Analysis of Entamoeba histolytica Excretory-Secretory Antigen and Identification of a New Potential Diagnostic Marker
|
|
|
- Roger Armstrong
- 5 years ago
- Views:
Transcription
1 CLINICAL AND VACCINE IMMUNOLOGY, Nov. 2011, p Vol. 18, No /11/$12.00 doi: /cvi Copyright 2011, American Society for Microbiology. All Rights Reserved. Analysis of Entamoeba histolytica Excretory-Secretory Antigen and Identification of a New Potential Diagnostic Marker Weng Kin Wong, 1 Zi Ning Tan, 2 Nurulhasanah Othman, 1 Boon Huat Lim, 2 Zeehaida Mohamed, 3 Alfonso Olivos Garcia, 4 and Rahmah Noordin 1 * Institute for Research in Molecular Medicine, Universiti Sains Malaysia, 11800, Penang, Malaysia 1 ; School of Health Sciences, Universiti Sains Malaysia, 16150, Kelantan, Malaysia 2 ; Department of Medical Microbiology and Parasitology, School of Medical Sciences, Universiti Sains Malaysia, 16150, Kelantan, Malaysia 3 ; and Departmento de Medicina Experimental, Facultad de Medicina, Universidad Nacional Autónoma de México, Mexico D.F., Mexico 4 Received 10 August 2011/Returned for modification 2 September 2011/Accepted 7 September 2011 Serodiagnosis of amoebiasis remains the preferred method for diagnosis of amoebic liver abscess (ALA). However, the commercially available kits are problematic in areas of endemicity due to the persistently high background antibody titers. Human serum samples (n 38) from patients with ALA who live in areas of endemicity were collected from Hospital Universiti Sains Malaysia during the period of 2008 to Western blots using excretory-secretory antigen (ESA) collected from axenically grown Entamoeba histolytica were probed with the above serum samples. Seven antigenic proteins of ESA with various reactivities were identified, i.e., 152 kda, 131 kda, 123 kda, 110 kda, 100 kda, 82 kda, and 76 kda. However, only the 152-kDa and 110-kDa proteins showed sensitivities above 80% in the Western blot analysis. All the antigenic proteins showed undetectable cross-reactivity when probed with healthy human serum samples (n 30) and serum samples from other infections (n 33). From the matrix-assisted laser desorption ionization two-stage time of flight (MALDI-TOF/TOF) analysis, the proteins were identified as heavy subunits of E. histolytica lectin and E. histolytica pyruvate phosphate dikinase, respectively. Use of the E. histolytica lectin for diagnosis of ALA has been well reported by researchers and is being used in commercialized kits. However, this is the first report on the potential use of pyruvate phosphate dikinase for diagnosis of ALA; thus, this molecule merits further evaluation on its diagnostic value using a larger panel of serum samples. Amoebiasis is caused by the enteric protozoan Entamoeba histolytica, which affects 50 million people worldwide and leads to 100,000 fatal cases annually (19, 21). Amoebic liver abscess (ALA) is the most common clinical manifestation of extraintestinal amoebiasis. It is due to the hematogenous spread of the E. histolytica trophozoites from intestine to liver through the portal vein. Patients with ALA present with hepatomegaly, right-upper-quadrant pain, tenderness of the liver, fever, jaundice, and nausea. It may lead to a fatal outcome if early diagnosis and treatment are not sought (1, 10). Diagnosis of ALA is often initiated with radiology imaging to examine the presence of abscess in the liver. If indicated, aspiration of the sample is performed for culture, DNA detection, and/or antigen detection. The indications include large abscesses, superficial abscesses, abscesses with severe pain or marked point tenderness, abscesses with marked diaphragm elevation, a clinical picture suggesting impending abscess perforation, and left lobe abscess (7). Absence of bacterial growth in the abscess culture could rule out the possibility of pyogenic liver abscess cases. The definitive diagnosis of ALA is by microscopic observation of trophozoites in the abscess fluid, but the sensitivity of microscopic examination is low as the trophozoites are easily disintegrated, and most of them reside at the peripheral margin of the abscess. Many reports showed that * Corresponding author. Mailing address: Institute for Research in Molecular Medicine, Universiti Sains Malaysia, 11800, Penang, Malaysia. Phone: Fax: Published ahead of print on 14 September DNA and antigen detection-based methods performed on the abscess sample (e.g., PCR, real-time PCR, and TechLab E. histolytica II antigen detection enzyme-linked immunosorbent assay [ELISA]) had high sensitivity (4, 11, 18). In addition to imaging, serological testing is the preferred choice for diagnosis. The available antigen detection tests such as TechLab E. histolytica II ELISA, which detects E. histolytica lectin antigen, can be used for diagnosis of acute ALA patients who have not received treatment (23). However, often patients who are admitted to the hospital with liver abscess have received treatment prior to investigation for ALA, which significantly reduces the sensitivity of the antigen detection test. Thus, antibody detection is currently the most common serological test used to detect ALA, either by indirect hemagglutination assay (IHA) or ELISA. However, these tests mostly use amoebic lysate antigen and are problematic for diagnosis in areas of endemicity where the background antiamoebic antibody titer is high. Thus, in areas of endemicity, low specificities of these tests were reported with the low cutoff values suggested by the manufacturer (22, 24). Comparison of crude soluble antigen (CSA) with excretory-secretory antigen (ESA) of E. histolytica showed that ESA demonstrated a higher positive detection rate when tested with sera of patients with acute amoebic dysentery and asymptomatic cyst passers and equal sensitivity for diagnosis of ALA (10, 15). Therefore, in our quest to identify new markers to improve the serodiagnosis of ALA, ESA of E. histolytica was produced and analyzed by SDS-PAGE, two-dimensional electrophoresis (2-DE), and Western blot- 1913
2 1914 WONG ET AL. CLIN. VACCINE IMMUNOL. FIG. 1. Representative IgG blot of ESA probed with human serum samples. Lanes 1 to 7, individual ALA serum samples from group A; lane 8, pooled IHA-negative serum sample (group C); lane 9, pooled ALA serum sample (positive control); lane 10, TBS. MW, molecular weights in thousands. ting. The identities of the potential candidates were then identified by mass spectrometry. MATERIALS AND METHODS Maintenance of E. histolytica trophozoites. E. histolytica axenic strain HM1: IMSS trophozoites were hermetically cultured in TYI-S-33 medium supplemented with 12.5% bovine serum (Invitrogen, New Zealand) and 1 Diamond s vitamin-tween 80 (Sigma) at 36 C. The medium was changed every 48 to 72 h (2). Collection and preparation of ESA. Mass cultures of E. histolytica trophozoites were collected at log phase and washed three time with RPMI medium supplemented with 0.1% L-cysteine and 0.02% ascorbic acid (RPMI-C-A medium) with centrifugation at 22 g for 2 min at room temperature (RT). Subsequently, the cell density was determined via the trypan blue exclusion method. Trophozoites were seeded into a culture tube 80% filled with RPMI-C-A medium at a cell density of cells per ml and incubated at 36 C for 6 h. Using this method, we have previously shown that 95% trophozoite viability can be maintained throughout the incubation period (20). Upon completion, culture tubes were subjected to centrifugation at 22 g for 2 min at 4 C. The supernatants in the culture tubes were collected and mixed with 0.5 M iodoacetamide to a final concentration of 1 mm. Next, the supernatant was again subjected to centrifugation at 10, 000 g for 5 min at 4 C and filtered through 0.2- m-pore-size membrane. Subsequently, ESA in the supernatant was concentrated 1,000 times using a U-tube concentrator with a molecular-weight cutoff (MWCO) of 10 kda. Cocktail protease inhibitor (Roche Diagnostic, Germany) was then added to the concentrated ESA. The protein concentration of the ESA was estimated using a Bradford protein assay (10, 15, 20). Serum samples and ethical approval. Serum samples in the current study were collected from Hospital Universiti Sains Malaysia (USM) during the period of 2008 to The procedures of collecting and handling the serum samples were approved by USM Human Ethical Committee (reference number USMKK/PPP/ JEPem[213.3(10)]). Human serum samples included in this study were divided into four groups: (i) group A, human ALA serum samples (n 24) with consistent clinical symptoms (i.e., fever, abdominal/right hepatic chest pain, hepatomegaly, and jaundice) and radiological image and IHA-positive results; (ii) group B, human ALA serum samples (n 14) from patients whose abscesses were positive by real-time PCR for E. histolytica DNA and negative by bacterial culture (the primers and probe sequences were as reported previously [9]); (iii) group C, healthy blood donor serum samples which were negative by IHA (n 30); (iv) group D, serum samples from patients with other infections (n 33), i.e., salmonellosis (n 5), shigellosis (n 1), Escherichia coli septicemia (n 2), Staphylococcus sp. septicemia (n 2), Helicobacter pylori (n 6), pyogenic liver abscess (n 4), Stenotrophomonas maltophilia septicemia (n 1), enteropathogenic E. coli (n 1), Ascaris lumbricoides (n 1), Klebsiella pneumoniae (n 1), and toxoplasmosis (n 9). These sera were negative by IHA for amoebiasis. SDS-PAGE. Protein samples were electrophoretically separated via SDS- PAGE using a Bio-Rad Mini-Protean III Electrophoresis Cell and Protean II xi Cell according to the Laemmli (8) protocol, with modifications. Prior to SDS- PAGE, ESA was mixed with 2 Laemmli sample buffer and boiled for 5 min. FIG. 2. Representative IgG blot of ESA when probed with human serum samples. Lane 1, TBS; lane 2, pooled ALA serum sample (positive control); lane 3, pooled IHA-negative serum sample (group D); lanes 4 to 10, individual ALA serum samples from group B. MW, molecular weights in thousands. Subsequently, ESA was separated using a 6% SDS-PAGE gel at a constant current of 25 ma per gel for about 1 h. Western blotting. Upon completion of SDS-PAGE, proteins in the gel were electrophoretically transferred onto a m-pore-size nitrocellulose paper (NCP) membrane using a semidry transblot apparatus (Bio-Rad) at a constant voltage of 15 V for 30 min. The NCP was blocked for 1hatRTwith 5% skim milk prepared in 10 mm Tris-buffered saline (TBS), ph 7.2. Subsequently, the NCP was washed (three times for 5 min each time) with TBS containing 0.1% Tween 20 (TBS-T). Then, the NCP was cut into multiple strips and incubated with human serum at a dilution of 1:200 (in TBS-T) for2hatrt.thencpstrips were then washed three times with TBS-T and then incubated with monoclonal mouse anti-human IgG conjugated with horseradish peroxides (HRP) at a dilution of 1:6,000 for 1 h. Subsequently, the NCP strips were again washed (three times for 5 min each time) with TBS-T. Western blot substrates, i.e., enhanced chemiluminescence (ECL) blotting reagent (Roche Diagnostics, Germany) or tetramethylbenzidine (TMB) substrate for membrane (Sigma), were used as substrates. The Western blot signal was captured using a camera (Lumix, Germany). 2-DE and Western blotting. Selected protein bands which showed potential diagnostic value were further analyzed using 2-DE to ensure that the bands were well separated. An Offgel Fractionator 3100 (Agilent Technologies, Germany) was used to separate the proteins by isoelectric point (pi) followed by SDS-PAGE and Western blotting. An Agilent Offgel kit, ph 3 to 10, with a 12-well setup frame was used. Sample was prepared by mixing 1,600 l of the ready stock solution (1.25 ) with 400 l of the sample with a total protein amount of 2 mg and then gently vortexed. Forty microliters of immobilized ph gradient (IPG) strip rehydration buffer was added into each well to swell the gel for 15 min. Wetted electrode pads were placed at the cathode and anode ends of the IPG strip gel surface. After reswelling of the gel, 150 l of protein sample was loaded into each well. Ten microliters of rehydration buffer was reapplied onto the electrode pads at each of the IPG gel ends. Cover fluid (mineral oil) was pipetted onto the gel strip ends. Subsequently, the sample was focused with a maximum power of 200 mw, maximum current of 50 ma, and typical voltages ranging from 500 to 4,500 V until 50 kvh was reached after 24 h. Upon completion, each of the 12 fractionated ESA samples was separately mixed with 5 Laemmli sample buffer without boiling and electrophoretically separated via SDS-PAGE. Western blotting was performed using pooled and individual human serum samples to identify the selected antigenic proteins. Mass spectrometry analysis and protein identification. The selected proteins were excised from a 2-D SDS-PAGE gel and sent for matrix-assisted laser desorption ionization two-stage time of flight ([MALDI-TOF/TOF] ABI 4800 instrument) analysis at the Proteomic Laboratory Service Center, Australia, and searched with a Swiss-Prot protein database.
3 VOL. 18, 2011 ENTAMOEBA HISTOLYTICA ESA OF DIAGNOSTIC POTENTIAL 1915 TABLE 1. Sensitivity and specificity of antigenic bands of ESA from IgG blots Antigenic band (kda) c Sensitivity (no. of positive tests % ) a Specificity (no. of negative tests % ) b (84) (45) (16) (82) (18) (39) (39) 100 a n 38, total number of tests within the groups with ALA (groups A and B). b n 63, total number of tests within the groups without ALA (groups C and D). c The bands with sensitivities above 80% are shown in boldface. RESULTS IgG blots of ESA. IgG blots of ESA probed with human ALA serum samples from group A showed seven antigenic bands with consistent reactivities (Fig. 1). In addition, these antigenic proteins were also similar to the bands present in the IgG blots probed with human ALA serum samples from group B (Fig. 2). However, mean sensitivities of the bands to detect ALA varied from 16% to 84%. Only two of the antigenic bands, i.e., 152-kDa and 110-kDa bands, showed high sensitivities of above 80% (Table 1). Neither of these two antigenic bands showed reactivity in IgG blots probed with serum samples from groups C and D, thus showing 100% specificity. In this study, sensitivity is defined as the number of serum samples which reacted with the band out of the total number of samples from ALA patients, i.e., group A and B sera. Meanwhile, specificity is defined as the number of serum samples which do not react with the band out of the total number of samples from individuals without ALA, i.e., group C and D sera. 2-DE Western blotting and protein identification. IgG blotting of 12 ESA fractions with pooled serum samples revealed FIG. 4. IgG blot of ESA separated by 2-DE and probed with human serum samples. H1 to H5, individual ALA serum samples from group B; HP, pooled ALA serum sample (positive control); HP, pooled IHA-negative serum sample (group D). Ctrl, control; MW, molecular weights in thousands. pi values are indicated for the fractions. that the 152-kDa and 110-kDa proteins were located in fraction 5 (pi 5.33 to 5.91) and fraction 6 (pi 5.91 to 6.5), respectively (Fig. 3). Further IgG blot analysis of these ESA fractions with individual serum samples (n 5) confirmed the location of these antigenic proteins (Fig. 4). These protein bands were excised and sent for MALDI-TOF/TOF analysis. According to the Mascot search result from the mass spectrometry database (MSDB) search engine, the 152-kDa protein matched the E. histolytica lectin protein (C4LTMO) with a protein score of 273. A score of 55 indicates identity or extensive homology at FIG. 3. IgG blot of ESA separated by 2-DE and probed with human serum samples. H, pooled positive ALA serum sample from group B; H, pooled IHA-negative serum sample (group C). MW, molecular weights in thousands. pi values are indicated for each fraction.
4 1916 WONG ET AL. CLIN. VACCINE IMMUNOL. a significant level (P 0.05). Seven peptides matched to the Gal/GalNAc lectin heavy subunit. The 110-kDa protein matched pyruvate phosphate dikinase (EHI_009530) with a protein score of 544, with nine matched peptides. Since pyruvate phosphate dikinase has not been reported as a diagnostic protein, the 110-kDa bands from separate gels were sent three times for the MALDI-TOF/TOF analysis, and same results were obtained each time. DISCUSSION Current diagnosis of ALA still depends on the results of clinical manifestations, radiology imaging, and serology since stool examination is inapplicable for diagnosis of extraintestinal amoebiasis and abscess aspiration is performed only when indicated. E. histolytica ESA contains proteins shed from trophozoites during active multiplication and metabolites released by trophozoites during incubation in RPMI-C-A medium. Although great care was taken to produce good quality ESA, there were probably still some partial proteins released from lysed trophozoites. In this study, the ESA antigenic bands that ranged from 97.2 to 158 kda consistently showed reactivities when incubated with human ALA serum samples (n 7). Analysis of the IgG blots showed that 152-kDa and 110-kDa proteins had higher association with serum samples from patients with PCR-positive abscess, i.e., 13/14 (93%) and 12/14 (86%), respectively, than serum samples from patients with unknown PCR results, i.e., 19/24 (79%) for both bands. In addition, the sensitivities of both the 152-kDa and 110-kDa bands were similar, i.e., 32/38 (84%) and 31/38 (82%), respectively. Both antigens showed high specificity as there were undetectable reactivities in the IgG blot of ESA probed with serum samples from healthy individuals and from those with other infections. The protein components of ESA in the current study were different from those reported by Sengupta et al. (15). In the latter study, the ESA proteins ranged between 200 to 20 kda, with the predominant protein bands below 66 kda (e.g., 45 kda and 29 kda), while the high-molecular-weight proteins ( 100 kda) were faint. This may be due to the differences in antigen preparations. The ESA in this study was concentrated 1,000 times instead of 10 times to enrich the low-abundance proteins. In addition, to enhance the reproducibility of ESA, protein-free defined RPMI-C-A medium was used in this study instead of serum- and vitamin-free TYI-S-33 medium. In addition, iodoacetamide was added to the RPMI-C-A medium containing the ESA upon its collection in order to protect the protein from degradation by proteases (3, 13, 15). Although we have previously optimized the growth conditions of E. histolytica trophozoites so that 95% of the parasites were alive when ESA was collected (20), nevertheless it is still possible that some of the antigenic bands may have come from ruptured trophozoites. 2-DE protein separation via an Agilent 3100 Offgel Fractionator followed by SDS-PAGE allowed only the selected ESA fractions to be tested with serum samples. The protein bands excised from the 2-DE gels were well separated, thus avoiding the presence of multiple proteins in each band. In this study, the 152-kDa protein was identified as an E. histolytica lectin protein, which has been reported to be sensitive for diagnosis of invasive amoebiasis (5, 6). Specific monoclonal antibody against this protein has been used for antigen detection in a TechLab Entamoeba histolytica II kit (TechLab Inc.). The 110-kDa protein, identified as the E. histolytica pyruvate phosphate dikinase, was also found to show similarly high sensitivity for diagnosis of ALA. This protein was reported to be a key enzyme in the anaerobic metabolism via pyrophosphate-dependent glycolysis and has no counterpart with proteins in human metabolism (14). Molecular modeling of this enzyme had been reported, and specific inhibitors to it for therapeutic purpose have been studied (17). The protein sequence of pyruvate phosphate dikinase showed high similarity with a closely related pathogenic intestinal anaerobic protozoon, Giardia lamblia. This suggests the possibility of producing specific antibody to pyruvate phosphate dikinase for simultaneous detection of both E. histolytica and G. lamblia species in fecal samples (14). To date, there is no report on the application of pyruvate phosphate kinase for diagnosis of amoebiasis. Nevertheless, to confirm the diagnostic value of pyruvate phosphate dikinase, it would be necessary to produce the recombinant form of the protein and then demonstrate that it is recognized by patient serum. This is indeed our aim for future research. ACKNOWLEDGMENTS This study was funded by FRGS grant number 203/CIPPM/ , USM-RU grant number 1001/PPSK/813009, and USM-RU-PGRS grant number 1001/INFORMM/ W.W.K. is a recipient of a USM Fellowship, and N.O. received financial support from the Vice Chancellor s Award. REFERENCES 1. Akgun, Y., I. H. Tacyildiz, and Y. Celik Amebic liver abscess: changing trends over 20 years. World J. Surg. 23: Diamond, L. S., D. R. Harlow, and C. C. Cunnick A new medium for the axenic cultivation of Entamoeba histolytica and other Entamoeba. Trans. R. Soc. Trop. Med. Hyg. 72: Flores, M. S., et al Preparation of Entamoeba histolytica antigens without enzymatic inhibitors. Parasitology 131: Fotedar, R., et al Laboratory diagnostic techniques for Entamoeba species. Clin. Microbiol. Rev. 20: Haque, R., et al Entamoeba histolytica and Entamoeba dispar infection in children in Bangladesh. J. Infect. Dis. 175: Haque, R., and W. A. Petri, Jr Diagnosis of amebiasis in Bangladesh. Arch. Med. Res. 37: Kapoor, O. P Amoebic liver abscess. S. S. Publishers, Bombay, India Laemmli, U. K Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227: Othman, N., et al Application of real-time polymerase chain reaction in detection of Entamoeba histolytica in pus aspirates of amoebic liver abscess patients. Foodborne Pathog. Dis. 7: Pal, S., et al Comparative evaluation of somatic and excretory-secretory antigens of Entamoeba histolytica in serodiagnosis of human amoebiasis by ELISA. Indian J. Med. Res. 104: Paul, J., S. Srivastava, and S. Bhattacharya Molecular methods for diagnosis of Entamoeba histolytica in a clinical setting: an overview. Exp. Parasitol. 116: Reference deleted. 13. Que, X., and S. L. Reed Cysteine proteinases and the pathogenesis of amebiasis. Clin. Microbiol. Rev. 13: Saavedra-Lira, E., L. Ramirez-Silva, and R. Perez-Montfort Expression and characterization of recombinant pyruvate phosphate dikinase from Entamoeba histolytica. Biochim. Biophys. Acta 1382: Sengupta, S., et al Role of excretory-secretory products of Entamoeba histolytica in human amebiasis. Arch. Med. Res. 31:S226 S Reference deleted. 17. Stephen, P., et al Molecular modeling on pyruvate phosphate dikinase of Entamoeba histolytica and in silico virtual screening for novel inhibitors. J. Comput. Aided Mol. Des. 22: Tanyuksel, M., and W. A. Petri, Jr Laboratory diagnosis of amebiasis. Clin. Microbiol. Rev. 16:
5 VOL. 18, 2011 ENTAMOEBA HISTOLYTICA ESA OF DIAGNOSTIC POTENTIAL Walsh, J. A Problems in recognition and diagnosis of amebiasis: estimation of the global magnitude of morbidity and mortality. Rev. Infect. Dis. 8: Wong, W. K., et al Comparison of protein-free defined media, and effect of L-cysteine and ascorbic acid supplementation on viability of axenic Entamoeba histolytica. Parasitol. Res. 108: World Health Organization WHO/PAHO/UNESCO report. A consultation with experts on amoebiasis. Mexico City, Mexico January, Epidemiol. Bull. 18: Zambrano-Villa, S. et al How protozoan parasites evade the immune response. Trends Parasitol. 18: Zeehaida, M., et al A study on the usefulness of TechLab Entamoeba histolytica II antigen detection ELISA in the diagnosis of amoebic liver abscess (ALA) at Hospital Universiti Sains Malaysia (HUSM), Kelantan, Malaysia. Trop. Biomed. 25: Zeehaida, M., et al Analysis of indirect hemagglutination assay results among patients with amoebic liver abscess. Int. Med. J. 16:
PARASITOLOGY CASE HISTORY 15 (HISTOLOGY) (Lynne S. Garcia)
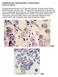 PARASITOLOGY CASE HISTORY 15 (HISTOLOGY) (Lynne S. Garcia) A biopsy was performed on a 27-year-old man with no known travel history, presenting with a perianal ulcer. The specimen was preserved in formalin
PARASITOLOGY CASE HISTORY 15 (HISTOLOGY) (Lynne S. Garcia) A biopsy was performed on a 27-year-old man with no known travel history, presenting with a perianal ulcer. The specimen was preserved in formalin
E. Histolytica IgG (Amebiasis)
 DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and 116, Woodland hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and 116, Woodland hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
INTERNATIONAL JOURNAL OF PHARMACEUTICAL RESEARCH AND BIO-SCIENCE
 INTERNATIONAL JOURNAL OF PHARMACEUTICAL RESEARCH AND BIO-SCIENCE SEROPREVALENCE OF AMOEBIASIS IN A TERTIARY CARE HOSPITAL DEEPINDER CHHINA, VEENU GUPTA, JASDEEP SINGH, POOJA SURI Department of Microbiology,
INTERNATIONAL JOURNAL OF PHARMACEUTICAL RESEARCH AND BIO-SCIENCE SEROPREVALENCE OF AMOEBIASIS IN A TERTIARY CARE HOSPITAL DEEPINDER CHHINA, VEENU GUPTA, JASDEEP SINGH, POOJA SURI Department of Microbiology,
Protocol for Gene Transfection & Western Blotting
 The schedule and the manual of basic techniques for cell culture Advanced Protocol for Gene Transfection & Western Blotting Schedule Day 1 26/07/2008 Transfection Day 3 28/07/2008 Cell lysis Immunoprecipitation
The schedule and the manual of basic techniques for cell culture Advanced Protocol for Gene Transfection & Western Blotting Schedule Day 1 26/07/2008 Transfection Day 3 28/07/2008 Cell lysis Immunoprecipitation
E. Histolytica IgG ELISA Kit
 E. Histolytica IgG ELISA Kit Catalog Number KA3193 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
E. Histolytica IgG ELISA Kit Catalog Number KA3193 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
Entamoeba histolytica/e. dispar. A. Haghighi,
 Entamoeba histolytica/e. dispar A. Haghighi, Wednesday, February 14, 2018 Classification of Protozoa? The protozoa are generally unicellular and may be divided for convenience, into four distinct groups
Entamoeba histolytica/e. dispar A. Haghighi, Wednesday, February 14, 2018 Classification of Protozoa? The protozoa are generally unicellular and may be divided for convenience, into four distinct groups
Amebiasis Ab E. histolytica IgG
 Code: HP003 96 Test distribuito da: Amebiasis Ab E. histolytica IgG Amebiasis E. histolytica lgg test is an enzyme immunoassay for the detection of specific lgg antibodies against Anti - Entamoeba histolytica
Code: HP003 96 Test distribuito da: Amebiasis Ab E. histolytica IgG Amebiasis E. histolytica lgg test is an enzyme immunoassay for the detection of specific lgg antibodies against Anti - Entamoeba histolytica
The Schedule and the Manual of Basic Techniques for Cell Culture
 The Schedule and the Manual of Basic Techniques for Cell Culture 1 Materials Calcium Phosphate Transfection Kit: Invitrogen Cat.No.K2780-01 Falcon tube (Cat No.35-2054:12 x 75 mm, 5 ml tube) Cell: 293
The Schedule and the Manual of Basic Techniques for Cell Culture 1 Materials Calcium Phosphate Transfection Kit: Invitrogen Cat.No.K2780-01 Falcon tube (Cat No.35-2054:12 x 75 mm, 5 ml tube) Cell: 293
Bacillary Dysentery (Shigellosis)
 Bacillary Dysentery (Shigellosis) An acute bacterial disease involving the large and distal small intestine, caused by the bacteria of the genus shigella. Infectious agent Shigella is comprised of four
Bacillary Dysentery (Shigellosis) An acute bacterial disease involving the large and distal small intestine, caused by the bacteria of the genus shigella. Infectious agent Shigella is comprised of four
TSH Receptor Monoclonal Antibody (49) Catalog Number MA3-218 Product data sheet
 Website: thermofisher.com Customer Service (US): 1 800 955 6288 ext. 1 Technical Support (US): 1 800 955 6288 ext. 441 TSH Receptor Monoclonal Antibody (49) Catalog Number MA3-218 Product data sheet Details
Website: thermofisher.com Customer Service (US): 1 800 955 6288 ext. 1 Technical Support (US): 1 800 955 6288 ext. 441 TSH Receptor Monoclonal Antibody (49) Catalog Number MA3-218 Product data sheet Details
Protein MultiColor Stable, Low Range
 Product Name: DynaMarker Protein MultiColor Stable, Low Range Code No: DM670L Lot No: ******* Size: 200 μl x 3 (DM670 x 3) (120 mini-gel lanes) Storage: 4 C Stability: 12 months at 4 C Storage Buffer:
Product Name: DynaMarker Protein MultiColor Stable, Low Range Code No: DM670L Lot No: ******* Size: 200 μl x 3 (DM670 x 3) (120 mini-gel lanes) Storage: 4 C Stability: 12 months at 4 C Storage Buffer:
Human HBcAb IgM ELISA kit
 Human HBcAb IgM ELISA kit Catalog number: NR-R10163 (96 wells) The kit is designed to qualitatively detect HBcAb IgM in human serum or plasma. FOR RESEARCH USE ONLY. NOT FOR DIAGNOSTIC OR THERAPEUTIC PURPOSES
Human HBcAb IgM ELISA kit Catalog number: NR-R10163 (96 wells) The kit is designed to qualitatively detect HBcAb IgM in human serum or plasma. FOR RESEARCH USE ONLY. NOT FOR DIAGNOSTIC OR THERAPEUTIC PURPOSES
Giardia lamblia ELISA Kit
 Giardia lamblia ELISA Kit Catalog Number KA2093 96 assay Version: 01 Intend for research use only www.abnova.com Introduction and Background A. Intended Use Giardia lamblia ELISA Kit is an Enzyme-Linked
Giardia lamblia ELISA Kit Catalog Number KA2093 96 assay Version: 01 Intend for research use only www.abnova.com Introduction and Background A. Intended Use Giardia lamblia ELISA Kit is an Enzyme-Linked
RayBio KinaseSTAR TM Akt Activity Assay Kit
 Activity Assay Kit User Manual Version 1.0 March 13, 2015 RayBio KinaseSTAR TM Akt Activity Kit Protocol (Cat#: 68AT-Akt-S40) RayBiotech, Inc. We Provide You With Excellent Support And Service Tel:(Toll
Activity Assay Kit User Manual Version 1.0 March 13, 2015 RayBio KinaseSTAR TM Akt Activity Kit Protocol (Cat#: 68AT-Akt-S40) RayBiotech, Inc. We Provide You With Excellent Support And Service Tel:(Toll
2009 H1N1 Influenza ( Swine Flu ) Hemagglutinin ELISA kit
 2009 H1N1 Influenza ( Swine Flu ) Hemagglutinin ELISA kit Catalog Number : SEK001 To achieve the best assay results, this manual must be read carefully before using this product and the assay is run as
2009 H1N1 Influenza ( Swine Flu ) Hemagglutinin ELISA kit Catalog Number : SEK001 To achieve the best assay results, this manual must be read carefully before using this product and the assay is run as
Entamoeba histolytica
 Entamoeba histolytica cosmopolitan distribution no animal reservoirs facultative pathogen most clear the infection spontaneous in 6-12 months with mild or no symptoms can cause a serious invasive disease
Entamoeba histolytica cosmopolitan distribution no animal reservoirs facultative pathogen most clear the infection spontaneous in 6-12 months with mild or no symptoms can cause a serious invasive disease
Cryptosporidium spp. ELISA Kit
 Cryptosporidium spp. ELISA Kit Catalog Number KA2094 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Cryptosporidium spp. ELISA Kit Catalog Number KA2094 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
HiPer Western Blotting Teaching Kit
 HiPer Western Blotting Teaching Kit Product Code: HTI009 Number of experiments that can be performed: 5/20 Duration of Experiment: ~ 2 days Day 1: 6-8 hours (SDS- PAGE and Electroblotting) Day 2: 3 hours
HiPer Western Blotting Teaching Kit Product Code: HTI009 Number of experiments that can be performed: 5/20 Duration of Experiment: ~ 2 days Day 1: 6-8 hours (SDS- PAGE and Electroblotting) Day 2: 3 hours
Influenza A H1N1 (Swine Flu 2009) Hemagglutinin / HA ELISA Pair Set
 Influenza A H1N1 (Swine Flu 2009) Hemagglutinin / HA ELISA Pair Set Catalog Number : SEK001 To achieve the best assay results, this manual must be read carefully before using this product and the assay
Influenza A H1N1 (Swine Flu 2009) Hemagglutinin / HA ELISA Pair Set Catalog Number : SEK001 To achieve the best assay results, this manual must be read carefully before using this product and the assay
Diagnosis of Amebic Liver Abscess and Intestinal Infection with the TechLab Entamoeba histolytica II Antigen Detection and Antibody Tests
 JOURNAL OF CLINICAL MICROBIOLOGY, Sept. 2000, p. 3235 3239 Vol. 38, No. 9 0095-1137/00/$04.00 0 Copyright 2000, American Society for Microbiology. All Rights Reserved. Diagnosis of Amebic Liver Abscess
JOURNAL OF CLINICAL MICROBIOLOGY, Sept. 2000, p. 3235 3239 Vol. 38, No. 9 0095-1137/00/$04.00 0 Copyright 2000, American Society for Microbiology. All Rights Reserved. Diagnosis of Amebic Liver Abscess
Recent Diagnostic Methods for Intestinal Parasitic Infections
 Recent Diagnostic Methods for Intestinal Parasitic Infections By Dr. Doaa Abdel Badie Salem Lecturer of Medical Parasitology, Mansoura Faculty of Medicine Agenda Intestinal parasites. Traditional Diagnostic
Recent Diagnostic Methods for Intestinal Parasitic Infections By Dr. Doaa Abdel Badie Salem Lecturer of Medical Parasitology, Mansoura Faculty of Medicine Agenda Intestinal parasites. Traditional Diagnostic
Human Immunodeficiency Virus type 1 (HIV-1) p24 / Capsid Protein p24 ELISA Pair Set
 Human Immunodeficiency Virus type 1 (HIV-1) p24 / Capsid Protein p24 ELISA Pair Set Catalog Number : SEK11695 To achieve the best assay results, this manual must be read carefully before using this product
Human Immunodeficiency Virus type 1 (HIV-1) p24 / Capsid Protein p24 ELISA Pair Set Catalog Number : SEK11695 To achieve the best assay results, this manual must be read carefully before using this product
INVASIVE AMEBIASIS: CHALLENGES IN DIAGNOSIS IN A NON-ENDEMIC COUNTRY (KUWAIT)
 Am. J. Trop. Med. Hyg., 65(4), 200, pp. 34 345 Copyright 200 by The American Society of Tropical Medicine and Hygiene INVASIVE AMEBIASIS: CHALLENGES IN DIAGNOSIS IN A NON-ENDEMIC COUNTRY (KUWAIT) PERSOTUM
Am. J. Trop. Med. Hyg., 65(4), 200, pp. 34 345 Copyright 200 by The American Society of Tropical Medicine and Hygiene INVASIVE AMEBIASIS: CHALLENGES IN DIAGNOSIS IN A NON-ENDEMIC COUNTRY (KUWAIT) PERSOTUM
Influenza A H1N1 HA ELISA Pair Set
 Influenza A H1N1 HA ELISA Pair Set for H1N1 ( A/Puerto Rico/8/1934 ) HA Catalog Number : SEK11684 To achieve the best assay results, this manual must be read carefully before using this product and the
Influenza A H1N1 HA ELISA Pair Set for H1N1 ( A/Puerto Rico/8/1934 ) HA Catalog Number : SEK11684 To achieve the best assay results, this manual must be read carefully before using this product and the
Problem in Amoebiasis Diagnosis in Clinical Setting: A Review from Conventional Microscopy to Advanced Molecular Based Diagnosis
 Volume 2, Issue 5 October 2013 257 REVIEW ARTICLE ISSN: 2278-5213 Problem in Amoebiasis Diagnosis in Clinical Setting: A Review from Conventional Microscopy to Advanced Molecular Based Diagnosis Joyobrato
Volume 2, Issue 5 October 2013 257 REVIEW ARTICLE ISSN: 2278-5213 Problem in Amoebiasis Diagnosis in Clinical Setting: A Review from Conventional Microscopy to Advanced Molecular Based Diagnosis Joyobrato
Protease Activity in Giardia duodenalis Trophozoites of Axenic Strains Isolated from Symptomatic and Asymptomatic Patients
 Mem Inst Oswaldo Cruz, Rio de Janeiro, Vol. 98: 000-000, 2003 1 Protease Activity in Giardia duodenalis Trophozoites of Axenic Strains Isolated from Symptomatic and Asymptomatic Patients Semíramis Guimarães/
Mem Inst Oswaldo Cruz, Rio de Janeiro, Vol. 98: 000-000, 2003 1 Protease Activity in Giardia duodenalis Trophozoites of Axenic Strains Isolated from Symptomatic and Asymptomatic Patients Semíramis Guimarães/
Mouse Cathepsin B ELISA Kit
 GenWay Biotech, Inc. 6777 Nancy Ridge Drive San Diego, CA 92121 Phone: 858.458.0866 Fax: 858.458.0833 Email: techline@genwaybio.com http://www.genwaybio.com Mouse Cathepsin B ELISA Kit Catalog No. GWB-ZZD154
GenWay Biotech, Inc. 6777 Nancy Ridge Drive San Diego, CA 92121 Phone: 858.458.0866 Fax: 858.458.0833 Email: techline@genwaybio.com http://www.genwaybio.com Mouse Cathepsin B ELISA Kit Catalog No. GWB-ZZD154
Evaluation of Recombinant Fragments of Entamoeba histolytica Gal/GalNAc Lectin Intermediate Subunit for Serodiagnosis of Amebiasis
 JOURNAL OF CLINICAL MICROBIOLOGY, Mar. 2004, p. 1069 1074 Vol. 42, No. 3 0095-1137/04/$08.00 0 DOI: 10.1128/JCM.42.3.1069 1074.2004 Copyright 2004, American Society for Microbiology. All Rights Reserved.
JOURNAL OF CLINICAL MICROBIOLOGY, Mar. 2004, p. 1069 1074 Vol. 42, No. 3 0095-1137/04/$08.00 0 DOI: 10.1128/JCM.42.3.1069 1074.2004 Copyright 2004, American Society for Microbiology. All Rights Reserved.
RIDA QUICK Entamoeba. Article no: N1702
 RIDA QUICK Entamoeba Article no: N1702 R-Biopharm AG, An der neuen Bergstraße 17, D-64297 Darmstadt, Germany Phone: +49 (0) 61 51 81 02-0 / Fax: +49 (0) 61 51 81 02-20 1. Intended use For in vitro diagnostic
RIDA QUICK Entamoeba Article no: N1702 R-Biopharm AG, An der neuen Bergstraße 17, D-64297 Darmstadt, Germany Phone: +49 (0) 61 51 81 02-0 / Fax: +49 (0) 61 51 81 02-20 1. Intended use For in vitro diagnostic
Amoebiasis. (Amoebic dysentery)
 Amoebiasis (Amoebic dysentery) Causative agent: Entamoeba histolytica Amoebiasis Harbouring of protozoa E. histolytica inside the body with or without disease only 10% of infected develop disease two types
Amoebiasis (Amoebic dysentery) Causative agent: Entamoeba histolytica Amoebiasis Harbouring of protozoa E. histolytica inside the body with or without disease only 10% of infected develop disease two types
Human Urokinase / PLAU / UPA ELISA Pair Set
 Human Urokinase / PLAU / UPA ELISA Pair Set Catalog Number : SEK10815 To achieve the best assay results, this manual must be read carefully before using this product and the assay is run as summarized
Human Urokinase / PLAU / UPA ELISA Pair Set Catalog Number : SEK10815 To achieve the best assay results, this manual must be read carefully before using this product and the assay is run as summarized
Characteristics of amebic liver abscess in patients with or without human immunodeficiency virus
 J Microbiol Immunol Infect. 2009;42:500-504 Characteristics of amebic liver abscess in patients with or without human virus Kuan-Jung Chen 1, Chin-Hui Yang 2,3, Yi-Chun Lin 4, Hsin-Yi Liu 5, Say-Tsung
J Microbiol Immunol Infect. 2009;42:500-504 Characteristics of amebic liver abscess in patients with or without human virus Kuan-Jung Chen 1, Chin-Hui Yang 2,3, Yi-Chun Lin 4, Hsin-Yi Liu 5, Say-Tsung
HIV-1 p24 ELISA Pair Set Cat#: orb54951 (ELISA Manual)
 HIV-1 p24 ELISA Pair Set Cat#: orb54951 (ELISA Manual) BACKGROUND Human Immunodeficiency Virus ( HIV ) can be divided into two major types, HIV type 1 (HIV-1) and HIV type 2 (HIV-2). HIV-1 is related to
HIV-1 p24 ELISA Pair Set Cat#: orb54951 (ELISA Manual) BACKGROUND Human Immunodeficiency Virus ( HIV ) can be divided into two major types, HIV type 1 (HIV-1) and HIV type 2 (HIV-2). HIV-1 is related to
Foodborne Disease in the Region of Peel
 Foodborne Disease in the Region of Peel HIGHLIGHTS The incidence of selected foodborne diseases was generally higher in Peel than in Ontario between 1993 and 22. A higher incidence was observed in Peel
Foodborne Disease in the Region of Peel HIGHLIGHTS The incidence of selected foodborne diseases was generally higher in Peel than in Ontario between 1993 and 22. A higher incidence was observed in Peel
Pathogens of the Digestive System
 Pathogens of the Digestive System Chapter 24 (Pages 625-661) 1. Digestive System Review (Pages 627-629) A. Oral Cavity B. Esophagus C. Stomach D. Small Intestine E. Pancreas F. Liver G. Gall Bladder H.
Pathogens of the Digestive System Chapter 24 (Pages 625-661) 1. Digestive System Review (Pages 627-629) A. Oral Cavity B. Esophagus C. Stomach D. Small Intestine E. Pancreas F. Liver G. Gall Bladder H.
Trichinella Cat #
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
SM Shamsuzzaman 1,2, Rashidul Haque 3, SK Ruhul Hasin 4, William A Petri Jr 5 and Yoshihisa Hashiguchi 2
 HEPATIC AMEBIASIS IN BANGLADESH SOCIOECONOMIC STATUS, CLINICAL FEATURES, LABORATORY AND PARASITOLOGICAL FINDINGS OF HEPATIC AMEBIASIS PATIENTS - A HOSPITAL BASED PROSPECTIVE STUDY IN BANGLADESH SM Shamsuzzaman
HEPATIC AMEBIASIS IN BANGLADESH SOCIOECONOMIC STATUS, CLINICAL FEATURES, LABORATORY AND PARASITOLOGICAL FINDINGS OF HEPATIC AMEBIASIS PATIENTS - A HOSPITAL BASED PROSPECTIVE STUDY IN BANGLADESH SM Shamsuzzaman
PREVALENCE OF DIFFERENT PROTOZOAN PARASITES IN PATIENTS VISITING AT ICDDR B HOSPITAL, DHAKA
 J. Asiat. Soc. Bangladesh, Sci. 39(1): 117-123, June 213 PREVALENCE OF DIFFERENT PROTOZOAN PARASITES IN PATIENTS VISITING AT ICDDR B HOSPITAL, DHAKA SHAHELA ALAM, HAMIDA KHANUM 1, RIMI FARHANA ZAMAN AND
J. Asiat. Soc. Bangladesh, Sci. 39(1): 117-123, June 213 PREVALENCE OF DIFFERENT PROTOZOAN PARASITES IN PATIENTS VISITING AT ICDDR B HOSPITAL, DHAKA SHAHELA ALAM, HAMIDA KHANUM 1, RIMI FARHANA ZAMAN AND
RIDA QUICK Entamoeba. Article no: N 1703
 RIDA QUICK Entamoeba Article no: N 1703 R-Biopharm AG, Landwehrstr. 54, D-64293 Darmstadt, Germany Tel: +49 (0) 61 51 81 02-0 / Fax: +49 (0) 61 51 81 02-20 1. Intended use For in vitro diagnostic use.
RIDA QUICK Entamoeba Article no: N 1703 R-Biopharm AG, Landwehrstr. 54, D-64293 Darmstadt, Germany Tel: +49 (0) 61 51 81 02-0 / Fax: +49 (0) 61 51 81 02-20 1. Intended use For in vitro diagnostic use.
Cell Lysis Buffer. Catalog number: AR0103
 Cell Lysis Buffer Catalog number: AR0103 Boster s Cell Lysis Buffer is a ready-to-use Western blot related reagent solution used for efficient extraction of total soluble protein in nondenatured state
Cell Lysis Buffer Catalog number: AR0103 Boster s Cell Lysis Buffer is a ready-to-use Western blot related reagent solution used for efficient extraction of total soluble protein in nondenatured state
HIV-1 p24 Antigen ELISA Catalog Number:
 INTENDED USE The RETRO-TEK HIV-1 p24 Antigen ELISA is supplied for research purposes only. It is not intended for use in the diagnosis or prognosis of disease, or for screening and may not be used as a
INTENDED USE The RETRO-TEK HIV-1 p24 Antigen ELISA is supplied for research purposes only. It is not intended for use in the diagnosis or prognosis of disease, or for screening and may not be used as a
Human LDL Receptor / LDLR ELISA Pair Set
 Human LDL Receptor / LDLR ELISA Pair Set Catalog Number : SEK10231 To achieve the best assay results, this manual must be read carefully before using this product and the assay is run as summarized in
Human LDL Receptor / LDLR ELISA Pair Set Catalog Number : SEK10231 To achieve the best assay results, this manual must be read carefully before using this product and the assay is run as summarized in
Toxocara. Cat # Toxocara ELISA. Enzyme Linked Immunosorbent Assay. ELISA-Indirect; Antigen Coated Plate
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Amoebiasis: current status in Australia
 Amoebiasis: current status in Australia Sebastiaan J van Hal, Damien J Stark, Rashmi Fotedar, Debbie Marriott, John T Ellis and Jock L Harkness Amoebiasis, a disease caused by the intestinal protozoan
Amoebiasis: current status in Australia Sebastiaan J van Hal, Damien J Stark, Rashmi Fotedar, Debbie Marriott, John T Ellis and Jock L Harkness Amoebiasis, a disease caused by the intestinal protozoan
Islet viability assay and Glucose Stimulated Insulin Secretion assay RT-PCR and Western Blot
 Islet viability assay and Glucose Stimulated Insulin Secretion assay Islet cell viability was determined by colorimetric (3-(4,5-dimethylthiazol-2-yl)-2,5- diphenyltetrazolium bromide assay using CellTiter
Islet viability assay and Glucose Stimulated Insulin Secretion assay Islet cell viability was determined by colorimetric (3-(4,5-dimethylthiazol-2-yl)-2,5- diphenyltetrazolium bromide assay using CellTiter
Mammalian Tissue Protein Extraction Reagent
 Mammalian Tissue Protein Extraction Reagent Catalog number: AR0101 Boster s Mammalian Tissue Protein Extraction Reagent is a ready-to-use Western blot related reagent solution used for efficient extraction
Mammalian Tissue Protein Extraction Reagent Catalog number: AR0101 Boster s Mammalian Tissue Protein Extraction Reagent is a ready-to-use Western blot related reagent solution used for efficient extraction
TECHNICAL BULLETIN. Catalog Number RAB0447 Storage Temperature 20 C
 Phospho-Stat3 (ptyr 705 ) and pan-stat3 ELISA Kit for detection of human, mouse, or rat phospho-stat3 (ptyr 705 ) and pan-stat3 in cell and tissue lysates Catalog Number RAB0447 Storage Temperature 20
Phospho-Stat3 (ptyr 705 ) and pan-stat3 ELISA Kit for detection of human, mouse, or rat phospho-stat3 (ptyr 705 ) and pan-stat3 in cell and tissue lysates Catalog Number RAB0447 Storage Temperature 20
ccess safe drinking wa r is everyone s right Protozoans that cause diarrheal disease
 ccess safe drinking wa r is everyone s right Protozoa: Protozoans that cause diarrheal disease 1. Giardia lamblia 2. Entameba histolytica 3. Cryptosporidium parvum 4. Cyclospora cayetanensis 1 Giardia
ccess safe drinking wa r is everyone s right Protozoa: Protozoans that cause diarrheal disease 1. Giardia lamblia 2. Entameba histolytica 3. Cryptosporidium parvum 4. Cyclospora cayetanensis 1 Giardia
Jyotika Sharma, Feng Dong, Mustak Pirbhai, and Guangming Zhong*
 INFECTION AND IMMUNITY, July 2005, p. 4414 4419 Vol. 73, No. 7 0019-9567/05/$08.00 0 doi:10.1128/iai.73.7.4414 4419.2005 Copyright 2005, American Society for Microbiology. All Rights Reserved. Inhibition
INFECTION AND IMMUNITY, July 2005, p. 4414 4419 Vol. 73, No. 7 0019-9567/05/$08.00 0 doi:10.1128/iai.73.7.4414 4419.2005 Copyright 2005, American Society for Microbiology. All Rights Reserved. Inhibition
Human TSH ELISA Kit. User Manual
 Human TSH ELISA Kit User Manual Catalog number: GTX15585 GeneTex Table of Contents A. Product Description... 2 B. Kit Components... 3 C. Additional Required Materials (not included)... 3 D. Reagent Preparation...
Human TSH ELISA Kit User Manual Catalog number: GTX15585 GeneTex Table of Contents A. Product Description... 2 B. Kit Components... 3 C. Additional Required Materials (not included)... 3 D. Reagent Preparation...
Anti-Lamin B1/LMNB1 Picoband Antibody
 Anti-Lamin B1/LMNB1 Picoband Antibody Catalog Number:PB9611 About LMNB1 Lamin-B1 is a protein that in humans is encoded by the LMNB1 gene. The nuclear lamina consists of a two-dimensional matrix of proteins
Anti-Lamin B1/LMNB1 Picoband Antibody Catalog Number:PB9611 About LMNB1 Lamin-B1 is a protein that in humans is encoded by the LMNB1 gene. The nuclear lamina consists of a two-dimensional matrix of proteins
Influenza B Hemagglutinin / HA ELISA Pair Set
 Influenza B Hemagglutinin / HA ELISA Pair Set Catalog Number : SEK11053 To achieve the best assay results, this manual must be read carefully before using this product and the assay is run as summarized
Influenza B Hemagglutinin / HA ELISA Pair Set Catalog Number : SEK11053 To achieve the best assay results, this manual must be read carefully before using this product and the assay is run as summarized
Cryptosporidium parvum. Cyclospora cayetanensis. Isospora belli. Entamoeba histolytica.
 Cryptosporidium parvum Cyclospora cayetanensis Isospora belli Entamoeba histolytica Giardia lamblia cellular immunity humoral immunity microsporidia Trichomonas vaginalis 12 64 http://www.aids-care.org.tw
Cryptosporidium parvum Cyclospora cayetanensis Isospora belli Entamoeba histolytica Giardia lamblia cellular immunity humoral immunity microsporidia Trichomonas vaginalis 12 64 http://www.aids-care.org.tw
Characterization of Entamoeba histolytica Antigens in Circulating Immune Complexes in Sera of Patients with Amoebiasis
 J HEALTH POPUL NUTR 2002 Sep;20(3):215-222 2002 ICDDR,B: Centre for Health and Population Research ISSN Circulating 1606-0997 immune $ complexes 5.00+0.20 in amoebiasis 215 Characterization of Entamoeba
J HEALTH POPUL NUTR 2002 Sep;20(3):215-222 2002 ICDDR,B: Centre for Health and Population Research ISSN Circulating 1606-0997 immune $ complexes 5.00+0.20 in amoebiasis 215 Characterization of Entamoeba
Human Immunodeficiency Virus type 1 (HIV-1) gp120 / Glycoprotein 120 ELISA Pair Set
 Human Immunodeficiency Virus type 1 (HIV-1) gp120 / Glycoprotein 120 ELISA Pair Set Catalog Number : SEK11233 To achieve the best assay results, this manual must be read carefully before using this product
Human Immunodeficiency Virus type 1 (HIV-1) gp120 / Glycoprotein 120 ELISA Pair Set Catalog Number : SEK11233 To achieve the best assay results, this manual must be read carefully before using this product
SIV p27 Antigen ELISA Catalog Number:
 INTENDED USE The RETRO-TEK SIV p27 Antigen ELISA is for research use only and is not intended for in vitro diagnostic use. The RETRO-TEK SIV p27 Antigen ELISA is an enzyme linked immunoassay used to detect
INTENDED USE The RETRO-TEK SIV p27 Antigen ELISA is for research use only and is not intended for in vitro diagnostic use. The RETRO-TEK SIV p27 Antigen ELISA is an enzyme linked immunoassay used to detect
Please use only the valid version of the package insert provided with the kit.
 Please use only the valid version of the package insert provided with the kit. Intended Use For the qualitative screening of serum IgG antibodies to Trichinella using an Enzyme Linked Immunoabsorbant Assay
Please use only the valid version of the package insert provided with the kit. Intended Use For the qualitative screening of serum IgG antibodies to Trichinella using an Enzyme Linked Immunoabsorbant Assay
Identification of Microbes Lecture: 12
 Diagnostic Microbiology Identification of Microbes Lecture: 12 Electron Microscopy 106 virus particles per ml required for visualization, 50,000-60,000 magnification normally used. Viruses may be detected
Diagnostic Microbiology Identification of Microbes Lecture: 12 Electron Microscopy 106 virus particles per ml required for visualization, 50,000-60,000 magnification normally used. Viruses may be detected
Mammalian Membrane Protein Extraction Kit
 Mammalian Membrane Protein Extraction Kit Catalog number: AR0155 Boster s Mammalian Membrane Protein Extraction Kit is a simple, rapid and reproducible method to prepare cellular protein fractions highly
Mammalian Membrane Protein Extraction Kit Catalog number: AR0155 Boster s Mammalian Membrane Protein Extraction Kit is a simple, rapid and reproducible method to prepare cellular protein fractions highly
EXOTESTTM. ELISA assay for exosome capture, quantification and characterization from cell culture supernatants and biological fluids
 DATA SHEET EXOTESTTM ELISA assay for exosome capture, quantification and characterization from cell culture supernatants and biological fluids INTRODUCTION Exosomes are small endosome-derived lipid nanoparticles
DATA SHEET EXOTESTTM ELISA assay for exosome capture, quantification and characterization from cell culture supernatants and biological fluids INTRODUCTION Exosomes are small endosome-derived lipid nanoparticles
Serodiagnosis of Canine Babesiosis
 Serodiagnosis of Canine Babesiosis Xuenan XUAN, DVM, PhD National Research Center for Protozoan Diseases Obihiro University of Agriculture and Veterinary Medicine Obihiro, Hokkaido 080-8555, Japan Tel.:+81-155-495648;
Serodiagnosis of Canine Babesiosis Xuenan XUAN, DVM, PhD National Research Center for Protozoan Diseases Obihiro University of Agriculture and Veterinary Medicine Obihiro, Hokkaido 080-8555, Japan Tel.:+81-155-495648;
Parasitology. Lab. Amoeba
 Parasitology. Lab. Kingdom : Protista Subkingdom : Protozoa Phylum : Sacromastigophora Subphylum : Sarcodina Superclass : Rhizopoda Class : Lobosea Order : Amoebida Amoeba Protozoa Amoebae geneus Entamoeba
Parasitology. Lab. Kingdom : Protista Subkingdom : Protozoa Phylum : Sacromastigophora Subphylum : Sarcodina Superclass : Rhizopoda Class : Lobosea Order : Amoebida Amoeba Protozoa Amoebae geneus Entamoeba
TECHNICAL BULLETIN. Phospho-Akt (pser 473 ) ELISA Kit for detection of human, mouse, or rat phospho-akt (pser 473 ) in cell and tissue lysates
 Phospho-Akt (pser 473 ) ELISA Kit for detection of human, mouse, or rat phospho-akt (pser 473 ) in cell and tissue lysates Catalog Number RAB0011 Storage Temperature 20 C TECHNICAL BULLETIN Product Description
Phospho-Akt (pser 473 ) ELISA Kit for detection of human, mouse, or rat phospho-akt (pser 473 ) in cell and tissue lysates Catalog Number RAB0011 Storage Temperature 20 C TECHNICAL BULLETIN Product Description
Mitochondrial Trifunctional Protein (TFP) Protein Quantity Microplate Assay Kit
 PROTOCOL Mitochondrial Trifunctional Protein (TFP) Protein Quantity Microplate Assay Kit DESCRIPTION Mitochondrial Trifunctional Protein (TFP) Protein Quantity Microplate Assay Kit Sufficient materials
PROTOCOL Mitochondrial Trifunctional Protein (TFP) Protein Quantity Microplate Assay Kit DESCRIPTION Mitochondrial Trifunctional Protein (TFP) Protein Quantity Microplate Assay Kit Sufficient materials
NF-κB p65 (Phospho-Thr254)
 Assay Biotechnology Company www.assaybiotech.com Tel: 1-877-883-7988 Fax: 1-877-610-9758 NF-κB p65 (Phospho-Thr254) Colorimetric Cell-Based ELISA Kit Catalog #: OKAG02015 Please read the provided manual
Assay Biotechnology Company www.assaybiotech.com Tel: 1-877-883-7988 Fax: 1-877-610-9758 NF-κB p65 (Phospho-Thr254) Colorimetric Cell-Based ELISA Kit Catalog #: OKAG02015 Please read the provided manual
STAT3 (py705)/ Pan STAT3 (Human/Mouse/Rat) ELISA Kit
 STAT3 (py705)/ Pan STAT3 (Human/Mouse/Rat) ELISA Kit Catalog Number KA2176 96 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Principle of the Assay...
STAT3 (py705)/ Pan STAT3 (Human/Mouse/Rat) ELISA Kit Catalog Number KA2176 96 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Principle of the Assay...
Influenza or flu is a
 Clinical and Research Area Infectious Diseases Influenza Virus Types A and B Influenza or flu is a respiratory illness that is caused by influenza viruses. Influenza viruses type A and type B cause seasonal
Clinical and Research Area Infectious Diseases Influenza Virus Types A and B Influenza or flu is a respiratory illness that is caused by influenza viruses. Influenza viruses type A and type B cause seasonal
Structural study of mosquito ovarian proteins participating in Transovarial transmission of dengue viruses
 ISSN: 2319-7706 Volume 3 Number 4 (2014) pp. 565-572 http://www.ijcmas.com Original Research Article Structural study of mosquito ovarian proteins participating in Transovarial transmission of dengue viruses
ISSN: 2319-7706 Volume 3 Number 4 (2014) pp. 565-572 http://www.ijcmas.com Original Research Article Structural study of mosquito ovarian proteins participating in Transovarial transmission of dengue viruses
Protocol for protein SDS PAGE and Transfer
 Protocol for protein SDS PAGE and Transfer According to Laemmli, (1970) Alaa El -Din Hamid Sayed, Alaa_h254@yahoo.com Serum Selection of a protein source cell cultures (bacteria, yeast, mammalian, etc.)
Protocol for protein SDS PAGE and Transfer According to Laemmli, (1970) Alaa El -Din Hamid Sayed, Alaa_h254@yahoo.com Serum Selection of a protein source cell cultures (bacteria, yeast, mammalian, etc.)
HSV-1 IgM ELISA. Catalog No (96 Tests) For Research Use Only. Not for use in Diagnostic Procedures.
 For Research Use Only. Not for use in Diagnostic Procedures. INTENDED USE The GenWay, Inc. HSV-1 IgM ELISA Kit is intended for the detection of IgM antibody to HSV-1 in human serum or plasma. SUMMARY AND
For Research Use Only. Not for use in Diagnostic Procedures. INTENDED USE The GenWay, Inc. HSV-1 IgM ELISA Kit is intended for the detection of IgM antibody to HSV-1 in human serum or plasma. SUMMARY AND
Influenza A IgG ELISA
 Influenza A IgG ELISA For the qualitative determination of IgG-class antibodies against Influenza A virus in human serum or plasma (citrate, heparin). For Research Use Only. Not For Use In Diagnostic Procedures.
Influenza A IgG ELISA For the qualitative determination of IgG-class antibodies against Influenza A virus in human serum or plasma (citrate, heparin). For Research Use Only. Not For Use In Diagnostic Procedures.
Influenza A H7N9 (A/Anhui/1/2013) Hemagglutinin / HA ELISA Pair Set
 Influenza A H7N9 (A/Anhui/1/2013) Hemagglutinin / HA ELISA Pair Set Catalog Number : SEK40103 To achieve the best assay results, this manual must be read carefully before using this product and the assay
Influenza A H7N9 (A/Anhui/1/2013) Hemagglutinin / HA ELISA Pair Set Catalog Number : SEK40103 To achieve the best assay results, this manual must be read carefully before using this product and the assay
Exosomes Immunocapture and Isolation Tools: Immunobeads
 Product Insert Exosomes Immunocapture and Isolation Tools: Immunobeads HBM-BOLF-##/##-# HBM-BOLC-##/##-# HBM-BTLF-##/##-# Overall Exosome capture from Human Biological fluids Overall Exosome capture from
Product Insert Exosomes Immunocapture and Isolation Tools: Immunobeads HBM-BOLF-##/##-# HBM-BOLC-##/##-# HBM-BTLF-##/##-# Overall Exosome capture from Human Biological fluids Overall Exosome capture from
FOCUS SubCell. For the Enrichment of Subcellular Fractions. (Cat. # ) think proteins! think G-Biosciences
 169PR 01 G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name FOCUS SubCell For the Enrichment of Subcellular Fractions (Cat. # 786 260) think
169PR 01 G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name FOCUS SubCell For the Enrichment of Subcellular Fractions (Cat. # 786 260) think
Exosome ELISA Complete Kits
 Exosome ELISA Complete Kits EXOEL-CD9A-1, EXOEL-CD63A-1, EXOEL-CD81A-1 User Manual See PAC for Storage Conditions for Individual Components Version 12 4/17/2017 A limited-use label license covers this
Exosome ELISA Complete Kits EXOEL-CD9A-1, EXOEL-CD63A-1, EXOEL-CD81A-1 User Manual See PAC for Storage Conditions for Individual Components Version 12 4/17/2017 A limited-use label license covers this
Supplementary material: Materials and suppliers
 Supplementary material: Materials and suppliers Electrophoresis consumables including tris-glycine, acrylamide, SDS buffer and Coomassie Brilliant Blue G-2 dye (CBB) were purchased from Ameresco (Solon,
Supplementary material: Materials and suppliers Electrophoresis consumables including tris-glycine, acrylamide, SDS buffer and Coomassie Brilliant Blue G-2 dye (CBB) were purchased from Ameresco (Solon,
Canine C-Reactive Protein ELISA
 Canine C-Reactive Protein ELISA Product Data Sheet Cat. No.: RH931CRP01DCR For Research Use Only Page 1 of 12 VERSION 51 020611 46 CONTENTS 1. INTENDED USE 3 2. PRINCIPLE OF ASSAY 3 3. REAGENTS AND MATERIALS
Canine C-Reactive Protein ELISA Product Data Sheet Cat. No.: RH931CRP01DCR For Research Use Only Page 1 of 12 VERSION 51 020611 46 CONTENTS 1. INTENDED USE 3 2. PRINCIPLE OF ASSAY 3 3. REAGENTS AND MATERIALS
Stitaya Sirisinha, Runglawan Chawengkirttikul.and. Department of Microbiology, Faculty of Science, Mahidol University, Bangkok 10400, Thailand.
 Stitaya Sirisinha, Runglawan Chawengkirttikul.and Rasana Sennswan Department of Microbiology, Faculty of Science, Mahidol University, Bangkok 10400, Thailand. Abstract. Monoclonal antibody-based enzyme-linked
Stitaya Sirisinha, Runglawan Chawengkirttikul.and Rasana Sennswan Department of Microbiology, Faculty of Science, Mahidol University, Bangkok 10400, Thailand. Abstract. Monoclonal antibody-based enzyme-linked
IVD. DRG Toxocara canis ELISA (EIA-3518) Revised 12 Feb rm (Vers. 6.1)
 Please use only the valid version of the package insert provided with the kit. Intended Use For the qualitative screening of serum IgG antibodies to Toxocara using an Enzyme-Linked Immunosorbent Assay
Please use only the valid version of the package insert provided with the kit. Intended Use For the qualitative screening of serum IgG antibodies to Toxocara using an Enzyme-Linked Immunosorbent Assay
POLYPEPTIDES ASSOCIATED WITH IN VITRO CYST FORMATION OF BLASTOCYSTIS HOMINIS
 IN VITRO CYST FORMATION OF B. HOMINIS POLYPEPTIDES ASSOCIATED WITH IN VITRO CYST FORMATION OF BLASTOCYSTIS HOMINIS I Init 1, JW Mak 2, S Top 3, Z Zulhainan 4, S Prummongkol 5, V Nissapatorn 1, WS Wan-Yusoff
IN VITRO CYST FORMATION OF B. HOMINIS POLYPEPTIDES ASSOCIATED WITH IN VITRO CYST FORMATION OF BLASTOCYSTIS HOMINIS I Init 1, JW Mak 2, S Top 3, Z Zulhainan 4, S Prummongkol 5, V Nissapatorn 1, WS Wan-Yusoff
Serological Techniques for Laboratory Diagnosis
 JOURNAL OF CLINICAL MICROBIOLOGY, Aug. 1979, p. 128-133 0095-1 137/79/08-0128/06$02.00/0 Vol. 10, No. 2 Entamoeba histolytica: Efficacy of Microscopic, Cultural, and Serological Techniques for Laboratory
JOURNAL OF CLINICAL MICROBIOLOGY, Aug. 1979, p. 128-133 0095-1 137/79/08-0128/06$02.00/0 Vol. 10, No. 2 Entamoeba histolytica: Efficacy of Microscopic, Cultural, and Serological Techniques for Laboratory
Product Manual. [pser 473/474 ]Akt1/2 ELISA kit TABLE OF CONTENTS
![Product Manual. [pser 473/474 ]Akt1/2 ELISA kit TABLE OF CONTENTS Product Manual. [pser 473/474 ]Akt1/2 ELISA kit TABLE OF CONTENTS](/thumbs/91/106555455.jpg) [pser 473/474 ]Akt1/2 ELISA kit Catalog # ADI-900-162 96 well enzyme-linked immunoassay kit For use with cell lysates All reagents, except standard, should be stored at 4 C. Store standard at -20 C. Check
[pser 473/474 ]Akt1/2 ELISA kit Catalog # ADI-900-162 96 well enzyme-linked immunoassay kit For use with cell lysates All reagents, except standard, should be stored at 4 C. Store standard at -20 C. Check
CHAPTER 4 RESULTS. showed that all three replicates had similar growth trends (Figure 4.1) (p<0.05; p=0.0000)
 CHAPTER 4 RESULTS 4.1 Growth Characterization of C. vulgaris 4.1.1 Optical Density Growth study of Chlorella vulgaris based on optical density at 620 nm (OD 620 ) showed that all three replicates had similar
CHAPTER 4 RESULTS 4.1 Growth Characterization of C. vulgaris 4.1.1 Optical Density Growth study of Chlorella vulgaris based on optical density at 620 nm (OD 620 ) showed that all three replicates had similar
Mouse C3 (Complement Factor 3) ELISA Kit
 Mouse C3 (Complement Factor 3) ELISA Kit Cat. No.:DEIA8289 Pkg.Size:96T Intended use The Mouse C3 (Complement Factor 3) ELISA Kit is a highly sensitive two-site enzyme linked immunoassay (ELISA) for measuring
Mouse C3 (Complement Factor 3) ELISA Kit Cat. No.:DEIA8289 Pkg.Size:96T Intended use The Mouse C3 (Complement Factor 3) ELISA Kit is a highly sensitive two-site enzyme linked immunoassay (ELISA) for measuring
Hepatitis A virus IgM ELISA Kit
 Hepatitis A virus IgM ELISA Kit Catalog Number KA0285 96 assays Version: 04 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Hepatitis A virus IgM ELISA Kit Catalog Number KA0285 96 assays Version: 04 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
H5N1 ( Avian Flu ) Hemagglutinin ELISA Pair Set
 H5N1 ( Avian Flu ) Hemagglutinin ELISA Pair Set Catalog Number : SEK002 To achieve the best assay results, this manual must be read carefully before using this product and the assay is run as summarized
H5N1 ( Avian Flu ) Hemagglutinin ELISA Pair Set Catalog Number : SEK002 To achieve the best assay results, this manual must be read carefully before using this product and the assay is run as summarized
Novel anti-citrullinated peptide autoantibodies identified by proteomics with. citrullinated proteins in patients with rheumatoid arthritis
 Original ArticleProteomic surveillance of ACPAs in RA 121 Original Article Novel anti-citrullinated peptide autoantibodies identified by proteomics with citrullinated proteins in patients with rheumatoid
Original ArticleProteomic surveillance of ACPAs in RA 121 Original Article Novel anti-citrullinated peptide autoantibodies identified by proteomics with citrullinated proteins in patients with rheumatoid
Protocol for Western Blo
 Protocol for Western Blo ng SDS-PAGE separa on 1. Make appropriate percentage of separa on gel according to the MW of target proteins. Related recommenda ons and rou ne recipes of separa on/stacking gels
Protocol for Western Blo ng SDS-PAGE separa on 1. Make appropriate percentage of separa on gel according to the MW of target proteins. Related recommenda ons and rou ne recipes of separa on/stacking gels
Entamoeba histolytica cyst as a causative pathogen of bloody diarrhea in children below 5 years
 EUROPEAN ACADEMIC RESEARCH Vol. III, Issue 6/ September 2015 ISSN 2286-4822 www.euacademic.org Impact Factor: 3.4546 (UIF) DRJI Value: 5.9 (B+) Entamoeba histolytica cyst as a causative pathogen of bloody
EUROPEAN ACADEMIC RESEARCH Vol. III, Issue 6/ September 2015 ISSN 2286-4822 www.euacademic.org Impact Factor: 3.4546 (UIF) DRJI Value: 5.9 (B+) Entamoeba histolytica cyst as a causative pathogen of bloody
CHAPTER 4: DISEASES SPREAD BY FOOD AND WATER
 CHAPTER 4: DISEASES SPREAD BY FOOD AND WATER Highlights The incidence of diseases spread by food and water was generally higher in Peel than Ontario with the exceptions of hepatitis A and verotoxinproducing
CHAPTER 4: DISEASES SPREAD BY FOOD AND WATER Highlights The incidence of diseases spread by food and water was generally higher in Peel than Ontario with the exceptions of hepatitis A and verotoxinproducing
Introduction. Methods RESEARCH FUND FOR THE CONTROL OF INFECTIOUS DISEASES. TCW Poon *, HLY Chan, HWC Leung, A Lo, RHY Lau, AY Hui, JJY Sung
 RESEARCH FUND FOR THE CONTROL OF INFECTIOUS DISEASES Liver specific glycoforms of serum proteins in chronic hepatitis B infection: identification by lectin affinity chromatography and quantitative proteomic
RESEARCH FUND FOR THE CONTROL OF INFECTIOUS DISEASES Liver specific glycoforms of serum proteins in chronic hepatitis B infection: identification by lectin affinity chromatography and quantitative proteomic
HIV-1 Virus-like Particle Budding Assay Nathan H Vande Burgt, Luis J Cocka * and Paul Bates
 HIV-1 Virus-like Particle Budding Assay Nathan H Vande Burgt, Luis J Cocka * and Paul Bates Department of Microbiology, Perelman School of Medicine at the University of Pennsylvania, Philadelphia, USA
HIV-1 Virus-like Particle Budding Assay Nathan H Vande Burgt, Luis J Cocka * and Paul Bates Department of Microbiology, Perelman School of Medicine at the University of Pennsylvania, Philadelphia, USA
Ameba has two stages of development: cyst and trophozoite
 Amebiasis A parasitic disease of worldwide public health importance Second to malaria in mortality due to protozoan parasites Invasive amebiasis results in up to 100,000 deaths / year Amebiasis is infection
Amebiasis A parasitic disease of worldwide public health importance Second to malaria in mortality due to protozoan parasites Invasive amebiasis results in up to 100,000 deaths / year Amebiasis is infection
Human Influenza A (Swine Flu) Rapid test
 Human Influenza A (Swine Flu) Rapid test Cat.No: DTSXY-Z9 Lot. No. (See product label) Size 20T Intended use The Influenza A (Swine Flu) test is a rapid chromatographic immunoassay for the qualitative
Human Influenza A (Swine Flu) Rapid test Cat.No: DTSXY-Z9 Lot. No. (See product label) Size 20T Intended use The Influenza A (Swine Flu) test is a rapid chromatographic immunoassay for the qualitative
Western Blot Analysis of Rat Pituitar Recognized by Human Antipituitary. y Antigens A. antibodies
 Endocrine Journal 1995, 42(1), 115-119 NOTE Western Blot Analysis of Rat Pituitar Recognized by Human Antipituitary y Antigens A ntibodies SHIGEKI YABE, MASAMI MURAKAMI*, KAYOKO MARUYAMA, HIDEKO MIWA,
Endocrine Journal 1995, 42(1), 115-119 NOTE Western Blot Analysis of Rat Pituitar Recognized by Human Antipituitary y Antigens A ntibodies SHIGEKI YABE, MASAMI MURAKAMI*, KAYOKO MARUYAMA, HIDEKO MIWA,
Mycoplasma pneumoniae IgG ELISA Kit
 Mycoplasma pneumoniae IgG ELISA Kit Catalog Number KA2260 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Mycoplasma pneumoniae IgG ELISA Kit Catalog Number KA2260 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
PRODUCT INFORMATION & MANUAL
 PRODUCT INFORMATION & MANUAL 0.4 micron for Overall Exosome Isolation (Cell Media) NBP2-49826 For research use only. Not for diagnostic or therapeutic procedures. www.novusbio.com - P: 303.730.1950 - P:
PRODUCT INFORMATION & MANUAL 0.4 micron for Overall Exosome Isolation (Cell Media) NBP2-49826 For research use only. Not for diagnostic or therapeutic procedures. www.novusbio.com - P: 303.730.1950 - P:
