PLATINUM Diversity: Outcomes with the Promus PREMIER TM Stent in Women and Minorities
|
|
|
- Loreen Baldwin
- 5 years ago
- Views:
Transcription
1 PLATINUM Diversity: Outcomes with the Promus PREMIER TM Stent in Women and Minorities
2 Long Term Outcomes After PCI with PES in Black Versus White Patients Black vs. White: Risk of MI and Stent Thrombosis Batchelor et al. J Interv Cardiol 2012
3 Disparities in Clinical Research Mirror those in Clinical Practice Women comprise 30% of PCI procedures and PCI clinical trial enrollment. Men Women TAXUS-IV 72% 28% 66% SPIRIT III 69% 31% 34% SIRIUS ENDEAVOR 71% 77% 29% 23% PCI Procedures 2009 Patients: N=596,000 National Hospital Discharge Survey/National Center for Health Statistics, 2009 Estimates are based on a sample of inpatient records from short-stay hospitals in the United States.. Heart Disease and Stroke Statistics-2012 Update. Circulation. 0% 20% 40% 60% 80% 100% Gender representation in PCI clinical trials Einstein et al Year Follow-Up From the ENDEAVOR II Trial. JACC: Cardiovascular Interventions, 2(12): Holmes et al Analysis of 1-Year Clinical Outcomes in the SIRIUS Trial. Circulation, 109: Stone et. al Everolimus-and Paclitaxel-Eluting Stents. Circulation, 119: Lansky et al Gender Differences After Paclitaxel-Eluting Stents. JACC, 45(8): Morice, MC XIENCE V SPIRIT WOMEN clinical trial: characterization of the female population undergoing stent implantation. Women s Health, 4(5):
4 Current Clinical Enrollees do not Reflect the US Population People of color are projected to outnumber U.S. white population by U.S. population years old, by race/ethnicity: July and projections to 2050 Sources: U.S. Bureau of the Census U.S. Census Bureau, 2009 National Projections supplement to the 2008 National Projections, August 14, 2008 Other Pacific AI/AN Asian Latino Black White Overall Male Female Registry ION and LIBERTE PMS: Pooled Patient Demographics Composition (%) US N=5305
5 Key Environmental Drivers of the Need for Increased Diversity in Clinical Trials SCIENTIFIC Testing the safety and effectiveness of new medicines should accurately reflects the changing demographic of the patient population that will eventually take them REGULATORY Both domestically and internationally, Regulators and other stakeholders are demanding more data from diverse populations ETHICAL Clinical trials in which minorities are underrepresented may not adequately serve the health needs of these communities COMMERCIAL Delays in recruitment cost billions and keep novel therapies away from the patients that will benefit from them Clinical Trial Engagement Network National Minority Quality Forum, 2014
6 PLATINUM Diversity Study Design Study Objective: To compile acute procedural performance and clinical outcomes data for the Promus PREMIER EES in understudied/underserved patient populations including women and minorities Enrollment: Primary Investigators: 1500 patients / Up to 65 US Sites Wayne Batchelor: Florida State College of Medicine, Tallahassee Research Institute, & Southern Medical Group, Tallahassee, FL Roxana Mehran: Icahn School of Medicine at Mount Sinai Hospital, New York, NY Observational Prospective Multicenter Open-label Single-arm 1 PREMIER Stent & one or more of the following: Female Black Hispanic/Latino American Indian or Alaskan Native Follow-up (telephone): 30 days 6 months 1 year Primary Endpoint: 12M Death/MI/TVR Demographic collection on socioeconomic status, level of education, insurance status, language, etc. Adherence to DAPT Aligned with Close The Gap educational initiative to address disparities in cardiovascular care for women and minorities
7 PLATINUM Diversity Study Design: Statistical Analysis Primary Statistical Hypothesis Patients combined from PE + Post Approval Study and PLATINUM Diversity: Female patients Minority patients (Black of African Heritage, Hispanic/Latino, American Indian or Alaska native) vs. Reference subset of Caucasian male patients from the PE+ PAS H 0 : P PE+ = P SG (Not Superior) H 1 : P PE+ P SG (Superior) Statistical Test Method Statistical Power Multivariate logistic regression model Adjusting for the baseline covariate imbalance Enrollment of patients from specific subgroups will be monitored /stopped as needed to ensure sufficient enrollment in other subgroups IF female patient cohort 12-month Death/MI/TVR rate of 10.1% 80% power to detect a significant difference between PE+ Caucasian male patients and the combined PE+ and PLATINUM Diversity female patients IF the combined minority patient cohort has a 12-month Death/MI/TVR rate of 10.6% 80% power to detect a significant difference between PE+ Caucasian male patients and the combined PE+ and PLATINUM Diversity minority patients
8 Thank you.
9 FDA Approved Product Labeling Promus PREMIER Stent System Caution: Federal Law (USA) restricts this device to sale by or on the order of a physician. Indications for Use: The Promus PREMIER Everolimus-Eluting Platinum Chromium Coronary Stent System is indicated for improving luminal diameter in patients with symptomatic heart disease or documented silent ischemia due to de novo lesions in native coronary arteries 2.25 mm to 4.00 mm in diameter in lesions 34 mm in length. Contraindications: Use of the Promus PREMIER Everolimus-Eluting Platinum Chromium Coronary Stent System is contraindicated in patients with known hypersensitivity to: 316L stainless steel or platinum everolimus or structurally-related compounds the polymers or their individual components (see Section , Primer Polymer and Drug Matrix Copolymer Carrier) Coronary Artery Stenting is contraindicated for use in: Patients judged to have a lesion that prevents complete inflation of an angioplasty balloon or proper placement of the stent or delivery device. Patients with uncorrected bleeding disorders or patients who cannot receive anticoagulation or antiplatelet aggregation therapy (see Section 6.2, Pre- and Post-Procedure Antiplatelet Regimen for more information). Warnings: To maintain sterility, the inner package should not be opened or damaged prior to use. The use of this product carries the risks associated with coronary artery stenting, including stent thrombosis, vascular complications, and/or bleeding events. This product should not be used in patients who are not likely to comply with recommended antiplatelet therapy. Precautions: General Precautions Only physicians who have received adequate training should perform implantation of the stent. Stent placement should only be performed at hospitals where emergency coronary artery bypass graft surgery can be readily performed. Subsequent stent blockage may require repeat dilatation of the arterial segment containing the stent. The long-term outcome following repeat dilatation of endothelialized stents is not well characterized. Consideration should be given to the risks and benefits of use in patients with history of severe reaction to contrast agents. Do not expose the delivery system to organic solvents such as alcohol or detergents. Care should be taken to control the position of the guide catheter tip during stent delivery, deployment and balloon withdrawal. Before withdrawing the stent delivery system, visually confirm complete balloon deflation by fluoroscopy to avoid guiding catheter movement into the vessel and subsequent arterial damage. Stent thrombosis is a low-frequency event that current drug-eluting stent (DES) clinical trials are not adequately powered to fully characterize. Stent thrombosis is frequently associated with myocardial infarction (MI) or death. In the clinical trials analyzed to date, differences in the incidence of stent thrombosis have not been associated with an increased risk of cardiac death, MI, or all-cause mortality. Additional data from longer-term follow-up of the PLATINUM clinical trials and analyses of stent thrombosis related to DES are expected and should be considered in making treatment decisions as data become available. When DES are used outside the specified Indications for Use, patient outcomes may differ from the results observed in the NG PROMUS and PLATINUM pivotal clinical trials. Compared to use within the specified Indications for Use, the use of DES in patients and lesions outside of the labeled indications may have an increased risk of adverse events, including stent thrombosis, stent embolization, MI or death. Orally-administered everolimus combined with cyclosporine is associated with increased serum cholesterol and triglyceride levels. The safety and effectiveness of the Promus PREMIER Stent have not been established in the cerebral, carotid, or peripheral vasculature or in the following patient populations: patients requiring multiple stents patients with prior brachytherapy of the target lesion patients in whom mechanical atherectomy are used in conjunction with the PROMUS Premier stent pediatric patients Patients with vessel thrombus at the lesion site. Patients with coronary artery reference vessel diameters <2.25 or >4.00 mm. Patients with coronary artery lesions longer than 34 mm or requiring more than one Promus PREMIER Stent. Patients with lesions located in the saphenous vein grafts, in the left main coronary artery, ostial lesions, or lesions located at a bifurcation. Patients with diffuse disease or poor flow distal to the identified lesions. Patients with tortuous vessels (>60 degrees) in the region of the obstruction or proximal to the lesion. Patients with a recent acute myocardial infarction where there is evidence of thrombus or poor flow. Patients with in-stent restenosis. Patients with moderate or severe calcification in the lesion or a chronic total occlusion. Patients with 3 vessel disease. 9.2 Potential Adverse Events Potential adverse events (in alphabetical order) which may be associated with the use of a coronary stent in native coronary arteries include but are not limited to: Abrupt stent closure Acute myocardial infarction Allergic reaction to anti-coagulant and/or antiplatelet therapy, contrast medium, or stent materials Angina Arrhythmias, including ventricular fibrillation and ventricular tachycardia Arteriovenous fistula Bleeding Cardiac tamponade Cardiogenic shock/pulmonary edema Coronary aneurysm Death Dissection Emboli, distal (air, tissue or thrombotic material or material from device(s) used in the procedure) Heart failure Hematoma Hemorrhage, which may require transfusion Hypotension/hypertension Infection, local or systemic Ischemia, myocardial Pain, access site Perforation or rupture of coronary artery Pericardial effusion Pseudoaneurysm, femoral Renal insufficiency or failure Respiratory failure Restenosis of stented segment Stent embolization or migration Stent deformation, collapse, or fracture Stent thrombosis/occlusion Stroke/cerebrovascular accident/transient ischemic attack Total occlusion of coronary artery Vessel spasm Vessel trauma requiring surgical repair or reintervention The following list includes the known risks of everolimus at the oral doses listed above. The amount of drug that circulates in the bloodstream following implantation of a Promus PREMIER Stent is several folds lower than that obtained with oral doses (1.5 mg to 20 mg/day, see Section 7.2, Pharmacokinetics). Abdominal pain (including upper abdominal pain) Anemia Angioedema Anorexia Asthenia Constipation Cough Delayed wound healing/fluid accumulation Diarrhea Dyslipidemia (including hyperlipidemia and hypercholesterolemia) Dysgeusia Dyspepsia Dyspnea Dysuria Dry skin Edema (peripheral) Epistaxis Fatigue Headache Hematuria Hyperglycemia (may include new onset of diabetes) Hyperkalemia Hyperlipidemia Hypertension Hypokalemia Hypomagnesemia Hypophosphatemia Increased serum creatinine Infections and serious infections: bacterial, viral, fungal, and protozoal infections (may include herpes virus infection, polyoma virus infection which may be associated with BK virus ssociated nephropathy, and/or other opportunistic infections) Insomnia Interaction with strong inhibitors and inducers of CYP3A4 Leukopenia Lymphoma and other malignancies (including skin cancer) Male infertility (azospermia and/or oligospermia) Mucosal inflammation (including oral ulceration and oral mucositis) Nausea Neutropenia Non-infectious pneumonitis Pain; extremity, incision site and procedural, back, chest, musculoskeletal Proteinuria Pruritus Pyrexia Rash Stomatitis Thrombocytopenia Thrombotic microangiopathy (TMA)/Thrombotic thrombocytopenic purpura (TTP)/Hemolytic uremic syndrome (HUS) Tremor Upper respiratory tract infection Urinary tract infection Vomiting Live vaccines should be avoided and close contact with those that have had live vaccines should be avoided. Fetal harm can occur when administered to a pregnant woman. There may be other potential adverse events that are unforeseen at this time. Prior to use, please reference the Instructions for Use at for more information on indications, contraindications, warnings, precautions, and adverse events.
Off-Label Use Expansion: The CTO Example
 Off-Label Use Expansion: The CTO Example Physician/Scientist Perspective Chuck Simonton MD, FACC, FSCAI Chief Medical Officer Divisional Vice President Abbott Vascular Santa Clara, CA TCT 2011 1 Disclosures
Off-Label Use Expansion: The CTO Example Physician/Scientist Perspective Chuck Simonton MD, FACC, FSCAI Chief Medical Officer Divisional Vice President Abbott Vascular Santa Clara, CA TCT 2011 1 Disclosures
Promus ELITE. Everolimus-Eluting Platinum Chromium Coronary Stent System. Patient Information Guide CV01
 Promus ELITE Everolimus-Eluting Platinum Chromium Coronary Stent System Patient Information Guide CV01 Promus ELITE Everolimus-Eluting Platinum Chromium Coronary Stent System PATIENT INFORMATION GUIDE
Promus ELITE Everolimus-Eluting Platinum Chromium Coronary Stent System Patient Information Guide CV01 Promus ELITE Everolimus-Eluting Platinum Chromium Coronary Stent System PATIENT INFORMATION GUIDE
REBEL. Platinum Chromium Coronary Stent System. Patient Information Guide
 REBEL Patient Information Guide REBEL PATIENT INFORMATION GUIDE You have recently had a REBEL bare metal stent implanted in the coronary arteries of your heart. The following information is important for
REBEL Patient Information Guide REBEL PATIENT INFORMATION GUIDE You have recently had a REBEL bare metal stent implanted in the coronary arteries of your heart. The following information is important for
Polymeric Bioresorbable Scaffolds: Are they Poised to Revolutionize Percutaneous Coronary Intervention?
 Polymeric Bioresorbable Scaffolds: Are they Poised to Revolutionize Percutaneous Coronary Intervention? Richard J. Rapoza, Divisional Vice President, R&D Product and Therapy Innovation 1 of 28 Outline
Polymeric Bioresorbable Scaffolds: Are they Poised to Revolutionize Percutaneous Coronary Intervention? Richard J. Rapoza, Divisional Vice President, R&D Product and Therapy Innovation 1 of 28 Outline
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service
 M AY. 6. 2011 10:37 A M F D A - C D R H - O D E - P M O N O. 4147 P. 1 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control
M AY. 6. 2011 10:37 A M F D A - C D R H - O D E - P M O N O. 4147 P. 1 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control
DURABLE. CONSISTENT. SAFE. IN.PACT Admiral Drug-Coated Balloon
 DURABLE. CONSISTENT. SAFE. Drug-Coated Balloon THE SCIENCE BEHIND THE OUTCOMES DCB has proven, long-term durable outcomes across multiple clinical trials, as well as across complex patient and lesion types.
DURABLE. CONSISTENT. SAFE. Drug-Coated Balloon THE SCIENCE BEHIND THE OUTCOMES DCB has proven, long-term durable outcomes across multiple clinical trials, as well as across complex patient and lesion types.
MICHAEL R. JAFF, DO MASSACHUSETTS, UNITED STATES. Medtronic Further. Together
 DRUG-COATED BALL0ON TREATMENT FOR PATIENTS WITH INTERMITTENT CLAUDICATION: INSIGHTS FROM THE IN.PACT GLOBAL FULL CLINICAL COHORT MICHAEL R. JAFF, DO MASSACHUSETTS, UNITED STATES Medtronic Further. Together
DRUG-COATED BALL0ON TREATMENT FOR PATIENTS WITH INTERMITTENT CLAUDICATION: INSIGHTS FROM THE IN.PACT GLOBAL FULL CLINICAL COHORT MICHAEL R. JAFF, DO MASSACHUSETTS, UNITED STATES Medtronic Further. Together
Sustained Release. Superior Results.
 ELUVIA Drug-Eluting Vascular Stent System Sustained Release. Superior Results. Superiority determined in Post Hoc Superiority Analysis. 12-Month Primary Patency rate of 86.8% in the Eluvia arm (n=309)
ELUVIA Drug-Eluting Vascular Stent System Sustained Release. Superior Results. Superiority determined in Post Hoc Superiority Analysis. 12-Month Primary Patency rate of 86.8% in the Eluvia arm (n=309)
The SplitWire Percutaneous Transluminal Angioplasty Scoring Device. Instructions for Use
 The SplitWire Percutaneous Transluminal Angioplasty Scoring Device Instructions for Use Contents Contains one (1) SplitWire device. Sterile. Sterilized with ethylene oxide gas. Radiopaque. For single use
The SplitWire Percutaneous Transluminal Angioplasty Scoring Device Instructions for Use Contents Contains one (1) SplitWire device. Sterile. Sterilized with ethylene oxide gas. Radiopaque. For single use
TCTAP Upendra Kaul MD,DM,FACC,FSCAI,FAMS,FCSI
 Indian TUXEDO Trial In Medically Treated Diabetics Upendra Kaul MD,DM,FACC,FSCAI,FAMS,FCSI Executive Director and Dean Escorts Heart Institute & Medical Research Center and Fortis Hospitals, New Delhi
Indian TUXEDO Trial In Medically Treated Diabetics Upendra Kaul MD,DM,FACC,FSCAI,FAMS,FCSI Executive Director and Dean Escorts Heart Institute & Medical Research Center and Fortis Hospitals, New Delhi
PROMUS Element Experience In AMC
 Promus Element Luncheon Symposium: PROMUS Element Experience In AMC Jung-Min Ahn, MD. University of Ulsan College of Medicine, Heart Institute, Asan Medical Center, Seoul, Korea PROMUS Element Clinical
Promus Element Luncheon Symposium: PROMUS Element Experience In AMC Jung-Min Ahn, MD. University of Ulsan College of Medicine, Heart Institute, Asan Medical Center, Seoul, Korea PROMUS Element Clinical
Talent Abdominal Stent Graft
 Talent Abdominal with THE Xcelerant Hydro Delivery System Expanding the Indications for EVAR Treat More Patients Short Necks The Talent Abdominal is the only FDA-approved device for proximal aortic neck
Talent Abdominal with THE Xcelerant Hydro Delivery System Expanding the Indications for EVAR Treat More Patients Short Necks The Talent Abdominal is the only FDA-approved device for proximal aortic neck
Patient Information. Peripheral Arterial Disease and the Lutonix 035 Balloon. Advancing Lives and the Delivery of Health Care
 Patient Information Peripheral Arterial Disease and the Lutonix 035 Balloon Advancing Lives and the Delivery of Health Care Contents Peripheral Arterial Disease (PAD) Peripheral Arterial Disease (PAD)
Patient Information Peripheral Arterial Disease and the Lutonix 035 Balloon Advancing Lives and the Delivery of Health Care Contents Peripheral Arterial Disease (PAD) Peripheral Arterial Disease (PAD)
1 Comparison of Warfarin and Aspirin for Symptomatic Intracranial Arterial Stenosis. N Engl J Med 352;13, March 31, 2005
 The risk of ischemic stroke in patients with Intracranial Atherosclerotic Disease (ICAD) ranges from 7 to 24%. 1,2 Developed specifically for the treatment of ICAD, the Wingspan Stent System and Gateway
The risk of ischemic stroke in patients with Intracranial Atherosclerotic Disease (ICAD) ranges from 7 to 24%. 1,2 Developed specifically for the treatment of ICAD, the Wingspan Stent System and Gateway
DISRUPT PAD. (( Data Summary )) DISRUPT PAD Data Summary SPL Rev. B 2016 Shockwave Medical Inc. All rights reserved.
 DISRUPT PAD (( Data Summary )) DISRUPT PAD Data Summary SPL 60971 Rev. B 1 Summary of the key findings from the DISRUPT PAD Study 99% of femoropopliteal lesions treated were moderately or severely calcified.
DISRUPT PAD (( Data Summary )) DISRUPT PAD Data Summary SPL 60971 Rev. B 1 Summary of the key findings from the DISRUPT PAD Study 99% of femoropopliteal lesions treated were moderately or severely calcified.
1 Description. 2 Indications. 3 Warnings ASPIRATION CATHETER
 Page 1 of 5 ASPIRATION CATHETER Carefully read all instructions prior to use, observe all warnings and precautions noted throughout these instructions. Failure to do so may result in complications. STERILE.
Page 1 of 5 ASPIRATION CATHETER Carefully read all instructions prior to use, observe all warnings and precautions noted throughout these instructions. Failure to do so may result in complications. STERILE.
Supplementary Online Content
 Supplementary Online Content Valle JA, Tamez H, Abbott JD, et al. Contemporary use and trends in unprotected left main coronary artery percutaneous coronary intervention in the United States: an analysis
Supplementary Online Content Valle JA, Tamez H, Abbott JD, et al. Contemporary use and trends in unprotected left main coronary artery percutaneous coronary intervention in the United States: an analysis
Nilotinib AEs (adverse events) in CML population:
 Nilotinib AEs (adverse events) in CML population: The percentages below were taken from a randomized trial of nilotinib 300mg BID in newly diagnosed Ph+ CML patients (N=279) taken from the Tasigna 2017
Nilotinib AEs (adverse events) in CML population: The percentages below were taken from a randomized trial of nilotinib 300mg BID in newly diagnosed Ph+ CML patients (N=279) taken from the Tasigna 2017
NON-COMPLIANT PTCA RAPID EXCHANGE DILATATION CATHETER
 Page 1 of 5 NON-COMPLIANT PTCA RAPID EXCHANGE DILATATION CATHETER STERILE. SINGLE USE ONLY. Sterilized with ethylene oxide gas. Non pyrogenic. Do not resterilize. Do not use opened or damaged packages.
Page 1 of 5 NON-COMPLIANT PTCA RAPID EXCHANGE DILATATION CATHETER STERILE. SINGLE USE ONLY. Sterilized with ethylene oxide gas. Non pyrogenic. Do not resterilize. Do not use opened or damaged packages.
Stellarex Drug-coated 0.035" Angioplasty Balloon. Featuring EnduraCoat Technology. The Clear DCB Choice
 Stellarex Drug-coated 0.035" Angioplasty Balloon Featuring EnduraCoat Technology The Clear DCB Choice Top-tier outcomes with the lowest therapeutic drug dose in all patients common to complex 89.0 % patency
Stellarex Drug-coated 0.035" Angioplasty Balloon Featuring EnduraCoat Technology The Clear DCB Choice Top-tier outcomes with the lowest therapeutic drug dose in all patients common to complex 89.0 % patency
Assurant Cobalt Iliac BALLOON EXPANDABLE STENT SYSTEM
 Assurant Cobalt Iliac BALLOON EXPANDABLE STENT SYSTEM Innovating for life. CONFORMABILIT Y 6 F S H E AT H C O M PAT I B I L I T Y THE ASSURANT COBALT ILIAC STENT, WITH ITS UNIQUE COBALT CHROMIUM MODULAR
Assurant Cobalt Iliac BALLOON EXPANDABLE STENT SYSTEM Innovating for life. CONFORMABILIT Y 6 F S H E AT H C O M PAT I B I L I T Y THE ASSURANT COBALT ILIAC STENT, WITH ITS UNIQUE COBALT CHROMIUM MODULAR
Effect of Intravascular Ultrasound- Guided vs. Angiography-Guided Everolimus-Eluting Stent Implantation: the IVUS-XPL Randomized Clinical Trial
 Effect of Intravascular Ultrasound- Guided vs. Angiography-Guided Everolimus-Eluting Stent Implantation: the IVUS-XPL Randomized Clinical Trial Myeong-Ki Hong, MD. PhD on behalf of the IVUS-XPL trial investigators
Effect of Intravascular Ultrasound- Guided vs. Angiography-Guided Everolimus-Eluting Stent Implantation: the IVUS-XPL Randomized Clinical Trial Myeong-Ki Hong, MD. PhD on behalf of the IVUS-XPL trial investigators
UNDERSTANDING TREATMENT OPTIONS FOR HEART DISEASE. Visit
 Visit www.absorbstent.com UNDERSTANDING TREATMENT OPTIONS FOR HEART DISEASE UNDERSTANDING YOUR BLOCKED ARTERIES CORONARY ARTERY DISEASE MAY BE AFFECTING YOU OR SOMEONE YOU LOVE. Your heart needs a constant
Visit www.absorbstent.com UNDERSTANDING TREATMENT OPTIONS FOR HEART DISEASE UNDERSTANDING YOUR BLOCKED ARTERIES CORONARY ARTERY DISEASE MAY BE AFFECTING YOU OR SOMEONE YOU LOVE. Your heart needs a constant
RECOMMENDED INSTRUCTIONS FOR USE
 Rapid Exchange PTCA Dilatation Catheter RECOMMENDED INSTRUCTIONS FOR USE Available in diameters 1.25mm to 4.5mm and in lengths 09mm to 40mm Caution: This device should be used only by physicians trained
Rapid Exchange PTCA Dilatation Catheter RECOMMENDED INSTRUCTIONS FOR USE Available in diameters 1.25mm to 4.5mm and in lengths 09mm to 40mm Caution: This device should be used only by physicians trained
TCT mdbuyline.com Clinical Trial Results Summary
 TCT 2012 Clinical Trial Results Summary FAME2 Trial: FFR (fractional flow reserve) guided PCI in all target lesions Patients with significant ischemia, randomized 1:1 Control arm: not hemodynamically significant
TCT 2012 Clinical Trial Results Summary FAME2 Trial: FFR (fractional flow reserve) guided PCI in all target lesions Patients with significant ischemia, randomized 1:1 Control arm: not hemodynamically significant
PERFORMANCE YOU CAN TRUST. EverFlex Self-expanding Peripheral Stent with Entrust Delivery System
 PERFORMANCE YOU CAN TRUST EverFlex Self-expanding Peripheral Stent with Entrust Delivery System The Entrust Delivery System is designed to provide improved patient outcomes and procedural efficiency. The
PERFORMANCE YOU CAN TRUST EverFlex Self-expanding Peripheral Stent with Entrust Delivery System The Entrust Delivery System is designed to provide improved patient outcomes and procedural efficiency. The
1. Whether the risks of stent thrombosis (ST) and major adverse cardiovascular and cerebrovascular events (MACCE) differ from BMS and DES
 1 Comparison of Ischemic and Bleeding Events After Drug- Eluting Stents or Bare Metal Stents in Subjects Receiving Dual Antiplatelet Therapy: Results from the Randomized Dual Antiplatelet Therapy (DAPT)
1 Comparison of Ischemic and Bleeding Events After Drug- Eluting Stents or Bare Metal Stents in Subjects Receiving Dual Antiplatelet Therapy: Results from the Randomized Dual Antiplatelet Therapy (DAPT)
LUTONIX DCB in BTK Update on the BTK clinical program & single center experience
 LUTONIX DCB in BTK Update on the BTK clinical program & single center experience Prof. Dr. med. Dierk Scheinert Department of Interventional Angiology University Hospital Leipzig Disclaimer 1. The information
LUTONIX DCB in BTK Update on the BTK clinical program & single center experience Prof. Dr. med. Dierk Scheinert Department of Interventional Angiology University Hospital Leipzig Disclaimer 1. The information
D. D. TSIKADERIS MD, FESC SAINT LUKES THESSALONIKI
 D. D. TSIKADERIS MD, FESC SAINT LUKES THESSALONIKI EPIDEMIOLOGY DIAGNOSIS RESPONSE TO THERAPY PROGNOSIS UNDERREPRESENTED IN TRIALS THE SUCCESS RATE OF THERAPY FOR CHD IS SIMILAR IN WOMEN AND MAN THE COMPLICATION
D. D. TSIKADERIS MD, FESC SAINT LUKES THESSALONIKI EPIDEMIOLOGY DIAGNOSIS RESPONSE TO THERAPY PROGNOSIS UNDERREPRESENTED IN TRIALS THE SUCCESS RATE OF THERAPY FOR CHD IS SIMILAR IN WOMEN AND MAN THE COMPLICATION
System.
 System www.penumbrainc.com POD System Case Examples 45 cm of POD Packing Coil in 1.5 to 3 mm diameter dilating vessel Bronchial Artery Embolization Dr. Amit Kakkar and Dr. Aksim Rivera, Bronx, NY Inflow:
System www.penumbrainc.com POD System Case Examples 45 cm of POD Packing Coil in 1.5 to 3 mm diameter dilating vessel Bronchial Artery Embolization Dr. Amit Kakkar and Dr. Aksim Rivera, Bronx, NY Inflow:
Clinical Study Age Differences in Long Term Outcomes of Coronary Patients Treated with Drug Eluting Stents at a Tertiary Medical Center
 Aging Research Volume 2013, Article ID 471026, 4 pages http://dx.doi.org/10.1155/2013/471026 Clinical Study Age Differences in Long Term Outcomes of Coronary Patients Treated with Drug Eluting Stents at
Aging Research Volume 2013, Article ID 471026, 4 pages http://dx.doi.org/10.1155/2013/471026 Clinical Study Age Differences in Long Term Outcomes of Coronary Patients Treated with Drug Eluting Stents at
LIBERTY 360 Study. 15-Jun-2018 Data 1. Olinic Dm, et al. Int Angiol. 2018;37:
 LIBERTY 360 Study LIBERTY is a prospective, observational, multi-center study to evaluate procedural and long-term clinical and economic outcomes of endovascular device interventions in patients with symptomatic
LIBERTY 360 Study LIBERTY is a prospective, observational, multi-center study to evaluate procedural and long-term clinical and economic outcomes of endovascular device interventions in patients with symptomatic
Excimer Laser for Coronary Intervention: Case Study RADIAL APPROACH: CORONARY LASER ATHERECTOMY FOR CTO OF THE LAD FOLLOWED BY PTCA NO STENTING
 Excimer Laser for Coronary Intervention: Case Study RADIAL APPROACH: CORONARY LASER ATHERECTOMY FOR CTO OF THE LAD FOLLOWED BY PTCA NO STENTING 1 2013 Spectranetics. All Rights Reserved. Approved for External
Excimer Laser for Coronary Intervention: Case Study RADIAL APPROACH: CORONARY LASER ATHERECTOMY FOR CTO OF THE LAD FOLLOWED BY PTCA NO STENTING 1 2013 Spectranetics. All Rights Reserved. Approved for External
Ruby Coil. Large Volume Detachable Coils
 Ruby Coil Ruby Case Examples 38 mm Hepatic Artery Aneurysm Pulmonary AVM 2 Coils Dr. James Benenati Miami Cardiac and Vascular Institute, FL Y90 Embolization Type 2 Endoleak Dr. J Moskovitz Florida Hospital,
Ruby Coil Ruby Case Examples 38 mm Hepatic Artery Aneurysm Pulmonary AVM 2 Coils Dr. James Benenati Miami Cardiac and Vascular Institute, FL Y90 Embolization Type 2 Endoleak Dr. J Moskovitz Florida Hospital,
Innovation by design. Technology that sets a new standard
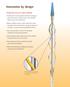 Innovation by design Technology that sets a new standard Flexible nitinol scoring element with three rectangular spiral struts works in tandem with a semi-compliant balloon to score the target lesion Balloon
Innovation by design Technology that sets a new standard Flexible nitinol scoring element with three rectangular spiral struts works in tandem with a semi-compliant balloon to score the target lesion Balloon
Online Supplementary Data. Country Number of centers Number of patients randomized
 A Randomized, Double-Blind, -Controlled, Phase-2B Study to Evaluate the Safety and Efficacy of Recombinant Human Soluble Thrombomodulin, ART-123, in Patients with Sepsis and Suspected Disseminated Intravascular
A Randomized, Double-Blind, -Controlled, Phase-2B Study to Evaluate the Safety and Efficacy of Recombinant Human Soluble Thrombomodulin, ART-123, in Patients with Sepsis and Suspected Disseminated Intravascular
COMPARE Trial Elvin Kedhi Maasstad Ziekenhuis Rotterdam The Netherlands
 COMPARE Trial Elvin Kedhi Maasstad Ziekenhuis Rotterdam The Netherlands TCTAP 2010 Seoul, Korea Disclosures Research Foundation of the Cardiology Department has received unrestricted research grants from:
COMPARE Trial Elvin Kedhi Maasstad Ziekenhuis Rotterdam The Netherlands TCTAP 2010 Seoul, Korea Disclosures Research Foundation of the Cardiology Department has received unrestricted research grants from:
Three-Year Clinical Outcomes with Everolimus-Eluting Bioresorbable Scaffolds: Results from the Randomized ABSORB III Trial Stephen G.
 Three-Year Clinical Outcomes with Everolimus-Eluting Bioresorbable Scaffolds: Results from the Randomized ABSORB III Trial Stephen G. Ellis MD Dean J. Kereiakes MD and Gregg W. Stone MD for the ABSORB
Three-Year Clinical Outcomes with Everolimus-Eluting Bioresorbable Scaffolds: Results from the Randomized ABSORB III Trial Stephen G. Ellis MD Dean J. Kereiakes MD and Gregg W. Stone MD for the ABSORB
Patient. Clinical data Indications: Operation date. Comorbidities: Patient code Birth date: / /
 Patient Patient code Birth date: / / Sex: Male Height (cm): Female Weight (kg): Risk Factors: Family history of coronary disease: Hypertension Dyslipidemia Peripheral disease Diabetes Comorbidities: No
Patient Patient code Birth date: / / Sex: Male Height (cm): Female Weight (kg): Risk Factors: Family history of coronary disease: Hypertension Dyslipidemia Peripheral disease Diabetes Comorbidities: No
ER REBOA Catheter. Instructions for Use
 ER REBOA Catheter Instructions for Use Prytime Medical Devices, Inc. 229 N. Main Street Boerne, TX 78006, USA feedback@prytimemedical.com www.prytimemedical.com US 1 210 340 0116 U.S. and Foreign Patents
ER REBOA Catheter Instructions for Use Prytime Medical Devices, Inc. 229 N. Main Street Boerne, TX 78006, USA feedback@prytimemedical.com www.prytimemedical.com US 1 210 340 0116 U.S. and Foreign Patents
An Important Message for Patients and Healthcare Professionals Who Depend on IMBRUVICA (ibrutinib)
 An Important Message for Patients and Healthcare Professionals Who Depend on IMBRUVICA (ibrutinib) IMBRUVICA (ibrutinib) is a meaningful and important treatment option for patients with chronic lymphocytic
An Important Message for Patients and Healthcare Professionals Who Depend on IMBRUVICA (ibrutinib) IMBRUVICA (ibrutinib) is a meaningful and important treatment option for patients with chronic lymphocytic
TAXUS Paclitaxel-Eluting Coronary Stent System
 Indications, contraindications, warnings and instructions for use can be found in the product labeling supplied with each product. CAUTION: Federal (U.S.A.) law and governing law outside the U.S.A. restricts
Indications, contraindications, warnings and instructions for use can be found in the product labeling supplied with each product. CAUTION: Federal (U.S.A.) law and governing law outside the U.S.A. restricts
Update from Korea on the Lutonix SFA registry 12 month data
 Update from Korea on the Lutonix SFA registry 12 month data Prospective, Multicenter, Post-Market Registy Assessing the Clinical Use and Safety of the Lutonix Drug Coated Balloon in Femoropopliteal Arteries
Update from Korea on the Lutonix SFA registry 12 month data Prospective, Multicenter, Post-Market Registy Assessing the Clinical Use and Safety of the Lutonix Drug Coated Balloon in Femoropopliteal Arteries
Understanding aneurysms and flow diversion treatment
 Surpass Streamline Flow Diverter See package insert for complete indications, contraindications, warnings and instructions for use. INTENDED USE / INDICATIONS FOR USE The Surpass Streamline Flow Diverter
Surpass Streamline Flow Diverter See package insert for complete indications, contraindications, warnings and instructions for use. INTENDED USE / INDICATIONS FOR USE The Surpass Streamline Flow Diverter
SHARED DECISION MAKING: AN EVIDENCE-BASED CORNERSTONE OF LAAC THERAPY
 SHARED DECISION MAKING: AN EVIDENCE-BASED CORNERSTONE OF LAAC THERAPY SHARED DECISION MAKING: AN EVIDENCE BASED CORNERSTONE OF LAAC THERAPY Shared decision making is a collaborative process that allows
SHARED DECISION MAKING: AN EVIDENCE-BASED CORNERSTONE OF LAAC THERAPY SHARED DECISION MAKING: AN EVIDENCE BASED CORNERSTONE OF LAAC THERAPY Shared decision making is a collaborative process that allows
Final Clinical and Angiographic Results From a Nationwide Registry of FIREBIRD Sirolimus- Eluting Stent: Firebird In China (FIC) Registry (PI R. Gao)
 The Microport FIREBIRD Polymer-based Sirolimus- Eluting Stent Clinical Trial Program Update: The FIC and FIREMAN Registries Junbo Ge, MD, FACC, FESC, FSCAI On behalf of Runlin Gao (FIC PI) and Haichang
The Microport FIREBIRD Polymer-based Sirolimus- Eluting Stent Clinical Trial Program Update: The FIC and FIREMAN Registries Junbo Ge, MD, FACC, FESC, FSCAI On behalf of Runlin Gao (FIC PI) and Haichang
TRIAS HR Pilot Study
 Late Breaking Clinical Trials TCT, October 22 nd 27, Washington, USA TRIAS HR Pilot Study RCT comparing Genous EPC capturing stent with Taxus Paclitaxel eluting stent Robbert J de Winter MD PhD FESC Academic
Late Breaking Clinical Trials TCT, October 22 nd 27, Washington, USA TRIAS HR Pilot Study RCT comparing Genous EPC capturing stent with Taxus Paclitaxel eluting stent Robbert J de Winter MD PhD FESC Academic
New Generation Drug- Eluting Stent in Korea
 New Generation Drug- Eluting Stent in Korea Young-Hak Kim, MD, PhD Department of Cardiology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea Purpose To briefly introduce the
New Generation Drug- Eluting Stent in Korea Young-Hak Kim, MD, PhD Department of Cardiology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea Purpose To briefly introduce the
Patient Brochure. Clearstream Technologies, Ltd. Moyne Upper Enniscorthy Co. Wexford, Ireland. PK Rev. 0 05/17
 Patient Brochure Clearstream Technologies, Ltd. Moyne Upper Enniscorthy Co. Wexford, Ireland PK1411100 Rev. 0 05/17 LIFESTREAM Patient Brochure If you or a member of your family has been diagnosed with
Patient Brochure Clearstream Technologies, Ltd. Moyne Upper Enniscorthy Co. Wexford, Ireland PK1411100 Rev. 0 05/17 LIFESTREAM Patient Brochure If you or a member of your family has been diagnosed with
TRIAL UPDATE 1. ISAR TRIPLE SECURITY Trial. Dr Deven Patel Royal Free Hospital
 TRIAL UPDATE 1 ISAR TRIPLE SECURITY Trial Dr Deven Patel Royal Free Hospital NO CONFLICT OF INTEREST TO DECLARE ISAR TRIPLE Comparison of 6 weeks vs 6 months Triple Therapy in patients on oral anticoagulation
TRIAL UPDATE 1 ISAR TRIPLE SECURITY Trial Dr Deven Patel Royal Free Hospital NO CONFLICT OF INTEREST TO DECLARE ISAR TRIPLE Comparison of 6 weeks vs 6 months Triple Therapy in patients on oral anticoagulation
VERNACULAR Trial & Clinical Experience with the VENOVO Venous Stent
 Stephen Black, MD VERNACULAR Trial & Clinical Experience with the VENOVO Venous Stent 1 Speaker Disclaimers The speakers presentation today is on behalf of Bard Peripheral Vascular, Inc. Any discussion
Stephen Black, MD VERNACULAR Trial & Clinical Experience with the VENOVO Venous Stent 1 Speaker Disclaimers The speakers presentation today is on behalf of Bard Peripheral Vascular, Inc. Any discussion
EXCEL vs. NOBLE: How to Treat Left Main Disease in 2017 AATS International Cardiovascular Symposium December 8-9, 2017
 EXCEL vs. NOBLE: How to Treat Left Main Disease in 2017 AATS International Cardiovascular Symposium December 8-9, 2017 Igor F. Palacios, MD Director of Interventional Cardiology Professor of Medicine Massachusetts
EXCEL vs. NOBLE: How to Treat Left Main Disease in 2017 AATS International Cardiovascular Symposium December 8-9, 2017 Igor F. Palacios, MD Director of Interventional Cardiology Professor of Medicine Massachusetts
Watchman. Left Atrial Appendage Closure Device. Uniquely engineered for the LAA 1-3 with proven safety and longterm efficacy. 4-8
 TM Watchman Left Atrial Appendage Closure Device PROOF OFLEADERSHIP Uniquely engineered for the LAA 1-3 with proven safety and longterm efficacy. 4-8 Patients with AF have a 5x increased risk of stroke.
TM Watchman Left Atrial Appendage Closure Device PROOF OFLEADERSHIP Uniquely engineered for the LAA 1-3 with proven safety and longterm efficacy. 4-8 Patients with AF have a 5x increased risk of stroke.
FROM THE EVERYDAY TO THE EXTRAORDINARY
 FROM THE EVERYDAY TO THE EXTRAORDINARY Created with the collaboration of more than 250 physicians around the world, ENDURANT empowers you to create stronger outcomes for more patients, including those
FROM THE EVERYDAY TO THE EXTRAORDINARY Created with the collaboration of more than 250 physicians around the world, ENDURANT empowers you to create stronger outcomes for more patients, including those
Coronary drug-eluting stents (DES) were first approved
 Thrombosis in Coronary Drug-Eluting Stents Report From the Meeting of the Circulatory System Medical Devices Advisory Panel of the Food and Drug Administration Center for Devices and Radiologic Health,
Thrombosis in Coronary Drug-Eluting Stents Report From the Meeting of the Circulatory System Medical Devices Advisory Panel of the Food and Drug Administration Center for Devices and Radiologic Health,
VIRTUS: Trial Design and Primary Endpoint Results
 VIRTUS: Trial Design and Primary Endpoint Results Mahmood K. Razavi, MD St. Joseph Cardiac and Vascular Center Orange, CA, USA IMPORTANT INFORMATION: These materials are intended to describe common clinical
VIRTUS: Trial Design and Primary Endpoint Results Mahmood K. Razavi, MD St. Joseph Cardiac and Vascular Center Orange, CA, USA IMPORTANT INFORMATION: These materials are intended to describe common clinical
Pathology of percutaneous interventions (PCI) in coronary arteries. Allard van der Wal, MD.PhD; Pathologie AMC, Amsterdam, NL
 Pathology of percutaneous interventions (PCI) in coronary arteries Allard van der Wal, MD.PhD; Pathologie AMC, Amsterdam, NL Percutaneous Coronary Intervention (PCI) Definition: transcatheter opening of
Pathology of percutaneous interventions (PCI) in coronary arteries Allard van der Wal, MD.PhD; Pathologie AMC, Amsterdam, NL Percutaneous Coronary Intervention (PCI) Definition: transcatheter opening of
PCI for In-Stent Restenosis. CardioVascular Research Foundation
 PCI for In-Stent Restenosis ISR of BMS Patterns of In-Stent Restenosis Pattern I : Focal Type IA: Articulation / Gap Type IB: Marginal Type IC: Focal body Type ID: Multifocal Pattern II,III,IV : Diffuse
PCI for In-Stent Restenosis ISR of BMS Patterns of In-Stent Restenosis Pattern I : Focal Type IA: Articulation / Gap Type IB: Marginal Type IC: Focal body Type ID: Multifocal Pattern II,III,IV : Diffuse
Single Pass MCA Revascularization with Trevo
 Single Pass MCA Revascularization with Trevo By Concentric Medical ProVueTM Retriever Case Reported By Sascha Prothmann, MD Klinikum rechts der Isar, Munich, Germany Images contained herein courtesy of
Single Pass MCA Revascularization with Trevo By Concentric Medical ProVueTM Retriever Case Reported By Sascha Prothmann, MD Klinikum rechts der Isar, Munich, Germany Images contained herein courtesy of
INSTRUCTIONS FOR USE
 PERICARDIUM COVERED STENT (PCS) INSTRUCTIONS FOR USE STERILE - Ethylene Oxide Sterilized. For single use only. Do not re-sterilize. THE CLINICAL TECHNIQUES AND PROCEDURES DESCRIBED HERE DO NOT REPRESENT
PERICARDIUM COVERED STENT (PCS) INSTRUCTIONS FOR USE STERILE - Ethylene Oxide Sterilized. For single use only. Do not re-sterilize. THE CLINICAL TECHNIQUES AND PROCEDURES DESCRIBED HERE DO NOT REPRESENT
VIVO-EU Results: Prospective European Study of the Zilver Vena TM Venous Stent in the Treatment of Symptomatic Iliofemoral Venous Outflow Obstruction
 VIVO-EU Results: Prospective European Study of the Zilver Vena TM Venous Stent in the Treatment of Symptomatic Iliofemoral Venous Outflow Obstruction Gerard J O Sullivan, M.D. and Jennifer McCann-Brown,
VIVO-EU Results: Prospective European Study of the Zilver Vena TM Venous Stent in the Treatment of Symptomatic Iliofemoral Venous Outflow Obstruction Gerard J O Sullivan, M.D. and Jennifer McCann-Brown,
BTK Case Studies Joseph Cardenas, MD AZ Heart & Vascular, Yuma, AZ
 BTK Case Studies Joseph Cardenas, MD AZ Heart & Vascular, Yuma, AZ 1 Case 1 78 yr. old female Rutherford Class II/III lesion 1 block claudicant 2 Pre Treatment Post Treatment Anterior Tibial Artery Occlusion
BTK Case Studies Joseph Cardenas, MD AZ Heart & Vascular, Yuma, AZ 1 Case 1 78 yr. old female Rutherford Class II/III lesion 1 block claudicant 2 Pre Treatment Post Treatment Anterior Tibial Artery Occlusion
ISAR-LEFT MAIN: A Randomized Clinical Trial on Drug-Eluting Stents for Unprotected Left Main Lesions
 Julinda Mehilli, MD Deutsches Herzzentrum Technische Universität Munich Germany ISAR-LEFT MAIN: A Randomized Clinical Trial on Drug-Eluting Stents for Unprotected Left Main Lesions Background Left main
Julinda Mehilli, MD Deutsches Herzzentrum Technische Universität Munich Germany ISAR-LEFT MAIN: A Randomized Clinical Trial on Drug-Eluting Stents for Unprotected Left Main Lesions Background Left main
Element Clinical Program Perseus Late Breaking News and the Platinum Study Design
 Element Clinical Program Perseus Late Breaking News and the Platinum Study Design Ian T. Meredith MBBS, BSc(Hons), Ph.D, FRACP, FACC, FCSANZ, FSCAI, FAHA, FAPSIC Professor and Director of Monash HEART
Element Clinical Program Perseus Late Breaking News and the Platinum Study Design Ian T. Meredith MBBS, BSc(Hons), Ph.D, FRACP, FACC, FCSANZ, FSCAI, FAHA, FAPSIC Professor and Director of Monash HEART
WARNING: MALIGNANCIES AND SERIOUS INFECTIONS,
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use ZORTRESS safely and effectively. See full prescribing information for ZORTRESS. ZORTRESS (everolimus)
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use ZORTRESS safely and effectively. See full prescribing information for ZORTRESS. ZORTRESS (everolimus)
Are Asian Patients Different? - Updates Of Biomatrix Experience In Regional Settings: BEACON II (3 Yr F up) &
 Are Asian Patients Different? - Updates Of Biomatrix Experience In Regional Settings: BEACON II (3 Yr F up) & Biomatrix TM Single Center Experience (Indonesia)(Final 5 Yr F up) T. Santoso University of
Are Asian Patients Different? - Updates Of Biomatrix Experience In Regional Settings: BEACON II (3 Yr F up) & Biomatrix TM Single Center Experience (Indonesia)(Final 5 Yr F up) T. Santoso University of
Prevention of Coronary Stent Thrombosis and Restenosis
 Prevention of Coronary Stent Thrombosis and Restenosis Seong-Wook Park, MD, PhD, FACC Division of Cardiology, Asan Medical Center University of Ulsan College of Medicine, Seoul, Korea 9/12/03 Coronary
Prevention of Coronary Stent Thrombosis and Restenosis Seong-Wook Park, MD, PhD, FACC Division of Cardiology, Asan Medical Center University of Ulsan College of Medicine, Seoul, Korea 9/12/03 Coronary
Drug Eluting Stents Sometimes Fail ESC Stockholm 29 Set 2010 Stent Thrombosis Alaide Chieffo
 Drug Eluting Stents Sometimes Fail ESC Stockholm 29 Set 2010 Stent Thrombosis 11.45-12.07 Alaide Chieffo San Raffaele Scientific Institute, Milan, Italy Historical Perspective 25 20 15 10 5 0 Serruys 1991
Drug Eluting Stents Sometimes Fail ESC Stockholm 29 Set 2010 Stent Thrombosis 11.45-12.07 Alaide Chieffo San Raffaele Scientific Institute, Milan, Italy Historical Perspective 25 20 15 10 5 0 Serruys 1991
Introduction 3. What is Peripheral Vascular Disease? 5. What Are Some of the Symptoms of Peripheral Vascular Disease? 6
 Patient Information Table of Contents Introduction 3 What is Peripheral Vascular Disease? 5 What Are Some of the Symptoms of Peripheral Vascular Disease? 6 What Causes Peripheral Vascular Disease? 7 How
Patient Information Table of Contents Introduction 3 What is Peripheral Vascular Disease? 5 What Are Some of the Symptoms of Peripheral Vascular Disease? 6 What Causes Peripheral Vascular Disease? 7 How
New Jersey Cardiac Catheterization Data Registry, Version 2.0 (Please report data only for patients 16 years or older.)
 A. ADMINISTRATIVE New Jersey Cardiac Catheterization Data Registry, Version 2.0 (Please report data only for patients 16 years or older.) 1. Facility Code: 2. Facility Name: 3. Procedure Type (Choose only
A. ADMINISTRATIVE New Jersey Cardiac Catheterization Data Registry, Version 2.0 (Please report data only for patients 16 years or older.) 1. Facility Code: 2. Facility Name: 3. Procedure Type (Choose only
MULTIVESSEL PCI. IN DRUG-ELUTING STENT RESTENOSIS DUE TO STENT FRACTURE, TREATED WITH REPEAT DES IMPLANTATION
 MULTIVESSEL PCI. IN DRUG-ELUTING STENT RESTENOSIS DUE TO STENT FRACTURE, TREATED WITH REPEAT DES IMPLANTATION C. Graidis, D. Dimitriadis, A. Ntatsios, V. Karasavvides Euromedica Kyanous Stavros, Thessaloniki.
MULTIVESSEL PCI. IN DRUG-ELUTING STENT RESTENOSIS DUE TO STENT FRACTURE, TREATED WITH REPEAT DES IMPLANTATION C. Graidis, D. Dimitriadis, A. Ntatsios, V. Karasavvides Euromedica Kyanous Stavros, Thessaloniki.
A PATIENT`S GUIDE TO CORONARY ARTERY DISEASE. AND YOUR NIRxcell CoCr CORONARY Stent on RX System
 A PATIENT`S GUIDE TO CORONARY ARTERY DISEASE AND YOUR NIRxcell CoCr CORONARY Stent on RX System TABLE OF CONTENTS ABOUT THIS BOOKLET PAGE 3 WHAT IS CORONARY ARTERY DISEASE (CAD) CORONARY ARTERY DISEASE
A PATIENT`S GUIDE TO CORONARY ARTERY DISEASE AND YOUR NIRxcell CoCr CORONARY Stent on RX System TABLE OF CONTENTS ABOUT THIS BOOKLET PAGE 3 WHAT IS CORONARY ARTERY DISEASE (CAD) CORONARY ARTERY DISEASE
Lecture 8 Cardiovascular Health Lecture 8 1. Introduction 2. Cardiovascular Health 3. Stroke 4. Contributing Factors
 Lecture 8 Cardiovascular Health 1 Lecture 8 1. Introduction 2. Cardiovascular Health 3. Stroke 4. Contributing Factors 1 Human Health: What s Killing Us? Health in America Health is the U.S Average life
Lecture 8 Cardiovascular Health 1 Lecture 8 1. Introduction 2. Cardiovascular Health 3. Stroke 4. Contributing Factors 1 Human Health: What s Killing Us? Health in America Health is the U.S Average life
Drug eluting stents. Where are we now and what can we expect in 2003? Tony Gershlick Leicester
 Drug eluting stents Where are we now and what can we expect in 2003? Tony Gershlick Leicester Trials Real World What we need i. Prevent restenosis cost effective Either : - Treat all at equivalent cost
Drug eluting stents Where are we now and what can we expect in 2003? Tony Gershlick Leicester Trials Real World What we need i. Prevent restenosis cost effective Either : - Treat all at equivalent cost
Nobori Clinical Studies Up-dates. Gian Battista DANZI, M.D. Ospedale Maggiore Policlinico University of Milan, Italy
 Nobori Clinical Studies Up-dates Gian Battista DANZI, M.D. Ospedale Maggiore Policlinico University of Milan, Italy Drug Eluting Stents High benefit in preventing restenosis and improving quality of life
Nobori Clinical Studies Up-dates Gian Battista DANZI, M.D. Ospedale Maggiore Policlinico University of Milan, Italy Drug Eluting Stents High benefit in preventing restenosis and improving quality of life
Komplexe Koronarintervention heute: Von Syntax zu bioresorbierbaren Stents
 Komplexe Koronarintervention heute: Von Syntax zu bioresorbierbaren Stents Prof. Dr. med. Julinda Mehilli Medizinische Klinik und Poliklinik I Klinikum der Universität München Campus Großhadern Key Factors
Komplexe Koronarintervention heute: Von Syntax zu bioresorbierbaren Stents Prof. Dr. med. Julinda Mehilli Medizinische Klinik und Poliklinik I Klinikum der Universität München Campus Großhadern Key Factors
10/8/2018. Lecture 9. Cardiovascular Health. Lecture Heart 2. Cardiovascular Health 3. Stroke 4. Contributing Factor
 Lecture 9 Cardiovascular Health 1 Lecture 9 1. Heart 2. Cardiovascular Health 3. Stroke 4. Contributing Factor 1 The Heart Muscular Pump The Heart Receives blood low pressure then increases the pressure
Lecture 9 Cardiovascular Health 1 Lecture 9 1. Heart 2. Cardiovascular Health 3. Stroke 4. Contributing Factor 1 The Heart Muscular Pump The Heart Receives blood low pressure then increases the pressure
JETSTREAM Atherectomy System DELIVERING VERSATILITY TO RESTORE FLOW
 JETSTREAM Atherectomy System DELIVERING VERSATILITY TO RESTORE FLOW DISCOVER THE VALUE OF VERSATILITY Versatility means not having to guess the morphology! Peripheral arterial lesions can present with
JETSTREAM Atherectomy System DELIVERING VERSATILITY TO RESTORE FLOW DISCOVER THE VALUE OF VERSATILITY Versatility means not having to guess the morphology! Peripheral arterial lesions can present with
IN-STENT RESTENOSIS. K.Boerlage-van Dijk CarVasZ 2014
 IN-STENT RESTENOSIS K.Boerlage-van Dijk CarVasZ 2014 Definition ISR Angiographic: recurrent diameter stenosis >50% at the stent segment or edges (5-mm segments adjacent to stent) Mehran system morphological
IN-STENT RESTENOSIS K.Boerlage-van Dijk CarVasZ 2014 Definition ISR Angiographic: recurrent diameter stenosis >50% at the stent segment or edges (5-mm segments adjacent to stent) Mehran system morphological
Full Novartis CTRD Results Template
 Full Novartis CTRD Results Template Sponsor Novartis Generic Drug Name vildagliptin Therapeutic Area of Trial Type 2 diabetes Approved Indication Type 2 diabetes Protocol Number CLAF237A23138E1 Title A
Full Novartis CTRD Results Template Sponsor Novartis Generic Drug Name vildagliptin Therapeutic Area of Trial Type 2 diabetes Approved Indication Type 2 diabetes Protocol Number CLAF237A23138E1 Title A
PEARL Registry Update Overview Venous Arterial AV Access
 PEARL Registry Update Overview Venous Arterial AV Access PEARL Registry Overview (as of 10 Sep12*) Overview Venous Arterial AV Access HOME Topic Data Support Comments Study Design Prospective, non-randomized,
PEARL Registry Update Overview Venous Arterial AV Access PEARL Registry Overview (as of 10 Sep12*) Overview Venous Arterial AV Access HOME Topic Data Support Comments Study Design Prospective, non-randomized,
Chapter 4 Section 9.1
 Surgery Chapter 4 Section 9.1 Issue Date: August 26, 1985 Authority: 32 CFR 199.4(c)(2) and (c)(3) 1.0 CPT 1 PROCEDURE CODES 33010-33130, 33140, 33141, 33200-37186, 37195-37785, 92950-93272, 93303-93581,
Surgery Chapter 4 Section 9.1 Issue Date: August 26, 1985 Authority: 32 CFR 199.4(c)(2) and (c)(3) 1.0 CPT 1 PROCEDURE CODES 33010-33130, 33140, 33141, 33200-37186, 37195-37785, 92950-93272, 93303-93581,
Clinical Investigations
 Clinical Investigations Clinical Outcomes for Single Stent and Multiple Stents in Contemporary Practice Qiao Shu Bin, MD; Liu Sheng Wen, MD; Xu Bo, BS; Chen Jue, MD; Liu Hai Bo, MD; Yang Yue Jin, MD; Chen
Clinical Investigations Clinical Outcomes for Single Stent and Multiple Stents in Contemporary Practice Qiao Shu Bin, MD; Liu Sheng Wen, MD; Xu Bo, BS; Chen Jue, MD; Liu Hai Bo, MD; Yang Yue Jin, MD; Chen
HEART AND SOUL STUDY OUTCOME EVENT - MORBIDITY REVIEW FORM
 REVIEW DATE REVIEWER'S ID HEART AND SOUL STUDY OUTCOME EVENT - MORBIDITY REVIEW FORM : DISCHARGE DATE: RECORDS FROM: Hospitalization ER Please check all that may apply: Myocardial Infarction Pages 2, 3,
REVIEW DATE REVIEWER'S ID HEART AND SOUL STUDY OUTCOME EVENT - MORBIDITY REVIEW FORM : DISCHARGE DATE: RECORDS FROM: Hospitalization ER Please check all that may apply: Myocardial Infarction Pages 2, 3,
Turbo-Power. Laser atherectomy catheter. The standard. for ISR
 Turbo-Power Laser atherectomy catheter The standard for ISR Vaporize the ISR challenge In-stent restenosis (ISR) Chance of recurring 7 115,000 + /year (U.S.) 1-6 Repeated narrowing of the arteries after
Turbo-Power Laser atherectomy catheter The standard for ISR Vaporize the ISR challenge In-stent restenosis (ISR) Chance of recurring 7 115,000 + /year (U.S.) 1-6 Repeated narrowing of the arteries after
Cardiothoracic and Cardiothoracic Surgery ICD-10-CM 2014: Reference Mapping Card
 2014: Reference Mapping Card 162.3 Malignant neoplasm upper lobe lung 162.5 Malignant neoplasm lower lobe lung 162.9 lung/bronchus 396.2 396.3 Mitral insufficiency, aortic stenosis Mitral aortic valve
2014: Reference Mapping Card 162.3 Malignant neoplasm upper lobe lung 162.5 Malignant neoplasm lower lobe lung 162.9 lung/bronchus 396.2 396.3 Mitral insufficiency, aortic stenosis Mitral aortic valve
Unprotected LM intervention
 Unprotected LM intervention Guideline for COMBAT Seung-Jung Park, MD, PhD Professor of Internal Medicine, Seoul, Korea Current Recommendation for unprotected LMCA Stenosis Class IIb C in ESC guideline
Unprotected LM intervention Guideline for COMBAT Seung-Jung Park, MD, PhD Professor of Internal Medicine, Seoul, Korea Current Recommendation for unprotected LMCA Stenosis Class IIb C in ESC guideline
Summary of Research and Writing Activities In Cardiovascular Disease
 Summary of Research and Writing Activities In Cardiovascular Disease Carole Alison Chrvala, PhD 919.545.2149 (Work) 919.951.5230 (Mobile) cchrvala@centurylink.net www.healthmattersmedwriting.com 1 Manuscripts
Summary of Research and Writing Activities In Cardiovascular Disease Carole Alison Chrvala, PhD 919.545.2149 (Work) 919.951.5230 (Mobile) cchrvala@centurylink.net www.healthmattersmedwriting.com 1 Manuscripts
Results of Ischemic Heart Disease
 Ischemic Heart Disease: Angina and Myocardial Infarction Ischemic heart disease; syndromes causing an imbalance between myocardial oxygen demand and supply (inadequate myocardial blood flow) related to
Ischemic Heart Disease: Angina and Myocardial Infarction Ischemic heart disease; syndromes causing an imbalance between myocardial oxygen demand and supply (inadequate myocardial blood flow) related to
DEB experience in Gachon Universtiy Gil Hospital (in ISR) Soon Yong Suh MD., PhD. Heart Center Gachon University Gil Hospital Seoul, Korea.
 DEB experience in Gachon Universtiy Gil Hospital (in ISR) Soon Yong Suh MD., PhD. Heart Center Gachon University Gil Hospital Seoul, Korea. In-stent restenosis (ISR) Remains important issue even in the
DEB experience in Gachon Universtiy Gil Hospital (in ISR) Soon Yong Suh MD., PhD. Heart Center Gachon University Gil Hospital Seoul, Korea. In-stent restenosis (ISR) Remains important issue even in the
BC Vascular Day. Contents. November 3, Abdominal Aortic Aneurysm 2 3. Peripheral Arterial Disease 4 6. Deep Venous Thrombosis 7 8
 BC Vascular Day Contents Abdominal Aortic Aneurysm 2 3 November 3, 2018 Peripheral Arterial Disease 4 6 Deep Venous Thrombosis 7 8 Abdominal Aortic Aneurysm Conservative Management Risk factor modification
BC Vascular Day Contents Abdominal Aortic Aneurysm 2 3 November 3, 2018 Peripheral Arterial Disease 4 6 Deep Venous Thrombosis 7 8 Abdominal Aortic Aneurysm Conservative Management Risk factor modification
Atherectomy: Jetstream and Directional. George S. Chrysant, M.D.
 Atherectomy: Jetstream and Directional George S. Chrysant, M.D. Disclosures Abbott Vascular: MAB, consultant, proctor Abiomed: consultant Boston Scientific: MAB, consultant, proctor Medicines Company:
Atherectomy: Jetstream and Directional George S. Chrysant, M.D. Disclosures Abbott Vascular: MAB, consultant, proctor Abiomed: consultant Boston Scientific: MAB, consultant, proctor Medicines Company:
Introduction What Causes Peripheral Vascular Disease? How Do Doctors Treat Peripheral Vascular Disease?... 9
 Patient Information Table of Contents Introduction... 3 What is Peripheral Vascular Disease?... 5 What Are Some of the Symptoms of Peripheral Vascular Disease?... 7 What Causes Peripheral Vascular Disease?...
Patient Information Table of Contents Introduction... 3 What is Peripheral Vascular Disease?... 5 What Are Some of the Symptoms of Peripheral Vascular Disease?... 7 What Causes Peripheral Vascular Disease?...
PRODUCT MONOGRAPH INCLUDING PATIENT MEDICATION INFORMATION
 PRODUCT MONOGRAPH INCLUDING PATIENT MEDICATION INFORMATION Pr Kyprolis carfilzomib for injection 10, 30, 60 mg per vial Antineoplastic Agent Amgen Canada Inc. 6775 Financial Drive, Suite 100 Mississauga,
PRODUCT MONOGRAPH INCLUDING PATIENT MEDICATION INFORMATION Pr Kyprolis carfilzomib for injection 10, 30, 60 mg per vial Antineoplastic Agent Amgen Canada Inc. 6775 Financial Drive, Suite 100 Mississauga,
The BIO revolution: bioadsorbable stents. Federico Conrotto Cardiologia 2 Città della Salute e della Scienza di Torino
 The BIO revolution: bioadsorbable stents Federico Conrotto Cardiologia 2 Città della Salute e della Scienza di Torino BVS stent (Abbot Vascular) Strut Material: Poly-L-Lactic acid Coating Material: Poly-D,L-lactide
The BIO revolution: bioadsorbable stents Federico Conrotto Cardiologia 2 Città della Salute e della Scienza di Torino BVS stent (Abbot Vascular) Strut Material: Poly-L-Lactic acid Coating Material: Poly-D,L-lactide
GORE EXCLUDER AAA Endoprosthesis ANNUAL CLINICAL UPDATE OCTOBER Section I Clinical experience. Section II Worldwide commercial experience
 GORE EXCLUDER AAA Endoprosthesis ANNUAL CLINICAL UPDATE OCTOBER 2018 Abstract This annual clinical update provides a review of the ongoing experience with the GORE EXCLUDER AAA Endoprosthesis used in the
GORE EXCLUDER AAA Endoprosthesis ANNUAL CLINICAL UPDATE OCTOBER 2018 Abstract This annual clinical update provides a review of the ongoing experience with the GORE EXCLUDER AAA Endoprosthesis used in the
CAROTID ARTERY ANGIOPLASTY
 CAROTID ARTERY ANGIOPLASTY Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Medical Coverage Guideline
CAROTID ARTERY ANGIOPLASTY Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Medical Coverage Guideline
OUTCOMES WITH LONGTERM DUAL ANTIPLATELET THERAPY AFTER CORONARY ANGIOPLASTY Ashok Kumar Arigonda, K. Nagendra Prasad, O. Hareesh, R.
 INDIAN JOURNAL OF CARDIOVACULAR DIEAE JOURNAL in women (IJCD) 016 VOL 1 IUE 3 ORIGINAL ARTICLE 1 OUTCOME WITH LONGTERM DUAL ANTIPLATELET THERAPY AFTER CORONARY ANGIOPLATY Ashok Kumar Arigonda, K. Nagendra
INDIAN JOURNAL OF CARDIOVACULAR DIEAE JOURNAL in women (IJCD) 016 VOL 1 IUE 3 ORIGINAL ARTICLE 1 OUTCOME WITH LONGTERM DUAL ANTIPLATELET THERAPY AFTER CORONARY ANGIOPLATY Ashok Kumar Arigonda, K. Nagendra
The present status of selfexpanding. for CLI: Why and when to use. Sean P Lyden MD Cleveland Clinic Cleveland, Ohio
 The present status of selfexpanding and balloonexpandable tibial BMS and DES for CLI: Why and when to use Sean P Lyden MD Cleveland Clinic Cleveland, Ohio Disclosure Speaker name: Sean Lyden, MD I have
The present status of selfexpanding and balloonexpandable tibial BMS and DES for CLI: Why and when to use Sean P Lyden MD Cleveland Clinic Cleveland, Ohio Disclosure Speaker name: Sean Lyden, MD I have
Instructions for Use Reprocessed LASSO Circular Mapping Diagnostic Electrophysiology (EP) Catheter
 Instructions for Use Reprocessed LASSO Circular Mapping Diagnostic Electrophysiology (EP) Catheter Caution: Federal (USA) law restricts this device to sale by or on the order of a physician. DEVICE DESCRIPTION
Instructions for Use Reprocessed LASSO Circular Mapping Diagnostic Electrophysiology (EP) Catheter Caution: Federal (USA) law restricts this device to sale by or on the order of a physician. DEVICE DESCRIPTION
