Drug-induced cardiotoxicity: screening methods, regulatory context, commercial environment. Beremans Ltd / Insight Pharma Reports April 2008
|
|
|
- Theodora Webster
- 6 years ago
- Views:
Transcription
1 Drug-induced cardiotoxicity: screening methods, regulatory context, commercial environment. Overview Beremans Ltd / Insight Pharma Reports April 2008 This report identifies and discusses methods, products and services designed to identify cardiotoxic compounds before they reach the market; discusses the current and possible future regulatory environment; outlines main commercial competitors; and suggests broad types of commercial opportunity and future M&A activity in this subsector. The primary focus is on identification of proarrhythmia, but some discussion is provided on identification of other forms of cardiotoxicity. We start by providing a primer on cardiac anatomy / physiology. Particular consideration is given to the various ion fluxes that contribute to the cardiac action potential. Next, drug-induced cardiotoxicity is discussed under two headings: direct cardiotoxicity (i.e. where the primary symptom of cardiotoxicity is not arrhythmia) and proarrhythmic cardiotoxicity. In each category, we provide extensive lists of drugs associated with cardiotoxicity, discuss factors which may predispose to druginduced cardiotoxicity, and outline current / proposed cardioprotective approaches. We then outline the history and status quo of the current regulatory environment pertinent to drug-induced proarrhythmia, with particular reference to guidelines S7B and E14. In addition, we discuss some of the main factors which may impact the regulators decisions regarding drug non-approval due to cardiotoxicity. Next, we provide a detailed discussion on methods for assessing the potential for drug-induced cardiotoxicity, with a primary focus on proarrhythmia screening. Various recommended and proposed surrogates for torsades de pointes (TdP) risk are compared. Preclinical and clinical proarrhythmia screens are discussed, including in silico methods, in vitro single cell / multicellular methods, in vivo methods and early clinical trial methods. Some discussion also is provided on mechanisms of screening for direct (non-proarrhythmic) cardiotoxicity. In addition, we provide and discuss the results of an industry survey undertaken for this report by online questionnaire and by one-to-one interviews with key experts. Questions were aimed at, inter alia, gathering information on attitudes to preclinical models and surrogates for TdP risk, and opinions as to how and when current guidelines might change. Results from previous industry surveys also are noted.
2 An overview of 50 commercial entities offering cardiotoxicity screening products / services is provided. Further analysis is included regarding the competitive positioning and ownership of 29 companies that have some clear cardiotoxicity screening focus. Broad consolidation / M&A opportunities are outlined in general terms. Finally, we provide a subjective opinion on the future of cardiotoxicity screening, suggest how regulatory guidelines might change in the future, and outline some commercial opportunities that might be associated with the current / future cardiotoxicity screening environment. Table of contents: Overview Executive summary Cardiac anatomy and physiology Overview Anatomy of the heart The cardiac cycle The resting potential The action potential Origin of the heartbeat Clinical assessment of cardiac function Cardiac ion channels Summary Cardiotoxicity Overview Directly cardiotoxic drugs Mechanism of toxicity of directly cardiotoxic drugs Anthracyclines / anthracycline-interacting anticancer drugs Other anti-cancer drugs NSAIDs Other drugs Direct cardiotoxicity predisposing factors Direct cardiotoxicity - damage limitation Pro-arrhythmic drugs Mechanism of cardiotoxicity of pro-arrhythmic drugs Molecular targets of proarrhythmic drugs Drug interactions with ion channels Arrhythmia generation Proarrhythmia - pre-disposing factors Proarrhythmia damage limitation Summary Regulatory environment and industry response Overview History ICH Guideline S7B: Pre-clinical QT studies ICH Guideline E14: clinical QT studies Other regulatory agency documents... 54
3 4.6 Regulatory decision-making Industry concerns Summary Assessing drug-induced cardiotoxicity Overview Surrogate markers for proarrhythmia Measures of ion channel flux Action potential morphology and duration Dispersion of action potential duration QT interval prolongation Combinations of measures Preclinical proarrhythmia screening in silico approaches Preclinical proarrhythmia screening single cell methods Cell types Non-patch clamp single cell assays Conventional patch clamping Automated MT/HT patch clamping Scanning patch clamping Preclinical proarrhythmia screening multicell methods Purkinje fibre and papillary muscle systems Ventricular wedge Whole heart systems Future developments in multicellular in vitro systems Preclinical proarrhythmia screening in vivo methods Anaesthetised animals Conscious, telemetrised animals Predisposed models Other models Clinical trials and post-marketing surveillance Low frequency of arrhythmia complicates trials Pharmacogenetics Measurements in the trial population Impact of QT effects discovered in clinical trials Screening for direct cardiotoxicity Markers of cardiac damage Animal models Summary Industry attitudes and survey results Overview Previous surveys Current survey questionnaire format Survey population Analysis of questionnaire responses Current survey interview format Survey population Analysis of interview responses Summary Commercial environment Overview Outline analysis of cardiotoxicity screening segment
4 7.3 Proarrhythmia screening product / service providers Summary Opinion: The future of cardiotoxicity screening in drug development Overview Proarrhythmia screening Early stage drug development Late stage drug development Screening for direct cardiotoxicity Chronic cardiotoxicity Acute cardiotoxicity Summary Appendix 1: References Appendix 2: Interview transcripts A2.1 Bush A2.2 Patel A2.3 Tyl A2.4 Antzelevitch A2.5 Tsaioun A2.6: Malik Appendix 3: Companies providing cardiotoxicity screening products or services Appendix 4: Outline of top 29 companies Appendix 5: Results of survey by questionnaire A5.1 Survey population A5.2 Survey responses To purchase this report, please contact Insight Pharma Reports ( To discuss bespoke projects covering similar subject matter, please contact Beremans Ltd.
5 Overview This report identifies and discusses methods, products and services designed to identify cardiotoxic compounds before they reach the market; discusses the current and possible future regulatory environment; outlines main commercial competitors; and suggests broad types of commercial opportunity and future M&A activity in this subsector. The primary focus is on identification of proarrhythmia, but some discussion is provided on identification of other forms of cardiotoxicity. We start by providing a primer on cardiac anatomy / physiology. Particular consideration is given to the various ion fluxes that contribute to the cardiac action potential. Next, drug-induced cardiotoxicity is discussed under two headings: direct cardiotoxicity (i.e. where the primary symptom of cardiotoxicity is not arrhythmia) and proarrhythmic cardiotoxicity. In each category, we provide extensive lists of drugs associated with cardiotoxicity, discuss factors which may predispose to druginduced cardiotoxicity, and outline current / proposed cardioprotective approaches. We then outline the history and status quo of the current regulatory environment pertinent to drug-induced proarrhythmia, with particular reference to guidelines S7B and E14. In addition, we discuss some of the main factors which may impact the regulators decisions regarding drug non-approval due to cardiotoxicity. Next, we provide a detailed discussion on methods for assessing the potential for drug-induced cardiotoxicity, with a primary focus on proarrhythmia screening. Various recommended and proposed surrogates for torsades de pointes (TdP) risk are compared. Preclinical and clinical proarrhythmia screens are discussed, including in silico methods, in vitro single cell / multicellular methods, in vivo methods and early clinical trial methods. Some discussion also is provided on mechanisms of screening for direct (non-proarrhythmic) cardiotoxicity. In addition, we provide and discuss the results of an industry survey undertaken for this report by online questionnaire and by one-to-one interviews with key experts. Questions were aimed at, inter alia, gathering information on attitudes to preclinical models and surrogates for TdP risk, and opinions as to how and when current guidelines might change. Results from previous industry surveys also are noted. An overview of 50 commercial entities offering cardiotoxicity screening products / services is provided. Further analysis is included regarding the competitive positioning and ownership of 29 companies that have some clear cardiotoxicity screening focus. Broad consolidation / M&A opportunities are outlined in general terms. Finally, we provide a subjective opinion on the future of cardiotoxicity screening, suggest how regulatory guidelines might change in the future, and outline some commercial opportunities that might be associated with the current / future cardiotoxicity screening environment.
In Vitro Assessment to Replace the Clinical TQT Study: The Comprehensive In Vitro ProArrhythmia Assay (CiPA) Initiative
 In Vitro Assessment to Replace the Clinical TQT Study: The Comprehensive In Vitro ProArrhythmia Assay (CiPA) Initiative Gary Gintant, AbbVie for the Comprehensive in Vitro ProArrhythmia Assay Group Hot
In Vitro Assessment to Replace the Clinical TQT Study: The Comprehensive In Vitro ProArrhythmia Assay (CiPA) Initiative Gary Gintant, AbbVie for the Comprehensive in Vitro ProArrhythmia Assay Group Hot
Preparing for Changing Cardiac Safety Regulations. Joy Olbertz, PharmD, PhD
 Preparing for Changing Cardiac Safety Regulations Joy Olbertz, PharmD, PhD Questions? Do I still have to assess proarrhythmic potential of my compound in a Thorough QT (TQT) study? If I have a TQT to conduct,
Preparing for Changing Cardiac Safety Regulations Joy Olbertz, PharmD, PhD Questions? Do I still have to assess proarrhythmic potential of my compound in a Thorough QT (TQT) study? If I have a TQT to conduct,
The QT interval as it relates to the safety of non-cardiac drugs
 European Heart Journal Supplements (2007) 9 (Supplement G), G3 G8 doi:10.1093/eurheartj/sum047 The QT interval as it relates to the safety of non-cardiac drugs Peter R. Kowey 1 * and Marek Malik 2,3 1
European Heart Journal Supplements (2007) 9 (Supplement G), G3 G8 doi:10.1093/eurheartj/sum047 The QT interval as it relates to the safety of non-cardiac drugs Peter R. Kowey 1 * and Marek Malik 2,3 1
Metrion Biosciences: the ion channel specialists
 Metrion Biosciences: the ion channel specialists In depth profiling of human ipsc cardiomyocytes: From electrophysiology to phenotypic assays Saïd El-Haou, PhD 30 November 2017 www.metrionbiosciences.com
Metrion Biosciences: the ion channel specialists In depth profiling of human ipsc cardiomyocytes: From electrophysiology to phenotypic assays Saïd El-Haou, PhD 30 November 2017 www.metrionbiosciences.com
Can the Thorough QT Study Be Replaced? Norman Stockbridge Division of Cardiovascular and Renal Products FDA/CDER
 Can the Thorough QT Study Be Replaced? Norman Stockbridge Division of Cardiovascular and Renal Products FDA/CDER Can the TQT study be replaced? Yes. Thank you. Any questions? Four genre of replacement
Can the Thorough QT Study Be Replaced? Norman Stockbridge Division of Cardiovascular and Renal Products FDA/CDER Can the TQT study be replaced? Yes. Thank you. Any questions? Four genre of replacement
CiPA, Pre-clinical Testing. Derek Leishman PhD DSP & ICH S7B
 CiPA, Pre-clinical Testing Derek Leishman PhD DSP & ICH S7B Outline Introduction Scenarios Conclusion Expressing an opinion What is not covered? Analysis/critique of the components too little time to try
CiPA, Pre-clinical Testing Derek Leishman PhD DSP & ICH S7B Outline Introduction Scenarios Conclusion Expressing an opinion What is not covered? Analysis/critique of the components too little time to try
Cover Page. The handle holds various files of this Leiden University dissertation.
 Cover Page The handle http://hdl.handle.net/1887/19939 holds various files of this Leiden University dissertation. Author: Chain, Anne S.Y. Title: Mind the gap : predicting cardiovascular risk during drug
Cover Page The handle http://hdl.handle.net/1887/19939 holds various files of this Leiden University dissertation. Author: Chain, Anne S.Y. Title: Mind the gap : predicting cardiovascular risk during drug
EMEA PUBLIC STATEMENT ON THE RECOMMENDATION TO SUSPEND THE MARKETING AUTHORISATION FOR ORLAAM (LEVACETYLMETHADOL) IN THE EUROPEAN UNION
 The European Agency for the Evaluation of Medicinal Products Evaluation of Medicines for Human Use London, 19 April 2001 EMEA/8776/01 EMEA PUBLIC STATEMENT ON THE RECOMMENDATION TO SUSPEND THE MARKETING
The European Agency for the Evaluation of Medicinal Products Evaluation of Medicines for Human Use London, 19 April 2001 EMEA/8776/01 EMEA PUBLIC STATEMENT ON THE RECOMMENDATION TO SUSPEND THE MARKETING
Design and Analysis of QT/QTc Studies Conceptional and Methodical Considerations Based on Experience
 Design and Analysis of QT/QTc Studies Conceptional and Methodical Considerations Based on Experience Dr. Manfred Wargenau, Institute, Düsseldorf OVERVIEW Clinical background The ICH E14 guideline / review
Design and Analysis of QT/QTc Studies Conceptional and Methodical Considerations Based on Experience Dr. Manfred Wargenau, Institute, Düsseldorf OVERVIEW Clinical background The ICH E14 guideline / review
Comparison of different proarrhythmia biomarkers in isolated rabbit hearts
 Comparison of different proarrhythmia biomarkers in isolated rabbit hearts Summary of PhD Thesis Szabolcs Orosz, MSc Supervisor: Attila Farkas MD, PhD 2nd Dept. of Internal Medicine and Cardiology Centre
Comparison of different proarrhythmia biomarkers in isolated rabbit hearts Summary of PhD Thesis Szabolcs Orosz, MSc Supervisor: Attila Farkas MD, PhD 2nd Dept. of Internal Medicine and Cardiology Centre
MEA assays using human ipsc-derived cardiomyocytes; challenges and opportunities
 MEA assays using human ipsc-derived cardiomyocytes; challenges and opportunities Event Presenter Date EMA Workshop 2017 Tessa de Korte, MSc 2017, October 5 Page 1 Ncardia at a glance Foundation Ncardia
MEA assays using human ipsc-derived cardiomyocytes; challenges and opportunities Event Presenter Date EMA Workshop 2017 Tessa de Korte, MSc 2017, October 5 Page 1 Ncardia at a glance Foundation Ncardia
Electrocardiographic Markers Associated with Sotalol-induced Torsades de Pointes
 Electrocardiographic Markers Associated with Sotalol-induced Torsades de Pointes Based on Data from the Telemetric and Holter ECG Warehouse (THEW) Initiative Jean-PhiIippe Couderc, PhD Center for Quantitative
Electrocardiographic Markers Associated with Sotalol-induced Torsades de Pointes Based on Data from the Telemetric and Holter ECG Warehouse (THEW) Initiative Jean-PhiIippe Couderc, PhD Center for Quantitative
QT QT Study Design and Statistical Evaluation of QT Prolongation Introduction and QT Interval Correction
 QT QT Study Design and Statistical Evaluation of QT Prolongation Introduction and QT Interval Correction Yasushi Orihashi Clinical Data and Biostatistics Department, Daiichi Sankyo Co., Ltd. email: orihashi.yasushi.c2@daiichisankyo.co.jp
QT QT Study Design and Statistical Evaluation of QT Prolongation Introduction and QT Interval Correction Yasushi Orihashi Clinical Data and Biostatistics Department, Daiichi Sankyo Co., Ltd. email: orihashi.yasushi.c2@daiichisankyo.co.jp
Russia Cardiac Assist Devices Market Outlook to 2021
 Russia Cardiac Assist Devices Market Outlook to 2021 Russia Cardiac Assist Devices Market Outlook to 2021 BioPortfolio has been marketing business and market research reports from selected publishers for
Russia Cardiac Assist Devices Market Outlook to 2021 Russia Cardiac Assist Devices Market Outlook to 2021 BioPortfolio has been marketing business and market research reports from selected publishers for
ICH E14 THE CLINICAL EVALUATION OF QT/QTc INTERVAL PROLONGATION AND PROARRHYTHMIC POTENTIAL FOR NON-ANTIARRHYTHMIC DRUGS.
 European Medicines Agency London, 25 May 2005 CHMP/ICH/2/04 ICH E14 THE CLINICAL EVALUATION OF QT/QTc INTERVAL PROLONGATION AND PROARRHYTHMIC POTENTIAL FOR NON-ANTIARRHYTHMIC DRUGS ICH Step 4 NOTE FOR
European Medicines Agency London, 25 May 2005 CHMP/ICH/2/04 ICH E14 THE CLINICAL EVALUATION OF QT/QTc INTERVAL PROLONGATION AND PROARRHYTHMIC POTENTIAL FOR NON-ANTIARRHYTHMIC DRUGS ICH Step 4 NOTE FOR
xcelligence System RTCA Cardio Instrument
 xcelligence System Application Note No. 15 January 2013 xcelligence System RTCA Cardio Instrument Preclinical Cardiac Safety Assessment using Human ipsc Derived icell TM Cardiomyocytes Introduction Over
xcelligence System Application Note No. 15 January 2013 xcelligence System RTCA Cardio Instrument Preclinical Cardiac Safety Assessment using Human ipsc Derived icell TM Cardiomyocytes Introduction Over
QT Assessment: The new paradigm, but now what? Joy Olbertz PharmD, PhD Senior Director, Cardiovascular Safety Services
 QT Assessment: The new paradigm, but now what? Joy Olbertz PharmD, PhD Senior Director, Cardiovascular Safety Services The Evolution of ICH E14 Points to Consider Joint Health Canada/FDA Concept Paper
QT Assessment: The new paradigm, but now what? Joy Olbertz PharmD, PhD Senior Director, Cardiovascular Safety Services The Evolution of ICH E14 Points to Consider Joint Health Canada/FDA Concept Paper
Effects of reference compounds on impedance signals from stem cellderived human cardiomyocytes
 Effects of reference compounds on impedance signals from stem cellderived human cardiomyocytes Herbert M. Himmel, Safety Pharmacology, Bayer Pharma AG, Wuppertal, Germany SPS Webinar Cardiac Safety Testing
Effects of reference compounds on impedance signals from stem cellderived human cardiomyocytes Herbert M. Himmel, Safety Pharmacology, Bayer Pharma AG, Wuppertal, Germany SPS Webinar Cardiac Safety Testing
Evolution of strategies to improve preclinical cardiac safety testing
 OPINION Evolution of strategies to improve preclinical cardiac safety testing Gary Gintant, Philip T. Sager and Norman Stockbridge Abstract The early and efficient assessment of cardiac safety liabilities
OPINION Evolution of strategies to improve preclinical cardiac safety testing Gary Gintant, Philip T. Sager and Norman Stockbridge Abstract The early and efficient assessment of cardiac safety liabilities
Drug-induced effects on the heart: insights from HPC simulations
 Drug-induced effects on the heart: insights from HPC simulations Blanca Rodriguez Computational Biology Group University of Oxford July 1 st, 2009 Acknowledgements Esther Pueyo, Lucia Romero, Alberto Corrias,
Drug-induced effects on the heart: insights from HPC simulations Blanca Rodriguez Computational Biology Group University of Oxford July 1 st, 2009 Acknowledgements Esther Pueyo, Lucia Romero, Alberto Corrias,
Drug-induced arrhythmogenesis. Prediction and Mechanisms. Blanca Rodriguez and Computational Biology Group University of Oxford
 Drug-induced arrhythmogenesis. Prediction and Mechanisms Blanca Rodriguez and Computational Biology Group University of Oxford Feb 3 rd, 29 Work done by Esther Pueyo, Lucia Romero, Alberto Corrias, Martin
Drug-induced arrhythmogenesis. Prediction and Mechanisms Blanca Rodriguez and Computational Biology Group University of Oxford Feb 3 rd, 29 Work done by Esther Pueyo, Lucia Romero, Alberto Corrias, Martin
Guidance for Industry
 Guidance for Industry E14 Clinical Evaluation of QT/QTc Interval Prolongation and Proarrhythmic Potential for Non-Antiarrhythmic Drugs U.S. Department of Health and Human Services Food and Drug Administration
Guidance for Industry E14 Clinical Evaluation of QT/QTc Interval Prolongation and Proarrhythmic Potential for Non-Antiarrhythmic Drugs U.S. Department of Health and Human Services Food and Drug Administration
For Discussion Purposes Only
 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36 37 38 39 40 41 42 43 Step 1 Draft 2 (July 17, 2003) Draft 3 (November 12, 2003), Draft 4 (June 10, 2004)
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36 37 38 39 40 41 42 43 Step 1 Draft 2 (July 17, 2003) Draft 3 (November 12, 2003), Draft 4 (June 10, 2004)
Preclinical Perspectives of QT Outlook from ICH Guidelines
 Biometry in Early Clinical Research QT/QTc Interval Workshop in Heidelberg, November 17-19, 2005 Preclinical Perspectives of QT Outlook from ICH Guidelines Gerd Bode, M.D.,Ph.D. Frelance Consultant, e-mail
Biometry in Early Clinical Research QT/QTc Interval Workshop in Heidelberg, November 17-19, 2005 Preclinical Perspectives of QT Outlook from ICH Guidelines Gerd Bode, M.D.,Ph.D. Frelance Consultant, e-mail
How to use market and clinical research together to develop claims
 We help healthcare businesses to grow. Whether it is new brands, new products or new markets, use our expertise to find the value maximising decision for your business. A brief introduction to market research
We help healthcare businesses to grow. Whether it is new brands, new products or new markets, use our expertise to find the value maximising decision for your business. A brief introduction to market research
Successes and Evolving Challenges Posed by the Comprehensive In Vitro Proarrhythmia (CiPA) Initiative
 Successes and Evolving Challenges Posed by the Comprehensive In Vitro Proarrhythmia (CiPA) Initiative Gary Gintant Dept. Integrative Pharmacology Integrated Sciences and Technology AbbVie For the CiPA
Successes and Evolving Challenges Posed by the Comprehensive In Vitro Proarrhythmia (CiPA) Initiative Gary Gintant Dept. Integrative Pharmacology Integrated Sciences and Technology AbbVie For the CiPA
Breakout #4 Phase 1 ECG:
 Breakout #4 Phase 1 ECG: Potential Role of ECG under CiPA HESI CSRC CiPA Meeting Washington, DC - May 22, 2018 Jose Vicente, PhD Christine Garnett, PharmD Division of Cardiovascular and Renal Products
Breakout #4 Phase 1 ECG: Potential Role of ECG under CiPA HESI CSRC CiPA Meeting Washington, DC - May 22, 2018 Jose Vicente, PhD Christine Garnett, PharmD Division of Cardiovascular and Renal Products
HHS Public Access Author manuscript Circ Res. Author manuscript; available in PMC 2016 June 05.
 Tissue and Animal Models of Sudden Cardiac Death Karim Sallam, MD *,1,2,3, Yingxin Li, PhD *,1,2,3, Philip T. Sager, MD 1,2, Steven R. Houser, PhD 4, and Joseph C. Wu, MD, PhD 1,2,3 1 Stanford Cardiovascular
Tissue and Animal Models of Sudden Cardiac Death Karim Sallam, MD *,1,2,3, Yingxin Li, PhD *,1,2,3, Philip T. Sager, MD 1,2, Steven R. Houser, PhD 4, and Joseph C. Wu, MD, PhD 1,2,3 1 Stanford Cardiovascular
Cardiac Safety Assessment. In-Vitro/In-Vivo Extrapolation
 Cardiac Safety Assessment In-Vitro/In-Vivo Extrapolation Drugs removed from the market for arrhythmia risk Courtesy of Dr. Norman Stockbridge Copyright 2014 Certara, L.P. All rights reserved. 2 Let s have
Cardiac Safety Assessment In-Vitro/In-Vivo Extrapolation Drugs removed from the market for arrhythmia risk Courtesy of Dr. Norman Stockbridge Copyright 2014 Certara, L.P. All rights reserved. 2 Let s have
Global Deaf/Hearing Aid Market: Trends and Opportunities ( )
 Global Deaf/Hearing Aid Market: Trends and Opportunities (2016-2020) View Report Details Scope of the Report The report titled Global Hearing Aid Market: Size, trends and Forecasts (2016-2020), provides
Global Deaf/Hearing Aid Market: Trends and Opportunities (2016-2020) View Report Details Scope of the Report The report titled Global Hearing Aid Market: Size, trends and Forecasts (2016-2020), provides
Industry Experience of the Concentration-QTc Relationship in Phase 1 Studies. Corina Dota, AstraZeneca
 Industry Experience of the Concentration-QTc Relationship in Phase 1 Studies Corina Dota, AstraZeneca CSRC-2 th February 2012 ICH E14 2.2.5 Alternative Strategies to Assess QT/QTc Interval Effects Alternatives
Industry Experience of the Concentration-QTc Relationship in Phase 1 Studies Corina Dota, AstraZeneca CSRC-2 th February 2012 ICH E14 2.2.5 Alternative Strategies to Assess QT/QTc Interval Effects Alternatives
Literature-based evaluation of four hard endpoint models for assessing drug-induced torsades de pointes liability
 British Journal of Pharmacology (2008) 154, 1523 1527 & 2008 Nature Publishing Group All rights reserved 0007 1188/08 $30.00 www.brjpharmacol.org REVIEW Literature-based evaluation of four hard endpoint
British Journal of Pharmacology (2008) 154, 1523 1527 & 2008 Nature Publishing Group All rights reserved 0007 1188/08 $30.00 www.brjpharmacol.org REVIEW Literature-based evaluation of four hard endpoint
CSRC White Papers. An update Dec. 2010
 CSRC White Papers An update Dec. 2010 Ignacio Rodriguez, MD Clinical Safety Risk Management Roche ignacio.rodriguez.ir1@roche.com CSRC White Papers Position papers usually cover challenging areas of cardiovascular
CSRC White Papers An update Dec. 2010 Ignacio Rodriguez, MD Clinical Safety Risk Management Roche ignacio.rodriguez.ir1@roche.com CSRC White Papers Position papers usually cover challenging areas of cardiovascular
TCP Transl Clin Pharmacol
 TCP 2018;26(4):145-149 https://doi.org/10.12793/tcp.2018.26.4.145 Five years of the CiPA project (2013 2018) - what did we learn? Dong-Seok Yim* Department of Clinical Pharmacology and Therapeutics, Seoul
TCP 2018;26(4):145-149 https://doi.org/10.12793/tcp.2018.26.4.145 Five years of the CiPA project (2013 2018) - what did we learn? Dong-Seok Yim* Department of Clinical Pharmacology and Therapeutics, Seoul
Asia-Pacific Bariatric Surgery Devices Market Outlook to 2020
 Asia-Pacific Bariatric Surgery Devices Market Outlook to 2020 Reference Code: GDMECR0195DB Publication Date: July 2014 Page 1 1 Table of Contents 1 Table of Contents... 2 1.1 List of Tables... 4 1.2 List
Asia-Pacific Bariatric Surgery Devices Market Outlook to 2020 Reference Code: GDMECR0195DB Publication Date: July 2014 Page 1 1 Table of Contents 1 Table of Contents... 2 1.1 List of Tables... 4 1.2 List
DBA Design Effectiveness Awards SoloSTAR. A disposable pen injector for insulin
 DBA Design Effectiveness Awards 2009 SoloSTAR A disposable pen injector for insulin Category: Sub category: Client company: Design consultancy: Product Consumer Sanofi Aventis GmbH DCA Design International
DBA Design Effectiveness Awards 2009 SoloSTAR A disposable pen injector for insulin Category: Sub category: Client company: Design consultancy: Product Consumer Sanofi Aventis GmbH DCA Design International
The Maximum Mean Difference
 The Maximum Mean Difference Statistical Problems in Assessing Cardiac Safety Georg Ferber, Novartis Pharma AG, Basel ROeS Seminar Bern, 10-13 Sep 2007 Overview QT prolongation some background The Thorough
The Maximum Mean Difference Statistical Problems in Assessing Cardiac Safety Georg Ferber, Novartis Pharma AG, Basel ROeS Seminar Bern, 10-13 Sep 2007 Overview QT prolongation some background The Thorough
ON SHELF AVAILABILITY ALIGNMENT PROJECT 2011 ASIA PAC SURVEY RESULTS
 ON SHELF AVAILABILITY ALIGNMENT PROJECT 2011 ASIA PAC SURVEY RESULTS Introduction The ECR Asia Pacific OSA working group conducted an online survey between July and September 2011 aimed at gaining insights
ON SHELF AVAILABILITY ALIGNMENT PROJECT 2011 ASIA PAC SURVEY RESULTS Introduction The ECR Asia Pacific OSA working group conducted an online survey between July and September 2011 aimed at gaining insights
Atrial Fibrillation - Pipeline Review, H1 2017
 Report Information More information from: https://www.wiseguyreports.com/reports/1095866-atrial-fibrillation-pipeline-review-h1-2017 Atrial Fibrillation - Pipeline Review, H1 2017 Report / Search Code:
Report Information More information from: https://www.wiseguyreports.com/reports/1095866-atrial-fibrillation-pipeline-review-h1-2017 Atrial Fibrillation - Pipeline Review, H1 2017 Report / Search Code:
ANNEX I LIST OF THE NAMES OF THE MEDICINAL PRODUCTS, MARKETING AUTHORISATION HOLDERS, PHARMACEUTICAL FORMS, STRENGTHS, ROUTE OF ADMINISTRATION,
 ANNEX I LIST OF THE NAMES OF THE MEDICINAL PRODUCTS, MARKETING AUTHORISATION HOLDERS, PHARMACEUTICAL FORMS, STRENGTHS, ROUTE OF ADMINISTRATION, PACKAGING AND PACKAGE SIZES IN THE MEMBER STATE 1 SERTINDOLE
ANNEX I LIST OF THE NAMES OF THE MEDICINAL PRODUCTS, MARKETING AUTHORISATION HOLDERS, PHARMACEUTICAL FORMS, STRENGTHS, ROUTE OF ADMINISTRATION, PACKAGING AND PACKAGE SIZES IN THE MEMBER STATE 1 SERTINDOLE
Mexico Ostomy Drainage Bags Market Outlook to 2020
 Mexico Ostomy Drainage Bags Market Outlook to 2020 Reference Code: GDMECC0990DB Publication Date: October 2014 Page 1 1 Table of Contents 1 Table of Contents... 2 1.1 List of Tables... 4 1.2 List of Figures...
Mexico Ostomy Drainage Bags Market Outlook to 2020 Reference Code: GDMECC0990DB Publication Date: October 2014 Page 1 1 Table of Contents 1 Table of Contents... 2 1.1 List of Tables... 4 1.2 List of Figures...
Global Magnetic Resonance Imaging (MRI) Systems Market Research Report 2016
 Published on Market Research Reports Inc. (https://www.marketresearchreports.com) Home > Global Magnetic Resonance Imaging (MRI) Systems Market Research Report 2016 Global Magnetic Resonance Imaging (MRI)
Published on Market Research Reports Inc. (https://www.marketresearchreports.com) Home > Global Magnetic Resonance Imaging (MRI) Systems Market Research Report 2016 Global Magnetic Resonance Imaging (MRI)
Cardiac Arrhythmias. For Pharmacists
 Cardiac Arrhythmias For Pharmacists Agenda Overview of the normal Classification Management Therapy Conclusion Cardiac arrhythmias Overview of the normal Arrhythmia: definition From the Greek a-, loss
Cardiac Arrhythmias For Pharmacists Agenda Overview of the normal Classification Management Therapy Conclusion Cardiac arrhythmias Overview of the normal Arrhythmia: definition From the Greek a-, loss
The Future of the Oils and Fats Market in Indonesia to 2018
 1. The Future of the Oils and Fats Market in Indonesia to 2018 Reference Code: FD1360MR Report Price: US$ 875 (Single Copy) www.canadean-winesandspirits.com Summary The Future of the Oils & Fats Market
1. The Future of the Oils and Fats Market in Indonesia to 2018 Reference Code: FD1360MR Report Price: US$ 875 (Single Copy) www.canadean-winesandspirits.com Summary The Future of the Oils & Fats Market
Interpreting Adult Human Thorough QT Studies: Are They Relevant to Pediatric Safety?
 Interpreting Adult Human Thorough QT Studies: Are They Relevant to Pediatric Safety? Hao Zhu, Ph.D. Scientific Lead, QT-IRT, FDA (Dec 10, 2010) Disclaimer: The views expressed in this talk are those of
Interpreting Adult Human Thorough QT Studies: Are They Relevant to Pediatric Safety? Hao Zhu, Ph.D. Scientific Lead, QT-IRT, FDA (Dec 10, 2010) Disclaimer: The views expressed in this talk are those of
Developing and Validating an In Silico Model for Proarrhythmia Risk Assessment Under the CiPA Initiative
 Developing and Validating an In Silico Model for Proarrhythmia Risk Assessment Under the CiPA Initiative May 2018 Zhihua Li, PhD for the CiPA In Silico Working Group US Food and Drug Administration This
Developing and Validating an In Silico Model for Proarrhythmia Risk Assessment Under the CiPA Initiative May 2018 Zhihua Li, PhD for the CiPA In Silico Working Group US Food and Drug Administration This
The Cigarette Market in Saudi Arabia
 The Cigarette Market in Saudi Arabia Product Code: CG0173MR Published Date: June 2015 Report Price: US$ 975 (Single Copy) Synopsis Cigarettes in Saudi Arabia report is a qualitative report providing extensive
The Cigarette Market in Saudi Arabia Product Code: CG0173MR Published Date: June 2015 Report Price: US$ 975 (Single Copy) Synopsis Cigarettes in Saudi Arabia report is a qualitative report providing extensive
Drugs Controlling Myocyte Excitability and Conduction at the AV node Singh and Vaughan-Williams Classification
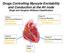 Drugs Controlling Myocyte Excitability and Conduction at the AV node Singh and Vaughan-Williams Classification Class I Na Channel Blockers Flecainide Propafenone Class III K channel Blockers Dofetilide,
Drugs Controlling Myocyte Excitability and Conduction at the AV node Singh and Vaughan-Williams Classification Class I Na Channel Blockers Flecainide Propafenone Class III K channel Blockers Dofetilide,
Drugs, herg and sudden death
 Cell Calcium 35 () 53 57 Drugs, herg and sudden death A.M. Brown a,b, a MetroHealth Campus, Case Western Reserve University, Cleveland, OH 8, USA b ChanTest, Inc., 656 Neo Parkway, Cleveland, OH 8, USA
Cell Calcium 35 () 53 57 Drugs, herg and sudden death A.M. Brown a,b, a MetroHealth Campus, Case Western Reserve University, Cleveland, OH 8, USA b ChanTest, Inc., 656 Neo Parkway, Cleveland, OH 8, USA
Global E-Cigarette & Vaporizer Market Research Report - Forecast 2011 to 2021
 Report Information More information from: https://www.marketresearchfuture.com/reports/949 Global E-Cigarette & Vaporizer Market Research Report - Forecast 2011 to 2021 Report / Search Code: MRFR/F-B &
Report Information More information from: https://www.marketresearchfuture.com/reports/949 Global E-Cigarette & Vaporizer Market Research Report - Forecast 2011 to 2021 Report / Search Code: MRFR/F-B &
Phase 2 Early Afterdepolarization as a Trigger of Polymorphic Ventricular Tachycardia in Acquired Long-QT Syndrome
 Phase 2 Early Afterdepolarization as a Trigger of Polymorphic Ventricular Tachycardia in Acquired Long-QT Syndrome Direct Evidence From Intracellular Recordings in the Intact Left Ventricular Wall Gan-Xin
Phase 2 Early Afterdepolarization as a Trigger of Polymorphic Ventricular Tachycardia in Acquired Long-QT Syndrome Direct Evidence From Intracellular Recordings in the Intact Left Ventricular Wall Gan-Xin
xcelligence RTCA Cardio Instrument Monitor Cardiomyocyte Beating in Real Time for Drug Discovery Research
 xcelligence RTCA Cardio Instrument Monitor Cardiomyocyte Beating in Real Time for Drug Discovery Research xcelligence RTCA Cardio Instrument A new way to test for compound cardiotoxicity Cardiac liability
xcelligence RTCA Cardio Instrument Monitor Cardiomyocyte Beating in Real Time for Drug Discovery Research xcelligence RTCA Cardio Instrument A new way to test for compound cardiotoxicity Cardiac liability
REFERENCE CODE GDHC378DFR PUBLICAT ION DATE M ARCH 2014 BOTOX (MIGRAINE) - FORECAST AND MARKET ANALYSIS TO 2023
 REFERENCE CODE GDHC378DFR PUBLICAT ION DATE M ARCH 2014 BOTOX (MIGRAINE) - Executive Summary Botox: Key Metrics in the 7MM* for Migraine, 2012-2023 2012 Market Sales US $434.3m 5 EU $68.3m Japan Total
REFERENCE CODE GDHC378DFR PUBLICAT ION DATE M ARCH 2014 BOTOX (MIGRAINE) - Executive Summary Botox: Key Metrics in the 7MM* for Migraine, 2012-2023 2012 Market Sales US $434.3m 5 EU $68.3m Japan Total
Safety Pharmacology Post Meeting Survey 2014
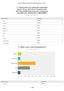 Q1 Please enter your personal information below. (If you want to be included in the 2014 Annual Meeting survey for a chance to win $200, this information is REQUIRED) Answered: 132 Skipped: 14 Name: Company:
Q1 Please enter your personal information below. (If you want to be included in the 2014 Annual Meeting survey for a chance to win $200, this information is REQUIRED) Answered: 132 Skipped: 14 Name: Company:
Spondyloarthropathies: Disease Perception Limits Market
 Spondyloarthropathies: Disease Perception Limits Market Psoriatic arthritis and ankylosing spondylitis form part of the group of diseases known as the spondyloarthropathies. Psoriatic arthritis is a form
Spondyloarthropathies: Disease Perception Limits Market Psoriatic arthritis and ankylosing spondylitis form part of the group of diseases known as the spondyloarthropathies. Psoriatic arthritis is a form
Europe Ginseng Market Report 2016
 Published on Market Research Reports Inc. (https://www.marketresearchreports.com) Home > Europe Ginseng Market Report 2016 Europe Ginseng Market Report 2016 Publication ID: QYR12167206 Publication December
Published on Market Research Reports Inc. (https://www.marketresearchreports.com) Home > Europe Ginseng Market Report 2016 Europe Ginseng Market Report 2016 Publication ID: QYR12167206 Publication December
Global Hearing Aids Market (by Devices, Implant Types, OTC Amplifiers, Diagnostic Instruments), Sales Volume, Company Analysis and Forecast to 2022
 Published on Market Research Reports Inc. (https://www.marketresearchreports.com) Home > Global Hearing Aids Market (by Devices, Implant Types, OTC Amplifiers, Diagnostic Instruments), Sales Volume, Company
Published on Market Research Reports Inc. (https://www.marketresearchreports.com) Home > Global Hearing Aids Market (by Devices, Implant Types, OTC Amplifiers, Diagnostic Instruments), Sales Volume, Company
Benchmarking In Silico Models and Candidate Metrics for Assessing the Risk of Torsade de Pointes
 Benchmarking In Silico Models and Candidate Metrics for Assessing the Risk of Torsade de Pointes Sara Dutta Division of Applied Regulatory Science/OCP/CDER CRSC/HESI/SPS/FDA Meeting December 11, 2014 Silver
Benchmarking In Silico Models and Candidate Metrics for Assessing the Risk of Torsade de Pointes Sara Dutta Division of Applied Regulatory Science/OCP/CDER CRSC/HESI/SPS/FDA Meeting December 11, 2014 Silver
Early drug development: assessment of proarrhythmic risk and cardiovascular safety
 EXPERT REVIEW OF CLINICAL PHARMACOLOGY, 2016 VOL. 9, NO. 12, 1611 1618 http://dx.doi.org/10.1080/17512433.2016.1245142 REVIEW Early drug development: assessment of proarrhythmic risk and cardiovascular
EXPERT REVIEW OF CLINICAL PHARMACOLOGY, 2016 VOL. 9, NO. 12, 1611 1618 http://dx.doi.org/10.1080/17512433.2016.1245142 REVIEW Early drug development: assessment of proarrhythmic risk and cardiovascular
QT prolongation and drug-drug interactions. Filip Josephson M.D., Ph.D Clinical Assessor Swedish Medical Products Agency
 QT prolongation and drug-drug interactions Filip Josephson M.D., Ph.D Clinical Assessor Swedish Medical Products Agency Disposition Introduction to QT prolongation Three relevant DDI scenarios Concluding
QT prolongation and drug-drug interactions Filip Josephson M.D., Ph.D Clinical Assessor Swedish Medical Products Agency Disposition Introduction to QT prolongation Three relevant DDI scenarios Concluding
Prof Dr Ir Ujang Sumarwan, MSc
 3 SURVEY Prof Dr Ir Ujang Sumarwan, MSc Graduate Program of Management and Business- IPB Metode Riset: 3 SURVEY Prof. Dr. Ir. Ujang Sumarwan, MSc 1 3. Survey Research What s going on? Just Asking Questions
3 SURVEY Prof Dr Ir Ujang Sumarwan, MSc Graduate Program of Management and Business- IPB Metode Riset: 3 SURVEY Prof. Dr. Ir. Ujang Sumarwan, MSc 1 3. Survey Research What s going on? Just Asking Questions
Tobacco Insights May
 Tobacco Insights 2015 - May Product Code: TI0017MR Published Date: June 2015 Report Price: US$ 150 (Single Copy) Synopsis Tobacco Insights 2015 - May report is a qualitative report providing extensive
Tobacco Insights 2015 - May Product Code: TI0017MR Published Date: June 2015 Report Price: US$ 150 (Single Copy) Synopsis Tobacco Insights 2015 - May report is a qualitative report providing extensive
PHARMACOLOGY OF ARRHYTHMIAS
 PHARMACOLOGY OF ARRHYTHMIAS Course: Integrated Therapeutics 1 Lecturer: Dr. E. Konorev Date: November 27, 2012 Materials on: Exam #5 Required reading: Katzung, Chapter 14 1 CARDIAC ARRHYTHMIAS Abnormalities
PHARMACOLOGY OF ARRHYTHMIAS Course: Integrated Therapeutics 1 Lecturer: Dr. E. Konorev Date: November 27, 2012 Materials on: Exam #5 Required reading: Katzung, Chapter 14 1 CARDIAC ARRHYTHMIAS Abnormalities
BRIC Transurethral Resection of the Prostate (TURP) Procedures Outlook to 2020
 BRIC Transurethral Resection of the Prostate (TURP) Procedures Outlook Reference Code: GDMECR0130PDB Publication Date: July 2014 Page 1 1 Table of Contents 1 Table of Contents... 2 1.1 List of Tables...
BRIC Transurethral Resection of the Prostate (TURP) Procedures Outlook Reference Code: GDMECR0130PDB Publication Date: July 2014 Page 1 1 Table of Contents 1 Table of Contents... 2 1.1 List of Tables...
September 30, Eric BASTINGS, MD. Acting Director Division of Neurology Products (DNP) Center for Drug Evaluation and Research (CDER)
 ISCTM Autumn Conference Options and methods to improve cognitive assessment in clinical trials of AD and its precursors September 30, 2013 Eric BASTINGS, MD Acting Director Division of Neurology Products
ISCTM Autumn Conference Options and methods to improve cognitive assessment in clinical trials of AD and its precursors September 30, 2013 Eric BASTINGS, MD Acting Director Division of Neurology Products
Asia-Pacific Electrophysiology Market Outlook to 2020
 Asia-Pacific Electrophysiology Market Outlook to 2020 Reference Code: GDMECR0114DB Publication Date: May 2014 Page 1 1 Table of Contents 1 Table of Contents... 2 1.1 List of Tables... 5 1.2 List of Figures...
Asia-Pacific Electrophysiology Market Outlook to 2020 Reference Code: GDMECR0114DB Publication Date: May 2014 Page 1 1 Table of Contents 1 Table of Contents... 2 1.1 List of Tables... 5 1.2 List of Figures...
China Cranio Maxillofacial Fixation (CMF) Market Outlook to 2020
 China Cranio Maxillofacial Fixation (CMF) Market Outlook to 2020 Reference Code: GDMECC0079DB Publication Date: March 2014 Page 1 1 Table of Contents 1 Table of Contents... 2 1.1 List of Tables... 4 1.2
China Cranio Maxillofacial Fixation (CMF) Market Outlook to 2020 Reference Code: GDMECC0079DB Publication Date: March 2014 Page 1 1 Table of Contents 1 Table of Contents... 2 1.1 List of Tables... 4 1.2
The overly-sensitive heart: sodium channel block and QRS interval prolongation
 BJP British Journal of Pharmacology DOI:10.1111/j.1476-5381.2011.01433.x www.brjpharmacol.org COMMENTARYbph_1433 254..259 The overly-sensitive heart: sodium channel block and QRS interval prolongation
BJP British Journal of Pharmacology DOI:10.1111/j.1476-5381.2011.01433.x www.brjpharmacol.org COMMENTARYbph_1433 254..259 The overly-sensitive heart: sodium channel block and QRS interval prolongation
NIH Public Access Author Manuscript Comput Cardiol (2010). Author manuscript; available in PMC 2011 July 11.
 NIH Public Access Author Manuscript Published in final edited form as: Comput Cardiol (2010). 2010 September 26; 2010: 353 356. Torsadogenic Drug-induced Increased Short-term Variability of JT-area Xiao
NIH Public Access Author Manuscript Published in final edited form as: Comput Cardiol (2010). 2010 September 26; 2010: 353 356. Torsadogenic Drug-induced Increased Short-term Variability of JT-area Xiao
Volume 9, Number 14 September 2015
 Arden and Greater East Midlands Commissioning Support Unit in association with Lincolnshire Clinical Commissioning Groups, Lincolnshire Community Health Services, United Lincolnshire Hospitals Trust and
Arden and Greater East Midlands Commissioning Support Unit in association with Lincolnshire Clinical Commissioning Groups, Lincolnshire Community Health Services, United Lincolnshire Hospitals Trust and
Annex I Scientific conclusions and grounds for the variation to the terms of the Marketing Authorisation(s)
 Annex I Scientific conclusions and grounds for the variation to the terms of the Marketing Authorisation(s) EMA/CMDh/766270/2017 1 Scientific conclusions Taking into account the PRAC Assessment Report
Annex I Scientific conclusions and grounds for the variation to the terms of the Marketing Authorisation(s) EMA/CMDh/766270/2017 1 Scientific conclusions Taking into account the PRAC Assessment Report
Global Standard Radiography Film X-Ray Film Printer Market Research Report 2016
 Published on Market Research Reports Inc. (https://www.marketresearchreports.com) Home > Global Standard Radiography Film X-Ray Film Printer Market Research Report 2016 Global Standard Radiography Film
Published on Market Research Reports Inc. (https://www.marketresearchreports.com) Home > Global Standard Radiography Film X-Ray Film Printer Market Research Report 2016 Global Standard Radiography Film
What key areas would you want to explore in order to understand Magna's decline in profitability?
 Magna Health Our client is Magna Health, a health care company in the Midwest. It both insures patients and provides health care services. Employers pay a fixed premium to Magna for each of their employees
Magna Health Our client is Magna Health, a health care company in the Midwest. It both insures patients and provides health care services. Employers pay a fixed premium to Magna for each of their employees
Ncardia. Assessment of pro-arrhythmic effects in Pluricyte Cardiomyocytes. using the Axion BioSystems Maestro TM MEA system
 Ncardia Stem cell experts Assessment of pro-arrhythmic effects in Pluricyte Cardiomyocytes using the Axion BioSystems Maestro TM MEA system Application note Version 2.0 Contents 1. Introduction 1 2. Assessment
Ncardia Stem cell experts Assessment of pro-arrhythmic effects in Pluricyte Cardiomyocytes using the Axion BioSystems Maestro TM MEA system Application note Version 2.0 Contents 1. Introduction 1 2. Assessment
Can non-clinical repolarization assays predict the results of clinical thorough QT studies? Results from a research consortium
 BJP British Journal of Pharmacology RESEARCH PAPER British Journal of Pharmacology (2018) 175 606 617 606 Can non-clinical repolarization assays predict the results of clinical thorough QT studies? Results
BJP British Journal of Pharmacology RESEARCH PAPER British Journal of Pharmacology (2018) 175 606 617 606 Can non-clinical repolarization assays predict the results of clinical thorough QT studies? Results
Quantitative and qualitative research
 Quantitative and qualitative research Quantitative and qualitative research 1 Qualitative vs quantitative 2 Overview of qualitative research 3 Qualitative research techniques 3 Overview of quantitative
Quantitative and qualitative research Quantitative and qualitative research 1 Qualitative vs quantitative 2 Overview of qualitative research 3 Qualitative research techniques 3 Overview of quantitative
1.1.1 Most common causes of infertility or sub-fertility 13
 CONTENTS REPORT AUTHOR FOREWORD LIST OF TABLES LIST OF FIGURES EXECUTIVE SUMMARY 11 1. MARKET 13 1.1 Introduction 13 1.1.1 Most common causes of infertility or sub-fertility 13 1.2 Definition 14 1.3 Size
CONTENTS REPORT AUTHOR FOREWORD LIST OF TABLES LIST OF FIGURES EXECUTIVE SUMMARY 11 1. MARKET 13 1.1 Introduction 13 1.1.1 Most common causes of infertility or sub-fertility 13 1.2 Definition 14 1.3 Size
Comparison of Two Highly Automated ECG Algorithms for Detection of Drug-Induced Cardiac Ion Channel Block
 Comparison of Two Highly Automated ECG Algorithms for Detection of Drug-Induced Cardiac Ion Channel Block Marina Brockway 1, Anthony A. Fossa 2 and Jay W. Mason 3,4 US Food and Drug Administration (FDA)
Comparison of Two Highly Automated ECG Algorithms for Detection of Drug-Induced Cardiac Ion Channel Block Marina Brockway 1, Anthony A. Fossa 2 and Jay W. Mason 3,4 US Food and Drug Administration (FDA)
Comparison of Electrocardiographic Biomarkers for Differentiating. Drug-induced Single vs. Multiple Cardiac Ion Channel Block
 Article Type: Article Comparison of Electrocardiographic Biomarkers for Differentiating Drug-induced Single vs. Multiple Cardiac Ion Channel Block Marina Brockway 1, Jay W. Mason 2,3, Brian P. Brockway
Article Type: Article Comparison of Electrocardiographic Biomarkers for Differentiating Drug-induced Single vs. Multiple Cardiac Ion Channel Block Marina Brockway 1, Jay W. Mason 2,3, Brian P. Brockway
The QT Interval Safety Endpoint for DR- TB trials. Kelly Dooley & Gary Maartens (Disclaimer: I know very little cardiac electrophysiology)
 The QT Interval Safety Endpoint for DR- TB trials Kelly Dooley & Gary Maartens (Disclaimer: I know very little cardiac electrophysiology) The ECG tracing Physiology of a cardiac myocyte Flow of ions (Na
The QT Interval Safety Endpoint for DR- TB trials Kelly Dooley & Gary Maartens (Disclaimer: I know very little cardiac electrophysiology) The ECG tracing Physiology of a cardiac myocyte Flow of ions (Na
HESI Technical Committee on Cardiovascular Safety: Advancing the clinical relevance of preclinical assessments
 HESI Technical Committee on Cardiovascular Safety: Advancing the clinical relevance of preclinical assessments Brian R. Berridge, DVM, PhD, DACVP Committee Co-Chair ILSI Health and Environmental Sciences
HESI Technical Committee on Cardiovascular Safety: Advancing the clinical relevance of preclinical assessments Brian R. Berridge, DVM, PhD, DACVP Committee Co-Chair ILSI Health and Environmental Sciences
(Incidence, Treatments, Key Companies, Pipeline and Trends)
 World Alzheimer's Disease Market (Incidence, Treatments, Key Companies, Pipeline and Trends) CHAPTER ONE: EXECUTIVE SUMMARY Background Scope and Methodology Size and Growth of the Market Key Issues and
World Alzheimer's Disease Market (Incidence, Treatments, Key Companies, Pipeline and Trends) CHAPTER ONE: EXECUTIVE SUMMARY Background Scope and Methodology Size and Growth of the Market Key Issues and
REFERENCE CODE GDHC1161DFR PUBLICAT ION DATE SEPTEMBER 2013 MENOMUNE (MENINGOCOCCAL VACCINES) - FORECAST AND MARKET ANALYSIS TO 2022
 REFERENCE CODE GDHC1161DFR PUBLICAT ION DATE SEPTEMBER 2013 MENOMUNE (MENINGOCOCCAL VACCINES) - Executive Summary Menomune: Key Metrics in the Nine Major Pharmaceutical Markets 2012 Market Sales US 5EU
REFERENCE CODE GDHC1161DFR PUBLICAT ION DATE SEPTEMBER 2013 MENOMUNE (MENINGOCOCCAL VACCINES) - Executive Summary Menomune: Key Metrics in the Nine Major Pharmaceutical Markets 2012 Market Sales US 5EU
Guidance for Industry
 Reprinted from FDA s website by Guidance for Industry E14 Clinical Evaluation of QT/QTc Interval Prolongation and Proarrhythmic Potential for Non-Antiarrhythmic Drugs Questions and Answers (R1) U.S. Department
Reprinted from FDA s website by Guidance for Industry E14 Clinical Evaluation of QT/QTc Interval Prolongation and Proarrhythmic Potential for Non-Antiarrhythmic Drugs Questions and Answers (R1) U.S. Department
Voltage-Gated Ion Channels as Drug Targets
 Voltage-Gated Ion Channels as Drug Targets Edited by David J. Triggle, Murali Gopalakrishnan, David Rampe, and Wei Zheng WILEY- VCH WILEY-VCH Verlag GmbH & Co. KGaA Preface XI 1 Introduction - On Ion Channels
Voltage-Gated Ion Channels as Drug Targets Edited by David J. Triggle, Murali Gopalakrishnan, David Rampe, and Wei Zheng WILEY- VCH WILEY-VCH Verlag GmbH & Co. KGaA Preface XI 1 Introduction - On Ion Channels
EU5 Bariatric Surgery Procedures Outlook to 2020
 EU5 Bariatric Surgery Procedures Outlook to 2020 Reference Code: GDMECR0089PDB Publication Date: July 2014 Page 1 1 Table of Contents 1 Table of Contents... 2 1.1 List of Tables... 3 1.2 List of Figures...
EU5 Bariatric Surgery Procedures Outlook to 2020 Reference Code: GDMECR0089PDB Publication Date: July 2014 Page 1 1 Table of Contents 1 Table of Contents... 2 1.1 List of Tables... 3 1.2 List of Figures...
Salisbury NHS Foundation Trust. Communications Plan Sustainability & Carbon Reduction
 Communication Plan Appendix D Salisbury NHS Foundation Trust Communications Plan Sustainability & Carbon Reduction Contents Introduction Aims Audiences Mechanisms Measurement Actions & Key Messages Introduction
Communication Plan Appendix D Salisbury NHS Foundation Trust Communications Plan Sustainability & Carbon Reduction Contents Introduction Aims Audiences Mechanisms Measurement Actions & Key Messages Introduction
Global Health Functional Food Market: Size, Trends & Forecasts ( ) December 2016
 Global Health Functional Food Market: Size, Trends & Forecasts (2016-2020) December 2016 Global Health Functional Food Market Report Scope of the Report The report titled Global Health Functional Food
Global Health Functional Food Market: Size, Trends & Forecasts (2016-2020) December 2016 Global Health Functional Food Market Report Scope of the Report The report titled Global Health Functional Food
DOWNLOAD OR READ : UPDATE ON BIOMARKERS IN ASTHMA AN ISSUE OF IMMUNOLOGY AND ALLERGY CLINICS PDF EBOOK EPUB MOBI
 DOWNLOAD OR READ : UPDATE ON BIOMARKERS IN ASTHMA AN ISSUE OF IMMUNOLOGY AND ALLERGY CLINICS PDF EBOOK EPUB MOBI Page 1 Page 2 update on biomarkers in asthma an issue of immunology and allergy clinics
DOWNLOAD OR READ : UPDATE ON BIOMARKERS IN ASTHMA AN ISSUE OF IMMUNOLOGY AND ALLERGY CLINICS PDF EBOOK EPUB MOBI Page 1 Page 2 update on biomarkers in asthma an issue of immunology and allergy clinics
CHAPTER 3 METHODOLOGY
 CHAPTER 3 METHODOLOGY The research will be conducted in several steps below: Figure 3.1 Research Frameworks 3.1 Literature Study Involve conceptual literature from book related to Integrated Marketing
CHAPTER 3 METHODOLOGY The research will be conducted in several steps below: Figure 3.1 Research Frameworks 3.1 Literature Study Involve conceptual literature from book related to Integrated Marketing
Noninvasive Glucose Monitors to 2022
 Noninvasive Glucose Monitors to 2022 Devices, Technologies, Players and Prospects Report Prospectus Greystone Research associates N o n i n v a s i v e G l u c o s e M o n i t o r s t o 2 0 2 2 Greystone
Noninvasive Glucose Monitors to 2022 Devices, Technologies, Players and Prospects Report Prospectus Greystone Research associates N o n i n v a s i v e G l u c o s e M o n i t o r s t o 2 0 2 2 Greystone
REFERENCE CODE GDHC386DFR PUBLICAT ION DATE M ARCH 2014 GARDASIL (PROPHYLACTIC HUMAN PAPILLOMAVIRUS VACCINES) - FORECAST AND MARKET ANALYSIS TO 2022
 REFERENCE CODE GDHC386DFR PUBLICAT ION DATE M ARCH 2014 GARDASIL (PROPHYLACTIC HUMAN PAPILLOMAVIRUS VACCINES) - FORECAST AND MARKET ANALYSIS TO 2022 Executive Summary Gardasil: Key Metrics in Nine Major
REFERENCE CODE GDHC386DFR PUBLICAT ION DATE M ARCH 2014 GARDASIL (PROPHYLACTIC HUMAN PAPILLOMAVIRUS VACCINES) - FORECAST AND MARKET ANALYSIS TO 2022 Executive Summary Gardasil: Key Metrics in Nine Major
Asia-Pacific Endoscopic Retrograde Cholangiopancreatography (ERCP) Procedures Outlook to 2020
 Asia-Pacific Endoscopic Retrograde Cholangiopancreatography (ERCP) Reference Code: GDMECR0107PDB Publication Date: July 2014 Page 1 1 Table of Contents 1 Table of Contents... 2 1.1 List of Tables... 3
Asia-Pacific Endoscopic Retrograde Cholangiopancreatography (ERCP) Reference Code: GDMECR0107PDB Publication Date: July 2014 Page 1 1 Table of Contents 1 Table of Contents... 2 1.1 List of Tables... 3
SPECIAL CARDIAC SAFETY CONCERNS
 SPECIAL CARDIAC SAFETY CONCERNS Shari L. Targum, MD, MPH, FACC Chief, General Medicine Branch 1 Division of Clinical Evaluation and Pharmacology/Toxicology (DCEPT) Office of Tissues and Advanced Therapies
SPECIAL CARDIAC SAFETY CONCERNS Shari L. Targum, MD, MPH, FACC Chief, General Medicine Branch 1 Division of Clinical Evaluation and Pharmacology/Toxicology (DCEPT) Office of Tissues and Advanced Therapies
The Cigarette Market in Greece
 The Cigarette Market in Greece Product Code: CG0145MR Published Date: April 2015 Report Price: US$ 975 (Single Copy) Synopsis Cigarettes in Greece report is a qualitative report providing extensive and
The Cigarette Market in Greece Product Code: CG0145MR Published Date: April 2015 Report Price: US$ 975 (Single Copy) Synopsis Cigarettes in Greece report is a qualitative report providing extensive and
Assessment of pro-arrhythmic effects using Pluricyte Cardiomyocytes. on the ACEA xcelligence RTCA CardioECR
 Assessment of pro-arrhythmic effects using Pluricyte Cardiomyocytes on the ACEA xcelligence RTCA CardioECR Application Note Version 2.1 / March 2018 Contents 1. Introduction 1 2. Assessment of pro-arrhythmic
Assessment of pro-arrhythmic effects using Pluricyte Cardiomyocytes on the ACEA xcelligence RTCA CardioECR Application Note Version 2.1 / March 2018 Contents 1. Introduction 1 2. Assessment of pro-arrhythmic
New Biomarkers The Path to Scientific and Regulatory Approval
 New Biomarkers The Path to Scientific and Regulatory Approval Boaz Mendzelevski, MD Cardiac Safety Consultant and Director of Cardiology Medifacts International Medicine is a science of uncertainty and
New Biomarkers The Path to Scientific and Regulatory Approval Boaz Mendzelevski, MD Cardiac Safety Consultant and Director of Cardiology Medifacts International Medicine is a science of uncertainty and
The pill-in-the-pocket strategy for paroxysmal atrial fibrillation
 The pill-in-the-pocket strategy for paroxysmal atrial fibrillation KONSTANTINOS P. LETSAS, MD, FEHRA LABORATORY OF CARDIAC ELECTROPHYSIOLOGY EVANGELISMOS GENERAL HOSPITAL OF ATHENS ARRHYTHMIAS UPDATE,
The pill-in-the-pocket strategy for paroxysmal atrial fibrillation KONSTANTINOS P. LETSAS, MD, FEHRA LABORATORY OF CARDIAC ELECTROPHYSIOLOGY EVANGELISMOS GENERAL HOSPITAL OF ATHENS ARRHYTHMIAS UPDATE,
Summary Public Assessment Report. Generics. Amiodarona Aurobindo. 200 mg, Tablets. (Amiodarone hydrochloride) PT/H/1444/001/DC
 Summary Public Assessment Report Generics 200 mg, Tablets (Amiodarone hydrochloride) Summary PAR Generics 1/9 Summary Public Assessment Report Generics 200 mg, Tablets Amiodarone hydrochloride 200 mg,
Summary Public Assessment Report Generics 200 mg, Tablets (Amiodarone hydrochloride) Summary PAR Generics 1/9 Summary Public Assessment Report Generics 200 mg, Tablets Amiodarone hydrochloride 200 mg,
