SUMMARY OF PRODUCT CHARACTERISTICS
|
|
|
- Joella Harrington
- 5 years ago
- Views:
Transcription
1 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT ACURMIL 25 mg/2.5 ml solution for intravenous injection ACURMIL 50 mg/5 ml solution for intravenous injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION ACURMIL 25 mg/2.5 ml solution for injection a 2.5 ml ampoule contains: Active principle: atracurium besylate: 25 mg ACURMIL 50 mg/5 ml solution for injection a 5 ml ampoule contains: Active principle: atracurium besylate: 50 mg For the full list of excipients reference is made to section PHARMACEUTICAL FORM Solution for intravenous injection containing 10 mg/ml of atracurium besylate 4. CLINICAL PARTICULARS 4.1. Therapeutic Indications M03AC04 ACURMIL is a highly-selective nondepolarizing neuromuscular blocker used in general anesthesia to facilitate endotracheal intubation and to obtain muscle relaxation in a wide range of surgical procedures and in controlled ventilation. It is also indicated to facilitate controlled ventilation in Intensive Care Unit patients. In addition, ACURMIL is indicated for the maintenance of muscle relaxation during cesarean sections Dosage and Administration ACURMIL should be administered intravenously. To avoid a temporary pressure drop, as may occur following a bolus injection, ACURMIL should be administered by slow injection. Adults - Injection Adult doses range from 0.3 to 0.6 mg/kg according to the required duration of the complete block, determining a state of suitable relaxation for times varying from 15 to 35 minutes. Additional doses of mg/kg, as required, may be administered to prolong the complete block. Administration of successive additional doses does not induce any cumulative effects. As a rule, endotracheal intubation may be carried out within 90 seconds of the intravenous injection of mg/kg of the drug. The neuromuscular block induced by ACURMIL may be rapidly and permanently reversed with standard doses of such anticholinesterase agents as neostigmine, in conjunction with an anticholinergic agent such as atropine. Recovery from a complete block occurs, without neostigmine, within approximately 35 minutes calculated from the first recovery of muscle twitch to 95% recovery of the normal neuromuscular function.
2 - Use by intravenous infusion After administration of an initial dose of mg/kg i.v., ACURMIL may be administered by continuous intravenous administration at the dose of mg/kg/min. in order to maintain the muscular block during extended surgical procedures. Accurate dosing is best achieved by using an infusion pump. ACURMIL may be administered by continuous infusion at the above-mentioned rates to patients undergoing cardiopulmonary bypass. The induced hypothermia, with body temperatures of 25 C or 26 C, reduces the inactivation speed of Atracurium and therefore, at such temperatures, complete neuromuscular block can be maintained by reducing the initial infusion rate to one half. ACURMIL is compatible with the following infusion solutions, for the following stability periods: Infusion solutions Physiologic solution 0.9% w/v Glucose 5% w/v Ringer Glucose with sodium chloride I (respectively 4.7% w/v and 0.18% w/v) Ringer lactate Stability periods 24 hours 8 hours 8 hours 8 hours 4 hours The above-mentioned solutions may be admixed with ACURMIL to obtain 0.5 to 0.9 mg/ml solutions. These ACURMIL infusion solutions remain stable, in daylight, at temperatures of up to 30 C. ACURMIL may also be diluted with water for injectable preparations until concentrations of mg/ml are achieved, though their use for continuous infusion is not recommended. Such dilutions remain stable for 8 hours at temperatures not exceeding 30 C. Pediatric patients The dosage used for children over 1 month old is the same as for adults, based on mg/kg of body weight. Neonates Use of ACURMIL in neonates is not recommended, as there are no sufficient data available. Elderly patients ACURMIL may be used at standard doses in elderly patients. Patients with kidney or liver insufficiency ACURMIL may be used at standard doses in patients with kidney or liver insufficiency. Patients with serious cardiovascular diseases The initial dose of ACURMIL should be given slowly over a period of 60 seconds to patients with significant cardiovascular diseases.
3 Intensive Care Unit (ICU) patients After an initial bolus dose of mg/kg i.v., ACURMIL may be given by continuous infusion at the dose of µg/kg/min ( mg/kg/hour) in order to maintain a state of neuromuscular block. Anyway, there may be wide interpatient variability in dosage requirements (from 4.5 µg/kg/min to 29.5 µg/kg/min). The available data indicate that such requirements may increase in the course of prolonged administration in ICU patients; especially in patients developing peripheral edemas. Recovery from neuromuscular block (TOF> 0.75) after an infusion with ACURMIL does not depend on the duration of administration. Spontaneous recovery occurs in about 60 minutes (range min.). 4.3 Contraindications Hypersensitivity to the active principle or to any of the excipients. Atracurium is contraindicated for patients with known hypersensitivity to atracurium, cisatracurium and benzenesulfonic acid Special Warnings and Precautions for Use ACURMIL, like all other neuromuscular blockers, causes the paralysis of the respiratory and skeletal muscles, but does not affect the state of consciousness. Therefore, it should be administered only with suitable general anesthesia and by an anesthetist or under his/her direct surveillance. Besides, suitable endotracheal and artificial ventilation equipment must be directly available. In predisposed patients the administration of ACURMIL may cause the release of histamine; for this reason special caution should be applied in the treatment of patients with significant cardiovascular diseases who may be more sensitive to the effects of temporary hypotension and of patients whose history (e.g. severe hypersensitivity reactions to multiple allergens and asthma) suggests a greater risk of histamine release. In particular, bronchospasm might occur in patients with a history of allergy and asthma. In these patients, ACURMIL should be administered by slow i.v. injection of fractionated doses. Besides, caution is recommended in administering atracurium to patients with a history of hypersensitivity to other neuromuscular blocking agents, as a high rate of cross allergy (over 50%) was observed between neuromuscular blocking agents (see section 4.3). ACURMIL should be administered with caution to patients with myasthenia gravis, other neuromuscular diseases, carcinomatosis, or significant electrolyte disorders, because, as with other non-depolarizing agents, greater sensitivity to atracurium is to be expected in patients with myasthenia gravis, other neuromuscular diseases and severe electrolyte disorders. Atracurium should be administered over a period of 60 seconds to patients potentially prone to a drop in arterial pressure, such as hypovolemic patients. In the recommended dosage range, atracurium does not present any significant vagal or ganglionic blocking properties. Consequently, atracurium has no clinically relevant effects on heart rate in the recommended dosage range. Bradycardia induced by other anesthetics or by vagal stimulation during surgery will not be counteracted by atracurium.
4 If the injection site of choice is a small vein, all residues of ACURMIL must be washed away with a small quantity of physiologic solution. If other anesthetics are to be administered through the same needle or cannula used for administration of ACURMIL, it is important that each drug is removed by introducing some sterile pyrogen-free water or physiologic solution into the needle or cannula inbetween administrations. When given to laboratory animals, laudanosine, one of atracurium metabolites, may cause excitatory effects on the CNS. While cases of seizures have been reported in ICU patients to whom ACURMIL was administered, these were not ascribed to laudanosine or atracurium, also in those cases in which ACURMIL had been administered as continuous infusion over longer periods. Atracurium is inactivated by high ph and therefore must not be admixed in the same syringe with thiopentone or any alkaline agents. Atracurium is hypotonic and must not be administered via the infusion system of a blood transfusion. Studies in malignant hyperthermia in susceptible animals (swine) and clinical studies in patients susceptible to malignant hyperthermia indicate that atracurium does not trigger this syndrome. In common with other non-depolarizing neuromuscular blocking agents, resistance may develop in patients suffering from burns. Such patients may require increased doses dependent on the time elapsed since the burn injury and the extent of the burn. ICU patients. When given to laboratory animals at high doses, laudanosine, one of atracurium metabolites, may cause temporary hypotension and, in some species, excitatory effects on the brain. While cases of seizures have been reported in ICU patients to whom atracurium was administered, it was not possible to establish a cause-effect relationship with laudanosine (see section 4.8) Interaction with other medicinal products and other forms of interaction Drugs that, if contextually administered, may enhance the neuromuscular blocking action of ACURMIL include such inhalation anesthetics as halothane, enflurane and isoflurane. As with other non-depolarizing neuromuscular blocking agents, the extent and/or duration of a non-depolarizing muscular block may be enhanced by the interaction with: - antibiotics including the aminoglycosides, polymyxins, spectinomycin, tetracyclines, lincomycin, and clindamycin; - antiarrhythmic medicinal products: propranolol, calcium-antagonists, lignocaine, procainamide and quinidine; - diuretics: furosemide and possibly mannitol, thiazide diuretics and acetazolamide; - magnesium sulfate; - ketamine; - lithium salts; - ganglion blocking agents: trimethaphan, hexamethonium. Seldom, certain medicinal products may aggravate or unmask latent myasthenia gravis or actually induce a myasthenic syndrome; increased sensitivity to atracurium besylate would follow. Such medicinal products include various antibiotics, beta-blockers (propranolol, oxprenolol), antiarrhythmics (procainamide, quinidine), antirheumatics
5 (chloroquine, D-penicillamine), trimethaphan, chlorpromazine, steroids, phenytoin, lithium. The onset of non-depolarizing neuromuscular block is likely to be lengthened and the duration of block shortened in patients receiving chronic anticonvulsant therapy. The administration of combinations of non-depolarizing neuromuscular blocking agents in conjunction with atracurium may produce a degree of neuromuscular blockade in excess of that which might be expected, were an equipotent total dose of atracurium administered. Any synergistic effect may vary between different medicinal product combinations. Depolarizing muscle relaxants such as suxamethonium chloride should not be administered to prolong the neuromuscular blocking effects of non-depolarizing blocking agents such as atracurium, as this may result in a prolonged and complex block, which can be difficult to reverse with anticholinesterase medicinal products. Anticholinesterase agents, such as donepezil, commonly used for the treatment of Alzheimer disease, may shorten the duration and reduce the extent of atracurium induced neuromuscular block Fertility, pregnancy and lactation Fertility No fertility studies have been conducted with atracurium besylate. Pregnancy Although animal studies indicate that ACURMIL has no negative effects on fetal development, however, as in the case of all neuromuscular blockers, ACURMIL should be used in pregnancy only if the potential benefit for the mother outweighs any potential risks for the fetus. ACURMIL may be used to maintain the neuromuscular block during a cesarean section, as within the recommended dose range it does not cross the placental barrier in clinically significant quantities. However, the possibility of respiratory depression in the newborn should be considered. Lactation It is not known whether ACURMIL is excreted in human milk Effects on the Ability to Drive and Operate Machines This precaution is not relevant for use of atracurium. As atracurium is always used in conjunction with a general anesthetic, the normal precautions concerning the performance of activities after general anesthesia apply Undesired effects ACURMIL exerts no appreciable vagal or ganglionic block effects. Therefore, the vagomimetic effects of the anesthetics used may be potentiated. As with most neuromuscular blockers, also with ACURMIL there is the possibility of histamine release in susceptible patients.
6 The adverse reactions most frequently observed during treatment are: hypotension (mild, transient) and flushing, both related to histamine release. More rarely serious anaphylactoid or anaphylactic reactions have been reported in patients who had received atracurium together with one or more anesthetics. Adverse reactions are classified by systems, organs and frequency. Frequency is defined as: very common > 1/10, common > 1/100 and < 1/10, uncommon > 1/1000 and < 1/100, rare > 1/ and < 1/1000, very rare < 1/ Very common, common and uncommon frequencies have been determined based on the data of clinical trials. Rare and very rare frequencies have been generally estimated based on spontaneous reports. Frequency defined as not known applied to those reactions that could not be estimated from the available data. Clinical trial data Vascular disorders Common: hypotension (mild, transient) #, flushing # Respiratory, thoracic and mediastinal disorders Uncommon: bronchospasm # Postmarketing data Immune system disorders Very rare: anaphylactic reaction, anaphylactoid reaction, including shock, circulatory failure and cardiac arrest. Very rarely serious anaphylactoid or anaphylactic reactions have been reported in patients who had received atracurium together with one or more anesthetics. Nervous system disorders Unknown: seizures There have been reports of seizures in patients in ICUs who had been receiving atracurium besylate simultaneously with other drugs. These patients generally had one or more medical conditions that made them susceptible to seizures (such as brain injury, cerebral edema, viral encephalitis, hypoxic encephalopathy, and uremia). It was impossible to establish a causative correlation with laudanosine. There appears to be no correlation between plasma laudanosine concentration and appearance of seizures in clinical trials. Skin and subcutaneous tissue disorders Rare: urticaria Musculoskeletal and connective tissue disorders Not known: myopathy, muscular weakness. After prolonged use of myorelaxants in severely ill ICU patients, myasthenia and/or myopathy have been observed. The majority of these patients received concomitant corticosteroids. Causal connection with atracurium besylate therapy is not established, as these events have been rarely observed with atracurium. # The events ascribed to histamine release have been marked with a # 4.9. Overdosage In case of overdosage or delayed recovery, patients must be given atropine and neostigmine, and should be artificially ventilated until spontaneous breathing is resumed. Signs and symptoms Prolonged muscle paralysis and its consequences are the main signs of overdose.
7 Treatment It is essential to maintain a patent airway together with assisted positive pressure ventilation until adequate spontaneous respiration reappears. Full sedation will be required if consciousness is not impaired. Recovery may be accelerated by the administration of anticholinesterase agents accompanied by atropine or glycopyrrolate, once evidence of spontaneous recovery is present. 5. PHARMACOLOGICAL PROPERTIES 5.1. Pharmacodynamic Properties Pharmacotherapeutic category: peripherally acting muscle relaxants ATC code: M03AC04. ACURMIL, atracurium besylate, acts on the neuromuscular junction competitively antagonizing the action of acetylcholine on the cholinergic receptors of the postsynaptic membrane, thus causing neuromuscular block. It has no effect on intraocular pressure, which makes it suitable for use in ophthalmic surgery. Pediatric population: Limited literature data on neonates suggest a variability in the onset time and duration of atracurium activity in this population as compared to older children (see section 4.2) 5.2. Pharmacokinetic Properties ACURMIL degradation to inactive metabolites is mainly carried out by a spontaneous mechanism of nonenzymatic decomposition called Hofmann elimination which takes place at physiological ph and temperature values. Reversal of ACURMIL-induced neuromuscular block is independent of its metabolism and excretion by liver or kidneys. Its action duration is probably not influenced by renal, liver or circulatory insufficiency. Degradation of a small ACURMIL aliquot is probably carried out by non-specific plasma esterases. Tests performed on the plasma of patients with low levels of pseudocholinesterase showed that ACURMIL inactivation proceeds without changes. Physiological changes in plasma ph or body temperature do not significantly influence the duration of ACURMIL action. Hemofiltration and hemodyalisis have minimal effects on the plasmatic levels of Atracurium and of its metabolites, including laudanosine. The effects of hemodyalisis and hemoperfusion on the plasmatic levels of Atracurium and its metabolites are not known Preclinical Safety Data No toxic effects were detected in patas monkeys following administration of nonparalyzing doses of 0.25 mg/kg of Atracurium s.c., administered 2-4 times a day for 28 days. 6. PHARMACEUTICAL PARTICULARS 6.1. List of Excipients Benzenesulfonic acid, water for injectable preparations
8 6.2. Incompatibilities ACURMIL should not be mixed in the same syringe with Thiopental or with any other alkaline substance, since high ph inactivates it Shelf-life 24 months 6.4. Special Storage Precautions Store at temperatures between 2 C and 8 C. Keep the ampoules in the outer carton in order to protect from light. Do not freeze Open, unused ampoules should be discarded Nature and contents of container Five 2.5 ml ampoules each containing 25 mg of Atracurium besylate Five 5.0 ml ampoules each containing 50 mg of Atracurium besylate 6.6 Instructions for Use See section Dosage and Administration 7. MARKETING AUTHORIZATION HOLDER Lab. It. Biochim.Farm.co LISAPHARMA S.p.A. Via Licinio ERBA (CO) 8. MARKETING AUTHORIZATION NUMBER(S) ACURMIL 25 five 2.5 ml ampoules AIC No ACURMIL 50 five 5.0 ml ampoules AIC No FIRST AUTHORIZATION /RENEWAL DATE / DATE OF THE LAST TEXT REVISION December 2013
TRACRIUM GlaxoSmithKline
 TRACRIUM GlaxoSmithKline Atracurium QUALITATIVE AND QUANTITATIVE COMPOSITION Injection: A sterile solution containing 10 mg atracurium besylate per ml, without an antimicrobial preservative, supplied in
TRACRIUM GlaxoSmithKline Atracurium QUALITATIVE AND QUANTITATIVE COMPOSITION Injection: A sterile solution containing 10 mg atracurium besylate per ml, without an antimicrobial preservative, supplied in
For the use only of Registered Medical Practitioners or a Hospital or a Laboratory TRACRIUM. Atracurium Besylate Injection IP
 For the use only of Registered Medical Practitioners or a Hospital or a Laboratory TRACRIUM Atracurium Besylate Injection IP QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml contains Atracurium Besylate
For the use only of Registered Medical Practitioners or a Hospital or a Laboratory TRACRIUM Atracurium Besylate Injection IP QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml contains Atracurium Besylate
PRODUCT INFORMATION. TRACRIUM INJECTION atracurium besylate DESCRIPTION:
 PRODUCT INFORMATION TRACRIUM INJECTION (atracurium besylate) NAME OF THE DRUG: atracurium besylate DESCRIPTION: TRACRIUM (atracurium besylate) is an intermediate-duration, non-depolarising, skeletal muscle
PRODUCT INFORMATION TRACRIUM INJECTION (atracurium besylate) NAME OF THE DRUG: atracurium besylate DESCRIPTION: TRACRIUM (atracurium besylate) is an intermediate-duration, non-depolarising, skeletal muscle
Core Safety Profile. Pharmaceutical form(s)/strength: Solution for Injection CZ/H/PSUR/0005/002 Date of FAR:
 Core Safety Profile Active substance: Rocuronium bromide Pharmaceutical form(s)/strength: Solution for Injection P-RMS: CZ/H/PSUR/0005/002 Date of FAR: 31.05.2012 4.3 Contraindications Hypersensitivity
Core Safety Profile Active substance: Rocuronium bromide Pharmaceutical form(s)/strength: Solution for Injection P-RMS: CZ/H/PSUR/0005/002 Date of FAR: 31.05.2012 4.3 Contraindications Hypersensitivity
PRODUCT INFORMATION CISATRACURIUM PFIZER NAME OF THE MEDICINE DESCRIPTION PHARMACOLOGY. Cisatracurium besylate
 PRODUCT INFORMATION CISATRACURIUM PFIZER Cisatracurium besylate NAME OF THE MEDICINE The chemical name of cisatracurium besylate is (1R,1 R,2R,2 R,)-2,2 -(3,11-dioxo-4,10- dioxatridecamethylene) bis (1,2,3,4-tetrahydro-6,7-dimethoxy-2-methyl-1-
PRODUCT INFORMATION CISATRACURIUM PFIZER Cisatracurium besylate NAME OF THE MEDICINE The chemical name of cisatracurium besylate is (1R,1 R,2R,2 R,)-2,2 -(3,11-dioxo-4,10- dioxatridecamethylene) bis (1,2,3,4-tetrahydro-6,7-dimethoxy-2-methyl-1-
PACKAGE LEAFLET: INFORMATION FOR THE USER. Atracurium Besilate 10 mg/ml Solution for Injection The active substance is atracurium besilate
 PACKAGE LEAFLET: INFORMATION FOR THE USER Atracurium Besilate 10 mg/ml Solution for Injection The active substance is atracurium besilate Read all of this leaflet carefully before you start using this
PACKAGE LEAFLET: INFORMATION FOR THE USER Atracurium Besilate 10 mg/ml Solution for Injection The active substance is atracurium besilate Read all of this leaflet carefully before you start using this
A Nondepolarizing Neuromuscular Blocking (NMB) Agent
 DOSING GUIDE A Nondepolarizing Neuromuscular Blocking (NMB) Agent Easy to remember dosing for the 0.20 mg/kg adult intubating doses of NIMBEX 1 *: For every 10 kg, give 1 ml of NIMBEX (2 mg/ml concentration)
DOSING GUIDE A Nondepolarizing Neuromuscular Blocking (NMB) Agent Easy to remember dosing for the 0.20 mg/kg adult intubating doses of NIMBEX 1 *: For every 10 kg, give 1 ml of NIMBEX (2 mg/ml concentration)
MIVACRON GlaxoSmithKline
 MIVACRON GlaxoSmithKline Mivacurium QUALITATIVE AND QUANTITATIVE COMPOSITION Injection: Sterile solution containing 2 mg mivacurium per ml as mivacurium chloride, without an antimicrobial preservative,
MIVACRON GlaxoSmithKline Mivacurium QUALITATIVE AND QUANTITATIVE COMPOSITION Injection: Sterile solution containing 2 mg mivacurium per ml as mivacurium chloride, without an antimicrobial preservative,
PRODUCT MONOGRAPH. Solution for Injection 20 mg/10 ml (2 mg/ml) Cisatracurium. Non-depolarizing Skeletal Neuromuscular Blocking Agent
 PRODUCT MONOGRAPH Pr CISATRACURIUM BESYLATE INJECTION Solution for Injection 20 mg/10 ml (2 mg/ml) Cisatracurium Non-depolarizing Skeletal Neuromuscular Blocking Agent This drug should be administered
PRODUCT MONOGRAPH Pr CISATRACURIUM BESYLATE INJECTION Solution for Injection 20 mg/10 ml (2 mg/ml) Cisatracurium Non-depolarizing Skeletal Neuromuscular Blocking Agent This drug should be administered
200 mg/20 ml (10 mg/ml) in single-dose vials (3)
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use NIMBEX safely and effectively. See full prescribing information for NIMBEX. NIMBEX (cisatracurium
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use NIMBEX safely and effectively. See full prescribing information for NIMBEX. NIMBEX (cisatracurium
FOR REPRESENTATIVE EDUCATION
 Neuromuscular Blockade in the ICU NIMBEX Indication 1 NIMBEX (cisatracurium besylate) is indicated as an adjunct to general anesthesia to facilitate tracheal intubation in adults and in pediatric patients
Neuromuscular Blockade in the ICU NIMBEX Indication 1 NIMBEX (cisatracurium besylate) is indicated as an adjunct to general anesthesia to facilitate tracheal intubation in adults and in pediatric patients
PACKAGE LEAFLET: INFORMATION FOR THE USER. Cisatracurium 2 mg/ml Solution for Injection/Infusion Cisatracurium 5 mg/ml Solution for Injection/Infusion
 PACKAGE LEAFLET: INFORMATION FOR THE USER Cisatracurium 2 mg/ml Solution for Injection/Infusion Cisatracurium 5 mg/ml Solution for Injection/Infusion Cisatracurium Read all of this leaflet carefully before
PACKAGE LEAFLET: INFORMATION FOR THE USER Cisatracurium 2 mg/ml Solution for Injection/Infusion Cisatracurium 5 mg/ml Solution for Injection/Infusion Cisatracurium Read all of this leaflet carefully before
08-15 NORCURON ORGANON
 08-15 NORCURON ORGANON 1. NAME OF THE MEDICINAL PRODUCT Norcuron 4 mg, powder for solution for injection Norcuron 10 mg, powder for solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Norcuron
08-15 NORCURON ORGANON 1. NAME OF THE MEDICINAL PRODUCT Norcuron 4 mg, powder for solution for injection Norcuron 10 mg, powder for solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Norcuron
2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Citanest with Octapressin Dental 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml contains Prilocaine Hydrochloride 30 mg (54 mg/1.8
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Citanest with Octapressin Dental 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml contains Prilocaine Hydrochloride 30 mg (54 mg/1.8
Protamine sulphate LEO Pharma 1400 anti-heparin IU/ml solution for injection and infusion.
 1. NAME OF THE MEDICINAL PRODUCT Protamine sulphate LEO Pharma 1400 anti-heparin IU/ml solution for injection and infusion. 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Protamine sulphate 1400 anti-heparin
1. NAME OF THE MEDICINAL PRODUCT Protamine sulphate LEO Pharma 1400 anti-heparin IU/ml solution for injection and infusion. 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Protamine sulphate 1400 anti-heparin
Please refer to the Summary of Product Characteristics (SPC) for further details on this product.
 [Aspen Logo] Information for the Physician Nimbex cisatracurium Please refer to the Summary of Product Characteristics (SPC) for further details on this product. Trade name of the medicinal product Nimbex
[Aspen Logo] Information for the Physician Nimbex cisatracurium Please refer to the Summary of Product Characteristics (SPC) for further details on this product. Trade name of the medicinal product Nimbex
NEW ZEALAND DATA SHEET. Injection 2 mg/ml: a clear, colourless, particle-free solution containing 2 mg/ml pancuronium bromide.
 NEW ZEALAND DATA SHEET NAME OF MEDICINE Pancuronium Bromide B.P. Injection 2 mg/ml PRESENTATION Injection 2 mg/ml: a clear, colourless, particle-free solution containing 2 mg/ml pancuronium bromide. INDICATIONS
NEW ZEALAND DATA SHEET NAME OF MEDICINE Pancuronium Bromide B.P. Injection 2 mg/ml PRESENTATION Injection 2 mg/ml: a clear, colourless, particle-free solution containing 2 mg/ml pancuronium bromide. INDICATIONS
Core Safety Profile. Pharmaceutical form(s)/strength: 5mg/ml and 25 mg/ml, Solution for injection, IM/IV FI/H/PSUR/0010/002 Date of FAR:
 Core Safety Profile Active substance: Esketamine Pharmaceutical form(s)/strength: 5mg/ml and 25 mg/ml, Solution for injection, IM/IV P-RMS: FI/H/PSUR/0010/002 Date of FAR: 29.05.2012 4.3 Contraindications
Core Safety Profile Active substance: Esketamine Pharmaceutical form(s)/strength: 5mg/ml and 25 mg/ml, Solution for injection, IM/IV P-RMS: FI/H/PSUR/0010/002 Date of FAR: 29.05.2012 4.3 Contraindications
This drug should be administered only by adequately trained individuals familiar with its actions, characteristics, and hazards.
 MIVACRON Injection (mivacurium chloride) This drug should be administered only by adequately trained individuals familiar with its actions, characteristics, and hazards. DESCRIPTION MIVACRON (mivacurium
MIVACRON Injection (mivacurium chloride) This drug should be administered only by adequately trained individuals familiar with its actions, characteristics, and hazards. DESCRIPTION MIVACRON (mivacurium
NIMBEX Injection. This information is intended for U.S. residents only. only. (cisatracurium besylate)
 This information is intended for U.S. residents only. NIMBEX Injection (cisatracurium besylate) only DESCRIPTION CLINICAL PHARMACOLOGY INDICATIONS AND USAGE CONTRAINDICATIONS WARNINGS PRECAUTIONS ADVERSE
This information is intended for U.S. residents only. NIMBEX Injection (cisatracurium besylate) only DESCRIPTION CLINICAL PHARMACOLOGY INDICATIONS AND USAGE CONTRAINDICATIONS WARNINGS PRECAUTIONS ADVERSE
NEW ZEALAND DATA SHEET
 NEW ZEALAND DATA SHEET VECURE 1. Product Name VECURE 10 mg, powder for injection. 2. Qualitative and Quantitative Composition VECURE 10 mg, 1 vial contains 10 mg vecuronium bromide. When reconstituted
NEW ZEALAND DATA SHEET VECURE 1. Product Name VECURE 10 mg, powder for injection. 2. Qualitative and Quantitative Composition VECURE 10 mg, 1 vial contains 10 mg vecuronium bromide. When reconstituted
ACETYLCYSTEINE INJECTION
 ACETYLCYSTEINE INJECTION 1. Name of the medicinal product Acetylcysteine 200 mg/ml Injection 2. Qualitative and quantitative composition Acetylcysteine 200mg per ml (as N-acetylcysteine) Each 10ml ampoule
ACETYLCYSTEINE INJECTION 1. Name of the medicinal product Acetylcysteine 200 mg/ml Injection 2. Qualitative and quantitative composition Acetylcysteine 200mg per ml (as N-acetylcysteine) Each 10ml ampoule
NIMBEX (cisatracurium besylate) injection
 NIMBEX (cisatracurium besylate) injection This drug should be administered only by adequately trained individuals familiar with its actions, characteristics, and hazards. NOT FOR USE IN NEONATES CONTAINS
NIMBEX (cisatracurium besylate) injection This drug should be administered only by adequately trained individuals familiar with its actions, characteristics, and hazards. NOT FOR USE IN NEONATES CONTAINS
Core Safety Profile. Date of FAR:
 Core Safety Profile Active substance: Levobupivicaine Pharmaceutical form(s)/strength: Solution for injection, concentrate for solution for infusion, 2,5 mg/ml, 5 mg/ml, 7,5 mg/ml, 0,625 mg/ml, 1,25 mg/ml
Core Safety Profile Active substance: Levobupivicaine Pharmaceutical form(s)/strength: Solution for injection, concentrate for solution for infusion, 2,5 mg/ml, 5 mg/ml, 7,5 mg/ml, 0,625 mg/ml, 1,25 mg/ml
DATA SHEET. Product Summary. 1. Trade Name of Medicinal Product. Protamine Sulphate Injection BP. 2. Qualitative and Quantitative Composition
 DATA SHEET Product Summary 1. Trade Name of Medicinal Product Protamine Sulphate Injection BP 2. Qualitative and Quantitative Composition Protamine Sulphate 10mg/ml 3. Pharmaceutical Form Solution for
DATA SHEET Product Summary 1. Trade Name of Medicinal Product Protamine Sulphate Injection BP 2. Qualitative and Quantitative Composition Protamine Sulphate 10mg/ml 3. Pharmaceutical Form Solution for
NEW ZEALAND DATA SHEET. Injection solution 2.5 mg/ml: a clear, colourless, particle-free solution containing 2.5 mg/ml Neostigmine methylsulphate.
 NEW ZEALAND DATA SHEET NAME OF MEDICINE NEOSTIGMINE METHYLSULPHATE INJECTION B.P. Solution for injection 2.5 mg/ml. PRESENTATIONS Injection solution 2.5 mg/ml: a clear, colourless, particle-free solution
NEW ZEALAND DATA SHEET NAME OF MEDICINE NEOSTIGMINE METHYLSULPHATE INJECTION B.P. Solution for injection 2.5 mg/ml. PRESENTATIONS Injection solution 2.5 mg/ml: a clear, colourless, particle-free solution
Package leaflet: Information for the user
 Package leaflet: Information for the user Cisatracurium 2 mg/ml Solution for injection/ infusion Cisatracurium 5 mg/ml Solution for injection/ infusion Cisatracurium The name of your medicine are Cisatracurium
Package leaflet: Information for the user Cisatracurium 2 mg/ml Solution for injection/ infusion Cisatracurium 5 mg/ml Solution for injection/ infusion Cisatracurium The name of your medicine are Cisatracurium
PACKAGE LEAFLET: INFORMATION FOR THE USER. Cisatracurium 2 mg/ml solution for injection/infusion Cisatracurium 5 mg/ml solution for injection/infusion
 PACKAGE LEAFLET: INFORMATION FOR THE USER Cisatracurium 2 mg/ml solution for injection/infusion Cisatracurium 5 mg/ml solution for injection/infusion Cisatracurium Read all of this leaflet carefully before
PACKAGE LEAFLET: INFORMATION FOR THE USER Cisatracurium 2 mg/ml solution for injection/infusion Cisatracurium 5 mg/ml solution for injection/infusion Cisatracurium Read all of this leaflet carefully before
Shaded areas=not MARKETED 24/2/09
 PACKAGE INSERT SCHEDULING STATUS Schedule 6 PROPRIETARY NAME AND DOSAGE FORM RAPIFEN 2 ml IV injection RAPIFEN 10 ml IV injection COMPOSITION Each ml contains alfentanil hydrochloride 0,544 mg (equivalent
PACKAGE INSERT SCHEDULING STATUS Schedule 6 PROPRIETARY NAME AND DOSAGE FORM RAPIFEN 2 ml IV injection RAPIFEN 10 ml IV injection COMPOSITION Each ml contains alfentanil hydrochloride 0,544 mg (equivalent
NERVOUS SYSTEM NERVOUS SYSTEM. Somatic nervous system. Brain Spinal Cord Autonomic nervous system. Sympathetic nervous system
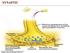 SYNAPTIC NERVOUS SYSTEM NERVOUS SYSTEM CENTRAL NERVOUS SYSTEM PERIPHERAL NERVOUS SYSTEM Brain Spinal Cord Autonomic nervous system Somatic nervous system Sympathetic nervous system Parasympathetic nervous
SYNAPTIC NERVOUS SYSTEM NERVOUS SYSTEM CENTRAL NERVOUS SYSTEM PERIPHERAL NERVOUS SYSTEM Brain Spinal Cord Autonomic nervous system Somatic nervous system Sympathetic nervous system Parasympathetic nervous
Chapter 18 Neuromuscular Blocking Agents Study Guide and Application Exercise
 Chapter 18 Neuromuscular Blocking Agents Study Guide and Application Exercise 1. Read chapter 2. Review objectives (p.305) 3. Review key terms and definitions (p.305) Add: Cholinesterase inhibitor Vagal
Chapter 18 Neuromuscular Blocking Agents Study Guide and Application Exercise 1. Read chapter 2. Review objectives (p.305) 3. Review key terms and definitions (p.305) Add: Cholinesterase inhibitor Vagal
2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 AUSTRALIAN PRODUCT INFORMATION NEOSTIGMINE INJECTION BP (NEOSTIGMINE METHYLSULFATE) 1 NAME OF THE MEDICINE Neostigmine methylsulfate. 2 QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml sterile solution of
AUSTRALIAN PRODUCT INFORMATION NEOSTIGMINE INJECTION BP (NEOSTIGMINE METHYLSULFATE) 1 NAME OF THE MEDICINE Neostigmine methylsulfate. 2 QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml sterile solution of
2. QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Acetylcysteine 200 mg/ml Injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Acetylcysteine 200 mg per ml. Each 10ml ampoule contains
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Acetylcysteine 200 mg/ml Injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Acetylcysteine 200 mg per ml. Each 10ml ampoule contains
NEUROMUSCULAR BLOCKING AGENTS
 NEUROMUSCULAR BLOCKING AGENTS Edward JN Ishac, Ph.D. Associate Professor, Pharmacology and Toxicology Smith 742, 828-2127, Email: eishac@vcu.edu Learning Objectives: 1. Understand the physiology of the
NEUROMUSCULAR BLOCKING AGENTS Edward JN Ishac, Ph.D. Associate Professor, Pharmacology and Toxicology Smith 742, 828-2127, Email: eishac@vcu.edu Learning Objectives: 1. Understand the physiology of the
New Zealand Data Sheet DBL TM Rocuronium Bromide Injection
 New Zealand Data Sheet DBL TM Rocuronium Bromide Injection NAME OF THE MEDICINE DBL TM Rocuronium Bromide Injection 50 mg/5 ml Vial PRESENTATION Rocuronium bromide is a quaternary aminosteroid and an analogue
New Zealand Data Sheet DBL TM Rocuronium Bromide Injection NAME OF THE MEDICINE DBL TM Rocuronium Bromide Injection 50 mg/5 ml Vial PRESENTATION Rocuronium bromide is a quaternary aminosteroid and an analogue
SUMMARY OF PRODUCT CHARACTERISTICS 2. QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS PRODUCT SUMMARY 1. NAME OF THE MEDICINAL PRODUCT Sterile Potassium Chloride Concentrate 15%. 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 15% of Potassium Chloride in
SUMMARY OF PRODUCT CHARACTERISTICS PRODUCT SUMMARY 1. NAME OF THE MEDICINAL PRODUCT Sterile Potassium Chloride Concentrate 15%. 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 15% of Potassium Chloride in
Benztropine and trihexyphenidyl: Centrally acting antimuscarinic agents used for treatment of Parkinson disease & extrapyramidal symptoms.
 Scopolamine: Tertiary amine plant alkaloid. Produces peripheral effects similar to those of atropine. Unlike atropine, scopolamine has greater action on the CNS (observed at therapeutic doses). It has
Scopolamine: Tertiary amine plant alkaloid. Produces peripheral effects similar to those of atropine. Unlike atropine, scopolamine has greater action on the CNS (observed at therapeutic doses). It has
2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 1 PRODUCT NAME TROPISETRON-AFT tropisetron hydrochloride (equivalent to 2 mg or 5 mg tropisetron) per ampoule. 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml contains 1 mg of tropisetron 1 2 ml ampoule
1 PRODUCT NAME TROPISETRON-AFT tropisetron hydrochloride (equivalent to 2 mg or 5 mg tropisetron) per ampoule. 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml contains 1 mg of tropisetron 1 2 ml ampoule
LACIPIL QUALITATIVE AND QUANTITATIVE COMPOSITION
 LACIPIL lacidipine QUALITATIVE AND QUANTITATIVE COMPOSITION Lacidipine, 2 mg - round shaped white engraved on one face. Lacidipine, 4 mg - oval white with break line on both faces. Lacidipine, 6 mg - oval
LACIPIL lacidipine QUALITATIVE AND QUANTITATIVE COMPOSITION Lacidipine, 2 mg - round shaped white engraved on one face. Lacidipine, 4 mg - oval white with break line on both faces. Lacidipine, 6 mg - oval
ULTIVA. Remifentanil hydrochloride
 ULTIVA Remifentanil hydrochloride QUALITATIVE AND QUANTITATIVE COMPOSITION Remifentanil for injection is a sterile, preservative-free, white to off white, lyophilised powder, to be reconstituted before
ULTIVA Remifentanil hydrochloride QUALITATIVE AND QUANTITATIVE COMPOSITION Remifentanil for injection is a sterile, preservative-free, white to off white, lyophilised powder, to be reconstituted before
2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Albunorm 5%, 50 g/l, solution for infusion 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Albunorm 5% is a solution containing 50 g/l of total
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Albunorm 5%, 50 g/l, solution for infusion 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Albunorm 5% is a solution containing 50 g/l of total
Glycopyrronium Bromide and Neostigmine Metilsulfate 0.5mg/2.5mg per ml
 New Zealand Datasheet Glycopyrronium Bromide and Neostigmine Metilsulfate 0.5mg/2.5mg per ml Solution for injection Glycopyrronium bromide 0.5 mg/ml & Neostigmine metilsulfate 2.5 mg/ml Presentation is
New Zealand Datasheet Glycopyrronium Bromide and Neostigmine Metilsulfate 0.5mg/2.5mg per ml Solution for injection Glycopyrronium bromide 0.5 mg/ml & Neostigmine metilsulfate 2.5 mg/ml Presentation is
ULTIVA GlaxoSmithKline
 ULTIVA GlaxoSmithKline Remifentanil QUALITATIVE AND QUANTITATIVE COMPOSITION Remifentanil for injection is a sterile, endotoxin-free, preservative-free, white to off white, lyophilised powder, to be reconstituted
ULTIVA GlaxoSmithKline Remifentanil QUALITATIVE AND QUANTITATIVE COMPOSITION Remifentanil for injection is a sterile, endotoxin-free, preservative-free, white to off white, lyophilised powder, to be reconstituted
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Comfora 595 mg film-coated tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION One film-coated tablet contains: glucosamine sulphate
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Comfora 595 mg film-coated tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION One film-coated tablet contains: glucosamine sulphate
SUMMARY OF PRODUCT CHARACTERISTICS. 1. NAME OF THE MEDICINAL PRODUCT Rocuronium Kabi 10 mg/ml solution for injection/infusion
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Rocuronium Kabi 10 mg/ml solution for injection/infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml of solution for injection
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Rocuronium Kabi 10 mg/ml solution for injection/infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml of solution for injection
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS Page 1 / 6 1. NAME OF THE VETERINARY MEDICINAL PRODUCT Fentadon 50 microgram/ml, solution for injection for dogs SE, DK: Fentadon Vet. 50 microgram/ml, solution for injection
SUMMARY OF PRODUCT CHARACTERISTICS Page 1 / 6 1. NAME OF THE VETERINARY MEDICINAL PRODUCT Fentadon 50 microgram/ml, solution for injection for dogs SE, DK: Fentadon Vet. 50 microgram/ml, solution for injection
MUSCLE RELAXANTS. Mr. D.Raju, M.pharm, Lecturer
 MUSCLE RELAXANTS Mr. D.Raju, M.pharm, Lecturer Muscle Relaxants are classified as: I)Peripherally acting A.Neuromuscular blocking agents:- 1) Depolarizing muscle relaxants. 2) Non-depolarizing muscle relaxants
MUSCLE RELAXANTS Mr. D.Raju, M.pharm, Lecturer Muscle Relaxants are classified as: I)Peripherally acting A.Neuromuscular blocking agents:- 1) Depolarizing muscle relaxants. 2) Non-depolarizing muscle relaxants
Granisetron Kabi, 1mg/ml, concentrate for solution for injection/infusion
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Granisetron Kabi, 1mg/ml, concentrate for solution for injection/infusion 2 QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml contains granisetron
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Granisetron Kabi, 1mg/ml, concentrate for solution for injection/infusion 2 QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml contains granisetron
SUMMARY OF PRODUCT CHARACTERISTICS. Each ml solution for injection contains phenylephrine hydrochloride corresponding to 0.1 mg phenylephrine.
 1. NAME OF THE MEDICINAL PRODUCT Fenylefrin Abcur 0.05 mg/ml, solution for injection Fenylefrin Abcur 0.1 mg/ml, solution for injection SUMMARY OF PRODUCT CHARACTERISTICS 2. QUALITATIVE AND QUANTITATIVE
1. NAME OF THE MEDICINAL PRODUCT Fenylefrin Abcur 0.05 mg/ml, solution for injection Fenylefrin Abcur 0.1 mg/ml, solution for injection SUMMARY OF PRODUCT CHARACTERISTICS 2. QUALITATIVE AND QUANTITATIVE
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Colecalciferol Meda 800 IU tablet 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains colecalciferol (vitamin D 3 ) 800 IU
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Colecalciferol Meda 800 IU tablet 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains colecalciferol (vitamin D 3 ) 800 IU
2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Rocuronium bromide 10 mg/ml solution for injection/ infusion 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml of solution of Rocuronium
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Rocuronium bromide 10 mg/ml solution for injection/ infusion 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml of solution of Rocuronium
SUMMARY OF PRODUCT CHARACTERISTICS 2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT SCANDONEST 3% PLAIN BIOCAINE 3% PLAIN, solution for injection 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Mepivacaine hydrochloride 30.00
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT SCANDONEST 3% PLAIN BIOCAINE 3% PLAIN, solution for injection 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Mepivacaine hydrochloride 30.00
SUMMARY OF PRODUCT CHARACTERISTICS. Flexbumin 200 g/l is a solution containing 200 g/l (20%) of total protein of which at least 95% is human albumin.
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Flexbumin 200g/l solution for infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Flexbumin 200 g/l is a solution containing 200 g/l
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Flexbumin 200g/l solution for infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Flexbumin 200 g/l is a solution containing 200 g/l
DBL MAGNESIUM SULFATE CONCENTRATED INJECTION
 DBL MAGNESIUM SULFATE CONCENTRATED INJECTION NAME OF MEDICINE Magnesium Sulfate BP DESCRIPTION DBL Magnesium Sulfate Concentrated Injection is a clear, colourless, sterile solution. Each ampoule contains
DBL MAGNESIUM SULFATE CONCENTRATED INJECTION NAME OF MEDICINE Magnesium Sulfate BP DESCRIPTION DBL Magnesium Sulfate Concentrated Injection is a clear, colourless, sterile solution. Each ampoule contains
Summary of Product Characteristics
 1 NAME OF THE MEDICINAL PRODUCT Etoflam 5% w/w Gel Summary of Product Characteristics 2 QUALITATIVE AND QUANTITATIVE COMPOSITION The gel contains 5% w/w etofenamate. For a full list of excipients, see
1 NAME OF THE MEDICINAL PRODUCT Etoflam 5% w/w Gel Summary of Product Characteristics 2 QUALITATIVE AND QUANTITATIVE COMPOSITION The gel contains 5% w/w etofenamate. For a full list of excipients, see
P-RMS: NO/H/PSUR/0009/001
 Core Safety Profile Active substance: Propofol Pharmaceutical form(s)/strength: Emulsion for injection, infusion, 10mg/ml Emulsion for infusion, 20mg/ml P-RMS: NO/H/PSUR/0009/001 Date of FAR: 30.06.2011
Core Safety Profile Active substance: Propofol Pharmaceutical form(s)/strength: Emulsion for injection, infusion, 10mg/ml Emulsion for infusion, 20mg/ml P-RMS: NO/H/PSUR/0009/001 Date of FAR: 30.06.2011
Pharmacology of the Neuromuscular Junction (NMJ)
 Pharmacology of the Neuromuscular Junction (NMJ) Edward JN Ishac, Ph.D. Professor Smith Building, Room 742 eishac@vcu.edu 828 2127 Department of Pharmacology and Toxicology Medical College of Virginia
Pharmacology of the Neuromuscular Junction (NMJ) Edward JN Ishac, Ph.D. Professor Smith Building, Room 742 eishac@vcu.edu 828 2127 Department of Pharmacology and Toxicology Medical College of Virginia
VENTOLIN RESPIRATOR SOLUTION
 VENTOLIN RESPIRATOR SOLUTION Salbutamol QUALITATIVE AND QUANTITATIVE COMPOSITION VENTOLIN Respirator Solution contains 5mg salbutamol, as sulphate, per ml of solution and is supplied in 10 ml bottles.
VENTOLIN RESPIRATOR SOLUTION Salbutamol QUALITATIVE AND QUANTITATIVE COMPOSITION VENTOLIN Respirator Solution contains 5mg salbutamol, as sulphate, per ml of solution and is supplied in 10 ml bottles.
SUMMARY OF PRODUCT CHARACTERISTICS. Albuman 40 g/l is a solution containing 40 g/l (4%) of total protein of which at least 95% is human albumin.
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Albuman 40 g/l solution for infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Albuman 40 g/l is a solution containing 40 g/l (4%)
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Albuman 40 g/l solution for infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Albuman 40 g/l is a solution containing 40 g/l (4%)
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS Page 1 of 9 1. NAME OF THE MEDICINAL PRODUCT Magnesium sulfate-kalceks 500 mg/ml solution for injection, BP * 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml of solution
SUMMARY OF PRODUCT CHARACTERISTICS Page 1 of 9 1. NAME OF THE MEDICINAL PRODUCT Magnesium sulfate-kalceks 500 mg/ml solution for injection, BP * 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml of solution
Pharmacology of the Neuromuscular Junction (NMJ)
 Pharmacology of the Neuromuscular Junction (NMJ) Edward JN Ishac, Ph.D. Professor Smith Building, Room 742 eishac@vcu.edu 828 2127 Department of Pharmacology and Toxicology Medical College of Virginia
Pharmacology of the Neuromuscular Junction (NMJ) Edward JN Ishac, Ph.D. Professor Smith Building, Room 742 eishac@vcu.edu 828 2127 Department of Pharmacology and Toxicology Medical College of Virginia
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT "Carbellon" Tablets. 2. Qualitative and Quantitative Composition Active Constituents: Activated Charcoal Ph.Eur. Magnesium Hydroxide Ph.Eur.
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT "Carbellon" Tablets. 2. Qualitative and Quantitative Composition Active Constituents: Activated Charcoal Ph.Eur. Magnesium Hydroxide Ph.Eur.
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT 2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Albunorm 20%, 200 g/l, solution for infusion 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Albunorm 20% is a solution containing 200 g/l
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Albunorm 20%, 200 g/l, solution for infusion 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Albunorm 20% is a solution containing 200 g/l
SUMMARY OF PRODUCT CHARACTERISTICS. Potassium Chloride 0.15% w/v & Sodium Chloride 0.9% w/v Solution for Infusion
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Potassium Chloride 0.15% w/v & Sodium Chloride 0.9% w/v Solution for Infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Potassium Chloride
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Potassium Chloride 0.15% w/v & Sodium Chloride 0.9% w/v Solution for Infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Potassium Chloride
50% Concentrated Injection
 NAME OF THE MEDICINE. The molecular weight of the compound is 246.5 and the CAS registry number is 10034-99-8. The molecular formula is MgSO4, 7H2O. DESCRIPTION MAGNESIUM SULFATE HEPTAHYDRATE 50% CONCENTRATED
NAME OF THE MEDICINE. The molecular weight of the compound is 246.5 and the CAS registry number is 10034-99-8. The molecular formula is MgSO4, 7H2O. DESCRIPTION MAGNESIUM SULFATE HEPTAHYDRATE 50% CONCENTRATED
SUMMARY OF PRODUCT CHARACTERISTICS 2. QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Lidokain Isdin 40 mg/g cream 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 g cream contains 40 mg lidocaine. Excipients: Propylene glycol
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Lidokain Isdin 40 mg/g cream 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 g cream contains 40 mg lidocaine. Excipients: Propylene glycol
3. PHARMACEUTICAL FORM Solution for infusion. A clear, slightly viscous liquid; it is almost colourless, yellow, amber or green.
 1. NAME OF THE MEDICINAL PRODUCT Albutein 250 g/l, solution for infusion. 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Human albumin Albutein 250 g/l is a solution containing 250 g/l of total protein of
1. NAME OF THE MEDICINAL PRODUCT Albutein 250 g/l, solution for infusion. 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Human albumin Albutein 250 g/l is a solution containing 250 g/l of total protein of
AUSTRALIAN PRODUCT INFORMATION REMIFENTANIL APOTEX (REMIFENTANIL HYDROCHLORIDE) POWDER FOR INJECTION
 AUSTRALIAN PRODUCT INFORMATION REMIFENTANIL APOTEX (REMIFENTANIL HYDROCHLORIDE) POWDER FOR INJECTION 1 NAME OF THE MEDICINE Remifentanil (as hydrochloride) 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each
AUSTRALIAN PRODUCT INFORMATION REMIFENTANIL APOTEX (REMIFENTANIL HYDROCHLORIDE) POWDER FOR INJECTION 1 NAME OF THE MEDICINE Remifentanil (as hydrochloride) 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each
DATA SHEET. Remifentanil hydrochloride for injection1mg and 2mg (remifentanil base) vials
 DATA SHEET 1. PRODUCT NAME ULTIVA Remifentanil hydrochloride for injection1mg and 2mg (remifentanil base) vials 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Active substance Remifentanil hydrochloride 1mg
DATA SHEET 1. PRODUCT NAME ULTIVA Remifentanil hydrochloride for injection1mg and 2mg (remifentanil base) vials 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Active substance Remifentanil hydrochloride 1mg
Pharmaceutical form(s)/strength: Solution: 5 mg/ml Suspensions: 2.5 and 5 mg/ml P-RMS:
 0BCore Safety Profile Active substance: Betaxolol eyedrops Pharmaceutical form(s)/strength: Solution: 5 mg/ml Suspensions: 2.5 and 5 mg/ml P-RMS: HU/H/PSUR/0010/002 Date of FAR: 20.03.2013 4.2 Posology
0BCore Safety Profile Active substance: Betaxolol eyedrops Pharmaceutical form(s)/strength: Solution: 5 mg/ml Suspensions: 2.5 and 5 mg/ml P-RMS: HU/H/PSUR/0010/002 Date of FAR: 20.03.2013 4.2 Posology
NEW ZEALAND DATA SHEET
 1 ONDANSETRON-CLARIS 2 mg/ml solution for injection Ondansetron-Claris Solution for Injection 2 mg/ml 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Ondansetron-Claris, Solution for Injection, is a clear colourless
1 ONDANSETRON-CLARIS 2 mg/ml solution for injection Ondansetron-Claris Solution for Injection 2 mg/ml 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Ondansetron-Claris, Solution for Injection, is a clear colourless
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Alburex 5, 50 g/l, solution for infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Alburex 5 is a solution containing 50 g/l of total
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Alburex 5, 50 g/l, solution for infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Alburex 5 is a solution containing 50 g/l of total
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT DOTAREM 0.5 mmol/ml solution for injection in vials 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml solution for injection contains
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT DOTAREM 0.5 mmol/ml solution for injection in vials 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml solution for injection contains
PROSTIGMIN (neostigmine bromide)
 PROSTIGMIN (neostigmine bromide) TABLETS DESCRIPTION: Prostigmin (neostigmine bromide), an anticholinesterase agent, is available for oral administration in 15 mg tablets. Each tablet also contains gelatin,
PROSTIGMIN (neostigmine bromide) TABLETS DESCRIPTION: Prostigmin (neostigmine bromide), an anticholinesterase agent, is available for oral administration in 15 mg tablets. Each tablet also contains gelatin,
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE VETERINARY MEDICINAL PRODUCT Tralieve 50 mg/ml solution for injection for dogs (AT, BE, BG, CY, CZ, DE, EL, ES, HR, HU, IE, IT, LU, NL, PT, RO,
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE VETERINARY MEDICINAL PRODUCT Tralieve 50 mg/ml solution for injection for dogs (AT, BE, BG, CY, CZ, DE, EL, ES, HR, HU, IE, IT, LU, NL, PT, RO,
Ganglion blocking agents
 Ganglion blocking agents -out of date -Specifically act on the nicotinic receptors of both parasymphatetic and sympathetic ganglia - no selectivity toward PG or SG -These drugs are non-depolarizing, competitive
Ganglion blocking agents -out of date -Specifically act on the nicotinic receptors of both parasymphatetic and sympathetic ganglia - no selectivity toward PG or SG -These drugs are non-depolarizing, competitive
Active ingredients: Metoclopramide Hydrochloride mg Equivalent to metoclopramide hydrochloride anhydrous mg
 Name Primperan 10 mg / 2 ml Metoclopramide hydrochloride anhydrous Solution for I.M. or I.V. injection (Ampoules) Composition Each 2 ml ampoule contains: Active ingredients: Metoclopramide Hydrochloride.
Name Primperan 10 mg / 2 ml Metoclopramide hydrochloride anhydrous Solution for I.M. or I.V. injection (Ampoules) Composition Each 2 ml ampoule contains: Active ingredients: Metoclopramide Hydrochloride.
Salapin: Salbutamol BP 2mg as sulphate in each 5mL of a raspberry cola flavoured, sugar free syrup.
 Salapin Salbutamol Syrup 2mg/5mL Qualitative and quantitative composition Salapin: Salbutamol BP 2mg as sulphate in each 5mL of a raspberry cola flavoured, sugar free syrup. Clinical particulars Therapeutic
Salapin Salbutamol Syrup 2mg/5mL Qualitative and quantitative composition Salapin: Salbutamol BP 2mg as sulphate in each 5mL of a raspberry cola flavoured, sugar free syrup. Clinical particulars Therapeutic
SUMMARY OF PRODUCT CHARACTERISTICS. 1 ml solution contains 75 micrograms of sufentanilcitrate, corresponding to 50 micrograms of sufentanil.
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Sufentanil Narcomed, 50 microgram / ml, solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml solution contains 75
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Sufentanil Narcomed, 50 microgram / ml, solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml solution contains 75
DBL NALOXONE HYDROCHLORIDE INJECTION USP
 Name of medicine Naloxone hydrochloride Data Sheet New Zealand DBL NALXNE HYDRCHLRIDE INJECTIN USP Presentation DBL Naloxone Hydrochloride Injection USP is a sterile, clear, colourless solution, free from
Name of medicine Naloxone hydrochloride Data Sheet New Zealand DBL NALXNE HYDRCHLRIDE INJECTIN USP Presentation DBL Naloxone Hydrochloride Injection USP is a sterile, clear, colourless solution, free from
PACKAGE LEAFLET: INFORMATION FOR THE USER. SODIPHOS 22mEq / 10ml Concentrate for solution for infusion. Disodium phosphate dihydrate
 PACKAGE LEAFLET: INFORMATION FOR THE USER SODIPHOS 22mEq / 10ml Concentrate for solution for infusion Disodium phosphate dihydrate Read all of this leaflet carefully before you start using this medicine.
PACKAGE LEAFLET: INFORMATION FOR THE USER SODIPHOS 22mEq / 10ml Concentrate for solution for infusion Disodium phosphate dihydrate Read all of this leaflet carefully before you start using this medicine.
COMPANY CORE PACKAGE INSERT CCPI (PI/CORE/ENGLISH)
 COMPANY CORE PACKAGE INSERT CCPI (PI/CORE/ENGLISH) HUMAN ALBUMIN 20 % BEHRING Rev.: 05-MAR-2008 / PEI approval 26.02.08 Supersedes previous versions Rev.: 28-NOV-2007 / Adaptation to Core SPC Rev.: 02-JAN-2007
COMPANY CORE PACKAGE INSERT CCPI (PI/CORE/ENGLISH) HUMAN ALBUMIN 20 % BEHRING Rev.: 05-MAR-2008 / PEI approval 26.02.08 Supersedes previous versions Rev.: 28-NOV-2007 / Adaptation to Core SPC Rev.: 02-JAN-2007
Human Albumin 200 g/l Baxter is a solution containing 200 g/l of total protein of which at least 95% is human albumin.
 1. NAME OF THE MEDICINAL PRODUCT Human Albumin 200 g/l Baxter Solution for Infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Human Albumin 200 g/l Baxter is a solution containing 200 g/l of total protein
1. NAME OF THE MEDICINAL PRODUCT Human Albumin 200 g/l Baxter Solution for Infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Human Albumin 200 g/l Baxter is a solution containing 200 g/l of total protein
(PP XI) Dr. Samir Matloob
 DRUGS ACTING ON THE CHOLINERGIC SYSTEM AND THE NEUROMUSCULAR BLOCKING DRUGS IV (NICOTINIC ANTAGONISTS) (PP XI) Dr. Samir Matloob Dept. of Pharmacology Baghdad College of Medicine Drugs acting on the cholinergic
DRUGS ACTING ON THE CHOLINERGIC SYSTEM AND THE NEUROMUSCULAR BLOCKING DRUGS IV (NICOTINIC ANTAGONISTS) (PP XI) Dr. Samir Matloob Dept. of Pharmacology Baghdad College of Medicine Drugs acting on the cholinergic
(human albumin solution) POM SUMMARY OF PRODUCT CHARACTERISTICS
 albunorm TM 20% (human albumin solution) POM SUMMARY OF PRODUCT CHARACTERISTICS UK IRELAND and Ireland Octapharma Limited The Zenith Building, 26 Spring Gardens Manchester M2 1AB United Kingdom 1. Name
albunorm TM 20% (human albumin solution) POM SUMMARY OF PRODUCT CHARACTERISTICS UK IRELAND and Ireland Octapharma Limited The Zenith Building, 26 Spring Gardens Manchester M2 1AB United Kingdom 1. Name
NEW ZEALAND DATA SHEET
 1 PRODUCT NAME Acetylcysteine 200 mg/ml Injection 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Acetylcysteine 200 mg per ml (as N-acetylcysteine) Each 10 ml ampoule contains 2 g N-acetylcysteine Excipients
1 PRODUCT NAME Acetylcysteine 200 mg/ml Injection 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Acetylcysteine 200 mg per ml (as N-acetylcysteine) Each 10 ml ampoule contains 2 g N-acetylcysteine Excipients
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Otrivin Menthol (without preservative) 1.0 mg/ml nasal spray, solution 2 QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml contains 1.0 mg
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Otrivin Menthol (without preservative) 1.0 mg/ml nasal spray, solution 2 QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml contains 1.0 mg
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Procto-Glyvenol, 400 mg + 40 mg, suppository Procto-Glyvenol, 5% + 2%, rectal cream 2. QUALITATIVE AND QUANTITATIVE COMPOSITION One suppository
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Procto-Glyvenol, 400 mg + 40 mg, suppository Procto-Glyvenol, 5% + 2%, rectal cream 2. QUALITATIVE AND QUANTITATIVE COMPOSITION One suppository
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT 2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Zaditen 0.25 mg/ml, eye drops, solution 2 QUALITATIVE AND QUANTITATIVE COMPOSITION One ml contains 0.345 mg ketotifen fumarate corresponding
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Zaditen 0.25 mg/ml, eye drops, solution 2 QUALITATIVE AND QUANTITATIVE COMPOSITION One ml contains 0.345 mg ketotifen fumarate corresponding
(human albumin solution) POM SUMMARY OF PRODUCT CHARACTERISTICS
 albunorm TM 5% (human albumin solution) POM SUMMARY OF PRODUCT CHARACTERISTICS UK IRELAND and Ireland Octapharma Limited The Zenith Building, 26 Spring Gardens Manchester M2 1AB United Kingdom 1. Name
albunorm TM 5% (human albumin solution) POM SUMMARY OF PRODUCT CHARACTERISTICS UK IRELAND and Ireland Octapharma Limited The Zenith Building, 26 Spring Gardens Manchester M2 1AB United Kingdom 1. Name
A specific opioid antagonist, such as naloxone immediately and completely reverses all actions of alfentanil.
 RAPIFEN DATA SHEET NAME OF THE MEDICINE RAPIFEN alfentanil 0.5 mg/ml injection PRESENTATION RAPIFEN is a sterile, preservative free, isotonic, aqueous solution containing alfentanil hydrochloride equivalent
RAPIFEN DATA SHEET NAME OF THE MEDICINE RAPIFEN alfentanil 0.5 mg/ml injection PRESENTATION RAPIFEN is a sterile, preservative free, isotonic, aqueous solution containing alfentanil hydrochloride equivalent
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Albunorm 20%, 200g/l, solution for infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Albunorm 20% is a solution containing 200 g/l
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Albunorm 20%, 200g/l, solution for infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Albunorm 20% is a solution containing 200 g/l
APPENDIX 1 SUMMARY OF PRODUCT CHARACTERISTICS
 SmPC Country: Lebanon Date of approval : 17/08/2011 Procedure : National APPENDIX 1 SUMMARY OF PRODUCT CHARACTERISTICS 1- NAME OF THE MEDICINAL PRODUCT DECAN, concentrate for solution for infusion 2- QUALITATIVE
SmPC Country: Lebanon Date of approval : 17/08/2011 Procedure : National APPENDIX 1 SUMMARY OF PRODUCT CHARACTERISTICS 1- NAME OF THE MEDICINAL PRODUCT DECAN, concentrate for solution for infusion 2- QUALITATIVE
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT WICK Chesty Cough Syrup 200 mg/15 ml 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 15 ml syrup contains 200 mg guaifenesin. Each ml syrup
SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT WICK Chesty Cough Syrup 200 mg/15 ml 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 15 ml syrup contains 200 mg guaifenesin. Each ml syrup
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Magnesium Trisilicate Mixture B.P. 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Magnesium Trisilicate B.P. 250 mg/5 ml Light Magnesium
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Magnesium Trisilicate Mixture B.P. 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Magnesium Trisilicate B.P. 250 mg/5 ml Light Magnesium
1. NAME OF THE MEDICINAL PRODUCT. Vicks Sinex, 0.5 mg/ml, nasal spray solution 2. QUALITATIVE AND QUANTITATIVE COMPOSITION
 1. NAME OF THE MEDICINAL PRODUCT Vicks Sinex, 0.5 mg/ml, nasal spray solution 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Oxymetazoline hydrochloride 0.5 mg/ml 1 spray (50 l) contains approximately 25
1. NAME OF THE MEDICINAL PRODUCT Vicks Sinex, 0.5 mg/ml, nasal spray solution 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Oxymetazoline hydrochloride 0.5 mg/ml 1 spray (50 l) contains approximately 25
למידע נוסף ניתן לעיין בעלון לרופא באתר משרד הבריאות או ליצור קשר טלפוני בטלפון
 חברת פרמה מדיס שמחה לבשר על תחילת שיווקו של תכשיר חדש. שם התכשיר: NEOSTIGMINE - HAMELN 2.5 MG/ML INJECTION צורת מינון: SOLUTION FOR INJECTION דרך מתן: I.M, I.V, S.C הרכב )חומר פעיל(: NEOSTIGMINE METHYLSULFATE
חברת פרמה מדיס שמחה לבשר על תחילת שיווקו של תכשיר חדש. שם התכשיר: NEOSTIGMINE - HAMELN 2.5 MG/ML INJECTION צורת מינון: SOLUTION FOR INJECTION דרך מתן: I.M, I.V, S.C הרכב )חומר פעיל(: NEOSTIGMINE METHYLSULFATE
1.3.1 SPC, Labelling and Package Leaflet (Voluven Fresenius 6% Solution for Infusion)
 1.3.1 SPC, Labelling and Package Leaflet (Voluven Fresenius 6% Solution for Infusion) 1.3.1 SUMMARY OF PRODUCT CHARACTERISTICS This medicinal product is subject to additional monitoring. This will allow
1.3.1 SPC, Labelling and Package Leaflet (Voluven Fresenius 6% Solution for Infusion) 1.3.1 SUMMARY OF PRODUCT CHARACTERISTICS This medicinal product is subject to additional monitoring. This will allow
This vaccine is indicated for the prevention of poliomyelitis in infants, children and adults, for primary and booster vaccinations.
 NAME OF THE MEDICINAL PRODUCT IMOVAX POLIO, suspension for injection in a prefilled syringe or multidose Poliomyelitis vaccine (inactivated) 2. QUALITATIVE AND QUANTITATIVE COMPOSITION One dose (0.5 ml)
NAME OF THE MEDICINAL PRODUCT IMOVAX POLIO, suspension for injection in a prefilled syringe or multidose Poliomyelitis vaccine (inactivated) 2. QUALITATIVE AND QUANTITATIVE COMPOSITION One dose (0.5 ml)
NEW ZEALAND DATASHEET
 NEW ZEALAND DATASHEET COLDREX HOT REMEDY COLD & FLU HOT LEMON Powder for Oral Solution Paracetamol (BP) 1000mg/sachet Presentation Pale yellow, free flowing heterogeneous powder with and odour of lemon
NEW ZEALAND DATASHEET COLDREX HOT REMEDY COLD & FLU HOT LEMON Powder for Oral Solution Paracetamol (BP) 1000mg/sachet Presentation Pale yellow, free flowing heterogeneous powder with and odour of lemon
