Varicella Vaccination in Australia and New Zealand
|
|
|
- Eleanor Booker
- 6 years ago
- Views:
Transcription
1 SUPPLEMENT ARTICLE Varicella Vaccination in Australia and New Zealand Kristine K. Macartney 1 and Margaret A. Burgess 2 1 National Centre for Immunisation Research and Surveillance, The Children s Hospital at Westmead, Westmead, and 2 Department of Paediatrics and Child Health, University of Sydney, Sydney, Australia Varicella-zoster virus has been responsible for a significant disease burden, including hospitalizations and deaths in Australia and New Zealand. Varicella vaccine has been available in Australia since 1999 and, since November 2005, has been funded under the National Immunisation Program for use in all children as a single dose at 18 months of age and in a school-based catch-up program at years of age. Recent hospitalization data from Australia show a decline in varicella hospitalizations in children 1 4 years of age, most likely related to vaccination. In New Zealand, varicella vaccine has been available since 1999 but is currently not recommended or funded on the New Zealand national immunization schedule. The anticipated licensure of combination measles-mumps-rubella-varicella vaccines in both countries may lead to future schedule changes. EPIDEMIOLOGY Australia. In Australia, varicella and herpes zoster (HZ) have not been notifiable diseases, although statebased surveillance for both varicella and HZ is currently being introduced. Estimates of the burden of varicellazoster virus (VZV) disease have relied on surveillance of hospitalizations and deaths, seroprevalence studies [1, 2], limited community-based surveys, and analyses of data on the prescription of pharmaceuticals [3]. It has been estimated that there are 240,000 cases of varicella in Australia each year, a number approximating the birth cohort [1, 2, 4]. In a recent study of seroprevalence, 83% of children years of age were seropositive for VZV IgG [1]. Age-specific attack rates were highest among children 0 9 years of age [1]. Al- Potential conflicts of interest: M.A.B. has received vaccine trial research support and given advice to both GlaxoSmithKline and Merck/CSL. K.K.M. reports no potential conflicts. Financial support: The authors are funded by the National Centre for Immunisation Research and Surveillance, Australia. The National Centre for Immunisation Research and Surveillance is supported by the Australian Government Department of Health and Ageing, the New South Wales Department of Health, the University of Sydney and the Children s Hospital at Westmead. Supplement sponsorship is detailed in the Acknowledgments. Reprints or correspondence: Dr. Kristine K. Macartney, National Centre for Immunisation Research and Surveillance, The Children s Hospital at Westmead, Locked Bag 4001, Westmead NSW 2145, Australia (kristinm@chw.edu.au). The Journal of Infectious Diseases 2008; 197:S by the Infectious Diseases Society of America. All rights reserved /2008/19705S2-0029$15.00 DOI: / though the incidence and seroconversion rates appear to be similar to those in many other countries with temperate climates, Australia may have a higher proportion of susceptible persons between the ages of 20 and 30 years (2% 10%) than does the United States [1]. Recent Australian hospitalization data indicate an average annual hospitalization rate for varicella of 8.7/100,000 population, or 5.5/100,000 population for varicella as the principal diagnosis [5]. Most hospitalizations were in the youngest age groups, with a median length of stay of 2 days; however, persons 60 years of age had the longest median length of stay (9 days). Approximately one-third of children admitted are immunocompromised or have chronic diseases [6]. There are 7 8 deaths due to varicella each year [5, 7]. Active surveillance for congenital varicella syndrome (CVS) and neonatal varicella before the introduction of vaccination indicated that CVS was notified at a rate of 0.8/100,000 live births, and neonatal varicella was notified at a rate of 5.8/100,000 live births [8]. Overall, the disease burden due to HZ is higher than that due to varicella, with average annual hospitalization rates of 24/100,000 population, or 10/100,000 population for HZ as the principal diagnosis [5]. The mortality due to HZ is 2 3 times higher than that due to varicella [3, 5]. It has been estimated that 1157,000 cases of HZ occurred in the Australian community in 1999, at a rate of 830/100,000 population [3]. Varicella Vaccination in Australia and New Zealand JID 2008:197 (Suppl 2) S191
2 New Zealand. In New Zealand, population-based studies of varicella have not been performed; however, the epidemiological pattern appears to be similar to patterns seen in all developed societies in temperate climates [9]. Hospital discharge information for varicella between 1970 and 2004 indicated that hospitalization rates for children 0 4 years of age were greatest, at 31/100,000 population, with the following hospitalization rates in other age brackets: 5 9 years of age, 12/ 100,000 population; years of age, 3/100,000 population; years of age, 1.5/100,000; years of age, 1.2/100,000; and 140 years of age, 0.5/100,000 [9]. The mean duration of hospital stay was 3 days, and only 4% of hospitalizations involved people with an underlying disease associated with immune suppression. On the basis of overseas rates, it is estimated that up to 1 case of CVS may be expected in New Zealand each year, although few have been reported. Mortality data are available for the period Nine deaths were attributed to chickenpox over the 14-year period , of which 4 occurred in children, 2 in infants, and 3 in adolescents or adults. None of the individuals who died had a contributory cause of death recorded. From 1994 to 2002, there were 9 deaths associated with varicella; 2 were in children 5 9 years of age, 4 were in adults years of age, and 3 were in adults 165 years of age [9]. It is estimated that, overall, there are 50,000 chickenpox cases each year in New Zealand, of which result in hospitalization, 1 2 result in residual long-term disability or death, and result in severe CVS [10]. The New Zealand Immunisation Handbook states that approximately two-thirds of this burden is borne by otherwise healthy children, and less than one-tenth is borne by children with a disease associated with immune suppression [10]. VACCINES AND RECOMMENDATIONS Australia. Varicella vaccines (Varivax Refrigerated [CSL Biotherapies/Merck] and Varilrix [GlaxoSmithKline]), have been licensed in Australia since In 2003, the Australian National Health and Medical Research Council recommended the addition of varicella vaccine to the National Immunisation Table 1. Recommendations for the use of varicella vaccine in Australia and New Zealand. Country [reference] Product (year available) Recommendations for use Funding Australia [4] Varivax Refrigerated (1999) Varilrix (1999) MMRV vaccines (?2007) New Zealand [10] Varivax Frozen (1999) Varivax Refrigerated (2004) Varilrix (1999) MMRV vaccines (?2007) One dose of VV is recommended for (1) all children aged 18 months (2) children years of age, unless they have already received a dose of VV or have a reliable clinical history of varicella (3) any nonimmune child Two doses of VV are recommended for nonimmune adolescents ( 14 years of age) and adults, especially (1) nonimmune people in high-risk occupations (2) nonimmune women before pregnancy (3) nonimmune parents of young children (4) nonimmune household contacts of immunosuppressed individuals Recommendations for use of MMRV vaccines in the Australian NIP are being considered. Varicella immunization is recommended, but not funded, for (1) adults and adolescents who were born and resident in tropical countries if they have no history of varicella (2) children with chronic liver disease who may in the future be candidates for transplantation; children with deteriorating renal function, as early as possible before transplantation; children likely to undergo solid organ transplant; and children with HIV infection at CDC stage N1 or A1 (2 doses) None Funding under the Australian NIP is provided for all children at 18 months of age and for 1 cohort of children at years of age; catch-up vaccine is delivered in a school-based program Varicella vaccine is not on the New Zealand National Immunisation Schedule; it is not funded NOTE. CDC, Centers for Disease Control and Prevention; MMRV, measles-mumps-rubella-varicella; NIP, National Immunisation Program; VV, varicella vaccine. S192 JID 2008:197 (Suppl 2) Macartney and Burgess
3 Program (NIP) for immunization of nonimmune children and nonimmune adolescents and adults [4], especially those in high-risk occupations or situations; however, the vaccine was not funded and cost at least US $50 (A $66) per dose to parents/ patients. Between 2003 and 2005, vaccine uptake was thought to be low; however, vaccine uptake estimates for children!4 years of age varied from 16% to 48% [11, 12]. Since November 2005, varicella vaccination has been funded under the NIP for all children at 18 months of age and in school-based programs for the catch-up cohort of children between 10 and 13 years of age [12] (table 1). The recommended age of 18 months was chosen for a number of reasons, including schedule crowding, with the only other immunization schedule point in the second year of life being 12 months of age, at which time measlesmumps-rubella, Haemophilus influenzae type b, and conjugate meningococcal C vaccine are given. The administration of childhood vaccines in the NIP is reported to the Australian Childhood Immunisation Register and should allow ongoing assessment of vaccine uptake [13]. The low coverage rates achieved with varicella vaccine over the past 5 years, before universal funding, are unlikely to have changed the epidemiology of varicella infection. Surveillance data from South Australia in 2002 and 2003 showed no apparent change in the number of cases of varicella and HZ [5]. However, preliminary analysis of national hospitalization data for varicella to June 2005 shows a downward trend in hospitalization rates in the years since 2003 among children 1 4 years of age (figure 1), who were most likely to have been vaccinated. New Zealand. Varicella vaccines (Varilrix and Varivax) have been available in New Zealand since Varivax was initially marketed as a frozen product; however, Varivax Refrigerated has been available since mid At present, varicella vaccine has not been added to the New Zealand National Immunisation Schedule, because of both the cost and the undesirability of adding another injection or immunization visit [10]. Vaccination is recommended, but not funded, for adults and adolescents who were born and resident in tropical countries and have no history of varicella and for a number of children with chronic medical conditions (table 1). The New Zealand Immunisation Handbook states that the vaccine can also be given to healthy children and susceptible adolescents whose parents request it. The cost of 1 dose of vaccine to parents/patients is at least US $36 (NZ $50). There are no available estimates of vaccine uptake. COST-EFFECTIVENESS OF VACCINATION Health economic studies in Australia and New Zealand, similar to those performed in the United States and other countries, Figure 1. Australian hospitalization rates for varicella by age, Varicella Vaccination in Australia and New Zealand JID 2008:197 (Suppl 2) S193
4 have modeled the costs and impact of 1 dose of universal varicella vaccine for a 30-year period [14, 15]. In the Australian study, on the basis of direct costs, a universal infant vaccination was considered to be the most effective but was very sensitive to vaccine price [15]. In New Zealand, from a health care payer s perspective, every dollar invested in a varicella vaccination program would return NZ $0.67; however, from a societal point of view (indirect costs), a vaccination program would return NZ $2.79 for every dollar invested. [14] FUTURE SCHEDULING Number of doses. In both Australia and New Zealand, consideration needs to be given to a 2-dose schedule, on the basis of the factors underpinning the decision to implement a 2- dose schedule in the United States [16]. Although potential differences in vaccine effectiveness may exist as a result of the use of different vaccine formulations, it is likely that the use of a 2-dose schedule would lead to greater population immunity to varicella. In Australia, such a schedule change would require a recommendation by the Australian Technical Advisory Group on Immunisation and a favorable cost-effectiveness assessment by the Pharmaceutical Benefits Advisory Committee. Use of measles-mumps-rubella-varicella (MMRV) vaccine. In both Australia and New Zealand, MMRV vaccines have been licensed but not marketed. A frozen formulation of ProQuad (Merck) is licensed but not marketed in both countries, because of its lack of suitability to existing cold-chain mechanisms. Similarly, Priorix-Tetra (GlaxoSmithKline) is licensed but not marketed in Australia, possibly because of the requirement of 2 doses of the present formulation to achieve adequate immunogenicity. It is likely that MMRV vaccines that are more suitable to the cold-chain systems and immunization schedules of both countries will be available in the future and could have an impact on vaccine scheduling at that point, provided they are considered to be cost-effective. SURVEILLANCE AND SAFETY An Australian study to predict the potential impact of universal varicella vaccination was performed [17], using the model developed by Brisson et al. [18] that proposes that immunity to varicella and HZ is boosted on re-exposure. Total morbidity due to varicella and HZ in Australia was predicted to decrease for the first 7 years of a program with 90% vaccination coverage and vaccine effectiveness estimated to be 93% [17]. However, 8 51 years after vaccination, total morbidity was predicted to be higher than prevaccination levels, with a peak in HZ cases occurring 15 years after the commencement of the program. The suggestions of such modeling, although not yet eventuating in the United States, underpin the need for effective surveillance of both varicella and HZ. This is currently being implemented on a state- and territory-wide basis in Australia through the National Notifiable Diseases Surveillance System (Australia) and is anticipated to utilize a number of active and passive surveillance approaches. In May 2006, the Australian Paediatric Surveillance Unit added CVS, neonatal varicella, and varicella complications requiring hospitalization in children 1 month to 15 years of age to its list of reportable diseases [19]. In addition, ongoing population-based serosurveys of immunity to varicella are planned to assess vaccine uptake and disease susceptibility. It will be important for these systems to address the question of vaccine effectiveness after the introduction of a universal program in Australia. In New Zealand, in the absence of widespread use of vaccine, hospitalization and death data are the means of surveillance. Adverse events following immunization are reported by immunization providers in Australia, by use of state-based mechanisms, to the Adverse Drug Reactions Advisory Committee [20]. In New Zealand, adverse events following immunization are reported to the Centre for Adverse Events Monitoring [10]. In 2005, Australia joined the United States and Canada in having a nationally funded universal varicella vaccination program. Careful ongoing assessment of the impact of this program on disease is required. Acknowledgments We thank Keith Grimwood of The University of Otago, New Zealand, for help with the manuscript, and Han Wang and Anita Heywood, of the National Centre for Immunisation Research and Surveillance, for producing the graph. Supplement sponsorship. This article was published as part of a supplement entitled Varicella Vaccine in the United States: A Decade of Prevention and the Way Forward, sponsored by the Research Foundation for Microbial Diseases of Osaka University, GlaxoSmithKline Biologicals, the Sabin Vaccine Institute, the Centers for Disease Control and Prevention, and the March of Dimes. References 1. Gidding HF, MacIntyre CR, Burgess MA, Gilbert GL. The seroepidemiology and transmission dynamics of varicella in Australia. Epidemiol Infect 2003; 131: Chant KG, Sullivan EA, Burgess MA, et al. Varicella-zoster virus infection in Australia. Aust N Z J Public Health 1998; 22:413 8 [erratum: Aust N Z J Public Health 1998; 22:630]. 3. MacIntyre CR, Chu CP, Burgess MA. Use of hospitalization and pharmaceutical prescribing data to compare the prevaccination burden of varicella and herpes zoster in Australia. Epidemiol Infect 2003; 131: National Health and Medical Research Council. Australian immunisation handbook, 8th ed. Canberra: Australian Government Department of Health and Ageing, Brotherton J, McIntyre P, Puech M, et al. Vaccine preventable diseases and vaccination coverage in Australia 2001 to Commun Dis Intell 2004; 28(Suppl 2):vii S Carapetis JR, Russell DM, Curtis N. The burden and cost of hospitalised varicella and zoster in Australian children. Vaccine 2004; 23: S194 JID 2008:197 (Suppl 2) Macartney and Burgess
5 7. McIntyre P, Gidding H, Gilmour R, et al. Vaccine preventable diseases and vaccination coverage in Australia, 1999 to Commun Dis Intell 2002; (Suppl):i xi, Forrest J, Mego S, Burgess M. Congenital and neonatal varicella in Australia. J Paediatr Child Health 2000; 36: Tobias M, Reid S, Lennon D, Meech R, Teele DW. Chickenpox immunisation in New Zealand. N Z Med J 1998; 111: Ministry of Health. Varicella (chickenpox and shingles). Immunisation handbook Wellington: New Zealand Ministry of Health, 2006: Marshall H, Ryan P, Roberton D. Uptake of varicella vaccine a cross sectional survey of parental attitudes to nationally recommended but unfunded varicella immunisation. Vaccine 2005; 23: Macartney KK, Beutels P, McIntyre P, Burgess MA. Varicella vaccination in Australia. J Paediatr Child Health 2005; 41: Hull B, Lawrence G, MacIntyre CR, McIntyre P. Immunisation coverage: Australia Canberra: Commonwealth Department of Health and Ageing, Scuffham P, Devlin N, Eberhart-Phillips J, Wilson-Salt R. The costeffectiveness of introducing a varicella vaccine to the New Zealand immunisation schedule. Soc Sci Med 1999; 49: Scuffham PA, Lowin AV, Burgess MA. The cost-effectiveness of varicella vaccine programs for Australia. Vaccine 1999; 18: Marin M, Guris D, Chaves SS, Schmid S, Seward JF, Advisory Committee on Immunization Practices. Prevention of varicella: recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Morb Mortal Wkly Rep 2007; 56(RR-4): Gidding HF, Brisson M, MacIntyre CR, Burgess MA. Modelling the impact of vaccination on the epidemiology of varicella zoster virus in Australia. Aust N Z J Public Health 2005; 29: Brisson M, Edmunds WJ, Gay NJ, Law B, De Serres G. Modelling the impact of immunization on the epidemiology of varicella zoster virus. Epidemiol Infect 2000; 125: Australian Paediatric Surveillance Unit (ASPU) website. Current studies October 1. Available at: Accessed 27 November Lawrence G, Menzies R, Burgess M, et al. Surveillance of adverse events following immunisation: Australia, Commun Dis Intell 2003; 27: Varicella Vaccination in Australia and New Zealand JID 2008:197 (Suppl 2) S195
Varicella and varicella vaccination An update
 THEME: School contagions Varicella and varicella vaccination An update John Litt, Margaret Burgess BACKGROUND Although varicella is generally mild in children, it is often more severe in adults and overall
THEME: School contagions Varicella and varicella vaccination An update John Litt, Margaret Burgess BACKGROUND Although varicella is generally mild in children, it is often more severe in adults and overall
EPIREVIEW INVASIVE PNEUMOCOCCAL DISEASE, NSW, 2002
 EPIREVIEW INVASIVE PNEUMOCOCCAL DISEASE, NSW, 2002 Robyn Gilmour Communicable Diseases Branch NSW Department of Health BACKGROUND Infection with the bacterium Streptococcus pneumoniae is a major cause
EPIREVIEW INVASIVE PNEUMOCOCCAL DISEASE, NSW, 2002 Robyn Gilmour Communicable Diseases Branch NSW Department of Health BACKGROUND Infection with the bacterium Streptococcus pneumoniae is a major cause
IMMUNISATION: A PUBLIC HEALTH SUCCESS CONTENTS
 NSW Public Health Bulletin IMMUNISATION: A PUBLIC HEALTH SUCCESS GUEST EDITORIAL Margaret Burgess National Centre for Immunisation Research The University of Sydney and The Children s Hospital at Westmead
NSW Public Health Bulletin IMMUNISATION: A PUBLIC HEALTH SUCCESS GUEST EDITORIAL Margaret Burgess National Centre for Immunisation Research The University of Sydney and The Children s Hospital at Westmead
Chickenpox vaccine for all?
 MODERN MEDICINE CPD ARTICLE NUMBER TWO: 1 point Chickenpox vaccine for all? MARK J FERSON, MB BS, MPH, MD, FRACP, FAFPHM A live attenuated varicella vaccine is now available in South Africa; it is approved
MODERN MEDICINE CPD ARTICLE NUMBER TWO: 1 point Chickenpox vaccine for all? MARK J FERSON, MB BS, MPH, MD, FRACP, FAFPHM A live attenuated varicella vaccine is now available in South Africa; it is approved
NSW Annual Vaccine-Preventable Disease Report, 2011
 NSW Annual Vaccine-Preventable Disease Report, 211 Alexander Rosewell A,B, Paula J. Spokes A and Robin E. Gilmour A A Health Protection NSW B Corresponding author. Email: arosw@doh.health.nsw.gov.au Abstract:
NSW Annual Vaccine-Preventable Disease Report, 211 Alexander Rosewell A,B, Paula J. Spokes A and Robin E. Gilmour A A Health Protection NSW B Corresponding author. Email: arosw@doh.health.nsw.gov.au Abstract:
Chickenpox Notification
 Goolwa Campus: (08) 8555 7500 Victor Harbor Campus: (08) 8551 0900 Investigator College Chickenpox Notification Date for Distribution: 2018 Dear Parents/Caregivers There has been a case of chickenpox reported
Goolwa Campus: (08) 8555 7500 Victor Harbor Campus: (08) 8551 0900 Investigator College Chickenpox Notification Date for Distribution: 2018 Dear Parents/Caregivers There has been a case of chickenpox reported
Varicella (Chickenpox) and Varicella Vaccines
 Centers for Disease Control and Prevention National Center for Immunization and Respiratory Diseases Varicella (Chickenpox) and Varicella Vaccines September 2018 Photographs and images included in this
Centers for Disease Control and Prevention National Center for Immunization and Respiratory Diseases Varicella (Chickenpox) and Varicella Vaccines September 2018 Photographs and images included in this
Changes in hospitalisations for acute gastroenteritis in Australia after the national rotavirus vaccination program
 Changes in hospitalisations for acute gastroenteritis in Australia after the national rotavirus vaccination program Rotavirus gastroenteritis is the leading cause of severe acute gastroenteritis (AGE)
Changes in hospitalisations for acute gastroenteritis in Australia after the national rotavirus vaccination program Rotavirus gastroenteritis is the leading cause of severe acute gastroenteritis (AGE)
SUPPLEMENT ARTICLE METHODS
 SUPPLEMENT ARTICLE Knowledge, Attitudes, and Practices Regarding Varicella Vaccination among Health Care Providers Participating in the Varicella Active Surveillance Project, Antelope Valley, California,
SUPPLEMENT ARTICLE Knowledge, Attitudes, and Practices Regarding Varicella Vaccination among Health Care Providers Participating in the Varicella Active Surveillance Project, Antelope Valley, California,
A PERTUSSIS EPIDEMIC IN NSW: HOW EPIDEMIOLOGY REFLECTS VACCINATION POLICY
 A PERTUSSIS EPIDEMIC IN NSW: HOW EPIDEMIOLOGY REFLECTS VACCINATION POLICY Julia Brotherton and Jeremy McAnulty Communicable Diseases Branch NSW Department of Health Pertussis has traditionally been considered
A PERTUSSIS EPIDEMIC IN NSW: HOW EPIDEMIOLOGY REFLECTS VACCINATION POLICY Julia Brotherton and Jeremy McAnulty Communicable Diseases Branch NSW Department of Health Pertussis has traditionally been considered
Health economic evaluation of vaccine: the example of varicella-zoster virus
 Health economic evaluation of vaccine: the example of varicella-zoster virus Benoît DERVAUX, CNRS, Catholic University of Lille Advances in infectious diseases modelling Annecy, Les Pensières, December
Health economic evaluation of vaccine: the example of varicella-zoster virus Benoît DERVAUX, CNRS, Catholic University of Lille Advances in infectious diseases modelling Annecy, Les Pensières, December
EPIDEMIOLOGY OF VACCINE-PREVENTABLE VIRAL INFECTIONS IN ABORIGINAL & TORRES STRAIT ISLANDER AUSTRALIANS
 EPIDEMIOLOGY OF VACCINE-PREVENTABLE VIRAL INFECTIONS IN ABORIGINAL & TORRES STRAIT ISLANDER AUSTRALIANS EMMA GOEMAN ADVANCED TRAINEE IN PAEDIATRIC INFECTIOUS DISEASES & MICROBIOLOGY Email: emma.goeman@health.nsw.gov.au
EPIDEMIOLOGY OF VACCINE-PREVENTABLE VIRAL INFECTIONS IN ABORIGINAL & TORRES STRAIT ISLANDER AUSTRALIANS EMMA GOEMAN ADVANCED TRAINEE IN PAEDIATRIC INFECTIOUS DISEASES & MICROBIOLOGY Email: emma.goeman@health.nsw.gov.au
Enhanced immunisation schedule Victoria
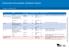 Children from March 2016 Age Disease Vaccine brand Birth Hepatitis B H-B-Vax-II Paediatric 2 months - from 6 weeks Diphtheria-tetanus-pertussis, poliomyelitis-hepatitis B- Reconstitute Site given Route
Children from March 2016 Age Disease Vaccine brand Birth Hepatitis B H-B-Vax-II Paediatric 2 months - from 6 weeks Diphtheria-tetanus-pertussis, poliomyelitis-hepatitis B- Reconstitute Site given Route
Immunisation schedule Victoria
 Infants and children July 2018 Birth H-B-Vax-II Paediatric or Paediatric 2 months (from 6 weeks) and 4 months Diphtheria-tetanus-pertussis, poliomyelitis-hepatitis B- Haemophilus influenzae type b Anterolateral
Infants and children July 2018 Birth H-B-Vax-II Paediatric or Paediatric 2 months (from 6 weeks) and 4 months Diphtheria-tetanus-pertussis, poliomyelitis-hepatitis B- Haemophilus influenzae type b Anterolateral
RACGP Immunisation Position Paper
 THE ROYAL AUSTRALIAN COLLEGE OF GENERAL PRACTITIONERS RACGP Immunisation Position Paper Aim The prevention of disease through vaccination is readily available to all Australian children Principles Prevention
THE ROYAL AUSTRALIAN COLLEGE OF GENERAL PRACTITIONERS RACGP Immunisation Position Paper Aim The prevention of disease through vaccination is readily available to all Australian children Principles Prevention
ROTAVIRUS VACCINES FOR AUSTRALIAN CHILDREN: INFORMATION FOR GPS AND IMMUNISATION PROVIDERS
 ROTAVIRUS VACCINES FOR AUSTRALIAN CHILDREN: INFORMATION FOR GPS AND IMMUNISATION PROVIDERS Summary Rotavirus is the most common cause of severe gastroenteritis in infants and young children, accounting
ROTAVIRUS VACCINES FOR AUSTRALIAN CHILDREN: INFORMATION FOR GPS AND IMMUNISATION PROVIDERS Summary Rotavirus is the most common cause of severe gastroenteritis in infants and young children, accounting
Early Learning Centre Immunisation Policy Legislation ACT Public Health Regulations (2000)
 Early Learning Centre Immunisation Policy Legislation ACT Public Health Regulations (2000) National Quality Standard / Education and Care Services National Regulations Standard 2.1 Each child s health
Early Learning Centre Immunisation Policy Legislation ACT Public Health Regulations (2000) National Quality Standard / Education and Care Services National Regulations Standard 2.1 Each child s health
Health benefits versus intussusception risk of rotavirus vaccination in Australia
 Health benefits versus intussusception risk of rotavirus vaccination in Australia Julie Bines 1 On behalf of co-authors: John Carlin 1, Kristine Macartney 2, Katherine Lee 1, Helen Quinn 2, Jim Buttery
Health benefits versus intussusception risk of rotavirus vaccination in Australia Julie Bines 1 On behalf of co-authors: John Carlin 1, Kristine Macartney 2, Katherine Lee 1, Helen Quinn 2, Jim Buttery
Varicella vaccination: a laboured take-off
 REVIEW 10.1111/1469-0691.12580 Varicella vaccination: a laboured take-off P. Carrillo-Santisteve and P. L. Lopalco Scientific Advice Section, ECDC, Stockholm, Sweden Abstract Varicella vaccines are highly
REVIEW 10.1111/1469-0691.12580 Varicella vaccination: a laboured take-off P. Carrillo-Santisteve and P. L. Lopalco Scientific Advice Section, ECDC, Stockholm, Sweden Abstract Varicella vaccines are highly
Monitoring vaccine-preventable diseases is
 New South Wales annual vaccinepreventable disease report, 2013 Surveillance Report Alexander Rosewell, a Paula Spokes a and Robin Gilmour a Correspondence to Robin Gilmour (e-mail: rgilm@doh.health.nsw.gov.au).
New South Wales annual vaccinepreventable disease report, 2013 Surveillance Report Alexander Rosewell, a Paula Spokes a and Robin Gilmour a Correspondence to Robin Gilmour (e-mail: rgilm@doh.health.nsw.gov.au).
Antigen Review for the New Zealand National Immunisation Schedule, 2016: Varicella-zoster virus (chickenpox)
 Antigen Review for the New Zealand National Immunisation Schedule, 2016: Varicella-zoster virus (chickenpox) Prepared as part of a Ministry of Health contract for services by the Immunisation Advisory
Antigen Review for the New Zealand National Immunisation Schedule, 2016: Varicella-zoster virus (chickenpox) Prepared as part of a Ministry of Health contract for services by the Immunisation Advisory
Routine Adult Immunization: American College of Preventive Medicine Practice Policy Statement, updated 2002
 Routine Adult Immunization: American College of Preventive Medicine Practice Policy Statement, updated 2002 Ann R. Fingar, MD, MPH, and Byron J. Francis, MD, MPH Burden of suffering Vaccines are available
Routine Adult Immunization: American College of Preventive Medicine Practice Policy Statement, updated 2002 Ann R. Fingar, MD, MPH, and Byron J. Francis, MD, MPH Burden of suffering Vaccines are available
An Economic Analysis of the Universal Varicella Vaccination Program in the United States
 SUPPLEMENT ARTICLE An Economic Analysis of the Universal Varicella Vaccination Program in the United States Fangjun Zhou, 1 Ismael R. Ortega-Sanchez, 1 Dalya Guris, 1,a Abigail Shefer, 1 Tracy Lieu, 2
SUPPLEMENT ARTICLE An Economic Analysis of the Universal Varicella Vaccination Program in the United States Fangjun Zhou, 1 Ismael R. Ortega-Sanchez, 1 Dalya Guris, 1,a Abigail Shefer, 1 Tracy Lieu, 2
A WORD FROM SIR GUSTAV NOSSAL ABOUT THE NCIRS. Our Vision. Our Purpose To lead and support collaborative research,
 1997 217 ABOUT THE NCIRS A WORD FROM SIR GUSTAV NOSSAL Our Vision To be a world leader in translational research optimising the population health benefits of immunisation The National Centre for Immunisation
1997 217 ABOUT THE NCIRS A WORD FROM SIR GUSTAV NOSSAL Our Vision To be a world leader in translational research optimising the population health benefits of immunisation The National Centre for Immunisation
Vaccination against shingles for adults aged 70 and 79 years of age Q&A s for healthcare professionals
 Vaccination against shingles for adults aged 70 and 79 years of age Q&A s for healthcare professionals Background In 2010, the Joint Committee on Vaccination and Immunisation (JCVI) 1 were asked by the
Vaccination against shingles for adults aged 70 and 79 years of age Q&A s for healthcare professionals Background In 2010, the Joint Committee on Vaccination and Immunisation (JCVI) 1 were asked by the
What are the new active vaccine recommendations in the Canadian Immunization Guide?
 154 CCDR 17 April 2014 Volume 40-8 https://doi.org/10.14745/ccdr.v40i08a03 1 What are the new active vaccine recommendations in the Canadian Immunization Guide? Warshawsky B 1 and Gemmill I 2 on behalf
154 CCDR 17 April 2014 Volume 40-8 https://doi.org/10.14745/ccdr.v40i08a03 1 What are the new active vaccine recommendations in the Canadian Immunization Guide? Warshawsky B 1 and Gemmill I 2 on behalf
Series of 2 doses, 6-12 months apart. One dose is 720 Elu/0.5ml (GSK) or 25 u/0.5 ml (Merck)
 UTAH PREVENTIVE CARE RECOMMENDATIONS Adult - Ages 19 and Above IMMUNIZATIONS CONTENTS: General Instructions Hepatitis A Hepatitis B Human Papilloma Virus Influenza Meningococcal A, C, Y, W (MCV4) Meningococcal
UTAH PREVENTIVE CARE RECOMMENDATIONS Adult - Ages 19 and Above IMMUNIZATIONS CONTENTS: General Instructions Hepatitis A Hepatitis B Human Papilloma Virus Influenza Meningococcal A, C, Y, W (MCV4) Meningococcal
Walter A. Orenstein, M.D. Professor of Medicine and Pediatrics Director, Emory Vaccine Policy and Development Associate Director, Emory Vaccine Center
 Could Vaccines be a Possible Model For Pediatric Drug Development? June 13, 2006 Walter A. Orenstein, M.D. Professor of Medicine and Pediatrics Director, Emory Vaccine Policy and Development Associate
Could Vaccines be a Possible Model For Pediatric Drug Development? June 13, 2006 Walter A. Orenstein, M.D. Professor of Medicine and Pediatrics Director, Emory Vaccine Policy and Development Associate
Current Immunisation Issues in the Nepean Blue Mountains Presented as part of the Immunisation Update education series in October 2017
 Current Immunisation Issues in the Nepean Blue Mountains 2017 Presented as part of the Immunisation Update education series in October 2017 Additional funded vaccines Catch up vaccines for all individuals
Current Immunisation Issues in the Nepean Blue Mountains 2017 Presented as part of the Immunisation Update education series in October 2017 Additional funded vaccines Catch up vaccines for all individuals
2018 Immunisation Update
 2018 Immunisation Update A puzzling time?!! Sometimes the hardest pieces of a puzzle to assemble, are the ones missing from the box. Dixie Waters, Author The Puzzle Pieces Immunisation coverage rates NSW
2018 Immunisation Update A puzzling time?!! Sometimes the hardest pieces of a puzzle to assemble, are the ones missing from the box. Dixie Waters, Author The Puzzle Pieces Immunisation coverage rates NSW
CVU: What s new? 2 nd December 2013
 CVU: What s new? 2 nd December 2013 Dr Margie Danchin Paediatrician, Department of General Medicine, RCH Senior Research Fellow, MCRI Senior Fellow, Department of Paediatrics, The University of Melbourne
CVU: What s new? 2 nd December 2013 Dr Margie Danchin Paediatrician, Department of General Medicine, RCH Senior Research Fellow, MCRI Senior Fellow, Department of Paediatrics, The University of Melbourne
immunisation in New Zealand
 This appendix details the history of. Section A1.1 is a brief summary of when each vaccine was introduced to the National Immunisation Schedule (the Schedule). This summary includes vaccines which were
This appendix details the history of. Section A1.1 is a brief summary of when each vaccine was introduced to the National Immunisation Schedule (the Schedule). This summary includes vaccines which were
Progress in varicella vaccine research
 40 1, 1, 1, 2 1., 230032; 2., 10032 : - ( VZV) VZV 1995,, Oka,,, VZV : - ; Oka ; Progress in varicella vaccine research GAN Lin 1, WANG Ming-Li 1, Jason Chen 1, 2 1. Department of Microbiology, Anhui Medical
40 1, 1, 1, 2 1., 230032; 2., 10032 : - ( VZV) VZV 1995,, Oka,,, VZV : - ; Oka ; Progress in varicella vaccine research GAN Lin 1, WANG Ming-Li 1, Jason Chen 1, 2 1. Department of Microbiology, Anhui Medical
Guidelines for Vaccinating Pregnant Women
 Guidelines for Vaccinating Pregnant Women March 2012 Guidelines for Vaccinating Pregnant Women Abstracted from recommendations of the Advisory Committee on Immunization Practices (ACIP) MARCH 2012 Risk
Guidelines for Vaccinating Pregnant Women March 2012 Guidelines for Vaccinating Pregnant Women Abstracted from recommendations of the Advisory Committee on Immunization Practices (ACIP) MARCH 2012 Risk
Dr Stewart Reid. General Practitioner Ropata Medical Centre Wellington
 Dr Stewart Reid General Practitioner Ropata Medical Centre Wellington 7:15-8:10 Medicines New Zealand Breakfast Session Adult Vaccination The Poor Cousin Adult Vaccination The poor cousin Stewart Reid
Dr Stewart Reid General Practitioner Ropata Medical Centre Wellington 7:15-8:10 Medicines New Zealand Breakfast Session Adult Vaccination The Poor Cousin Adult Vaccination The poor cousin Stewart Reid
Impact and effectiveness of national immunisation programmes. David Green, Nurse Consultant, Immunisations Public Health England
 Impact and effectiveness of national immunisation programmes David Green, Nurse Consultant, Immunisations Public Health England Session objectives Immunisation planning and implementation The impact and
Impact and effectiveness of national immunisation programmes David Green, Nurse Consultant, Immunisations Public Health England Session objectives Immunisation planning and implementation The impact and
Director of Public Health Board Paper No. 13/13
 Greater Glasgow and Clyde NHS Board Director of Public Health Board Paper No. 13/13 Report of the Director of Public Health : Major Development to Immunisation Programmes in Scotland Implications for NHSGGC
Greater Glasgow and Clyde NHS Board Director of Public Health Board Paper No. 13/13 Report of the Director of Public Health : Major Development to Immunisation Programmes in Scotland Implications for NHSGGC
HERPES ZOSTER VACCINATION
 HERPES ZOSTER VACCINATION Herpes Zoster (shingles) is a reactivation of the varicella zoster infection in some-one who has previously had varicella(chicken pox) disease Painful, unilateral, self-limiting
HERPES ZOSTER VACCINATION Herpes Zoster (shingles) is a reactivation of the varicella zoster infection in some-one who has previously had varicella(chicken pox) disease Painful, unilateral, self-limiting
MarketVIEW: MMR, MMRV and varicella vaccines
 ****Published October 2014*** MarketVIEW: MMR, MMRV and varicella vaccines Product Name : MarketVIEW: MMR, MMRV and varicella vaccines Description : Global vaccine commercial opportunity assessment Contents
****Published October 2014*** MarketVIEW: MMR, MMRV and varicella vaccines Product Name : MarketVIEW: MMR, MMRV and varicella vaccines Description : Global vaccine commercial opportunity assessment Contents
Immunisation Subcommittee of PTAC Meeting held 10 February (minutes for web publishing)
 Immunisation Subcommittee of PTAC Meeting held 10 February 2014 (minutes for web publishing) Immunisation Subcommittee minutes are published in accordance with the Terms of Reference for the Pharmacology
Immunisation Subcommittee of PTAC Meeting held 10 February 2014 (minutes for web publishing) Immunisation Subcommittee minutes are published in accordance with the Terms of Reference for the Pharmacology
VARICELLA. Dr Louise Cooley Royal Hobart Hospital
 VARICELLA Dr Louise Cooley Royal Hobart Hospital Varicella Zoster Virus (VZV): The Basics Herpes virus Exclusively human infection Primary infection: varicella (chickenpox) Neurotropic, establishing latency
VARICELLA Dr Louise Cooley Royal Hobart Hospital Varicella Zoster Virus (VZV): The Basics Herpes virus Exclusively human infection Primary infection: varicella (chickenpox) Neurotropic, establishing latency
Victorian immunisation catch-up tool for 10 to 19 year olds
 Victorian immunisation catch-up tool for 10 to 19 year olds January 2018 Guidelines for 10 to 19 year olds with no documented history of vaccine administration The objective of catch-up vaccination is
Victorian immunisation catch-up tool for 10 to 19 year olds January 2018 Guidelines for 10 to 19 year olds with no documented history of vaccine administration The objective of catch-up vaccination is
Varicella Vaccination in New Zealand: NHC Assessment 2012
 Varicella Vaccination in New Zealand: NHC Assessment 2012 In 2014 the National Health Committee (NHC) received a request for a copy of the NHC s assessment report on varicella (chicken pox) vaccination.
Varicella Vaccination in New Zealand: NHC Assessment 2012 In 2014 the National Health Committee (NHC) received a request for a copy of the NHC s assessment report on varicella (chicken pox) vaccination.
H igh rates of hospitalisation have been recognised in
 20 ORIGINAL ARTICLE related hospitalisations in Sydney, New South Wales, Australia F Beard, P McIntyre, H Gidding, M Watson... See end of article for authors affiliations... Correspondence to: Dr F Beard,
20 ORIGINAL ARTICLE related hospitalisations in Sydney, New South Wales, Australia F Beard, P McIntyre, H Gidding, M Watson... See end of article for authors affiliations... Correspondence to: Dr F Beard,
Ms Kim Hunter. Clinical Nurse Coordinator Immunisation Population Health Waikato District Health Board
 Ms Kim Hunter Clinical Nurse Coordinator Immunisation Population Health Waikato District Health Board 11:30-12:00 A Hitchhiker's Guide to Vaccine Preventable Disease Hitchhikers Guide to Vaccines and Preventable
Ms Kim Hunter Clinical Nurse Coordinator Immunisation Population Health Waikato District Health Board 11:30-12:00 A Hitchhiker's Guide to Vaccine Preventable Disease Hitchhikers Guide to Vaccines and Preventable
Introduction and overview of the program; new vaccine pipeline and prioritization process
 Immunization for the Modern Family: Western Canada Immunization Forum 2011 Introduction and overview of the program; new vaccine pipeline and prioritization process Monika Naus, MD, MHSc, FRCPC, FACPM
Immunization for the Modern Family: Western Canada Immunization Forum 2011 Introduction and overview of the program; new vaccine pipeline and prioritization process Monika Naus, MD, MHSc, FRCPC, FACPM
Vaccine Update Paul A. Offit, MD Division of Infectious Diseases Vaccine Education Center The Children
 Vaccine Update Paul A. Offit, MD Division of Infectious Diseases Vaccine Education Center The Children s Hospital of Philadelphia Perelman School of Medicine The University of Pennsylvania Topics Hep A:
Vaccine Update Paul A. Offit, MD Division of Infectious Diseases Vaccine Education Center The Children s Hospital of Philadelphia Perelman School of Medicine The University of Pennsylvania Topics Hep A:
BCG vaccine and tuberculosis
 PART 2: Vaccination for special risk groups 2.1 Vaccination for Aboriginal and Torres Strait Islander people Aboriginal and Torres Strait Islander people historically had a very high burden of infectious
PART 2: Vaccination for special risk groups 2.1 Vaccination for Aboriginal and Torres Strait Islander people Aboriginal and Torres Strait Islander people historically had a very high burden of infectious
Guidelines for Vaccinating Pregnant Women
 Guidelines for Vaccinating Pregnant Women April 2013 Guidelines for Vaccinating Pregnant Women Abstracted from recommendations of the Advisory Committee on Immunization Practices (ACIP) April 2013 Risk
Guidelines for Vaccinating Pregnant Women April 2013 Guidelines for Vaccinating Pregnant Women Abstracted from recommendations of the Advisory Committee on Immunization Practices (ACIP) April 2013 Risk
The University of Toledo Medical Center and its Medical Staff, Residents, Fellows, Salaried and Hourly employees
 Name of Policy: Policy Number: Department: Approving Officer: Responsible Agent: Scope: Healthcare Worker Immunizations 3364-109-EH-603 Infection Prevention and Control Hospital Administration Medical
Name of Policy: Policy Number: Department: Approving Officer: Responsible Agent: Scope: Healthcare Worker Immunizations 3364-109-EH-603 Infection Prevention and Control Hospital Administration Medical
Persistence of Immunity to Live Attenuated Varicella Vaccine in Healthy Adults
 MAJOR ARTICLE Persistence of Immunity to Live Attenuated Varicella Vaccine in Healthy Adults Krow Ampofo, 1 Lisa Saiman, 1,2 Philip LaRussa, 1 Sharon Steinberg, 1 Paula Annunziato, 1 and Anne Gershon 1
MAJOR ARTICLE Persistence of Immunity to Live Attenuated Varicella Vaccine in Healthy Adults Krow Ampofo, 1 Lisa Saiman, 1,2 Philip LaRussa, 1 Sharon Steinberg, 1 Paula Annunziato, 1 and Anne Gershon 1
Andrea Streng, Johannes G. Liese
 Editorial Fifteen years of routine childhood varicella vaccination in the United States strong decrease in the burden of varicella disease and no negative effects on the population level thus far Andrea
Editorial Fifteen years of routine childhood varicella vaccination in the United States strong decrease in the burden of varicella disease and no negative effects on the population level thus far Andrea
VARICELLA EPIDEMIOLOGY AND COST-EFFECTIVENESS ANALYSIS OF UNIVERSAL VARICELLA VACCINATION PROGRAM IN TAIWAN
 VARICELLA EPIDEMIOLOGY AND COST-EFFECTIVENESS ANALYSIS OF UNIVERSAL VARICELLA VACCINATION PROGRAM IN TAIWAN HF Tseng 1, HF Tan 2 and CK Chang 3 1 Institute of Medical Research, Chang-Jung Christian University,
VARICELLA EPIDEMIOLOGY AND COST-EFFECTIVENESS ANALYSIS OF UNIVERSAL VARICELLA VACCINATION PROGRAM IN TAIWAN HF Tseng 1, HF Tan 2 and CK Chang 3 1 Institute of Medical Research, Chang-Jung Christian University,
Vaccinations for Adults
 Case: Vaccinations for Adults Lisa Winston, MD University of California, San Francisco San Francisco General Hospital A 30-year old healthy woman comes for a routine visit. She is recently married and
Case: Vaccinations for Adults Lisa Winston, MD University of California, San Francisco San Francisco General Hospital A 30-year old healthy woman comes for a routine visit. She is recently married and
UPDATE ON IMMUNIZATION GUIDELINES AND PRACTICES
 DISCLOSURES UPDATE ON IMMUNIZATION GUIDELINES AND PRACTICES Nothing to disclose Kylie Mueller, Pharm.D., BCPS Clinical Specialist, Infectious Diseases Spartanburg Regional Medical Center LEARNING OBJECTIVES
DISCLOSURES UPDATE ON IMMUNIZATION GUIDELINES AND PRACTICES Nothing to disclose Kylie Mueller, Pharm.D., BCPS Clinical Specialist, Infectious Diseases Spartanburg Regional Medical Center LEARNING OBJECTIVES
Appendix An Assessment Tool to Determine the Validity of Vaccine Doses
 Appendix 4.4 - An Assessment Tool to Determine the Validity of Vaccine Doses Note: Refer to the Canadian Immunization Guide and New Brunswick (NB) immunization program directives for recommendations for
Appendix 4.4 - An Assessment Tool to Determine the Validity of Vaccine Doses Note: Refer to the Canadian Immunization Guide and New Brunswick (NB) immunization program directives for recommendations for
Immunization Guidelines for the Use of State Supplied Vaccine April 18, 2013
 DTaP / DT DTaP/IPV/Hep B Combination (Pediarix ) Children from 6 weeks of age up to the 7 th birthday Children from 2 months of age up to the 7th birthday: Indicated for the primary doses of DTaP, IPV,
DTaP / DT DTaP/IPV/Hep B Combination (Pediarix ) Children from 6 weeks of age up to the 7 th birthday Children from 2 months of age up to the 7th birthday: Indicated for the primary doses of DTaP, IPV,
Mathematical Modelling of Infectious Diseases. Raina MacIntyre
 Mathematical Modelling of Infectious Diseases Raina MacIntyre A little bit of EBM is a dangerous thing Research question: Does smoking cause lung cancer? Answer: I couldn t find a meta-analysis or even
Mathematical Modelling of Infectious Diseases Raina MacIntyre A little bit of EBM is a dangerous thing Research question: Does smoking cause lung cancer? Answer: I couldn t find a meta-analysis or even
IMMUNISATION POLICY. Explanation: It is imperative that children are kept safe and healthy at all times in the centre environment.
 IMMUNISATION POLICY Aim: Immunisation is a simple, safe and effective way of protecting people against harmful diseases before they come into contact with them in the community. Immunisation not only protects
IMMUNISATION POLICY Aim: Immunisation is a simple, safe and effective way of protecting people against harmful diseases before they come into contact with them in the community. Immunisation not only protects
No Jab, No Pay New Immunisation Requirements for Family Assistance Payments
 No Jab, No Pay New Immunisation Requirements for Family Assistance Payments SUMMARY From 1 January 2016: Only parents of children (less than 20 years of age) who are fully immunised or are on a recognised
No Jab, No Pay New Immunisation Requirements for Family Assistance Payments SUMMARY From 1 January 2016: Only parents of children (less than 20 years of age) who are fully immunised or are on a recognised
Decline in Mortality Due to Varicella after Implementation of Varicella Vaccination in the United States
 The new england journal of medicine original article Decline in Mortality Due to Varicella after Implementation of Varicella Vaccination in the United States Huong Q. Nguyen, M.P.H., Aisha O. Jumaan, Ph.D.,
The new england journal of medicine original article Decline in Mortality Due to Varicella after Implementation of Varicella Vaccination in the United States Huong Q. Nguyen, M.P.H., Aisha O. Jumaan, Ph.D.,
2016 Vaccine Preventable Disease Summary
 2016 Vaccine Preventable Disease Summary 12251 James Street Holland, MI 49424 www.miottawa.org/healthdata Prepared October 2017 2016 Summary of Vaccine Preventable Diseases (VPDs) Reported to Ottawa County
2016 Vaccine Preventable Disease Summary 12251 James Street Holland, MI 49424 www.miottawa.org/healthdata Prepared October 2017 2016 Summary of Vaccine Preventable Diseases (VPDs) Reported to Ottawa County
Vaccinology Overview. Complexity of the Vaccine Approval Process Including Lessons Learned
 Vaccinology Overview Complexity of the Vaccine Approval Process Including Lessons Learned Larry K. Pickering, MD, FAAP, FIDSA, FPIDS August 18, 2018 Faculty Disclosure Information In the past 12 months,
Vaccinology Overview Complexity of the Vaccine Approval Process Including Lessons Learned Larry K. Pickering, MD, FAAP, FIDSA, FPIDS August 18, 2018 Faculty Disclosure Information In the past 12 months,
Immunization Guidelines For the Use of State Supplied Vaccine July 1, 2011
 DTaP / DT DTaP/IPV/Hep B Combination (Pediarix ) Children from 6 weeks of age up to the 7 th birthday Children from 2 months of age up to the 7th birthday: Indicated for the primary doses of DTaP, IPV,
DTaP / DT DTaP/IPV/Hep B Combination (Pediarix ) Children from 6 weeks of age up to the 7 th birthday Children from 2 months of age up to the 7th birthday: Indicated for the primary doses of DTaP, IPV,
Changes to the National Immunisation Schedule
 17 December 2013 Changes to the National Immunisation Schedule PHARMAC is pleased to announce decisions related to the National Immunisation Schedule (NIS) that will take effect from 1 July 2014. This
17 December 2013 Changes to the National Immunisation Schedule PHARMAC is pleased to announce decisions related to the National Immunisation Schedule (NIS) that will take effect from 1 July 2014. This
Measles and Measles Vaccine
 Measles and Measles Vaccine Epidemiology and Prevention of Vaccine- Preventable Diseases Note to presenters: Images of vaccine-preventable diseases are available from the Immunization Action Coalition
Measles and Measles Vaccine Epidemiology and Prevention of Vaccine- Preventable Diseases Note to presenters: Images of vaccine-preventable diseases are available from the Immunization Action Coalition
T he introduction of universal varicella vaccination in the
 862 ORIGINAL ARTICLE Varicella vaccination in England and Wales: cost-utility analysis M Brisson, W J Edmunds... Arch Dis Child 2003;88:862 869 See end of article for authors affiliations... Correspondence
862 ORIGINAL ARTICLE Varicella vaccination in England and Wales: cost-utility analysis M Brisson, W J Edmunds... Arch Dis Child 2003;88:862 869 See end of article for authors affiliations... Correspondence
Washtenaw County Community Mental Health HEALTH CARE PERSONNEL (HCP) VACCINES (RECOMMENDED EMPLOYEE IMMUNIZATIONS)
 Washtenaw County Community Mental Health HEALTH CARE PERSONNEL (HCP) VACCINES (RECOMMENDED EMPLOYEE IMMUNIZATIONS) PURPOSE To reduce the risk of exposure of Washtenaw County Community Mental Health (CMH)
Washtenaw County Community Mental Health HEALTH CARE PERSONNEL (HCP) VACCINES (RECOMMENDED EMPLOYEE IMMUNIZATIONS) PURPOSE To reduce the risk of exposure of Washtenaw County Community Mental Health (CMH)
Update on Vaccine Recommendations. Objectives. Childhood Immunization Schedule At the Turn of the Century. New Horizons in Pediatrics April 30, 2017
 Centers for for Disease Disease Control Control and and Prevention Prevention National Center for Immunization and Respiratory Diseases Update on Vaccine Recommendations New Horizons in Pediatrics April
Centers for for Disease Disease Control Control and and Prevention Prevention National Center for Immunization and Respiratory Diseases Update on Vaccine Recommendations New Horizons in Pediatrics April
VACCINES FOR ADULTS. Developing an Underutilised Health Resource. 13 November 2007
 VACCINES FOR ADULTS Developing an Underutilised Health Resource 13 November 2007 OUTLINE background to adult immunisation in Canada roles and recommendations of immunisation advisory committees in Canada
VACCINES FOR ADULTS Developing an Underutilised Health Resource 13 November 2007 OUTLINE background to adult immunisation in Canada roles and recommendations of immunisation advisory committees in Canada
Rationale for recommendations
 Australian Technical Advisory Group on Immunisation (ATAGI) Statement Clinical advice for immunisation providers on resumption of the use of 2010 trivalent seasonal vaccines in children less than 5 years
Australian Technical Advisory Group on Immunisation (ATAGI) Statement Clinical advice for immunisation providers on resumption of the use of 2010 trivalent seasonal vaccines in children less than 5 years
Healthcare Personnel Immunization Recommendations
 Healthcare Personnel Immunization Recommendations Kathleen Harriman, PhD, MPH, RN California Department of Public Health Immunization Branch Vaccine Preventable Disease Epidemiology Section kathleen.harriman@cdph.ca.gov
Healthcare Personnel Immunization Recommendations Kathleen Harriman, PhD, MPH, RN California Department of Public Health Immunization Branch Vaccine Preventable Disease Epidemiology Section kathleen.harriman@cdph.ca.gov
Immunization Guidelines for the Use of State Supplied Vaccine May 17, 2015
 DTaP / DT DTaP/IPV/Hep B Combination (Pediarix ) Children from 6 weeks of age up to the 7 th birthday Children from 2 months of age up to the 7th birthday: Indicated for the primary doses of DTaP, IPV,
DTaP / DT DTaP/IPV/Hep B Combination (Pediarix ) Children from 6 weeks of age up to the 7 th birthday Children from 2 months of age up to the 7th birthday: Indicated for the primary doses of DTaP, IPV,
Immunizations: new and sometimes confusing recommendations MARK H. SAWYER, MD UCSD SCHOOL OF MEDICINE AND RADY CHILDREN S HOSPITAL SAN DIEGO
 Immunizations: new and sometimes confusing recommendations MARK H. SAWYER, MD UCSD SCHOOL OF MEDICINE AND RADY CHILDREN S HOSPITAL SAN DIEGO Disclosures I have no financial disclosures related to this
Immunizations: new and sometimes confusing recommendations MARK H. SAWYER, MD UCSD SCHOOL OF MEDICINE AND RADY CHILDREN S HOSPITAL SAN DIEGO Disclosures I have no financial disclosures related to this
Varicella among Adults: Data from an Active Surveillance Project,
 SUPPLEMENT ARTICLE Varicella among Adults: Data from an Active Surveillance Project, 1995 2005 Mona Marin, 1 Tureka L. Watson, 1 Sandra S. Chaves, 1 Rachel Civen, 2 Barbara M. Watson, 3 John X. Zhang,
SUPPLEMENT ARTICLE Varicella among Adults: Data from an Active Surveillance Project, 1995 2005 Mona Marin, 1 Tureka L. Watson, 1 Sandra S. Chaves, 1 Rachel Civen, 2 Barbara M. Watson, 3 John X. Zhang,
Maternal immunisation and infant immunisation: core business for GPs What s new in 2017?
 Maternal immunisation and infant immunisation: core business for GPs What s new in 2017? Peter McIntyre Director, National Centre for Immunisation Research & Surveillance Senior Staff Specialist, Children
Maternal immunisation and infant immunisation: core business for GPs What s new in 2017? Peter McIntyre Director, National Centre for Immunisation Research & Surveillance Senior Staff Specialist, Children
Vaccinology 2017 Hanoi, Vietnam October 2017
 Vaccinology 2017 Hanoi, Vietnam October 2017 Active surveillance to assess vaccine benefits and risks Associate Professor Kristine Macartney National Centre for Immunisation Research and Surveillance University
Vaccinology 2017 Hanoi, Vietnam October 2017 Active surveillance to assess vaccine benefits and risks Associate Professor Kristine Macartney National Centre for Immunisation Research and Surveillance University
Immunisation Policy. Country Children s Early Learning Ph: M:
 Immunisation Policy Published June 2018 Review Date December 2019 Sources Education and Care Services National Regulations, October 2017 Guide to the Education and Care Services National Law and the Education
Immunisation Policy Published June 2018 Review Date December 2019 Sources Education and Care Services National Regulations, October 2017 Guide to the Education and Care Services National Law and the Education
ACIP Meeting Update, New Recommendations and Pending Influenza Season
 ACIP Meeting Update, New Recommendations and Pending Influenza Season February 17 th 2011 www.immunizetexas.com ACIP Upcoming Agenda and New Recommendations ACIP (February 23-24 th 2011) Topics for meeting
ACIP Meeting Update, New Recommendations and Pending Influenza Season February 17 th 2011 www.immunizetexas.com ACIP Upcoming Agenda and New Recommendations ACIP (February 23-24 th 2011) Topics for meeting
Routine Immunization Schedules. Section 2. Newfoundland and Labrador Immunization Manual. Routine Immunization Schedules
 Newfoundland and Labrador Immunization Manual Section 2... 2.1 Routine and Delayed Immunization Schedules for Infants and Children... 2.2 Recommended Immunizations for Adults... 2.3 (Provinces and Territories)...
Newfoundland and Labrador Immunization Manual Section 2... 2.1 Routine and Delayed Immunization Schedules for Infants and Children... 2.2 Recommended Immunizations for Adults... 2.3 (Provinces and Territories)...
OVERVIEW OF THE NATIONAL CHILDHOOD IMMUNISATION PROGRAMME IN SINGAPORE
 OVERVIEW OF THE NATIONAL CHILDHOOD IMMUNISATION PROGRAMME IN SINGAPORE Dr Tiong Wei Wei, MD, MPH Senior Assistant Director Policy and Control Branch, Communicable Diseases Division Ministry of Health 9
OVERVIEW OF THE NATIONAL CHILDHOOD IMMUNISATION PROGRAMME IN SINGAPORE Dr Tiong Wei Wei, MD, MPH Senior Assistant Director Policy and Control Branch, Communicable Diseases Division Ministry of Health 9
Healthy People 2020 objectives were released in 2010, with a 10-year horizon to achieve the goals by 2020.
 Appendix 1: Healthy People 2020 Immunization-related Objectives Healthy People provides science-based, 10-year national objectives for improving the health of all Americans. For three decades, Healthy
Appendix 1: Healthy People 2020 Immunization-related Objectives Healthy People provides science-based, 10-year national objectives for improving the health of all Americans. For three decades, Healthy
Significant events in immunisation policy and practice* in Australia
 Significant events in immunisation policy and practice* in Australia Year 1804 First vaccine (for smallpox) used in Australia 1916 Commonwealth Serum Laboratories (CSL) established in Victoria to produce
Significant events in immunisation policy and practice* in Australia Year 1804 First vaccine (for smallpox) used in Australia 1916 Commonwealth Serum Laboratories (CSL) established in Victoria to produce
3 rd dose. 3 rd or 4 th dose, see footnote 5. see footnote 13. for certain high-risk groups
 Figure 1. Recommended immunization schedule for persons aged 0 through 18 years 2013. (FOR THOSE WHO FALL BEHIND OR START LATE, SEE THE CATCH-UP SCHEDULE [FIGURE 2]). These recommendations must be read
Figure 1. Recommended immunization schedule for persons aged 0 through 18 years 2013. (FOR THOSE WHO FALL BEHIND OR START LATE, SEE THE CATCH-UP SCHEDULE [FIGURE 2]). These recommendations must be read
Vaccines for Primary Care Pneumococcal, Shingles, Pertussis
 Vaccines for Primary Care Pneumococcal, Shingles, Pertussis Devang Patel, M.D. Assistant Professor Chief of Service, MICU ID Service University of Maryland School of Medicine Pneumococcal Vaccine Pneumococcal
Vaccines for Primary Care Pneumococcal, Shingles, Pertussis Devang Patel, M.D. Assistant Professor Chief of Service, MICU ID Service University of Maryland School of Medicine Pneumococcal Vaccine Pneumococcal
COFM Immunization Policy
 COUNCIL OF ONTARIO FACULTIES OF MEDICINE An affiliate of the Council of Ontario Universities COFM Immunization Policy This policy applies to all undergraduate medical students attending an Ontario medical
COUNCIL OF ONTARIO FACULTIES OF MEDICINE An affiliate of the Council of Ontario Universities COFM Immunization Policy This policy applies to all undergraduate medical students attending an Ontario medical
Recommended Health Screenings
 Recommended Health Screenings UnitedHealthcare appreciates the preventive care you deliver to our members. Please use the below health screening chart to schedule screenings based on the member s age and
Recommended Health Screenings UnitedHealthcare appreciates the preventive care you deliver to our members. Please use the below health screening chart to schedule screenings based on the member s age and
Agenda. Richard Moriarty, MD, FAAP, Co-Director, MCAAP Immunization Initiative. Pentacel Vaccine Shortage Update MMRV Vaccine (Proquad ) Update
 Massachusetts Department of Public Health Division of Epidemiology and Immunization Agenda Introduction Richard Moriarty, MD, FAAP, Co-Director, MCAAP Immunization Initiative Pentacel Vaccine Shortage
Massachusetts Department of Public Health Division of Epidemiology and Immunization Agenda Introduction Richard Moriarty, MD, FAAP, Co-Director, MCAAP Immunization Initiative Pentacel Vaccine Shortage
Infectious Disease Surveillance in NZ. Michael Baker Department of Public Health, University of Otago, Wellington
 Infectious Disease Surveillance in NZ Michael Baker Department of Public Health, University of Otago, Wellington michael.baker@otago.ac.nz Outline Current best practice High quality surveillance of specific
Infectious Disease Surveillance in NZ Michael Baker Department of Public Health, University of Otago, Wellington michael.baker@otago.ac.nz Outline Current best practice High quality surveillance of specific
Effects of varicella vaccination on herpes zoster incidence S. Wagenpfeil 1, A. Neiss 1 and P. Wutzler 2
 REVIEW 10.1111/j.1469-0691.2004.01020.x Effects of varicella vaccination on herpes zoster incidence S. Wagenpfeil 1, A. Neiss 1 and P. Wutzler 2 1 Institute for Medical Statistics and Epidemiology, Technical
REVIEW 10.1111/j.1469-0691.2004.01020.x Effects of varicella vaccination on herpes zoster incidence S. Wagenpfeil 1, A. Neiss 1 and P. Wutzler 2 1 Institute for Medical Statistics and Epidemiology, Technical
NSW Annual Vaccine-Preventable Disease Report, 2009
 NSW Annual Vaccine-Preventable Disease Report, 29 Paula J. Spokes A,B and Robin E. Gilmour A A Communicable Diseases Branch, NSW Department of Health B Corresponding author. Email: pspok@doh.health.nsw.gov.au
NSW Annual Vaccine-Preventable Disease Report, 29 Paula J. Spokes A,B and Robin E. Gilmour A A Communicable Diseases Branch, NSW Department of Health B Corresponding author. Email: pspok@doh.health.nsw.gov.au
Wherever possible it is recommended that a child receive their vaccination on a day when they will not be attending the centre..
 Policy Hierarchy link Responsible Officer Contact Officer Superseded Documents Children (Education and Care Services National Law Application) Act 2010 Education and Care Services National Regulations
Policy Hierarchy link Responsible Officer Contact Officer Superseded Documents Children (Education and Care Services National Law Application) Act 2010 Education and Care Services National Regulations
Policy Document. Vaccination Policy. Background
 Policy Document Vaccination Policy Background The Australian Medical Students Association (AMSA) is the peak representative body of Australia s medical students. AMSA believes that all communities have
Policy Document Vaccination Policy Background The Australian Medical Students Association (AMSA) is the peak representative body of Australia s medical students. AMSA believes that all communities have
Disease and Contemporary Society
 Disease and Contemporary Society Alan Mortimer PhD Lecture 1 The notes to accompany this lecture series are provided for the educational use of the course participants. It is believed that images may be
Disease and Contemporary Society Alan Mortimer PhD Lecture 1 The notes to accompany this lecture series are provided for the educational use of the course participants. It is believed that images may be
THE EPIDEMIOLOGY OF RESPIRATORY SYNCYTIAL VIRUS INFECTIONS IN NSW CHILDREN,
 THE EPIDEMIOLOGY OF RESPIRATORY SYNCYTIAL VIRUS INFECTIONS IN NSW CHILDREN, 1992 1997 Susan Lister * and Peter McIntyre National Centre for Immunisation Research and Surveillance of Vaccine Preventable
THE EPIDEMIOLOGY OF RESPIRATORY SYNCYTIAL VIRUS INFECTIONS IN NSW CHILDREN, 1992 1997 Susan Lister * and Peter McIntyre National Centre for Immunisation Research and Surveillance of Vaccine Preventable
Quote. These are my principles. If you don t like them I have others. Groucho Marx
 Quote These are my principles. If you don t like them I have others. Groucho Marx Analysis of the Auckland 2014 Measles Outbreak Indicates that Adolescents and Young Adults could Benefit from Catch up
Quote These are my principles. If you don t like them I have others. Groucho Marx Analysis of the Auckland 2014 Measles Outbreak Indicates that Adolescents and Young Adults could Benefit from Catch up
Measles Elimination in NZ. Dr Tom Kiedrzynski Principal Adviser, Communicable diseases, Ministry of Health
 Measles Elimination in NZ Dr Tom Kiedrzynski Principal Adviser, Communicable diseases, Ministry of Health Presentation Outline International background WHO requirements Overview of the measles situation
Measles Elimination in NZ Dr Tom Kiedrzynski Principal Adviser, Communicable diseases, Ministry of Health Presentation Outline International background WHO requirements Overview of the measles situation
Manitoba Health, Healthy Living and Seniors
 Manitoba Health, Healthy Living and Seniors Manitoba Annual Immunization Surveillance Report, 2012 and 2013 January 1, 2012 to December 31, 2013 with 5-year average comparison (January 1, 2007 to December
Manitoba Health, Healthy Living and Seniors Manitoba Annual Immunization Surveillance Report, 2012 and 2013 January 1, 2012 to December 31, 2013 with 5-year average comparison (January 1, 2007 to December
Immunisation Subcommittee of PTAC Meeting held 18 February 2015
 Immunisation Subcommittee of PTAC Meeting held 18 February 2015 (minutes for web publishing) Immunisation Subcommittee minutes are published in accordance with the Terms of Reference for the Pharmacology
Immunisation Subcommittee of PTAC Meeting held 18 February 2015 (minutes for web publishing) Immunisation Subcommittee minutes are published in accordance with the Terms of Reference for the Pharmacology
INFECTIOUS DISEASES PROCEDURE
 INFECTIOUS DISEASES PROCEDURE Policy Hierarchy link Responsible Officer Contact Officer Superseded Documents Associated Documents Children (Education and Care Services National Law Application) Act 2010
INFECTIOUS DISEASES PROCEDURE Policy Hierarchy link Responsible Officer Contact Officer Superseded Documents Associated Documents Children (Education and Care Services National Law Application) Act 2010
