SUPPLEMENTARY APPENDIX 1: Methods
|
|
|
- Arnold Ramsey
- 5 years ago
- Views:
Transcription
1 SUPPLEMENTARY APPENDIX 1: Methods Methodology Overview We developed this guideline following the ACR guideline development process ( Guidelines). This process includes the Grading of Recommendations Assessment, Development, and Evaluation (GRADE) methodology ( (1-3). Teams Involved A Core Leadership Team (4 members) supervised the project and was responsible for defining the scope, drafting the clinical (Patient/Intervention/Comparator/Outcomes - PICO) questions, coordinating with the Literature Review Team, overseeing the voting process, and drafting the manuscript. The Core Team, together with the Literature Review Team, was comprised of individuals with content and methodological expertise, and included a GRADE methodologist who advised on the process of developing and presenting the evidence and provided input on the quality assessment of evidence and summary of findings (SoF) tables (provided in Supplementary Appendix 3). The Literature Review Team (5 members) conducted a systematic search, assessed study quality, extracted data, computed pooled estimates of outcomes, graded the quality of overall evidence, generated the SoF tables, and compiled the resulting evidence into an evidence report. The role of the Expert Panel, composed of 9 content experts, was to provide consultation and feedback on the PICO questions. The Voting Panel (16 members) included adult and pediatric rheumatologists, internists, a nephrologist, a pulmonologist, a gastroenterologist, medical specialists with expertise and clinical experience in treating glucocorticoid induced OP (GIOP), and a patient representative. Their role was to participate in the development of the PICO questions, including
2 making judgments regarding the relative importance of the outcomes, and vote on the PICO questions based on the evidence report and their expertise. The ACR provided training for everyone involved in the development of this guideline, which included sessions on the ACR guideline process and GRADE methodology. See Supplementary Appendix 2 for team/panel rosters. Disclosures and Management of Conflicts of Interest Per ACR policy, everyone who was intellectually involved in the project (i.e., considered for guideline authorship) disclosed all relationships ( Summary.pdf). Disclosures were compared against a previously drafted list of affected companies (i.e., companies or organizations that were considered reasonably likely to be positively or negatively affected by care delivered in accordance with the guideline) to determine which relationships were considered conflicts of interest for purposes of this project. Individuals whose primary employment (> 51% of work time/effort) was with a company that manufactured or sold therapeutics or diagnostics were not eligible to participate. The project s principal investigator (PI) and literature review leader had no relevant conflicts of interest for the full 12 months before this project began, and all but one of the guideline development team members, including the PI and literature review leader, had no relevant conflicts of interest for the duration of the project. A participant who had any relationship with an affected company was counted as conflicted (i.e., toward the allowed threshold) regardless of the type or subject of the relationship. Intellectual conflicts, such as a prior publication or scientific presentation on a GIOP therapy, were recognized as important and were required to be disclosed, but because they were ubiquitous, participants with intellectual conflicts were not counted as conflicted (i.e., toward the allowed threshold) based on their intellectual conflict alone.
3 Participant disclosures were included in the project plan that was posted online for public comment (see description below). In addition, disclosures of all participants were shared, in writing, with each project participant. At the face-to-face Voting Panel meeting, verbal disclosures were provided before any content discussion. Updated participant disclosures, as well as ACR committee reviewer disclosures, are included online with this manuscript. Finally, author disclosures are also included in this paper. Scope and Target Audience The scope of this project included the assessment, prevention and treatment of OP and fractures in children and adults taking glucocorticoids (prednisone dose of > 2.5 mg of prednisone for 3 months), including patients with organ transplant who are treated with GCs. Clinical situations not addressed by this guideline include treatment of people with stage 4-5 chronic kidney disease and people who use inhaled GCs. The target audience for this guideline includes patients with or at risk for GIOP and their clinicians. The ACR plans to develop derivative products, including a pocket card, an app version of this guideline, and a patient education tool to facilitate implementation. Establishing Key Principles and PICO Development The Core Team collaborated with the Expert Panel to develop the initial set of PICO-formatted clinical questions for the guideline (4). The panel ranked fracture (hip, vertebral, non-vertebral) as the only critically important outcome for treatment evaluation. Important outcomes included adverse effects of treatments, in particular the incidence of serious and total adverse events (AEs) in all clinical scenarios. These events included upper gastrointestinal AEs for bisphosphonates; rates of transplant rejections, mortality, and hypo-or hypercalcaemia in transplant recipients; and maternal and fetal risks for women of child-bearing potential. The PICO questions were posted for 30 days on the ACR web site for public comment and revised accordingly.
4 Framework for the GIOP Guideline Development At the initial scoping meeting, the group agreed that the scope of the populations to be addressed would include adult men and postmenopausal women and special populations that have unique risks, such as organ transplant recipients, women of childbearing potential, children, and people receiving very high-dose GCs (defined as one or more courses of high dose GCs (initial dose of 30 mg/day of prednisone or equivalent) and a cumulative dose of 5 grams (5,6). Chronic GC use was defined as 3 months in duration. Adult men and women were divided into two groups based on age ( 40 years, < 40 years) because tools to predict absolute fracture risk are available only for adults 40 years. After defining population risk groups, interventions and comparators were specified for each PICO question (see list of PICO questions in Supplementary Table 1). The Core Team agreed that while bone mineral density (BMD) data would be examined, the critical outcome for the analysis and literature search was fracture particularly vertebral fracture. Vertebral fractures are more common than femoral fractures in GC treated patients and GIOP clinical trials are not of adequate size to assess the impact of interventions on femoral fractures. When necessary to use BMD to support a recommendation (which was the case in only four PICO questions, all addressing pediatric GIOP patients), the Voting Panel rated down the quality of evidence for indirectness. PICO questions included assessment and reassessment of fracture risks, treatment comparisons, and questions about duration and reassessment of treatment. Systematic Synthesis of the Literature Literature Searches To identify relevant evidence for the PICO questions, a medical librarian, in collaboration with the Literature Review Team, performed systematic searches of the
5 published English language literature. We searched OVID Medline, PubMed, Embase, and the Cochrane Library (including Cochrane Database of Systematic Reviews; Database of Abstracts of Reviews of Effects (DARE); Cochrane Central Register of Controlled Trials (CENTRAL); and Health Technology Assessments (HTA)) from the beginning of each database through October 6-8, 2015 (Supplementary Appendix 4), and update searches were conducted on April 23, For PICO questions for which we found no direct evidence in the GIOP field, we sought indirect evidence, in particular meta-analyses of randomized trials, in the non-giop populations. Study Selection We used DistillerSR software ( to screen the literature search results. We performed duplicate screening of each title and abstract using two independent reviewers from among a pool of reviewers, with a third reviewer resolving conflicts. Eligible articles underwent full-text screening by two independent reviewers. Selected manuscripts were matched to PICO questions. See Supplementary Appendix 5 for details related to the study selection process. Data Extraction and Analysis We extracted data from RCTs for each PICO question into RevMan software ( The quality of each study was assessed using the Cochrane risk of bias tool ( The critical/important outcomes (fractures and adverse events) selected for this guideline were binary, and they were analyzed using the Mantel-Haenszel method in a random effects model and reported as relative risks with 95% confidence intervals. In clinical scenarios not addressed by RCT data (e.g., certain special populations, such as patients on high-dose steroids), we used data from observational cohort studies to estimate relative effects. In situations in which the question had not
6 been tested in a sample taking glucocorticoids but had been tested in a non-giop population, we applied the relative risk values from that study, making the assumption that the effect was generalizable but rating down the quality of evidence for indirectness. After calculating relative treatment effects for the pooled data for each PICO question, we quantified overall absolute risk reduction using pooled control group fracture rates. Next, in order to project absolute risk reduction within each risk strata, we constructed a risk calculator to display the absolute risk reduction in vertebral fracture rates over five years, depending on hypothetical baseline fracture risk ranging from 1% to 20%. We used the following cut-points to stratify levels of risk: low risk, viewed as < 5% incidence of vertebral fractures over 5 years; medium risk, 5 to < 10%; and high risk, 10%. The Voting Panel then made recommendations based on absolute fracture reduction with treatment in each of these strata. We focused on vertebral fracture rates because this outcome was more consistently reported in the literature and because of the greater effects of GCs on trabecular bone. Evidence Report Formulation We exported RevMan files into GRADEpro software to formulate a GRADE summary of findings (SoF) table for each PICO question (4). The quality of evidence for each outcome was evaluated in duplicate by three independent reviewers using GRADE quality assessment criteria (1) with discordance resolved by discussion. We compiled the resulting SoF tables in an evidence report (Supplementary Appendix 3). The Core Team reviewed the evidence report and addressed possible evidence gaps prior to presentation to the Voting Panel. Moving from Evidence to Recommendations GRADE methodology specifies that panels make recommendations based on a consideration of the balance of relative benefits and harms of the treatment options
7 under consideration, the quality of the evidence (i.e., confidence in the effect estimates), and patients values and preferences. Key to the recommendation is the trade-off between desirable and undesirable outcomes; recommendations require estimating the relative value patients place in the outcomes. A recommendation could be either in favor or against the proposed intervention and either strong or conditional. According to GRADE, a recommendation is categorized as strong if the panel is very confident that the benefits of an intervention clearly outweigh the harms (or vice versa); a conditional recommendation denotes uncertainty over the balance of benefits and harms, such as when the evidence quality is low or very low, or when patient preferences or costs are expected to impact the decision. Thus, conditional recommendations refer to decisions where incorporation of patient preferences is an essential element of decision making. The Voting Panel s assessment was that patients would be willing to use calcium and vitamin D with only a very small absolute risk reduction, and that all or virtually all would be willing to use bisphosphonates to achieve a 5-year absolute reduction in vertebral fracture risk of 5%, and that most would choose to use oral bisphosphonates if the fracture reduction were 3% to <5% (leading to a conditional recommendation in favor) The 5-year time period was chosen because few clinical trials have data on fracture risk reduction past 3-5 years. Further, the panel thought that most patients would decline oral bisphosphonates with an absolute reduction in 5-year risk of vertebral fractures of 1.6% to 2.9% (leading to a conditional recommendation against) and all or virtually all would decline if the risk reduction were less than 1.5% (leading, in the presence of high or moderate quality evidence, to a strong recommendation against). For IV bisphosphonates, denosumab, raloxifene, and teriparatide, which have greater harms or burden of treatment, the threshold was higher, though the panel did not specify a threshold value. Because raloxifene may increase the risk of death due to
8 stroke in postmenopausal women with documented coronary heart disease or at increased risk for major coronary events and/or increase the risk of deep vein thrombosis and pulmonary embolism (7), and there is no evidence of its benefit in fracture reduction in GC-treated patients, the Voting Panel considered the drug as a treatment option only for postmenopausal women with contraindications to all other treatments. We are unaware of published literature exploring patient values and preferences regarding these issues. Our judgments are based on the experience of the panel members (which included a patient) in shared decision making with their patients. Consensus Building The Voting Panel received the evidence report for review before it met to discuss and decide on the final recommendations. During a face-to-face meeting held on May 14, 2016, and subsequent conference calls and s, the Voting Panel, for each PICO question, reviewed the evidence and provided votes on the direction and strength of the related recommendation. The initial voting process was conducted using Poll Everywhere software ( with follow-up conference calls to vote on unresolved questions. An 80% consensus was used as the threshold for a recommendation; if 80% consensus was not achieved during an initial vote, the panel members held additional discussions before re-voting. In some instances, the Voting Panel decided to combine recommendation statements comparing treatment options (calcium and vitamin D, lifestyle changes) for all patient groups. They then voted on a new recommendation statement that combined those treatment options instead of considering the interventions separately for each risk group. When considering post-transplant patient populations with GFR 30 and no evidence of metabolic bone disease, the panel decided that despite the availability of transplant-specific evidence, the recommendations should be identical to
9 the same-age recommendations for adults without transplants. Those with GFR < 30 should be referred to the care of an expert in osteoporosis and metabolic bone disease because of the difference in harms and benefits of treatment in this group. In addition, the Voting Panel dropped a number of PICO questions because the clinical scenario was uncommon, irrelevant, or redundant (see Supplementary Table 1), or because one of the treatment options for that scenario had been eliminated by another recommendation. Consistent with GRADE guidance, in some instances, the Voting Panel chose to provide a strong recommendation despite a low quality rating of evidence (3). In such cases, a written explanation is provided describing the reasons behind this decision. Final Review and Approval of the Manuscript by the ACR In additional to journal peer reviews, the manuscript was reviewed by the ACR Guideline Subcommittee, ACR Quality of Care Committee, and the ACR Board of Directors including over 40 reviewers ( Summary.pdf). These ACR oversight groups did not mandate that certain recommendations be made within the guideline, but rather, these ACR committees served as peer reviewers. Moving From Recommendations to Practice When applying these risk-stratified recommendations in clinical settings, adults 40 years of age on chronic GCs should be designated to be at high, moderate, or low risk of fracture (Table 1) based on BMD, history of fracture, and 10-year risk of major OP fracture and hip fracture using a tool that combines risk factors with GC dose. Although many tools are available, the Voting Panel suggested using FRAX (Figure 1) for fracture risk assessment. When GC use is included as a risk factor in FRAX, the fracture risk generated is the risk associated with a prednisolone dose of mg of per day
10 (prednisolone and prednisone doses are nearly equivalent). For people taking 7.5 mg per day, the fracture risk generated by FRAX should be increased by a relative 15% for major osteoporotic fracture (MOF) and 20% for hip fracture risk (8). For example, if the 10-year hip fracture risk was 2.0% with glucocorticoids entered in FRAX, the risk should be increased to 2.4% if the prednisone dose is 7.5%. There are no tools available to estimate absolute fracture risk in children or adults < 40 years of age. These groups were considered to be at high fracture risk if they had already sustained an OP fracture. The Voting Panel designated men and women < 40 years of age to be moderate to high fracture risk if they were expected to continue GC treatment at 7.5 mg per day for 6 months and had a BMD Z score < -3 at the hip or spine or a rapid decline in BMD (equivalent to 10% at the hip or spine in 1 year) during GC treatment.
11 REFERENCES 1. Guyatt GH, Oxman AD, Vist GE, Kunz R, Falck-Ytter Y, Alonso-Coello P, et al. GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. BMJ. 2008;336(7650): Andrews J, Guyatt G, Oxman AD, Alderson P, Dahm P, Falck-Ytter Y, et al. GRADE guidelines: 14. Going from evidence to recommendations: the significance and presentation of recommendations. J Clin Epidemiol. 2013;66(7): Andrews JC, Schunemann HJ, Oxman AD, Pottie K, Meerpohl JJ, Coello PA, et al. GRADE guidelines: 15. Going from evidence to recommendation-determinants of a recommendation's direction and strength. J Clin Epidemiol. 2013;66(7): Guyatt GH, Oxman AD, Kunz R, Atkins D, Brozek J, Vist G, et al. GRADE guidelines: 2. Framing the question and deciding on important outcomes. J Clin Epidemiol 2011;64: van Staa TP, Geusens P, Pols HA, de Laet C, Leufkens HG, Cooper C. A simple score for estimating the long term risk of fracture in patients using oral glucocorticoids. QJM 2005;98: De Vries F, Bracke M, Leufkens HG, Lammers JW, Cooper C, van Staa TP. Fracture risk with intermittent high-dose oral glucocorticoid therapy. Arthritis Rheum. 2007:56: EVISTA (raloxifene hydrochloride) Tablet for Oral Use Initial U.S. Approval: URL: 8. Guyatt GH, Schünemann HJ, Djulbegovic B, Akl EA. Guideline panels should not GRADE good practice statements. J Clin Epidemiol. 2015;68(5):
SSAI Clinical Practice Committee guideline work flow v2. A. Formal matters
 SSAI Clinical Practice Committee guideline work flow v2 The following document outlines key steps (Figure 1) in the development and/or endorsement of Scandinavian SSAI clinical practice guidelines. A.
SSAI Clinical Practice Committee guideline work flow v2 The following document outlines key steps (Figure 1) in the development and/or endorsement of Scandinavian SSAI clinical practice guidelines. A.
Osteoporosis/Fracture Prevention
 Osteoporosis/Fracture Prevention NATIONAL GUIDELINE SUMMARY This guideline was developed using an evidence-based methodology by the KP National Osteoporosis/Fracture Prevention Guideline Development Team
Osteoporosis/Fracture Prevention NATIONAL GUIDELINE SUMMARY This guideline was developed using an evidence-based methodology by the KP National Osteoporosis/Fracture Prevention Guideline Development Team
Osteoporosis: A Tale of 3 Task Forces!
 Osteoporosis: A Tale of 3 Task Forces! Robert A. Adler, MD McGuire Veterans Affairs Medical Center Virginia Commonwealth University Richmond, Virginia, USA Disclosures The opinions are those of the speaker
Osteoporosis: A Tale of 3 Task Forces! Robert A. Adler, MD McGuire Veterans Affairs Medical Center Virginia Commonwealth University Richmond, Virginia, USA Disclosures The opinions are those of the speaker
Osteoporosis: A Tale of 3 Task Forces!
 Osteoporosis: A Tale of 3 Task Forces! Robert A. Adler, MD McGuire Veterans Affairs Medical Center Virginia Commonwealth University Richmond, Virginia, USA Disclosures The opinions are those of the speaker
Osteoporosis: A Tale of 3 Task Forces! Robert A. Adler, MD McGuire Veterans Affairs Medical Center Virginia Commonwealth University Richmond, Virginia, USA Disclosures The opinions are those of the speaker
Guideline development in TB diagnostics. Karen R Steingart, MD, MPH McGill University, Montreal, July 2011
 Guideline development in TB diagnostics Karen R Steingart, MD, MPH McGill University, Montreal, July 2011 karenst@uw.edu Overview What are guidelines? Quality of guidelines The Grade approach IOM standards
Guideline development in TB diagnostics Karen R Steingart, MD, MPH McGill University, Montreal, July 2011 karenst@uw.edu Overview What are guidelines? Quality of guidelines The Grade approach IOM standards
Systematic reviews and meta-analyses of observational studies (MOOSE): Checklist.
 Systematic reviews and meta-analyses of observational studies (MOOSE): Checklist. MOOSE Checklist Infliximab reduces hospitalizations and surgery interventions in patients with inflammatory bowel disease:
Systematic reviews and meta-analyses of observational studies (MOOSE): Checklist. MOOSE Checklist Infliximab reduces hospitalizations and surgery interventions in patients with inflammatory bowel disease:
Evaluating the Strength of Clinical Recommendations in the Medical Literature: GRADE, SORT, and AGREE
 Evaluating the Strength of Clinical Recommendations in the Medical Literature: GRADE, SORT, and AGREE Mayra Buainain de Castro Maymone 1, Stephanie D. Gan 2 and Michael Bigby 3 Journal of Investigative
Evaluating the Strength of Clinical Recommendations in the Medical Literature: GRADE, SORT, and AGREE Mayra Buainain de Castro Maymone 1, Stephanie D. Gan 2 and Michael Bigby 3 Journal of Investigative
8/6/2018. Glucocorticoid induced osteoporosis: overlooked and undertreated? Disclosure. Objectives. Overview
 Disclosure Glucocorticoid induced osteoporosis: overlooked and undertreated? I have no financial disclosure relevant to this presentation Tasma Harindhanavudhi, MD Division of Diabetes and Endocrinology
Disclosure Glucocorticoid induced osteoporosis: overlooked and undertreated? I have no financial disclosure relevant to this presentation Tasma Harindhanavudhi, MD Division of Diabetes and Endocrinology
1
 www.osteoporosis.ca 1 2 Overview of the Presentation Osteoporosis: An Overview Bone Basics Diagnosis of Osteoporosis Drug Therapies Risk Reduction Living with Osteoporosis 3 What is Osteoporosis? Osteoporosis:
www.osteoporosis.ca 1 2 Overview of the Presentation Osteoporosis: An Overview Bone Basics Diagnosis of Osteoporosis Drug Therapies Risk Reduction Living with Osteoporosis 3 What is Osteoporosis? Osteoporosis:
Systematic Reviews and Meta- Analysis in Kidney Transplantation
 Systematic Reviews and Meta- Analysis in Kidney Transplantation Greg Knoll MD MSc Associate Professor of Medicine Medical Director, Kidney Transplantation University of Ottawa and The Ottawa Hospital KRESCENT
Systematic Reviews and Meta- Analysis in Kidney Transplantation Greg Knoll MD MSc Associate Professor of Medicine Medical Director, Kidney Transplantation University of Ottawa and The Ottawa Hospital KRESCENT
AMERICAN COLLEGE OF RHEUMATOLOGY POSITION STATEMENT. Committee on Rheumatologic Care
 AMERICAN COLLEGE OF RHEUMATOLOGY POSITION STATEMENT SUBJECT: PRESENTED BY: FOR DISTRIBUTION TO: Bone Mineral Density Measurement and the Role of Rheumatologists in the Management of Osteoporosis Committee
AMERICAN COLLEGE OF RHEUMATOLOGY POSITION STATEMENT SUBJECT: PRESENTED BY: FOR DISTRIBUTION TO: Bone Mineral Density Measurement and the Role of Rheumatologists in the Management of Osteoporosis Committee
Grading the Evidence Developing the Typhoid Statement. Manitoba 10 th Annual Travel Conference April 26, 2012
 Grading the Evidence Developing the Typhoid Statement Manitoba 10 th Annual Travel Conference April 26, 2012 Disclosure of Potential Conflict of Interest Alexandra Henteleff MEd, BN, RN Certificate in
Grading the Evidence Developing the Typhoid Statement Manitoba 10 th Annual Travel Conference April 26, 2012 Disclosure of Potential Conflict of Interest Alexandra Henteleff MEd, BN, RN Certificate in
GRADE. Grading of Recommendations Assessment, Development and Evaluation. British Association of Dermatologists April 2018
 GRADE Grading of Recommendations Assessment, Development and Evaluation British Association of Dermatologists April 2018 Previous grading system Level of evidence Strength of recommendation Level of evidence
GRADE Grading of Recommendations Assessment, Development and Evaluation British Association of Dermatologists April 2018 Previous grading system Level of evidence Strength of recommendation Level of evidence
Guideline Development at the American College of Physicians. American College of Physicians
 Guideline Development at the American College of Physicians Melissa Starkey, PhD American College of Physicians Institute of Medicine Committee on Preventive Services for Women March 9, 2011 Who is ACP?
Guideline Development at the American College of Physicians Melissa Starkey, PhD American College of Physicians Institute of Medicine Committee on Preventive Services for Women March 9, 2011 Who is ACP?
Introduzione al metodo GRADE
 Introduzione al metodo GRADE Atto Billio MD, MSc EBHC Ematologia e TMO-Bolzano Gruppo linee guida SIE Critique of EBM De-emphasizes patient values Doesn t account for individual variation Devalues clinical
Introduzione al metodo GRADE Atto Billio MD, MSc EBHC Ematologia e TMO-Bolzano Gruppo linee guida SIE Critique of EBM De-emphasizes patient values Doesn t account for individual variation Devalues clinical
The Joanna Briggs Institute Reviewers Manual 2014
 The Joanna Briggs Institute Reviewers Manual 2014 Summary of Findings Tables for Joanna Briggs Institute Systematic Reviews www.joannabriggs.org Joanna Briggs Institute Reviewers Manual: 2014 edition/supplement
The Joanna Briggs Institute Reviewers Manual 2014 Summary of Findings Tables for Joanna Briggs Institute Systematic Reviews www.joannabriggs.org Joanna Briggs Institute Reviewers Manual: 2014 edition/supplement
Learning Objectives. Controversies in Osteoporosis Prevention and Management. Etiology. Presenter Disclosure Information. Epidemiology.
 12:45 1:30pm Controversies in Osteoporosis Prevention and Management SPEAKER Carolyn Crandall, MD, MS Presenter Disclosure Information The following relationships exist related to this presentation: Carolyn
12:45 1:30pm Controversies in Osteoporosis Prevention and Management SPEAKER Carolyn Crandall, MD, MS Presenter Disclosure Information The following relationships exist related to this presentation: Carolyn
Disclosures. Diagnostic Challenges in Osteoporosis: Whom To Treat 9/25/2014
 Disclosures Diagnostic Challenges in Osteoporosis: Whom To Treat Ethel S. Siris, MD Columbia University Medical Center New York, NY Consultant on scientific issues for: AgNovos Amgen Eli Lilly Merck Novartis
Disclosures Diagnostic Challenges in Osteoporosis: Whom To Treat Ethel S. Siris, MD Columbia University Medical Center New York, NY Consultant on scientific issues for: AgNovos Amgen Eli Lilly Merck Novartis
Chapter 1: Introduction and Methodology
 Chapter 1: Introduction and Methodology These guidelines were developed by an expert panel composed of health care professionals with expertise in family medicine, general internal medicine, adult and
Chapter 1: Introduction and Methodology These guidelines were developed by an expert panel composed of health care professionals with expertise in family medicine, general internal medicine, adult and
Submission to the National Institute for Clinical Excellence on
 Submission to the National Institute for Clinical Excellence on Strontium ranelate for the prevention of osteoporotic fractures in postmenopausal women with osteoporosis by The Society for Endocrinology
Submission to the National Institute for Clinical Excellence on Strontium ranelate for the prevention of osteoporotic fractures in postmenopausal women with osteoporosis by The Society for Endocrinology
ACR OA Guideline Development Process Knee and Hip
 ACR OA Guideline Development Process Knee and Hip 1. Literature searching frame work Literature searches were developed based on the scenarios. A comprehensive search strategy was used to guide the process
ACR OA Guideline Development Process Knee and Hip 1. Literature searching frame work Literature searches were developed based on the scenarios. A comprehensive search strategy was used to guide the process
Guidelines for the Pharmaceutical Management of Osteoporosis in Adult WA Public Hospitals
 WA.DRUG EVALUATION PANEL Guidelines for the Pharmaceutical Management of Osteoporosis in Adult WA Public Hospitals Introduction Osteoporotic fracture-related hospitalisations impose a substantial financial
WA.DRUG EVALUATION PANEL Guidelines for the Pharmaceutical Management of Osteoporosis in Adult WA Public Hospitals Introduction Osteoporotic fracture-related hospitalisations impose a substantial financial
Systematic Review & Course outline. Lecture (20%) Class discussion & tutorial (30%)
 Systematic Review & Meta-analysisanalysis Ammarin Thakkinstian, Ph.D. Section for Clinical Epidemiology and Biostatistics Faculty of Medicine, Ramathibodi Hospital Tel: 02-201-1269, 02-201-1762 Fax: 02-2011284
Systematic Review & Meta-analysisanalysis Ammarin Thakkinstian, Ph.D. Section for Clinical Epidemiology and Biostatistics Faculty of Medicine, Ramathibodi Hospital Tel: 02-201-1269, 02-201-1762 Fax: 02-2011284
Allergen immunotherapy for the treatment of allergic rhinitis and/or asthma
 ril 2014 Allergen immunotherapy for the treatment of allergic rhinitis and/or asthma FINAL COMPREHENSIVE RESEARCH PLAN June 2015 Study Team: Systematic Review Unit FINAL COMPREHENSIVE RESEARCH PLAN: Systematic
ril 2014 Allergen immunotherapy for the treatment of allergic rhinitis and/or asthma FINAL COMPREHENSIVE RESEARCH PLAN June 2015 Study Team: Systematic Review Unit FINAL COMPREHENSIVE RESEARCH PLAN: Systematic
SCORECARD Questionnaire September 2012
 Introduction SCORECARD Questionnaire September 2012 The Osteoporosis Advisory Panel is an independent project chaired by our president Professor John Kanis, supported by a multi-stakeholder group of experts
Introduction SCORECARD Questionnaire September 2012 The Osteoporosis Advisory Panel is an independent project chaired by our president Professor John Kanis, supported by a multi-stakeholder group of experts
An evidence-based laboratory medicine approach to evaluate new laboratory tests
 Meeting the needs of Mediterranean nations: improving efficiency in laboratory medicine This is a Platinum Open Access Journal distributed under the terms of the Creative Commons Attribution Non-Commercial
Meeting the needs of Mediterranean nations: improving efficiency in laboratory medicine This is a Platinum Open Access Journal distributed under the terms of the Creative Commons Attribution Non-Commercial
Osteodensitometry in primary and secondary osteoporosis
 Osteodensitometry in primary and secondary osteoporosis Institut für Qualität und Wirtschaftlichkeit im Gesundheitswesen (IQWiG) Research question The main goal of the present research was the assessment
Osteodensitometry in primary and secondary osteoporosis Institut für Qualität und Wirtschaftlichkeit im Gesundheitswesen (IQWiG) Research question The main goal of the present research was the assessment
CADTH Therapeutic Review
 Canadian Agency for Drugs and Technologies in Health Agence canadienne des médicaments et des technologies de la santé CADTH Therapeutic Review August 2012 Volume 1, Issue 1A Antithrombotic Therapy for
Canadian Agency for Drugs and Technologies in Health Agence canadienne des médicaments et des technologies de la santé CADTH Therapeutic Review August 2012 Volume 1, Issue 1A Antithrombotic Therapy for
USDA Nutrition Evidence Library: Systematic Review Methodology
 USDA Nutrition Evidence Library: Systematic Review Methodology Julie E. Obbagy, PhD, RD USDA Center for Nutrition Policy & Promotion Meeting #2 October 17, 2016 The National Academies of Sciences, Engineering,
USDA Nutrition Evidence Library: Systematic Review Methodology Julie E. Obbagy, PhD, RD USDA Center for Nutrition Policy & Promotion Meeting #2 October 17, 2016 The National Academies of Sciences, Engineering,
ACR Appropriateness Criteria Rating Round Information
 Revised September 2017 ACR Appropriateness Criteria Rating Round Information Overview The purpose of the rating rounds is to systematically and transparently determine the panels recommendations while
Revised September 2017 ACR Appropriateness Criteria Rating Round Information Overview The purpose of the rating rounds is to systematically and transparently determine the panels recommendations while
American Thoracic Society Clinical Practice Guideline Development Manual. Audience: Guideline committee members
 American Thoracic Society Clinical Practice Guideline Development Manual Audience: Guideline committee members Last updated: April 17, 2018 1 TABLE OF CONTENTS 1. Guideline development committee 2. Conflict
American Thoracic Society Clinical Practice Guideline Development Manual Audience: Guideline committee members Last updated: April 17, 2018 1 TABLE OF CONTENTS 1. Guideline development committee 2. Conflict
Using the FRAX Tool. Osteoporosis Definition
 How long will your bones remain standing? Using the FRAX Tool Gary Salzman M.D. Director Banner Good Samaritan/ Hayden VAMC Internal Medicine Geriatric Fellowship Program Phoenix, Arizona Using the FRAX
How long will your bones remain standing? Using the FRAX Tool Gary Salzman M.D. Director Banner Good Samaritan/ Hayden VAMC Internal Medicine Geriatric Fellowship Program Phoenix, Arizona Using the FRAX
Osteoporosis: An Overview. Carolyn J. Crandall, MD, MS
 Osteoporosis: An Overview Carolyn J. Crandall, MD, MS Osteoporosis: An Overview Carolyn J. Crandall, MD, MS Professor of Medicine David Geffen School of Medicine at UCLA Objectives Review osteoporosis
Osteoporosis: An Overview Carolyn J. Crandall, MD, MS Osteoporosis: An Overview Carolyn J. Crandall, MD, MS Professor of Medicine David Geffen School of Medicine at UCLA Objectives Review osteoporosis
NICE SCOOP OF THE DAY FRAX with NOGG. Eugene McCloskey Professor of Adult Bone Diseases University of Sheffield
 NICE SCOOP OF THE DAY FRAX with NOGG Eugene McCloskey Professor of Adult Bone Diseases University of Sheffield Disclosures Consultant/Advisor/Speaker for: o ActiveSignal, Amgen, AstraZeneca, Consilient
NICE SCOOP OF THE DAY FRAX with NOGG Eugene McCloskey Professor of Adult Bone Diseases University of Sheffield Disclosures Consultant/Advisor/Speaker for: o ActiveSignal, Amgen, AstraZeneca, Consilient
MINDFULNESS-BASED INTERVENTIONS IN EPILEPSY
 03 March 2016; v.1 MINDFULNESS-BASED INTERVENTIONS IN EPILEPSY AIM This review aimed to evaluate the effectiveness of mindfulness as a therapeutic intervention for people with epilepsy. METHODS Criteria
03 March 2016; v.1 MINDFULNESS-BASED INTERVENTIONS IN EPILEPSY AIM This review aimed to evaluate the effectiveness of mindfulness as a therapeutic intervention for people with epilepsy. METHODS Criteria
Results. NeuRA Hypnosis June 2016
 Introduction may be experienced as an altered state of consciousness or as a state of relaxation. There is no agreed framework for administering hypnosis, but the procedure often involves induction (such
Introduction may be experienced as an altered state of consciousness or as a state of relaxation. There is no agreed framework for administering hypnosis, but the procedure often involves induction (such
Learning Objectives. Controversies in Osteoporosis Prevention and Management. Definition. Presenter Disclosure Information.
 4 4:45 pm Controversies in Osteoporosis Prevention and Management SPEAKER Carolyn Crandall, MD, MS Presenter Disclosure Information The following relationships exist related to this presentation: Carolyn
4 4:45 pm Controversies in Osteoporosis Prevention and Management SPEAKER Carolyn Crandall, MD, MS Presenter Disclosure Information The following relationships exist related to this presentation: Carolyn
SCHEDULE 2 THE SERVICES. A. Service Specifications
 SCHEDULE 2 THE SERVICES A. Service Specifications Service Specification No. 04/MSKT/0013 Service PAN DORSET FRACTURE LIAISON SERVICE Commissioner Lead CCP for Musculoskeletal & Trauma Provider Lead Deputy
SCHEDULE 2 THE SERVICES A. Service Specifications Service Specification No. 04/MSKT/0013 Service PAN DORSET FRACTURE LIAISON SERVICE Commissioner Lead CCP for Musculoskeletal & Trauma Provider Lead Deputy
Osteoporosis: fragility fracture risk. Costing report. Implementing NICE guidance
 Osteoporosis: fragility fracture risk Costing report Implementing NICE guidance August 2012 NICE clinical guideline 146 1 of 15 This costing report accompanies the clinical guideline: Osteoporosis: assessing
Osteoporosis: fragility fracture risk Costing report Implementing NICE guidance August 2012 NICE clinical guideline 146 1 of 15 This costing report accompanies the clinical guideline: Osteoporosis: assessing
Osteoporosis challenges
 Osteoporosis challenges Osteoporosis challenges Who should have a fracture risk assessment? Who to treat? Drugs, holidays and unusual adverse effects Fracture liaison service? The size of the problem 1
Osteoporosis challenges Osteoporosis challenges Who should have a fracture risk assessment? Who to treat? Drugs, holidays and unusual adverse effects Fracture liaison service? The size of the problem 1
North Central London Joint Formulary Committee
 North Central London Joint Formulary Committee Calcium + vitamin D supplementation for the prevention of osteoporotic fragility fractures Disclaimer This guideline is registered at North Central London
North Central London Joint Formulary Committee Calcium + vitamin D supplementation for the prevention of osteoporotic fragility fractures Disclaimer This guideline is registered at North Central London
Analyses of cost-effective BMD scanning and treatment strategies for generic alendronate, and the costeffectiveness
 Analyses of cost-effective BMD scanning and treatment strategies for generic alendronate, and the costeffectiveness of risedronate and strontium ranelate in those people who would be treated with generic
Analyses of cost-effective BMD scanning and treatment strategies for generic alendronate, and the costeffectiveness of risedronate and strontium ranelate in those people who would be treated with generic
Thank you for agreeing to give us a statement on your organisation s view of the technology and the way it should be used in the NHS.
 Appendix I - Thank you for agreeing to give us a statement on your organisation s view of the technology and the way it should be used in the NHS. Healthcare professionals can provide a unique perspective
Appendix I - Thank you for agreeing to give us a statement on your organisation s view of the technology and the way it should be used in the NHS. Healthcare professionals can provide a unique perspective
The MASCC Guidelines Policy
 The MASCC Guidelines Policy Recommendations for MASCC Guideline Construction and the Endorsement of Externally Generated Guidelines Preamble MASCC recognizes that providing supportive care facilitates
The MASCC Guidelines Policy Recommendations for MASCC Guideline Construction and the Endorsement of Externally Generated Guidelines Preamble MASCC recognizes that providing supportive care facilitates
Guideline Development At WHO
 Guideline Development At WHO Susan L. Norris, MD MPH MSc Guidelines Review Committee Secretariat September 24, 2015 1 Questions Who has ever used a clinical practice guideline? Who regularly uses clinical
Guideline Development At WHO Susan L. Norris, MD MPH MSc Guidelines Review Committee Secretariat September 24, 2015 1 Questions Who has ever used a clinical practice guideline? Who regularly uses clinical
May Professor Matt Stevenson School of Health and Related Research, University of Sheffield
 ASSESSING THE FEASIBILITY OF TRANSFORMING THE RECOMMENDATIONS IN TA160, TA161 AND TA204 INTO ABSOLUTE 10-YEAR RISK OF FRACTURE A REPORT PRODUCED BY THE DECISION SUPPORT UNIT IN THE CONTEXT OF THE REVIEW
ASSESSING THE FEASIBILITY OF TRANSFORMING THE RECOMMENDATIONS IN TA160, TA161 AND TA204 INTO ABSOLUTE 10-YEAR RISK OF FRACTURE A REPORT PRODUCED BY THE DECISION SUPPORT UNIT IN THE CONTEXT OF THE REVIEW
Post-traumatic stress disorder
 National Institute for Health and Care Excellence Final Post-traumatic stress disorder Methods NICE guideline NG116 Supplement 1 December 2018 Final These evidence reviews were developed by the National
National Institute for Health and Care Excellence Final Post-traumatic stress disorder Methods NICE guideline NG116 Supplement 1 December 2018 Final These evidence reviews were developed by the National
DEXA SCAN POLICY CRITERIA BASED ACCESS
 DEXA SCAN POLICY Version: 1718.v2 Recommendation by: Somerset CCG Clinical Commissioning Policy Forum (CCPF) Date Ratified: 01 November 2017 Name of Originator/Author: Approved by Responsible Committee/Individual:
DEXA SCAN POLICY Version: 1718.v2 Recommendation by: Somerset CCG Clinical Commissioning Policy Forum (CCPF) Date Ratified: 01 November 2017 Name of Originator/Author: Approved by Responsible Committee/Individual:
The 2017 CMS Merit-based Incentive Payment System includes 2 inflammatory bowel disease (IBD) measures.
 The 2017 CMS Merit-based Incentive Payment System includes 2 inflammatory bowel disease (IBD) measures. Measure Specifications for Registry Reporting MIPS #271: Inflammatory Bowel Disease (IBD): Preventive
The 2017 CMS Merit-based Incentive Payment System includes 2 inflammatory bowel disease (IBD) measures. Measure Specifications for Registry Reporting MIPS #271: Inflammatory Bowel Disease (IBD): Preventive
Primary and secondary prevention of cardiovascular disease: antithrombotic therapy and prevention of thrombosis, 9th ed: American College
 General Guideline Title Primary and secondary prevention of cardiovascular disease: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians evidence-based clinical
General Guideline Title Primary and secondary prevention of cardiovascular disease: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians evidence-based clinical
FRAX Based Lebanese Osteoporosis Guidelines Second Update for Lebanese Guidelines for Osteoporosis Assessment and Treatment
 These guidelines are endorsed by the following Lebanese Scientific Societies and Associations: Lebanese Society of Endocrinology Diabetes and Lipids, Lebanese Society of Rheumatology, Lebanese Society
These guidelines are endorsed by the following Lebanese Scientific Societies and Associations: Lebanese Society of Endocrinology Diabetes and Lipids, Lebanese Society of Rheumatology, Lebanese Society
Osteoporosis. Overview
 v2 Osteoporosis Overview Osteoporosis is defined as compromised bone strength that increases risk of fracture (NIH Consensus Conference, 2000). Bone strength is characterized by bone mineral density (BMD)
v2 Osteoporosis Overview Osteoporosis is defined as compromised bone strength that increases risk of fracture (NIH Consensus Conference, 2000). Bone strength is characterized by bone mineral density (BMD)
The QUOROM Statement: revised recommendations for improving the quality of reports of systematic reviews
 The QUOROM Statement: revised recommendations for improving the quality of reports of systematic reviews David Moher 1, Alessandro Liberati 2, Douglas G Altman 3, Jennifer Tetzlaff 1 for the QUOROM Group
The QUOROM Statement: revised recommendations for improving the quality of reports of systematic reviews David Moher 1, Alessandro Liberati 2, Douglas G Altman 3, Jennifer Tetzlaff 1 for the QUOROM Group
Osteoporosis Screening and Treatment in Type 2 Diabetes
 Osteoporosis Screening and Treatment in Type 2 Diabetes Ann Schwartz, PhD! Dept. of Epidemiology and Biostatistics! University of California San Francisco! October 2011! Presenter Disclosure Information
Osteoporosis Screening and Treatment in Type 2 Diabetes Ann Schwartz, PhD! Dept. of Epidemiology and Biostatistics! University of California San Francisco! October 2011! Presenter Disclosure Information
Background: Traditional rehabilitation after total joint replacement aims to improve the muscle strength of lower limbs,
 REVIEWING THE EFFECTIVENESS OF BALANCE TRAINING BEFORE AND AFTER TOTAL KNEE AND TOTAL HIP REPLACEMENT: PROTOCOL FOR A SYSTEMATIC RE- VIEW AND META-ANALYSIS Background: Traditional rehabilitation after
REVIEWING THE EFFECTIVENESS OF BALANCE TRAINING BEFORE AND AFTER TOTAL KNEE AND TOTAL HIP REPLACEMENT: PROTOCOL FOR A SYSTEMATIC RE- VIEW AND META-ANALYSIS Background: Traditional rehabilitation after
Download slides:
 Download slides: https://www.tinyurl.com/m67zcnn https://tinyurl.com/kazchbn OSTEOPOROSIS REVIEW AND UPDATE Boca Raton Regional Hospital Internal Medicine Conference 2017 Benjamin Wang, M.D., FRCPC Division
Download slides: https://www.tinyurl.com/m67zcnn https://tinyurl.com/kazchbn OSTEOPOROSIS REVIEW AND UPDATE Boca Raton Regional Hospital Internal Medicine Conference 2017 Benjamin Wang, M.D., FRCPC Division
Alcohol interventions in secondary and further education
 National Institute for Health and Care Excellence Guideline version (Draft for Consultation) Alcohol interventions in secondary and further education NICE guideline: methods NICE guideline Methods
National Institute for Health and Care Excellence Guideline version (Draft for Consultation) Alcohol interventions in secondary and further education NICE guideline: methods NICE guideline Methods
Lorne A. Becker MD Emeritus Professor SUNY Upstate Medical University. Co-Chair, Cochrane Collaboration Steering Group
 Lorne A. Becker MD Emeritus Professor SUNY Upstate Medical University Co-Chair, Cochrane Collaboration Steering Group Many different types Vary in Complexity Trustworthiness of conclusions http://www.ahrq.gov/clinic/uspstf/uspscopd.htm
Lorne A. Becker MD Emeritus Professor SUNY Upstate Medical University Co-Chair, Cochrane Collaboration Steering Group Many different types Vary in Complexity Trustworthiness of conclusions http://www.ahrq.gov/clinic/uspstf/uspscopd.htm
nogg Guideline for the diagnosis and management of osteoporosis in postmenopausal women and men from the age of 50 years in the UK
 nogg NATIONAL OSTEOPOROSIS GUIDELINE GROUP Guideline for the diagnosis and management of osteoporosis in postmenopausal women and men from the age of 50 years in the UK Produced by J Compston, A Cooper,
nogg NATIONAL OSTEOPOROSIS GUIDELINE GROUP Guideline for the diagnosis and management of osteoporosis in postmenopausal women and men from the age of 50 years in the UK Produced by J Compston, A Cooper,
Modeling the annual costs of postmenopausal prevention therapy: raloxifene, alendronate, or estrogen-progestin therapy Mullins C D, Ohsfeldt R L
 Modeling the annual costs of postmenopausal prevention therapy: raloxifene, alendronate, or estrogen-progestin therapy Mullins C D, Ohsfeldt R L Record Status This is a critical abstract of an economic
Modeling the annual costs of postmenopausal prevention therapy: raloxifene, alendronate, or estrogen-progestin therapy Mullins C D, Ohsfeldt R L Record Status This is a critical abstract of an economic
PGY1 Learning activities-ebcp Scripts
 PGY1 Learning activities-ebcp Scripts Interns will meet in small groups, once per month, to discuss EBCP scripts designed to promote understanding of basic EBCP concepts. EBCP scripts (see example below)
PGY1 Learning activities-ebcp Scripts Interns will meet in small groups, once per month, to discuss EBCP scripts designed to promote understanding of basic EBCP concepts. EBCP scripts (see example below)
Horizon Scanning Centre March Denosumab for glucocorticoidinduced SUMMARY NIHR HSC ID: 6329
 Horizon Scanning Centre March 2014 Denosumab for glucocorticoidinduced osteoporosis SUMMARY NIHR HSC ID: 6329 This briefing is based on information available at the time of research and a limited literature
Horizon Scanning Centre March 2014 Denosumab for glucocorticoidinduced osteoporosis SUMMARY NIHR HSC ID: 6329 This briefing is based on information available at the time of research and a limited literature
Advanced medicine conference. Monday 20 Tuesday 21 June 2016
 Advanced medicine conference Monday 20 Tuesday 21 June 2016 Osteoporosis: recent advances in risk assessment and management Juliet Compston Emeritus Professor of Bone Medicine Cambridge Biomedical Campus
Advanced medicine conference Monday 20 Tuesday 21 June 2016 Osteoporosis: recent advances in risk assessment and management Juliet Compston Emeritus Professor of Bone Medicine Cambridge Biomedical Campus
What is the Cochrane Collaboration? What is a systematic review?
 1 What is the Cochrane Collaboration? What is a systematic review? Archie Cochrane (1909-1988) It is surely a great criticism of our profession that we have not organised a critical summary, by specialty
1 What is the Cochrane Collaboration? What is a systematic review? Archie Cochrane (1909-1988) It is surely a great criticism of our profession that we have not organised a critical summary, by specialty
Prophylactic treatment for osteoporosis: Student EBM Presentation
 Prophylactic treatment for osteoporosis: Student EBM Presentation Callum Harris & Ealish Swift University of Oxford October 2015 Example patient JS is a 67 year old lady who needs to start taking long-term
Prophylactic treatment for osteoporosis: Student EBM Presentation Callum Harris & Ealish Swift University of Oxford October 2015 Example patient JS is a 67 year old lady who needs to start taking long-term
Uses and misuses of the STROBE statement: bibliographic study
 Uses and misuses of the STROBE statement: bibliographic study Bruno R. da Costa 1, Myriam Cevallos 1, 2, Douglas G. Altman 3, Anne W.S. Rutjes 1, Matthias Egger 1 1. Institute of Social & Preventive Medicine
Uses and misuses of the STROBE statement: bibliographic study Bruno R. da Costa 1, Myriam Cevallos 1, 2, Douglas G. Altman 3, Anne W.S. Rutjes 1, Matthias Egger 1 1. Institute of Social & Preventive Medicine
Cochrane Breast Cancer Group
 Cochrane Breast Cancer Group Version and date: V3.2, September 2013 Intervention Cochrane Protocol checklist for authors This checklist is designed to help you (the authors) complete your Cochrane Protocol.
Cochrane Breast Cancer Group Version and date: V3.2, September 2013 Intervention Cochrane Protocol checklist for authors This checklist is designed to help you (the authors) complete your Cochrane Protocol.
Results. NeuRA Treatments for internalised stigma December 2017
 Introduction Internalised stigma occurs within an individual, such that a person s attitude may reinforce a negative self-perception of mental disorders, resulting in reduced sense of selfworth, anticipation
Introduction Internalised stigma occurs within an individual, such that a person s attitude may reinforce a negative self-perception of mental disorders, resulting in reduced sense of selfworth, anticipation
Webinar 3 Systematic Literature Review: What you Need to Know
 Webinar 3 Systematic Literature Review: What you Need to Know Camille Kolotylo RN, PhD Andrea Baumann RN, PhD Nursing Health Services Research Unit (NHSRU) McMaster University Date: Thursday May 29, 2014
Webinar 3 Systematic Literature Review: What you Need to Know Camille Kolotylo RN, PhD Andrea Baumann RN, PhD Nursing Health Services Research Unit (NHSRU) McMaster University Date: Thursday May 29, 2014
Technology appraisal guidance Published: 9 August 2017 nice.org.uk/guidance/ta464
 Bisphosphonates for treating osteoporosis Technology appraisal guidance Published: 9 August 2017 nice.org.uk/guidance/ta464 NICE 2017. All rights reserved. Subject to Notice of rights (https://www.nice.org.uk/terms-and-conditions#notice-ofrights).
Bisphosphonates for treating osteoporosis Technology appraisal guidance Published: 9 August 2017 nice.org.uk/guidance/ta464 NICE 2017. All rights reserved. Subject to Notice of rights (https://www.nice.org.uk/terms-and-conditions#notice-ofrights).
Committee of Senior Representatives Tenth Meeting Oslo, Norway 11 December 2006
 Committee of Senior Representatives Tenth Meeting Oslo, Norway 11 December 2006 Reference CSR 10/7.1/1 Title Proposed Terms of Reference for the EG on HIV/AIDS Submitted by Secretariat Summary / Note As
Committee of Senior Representatives Tenth Meeting Oslo, Norway 11 December 2006 Reference CSR 10/7.1/1 Title Proposed Terms of Reference for the EG on HIV/AIDS Submitted by Secretariat Summary / Note As
Component of CPG development ILAE Recommendation Document Identifying topic and developing clinical. S3 research question
 S1: Summary of recommendations for developing Clinical Practice Guidelines Minimum CPG development requirements (where resources are more limited) are bolded. When full resources are available, every recommendation
S1: Summary of recommendations for developing Clinical Practice Guidelines Minimum CPG development requirements (where resources are more limited) are bolded. When full resources are available, every recommendation
TITLE: Acetylsalicylic Acid for Venous Thromboembolism Prophylaxis: A Review of Clinical Evidence, Benefits and Harms
 TITLE: Acetylsalicylic Acid for Venous Thromboembolism Prophylaxis: A Review of Clinical Evidence, Benefits and Harms DATE: 23 August 2011 CONTEXT AND POLICY ISSUES: Thromboembolism occurs when a blood
TITLE: Acetylsalicylic Acid for Venous Thromboembolism Prophylaxis: A Review of Clinical Evidence, Benefits and Harms DATE: 23 August 2011 CONTEXT AND POLICY ISSUES: Thromboembolism occurs when a blood
Page 1
 Osteoporosis Osteoporosis is a condition characterised by weakened bones that fracture easily. After menopause many women are at risk of developing osteoporosis. Peak bone mass is usually reached during
Osteoporosis Osteoporosis is a condition characterised by weakened bones that fracture easily. After menopause many women are at risk of developing osteoporosis. Peak bone mass is usually reached during
Practical Use of Decision Aids in Primary Care. Michael Soung, MD, FACP Virginia Mason Medical Center General Internal Medicine
 Practical Use of Decision Aids in Primary Care Michael Soung, MD, FACP Virginia Mason Medical Center General Internal Medicine Question #1 40 yo man with total cholesterol 170, HDL 50, LDL 90. African
Practical Use of Decision Aids in Primary Care Michael Soung, MD, FACP Virginia Mason Medical Center General Internal Medicine Question #1 40 yo man with total cholesterol 170, HDL 50, LDL 90. African
Implementing scientific evidence into clinical practice guidelines
 Evidence Based Dentistry Implementing scientific evidence into clinical practice guidelines Asbjørn Jokstad University of Oslo, Norway 15/07/2004 1 PRACTICE GUIDELINES IN DENTISTRY (Medline) 2100 1945
Evidence Based Dentistry Implementing scientific evidence into clinical practice guidelines Asbjørn Jokstad University of Oslo, Norway 15/07/2004 1 PRACTICE GUIDELINES IN DENTISTRY (Medline) 2100 1945
The health economics of calcium and vitamin D3 for the prevention of osteoporotic hip fractures in Sweden Willis M S
 The health economics of calcium and vitamin D3 for the prevention of osteoporotic hip fractures in Sweden Willis M S Record Status This is a critical abstract of an economic evaluation that meets the criteria
The health economics of calcium and vitamin D3 for the prevention of osteoporotic hip fractures in Sweden Willis M S Record Status This is a critical abstract of an economic evaluation that meets the criteria
Case Finding and Risk Assessment for Osteoporosis
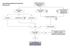 Case Finding and Risk Assessment for Osteoporosis Patient may present as a fragility fracture or risk fracture Fragility fracture age 50 Clinical risk factors aged 50 Very strong clinical risk factors
Case Finding and Risk Assessment for Osteoporosis Patient may present as a fragility fracture or risk fracture Fragility fracture age 50 Clinical risk factors aged 50 Very strong clinical risk factors
COPD-Related Musculoskeletal Disease. Jessica Bon Field, MD, MS 2017 Update in Internal Medicine October 20, 2017
 COPD-Related Musculoskeletal Disease Jessica Bon Field, MD, MS 2017 Update in Internal Medicine October 20, 2017 A 60-year old man with COPD comes into your office for a routine office visit. He is a former
COPD-Related Musculoskeletal Disease Jessica Bon Field, MD, MS 2017 Update in Internal Medicine October 20, 2017 A 60-year old man with COPD comes into your office for a routine office visit. He is a former
Clinical Evidence. A BMJ database of the very best evidence for effective health care
 Clinical Evidence A BMJ database of the very best evidence for effective health care Myleen Oosthuizen Academic Information Service School for Health Sciences What is Clinical Evidence? An international
Clinical Evidence A BMJ database of the very best evidence for effective health care Myleen Oosthuizen Academic Information Service School for Health Sciences What is Clinical Evidence? An international
Treatments for Osteoporosis Expected Benefits, Potential Harms and Drug Holidays. Suzanne Morin MD FRCP FACP McGill University May 2014
 Treatments for Osteoporosis Expected Benefits, Potential Harms and Drug Holidays Suzanne Morin MD FRCP FACP McGill University May 2014 Learning Objectives Overview of osteoporosis management Outline efficacy
Treatments for Osteoporosis Expected Benefits, Potential Harms and Drug Holidays Suzanne Morin MD FRCP FACP McGill University May 2014 Learning Objectives Overview of osteoporosis management Outline efficacy
Osteoporosis - New Guidelines. Michelle Glass B.Sc. (Pharm) June 15, 2011
 Osteoporosis - New Guidelines Michelle Glass B.Sc. (Pharm) June 15, 2011 Outline What is Osteoporosis? Who is at risk? What treatments are available? Role of the Pharmacy technician Definition of Osteoporosis
Osteoporosis - New Guidelines Michelle Glass B.Sc. (Pharm) June 15, 2011 Outline What is Osteoporosis? Who is at risk? What treatments are available? Role of the Pharmacy technician Definition of Osteoporosis
Traumatic brain injury
 Introduction It is well established that traumatic brain injury increases the risk for a wide range of neuropsychiatric disturbances, however there is little consensus on whether it is a risk factor for
Introduction It is well established that traumatic brain injury increases the risk for a wide range of neuropsychiatric disturbances, however there is little consensus on whether it is a risk factor for
Initial Pathway for DEXA Referral and Treatment for Fracture Risk Reduction in Postmenopausal Women and Men Age 50 or Above
 Initial Pathway for DEXA Referral and Treatment for Fracture Risk Reduction in Postmenopausal Women and Men Age 50 or Above 2 or > vertebral fractures Low trauma fracture In past 5 years Risk Factors (table1)
Initial Pathway for DEXA Referral and Treatment for Fracture Risk Reduction in Postmenopausal Women and Men Age 50 or Above 2 or > vertebral fractures Low trauma fracture In past 5 years Risk Factors (table1)
GRADE. Grading of Recommendations Assessment, Development and Evaluation. British Association of Dermatologists April 2014
 GRADE Grading of Recommendations Assessment, Development and Evaluation British Association of Dermatologists April 2014 Previous grading system Level of evidence Strength of recommendation Level of evidence
GRADE Grading of Recommendations Assessment, Development and Evaluation British Association of Dermatologists April 2014 Previous grading system Level of evidence Strength of recommendation Level of evidence
SUPPLEMENTARY DATA. Supplementary Figure S1. Search terms*
 Supplementary Figure S1. Search terms* *mh = exploded MeSH: Medical subject heading (Medline medical index term); tw = text word; pt = publication type; the asterisk (*) stands for any character(s) #1:
Supplementary Figure S1. Search terms* *mh = exploded MeSH: Medical subject heading (Medline medical index term); tw = text word; pt = publication type; the asterisk (*) stands for any character(s) #1:
Osteoporosis Clinical Guideline. Rheumatology January 2017
 Osteoporosis Clinical Guideline Rheumatology January 2017 Introduction Osteoporosis is a condition of low bone mass leading to an increased risk of low trauma fractures. The prevalence of osteoporosis
Osteoporosis Clinical Guideline Rheumatology January 2017 Introduction Osteoporosis is a condition of low bone mass leading to an increased risk of low trauma fractures. The prevalence of osteoporosis
FRAX, NICE and NOGG. Eugene McCloskey Professor of Adult Bone Diseases University of Sheffield
 FRAX, NICE and NOGG Eugene McCloskey Professor of Adult Bone Diseases University of Sheffield Disclosures Research funding and/or honoraria and/or advisory boards for: o ActiveSignal, Amgen, Bayer, Boehringer
FRAX, NICE and NOGG Eugene McCloskey Professor of Adult Bone Diseases University of Sheffield Disclosures Research funding and/or honoraria and/or advisory boards for: o ActiveSignal, Amgen, Bayer, Boehringer
Standards for the reporting of new Cochrane Intervention Reviews
 Methodological Expectations of Cochrane Intervention Reviews (MECIR) Standards for the reporting of new Cochrane Intervention Reviews 24 September 2012 Preface The standards below summarize proposed attributes
Methodological Expectations of Cochrane Intervention Reviews (MECIR) Standards for the reporting of new Cochrane Intervention Reviews 24 September 2012 Preface The standards below summarize proposed attributes
Beyond the T-score: New Thinking in Osteoporosis 2
 Transcript Details This is a transcript of an educational program accessible on the ReachMD network. Details about the program and additional media formats for the program are accessible by visiting: https://reachmd.com/programs/clinicians-roundtable/beyond-the-t-score-new-thinking-in-osteoporosis-
Transcript Details This is a transcript of an educational program accessible on the ReachMD network. Details about the program and additional media formats for the program are accessible by visiting: https://reachmd.com/programs/clinicians-roundtable/beyond-the-t-score-new-thinking-in-osteoporosis-
Osteoporosis/Fracture Prevention Clinical Practice Guidelines
 NATIONAL CLINICAL PRACTICE GUIDELINES Osteoporosis/Fracture Prevention Clinical Practice Guidelines Reviewed/Approved by the National Guideline Directors September 2017 Next Review/Approval: September
NATIONAL CLINICAL PRACTICE GUIDELINES Osteoporosis/Fracture Prevention Clinical Practice Guidelines Reviewed/Approved by the National Guideline Directors September 2017 Next Review/Approval: September
CIHRT Exhibit P-2592 Page 1 APPENDIX. ADAPTE Process for the Treatment of In situ Breast Carcinoma. Eastern Health Breast Disease Site Group
 CIHRT Exhibit P-2592 Page 1 APPENDIX ADAPTE Process for the Treatment of In situ Breast Carcinoma Eastern Health Breast Disease Site Group Phase I - Setup The Breast Disease Site Group was formed in the
CIHRT Exhibit P-2592 Page 1 APPENDIX ADAPTE Process for the Treatment of In situ Breast Carcinoma Eastern Health Breast Disease Site Group Phase I - Setup The Breast Disease Site Group was formed in the
denosumab (Prolia ) Policy # Original Effective Date: 07/21/2011 Current Effective Date: 04/19/2017
 Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Based on review of available data, the Company may consider the use of denosumab (Prolia) for the
 Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
[If no, skip to question 10.] Y N. 2. Does the member have a diagnosis of Paget s disease of bone? Y N. [If no, skip to question 4.
![[If no, skip to question 10.] Y N. 2. Does the member have a diagnosis of Paget s disease of bone? Y N. [If no, skip to question 4. [If no, skip to question 10.] Y N. 2. Does the member have a diagnosis of Paget s disease of bone? Y N. [If no, skip to question 4.](/thumbs/86/94943423.jpg) Pharmacy Prior Authorization AETA BETTER HEALTH EW JERSE (MEDICAID) Zoledronic Acid (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information,
Pharmacy Prior Authorization AETA BETTER HEALTH EW JERSE (MEDICAID) Zoledronic Acid (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information,
Acute Dialysis Quality Inititative 8 th Scientific Meeting: Hepatorenal Syndrome March 16 th -19 th, 2010
 Acute Dialysis Quality Inititative 8 th Scientific Meeting: Hepatorenal Syndrome March 16 th -19 th, 2010 Acute Dialysis Quality Initiative (ADQI) started in response to concerns about the quality of care
Acute Dialysis Quality Inititative 8 th Scientific Meeting: Hepatorenal Syndrome March 16 th -19 th, 2010 Acute Dialysis Quality Initiative (ADQI) started in response to concerns about the quality of care
Problem solving therapy
 Introduction People with severe mental illnesses such as schizophrenia may show impairments in problem-solving ability. Remediation interventions such as problem solving skills training can help people
Introduction People with severe mental illnesses such as schizophrenia may show impairments in problem-solving ability. Remediation interventions such as problem solving skills training can help people
Method. NeuRA Biofeedback May 2016
 Introduction is a technique in which information about the person s body is fed back to the person so that they may be trained to alter the body s conditions. Physical therapists use biofeedback to help
Introduction is a technique in which information about the person s body is fed back to the person so that they may be trained to alter the body s conditions. Physical therapists use biofeedback to help
3. Has bone specific alkaline phosphatase level increased OR does the member have symptoms related to active Paget s?
 Pharmacy Prior Authorization AETA BETTER HEALTH VIRGIIA CCC PLUS and MEDALLIO/FAMIS 4.0 Zoledronic Acid (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review
Pharmacy Prior Authorization AETA BETTER HEALTH VIRGIIA CCC PLUS and MEDALLIO/FAMIS 4.0 Zoledronic Acid (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review
Osteoporosis: How to Manage Long- Term Use of Bisphosphonates AKA Now What? David E Feinstein, DO, CCD November 15 th, 2017
 Osteoporosis: How to Manage Long- Term Use of Bisphosphonates AKA Now What? David E Feinstein, DO, CCD November 15 th, 2017 Introduction A fracture due to OP occurs every 3 seconds around the world. 1
Osteoporosis: How to Manage Long- Term Use of Bisphosphonates AKA Now What? David E Feinstein, DO, CCD November 15 th, 2017 Introduction A fracture due to OP occurs every 3 seconds around the world. 1
