PRESCRIBING INFORMATION LEVOPHED. (norepinephrine bitartrate injection USP) 1 mg norepinephrine/ ml
|
|
|
- Charla Merritt
- 5 years ago
- Views:
Transcription
1 PRESCRIBING INFORMATION LEVOPHED (norepinephrine bitartrate injection USP) 1 mg norepinephrine/ ml Sympathomimetic Amine for Use in Hypotensive Emergencies Pfizer Canada Inc Trans-Canada Highway Kirkland, Québec H9J 2M5 Date of Revision: July 17, 2017 Control No.:
2 DESCRIPTION Norepinephrine (sometimes referred to as l-arterenol/levarterenol or l-norepinephrine) is a sympathomimetic amine which differs from epinephrine by the absence of a methyl group on the nitrogen atom. ACTIONS Levophed (norepinephrine bitartrate injection) functions as a powerful peripheral vasoconstrictor (alpha-adrenergic action) and as a potent inotropic stimulator of the heart and dilator of coronary arteries (beta-adrenergic action). Both of these actions result in an increase in systemic blood pressure and coronary artery blood flow. Cardiac output will vary reflexly in response to systemic hypertension but is usually increased in hypotensive man when the blood pressure is raised to an optimal level. In myocardial infarction accompanied by hypotension. Levophed usually increases aortic blood pressure, coronary artery blood flow, and myocardial oxygenation, thereby helping to limit the area of myocardial ischemia and infarction. Venous return is increased and the heart tends to resume a more normal rate and rhythm than in the hypotensive state. In hypotension that persists after correction of blood volume deficits, Levophed helps raise the blood pressure to an optimal level and establish a more adequate circulation. In myocardial infarction, Levophed has been shown to increase greatly the patient survival rate. Levophed not only corrects systemic shock (through cardiotonic and peripheral vasoconstrictor action), but also markedly dilates the coronary arteries, thereby increasing coronary blood flow, reducing the area of ischemia and promoting myocardial oxygenation. There is increased venous return and the heart tends to resume a more normal rate and rhythm. On the coronary arteries, Levophed causes about two and one half times the degree of vasodilatation that epinephrine produces and therefore has a greater effect in increasing coronary 2
3 flow. It has only a slight effect on sugar metabolism, its hyperglycemic action being far less pronounced than epinephrine, and is not contraindicated in diabetic patients. NOTE: With Levophed administration, bradycardia sometimes occurs, probably as a direct result of the rise in blood pressure to normal levels. INDICATIONS Levophed (norepinephrine bitartrate injection) is recommended for the restoration and maintenance of blood pressure in all acute hypotensive or shock states which may result from surgery, trauma, hemorrhage, myocardial infarction, pheochromocytomectomy, sympathectomy, spinal anesthesia, septicemia, drug reactions, poliomyelitis and blood transfusion reactions. Because of the selective peripheral vasoconstrictive action of Levophed, pooled or stagnant blood in the dilated capillaries is driven into the central circulation, thus maintaining vital functions (e.g.-brain, heart, kidneys, etc.) It may also be useful as an adjunct in the treatment of cardiac arrest and profound hypotension. CONTRAINDICATIONS Levophed (norepinephrine bitartrate injection) should not be given to patients who are hypotensive from blood volume deficits except as an emergency measure to maintain coronary and cerebral artery perfusion until blood volume replacement therapy can be completed. If Levophed is continuously administered to maintain blood pressure in the absence of blood volume replacement, the following may occur: severe peripheral and visceral vasoconstriction, decreased renal perfusion and urine output, poor systemic blood flow despite "normal" blood pressure, tissue hypoxia, and lactate acidosis. 3
4 Levophed should also not be given to patients with mesenteric or peripheral vascular thrombosis (because of the risk of increasing ischemia and extending the area of infarction) unless, in the opinion of the attending physician, the administration of Levophed is necessary as a life-saving procedure. Cyclopropane and halothane anesthetics increase cardiac autonomic irritability and therefore seem to sensitize the myocardium to the action of intravenously administered epinephrine or levarterenol. Hence, the use of Levophed during cyclopropane and halothane anesthesia is generally considered contraindicated because of the risk of producing ventricular tachycardia or fibrillation. The same type of cardiac arrhythmias may result from the use of Levophed in patients with profound hypoxia or hypercarbia. WARNING Levophed (norepinephrine bitartrate injection) should be used with extreme caution in patients receiving monoamine oxidase (MAO) inhibitors or antidepressants of the triptyline or imipramine types, because severe, prolonged hypertension may result. Levophed contains sodium metabisulfite, a sulfite that may cause allergic-type reactions including anaphylactic symptoms and life-threatening or less severe asthmatic episodes in certain susceptible people. The overall prevalence of sulfite sensitivity in the general population is unknown. Sulfite sensitivity is seen more frequently in asthmatic than in nonasthmatic people. PRECAUTIONS Avoid Hypertension: Because of the potency of Levophed (norepinephrine bitartrate injection) and because of varying response to pressor substances, the possibility always exists that dangerously high blood pressure may be produced with overdoses of this pressor agent. It is desirable, therefore, to record the blood pressure every two minutes from the time administration 4
5 is started until the desired blood pressure is obtained, then every five minutes if administration is to be continued. The rate of flow must be watched constantly, and the patient should never be left unattended while receiving Levophed. Headache may be a symptom of hypertension due to overdosage. Site of infusion: Whenever possible, Levophed should be given into a large vein, particularly an antecubital vein because, when administered into this vein, the risk of necrosis of the overlying skin from prolonged vasoconstriction is apparently very slight. Some authors have indicated that the femoral vein is also an acceptable route of administration. A catheter tie-in technique should be avoided, if possible, since the obstruction to blood flow around the tubing may cause stasis and increased local concentration of the drug. Occlusive vascular diseases (for example, atherosclerosis, arteriosclerosis, diabetic endarteritis, Buerger s disease) are more likely to occur in the lower than in the upper extremity. Therefore, one should avoid the veins of the leg or dorsum of the hand in elderly patients, or in those suffering from such disorders. Gangrene has been reported in a lower extremity when Levophed was given in an ankle vein. Extravasation: The infusion site should be checked frequently for free flow. Care should be taken to avoid extravasation of Levophed into the tissues, as local necrosis might ensue due to the vasoconstrictive action of the drug. Blanching along the course of the infused vein, sometimes without obvious extravasation, has been attributed to vasa vasorum constriction with increased permeability of the vein wall, permitting some leakage. This also may progress on rare occasions to superficial slough, particularly during infusion into leg veins, in elderly patients or in those suffering from obliterative vascular disease. Hence, if blanching occurs, consideration should be given to the advisability of changing the infusion site at intervals to allow the effects of local vasoconstriction to subside. It is not known whether Levophed can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Levophed should be given to a pregnant woman only if clearly needed. 5
6 It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when Levophed is administered to a nursing woman. IMPORTANT: Antidote for Extravasation Ischemia: To prevent sloughing and necrosis in areas in which extravasation has taken place, the area should be infiltrated as soon as possible with 10 to 15 ml of saline solution containing from 5 to 10 mg of phentolamine, an adrenergic blocking agent. A syringe with a fine hypodermic needle is used, and the solution is infiltrated liberally throughout the area, which is easily identified by its cold, hard, and pallid appearance. Sympathetic blockage with phentolamine causes immediate and conspicuous local hyperemic changes if the area is infiltrated within 12 hours. Therefore, phentolamine should be given as soon as possible after the extravasation is noted. Some investigators 1 add phentolamine (5 to 10 mg) directly to the infusion flask because it is believed that the drug used in this manner is an effective antidote against sloughing should extravasation occur, whereas the systemic vasopressor activity of the norepinephrine is not impaired. Two investigators 2 stated that, in the treatment of patients with severe hypotension following myocardial infarction, thrombosis in the infused vein and perivenous reactions and necrosis may usually be prevented if 10 mg of heparin are added to each 500 ml of infusion fluid (5 % dextrose) containing norepinephrine. Sympathetic nerve block has also been suggested. 6
7 ADVERSE REACTIONS The following reactions can occur: Body As A Whole: Ischemic injury due to potent vasoconstrictor action and tissue hypoxia. Cardiovascular System: Bradycardia, probably as a reflex result of a rise in blood pressure, arrhythmias. Nervous System: Anxiety, transient headache. Respiratory System: Respiratory difficulty. Skin and Appendages: Extravasation necrosis at injection site. Prolonged administration of any potent vasopressor may result in plasma volume depletion which should be continuously corrected by appropriate fluid and electrolyte replacement therapy. If plasma volumes are not corrected, hypotension may recur when Levophed is discontinued, or blood pressure may be maintained at the risk of severe peripheral and visceral vasoconstriction (e.g., decreased renal perfusion) with diminution in blood flow and tissue perfusion with subsequent tissue hypoxia and lactic acidosis and possible ischemic injury. Gangrene of extremities has been rarely reported. Overdoses or conventional doses in hypersensitive persons {eg. hyperthyroid patients) cause severe hypertension with violent headache, photophobia, stabbing retrosternal pain, pallor, intense sweating and vomiting. You can report any suspected adverse reactions associated with the use of health products to the Canada Vigilance Program by one of the following 3 ways: Report online at Call toll-free at
8 Complete a Canada Vigilance Reporting Form and: - Fax toll-free to , or - Mail to: Canada Vigilance Program Health Canada Postal Locator 1908C Ottawa, Ontario K1A 0K9 Postage paid labels, Canada Vigilance Reporting Form and the adverse reaction reporting guidelines are available on the MedEffect Canada Web site at NOTE: Should you require information related to the management of side effects, contact your health professional. The Canada Vigilance Program does not provide medical advice OVERDOSAGE Overdosage with Levophed may result in headache, severe hypertension, reflex bradycardia, marked increase in peripheral resistance, and decreased cardiac output. In case of accidental overdosage, as evidenced by excessive blood pressure elevation, discontinue Levophed until the condition of the patient stabilizes. For management of a suspected drug overdose, contact your regional Poison Control Centre. DOSAGE AND ADMINISTRATION Restoration of Blood Pressure in Acute Hypotensive States Blood volume depletion should always be corrected as fully as possible before any vasopressor is administered. When, as an emergency measure, intraaortic pressures must be maintained to 8
9 prevent cerebral or coronary artery ischemia, Levophed (norepinephrine bitartrate injection) can be administered before and concurrently with blood volume replacement. Diluent: Levophed solution should be administered in 5 % dextrose solution in distilled water or 5 % dextrose in saline solution. These fluids containing dextrose are protection against significant loss of potency due to oxidation. Administration in saline solution alone is not recommended. Whole blood or plasma, if indicated to increase blood volume, should be administered separately (for example by use of a Y-tube and individual flasks if given simultaneously). Average Dosage: Add 4 ml of Levophed solution to 1000 ml of 5 % dextrose solution. Each 1 ml of this dilution contains 4 mcg of Levophed base. Give this dilution intravenously. Insert a plastic intravenous catheter through a suitable bore needle well advanced centrally into the vein and securely fixed with adhesive tape, avoiding if possible, a catheter tie-in technique as this promotes stasis. A drip bulb is necessary to permit an accurate estimation of the rate of flow in drops per minute. After observing the response to an initial dose of 2 to 3 ml (from 8 to 12 mcg of base) per minute, adjust the rate of flow to establish and maintain a low normal blood pressure {usually 80 to 100 mm Hg systolic) sufficient to maintain the circulation to vital organs, in previously hypertensive patients, it is recommended that the blood pressure should be raised no higher than 40 mm Hg below the preexisting systolic pressure. The average maintenance dose ranges from 0.5 to 1 ml per minute (from 2 to 4 mcg of base). High Dosage: Great individual variation occurs in the dose required to attain and maintain adequate blood pressure. In all cases, dosage of Levophed should be titrated according to response of the patient. Occasionally much larger or even enormous daily doses (as high 68 mg base or 17 ampoules) may be necessary if the patient remains hypotensive, but occult blood volume depletion should always be suspected and corrected when present. Central venous pressure monitoring is usually helpful in detecting and treating this situation. Fluid Intake: The degree of dilution depends on clinical fluid volume requirements. If large volumes of fluid (dextrose) are needed at a flow rate that would involve an excessive dose of the 9
10 pressor agent per unit of time, a more dilute solution than 4 mcg per ml should be used. On the other hand, when large volumes of fluid are clinically undesirable, a concentration greater than 4 mcg per ml may be used. Duration of Therapy: The infusion should be continued until adequate blood pressure and tissue perfusion are maintained without therapy. Levophed infusion should be reduced gradually, avoiding abrupt withdrawal. In some of the reported cases of vascular collapse due to acute myocardial infarction, treatment was required for up to six days. Adjunctive Treatment in Cardiac Arrest Levophed is usually administered intravenously during cardiac resuscitation to restore and maintain an adequate blood pressure after an effective heartbeat and ventilation have been established by other means. (Levophed s powerful beta-adrenergic stimulating action is also thought to increase the strength and effectiveness of systolic contractions once they occur.) Average Dosage: To maintain systemic blood pressure during the management of cardiac arrest. Levophed is used in the same manner as described under Restoration of Blood Pressure in Acute Hypotensive States. HOW SUPPLIED Levophed (norepinephrine bitartrate injection) is supplied in a sterile aqueous solution in the form of the bitartrate. Each ml contains 1 mg norepinephrine bitartrate base and as non-medicinal ingredients, not more than 2 mg sodium metabisulfite and sodium chloride to make isotonic in Water for Injection. The air in the ampoules has been displaced by nitrogen gas. Ampoules of 4 ml, boxes of 10. Caution - Destroy when expired and do not use if the solution is pinkish or darker than slightly yellow or contains a precipitate. Protect from light and freezing. Store ampoules inside carton. 10
11 REFERENCES 1. Zucker. G. et al.: Circulation Nov Sampson. J. and Griffith, G.: Geriatrics 11:60. Feb
LEVOPHED TM 1:1000. Name of medicine. Presentation LEVOPHED TM 1:1000 is a sterile noradrenaline concentrated solution for injection.
 LEVOPHED TM 1:1000 Name of medicine LEVOPHED TM 1:1000 Presentation LEVOPHED TM 1:1000 is a sterile noradrenaline concentrated solution for injection. Each ampoule contains noradrenaline 2 mg in 2 ml (1:1000),
LEVOPHED TM 1:1000 Name of medicine LEVOPHED TM 1:1000 Presentation LEVOPHED TM 1:1000 is a sterile noradrenaline concentrated solution for injection. Each ampoule contains noradrenaline 2 mg in 2 ml (1:1000),
Norepinephrine (Levophed )
 Norepinephrine (Levophed ) Scope C3IFT CCT Generic Name: Norepinephrine Trade Name: Levophed Chemical Class: Therapeutic Class: Actions: Pharmacokinetics: Vasopressor Vasopressor Mechanism of Action: Norepinephrine
Norepinephrine (Levophed ) Scope C3IFT CCT Generic Name: Norepinephrine Trade Name: Levophed Chemical Class: Therapeutic Class: Actions: Pharmacokinetics: Vasopressor Vasopressor Mechanism of Action: Norepinephrine
Neosynephrine. Name of the Medicine
 Name of the Medicine Neosynephrine Phenylephrine hydrochloride 1% injection Neosynephrine Presentation Neosynephrine is a clear, colourless, aqueous solution, free from visible particulates, in sterile
Name of the Medicine Neosynephrine Phenylephrine hydrochloride 1% injection Neosynephrine Presentation Neosynephrine is a clear, colourless, aqueous solution, free from visible particulates, in sterile
PHENTOLAMINE MESYLATE INJECTION SANDOZ STANDARD 5 mg/ ml THERAPEUTIC CLASSIFICATION Alpha-adrenoreceptor Blocker
 PACKAGE INSERT Pr PHENTOLAMINE MESYLATE INJECTION SANDOZ STANDARD 5 mg/ ml THERAPEUTIC CLASSIFICATION Alpha-adrenoreceptor Blocker ACTIONS AND CLINICAL PHARMACOLOGY Phentolamine produces an alpha-adrenergic
PACKAGE INSERT Pr PHENTOLAMINE MESYLATE INJECTION SANDOZ STANDARD 5 mg/ ml THERAPEUTIC CLASSIFICATION Alpha-adrenoreceptor Blocker ACTIONS AND CLINICAL PHARMACOLOGY Phentolamine produces an alpha-adrenergic
PRESCRIBING INFORMATION ADRENALIN TOPICAL SOLUTION. Epinephrine Chloride Topical Solution, manufacturer standard 30 mg / 30 ml (1 mg/ml)
 PRESCRIBING INFORMATION ADRENALIN TOPICAL SOLUTION Epinephrine Chloride Topical Solution, manufacturer standard 30 mg / 30 ml (1 mg/ml) HEMOSTATIC AGENT For Topical Application Only DO NOT INJECT DATE
PRESCRIBING INFORMATION ADRENALIN TOPICAL SOLUTION Epinephrine Chloride Topical Solution, manufacturer standard 30 mg / 30 ml (1 mg/ml) HEMOSTATIC AGENT For Topical Application Only DO NOT INJECT DATE
SUMMARY OF PRODUCT CHARACTERISTICS
 Page 1 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Sinora 1 mg/ml concentrate for solution for infusion. 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml concentrate for solution
Page 1 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Sinora 1 mg/ml concentrate for solution for infusion. 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml concentrate for solution
Titrating Critical Care Medications
 Titrating Critical Care Medications Chad Johnson, MSN (NED), RN, CNCC(C), CNS-cc Clinical Nurse Specialist: Critical Care and Neurosurgical Services E-mail: johnsoc@tbh.net Copyright 2017 1 Learning Objectives
Titrating Critical Care Medications Chad Johnson, MSN (NED), RN, CNCC(C), CNS-cc Clinical Nurse Specialist: Critical Care and Neurosurgical Services E-mail: johnsoc@tbh.net Copyright 2017 1 Learning Objectives
SUMMARY OF PRODUCT CHARACTERISTICS. Each ml solution for injection contains phenylephrine hydrochloride corresponding to 0.1 mg phenylephrine.
 1. NAME OF THE MEDICINAL PRODUCT Fenylefrin Abcur 0.05 mg/ml, solution for injection Fenylefrin Abcur 0.1 mg/ml, solution for injection SUMMARY OF PRODUCT CHARACTERISTICS 2. QUALITATIVE AND QUANTITATIVE
1. NAME OF THE MEDICINAL PRODUCT Fenylefrin Abcur 0.05 mg/ml, solution for injection Fenylefrin Abcur 0.1 mg/ml, solution for injection SUMMARY OF PRODUCT CHARACTERISTICS 2. QUALITATIVE AND QUANTITATIVE
Clinical Competency Name: Post-operative use of Metaraminol in Recovery
 Clinical Competency Name: Post-operative use of Metaraminol in Recovery Trainee name: Title: Ward or department: Method of assessment: Clinical assessor: Name: Title: Professional assessor: Name: Title:
Clinical Competency Name: Post-operative use of Metaraminol in Recovery Trainee name: Title: Ward or department: Method of assessment: Clinical assessor: Name: Title: Professional assessor: Name: Title:
EPINEPHRINE Injection, USP 1:1000 (1 mg/ml) Ampul Protect from light until ready to use.
 EPINEPHRINE Injection, USP 1:1000 (1 mg/ml) Ampul Protect from light until ready to use. DESCRIPTION Epinephrine Injection, USP 1:1000 is a sterile, nonpyrogenic solution. Each ml contains epinephrine
EPINEPHRINE Injection, USP 1:1000 (1 mg/ml) Ampul Protect from light until ready to use. DESCRIPTION Epinephrine Injection, USP 1:1000 is a sterile, nonpyrogenic solution. Each ml contains epinephrine
DBL EPHEDRINE SULFATE INJECTION
 Ephedrine Sulfate USP Description DBL EPHEDRINE SULFATE INJECTION The molecular formula of Ephedrine Sulfate is (C 10 H 15 NO) 2 H 2 SO 4. Its molecular weight is 428.5. The CAS registry number of Ephedrine
Ephedrine Sulfate USP Description DBL EPHEDRINE SULFATE INJECTION The molecular formula of Ephedrine Sulfate is (C 10 H 15 NO) 2 H 2 SO 4. Its molecular weight is 428.5. The CAS registry number of Ephedrine
PRESCRIBING INFORMATION
 PRESCRIBING INFORMATION Bacitracin USP (Bacitracin for Injection USP) For Topical or Intramuscular Use in Solution 50 000 IU Sterile Powder Pfizer Canada Inc Date of Preparation: 17,300 Trans-Canada Highway
PRESCRIBING INFORMATION Bacitracin USP (Bacitracin for Injection USP) For Topical or Intramuscular Use in Solution 50 000 IU Sterile Powder Pfizer Canada Inc Date of Preparation: 17,300 Trans-Canada Highway
DBL NALOXONE HYDROCHLORIDE INJECTION USP
 Name of medicine Naloxone hydrochloride Data Sheet New Zealand DBL NALXNE HYDRCHLRIDE INJECTIN USP Presentation DBL Naloxone Hydrochloride Injection USP is a sterile, clear, colourless solution, free from
Name of medicine Naloxone hydrochloride Data Sheet New Zealand DBL NALXNE HYDRCHLRIDE INJECTIN USP Presentation DBL Naloxone Hydrochloride Injection USP is a sterile, clear, colourless solution, free from
Molecular Formula: C9 H13 NO 2 HCl Molecular Weig ht:
 PHENYLEPHRINE HYDROCHLORIDE- phenylephrine hydrochloride injection, solution Sandoz Inc. Disclaimer: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved
PHENYLEPHRINE HYDROCHLORIDE- phenylephrine hydrochloride injection, solution Sandoz Inc. Disclaimer: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved
DOPamine HYDROCHLORIDE in 5% Dextrose Injection, USP
 DOPamine HYDROCHLORIDE in 5% Dextrose Injection, USP Aqueous Solutions for Acute Correction of Hemodynamics in Shock States Flexible Plastic Container Rx only DESCRIPTION Dopamine Hydrochloride in 5% Dextrose
DOPamine HYDROCHLORIDE in 5% Dextrose Injection, USP Aqueous Solutions for Acute Correction of Hemodynamics in Shock States Flexible Plastic Container Rx only DESCRIPTION Dopamine Hydrochloride in 5% Dextrose
ADULT DRUG REFERENCE Drug Indication Adult Dosage Precautions / Comments
 ADENOSINE Paroxysmal SVT 1 st Dose 6 mg rapid IV 2 nd & 3 rd Doses 12 mg rapid IV push Follow each dose with rapid bolus of 20 ml NS May cause transient heart block or asystole. Side effects include chest
ADENOSINE Paroxysmal SVT 1 st Dose 6 mg rapid IV 2 nd & 3 rd Doses 12 mg rapid IV push Follow each dose with rapid bolus of 20 ml NS May cause transient heart block or asystole. Side effects include chest
PRESCRIBING INFORMATION. Dextrose Injection USP. (Concentrated Dextrose for Intravenous Administration) 50% (500 mg/ml) Fluid and Nutrient Replenisher
 PRESCRIBING INFORMATION Dextrose Injection USP (Concentrated Dextrose for Intravenous Administration) 50% (500 mg/ml) Fluid and Nutrient Replenisher Pfizer Canada Inc. 17300 Trans-Canada Highway Kirkland,
PRESCRIBING INFORMATION Dextrose Injection USP (Concentrated Dextrose for Intravenous Administration) 50% (500 mg/ml) Fluid and Nutrient Replenisher Pfizer Canada Inc. 17300 Trans-Canada Highway Kirkland,
DOBUTamine INJECTION, USP R x only
 DOBUTamine INJECTION, USP R x only DESCRIPTION Dobutamine Injection, USP is 1,2-benzenediol, 4-[2-[[3-(4-hydro-xyphenyl)-1- methylpropyl]amino]ethyl]-hydrochloride, (±). It is a synthetic catecholamine.
DOBUTamine INJECTION, USP R x only DESCRIPTION Dobutamine Injection, USP is 1,2-benzenediol, 4-[2-[[3-(4-hydro-xyphenyl)-1- methylpropyl]amino]ethyl]-hydrochloride, (±). It is a synthetic catecholamine.
CLINICAL PHARMACOLOGY
 EPHEDRINE SULFATE- ephedrine sulfate injection, solution Sandoz Inc Disclaimer: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further
EPHEDRINE SULFATE- ephedrine sulfate injection, solution Sandoz Inc Disclaimer: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further
PRESCRIBING INFORMATION. EPINEPHRINE INJECTION USP 1 mg / ml (ampoules) 0.1 mg / ml (Single-Use Syringes) Sterile Solution.
 PRESCRIBING INFORMATION EPINEPHRINE INJECTION USP 1 mg / ml (ampoules) 0.1 mg / ml (Single-Use Syringes) Sterile Solution (Sympathomimetic) Pfizer Canada Inc. 17300 Trans-Canada Highway Kirkland, Québec
PRESCRIBING INFORMATION EPINEPHRINE INJECTION USP 1 mg / ml (ampoules) 0.1 mg / ml (Single-Use Syringes) Sterile Solution (Sympathomimetic) Pfizer Canada Inc. 17300 Trans-Canada Highway Kirkland, Québec
Physiological Response to Hypovolemic Shock Dr Khwaja Mohammed Amir MD Assistant Professor(Physiology) Objectives At the end of the session the
 Physiological Response to Hypovolemic Shock Dr Khwaja Mohammed Amir MD Assistant Professor(Physiology) Objectives At the end of the session the students should be able to: List causes of shock including
Physiological Response to Hypovolemic Shock Dr Khwaja Mohammed Amir MD Assistant Professor(Physiology) Objectives At the end of the session the students should be able to: List causes of shock including
BETAGAN Allergan Levobunolol HCl Glaucoma Therapy
 BETAGAN Allergan Levobunolol HCl Glaucoma Therapy Action And Clinical Pharmacology: Levobunolol is a noncardioselective beta- adrenoceptor antagonist, equipotent at both beta1 and beta2 receptors. Levobunolol
BETAGAN Allergan Levobunolol HCl Glaucoma Therapy Action And Clinical Pharmacology: Levobunolol is a noncardioselective beta- adrenoceptor antagonist, equipotent at both beta1 and beta2 receptors. Levobunolol
Angina pectoris due to coronary atherosclerosis : Atenolol is indicated for the long term management of patients with angina pectoris.
 Lonet Tablet Description Lonet contains Atenolol, a synthetic β1 selective (cardioselective) adrenoreceptor blocking agent without membrane stabilising or intrinsic sympathomimetic (partial agonist) activity.
Lonet Tablet Description Lonet contains Atenolol, a synthetic β1 selective (cardioselective) adrenoreceptor blocking agent without membrane stabilising or intrinsic sympathomimetic (partial agonist) activity.
CONTRAINDICATIONS None (4)
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use VAZCULEP safely and effectively. See full prescribing information for VAZCULEP. VAZCULEP (phenylephrine
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use VAZCULEP safely and effectively. See full prescribing information for VAZCULEP. VAZCULEP (phenylephrine
BRICANYL INJECTION. terbutaline sulfate PRODUCT INFORMATION
 BRICANYL INJECTION terbutaline sulfate PRODUCT INFORMATION NAME OF THE MEDICINE Terbutaline sulfate, 2-(tert-butylamino)-1-(3,5-dihydroxyphenyl) ethanol sulfate, a sympathomimetic bronchodilator with a
BRICANYL INJECTION terbutaline sulfate PRODUCT INFORMATION NAME OF THE MEDICINE Terbutaline sulfate, 2-(tert-butylamino)-1-(3,5-dihydroxyphenyl) ethanol sulfate, a sympathomimetic bronchodilator with a
PRODUCT MONOGRAPH METHYLENE BLUE INJECTION. (Tetramethylthionine Chloride Trihydrate) Solution, 10 mg/ml USP METHEMOGLOBINEMIA/DIAGNOSTIC AID
 PRODUCT MONOGRAPH METHYLENE BLUE INJECTION (Tetramethylthionine Chloride Trihydrate) Solution, 10 mg/ml USP METHEMOGLOBINEMIA/DIAGNOSTIC AID Alveda Pharmaceuticals Inc. Date of Preparation: 21 St. Clair
PRODUCT MONOGRAPH METHYLENE BLUE INJECTION (Tetramethylthionine Chloride Trihydrate) Solution, 10 mg/ml USP METHEMOGLOBINEMIA/DIAGNOSTIC AID Alveda Pharmaceuticals Inc. Date of Preparation: 21 St. Clair
Core Safety Profile. Date of FAR:
 Core Safety Profile Active substance: Levobupivicaine Pharmaceutical form(s)/strength: Solution for injection, concentrate for solution for infusion, 2,5 mg/ml, 5 mg/ml, 7,5 mg/ml, 0,625 mg/ml, 1,25 mg/ml
Core Safety Profile Active substance: Levobupivicaine Pharmaceutical form(s)/strength: Solution for injection, concentrate for solution for infusion, 2,5 mg/ml, 5 mg/ml, 7,5 mg/ml, 0,625 mg/ml, 1,25 mg/ml
Core Safety Profile. Pharmaceutical form(s)/strength: Film-coated tablets 1.25 mg, 2.5 mg, 3.75 mg, 5 mg, 7.5 mg and 10 mg. Date of FAR:
 Core Safety Profile Active substance: Bisoprolol Pharmaceutical form(s)/strength: Film-coated tablets 1.25 mg, 2.5 mg, 3.75 mg, 5 mg, 7.5 mg and 10 mg P - RMS: FI/H/PSUR/0002/002 Date of FAR: 13.12.2011
Core Safety Profile Active substance: Bisoprolol Pharmaceutical form(s)/strength: Film-coated tablets 1.25 mg, 2.5 mg, 3.75 mg, 5 mg, 7.5 mg and 10 mg P - RMS: FI/H/PSUR/0002/002 Date of FAR: 13.12.2011
Adult Drug Reference. Dopamine Drip Chart. Pediatric Drug Reference. Pediatric Drug Dosage Charts DRUG REFERENCES
 Adult Drug Reference Dopamine Drip Chart Pediatric Drug Reference Pediatric Drug Dosage Charts DRUG REFERENCES ADULT DRUG REFERENCE Drug Indication Adult Dosage Precautions / Comments ADENOSINE Paroxysmal
Adult Drug Reference Dopamine Drip Chart Pediatric Drug Reference Pediatric Drug Dosage Charts DRUG REFERENCES ADULT DRUG REFERENCE Drug Indication Adult Dosage Precautions / Comments ADENOSINE Paroxysmal
PACKAGE LEAFLET: INFORMATION FOR THE USER. Active substance: enoximone
 PACKAGE LEAFLET 1 PACKAGE LEAFLET: INFORMATION FOR THE USER Perfan Injection 100 mg/20 ml Concentrate for Solution for Injection Active substance: enoximone Read all of this leaflet carefully before you
PACKAGE LEAFLET 1 PACKAGE LEAFLET: INFORMATION FOR THE USER Perfan Injection 100 mg/20 ml Concentrate for Solution for Injection Active substance: enoximone Read all of this leaflet carefully before you
Vasoactive Medications. Matthew J. Korobey Pharm.D., BCCCP Critical Care Clinical Specialist Mercy St. Louis
 Vasoactive Medications Matthew J. Korobey Pharm.D., BCCCP Critical Care Clinical Specialist Mercy St. Louis Objectives List components of physiology involved in blood pressure Review terminology related
Vasoactive Medications Matthew J. Korobey Pharm.D., BCCCP Critical Care Clinical Specialist Mercy St. Louis Objectives List components of physiology involved in blood pressure Review terminology related
P-RMS: IE/H/PSUR/0014/002
 Core Safety Profile Active substance: Nitroglycerin Pharmaceutical form(s)/strength: Transdermal patch 25mg, 50mg, 75mg (corresponding to 5, 10 and 15mg per 24 hours respectively P-RMS: IE/H/PSUR/0014/002
Core Safety Profile Active substance: Nitroglycerin Pharmaceutical form(s)/strength: Transdermal patch 25mg, 50mg, 75mg (corresponding to 5, 10 and 15mg per 24 hours respectively P-RMS: IE/H/PSUR/0014/002
PACKAGE INSERT TEMPLATE FOR SALBUTAMOL TABLET & SALBUTAMOL SYRUP
 PACKAGE INSERT TEMPLATE FOR SALBUTAMOL TABLET & SALBUTAMOL SYRUP Brand or Product Name [Product name] Tablet 2mg [Product name] Tablet 4mg [Product name] Syrup 2mg/5ml Name and Strength of Active Substance(s)
PACKAGE INSERT TEMPLATE FOR SALBUTAMOL TABLET & SALBUTAMOL SYRUP Brand or Product Name [Product name] Tablet 2mg [Product name] Tablet 4mg [Product name] Syrup 2mg/5ml Name and Strength of Active Substance(s)
Diagnosis and Management of Acute Myocardial Infarction
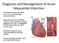 Diagnosis and Management of Acute Myocardial Infarction Acute Myocardial Infarction (AMI) occurs as a result of prolonged myocardial ischemia Atherosclerosis leads to endothelial rupture or erosion that
Diagnosis and Management of Acute Myocardial Infarction Acute Myocardial Infarction (AMI) occurs as a result of prolonged myocardial ischemia Atherosclerosis leads to endothelial rupture or erosion that
DOPamine Hydrochloride and 5% Dextrose Injection, USP
 DOPamine Hydrochloride and 5% Dextrose Injection, USP in Plastic Container VIAFLEX Plus Container DESCRIPTION Dopamine Hydrochloride and 5% Dextrose Injection, USP is a sterile, nonpyrogenic solution of
DOPamine Hydrochloride and 5% Dextrose Injection, USP in Plastic Container VIAFLEX Plus Container DESCRIPTION Dopamine Hydrochloride and 5% Dextrose Injection, USP is a sterile, nonpyrogenic solution of
ADVERSE REACTIONS Most common adverse reactions during treatment: nausea, vomiting, and tachycardia. (6)
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use AKOVAZ safely and effectively. See full prescribing information for AKOVAZ. AKOVAZ (ephedrine sulfate
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use AKOVAZ safely and effectively. See full prescribing information for AKOVAZ. AKOVAZ (ephedrine sulfate
*Sections or subsections omitted from the full prescribing information are not listed.
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use GIAPREZA TM safely and effectively. See full prescribing information for GIAPREZA. GIAPREZA (angiotensin
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use GIAPREZA TM safely and effectively. See full prescribing information for GIAPREZA. GIAPREZA (angiotensin
LESSON ASSIGNMENT Given the trade and/or generic name of an adrenergic blocking agent, classify that agent as either an alpha or beta blocker.
 LESSON ASSIGNMENT LESSON 8 Adrenergic Blocking Agents. TEXT ASSIGNMENT Paragraphs 8-1 through 8-5. LESSON OBJECTIVES 8-1. Given a group of statements, select the statement that best describes one of the
LESSON ASSIGNMENT LESSON 8 Adrenergic Blocking Agents. TEXT ASSIGNMENT Paragraphs 8-1 through 8-5. LESSON OBJECTIVES 8-1. Given a group of statements, select the statement that best describes one of the
PACKAGE LEAFLET: INFORMATION FOR THE USER. EPHEDRINE HCl STEROP 10mg/ml EPHEDRINE HCl STEROP 50mg/ml Solution for injection. Ephedrine Hydrochloride
 PACKAGE LEAFLET: INFORMATION FOR THE USER EPHEDRINE HCl STEROP 10mg/ml EPHEDRINE HCl STEROP 50mg/ml Solution for injection Ephedrine Hydrochloride Read all of this leaflet carefully before you start using
PACKAGE LEAFLET: INFORMATION FOR THE USER EPHEDRINE HCl STEROP 10mg/ml EPHEDRINE HCl STEROP 50mg/ml Solution for injection Ephedrine Hydrochloride Read all of this leaflet carefully before you start using
Contra Costa County Emergency Medical Services Drug Reference. Indication Dosing Cautions Comments
 Drug Adenosine Albuterol Indication Dosing Cautions Comments Narrow complex tachycardia Bronchospasm Crush injury - hyperkalemia Initial 6mg rapid IV Repeat 12mg rapid IV Follow each dose with 20ml NS
Drug Adenosine Albuterol Indication Dosing Cautions Comments Narrow complex tachycardia Bronchospasm Crush injury - hyperkalemia Initial 6mg rapid IV Repeat 12mg rapid IV Follow each dose with 20ml NS
IMPORTANT: PLEASE READ
 PART III: CONSUMER INFORMATION ALPROLIX [pronounced all prō liks] Coagulation Factor IX (Recombinant), Fc Fusion Protein This leaflet is part III of a three-part "Product Monograph" published when ALPROLIX
PART III: CONSUMER INFORMATION ALPROLIX [pronounced all prō liks] Coagulation Factor IX (Recombinant), Fc Fusion Protein This leaflet is part III of a three-part "Product Monograph" published when ALPROLIX
PRODUCT INFORMATION ALCAINE. Proparacaine Hydrochloride Sterile Ophthalmic Solution, USP. 5 mg/ml. Topical Anesthetic
 PRODUCT INFORMATION ALCAINE Proparacaine Hydrochloride Sterile Ophthalmic Solution, USP 5 mg/ml Topical Anesthetic Alcon Canada Inc. 2665 Meadowpine Blvd. Mississauga, ON L5N 8C7 www.alcon.ca Date of Preparation:
PRODUCT INFORMATION ALCAINE Proparacaine Hydrochloride Sterile Ophthalmic Solution, USP 5 mg/ml Topical Anesthetic Alcon Canada Inc. 2665 Meadowpine Blvd. Mississauga, ON L5N 8C7 www.alcon.ca Date of Preparation:
(angiotensin II) injection for intravenous infusion
 ADMINISTERING GIAPREZA TM (angiotensin II) injection for intravenous infusion Visit www.giapreza.com INITIATE Recommended starting dose of GIAPREZA is 20 ng/kg/min, which is equivalent to 0.02 mcg/kg/min
ADMINISTERING GIAPREZA TM (angiotensin II) injection for intravenous infusion Visit www.giapreza.com INITIATE Recommended starting dose of GIAPREZA is 20 ng/kg/min, which is equivalent to 0.02 mcg/kg/min
(Dalteparin Sodium Injection)
 PART III: CONSUMER INFORMATION Pr FRAGMIN (Dalteparin Sodium Injection) This leaflet is part III of a three-part "Product Monograph" published when FRAGMIN was approved for sale in Canada and is designed
PART III: CONSUMER INFORMATION Pr FRAGMIN (Dalteparin Sodium Injection) This leaflet is part III of a three-part "Product Monograph" published when FRAGMIN was approved for sale in Canada and is designed
PACKAGE LEAFLET: INFORMATION FOR THE USER. Glucose Intravenous Infusion BP 10% w/v solution for infusion Glucose (as glucose monohydrate)
 PACKAGE LEAFLET: INFORMATION FOR THE USER Glucose Intravenous Infusion BP 10% w/v solution for infusion Glucose (as glucose monohydrate) Read all of this leaflet carefully before you start using this medicine
PACKAGE LEAFLET: INFORMATION FOR THE USER Glucose Intravenous Infusion BP 10% w/v solution for infusion Glucose (as glucose monohydrate) Read all of this leaflet carefully before you start using this medicine
COMPANY CORE PACKAGE INSERT CCPI (PI/CORE/ENGLISH)
 COMPANY CORE PACKAGE INSERT CCPI (PI/CORE/ENGLISH) HUMAN ALBUMIN 20 % BEHRING Rev.: 05-MAR-2008 / PEI approval 26.02.08 Supersedes previous versions Rev.: 28-NOV-2007 / Adaptation to Core SPC Rev.: 02-JAN-2007
COMPANY CORE PACKAGE INSERT CCPI (PI/CORE/ENGLISH) HUMAN ALBUMIN 20 % BEHRING Rev.: 05-MAR-2008 / PEI approval 26.02.08 Supersedes previous versions Rev.: 28-NOV-2007 / Adaptation to Core SPC Rev.: 02-JAN-2007
Systemic Pharmacology Lecture 7: Neuropharmacology
 Systemic Pharmacology Lecture 7: Neuropharmacology Drugs act on Sympathetic NS (adrenergic system) Adrenergic Drugs (Sympathomimetics), adrenergic agonists, or alpha- and beta-adrenergic agonists Antiadrenergic
Systemic Pharmacology Lecture 7: Neuropharmacology Drugs act on Sympathetic NS (adrenergic system) Adrenergic Drugs (Sympathomimetics), adrenergic agonists, or alpha- and beta-adrenergic agonists Antiadrenergic
NAPHCON-A Eye Drops naphazoline hydrochloride 0.025% and pheniramine maleate 0.3%.
 NEW ZEALAND DATA SHEET 1. PRODUCT NAME NAPHCON-A Eye Drops naphazoline hydrochloride 0.025% and pheniramine maleate 0.3%. 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Naphcon-A contains naphazoline hydrochloride
NEW ZEALAND DATA SHEET 1. PRODUCT NAME NAPHCON-A Eye Drops naphazoline hydrochloride 0.025% and pheniramine maleate 0.3%. 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Naphcon-A contains naphazoline hydrochloride
What would be the response of the sympathetic system to this patient s decrease in arterial pressure?
 CASE 51 A 62-year-old man undergoes surgery to correct a herniated disc in his spine. The patient is thought to have an uncomplicated surgery until he complains of extreme abdominal distention and pain
CASE 51 A 62-year-old man undergoes surgery to correct a herniated disc in his spine. The patient is thought to have an uncomplicated surgery until he complains of extreme abdominal distention and pain
ALS MODULE 7 Pharmacology
 ALS MODULE 7 Pharmacology Relates to HLT404C Apply Advanced Resuscitation Techniques Introduction There are no studies that addressed the order of drug administration. There is inadequate evidence to define
ALS MODULE 7 Pharmacology Relates to HLT404C Apply Advanced Resuscitation Techniques Introduction There are no studies that addressed the order of drug administration. There is inadequate evidence to define
PRESCRIBING INFORMATION WITH CONSUMER INFORMATION MYDRIACYL. tropicamide ophthalmic solution, USP. 0.5% and 1% w/v.
 PRESCRIBING INFORMATION WITH CONSUMER INFORMATION Pr MYDRIACYL tropicamide ophthalmic solution, USP 0.5% and 1% w/v Anticholinergic Alcon Canada Inc. 2665 Meadowpine Blvd. Mississauga, ON L5N 8C7 www.alcon.ca
PRESCRIBING INFORMATION WITH CONSUMER INFORMATION Pr MYDRIACYL tropicamide ophthalmic solution, USP 0.5% and 1% w/v Anticholinergic Alcon Canada Inc. 2665 Meadowpine Blvd. Mississauga, ON L5N 8C7 www.alcon.ca
Intravenous Infusions
 Intravenous Infusions 1) An IV insulin infusion can be used for patients: a) with out of control diabetes b) with DKA (Diabetic Ketoacidosis) c) after a heart attack 2) Hyperglycemia is an adaptive response
Intravenous Infusions 1) An IV insulin infusion can be used for patients: a) with out of control diabetes b) with DKA (Diabetic Ketoacidosis) c) after a heart attack 2) Hyperglycemia is an adaptive response
Taking the shock factor out of shock
 Taking the shock factor out of shock Julie Antonellis, BS, LVT, VTS (ECC) Northern Virginia Regional Director for the VALVT Technician Supervisor VCA Animal Emergency Critical Care Business owner Antonellis
Taking the shock factor out of shock Julie Antonellis, BS, LVT, VTS (ECC) Northern Virginia Regional Director for the VALVT Technician Supervisor VCA Animal Emergency Critical Care Business owner Antonellis
Swans and Pressors. Vanderbilt Surgery Summer School Ricky Shinall
 Swans and Pressors Vanderbilt Surgery Summer School Ricky Shinall Shock, Swans, Pressors in 15 minutes 4 Reasons for Shock 4 Swan numbers to know 7 Pressors =15 things to know 4 Reasons for Shock Not enough
Swans and Pressors Vanderbilt Surgery Summer School Ricky Shinall Shock, Swans, Pressors in 15 minutes 4 Reasons for Shock 4 Swan numbers to know 7 Pressors =15 things to know 4 Reasons for Shock Not enough
Swans and Pressors. Vanderbilt Surgery Summer School Ricky Shinall
 Swans and Pressors Vanderbilt Surgery Summer School Ricky Shinall SHOCK Hypotension SHOCK Hypotension SHOCK=Reduction of systemic tissue perfusion, resulting in decreased oxygen delivery to the tissues.
Swans and Pressors Vanderbilt Surgery Summer School Ricky Shinall SHOCK Hypotension SHOCK Hypotension SHOCK=Reduction of systemic tissue perfusion, resulting in decreased oxygen delivery to the tissues.
EMT. Chapter 14 Review
 EMT Chapter 14 Review Review 1. All of the following are common signs and symptoms of cardiac ischemia, EXCEPT: A. headache. B. chest pressure. C. shortness of breath. D. anxiety or restlessness. Review
EMT Chapter 14 Review Review 1. All of the following are common signs and symptoms of cardiac ischemia, EXCEPT: A. headache. B. chest pressure. C. shortness of breath. D. anxiety or restlessness. Review
Adrenaline 1mg in 10mL (1:10,000) Pre-filled syringe 3 Amiodarone 300mg/10mL Pre-filled syringe 5
 Quick Reference Guide for: Cardiac Arrest Medicines Box (BLUE) Please Note: Any medicines given must form part of an Airway, Breathing, Circulation, Disability and Exposure (ABCDE) Assessment (9)999 must
Quick Reference Guide for: Cardiac Arrest Medicines Box (BLUE) Please Note: Any medicines given must form part of an Airway, Breathing, Circulation, Disability and Exposure (ABCDE) Assessment (9)999 must
Physiology and Pharmacology
 Pharmacokinetics Physiology and Pharmacology Pharmacokinetics of Local Anesthetics Uptake Oral Route Topical Route Injection Distribution Metabolism (Biotransformation) Excretion Uptake Vasoactivity Local
Pharmacokinetics Physiology and Pharmacology Pharmacokinetics of Local Anesthetics Uptake Oral Route Topical Route Injection Distribution Metabolism (Biotransformation) Excretion Uptake Vasoactivity Local
Nothing to Disclose. Severe Pulmonary Hypertension
 Severe Ronald Pearl, MD, PhD Professor and Chair Department of Anesthesiology Stanford University Rpearl@stanford.edu Nothing to Disclose 65 year old female Elective knee surgery NYHA Class 3 Aortic stenosis
Severe Ronald Pearl, MD, PhD Professor and Chair Department of Anesthesiology Stanford University Rpearl@stanford.edu Nothing to Disclose 65 year old female Elective knee surgery NYHA Class 3 Aortic stenosis
PRESCRIBING INFORMATION with the Consumer Information FLUORESCITE* fluorescein injection, USP. fluorescein 10% w/v (as fluorescein sodium) Sterile
 PRESCRIBING INFORMATION with the Consumer Information FLUORESCITE* fluorescein injection, USP fluorescein 10% w/v (as fluorescein sodium) Sterile Diagnostic Agent Alcon Canada Inc. 2665 Meadowpine Blvd.
PRESCRIBING INFORMATION with the Consumer Information FLUORESCITE* fluorescein injection, USP fluorescein 10% w/v (as fluorescein sodium) Sterile Diagnostic Agent Alcon Canada Inc. 2665 Meadowpine Blvd.
PRESCRIBING INFORMATION. Sodium Bicarbonate Injection USP 42mg/ml (4.2%), 75mg/ml (7.5%), 84mg/ml (8.4%) Sterile solution ALKALIZER
 PRESCRIBING INFORMATION Sodium Bicarbonate Injection USP 42mg/ml (4.2%), 75mg/ml (7.5%), 84mg/ml (8.4%) Sterile solution Lifeshield ABBOJECT Single-dose Syringes and Flipp Vial ALKALIZER FOR CORRECTION
PRESCRIBING INFORMATION Sodium Bicarbonate Injection USP 42mg/ml (4.2%), 75mg/ml (7.5%), 84mg/ml (8.4%) Sterile solution Lifeshield ABBOJECT Single-dose Syringes and Flipp Vial ALKALIZER FOR CORRECTION
VENTOLIN RESPIRATOR SOLUTION
 VENTOLIN RESPIRATOR SOLUTION Salbutamol QUALITATIVE AND QUANTITATIVE COMPOSITION VENTOLIN Respirator Solution contains 5mg salbutamol, as sulphate, per ml of solution and is supplied in 10 ml bottles.
VENTOLIN RESPIRATOR SOLUTION Salbutamol QUALITATIVE AND QUANTITATIVE COMPOSITION VENTOLIN Respirator Solution contains 5mg salbutamol, as sulphate, per ml of solution and is supplied in 10 ml bottles.
READ THIS FOR SAFE AND EFFECTIVE USE OF YOUR MEDICINE PATIENT MEDICATION INFORMATION. (em-plis-city)
 READ THIS FOR SAFE AND EFFECTIVE USE OF YOUR MEDICINE PATIENT MEDICATION INFORMATION Pr Empliciti TM (em-plis-city) (elotuzumab 300 & 400 mg powder for concentrate for solution for infusion) Read this
READ THIS FOR SAFE AND EFFECTIVE USE OF YOUR MEDICINE PATIENT MEDICATION INFORMATION Pr Empliciti TM (em-plis-city) (elotuzumab 300 & 400 mg powder for concentrate for solution for infusion) Read this
P-RMS: NO/H/PSUR/0009/001
 Core Safety Profile Active substance: Propofol Pharmaceutical form(s)/strength: Emulsion for injection, infusion, 10mg/ml Emulsion for infusion, 20mg/ml P-RMS: NO/H/PSUR/0009/001 Date of FAR: 30.06.2011
Core Safety Profile Active substance: Propofol Pharmaceutical form(s)/strength: Emulsion for injection, infusion, 10mg/ml Emulsion for infusion, 20mg/ml P-RMS: NO/H/PSUR/0009/001 Date of FAR: 30.06.2011
NORMOSOL -R MULTIPLE ELECTROLYTES INJECTION TYPE 1, USP For Replacing Acute Losses of Extracellular Fluid Flexible Plastic Container
 NORMOSOL -R MULTIPLE ELECTROLYTES INJECTION TYPE 1, USP For Replacing Acute Losses of Extracellular Fluid Flexible Plastic Container R x only DESCRIPTION Normosol-R is a sterile, nonpyrogenic isotonic
NORMOSOL -R MULTIPLE ELECTROLYTES INJECTION TYPE 1, USP For Replacing Acute Losses of Extracellular Fluid Flexible Plastic Container R x only DESCRIPTION Normosol-R is a sterile, nonpyrogenic isotonic
PACKAGE LEAFLET: INFORMATION FOR THE PATIENT. Dobutamine 12.5mg/ml Concentrate for Solution for Infusion Dobutamine Hydrochloride
 PACKAGE LEAFLET: INFORMATION FOR THE PATIENT Dobutamine 12.5mg/ml Concentrate for Solution for Infusion Dobutamine Hydrochloride Read all of this leaflet carefully before you start using this medicine
PACKAGE LEAFLET: INFORMATION FOR THE PATIENT Dobutamine 12.5mg/ml Concentrate for Solution for Infusion Dobutamine Hydrochloride Read all of this leaflet carefully before you start using this medicine
Salapin: Salbutamol BP 2mg as sulphate in each 5mL of a raspberry cola flavoured, sugar free syrup.
 Salapin Salbutamol Syrup 2mg/5mL Qualitative and quantitative composition Salapin: Salbutamol BP 2mg as sulphate in each 5mL of a raspberry cola flavoured, sugar free syrup. Clinical particulars Therapeutic
Salapin Salbutamol Syrup 2mg/5mL Qualitative and quantitative composition Salapin: Salbutamol BP 2mg as sulphate in each 5mL of a raspberry cola flavoured, sugar free syrup. Clinical particulars Therapeutic
READ THIS FOR SAFE AND EFFECTIVE USE OF YOUR MEDICINE PATIENT MEDICATION INFORMATION. IOPIDINE 1% Apraclonidine Ophthalmic Solution, USP
 READ THIS FOR SAFE AND EFFECTIVE USE OF YOUR MEDICINE PATIENT MEDICATION INFORMATION Pr IOPIDINE 1% Apraclonidine Ophthalmic Solution, USP Read this carefully before you start taking IOPIDINE 1% and each
READ THIS FOR SAFE AND EFFECTIVE USE OF YOUR MEDICINE PATIENT MEDICATION INFORMATION Pr IOPIDINE 1% Apraclonidine Ophthalmic Solution, USP Read this carefully before you start taking IOPIDINE 1% and each
CSL Behring LLC Albuminar -25 US Package Insert Albumin (Human) USP, 25% Revised: 01/2008 Page 1
 Page 1 CSL Behring Albuminar -25 Albumin (Human) USP, 25% R x only DESCRIPTION Albuminar -25, Albumin (Human) 25%, is a sterile aqueous solution of albumin obtained from large pools of adult human venous
Page 1 CSL Behring Albuminar -25 Albumin (Human) USP, 25% R x only DESCRIPTION Albuminar -25, Albumin (Human) 25%, is a sterile aqueous solution of albumin obtained from large pools of adult human venous
PACKAGE LEAFLET: INFORMATION FOR THE USER. CALCICLO STEROP 11 meq 10 ml Solution for injection. Dihydrated Calcium Chloride
 PACKAGE LEAFLET: INFORMATION FOR THE USER CALCICLO STEROP 11 meq 10 ml Solution for injection Dihydrated Calcium Chloride Read all of this leaflet carefully before you start using this medicine because
PACKAGE LEAFLET: INFORMATION FOR THE USER CALCICLO STEROP 11 meq 10 ml Solution for injection Dihydrated Calcium Chloride Read all of this leaflet carefully before you start using this medicine because
SYMJEPI TM (epinephrine) injection, for intramuscular or subcutaneous use Initial U.S. Approval: 1939
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use SYMJEPI TM safely and effectively. See full prescribing information for SYMJEPI TM. SYMJEPI TM (epinephrine)
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use SYMJEPI TM safely and effectively. See full prescribing information for SYMJEPI TM. SYMJEPI TM (epinephrine)
Instil one or two drops in the conjunctival sac(s) every three to four hours as needed.
 NEW ZEALAND DATA SHEET 1. PRODUCT NAME NAPHCON FORTE Eye Drops 0.1% 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Naphcon Forte contains naphazoline hydrochloride 1.0 mg in 1 ml (0.1%). Excipient with known
NEW ZEALAND DATA SHEET 1. PRODUCT NAME NAPHCON FORTE Eye Drops 0.1% 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Naphcon Forte contains naphazoline hydrochloride 1.0 mg in 1 ml (0.1%). Excipient with known
Shock is defined as a state of cellular and tissue hypoxia due to : reduced oxygen delivery and/or increased oxygen consumption or inadequate oxygen
 Shock is defined as a state of cellular and tissue hypoxia due to : reduced oxygen delivery and/or increased oxygen consumption or inadequate oxygen utilization The effects of shock are initially reversible
Shock is defined as a state of cellular and tissue hypoxia due to : reduced oxygen delivery and/or increased oxygen consumption or inadequate oxygen utilization The effects of shock are initially reversible
PRESCRIBING INFORMATION. DOPAMINE HYDROCHLORIDE AND 5% DEXTROSE INJECTION, USP (Dopamine Hydrochloride and 5% Dextrose) Sympathomimetic Agent
 PRESCRIBING INFORMATION DOPAMINE HYDROCHLORIDE AND 5% DEXTROSE INJECTION, USP (Dopamine Hydrochloride and 5% Dextrose) 800 :g/ml, 1600 :g/ml and 3200 :g/ml Sympathomimetic Agent Hospira Healthcare Corporation
PRESCRIBING INFORMATION DOPAMINE HYDROCHLORIDE AND 5% DEXTROSE INJECTION, USP (Dopamine Hydrochloride and 5% Dextrose) 800 :g/ml, 1600 :g/ml and 3200 :g/ml Sympathomimetic Agent Hospira Healthcare Corporation
Composition Each ml of Ventol solution for inhalation contains 5 mg Salbutamol (as sulphate).
 VENTOL Composition Each ml of Ventol solution for inhalation contains 5 mg Salbutamol (as sulphate). Respiratory Solution Action Salbutamol is a short-acting, relatively selective beta2-adrenoceptor agonist.
VENTOL Composition Each ml of Ventol solution for inhalation contains 5 mg Salbutamol (as sulphate). Respiratory Solution Action Salbutamol is a short-acting, relatively selective beta2-adrenoceptor agonist.
READ THIS FOR SAFE AND EFFECTIVE USE OF YOUR MEDICINE PATIENT MEDICATION INFORMATION. (Pentazocine Lactate Injection, USP)
 READ THIS FOR SAFE AND EFFECTIVE USE OF YOUR MEDICINE PATIENT MEDICATION INFORMATION N Talwin (Pentazocine Lactate Injection, USP) Read this carefully before you start taking Talwin. This leaflet is a
READ THIS FOR SAFE AND EFFECTIVE USE OF YOUR MEDICINE PATIENT MEDICATION INFORMATION N Talwin (Pentazocine Lactate Injection, USP) Read this carefully before you start taking Talwin. This leaflet is a
Autonomic Nervous System (ANS) وحدة اليوزبكي Department of Pharmacology- College of Medicine- University of Mosul
 Autonomic Nervous System (ANS) د. م. أ. وحدة اليوزبكي Department of Pharmacology- College of Medicine- University of Mosul Sympathetic (Adrenergic) nervous system 3 Objectives At end of this lecture, the
Autonomic Nervous System (ANS) د. م. أ. وحدة اليوزبكي Department of Pharmacology- College of Medicine- University of Mosul Sympathetic (Adrenergic) nervous system 3 Objectives At end of this lecture, the
a)-catecholamines // these are compounds which have the catechol nucleus as adrenaline, noradrenaline, isoprenaline, dopamine, dobutamine
 1 Adrenergic or sympathetic agonist: Are classified according to: I)- according to the chemical structure a)-catecholamines // these are compounds which have the catechol nucleus as adrenaline, noradrenaline,
1 Adrenergic or sympathetic agonist: Are classified according to: I)- according to the chemical structure a)-catecholamines // these are compounds which have the catechol nucleus as adrenaline, noradrenaline,
PACKAGE INSERT CALCIUM GLUCONATE INJECTION, USP 10% Electrolyte Replenisher mosmol/ml 680 mosmol/l Ca meq/ml ph
 CALCIUM GLUCONATE INJECTION, USP 10% Electrolyte Replenisher 0.68 mosmol/ml 680 mosmol/l Ca ++ 0.465 meq/ml ph 6.0 8.2 DESCRIPTION Calcium Gluconate Injection, USP is a sterile, nonpyrogenic supersaturated
CALCIUM GLUCONATE INJECTION, USP 10% Electrolyte Replenisher 0.68 mosmol/ml 680 mosmol/l Ca ++ 0.465 meq/ml ph 6.0 8.2 DESCRIPTION Calcium Gluconate Injection, USP is a sterile, nonpyrogenic supersaturated
Document 3 Summary of Data and Guidance for Medical Professionals
 Document 3 Summary of Data and Guidance for Medical Professionals Anvirzel Anvirzel is a novel antitumor compound extracted from the plant Nerium oleander, which belongs to the family Apocynaceae. Anvirzel
Document 3 Summary of Data and Guidance for Medical Professionals Anvirzel Anvirzel is a novel antitumor compound extracted from the plant Nerium oleander, which belongs to the family Apocynaceae. Anvirzel
The adrenergic drugs affect receptors that are stimulated by norepinephrine or epinephrine. Some adrenergic drugs act directly on the adrenergic
 Adrenergic drugs The adrenergic drugs affect receptors that are stimulated by norepinephrine or epinephrine. Some adrenergic drugs act directly on the adrenergic receptor (adrenoceptor) by activating it
Adrenergic drugs The adrenergic drugs affect receptors that are stimulated by norepinephrine or epinephrine. Some adrenergic drugs act directly on the adrenergic receptor (adrenoceptor) by activating it
DBL DOBUTAMINE HYDROCHLORIDE INJECTION
 Name of medicine Dobutamine Hydrochloride DBL DOBUTAMINE HYDROCHLORIDE INJECTION Data Sheet New Zealand Presentation DBL Dobutamine Hydrochloride Injection is a sterile solution containing in each 20 ml
Name of medicine Dobutamine Hydrochloride DBL DOBUTAMINE HYDROCHLORIDE INJECTION Data Sheet New Zealand Presentation DBL Dobutamine Hydrochloride Injection is a sterile solution containing in each 20 ml
PRESCRIBING INFORMATION
 PRESCRIBING INFORMATION Pr ATROPINE SULFATE INJECTION USP Anticholinergic (spasmolytic agent) Sandoz Canada Inc. Date of revision: March 4, 2011 145 Jules-Léger Boucherville, QC, Canada J4B 7K8 Control
PRESCRIBING INFORMATION Pr ATROPINE SULFATE INJECTION USP Anticholinergic (spasmolytic agent) Sandoz Canada Inc. Date of revision: March 4, 2011 145 Jules-Léger Boucherville, QC, Canada J4B 7K8 Control
-Cardiogenic: shock state resulting from impairment or failure of myocardium
 Shock chapter Shock -Condition in which tissue perfusion is inadequate to deliver oxygen, nutrients to support vital organs, cellular function -Affects all body systems -Classic signs of early shock: Tachycardia,tachypnea,restlessness,anxiety,
Shock chapter Shock -Condition in which tissue perfusion is inadequate to deliver oxygen, nutrients to support vital organs, cellular function -Affects all body systems -Classic signs of early shock: Tachycardia,tachypnea,restlessness,anxiety,
Surgical Care at the District Hospital. EMERGENCY & ESSENTIAL SURGICAL CARE
 Surgical Care at the District Hospital 1 13 Resuscitation and Preparation for Anesthesia & Surgery Key Points 2 13.1 Management of Emergencies and Cardiopulmonary Resuscitation The emergency measures that
Surgical Care at the District Hospital 1 13 Resuscitation and Preparation for Anesthesia & Surgery Key Points 2 13.1 Management of Emergencies and Cardiopulmonary Resuscitation The emergency measures that
Platelet aggregation inhibitor. Cardiac chest pain or suspected Myocardial Infarction.
 s Aspirin Platelet aggregation inhibitor. Anti-inflammatory agent and an inhibitor of platelet function. Useful agent in the treatment of various thromboembolic diseases such as acute myocardial infarction.
s Aspirin Platelet aggregation inhibitor. Anti-inflammatory agent and an inhibitor of platelet function. Useful agent in the treatment of various thromboembolic diseases such as acute myocardial infarction.
BRIMOPRESS-T EYE DROPS. COMPOSITION Each ml of BRIMOPRESS-T EYE DROPS contains. Brimonidine Tartarate 0.2% Timolol Maleate 0.5%
 BRIMOPRESS-T EYE DROPS COMPOSITION Each ml of BRIMOPRESS-T EYE DROPS contains Brimonidine Tartarate 0.2% Timolol Maleate 0.5% BRIMOPRESS-T ophthalmic solution is a combination of Brimonidine Tartrate,
BRIMOPRESS-T EYE DROPS COMPOSITION Each ml of BRIMOPRESS-T EYE DROPS contains Brimonidine Tartarate 0.2% Timolol Maleate 0.5% BRIMOPRESS-T ophthalmic solution is a combination of Brimonidine Tartrate,
PRESCRIBING INFORMATION AND CONSUMER INFORMATION ISOPTO * CARPINE. pilocarpine hydrochloride ophthalmic solution, USP.
 PRESCRIBING INFORMATION AND CONSUMER INFORMATION Pr ISOPTO * CARPINE pilocarpine hydrochloride ophthalmic solution, USP 1%, 2% and 4% w/v Parasympathomimetic Alcon Canada Inc. 2665 Meadowpine Blvd. Mississauga,
PRESCRIBING INFORMATION AND CONSUMER INFORMATION Pr ISOPTO * CARPINE pilocarpine hydrochloride ophthalmic solution, USP 1%, 2% and 4% w/v Parasympathomimetic Alcon Canada Inc. 2665 Meadowpine Blvd. Mississauga,
Public Assessment Report Scientific discussion. Nepipe Junior, Nepipe (former Adrenalin MEDA) (adrenaline) SE/H/1287/01-02/DC
 Public Assessment Report Scientific discussion Nepipe Junior, Nepipe (former Adrenalin MEDA) (adrenaline) SE/H/1287/01-02/DC This module reflects the scientific discussion for the approval of Nepipe Junior
Public Assessment Report Scientific discussion Nepipe Junior, Nepipe (former Adrenalin MEDA) (adrenaline) SE/H/1287/01-02/DC This module reflects the scientific discussion for the approval of Nepipe Junior
The Pharmacology of Hypotension: Vasopressor Choices for HIE patients. Keliana O Mara, PharmD August 4, 2018
 The Pharmacology of Hypotension: Vasopressor Choices for HIE patients Keliana O Mara, PharmD August 4, 2018 Objectives Review the pathophysiology of hypotension in neonates Discuss the role of vasopressors
The Pharmacology of Hypotension: Vasopressor Choices for HIE patients Keliana O Mara, PharmD August 4, 2018 Objectives Review the pathophysiology of hypotension in neonates Discuss the role of vasopressors
Bisoprolol Fumarate 2.5 mg, 5 mg and 10 mg Tablet
 Bisoprolol Fumarate 2.5 mg, 5 mg and 10 mg Tablet Description Betapro is a preparation of Bisoprolol Fumarate. Bisoprolol is used alone or in combination with other medications to treat high blood pressure.
Bisoprolol Fumarate 2.5 mg, 5 mg and 10 mg Tablet Description Betapro is a preparation of Bisoprolol Fumarate. Bisoprolol is used alone or in combination with other medications to treat high blood pressure.
BETALOL Esmolol hydrochloride HIKMA PHARMACEUTICALS
 09-15 BETALOL Esmolol hydrochloride HIKMA PHARMACEUTICALS ACTION Esmolol hydrochloride is a beta 1-selective (cardioselective) adrenergic receptor blocking agent with rapid onset, a very short duration
09-15 BETALOL Esmolol hydrochloride HIKMA PHARMACEUTICALS ACTION Esmolol hydrochloride is a beta 1-selective (cardioselective) adrenergic receptor blocking agent with rapid onset, a very short duration
Package leaflet: Information for the user
 Package leaflet: Information for the user Emerade 150 micrograms solution for injection in pre-filled pen Emerade 300 micrograms solution for injection in pre-filled pen Emerade 500 micrograms solution
Package leaflet: Information for the user Emerade 150 micrograms solution for injection in pre-filled pen Emerade 300 micrograms solution for injection in pre-filled pen Emerade 500 micrograms solution
HYPOTENSION IS DANGEROUS C. R Y A N K E A Y, M D, F A C E P 1 6 M A R C H
 HYPOTENSION IS DANGEROUS C. R Y A N K E A Y, M D, F A C E P 1 6 M A R C H 2 0 1 8 OBJECTIVES Case-based overview of pressors Debunking pressor myths Utilizing push-dose pressors CASE 1 82-year old male,
HYPOTENSION IS DANGEROUS C. R Y A N K E A Y, M D, F A C E P 1 6 M A R C H 2 0 1 8 OBJECTIVES Case-based overview of pressors Debunking pressor myths Utilizing push-dose pressors CASE 1 82-year old male,
Intermediate Medications. Epinephrine cardiac Epinephrine anaphylaxis Dextrose Atropine Narcan Thiamine Albuterol
 Intermediate Medications Epinephrine cardiac Epinephrine anaphylaxis Dextrose Atropine Narcan Thiamine Albuterol Needle Handling Precautions Minimize the tasks performed in a moving ambulance Balance the
Intermediate Medications Epinephrine cardiac Epinephrine anaphylaxis Dextrose Atropine Narcan Thiamine Albuterol Needle Handling Precautions Minimize the tasks performed in a moving ambulance Balance the
Chapter 13. Learning Objectives. Learning Objectives 9/11/2012. Poisonings, Overdoses, and Intoxications
 Chapter 13 Poisonings, Overdoses, and Intoxications Learning Objectives Discuss use of activated charcoal in treatment of poisonings List treatment options for acetaminophen overdose List clinical manifestations
Chapter 13 Poisonings, Overdoses, and Intoxications Learning Objectives Discuss use of activated charcoal in treatment of poisonings List treatment options for acetaminophen overdose List clinical manifestations
HOW LOW CAN YOU GO? HYPOTENSION AND THE ANESTHETIZED PATIENT.
 HOW LOW CAN YOU GO? HYPOTENSION AND THE ANESTHETIZED PATIENT. Donna M. Sisak, CVT, LVT, VTS (Anesthesia/Analgesia) Seattle Veterinary Specialists Kirkland, WA dsisak@svsvet.com THE ANESTHETIZED PATIENT
HOW LOW CAN YOU GO? HYPOTENSION AND THE ANESTHETIZED PATIENT. Donna M. Sisak, CVT, LVT, VTS (Anesthesia/Analgesia) Seattle Veterinary Specialists Kirkland, WA dsisak@svsvet.com THE ANESTHETIZED PATIENT
DRUG GUIDELINE. HYDRALAZINE (Intravenous severe hypertension in pregnancy)
 DRUG GUIDELINE HYDRALAZINE (Intravenous severe hypertension SCOPE (Area): FOR USE IN: Labour Ward, HDU, Theatre and ED EXCLUSIONS: Paediatrics (seek Paediatrician advice) and other general wards. SCOPE
DRUG GUIDELINE HYDRALAZINE (Intravenous severe hypertension SCOPE (Area): FOR USE IN: Labour Ward, HDU, Theatre and ED EXCLUSIONS: Paediatrics (seek Paediatrician advice) and other general wards. SCOPE
