10/17/2017 CHRONIC MIGRAINES BOTOX: TO INJECT OR NOT INJECT? IN CHRONIC MIGRAINE PROPHYLAXIS OBJECTIVES PATIENT CASE EPIDEMIOLOGY EPIDEMIOLOGY
|
|
|
- Christian Dalton
- 6 years ago
- Views:
Transcription
1 BOTOX: TO INJECT OR NOT INJECT? IN CHRONIC MIGRAINE PROPHYLAXIS OBJECTIVES JENNIFER SHIN, PHARMD PGY2 AMBULATORY CARE PHARMACY RESIDENT COMMUNITYCARE HEALTH CENTERS PHARMACOTHERAPY ROUNDS OCTOBER 20, 2017 State the definition, background, and pathophysiology of chronic migraines (CM) Identify the current guideline recommendations for standard treatment Discuss outcomes from literature about botox and its place in therapy for CM prophylaxis 2 PATIENT CASE RW is a 35 year old female PMH: DM2, HTN, migraines, asthma, and seasonal allergies Current regimen: Sumatriptan 25 mg twice daily PRN migraines Topiramate 100 mg daily CHRONIC MIGRAINES How many of you would say this is an appropriate regimen? 3 4 EPIDEMIOLOGY EPIDEMIOLOGY Ranked 19 th by the World Health Organization (WHO) among causes for years lived with disability Often begins at puberty and most affects those aged between 35 and 45 years More common in women, usually by a factor of about 2: Migraine Research Foundation 5 Current Pain and Headache Reports, 16(1),
2 PATHOPHYSIOLOGY BACKGROUND The Neurology & Headache Treatment Center 7 Divided into two groups based on headache days per month Can present with or without aura Headache attacks Clinical presentation Throbbing headache Nausea, vomiting, diarrhea Lightheadedness Dizziness 8 DEFINITION RISK FACTORS Chronic migraines(cm) is: A headache (HA) occurring on at least 15 days per month For more than 3 months With typical features of migraine on at least 8 days per month Modifiable Obesity Depression Medication overuse Sleep related problems Caffeine overuse Non-modifiable Age Female sex Caucasian race Low educational level/socioeconomic status Head injury American Headache Society 9 Curr Pain Headache Rep Feb; 16(1): DIAGNOSIS: INTERNATIONAL CLASSIFICATION OF HEADACHE DISORDERS DIAGNOSIS: INTERNATIONAL CLASSIFICATION OF HEADACHE DISORDERS 1. At least five attacks fulfilling criteria (2) (4) 2. Headache attacks lasting 4 72 h (untreated or unsuccessfully treated) 3. Headache has at least two of the following four characteristics: a) Unilateral location b) Pulsating quality c) Moderate or severe pain intensity 4. During headache at least one of the following: 1. Nausea and/or vomiting 2. Photophobia and phonophobia 5. Not better accounted for by another ICHD-3 diagnosis. d) Aggravation by or causing avoidance of routine physical activity (e.g. walking or climbing stairs) Headache Classification Committee of the International Headache Society (IHS) 11 Headache Classification Committee of the International Headache Society (IHS) 12 2
3 GOALS OF THERAPY PROPHYLAXIS INITIATION Reduce attack frequency, severity, and disability Reduce reliance on poorly tolerated, ineffective, or unwanted acute pharmacotherapies Improve quality of life Avoid acute headache medication escalation Educate and enable patients to manage their disease to enhance personal control of their migraine Reduce headache-related distress and psychological symptoms Headache Classification Committee of the International Headache Society (IHS) 13 Initiate At least six headache days per month At least four headache days with at least some impairment At least three headache days with severe impairment or requiring bed rest Consider Four to five migraine days per month with normal functioning Two to three migraine days per month with some impairment Two migraine days with severe impairment Not indicated Less than four headache days per month and no impairment Zero or one headache day per month regardless of impairment Neuropsychiatric Disease and Treatment 2013:9 14 OPTIMIZING THERAPY AHS/AAN AND NICE GUIDELINE RECOMMENDATIONS: PROPHYLAXIS MANAGEMENT Start at a low dose Give each preventive medication an adequate trial, > 2 months Reevaluating therapy and follow up is important Choose a drug based on efficacy, patient s preferences, headache profile, the drug s side effects, and the presence or absence of coexisting or comorbid conditions Initiate therapy with medications that have the highest level of evidence-based efficacy Initiate therapy with the lowest effective dose of the drug Avoid interfering medications (e.g., overuse of acute medications) Use of a long-acting formulation may improve compliance 15 Neurology April 24, 2012 vol. 78 no AMERICAN ACADEMY OF NEUROLOGY AND THE AMERICAN HEADACHE SOCIETY GUIDELINES FOR PROPHYLAXIS Antiepileptic drugs Level A Divalproex sodium Topiramate Level B β blockers Propranolol Atenolol Metoprolol Timolol Nadalol Triptans (MRM*) Frovatriptan Naratriptan Antidepressants NSAIDs Herbals/vitamins/minerals *MRM = Menstrual related migraines Petasites Zolmitriptan Amitriptyline Venlafaxine Ibuprofen/Naproxen Magnesium, Riboflavin, Histamines Neurology April 24, 2012 vol. 78 no PATIENT CASE Current regimen: Sumatriptan 25 mg twice daily PRN migraines Topiramate 100 mg daily Headache diary 18 headache days per month 5-6 months Migraine features 10 days per month How many of you would say this is an appropriate regimen? A) True B) False C) Still not sure 18 3
4 BOTOX BACKGROUND ONABOTULINUMTOXINA BOTOX (ONABOTULINUMTOXINA) A potent purified neurotoxin complex produced by anaerobic bacteria Clostridium botulinum Approved October 2010 for prophylaxis in adult patients with CM Dosing: 155 units once every 12 weeks Equally divided and administered bilaterally, into 31 total sites 19 Lexicomp Online, Pediatric & Neonatal Lexi-Drugs, Hudson, Ohio: Lexi-Comp, Inc.; August BOTOX (ONABOTULINUMTOXINA) BOTOX SUSPECTED MECHANISM A. Corrugator: 5 U each side B. Procerus: 5U (one site) C. Frontalis: 10 U each side D. Temporalis: 20 U each side E. Occipitalis: 15 U each side F. Cervical paraspinal: 10 U each side G. Trapezius: 15 U each side Lexicomp Online, Pediatric & Neonatal Lexi-Drugs, Hudson, Ohio: Lexi-Comp, Inc.; August Toxins (Basel) Jul; 7(7): BOTULINUM TOXIN IN THE MANAGEMENT OF CHRONIC MIGRAINE: CLINICAL EVIDENCE AND EXPERIENCE. PREEMPT I (n=679) Primary endpoint: reduction of migraine episodes (non-significant) Significant differences in reduction of headache and migraine days PREEMPT II (n=705) Primary endpoint: confirmed efficacy of botox in reduction of headache days Identical study design, slightly different end points Commonly reported adverse events: neck pain, injection site pain, and eyelid ptosis Led to the approval of botox in prophylaxis of chronic migraine BOTOX: GUIDELINE RECOMMENDATIONS Guidelines of the American Academy of Neurology state that botox is effective and should be offered to patients with CM NICE guidelines recommend botox as a prophylaxis medication treatment for CM in patients who did not respond to at least 3 prior prophylaxis therapies Ther Adv Neurol Disord Feb; 10(2):
5 BOTOX FOR CHRONIC MIGRAINE: SAFE AND EFFECTIVE? WHEN SHOULD WE USE IT? CLINICAL QUESTION ONABOTULINUMTOXINA FOR CHRONIC MIGRAINE: EFFICACY, SAFETY, AND TOLERABILITY IN PATIENTS WHO RECEIVED ALL FIVE TREATMENT CYCLES IN THE PREEMPT CLINICAL PROGRAM AURORA, ET AL (2014) METHODS: AURORA, ET AL METHODS: AURORA, ET AL Objectives: Assess efficacy, safety and tolerability of botox as headache prophylaxis in adults with chronic migraine Phase III Research Evaluating Migraine Prophylaxis Therapy (PREEMPT) 24 week, double-blind, parallel-group, placebo controlled phase followed by a 32-week open-label phase Botox or placebo every 12 weeks for two cycles, followed by botox for three cycles 27 Inclusion Patients that completed all five cycles years with a history of migraine defined by International classification of headache disorders Had to have headache occurring on 15 days/4 weeks, with each day consisting of 4 h of continuous headache, and 50% of headache days being migraine or probable migraine days Experience 4 distinct headache episodes, each lasting 4 h during this period Naïve to botox prior to trial Exclusion Headache prophylactic medication within 4 weeks prior to start date of baseline 28 METHODS: AURORA, ET AL METHODS: AURORA, ET AL Primary endpoint Frequency of headache days at 24 weeks Secondary endpoint Frequency of migraine days Moderate/severe headache days Headache episodes Migraine episodes Acute headache medication intake
6 RESULTS: AURORA, ET AL RESULTS: AURORA, ET AL weeks Mean change from baseline Botox Placebo P value Frequency of HA days <0.001 Frequency of migraine days <0.001 Frequency of moderate/severe HA days Total cumulative HA hours on HA days < <0.001 Frequency of HA episodes <0.001 Frequency of migraine episodes Frequency of acute HA medication intakes < RESULTS: AURORA, ET AL RESULTS: AURORA, ET AL 56 weeks Mean change from baseline Botox Placebo P value Frequency of HA days Frequency of migraine days Frequency of moderate/severe HA days Total cumulative HA hours on HA days Frequency of HA episodes Frequency of migraine episodes Frequency of acute HA medication intakes AUTHOR S CONCLUSIONS CRITICAL APPRAISAL Patients treated earlier with botox had better outcomes at week 56 There is a continued need and cumulative benefit over time with continued prophylaxis Botox is a safe and effective option for use in CM Strengths Well designed and largest trial thus far investigating use of botox in CM Resulted in FDA approval for botox use in CM Revealed earlier treatment with botox had better outcomes Weaknesses Placebo response noted in both studies Lacking report on differences between those with or without medication use headaches
7 METHODS: MATHARU, ET AL UTILIZATION AND SAFETY OF ONABOTULINUMTOXINA FOR THE PROPHYLACTIC TREATMENT OF CHRONIC MIGRAINE MATHARU, ET AL (2017) Objectives: Examine utilization patterns and safety of botox for prophylactic treatment of chronic migraine in routine clinical practice Prospective, observational post-authorization study Data collection First study injection then every 3 months < 52 weeks for utilization < 64 weeks for safety data METHODS RESULTS: MATHARU, ET AL 1400 Inclusion Botox treated and Botox naïve patients recruited by their physicians 18 years or older with a new or established physician diagnosis of CM Decision to initiate or continue treatment was independent of patient s enrollment in the study. Completed at least one botox treatment session Treatment 1 (n=1160) Treatment 2 (n=1034) Treatment 3 (n=885) Treatment 4 (n=712) Treatment 5 (n=210) botox treatment sessions administered total 40 RESULTS: MATHARU, ET AL RESULTS: MATHARU, ET AL n (%) Patients with > 1 adverse event 478 (41.2) Serious adverse events 61 (5.3%) Treatment discontinued due to adverse event 51 (4.4%) Fatal adverse events 2 (0.2%) Patients with > 1 treatment related adverse event 291 (25.1%) Serious treatment related adverse event 1 (< 0.1%) Fatal treatment related adverse events 0 (0%) 41 Adverse events,n (%) Treatment naïve patients (n=556) Overall (n=1160) Neck pain 30 (5.4) 51 (4.4) Eyelid ptosis 24 (4.3) 47 (4.1) Muscular weakness 16 (2.9) 31(2.7) Headache 14 (2.5) 26 (2.2) Musculoskeletal stiffness 14 (2.5) 23 (2.0) Migraine 12 (2.2) 34 (2.9) Facial paresis 7 (1.3) 15 (1.3) Facial spasm 7 (1.3) 11 (0.9) Myalgia 7 (1.3) 11 (0.9) Pruritus 7 (1.3) 7 (0.6) Musculoskeletal pain 6 (1.1) 10 (0.9) 42 7
8 RESULTS: MATHARU, ET AL RESULTS: MATHARU, ET AL AUTHOR S CONCLUSIONS CRITICAL APPRAISAL Adverse event findings are similar to the PREEMPT studies Adverse event incidence rate decreased with each treatment session Data adds onto support the favorable safety profile of botox for CM prophylaxis Strengths Observe and assess patient with CM in a real world setting Large observational study in multiple settings Weaknesses Observational study: increases generalizability METHODS: CADY, ET AL (2011) A MULTI-CENTER DOUBLE-BLIND PILOT COMPARISON OF ONABOTULINUMTOXINA AND TOPIRAMATE FOR THE PROPHYLACTIC TREATMENT OF CHRONIC MIGRAINE. CADY, ET AL (2011) 3-center double-blind randomized pilot study Objectives: Direct comparison of topiramate vs onabotulinumtoxina Group 1 : Topiramate plus placebo injections Group 2: onabotulinumtoxina injections plus placebo tablets Daily headache diaries over 4 week baseline period and 12- week active study period
9 METHODS: CADY, ET AL (2011) METHODS: CADY, ET AL (2011) Inclusion 18 to 65 years of age Subjects met criteria for CM as defined by Second Edition of the International Classification for Headache Disorders Exclusion Female subjects who were pregnant, breast feeding, or planning to become pregnant Individuals with headache disorders other than CM Subjects who had previously used botulinum toxin of any type or topiramate regardless of indication Primary endpoint Physician global assessment: treatment responder rate Indicated improvement in both groups over 12 weeks Secondary endpoint Measured at weeks 4 and 12 Headache days per month, migraine days, headache free-days, days on acute medication, and severity of headache episodes RESULTS: CADY, ET AL (2011) RESULTS: CADY, ET AL (2011) Week 4 Group I Topiramate Group 2 Botox P-value N=27 N= No change 4 (14.8) 9 (32.1) Slight improvement 8 (29.6) 10 (35.7) Moderate improvement Marked improvement 9 (33.3) 3 (10.7) 3 (11.1) 4 (14.3) Week 12 Group 1 Topiramate Group 2 Botox P-value N=24 N= No change 5 (20.8) 3 (12.5) Slight improvement 1 (4.2) 5 (20.8) Moderate improvement Marked improvement 6 (25.0) 4 (16.7) 10 (41.7) 10 (41.7) RESULTS: CADY, ET AL (2011) AUTHOR S CONCLUSIONS Group 1 Topiramate Headache free days/month Group 2 Group 1 Botox Topiramate Week 4 Week 12 Group 2 Botox 53 Topiramate and botox demonstrated efficacy in treating subjects with CM Improvements were noted from both medications Support the use of botox for patients with frequent migraine 54 9
10 CRITICAL APPRAISAL LITERATURE SUMMARY Strengths Aurora, et al Matharu, et al Cady, et al Completed a direct comparison between the two drugs Subjects were asked to pursue an open label Weaknesses Small sample size Questionable blinding due to injection reactions Lack a placebo arm Botox vs. placebo Safety and efficacy Reduced frequency of HA and migraine days with botox Botox observational Safety and tolerability Safety outcomes similar to PREEMPT trials Botox vs. topiramate Efficacy Similar efficacy outcomes RW PATIENT CASE Assessment: Controlled on current regimen Topiramate 100mg daily Sumatriptan 25mg twice daily PRN Is this patient a candidate for Botox? No: Patient is currently controlled on current regimen CONCLUSION Diagnosis and treatment for chronic migraines is complex and clinicians should be mindful refractory to treatment is common. Botox has proven safety outcomes and efficacy outcomes in primary endpoints for reducing migraine days Guidelines of the American Academy of Neurology and NICE both recommend botox in CM Would recommend for patients that have failed two previous agents level A and/or B medications FUTURE TOPICS FOR RESEARCH ACKNOWLEDGEMENTS Studies comparing botox vs other approved agents on the market Studies to determine if botox reaches a plateau effect Studies in patients with comorbidities in addition to CM Studies to determine impact of botox on indirect and direct costs of CM compared to the agents well studied in CM Review committee Jason Jokerst, Pharm.D., BCPS April Hinds, PharmD, BCACP Evaluator Lucas Hill, PharmD, BCPS
11 BOTOX: TO INJECT OR NOT INJECT? IN CHRONIC MIGRAINES JENNIFER SHIN, PHARMD PGY2 AMBULATORY CARE PHARMACY RESIDENT COMMUNITYCARE HEALTH CENTERS PHARMACOTHERAPY ROUNDS OCTOBER 20,
I have no financial relationships to disclose. I will not discuss investigational use of medication in my presentation.
 I have no financial relationships to disclose. I will not discuss investigational use of medication in my presentation. In 1962, Bille published landmark epidemiologic survey of headache among 9,000 school
I have no financial relationships to disclose. I will not discuss investigational use of medication in my presentation. In 1962, Bille published landmark epidemiologic survey of headache among 9,000 school
Update on Diagnosis and Management of Migraines
 Update on Diagnosis and Management of Migraines Joel J. Heidelbaugh, MD, FAAFP, FACG Clinical Professor Departments of Family Medicine and Urology University of Michigan Learning Objectives To distinguish
Update on Diagnosis and Management of Migraines Joel J. Heidelbaugh, MD, FAAFP, FACG Clinical Professor Departments of Family Medicine and Urology University of Michigan Learning Objectives To distinguish
Prevention and Treatment of Migraines CAITLIN BARNES, PHARM.D. CANDIDATE AMBULATORY CARE JOE CAMMILLERI, PHARM.D. NATOHYA MALLORY, PHARM.D.
 Prevention and Treatment of Migraines CAITLIN BARNES, PHARM.D. CANDIDATE AMBULATORY CARE JOE CAMMILLERI, PHARM.D. NATOHYA MALLORY, PHARM.D. Objectives Present patient case Review epidemiology/pathophysiology
Prevention and Treatment of Migraines CAITLIN BARNES, PHARM.D. CANDIDATE AMBULATORY CARE JOE CAMMILLERI, PHARM.D. NATOHYA MALLORY, PHARM.D. Objectives Present patient case Review epidemiology/pathophysiology
MEASURE #3: PREVENTIVE MIGRAINE MEDICATION PRESCRIBED Headache
 MEASURE #3: PREVENTIVE MIGRAINE MEDICATION PRESCRIBED Headache Measure Description Percentage of patients age 18 years old and older diagnosed with migraine headache whose migraine frequency is 4 migraine
MEASURE #3: PREVENTIVE MIGRAINE MEDICATION PRESCRIBED Headache Measure Description Percentage of patients age 18 years old and older diagnosed with migraine headache whose migraine frequency is 4 migraine
What is the Effectiveness of OnabotulinumtoxinA (Botox ) in Reducing the Number of Chronic Migraines (CM) in Patients Years Old?
 Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 2013 What is the Effectiveness of OnabotulinumtoxinA
Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 2013 What is the Effectiveness of OnabotulinumtoxinA
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 CGRP Page 1 of 8 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: CGRP (calcitonin gene-related peptide) Prime Therapeutics will review Prior Authorization requests
CGRP Page 1 of 8 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: CGRP (calcitonin gene-related peptide) Prime Therapeutics will review Prior Authorization requests
Headache Master School Japan-Osaka 2016 (HMSJ-Osaka2016) October 23, II. Management of Refractory Headaches
 Headache Master School Japan-Osaka 2016 (HMSJ-Osaka2016) October 23, 2016 II. Management of Refractory Headaches Case presentation 1: A case of intractable daily-persistent headache Keio University School
Headache Master School Japan-Osaka 2016 (HMSJ-Osaka2016) October 23, 2016 II. Management of Refractory Headaches Case presentation 1: A case of intractable daily-persistent headache Keio University School
This policy addresses only the type B formulation rimabotulinumtoxinb marketed as Myobloc.
 RimabotulinumtoxinB NDC CODE(S) 10454-0710-XX Myobloc 2500 UNIT/0.5ML SOLN (SOLSTICE NEUROSCIENCES) 10454-0711-XX Myobloc 5000 UNIT/ML SOLN (SOLSTICE NEUROSCIENCES) 10454-0712-XX Myobloc 10000 UNIT/2ML
RimabotulinumtoxinB NDC CODE(S) 10454-0710-XX Myobloc 2500 UNIT/0.5ML SOLN (SOLSTICE NEUROSCIENCES) 10454-0711-XX Myobloc 5000 UNIT/ML SOLN (SOLSTICE NEUROSCIENCES) 10454-0712-XX Myobloc 10000 UNIT/2ML
How do we treat migraine? New SIGN Guidelines
 How do we treat migraine? New SIGN Guidelines Managing your migraine Migraine Trust, Edinburgh 2018 Callum Duncan Consultant Neurologist Aberdeen Royal Infirmary Chair SIGN Guideline 155 Premonitory Mood
How do we treat migraine? New SIGN Guidelines Managing your migraine Migraine Trust, Edinburgh 2018 Callum Duncan Consultant Neurologist Aberdeen Royal Infirmary Chair SIGN Guideline 155 Premonitory Mood
David W. Dodick M.D. Professor Director of Headache Medicine Department of Neurology Mayo Clinic Phoenix Arizona USA
 Headache Masters School 2013 in Asia Sunday March 24, 2013 Procedural Medicine Workshop Onabotulinumtoxin A: Evidence, Injection Technique, and Mechanism of Action David W. Dodick M.D. Professor Director
Headache Masters School 2013 in Asia Sunday March 24, 2013 Procedural Medicine Workshop Onabotulinumtoxin A: Evidence, Injection Technique, and Mechanism of Action David W. Dodick M.D. Professor Director
Chronic Migraine in Primary Care. December 11 th, 2017 Werner J. Becker University of Calgary
 Chronic Migraine in Primary Care December 11 th, 2017 Werner J. Becker University of Calgary Disclosures Faculty: Werner J. Becker Relationships with commercial interests: Grants/Research Support: Clinical
Chronic Migraine in Primary Care December 11 th, 2017 Werner J. Becker University of Calgary Disclosures Faculty: Werner J. Becker Relationships with commercial interests: Grants/Research Support: Clinical
Dubai Standards of Care (Migraine)
 Dubai Standards of Care 2018 (Migraine) Preface Migraine is one of the most common problem dealt with in daily practice. In Dubai, the management of migraine is done through various different strategies.
Dubai Standards of Care 2018 (Migraine) Preface Migraine is one of the most common problem dealt with in daily practice. In Dubai, the management of migraine is done through various different strategies.
Get ahead of the ACHE: Monoclonal Antibodies in Migraine Prevention
 Get ahead of the ACHE: Monoclonal Antibodies in Migraine Prevention Amanda Janisch, PharmD PGY2 Ambulatory Care Pharmacy Resident MCHS SWMN, Mankato, MN 2018 MFMER slide-1 Disclosures No financial interest
Get ahead of the ACHE: Monoclonal Antibodies in Migraine Prevention Amanda Janisch, PharmD PGY2 Ambulatory Care Pharmacy Resident MCHS SWMN, Mankato, MN 2018 MFMER slide-1 Disclosures No financial interest
Jessica Ailani MD FAHS Director, Georgetown Headache Center Associate Professor Neurology Medstar Georgetown University Hospital
 Jessica Ailani MD FAHS Director, Georgetown Headache Center Associate Professor Neurology Medstar Georgetown University Hospital Honorarium from Current Pain and Headache Reports; Section Editor Unusual
Jessica Ailani MD FAHS Director, Georgetown Headache Center Associate Professor Neurology Medstar Georgetown University Hospital Honorarium from Current Pain and Headache Reports; Section Editor Unusual
A case of a patient with chronic headache. Focus on Migraine. None related to the presentation Grants to conduct clinical trials from: Speaker bureau:
 Chronic Daily Headache Bassel F. Shneker, MD, MBA Associate Professor Vice Chair, OSU Neurology The Ohio State University Wexner Medical Center Financial Disclosures None related to the presentation Grants
Chronic Daily Headache Bassel F. Shneker, MD, MBA Associate Professor Vice Chair, OSU Neurology The Ohio State University Wexner Medical Center Financial Disclosures None related to the presentation Grants
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 CGRP Page 1 of 13 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: CGRP (calcitonin gene-related peptide) Prime Therapeutics will review Prior Authorization requests
CGRP Page 1 of 13 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: CGRP (calcitonin gene-related peptide) Prime Therapeutics will review Prior Authorization requests
Preventive treatment of migraine. Rebecca Burch, MD Brigham and Women s Faulkner Hospital Harvard Medical School Boston, MA
 Preventive treatment of migraine Rebecca Burch, MD Brigham and Women s Faulkner Hospital Harvard Medical School Boston, MA No disclosures Disclosures Many preventive treatments for migraine are not FDA-approved
Preventive treatment of migraine Rebecca Burch, MD Brigham and Women s Faulkner Hospital Harvard Medical School Boston, MA No disclosures Disclosures Many preventive treatments for migraine are not FDA-approved
Migraine: Past, Present and Future Edward O Sullivan September 12 th 2015 Dublin 12/09/2015
 1 Migraine: Past, Present and Future Edward O Sullivan September 12 th 2015 Dublin Tour de France 2015: Mark Renshaw Yesterday at the end of the stage 17 I came down with a migraine before the final climb
1 Migraine: Past, Present and Future Edward O Sullivan September 12 th 2015 Dublin Tour de France 2015: Mark Renshaw Yesterday at the end of the stage 17 I came down with a migraine before the final climb
Emerging Challenges in Primary Care: Brainstorm: A Symposium on Migraine Treatment and Management
 Emerging Challenges in Primary Care: 2017 Brainstorm: A Symposium on Migraine Treatment and Management 1 Faculty Jeff Unger, MD, ABFM, FACE Director, Unger Primary Care Medical Group Rancho Cucamonga,
Emerging Challenges in Primary Care: 2017 Brainstorm: A Symposium on Migraine Treatment and Management 1 Faculty Jeff Unger, MD, ABFM, FACE Director, Unger Primary Care Medical Group Rancho Cucamonga,
Headache. Section 1. Migraine headache. Clinical presentation
 Section 1 Headache Migraine headache 1 Clinical presentation It is important to recognize just how significant a problem migraine headache is. It has been estimated that migraine affects 11% of the United
Section 1 Headache Migraine headache 1 Clinical presentation It is important to recognize just how significant a problem migraine headache is. It has been estimated that migraine affects 11% of the United
Headache Mary D. Hughes, MD Neuroscience Associates
 Headache Mary D. Hughes, MD Neuroscience Associates Case 1 22 year old female presents with recurrent headaches. She has had headaches for the past 3 years. They start on the right side of her head and
Headache Mary D. Hughes, MD Neuroscience Associates Case 1 22 year old female presents with recurrent headaches. She has had headaches for the past 3 years. They start on the right side of her head and
Strategies in Migraine Care
 Strategies in Migraine Care Julie L. Roth, MD Rhode Island Hospital Assistant Professor, Neurology The Warren Alpert Medical School of Brown University March 28, 2015 Financial Disclosures None. Objectives
Strategies in Migraine Care Julie L. Roth, MD Rhode Island Hospital Assistant Professor, Neurology The Warren Alpert Medical School of Brown University March 28, 2015 Financial Disclosures None. Objectives
Subject: CGRP Inhibitors
 ARCHIVED (NOT ACTIVE RETIRED) Archived: 08/01/18 09-J2000-98 Original Effective Date: 06/15/18 Reviewed: 05/09/18 Revised: 08/01/18 Next Review: ARCHIVED (NOT ACTIVE RETIRED) Subject: CGRP Inhibitors THIS
ARCHIVED (NOT ACTIVE RETIRED) Archived: 08/01/18 09-J2000-98 Original Effective Date: 06/15/18 Reviewed: 05/09/18 Revised: 08/01/18 Next Review: ARCHIVED (NOT ACTIVE RETIRED) Subject: CGRP Inhibitors THIS
Clinical case. Clinical case 3/15/2018 OVERVIEW. Refractory headaches and update on novel treatment. Refractory headache.
 OVERVIEW Refractory headaches and update on novel treatment Definition of refractory headache Treatment approach Medications Neuromodulation In the pipeline Juliette Preston, MD OHSU Headache Center Refractory
OVERVIEW Refractory headaches and update on novel treatment Definition of refractory headache Treatment approach Medications Neuromodulation In the pipeline Juliette Preston, MD OHSU Headache Center Refractory
Current Migraine Treatment Therapy. Daniel Kassicieh, DO, FAAN
 Current Migraine Treatment Therapy Daniel Kassicieh, DO, FAAN Migraine a Disease Process Migraines are a chronic disease process similar to many other chronic medical conditions Migraine has a low mortality
Current Migraine Treatment Therapy Daniel Kassicieh, DO, FAAN Migraine a Disease Process Migraines are a chronic disease process similar to many other chronic medical conditions Migraine has a low mortality
Mark W. Green, MD, FAAN
 Mark W. Green, MD, FAAN Professor of Neurology, Anesthesiology, and Rehabilitation Medicine Director of Headache and Pain Medicine Icahn School of Medicine at Mt Sinai New York Pain-sensitive structures
Mark W. Green, MD, FAAN Professor of Neurology, Anesthesiology, and Rehabilitation Medicine Director of Headache and Pain Medicine Icahn School of Medicine at Mt Sinai New York Pain-sensitive structures
OH, MY ACHING HEAD! I HAVE NO DISCLOSURES OR CONFLICTS OF INTERESTS TO DECLARE MANAGING HEADACHE IN THE OUTPATIENT SETTING SECONDARY HEADACHES
 1 JUSTIN A. OSSMAN, MD CHATTANOOGA FAMILY MEDICINE UPDATE OH, MY ACHING HEAD! MANAGING HEADACHE IN THE OUTPATIENT SETTING 2 I HAVE NO DISCLOSURES OR CONFLICTS OF INTERESTS TO DECLARE OBJECTIVES International
1 JUSTIN A. OSSMAN, MD CHATTANOOGA FAMILY MEDICINE UPDATE OH, MY ACHING HEAD! MANAGING HEADACHE IN THE OUTPATIENT SETTING 2 I HAVE NO DISCLOSURES OR CONFLICTS OF INTERESTS TO DECLARE OBJECTIVES International
ARxCH. Annual Review of Changes in Healthcare. Calcitonin Gene-Related Peptide Receptors and the Prevention of Migraines. Abstract
 Calcitonin Gene-Related Peptide Receptors and the Prevention of Migraines Brian Schuler, PharmD Candidate 2018 1 1 University of Findlay College of Pharmacy Abstract Migraines are the third most prevalent
Calcitonin Gene-Related Peptide Receptors and the Prevention of Migraines Brian Schuler, PharmD Candidate 2018 1 1 University of Findlay College of Pharmacy Abstract Migraines are the third most prevalent
Case Presentation. Case Presentation. Case Presentation. Truths about Headaches (2017) Most headaches were muscle-tension headaches
 Agenda Case presentation Migraine Morphology Primary and Premonitory Phase Secondary Headache Aura Headache Primer on Pain Medication Overuse Headache Case Presentation RT is a 25 year old woman with daily
Agenda Case presentation Migraine Morphology Primary and Premonitory Phase Secondary Headache Aura Headache Primer on Pain Medication Overuse Headache Case Presentation RT is a 25 year old woman with daily
Botulinum toxin type A for the prevention of headaches in adults with chronic migraine
 Botulinum toxin type A for the prevention of headaches in adults with chronic migraine Issued: June 2012 guidance.nice.org.uk/ta260 NICE has accredited the process used by the Centre for Health Technology
Botulinum toxin type A for the prevention of headaches in adults with chronic migraine Issued: June 2012 guidance.nice.org.uk/ta260 NICE has accredited the process used by the Centre for Health Technology
10/19/2018. Disclosures MIGRAINE PROPHYLAXIS. Objectives. Definitions Slide. What do you think the aooe stands for at the end of erenumab-aooe?
 Disclosures MIGRAINE PROPHYLAXIS Erenumab-aooe (AIMOVIG TM ) Calcitonin Gene Related Peptide Receptor Antagonist No conflicts of interest to disclose Chelsey Roscoe, PharmD PGY1 Resident - CTVHCS 2 3 Definitions
Disclosures MIGRAINE PROPHYLAXIS Erenumab-aooe (AIMOVIG TM ) Calcitonin Gene Related Peptide Receptor Antagonist No conflicts of interest to disclose Chelsey Roscoe, PharmD PGY1 Resident - CTVHCS 2 3 Definitions
Clinical Trials. Hans-Christoph Diener Senior Professor of Clinical Neuroscienes Medical Faculty University Duisburg-Essen Germany
 Clinical Trials Hans-Christoph Diener Senior Professor of Clinical Neuroscienes Medical Faculty University Duisburg-Essen Germany Conflict of Interest Statement German Research Council German Ministry
Clinical Trials Hans-Christoph Diener Senior Professor of Clinical Neuroscienes Medical Faculty University Duisburg-Essen Germany Conflict of Interest Statement German Research Council German Ministry
Proposed Project Scope. OPTIMAL USE OnabotulinumtoxinA for the Prevention of Chronic Migraine Clinical Evidence, Policies and Practice
 Proposed Project Scope OPTIMAL USE OnabotulinumtoxinA for the Prevention of Chronic Migraine Clinical Evidence, Policies and Practice May 2018 1. BACKGROUND AND RATIONALE Migraine is a common, debilitating
Proposed Project Scope OPTIMAL USE OnabotulinumtoxinA for the Prevention of Chronic Migraine Clinical Evidence, Policies and Practice May 2018 1. BACKGROUND AND RATIONALE Migraine is a common, debilitating
Migraine Management. Roger Cady, MD Headache Care Center Springfield, MO
 Migraine Management Roger Cady, MD Headache Care Center Springfield, MO Disclosures Objectives The evolution of migraine From benign episodic (benign) headache to potentially a devastating chronic disease
Migraine Management Roger Cady, MD Headache Care Center Springfield, MO Disclosures Objectives The evolution of migraine From benign episodic (benign) headache to potentially a devastating chronic disease
MIGRAINE UPDATE. Objectives & Disclosures. Learn techniques used to diagnose headaches. Become familiar with medications used for headache treatment.
 MIGRAINE UPDATE Karen L. Bremer, MD November 16, 2018 Objectives & Disclosures Learn techniques used to diagnose headaches. Become familiar with medications used for headache treatment. Disclosure: I am
MIGRAINE UPDATE Karen L. Bremer, MD November 16, 2018 Objectives & Disclosures Learn techniques used to diagnose headaches. Become familiar with medications used for headache treatment. Disclosure: I am
HMFP Comprehensive Headache Center Department of Anesthesia, Critical Care and Pain Medicine Beth Israel Deaconess Medical Center Instructor in
 HMFP Comprehensive Headache Center Department of Anesthesia, Critical Care and Pain Medicine Beth Israel Deaconess Medical Center Instructor in Anesthesia and Neurology Harvard Medical School Limited time
HMFP Comprehensive Headache Center Department of Anesthesia, Critical Care and Pain Medicine Beth Israel Deaconess Medical Center Instructor in Anesthesia and Neurology Harvard Medical School Limited time
ปวดศ รษะมา 5 ป ก นยาแก ปวดก ย งไม ข น นพ.พาว ฒ เมฆว ช ย โรงพยาบาลนครราชส มา
 ปวดศ รษะมา 5 ป ก นยาแก ปวดก ย งไม ข น นพ.พาว ฒ เมฆว ช ย โรงพยาบาลนครราชส มา 1 CONTENT 1 2 3 Chronic Daily Headache Medical Overused Headache Management Headaches are one of the most common symptoms List
ปวดศ รษะมา 5 ป ก นยาแก ปวดก ย งไม ข น นพ.พาว ฒ เมฆว ช ย โรงพยาบาลนครราชส มา 1 CONTENT 1 2 3 Chronic Daily Headache Medical Overused Headache Management Headaches are one of the most common symptoms List
Outpatient Headache Care Guideline
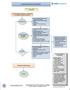 1 Outpatient Care Guideline Inclusion criteria: children > 3 yrs with headaches Is urgent emergency department, neuroimaging, or Neurology consultation indicated? Referral to ED if: New severe headache
1 Outpatient Care Guideline Inclusion criteria: children > 3 yrs with headaches Is urgent emergency department, neuroimaging, or Neurology consultation indicated? Referral to ED if: New severe headache
ABORTIVE AGENTS. Average cost per 30 days. Form Limits SEROTONIN AGONISTS $ $ Reserved for treatment failure to either Sumatriptan PA; QL
 MEDICATION COVERAGE POLICY PHARMACY AND THERAPEUTICS ADVISORY COMMITTEE POLICY: Migraine Therapy P&T DATE: 12/11/2018 CLASS: Neurological Disorders REVIEW HISTORY 9/17, 12/16, 9/15, 2/15, 2/10, LOB: MCL
MEDICATION COVERAGE POLICY PHARMACY AND THERAPEUTICS ADVISORY COMMITTEE POLICY: Migraine Therapy P&T DATE: 12/11/2018 CLASS: Neurological Disorders REVIEW HISTORY 9/17, 12/16, 9/15, 2/15, 2/10, LOB: MCL
Anti-Migraine Agents
 DRUG POLICY BENEFIT APPLICATION Anti-Migraine Agents Benefit determinations are based on the applicable contract language in effect at the time the services were rendered. Exclusions, limitations or exceptions
DRUG POLICY BENEFIT APPLICATION Anti-Migraine Agents Benefit determinations are based on the applicable contract language in effect at the time the services were rendered. Exclusions, limitations or exceptions
Page: 1 of 6. Aimovig (erenumab-aooe) injection, Ajovy (fremanezumab-vfrm) injection, Emgality (galcanezumab-gnim)
 Page: 1 of 6 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Last Review Date: November 30, 2018 Description Aimovig (erenumab-aooe) injection, Ajovy (fremanezumab-vfrm)
Page: 1 of 6 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Last Review Date: November 30, 2018 Description Aimovig (erenumab-aooe) injection, Ajovy (fremanezumab-vfrm)
6/20/2018 PROCEDURAL TREATMENTS FOR HEADACHE. Case study: Emily. Diary
 Headache severity (Scale 0 10) Topiramate started on July 20, D/C July 29 Amitriptyline initiated, D/C on Sep 23 PROCEDURAL TREATMENTS FOR HEADACHE Andrew M. Blumenfeld Director of the Headache Center
Headache severity (Scale 0 10) Topiramate started on July 20, D/C July 29 Amitriptyline initiated, D/C on Sep 23 PROCEDURAL TREATMENTS FOR HEADACHE Andrew M. Blumenfeld Director of the Headache Center
National Institute for Health and Clinical Excellence. Single Technology Appraisal (STA)
 National Institute for Health and Clinical Excellence Comment 1: the draft remit Single Technology Appraisal (STA) Botulinum toxin type A for the prophylaxis of headaches in adults with chronic migraine
National Institute for Health and Clinical Excellence Comment 1: the draft remit Single Technology Appraisal (STA) Botulinum toxin type A for the prophylaxis of headaches in adults with chronic migraine
Lost in Translation: Making Sense of Clinical Treatment Guidelines
 Lost in Translation: Making Sense of Clinical Treatment Guidelines Charles E. Argoff, MD, CPE Disclosures: Charles Argoff Financial Disclosure: Consultant: Teva, Daiichi Sakyo, Pfizer, Nektar, Purdue,
Lost in Translation: Making Sense of Clinical Treatment Guidelines Charles E. Argoff, MD, CPE Disclosures: Charles Argoff Financial Disclosure: Consultant: Teva, Daiichi Sakyo, Pfizer, Nektar, Purdue,
Is Topiramate Effective in Preventing Pediatric Migraines?
 Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 2013 Is Topiramate Effective in Preventing
Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 2013 Is Topiramate Effective in Preventing
Management of headache
 Management of headache TJ Steiner Imperial College London Based on European principles of management of common headache disorders TJ Steiner, K Paemeleire, R Jensen, D Valade, L Savi, MJA Lainez, H-C Diener,
Management of headache TJ Steiner Imperial College London Based on European principles of management of common headache disorders TJ Steiner, K Paemeleire, R Jensen, D Valade, L Savi, MJA Lainez, H-C Diener,
Subject: Aimovig (erenumab) Original Effective Date: 7/10/2018. Policy Number: MCP-320. Revision Date(s):
 Subject: Aimovig (erenumab) Original Effective Date: 7/10/2018 Policy Number: MCP-320 Revision Date(s): Review Date(s): MCPC Approval Date: 7/10/2018 DISCLAIMER This Molina Clinical Policy (MCP) is intended
Subject: Aimovig (erenumab) Original Effective Date: 7/10/2018 Policy Number: MCP-320 Revision Date(s): Review Date(s): MCPC Approval Date: 7/10/2018 DISCLAIMER This Molina Clinical Policy (MCP) is intended
Goals. Primary Headache Syndromes. One-Year Prevalence of Common Headache Disorders
 Goals One-Year Prevalence of Common Headache Disorders Impact of primary headache syndromes Non pharmacologic Rx of migraine individualized to patient triggers Complementary and alternative Rx of migraine
Goals One-Year Prevalence of Common Headache Disorders Impact of primary headache syndromes Non pharmacologic Rx of migraine individualized to patient triggers Complementary and alternative Rx of migraine
COMBINATION THERAPIES PREVENTATIVE THERAPIES BETA BLOCKERS
 ACUTE THEAPIES TIPTANS TICYCLIC ANTIDEPESSANTS When starting acute treatment, healthcare professionals should warn patients about the risk of developing medication-overuse headache. ASPIIN Aspirin (900
ACUTE THEAPIES TIPTANS TICYCLIC ANTIDEPESSANTS When starting acute treatment, healthcare professionals should warn patients about the risk of developing medication-overuse headache. ASPIIN Aspirin (900
10/31/2017 PRIMARY CARE AND HEADACHE DISCLOSURES WHERE DO THOSE WITH HEADACHE SEEK MEDICAL CARE? Primary Care 67%
 PRIMARY CARE AND HEADACHE Sonja Potrebic MD PhD Regional Headache Specialist Kaiser LAMC 1 WHERE DO THOSE WITH HEADACHE SEEK MEDICAL CARE? Column1 Primary Care 67% Primary Care Headache Specialty Other
PRIMARY CARE AND HEADACHE Sonja Potrebic MD PhD Regional Headache Specialist Kaiser LAMC 1 WHERE DO THOSE WITH HEADACHE SEEK MEDICAL CARE? Column1 Primary Care 67% Primary Care Headache Specialty Other
Migraine Management. Jane Melling Headache nurse Mater Misericordiae Hospital
 Migraine Management Jane Melling Headache nurse Mater Misericordiae Hospital Migraine facts Among the most common disorders of the nervous system 3 rd most prevalent medical disorder on the planet (lancet
Migraine Management Jane Melling Headache nurse Mater Misericordiae Hospital Migraine facts Among the most common disorders of the nervous system 3 rd most prevalent medical disorder on the planet (lancet
Neuromuscular Blocking Agents
 Neuromuscular Blocking Agents DRUG POLICY This Prior Authorization request will be reviewed for medical necessity only. Benefits are subject to the terms and conditions of the patient s contract. Please
Neuromuscular Blocking Agents DRUG POLICY This Prior Authorization request will be reviewed for medical necessity only. Benefits are subject to the terms and conditions of the patient s contract. Please
Disclosures. Triptans for Kids 5/16/13
 5/16/13 Disclosures Triptans for Kids Amy A. Gelfand, MD GelfandA@neuropeds.ucsf.edu Departments of Neurology and Pediatrics UCSF Child Neurology and Headache Center I receive grant funding from: NIH/NINDS
5/16/13 Disclosures Triptans for Kids Amy A. Gelfand, MD GelfandA@neuropeds.ucsf.edu Departments of Neurology and Pediatrics UCSF Child Neurology and Headache Center I receive grant funding from: NIH/NINDS
ASPS Insurance Coverage Criteria for Third-Party Payers Nerve Decompression for Migraine Surgery
 ASPS Insurance Coverage Criteria for Third-Party Payers Nerve Decompression for Migraine Surgery BACKGROUND Migraine headache is a debilitating neurovascular disorder that affects 35 million Americans
ASPS Insurance Coverage Criteria for Third-Party Payers Nerve Decompression for Migraine Surgery BACKGROUND Migraine headache is a debilitating neurovascular disorder that affects 35 million Americans
ADVANCES IN MIGRAINE MANAGEMENT
 ADVANCES IN MIGRAINE MANAGEMENT Joanna Girard Katzman, M.D.MSPH Assistant Professor, Dept. of Neurology Project ECHO, Chronic Pain Program University of New Mexico Outline Migraine throughout the decades
ADVANCES IN MIGRAINE MANAGEMENT Joanna Girard Katzman, M.D.MSPH Assistant Professor, Dept. of Neurology Project ECHO, Chronic Pain Program University of New Mexico Outline Migraine throughout the decades
2 nd Resubmission. 13 January 2017
 2 nd Resubmission botulinum toxin A, 50 Allergan units, 100 Allergan units, 200 Allergan units, powder for solution for injection (Botox ) SMC No. (692/11) Allergan Limited 13 January 2017 The Scottish
2 nd Resubmission botulinum toxin A, 50 Allergan units, 100 Allergan units, 200 Allergan units, powder for solution for injection (Botox ) SMC No. (692/11) Allergan Limited 13 January 2017 The Scottish
Technology appraisal guidance Published: 27 June 2012 nice.org.uk/guidance/ta260
 Botulinum toxin type A for the prevention ention of headaches in adults with chronic migraine Technology appraisal guidance Published: 27 June 2012 nice.org.uk/guidance/ta260 NICE 2018. All rights reserved.
Botulinum toxin type A for the prevention ention of headaches in adults with chronic migraine Technology appraisal guidance Published: 27 June 2012 nice.org.uk/guidance/ta260 NICE 2018. All rights reserved.
Description. Section: Surgery Effective Date: January 15, 2016 Subsection: Surgery Original Policy Date: December 6, 2012 Subject:
 Last Review Status/Date: December 2015 Description Page: 1 of 11 Surgical deactivation of trigger sites is a proposed treatment of migraine headache. The procedure involves identifying a patient s predominant
Last Review Status/Date: December 2015 Description Page: 1 of 11 Surgical deactivation of trigger sites is a proposed treatment of migraine headache. The procedure involves identifying a patient s predominant
Migraine: Developing Drugs for Acute Treatment Guidance for Industry
 Migraine: Developing Drugs for Acute Treatment Guidance for Industry U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER) February 2018
Migraine: Developing Drugs for Acute Treatment Guidance for Industry U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER) February 2018
Study No.: Title: Rationale: Phase: Study Period: Study Design: Centres: Indication: Treatment: Objectives: Primary Outcome/Efficacy Variable:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
1/25/2018 ARE CGRP ANTAGONISTS ANY BETTER THAN CURRENT EVIDENCE BASED TREATMENTS? Disclosures: Objectives: Headache Division
 ARE CGRP ANTAGONISTS ANY BETTER THAN CURRENT EVIDENCE BASED TREATMENTS? Lawrence C Newman, MD, FAHS, FAAN Clinical Professor of Neurology Disclosures: Advisory Board: Alder, Allergan, Amgen, Lilly, Supernus,
ARE CGRP ANTAGONISTS ANY BETTER THAN CURRENT EVIDENCE BASED TREATMENTS? Lawrence C Newman, MD, FAHS, FAAN Clinical Professor of Neurology Disclosures: Advisory Board: Alder, Allergan, Amgen, Lilly, Supernus,
Abortive Agents. Available Strengths. Formulary Limits. Tablet: 5mg, 10mg ODT: 5mg, 10 mg 25mg, 50mg, 100mg. 5mg/act, 20mg/act
 MEDICATION COVERAGE POLICY PHARMACY AND THERAPEUTICS ADVISORY COMMITTEE POLICY: Migraine Therapy P&T DATE: 9/12/2017 CLASS: Neurological Disorders REVIEW HISTORY 12/16, 9/15, 2/15, 2/10, 5/07 LOB: MCL
MEDICATION COVERAGE POLICY PHARMACY AND THERAPEUTICS ADVISORY COMMITTEE POLICY: Migraine Therapy P&T DATE: 9/12/2017 CLASS: Neurological Disorders REVIEW HISTORY 12/16, 9/15, 2/15, 2/10, 5/07 LOB: MCL
They are updated regularly as new NICE guidance is published. To view the latest version of this NICE Pathway see:
 bring together everything NICE says on a topic in an interactive flowchart. are interactive and designed to be used online. They are updated regularly as new NICE guidance is published. To view the latest
bring together everything NICE says on a topic in an interactive flowchart. are interactive and designed to be used online. They are updated regularly as new NICE guidance is published. To view the latest
Long-Term Care Updates
 Long-Term Care Updates September 2017 By Lindsay Slowiczek, PharmD Migraines are often considered to be a condition affecting younger or middle-aged patients, during which patients experience episodic,
Long-Term Care Updates September 2017 By Lindsay Slowiczek, PharmD Migraines are often considered to be a condition affecting younger or middle-aged patients, during which patients experience episodic,
Policy. Medical Policy Manual Approved Revised: Do Not Implement Until 4/2/19. IncobotulinumtoxinA
 IncobotulinumtoxinA NDC CODE(S) 00259-1605-XX Xeomin 50 UNIT SOLR (MERZ PHARMACEUTICAL) 00259-1610-XX Xeomin 100 UNIT SOLR (MERZ PHARMACEUTICAL) 00259-1620-XX Xeomin 200 UNIT SOLR (MERZ PHARMACEUTICAL)
IncobotulinumtoxinA NDC CODE(S) 00259-1605-XX Xeomin 50 UNIT SOLR (MERZ PHARMACEUTICAL) 00259-1610-XX Xeomin 100 UNIT SOLR (MERZ PHARMACEUTICAL) 00259-1620-XX Xeomin 200 UNIT SOLR (MERZ PHARMACEUTICAL)
Management options for Migraine. Prof. Dr. Khwaja Nazimuddin Head Dept. of Internal Medicine BIRDEM
 Management options for Migraine Prof. Dr. Khwaja Nazimuddin Head Dept. of Internal Medicine BIRDEM Assessment The Migraine Disability Assessment Score MIDAS Complete loss of work Partial loss of work Off
Management options for Migraine Prof. Dr. Khwaja Nazimuddin Head Dept. of Internal Medicine BIRDEM Assessment The Migraine Disability Assessment Score MIDAS Complete loss of work Partial loss of work Off
Headache evaluation and management after concussion. Assistant Professor
 Headache evaluation and management after concussion Juliette Preston, M.D. Assistant Professor OHSU Headache Center OVERVIEW Introduction Definition Acute post-traumatic headache Red flags Persistent post-traumatic
Headache evaluation and management after concussion Juliette Preston, M.D. Assistant Professor OHSU Headache Center OVERVIEW Introduction Definition Acute post-traumatic headache Red flags Persistent post-traumatic
BOTULINUM TOXIN POLICY TO INCLUDE:
 BOTULINUM TOXIN POLICY TO INCLUDE: Blepharospasm in adults, Hemi facial spasm in adults, spasmodic torticollis (cervical dystonia), focal spasticity treatment of dynamic equinus foot deformity, focal spasticity
BOTULINUM TOXIN POLICY TO INCLUDE: Blepharospasm in adults, Hemi facial spasm in adults, spasmodic torticollis (cervical dystonia), focal spasticity treatment of dynamic equinus foot deformity, focal spasticity
Faculty Disclosure. Karen L. Bremer, MD. Dr. Bremer has listed no financial interest/arrangement that would be considered a conflict of interest.
 Faculty Disclosure Karen L. Bremer, MD Dr. Bremer has listed no financial interest/arrangement that would be considered a conflict of interest. HEADACHE UPDATE Karen L. Bremer, MD November 10, 2017 karen.bremer@creighton.edu
Faculty Disclosure Karen L. Bremer, MD Dr. Bremer has listed no financial interest/arrangement that would be considered a conflict of interest. HEADACHE UPDATE Karen L. Bremer, MD November 10, 2017 karen.bremer@creighton.edu
Is Yoga an Effective Treatment for Reducing the Frequency of Episodic Migraine?
 Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 2018 Is Yoga an Effective Treatment for Reducing
Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 2018 Is Yoga an Effective Treatment for Reducing
HARVARD PILGRIM HEALTH CARE RECOMMENDED MEDICATION REQUEST GUIDELINES
 Generic Brand HICL GCN Exception/Other ONABOTULINUMTOXINA BOTOX 04867 BRAND BOTOX COSMETIC ABOBOTULINUMTOXINA DYSPORT 36477 RIMABOTULINUMTOXINB MYOBLOC 21869 INCOBOTULINUMTOXINA XEOMIN 36687 Please use
Generic Brand HICL GCN Exception/Other ONABOTULINUMTOXINA BOTOX 04867 BRAND BOTOX COSMETIC ABOBOTULINUMTOXINA DYSPORT 36477 RIMABOTULINUMTOXINB MYOBLOC 21869 INCOBOTULINUMTOXINA XEOMIN 36687 Please use
Treatment of Primary Headache Syndromes
 Presenter Disclosure Information 2:45 3:45pm Treatment of Primary Headache Syndromes SPEAKER Gerald W. Smetana, MD The following relationships exist related to this presentation: Gerald W.Smetana, MD,
Presenter Disclosure Information 2:45 3:45pm Treatment of Primary Headache Syndromes SPEAKER Gerald W. Smetana, MD The following relationships exist related to this presentation: Gerald W.Smetana, MD,
Headache: A case-based approach
 Headache: A case-based approach Rebecca Burch, MD John R. Graham Headache Center Department of Neurology Brigham and Women s Faulkner Hospital Harvard Medical School Boston, MA Disclosure of Financial
Headache: A case-based approach Rebecca Burch, MD John R. Graham Headache Center Department of Neurology Brigham and Women s Faulkner Hospital Harvard Medical School Boston, MA Disclosure of Financial
Triptan Quantity Limit
 *- Florida Healthy Kids Triptan Quantity Limit Override(s) Quantity Limit Approval Duration 1 year Oral Tablets Axert (almotriptan) tablets Relpax (eletriptan) tablets 6 tablets (6.25 mg) 12 tablets (12.5
*- Florida Healthy Kids Triptan Quantity Limit Override(s) Quantity Limit Approval Duration 1 year Oral Tablets Axert (almotriptan) tablets Relpax (eletriptan) tablets 6 tablets (6.25 mg) 12 tablets (12.5
Paediatric headaches. Dr Jaycen Cruickshank Director of Clinical Training Ballarat Health Services. Brevity, levity, repetition
 Paediatric headaches Dr Jaycen Cruickshank Director of Clinical Training Ballarat Health Services Brevity, levity, repetition Paediatric)headache?)! Headache!in!children!is!not!that!common.!The!question!is!which!headaches!do!I!
Paediatric headaches Dr Jaycen Cruickshank Director of Clinical Training Ballarat Health Services Brevity, levity, repetition Paediatric)headache?)! Headache!in!children!is!not!that!common.!The!question!is!which!headaches!do!I!
Guidance for Industry Migraine: Developing Drugs for Acute Treatment
 Guidance for Industry Migraine: Developing Drugs for Acute Treatment DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions regarding this draft
Guidance for Industry Migraine: Developing Drugs for Acute Treatment DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions regarding this draft
Transcutaneous stimulation of the cervical branch of the vagus nerve for cluster headache and migraine
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Interventional procedure consultation document Transcutaneous stimulation of the cervical branch of the vagus nerve for cluster headache and migraine Cluster
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Interventional procedure consultation document Transcutaneous stimulation of the cervical branch of the vagus nerve for cluster headache and migraine Cluster
Migraine much more than just a headache
 Migraine much more than just a headache Session hosted by Teva UK Limited PUU4 11:15 12:15 UK/NHSS/18/0021b Date of Preparation: August 2018 The views expressed in this presentation are those of the speaker
Migraine much more than just a headache Session hosted by Teva UK Limited PUU4 11:15 12:15 UK/NHSS/18/0021b Date of Preparation: August 2018 The views expressed in this presentation are those of the speaker
Disclosures. Objectives 6/2/2017
 Classification: Migraine and Trigeminal Autonomic Cephalalgias Lauren Doyle Strauss, DO, FAHS Assistant Professor, Child Neurology Assistant Director, Child Neurology Residency @StraussHeadache No disclosures
Classification: Migraine and Trigeminal Autonomic Cephalalgias Lauren Doyle Strauss, DO, FAHS Assistant Professor, Child Neurology Assistant Director, Child Neurology Residency @StraussHeadache No disclosures
10/13/17. Christy M. Jackson, MD Director, Dalessio Headache Center Scripps Clinic, La Jolla Clinical Professor, Neurosciences UCSD
 Christy M. Jackson, MD Director, Dalessio Headache Center Scripps Clinic, La Jolla Clinical Professor, Neurosciences UCSD } Depomed Consultant 2014 to present } Avanir Consultant 2014 to present } Amgen
Christy M. Jackson, MD Director, Dalessio Headache Center Scripps Clinic, La Jolla Clinical Professor, Neurosciences UCSD } Depomed Consultant 2014 to present } Avanir Consultant 2014 to present } Amgen
Ana Podgorac Belgrade, May 2012
 Headache and reproductive life Ana Podgorac Belgrade, May 2012 52 years old woman, English teacher in primary school, married, mother of two, with a history of migraine without aura. Over the last 6 months
Headache and reproductive life Ana Podgorac Belgrade, May 2012 52 years old woman, English teacher in primary school, married, mother of two, with a history of migraine without aura. Over the last 6 months
& MIGRAINE DIARY YOUR HEADACHE. Talk with your doctor about BOTOX and Chronic Migraine
 YOUR HEADACHE & MIGRAINE DIARY Do you really know how many headaches and migraines you have in a month? Keeping a diary like this one can help you find out. If you have 15 or more headache days a month,
YOUR HEADACHE & MIGRAINE DIARY Do you really know how many headaches and migraines you have in a month? Keeping a diary like this one can help you find out. If you have 15 or more headache days a month,
Headache and Facial Pain. Mohammed ALEssa MBBS, FRCSC Assistant Professor Consultant Otolaryngology,Head & Neck Surgical Oncology
 Headache and Facial Pain Mohammed ALEssa MBBS, FRCSC Assistant Professor Consultant Otolaryngology,Head & Neck Surgical Oncology Introduction It is the most common neurologic complain The diagnosis usually
Headache and Facial Pain Mohammed ALEssa MBBS, FRCSC Assistant Professor Consultant Otolaryngology,Head & Neck Surgical Oncology Introduction It is the most common neurologic complain The diagnosis usually
Ishaq Abu Arafeh Consultant Paediatrician Royal Hospital for Children, Glasgow Forth Valley Royal Hospital, Larbert
 Ishaq Abu Arafeh Consultant Paediatrician Royal Hospital for Children, Glasgow Forth Valley Royal Hospital, Larbert Childhood headache: Is it really difficult to manage? It shouldn t be... But it can be...
Ishaq Abu Arafeh Consultant Paediatrician Royal Hospital for Children, Glasgow Forth Valley Royal Hospital, Larbert Childhood headache: Is it really difficult to manage? It shouldn t be... But it can be...
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Aimovig) Reference Number: CP.PHAR.128 Effective Date: 07.10.18 Last Review Date: 08.18 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this policy
Clinical Policy: (Aimovig) Reference Number: CP.PHAR.128 Effective Date: 07.10.18 Last Review Date: 08.18 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this policy
[If yes, no further questions.]
![[If yes, no further questions.] [If yes, no further questions.]](/thumbs/94/118728940.jpg) 05/30/2014 Prior Authorization AETA BETTER HEALTH OF ILLIOIS MEDICAID Botox (IL88) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign and
05/30/2014 Prior Authorization AETA BETTER HEALTH OF ILLIOIS MEDICAID Botox (IL88) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign and
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Emgality) Reference Number: CP.PHAR.## Effective Date: 11.13.18 Last Review Date: 02.19 Line of Business: Commercial, TBD HIM*, Medicaid Revision Log See Important Reminder at the end
Clinical Policy: (Emgality) Reference Number: CP.PHAR.## Effective Date: 11.13.18 Last Review Date: 02.19 Line of Business: Commercial, TBD HIM*, Medicaid Revision Log See Important Reminder at the end
This policy addresses only the type A formulation abobotulinumtoxina marketed as Dysport.
 AbobotulinumtoxinA NDC CODE(S) 00299-5962-xx Dysport 300 UNIT VIAL (GALDERMA LABORA) 15054-0500-XX Dysport 500 UNIT SOLR (IPSEN BIOPHARMACEUTICALS) 15054-0530-XX Dysport 300 UNIT SOLR (IPSEN BIOPHARMACEUTICALS)
AbobotulinumtoxinA NDC CODE(S) 00299-5962-xx Dysport 300 UNIT VIAL (GALDERMA LABORA) 15054-0500-XX Dysport 500 UNIT SOLR (IPSEN BIOPHARMACEUTICALS) 15054-0530-XX Dysport 300 UNIT SOLR (IPSEN BIOPHARMACEUTICALS)
Faculty Disclosures. Learning Objectives. Acute Treatment Strategies
 WWW.AMERICANHEADACHESOCIETY.ORG Acute Treatment Strategies Content developed by: Lawrence C. Newman, MD, FAHS Donna Gutterman, PharmD Faculty Disclosures LAWRENCE C. NEWMAN, MD, FAHS Dr. Newman has received
WWW.AMERICANHEADACHESOCIETY.ORG Acute Treatment Strategies Content developed by: Lawrence C. Newman, MD, FAHS Donna Gutterman, PharmD Faculty Disclosures LAWRENCE C. NEWMAN, MD, FAHS Dr. Newman has received
Triptans Quantity Limit Program Summary
 Triptans Quantity Limit Program Summary FDA APPROVED INDICATIONS AND DOSAGE 1-13,14,23,24 Agents Amerge (naratriptan) 1, 2.5 tablets Axert (almotriptan) 6.25, 12.5 tablets migraine attacks with/without
Triptans Quantity Limit Program Summary FDA APPROVED INDICATIONS AND DOSAGE 1-13,14,23,24 Agents Amerge (naratriptan) 1, 2.5 tablets Axert (almotriptan) 6.25, 12.5 tablets migraine attacks with/without
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Surgical Deactivation of Headache Trigger Sites Page 1 of 12 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Surgical Deactivation of Headache Trigger Sites Professional
Surgical Deactivation of Headache Trigger Sites Page 1 of 12 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Surgical Deactivation of Headache Trigger Sites Professional
SIGN on the pharmacological management of migraine
 GUIDELINES SIGN on the pharmacological management of migraine STEVE CHAPLIN In February 2018, the Scottish Intercollegiate Guidelines Network (SIGN) published a new guideline on the pharmacological management
GUIDELINES SIGN on the pharmacological management of migraine STEVE CHAPLIN In February 2018, the Scottish Intercollegiate Guidelines Network (SIGN) published a new guideline on the pharmacological management
Study No.:MPX Title: Rationale: Phase: IIB Study Period: Study Design: Centres: Indication: Treatment: Objectives:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Headache A Practical Approach
 Headache A Practical Approach Integrated Pain Symposium December 1, 2017 Alyssa Lettich. MD Neurosciences Institute/Neurosciences Clinical Program Medical Director Headache and Pain Development Teams Disclosures:
Headache A Practical Approach Integrated Pain Symposium December 1, 2017 Alyssa Lettich. MD Neurosciences Institute/Neurosciences Clinical Program Medical Director Headache and Pain Development Teams Disclosures:
Medication Prior Authorization Form
 (OnabotulinumtoxinA) Dysport (abobotulinumtoxina) Myobloc (rimabotulinumtoxinb) Xeomin (incobotulinumtoxina) Policy Number: 1042 Policy History Approve Date: 12/11/2015 Revise Dates: 07/07/2017 Next Review:
(OnabotulinumtoxinA) Dysport (abobotulinumtoxina) Myobloc (rimabotulinumtoxinb) Xeomin (incobotulinumtoxina) Policy Number: 1042 Policy History Approve Date: 12/11/2015 Revise Dates: 07/07/2017 Next Review:
OnabotulinumtoxinA in the treatment of patients with chronic migraine: clinical evidence and experience
 731521TAN0010.1177/1756285617731521Therapeutic Advances in Neurological DisordersC-C Chiang and AJ Starling research-article2017 Therapeutic Advances in Neurological Disorders Review OnabotulinumtoxinA
731521TAN0010.1177/1756285617731521Therapeutic Advances in Neurological DisordersC-C Chiang and AJ Starling research-article2017 Therapeutic Advances in Neurological Disorders Review OnabotulinumtoxinA
Botox. Botox (onabotulinum toxin A) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.75.01 Subject: Botox Page: 1 of 8 Last Review Date: September 15, 2017 Botox Description Botox (onabotulinum
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.75.01 Subject: Botox Page: 1 of 8 Last Review Date: September 15, 2017 Botox Description Botox (onabotulinum
Botox (onabotulinumtoxina) Dysport (abobotulinumtoxina) Xeomin (incobotulinumtoxina) Myobloc (rimabotulinumtoxinb)
 Botox (onabotulinumtoxina) Dysport (abobotulinumtoxina) Xeomin (incobotulinumtoxina) Myobloc (rimabotulinumtoxinb) Line(s) of Business: HMO; PPO; QUEST Integration Akamai Advantage Original Effective Date:
Botox (onabotulinumtoxina) Dysport (abobotulinumtoxina) Xeomin (incobotulinumtoxina) Myobloc (rimabotulinumtoxinb) Line(s) of Business: HMO; PPO; QUEST Integration Akamai Advantage Original Effective Date:
