Immunisation Schedule. July Guide
|
|
|
- Rosamond Cummings
- 6 years ago
- Views:
Transcription
1 Immunisation Schedule July 2013 Guide This manual is for clinical staff and contains important information for all Medtech 32 users. Please ensure this document is circulated amongst all your clinical staff. We suggest these should be filed safely for future reference.
2 Table of Contents Introduction... 3 Pre-requisites... 4 Add a new Vaccine Code... 5 Add a New Vaccine Age Group... 7 Add & Map a New Vaccine Product... 9 Immunisation Schedule Utility...12 Add a New Schedule Path...13 Appendix A...17 Standard Vaccines...17 Non-Standard Vaccines...18 Sub-Population Vaccines...18 Queensland Providers Only...18 Version 9.0 Page 2 of 18
3 Introduction The Medtech32 Immunisation Schedule Guide has been written to provide assistance to your Clinical staff in the creation of new vaccines, how to edit existing vaccines, and how to add new vaccines to the Medtech32 Vaccine Product Mapping screen. This document will also assist with the new July 2013 Immunisation Schedule changes for: The addition of 3 new Influenza vaccines: 1. Agrippal (AGRPAL) 2. Fluvax Junior 3. Vaxigrip Junior (VGRJNR) The addition also of two new vaccine code edits for the new: 1. Measles, Mumps, Rubella and Varicella (MMRV) 2. Priorix-Tetra and ProQuad (which will be available from 1 July 2013). Setup and Configuration instructions on the use of the Immunisation Schedule Utility have been broken down in to separate sections so that you may select and use those that are relevant to your needs. These are covered below and describe how to: Add new Vaccines Add new Vaccine Groups Add new Age Range groups Add new Immunisation Path (as per your State and Practice requirements). Please refer to the Immunisation link for further information on the requirements for your State The Medtech Immunisation Schedule Utility is available from our website as per the link: 8/useful-tools-and-documents.html Please download and save this Utility - Ensure that it is saved in to the correct folder path described below (where your MT32 Bin resides) so that you may access this Utility at any time from within the Utility Menu in your Medtech32 system. A screenshot of the folder is shown below. (Drive location where you MT32 lives)\mt32\bin\tools\addins folder. Version 9.0 Page 3 of 18
4 Pre-requisites Medtech32 Immunisation Schedule Guide Please ensure that the person who will be altering or creating new Pathways or Vaccines in your Medtech32 system has read and fully understands the process as described in this instruction guide. The requirements for the successful management of your Immunisation Schedule have been listed below, and must be applied prior to adding any new vaccines or pathways: Ensure that the staff member updating the Immunisation Schedule has the correct Medtech32 Administration and Clinical Rights and access. Ensure that you have run the latest Medtech32 Vaccine Product Mapping Utility (March 2013) from our website on all your Medtech32 databases. The March 2013 update will add/edit the following vaccines and complete the additional tasks as numbered below. Priorix-Tetra ProQuad Agrippal Vaxigrip Junior ACIR Vaccine Code / Description (Old) PRXTET PROQAD AGRPAL VGRJNR ACIR Vaccine Code / Description (New) 1. It will add the new Medicare ACIR vaccine as listed above. 2. It will recreate any current Medicare ACIR vaccines that are missing from your Medtech32 database. 3. It will remove any invalid or obsolete Medicare ACIR vaccines from your Medtech32 database. 4. It will remove any duplicated Medicare ACIR vaccines from your Medtech32 database. NOTE: If the Utility is unable to determine which of the duplicated records should be kept, all duplicates will be deleted and the corresponding Medicare ACIR vaccine will then be recreated. 5. For all newly added or recreated Medicare ACIR vaccines, the Utility will default the Medtech32 Vaccine Code (MT32 Vaccine) to 'TEST', and the Medtech32 Vaccine Product Code (MT32 Code) to 'TEST' under Vaccine Product Mapping. 6. All existing Vaccine Product Mapping WILL NOT be altered, except for those Medicare ACIR vaccines that are recreated by the Utility. Version 9.0 Page 4 of 18
5 Add a new Vaccine Code Medtech32 Immunisation Schedule Guide 1. Go to the menu option Setup > Recall / Screening > Vaccine. 2. To add the new Vaccine, click on the first icon at the top of the screen to Add the new vaccine. This will open a new screen for you to add the Code, and Description of the Vaccine. Example in the screenshot below is for the vaccine Menitorix Important Note: Ensure that the Code used is of the correct standards as per Appendix A of this document. 3. After adding the Code and Description, click on the Products tab of the screen. Then click on the Add button to the right to Add the Product details in here. Version 9.0 Page 5 of 18
6 Enter in the relevant information: PRODUCT CODE: < Refer to the correct Product Code Table in Appendix A> PRODUCT DESCRIPTION: < Refer to the correct Product Description in Appendix A> Add Batch No: (use your existing batch details) Batch Expiry: (use your existing expiry date details) 4. The Outcome Tab will be filled in automatically with the default outcome responses. 5. Click on the Add button to add new Outcomes if required. 6. Click on the OK button to save the Product Code entry. Version 9.0 Page 6 of 18
7 Add a New Vaccine Age Group 1. To include a new Age Range for a Vaccine Group to appear in the Immunisation Schedule screen - go to the Setup > Recall / Screening > Vaccine Group menu option. The Vaccine Group screen will appear as shown below. 2. To add a new Code, click on the new icon to open up the relevant screen. Version 9.0 Page 7 of 18
8 3. Enter in the unique Code and a meaningful Description in to the relevant fields as shown in the screenshot sample below. 4. Click on the OK button to save the new Code entry. Below is a sample screenshot of various new Codes and Descriptions created, to accommodate an advanced Age Range selection. Version 9.0 Page 8 of 18
9 Add & Map a New Vaccine Product Important Note: Before you can correctly configure your new Vaccines in to your Vaccine Product Mapping screen, you must first have ensured that the latest Vaccine Product Mapping Utility has been successfully run on your Medtech32 database. This utility can be downloaded from our Medtech website 1. Select the Setup > Recall / Screening > Vaccine Product Mapping menu option. The following screen will be displayed. 2. The Vaccine Product Mapping screen consists of four columns. Medicare Australia Vaccine Code o This is the Medicare accepted code that will be sent electronically to ACIR. MT32 Vaccine o This is from your Vaccine that you created in Medtech (also shows the associated Code used) MT32 Code o This is the Code that you created for entering your Vaccine details MT32 Vaccine Product o This contains the details of the Product names that make up your vaccine (populated from the Vaccines Product Tab). Version 9.0 Page 9 of 18
10 3. Using the first column as your guide Medicare Aust. Vaccine - scroll down the list until you view the new vaccine Menitorix that we created at the start of this document. Note: the new Medicare Vaccine codes were added to your Medtech32 database via the Vaccine Product Mapping (March) 2013 utility that was run earlier. 4. The Mt32 Vaccine, Mt32 Code, and Mt32 Vaccine Product columns are set to the default of Test Vaccine (TEST) as a place holder for you to correctly map your Vaccine to the correct Medicare Codes. To change this, click on the MT32 Vaccine column to alter the code from Test Vaccine (TEST) by using the drop down menu to select your new Vaccine code MNTRX. Version 9.0 Page 10 of 18
11 5. Once the MT32 Vaccine code has been selected-repeat the process in the next column. 6. The end result will be the updated Vaccine Product Mapping screen as shown in the screenshot sample below. 7. Click on the OK button to save the changes. Version 9.0 Page 11 of 18
12 Immunisation Schedule Utility Important Note: Only those Vaccines entered in your Vaccine Product Mapping list will be displayed as a valid option in the Immunisation Schedule Utility. 1. From the Utility > Immunisation Schedule menu, select the right arrow option to open up the Immunisation Schedule. 2. The Immunisation Schedule screen will open with the existing Pathways shown, as per the screenshot sample below: Note : The schedules shown in the screenshot above may differ greatly (or only slightly) from the Immunisations Schedule screen in your Practice and is for demonstration purposes only. Version 9.0 Page 12 of 18
13 Add a New Schedule Path Medtech32 Immunisation Schedule Guide 1. From the Immunisation Schedule screen, click on the Add New Path button to create your new Immunisation Schedule Pathway. 2. The Add New Path button will open up the following screen to allow you to create your new entry. Enter in the Description and the Start Date in to the relevant fields. Click on the OK button to save the new entry. 3. Once you have clicked on the OK button, the next screen that appears is the actual Immunisation Schedule configuration process screen. 4. This screen will allow you to define the Vaccines for each Age Range, and is broken in to three sections: The first section contains details of the name and description of the Path, along with the valid start time of the schedule entry. Version 9.0 Page 13 of 18
14 The second section (left hand side of the screen) contains the Age Range from the drop down list, and the various Vaccines that may be selected/ticked for inclusion of the various Age Range vaccines. The third section (right hand side of the screen) contains the list of specified Vaccines for each Age Range as selected from the drop down list. Note: You need to alter the Age Range each time to add/exclude vaccines to the list to the right. Then use the right arrow icon right hand side of the screen. to move the selected Vaccine to appear on the If a vaccine selection has been made in error, use the right hand side of the screen. icon to remove it from the Version 9.0 Page 14 of 18
15 Note: From the example below, we have selected BIRTH from the drop down list, and have then selected/ticked the Hepatitis B vaccine directly from the vaccine list below. 5. The next Age Range is 2 Months. In the screenshot below, we have added the Infanrix Hexa to the schedule for 2 Months already (shown on the right hand side of the screen). We have ticked the two vaccines on the left hand side of the screen in readiness to move them over to be included in the 2 Months vaccines. Version 9.0 Page 15 of 18
16 6. The next Age Range is 4 Months, so we will need to look at is the 4 Month from the Drop Down menu option. Work your way through the list using the Drop Down arrow to select the next appropriate Age Range, as shown in the next screenshot example. 7. Continue to work through the Age Range, and add the required vaccines to the right hand side of the screen. The end result (continuing on from the Queensland example) will result in the final screen showing the new entries as shown in the sample screenshot below. Version 9.0 Page 16 of 18
17 Appendix A Medtech32 Immunisation Schedule Guide Standard Vaccines Vaccine/Brand Name Vaccine Code Max doses ActHIB PRPT 4 CDT - combined diphtheria, tetanus CDT 4 Comvax CMX 3 Engerix B (paediatric) ENGP 3 Generic Diphtheria GNDIP 4 Generic DTPa DTPA 4 Generic HBV HBV 3 Generic Hepatitis B GNHEP 3 Generic HIB GNHIB 3 Generic Measles GNMEA 3 Generic Meningococcal C GNMEN 3 Generic MMR MMR 3 Generic Mumps GNMUM 3 Generic (Monovalent) Pertussis P 5 Generic Pneumococcal GNPNE 3 Generic Poliomyelitis GNPOL 4 Generic Rubella GNRUB 3 Generic Tetanus GNTET 4 Generic Varicella-Zoster GNVAR 1 HBVAX II (paediatric) HBVP 3 Hiberix HBX 4 HibTITER HBOC 4 Infanrix IFX 4 Infanrix-HepB IFXB 3 Infanrix Hexa IFHX 3 Infanrix-IPV IFIP 4 Infanrix Penta IFPA 3 IPOL - inactivated polio vaccine IPV 4 Meningitec MENTEC 3 Menitorix MNTRX 3 Menjugate MENJUG 3 MMR II MMRCSL 3 Neis Vac-C NEISVC 3 Oral Polio (Polio Sabin) OPV 4 Pediacel PDCL 4 PedvaxHIB PRPOMP 3 Poliacel PLCL 4 Prevenar 7 PRVNR 3 Prevenar 13 PRVTH 4 Priorix MMRSKB 3 Priorix-Tetra PRXTET 3 ProHIBit PRPD 4 ProQuad PROQAD 3 Quadracel QDCL 4 Rotarix ROTRIX 2 RotaTeq ROTTEQ 3 Synflorix SYNFLX 4 Version 9.0 Page 17 of 18
18 Tripacel TCL 4 Triple Antigen DTP 4 Varilrix VLRIX 1 Varivax VRVAX 1 Non-Standard Vaccines Vaccine/Brand Name Vaccine Code Agrippal AGRPAL 8 Max doses Avaxim AVAXM 3 BCG tuberculosis BCG 1 Fluarix FLRIX 8 Fluvax FLUVAX 8 Fluvax Junior FVXJNR 8 Fluvirin FLVRN 8 Generic Hepatitis A GNHPA 3 Generic Influenza GNFLU 8 Generic Japanese Encephalitis GNJEN 4 Havrix Junior HAVJ 3 Influvac INFLUV 8 JE-VAX JEVAX 4 Mencevax ACWY MENVAX 3 Menomune MENUME 3 Panvax PANVAX 8 Twinrix Junior HATWNJ 3 Vaqta Paediatric/Adolescent formulation HAVAQ 3 Vaxigrip VAXGRP 8 Vaxigrip Junior VGRJNR 8 Sub-Population Vaccines Vaccine/Brand Name Vaccine Code Pneumovax 23 PNEUMO 1 Queensland Providers Only Max doses Max Vaccine/Brand Name Vaccine Code doses ADT ADT 5 Engerix B adult ENGBA 3 HBVaxII (thiomersal free) HBVXII 3 HBVaxII adult HBVXA 3 Hepatitis B Immunoglobulin HBIG 1 Q fever vaccine QFEVER 1 Rabies IMOG 10ml RIGTEN 3 Rabies IMOG 2ml RIGTWO 9 Rabies vaccine HDCV 5 Tet-tox TET 3 If you have any queries with the above process, or experience any difficulty in setting up your Immunisation Schedule, please contact the Medtech Helpdesk on Version 9.0 Page 18 of 18
Medtech Evolution Immunisation Schedule. (July 2018)
 Medtech Evolution Immunisation Schedule (July 2018) These Instructions contain important information for all Medtech Evolution users. We suggest that these notes are filed safely for future reference Copyright
Medtech Evolution Immunisation Schedule (July 2018) These Instructions contain important information for all Medtech Evolution users. We suggest that these notes are filed safely for future reference Copyright
Release Notes Medtech32
 Release Notes Medtech32 Medicare Australia Online Vaccine Product Mapping Update (September 2016) These release notes contain important information for Medtech32 users. Please ensure that they are circulated
Release Notes Medtech32 Medicare Australia Online Vaccine Product Mapping Update (September 2016) These release notes contain important information for Medtech32 users. Please ensure that they are circulated
Enhanced immunisation schedule Victoria
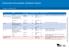 Children from March 2016 Age Disease Vaccine brand Birth Hepatitis B H-B-Vax-II Paediatric 2 months - from 6 weeks Diphtheria-tetanus-pertussis, poliomyelitis-hepatitis B- Reconstitute Site given Route
Children from March 2016 Age Disease Vaccine brand Birth Hepatitis B H-B-Vax-II Paediatric 2 months - from 6 weeks Diphtheria-tetanus-pertussis, poliomyelitis-hepatitis B- Reconstitute Site given Route
STANWOOD-CAMANO SCHOOL DISTRICT REQUEST FOR PART-TIME ATTENDANCE OR ANCILLARY SERVICES FROM PRIVATE SCHOOL PUPIL
 STANWOOD-CAMANO SCHOOL DISTRICT REQUEST FOR PART-TIME ATTENDANCE OR ANCILLARY SERVICES FROM PRIVATE SCHOOL PUPIL 114 F2 Students Please complete ALL information: Name of Pupil Grade Address of Pupil City
STANWOOD-CAMANO SCHOOL DISTRICT REQUEST FOR PART-TIME ATTENDANCE OR ANCILLARY SERVICES FROM PRIVATE SCHOOL PUPIL 114 F2 Students Please complete ALL information: Name of Pupil Grade Address of Pupil City
New Jersey Department of Health Vaccine Preventable Disease Program Childhood and Adolescent Recommended Vaccines
 New Jersey Department of Health Vaccine Preventable Disease Program Childhood and Adolescent Recommended Vaccines Antigens Vaccine Approved Age Daptacel Diphtheria, Tetanus, and acellular Pertussis (DTaP)
New Jersey Department of Health Vaccine Preventable Disease Program Childhood and Adolescent Recommended Vaccines Antigens Vaccine Approved Age Daptacel Diphtheria, Tetanus, and acellular Pertussis (DTaP)
TRICARE Retail Vaccination Program Vaccine List - September 2018*
 Diphtheria, tetanus and pertussis Diphtheria and tetanus toxoids adsorbed acellular pertussis adsorbed DT DTaP Diphtheria and tetanus toxoids adsorbed Daptacel, Infanrix Tetanus and diphtheria toxoids
Diphtheria, tetanus and pertussis Diphtheria and tetanus toxoids adsorbed acellular pertussis adsorbed DT DTaP Diphtheria and tetanus toxoids adsorbed Daptacel, Infanrix Tetanus and diphtheria toxoids
All Kindergarteners and 4-6 year old transfer students. 4 doses DTP or DTaP 1 dose must be at or after 4 years of age. None
 Parents Vaccines Required for School Entry in Michigan Whenever children are brought into group settings, there is a chance for diseases to spread. Children must follow state vaccine laws in order to attend
Parents Vaccines Required for School Entry in Michigan Whenever children are brought into group settings, there is a chance for diseases to spread. Children must follow state vaccine laws in order to attend
Twinrix Combined Hep A and B
 Twinrix Combined Hep A and B Cost $70.00 per injection. 3 required Standard course: 0, 1, 6 months Blood test recommended after course complete site of injection Vaqta Hepatitis A Vaccine Adults: cost
Twinrix Combined Hep A and B Cost $70.00 per injection. 3 required Standard course: 0, 1, 6 months Blood test recommended after course complete site of injection Vaqta Hepatitis A Vaccine Adults: cost
PREVENTIVE IMMUNIZATIONS. PREVENTIVE IMMUNIZATIONS These codes do not have a diagnosis code requirement for preventive benefits to apply.
 An immunization that does not fall under one of the exclusions in the Certificate of Coverage is considered covered after the following conditions are satisfied: (1) FDA approval; (2) explicit ACIP recommendation
An immunization that does not fall under one of the exclusions in the Certificate of Coverage is considered covered after the following conditions are satisfied: (1) FDA approval; (2) explicit ACIP recommendation
I certify that the information provided on this form is correct and verifiable. Month Day Year
 Certificate of Immunization Status (CIS) DOH 48-0 January 05 Please print. See back for instructions on how to fill out this form or get it printed from the Immunization Information System. Child s Last
Certificate of Immunization Status (CIS) DOH 48-0 January 05 Please print. See back for instructions on how to fill out this form or get it printed from the Immunization Information System. Child s Last
Immunisation schedule Victoria
 Infants and children July 2018 Birth H-B-Vax-II Paediatric or Paediatric 2 months (from 6 weeks) and 4 months Diphtheria-tetanus-pertussis, poliomyelitis-hepatitis B- Haemophilus influenzae type b Anterolateral
Infants and children July 2018 Birth H-B-Vax-II Paediatric or Paediatric 2 months (from 6 weeks) and 4 months Diphtheria-tetanus-pertussis, poliomyelitis-hepatitis B- Haemophilus influenzae type b Anterolateral
Coverage of Vaccines Medicaid and Child Health Plus Members
 Coverage of Vaccines Medicaid and Child Health Plus Members For children between the ages 0-18, routine recommended vaccinations are covered through Vaccines for Children program [VFC]. Fidelis Care will
Coverage of Vaccines Medicaid and Child Health Plus Members For children between the ages 0-18, routine recommended vaccinations are covered through Vaccines for Children program [VFC]. Fidelis Care will
Immunisation Policy. Country Children s Early Learning Ph: M:
 Immunisation Policy Published June 2018 Review Date December 2019 Sources Education and Care Services National Regulations, October 2017 Guide to the Education and Care Services National Law and the Education
Immunisation Policy Published June 2018 Review Date December 2019 Sources Education and Care Services National Regulations, October 2017 Guide to the Education and Care Services National Law and the Education
WVSIIS Vaccine Type Cheat Sheet Updated June 2010
 DTaP / DTP / DT / Td / Tdap Combo Vaccines Comvax HepB / Hib Merck (MSD) 1996 - Present 90748 Kinrix DTaP / IPV GlaxoSmithKline (SKB) 2008 - Present 90696 Pediarix DTaP / HepB / IPV GlaxoSmithKline (SKB)
DTaP / DTP / DT / Td / Tdap Combo Vaccines Comvax HepB / Hib Merck (MSD) 1996 - Present 90748 Kinrix DTaP / IPV GlaxoSmithKline (SKB) 2008 - Present 90696 Pediarix DTaP / HepB / IPV GlaxoSmithKline (SKB)
Advisory Committee on Immunization Practices VACCINE ACRONYMS
 Vaccine Acronyms Page 1 of 5 Advisy Committee on Immunization Practices VACCINE ACRONYMS Vaccines Included in the Immunization Schedules f Children, Adolescents, and Adults Following is a table of standardized
Vaccine Acronyms Page 1 of 5 Advisy Committee on Immunization Practices VACCINE ACRONYMS Vaccines Included in the Immunization Schedules f Children, Adolescents, and Adults Following is a table of standardized
PREVENTIVE IMMUNIZATIONS. PREVENTIVE IMMUNIZATIONS These codes do not have a diagnosis code requirement for preventive benefits to apply.
 An immunization that does not fall under one of the exclusions in the Certificate of Coverage is considered covered after the following conditions are satisfied: (1) FDA approval; (2) explicit ACIP recommendation
An immunization that does not fall under one of the exclusions in the Certificate of Coverage is considered covered after the following conditions are satisfied: (1) FDA approval; (2) explicit ACIP recommendation
GENERAL IMMUNIZATION GUIDE FOR CHILDCARE PROVIDERS August 2018 **CHILD VACCINES** DIPHTHERIA, TETANUS, PERTUSSIS VACCINES
 GENERAL IMMUNIZATION GUIDE FOR CHILDCARE PROVIDERS August 2018 **CHILD VACCINES** DIPHTHERIA, TETANUS, PERTUSSIS VACCINES DTaP: Diphtheria, Tetanus, acellular Pertussis Vaccine Infanrix Licensed in 1997
GENERAL IMMUNIZATION GUIDE FOR CHILDCARE PROVIDERS August 2018 **CHILD VACCINES** DIPHTHERIA, TETANUS, PERTUSSIS VACCINES DTaP: Diphtheria, Tetanus, acellular Pertussis Vaccine Infanrix Licensed in 1997
EARLY CHILDHOOD DEVELOPMENT & EDUCATION
 A division of Pawsitively Bassett, Inc. EARLY CHILDHOOD DEVELOPMENT & EDUCATION 08-09 67 N. Monroe St., Spokane WA 9908 www.nlcpreschool.com (509) 8-00 Dear Parents, Thank you, for your interest in the
A division of Pawsitively Bassett, Inc. EARLY CHILDHOOD DEVELOPMENT & EDUCATION 08-09 67 N. Monroe St., Spokane WA 9908 www.nlcpreschool.com (509) 8-00 Dear Parents, Thank you, for your interest in the
Advisory Committee on Immunization Practices VACCINE ACRONYMS
 May 1, 2015 Vaccine Acronyms Page 1 of 5 Advisy Committee on Immunization Practices VACCINE ACRONYMS Vaccines Included in the Immunization Schedules f Children, Adolescents, and Adults Following is a table
May 1, 2015 Vaccine Acronyms Page 1 of 5 Advisy Committee on Immunization Practices VACCINE ACRONYMS Vaccines Included in the Immunization Schedules f Children, Adolescents, and Adults Following is a table
Victorian immunisation catch-up tool for 10 to 19 year olds
 Victorian immunisation catch-up tool for 10 to 19 year olds January 2018 Guidelines for 10 to 19 year olds with no documented history of vaccine administration The objective of catch-up vaccination is
Victorian immunisation catch-up tool for 10 to 19 year olds January 2018 Guidelines for 10 to 19 year olds with no documented history of vaccine administration The objective of catch-up vaccination is
CVU: What s new? 2 nd December 2013
 CVU: What s new? 2 nd December 2013 Dr Margie Danchin Paediatrician, Department of General Medicine, RCH Senior Research Fellow, MCRI Senior Fellow, Department of Paediatrics, The University of Melbourne
CVU: What s new? 2 nd December 2013 Dr Margie Danchin Paediatrician, Department of General Medicine, RCH Senior Research Fellow, MCRI Senior Fellow, Department of Paediatrics, The University of Melbourne
Vaccination Decision Making: What Providers Need to Know
 Objectives Vaccination Decision Making: What Providers Need to Know Kelli Smith, RN, BSN Iowa Department of Public Health Immunization Program Catch-up Schedule: how to most efficiently bring persons up-to-date
Objectives Vaccination Decision Making: What Providers Need to Know Kelli Smith, RN, BSN Iowa Department of Public Health Immunization Program Catch-up Schedule: how to most efficiently bring persons up-to-date
Early Learning Centre Immunisation Policy Legislation ACT Public Health Regulations (2000)
 Early Learning Centre Immunisation Policy Legislation ACT Public Health Regulations (2000) National Quality Standard / Education and Care Services National Regulations Standard 2.1 Each child s health
Early Learning Centre Immunisation Policy Legislation ACT Public Health Regulations (2000) National Quality Standard / Education and Care Services National Regulations Standard 2.1 Each child s health
State of Alaska Child Care & School Immunization Requirements Packet Revised 08/2010
 State of Alaska Child Care & School Immunization Requirements Packet Revised 08/2010 ALASKA IMMUNIZATION PROGRAM (907) 269-8000 OR 1-888-430-4321 http://www.epi.hss.state.ak.us/id/immune.stm (This page
State of Alaska Child Care & School Immunization Requirements Packet Revised 08/2010 ALASKA IMMUNIZATION PROGRAM (907) 269-8000 OR 1-888-430-4321 http://www.epi.hss.state.ak.us/id/immune.stm (This page
Primary Health Care forum August 2015 Sue Casey
 Primary Health Care forum August 2015 Sue Casey A needs analysis of catch-up immunisation in refugee background and asylum seeker communities in Victoria Authors: Dr Georgia Paxton Dr Hamish Graham Peter
Primary Health Care forum August 2015 Sue Casey A needs analysis of catch-up immunisation in refugee background and asylum seeker communities in Victoria Authors: Dr Georgia Paxton Dr Hamish Graham Peter
Victorian immunisation catch-up tool for 10 to 19 year olds
 Victorian immunisation catch-up tool for 10 to 19 year olds For healthy children with no documented history of vaccine administration The objective of catch-up vaccination is to complete a course of age
Victorian immunisation catch-up tool for 10 to 19 year olds For healthy children with no documented history of vaccine administration The objective of catch-up vaccination is to complete a course of age
Vaccine Label Examples
 Vaccine Label Examples With the large amount of vaccine carried in most clinics, staff can easily become confused about vaccines within the storage unit. Labeling the area where vaccines are stored can
Vaccine Label Examples With the large amount of vaccine carried in most clinics, staff can easily become confused about vaccines within the storage unit. Labeling the area where vaccines are stored can
DATE: & 5, 2015 SUBJECT:
 TO: Immunization Program Directors of Licensed Child Day Care Centers & Group Day Care Homes FROM: Mick Bolduc-Epidemiologist, Immunization Program DATE: January 5, 2015 SUBJECT: Annual Immunization Survey
TO: Immunization Program Directors of Licensed Child Day Care Centers & Group Day Care Homes FROM: Mick Bolduc-Epidemiologist, Immunization Program DATE: January 5, 2015 SUBJECT: Annual Immunization Survey
Release Notes Medtech Evolution General Practice
 Release Notes Medtech Evolution General Practice Version 1.9 Build 1.9.0.15 (June 2017) These release notes contain important information for Medtech Evolution users. Please ensure that they are circulated
Release Notes Medtech Evolution General Practice Version 1.9 Build 1.9.0.15 (June 2017) These release notes contain important information for Medtech Evolution users. Please ensure that they are circulated
National Immunisation News
 July 2015 National Immunisation News The newsletter of the HSE National Immunisation Office CONTENTS Changes to the Primary Childhood Immunisation Programme BCG Shortage Pertussis Vaccine Mumps Order Vaccines
July 2015 National Immunisation News The newsletter of the HSE National Immunisation Office CONTENTS Changes to the Primary Childhood Immunisation Programme BCG Shortage Pertussis Vaccine Mumps Order Vaccines
Detailed cost of vaccination/ Immunization in India (Birth to 10 years of age)
 Detailed cost of vaccination/ Immunization in India (Birth to 10 years of age) Glossary: IPV= Injectable Polio vaccine OPV= Oral polio vaccine (these are oral drops, which is also given in "Pulse Polio
Detailed cost of vaccination/ Immunization in India (Birth to 10 years of age) Glossary: IPV= Injectable Polio vaccine OPV= Oral polio vaccine (these are oral drops, which is also given in "Pulse Polio
Childhood Immunisations Template Guide 2017
 Childhood Immunisations Template Guide 2017 1 Template Control Page Childhood Immunisations Template Guide Title Childhood Immunisations Author CEG Description Supports the schedule of Childhood Immunisations
Childhood Immunisations Template Guide 2017 1 Template Control Page Childhood Immunisations Template Guide Title Childhood Immunisations Author CEG Description Supports the schedule of Childhood Immunisations
immunisation in New Zealand
 This appendix details the history of. Section A1.1 is a brief summary of when each vaccine was introduced to the National Immunisation Schedule (the Schedule). This summary includes vaccines which were
This appendix details the history of. Section A1.1 is a brief summary of when each vaccine was introduced to the National Immunisation Schedule (the Schedule). This summary includes vaccines which were
Vaccine Label Examples
 Vaccine Label Examples With the large amount of vaccine carried in most clinics, staff can easily become confused about vaccines within the storage unit. Labeling the area where vaccines are stored can
Vaccine Label Examples With the large amount of vaccine carried in most clinics, staff can easily become confused about vaccines within the storage unit. Labeling the area where vaccines are stored can
Immunization Program. Directors of Licensed Child Day Care Centers & Group Day Care Homes. FROM: Mick Bolduc-Epidemiologist Debra L.
 Immunization Program TO: Directors of Licensed Child Day Care Centers & Group Day Care Homes FROM: Mick Bolduc-Epidemiologist Debra L. Johnson-Chief Immunization Program Community Based Regulation Section
Immunization Program TO: Directors of Licensed Child Day Care Centers & Group Day Care Homes FROM: Mick Bolduc-Epidemiologist Debra L. Johnson-Chief Immunization Program Community Based Regulation Section
SKAGIT VALLEY FAMILY YMCA
 Central Preschool Registration 2019-2020 Child s First : MI: Last : Address: City: State: Zip Home Phone: ( ) Gender: Birth : Age: Child lives w/ Mother/Guardian: Cell Phone: ( ) Employer: Work Phone:
Central Preschool Registration 2019-2020 Child s First : MI: Last : Address: City: State: Zip Home Phone: ( ) Gender: Birth : Age: Child lives w/ Mother/Guardian: Cell Phone: ( ) Employer: Work Phone:
Refer to the PCS Clinical Audit Tool Quick Reference Guide for installation instructions and an overview of CAT features.
 Getting Started Installation Refer to the PCS Clinical Audit Tool Quick Reference Guide for installation instructions and an overview of CAT features. CAT Immunisations > Childhood Schedule Graph and Worksheet
Getting Started Installation Refer to the PCS Clinical Audit Tool Quick Reference Guide for installation instructions and an overview of CAT features. CAT Immunisations > Childhood Schedule Graph and Worksheet
INFECTIOUS DISEASES PROCEDURE
 INFECTIOUS DISEASES PROCEDURE Policy Hierarchy link Responsible Officer Contact Officer Superseded Documents Associated Documents Children (Education and Care Services National Law Application) Act 2010
INFECTIOUS DISEASES PROCEDURE Policy Hierarchy link Responsible Officer Contact Officer Superseded Documents Associated Documents Children (Education and Care Services National Law Application) Act 2010
14: Immunological products and vaccines
 File name: Immunological Products and s Formulary BNF Original Date of issue: 30/10/2005 Last Reviewed: 23/11/2012 Version:4 Page 1 of 6 14: Immunological products and vaccines 14.1 Active immunity Live
File name: Immunological Products and s Formulary BNF Original Date of issue: 30/10/2005 Last Reviewed: 23/11/2012 Version:4 Page 1 of 6 14: Immunological products and vaccines 14.1 Active immunity Live
Release Notes. Medtech Evolution General Practice. Version Build (June 2016)
 Release Notes Medtech Evolution General Practice Version 1.5.7 Build 1.5.7.72 (June 2016) These release notes contain important information for Medtech Evolution users. Please ensure that they are circulated
Release Notes Medtech Evolution General Practice Version 1.5.7 Build 1.5.7.72 (June 2016) These release notes contain important information for Medtech Evolution users. Please ensure that they are circulated
K 6 th Express Billing Contract
 2017-2018 K 6 th Express Billing Contract Child Information: First Middle Initial Last Parent/Guardian Information: First Middle Initial Last Address Phone Requested Start Will you be receiving state/other
2017-2018 K 6 th Express Billing Contract Child Information: First Middle Initial Last Parent/Guardian Information: First Middle Initial Last Address Phone Requested Start Will you be receiving state/other
Armed Services Blood Program Immunization List
 Immunization List NOTICE: The Department of Defense (DoD) assumes no risk for the use of this information by non-dod personnel, blood programs, or individual medical institutions. The use of this information
Immunization List NOTICE: The Department of Defense (DoD) assumes no risk for the use of this information by non-dod personnel, blood programs, or individual medical institutions. The use of this information
National Immunisation News The newsletter of the HSE National Immunisation Office
 February 2015 National Immunisation News The newsletter of the HSE National Immunisation Office CONTENTS Changes to the PCI programme Uptake statistics Pneumovax name change Flu season Common queries School
February 2015 National Immunisation News The newsletter of the HSE National Immunisation Office CONTENTS Changes to the PCI programme Uptake statistics Pneumovax name change Flu season Common queries School
RE: 2017 Immunization Requirements for Children Attending Michigan Preschool Programs or Licensed Childcare Centers
 RICK SNYDER GOVERNOR STATE OF MICHIGAN DEPARTMENT OF HEAL TH AND HUMAN SERVICES LANSING NICK LYON DIRECTOR April 24, 2017 RE: 2017 Immunization Requirements for Children Attending Michigan Preschool Programs
RICK SNYDER GOVERNOR STATE OF MICHIGAN DEPARTMENT OF HEAL TH AND HUMAN SERVICES LANSING NICK LYON DIRECTOR April 24, 2017 RE: 2017 Immunization Requirements for Children Attending Michigan Preschool Programs
Download CoCASA Software Application
 Comprehensive Clinic Assessment Software Application (CoCASA) 7.0 Instructions Guide for Immunization Service Contractors February 6, 2012 The Comprehensive Clinic Assessment Software Application (CoCASA)
Comprehensive Clinic Assessment Software Application (CoCASA) 7.0 Instructions Guide for Immunization Service Contractors February 6, 2012 The Comprehensive Clinic Assessment Software Application (CoCASA)
Public Statement: Medical Policy. Effective Date: 01/01/2012 Revision Date: 03/24/2014 Code(s): Many. Document: ARB0454:04.
 ARBenefits Approval: 01/01/2012 Effective Date: 01/01/2012 Revision Date: 03/24/2014 Code(s): Many Medical Policy Title: Immunization Coverage Document: ARB0454:04 Administered by: Public Statement: 1.
ARBenefits Approval: 01/01/2012 Effective Date: 01/01/2012 Revision Date: 03/24/2014 Code(s): Many Medical Policy Title: Immunization Coverage Document: ARB0454:04 Administered by: Public Statement: 1.
Child Care Registration Form
 Child Care Registration Form child entered care Child s name Last First Middle Name (Nickname) used Birthdate Street address City Zip code Child s parent/guardian name home phone # cell phone# child left
Child Care Registration Form child entered care Child s name Last First Middle Name (Nickname) used Birthdate Street address City Zip code Child s parent/guardian name home phone # cell phone# child left
Kadina Child Care Centre Enrolment Form
 Court Orders Are there any court orders (including Parenting Orders and Parent Plans) relating to the powers and responsibilites of the parents in relation to the child or access to the child? No Yes go
Court Orders Are there any court orders (including Parenting Orders and Parent Plans) relating to the powers and responsibilites of the parents in relation to the child or access to the child? No Yes go
State of Alaska CHILD CARE & SCHOOL IMMUNIZATION REQUIREMENTS Packet Revised 06/2017
 State of Alaska CHILD CARE & SCHOOL IMMUNIZATION REQUIREMENTS Packet Revised 06/2017 ALASKA IMMUNIZATION PROGRAM (907) 269-8088 OR 1-888-430-4321 http://www.epi.alaska.gov/immunize Updates in this packet
State of Alaska CHILD CARE & SCHOOL IMMUNIZATION REQUIREMENTS Packet Revised 06/2017 ALASKA IMMUNIZATION PROGRAM (907) 269-8088 OR 1-888-430-4321 http://www.epi.alaska.gov/immunize Updates in this packet
Release Notes. Medtech32. Version Build 3347 Update HbA1c Laboratory Data Format Change Advanced Forms Licensing Renewal (September 2011)
 Release Notes Medtech32 Version 19.11 Build 3347 Update HbA1c Laboratory Data Format Change Advanced Forms Licensing Renewal (September 2011) IMPORTANT NOTE Medtech recommends that all Medtech upgrades
Release Notes Medtech32 Version 19.11 Build 3347 Update HbA1c Laboratory Data Format Change Advanced Forms Licensing Renewal (September 2011) IMPORTANT NOTE Medtech recommends that all Medtech upgrades
Michigan Care Improvement Registry (MCIR) Meaningful Use Follow Up Submission/ Quality Assurance Testing Guide
 MCIR Meaningful Use Follow Up Submission/Quality Assurance Testing Guide Michigan Care Improvement Registry (MCIR) Meaningful Use Follow Up Submission/ Quality Assurance Testing Guide Document Description
MCIR Meaningful Use Follow Up Submission/Quality Assurance Testing Guide Michigan Care Improvement Registry (MCIR) Meaningful Use Follow Up Submission/ Quality Assurance Testing Guide Document Description
Approval / Acknowledgement Letter Ref No. / Diary no F.No.12-48/GSK/PAC- Synflorix/15-BD 25-Mar-15 2 GlaxoSmithKline Pharmaceuticals Limited.
 1 GlaxoSmithKline Deletion of a n-significant in-process test limit during the purification process of the tetanus / diphtheria toxoids used in the manufacture of GSK's PCV vaccine [brand name: Synflorix]
1 GlaxoSmithKline Deletion of a n-significant in-process test limit during the purification process of the tetanus / diphtheria toxoids used in the manufacture of GSK's PCV vaccine [brand name: Synflorix]
Yukon Immunization Program Manual ADMENDMENTS & ADDITIONS
 2012 August Page 1 Manual ADMENDMENTS & ADDITIONS Number & 11-01 2011 June 09 11-02 2011 October 11 12-01 2012 Feb 9 Topic Additions to Yukon Immunization Program Manual Twinrix, Twinrix Junior, Menamune,
2012 August Page 1 Manual ADMENDMENTS & ADDITIONS Number & 11-01 2011 June 09 11-02 2011 October 11 12-01 2012 Feb 9 Topic Additions to Yukon Immunization Program Manual Twinrix, Twinrix Junior, Menamune,
Childhood Immunisations Template Guide 2016
 Childhood Immunisations Template Guide 2016 1 Template Control Page Childhood Immunisations Template Guide Title Childhood Immunisations Author CEG Version 9 Description Supports the schedule of Childhood
Childhood Immunisations Template Guide 2016 1 Template Control Page Childhood Immunisations Template Guide Title Childhood Immunisations Author CEG Version 9 Description Supports the schedule of Childhood
National Immunisation News
 National Immunisation News The newsletter of the HSE National Immunisation Office July 2016 Changes to the Primary Childhood Immunisation Programme The National Immunisation Advisory Committee (NIAC) has
National Immunisation News The newsletter of the HSE National Immunisation Office July 2016 Changes to the Primary Childhood Immunisation Programme The National Immunisation Advisory Committee (NIAC) has
Syrian Programme Refugees Advice on assessment of immunisation status and recommendations for additional immunisation
 Syrian Programme Refugees Advice on assessment of immunisation status and recommendations for additional immunisation Background Information Immunisation services in Syria Syria had good immunisation services,
Syrian Programme Refugees Advice on assessment of immunisation status and recommendations for additional immunisation Background Information Immunisation services in Syria Syria had good immunisation services,
State of Alaska CHILD CARE & SCHOOL IMMUNIZATION REQUIREMENTS Packet
 State of Alaska CHILD CARE & SCHOOL IMMUNIZATION REQUIREMENTS Packet Revised 6/2013 ALASKA IMMUNIZATION PROGRAM (907) 269-8088 OR 1-888-430-4321 http://www.epi.alaska.gov/immunize (This page intentionally
State of Alaska CHILD CARE & SCHOOL IMMUNIZATION REQUIREMENTS Packet Revised 6/2013 ALASKA IMMUNIZATION PROGRAM (907) 269-8088 OR 1-888-430-4321 http://www.epi.alaska.gov/immunize (This page intentionally
Hexavalent Vaccines: Hepatitis B antibody response and co-administration with other vaccines
 Viral Hepatitis Prevention Board Hanoi, 25 26 July 2018 Hexavalent Vaccines: Hepatitis B antibody response and co-administration with other vaccines Prof. Timo Vesikari Vaccine Research Center University
Viral Hepatitis Prevention Board Hanoi, 25 26 July 2018 Hexavalent Vaccines: Hepatitis B antibody response and co-administration with other vaccines Prof. Timo Vesikari Vaccine Research Center University
K 6 th Express Billing Contract
 05-06 K 6 th Express Billing Contract Child Information: First Middle Initial Last Parent/Guardian Information: First Middle Initial Last Address Phone Requested Start Will you be receiving state/other
05-06 K 6 th Express Billing Contract Child Information: First Middle Initial Last Parent/Guardian Information: First Middle Initial Last Address Phone Requested Start Will you be receiving state/other
2018 Immunisation Update
 2018 Immunisation Update A puzzling time?!! Sometimes the hardest pieces of a puzzle to assemble, are the ones missing from the box. Dixie Waters, Author The Puzzle Pieces Immunisation coverage rates NSW
2018 Immunisation Update A puzzling time?!! Sometimes the hardest pieces of a puzzle to assemble, are the ones missing from the box. Dixie Waters, Author The Puzzle Pieces Immunisation coverage rates NSW
Changes to the National Immunisation Schedule
 17 December 2013 Changes to the National Immunisation Schedule PHARMAC is pleased to announce decisions related to the National Immunisation Schedule (NIS) that will take effect from 1 July 2014. This
17 December 2013 Changes to the National Immunisation Schedule PHARMAC is pleased to announce decisions related to the National Immunisation Schedule (NIS) that will take effect from 1 July 2014. This
National Pandemic Vaccination Programme
 March 2010 National Pandemic Vaccination Programme Swine Flu Vaccination Programme Pneumococcal Catch-up Campaign Measles Outbreak National Immunisation Guidelines NIO Website HPV Vaccine Campaign Vaccine
March 2010 National Pandemic Vaccination Programme Swine Flu Vaccination Programme Pneumococcal Catch-up Campaign Measles Outbreak National Immunisation Guidelines NIO Website HPV Vaccine Campaign Vaccine
Copyright regulations Warning
 COMMONWEALTH OF AUSTRALIA Copyright regulations 1969 Warning This material has been reproduced and communicated to you by or on behalf of the University of Melbourne pursuant to part VB of the Copyright
COMMONWEALTH OF AUSTRALIA Copyright regulations 1969 Warning This material has been reproduced and communicated to you by or on behalf of the University of Melbourne pursuant to part VB of the Copyright
National Immunisation NEWS The newsletter of the National Immunisation Office Directorate of Population Health
 Page 1 of 8 National Immunisation News February 2008 National Immunisation NEWS The newsletter of the National Immunisation Office Directorate of Population Health 2008: Issue 5 HSE National Cold Chain
Page 1 of 8 National Immunisation News February 2008 National Immunisation NEWS The newsletter of the National Immunisation Office Directorate of Population Health 2008: Issue 5 HSE National Cold Chain
THE AFIX PRODUCT TRAINING MANUAL
 THE AFIX PRODUCT TRAINING MANUAL Last Updated: 11/30/2018 Table of Contents The AFIX Product End User Training AFIX Cohort. 4 Provider Selection... 4 Assessment Selection..... 7 Reports.......10 Flexible
THE AFIX PRODUCT TRAINING MANUAL Last Updated: 11/30/2018 Table of Contents The AFIX Product End User Training AFIX Cohort. 4 Provider Selection... 4 Assessment Selection..... 7 Reports.......10 Flexible
Role of Partnerships in Developing Innovative Vaccines: Brazil
 Role of Partnerships in Developing Innovative Vaccines: Brazil SAGE Meeting, October 2010 Reinaldo Guimarães, M.D, MSc. Secretary of Science, Technology and Strategic Inputs Ministry of Health of Brazil
Role of Partnerships in Developing Innovative Vaccines: Brazil SAGE Meeting, October 2010 Reinaldo Guimarães, M.D, MSc. Secretary of Science, Technology and Strategic Inputs Ministry of Health of Brazil
Modern Drug House.
 +91-8048941768 Modern Drug House https://www.moderndrughouse.in/ We are one of the leading suppliers and traders of a wide range of Vaccine and Pharmaceutical Products. Owing to its effectiveness, purity
+91-8048941768 Modern Drug House https://www.moderndrughouse.in/ We are one of the leading suppliers and traders of a wide range of Vaccine and Pharmaceutical Products. Owing to its effectiveness, purity
How Immunisations work in Best Practice?
 How Immunisations work in Best Practice? There are a number of areas in Best Practice related to Immunisations:- Recording and updating Immunisation records Searching and Printing Immunisations List Printing
How Immunisations work in Best Practice? There are a number of areas in Best Practice related to Immunisations:- Recording and updating Immunisation records Searching and Printing Immunisations List Printing
Current National Immunisation Schedule Dr Brenda Corcoran National Immunisation Office.
 Current National Immunisation Schedule 2012 Dr Brenda Corcoran National Immunisation Office Objectives To outline immunisation schedules in Ireland Primary childhood schedule Vaccine uptake rates School
Current National Immunisation Schedule 2012 Dr Brenda Corcoran National Immunisation Office Objectives To outline immunisation schedules in Ireland Primary childhood schedule Vaccine uptake rates School
Guidelines for the immunisation of children following treatment with Standard-Dose Chemotherapy
 Guidelines for the immunisation of children following treatment with Standard-Dose Chemotherapy Version 2.0 Approved by Haem / Onc Senior Clinical Management team Date Approved March 2015 Ratified by:
Guidelines for the immunisation of children following treatment with Standard-Dose Chemotherapy Version 2.0 Approved by Haem / Onc Senior Clinical Management team Date Approved March 2015 Ratified by:
Routine Immunization Schedules. Section 2. Newfoundland and Labrador Immunization Manual. Routine Immunization Schedules
 Newfoundland and Labrador Immunization Manual Section 2 Routine Immunization Schedules Routine Immunization Schedules... 2.1-1 Policy on Routine Immunization Schedules... 2.1-2 Routine and Delayed Immunization
Newfoundland and Labrador Immunization Manual Section 2 Routine Immunization Schedules Routine Immunization Schedules... 2.1-1 Policy on Routine Immunization Schedules... 2.1-2 Routine and Delayed Immunization
BabyJabs Vaccines. All vaccines are mercury- free We use aluminium- free vaccines wherever possible
 BabyJabs Vaccines All vaccines are mercury- free We use aluminium- free vaccines wherever possible BabyJabs is a dedicated children s immunisation service, offering a choice of single and small combination
BabyJabs Vaccines All vaccines are mercury- free We use aluminium- free vaccines wherever possible BabyJabs is a dedicated children s immunisation service, offering a choice of single and small combination
1.0 ROUTINE SCHEDULES...
 August 2012 TABLE OF CONTENTS 1.0 ROUTINE SCHEDULES... 1 1.1 SCHEDULE A: BASIC IMMUNIZATION WHEN STARTING WITH INFANRIX HEXA VACCINE... 1 1.1.1 SCHEDULE A: BASIC IMMUNIZATION WHEN STARTING WITH PEDIACEL
August 2012 TABLE OF CONTENTS 1.0 ROUTINE SCHEDULES... 1 1.1 SCHEDULE A: BASIC IMMUNIZATION WHEN STARTING WITH INFANRIX HEXA VACCINE... 1 1.1.1 SCHEDULE A: BASIC IMMUNIZATION WHEN STARTING WITH PEDIACEL
Appendix An Assessment Tool to Determine the Validity of Vaccine Doses
 Appendix 4.4 - An Assessment Tool to Determine the Validity of Vaccine Doses Note: Refer to the Canadian Immunization Guide and New Brunswick (NB) immunization program directives for recommendations for
Appendix 4.4 - An Assessment Tool to Determine the Validity of Vaccine Doses Note: Refer to the Canadian Immunization Guide and New Brunswick (NB) immunization program directives for recommendations for
Immunise your child on time. It s their best protection
 Immunise your child on time It s their best protection If you are a parent or caregiver, this pamphlet is for you. It tells you about the recommended ages for your baby to receive their immunisations.
Immunise your child on time It s their best protection If you are a parent or caregiver, this pamphlet is for you. It tells you about the recommended ages for your baby to receive their immunisations.
Return those vaccines to the storage unit and repeat for all VFC vaccines before completing the rest of this worksheet.
 Instructions: Complete the worksheet that matches your provider category (e.g., low-, medium-, or high-volume) before submitting routine vaccine orders. Refer to the VFC Program Provider Operations Manual
Instructions: Complete the worksheet that matches your provider category (e.g., low-, medium-, or high-volume) before submitting routine vaccine orders. Refer to the VFC Program Provider Operations Manual
National Immunisation News The newsletter of the HSE National Immunisation Office
 February 2014 National Immunisation News The newsletter of the HSE National Immunisation Office CONTENTS Adverse Local Reactions following 4 in 1 booster 2013 Immunisation Guidelines for Ireland Flu Season
February 2014 National Immunisation News The newsletter of the HSE National Immunisation Office CONTENTS Adverse Local Reactions following 4 in 1 booster 2013 Immunisation Guidelines for Ireland Flu Season
VACCINATION. DR.FATIMA ALKHALEDY M.B.Ch.B;F.I.C.M.S/C.M.
 VACCINATION DR.FATIMA ALKHALEDY M.B.Ch.B;F.I.C.M.S/C.M. IMMUNIZATION Immunization is defined as the procedure by which the body is prepared to fight against a specific disease. It is used to induce the
VACCINATION DR.FATIMA ALKHALEDY M.B.Ch.B;F.I.C.M.S/C.M. IMMUNIZATION Immunization is defined as the procedure by which the body is prepared to fight against a specific disease. It is used to induce the
BabyJabs Vaccines. All vaccines are mercury-free We use aluminium-free vaccines wherever possible
 BabyJabs Vaccines All vaccines are mercury-free We use aluminium-free vaccines wherever possible BabyJabs is a dedicated children s immunisation service, offering a choice of single and small combination
BabyJabs Vaccines All vaccines are mercury-free We use aluminium-free vaccines wherever possible BabyJabs is a dedicated children s immunisation service, offering a choice of single and small combination
BabyJabs Vaccines. All vaccines are mercury-free We use aluminium-free vaccines wherever possible
 BabyJabs Vaccines All vaccines are mercury-free We use aluminium-free vaccines wherever possible BabyJabs is a dedicated children s immunisation service, offering a choice of single and small combination
BabyJabs Vaccines All vaccines are mercury-free We use aluminium-free vaccines wherever possible BabyJabs is a dedicated children s immunisation service, offering a choice of single and small combination
CHILDHOOD VACCINATION
 EPI (3) Age of Child How and Where is it given? CHILDHOOD VACCINATION Nicolette du Plessis Block 10 28/02/2012 10 weeks DTaP-IPV/Hib (2) Diphtheria, Tetanus, Acellular pertussis, Inactivated polio vaccine,
EPI (3) Age of Child How and Where is it given? CHILDHOOD VACCINATION Nicolette du Plessis Block 10 28/02/2012 10 weeks DTaP-IPV/Hib (2) Diphtheria, Tetanus, Acellular pertussis, Inactivated polio vaccine,
Routine Immunization Schedules. Section 2. Newfoundland and Labrador Immunization Manual. Routine Immunization Schedules
 Newfoundland and Labrador Immunization Manual Section 2... 2.1 Routine and Delayed Immunization Schedules for Infants and Children... 2.2 Recommended Immunizations for Adults... 2.3 (Provinces and Territories)...
Newfoundland and Labrador Immunization Manual Section 2... 2.1 Routine and Delayed Immunization Schedules for Infants and Children... 2.2 Recommended Immunizations for Adults... 2.3 (Provinces and Territories)...
Immunizations are among the most cost effective and widely used public health interventions.
 Focused Issue of This Month Recommended by the Korean Pediatric Society, 2008 Hoan Jong Lee, MD Department of Pediatrics, Seoul National University College of Medicine E mail : hoanlee@snu.ac.kr J Korean
Focused Issue of This Month Recommended by the Korean Pediatric Society, 2008 Hoan Jong Lee, MD Department of Pediatrics, Seoul National University College of Medicine E mail : hoanlee@snu.ac.kr J Korean
The hexavalent DTaP/IPV/Hib/HepB combination vaccine
 The hexavalent combination vaccine Information for registered healthcare practitioners about the neonatal selective immunisation programme for babies at high risk (babies born to hepatitis B positive mothers)
The hexavalent combination vaccine Information for registered healthcare practitioners about the neonatal selective immunisation programme for babies at high risk (babies born to hepatitis B positive mothers)
UNSCHEDULED VACCINATION OF CHILDREN AND YOUNG PEOPLE WHO HAVE OUTSTANDING ROUTINE IMMUNISATIONS. Service Specification
 UNSCHEDULED VACCINATION OF CHILDREN AND YOUNG PEOPLE WHO HAVE OUTSTANDING ROUTINE IMMUNISATIONS Service Specification National Enhanced Service Specification For The Unscheduled Vaccination Of Children
UNSCHEDULED VACCINATION OF CHILDREN AND YOUNG PEOPLE WHO HAVE OUTSTANDING ROUTINE IMMUNISATIONS Service Specification National Enhanced Service Specification For The Unscheduled Vaccination Of Children
Objectives. Immunity. Childhood Immunization Risk of Non-Vaccinated Children 12/22/2015
 Childhood Immunization Risk of Non-Vaccinated Children Bertha P. Rojas, Pharm.D. PGY-1 Pharmacy Resident South Miami Hospital Objectives Understand the definition of herd immunity Identify vaccine-preventable
Childhood Immunization Risk of Non-Vaccinated Children Bertha P. Rojas, Pharm.D. PGY-1 Pharmacy Resident South Miami Hospital Objectives Understand the definition of herd immunity Identify vaccine-preventable
Alaska School and Child Care Facility Immunization Manual
 Alaska School and Child Care Facility Immunization Manual Alaska Dept. of Health and Social Services For many years the backbone of Alaska s disease prevention efforts has been the appropriate immunization
Alaska School and Child Care Facility Immunization Manual Alaska Dept. of Health and Social Services For many years the backbone of Alaska s disease prevention efforts has been the appropriate immunization
Supported MCIR Vaccine Codes Including U.S. Licensed CVX, CPT-4, and MVX
 Supported MCIR Vaccine s Including U.S. Licensed CVX, CPT-4, and MVX Name Product Name Vaccine Name (CPT ) Adenovirus 4,7, live, Adenovirus types 4 Barr 143 Adenovirus types 4 and 7 BRR oral and 7 Laboratories
Supported MCIR Vaccine s Including U.S. Licensed CVX, CPT-4, and MVX Name Product Name Vaccine Name (CPT ) Adenovirus 4,7, live, Adenovirus types 4 Barr 143 Adenovirus types 4 and 7 BRR oral and 7 Laboratories
WELSH HEALTH CIRCULAR
 WELSH HEALTH CIRCULAR WHC (2017) 022 Issue Date: 12 May 2017 STATUS: ACTION CATEGORY: PUBLIC HEALTH Title: Change of vaccine for the routine primary infant immunisation Date of Expiry / Review N/A For
WELSH HEALTH CIRCULAR WHC (2017) 022 Issue Date: 12 May 2017 STATUS: ACTION CATEGORY: PUBLIC HEALTH Title: Change of vaccine for the routine primary infant immunisation Date of Expiry / Review N/A For
Healthy People 2020 objectives were released in 2010, with a 10-year horizon to achieve the goals by 2020.
 Appendix 1: Healthy People 2020 Immunization-related Objectives Healthy People provides science-based, 10-year national objectives for improving the health of all Americans. For three decades, Healthy
Appendix 1: Healthy People 2020 Immunization-related Objectives Healthy People provides science-based, 10-year national objectives for improving the health of all Americans. For three decades, Healthy
Whittier Kids Child Enrollment Form. Grade School Year: Child s Full Name: Birthdate: Nickname/Preferred Name: Gender: Preschool Program
 Whittier Kids Child Enrollment Form 2017-18 School Year Child s Full Name: Birthdate: Nickname/Preferred Name: Gender: School Age Program Preschool Program Grade 2017-18 School Year: Parent/Guardian Name:
Whittier Kids Child Enrollment Form 2017-18 School Year Child s Full Name: Birthdate: Nickname/Preferred Name: Gender: School Age Program Preschool Program Grade 2017-18 School Year: Parent/Guardian Name:
REACHING OUR GOALS: CHILDHOOD & ADOLESCENT IMMUNIZATION Illinois Chapter, American Academy of Pediatrics
 REACHING OUR GOALS: CHILDHOOD & ADOLESCENT IMMUNIZATION 2017 Illinois Chapter, American Academy of Pediatrics Learning Objectives Provide the updates from 2017 Advisory Committee on Immunizations Practices
REACHING OUR GOALS: CHILDHOOD & ADOLESCENT IMMUNIZATION 2017 Illinois Chapter, American Academy of Pediatrics Learning Objectives Provide the updates from 2017 Advisory Committee on Immunizations Practices
Selected vaccine introduction status into routine immunization
 Selected introduction status into routine infant immunization worldwide, 2003 This report summarizes the current status of national immunization schedules in 2003, as reported by Member States in the /UNICEF
Selected introduction status into routine infant immunization worldwide, 2003 This report summarizes the current status of national immunization schedules in 2003, as reported by Member States in the /UNICEF
Medtech Training Guide
 Medtech Training Guide Clinical Audit Tool Copyright Medtech Limited Page 1 of 80 Clinical Audit Tool v4.0 Contents Chapter 1: Getting Started... 3 Logging In... 3 Logging Out... 3 Setting the Preliminary
Medtech Training Guide Clinical Audit Tool Copyright Medtech Limited Page 1 of 80 Clinical Audit Tool v4.0 Contents Chapter 1: Getting Started... 3 Logging In... 3 Logging Out... 3 Setting the Preliminary
Vaccinations Outside Recommended Ages 2014; Six Immunization Information System Sentinel Sites
 National Center for Immunization & Respiratory Diseases Vaccinations Outside Recommended Ages 2014; Six Immunization Information System Sentinel Sites Loren Rodgers, Ph.D. CDC Immunization Information
National Center for Immunization & Respiratory Diseases Vaccinations Outside Recommended Ages 2014; Six Immunization Information System Sentinel Sites Loren Rodgers, Ph.D. CDC Immunization Information
Wherever possible it is recommended that a child receive their vaccination on a day when they will not be attending the centre..
 Policy Hierarchy link Responsible Officer Contact Officer Superseded Documents Children (Education and Care Services National Law Application) Act 2010 Education and Care Services National Regulations
Policy Hierarchy link Responsible Officer Contact Officer Superseded Documents Children (Education and Care Services National Law Application) Act 2010 Education and Care Services National Regulations
Please note the following changes to the Immunization Program:
 1 August 22, 2003 ATTN: Medical Health Officers and Branch Offices Public Health Nursing Administrators and Assistant Administrators Holders of Communicable Disease Control Manuals Re: Revisions to the
1 August 22, 2003 ATTN: Medical Health Officers and Branch Offices Public Health Nursing Administrators and Assistant Administrators Holders of Communicable Disease Control Manuals Re: Revisions to the
Family and Travel Vaccinations
 Family and Travel Vaccinations We offer the full range of baby, child and family vaccinations. We are able to tailor schedules to your child s needs or international schedule. We have a suggested vaccination
Family and Travel Vaccinations We offer the full range of baby, child and family vaccinations. We are able to tailor schedules to your child s needs or international schedule. We have a suggested vaccination
Adolescent vaccination strategies
 Adolescent vaccination strategies Gregory Hussey Vaccines for Africa Initiative Institute of Infectious Diseases & Molecular Medicine University of Cape Town www.vacfa.uct.ac.za gregory.hussey@uct.ac.za
Adolescent vaccination strategies Gregory Hussey Vaccines for Africa Initiative Institute of Infectious Diseases & Molecular Medicine University of Cape Town www.vacfa.uct.ac.za gregory.hussey@uct.ac.za
Immunisation records and data explained: A guide for immunisation providers
 Immunisation records and data explained: A guide for immunisation providers 2 nd edition, April 2017 Immunisation records and data explained: A guide for immunisation providers, 2 nd edition Published
Immunisation records and data explained: A guide for immunisation providers 2 nd edition, April 2017 Immunisation records and data explained: A guide for immunisation providers, 2 nd edition Published
