BIOCHEMICAL EVIDENCE FOR THE PRESENCE OF LYSOSOMES IN THE EPIDERMIS*
|
|
|
- Phillip Harper
- 5 years ago
- Views:
Transcription
1 ThE JOURNAL OP INVESTIGATiVE DERMATOLOGY Copyright 1966 by The Williams & Wilkins Co. Vol. 47, No. 5 Printed in U.S.A. BIOCHEMICAL EVIDENCE FOR THE PRESENCE OF LYSOSOMES IN THE EPIDERMIS* CHARLES H. DICKEN, M.D. AND RICHARD H. DECKER, PH.D Reviews (1, 2) of studies on lysosomes suggest that these organelles may play an important role in normal processes and in diverse pathologic conditions. Since lysosomes of different tissues may vary to a considerable extent, it is important to identify and characterize these organelles in the particular tissue to be rnvestigated. The presently available evidence for the presence of lysosomes in the epidermis is morphologic or histochemical. In electron micrographs, subcellular structures which resemble lysosomes have been seen in keratinocytes (3), in Langerhans cells (4, 5), and in intraepiderma! eccrine duct cells (6). Subeellular particles containing acid phosphatase have also been demonstrated in electron micrographs of keratinocytes of normal (7) and abnormal epidermis (8). Diengdoh (9) reported latency (time-related appearance) of acid phosphatase in mouse epidermis by using a histochemical technique and the light microscope. However, Eisen and coworkers (10) were unable to confirm this in human epidermis. Morphologic and histochemical methods cannot fulfil all the criteria for lysosomes (1). We have used biochemical methods for the demonstration of lysosomes in the epidermis of man and of the hairless mouse. METHODS Human skin was obtained from the abdomen at autopsy or from surgical amputations. Skin from the backs of hairless mice was also used. Epidermis was removed by the stretch method (11) and was immediately chilled in ice-cold, 0.3M sucrose buffered with 0.O1M phosphate buffer, ph 7.3. The tissue was homogenized in small batches for 10-second intervals at one-third to one-half speed in a VirTis Model 23 with the micro attachment; the Mayo Clinic and Mayo Foundation, Rochester, Minnesota. This investigation was supported in part by the Kieckhefer Research Fund and by Research Grant 2A-5299 from the National Institutes of Health, Pubhc Health Service. Presented at the Twenty-seventh Annual Meeting of The Society for Investigative Dermatology, Inc., Chicago, Illinois, June 26, the homogenate was filtered through one or two layers of cheesecloth to remove gross tissue fragments. The cloudy suspension was separated into three fractions, designated "nuclear," "lysosomal," and "supernatant" fractions, by the procedure of differential centrifugation indicated in Table I. Acid phosphatase was determined fluorometrically by adapting the procedure of Campbell and Moss (12) to the Turner Model 111 fluorometer with primary filter no (360 me) and secondary filter no. 47B (415 mae). Acid phosphatase activity was used to determine latency and specific activity. p-glucuronidase was assayed by a modification of the method of Fishman and coworkers (13), aryl sulfatase was determined by the method of Roy (14), and cathepsin was determined by the method of Gianetto and de Duve (15); these enzymes were measured only on homogenates because of the relative insensitivity of the assay procedures for their detection. Two technics were used to study the latent properties of the isolated lysosomes. According to the method of de Duve and co-workers (16), the lysosome suspension was incubated with substrate with or without exposure to disrupting conditions (for example, Triton-X-100). Thus, the access of substrate to enzymes still associated with granules is measured. This assay necessitates short incubation times since the ph of assay is sufficient to cause disruption of lysosomes. The acid phosphatase assay was the only procedure sensitive enough for this technic. By following the alternate method of Dingle (17), the particles were removed by centrifugation after pre-incubation with disrupting agents. The enzyme activity found in the superliate measures the release of enzyme from the granules caused by the test conditions. Protein was determined in the fractions by the method of Lowry and co-workers (18). Enzyme activities of the three fractions were measured after treatment with 0,1% Triton-X-100 for calculation of the specific and total activities because this agent increased the activity in the nuclear and lysosomal fractions. RESULTS We were able to demonstrate the activity of four of the enzymes associated with lysosomes in homogenates of the epidermis from man and from the hairless mouse. These enzymes are shown in Table II along with other enzymes that are probably associated with lysosomes and that have been demonstrated by biochemical or histochemical methods to be in epidermis. Acid phosphatase was assayed to determine the die-
2 LYSOSOMES IN EPIDERMIS 427 TAI3LE I Fractionation procedure Homogenate (0.3M sucrose in 0.O1M phosphate buffer) Filter through cheese cloth Filtrate Residu discarded Centrifuged (700 X g, 10 minutes) Nuclear fraction Supernatan fraction Centrifuged (12,000 X g, 10 minutes) Lysosomal fraction Supernatan1 fraction Resuspended in 0.25M sucrose Centrifuged (12,000 X g, 10 minutes) Supernat discarded tribution of activity and latency of the fractions. Although lysosomal acid phosphatase is usually measured with /3-glycerol phosphate as substrate, we employed a-naphthyl phosphate in a fluorometric assay method in order to obtain the sensitivity necessary for these studies. We found that the activity measured by this method was lysosomal acid phosphatase because it was sedimentable, showed latent activity, and had the same distribution as /3-glucuronidase. The distribution of acid phosphatase activity among the nuclear, lysosomal, and supernatant fractions is shown in Table III. Although the lysosomal fraction contained a relatively small part of the total activity, it also contained a smaller percentage of the total protein. That the fractionation procedure resulted in a purification of lysosomes is indicated by the increase in specific activity (enzyme activity per amount of protein) of the lysosomal fraction compared to the other fractions (Fig. 1). The relative specific activity of the iysosomal fraction could usually be increased, but Lysosomal fraction resuspended in 0.25M sucrose TABLE II Some lysosomal hydrotases found in epidermis Enzymes found, this study Other enzymes reported in literature Acid phosphatase -Glucuronidase Aryl sulfatase Cathepsin TABLE III Distribution of acid phosphatase activity in )ractions of epidermal homogenate Fraction Nuclear Supernatant Lysosomal Nonspecific esterases (19) Lipases (19) Ribonuclease (20) Deoxyribonuelease (20) Per cent of total activity (mean) Hairless mouse Human
3 428 THE JOURNAL OF INVESTIGATIVE DERMATOLOGY this generally occurred at the expense of the total activity of the fraction. Latency of the lysosomal fraction was demonstrated most effectively with Triton-X-100 g,.- $ Human epidermis Fm. 1. Relative specific activity of acid phosphatase in fractions of homogenates of epidermis from man and from hairless mouse. N = nuclear; L lysosomal; S = supernatant. and the centrifugation technic of Dingle (17) (Fig. 2 right). With mouse preparations, Tnton-X-100 caused a fivefold increase in activity compared with a control; sodium cholate caused a 2.7-fold increase (unpublished data). Hypotonic conditions were less effective, while freezing and thawing and acidic ph were without effect. Acid ph and hypotonic conditions caused only modest stimulation compared with that induced by Triton-X-100 when latency was determined by the procedure of de Duve and co-workers (16) (Fig. 2 left). Results with human skin (Fig. 3) were nearly identical to those obtained with mouse epidermis. COMMENT Four biochemical criteria for establishing the presence of lysosomes have been fulfilled for the epidermis from man and from the hairless mouse: (1) epidermal preparations contained at least eight acid hydrolases which are known to be associated with lysosomes; (2) a portion of one of the enzymes, acid phosphatase, was lincentrifuged Centrifuged nrill 10 - HnnnhI 0 I C Ff1 ph H NaF T C F+T p1-i H NaF Fm. 2. Demonstration of latency of acid phosphatase in lysosomal fraction from epidermis of hairless mouse. Abbreviations: T = 0.1% Triton-X-100; C = control; F + T freezing and thawing xlo; ph ph5; H = hypotonic conditions (distilled water); NaF.O1M sodium fluoride. Left, Method of de Duve and co-workers (16). Right, Method of Dingle (17).
4 LYSOSOMES IN EPIDERMIS 429 Fia. 3. Demonstration of latency of acid phosphatase in lysosomal fraction from human epidermis. Abbreviations and methods are the same as in Figure 2. sedimented by centrifugation under conditions which, with liver, yield a fraction known to contain lysosomes; (3) the acid phosphatase in the sedimented fraction possessed the highest specific activity of all fractions containing the enzyrne; and (4) the fraction contained latent acid phosphatase activity, demonstrable with Triton-X-100. Morphologic studies indicate that the lysosomal content of the epidermis is not dependent on one cell type. The keratinocyte (3, 7, 8), intraepidermal eccrine duct cell (6), and Langerhans cell (4, 5) all probably contribute to the epidermal lysosome content. The extent to which each contributes to the total would be expected to vary with cell population and with rate of metabolism of each cell type. The distribution of activity (Table III) depended to some extent on the technic employed during homogenization, quite likely because the force required to break the cells also breaks many lysosonies. Human skin was difficult to homogenize, and the yield of lysosomal fraction was low. The homogenization of mouse epidermis was accomplished more easily, and the higher yield of enzyme activity in the lysosomal fraction reflects this. Histochemically, acid phosphatase is found in significant amounts in the stratum granulosum and upper stratum corneum. The acid phosphatase activity in this location is not membrane limited. The high enzyme activity in a wash of the scraped-off epidermis before any homogenization suggests that this may be a significant factor in the high enzyme activity found in the supernatant fraction. The amount of activity that would be sedimented with the microsomal fraction was not determined, but it would be of interest in view of the finding of significant amounts of enzyme activity in this fraction in liver (21). The breaking of the lipoprotein membrane of the lysosome is usually associated with release or solubilization of the hydrolytic enzymes. The method of de Duve and co-workers (16) does not necessarily measure the solubilization of the enzyme(s) because the lysosomes are not centrifuged after releasing
5 430 THE JOURNAL OF INVESTIGATIVE DERMATOLOGY agents have been used. In this method intact lysosomes are used as a control and assays are short (10 minutes). In this manner, increased activity of the lysosomal enzymes in those samples previously treated with disrupting agents is demonstrated compared to the untreated lysosome samples. If the disrupting agents are used on the lysosomes and the solution is then centrifuged, the enzymic activity released into the supernate is less than the enzyme activity of a similarly treated duplicate that is not centrifuged. This is usually explained by stating that the difference is due to absorption of enzyme(s) on the walls of the disrupted lysocomes and that the fragments are then sedimentable. Another explanation or extension to the lysosome concept was suggested by Koenig (22): that the internal matrix of the lysosome is composed of macromolecules of enzyme and nonenzyme protein conjugated to acidic glycolipids by ionic, covalent, or other bonds. Electron micrographs of lysosornes do indicate that an internal structure exists (8, 23), and Koenig and Jibril (24) showed release of lysosomal enzymes with diverse cationic molecules. Acid ph, hypotonic solutions, and freezing and thawing readily release liver lysosomal enzymes (1). This means that these agents must disrupt the membrane and the internal matrix. We found little or no solubiization of enyme with acid ph, hypotonic solutions, or freezing and thawing, but an epidermal lysosome solution at ph 5 showed 50% of the activity of an epidermal lysosome solution made with 0.1% Triton-X-100. This finding suggests that the membrane may be broken by the above agents but that the internal matrix or large pieces of it continue to bind the enzyme and that it is therefore still sedimentable although 50% of the enzyme has been activated by the lowered ph. The increased activity and solubilization found with Triton-X-100 would be due to disruption of the membrane and the internal matrix. The process of keratinization is in many ways similar to cell death or autolysis. As epidermal cells progress from the basal layer to the stratum corneum, there is a disappearance of such organelles as the mitochondria and the nucleus. It would appear then that lysosomes may play a role in the degradative processes associated with formation of the stratum corneum. Lysosomes have the enzymes capable of this function, such as deoxyribonuclease, ribonuclease, protease, esterase, and lipase, and the acid ph in the upper layers of the epidermis is optimal for these acid hydrolases. The lysosomes tentatively appear to be implicated in tissue injury and inflammation, immune response, connective tissue disease, mitosis, fever, metabolic diseases, photosensitivity, and specific organ diseases (2, 25). The present evidence is circumstantial for lysosomal involvement in all of these processes, except in the case of some storage diseases, in which specific lack or diminution of a lysosomal enzyme has been shown (26 28). The role of lysosomes in skin diseases should be investigated, especially in view of their reported labiization or stabilization by such agents as ultraviolet irradiation, phylloerythrin, cantharidin, vitamin A, hydrocortisone, chioroquine, and antihistamines. SUMMARY Four biochemical tests have demonstrated the presence of lysosomes in the epidermis of man and of the hairless mouse: (1) presence of acid hydrolases, (2) sedimentability, (3) increased specific activity, (4) structure-linked latency. Epidermal homogenates from man and from the hairless mouse have been shown to contain four lysosomal enzymes: acid phosphatase, cathepsin, /3-glucuronidase, and aryl sulfatase. Acid phosphatase activity was used to demonstrate sedimentability of the lysosomal fraction and structure-linked latency. Acid ph, hypotonic solution, and freezing and thawing did not solubilize epidermal lysosomes. Triton- X-100 produced time-dependent solubiization (latency) of epidermal lysosomes by two methods. REFERENCES 1. De Duve, Christian: The lysosome concept. In De Reuck, A. V. S. and Cameron, Margaret P.: Lysosomes, pp Boston, Mass., Little, Brown & Company, Weissmann, Gerald: Lysosomes. New Eng. J. Med., 73: 1084; 1143, Mishima, Y.: Macromolecular changes in pigmentary disorders. Arch. Derm. (Chicago), 91: 519, Breathnach, A. S.: Observations on cytoplasmic organelles in Langerhans cells of human epidermis. J. Anat., 98: 265, 1964.
6 LYSOSOMES IN EPIDERMIS Zelickson, A. S.: The Langerhans cell. J. Invest. Derm., 44: 201, Hashimoto, Ken, Gross, B. G. and Lever, W. F.: The ultrastructure of the skin of human embryos. I. The intraepidermal cccrine sweat duct. J. Invest. Derm., 45: 139, Olson, R. L. and Nordquist, R. E.: Ultramicroscopic localization of acid phosphatase in human epidermis. J. Invest. Derm., 46: 431, Prose, P. H., Sedlis, Emilia and Bigelow, Monica: The demonstration of lysosomes in the diseased skin of infants with infantile eczema. J. Invest. Derm., 45: 448, Diengdoh, J. V.: The demonstration of lysosomes in mouse skin. Quart. J. Micr. Sci., 105: 73, Eisen, A. Z., Arndt, K. A. and Clark, W. H., Jr.: The ultrastructural localization of acid phosphatase in human epidermis. J. Invest. Derm., 43: 319, Van Scott, E. J.: Mechanical separation of epidermis from the corium. J. Invest. Derm., 18: 377, Campbell, Diana M. and Moss, D. W.: Spectrofluorimetric determination of acid phosphatase activity. Clin. Chem. Acta, 6: 307, Fishman, W. H., Springer, B. and Brunetti, R.: Application of an improved glucuronidase assay method to the study of human blood p-glucuronidase. J. Biol. Chem., 173: 449, Roy, A. B.: The suiphatase of ox liver. I. The complex nature of the enzyme. Biochem. J., 53: 12, Gianetto, R. and De Duve, C.: Tissue fractionation studies. IV. Comparative study of the binding of acid phosphatase, p-glucuronidase and eathepsin by rat-liver particles. Biochem. J., 59: 433, De Duve, C. Pressman, B. C., Gianetto, R., Wattiaux,. and Appelmans, F.: Tissue fractionation studies. VI. Intracellular distribution patterns of enzymes in rat-liver tissue. Biochem. J., 60: 604, Dingle, J. T.: Studies on the mode of action of excess of vitamin A. III. Release of a. bound protease by the action of vitamin A.. Biochem. J., 79: 509, Lowry, 0. II., Rosebrough, Nira J., Farr, A.. L. and Randall, Rose J.: Protein measure-. ment with the folin phenol reagent. J. BioL Chem., 193: 265, Montagna, William: The Structure and Function of Skin, 2nd edition, 454 pp. New York,. Academic Press, Inc., Santoianni, P. and Rothman, S.: Nucleic acid splitting enzymes in human epidermis and! their possible role in keratinization. J. In vest. Derm., 37: 489, Wattiaux, R., Baudhuin, P., Berleur, Anne-- Marie and De Duve, C.: Tissue fractionation studies. VIII. Cellular localization of bound enzymes. Biochem. J., 63: 608, Koenig, H.: Histological distribution of brain gangliosides: Lysosomes as glycolipoprotein granules. Nature, London, 195: 782, Daems, W. Th. ind Van Rijssel, Th. G.: The fine structure of the peribiliary dense bodies in mouse liver tissue. J. Ultrastruct. Res., 5: 263, Koenig, H. and Jibril, A.: Acidic glycolipids and the role of ionic bonds in the structurelinked latency of lysosomal hydrolases. Biochim Biophys. Acta, 65: 543, Slater, T. F. and Riley, P. A.: Photosensitization and lysosomal damage. Nature, London, 209: 151, Hers, H. G.: Glycogen storage disease, Type II. In Whelan, W. J. and Cameron, Margaret P.: Ciba Foundation Symposium on Control of Glycogen Metabolism, pp Boston, Mass., Little, Brown & Company, Austin, James, McAfee, Donald, Armstrong, Donald, O'Rourke, Michael, Shearer, Leslie and Bachhawat, Bimal: Abnormal sulphatase activities in two human diseases (metachromatic leucodystrophy and gargoylism). Biochem. J., 93: 15c, Bean, M. A. and Janoff, A.: Lysosomal enzymes in Gaucher's disease. (Abstr.) Fed. Proc., 24: 617, 1965.
the structure of their ducts has been
 Tza JOURNAL 0? INVEa'riGATrVN DEBMATOLOOT Copyright t 1966 by The Williams & Wilkins Co. Vol. 46, No. I Printed in U.S.A. AN ELECTRON MICROSCOPIC STUDY OF THE ADULT HUMAN APOCRINE DUCT* KEN HASHIMOTO,
Tza JOURNAL 0? INVEa'riGATrVN DEBMATOLOOT Copyright t 1966 by The Williams & Wilkins Co. Vol. 46, No. I Printed in U.S.A. AN ELECTRON MICROSCOPIC STUDY OF THE ADULT HUMAN APOCRINE DUCT* KEN HASHIMOTO,
Acid phosphatase activity in the neutral red granules of mouse exocrine pancreas cells
 343 Acid phosphatase activity in the neutral red granules of mouse exocrine pancreas cells By JENNIFER M. BYRNE (From the Cytological Laboratory, Department of Zoology, University Museum, Oxford) With
343 Acid phosphatase activity in the neutral red granules of mouse exocrine pancreas cells By JENNIFER M. BYRNE (From the Cytological Laboratory, Department of Zoology, University Museum, Oxford) With
Deterioration of Rat -Liver Mitochondria during Isopycnic Centrifugation in an Isoosmotic Medium
 Eur. J. Biochem. 51, 603-608 (1975) Deterioration of Rat -Liver Mitochondria during Isopycnic Centrifugation in an Isoosmotic Medium Michkle COLLOT, Simone WATTIAUX-DE CONINCK, and Robert WATTIAUX Laboratoire
Eur. J. Biochem. 51, 603-608 (1975) Deterioration of Rat -Liver Mitochondria during Isopycnic Centrifugation in an Isoosmotic Medium Michkle COLLOT, Simone WATTIAUX-DE CONINCK, and Robert WATTIAUX Laboratoire
Nuclear Extraction Kit
 Nuclear Extraction Kit Catalog Number KA1346 50 assays Version: 07 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Principle of the Assay... 3 General Information... 4
Nuclear Extraction Kit Catalog Number KA1346 50 assays Version: 07 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Principle of the Assay... 3 General Information... 4
Trident Membrane Protein Extraction Kit
 Cat. No. Size Shelf life GTX16373 5/ 20 tests 12 months at the appropriate storage temperatures (see below) Contents Component Storage Amount for 5 tests Amount for 20 tests Buffer A -20 o C 2.5 ml 10
Cat. No. Size Shelf life GTX16373 5/ 20 tests 12 months at the appropriate storage temperatures (see below) Contents Component Storage Amount for 5 tests Amount for 20 tests Buffer A -20 o C 2.5 ml 10
Changes in the Activities of Lysosomal Enzymes in Infarcted Canine Heart Muscle
 Changes in the Activities of Lysosomal Enzymes in Infarcted Canine Heart Muscle By Kurt G. Ravens, M.D., and S. Gudbjarnason, Ph.D. ABSTRACT Experimental myocardial infarction was produced in 32 mongrel
Changes in the Activities of Lysosomal Enzymes in Infarcted Canine Heart Muscle By Kurt G. Ravens, M.D., and S. Gudbjarnason, Ph.D. ABSTRACT Experimental myocardial infarction was produced in 32 mongrel
the Golgi complex with the formation of
 THE JOURNAL OF INVESTIGATIVE DERMATOLOGY Copyright 1968 by The Williams & Wilkins Co. Vol. 51, No. I Printed in U.S.A. ARYL SUTJFATASE IN THE SEBACEOUS GLANDS OF MOUSE SKIN A COMBINED ELECTRON MICROSCOPE
THE JOURNAL OF INVESTIGATIVE DERMATOLOGY Copyright 1968 by The Williams & Wilkins Co. Vol. 51, No. I Printed in U.S.A. ARYL SUTJFATASE IN THE SEBACEOUS GLANDS OF MOUSE SKIN A COMBINED ELECTRON MICROSCOPE
TRANSPORT OF AMINO ACIDS IN INTACT 3T3 AND SV3T3 CELLS. Binding Activity for Leucine in Membrane Preparations of Ehrlich Ascites Tumor Cells
 Journal of Supramolecular Structure 4:441 (401)-447 (407) (1976) TRANSPORT OF AMINO ACIDS IN INTACT 3T3 AND SV3T3 CELLS. Binding Activity for Leucine in Membrane Preparations of Ehrlich Ascites Tumor Cells
Journal of Supramolecular Structure 4:441 (401)-447 (407) (1976) TRANSPORT OF AMINO ACIDS IN INTACT 3T3 AND SV3T3 CELLS. Binding Activity for Leucine in Membrane Preparations of Ehrlich Ascites Tumor Cells
Effect of lysosomes on cultured rabbit corneal endothelial cells* Dharmendra V. Arya, Jean Mannagh, and A. Ray Irvine, Jr.
 Effect of lysosomes on cultured rabbit corneal endothelial cells* Dharmendra V. Arya, Jean Mannagh, and A. Ray Irvine, Jr. Lysosomal preparations induced marked changes in cultured corneal endothelial
Effect of lysosomes on cultured rabbit corneal endothelial cells* Dharmendra V. Arya, Jean Mannagh, and A. Ray Irvine, Jr. Lysosomal preparations induced marked changes in cultured corneal endothelial
Minute TM Plasma Membrane Protein Isolation and Cell Fractionation Kit User Manual (v5)
 Minute TM Plasma Membrane Protein Isolation and Cell Fractionation Kit Catalog number: SM-005 Description Minute TM plasma membrane (PM) protein isolation kit is a novel and patented native PM protein
Minute TM Plasma Membrane Protein Isolation and Cell Fractionation Kit Catalog number: SM-005 Description Minute TM plasma membrane (PM) protein isolation kit is a novel and patented native PM protein
PREPARATION OF IF- ENRICHED CYTOSKELETAL PROTEINS
 TMM,5-2011 PREPARATION OF IF- ENRICHED CYTOSKELETAL PROTEINS Ice-cold means cooled in ice water. In order to prevent proteolysis, make sure to perform all steps on ice. Pre-cool glass homogenizers, buffers
TMM,5-2011 PREPARATION OF IF- ENRICHED CYTOSKELETAL PROTEINS Ice-cold means cooled in ice water. In order to prevent proteolysis, make sure to perform all steps on ice. Pre-cool glass homogenizers, buffers
:1c.c :& Preliminary and Short Report GRANULE FORMATION IN THE LANGERHANS CELL* structure with rounded ends and a striated lamella
 THE JOURNAL OF INVESTIGATIVE DERMATOLOGY Copyright 1566 by The Williams & Wilkins Co. Vol. 7, No. 5 Printed in U.S.A. Preliminary and Short Report GRANULE FORMATION IN THE LANGERHANS CELL* ALVIN S. ZELICKSON,
THE JOURNAL OF INVESTIGATIVE DERMATOLOGY Copyright 1566 by The Williams & Wilkins Co. Vol. 7, No. 5 Printed in U.S.A. Preliminary and Short Report GRANULE FORMATION IN THE LANGERHANS CELL* ALVIN S. ZELICKSON,
ELECTRON MICROSCOPIC STUDY OF MELANIN-PHAGOCYTOSIS BY CUTANEOUS VESSELS IN CELLULAR BLUE NEVUS*
 THE JOURNAL 05' INVESTIGATIVE DERMATOLOGY Copyright 1969 by The Williams & Wilkinl Co. Vol. 62, No. 6 Printed in U.S.A. ELECTRON MICROSCOPIC STUDY OF MELANIN-PHAGOCYTOSIS BY CUTANEOUS VESSELS IN CELLULAR
THE JOURNAL 05' INVESTIGATIVE DERMATOLOGY Copyright 1969 by The Williams & Wilkinl Co. Vol. 62, No. 6 Printed in U.S.A. ELECTRON MICROSCOPIC STUDY OF MELANIN-PHAGOCYTOSIS BY CUTANEOUS VESSELS IN CELLULAR
Mammalian Membrane Protein Extraction Kit
 Mammalian Membrane Protein Extraction Kit Catalog number: AR0155 Boster s Mammalian Membrane Protein Extraction Kit is a simple, rapid and reproducible method to prepare cellular protein fractions highly
Mammalian Membrane Protein Extraction Kit Catalog number: AR0155 Boster s Mammalian Membrane Protein Extraction Kit is a simple, rapid and reproducible method to prepare cellular protein fractions highly
Nuclear Extraction Kit
 Nuclear Extraction Kit Item No. 10009277 www.caymanchem.com Customer Service 800.364.9897 Technical Support 888.526.5351 1180 E. Ellsworth Rd Ann Arbor, MI USA TABLE OF CONTENTS GENERAL INFORMATION 3 Materials
Nuclear Extraction Kit Item No. 10009277 www.caymanchem.com Customer Service 800.364.9897 Technical Support 888.526.5351 1180 E. Ellsworth Rd Ann Arbor, MI USA TABLE OF CONTENTS GENERAL INFORMATION 3 Materials
(From Arnold Biological Laboratory, Brown University, ProVence) P~s 3 To 5. Materials and Methods
 Published Online: 25 January, 1955 Supp Info: http://doi.org/10.1083/jcb.1.1.13 Downloaded from jcb.rupress.org on June 21, 2018 HISTOLOGY AND CYTOCHEMISTRY OF HUMAN SKIN IX. THE Dm~mUT~ON Ot NoN-SP~.c~Ic
Published Online: 25 January, 1955 Supp Info: http://doi.org/10.1083/jcb.1.1.13 Downloaded from jcb.rupress.org on June 21, 2018 HISTOLOGY AND CYTOCHEMISTRY OF HUMAN SKIN IX. THE Dm~mUT~ON Ot NoN-SP~.c~Ic
Acid hydrolases of the retinal pigment epithelium. E. R. Berman
 Acid hydrolases of the retinal pigment epithelium E. R. Berman Two enymes, P-galactosidase and N-acetyl-fi-glucosaminidase, have been detected in pigment epithelial cells of cattle. The optimum activity
Acid hydrolases of the retinal pigment epithelium E. R. Berman Two enymes, P-galactosidase and N-acetyl-fi-glucosaminidase, have been detected in pigment epithelial cells of cattle. The optimum activity
AND LEWIS THOMAS. 1 Hoffman-LaRoche Co., Nutley, N. J. 2 Nutritional Biochemicals, Inc., Cleveland, Ohio.
 Journal of Clinical Investigation Vol. 42, No. 5, 1963 STUDIES ON LYSOSOMES. II. THE EFFECT OF CORTISONE ON THE RELEASE OF ACID HYDROLASES FROM A LARGE GRANULE FRACTION OF RABBIT LIVER INDUCED BY AN EXCESS
Journal of Clinical Investigation Vol. 42, No. 5, 1963 STUDIES ON LYSOSOMES. II. THE EFFECT OF CORTISONE ON THE RELEASE OF ACID HYDROLASES FROM A LARGE GRANULE FRACTION OF RABBIT LIVER INDUCED BY AN EXCESS
FOCUS SubCell. For the Enrichment of Subcellular Fractions. (Cat. # ) think proteins! think G-Biosciences
 169PR 01 G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name FOCUS SubCell For the Enrichment of Subcellular Fractions (Cat. # 786 260) think
169PR 01 G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name FOCUS SubCell For the Enrichment of Subcellular Fractions (Cat. # 786 260) think
(From The Rockefeller Institute) Materials and Methods. Observations with the Electron Microscope
 ELECTRON MICROSCOPE STUDY OF THE DEVELOPMENT OF THE PAPILLOMA VIRUS IN THE SKIN OF THE RABBIT* BY ROBERT S. STONE,~ M.D., RICHARD E. SHOPE, M.D., DAN H. MOORE, P,~.D. (From The Rockefeller Institute) PLATES
ELECTRON MICROSCOPE STUDY OF THE DEVELOPMENT OF THE PAPILLOMA VIRUS IN THE SKIN OF THE RABBIT* BY ROBERT S. STONE,~ M.D., RICHARD E. SHOPE, M.D., DAN H. MOORE, P,~.D. (From The Rockefeller Institute) PLATES
ab Nuclear Extract Kit
 Version 1 Last updated 10 November 2017 ab221978 Nuclear Extract Kit For the preparation of nuclear extracts from mammalian and tissue. This product is for research use only and is not intended for diagnostic
Version 1 Last updated 10 November 2017 ab221978 Nuclear Extract Kit For the preparation of nuclear extracts from mammalian and tissue. This product is for research use only and is not intended for diagnostic
tt 't'.swrt. I i LOCALIZATION OF GOLD CHLORIDE AND ADENOSINE TRIPHOSPHATASE IN HUMAN LANGERHANS CELLS*
 THE JOURNAL OF INVESTIGATIVE DERMATOLOGY Copyright 1968 by The Williams & Wilkins Co. Vol. 51, No. 5 Printed in U.S.A. LOCALIZATION OF GOLD CHLORIDE AND ADENOSINE TRIPHOSPHATASE IN HUMAN LANGERHANS CELLS*
THE JOURNAL OF INVESTIGATIVE DERMATOLOGY Copyright 1968 by The Williams & Wilkins Co. Vol. 51, No. 5 Printed in U.S.A. LOCALIZATION OF GOLD CHLORIDE AND ADENOSINE TRIPHOSPHATASE IN HUMAN LANGERHANS CELLS*
(From the Arnold Biological Laboratory, Brown University, Providence, Rhode Island) Materials and Methods
 HISTOLOGY AND CYTOCHEMISTRY OF HUMAN SKIN XI. THF. DISTRIBUTION OF /~-GLucURONIDASE* BY WILLIAM MONTAGNA, PH.D. (From the Arnold Biological Laboratory, Brown University, Providence, Rhode Island) PLATES
HISTOLOGY AND CYTOCHEMISTRY OF HUMAN SKIN XI. THF. DISTRIBUTION OF /~-GLucURONIDASE* BY WILLIAM MONTAGNA, PH.D. (From the Arnold Biological Laboratory, Brown University, Providence, Rhode Island) PLATES
OF TRANSAMINASE IN RAT TISUES
 OF TRANSAMINASE IN RAT TISUES KOZO YAMADA, SHUNJI SAWAKI, AKIRA FUKUMURA AND MASARU HAYASHID epartment of Internal Mcdicine, Faculty of Medicine, Nagoya University, agoya Showa-ku, N (Received July 30,
OF TRANSAMINASE IN RAT TISUES KOZO YAMADA, SHUNJI SAWAKI, AKIRA FUKUMURA AND MASARU HAYASHID epartment of Internal Mcdicine, Faculty of Medicine, Nagoya University, agoya Showa-ku, N (Received July 30,
ab65336 Triglyceride Quantification Assay Kit (Colorimetric/ Fluorometric)
 Version 10 Last updated 19 December 2017 ab65336 Triglyceride Quantification Assay Kit (Colorimetric/ Fluorometric) For the measurement of triglycerides in various samples. This product is for research
Version 10 Last updated 19 December 2017 ab65336 Triglyceride Quantification Assay Kit (Colorimetric/ Fluorometric) For the measurement of triglycerides in various samples. This product is for research
Integumentary System and Body Membranes
 Integumentary System and Body Membranes The Skin and its appendages hair, nails, and skin glands Anatomy/Physiology NHS http://www.lab.anhb.uwa.edu.au/mb140/corepages/integumentary/integum.htm I. System
Integumentary System and Body Membranes The Skin and its appendages hair, nails, and skin glands Anatomy/Physiology NHS http://www.lab.anhb.uwa.edu.au/mb140/corepages/integumentary/integum.htm I. System
Do Now Makeups. 4. In which organelle would water and dissolved materials be stored? A. 1 B. 2 C. 3 D. 5. A. mitochondria B.
 Do Now Makeups Name: Date: 1. Which organelle is primarily concerned with the conversion of potential energy of organic compounds into suitable form for immediate use by the cell? A. mitochondria B. centrosomes
Do Now Makeups Name: Date: 1. Which organelle is primarily concerned with the conversion of potential energy of organic compounds into suitable form for immediate use by the cell? A. mitochondria B. centrosomes
(From the Department of Medicine, New York University School of Medicine, New York)
 STUDIES ON LYSOSOMES I. THE EFFECTS O]~ ENDOTOXIN, ENDOTOX.IN TOLERANCE, AND CORTISONE ON THE RELEASE OF ACID HYDROLASES ]~RO~I A GRANULAR FRACTION OF RABBIT LIVER* BY GERALD WEISSMANN,$ M.D., A~D LEWIS
STUDIES ON LYSOSOMES I. THE EFFECTS O]~ ENDOTOXIN, ENDOTOX.IN TOLERANCE, AND CORTISONE ON THE RELEASE OF ACID HYDROLASES ]~RO~I A GRANULAR FRACTION OF RABBIT LIVER* BY GERALD WEISSMANN,$ M.D., A~D LEWIS
Instructions. Fuse-It-mRNA easy. Shipping and Storage. Overview. Kit Contents. Specifications. Note: Important Guidelines
 Membrane fusion is a highly efficient method for transfecting various molecules and particles into mammalian cells, even into sensitive and primary cells. The Fuse-It reagents are cargo-specific liposomal
Membrane fusion is a highly efficient method for transfecting various molecules and particles into mammalian cells, even into sensitive and primary cells. The Fuse-It reagents are cargo-specific liposomal
TRANSFER OF PREMELANOSOMES INTO THE KERATINIZING CELLS OF ALBINO HAIR FOLLICLE
 TRANSFER OF PREMELANOSOMES INTO THE KERATINIZING CELLS OF ALBINO HAIR FOLLICLE PAUL F. PARAKKAL. From the Department of Dermatology, Boston University School of Medicine, Boston, Massachusetts 02118 INTRODUCTION
TRANSFER OF PREMELANOSOMES INTO THE KERATINIZING CELLS OF ALBINO HAIR FOLLICLE PAUL F. PARAKKAL. From the Department of Dermatology, Boston University School of Medicine, Boston, Massachusetts 02118 INTRODUCTION
Plasma Membrane Protein Extraction Kit
 ab65400 Plasma Membrane Protein Extraction Kit Instructions for Use For the rapid and sensitive extraction and purification of Plasma Membrane proteins from cultured cells and tissue samples. This product
ab65400 Plasma Membrane Protein Extraction Kit Instructions for Use For the rapid and sensitive extraction and purification of Plasma Membrane proteins from cultured cells and tissue samples. This product
Midi Plant Genomic DNA Purification Kit
 Midi Plant Genomic DNA Purification Kit Cat #:DP022MD/ DP022MD-50 Size:10/50 reactions Store at RT For research use only 1 Description: The Midi Plant Genomic DNA Purification Kit provides a rapid, simple
Midi Plant Genomic DNA Purification Kit Cat #:DP022MD/ DP022MD-50 Size:10/50 reactions Store at RT For research use only 1 Description: The Midi Plant Genomic DNA Purification Kit provides a rapid, simple
CELLS CONTAINING LANGERHANS GRANULES IN HUMAN LYMPH NODES OF DERMATOPATHIC LYMPHADENOPATHY*
 THS JOURNAL OF INVEBTIOATIVR DERMATOLOGY Copyright 1969 by The Williams & Wilkins Co. Vol. 93, No. 4 Printed in U.S.A. CELLS CONTAINING LANGERHANS GRANULES IN HUMAN LYMPH NODES OF DERMATOPATHIC LYMPHADENOPATHY*
THS JOURNAL OF INVEBTIOATIVR DERMATOLOGY Copyright 1969 by The Williams & Wilkins Co. Vol. 93, No. 4 Printed in U.S.A. CELLS CONTAINING LANGERHANS GRANULES IN HUMAN LYMPH NODES OF DERMATOPATHIC LYMPHADENOPATHY*
DEBRIDEMENT: ANATOMY and PHYSIOLOGY. Professor Donald G. MacLellan Executive Director Health Education & Management Innovations
 DEBRIDEMENT: ANATOMY and PHYSIOLOGY Professor Donald G. MacLellan Executive Director Health Education & Management Innovations ANATOMY and PHYSIOLOGY Epidermal Layers ECM Structure Dermis Structure Skin
DEBRIDEMENT: ANATOMY and PHYSIOLOGY Professor Donald G. MacLellan Executive Director Health Education & Management Innovations ANATOMY and PHYSIOLOGY Epidermal Layers ECM Structure Dermis Structure Skin
A rapid procedure for the isolation of lysosomes from kidney cortex by Percoll density gradient centrifugation
 J. Biosci., Vol. 14, Number 3, September 1989, pp. 269-277. Printed in India. A rapid procedure for the isolation of lysosomes from kidney cortex by Percoll density gradient centrifugation P. HARIKUMAR,
J. Biosci., Vol. 14, Number 3, September 1989, pp. 269-277. Printed in India. A rapid procedure for the isolation of lysosomes from kidney cortex by Percoll density gradient centrifugation P. HARIKUMAR,
For the quantitative measurement of ATP Synthase Specific activity in samples from Human, Rat and Cow
 ab109716 ATP Synthase Specific Activity Microplate Assay Kit Instructions for Use For the quantitative measurement of ATP Synthase Specific activity in samples from Human, Rat and Cow This product is for
ab109716 ATP Synthase Specific Activity Microplate Assay Kit Instructions for Use For the quantitative measurement of ATP Synthase Specific activity in samples from Human, Rat and Cow This product is for
INTEGUMENTARY 1-Epidermis, 2-Dermis, Structure of thick and thin skin I- Epidermis . Stratum basale
 INTEGUMENTARY The skin (integument, cutis ) and its derivatives constitute the integumentary system. It form the external covering of the body and is the largest organ of the body. The skin consists of
INTEGUMENTARY The skin (integument, cutis ) and its derivatives constitute the integumentary system. It form the external covering of the body and is the largest organ of the body. The skin consists of
PRODUCT INFORMATION & MANUAL
 PRODUCT INFORMATION & MANUAL Mitochondrial Extraction Kit NBP2-29448 Research use only. Not for diagnostic or therapeutic procedures www.novusbio.com P: 303.760.1950 P: 888.506.6887 F: 303.730.1966 technical@novusbio.com
PRODUCT INFORMATION & MANUAL Mitochondrial Extraction Kit NBP2-29448 Research use only. Not for diagnostic or therapeutic procedures www.novusbio.com P: 303.760.1950 P: 888.506.6887 F: 303.730.1966 technical@novusbio.com
ab Membrane Fractionation Kit Instructions for Use For the rapid and simple separation of membrane, cytosolic and nuclear cellular fractions.
 ab139409 Membrane Fractionation Kit Instructions for Use For the rapid and simple separation of membrane, cytosolic and nuclear cellular fractions. This product is for research use only and is not intended
ab139409 Membrane Fractionation Kit Instructions for Use For the rapid and simple separation of membrane, cytosolic and nuclear cellular fractions. This product is for research use only and is not intended
Cell morphology. Cell organelles structure and function. Chapter 1: UNIT 1. Dr. Charushila Rukadikar
 UNIT 1 Cell morphology Cell organelles structure and function Chapter 1: Dr. Charushila Rukadikar Assistant Professor Department Of Physiology ZMCH, Dahod Physiology The science that is concerned with
UNIT 1 Cell morphology Cell organelles structure and function Chapter 1: Dr. Charushila Rukadikar Assistant Professor Department Of Physiology ZMCH, Dahod Physiology The science that is concerned with
Mitochondria Isolation Kit for Tissue
 ab110168 Mitochondria Isolation Kit for Tissue Instructions for Use For mitochondria isolations from mammalian tissue samples This product is for research use only and is not intended for diagnostic use.
ab110168 Mitochondria Isolation Kit for Tissue Instructions for Use For mitochondria isolations from mammalian tissue samples This product is for research use only and is not intended for diagnostic use.
Mediators of Histamine Release from Human Platelets, Lymphocytes, and Granulocytes
 Mediators of Histamine Release from Human Platelets, Lymphocytes, and Granulocytes MICHAEL T. KELLY, R. RUSSELL MARTIN, and ARTHuR WHITE From the Infectious Disease Division, Department of Medicine, Indiana
Mediators of Histamine Release from Human Platelets, Lymphocytes, and Granulocytes MICHAEL T. KELLY, R. RUSSELL MARTIN, and ARTHuR WHITE From the Infectious Disease Division, Department of Medicine, Indiana
FOR RESEARCH USE ONLY. NOT FOR HUMAN OR DIAGNOSTIC USE.
 User Protocol 539790 Rev. 28 March 05 JSW Page 1 of 17 ProteoExtract Subcellular Proteome Extraction Kit Cat. No. 539790 User Protocol 539790 Rev. 28 March 05 JSW Page 2 of 17 1. Introduction One major
User Protocol 539790 Rev. 28 March 05 JSW Page 1 of 17 ProteoExtract Subcellular Proteome Extraction Kit Cat. No. 539790 User Protocol 539790 Rev. 28 March 05 JSW Page 2 of 17 1. Introduction One major
Threonine Aldolase and Allothreonine Aldolase in Rat Liver
 European J. Biochem. 8 (1969) 88-92 Threonine Aldolase and Allothreonine Aldolase in Rat Liver G. RIARIO-SFORZA, R. PAGANI, and E. MARINELLO Istituto di Chimica Biologica, UniversitA di Siena (Received
European J. Biochem. 8 (1969) 88-92 Threonine Aldolase and Allothreonine Aldolase in Rat Liver G. RIARIO-SFORZA, R. PAGANI, and E. MARINELLO Istituto di Chimica Biologica, UniversitA di Siena (Received
THE GOLGI APPARATUS IN SMALL LYMPHOCYTES AND IN
 THE GOLG APPARATUS N SMALL LYMPHOCYTES AND N TRANSFORMED BLAST CELLS. By A. S. COULSON. From the Department of Pathology, Cambridge University. (Received for publication 23rd December 1964) The Golgi apparatus
THE GOLG APPARATUS N SMALL LYMPHOCYTES AND N TRANSFORMED BLAST CELLS. By A. S. COULSON. From the Department of Pathology, Cambridge University. (Received for publication 23rd December 1964) The Golgi apparatus
Cathepsin K Activity Assay Kit
 Cathepsin K Activity Assay Kit Catalog Number KA0769 100 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Background... 3 General Information... 4 Materials
Cathepsin K Activity Assay Kit Catalog Number KA0769 100 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Background... 3 General Information... 4 Materials
Lipase Activity of Guinea Pig Peritoneal Macrophages and Mycobacterial Lipase Inhibitor*)
 Hiroshima Journal of Medical Sciences Vol. 32, No. 3, 267271, September, 1983 HIJM 32-42 267 Lipase Activity of Guinea Pig Peritoneal Macrophages and Mycobacterial Lipase Inhibitor*) Katsuhiro KIYOT ANI,
Hiroshima Journal of Medical Sciences Vol. 32, No. 3, 267271, September, 1983 HIJM 32-42 267 Lipase Activity of Guinea Pig Peritoneal Macrophages and Mycobacterial Lipase Inhibitor*) Katsuhiro KIYOT ANI,
Lesson Plan: Integumentary System
 Integumentary System The skin is no more separate from the brain than the surface of a lake is separate from its depths. They are two different locations in a continuous medium. To touch the surface is
Integumentary System The skin is no more separate from the brain than the surface of a lake is separate from its depths. They are two different locations in a continuous medium. To touch the surface is
Lysosomes, Peroxisomes and Centrioles. Hüseyin Çağsın
 Lysosomes, Peroxisomes and Centrioles Hüseyin Çağsın Lysosomes Outline Endosomes Molecule transport to the lysosomes Endocytosis Exocytosis Autophagy Vacuoles Peroxisomes Centrioles Lysosomes Lysosomes
Lysosomes, Peroxisomes and Centrioles Hüseyin Çağsın Lysosomes Outline Endosomes Molecule transport to the lysosomes Endocytosis Exocytosis Autophagy Vacuoles Peroxisomes Centrioles Lysosomes Lysosomes
PRODUCT: RNAzol BD for Blood May 2014 Catalog No: RB 192 Storage: Store at room temperature
 PRODUCT: RNAzol BD for Blood May 2014 Catalog No: RB 192 Storage: Store at room temperature PRODUCT DESCRIPTION. RNAzol BD is a reagent for isolation of total RNA from whole blood, plasma or serum of human
PRODUCT: RNAzol BD for Blood May 2014 Catalog No: RB 192 Storage: Store at room temperature PRODUCT DESCRIPTION. RNAzol BD is a reagent for isolation of total RNA from whole blood, plasma or serum of human
RayBio Cathepsin D Activity Assay Kit
 RayBio Cathepsin D Activity Assay Kit User Manual Version 1.0 October 1, 2014 RayBio Cathepsin D Activity Assay (Cat#: 68AT-CathD-S100) RayBiotech, Inc. We Provide You With Excellent Support And Service
RayBio Cathepsin D Activity Assay Kit User Manual Version 1.0 October 1, 2014 RayBio Cathepsin D Activity Assay (Cat#: 68AT-CathD-S100) RayBiotech, Inc. We Provide You With Excellent Support And Service
2.1.1 Biological Molecules
 2.1.1 Biological Molecules Relevant Past Paper Questions Paper Question Specification point(s) tested 2013 January 4 parts c and d p r 2013 January 6 except part c j k m n o 2012 June 1 part ci d e f g
2.1.1 Biological Molecules Relevant Past Paper Questions Paper Question Specification point(s) tested 2013 January 4 parts c and d p r 2013 January 6 except part c j k m n o 2012 June 1 part ci d e f g
THE SITE OF STEROL AND SQUALENE SYNTHESIS IN THE HUMAN SKIN123
 THE SITE OF STEROL AND SQUALENE SYNTHESIS IN THE HUMAN SKIN123 N. NICOLAIDES, PH.D. AND STEPHEN ROTHMAN, M.D. In earlier work (1) it was demonstrated that human scalp skin is an efficient organ for synthesizing
THE SITE OF STEROL AND SQUALENE SYNTHESIS IN THE HUMAN SKIN123 N. NICOLAIDES, PH.D. AND STEPHEN ROTHMAN, M.D. In earlier work (1) it was demonstrated that human scalp skin is an efficient organ for synthesizing
STAT3 (py705) (Human/Mouse/Rat) ELISA Kit
 STAT3 (py705) (Human/Mouse/Rat) ELISA Kit Catalog Number KA2175 96 assays Version: 01 Intended for research use only www.abnova.com I. INTRODUCTION STAT3 (py705) (Human/Mouse/Rat) ELISA (Enzyme-Linked
STAT3 (py705) (Human/Mouse/Rat) ELISA Kit Catalog Number KA2175 96 assays Version: 01 Intended for research use only www.abnova.com I. INTRODUCTION STAT3 (py705) (Human/Mouse/Rat) ELISA (Enzyme-Linked
Procaspase-3. Cleaved caspase-3. actin. Cytochrome C (10 M) Z-VAD-fmk. Procaspase-3. Cleaved caspase-3. actin. Z-VAD-fmk
 A HeLa actin - + + - - + Cytochrome C (1 M) Z-VAD-fmk PMN - + + - - + actin Cytochrome C (1 M) Z-VAD-fmk Figure S1. (A) Pan-caspase inhibitor z-vad-fmk inhibits cytochrome c- mediated procaspase-3 cleavage.
A HeLa actin - + + - - + Cytochrome C (1 M) Z-VAD-fmk PMN - + + - - + actin Cytochrome C (1 M) Z-VAD-fmk Figure S1. (A) Pan-caspase inhibitor z-vad-fmk inhibits cytochrome c- mediated procaspase-3 cleavage.
Mitochondrial Trifunctional Protein (TFP) Protein Quantity Microplate Assay Kit
 PROTOCOL Mitochondrial Trifunctional Protein (TFP) Protein Quantity Microplate Assay Kit DESCRIPTION Mitochondrial Trifunctional Protein (TFP) Protein Quantity Microplate Assay Kit Sufficient materials
PROTOCOL Mitochondrial Trifunctional Protein (TFP) Protein Quantity Microplate Assay Kit DESCRIPTION Mitochondrial Trifunctional Protein (TFP) Protein Quantity Microplate Assay Kit Sufficient materials
SensoLyte pnpp Alkaline Phosphatase Assay Kit *Colorimetric*
 SensoLyte pnpp Alkaline Phosphatase Assay Kit *Colorimetric* Catalog # 72146 Kit Size 500 Assays (96-well plate) Optimized Performance: This kit is optimized to detect alkaline phosphatase activity Enhanced
SensoLyte pnpp Alkaline Phosphatase Assay Kit *Colorimetric* Catalog # 72146 Kit Size 500 Assays (96-well plate) Optimized Performance: This kit is optimized to detect alkaline phosphatase activity Enhanced
Chapter 3 Cell Structures & Functions
 Biology 12 Name: Cell Biology Per: Date: Chapter 3 Cell Structures & Functions Complete using BC Biology 12, pages 62-107 Diagnostic Questions (mark using the answer key on page 527) 1. 2. 3. 4. 9. What
Biology 12 Name: Cell Biology Per: Date: Chapter 3 Cell Structures & Functions Complete using BC Biology 12, pages 62-107 Diagnostic Questions (mark using the answer key on page 527) 1. 2. 3. 4. 9. What
Differentially Localized Incorporation of Amino Acids. in Relation to Epidermal Keratinization in the Newborn Rat '
 Differentially Localized Incorporation of Amino Acids - in Relation to Epidermal Keratinization in the Newborn Rat ' KIMIE FUKUYAMA, TOSHIO NAKAMURA AND I. A. BERNSTEIN Department of Dermutology, Tokyo
Differentially Localized Incorporation of Amino Acids - in Relation to Epidermal Keratinization in the Newborn Rat ' KIMIE FUKUYAMA, TOSHIO NAKAMURA AND I. A. BERNSTEIN Department of Dermutology, Tokyo
PRODUCT INFORMATION & MANUAL
 PRODUCT INFORMATION & MANUAL Nuclear Extraction Kit NBP2-29447 Research use only. Not for diagnostic or therapeutic procedures. www.novusbio.com - P: 888.506.6887 - technical@novusbio.com Novus kits are
PRODUCT INFORMATION & MANUAL Nuclear Extraction Kit NBP2-29447 Research use only. Not for diagnostic or therapeutic procedures. www.novusbio.com - P: 888.506.6887 - technical@novusbio.com Novus kits are
Chapter 5: Integumentary System
 Chapter 5: Integumentary System I. Overview of the Integumentary System A. List the five major functions of the integumentary system: 1. 2. 3. 4. 5. Il. Skin A. Epidermis 1. The epidermis consists of 2.
Chapter 5: Integumentary System I. Overview of the Integumentary System A. List the five major functions of the integumentary system: 1. 2. 3. 4. 5. Il. Skin A. Epidermis 1. The epidermis consists of 2.
High-density Lipoprotein Cholesterol (HDL-C) Assay Kit
 (FOR RESEARCH USE ONLY. DO NOT USE IT IN CLINICAL DIAGNOSIS!) High-density Lipoprotein Cholesterol (HDL-C) Assay Kit (Double reagents) Catalog No: E-BC-K221 Method: Colorimetric method Specification: 96T
(FOR RESEARCH USE ONLY. DO NOT USE IT IN CLINICAL DIAGNOSIS!) High-density Lipoprotein Cholesterol (HDL-C) Assay Kit (Double reagents) Catalog No: E-BC-K221 Method: Colorimetric method Specification: 96T
Procine sphingomyelin ELISA Kit
 Procine sphingomyelin ELISA Kit For the quantitative in vitro determination of Procine sphingomyelin concentrations in serum - plasma - celiac fluid - tissue homogenate - body fluid FOR LABORATORY RESEARCH
Procine sphingomyelin ELISA Kit For the quantitative in vitro determination of Procine sphingomyelin concentrations in serum - plasma - celiac fluid - tissue homogenate - body fluid FOR LABORATORY RESEARCH
Lysosomes. Gr: lysis solution, soma body. Membrane bounded vesicles. Usually round ovoid or irregular electron dense bodies m.
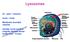 Lysosomes Gr: lysis solution, soma body Membrane bounded vesicles Usually round ovoid or irregular electron dense bodies 0.05 0.5 m. Lysosomes No. varies from a few to several hundred per cell, in different
Lysosomes Gr: lysis solution, soma body Membrane bounded vesicles Usually round ovoid or irregular electron dense bodies 0.05 0.5 m. Lysosomes No. varies from a few to several hundred per cell, in different
Work-flow: protein sample preparation Precipitation methods Removal of interfering substances Specific examples:
 Dr. Sanjeeva Srivastava IIT Bombay Work-flow: protein sample preparation Precipitation methods Removal of interfering substances Specific examples: Sample preparation for serum proteome analysis Sample
Dr. Sanjeeva Srivastava IIT Bombay Work-flow: protein sample preparation Precipitation methods Removal of interfering substances Specific examples: Sample preparation for serum proteome analysis Sample
Histochemical Demonstration of Certain Hydrolytic Enzymes within Cytoplasmic Particles of Botrytis cinerea Fr.
 J. gen. Microbiol. (1968), 52, 67-75 With 2 plates Printed in Great Britain 67 Histochemical Demonstration of Certain Hydrolytic Enzymes within Cytoplasmic Particles of Botrytis cinerea Fr. By D. PITT
J. gen. Microbiol. (1968), 52, 67-75 With 2 plates Printed in Great Britain 67 Histochemical Demonstration of Certain Hydrolytic Enzymes within Cytoplasmic Particles of Botrytis cinerea Fr. By D. PITT
ab Lipid Peroxidation (MDA) Assay kit (Colorimetric/ Fluorometric)
 Version 10b Last updated 19 December 2018 ab118970 Lipid Peroxidation (MDA) Assay kit (Colorimetric/ Fluorometric) For the measurement of Lipid Peroxidation in plasma, cell culture and tissue extracts.
Version 10b Last updated 19 December 2018 ab118970 Lipid Peroxidation (MDA) Assay kit (Colorimetric/ Fluorometric) For the measurement of Lipid Peroxidation in plasma, cell culture and tissue extracts.
Effects of Listeria monocytogenes Hemolysin on Phagocytic Cells and Lysosomes
 INFECTION AND IMMUNITy, Apr. 197, p. 356-362 Copyright 197 American Society for Microbiology Vol. 1, No. 4 Printed in U.S.A. Effects of Listeria monocytogenes Hemolysin on Phagocytic Cells and Lysosomes
INFECTION AND IMMUNITy, Apr. 197, p. 356-362 Copyright 197 American Society for Microbiology Vol. 1, No. 4 Printed in U.S.A. Effects of Listeria monocytogenes Hemolysin on Phagocytic Cells and Lysosomes
OCHROMONAS MALHAMENSIS
 THE INFLUENCE OF THE MODE OF NUTRITION ON THE DIGESTIVE SYSTEM OF OCHROMONAS MALHAMENSIS H. J. STOLTZE, N. S. T. LUI, O. R. ANDERSON, and O. A. ROELS From the Marine Biology Division of the Lamont-Doherty
THE INFLUENCE OF THE MODE OF NUTRITION ON THE DIGESTIVE SYSTEM OF OCHROMONAS MALHAMENSIS H. J. STOLTZE, N. S. T. LUI, O. R. ANDERSON, and O. A. ROELS From the Marine Biology Division of the Lamont-Doherty
I mutants accumulate pyruvate when growing in the presence of isoleucine and
 THE iv-3 MUTANTS OF NEUROSPORA CRASSA 11. ACTIVITY OF ACETOHYDROXY ACID SYNTHETASE DINA F. CAROLINE, ROY W. HARDINGZ, HOMARE KUWANA3, T. SATYANARAYANA AND R.P. WAGNER4 Genetics Foundation, The University
THE iv-3 MUTANTS OF NEUROSPORA CRASSA 11. ACTIVITY OF ACETOHYDROXY ACID SYNTHETASE DINA F. CAROLINE, ROY W. HARDINGZ, HOMARE KUWANA3, T. SATYANARAYANA AND R.P. WAGNER4 Genetics Foundation, The University
Cathepsin D Activity Assay Kit
 Cathepsin D Activity Assay Kit Catalog Number KA0767 100 assays Version: 04 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Background... 3 General Information... 4 Materials
Cathepsin D Activity Assay Kit Catalog Number KA0767 100 assays Version: 04 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Background... 3 General Information... 4 Materials
Basic Structure of a Cell
 Basic Structure of a Cell 1 Introduction to Cells Cells are the basic units of organisms Cells can only be observed under microscope Basic types of cells: Animal Cell Plant Cell Bacterial Cell 2 Number
Basic Structure of a Cell 1 Introduction to Cells Cells are the basic units of organisms Cells can only be observed under microscope Basic types of cells: Animal Cell Plant Cell Bacterial Cell 2 Number
4. Lysosomes, Smooth Endoplasmic Reticulum, Mitochondria, and Inclusions
 4. Lysosomes, Smooth Endoplasmic Reticulum, Mitochondria, and Inclusions Undergraduate Graduate Histology Lecture Series Larry Johnson, Professor Veterinary Integrative Biosciences Texas A&M University
4. Lysosomes, Smooth Endoplasmic Reticulum, Mitochondria, and Inclusions Undergraduate Graduate Histology Lecture Series Larry Johnson, Professor Veterinary Integrative Biosciences Texas A&M University
Biochemical evaluation of the corneal endothelium
 Isolation of the plasma membrane from corneal endothelial cells Z. Suzanne Zam, James Cerda,* and Frank M. Polack The plasma membranes of normal rabbit endothelail cells were isolated by the use of an
Isolation of the plasma membrane from corneal endothelial cells Z. Suzanne Zam, James Cerda,* and Frank M. Polack The plasma membranes of normal rabbit endothelail cells were isolated by the use of an
Explain the reason for this difference in resolving power.
 1. (a) An electron microscope has a much greater resolving power than an optical microscope. (i) Explain the meaning of the term resolving power. Explain the reason for this difference in resolving power.
1. (a) An electron microscope has a much greater resolving power than an optical microscope. (i) Explain the meaning of the term resolving power. Explain the reason for this difference in resolving power.
The lipoprotein lipase of cow s milk
 J. Lipid Research, April, 1962 Volume 3. Number 2 The lipoprotein lipase of cow s milk EDWARD D. KORN Laboratory of Cellular Physiology and Metabolism, National Heart stitute, Bethesda 14, Maryland [Itcceived
J. Lipid Research, April, 1962 Volume 3. Number 2 The lipoprotein lipase of cow s milk EDWARD D. KORN Laboratory of Cellular Physiology and Metabolism, National Heart stitute, Bethesda 14, Maryland [Itcceived
BIOLOGICAL MOLECULES REVIEW-UNIT 1 1. The factor being tested in an experiment is the A. data. B. variable. C. conclusion. D. observation. 2.
 BIOLOGICAL MOLECULES REVIEW-UNIT 1 1. The factor being tested in an experiment is the A. data. B. variable. C. conclusion. D. observation. 2. A possible explanation for an event that occurs in nature is
BIOLOGICAL MOLECULES REVIEW-UNIT 1 1. The factor being tested in an experiment is the A. data. B. variable. C. conclusion. D. observation. 2. A possible explanation for an event that occurs in nature is
ATTACHMENT OF RIBOSOMES TO MEMBRANES DURING POLYSOME FORMATION IN MOUSE SARCOMA 180 CELLS
 Published Online: 1 June, 1971 Supp Info: http://doi.org/10.1083/jcb.49.3.683 Downloaded from jcb.rupress.org on November 2, 2018 ATTACHMENT OF RIBOSOMES TO MEMBRANES DURING POLYSOME FORMATION IN MOUSE
Published Online: 1 June, 1971 Supp Info: http://doi.org/10.1083/jcb.49.3.683 Downloaded from jcb.rupress.org on November 2, 2018 ATTACHMENT OF RIBOSOMES TO MEMBRANES DURING POLYSOME FORMATION IN MOUSE
ab ATP Synthase Enzyme Activity Microplate Assay Kit
 ab109714 ATP Synthase Enzyme Activity Microplate Assay Kit Instructions for Use For the quantitative measurement of ATP Synthase activity in samples from Human, Rat and Cow This product is for research
ab109714 ATP Synthase Enzyme Activity Microplate Assay Kit Instructions for Use For the quantitative measurement of ATP Synthase activity in samples from Human, Rat and Cow This product is for research
STAT3 (py705)/ Pan STAT3 (Human/Mouse/Rat) ELISA Kit
 STAT3 (py705)/ Pan STAT3 (Human/Mouse/Rat) ELISA Kit Catalog Number KA2176 96 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Principle of the Assay...
STAT3 (py705)/ Pan STAT3 (Human/Mouse/Rat) ELISA Kit Catalog Number KA2176 96 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Principle of the Assay...
ab CytoPainter Golgi/ER Staining Kit
 ab139485 CytoPainter Golgi/ER Staining Kit Instructions for Use Designed to detect Golgi bodies and endoplasmic reticulum by microscopy This product is for research use only and is not intended for diagnostic
ab139485 CytoPainter Golgi/ER Staining Kit Instructions for Use Designed to detect Golgi bodies and endoplasmic reticulum by microscopy This product is for research use only and is not intended for diagnostic
New aspect of hepatic nuclear glycogenosis
 J. clin. Path. (1968), 21, 19 New aspect of hepatic nuclear glycogenosis in diabetes1 F. CARAMIA, F. G. GHERGO, C. BRANCIARI, AND G. MENGHINI From the Institute of General Pathology, University of Rome,
J. clin. Path. (1968), 21, 19 New aspect of hepatic nuclear glycogenosis in diabetes1 F. CARAMIA, F. G. GHERGO, C. BRANCIARI, AND G. MENGHINI From the Institute of General Pathology, University of Rome,
A COMPARISON OF MEMBRANE FRACTURE FACES OF FIXED AND UNFIXED GLYCERINATED TISSUE
 J. Cell Set. 21, 437-448 (1976) 43-7 Printed in Great Britain A COMPARISON OF MEMBRANE FRACTURE FACES OF FIXED AND UNFIXED GLYCERINATED TISSUE A. S. BREATHNACH, M. GROSS, B. MARTIN AND C. STOLINSKI Department
J. Cell Set. 21, 437-448 (1976) 43-7 Printed in Great Britain A COMPARISON OF MEMBRANE FRACTURE FACES OF FIXED AND UNFIXED GLYCERINATED TISSUE A. S. BREATHNACH, M. GROSS, B. MARTIN AND C. STOLINSKI Department
ELECTRON MICROSCOPY OF A SMALL PIGMENTED CUTANEOUS LESION*
 ELECTRON MICROSCOPY OF A SMALL PIGMENTED CUTANEOUS LESION* The description of the lesion in the title of this rcport is intentionally non-committal. Diagnosed clinically as a lentigo, it was removed as
ELECTRON MICROSCOPY OF A SMALL PIGMENTED CUTANEOUS LESION* The description of the lesion in the title of this rcport is intentionally non-committal. Diagnosed clinically as a lentigo, it was removed as
ab Histone Deacetylase (HDAC) Activity Assay Kit (Fluorometric)
 ab156064 Histone Deacetylase (HDAC) Activity Assay Kit (Fluorometric) Instructions for Use For the quantitative measurement of Histone Deacetylase activity in cell lysates This product is for research
ab156064 Histone Deacetylase (HDAC) Activity Assay Kit (Fluorometric) Instructions for Use For the quantitative measurement of Histone Deacetylase activity in cell lysates This product is for research
LYSOSOMAL ENZYMES OF RAT
 Published Online: 1 November, 1964 Supp Info: http://doi.org/10.1083/jcb.23.2.233 Downloaded from jcb.rupress.org on April 29, 2018 LYSOSOMAL ENZYMES OF RAT INTESTINAL MUCOSA LICHU HSU, Ph.D., and A. L.
Published Online: 1 November, 1964 Supp Info: http://doi.org/10.1083/jcb.23.2.233 Downloaded from jcb.rupress.org on April 29, 2018 LYSOSOMAL ENZYMES OF RAT INTESTINAL MUCOSA LICHU HSU, Ph.D., and A. L.
STAT1 (ps727) (Human/Mouse) ELISA Kit
 STAT1 (ps727) (Human/Mouse) ELISA Kit Catalog Number KA2171 96 assays Version: 01 Intended for research use only www.abnova.com I. INTRODUCTION STAT1 (ps727) (Human/Mouse) ELISA (Enzyme-Linked Immunosorbent
STAT1 (ps727) (Human/Mouse) ELISA Kit Catalog Number KA2171 96 assays Version: 01 Intended for research use only www.abnova.com I. INTRODUCTION STAT1 (ps727) (Human/Mouse) ELISA (Enzyme-Linked Immunosorbent
Mammalian Melanosomal Proteins: Characterization by Polyacrylamide Gel Electrophoresis
 YALE JOURNAL OF BIOLOGY AND MEDICINE 46, 553-559 (1973) Mammalian Melanosomal Proteins: Characterization by Polyacrylamide Gel Electrophoresis VINCENT J. HEARING AND MARVIN A. LUTZNER Dermatology Branch,
YALE JOURNAL OF BIOLOGY AND MEDICINE 46, 553-559 (1973) Mammalian Melanosomal Proteins: Characterization by Polyacrylamide Gel Electrophoresis VINCENT J. HEARING AND MARVIN A. LUTZNER Dermatology Branch,
Science that studies adverse skin effects and the substances that produce them
 Science that studies adverse skin effects and the substances that produce them Leena A. Nylander-French, Ph.D., CIH 159 Rosenau Tel: 966.3826 E-mail: leena_french@unc.edu Occupational skin diseases are
Science that studies adverse skin effects and the substances that produce them Leena A. Nylander-French, Ph.D., CIH 159 Rosenau Tel: 966.3826 E-mail: leena_french@unc.edu Occupational skin diseases are
Cathepsin K Activity Assay Kit (Fluorometric)
 ab65303 Cathepsin K Activity Assay Kit (Fluorometric) Instructions for Use For the rapid, sensitive and accurate measurement of Cathepsin K activity in various samples. This product is for research use
ab65303 Cathepsin K Activity Assay Kit (Fluorometric) Instructions for Use For the rapid, sensitive and accurate measurement of Cathepsin K activity in various samples. This product is for research use
Biology 2180 Laboratory #3. Enzyme Kinetics and Quantitative Analysis
 Biology 2180 Laboratory #3 Name Introduction Enzyme Kinetics and Quantitative Analysis Catalysts are agents that speed up chemical processes and the catalysts produced by living cells are called enzymes.
Biology 2180 Laboratory #3 Name Introduction Enzyme Kinetics and Quantitative Analysis Catalysts are agents that speed up chemical processes and the catalysts produced by living cells are called enzymes.
CoQ10(Coenzyme Q10) ELISA Kit
 CoQ10(Coenzyme Q10) ELISA Kit Catalogue No.: EU0196 Size: 48T/96T Reactivity: Universal Detection Range: 0.781-50ng/ml Sensitivity:
CoQ10(Coenzyme Q10) ELISA Kit Catalogue No.: EU0196 Size: 48T/96T Reactivity: Universal Detection Range: 0.781-50ng/ml Sensitivity:
Synopsis. Received March 2, adrenaline. Mosinger and Kujalova (1964) reported that adrenaline-induced lipolysis
 Studies on Reduction of Lipolysis in Adipose Tissue on Freezing and Thawing YASUSHI SAITO1, NoBUO MATSUOKA1, AKIRA KUMAGAI1, HIROMICHI OKUDA2, AND SETSURO FUJII3 Chiba University, Chiba 280, Japan, 2Department
Studies on Reduction of Lipolysis in Adipose Tissue on Freezing and Thawing YASUSHI SAITO1, NoBUO MATSUOKA1, AKIRA KUMAGAI1, HIROMICHI OKUDA2, AND SETSURO FUJII3 Chiba University, Chiba 280, Japan, 2Department
BIOLOGY 111. CHAPTER 2: The Chemistry of Life Biological Molecules
 BIOLOGY 111 CHAPTER 2: The Chemistry of Life Biological Molecules The Chemistry of Life : Learning Outcomes 2.4) Describe the significance of carbon in forming the basis of the four classes of biological
BIOLOGY 111 CHAPTER 2: The Chemistry of Life Biological Molecules The Chemistry of Life : Learning Outcomes 2.4) Describe the significance of carbon in forming the basis of the four classes of biological
Principles of Anatomy and Physiology
 Principles of Anatomy and Physiology 14 th Edition CHAPTER 5 The Integumentary System Introduction The organs of the integumentary system include the skin and its accessory structures including hair, nails,
Principles of Anatomy and Physiology 14 th Edition CHAPTER 5 The Integumentary System Introduction The organs of the integumentary system include the skin and its accessory structures including hair, nails,
