Neuropathic pain: aetiology, s y m p t o m s, m e c h a n i s m s, a n d m a n a g e m e n t
|
|
|
- Virginia Wheeler
- 6 years ago
- Views:
Transcription
1 Pain Neuropathic pain: aetiology, s y m p t o m s, m e c h a n i s m s, a n d m a n a g e m e n t Clifford J Woolf, Richard J Mannion We highlight current theories about peripheral neuropathic pain and show that progress in management is contingent on targeting treatment not at the aetiological factors or the symptoms but at the mechanisms that operate to produce the symptoms. This approach will require substantial progress in our understanding of the pathophysiology of neuropathic pain, the development of accurate diagnostic tools to discover what mechanisms contribute to the pain syndrome in an individual, and effective treatments aimed specifically at the mechanisms. Neuropathic pain is a pathological pain The capacity to experience pain has a protective role: it warns us of imminent or actual tissue damage and elicits coordinated reflex and behavioural responses to keep such damage to a minimum. If tissue damage is unavoidable, a set of excitability changes in the peripheral and central nervous system establish a profound but reversible pain hypersensitivity in the inflamed and surrounding tissue. This process assists wound repair because any contact w i t h the damaged part is avoided until healing has occurred. By contrast, persistent pain syndromes offer no biological advantage and cause suffering and distress. Such maladaptive pain typically results from damage to the nervous system the peripheral nerve, the dorsal root ganglion or dorsal root, or the central nervous system and is known as neuropathic pain. Such syndromes comprise a complex combination of negative symptoms or sensory deficits, such as partial or complete loss of sensation, and positive symptoms that include dysaethesia, paraesthesia, and pain. Apart from trigeminal neuralgia, which responds well to c a r b a m a z e p i n e, 1 pharmacotherapy for neuropathic pain has been disappointing. Patients with neuropathic pain do not respond to non-steroidal anti-inflammatory drugs and resistance or insensitivity to opiates is common. Patients are usually treated empirically with tricyclic or serotonin and norepinephrine uptake inhibitors, antidepressants, and anticonvulsants that all have limited efficacy and undesirable s i d e - e f f e c t s. 2 Neurosurgical lesions have a negligible role and functional neurosurgery, including dorsal column or brain stimulation, is controversial, although transcutaneous nerve stimulation may provide some relief. Local anaesthetic blocks targeted at trigger points, peripheral nerves, plexi, dorsal roots, and the sympathetic nervous system have useful but short-lived effects; longer lasting blocks by phenol injection or cryotherapy risk irreversible functional impairment and have not been tested in placebo-controlled trials. Chronic epidural administration of drugs such as clonidine, steroids, opioids, or midazolam is invasive, has side-effects, and the efficacy of these drugs has not been adequately assessed. There is no treatment to prevent the development of neuropathic pain, 3 nor to adequately, predictably, and Lancet 1999; 353: Neural Plasticity Research Group, Department of Anesthesia and Critical Care, Massachusetts General Hospital and Harvard Medical School, Charlestown, MA 02129, USA (Prof C J Woolf MRCP, R J Mannion PhD) Correspondence to: Prof Clifford J Woolf Figure 1: Aetiology, mechanisms, and symptoms specifically control established neuropathic pain. 2, 4 T h e a i m of treatment, therefore, is often just to help the patient cope by means of psychological or occupational therapy, rather than to eliminate the pain. Thus, there is an unmet clinical need and a challenge to develop more effective therapy can be achieved only if the relation between the aetiology, mechanisms, and symptoms of neuropathic pain are understood. Aetiology of neuropathic pain Neuropathic pain is currently classified on the basis of the aetiology of the insult to the nervous system or the anatomical distribution of the pain. Although this classification has some use for the differential diagnosis of the neuropathy, and for disease-modifying treatment if available, it offers no framework for clinical management of the pain. The relation between aetiology, mechanisms, and symptoms in this condition is complex (figure 1). The p a i n that manifests in diverse diseases may operate through common mechanisms. No pain mechanism is an inevitable consequence of a particular disease process; only a few patients are affected and there are no predictors to indicate which patient will develop neuropathic pain. One mechanism could be responsible for many different symptoms. Furthermore, the same symptom in two patients may be caused by different mechanisms. Finally, more than one mechanism can operate in a single patient, and these mechanisms may change with time. Thus, in patients with neuropathic pain it is impossible to predict THE LANCET Vol 353 June 5,
2 Figure 2: Stimulus-independent pain the mechanisms responsible for their pain on the basis of only the aetiology of the neuropathy or on the distribution and nature of the symptoms. Without identification of the mechanisms, the optimum treatment strategy for the patient s pain cannot be selected. A further confounding issue is that the extent to which common pain syndromes such as low back or myofascial pain have a neuropathic component is not known, because of the absence of specific diagnostic tools. This review only looks at peripheral neuropathic pain. The pain that results from spinal cord, brain stem, or thalamic/cortical damage is less common and may have different mechanisms. Why does nerve injury cause pain? Pain is normally elicited only when intense or damaging noxious stimuli activate high-threshold nociceptor primary sensory neurons. Peripheral neuropathic pain manifests a s spontaneous pain (stimulus-independent pain) or pain hypersensitivity elicited by a stimulus after damage to or alterations in sensory neurons (stimulus-evoked pain). Normal neuronal function is contingent on the neuron itself, its supporting glial cells, and the environment with which it interacts. For example, the chemicals that surround the peripheral terminals of nociceptors in the skin determine baseline sensitivity and activation threshold; inflammatory mediators and cytokines can sensitise nociceptor terminals such that they begin to respond to normally innocuous thermal and mechanical stimuli. Inflammation within the nerve or ganglion may also alter neurons by introducing new chemical messengers that alter the function, chemistry, or even survival of cells. Loss of cells or axons reduces sensibility and induces changes in the surrounding neurons that survive. Indeed, loss of sensory axons within the epidermis of patients with postherpetic neuralgia has been positively correlated with sensory deficits 5 and pain. 6 Constitutive availability of growth factors in normal s k i n is needed to maintain neuronal phenotype. Nerve growth factor is retrogradely transported from innervation targets to sensory neuron cell bodies where it regulates concentrations of neuropeptide transmitter. 7 T r a u m a t i c injury to axons or axonal transport blockade from the use of cytostatic drugs (eg, vincrisitine or taxol) will initiate profound phenotypic changes through the disruption of t h e constant communication that a cell body maintains with its target. The local axon environment is also a key determinant of sensory neuron phenotype. Schwann cells not only insulate axons, but also control sensory neuron f u n c t i o n, 8 for example, through sodium-channel expression and distribution along the axon. 9, 10 Schwann cell dedifferentiation initiated by nerve injury causes a switch away from the production of myelin to the synthesis o f growth factors, which have profound effects on neighbouring intact and injured neurons. Many examples of activity-dependent gene expression have been described within the nervous system, and insults that cause changes in activity or sensory input to the spinal cord are likely to have long-term effects on neuronal phenotype and function. Normal sensory function is the product of an actively maintained equilibrium between neurons and their environment. Any disruption of this equilibrium that results from changes in sensitivity, excitability, transmission, growth status, and survival c a n initiate profound changes in sensory function, which explains why diverse diseases can manifest as pain. A genetic component probably contributes to the diverse phenotype of individuals with apparently similar lesions, and would explain, for example, varied susceptibility to developing postherpetic neuralgia after an attack of shingles. Experimental models show enormous strain differences in pain behaviour and analgesic sensitivity, 11 and is an area ripe for further genomic research (eg, quantitative trait locus analysis). Ophoff and colleagues 12 discovered that a familial migraine syndrome was a channelopathy with a mutation in a calcium channel and similar changes may underlie some neuropathic pains. Symptoms of neuropathic pain Many patients with neuropathic pain exhibit persistent or paroxysmal pain that is independent of a stimulus. This stimulus-independent pain can be shooting, lancinating, or burning and may depend on activity in the sympathetic nervous system. Spontaneous activity in nociceptor C fibres is thought to be responsible for persistent burning pain and the sensitisation of dorsal horn neurons. Similarly, spontaneous activity in large myelinated A fibres (which normally signal innocuous sensations) is related to stimulus-independent paraesthesias and, after central sensitisation, to dysaesthesias and pain. Stimulus-evoked pain is a common component of peripheral nerve injury or damage and has two key features: hyperalgesia and allodynia. Hyperalgesia is an increased pain response to a suprathreshold noxious stimulus and is the result of abnormal processing of nociceptor input. Allodynia is the sensation of pain elicited by a non-noxious stimulus and can be produced in two ways: by the action of low threshold myelinated A fibres on an altered central nervous system; and by a reduction in the threshold of nociceptor terminals in the periphery. Because allodynia a s a clinical diagnosis does not implicate a particular mechanism, it may be more useful to subclassify it under the umbrella of hyperalgesia, so that the diagnosis of symptoms can be more suitably aligned with mechanisms. Stimulus-evoked hyperalgesias are commonly classified into subgroups on the basis of modality ie, mechanical, 1960 THE LANCET Vol 353 June 5, 1999
3 Figure 3: Disinhibition in which nerve injury reduces inhibition in dorsal horn through various mechanisms thermal, or chemical. Mechanical hyperalgesias are further classified as brush-evoked (dynamic), pressure-evoked (static), and punctate hyperalgesias. Abnormal sensitivity is not confined to the periphery. The neuroma, a swelling a t the proximal end of the injured nerve which contains regenerative axon sprouts, commonly exhibits exquisite mechanical sensitivity because of altered membrane properties of both C and A fibre axons. Mechanisms of peripheral neuropathic pain Stimulus-independent pain Two types of sodium channel are found in sensory neurons; the first type are sensitive to tetrodotoxin, a potent puffer-fish toxin, and the second type are insensitive to tetrodotoxin. The channels that are sensitive to tetrodotoxin are responsible for the initiation of the action potential and exist in all sensory neurons. By contrast, channels that are insensitive to tetrodotoxin are found o n l y on nociceptor sensory neurons, have much slower activation and inactivation kinetics than the tetrodotoxinsensitive channels, and are implicated in pathological pain s t a t e s. 13 After nerve injury, both types of sodium channels begin to accumulate in the axon at the neuroma site and along the length of the axon 14 and result in foci of hyperexcitability and ectopic action potential discharge in the axon and cell body of injured sensory neurons ( f i g u r e 2). Ochoa and colleagues 15 reported spontaneous firing of C fibre nociceptors and low threshold A f i b r e mechanoreceptors after nerve injury in human beings. After nerve insult, injured and uninjured sensory afferents may display ectopic discharge properties with the tetrodotoxin-insensitive sodium channels particularly implicated in the latter. 13 Unfortunately, the available sodium-channel blockers are not selective so they act on neural and cardiovascular sodium ion channels and produce central-nervous-system and cardiovascular-system side-effects. Nevertheless, such drugs are the mainstay of much treatment of neuropathic pain, including local Figure 4: Brush-evoked mechanical hyperplasia and central s e n s i t i s a t i o n Central sensitisation as a result of increased nociceptor drive or disinhibition after nerve injury leads to exaggerated dorsal horn response to A -fibre input and tactile hyperalgesia. anaesthetics, antiarrhythmic and antiepileptic agents, and tricyclic antidepressants. In a few patients stimulus-independent pain is sympathetically maintained. After partial nerve injury, injured and uninjured axons begin to express - a d r e n o- receptors, which renders them sensitive to circulating catecholamines and noradrenaline released from postganglionic sympathetic terminals (figure 2). Nerve injury also induces the sprouting of sympathetic axons into the dorsal root ganglion where they form baskets around the cell bodies of sensory neurons 16 and may constitute a mechanism in which sympathetic activity initiates activity in sensory fibres. Sympathetically maintained pain theoretically requires specific treatment, such as sympathetic blocks, guanethidine, or 1 antagonists that would not be appropriate for patients with sympathetically independent pain, although Kingery s meta-analysis 2 s h o w s poor outcome with sympatholytic approaches. Not all stimulus-independent pain is mediated by spontaneous activity in primary sensory neurons. Neurons in the dorsal horn of the spinal cord receive input from primary afferents. The firing of dorsal horn projection neurons, which process and transfer information about peripheral stimuli to the brain, is determined not only by the excitatory input they receive, but also by inhibitory inputs that can be segmental (restricted to the spinal cord) or descend from the brain. Thus, increased inhibition will reduce activity in dorsal horn neurons and act as a spinal gate. This process is the basis of transcutaneous electrical nerve stimulation, which activate segmental inhibitory pathways, or brain stimulators, which activate descending inhibitory pathways. Peripheral nerve injury may reduce the amount of inhibitory control ie, disinhibition over dorsal horn neurons through various mechanisms (figure 3). GABA, an inhibitory transmitter in the dorsal horn, is reduced, and GABA receptors and opioid receptors, which exist presynaptically on primary sensory neurons and postsynaptically on dorsal horn neurons, are downregulated. The expression of cholycystokinin, an endogenous inhibitor of opiate THE LANCET Vol 353 June 5,
4 Figure 5: Mechanical and thermal hyperplasia Innocuous stimuli cause pain if peripheral terminals are sensitised such that they have a much lower activation threshold. This sensitisation can result from antidromic activation, neurogenic inflammation, and increased sensory neuron exposure to molecules such as nerve growth factor (NGF). receptors, is upregulated in injured sensory neurons. Furthermore, interneurons in lamina II, many of which a r ei n h i b i t o r y, 17 are thought to die after peripheral nerve injury, possibly through an excitotoxic mechanism. 18 T h e disinhibition that occurs as a result of these processes increases the likelihood that a dorsal horn neuron will fire spontaneously or in an exaggerated way in response to primary afferent input, and would be sensitive to agents that increase inhibition and use-dependent sodiumchannel blockers. An attempt to mimic the action of GABA lead to the development of the antiepileptic gabapentin, which reduces neuropathic pain, 19 but does not act on GABA receptors or any currently known target. Nevertheless, augmenting central inhibition can be a useful therapeutic approach with opiates, GABA-enhancing drugs, or drugs such as clonidine that mimic descending i n h i b i t i o n. Stimulus-evoked pain Mechanical hyperalgesias are one of the most common manifestations of neuropathy. Brush-evoked (dynamic) hyperalgesia is the consequence of an increased central response to A fibre input. Continual input to the dorsal horn as a result of spontaneous firing in C fibre sensory neurons causes sensitisation of dorsal horn neurons, which increases their excitability such that they respond to normal inputs in an exaggerated and extended way. Thus, stimuli that would normally be innocuous are now painful. The main neurotransmitter in primary afferents is the excitatory aminoacid glutamate. Activation of nociceptors causes the release of glutamate from central terminals that acts on the ionotropic glutamate receptor amino-3-h y d r o x y methylisoxazole-4-proprionic acid postsynaptically to cause a rapid depolarisation in dorsal horn neurons and, if threshold is reached, action potential discharge. Another ionotropic glutamate receptor, N-methyl-D-aspartate (NMDA), is closed at resting membrane potentials, because a magnesium ion blocks the channel pore so that when glutamate binds to the receptor it does not activate the cell. However, depolarisation causes this channel to open. Neuropeptides such as substance P, colocalised in nociceptor central terminals with glutamate, are also released and act on neurokinin 1 receptors, extend depolarisation, and increase intracellular concentrations of calcium. This process activates protein kinase C which phosphorylates the NMDA receptor at serine and Figure 6: Sprouting of A fibres threonine residues, 20 removes the magnesium block, and thereby increases excitability. Phosphorylation of the NMDA receptor at tyrosine residues by tyrosine kinases also increases excitability by increasing the probability of an open channel. Thus, nociceptive input to the dorsal horn increases the excitability of neuronal membranes the phenomenon of central sensitisation which changes the way the neurons respond to subsequent inputs. 21 Central sensitisation can manifest in three ways: enlargement of the area in the periphery where a stimulus will activate neurons; increased response to a suprathreshold input; and previously subthreshold inputs reach threshold and initiate action potential discharge. These changes manifest as hypersensitivity to pain that spreads from the site of injury and includes tactile A - f i b r e - mediated hyperalgesia (figure 4). 22, 23 Spread of pain beyond the territory of an affected nerve is a common feature, once thought to indicate hysteria but now recognised as the result of central sensitisation. The role that central sensitisation and the NMDA receptor have in pathological pain is highlighted by studies that show blocking of central sensitisation with NMDA antagonists abolishes pain hypersensitivity in patients with neuropathic pain. 24 T h e r e is much evidence to support the role of central sensitisation in neuropathic pain. Stimulus-response latencies in psychophysical studies on brush-evoked hyperalgesia indicate activity along fast conducting myelinated axons. Electrical stimulation of A fibres elicits painful dysaesthesias in these patients, and selective A - f i b r e conduction block with ischaemia or pressure abolishes the allodynic features while leaving thermal and high threshold mechanical modalities intact. 25 Finally, disruption of constant activity in nociceptors by local anaesthetic 1962 THE LANCET Vol 353 June 5, 1999
5 Drug treatment of neuropathic pain and new agents under development Mechanism Symptom Target Drug Sodium-channel accumulation, Spontaneous pain, paraesthesia, Sodium channels sensitive to Sodium-channel blockers redistribution, altered neuroma sign tetrodotoxin Antiepileptic agents (carbamazipine expression Sodium channels resistant to lamotrigine) tetrodotoxin Antiarrhythmic agents (mexilitine tricyclic antidepressants) Blockers with greater analgesic than anticonvulsant index* Ion-channel selective blockers Central sensitisation Tactile (dynamic) hyperalgesia NMDA-R NMDA antagonists Cold hyperalgesia Neurokinin 1-R Ketamine, dextramethorphan, amantidine Pin-prick hyperalgesia Neuronal nitric oxide synthase Glycine site antagonists* Protein kinase Subunit specific antagonists Neurokinin-1-R antagonists* Neuronal nitric oxide synthase, protein kinase C inhibitors Peripheral sensitisation Pressure (static) hyperalgesia Vanilloid receptor-1-desensitisation Capsaicin Thermal hyperalgesia Neurokinin 1 Neurokinin-1-R antagonists* Spontaneous pain Sodium channels resistant to Blockers of sodium channels resistant to Neurogenic inflammation tetrodotoxin tetrodotoxin Nerve growth factor Nerve growth factors -receptor expression Spontaneous pain -receptor antagonists Phentolamine Sympathetic sprouting Nerve growth factor/trka Guanethidine Nerve growth factor antagonists Increased transmision Spontaneous pain N-type calcium channels Conotoxin Reduced inhibition Hyperalgesia Receptors Opiates *In clinical development. In preclinical development. (MOR, 2, GABA, neurokinin 1 adenosine, P2X 3, kainate, mglur, CCK, nachr) Gabapentin Clonidine Tricyclic antidepressants SNRIs blockers or sympathetic ganglion blockade can abolish mechanical allodynia without blocking activity in large myelinated fibres. This effect indicates that A-fibremediated mechanical allodynia as a result of central sensitisation in some patients requires some continual input to maintain the central hyperexcitability. 22, 23, 26 C o l d allodynia and mechanical hyperalgesia to punctate stimuli also are the result of central sensitisation, although in the later case, the symptom is not thought to rely on continual input to the dorsal horn. Jensen 27 suggested a similar mechanism for hyperpathia, in which sensory thresholds are increased yet a suprathreshold stimulus suddenly elicits an explosive response. Some patients exhibit a brush-evoked allodynia without any signs of persistent or spontaneous pain. Although low-level continual input may occur below the threshold of conscious detection, it is more likely that these patients have an A-fibre-mediated pain mechanism that does not require constant nociceptor drive. These mechanisms include: disinhibition as shown (figure 3), A fibre sprouting in the spinal cord (figure 5), and A fibre phenotypic switching after peripheral nerve injury. A fibre central terminals occur in all laminae of the dorsal horn, other than lamina II in the superficial dorsal horn which receives nociceptor C-fibre innervation exclusively (figure 5). Peripheral nerve injury, however, induces sprouting of A fibre central terminals into lamina II. 28 This sprouting i s probably dependent on injury to the peripheral axons o f C f i b r e s. 29 Thus, intrathecally supplied neurotrophic factors, which may act as a C fibre therapy, can prevent A fibre sprouting. 30 The functional importance of A fibre sprouting is that if lamina II, which normally receives only nociceptor information, begins to receive information about non-noxious stimuli, this information may be misinterpreted by the nervous system as noxious an anatomical substrate for mechanical allodynia. 31 T h i s rewiring of synaptic connections could explain why tactile allodynia occurs in patients with postherpetic neuralgia who have absent nociceptor innervation of the skin. 32 The nociceptors substance P and calcitonin-gene-related peptide are normally expressed by nociceptor primary afferent C fibres and A fibres, and are strongly implicated in sensory transmission between nociceptors, and the central nervous system, and the generation of central sensitisation. After peripheral nervy injury, the expression of these neuropeptides by these cells is downregulated. However, large myelinated A fibres begin to express these neuropeptides a phenotypic switch. 33 Thus, low threshold stimuli, activating A fibres, may cause release of substance P in the dorsal horn and thereby generate a state of central hyperexcitability that is normally produced only by nociceptor drive. The perception of pain in response to innocuous thermal stimuli in a small group of patients with spontaneously firing intact nociceptors has been reported and attributed to a reduction in the activation threshold of nociceptor peripheral terminals peripheral sensitisation. Action potentials can travel in both directions in sensory axons and ectopic discharge will result in invasion by antidromic action potentials of the peripheral terminal where they elicit the release of neuropeptides, such as substance P and calcitonin-gene-related peptide (figure 6). Release of these substances may lead to the sensitisation of peripheral sensory terminals of injured and uninjured fibres. A change in the function, chemistry, and structure of neurons (neural plasticity) clearly underlies the production of the altered sensitivity characteristic of neuropathic pain. These changes have been most extensively investigated in sensory and spinal cord neurons, but similar changes occur in the brain, particularly in the cortex and can be measured THE LANCET Vol 353 June 5,
6 experimentally and by functional magnetic resonance imaging or positron-emission tomography. Dramatic alterations in cortical spatial maps can be detected after nerve injury that may contribute to phantom pains. 3 4 Mechanisms as the target of management The relation between the mechanisms responsible for the symptoms of neuropathic pain, the molecular targets that underlie these mechanisms, current drug therapy of neuropathic pain, and the new agents under development are shown in the panel. Knowledge about the mechanisms that produce neuropathic pain has advanced through laboratory investigation and quantitative sensory testing of symptoms in patients, the latter shows whether the pain is mediated centrally or peripherally and which fibre types and modalities are involved. A symptom-based analysis of neuropathic pain is important for assessments of disease progression and treatment outcome. However, as a tool to define treatment strategy, symptoms alone are not useful because they are not equivalent to mechanisms. Sensitive and specific diagnostic tools are needed to reveal the particular pathological processes that are responsible for pain in an individual. Accurate diagnosis of mechanisms will be of use only if the mechanisms can be adequately targeted with specific therapy. Sodium channels that are insensitive to tetrodotoxin, for example, offer a new target for treatment because their sensory neuron specificity would avoid the side-effects of other membrane stabilisers, such as tricyclic antidepressants, sodiumchannel blockers, and anticonvulsants. Such new treatments will require carefully controlled clinical trials, the cohorts of which should be selected not by aetiology, which has no relation to treatment outcome, 2 b u t according to closely related symptoms, such as stimulusindependent pain or brush-evoked pain, to maximise the chance of discovering mechanism-specific treatments. Only when we have the tools to identify the mechanism responsible for the pain in a particular individual, and t h e n the capacity to reverse the mechanisms, will the management of neuropathic pain really advance. The onus on the clinician will then be to use the history, examination, investigation, and diagnostic tools as a way to identify the mechanisms that operate in their patients and use this information to select appropriate treatment. R e f e r e n c e s 1 Green MW, Selman JE. Review article: the medical management of trigeminal neuralgia. H e a d a c h e 1991; 3 1 : Kingery WS. A critical review of controlled clinical trials for peripheral neuropathic pain and complex regional pain syndromes. P a i n 1997; 7 3 : Nikolajsen L, Ilkjaer S, Christensen JH, et al. Randomised trial of epidural bupivacaine and morphine in prevention of stump and phantom pain lower-limb amputation. L a n c e t 1997; : Koltzenburg M. Painful neuropathies. Curr Opinion Neurol 1998; ii: Rowbotham MC, Yosipovitch G, Connolly MK, et al. Cutaneous innervation density in the allodynic form of postherpetic neuralgia. Neurobiol Dis 1996; 3 : Oaklander AL, Romans K, Horasek S, Stocks A, Haver P, Meyer RA. Unilateral postherpetic neuralgia is associated with bilateral neuron damage. Ann Neurol 1998; 44: Lindsay RM, Harmar AJ. Nerve growth factor regulates expression of neuropeptide genes in adult sensory neurons. N a t u r e 1989; : Reynolds ML, Woolf C. Reciprocal Schwann cell-axon interactions. Curr Opin Neurobiol 1993; 3 : Vabnick I, Novakovic SD, Levinson SR, Schachner M, Shrager P. The clustering of axonal sodium channels during development of the peripheral nervous system. J N e u r o s c i 1996; 1 6 : Hinson AW, Gu XQ, Dib-Hajj S, Black JA, Waxman SG. Schwann cells modulate sodium channel expression in spinal sensory neurons in vitro. G l i a 1997; 2 1 : Devor M, Raber P. Heritability of symptoms in an experimental model of neuropathic pain. P a i n 1990; 4 2 : Ophoff RA, Terwindt GM, Vergouwe MN, et al. Familial hemiplegic migraine and episodic ataxia type-2 are caused by mutations in the Ca2+ channel gene CACNL1A4. C e l l 1996; 8 7 : Novakovic SD, Tzoumaka E, McGivern JG, et al. Distribution of the tetrodotoxin-resistant sodium channel PN3 in rat sensory neurons in normal and neuropathic conditions. J N e u r o s c i 1998; 1 8 : Devor M. The pathophysiology of damaged peripheral nerves. In: Wall PD, Melzack R, eds. Textbook of pain. Edinburgh: Churchill Livingstone, 1994: Ochoa J, Torebjork HE, Culp WJ, Schady W. Abnormal spontaneous activity in single sensory nerve fibres in humans. Muscle Nerve 1982; 5 : S 7 4 S McLachlan EM, Janig W, Devor M, Michaelis M. Peripheral nerve injury triggers noradrenergic sprouting within dorsal root ganglia. N a t u r e 1993; : Willis WD, Coggeshall RE. Sensory mechanisms of the spinal cord. New York: Plenum Press, Sugimoto T, Bennett GJ, Kajander KC. Transsynaptic degeneration in the superficial dorsal horn after sciatic nerve injury: effects of a chronic constriction injury, transection, and strychnine. P a i n 1990; 4 2 : Rosenberg JM, Harrell C, Ristic H, Werner RA, de Rosayro AM. The effect of gabapentin on neuropathic pain. Clin J P a i n 1997; 1 3 : Chen L, Huang LY. Protein kinase C reduces Mg2+ block of NMDAreceptor channels as a mechanism of modulation. N a t u r e 1992; : Woolf CJ. Evidence for a central component of post-injury pain hypersensitivity. N a t u r e 1983; : Treede RD, Davis KD, Campbell JN, Raja SN. The plasticity of cutaneous hyperalgesia during sympathetic ganglion blockade in patients with neuropathic pain. B r a i n 1992; : Gracely RH, Lynch SA, Bennett GJ. Painful neuropathy: altered central processing maintained dynamically by peripheral input. P a i n 1992; 5 1 : Nelson KA, Park KM, Robinovitz E, Tsigos C, Max MB. High-dose oral dextromethorphan versus placebo in painful diabetic neuropathy and postherpetic neuralgia. N e u r o l o g y 1997; 4 8 : Ochoa JL, Yarnitsky D. Mechanical hyperalgesias in neuropathic pain patients: dynamic and static subtypes. Ann Neurol 1993; 3 3 : Koltzenburg M, Torebjork HE, Wahren LK. Nociceptor modulated central sensitisation causes mechanical hyperalgesia in acute chemogenic and chronic neuropathic pain. Brain 1994; : Jensen TS. Mechanisms of neuropathic pain. Seattle: IASP Press, Woolf CJ, Shortland P, Coggeshall RE. Peripheral nerve injury t r i g g e r s central sprouting of myelinated afferents. N a t u r e ; : Mannion RJ, Doubell TP, Coggeshall RE, Woolf CJ. Collateral sprouting of uninjured primary afferent A-fibres into the superficial dorsal horn of the adult rat spinal cord after topical capsaicin treatment to the sciatic nerve. JN e u r o s c i 1996; 1 6 : Bennett GJ. Neuropathic pain. In: Wall PD, Melzack R, eds. Textbook of pain. Edinburgh: Churchill Livingstone, 1994; Woolf CJ, Doubell TP. The pathophysiology of chronic pain increased sensitivity to low threshold A beta-fibre inputs. Curr Opin Neurobiol 1994; 4 : Baron R, Saguer M. Postherpetic neuralgia. Are C-nociceptors involved in signalling and maintenance of tactile allodynia? B r a i n 1993; : Miki K, Fukuoka T, Tokunaga A, Noguchi K. Calcitonin gene-related peptide increase in the rat spinal horn and dorsal column nucleus following peripheral nerve injury: up-regulation in a subpopulation of primary afferent sensory neurons. N e u r o s c i e n c e 1998; 8 2 : Flor H, Elbert T, Knecht S, et al. Phantom-limb pain as a perceptual correlate of cortical reorganisation following arm amputation. N a t u r e 1995; : THE LANCET Vol 353 June 5, 1999
Seizure: the clinical manifestation of an abnormal and excessive excitation and synchronization of a population of cortical
 Are There Sharing Mechanisms of Epilepsy, Migraine and Neuropathic Pain? Chin-Wei Huang, MD, PhD Department of Neurology, NCKUH Basic mechanisms underlying seizures and epilepsy Seizure: the clinical manifestation
Are There Sharing Mechanisms of Epilepsy, Migraine and Neuropathic Pain? Chin-Wei Huang, MD, PhD Department of Neurology, NCKUH Basic mechanisms underlying seizures and epilepsy Seizure: the clinical manifestation
Pharmacology of Pain Transmission and Modulation
 Pharmacology of Pain Transmission and Modulation 2 Jürg Schliessbach and Konrad Maurer Nociceptive Nerve Fibers Pain is transmitted to the central nervous system via thinly myelinated Aδ and unmyelinated
Pharmacology of Pain Transmission and Modulation 2 Jürg Schliessbach and Konrad Maurer Nociceptive Nerve Fibers Pain is transmitted to the central nervous system via thinly myelinated Aδ and unmyelinated
Neuropathic Pain in Palliative Care
 Neuropathic Pain in Palliative Care Neuropathic Pain in Advanced Cancer Affects 40% of patients Multiple concurrent pains are common Often complex pathophysiology with mixed components Nocioceptive Neuropathic
Neuropathic Pain in Palliative Care Neuropathic Pain in Advanced Cancer Affects 40% of patients Multiple concurrent pains are common Often complex pathophysiology with mixed components Nocioceptive Neuropathic
Pain Pathways. Dr Sameer Gupta Consultant in Anaesthesia and Pain Management, NGH
 Pain Pathways Dr Sameer Gupta Consultant in Anaesthesia and Pain Management, NGH Objective To give you a simplistic and basic concepts of pain pathways to help understand the complex issue of pain Pain
Pain Pathways Dr Sameer Gupta Consultant in Anaesthesia and Pain Management, NGH Objective To give you a simplistic and basic concepts of pain pathways to help understand the complex issue of pain Pain
The anatomy and physiology of pain
 The anatomy and physiology of pain Charlotte E Steeds Abstract Pain is an unpleasant experience that results from both physical and psychological responses to injury. A complex set of pathways transmits
The anatomy and physiology of pain Charlotte E Steeds Abstract Pain is an unpleasant experience that results from both physical and psychological responses to injury. A complex set of pathways transmits
Pathophysiology of Pain
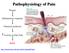 Pathophysiology of Pain Wound Inflammatory response Chemical mediators Activity in Pain Path PAIN http://neuroscience.uth.tmc.edu/s2/chapter08.html Chris Cohan, Ph.D. Dept. of Pathology/Anat Sci University
Pathophysiology of Pain Wound Inflammatory response Chemical mediators Activity in Pain Path PAIN http://neuroscience.uth.tmc.edu/s2/chapter08.html Chris Cohan, Ph.D. Dept. of Pathology/Anat Sci University
Introduction to some interesting research questions: Molecular biology of the primary afferent nociceptor
 Introduction to some interesting research questions: Molecular biology of the primary afferent nociceptor NOCICEPTORS ARE NOT IDENTICAL PEPTIDE SubP/CGRP Trk A NON-PEPTIDE IB4 P2X 3 c-ret Snider and McMahon
Introduction to some interesting research questions: Molecular biology of the primary afferent nociceptor NOCICEPTORS ARE NOT IDENTICAL PEPTIDE SubP/CGRP Trk A NON-PEPTIDE IB4 P2X 3 c-ret Snider and McMahon
Update on the Neurophysiology of Pain Transmission and Modulation: Focus on the NMDA-Receptor
 S2 Journal of Pain and Symptom Management Vol. 19 No. 1(Suppl.) January 2000 Proceedings Supplement NMDA-Receptor Antagonists: Evolving Role in Analgesia Update on the Neurophysiology of Pain Transmission
S2 Journal of Pain and Symptom Management Vol. 19 No. 1(Suppl.) January 2000 Proceedings Supplement NMDA-Receptor Antagonists: Evolving Role in Analgesia Update on the Neurophysiology of Pain Transmission
Pain Mechanisms. Prof Michael G Irwin MD, FRCA, FANZCA FHKAM Head Department of Anaesthesiology University of Hong Kong. The Somatosensory System
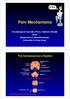 ain Mechanisms rof Michael G Irwin MD, FRCA, FANZCA FHKAM Head Department of Anaesthesiology University of Hong Kong The Somatosensory System Frontal cortex Descending pathway eriaqueductal gray matter
ain Mechanisms rof Michael G Irwin MD, FRCA, FANZCA FHKAM Head Department of Anaesthesiology University of Hong Kong The Somatosensory System Frontal cortex Descending pathway eriaqueductal gray matter
Cancer-induced bone pain
 Cancer-induced bone pain Common Prevalent in particular cancers: breast (73%), prostate (68%), thyroid (42%), lung (36%), renal (35%), colon (5%) Correlates with an increased morbidity Reduced performance
Cancer-induced bone pain Common Prevalent in particular cancers: breast (73%), prostate (68%), thyroid (42%), lung (36%), renal (35%), colon (5%) Correlates with an increased morbidity Reduced performance
Somatosensory Physiology (Pain And Temperature) Richard M. Costanzo, Ph.D.
 Somatosensory Physiology (Pain And Temperature) Richard M. Costanzo, Ph.D. OBJECTIVES After studying the material of this lecture the student should be familiar with: 1. The relationship between nociception
Somatosensory Physiology (Pain And Temperature) Richard M. Costanzo, Ph.D. OBJECTIVES After studying the material of this lecture the student should be familiar with: 1. The relationship between nociception
Chronic pain: We should not underestimate the contribution of neural plasticity. *Gwyn N Lewis 1, David A Rice 1,2
 Chronic pain: We should not underestimate the contribution of neural plasticity *Gwyn N Lewis 1, David A Rice 1,2 1 Health and Rehabilitation Research Institute, AUT University, Auckland, New Zealand 2
Chronic pain: We should not underestimate the contribution of neural plasticity *Gwyn N Lewis 1, David A Rice 1,2 1 Health and Rehabilitation Research Institute, AUT University, Auckland, New Zealand 2
San Francisco Chronicle, June 2001
 PAIN San Francisco Chronicle, June 2001 CONGENITAL INSENSITIVITY TO PAIN PAIN IS A SUBJECTIVE EXPERIENCE: It is not a stimulus MAJOR FEATURES OF THE PAIN EXPERIENCE: Sensory discriminative Affective (emotional)
PAIN San Francisco Chronicle, June 2001 CONGENITAL INSENSITIVITY TO PAIN PAIN IS A SUBJECTIVE EXPERIENCE: It is not a stimulus MAJOR FEATURES OF THE PAIN EXPERIENCE: Sensory discriminative Affective (emotional)
Dissecting out mechanisms responsible for peripheral neuropathic pain: Implications for diagnosis and therapy
 Life Sciences 74 (2004) 2605 2610 www.elsevier.com/locate/lifescie Dissecting out mechanisms responsible for peripheral neuropathic pain: Implications for diagnosis and therapy Clifford J. Woolf* Neural
Life Sciences 74 (2004) 2605 2610 www.elsevier.com/locate/lifescie Dissecting out mechanisms responsible for peripheral neuropathic pain: Implications for diagnosis and therapy Clifford J. Woolf* Neural
211MDS Pain theories
 211MDS Pain theories Definition In 1986, the International Association for the Study of Pain (IASP) defined pain as a sensory and emotional experience associated with real or potential injuries, or described
211MDS Pain theories Definition In 1986, the International Association for the Study of Pain (IASP) defined pain as a sensory and emotional experience associated with real or potential injuries, or described
Spinal Cord Injury Pain. Michael Massey, DO CentraCare Health St Cloud, MN 11/07/2018
 Spinal Cord Injury Pain Michael Massey, DO CentraCare Health St Cloud, MN 11/07/2018 Objectives At the conclusion of this session, participants should be able to: 1. Understand the difference between nociceptive
Spinal Cord Injury Pain Michael Massey, DO CentraCare Health St Cloud, MN 11/07/2018 Objectives At the conclusion of this session, participants should be able to: 1. Understand the difference between nociceptive
A role for uninjured afferents in neuropathic pain
 Acta Physiologica Sinica, October 25, 2008, 60 (5): 605-609 http://www.actaps.com.cn 605 Review A role for uninjured afferents in neuropathic pain Richard A. Meyer 1,2,3,*, Matthias Ringkamp 1 Departments
Acta Physiologica Sinica, October 25, 2008, 60 (5): 605-609 http://www.actaps.com.cn 605 Review A role for uninjured afferents in neuropathic pain Richard A. Meyer 1,2,3,*, Matthias Ringkamp 1 Departments
Pathophysiological Classification of Pain
 PATHOPHYSIOLOGY Overview Pathophysiological Classification of Pain Central sensitization/ dysfunctional pain Nociceptive pain - Somatic - Visceral Multiple pain mechanisms may coexist (mixed pain) Neuropathic
PATHOPHYSIOLOGY Overview Pathophysiological Classification of Pain Central sensitization/ dysfunctional pain Nociceptive pain - Somatic - Visceral Multiple pain mechanisms may coexist (mixed pain) Neuropathic
Pain teaching. Muhammad Laklouk
 Pain teaching Muhammad Laklouk Definition Pain An unpleasant sensory and emotional experience associated with actual or potential tissue damage or described in terms of such damage. Sensory (discriminatiory)
Pain teaching Muhammad Laklouk Definition Pain An unpleasant sensory and emotional experience associated with actual or potential tissue damage or described in terms of such damage. Sensory (discriminatiory)
Pathophysiology of Pain. Ramon Go MD Assistant Professor Anesthesiology and Pain medicine NYP-CUMC
 Pathophysiology of Pain Ramon Go MD Assistant Professor Anesthesiology and Pain medicine NYP-CUMC Learning Objectives Anatomic pathway of nociception Discuss the multiple target sites of pharmacological
Pathophysiology of Pain Ramon Go MD Assistant Professor Anesthesiology and Pain medicine NYP-CUMC Learning Objectives Anatomic pathway of nociception Discuss the multiple target sites of pharmacological
Brian Kahan, D.O. FAAPMR, DABPM, DAOCRM, FIPP Center for Pain Medicine and Physiatric Rehabilitation 2002 Medical Parkway Suite 150 Annapolis, MD
 Brian Kahan, D.O. FAAPMR, DABPM, DAOCRM, FIPP Center for Pain Medicine and Physiatric Rehabilitation 2002 Medical Parkway Suite 150 Annapolis, MD 1630 Main Street Suite 215 Chester, MD 410-571-9000 www.4-no-pain.com
Brian Kahan, D.O. FAAPMR, DABPM, DAOCRM, FIPP Center for Pain Medicine and Physiatric Rehabilitation 2002 Medical Parkway Suite 150 Annapolis, MD 1630 Main Street Suite 215 Chester, MD 410-571-9000 www.4-no-pain.com
Section: Chapter 5: Multiple Choice. 1. The structure of synapses is best viewed with a(n):
 Section: Chapter 5: Multiple Choice 1. The structure of synapses is best viewed with a(n): p.155 electron microscope. light microscope. confocal microscope. nissle-stained microscopic procedure. 2. Electron
Section: Chapter 5: Multiple Choice 1. The structure of synapses is best viewed with a(n): p.155 electron microscope. light microscope. confocal microscope. nissle-stained microscopic procedure. 2. Electron
PAIN MANAGEMENT in the CANINE PATIENT
 PAIN MANAGEMENT in the CANINE PATIENT Laurie Edge-Hughes, BScPT, MAnimSt (Animal Physio), CAFCI, CCRT Part 1: Laurie Edge-Hughes, BScPT, MAnimSt (Animal Physio), CAFCI, CCRT 1 Pain is the most common reason
PAIN MANAGEMENT in the CANINE PATIENT Laurie Edge-Hughes, BScPT, MAnimSt (Animal Physio), CAFCI, CCRT Part 1: Laurie Edge-Hughes, BScPT, MAnimSt (Animal Physio), CAFCI, CCRT 1 Pain is the most common reason
Proceedings of the World Small Animal Veterinary Association Sydney, Australia 2007
 Proceedings of the World Small Animal Sydney, Australia 2007 Hosted by: Next WSAVA Congress REDUCING THE PAIN FACTOR AN UPDATE ON PERI-OPERATIVE ANALGESIA Sandra Forysth, BVSc DipACVA Institute of Veterinary,
Proceedings of the World Small Animal Sydney, Australia 2007 Hosted by: Next WSAVA Congress REDUCING THE PAIN FACTOR AN UPDATE ON PERI-OPERATIVE ANALGESIA Sandra Forysth, BVSc DipACVA Institute of Veterinary,
Pain and impulse conduction
 1 Pain and impulse conduction L.H.D.J. Booij According to the World Health Organisation pain is defined as an unpleasant sensation that occurs from imminent tissue damage. From a physiological perspective,
1 Pain and impulse conduction L.H.D.J. Booij According to the World Health Organisation pain is defined as an unpleasant sensation that occurs from imminent tissue damage. From a physiological perspective,
SOMATOSENSORY SYSTEMS AND PAIN
 SOMATOSENSORY SYSTEMS AND PAIN A 21 year old man presented with a stab wound of the right side of the neck (Panel A). Neurological examination revealed right hemiplegia and complete right-sided loss of
SOMATOSENSORY SYSTEMS AND PAIN A 21 year old man presented with a stab wound of the right side of the neck (Panel A). Neurological examination revealed right hemiplegia and complete right-sided loss of
What it Takes to be a Pain
 What it Takes to be a Pain Pain Pathways and the Neurophysiology of pain Dennis S. Pacl, MD, FACP, FAChPM Austin Palliative Care/ Hospice Austin A Definition of Pain complex constellation of unpleasant
What it Takes to be a Pain Pain Pathways and the Neurophysiology of pain Dennis S. Pacl, MD, FACP, FAChPM Austin Palliative Care/ Hospice Austin A Definition of Pain complex constellation of unpleasant
CHAPTER 4 PAIN AND ITS MANAGEMENT
 CHAPTER 4 PAIN AND ITS MANAGEMENT Pain Definition: An unpleasant sensory and emotional experience associated with actual or potential tissue damage, or described in terms of such damage. Types of Pain
CHAPTER 4 PAIN AND ITS MANAGEMENT Pain Definition: An unpleasant sensory and emotional experience associated with actual or potential tissue damage, or described in terms of such damage. Types of Pain
NMDA-Receptor Antagonists and Opioid Receptor Interactions as Related to Analgesia and Tolerance
 Vol. 19 No. 1(Suppl.) January 2000 Journal of Pain and Symptom Management S7 Proceedings Supplement NDMA-Receptor Antagonists: Evolving Role in Analgesia NMDA-Receptor Antagonists and Opioid Receptor Interactions
Vol. 19 No. 1(Suppl.) January 2000 Journal of Pain and Symptom Management S7 Proceedings Supplement NDMA-Receptor Antagonists: Evolving Role in Analgesia NMDA-Receptor Antagonists and Opioid Receptor Interactions
Neurons Chapter 7 2/19/2016. Learning Objectives. Cells of the Nervous System. Cells of the Nervous System. Cells of the Nervous System
 Learning Objectives Neurons Chapter 7 Identify and describe the functions of the two main divisions of the nervous system. Differentiate between a neuron and neuroglial cells in terms of structure and
Learning Objectives Neurons Chapter 7 Identify and describe the functions of the two main divisions of the nervous system. Differentiate between a neuron and neuroglial cells in terms of structure and
Receptors and Neurotransmitters: It Sounds Greek to Me. Agenda. What We Know About Pain 9/7/2012
 Receptors and Neurotransmitters: It Sounds Greek to Me Cathy Carlson, PhD, RN Northern Illinois University Agenda We will be going through this lecture on basic pain physiology using analogies, mnemonics,
Receptors and Neurotransmitters: It Sounds Greek to Me Cathy Carlson, PhD, RN Northern Illinois University Agenda We will be going through this lecture on basic pain physiology using analogies, mnemonics,
Chapter 11 Introduction to the Nervous System and Nervous Tissue Chapter Outline
 Chapter 11 Introduction to the Nervous System and Nervous Tissue Chapter Outline Module 11.1 Overview of the Nervous System (Figures 11.1-11.3) A. The nervous system controls our perception and experience
Chapter 11 Introduction to the Nervous System and Nervous Tissue Chapter Outline Module 11.1 Overview of the Nervous System (Figures 11.1-11.3) A. The nervous system controls our perception and experience
Neuropathic pain, pain matrix dysfunction, and pain syndromes
 Neuropathic pain, pain matrix dysfunction, and pain syndromes MSTN121 - Neurophysiology Session 3 Department of Myotherapy Session objectives Describe the mechanism of nociceptive chronic pain. Define
Neuropathic pain, pain matrix dysfunction, and pain syndromes MSTN121 - Neurophysiology Session 3 Department of Myotherapy Session objectives Describe the mechanism of nociceptive chronic pain. Define
What is pain?: An unpleasant sensation. What is an unpleasant sensation?: Pain. - Aristotle.
 What is pain?: An unpleasant sensation. What is an unpleasant sensation?: Pain. - Aristotle. Nociception The detection of tissue damage or impending tissue damage, but There can be tissue damage without
What is pain?: An unpleasant sensation. What is an unpleasant sensation?: Pain. - Aristotle. Nociception The detection of tissue damage or impending tissue damage, but There can be tissue damage without
Chapter Nervous Systems
 The Nervous System Chapter Nervous Systems Which animals have nervous systems? (Which do not) What are the basic components of a NS? What kind of fish performs brain operations? What differentiates one
The Nervous System Chapter Nervous Systems Which animals have nervous systems? (Which do not) What are the basic components of a NS? What kind of fish performs brain operations? What differentiates one
What are the 6 types of neuroglia and their functions?!
 Warm Up! Take out your 11C Notes What are the 6 types of neuroglia and their functions?! Astrocytes Microglia Ependymal Cells Satellite Cells Schwann Cells Oligodendrocytes Support, brace, & nutrient transfer
Warm Up! Take out your 11C Notes What are the 6 types of neuroglia and their functions?! Astrocytes Microglia Ependymal Cells Satellite Cells Schwann Cells Oligodendrocytes Support, brace, & nutrient transfer
Lecture 22: A little Neurobiology
 BIO 5099: Molecular Biology for Computer Scientists (et al) Lecture 22: A little Neurobiology http://compbio.uchsc.edu/hunter/bio5099 Larry.Hunter@uchsc.edu Nervous system development Part of the ectoderm
BIO 5099: Molecular Biology for Computer Scientists (et al) Lecture 22: A little Neurobiology http://compbio.uchsc.edu/hunter/bio5099 Larry.Hunter@uchsc.edu Nervous system development Part of the ectoderm
The perception of pain is a complex process,
 SPECIAL TOPIC Pathophysiology of Pain: A Practical Primer Adeline Fong, MRCP, MRCA, FANZCA Stephan A. Schug, MD, FANZCA, FFPMANZCA Perth, Australia Summary: The experience of pain is a subjective one and
SPECIAL TOPIC Pathophysiology of Pain: A Practical Primer Adeline Fong, MRCP, MRCA, FANZCA Stephan A. Schug, MD, FANZCA, FFPMANZCA Perth, Australia Summary: The experience of pain is a subjective one and
Fundamentals of the Nervous System and Nervous Tissue: Part C
 PowerPoint Lecture Slides prepared by Janice Meeking, Mount Royal College C H A P T E R 11 Fundamentals of the Nervous System and Nervous Tissue: Part C Warm Up What is a neurotransmitter? What is the
PowerPoint Lecture Slides prepared by Janice Meeking, Mount Royal College C H A P T E R 11 Fundamentals of the Nervous System and Nervous Tissue: Part C Warm Up What is a neurotransmitter? What is the
PAIN IS A SUBJECTIVE EXPERIENCE: It is not a stimulus. MAJOR FEATURES OF THE PAIN EXPERIENCE: Sensory discriminative Affective (emotional) Cognitive
 PAIN PAIN IS A SUBJECTIVE EXPERIENCE: It is not a stimulus MAJOR FEATURES OF THE PAIN EXPERIENCE: Sensory discriminative Affective (emotional) Cognitive MEASUREMENT OF PAIN: A BIG PROBLEM Worst pain ever
PAIN PAIN IS A SUBJECTIVE EXPERIENCE: It is not a stimulus MAJOR FEATURES OF THE PAIN EXPERIENCE: Sensory discriminative Affective (emotional) Cognitive MEASUREMENT OF PAIN: A BIG PROBLEM Worst pain ever
Overview of Neuropathic pain
 Overview of Neuropathic pain Kongkiat Kulkantrakorn,M.D. Neurology division Thammasat University 1 Contents Overview of pain New concepts and mechanism Treatment options New data in management 2 3 Breaking
Overview of Neuropathic pain Kongkiat Kulkantrakorn,M.D. Neurology division Thammasat University 1 Contents Overview of pain New concepts and mechanism Treatment options New data in management 2 3 Breaking
Various Types of Pain Defined
 Various Types of Pain Defined Pain: The International Association for the Study of Pain describes pain as, An unpleasant sensory and emotional experience associated with actual or potential tissue damage,
Various Types of Pain Defined Pain: The International Association for the Study of Pain describes pain as, An unpleasant sensory and emotional experience associated with actual or potential tissue damage,
Action Potentials and Synaptic Transmission. BIO 219 Napa Valley College Dr. Adam Ross
 Action Potentials and Synaptic Transmission BIO 219 Napa Valley College Dr. Adam Ross Review of action potentials Nodes of Ranvier Nucleus Dendrites Cell body In saltatory conduction, the nerve impulses
Action Potentials and Synaptic Transmission BIO 219 Napa Valley College Dr. Adam Ross Review of action potentials Nodes of Ranvier Nucleus Dendrites Cell body In saltatory conduction, the nerve impulses
A Review of Neuropathic Pain: From Diagnostic Tests to Mechanisms
 DOI 10.1007/s40122-017-0085-2 REVIEW A Review of Neuropathic Pain: From Diagnostic Tests to Mechanisms Andrea Truini Received: September 19, 2017 Ó The Author(s) 2017. This article is an open access publication
DOI 10.1007/s40122-017-0085-2 REVIEW A Review of Neuropathic Pain: From Diagnostic Tests to Mechanisms Andrea Truini Received: September 19, 2017 Ó The Author(s) 2017. This article is an open access publication
Neurobiology of Pain Adjuvant analgesia
 Neurobiology of Pain Adjuvant analgesia Jason Brooks Consultant Anaesthesia and Pain Management BCH March 2017 The Brief A broad overview of the abnormal and normal anatomy and physiology of pain pathways
Neurobiology of Pain Adjuvant analgesia Jason Brooks Consultant Anaesthesia and Pain Management BCH March 2017 The Brief A broad overview of the abnormal and normal anatomy and physiology of pain pathways
Sensory coding and somatosensory system
 Sensory coding and somatosensory system Sensation and perception Perception is the internal construction of sensation. Perception depends on the individual experience. Three common steps in all senses
Sensory coding and somatosensory system Sensation and perception Perception is the internal construction of sensation. Perception depends on the individual experience. Three common steps in all senses
Pain. Pain. Pain: One definition. Pain: One definition. Pain: One definition. Pain: One definition. Psyc 2906: Sensation--Introduction 9/27/2006
 Pain Pain Pain: One Definition Classic Paths A new Theory Pain and Drugs According to the international Association for the Study (Merskey & Bogduk, 1994), Pain is an unpleasant sensory and emotional experience
Pain Pain Pain: One Definition Classic Paths A new Theory Pain and Drugs According to the international Association for the Study (Merskey & Bogduk, 1994), Pain is an unpleasant sensory and emotional experience
The biochemical origin of pain: The origin of all pain is inflammation and the inflammatory response: Inflammatory profile of pain syndromes
 The biochemical origin of pain: The origin of all pain is inflammation and the inflammatory response: Inflammatory profile of pain syndromes 1 Medical Hypothesis 2007, Vol. 69, pp. 1169 1178 Sota Omoigui
The biochemical origin of pain: The origin of all pain is inflammation and the inflammatory response: Inflammatory profile of pain syndromes 1 Medical Hypothesis 2007, Vol. 69, pp. 1169 1178 Sota Omoigui
S E C T I O N I M E C H A N I S M S A N D E P I D E M I O L O G Y
 SECTION I MECHANISMS AND EPIDEMIOLOGY 1 Nociception: basic principles rie suzuki, shafaq sikandar, and anthony h. dickenson University College London Introduction Pain has been a major concern in the clinic
SECTION I MECHANISMS AND EPIDEMIOLOGY 1 Nociception: basic principles rie suzuki, shafaq sikandar, and anthony h. dickenson University College London Introduction Pain has been a major concern in the clinic
Memory Systems II How Stored: Engram and LTP. Reading: BCP Chapter 25
 Memory Systems II How Stored: Engram and LTP Reading: BCP Chapter 25 Memory Systems Learning is the acquisition of new knowledge or skills. Memory is the retention of learned information. Many different
Memory Systems II How Stored: Engram and LTP Reading: BCP Chapter 25 Memory Systems Learning is the acquisition of new knowledge or skills. Memory is the retention of learned information. Many different
SYLLABUS SPRING 2011 COURSE: NSC NEUROBIOLOGY OF PAIN
 SYLLABUS NSC 4358 NEUROBIOLOGY OF PAIN SPRING 2011 1 SYLLABUS SPRING 2011 COURSE: NSC 4358 001 NEUROBIOLOGY OF PAIN Instructor: Aage R. Møller PhD E-mail: AMOLLER@UTDALLAS.EDU Class schedule: Main Campus:
SYLLABUS NSC 4358 NEUROBIOLOGY OF PAIN SPRING 2011 1 SYLLABUS SPRING 2011 COURSE: NSC 4358 001 NEUROBIOLOGY OF PAIN Instructor: Aage R. Møller PhD E-mail: AMOLLER@UTDALLAS.EDU Class schedule: Main Campus:
Organization of the nervous system. [See Fig. 48.1]
![Organization of the nervous system. [See Fig. 48.1] Organization of the nervous system. [See Fig. 48.1]](/thumbs/90/103926552.jpg) Nervous System [Note: This is the text version of this lecture file. To make the lecture notes downloadable over a slow connection (e.g. modem) the figures have been replaced with figure numbers as found
Nervous System [Note: This is the text version of this lecture file. To make the lecture notes downloadable over a slow connection (e.g. modem) the figures have been replaced with figure numbers as found
Chapter 12 Nervous Tissue. Copyright 2009 John Wiley & Sons, Inc. 1
 Chapter 12 Nervous Tissue Copyright 2009 John Wiley & Sons, Inc. 1 Terms to Know CNS PNS Afferent division Efferent division Somatic nervous system Autonomic nervous system Sympathetic nervous system Parasympathetic
Chapter 12 Nervous Tissue Copyright 2009 John Wiley & Sons, Inc. 1 Terms to Know CNS PNS Afferent division Efferent division Somatic nervous system Autonomic nervous system Sympathetic nervous system Parasympathetic
The Nervous System -The master controlling and communicating system of the body
 The Nervous System -The master controlling and communicating system of the body Functions: -Sensory input -Integration -Motor output Organization of the Nervous System Central nervous system (CNS) -Brain
The Nervous System -The master controlling and communicating system of the body Functions: -Sensory input -Integration -Motor output Organization of the Nervous System Central nervous system (CNS) -Brain
Synaptic transmission
 Outline Synaptic transmission Sompol Tapechum M.D., Ph.D. Department of Physiology Faculty of Medicine Siriraj Hospital, Bangkok, Thailand. sisth@mahidol.ac.th 2 Structure of synapse Modes of synaptic
Outline Synaptic transmission Sompol Tapechum M.D., Ph.D. Department of Physiology Faculty of Medicine Siriraj Hospital, Bangkok, Thailand. sisth@mahidol.ac.th 2 Structure of synapse Modes of synaptic
Chapter 16. Sense of Pain
 Chapter 16 Sense of Pain Pain Discomfort caused by tissue injury or noxious stimulation, and typically leading to evasive action important /// helps to protect us lost of pain in diabetes mellitus = diabetic
Chapter 16 Sense of Pain Pain Discomfort caused by tissue injury or noxious stimulation, and typically leading to evasive action important /// helps to protect us lost of pain in diabetes mellitus = diabetic
Venipuncture-induced neuropathic pain: the clinical syndrome, with comparisons to experimental nerve injury models
 Pain 94 (2001) 225 229 Topical review Venipuncture-induced neuropathic pain: the clinical syndrome, with comparisons to experimental nerve injury models Steven H. Horowitz* Division of Neurology, University
Pain 94 (2001) 225 229 Topical review Venipuncture-induced neuropathic pain: the clinical syndrome, with comparisons to experimental nerve injury models Steven H. Horowitz* Division of Neurology, University
Anesthesiology, V 92, No 3, Mar 2000
 691 Anesthesiology 2000; 92:691 8 2000 American Society of Anesthesiologists, Inc. Lippincott Williams & Wilkins, Inc. Heterogenous Patterns of Sensory Dysfunction in Postherpetic Neuralgia Suggest Multiple
691 Anesthesiology 2000; 92:691 8 2000 American Society of Anesthesiologists, Inc. Lippincott Williams & Wilkins, Inc. Heterogenous Patterns of Sensory Dysfunction in Postherpetic Neuralgia Suggest Multiple
Virtually everyone has experienced pain in one
 Transfer of Advances in Sciences into Dental Education Recent Insights into Brainstem Mechanisms Underlying Craniofacial Pain Barry J. Sessle, B.D.S., M.D.S., B.Sc., Ph.D., F.R.S.C., D.Sc. (honorary) Abstract:
Transfer of Advances in Sciences into Dental Education Recent Insights into Brainstem Mechanisms Underlying Craniofacial Pain Barry J. Sessle, B.D.S., M.D.S., B.Sc., Ph.D., F.R.S.C., D.Sc. (honorary) Abstract:
Physiology of synapses and receptors
 Physiology of synapses and receptors Dr Syed Shahid Habib Professor & Consultant Clinical Neurophysiology Dept. of Physiology College of Medicine & KKUH King Saud University REMEMBER These handouts will
Physiology of synapses and receptors Dr Syed Shahid Habib Professor & Consultant Clinical Neurophysiology Dept. of Physiology College of Medicine & KKUH King Saud University REMEMBER These handouts will
What is Pain? An unpleasant sensory and emotional experience associated with actual or potential tissue damage. Pain is always subjective
 Pain & Acupuncture What is Pain? An unpleasant sensory and emotional experience associated with actual or potential tissue damage. NOCICEPTION( the neural processes of encoding and processing noxious stimuli.)
Pain & Acupuncture What is Pain? An unpleasant sensory and emotional experience associated with actual or potential tissue damage. NOCICEPTION( the neural processes of encoding and processing noxious stimuli.)
TRANSCUTANEOUS ELECTRICAL STIMULATION
 TRANSCUTANEOUS ELECTRICAL STIMULATION Transcutaneous electrical stimulation (TENS) Transcutaneous electrical stimulation ; An electronic device that produces electrical signals used to stimulate nerve
TRANSCUTANEOUS ELECTRICAL STIMULATION Transcutaneous electrical stimulation (TENS) Transcutaneous electrical stimulation ; An electronic device that produces electrical signals used to stimulate nerve
Understanding the experience and physiology of pain
 If you would like to contribute to the art and science section contact: Gwen Clarke, art and science editor, Nursing Standard, The Heights, 59-65 Lowlands Road, Harrow-on-the-Hill, Middlesex HA1 3AW. email:
If you would like to contribute to the art and science section contact: Gwen Clarke, art and science editor, Nursing Standard, The Heights, 59-65 Lowlands Road, Harrow-on-the-Hill, Middlesex HA1 3AW. email:
Pain. Types of Pain. Types of Pain 8/21/2013
 Pain 1 Types of Pain Acute Pain Complex combination of sensory, perceptual, & emotional experiences as a result of a noxious stimulus Mediated by rapidly conducting nerve pathways & associated with increased
Pain 1 Types of Pain Acute Pain Complex combination of sensory, perceptual, & emotional experiences as a result of a noxious stimulus Mediated by rapidly conducting nerve pathways & associated with increased
Function of the Nervous System
 Nervous System Function of the Nervous System Receive sensory information, interpret it, and send out appropriate commands to form a response Composed of neurons (functional unit of the nervous system)
Nervous System Function of the Nervous System Receive sensory information, interpret it, and send out appropriate commands to form a response Composed of neurons (functional unit of the nervous system)
Somatic Sensation (MCB160 Lecture by Mu-ming Poo, Friday March 9, 2007)
 Somatic Sensation (MCB160 Lecture by Mu-ming Poo, Friday March 9, 2007) Introduction Adrian s work on sensory coding Spinal cord and dorsal root ganglia Four somatic sense modalities Touch Mechanoreceptors
Somatic Sensation (MCB160 Lecture by Mu-ming Poo, Friday March 9, 2007) Introduction Adrian s work on sensory coding Spinal cord and dorsal root ganglia Four somatic sense modalities Touch Mechanoreceptors
GENERAL PAIN DEFINITIONS
 I. OVERVIEW GENERAL PAIN DEFINITIONS Charles E. Argoff, MD CHAPTER 1 1. What is pain? Some dictionaries define pain as An unpleasant sensation, occurring in varying degrees of severity as a consequence
I. OVERVIEW GENERAL PAIN DEFINITIONS Charles E. Argoff, MD CHAPTER 1 1. What is pain? Some dictionaries define pain as An unpleasant sensation, occurring in varying degrees of severity as a consequence
Neurons, Synapses, and Signaling
 Neurons, Synapses, and Signaling The Neuron is the functional unit of the nervous system. Neurons are composed of a cell body, which contains the nucleus and organelles; Dendrites which are extensions
Neurons, Synapses, and Signaling The Neuron is the functional unit of the nervous system. Neurons are composed of a cell body, which contains the nucleus and organelles; Dendrites which are extensions
CHAPTER 10 THE SOMATOSENSORY SYSTEM
 CHAPTER 10 THE SOMATOSENSORY SYSTEM 10.1. SOMATOSENSORY MODALITIES "Somatosensory" is really a catch-all term to designate senses other than vision, hearing, balance, taste and smell. Receptors that could
CHAPTER 10 THE SOMATOSENSORY SYSTEM 10.1. SOMATOSENSORY MODALITIES "Somatosensory" is really a catch-all term to designate senses other than vision, hearing, balance, taste and smell. Receptors that could
PART IV: NEUROPATHIC PAIN SYNDROMES JILL SINDT FEBRUARY 7, 2019
 PART IV: NEUROPATHIC PAIN SYNDROMES JILL SINDT FEBRUARY 7, 2019 NEUROPATHIC PAIN PAIN ARISING AS DIRECT CONSEQUENCE OF A LESION OR DISEASE AFFECTING THE SOMATOSENSORY SYSTEM AFFECTS 3-8% OF POPULATION
PART IV: NEUROPATHIC PAIN SYNDROMES JILL SINDT FEBRUARY 7, 2019 NEUROPATHIC PAIN PAIN ARISING AS DIRECT CONSEQUENCE OF A LESION OR DISEASE AFFECTING THE SOMATOSENSORY SYSTEM AFFECTS 3-8% OF POPULATION
ANAT2010. Concepts of Neuroanatomy (II) S2 2018
 ANAT2010 Concepts of Neuroanatomy (II) S2 2018 Table of Contents Lecture 13: Pain and perception... 3 Lecture 14: Sensory systems and visual pathways... 11 Lecture 15: Techniques in Neuroanatomy I in vivo
ANAT2010 Concepts of Neuroanatomy (II) S2 2018 Table of Contents Lecture 13: Pain and perception... 3 Lecture 14: Sensory systems and visual pathways... 11 Lecture 15: Techniques in Neuroanatomy I in vivo
The Nervous System Mark Stanford, Ph.D.
 The Nervous System Functional Neuroanatomy and How Neurons Communicate Mark Stanford, Ph.D. Santa Clara Valley Health & Hospital System Addiction Medicine and Therapy Services The Nervous System In response
The Nervous System Functional Neuroanatomy and How Neurons Communicate Mark Stanford, Ph.D. Santa Clara Valley Health & Hospital System Addiction Medicine and Therapy Services The Nervous System In response
NEURAL TISSUE (NEUROPHYSIOLOGY) PART I (A): NEURONS & NEUROGLIA
 PART I (A): NEURONS & NEUROGLIA Neural Tissue Contains 2 kinds of cells: neurons: cells that send and receive signals neuroglia (glial cells): cells that support and protect neurons Neuron Types Sensory
PART I (A): NEURONS & NEUROGLIA Neural Tissue Contains 2 kinds of cells: neurons: cells that send and receive signals neuroglia (glial cells): cells that support and protect neurons Neuron Types Sensory
BIOLOGY 12 NERVOUS SYSTEM PRACTICE
 1 Name: BIOLOGY 12 NERVOUS SYSTEM PRACTICE Date: 1) Identify structures X, Y and Z and give one function of each. 2) Which processes are involved in the movement of molecule Y from point X to point Z?
1 Name: BIOLOGY 12 NERVOUS SYSTEM PRACTICE Date: 1) Identify structures X, Y and Z and give one function of each. 2) Which processes are involved in the movement of molecule Y from point X to point Z?
QUIZ YOURSELF COLOSSAL NEURON ACTIVITY
 QUIZ YOURSELF What are the factors that produce the resting potential? How is an action potential initiated and what is the subsequent flow of ions during the action potential? 1 COLOSSAL NEURON ACTIVITY
QUIZ YOURSELF What are the factors that produce the resting potential? How is an action potential initiated and what is the subsequent flow of ions during the action potential? 1 COLOSSAL NEURON ACTIVITY
Name Biology 125 Midterm #2 ( ) Total Pages: 9
 Name - 1 - Biology 125 Midterm #2 (11-15-07) Part 1: (30 pts) Part 2: (40 pts) Part 3: (6 pts) Part 4: (12 pts) Part 5: (12 pts) Total Pages: 9 Total Score: (100 pts) 1 Name - 2 - Part 1: True or False.
Name - 1 - Biology 125 Midterm #2 (11-15-07) Part 1: (30 pts) Part 2: (40 pts) Part 3: (6 pts) Part 4: (12 pts) Part 5: (12 pts) Total Pages: 9 Total Score: (100 pts) 1 Name - 2 - Part 1: True or False.
Applied physiology: neuropathic pain
 1 Applied physiology: neuropathic pain VICTORIA CJ WALLACE AND ANDREW SC RICE Introduction 3 Animal models of neuropathic pain 4 Mechanisms of neuropathic hypersensitivity 7 Conclusions 16 References 16
1 Applied physiology: neuropathic pain VICTORIA CJ WALLACE AND ANDREW SC RICE Introduction 3 Animal models of neuropathic pain 4 Mechanisms of neuropathic hypersensitivity 7 Conclusions 16 References 16
3) Most of the organelles in a neuron are located in the A) dendritic region. B) axon hillock. C) axon. D) cell body. E) axon terminals.
 Chapter 48 Neurons, Synapses, and Signaling Multiple-Choice Questions 1) A simple nervous system A) must include chemical senses, mechanoreception, and vision. B) includes a minimum of 12 ganglia. C) has
Chapter 48 Neurons, Synapses, and Signaling Multiple-Choice Questions 1) A simple nervous system A) must include chemical senses, mechanoreception, and vision. B) includes a minimum of 12 ganglia. C) has
Mechanism of Pain Production
 Mechanism of Pain Production Pain conducting nerve fibers are small myelinated (A-delta) or unmyelinated nerve fibers (C-fibers). Cell bodies are in the dorsal root ganglia (DRG) or sensory ganglia of
Mechanism of Pain Production Pain conducting nerve fibers are small myelinated (A-delta) or unmyelinated nerve fibers (C-fibers). Cell bodies are in the dorsal root ganglia (DRG) or sensory ganglia of
9/28/2016. Neuron. Multipolar Neuron. Astrocytes Exchange Materials With Neurons. Glia or Glial Cells ( supporting cells of the nervous system)
 Neuron Multipolar Neuron https://www.youtube.com/watch?v=lw-psbnu5xago to :38 Glia or Glial Cells ( supporting cells of the nervous system) 10X more numerous than neurons but one-tenth the size make up
Neuron Multipolar Neuron https://www.youtube.com/watch?v=lw-psbnu5xago to :38 Glia or Glial Cells ( supporting cells of the nervous system) 10X more numerous than neurons but one-tenth the size make up
Biomechanics of Pain: Dynamics of the Neuromatrix
 Biomechanics of Pain: Dynamics of the Neuromatrix Partap S. Khalsa, D.C., Ph.D. Department of Biomedical Engineering The Neuromatrix From: Melzack R (1999) Pain Suppl 6:S121-6. NIOSH STAR Symposium May
Biomechanics of Pain: Dynamics of the Neuromatrix Partap S. Khalsa, D.C., Ph.D. Department of Biomedical Engineering The Neuromatrix From: Melzack R (1999) Pain Suppl 6:S121-6. NIOSH STAR Symposium May
By the end of this lecture the students will be able to:
 UNIT VII: PAIN Objectives: By the end of this lecture the students will be able to: Review the concept of somatosensory pathway. Describe the function of Nociceptors in response to pain information. Describe
UNIT VII: PAIN Objectives: By the end of this lecture the students will be able to: Review the concept of somatosensory pathway. Describe the function of Nociceptors in response to pain information. Describe
Portions from Chapter 6 CHAPTER 7. The Nervous System: Neurons and Synapses. Chapter 7 Outline. and Supporting Cells
 CHAPTER 7 The Nervous System: Neurons and Synapses Chapter 7 Outline Neurons and Supporting Cells Activity in Axons The Synapse Acetylcholine as a Neurotransmitter Monoamines as Neurotransmitters Other
CHAPTER 7 The Nervous System: Neurons and Synapses Chapter 7 Outline Neurons and Supporting Cells Activity in Axons The Synapse Acetylcholine as a Neurotransmitter Monoamines as Neurotransmitters Other
Nervous System Notes
 Nervous System Notes The nervous system consists of a network of nerve cells or neurons. I. A nervous system is an important part of a cell s (or an organism s) ability to respond to the environment. A.
Nervous System Notes The nervous system consists of a network of nerve cells or neurons. I. A nervous system is an important part of a cell s (or an organism s) ability to respond to the environment. A.
LECTURE STRUCTURE ASC171 NERVOUS SYSTEM PART 1: BACKGROUND 26/07/2015. Module 5
 LECTURE STRUCTURE PART 1: Background / Introduction PART 2: Structure of the NS, how it operates PART 3: CNS PART 4: PNS Why did the action potential cross the synaptic junction? To get to the other side
LECTURE STRUCTURE PART 1: Background / Introduction PART 2: Structure of the NS, how it operates PART 3: CNS PART 4: PNS Why did the action potential cross the synaptic junction? To get to the other side
NEURONS COMMUNICATE WITH OTHER CELLS AT SYNAPSES 34.3
 NEURONS COMMUNICATE WITH OTHER CELLS AT SYNAPSES 34.3 NEURONS COMMUNICATE WITH OTHER CELLS AT SYNAPSES Neurons communicate with other neurons or target cells at synapses. Chemical synapse: a very narrow
NEURONS COMMUNICATE WITH OTHER CELLS AT SYNAPSES 34.3 NEURONS COMMUNICATE WITH OTHER CELLS AT SYNAPSES Neurons communicate with other neurons or target cells at synapses. Chemical synapse: a very narrow
Acetylcholine (ACh) Action potential. Agonists. Drugs that enhance the actions of neurotransmitters.
 Acetylcholine (ACh) The neurotransmitter responsible for motor control at the junction between nerves and muscles; also involved in mental processes such as learning, memory, sleeping, and dreaming. (See
Acetylcholine (ACh) The neurotransmitter responsible for motor control at the junction between nerves and muscles; also involved in mental processes such as learning, memory, sleeping, and dreaming. (See
The Nervous System: Neural Tissue Pearson Education, Inc.
 13 The Nervous System: Neural Tissue Introduction Nervous System Characteristics Controls and adjust the activity of the body Provides swift but brief responses The nervous system includes: Central Nervous
13 The Nervous System: Neural Tissue Introduction Nervous System Characteristics Controls and adjust the activity of the body Provides swift but brief responses The nervous system includes: Central Nervous
MANAGING CHRONIC PAIN
 George Hardas MANAGING CHRONIC PAIN The guide to understanding chronic pain and how to manage it. George Hardas MMed (UNSW) MScMed (Syd) MChiro (Macq) BSc (Syd) Grad Cert Pain Management (Syd) Cognitive
George Hardas MANAGING CHRONIC PAIN The guide to understanding chronic pain and how to manage it. George Hardas MMed (UNSW) MScMed (Syd) MChiro (Macq) BSc (Syd) Grad Cert Pain Management (Syd) Cognitive
Risk Factors That Predispose Patients to Orofacial Pain
 Risk Factors That Predispose Patients to Orofacial Pain Paul Durham, PhD Distinguished Professor of Cell Biology Director Center for Biomedical & Life Sciences Missouri State University Cluster Headache
Risk Factors That Predispose Patients to Orofacial Pain Paul Durham, PhD Distinguished Professor of Cell Biology Director Center for Biomedical & Life Sciences Missouri State University Cluster Headache
Chapter 2. The Cellular and Molecular Basis of Cognition Cognitive Neuroscience: The Biology of the Mind, 2 nd Ed.,
 Chapter 2. The Cellular and Molecular Basis of Cognition Cognitive Neuroscience: The Biology of the Mind, 2 nd Ed., M. S. Gazzaniga, R. B. Ivry, and G. R. Mangun, Norton, 2002. Summarized by B.-W. Ku,
Chapter 2. The Cellular and Molecular Basis of Cognition Cognitive Neuroscience: The Biology of the Mind, 2 nd Ed., M. S. Gazzaniga, R. B. Ivry, and G. R. Mangun, Norton, 2002. Summarized by B.-W. Ku,
Chapter 2. The Cellular and Molecular Basis of Cognition
 Chapter 2. The Cellular and Molecular Basis of Cognition Cognitive Neuroscience: The Biology of the Mind, 2 nd Ed., M. S. Gazzaniga,, R. B. Ivry,, and G. R. Mangun,, Norton, 2002. Summarized by B.-W. Ku,
Chapter 2. The Cellular and Molecular Basis of Cognition Cognitive Neuroscience: The Biology of the Mind, 2 nd Ed., M. S. Gazzaniga,, R. B. Ivry,, and G. R. Mangun,, Norton, 2002. Summarized by B.-W. Ku,
Neuropathic Pain. Scott Magnuson, MD Pain Management of North Idaho, PLLC
 Neuropathic Pain Scott Magnuson, MD Pain Management of North Idaho, PLLC Pain is our friend "An unpleasant sensory and emotional experience associated with actual or potential tissue damage, or described
Neuropathic Pain Scott Magnuson, MD Pain Management of North Idaho, PLLC Pain is our friend "An unpleasant sensory and emotional experience associated with actual or potential tissue damage, or described
STRUCTURAL ELEMENTS OF THE NERVOUS SYSTEM
 STRUCTURAL ELEMENTS OF THE NERVOUS SYSTEM STRUCTURE AND MAINTENANCE OF NEURONS (a) (b) Dendrites Cell body Initial segment collateral terminals (a) Diagrammatic representation of a neuron. The break in
STRUCTURAL ELEMENTS OF THE NERVOUS SYSTEM STRUCTURE AND MAINTENANCE OF NEURONS (a) (b) Dendrites Cell body Initial segment collateral terminals (a) Diagrammatic representation of a neuron. The break in
CHAPTER 4 PAIN AND ITS MANAGEMENT
 CHAPTER 4 PAIN AND ITS MANAGEMENT Pain Definition: An unpleasant sensory and emotional experience associated with actual or potential tissue damage, or described in terms of such damage. Types of Pain
CHAPTER 4 PAIN AND ITS MANAGEMENT Pain Definition: An unpleasant sensory and emotional experience associated with actual or potential tissue damage, or described in terms of such damage. Types of Pain
Acute Pain NETP: SEPTEMBER 2013 COHORT
 Acute Pain NETP: SEPTEMBER 2013 COHORT Pain & Suffering an unpleasant sensory & emotional experience associated with actual or potential tissue damage, or described in terms of such damage International
Acute Pain NETP: SEPTEMBER 2013 COHORT Pain & Suffering an unpleasant sensory & emotional experience associated with actual or potential tissue damage, or described in terms of such damage International
Chapter 17 Nervous System
 Chapter 17 Nervous System 1 The Nervous System Two Anatomical Divisions Central Nervous System (CNS) Brain and Spinal Cord Peripheral Nervous System (PNS) Two Types of Cells Neurons Transmit nerve impulses
Chapter 17 Nervous System 1 The Nervous System Two Anatomical Divisions Central Nervous System (CNS) Brain and Spinal Cord Peripheral Nervous System (PNS) Two Types of Cells Neurons Transmit nerve impulses
The Nervous System. Nervous System Functions 1. gather sensory input 2. integration- process and interpret sensory input 3. cause motor output
 The Nervous System Nervous System Functions 1. gather sensory input 2. integration- process and interpret sensory input 3. cause motor output The Nervous System 2 Parts of the Nervous System 1. central
The Nervous System Nervous System Functions 1. gather sensory input 2. integration- process and interpret sensory input 3. cause motor output The Nervous System 2 Parts of the Nervous System 1. central
Peripheral nerve injury alters excitatory synaptic transmission in lamina II of the rat dorsal horn
 J Physiol (2003), 548.1, pp. 131 138 DOI: 10.1113/jphysiol.2002.036186 The Physiological Society 2003 www.jphysiol.org Peripheral nerve injury alters excitatory synaptic transmission in lamina II of the
J Physiol (2003), 548.1, pp. 131 138 DOI: 10.1113/jphysiol.2002.036186 The Physiological Society 2003 www.jphysiol.org Peripheral nerve injury alters excitatory synaptic transmission in lamina II of the
The Nervous System. Chapter 4. Neuron 3/9/ Components of the Nervous System
 Chapter 4 The Nervous System 1. Components of the Nervous System a. Nerve cells (neurons) Analyze and transmit information Over 100 billion neurons in system Four defined regions Cell body Dendrites Axon
Chapter 4 The Nervous System 1. Components of the Nervous System a. Nerve cells (neurons) Analyze and transmit information Over 100 billion neurons in system Four defined regions Cell body Dendrites Axon
