PFIZER INC. Study Initiation Date and Primary Completion or Completion Dates: March 1991 to August 1991
|
|
|
- Ernest Boyd
- 5 years ago
- Views:
Transcription
1 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography. PROPRIETARY DRUG NAME /GENERIC DRUG NAME: Fasigyn /Tinidazole PROTOCOL NO.: TND-II PROTOCOL TITLE: A Randomized Assesser-blind, Parallel-group Comparison of Fasigyn DS Tablets (1 BD for 3 days) and Entamizole Forte Tablets (1 TID for 5 days) in Intestinal Amebiasis with Colonic symptoms. Study Centre: One centre in India Study Initiation Date and Primary Completion or Completion Dates: March 1991 to August 1991 Phase of Development: Phase 3 Study Objective(s): To compare the efficacy and tolerability of Fasigyn DS (1 tablet BD for 3 days) and of Entamizole (1 tablet TID for 5 days) in subjects with intestinal amebiasis and colonic symptoms. METHODS Study Design: This was a randomised, assessor-blind, parallel-group trial to compare the efficacy and tolerability of Fasigyn DS and Entamizole in subjects having colonic symptoms and Entamoeba histolytica (trophozoites, cysts, or both) in their stools. The study was carried out between March and August The subjects were required to make at least 3 visits. The first study visit was to collect information on subject history and symptoms, to examine the stools and for the subject to collect the study drug. The second visit (one week later) was for reporting compliance and adverse events (if any). The third visit (3 weeks later), was the final assessment of symptoms and stool examination. In all the visits, the microscopist was unaware of the treatment given to the subject. Number of Subjects (Planned and Analyzed): In total, 100 subjects were screened and enrolled in the study. Diagnosis and Main Criteria for Inclusion: The study included subjects of either sex, aged years, with intestinal amebiasis that was diagnosed by microscopic examination of stools by formol-ether concentration method for trophozoites, cysts, or both, of E histolytica, and symptoms referable to the colon. The subjects were not to have taken any amebicidal drug for at least one month before entering the trial and should not have had any history of irritable bowel syndrome (IBS), e.g. repeated episodes of colonic dysfunction with only mucus in stools, and with temporary relief of symptoms after a variety of specific and/or Page 1
2 nonspecific therapy. Subjects were also to have a high likelihood of returning for follow-up after 1 week and 4 weeks. Study Treatment: Overall, 50 subjects were given Fasigyn DS, 1 tablet twice daily for 3 days, after dinner, immediately after the first stool examination. Entamizole was given to the other 50 subjects at a dose of 1 tablet three times daily for 5 days, after meals, immediately after the first stool examination. Allocation of the subjects to the regimens was randomised in blocks of 10 subjects and recorded on each subject s form. Efficacy Evaluations: Primary Efficacy Evaluation: The primary efficacy endpoint was evaluated by stool examination at the third visit. The parasitic response was assessed as cure if the stool was negative for E histolytica and failure if it was positive. Secondary Efficacy Evaluation: The secondary efficacy endpoint was evaluated by assessing the symptomatic relief of the subjects. The clinical response was assessed as relieved or not relieved. Other Miscellaneous Efficacy Evaluation: Compliance was evaluated by assessing the number of subjects missing one or more tablets, and the number of tablets missed by the noncomplying subjects. Non-compliance and its extent were correlated with the parasitic response. Safety Evaluations: Safety was determined by the following assessments: Monitoring adverse events and their outcome. Severity and duration of adverse events, as well as their relationship to the study drug. Statistical Methods: Comparison between groups was done by means of student s t-test for measured variables and chi-square test for attributes. RESULTS Subject Disposition and Demography: In total, 100 subjects were screened and assigned study treatment. In total, 50 subjects received Fasigyn DS, 1 tablet twice daily for 3 days and another 50 subjects received Entamizole 1 tablet thrice daily for 5 days. One subject in the Entamizole group was excluded from the analysis when his symptoms returned on Day 12 after initial relief. The subject s stool was positive for E. histolytica. The subject took clioquinol tablets for 4 days. The subject s stool was negative for E. histolytica on Day 30; however, the subject was still excluded from the analysis so as not to confound the results. Demography characteristics are summarized in Table 1. Page 2
3 Table 1. Demographic Characteristics of the Subjects Characteristic Fasigyn (N=50) Entamizole (N=49) Sex Male Female Age (yr) Mean SEM Wt (Kg) Mean SEM N=Total number of subjects; Yr=Year; Wt=Weight; Kg=Kilogram; SEM=Standard error of mean. Prevalence of symptoms and parasites are summarized in Table 2 and Table 3, respectively. Pain in the abdomen was significantly more frequent in the Fasigyn group (100% vs 78%, p<0.01). All subjects had cysts of ameba in the stools, but trophozoites were not found. The other parasites present were A. lumricoides, hookworm and G. lamblia. The Fasigyn group had a significantly lower prevalence of A lumbricoides (32% vs 57%, p<0.01). Table 2. Prevalence of Symptoms Symptom Fasigyn (N=50) Entamizole (N=49) Pain in abdomen 50 (100%) 38 (78%) Diarrhea 33 (66%) 41 (84%) Constipation 41 (82%) 38 (78%) Mucus in stools 36 (72%) 39 (80%) Blood in stool 0 (0%) 3 (6%) Tenderness on liver 0 (0%) 5 (10%) p<0.05 for difference between groups. Table 3. Prevalence of Parasites in Stools Parasite Fasigyn (N=50) Entamizole (N=49) E. histolytica Trophozoites 0 0 Cysts 50 (100%) 49 (100%) A. lumbricoides* 16 (32%) 28 (57%) Hookworm 13 (26%) 12 (24%) G. lamblia 3 (6%) 6 (12%) p<0.01 for difference between groups. Efficacy Results: Primary Efficacy Endpoint: On Day 30, the stools of 46 subjects in the Fasigyn group were free from E. histolytica, which showed that the cure rate was 92% (46/50). In the Entamizole group, E. histolytica was absent from the stools of 38 subjects, which showed that the cure rate was 76% (38/50). The difference of 16% between the groups narrowly failed to reach significance (p=0.08). Analysis of the results in compliers revealed that Fasigyn cured 92% of subjects (46/50), whereas Entamizole cured 85% of subjects (33/39) (Table 4). Page 3
4 Table 4. Parasitic Cure Response Fasigyn Entamizole Cure 46/50 (92%) 38/49 (78%) Failure 4/50 (8%) 11/49 (22%) For difference between groups, p=0.08 In the Fasigyn group, 3 subjects who had giardiasis were cured. However, in the Entamizole group, 2/6 subjects who had giardiasis were cured. Secondary Efficacy Endpoint: The subjects showed symptomatic relief in both groups (Table 5). Relief of pain in the abdomen, constipation, and mucus in the stools was significantly higher in the Fasigyn group (p<0.05). Table 5. Symptomatic Relief Symptom Fasigyn (N=50) Entamizole (N=49) Pain in abdomen 46/50 (92%) 26/38 (68%) Diarrhea 33/33 (100%) 39/41 (95%) Constipation 38/41 (93%) 28/38 (74%) Mucus in stools 36/36 (100%) 30/39 (77%) p<0.05 and p<0.01 for difference between groups. N=total number of subjects Other Miscellaneous Efficacy Endpoint: In the Fasigyn group, subjects took all tablets as prescribed and thus compliance was 100%. In the Entamizole group, 10 subjects (20%) failed to take one or more tablets. The difference in compliance between the two groups was significant (p<0.01). The average number of missed tablets was 3.1 (range: 1 to 6), which represented 20% of the doses (range: 7% to 40%). Out of 10 subjects in the Entamizole group who failed to comply with the prescribed dosage regimen, 5 subjects were cured and 5 failed. Further analysis showed that the average number of tablets missed by those who failed was 4.2 (28% of doses), whereas the average number of tablets missed by those who cured was 2.0 (14% of doses), and the difference narrowly failed to reach significance (p=0.08). Safety Results: The safety results are summarized in Table 6. The most frequent adverse event during the study was altered taste (11 subjects [22%] in the Fasigyn group and 3 subjects [6%] in the Entamizole group). The difference between the treatment groups in the incidence of altered taste was not significant (p=0.08). No subjects in the Fasigyn group defaulted, probably because the treatment was over in just 3 days. Page 4
5 Table 6. Summary of Adverse Experiences Adverse Event Fasigyn (N=50) Entamizole (N=49) Altered taste 11 (22%) 3 (6%) Nausea 2 0 Headache 1 0 Gastritis 1 0 Constipation 0 1 Drowsiness 0 1 p=0.08 for difference between groups. CONCLUSION(S): In cyst-passing subjects of amebiasis with colonic symptoms, Fasigyn 1 g daily for 3 days was as effective as, but operationally more effective than, Entamizole 1 tablet three times daily for 5 days. While the Fasigyn regimen was conducive to good compliance, the Entamizole regimen predisposes to non-compliance to the extent of 20%, which was probably the reason for its lower use-effectiveness as revealed in this study. Page 5
PFIZER INC. Study Initiation Date and Completion Dates: Information not available (Date of Statistical Report: 16 May 2004)
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. Study Initiation Date and Completion Dates: 09 March 2000 to 09 August 2001.
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. THERAPEUTIC AREA AND FDA APPROVED INDICATIONS: See USPI.
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
Ameba has two stages of development: cyst and trophozoite
 Amebiasis A parasitic disease of worldwide public health importance Second to malaria in mortality due to protozoan parasites Invasive amebiasis results in up to 100,000 deaths / year Amebiasis is infection
Amebiasis A parasitic disease of worldwide public health importance Second to malaria in mortality due to protozoan parasites Invasive amebiasis results in up to 100,000 deaths / year Amebiasis is infection
PFIZER INC. THERAPEUTIC AREA AND FDA APPROVED INDICATIONS: See USPI.
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. GENERIC DRUG NAME and/or COMPOUND NUMBER: 5% Spironolactone Cream/ PF
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. THERAPEUTIC AREA AND FDA APPROVED INDICATIONS: See United States Package Insert (USPI)
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. THERAPEUTIC AREA AND FDA APPROVED INDICATIONS: See United States Package Insert (USPI)
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. Study Initiation Date and Primary Completion or Completion Dates: 11 November 1998 to 17 September 1999
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. Study Center(s): A total of 6 centers took part in the study, including 2 in France and 4 in the United States.
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
Bacillary Dysentery (Shigellosis)
 Bacillary Dysentery (Shigellosis) An acute bacterial disease involving the large and distal small intestine, caused by the bacteria of the genus shigella. Infectious agent Shigella is comprised of four
Bacillary Dysentery (Shigellosis) An acute bacterial disease involving the large and distal small intestine, caused by the bacteria of the genus shigella. Infectious agent Shigella is comprised of four
Amoebiasis. (Amoebic dysentery)
 Amoebiasis (Amoebic dysentery) Causative agent: Entamoeba histolytica Amoebiasis Harbouring of protozoa E. histolytica inside the body with or without disease only 10% of infected develop disease two types
Amoebiasis (Amoebic dysentery) Causative agent: Entamoeba histolytica Amoebiasis Harbouring of protozoa E. histolytica inside the body with or without disease only 10% of infected develop disease two types
PFIZER INC. Study Centres: A total of 8 centres in France took part in the study.
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
Study No.: Title: Rationale: Phase: Study Period: Study Design: Centres: Indication: Treatment: Objectives: Primary Outcome/Efficacy Variable:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
SYNOPSIS. Co-ordinating investigator Not applicable. Study centre(s) This study was conducted in Japan (57 centres).
 Drug product: Symbicort Turbuhaler Drug substance(s): ST (Symbicort Turbuhaler ) Edition No.: 1.0 Study code: D5890C00010 Date: 15 March 2007 SYNOPSIS An 8-week, randomised, double blind, parallel-group,
Drug product: Symbicort Turbuhaler Drug substance(s): ST (Symbicort Turbuhaler ) Edition No.: 1.0 Study code: D5890C00010 Date: 15 March 2007 SYNOPSIS An 8-week, randomised, double blind, parallel-group,
Clinical Study Synopsis
 Clinical Study Synopsis This document is not intended to replace the advice of a healthcare professional and should not be considered as a recommendation. Patients should always seek medical advice before
Clinical Study Synopsis This document is not intended to replace the advice of a healthcare professional and should not be considered as a recommendation. Patients should always seek medical advice before
PhRMA Clinical Study Synopsis Protocol CTN / (A /A ) 21 August 2006 Final PFIZER INC.
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert.
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. PROPRIETARY DRUG NAME / GENERIC DRUG NAME: Advil / Ibuprofen
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. PROPRIETARY DRUG NAME / GENERIC DRUG NAME: Advil / Ibuprofen
Study No.: Title: Rationale: Phase: Study Period: Study Design: Centres: Indication: Treatment: Objectives: Primary Outcome/Efficacy Variable:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Study No: Title: Rationale: . Phase: Study Period: Study Design: Centres: Indication: Treatment: Objectives: Primary Outcome/Efficacy Variable:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
PFIZER INC. PROPRIETARY DRUG NAME /GENERIC DRUG NAME: Cerebyx / Fosphenytoin Sodium
 PFIZER INC These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
METRONIDAZOLE. General Monograph, Antibacterial - Antiprotozoal
 METRONIDAZOLE General Monograph, Antibacterial - Antiprotozoal Pharmacology: Metronidazole is amebicidal, trichomonacidal and bactericidal. A chemically reactive, reduced form of metronidazole is thought
METRONIDAZOLE General Monograph, Antibacterial - Antiprotozoal Pharmacology: Metronidazole is amebicidal, trichomonacidal and bactericidal. A chemically reactive, reduced form of metronidazole is thought
1. Developed January Revised September 2017; August 2015; December 2013; February 2012; April 2010; March 2007.
 Texas Vendor Drug Program Drug Use Criteria: Nitazoxanide (Alinia ) Publication History 1. Developed January 2007. 2. Revised September 2017; August 2015; December 2013; February 2012; April 2010; March
Texas Vendor Drug Program Drug Use Criteria: Nitazoxanide (Alinia ) Publication History 1. Developed January 2007. 2. Revised September 2017; August 2015; December 2013; February 2012; April 2010; March
PFIZER INC. THERAPEUTIC AREA AND FDA APPROVED INDICATIONS: See USPI
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
Study No.: Title: Rationale: Phase: Study Period: Study Design: Centres: Indication: Treatment: Objectives: Primary Outcome/Efficacy Variable:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
SYNOPSIS. Study Coordinator. Study centre(s)
 Drug product: Seroquel Drug substance(s): Quetiapine Document No.: 1 Edition No.: 1 Study code: D1449C00005 Date: 02 January 2007 SYNOPSIS A Randomized, Parallel Group, Open Trial Examining the Safety,
Drug product: Seroquel Drug substance(s): Quetiapine Document No.: 1 Edition No.: 1 Study code: D1449C00005 Date: 02 January 2007 SYNOPSIS A Randomized, Parallel Group, Open Trial Examining the Safety,
Labeled Uses: Treatment of Clostiridum Difficile associated diarrhea (CDAD)
 Brand Name: Dificid Generic Name: fidaxomicin Manufacturer 1,2,3,4,5 : Optimer Pharmaceuticals, Inc. Drug Class 1,2,3,4,5 : Macrolide Antibiotic Uses 1,2,3,4,5 : Labeled Uses: Treatment of Clostiridum
Brand Name: Dificid Generic Name: fidaxomicin Manufacturer 1,2,3,4,5 : Optimer Pharmaceuticals, Inc. Drug Class 1,2,3,4,5 : Macrolide Antibiotic Uses 1,2,3,4,5 : Labeled Uses: Treatment of Clostiridum
PFIZER INC. Study Initiation Date: 15 June 1995; Completion Date: 22 April 1996
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. PROPRIETARY DRUG NAME /GENERIC DRUG NAME: Cerebyx /fosphenytoin
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. PROPRIETARY DRUG NAME /GENERIC DRUG NAME: Cerebyx /fosphenytoin
Parasites List of Pinworm (Enterobius vermicularis) Giardia ( Giardia lamblia Coccidia ( Cryptosporidium
 Parasites List of Pinworm (Enterobius vermicularis) Pinworm, also known as seatworm or threadworm, is one of the most prevalent intestinal parasites in the United States, with approximately 40 million
Parasites List of Pinworm (Enterobius vermicularis) Pinworm, also known as seatworm or threadworm, is one of the most prevalent intestinal parasites in the United States, with approximately 40 million
PRESCRIBING INFORMATION PRODUCT MONOGRAPH HUMATIN*CAP
 PRESCRIBING INFORMATION PRODUCT MONOGRAPH HUMATIN*CAP (Paromomycin Sulfate Capsules MANUFACTURER STANDARD) 250 mg Capsules ANTIBIOTIC DATE OF PREPARATION January 08, 2001 8250 Décarie Blvd, suite 110 Montréal,
PRESCRIBING INFORMATION PRODUCT MONOGRAPH HUMATIN*CAP (Paromomycin Sulfate Capsules MANUFACTURER STANDARD) 250 mg Capsules ANTIBIOTIC DATE OF PREPARATION January 08, 2001 8250 Décarie Blvd, suite 110 Montréal,
Sponsor / Company: Sanofi Drug substance(s): AMARYL M (1/250 mg) / HOE490
 These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor / Company: Sanofi Drug substance(s):
These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor / Company: Sanofi Drug substance(s):
PFIZER INC. THERAPEUTIC AREA AND FDA APPROVED INDICATIONS: See USPI.
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert.
 Public Disclosure Synopsis Protocol A7772 September 25 Final PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert.
Public Disclosure Synopsis Protocol A7772 September 25 Final PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert.
Anton van Leeuwenhoek. Protozoa: This is what he saw in his own stool sample. Morphology 10/14/2009. Protozoans that cause diarrheal disease
 Access to safe drinking water is everyone s right Anton van Leeuwenhoek Protozoa: Protozoans that cause diarrheal disease This is what he saw in his own stool sample 1. Giardia lamblia 2. Entameba histolytica
Access to safe drinking water is everyone s right Anton van Leeuwenhoek Protozoa: Protozoans that cause diarrheal disease This is what he saw in his own stool sample 1. Giardia lamblia 2. Entameba histolytica
Study No.: Title: Rationale: Phase: Study Period: Study Design: Centers Indication: Treatment: Objectives: Primary Outcome/Efficacy Variable:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Access to safe drinking water is everyone s right. Protozoans that cause diarrheal disease
 Access to safe drinking water is everyone s right Protozoa: Protozoans that cause diarrheal disease 1. Giardia lamblia 2. Entameba histolytica 3. Cryptosporidium parvum 4. Cyclospora cayetanensis 1 Giardia
Access to safe drinking water is everyone s right Protozoa: Protozoans that cause diarrheal disease 1. Giardia lamblia 2. Entameba histolytica 3. Cryptosporidium parvum 4. Cyclospora cayetanensis 1 Giardia
3-Mutualism: It is a relationship between two. organisms; one called. or harmless. 2-Commensalism: It is a relationship in which one
 Host-Parasite relationships Symbiosis: It is the permanent relationship between two dissimilar organisms which depending each others. They are three types 1- Parasitism : It is a relationship between two
Host-Parasite relationships Symbiosis: It is the permanent relationship between two dissimilar organisms which depending each others. They are three types 1- Parasitism : It is a relationship between two
ccess safe drinking wa r is everyone s right Protozoans that cause diarrheal disease
 ccess safe drinking wa r is everyone s right Protozoa: Protozoans that cause diarrheal disease 1. Giardia lamblia 2. Entameba histolytica 3. Cryptosporidium parvum 4. Cyclospora cayetanensis 1 Giardia
ccess safe drinking wa r is everyone s right Protozoa: Protozoans that cause diarrheal disease 1. Giardia lamblia 2. Entameba histolytica 3. Cryptosporidium parvum 4. Cyclospora cayetanensis 1 Giardia
IRBES_R_04320 Generic drug name: Irbesartan + amlodipine Date: Study Code: 31 active centers: 20 in Korea, 7 in India and 4 in Philippines.
 These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription Sponsor/company: sanofi-aventis ClinicalTrials.gov
These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription Sponsor/company: sanofi-aventis ClinicalTrials.gov
Irritable Bowel Syndrome
 What I need to know about Irritable Bowel Syndrome NATIONAL INSTITUTES OF HEALTH National Digestive Diseases Information Clearinghouse U.S. Department of Health and Human Services What I need to know
What I need to know about Irritable Bowel Syndrome NATIONAL INSTITUTES OF HEALTH National Digestive Diseases Information Clearinghouse U.S. Department of Health and Human Services What I need to know
Inlyta (axitinib) for Kidney Cancer
 Inlyta (axitinib) for Kidney Cancer Inlyta is a medication used to treat advanced kidney cancer in adults when one prior drug treatment for this disease has not worked Dosage: 5mg taken twice a day How
Inlyta (axitinib) for Kidney Cancer Inlyta is a medication used to treat advanced kidney cancer in adults when one prior drug treatment for this disease has not worked Dosage: 5mg taken twice a day How
PFIZER INC. THERAPEUTIC AREA AND FDA APPROVED INDICATIONS: See USPI.
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
EVALUATION OF PROTOZOAN PARASITES CAUSING DIARRHOEA IN HIV POSITIVE PATIENTS
 EVALUATION OF PROTOZOAN PARASITES CAUSING DIARRHOEA IN HIV POSITIVE PATIENTS Uday Shankar 1, Saral J. Ghosh 2 1Post Graduate Student, Department of Microbiology, D. Y. Patil Medical College, Kolhapur,
EVALUATION OF PROTOZOAN PARASITES CAUSING DIARRHOEA IN HIV POSITIVE PATIENTS Uday Shankar 1, Saral J. Ghosh 2 1Post Graduate Student, Department of Microbiology, D. Y. Patil Medical College, Kolhapur,
Study No.: Title: Rationale: Phase: Study Period: Study Design: Centers: Indication: Treatment: Objective: Primary Outcome/Efficacy Variable:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Clinical Trial Results Database Page 1
 Clinical Trial Results Database Page 1 Sponsor Novartis Pharmaceuticals Corporation Generic Drug Name Therapeutic Area of Trial Major Depressive Disorder (MDD) Approved Indication Treatment of major depressive
Clinical Trial Results Database Page 1 Sponsor Novartis Pharmaceuticals Corporation Generic Drug Name Therapeutic Area of Trial Major Depressive Disorder (MDD) Approved Indication Treatment of major depressive
SYNOPSIS. The study results and synopsis are supplied for informational purposes only.
 SYNOPSIS INN : FEXOFENADINE Study number : PJPR0024 Study title : A double-blind, randomized, placebo-controlled, parallel study comparing the efficacy and safety of three dosage strengths of MDL 16,455A
SYNOPSIS INN : FEXOFENADINE Study number : PJPR0024 Study title : A double-blind, randomized, placebo-controlled, parallel study comparing the efficacy and safety of three dosage strengths of MDL 16,455A
Study No.: Title: Rationale: Phase: Study Period: Study Design: Centers: Indication: Treatment: Objectives: Primary Outcome/Efficacy Variable:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
PFIZER INC. THERAPEUTIC AREA AND FDA APPROVED INDICATIONS: See USPI
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert.
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. PROPRIETARY DRUG NAME / GENERIC DRUG NAME: Lyrica / Pregabalin
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. PROPRIETARY DRUG NAME / GENERIC DRUG NAME: Lyrica / Pregabalin
SYNOPSIS. The study results and synopsis are supplied for informational purposes only.
 SYNOPSIS INN : LEFLUNOMIDE Study number : HMR486/1037 et HMR486/3503 Study title : Population pharmacokinetics of A77 1726 (M1) after oral administration of leflunomide in pediatric subjects with polyarticular
SYNOPSIS INN : LEFLUNOMIDE Study number : HMR486/1037 et HMR486/3503 Study title : Population pharmacokinetics of A77 1726 (M1) after oral administration of leflunomide in pediatric subjects with polyarticular
Indian Journal of Basic and Applied Medical Research; September 2014: Vol.-3, Issue- 4, P
 Original article: Comparison of stool concentration methods for detection of prevalence of enteroparasitic infection in rural tertiary care teaching hospital of Maharashtra 1 Dr. Jaishree Puri, 2 Dr. S.
Original article: Comparison of stool concentration methods for detection of prevalence of enteroparasitic infection in rural tertiary care teaching hospital of Maharashtra 1 Dr. Jaishree Puri, 2 Dr. S.
Sponsor / Company: Sanofi Drug substance(s): SSR103371
 These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor / Company: Sanofi Drug substance(s):
These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor / Company: Sanofi Drug substance(s):
PFIZER INC. PROTOCOL TITLE: Efficacy and Safety of the Authentic Recombinant Human Somatropin Genotropin in Children with Familial Short Stature
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
A randomised, double-blind, parallel group, multicentre study to compare the tolerability, safety, and efficacy of oxycodone with morphine in
 A randomised, double-blind, parallel group, multicentre study to compare the tolerability, safety, and efficacy of oxycodone with morphine in patients using i.v. patient-controlled analgesia (PCA) for
A randomised, double-blind, parallel group, multicentre study to compare the tolerability, safety, and efficacy of oxycodone with morphine in patients using i.v. patient-controlled analgesia (PCA) for
Is one of the most common chronic disorders. causing patients to seek medical treatment.
 ILOs After this lecture you should be able to : Define IBS Identify causes and risk factors of IBS Determine the appropriate therapeutic options for IBS Is one of the most common chronic disorders causing
ILOs After this lecture you should be able to : Define IBS Identify causes and risk factors of IBS Determine the appropriate therapeutic options for IBS Is one of the most common chronic disorders causing
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert.
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. PROPRIETARY DRUG NAME /GENERIC DRUG NAME: Vfend /Voriconazole
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. PROPRIETARY DRUG NAME /GENERIC DRUG NAME: Vfend /Voriconazole
Individual Study Table Referring to Part of the Dossier. Volume: Page:
 1 SYNOPSIS (CR002878) Title of Study: The effect of on vasomotor symptoms in healthy postmenopausal women: a double-blind placebo controlled pilot study Investigators: Multiple, see Section 4, Investigators
1 SYNOPSIS (CR002878) Title of Study: The effect of on vasomotor symptoms in healthy postmenopausal women: a double-blind placebo controlled pilot study Investigators: Multiple, see Section 4, Investigators
PFIZER INC. PROTOCOL TITLE: Growth hormone therapy in short children after renal transplantation for chronic renal failure in Germany.
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
Study No.: Title: Rationale: Phase: Study Period: Study Design: Centres: Indication: Treatment: Objectives: Primary Outcome/Efficacy Variable:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Clinical Study Synopsis
 Clinical Study Synopsis This document is not intended to replace the advice of a healthcare professional and should not be considered as a recommendation. Patients should always seek medical advice before
Clinical Study Synopsis This document is not intended to replace the advice of a healthcare professional and should not be considered as a recommendation. Patients should always seek medical advice before
Summary of Results for Laypersons
 What was the Study Called? Summary of Results for Laypersons A Phase IIIb/IV Randomized, Controlled, Open-Label, Parallel Group Study to Compare the Efficacy of Vancomycin Therapy to Extended Duration
What was the Study Called? Summary of Results for Laypersons A Phase IIIb/IV Randomized, Controlled, Open-Label, Parallel Group Study to Compare the Efficacy of Vancomycin Therapy to Extended Duration
PFIZER INC. THERAPEUTIC AREA AND FDA APPROVED INDICATIONS: See United States Package Insert (USPI)
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
Zinplava. (bezlotoxumab) New Product Slideshow
 Zinplava (bezlotoxumab) New Product Slideshow Introduction Brand name: Zinplava Generic name: Bezlotoxumab Pharmacological class: Human IgG1 monoclonal antibody Strength and Formulation: 25mg/mL; solution
Zinplava (bezlotoxumab) New Product Slideshow Introduction Brand name: Zinplava Generic name: Bezlotoxumab Pharmacological class: Human IgG1 monoclonal antibody Strength and Formulation: 25mg/mL; solution
Is Physical Activity Effective In Reducing The Gastrointestinal Symptoms Associated with Irritable Bowel Syndrome?
 Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 2018 Is Physical Activity Effective In Reducing
Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 2018 Is Physical Activity Effective In Reducing
Study No.:MPX Title: Rationale: Phase: IIB Study Period: Study Design: Centres: Indication: Treatment: Objectives:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only.
 The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
PFIZER INC. PROTOCOL TITLE: Metabolic effects of growth hormone (Genotonorm ) in girls with Turner syndrome.
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
SYNOPSIS 2/198 CSR_BDY-EFC5825-EN-E02. Name of company: TABULAR FORMAT (For National Authority Use only)
 SYNOPSIS Title of the study: A randomized, double-blind, placebo-controlled, parallel-group, fixed-dose (rimonabant 20 mg) multicenter study of long-term glycemic control with rimonabant in treatment-naïve
SYNOPSIS Title of the study: A randomized, double-blind, placebo-controlled, parallel-group, fixed-dose (rimonabant 20 mg) multicenter study of long-term glycemic control with rimonabant in treatment-naïve
COMPLETE DIGESTIVE STOOL ANALYSIS - Level 5
 COMPLETE DIGESTIVE STOOL ANALYSIS - Level 5 MACROSCOPIC DESCRIPTION Stool Colour Brown Result Range Brown Markers Colour - Brown is the colour of normal stool. Other colours may indicate abnormal GIT conditions.
COMPLETE DIGESTIVE STOOL ANALYSIS - Level 5 MACROSCOPIC DESCRIPTION Stool Colour Brown Result Range Brown Markers Colour - Brown is the colour of normal stool. Other colours may indicate abnormal GIT conditions.
Sponsor / Company: Sanofi Drug substance(s): Insulin Glargine (HOE901) Insulin Glulisine (HMR1964)
 These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor / Company: Sanofi Drug substance(s):
These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor / Company: Sanofi Drug substance(s):
8/29/2016 DIVERTICULAR DISEASE: WHAT EVERY NURSE PRACTITIONER SHOULD KNOW. LENORE LAMANNA Ed.D, ANP-C LEARNING OBJECTIVES
 DIVERTICULAR DISEASE: WHAT EVERY NURSE PRACTITIONER SHOULD KNOW LENORE LAMANNA Ed.D, ANP-C LEARNING OBJECTIVES Define Diverticular Disease Discuss Epidemiology and Pathophysiology of Diverticular disease
DIVERTICULAR DISEASE: WHAT EVERY NURSE PRACTITIONER SHOULD KNOW LENORE LAMANNA Ed.D, ANP-C LEARNING OBJECTIVES Define Diverticular Disease Discuss Epidemiology and Pathophysiology of Diverticular disease
THERAPEUTIC AREA AND FDA APPROVED INDICATIONS: See USPI.
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
Product: Femmerol. Client: Solutions for Women, LLC 315 North Village Avenue New York, (516)
 CLINICAL TRIAL In 2002 the company completed an IRB (Institutional Review Board) Prospective, Randomized, Placebo-Controlled Clinical Trial. In Conclusion: Femmerol is effective in relieving a number of
CLINICAL TRIAL In 2002 the company completed an IRB (Institutional Review Board) Prospective, Randomized, Placebo-Controlled Clinical Trial. In Conclusion: Femmerol is effective in relieving a number of
Prevalence of Intestinal Parasitic Infections in HIV-Positive Patients
 ISSN: 2319-7706 Volume 4 Number 5 (2015) pp. 269-273 http://www.ijcmas.com Original Research Article Prevalence of Intestinal Parasitic Infections in HIV-Positive Patients Vasundhara*, Haris M.Khan, Harekrishna
ISSN: 2319-7706 Volume 4 Number 5 (2015) pp. 269-273 http://www.ijcmas.com Original Research Article Prevalence of Intestinal Parasitic Infections in HIV-Positive Patients Vasundhara*, Haris M.Khan, Harekrishna
PFIZER INC. THERAPEUTIC AREA AND FDA APPROVED INDICATIONS: This drug is not marketed in the United States.
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
-2002: Rectal blood loss, UC? (no definite diagnosis) rectal mesalazine. -June 2008: Recurrence of rectal blood loss and urgency
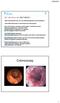 SD, male 40 yrs. old. (680718M467.) -2002: Rectal blood loss, UC? (no definite diagnosis) rectal mesalazine -June 2008: Recurrence of rectal blood loss and urgency Total colonoscopy: ulcerative rectitis,
SD, male 40 yrs. old. (680718M467.) -2002: Rectal blood loss, UC? (no definite diagnosis) rectal mesalazine -June 2008: Recurrence of rectal blood loss and urgency Total colonoscopy: ulcerative rectitis,
CENTENE PHARMACY AND THERAPEUTICS DRUG REVIEW 2Q17 April May
 BRAND NAME Trulance GENERIC NAME Plecanatide MANUFACTURER Synergy Pharmaceuticals, Inc. DATE OF APPROVAL January 19, 2017 PRODUCT LAUNCH DATE Anticipated in 1Q2017 REVIEW TYPE Review type 1 (RT1): New
BRAND NAME Trulance GENERIC NAME Plecanatide MANUFACTURER Synergy Pharmaceuticals, Inc. DATE OF APPROVAL January 19, 2017 PRODUCT LAUNCH DATE Anticipated in 1Q2017 REVIEW TYPE Review type 1 (RT1): New
These results are supplied for informational purposes only.
 These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription Sponsor/company: sanofi-aventis ClinialTrials.gov
These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription Sponsor/company: sanofi-aventis ClinialTrials.gov
[Conditions for approval] The applicant is required to develop and appropriately implement a risk management plan.
![[Conditions for approval] The applicant is required to develop and appropriately implement a risk management plan. [Conditions for approval] The applicant is required to develop and appropriately implement a risk management plan.](/thumbs/83/87676291.jpg) Report on the Deliberation Results May 14, 2015 Evaluation and Licensing Division, Pharmaceutical and Food Safety Bureau, Ministry of Health, Labour and Welfare [Brand name] Irribow Tablets 2.5 μg, Irribow
Report on the Deliberation Results May 14, 2015 Evaluation and Licensing Division, Pharmaceutical and Food Safety Bureau, Ministry of Health, Labour and Welfare [Brand name] Irribow Tablets 2.5 μg, Irribow
PACKAGE LEAFLET: Information for the patient. METRONIDAZOL Tablets 250 mg Solution for infusion 0.5 % Ovules mg (Metronidazole)
 PACKAGE LEAFLET: Information for the patient METRONIDAZOL Tablets 250 mg Solution for infusion 0.5 % Ovules - 500 mg (Metronidazole) Read this leaflet carefully before you start taking this medicine. Keep
PACKAGE LEAFLET: Information for the patient METRONIDAZOL Tablets 250 mg Solution for infusion 0.5 % Ovules - 500 mg (Metronidazole) Read this leaflet carefully before you start taking this medicine. Keep
What is Irritable Bowel Syndrome (IBS)?
 What is Irritable Bowel Syndrome (IBS)? Irritable bowel syndrome (IBS) is a health issue found in your intestines (gut). IBS can cause symptoms such as: Belly pain. Cramping. Gas. Bloating (or swelling)
What is Irritable Bowel Syndrome (IBS)? Irritable bowel syndrome (IBS) is a health issue found in your intestines (gut). IBS can cause symptoms such as: Belly pain. Cramping. Gas. Bloating (or swelling)
Supplemental Table 1: Moderate and severe definitions of Celiac Disease Symptom Diary
 Supplemental Table 1: Moderate and severe definitions of Celiac Disease Symptom Diary symptoms CDSD Symptom Diarrhea Constipation Abdominal Pain Bloating Nausea Tiredness Moderate Once or twice between
Supplemental Table 1: Moderate and severe definitions of Celiac Disease Symptom Diary symptoms CDSD Symptom Diarrhea Constipation Abdominal Pain Bloating Nausea Tiredness Moderate Once or twice between
Parasite Organism Chart Parasite Description Habitat/Sources of Isolation Blastocystis hominis
 Blastocystis hominis B. hominis has recently been reclassified as a protozoan, of which there are thought to be four separate serologic groups. 1 This organism is transmitted via the fecal-oral route or
Blastocystis hominis B. hominis has recently been reclassified as a protozoan, of which there are thought to be four separate serologic groups. 1 This organism is transmitted via the fecal-oral route or
PARASITOLOGY CASE HISTORY 15 (HISTOLOGY) (Lynne S. Garcia)
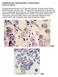 PARASITOLOGY CASE HISTORY 15 (HISTOLOGY) (Lynne S. Garcia) A biopsy was performed on a 27-year-old man with no known travel history, presenting with a perianal ulcer. The specimen was preserved in formalin
PARASITOLOGY CASE HISTORY 15 (HISTOLOGY) (Lynne S. Garcia) A biopsy was performed on a 27-year-old man with no known travel history, presenting with a perianal ulcer. The specimen was preserved in formalin
William D Chey, 1 Anthony J Lembo, 2 James A Phillips, 3 David P Rosenbaum 4
 Efficacy and safety of tenapanor in patients with constipationpredominant irritable bowel syndrome: a 12-week, double-blind, placebocontrolled, randomized phase 2b trial William D Chey, 1 Anthony J Lembo,
Efficacy and safety of tenapanor in patients with constipationpredominant irritable bowel syndrome: a 12-week, double-blind, placebocontrolled, randomized phase 2b trial William D Chey, 1 Anthony J Lembo,
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only.
 The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
Clinical Policy: Alosetron (Lotronex) Reference Number: CP.CPA.65 Effective Date: Last Review Date: Line of Business: Medicaid Medi-Cal
 Clinical Policy: (Lotronex) Reference Number: CP.CPA.65 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Medicaid Medi-Cal Revision Log See Important Reminder at the end of this policy
Clinical Policy: (Lotronex) Reference Number: CP.CPA.65 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Medicaid Medi-Cal Revision Log See Important Reminder at the end of this policy
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION. 20 October 2010
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 20 October 2010 MEZAVANT LP 1200 mg, prolonged-release gastro-resistant tablets B/60 (CIP code: 378 689-2) Applicant
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 20 October 2010 MEZAVANT LP 1200 mg, prolonged-release gastro-resistant tablets B/60 (CIP code: 378 689-2) Applicant
NUR 568: Critical Appraisal Topic Lactobacillus in the Management of Abdominal Pain in Children Christina A. Dahl, RN, FNP-s University of Mary
 Running Head: NUR 568 Critical Appraisal Topic 1 NUR 568: Critical Appraisal Topic Lactobacillus in the Management of Abdominal Pain in Children Christina A. Dahl, RN, FNP-s University of Mary Running
Running Head: NUR 568 Critical Appraisal Topic 1 NUR 568: Critical Appraisal Topic Lactobacillus in the Management of Abdominal Pain in Children Christina A. Dahl, RN, FNP-s University of Mary Running
ClinicalTrials.gov Identifier: sanofi-aventis. Sponsor/company:
 These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription Sponsor/company: sanofi-aventis ClinicalTrials.gov
These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription Sponsor/company: sanofi-aventis ClinicalTrials.gov
WHAT EVERYONE SHOULD KNOW ABOUT GASTROINTESTINAL DISORDERS IN WOMEN. By Robyn G. Karlstadt, M.D., FACG
 WHAT EVERYONE SHOULD KNOW ABOUT GASTROINTESTINAL DISORDERS IN WOMEN By Robyn G. Karlstadt, M.D., FACG Originally published in October 2002. Updated by Dr. Robyn G. Karlstadt, M.D., MACG in April 2007.
WHAT EVERYONE SHOULD KNOW ABOUT GASTROINTESTINAL DISORDERS IN WOMEN By Robyn G. Karlstadt, M.D., FACG Originally published in October 2002. Updated by Dr. Robyn G. Karlstadt, M.D., MACG in April 2007.
At the outset, we want to clear up some terminology issues. IBS is COPYRIGHTED MATERIAL. What Is IBS?
 1 What Is IBS? At the outset, we want to clear up some terminology issues. IBS is the abbreviation that doctors use for irritable bowel syndrome, often when they are talking about people with IBS. We will
1 What Is IBS? At the outset, we want to clear up some terminology issues. IBS is the abbreviation that doctors use for irritable bowel syndrome, often when they are talking about people with IBS. We will
Chapter 19. Assisting With Bowel Elimination. Elsevier items and derived items 2014, 2010 by Mosby, an imprint of Elsevier Inc. All rights reserved.
 Chapter 19 Assisting With Bowel Elimination Normal Bowel Elimination Time and frequency of bowel movements (BMs) vary. To assist with bowel elimination, you need to know these terms: Defecation is the
Chapter 19 Assisting With Bowel Elimination Normal Bowel Elimination Time and frequency of bowel movements (BMs) vary. To assist with bowel elimination, you need to know these terms: Defecation is the
CHAPTER 4: DISEASES SPREAD BY FOOD AND WATER
 CHAPTER 4: DISEASES SPREAD BY FOOD AND WATER Highlights The incidence of diseases spread by food and water was generally higher in Peel than Ontario with the exceptions of hepatitis A and verotoxinproducing
CHAPTER 4: DISEASES SPREAD BY FOOD AND WATER Highlights The incidence of diseases spread by food and water was generally higher in Peel than Ontario with the exceptions of hepatitis A and verotoxinproducing
BRL /RSD-101RLL/1/CPMS-716. Report Synopsis
 Report Synopsis Study Title: A Multicenter, Open-label, Six-Month Extension Study to Assess the Long-term Safety of Paroxetine in Children and Adolescents with Major Depressive Disorder (MDD) or Obsessive-Compulsive
Report Synopsis Study Title: A Multicenter, Open-label, Six-Month Extension Study to Assess the Long-term Safety of Paroxetine in Children and Adolescents with Major Depressive Disorder (MDD) or Obsessive-Compulsive
Recent Diagnostic Methods for Intestinal Parasitic Infections
 Recent Diagnostic Methods for Intestinal Parasitic Infections By Dr. Doaa Abdel Badie Salem Lecturer of Medical Parasitology, Mansoura Faculty of Medicine Agenda Intestinal parasites. Traditional Diagnostic
Recent Diagnostic Methods for Intestinal Parasitic Infections By Dr. Doaa Abdel Badie Salem Lecturer of Medical Parasitology, Mansoura Faculty of Medicine Agenda Intestinal parasites. Traditional Diagnostic
PARASITE MRS. OHOUD S.ALHUMAIDAN
 PARASITE MRS. OHOUD S.ALHUMAIDAN OUTLINE Intruduction Important terms classification of hosts Mode of parasitic infections General Classification of parasites Specific Classification of parasites Protozoa
PARASITE MRS. OHOUD S.ALHUMAIDAN OUTLINE Intruduction Important terms classification of hosts Mode of parasitic infections General Classification of parasites Specific Classification of parasites Protozoa
(sunitinib malate) for Kidney Cancer
 Sutent (sunitinib malate) for Kidney Cancer Sutent is a medication used to treat adult patients with kidney cancer that has been surgically removed and at high risk of recurrence, or advanced kidney cancer
Sutent (sunitinib malate) for Kidney Cancer Sutent is a medication used to treat adult patients with kidney cancer that has been surgically removed and at high risk of recurrence, or advanced kidney cancer
Xifaxan. Xifaxan (rifaximin) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.01.34 Subject: Xifaxan Page: 1 of 6 Last Review Date: December 8, 2017 Xifaxan Description Xifaxan (rifaximin)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.01.34 Subject: Xifaxan Page: 1 of 6 Last Review Date: December 8, 2017 Xifaxan Description Xifaxan (rifaximin)
Ovarian cyst, indigestion, constipation, back pain bloating
 P ford residence southampton, ny Ovarian cyst, indigestion, constipation, back pain bloating Mar 15, 2017. 33-year-old Fiona thought her bloating and stomach cramps were normal digestion problems. She
P ford residence southampton, ny Ovarian cyst, indigestion, constipation, back pain bloating Mar 15, 2017. 33-year-old Fiona thought her bloating and stomach cramps were normal digestion problems. She
