A Randomized Trial of High-Dose Compared with Low-Dose Omega-3 Fatty Acids in Severe IgA Nephropathy
|
|
|
- Claribel Malone
- 6 years ago
- Views:
Transcription
1 J Am Soc Nephrol 12: , 2001 A Randomized Trial of High-Dose Compared with Low-Dose Omega-3 Fatty Acids in Severe IgA Nephropathy JAMES V. DONADIO, JR.,* TIMOTHY S. LARSON,* ERIK J. BERGSTRALH, and JOSEPH P. GRANDE,* for the Mayo Nephrology Collaborative Group 1 *Division of Nephrology, Department of Internal Medicine, Division of Anatomic Pathology, Department of Laboratory Medicine and Pathology, and the Section of Biostatistics, Mayo Foundation, Rochester, Minnesota. Abstract. Tested was the hypothesis that high-dose omega ( )-3 fatty acids will be more effective than low-dose -3 fatty acids in preserving renal function in patients with severe IgA nephropathy in a randomized, open-label, parallel-group clinical trial. Patients were assigned to receive either high-dose fatty acids (EPA 3.76 g and DHA 2.94 g) or low-dose fatty acids (EPA 1.88 g and DHA 1.47 g), both given daily in a highly purified ethyl ester concentrate (Omacor). Patients were treated for a minimum of 2 yr in the absence of a treatment failure or until study closure (January 2000). Seventy-three patients were enrolled in the trial with two ranges of elevated serum creatinine (SC): 63 patients (86%) with a range of 1.5 to 2.9 mg/dl and 10 patients (14%) with a range of 3.0 to 4.9 mg/dl. The primary end point, within-patient rates of change in SC (2-yr minimum), showed an annualized median increase in SC of 0.08 mg/dl per yr in the low-dose group and 0.10 mg/dl per yr in the high-dose group (P 0.51). Patients in the lower entry SC range had lower SC slopes (P 0.02) and less end-stage renal disease (ESRD) (P 0.001) compared with those in the higher entry SC range. No patient died, and 18 patients developed ESRD: 10 in the low-dose group and 8 in the high-dose group (P 0.56). SC slopes were significantly lower, and survival free of ESRD was significantly higher (both, P 0.04) in the 63 Omacor-treated patients compared with the 22 placebo-treated patients from our previously reported clinical trial in which both groups had a similar level of renal impairment. Patient compliance was excellent, and no serious adverse events were noted. Low-dose and high-dose -3 fatty acids were similar in slowing the rate of renal function loss in high-risk patients with IgA nephropathy, particularly those with moderately advanced disease. We previously reported that omega ( )-3 polyunsaturated fatty acids significantly reduced renal disease progression in patients with idiopathic IgA nephropathy in a multicenter, placebocontrolled, randomized, 2-yr clinical trial (1). In a follow-up observational study extending beyond the 2-yr trial, long-term treatment with -3 fatty acids retarded renal progression consistent with the findings in the 2-yr trial (2). Both the primary end point an increase in serum creatinine of 50% or more and end-stage renal disease (ESRD) were substantially lower in the fatty acid treated group in observations extending 6.4 yr from randomization to last follow-up. Despite these encouraging results, in large cohort studies of patients with IgA nephropathy as many as 30 to 50% develop ESRD over a 20-yr period after diagnosis (3 5). More progressive disease relates to higher prevalence rates of well-recognized clinical markers of disease progression in patients with hypertension, reduced Received June 28, Accepted September 1, Correspondence to Dr. James V. Donadio, Jr., Emeritus Staff, Mayo Clinic, Mayo Foundation, MNCG Study Desk-Eisenberg S24, 200 First Street, S.W., Rochester, MN Phone: ; Fax: ; donadio.james@mayo.edu 1 The investigators of the Mayo Nephrology Collaborative Group are listed in the appendix / Journal of the American Society of Nephrology Copyright 2001 by the American Society of Nephrology renal function and abnormal proteinuria at diagnosis, and high glomerular histopathologic scores in their renal biopsy specimens (5). In 1995, in a new study, we proposed the hypothesis that high-dose -3 fatty acids will be more effective than low-dose -3 fatty acids in preserving renal function in patients with IgA nephropathy who are at high risk for developing progressive renal disease. We designed a prospective, randomized, comparative two-dose study of -3 fatty acids using a highly purified ethyl ester concentrate of -3 fatty acids, and we report here the effects of treatment on the renal outcome of patients with severe IgA nephropathy who participated in the trial. Materials and Methods Study Design The study was a randomized, open-label, parallel-group comparison of treatment for 2 yr with high-dose and low-dose -3 fatty acids. As this was an open-label study, patients who completed a minimum of 2 yr of treatment (in the absence of failure) were continued on their same regimens, either low dose or high dose, until study closure (January 2000). The quantities of -3 fatty acids we chose for the low-dose group approximated the amounts of C20:5 3 (EPA) and C22:6 3 (DHA) that were given to patients in the clinical trial (1) and extended observational study (2). In these earlier studies, patients were given 1.68 to 1.87 g/d EPA and 0.97 to 1.36 g/d DHA. In the present study, patients in the low-dose group received 1.88 g/d EPA and 1.47 g/d DHA; in the high-dose group, patients received 2 this
2 792 Journal of the American Society of Nephrology J Am Soc Nephrol 12: , 2001 amount. We did not consider it justifiable to have a placebo-control group because the primary renal end point occurred in only 6% of -3 fatty acid treated patients compared with 33% in the placebo group in our previously reported clinical trial (1). The trial was performed in 14 centers of the Mayo Nephrology Collaborative Group (see the Appendix). The study protocol was approved by the institutional review boards at each site, and all patients gave written, informed consent. Patient Selection and Treatment Patients aged 18 yr and older with a baseline serum creatinine of 1.5 to 4.9 mg/dl were eligible for the study if they had renal biopsyproven IgA nephropathy. The diagnosis of IgA nephropathy was based on histologic assessment of renal biopsy tissue performed by one investigator (J.P.G.) and was confirmed by immunofluorescence studies showing predominant or co-dominant mesangial deposition of IgA (5,6). Histopathologic assessment of renal injury was done by a semiquantitative scoring system, as described previously (5). In a previous study of 148 patients with IgA nephropathy, total glomerular score, derived from extent of mesangial cell proliferation, matrix increase, capillary loop narrowing or disruption, glomerular sclerosis, cellular crescents, and fibrous adhesions, was an independent predictor of adverse outcome (5). Therefore, total glomerular score at time of renal biopsy was assessed in this study. At study entry, patients were randomized within strata determined by (1) two ranges of elevated serum creatinine concentrations (1.5 to 2.9 mg/dl and 3.0 to 4.9 mg/dl), (2) previous treatment with -3 fatty acids, and (3) previous treatment with corticosteroids. One patient who had a central laboratory baseline serum creatinine of 1.4 mg/dl was randomized to low-dose Omacor (Pronova Biocare, Oslo, Norway). This patient s initial serum creatinine was 1.5 mg/dl measured 1 mo before in her local laboratory. We elected to include the patient in all analyses (in the 1.5 to 2.9 serum creatinine group). Omacor soft gelatin capsules, the study medicine used in this study, contain 1 g of a long-chain polyunsaturated -3 fatty acid ethyl ester concentrate including 4 mg of -tocopherol, which is added as an antioxidant. The concentrate is produced from high-quality fish oil and contains 47% EPA, 37% DHA, and 5 to 10% of other -3 fatty acids. In the manufacturing process, impurities, cholesterol, vitamins A and D, and potentially toxic compounds such as heavy metals, dioxins, and pesticides are removed below detection limits (7). The patients were randomly assigned to receive either Omacor 8 g/d given as eight 1-g soft gelatin capsules containing 3.76 g of EPA and 2.94 g of DHA for a total of 6.7 g of -3 fatty acids the high-dose group or Omacor 4 g/d given as four 1-g soft gelatin capsules containing 1.88 g of EPA and 1.47 g of DHA for a total of 3.35 g of -3 fatty acids the low-dose group. Dosing instruction was eight capsules or four capsules once a day ingested with a meal. Patients with hypertension were treated with the angiotensin-converting enzyme inhibitor enalapril (Vasotec; Merck Sharp & Dohme Laboratories, West Point, PA). When the target BP of 140/85 mmhg was not achieved, other antihypertensive drugs were added at the physician s discretion. Mild dietary sodium restriction limited to 90 mmol/d was advised. Patient Monitoring At study entry, complete medical histories were taken and physical examinations were performed for all patients. Initial clinical and laboratory results were sent to the coordinating center. Follow-up patient examinations and measurements of serum creatinine; total, high-density lipoprotein, and low-density lipoprotein cholesterol; triglycerides; and 24-h urine total protein were scheduled after 1.5, 6, 12, 18, and 24 mo of treatment. Patients who remained at risk for a renal failure event after 24 mo of treatment were continued on the same dose regimens to which they were originally assigned, and they had scheduled visits and the above named laboratory tests performed at 6-mo intervals until study closure (January 2000). Also, first morning urinalysis, hemoglobin, hematocrit, peripheral blood leukocytes, platelets, and serum potassium were collected and analyzed at the local center at each scheduled visit. All clinical and laboratory results were recorded on case report forms, forwarded to the coordinating center, and entered for data processing. To reduce variability, we analyzed each sample for serum creatinine and lipids and 24-h urine total protein at the Mayo Medical Laboratories (Rochester, MN) using standard methods. Patient compliance was ascertained by measuring the plasma phospholipid fatty acids EPA, DHA, and C20:4 6 (AA) at 6 wk and 6 mo versus baseline within and between the two Omacor-supplemented groups. The EPA/AA ratio, a particularly sensitive indicator of compliance, was also calculated. The fatty acid composition including -6 and -3 fatty acids of plasma phospholipid was measured by capillary gas-liquid chromatography (8,9). Study End Points The primary end point of the study was the estimated annual within-patient slope in serum creatinine, a determinant of change in renal function, over the entire time in study for each patient (2-yr minimum). Secondary end points were time to ESRD (defined as chronic, repetitive dialysis or receiving a renal transplant) and time to treatment failure, defined as the first occurrence of any of the following: (1) the development of ESRD, (2) a serious side effect leading to the inability to continue study medicines and death from any cause, and (3) refusal to continue participation (noncompliance). For noncompliant patients, we attempted to obtain follow-up information on subsequent renal failure and vital status following the intent-to-treat principle. Other variables monitored included changes in BP and 24-h urine total protein and changes in serum lipid profiles, peripheral blood counts, and serum potassium for safety. Statistical Analyses Univariate baseline comparisons between the two dose groups were done using the rank-sum (continuous data) or 2 (nominal data). Continuous factors were summarized using means and SD. Distributions that were particularly skewed or that had severe outliers (urine protein, triglycerides, serum creatinine slopes) were summarized using medians and the estimated interquartile range (25th, 75th percentiles). For patients who developed ESRD, only laboratory values taken before starting dialysis or receiving a renal transplant are included in the analyses. The primary end point for the study was rate of change in serum creatinine. Changes in serum creatinine over time were estimated in two ways. First, simple linear regression analysis (Y serum creatinine, X years since start of Omacor) was used to estimate annualized rates of change (slopes) in serum creatinine and reciprocal serum creatinine for each patient. Although reciprocal serum creatinine values were more linear over time than direct values, both measurements are presented as the interpretation of the direct changes is more familiar to readers. The rank-sum test was then used to compare dose groups with respect to serum creatinine slopes. Analyses followed intent-to-treat principles, with every attempt made to include all randomized patients in the analysis. However, one patient, randomized to high-dose Omacor, left the study before beginning treatment and was excluded from this analysis as only a single baseline serum creatinine reading was available.
3 J Am Soc Nephrol 12: , 2001 Omega-3 Fatty Acids in IgA Nephropathy 793 Second, generalized estimating equations were used to assess the average change over time in serum creatinine readings taken from low-dose versus high-dose Omacor-treated patients (10). In contrast to the first approach, this method eliminates the need first to estimate within patient slopes, while taking into account not only the repeated nature of the data but also the variable number of readings per patient. Furthermore, all patients contributed to the analysis. The dependent variable was serum creatinine (and also reciprocal serum creatinine). Predictor variables were Omacor dose group, time since start of therapy, and dose by time interaction. As readings within patients tend to be correlated, patient was used as a clustering factor. Both an autoregressive reading (correlations between readings within a patient assumed to decrease with time) and an exchangeable reading (correlations between readings within a patient assumed constant across time) were used. Secondary end points included time from start of therapy to development of ESRD and time to either development of ESRD or discontinuation of treatment as a result of an adverse event or noncompliance (refusal to continue). The cumulative percentage of patients who were free of these events was estimated using the Kaplan-Meier method. Dose groups were compared using the log-rank test. All randomized patients were included in these comparisons. The one patient who dropped out before beginning therapy was censored with 0 follow-up for the ESRD end point and counted as an event with 0 follow-up for the ESRD or discontinuation of treatment end point. Within-dose group 1-yr and 2-yr changes in BP, urine protein, serum lipids, peripheral blood counts, and serum potassium were tested using the sign-rank test. All tests were two-sided with -level Results Between October 1995 and January 1998, 103 patients with IgA nephropathy were screened for study. Seventy-three patients met the clinical criteria for eligibility and were willing to enter the study. Except for one center that recruited 33 patients, the remaining 13 centers all had 7 or fewer patients each, precluding the analysis of an effect of study center. In the prerandomization strata, 32 patients who were allocated to the low-dose Omacor group and 31 patients who were allocated to the high-dose Omacor group had baseline serum creatinines of 1.5 to 2.9 mg/dl, whereas 5 patients who were assigned to each treatment group had baseline serum creatinines of 3.0 to 4.9 mg/dl. Twenty-one patients in the low-dose group and 22 in the high-dose group had previous treatment with -3 fatty acids, and 4 patients and 5 patients, respectively, had previous treatment with corticosteroids. Thirty-seven patients were assigned to receive low-dose Omacor, and 36 were assigned to receive high-dose Omacor. The clinical and laboratory characteristics of the patients in the two treatment groups were similar (Table 1), as were preceding illnesses, including upper respiratory infections, episodic macroscopic hematuria, fever, and flu-like illnesses. There was no difference in glomerular histopathologic score of patients (Table 1), indicating that the extent of histopathologic injury was similar in the two groups. In the follow-up of the 73 patients who entered the trial, there were no deaths and 18 developed ESRD. Of the remaining 55 patients, 42 stayed on treatment through July 1, 1999 (the earliest possible date for completing the final study visit), whereas 13 patients left the study early because of refusal to continue participation (8 patients), moving away from a study center (2 patients), lack of follow-up (1 patient), or sustaining an adverse event (2 patients). Of the two patients who withdrew from the study early because of an adverse event, one patient had gastrointestinal intolerance (indigestion) after the patient had taken Omacor 8 g/d for 18 mo. The symptom resolved promptly after the drug was stopped. The second patient, with a history of Barrett s esophagus, discontinued medication (4 g/d) approximately 6 wk after study entry as a result of an exacerbation of reflux esophagitis. Outcome: Annual within-patient Slopes in Serum Creatinine Based on within-patient serum creatinine profiles (Figure 1), we estimated within-patient annualized rates of change (slopes) for serum creatinine. These slopes are summarized in Table 2 and Figure 2. Although both groups demonstrated slopes that were significantly different from 0 (4 g, P 0.008; 8 g, P [sign-rank test]), no significant differences were noted between the two Omacor dose groups (P 0.51 [rank-sum test]). Additional analyses based on slopes derived from reciprocal serum creatinine profiles also showed no significant differences between Omacor dose groups (P 0.58). Further analyses based on generalized estimating equations (see the Materials and Methods section) also found no significant evidence of an Omacor dose effect on follow-up serum creatinine levels (all P values 0.20). For example, the mean slope in reciprocal serum creatinine was estimated to be 0.03 dl/mg per yr for the low-dose group compared with 0.02 dl/mg per yr for the high-dose group (P 0.58). Annualized median change in serum creatinine was not different in previously -3 fatty acid treated (0.107 mg/dl per yr) versus non-previously -3 fatty acid treated patients (0.064 mg/dl per yr; P 0.36 [rank-sum test]) or in reciprocal serum creatinine slopes comparing previously -3 fatty acid treated ( dl/mg per yr) with non-previously -fatty acid treated ( dl/mg per yr) patients (P 0.71 [rank-sum test]). It is apparent that the majority of patients who had baseline serum creatinines of 1.5 to 2.9 mg/dl at study entry (n 63) had stable renal function in both the low-dose and high-dose Omacor groups (Figure 1). Patients with initial serum creatinine levels of 1.5 to 2.9 mg/dl had significantly lower rates of deterioration in renal function (median creatinine slope 0.08 versus 1.3 mg/dl per yr [P 0.019]; median reciprocal creatinine slope 0.02 versus 0.07 [P 0.062]) and less ESRD (13 versus 85% at 3 yr; P [log-rank test]) when compared with those with initial serum creatinines of 3.0 to 4.9 mg/dl. Large serum creatinine slopes ( 0.5 mg/dl per yr) were observed in 14 of 62 patients (23%) with baseline serum creatinines of 1.5 to 2.9 mg/dl compared with 7 of 10 (70%) patients with baseline serum creatinines of 3.0 to 4.9 mg/dl and in 17 of 18 patients who subsequently developed ESRD. Although we recognize that serum creatinine slopes 0.5 mg/dl per yr are nonlinear for expressing changes in GFR, such large slopes have clinical interpretation and correlate with the development of ESRD.
4 794 Journal of the American Society of Nephrology J Am Soc Nephrol 12: , 2001 Table 1. Characteristics of the patients in the low-dose and high-dose Omacor treatment groups at baseline a Characteristic Low [Mean (SD); N 37] Omacor Dose b High [Mean (SD); N 36] Age at randomization (yr) 46 (13) 45 (13) Male gender 84% 81% White race 92% 92% Hypertension c 92% 92% Blood pressure (mmhg) systolic 139 (16) 136 (16) diastolic 84 (10) 82 (9) Serum creatinine (mg/dl) 2.1 (0.7) 2.3 (0.7) Hematocrit (vol %) 39 (6.2) 38 (5.5) Peripheral blood leukocytes ( 10 9 /L) 7.2 (2.5) 7.1 (2.0) Platelets ( 10 9 /L) 214 (50) 251 (64) Serum potassium (meq/l) 4.8 (0.62) 4.8 (0.47) Serum cholesterol (mg/dl) total 217 (36) 228 (53) HDL 41 (15) 44 (14) LDL 135 (28) 146 (47) Serum triglycerides (mg/dl) d 178 (109, 277) 156 (114, 235) 24-h urine protein (mg/24 h) d 1791 (757, 2569) 1526 (702, 3662) Time from biopsy to randomization (yr) d 3.7 (2.1, 9.4) 2.6 (0.4, 6.0) Total glomerular histopathologic score e 5.5 (2.4) 4.9 (2.2) scores 5 (%) a HDL, high-density lipoprotein; LDL, low-density lipoprotein. b All P values for the comparison between the two groups were 0.05, except for platelets (P 0.05). c Hypertension was defined as a systolic pressure 140 mmhg or a diastolic pressure 85 mmhg or currently being treated for hypertension. d Median (25th, 75th percentiles) are presented due to extreme skewness. e Total scores based on a semiquantitative scoring system (see Materials and Methods section). Renal Failure Events Survival free of ESRD was similar in the two treatment groups (Table 2, Figure 3), and survival without sustaining any event ESRD, an adverse event leading to discontinuation of treatment, or refusal to continue therapy was also similar in the low-dose and high-dose Omacor treatment groups (Table 2). Changes in BP, Proteinuria, Serum Lipids, Peripheral Blood Counts, and Potassium There were no significant within-dose group time trends in BP, proteinuria, serum lipids, peripheral blood counts, and potassium, with the exception that BP and serum triglycerides were lower in the low-dose Omacor group at 12 mo compared with baseline, and high-density lipoprotein and low-density lipoprotein cholesterol were lower in the high-dose Omacor group at 12 mo compared with baseline (Table 3). Although there was a decline in urine protein excretion over time in the low-dose group compared with no change in the high-dose group, the median annual slopes in proteinuria were not significantly different between treatment groups (P 0.17 [ranksum test]). Compliance with Treatment As a measure of compliance, in 56 patients who were not taking -3 fatty acids at study entry, plasma phospholipid fatty acid changes from pretreatment were significant for all studied fatty acids (Table 4). There were significant increases in EPA and DHA and significant reductions in AA levels at 6 wk and 6 mo after supplementation (P 0.01 for all values [paired t test]). With the exception of AA, fatty acid changes from baseline were, in general, higher for the high-dose compared with the low-dose group (Table 4). EPA/AA ratios were also significantly increased in both treatment groups and were higher at both 6 wk (P 0.007, two-tailed P value from rank-sum test) and 6 mo (P 0.002) in the high-dose group (Table 4). Adverse Events A total of nine patients (five at 4 g/d, four at 8 g/d) had adverse events, including the two aforementioned patients who withdrew early as a result of adverse events. Of the remaining seven patients, one patient (4 g/d) had episodes of cryptogenic gastrointestinal bleeding before, during, and after study participation. Omacor was discontinued in this patient 2 yr after
5 J Am Soc Nephrol 12: , 2001 Omega-3 Fatty Acids in IgA Nephropathy 795 Figure 1. Serum creatinine profiles in patients who had IgA nephropathy and who were treated with low-dose (A) and high-dose (B) Omacor. The serum creatinines are plotted and connected for each patient and include all readings before renal failure (if present). Figure 2. Annualized rate of change in renal function in patients who had IgA nephropathy and who were treated with low-dose and high-dose Omacor. The annual rates of change (slopes) for serum creatinine were computed for each patient by linear regression analysis using all results before renal failure (if present). The annualized median change in serum creatinine was 0.08 mg/dl per yr in the low-dose group and 0.10 mg/dl per yr in the high-dose group (P 0.51)., median. study enrollment during an episode of gastrointestinal bleeding 1 mo before the patient was started on maintenance hemodialysis for ESRD. Another patient (4 g/d) discontinued therapy after development of rectal bleeding from hemorrhoids 48 mo after study entry. This patient had advanced renal failure at the time. Other single adverse events noted during the course of study participation included one episode of diverticulitis (4 g/d), an episode of pneumonitis and possible pancreatitis (8 g/d), development of asymptomatic atrial fibrillation (8 g/d), hyperkalemia (4 g/d) that resolved after discontinuing angiotensin-converting enzyme inhibitor therapy, and development of hemiparesis as a result of ischemic cerebrovascular disease (8 g/d). None of these events was thought to be related directly to study medication. Discussion In this multicenter, randomized, 2-yr, comparative two-dose trial, both low-dose and high-dose -3 fatty acids, given daily
6 796 Journal of the American Society of Nephrology J Am Soc Nephrol 12: , 2001 Table 2. Results of low-dose versus high-dose Omacor regarding primary and secondary end points a End Point Omacor Dose Low (n 37) High (n 36) P Value Primary slopes in serum creatinine (mg/dl per yr) median (25th, 75th percentile) 0.08 ( 0.03, 0.79) 0.10 d ( 0.02, 0.70) 0.51 b slope 0.5 mg/dl per yr (no.) Secondary ESRD number of events c event-free [% (SEM)] at 2 yr 86 (6.0) 80 (6.9) at 3 yr 73 (8.4) 76 (7.6) ESRD, adverse event, or noncompliant e number of events c event-free [% (SEM)] at 2 yr 78 (6.9) 72 (7.5) at 3 yr 62 (9.2) 65 (8.2) a ESRD, end-stage renal disease. b Based on two-sided rank-sum test. Note that similar results obtained when using slopes based on reciprocal serum creatinine (medians 0.02 low dose, 0.03 high dose; P 0.58). c Based on two-sided log-rank test. d Excludes one patient with a single serum creatinine who dropped out of the study before beginning treatment. This patient is included in the analyses of secondary end points. e Includes patients with ESRD or an adverse event leading to discontinuation of treatment or refusal to continue therapy. Figure 3. Cumulative percentage of patients who had IgA nephropathy and who were treated with low-dose and high-dose Omacor and did not have renal failure during the treatment period. In the low-dose group, 73% of the patients (SEM 8.4%) were free of renal failure at 3 yr compared with 76% (7.6) in the high-dose group (P 0.56). Numbers of patients who were at risk at 3 yr were 12 in the low-dose group and 15 in the high-dose group. as a highly purified ethyl ester concentrate of EPA and DHA (Omacor), were equally effective in slowing the rate of loss of renal function in patients who were at high risk for renal progression, all having impaired renal function at the start of treatment. Changes in renal function (serum creatinine) were only 0.08 mg/dl per yr in the low-dose group and 0.10 mg/dl per yr in the high-dose group. Patients who had the most stable renal course in both the low-dose and high-dose groups were those who had less impaired renal function upon entering the study with a baseline
7 J Am Soc Nephrol 12: , 2001 Omega-3 Fatty Acids in IgA Nephropathy 797 Table 3. Time trends in BP, urine protein, lipids, and CBC by Omacor dose a Low Omacor Dose High Omacor Dose Clinical Variable Study Visit Study Visit 0 (Baseline) (n 37) 12 Mo (n 32) 24 Mo (n 28) 0 (Baseline) (n 36) 12 Mo (n 32) 24 Mo (n 26) Systolic BP (mmhg) 139 (16) 132 (16) b 136 (17) 136 (16) 135 (20) 136 (13) Diastolic BP (mmhg) 84 (10) 80 (9.4) b 81 (8.9) 82 (9) 80 (8.0) 83 (8.0) Urine protein (mg/24 h) Cholesterol (mg/dl) 217 (36) 204 (63) 206 (50) 228 (53) 214 (56) 212 (57) HDL (mg/dl) 41 (15) 40 (15) 39 (13) 44 (14) 42 (10) b 42 (13) LDL (mg/dl) 135 (28) 124 (47) 132 (45) 146 (47) 132 (51) b 130 (48) Triglycerides (mg/dl) b Hgb (g/dl) 13.6 (3.1) 13.4 (2.0) 13.7 (2.1) 13.1 (1.9) 13.2 (2.4) 13.7 (2.0) Hct (vol %) 39 (6.2) 39 (5.9) 40 (6.2) 38 (5.5) 39 (7.0) 40 (6.7) Erythrocytes ( /L) 4.4 (.66) 4.4 (.62) 4.5 (.67) 4.3 (.61) 4.3 (.82) 4.5 (.70) Leukocytes ( 10 9 /L) 7.2 (2.5) 7.1 (2.3) 6.9 (2.2) 7.1 (2.0) 6.8 (2.4) 6.6 (1.2) Platelets/1000 ( 10 9 /L) 214 (50) 210 (53) 203 (53) 251 (64) 242 (57) 233 (48) Potassium (meq/l) 4.8 (.62) 4.7 (.65) 4.7 (.45) 4.8 (.47) 4.8 (.68) 4.7 (.60) a Tabled values are mean (SD). Readings after ESRD (10 patients in low-dose group, 8 patients in high-dose group) are excluded. Medians presented for urine protein and triglycerides due to extreme skewness. Hgb, hemoglobin; Hct, hematocrit. b P 0.05 for significant change from baseline to 12 mo. There were no significant changes from baseline to 24 mo. serum creatinine that ranged from 1.5 to 2.9 mg/dl. Sixty-three of the 73 patients (86%) who were enrolled in the trial fell into this category. Patients in this lower entry serum creatinine range had significantly lower serum creatinine slopes and less ESRD when compared with those in the higher entry serum creatinine range. Furthermore, large serum creatinine slopes 0.5 mg/dl per yr, a widely recognized clinical change in renal function were observed in 14 of 62 patients (23%) with lower baseline serum creatinine levels compared with 7 of 10 patients (70%) with higher baseline serum creatinines and in 17 of 18 patients who subsequently developed ESRD. This is not a surprising finding because most patients in the late stages of IgA nephropathy develop progressive renal failure despite various therapies that have been tried (11,12). Survival free of ESRD was also similar in the low-dose and high-dose Omacor treatment groups. An event-free survival rate from ESRD was 75% at 3 yr, representing a favorable outcome in view of the high-risk profile in the cohort of patients in this trial. To expand on this point, we compared both the primary endpoint changes in renal function by annualized rates of change in serum creatinine and survival free of ESRD in patients in the present study with patients in a placebo-control group in our previously reported clinical trial (1). Time to ESRD was defined in both study groups as time from start of therapy to the start of chronic, repetitive dialysis or receiving a renal transplant, whichever occurred first. To make the analysis comparable between the two groups, we compared outcomes in the 63 patients in the current trial with 22 patients in the placebo-treated group whose baseline serum creatinine concentrations were in the 1.4 to 2.9 mg/dl range (Table 5). Changes in serum creatinine slopes were significantly lower, and survival free of ESRD was significantly higher in the Omacor-treated patients from the present study compared with the placebo-treated patients from our previous study (1). Although this is not a comparison between patients who were contemporaneously randomized to treatment, it does examine the effects of treatment for patients with similar degrees of renal impairment (median serum creatinine is 1.8 mg/dl for both groups). That the patients in the present study were at high risk for developing progressive renal disease is evidenced not only by the aforementioned impaired pretreatment renal function that was part of the study design but also by the observations that 92% were hypertensive and that urinary protein excretion was in excess of 1.5 g/24 h at study entry. These clinical variables are important predictors of poor outcome in IgA nephropathy (5,13 17). In addition, total glomerular histopathologic score, a semiquantitative index of renal injury and an independent predictor of renal failure (5), was increased in the renal biopsy specimens of 50% of the patients. In a previous study of 148 patients with IgA nephropathy, we found that total glomerular histopathologic scores greater than 5 were associated with adverse outcome (5). Patient compliance was excellent as ascertained by the expected enhancement of plasma phospholipid EPA and DHA and suppression of AA after Omacor supplementation (1,18). EPA and DHA levels on treatment were higher in the highdose group compared with the low-dose group. Yet, as already shown, treatment with high-dose -3 fatty acids had no added beneficial effect on preserving renal function. Omacor was well tolerated as only two patients discontinued treatment as a result of gastrointestinal intolerance. There were no unfavorable effects on serum lipid profiles, hematocrits, peripheral blood leukocytes, or platelets.
8 798 Journal of the American Society of Nephrology J Am Soc Nephrol 12: , 2001 Table 4. Fatty acid composition of plasma phospholipid in low-dose and high-dose Omacor-treated patients with IgA nephropathy who were not taking omega-3 fatty acids at study entry a Omacor Dose Fatty Acid Visit Low High N Mean (SD) N Mean (SD) Low Versus High P Value b C20:5 3 (EPA) (0.5) (0.6) wk (1.3) (1.3) 6 mo (1.3) (1.8) 6 wk (1.2) c (1.5) c mo (1.3) c (1.9) c C22:6 3 (DHA) (1.5) (1.4) wk (1.2) (1.3) 6 mo (1.3) (1.6) 6 wk (1.5) c (1.7) c mo (1.5) c (2.0) c C22:4 6 (AA) (2.5) (2.6) wk (1.8) (1.9) 6 mo (1.9) (1.8) 6 wk (2.0) c (1.8) c mo (2.0) c (1.8) c 0.66 EPA/AA (0.07) (0.08) wk (0.29) (0.22) 6 mo (0.40) (0.31) 6 wk (0.26) c (0.24) c mo (0.39) c (0.33) c a Data for each fatty acid are expressed as the weight percentage of total fatty acids and calculated as millimoles per mole divided by 100. b P value from two-sided rank-sum test comparing low-dose versus high-dose Omacor. c P value from two-sided paired t test for within-dose group test of no significant change from pretreatment (visit 0) is Because treatment with high-dose -3 fatty acids provided no added benefit, we believe that it is appropriate to recommend low-dose -3 fatty acids in the treatment of high-risk patients with IgA nephropathy, including those with moderately advanced renal disease, for example, for those patients whose serum creatinines were in the lower entry range of 1.5 to 2.9 mg/dl. We previously determined that this lower dose of -3 fatty acids, composed of 1.9 g of EPA and of 1.4 g DHA, efficiently enhanced the EPA and DHA and total -3 polyunsaturated fatty acids of plasma phospholipids (18) and can be recommended on the strength of the present findings. For convenience, the ethyl ester -3 fatty acid concentrate Omacor, which provides a concentrated preparation of EPA and DHA and was used in this trial, allowed patients who were in the Table 5. Comparison of renal end points in Omacor- and placebo-treated patients who had pretreatment serum creatinine levels ranging from 1.4- to 2.9 mg/dl End Point Omacor (n 63) Treatment Group Placebo (n 22) Omacor Versus Placebo P Value Slopes in serum creatinine (mg/dl per yr) median (25th, 75th percentile) ( 0.02, 0.39) (0.035, 0.98) ESRD number of events 10 9 event-free at 3 yr [% (SE)] 85 (5.1) 64 (11.1) 0.04 a 0.04 b a Based on two-sided rank-sum test. One patient from each group did not have sufficient data for a serum creatinine slope. b Based on a two-sided log-rank test.
9 J Am Soc Nephrol 12: , 2001 Omega-3 Fatty Acids in IgA Nephropathy 799 low-dose group to consume only four soft gelatin capsules daily. This compares with 12 capsules daily necessary to take in 1.9 g of EPA and 1.4 g of DHA contained in the over-thecounter products that generally are 30% concentrates of -3 fatty acids. As no other comparative-dose studies have been performed using -3 fatty acids, we do not know whether a lower dose of -3 fatty acids might be effective in slowing renal progression for high-risk patients with IgA nephropathy. Appendix: The Mayo Nephrology Collaborative Group Clinical Coordinating Center: T. Larson, D. Spencer, J. Grande, and J. Donadio; Section of Biostatistics: E. Bergstralh and D. Rademacher; Mayo Clinic (Rochester, MN): J. Gloor, J. McCarthy, J. Mitchell, T. Schwab, and V. Torres; Mayo Clinic (Scottsdale, AZ): R. Heilman and D. Wochos; Duluth Clinic, Ltd. (Duluth, MN): R. Hellman and T. Russ; Marshfield Clinic (Marshfield, WI): R. Dart; Consultative Nephrology (Lincoln, NE): S. Youngberg; McFarland Clinic (Ames, IA): J. Graves; Arms, Dodge, Robinson, Wilber & Crouch, Inc. (Kansas City, MO): J. Mertz; Columbia Nephrology Association (Columbia, SC): W. Edwards; Nephrology and Hypertension (Columbia, MO): M. Vaporean and W. Winkelmeyer; West Michigan Nephrology, P.C. (Muskegon, MI): G. Downer; Renal Associates, P.A. (San Antonio, TX): M. Isbell; Nephrology Associates of Syracuse, P.C. (Syracuse, NY): R. Scheer; Lynchburg Nephrology (Lynchburg, VA): J. Nielsen; Gunderson Clinic (LaCrosse, WI): J. Singer. Acknowledgments The authors acknowledge Dorothy Spencer for her assistance as Clinical Coordinator at the Clinical Coordinating Center (Rochester, MN); Diana Rademacher for her assistance with statistical analyses; Cherish Grabau for secretarial support; and Dr. Bruce Holub, Department of Nutritional Sciences, University of Guelph (Guelph, Ontario, Canada), for the measurement of plasma phospholipids. This work was supported by research grants from Pronova Biocare a.s. (Oslo, Norway) and Mayo Foundation (Rochester, MN). References 1. Donadio JV Jr, Bergstralh EJ, Offord KP, Spencer DC, Holley KE, for the Mayo Nephrology Collaborative Group: A controlled trial of fish oil in IgA nephropathy. N Engl J Med 331: , Donadio JV Jr, Grande JP, Bergstralh EJ, Dart RA, Larson TS, Spencer DC, for the Mayo Nephrology Collaborative Group: The long-term outcome of patients with IgA nephropathy treated with fish oil in a controlled trial. J Am Soc Nephrol 10: , Velo M, Lozano L, Egido J, Guiterrez-Millet V, Hernando L: Natural history of IgA nephropathy in patients followed up for more than ten years in Spain. Semin Nephrol 7: , Droz D: Natural history of primary glomerulonephritis with mesangial deposits of IgA. Contrib Nephrol 2: , Radford GM Jr, Donadio JV Jr, Bergstralh EJ, Grande JP: Predicting renal outcome in IgA nephropathy. J Am Soc Nephrol 8: , Donadio JV Jr, Grande JP: Immunoglobulin A nephropathy: A clinical perspective. J Am Soc Nephrol 8: , Omacor Investigator Brochure, Oslo, Norway, Pronova Biocare, a.s., August Bligh EG, Dyer WJ: A rapid method of total lipid extraction and purification. Can J Biochem Physiol 37: , Holub BJ, Skeaff CM: Nutritional regulation of cellular phosphatidylinositol. Methods Enzymol 141: , Liang KY, Zeger SL: Longitudinal data analysis using generalized linear models. Biometrika 73: 13 22, Cheng IKP, Chan PCK, Chan MK: The effect of fish oil dietary supplement on the progression of mesangial IgA glomerulonephritis. Nephrol Dial Transplant 5: , Fung SM, Ferrill MJ, Norton LL: Fish oil therapy in IgA nephropathy. Ann Pharmacother 31: , Beukof JR, Kardaun O, Schaafsma W, Poortema K, Donker AJ, Hoedemaeker PJ, van der Hem GK: Toward individual prognosis of IgA nephropathy. Clin Invest 29: , Rekola S, Bergstrand A, Bucht H: IgA nephropathy: A retrospective evaluation of prognostic indices in 176 patients. Scand J Urol Nephrol 23: 37 50, Bogenschutz O, Bohle A, Batz C, Wehrmann M, Pressler H, Kendziorra H, Gartner HV: IgA nephritis: On the importance of morphological and clinical parameters in the long-term prognosis of 239 patients. Am J Nephrol 10: , Ibels LS, Gyory AZ: IgA nephropathy: Analysis of the natural history, important factors in the progression of renal disease, and a review of the literature. Medicine 73: , Hogg RJ, Silva FG, Wyatt RJ, Reisch JS, Argyle JC, Savino DA: Prognostic indicators in children with IgA nephropathy: Report of the Southwest Pediatric Nephrology Study Group. Pediatr Nephrol 8: 15 20, Holman RT, Johnson SB, Bibus D, Spencer DC, Donadio JV Jr: Essential fatty acid deficiency profiles in idiopathic immunoglobulin A nephropathy. Am J Kidney Dis 23: , 1994 Access to UpToDate on-line is available for additional clinical information at
The CARI Guidelines Caring for Australasians with Renal Impairment. Specific management of IgA nephropathy: role of fish oil
 Specific management of IgA nephropathy: role of fish oil Date written: July 2005 Final submission: September 2005 Author: Merlin Thomas GUIDELINES Early and prolonged treatment with fish oil may retard
Specific management of IgA nephropathy: role of fish oil Date written: July 2005 Final submission: September 2005 Author: Merlin Thomas GUIDELINES Early and prolonged treatment with fish oil may retard
The CARI Guidelines Caring for Australasians with Renal Impairment. Specific management of IgA nephropathy: role of steroid therapy GUIDELINES
 Specific management of IgA nephropathy: role of steroid therapy Date written: July 2005 Final submission: September 2005 Author: Merlin Thomas GUIDELINES Steroid therapy may protect against progressive
Specific management of IgA nephropathy: role of steroid therapy Date written: July 2005 Final submission: September 2005 Author: Merlin Thomas GUIDELINES Steroid therapy may protect against progressive
Reducing proteinuria
 Date written: May 2005 Final submission: October 2005 Author: Adrian Gillin Reducing proteinuria GUIDELINES a. The beneficial effect of treatment regimens that include angiotensinconverting enzyme inhibitors
Date written: May 2005 Final submission: October 2005 Author: Adrian Gillin Reducing proteinuria GUIDELINES a. The beneficial effect of treatment regimens that include angiotensinconverting enzyme inhibitors
The CARI Guidelines Caring for Australasians with Renal Impairment
 Specific management of IgA nephropathy: role of triple therapy and cytotoxic therapy Date written: July 2005 Final submission: September 2005 Author: Merlin Thomas GUIDELINES a. Triple therapy with cyclophosphamide,
Specific management of IgA nephropathy: role of triple therapy and cytotoxic therapy Date written: July 2005 Final submission: September 2005 Author: Merlin Thomas GUIDELINES a. Triple therapy with cyclophosphamide,
The CARI Guidelines Caring for Australasians with Renal Impairment. Membranous nephropathy role of steroids GUIDELINES
 Membranous nephropathy role of steroids Date written: July 2005 Final submission: September 2005 Author: Merlin Thomas GUIDELINES There is currently no data to support the use of short-term courses of
Membranous nephropathy role of steroids Date written: July 2005 Final submission: September 2005 Author: Merlin Thomas GUIDELINES There is currently no data to support the use of short-term courses of
Diabetic Nephropathy
 Diabetic Nephropathy Outline Introduction of diabetic nephropathy Manifestations of diabetic nephropathy Staging of diabetic nephropathy Microalbuminuria Diagnosis of diabetic nephropathy Treatment of
Diabetic Nephropathy Outline Introduction of diabetic nephropathy Manifestations of diabetic nephropathy Staging of diabetic nephropathy Microalbuminuria Diagnosis of diabetic nephropathy Treatment of
The Diabetes Kidney Disease Connection Missouri Foundation for Health February 26, 2009
 The Diabetes Kidney Disease Connection Missouri Foundation for Health February 26, 2009 Teresa Northcutt, RN BSN Primaris Program Manager, Prevention - CKD MO-09-01-CKD This material was prepared by Primaris,
The Diabetes Kidney Disease Connection Missouri Foundation for Health February 26, 2009 Teresa Northcutt, RN BSN Primaris Program Manager, Prevention - CKD MO-09-01-CKD This material was prepared by Primaris,
Supplementary Appendix
 Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Wanner C, Inzucchi SE, Lachin JM, et al. Empagliflozin and
Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Wanner C, Inzucchi SE, Lachin JM, et al. Empagliflozin and
CHAPTER 2 PRIMARY GLOMERULONEPHRITIS
 CHAPTER 2 Sunita Bavanandan Lim Soo Kun 19 5th Report of the 2.1: Introduction This chapter covers the main primary glomerulonephritis that were reported to the MRRB from the years 2005-2012. Minimal change
CHAPTER 2 Sunita Bavanandan Lim Soo Kun 19 5th Report of the 2.1: Introduction This chapter covers the main primary glomerulonephritis that were reported to the MRRB from the years 2005-2012. Minimal change
Protocol GTC : A Randomized, Open Label, Parallel Design Study of Sevelamer Hydrochloride (Renagel ) in Chronic Kidney Disease Patients.
 Protocol GTC-68-208: A Randomized, Open Label, Parallel Design Study of Sevelamer Hydrochloride (Renagel ) in Chronic Kidney Disease Patients. These results are supplied for informational purposes only.
Protocol GTC-68-208: A Randomized, Open Label, Parallel Design Study of Sevelamer Hydrochloride (Renagel ) in Chronic Kidney Disease Patients. These results are supplied for informational purposes only.
Current treatment recommendations in children with IgA nephropathy Selçuk Yüksel
 Current treatment recommendations in children with IgA nephropathy Selçuk Yüksel Department of Pediatric Nephrology Pamukkale University School of Medicine IgA Nephropathy The most common cause of primary
Current treatment recommendations in children with IgA nephropathy Selçuk Yüksel Department of Pediatric Nephrology Pamukkale University School of Medicine IgA Nephropathy The most common cause of primary
Objectives. Pre-dialysis CKD: The Problem. Pre-dialysis CKD: The Problem. Objectives
 The Role of the Primary Physician and the Nephrologist in the Management of Chronic Kidney Disease () By Brian Young, M.D. Assistant Clinical Professor of Medicine David Geffen School of Medicine at UCLA
The Role of the Primary Physician and the Nephrologist in the Management of Chronic Kidney Disease () By Brian Young, M.D. Assistant Clinical Professor of Medicine David Geffen School of Medicine at UCLA
Fish Oil to Increase Graft Patency. Fish Oil: Basic Mechanisms. Fish Oil. Inclusion/Exclusion Criteria. FISH Study. FISH Study
 Annual Scientific Meeting to Increase Graft Patency AKA Charmaine E. Lok, MD, MSc,FRCPC FISH Study Toronto General Hospital Toronto, On ASDIN, Washington, DC Annual Scientific Meeting February 6, 3 Annual
Annual Scientific Meeting to Increase Graft Patency AKA Charmaine E. Lok, MD, MSc,FRCPC FISH Study Toronto General Hospital Toronto, On ASDIN, Washington, DC Annual Scientific Meeting February 6, 3 Annual
The CARI Guidelines Caring for Australasians with Renal Impairment. Specific management of IgA nephropathy: role of tonsillectomy GUIDELINES
 Specific management of IgA nephropathy: role of tonsillectomy Date written: July 2005 Final submission: September 2005 Author: Merlin Thomas GUIDELINES No recommendation possible based on Level I or II
Specific management of IgA nephropathy: role of tonsillectomy Date written: July 2005 Final submission: September 2005 Author: Merlin Thomas GUIDELINES No recommendation possible based on Level I or II
Antihypertensive Trial Design ALLHAT
 1 U.S. Department of Health and Human Services Major Outcomes in High Risk Hypertensive Patients Randomized to Angiotensin-Converting Enzyme Inhibitor or Calcium Channel Blocker vs Diuretic National Institutes
1 U.S. Department of Health and Human Services Major Outcomes in High Risk Hypertensive Patients Randomized to Angiotensin-Converting Enzyme Inhibitor or Calcium Channel Blocker vs Diuretic National Institutes
1. Albuminuria an early sign of glomerular damage and renal disease. albuminuria
 1. Albuminuria an early sign of glomerular damage and renal disease albuminuria Cardio-renal continuum REGRESS Target organ damage Asymptomatic CKD New risk factors Atherosclerosis Target organ damage
1. Albuminuria an early sign of glomerular damage and renal disease albuminuria Cardio-renal continuum REGRESS Target organ damage Asymptomatic CKD New risk factors Atherosclerosis Target organ damage
Supplementary Appendix
 Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Serra AL, Poster D, Kistler AD, et al. Sirolimus and kidney
Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Serra AL, Poster D, Kistler AD, et al. Sirolimus and kidney
Individual Study Table Referring to Part of Dossier: Volume: Page:
 Synopsis Abbott Laboratories Name of Study Drug: Paricalcitol Capsules (ABT-358) (Zemplar ) Name of Active Ingredient: Paricalcitol Individual Study Table Referring to Part of Dossier: Volume: Page: (For
Synopsis Abbott Laboratories Name of Study Drug: Paricalcitol Capsules (ABT-358) (Zemplar ) Name of Active Ingredient: Paricalcitol Individual Study Table Referring to Part of Dossier: Volume: Page: (For
Predictors of the progression of renal disease in the Modification of Diet in Renal Disease Study
 Kidney International, Vol. 51 (/997), pp. 198 19/9 Predictors of the progression of renal disease in the Modification of Diet in Renal Disease Study MODIFICATION OF DIET IN RENAL DISEASE STUDY GROUP, prepared
Kidney International, Vol. 51 (/997), pp. 198 19/9 Predictors of the progression of renal disease in the Modification of Diet in Renal Disease Study MODIFICATION OF DIET IN RENAL DISEASE STUDY GROUP, prepared
NDT Advance Access published August 6, 2008
 NDT Advance Access published August 6, 2008 Nephrol Dial Transplant (2008) 1 5 doi: 10.1093/ndt/gfn454 Original Article Combined treatment with renin angiotensin system blockers and polyunsaturated fatty
NDT Advance Access published August 6, 2008 Nephrol Dial Transplant (2008) 1 5 doi: 10.1093/ndt/gfn454 Original Article Combined treatment with renin angiotensin system blockers and polyunsaturated fatty
C1q nephropathy the Diverse Disease
 C1q nephropathy the Diverse Disease Danica Galešić Ljubanović School of Medicine, University of Zagreb Dubrava University Hospital Zagreb, Croatia Definition Dominant or codominant ( 2+), mesangial staining
C1q nephropathy the Diverse Disease Danica Galešić Ljubanović School of Medicine, University of Zagreb Dubrava University Hospital Zagreb, Croatia Definition Dominant or codominant ( 2+), mesangial staining
SLOWING PROGRESSION OF KIDNEY DISEASE. Mark Rosenberg MD University of Minnesota
 SLOWING PROGRESSION OF KIDNEY DISEASE Mark Rosenberg MD University of Minnesota OUTLINE 1. Epidemiology of progression 2. Therapy to slow progression a. Blood Pressure control b. Renin-angiotensin-aldosterone
SLOWING PROGRESSION OF KIDNEY DISEASE Mark Rosenberg MD University of Minnesota OUTLINE 1. Epidemiology of progression 2. Therapy to slow progression a. Blood Pressure control b. Renin-angiotensin-aldosterone
BASELINE CHARACTERISTICS OF THE STUDY POPULATION
 COMPARISON OF TREATING METABOLIC ACIDOSIS IN CKD STAGE 4 HYPERTENSIVE KIDNEY DISEASE WITH FRUITS & VEGETABLES OR SODIUM BICARBONATE This was a 1-year, single-center, prospective, randomized, interventional
COMPARISON OF TREATING METABOLIC ACIDOSIS IN CKD STAGE 4 HYPERTENSIVE KIDNEY DISEASE WITH FRUITS & VEGETABLES OR SODIUM BICARBONATE This was a 1-year, single-center, prospective, randomized, interventional
Marshall Tulloch-Reid, MD, MPhil, DSc, FACE Epidemiology Research Unit Tropical Medicine Research Institute The University of the West Indies, Mona,
 Marshall Tulloch-Reid, MD, MPhil, DSc, FACE Epidemiology Research Unit Tropical Medicine Research Institute The University of the West Indies, Mona, Jamaica At the end of this presentation the participant
Marshall Tulloch-Reid, MD, MPhil, DSc, FACE Epidemiology Research Unit Tropical Medicine Research Institute The University of the West Indies, Mona, Jamaica At the end of this presentation the participant
CHAPTER 2. Primary Glomerulonephritis
 2nd Report of the PRIMARY GLOMERULONEPHRITIS CHAPTER 2 Primary Glomerulonephritis Sunita Bavanandan Lee Han Wei Lim Soo Kun 21 PRIMARY GLOMERULONEPHRITIS 2nd Report of the 2.1 Introduction This chapter
2nd Report of the PRIMARY GLOMERULONEPHRITIS CHAPTER 2 Primary Glomerulonephritis Sunita Bavanandan Lee Han Wei Lim Soo Kun 21 PRIMARY GLOMERULONEPHRITIS 2nd Report of the 2.1 Introduction This chapter
Trial to Reduce. Aranesp* Therapy. Cardiovascular Events with
 Trial to Reduce Cardiovascular Events with Aranesp* Therapy John J.V. McMurray, Hajime Uno, Petr Jarolim, Akshay S. Desai, Dick de Zeeuw, Kai-Uwe Eckardt, Peter Ivanovich, Andrew S. Levey, Eldrin F. Lewis,
Trial to Reduce Cardiovascular Events with Aranesp* Therapy John J.V. McMurray, Hajime Uno, Petr Jarolim, Akshay S. Desai, Dick de Zeeuw, Kai-Uwe Eckardt, Peter Ivanovich, Andrew S. Levey, Eldrin F. Lewis,
Long-term outcomes in nondiabetic chronic kidney disease
 original article http://www.kidney-international.org & 28 International Society of Nephrology Long-term outcomes in nondiabetic chronic kidney disease V Menon 1, X Wang 2, MJ Sarnak 1, LH Hunsicker 3,
original article http://www.kidney-international.org & 28 International Society of Nephrology Long-term outcomes in nondiabetic chronic kidney disease V Menon 1, X Wang 2, MJ Sarnak 1, LH Hunsicker 3,
HYPERTENSION GUIDELINES WHERE ARE WE IN 2014
 HYPERTENSION GUIDELINES WHERE ARE WE IN 2014 Donald J. DiPette MD FACP Special Assistant to the Provost for Health Affairs Distinguished Health Sciences Professor University of South Carolina University
HYPERTENSION GUIDELINES WHERE ARE WE IN 2014 Donald J. DiPette MD FACP Special Assistant to the Provost for Health Affairs Distinguished Health Sciences Professor University of South Carolina University
The Seventh Report of the Joint National Commission
 The Effect of a Lower Target Blood Pressure on the Progression of Kidney Disease: Long-Term Follow-up of the Modification of Diet in Renal Disease Study Mark J. Sarnak, MD; Tom Greene, PhD; Xuelei Wang,
The Effect of a Lower Target Blood Pressure on the Progression of Kidney Disease: Long-Term Follow-up of the Modification of Diet in Renal Disease Study Mark J. Sarnak, MD; Tom Greene, PhD; Xuelei Wang,
Table 1 Baseline characteristics of 60 hemodialysis patients with atrial fibrillation and warfarin use
 Table 1 Baseline characteristics of 60 hemodialysis patients with atrial fibrillation and warfarin use Baseline characteristics Users (n = 28) Non-users (n = 32) P value Age (years) 67.8 (9.4) 68.4 (8.5)
Table 1 Baseline characteristics of 60 hemodialysis patients with atrial fibrillation and warfarin use Baseline characteristics Users (n = 28) Non-users (n = 32) P value Age (years) 67.8 (9.4) 68.4 (8.5)
Nephrology Grand Rounds. Mansi Mehta November 24, 2015
 Nephrology Grand Rounds Mansi Mehta November 24, 2015 Case 51yo F with PMH significant for Hypertension referred to renal clinic for evaluation of elevated Cr. no known history of CKD; baseline creatinine
Nephrology Grand Rounds Mansi Mehta November 24, 2015 Case 51yo F with PMH significant for Hypertension referred to renal clinic for evaluation of elevated Cr. no known history of CKD; baseline creatinine
Additional file 2: Details of cohort studies and randomised trials
 Reference Randomised trials Ye et al. 2001 Abstract 274 R=1 WD=0 Design, numbers, treatments, duration Randomised open comparison of: (45 patients) 1.5 g for 3, 1 g for 3, then 0.5 to 0.75 g IV cyclophosphamide
Reference Randomised trials Ye et al. 2001 Abstract 274 R=1 WD=0 Design, numbers, treatments, duration Randomised open comparison of: (45 patients) 1.5 g for 3, 1 g for 3, then 0.5 to 0.75 g IV cyclophosphamide
MARINE Study Results
 TM MARINE Study Results Nasdaq: AMRN www.amarincorp.com 1 Forward-Looking Statement This presentation contains forward-looking statements, including those relating to the Company s product development
TM MARINE Study Results Nasdaq: AMRN www.amarincorp.com 1 Forward-Looking Statement This presentation contains forward-looking statements, including those relating to the Company s product development
Renal Protection Staying on Target
 Update Staying on Target James Barton, MD, FRCPC As presented at the University of Saskatchewan's Management of Diabetes & Its Complications (May 2004) Gwen s case Gwen, 49, asks you to take on her primary
Update Staying on Target James Barton, MD, FRCPC As presented at the University of Saskatchewan's Management of Diabetes & Its Complications (May 2004) Gwen s case Gwen, 49, asks you to take on her primary
Chapter 4: Steroid-resistant nephrotic syndrome in children Kidney International Supplements (2012) 2, ; doi: /kisup.2012.
 http://www.kidney-international.org & 2012 KDIGO Chapter 4: Steroid-resistant nephrotic syndrome in children Kidney International Supplements (2012) 2, 172 176; doi:10.1038/kisup.2012.17 INTRODUCTION This
http://www.kidney-international.org & 2012 KDIGO Chapter 4: Steroid-resistant nephrotic syndrome in children Kidney International Supplements (2012) 2, 172 176; doi:10.1038/kisup.2012.17 INTRODUCTION This
Lab Values Explained. working at full strength. Other possible causes of an elevated BUN include dehydration and heart failure.
 Patient Education Lab Values Explained Common Tests to Help Diagnose Kidney Disease Lab work, urine samples and other tests may be given as you undergo diagnosis and treatment for renal failure. The test
Patient Education Lab Values Explained Common Tests to Help Diagnose Kidney Disease Lab work, urine samples and other tests may be given as you undergo diagnosis and treatment for renal failure. The test
Out of date SUGGESTIONS FOR CLINICAL CARE (Suggestions are based on level III and IV evidence)
 Membranous nephropathy role of cyclosporine therapy Date written: July 2005 Final submission: September 2005 Author: Merlin Thomas GUIDELINES a. The use of cyclosporine therapy alone to prevent progressive
Membranous nephropathy role of cyclosporine therapy Date written: July 2005 Final submission: September 2005 Author: Merlin Thomas GUIDELINES a. The use of cyclosporine therapy alone to prevent progressive
Special Challenges and Co-Morbidities
 Special Challenges and Co-Morbidities Renal Disease/ Hypertension/ Diabetes in African-Americans M. Keith Rawlings, MD Medical Director Peabody Health Center AIDS Arms, Inc Dallas, TX Chair, Internal Medicine
Special Challenges and Co-Morbidities Renal Disease/ Hypertension/ Diabetes in African-Americans M. Keith Rawlings, MD Medical Director Peabody Health Center AIDS Arms, Inc Dallas, TX Chair, Internal Medicine
1. INSTRUCTIONS 2. DEFINITION OF HUS
 CQ_IBK_aHUS_01 / version 25/11/09 European Paediatric Research Group for HUS and related disorders Case questionnaire for diarrhoea negative/vtec (STEC) negative cases acute phase 1. INSTRUCTIONS Please
CQ_IBK_aHUS_01 / version 25/11/09 European Paediatric Research Group for HUS and related disorders Case questionnaire for diarrhoea negative/vtec (STEC) negative cases acute phase 1. INSTRUCTIONS Please
Glomerular tip adhesions predict the progression of IgA nephropathy
 Maeda et al. BMC Nephrology 2013, 14:272 RESEARCH ARTICLE Open Access Glomerular tip adhesions predict the progression of IgA nephropathy Kunihiro Maeda 1, Shogo Kikuchi 2, Naoto Miura 1, Keisuke Suzuki
Maeda et al. BMC Nephrology 2013, 14:272 RESEARCH ARTICLE Open Access Glomerular tip adhesions predict the progression of IgA nephropathy Kunihiro Maeda 1, Shogo Kikuchi 2, Naoto Miura 1, Keisuke Suzuki
Validation of the Oxford Classification of IgA Nephropathy: A Single-Center Study in Korean Adults
 original article korean j intern med 202;27:293-300 pissn 226-3303 eissn 2005-6648 Validation of the Oxford Classification of IgA Nephropathy: A Single-Center Study in Korean Adults Hoyoung Lee, Sul Hee
original article korean j intern med 202;27:293-300 pissn 226-3303 eissn 2005-6648 Validation of the Oxford Classification of IgA Nephropathy: A Single-Center Study in Korean Adults Hoyoung Lee, Sul Hee
Case Presentation Turki Al-Hussain, MD
 Case Presentation Turki Al-Hussain, MD Director, Renal Pathology Chapter Saudi Society of Nephrology & Transplantation Consultant Nephropathologist & Urological Pathologist Department of Pathology & Laboratory
Case Presentation Turki Al-Hussain, MD Director, Renal Pathology Chapter Saudi Society of Nephrology & Transplantation Consultant Nephropathologist & Urological Pathologist Department of Pathology & Laboratory
Predicting Renal Outcome in IgA Nephropathy
 Predicting Renal Outcome in IgA Nephropathy M. GENE RADFORD, JR.,* JAMES V. DONADIO, JR.,* ERIK J. BERGSTRALH,t and JOSEPH P. GRANDEt *Division of Nephrology, tthe Section of Biostatistics, and the Department
Predicting Renal Outcome in IgA Nephropathy M. GENE RADFORD, JR.,* JAMES V. DONADIO, JR.,* ERIK J. BERGSTRALH,t and JOSEPH P. GRANDEt *Division of Nephrology, tthe Section of Biostatistics, and the Department
Secondary IgA Nephropathy & HSP
 Secondary IgA Nephropathy & HSP Anjali Gupta, MD 1/11/11 AKI sec to Hematuria? 65 cases of ARF after an episode of macroscopic hematuria have been reported in the literature in patients with GN. The main
Secondary IgA Nephropathy & HSP Anjali Gupta, MD 1/11/11 AKI sec to Hematuria? 65 cases of ARF after an episode of macroscopic hematuria have been reported in the literature in patients with GN. The main
The nephrotic syndrome defined as urinary protein
 Original Article Comparative Study of Angiotensin Converting Enzyme Inhibitor and Calcium Channel Blocker in the Treatment of Steroid-Resistant Idiopathic Nephrotic Syndrome NS Kumar*, AK Singh*, RN Mishra**,
Original Article Comparative Study of Angiotensin Converting Enzyme Inhibitor and Calcium Channel Blocker in the Treatment of Steroid-Resistant Idiopathic Nephrotic Syndrome NS Kumar*, AK Singh*, RN Mishra**,
The CARI Guidelines Caring for Australians with Renal Impairment. Specific effects of calcium channel blockers in diabetic nephropathy GUIDELINES
 Specific effects of calcium channel blockers in diabetic nephropathy Date written: September 2004 Final submission: September 2005 Author: Kathy Nicholls GUIDELINES a. Non-dihydropyridine calcium channel
Specific effects of calcium channel blockers in diabetic nephropathy Date written: September 2004 Final submission: September 2005 Author: Kathy Nicholls GUIDELINES a. Non-dihydropyridine calcium channel
CKD FOR INTERNISTS. Dr Ahmed Hossain Associate professor Medicine Sir Salimullah Medical College
 CKD FOR INTERNISTS Dr Ahmed Hossain Associate professor Medicine Sir Salimullah Medical College INTRODUCTION In 2002, the National Kidney Foundation s Kidney Disease Outcomes Quality Initiative(KDOQI)
CKD FOR INTERNISTS Dr Ahmed Hossain Associate professor Medicine Sir Salimullah Medical College INTRODUCTION In 2002, the National Kidney Foundation s Kidney Disease Outcomes Quality Initiative(KDOQI)
The CARI Guidelines Caring for Australasians with Renal Impairment. Blood Pressure Control role of specific antihypertensives
 Blood Pressure Control role of specific antihypertensives Date written: May 2005 Final submission: October 2005 Author: Adrian Gillian GUIDELINES a. Regimens that include angiotensin-converting enzyme
Blood Pressure Control role of specific antihypertensives Date written: May 2005 Final submission: October 2005 Author: Adrian Gillian GUIDELINES a. Regimens that include angiotensin-converting enzyme
Launch Meeting 3 rd April 2014, Lucas House, Birmingham
 Angiotensin Converting Enzyme inhibitor (ACEi) / Angiotensin Receptor Blocker (ARB) To STOP OR Not in Advanced Renal Disease Launch Meeting 3 rd April 2014, Lucas House, Birmingham Prof Sunil Bhandari
Angiotensin Converting Enzyme inhibitor (ACEi) / Angiotensin Receptor Blocker (ARB) To STOP OR Not in Advanced Renal Disease Launch Meeting 3 rd April 2014, Lucas House, Birmingham Prof Sunil Bhandari
Individual Study Table Referring to Part of the Dossier. Use only) Name of Finished Product:
 SYNOPSIS Fresenius Title of the study: A double-blind, randomized study comparing the safety and torelance of SMOFlipid 20% and Intralipid 20% in long-term treatment with parenteral nutrition Coordinating
SYNOPSIS Fresenius Title of the study: A double-blind, randomized study comparing the safety and torelance of SMOFlipid 20% and Intralipid 20% in long-term treatment with parenteral nutrition Coordinating
Predicting and changing the future for people with CKD
 Predicting and changing the future for people with CKD I. David Weiner, M.D. Co-holder, C. Craig and Audrae Tisher Chair in Nephrology Professor of Medicine and Physiology and Functional Genomics University
Predicting and changing the future for people with CKD I. David Weiner, M.D. Co-holder, C. Craig and Audrae Tisher Chair in Nephrology Professor of Medicine and Physiology and Functional Genomics University
VA/DoD Clinical Practice Guideline for the Management of Chronic Kidney Disease in Primary Care (2008) PROVIDER REFERENCE CARDS Chronic Kidney Disease
 VA/DoD Clinical Practice Guideline for the Management of Chronic Kidney Disease in Primary Care (2008) PROVIDER REFERECE CARDS Chronic Kidney Disease CKD VA/DoD Clinical Practice Guideline for the Management
VA/DoD Clinical Practice Guideline for the Management of Chronic Kidney Disease in Primary Care (2008) PROVIDER REFERECE CARDS Chronic Kidney Disease CKD VA/DoD Clinical Practice Guideline for the Management
Tread Carefully Because you Tread on my Nephrons. Prescribing Hints in Renal Disease
 Tread Carefully Because you Tread on my Nephrons Prescribing Hints in Renal Disease David WP Lappin,, MB PhD FRCPI Clinical Lecturer in Medicine and Consultant Nephrologist and General Physician, Merlin
Tread Carefully Because you Tread on my Nephrons Prescribing Hints in Renal Disease David WP Lappin,, MB PhD FRCPI Clinical Lecturer in Medicine and Consultant Nephrologist and General Physician, Merlin
Corticosteroid Effectiveness in IgA Nephropathy: Long-Term Results of a Randomized, Controlled Trial
 J Am Soc Nephrol 15: 157 163, 2004 Corticosteroid Effectiveness in IgA Nephropathy: Long-Term Results of a Randomized, Controlled Trial CLAUDIO POZZI,* SIMEONE ANDRULLI,* LUCIA DEL VECCHIO,* PATRIZIA MELIS,
J Am Soc Nephrol 15: 157 163, 2004 Corticosteroid Effectiveness in IgA Nephropathy: Long-Term Results of a Randomized, Controlled Trial CLAUDIO POZZI,* SIMEONE ANDRULLI,* LUCIA DEL VECCHIO,* PATRIZIA MELIS,
Clinical Trial Synopsis TL-OPI-525, NCT#
 Clinical Trial Synopsis, NCT#00762736 Title of Study: A Phase II, Double-Blind, Randomized, Placebo-Controlled, Proof-of-Concept Study of the Efficacy, Safety, and Tolerability of Pioglitazone HCl (ACTOS
Clinical Trial Synopsis, NCT#00762736 Title of Study: A Phase II, Double-Blind, Randomized, Placebo-Controlled, Proof-of-Concept Study of the Efficacy, Safety, and Tolerability of Pioglitazone HCl (ACTOS
PRIMARY PHASE 2 ANALYSES FROM CARDINAL: A PHASE 2/3 STUDY OF BARDOXOLONE METHYL IN PATIENTS WITH ALPORT SYNDROME
 PRIMARY PHASE 2 ANALYSES FROM CARDINAL: A PHASE 2/3 STUDY OF BARDOXOLONE METHYL IN PATIENTS WITH ALPORT SYNDROME 217 American Society of Nephrology Meeting Geoffrey A. Block 1, Pablo E. Pergola 2, Lesley
PRIMARY PHASE 2 ANALYSES FROM CARDINAL: A PHASE 2/3 STUDY OF BARDOXOLONE METHYL IN PATIENTS WITH ALPORT SYNDROME 217 American Society of Nephrology Meeting Geoffrey A. Block 1, Pablo E. Pergola 2, Lesley
Omega-3 Fatty Acids. Alison L. Bailey MD, FACC Erlanger Heart and Lung Institute/University of Tennessee COM
 Omega-3 Fatty Acids Alison L. Bailey MD, FACC Erlanger Heart and Lung Institute/University of Tennessee COM Chattanooga @a_l_bailey Disclosures None Objectives Describe omega-3 fatty acids Review the historic
Omega-3 Fatty Acids Alison L. Bailey MD, FACC Erlanger Heart and Lung Institute/University of Tennessee COM Chattanooga @a_l_bailey Disclosures None Objectives Describe omega-3 fatty acids Review the historic
BASELINE CHARACTERISTICS OF THE STUDY POPULATION
 Study Summary DAILY ORAL SODIUM BICARBONATE PRESERVES GLOMERULAR FILTRATION RATE BY SLOWING ITS DECLINE IN EARLY HYPERTENSIVE NEPHROPATHY This was a 5-year, single-center, prospective, randomized, placebo-controlled,
Study Summary DAILY ORAL SODIUM BICARBONATE PRESERVES GLOMERULAR FILTRATION RATE BY SLOWING ITS DECLINE IN EARLY HYPERTENSIVE NEPHROPATHY This was a 5-year, single-center, prospective, randomized, placebo-controlled,
Study No.: Title: Rationale: Phase: Study Period: Study Design: Centres: Indication: Treatment: Objectives: Primary Outcome/Efficacy Variable:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Stages of Chronic Kidney Disease (CKD)
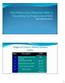 Early Treatment is the Key Stages of Chronic Kidney Disease (CKD) Stage Description GFR (ml/min/1.73 m 2 ) >90 1 Kidney damage with normal or GFR 2 Mild decrease in GFR 60-89 3 Moderate decrease in GFR
Early Treatment is the Key Stages of Chronic Kidney Disease (CKD) Stage Description GFR (ml/min/1.73 m 2 ) >90 1 Kidney damage with normal or GFR 2 Mild decrease in GFR 60-89 3 Moderate decrease in GFR
Proceedings of the 34th World Small Animal Veterinary Congress WSAVA 2009
 www.ivis.org Proceedings of the 34th World Small Animal Veterinary Congress WSAVA 2009 São Paulo, Brazil - 2009 Next WSAVA Congress : Reprinted in IVIS with the permission of the Congress Organizers PROTEINURIA
www.ivis.org Proceedings of the 34th World Small Animal Veterinary Congress WSAVA 2009 São Paulo, Brazil - 2009 Next WSAVA Congress : Reprinted in IVIS with the permission of the Congress Organizers PROTEINURIA
Efficacy and Safety of Prescription Omega-3-Acid Esters (P-OM3) for the Prevention of Symptomatic Atrial Fibrillation
 Efficacy and Safety of Prescription Omega-3-Acid Esters (P-OM3) for the Prevention of Symptomatic Atrial Fibrillation PR Kowey, JA Reiffel, KA Ellenbogen, GV Naccarelli, and CM Pratt for the OM-8 Clinical
Efficacy and Safety of Prescription Omega-3-Acid Esters (P-OM3) for the Prevention of Symptomatic Atrial Fibrillation PR Kowey, JA Reiffel, KA Ellenbogen, GV Naccarelli, and CM Pratt for the OM-8 Clinical
6/10/2014. Chronic Kidney Disease - General management and standard of care. Management of CKD according to stage (KDOQI 2002)
 Chronic Kidney Disease - General management and standard of care Dr Nathalie Demoulin, Prof Michel Jadoul Cliniques universitaires Saint-Luc Université Catholique de Louvain What should and can be done
Chronic Kidney Disease - General management and standard of care Dr Nathalie Demoulin, Prof Michel Jadoul Cliniques universitaires Saint-Luc Université Catholique de Louvain What should and can be done
Hot Topics in Diabetic Kidney Disease a primary care perspective
 Hot Topics in Diabetic Kidney Disease a primary care perspective DR SARAH DAVIES GP PARTNER WITH SPECIAL INTEREST IN DIABETES, CARDIFF DUK CLINICAL CHAMPION NB MEDICAL HOT TOPICS PRESENTER AND DIABETES
Hot Topics in Diabetic Kidney Disease a primary care perspective DR SARAH DAVIES GP PARTNER WITH SPECIAL INTEREST IN DIABETES, CARDIFF DUK CLINICAL CHAMPION NB MEDICAL HOT TOPICS PRESENTER AND DIABETES
Fructose, Uric Acid and Hypertension in Children and Adolescents
 Fructose, Uric Acid and Hypertension in Children and Adolescents Daniel I. Feig, MD, PhD, MS Director, Division of Nephrology Department of Pediatrics University of Alabama, Birmingham Topics for Discussion
Fructose, Uric Acid and Hypertension in Children and Adolescents Daniel I. Feig, MD, PhD, MS Director, Division of Nephrology Department of Pediatrics University of Alabama, Birmingham Topics for Discussion
Random forest can accurately predict the development of end-stage renal disease in immunoglobulin a nephropathy patients
 Original Article Page 1 of 8 Random forest can accurately predict the development of end-stage renal disease in immunoglobulin a nephropathy patients Xin Han 1#, Xiaonan Zheng 2#, Ying Wang 3, Xiaoru Sun
Original Article Page 1 of 8 Random forest can accurately predict the development of end-stage renal disease in immunoglobulin a nephropathy patients Xin Han 1#, Xiaonan Zheng 2#, Ying Wang 3, Xiaoru Sun
A: Epidemiology update. Evidence that LDL-C and CRP identify different high-risk groups
 A: Epidemiology update Evidence that LDL-C and CRP identify different high-risk groups Women (n = 27,939; mean age 54.7 years) who were free of symptomatic cardiovascular (CV) disease at baseline were
A: Epidemiology update Evidence that LDL-C and CRP identify different high-risk groups Women (n = 27,939; mean age 54.7 years) who were free of symptomatic cardiovascular (CV) disease at baseline were
Clinical Practice Guideline
 Clinical Practice Guideline Secondary Prevention for Patients with Coronary and Other Vascular Disease Since the 2001 update of the American Heart Association (AHA)/American College of Cardiology (ACC)
Clinical Practice Guideline Secondary Prevention for Patients with Coronary and Other Vascular Disease Since the 2001 update of the American Heart Association (AHA)/American College of Cardiology (ACC)
(For National Authority Use Only) Name of Study Drug: to Part of Dossier:
 2.0 Synopsis Abbott Laboratories Individual Study Table Referring to Part of Dossier: (For National Authority Use Only) Name of Study Drug: Volume: ABT-335 Name of Active Ingredient: Page: ABT-335, A-7770335.115
2.0 Synopsis Abbott Laboratories Individual Study Table Referring to Part of Dossier: (For National Authority Use Only) Name of Study Drug: Volume: ABT-335 Name of Active Ingredient: Page: ABT-335, A-7770335.115
Chronic Kidney Disease. Basics of CKD Terms Diagnosis Management
 Chronic Kidney Disease Basics of CKD Terms Diagnosis Management Review the prevalence of chronic kidney disease (CKD) Review how CKD develops Review populations at risk for CKD Review CKD diagnosis Objectives
Chronic Kidney Disease Basics of CKD Terms Diagnosis Management Review the prevalence of chronic kidney disease (CKD) Review how CKD develops Review populations at risk for CKD Review CKD diagnosis Objectives
SUPPLEMENTAL MATERIAL
 SUPPLEMENTAL MATERIAL Randomized Controlled Trial of to Increase Serum Bicarbonate in Chronic Kidney Disease Patients David A. Bushinsky a, Thomas Hostetter b, Gerrit Klaerner c, Yuri Stasiv c, Claire
SUPPLEMENTAL MATERIAL Randomized Controlled Trial of to Increase Serum Bicarbonate in Chronic Kidney Disease Patients David A. Bushinsky a, Thomas Hostetter b, Gerrit Klaerner c, Yuri Stasiv c, Claire
2.0 Synopsis. Paricalcitol Capsules M Clinical Study Report R&D/15/0380. (For National Authority Use Only)
 2.0 Synopsis AbbVie Inc. Name of Study Drug: ABT-358/Zemplar (paricalcitol) Capsules Name of Active Ingredient: paricalcitol Individual Study Table Referring to Part of Dossier: Volume: Page: (For National
2.0 Synopsis AbbVie Inc. Name of Study Drug: ABT-358/Zemplar (paricalcitol) Capsules Name of Active Ingredient: paricalcitol Individual Study Table Referring to Part of Dossier: Volume: Page: (For National
The CARI Guidelines Caring for Australians with Renal Impairment. Membranous nephropathy Role of alkylating agents GUIDELINES
 Membranous nephropathy Role of alkylating agents Date written: July 2005 Final submission: September 2005 Author: Merlin Thomas GUIDELINES a. Treatment with alkylating agents is associated with an increased
Membranous nephropathy Role of alkylating agents Date written: July 2005 Final submission: September 2005 Author: Merlin Thomas GUIDELINES a. Treatment with alkylating agents is associated with an increased
IgA-Nephropathy: an update on treatment Jürgen Floege
 IgA-Nephropathy: an update on treatment Jürgen Floege Division of Nephrology & Immunology juergen.floege@rwth-aachen.de Floege & Feehally, Nat Rev Nephrol 2013 Floege & Eitner, J Am Soc Nephrol. 2011 If
IgA-Nephropathy: an update on treatment Jürgen Floege Division of Nephrology & Immunology juergen.floege@rwth-aachen.de Floege & Feehally, Nat Rev Nephrol 2013 Floege & Eitner, J Am Soc Nephrol. 2011 If
GALECTIN-3 PREDICTS LONG TERM CARDIOVASCULAR DEATH IN HIGH-RISK CORONARY ARTERY DISEASE PATIENTS
 GALECTIN-3 PREDICTS LONG TERM CARDIOVASCULAR DEATH IN HIGH-RISK CORONARY ARTERY DISEASE PATIENTS Table of Contents List of authors pag 2 Supplemental figure I pag 3 Supplemental figure II pag 4 Supplemental
GALECTIN-3 PREDICTS LONG TERM CARDIOVASCULAR DEATH IN HIGH-RISK CORONARY ARTERY DISEASE PATIENTS Table of Contents List of authors pag 2 Supplemental figure I pag 3 Supplemental figure II pag 4 Supplemental
Elevation of Serum Creatinine: When to Screen, When to Refer. Bruce F. Culleton, MD, FRCPC; and Jolanta Karpinski, MD, FRCPC
 Elevation of Serum Creatinine: When to Screen, When to Refer Bruce F. Culleton, MD, FRCPC; and Jolanta Karpinski, MD, FRCPC Presented at the University of Calgary s CME and Professional Development 2006-2007
Elevation of Serum Creatinine: When to Screen, When to Refer Bruce F. Culleton, MD, FRCPC; and Jolanta Karpinski, MD, FRCPC Presented at the University of Calgary s CME and Professional Development 2006-2007
FIBRILLARY GLOMERULONEPHRITIS DIAGNOSTIC CRITERIA, PITFALLS, AND DIFFERENTIAL DIAGNOSIS
 FIBRILLARY GLOMERULONEPHRITIS DIAGNOSTIC CRITERIA, PITFALLS, AND DIFFERENTIAL DIAGNOSIS Guillermo A. Herrera MD Louisiana State University, Shreveport Fibrils in bundles 10-20 nm d Diabetic fibrillosis
FIBRILLARY GLOMERULONEPHRITIS DIAGNOSTIC CRITERIA, PITFALLS, AND DIFFERENTIAL DIAGNOSIS Guillermo A. Herrera MD Louisiana State University, Shreveport Fibrils in bundles 10-20 nm d Diabetic fibrillosis
SUPPLEMENTARY DATA. Supplementary Table 1. Baseline Patient Characteristics
 Supplementary Table 1. Baseline Patient Characteristics Normally distributed data are presented as mean (±SD), data that were not of a normal distribution are presented as median (ICR). The baseline characteristics
Supplementary Table 1. Baseline Patient Characteristics Normally distributed data are presented as mean (±SD), data that were not of a normal distribution are presented as median (ICR). The baseline characteristics
Research Article Serum Galactose-Deficient IgA1 Level Is Not Associated with Proteinuria in Children with IgA Nephropathy
 International Nephrology Volume 212, Article ID 315467, 7 pages doi:1.1155/212/315467 Research Article Serum Galactose-Deficient IgA1 Level Is Not Associated with Proteinuria in Children with IgA Nephropathy
International Nephrology Volume 212, Article ID 315467, 7 pages doi:1.1155/212/315467 Research Article Serum Galactose-Deficient IgA1 Level Is Not Associated with Proteinuria in Children with IgA Nephropathy
HHS Public Access Author manuscript Am J Kidney Dis. Author manuscript; available in PMC 2017 July 05.
 HHS Public Access Author manuscript Published in final edited form as: Am J Kidney Dis. 2017 March ; 69(3): 482 484. doi:10.1053/j.ajkd.2016.10.021. Performance of the Chronic Kidney Disease Epidemiology
HHS Public Access Author manuscript Published in final edited form as: Am J Kidney Dis. 2017 March ; 69(3): 482 484. doi:10.1053/j.ajkd.2016.10.021. Performance of the Chronic Kidney Disease Epidemiology
Nephrotic syndrome minimal change disease vs. IgA nephropathy. Hadar Meringer Internal medicine B Sheba
 Nephrotic syndrome minimal change disease vs. IgA nephropathy Hadar Meringer Internal medicine B Sheba The Case 29 year old man diagnosed with nephrotic syndrome 2 weeks ago and complaining now about Lt.flank
Nephrotic syndrome minimal change disease vs. IgA nephropathy Hadar Meringer Internal medicine B Sheba The Case 29 year old man diagnosed with nephrotic syndrome 2 weeks ago and complaining now about Lt.flank
Mipomersen (ISIS ) Page 2 of 1979 Clinical Study Report ISIS CS3
 (ISIS 301012) Page 2 of 1979 2 SYNOPSIS ISIS 301012-CS3 synopsis Page 1 Title of Study: A Phase 2, Randomized, Double-Blind, Placebo-Controlled Study to Assess the Safety, Tolerability, Pharmacokinetics,
(ISIS 301012) Page 2 of 1979 2 SYNOPSIS ISIS 301012-CS3 synopsis Page 1 Title of Study: A Phase 2, Randomized, Double-Blind, Placebo-Controlled Study to Assess the Safety, Tolerability, Pharmacokinetics,
RENAAL, IRMA-2 and IDNT. Three featured trials linking a disease spectrum IDNT RENAAL. Death IRMA 2
 Treatment of Diabetic Nephropathy and Proteinuria Background End stage renal disease is a major cause of death and disability among diabetics BP reduction is important to slow the progression of diabetic
Treatment of Diabetic Nephropathy and Proteinuria Background End stage renal disease is a major cause of death and disability among diabetics BP reduction is important to slow the progression of diabetic
Inflammation in Renal Disease
 Inflammation in Renal Disease Donald G. Vidt, MD Inflammation is a component of the major modifiable risk factors in renal disease. Elevated high-sensitivity C-reactive protein (hs-crp) levels have been
Inflammation in Renal Disease Donald G. Vidt, MD Inflammation is a component of the major modifiable risk factors in renal disease. Elevated high-sensitivity C-reactive protein (hs-crp) levels have been
Clinical Pearls in Renal Medicine
 Clinical Pearls in Renal Medicine Joel A. Gordon MD Professor of Medicine Nephrology Division Staff Physician Kidney Disease and Blood Pressure Clinic Disclosures None of my financial holdings will have
Clinical Pearls in Renal Medicine Joel A. Gordon MD Professor of Medicine Nephrology Division Staff Physician Kidney Disease and Blood Pressure Clinic Disclosures None of my financial holdings will have
Am J Nephrol 2015;41: DOI: /
 American Journal of Nephrology Original Report: Patient-Oriented, Translational Research Received: September 1, 214 Accepted: March 2, 215 Published online: April 9, 215 Addition of egfr and Age Improves
American Journal of Nephrology Original Report: Patient-Oriented, Translational Research Received: September 1, 214 Accepted: March 2, 215 Published online: April 9, 215 Addition of egfr and Age Improves
SYNOPSIS. Issue Date: 04 February 2009 Document No.: EDMS -USRA
 SYNOPSIS Issue Date: 04 February 2009 Document No.: EDMS -USRA-10751204 Name of Sponsor/Company Name of Finished Product Name of Active Ingredient(s) Johnson & Johnson Pharmaceutical Research & Development,
SYNOPSIS Issue Date: 04 February 2009 Document No.: EDMS -USRA-10751204 Name of Sponsor/Company Name of Finished Product Name of Active Ingredient(s) Johnson & Johnson Pharmaceutical Research & Development,
Hypertension targets: sorting out the confusion. Brian Rayner, Division of Nephrology and Hypertension, University of Cape Town
 Hypertension targets: sorting out the confusion Brian Rayner, Division of Nephrology and Hypertension, University of Cape Town Historical Perspective The most famous casualty of this approach was the
Hypertension targets: sorting out the confusion Brian Rayner, Division of Nephrology and Hypertension, University of Cape Town Historical Perspective The most famous casualty of this approach was the
PEER REVIEW HISTORY ARTICLE DETAILS VERSION 1 - REVIEW. Kristine Hommel Department of nephrology, Herlev Hospital, Denmark 17-Nov-2015
 PEER REVIEW HISTORY BMJ Open publishes all reviews undertaken for accepted manuscripts. Reviewers are asked to complete a checklist review form (http://bmjopen.bmj.com/site/about/resources/checklist.pdf)
PEER REVIEW HISTORY BMJ Open publishes all reviews undertaken for accepted manuscripts. Reviewers are asked to complete a checklist review form (http://bmjopen.bmj.com/site/about/resources/checklist.pdf)
Classification of Glomerular Diseases and Defining Individual Glomerular Lesions: Developing International Consensus
 Classification of Glomerular Diseases and Defining Individual Glomerular Lesions: Developing International Consensus Mark Haas MD, PhD Department of Pathology & Laboratory Medicine Cedars-Sinai Medical
Classification of Glomerular Diseases and Defining Individual Glomerular Lesions: Developing International Consensus Mark Haas MD, PhD Department of Pathology & Laboratory Medicine Cedars-Sinai Medical
Primary Endpoint The primary endpoint is overall survival, measured as the time in weeks from randomization to date of death due to any cause.
 CASE STUDY Randomized, Double-Blind, Phase III Trial of NES-822 plus AMO-1002 vs. AMO-1002 alone as first-line therapy in patients with advanced pancreatic cancer This is a multicenter, randomized Phase
CASE STUDY Randomized, Double-Blind, Phase III Trial of NES-822 plus AMO-1002 vs. AMO-1002 alone as first-line therapy in patients with advanced pancreatic cancer This is a multicenter, randomized Phase
IgA Nephropathy: Morphologic Findings Associated with Disease Progression and Therapeutic Response A Working Group Approach
 I IgA Nephropathy: Morphologic Findings Associated with Disease Progression and Therapeutic Response A Working Group Approach Mark Haas Department of Pathology & Lab Medicine Cedars-Sinai Medical Center
I IgA Nephropathy: Morphologic Findings Associated with Disease Progression and Therapeutic Response A Working Group Approach Mark Haas Department of Pathology & Lab Medicine Cedars-Sinai Medical Center
Irish Practice Nurses Association Annual Conference Tullamore Court Hotel OCTOBER 6 th 2012
 Irish Practice Nurses Association Annual Conference Tullamore Court Hotel OCTOBER 6 th 2012 Susan McKenna Renal Clinical Nurse Specialist Cavan General Hospital Renal patient population ACUTE RENAL FAILURE
Irish Practice Nurses Association Annual Conference Tullamore Court Hotel OCTOBER 6 th 2012 Susan McKenna Renal Clinical Nurse Specialist Cavan General Hospital Renal patient population ACUTE RENAL FAILURE
Fabry Disease and the Kidneys
 Department of Human Genetics Division of Medical Genetics Lysosomal Storage Disease Center www.genetics.emory.edu Fabry Disease and the Kidneys What is Fabry Disease? Fabry disease (FD) is an X-linked
Department of Human Genetics Division of Medical Genetics Lysosomal Storage Disease Center www.genetics.emory.edu Fabry Disease and the Kidneys What is Fabry Disease? Fabry disease (FD) is an X-linked
Omega-3 Fatty Acids for Prevention of Post-Operative Atrial Fibrillation
 Omega-3 Fatty Acids for Prevention of Post-Operative Atrial Fibrillation Roberto Marchioli, MD, on behalf of the OPERA Investigators American Heart Association, Los Angeles November 5, 2012 Published online
Omega-3 Fatty Acids for Prevention of Post-Operative Atrial Fibrillation Roberto Marchioli, MD, on behalf of the OPERA Investigators American Heart Association, Los Angeles November 5, 2012 Published online
RENAL FAILURE IN CHILDREN Dr. Mai Mohamed Elhassan Assistant Professor Jazan University
 RENAL FAILURE IN CHILDREN Dr. Mai Mohamed Elhassan Assistant Professor Jazan University OBJECTIVES By the end of this lecture each student should be able to: Define acute & chronic kidney disease(ckd)
RENAL FAILURE IN CHILDREN Dr. Mai Mohamed Elhassan Assistant Professor Jazan University OBJECTIVES By the end of this lecture each student should be able to: Define acute & chronic kidney disease(ckd)
PFIZER INC. THERAPEUTIC AREA AND FDA APPROVED INDICATIONS: See USPI
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
The CARI Guidelines Caring for Australasians with Renal Impairment. Protein Restriction to prevent the progression of diabetic nephropathy GUIDELINES
 Protein Restriction to prevent the progression of diabetic nephropathy Date written: September 2004 Final submission: September 2005 Author: Kathy Nicholls GUIDELINES a. A small volume of evidence suggests
Protein Restriction to prevent the progression of diabetic nephropathy Date written: September 2004 Final submission: September 2005 Author: Kathy Nicholls GUIDELINES a. A small volume of evidence suggests
Jo Abraham MD Division of Nephrology University of Utah
 Jo Abraham MD Division of Nephrology University of Utah 1. To highlight the link between AKI and progression to CKD 2. To discuss the newer agents that have come available for the treatment of hyperkalemia
Jo Abraham MD Division of Nephrology University of Utah 1. To highlight the link between AKI and progression to CKD 2. To discuss the newer agents that have come available for the treatment of hyperkalemia
