Different effects of mexiletine on two mutant sodium channels causing paramyotonia congenita and hyperkalemic periodic paralysis
|
|
|
- Dorthy Nash
- 6 years ago
- Views:
Transcription
1 Neuromuscular Disorders 10 (2000) 31±39 Different effects of mexiletine on two mutant sodium channels causing paramyotonia congenita and hyperkalemic periodic paralysis K. Weckbecker a, b, 1,A.WuÈrz a,1, B. Mohammadi a, T. Mansuroglu a, A.L. George Jr. d, H. Lerche b, c, R. Dengler a, F. Lehmann-Horn b, N. Mitrovic b, c, * a Department of Neurology, Medizinische Hochschule, Hannover, Germany b Department of Applied Physiology, University of Ulm, Ulm, Germany c Department of Neurology, University of Ulm, Ulm, Germany d Department of Medicine, Vanderbilt University Medical Center, Nashville TN, USA Received 2 December 1998; received in revised form 6 April 1999; accepted 29 May 1999 Abstract Effects of the antiarrhythmic and antimyotonic drug mexiletine were studied on two sodium channel mutants causing paramyotonia congenita (R1448H) and an overlap paramyotonic and hyperkalemic paralytic syndrome (M1360V). Channels were expressed in human embryonic kidney cells and studied electrophysiologically, using the whole-cell patch-clamp technique. Compared to the wild-type, channel, both mutants showed alterations of inactivation, i.e. slower inactivation, left shift of steady-state inactivation and faster recovery from inactivation. Mexiletine caused a signi cantly larger use-dependent block of the R1448H mutant when compared to M1360V and wild-type channels. This can be explained by a prolonged recovery from mexiletine block as observed for R1448H channels, since the af nity of mexiletine for the inactivated state was similar for all three clones. The use-dependent block of sodium channels by mexiletine reduces repetitive series of action potentials and therefore improves muscle stiffness in myotonic patients. The enhanced use-dependent block as seen with R1448H may explain the extraordinary therapeutic ef cacy of mexiletine in most patients with paramyotonia congenita. q 2000 Elsevier Science B.V. All rights reserved. Keywords: Local anaesthetics; Channelopathies; Myotonia; Patch-clamp 1. Introduction To date about 20 different naturally occurring mutations have been found in the human skeletal muscle sodium channel a-subunit which may cause either of three neurological syndromes, hyperkalemic periodic paralysis (HyperPP), paramyotonia congenita (PC) and potassium-aggravated myotonia (PAM) [1]. Hallmarks of PC are paradoxical myotonia and cold-induced muscle weakness. Paradoxical myotonia is muscle stiffness increasing with continued exercise, which is in contrast to the `warm-up' phenomenon observed with the chloride channel myotonias Thomsen and Becker. In contrast, temperature sensitivity is not a typical sign of HyperPP and PAM [1,2]. HyperPP is characterised by episodic weakness sometimes accompanied by myotonia. Weakness usually appears at rest after heavy * Corresponding author. Department of Neurology/Applied Physiology, University of Ulm, Helmholzstrasse 8-1, Ulm, Germany. Tel.: ; fax: address: nenad.mitrovic@medizin.uni-ulm.de (N. Mitrovic) 1 These authors contributed equally to this study. exercise or is precipitated by depolarising agents such as potassium. Potassium-aggravated myotonia is characterised by muscle stiffness induced or aggravated by a potassium load occurring typically without weakness [2]. Normal excitability of muscle bres requires a high resting potential and short-lasting action potentials. These requirements are not ful lled in PC and HyperPP as shown with microelectrode recordings on excised muscle bres from patients. Voltage-clamp studies on single bres have revealed that inactivation of sodium currents is incomplete [3]. A defective inactivation with increased sodium in ux results in depolarisation of muscle bres. The degree of depolarisation determines the clinical phenotype; a slight depolarisation causes membrane hyperexcitability and muscle stiffness, whereas a substantial and sustained depolarisation induces membrane inexcitability and muscle paralysis. Patch-clamp studies on mutant sodium channel causing PC, PAM or HyperPP expressed in human cell lines not only revealed detailed mechanisms of the pathophysiology of these diseases but also pointed to parts of the protein important for channel function [4±8] /00/$ - see front matter q 2000 Elsevier Science B.V. All rights reserved. PII: S (99)
2 32 K. Weckbecker et al. / Neuromuscular Disorders 10 (2000) 31±39 The standard therapy in myotonia patients is the administration of sodium channel blocking agents of the lidocaine type. Mexiletine is favoured because of less serious sideeffects compared to tocainide or lidocaine [9]. Under therapy with mexiletine or tocainide, the muscle stiffness and weakness in patients with PC, as well as myotonia in other myotonic syndromes, tend to disappear completely [10,11]. The therapeutic effect of mexiletine in HyperPP patients is not satisfactory [12]. The severity and duration of symptoms can be reduced by administration of hydrochlorothiazide, acetazolamide or b-mimetic agents. Lidocaine causes a resting, or tonic block of sodium channels and a phasic block, also called use-dependent block. The blocking mechanism can be explained by a change of the channel af nity for the drug in different kinetic states. The resting block of sodium current by lidocaine-like local anaesthetics is attributed to the af nity of the drug to the resting state of the channel, whereas the additional phasic block occurs due to an increased af nity to the inactivated state [13]. Regions in the sodium channel that can be involved in binding of local anaesthetics are the cytoplasmic end of segment S6 in the domain IV and the intracellular loop between domains III±IV containing the proposed inactivation particle [14,15]. We used the patch-clamp technique to study the effects of mexiletine on two sodium channel mutants causing paramyotonic and/or hyperkalemic paralytic syndromes. Although both mutants show similar inactivation defects, mexiletine induced a larger resting and phasic block in the PC-causing mutant. Our results indicate that an increased mexiletine block of mutant channels reduces the pathologic sodium inward current in a diseased muscle, and causes a relief of symptoms in PC. 2. Methods The Arginine-1448-Histidine (R1448H) and Methionine Valine (M1360V) substitutions were created by sitedirected mutagenesis using the altered sites system (Promega Corporation, Madison, WI) as described [6,16]. Wild-type (WT) and mutant constructs were assembled in the mammalian expression vector prc/cmv and transfected into human embryonic kidney cells (HEK293) by the calcium phosphate precipitation method. Since the channel expression after transient transfection was low (,10%) stable cell lines were obtained by antibiotic selection as described [7]. Standard whole cell patch-clamp recordings [17] were performed at 208C (temperature controller II, Luigs and Neumann, Ratingen, Germany) using an EPC-9 ampli er with `Pulse' software (HEKA, Lambrecht, Germany). The voltage error due to series resistance was,5 mv. Leakage and capacitative currents were subtracted automatically by a prepulse protocol (-P/4). All data were ltered at 5 khz and digitised at 20 khz. Analysis was based on HEKA, Excel (Microsoft corporation) and Sigma Plot (Jandel Scienti c, San Rafael, CA) software. The solutions were as follows (in mm, ph 7.4): pipette: 135 CsCl, 5 NaCl, 2 MgCl2, 5 EGTA and 10 HEPES; bath: 140 NaCl, 4 KCl, 2 CaCl2, 1 MgCl2, 4 dextrose and 5 HEPES. Mexiletine was added to the bathing solution at concentrations of 0.1, 0.5 or 1.0 mm. The amplitude of sodium currents in non-transfected cells was always below 0.5 na (0:25 ^ 0:05 na, n ˆ 8). A maximum peak current in transfected cells was up to 20 na. To minimise both serial resistance and contribution of endogenous sodium channels, data were only recorded from cells with currents between 2 and 5 na (WT: 2:63 ^ 0:4 na; R1448H: 2:75 ^ 0:4 na, M1360V: 2:81^ 0:6 na, n $ 14). The mean cell capacitance was 12:9 ^ 0:18 pf with no signi cant difference between cells expressing mutant and wild-type channels. Student's t-test for comparison of two means and the Bonferroni t-test for comparison of more than two means were applied for statistic analysis. All data are presented as mean ^ SEM. 3. Results The S4 segments in all four domains contain positively charged residues, either arginines or lysines, and are proposed to be the voltage sensors of voltage-gated channels [18±20]. For the PC causing R1448H mutant, a positively charged arginine in the S4 segment of domain IV of the skeletal muscle sodium channel a-subunit is substituted by a histidine which is predominantly neutral at ph 7.4 (Fig. 1). The M1360V mutation is located in the S1 segment of domain IV (Fig. 1). Although the exact role of this segment in sodium channel function is not known, there is evidence that this part of the channel is important for inactivation [16] Characterization of the mutant channels Inactivation time course in WT and mutant channels Fig. 2A shows typical sodium currents of WT, R1448H and M1360V elicited by a depolarising step to 0 mv from a holding potential of 2100 mv after a 10 ms prepulse to 2120 mv. The inactivation time course of either mutant, in particular that of R1448H, is slower than that of the WT. The current decay was best described by a sum of two exponentials yielding two time constants of inactivation, t h1 and t h2, with a relative weight of the faster time constant, t h1, of more than 90 %. t h1 was signi cantly larger for R1448H. M1360V than for WT channels (Fig. 2B). In addition, for R1448H, t h1 showed an altered voltage dependence (Fig. 2B). t h2 was not signi cantly different between WT and mutant channels (not shown) Steady-state inactivation To characterise steady-state inactivation, which determines the probability of Na 1 channels being inactivated, a
3 K. Weckbecker et al. / Neuromuscular Disorders 10 (2000) 31±39 33 Fig. 1. Model of the human skeletal muscle sodium channel a-subunit containing four domains each having six transmembrane segments. The M1360V mutation causing a hyperkalemic paralytic and paramyotonic syndrome is located in the rst segment of the fourth domain (D4/S1). The R1448H mutation causing paramyotonia congenita is located in the fourth segment of the same domain (D4/S4). variable 20-ms prepulse ranging from 2150 to 25 mv in 5- mv steps was applied prior to a test pulse to 0 mv. The steady-state inactivation curve was t by a standard Boltzmann equation I=I max ˆ 1 1 exp q V 2 V 0:5 =kš 21.The midpoint of the inactivation curve (V 0.5 ) was shifted for both mutants to the left (Fig. 2C, Table 1). The shift was more pronounced and the slope of the curve more markedly reduced for R1448H than for M1360V (Table 1). These changes have two important implications with regard to the weakness in PC: (i) at the resting potential, the number of Na 1 channels available for an action potential is markedly reduced (at 280 mv only 70% of mutant channels versus 95% of WT) and (ii) at less negative potentials, the increased overlap of the activation and inactivation curves results in a larger permanent `window' current. The rst point directly reduces excitability, the second furthers membrane depolarisation, i.e. also reduces excitability Steady-state activation Steady-state activation was determined by using a series of depolarizations in 5 mv steps, starting with 250 mv from a holding potential of 2100 mv. Boltzmann ts of the data revealed a 26 mv shift of the steady-state activation curve for R1448H (V 0.5 in mv: WT: 215:0 ^ 2:0 versus R1448H: 221:1 ^ 4:3 versus M1360V: ^ 1.8; n $ 4). This shift might represent a real alteration of the activation process, but it could also just re ect the in uence of slowed inactivation on our usual procedure for determining the activation curve: all currents are normalised to the largest peak current which was delayed in R1448H. Table 1 Midpoint (V 0.5 ) and slope of the steady-state inactivation curve in control solution and mexiletine (0.1, 0.5, 1.0 mm) WT n R1448H n M1360V n Control solution V 0.5 (mv) ^ ^ 3.4 a ^ 2.4 a 4 Slope 2.8 ^ ^ 0.1 a 2.8 ^ 0.1 Mexiletine 0.1 mm V 0.5 (mv) ^ ^ 2.3 a ^ 0.6 a 4 Slope 2.0 ^ ^ ^ 0.1 Mexiletine 0.5 mm V 0.5 (mv) ^ ^ 2.3 a ^ 1.3 a 3 Slope 1.9 ^ ^ 0.1 a 2.2 ^ 0.2 Mexiletine 1.0 mm V 0.5 (mv) ^ ^ 4.1 a ^ Slope 1.5 ^ ^ ^ 0.1 a Data was signi cantly different for mutant channels when compared to WT (mean ^ SEM, P, 0:01).
4 34 K. Weckbecker et al. / Neuromuscular Disorders 10 (2000) 31±39 Fig. 2. Inactivation and recovery from inactivation. (A) Normalised sodium currents through WT, M1360V and R1448H channels were elicited by depolarisation from 2100 to 0 mv for 10 ms. Current amplitudes were 2.33, 3.01 and 3.71 na for WT, R1448H and M1360V channels respectively. Both mutations inactivate more slowly than the WT channel.( B) Fast time constant of inactivation t h plotted versus test potential for all three clones (n ˆ 326). (C) Steady-state inactivation curves for WT, R1448H and M1360V channels. Lines represent ts of standard Boltzmann functions with parameters given in Table 1. (D) Recovery from inactivation was measured at -100 mv. Na 1 channels were inactivated by a 36 ms prepulse to 0 mv from a holding potential of 2100 mv, and repolarised to 2100 mv recovery potential with increasing duration prior to the test pulse to 0 mv. The time course of recovery from inactivation was best described by a single exponential. Lines represent exponential ts. The values of the recovery time constant t r are given in the text and Table 2 (n ˆ 527) Recovery from inactivation Recovery from inactivation determines the refractory period after an action potential and therefore limits the maximal ring rate of nerve and muscle cells. Recovery was determined as follows: in order to inactivate all sodium channels, cells were depolarised for 36 ms to 0 mv, then repolarised with increasing duration to 2100 mv prior to the test pulse to 0 mv. The time course of recovery from inactivation was well described with a single exponential yielding the time constant t r. t r was faster for both mutants than for the WT (Fig. 2D, Table 2, t r at 2100 mv in ms: WT: 3:06 ^ 0:13 versus R1448H: 2:13 ^ 0:18 versus M1360V: 2:72 ^ 0:09 ms; for R1448H versus WT, P, 0:01; n ˆ 527, for R1448H versus M1360V P, 0:05). The faster recovery from inactivation promotes the development of myotonia by shortening the refractory period after a muscle action potential Effects of mexiletine Mexiletine block Mexiletine did not signi cantly affect the time course of inactivation and recovery from inactivation, and steadystate activation for either of the investigated channels
5 K. Weckbecker et al. / Neuromuscular Disorders 10 (2000) 31±39 35 Table 2 Recovery from inactivation and recovery from mexiletine block for WT and mutant channels WT n R1448H n M1360V n Control solution t r (ms) 3.06 ^ ^ ^ Mexiletine 0.1 mm t r (ms) 2.91 ^ ^ ^ t mex (ms) 246 ^ ^ 24* 351 ^ 32 A(t mex ) % 17.0 ^ ^ ^ 2.2 Mexiletine 0.5 mm t mex (ms) 395 ^ ^ ^ 63 3 A(t mex ) % 51.0 ^ ^ ^ 5.5 Under mexiletine a second exponential yielding a slow time constant t mex (ms) was necessary to t the data. Time constants of recovery from inactivation, t r, time constants of recovery from mexiletine block, t mex, and relative amplitudes of t mex are shown as mean values ^ SEM. At a concentration of 0.1 mm, recovery from mexiletine block was signi cantly slower for R1448H mutation than for WT and M1360V channels. *P, 0:01. (data not shown). Sodium channel block by mexiletine could be separated into a resting block and a phasic block [13]. The resting block was determined at 2100 mv holding potential, as a reduction of the peak current after administration of mexiletine. The block was signi cantly larger for R1448H channels (P, 0:05) when compared to WT and M1360V (Table 3). The larger block may be caused by an increased af nity of mexiletine for the resting state of the R1448H channel. However, it may also re ect the binding of mexiletine to the inactivated state, since some of R1448H channels at 2100 mv are already inactivated, due to the left shift of steady-state inactivation. To discriminate between these two possibilities we measured the resting block at 2150 mv holding potential where all channels are noninactivated. At this potential, block was similar for all three clones (tonic block for 0.1 mm versus 1.0 mm at 2150 mv in %: WT: 11:0 ^ 2:0 versus 56:5 ^ 4:7 R1448H: 10:3 ^ 2:5 versus 55:7 ^ 5:0; n ˆ 3 2 5) suggesting that the larger block for R1448H at 2100 mv is due to the shift of the steady-state inactivation curve. To investigate the phasic block, a series of 10 depolarising pulses to 0 mv at different frequencies (from 1 up to 100 Hz) was applied and the reduction of the current amplitude between the 1st and 10th depolarising pulse was observed. Under control conditions, i.e. in the absence of mexiletine, none of the channels showed a signi cant reduction of the peak amplitude at frequencies up to 50 Hz (Fig. 3B). Mexiletine induced a pronounced, concentration-dependent phasic block in all clones (Fig. 3A,B). At a concentration of 0.1 mm, the block was more pronounced for R1448H channels than for M1360V and WT at 2100 mv (Fig. 3B), but also at 2150 mv holding potential (data not shown). At higher concentrations (0.5 and 1 mm) mexiletine blocked all channels to a similar extent (Fig. 3B). The increased phasic block for R1448H may be either due to a higher af nity of mexiletine for the inactivated state of the mutant channel or due to a slower recovery from block. We explored both possibilities. The af nity of mexiletine to the inactivated state was estimated by determining of steady-state inactivation under different concentrations of mexiletine. Boltzmann parameters of steady-state inactivation for WT and mutant channels are summarised in Table 1. Mexiletine caused a concentration-dependent left shift of the midpoint of the inactivation curve, but the extent of the shift was not different between WT and both mutants (Fig. 4A). The left shift is due to a higher af nity of mexiletine to the inactivated state affecting the equilibrium between closed and inactivated channels. In order to calculate the af nity of mexiletine for the inactivated state, we used the following equation: V 1=2 ˆ k*ln 1= 1 1 mexiletineš=k d 1 V 1=2c [15,21], where V 1/2 and V 1/2c are the midpoints of steady-state inactivation for mexiletine and control solutions respectively, k is the slope of the steady-state inactivation curve, and k d is the dissociation constant for mexiletine from the inactivated state. Fits revealed that the k d for R1448H was not signi cantly different when compared to WT and M1360V channels (Fig. 4B) Recovery from mexiletine block As mentioned above mexiletine did not affect recovery from fast inactivation for any mutant, but it introduced a second exponential with a very slow time constant, t mex, representing the recovery from block (Fig. 4C, Table 2). The pulse protocol used to study recovery from mexiletine block was the same as for recovery from inactivation (see above or legend to Fig. 2). The equation I=I max ˆ 1 2 A*exp 2t=t r A *exp 2t=t mex )) was tted to the data and this showed that t mex was signi cantly larger for R1448H channels when compared to WT and M1360V in 0.1 mm mexiletine (P, 0:01, Table 2). This indicates slower recovery from mexiletine block. Increasing the mexiletine concentration to 0.5 mm this difference was no longer signi cant (Table 2). Since the af nity of mexiletine for the inactivated state of the WT channel and two mutants was not different (see above), the slower recovery from mexiletine block for R1448H explains the increased phasic block for this mutant. In accordance to the phasic block by mexiletine, a signi cant difference between R1448H and WT channels was only observed at a concentration of Table 3 Tonic block for WT and mutant channels, holding potential 2100 mv WT n R1448H n M1360V n Mexiletine 0.1 mm 18.7 ^ ^ 2.5* ^ Mexiletine 0.5 mm 22.0 ^ ^ 0.8* ^ Mexiletine 1.0 mm 66.7 ^ ^ 2.2* ^ Given are mean values ^ SEM. Tonic block was increased for R1448H channels, *P, 0:01.
6 36 K. Weckbecker et al. / Neuromuscular Disorders 10 (2000) 31±39 curve, increasing the permanent `window current' [5], and/ or a defect of slow inactivation [25,26] Mexiletine block Fig. 3. Phasic block in WT and mutant sodium channels induced by mexiletine. (A) Currents elicited by depolarizations from 2100 to 0 mv for 10 ms at 10 Hz frequency; extracellular mexiletine concentration 0.1 mm. Lines represent exponential ts. (B) Mean values of the phasic block at different pulse frequencies in the control solution, 0.1 and 1 mm mexiletine. Phasic block is more pronounced for R1448H channels in 0.1 mm mexiletine. 0.1 mm. At higher concentrations both phasic block and recovery from block were similar for all three clones. 4. Discussion Mexiletine block was studied on two mutants, R1448H and M1360V, in the a-subunit of the human skeletal muscle sodium channel causing either pure paramyotonia congenita (PC), with paradoxical myotonia and cold-induced muscle weakness, or an overlap syndrome, with hyperkalemic paralytic attacks and cold-induced muscle stiffness and weakness. Interestingly, both mutants showed a similar electrophysiological phenotype: slowing of inactivation, left shift and reduced voltage dependence of steady-state inactivation and faster recovery from inactivation, although more pronounced for R1448H, as shown in previous reports [6,16]. Similar alterations of sodium channel gating were found with other PC mutants [6,22±24]. In contrast, HyperPP-causing mutants show a large increase of the persistent current (,6% [4]), a left shift of the activation Although both mutants caused a similar electrophysiological phenotype, the effects of mexiletine were different in various aspects. Mexiletine caused a more pronounced resting block for the R1448H mutant, than for WT and M1360V channels at a physiologically relevant holding potential. Phasic block was also increased for the R1448H mutant in comparison to WT and M1360V, at a concentration of 0.1 mm mexiletine which is close to the therapeutic serum concentration of 0.01 mm [27]. The increased phasic block for R1448H is explained by a slower recovery from mexiletine block, since the af nity of the drug for the inactivated state was not changed by the mutation. Several models have been proposed to explain the complex interactions between inactivation of sodium channels and the binding of local anaesthetics. Most of them predict the stabilisation of the inactivated state after the drug binding resulting in a shift of the steady-state inactivation curve to the left and a decrease of the rate of recovery from inactivation [15,21]. Our observations are in agreement with this model. Since the recovery from mexiletine block was slower for R1448H one could expect that the af nity of mexiletine for the inactivated state must be increased. This was not the case in our study but can be explained using the model described in Fan et al. (1996) as follows. The af nity is determined by two rate constants, k on and k off. k on and k off determine the entry and the recovery from the blocked state. In the case that both rate constants decrease to a similar extent, the af nity would not change signi cantly (k d ˆ k off =k on ) and time constant of the recovery from block, which is de ned as 1= k on 1 k off ) would be prolonged. The R1448H mutant is located in the voltage sensor D4/ S4 of the channel which moves in response to changes of the membrane potential and this movement is disturbed by the neutralisation of positive charges such as occur in an arginine to cysteine mutation [6]. The disturbed inward movement of D4/S4 upon membrane hyperpolarization could slow the unbinding of mexiletine, prolong the recovery from block and therefore increase the phasic block. Another explanation for the large effect of mexiletine on R1448H channels could be an accumulation of mexiletine within the cell due to a reduction of the membrane surface charge. Local anaesthetics are presumed to block sodium channels in their charged form, i.e. with an ammonium group located in the pore and a lipophilic part in the membrane. Protons are known to be able to reach bound drug molecules from the extracellular side [13]. A substitution of the extracellularly located positively charged R1448 to a predominantly neutral histidine could facilitate the access of protons to neutral mexiletine molecules which
7 K. Weckbecker et al. / Neuromuscular Disorders 10 (2000) 31±39 37 Fig. 4. Steady-state inactivation and recovery from mexiletine block. (A) Steady-state inactivation curves for WT, M1360V and R1448H channels in control solution (closed symbols) and 1 mm mexiletine (open symbols). Lines represent ts of standard Boltzmann functions with parameters given in Table 1. (B) Mexiletine induced a left-shift of the midpoint potential of inactivation (V 1/2 ) in wild-type, R1448H and M1360V channels. The relative extent of the shift was similar for all three clones. Symbols are mean values ^ SEM. Lines are ttings by the equation given in the text. k d was not statistically different for R1448H, WT and M1360V channels. (C) Recovery from inactivation (closed symbols) and recovery from mexiletine block (open symbols) at 2100 mv. Mexiletine was continuously present in the bath solution. Pulse protocol was as described in Fig. 2D. The time course of recovery was best described by a sum of two exponentials. Lines represent ts. The values of t mex and corresponding relative amplitudes are given in the Table 2 (n ˆ 325). reach the channel from the cytoplasmic space or through the membrane. This could promote the formation of mexiletine `cations', which are no longer able to escape from the cell and remain trapped in the cell prolonging a blocking effect of mexiletine. Considering that recovery from block is in uenced by drug unbinding this would account for the increased value of t mex and the greater phasic block. The effects of lidocaine were tested recently on two PCcausing mutants (R1448C, T1313M): for R1448C the phasic block was increased whereas for T1313M it was decreased when compared to the WT channel [15]. T1313M is located in the III±IV interlinker of the sodium channel which is proposed to contain an inactivation particle of the sodium channel blocking the ionic pore from the intracellular side of the membrane. For T1313M Fan and colleagues [15] showed a decreased af nity of mexiletine for the inactivated state, suggesting an interaction of the inactivation ball and lidocaine molecule. Similarly Bennett and colleagues [28] have shown a decreased af nity of lidocaine for the inactivated state for the IFM±QQQ mutation also located in the III±IV interlinker. In our study, neither the R1448H nor the M1360V mutant affected the af nity of mexiletine for the inactivated state Therapeutic effect of mexiletine Due to a phasic block of sodium channels, mexiletine is a very effective inhibitor of high frequent trains of action potentials which are the electrophysiological correlate of myotonia. This unspeci c effect explains why all forms of myotonia, also the chloride channel diseases myotonia congenita Thomsen and Becker, generally respond well to
8 38 K. Weckbecker et al. / Neuromuscular Disorders 10 (2000) 31±39 treatment with mexiletine [1,29]. Cold-induced muscle weakness in PC patients also responds well to mexiletine whereas the paralytic attacks of HyperPP patients do not [10±12]. The mechanism of weakness in both diseases is probably different. In PC, weakness develops after a period of muscle stiffness in a cold environment. The pathological sodium inward current ows mainly during an action potential, since fast inactivation is slowed but the persistent sodium current is only slightly increased and slow inactivation is normal [1,6,23,24]. This is why weakness develops in the course of prolonged series of action potentials providing enough sodium in ux, so that the depolarisation exceeds some point where most sodium channels get inactivated. Such series of action potentials are blocked by mexiletine. In contrast, weakness and paralysis in HyperPP, only seldom preceded by myotonia, are caused by a large persistent sodium current and impaired slow inactivation [4,25,26]. This inducies a substantial and sustained membrane depolarisation which may be reached without repetitive action potentials. The mechanism of mexiletine block is probably not appropriate to reduce the persistent sodium current to a greater degree. Therefore, mexiletine may not be able to prevent paralytic attacks in HyperPP. Altogether, our results show a pronounced phasic block of mexiletine for R1448H channels thus explaining a very good therapeutic effect of the drug in PC patients carrying this mutation. In agreement with the extent of phasic block in electrophysiological experiments, several of our patients carrying the R1448H or the R1448C mutation respond very well to mexiletine therapy. In contrast, two patients with the T1313M mutation showed a slight or no response to mexiletine, con rming the importance of the phasic block for the clinical ef cacy of mexiletine. Acknowledgements We thank Dr. A.C. Ludolph for helpful discussion and U. Pika-Hartlaub for performing cell culture. This study was supported by the Deutsche Forschungsgemeinschaft (Le481/3), the Muscular Dystrophy Association and the National Institute of Health (NS32387). References [1] Lehmann-Horn F, RuÈdel R. Molecular pathophysiology of voltagegated ion channels. Rev Physiol Biochem Pharmacol 1996;128:195± 268. [2] Ricker K, Moxley RT, Heine R, Lehmann-Horn F. Myotonia uctuans. A third type of muscle sodium channel disease. Arch Neurol 1994;51:1095±1102. [3] Lehmann-Horn F, RuÈdel R, Ricker R. Membrane defects in paramyotonia congenita (Eulenburg). Muscle Nerve 1987;10:633±641. [4] Cannon SC, Strittmatter SM. Functional expression of Na 1 channel mutations identi ed in families with periodic paralysis. Neuron 1993;10:317±326. [5] Cummins TR, Zhou J, Sigworth FJ, Ukomadu C, Stephan M, Ptacek LJ, Agnew WS. Functional consequences of a Na 1 channel mutation causing hyperkalemic periodic paralysis. Neuron 1993;10:667± 678. [6] Chahine M, George Jr AL, Zhou M, Ji S, Sun W, Barchi RL, Horn R. Sodium channel mutations in paramyotonia congenita uncouple inactivation from activation. Neuron 1994;12:281±294. [7] Mitrovic N, George Jr AL, Heine R, Wagner S, Pika U, Hartlaub U, Zhou M, Lerche H, Fahlke C, Lehmann-Horn F. K 1 -aggravated myotonia: destabilization of the inactivated state of the human muscle Na 1 channel by the V1589M mutation. J Physiol 1994;478:395±402. [8] Mitrovic N, George Jr AL, Lerche H, Wagner S, Fahlke CH, Lehmann-Horn F. Different effects on gating of three myotonia-causing mutations in the inactivation gate of the human muscle sodium channel. J Physiol 1995;487:107±114. [9] Witt TN, Lane RJM. Myotonias. In: Brandt T, Caplan LR, Dichgans J, Diener HC, Kennard C, editors. Neurological Disorders. Course and Treatment, San Diego, CA: Academic Press, pp. 997±1002. [10] Ricker K, BoÈhlen R, Rohkamm R. Different effectiveness of tocainide and hydrochlorothiazide in paramyotonia congenita with hyperkalemic episodic paralysis. Neurology 1983;33:1615±1618. [11] Kwiecinski H, Ryniewicz B, Ostrzycki A. Treatment of myotonia with antiarrhythmic drugs. Acta Neurol Scand 1992;86:371±375. [12] Ricker K, Rohkamm R, BoÈhlen R. Adynamia episodica and paralysis periodica paramyotonica. Neurology 1986;36:682±686. [13] Hille B. Mechanisms of block. In: Hille B, editor. Ionic channels of excitable membranes, second ed., Sunderland: Sinauer, pp. 390±422. [14] Ragsdale DS, McPhee JC, Scheuer T, Catterall WA. Molecular determinants of state-dependent block of Na 1 channels by local anesthetics. Science 1994;265:1724±1728. [15] Fan Z, George AL, Kyle JW, Makielski JC. Two human paramyotonia congenita mutations have opposite effects on lidocaine block of Na 1 channels expressed in a mammalian cell line. J Physiol 1996;496:275±286. [16] Wagner S, Mitrovic N, Heine R, Lerche H, George AL, Lehmann- Horn F. A new single point mutation in human muscle sodium channel causing hyperkalemic periodic paralysis. Neurology 1997;49:1018±1025. [17] Hamill OP, Marty A, Neher E, Sakmann B, Sigworth FJ. Improved patch-clamp techniques for high-resolution current recording from cells and cell-free membrane patches. P uègers Arch 1981;391:85± 100. [18] Noda M, Shimizu S, Tanabe T, et al. Primary structure of Electrophorus electricus sodium channel deduced from cdna sequence. Nature 1984;312:121±127. [19] Catterall WA. Structure and function of voltage-gated ion channels. Annu Rev Biochem 1995;64:493±531. [20] Yang N, George Jr AL, Horn R. Molecular basis of charge movement in voltage gated Na 1 channels. Neuron 1996;16:113±122. [21] Bean B, Cohen CJ, Tsien RW. Lidocain block of cardiac sodium channels. J Gen Physiol 1983;81:613±642. [22] Yang N, Ji S, Zhou M, Ptacek LJ, Barchi RL, Horn R, George Jr AL. Sodium channel mutations in paramyotonia congenita exhibit similar biophysical phenotypes in vitro. Proc Natl Acad Sci USA 1994;91:12785± [23] Mitrovic N, Lerche H, Heine R, Fleischhauer R, Pika-Hartlaub U, Hartlaub U, George Jr AL, Lehmann-Horn F. Role in fast inactivation of conserved amino acids in the IV/S4-S5 loop of the human muscle Na 1 channel. Neurosci Lett 1996;214:9±12. [24] Lerche H, Mitrovic N, Dubovitz V, Lehmann-Horn F. Paramyotonia congenita: the R1448P Na 1 channel mutation in adult human skeletal muscle. Ann Neurol 1996;39:599±608.
9 K. Weckbecker et al. / Neuromuscular Disorders 10 (2000) 31±39 39 [25] Cummins TR, Sigworth FJ. Impaired slow inactivation in mutant sodium channels. Biophys J 1996;71:227±236. [26] Hayward LJ, Brown Jr RH, Cannon SC. Slow inactivation differs among mutant Na 1 channels associated with myotonia and periodic paralysis. Biophys J 1997;72:1204±1219. [27] Cereghino JJ, Brock JT, Van-Meter JC, Penry JK, Kupferberg HJ, Smith LD, White BG. A multiple-dose study of mexiletine (Ko 1173). Epilepsia 1975;16:673±677. [28] Bennett PB, Valenzuela C, Chen LQ, Kallen RG. On the molecular nature of the lidocaine receptor of cardiac Na 1 channel. Modi cation of block by alterations in the alpha-subunit III±IV interdomain. Circ Res 1995;77:584±592. [29] Lehmann-Horn F, Engel AG, Ricker K, RuÈdel R. The periodic paralyses and paramyotonia congenita. In: Engel AG, Franzini- Armstrong, editors. Myology, second ed., New York: McGraw-Hill, pp. 1303±1334.
Mechanisms of cold sensitivity of paramyotonia congenita mutation R1448H and overlap syndrome mutation M1360V
 J Physiol (2003), 547.3, pp. 691 698 DOI: 10.1113/jphysiol.2002.033928 The Physiological Society 2003 www.jphysiol.org Mechanisms of cold sensitivity of paramyotonia congenita mutation R1448H and overlap
J Physiol (2003), 547.3, pp. 691 698 DOI: 10.1113/jphysiol.2002.033928 The Physiological Society 2003 www.jphysiol.org Mechanisms of cold sensitivity of paramyotonia congenita mutation R1448H and overlap
a Department of Neurology, Medical School Hannover, Hannover and b Department of Applied Physiology, Ulm University, Ulm, Germany.
 Original article 235 Preferred mexiletine block of human sodium channels with IVS4 mutations and its ph-dependence* Bahram Mohammadi a, Karin Jurkat-Rott b, Alexi Alekov b, Reinhard Dengler a, Johannes
Original article 235 Preferred mexiletine block of human sodium channels with IVS4 mutations and its ph-dependence* Bahram Mohammadi a, Karin Jurkat-Rott b, Alexi Alekov b, Reinhard Dengler a, Johannes
Common molecular determinants of local anesthetic, antiarrhythmic, and anticonvulsant block of voltage-gated Na channels
 Proc. Natl. Acad. Sci. USA Vol. 93, pp. 9270 9275, August 1996 Pharmacology Common molecular determinants of local anesthetic, antiarrhythmic, and anticonvulsant block of voltage-gated Na channels DAVID
Proc. Natl. Acad. Sci. USA Vol. 93, pp. 9270 9275, August 1996 Pharmacology Common molecular determinants of local anesthetic, antiarrhythmic, and anticonvulsant block of voltage-gated Na channels DAVID
inactivation, inwhich a Role in fast inactivation of the IV/S4-S5 loop of the human muscle Nae channel probed by cysteine mutagenesis
 7283 Journal of Physiology (1997), 55.2, pp.345-352 345 Role in fast inactivation of the IV/S4-S5 loop of the human muscle Nae channel probed by cysteine mutagenesis H. Lerche*tt, W. Peter*t, R. Fleischhauer*,
7283 Journal of Physiology (1997), 55.2, pp.345-352 345 Role in fast inactivation of the IV/S4-S5 loop of the human muscle Nae channel probed by cysteine mutagenesis H. Lerche*tt, W. Peter*t, R. Fleischhauer*,
Interaction of Scorpion -Toxins with Cardiac Sodium Channels: Binding Properties and Enhancement of Slow Inactivation
 Interaction of Scorpion -Toxins with Cardiac Sodium Channels: Binding Properties and Enhancement of Slow Inactivation Haijun Chen and Stefan H. Heinemann From the Research Unit Molecular and Cellular Biophysics,
Interaction of Scorpion -Toxins with Cardiac Sodium Channels: Binding Properties and Enhancement of Slow Inactivation Haijun Chen and Stefan H. Heinemann From the Research Unit Molecular and Cellular Biophysics,
Two human paramyotonia congenita mutations have opposite effects on lidocaine block of Nae channels expressed in a mammalian cell line
 5575 Journal of Physiology (1996), 496.1, pp. 275-286 275 Two human paramyotonia congenita mutations have opposite effects on lidocaine block of Nae channels expressed in a mammalian cell line Zheng Fan,
5575 Journal of Physiology (1996), 496.1, pp. 275-286 275 Two human paramyotonia congenita mutations have opposite effects on lidocaine block of Nae channels expressed in a mammalian cell line Zheng Fan,
Activation and Inactivation of the Voltage-Gated Sodium Channel: Role of Segment S5 Revealed by a Novel Hyperkalaemic Periodic Paralysis Mutation
 The Journal of Neuroscience, June 15, 1999, 19(12):4762 4771 Activation and Inactivation of the Voltage-Gated Sodium Channel: Role of Segment S5 Revealed by a Novel Hyperkalaemic Periodic Paralysis Mutation
The Journal of Neuroscience, June 15, 1999, 19(12):4762 4771 Activation and Inactivation of the Voltage-Gated Sodium Channel: Role of Segment S5 Revealed by a Novel Hyperkalaemic Periodic Paralysis Mutation
Spectrum of sodium channel disturbances in the nondystrophic myotonias and periodic paralyses
 Kidney International, Vol. 57 (2000), pp. 772 779 Spectrum of sodium channel disturbances in the nondystrophic myotonias and periodic paralyses STEPHEN C. CANNON Department of Neurobiology, Harvard Medical
Kidney International, Vol. 57 (2000), pp. 772 779 Spectrum of sodium channel disturbances in the nondystrophic myotonias and periodic paralyses STEPHEN C. CANNON Department of Neurobiology, Harvard Medical
A novel N440K sodium channel mutation causes myotonia with exercise-induced weakness - exclusion of CLCN1 exon deletion/duplication by MLPA
 Acta Myologica 2011; XXX: p. 133-137 A novel N440K sodium channel mutation causes myotonia with exercise-induced weakness - exclusion of CLCN1 exon deletion/duplication by MLPA F. Lehmann-Horn, M. Orth
Acta Myologica 2011; XXX: p. 133-137 A novel N440K sodium channel mutation causes myotonia with exercise-induced weakness - exclusion of CLCN1 exon deletion/duplication by MLPA F. Lehmann-Horn, M. Orth
Paroxysmal extreme pain disorder mutations within the D3/S4 S5 linker of Nav1.7 cause moderate destabilization of fast inactivation
 J Physiol 586.17 (2008) pp 4137 4153 4137 Paroxysmal extreme pain disorder mutations within the D3/S4 S5 linker of Nav1.7 cause moderate destabilization of fast inactivation Brian W. Jarecki, Patrick L.
J Physiol 586.17 (2008) pp 4137 4153 4137 Paroxysmal extreme pain disorder mutations within the D3/S4 S5 linker of Nav1.7 cause moderate destabilization of fast inactivation Brian W. Jarecki, Patrick L.
Tetrapentylammonium block of chloramine-t and veratridine modi ed rat brain type IIA sodium channels
 British Journal of Pharmacology (2001) 132, 1755 ± 1760 ã 2001 Nature Publishing Group All rights reserved 0007 ± 1188/01 $15.00 Tetrapentylammonium block of chloramine-t and veratridine modi ed rat brain
British Journal of Pharmacology (2001) 132, 1755 ± 1760 ã 2001 Nature Publishing Group All rights reserved 0007 ± 1188/01 $15.00 Tetrapentylammonium block of chloramine-t and veratridine modi ed rat brain
Anaesthesia pearls and pitfalls in periodic paralysis
 Biannual Meeting of the PPA Orlando, FL, 2013 Anaesthesia pearls and pitfalls in periodic paralysis Department of Neurology Military Hospital Ulm Germany Frank Weber Frank Lehmann Horn, Senior Research
Biannual Meeting of the PPA Orlando, FL, 2013 Anaesthesia pearls and pitfalls in periodic paralysis Department of Neurology Military Hospital Ulm Germany Frank Weber Frank Lehmann Horn, Senior Research
β-scorpion toxin effects suggest electrostatic interactions in domain II of voltage-dependent sodium channels
 J Physiol 568.1 (2005) pp 13 30 13 β-scorpion toxin effects suggest electrostatic interactions in domain II of voltage-dependent sodium channels Massimo Mantegazza 1 and Sandrine Cestèle 1,2 1 Dipartimento
J Physiol 568.1 (2005) pp 13 30 13 β-scorpion toxin effects suggest electrostatic interactions in domain II of voltage-dependent sodium channels Massimo Mantegazza 1 and Sandrine Cestèle 1,2 1 Dipartimento
Neuroscience 201A Problem Set #1, 27 September 2016
 Neuroscience 201A Problem Set #1, 27 September 2016 1. The figure above was obtained from a paper on calcium channels expressed by dentate granule cells. The whole-cell Ca 2+ currents in (A) were measured
Neuroscience 201A Problem Set #1, 27 September 2016 1. The figure above was obtained from a paper on calcium channels expressed by dentate granule cells. The whole-cell Ca 2+ currents in (A) were measured
A Critical Role for the S4-S5 Intracellular Loop in Domain IV of the Sodium Channel -Subunit in Fast Inactivation*
 THE JOURNAL OF BIOLOGICAL CHEMISTRY Vol. 273, No. 2, Issue of January 9, pp. 1121 1129, 1998 1998 by The American Society for Biochemistry and Molecular Biology, Inc. Printed in U.S.A. A Critical Role
THE JOURNAL OF BIOLOGICAL CHEMISTRY Vol. 273, No. 2, Issue of January 9, pp. 1121 1129, 1998 1998 by The American Society for Biochemistry and Molecular Biology, Inc. Printed in U.S.A. A Critical Role
Cooperative effect of S4 S5 loops in domains D3 and D4 on fast inactivation of the Na + channel
 J Physiol 561.1 (2004) pp 39 51 39 Cooperative effect of S4 S5 loops in domains D3 and D4 on fast inactivation of the Na + channel M. Oana Popa 1,Alexi K. Alekov 1,SigridBail 1,Frank Lehmann-Horn 1 and
J Physiol 561.1 (2004) pp 39 51 39 Cooperative effect of S4 S5 loops in domains D3 and D4 on fast inactivation of the Na + channel M. Oana Popa 1,Alexi K. Alekov 1,SigridBail 1,Frank Lehmann-Horn 1 and
Lihui Tang,* Nabil Chehab,* Steven J. Wieland, and Roland G. Kallen* action potential can be triggered reliably, that the depolarization
 Glutamine Substitution at Alanine 1649 in the S4 S5 Cytoplasmic Loop of Domain 4 Removes the Voltage Sensitivity of Fast Inactivation in the Human Heart Sodium Channel Lihui Tang,* Nabil Chehab,* Steven
Glutamine Substitution at Alanine 1649 in the S4 S5 Cytoplasmic Loop of Domain 4 Removes the Voltage Sensitivity of Fast Inactivation in the Human Heart Sodium Channel Lihui Tang,* Nabil Chehab,* Steven
Open- and closed-state fast inactivation in sodium channels Differential effects of a site-3 anemone toxin
 Research paper Channels 5:1, 1-16; January/February 2011; 2011 Landes Bioscience research paper Open- and closed-state fast inactivation in sodium channels Differential effects of a site-3 anemone toxin
Research paper Channels 5:1, 1-16; January/February 2011; 2011 Landes Bioscience research paper Open- and closed-state fast inactivation in sodium channels Differential effects of a site-3 anemone toxin
Chapter 3 subtitles Action potentials
 CELLULAR NEUROPHYSIOLOGY CONSTANCE HAMMOND Chapter 3 subtitles Action potentials Introduction (3:15) This third chapter explains the calcium current triggered by the arrival of the action potential in
CELLULAR NEUROPHYSIOLOGY CONSTANCE HAMMOND Chapter 3 subtitles Action potentials Introduction (3:15) This third chapter explains the calcium current triggered by the arrival of the action potential in
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature19102 Supplementary Discussion Benzothiazepine Binding in Ca V Ab Diltiazem and other benzothiazepines inhibit Ca V 1.2 channels in a frequency-dependent manner consistent with pore block
doi:10.1038/nature19102 Supplementary Discussion Benzothiazepine Binding in Ca V Ab Diltiazem and other benzothiazepines inhibit Ca V 1.2 channels in a frequency-dependent manner consistent with pore block
Department of Medicinal Chemistry, Faculty of Pharmacy, University of Bari, I Bari, Italy
 British Journal of Pharmacology (1999) 128, 1165 ± 1174 ã 1999 Stockton Press All rights reserved 0007 ± 1188/99 $15.00 http://www.stockton-press.co.uk/bjp Increased hindrance on the chiral carbon atom
British Journal of Pharmacology (1999) 128, 1165 ± 1174 ã 1999 Stockton Press All rights reserved 0007 ± 1188/99 $15.00 http://www.stockton-press.co.uk/bjp Increased hindrance on the chiral carbon atom
During an action potential, sodium channels first activate,
 Na channel inactivation from open and closed states Clay M. Armstrong* Department of Physiology, University of Pennsylvania, Philadelphia, PA 19104-6085 Contributed by Clay M. Armstrong, September 26,
Na channel inactivation from open and closed states Clay M. Armstrong* Department of Physiology, University of Pennsylvania, Philadelphia, PA 19104-6085 Contributed by Clay M. Armstrong, September 26,
PROGRESSION OF SYMPTOMS AND DIFFERENCES IN THE RESPONSE OF DIFFERENT SKELETAL MUSCLES TO HYPERKALEMIC PERIODIC PARALYSIS.
 PROGRESSION OF SYMPTOMS AND DIFFERENCES IN THE RESPONSE OF DIFFERENT SKELETAL MUSCLES TO THE M1592V MUTATION OF Na V1.4 THAT CAUSES HYPERKALEMIC PERIODIC PARALYSIS By Shiemaa Khogali Thesis submitted to
PROGRESSION OF SYMPTOMS AND DIFFERENCES IN THE RESPONSE OF DIFFERENT SKELETAL MUSCLES TO THE M1592V MUTATION OF Na V1.4 THAT CAUSES HYPERKALEMIC PERIODIC PARALYSIS By Shiemaa Khogali Thesis submitted to
The Journal of Physiology
 J Physiol 589.13 (2011) pp 3115 3124 3115 Mechanisms underlying a life-threatening skeletal muscle Na + channel disorder Dina Simkin 1,2,IsabelleLéna 1, Pierre Landrieu 3, Laurence Lion-François 4, Damien
J Physiol 589.13 (2011) pp 3115 3124 3115 Mechanisms underlying a life-threatening skeletal muscle Na + channel disorder Dina Simkin 1,2,IsabelleLéna 1, Pierre Landrieu 3, Laurence Lion-François 4, Damien
A revised view of cardiac sodium channel blockade in the long-qt syndrome
 A revised view of cardiac sodium channel blockade in the long-qt syndrome Nicholas G. Kambouris, 1 H. Bradley Nuss, 2 David C. Johns, 2 Eduardo Marbán, 2 Gordon F. Tomaselli, 2 and Jeffrey R. Balser 3
A revised view of cardiac sodium channel blockade in the long-qt syndrome Nicholas G. Kambouris, 1 H. Bradley Nuss, 2 David C. Johns, 2 Eduardo Marbán, 2 Gordon F. Tomaselli, 2 and Jeffrey R. Balser 3
antiarrhythmic, and anticonvulsant block of
 Proc. Natl. Acad. Sci. USA Vol. 93, pp. 927-9275, August 1996 Pharmacology ommon molecular determinants of local anesthetic, antiarrhythmic, and anticonvulsant block of voltage-gated Na+ channels DAVID
Proc. Natl. Acad. Sci. USA Vol. 93, pp. 927-9275, August 1996 Pharmacology ommon molecular determinants of local anesthetic, antiarrhythmic, and anticonvulsant block of voltage-gated Na+ channels DAVID
Functional Effects of Two Voltage-Gated Sodium Channel Mutations That Cause Generalized Epilepsy with Febrile Seizures Plus Type 2
 The Journal of Neuroscience, October 1, 2001, 21(19):7481 7490 Functional Effects of Two Voltage-Gated Sodium Channel Mutations That Cause Generalized Epilepsy with Febrile Seizures Plus Type 2 Jay Spampanato,
The Journal of Neuroscience, October 1, 2001, 21(19):7481 7490 Functional Effects of Two Voltage-Gated Sodium Channel Mutations That Cause Generalized Epilepsy with Febrile Seizures Plus Type 2 Jay Spampanato,
Electrical Properties of Neurons. Steven McLoon Department of Neuroscience University of Minnesota
 Electrical Properties of Neurons Steven McLoon Department of Neuroscience University of Minnesota 1 Neuronal Communication Neurons communicate with other cells, often over long distances. The electrical
Electrical Properties of Neurons Steven McLoon Department of Neuroscience University of Minnesota 1 Neuronal Communication Neurons communicate with other cells, often over long distances. The electrical
A novel sodium channel mutation causing a hyperkalemic paralytic and paramyotonic syndrome with variable clinical expressivity
 A novel sodium channel mutation causing a hyperkalemic paralytic and paramyotonic syndrome with variable clinical expressivity S. Wagner, H. Lerche, N. Mitrovic, et al. Neurology 1997;49;1018 This information
A novel sodium channel mutation causing a hyperkalemic paralytic and paramyotonic syndrome with variable clinical expressivity S. Wagner, H. Lerche, N. Mitrovic, et al. Neurology 1997;49;1018 This information
Fast Calcium Currents in Cut Skeletal Muscle Fibres of the Frogs Rana temporaria and Xenopus laevis
 Gen. Physiol. Biophys. (1988), 7, 651-656 65! Short communication Fast Calcium Currents in Cut Skeletal Muscle Fibres of the Frogs Rana temporaria and Xenopus laevis M. HENČĽK, D. ZACHAROVÁ and J. ZACHAR
Gen. Physiol. Biophys. (1988), 7, 651-656 65! Short communication Fast Calcium Currents in Cut Skeletal Muscle Fibres of the Frogs Rana temporaria and Xenopus laevis M. HENČĽK, D. ZACHAROVÁ and J. ZACHAR
THE INFLUENCE OF PROTONS AND ZINC IONS ON THE STEADY- STATE INACTIVATION OF Kv1.3 POTASSIUM CHANNELS
 CELLULAR & MOLECULAR BIOLOGY LETTERS Volume 12 (2007) pp 220-230 http://www.cmbl.org.pl Received: 27 July 2006 Revised form accepted: 12 October 2006 Published online: 12 December 2006 DOI: 10.2478/s11658-006-0067-6
CELLULAR & MOLECULAR BIOLOGY LETTERS Volume 12 (2007) pp 220-230 http://www.cmbl.org.pl Received: 27 July 2006 Revised form accepted: 12 October 2006 Published online: 12 December 2006 DOI: 10.2478/s11658-006-0067-6
Human TRPC6 Ion Channel Cell Line
 TECHNICAL DATA SHEET ValiScreen Ion Channel Cell Line Caution: For Laboratory Use. A research product for research purposes only Human TRPC6 Ion Channel Cell Line Product No.: AX-012-C Lot No.: 512-548-A
TECHNICAL DATA SHEET ValiScreen Ion Channel Cell Line Caution: For Laboratory Use. A research product for research purposes only Human TRPC6 Ion Channel Cell Line Product No.: AX-012-C Lot No.: 512-548-A
Differential modulation of Na v 1.7 and Na v 1.8 peripheral nerve sodium channels by the local anesthetic lidocaine
 British Journal of Pharmacology (2004) 142, 576 584 & 2004 Nature Publishing Group All rights reserved 0007 1188/04 $30.00 www.nature.com/bjp Differential modulation of Na v 1.7 and Na v 1.8 peripheral
British Journal of Pharmacology (2004) 142, 576 584 & 2004 Nature Publishing Group All rights reserved 0007 1188/04 $30.00 www.nature.com/bjp Differential modulation of Na v 1.7 and Na v 1.8 peripheral
Tryptophan Scanning of D1S6 and D4S6 C-Termini in Voltage-Gated Sodium Channels
 Biophysical Journal Volume 85 August 2003 911 920 911 Tryptophan Scanning of D1S6 and D4S6 C-Termini in Voltage-Gated Sodium Channels Sho-Ya Wang,* Kaitlin Bonner,* Corinna Russell, y and Ging Kuo Wang
Biophysical Journal Volume 85 August 2003 911 920 911 Tryptophan Scanning of D1S6 and D4S6 C-Termini in Voltage-Gated Sodium Channels Sho-Ya Wang,* Kaitlin Bonner,* Corinna Russell, y and Ging Kuo Wang
Supplemental material to this article can be found at:
 Supplemental material to this article can be found at: http://molpharm.aspetjournals.org/content/suppl/2010/09/20/mol.110.066332.dc1 0026-895X/10/7806-1124 1134$20.00 MOLECULAR PHARMACOLOGY Vol. 78, No.
Supplemental material to this article can be found at: http://molpharm.aspetjournals.org/content/suppl/2010/09/20/mol.110.066332.dc1 0026-895X/10/7806-1124 1134$20.00 MOLECULAR PHARMACOLOGY Vol. 78, No.
Irreversible Block of Human Heart (hh1) Sodium Channels by the Plant Alkaloid Lappaconitine
 0026-895X/01/5902-183 192$3.00 MOLECULAR PHARMACOLOGY Vol. 59, No. 2 Copyright 2001 The American Society for Pharmacology and Experimental Therapeutics 381/877513 Mol Pharmacol 59:183 192, 2001 Printed
0026-895X/01/5902-183 192$3.00 MOLECULAR PHARMACOLOGY Vol. 59, No. 2 Copyright 2001 The American Society for Pharmacology and Experimental Therapeutics 381/877513 Mol Pharmacol 59:183 192, 2001 Printed
Chapter 5 subtitles GABAergic synaptic transmission
 CELLULAR NEUROPHYSIOLOGY CONSTANCE HAMMOND Chapter 5 subtitles GABAergic synaptic transmission INTRODUCTION (2:57) In this fifth chapter, you will learn how the binding of the GABA neurotransmitter to
CELLULAR NEUROPHYSIOLOGY CONSTANCE HAMMOND Chapter 5 subtitles GABAergic synaptic transmission INTRODUCTION (2:57) In this fifth chapter, you will learn how the binding of the GABA neurotransmitter to
Title Page. The skeletal muscle channelopathies: phenotype, genotype and pathogenesis. Emma Louise Matthews UCL. Submission for PhD examination
 Title Page The skeletal muscle channelopathies: phenotype, genotype and pathogenesis Emma Louise Matthews UCL Submission for PhD examination Declaration I, Emma Louise Matthews confirm that the work presented
Title Page The skeletal muscle channelopathies: phenotype, genotype and pathogenesis Emma Louise Matthews UCL Submission for PhD examination Declaration I, Emma Louise Matthews confirm that the work presented
Muscle Na channelopathies
 Published Ahead of Print on August 23, 2006 as 10.1212/01.wnl.0000233841.75824.0f Muscle Na channelopathies MRI detects intracellular 23 Na accumulation during episodic weakness M.-A. Weber, MD; S. Nielles-Vallespin,
Published Ahead of Print on August 23, 2006 as 10.1212/01.wnl.0000233841.75824.0f Muscle Na channelopathies MRI detects intracellular 23 Na accumulation during episodic weakness M.-A. Weber, MD; S. Nielles-Vallespin,
Facilitation of Recovery from Inactivation by External Na and Location of the Activation Gate in Neuronal Na Channels
 The Journal of Neuroscience, August 1, 2000, 20(15):5639 5646 Facilitation of Recovery from Inactivation by External Na and Location of the Activation Gate in Neuronal Na Channels Chung-Chin Kuo 1,2 and
The Journal of Neuroscience, August 1, 2000, 20(15):5639 5646 Facilitation of Recovery from Inactivation by External Na and Location of the Activation Gate in Neuronal Na Channels Chung-Chin Kuo 1,2 and
Chloride channelopathies Conclusion Introduction
 1 Frank Lehmann-Horn, Reinhardt Rüdel. Molecular pathophysiology of voltage-gated ion channels. Chapter 128 in: Reviews of Physiology Biochemistry and Pharmacology 1996; Editors: M.P. Blaustein, Baltimore;
1 Frank Lehmann-Horn, Reinhardt Rüdel. Molecular pathophysiology of voltage-gated ion channels. Chapter 128 in: Reviews of Physiology Biochemistry and Pharmacology 1996; Editors: M.P. Blaustein, Baltimore;
Rare Monogenic Disorders. Function. Pathophysiology
 Rare Monogenic Disorders Function Pathophysiology Protein Gene Episodic Nervous System Diseases Migraine Epilepsy Periodic Paralysis LQTS Episodic Ataxia Paroxysmal Dyskinesias Phenotypes Muscle diseases
Rare Monogenic Disorders Function Pathophysiology Protein Gene Episodic Nervous System Diseases Migraine Epilepsy Periodic Paralysis LQTS Episodic Ataxia Paroxysmal Dyskinesias Phenotypes Muscle diseases
The Journal of Physiology
 J Physiol 590.7 (2012) pp 1599 1614 1599 Altered Kv3.3 channel gating in early-onset spinocerebellar ataxia type 13 Natali A. Minassian 1, Meng-Chin A. Lin 1 and Diane M. Papazian 1,2,3 1 Department of
J Physiol 590.7 (2012) pp 1599 1614 1599 Altered Kv3.3 channel gating in early-onset spinocerebellar ataxia type 13 Natali A. Minassian 1, Meng-Chin A. Lin 1 and Diane M. Papazian 1,2,3 1 Department of
THE EFFECT OF ZINC ON CALCIUM AND HYDROGEN ION CURRENTS IN INTACT SNAIL NEURONES BY M. P. MAHAUT-SMITH*
 /. exp. Biol. 145, 455-464 (1989) 455 Printed in Great Britain The Company of Biologists Limited 1989 THE EFFECT OF ZINC ON CALCIUM AND HYDROGEN ION CURRENTS IN INTACT SNAIL NEURONES BY M. P. MAHAUT-SMITH*
/. exp. Biol. 145, 455-464 (1989) 455 Printed in Great Britain The Company of Biologists Limited 1989 THE EFFECT OF ZINC ON CALCIUM AND HYDROGEN ION CURRENTS IN INTACT SNAIL NEURONES BY M. P. MAHAUT-SMITH*
Cellular/Molecular The Journal of Neuroscience, November 29, (48):
 12566 The Journal of Neuroscience, November 29, 2006 26(48):12566 12575 Cellular/Molecular Na v 1.7 Mutant A863P in Erythromelalgia: Effects of Altered Activation and Steady-State Inactivation on Excitability
12566 The Journal of Neuroscience, November 29, 2006 26(48):12566 12575 Cellular/Molecular Na v 1.7 Mutant A863P in Erythromelalgia: Effects of Altered Activation and Steady-State Inactivation on Excitability
7.06 Spring of PROBLEM SET #6
 7.6 Spring 23 1 of 6 7.6 PROBLEM SET #6 1. You are studying a mouse model of hypercholesterolemia, a disease characterized by high levels of cholesterol in the blood. In normal cells, LDL particles in
7.6 Spring 23 1 of 6 7.6 PROBLEM SET #6 1. You are studying a mouse model of hypercholesterolemia, a disease characterized by high levels of cholesterol in the blood. In normal cells, LDL particles in
Chapter 3 Neurotransmitter release
 NEUROPHYSIOLOGIE CELLULAIRE CONSTANCE HAMMOND Chapter 3 Neurotransmitter release In chapter 3, we proose 3 videos: Observation Calcium Channel, Ca 2+ Unitary and Total Currents Ca 2+ and Neurotransmitter
NEUROPHYSIOLOGIE CELLULAIRE CONSTANCE HAMMOND Chapter 3 Neurotransmitter release In chapter 3, we proose 3 videos: Observation Calcium Channel, Ca 2+ Unitary and Total Currents Ca 2+ and Neurotransmitter
Sample Lab Report 1 from 1. Measuring and Manipulating Passive Membrane Properties
 Sample Lab Report 1 from http://www.bio365l.net 1 Abstract Measuring and Manipulating Passive Membrane Properties Biological membranes exhibit the properties of capacitance and resistance, which allow
Sample Lab Report 1 from http://www.bio365l.net 1 Abstract Measuring and Manipulating Passive Membrane Properties Biological membranes exhibit the properties of capacitance and resistance, which allow
Identification of Three Cysteines as Targets for the Zn 2 Blockade of the Human Skeletal Muscle Chloride Channel*
 THE JOURNAL OF BIOLOGICAL CHEMISTRY Vol. 274, No. 17, Issue of April 23, pp. 11687 11692, 1999 1999 by The American Society for Biochemistry and Molecular Biology, Inc. Printed in U.S.A. Identification
THE JOURNAL OF BIOLOGICAL CHEMISTRY Vol. 274, No. 17, Issue of April 23, pp. 11687 11692, 1999 1999 by The American Society for Biochemistry and Molecular Biology, Inc. Printed in U.S.A. Identification
Sodium channelopathies of skeletal muscle result from gain or loss of function
 Pflugers Arch - Eur J Physiol (2010) 460:239 248 DOI 10.1007/s00424-010-0814-4 INVITED REVIEW Sodium channelopathies of skeletal muscle result from gain or loss of function Karin Jurkat-Rott & Boris Holzherr
Pflugers Arch - Eur J Physiol (2010) 460:239 248 DOI 10.1007/s00424-010-0814-4 INVITED REVIEW Sodium channelopathies of skeletal muscle result from gain or loss of function Karin Jurkat-Rott & Boris Holzherr
Supplementary Information
 Hyperpolarization-activated cation channels inhibit EPSPs by interactions with M-type K + channels Meena S. George, L.F. Abbott, Steven A. Siegelbaum Supplementary Information Part 1: Supplementary Figures
Hyperpolarization-activated cation channels inhibit EPSPs by interactions with M-type K + channels Meena S. George, L.F. Abbott, Steven A. Siegelbaum Supplementary Information Part 1: Supplementary Figures
Absence of Ca2+ current facilitation in skeletal muscle of transgenic mice lacking the type 1 ryanodine receptor
 592 Journal of Physiology (1996), 496.2, pp.339-345 Absence of Ca2+ current facilitation in skeletal muscle of transgenic mice lacking the type 1 ryanodine receptor Andrea Fleig, Hiroshi Takeshima * and
592 Journal of Physiology (1996), 496.2, pp.339-345 Absence of Ca2+ current facilitation in skeletal muscle of transgenic mice lacking the type 1 ryanodine receptor Andrea Fleig, Hiroshi Takeshima * and
and paralysis periodica paramyotonia
 618 Department of Neurology, College of Medicine, Chungnam National University, 640 Daesa-dong, Joong-ku, Taejon, Korea J Kim E H Sohn J M Kim Department of Biological Sciences, Korea Advanced Institute
618 Department of Neurology, College of Medicine, Chungnam National University, 640 Daesa-dong, Joong-ku, Taejon, Korea J Kim E H Sohn J M Kim Department of Biological Sciences, Korea Advanced Institute
Acta Physiologica Sinica
 , 1999 4, 51 (2), 187 192 187 Acta Physiologica Sinica 3 1998204222 1998206203 3 (No139500052) 3 3, 221002 3 3 3 3 3 (, 200031) ( Ito), 28 d (H28, 6 h/ d), Ito (16118 4161 6132 1135 pa/ pf, P < 0105),
, 1999 4, 51 (2), 187 192 187 Acta Physiologica Sinica 3 1998204222 1998206203 3 (No139500052) 3 3, 221002 3 3 3 3 3 (, 200031) ( Ito), 28 d (H28, 6 h/ d), Ito (16118 4161 6132 1135 pa/ pf, P < 0105),
DOI: /jphysiol The Physiological Society Rapid Report
 Journal of Physiology (2002), 541.3, pp. 665 672 DOI: 10.1113/jphysiol.2002.020503 The Physiological Society 2002 www.jphysiol.org Rapid Report Phosphorylation-dependent differences in the activation properties
Journal of Physiology (2002), 541.3, pp. 665 672 DOI: 10.1113/jphysiol.2002.020503 The Physiological Society 2002 www.jphysiol.org Rapid Report Phosphorylation-dependent differences in the activation properties
Aberrant epilepsy-associated mutant Na v 1.6 sodium channel activity can be targeted with cannabidiol
 doi:10.1093/brain/aww129 BRAIN 2016: 139; 2164 2181 2164 Aberrant epilepsy-associated mutant Na v 1.6 sodium channel activity can be targeted with cannabidiol Reesha R. Patel, 1,2 Cindy Barbosa, 3 Tatiana
doi:10.1093/brain/aww129 BRAIN 2016: 139; 2164 2181 2164 Aberrant epilepsy-associated mutant Na v 1.6 sodium channel activity can be targeted with cannabidiol Reesha R. Patel, 1,2 Cindy Barbosa, 3 Tatiana
The plant alkaloid harmaline blocks the human voltage-gated sodium channel Na v 1.7 expressed in CHO cells: a study using automated patch clamp
 Ernst Moritz Arndt University of Greifswald Institute of Pathophysiology The plant alkaloid harmaline blocks the human voltage-gated sodium channel Na v 1.7 expressed in CHO cells: a study using automated
Ernst Moritz Arndt University of Greifswald Institute of Pathophysiology The plant alkaloid harmaline blocks the human voltage-gated sodium channel Na v 1.7 expressed in CHO cells: a study using automated
Inherited disorders of voltage-gated sodium channels
 Review series Inherited disorders of voltage-gated sodium channels Alfred L. George Jr. Division of Genetic Medicine, Departments of Medicine and Pharmacology, Vanderbilt University, Nashville, Tennessee,
Review series Inherited disorders of voltage-gated sodium channels Alfred L. George Jr. Division of Genetic Medicine, Departments of Medicine and Pharmacology, Vanderbilt University, Nashville, Tennessee,
2/7/16. Neurons maintain a negative membrane potential. Membrane potential. Ion conductances determine the membrane potential
 Neurons maintain a negative membrane potential. V Ion channels are key regulators of membrane potential. Low Na + 2mM High K + 125mM Low Ca + (10-7 ) Low Cl - (5mM) Membrane potential. V ENa= RT/nF ln[na+]o/[na+]in
Neurons maintain a negative membrane potential. V Ion channels are key regulators of membrane potential. Low Na + 2mM High K + 125mM Low Ca + (10-7 ) Low Cl - (5mM) Membrane potential. V ENa= RT/nF ln[na+]o/[na+]in
Cold Extends Electromyography Distinction between Ion Channel Mutations Causing Myotonia
 Cold Extends Electromyography Distinction between Ion Channel Mutations Causing Myotonia Emmanuel Fournier, MD, PhD, 1 5 Karine Viala, MD, 1,2 Hélène Gervais, MD, 6 Damien Sternberg, MD, PhD, 7 Marianne
Cold Extends Electromyography Distinction between Ion Channel Mutations Causing Myotonia Emmanuel Fournier, MD, PhD, 1 5 Karine Viala, MD, 1,2 Hélène Gervais, MD, 6 Damien Sternberg, MD, PhD, 7 Marianne
Voltage Gated Ion Channels
 Voltage Gated Ion Channels The Machines That Make It Possible... Topics I Introduction Electrochemical Gradients Passive Membrane Properties Action Potential Voltage-Gated Ion Channels Ligand-Gated Ion
Voltage Gated Ion Channels The Machines That Make It Possible... Topics I Introduction Electrochemical Gradients Passive Membrane Properties Action Potential Voltage-Gated Ion Channels Ligand-Gated Ion
BIONB/BME/ECE 4910 Neuronal Simulation Assignments 1, Spring 2013
 BIONB/BME/ECE 4910 Neuronal Simulation Assignments 1, Spring 2013 Tutorial Assignment Page Due Date Week 1/Assignment 1: Introduction to NIA 1 January 28 The Membrane Tutorial 9 Week 2/Assignment 2: Passive
BIONB/BME/ECE 4910 Neuronal Simulation Assignments 1, Spring 2013 Tutorial Assignment Page Due Date Week 1/Assignment 1: Introduction to NIA 1 January 28 The Membrane Tutorial 9 Week 2/Assignment 2: Passive
Correlation between Membrane Potential Responses and Tentacle Movement in the Dinoflagellate Noctiluca miliaris
 ZOOLOGICAL SCIENCE 21: 131 138 (2004) 2004 Zoological Society of Japan Correlation between Membrane Potential Responses and Tentacle Movement in the Dinoflagellate Noctiluca miliaris Kazunori Oami* Institute
ZOOLOGICAL SCIENCE 21: 131 138 (2004) 2004 Zoological Society of Japan Correlation between Membrane Potential Responses and Tentacle Movement in the Dinoflagellate Noctiluca miliaris Kazunori Oami* Institute
Sodium and Gating Current Time Shifts Resulting from Changes in Initial Conditions
 Sodium and Gating Current Time Shifts Resulting from Changes in Initial Conditions ROBERT E. TAYLOR and FRANCISCO BEZANILLA From the Laboratory of Biophysics, National Institute of Neurological and Communicative
Sodium and Gating Current Time Shifts Resulting from Changes in Initial Conditions ROBERT E. TAYLOR and FRANCISCO BEZANILLA From the Laboratory of Biophysics, National Institute of Neurological and Communicative
nachr α 4 β 2 CHO Cell Line
 B SYS GmbH nachr α 4 β 2 CHO Cell Line Cell Culture Conditions B SYS GmbH B SYS GmbH nachr α 4 β 2 CHO Page 2 TABLE OF CONTENTS 1 BACKGROUND...3 1.1 Human Nicotinic Acetylcholine Receptors...3 1.2 B SYS
B SYS GmbH nachr α 4 β 2 CHO Cell Line Cell Culture Conditions B SYS GmbH B SYS GmbH nachr α 4 β 2 CHO Page 2 TABLE OF CONTENTS 1 BACKGROUND...3 1.1 Human Nicotinic Acetylcholine Receptors...3 1.2 B SYS
Novel CLCN1 mutations with unique clinical and electrophysiological consequences
 Brain (2002), 125, 2392±2407 Novel CLCN1 mutations with unique clinical and electrophysiological consequences Fen-Fen Wu, 1,3 Aisling Ryan, 8 Joseph Devaney, 3,4 Maike Warnstedt, 9 Zeljka Korade-Mirnics,
Brain (2002), 125, 2392±2407 Novel CLCN1 mutations with unique clinical and electrophysiological consequences Fen-Fen Wu, 1,3 Aisling Ryan, 8 Joseph Devaney, 3,4 Maike Warnstedt, 9 Zeljka Korade-Mirnics,
Primary Periodic Paralysis. A Complex Disorder. A Challenging Diagnosis.
 Primary Periodic Paralysis A Complex Disorder. A Challenging Diagnosis. A Complex Disorder A Group of Rare Channelopathies With Varying Subtypes and Triggers Primary periodic paralysis causes recurrent,
Primary Periodic Paralysis A Complex Disorder. A Challenging Diagnosis. A Complex Disorder A Group of Rare Channelopathies With Varying Subtypes and Triggers Primary periodic paralysis causes recurrent,
1. (1 pt) At the equilibrium potential of an ion, what two things are equal? Electrical potential (voltage) and chemical potential (concentration)
 MIDERM REVIEW QUESIONS: IO 3411 (hese are questions from 3 of the previous years midterms) 1. (1 pt) t the equilibrium potential of an ion, what two things are equal? Electrical potential (voltage) and
MIDERM REVIEW QUESIONS: IO 3411 (hese are questions from 3 of the previous years midterms) 1. (1 pt) t the equilibrium potential of an ion, what two things are equal? Electrical potential (voltage) and
Multiple pore conformations driven by asynchronous movements of voltage sensors in a eukaryotic sodium channel
 Received 17 Sep 212 Accepted 4 Dec 212 Published 15 Jan 213 DOI: 1.138/ncomms2356 Multiple pore conformations driven by asynchronous movements of voltage sensors in a eukaryotic sodium channel Marcel P.
Received 17 Sep 212 Accepted 4 Dec 212 Published 15 Jan 213 DOI: 1.138/ncomms2356 Multiple pore conformations driven by asynchronous movements of voltage sensors in a eukaryotic sodium channel Marcel P.
Lidocaine alters the input resistance and evokes neural activity in crayfish sensory neurons
 20 Gen. Physiol. Biophys. (2007), 26, 20 26 Lidocaine alters the input resistance and evokes neural activity in crayfish sensory neurons M. B. Keceli and N. Purali Hacettepe University, Medical Faculty,
20 Gen. Physiol. Biophys. (2007), 26, 20 26 Lidocaine alters the input resistance and evokes neural activity in crayfish sensory neurons M. B. Keceli and N. Purali Hacettepe University, Medical Faculty,
The "Pacemaker" Function of the Transient Outward Current in the Rabbit Myocardium
 Gen. Physiol. Biophys. (1988). 7. 235 242 235 The "Pacemaker" Function of the Transient Outward Current in the Rabbit Myocardium R. Z. GAINULLIN 1, N. I. KUKUSHKIN 1, R. E. KISELEVA 2 and E. A. SOSUNOV
Gen. Physiol. Biophys. (1988). 7. 235 242 235 The "Pacemaker" Function of the Transient Outward Current in the Rabbit Myocardium R. Z. GAINULLIN 1, N. I. KUKUSHKIN 1, R. E. KISELEVA 2 and E. A. SOSUNOV
Quantal Analysis Problems
 Quantal Analysis Problems 1. Imagine you had performed an experiment on a muscle preparation from a Drosophila larva. In this experiment, intracellular recordings were made from an identified muscle fibre,
Quantal Analysis Problems 1. Imagine you had performed an experiment on a muscle preparation from a Drosophila larva. In this experiment, intracellular recordings were made from an identified muscle fibre,
Mutations of the S4 S5 linker alter activation properties of HERG potassium channels expressed in Xenopus oocytes
 Keywords: Potassium channel, Xenopus 8564 Journal of Physiology (1999), 514.3, pp. 667 675 667 Mutations of the S4 S5 linker alter activation properties of HERG potassium channels expressed in Xenopus
Keywords: Potassium channel, Xenopus 8564 Journal of Physiology (1999), 514.3, pp. 667 675 667 Mutations of the S4 S5 linker alter activation properties of HERG potassium channels expressed in Xenopus
OPEN.
 www.nature.com/scientificreports Received: 28 February 2018 Accepted: 20 June 2018 Published: xx xx xxxx OPEN Na V 1.4 DI-S4 periodic paralysis mutation R222W enhances inactivation and promotes leak current
www.nature.com/scientificreports Received: 28 February 2018 Accepted: 20 June 2018 Published: xx xx xxxx OPEN Na V 1.4 DI-S4 periodic paralysis mutation R222W enhances inactivation and promotes leak current
BRIEF COMMUNICATION CALCIUM- AND VOLTAGE-ACTIVATED POTASSIUM CHANNELS IN HUMAN MACROPHAGES. frequency of channel opening increased with depolarization
 BRIEF COMMUNICATION CALCIUM- AND VOLTAGE-ACTIVATED POTASSIUM CHANNELS IN HUMAN MACROPHAGES ELAINE K. GALLIN Physiology Department, Armed Forces Radiobiology Research Institute, Bethesda, Maryland 20814
BRIEF COMMUNICATION CALCIUM- AND VOLTAGE-ACTIVATED POTASSIUM CHANNELS IN HUMAN MACROPHAGES ELAINE K. GALLIN Physiology Department, Armed Forces Radiobiology Research Institute, Bethesda, Maryland 20814
Drugs, Drug Targets and You: Patch Clamping
 Drugs, Drug Targets and You: Patch Clamping Introduction To elucidate how an ion channel operates, one needs to examine the factors that influence its opening and closing as well as measure the resulting
Drugs, Drug Targets and You: Patch Clamping Introduction To elucidate how an ion channel operates, one needs to examine the factors that influence its opening and closing as well as measure the resulting
The Batrachotoxin Receptor on the Voltage-Gated Sodium Channel is Guarded by the Channel Activation Gate
 0026-895X/02/6104-905 912$7.00 MOLECULAR PHARMACOLOGY Vol. 61, No. 4 Copyright 2002 The American Society for Pharmacology and Experimental Therapeutics 1171/974120 Mol Pharmacol 61:905 912, 2002 Printed
0026-895X/02/6104-905 912$7.00 MOLECULAR PHARMACOLOGY Vol. 61, No. 4 Copyright 2002 The American Society for Pharmacology and Experimental Therapeutics 1171/974120 Mol Pharmacol 61:905 912, 2002 Printed
Neurophysiology of Nerve Impulses
 M52_MARI0000_00_SE_EX03.qxd 8/22/11 2:47 PM Page 358 3 E X E R C I S E Neurophysiology of Nerve Impulses Advance Preparation/Comments Consider doing a short introductory presentation with the following
M52_MARI0000_00_SE_EX03.qxd 8/22/11 2:47 PM Page 358 3 E X E R C I S E Neurophysiology of Nerve Impulses Advance Preparation/Comments Consider doing a short introductory presentation with the following
Synaptic Integration
 Synaptic Integration 3 rd January, 2017 Touqeer Ahmed PhD Atta-ur-Rahman School of Applied Biosciences National University of Sciences and Technology Excitatory Synaptic Actions Excitatory Synaptic Action
Synaptic Integration 3 rd January, 2017 Touqeer Ahmed PhD Atta-ur-Rahman School of Applied Biosciences National University of Sciences and Technology Excitatory Synaptic Actions Excitatory Synaptic Action
Membrane Structure, Resting membrane potential, Action potential. Biophysics seminar
 Membrane Structure, Resting membrane potential, Action potential Biophysics seminar 09.09.2013. Membrane structure Biological membranes consists of lipids and proteins to bind with non-covalent bond. Phospholipids
Membrane Structure, Resting membrane potential, Action potential Biophysics seminar 09.09.2013. Membrane structure Biological membranes consists of lipids and proteins to bind with non-covalent bond. Phospholipids
Local Anesthetics. Xiaoping Du Room E417 MSB Department of Pharmacology Phone (312) ;
 Local Anesthetics Xiaoping Du Room E417 MSB Department of Pharmacology Phone (312)355 0237; Email: xdu@uic.edu Summary: Local anesthetics are drugs used to prevent or relieve pain in the specific regions
Local Anesthetics Xiaoping Du Room E417 MSB Department of Pharmacology Phone (312)355 0237; Email: xdu@uic.edu Summary: Local anesthetics are drugs used to prevent or relieve pain in the specific regions
The Journal of Neuroscience, August 15, 2001, 21(16):
 The Journal of Neuroscience, August 15, 2001, 21(16):5952 5961 Nav1.3 Sodium Channels: Rapid Repriming and Slow Closed-State Inactivation Display Quantitative Differences after Expression in a Mammalian
The Journal of Neuroscience, August 15, 2001, 21(16):5952 5961 Nav1.3 Sodium Channels: Rapid Repriming and Slow Closed-State Inactivation Display Quantitative Differences after Expression in a Mammalian
Name: Answer Key. Question 1.
 2007 7.013 Problem Set 6 Due before 5 PM on FRIDAY, April 27, 2007. Turn answers in to the box outside of 68-120. PLEASE WRITE YOUR ANSWERS ON THIS PRINTOUT. Question 1. 1a. This is a diagram showing changes
2007 7.013 Problem Set 6 Due before 5 PM on FRIDAY, April 27, 2007. Turn answers in to the box outside of 68-120. PLEASE WRITE YOUR ANSWERS ON THIS PRINTOUT. Question 1. 1a. This is a diagram showing changes
GABA-induced current and circadian regulation of chloride in neurones of the rat suprachiasmatic nucleus
 12743 Journal of Physiology (2001), 537.3, pp.853 869 853 GABA-induced current and circadian regulation of chloride in neurones of the rat suprachiasmatic nucleus Shlomo Wagner, Noa Sagiv and Yosef Yarom
12743 Journal of Physiology (2001), 537.3, pp.853 869 853 GABA-induced current and circadian regulation of chloride in neurones of the rat suprachiasmatic nucleus Shlomo Wagner, Noa Sagiv and Yosef Yarom
Bear: Neuroscience: Exploring the Brain 3e
 Bear: Neuroscience: Exploring the Brain 3e Chapter 03: The Neuronal Membrane at Rest Introduction Action potential in the nervous system Action potential vs. resting potential Slide 1 Slide 2 Cytosolic
Bear: Neuroscience: Exploring the Brain 3e Chapter 03: The Neuronal Membrane at Rest Introduction Action potential in the nervous system Action potential vs. resting potential Slide 1 Slide 2 Cytosolic
Cellular Neurobiology BIPN140. 1st Midterm Exam October 18 th, Tuesday Material covered: Lectures 1-6 & Reading
 Cellular Neurobiology BIPN140 1st Midterm Exam October 18 th, Tuesday Material covered: Lectures 1-6 & Reading Review session October 17 th 3500 Pacitic Hall, 6-8 pm (access code is 127895) Come with questions!
Cellular Neurobiology BIPN140 1st Midterm Exam October 18 th, Tuesday Material covered: Lectures 1-6 & Reading Review session October 17 th 3500 Pacitic Hall, 6-8 pm (access code is 127895) Come with questions!
Interaction Between the Sodium Channel Inactivation Linker and Domain
 Biophysical Journal Volume 73 October 1997 1885-1895 1 885 Interaction Between the Sodium Channel Inactivation Linker and Domain III S4-S5 Marianne R. Smith and Alan L. Goldin Departments of Microbiology
Biophysical Journal Volume 73 October 1997 1885-1895 1 885 Interaction Between the Sodium Channel Inactivation Linker and Domain III S4-S5 Marianne R. Smith and Alan L. Goldin Departments of Microbiology
THREE TYPES OF VOLTAGE-DEPENDENT CALCIUM CURRENTS DEVELOPING IN CULTURED HUMAN NEUROBLASTOMA CELLS
 ORIGINAL PAPER Nagoya 1. Med. Sci. 62. 39-45, 1999 THREE TYPES OF VOLTAGE-DEPENDENT CALCIUM CURRENTS DEVELOPING IN CULTURED HUMAN NEUROBLASTOMA CELLS MASAO KITO [, MITSUO MAEHARA 2 and KAZUYOSHI WATANABE
ORIGINAL PAPER Nagoya 1. Med. Sci. 62. 39-45, 1999 THREE TYPES OF VOLTAGE-DEPENDENT CALCIUM CURRENTS DEVELOPING IN CULTURED HUMAN NEUROBLASTOMA CELLS MASAO KITO [, MITSUO MAEHARA 2 and KAZUYOSHI WATANABE
Questions. Question 1!
 Questions Question 1 In a laboratory, scientists often study neurons in isolation, outside of a living creature, in a dish. In this setting, one can have a good deal of control over the local ionic environment
Questions Question 1 In a laboratory, scientists often study neurons in isolation, outside of a living creature, in a dish. In this setting, one can have a good deal of control over the local ionic environment
KEVEYIS (dichlorphenamide) oral tablet
 KEVEYIS (dichlorphenamide) oral tablet Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Pharmacy
KEVEYIS (dichlorphenamide) oral tablet Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Pharmacy
Transient Sodium Current at Subthreshold Voltages: Activation by EPSP Waveforms
 Article Transient Sodium Current at Subthreshold Voltages: Activation by EPSP Waveforms Brett C. Carter, 1 Andrew J. Giessel, 2 Bernardo L. Sabatini, 2 and Bruce P. Bean 1, * 1 Department of Neurobiology,
Article Transient Sodium Current at Subthreshold Voltages: Activation by EPSP Waveforms Brett C. Carter, 1 Andrew J. Giessel, 2 Bernardo L. Sabatini, 2 and Bruce P. Bean 1, * 1 Department of Neurobiology,
ANC: Section 1. Block AP - 1 J.Thomas.Mortimer &Narendra Bhadra.
 ANC: Section 1. Block AP - 1 J.Thomas.Mortimer &Narendra Bhadra. Introduction In some clinical applications, it is desirable to arrest or block propagating action potentials and eliminate the possibility
ANC: Section 1. Block AP - 1 J.Thomas.Mortimer &Narendra Bhadra. Introduction In some clinical applications, it is desirable to arrest or block propagating action potentials and eliminate the possibility
nerves of the toad Bufo marinus and voltage clamped as described by Dodge and Frankenhaeuser (7). Neurotoxins
 BRIEF COMMUNICATION SIMULTANEOUS MODIFICATIONS OF SODIUM CHANNEL GATING BY TWO SCORPION TOXINS GING Kuo WANG AND GARY STRICHARTZ Department ofanesthesia Research Laboratories, Harvard Medical School, Boston,
BRIEF COMMUNICATION SIMULTANEOUS MODIFICATIONS OF SODIUM CHANNEL GATING BY TWO SCORPION TOXINS GING Kuo WANG AND GARY STRICHARTZ Department ofanesthesia Research Laboratories, Harvard Medical School, Boston,
Electrophysiology. General Neurophysiology. Action Potentials
 5 Electrophysiology Cochlear implants should aim to reproduce the coding of sound in the auditory system as closely as possible, for best sound perception. The cochlear implant is in part the result of
5 Electrophysiology Cochlear implants should aim to reproduce the coding of sound in the auditory system as closely as possible, for best sound perception. The cochlear implant is in part the result of
The Nondystrophic Myotonias
 Neurotherapeutics: The Journal of the American Society for Experimental NeuroTherapeutics The Nondystrophic Myotonias Chad R. Heatwole* and Richard T. Moxley III* *Department of Neurology and Department
Neurotherapeutics: The Journal of the American Society for Experimental NeuroTherapeutics The Nondystrophic Myotonias Chad R. Heatwole* and Richard T. Moxley III* *Department of Neurology and Department
Voltage-gated Na channels are essential for the generation
 UltraRapid Communication Isoform-Specific Modulation of Voltage-Gated Na Channels by Calmodulin Isabelle Deschênes, Nathalie Neyroud, Deborah DiSilvestre, Eduardo Marbán, David T. Yue, Gordon F. Tomaselli
UltraRapid Communication Isoform-Specific Modulation of Voltage-Gated Na Channels by Calmodulin Isabelle Deschênes, Nathalie Neyroud, Deborah DiSilvestre, Eduardo Marbán, David T. Yue, Gordon F. Tomaselli
Sodium channel isoform-speci c e ects of halothane: protein kinase C co-expression and slow inactivation gating
 British Journal of Pharmacology (2000) 130, 1785 ± 1792 ã 2000 Macmillan Publishers Ltd All rights reserved 0007 ± 1188/00 $15.00 www.nature.com/bjp Sodium channel isoform-speci c e ects of halothane:
British Journal of Pharmacology (2000) 130, 1785 ± 1792 ã 2000 Macmillan Publishers Ltd All rights reserved 0007 ± 1188/00 $15.00 www.nature.com/bjp Sodium channel isoform-speci c e ects of halothane:
SUPPLEMENTARY INFORMATION
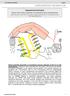 doi: 1.138/nature588 SUPPLEMENTARY INFORMATION Supplemental Information Sensory neuron sodium channel Na v 1.8 is essential for pain at cold temperatures Katharina Zimmermann*, Andreas Leffler*, Alexandru
doi: 1.138/nature588 SUPPLEMENTARY INFORMATION Supplemental Information Sensory neuron sodium channel Na v 1.8 is essential for pain at cold temperatures Katharina Zimmermann*, Andreas Leffler*, Alexandru
The Contribution of Resurgent Sodium Current to High- Frequency Firing in Purkinje Neurons: An Experimental and Modeling Study
 The Journal of Neuroscience, June 15, 2003 23(12):4899 4912 4899 Cellular/Molecular The Contribution of Resurgent Sodium Current to High- Frequency Firing in Purkinje Neurons: An Experimental and Modeling
The Journal of Neuroscience, June 15, 2003 23(12):4899 4912 4899 Cellular/Molecular The Contribution of Resurgent Sodium Current to High- Frequency Firing in Purkinje Neurons: An Experimental and Modeling
Clinical Neurophysiology
 Clinical Neurophysiology 2013 Dept. of Neurology Basic and clinical Neurophysiology Basic neurophysiology Identify mechanisms that make the nervous system work Molecules, cells, groups of cells, functional
Clinical Neurophysiology 2013 Dept. of Neurology Basic and clinical Neurophysiology Basic neurophysiology Identify mechanisms that make the nervous system work Molecules, cells, groups of cells, functional
