CLINICAL INVESTIGATION OF ORAL CONTRACEPTIVES
|
|
|
- Andrew Parks
- 5 years ago
- Views:
Transcription
1 CLINICAL INVESTIGATION OF ORAL CONTRACEPTIVES Guideline Title Clinical Investigation of Oral Contraceptives Legislative basis Directive 75/318/EEC as amended Date of first adoption February 1987 Date of entry into August 1987 force Status Last revised 1987 Previous titles/other None references Additional Notes This note for guidance is intended to assist applicants for a marketing authorisation in the interpretation of Directive 75/318/EEC as amended with respect to specific clinical problems involved in establishing the safety and efficacy of oral contraceptives. A contraceptive preparation consisting of two or more components should also be investigated so as to elucidate the points listed in the note for guidance on Fixed-Combination Medicinal Products. CONTENTS 1. GENERAL 2. CLINICAL-PHARMACOLOGICAL STUDIES 3. CLINICAL INVESTIGATIONS OF EFFICACY AND SAFETY 341
2 CLINICAL INVESTIGATION OF ORAL CONTRACEPTIVES 1. GENERAL This note for guidance should be read in the light of the annex to Directive 75/318/EEC as amended as well as Directive 65/65/EEC as amended; it is intended solely to assist applicants in the interpretation of these documents with respect to the specific clinical problems involved in establishing the safety and efficacy of oral contraceptives. This note has been compiled primarily with those contraceptives in mind which have hormonal activity and are administered orally to women. It is evident that for other contraceptive products subject to medicines legislation the investigational approach required to assess efficacy and safety will be analogous but not necessarily identical. A contraceptive preparation consisting of two or more components should also be investigated so as to elucidate the points listed in the note for guidance on Fixed Combination Medicinal Products, i.e. the properties of the individual components and their contribution to the total effect should be known. Where a new contraceptive can be considered as a modification of one already recognised as being efficacious and safe (particularly where there is merely a minor change in dosage, or where one oestrogen is replaced by another accepted oestrogen) it may be possible to simplify the investigations provided the theoretical basis for the new formulation appears to be sound. 2. CLINICAL-PHARMACOLOGICAL STUDIES Clinical-pharmacological studies performed with an oral contraceptive (and in the case of a fixed combination also with its components) are most likely to meet the standards set by the annex to Directive 75/318/EEC as amended if they are designed to provide data on: a) the pharmacological action or actions in man by virtue of which a contraceptive effect is attained; b) other pharmacological effects on the reproductive system and process, including those on hypothalamic and pituitary activity, ovarian endocrine secretion, ovulation, endometrial histology and biochemical activity, cervical mucus and vaginal cytology and secretion. Effects on tubal function, which cannot currently be studied in human subjects, can be investigated in animals; c) the degree of progestogenic, oestrogenic, androgenic, corticosteroid and other hormonal or anti-hormonal activity of the product and its components in man. It is appreciated that the quantitative study of some of these effects (particularly androgenicity and antiandrogenicity) in the clinical pharmacological phase may be difficult, but conclusions on these points may also be drawn from animal studies and from adverse reactions occurring during efficacy investigations; d) the nature and hormonal activity of the principal metabolites; e) suspected medicinal product interactions likely to impair the efficacy of the product; 343
3 3CC25a f ) those adverse effects likely to be detectable with products of this type even in a limited population, including those involving liver function, adrenal activity, lipid and carbohydrate metabolism, the thyroid and haemostatic mechanisms. 3. CLINICAL INVESTIGATIONS OF EFFICACY AND SAFETY Clinical studies with an oral contraceptive are most likely to meet the standards set by the annex to Directive 75/318/EEC as amended if the following principles are borne in mind: a) Trial population The population studied should be reasonably comparable to that in the country or countries i n which it is proposed to introduce the product. It should be realised that dietary habits, endemic diseases, body weight, illiteracy, etc. can substantially affect the results obtained with a given contraceptive method. b ) Scope of trials The clinical investigation should be sufficiently large to render possible a reliable calculation of efficacy (in terms of the Pearl Index and the Life Table method) and of the incidence of adverse reactions. In practice it generally proves desirable for an entirely new contraceptive product (e.g. one incorporating a new progestagen) to study some cycles of treatment. Where a modification of an existing product is investigated, valid conclusions may sometimes be drawn from more limited material. Since both the effect of the product and the degree of precision with which it is used may alter during long-term use, a substantial part of the total population studied should have used the contraceptive for a period of not less than 12 months, e.g. about a quarter of the total data available should relate to prolonged use. Although any large-scale study of an oral contraceptive will as a rule have to be conducted on a multicentre basis, only data from those centres which have gained substantial experience with the product should be included in the total analysis. c ) Studies on admission History-taking and clinical examination at the time of admission to the trial should provide a detailed record of any risk factors, relative contra-indications or other elements which may subsequently be relevant to the assessment of efficacy and supposed adverse effects, e.g.: age, obesity, smoking habits, alcoholism, cardiovascular disease, psychic disorders or symptoms, migraine, endocrine and metabolic disorders, epilepsy, anaemia, 344
4 disorders of the haemostatic system, renal disorders, hepatic disorders, tumours. The prior obstetric, gynaecological and contraceptive history should be known and recent and current medicinal product intake recorded. The findings on certain of the above points will clearly lead to the exclusion of some subjects from the trial. d ) Recording of data It is recommended that in a contraceptive study individual patient data be recorded on a well-recognised form, such as that advocated by the World Health Organisation. Trial subjects should be seen by investigators at intervals of not more than three months. In all subjects taking part in such studies there should be a periodic gynaecological examination including cervical smear, as well as examination of the breast, weight and blood pressure, a test of glycosuria, and a close record of the menstrual history and any suspected adverse reactions. Intercurrent illness as well as adverse events noted by the patient should be recorded and any change in libido registered. In a subgroup detailed laboratory examinations should be performed to detect any change i n the normal endometrium, hepatic function, lipid metabolism, haematological parameters, protein spectrum, serum electrolytes urine composition, adrenal activity, carbohydrate metabolism, and any parameter an effect on which might be anticipated in view of the pharmacological and toxicological findings. Where clear abnormalities are detected, the patient should be examined clinically and the findings recorded, irrespective of whether the subject continues to take part in the trial or not. Any patients admitted to the study despite the presence of certain risk factors or functional disorders at the time of admission, should undergo regular re-examination in these respects during the trial. In cases of contraceptive failure, data on the pregnancy and the condition of the neonate or embryo should be recorded and the possibility of patient failure assessed. Where a subject withdraws from the study the reasons for withdrawal should be recorded and where possible the subject should be followed up to determine the time of resumption of menstruation and fertility and any possible effect on subsequent pregnancy. Follow-up. Any subject who has shown significant variation in metabolic functions should be followed up to determine if and when these return to normal after termination of the trial. e ) Analysis of data i) General Data relating to efficacy, cycle control, adverse reactions and laboratory findings should be presented for the investigational programme as a whole and for the individual studies and also analysed to try and find correlations with factors likely to be capable of affecting the findings. ii) Efficacy If pregnancies have occurred during the study, a detailed analysis of each individual case should be presented. 345
5 3CC25a iii) iv) Cycle control Data on cycle control should be recorded and presented in such a manner that the incidence and severity of menstrual irregularity, spotting, breakthrough bleeding and amenorrhoea are clear, and so that any variation therein between individuals or over a period of time can be discerned. It is useful to indicate to what extent such events have been regarded as acceptable by the trial subjects and the investigators. Laboratory findings Abnormal laboratory findings should be analysed, inter alia, with respect to any possible correlations with clinical findings in the subjects concerned. f ) Absolute and relative efficacy and safety An oral contraceptive is likely to be regarded as effective if the degree of contraceptive efficacy attained when the product is employed under field conditions by a normal population is not less than that currently attained with other contraceptive methods which have obtained wide acceptance. Some lesser degree of efficacy may be acceptable if it is outweighed by advantages in terms of safety and tolerance, and provided the chances of contraceptive failure can be quantified and are clearly explained in the texts made available to the user. An oral contraceptive is likely to be regarded as not harmful if its adverse effects are not more severe or prolonged than those of oral contraceptives in current use, and provided that it does not result in persistent derangement of the menstrual bleeding pattern during longterm use or persistent changes after withdrawal. g) Post-marketing studies Whilst Directives 65/65/EEC as amended and 75/318/EEC as amended impose no requirements that post-marketing studies be conducted, applicants proposing to market oral contraceptives of a new type are urged to consider the possibility of continuing long-term clinical investigations and monitoring subsequent to introduction of the products concerned; this will greatly facilitate the assessment of reports on supposed adverse reactions. 346
CLINICAL INVESTIGATION OF ANTI-ANGINAL MEDICINAL PRODUCTS IN STABLE ANGINA PECTORIS
 CLINICAL INVESTIGATION OF ANTI-ANGINAL MEDICINAL PRODUCTS IN STABLE ANGINA PECTORIS Guideline Title Clinical Investigation of Anti-Anginal Medicinal Products in Stable Angina Pectoris Legislative basis
CLINICAL INVESTIGATION OF ANTI-ANGINAL MEDICINAL PRODUCTS IN STABLE ANGINA PECTORIS Guideline Title Clinical Investigation of Anti-Anginal Medicinal Products in Stable Angina Pectoris Legislative basis
DOSE SELECTION FOR CARCINOGENICITY STUDIES OF PHARMACEUTICALS *)
 DOSE SELECTION FOR CARCINOGENICITY STUDIES OF PHARMACEUTICALS *) Guideline Title Dose Selection for Carcinogenicity Studies of Pharmaceuticals *) Legislative basis Directive 75/318/EEC as amended Date
DOSE SELECTION FOR CARCINOGENICITY STUDIES OF PHARMACEUTICALS *) Guideline Title Dose Selection for Carcinogenicity Studies of Pharmaceuticals *) Legislative basis Directive 75/318/EEC as amended Date
CLINICAL INVESTIGATION OF MEDICINAL PRODUCTS IN GERIATRICS *)
 3CC9a CLINICAL INVESTIGATION OF MEDICINAL PRODUCTS IN GERIATRICS *) Guideline Title Clinical Investigation of Medicinal Products in Geriatrics*) Legislative basis Directive 75/318/EEC, as amended Date
3CC9a CLINICAL INVESTIGATION OF MEDICINAL PRODUCTS IN GERIATRICS *) Guideline Title Clinical Investigation of Medicinal Products in Geriatrics*) Legislative basis Directive 75/318/EEC, as amended Date
12/13/2017. Important references for PCOS. Polycystic Ovarian Syndrome (PCOS) for the Family Physician. 35 year old obese woman
 Polycystic Ovarian Syndrome (PCOS) for the Family Physician Barbara S. Apgar MD, MS Professor or Family Medicine University of Michigan Ann Arbor, Michigan Important references for PCOS Endocrine Society
Polycystic Ovarian Syndrome (PCOS) for the Family Physician Barbara S. Apgar MD, MS Professor or Family Medicine University of Michigan Ann Arbor, Michigan Important references for PCOS Endocrine Society
Levosert levonorgestrel 20mcg/24hour intrauterine device
 Levosert levonorgestrel 20mcg/24hour intrauterine device Verdict: Formulary inclusion: Formulary category: Restrictions: Reason for inclusion: Link to formulary: Link to medicine review summary: Levosert
Levosert levonorgestrel 20mcg/24hour intrauterine device Verdict: Formulary inclusion: Formulary category: Restrictions: Reason for inclusion: Link to formulary: Link to medicine review summary: Levosert
Official Journal of the European Union. (Non-legislative acts) REGULATIONS
 21.2.2015 L 50/1 II (Non-legislative acts) REGULATIONS COMMISSION REGULATION (EU) 2015/282 of 20 February 2015 amending Annexes VIII, IX and X to Regulation (EC) No 1907/2006 of the European Parliament
21.2.2015 L 50/1 II (Non-legislative acts) REGULATIONS COMMISSION REGULATION (EU) 2015/282 of 20 February 2015 amending Annexes VIII, IX and X to Regulation (EC) No 1907/2006 of the European Parliament
The 28-day pack (Every-Day pack) contains 21 hormonal tablets, 6 tablets each with gestodene (17αethinyl-13-ethyl-17β-hydroxy-4,15-gonadiene-3-one)
 REGISTERED PACKAGE INSERT SCHEDULING STATUS S3 PROPRIETARY NAME AND DOSAGE FORM TRIODENE ED Tablets COMPOSITION The 28-day pack (Every-Day pack) contains 21 hormonal tablets, 6 tablets each with gestodene
REGISTERED PACKAGE INSERT SCHEDULING STATUS S3 PROPRIETARY NAME AND DOSAGE FORM TRIODENE ED Tablets COMPOSITION The 28-day pack (Every-Day pack) contains 21 hormonal tablets, 6 tablets each with gestodene
INFERTILITY CAUSES. Basic evaluation of the female
 INFERTILITY Infertility is the inability to conceive after 12 months of unprotected intercourse. There are multiple causes of infertility and a systematic way to evaluate the condition. Let s look at some
INFERTILITY Infertility is the inability to conceive after 12 months of unprotected intercourse. There are multiple causes of infertility and a systematic way to evaluate the condition. Let s look at some
CURRENT HORMONAL CONTRACEPTION - LIMITATIONS
 CURRENT HORMONAL CONTRACEPTION - LIMITATIONS Oral Contraceptives - Features MERITS Up to 99.9% efficacy if used correctly and consistently Reversible method rapid return of fertility Offer non-contraceptive
CURRENT HORMONAL CONTRACEPTION - LIMITATIONS Oral Contraceptives - Features MERITS Up to 99.9% efficacy if used correctly and consistently Reversible method rapid return of fertility Offer non-contraceptive
SUMMARY OF PRODUCT CHARACTERISTICS FOR BENZODIAZEPINES AS ANXIOLYTICS OR HYPNOTICS
 SUMMARY OF PRODUCT CHARACTERISTICS FOR BENZODIAZEPINES AS ANXIOLYTICS OR HYPNOTICS Guideline Title Summary of Product Characteristics for Benzodiazepines as Anxiolytics or Hypnotics Legislative basis Directive
SUMMARY OF PRODUCT CHARACTERISTICS FOR BENZODIAZEPINES AS ANXIOLYTICS OR HYPNOTICS Guideline Title Summary of Product Characteristics for Benzodiazepines as Anxiolytics or Hypnotics Legislative basis Directive
Menopause management NICE Implementation
 Menopause management NICE Implementation Dr Paula Briggs Consultant in Sexual & Reproductive Health Southport and Ormskirk NHS Hospital Trust Why a NICE guideline (NG 23) Media reports about HRT have not
Menopause management NICE Implementation Dr Paula Briggs Consultant in Sexual & Reproductive Health Southport and Ormskirk NHS Hospital Trust Why a NICE guideline (NG 23) Media reports about HRT have not
Dr Mary Birdsall. Fertility Associates Auckland
 Dr Mary Birdsall Fertility Associates Auckland Period Problems Mary Birdsall Medical Director Fertility Associates Auckland Period Problems Basic Physiology No Periods Irregular Periods Heavy Periods
Dr Mary Birdsall Fertility Associates Auckland Period Problems Mary Birdsall Medical Director Fertility Associates Auckland Period Problems Basic Physiology No Periods Irregular Periods Heavy Periods
Cervical Cancer - Suspected
 Cervical Cancer - Suspected Presentation for patients Asymptomatic presentation Symptomatic presentation History and examination Consider differential diagnoses RED FLAG! Cervix appears normal after examination
Cervical Cancer - Suspected Presentation for patients Asymptomatic presentation Symptomatic presentation History and examination Consider differential diagnoses RED FLAG! Cervix appears normal after examination
MORE THAN FERTILITY SAFEGUARDING YOUR REPRODUCTIVE HEALTH WITH THE BILLINGS OVULATION METHOD
 MORE THAN FERTILITY SAFEGUARDING YOUR REPRODUCTIVE HEALTH WITH THE BILLINGS OVULATION METHOD 1 Introduction If you have heard of the Billings Ovulation Method, you probably associate it with teaching women
MORE THAN FERTILITY SAFEGUARDING YOUR REPRODUCTIVE HEALTH WITH THE BILLINGS OVULATION METHOD 1 Introduction If you have heard of the Billings Ovulation Method, you probably associate it with teaching women
SUBSPECIALIST TRAINING PROGRAMME
 EUROPEAN BOARD AND COLLEGE OF OBSTETRICIANS AND GYNAECOLOGISTS (EBCOG) AND EUROPEAN SOCIETY OF GYNAECOLOGICAL ONCOLOGY (ESGO) SUBSPECIALIST TRAINING PROGRAMME IN GYNAECOLOGICAL ONCOLOGY This Programme
EUROPEAN BOARD AND COLLEGE OF OBSTETRICIANS AND GYNAECOLOGISTS (EBCOG) AND EUROPEAN SOCIETY OF GYNAECOLOGICAL ONCOLOGY (ESGO) SUBSPECIALIST TRAINING PROGRAMME IN GYNAECOLOGICAL ONCOLOGY This Programme
The 6 th Scientific Meeting of the Asia Pacific Menopause Federation
 Abnormal uterine bleeding in the perimenopause Perimenopausal menstrual problems are among the most common causes for family practitioner and specialist referral. Often it is due to the hormone changes
Abnormal uterine bleeding in the perimenopause Perimenopausal menstrual problems are among the most common causes for family practitioner and specialist referral. Often it is due to the hormone changes
DIRECTIVE 2004/24/EC OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL. of 31 March 2004
 30.4.2004 Official Journal of the European Union L 136/85 DIRECTIVE 2004/24/EC OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL of 31 March 2004 amending, as regards traditional herbal medicinal products,
30.4.2004 Official Journal of the European Union L 136/85 DIRECTIVE 2004/24/EC OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL of 31 March 2004 amending, as regards traditional herbal medicinal products,
Polycystic Ovary Syndrome (PCOS)
 Polycystic Ovary Syndrome (PCOS) What are Polycystic Ovaries? Polycystic ovaries are slightly larger than normal ovaries and have twice the number of follicles (small cysts). Polycystic ovaries are very
Polycystic Ovary Syndrome (PCOS) What are Polycystic Ovaries? Polycystic ovaries are slightly larger than normal ovaries and have twice the number of follicles (small cysts). Polycystic ovaries are very
Title of Guideline (must include the word Guideline (not protocol, policy, procedure etc)
 Title of Guideline (must include the word Guideline (not protocol, policy, procedure etc) Guideline for the Investigation and Management of Polycystic Ovary Syndrome Author: Contact Name and Job Title
Title of Guideline (must include the word Guideline (not protocol, policy, procedure etc) Guideline for the Investigation and Management of Polycystic Ovary Syndrome Author: Contact Name and Job Title
Fertility. Assessment and treatment for people with fertility problems. Issued: February NICE clinical guideline 156. guidance.nice.org.
 Fertility Assessment and treatment for people with fertility problems Issued: February 2013 NICE clinical guideline 156 guidance.nice.org.uk/cg156 NICE has accredited the process used by the Centre for
Fertility Assessment and treatment for people with fertility problems Issued: February 2013 NICE clinical guideline 156 guidance.nice.org.uk/cg156 NICE has accredited the process used by the Centre for
Please fill out the following information and have it returned to our office prior to your consultation.
 Please fill out the following information and have it returned to our office prior to your consultation. Patient s Name Partner s Name Address: City: State: Zip: Phone (day#): ( ) (eve#) ( ) (cell) ( )
Please fill out the following information and have it returned to our office prior to your consultation. Patient s Name Partner s Name Address: City: State: Zip: Phone (day#): ( ) (eve#) ( ) (cell) ( )
Annex I. Scientific conclusions and grounds for the variation to the terms of the Marketing Authorisation(s)
 Annex I Scientific conclusions and grounds for the variation to the terms of the Marketing Authorisation(s) 1 Scientific conclusions Taking into account the PRAC Assessment Report on the PSUR(s) for dexamethasone
Annex I Scientific conclusions and grounds for the variation to the terms of the Marketing Authorisation(s) 1 Scientific conclusions Taking into account the PRAC Assessment Report on the PSUR(s) for dexamethasone
Picking the Perfect Pill How to Effectively Choose an Oral Contraceptive
 Focus on CME at Queen s University Picking the Perfect Pill How to Effectively Choose an Oral Contraceptive By Susan Chamberlain, MD, FRCSC There are over 20 oral contraceptive (OC) preparations on the
Focus on CME at Queen s University Picking the Perfect Pill How to Effectively Choose an Oral Contraceptive By Susan Chamberlain, MD, FRCSC There are over 20 oral contraceptive (OC) preparations on the
WWF's RESPONSE TO THE COMMUNITY STRATEGY FOR ENDOCRINE DISRUPTORS
 WWF's RESPONSE TO THE COMMUNITY STRATEGY FOR ENDOCRINE DISRUPTORS WWF WWF is the world s largest and most experienced independent conservation organisation. It has 4.7 million regular supporters and a
WWF's RESPONSE TO THE COMMUNITY STRATEGY FOR ENDOCRINE DISRUPTORS WWF WWF is the world s largest and most experienced independent conservation organisation. It has 4.7 million regular supporters and a
MODUS TABLETS. Medroxyprogesterone Tablets IP
 For the use only of a Registered Medical Practitioner or a Hospital or a Laboratory MODUS TABLETS Medroxyprogesterone Tablets IP QUALITATIVE AND QUANTITATIVE COMPOSITION MODUS TABLET 2.5 mg Each uncoated
For the use only of a Registered Medical Practitioner or a Hospital or a Laboratory MODUS TABLETS Medroxyprogesterone Tablets IP QUALITATIVE AND QUANTITATIVE COMPOSITION MODUS TABLET 2.5 mg Each uncoated
Annex III. Amendments to relevant sections of the Product Information
 Annex III Amendments to relevant sections of the Product Information Note: These amendments to the relevant sections of the Summary of Product Characteristics and package leaflet are the outcome of the
Annex III Amendments to relevant sections of the Product Information Note: These amendments to the relevant sections of the Summary of Product Characteristics and package leaflet are the outcome of the
Biology of fertility control. Higher Human Biology
 Biology of fertility control Higher Human Biology Learning Intention Compare fertile periods in females and males What is infertility? Infertility is the inability of a sexually active, non-contracepting
Biology of fertility control Higher Human Biology Learning Intention Compare fertile periods in females and males What is infertility? Infertility is the inability of a sexually active, non-contracepting
Topics. Periods Menopause & HRT Contraception Vulva problems
 Girls stuff Topics Periods Menopause & HRT Contraception Vulva problems Menorrhagia Excessive menstrual loss occurring with regular or irregular cycles Ovulatory Anovulatory Usual blood loss 30-40ml per
Girls stuff Topics Periods Menopause & HRT Contraception Vulva problems Menorrhagia Excessive menstrual loss occurring with regular or irregular cycles Ovulatory Anovulatory Usual blood loss 30-40ml per
West Hampshire Clinical Commissioning Group Board
 West Hampshire Clinical Commissioning Group Board Date of meeting 25 July 2013 Agenda Item 9 Paper No WHCCG13/089 Priorities Committee Statement Assisted Conception/IVF Key issues An Interim Policy Statement
West Hampshire Clinical Commissioning Group Board Date of meeting 25 July 2013 Agenda Item 9 Paper No WHCCG13/089 Priorities Committee Statement Assisted Conception/IVF Key issues An Interim Policy Statement
Information for you. What is polycystic ovary syndrome? Polycystic ovary syndrome: what it means for your long-term health
 aashara Polycystic ovary syndrome: what it means for your long-term health Information for you Published in February 2005, minor amendments in June 2005 Revised 2009 What is polycystic ovary syndrome?
aashara Polycystic ovary syndrome: what it means for your long-term health Information for you Published in February 2005, minor amendments in June 2005 Revised 2009 What is polycystic ovary syndrome?
Polycystic Ovary Syndrome
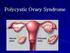 Polycystic Ovary Syndrome Definition: the diagnostic criteria Evidence of hyperandrogenism, biochemical &/or clinical (hirsutism, acne & male pattern baldness). Ovulatory dysfunction; amenorrhoea; oligomenorrhoea
Polycystic Ovary Syndrome Definition: the diagnostic criteria Evidence of hyperandrogenism, biochemical &/or clinical (hirsutism, acne & male pattern baldness). Ovulatory dysfunction; amenorrhoea; oligomenorrhoea
Emergency contraception is an occasional method. It should in no instance replace a regular contraceptive method.
 1. NAME OF THE MEDICINAL PRODUCT: Levonorgestrel Tablets 1.5 mg 2. QUALITATIVE AND QUANTITATIVE COMPOSITION: Each tablet contains levonorgestrel 1.5 mg. Excipient with known effect: Each tablet contains
1. NAME OF THE MEDICINAL PRODUCT: Levonorgestrel Tablets 1.5 mg 2. QUALITATIVE AND QUANTITATIVE COMPOSITION: Each tablet contains levonorgestrel 1.5 mg. Excipient with known effect: Each tablet contains
Contraception and cancerepidemiological
 Contraception and cancerepidemiological evidence Hormonal contraception Combined Progestogen-only Philip C Hannaford University of Aberdeen Breast cancer and combined oral Re-analysis of individual data
Contraception and cancerepidemiological evidence Hormonal contraception Combined Progestogen-only Philip C Hannaford University of Aberdeen Breast cancer and combined oral Re-analysis of individual data
RAISING THE AWARENESS OF GYNAECOLOGICAL CANCER. Penny Bognuda CNS Gynaecologic Oncology ADHB. June 2015.
 RAISING THE AWARENESS OF GYNAECOLOGICAL CANCER Penny Bognuda CNS Gynaecologic Oncology ADHB. June 2015. AIMS AND OBJECTIVES OF THE NEXT ½ HOUR. Brief overview of the different types of gynaecological cancers
RAISING THE AWARENESS OF GYNAECOLOGICAL CANCER Penny Bognuda CNS Gynaecologic Oncology ADHB. June 2015. AIMS AND OBJECTIVES OF THE NEXT ½ HOUR. Brief overview of the different types of gynaecological cancers
Heavy Menstrual Bleeding. Mr Nick Nicholas MD FRCOG Grad Dip Law. Consultant Gynaecologist
 Heavy Menstrual Bleeding Mr Nick Nicholas MD FRCOG Grad Dip Law. Consultant Gynaecologist Why is HMB so important? 1:20 women aged 30-49 consult their GP with HMB Once referred to gynaecologist, surgical
Heavy Menstrual Bleeding Mr Nick Nicholas MD FRCOG Grad Dip Law. Consultant Gynaecologist Why is HMB so important? 1:20 women aged 30-49 consult their GP with HMB Once referred to gynaecologist, surgical
2. Studies of Cancer in Humans
 346 IARC MONOGRAPHS VOLUME 72 2. Studies of Cancer in Humans 2.1 Breast cancer 2.1.1 Results of published studies Eight studies have been published on the relationship between the incidence of breast cancer
346 IARC MONOGRAPHS VOLUME 72 2. Studies of Cancer in Humans 2.1 Breast cancer 2.1.1 Results of published studies Eight studies have been published on the relationship between the incidence of breast cancer
St Helens CCG NHS Funded Treatment for Subfertility Policy 2015/16
 St Helens CCG NHS Funded Treatment for Subfertility Policy 2015/16 1 Standard Operating Procedure St Helens CCG NHS Funded Treatment for Sub Fertility Policy Version 1 Implementation Date May 2015 Review
St Helens CCG NHS Funded Treatment for Subfertility Policy 2015/16 1 Standard Operating Procedure St Helens CCG NHS Funded Treatment for Sub Fertility Policy Version 1 Implementation Date May 2015 Review
PACKAGE LEAFLET TEXT ZOLADEX LA 10.8MG. (goserelin)
 ONC.000-092-861.10.0 PACKAGE LEAFLET TEXT ZOLADEX LA 10.8MG (goserelin) Name of the medicinal product Zoladex LA 10.8mg depot Qualitative and quantitative composition Goserelin acetate (equivalent to 10.8
ONC.000-092-861.10.0 PACKAGE LEAFLET TEXT ZOLADEX LA 10.8MG (goserelin) Name of the medicinal product Zoladex LA 10.8mg depot Qualitative and quantitative composition Goserelin acetate (equivalent to 10.8
Sexual dysfunction of chronic kidney disease. Razieh salehian.md psychiatrist
 Sexual dysfunction of chronic kidney disease Razieh salehian.md psychiatrist Disturbances in sexual function are a common feature of chronic renal failure. Sexual dysfunction is inversely associated with
Sexual dysfunction of chronic kidney disease Razieh salehian.md psychiatrist Disturbances in sexual function are a common feature of chronic renal failure. Sexual dysfunction is inversely associated with
SAMPLE REPORT. Order Number: PATIENT. Age: 40 Sex: F MRN:
 Patient: Age: 40 Sex: F MRN: SAMPLE PATIENT Order Number: Completed: Received: Collected: SAMPLE REPORT Progesterone ng/ml 0.34 0.95 21.00 DHEA-S mcg/dl Testosterone ng/ml 48 35 0.10 0.54 0.80 430 Sex
Patient: Age: 40 Sex: F MRN: SAMPLE PATIENT Order Number: Completed: Received: Collected: SAMPLE REPORT Progesterone ng/ml 0.34 0.95 21.00 DHEA-S mcg/dl Testosterone ng/ml 48 35 0.10 0.54 0.80 430 Sex
Infertility Investigations. Patient Information
 Infertility Investigations Patient Information Author ID: PH Leaflet Number: Gyn 048 Version: 4 Name of Leaflet: Infertility Investigations Date Produced: March 2017 Review Date: March 2019 Please be aware
Infertility Investigations Patient Information Author ID: PH Leaflet Number: Gyn 048 Version: 4 Name of Leaflet: Infertility Investigations Date Produced: March 2017 Review Date: March 2019 Please be aware
Instruction for the patient
 WS 4 Case 3 STI and IUD Your situation Instruction for the patient You are 32 years old, divorced and have one child; you have just started a new relationship You underwent surgical resection of the left
WS 4 Case 3 STI and IUD Your situation Instruction for the patient You are 32 years old, divorced and have one child; you have just started a new relationship You underwent surgical resection of the left
They are updated regularly as new NICE guidance is published. To view the latest version of this NICE Pathway see:
 bring together everything NICE says on a topic in an interactive flowchart. are interactive and designed to be used online. They are updated regularly as new NICE guidance is published. To view the latest
bring together everything NICE says on a topic in an interactive flowchart. are interactive and designed to be used online. They are updated regularly as new NICE guidance is published. To view the latest
5. Summary of Data Reported and Evaluation
 288 5. Summary of Data Reported and Evaluation 5.1 Exposure Oral contraceptives have been used since the early 1960s and are now used by about 90 million women worldwide. The pill is given as a combination
288 5. Summary of Data Reported and Evaluation 5.1 Exposure Oral contraceptives have been used since the early 1960s and are now used by about 90 million women worldwide. The pill is given as a combination
One of the commonest gynecological cancers,especially in white Americans.
 Gynaecology Dr. Rozhan Lecture 6 CARCINOMA OF THE ENDOMETRIUM One of the commonest gynecological cancers,especially in white Americans. It is a disease of postmenopausal women with a peak incidence in
Gynaecology Dr. Rozhan Lecture 6 CARCINOMA OF THE ENDOMETRIUM One of the commonest gynecological cancers,especially in white Americans. It is a disease of postmenopausal women with a peak incidence in
Hormone Replacement Therapy (HRT) Benefits & Risks - The Facts
 Hormone Replacement Therapy (HRT) Benefits & Risks - The Facts HRT is a prescription only treatment that replaces some of the lost oestrogen and progesterone hormones which occur during menopause. It can
Hormone Replacement Therapy (HRT) Benefits & Risks - The Facts HRT is a prescription only treatment that replaces some of the lost oestrogen and progesterone hormones which occur during menopause. It can
Female fertility problems How Chinese medicine may help
 Female fertility problems How Chinese medicine may help Prevalence of fertility problems According to figures issued by the Human Fertility and Embryology Authority (HFEA) in 2006, between 1 in 6 or 7
Female fertility problems How Chinese medicine may help Prevalence of fertility problems According to figures issued by the Human Fertility and Embryology Authority (HFEA) in 2006, between 1 in 6 or 7
Polycystic Ovary Syndrome
 Polycystic Ovary Syndrome Polycystic ovary syndrome (PCOS) is common. It can cause period problems, reduced fertility, excess hair growth, and acne. Many women with PCOS are also overweight. Treatment
Polycystic Ovary Syndrome Polycystic ovary syndrome (PCOS) is common. It can cause period problems, reduced fertility, excess hair growth, and acne. Many women with PCOS are also overweight. Treatment
Female Reproductive Physiology. Dr Raelia Lew CREI, FRANZCOG, PhD, MMed, MBBS Fertility Specialist, Melbourne IVF
 Female Reproductive Physiology Dr Raelia Lew CREI, FRANZCOG, PhD, MMed, MBBS Fertility Specialist, Melbourne IVF REFERENCE Lew, R, Natural History of ovarian function including assessment of ovarian reserve
Female Reproductive Physiology Dr Raelia Lew CREI, FRANZCOG, PhD, MMed, MBBS Fertility Specialist, Melbourne IVF REFERENCE Lew, R, Natural History of ovarian function including assessment of ovarian reserve
Premature Menopause : Diagnosis and Management
 Guideline Number 3 : August 2010 Premature Menopause : Diagnosis and Management Introduction : Premature menopause is a serious condition that affects young women and remains an enigma. The challenges
Guideline Number 3 : August 2010 Premature Menopause : Diagnosis and Management Introduction : Premature menopause is a serious condition that affects young women and remains an enigma. The challenges
Infertility: failure to conceive within one year of unprotected regular sexual intercourse. Primary secondary
 Subfertility Infertility: failure to conceive within one year of unprotected regular sexual intercourse. Primary secondary Infertility affects about 15 % of couples. age of the female. Other factors that
Subfertility Infertility: failure to conceive within one year of unprotected regular sexual intercourse. Primary secondary Infertility affects about 15 % of couples. age of the female. Other factors that
Recommended Interim Policy Statement 150: Assisted Conception Services
 Southampton City Clinical Commissioning Group (CCG) took on commissioning responsibility for Assisted Conception Services from 1 April 2013 for its population and agreed to adopt the interim policy recommendations
Southampton City Clinical Commissioning Group (CCG) took on commissioning responsibility for Assisted Conception Services from 1 April 2013 for its population and agreed to adopt the interim policy recommendations
Evaluation of the Infertile Couple
 Overview and Definition Infertility is defined as the inability of a couple to fall pregnant after one year of unprotected intercourse. Infertility is a very common condition as in any given year about
Overview and Definition Infertility is defined as the inability of a couple to fall pregnant after one year of unprotected intercourse. Infertility is a very common condition as in any given year about
They are updated regularly as new NICE guidance is published. To view the latest version of this NICE Pathway see:
 bring together everything NICE says on a topic in an interactive flowchart. are interactive and designed to be used online. They are updated regularly as new NICE guidance is published. To view the latest
bring together everything NICE says on a topic in an interactive flowchart. are interactive and designed to be used online. They are updated regularly as new NICE guidance is published. To view the latest
Working Document prepared by the Commission services - does not prejudice the Commission's final decision 3/2/2014 COMMISSION STAFF WORKING DOCUMENT
 COMMISSION STAFF WORKING DOCUMENT on certain requirements for FSMPs [Supporting Document for the Expert Group meeting of 7 February 2014] Introduction Following the discussions in the Expert Group meeting
COMMISSION STAFF WORKING DOCUMENT on certain requirements for FSMPs [Supporting Document for the Expert Group meeting of 7 February 2014] Introduction Following the discussions in the Expert Group meeting
Women s Issues in Epilepsy. Esther Bui, Epilepsy Fellow MD, FRCPC
 Women s Issues in Epilepsy Esther Bui, Epilepsy Fellow MD, FRCPC How are women different? Different habitus Different metabolism Different co-morbidities Different psychosocial stigma Different hormonal
Women s Issues in Epilepsy Esther Bui, Epilepsy Fellow MD, FRCPC How are women different? Different habitus Different metabolism Different co-morbidities Different psychosocial stigma Different hormonal
DRAFT Policy for the Provision of NHS funded Gamete Retrieval and Cryopreservation for the Preservation of Fertility
 NHS Birmingham and Solihull Clinical Commissioning Group NHS Sandwell and West Birmingham Clinical Commissioning Group DRAFT Policy for the Provision of NHS funded Gamete Retrieval and Cryopreservation
NHS Birmingham and Solihull Clinical Commissioning Group NHS Sandwell and West Birmingham Clinical Commissioning Group DRAFT Policy for the Provision of NHS funded Gamete Retrieval and Cryopreservation
COMMITTEE FOR MEDICINAL PRODUCTS FOR HUMAN USE (CHMP)
 European Medicines Agency London, 15 November 2007 Doc. Ref. EMEA/CHMP/EWP/517497/2007 COMMITTEE FOR MEDICINAL PRODUCTS FOR HUMAN USE (CHMP) GUIDELINE ON CLINICAL EVALUATION OF MEDICINAL PRODUCTS USED
European Medicines Agency London, 15 November 2007 Doc. Ref. EMEA/CHMP/EWP/517497/2007 COMMITTEE FOR MEDICINAL PRODUCTS FOR HUMAN USE (CHMP) GUIDELINE ON CLINICAL EVALUATION OF MEDICINAL PRODUCTS USED
CASE 41. What is the pathophysiologic cause of her amenorrhea? Which cells in the ovary secrete estrogen?
 CASE 41 A 19-year-old woman presents to her gynecologist with complaints of not having had a period for 6 months. She reports having normal periods since menarche at age 12. She denies sexual activity,
CASE 41 A 19-year-old woman presents to her gynecologist with complaints of not having had a period for 6 months. She reports having normal periods since menarche at age 12. She denies sexual activity,
Fertility care for women diagnosed with cancer
 Saint Mary s Hospital Department of Reproductive Medicine Information for Patients Fertility care for women diagnosed with cancer Contents Page Overview... 2 Our service... 2 Effects of cancer treatment
Saint Mary s Hospital Department of Reproductive Medicine Information for Patients Fertility care for women diagnosed with cancer Contents Page Overview... 2 Our service... 2 Effects of cancer treatment
The 6 th Scientific Meeting of the Asia Pacific Menopause Federation
 Predicting the menopause The menopause marks the end of ovarian follicular activity and is said to have occurred after 12 months amenorrhoea. The average age of the menopause is between 45 and 60 years
Predicting the menopause The menopause marks the end of ovarian follicular activity and is said to have occurred after 12 months amenorrhoea. The average age of the menopause is between 45 and 60 years
Menopause: diagnosis and management NICE guideline NG23. Published November 2015
 Menopause: diagnosis and management NICE guideline NG23 Published November 2015 1 Full title: Menopause : diagnosis and management Available at: http://www.nice.org.uk/guidance/ng23 Guideline published
Menopause: diagnosis and management NICE guideline NG23 Published November 2015 1 Full title: Menopause : diagnosis and management Available at: http://www.nice.org.uk/guidance/ng23 Guideline published
Hormonal Changes Following Low-Dosage Irradiation of Pituitary and Ovaries in Anovulatory Women
 Hormonal Changes Following Low-Dosage Irradiation of Pituitary and Ovaries in Anovulatory Women Further Studies A. E. Rakoff, M.D. Tms PRESENTATION is a second progress report in a long-term study of the
Hormonal Changes Following Low-Dosage Irradiation of Pituitary and Ovaries in Anovulatory Women Further Studies A. E. Rakoff, M.D. Tms PRESENTATION is a second progress report in a long-term study of the
FEMALE MEDICAL HISTORY
 Name: Surname: Date of birth: Dear patient, filling out this questionnaire correctly and completely is very important, because this allows us to assess your situation faster during the consultation and
Name: Surname: Date of birth: Dear patient, filling out this questionnaire correctly and completely is very important, because this allows us to assess your situation faster during the consultation and
COMMISSION REGULATION (EU) / of XXX
 Ref. Ares(2016)5616438-28/09/2016 EUROPEAN COMMISSION Brussels, XXX [ ](2016) XXX draft COMMISSION REGULATION (EU) / of XXX amending Annex III to Directive 2008/98/EC of the European Parliament and of
Ref. Ares(2016)5616438-28/09/2016 EUROPEAN COMMISSION Brussels, XXX [ ](2016) XXX draft COMMISSION REGULATION (EU) / of XXX amending Annex III to Directive 2008/98/EC of the European Parliament and of
Health screening questionnaire
 Health screening questionnaire High Road Buckhurst Hill Essex IG9 5HX Tel: 020 8936 1202 Fax: 020 8936 1191 Visit: theholly.com Title: Surname: Forenames: Date of birth: Age: Address: Tel no. (Home): Tel
Health screening questionnaire High Road Buckhurst Hill Essex IG9 5HX Tel: 020 8936 1202 Fax: 020 8936 1191 Visit: theholly.com Title: Surname: Forenames: Date of birth: Age: Address: Tel no. (Home): Tel
Adrenal Stress Profile (Saliva)
 Adrenal Stress Profile (Saliva) Ireland Cortisol Levels Sample 1 Post Awakening Sample 2 (+ 4-5 Hours) Sample 3 (+ 4-5 Hours) Sample 4 (Prior to Sleep) 11.42 2.10 1.60 0.47 Sum of Cortisol 15.590 DHEA
Adrenal Stress Profile (Saliva) Ireland Cortisol Levels Sample 1 Post Awakening Sample 2 (+ 4-5 Hours) Sample 3 (+ 4-5 Hours) Sample 4 (Prior to Sleep) 11.42 2.10 1.60 0.47 Sum of Cortisol 15.590 DHEA
COMMISSION OF THE EUROPEAN COMMUNITIES REPORT FROM THE COMMISSION TO THE COUNCIL AND THE EUROPEAN PARLIAMENT
 EN EN EN COMMISSION OF THE EUROPEAN COMMUNITIES Brussels, 5.12.2008 COM(2008) 824 final REPORT FROM THE COMMISSION TO THE COUNCIL AND THE EUROPEAN PARLIAMENT on the use of substances other than vitamins
EN EN EN COMMISSION OF THE EUROPEAN COMMUNITIES Brussels, 5.12.2008 COM(2008) 824 final REPORT FROM THE COMMISSION TO THE COUNCIL AND THE EUROPEAN PARLIAMENT on the use of substances other than vitamins
Y11 Homeostasis & Response
 Key words: 1. Homeostasis: Maintaining a constant internal environment 2. Receptor: Detects a change in the environment 3. Effector: Muscle or gland that carries out a response 4. Stimulus: A change in
Key words: 1. Homeostasis: Maintaining a constant internal environment 2. Receptor: Detects a change in the environment 3. Effector: Muscle or gland that carries out a response 4. Stimulus: A change in
Annex I. Scientific conclusions and grounds for the variation to the terms of the Marketing Authorisation(s)
 Annex I Scientific conclusions and grounds for the variation to the terms of the Marketing Authorisation(s) 1 Scientific conclusions Taking into account the PRAC Assessment Report on the PSUR(s) for sodium
Annex I Scientific conclusions and grounds for the variation to the terms of the Marketing Authorisation(s) 1 Scientific conclusions Taking into account the PRAC Assessment Report on the PSUR(s) for sodium
Annex III. Amendments to relevant sections of the summary of product characteristics and package leaflets
 Annex III Amendments to relevant sections of the summary of product characteristics and package leaflets Note: These amendments to the relevant sections of the Summary of Product Characteristics and package
Annex III Amendments to relevant sections of the summary of product characteristics and package leaflets Note: These amendments to the relevant sections of the Summary of Product Characteristics and package
NHS FUNDED TREATMENT FOR SUBFERTILITY. ELIGIBILITY CRITERIA POLICY GUIDANCE/OPTIONS FOR CCGs
 NHS FUNDED TREATMENT FOR SUBFERTILITY ELIGIBILITY CRITERIA POLICY GUIDANCE/OPTIONS FOR CCGs CONTENTS Page 1. INTRODUCTION 2 2. GENERAL PRINCIPLES 2 3. DEFINITION OF SUBFERTILITY AND TIMING OF ACCESS TO
NHS FUNDED TREATMENT FOR SUBFERTILITY ELIGIBILITY CRITERIA POLICY GUIDANCE/OPTIONS FOR CCGs CONTENTS Page 1. INTRODUCTION 2 2. GENERAL PRINCIPLES 2 3. DEFINITION OF SUBFERTILITY AND TIMING OF ACCESS TO
REPRODUCTION The diagram below shows a section through seminiferous tubules in a testis.
 1. The diagram below shows a section through seminiferous tubules in a testis. Which cell produces testosterone? 2. A function of the interstitial cells in the testes is to produce A sperm B testosterone
1. The diagram below shows a section through seminiferous tubules in a testis. Which cell produces testosterone? 2. A function of the interstitial cells in the testes is to produce A sperm B testosterone
Contraceptive case studies. Dr Christine Roke National Medical Advisor Family Planning June 2015
 Contraceptive case studies Dr Christine Roke National Medical Advisor Family Planning June 2015 Case 1 Mary is a 47 year old who has come in for a routine cervical smear. She asks when her Multiload IUD
Contraceptive case studies Dr Christine Roke National Medical Advisor Family Planning June 2015 Case 1 Mary is a 47 year old who has come in for a routine cervical smear. She asks when her Multiload IUD
By Dr Rukhsana Hussain 5 th April 2016
 By Dr Rukhsana Hussain 5 th April 2016 1. Is it indicated? Vasomotor symptoms (flushes/sweats) Vaginal atrophy Cycle control (In perimenopause consider low dose COCP (age
By Dr Rukhsana Hussain 5 th April 2016 1. Is it indicated? Vasomotor symptoms (flushes/sweats) Vaginal atrophy Cycle control (In perimenopause consider low dose COCP (age
Hormone Treatments and the Risk of Breast Cancer
 Cornell University Program on Breast Cancer and Environmental Risk Factors in New York State (BCERF) Updated July 2002 Hormone Treatments and the Risk of Breast Cancer 1) Hormone Treatment After Menopause
Cornell University Program on Breast Cancer and Environmental Risk Factors in New York State (BCERF) Updated July 2002 Hormone Treatments and the Risk of Breast Cancer 1) Hormone Treatment After Menopause
PCOS and Obesity DUB is better treated by OCPs
 PCOS and Obesity DUB is better treated by OCPs Dr. Ritu Joshi Senior consultant Fortis escorts Hospital, Jaipur Chairperson Family welfare com. FOGSI (20092012) Vice President FOGSI 2014 Introduction One
PCOS and Obesity DUB is better treated by OCPs Dr. Ritu Joshi Senior consultant Fortis escorts Hospital, Jaipur Chairperson Family welfare com. FOGSI (20092012) Vice President FOGSI 2014 Introduction One
Clinical Study Synopsis
 Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Polycystic ovary syndrome (PCOS)
 Information for patients Polycystic ovary syndrome (PCOS) What is polycystic ovary syndrome? Polycystic ovary syndrome (PCOS) is a condition which can affect a woman s menstrual cycle, fertility, hormones
Information for patients Polycystic ovary syndrome (PCOS) What is polycystic ovary syndrome? Polycystic ovary syndrome (PCOS) is a condition which can affect a woman s menstrual cycle, fertility, hormones
BACKGROUNDER. Pregnitude A Fertility Dietary Supplement for Reproductive Support Offers an Additional Option for Women Who Are Trying to Conceive
 BACKGROUNDER Pregnitude A Fertility Dietary Supplement for Reproductive Support Offers an Additional Option for Women Who Are Trying to Conceive Studies find that increased chances of ovulation, menstrual
BACKGROUNDER Pregnitude A Fertility Dietary Supplement for Reproductive Support Offers an Additional Option for Women Who Are Trying to Conceive Studies find that increased chances of ovulation, menstrual
FEMALE PATIENT HISTORY
 ew Hope. ew Life. ew Beginnings. A Division of MID-ATLATIC WOME S CARE, PLC FEMALE PATIET HISTORY PLEASE OTE: Infertility patients please complete ALL sections. All other patients, complete section 1.,
ew Hope. ew Life. ew Beginnings. A Division of MID-ATLATIC WOME S CARE, PLC FEMALE PATIET HISTORY PLEASE OTE: Infertility patients please complete ALL sections. All other patients, complete section 1.,
Medical treatment for uterine fibroids
 Medical treatment for uterine fibroids Prof Mary Ann Lumsden Prof of Gynaecology and Medical Education University of Glasgow Senior Vice President RCOG Conflict of Interest Chair, Guideline development
Medical treatment for uterine fibroids Prof Mary Ann Lumsden Prof of Gynaecology and Medical Education University of Glasgow Senior Vice President RCOG Conflict of Interest Chair, Guideline development
EVALUATION OF MALE AND FEMALE INFERTILITY ANDREA BARRUECO AMERICAN CENTER FOR REPRODUCTIVE MEDICINE CLEVELAND CLINIC ART TRAINING 2018
 EVALUATION OF MALE AND FEMALE INFERTILITY ANDREA BARRUECO AMERICAN CENTER FOR REPRODUCTIVE MEDICINE CLEVELAND CLINIC ART TRAINING 2018 The evaluation of an infertile couple requires an understanding of
EVALUATION OF MALE AND FEMALE INFERTILITY ANDREA BARRUECO AMERICAN CENTER FOR REPRODUCTIVE MEDICINE CLEVELAND CLINIC ART TRAINING 2018 The evaluation of an infertile couple requires an understanding of
COUNCIL OF THE EUROPEAN UNION. Brussels, 7 September 2009 (OR. en) 11261/09 Interinstitutional File: 2008/0002 (COD) DENLEG 51 CODEC 893
 COUNCIL OF THE EUROPEAN UNION Brussels, 7 September 2009 (OR. en) 11261/09 Interinstitutional File: 2008/0002 (COD) DLEG 51 CODEC 893 LEGISLATIVE ACTS AND OTHER INSTRUMTS Subject: Common Position with
COUNCIL OF THE EUROPEAN UNION Brussels, 7 September 2009 (OR. en) 11261/09 Interinstitutional File: 2008/0002 (COD) DLEG 51 CODEC 893 LEGISLATIVE ACTS AND OTHER INSTRUMTS Subject: Common Position with
Oral Contraceptives. Mike Williams GPST2
 Oral Contraceptives Mike Williams GPST2 Curriculum Mechanism Efficacy Advantages/Disadvantages Starting Continuing Problems/complications Contraception: effectiveness rates, risks, benefits and appropriate
Oral Contraceptives Mike Williams GPST2 Curriculum Mechanism Efficacy Advantages/Disadvantages Starting Continuing Problems/complications Contraception: effectiveness rates, risks, benefits and appropriate
Birth Control- an Overview. Keith Merritt, MD. Remember, all methods of birth control are safer and have fewer side effects than pregnancy
 Birth Control- an Overview Keith Merritt, MD Basics Remember, all methods of birth control are safer and have fewer side effects than pregnancy Even with perfect use, each method of birth control has a
Birth Control- an Overview Keith Merritt, MD Basics Remember, all methods of birth control are safer and have fewer side effects than pregnancy Even with perfect use, each method of birth control has a
Reproductive System. Testes. Accessory reproductive organs. gametogenesis hormones. Reproductive tract & Glands
 Reproductive System Testes gametogenesis hormones Accessory reproductive organs Reproductive tract & Glands transport gametes provide nourishment for gametes Hormonal regulation in men Hypothalamus - puberty
Reproductive System Testes gametogenesis hormones Accessory reproductive organs Reproductive tract & Glands transport gametes provide nourishment for gametes Hormonal regulation in men Hypothalamus - puberty
ASSISTED CONCEPTION NHS FUNDED TREATMENT FOR SUBFERTILITY ELIGIBILITY CRITERIA & POLICY GUIDANCE
 ASSISTED CONCEPTION NHS FUNDED TREATMENT FOR SUBFERTILITY ELIGIBILITY CRITERIA & POLICY GUIDANCE Version 1.0 Page 1 of 11 MARCH 2014 POLICY DOCUMENT VERSION CONTROL CERTIFICATE TITLE Title: Assisted Conception
ASSISTED CONCEPTION NHS FUNDED TREATMENT FOR SUBFERTILITY ELIGIBILITY CRITERIA & POLICY GUIDANCE Version 1.0 Page 1 of 11 MARCH 2014 POLICY DOCUMENT VERSION CONTROL CERTIFICATE TITLE Title: Assisted Conception
Migraine: Developing Drugs for Acute Treatment Guidance for Industry
 Migraine: Developing Drugs for Acute Treatment Guidance for Industry U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER) February 2018
Migraine: Developing Drugs for Acute Treatment Guidance for Industry U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER) February 2018
trans-1-[4-(2-dimethylaminoethoxy)phenyl]1,2-diphenyl-1-butene
![trans-1-[4-(2-dimethylaminoethoxy)phenyl]1,2-diphenyl-1-butene trans-1-[4-(2-dimethylaminoethoxy)phenyl]1,2-diphenyl-1-butene](/thumbs/95/124190064.jpg) Genox Tamoxifen citrate PRODUCT INFORMATION NAME OF THE MEDICINE Active ingredient: Chemical name: Tamoxifen (as citrate) trans-1-[4-(2-dimethylaminoethoxy)phenyl]1,2-diphenyl-1-butene Structural formula:
Genox Tamoxifen citrate PRODUCT INFORMATION NAME OF THE MEDICINE Active ingredient: Chemical name: Tamoxifen (as citrate) trans-1-[4-(2-dimethylaminoethoxy)phenyl]1,2-diphenyl-1-butene Structural formula:
Infertility DR. RAHUL BEVARA
 Infertility DR. RAHUL BEVARA Definitions Infertility is defined as the inability to conceive after one year of unprotected coitus. Affects 10-15% of couples Primary Infertility, that is inability to conceive
Infertility DR. RAHUL BEVARA Definitions Infertility is defined as the inability to conceive after one year of unprotected coitus. Affects 10-15% of couples Primary Infertility, that is inability to conceive
 Understanding ovarian cancer An information sheet for women with ovarian cancer, their family and friends If you or someone you love has recently been diagnosed with ovarian cancer, you re sure to have
Understanding ovarian cancer An information sheet for women with ovarian cancer, their family and friends If you or someone you love has recently been diagnosed with ovarian cancer, you re sure to have
Fact Sheet. Quick guide to infertility and treatment options
 Fact Sheet Quick guide to infertility and treatment options www.ptafertility.co.za info@ptafertility.co.za +27 12 998 8854 Ovarian Hyper stimulation syndrome OHSS is a potentially life threatening complication
Fact Sheet Quick guide to infertility and treatment options www.ptafertility.co.za info@ptafertility.co.za +27 12 998 8854 Ovarian Hyper stimulation syndrome OHSS is a potentially life threatening complication
Unit 2 Physiology and Health Part (a) The Reproductive System HOMEWORK BOOKLET
 Unit 2 Physiology and Health Part (a) The Reproductive System HOMEWORK BOOKLET Name: Homework Date Due Mark % Key Area 1 The structure and function of reproductive organs Key Area 2 Hormonal control of
Unit 2 Physiology and Health Part (a) The Reproductive System HOMEWORK BOOKLET Name: Homework Date Due Mark % Key Area 1 The structure and function of reproductive organs Key Area 2 Hormonal control of
Y11 Homeostasis & Response
 Key words: 1. Homeostasis: Maintaining a constant internal environment 2. Receptor: Detect a change in the environment 3. Effector: Muscle or gland that carries out a response 4. Stimulus: A change in
Key words: 1. Homeostasis: Maintaining a constant internal environment 2. Receptor: Detect a change in the environment 3. Effector: Muscle or gland that carries out a response 4. Stimulus: A change in
Dr Catherine Black. Head of WOOMB NZ
 Dr Catherine Black Head of WOOMB NZ HRT AND NATURAL FERTILITY REGULATION Dr Catherine Black MB ChB DCH FRNZCGP Head of WOOMB NZ Summary Physiology of menopause Managing fertility Symptoms of menopause
Dr Catherine Black Head of WOOMB NZ HRT AND NATURAL FERTILITY REGULATION Dr Catherine Black MB ChB DCH FRNZCGP Head of WOOMB NZ Summary Physiology of menopause Managing fertility Symptoms of menopause
Summary CHAPTER 1. Introduction
 Summary This thesis aims to evaluate the diagnostic work-up in postmenopausal women presenting with abnormal vaginal bleeding. The Society of Dutch Obstetrics and Gynaecology composed a guideline, which
Summary This thesis aims to evaluate the diagnostic work-up in postmenopausal women presenting with abnormal vaginal bleeding. The Society of Dutch Obstetrics and Gynaecology composed a guideline, which
Web Activity: Simulation Structures of the Female Reproductive System
 differentiate. The epididymis is a coiled tube found along the outer edge of the testis where the sperm mature. 3. Testosterone is a male sex hormone produced in the interstitial cells of the testes. It
differentiate. The epididymis is a coiled tube found along the outer edge of the testis where the sperm mature. 3. Testosterone is a male sex hormone produced in the interstitial cells of the testes. It
Primary and secondary amenorrhoea
 BRITISH MEDICAL JOURNAL VOLUME 294 28 MARCH 1987 815 Clinical Aalgorithms Primary and secondary amenorrhoea S FRANKS Amenorrhoea, which is usually defined as no periods for six months or more, occurs in
BRITISH MEDICAL JOURNAL VOLUME 294 28 MARCH 1987 815 Clinical Aalgorithms Primary and secondary amenorrhoea S FRANKS Amenorrhoea, which is usually defined as no periods for six months or more, occurs in
Annex II. Scientific conclusions and grounds for variation to the terms of the marketing authorisations
 Annex II Scientific conclusions and grounds for variation to the terms of the marketing authorisations 12 Scientific conclusions and grounds for variation to the terms of the marketing authorisations The
Annex II Scientific conclusions and grounds for variation to the terms of the marketing authorisations 12 Scientific conclusions and grounds for variation to the terms of the marketing authorisations The
