Performance Characteristics of Eight Estradiol Immunoassays
|
|
|
- Avice West
- 5 years ago
- Views:
Transcription
1 Clinical Chemistry / EVALUATION OF EIGHT ESTRADIOL IMMUNOASSAYS Performance Characteristics of Eight Estradiol Immunoassays David T. Yang, MD, 1 William E. Owen, MT(ASCP), 2 Carol S. Ramsay, 3 Hui Xie, MS, 3 and William L. Roberts, MD, PhD, 1,2 Key Words: Gas chromatography; Mass spectrometry; Accuracy; Imprecision DOI: 1.139/5N2R4HT4GMAGPBY Abstract Measurement of estradiol is useful in assisted reproduction, evaluation of infertility, menopause, and male feminization. The analytic performance of 8 estradiol immunoassays was evaluated. The imprecision and accuracy of the Access, ADVIA Centaur, ARCHITECT i2, AutoDELFIA, Elecsys 21, IMMULITE 2, and Vitros ECi estradiol assays (see text for proprietary information) were evaluated by using an isotope dilution gas chromatography mass spectrometry (ID-GC-MS) reference method. The coefficient of variation (CV) ranged from 6.9% on the Elecsys 21 to 42.6% on the ADVIA Centaur at an estradiol concentration of 18 pg/ml (66 pmol/l), with the ARCHITECT i2 assay in development and the Vitros ECi having a CV below 1% at this estradiol concentration. Agreement between the automated assays and ID-GC-MS was variable, with slopes ranging from.87 to 1.2. The Access, ARCHITECT i2 in development, and the IMMULITE 2 were the most accurate, with slopes of.99,.98, and 1.3, respectively. These findings indicate that the ARCHITECT i2 estradiol assay in development had the best precision and accuracy of the assays evaluated for measurement of serum estradiol concentrations. Estradiol, a steroid hormone, is the most potent of the naturally occurring estrogens, and in the nonpregnant female, it is derived almost exclusively from the ovaries. Estradiol is responsible for the development and maintenance of the female sex organs and secondary sex characteristics. It also has important roles in regulation of the menstrual cycle and in maintenance of pregnancy. Estradiol measurements frequently are used during evaluation of female infertility and menopause and feminization in males and in assisted reproduction to monitor follicular development. 1 Physiologic levels are low, often less than 5 pg/ml (184 pmol/l), in men, children, and postmenopausal women and can range widely, from 5 to 41 pg/ml (18-1,55 pmol/l), in menstruating women. 2 The wide reference intervals and the clinical applications of this assay dictate the need for high sensitivity, excellent accuracy and precision through a wide analytical range, and a prompt turnaround time. Concern regarding these requirements, accuracy in particular, led to efforts toward standardization of serum estradiol measurements through a reference serum panel with estradiol concentrations determined by isotope dilution gas chromatography mass spectrometry (ID-GC-MS). 3,4 Recently, estradiol assays have become available on a number of fully automated immunoassay analyzers. While these systems provide rapid turnaround times and high throughput, there presently are only limited reports regarding the analytic performance of these assays, and most do not assess accuracy with ID-GC-MS as the reference method. 5-8 The aim of our study was to assess the imprecision and accuracy of 8 automated estradiol assays with ID-GC-MS as the reference method. 332 Am J Clin Pathol 24;122: DOI: 1.139/5N2R4HT4GMAGPBY
2 Clinical Chemistry / ORIGINAL ARTICLE Materials and Methods The following automated methods and instruments were evaluated: the Access analyzer (Beckman Coulter, Brea, CA), the ADVIA Centaur analyzer (Bayer Diagnostics, Tarrytown, NY), the ARCHITECT i2 analyzer using current reagents and reagents for a method that is in development (Abbott Diagnostics, Abbott Park, IL), the AutoDELFIA automatic immunoassay system (Wallac Oy, Turku, Finland), the Elecsys 21 analyzer (Roche Diagnostics, Indianapolis, IN), the IMMULITE 2 analyzer (Diagnostics Products, Los Angeles, CA), and the Vitros ECi analyzer (Ortho-Clinical Diagnostics, Raritan, NJ). All of these immunoassay methods use a competitive format. All methods use rabbit antiestradiol antibodies except for the Vitros ECi assay, which uses both rabbit and sheep antiestradiol antibodies. The ARCHITECT i2 in development assay requires 15 µl of sample compared with 1 µl for the current assay and has a comparable resistance to interference by hemolysis, icterus, and lipemia. The Elecsys 21 assay adds mesterolone to release estradiol from the sample. Assay imprecision was assessed by assaying aliquots of 5 serum pools with estradiol concentrations of 18, 31, 93, 387, and 846 pg/ml (66, 114, 341, 1,42, and 3,15 pmol/l, respectively) in 6 replicates each on 2 different days for a total of 12 measurements. The estradiol concentration of these samples was determined by ID-GC-MS as previously described. 9 Functional sensitivity, defined as the estradiol concentration that would produce a coefficient of variation (CV) of 1%, was determined from a power curve fit of the CV plotted against the estradiol concentration determined by ID-GC-MS. A method comparison study between each of the 8 estradiol assays and the ID-GC-MS reference method was performed using 61 samples with estradiol concentrations ranging from to 1,11 pg/ml (-4,41 pmol/l). EP Evaluator Release 5 software (David G. Rhoads Associates, Kennett Square, PA) was used for simple precision calculations and Deming regression analysis. For Deming regression analysis, the same SD was used for each method. Results A summary of data from the imprecision study is shown in Table 1. CVs ranged from 1.2% to 42.6% for estradiol concentrations from 18 to 846 pg/ml (66-3,15 pmol/l). At 18 pg/ml (66 pmol/l), the range of CV observed for each method was broad, ranging from 6.9% on the Elecsys 21 to 42.6% on the ADVIA Centaur. At 93 pg/ml, CVs became more uniform and ranged from 2.5% on the ARCHITECT i2 in development to 9.4% on the IMMULITE 2. The functional sensitivities for a cutoff CV of 1% were determined from the data and are listed in Table 2. The ARCHI- TECT i2 in development, Elecsys 21, and Vitros ECi are capable of determining estradiol concentrations as low as 18 pg/ml (66 pmol/l) while maintaining CVs of less than 1%. The remaining instruments had functional sensitivities ranging from 35 to 8 pg/ml ( pmol/l). It is noteworthy that the mean estradiol concentrations obtained by some immunoassay methods for the sample with the lowest estradiol concentration were substantially higher than the ID- GC-MS reference method. Comparison of results obtained for samples collected from individual subjects using the 8 automated estradiol immunoassays and the reference ID-GC-MS method by Deming regression analysis is shown Figure 1. There were varying degrees of assay accuracy apparent, with slopes ranging from.87 to 1.2. The Access, ARCHI- TECT i2 in development, and IMMULITE 2 assays had the best accuracy with slopes of.99,.98, and 1.3 respectively. The AutoDELFIA and Elecsys 21 assays showed the poorest accuracy with slopes of.87 and 1.2, respectively. Difference plots for the same data are shown Figure 2. The dispersion of data around the regression line, as reflected in the standard error of the estimate (S y/x ) and the correlation coefficient (r) and represented graphically in the difference plot, showed that the ARCHITECT i2 assay in development had the best agreement with the reference method with an S y/x of 23 pg/ml (84 pmol/l), r =.997, followed by the Elecsys 21 method with an S y/x of 28 pg/ml (13 pmol/l) and r =.997. The ADVIA Centaur had the largest amount of dispersion with an S y/x of 66 pg/ml (242 pmol/l) and r =.98. Discussion Our protocol for assessing imprecision was abbreviated owing to limited quantities of serum assayed by the ID-GC- MS reference method. Therefore, the true overall imprecision of the estradiol immunoassays during multiple days of analysis and multiple reagent lots is likely to be substantially higher than our data would indicate. Nevertheless, our results reflect the relative imprecision of each method. Of the 8 estradiol immunoassays evaluated, the ARCHITECT i2 assay in development, Elecsys 21, and Vitros ECi methods were the most precise at low estradiol concentrations, being able to quantify estradiol concentrations as low as 18 pg/ml (66 pmol/l) while maintaining CVs of less than 1%. The Access, ARCHITECT i2 assay in development, and IMMULITE 2 had the best average results compared with the reference method, with Am J Clin Pathol 24;122: DOI: 1.139/5N2R4HT4GMAGPBY 333
3 Yang et al / EVALUATION OF EIGHT ESTRADIOL IMMUNOASSAYS Table 1 Summary of Imprecision Data * Estradiol Concentration (pg/ml) Method Serum Pool ID-GC-MS Method Mean Coefficient of Variation (%) Access ADVIA Centaur , ARCHITECT i >1, Not determined ARCHITECT i2 in development AutoDELFIA Elecsys , IMMULITE Vitros ECi ID-GC-MS, isotope dilution gas chromatography mass spectrometry. * Values are given in conventional units; for conversion to Système International units (pmol/l), multiply by For proprietary information, see the text. Table 2 Estimated Functional Sensitivity * Assay Functional Sensitivity (pg/ml) Access 37 ADVIA Centaur 116 (61) ARCHITECT i2 35 ARCHITECT i2 in development <18 AutoDELFIA 74 (57) Elecsys 21 <18 IMMULITE 2 8 Vitros ECi <18 * The functional sensitivity was defined as the estradiol concentration that would produce a coefficient of variation (CV) of 1%. It was estimated from a power curve fit of the data in Table 1 as shown in Figure 1. For proprietary information, see the text. Inspection of the power curves in Figure 1 revealed that the concentration of estradiol corresponding to a CV of 1% was overestimated for the ADVIA Centaur and AutoDELFIA methods. The imprecision data for the 18, 31, and 93 pg/ml samples were refitted with a power curve, and the result shown in parentheses provides a better estimate of the true functional sensitivity of these assays. slopes very close to 1.. The remainder of the instruments showed varying degrees of bias and possibly could benefit from improved calibration. This is especially true for the Elecsys 21, which demonstrated excellent precision but had a positive proportional bias. Ovarian stimulation protocols require serial monitoring of estradiol levels to direct dosage adjustments of gonadotropins for optimization of follicular growth and prevention of ovarian overstimulation. 1,11 Most of these decision points are between estradiol concentrations of 2 and 2, pg/ml (734-7,34 pmol/l), 1 and all 8 instruments have adequate precision within this range with CVs of less than 1%. Before initiating an ovarian stimulation protocol, differentiating populations with poor stimulation 334 Am J Clin Pathol 24;122: DOI: 1.139/5N2R4HT4GMAGPBY
4 Clinical Chemistry / ORIGINAL ARTICLE A B C D Access (pg/ml) ADVIA Centaur (pg/ml) ARCHITECT i2 (pg/ml) ARCHITECT i2 in Development (pg/ml) E F G H AutoDELFIA (pg/ml) Elecsys 21 (pg/ml) IMMULITE 2 (pg/ml) Vitros ECi (pg/ml) Figure 1 Comparison of estradiol immunoassay and isotope dilution gas chromatography mass spectrometry (ID-GC-MS) results using Deming regression. The solid lines are from Deming regression analysis, and the dashed lines are x = y. Data are given for the assay compared with the ID-GC-MS reference method. A, Access: slope,.99; intercept, 21 pg/ml; standard error of the estimate (S y/x ), 38 pg/ml; r =.992. B, ADVIA Centaur: slope, 1.1; intercept, 9 pg/ml; S y/x, 66 pg/ml; r =.98. C, ARCHITECT i2: slope, 1.12; intercept, 41 pg/ml; S y/x, 4 pg/ml; r =.993. D, ARCHITECT i2 in development: slope,.98; intercept, 1 pg/ml; S y/x, 23 pg/ml; r =.997. E, AutoDELFIA: slope,.87; intercept, 17 pg/ml; S y/x, 32 pg/ml; r =.993. F, Elecsys 21: slope, 1.2; intercept, 1 pg/ml; S y/x, 28 pg/ml; r =.997. G, IMMULITE 2: slope, 1.3; intercept, 7 pg/ml; S y/x, 36 pg/ml; r =.993. H, Vitros ECi: slope,.92; intercept, 9 pg/ml; S y/x, 32 pg/ml; r =.993. GC-MS, gas chromatography mass spectrometry. Intercept and S y/x values are given in conventional units; for conversion to Système International units (pmol/l), multiply by For proprietary information, see the text. characteristics from those with good stimulation characteristics is a prognostic necessity to select the appropriate stimulation protocol. This can be done by measuring basal levels of estradiol and follicle stimulating hormone on day 3 of the menstrual cycle, and the determination point is an estradiol concentration of 6 pg/ml (22 pmol/l). 12 All of the assays except the IMMULITE 2 demonstrated what we consider to be adequate precision at low estradiol concentrations for this task (CV <1%). While the Access, ARCHITECT i2 assay in development, and IMMULITE 2 assays demonstrated good concordance with the reference method, additional calibration standardization of the ADVIA Centaur, AutoDELFIA, Elecsys 21, and Vitros ECi assays might improve their accuracy and could lead to better harmonization of estradiol results. Additional studies to examine interfering substances, particularly endogenous and exogenous estrogens, need to be performed because the presence of some potential crossreacting substances could contribute to the bias and scatter observed for some of the methods. The clinical applications of estradiol assays can be diverse. This study shows that the ARCHITECT i2 assay in development, Elecsys 21, and Vitros ECi assays have adequate low-end precision for the measurement of estradiol levels in men and postmenopausal women, but the accuracy of the Elecsys 21 method needs some improvement. The rapid analysis time and low technical demands of automated immunoassays have made these instruments popular in the field of assisted reproduction. Within the range of estradiol concentrations typically encountered in this application, the Access, ARCHITECT i2 in development, and Vitros ECi assays seem to have adequate precision and accuracy. The Am J Clin Pathol 24;122: DOI: 1.139/5N2R4HT4GMAGPBY 335
5 Yang et al / EVALUATION OF EIGHT ESTRADIOL IMMUNOASSAYS A B C D Access ADVIA Centaur ARCHITECT i ARCHITECT i2 in Development E F G H AutoDELFIA Elecsys IMMULITE Vitros ECi Figure 2 Comparison of estradiol immunoassays with isotope dilution gas chromatography mass spectrometry by difference plots. The solid lines are the ± 2 SD limits of agreement, and the dashed lines are the mean bias. All values are given in conventional units (pg/ml); for conversion to Système International units (pmol/l), multiply by A, Access: mean bias, +2; limits of agreement, 55 and +95. B, ADVIA Centaur: mean bias, +34; limits of agreement, 14 and C, ARCHITECT i2: mean bias, +7; limits of agreement, 32 and D, ARCHITECT i2 in development: mean bias, 5; limits of agreement, 54 and +44. E, AutoDELFIA: mean bias, 17; limits of agreement, 122 and +89. F, Elecsys 21: mean bias, 48; limits of agreement, 79 and G, IMMULITE 2: mean bias, ; limits of agreement, 72 and +72. H, Vitros ECi: mean bias, 29; limits of agreement, 113 and +54. GC-MS, gas chromatography mass spectrometry. For proprietary information, see the text. changes made to the reagents for the ARCHITECT i2 in development have resulted in an improvement in assay performance compared with the current ARCHITECT i2 reagents. The IMMULITE 2, while accurate, lacked adequate precision at low estradiol concentrations for this application, and, conversely, the Elecsys 21, while precise, lacked the requisite accuracy for this application. From the 1 Department of Pathology, University of Utah Health Sciences Center, Salt Lake City; 2 ARUP Institute for Clinical and Experimental Pathology, Salt Lake City; and 3 Abbott Diagnostics Division, Abbott Park, IL. Supported by the Diagnostics Division of Abbott Laboratories and the ARUP Institute for Clinical and Experimental Pathology. Address reprint requests to Dr Roberts: ARUP Laboratories, 5 Chipeta Way, Salt Lake City, UT References 1. Gronowski AM, Landau-Levine M. Reproductive endocrine function. In: Burtis CA, Ashwood ER, eds. Tietz Textbook of Clinical Chemistry. 3rd ed. Philadelphia, PA: Saunders; 1999: Painter PC, Cope JY, Smith JL. Reference information for the clinical laboratory. In: Burtis CA, Ashwood ER, eds. Tietz Textbook of Clinical Chemistry. 3rd ed. Philadelphia, PA: Saunders; 1999: Thienpont L. Meeting report: first and second estradiol international workshops [letter]. Clin Chem. 1996;42: Thienpont LM, De Leenheer AP. Efforts by industry toward standardization of serum estradiol-17β measurements. Clin Chem. 1998;44: Levesque A, Letellier M, Dillon P, et al. Analytical performance of Bayer Immuno I estradiol and progesterone assays. Clin Chem. 1997;43: Am J Clin Pathol 24;122: DOI: 1.139/5N2R4HT4GMAGPBY
6 Clinical Chemistry / ORIGINAL ARTICLE 6. Dancoine F, Couplet G, Buvat J, et al. Analytical and clinical evaluation of the Immulite estradiol assay in serum from patients undergoing in vitro fertilization: estradiol increase in mature follicles. Clin Chem. 1997;43: Rodriguez-Espinoza J, Otal-Entraigas C, Gascon-Roche N, et al. Analytical and clinical performance of an automated immunoassay system (Immulite) for estradiol in serum. Clin Chem Lab Med. 1998;36: Reinsberg J, Jost E. Analytical performance of the fully automated AxSYM estradiol assay. Clin Chem Lab Med. 2;38: Wu H, Ramsay C, Ozaeta P, et al. Serum estradiol quantified by isotope dilution gas chromatography/mass spectrometry. Clin Chem. 22;48: Maclin VM. Ovarian stimulation and ovulation induction. In: Keel BA, May JV, De Jonge CJ, eds. Handbook of the Assisted Reproduction Laboratory. Boca Raton, FL: CRC Press; 2: Navot D, Bergh PA, Laufer N. Ovarian hyperstimulation syndrome in novel reproductive technologies: prevention and treatment. Fertil Steril. 1992;58: Evers JL, Slaats P, Land JA, et al. Elevated levels of basal estradiol-17β predict poor response in patients with normal basal levels of follicle-stimulating hormone undergoing in vitro fertilization. Fertil Steril. 1998;69: Am J Clin Pathol 24;122: DOI: 1.139/5N2R4HT4GMAGPBY 337
Comparison of Five Automated Serum and Whole Blood Folate Assays
 Clinical Chemistry / FIVE AUTOMATED FOLATE ASSAYS Comparison of Five Automated Serum and Whole Blood Folate Assays William E. Owen, MT(ASCP), 1 and William L. Roberts, MD, PhD 2 Key Words: Hemolysate;
Clinical Chemistry / FIVE AUTOMATED FOLATE ASSAYS Comparison of Five Automated Serum and Whole Blood Folate Assays William E. Owen, MT(ASCP), 1 and William L. Roberts, MD, PhD 2 Key Words: Hemolysate;
Immunoassay of Estradiol: Unanticipated Suppression by Unconjugated Estriol
 Clinical Chemistry 50:1 160 165 (2004) Endocrinology and Metabolism Immunoassay of Estradiol: Unanticipated Suppression by Unconjugated Estriol Zhimin (Tim) Cao, 1* Thomas A. Swift, 2 Clint A. West, 1
Clinical Chemistry 50:1 160 165 (2004) Endocrinology and Metabolism Immunoassay of Estradiol: Unanticipated Suppression by Unconjugated Estriol Zhimin (Tim) Cao, 1* Thomas A. Swift, 2 Clint A. West, 1
Intermethod Differences in Results for Total PSA, Free PSA, and Percentage of Free PSA
 Clinical Chemistry / PSA and Free PSA Method Differences Intermethod Differences in Results for Total PSA, Free PSA, and Percentage of Free PSA Patricia R. Slev, PhD, Sonia L. La ulu, and William L. Roberts,
Clinical Chemistry / PSA and Free PSA Method Differences Intermethod Differences in Results for Total PSA, Free PSA, and Percentage of Free PSA Patricia R. Slev, PhD, Sonia L. La ulu, and William L. Roberts,
Chapter 2. High-sensitivity C-reactive protein methods examined
 Chapter High-sensitivity C-reactive protein methods examined Snježana Rothkrantz-Kos, Maria PJ Schmitz, Otto Bekers, Paul PCA Menheere, Marja P van Dieijen-Visser Clin Chem ;48:359-6 3 Chapter Abstract
Chapter High-sensitivity C-reactive protein methods examined Snježana Rothkrantz-Kos, Maria PJ Schmitz, Otto Bekers, Paul PCA Menheere, Marja P van Dieijen-Visser Clin Chem ;48:359-6 3 Chapter Abstract
IDEXX Catalyst One Chemistry Analyzer for In-house Measurement of Total Thyroxine (TT 4 ) Concentration in Serum from Dogs and Cats
 IDEXX Catalyst One Chemistry Analyzer for In-house Measurement of Total Thyroxine (TT 4 ) Concentration in Serum from Dogs and Cats Authors: Kate Cote, Ph.D., Graham Bilbrough, MA, VetMB, CertVA, MRCVS,
IDEXX Catalyst One Chemistry Analyzer for In-house Measurement of Total Thyroxine (TT 4 ) Concentration in Serum from Dogs and Cats Authors: Kate Cote, Ph.D., Graham Bilbrough, MA, VetMB, CertVA, MRCVS,
Determination of hemoglobin is one of the most commonly
 ORIGINAL ARTICLE Multiple-Site Analytic Evaluation of a New Portable Analyzer, HemoCue Hb 201+, for Point-of-Care Testing Sten-Erik Bäck, PhD,* Carl G. M. Magnusson, PhD, Lena K. Norlund, MD, PhD, Henning
ORIGINAL ARTICLE Multiple-Site Analytic Evaluation of a New Portable Analyzer, HemoCue Hb 201+, for Point-of-Care Testing Sten-Erik Bäck, PhD,* Carl G. M. Magnusson, PhD, Lena K. Norlund, MD, PhD, Henning
Analytic Bias of Thyroid Function Tests. Analysis of a College of American Pathologists Fresh Frozen Serum Pool by 3900 Clinical Laboratories
 Analytic Bias of Thyroid Function Tests Analysis of a College of American Pathologists Fresh Frozen Serum Pool by 3900 Clinical Laboratories Bernard W. Steele, MD; Edward Wang, PhD; George G. Klee, MD,
Analytic Bias of Thyroid Function Tests Analysis of a College of American Pathologists Fresh Frozen Serum Pool by 3900 Clinical Laboratories Bernard W. Steele, MD; Edward Wang, PhD; George G. Klee, MD,
USING THE ACCESS AMH ASSAY IN YOUR LABORATORY
 INFORMATION BULLETIN USING THE ACCESS AMH ASSAY IN YOUR LABORATORY ///////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////
INFORMATION BULLETIN USING THE ACCESS AMH ASSAY IN YOUR LABORATORY ///////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////////
ACCESS hstni SCIENTIFIC LITERATURE
 ACCESS hstni SCIENTIFIC LITERATURE 2017 2018 Table of contents Performance Evaluation of Access hstni A critical evaluation of the Beckman Coulter Access hstni: Analytical performance, reference interval
ACCESS hstni SCIENTIFIC LITERATURE 2017 2018 Table of contents Performance Evaluation of Access hstni A critical evaluation of the Beckman Coulter Access hstni: Analytical performance, reference interval
Evaluation of the LIAISON 25 OH Vitamin D TOTAL Assay on the new LIAISON XL analyser
 Evaluation of the LIAISON 25 OH Vitamin D TOTAL Assay on the new LIAISON XL analyser Prof. Etienne Cavalier Department of Clinical Chemistry University of Liège, CHU Sart-Tilman Liège, Belgium etienne.cavalier@chu.ulg.ac.be
Evaluation of the LIAISON 25 OH Vitamin D TOTAL Assay on the new LIAISON XL analyser Prof. Etienne Cavalier Department of Clinical Chemistry University of Liège, CHU Sart-Tilman Liège, Belgium etienne.cavalier@chu.ulg.ac.be
In Vitro Diagnostic Glucose Test System
 CDRH Final Guidance 2-Page Cover Sheet Guidance for Industry In Vitro Diagnostic Glucose Test System Document issued on: July 6, 1998 U.S. Department Of Health and Human Services Food and Drug Administration
CDRH Final Guidance 2-Page Cover Sheet Guidance for Industry In Vitro Diagnostic Glucose Test System Document issued on: July 6, 1998 U.S. Department Of Health and Human Services Food and Drug Administration
Fertility Diagnostics
 Fertility Diagnostics Fertility hormones measured on PATHFAST For internal use only Diagnostics PATHFAST Chemiluminescence-immuno-analyzer 1 Content: page 1. Fertility hormones - general aspects 1.1 Reproductive
Fertility Diagnostics Fertility hormones measured on PATHFAST For internal use only Diagnostics PATHFAST Chemiluminescence-immuno-analyzer 1 Content: page 1. Fertility hormones - general aspects 1.1 Reproductive
Establishment of Reference Intervals for Thyroid- Stimulating Hormone and Free Thyroxine in Amniotic Fluid Using the Bayer ADVIA Centaur
 Clinical Chemistry / THYROID HORMONES IN AMNIOTIC FLUID Establishment of Reference Intervals for Thyroid- Stimulating Hormone and Free Thyroxine in Amniotic Fluid Using the Bayer ADVIA Centaur Nikola A.
Clinical Chemistry / THYROID HORMONES IN AMNIOTIC FLUID Establishment of Reference Intervals for Thyroid- Stimulating Hormone and Free Thyroxine in Amniotic Fluid Using the Bayer ADVIA Centaur Nikola A.
Keywords: albuminuria; albumin/creatinine ratio (ACR); measurements. Introduction
 Clin Chem Lab Med 2015; 53(11): 1737 1743 Beryl E. Jacobson, David W. Seccombe*, Alex Katayev and Adeera Levin A study examining the bias of albumin and albumin/creatinine ratio measurements in urine DOI
Clin Chem Lab Med 2015; 53(11): 1737 1743 Beryl E. Jacobson, David W. Seccombe*, Alex Katayev and Adeera Levin A study examining the bias of albumin and albumin/creatinine ratio measurements in urine DOI
Endocrinology of the Female Reproductive Axis
 Endocrinology of the Female Reproductive Axis girlontheriver.com Geralyn Lambert-Messerlian, PhD, FACB Professor Women and Infants Hospital Alpert Medical School at Brown University Women & Infants BROWN
Endocrinology of the Female Reproductive Axis girlontheriver.com Geralyn Lambert-Messerlian, PhD, FACB Professor Women and Infants Hospital Alpert Medical School at Brown University Women & Infants BROWN
Table. Analytical characteristics of commercial and research cardiac troponin I and T assays declared by the manufacturer.
 Table. Analytical characteristics of commercial and research cardiac troponin I and T assays declared by the manufacturer. Commercially available assays - Company/ platform(s)/ assay LoB a LoD b 99 th
Table. Analytical characteristics of commercial and research cardiac troponin I and T assays declared by the manufacturer. Commercially available assays - Company/ platform(s)/ assay LoB a LoD b 99 th
Shyamali Pal. Page 315 ejifcc2017vol28no4pp R B Diagnostic Private Limited
 Evaluation of the correlation coefficient of polyethylene glycol treated and direct prolactin results and comparability with different assay system results Shyamali Pal R B Diagnostic Private Limited INFO
Evaluation of the correlation coefficient of polyethylene glycol treated and direct prolactin results and comparability with different assay system results Shyamali Pal R B Diagnostic Private Limited INFO
The Virtues and Pitfalls of Implementing a New Test
 The Virtues and Pitfalls of Implementing a New Test James H. Nichols, Ph.D., DABCC, FACB Professor of Clinical Pathology, Microbiology and Immunology Associate Medical Director for Clinical Operations
The Virtues and Pitfalls of Implementing a New Test James H. Nichols, Ph.D., DABCC, FACB Professor of Clinical Pathology, Microbiology and Immunology Associate Medical Director for Clinical Operations
Harmonisation of TFTs
 Royal Prince Alfred Hospital and University of Sydney Harmonisation of TFTs Harmonisation Workshop May 2015 Paul Williams Harmonisation May 2015 Talk Outline IFCC initiatives C-STFT (Committee for standardisation
Royal Prince Alfred Hospital and University of Sydney Harmonisation of TFTs Harmonisation Workshop May 2015 Paul Williams Harmonisation May 2015 Talk Outline IFCC initiatives C-STFT (Committee for standardisation
Cortisol Assays. The Good, The Bad and The Indifferent. David Ducroq. Cardiff and Vale ulhb WEQAS. Unit 6, Parc Tŷ Glas. Llanishen.
 Cortisol Assays The Good, The Bad and The Indifferent David Ducroq Cardiff and Vale ulhb WEQAS Unit 6, Parc Tŷ Glas Llanishen Cardiff www.weqas.com Summary Brief overview of current methods and challenges
Cortisol Assays The Good, The Bad and The Indifferent David Ducroq Cardiff and Vale ulhb WEQAS Unit 6, Parc Tŷ Glas Llanishen Cardiff www.weqas.com Summary Brief overview of current methods and challenges
Multicenter Comparison of Four Automated Immunoassay Analyzers for Prostate Specific Antigen
 Letter to the Editor Clinical Chemistry Ann Lab Med 19;39:6-41 https://doi.org/1.3343/alm.19.39.4.6 ISSN 2234-36 eissn 2234-3814 Multicenter Comparison of Four Automated Immunoassay Analyzers for Prostate
Letter to the Editor Clinical Chemistry Ann Lab Med 19;39:6-41 https://doi.org/1.3343/alm.19.39.4.6 ISSN 2234-36 eissn 2234-3814 Multicenter Comparison of Four Automated Immunoassay Analyzers for Prostate
Analytic Performance of the iq200 Automated Urine Microscopy Analyzer and Comparison With Manual Counts Using Fuchs-Rosenthal Cell Chambers
 Clinical Chemistry / PERFORMANCE OF THE IQ2 URINALYSIS SYSTEM Analytic Performance of the iq2 Automated Urine Microscopy Analyzer and Comparison With Manual Counts Using Fuchs-Rosenthal Cell Chambers David
Clinical Chemistry / PERFORMANCE OF THE IQ2 URINALYSIS SYSTEM Analytic Performance of the iq2 Automated Urine Microscopy Analyzer and Comparison With Manual Counts Using Fuchs-Rosenthal Cell Chambers David
Reproductive FSH. Analyte Information
 Reproductive FSH Analyte Information 1 Follicle-stimulating hormone Introduction Follicle-stimulating hormone (FSH, also known as follitropin) is a glycoprotein hormone secreted by the anterior pituitary
Reproductive FSH Analyte Information 1 Follicle-stimulating hormone Introduction Follicle-stimulating hormone (FSH, also known as follitropin) is a glycoprotein hormone secreted by the anterior pituitary
N. Shirazian, MD. Endocrinologist
 N. Shirazian, MD Internist, Endocrinologist Inside the ovary Day 15-28: empty pyfollicle turns into corpus luteum (yellow body) Immature eggs Day 1-13: 13: egg developing inside the growing follicle Day
N. Shirazian, MD Internist, Endocrinologist Inside the ovary Day 15-28: empty pyfollicle turns into corpus luteum (yellow body) Immature eggs Day 1-13: 13: egg developing inside the growing follicle Day
Gentian Canine CRP Immunoassay Application Note for Abbott Architect * c4000
 v2-jan214 Gentian Canine CRP Immunoassay Application Note for Abbott Architect * c4 Intended Use The canine CRP immunoassay on Abbott's Architect c4 is an in vitro diagnostic test for quantitative determination
v2-jan214 Gentian Canine CRP Immunoassay Application Note for Abbott Architect * c4 Intended Use The canine CRP immunoassay on Abbott's Architect c4 is an in vitro diagnostic test for quantitative determination
Standardization and Certification of the ADVIA Centaur Vitamin D Total Assay
 Standardization and Certification of the ADVIA Centaur Vitamin D Total Assay Authors: James Freeman Paul Sibley, PhD Neil Parker, MSc Ryan Spears Kimberly S. Wilson H. Roma Levy, MS Affiliations: Siemens
Standardization and Certification of the ADVIA Centaur Vitamin D Total Assay Authors: James Freeman Paul Sibley, PhD Neil Parker, MSc Ryan Spears Kimberly S. Wilson H. Roma Levy, MS Affiliations: Siemens
CASE STUDY 3: PSA-STANDARDISATION & VARIABILITY COMPARATIVE STUDY IN HOSPITALS
 CASE STUDY 3: PSA-STANDARDISATION & VARIABILITY COMPARATIVE STUDY IN HOSPITALS Dr Ophelia Blake FRCPath University Hospital Limerick, IRELAND JCTLM Meeting BIPM, Sevres,France 4 th December 2013 Prostate
CASE STUDY 3: PSA-STANDARDISATION & VARIABILITY COMPARATIVE STUDY IN HOSPITALS Dr Ophelia Blake FRCPath University Hospital Limerick, IRELAND JCTLM Meeting BIPM, Sevres,France 4 th December 2013 Prostate
Ultra-Sensitive Estradiol lumelisa Catalog No. GWB-AEB745, legacy id (96 Tests)
 For Research Use Only. Not for use in Diagnostic Procedures. INTENDED USE The GenWay, Inc. Ultra Sensitive Estradiol (E2) (Chemiluminescence Enzyme Linked Immunosorbent Assay) is used for the ultra sensitive
For Research Use Only. Not for use in Diagnostic Procedures. INTENDED USE The GenWay, Inc. Ultra Sensitive Estradiol (E2) (Chemiluminescence Enzyme Linked Immunosorbent Assay) is used for the ultra sensitive
Method Comparison Report Semi-Annual 1/5/2018
 Method Comparison Report Semi-Annual 1/5/2018 Prepared for Carl Commissioner Regularatory Commission 123 Commission Drive Anytown, XX, 12345 Prepared by Dr. Mark Mainstay Clinical Laboratory Kennett Community
Method Comparison Report Semi-Annual 1/5/2018 Prepared for Carl Commissioner Regularatory Commission 123 Commission Drive Anytown, XX, 12345 Prepared by Dr. Mark Mainstay Clinical Laboratory Kennett Community
Toward Standardization of Insulin Immunoassays
 Clinical Chemistry 55:5 1011 1018 (2009) General Clinical Chemistry Toward Standardization of Insulin Immunoassays W. Greg Miller, 1* Linda M. Thienpont, 2 Katleen Van Uytfanghe, 2 Penelope M. Clark, 3
Clinical Chemistry 55:5 1011 1018 (2009) General Clinical Chemistry Toward Standardization of Insulin Immunoassays W. Greg Miller, 1* Linda M. Thienpont, 2 Katleen Van Uytfanghe, 2 Penelope M. Clark, 3
ImmunoCAP Systems. Optimized for allergy testing - Precision - Accuracy - Consistency
 ImmunoCAP Systems Optimized for allergy testing - Precision - Accuracy - Consistency 1 The ImmunoCAP systems from Phadia offer an optimal allergy testing solution for every laboratory, regardless of testing
ImmunoCAP Systems Optimized for allergy testing - Precision - Accuracy - Consistency 1 The ImmunoCAP systems from Phadia offer an optimal allergy testing solution for every laboratory, regardless of testing
lbt lab tests t Conrolled Ovarian Hyperstimulation Dr Soheila Ansaripour
 lbt lab tests t and Conrolled Ovarian Hyperstimulation Dr Soheila Ansaripour Research Instituteof Avicenna 4/23/2012 Why good prediction of poor response good prediction i of OHSS application appropriate
lbt lab tests t and Conrolled Ovarian Hyperstimulation Dr Soheila Ansaripour Research Instituteof Avicenna 4/23/2012 Why good prediction of poor response good prediction i of OHSS application appropriate
1. PROTOCOL. Comparison Study Summary. Tuality Healthcare 324 SE 9 th Ave. Suite E Hillsboro, OR July 30, 2014
 Comparison Study Summary Tuality Healthcare 324 SE 9 th Ave. Suite E Hillsboro, OR 97123 July 30, 2014 1. PROTOCOL This study was conducted on July 24 th, 2014 at Tuality Healthcare, Hillsboro, OR. The
Comparison Study Summary Tuality Healthcare 324 SE 9 th Ave. Suite E Hillsboro, OR 97123 July 30, 2014 1. PROTOCOL This study was conducted on July 24 th, 2014 at Tuality Healthcare, Hillsboro, OR. The
IDEXX Catalyst SDMA Test for in-house measurement of SDMA concentration in serum from dogs and cats
 IDEXX Catalyst SDMA Test for in-house measurement of SDMA in serum from dogs and cats By Graham Bilbrough, Barbara Evert, Karen Hathaway, Gina Panagakos, Jane Robertson, and Maha Yerramilli Introduction
IDEXX Catalyst SDMA Test for in-house measurement of SDMA in serum from dogs and cats By Graham Bilbrough, Barbara Evert, Karen Hathaway, Gina Panagakos, Jane Robertson, and Maha Yerramilli Introduction
in vitro fertilization
 FERTILITY AND STERILITY VOL 69, NO. 6, JUNE 1998 Copyright (#1998 American Society for Reproductive Medicine Published by Elsevier Science Inc. Printed on acid-free paper in U.S.A. Elevated levels of basal
FERTILITY AND STERILITY VOL 69, NO. 6, JUNE 1998 Copyright (#1998 American Society for Reproductive Medicine Published by Elsevier Science Inc. Printed on acid-free paper in U.S.A. Elevated levels of basal
Hormone Balance - Female Report SAMPLE. result graph based on Luteal Phase. result graph based on Luteal Phase
 Patient Name: Patient DOB: Gender: Physician: Test Hormone Balance - Female Report SAMPLE Grote, Mary Jane Batch Number: B6437 2/16/1954 Accession Number: N52281 F Date Received: 2/3/2015 Any Lab Test
Patient Name: Patient DOB: Gender: Physician: Test Hormone Balance - Female Report SAMPLE Grote, Mary Jane Batch Number: B6437 2/16/1954 Accession Number: N52281 F Date Received: 2/3/2015 Any Lab Test
Lab Tests Made Simple
 Body Fluids Commonly Used for Testing Steroid Hormones Blood Serum (venipuncture) Plasma (venipuncture) Capillary Blood (finger/heel stick) Urine Saliva Limitations of Hormone Testing in Different Body
Body Fluids Commonly Used for Testing Steroid Hormones Blood Serum (venipuncture) Plasma (venipuncture) Capillary Blood (finger/heel stick) Urine Saliva Limitations of Hormone Testing in Different Body
Serology and International units
 Serology and International units L. Grangeot-Keros, National Reference Laboratory for Rubella, Virology Department, A. Béclère Hospital, Clamart, France Detection of rubella-specific IgG antibody Assays
Serology and International units L. Grangeot-Keros, National Reference Laboratory for Rubella, Virology Department, A. Béclère Hospital, Clamart, France Detection of rubella-specific IgG antibody Assays
College of American Pathologists (CAP) GH2 Survey Data: (updated 12/09) 2009 GH2-B (fresh pooled samples) * = NGSP certified at the time of the survey
 College of American Pathologists (CAP) GH2 Survey Data: (updated 12/09) The American Diabetes Association (ADA) recommends that laboratories use only HbA1c assay methods that have been NGSP certified and
College of American Pathologists (CAP) GH2 Survey Data: (updated 12/09) The American Diabetes Association (ADA) recommends that laboratories use only HbA1c assay methods that have been NGSP certified and
Evaluation of the UniCel TM DxI 800 Immunoassay Analyzer in Measuring Five Tumor Markers
 Original Article http://dx.doi.org/1.3349/ymj.212.53.3.557 pissn: 513-5796, eissn: 1976-2437 Yonsei Med J 53(3):557-564, 212 Evaluation of the UniCel TM DxI 8 Immunoassay Analyzer in Measuring Five Tumor
Original Article http://dx.doi.org/1.3349/ymj.212.53.3.557 pissn: 513-5796, eissn: 1976-2437 Yonsei Med J 53(3):557-564, 212 Evaluation of the UniCel TM DxI 8 Immunoassay Analyzer in Measuring Five Tumor
Traditional approaches to Reference Intervals
 Establishing reference intervals: Unique challenges for the pediatric population Amy L. Pyle, PhD, DABCC Traditional approaches to Reference Intervals Decision Limits Establish Verify Transfer 1 Decision
Establishing reference intervals: Unique challenges for the pediatric population Amy L. Pyle, PhD, DABCC Traditional approaches to Reference Intervals Decision Limits Establish Verify Transfer 1 Decision
1 PROTOCOL. Comparison Study Summary. Springs Memorial Hospital. Springs Memorial Hospital 800 W. Meeting St. Lancaster, SC (803)
 Comparison Study Summary Springs Memorial Hospital 800 W. Meeting St. Lancaster, SC 29720 (803) 286-1480 February 10, 2016 1 PROTOCOL This evaluation was conducted on February 10, 2016 at Springs Memorial
Comparison Study Summary Springs Memorial Hospital 800 W. Meeting St. Lancaster, SC 29720 (803) 286-1480 February 10, 2016 1 PROTOCOL This evaluation was conducted on February 10, 2016 at Springs Memorial
MARTIN FLEISHER, PH.D., FACB MEMORIAL SLOAN-KETTERING CANCER CENTER NEW YORK, N.Y.
 MARTIN FLEISHER, PH.D., FACB MEMORIAL SLOAN-KETTERING CANCER CENTER NEW YORK, N.Y. FLEISHEM@MSKCC.ORG The Ideal Tumor Marker Quantitative level of TM reflects tumor burden with high diagnostic sensitivity
MARTIN FLEISHER, PH.D., FACB MEMORIAL SLOAN-KETTERING CANCER CENTER NEW YORK, N.Y. FLEISHEM@MSKCC.ORG The Ideal Tumor Marker Quantitative level of TM reflects tumor burden with high diagnostic sensitivity
This supplementary material has been provided by the authors to give readers
 Supplementary Online Content LI D, Radulescu A, Shrestha RT, et al. Association of biotin ingestion with performance of hormone and nonhormone assays in healthy adults. JAMA. doi:1.11/jama.217.1375 efigure
Supplementary Online Content LI D, Radulescu A, Shrestha RT, et al. Association of biotin ingestion with performance of hormone and nonhormone assays in healthy adults. JAMA. doi:1.11/jama.217.1375 efigure
Superovulation of Beef Heifers with Follicle Stimulating Hormone or Human Menopausal Gonadotropin: Acute Effects on Hormone Secretion
 Superovulation of Beef Heifers with Follicle Stimulating Hormone or Human Menopausal Gonadotropin: Acute Effects on Hormone Secretion A.S. Leaflet R1362 Acacia A. Alcivar, graduate research assistant,
Superovulation of Beef Heifers with Follicle Stimulating Hormone or Human Menopausal Gonadotropin: Acute Effects on Hormone Secretion A.S. Leaflet R1362 Acacia A. Alcivar, graduate research assistant,
Photo (compulsory) Columbia University Medical Center, New York, NY 2. University of Southern California, Keck School of Medicine, Los Angeles, CA 3
 Progesterone bioavailability for preventing endometrial stimulation with a continuous-combined regimen of TX-001HR (oral estradiol and micronized progesterone capsules) Photo (compulsory) Rogerio A. Lobo,
Progesterone bioavailability for preventing endometrial stimulation with a continuous-combined regimen of TX-001HR (oral estradiol and micronized progesterone capsules) Photo (compulsory) Rogerio A. Lobo,
Guide to Fulfillment of Measurement Uncertainty
 DIAGNOSTIC ACCREDITATION PROGRAM College of Physicians and Surgeons of British Columbia 300 669 Howe Street Telephone: 604-733-7758 ext. 2635 Vancouver BC V6C 0B4 Toll Free: 1-800-461-3008 (in BC) www.cpsbc.ca
DIAGNOSTIC ACCREDITATION PROGRAM College of Physicians and Surgeons of British Columbia 300 669 Howe Street Telephone: 604-733-7758 ext. 2635 Vancouver BC V6C 0B4 Toll Free: 1-800-461-3008 (in BC) www.cpsbc.ca
Total Cost of Ownership (TCO): An evidence-based approach to compare laboratory equipment
 Total Cost of Ownership (TCO): An evidence-based approach to compare laboratory equipment P.C.G. Gontard 1, L.I. Stankevich 1, B.G. Gorodetsky 1 SUMMARY Clinical laboratories across the globe operate in
Total Cost of Ownership (TCO): An evidence-based approach to compare laboratory equipment P.C.G. Gontard 1, L.I. Stankevich 1, B.G. Gorodetsky 1 SUMMARY Clinical laboratories across the globe operate in
Measurement of total thyroxine concentration in serum from dogs and cats by use of various methods Objective Sample Population Procedure Results
 Measurement of total thyroxine concentration in serum from dogs and cats by use of various methods Robert J. Kemppainen, DVM, PhD, and Jeremy R. Birchfield, DVM Objective To compare results obtained from
Measurement of total thyroxine concentration in serum from dogs and cats by use of various methods Robert J. Kemppainen, DVM, PhD, and Jeremy R. Birchfield, DVM Objective To compare results obtained from
SCIEX Vitamin D 200M Assay for the Topaz System
 The First FDA-Cleared LC-MS/MS Assay for Vitamin D SCIEX Vitamin D 200M Assay for the Topaz System The first FDA-cleared LC-MS/MS assay for Vitamin D Vitamin D is an important building block for human
The First FDA-Cleared LC-MS/MS Assay for Vitamin D SCIEX Vitamin D 200M Assay for the Topaz System The first FDA-cleared LC-MS/MS assay for Vitamin D Vitamin D is an important building block for human
Fish follicle-stimulating hormone, FSH ELISA Kit
 Fish follicle-stimulating hormone, FSH ELISA Kit Catalog No: E0830f 96 Tests Operating instruction www.eiaab.com FOR RESEARCH USE ONLY; NOT FOR THERAPEUTIC OR DIAGNOSTIC APPLICATIONS! PLEASE READ THROUGH
Fish follicle-stimulating hormone, FSH ELISA Kit Catalog No: E0830f 96 Tests Operating instruction www.eiaab.com FOR RESEARCH USE ONLY; NOT FOR THERAPEUTIC OR DIAGNOSTIC APPLICATIONS! PLEASE READ THROUGH
METHOD VALIDATION: WHY, HOW AND WHEN?
 METHOD VALIDATION: WHY, HOW AND WHEN? Linear-Data Plotter a,b s y/x,r Regression Statistics mean SD,CV Distribution Statistics Comparison Plot Histogram Plot SD Calculator Bias SD Diff t Paired t-test
METHOD VALIDATION: WHY, HOW AND WHEN? Linear-Data Plotter a,b s y/x,r Regression Statistics mean SD,CV Distribution Statistics Comparison Plot Histogram Plot SD Calculator Bias SD Diff t Paired t-test
Reproductive. Estradiol Analyte Information
 Reproductive Estradiol Analyte Information - 1 - Estradiol Introduction Estradiol (E2 or 17β-estradiol) is the major estrogen in humans. Although it is often called the "female" hormone, it is also present
Reproductive Estradiol Analyte Information - 1 - Estradiol Introduction Estradiol (E2 or 17β-estradiol) is the major estrogen in humans. Although it is often called the "female" hormone, it is also present
Automated Quantitation of Hemoglobin-Based Blood Substitutes in Whole Blood Samples
 Hematopathology / HEMOGLOBIN-BASED BLOOD SUBSTITUTES Automated Quantitation of Hemoglobin-Based Blood Substitutes in Whole Blood Samples Jolanta Kunicka, PhD, 1 Michael Malin, PhD, 1 David Zelmanovic,
Hematopathology / HEMOGLOBIN-BASED BLOOD SUBSTITUTES Automated Quantitation of Hemoglobin-Based Blood Substitutes in Whole Blood Samples Jolanta Kunicka, PhD, 1 Michael Malin, PhD, 1 David Zelmanovic,
Evaluation Report: Eurolyser CRP test (ST0100 and ST0102) on. CUBE analyser (CA0100)
 Evaluation Report: Eurolyser CRP test (ST0100 and ST0102) on CUBE analyser (CA0100) Location Location: Eurolyser Diagnostica GmbH Operators: Simone Wieser; Franz Helminger; Michael Gruber Date: July-November
Evaluation Report: Eurolyser CRP test (ST0100 and ST0102) on CUBE analyser (CA0100) Location Location: Eurolyser Diagnostica GmbH Operators: Simone Wieser; Franz Helminger; Michael Gruber Date: July-November
The Adiponectin Turbidimetric Immunoassay Reagent Kit
 The Adiponectin Turbidimetric Immunoassay Reagent Kit Catalogue number: 51010 For the quantitative determination of Adiponectin in human serum and plasma This package insert must be read in its entirety
The Adiponectin Turbidimetric Immunoassay Reagent Kit Catalogue number: 51010 For the quantitative determination of Adiponectin in human serum and plasma This package insert must be read in its entirety
8.2 Principles of Quantitative Hematologic Determinations (1)
 3 8. FIELD CENTER HEMATOLOGY SERVICES 8.1 Clinical Significance Quantitation of the formed elements of the blood (erythrocytes -RBCs, leukocytes - WBCs, and platelets) is important in the ARIC study primarily
3 8. FIELD CENTER HEMATOLOGY SERVICES 8.1 Clinical Significance Quantitation of the formed elements of the blood (erythrocytes -RBCs, leukocytes - WBCs, and platelets) is important in the ARIC study primarily
Gentian Canine CRP Immunoassay Application Note for scil VitroVet*
 v02-jan2014 Gentian Canine CRP Immunoassay Application Note for scil VitroVet* Intended Use The canine CRP immunoassay on scil VitroVet is an in vitro diagnostic test for quantitative determination of
v02-jan2014 Gentian Canine CRP Immunoassay Application Note for scil VitroVet* Intended Use The canine CRP immunoassay on scil VitroVet is an in vitro diagnostic test for quantitative determination of
QMS TACROLIMUS APPLICATION BECKMAN COULTER AU480 /AU680 /AU5800
 QMS TACROLIMUS APPLICATION BECKMAN COULTER AU480 /AU680 /AU5800 Beckman Coulter Reagent REF A53727 The QMS Tacrolimus Immunoassay is intended for the quantitative determination of tacrolimus in human whole
QMS TACROLIMUS APPLICATION BECKMAN COULTER AU480 /AU680 /AU5800 Beckman Coulter Reagent REF A53727 The QMS Tacrolimus Immunoassay is intended for the quantitative determination of tacrolimus in human whole
Commentary by R. Little, Ph.D., NGSP Network Coordinator for the NGSP Steering Committee
 College of American Pathologists (CAP) GH5 Survey Data: (updated 8/17) The American Diabetes Association (ADA) recommends that The A1C test should be performed using a method that is certified by the NGSP.
College of American Pathologists (CAP) GH5 Survey Data: (updated 8/17) The American Diabetes Association (ADA) recommends that The A1C test should be performed using a method that is certified by the NGSP.
Total Thyroxine ELISA (T4)
 Design Verification Total Thyroxine ELISA (T4) Contents 1 Assay Principle... 2 2 Imprecision... 2 Within-run Imprecision... 2 Between run Imprecision... 2 3 Comparison of Methods, Accuracy... 2 4 Linearity...
Design Verification Total Thyroxine ELISA (T4) Contents 1 Assay Principle... 2 2 Imprecision... 2 Within-run Imprecision... 2 Between run Imprecision... 2 3 Comparison of Methods, Accuracy... 2 4 Linearity...
1. During the follicular phase of the ovarian cycle, the hypothalamus releases GnRH.
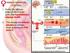 1. During the follicular phase of the ovarian cycle, the hypothalamus releases GnRH. 2. This causes the anterior pituitary to secrete small quantities of FSH and LH. 3. At this time, the follicles in the
1. During the follicular phase of the ovarian cycle, the hypothalamus releases GnRH. 2. This causes the anterior pituitary to secrete small quantities of FSH and LH. 3. At this time, the follicles in the
Evaluation of the Abbott CELL-DYN 4000 Hematology Analyzer
 Hematopathology / EVALUATION OF THE ABBOTT CELL-DYN 4 HEMATOLOGY ANALYZER Evaluation of the Abbott CELL-DYN 4 Hematology Analyzer Ernesto Grimaldi, MD, and Francesco Scopacasa, PhD Key Words: Abbott CD
Hematopathology / EVALUATION OF THE ABBOTT CELL-DYN 4 HEMATOLOGY ANALYZER Evaluation of the Abbott CELL-DYN 4 Hematology Analyzer Ernesto Grimaldi, MD, and Francesco Scopacasa, PhD Key Words: Abbott CD
Superovulation of Beef Heifers with Follicle Stimulating Hormone or Human Menopausal Gonadotropin: Acute Effects on Hormone Secretion
 Beef Research Report, 1996 Animal Science Research Reports 1997 Superovulation of Beef Heifers with Follicle Stimulating Hormone or Human Menopausal Gonadotropin: Acute Effects on Hormone Secretion Acacia
Beef Research Report, 1996 Animal Science Research Reports 1997 Superovulation of Beef Heifers with Follicle Stimulating Hormone or Human Menopausal Gonadotropin: Acute Effects on Hormone Secretion Acacia
Rat Mullerian Inhibiting Substance/Anti-Mullerian hormone, MIS/AMH ELISA kit
 Rat Mullerian Inhibiting Substance/Anti-Mullerian hormone, MIS/AMH ELISA kit Catalog No. E0228r (96 tests) Operating instruction www.eiaab.com FOR RESEARCH USE ONLY; NOT FOR THERAPEUTIC OR DIAGNOSTIC APPLICATIONS!
Rat Mullerian Inhibiting Substance/Anti-Mullerian hormone, MIS/AMH ELISA kit Catalog No. E0228r (96 tests) Operating instruction www.eiaab.com FOR RESEARCH USE ONLY; NOT FOR THERAPEUTIC OR DIAGNOSTIC APPLICATIONS!
Commentary by R. Little, Ph.D., NGSP Network Coordinator for the NGSP Steering Committee
 College of American Pathologists (CAP) GH5 Survey Data: (updated 12/17) The American Diabetes Association (ADA) recommends that The A1C test should be performed using a method that is certified by the
College of American Pathologists (CAP) GH5 Survey Data: (updated 12/17) The American Diabetes Association (ADA) recommends that The A1C test should be performed using a method that is certified by the
Commercial insulin immunoassays fail to detect commonly prescribed insulin analogues
 Commercial insulin immunoassays fail to detect commonly prescribed insulin analogues Ceri Parfitt Trainee Clinical Scientist Blood Sciences Department Royal Devon and Exeter NHS Foundation Trust November
Commercial insulin immunoassays fail to detect commonly prescribed insulin analogues Ceri Parfitt Trainee Clinical Scientist Blood Sciences Department Royal Devon and Exeter NHS Foundation Trust November
New York State Soluble Tumor Markers Proficiency Test
 ADREW M. CUOMO Governor October 5, 2016 HOWARD A. ZUCKER, M.D., J.D. Commissioner SALLY DRESLI, M.S., R.. Executive Deputy Commissioner ew York State Soluble Tumor Markers Proficiency Test 9-2016 1 Dear
ADREW M. CUOMO Governor October 5, 2016 HOWARD A. ZUCKER, M.D., J.D. Commissioner SALLY DRESLI, M.S., R.. Executive Deputy Commissioner ew York State Soluble Tumor Markers Proficiency Test 9-2016 1 Dear
Analytical Variability Among Methods for the Measurement of 25-Hydroxyvitamin D. Still Adding to the Noise
 Analytical Variability Among Methods for the Measurement of 25-Hydroxyvitamin D Still Adding to the Noise Earle W. Holmes, PhD, 1 Jean Garbincius, 2 and Kathleen M. McKenna, MBA, MT 2 From the 1 Departments
Analytical Variability Among Methods for the Measurement of 25-Hydroxyvitamin D Still Adding to the Noise Earle W. Holmes, PhD, 1 Jean Garbincius, 2 and Kathleen M. McKenna, MBA, MT 2 From the 1 Departments
All stocks and calibration levels were prepared in water: methanol (50:50) v/v to cover range of all steroid concentrations (refer Table 1).
 Application LCMS-8040 Simultaneous determination of 11 steroids and Vitamin D2/D3 in human serum using LC/MS/MS - Introduction Quantification of endogenous hormonal steroids and their precursors is essential
Application LCMS-8040 Simultaneous determination of 11 steroids and Vitamin D2/D3 in human serum using LC/MS/MS - Introduction Quantification of endogenous hormonal steroids and their precursors is essential
A validation study of after reconstitution stability of diabetes: level 1 and diabetes level 2 controls
 A validation study of after reconstitution stability of diabetes: level 1 and diabetes level 2 controls Shyamali Pal R B Diagnostic Private Limited, Lake Town, Kolkata, India INFO ABSTRACT Corresponding
A validation study of after reconstitution stability of diabetes: level 1 and diabetes level 2 controls Shyamali Pal R B Diagnostic Private Limited, Lake Town, Kolkata, India INFO ABSTRACT Corresponding
Elevated Urine Zinc Concentration Reduces the Detection of Methamphetamine, Cocaine, THC and Opiates in Urine by EMIT
 Journal of Analytical Toxicology 2013;37:665 669 doi:10.1093/jat/bkt056 Advance Access publication July 10, 2013 Article Elevated Urine Zinc Concentration Reduces the Detection of Methamphetamine, Cocaine,
Journal of Analytical Toxicology 2013;37:665 669 doi:10.1093/jat/bkt056 Advance Access publication July 10, 2013 Article Elevated Urine Zinc Concentration Reduces the Detection of Methamphetamine, Cocaine,
International Journal of Pharma and Bio Sciences ENDOCRINE MARKERS AND DECLINE IN REPRODUCTIVE POTENTIAL OF WOMEN ABSTRACT
 Research Article Biochemistry International Journal of Pharma and Bio Sciences ISSN 0975-6299 ENDOCRINE MARKERS AND DECLINE IN REPRODUCTIVE POTENTIAL OF WOMEN BUSHRA FIZA *, 1, 2, RATI MATHUR 2, MAHEEP
Research Article Biochemistry International Journal of Pharma and Bio Sciences ISSN 0975-6299 ENDOCRINE MARKERS AND DECLINE IN REPRODUCTIVE POTENTIAL OF WOMEN BUSHRA FIZA *, 1, 2, RATI MATHUR 2, MAHEEP
FreeStyle Lite A Blood Glucose Meter That Requires No Coding
 Journal of Diabetes Science and Technology Volume 2, Issue 4, July 2008 Diabetes Technology Society SYMPOSIUM Shridhara, Ph.D. Abstract Background: Abbott Diabetes Care introduced the FreeStyle Lite blood
Journal of Diabetes Science and Technology Volume 2, Issue 4, July 2008 Diabetes Technology Society SYMPOSIUM Shridhara, Ph.D. Abstract Background: Abbott Diabetes Care introduced the FreeStyle Lite blood
BNP Fragment EIA (Cat.No. BI-20852W) For the Determination of BNP Fragment in Human Samples
 BNP Fragment EIA (Cat.No. BI-20852W) For the Determination of BNP Fragment in Human Samples ASSAY CHARACTERISTICS Method Competitive Enzyme Immunoassay, HRP/TMB. Microtiter plates are coated with a polyclonal
BNP Fragment EIA (Cat.No. BI-20852W) For the Determination of BNP Fragment in Human Samples ASSAY CHARACTERISTICS Method Competitive Enzyme Immunoassay, HRP/TMB. Microtiter plates are coated with a polyclonal
POTASSIUM/K. 3.6 mmol/l
 POTASSIUM/K Potassium is measured by ion-selective electrode potentiometry. In the calculation of results for potassium, concentration is related to potential through the Nernst equation. The i-stat System
POTASSIUM/K Potassium is measured by ion-selective electrode potentiometry. In the calculation of results for potassium, concentration is related to potential through the Nernst equation. The i-stat System
For In Vitro Diagnostic Use
 SYNCHRON System(s) Chemistry Information Sheet Lactate REF A95550 For In Vitro Diagnostic Use ANNUAL REVIEW Refer to Review and Revision Coversheet at the front of each method. PRINCIPLE INTENDED USE reagent,
SYNCHRON System(s) Chemistry Information Sheet Lactate REF A95550 For In Vitro Diagnostic Use ANNUAL REVIEW Refer to Review and Revision Coversheet at the front of each method. PRINCIPLE INTENDED USE reagent,
Diagnostic reagent for quantitative in vitro determination of Urea / Blood Urea Nitrogen in serum, plasma and urine by colorimetry.
 2013/07/30 A93A01315AUS A11A01641 60 ml 15 ml Pentra C400 Diagnostic reagent for quantitative in vitro determination of Urea / Blood Urea Nitrogen in serum, plasma and urine by colorimetry. QUAL-QA-TEMP-0846
2013/07/30 A93A01315AUS A11A01641 60 ml 15 ml Pentra C400 Diagnostic reagent for quantitative in vitro determination of Urea / Blood Urea Nitrogen in serum, plasma and urine by colorimetry. QUAL-QA-TEMP-0846
CMV DNA Quantification Using an Automated Platform for Nucleic Acid Extraction and Real- time PCR Assay Set-up
 JCM Accepts, published online ahead of print on 11 May 2011 J. Clin. Microbiol. doi:10.1128/jcm.00721-11 Copyright 2011, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights
JCM Accepts, published online ahead of print on 11 May 2011 J. Clin. Microbiol. doi:10.1128/jcm.00721-11 Copyright 2011, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights
Week 17 and 21 Comparing two assays and Measurement of Uncertainty Explain tools used to compare the performance of two assays, including
 Week 17 and 21 Comparing two assays and Measurement of Uncertainty 2.4.1.4. Explain tools used to compare the performance of two assays, including 2.4.1.4.1. Linear regression 2.4.1.4.2. Bland-Altman plots
Week 17 and 21 Comparing two assays and Measurement of Uncertainty 2.4.1.4. Explain tools used to compare the performance of two assays, including 2.4.1.4.1. Linear regression 2.4.1.4.2. Bland-Altman plots
Reproductive. Estrone sulfate Analyte Information
 Reproductive Estrone sulfate Analyte Information - 1 - Estrone sulfate Introduction Estrone sulfate (E1-S) is a sulfate derivative of estrone, and is the most abundant form of circulating estrogens in
Reproductive Estrone sulfate Analyte Information - 1 - Estrone sulfate Introduction Estrone sulfate (E1-S) is a sulfate derivative of estrone, and is the most abundant form of circulating estrogens in
Performance Characteristics of a New Single-Sample Freezing Point Depression Osmometer
 AACC, July 2007 Performance Characteristics of a New Single-Sample Freezing Point Depression Osmometer E. Garry, M. Pesta, N. Zampa Advanced Instruments, Inc., Norwood, MA 02062 AbstracT The Advanced Model
AACC, July 2007 Performance Characteristics of a New Single-Sample Freezing Point Depression Osmometer E. Garry, M. Pesta, N. Zampa Advanced Instruments, Inc., Norwood, MA 02062 AbstracT The Advanced Model
Bias in clinical chemistry. Elvar Theodorsson EFLM and Linköping University
 Bias in clinical chemistry Elvar Theodorsson EFLM and Linköping University Bias a controversial subject Different perspectives Reseachers Users Regulatory bodies Standardisation organisations Metrologists
Bias in clinical chemistry Elvar Theodorsson EFLM and Linköping University Bias a controversial subject Different perspectives Reseachers Users Regulatory bodies Standardisation organisations Metrologists
Evaluation of Cheongmeak DCS TM Reagents for Chemistry Analyzers
 임상검사와정도관리 J Lab Med Qual Assur 2010 ; 32:197-204 ISSN 1225-097X Evaluation of Cheongmeak DCS TM Reagents for Chemistry Analyzers Kyeong Seob Shin, Taek Eun Jeong, and Bo Ra Son Department of Laboratory
임상검사와정도관리 J Lab Med Qual Assur 2010 ; 32:197-204 ISSN 1225-097X Evaluation of Cheongmeak DCS TM Reagents for Chemistry Analyzers Kyeong Seob Shin, Taek Eun Jeong, and Bo Ra Son Department of Laboratory
For in vitro diagnostic use only. For Rx use only. Reviewed by Date Reviewed by Date
 AU US Instructions For Use 2018 Beckman Coulter, Inc. All rights reserved. TRIG TRIGLYCERIDE OSR60118 4 x 20 ml R1, 4 x 5 ml R2 OSR61118 4 x 50 ml R1, 4 x 12.5 ml R2 OSR66118 4 x 173 ml R1, 4 x 48 ml R2
AU US Instructions For Use 2018 Beckman Coulter, Inc. All rights reserved. TRIG TRIGLYCERIDE OSR60118 4 x 20 ml R1, 4 x 5 ml R2 OSR61118 4 x 50 ml R1, 4 x 12.5 ml R2 OSR66118 4 x 173 ml R1, 4 x 48 ml R2
Bio-Rad Laboratories. Cardiac Assessment Controls. Value Assigned for Clinical Laboratory and Point of Care Test Systems
 Bio-Rad Laboratories Cardiac Assessment Controls Cardiac Assessment Controls Value Assigned for Clinical Laboratory and Point of Care Test Systems Solving the Challenges of Troponin I Testing for the Central
Bio-Rad Laboratories Cardiac Assessment Controls Cardiac Assessment Controls Value Assigned for Clinical Laboratory and Point of Care Test Systems Solving the Challenges of Troponin I Testing for the Central
Hormonal Control of Human Reproduction
 Hormonal Control of Human Reproduction Bởi: OpenStaxCollege The human male and female reproductive cycles are controlled by the interaction of hormones from the hypothalamus and anterior pituitary with
Hormonal Control of Human Reproduction Bởi: OpenStaxCollege The human male and female reproductive cycles are controlled by the interaction of hormones from the hypothalamus and anterior pituitary with
Verification and validation of diagnostic laboratory tests in clinical virology
 Journal of Clinical Virology 40 (2007) 93 98 Review Verification and validation of diagnostic laboratory tests in clinical virology Holger F. Rabenau a,, Harald H. Kessler b, Marhild Kortenbusch a, Andreas
Journal of Clinical Virology 40 (2007) 93 98 Review Verification and validation of diagnostic laboratory tests in clinical virology Holger F. Rabenau a,, Harald H. Kessler b, Marhild Kortenbusch a, Andreas
ASSESSMENT OF A POINT-OF-CARE DEVICE FOR MEASURING CREATININE IN A COMMUNITY SCREENING PROGRAM FOR CHRONIC KIDNEY DISEASE
 ASSESSMENT OF A POINT-OF-CARE DEVICE FOR MEASURING CREATININE IN A COMMUNITY SCREENING PROGRAM FOR CHRONIC KIDNEY DISEASE Brooke Ann Spaeth, Anne K Shephard, Mark DS Shephard, Timothy H Mathew ABSTRACT
ASSESSMENT OF A POINT-OF-CARE DEVICE FOR MEASURING CREATININE IN A COMMUNITY SCREENING PROGRAM FOR CHRONIC KIDNEY DISEASE Brooke Ann Spaeth, Anne K Shephard, Mark DS Shephard, Timothy H Mathew ABSTRACT
The effect of serum matrix and gender on cortisol measurement by commonly used immunoassays
 Original Article Annals of Clinical Biochemistry 214, Vol. 51(3) 379 385! The Author(s) 213 Reprints and permissions: sagepub.co.uk/journalspermissions.nav DOI: 1.1177/4563213514567 acb.sagepub.com The
Original Article Annals of Clinical Biochemistry 214, Vol. 51(3) 379 385! The Author(s) 213 Reprints and permissions: sagepub.co.uk/journalspermissions.nav DOI: 1.1177/4563213514567 acb.sagepub.com The
HCG (human chorionic gonadotropin); Novarel Pregnyl (chorionic gonadotropin); Ovidrel (choriogonadotropin alfa)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.08.09 Subject: HCG Page: 1 of 5 Last Review Date: June 19, 2015 HCG Powder, Novarel, Pregnyl, Ovidrel
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.08.09 Subject: HCG Page: 1 of 5 Last Review Date: June 19, 2015 HCG Powder, Novarel, Pregnyl, Ovidrel
Comparison of Three Whole Blood Creatinine Methods for Estimation of Glomerular Filtration Rate Before Radiographic Contrast Administration
 Clinical Chemistry / Whole Blood Creatinine for egfr Comparison of Three Whole Blood Creatinine Methods for Estimation of Glomerular Filtration Rate Before Radiographic Contrast Administration Nichole
Clinical Chemistry / Whole Blood Creatinine for egfr Comparison of Three Whole Blood Creatinine Methods for Estimation of Glomerular Filtration Rate Before Radiographic Contrast Administration Nichole
Introduction. Etienne Cavalier*, Olivier Rousselle, Nunzio Ferrante, Agnes Carlisi, Caroline Le Goff and Jean-Claude Souberbielle.
 DOI 1.1515/cclm-13-138 Clin Chem Lab Med 13; aop Etienne Cavalier*, Olivier Rousselle, Nunzio Ferrante, Agnes Carlisi, Caroline Le Goff and Jean-Claude Souberbielle Technical and clinical evaluation of
DOI 1.1515/cclm-13-138 Clin Chem Lab Med 13; aop Etienne Cavalier*, Olivier Rousselle, Nunzio Ferrante, Agnes Carlisi, Caroline Le Goff and Jean-Claude Souberbielle Technical and clinical evaluation of
MICROWELL ELISA LUTEINIZING HORMONE (LH) ENZYMEIMMUNOASSAY TEST KIT LH ELISA. Cat # 4225Z
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Persistent Low Concentration of Human Chorionic Gonadotropin in a Nonpregnant Woman
 Clinical Chemistry 54:1 209 214 (2008) Clinical Case Study Persistent Low Concentration of Human Chorionic Gonadotropin in a Nonpregnant Woman Christopher R. McCudden, Monte S. Willis, and David G. Grenache
Clinical Chemistry 54:1 209 214 (2008) Clinical Case Study Persistent Low Concentration of Human Chorionic Gonadotropin in a Nonpregnant Woman Christopher R. McCudden, Monte S. Willis, and David G. Grenache
Establishing Reference Intervals for Sex Hormones on the Analytical Platforms Advia Centaur and Immulite 2000XP
 Brief Communication Clinical Chemistry Ann Lab Med 2016;36:5559 http://dx.doi.org/10.3343/alm.2016.36.1.55 ISSN 22343806 eissn 22343814 Establishing Reference Intervals for Sex Hormones on the Analytical
Brief Communication Clinical Chemistry Ann Lab Med 2016;36:5559 http://dx.doi.org/10.3343/alm.2016.36.1.55 ISSN 22343806 eissn 22343814 Establishing Reference Intervals for Sex Hormones on the Analytical
Cortisol (Sheep) ELISA Kit
 Cortisol (Sheep) ELISA Kit Catalog Number KA0919 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of the
Cortisol (Sheep) ELISA Kit Catalog Number KA0919 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of the
Annual Review Contents. Page 1. Introduction, scheme-wide trends, future developments Insulin C-Peptide 8-10
 UK NEQAS Guildford Peptide Hormones Annual Review 212 Contents Page 1. Introduction, scheme-wide trends, future developments 1-3 2. Insulin 3-7 3. C-Peptide 8-1 4. Insulin interpretative exercise 1-12
UK NEQAS Guildford Peptide Hormones Annual Review 212 Contents Page 1. Introduction, scheme-wide trends, future developments 1-3 2. Insulin 3-7 3. C-Peptide 8-1 4. Insulin interpretative exercise 1-12
ab Estradiol E2 Human ELISA Kit
 ab108640 Estradiol E2 Human ELISA Kit Instructions for Use A competitive immunoenzymatic assay for the quantitative measurement of Estradiol E2 in Human Serum. This product is for research use only and
ab108640 Estradiol E2 Human ELISA Kit Instructions for Use A competitive immunoenzymatic assay for the quantitative measurement of Estradiol E2 in Human Serum. This product is for research use only and
