Switch protocol: Insulin pen needles to cost effective brand
|
|
|
- Norman Ellis
- 6 years ago
- Views:
Transcription
1 Switch protocol: Insulin pen needles to cost effective brand Applies to HaRD CCG employed Pharmacists and Medicines Optimisation Technicians. These protocols are produced by the NY&AWC MM team hosted by HaRD CCG for use by their employed MM team members. They can be adopted for use by other healthcare staff working in GP practices across NY&AWC CCGs but HaRD CCG accepts no responsibility for the use and application of these protocols in these situations. External staff working to these protocols must agree with their own employer whether they are competent and able to work to these protocols Rationale Pen needles are sterile single use disposable needles for subcutaneous injection of insulin with pre-filled syringes or pen injection devices. Insulin pen needles come in a variety of lengths and widths to suit all body types. From 4 to 12mm, the needle length the patient requires is likely to be dictated by the size of the patient, the thickness of their subcutaneous fat layer and injection technique. Most patients are likely to benefit from the shorter 4 and 6mm needles with less risk of pain on injection or accidental intramuscular injection. There is no clinical reason to recommend a needle length of over 8mm, unless the patient in historically used to pinching the skin, and no reason for over 6mm in children and adolescents (Frid, et al., 2010) (Hicks, et al., 2011). The measurements that are part of the needle descriptions relate to how long the needle is. There is also a gauge size. This denotes the thickness, size, or capacity. The two factors of gauge and needle length will contribute to perceptions of pain when injecting. The longer the needle is, the harder it is to pressurize the insulin through, so it needs a wider hole down the middle, the lower the gauge number the wider the aperture and the thicker the needle is. The use of a shorter needle encourages best practice and helps to prevent intramuscular injections which can lead to hypoglycaemic shock. It is proposed that repeat prescriptions for pen needles can be switched to lower cost branded alternatives. Patients can be a prescribed a needle length similar to but not the same as their current length without clinical impact or any differences in administration (Frid, et al., 2010).This switch does not address changing those on needle lengths greater the 8mm to the short lengths and covers only a like for like switch. Patients with the longer needle lengths would potentially need to be retrained before such a switch. There is a large variation in both the utilisation of insulin pen needles and the uptake of newer, cost-effective alternative products across the CCGs within England. Cost-effectiveness, product range, quality and availability should be considered when making these changes. Appendix 1 shows a sample of common pen needles along with a switch illustration. (NHSBSA, 2016). The CCG selected brand to switch to can be found on the CCG formulary. Insulin Pen Needles to low cost brand Page 1 of 7 NYAWC MMT
2 Method 1. Check the practice has agreed to the protocol and a signed copy is in place. 2. Check for any extra exclusions or amendments to the protocol made by the practice. 3. Rearrange practice formulary/add do not use codes where used to prevent future inadvertent prescribing. Check prescribing support software prompts are in place where relevant. 4. Notify local pharmacies/dispensary of work being undertaken and inform any relevant practice staff e.g. dispensary staff. 5. Run a computer search to identify patients who are currently receiving prescriptions for all sizes of needles both brand and generic. See Appendix 1 for a list of needle options. 6. Use the data collection form (Appendix 2) and the medical records to record the following: Patient identifier Age Allergies Current prescription (generic or brand) Previous prescriptions for the CCG selected brand needles Concerns Exclusions Patient referred to prescriber Patient switched and letter sent 7. Identify patients to exclude, those considered suitable to switch now and those needing special consideration by prescriber. 8. If a patient is excluded, add a consultation note listing reasons why they are unsuitable. 9. For those patients who can be switched now or approved by prescriber; change repeat medication on computer to: CCG selected brand (nearest needle size) (Same instructions) 10. Ensure that the old pen needle prescription is archived. 11. Send a letter to the patient advising them of the change. Liaise with practice staff to organise mail merge of letters and posting. At the end of the session, for all those changes that have been completed, a letter must be ready to send to the patient for information. 12. Add READ code 8BIr or XaJKo (on Systm One) drug changed to cost effective alternative for all patients switched. 13. Problem link drug to disease (where possible). 14. Inform relevant practice staff. Insulin Pen Needles to low cost brand Page 2 of 7 NYAWC MMT
3 15. Record the numbers/patients changed using an activity log. 16. Continue to monitor the long term outcomes of the switch e.g. cost savings via PPD data, complaints, problems encountered etc. Exclusions - do not switch 1. Those patients receiving snap on pen needles 2. Patients already prescribed safety needles see also comment below in flag up for special consideration by prescriber section. 3. Those previously recorded as having received the CCG selected brand needles with a valid reason recorded for them to have been stopped. To flag up for special consideration by prescriber approval required via electronic task 1. Any patient that you are concerned may be unsuitable for switching, or other prescribing issue that you are concerned about. 2. Children under 16 parents or carers should be notified and an appointment may be necessary. 3. Those on a long needle length >8mm if agreed with the practice. 4. Patients prescribed safety needles PST member to check whether these are actually indicated. Appropriate circumstance might include: patients whose insulin is administered by a non-professional healthcare worker, e.g. carer, AND who is known to have a blood born virus e.g. HIV, hepatitis, OR who is known to be aggressive. 5. Those previously recorded as having received the CCG selected brand needles without a valid reason recorded for them to have been stopped. Points to discuss with practice 1. Who is the contact in the practice for the project? 2. How will under 16s be approached and managed? 3. Should those on a long needle length >8mm be brought in for a review appointment with a view to change to a short length and check of injection technique? If so exclude from the switch and flag to prescriber only. 4. Those patients using snap on needles can they be invited for review to switch to screw on? If so flag them to the prescriber. Switch only after recorded prescriber consent. 5. Seek agreement from practice as to whether palliative care patients can be switched. Patients considered to be in the last few weeks of life would not normally be recommended for the switch. 6. Agree content of patient letter a possible form of words is attached below 7. Agree the number of repeats to issue for patients who are switched. 8. Any practice additions, deletions or amendments to the protocol. Insulin Pen Needles to low cost brand Page 3 of 7 NYAWC MMT
4 References Frid, A., Hirsch, L., Gaspar, R. & Strauss, K., New injection recommendations for patients with diabetes. Diabetes & Metabolism 36 Suppl 2(Suppl 2):S3-18, 36(Supplement 2), pp. s3-18. Hicks, D., Kirkland, F., Pledger, J. & Down, S., The First UK Injection Technique Recommendatins 2nd Edition. [Online] Available at: [Accessed ]. NHSBSA, DM+D. [Online] Available at: [Accessed ]. Agreement to protocol Please detail any amendments to the protocol here/or attach a copy of agreed changes: Signature of practice prescribing lead/ manager Practice name Date Signed on behalf of NYAWC MMT The practice representative signing this protocol agrees to take responsibility for the notification of all relevant practice staff. Document version control Version State changes New version Actioned by Date Omnican Pen needles 0.02 Rewritten as interchangeable protocol. Statements checks, format update. Allergies and safety needle statements added. Clarity and safety revisions 1 00 SK March 2017 Insulin Pen Needles to low cost brand Page 4 of 7 NYAWC MMT
5 Possible letter Dear ~[Title/Initial/Surname] Your repeat prescription for insulin needles The practice has been reviewing its prescribing of insulin needles. This is to ensure our patients receive the best treatment offering the best value to the NHS. As a result we have changed your future prescriptions from (insert previous brand) to [CCG selected brand] (insert needle size). You should notice no effect on your treatment and you do not need to alter your injection technique. Your prescription will be changed automatically so please order your next prescription in the usual way. It would help the NHS save money if you would use up any remaining needle type first. If you have any queries regarding this letter or these new needles please contact [insert name of contact, practice] and or [insert of local CCG patient relations team] All medicines should be safely stored out of the reach of children. Yours sincerely ~ [Usual prescriber/registered prescriber/gp Prescribing Lead/Other] Agreement to letter Please detail any amendments to the letter here/or attach a copy of agreed changes: Signature of practice prescribing lead/ manager Practice name Date Signed on behalf of NYAWC MMT Insulin Pen Needles to low cost brand Page 5 of 7 NYAWC MMT
6 Appendix 1 Switch Illustration (NB. CCG choice can be found on individual CCG s formulary) From BD AutoShield Duo Needles Pen Inj Screw On 5mm/30 BD Micro-Fine Ultra Needles Pen Inj Screw On 4mm BD Microfine+ Needles Pen Inj Screw On 5mm BD Microfine+ Needles Pen Inj Screw On 8mm CarePoint Needles Pen Inj Screw On 12mm CarePoint Needles Pen Inj Screw On 4mm CarePoint Point Needles Pen Inj Screw On 5mm CarePoint Point Needles Pen Inj Screw On 6mm CarePoint Point Needles Pen Inj Screw On 8mm IME-FINE Needles Pen Inj Screw On 6mm/31 IME-FINE Needles Pen Inj Screw On 8mm/31 Nanopass Needles Pen Inj Screw On 6mm/32.5 Mylife Clickfine AutoProtect Pen Inj Screw On 8mm/29 Mylife Clickfine AutoProtect Pen Inj Screw On 5mm/31 Mylife Clickfine Needles Pen Inj Snap On 10mm Mylife Clickfine Needles Pen Inj Snap On 12mm Mylife Clickfine Needles Pen Inj Snap On 4mm Mylife Clickfine Needles Pen Inj Snap On 6mm Mylife Clickfine Needles Pen Inj Snap On 8mm Nanopass Needles Pen Inj Screw On 4mm/32.5 Nanopass Needles Pen Inj Screw On 8mm/32.5 NeedleBay Fine Pen Needles Pen Inj Screw On 4mm/32 NeedleBay Fine Pen Needles Pen Inj Screw On 5mm/31 NeedleBay Fine Pen Needles Pen Inj Screw On 8mm/31 NovoFine Autocover Pen Inj Screw On 8mm NovoFine Needles Pen Inj Screw On 12mm NovoFine Needles Pen Inj Screw On 6mm NovoFine Needles Pen Inj Screw On 8mm NovoTwist Needles Pen Inj Screw On 5mm/32 Unifine Pentips Needles Pen Inj Screw On 12mm/29 Unifine Pentips Needles Pen Inj Screw On 4mm/32 Unifine Pentips Needles Pen Inj Screw On 5mm/31 Unifine Pentips Needles Pen Inj Screw On 6mm/31 Unifine Pentips Needles Pen Inj Screw On 8mm/31 To GlucoRX FinePoint Needles Pen Inj Screw On 5mm/31 GlucoRX FinePoint Needles Pen Inj Screw On 4mm/31 GlucoRX FinePoint Needles Pen Inj Screw On 5mm/31 GlucoRX FinePoint Needles Pen Inj Screw On 4mm/31 GlucoRX FinePoint Needles Pen Inj Screw On 5mm/31 GlucoRX FinePoint Needles Pen Inj Screw On 6mm/31 GlucoRX FinePoint Needles Pen Inj Screw On 6mm/31 GlucoRX FinePoint Needles Pen Inj Screw On 6mm/31 GlucoRX FinePoint Needles Pen Inj Screw On 5mm/31 GlucoRX FinePoint Needles Pen Inj Screw On 10mm/29 GlucoRX FinePoint Needles Pen Inj Screw On 4mm/31 GlucoRX FinePoint Needles Pen Inj Screw On 6mm/31 GlucoRX FinePoint Needles Pen Inj Screw On 4mm/31 GlucoRX FinePoint Needles Pen Inj Screw On 4mm/31 GlucoRX FinePoint Needles Pen Inj Screw On 5mm/31 GlucoRX FinePoint Needles Pen Inj Screw On 6mm/31 GlucoRX FinePoint Needles Pen Inj Screw On 5mm/31 GlucoRX FinePoint Needles Pen Inj Screw On 4mm/31 GlucoRX FinePoint Needles Pen Inj Screw On 5mm/31 GlucoRX FinePoint Needles Pen Inj Screw On 6mm/31 Insulin Pen Needles to low cost brand Page 6 of 7 NYAWC MMT
7 Appendix 2 Insulin Pen Needles to Cost Effective Brand - Data Collection Form Patient identifier: Age Allergies e.g.: Latex Current needle [insert name] size and quantity Previously had CCG selected brand Y/N. If Yes, record reason why stopped Any contraindications/cautions If Yes, list and flag to prescriber Patient has an exclusion to switch to CCG selected brand? Flag to prescriber? Y/N Record reason Patient switched? Y/N Letter sent? Y/N Insulin Pen Needles to low cost brand Page 7 of 7 NYAWC MMT amendments, including updated hyperlinks. Please use the online version to ensure use of the latest version.
Switch protocol: Insulin pen needles to cost effective brand
 Switch protocol: Insulin pen needles to cost effective brand Applies to HaRD CCG employed Pharmacists and Medicines Optimisation Technicians. These protocols are produced by the NY&AWC MM team hosted by
Switch protocol: Insulin pen needles to cost effective brand Applies to HaRD CCG employed Pharmacists and Medicines Optimisation Technicians. These protocols are produced by the NY&AWC MM team hosted by
Metformin MR to low cost branded generic (Sukkarto SR) Switch Protocol
 Metformin MR to low cost branded generic (Sukkarto SR) Switch Protocol Applies to HaRD CCG employed Pharmacists and Medicines Optimisation Technicians. These protocols are produced by the NY&AWC MM team
Metformin MR to low cost branded generic (Sukkarto SR) Switch Protocol Applies to HaRD CCG employed Pharmacists and Medicines Optimisation Technicians. These protocols are produced by the NY&AWC MM team
Fencino patch (5) 2. Check the practice has agreed to the protocol and a signed copy is in place.
 Switch protocol: Fentanyl/Durogesic patches to Fencino /Matrifen /Mezolar brand switch Applies to HaRD CCG employed Pharmacists and Medicines Optimisation Technicians. These protocols are produced by the
Switch protocol: Fentanyl/Durogesic patches to Fencino /Matrifen /Mezolar brand switch Applies to HaRD CCG employed Pharmacists and Medicines Optimisation Technicians. These protocols are produced by the
Asacol / Mesalazine to Octasa Switch protocol
 Asacol / Mesalazine to Octasa Switch protocol Applies to HaRD MMT Technicians, Pharmacists These protocols are produced by the NY&AWC MM team hosted by HaRD CCG for use by their employed MM team members.
Asacol / Mesalazine to Octasa Switch protocol Applies to HaRD MMT Technicians, Pharmacists These protocols are produced by the NY&AWC MM team hosted by HaRD CCG for use by their employed MM team members.
Seretide 250mcg/125mcg Evohaler switch to Fostair 100/6 MDI in COPD.
 Seretide 250mcg/125mcg Evohaler switch to Fostair 100/6 MDI in COPD. Applies to HaRD CCG employed Pharmacists and Medicines Optimisation Technicians These protocols are produced by the NY&AWC MM team hosted
Seretide 250mcg/125mcg Evohaler switch to Fostair 100/6 MDI in COPD. Applies to HaRD CCG employed Pharmacists and Medicines Optimisation Technicians These protocols are produced by the NY&AWC MM team hosted
Optimising Insulin Pen Needles
 Optimising Insulin Pen Needles Following a review of insulin pen needles NHS Telford and Wrekin CCG now recommends the use of GlucoRx FinePoint Pen Needles. GlucoRx FinePoint Pen Needles are a high quality
Optimising Insulin Pen Needles Following a review of insulin pen needles NHS Telford and Wrekin CCG now recommends the use of GlucoRx FinePoint Pen Needles. GlucoRx FinePoint Pen Needles are a high quality
Review of Oral Nutritional Supplement (ONS) prescribing
 Applies to Review of Oral Nutritional Supplement (ONS) prescribing HaRD CCG employed Pharmacists and Medicines Optimisation Technicians. These protocols are produced by the NY&AWC MM team hosted by HaRD
Applies to Review of Oral Nutritional Supplement (ONS) prescribing HaRD CCG employed Pharmacists and Medicines Optimisation Technicians. These protocols are produced by the NY&AWC MM team hosted by HaRD
Medicines Optimisation Team Standard Operating Procedure Insulin Quantities Audit
 Medicines Optimisation Team Standard Operating Procedure Insulin Quantities Audit Aim Review and rationalise the insulin quantities based on dosage instructions for diabetic patients in order to align
Medicines Optimisation Team Standard Operating Procedure Insulin Quantities Audit Aim Review and rationalise the insulin quantities based on dosage instructions for diabetic patients in order to align
Protocol to support reducing the use of and the effective use of laxatives. HaRD CCG employed Pharmacists and Medicines Optimisation Technicians.
 NY&AWC Medicines Management Team Protocol to support reducing the use of and the effective use of laxatives Applies to HaRD CCG employed Pharmacists and Medicines Optimisation Technicians. These protocols
NY&AWC Medicines Management Team Protocol to support reducing the use of and the effective use of laxatives Applies to HaRD CCG employed Pharmacists and Medicines Optimisation Technicians. These protocols
Type 2 Diabetes Recommended SMBG
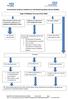 Herefordshire Diabetes Guideline for Self-Monitoring Blood Glucose (SMBG) Type 2 Diabetes Recommended SMBG Diet controlled/ metformin only/ combination of metformin and pioglitazone/dpp4/sglt2 or GLP1
Herefordshire Diabetes Guideline for Self-Monitoring Blood Glucose (SMBG) Type 2 Diabetes Recommended SMBG Diet controlled/ metformin only/ combination of metformin and pioglitazone/dpp4/sglt2 or GLP1
Guidance on Bulk Prescribing for Care Home Patients
 Guidance on Bulk Prescribing for Care Home Patients Introduction Many patients in care homes taking medicines when required (PRN) can inevitably present problems for the prescriber in determining the quantity
Guidance on Bulk Prescribing for Care Home Patients Introduction Many patients in care homes taking medicines when required (PRN) can inevitably present problems for the prescriber in determining the quantity
Medicines Optimisation Team Standard Operating procedure for Alogliptin Audit:
 Medicines Optimisation Team Standard Operating procedure for Alogliptin Audit: To review type 2 diabetic patients prescribed dipedtidylpeptidase-4 (DPP-4) inhibitors (linagliptin (Trajenta, saxagliptin
Medicines Optimisation Team Standard Operating procedure for Alogliptin Audit: To review type 2 diabetic patients prescribed dipedtidylpeptidase-4 (DPP-4) inhibitors (linagliptin (Trajenta, saxagliptin
Product Cost per Pack Cost per 28 days (assuming 2 sprays od) Flixonase years 50mcg in both nostrils od (increasing to max bd prn)
 Protocol for Flixonase to Avamys Nasal Spray Switch May 2010 1. Reason Avamys contains the corticosteroid fluticasone furoate (27.5mcg/spray) and Flixonase nasal spray contains fluticasone propionate (50mcg/spray).
Protocol for Flixonase to Avamys Nasal Spray Switch May 2010 1. Reason Avamys contains the corticosteroid fluticasone furoate (27.5mcg/spray) and Flixonase nasal spray contains fluticasone propionate (50mcg/spray).
Immediate Release Nifedipine Review July 2011
 Title of Project: NHS Dumfries & Galloway - Clinical Safety Review Immediate Release Nifedipine Review July 2011 1 Reason for the review 2 Aim Immediate release nifedipine capsules are associated with
Title of Project: NHS Dumfries & Galloway - Clinical Safety Review Immediate Release Nifedipine Review July 2011 1 Reason for the review 2 Aim Immediate release nifedipine capsules are associated with
Brook Green Centre for Learning. Policy and Guidance for Supporting Pupils with Medical Needs
 Brook Green Centre for Learning Policy and Guidance for Supporting Pupils with Medical Needs This document was written in line with recommendations made in the DfE s information pack Supporting Pupils
Brook Green Centre for Learning Policy and Guidance for Supporting Pupils with Medical Needs This document was written in line with recommendations made in the DfE s information pack Supporting Pupils
GENERIC MEDICINES (Non-Innovator Brand) PRESCRIBING POLICY
 GENERIC MEDICINES (Non-Innovator Brand) PRESCRIBING POLICY First issued by/date Issue Version Purpose of Issue/Description of Change Planned Review Date August 2018 1.0 Promoting generic prescribing whenever
GENERIC MEDICINES (Non-Innovator Brand) PRESCRIBING POLICY First issued by/date Issue Version Purpose of Issue/Description of Change Planned Review Date August 2018 1.0 Promoting generic prescribing whenever
If you inject diabetes medication, you should know how important correct injection technique is for effectively managing your diabetes.
 Insulin injection technique best practices Expert recommendations on injecting insulin with pen and insulin syringe needles If you inject diabetes medication, you should know how important correct injection
Insulin injection technique best practices Expert recommendations on injecting insulin with pen and insulin syringe needles If you inject diabetes medication, you should know how important correct injection
Title of Project: NHS Dumfries & Galloway Respiratory Bundle Asthma: Bronchodilator Overuse Review April 2015
 Title of Project: NHS Dumfries & Galloway Respiratory Bundle Asthma: Bronchodilator Overuse Review April 2015 1 Reason for the review Respiratory prescribing is long term and can be costly. Appropriate
Title of Project: NHS Dumfries & Galloway Respiratory Bundle Asthma: Bronchodilator Overuse Review April 2015 1 Reason for the review Respiratory prescribing is long term and can be costly. Appropriate
Guidance on the Self-Monitoring of Blood Glucose in Adults with Diabetes
 Introduction Guidance on the Self-Monitoring of Blood Glucose in Adults with Diabetes This guideline is designed to offer guidance for primary and secondary care on the use of selfmonitoring of blood glucose
Introduction Guidance on the Self-Monitoring of Blood Glucose in Adults with Diabetes This guideline is designed to offer guidance for primary and secondary care on the use of selfmonitoring of blood glucose
Management of Migraine
 Title of Project: NHS Dumfries & Galloway June 2013 Management of Migraine 1 Reason for the review SIGN 107 Diagnosis and management of headache in adults, advises that patients with migraine and those
Title of Project: NHS Dumfries & Galloway June 2013 Management of Migraine 1 Reason for the review SIGN 107 Diagnosis and management of headache in adults, advises that patients with migraine and those
Guidance on the Self-Monitoring of Blood Glucose in Adults with Diabetes
 Introduction Guidance on the Self-Monitoring of Blood Glucose in Adults with Diabetes This guideline is designed to offer guidance for primary and secondary care on the use of selfmonitoring of blood glucose
Introduction Guidance on the Self-Monitoring of Blood Glucose in Adults with Diabetes This guideline is designed to offer guidance for primary and secondary care on the use of selfmonitoring of blood glucose
MedsCheck Reviews. Ontario
 MedsCheck Reviews Ontario Contents Configuration... 1 Configuring Electronic Signatures... 1 Configuring Electronic MedsCheck Reviews... 2 Creating an ODB MedsCheck Consent Record... 3 Electronic MedsCheck
MedsCheck Reviews Ontario Contents Configuration... 1 Configuring Electronic Signatures... 1 Configuring Electronic MedsCheck Reviews... 2 Creating an ODB MedsCheck Consent Record... 3 Electronic MedsCheck
Smoking cessation interventions and services
 National Institute for Health and Care Excellence Guideline version (Final) Smoking cessation interventions and services [E] Evidence reviews for advice NICE guideline NG92 Evidence reviews FINAL These
National Institute for Health and Care Excellence Guideline version (Final) Smoking cessation interventions and services [E] Evidence reviews for advice NICE guideline NG92 Evidence reviews FINAL These
The prescribing newsletter for GPs, nurses and pharmacists NHS Northamptonshire Failure to respond to first choice antibiotics
 Tablet Press EXTRA The prescribing newsletter for GPs, nurses and pharmacists NHS Northamptonshire Failure to respond to first choice antibiotics March 2017 Use of Buccolam (buccal midazolam) for breakthrough
Tablet Press EXTRA The prescribing newsletter for GPs, nurses and pharmacists NHS Northamptonshire Failure to respond to first choice antibiotics March 2017 Use of Buccolam (buccal midazolam) for breakthrough
A new model for prescribing varenicline
 Pharmacist Independent Prescribers in partnership with A new model for prescribing varenicline Dear Stop Smoking Advisor You will be aware of the stop smoking drug varenicline that goes under the brand
Pharmacist Independent Prescribers in partnership with A new model for prescribing varenicline Dear Stop Smoking Advisor You will be aware of the stop smoking drug varenicline that goes under the brand
Document Details. Patient Group Direction
 Document Details Title Patient Group Direction (PGD) Hepatitis B Vaccine (Engerix ) Trust Ref No 1505-41182 Local Ref (optional) Main points the Immunisation against Hepatitis B document covers Who is
Document Details Title Patient Group Direction (PGD) Hepatitis B Vaccine (Engerix ) Trust Ref No 1505-41182 Local Ref (optional) Main points the Immunisation against Hepatitis B document covers Who is
Blood glucose monitoring and compatibility equipment checker
 Introduction This resource has been created to assist pharmacists, and other appropriately trained members of the pharmacy team, to ensure compatibility of insulin products/devices and blood glucose monitoring
Introduction This resource has been created to assist pharmacists, and other appropriately trained members of the pharmacy team, to ensure compatibility of insulin products/devices and blood glucose monitoring
E096. Essential Shared Care Agreement: Melatonin (Circadin ) in Children, adolescents and adults only with Learning Disability
 E096 Essential Shared Care Agreement: Melatonin (Circadin ) in Children, adolescents and adults only with Learning Disability Please complete the following details: Patient s name, address, date of birth,
E096 Essential Shared Care Agreement: Melatonin (Circadin ) in Children, adolescents and adults only with Learning Disability Please complete the following details: Patient s name, address, date of birth,
Ref. Version Supersedes Approved for Date approved Document Owner N/A MMT 17/01/2017 Lois Taylor. Switching to Alogliptin (Vipidia )
 Ref. Version Supersedes Approved for Date approved Document number use by Owner 001 1.0 N/A MMT 17/01/2017 Lois Taylor Aim: Switching to Alogliptin (Vipidia ) To reduce our spend on gliptins (DDP-4 drugs)
Ref. Version Supersedes Approved for Date approved Document number use by Owner 001 1.0 N/A MMT 17/01/2017 Lois Taylor Aim: Switching to Alogliptin (Vipidia ) To reduce our spend on gliptins (DDP-4 drugs)
venue Infant School MEDICINES PROTOCOL DOB: Insert photograph of child: This means..must avoid ALL substances which contain or may contain
 The venue Infant School MEDICINES PROTOCOL Name of Child: Address: Age of Child: DOB: Insert photograph of child: Parent/Carers name and contact numbers: Additional contacts: GP contact details: ALLERGY
The venue Infant School MEDICINES PROTOCOL Name of Child: Address: Age of Child: DOB: Insert photograph of child: Parent/Carers name and contact numbers: Additional contacts: GP contact details: ALLERGY
Unit 204 Assist with the assembly of prescribed items
 Element 1 Assemble prescribed 171 172 Element 1 Assemble prescribed Background Assembling prescribed is a complex process. The two main components of this process are labelling and dispensing. It is essential
Element 1 Assemble prescribed 171 172 Element 1 Assemble prescribed Background Assembling prescribed is a complex process. The two main components of this process are labelling and dispensing. It is essential
Document Details Patient Group Direction Hepatitis A vaccine (Havrix Monodose ) Trust Ref No Local Ref (optional) Main points the
 Document Details Title Patient Group Direction Hepatitis A vaccine (Havrix Monodose ) Trust Ref No 1506-41174 Local Ref (optional) Main points the Active immunisation against infection caused by Hepatitis
Document Details Title Patient Group Direction Hepatitis A vaccine (Havrix Monodose ) Trust Ref No 1506-41174 Local Ref (optional) Main points the Active immunisation against infection caused by Hepatitis
Paliperidone Palmitate 3-monthly Long-Acting Injection (Trevicta ) Guidelines for Prescribing and Administration (Version 1 October 2016)
 Paliperidone Palmitate 3-monthly Long-Acting Injection (Trevicta ) Guidelines for Prescribing and Administration (Version 1 October 2016) 1. Key Points 1.1 Paliperidone palmitate 3-monthly long-acting
Paliperidone Palmitate 3-monthly Long-Acting Injection (Trevicta ) Guidelines for Prescribing and Administration (Version 1 October 2016) 1. Key Points 1.1 Paliperidone palmitate 3-monthly long-acting
Policy and Procedure for the Development, Approval and Implementation of Patient Group Directions in NHS Haringey Clinical Commissioning Group
 BEFORE USING THIS POLICY ALWAYS ENSURE YOU ARE USING THE MOST UP TO DATE VERSION Policy and Procedure for the Development, Approval and Implementation of Patient Group Directions in NHS Haringey Clinical
BEFORE USING THIS POLICY ALWAYS ENSURE YOU ARE USING THE MOST UP TO DATE VERSION Policy and Procedure for the Development, Approval and Implementation of Patient Group Directions in NHS Haringey Clinical
BEDFORDSHIRE AND LUTON JOINT PRESCRIBING COMMITTEE (JPC)
 BEDFORDSHIRE AND LUTON JOINT PRESCRIBING COMMITTEE (JPC) Sept 2015 Review: Sept 2018 Bulletin 227: EXOGEN ultrasound bone healing system used for the management of long bone fractures JPC Recommendations:
BEDFORDSHIRE AND LUTON JOINT PRESCRIBING COMMITTEE (JPC) Sept 2015 Review: Sept 2018 Bulletin 227: EXOGEN ultrasound bone healing system used for the management of long bone fractures JPC Recommendations:
HCV Action and Bristol & Severn ODN workshop, 14 th September 2017: Summary report
 HCV Action and Bristol & Severn ODN workshop, 14 th September 2017: Summary report About HCV Action HCV Action is a network, co-ordinated by The Hepatitis C Trust, that brings together health professionals
HCV Action and Bristol & Severn ODN workshop, 14 th September 2017: Summary report About HCV Action HCV Action is a network, co-ordinated by The Hepatitis C Trust, that brings together health professionals
Volume 11; Number 5 February 2017 REVIEW OF GONADOTROPIN RELEASING HORMONE AGONISTS FOR PROSTATE CANCER (REVISED EDITION)
 Optum in association with Lincolnshire Clinical Commissioning Groups, Lincolnshire Community Health Services, United Lincolnshire Hospitals Trust and Lincolnshire Partnership Foundation Trust Volume 11;
Optum in association with Lincolnshire Clinical Commissioning Groups, Lincolnshire Community Health Services, United Lincolnshire Hospitals Trust and Lincolnshire Partnership Foundation Trust Volume 11;
Essential Shared Care Agreement (South Staffordshire): Aciclovir Administration in the Community
 E088 Essential Shared Care Agreement (South Staffordshire): Aciclovir Administration in the Community Patient s name: DOB NHS Number Patient s address: Consultant Note: Shared care agreement sets out a
E088 Essential Shared Care Agreement (South Staffordshire): Aciclovir Administration in the Community Patient s name: DOB NHS Number Patient s address: Consultant Note: Shared care agreement sets out a
Gentle and safe injections. Tips and tricks for injecting insulin.
 Gentle and safe injections. Tips and tricks for injecting insulin. Freedom. Confidence. With mylife. Gentle and safe injections The correct injection technique The insulin is injected into the subcutaneous
Gentle and safe injections. Tips and tricks for injecting insulin. Freedom. Confidence. With mylife. Gentle and safe injections The correct injection technique The insulin is injected into the subcutaneous
Shared Care Agreement for Donepezil
 ESCA: for the treatment of Alzheimer s disease SECONDARY CARE SECTION TO BE COMPLETED BY INITIATING DOCTOR Patient s Name: Date of Birth: NHS Number: ESCA Date: One copy of information leaflet given to
ESCA: for the treatment of Alzheimer s disease SECONDARY CARE SECTION TO BE COMPLETED BY INITIATING DOCTOR Patient s Name: Date of Birth: NHS Number: ESCA Date: One copy of information leaflet given to
Safety Regulations and Procedures Occupational Health Bloodborne Pathogens Exposure Control Plan S80.10, updated, May Contains information for:
 APPENDIX A Safety Regulations and Procedures Occupational Health Bloodborne Pathogens Exposure Control Plan S80.10, updated, May 2018 BLOODBORNE PATHOGEN EXPOSURE INCIDENT PACKET Contains information for:
APPENDIX A Safety Regulations and Procedures Occupational Health Bloodborne Pathogens Exposure Control Plan S80.10, updated, May 2018 BLOODBORNE PATHOGEN EXPOSURE INCIDENT PACKET Contains information for:
SHARED CARE PROTOCOL FOR THE PRESCRIBING AND MONITORING OF MEDICINES FOR ATTENTION-DEFICIT HYPERACTIVITY DISORDER (ADHD)
 SHARED CARE PROTOCOL FOR THE PRESCRIBING AND MONITORING OF MEDICINES FOR ATTENTION-DEFICIT HYPERACTIVITY DISORDER (ADHD) 1. Introduction This protocol describes how patients prescribed medicines for ADHD
SHARED CARE PROTOCOL FOR THE PRESCRIBING AND MONITORING OF MEDICINES FOR ATTENTION-DEFICIT HYPERACTIVITY DISORDER (ADHD) 1. Introduction This protocol describes how patients prescribed medicines for ADHD
NHS Dumfries & Galloway Ferrous Salt Review Protocol November 09
 Title of Project: NHS Dumfries & Galloway Ferrous Salt Review Protocol November 09 1 Reason for the review Choice of iron preparation is based on cost and incidence of side effects (BNF). There is little
Title of Project: NHS Dumfries & Galloway Ferrous Salt Review Protocol November 09 1 Reason for the review Choice of iron preparation is based on cost and incidence of side effects (BNF). There is little
ProScript User Guide. Pharmacy Access Medicines Manager
 User Guide Pharmacy Access Medicines Manager Version 3.0.0 Release Date 01/03/2014 Last Reviewed 11/04/2014 Author Rx Systems Service Desk (T): 01923 474 600 Service Desk (E): servicedesk@rxsystems.co.uk
User Guide Pharmacy Access Medicines Manager Version 3.0.0 Release Date 01/03/2014 Last Reviewed 11/04/2014 Author Rx Systems Service Desk (T): 01923 474 600 Service Desk (E): servicedesk@rxsystems.co.uk
Approval of Multiple Diabetes Management Products and Mesalazine proposals
 8 June Approval of Multiple Diabetes Management Products and Mesalazine proposals PHARMAC is pleased to announce the approval of five agreements in relation to funding of blood glucose, blood glucose diagnostic
8 June Approval of Multiple Diabetes Management Products and Mesalazine proposals PHARMAC is pleased to announce the approval of five agreements in relation to funding of blood glucose, blood glucose diagnostic
Emergency Inhaler Policy. March 2016
 Emergency Inhaler Policy March 2016 This Policy was adopted by the Governors of Presteigne Primary School in March 2016 Updated September 2017 Signed:..(Chair of Governors) Introduction Following agreement
Emergency Inhaler Policy March 2016 This Policy was adopted by the Governors of Presteigne Primary School in March 2016 Updated September 2017 Signed:..(Chair of Governors) Introduction Following agreement
STANDARD OPERATING PROCEDURE
 STANDARD OPERATING PROCEDURE Title Reference Number Site Initiation SOP-RES-010 Version Number 2 Issue Date 15 Apr 2016 Effective Date 26 May 2016 Review Date 26 May 2018 Author(s) Reviewer(s) Rachelle
STANDARD OPERATING PROCEDURE Title Reference Number Site Initiation SOP-RES-010 Version Number 2 Issue Date 15 Apr 2016 Effective Date 26 May 2016 Review Date 26 May 2018 Author(s) Reviewer(s) Rachelle
Flu season. Making the most of online appointments. August 2018
 Flu season Making the most of online appointments August 2018 Dear colleagues, Over the last four years GP Online Services have been implemented by practices across England. Over 14 million patients -
Flu season Making the most of online appointments August 2018 Dear colleagues, Over the last four years GP Online Services have been implemented by practices across England. Over 14 million patients -
Frequently Used PINs for Billing Purposes November 2018
 Diabetic Test Strips PIN Description 97799824 ACCU-CHEK ADVANTAGE STRIPS (100) 97799823 ACCU-CHEK ADVANTAGE STRIPS (50) 97799814 ACCU-CHEK AVIVA TEST STRIPS (100) 97799815 ACCU-CHEK AVIVA TEST STRIPS (50)
Diabetic Test Strips PIN Description 97799824 ACCU-CHEK ADVANTAGE STRIPS (100) 97799823 ACCU-CHEK ADVANTAGE STRIPS (50) 97799814 ACCU-CHEK AVIVA TEST STRIPS (100) 97799815 ACCU-CHEK AVIVA TEST STRIPS (50)
Emis Web Technical Guidance
 Emis Web Technical Guidance Updated February 2018 1 Dementia 18-19 Technical Solution Emis Web Implementation Guide Summary A resource to provide system support to clinicians and CCGs to: Data cleansing
Emis Web Technical Guidance Updated February 2018 1 Dementia 18-19 Technical Solution Emis Web Implementation Guide Summary A resource to provide system support to clinicians and CCGs to: Data cleansing
This document expires on Patient Group Direction No. VI 7
 Patient Group Direction (PGD) for The Administration of Combined low-dose Diphtheria, Tetanus, and Inactivated Polio Vaccine (Revaxis ) to individuals aged over 10 years, by Registered Nurses employed
Patient Group Direction (PGD) for The Administration of Combined low-dose Diphtheria, Tetanus, and Inactivated Polio Vaccine (Revaxis ) to individuals aged over 10 years, by Registered Nurses employed
Newfoundland and Labrador Pharmacy Board Standards of Practice
 Newfoundland and Labrador Pharmacy Board Standards of Practice Standards for the Safe and Effective Administration of Drug Therapy by Inhalation or Injection June 2015 Table of Contents 1) Introduction...
Newfoundland and Labrador Pharmacy Board Standards of Practice Standards for the Safe and Effective Administration of Drug Therapy by Inhalation or Injection June 2015 Table of Contents 1) Introduction...
Guide to Interchangeable Medicines
 Guide to Interchangeable Medicines AUT-G0115-6 01 JUNE 2018 This guide does not purport to be an interpretation of law and/or regulations and is for guidance purposes only. CONTENTS 1 BACKGROUND 3 1.1
Guide to Interchangeable Medicines AUT-G0115-6 01 JUNE 2018 This guide does not purport to be an interpretation of law and/or regulations and is for guidance purposes only. CONTENTS 1 BACKGROUND 3 1.1
Impact of Unifine Pentips Plus on pen needle changing behaviour amongst people with diabetes medicating with injectable formats.
 Impact of Unifine Pentips Plus on pen needle changing behaviour amongst people with diabetes medicating with injectable formats September 2014 September 2014 01 Impact of Unifine Pentips Plus on pen needle
Impact of Unifine Pentips Plus on pen needle changing behaviour amongst people with diabetes medicating with injectable formats September 2014 September 2014 01 Impact of Unifine Pentips Plus on pen needle
Costing report: Lipid modification Implementing the NICE guideline on lipid modification (CG181)
 Putting NICE guidance into practice Costing report: Lipid modification Implementing the NICE guideline on lipid modification (CG181) Published: July 2014 This costing report accompanies Lipid modification:
Putting NICE guidance into practice Costing report: Lipid modification Implementing the NICE guideline on lipid modification (CG181) Published: July 2014 This costing report accompanies Lipid modification:
Do not re use needles. If you do you might not get your dose (underdosing) or get too much (overdosing) as the needle could block.
 What are the ingredients in TOUJEO? Active ingredient: insulin glargine Inactive ingredients: zinc, m cresol, glycerol and water for injection Hydrochloric acid and sodium hydroxide may be added to adjust
What are the ingredients in TOUJEO? Active ingredient: insulin glargine Inactive ingredients: zinc, m cresol, glycerol and water for injection Hydrochloric acid and sodium hydroxide may be added to adjust
USE OF UNLICENSED MEDICINES AND OFF-LABEL MEDICINES WHERE A LICENSED MEDICINE IS AVAILABLE
 NHS Scotland Directors of Pharmacy and Scottish Association of Medical Directors USE OF UNLICENSED MEDICINES AND OFF-LABEL MEDICINES WHERE A LICENSED MEDICINE IS AVAILABLE CONSENSUS STATEMENT This consensus
NHS Scotland Directors of Pharmacy and Scottish Association of Medical Directors USE OF UNLICENSED MEDICINES AND OFF-LABEL MEDICINES WHERE A LICENSED MEDICINE IS AVAILABLE CONSENSUS STATEMENT This consensus
Safe and Gentle Injection of Insulin. Guide for correct injection technique. Englische Ausgabe
 Safe and Gentle Injection of Insulin Guide for correct injection technique Englische Ausgabe Insulin injection basics 1 Insulin has to be injected into the subcutaneous fat layer 3 Rotate the injection
Safe and Gentle Injection of Insulin Guide for correct injection technique Englische Ausgabe Insulin injection basics 1 Insulin has to be injected into the subcutaneous fat layer 3 Rotate the injection
ADVANCE PREPARATION OF INSULIN FOR PATIENTS TO ADMINISTER AT HOME
 STANDARD OPERATING PROCEDURE ADVANCE PREPARATION OF INSULIN FOR PATIENTS TO ADMINISTER AT HOME First Issued July 08 Issue Version Two Purpose of Issue/Description of Change Planned Review Date To promote
STANDARD OPERATING PROCEDURE ADVANCE PREPARATION OF INSULIN FOR PATIENTS TO ADMINISTER AT HOME First Issued July 08 Issue Version Two Purpose of Issue/Description of Change Planned Review Date To promote
MAGDALENE CATHOLIC HIGH SCHOOL Because I have seen the Lord. Anaphylaxis Policy
 MAGDALENE CATHOLIC HIGH SCHOOL Because I have seen the Lord Rationale Anaphylaxis Policy Magdalene Catholic High School endeavours to provide a safe and supportive school environment for all students.
MAGDALENE CATHOLIC HIGH SCHOOL Because I have seen the Lord Rationale Anaphylaxis Policy Magdalene Catholic High School endeavours to provide a safe and supportive school environment for all students.
ESCA: Cinacalcet (Mimpara )
 ESCA: Cinacalcet (Mimpara ) Effective Shared Care Agreement for the Treatment of Primary hyperparathyroidism when parathyroidectomy is contraindicated or not clinically appropriate. Specialist details
ESCA: Cinacalcet (Mimpara ) Effective Shared Care Agreement for the Treatment of Primary hyperparathyroidism when parathyroidectomy is contraindicated or not clinically appropriate. Specialist details
Policy for Supporting. Children and Young People with. Diabetes in Education
 Policy for Supporting Children and Young People with Diabetes in Education December 2011 POLICY FOR SUPPORTING CHILDREN AND YOUNG PEOPLE WITH DIABETES IN EDUCATION Contents 1. Introduction 2. Before a
Policy for Supporting Children and Young People with Diabetes in Education December 2011 POLICY FOR SUPPORTING CHILDREN AND YOUNG PEOPLE WITH DIABETES IN EDUCATION Contents 1. Introduction 2. Before a
Essential Shared Care Agreement: Melatonin (Circadin, Bio-Melatonin)
 Ref No: E056 Essential Shared Care Agreement: Melatonin (Circadin, Bio-Melatonin) Please complete the following details: Patient s name, address, date of birth, and either complete when the consultant
Ref No: E056 Essential Shared Care Agreement: Melatonin (Circadin, Bio-Melatonin) Please complete the following details: Patient s name, address, date of birth, and either complete when the consultant
This document expires on Patient Group Direction No. VI 5
 Patient Group Direction (PGD) for The Administration of Combined Adsorbed Diphtheria, Tetanus, Pertussis, (Acellular Component) and Inactivated Poliomyelitis Vaccine (Infanrix-IPV ) to children aged between
Patient Group Direction (PGD) for The Administration of Combined Adsorbed Diphtheria, Tetanus, Pertussis, (Acellular Component) and Inactivated Poliomyelitis Vaccine (Infanrix-IPV ) to children aged between
Berkshire School Aged Immunisation Team Seasonal Influenza 2018/19
 Dear Parent/ Carer Re: Seasonal Influenza Vaccinations (flu) for children from Reception to year 5 This autumn the school immunisation team will be returning to your child s school to offer the seasonal
Dear Parent/ Carer Re: Seasonal Influenza Vaccinations (flu) for children from Reception to year 5 This autumn the school immunisation team will be returning to your child s school to offer the seasonal
APIDRA (insulin glulisine) injection vial APIDRA SOLOSTAR (insulin glulisine) subcutaneous solution pen-injector
 APIDRA SOLOSTAR (insulin glulisine) subcutaneous solution pen-injector Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific
APIDRA SOLOSTAR (insulin glulisine) subcutaneous solution pen-injector Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific
PROCEDURE FOR BLOOD GLUCOSE MONITORING
 PROCEDURE FOR BLOOD GLUCOSE MONITORING First Issued Issue Version Two Purpose of Issue/Description of Change Planned Review Date To promote safe and effective blood glucose monitoring using Trust equipment
PROCEDURE FOR BLOOD GLUCOSE MONITORING First Issued Issue Version Two Purpose of Issue/Description of Change Planned Review Date To promote safe and effective blood glucose monitoring using Trust equipment
Denosumab for the treatment of osteoporosis in postmenopausal women at increased risk of fractures
 APper apc15-0avgfh7 Shared Care Guideline Denosumab for the treatment of osteoporosis in postmenopausal women at increased risk of fractures For the latest information on interactions and adverse effects,
APper apc15-0avgfh7 Shared Care Guideline Denosumab for the treatment of osteoporosis in postmenopausal women at increased risk of fractures For the latest information on interactions and adverse effects,
OPTOMETRY PRACTICE INFORMATION: SUPPORTING GUIDANCE NOTES ON THE USE OF THE CLINICAL COMMUNICATIONS GATEWAY FOR ELECTRONIC REFERRAL
 OPTOMETRY PRACTICE INFORMATION: SUPPORTING GUIDANCE NOTES ON THE USE OF THE CLINICAL COMMUNICATIONS GATEWAY FOR ELECTRONIC REFERRAL Ophthalmic Contractors and their staff should read and implement the
OPTOMETRY PRACTICE INFORMATION: SUPPORTING GUIDANCE NOTES ON THE USE OF THE CLINICAL COMMUNICATIONS GATEWAY FOR ELECTRONIC REFERRAL Ophthalmic Contractors and their staff should read and implement the
WHAT WORKS? A comprehensive guide to safer needle and syringe selection for injecting drug users. tools for harm reduction
 WHAT WORKS? A comprehensive guide to safer needle and syringe selection for injecting drug users. tools for harm reduction What works? (product code: A299) Written by: Andrew Preston, Jon Derricott, and
WHAT WORKS? A comprehensive guide to safer needle and syringe selection for injecting drug users. tools for harm reduction What works? (product code: A299) Written by: Andrew Preston, Jon Derricott, and
Streamlining the lung diagnostic pathway (A87)
 Streamlining the lung diagnostic pathway (A87) Crawley CCG with Surrey and Sussex Healthcare NHS Trust Evaluation January 2017 Summary A new Straight-to-CT pathway for patients with an abnormal CXR result
Streamlining the lung diagnostic pathway (A87) Crawley CCG with Surrey and Sussex Healthcare NHS Trust Evaluation January 2017 Summary A new Straight-to-CT pathway for patients with an abnormal CXR result
ESSENTIAL SHARED CARE AGREEMENT FOR Risperidone, Olanzapine, Quetiapine, Aripiprazole, Amisulpride (South Staffordshire Only)
 E099 ESSENTIAL SHARED CARE AGREEMENT FOR Risperidone, Olanzapine, Quetiapine, Aripiprazole, Amisulpride (South Staffordshire Only) NOTE: Please complete details on P1 &3 Send one copy to GP, Patient and
E099 ESSENTIAL SHARED CARE AGREEMENT FOR Risperidone, Olanzapine, Quetiapine, Aripiprazole, Amisulpride (South Staffordshire Only) NOTE: Please complete details on P1 &3 Send one copy to GP, Patient and
ADMINISTRATION OF INSULIN
 STANDARD OPERATING PROCEDURE ADMINISTRATION OF INSULIN Issue History First issued April 2012 Issue Version Two Purpose of Issue/Description of Change Planned Review Date To promote the safe administration
STANDARD OPERATING PROCEDURE ADMINISTRATION OF INSULIN Issue History First issued April 2012 Issue Version Two Purpose of Issue/Description of Change Planned Review Date To promote the safe administration
Medicines Optimisation Team Standard Operating Procedure for Audit: High Dose Inhaled Corticosteroids
 Cannock Chase Clinical Commissioning Group South East Staffordshire and Seisdon Peninsula Clinical Commissioning Group Stafford and Surrounds Clinical Commissioning Group East Staffordshire Clinical Commissioning
Cannock Chase Clinical Commissioning Group South East Staffordshire and Seisdon Peninsula Clinical Commissioning Group Stafford and Surrounds Clinical Commissioning Group East Staffordshire Clinical Commissioning
Essential Shared Care Agreement: Lithium
 Ref No. E042 Essential Shared Care Agreement: Lithium Please complete the following details: Patient s name, address, date of birth Treatment (indication, dose regimen, brand name) Monitoring (proposed
Ref No. E042 Essential Shared Care Agreement: Lithium Please complete the following details: Patient s name, address, date of birth Treatment (indication, dose regimen, brand name) Monitoring (proposed
Consultation Group: Dr Amalia Mayo, Paediatric Consultant. Review Date: March Uncontrolled when printed. Version 2. Executive Sign-Off
 Policy For The Adjustment Of Insulin Injections By Paediatric Diabetes Specialist Nurses/Community Paediatric Nurses Diabetes Working With Children Within NHS Grampian Co-ordinators: Lead Paediatric Diabetes
Policy For The Adjustment Of Insulin Injections By Paediatric Diabetes Specialist Nurses/Community Paediatric Nurses Diabetes Working With Children Within NHS Grampian Co-ordinators: Lead Paediatric Diabetes
POSITION STATEMENT. Diabetic eye screening April Key points
 POSITION STATEMENT Title Date Diabetic eye screening April 2013 Key points Diabetic retinopathy is the most common cause of sight loss in the working age population (1) All people with any type of diabetes
POSITION STATEMENT Title Date Diabetic eye screening April 2013 Key points Diabetic retinopathy is the most common cause of sight loss in the working age population (1) All people with any type of diabetes
SystmOne Technical Guidance
 SystmOne Technical Guidance Updated February 2018 1 Dementia 18-19 Technical Solution SystmOne Implementation Guide Summary A resource to provide system support to clinicians and CCGs to: Data cleansing
SystmOne Technical Guidance Updated February 2018 1 Dementia 18-19 Technical Solution SystmOne Implementation Guide Summary A resource to provide system support to clinicians and CCGs to: Data cleansing
SENSOR ELECTRODE/PRECISION (Also Medisense Companion)
 DIABETIC PSEUDO-DIN LIST - Revised Mar 2017 For Provinces not listed, please submit using the diabetic pseudo DIN listed in your provincial formulary or the AB/ON pseudo DIN (except for insulin). Pricing
DIABETIC PSEUDO-DIN LIST - Revised Mar 2017 For Provinces not listed, please submit using the diabetic pseudo DIN listed in your provincial formulary or the AB/ON pseudo DIN (except for insulin). Pricing
The Society welcomes the opportunity to respond the Department of Health s consultation on generic substitution.
 Lindsey Gilpin Chairman of the English Pharmacy Board Telephone: 020 7572 2519 Fax: 0207 572 2501 e-mail: howard.duff@rpsgb.org 25 th March 2010 Dear Sir/Madam, Consultation on the proposals to implement
Lindsey Gilpin Chairman of the English Pharmacy Board Telephone: 020 7572 2519 Fax: 0207 572 2501 e-mail: howard.duff@rpsgb.org 25 th March 2010 Dear Sir/Madam, Consultation on the proposals to implement
for adults engaged with the Family Wellbeing Service Isle of Wight In Community Pharmacy for Isle of Wight Public Health Commissioned Services
 The supply of Champix (Varenicline) Tablets 500mcg and 1mg by registered community pharmacists for smoking cessation / management of nicotine withdrawal for adults engaged with the Family Wellbeing Service
The supply of Champix (Varenicline) Tablets 500mcg and 1mg by registered community pharmacists for smoking cessation / management of nicotine withdrawal for adults engaged with the Family Wellbeing Service
CCG/PH updates FAO Community Pharmacies
 CCG/PH updates FAO Community Pharmacies 1. Stop smoking county update 2.Gluten Free Prescribing East and West CCG 3.Discontinuation of Minor Ailments West CCG 4.Changes to OTC prescribing East CCG 5.changes
CCG/PH updates FAO Community Pharmacies 1. Stop smoking county update 2.Gluten Free Prescribing East and West CCG 3.Discontinuation of Minor Ailments West CCG 4.Changes to OTC prescribing East CCG 5.changes
Injection technique for different needle lengths
 Injection technique for different needle lengths Use the correct technique to better manage your diabetes 1 Why it s important to use the shortest needles available Reduce your risk of injecting into the
Injection technique for different needle lengths Use the correct technique to better manage your diabetes 1 Why it s important to use the shortest needles available Reduce your risk of injecting into the
Medicines Optimisation Prescribing Audit: Long acting muscarinic antagonist (LAMA ) review
 Medicines Optimisation Prescribing Audit: Long acting muscarinic antagonist (LAMA ) review Aim To identify all patients prescribed long acting muscarinic antagonists (LAMAs) and where clinically appropriate
Medicines Optimisation Prescribing Audit: Long acting muscarinic antagonist (LAMA ) review Aim To identify all patients prescribed long acting muscarinic antagonists (LAMAs) and where clinically appropriate
medicines_management/correspondence/pathway-for-the-managed- Access-of-FreeStyle-Libre.
 Pathway for the Managed Access of FreeStyle Libre (Flash Glucose monitoring) for Adults and Children in the care of Trust Specialist Diabetes Clinics in Northern Ireland www.hscboard.hscni.net/download/publications/pharmacy_and_
Pathway for the Managed Access of FreeStyle Libre (Flash Glucose monitoring) for Adults and Children in the care of Trust Specialist Diabetes Clinics in Northern Ireland www.hscboard.hscni.net/download/publications/pharmacy_and_
Putting NICE guidance into practice. Resource impact report: Hearing loss in adults: assessment and management (NG98)
 Putting NICE guidance into practice Resource impact report: Hearing loss in adults: assessment and management (NG98) Published: June 2018 Summary This report focuses on the recommendation from NICE s guideline
Putting NICE guidance into practice Resource impact report: Hearing loss in adults: assessment and management (NG98) Published: June 2018 Summary This report focuses on the recommendation from NICE s guideline
North of England Cancer Network. Policies and Procedures. Standards for the Safe Use of Oral Anticancer Medicines
 \ North of England Cancer Network Policies and Procedures Standards for the Safe Use of Oral Anticancer Medicines NECN Oral Anticancer medicine Policy version 1.6 Page 1 of 17 Issue Date: Feb 2017 Contents
\ North of England Cancer Network Policies and Procedures Standards for the Safe Use of Oral Anticancer Medicines NECN Oral Anticancer medicine Policy version 1.6 Page 1 of 17 Issue Date: Feb 2017 Contents
Patient Group Direction for Varicella vaccine Version: Varicella Start Date: 1 st May 2014 Expiry Date: 30 th April 2017
 Patient Group Direction for Varicella vaccine Version: Varicella 2014.1 Start Date: 1 st May 2014 Expiry Date: 30 th April 2017 THIS PATIENT GROUP DIRECTION HAS BEEN AGREED BY THE FOLLOWING ORGANISATIONS:
Patient Group Direction for Varicella vaccine Version: Varicella 2014.1 Start Date: 1 st May 2014 Expiry Date: 30 th April 2017 THIS PATIENT GROUP DIRECTION HAS BEEN AGREED BY THE FOLLOWING ORGANISATIONS:
Country Health SA Local Health Network. Title: Subcutaneous Insulin Administration in the Community Setting
 Protocol (Clinical) Title: Subcutaneous Insulin Administration in the Community Setting Protocol developed by: CHSALHN Diabetes Service Protocol Sponsor: CHSALHN, Executive Director, Medical Services Approved
Protocol (Clinical) Title: Subcutaneous Insulin Administration in the Community Setting Protocol developed by: CHSALHN Diabetes Service Protocol Sponsor: CHSALHN, Executive Director, Medical Services Approved
Procedure for the prescribing and administration of insulin
 Procedure for the prescribing and administration of insulin Author: Lilian Baxendale and Heather Beadle Designation: Clinical Governance Pharmacists Version: Version 3 Date: December 2016 Date Approved:
Procedure for the prescribing and administration of insulin Author: Lilian Baxendale and Heather Beadle Designation: Clinical Governance Pharmacists Version: Version 3 Date: December 2016 Date Approved:
ADMELOG, NOVOLIN, NOVOLOG, and FIASP
 ADMELOG, NOVOLIN, NOVOLOG, and FIASP Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Pharmacy
ADMELOG, NOVOLIN, NOVOLOG, and FIASP Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Pharmacy
All Saints First School Administering of Medicines Policy
 All Saints First School Administering of Medicines Policy Success Indicators The following indicators will demonstrate the level of compliance with this policy and its procedures: a) Employees who are
All Saints First School Administering of Medicines Policy Success Indicators The following indicators will demonstrate the level of compliance with this policy and its procedures: a) Employees who are
This paper outlines the engagement activity that took place, and provides key themes from the 57 written responses received.
 Agenda item: 5.4 Subject: Presented by: Prepared by: Submitted to: Specialist Fertility Services Dr Dustyn Saint SNCCG Commissioning Team SNCCG Communications and Engagement Team SNCCG Governing Body Date:
Agenda item: 5.4 Subject: Presented by: Prepared by: Submitted to: Specialist Fertility Services Dr Dustyn Saint SNCCG Commissioning Team SNCCG Communications and Engagement Team SNCCG Governing Body Date:
LEVEMIR (insulin detemir) subcutaneous solution LEVEMIR FLEXTOUCH (insulin detemir) subcutaneous solution pen-injector
 LEVEMIR FLEXTOUCH (insulin detemir) subcutaneous solution pen-injector Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific
LEVEMIR FLEXTOUCH (insulin detemir) subcutaneous solution pen-injector Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific
URGENT: Important Safety Information
 July 2018 URGENT: Important Safety Information Subject: Notice of New Special Handling Instructions due to Potential for Cracked Needle Hubs and Particulate in Multiple Carpuject Luer Lock Glass Products-Corrected
July 2018 URGENT: Important Safety Information Subject: Notice of New Special Handling Instructions due to Potential for Cracked Needle Hubs and Particulate in Multiple Carpuject Luer Lock Glass Products-Corrected
Stoma Additional Products Savings Guidance
 Stoma Additional Products Savings Guidance Stoma care additional products (also known as ancillary products) are prescribed for individual patients to help prevent sore skin and other problems developing
Stoma Additional Products Savings Guidance Stoma care additional products (also known as ancillary products) are prescribed for individual patients to help prevent sore skin and other problems developing
How can I access flash glucose monitoring if I need it? Support pack. This pack will help you to find out more about flash and how you can access it.
 How can I access flash glucose monitoring if I need it? Support pack This pack will help you to find out more about flash and how you can access it. Reviewed March 2019 Introduction Following several major
How can I access flash glucose monitoring if I need it? Support pack This pack will help you to find out more about flash and how you can access it. Reviewed March 2019 Introduction Following several major
