Synthesis of Thiochromanes Using an Imidazoline-aminophenol aminophenol (IAP)-Ni Catalyzed Michael/Henry
Supporting information Diversity Oriented Asymmetric Catalysis (DOAC): Stereochemically Divergent Synthesis of Thiochromanes Using an Imidazoline-aminophenol aminophenol (IAP)-Ni Catalyzed Michael/Henry
Novel D-erythro N-Octanoyl Sphingosine Analogs As Chemo- and Endocrine. Resistant Breast Cancer Therapeutics
 Page 11 of 32 Cancer Chemotherapy and Pharmacology Novel D-erythro N-Octanoyl Sphingosine Analogs As Chemo- and Endocrine Resistant Breast Cancer Therapeutics James W. Antoon, Jiawang Liu, Adharsh P. Ponnapakkam,
Page 11 of 32 Cancer Chemotherapy and Pharmacology Novel D-erythro N-Octanoyl Sphingosine Analogs As Chemo- and Endocrine Resistant Breast Cancer Therapeutics James W. Antoon, Jiawang Liu, Adharsh P. Ponnapakkam,
Oxylipin Analysis in Biological Systems
 Oxylipin Analysis in Biological Systems Dr Jacqui Adcock ARC DECRA Fellow Deakin University Centre for Chemistry and Biotechnology School of Life and Environmental Sciences Waurn Ponds, Victoria, Australia
Oxylipin Analysis in Biological Systems Dr Jacqui Adcock ARC DECRA Fellow Deakin University Centre for Chemistry and Biotechnology School of Life and Environmental Sciences Waurn Ponds, Victoria, Australia
Supporting Information
 Supporting Information Wiley-VCH 2008 69451 Weinheim, Germany Supporting Information Enantioselective Cu-catalyzed 1,4-Addition of Various Grignard Reagents to Cyclohexenone using Taddol-derived Phosphine-Phosphite
Supporting Information Wiley-VCH 2008 69451 Weinheim, Germany Supporting Information Enantioselective Cu-catalyzed 1,4-Addition of Various Grignard Reagents to Cyclohexenone using Taddol-derived Phosphine-Phosphite
Masatoshi Shibuya,Takahisa Sato, Masaki Tomizawa, and Yoshiharu Iwabuchi* Department of Organic Chemistry, Graduate School of Pharmaceutical Sciences,
 Oxoammonium ion/naclo 2 : An Expedient, Catalytic System for One-pot Oxidation of Primary Alcohols to Carboxylic Acid with Broad Substrate Applicability Masatoshi Shibuya,Takahisa Sato, Masaki Tomizawa,
Oxoammonium ion/naclo 2 : An Expedient, Catalytic System for One-pot Oxidation of Primary Alcohols to Carboxylic Acid with Broad Substrate Applicability Masatoshi Shibuya,Takahisa Sato, Masaki Tomizawa,
Naoya Takahashi, Keiya Hirota and Yoshitaka Saga* Supplementary material
 Supplementary material Facile transformation of the five-membered exocyclic E-ring in 13 2 -demethoxycarbonyl chlorophyll derivatives by molecular oxygen with titanium oxide in the dark Naoya Takahashi,
Supplementary material Facile transformation of the five-membered exocyclic E-ring in 13 2 -demethoxycarbonyl chlorophyll derivatives by molecular oxygen with titanium oxide in the dark Naoya Takahashi,
Enantioselective synthesis of anti- and syn-β-hydroxy-α-phenyl carboxylates via boron-mediated asymmetric aldol reaction
 Enantioselective synthesis of anti- and syn-β-hydroxy-α-phenyl carboxylates via boron-mediated asymmetric aldol reaction P. Veeraraghavan Ramachandran* and Prem B. Chanda Department of Chemistry, Purdue
Enantioselective synthesis of anti- and syn-β-hydroxy-α-phenyl carboxylates via boron-mediated asymmetric aldol reaction P. Veeraraghavan Ramachandran* and Prem B. Chanda Department of Chemistry, Purdue
Thiol-Activated gem-dithiols: A New Class of Controllable. Hydrogen Sulfide (H 2 S) Donors
 Thiol-Activated gem-dithiols: A New Class of Controllable Hydrogen Sulfide (H 2 S) Donors Yu Zhao, Jianming Kang, Chung-Min Park, Powell E. Bagdon, Bo Peng, and Ming Xian * Department of Chemistry, Washington
Thiol-Activated gem-dithiols: A New Class of Controllable Hydrogen Sulfide (H 2 S) Donors Yu Zhao, Jianming Kang, Chung-Min Park, Powell E. Bagdon, Bo Peng, and Ming Xian * Department of Chemistry, Washington
THE JOURNAL OF ANTIBIOTICS. Polyketomycin, a New Antibiotic from Streptomyces sp. MK277-AF1. II. Structure Determination
 THE JOURNAL OF ANTIBIOTICS Polyketomycin, a New Antibiotic from Streptomyces sp. MK277-AF1 II. Structure Determination ISAO MOMOSE, WEI CHEN, HIKARU NAKAMURA, HIROSHI NAGANAWA, HIRONOBU IINUMA and TOMIO
THE JOURNAL OF ANTIBIOTICS Polyketomycin, a New Antibiotic from Streptomyces sp. MK277-AF1 II. Structure Determination ISAO MOMOSE, WEI CHEN, HIKARU NAKAMURA, HIROSHI NAGANAWA, HIRONOBU IINUMA and TOMIO
Identification of novel endophenaside antibiotics produced by Kitasatospora sp. MBT66
 SUPPORTING INFORMATION belonging to the manuscript: Identification of novel endophenaside antibiotics produced by Kitasatospora sp. MBT66 by Changsheng Wu 1, 2, Gilles P. van Wezel 1, *, and Young Hae
SUPPORTING INFORMATION belonging to the manuscript: Identification of novel endophenaside antibiotics produced by Kitasatospora sp. MBT66 by Changsheng Wu 1, 2, Gilles P. van Wezel 1, *, and Young Hae
Developing a Biocascade Process: Concurrent Ketone Reduction-Nitrile Hydrolysis of 2-Oxocycloalkanecarbonitriles
 Developing a Biocascade Process: Concurrent Ketone Reduction-Nitrile Hydrolysis of 2-Oxocycloalkanecarbonitriles Elisa Liardo, a Nicolás Ríos-Lombardía, b Francisco Morís, b Javier González-Sabín,*,b and
Developing a Biocascade Process: Concurrent Ketone Reduction-Nitrile Hydrolysis of 2-Oxocycloalkanecarbonitriles Elisa Liardo, a Nicolás Ríos-Lombardía, b Francisco Morís, b Javier González-Sabín,*,b and
Asymmetric organocatalytic diboration of alkenes
 Asymmetric organocatalytic diboration of alkenes Amadeu Bonet, a Cristina Solé, Henrik Gulyás,* Elena Fernández* a Dept. Química Física i Inorgànica, University Rovira i Virgili, C/Marcel lí Domingo s/n,
Asymmetric organocatalytic diboration of alkenes Amadeu Bonet, a Cristina Solé, Henrik Gulyás,* Elena Fernández* a Dept. Química Física i Inorgànica, University Rovira i Virgili, C/Marcel lí Domingo s/n,
SUPPORTING INFORMATION SECTION: The manufacture of a homochiral 4-silyloxy-cyclopentenone intermediate for. the synthesis of prostaglandin analogues
 SUPPORTING INFORMATION SECTION: The manufacture of a homochiral 4-silyloxy-cyclopentenone intermediate for the synthesis of prostaglandin analogues Julian P. Henschke,* a Yuanlian Liu, b Xiaohong Huang,
SUPPORTING INFORMATION SECTION: The manufacture of a homochiral 4-silyloxy-cyclopentenone intermediate for the synthesis of prostaglandin analogues Julian P. Henschke,* a Yuanlian Liu, b Xiaohong Huang,
Non-Conjugated Double Bonds
 1 H-NMR Spectroscopy of Fatty Acids and Their Derivatives Non-Conjugated Double Bonds The introduction of one double bond gives rise to several peaks in the NMR spectrum compared to the saturated chains
1 H-NMR Spectroscopy of Fatty Acids and Their Derivatives Non-Conjugated Double Bonds The introduction of one double bond gives rise to several peaks in the NMR spectrum compared to the saturated chains
Supporting information
 Supporting information Figure legends Supplementary Table 1. Specific product ions obtained from fragmentation of lithium adducts in the positive ion mode comparing the different positional isomers of
Supporting information Figure legends Supplementary Table 1. Specific product ions obtained from fragmentation of lithium adducts in the positive ion mode comparing the different positional isomers of
Supporting Information. An Efficient Synthesis of Optically Active Physostigmine from Tryptophan via Alkylative Cyclization
 Supporting Information An Efficient Synthesis of Optically Active Physostigmine from Tryptophan via Alkylative Cyclization Michiaki, Kawahara, Atsushi Nishida, Masako Nakagawa* Faculty of Pharmaceutical
Supporting Information An Efficient Synthesis of Optically Active Physostigmine from Tryptophan via Alkylative Cyclization Michiaki, Kawahara, Atsushi Nishida, Masako Nakagawa* Faculty of Pharmaceutical
Tenofovir disoproxil fumarate (Tenofoviri disoproxili fumaras)
 C 19 H 30 N 5 O 10 P. C 4 H 4 O 4 Relative molecular mass. 635.5. Chemical names. bis(1-methylethyl) 5-{[(1R)-2-(6-amino-9H-purin-9-yl)-1-methylethoxy]methyl}-5-oxo-2,4,6,8-tetraoxa-5-λ 5 - phosphanonanedioate
C 19 H 30 N 5 O 10 P. C 4 H 4 O 4 Relative molecular mass. 635.5. Chemical names. bis(1-methylethyl) 5-{[(1R)-2-(6-amino-9H-purin-9-yl)-1-methylethoxy]methyl}-5-oxo-2,4,6,8-tetraoxa-5-λ 5 - phosphanonanedioate
Topic 6 Structure Determination Revision Notes
 1) Introduction Topic 6 Structure Determination Revision Notes Mass spectrometry, infrared spectroscopy and NMR spectroscopy can be used to determine the structure of unknown compounds 2) Mass spectrometry
1) Introduction Topic 6 Structure Determination Revision Notes Mass spectrometry, infrared spectroscopy and NMR spectroscopy can be used to determine the structure of unknown compounds 2) Mass spectrometry
Singlet oxygen photosensitisation by the fluorescent probe Singlet Oxygen Sensor Green
 Singlet oxygen photosensitisation by the fluorescent probe Singlet Oxygen Sensor Green Xavier Ragàs, Ana Jiménez-Banzo, David Sánchez-García, Xavier Batllori and Santi Nonell* Grup d Enginyeria Molecular,
Singlet oxygen photosensitisation by the fluorescent probe Singlet Oxygen Sensor Green Xavier Ragàs, Ana Jiménez-Banzo, David Sánchez-García, Xavier Batllori and Santi Nonell* Grup d Enginyeria Molecular,
Supporting Materials. Experimental Section. internal standard TMS (0 ppm). The peak patterns are indicated as follows: s, singlet; d,
 CuBr-Catalyzed Efficient Alkynylation of sp 3 C-H Bonds Adjacent to a itrogen Atom Zhiping Li and Chao-Jun Li* Department of Chemistry, McGill University, 801 Sherbrooke St. West, Montreal, Quebec H3A
CuBr-Catalyzed Efficient Alkynylation of sp 3 C-H Bonds Adjacent to a itrogen Atom Zhiping Li and Chao-Jun Li* Department of Chemistry, McGill University, 801 Sherbrooke St. West, Montreal, Quebec H3A
Supporting Information. were prepared from commercially available ethyl acetoacetate by alkylation with the
 ighly Stereoselective Reductions of α-alkyl-1,3-diketones and α- Alkyl-β-keto esters Catalyzed by Isolated NADP-dependent Ketoreductases Dimitris Kalaitzakis, a David J. Rozzell b, Spiros Kambourakis *b
ighly Stereoselective Reductions of α-alkyl-1,3-diketones and α- Alkyl-β-keto esters Catalyzed by Isolated NADP-dependent Ketoreductases Dimitris Kalaitzakis, a David J. Rozzell b, Spiros Kambourakis *b
List of Figure. Figure 1.1. Selective pathways for the metabolism of arachidonic acid
 List of Figure Figure 1.1. Selective pathways for the metabolism of arachidonic acid Figure 1.2. Arachidonic acid transformation to ω and ω-1 hydroxylase 14 19 reaction mediated by cytochrome P450 enzyme
List of Figure Figure 1.1. Selective pathways for the metabolism of arachidonic acid Figure 1.2. Arachidonic acid transformation to ω and ω-1 hydroxylase 14 19 reaction mediated by cytochrome P450 enzyme
SI APPENDIX. for Polyunsaturated fatty acid saturation by gut lactic acid bacteria affecting host lipid composition
 SI APPENDIX for Polyunsaturated fatty acid saturation by gut lactic acid bacteria affecting host lipid composition Shigenobu Kishino, Michiki Takeuchi, Si-Bum Park, Akiko Hirata, Nahoko Kitamura, Jun Kunisawa,
SI APPENDIX for Polyunsaturated fatty acid saturation by gut lactic acid bacteria affecting host lipid composition Shigenobu Kishino, Michiki Takeuchi, Si-Bum Park, Akiko Hirata, Nahoko Kitamura, Jun Kunisawa,
L-Carnosine-Derived Fmoc-Tripeptides Forming ph- Sensitive and Proteolytically Stable Supramolecular
 Supporting Information: L-Carnosine-Derived Fmoc-Tripeptides Forming ph- Sensitive and Proteolytically Stable Supramolecular Hydrogels Rita Das Mahapatra, a Joykrishna Dey* a, and Richard G. Weiss b a
Supporting Information: L-Carnosine-Derived Fmoc-Tripeptides Forming ph- Sensitive and Proteolytically Stable Supramolecular Hydrogels Rita Das Mahapatra, a Joykrishna Dey* a, and Richard G. Weiss b a
Biomimetic Chirality Sensing with Pyridoxal-5 -phosphate
 Supporting Information Biomimetic Chirality Sensing with Pyridoxal-5 -phosphate Samantha L. Pilicer, Pegah R. Bakhshi, Keith W. Bentley, Christian Wolf Department of Chemistry, Georgetown University, Washington,
Supporting Information Biomimetic Chirality Sensing with Pyridoxal-5 -phosphate Samantha L. Pilicer, Pegah R. Bakhshi, Keith W. Bentley, Christian Wolf Department of Chemistry, Georgetown University, Washington,
Supporting Information
 Supporting Information Wiley-VCH 2008 69451 Weinheim, Germany Enantioselective Rhodium-catalyzed Addition of Arylboronic Acids to α-ketoesters Hai-Feng Duan, Jian-Hua Xie, Xiang-Chen Qiao, Li-Xin Wang,
Supporting Information Wiley-VCH 2008 69451 Weinheim, Germany Enantioselective Rhodium-catalyzed Addition of Arylboronic Acids to α-ketoesters Hai-Feng Duan, Jian-Hua Xie, Xiang-Chen Qiao, Li-Xin Wang,
Edgar Naegele. Abstract
 Simultaneous determination of metabolic stability and identification of buspirone metabolites using multiple column fast LC/TOF mass spectrometry Application ote Edgar aegele Abstract A recent trend in
Simultaneous determination of metabolic stability and identification of buspirone metabolites using multiple column fast LC/TOF mass spectrometry Application ote Edgar aegele Abstract A recent trend in
# Supplementary Material (ESI) for Chemical Communications # This journal is The Royal Society of Chemistry 2005
 Electronic Supplementary Information for: (Z)-Selective cross-dimerization of arylacetylenes with silylacetylenes catalyzed by vinylideneruthenium complexes Hiroyuki Katayama,* Hiroshi Yari, Masaki Tanaka,
Electronic Supplementary Information for: (Z)-Selective cross-dimerization of arylacetylenes with silylacetylenes catalyzed by vinylideneruthenium complexes Hiroyuki Katayama,* Hiroshi Yari, Masaki Tanaka,
Divergent Construction of Pyrazoles via Michael Addition of N-Aryl Hydrazones to 1,2-Diaza-1,3-dienes
 Divergent Construction of Pyrazoles via Michael Addition of N-Aryl Hydrazones to 1,2-Diaza-1,3-dienes Serena Mantenuto, Fabio Mantellini, Gianfranco Favi,* and Orazio A. Attanasi Department of Biomolecular
Divergent Construction of Pyrazoles via Michael Addition of N-Aryl Hydrazones to 1,2-Diaza-1,3-dienes Serena Mantenuto, Fabio Mantellini, Gianfranco Favi,* and Orazio A. Attanasi Department of Biomolecular
Stereoselective Aza-Darzens Reactions of Tert- Butanesulfinimines: Convenient Access to Chiral Aziridines
 Stereoselective Aza-Darzens Reactions of Tert- Butanesulfinimines: Convenient Access to Chiral Aziridines Toni Moragas Solá, a Ian Churcher, b William Lewis a and Robert A. Stockman* a Supplementary Information
Stereoselective Aza-Darzens Reactions of Tert- Butanesulfinimines: Convenient Access to Chiral Aziridines Toni Moragas Solá, a Ian Churcher, b William Lewis a and Robert A. Stockman* a Supplementary Information
Preparation and Characterization of Cysteine Adducts of Deoxynivalenol
 Preparation and Characterization of Cysteine Adducts of Deoxynivalenol Ana Stanic, Silvio Uhlig, Anita Solhaug, Frode Rise, Alistair L. Wilkins, Christopher O. Miles S1 Figure S1. 1 H spectrum of 1 (DON)
Preparation and Characterization of Cysteine Adducts of Deoxynivalenol Ana Stanic, Silvio Uhlig, Anita Solhaug, Frode Rise, Alistair L. Wilkins, Christopher O. Miles S1 Figure S1. 1 H spectrum of 1 (DON)
Manganese powder promoted highly efficient and selective synthesis of fullerene mono- and biscycloadducts at room temperature
 Supplementary Information Manganese powder promoted highly efficient and selective synthesis of fullerene mono- and biscycloadducts at room temperature Weili Si 1, Xuan Zhang 1, Shirong Lu 1, Takeshi Yasuda
Supplementary Information Manganese powder promoted highly efficient and selective synthesis of fullerene mono- and biscycloadducts at room temperature Weili Si 1, Xuan Zhang 1, Shirong Lu 1, Takeshi Yasuda
Supplementary Material (ESI) for Chemical Communications This journal is (c) The Royal Society of Chemistry 2008
 Experimental Details Unless otherwise noted, all chemicals were purchased from Sigma-Aldrich Chemical Company and were used as received. 2-DOS and neamine were kindly provided by Dr. F. Huang. Paromamine
Experimental Details Unless otherwise noted, all chemicals were purchased from Sigma-Aldrich Chemical Company and were used as received. 2-DOS and neamine were kindly provided by Dr. F. Huang. Paromamine
Supporting Information. for. Synthesis of 2,1-benzisoxazole-3(1H)-ones by basemediated. photochemical N O bond-forming
 Supporting Information for Synthesis of 2,1-benzisoxazole-3(1H)-ones by basemediated photochemical N O bond-forming cyclization of 2-azidobenzoic acids Daria Yu. Dzhons and Andrei V. Budruev* Address:
Supporting Information for Synthesis of 2,1-benzisoxazole-3(1H)-ones by basemediated photochemical N O bond-forming cyclization of 2-azidobenzoic acids Daria Yu. Dzhons and Andrei V. Budruev* Address:
Electronic Supporting Information
 Electronic Supporting Information Detection of Hg 2+ by Cyanobacteria in Aqueous media. Moorthy Suresh, Sanjiv K. Mishra, Sandhya Mishra* and Amitava Das* Contents 1. Absorption spectrum of C-Phycocyanin
Electronic Supporting Information Detection of Hg 2+ by Cyanobacteria in Aqueous media. Moorthy Suresh, Sanjiv K. Mishra, Sandhya Mishra* and Amitava Das* Contents 1. Absorption spectrum of C-Phycocyanin
SUPPLEMENTARY MATERIAL
 SUPPLEMENTARY MATERIAL Chemical constituents from Agrimonia pilosa Ledeb. and their chemotaxonomic significance Wei-jie Liu, Xue-qian Hou, Hao Chen, Jing-yu Liang*, Jian-bo Sun** Department of Natural
SUPPLEMENTARY MATERIAL Chemical constituents from Agrimonia pilosa Ledeb. and their chemotaxonomic significance Wei-jie Liu, Xue-qian Hou, Hao Chen, Jing-yu Liang*, Jian-bo Sun** Department of Natural
What happens after activation of ascaridole? Reactive compounds and their implications for skin sensitization.
 What happens after activation of ascaridole? Reactive compounds and their implications for skin sensitization. Amar G. Chittiboyina,, * Cristina Avonto, and Ikhlas A. Khan., National Center for Natural
What happens after activation of ascaridole? Reactive compounds and their implications for skin sensitization. Amar G. Chittiboyina,, * Cristina Avonto, and Ikhlas A. Khan., National Center for Natural
Supporting Information
 Investigation of self-immolative linkers in the design of hydrogen peroxide metalloprotein inhibitors Jody L. Major Jourden, Kevin B. Daniel, and Seth M. Cohen* Department of Chemistry and Biochemistry,
Investigation of self-immolative linkers in the design of hydrogen peroxide metalloprotein inhibitors Jody L. Major Jourden, Kevin B. Daniel, and Seth M. Cohen* Department of Chemistry and Biochemistry,
Supporting Information
 Supporting Information A single design strategy for dual sensitive ph probe with a suitable range to map ph in living cells Kang-Kang Yu, Ji-Ting Hou, Kun Li, * Qian Yao, Jin Yang, Ming-Yu Wu, Yong-Mei
Supporting Information A single design strategy for dual sensitive ph probe with a suitable range to map ph in living cells Kang-Kang Yu, Ji-Ting Hou, Kun Li, * Qian Yao, Jin Yang, Ming-Yu Wu, Yong-Mei
Solid Phase Peptide Synthesis (SPPS) and Solid Phase. Fragment Coupling (SPFC) Mediated by Isonitriles
 Solid Phase Peptide Synthesis (SPPS) and Solid Phase Fragment Coupling (SPFC) Mediated by Isonitriles Ting Wang a and Samuel J. Danishefsky a,b,* alaboratory for Bioorganic Chemistry, Sloan- Kettering
Solid Phase Peptide Synthesis (SPPS) and Solid Phase Fragment Coupling (SPFC) Mediated by Isonitriles Ting Wang a and Samuel J. Danishefsky a,b,* alaboratory for Bioorganic Chemistry, Sloan- Kettering
Synergistic Cu-amine catalysis for the enantioselective synthesis of chiral cyclohexenones
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2015 Synergistic Cu-amine catalysis for the enantioselective synthesis of chiral cyclohexenones Adrien
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2015 Synergistic Cu-amine catalysis for the enantioselective synthesis of chiral cyclohexenones Adrien
ph Switchable and Fluorescent Ratiometric Squarylium Indocyanine Dyes as Extremely Alkaline Sensors
 ph Switchable and Fluorescent Ratiometric Squarylium Indocyanine Dyes as Extremely Alkaline Sensors Jie Li, Chendong Ji, Wantai Yang, Meizhen Yin* State Key Laboratory of Chemical Resource Engineering,
ph Switchable and Fluorescent Ratiometric Squarylium Indocyanine Dyes as Extremely Alkaline Sensors Jie Li, Chendong Ji, Wantai Yang, Meizhen Yin* State Key Laboratory of Chemical Resource Engineering,
Highly enantioselective tandem enzyme-organocatalyst crossed aldol reactions. with acetaldehyde in deep-eutectic-solvents.
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Highly enantioselective tandem enzyme-organocatalyst crossed aldol reactions with acetaldehyde
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Highly enantioselective tandem enzyme-organocatalyst crossed aldol reactions with acetaldehyde
Supporting Information. Copyright Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim, 2007
 Supporting Information Copyright Wiley-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim, 2007 Organocatalytic Asymmetric Sulfa-Michael Addition to α,β- Unsaturated Ketones Paolo Ricci, Armando Carlone, Giuseppe
Supporting Information Copyright Wiley-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim, 2007 Organocatalytic Asymmetric Sulfa-Michael Addition to α,β- Unsaturated Ketones Paolo Ricci, Armando Carlone, Giuseppe
Asymmetric organocatalytic synthesis of tertiary azomethyl alcohols: key intermediates towards azoxy compounds and α-hydroxy-β-amino esters
 Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2017 Asymmetric organocatalytic synthesis of tertiary azomethyl alcohols: key
Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2017 Asymmetric organocatalytic synthesis of tertiary azomethyl alcohols: key
Supporting Information for. Use of the Curtius Rearrangement of Acryloyl Azides in the Synthesis of. 3,5-Disubstituted Pyridines: Mechanistic Studies
 Supporting Information for Use of the Curtius Rearrangement of Acryloyl Azides in the Synthesis of 3,5-Disubstituted Pyridines: Mechanistic Studies Ta-Hsien Chuang* a, Yu-Chi Chen b and Someshwar Pola
Supporting Information for Use of the Curtius Rearrangement of Acryloyl Azides in the Synthesis of 3,5-Disubstituted Pyridines: Mechanistic Studies Ta-Hsien Chuang* a, Yu-Chi Chen b and Someshwar Pola
Surafel Mulugeta, Takashi Suzuki, Noemi Tejera Hernandez, Markus Griesser 1, William E. Boeglin, and Claus Schneider
 Identification and absolute configuration of dihydroxy-arachidonic acids formed by oxygenation of 5S-hydroxyeicosatetraenoic acid by native and aspirin-acetylated cyclooxygenase-2 Surafel Mulugeta, Takashi
Identification and absolute configuration of dihydroxy-arachidonic acids formed by oxygenation of 5S-hydroxyeicosatetraenoic acid by native and aspirin-acetylated cyclooxygenase-2 Surafel Mulugeta, Takashi
Eszopiclone (Lunesta ): An Analytical Profile
 Eszopiclone (Lunesta ): An Analytical Profile Roxanne E. Franckowski, M.S.* and Robert A. Thompson, Ph.D. U.S. Department of Justice Drug Enforcement Administration Special Testing and Research Laboratory
Eszopiclone (Lunesta ): An Analytical Profile Roxanne E. Franckowski, M.S.* and Robert A. Thompson, Ph.D. U.S. Department of Justice Drug Enforcement Administration Special Testing and Research Laboratory
You must answer FOUR of the SIX questions. Use a SEPARATE answer book for EACH question.
 UNIVERSITY OF EAST ANGLIA School of Pharmacy Main Series UG Examination 2016-17 DRUG DESIGN AND MECHANISMS OF DRUG ACTION PHA-5001Y Time allowed: 2 hours You must answer FOUR of the SIX questions. Use
UNIVERSITY OF EAST ANGLIA School of Pharmacy Main Series UG Examination 2016-17 DRUG DESIGN AND MECHANISMS OF DRUG ACTION PHA-5001Y Time allowed: 2 hours You must answer FOUR of the SIX questions. Use
Structure Determination of a New Juvenile Hormone from a Heteropteran Insect Toyomi Kotaki 1 *, Tetsuro Shinada 2*, Kanako Kaihara 2, Yasufumi Ohfune,
 Structure Determination of a New Juvenile Hormone from a Heteropteran Insect Toyomi Kotaki 1 *, Tetsuro Shinada 2*, Kanako Kaihara 2, Yasufumi hfune, 2 and Hideharu Numata 2 1 National Institute of Agrobiological
Structure Determination of a New Juvenile Hormone from a Heteropteran Insect Toyomi Kotaki 1 *, Tetsuro Shinada 2*, Kanako Kaihara 2, Yasufumi hfune, 2 and Hideharu Numata 2 1 National Institute of Agrobiological
Scheme S1. Synthesis of glycose-amino ligand.
 Scheme S1. Synthesis of glycose-amino ligand. 5-Chloro-1-pentyl-2,3,4,6-tetra-O-acetyl-ß-D-glucopyranoside S2 To a solution of penta-o-acetyl-ß-d-glucopyranoside S1 (3.0 g, 7.69 mmol) and 5-chloropentan-1-ol
Scheme S1. Synthesis of glycose-amino ligand. 5-Chloro-1-pentyl-2,3,4,6-tetra-O-acetyl-ß-D-glucopyranoside S2 To a solution of penta-o-acetyl-ß-d-glucopyranoside S1 (3.0 g, 7.69 mmol) and 5-chloropentan-1-ol
Supporting Information
 Electronic Supplementary Material (ESI) for rganic Chemistry Frontiers. This journal is the Partner rganisations 2016 Supporting Information Fangyi Li, Changgui Zhao, and Jian Wang* Department of Pharmacology
Electronic Supplementary Material (ESI) for rganic Chemistry Frontiers. This journal is the Partner rganisations 2016 Supporting Information Fangyi Li, Changgui Zhao, and Jian Wang* Department of Pharmacology
Biomass Oxidation to Formic Acid in Aqueous Media Using Polyoxometalate Catalysts Boosting FA Selectivity by In-situ Extraction
 Electronic Supplementary Material (ESI) for Energy & Environmental Science. This journal is The Royal Society of Chemistry 2015 Biomass Oxidation to Formic Acid in Aqueous Media Using Polyoxometalate Catalysts
Electronic Supplementary Material (ESI) for Energy & Environmental Science. This journal is The Royal Society of Chemistry 2015 Biomass Oxidation to Formic Acid in Aqueous Media Using Polyoxometalate Catalysts
DEPARTMENT OF CHEMISTRY AND CHEMICAL ORGANIC CHEMISTRY II 202-BZG-05 03
 DEPARTMENT OF CHEMISTRY AND CHEMICAL TECHNOLOGY ORGANIC CHEMISTRY II 202-BZG-05 03 TEST 1 11 MARCH 2010 INSTRUCTOR: I. DIONNE PRINT YOUR NAME: Answers INSTRUCTIONS: Answer all questions in the space provided.
DEPARTMENT OF CHEMISTRY AND CHEMICAL TECHNOLOGY ORGANIC CHEMISTRY II 202-BZG-05 03 TEST 1 11 MARCH 2010 INSTRUCTOR: I. DIONNE PRINT YOUR NAME: Answers INSTRUCTIONS: Answer all questions in the space provided.
Copper-Catalyzed Cascade Cycloamination of alpha-csp 3 -H Bond of N-Aryl Ketimines with Azides: Access to Quinoxalines. Supporting Information
 Copper-Catalyzed Cascade Cycloamination of alpha-csp 3 -H Bond of N-Aryl Ketimines with Azides: Access to Quinoxalines Tengfei Chen, Xun Chen, Jun Wei, Dongen Lin *, Ying Xie, and Wei Zeng * School of
Copper-Catalyzed Cascade Cycloamination of alpha-csp 3 -H Bond of N-Aryl Ketimines with Azides: Access to Quinoxalines Tengfei Chen, Xun Chen, Jun Wei, Dongen Lin *, Ying Xie, and Wei Zeng * School of
Zinc Chloride Promoted Formal Oxidative Coupling of Aromatic Aldehydes and Isocyanides to α- Ketoamides
 Supporting information for Zinc Chloride Promoted Formal xidative Coupling of Aromatic Aldehydes and Isocyanides to α- Ketoamides Marinus Bouma, Géraldine Masson* and Jieping Zhu* Institut de Chimie des
Supporting information for Zinc Chloride Promoted Formal xidative Coupling of Aromatic Aldehydes and Isocyanides to α- Ketoamides Marinus Bouma, Géraldine Masson* and Jieping Zhu* Institut de Chimie des
Electronic Supplementary Information
 Electronic Supplementary Information A Novel and Facile Zn-mediated Intramolecular Five-membered Cyclization of β-tetraarylporphyrin Radicals from β-bromotetraarylporphyrins Dong-Mei Shen, Chao Liu, Qing-Yun
Electronic Supplementary Information A Novel and Facile Zn-mediated Intramolecular Five-membered Cyclization of β-tetraarylporphyrin Radicals from β-bromotetraarylporphyrins Dong-Mei Shen, Chao Liu, Qing-Yun
Neuroprotective and Antioxidant Constituents from Curcuma zedoaria Rhizomes
 Supporting Information Rec. Nat. Prod. 9:3 (2015) 349-355 Neuroprotective and Antioxidant Constituents from Curcuma zedoaria Rhizomes Omer Abdalla Ahmed Hamdi 1, Lo Jia Ye 2, Muhamad Noor Alfarizal Kamarudin
Supporting Information Rec. Nat. Prod. 9:3 (2015) 349-355 Neuroprotective and Antioxidant Constituents from Curcuma zedoaria Rhizomes Omer Abdalla Ahmed Hamdi 1, Lo Jia Ye 2, Muhamad Noor Alfarizal Kamarudin
Supporting Information
 Supporting Information Asymmetric Catalysis of the Carbonyl-Amine Condensation: Kinetic Resolution of Primary Amines Sayantani Das, Nilanjana Majumdar, Chandra Kanta De, Dipti Sankar Kundu, Arno Döhring,
Supporting Information Asymmetric Catalysis of the Carbonyl-Amine Condensation: Kinetic Resolution of Primary Amines Sayantani Das, Nilanjana Majumdar, Chandra Kanta De, Dipti Sankar Kundu, Arno Döhring,
Preparation, isolation and characterization of N α -Fmoc-peptide isocyanates: Solution synthesis of oligo-α-peptidyl ureas
 SUPPORTING INFORMATION Preparation, isolation and characterization of N α -Fmoc-peptide isocyanates: Solution synthesis of oligo-α-peptidyl ureas Vommina V. Suresh Babu*, Basanagoud S. Patil, and Rao Venkataramanarao
SUPPORTING INFORMATION Preparation, isolation and characterization of N α -Fmoc-peptide isocyanates: Solution synthesis of oligo-α-peptidyl ureas Vommina V. Suresh Babu*, Basanagoud S. Patil, and Rao Venkataramanarao
Supporting Information. as the nitro source
 Supporting Information Efficient ipso-nitration of arylboronic acids with iron nitrate as the nitro source Min Jiang, a,b Haijun Yang,* a,b Yong Li, a,b Zhiying Jia b and Hua Fu b a Beijing Key Laboratory
Supporting Information Efficient ipso-nitration of arylboronic acids with iron nitrate as the nitro source Min Jiang, a,b Haijun Yang,* a,b Yong Li, a,b Zhiying Jia b and Hua Fu b a Beijing Key Laboratory
Supporting Information for. A convenient Method for Epoxidation of Alkenes using Aqueous. Hydrogen Peroxide
 Supporting Information for A convenient Method for Epoxidation of Alkenes using Aqueous Hydrogen Peroxide Man Kin Tse, Markus Klawonn, Santosh Bhor, Christian Döbler, Gopinathan Anilkumar, and Matthias
Supporting Information for A convenient Method for Epoxidation of Alkenes using Aqueous Hydrogen Peroxide Man Kin Tse, Markus Klawonn, Santosh Bhor, Christian Döbler, Gopinathan Anilkumar, and Matthias
Electronic Supplementary Information
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2015 Electronic Supplementary Information ovel pseudo[2]rotaxanes constructed by selfassembly of dibenzyl
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2015 Electronic Supplementary Information ovel pseudo[2]rotaxanes constructed by selfassembly of dibenzyl
Continuous flow retro-diels Alder reaction: an. efficient method for the preparation of pyrimidinone
 Supporting Information for Continuous flow retro-diels Alder reaction: an efficient method for the preparation of pyrimidinone derivatives Imane Nekkaa 1, Márta Palkó 1, István M. Mándity 1, and Ferenc
Supporting Information for Continuous flow retro-diels Alder reaction: an efficient method for the preparation of pyrimidinone derivatives Imane Nekkaa 1, Márta Palkó 1, István M. Mándity 1, and Ferenc
Supporting Information
 Supporting Information Unconventional Passerini Reaction towards α-aminoxyamides Ajay L. Chandgude, Alexander Dömling* Department of Drug Design, University of Groningen, Antonius Deusinglaan 1, 9713 AV
Supporting Information Unconventional Passerini Reaction towards α-aminoxyamides Ajay L. Chandgude, Alexander Dömling* Department of Drug Design, University of Groningen, Antonius Deusinglaan 1, 9713 AV
Catalytic decarboxylative alkylation of β-keto acids with sulfonamides via the cleavage of carbon nitrogen and carbon carbon bonds
 Catalytic decarboxylative alkylation of β-keto acids with sulfonamides via the cleavage of carbon nitrogen and carbon carbon bonds Cui-Feng Yang, Jian-Yong Wang and Shi-Kai Tian* Joint Laboratory of Green
Catalytic decarboxylative alkylation of β-keto acids with sulfonamides via the cleavage of carbon nitrogen and carbon carbon bonds Cui-Feng Yang, Jian-Yong Wang and Shi-Kai Tian* Joint Laboratory of Green
Supporting Information
 Supporting Information Direct Synthesis of Benzimidazoles by Dehydrogenative Coupling of Aromatic Diamines and Alcohols Catalyzed by Cobalt Prosenjit Daw, Yehoshoa Ben-David, and David Milstein* Department
Supporting Information Direct Synthesis of Benzimidazoles by Dehydrogenative Coupling of Aromatic Diamines and Alcohols Catalyzed by Cobalt Prosenjit Daw, Yehoshoa Ben-David, and David Milstein* Department
Methyltrioxorhenium-Catalyzed Highly Selective Dihydroxylation of 1,2-Allenylic Diphenyl Phosphine Oxides
 Electronic Supplementary Material (ESI) for Chemical Communications. This journal is The Royal Society of Chemistry 2015 Methyltrioxorhenium-Catalyzed Highly Selective Dihydroxylation of 1,2-Allenylic
Electronic Supplementary Material (ESI) for Chemical Communications. This journal is The Royal Society of Chemistry 2015 Methyltrioxorhenium-Catalyzed Highly Selective Dihydroxylation of 1,2-Allenylic
Preparation of Stable Aziridinium Ions and Their Ring Openings
 Supplementary Information Preparation of Stable Aziridinium Ions and Their Ring Openings Yongeun Kim a Hyun-Joon Ha*, a Sae Young Yun b and Won Koo Lee,*,b a Department of Chemistry and Protein Research
Supplementary Information Preparation of Stable Aziridinium Ions and Their Ring Openings Yongeun Kim a Hyun-Joon Ha*, a Sae Young Yun b and Won Koo Lee,*,b a Department of Chemistry and Protein Research
Studies on stereoselective separations of the azole antifungal drugs ketoconazole and itraconazole using HPLC and SFC on silica-based polysaccharides
 SFC Dossier Studies on stereoselective separations of the azole antifungal drugs ketoconazole and itraconazole using HPLC and SFC on silica-based polysaccharides A. Thienpont 1, J. Gal 2, C. Aeschlimann
SFC Dossier Studies on stereoselective separations of the azole antifungal drugs ketoconazole and itraconazole using HPLC and SFC on silica-based polysaccharides A. Thienpont 1, J. Gal 2, C. Aeschlimann
Applications Summary Waters Corporation 1
 Applications Summary 2012 Waters Corporation 1 UPC ² Key Messages UPC² is an exclusive, innovative separations tool which applies Waters proven UPLC technology to the traditional technique of SFC, broadening
Applications Summary 2012 Waters Corporation 1 UPC ² Key Messages UPC² is an exclusive, innovative separations tool which applies Waters proven UPLC technology to the traditional technique of SFC, broadening
Chukvelutins A-C, 16-norphragmalin limonoids with unprecedented skeletons from Chukrasia tabularis var. velutina
 Chukvelutins A-C, 16-norphragmalin limonoids with unprecedented skeletons from Chukrasia tabularis var. velutina Jun Luo, Jun-Song Wang, Jian-Guang Luo, Xiao-Bing Wang, and Ling-Yi Kong* Department of
Chukvelutins A-C, 16-norphragmalin limonoids with unprecedented skeletons from Chukrasia tabularis var. velutina Jun Luo, Jun-Song Wang, Jian-Guang Luo, Xiao-Bing Wang, and Ling-Yi Kong* Department of
Lewis acid-catalyzed regioselective synthesis of chiral α-fluoroalkyl amines via asymmetric addition of silyl dienolates to fluorinated sulfinylimines
 Supporting Information for Lewis acid-catalyzed regioselective synthesis of chiral α-fluoroalkyl amines via asymmetric addition of silyl dienolates to fluorinated sulfinylimines Yingle Liu a, Jiawang Liu
Supporting Information for Lewis acid-catalyzed regioselective synthesis of chiral α-fluoroalkyl amines via asymmetric addition of silyl dienolates to fluorinated sulfinylimines Yingle Liu a, Jiawang Liu
A biocatalytic hydrogenation of carboxylic acids
 Electronic Supplementary Information (ESI) for: A biocatalytic hydrogenation of carboxylic acids Yan Ni, Peter-Leon Hagedoorn,* Jian-He Xu, Isabel Arends, Frank Hollmann* 1. General Chemicals All the carboxylic
Electronic Supplementary Information (ESI) for: A biocatalytic hydrogenation of carboxylic acids Yan Ni, Peter-Leon Hagedoorn,* Jian-He Xu, Isabel Arends, Frank Hollmann* 1. General Chemicals All the carboxylic
Identification of Aromatic Fatty Acid Ethyl Esters
 Chapter 3.2 Identification of Aromatic Fatty Acid Ethyl Esters The only use of gas chromatography is not sufficient to determine which compounds are eluting from the catalytic bed. At the beginning of
Chapter 3.2 Identification of Aromatic Fatty Acid Ethyl Esters The only use of gas chromatography is not sufficient to determine which compounds are eluting from the catalytic bed. At the beginning of
Supporting Information
 Supporting Information P-Chirogenic Xantphos Ligands and Related Ether iphosphines Synthesis and Application in the Rhodium Catalyzed Asymmetric Hydrogenation Jens Holz,* a Katharina Rumpel, a Anke Spannenberg,
Supporting Information P-Chirogenic Xantphos Ligands and Related Ether iphosphines Synthesis and Application in the Rhodium Catalyzed Asymmetric Hydrogenation Jens Holz,* a Katharina Rumpel, a Anke Spannenberg,
Supporting Information
 J. Am. Chem. Soc. Supporting Information S 1 Enantioselective rganocatalytic Indole Alkylations. Design of a New and Highly Effective Chiral Amine for Iminium Catalysis. Joel F. Austin and David W. C.
J. Am. Chem. Soc. Supporting Information S 1 Enantioselective rganocatalytic Indole Alkylations. Design of a New and Highly Effective Chiral Amine for Iminium Catalysis. Joel F. Austin and David W. C.
University of Groningen
 University of Groningen Copper phosphoramidite-catalyzed enantioselective desymmetrization of meso-cyclic allylic bisdiethyl phosphates Piarulli, Umberto; Claverie, Christelle; Daubos, Philippe; Gennari,
University of Groningen Copper phosphoramidite-catalyzed enantioselective desymmetrization of meso-cyclic allylic bisdiethyl phosphates Piarulli, Umberto; Claverie, Christelle; Daubos, Philippe; Gennari,
Enzymatic resolution and evaluation of enantiomers of. cis-5 -hydroxythalidomide
 Electronic Supplementary Information Enzymatic resolution and evaluation of enantiomers of cis-5 -hydroxythalidomide Takeshi Yamamoto, a orio Shibata, a * Masayuki Takashima, a Shuichi akamura, a Takeshi
Electronic Supplementary Information Enzymatic resolution and evaluation of enantiomers of cis-5 -hydroxythalidomide Takeshi Yamamoto, a orio Shibata, a * Masayuki Takashima, a Shuichi akamura, a Takeshi
CHAPTER-6 IDENTIFICATION, AND CHARACTERISATION OF DEGRADATION IMPURITY IN VALSARTAN TABLETS
 129 CHAPTER-6 IDENTIFICATION, AND CHARACTERISATION OF DEGRADATION IMPURITY IN VALSARTAN TABLETS 130 6.1. Introduction Valsartan is an orally active specific angiotensin II blocker effective in lowering
129 CHAPTER-6 IDENTIFICATION, AND CHARACTERISATION OF DEGRADATION IMPURITY IN VALSARTAN TABLETS 130 6.1. Introduction Valsartan is an orally active specific angiotensin II blocker effective in lowering
Supplementary Information Titles
 CRRESPNDING AUTHR: MS NUMBER: MS TYPE: Fasan NCHEMB-BC080803093A Brief Communication Supplementary Information Titles Journal: Nature Chemical Biology Article Title: Corresponding Author: Supplementary
CRRESPNDING AUTHR: MS NUMBER: MS TYPE: Fasan NCHEMB-BC080803093A Brief Communication Supplementary Information Titles Journal: Nature Chemical Biology Article Title: Corresponding Author: Supplementary
Improved Pirkie Type 1 Amide Type: Asymmetric carbon atoms are bonded directly with CONH group
 1 sur 6 26/06/2013 14:16 Special Merits of SUMICHIRAL OA 1.The large number of theoretical plates of the columns offer high resolution. 2.The packing materials have chemical stability and the columns have
1 sur 6 26/06/2013 14:16 Special Merits of SUMICHIRAL OA 1.The large number of theoretical plates of the columns offer high resolution. 2.The packing materials have chemical stability and the columns have
Supporting Information
 Supporting Information Developing Activity Localization Fluorescence Peptide Probe Using Thiol-Ene Click Reaction for Spatially Resolved Imaging of Caspase-8 in Live Cells Wei Liu,, Si-Jia Liu,, Yong-Qing
Supporting Information Developing Activity Localization Fluorescence Peptide Probe Using Thiol-Ene Click Reaction for Spatially Resolved Imaging of Caspase-8 in Live Cells Wei Liu,, Si-Jia Liu,, Yong-Qing
Supporting Information
 Supporting Information B(C 6 F 5 ) 3 -catalyzed Regioselective Deuteration of Electronrich Aromatic and Heteroaromatic compounds Wu Li, Ming-Ming Wang, Yuya Hu and Thomas Werner* Leibniz-Institute of Catalysis
Supporting Information B(C 6 F 5 ) 3 -catalyzed Regioselective Deuteration of Electronrich Aromatic and Heteroaromatic compounds Wu Li, Ming-Ming Wang, Yuya Hu and Thomas Werner* Leibniz-Institute of Catalysis
Acyl Radical Reactions in Fullerene Chemistry: Direct Acylation of. [60]Fullerene through an Efficient Decatungstate-Photomediated Approach.
![Acyl Radical Reactions in Fullerene Chemistry: Direct Acylation of. [60]Fullerene through an Efficient Decatungstate-Photomediated Approach. Acyl Radical Reactions in Fullerene Chemistry: Direct Acylation of. [60]Fullerene through an Efficient Decatungstate-Photomediated Approach.](/thumbs/96/126778552.jpg) Supporting information Acyl Radical Reactions in Fullerene Chemistry: Direct Acylation of [60]Fullerene through an Efficient Decatungstate-Photomediated Approach. Manolis D. Tzirakis and Michael rfanopoulos
Supporting information Acyl Radical Reactions in Fullerene Chemistry: Direct Acylation of [60]Fullerene through an Efficient Decatungstate-Photomediated Approach. Manolis D. Tzirakis and Michael rfanopoulos
Supporting Information. for. Synthesis of dye/fluorescent functionalized. dendrons based on cyclotriphosphazene
 Supporting Information for Synthesis of dye/fluorescent functionalized dendrons based on cyclotriphosphazene Aurélien Hameau 1,2, Sabine Fuchs 1,2, Régis Laurent 1,2, Jean-Pierre Majoral* 1,2 and Anne-Marie
Supporting Information for Synthesis of dye/fluorescent functionalized dendrons based on cyclotriphosphazene Aurélien Hameau 1,2, Sabine Fuchs 1,2, Régis Laurent 1,2, Jean-Pierre Majoral* 1,2 and Anne-Marie
Specific N-Alkylation of Hydroxypyridines Achieved by a Catalyst- and Base-Free Reaction with Organohalides
 Supporting Information Specific N-Alkylation of Hydroxypyridines Achieved by a Catalyst- and Base-Free Reaction with Organohalides Bin Feng, Yang Li, Huan Li, Xu Zhang, Huamei Xie, Hongen Cao, Lei Yu,
Supporting Information Specific N-Alkylation of Hydroxypyridines Achieved by a Catalyst- and Base-Free Reaction with Organohalides Bin Feng, Yang Li, Huan Li, Xu Zhang, Huamei Xie, Hongen Cao, Lei Yu,
A pillar[2]arene[3]hydroquinone which can self-assemble to a molecular zipper in the solid state
![A pillar[2]arene[3]hydroquinone which can self-assemble to a molecular zipper in the solid state A pillar[2]arene[3]hydroquinone which can self-assemble to a molecular zipper in the solid state](/thumbs/86/93673402.jpg) A pillar[2]arene[3]hydroquinone which can self-assemble to a molecular zipper in the solid state Mingguang Pan, Min Xue* Department of Chemistry, Zhejiang University, Hangzhou 310027, P. R. China Fax:
A pillar[2]arene[3]hydroquinone which can self-assemble to a molecular zipper in the solid state Mingguang Pan, Min Xue* Department of Chemistry, Zhejiang University, Hangzhou 310027, P. R. China Fax:
Chiral Squaramide Derivatives are Excellent Hydrogen Bond Donor Catalysts. Jeremiah P. Malerich, Koji Hagihara, and Viresh H.
 Chiral Squaramide Derivatives are Excellent ydrogen Bond Donor Catalysts Jeremiah P. Malerich, Koji agihara, and Viresh. Rawal* Department of Chemistry, University of Chicago, Chicago, Illinois 60637 E-mail:
Chiral Squaramide Derivatives are Excellent ydrogen Bond Donor Catalysts Jeremiah P. Malerich, Koji agihara, and Viresh. Rawal* Department of Chemistry, University of Chicago, Chicago, Illinois 60637 E-mail:
Stereoselective preparations of epoxy-, fluoro- and related derivatives of ricinoleic acid and 13(S)-hydroxyoctadeca-9(Z),11(E)-dienoic acid
 Issue in Honor of Prof. A. McKervey ARKIVC 2003 (vii) 190-199 Stereoselective preparations of epoxy-, fluoro- and related derivatives of ricinoleic acid and 13(S)-hydroxyoctadeca-9(Z),11(E)-dienoic acid
Issue in Honor of Prof. A. McKervey ARKIVC 2003 (vii) 190-199 Stereoselective preparations of epoxy-, fluoro- and related derivatives of ricinoleic acid and 13(S)-hydroxyoctadeca-9(Z),11(E)-dienoic acid
On the Utility of S-Mesitylsulfinimines for the Stereoselective Synthesis of Chiral Amines and Aziridines
 n the Utility of S-Mesitylsulfinimines for the Stereoselective Synthesis of Chiral Amines and Aziridines Caroline Roe, a Toni Moragas Solá, a Leonid Sasraku-eequaye, b eather obbs, c Ian Churcher, c David
n the Utility of S-Mesitylsulfinimines for the Stereoselective Synthesis of Chiral Amines and Aziridines Caroline Roe, a Toni Moragas Solá, a Leonid Sasraku-eequaye, b eather obbs, c Ian Churcher, c David
