Shared Care Guideline Metolazone for fluid management in CKD (Adults)
|
|
|
- Dina Cook
- 5 years ago
- Views:
Transcription
1 Shared Care Guideline Metolazone for fluid management in CKD (Adults) It is vital for safe and appropriate patient care that there is a clear understanding of where clinical and prescribing responsibility rests between Consultants and General Practitioners (GPs). This guideline reinforces the basic premise that: When clinical and / or prescribing responsibility for a patient is transferred from hospital to GP, the GP should have full confidence to prescribe the necessary medicines. Therefore, it is essential that a transfer of care involving medicines that a GP would not normally be familiar with, should not take place without the sharing of information with the individual GP and their mutual agreement to the transfer of care. These are not rigid guidelines. On occasions, Consultants and GPs may agree to work outside of this guidance. As always, the doctor who prescribes the medication has the clinical responsibility for the drug and the consequences of its use. Indication: Metolazone is approved for use in combination with a loop diuretic for management of CKD 4/5 by nephrologists. This is a second line agent for the above indication for the few patients that do not respond sufficiently to loop diuretics alone, but have adequate urine output to warrant pursuing intensive diuresis with metolazone. Contraindications: Refractory hypokalaemia Hyponatraemia Hepatic impairment Symptomatic hyperuricaemia Anuria Symptomatic hypercalcaemia Addison s disease Dosage and administration: Starting dose: 2.5mg on alternative days or daily. Titrated by 2.5mg dose units according to response. Maximum dose: 7.5mg daily. Cautions: Diabetes and gout may be aggravated Hepatic impairment Pregnancy SLE Nephrotic Syndrome Additional Information Sanofi-Aventis discontinued the manufacturing of metolazone in Metolazone is now only available as an unlicensed special order item in the UK. Metolazone can promote dramatic diuresis and disturbance in fluid balance and electrolytes. Patients must be closely monitored and specialist initiation & advice is required. Treatment in the majority of patients will be short term & undertaken in secondary care inpatient setting only. There may however be a very small number of cases where maintenance therapy with metolazone needs to continue on discharge from the hospital, or it may need to be initiated in the community heart failure clinics to prevent an admission to hospital or for palliation. It is proposed that in these circumstances that continuing supply of the medicine is available in primary care. Patients should generally fall into one of the following 3 scenarios: 1. Discharged from hospital on metolazone for short term treatment ( 4 weeks) hospital to supply. 2. Discharged from hospital on metolazone long term hospital to supply 2 weeks then GP to prescribe 3. Initiated at outpatient appointment GP to prescribe initial supply unless there is need to initiate metolazone within 5 days (it may take 5 days before metolazone reaches the patient via a GP prescription because of the time taken to issue a prescription and for community pharmacy to procure the drug). In such cases the consultant should consider issuing a hospital outpatient prescription. Approved by MCGT Sept 2017 (Published 15/09/17) Review by: August 2020 Page 1 of 5
2 Monitoring Responsibility for monitoring is shared between the specialist clinicians and the GP. Monitoring will be co-ordinated with the GP practice. GPs are responsible for arranging all blood tests. Monitoring requirements: Before (i.e. baseline) and during metolazone treatment: Urea and electrolytes (U&Es) Creatinine Blood pressure Weight The interval for monitoring these parameters whilst a patient is on metolazone depends on values for blood pressure, renal function and serum potassium concentration; these indicate likelihood of occurrence of significant adverse effects. Patients at higher risk of adverse events should be monitored more frequently. Where any one of these parameters is within the range which places the patient at higher risk of adverse effects they should be monitored more frequently - as indicated in the table below. Risk of patient developing adverse effects: Normal risk Increased risk Initial monitoring interval (i.e. after initiation or increase in dose) SBP (mmhg) >120mmHg <120mmHg Serum potassium concentration >4.5 mmol/l <4.5 mmol/l Monitoring interval At least every 2 weeks until stable Monitoring interval once patient is stable on metolazone dose At least weekly until stable SBP (mmhg) >120mmHg <120mmHg Serum potassium concentration >4.5 mmol/l <4.5 mmol/l GFR decline (ml/min) Decrease equal to or less than 15mL/min Decrease of more than 15mL/min Increase in creatinine <30% >30% Monitoring interval At least every 6months Every 1 to 3months depending on patient stability Patients should be weighed or encouraged to self-weigh daily. Aim of maintenance therapy is to maintain a constant dry weight, and patients should be advised to contact GP or nephrologist for advice if weight is increasing or decreasing. Aim of acute metolazone diuresis is for patient to reach a target dry weight. Patients should be advised to contact GP or nephrologist once target dry weight has been reached for advice. Approved by MCGT Sept 2017 (Published 15/09/17) Review by: August 2020 Page 2 of 5
3 Action to be taken if abnormal results/adverse effects: This should be a shared care approach. The named nephrologist for the patient should be available to offer advice, where necessary, regarding clinical parameters and blood tests. Alternatively, in their absence, the nephrologist of the week can provide advice. For contact details of the nephrologists see the advice and support section below. The following provides some guidance for GPs if abnormal results are picked up. Weight Change Weight loss beyond target dry weight consider reducing dose and/or stopping temporarily. Weight gain associated with worsening symptoms (increased dyspnoea, fatigue, oedema, weight gain) - confirm compliance and consider increasing dose. Creatinine levels: An increase of less than 30% from baseline does not normally require action. However recheck creatinine within 2 weeks to confirm no further increase. If creatinine increases by 30 50% - review volume status and then reduce dose or stop diuretics (if the person is hypovolaemic). Re-measure renal function within 1 week. If creatinine increases by more than 50% - assess volume status, check blood pressure and review other renal function tests, including electrolytes and proteinuria. If the person is hypovolaemic, stop the diuretic. If there is any uncertainty, contact nephrologist / heart failure nurses / cardiologist urgently. Potassium: If potassium level <3 mmol/l (or <4 mmol/l in patients at high risk of cardiac arrhythmias), ensure patient is reviewed urgently to: o prevent further drop in potassium by initiating potassium supplements. o assess if admission to hospital is required for urgent potassium replacement. People at high risk of cardiac arrhythmias with even mild hypokalaemia include: o Those taking digoxin or drugs that prolong the QT interval (such as amiodarone). o Those with paroxysmal arrhythmias, unstable angina, or chronic liver disease. Give potassium supplements: Sando K 24mmol TDS for 2-3days, repeat prescriptions until potassium is >4mmol/l. Beware that levels will continue to rise once supplements have been discontinued. Potassium levels should be rechecked on a 24-48hourly basis until >4mmol/L. Then repeat U&Es within 7days. If potassium concentration < 2.5 mmol/l (or 3.5 mmol/l in high-risk people) admit patient to hospital for urgent potassium replacement. Sodium: If <131mmol/L repeat level the next day. Consider reduction in diuretic therapy if clinically stable. Symptomatic Hypotension:(Systolic pressure < 90 mmhg associated with dizziness, fainting, confusion) Review antihypertensives, check blood chemistry to exclude other causes for symptoms, consider reduction in diuretic therapy if clinically stable. Adverse Effects: Mild gastro-intestinal disturbances, postural hypotension, altered plasma-lipid concentrations, metabolic and electrolyte disturbances including hypokalaemia (see abnormal results section above), hyponatraemia, hypomagnesaemia, hypercalcaemia, hyperglycaemia, hypochloraemic alkalosis, hyperuricaemia, and gout. Less common side-effects include blood disorders such as agranulocytosis, leucopenia, and thrombocytopenia, and impotence. Pancreatitis, intrahepatic cholestasis, cardiac arrhythmias, headache, dizziness, paraesthesia, visual disturbances, and hypersensitivity reactions (including pneumonitis, pulmonary oedema, photosensitivity, and severe skin reactions) have also been reported. Drug interactions: Hypokalaemia caused by thiazides and related diuretics increases cardiac toxicity with cardiac glycosides, flecainide, lidocaine, disopyramide, sotalol. Hypokalaemia caused by diuretics increases risk of ventricular arrhythmias with amisulpride, atomoxetine, pimozide (avoid concurrent use with pimozide) Hypokalaemia caused by thiazides and related diuretics antagonises action of lidocaine (less likely with topical lidocaine) Thiazides and related diuretics reduce excretion of lithium (increased plasma concentration and risk of toxicity) Enhanced hypotensive effect when diuretics given with ACE inhibitors, alpha-blockers, angiotensin-ii receptor antagonists Diuretics increase risk of nephrotoxicity of NSAIDs, also antagonism of diuretic effect Please see BNF for further details of interactions Approved by MCGT Sept 2017 (Published 15/09/17) Review by: August 2020 Page 3 of 5
4 Specialist responsibilities: 1. Confirm diagnosis and indication for metolazone use 2. Discuss benefits and side effects of metolazone treatment with the patient and document that the patient has given informed consent to the unlicensed use of metolazone. 3. Ensure baseline monitoring of biochemical profile & pre-treatment weight of patient completed 4. Ensure that the patient understands and accepts their responsibilities (see section below) 5. Ensure patient is aware of the signs of over diuresis or worsening symptoms, when to seek medical advice from their GP. 6. Arrange for initiation of metolazone as outline above. 7. Provide written instruction to the GP for the initiation and on-going management of metolazone treatment. 8. Ensure patient is aware that GP has to issue a prescription and then it may take 1 or 2 days for community pharmacy to supply as unlicensed. 9. Ensure that a clear management plan is supplied to the GP for the individual patient and that systems exist for GPs to obtain specialist advice and support. 10. Review patient at regular intervals or on GP request to monitor disease progress and continued need for metolazone therapy. 11. Promptly communicate with the GP via a clinic letter any changes in treatment, results of tests undertaken and assessment of adverse events. Clinic letter should clearly state whether the dose has remained the same or if a dose adjustment has been made specifically highlighting the new dose and for how long treatment should continue. 12. Discontinue metolazone if patient has an adverse event or no longer required diuresis. 13. Report serious adverse events to MHRA on yellow card GP s responsibilities: 1. Initial referral to secondary care for assessment of CKD. 2. Provide the patient with monthly repeat prescriptions of metolazone at the nephrologist s request as detailed above. 3. Ensure patient is having regular monitoring done as outlined above and ensure results are available on clinical system. 4. Check recent blood results are within range as per above guidance before issuing each repeat prescription of metolazone. 5. Carry out further dose titration according to response or discontinue medication when necessary or requested. Whenever necessary discuss with nephrologist. 6. Ensure practice computer is updated with any dose changes. 7. Seek advice from nephrologist if the patient s condition deteriorates or if there is a change in the patient s status. 8. Contact a nephrologist if not in agreement with the treatment recommendation, or if there is a perceived problem with compliance or concordance, or if they have any questions about the management plan. 9. Report any adverse effects to the nephrologist managing the patient. 14. Report serious adverse events to MHRA on yellow card Patient s responsibilities: 1. Read the written information provided about the drug from the community pharmacy (Patient Information Leaflet) and have a clear understanding of the risks / benefits of metolazone treatment 2. MUST attend for blood tests. 3. Allow at least 48 hours for the prescription from the GP to be generated (once the GP has agreed to take on the prescribing.) 4. Allow at least 48 hours for the community pharmacy to obtain the metolazone as it is an unlicensed medicine. 5. Report any adverse effects, concerns or lack of understanding of the treatment to the GP or specialist. 6. Weigh themselves regularly as requested by clinicians. 7. Ensure they report signs of over diuresis or any signs of worsening symptoms Community Pharmacy responsibilities: 1. Ensure prescriber is aware of unlicensed status of medicine 2. Obtain appropriate product (see section below re metolazone availability). 3. Supply an English PIL (supplied with product from IDIS) 4. Maintain named patient records as it is an unlicensed medicine Approved by MCGT Sept 2017 (Published 15/09/17) Review by: August 2020 Page 4 of 5
5 Metolazone availability: It is only available as an unlicensed medicine in the UK. Please be aware that there are a number of products from different countries with the same brand name Zaroxolyn. The products differ in strengths and the colourants that are used in them. The preferred product to be used on Wirral is metolazone (Zaroxolyn ) 2.5mg from Canada as it is the only product that is available as a 2.5mg tablet (the most commonly used strength). Advice and support: Principal contact: Named nephrologist for the patient via the nephrology secretaries. Contact via switchboard. Telephone: and ask for the secretary of the relevant nephrologist. Emergency out-of-hours advice only: Nephrologist of the week, available via the on-call junior renal doctor 7 days a week 9-8pm. Contact via switchboard. Telephone: and ask for Bleep Written By: Rachael Pugh, Prescribing Advisor, Medicines Management Team, MLCSU Victoria Keers, Highly Specialist Pharmacist for Renal Medicine, WUTH ( Updated Aine McManus August 2017) Approved by MCGT Sept 2017 (Published 15/09/17) Review by: August 2020 Page 5 of 5
keyword: diuretics Drug monitoring Monitoring diuretics in primary care 2 March 2009 best tests
 www.bpac.org.nz keyword: diuretics Drug monitoring Monitoring diuretics in primary care 2 March 2009 best tests Why do we monitor patients taking diuretics and what do we monitor? Monitoring a person on
www.bpac.org.nz keyword: diuretics Drug monitoring Monitoring diuretics in primary care 2 March 2009 best tests Why do we monitor patients taking diuretics and what do we monitor? Monitoring a person on
ESCA: Cinacalcet (Mimpara )
 ESCA: Cinacalcet (Mimpara ) Effective Shared Care Agreement for the Treatment of Primary hyperparathyroidism when parathyroidectomy is contraindicated or not clinically appropriate. Specialist details
ESCA: Cinacalcet (Mimpara ) Effective Shared Care Agreement for the Treatment of Primary hyperparathyroidism when parathyroidectomy is contraindicated or not clinically appropriate. Specialist details
Drug Name: Lithium Clinical Indications: Treatment and prophylaxis of mania; bipolar disorder; augmentation therapy in treatment resistant depression
 SHARED CARE PROTOCOL AND INFORMATION FOR GPS Drug Name: Lithium Clinical Indications: Treatment and prophylaxis of mania; bipolar disorder; augmentation therapy in treatment resistant depression Version:
SHARED CARE PROTOCOL AND INFORMATION FOR GPS Drug Name: Lithium Clinical Indications: Treatment and prophylaxis of mania; bipolar disorder; augmentation therapy in treatment resistant depression Version:
Frimley Health Area Prescribing Committee
 Frimley Health Area Prescribing Committee Frimley Health NHS Foundation Trust North East Hampshire and Farnham CCG East Berkshire CCG Surrey Heath CCG Buckinghamshire CCG SHARED CARE Guideline Amber Traffic
Frimley Health Area Prescribing Committee Frimley Health NHS Foundation Trust North East Hampshire and Farnham CCG East Berkshire CCG Surrey Heath CCG Buckinghamshire CCG SHARED CARE Guideline Amber Traffic
SHARED CARE GUIDELINE
 SHARED CARE GUIDELINE Title: Lithium Treatment in Adults aged 18-65 years Scope: Pennine Care NHS Foundation Trust NHS Bury NHS Oldham NHS Heywood, Middleton and Rochdale NHS Stockport NHS Tameside & Glossop
SHARED CARE GUIDELINE Title: Lithium Treatment in Adults aged 18-65 years Scope: Pennine Care NHS Foundation Trust NHS Bury NHS Oldham NHS Heywood, Middleton and Rochdale NHS Stockport NHS Tameside & Glossop
NHS Kent and Medway Medicines Management. Dronedarone (Multaq ) Shared Care Guideline For Prescribing
 NHS Kent and Medway Medicines Management Dronedarone (Multaq ) Shared Care Guideline For Prescribing Issue No: 2 Review Date (If Applicable): Accountable Officer: Heather Lucas Contact Details: 01233 618158
NHS Kent and Medway Medicines Management Dronedarone (Multaq ) Shared Care Guideline For Prescribing Issue No: 2 Review Date (If Applicable): Accountable Officer: Heather Lucas Contact Details: 01233 618158
Guidelines for the Prescribing of Sacubitril / Valsartan
 Hull & East Riding Prescribing Committee Guidelines for the Prescribing of Sacubitril / Valsartan 1. BACKGROUND Sacubitril valsartan is an angiotensin receptor neprilysin inhibitor, including both a neprilysin
Hull & East Riding Prescribing Committee Guidelines for the Prescribing of Sacubitril / Valsartan 1. BACKGROUND Sacubitril valsartan is an angiotensin receptor neprilysin inhibitor, including both a neprilysin
Prescribing Framework for Methotrexate for Immunosuppression in ADULTS
 Hull & East Riding Prescribing Committee Prescribing Framework for Methotrexate for Immunosuppression in ADULTS Patient s Name:.. NHS Number: Patient s Address:... (Use addressograph sticker) GP s Name:...
Hull & East Riding Prescribing Committee Prescribing Framework for Methotrexate for Immunosuppression in ADULTS Patient s Name:.. NHS Number: Patient s Address:... (Use addressograph sticker) GP s Name:...
Treatment monitoring protocol for Dimethyl fumarate therapy in active Relapsing Remitting Multiple Sclerosis
 Treatment monitoring protocol for Dimethyl fumarate therapy in active Relapsing Remitting Multiple Sclerosis This protocol provides monitoring guidance for adult patients requiring Dimethyl fumarate therapy
Treatment monitoring protocol for Dimethyl fumarate therapy in active Relapsing Remitting Multiple Sclerosis This protocol provides monitoring guidance for adult patients requiring Dimethyl fumarate therapy
Essential Shared Care Agreement: Lithium
 Ref No. E042 Essential Shared Care Agreement: Lithium Please complete the following details: Patient s name, address, date of birth Treatment (indication, dose regimen, brand name) Monitoring (proposed
Ref No. E042 Essential Shared Care Agreement: Lithium Please complete the following details: Patient s name, address, date of birth Treatment (indication, dose regimen, brand name) Monitoring (proposed
Western Locality Shared care Information ~ Penicillamine, Rheumatology April 2013
 Western Locality Shared care Information ~ Penicillamine, Rheumatology April 2013 Penicillamine Treatment of: Rheumatoid arthritis Specialist: Please complete the Shared Care letter sending a request to
Western Locality Shared care Information ~ Penicillamine, Rheumatology April 2013 Penicillamine Treatment of: Rheumatoid arthritis Specialist: Please complete the Shared Care letter sending a request to
EFFECTIVE SHARE CARE AGREEMENT. FOR THE off license use of GLP1 mimetics in combination with insulin IN DUDLEY
 Specialist details Patient identifier Name Tel: EFFECTIVE SHARE CARE AGREEMENT FOR THE off license use of GLP1 mimetics in combination with insulin IN DUDLEY The aim of Effective Shared Care Guidelines
Specialist details Patient identifier Name Tel: EFFECTIVE SHARE CARE AGREEMENT FOR THE off license use of GLP1 mimetics in combination with insulin IN DUDLEY The aim of Effective Shared Care Guidelines
Chronic heart failure: management of chronic heart failure in adults in primary and secondary care (partial update)
 Chronic heart failure: management of chronic heart failure in adults in primary and secondary care (partial update) NICE guideline Apendix C The algorithms Draft for consultation, January 2010 Chronic
Chronic heart failure: management of chronic heart failure in adults in primary and secondary care (partial update) NICE guideline Apendix C The algorithms Draft for consultation, January 2010 Chronic
Shared Care Guideline for Olanzapine (Zyprexa )
 Shared Care Guideline for Olanzapine (Zyprexa ) Development Process This guidance has been produced by Sarah Hudson Lead Pharmacist SWYPFT following an AMBER classification status of Olanzapine by the
Shared Care Guideline for Olanzapine (Zyprexa ) Development Process This guidance has been produced by Sarah Hudson Lead Pharmacist SWYPFT following an AMBER classification status of Olanzapine by the
Atomoxetine Effective Shared Care Agreement For Attention Deficit Hyperactivity Disorder (ADHD)
 Atomoxetine Effective Shared Care Agreement For Attention Deficit Hyperactivity Disorder (ADHD) Section 1: Shared Care arrangements and responsibilities Section 1.1 Agreement to transfer of prescribing
Atomoxetine Effective Shared Care Agreement For Attention Deficit Hyperactivity Disorder (ADHD) Section 1: Shared Care arrangements and responsibilities Section 1.1 Agreement to transfer of prescribing
PRESCRIBING GUIDANCE METHOTREXATE for the treatment of vasculitis
 PRESCRIBING GUIDANCE METHOTREXATE for the treatment of vasculitis For the latest information on interactions and adverse effects, always consult the latest version of the Summary of Product Characteristics
PRESCRIBING GUIDANCE METHOTREXATE for the treatment of vasculitis For the latest information on interactions and adverse effects, always consult the latest version of the Summary of Product Characteristics
Younger adults with a family history of premature artherosclerotic disease should have their cardiovascular risk factors measured.
 Appendix 2A - Guidance on Management of Hypertension Measurement of blood pressure All adults from 40 years should have blood pressure measured as part of opportunistic cardiovascular risk assessment.
Appendix 2A - Guidance on Management of Hypertension Measurement of blood pressure All adults from 40 years should have blood pressure measured as part of opportunistic cardiovascular risk assessment.
Lithium Shared Care Protocol and Information for Oxfordshire GPs
 Summary Lithium Shared Care Protocol and Information for Oxfordshire GPs 1. Lithium is predominantly used in the following situations: a. The acute treatment of mania b. Prophylaxis in bipolar disorder
Summary Lithium Shared Care Protocol and Information for Oxfordshire GPs 1. Lithium is predominantly used in the following situations: a. The acute treatment of mania b. Prophylaxis in bipolar disorder
Factsheet Sacubitril valsartan (Entresto ) for chronic heart failure with reduced ejection fraction
 North Central London Joint Formulary Committee Factsheet Sacubitril valsartan (Entresto ) for chronic heart failure with reduced ejection fraction Start date: July 2016 Review date: July 2019 Document
North Central London Joint Formulary Committee Factsheet Sacubitril valsartan (Entresto ) for chronic heart failure with reduced ejection fraction Start date: July 2016 Review date: July 2019 Document
AREAS OF RESPONSIBILITY FOR THE SHARING OF CARE
 Effective Shared Care Agreement (ESCA) Midodrine ESCA: For the treatment of severe orthostatic hypotension due to autonomic dysfunction when corrective factors have been ruled out and other forms of treatment
Effective Shared Care Agreement (ESCA) Midodrine ESCA: For the treatment of severe orthostatic hypotension due to autonomic dysfunction when corrective factors have been ruled out and other forms of treatment
Prescribing Framework for Galantamine in the Treatment and Management of Dementia
 Hull & East Riding Prescribing Committee Prescribing Framework for Galantamine in the Treatment and Management of Dementia Patients Name:.. NHS Number: Patients Address:... (Use addressograph sticker)
Hull & East Riding Prescribing Committee Prescribing Framework for Galantamine in the Treatment and Management of Dementia Patients Name:.. NHS Number: Patients Address:... (Use addressograph sticker)
Patient details GP details Specialist details Name GP Name Dr Specialist Name Dr R. Horton
 Rationale for Initiation, Continuation and Discontinuation (RICaD) Sacubitril/Valsartan (Entresto) For the treatment of symptomatic heart failure with reduced ejection fraction (NICE TA388) This document
Rationale for Initiation, Continuation and Discontinuation (RICaD) Sacubitril/Valsartan (Entresto) For the treatment of symptomatic heart failure with reduced ejection fraction (NICE TA388) This document
ZAROXOLYN 5mg tablets ZAROXOLYN 10 mg tablets Metolazone. PHARMACOTHERAPEUTIC CATEGORY Diuretic with minor diuretic action, non-associated.
 ZAROXOLYN 5mg tablets ZAROXOLYN 10 mg tablets Metolazone PHARMACOTHERAPEUTIC CATEGORY Diuretic with minor diuretic action, non-associated. THERAPEUTIC INDICATIONS Zaroxolyn is indicated in the treatment
ZAROXOLYN 5mg tablets ZAROXOLYN 10 mg tablets Metolazone PHARMACOTHERAPEUTIC CATEGORY Diuretic with minor diuretic action, non-associated. THERAPEUTIC INDICATIONS Zaroxolyn is indicated in the treatment
SHARED CARE GUIDELINE For
 SHARED CARE GUIDELINE For Leuprorelin (Prostap 3 DCS ) for advanced hormone dependent prostate cancer Implementation Date: Review Date: This guidance has been prepared and approved for use within Gateshead,
SHARED CARE GUIDELINE For Leuprorelin (Prostap 3 DCS ) for advanced hormone dependent prostate cancer Implementation Date: Review Date: This guidance has been prepared and approved for use within Gateshead,
Effective Shared Care Agreement (ESCA) for drugs used in dementia- Donepezil, Galantamine, Rivastigmine and Memantine
 Effective Shared Care Agreement (ESCA) for drugs used in dementia- Donepezil, Galantamine, Rivastigmine and Memantine for the treatment of dementia AREAS OF RESPONSIBILITY FOR THE SHARING OF CARE This
Effective Shared Care Agreement (ESCA) for drugs used in dementia- Donepezil, Galantamine, Rivastigmine and Memantine for the treatment of dementia AREAS OF RESPONSIBILITY FOR THE SHARING OF CARE This
Heart Failure (HF) - Primary Care Flow Charts. Pre diagnosis Symptoms or signs suggestive of HF
 Heart Failure (HF) - Primary Care Flow Charts Pre diagnosis Symptoms or signs suggestive of HF 12 lead ECG Normal examination and 12 lead ECG HF highly unlikely Abnormal 12 lead ECG HF Possible Arrange
Heart Failure (HF) - Primary Care Flow Charts Pre diagnosis Symptoms or signs suggestive of HF 12 lead ECG Normal examination and 12 lead ECG HF highly unlikely Abnormal 12 lead ECG HF Possible Arrange
Heart Failure (HF) - Primary Care Flow Charts. Symptoms or signs suggestive of HF. Pre diagnosis. Refer to the Heart Failure Clinic at VHK for
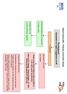 Heart Failure (HF) - Primary Care Flow Charts Pre diagnosis Symptoms or signs suggestive of HF 12 lead ECG Normal examination and 12 lead ECG HF highly unlikely Abnormal 12 lead ECG HF Possible Arrange
Heart Failure (HF) - Primary Care Flow Charts Pre diagnosis Symptoms or signs suggestive of HF 12 lead ECG Normal examination and 12 lead ECG HF highly unlikely Abnormal 12 lead ECG HF Possible Arrange
Essential Shared Care Agreement Drugs for Dementia
 E098 Essential Shared Care Agreement Drugs for Dementia Please complete the following details: Patient s name, address, date of birth Consultant s contact details (p.3) And send One copy to: 1. the patient
E098 Essential Shared Care Agreement Drugs for Dementia Please complete the following details: Patient s name, address, date of birth Consultant s contact details (p.3) And send One copy to: 1. the patient
SHARED CARE GUIDELINE: Mycophenolate mofetil or mycophenolic acid for Maintenance of Immunosuppression after Kidney Transplantation in Adults
 NHS LINCOLNSHIRE in association with UNITED LINCOLNSHIRE HOSPITALS TRUST SHARED CARE GUIDELINE: Mycophenolate mofetil or mycophenolic acid for Maintenance of Immunosuppression after Kidney Transplantation
NHS LINCOLNSHIRE in association with UNITED LINCOLNSHIRE HOSPITALS TRUST SHARED CARE GUIDELINE: Mycophenolate mofetil or mycophenolic acid for Maintenance of Immunosuppression after Kidney Transplantation
SUMMARY OF PRODUCT CHARACTERISTICS 2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Bendroflumethiazide 2.5mg Tablets Neo-Naclex 2.5mg Tablets 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains Bendroflumethiazide
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Bendroflumethiazide 2.5mg Tablets Neo-Naclex 2.5mg Tablets 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains Bendroflumethiazide
NHS GG&C -Mental Health Services. Lithium Ward Bundle
 Lithium Ward Bundle Lithium is a useful drug, particularly in the maintenance treatment of bipolar affective disorder, recurrent depression and self injurious behaviour. It is widely used and most patients
Lithium Ward Bundle Lithium is a useful drug, particularly in the maintenance treatment of bipolar affective disorder, recurrent depression and self injurious behaviour. It is widely used and most patients
BNSSG Shared Care Guidance Please complete all sections
 NHS Bristol CCG NHS North Somerset CCG NHS South Gloucestershire CCG North Bristol NHS Trust University Hospitals Bristol NHS Foundation Trust Weston Area Health NHS Trust BNSSG Shared Care Guidance Please
NHS Bristol CCG NHS North Somerset CCG NHS South Gloucestershire CCG North Bristol NHS Trust University Hospitals Bristol NHS Foundation Trust Weston Area Health NHS Trust BNSSG Shared Care Guidance Please
PUBLIC SUMMARY OF RISK MANAGEMENT PLAN (RMP) CANDESARTAN/HYDROCHLOROTHIAZIDE ORION 8 MG/12.5 MG, 16 MG/12.5 MG, 32 MG/12.5 MG, 32 MG/25 MG ORION
 PUBLIC SUMMARY OF RISK MANAGEMENT PLAN (RMP) CANDESARTAN/HYDROCHLOROTHIAZIDE ORION 8 MG/12.5 MG, 16 MG/12.5 MG, 32 MG/12.5 MG, 32 MG/25 MG ORION CORPORATION DATE: 17-04-2015, VERSION 2 VI.2 Elements for
PUBLIC SUMMARY OF RISK MANAGEMENT PLAN (RMP) CANDESARTAN/HYDROCHLOROTHIAZIDE ORION 8 MG/12.5 MG, 16 MG/12.5 MG, 32 MG/12.5 MG, 32 MG/25 MG ORION CORPORATION DATE: 17-04-2015, VERSION 2 VI.2 Elements for
EXENATIDE (BYETTA ) PROTOCOL, 5mcg and 10mcg SC injection pre-filled pens
 EXENATIDE (BYETTA ) PROTOCOL, 5mcg and 10mcg SC injection pre-filled pens This document should be read in conjunction with the current Summary of Product Characteristics http://www.medicines.org.uk 1.
EXENATIDE (BYETTA ) PROTOCOL, 5mcg and 10mcg SC injection pre-filled pens This document should be read in conjunction with the current Summary of Product Characteristics http://www.medicines.org.uk 1.
Specialist Palliative Care Audit and Guidelines Group (SPAGG)
 Specialist Palliative Care Audit and Guidelines Group (SPAGG) Clinical Guideline for the Prescribing and Administration of Furosemide via continuous subcutaneous infusion (CSCI) for Heart Failure Patients
Specialist Palliative Care Audit and Guidelines Group (SPAGG) Clinical Guideline for the Prescribing and Administration of Furosemide via continuous subcutaneous infusion (CSCI) for Heart Failure Patients
Dorset Health Technologies Forum SHARED CARE GUIDELINE FOR PRESCRIBING EPLERENONE (INSPRA )
 INDICATION SHARED CARE GUIDELINE FOR PRESCRIBING EPLERENONE (INSPRA ) Eplerenone is an aldosterone antagonist licensed to be used in addition to standard therapy including beta-blockers, to reduce the
INDICATION SHARED CARE GUIDELINE FOR PRESCRIBING EPLERENONE (INSPRA ) Eplerenone is an aldosterone antagonist licensed to be used in addition to standard therapy including beta-blockers, to reduce the
Shared Care Agreement for Donepezil
 ESCA: for the treatment of Alzheimer s disease SECONDARY CARE SECTION TO BE COMPLETED BY INITIATING DOCTOR Patient s Name: Date of Birth: NHS Number: ESCA Date: One copy of information leaflet given to
ESCA: for the treatment of Alzheimer s disease SECONDARY CARE SECTION TO BE COMPLETED BY INITIATING DOCTOR Patient s Name: Date of Birth: NHS Number: ESCA Date: One copy of information leaflet given to
Tacrolimus (Adoport, Prograf, Modigraf or Advagraf )
 Shared Care Guideline DRUG: Tacrolimus (Adoport, Prograf, Modigraf or Advagraf ) for Renal Transplant (Adults) Introduction: Indication: Prophylaxis of transplant rejection in kidney recipients Tacrolimus
Shared Care Guideline DRUG: Tacrolimus (Adoport, Prograf, Modigraf or Advagraf ) for Renal Transplant (Adults) Introduction: Indication: Prophylaxis of transplant rejection in kidney recipients Tacrolimus
Analgesia in patients with impaired renal function Formulary Guidance
 Analgesia in patients with impaired renal function Formulary Guidance Approved by Trust D&TC: January 2010 Revised March 2017 Contents Paragraph Page 1 Aim 4 2 Introduction 4 3 Assessment of renal function
Analgesia in patients with impaired renal function Formulary Guidance Approved by Trust D&TC: January 2010 Revised March 2017 Contents Paragraph Page 1 Aim 4 2 Introduction 4 3 Assessment of renal function
SHARED CARE AGREEMENT: METHOTREXATE S/C
 NB: This document should be read in conjunction with the current Summary of Product Characteristics (SPC) and BSR Guideline for disease-modifying anti-rheumatic drug (DMARD) therapy (available at www.rheumatology.org.uk/resources/guidelines/bsr_guidelines.aspx
NB: This document should be read in conjunction with the current Summary of Product Characteristics (SPC) and BSR Guideline for disease-modifying anti-rheumatic drug (DMARD) therapy (available at www.rheumatology.org.uk/resources/guidelines/bsr_guidelines.aspx
Prescribing Framework for Rivastigmine in the Treatment and Management of Dementia
 Hull & East Riding Prescribing Committee Prescribing Framework for Rivastigmine in the Treatment and Management of Dementia Patients Name:.. NHS Number: Patients Address:... (Use addressograph sticker)
Hull & East Riding Prescribing Committee Prescribing Framework for Rivastigmine in the Treatment and Management of Dementia Patients Name:.. NHS Number: Patients Address:... (Use addressograph sticker)
NHS LINCOLNSHIRE in association with UNITED LINCOLNSHIRE HOSPITALS TRUST
 NHS LINCOLNSHIRE in association with UNITED LINCOLNSHIRE HOSPITALS TRUST SHARED CARE GUIDELINE: CINACALCET in the management of secondary hyperparathyroidism in adult patients with end-stage renal disease
NHS LINCOLNSHIRE in association with UNITED LINCOLNSHIRE HOSPITALS TRUST SHARED CARE GUIDELINE: CINACALCET in the management of secondary hyperparathyroidism in adult patients with end-stage renal disease
SULFASALAZINE (Adults)
 Shared Care Guideline DRUG: Introduction: SULFASALAZINE (Adults) Indication: Disease modifying drug for rheumatoid arthritis, psoriatic arthritis, undifferentiated arthritis, spondyloarthropathies, Crohn
Shared Care Guideline DRUG: Introduction: SULFASALAZINE (Adults) Indication: Disease modifying drug for rheumatoid arthritis, psoriatic arthritis, undifferentiated arthritis, spondyloarthropathies, Crohn
Essential Shared Care Agreement Drugs for Dementia
 Ref No. E052 Please complete the following details: Patient s name, address, date of birth Consultant s contact details (p.3) And send One copy to: 1. the patient s GP 2. put one copy in care plan 3. give
Ref No. E052 Please complete the following details: Patient s name, address, date of birth Consultant s contact details (p.3) And send One copy to: 1. the patient s GP 2. put one copy in care plan 3. give
Hydroxychloroquine (Adults)
 Shared Care Guideline DRUG: Introduction: Contraindications & Warnings: Hydroxychloroquine (Adults) Indication: Rheumatoid arthritis and inflammatory osteoarthritis, discoid and systemic lupus erythematosus,
Shared Care Guideline DRUG: Introduction: Contraindications & Warnings: Hydroxychloroquine (Adults) Indication: Rheumatoid arthritis and inflammatory osteoarthritis, discoid and systemic lupus erythematosus,
Prescribing Guidelines Prescribing arrangement for the management of patients transferring from Secondary Care to Primary Care
 Berkshire West Integrated Care System Representing Berkshire West Clinical Commisioning Group Royal Berkshire NHS Foundation Trust Berkshire Healthcare NHS Foundation Trust Berkshire West Primary Care
Berkshire West Integrated Care System Representing Berkshire West Clinical Commisioning Group Royal Berkshire NHS Foundation Trust Berkshire Healthcare NHS Foundation Trust Berkshire West Primary Care
patient group direction
 ACICLOVIR v01 1/8 ACICLOVIR PGD Details Version 1.0 Legal category Staff grades Approved by POM Paramedic (Non-ECP) Nurse (Non-ECP) Emergency Care Practitioner (Paramedic) Emergency Care Practitioner (Nurse)
ACICLOVIR v01 1/8 ACICLOVIR PGD Details Version 1.0 Legal category Staff grades Approved by POM Paramedic (Non-ECP) Nurse (Non-ECP) Emergency Care Practitioner (Paramedic) Emergency Care Practitioner (Nurse)
Heart Failure Nurse Specialist: Pharmacological Management of Heart Failure
 Heart Failure Nurse Specialist: Pharmacological Management of Heart Failure Author(s) & Designation Lead Clinician if appropriate In consultation with To be read in association with Ratified by Linda Edmunds,
Heart Failure Nurse Specialist: Pharmacological Management of Heart Failure Author(s) & Designation Lead Clinician if appropriate In consultation with To be read in association with Ratified by Linda Edmunds,
1. Despite the plethora of new ACE-inhibitors they offer little advantage over the earlier products captopril and enalapril.
 SUMMARY 1. Despite the plethora of new ACE-inhibitors they offer little advantage over the earlier products captopril and enalapril. 2. While diuretics and beta-blockers remain first-line antihypertensive
SUMMARY 1. Despite the plethora of new ACE-inhibitors they offer little advantage over the earlier products captopril and enalapril. 2. While diuretics and beta-blockers remain first-line antihypertensive
ACE. Inhibitors. Quiz feedback
 ACE Inhibitors Quiz feedback bpac nz better medicin e bpac nz Quiz feedback, ACE inhibitors, 2006 Best Practice Advocacy Centre ACE inhibitors quiz feedback bpac nz Development Team: Rachael Clarke Sonia
ACE Inhibitors Quiz feedback bpac nz better medicin e bpac nz Quiz feedback, ACE inhibitors, 2006 Best Practice Advocacy Centre ACE inhibitors quiz feedback bpac nz Development Team: Rachael Clarke Sonia
CLINICAL GUIDELINES ID TAG
 CLINICAL GUIDELINES ID TAG Title: Treatment of Hypomagnesaemia in adults Author: Speciality / Division: Directorate: Dr Peter Sharpe, Dr Neal Morgan, Jillian Redpath Chemical Pathology/Nephrology/Pharmacy
CLINICAL GUIDELINES ID TAG Title: Treatment of Hypomagnesaemia in adults Author: Speciality / Division: Directorate: Dr Peter Sharpe, Dr Neal Morgan, Jillian Redpath Chemical Pathology/Nephrology/Pharmacy
PRESCRIBING GUIDANCE TACROLIMUS for the treatment of INFLAMATORY BOWEL DISEASE (IBD)
 PRESCRIBING GUIDANCE TACROLIMUS for the treatment of INFLAMATORY BOWEL DISEASE (IBD) For the latest information on interactions and adverse effects, always consult the latest version of the Summary of
PRESCRIBING GUIDANCE TACROLIMUS for the treatment of INFLAMATORY BOWEL DISEASE (IBD) For the latest information on interactions and adverse effects, always consult the latest version of the Summary of
Doncaster & Bassetlaw. AKI guidelines for primary care
 Doncaster & Bassetlaw AKI guidelines for primary care Contents: FLOW DIAGRAM: MANAGEMENT OF PATIENTS WITH AKI DETECTED IN PRIMARY CARE... 2 FLOW DIAGRAM: MANAGEMENT OF HYPERKALAEMIA.... 3 FLOW DIAGRAM:
Doncaster & Bassetlaw AKI guidelines for primary care Contents: FLOW DIAGRAM: MANAGEMENT OF PATIENTS WITH AKI DETECTED IN PRIMARY CARE... 2 FLOW DIAGRAM: MANAGEMENT OF HYPERKALAEMIA.... 3 FLOW DIAGRAM:
Bournemouth, Dorset and Poole Prescribing Forum
 SHARED CARE GUIDELINE FOR THE USE OF ATOMOXETINE IN ADULTS WITH ATTENTION DEFICIT HYPERACTIVITY DISORDER INDICATION Atomoxetine is a non-stimulant non-amphetamine inhibitor of noradrenaline reuptake. It
SHARED CARE GUIDELINE FOR THE USE OF ATOMOXETINE IN ADULTS WITH ATTENTION DEFICIT HYPERACTIVITY DISORDER INDICATION Atomoxetine is a non-stimulant non-amphetamine inhibitor of noradrenaline reuptake. It
Review guidance for patients on long-term amiodarone treatment
 Review guidance for patients on long-term amiodarone treatment This review guidance document has been produced in response to: 1. Current supply shortages of branded and generic versions of 100mg and 200mg
Review guidance for patients on long-term amiodarone treatment This review guidance document has been produced in response to: 1. Current supply shortages of branded and generic versions of 100mg and 200mg
for adults engaged with the Family Wellbeing Service Isle of Wight In Community Pharmacy for Isle of Wight Public Health Commissioned Services
 The supply of Champix (Varenicline) Tablets 500mcg and 1mg by registered community pharmacists for smoking cessation / management of nicotine withdrawal for adults engaged with the Family Wellbeing Service
The supply of Champix (Varenicline) Tablets 500mcg and 1mg by registered community pharmacists for smoking cessation / management of nicotine withdrawal for adults engaged with the Family Wellbeing Service
Mycophenolate Mofetil (MMF)
 SCG: For Transplant patients The following guidelines are designed to provide information relating to mycophenolate mofetil and to outline the responsibilities of the primary and secondary care teams in
SCG: For Transplant patients The following guidelines are designed to provide information relating to mycophenolate mofetil and to outline the responsibilities of the primary and secondary care teams in
HYPONATRAEMIA: NUH GUIDELINE FOR INITIAL ASSESSMENT AND MANAGEMENT.
 HYPONATRAEMIA: NUH GUIDELINE FOR INITIAL ASSESSMENT AND MANAGEMENT. HYPONATRAEMIA: SODIUM < 130 MMOL/L SIGNIFICANT. Symptoms/signs usually only occur when sodium < 125 mmol/l. Acute hyponatraemia is less
HYPONATRAEMIA: NUH GUIDELINE FOR INITIAL ASSESSMENT AND MANAGEMENT. HYPONATRAEMIA: SODIUM < 130 MMOL/L SIGNIFICANT. Symptoms/signs usually only occur when sodium < 125 mmol/l. Acute hyponatraemia is less
EFFECTIVE SHARED CARE AGREEMENT. Dronedarone - For the treatment and management of atrial fibrillation
 EFFECTIVE SHARED CARE AGREEMENT Dronedarone - For the treatment and management of atrial fibrillation Specialist details Patient identifier Name INTRODUCTION This shared care agreement outlines how the
EFFECTIVE SHARED CARE AGREEMENT Dronedarone - For the treatment and management of atrial fibrillation Specialist details Patient identifier Name INTRODUCTION This shared care agreement outlines how the
New Zealand Data Sheet. Arrow-Bendrofluazide Bendroflumethiazide (also known as Bendrofluazide) Tablets 2.5mg and 5mg
 New Zealand Data Sheet Arrow-Bendrofluazide Bendroflumethiazide (also known as Bendrofluazide) Tablets 2.5mg and 5mg Description Arrow-Bendrofluazide Tablets contain 2.5 and 5 mg of the active ingredient
New Zealand Data Sheet Arrow-Bendrofluazide Bendroflumethiazide (also known as Bendrofluazide) Tablets 2.5mg and 5mg Description Arrow-Bendrofluazide Tablets contain 2.5 and 5 mg of the active ingredient
Ivabradine for treating chronic heart failure: NICE technology appraisal guidance 267 Information for East Kent GPs revised July 2015
 Ivabradine for treating chronic heart failure: NICE technology appraisal guidance 267 Information for East Kent GPs revised July 2015 In November 2012 NICE recommended that: 1.1 Ivabradine is recommended
Ivabradine for treating chronic heart failure: NICE technology appraisal guidance 267 Information for East Kent GPs revised July 2015 In November 2012 NICE recommended that: 1.1 Ivabradine is recommended
Prescribing Guidelines Prescribing arrangement for the management of patients transferring from Secondary Care to Primary Care
 Berkshire West Integrated Care System Representing Berkshire West Clinical Commisioning Group Royal Berkshire NHS Foundation Trust Berkshire Healthcare NHS Foundation Trust Berkshire West Primary Care
Berkshire West Integrated Care System Representing Berkshire West Clinical Commisioning Group Royal Berkshire NHS Foundation Trust Berkshire Healthcare NHS Foundation Trust Berkshire West Primary Care
HYDROXYCARBAMIDE for Haematological conditions (Adults)
 Shared Care Guideline DRUG: Introduction: Contraindications & Warnings: HYDROXYCARBAMIDE for Haematological conditions (Adults) Indication: Hydroxycarbamide is an established treatment in haematological
Shared Care Guideline DRUG: Introduction: Contraindications & Warnings: HYDROXYCARBAMIDE for Haematological conditions (Adults) Indication: Hydroxycarbamide is an established treatment in haematological
NPSA SAFER LITHIUM THERAPY GUIDELINES FOR NHS LANARKSHIRE
 NPSA SAFER LITHIUM THERAPY GUIDELINES FOR NHS LANARKSHIRE Implementation Date Spring 2013 Review Date Spring 2016 NPSA Safer Lithium Therapy Guidelines for NHS Lanarkshire BACKGROUND On 1 st December 2009
NPSA SAFER LITHIUM THERAPY GUIDELINES FOR NHS LANARKSHIRE Implementation Date Spring 2013 Review Date Spring 2016 NPSA Safer Lithium Therapy Guidelines for NHS Lanarkshire BACKGROUND On 1 st December 2009
EFFECTIVE SHARED CARE AGREEMENT (ESCA)
 WORKING IN PARTNERSHIP WITH EFFECTIVE SHARED CARE AGREEMENT (ESCA) DRUG NAME: ACETYLCHOLINESTERASE-INHIBITORS AND MEMANTINE INDICATION/S COVERED: Dementia in Alzheimers Disease Coastal West Sussex Traffic
WORKING IN PARTNERSHIP WITH EFFECTIVE SHARED CARE AGREEMENT (ESCA) DRUG NAME: ACETYLCHOLINESTERASE-INHIBITORS AND MEMANTINE INDICATION/S COVERED: Dementia in Alzheimers Disease Coastal West Sussex Traffic
PACKAGE LEAFLET: Information for the patient FUROSEMID Tablets - 40 mg Solution for injection - 20 mg / 2 ml (Furosemide)
 PACKAGE LEAFLET: Information for the patient FUROSEMID Tablets - 40 mg Solution for injection - 20 mg / 2 ml (Furosemide) Read this leaflet carefully before you start taking this medicine. - Keep this
PACKAGE LEAFLET: Information for the patient FUROSEMID Tablets - 40 mg Solution for injection - 20 mg / 2 ml (Furosemide) Read this leaflet carefully before you start taking this medicine. - Keep this
Composition: Each Tablet contains. Pharmacokinetic properties:
 Composition: Each Tablet contains Torsemide 5/10/20/40/100mg Pharmacokinetic properties: Torsemide is well absorbed from the gastrointestinal tract. Peak serum concentrations are achieved within 1 hour
Composition: Each Tablet contains Torsemide 5/10/20/40/100mg Pharmacokinetic properties: Torsemide is well absorbed from the gastrointestinal tract. Peak serum concentrations are achieved within 1 hour
Amiodarone. Traffic light classification- Amber 2 Information sheet for Primary Care Prescribers
 Amiodarone Traffic light classification- Amber 2 Information sheet for Primary Care Prescribers Licensed Indications Tachyarrhythmias associated with Wolff-Parkinson-White syndrome All types of tachyarrhythmias
Amiodarone Traffic light classification- Amber 2 Information sheet for Primary Care Prescribers Licensed Indications Tachyarrhythmias associated with Wolff-Parkinson-White syndrome All types of tachyarrhythmias
Ciclosporin 25mg, 50mg, 100mg capsules Ciclosporin oral solution 100mg/ml
 Shared Care Protocol Ciclosporin for the treatment of rheumatoid arthritis Name of drug, form and strength Background Ciclosporin 25mg, 50mg, 100mg capsules Ciclosporin oral solution 100mg/ml Ciclosporin
Shared Care Protocol Ciclosporin for the treatment of rheumatoid arthritis Name of drug, form and strength Background Ciclosporin 25mg, 50mg, 100mg capsules Ciclosporin oral solution 100mg/ml Ciclosporin
Document Details Shared Care Agreement Lisdexamfetamine Trust Ref No Local Ref (optional) Main points the document covers
 Document Details Title Shared Care Agreement Lisdexamfetamine Trust Ref No 1989-33708 Local Ref (optional) Main points the document covers The responsibilities of each partner entering into the shared
Document Details Title Shared Care Agreement Lisdexamfetamine Trust Ref No 1989-33708 Local Ref (optional) Main points the document covers The responsibilities of each partner entering into the shared
Patient Group Direction for SALBUTAMOL INHALER (Version 02) Valid From 1 October September 2019
 Version Control This PGD has been agreed by the following organisations FCMS PDS Medical Doncaster CCG Lancashire CCGs including East Lancashire, Fylde and Wyre and North Lancashire CCGs Change history
Version Control This PGD has been agreed by the following organisations FCMS PDS Medical Doncaster CCG Lancashire CCGs including East Lancashire, Fylde and Wyre and North Lancashire CCGs Change history
EFFECTIVE SHARE CARE AGREEMENT. For the specialist use of LIOTHYRONINE for patients registered with a Dudley GP.
 Specialist details Patient identifier Name Tel: EFFECTIVE SHARE CARE AGREEMENT For the specialist use of LIOTHYRONINE for patients registered with a Dudley GP. The aim of an Effective Shared Care Agreement
Specialist details Patient identifier Name Tel: EFFECTIVE SHARE CARE AGREEMENT For the specialist use of LIOTHYRONINE for patients registered with a Dudley GP. The aim of an Effective Shared Care Agreement
GREATER MANCHESTER INTERFACE PRESCRIBING GROUP
 GREATER MANCHESTER INTERFACE PRESCRIBING GROUP On behalf of the GREATER MANCHESTER MEDICINES MANAGEMENT GROUP SHARED CARE GUIDELINE FOR THE PRESCRIBING OF SELECTIVE SEROTONIN REUPTAKE INHIBITORS (SSRIs)
GREATER MANCHESTER INTERFACE PRESCRIBING GROUP On behalf of the GREATER MANCHESTER MEDICINES MANAGEMENT GROUP SHARED CARE GUIDELINE FOR THE PRESCRIBING OF SELECTIVE SEROTONIN REUPTAKE INHIBITORS (SSRIs)
Patient Group Direction for PROCHLORPERAZINE (Version 02) Valid From 1 October September 2019
 Version Control This PGD has been agreed by the following organisations FCMS PDS Medical Doncaster CCG Lancashire CCGs including East Lancashire, Fylde and Wyre and North Lancashire CCGs Change history
Version Control This PGD has been agreed by the following organisations FCMS PDS Medical Doncaster CCG Lancashire CCGs including East Lancashire, Fylde and Wyre and North Lancashire CCGs Change history
EFFECTIVE SHARE CARE AGREEMENT
 Specialist details Patient identifier Name: Tel: EFFECTIVE SHARE CARE AGREEMENT For the specialist use of Erythropoietin Stimulating Agent (ESA) Therapy (formerly known as EPO) for the correction of Anaemia
Specialist details Patient identifier Name: Tel: EFFECTIVE SHARE CARE AGREEMENT For the specialist use of Erythropoietin Stimulating Agent (ESA) Therapy (formerly known as EPO) for the correction of Anaemia
PRODUCT INFORMATION. HYGROTON (Chlorthalidone Tablets 25 mg)
 NAME OF THE MEDICINE PRODUCT INFORMATION HYGROTON (Chlorthalidone Tablets 25 mg) Chlorthalidone 2-chloro-5-(1-hydroxy-3-oxo-1,2-dihydroisoindol-1-yl)- benzenesulfonamide Molecular weight - 338.767 CAS
NAME OF THE MEDICINE PRODUCT INFORMATION HYGROTON (Chlorthalidone Tablets 25 mg) Chlorthalidone 2-chloro-5-(1-hydroxy-3-oxo-1,2-dihydroisoindol-1-yl)- benzenesulfonamide Molecular weight - 338.767 CAS
Effective Shared Care Agreement for the treatment of severe motor complications in people with Parkinson Disease with apomorphine (APO-go )
 Effective Shared Care Agreement for the treatment of severe motor complications in people with Parkinson Disease with apomorphine (APO-go ) This shared care agreement outlines the ways in which the responsibilities
Effective Shared Care Agreement for the treatment of severe motor complications in people with Parkinson Disease with apomorphine (APO-go ) This shared care agreement outlines the ways in which the responsibilities
Cardiff and Vale (C&V) UHB Corporate Medicines Management Group Shared Care Committee SHARED CARE
 Cardiff and Vale (C&V) UHB Corporate Medicines Management Group Shared Care Committee SHARED CARE Drug: RILUZOLE Protocol number: CV 39 Indication: AMYOTROPHIC LATERAL SCLEROSIS (ALS) General Guidance
Cardiff and Vale (C&V) UHB Corporate Medicines Management Group Shared Care Committee SHARED CARE Drug: RILUZOLE Protocol number: CV 39 Indication: AMYOTROPHIC LATERAL SCLEROSIS (ALS) General Guidance
Patient Group Direction for the supply and/or administration of Ibuprofen 400mg tablets to patients attending NHS Borders services
 Patient Group Direction for the supply and/or administration of Ibuprofen 400mg tablets to patients attending NHS Borders services This document authorises the supply and/or administration of Ibuprofen
Patient Group Direction for the supply and/or administration of Ibuprofen 400mg tablets to patients attending NHS Borders services This document authorises the supply and/or administration of Ibuprofen
Primary Care Prescribing Protocol to Support the Diagnosis and Management of People with Dementia
 Primary Care Prescribing Protocol to Support the Diagnosis and Management of People with Dementia This prescribing guideline provides the necessary information and guidance to support clinicians in the
Primary Care Prescribing Protocol to Support the Diagnosis and Management of People with Dementia This prescribing guideline provides the necessary information and guidance to support clinicians in the
HEART FAILURE SUMMARY. and is associated with significant morbidity and mortality. the cornerstone of heart failure treatment.
 HEART FAILURE SUMMARY + Heart Failure is a condition affecting a large number of Irish people and is associated with significant morbidity and mortality. + ACE inhibitors, in combination with diuretics,
HEART FAILURE SUMMARY + Heart Failure is a condition affecting a large number of Irish people and is associated with significant morbidity and mortality. + ACE inhibitors, in combination with diuretics,
Azathioprine Shared Care Guideline for GPs
 Indication: Azathioprine Shared Care Guideline for GPs Active rheumatoid arthritis and other types of inflammatory arthritis, systemic lupus erythematosus, dermatomyositis and polymyositis, vasculitis
Indication: Azathioprine Shared Care Guideline for GPs Active rheumatoid arthritis and other types of inflammatory arthritis, systemic lupus erythematosus, dermatomyositis and polymyositis, vasculitis
patient group direction
 NAPROXEN v01 1/10 NAPROXEN PGD Details Version 1.0 Legal category Staff grades Approved by POM Paramedic (Non-ECP) Nurse (Non-ECP) Emergency Care Practitioner (Paramedic) Emergency Care Practitioner (Nurse)
NAPROXEN v01 1/10 NAPROXEN PGD Details Version 1.0 Legal category Staff grades Approved by POM Paramedic (Non-ECP) Nurse (Non-ECP) Emergency Care Practitioner (Paramedic) Emergency Care Practitioner (Nurse)
DRUG: Introduction. `Shared Care Guidelines. They can also be used long term for chronic pulmonary Pseudomonas aeruginosa infection.
 `Shared Care Guidelines DRUG: Introduction Inhaled antibiotics (Adults and Paediatrics) in Cystic Fibrosis. EXISTING ESTABLISHED PATIENTS ONLY Colistimethate (Colomycin, Promixin ) Tobramycin (Tobi Bramitob
`Shared Care Guidelines DRUG: Introduction Inhaled antibiotics (Adults and Paediatrics) in Cystic Fibrosis. EXISTING ESTABLISHED PATIENTS ONLY Colistimethate (Colomycin, Promixin ) Tobramycin (Tobi Bramitob
Name of Shared Care Agreement: AZATHIOPRINE/6-MERCAPTOPURINE: Oral immunomodulating drugs for inflammatory bowel disease. Reference number: 01/2008
 Name of Shared Care Agreement: AZATHIOPRINE/6-MERCAPTOPURINE: Oral immunomodulating drugs for inflammatory bowel disease. Reference number: 01/2008 Shared care agreement has been developed appropriately
Name of Shared Care Agreement: AZATHIOPRINE/6-MERCAPTOPURINE: Oral immunomodulating drugs for inflammatory bowel disease. Reference number: 01/2008 Shared care agreement has been developed appropriately
Shared Care Guidelines. In partnership with Acute Trust & PCTs. SIROLIMUS In renal transplant. August 2016
 Shared Care Guidelines In partnership with Acute Trust & PCTs SIROLIMUS In renal transplant August 2016 Page 0 of 12 Table of Contents Introduction 2 Licensing 2 Referral 2 Initiation and maintenance of
Shared Care Guidelines In partnership with Acute Trust & PCTs SIROLIMUS In renal transplant August 2016 Page 0 of 12 Table of Contents Introduction 2 Licensing 2 Referral 2 Initiation and maintenance of
Swindon Diabetes Guidelines: Management of Chronic Kidney Disease Associated with Diabetes Mellitus
 Swindon Diabetes Guidelines: Management of Chronic Kidney Disease Associated with Diabetes Mellitus 1 Contents Executive Summary... 3 How to Screen for Diabetic Nephropathy... 4 What to Measure... 4 Frequency
Swindon Diabetes Guidelines: Management of Chronic Kidney Disease Associated with Diabetes Mellitus 1 Contents Executive Summary... 3 How to Screen for Diabetic Nephropathy... 4 What to Measure... 4 Frequency
EFFECTIVE SHARED CARE AGREEMENT (ESCA)
 EFFECTIVE SHARED CARE AGREEMENT (ESCA) DRUG NAME: HYDROXYCHLOROQUINE INDICATION/S COVERED: active rheumatoid arthritis, juvenile idiopathic arthritis, discoid and systemic lupus erythematosus Coastal West
EFFECTIVE SHARED CARE AGREEMENT (ESCA) DRUG NAME: HYDROXYCHLOROQUINE INDICATION/S COVERED: active rheumatoid arthritis, juvenile idiopathic arthritis, discoid and systemic lupus erythematosus Coastal West
Essential Shared Care Agreement Naltrexone
 In partnership with as part of Please complete the following details: Patient s name, address, date of birth Consultant s contact details (p.3) And send One copy to: 1. The patient s GP 2. Put one copy
In partnership with as part of Please complete the following details: Patient s name, address, date of birth Consultant s contact details (p.3) And send One copy to: 1. The patient s GP 2. Put one copy
Title of Guideline (must include the word Guideline Guideline for the Treatment of Hypokalaemia in Adults
 Title of Guideline (must include the word Guideline Guideline for the Treatment of (not protocol, policy, procedure etc) Hypokalaemia in Adults Contact Name and Job Title (author) Emily Snow, Pharmacist
Title of Guideline (must include the word Guideline Guideline for the Treatment of (not protocol, policy, procedure etc) Hypokalaemia in Adults Contact Name and Job Title (author) Emily Snow, Pharmacist
PHARMACOLOGY AND PHARMACOKINETICS
 DRUG GUIDELINE Insulin, human neutral (Actrapid ) Intravenous Infusion for SCOPE (Area): FOR USE IN: Critical Care Unit, Emergency Department and Operating Suite EXCLUSIONS: Paediatrics (seek Paediatrician
DRUG GUIDELINE Insulin, human neutral (Actrapid ) Intravenous Infusion for SCOPE (Area): FOR USE IN: Critical Care Unit, Emergency Department and Operating Suite EXCLUSIONS: Paediatrics (seek Paediatrician
SHARED CARE PRESCRIBING GUIDELINE
 SHARED CARE PRESCRIBING GUIDELINE RILUZOLE for the Treatment of MOTOR NEURONE DISEASE NHS Surrey s Medicines Management Committee classification: Amber N.B. The eligibility criteria included here apply
SHARED CARE PRESCRIBING GUIDELINE RILUZOLE for the Treatment of MOTOR NEURONE DISEASE NHS Surrey s Medicines Management Committee classification: Amber N.B. The eligibility criteria included here apply
Denosumab (Prolia 60 mg) Effective Shared Care Agreement For the treatment of Osteoporosis. Date: Date:
 Denosumab (Prolia 60 mg) Effective Shared Care Agreement For the treatment of Osteoporosis Section 1: Shared care arrangements and responsibilities Section 1.1 Agreement for transfer of prescribing to
Denosumab (Prolia 60 mg) Effective Shared Care Agreement For the treatment of Osteoporosis Section 1: Shared care arrangements and responsibilities Section 1.1 Agreement for transfer of prescribing to
SHARED CARE PROTOCOL FOR THE PRESCRIBING AND MONITORING OF MEDICINES FOR ATTENTION-DEFICIT HYPERACTIVITY DISORDER (ADHD)
 SHARED CARE PROTOCOL FOR THE PRESCRIBING AND MONITORING OF MEDICINES FOR ATTENTION-DEFICIT HYPERACTIVITY DISORDER (ADHD) 1. Introduction This protocol describes how patients prescribed medicines for ADHD
SHARED CARE PROTOCOL FOR THE PRESCRIBING AND MONITORING OF MEDICINES FOR ATTENTION-DEFICIT HYPERACTIVITY DISORDER (ADHD) 1. Introduction This protocol describes how patients prescribed medicines for ADHD
Elements for a public summary. Overview of disease epidemiology
 VI.2 Elements for a public summary VI.2.1 Overview of disease epidemiology Bendroflumethiazide/ potassium chloride contains thiazide diuretic bendroflumethiazide that is used in therapy of oedema, arterial
VI.2 Elements for a public summary VI.2.1 Overview of disease epidemiology Bendroflumethiazide/ potassium chloride contains thiazide diuretic bendroflumethiazide that is used in therapy of oedema, arterial
patient group direction
 ASPIRIN v01 1/8 ASPIRIN PGD Details Version 1.0 Legal category P Staff grades Approved by Paramedic (Non-ECP) Nurse (Non-ECP) Emergency Care Practitioner (Paramedic) Emergency Care Practitioner (Nurse)
ASPIRIN v01 1/8 ASPIRIN PGD Details Version 1.0 Legal category P Staff grades Approved by Paramedic (Non-ECP) Nurse (Non-ECP) Emergency Care Practitioner (Paramedic) Emergency Care Practitioner (Nurse)
Clinical guideline for the introduction of Sacubitril Valsartan in primary and secondary care in Cornwall
 Clinical guideline for the introduction of Sacubitril Valsartan in primary and secondary care in Cornwall Page 1 of 15 Summary Patient clinically assessed and reviewed by a Consultant Cardiologist, Cardiology
Clinical guideline for the introduction of Sacubitril Valsartan in primary and secondary care in Cornwall Page 1 of 15 Summary Patient clinically assessed and reviewed by a Consultant Cardiologist, Cardiology
SHARED CARE GUIDELINE FOR THE MANAGEMENT OF PATIENTS ON NALTREXONE FOR OPIOID DEPENDENCE
 SHARED CARE GUIDELINE FOR THE MANAGEMENT OF PATIENTS ON NALTREXONE FOR OPIOID DEPENDENCE INDICATION Naltrexone is a pure opiate antagonist licensed as an adjunctive prophylactic therapy in the maintenance
SHARED CARE GUIDELINE FOR THE MANAGEMENT OF PATIENTS ON NALTREXONE FOR OPIOID DEPENDENCE INDICATION Naltrexone is a pure opiate antagonist licensed as an adjunctive prophylactic therapy in the maintenance
