Decision to amend access criteria for some vaccines
|
|
|
- Kelly Crawford
- 5 years ago
- Views:
Transcription
1 3 July 2015 Decision to amend access criteria for some vaccines PHARMAC is pleased to announce the following changes to the funding access criteria for a number of vaccines. The changes in criteria follow consideration by PHARMAC of recommendations made by the Immunisation Subcommittee of PTAC during its September 2014 and February 2015 meetings. PHARMAC did not consult on these changes as they reflect current practice and/or aim to clarify the intent current funding criteria. In summary the main effects of the decision area that from 1 July 2015: Influenza vaccine access will be widened to include vaccination of people with cochlea implants, errors of metabolism at risk of major metabolic decomposition, Down syndrome and for people who are pre and post splenectomy. Diphtheria tetanus and pertussis vaccine, hexavalent vaccine (DTaP-IPV-HepB/Hib), haemophilus influenza type B and the meningococcal vaccines (monovalent and quadrivalent) clarification of the access criteria for immunosuppressed patients. Hepatitis B vaccine access will be widened to include vaccination following nonconsensual sexual intercourse or needle stick injury. Pneumococcal (PCV13) vaccine access will be clarified to include patients with complement deficiency (acquired or inherited), primary immunodeficiency and people with cochlear implants. Human papillomavirus (6, 11, 16 and 18) vaccine (HPV) and measles, mumps and rubella vaccines - minor changes to the access criteria including an additional dose being funded for patients under 26 years of age post chemotherapy. Details of the decision are as follows: Details of the decision Influenza vaccine will remain listed in Section I of the Pharmaceutical Schedule from 1 July 2015 with changes to the funding criteria as follows (deletions in strike through, additions in bold): A) is available each year for patients who meet the following criteria, as set by PHARMAC: a) all people 65 years of age and over; or b) people under 65 years of age who: i) have any of the following cardiovascular diseases: a) ischaemic heart disease, or b) congestive heart disease failure, or c) rheumatic heart disease, or d) congenital heart disease, or e) cerebo-vascular disease; or ii) have either of the following chronic respiratory diseases: a) asthma, if on a regular preventative therapy, or b) other chronic respiratory disease with impaired lung function; or iii) have diabetes; or iv) have chronic renal disease; or A Page 1 of 5
2 v) have any cancer, excluding basal and squamous skin cancers if not invasive; or vi) have any of the following other conditions: a) autoimmune disease, or b) immune suppression or immune deficiency, or c) HIV, or d) transplant recipients, or e) neuromuscular and CNS diseases/disorders, or f) haemoglobinopathies, or g) are children on long term aspirin, or h) have a cochlear implant, or (i) errors of metabolism at risk of major metabolic decomposition, or (j) pre and post splenectomy, or (k) down syndrome; or vii) are pregnant; or c) children aged four years and under who have been hospitalised for respiratory illness or have a history of significant respiratory illness. Unless meeting the criteria set out above, the following conditions are excluded from funding: a) asthma not requiring regular preventative therapy. b) hypertension and/or dyslipidaemia without evidence of end-organ disease. B) Doctors are the only Contractors entitled to claim payment from the Funder for the supply of influenza vaccine to patients eligible under the above criteria for subsidised immunisation and they may only do so in respect of the influenza vaccine listed in the Pharmaceutical Schedule. C) Individual DHBs may fund patients over and above the above criteria. The claiming process for these additional patients should be determined between the DHB and Contractor, or D) Stock of the seasonal influenza vaccine is typically available from February until late July with suppliers being required to ensure supply until at least 30 June. Exact start and end dates for each season will be notified each year. influenza vaccine will remain listed in Part II of Section H of the Pharmaceutical Schedule from 1 July 2015 with changes to the restriction as follows (deletions in strike through, additions in bold): 1 All people 65 years of age and over; or 2 People under 65 years of age who: 2.1 Have any of the following cardiovascular diseases: Ischaemic heart disease; or Congestive heart disease failure; or Rheumatic heart disease; or Congenital heart disease; or Cerebro-vascular disease; or 2.2 Have any of the following chronic respiratory diseases: Asthma, if on a regular preventative therapy; or Other chronic respiratory disease with impaired lung function; or 2.3 Have diabetes; or 2.4 Have chronic renal disease; or 2.5 Have any cancer, excluding basal and squamous skin cancers if not invasive; or 2.6 Have any of the following other conditions: Autoimmune disease; or Immune suppression or immune deficiency; or HIV; or Transplant recipients; or Neuromuscular and CNS diseases/disorders; or Haemoglobinopathies; or Are children on long term aspirin; or Have a cochlear implant, or Errors of metabolism at risk of major metabolic decomposition, or Pre and post splenectomy, or Down syndrome; or 2.7 Are pregnant, or 2.8 Are children aged four and under who have been hospitalised for respiratory illness or have a history of significant respiratory illness; or 3 Patients who are compulsorily detained long-term in a forensic unit within a DHB hospital in the 2015 season. Note: The following conditions are excluded from funding: asthma not requiring regular preventative therapy; and A Page 2 of 5
3 hypertension and/or dyslipidaemia without evidence of end-organ disease. Diphtheria, tetanus and pertussis vaccine (Boostrix) will remain listed in Section I and Part II of Section H of the Pharmaceutical Schedule from 1 July Funded for any of the following criteria: 1) A single vaccine for pregnant woman between gestational weeks 28 and 38 during epidemics; or 2) A course of up to four vaccines is funded for children from age 7 up to the age of 18 inclusive to complete full primary immunisation; or 3) A course of up to four vaccines is funded for children from age 7 to 17 years inclusive for reimmunisation following immunosuppression. 3) An additional four doses (as appropriate) are funded for (re-)immunisation for patients post haematopoietic stem cell transplantation or chemotherapy; pre or post splenectomy; pre- or post solid organ transplant, renal dialysis and other severely immunosuppressive regimens. Notes: Tdap is not registered for patients aged less than 10 years. Please refer to the Immunisation Handbook for appropriate schedule for catch up programmes. Diphtheria, tetanus, pertussis, polio, hepatitis B and haemophilus influenzae Type B vaccine (Infanrix-hexa) will remain listed in Section I and Part II of Section H of the Pharmaceutical Schedule from 1 July 2015 as follows (deletions in strike through, additions in bold): Funded for patients meeting any of the following criteria: 1) Up to four doses for children up to and under the age of 10 for primary immunisation; or 2) Up to four doses (as appropriate) for children individuals are funded for (re)immunisation for patients post HSCT, or chemotherapy; pre or post splenectomy; renal dialysis and other severely immunosuppressive regimens; or 2) An additional four doses (as appropriate) are funded for (re-)immunisation for children up to and under the age of 10 who are patients post haematopoietic stem cell transplantation, or chemotherapy; pre or post splenectomy; pre- or post solid organ transplant, renal dialysis and other severely immunosuppressive regimens; or 3) Up to five doses for children up to and under the age of 10 receiving solid organ transplantation. Note: A course of up-to four vaccines is funded for catch up programmes for children (up to and under the age of 10 years) to complete full primary immunisation. Please refer to the Immunisation Handbook for the appropriate schedule for catch up programmes. Haemophilus influenzae Type B vaccine (Act-HIB) will remain listed in Section I and Part II of Section H of the Pharmaceutical Schedule effective from 1 July One dose for patients meeting any of the following: 1) For primary vaccination in children; or 2) For revaccination of children following immunosuppression; or 3) For children aged 0 18 years with functional asplenia; or 4) For patients pre and post splenectomy; or 2) An additional dose (as appropriate) is funded for (re-)immunisation for patients post haematopoietic stem cell transplantation, or chemotherapy; pre or post splenectomy; pre- or post solid organ transplant, pre- or post cochlear implants, renal dialysis and other severely immunosuppressive regimens; or 3 5) For use in testing for primary immunodeficiency diseases, on the recommendation of an internal medicine physician or paediatrician. Hepatitis B vaccine (HBvaxPRO) inj 5 mcg per 0.5 ml vial and inj 10 mcg per 1 ml vial will remain listed in Section I and Part II of Section H of the A Page 3 of 5
4 Pharmaceutical Schedule effective from 1 July 2015 as follows (deletions in strike through, additions in bold): Funded for patients meeting any of the following criteria: 1) for household or sexual contacts of known acute hepatitis B patients or hepatitis B carriers; or 2) for children born to mothers who are hepatitis B surface antigen (HBsAg) positive; or 3) for children up to and under the age of 18 years inclusive who are considered not to have achieved a positive serology and require additional vaccination; or 4) for HIV positive patients; or 5) for hepatitis C positive patients; or 6) for patients following non-consensual sexual intercourse; or 7 6) for patients following immunosuppression; or 8 7) for transplant patients; or 9) following needle stick injury. Human papillomavirus (6, 11, 16 and 18) vaccine (Gardasil) will remain listed in Section I and Part II of Section H of the Pharmaceutical Schedule effective from 1 July Maximum of three doses for patient meeting any of the following criteria: 1) Females aged under 20 years old; or 2) Patients aged under 26 years old with confirmed HIV infection; or 3) For use in transplant (including stem cell) patients; or 4) An additional dose for patients under 26 years of age post chemotherapy. Measles, mumps and rubella vaccine (M-M-R II) will remain listed in Section I and Part II of Section H of the Pharmaceutical Schedule effective from 1 July A maximum of two doses for any patient meeting the following criteria: 1) For primary vaccination in children; or 2) For revaccination following immunosuppression; or 3) For any individual susceptible to measles, mumps or rubella; or 4) A maximum of three doses for children who have had their first dose prior to 12 months. Note: Please refer to the Immunisation Handbook for appropriate schedule for catch up programmes. Meningococcal (groups A, C, Y, W-135) conjugate vaccine (Menactra) will remain listed in Section I and Part II of Section H of the Pharmaceutical Schedule effective from 1 July 2015 as follows (deletions in strike through, additions in bold): 1) Up to three doses and a booster every five years for patients pre- and post splenectomy and for patients with functional or anatomic asplenia, HIV, complement deficiency (acquired or inherited), or pre or post solid organ transplant; or 2) One dose every five years for patients with HIV, complement deficiency (acquired or inherited), functional or anatomic asplenia or pre or post solid organ transplant; or 2 3) One dose for close contacts of meningococcal cases; or 3 4) A maximum of two doses for bone marrow transplant patients; or 4 5) A maximum of two doses for patients following immunosuppression*. Note: children under seven years of age require two doses 8 weeks apart, a booster dose three years after the primary series and then five yearly a second dose three years after the first and then five yearly. *Immunosuppression due to steroid or other immunosuppressive therapy must be for a period of greater than 28 days. Meningococcal C conjugated vaccine (Neisvac-C) will remain listed in Section I and Part II of Section H of the Pharmaceutical Schedule effective from 1 July A Page 4 of 5
5 1) Up to three doses and a booster every five years for patients pre- and post splenectomy and for patients with functional or anatomic asplenia, HIV, complement deficiency (acquired or inherited or pre or post solid organ transplant; or 2) One dose every five years for patients with HIV, complement deficiency (acquired or inherited), functional or anatomic asplenia or pre or post solid organ transplant; or 2 3) One dose for close contacts of meningococcal cases; or 3 4) A maximum of two doses for bone marrow transplant patients; or 4 5) A maximum of two doses for patients following immunosuppression*. Note: children under seven years of age require two doses 8 weeks apart, a booster dose three years after the primary series and then five yearly a second dose three years after the first and then five yearly. *Immunosuppression due to steroid or other immunosuppressive therapy must be for a period of greater than 28 days. Pneumococcal (PCV13) vaccine (Prevenar 13) will remain listed in Section I and Part II of Section H of the Pharmaceutical Schedule effective from 1 July 1) A primary course of four doses for previously unvaccinated individuals up to the age of 59 months inclusive; or 2) Up to three doses as appropriate to complete the primary course of immunisation for individuals under the age of 59 months who have received one to three doses of PCV10; or 3) One dose is funded for high risk children (over the age of 17 months and up to the age of 18) who have previously received four doses of PCV10; or 4) Up to an additional four doses (as appropriate) are funded for (re-)immunisation of patients with HIV, for patients post haematopoietic stem cell transplantationhsct, or chemotherapy; preor post splenectomy; functional asplenia, pre- or post- solid organ transplant, renal dialysis, complement deficiency (acquired or inherited), cochlear implants, primary immunodeficiency and other severely immunosuppressive regimens up to under the age of 18; or 5) For use in testing for primary immunodeficiency diseases, on the recommendation of an internal medicine physician or paediatrician. Note: please refer to the Immunisation Handbook for the appropriate schedule for catch up programmes More information If you have any questions about this decision, you can us at enquiry@pharmac.govt.nz or call our toll free number (9 am to 5 pm, Monday to Friday) on A Page 5 of 5
Changes to the National Immunisation Schedule
 17 December 2013 Changes to the National Immunisation Schedule PHARMAC is pleased to announce decisions related to the National Immunisation Schedule (NIS) that will take effect from 1 July 2014. This
17 December 2013 Changes to the National Immunisation Schedule PHARMAC is pleased to announce decisions related to the National Immunisation Schedule (NIS) that will take effect from 1 July 2014. This
Immunisation Subcommittee of PTAC Meeting held 10 February (minutes for web publishing)
 Immunisation Subcommittee of PTAC Meeting held 10 February 2014 (minutes for web publishing) Immunisation Subcommittee minutes are published in accordance with the Terms of Reference for the Pharmacology
Immunisation Subcommittee of PTAC Meeting held 10 February 2014 (minutes for web publishing) Immunisation Subcommittee minutes are published in accordance with the Terms of Reference for the Pharmacology
Immunisation Subcommittee of PTAC Meeting held 23 April (minutes for web publishing)
 Immunisation Subcommittee of PTAC Meeting held 23 April 2013 (minutes for web publishing) Immunisation Subcommittee minutes are published in accordance with the Terms of Reference for the Pharmacology
Immunisation Subcommittee of PTAC Meeting held 23 April 2013 (minutes for web publishing) Immunisation Subcommittee minutes are published in accordance with the Terms of Reference for the Pharmacology
immunisation in New Zealand
 This appendix details the history of. Section A1.1 is a brief summary of when each vaccine was introduced to the National Immunisation Schedule (the Schedule). This summary includes vaccines which were
This appendix details the history of. Section A1.1 is a brief summary of when each vaccine was introduced to the National Immunisation Schedule (the Schedule). This summary includes vaccines which were
What are the new active vaccine recommendations in the Canadian Immunization Guide?
 154 CCDR 17 April 2014 Volume 40-8 https://doi.org/10.14745/ccdr.v40i08a03 1 What are the new active vaccine recommendations in the Canadian Immunization Guide? Warshawsky B 1 and Gemmill I 2 on behalf
154 CCDR 17 April 2014 Volume 40-8 https://doi.org/10.14745/ccdr.v40i08a03 1 What are the new active vaccine recommendations in the Canadian Immunization Guide? Warshawsky B 1 and Gemmill I 2 on behalf
IMMUNISATION OF CHILDREN DURING and AFTER CANCER THERAPY
 NATIONAL GUIDELINES PAEDIATRIC ONCOLOGY AND HAEMATOLOGY For the Care of Childhood Cancer in Specialist Child Cancer and Shared Care Centres. IMMUNISATION OF CHILDREN DURING and AFTER CANCER THERAPY Date
NATIONAL GUIDELINES PAEDIATRIC ONCOLOGY AND HAEMATOLOGY For the Care of Childhood Cancer in Specialist Child Cancer and Shared Care Centres. IMMUNISATION OF CHILDREN DURING and AFTER CANCER THERAPY Date
3 rd dose. 3 rd or 4 th dose, see footnote 5. see footnote 13. for certain high-risk groups
 Figure 1. Recommended immunization schedule for persons aged 0 through 18 years 2013. (FOR THOSE WHO FALL BEHIND OR START LATE, SEE THE CATCH-UP SCHEDULE [FIGURE 2]). These recommendations must be read
Figure 1. Recommended immunization schedule for persons aged 0 through 18 years 2013. (FOR THOSE WHO FALL BEHIND OR START LATE, SEE THE CATCH-UP SCHEDULE [FIGURE 2]). These recommendations must be read
PHARMAC invites proposals for the supply of various vaccines in New Zealand.
 28 June 2013 Dear Supplier REQUEST FOR PROPOSALS SUPPLY OF VARIOUS VACCINES PHARMAC invites proposals for the supply of various vaccines in New Zealand. This request for proposals (RFP) letter incorporates
28 June 2013 Dear Supplier REQUEST FOR PROPOSALS SUPPLY OF VARIOUS VACCINES PHARMAC invites proposals for the supply of various vaccines in New Zealand. This request for proposals (RFP) letter incorporates
NOTE: The above recommendations must be read along with the footnotes of this schedule.
 Figure 1. Recommended immunization schedule for persons aged 0 through 18 years United States, 2014. (FOR THOSE WHO FALL BEHIND OR START LATE, SEE THE CATCH-UP SCHEDULE [FIGURE 2]). These recommendations
Figure 1. Recommended immunization schedule for persons aged 0 through 18 years United States, 2014. (FOR THOSE WHO FALL BEHIND OR START LATE, SEE THE CATCH-UP SCHEDULE [FIGURE 2]). These recommendations
Immunisations in Adult End stage kidney disease update 2016
 Additional Notes During work up (predialysis or dialysis) Awaiting (live donor or on deceased donor list) Post Renal Transplant on immunosuppression Vaccinate Annually: Influenza (eg Influvac, Flurix)
Additional Notes During work up (predialysis or dialysis) Awaiting (live donor or on deceased donor list) Post Renal Transplant on immunosuppression Vaccinate Annually: Influenza (eg Influvac, Flurix)
The National Immunisation Schedule Update and Current issues. Dr Brenda Corcoran National Immunisation Office.
 The National Immunisation Schedule Update and Current issues Dr Brenda Corcoran National Immunisation Office : Dates vaccines introduced into the Irish immunisation schedule Vaccine 1937-1999 Date introduced
The National Immunisation Schedule Update and Current issues Dr Brenda Corcoran National Immunisation Office : Dates vaccines introduced into the Irish immunisation schedule Vaccine 1937-1999 Date introduced
Vaccinations for Adults
 Case: Vaccinations for Adults Lisa Winston, MD University of California, San Francisco San Francisco General Hospital A 30-year old healthy woman comes for a routine visit. She is recently married and
Case: Vaccinations for Adults Lisa Winston, MD University of California, San Francisco San Francisco General Hospital A 30-year old healthy woman comes for a routine visit. She is recently married and
NOTE: The above recommendations must be read along with the footnotes of this schedule.
 Figure 1. Recommended immunization schedule for persons aged 0 through 18 years 2013. (FOR THOSE WHO FALL BEHIND OR START LATE, SEE THE CATCH-UP SCHEDULE [FIGURE 2]). These recommendations must be read
Figure 1. Recommended immunization schedule for persons aged 0 through 18 years 2013. (FOR THOSE WHO FALL BEHIND OR START LATE, SEE THE CATCH-UP SCHEDULE [FIGURE 2]). These recommendations must be read
Recommended Immunization Schedules for Persons Aged 0 Through 18 Years UNITED STATES, 2016
 Recommended Immunization Schedules for Persons Aged 0 Through 18 Years UNITED STATES, 2016 This schedule includes recommendations in effect as of January 1, 2016. Any dose not administered at the recommended
Recommended Immunization Schedules for Persons Aged 0 Through 18 Years UNITED STATES, 2016 This schedule includes recommendations in effect as of January 1, 2016. Any dose not administered at the recommended
Immunization Guidelines for the Use of State Supplied Vaccine April 18, 2013
 DTaP / DT DTaP/IPV/Hep B Combination (Pediarix ) Children from 6 weeks of age up to the 7 th birthday Children from 2 months of age up to the 7th birthday: Indicated for the primary doses of DTaP, IPV,
DTaP / DT DTaP/IPV/Hep B Combination (Pediarix ) Children from 6 weeks of age up to the 7 th birthday Children from 2 months of age up to the 7th birthday: Indicated for the primary doses of DTaP, IPV,
Immunization Guidelines For the Use of State Supplied Vaccine July 1, 2011
 DTaP / DT DTaP/IPV/Hep B Combination (Pediarix ) Children from 6 weeks of age up to the 7 th birthday Children from 2 months of age up to the 7th birthday: Indicated for the primary doses of DTaP, IPV,
DTaP / DT DTaP/IPV/Hep B Combination (Pediarix ) Children from 6 weeks of age up to the 7 th birthday Children from 2 months of age up to the 7th birthday: Indicated for the primary doses of DTaP, IPV,
Series of 2 doses, 6-12 months apart. One dose is 720 Elu/0.5ml (GSK) or 25 u/0.5 ml (Merck)
 UTAH PREVENTIVE CARE RECOMMENDATIONS Adult - Ages 19 and Above IMMUNIZATIONS CONTENTS: General Instructions Hepatitis A Hepatitis B Human Papilloma Virus Influenza Meningococcal A, C, Y, W (MCV4) Meningococcal
UTAH PREVENTIVE CARE RECOMMENDATIONS Adult - Ages 19 and Above IMMUNIZATIONS CONTENTS: General Instructions Hepatitis A Hepatitis B Human Papilloma Virus Influenza Meningococcal A, C, Y, W (MCV4) Meningococcal
7.0 Nunavut Childhood and Adult Immunization Schedules and Catch-up Aids
 7.0 Nunavut Childhood and Adult Immunization Schedules and Catch-up Aids Contents Introduction Nunavut Recommended Childhood Immunization Schedule Nunavut Routine Adult Immunization Schedule Nunavut Immunization
7.0 Nunavut Childhood and Adult Immunization Schedules and Catch-up Aids Contents Introduction Nunavut Recommended Childhood Immunization Schedule Nunavut Routine Adult Immunization Schedule Nunavut Immunization
Immunization Guidelines for the Use of State Supplied Vaccine May 17, 2015
 DTaP / DT DTaP/IPV/Hep B Combination (Pediarix ) Children from 6 weeks of age up to the 7 th birthday Children from 2 months of age up to the 7th birthday: Indicated for the primary doses of DTaP, IPV,
DTaP / DT DTaP/IPV/Hep B Combination (Pediarix ) Children from 6 weeks of age up to the 7 th birthday Children from 2 months of age up to the 7th birthday: Indicated for the primary doses of DTaP, IPV,
Revisions to Communicable Disease Control Manual: Chapter 2 Immunization Program
 January 5, 2010 ATTN: Medical Health Officers and Branch Offices Public Health Nursing Administrators and Assistant Administrators Holders of Communicable Disease Control Manuals Re: Revisions to Communicable
January 5, 2010 ATTN: Medical Health Officers and Branch Offices Public Health Nursing Administrators and Assistant Administrators Holders of Communicable Disease Control Manuals Re: Revisions to Communicable
What DO the childhood immunization footnotes reveal? Questions and answers
 What DO the childhood immunization footnotes reveal? Questions and answers Stanley E. Grogg, DO, FACOP, FAAP he Advisory Committee on Immunization Practices (ACIP) recommends the childhood vaccination
What DO the childhood immunization footnotes reveal? Questions and answers Stanley E. Grogg, DO, FACOP, FAAP he Advisory Committee on Immunization Practices (ACIP) recommends the childhood vaccination
Post-Transplant Vaccination and Re-Immunisation Procedure
 Post-Transplant Vaccination and Re-Immunisation Procedure Table of Contents Purpose... 1 Scope/Audience... 1 Associated documents and forms... 1 Definitions... 2 Background... 2 Vaccination Recommendations...
Post-Transplant Vaccination and Re-Immunisation Procedure Table of Contents Purpose... 1 Scope/Audience... 1 Associated documents and forms... 1 Definitions... 2 Background... 2 Vaccination Recommendations...
Enhanced immunisation schedule Victoria
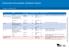 Children from March 2016 Age Disease Vaccine brand Birth Hepatitis B H-B-Vax-II Paediatric 2 months - from 6 weeks Diphtheria-tetanus-pertussis, poliomyelitis-hepatitis B- Reconstitute Site given Route
Children from March 2016 Age Disease Vaccine brand Birth Hepatitis B H-B-Vax-II Paediatric 2 months - from 6 weeks Diphtheria-tetanus-pertussis, poliomyelitis-hepatitis B- Reconstitute Site given Route
Vaccines, Not Just for Babies
 Vaccines, Not Just for Babies Meg Fisher, MD Medical Director Disclosures I have no relevant financial relationships with the manufacturers of any commercial products or commercial services discussed in
Vaccines, Not Just for Babies Meg Fisher, MD Medical Director Disclosures I have no relevant financial relationships with the manufacturers of any commercial products or commercial services discussed in
Immunisation Subcommittee of the Pharmacology and Therapeutics Advisory Committee (PTAC) Meeting held on 26 July (minutes for web publishing)
 Immunisation Subcommittee of the Pharmacology and Therapeutics Advisory Committee (PTAC) Meeting held on 26 July 2017 (minutes for web publishing) Immunisation Subcommittee minutes are published in accordance
Immunisation Subcommittee of the Pharmacology and Therapeutics Advisory Committee (PTAC) Meeting held on 26 July 2017 (minutes for web publishing) Immunisation Subcommittee minutes are published in accordance
2018/19 Immunisation programmes list of additional and enhanced services
 2018/19 Immunisation programmes list of additional and enhanced services 2018/19 Vaccination and Immunisation list of additional and enhanced services Version number: 1 First published: April 2018 Prepared
2018/19 Immunisation programmes list of additional and enhanced services 2018/19 Vaccination and Immunisation list of additional and enhanced services Version number: 1 First published: April 2018 Prepared
New NSW Immunisation Schedule
 New NSW Immunisation Schedule GP Update Prepared by Kym Bush Immunisation Coordinator Public Health Unit July 2018 Rationale for changes on 1 July 2018 Increase in cases of IPD in kiddies > 12 months Increase
New NSW Immunisation Schedule GP Update Prepared by Kym Bush Immunisation Coordinator Public Health Unit July 2018 Rationale for changes on 1 July 2018 Increase in cases of IPD in kiddies > 12 months Increase
New Jersey Department of Health Vaccine Preventable Disease Program Childhood and Adolescent Recommended Vaccines
 New Jersey Department of Health Vaccine Preventable Disease Program Childhood and Adolescent Recommended Vaccines Antigens Vaccine Approved Age Daptacel Diphtheria, Tetanus, and acellular Pertussis (DTaP)
New Jersey Department of Health Vaccine Preventable Disease Program Childhood and Adolescent Recommended Vaccines Antigens Vaccine Approved Age Daptacel Diphtheria, Tetanus, and acellular Pertussis (DTaP)
PHARMAC invites proposals for the supply of various vaccines in New Zealand.
 15 February 2016 Dear Supplier REQUEST FOR PROPOSALS SUPPLY OF VARIOUS VACCINES PHARMAC invites proposals for the supply of various vaccines in New Zealand. This request for proposals (RFP) letter incorporates
15 February 2016 Dear Supplier REQUEST FOR PROPOSALS SUPPLY OF VARIOUS VACCINES PHARMAC invites proposals for the supply of various vaccines in New Zealand. This request for proposals (RFP) letter incorporates
2017/18 Immunisation programmes list of additional and enhanced services
 2017/18 Immunisation programmes list of additional and enhanced services 2017/18 Vaccination and Immunisation list of additional and enhanced services Version number: 1 First published: April 2017 Prepared
2017/18 Immunisation programmes list of additional and enhanced services 2017/18 Vaccination and Immunisation list of additional and enhanced services Version number: 1 First published: April 2017 Prepared
Immunisation Policy. Country Children s Early Learning Ph: M:
 Immunisation Policy Published June 2018 Review Date December 2019 Sources Education and Care Services National Regulations, October 2017 Guide to the Education and Care Services National Law and the Education
Immunisation Policy Published June 2018 Review Date December 2019 Sources Education and Care Services National Regulations, October 2017 Guide to the Education and Care Services National Law and the Education
RECOMMENDED IMMUNIZATIONS
 Recommended Immunization Schedule for Persons Aged 0 Through 6 Years United States 2010 1 2 4 6 12 15 18 19 23 2 3 4 6 Vaccine Age Birth month months months months months months months months years years
Recommended Immunization Schedule for Persons Aged 0 Through 6 Years United States 2010 1 2 4 6 12 15 18 19 23 2 3 4 6 Vaccine Age Birth month months months months months months months months years years
Healthy People 2020 objectives were released in 2010, with a 10-year horizon to achieve the goals by 2020.
 Appendix 1: Healthy People 2020 Immunization-related Objectives Healthy People provides science-based, 10-year national objectives for improving the health of all Americans. For three decades, Healthy
Appendix 1: Healthy People 2020 Immunization-related Objectives Healthy People provides science-based, 10-year national objectives for improving the health of all Americans. For three decades, Healthy
Current Immunisation Issues in the Nepean Blue Mountains Presented as part of the Immunisation Update education series in October 2017
 Current Immunisation Issues in the Nepean Blue Mountains 2017 Presented as part of the Immunisation Update education series in October 2017 Additional funded vaccines Catch up vaccines for all individuals
Current Immunisation Issues in the Nepean Blue Mountains 2017 Presented as part of the Immunisation Update education series in October 2017 Additional funded vaccines Catch up vaccines for all individuals
The National Immunisation Schedule Update and Current issues. Dr Brenda Corcoran National Immunisation Office.
 The National Immunisation Schedule Update and Current issues Dr Brenda Corcoran National Immunisation Office Objectives To outline immunisation schedules in Ireland Primary childhood schedule Vaccine uptake
The National Immunisation Schedule Update and Current issues Dr Brenda Corcoran National Immunisation Office Objectives To outline immunisation schedules in Ireland Primary childhood schedule Vaccine uptake
Massachusetts Department of Public Health Recommended Immunization Schedule for Persons Aged 0-6 Years, 2007
 Vaccine Hepatitis B 1 Birth HepB 1 month Rotavirus 2 Rota Rota Rota Diphtheria, Tetanus, Pertussis 3 DTaP DTaP DTaP Haemophilus influenzae type b 4 Hib Hib Hib 4 Pneumococcal 5 PCV PCV PCV Inactivated
Vaccine Hepatitis B 1 Birth HepB 1 month Rotavirus 2 Rota Rota Rota Diphtheria, Tetanus, Pertussis 3 DTaP DTaP DTaP Haemophilus influenzae type b 4 Hib Hib Hib 4 Pneumococcal 5 PCV PCV PCV Inactivated
Immunisation Subcommittee of PTAC Meeting held 23 May (minutes for web publishing)
 Immunisation Subcommittee of PTAC Meeting held 23 May 2016 (minutes for web publishing) Immunisation Subcommittee minutes are published in accordance with the Terms of Reference for the Pharmacology and
Immunisation Subcommittee of PTAC Meeting held 23 May 2016 (minutes for web publishing) Immunisation Subcommittee minutes are published in accordance with the Terms of Reference for the Pharmacology and
Syrian Programme Refugees Advice on assessment of immunisation status and recommendations for additional immunisation
 Syrian Programme Refugees Advice on assessment of immunisation status and recommendations for additional immunisation Background Information Immunisation services in Syria Syria had good immunisation services,
Syrian Programme Refugees Advice on assessment of immunisation status and recommendations for additional immunisation Background Information Immunisation services in Syria Syria had good immunisation services,
2016/17 Vaccination and Immunisation list of additional services and enhanced services
 2016/17 Vaccination and Immunisation list of additional services and enhanced services 2016/17 Vaccination and Immunisation list of additional services and enhanced services Version number: 1 First published:
2016/17 Vaccination and Immunisation list of additional services and enhanced services 2016/17 Vaccination and Immunisation list of additional services and enhanced services Version number: 1 First published:
POLICIES AND PROCEDURES
 Purpose: To establish guidelines for Childhood Preventive Care Screening.To specify and define the Alliance guidelines for periodic health screening and preventive health services for members up to 21
Purpose: To establish guidelines for Childhood Preventive Care Screening.To specify and define the Alliance guidelines for periodic health screening and preventive health services for members up to 21
UPDATE ON IMMUNIZATION GUIDELINES AND PRACTICES
 DISCLOSURES UPDATE ON IMMUNIZATION GUIDELINES AND PRACTICES Nothing to disclose Kylie Mueller, Pharm.D., BCPS Clinical Specialist, Infectious Diseases Spartanburg Regional Medical Center LEARNING OBJECTIVES
DISCLOSURES UPDATE ON IMMUNIZATION GUIDELINES AND PRACTICES Nothing to disclose Kylie Mueller, Pharm.D., BCPS Clinical Specialist, Infectious Diseases Spartanburg Regional Medical Center LEARNING OBJECTIVES
Take advantage of preventive care to help manage your health
 Take advantage of preventive care to help manage your health Preventing disease and detecting health issues at an early stage, if they occur, are important to living a healthy life. Following the recommended
Take advantage of preventive care to help manage your health Preventing disease and detecting health issues at an early stage, if they occur, are important to living a healthy life. Following the recommended
Guidelines for the immunisation of children following treatment with Standard-Dose Chemotherapy
 Guidelines for the immunisation of children following treatment with Standard-Dose Chemotherapy Version 2.0 Approved by Haem / Onc Senior Clinical Management team Date Approved March 2015 Ratified by:
Guidelines for the immunisation of children following treatment with Standard-Dose Chemotherapy Version 2.0 Approved by Haem / Onc Senior Clinical Management team Date Approved March 2015 Ratified by:
Recommended Health Screenings
 Recommended Health Screenings UnitedHealthcare appreciates the preventive care you deliver to our members. Please use the below health screening chart to schedule screenings based on the member s age and
Recommended Health Screenings UnitedHealthcare appreciates the preventive care you deliver to our members. Please use the below health screening chart to schedule screenings based on the member s age and
National Immunisation News The newsletter of the HSE National Immunisation Office
 February 2015 National Immunisation News The newsletter of the HSE National Immunisation Office CONTENTS Changes to the PCI programme Uptake statistics Pneumovax name change Flu season Common queries School
February 2015 National Immunisation News The newsletter of the HSE National Immunisation Office CONTENTS Changes to the PCI programme Uptake statistics Pneumovax name change Flu season Common queries School
Appendix An Assessment Tool to Determine the Validity of Vaccine Doses
 Appendix 4.4 - An Assessment Tool to Determine the Validity of Vaccine Doses Note: Refer to the Canadian Immunization Guide and New Brunswick (NB) immunization program directives for recommendations for
Appendix 4.4 - An Assessment Tool to Determine the Validity of Vaccine Doses Note: Refer to the Canadian Immunization Guide and New Brunswick (NB) immunization program directives for recommendations for
Summary of Recommendations for Adult Immunization (Age 19 years and older) (Page 2 of 5)
 Summary of Recommendations for Adult Immunization (Age 19 years and older) (Page 1 of 5) Influenza Inactivated Influenza (IIV*) or ID (intradermally) *includes recombinant influenza (RIV) Live attenuated
Summary of Recommendations for Adult Immunization (Age 19 years and older) (Page 1 of 5) Influenza Inactivated Influenza (IIV*) or ID (intradermally) *includes recombinant influenza (RIV) Live attenuated
Guidelines for immunisation of children following treatment with high dose chemotherapy and Haematopoietic Stem Cell Transplantation (HSCT)
 Guidelines for immunisation of children following treatment with high dose chemotherapy and Haematopoietic Stem Cell Transplantation (HSCT) Version 2.0 Approved by Haem / Onc Senior Clinical Management
Guidelines for immunisation of children following treatment with high dose chemotherapy and Haematopoietic Stem Cell Transplantation (HSCT) Version 2.0 Approved by Haem / Onc Senior Clinical Management
Immunisation schedule Victoria
 Infants and children July 2018 Birth H-B-Vax-II Paediatric or Paediatric 2 months (from 6 weeks) and 4 months Diphtheria-tetanus-pertussis, poliomyelitis-hepatitis B- Haemophilus influenzae type b Anterolateral
Infants and children July 2018 Birth H-B-Vax-II Paediatric or Paediatric 2 months (from 6 weeks) and 4 months Diphtheria-tetanus-pertussis, poliomyelitis-hepatitis B- Haemophilus influenzae type b Anterolateral
National Immunisation News
 July 2015 National Immunisation News The newsletter of the HSE National Immunisation Office CONTENTS Changes to the Primary Childhood Immunisation Programme BCG Shortage Pertussis Vaccine Mumps Order Vaccines
July 2015 National Immunisation News The newsletter of the HSE National Immunisation Office CONTENTS Changes to the Primary Childhood Immunisation Programme BCG Shortage Pertussis Vaccine Mumps Order Vaccines
These slides are the property of the presenter. Do not duplicate without express written consent.
 Cancer Survivorship Protecting Against Vaccine Preventable Diseases Heidi Loynes BSN, RN Immunization Nurse Educator Michigan Department of health and Human Services (MDHHS) loynesh@michigan.gov Are Vaccine-Preventable
Cancer Survivorship Protecting Against Vaccine Preventable Diseases Heidi Loynes BSN, RN Immunization Nurse Educator Michigan Department of health and Human Services (MDHHS) loynesh@michigan.gov Are Vaccine-Preventable
Appendice History of Vaccine Use in New Brunswick- Chronology of vaccine use in New Brunswick Public Health programs*
 Appendice 4.3 - History of Vaccine Use in New Brunswick- 4.3.1 - History of Vaccine Use in New Brunswick - chronological listing Chronology of vaccine use in New Brunswick Public Health programs* * Please
Appendice 4.3 - History of Vaccine Use in New Brunswick- 4.3.1 - History of Vaccine Use in New Brunswick - chronological listing Chronology of vaccine use in New Brunswick Public Health programs* * Please
4 Immunisation of special groups
 4 special groups This chapter discusses the special immunisation requirements of individuals at risk of vaccine-preventable diseases due to certain conditions or underlying disease, or through their occupation
4 special groups This chapter discusses the special immunisation requirements of individuals at risk of vaccine-preventable diseases due to certain conditions or underlying disease, or through their occupation
Take advantage of preventive care to help manage your health
 UnitedHealthcare Preventive Plan Design Employee Take advantage of preventive care to help manage your health Preventing disease and detecting health issues at an early stage, if they occur, are important
UnitedHealthcare Preventive Plan Design Employee Take advantage of preventive care to help manage your health Preventing disease and detecting health issues at an early stage, if they occur, are important
Note from the National Guideline Clearinghouse (NGC): The guideline recommendations are presented in the form of tables with footnotes (see below).
 Brief Summary GUIDELINE TITLE Recommended immunization schedules for persons aged 0 through 18 years: United States, 2009. BIBLIOGRAPHIC SOURCE(S) American Academy of Pediatrics Committee on Infectious
Brief Summary GUIDELINE TITLE Recommended immunization schedules for persons aged 0 through 18 years: United States, 2009. BIBLIOGRAPHIC SOURCE(S) American Academy of Pediatrics Committee on Infectious
Release Notes. Medtech Evolution General Practice. Version Build (June 2016)
 Release Notes Medtech Evolution General Practice Version 1.5.7 Build 1.5.7.72 (June 2016) These release notes contain important information for Medtech Evolution users. Please ensure that they are circulated
Release Notes Medtech Evolution General Practice Version 1.5.7 Build 1.5.7.72 (June 2016) These release notes contain important information for Medtech Evolution users. Please ensure that they are circulated
kernfamilyhealthcare.com. Si necesita esta información en español, por favor llámenos.
 Together in... p revention Preventive Care Gu id e Kern Family Health Care wants you to get good health care. These preventive care guidelines help you stay healthy by preventing diseases or by finding
Together in... p revention Preventive Care Gu id e Kern Family Health Care wants you to get good health care. These preventive care guidelines help you stay healthy by preventing diseases or by finding
Immunise your child on time. It s their best protection
 Immunise your child on time It s their best protection If you are a parent or caregiver, this pamphlet is for you. It tells you about the recommended ages for your baby to receive their immunisations.
Immunise your child on time It s their best protection If you are a parent or caregiver, this pamphlet is for you. It tells you about the recommended ages for your baby to receive their immunisations.
Immunization Update. 23 rd October Natasha S. Crowcroft Chief, Infectious Diseases Public Health Ontario
 Immunization Update 23 rd October 2013 Natasha S. Crowcroft Chief, Infectious Diseases Public Health Ontario Immunization: It s Your Best Shot! Faculty/Presenter Disclosure Faculty: Dr. Natasha Crowcroft
Immunization Update 23 rd October 2013 Natasha S. Crowcroft Chief, Infectious Diseases Public Health Ontario Immunization: It s Your Best Shot! Faculty/Presenter Disclosure Faculty: Dr. Natasha Crowcroft
National Immunisation News
 National Immunisation News The newsletter of the HSE National Immunisation Office July 2016 Changes to the Primary Childhood Immunisation Programme The National Immunisation Advisory Committee (NIAC) has
National Immunisation News The newsletter of the HSE National Immunisation Office July 2016 Changes to the Primary Childhood Immunisation Programme The National Immunisation Advisory Committee (NIAC) has
ACIP Meeting Update, New Recommendations and Pending Influenza Season
 ACIP Meeting Update, New Recommendations and Pending Influenza Season February 17 th 2011 www.immunizetexas.com ACIP Upcoming Agenda and New Recommendations ACIP (February 23-24 th 2011) Topics for meeting
ACIP Meeting Update, New Recommendations and Pending Influenza Season February 17 th 2011 www.immunizetexas.com ACIP Upcoming Agenda and New Recommendations ACIP (February 23-24 th 2011) Topics for meeting
ANNUAL HEALTH SCREENINGS AND IMMUNIZATIONS GUIDE MEN WOMEN ALL ADULTS CHILDREN
 AND IMMUNIZATIONS GUIDE MEN WOMEN ALL ADULTS CHILDREN MEN PROSTATE CANCER Testicular exam Age 18+ PSA test Ages 50-75, based on risk WOMEN BREAST CANCER Self breast exam Monthly Clinical breast exam Annually
AND IMMUNIZATIONS GUIDE MEN WOMEN ALL ADULTS CHILDREN MEN PROSTATE CANCER Testicular exam Age 18+ PSA test Ages 50-75, based on risk WOMEN BREAST CANCER Self breast exam Monthly Clinical breast exam Annually
WELSH HEALTH CIRCULAR
 WELSH HEALTH CIRCULAR WHC (2017) 022 Issue Date: 12 May 2017 STATUS: ACTION CATEGORY: PUBLIC HEALTH Title: Change of vaccine for the routine primary infant immunisation Date of Expiry / Review N/A For
WELSH HEALTH CIRCULAR WHC (2017) 022 Issue Date: 12 May 2017 STATUS: ACTION CATEGORY: PUBLIC HEALTH Title: Change of vaccine for the routine primary infant immunisation Date of Expiry / Review N/A For
Physicians Update 1 September 2015 From the Office of the Chief Medical Health Officer
 1 September 2015 Immunization Bulletin (Please share with your medical office assistants) 1. New publicly funded vaccines a. Expanded eligibility for free HPV vaccine for select males 9-26 years of age
1 September 2015 Immunization Bulletin (Please share with your medical office assistants) 1. New publicly funded vaccines a. Expanded eligibility for free HPV vaccine for select males 9-26 years of age
Andrew Kroger, MD, MPH National Center for Immunization and Respiratory Diseases MCH & Immunization Conference Anchorage, AK September 28, 2010
 2010 Immunization Update Andrew Kroger, MD, MPH National Center for Immunization and Respiratory Diseases MCH & Immunization Conference Anchorage, AK September 28, 2010 Disclosures No financial conflict
2010 Immunization Update Andrew Kroger, MD, MPH National Center for Immunization and Respiratory Diseases MCH & Immunization Conference Anchorage, AK September 28, 2010 Disclosures No financial conflict
Streptococcus pneumoniae CDC
 Streptococcus pneumoniae CDC Pneumococcal Disease Infection caused by the bacteria, Streptococcus pneumoniae» otitis media 20 million office visits (28-55% Strep)» pneumonia 175,000 cases annually» meningitis
Streptococcus pneumoniae CDC Pneumococcal Disease Infection caused by the bacteria, Streptococcus pneumoniae» otitis media 20 million office visits (28-55% Strep)» pneumonia 175,000 cases annually» meningitis
Preventive care is important at every age. Making good health choices now can boost your health and well-being for a lifetime.
 Adult Recommendations Preventive care is important at every age. Making good health choices now can boost your health and well-being for a lifetime. Asthma and COPD Well-Child Visits Children s Immunization
Adult Recommendations Preventive care is important at every age. Making good health choices now can boost your health and well-being for a lifetime. Asthma and COPD Well-Child Visits Children s Immunization
1.0 ROUTINE SCHEDULES...
 August 2012 TABLE OF CONTENTS 1.0 ROUTINE SCHEDULES... 1 1.1 SCHEDULE A: BASIC IMMUNIZATION WHEN STARTING WITH INFANRIX HEXA VACCINE... 1 1.1.1 SCHEDULE A: BASIC IMMUNIZATION WHEN STARTING WITH PEDIACEL
August 2012 TABLE OF CONTENTS 1.0 ROUTINE SCHEDULES... 1 1.1 SCHEDULE A: BASIC IMMUNIZATION WHEN STARTING WITH INFANRIX HEXA VACCINE... 1 1.1.1 SCHEDULE A: BASIC IMMUNIZATION WHEN STARTING WITH PEDIACEL
Childhood Immunisations Template Guide 2017
 Childhood Immunisations Template Guide 2017 1 Template Control Page Childhood Immunisations Template Guide Title Childhood Immunisations Author CEG Description Supports the schedule of Childhood Immunisations
Childhood Immunisations Template Guide 2017 1 Template Control Page Childhood Immunisations Template Guide Title Childhood Immunisations Author CEG Description Supports the schedule of Childhood Immunisations
IMMUNIZATION IN CHILDREN WITH CANCER
 SIOP PODC Supportive Care Education Presentation Date: 05 th September 2014 Recording Link at www.cure4kids.org: http://www.cure4kids.org/ums/home/conference_rooms/enter.php?room=p2xokm5imdj Email: ahmed.naqvi@sickkids.ca
SIOP PODC Supportive Care Education Presentation Date: 05 th September 2014 Recording Link at www.cure4kids.org: http://www.cure4kids.org/ums/home/conference_rooms/enter.php?room=p2xokm5imdj Email: ahmed.naqvi@sickkids.ca
Immunisation Subcommittee of PTAC Meeting held 28 October 2015
 Immunisation Subcommittee of PTAC Meeting held 28 October 2015 (minutes for web publishing) Immunisation Subcommittee minutes are published in accordance with the Terms of Reference for the Pharmacology
Immunisation Subcommittee of PTAC Meeting held 28 October 2015 (minutes for web publishing) Immunisation Subcommittee minutes are published in accordance with the Terms of Reference for the Pharmacology
Preventative Vaccines. Vaccines for Special Populations. Vaccinations for Adults: An Update. Vaccines Generally Available in the U.S.
 Vaccinations for Adults: An Update Preventative Vaccines Need to be extremely safe Even greater issue as disease prevalence wanes or uncommon diseases targeted Lisa G. Winston, MD University of California,
Vaccinations for Adults: An Update Preventative Vaccines Need to be extremely safe Even greater issue as disease prevalence wanes or uncommon diseases targeted Lisa G. Winston, MD University of California,
Proof of residency in East Orange is mandatory (see Residency Requirements)
 Pre-K Registration Requirements Child must be at least 3 or 4 years old by October 1st of the current school year Immunization (shot records) are mandatory Age appropriate vaccinations for children entering
Pre-K Registration Requirements Child must be at least 3 or 4 years old by October 1st of the current school year Immunization (shot records) are mandatory Age appropriate vaccinations for children entering
Recommended Immunization Schedule for Adults Aged 19 Years or Older, United States, 2017
 Caring for Our Children: National Health and Safety Performance Standards H Recommended Immunization Schedule for Adults Aged 19 Years or Older, United States, 2017 In February 2017, the Recommended Immunization
Caring for Our Children: National Health and Safety Performance Standards H Recommended Immunization Schedule for Adults Aged 19 Years or Older, United States, 2017 In February 2017, the Recommended Immunization
APEC Guidelines Immunizations
 Pregnancy provides an excellent opportunity to enhance a woman s protection against disease and to provide protection to the neonate during the first 3 to 6 months of life. Women of childbearing age should
Pregnancy provides an excellent opportunity to enhance a woman s protection against disease and to provide protection to the neonate during the first 3 to 6 months of life. Women of childbearing age should
Immunizations (Guideline Intervals Using The Rule of Six for Vaccines Birth to Six Years)
 Immunizations (Guideline Intervals Using The Rule of Six for Vaccines Birth to Six Years) Guideline developed by Shelly Baldwin, MD, in collaboration with the ANGELS Team. Last reviewed by Shelly Baldwin,
Immunizations (Guideline Intervals Using The Rule of Six for Vaccines Birth to Six Years) Guideline developed by Shelly Baldwin, MD, in collaboration with the ANGELS Team. Last reviewed by Shelly Baldwin,
type b (Hib) disease
 6 Haemophilus influenzae Haemophilus influenzae Key information Mode of transmission Incubation period Period of communicability Disease burden Funded vaccines Dose, presentation, route By inhalation of
6 Haemophilus influenzae Haemophilus influenzae Key information Mode of transmission Incubation period Period of communicability Disease burden Funded vaccines Dose, presentation, route By inhalation of
TDCJ OFFENDER IMMUNIZATION GUIDELINES
 TDCJ OFFENDER IMMUNIZATION GUIDELINES B-14.07 Immunizations Attachment A VACCINE WHO GETS IT * WHEN THEY GET IT DOSE/ROUTE HPV Vaccine Females age 9 to 26 years if not previously vaccinated. On Intake
TDCJ OFFENDER IMMUNIZATION GUIDELINES B-14.07 Immunizations Attachment A VACCINE WHO GETS IT * WHEN THEY GET IT DOSE/ROUTE HPV Vaccine Females age 9 to 26 years if not previously vaccinated. On Intake
Immunization for Child Hematopoietic Stem Cell Transplant (HSCT) Recipients
 Immunization for Child Recipients January 4, 201 Immunization for Child Hematopoietic Stem Cell Transplant () Recipients Revision Date: January 4, 201 Note: This guide is meant to supplement existing recommendations
Immunization for Child Recipients January 4, 201 Immunization for Child Hematopoietic Stem Cell Transplant () Recipients Revision Date: January 4, 201 Note: This guide is meant to supplement existing recommendations
Routine Immunization Products
 Newfoundland and Labrador Immunization Manual Section 3 Routine Immunization Products Immunization with Diphtheria, Pertussis, Tetanus, Polio and Hib Vaccine (DTap-IPV- Hib 3.1 Immunization with Diphtheria,
Newfoundland and Labrador Immunization Manual Section 3 Routine Immunization Products Immunization with Diphtheria, Pertussis, Tetanus, Polio and Hib Vaccine (DTap-IPV- Hib 3.1 Immunization with Diphtheria,
CVU: What s new? 2 nd December 2013
 CVU: What s new? 2 nd December 2013 Dr Margie Danchin Paediatrician, Department of General Medicine, RCH Senior Research Fellow, MCRI Senior Fellow, Department of Paediatrics, The University of Melbourne
CVU: What s new? 2 nd December 2013 Dr Margie Danchin Paediatrician, Department of General Medicine, RCH Senior Research Fellow, MCRI Senior Fellow, Department of Paediatrics, The University of Melbourne
Chapter 16 Pneumococcal Infection. Pneumococcal Infection. August 2015
 Chapter 16 16 PPV introduced for at risk 1996 PCV7 introduced for at risk 2002 and as routine 2008 PCV13 replaced PCV7 in 2010 NOTIFIABLE In some circumstances, advice in these guidelines may differ from
Chapter 16 16 PPV introduced for at risk 1996 PCV7 introduced for at risk 2002 and as routine 2008 PCV13 replaced PCV7 in 2010 NOTIFIABLE In some circumstances, advice in these guidelines may differ from
Immunisation Handbook. (2 nd edition, March 2018)
 Immunisation Handbook 2017 (2 nd edition, March 2018) Released March 2018 health.govt.nz Disclaimer This publication, which has been prepared for, and is published by, the Ministry of Health, is for the
Immunisation Handbook 2017 (2 nd edition, March 2018) Released March 2018 health.govt.nz Disclaimer This publication, which has been prepared for, and is published by, the Ministry of Health, is for the
Routine Immunization Schedules. Section 2. Newfoundland and Labrador Immunization Manual. Routine Immunization Schedules
 Newfoundland and Labrador Immunization Manual Section 2... 2.1 Routine and Delayed Immunization Schedules for Infants and Children... 2.2 Recommended Immunizations for Adults... 2.3 (Provinces and Territories)...
Newfoundland and Labrador Immunization Manual Section 2... 2.1 Routine and Delayed Immunization Schedules for Infants and Children... 2.2 Recommended Immunizations for Adults... 2.3 (Provinces and Territories)...
Current National Immunisation Schedule Dr Brenda Corcoran National Immunisation Office.
 Current National Immunisation Schedule 2012 Dr Brenda Corcoran National Immunisation Office Objectives To outline immunisation schedules in Ireland Primary childhood schedule Vaccine uptake rates School
Current National Immunisation Schedule 2012 Dr Brenda Corcoran National Immunisation Office Objectives To outline immunisation schedules in Ireland Primary childhood schedule Vaccine uptake rates School
Immunisation Subcommittee of PTAC Meeting held 18 February 2015
 Immunisation Subcommittee of PTAC Meeting held 18 February 2015 (minutes for web publishing) Immunisation Subcommittee minutes are published in accordance with the Terms of Reference for the Pharmacology
Immunisation Subcommittee of PTAC Meeting held 18 February 2015 (minutes for web publishing) Immunisation Subcommittee minutes are published in accordance with the Terms of Reference for the Pharmacology
Vaccines Indicated for Infants, Children, and Adolescents Based on Medical and Other Indications
 Vaccines Indicated for Infants, Children, and Adolescents Based on Medical and Other Indications Vaccine Prematurity 1 Altered Immunocompetence 2 (excluding human immunodefi ciency virus [HIV] infection)
Vaccines Indicated for Infants, Children, and Adolescents Based on Medical and Other Indications Vaccine Prematurity 1 Altered Immunocompetence 2 (excluding human immunodefi ciency virus [HIV] infection)
Take advantage of preventive care to help manage your health
 Take advantage of preventive care to help manage your health Preventing disease and detecting health issues at an early stage, if they occur, are important to living a healthy life. Following the recommended
Take advantage of preventive care to help manage your health Preventing disease and detecting health issues at an early stage, if they occur, are important to living a healthy life. Following the recommended
Immunization Update: New CDC Recommendations. Blaise L. Congeni M.D. 2012
 Immunization Update: New CDC Recommendations Blaise L. Congeni M.D. 2012 Polysaccharide Vaccines Vaccine Hib capsule polysaccharide PRP (polyribose ribitol phosphate) Not protective in infants
Immunization Update: New CDC Recommendations Blaise L. Congeni M.D. 2012 Polysaccharide Vaccines Vaccine Hib capsule polysaccharide PRP (polyribose ribitol phosphate) Not protective in infants
Acknowledgements. Introduction. Structure of the video
 Educators Guide Acknowledgements The Ministry of Health would like to thank Blue Bicycle Flicks. Thanks also to the staff and students from Evans Bay Intermediate School who contributed to the shooting
Educators Guide Acknowledgements The Ministry of Health would like to thank Blue Bicycle Flicks. Thanks also to the staff and students from Evans Bay Intermediate School who contributed to the shooting
Take advantage of preventive care to help manage your health
 Take advantage of preventive care to help manage your health UnitedHealthcare is dedicated to helping people live healthier lives, and we encourage our members to receive age and gender appropriate preventive
Take advantage of preventive care to help manage your health UnitedHealthcare is dedicated to helping people live healthier lives, and we encourage our members to receive age and gender appropriate preventive
Birth 6 wk 10 wk 14 wk 18 wk 6 mo 9 mo 12 mo 15 mo 18 mo mo 2-3 Yr 4-6 Yr 7-10Yr Yr 13-18Yr
 Vaccine BCG Age Birth 6 wk 10 wk 14 wk 18 wk 6 mo 9 mo 12 mo 15 mo 18 mo 19-23 mo 2-3 Yr 4-6 Yr 7-10Yr 11-12 Yr 13-18Yr BCG Hep B Hep B1 Hep B2 Hep B3 Polio OPV 0 IPV1 IPV2 IPV3 OPV1 OPV2 IPV B1 OPV3 DTP
Vaccine BCG Age Birth 6 wk 10 wk 14 wk 18 wk 6 mo 9 mo 12 mo 15 mo 18 mo 19-23 mo 2-3 Yr 4-6 Yr 7-10Yr 11-12 Yr 13-18Yr BCG Hep B Hep B1 Hep B2 Hep B3 Polio OPV 0 IPV1 IPV2 IPV3 OPV1 OPV2 IPV B1 OPV3 DTP
Mandates and More. Julie Morita, M.D. Deputy Commissioner Chicago Department of Public Health. Chicago Department of Public Health
 Mandates and More Julie Morita, M.D. Deputy Chicago Department of Public Health Why are vaccines required for school entry? School Vaccine Requirements Small pox vaccine required in Massachusetts 1855
Mandates and More Julie Morita, M.D. Deputy Chicago Department of Public Health Why are vaccines required for school entry? School Vaccine Requirements Small pox vaccine required in Massachusetts 1855
HIT ME WITH YOUR BEST SHOT: UPDATE ON IMMUNIZATIONS. Karen Hoang, PharmD Clinical Pharmacy Specialist, MTM Services UPMC Health Plan
 HIT ME WITH YOUR BEST SHOT: UPDATE ON IMMUNIZATIONS Karen Hoang, PharmD Clinical Pharmacy Specialist, MTM Services UPMC Health Plan DISCLOSURES No financial or other conflicts to disclose LEARNING OBJECTIVES
HIT ME WITH YOUR BEST SHOT: UPDATE ON IMMUNIZATIONS Karen Hoang, PharmD Clinical Pharmacy Specialist, MTM Services UPMC Health Plan DISCLOSURES No financial or other conflicts to disclose LEARNING OBJECTIVES
Immunization Resources for Pharmacists
 Immunization Resources for Pharmacists TABLE OF CONTENTS Child / Teen Resources Screening Questionnaire Child/Teen... 3 Vaccine Schedule Age 0-6yrs... 5 Catch-Up Schedule 4mo-18yrs... 6 Vaccine Schedule
Immunization Resources for Pharmacists TABLE OF CONTENTS Child / Teen Resources Screening Questionnaire Child/Teen... 3 Vaccine Schedule Age 0-6yrs... 5 Catch-Up Schedule 4mo-18yrs... 6 Vaccine Schedule
UNSCHEDULED VACCINATION OF CHILDREN AND YOUNG PEOPLE WHO HAVE OUTSTANDING ROUTINE IMMUNISATIONS. Service Specification
 UNSCHEDULED VACCINATION OF CHILDREN AND YOUNG PEOPLE WHO HAVE OUTSTANDING ROUTINE IMMUNISATIONS Service Specification National Enhanced Service Specification For The Unscheduled Vaccination Of Children
UNSCHEDULED VACCINATION OF CHILDREN AND YOUNG PEOPLE WHO HAVE OUTSTANDING ROUTINE IMMUNISATIONS Service Specification National Enhanced Service Specification For The Unscheduled Vaccination Of Children
16 November 2017 National Immunisation Advisory Committee Recommendations for the 2017/2018 Influenza Vaccination Campaign
 16 November 2017 National Immunisation Advisory Committee Recommendations for the 2017/2018 Influenza Vaccination Campaign Please note the National Immunisation Advisory Committee (NIAC) has updated the
16 November 2017 National Immunisation Advisory Committee Recommendations for the 2017/2018 Influenza Vaccination Campaign Please note the National Immunisation Advisory Committee (NIAC) has updated the
Vaccine Preventable Diseases. Rebecca Ward Utah Department of Health Bureau of Epidemiology October 9, 2013
 Vaccine Preventable Diseases In Utah: 2013 Rebecca Ward Utah Department of Health Bureau of Epidemiology October 9, 2013 VPD Overview H3N2v Influenza Seasonal Influenza Pertussis Haemophilus influenzae
Vaccine Preventable Diseases In Utah: 2013 Rebecca Ward Utah Department of Health Bureau of Epidemiology October 9, 2013 VPD Overview H3N2v Influenza Seasonal Influenza Pertussis Haemophilus influenzae
VACCINATION FOR WOMEN (ADOLESCENCE TO SENESCENCE)
 VACCINATION FOR WOMEN (ADOLESCENCE TO SENESCENCE) Dr.T.K.SHAANTHY GUNASINGH M.D., D.G.O., FICOG PROFESSOR AND HEAD OF THE DEPARTMENT OF OBSTETRICS AND GYNAECOLOGY PROGRAMME DIRECTOR CENTRE OF EXCELLENCE
VACCINATION FOR WOMEN (ADOLESCENCE TO SENESCENCE) Dr.T.K.SHAANTHY GUNASINGH M.D., D.G.O., FICOG PROFESSOR AND HEAD OF THE DEPARTMENT OF OBSTETRICS AND GYNAECOLOGY PROGRAMME DIRECTOR CENTRE OF EXCELLENCE
