MIVACRON GlaxoSmithKline
|
|
|
- Abner Garey Andrews
- 5 years ago
- Views:
Transcription
1 MIVACRON GlaxoSmithKline Mivacurium QUALITATIVE AND QUANTITATIVE COMPOSITION Injection: Sterile solution containing 2 mg mivacurium per ml as mivacurium chloride, without an antimicrobial preservative, supplied in ampoules. Multi-dose vial: Injections containing 2 mg mivacurium per ml as mivacurium chloride with 0.9% w/v benzyl alcohol as an antimicrobial preservative, supplied in vials. PHARMACEUTICAL FORM Solution for injection or infusion. CLINICAL PARTICULARS Indications MIVACRON is a highly selective, short-acting, nondepolarising neuromuscular blocking agent with a fast recovery profile. MIVACRON is used as an adjunct to general anaesthesia to relax skeletal muscles and to facilitate tracheal intubation and mechanical ventilation. The injection formulation contains no antimicrobial preservative and is intended for single patient use. The multi-dose vial contains 0.9% w/v benzyl alcohol as an antimicrobial preservative and is intended for multiple use in one or more patients. Dosage and Administration IN COMMON WITH ALL NEUROMUSCULAR BLOCKING AGENTS, MONITORING OF NEURO- MUSCULAR FUNCTION IS RECOMMENDED DURING THE USE OF MIVACRON IN ORDER TO INDIVIDUALISE DOSAGE REQUIREMENTS. With MIVACRON, significant train-of-four fade is not seen during onset. It is often possible to intubate the trachea before complete abolition of the trainof-four response of the adductor pollicis muscle has occurred. Adults MIVACRON is administered by i.v. injection. The mean dose required to produce 95% suppression of the adductor pollicis single twitch response to ulnar nerve stimulation (ED95) is 0.07 mg/kg (range 0.06 to 0.09) in adults receiving narcotic anaesthesia. The following dose regimens are recommended for tracheal intubation: I. A dose of 0.2 mg/kg, administered over 30 seconds, produces good to excellent conditions for tracheal intubation within 2.0 to 2.5 min. II. A dose of 0.25 mg/kg, administered as a divided dose (0.15 mg/kg followed 30 seconds later by 0.1 mg/kg), produces good to excellent conditions for tracheal intubation within 1.5 to 2.0 min of completion of administration of the first dose portion. The recommended bolus dose range for healthy adults is 0.07 to 0.25 mg/kg. The duration of neuromuscular block is related to the dose. Doses of 0.07, 0.15, 0.20 and 0.25 mg/kg produce clinically effective block for approximately 13, 16, 20 and 23 min, respectively. Doses of up to 0.15 mg/kg may be administered over 5 to 15 seconds. Higher doses should be administered over 30 seconds in order to minimise the possibility of occurrence of cardiovascular effects. Full block can be prolonged with maintenance doses of MIVACRON. Doses of 0.1 mg/kg administered during narcotic anaesthesia each provide approximately 15 min of additional clinically effective block. Successive supplementary doses do not give rise to accumulation of neuromuscular blocking effect. The neuromuscular blocking action of MIVACRON is potentiated by isoflurane or enflurane anaesthesia. If steady-state anaesthesia with isoflurane or enflurane has been established, the recommended initial dose of MIVACRON should be reduced by up to 25%. Halothane appears to have only a minimal potentiating effect on MIVACRON and dose reduction is probably not necessary.
2 Once spontaneous recovery is underway, it is complete in approximately 15 min and is independent of the dose administered. The neuromuscular block produced by MIVACRON can be rapidly reversed with standard doses of anticholinesterase agents. However, because spontaneous recovery after MIVACRON is rapid, reversal may not be routinely required since it shortens recovery time by only 5 to 6 min. Use by infusion (Injection formulation only): Continuous infusion of MIVACRON may be used to maintain neuromuscular block. Upon early evidence of spontaneous recovery from an initial MIVACRON dose, an infusion rate of 8 to 10 micrograms/kg/min (0.5 to 0.6 mg/kg/h) is recommended. The initial infusion rate should be adjusted according to the patient s response to peripheral nerve stimulation and clinical criteria. Adjustments of the infusion rate should be made in increments of approximately 1 microgram/kg/min (0.06 mg/kg/h). In general a given rate should be maintained for at least 3 min before a rate change is made. On average, an infusion rate of 6 to 7 micrograms/ kg/min will maintain neuromuscular block within the range of 89% to 99% for extended periods in adults receiving narcotic anaesthesia. During steady-state isoflurane or enflurane anaesthesia, reduction in the infusion rate by up to 40% should be considered. A study has shown that the MIVACRON infusion rate requirement should be reduced by up to 50% with sevoflurane. With halothane, smaller reductions in infusion rate may be required. Spontaneous recovery after infusion of MIVACRON is independent of the duration of infusion and comparable to recovery reported for single doses. Continuous infusion of MIVACRON has not been associated with the development of tachyphylaxis or cumulative neuromuscular blockade. MIVACRON injection (2 mg/ml) may be used undiluted for infusion. It is compatible with the following infusion fluids: - sodium chloride i.v. infusion (0.9% w/v) - glucose i.v. infusion (5% w/v) - sodium chloride (0.18% w/v) and glucose (4% w/v) i.v. infusion - lactated Ringer s injection, USP. When diluted with the listed infusion solutions in the proportion of 1 plus 3 (i.e. to give 0.5 mg/ml) MIVACRON injection has been shown to be chemically and physically stable for at least 48 h at 30oC. However, since the product contains no antimicrobial preservative dilution should be carried out immediately prior to use, administration should commence as soon as possible thereafter and any remaining solution should be discarded. Children aged 7 months to 12 years MIVACRON has a higher ED95 (approximately 0.1 mg/kg), faster onset, shorter clinically effective duration of action and more rapid spontaneous recovery in infants and children aged 7 months to 12 years, than in adults. The recommended bolus dose range for infants and children aged 7 months to 12 years is 0.1 to 0.2 mg/ kg administered over 5 to 15 seconds. When administered during stable narcotic or halothane anaesthesia a dose of 0.2 mg/kg produces clinically effective block for an average of 9 min. A MIVACRON dose of 0.2 mg/kg is recommended for tracheal intubation in infants and children aged 7 months to 12 years. Maximum block is usually achieved within 2 min following administration of this dose and intubation should be possible within this time. Maintenance doses are generally required more frequently in infants and children than in adults. Available data suggest that a maintenance dose of 0.1 mg/kg will give approximately 6 to 9 min of additional clinically effective block during narcotic or halothane anaesthesia. Once spontaneous recovery is underway, it is complete in approximately 10 min. Use by infusion (Injection formulation only): Infants and children generally require higher infusion rates than adults. During halothane anaesthesia, the
3 mean infusion rate required to maintain 89 to 99% neuromuscular block in patients aged 7 to 23 months is approximately 11 micrograms/kg/min (approximately 0.7 mg/kg/h) [range 3 to 26 micrograms/kg/min (approximately 0.2 to 1.6 mg/kg/h)]. For children aged 2 to 12 years the equivalent mean infusion rate is approximately 13 to 14 micrograms/ kg/min (approximately 0.8 mg/kg/h) [range 5 to 31 micrograms/kg/min (approximately 0.3 to 1.9 mg/ kg/h)] under halothane or narcotic anaesthesia. The neuromuscular blocking action of MIVACRON is potentiated by inhalational agents. A study has shown that the MIVACRON infusion rate requirement should be reduced by up to 70% with sevoflurane in children aged 2 to 12 years. Children aged 2 to 6 months MIVACRON has a similar ED95 to that in adults (0.07 mg/kg), but a faster onset, shorter clinically effective duration and more rapid spontaneous recovery in infants aged 2 to 6 months than in adults. The recommended bolus dose range for infants aged 2 to 6 months is 0.1 to 0.15 mg/kg administered over 5 to 15 seconds. When administered during stable halothane anaesthesia a dose of 0.15 mg/ kg produces clinically effective block for an average of 9 min. A MIVACRON dose of 0.15 mg/kg is recommended for tracheal intubation in infants aged 2 to 6 months. Maximum block is achieved approximately 1.4 min following administration of this dose and intubation should be possible within this time. Maintenance doses are generally required more frequently in infants aged 2 to 6 months than in adults. Available data suggest that a maintenance dose of 0.1 mg/kg will give approximately 7 min of additional clinically effective block during halothane anaesthesia. Once spontaneous recovery is underway, it is complete in approximately 10 min. Use by infusion (Injection formulation only): Infants aged 2 to 6 months generally require higher infusion rates than adults. During halothane anaesthesia the mean infusion rate required to maintain 89 to 99% neuromuscular block is approximately 11 micrograms/kg/min (approximately 0.7 mg/kg/h) [range 4 to 24 micrograms/kg/min (approximately 0.2 to 1.5 mg/kg/h)]. Neonates and infants under two months of age No dose recommendations for neonates and infants under two months of age can be made until further information becomes available. Elderly In elderly patients receiving single bolus doses of MIVACRON, the onset time, duration of action and recovery rate may be extended relative to younger patients by 20 to 30%. Elderly patients may also require smaller or less frequent maintenance bolus doses. Use by infusion (Injection formulation only): Elderly patients may also require decreased infusion rates. Patients with cardiovascular disease In patients with clinically significant cardiovascular disease, the initial dose of MIVACRON should be administered over 60 seconds. MIVACRON has been administered in this way with minimal haemodynamic effects to patients undergoing cardiac surgery. Patients with reduced renal function In patients with end-stage renal failure the clinically effective duration of block produced by 0.15 mg/kg MIVACRON is approximately 1.5 times longer than in patients with normal renal function. Subsequently, dosage should be adjusted according to individual clinical response. Patients with reduced hepatic function In patients with end-stage hepatic failure the clinically effective duration of block produced by 0.15 mg/kg MIVACRON is approximately three times longer than in patients with normal hepatic function. This prolongation is related to the markedly reduced plasma cholinesterase activity seen in these patients. Subsequently, dosage should be adjusted according to individual clinical response.
4 Patients with reduced plasma cholinesterase activity MIVACRON is metabolized by plasma cholinesterase. Plasma cholinesterase activity may be diminished in the presence of genetic abnormalities of plasma cholinesterase (e.g. patients heterozygous or homozygous for the atypical plasma cholinesterase gene), and in various pathologic conditions (see Dosage and Administration: Patients with reduced hepatic function) and by administration of certain drugs (see Interactions). The possibility of prolonged neuromuscular block following administration of MIVACRON must be considered in patients with reduced plasma cholinesterase activity. Mild reductions (i.e. within 20% of the lower limit of the normal range) are not associated with clinically significant effects on duration. In patients heterozygous for the atypical plasma cholinesterase gene, the clinically effective duration of block of MIVACRON 0.15 mg/kg is approximately 10 min longer than in control patients. Obese patients In obese patients (those weighing 30% or more above their ideal bodyweight for height), the initial dose of MIVACRON should be based upon ideal bodyweight and not actual bodyweight. Contraindications - MIVACRON is contraindicated in patients known to be homozygous for the atypical plasma cholinesterase gene (see Warnings and Precautions). - MIVACRON should not be administered to patients known to have a hypersensitivity to mivacurium or excipients. Warnings and Precautions IN COMMON WITH ALL THE OTHER - BLOCKING AGENTS, MIVACRON PARALYSES THE RESPI- RATORY MUSCLES AS WELL AS OTHER SKEL- ETAL MUSCLES BUT HAS NO EFFECT ON CON- SCIOUSNESS. MIVACRON SHOULD BE ADMIN- ISTERED ONLY BY OR UNDER THE CLOSE SUP- ERVISION OF AN EXPERIENCED ANAESTHETIST WITH ADEQUATE FACILITIES FOR ENDOTRACH- EAL INTUBATION AND ARTIFICIAL VENTILATION. In common with suxamethonium/succinylcholine, patients homozygous for the atypical plasma cholinesterase gene (1 in 2500 patients) are extremely sensitive to the neuromuscular blocking effect of MIVACRON. In three such adult patients, a small dose of MIVACRON 0.03 mg/kg (approximately the ED10-20 in genotypically normal patients), produced complete neuromuscular block for 26 to 128 min. Once spontaneous recovery had begun, neuromuscular block in these patients was antagonized with conventional doses of neostigmine. Caution should be exercised in administering MIVACRON to patients with a history suggestive of an increased sensitivity to the effects of histamine, e.g. asthma. If MIVACRON is used in this group of patients it should be administered over 60 seconds. Caution should also be exercised when administering MIVACRON to patients who have shown hypersensitivity to other neuromuscular blocking agents since cross-sensitivity between neuromuscular blocking agents has been reported. MIVACRON should be administered over a period of 60 seconds to patients who may be unusually sensitive to falls in arterial blood pressure, for example those who are hypovolaemic. In adults, doses of MIVACRON of greater than or equal to 0.2 mg/kg (greater than or equal to 3x ED95) have been associated with histamine release when administered by rapid bolus injection. However, the slower administration of the 0.2 mg/ kg MIVACRON dose and the divided administration of the 0.25 mg/kg MIVACRON dose (see Dosage and Administration) minimise the cardiovascular effects of these doses. Cardiovascular safety did not appear to be compromised in children given a rapid bolus dose of 0.2 mg/kg in clinical studies. MIVACRON does not have significant vagal or ganglion blocking properties in the recommended dosage range. Recommended doses of MIVACRON consequently have no clinically significant effects on heart rate and will not counteract the bradycardia produced by many anaesthetic agents or by vagal stimulation during surgery.
5 In common with other non-depolarising neuromuscular blocking agents, increased sensitivity to MIVACRON can be expected in patients with myasthenia gravis, other forms of neuromuscular disease and cachectic patients. Severe acid-base or electrolyte abnormalities may increase or reduce sensitivity to MIVACRON. MIVACRON solution is acidic (approximately ph 4.5) and should not be mixed in the same syringe or administered simultaneously through the same needle with highly alkaline solutions (e.g. barbiturate solutions). It has been shown to be compatible with some commonly used peri-operative drugs supplied as acidic solutions, e.g. fentanyl, alfentanil, sufentanil, droperidol and midazolam. Where other anaesthetic agents are administered through the same indwelling needle or cannula as used for MIVACRON, and compatibility has not been demonstrated, it is recommended that each drug is flushed through with physiological saline. Patients with burns may develop resistance to nondepolarising neuromuscular blocking agents and require increased doses. However, such patients may also have reduced plasma cholinesterase activity, requiring dose reduction. Consequently, burn patients should be given a test dose of to mg/kg MIVACRON followed by appropriate dosing guided by monitoring of block with a nerve stimulator. Studies in malignant hyperthermia-susceptible pigs indicated that MIVACRON does not trigger this syndrome. MIVACRON has not been studied in malignant hyperthermia-susceptible patients. Reversal of neuromuscular block: as with other neuromuscular blocking agents, evidence of spontaneous recovery should be observed prior to administration of reversal agent (e.g. neostigmine). The use of a peripheral nerve stimulator to evaluate recovery prior to and following reversal of neuromuscular block is strongly recommended. No data are available on the long-term use of MIVACRON injection in patients undergoing mechanical ventilation in the intensive care unit. Benzyl alcohol is used as an antimicrobial preservative in many parenteral drug formulations, including MIVACRON multi-dose vial. Theoretically, certain patient groups, e.g. those with liver failure, may possess a reduced ability to metabolise benzyl alcohol. There have been reports linking the use of parenteral drug formulations containing benzyl alcohol to morbidity and mortality amongst low weight neonates. Interactions The neuromuscular block produced by MIVACRON may be potentiated by the concomitant use of inhalational anaesthetics such as enflurane, isoflurane, sevoflurane and halothane. MIVACRON has been safely administered following suxamethonium-facilitated tracheal intubation. Evidence of spontaneous recovery from suxamethonium should be observed prior to administration of MIVACRON. In common with all non-depolarising neuromuscular blocking agents the magnitude and/or duration of a non-depolarising neuromuscular block may be increased and maintenance requirements may be reduced as a result of interaction with: - antibiotics: including the aminoglycosides, polymyxins, spectinomycin, tetracyclines, lincomycin and clindamycin - anti-arrhythmic drugs: propranolol, calcium channel blockers, lignocaine, procainamide and quinidine - diuretics: frusemide and possibly thiazides, mannitol and acetazolamide - magnesium salts - ketamine - lithium salts - ganglion blocking drugs: trimetaphan, hexamethonium. Drugs that may reduce plasma cholinesterase activity may also prolong the neuromuscular blocking action of MIVACRON. These include anti-mitotic drugs, monoamine oxidase inhibitors, ecothiopate iodide, pancuronium, organophosphates, anticholinesterases, certain hormones, bambuterol. Rarely, certain drugs may aggravate or unmask latent myasthenia gravis or actually induce a
6 myasthenic syndrome; increased sensitivity to MIVACRON would be consequent on such a development. Such drugs include various antibiotics, beta-blockers (propranolol, oxprenolol), antiarrhythmic drugs (procainamide, quinidine), antirheumatic drugs (chloroquine, D-pencillamine), trimetaphan, chlorpromazine, steroids, phenytoin and lithium. The administration of combinations of non-depolarising neuromuscular blocking agents in conjunction with MIVACRON may produce a degree of neuromuscular blockade in excess of that which might be expected from an equipotent total dose of MIVACRON. Any synergistic effect may vary between different drug combinations. A depolarising muscle relaxant such as suxamethonium chloride should not be administered to prolong the neuromuscular blocking effects of non-depolarising agents, as this may result in a prolonged and complex block which can be difficult to reverse with anti-cholinesterase drugs. Pregnancy and Lactation Fertility studies have not been performed. Animal studies have indicated that mivacurium has no adverse effect on foetal development. There is no information on the use of MIVACRON in pregnant women therefore MIVACRON should not be used during pregnancy unless the expected clinical benefit to the mother outweighs any potential risk to the foetus. Plasma cholinesterase levels decrease during pregnancy. MIVACRON injection has been used to maintain neuromuscular block during Caesarean section, but due to the reduced levels of plasma cholinesterase, dosage adjustments to the infusion rate were necessary. A further reduction in the infusion rate may also be required during Caesarean section in patients pretreated with MgSO4, due to the potentiating effects of Mg2+. There has been no experience of MIVACRON multi-dose vials during Caesarean section. It is not known whether mivacurium is excreted in human milk. Effects on Ability to Drive and Use Machines No data. Adverse Reactions Adverse events are listed below by system organ class and frequency. Frequencies are defined as: very common: 1 in 10 common: 1 in 100 and <1 in 10 uncommon: 1 in 1,000 and <1 in 100 rare: 1 in 10,000 and <1 in 1,000 very rare: <1/10,000 including isolated reports. Immune disorders Very rare: Severe anaphylactic or anaphylactoid reactions have been reported in patients receiving MIVACRON chloride in conjunction with one or more anaesthetic agents. Cardiac disorders Uncommon: Transient tachycardia Vascular disorders Very common: Flushing Uncommon: Hypotension Respiratory, thoracic and mediastinal disorders Uncommon: Bronchospasm Skin and subcutaneous tissue disorders Uncommon: Erythema, urticaria Associated with the use of MIVACRON there have been reports of skin flushing, erythema, urticaria, hypotension, transient tachycardia or bronchospasm which have been attributed to histamine release. These effects are dose-related and more common following initial doses of 0.2 mg/kg or more when given rapidly and are reduced if MIVACRON is injected over 30 to 60 seconds or in divided doses over 30 seconds. Overdose Prolonged muscle paralysis and its consequences are the main effects of overdosage with neuromuscular blocking agents. However, the risk of haemodynamic side effects, especially decreases in blood pressure may be increased. It is essential to maintain a patent airway together with assisted positive pressure ventilation until spontaneous respiration is adequate.
7 Full sedation will be required since consciousness is not impaired. Recovery may be hastened by the administration of anticholinesterase agents accompanied by atropine or glycopyrrolate, once evidence of spontaneous recovery is present. Cardiovascular support may be provided by proper positioning of the patient and administration of fluids or vasopressor agents as required. PHARMACOLOGICAL PROPERTIES Pharmacodynamics Mivacurium is a highly selective, short-acting, nondepolarising neuromuscular blocking agent with a fast recovery profile. MIVACRON is a mixture of three mivacurium stereoisomers. The trans-trans and cis- trans stereoisomers comprise 92% to 96% of MIVACRON and when studied in cats their neuromuscular blocking potencies are not significantly different from each other or from mivacurium chloride. The cis-cis isomer has been estimated from studies in cats to have one-tenth of the neuromuscular blocking potency of the other two stereoisomers. Pharmacokinetics Enzymatic hydrolysis by plasma cholinesterase is the primary mechanism for inactivation of mivacurium and yields a quaternary alcohol and a quaternary monoester metabolite. Pharmacological studies in cats and dogs have shown that the metabolites possess insignificant neuromuscular, autonomic or cardiovascular activity at concentrations higher than seen in man. The termination of the neuromuscular blocking action of MIVACRON is mainly dependent on hydrolysis by plasma pseudocholinesterase, which is present at high levels in human plasma. Multiple degradation/elimination pathways appear to exist for mivacurium (e.g. hydrolysis by liver esterases, elimination in bile and renal excretion). Pre-clinical Safety Data Mivacurium has been evaluated in four short-term mutagenicity tests. Mivacurium was non-mutagenic in the Ames Salmonella assay, the mouse lymphoma assay, the human lymphocyte assay and the in vivo rat bone marrow cytogenetic assay. There is no information available on whether mivacurium has carcinogenic potential. PHARMACEUTICAL PARTICULARS List of Excipients MIVACRON multi-dose vial contains benzyl alcohol. Incompatibilities MIVACRON is acidic (approximately ph 4.5) and should not be mixed with highly alkaline solutions, e.g. barbiturates. Shelf Life The expiry date is indicated on the packaging. Special Precautions for Storage Injection: Store below 25oC. Protect from light. Do not freeze. Multi-dose vial: As registered locally. Nature and Contents of Container As registered locally. Instructions for Use/Handling MIVACRON injection has been shown to be compatible with some commonly used peri- operative drugs supplied as acidic solutions. Where such agents are administered through the same indwelling needle or cannula as used for MIVACRON injection, and compatibility has not been demonstrated, it is recommended that each drug is flushed through with physiological saline. Injection: Since no antimicrobial preservative is included, MIVACRON injection must be used under full aseptic conditions and any dilution carried out immediately before use. Any unused solution in open ampoules should be discarded. MIVACRON injection is compatible with the following infusion fluids:
8 - sodium chloride i.v. infusion (0.9% w/v) - glucose i.v. infusion (5% w/v) - sodium chloride (0.18% w/v) and glucose (4% w/v) i.v. infusion - lactated Ringer s injection, USP. When diluted with the listed infusion solutions in the proportion of 1 plus 3 (i.e. to give 0.5 mg/ml) MIVACRON injection has been shown to be chemically and physically stable for at least 48 h at 30oC. However, since the product contains no antimicrobial preservative dilution should be carried out immediately prior to use, administration should commence as soon as possible thereafter and any remaining solution should be discarded. Multi-dose vial: MIVACRON Multi-dose vials contain 0.9% w/v benzyl alcohol as an antimicrobial preservative and is intended for use in one or more patients. It is good clinical practice to discard any partly used vials at the end of an operating day. Not all presentations are available in every country. Version number: GDS09/IPI02 Date of issue: 22 April 2005 MIVACRON is a trademark of the GlaxoSmithKline group of companies
TRACRIUM GlaxoSmithKline
 TRACRIUM GlaxoSmithKline Atracurium QUALITATIVE AND QUANTITATIVE COMPOSITION Injection: A sterile solution containing 10 mg atracurium besylate per ml, without an antimicrobial preservative, supplied in
TRACRIUM GlaxoSmithKline Atracurium QUALITATIVE AND QUANTITATIVE COMPOSITION Injection: A sterile solution containing 10 mg atracurium besylate per ml, without an antimicrobial preservative, supplied in
For the use only of Registered Medical Practitioners or a Hospital or a Laboratory TRACRIUM. Atracurium Besylate Injection IP
 For the use only of Registered Medical Practitioners or a Hospital or a Laboratory TRACRIUM Atracurium Besylate Injection IP QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml contains Atracurium Besylate
For the use only of Registered Medical Practitioners or a Hospital or a Laboratory TRACRIUM Atracurium Besylate Injection IP QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml contains Atracurium Besylate
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT ACURMIL 25 mg/2.5 ml solution for intravenous injection ACURMIL 50 mg/5 ml solution for intravenous injection 2. QUALITATIVE AND QUANTITATIVE
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT ACURMIL 25 mg/2.5 ml solution for intravenous injection ACURMIL 50 mg/5 ml solution for intravenous injection 2. QUALITATIVE AND QUANTITATIVE
PRODUCT INFORMATION. TRACRIUM INJECTION atracurium besylate DESCRIPTION:
 PRODUCT INFORMATION TRACRIUM INJECTION (atracurium besylate) NAME OF THE DRUG: atracurium besylate DESCRIPTION: TRACRIUM (atracurium besylate) is an intermediate-duration, non-depolarising, skeletal muscle
PRODUCT INFORMATION TRACRIUM INJECTION (atracurium besylate) NAME OF THE DRUG: atracurium besylate DESCRIPTION: TRACRIUM (atracurium besylate) is an intermediate-duration, non-depolarising, skeletal muscle
PRODUCT INFORMATION CISATRACURIUM PFIZER NAME OF THE MEDICINE DESCRIPTION PHARMACOLOGY. Cisatracurium besylate
 PRODUCT INFORMATION CISATRACURIUM PFIZER Cisatracurium besylate NAME OF THE MEDICINE The chemical name of cisatracurium besylate is (1R,1 R,2R,2 R,)-2,2 -(3,11-dioxo-4,10- dioxatridecamethylene) bis (1,2,3,4-tetrahydro-6,7-dimethoxy-2-methyl-1-
PRODUCT INFORMATION CISATRACURIUM PFIZER Cisatracurium besylate NAME OF THE MEDICINE The chemical name of cisatracurium besylate is (1R,1 R,2R,2 R,)-2,2 -(3,11-dioxo-4,10- dioxatridecamethylene) bis (1,2,3,4-tetrahydro-6,7-dimethoxy-2-methyl-1-
A Nondepolarizing Neuromuscular Blocking (NMB) Agent
 DOSING GUIDE A Nondepolarizing Neuromuscular Blocking (NMB) Agent Easy to remember dosing for the 0.20 mg/kg adult intubating doses of NIMBEX 1 *: For every 10 kg, give 1 ml of NIMBEX (2 mg/ml concentration)
DOSING GUIDE A Nondepolarizing Neuromuscular Blocking (NMB) Agent Easy to remember dosing for the 0.20 mg/kg adult intubating doses of NIMBEX 1 *: For every 10 kg, give 1 ml of NIMBEX (2 mg/ml concentration)
This drug should be administered only by adequately trained individuals familiar with its actions, characteristics, and hazards.
 MIVACRON Injection (mivacurium chloride) This drug should be administered only by adequately trained individuals familiar with its actions, characteristics, and hazards. DESCRIPTION MIVACRON (mivacurium
MIVACRON Injection (mivacurium chloride) This drug should be administered only by adequately trained individuals familiar with its actions, characteristics, and hazards. DESCRIPTION MIVACRON (mivacurium
ULTIVA GlaxoSmithKline
 ULTIVA GlaxoSmithKline Remifentanil QUALITATIVE AND QUANTITATIVE COMPOSITION Remifentanil for injection is a sterile, endotoxin-free, preservative-free, white to off white, lyophilised powder, to be reconstituted
ULTIVA GlaxoSmithKline Remifentanil QUALITATIVE AND QUANTITATIVE COMPOSITION Remifentanil for injection is a sterile, endotoxin-free, preservative-free, white to off white, lyophilised powder, to be reconstituted
PACKAGE LEAFLET: INFORMATION FOR THE USER. Atracurium Besilate 10 mg/ml Solution for Injection The active substance is atracurium besilate
 PACKAGE LEAFLET: INFORMATION FOR THE USER Atracurium Besilate 10 mg/ml Solution for Injection The active substance is atracurium besilate Read all of this leaflet carefully before you start using this
PACKAGE LEAFLET: INFORMATION FOR THE USER Atracurium Besilate 10 mg/ml Solution for Injection The active substance is atracurium besilate Read all of this leaflet carefully before you start using this
Core Safety Profile. Pharmaceutical form(s)/strength: Solution for Injection CZ/H/PSUR/0005/002 Date of FAR:
 Core Safety Profile Active substance: Rocuronium bromide Pharmaceutical form(s)/strength: Solution for Injection P-RMS: CZ/H/PSUR/0005/002 Date of FAR: 31.05.2012 4.3 Contraindications Hypersensitivity
Core Safety Profile Active substance: Rocuronium bromide Pharmaceutical form(s)/strength: Solution for Injection P-RMS: CZ/H/PSUR/0005/002 Date of FAR: 31.05.2012 4.3 Contraindications Hypersensitivity
Please refer to the Summary of Product Characteristics (SPC) for further details on this product.
 [Aspen Logo] Information for the Physician Nimbex cisatracurium Please refer to the Summary of Product Characteristics (SPC) for further details on this product. Trade name of the medicinal product Nimbex
[Aspen Logo] Information for the Physician Nimbex cisatracurium Please refer to the Summary of Product Characteristics (SPC) for further details on this product. Trade name of the medicinal product Nimbex
ULTIVA. Remifentanil hydrochloride
 ULTIVA Remifentanil hydrochloride QUALITATIVE AND QUANTITATIVE COMPOSITION Remifentanil for injection is a sterile, preservative-free, white to off white, lyophilised powder, to be reconstituted before
ULTIVA Remifentanil hydrochloride QUALITATIVE AND QUANTITATIVE COMPOSITION Remifentanil for injection is a sterile, preservative-free, white to off white, lyophilised powder, to be reconstituted before
LACIPIL QUALITATIVE AND QUANTITATIVE COMPOSITION
 LACIPIL lacidipine QUALITATIVE AND QUANTITATIVE COMPOSITION Lacidipine, 2 mg - round shaped white engraved on one face. Lacidipine, 4 mg - oval white with break line on both faces. Lacidipine, 6 mg - oval
LACIPIL lacidipine QUALITATIVE AND QUANTITATIVE COMPOSITION Lacidipine, 2 mg - round shaped white engraved on one face. Lacidipine, 4 mg - oval white with break line on both faces. Lacidipine, 6 mg - oval
PACKAGE LEAFLET: INFORMATION FOR THE USER. Cisatracurium 2 mg/ml Solution for Injection/Infusion Cisatracurium 5 mg/ml Solution for Injection/Infusion
 PACKAGE LEAFLET: INFORMATION FOR THE USER Cisatracurium 2 mg/ml Solution for Injection/Infusion Cisatracurium 5 mg/ml Solution for Injection/Infusion Cisatracurium Read all of this leaflet carefully before
PACKAGE LEAFLET: INFORMATION FOR THE USER Cisatracurium 2 mg/ml Solution for Injection/Infusion Cisatracurium 5 mg/ml Solution for Injection/Infusion Cisatracurium Read all of this leaflet carefully before
NEW ZEALAND DATA SHEET. Injection 2 mg/ml: a clear, colourless, particle-free solution containing 2 mg/ml pancuronium bromide.
 NEW ZEALAND DATA SHEET NAME OF MEDICINE Pancuronium Bromide B.P. Injection 2 mg/ml PRESENTATION Injection 2 mg/ml: a clear, colourless, particle-free solution containing 2 mg/ml pancuronium bromide. INDICATIONS
NEW ZEALAND DATA SHEET NAME OF MEDICINE Pancuronium Bromide B.P. Injection 2 mg/ml PRESENTATION Injection 2 mg/ml: a clear, colourless, particle-free solution containing 2 mg/ml pancuronium bromide. INDICATIONS
PRODUCT MONOGRAPH. Solution for Injection 20 mg/10 ml (2 mg/ml) Cisatracurium. Non-depolarizing Skeletal Neuromuscular Blocking Agent
 PRODUCT MONOGRAPH Pr CISATRACURIUM BESYLATE INJECTION Solution for Injection 20 mg/10 ml (2 mg/ml) Cisatracurium Non-depolarizing Skeletal Neuromuscular Blocking Agent This drug should be administered
PRODUCT MONOGRAPH Pr CISATRACURIUM BESYLATE INJECTION Solution for Injection 20 mg/10 ml (2 mg/ml) Cisatracurium Non-depolarizing Skeletal Neuromuscular Blocking Agent This drug should be administered
200 mg/20 ml (10 mg/ml) in single-dose vials (3)
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use NIMBEX safely and effectively. See full prescribing information for NIMBEX. NIMBEX (cisatracurium
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use NIMBEX safely and effectively. See full prescribing information for NIMBEX. NIMBEX (cisatracurium
NEW ZEALAND DATA SHEET. Injection solution 2.5 mg/ml: a clear, colourless, particle-free solution containing 2.5 mg/ml Neostigmine methylsulphate.
 NEW ZEALAND DATA SHEET NAME OF MEDICINE NEOSTIGMINE METHYLSULPHATE INJECTION B.P. Solution for injection 2.5 mg/ml. PRESENTATIONS Injection solution 2.5 mg/ml: a clear, colourless, particle-free solution
NEW ZEALAND DATA SHEET NAME OF MEDICINE NEOSTIGMINE METHYLSULPHATE INJECTION B.P. Solution for injection 2.5 mg/ml. PRESENTATIONS Injection solution 2.5 mg/ml: a clear, colourless, particle-free solution
Package leaflet: Information for the user
 Package leaflet: Information for the user Cisatracurium 2 mg/ml Solution for injection/ infusion Cisatracurium 5 mg/ml Solution for injection/ infusion Cisatracurium The name of your medicine are Cisatracurium
Package leaflet: Information for the user Cisatracurium 2 mg/ml Solution for injection/ infusion Cisatracurium 5 mg/ml Solution for injection/ infusion Cisatracurium The name of your medicine are Cisatracurium
PACKAGE LEAFLET: INFORMATION FOR THE USER. Cisatracurium 2 mg/ml solution for injection/infusion Cisatracurium 5 mg/ml solution for injection/infusion
 PACKAGE LEAFLET: INFORMATION FOR THE USER Cisatracurium 2 mg/ml solution for injection/infusion Cisatracurium 5 mg/ml solution for injection/infusion Cisatracurium Read all of this leaflet carefully before
PACKAGE LEAFLET: INFORMATION FOR THE USER Cisatracurium 2 mg/ml solution for injection/infusion Cisatracurium 5 mg/ml solution for injection/infusion Cisatracurium Read all of this leaflet carefully before
NEW ZEALAND DATA SHEET
 NEW ZEALAND DATA SHEET VECURE 1. Product Name VECURE 10 mg, powder for injection. 2. Qualitative and Quantitative Composition VECURE 10 mg, 1 vial contains 10 mg vecuronium bromide. When reconstituted
NEW ZEALAND DATA SHEET VECURE 1. Product Name VECURE 10 mg, powder for injection. 2. Qualitative and Quantitative Composition VECURE 10 mg, 1 vial contains 10 mg vecuronium bromide. When reconstituted
New Zealand Data Sheet DBL TM Rocuronium Bromide Injection
 New Zealand Data Sheet DBL TM Rocuronium Bromide Injection NAME OF THE MEDICINE DBL TM Rocuronium Bromide Injection 50 mg/5 ml Vial PRESENTATION Rocuronium bromide is a quaternary aminosteroid and an analogue
New Zealand Data Sheet DBL TM Rocuronium Bromide Injection NAME OF THE MEDICINE DBL TM Rocuronium Bromide Injection 50 mg/5 ml Vial PRESENTATION Rocuronium bromide is a quaternary aminosteroid and an analogue
2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Rocuronium bromide 10 mg/ml solution for injection/ infusion 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml of solution of Rocuronium
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Rocuronium bromide 10 mg/ml solution for injection/ infusion 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml of solution of Rocuronium
DATA SHEET. Remifentanil hydrochloride for injection1mg and 2mg (remifentanil base) vials
 DATA SHEET 1. PRODUCT NAME ULTIVA Remifentanil hydrochloride for injection1mg and 2mg (remifentanil base) vials 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Active substance Remifentanil hydrochloride 1mg
DATA SHEET 1. PRODUCT NAME ULTIVA Remifentanil hydrochloride for injection1mg and 2mg (remifentanil base) vials 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Active substance Remifentanil hydrochloride 1mg
DBL NALOXONE HYDROCHLORIDE INJECTION USP
 Name of medicine Naloxone hydrochloride Data Sheet New Zealand DBL NALXNE HYDRCHLRIDE INJECTIN USP Presentation DBL Naloxone Hydrochloride Injection USP is a sterile, clear, colourless solution, free from
Name of medicine Naloxone hydrochloride Data Sheet New Zealand DBL NALXNE HYDRCHLRIDE INJECTIN USP Presentation DBL Naloxone Hydrochloride Injection USP is a sterile, clear, colourless solution, free from
SUMMARY OF PRODUCT CHARACTERISTICS. 1. NAME OF THE MEDICINAL PRODUCT Rocuronium Kabi 10 mg/ml solution for injection/infusion
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Rocuronium Kabi 10 mg/ml solution for injection/infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml of solution for injection
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Rocuronium Kabi 10 mg/ml solution for injection/infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml of solution for injection
VENTOLIN RESPIRATOR SOLUTION
 VENTOLIN RESPIRATOR SOLUTION Salbutamol QUALITATIVE AND QUANTITATIVE COMPOSITION VENTOLIN Respirator Solution contains 5mg salbutamol, as sulphate, per ml of solution and is supplied in 10 ml bottles.
VENTOLIN RESPIRATOR SOLUTION Salbutamol QUALITATIVE AND QUANTITATIVE COMPOSITION VENTOLIN Respirator Solution contains 5mg salbutamol, as sulphate, per ml of solution and is supplied in 10 ml bottles.
AUSTRALIAN PRODUCT INFORMATION REMIFENTANIL APOTEX (REMIFENTANIL HYDROCHLORIDE) POWDER FOR INJECTION
 AUSTRALIAN PRODUCT INFORMATION REMIFENTANIL APOTEX (REMIFENTANIL HYDROCHLORIDE) POWDER FOR INJECTION 1 NAME OF THE MEDICINE Remifentanil (as hydrochloride) 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each
AUSTRALIAN PRODUCT INFORMATION REMIFENTANIL APOTEX (REMIFENTANIL HYDROCHLORIDE) POWDER FOR INJECTION 1 NAME OF THE MEDICINE Remifentanil (as hydrochloride) 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each
08-15 NORCURON ORGANON
 08-15 NORCURON ORGANON 1. NAME OF THE MEDICINAL PRODUCT Norcuron 4 mg, powder for solution for injection Norcuron 10 mg, powder for solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Norcuron
08-15 NORCURON ORGANON 1. NAME OF THE MEDICINAL PRODUCT Norcuron 4 mg, powder for solution for injection Norcuron 10 mg, powder for solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Norcuron
FOR REPRESENTATIVE EDUCATION
 Neuromuscular Blockade in the ICU NIMBEX Indication 1 NIMBEX (cisatracurium besylate) is indicated as an adjunct to general anesthesia to facilitate tracheal intubation in adults and in pediatric patients
Neuromuscular Blockade in the ICU NIMBEX Indication 1 NIMBEX (cisatracurium besylate) is indicated as an adjunct to general anesthesia to facilitate tracheal intubation in adults and in pediatric patients
2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 AUSTRALIAN PRODUCT INFORMATION NEOSTIGMINE INJECTION BP (NEOSTIGMINE METHYLSULFATE) 1 NAME OF THE MEDICINE Neostigmine methylsulfate. 2 QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml sterile solution of
AUSTRALIAN PRODUCT INFORMATION NEOSTIGMINE INJECTION BP (NEOSTIGMINE METHYLSULFATE) 1 NAME OF THE MEDICINE Neostigmine methylsulfate. 2 QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml sterile solution of
Shaded areas=not MARKETED 24/2/09
 PACKAGE INSERT SCHEDULING STATUS Schedule 6 PROPRIETARY NAME AND DOSAGE FORM RAPIFEN 2 ml IV injection RAPIFEN 10 ml IV injection COMPOSITION Each ml contains alfentanil hydrochloride 0,544 mg (equivalent
PACKAGE INSERT SCHEDULING STATUS Schedule 6 PROPRIETARY NAME AND DOSAGE FORM RAPIFEN 2 ml IV injection RAPIFEN 10 ml IV injection COMPOSITION Each ml contains alfentanil hydrochloride 0,544 mg (equivalent
SUMMARY OF PRODUCT CHARACTERISTICS. 1 ml solution contains 75 micrograms of sufentanilcitrate, corresponding to 50 micrograms of sufentanil.
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Sufentanil Narcomed, 50 microgram / ml, solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml solution contains 75
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Sufentanil Narcomed, 50 microgram / ml, solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml solution contains 75
NEW ZEALAND DATA SHEET
 NEW ZEALAND DATA SHEET 1. PRODUCT NAME SUXAMETHONIUM CHLORIDE INJECTION B.P. 50 mg/ml 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each 2 ml of solution contains 100 mg of suxamethonium chloride BP For
NEW ZEALAND DATA SHEET 1. PRODUCT NAME SUXAMETHONIUM CHLORIDE INJECTION B.P. 50 mg/ml 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each 2 ml of solution contains 100 mg of suxamethonium chloride BP For
P-RMS: NO/H/PSUR/0009/001
 Core Safety Profile Active substance: Propofol Pharmaceutical form(s)/strength: Emulsion for injection, infusion, 10mg/ml Emulsion for infusion, 20mg/ml P-RMS: NO/H/PSUR/0009/001 Date of FAR: 30.06.2011
Core Safety Profile Active substance: Propofol Pharmaceutical form(s)/strength: Emulsion for injection, infusion, 10mg/ml Emulsion for infusion, 20mg/ml P-RMS: NO/H/PSUR/0009/001 Date of FAR: 30.06.2011
ZOFRAN INJECTION GlaxoSmithKline
 ZOFRAN INJECTION GlaxoSmithKline Ondansetron QUALITATIVE AND QUANTITATIVE COMPOSITION Each 1 ml of aqueous solution contains 2 mg ondansetron as hydrochloride dihydrate. PHARMACEUTICAL FORM A clear, colourless,
ZOFRAN INJECTION GlaxoSmithKline Ondansetron QUALITATIVE AND QUANTITATIVE COMPOSITION Each 1 ml of aqueous solution contains 2 mg ondansetron as hydrochloride dihydrate. PHARMACEUTICAL FORM A clear, colourless,
2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Citanest with Octapressin Dental 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml contains Prilocaine Hydrochloride 30 mg (54 mg/1.8
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Citanest with Octapressin Dental 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml contains Prilocaine Hydrochloride 30 mg (54 mg/1.8
A specific opioid antagonist, such as naloxone immediately and completely reverses all actions of alfentanil.
 RAPIFEN DATA SHEET NAME OF THE MEDICINE RAPIFEN alfentanil 0.5 mg/ml injection PRESENTATION RAPIFEN is a sterile, preservative free, isotonic, aqueous solution containing alfentanil hydrochloride equivalent
RAPIFEN DATA SHEET NAME OF THE MEDICINE RAPIFEN alfentanil 0.5 mg/ml injection PRESENTATION RAPIFEN is a sterile, preservative free, isotonic, aqueous solution containing alfentanil hydrochloride equivalent
2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 1 PRODUCT NAME TROPISETRON-AFT tropisetron hydrochloride (equivalent to 2 mg or 5 mg tropisetron) per ampoule. 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml contains 1 mg of tropisetron 1 2 ml ampoule
1 PRODUCT NAME TROPISETRON-AFT tropisetron hydrochloride (equivalent to 2 mg or 5 mg tropisetron) per ampoule. 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml contains 1 mg of tropisetron 1 2 ml ampoule
Suxamethonium Chloride Injection BP PRODUCT INFORMATION
 Suxamethonium Chloride Injection BP Product Information 1 (8) Suxamethonium Chloride Injection BP PRODUCT INFORMATION NAME OF DRUG The Australian Approved Name is suxamethonium chloride. The CAS number
Suxamethonium Chloride Injection BP Product Information 1 (8) Suxamethonium Chloride Injection BP PRODUCT INFORMATION NAME OF DRUG The Australian Approved Name is suxamethonium chloride. The CAS number
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS Page 1 / 6 1. NAME OF THE VETERINARY MEDICINAL PRODUCT Fentadon 50 microgram/ml, solution for injection for dogs SE, DK: Fentadon Vet. 50 microgram/ml, solution for injection
SUMMARY OF PRODUCT CHARACTERISTICS Page 1 / 6 1. NAME OF THE VETERINARY MEDICINAL PRODUCT Fentadon 50 microgram/ml, solution for injection for dogs SE, DK: Fentadon Vet. 50 microgram/ml, solution for injection
ACETYLCYSTEINE INJECTION
 ACETYLCYSTEINE INJECTION 1. Name of the medicinal product Acetylcysteine 200 mg/ml Injection 2. Qualitative and quantitative composition Acetylcysteine 200mg per ml (as N-acetylcysteine) Each 10ml ampoule
ACETYLCYSTEINE INJECTION 1. Name of the medicinal product Acetylcysteine 200 mg/ml Injection 2. Qualitative and quantitative composition Acetylcysteine 200mg per ml (as N-acetylcysteine) Each 10ml ampoule
Glycopyrronium Bromide and Neostigmine Metilsulfate 0.5mg/2.5mg per ml
 New Zealand Datasheet Glycopyrronium Bromide and Neostigmine Metilsulfate 0.5mg/2.5mg per ml Solution for injection Glycopyrronium bromide 0.5 mg/ml & Neostigmine metilsulfate 2.5 mg/ml Presentation is
New Zealand Datasheet Glycopyrronium Bromide and Neostigmine Metilsulfate 0.5mg/2.5mg per ml Solution for injection Glycopyrronium bromide 0.5 mg/ml & Neostigmine metilsulfate 2.5 mg/ml Presentation is
(PP XI) Dr. Samir Matloob
 DRUGS ACTING ON THE CHOLINERGIC SYSTEM AND THE NEUROMUSCULAR BLOCKING DRUGS IV (NICOTINIC ANTAGONISTS) (PP XI) Dr. Samir Matloob Dept. of Pharmacology Baghdad College of Medicine Drugs acting on the cholinergic
DRUGS ACTING ON THE CHOLINERGIC SYSTEM AND THE NEUROMUSCULAR BLOCKING DRUGS IV (NICOTINIC ANTAGONISTS) (PP XI) Dr. Samir Matloob Dept. of Pharmacology Baghdad College of Medicine Drugs acting on the cholinergic
Core Safety Profile. Date of FAR:
 Core Safety Profile Active substance: Levobupivicaine Pharmaceutical form(s)/strength: Solution for injection, concentrate for solution for infusion, 2,5 mg/ml, 5 mg/ml, 7,5 mg/ml, 0,625 mg/ml, 1,25 mg/ml
Core Safety Profile Active substance: Levobupivicaine Pharmaceutical form(s)/strength: Solution for injection, concentrate for solution for infusion, 2,5 mg/ml, 5 mg/ml, 7,5 mg/ml, 0,625 mg/ml, 1,25 mg/ml
RAPIFEN DATA SHEET 1. NAME OF THE MEDICINE 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 3. PHARMACEUTICAL FORM 4. CLINICAL PARTICULARS
 RAPIFEN DATA SHEET 1. NAME OF THE MEDICINE RAPIFEN alfentanil 0.5 mg/ml injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml of RAPIFEN contains alfentanil hydrochloride 544 micrograms, equivalent
RAPIFEN DATA SHEET 1. NAME OF THE MEDICINE RAPIFEN alfentanil 0.5 mg/ml injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml of RAPIFEN contains alfentanil hydrochloride 544 micrograms, equivalent
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE VETERINARY MEDICINAL PRODUCT Tralieve 50 mg/ml solution for injection for dogs (AT, BE, BG, CY, CZ, DE, EL, ES, HR, HU, IE, IT, LU, NL, PT, RO,
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE VETERINARY MEDICINAL PRODUCT Tralieve 50 mg/ml solution for injection for dogs (AT, BE, BG, CY, CZ, DE, EL, ES, HR, HU, IE, IT, LU, NL, PT, RO,
NEW ZEALAND DATA SHEET
 1 ONDANSETRON-CLARIS 2 mg/ml solution for injection Ondansetron-Claris Solution for Injection 2 mg/ml 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Ondansetron-Claris, Solution for Injection, is a clear colourless
1 ONDANSETRON-CLARIS 2 mg/ml solution for injection Ondansetron-Claris Solution for Injection 2 mg/ml 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Ondansetron-Claris, Solution for Injection, is a clear colourless
2. QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Acetylcysteine 200 mg/ml Injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Acetylcysteine 200 mg per ml. Each 10ml ampoule contains
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Acetylcysteine 200 mg/ml Injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Acetylcysteine 200 mg per ml. Each 10ml ampoule contains
Salapin: Salbutamol BP 2mg as sulphate in each 5mL of a raspberry cola flavoured, sugar free syrup.
 Salapin Salbutamol Syrup 2mg/5mL Qualitative and quantitative composition Salapin: Salbutamol BP 2mg as sulphate in each 5mL of a raspberry cola flavoured, sugar free syrup. Clinical particulars Therapeutic
Salapin Salbutamol Syrup 2mg/5mL Qualitative and quantitative composition Salapin: Salbutamol BP 2mg as sulphate in each 5mL of a raspberry cola flavoured, sugar free syrup. Clinical particulars Therapeutic
Quelicin TM SUCCINYLCHOLINE CHLORIDE INJECTION, USP
 Quelicin TM SUCCINYLCHOLINE CHLORIDE INJECTION, USP A short-acting depolarizing skeletal muscle relaxant. Fliptop Vial Rx only WARNING RISK OF CARDIAC ARREST FROM HYPERKALEMIC RHABDOMYOLYSIS There have
Quelicin TM SUCCINYLCHOLINE CHLORIDE INJECTION, USP A short-acting depolarizing skeletal muscle relaxant. Fliptop Vial Rx only WARNING RISK OF CARDIAC ARREST FROM HYPERKALEMIC RHABDOMYOLYSIS There have
Final Core Safety Profile for propofol 10 mg/ml (1%) and 20 mg/ml (2%) emulsion for injection or infusion
 CSP Drug Substance Propofol Date September 04 2014 Final Core Safety Profile for propofol 10 mg/ml (1%) and 20 mg/ml (2%) emulsion for injection or infusion CORE SAFETY PROFILE 4.3 Contraindications Propofol
CSP Drug Substance Propofol Date September 04 2014 Final Core Safety Profile for propofol 10 mg/ml (1%) and 20 mg/ml (2%) emulsion for injection or infusion CORE SAFETY PROFILE 4.3 Contraindications Propofol
PRODUCT INFORMATION. RAPIFEN (alfentanil hydrochloride) is a potent short-acting narcotic analgesic for intravenous use by anaesthetists.
 RAPIFEN INJECTION PRODUCT INFORMATION NAME OF THE MEDICINE Alfentanil Hydrochloride (equivalent to 0.5mg/mL alfentanil) DESCRIPTION RAPIFEN (alfentanil hydrochloride) is a potent short-acting narcotic
RAPIFEN INJECTION PRODUCT INFORMATION NAME OF THE MEDICINE Alfentanil Hydrochloride (equivalent to 0.5mg/mL alfentanil) DESCRIPTION RAPIFEN (alfentanil hydrochloride) is a potent short-acting narcotic
Core Safety Profile. Pharmaceutical form(s)/strength: 5mg/ml and 25 mg/ml, Solution for injection, IM/IV FI/H/PSUR/0010/002 Date of FAR:
 Core Safety Profile Active substance: Esketamine Pharmaceutical form(s)/strength: 5mg/ml and 25 mg/ml, Solution for injection, IM/IV P-RMS: FI/H/PSUR/0010/002 Date of FAR: 29.05.2012 4.3 Contraindications
Core Safety Profile Active substance: Esketamine Pharmaceutical form(s)/strength: 5mg/ml and 25 mg/ml, Solution for injection, IM/IV P-RMS: FI/H/PSUR/0010/002 Date of FAR: 29.05.2012 4.3 Contraindications
NIMBEX Injection. This information is intended for U.S. residents only. only. (cisatracurium besylate)
 This information is intended for U.S. residents only. NIMBEX Injection (cisatracurium besylate) only DESCRIPTION CLINICAL PHARMACOLOGY INDICATIONS AND USAGE CONTRAINDICATIONS WARNINGS PRECAUTIONS ADVERSE
This information is intended for U.S. residents only. NIMBEX Injection (cisatracurium besylate) only DESCRIPTION CLINICAL PHARMACOLOGY INDICATIONS AND USAGE CONTRAINDICATIONS WARNINGS PRECAUTIONS ADVERSE
GlaxoSmithKline. Renal impairment. Hepatic impairment
 RELENZA GlaxoSmithKline Zanamivir QUALITATIVE AND QUANTITATIVE COMPOSITION Each RELENZA ROTADISK consists of four regularly spaced double foil blisters each containing a white to off-white micronised powder
RELENZA GlaxoSmithKline Zanamivir QUALITATIVE AND QUANTITATIVE COMPOSITION Each RELENZA ROTADISK consists of four regularly spaced double foil blisters each containing a white to off-white micronised powder
ANNEX III LABELLING AND PACKAGE LEAFLET
 ANNEX III LABELLING AND PACKAGE LEAFLET 1 A. LABELLING 2 PARTICULARS TO APPEAR ON THE OUTER PACKAGE AND THE IMMEDIATE PACKAGE Outer carton Multi-pack 1. NAME OF THE VETERINARY MEDICINAL PRODUCT Tralieve
ANNEX III LABELLING AND PACKAGE LEAFLET 1 A. LABELLING 2 PARTICULARS TO APPEAR ON THE OUTER PACKAGE AND THE IMMEDIATE PACKAGE Outer carton Multi-pack 1. NAME OF THE VETERINARY MEDICINAL PRODUCT Tralieve
ZOFRAN TABLETS GlaxoSmithKline
 ZOFRAN TABLETS GlaxoSmithKline Ondansetron QUALITATIVE AND QUANTITATIVE COMPOSITION ZOFRAN tablets 4 mg: Each tablet contains ondansetron 4 mg as hydrochloride dihydrate. ZOFRAN tablets 8 mg: Each tablet
ZOFRAN TABLETS GlaxoSmithKline Ondansetron QUALITATIVE AND QUANTITATIVE COMPOSITION ZOFRAN tablets 4 mg: Each tablet contains ondansetron 4 mg as hydrochloride dihydrate. ZOFRAN tablets 8 mg: Each tablet
50% Concentrated Injection
 NAME OF THE MEDICINE. The molecular weight of the compound is 246.5 and the CAS registry number is 10034-99-8. The molecular formula is MgSO4, 7H2O. DESCRIPTION MAGNESIUM SULFATE HEPTAHYDRATE 50% CONCENTRATED
NAME OF THE MEDICINE. The molecular weight of the compound is 246.5 and the CAS registry number is 10034-99-8. The molecular formula is MgSO4, 7H2O. DESCRIPTION MAGNESIUM SULFATE HEPTAHYDRATE 50% CONCENTRATED
DBL MAGNESIUM SULFATE CONCENTRATED INJECTION
 DBL MAGNESIUM SULFATE CONCENTRATED INJECTION NAME OF MEDICINE Magnesium Sulfate BP DESCRIPTION DBL Magnesium Sulfate Concentrated Injection is a clear, colourless, sterile solution. Each ampoule contains
DBL MAGNESIUM SULFATE CONCENTRATED INJECTION NAME OF MEDICINE Magnesium Sulfate BP DESCRIPTION DBL Magnesium Sulfate Concentrated Injection is a clear, colourless, sterile solution. Each ampoule contains
PROSTIGMIN (neostigmine bromide)
 PROSTIGMIN (neostigmine bromide) TABLETS DESCRIPTION: Prostigmin (neostigmine bromide), an anticholinesterase agent, is available for oral administration in 15 mg tablets. Each tablet also contains gelatin,
PROSTIGMIN (neostigmine bromide) TABLETS DESCRIPTION: Prostigmin (neostigmine bromide), an anticholinesterase agent, is available for oral administration in 15 mg tablets. Each tablet also contains gelatin,
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Addaven concentrate for solution for infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Addaven contains: 1 ml 1 ampoule (10 ml) Chromic
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Addaven concentrate for solution for infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Addaven contains: 1 ml 1 ampoule (10 ml) Chromic
MINIMS AMETHOCAINE EYE DROPS
 MINIMS AMETHOCAINE EYE DROPS NAME OF THE MEDICINE Amethocaine hydrochloride Synonyms: Tetracaine hydrochloride Structural formula: Chemical name: 2-(dimethylamino)ethyl 4-(butylamino)benzoate hydrochloride
MINIMS AMETHOCAINE EYE DROPS NAME OF THE MEDICINE Amethocaine hydrochloride Synonyms: Tetracaine hydrochloride Structural formula: Chemical name: 2-(dimethylamino)ethyl 4-(butylamino)benzoate hydrochloride
NIMBEX (cisatracurium besylate) injection
 NIMBEX (cisatracurium besylate) injection This drug should be administered only by adequately trained individuals familiar with its actions, characteristics, and hazards. NOT FOR USE IN NEONATES CONTAINS
NIMBEX (cisatracurium besylate) injection This drug should be administered only by adequately trained individuals familiar with its actions, characteristics, and hazards. NOT FOR USE IN NEONATES CONTAINS
NEUROMUSCULAR BLOCKING AGENTS
 NEUROMUSCULAR BLOCKING AGENTS Edward JN Ishac, Ph.D. Associate Professor, Pharmacology and Toxicology Smith 742, 828-2127, Email: eishac@vcu.edu Learning Objectives: 1. Understand the physiology of the
NEUROMUSCULAR BLOCKING AGENTS Edward JN Ishac, Ph.D. Associate Professor, Pharmacology and Toxicology Smith 742, 828-2127, Email: eishac@vcu.edu Learning Objectives: 1. Understand the physiology of the
PRODUCT INFORMATION. Alfentanil hydrochloride
 RAPIFEN INJECTION PRODUCT INFORMATION NAME OF THE MEDICINE Alfentanil Hydrochloride (equivalent to 0.5mg/mL alfentanil) Chemical Structure HCl CAS Number 71195-58-9 Alfentanil hydrochloride DESCRIPTION
RAPIFEN INJECTION PRODUCT INFORMATION NAME OF THE MEDICINE Alfentanil Hydrochloride (equivalent to 0.5mg/mL alfentanil) Chemical Structure HCl CAS Number 71195-58-9 Alfentanil hydrochloride DESCRIPTION
TRAPADOL INJECTION FOR I.V./I.M. USE ONLY
 TRAPADOL INJECTION FOR I.V./I.M. USE ONLY Composition : Each 2ml. contains : Tramadol Hydrochloride I.P. Water for injection I.P. 100mg. q.s. CLINICAL PHARMACOLOGY : Pharmacodynamics Tramadol is a centrally
TRAPADOL INJECTION FOR I.V./I.M. USE ONLY Composition : Each 2ml. contains : Tramadol Hydrochloride I.P. Water for injection I.P. 100mg. q.s. CLINICAL PHARMACOLOGY : Pharmacodynamics Tramadol is a centrally
DATA SHEET. Product Summary. 1. Trade Name of Medicinal Product. Protamine Sulphate Injection BP. 2. Qualitative and Quantitative Composition
 DATA SHEET Product Summary 1. Trade Name of Medicinal Product Protamine Sulphate Injection BP 2. Qualitative and Quantitative Composition Protamine Sulphate 10mg/ml 3. Pharmaceutical Form Solution for
DATA SHEET Product Summary 1. Trade Name of Medicinal Product Protamine Sulphate Injection BP 2. Qualitative and Quantitative Composition Protamine Sulphate 10mg/ml 3. Pharmaceutical Form Solution for
ROSOBAC-1GM / ROSOBAC-FORT
 ROSOBAC-1GM / ROSOBAC-FORT ROSOBAC - 1GM. COMPOSITION : Each vial contains Sterile Cefoperazone Sodium IP Eq. to Anhydrous Cefoperazone - Sterile Sulbactam Sodium USP Eq. to Anhydrous Sulbactam - ROSOBAC
ROSOBAC-1GM / ROSOBAC-FORT ROSOBAC - 1GM. COMPOSITION : Each vial contains Sterile Cefoperazone Sodium IP Eq. to Anhydrous Cefoperazone - Sterile Sulbactam Sodium USP Eq. to Anhydrous Sulbactam - ROSOBAC
Granisetron Kabi, 1mg/ml, concentrate for solution for injection/infusion
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Granisetron Kabi, 1mg/ml, concentrate for solution for injection/infusion 2 QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml contains granisetron
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Granisetron Kabi, 1mg/ml, concentrate for solution for injection/infusion 2 QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml contains granisetron
NEW ZEALAND DATA SHEET
 1. PRODUCT NAME ADDAVEN (infusion, solution concentrate) NEW ZEALAND DATA SHEET 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each 10 ml ampoule of Addaven contains: Chromic chloride hexahydrate 53.33 µg
1. PRODUCT NAME ADDAVEN (infusion, solution concentrate) NEW ZEALAND DATA SHEET 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each 10 ml ampoule of Addaven contains: Chromic chloride hexahydrate 53.33 µg
למידע נוסף ניתן לעיין בעלון לרופא באתר משרד הבריאות או ליצור קשר טלפוני בטלפון
 חברת פרמה מדיס שמחה לבשר על תחילת שיווקו של תכשיר חדש. שם התכשיר: NEOSTIGMINE - HAMELN 2.5 MG/ML INJECTION צורת מינון: SOLUTION FOR INJECTION דרך מתן: I.M, I.V, S.C הרכב )חומר פעיל(: NEOSTIGMINE METHYLSULFATE
חברת פרמה מדיס שמחה לבשר על תחילת שיווקו של תכשיר חדש. שם התכשיר: NEOSTIGMINE - HAMELN 2.5 MG/ML INJECTION צורת מינון: SOLUTION FOR INJECTION דרך מתן: I.M, I.V, S.C הרכב )חומר פעיל(: NEOSTIGMINE METHYLSULFATE
Benztropine and trihexyphenidyl: Centrally acting antimuscarinic agents used for treatment of Parkinson disease & extrapyramidal symptoms.
 Scopolamine: Tertiary amine plant alkaloid. Produces peripheral effects similar to those of atropine. Unlike atropine, scopolamine has greater action on the CNS (observed at therapeutic doses). It has
Scopolamine: Tertiary amine plant alkaloid. Produces peripheral effects similar to those of atropine. Unlike atropine, scopolamine has greater action on the CNS (observed at therapeutic doses). It has
Active ingredients: Metoclopramide Hydrochloride mg Equivalent to metoclopramide hydrochloride anhydrous mg
 Name Primperan 10 mg / 2 ml Metoclopramide hydrochloride anhydrous Solution for I.M. or I.V. injection (Ampoules) Composition Each 2 ml ampoule contains: Active ingredients: Metoclopramide Hydrochloride.
Name Primperan 10 mg / 2 ml Metoclopramide hydrochloride anhydrous Solution for I.M. or I.V. injection (Ampoules) Composition Each 2 ml ampoule contains: Active ingredients: Metoclopramide Hydrochloride.
NERVOUS SYSTEM NERVOUS SYSTEM. Somatic nervous system. Brain Spinal Cord Autonomic nervous system. Sympathetic nervous system
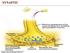 SYNAPTIC NERVOUS SYSTEM NERVOUS SYSTEM CENTRAL NERVOUS SYSTEM PERIPHERAL NERVOUS SYSTEM Brain Spinal Cord Autonomic nervous system Somatic nervous system Sympathetic nervous system Parasympathetic nervous
SYNAPTIC NERVOUS SYSTEM NERVOUS SYSTEM CENTRAL NERVOUS SYSTEM PERIPHERAL NERVOUS SYSTEM Brain Spinal Cord Autonomic nervous system Somatic nervous system Sympathetic nervous system Parasympathetic nervous
Protamine sulphate LEO Pharma 1400 anti-heparin IU/ml solution for injection and infusion.
 1. NAME OF THE MEDICINAL PRODUCT Protamine sulphate LEO Pharma 1400 anti-heparin IU/ml solution for injection and infusion. 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Protamine sulphate 1400 anti-heparin
1. NAME OF THE MEDICINAL PRODUCT Protamine sulphate LEO Pharma 1400 anti-heparin IU/ml solution for injection and infusion. 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Protamine sulphate 1400 anti-heparin
PRODUCT INFORMATION. NAME OF THE MEDICINE Compound Sodium Lactate (Hartmann's) Solution for Injection
 PRODUCT INFORMATION NAME OF THE MEDICINE Compound Sodium Lactate (Hartmann's) Solution for Injection DESCRIPTION Molecular formulae. Potassium chloride: KCl; sodium chloride: NaCl; calcium chloride dihydrate:
PRODUCT INFORMATION NAME OF THE MEDICINE Compound Sodium Lactate (Hartmann's) Solution for Injection DESCRIPTION Molecular formulae. Potassium chloride: KCl; sodium chloride: NaCl; calcium chloride dihydrate:
SUMMARY OF PRODUCT CHARACTERISTICS 2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Fexofenadine hydrochloride 180 mg film-coated tablets 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each film coated tablet contains 180mg
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Fexofenadine hydrochloride 180 mg film-coated tablets 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each film coated tablet contains 180mg
SUMMARY OF PRODUCT CHARACTERISTICS 2. QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Lidokain Isdin 40 mg/g cream 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 g cream contains 40 mg lidocaine. Excipients: Propylene glycol
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Lidokain Isdin 40 mg/g cream 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 g cream contains 40 mg lidocaine. Excipients: Propylene glycol
*Sections or subsections omitted from the full prescribing information are not listed.
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use GIAPREZA TM safely and effectively. See full prescribing information for GIAPREZA. GIAPREZA (angiotensin
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use GIAPREZA TM safely and effectively. See full prescribing information for GIAPREZA. GIAPREZA (angiotensin
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS Page 1 of 9 1. NAME OF THE MEDICINAL PRODUCT Magnesium sulfate-kalceks 500 mg/ml solution for injection, BP * 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml of solution
SUMMARY OF PRODUCT CHARACTERISTICS Page 1 of 9 1. NAME OF THE MEDICINAL PRODUCT Magnesium sulfate-kalceks 500 mg/ml solution for injection, BP * 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml of solution
1.3.1 SPC, Labelling and Package Leaflet (Voluven Fresenius 6% Solution for Infusion)
 1.3.1 SPC, Labelling and Package Leaflet (Voluven Fresenius 6% Solution for Infusion) 1.3.1 SUMMARY OF PRODUCT CHARACTERISTICS This medicinal product is subject to additional monitoring. This will allow
1.3.1 SPC, Labelling and Package Leaflet (Voluven Fresenius 6% Solution for Infusion) 1.3.1 SUMMARY OF PRODUCT CHARACTERISTICS This medicinal product is subject to additional monitoring. This will allow
Propofol 1% (10 mg/1 ml) MCT Fresenius emulsion for injection or infusion
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Propofol 1% (10 mg/1 ml) MCT Fresenius emulsion for injection or infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml emulsion contains
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Propofol 1% (10 mg/1 ml) MCT Fresenius emulsion for injection or infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml emulsion contains
SUMMARY OF PRODUCT CHARACTERISTICS 2. QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS PRODUCT SUMMARY 1. NAME OF THE MEDICINAL PRODUCT Sterile Potassium Chloride Concentrate 15%. 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 15% of Potassium Chloride in
SUMMARY OF PRODUCT CHARACTERISTICS PRODUCT SUMMARY 1. NAME OF THE MEDICINAL PRODUCT Sterile Potassium Chloride Concentrate 15%. 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 15% of Potassium Chloride in
1. NAME OF THE MEDICINAL PRODUCT
 08-15 BRIDIO Solution for Injection ORGAO Brand of sugammadex FOR ITRAVEOUS IJECTIO 1. AME OF THE MEDICIAL PRODUCT Bridion 100 mg/ml solution for injection 2. QUALITATIVE AD QUATITATIVE COMPOSITIO 1 ml
08-15 BRIDIO Solution for Injection ORGAO Brand of sugammadex FOR ITRAVEOUS IJECTIO 1. AME OF THE MEDICIAL PRODUCT Bridion 100 mg/ml solution for injection 2. QUALITATIVE AD QUATITATIVE COMPOSITIO 1 ml
SUMMARY OF PRODUCT CHARACTERISTICS. Each ml solution for injection contains phenylephrine hydrochloride corresponding to 0.1 mg phenylephrine.
 1. NAME OF THE MEDICINAL PRODUCT Fenylefrin Abcur 0.05 mg/ml, solution for injection Fenylefrin Abcur 0.1 mg/ml, solution for injection SUMMARY OF PRODUCT CHARACTERISTICS 2. QUALITATIVE AND QUANTITATIVE
1. NAME OF THE MEDICINAL PRODUCT Fenylefrin Abcur 0.05 mg/ml, solution for injection Fenylefrin Abcur 0.1 mg/ml, solution for injection SUMMARY OF PRODUCT CHARACTERISTICS 2. QUALITATIVE AND QUANTITATIVE
Health Products Regulatory Authority
 1 NAME OF THE VETERINARY MEDICINAL PRODUCT Procycline LA 200 mg/ml solution for injection 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml contains: Active Substance Oxytetracycline (as dihydrate) 200.0
1 NAME OF THE VETERINARY MEDICINAL PRODUCT Procycline LA 200 mg/ml solution for injection 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml contains: Active Substance Oxytetracycline (as dihydrate) 200.0
Nausicalm solution for injection is a clear colourless solution, presented in 1 ml ampoules.
 Nausicalm Cyclizine lactate 50 mg/ml solution for injection Presentation Nausicalm solution for injection is a clear colourless solution, presented in 1 ml ampoules. Uses Actions Cyclizine is a piperazine
Nausicalm Cyclizine lactate 50 mg/ml solution for injection Presentation Nausicalm solution for injection is a clear colourless solution, presented in 1 ml ampoules. Uses Actions Cyclizine is a piperazine
PRODUCT INFORMATION. DESCRIPTION Addaven is a concentrated trace element solution for infusion which is clear and colourless to slightly yellow.
 PRODUCT INFORMATION Addaven NAME OF MEDICINE Addaven is a concentrated trace element solution containing the following simple salts: chromic chloride hexahydrate (CAS No.:10060-12-5), cupric chloride dihydrate
PRODUCT INFORMATION Addaven NAME OF MEDICINE Addaven is a concentrated trace element solution containing the following simple salts: chromic chloride hexahydrate (CAS No.:10060-12-5), cupric chloride dihydrate
Neosynephrine. Name of the Medicine
 Name of the Medicine Neosynephrine Phenylephrine hydrochloride 1% injection Neosynephrine Presentation Neosynephrine is a clear, colourless, aqueous solution, free from visible particulates, in sterile
Name of the Medicine Neosynephrine Phenylephrine hydrochloride 1% injection Neosynephrine Presentation Neosynephrine is a clear, colourless, aqueous solution, free from visible particulates, in sterile
NEW ZEALAND DATA SHEET
 1. PRODUCT NAME Sudomyl, Tablet, 60 mg 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Name and strength of the active substance Pseudoephedrine Hydrochloride 60mg Excipient(s) with known effect For the full
1. PRODUCT NAME Sudomyl, Tablet, 60 mg 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Name and strength of the active substance Pseudoephedrine Hydrochloride 60mg Excipient(s) with known effect For the full
Ventolin Injection 500 micrograms (0.5mg), salbutamol, as sulphate, in 1ml (500 micrograms/ml).
 Ventolin parenteral preparations Salbutamol QUALITATIVE AND QUANTITATIVE COMPOSITION Ventolin Injection 500 micrograms (0.5mg), salbutamol, as sulphate, in 1ml (500 micrograms/ml). Ventolin Solution for
Ventolin parenteral preparations Salbutamol QUALITATIVE AND QUANTITATIVE COMPOSITION Ventolin Injection 500 micrograms (0.5mg), salbutamol, as sulphate, in 1ml (500 micrograms/ml). Ventolin Solution for
NEW ZEALAND DATA SHEET SEREVENT Accuhaler
 NEW ZEALAND DATA SHEET SEREVENT Accuhaler Salmeterol xinafoate (50 mcg per inhalation) Presentation SEREVENT Accuhaler is a moulded plastic device containing a foil strip with 60 regularly placed blisters
NEW ZEALAND DATA SHEET SEREVENT Accuhaler Salmeterol xinafoate (50 mcg per inhalation) Presentation SEREVENT Accuhaler is a moulded plastic device containing a foil strip with 60 regularly placed blisters
Core Safety Profile. Pharmaceutical form(s)/strength: Film-coated tablets 1.25 mg, 2.5 mg, 3.75 mg, 5 mg, 7.5 mg and 10 mg. Date of FAR:
 Core Safety Profile Active substance: Bisoprolol Pharmaceutical form(s)/strength: Film-coated tablets 1.25 mg, 2.5 mg, 3.75 mg, 5 mg, 7.5 mg and 10 mg P - RMS: FI/H/PSUR/0002/002 Date of FAR: 13.12.2011
Core Safety Profile Active substance: Bisoprolol Pharmaceutical form(s)/strength: Film-coated tablets 1.25 mg, 2.5 mg, 3.75 mg, 5 mg, 7.5 mg and 10 mg P - RMS: FI/H/PSUR/0002/002 Date of FAR: 13.12.2011
NEW ZEALAND DATA SHEET
 NEW ZEALAND DATA SHEET TRANDATE Injection labetalol hydrochloride injection 5 mg/ml Pharmaceutical Form Solution for injection. Qualitative and Quantitative Composition TRANDATE Injection: 20 ml glass
NEW ZEALAND DATA SHEET TRANDATE Injection labetalol hydrochloride injection 5 mg/ml Pharmaceutical Form Solution for injection. Qualitative and Quantitative Composition TRANDATE Injection: 20 ml glass
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE VETERINARY MEDICINAL PRODUCT Buprecare Multidose 0.3 mg/ml Solution for Injection for Dogs and Cats (UK, BE, FR, IE, LU, NL, ES) Buprenovet Multidose 0.3
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE VETERINARY MEDICINAL PRODUCT Buprecare Multidose 0.3 mg/ml Solution for Injection for Dogs and Cats (UK, BE, FR, IE, LU, NL, ES) Buprenovet Multidose 0.3
APPENDIX 1 SUMMARY OF PRODUCT CHARACTERISTICS
 SmPC Country: Lebanon Date of approval : 17/08/2011 Procedure : National APPENDIX 1 SUMMARY OF PRODUCT CHARACTERISTICS 1- NAME OF THE MEDICINAL PRODUCT DECAN, concentrate for solution for infusion 2- QUALITATIVE
SmPC Country: Lebanon Date of approval : 17/08/2011 Procedure : National APPENDIX 1 SUMMARY OF PRODUCT CHARACTERISTICS 1- NAME OF THE MEDICINAL PRODUCT DECAN, concentrate for solution for infusion 2- QUALITATIVE
(angiotensin II) injection for intravenous infusion
 ADMINISTERING GIAPREZA TM (angiotensin II) injection for intravenous infusion Visit www.giapreza.com INITIATE Recommended starting dose of GIAPREZA is 20 ng/kg/min, which is equivalent to 0.02 mcg/kg/min
ADMINISTERING GIAPREZA TM (angiotensin II) injection for intravenous infusion Visit www.giapreza.com INITIATE Recommended starting dose of GIAPREZA is 20 ng/kg/min, which is equivalent to 0.02 mcg/kg/min
SUMMARY OF PRODUCT CHARACTERISTICS. Flexbumin 200 g/l is a solution containing 200 g/l (20%) of total protein of which at least 95% is human albumin.
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Flexbumin 200g/l solution for infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Flexbumin 200 g/l is a solution containing 200 g/l
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Flexbumin 200g/l solution for infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Flexbumin 200 g/l is a solution containing 200 g/l
