After successful resuscitation from cardiac arrest (CA),
|
|
|
- Mary Gibbs
- 5 years ago
- Views:
Transcription
1 Critical Time Window for Intra-Arrest Cooling With Cold Saline Flush in a Dog Model of Cardiopulmonary Resuscitation Ala Nozari, MD, PhD; Peter Safar, MD ; S. William Stezoski; Xianren Wu, MD; Scott Kostelnik; Ann Radovsky, DVM, PhD; Samuel Tisherman, MD; Patrick M. Kochanek, MD Background Mild hypothermia improves outcome when induced after cardiac arrest in humans. Recent studies in both dogs and mice suggest that induction of mild hypothermia during cardiopulmonary resuscitation (CPR) greatly enhances its efficacy. In this study, we evaluate the time window for the beneficial effect of intra-arrest cooling in the setting of prolonged CPR in a clinically relevant large-animal model. Methods and Results Seventeen dogs had ventricular fibrillation cardiac arrest no flow of 3 minutes, followed by 7 minutes of CPR basic life support and 50 minutes of advanced life support. In the early hypothermia group (n 9), mild hypothermia (34 C) was induced with an intravenous fluid bolus flush and venovenous blood shunt cooling after 10 minutes of ventricular fibrillation. In the delayed hypothermia group (n 8), hypothermia was induced at ventricular fibrillation 20 minutes. After 60 minutes of ventricular fibrillation, restoration of spontaneous circulation was achieved with cardiopulmonary bypass for 4 hours, and intensive care was given for 96 hours. In the early hypothermia group, 7 of 9 dogs survived to 96 hours, 5 with good neurological outcome. In contrast, 7 of 8 dogs in the delayed hypothermia group died within 37 hours with multiple organ failure (P 0.012). Conclusions Early application of mild hypothermia with cold saline during prolonged CPR enables intact survival. Delay in the induction of mild hypothermia in this setting markedly reduces its efficacy. Our data suggest that if mild hypothermia is used during CPR, it should be applied as early as possible. (Circulation. 2006;113: ) Key Words: cardiopulmonary resuscitation cooling heart arrest hypothermia resuscitation After successful resuscitation from cardiac arrest (CA), hypothermia has been shown in several experimental studies to improve cerebral outcome. 1 5 On the basis of recent clinical studies, therapeutic mild hypothermia is recommended by the American Heart Association and the International Liaison Committee on Resuscitation for Unconscious Survivors of CA. 6,7 Despite a relatively late and slow surface cooling technique, these clinical trials in Europe and Australia documented neurological benefits with mild hypothermia in survivors of out-of-hospital CA. 8,9 Because evidence exists that a delay in cooling negates the beneficial effect of mild hypothermia, 4,10 some have suggested that hypothermia should be initiated as soon as possible after resuscitation or, preferably, during cardiopulmonary resuscitation (CPR) attempts. 5,10 In a recent study of CA in mice, application of mild hypothermia during CPR was shown to enhance outcome compared with its application after restoration of spontaneous circulation (ROSC). 11 Similarly, in a clinically relevant study of prolonged ventricular fibrillation (VF) in dogs, we documented that mild or moderate hypothermia Clinical Perspective p 2696 induced during 40 minutes of CPR attempts preserves organ viability and significantly improves outcome. 12 Intact survival was achieved despite 40 minutes of VF, indicating that effective closed-chest CPR with mild hypothermia opens a therapeutic window of at least 40 minutes for the institution of advanced techniques to restore spontaneous circulation (including cardiopulmonary bypass [CPB]). In clinical cases, however, 60 minutes often is required to initiate CPB In the present study, therefore, we sought to examine the time window for successful application of mild hypothermia during 60 minutes of CPR-resistant VF. We hypothesized that in contrast to delayed induction of hypothermia, early application of mild hypothermia minimizes organ injury during prolonged CPR and enables intact survival. Methods The experimental protocol was approved by the Institutional Animal Care and Use committees of the University of Pittsburgh. All surgery Received February 3, 2006; revision received April 10, 2006; accepted April 17, From the Safar Center for Resuscitation Research (A.N., P.S., S.W.S., X.W., S.K., S.T., P.M.K.) and Departments of Critical Care Medicine (A.N., P.S., S.W.S., X.W., S.T., P.M.K.), Anesthesiology (A.N., P.S., S.W.S., X.W.), Surgery (S.T.), and Pediatrics (P.M.K.), University of Pittsburgh, Pittsburgh, Pa; and WIL Research Laboratories (A.R.), Ashland, Ohio. Deceased. Correspondence to Ala Nozari, MD, PhD, Department of Anesthesia and Critical Care, Massachusetts General Hospital, Harvard Medical School, 55 Fruit St, Boston, MA anozari@partners.org 2006 American Heart Association, Inc. Circulation is available at DOI: /CIRCULATIONAHA
2 Nozari et al Time Window for Intra-Arrest Hypothermia 2691 Figure 1. Study protocol. Three minutes of normovolemic VF was followed by 7 minutes of BLS and 50 minutes of ALS. V-v indicates venovenous. was performed in our animal intensive care unit (ICU) with sterile techniques. 16,17 Protocol The protocol (Figure 1) simulated nonresponsive VF CA that was bridged during closed-chest CPR of 60 minutes from collapse via transport to initiation of CPB in the hospital emergency department Seventeen custom-bred hunting dogs (body weight, 19 to 25 kg; age, 8 to 12 months; Pat s Pine Tree Farm, Williford, Ark) were used. All dogs received the same standardized anesthesia and instrumentation, as described previously. 12,21 Briefly, anesthesia was induced and maintained with titrated doses of halothane in N 2 O/O 2 50%/50%. Gastric and bladder catheters and temperature probes were inserted, and the tympanic membrane temperature (Tty) was controlled at C with heating blankets and heating lamps. Catheters were inserted into the left femoral artery for monitoring of arterial pressure and blood sampling and into the right femoral artery for later CPB. A pulmonary artery catheter was inserted into the left femoral vein and advanced into wedge position. For rapidly controlling Tty, a venovenous extracorporeal shunt cooling system was used. 22 A 13F catheter was inserted into the femoral vein 20 cm into the inferior vena cava and connected to a 15-m-long tubing (3-mm inner diameter; primed with 120 ml isotonic saline and 500 IU heparin) immersed in ice water. There was no additional systemic heparinization. A shunt flow of 10 ml kg 1 min 1 by a roller pump returned the cooled blood via the right external jugular vein into the superior vena cava using a multipleholed 19F catheter. After baseline measurements, the dogs were weaned to spontaneous breathing, and VF was induced with a 95-V AC, 60-Hz transthoracic shock of 2 seconds using subcutaneous needles. Pulselessness was allowed to persist for 3 minutes before initiation of CPR. CPR basic life support (BLS) was then begun with left parasternal chest compressions (dogs were placed supine, then turned 25% to the right to exert pressure over the heart) at a rate of 80 compressions per minute with a mechanical thumper (Michigan Instruments, Grand Rapids, Mich) and bag ventilation (FiO ) at a ratio of 5 compressions to 1 ventilation. After 7 minutes of BLS (10 minutes of VF), the dogs were randomized into 2 treatment groups. In the early hypothermia (EH) group (n 9), cooling was initiated at 10 minutes of VF, together with the advanced life support (ALS), simulating immediate cooling by the paramedics. FiO 2 was increased to 1.0, and 20- g/kg IV boluses of epinephrine were given at 5-minute intervals. Three external transthoracic DC countershocks of 50 J (purposely low to fail to defibrillate) were delivered in rapid sequence. Cooling was induced with a bolus of 20 ml/kg of normal saline at 2 C into the superior vena cava, followed by venovenous extracorporeal pumping at 10 ml kg 1 min 1 until a Tty of 34 C was achieved. The dogs were purposefully maintained in VF for a total of 60 minutes. The delayed hypothermia (DH) group (n 8) was subjected to the same insult, but ALS was provided for another 10 minutes of normothermic VF before cooling was induced. Reperfusion after 60 minutes of VF was achieved with CPB, as described previously The CPB system included a centrifugal pump (Biomedicus, Eden Prairie, Minn), a hollow-fiber membrane oxygenator, and a heat exchanger unit (Medtronic, Anaheim, Calif) and was primed with 10% dextran 40 in isotonic saline:ringer s solution (1:1), with 2 meq/kg sodium bicarbonate and 500 U heparin. Flow of 100% O 2 through the oxygenator was adjusted to keep PaCO 2 at 30 to 35 mm Hg. The CPB flow rate was kept at 100 ml kg 1 min 1 until 120 minutes, at which time it was reduced to 50 ml kg 1 min 1 until the dogs were weaned from CPB at 4 hours. After 15 minutes of recirculation with CPB, DC countershocks of 150 J were delivered; they were increased if needed by 50 J for repeated shocks. If necessary for ROSC, epinephrine 5 g/kg IV was administered and repeated as needed. After ROSC, a norepinephrine infusion was started and titrated to maintain the mean arterial pressure at 90 to 120 mm Hg. To comply with current International Liaison Committee on Resuscitation guidelines, Tty of 34 C was maintained by external means (application of ice, warming/cooling blankets, and heating lamps) until 12 hours in both groups. Controlled ventilation was continued to 48 hours, and intensive care was provided until 96 hours or earlier death. Analgesia and sedation were provided with N 2 O/O 2 50%/50% and boluses of morphine (0.1 to 0.3 mg/kg IV) and diazepam (0.1 to 0.3 mg/kg IV) as needed. At 44 to 48 hours, the dogs were weaned from controlled ventilation and transferred to a step-down ICU, where they were monitored until 96 hours. Outcome Evaluation Performance was evaluated according to overall performance categories (OPC; 1 normal, 2 moderate disability, 3 severe disability but conscious, 4 coma, and 5 death). 16,18 20 Neurological function was evaluated as neurological deficit scores (NDS; 0% to 10% normal, 100% brain death) OPC and NDS were evaluated by 3 independent observers (not blinded) every 8 hours after extubation to obtain best and final values. If different scores were assigned by the observers, an independent blinded physician was asked to evaluate the performance and neurological outcome of the dogs. After final evaluation at 96 hours, the dogs were reanesthetized for morphological studies and brain perfusion fixation for histological damage scoring. 16,17 A pathologist, blinded to treatment, scored 19 distinct anatomic brain regions for severity and extent of ischemic neuronal changes, infarcts, and edema. 17 A total brain histological damage score (HDS) 40 represents moderate damage, and HDS 100 represents severe damage. Myocardial injury was quantified sepa-
3 2692 Circulation June 13, 2006 Figure 2. Tty during CA. Gr indicates group. rately in the right and left ventricles. The degree of myocardial damage was scored from 0 to 5 (0 absent, 1 minimal, 2 mild, 3 moderate, 4 marked, 5 severe) on the basis of the size and anatomic distribution (endocardial, epicardial, or transmural). The myocardial lesions also were scored for pattern (1 focal, 2 multifocal, 3 focally extensive, 4 diffuse), for anatomic location (1 primarily endocardial, 2 primarily epicardial, 3 extensively endocardial and epicardial, 4 transmural), and for appearance (1 exclusively pale, 2 mottled pale and hemorrhagic). The total myocardial damage score was the indexed sum of all the scores (0 no damage, 100 severe damage). Statistical Analysis Repeated-measures ANOVA was performed, followed by Bonferroni/Dunn post hoc tests to identify differences in hemodynamic and arterial blood gas parameters between groups over time. The Fisher exact test was used to assess differences in survival and OPC proportions (dichotomized to OPC 1 and 2 good outcome; OPC 3, 4, or death bad outcome) between groups. Resuscitation variables, HDS, and myocardial damage scores were analyzed with the Mann-Whitney U test. A value of P 0.05 was considered statistically significant. The authors had full access to the data and take full responsibility for their integrity. All authors have read and agree to the manuscript as written. Results Sixteen of 17 dogs remained in protocol for the duration of study. One dog in the EH group was excluded because of accidental extubation leading to CA in the ICU. There were no group differences in body weight or in baseline values of any observed or controlled variables. Temperature (Figure 2) and blood pressures (Figure 3) changed as expected, according to protocol. Changes in arterial ph, serum sodium and potassium concentrations, and norepinephrine infusion rate are summarized in Figure 4. Resuscitation variables are summarized in the Table. ROSC was achieved after 15 to 235 minutes of CPB. All but 1 dog in the EH group survived to 96 hours. In contrast, all but 1 dog in the DH group died within 37 hours with multiple organ failure, including cardiovascular failure (P 0.01). During closed-chest CPR, mean arterial blood pressure was 45 4 mm Hg, with a diastolic pressure of 24 5 mm Hg, without overall group differences (Figure 3). These pressures increased in both groups transiently after the intravenous flush of cold normal saline at 10 or 20 minutes of VF, respectively, and returned to preinfusion levels within 3 minutes, without statistical differences between groups. Venovenous shunt cooling was initiated in both groups with an intravenous saline flush, which decreased Tty from 37.5 C to C (Figure 2). Tty of Figure 3. Mean arterial pressure (MAP) and coronary perfusion pressure (CPP) during and after circulatory arrest. Data are presented as means and SD.
4 Nozari et al Time Window for Intra-Arrest Hypothermia 2693 Figure 5. Final 96-hour outcome. Each dot represents a dog. Values are expressed as median (range). Values in brackets represent HDS at 4 to 37 hours of reperfusion. MDS indicates gross myocardial damage score. Figure 4. ph, serum sodium, and potassium concentrations (mmol/l) and norepinephrine infusion rate ( g/min). 34 C was achieved at minutes after the initiation of cooling (3.5 minutes after the start of venovenous shunt cooling) in both groups (after 16.6 and 25.4 minutes of VF in the EH and DH groups, respectively). In the DH group, unstoppable deterioration after ROSC was due to extracerebral organ failure. In 4 dogs, increased microvascular fluid extravasation resulting in general edema, ascites, and pleural effusion was followed by vasopressor-resistant shock. Oliguria or anuria also was observed in 2 of these dogs, despite normal central venous and pulmonary artery wedge pressures. Two dogs in the DH group developed vasopressor-resistant shock and were euthanized at 27 and 36 hours, respectively; their brains were processed for histological scoring. The 3 remaining dogs died in secondary VF that was resistant to vigorous CPR with antiarrhythmic treatment and repeated countershocks. The only nonsurviving dog in the EH group died with pulmonary edema and hemoptysis. Myocardial necrosis was present in all dogs in both groups despite patent coronary arteries. Superficial subendocardial hemorrhage and papillary muscle necrosis were observed in both ventricles in 2 dogs in the EH group and in 6 of 8 dogs in the DH group (P NS). Milder lesions, consisting of focal areas of subendocardial or subepicardial infarctions, and focally extensive hemorrhagic lesions were present in all dogs, resulting in a total myocardial damage score of 58.5 (range, 43 to 93) in the EH group and 68.5 (range, 47 to 93) in the DH group (P NS). In the EH group, 5 of 7 surviving dogs were functionally normal (OPC 1 or 2), 1 had OPC 3, and 1 had OPC 4 (coma) at 96 hours of recovery. Histologically, 4 of 8 dogs in this group were normal (HDS 0), 1 had HDS 16, 1 had 22, and 1 had 98. The only surviving dog in the DH group was functionally normal at 96 hours (OPC 1, NDS 0) and had an HDS of 32 (mild injury) (Figure 5). Because of early deaths, only 2 additional brains in this group could be studied histologically. One had an HDS score of 38 and the other had 45 (Figure 5). Histopathological changes consisted mainly of focal infarctions and scattered ischemic neurons in the frontal, parietal, and temporal cortexes, as well as multifocal gliosis and vasculitis involving primarily the basal ganglia. Discussion In the present study, we demonstrated that early induction of mild hypothermia preserves the organism in a scenario modeling prolonged unsuccessful CPR attempts, enabling intact survival after up to 60 minutes of VF. Importantly, a 20-minute versus 10-minute delay in cooling negates the beneficial effects of hypothermia in this model of prolonged VF CA. The benefit derived from mild hypothermia after ROSC for cerebral recovery has been well documented. 1 5,8,9 Using a mouse model of CA, Abella et al 11 recently reported improved outcome when cooling was induced during CA but not after ROSC. Similarly, in a clinically relevant large-animal model of CA, we reported survival with full neurological recovery after 40 minutes of VF if mild or moderate hypothermia was initiated during ROSC
5 2694 Circulation June 13, 2006 Resuscitation Variables Group DH EH P Countershocks, total 13 (1 58) 1 (1 8) Countershocks, total energy, J 2755 ( ) 185 ( ) ROSC, min of CPB 51 (15 235) 16.5 (15 80) Total bicarbonate, meq 107 (55 175) 95 (40 230) Total epinephrine, mg 2.45 ( ) 0.75 (0.2 3) 0.01 Total NE, mg ( ) ( ) Duration of NE infusion, h 5.3 ( ) 20.5 ( ) Survival, h 21 (4 96) 96 (48 96) NE indicates norepinephrine. Data are given as median (range). attempts, but not after ROSC. 12 Cooling was started 20 minutes after normothermic CA (3 minutes of no flow, 7 minutes of BLS, and 10 minutes of ALS), simulating the time required for the ambulance to arrive and for the paramedics to attempt ROSC. Our present study is based on these 2 reports and suggests a critical time window to maximize what can be a dramatic benefit of mild hypothermia in this setting. We believe that our data may have broad implications for the potential application of mild cooling during CPR. Moreover, our data suggest that for mild hypothermia to be maximally effective, it should probably be applied as early as possible. Future clinical trials will need to address the safety and efficacy of mild cooling during CPR. In a recent study in patients with acute myocardial infarction, Ly et al 23 failed to document hemodynamic instability or arrhythmias when hypothermia was induced before reperfusion therapy. Recent studies also have shown the benefit of mild and moderate hypothermia on defibrillation success during VF. 24,25 However, it is not clear if there is a critical time window for this additional benefit of intra-arrest hypothermia with regard to defibrillation success. Nevertheless, the use of mild or moderate hypothermia would be expected to facilitate rather than reduce defibrillation success. Thus, our work also builds on these important parallel findings. Our study could also have potential implications for situations in which CPB is attempted to achieve ROSC after prolonged CPR. Clinical studies indicate that, even for witnessed in-hospital CA victims, up to 1 hour often is required for cannulation and implementation of CPB These studies also have shown that a delay 30 minutes in the initiation of CPB can lead to poor outcome despite vigorous CPR. Therefore, we aimed to explore whether mild hypothermia would enable intact survival after 60 minutes of VF, giving a considerable window of time for transportation of the CA victim and implementation of CPB. Our results support this hypothesis and indicate that cooling should be initiated as early as possible to achieve this goal. The fluid load (20 ml/kg) used to initiate the cooling also may affect the chance of survival by altering the blood rheology and improving myocardial and cerebral blood flow. A recent study by Bernard et al 26 demonstrated that a fluid bolus of 30 ml/kg ice-cold lactated Ringer s solution in comatose survivors of CA decreased core temperature by 1.6 C over 25 minutes and improved blood pressure. Nordmark et al 27 also reported a 1.6 C decrease in core body temperature using 30 ml/kg infusion of acetated Ringer s solution at 4 C in pigs. Thus, in the present study, an earlier augmentation of the organ blood flow during the low-flow state of CPR may have resulted in an improved outcome in the EH group. To differentiate between the effects of EH and early fluid load, additional studies are needed with normothermic flush at 10 minutes of VF but cooling at 20 minutes of VF. In additional experiments at our laboratory (n 2), however, early volume expansion with delayed cooling did not result in good outcome: Both dogs died within 26 hours after ROSC in vasopressor-resistant shock. Prior reports on the role of fluid boluses during CPR similarly suggest that the benefit observed in the present study is due more likely to cooling than to volume expansion. 28 These findings support the hypothesis that good outcome in the EH group is, in fact, the result of EH and not a single-volume bolus during CPR. Nevertheless, we cannot rule out a possible synergistic effect of hypothermia and volume administration. The unexpected failure of EH to reduce myocardial damage scores may be attributed to the difference in survival time between groups, which alters the evolution of histological changes after myocardial ischemia and reperfusion injury. 29 Alternatively, this finding could suggest that EH influences myocardial function more than its morphological outcome in this paradigm. One other possibility is that myocardial protection would be enhanced with either a lower target temperature or longer post- ROSC application of mild cooling. Ao et al 30 reported histological myocardial protection with 20 hours of mild cooling in a 15-minute VF model, followed by extracorporeal support. Likewise, in a rabbit model of myocardial infarction, Hale et al 31 reported myocardial protection with regional hypothermia induced early after ischemia. These findings parallel our recent work showing that sustained mild cooling further enhances neuronal protection after CA. 32 Furthermore, we cannot rule out the possible interaction between central nervous system injury and multiple organ failure, with EH indirectly improving survival by mitigating central nervous system injury. We also recog-
6 Nozari et al Time Window for Intra-Arrest Hypothermia 2695 nize the limitation that our model does not incorporate coronary artery disease, which often complicates refractory CA. Further studies of the specific mechanisms underlying the potential benefits of early application of mild hypothermia during CPR are needed. We recognize that the effect of hypothermia on the capability of achieving ROSC requires further studies. Our findings, however, magnify the importance of those studies because earlier application of mild hypothermia during CPR was dramatically effective. For CPR-resistant cases, nevertheless, the authors suggest that mild hypothermia should be considered as soon as possible during CPR in cases in which a bridge to prolonged CPB for delayed resuscitation is being considered. Early, but not delayed, hypothermia enables intact survival after prolonged nonresponsive VF CA in our model. Acknowledgments Jeremy Henchir, Sherman Culver, and Alan Abraham provided excellent technical support. Fran Mistrick and Valerie Sabo helped with the preparation of the manuscript. This article was written in honor of the late Dr Peter Safar. Sources of Funding This work was supported by grant DAMD from the US Army MRMC / TATRC and the Laerdal Foundation for Acute Medicine. A. Nozari received research grants from Laerdal Foundation for Acute Medicine, Swedish Heart and Lung Foundation, and Olof Norlanders Foundation, Karolinska Institute. Disclosures S.W. Stezoski, X. Wu, P.M. Kochanek, and S.A. Tisherman have ownership interest in the US provisional patent Method of Inducing Suspended Animation Following Cardiopulmonary Arrest. S.A. Tisherman received research grants from the Resuscitation Outcomes Consortium s Multicenter Clinical Trials Network for Trauma and Cardiac Arrest and Novel Resuscitation From Lethal Hemorrhage and Suspended Animation for Delayed Resuscitation. He also received the Laerdal Award from the Society of Critical Care Medicine and serves as a consultant and on the advisory board for Biopure, Trauma Studies Committee. P.M. Kochanek received research grants titled Adenosine and Traumatic Brain Injury and Training in Pediatric Neurointensive Care and Resuscitation Research from the NIH and a research grant titled Novel Nitroxide Resuscitation Strategies in Experimental Traumatic Brain Injury from the United States Army. The remaining authors report no conflicts. References 1. Leonov Y, Sterz F, Safar P, Radovsky A, Oku K, Tisherman S, Stezoski SW. Mild cerebral hypothermia during and after cardiac arrest improves neurologic outcome in dogs. J Cereb Blood Flow Metab. 1990;10: Sterz F, Safar P, Tisherman S, Radovsky A, Kuboyama K, Oku K. Mild hypothermic cardiopulmonary resuscitation improves outcome after prolonged cardiac arrest in dogs. Crit Care Med. 1991;19: Weinrauch V, Safar P, Tisherman S, Kuboyama K, Radovsky A. Beneficial effect of mild hypothermia and detrimental effect of deep hypothermia after cardiac arrest in dogs. Stroke. 1992;23: Kuboyama K, Safar P, Radovsky A, Tisherman SA, Stezoski SW, Alexander H. Delay in cooling negates the beneficial effect of mild resuscitative cerebral hypothermia after cardiac arrest in dogs: a prospective, randomized study. Crit Care Med. 1993;21: Safar P, Xiao F, Radovsky A, Tanigawa K, Ebmeyer U, Bircher N, Alexander H, Stezoski SW. Improved cerebral resuscitation from cardiac arrest in dogs with mild hypothermia plus blood flow promotion. Stroke. 1996;27: American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care, Part 7.5: Postresuscitation support. Circulation. 2005;112(suppl IV):IV-84 IV Nolan JP, Morley PT, Vanden Hoek TL, Hickey RW, Kloeck WG, Billi J, Bottiger BW, Morley PT, Nolan JP, Okada K, Reyes C, Shuster M, Steen PA, Weil MH, Wenzel V, Hickey RW, Carli P, Vanden Hoek TL, Atkins D, for the International Liaison Committee on Resuscitation. Therapeutic hypothermia after cardiac arrest: an advisory statement by the Advanced Life Support Task Force of the International Liaison Committee on Resuscitation. Circulation. 2003; 108: Hypothermia After Cardiac Arrest Study Group. Mild therapeutic hypothermia to improve the neurologic outcome after cardiac arrest. N Engl J Med. 2002;346: Bernard SA, Gray TW, Buist MD, Jones BM, Silvester W, Gutteridge G, Smith K. Treatment of comatose survivors of out-of-hospital cardiac arrest with induced hypothermia. N Engl J Med. 2002;346: Takata K, Takeda Y, Sato T, Nakatsuka H, Yokoyama M, Morita K. Effects of hypothermia for a short period on histologic outcome and extracellular glutamate concentration during and after cardiac arrest in rats. Crit Care Med. 2005;33: Abella B, Zhao D, Alvarado J, Hamann K, Vanden Hoek T, Becker L. Intra-arrest cooling improves outcomes in a murine cardiac arrest model. Circulation. 2004;109: Nozari A, Safar P, Stezoski S, Wu X, Henchir J, Radovsky A, Hanson K, Klein E, Kochanek PM, Tisherman SA. Mild hypothermia during prolonged cardiopulmonary cerebral resuscitation increases conscious survival in dogs. Crit Care Med. 2004;32: Chen YS, Chao A, Yu HY, Ko WJ, Wu IH, Chen RJ, Huang SC, Lin FY, Wang SS. Analysis and results of prolonged resuscitation in cardiac arrest patients rescued by extracorporeal membrane oxygenation. J Am Coll Cardiol. 2003;41: Kurusz M, Zwischenberger JB. Percutaneous cardiopulmonary bypass for cardiac emergencies. Perfusion. 2002;17: Wittenmyer BL, Pomerants BJ, Duff SB, Watson WD, Blackford JM. Single hospital experience with emergency cardiopulmonary bypass using the portable CPS (Bard) system. J Extra Corpor Technol. 1997;29: Safar P, Gisvold S, Vaagenes P, Hedrickx H, Bar-Joseph G, Bircher N, Stezoski W, Alexander H. Long-term animal models for the study of global brain ischemia. In: Wauquier A, Borgers M, Amery WK, eds. Protection of Tissue Against Hypoxia. Amsterdam: Elsevier; 1982: Radovsky A, Safar P, Sterz F, Leonov Y, Reich H, Kuboyama K. Regional prevalence and distribution of ischemic neurons in dog brains 96 hours after cardiac arrest of 0 to 20 minutes. Stroke. 1995;26: ; discussion Reich H, Angelos M, Safar P, Sterz F, Leonov Y. Cardiac resuscitability with cardiopulmonary bypass after increasing ventricular fibrillation times in dogs. Ann Emerg Med. 1990;19: Angelos M, Safar P, Reich H. A comparison of cardiopulmonary resuscitation with cardiopulmonary bypass after prolonged cardiac arrest in dogs: reperfusion pressures and neurologic recovery. Resuscitation. 1991;21: Safar P, Abramson NS, Angelos M, Cantadore R, Leonov Y, Levine R, Pretto E, Reich H, Sterz F, Stezoski SW. Emergency cardiopulmonary bypass for resuscitation from prolonged cardiac arrest. Am J Emerg Med. 1990;8: Behringer W, Safar P, Wu X, Kentner R, Radovsky A, Kochanek PM, Dixon CE, Tisherman SA. Survival without brain damage after clinical death of mins in dogs using suspended animation by profound hypothermia. Crit Care Med. 2003;31: Behringer W, Safar P, Wu X, Nozari A, Abdullah A, Stezoski SW, Tisherman SA. Veno-venous extracorporeal blood shunt cooling to induce mild hypothermia in dog experiments and review of cooling methods. Resuscitation. 2002;54: Ly HQ, Denault A, Dupuis J, Vadeboncoeur A, Harel F, Arsenault A, Gibson CM, Bonan R. A pilot study: the Noninvasive Surface Cooling Thermoregulatory System for Mild Hypothermia Induction in Acute Myocardial Infarction (the NICAMI Study). Am Heart J. 2005; 150:933.
7 2696 Circulation June 13, Boddicker KA, Zhang Y, Zimmerman MB, Davies LR, Kerber RE. Hypothermia improves defibrillation success and resuscitation outcomes from ventricular fibrillation. Circulation. 2005;111: Rhee BJ, Zhang Y, Boddicker KA, Davies LR, Kerber RE. Effect of hypothermia on transthoracic defibrillation in a swine model. Resuscitation. 2005;65: Bernard SA, Buist M, Monteiro O, Smith K. Induced hypothermia using large volume, ice-cold intravenous fluid in comatose survivors of out-of-hospital cardiac arrest: a preliminary report. Resuscitation. 2003;56: Nordmark J, Rubertsson S. Induction of mild hypothermia with infusion of cold (4 degrees C) fluid during ongoing experimental CPR. Resuscitation. 2005;66: Nozari A, Rubertsson S, Gedeborg R, Nordgren A, Wiklund L. Maximisation of cerebral blood flow during experimental cardiopulmonary resuscitation does not ameliorate post-resuscitation hypoperfusion. Resuscitation. 1999;40: Buja L. Myocardial ischemia and reperfusion injury. Cardiovasc Pathol. 2005;14: Ao H, Tanimoto H, Yoshitake A, Moon JK, Terasaki H. Long-term mild hypothermia with extracorporeal lung and heart assist improves survival from prolonged cardiac arrest in dogs. Resuscitation. 2001; 48: Hale SL, Kloner RA. Myocardial temperature in acute myocardial infarction: protection with mild regional hypothermia. Am J Physiol. 1997;273(pt 2):H220 H Wu X, Drabek T, Kochanek PM, Henchir J, Stezoski SW, Stezoski J, Cochran K, Garman R, Tisherman SA. Induction of profound hypothermia for emergency preservation and resuscitation allows intact survival from cardiac arrest resulting from prolonged lethal hemorrhage and trauma in dogs. Circulation. 2006;113: CLINICAL PERSPECTIVE Innovative clinical approaches are needed to improve the poor prognosis after cardiac arrest (CA). Mild therapeutic hypothermia is promising. Based on the evidence evaluation from the 2005 International Consensus Conference on Cardiopulmonary Resuscitation, the International Liaison Committee on Resuscitation recently recommended avoiding active rewarming of CA survivors and instead actively inducing mild hypothermia in hemodynamically stable comatose survivors. During circulatory arrest for cardiac surgery, protective hypothermia (induced before circulatory arrest) is considered the single most important measure to minimize ischemic brain injury. Similarly, CA victims could potentially benefit from an earlier induction of hypothermia during CA (intra-arrest cooling). In the present study, we show that there is a critical time window for intra-arrest hypothermic protection in a canine model of CA treated with cardiopulmonary resuscitation. Early (but not late) intra-arrest cooling by infusion of cooled saline reduced neurological injury and enabled intact survival from prolonged CA. These findings suggest that hypothermia initiated early during resuscitation may improve CA outcomes and merits clinical testing. In addition, when CA is resistant to standard resuscitative measures, early intra-arrest cooling may delay neurological injury sufficiently to warrant implementation of cardiopulmonary bypass or other mechanical support for more arrest victims.
Mild hypothermia during prolonged cardiopulmonary cerebral resuscitation increases conscious survival in dogs*
 Neurologic Critical Care Mild hypothermia during prolonged cardiopulmonary cerebral resuscitation increases conscious survival in dogs* Ala Nozari, MD, PhD; Peter Safar, MD, FCCM; S. William Stezoski;
Neurologic Critical Care Mild hypothermia during prolonged cardiopulmonary cerebral resuscitation increases conscious survival in dogs* Ala Nozari, MD, PhD; Peter Safar, MD, FCCM; S. William Stezoski;
Preventive Cardiology
 Preventive Cardiology Pilot Study of Rapid Infusion of 2 L of 4 C Normal Saline for Induction of Mild Hypothermia in Hospitalized, Comatose Survivors of Out-of-Hospital Cardiac Arrest Francis Kim, MD;
Preventive Cardiology Pilot Study of Rapid Infusion of 2 L of 4 C Normal Saline for Induction of Mild Hypothermia in Hospitalized, Comatose Survivors of Out-of-Hospital Cardiac Arrest Francis Kim, MD;
The Evidence Base. Stephan A. Mayer, MD. Columbia University New York, NY
 Hypothermic for Cardiac Arrest The Evidence Base Stephan A. Mayer, MD Director, Neuro-ICU Columbia University New York, NY Disclosures Columbia University Clinical Trials Pilot Award Radiant Medical, Inc.
Hypothermic for Cardiac Arrest The Evidence Base Stephan A. Mayer, MD Director, Neuro-ICU Columbia University New York, NY Disclosures Columbia University Clinical Trials Pilot Award Radiant Medical, Inc.
After resuscitation from cardiac arrest, brain injury is a
 Pilot Randomized Clinical Trial of Prehospital Induction of Mild Hypothermia in Out-of-Hospital Cardiac Arrest Patients With a Rapid Infusion of 4 C Normal Saline Francis Kim, MD; Michele Olsufka, RN;
Pilot Randomized Clinical Trial of Prehospital Induction of Mild Hypothermia in Out-of-Hospital Cardiac Arrest Patients With a Rapid Infusion of 4 C Normal Saline Francis Kim, MD; Michele Olsufka, RN;
Mild. Moderate. Severe. 32 to to and below
 Mohamud Daya MD, MS Mild 32 to 34 Moderate 28 to 32 Severe 28 and below Jon Rittenberger Shervin Ayati Protocol Development Committee Hypothermia Working Group Lynn Wittwer Jon Jui John Stouffer Scott
Mohamud Daya MD, MS Mild 32 to 34 Moderate 28 to 32 Severe 28 and below Jon Rittenberger Shervin Ayati Protocol Development Committee Hypothermia Working Group Lynn Wittwer Jon Jui John Stouffer Scott
Emergency Preservation and Resuscitation
 Emergency Preservation and Resuscitation Samuel A. Tisherman, MD, FACS, FCCM Director, Center for Critical Care and Trauma Education Director, SICU RA Cowley Shock Trauma Center Disclosures Co-author of
Emergency Preservation and Resuscitation Samuel A. Tisherman, MD, FACS, FCCM Director, Center for Critical Care and Trauma Education Director, SICU RA Cowley Shock Trauma Center Disclosures Co-author of
WORKSHEET for Evidence-Based Review of Science for Veterinary CPCR
 RECOVER 2011 1 of 11 WORKSHEET for Evidence-Based Review of Science for Veterinary CPCR 1. Basic Demographics Worksheet author(s) Shannon Axiak Date Submitted for review: 7.7.2011 Mailing address: Giessereiweg
RECOVER 2011 1 of 11 WORKSHEET for Evidence-Based Review of Science for Veterinary CPCR 1. Basic Demographics Worksheet author(s) Shannon Axiak Date Submitted for review: 7.7.2011 Mailing address: Giessereiweg
RACE CARS: Hospital Response. David A. Pearson, MD Department of Emergency Medicine Carolinas Medical Center February 23, 2012
 L MODULE 9 RACE CARS: Hospital Response David A. Pearson, MD Department of Emergency Medicine Carolinas Medical Center February 23, 2012 2 Objectives: Post-cardiac arrest syndrome Therapeutic hypothermia
L MODULE 9 RACE CARS: Hospital Response David A. Pearson, MD Department of Emergency Medicine Carolinas Medical Center February 23, 2012 2 Objectives: Post-cardiac arrest syndrome Therapeutic hypothermia
RESEARCH IN BASIC SCIENCE
 RESEARCH IN BASIC SCIENCE Effect of High-Dose Sodium Bicarbonate on the Vasopressor Effects of Epinephrine During Cardiopulmonary Resuscitation Barry E. Bleske, Pharm.D., Eric W Warren, Pharm.D., Ted L.
RESEARCH IN BASIC SCIENCE Effect of High-Dose Sodium Bicarbonate on the Vasopressor Effects of Epinephrine During Cardiopulmonary Resuscitation Barry E. Bleske, Pharm.D., Eric W Warren, Pharm.D., Ted L.
INDUCED HYPOTHERMIA. F. Ben Housel, M.D.
 INDUCED HYPOTHERMIA F. Ben Housel, M.D. Historical Use of Induced Hypothermia 1950 s - Moderate hypothermia (30-32º C) in open heart surgery to protect brain against global ischemia 1960-1980 s - Use of
INDUCED HYPOTHERMIA F. Ben Housel, M.D. Historical Use of Induced Hypothermia 1950 s - Moderate hypothermia (30-32º C) in open heart surgery to protect brain against global ischemia 1960-1980 s - Use of
Patient Case. Post cardiac arrest pathophysiology 10/19/2017. Disclosure. Objectives. Patient Case-TM
 Disclosure TARGETED TEMPERATURE MANAGEMENT POST CARDIAC ARREST I have nothing to disclose concerning possible financial or personal relationships with commercial entities that may have a direct or indirect
Disclosure TARGETED TEMPERATURE MANAGEMENT POST CARDIAC ARREST I have nothing to disclose concerning possible financial or personal relationships with commercial entities that may have a direct or indirect
Resuscitation Science : Advancing Care for the Sickest Patients
 Resuscitation Science : Advancing Care for the Sickest Patients William Hallinan University of Rochester What is resuscitation science? Simply the science of resuscitation : Pre arrest Arrest care Medical
Resuscitation Science : Advancing Care for the Sickest Patients William Hallinan University of Rochester What is resuscitation science? Simply the science of resuscitation : Pre arrest Arrest care Medical
Hypothermia: The Science and Recommendations (In-hospital and Out)
 Hypothermia: The Science and Recommendations (In-hospital and Out) L. Kristin Newby, MD, MHS Professor of Medicine Duke University Medical Center Chair, Council on Clinical Cardiology, AHA President, Society
Hypothermia: The Science and Recommendations (In-hospital and Out) L. Kristin Newby, MD, MHS Professor of Medicine Duke University Medical Center Chair, Council on Clinical Cardiology, AHA President, Society
WORKSHEET for Evidence-Based Review of Science for Veterinary CPCR
 RECOVER 2011 1 of 12 WORKSHEET for Evidence-Based Review of Science for Veterinary CPCR 1. Basic Demographics Worksheet author(s) Jane Quandt Mailing address: 501 DW Brooks Drive Small Animal Medicine
RECOVER 2011 1 of 12 WORKSHEET for Evidence-Based Review of Science for Veterinary CPCR 1. Basic Demographics Worksheet author(s) Jane Quandt Mailing address: 501 DW Brooks Drive Small Animal Medicine
New Therapeutic Hypothermia Techniques
 New Therapeutic Hypothermia Techniques Joseph P. Ornato, MD, FACP, FACC, FACEP Professor & Chairman, Emergency Medicine Virginia Commonwealth University Health System Richmond, VA Medical Director Richmond
New Therapeutic Hypothermia Techniques Joseph P. Ornato, MD, FACP, FACC, FACEP Professor & Chairman, Emergency Medicine Virginia Commonwealth University Health System Richmond, VA Medical Director Richmond
Advanced Resuscitation - Child
 C02C Resuscitation 2017-03-23 1 up to 10 years Office of the Medical Director Advanced Resuscitation - Child Intermediate Advanced Critical From PRIMARY ASSESSMENT Known or suspected hypothermia Algorithm
C02C Resuscitation 2017-03-23 1 up to 10 years Office of the Medical Director Advanced Resuscitation - Child Intermediate Advanced Critical From PRIMARY ASSESSMENT Known or suspected hypothermia Algorithm
Advanced Resuscitation - Adult
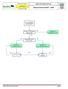 C02A Resuscitation 2017-03-23 17 years & older Office of the Medical Director Advanced Resuscitation - Adult Intermediate Advanced Critical From PRIMARY ASSESSMENT Known or suspected hypothermia Algorithm
C02A Resuscitation 2017-03-23 17 years & older Office of the Medical Director Advanced Resuscitation - Adult Intermediate Advanced Critical From PRIMARY ASSESSMENT Known or suspected hypothermia Algorithm
Outcomes of Therapeutic Hypothermia in Cardiac Arrest. Saad Mohammed Shariff, MBBS Aravind Herle, MD, FACC
 Outcomes of Therapeutic Hypothermia in Cardiac Arrest Saad Mohammed Shariff, MBBS Aravind Herle, MD, FACC https://my.americanheart.org/idc/groups/ahamah-public/@wcm/@sop/@scon/documents/downloadable/ucm_427331.pdf
Outcomes of Therapeutic Hypothermia in Cardiac Arrest Saad Mohammed Shariff, MBBS Aravind Herle, MD, FACC https://my.americanheart.org/idc/groups/ahamah-public/@wcm/@sop/@scon/documents/downloadable/ucm_427331.pdf
Update on Sudden Cardiac Death and Resuscitation
 Update on Sudden Cardiac Death and Resuscitation Ashish R. Panchal, MD, PhD Medical Director Center for Emergency Medical Services Assistant Professor Clinical Department of Emergency Medicine The Ohio
Update on Sudden Cardiac Death and Resuscitation Ashish R. Panchal, MD, PhD Medical Director Center for Emergency Medical Services Assistant Professor Clinical Department of Emergency Medicine The Ohio
Case Presentation. Cooling. Case Presentation. New Developments in Cardiopulmonary Arrest: Therapeutic Hypothermia in Resuscitation
 New Developments in Cardiopulmonary Arrest: Therapeutic Hypothermia in Resuscitation Michael Sayre, MD Emergency Medicine and LeRoy Essig, MD Pulmonary/Critical Care Medicine Case Presentation 3:40 (+
New Developments in Cardiopulmonary Arrest: Therapeutic Hypothermia in Resuscitation Michael Sayre, MD Emergency Medicine and LeRoy Essig, MD Pulmonary/Critical Care Medicine Case Presentation 3:40 (+
Frank Guyette, MD, MS, MPH Jon Rittenberger, MD, MSc Cliff Callaway, MD, PhD University of Pittsburgh Department of Emergency Medicine
 Frank Guyette, MD, MS, MPH Jon Rittenberger, MD, MSc Cliff Callaway, MD, PhD University of Pittsburgh Department of Emergency Medicine Disclosures Philips Healthcare: Faculty Learning Objectives Upon completion
Frank Guyette, MD, MS, MPH Jon Rittenberger, MD, MSc Cliff Callaway, MD, PhD University of Pittsburgh Department of Emergency Medicine Disclosures Philips Healthcare: Faculty Learning Objectives Upon completion
In-hospital Care of the Post-Cardiac Arrest Patient. David A. Pearson, MD, FACEP, FAAEM Associate Program Director Department of Emergency Medicine
 In-hospital Care of the Post-Cardiac Arrest Patient David A. Pearson, MD, FACEP, FAAEM Associate Program Director Department of Emergency Medicine Disclosures I have no financial interest, arrangement,
In-hospital Care of the Post-Cardiac Arrest Patient David A. Pearson, MD, FACEP, FAAEM Associate Program Director Department of Emergency Medicine Disclosures I have no financial interest, arrangement,
Advanced Resuscitation - Adolescent
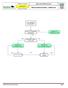 C02B Resuscitation 2017-03-23 10 up to 17 years Office of the Medical Director Advanced Resuscitation - Adolescent Intermediate Advanced Critical From PRIMARY ASSESSMENT Known or suspected hypothermia
C02B Resuscitation 2017-03-23 10 up to 17 years Office of the Medical Director Advanced Resuscitation - Adolescent Intermediate Advanced Critical From PRIMARY ASSESSMENT Known or suspected hypothermia
Post-Cardiac Arrest Syndrome. MICU Lecture Series
 Post-Cardiac Arrest Syndrome MICU Lecture Series Case 58 y/o female collapses at home, family attempts CPR, EMS arrives and notes VF, defibrillation x 3 with return of spontaneous circulation, brought
Post-Cardiac Arrest Syndrome MICU Lecture Series Case 58 y/o female collapses at home, family attempts CPR, EMS arrives and notes VF, defibrillation x 3 with return of spontaneous circulation, brought
Cardiovascular disease is a leading cause of premature
 Induction of Therapeutic Hypothermia by Paramedics After Resuscitation From Out-of-Hospital Ventricular Fibrillation Cardiac Arrest A Randomized Controlled Trial Stephen A. Bernard, MD; Karen Smith, BSc,
Induction of Therapeutic Hypothermia by Paramedics After Resuscitation From Out-of-Hospital Ventricular Fibrillation Cardiac Arrest A Randomized Controlled Trial Stephen A. Bernard, MD; Karen Smith, BSc,
CPR What Works, What Doesn t
 Resuscitation 2017 ECMO and ECLS April 1, 2017 Corey M. Slovis, M.D. Vanderbilt University Medical Center Metro Nashville Fire Department Nashville International Airport Nashville, TN Circulation 2013;128:417-35
Resuscitation 2017 ECMO and ECLS April 1, 2017 Corey M. Slovis, M.D. Vanderbilt University Medical Center Metro Nashville Fire Department Nashville International Airport Nashville, TN Circulation 2013;128:417-35
Peter Safar, MD, Drhc, FCCM, FCCP; Wilhelm Behringer, MD; Bernd W. Böttiger, MD, PhD; Fritz Sterz, MD, PhD
 Cerebral resuscitation potentials for cardiac arrest Peter Safar, MD, Drhc, FCCM, FCCP; Wilhelm Behringer, MD; Bernd W. Böttiger, MD, PhD; Fritz Sterz, MD, PhD Permanent brain damage after cardiac arrest
Cerebral resuscitation potentials for cardiac arrest Peter Safar, MD, Drhc, FCCM, FCCP; Wilhelm Behringer, MD; Bernd W. Böttiger, MD, PhD; Fritz Sterz, MD, PhD Permanent brain damage after cardiac arrest
Disclosures. Extra-Corporeal Membrane Oxygenation During Cardio- Pulmonary Resuscitation ECPR April 22, 2016 ECG. Case. Case. Case Summary 4/22/2016
 Extra-Corporeal Membrane Oxygenation During Cardio- Pulmonary Resuscitation ECPR April 22, 2016 Nothing to disclose. Disclosures Ivan J Chavez MD Case ECG History 60 y/o male No prior history of CAD In
Extra-Corporeal Membrane Oxygenation During Cardio- Pulmonary Resuscitation ECPR April 22, 2016 Nothing to disclose. Disclosures Ivan J Chavez MD Case ECG History 60 y/o male No prior history of CAD In
CHILL OUT! Induced Hypothermia: Challenges & Successes in the
 CHILL OUT! Induced Hypothermia: Challenges & Successes in the ICU Colleen Bell RN, BS, CCRN, Donna Brault RN, BSN, CCRN, Cathy Patnode RN, BSN, CCRN Champlain Valley Physician Hospital November 2012 Objectives
CHILL OUT! Induced Hypothermia: Challenges & Successes in the ICU Colleen Bell RN, BS, CCRN, Donna Brault RN, BSN, CCRN, Cathy Patnode RN, BSN, CCRN Champlain Valley Physician Hospital November 2012 Objectives
Beneficial Effect of Mild Hypothermia and Detrimental Effect of Deep Hypothermia After Cardiac Arrest in Dogs
 1454 Beneficial Effect of Mild Hypothermia and Detrimental Effect of Deep Hypothermia After Cardiac Arrest in Dogs Viktor Weinrauch, MD; Peter Safar, MD; Samuel Tisherman, MD; Kazutoshi Kuboyama, MD; and
1454 Beneficial Effect of Mild Hypothermia and Detrimental Effect of Deep Hypothermia After Cardiac Arrest in Dogs Viktor Weinrauch, MD; Peter Safar, MD; Samuel Tisherman, MD; Kazutoshi Kuboyama, MD; and
Protective head-cooling during cardiac arrest and cardiopulmonary resuscitation: the original animal studies
 Neurology International 2010; volume 2:e3 Protective head-cooling during cardiac arrest and cardiopulmonary resuscitation: the original animal studies Eric W. Brader, 1 Dietrich Jehle, 2 Michael Mineo,
Neurology International 2010; volume 2:e3 Protective head-cooling during cardiac arrest and cardiopulmonary resuscitation: the original animal studies Eric W. Brader, 1 Dietrich Jehle, 2 Michael Mineo,
Curricullum Vitae. Dr. Isman Firdaus, SpJP (K), FIHA
 Curricullum Vitae Dr. Isman Firdaus, SpJP (K), FIHA Email: ismanf@yahoo.com Qualification : o GP 2001 (FKUI) o Cardiologist 2007 (FKUI) o Cardiovascular Intensivist 2010 - present o Cardiovascular Intervensionist
Curricullum Vitae Dr. Isman Firdaus, SpJP (K), FIHA Email: ismanf@yahoo.com Qualification : o GP 2001 (FKUI) o Cardiologist 2007 (FKUI) o Cardiovascular Intensivist 2010 - present o Cardiovascular Intervensionist
Michigan Pediatric Cardiac Protocols. Date: November 15, 2012 Page 1 of 1 TABLE OF CONTENTS
 Date: November 15, 2012 Page 1 of 1 TABLE OF CONTENTS Pediatric Asystole Section 4-1 Pediatric Bradycardia Section 4-2 Pediatric Cardiac Arrest General Section 4-3 Pediatric Narrow Complex Tachycardia
Date: November 15, 2012 Page 1 of 1 TABLE OF CONTENTS Pediatric Asystole Section 4-1 Pediatric Bradycardia Section 4-2 Pediatric Cardiac Arrest General Section 4-3 Pediatric Narrow Complex Tachycardia
UTSW/BioTel EMS TRAINING BULLETIN January EMS TB Accidental Hypothermia
 UTSW/BioTel EMS TRAINING BULLETIN January 2015 EMS TB 15-001 Accidental Hypothermia Purpose: 1. To provide patient assessment and management guidance to UTSW/BioTel EMS Providers about Accidental Hypothermia
UTSW/BioTel EMS TRAINING BULLETIN January 2015 EMS TB 15-001 Accidental Hypothermia Purpose: 1. To provide patient assessment and management guidance to UTSW/BioTel EMS Providers about Accidental Hypothermia
Post-Arrest Care: Beyond Hypothermia
 Post-Arrest Care: Beyond Hypothermia Damon Scales MD PhD Department of Critical Care Medicine Sunnybrook Health Sciences Centre University of Toronto Disclosures CIHR Physicians Services Incorporated Main
Post-Arrest Care: Beyond Hypothermia Damon Scales MD PhD Department of Critical Care Medicine Sunnybrook Health Sciences Centre University of Toronto Disclosures CIHR Physicians Services Incorporated Main
Michigan Pediatric Cardiac Protocols. Date: November 15, 2012 Page 1 of 1 TABLE OF CONTENTS
 Date: November 15, 2012 Page 1 of 1 TABLE OF CONTENTS Pediatric Asystole Section 4-1 Pediatric Bradycardia Section 4-2 Pediatric Cardiac Arrest General Section 4-3 Pediatric Narrow Complex Tachycardia
Date: November 15, 2012 Page 1 of 1 TABLE OF CONTENTS Pediatric Asystole Section 4-1 Pediatric Bradycardia Section 4-2 Pediatric Cardiac Arrest General Section 4-3 Pediatric Narrow Complex Tachycardia
Cardiopulmonary Resuscitation in Adults
 Cardiopulmonary Resuscitation in Adults Fatma Özdemir, MD Emergency Deparment of Uludag University Faculty of Medicine OVERVIEW Introduction Pathophysiology BLS algorithm ALS algorithm Post resuscitation
Cardiopulmonary Resuscitation in Adults Fatma Özdemir, MD Emergency Deparment of Uludag University Faculty of Medicine OVERVIEW Introduction Pathophysiology BLS algorithm ALS algorithm Post resuscitation
Induced Hypothermia Following Out-of-Hospital Cardiac Arrest; Initial Experience in a Community Hospital
 Clin. Cardiol. 29, 525 529 (2006) Induced Hypothermia Following Out-of-Hospital Cardiac Arrest; Initial Experience in a Community Hospital Brook D. Scott, M.D., FACC, Tammy Hogue, R.N., M.S., C.C.N.S.,
Clin. Cardiol. 29, 525 529 (2006) Induced Hypothermia Following Out-of-Hospital Cardiac Arrest; Initial Experience in a Community Hospital Brook D. Scott, M.D., FACC, Tammy Hogue, R.N., M.S., C.C.N.S.,
Reperfusion Effects After Cardiac Ischemia
 Reperfusion Effects After Cardiac Ischemia Dave Milzman, MD, FACEP Professor and Assistant Dean for Clinical Research Georgetown University School of Medicine Research Director, Depts of Trauma and Emerg
Reperfusion Effects After Cardiac Ischemia Dave Milzman, MD, FACEP Professor and Assistant Dean for Clinical Research Georgetown University School of Medicine Research Director, Depts of Trauma and Emerg
Therapeutic hypothermia Transcutaneous pacing Sodium bicarbonate Rx Calcium, Magnesium Fluids and Pressors Antiarrhythmic Rx Epi/Vasopressin O 2
 Resuscitation Arsenal Therapeutic hypothermia Transcutaneous pacing Sodium bicarbonate Rx Calcium, Magnesium Fluids and Pressors Antiarrhythmic Rx Epi/Vasopressin O 2 /intubation Shock CPR ` 1994-96 Standing
Resuscitation Arsenal Therapeutic hypothermia Transcutaneous pacing Sodium bicarbonate Rx Calcium, Magnesium Fluids and Pressors Antiarrhythmic Rx Epi/Vasopressin O 2 /intubation Shock CPR ` 1994-96 Standing
Chapter 19 Detection of ROSC in Patients with Cardiac Arrest During Chest Compression Using NIRS: A Pilot Study
 Chapter 19 Detection of ROSC in Patients with Cardiac Arrest During Chest Compression Using NIRS: A Pilot Study Tsukasa Yagi, Ken Nagao, Tsuyoshi Kawamorita, Taketomo Soga, Mitsuru Ishii, Nobutaka Chiba,
Chapter 19 Detection of ROSC in Patients with Cardiac Arrest During Chest Compression Using NIRS: A Pilot Study Tsukasa Yagi, Ken Nagao, Tsuyoshi Kawamorita, Taketomo Soga, Mitsuru Ishii, Nobutaka Chiba,
ECG Changes in Patients Treated with Mild Hypothermia after Cardio-pulmonary Resuscitation for Out-of-hospital Cardiac Arrest
 ECG Changes in Patients Treated with Mild Hypothermia after Cardio-pulmonary Resuscitation for Out-of-hospital Cardiac Arrest R. Schneider, S. Zimmermann, W.G. Daniel, S. Achenbach Department of Internal
ECG Changes in Patients Treated with Mild Hypothermia after Cardio-pulmonary Resuscitation for Out-of-hospital Cardiac Arrest R. Schneider, S. Zimmermann, W.G. Daniel, S. Achenbach Department of Internal
The ABC of CAB- Circulation, Airway, Breathing: PALS/Resuscitation Update
 The ABC of CAB- Circulation, Airway, Breathing: PALS/Resuscitation Update Jennifer K. Lee, MD Johns Hopkins University Dept. of Anesthesia, Division of Pediatric Anesthesia Disclosures I have research
The ABC of CAB- Circulation, Airway, Breathing: PALS/Resuscitation Update Jennifer K. Lee, MD Johns Hopkins University Dept. of Anesthesia, Division of Pediatric Anesthesia Disclosures I have research
EXTRACORPOREAL LIFE SUPPORT FOR PROLONGED CARDIAC ARREST
 EXTRACORPOREAL LIFE SUPPORT FOR PROLONGED CARDIAC ARREST Massimo MASSETTI Cardiac Surgery Department University of Sacred Heart, Gemelli Hospital Rome «No financial interest to declare» ESC CONGRESS 2012
EXTRACORPOREAL LIFE SUPPORT FOR PROLONGED CARDIAC ARREST Massimo MASSETTI Cardiac Surgery Department University of Sacred Heart, Gemelli Hospital Rome «No financial interest to declare» ESC CONGRESS 2012
CrackCast Episode 8 Brain Resuscitation
 CrackCast Episode 8 Brain Resuscitation Episode Overview: 1) Describe 6 therapeutic interventions for the post-arrest brain 2) List 5 techniques for initiating therapeutic hypothermia 3) List 4 mechanisms
CrackCast Episode 8 Brain Resuscitation Episode Overview: 1) Describe 6 therapeutic interventions for the post-arrest brain 2) List 5 techniques for initiating therapeutic hypothermia 3) List 4 mechanisms
Management of Cardiac Arrest Based on : 2010 American Heart Association Guidelines
 Management of Cardiac Arrest Based on : 2010 American Heart Association Guidelines www.circ.ahajournals.org Elham Pishbin. M.D Assistant Professor of Emergency Medicine MUMS C H E S Advanced Life Support
Management of Cardiac Arrest Based on : 2010 American Heart Association Guidelines www.circ.ahajournals.org Elham Pishbin. M.D Assistant Professor of Emergency Medicine MUMS C H E S Advanced Life Support
ECMO CPR. Ravi R. Thiagarajan MBBS, MPH. Cardiac Intensive Care Unit
 ECMO CPR Ravi R. Thiagarajan MBBS, MPH Staff Intensivist Cardiac Intensive Care Unit Children s Hospital Boston PCICS 2008, Miami, FL No disclosures Disclosures Outline Outcomes for Pediatric in-hospital
ECMO CPR Ravi R. Thiagarajan MBBS, MPH Staff Intensivist Cardiac Intensive Care Unit Children s Hospital Boston PCICS 2008, Miami, FL No disclosures Disclosures Outline Outcomes for Pediatric in-hospital
MICHIGAN. State Protocols. Pediatric Cardiac Table of Contents 6.1 General Pediatric Cardiac Arrest 6.2 Bradycardia 6.
 MICHIGAN State Protocols Protocol Number Protocol Name Pediatric Cardiac Table of Contents 6.1 General Pediatric Cardiac Arrest 6.2 Bradycardia 6.3 Tachycardia PEDIATRIC CARDIAC PEDIATRIC CARDIAC ARREST
MICHIGAN State Protocols Protocol Number Protocol Name Pediatric Cardiac Table of Contents 6.1 General Pediatric Cardiac Arrest 6.2 Bradycardia 6.3 Tachycardia PEDIATRIC CARDIAC PEDIATRIC CARDIAC ARREST
Neurologic Recovery Following Prolonged Out-of-Hospital Cardiac Arrest With Resuscitation Guided by Continuous Capnography
 CASE REPORT FULL RECOVERY AFTER PROLONGED CARDIAC ARREST AND RESUSCITATION WITH CAPNOGRAPHY GUIDANCE Neurologic Recovery Following Prolonged Out-of-Hospital Cardiac Arrest With Resuscitation Guided by
CASE REPORT FULL RECOVERY AFTER PROLONGED CARDIAC ARREST AND RESUSCITATION WITH CAPNOGRAPHY GUIDANCE Neurologic Recovery Following Prolonged Out-of-Hospital Cardiac Arrest With Resuscitation Guided by
ANZCOR Guideline 11.1 Introduction to Advanced Life Support
 ANZCOR Guideline 11.1 Introduction to Advanced Life Support Who does this guideline apply to? Summary This guideline applies to adults who require advanced life support. Who is the audience for this guideline?
ANZCOR Guideline 11.1 Introduction to Advanced Life Support Who does this guideline apply to? Summary This guideline applies to adults who require advanced life support. Who is the audience for this guideline?
Hypertension With or Without Hemodilution After Cardiac Arrest in Dogs
 1178 Hypertension With or Without Hemodilution After Cardiac Arrest in Dogs Fritz Sterz, MD, Yuval Leonov, MD, Peter Safar, MD, Ann Radovsky, DVM, PhD, Samuel A. Tisherman, MD, and Ken-ichi Oku, MD We
1178 Hypertension With or Without Hemodilution After Cardiac Arrest in Dogs Fritz Sterz, MD, Yuval Leonov, MD, Peter Safar, MD, Ann Radovsky, DVM, PhD, Samuel A. Tisherman, MD, and Ken-ichi Oku, MD We
Update on Sudden Cardiac Death and Resuscitation
 Update on Sudden Cardiac Death and Resuscitation Ashish R. Panchal, MD, PhD Medical Director Center for Emergency Medical Services Assistant Professor Clinical Department of Emergency Medicine The Ohio
Update on Sudden Cardiac Death and Resuscitation Ashish R. Panchal, MD, PhD Medical Director Center for Emergency Medical Services Assistant Professor Clinical Department of Emergency Medicine The Ohio
Epinephrine Cardiovascular Emergencies Symposium 2018
 Epinephrine Cardiovascular Emergencies Symposium 218 Corey M. Slovis, M.D. Vanderbilt University Medical Center Metro Nashville Fire Department Nashville International Airport Nashville, TN High Quality
Epinephrine Cardiovascular Emergencies Symposium 218 Corey M. Slovis, M.D. Vanderbilt University Medical Center Metro Nashville Fire Department Nashville International Airport Nashville, TN High Quality
Ameliorating Reperfusion Injury During Resuscitation from Cardiac Arrest
 Ameliorating Reperfusion Injury During Resuscitation from Cardiac Arrest Scott T. Youngquist, MD, MSc Associate Professor, Emergency Medicine University of Utah School of Medicine Medical Director, Salt
Ameliorating Reperfusion Injury During Resuscitation from Cardiac Arrest Scott T. Youngquist, MD, MSc Associate Professor, Emergency Medicine University of Utah School of Medicine Medical Director, Salt
WORKSHEET for Evidence-Based Review of Science for Veterinary CPCR
 RECOVER 2011 1 of 7 WORKSHEET for Evidence-Based Review of Science for Veterinary CPCR 1. Basic Demographics Worksheet author(s) Kate Hopper Mailing address: Dept Vet Surgical & Radiological Sciences Room
RECOVER 2011 1 of 7 WORKSHEET for Evidence-Based Review of Science for Veterinary CPCR 1. Basic Demographics Worksheet author(s) Kate Hopper Mailing address: Dept Vet Surgical & Radiological Sciences Room
Student Contribution THE USE OF VASOPRESSIN IN THE TREATMENT OF CARDIAC ARREST A CASE STUDY. Aaron Turner, SA Ambulance Service
 ISSN 1447-4999 Student Contribution THE USE OF VASOPRESSIN IN THE TREATMENT OF CARDIAC ARREST A CASE STUDY Aaron Turner, SA Ambulance Service INTRODUCTION Despite widespread use, current methods of treatment
ISSN 1447-4999 Student Contribution THE USE OF VASOPRESSIN IN THE TREATMENT OF CARDIAC ARREST A CASE STUDY Aaron Turner, SA Ambulance Service INTRODUCTION Despite widespread use, current methods of treatment
Post Resuscitation (ROSC) Care
 Standard Operating Procedure 2.10 Post Resuscitation (ROSC) Care Position Responsible: Medical Director Approved: Clinical Governance Committee Related Documents: This document is the intellectual property
Standard Operating Procedure 2.10 Post Resuscitation (ROSC) Care Position Responsible: Medical Director Approved: Clinical Governance Committee Related Documents: This document is the intellectual property
PALS NEW GUIDELINES 2010
 PALS NEW GUIDELINES 2010 DR WALEED ALAMRI PEDIATRIC EMERGENCY CONSULTANT FEB 24, 2011 Pediatric Basic Life Support Change in CPR Sequence (C-A-B Rather Than A-B-C) 2010 (New): Initiate CPR for infants
PALS NEW GUIDELINES 2010 DR WALEED ALAMRI PEDIATRIC EMERGENCY CONSULTANT FEB 24, 2011 Pediatric Basic Life Support Change in CPR Sequence (C-A-B Rather Than A-B-C) 2010 (New): Initiate CPR for infants
DELINEATION OF CLINICAL PRIVILEGES SURGERY - THORACIC AND CARDIOVASCULAR SURGERY
 Basic Education: MD or DO (Applicants must meet the following criteria) Be certified by or be currently qualified to take the board certification examination of a board recognized by the American Board
Basic Education: MD or DO (Applicants must meet the following criteria) Be certified by or be currently qualified to take the board certification examination of a board recognized by the American Board
State of the art lecture: 21st Century Post resuscitation management
 State of the art lecture: 21st Century Post resuscitation management ACCA Masterclass 2017 Prof Alain CARIOU Intensive Care Unit - Cochin Hospital (APHP) Paris Descartes University INSERM U970 - France
State of the art lecture: 21st Century Post resuscitation management ACCA Masterclass 2017 Prof Alain CARIOU Intensive Care Unit - Cochin Hospital (APHP) Paris Descartes University INSERM U970 - France
Johnson County Emergency Medical Services Page 23
 Non-resuscitation Situations: Resuscitation should not be initiated in the following situations: Prolonged arrest as evidenced by lividity in dependent parts, rigor mortis, tissue decomposition, or generalized
Non-resuscitation Situations: Resuscitation should not be initiated in the following situations: Prolonged arrest as evidenced by lividity in dependent parts, rigor mortis, tissue decomposition, or generalized
2015 AHA Guidelines: Pediatric Updates
 2015 AHA Guidelines: Pediatric Updates Advances in Pediatric Emergency Medicine December 9, 2016 Karen O Connell, MD, MEd Associate Professor of Pediatrics and Emergency Medicine Emergency Medicine and
2015 AHA Guidelines: Pediatric Updates Advances in Pediatric Emergency Medicine December 9, 2016 Karen O Connell, MD, MEd Associate Professor of Pediatrics and Emergency Medicine Emergency Medicine and
Out-of-hospital Cardiac Arrest. Franz R. Eberli MD, FESC, FAHA Cardiology Triemli Hospital Zurich, Switzerland
 Out-of-hospital Cardiac Arrest Franz R. Eberli MD, FESC, FAHA Cardiology Triemli Hospital Zurich, Switzerland Conflict of Interest I have no conflict of interest to disclose regarding this presentation.
Out-of-hospital Cardiac Arrest Franz R. Eberli MD, FESC, FAHA Cardiology Triemli Hospital Zurich, Switzerland Conflict of Interest I have no conflict of interest to disclose regarding this presentation.
Update of CPR AHA Guidelines
 Update of CPR AHA Guidelines Donald Hal Shaffner Course objective is to have an updated understanding of the American Heart Association s treatment algorithms for the management of cardiac decompensation
Update of CPR AHA Guidelines Donald Hal Shaffner Course objective is to have an updated understanding of the American Heart Association s treatment algorithms for the management of cardiac decompensation
3/6/2017. Endovascular Selective Cerebral Hypothermia First-in-Human Experience
 Endovascular Selective Cerebral Hypothermia First-in-Human Experience Ronald Jay Solar, Ph.D. San Diego, CA 32 nd Annual Snowmass Symposium March 5-10, 2017 Introduction Major limitations in acute ischemic
Endovascular Selective Cerebral Hypothermia First-in-Human Experience Ronald Jay Solar, Ph.D. San Diego, CA 32 nd Annual Snowmass Symposium March 5-10, 2017 Introduction Major limitations in acute ischemic
Advance Publication by J-STAGE
 Circulation Journal Official Journal of the Japanese Circulation Society http://www.j-circ.or.jp Early Induction of Hypothermia During Cardiac Arrest Improves Neurological Outcomes in Patients With Out-of-Hospital
Circulation Journal Official Journal of the Japanese Circulation Society http://www.j-circ.or.jp Early Induction of Hypothermia During Cardiac Arrest Improves Neurological Outcomes in Patients With Out-of-Hospital
Post-Resuscitation Care. Prof. Wilhelm Behringer Center of Emergency Medicine University of Jena
 Post-Resuscitation Care Prof. Wilhelm Behringer Center of Emergency Medicine University of Jena Conflict of interest Emcools Shareholder and founder, honoraria Zoll: honoraria Bard: honoraria, nephew works
Post-Resuscitation Care Prof. Wilhelm Behringer Center of Emergency Medicine University of Jena Conflict of interest Emcools Shareholder and founder, honoraria Zoll: honoraria Bard: honoraria, nephew works
Patient Management Code Blue in the CT Suite
 Patient Management Code Blue in the CT Suite David Stultz, MD November 28, 2001 Case Presentation A 53-year-old woman experienced acute respiratory distress during an IV contrast enhanced CT scan of the
Patient Management Code Blue in the CT Suite David Stultz, MD November 28, 2001 Case Presentation A 53-year-old woman experienced acute respiratory distress during an IV contrast enhanced CT scan of the
The ARREST Trial: Amiodarone for Resuscitation After Out-of-Hospital Cardiac Arrest Due to Ventricular Fibrillation
 The ARREST Trial: Amiodarone for Resuscitation After Out-of-Hospital Cardiac Arrest Due to Ventricular Fibrillation Introduction The ARREST (Amiodarone in out-of-hospital Resuscitation of REfractory Sustained
The ARREST Trial: Amiodarone for Resuscitation After Out-of-Hospital Cardiac Arrest Due to Ventricular Fibrillation Introduction The ARREST (Amiodarone in out-of-hospital Resuscitation of REfractory Sustained
The ALS Algorithm and Post Resuscitation Care
 The ALS Algorithm and Post Resuscitation Care CET - Ballarat Health Services Valid from 1 st July 2018 to 30 th June 2020 2 Defibrillation Produces simultaneous mass depolarisation of myocardial cells
The ALS Algorithm and Post Resuscitation Care CET - Ballarat Health Services Valid from 1 st July 2018 to 30 th June 2020 2 Defibrillation Produces simultaneous mass depolarisation of myocardial cells
Developments in Cardiopulmonary Resuscitation Guidelines
 Developments in Cardiopulmonary Resuscitation Guidelines Bernd W. Böttiger Seite 1 To preserve human life by making high quality resuscitation available to all Outcome after CPR in Germany ROSC ( Return
Developments in Cardiopulmonary Resuscitation Guidelines Bernd W. Böttiger Seite 1 To preserve human life by making high quality resuscitation available to all Outcome after CPR in Germany ROSC ( Return
In the past decade, two large randomized
 Mild therapeutic hypothermia improves outcomes compared with normothermia in cardiac-arrest patients a retrospective chart review* David Hörburger, MD; Christoph Testori, MD; Fritz Sterz, MD; Harald Herkner,
Mild therapeutic hypothermia improves outcomes compared with normothermia in cardiac-arrest patients a retrospective chart review* David Hörburger, MD; Christoph Testori, MD; Fritz Sterz, MD; Harald Herkner,
Therapeutic hypothermia following cardiac arrest
 TITLE: Therapeutic hypothermia following cardiac arrest AUTHOR: Jeffrey A. Tice, MD Assistant Professor of Medicine Division of General Internal Medicine Department of Medicine University of California
TITLE: Therapeutic hypothermia following cardiac arrest AUTHOR: Jeffrey A. Tice, MD Assistant Professor of Medicine Division of General Internal Medicine Department of Medicine University of California
Accidental Hypothermia
 Accidental Hypothermia Doug Brown, MD, FRCPC Emergency Physician Royal Columbian & Eagle Ridge Hospital October, 2013 Objectives 45min lecture, 15min discussion & questions: Inspirational case & overview
Accidental Hypothermia Doug Brown, MD, FRCPC Emergency Physician Royal Columbian & Eagle Ridge Hospital October, 2013 Objectives 45min lecture, 15min discussion & questions: Inspirational case & overview
Therapeutic Hypothermia
 Objectives Overview Therapeutic Hypothermia Nerissa U. Ko, MD, MAS UCSF Department of Neurology Critical Care Medicine and Trauma June 4, 2011 Hypothermia as a neuroprotectant Proven indications: Adult
Objectives Overview Therapeutic Hypothermia Nerissa U. Ko, MD, MAS UCSF Department of Neurology Critical Care Medicine and Trauma June 4, 2011 Hypothermia as a neuroprotectant Proven indications: Adult
ACLS/ACS Updates 2015
 ACLS/ACS Updates 2015 Advanced Cardiovascular Life Support by: Fareed Al Nozha, JBIM, ABIM, FKFSH&RC(Cardiology) Consultant Cardiologist Faculty, National CPR Committee, ACLS Program Head, SHA Dr Abdulhalim
ACLS/ACS Updates 2015 Advanced Cardiovascular Life Support by: Fareed Al Nozha, JBIM, ABIM, FKFSH&RC(Cardiology) Consultant Cardiologist Faculty, National CPR Committee, ACLS Program Head, SHA Dr Abdulhalim
Hypothermia After Cardiac Arrest: Where Are We Now?
 Hypothermia After Cardiac Arrest: Where Are We Now? David A. Pearson, MD, MS Associate Professor Director of Cardiac Arrest Resuscitation Carolinas HealthCare System Disclosures I have no financial interest,
Hypothermia After Cardiac Arrest: Where Are We Now? David A. Pearson, MD, MS Associate Professor Director of Cardiac Arrest Resuscitation Carolinas HealthCare System Disclosures I have no financial interest,
2015 Interim Training Materials
 2015 Interim Training Materials ACLS Manual and ACLS EP Manual Comparison Chart Assessment sequence Manual, Part 2: The Systematic Approach, and Part BLS Changes The HCP should check for response while
2015 Interim Training Materials ACLS Manual and ACLS EP Manual Comparison Chart Assessment sequence Manual, Part 2: The Systematic Approach, and Part BLS Changes The HCP should check for response while
Prehospital Resuscitation for the 21 st Century Simulation Case. VF/Asystole
 Prehospital Resuscitation for the 21 st Century Simulation Case VF/Asystole Case History 1 (hypovolemic cardiac arrest secondary to massive upper GI bleed) 56 year-old male patient who fainted in the presence
Prehospital Resuscitation for the 21 st Century Simulation Case VF/Asystole Case History 1 (hypovolemic cardiac arrest secondary to massive upper GI bleed) 56 year-old male patient who fainted in the presence
Hanna K. Al-Makhamreh, M.D., FACC Interventional Cardiologist
 Hanna K. Al-Makhamreh, M.D., FACC Interventional Cardiologist Introduction. Basic Life Support (BLS). Advanced Cardiac Life Support (ACLS). Cardiovascular diseases (CVDs) are the number one cause of death
Hanna K. Al-Makhamreh, M.D., FACC Interventional Cardiologist Introduction. Basic Life Support (BLS). Advanced Cardiac Life Support (ACLS). Cardiovascular diseases (CVDs) are the number one cause of death
Don t let your patients turn blue! Isn t it about time you used etco 2?
 Don t let your patients turn blue! Isn t it about time you used etco 2? American Association of Critical Care Nurses National Teaching Institute Expo Ed 2013 Susan Thibeault MS, CRNA, APRN, CCRN, EMT-P
Don t let your patients turn blue! Isn t it about time you used etco 2? American Association of Critical Care Nurses National Teaching Institute Expo Ed 2013 Susan Thibeault MS, CRNA, APRN, CCRN, EMT-P
AllinaHealthSystem 1
 : Definition End-organ hypoperfusion secondary to cardiac failure Venoarterial ECMO: Patient Selection Michael A. Samara, MD FACC Advanced Heart Failure, Cardiac Transplant & Mechanical Circulatory Support
: Definition End-organ hypoperfusion secondary to cardiac failure Venoarterial ECMO: Patient Selection Michael A. Samara, MD FACC Advanced Heart Failure, Cardiac Transplant & Mechanical Circulatory Support
Nassau Regional Emergency Medical Services. Advanced Life Support Pediatric Protocol Manual
 Nassau Regional Emergency Medical Services Advanced Life Support Pediatric Protocol Manual 2014 PEDIATRIC ADVANCED LIFE SUPPORT PROTOCOLS TABLE OF CONTENTS Approved Effective Newborn Resuscitation P 1
Nassau Regional Emergency Medical Services Advanced Life Support Pediatric Protocol Manual 2014 PEDIATRIC ADVANCED LIFE SUPPORT PROTOCOLS TABLE OF CONTENTS Approved Effective Newborn Resuscitation P 1
Targeted temperature management after post-anoxic brain insult: where do we stand?
 Targeted temperature management after post-anoxic brain insult: where do we stand? Alain Cariou Intensive Care Unit Cochin University Hospital Paris Descartes University INSERM U970 (France) COI Disclosure
Targeted temperature management after post-anoxic brain insult: where do we stand? Alain Cariou Intensive Care Unit Cochin University Hospital Paris Descartes University INSERM U970 (France) COI Disclosure
Tina Yoo, PharmD Clinical Pharmacist Alameda Health System Highland Hospital
 Tina Yoo, PharmD Clinical Pharmacist Alameda Health System Highland Hospital 1 Review changes in the 2015 AHA ACLS guidelines with emphasis on changes in therapeutic hypothermia Provide overview of ACLS
Tina Yoo, PharmD Clinical Pharmacist Alameda Health System Highland Hospital 1 Review changes in the 2015 AHA ACLS guidelines with emphasis on changes in therapeutic hypothermia Provide overview of ACLS
Big Chill in the Big Apple: Why FDNY is Not Getting the Cold Shoulder
 Big Chill in the Big Apple: Why FDNY is Not Getting the Cold Shoulder John Freese, MD Medical Director of Training Director of Prehospital Research OLMC Medical Director New York City Fire Department Complexities
Big Chill in the Big Apple: Why FDNY is Not Getting the Cold Shoulder John Freese, MD Medical Director of Training Director of Prehospital Research OLMC Medical Director New York City Fire Department Complexities
SURVIVAL after out-of-hospital cardiac arrest (OH-
 Acta Anaesthesiol Scand 2007; 51: 137 142 Printed in Singapore. All rights reserved # 2006 The Authors Journal compilation # 2006 Acta Anaesthesiol Scand ACTA ANAESTHESIOLOGICA SCANDINAVICA doi: 10.1111/j.1399-6576.2006.01209.x
Acta Anaesthesiol Scand 2007; 51: 137 142 Printed in Singapore. All rights reserved # 2006 The Authors Journal compilation # 2006 Acta Anaesthesiol Scand ACTA ANAESTHESIOLOGICA SCANDINAVICA doi: 10.1111/j.1399-6576.2006.01209.x
Use of an Inspiratory Impedance Valve Improves Neurologically Intact Survival in a Porcine Model of Ventricular Fibrillation
 Use of an Inspiratory Impedance Valve Improves Neurologically Intact Survival in a Porcine Model of Ventricular Fibrillation Keith G. Lurie, MD; Todd Zielinski, MS; Scott McKnite, BS; Tom Aufderheide,
Use of an Inspiratory Impedance Valve Improves Neurologically Intact Survival in a Porcine Model of Ventricular Fibrillation Keith G. Lurie, MD; Todd Zielinski, MS; Scott McKnite, BS; Tom Aufderheide,
ACLS Review. Pulse Oximetry to be between 94 99% to avoid hyperoxia (high oxygen tension can lead to tissue death
 ACLS Review BLS CPR BLS CPR changed in 2010. The primary change is from the ABC format to CAB. After establishing unresponsiveness and calling for a code, check for a pulse less than 10 seconds then begin
ACLS Review BLS CPR BLS CPR changed in 2010. The primary change is from the ABC format to CAB. After establishing unresponsiveness and calling for a code, check for a pulse less than 10 seconds then begin
TOO COOL OR NOT TOO COOL- THERAPEUTIC HYPOTHERMIA IN THE ICU SCCM TX 2017 TED WU MD PEDIATRIC CRITICAL CARE UNIVERSITY OF TEXAS HEALTH SAN ANTONIO
 TOO COOL OR NOT TOO COOL- THERAPEUTIC HYPOTHERMIA IN THE ICU SCCM TX 2017 TED WU MD PEDIATRIC CRITICAL CARE UNIVERSITY OF TEXAS HEALTH SAN ANTONIO DISCLOSURE I have no relationships with commercial companies
TOO COOL OR NOT TOO COOL- THERAPEUTIC HYPOTHERMIA IN THE ICU SCCM TX 2017 TED WU MD PEDIATRIC CRITICAL CARE UNIVERSITY OF TEXAS HEALTH SAN ANTONIO DISCLOSURE I have no relationships with commercial companies
Post Cardiac Arrest Care. From : 2010 American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care
 Post Cardiac Arrest Care From : 2010 American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care Initial Objectives of Post cardiac Arrest Care Optimize cardiopulmonary
Post Cardiac Arrest Care From : 2010 American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care Initial Objectives of Post cardiac Arrest Care Optimize cardiopulmonary
Use of Blood Lactate Measurements in the Critical Care Setting
 Use of Blood Lactate Measurements in the Critical Care Setting John G Toffaletti, PhD Director of Blood Gas and Clinical Pediatric Labs Professor of Pathology Duke University Medical Center Chief, VAMC
Use of Blood Lactate Measurements in the Critical Care Setting John G Toffaletti, PhD Director of Blood Gas and Clinical Pediatric Labs Professor of Pathology Duke University Medical Center Chief, VAMC
The Hypotensive Poisoned Patient. Robert S. Hoffman, MD Director, NYC PCC
 The Hypotensive Poisoned Patient Robert S. Hoffman, MD Director, NYC PCC Some Definitions Hypotension = Low blood pressure Failure of macrocirculation Shock = Poor tissue perfusion Failure of microcirculation
The Hypotensive Poisoned Patient Robert S. Hoffman, MD Director, NYC PCC Some Definitions Hypotension = Low blood pressure Failure of macrocirculation Shock = Poor tissue perfusion Failure of microcirculation
Index. Note: Page numbers of article titles are in boldface type.
 Index Note: Page numbers of article titles are in boldface type. A Acute coronary syndrome(s), anticoagulant therapy in, 706, 707 antiplatelet therapy in, 702 ß-blockers in, 703 cardiac biomarkers in,
Index Note: Page numbers of article titles are in boldface type. A Acute coronary syndrome(s), anticoagulant therapy in, 706, 707 antiplatelet therapy in, 702 ß-blockers in, 703 cardiac biomarkers in,
INDUCED HYPOTHERMIA A Hot Topic. R. Darrell Nelson, MD, FACEP Emergency Medicine Wake Forest University Health Sciences
 INDUCED HYPOTHERMIA A Hot Topic R. Darrell Nelson, MD, FACEP Emergency Medicine Wake Forest University Health Sciences Conflicts of Interest Sadly, we have no financial or industrial conflicts of interest
INDUCED HYPOTHERMIA A Hot Topic R. Darrell Nelson, MD, FACEP Emergency Medicine Wake Forest University Health Sciences Conflicts of Interest Sadly, we have no financial or industrial conflicts of interest
Post Cardiac Arrest Care 2015 American Heart Association Guideline Update for CPR and Emergency Cardiovascular Care
 Post Cardiac Arrest Care 2015 American Heart Association Guideline Update for CPR and Emergency Cardiovascular Care รศ.ดร.พญ.ต นหยง พ พานเมฆาภรณ ภาคว ชาว ส ญญ ว ทยา คณะแพทยศาสตร มหาว ทยาล ยเช ยงใหม System
Post Cardiac Arrest Care 2015 American Heart Association Guideline Update for CPR and Emergency Cardiovascular Care รศ.ดร.พญ.ต นหยง พ พานเมฆาภรณ ภาคว ชาว ส ญญ ว ทยา คณะแพทยศาสตร มหาว ทยาล ยเช ยงใหม System
ECMO Primer A View to the Future
 ECMO Primer A View to the Future Todd J. Kilbaugh Assistant Professor of Anesthesiology, Critical Care Medicine, and Pediatrics Director of The ECMO Center at the Children s Hospital of Philadelphia Disclosures
ECMO Primer A View to the Future Todd J. Kilbaugh Assistant Professor of Anesthesiology, Critical Care Medicine, and Pediatrics Director of The ECMO Center at the Children s Hospital of Philadelphia Disclosures
Stayin Alive: Pediatric Advanced Life Support (PALS) Updated Guidelines
 Stayin Alive: Pediatric Advanced Life Support (PALS) Updated Guidelines Margaret Oates, PharmD, BCPPS Pediatric Critical Care Specialist GSHP Summer Meeting July 16, 2016 Disclosures I have nothing to
Stayin Alive: Pediatric Advanced Life Support (PALS) Updated Guidelines Margaret Oates, PharmD, BCPPS Pediatric Critical Care Specialist GSHP Summer Meeting July 16, 2016 Disclosures I have nothing to
Advanced Cardiac Life Support (ACLS) Science Update 2015
 1 2 3 4 5 6 7 8 9 Advanced Cardiac Life Support (ACLS) Science Update 2015 What s New in ACLS for 2015? Adult CPR CPR remains (Compressions, Airway, Breathing Chest compressions has priority over all other
1 2 3 4 5 6 7 8 9 Advanced Cardiac Life Support (ACLS) Science Update 2015 What s New in ACLS for 2015? Adult CPR CPR remains (Compressions, Airway, Breathing Chest compressions has priority over all other
