ClinicalTrials.gov Protocol Registration and Results System (PRS) Receipt Release Date: 01/20/2014. ClinicalTrials.gov ID: NCT
|
|
|
- Tamsin Cummings
- 5 years ago
- Views:
Transcription
1 ClinicalTrials.gov Protocol Registration and Results System (PRS) Receipt Release Date: 01/20/2014 ClinicalTrials.gov ID: NCT Study Identification Unique Protocol ID: Brief Title: Enriched in Ovulation Induction Official Title: A Phase II, Multicentre, Randomised, Assessor-blinded, Active-comparator, Parallel-group Dose Finding Trial to Evaluate enriched Versus Follitropin Alfa (GONAL-f ) in Oligo-anovulatory Infertile Women Undergoing Ovulation Induction (OI) Secondary IDs: Study Status Record Verification: January 2014 Overall Status: Terminated Study Start: December 2007 Primary Completion: March 2009 [Actual] Study Completion: March 2009 [Actual] Sponsor/Collaborators Sponsor: Merck KGaA Responsible Party: Sponsor Collaborators: Oversight FDA Regulated?: No - Page 1 of 24 -
2 IND/IDE Protocol?: No Review Board: Approval Status: Approved Approval Number: 8 Oct 2007 Board Name: The Epworth Healthcare Human Researchand Ethics Committee (HREC) Board Affiliation: The Epworth Healthcare Human Researchand Ethics Committee (HREC) Phone: Data Monitoring?: Plan to Share Data?: Oversight Authorities: Argentina: Human Research Bioethics Committee Australia: Human Research Ethics Committee Belgium: Institutional Review Board Chile: Instituto de Salud Pública de Chile Denmark: Ethics Committee France: Institutional Ethical Committee Germany: Ethics Commission Israel: Ethics Commission Italy: Ethics Committee Netherlands: The Central Committee on Research Involving Human Subjects (CCMO) Spain: Comité Ético de Investigación Clínica Sweden: Regional Ethical Review Board Turkey: Ethics Committee United Kingdom: Research Ethics Committee Study Brief Summary: This is a Phase 2, interventional, multi-center, randomized, assessor-blind, active-comparator, dose-finding study to evaluate a new investigational long-acting follicle stimulating hormone (FSH) in oligo-anovulatory women undergoing ovulation induction (OI). This study will compare 4 doses of the investigational drug versus a currently marketed drug follitropin alfa (Gonal-f ) prefilled pen with regards to ovulation rate. Detailed : The study was terminated after Merck Serono had taken the decision not to pursue the development of enriched in ovulation induction (OI). This decision was not related to any safety or efficacy concerns over the use of Enriched in OI. Conditions Conditions: Ovulation Induction Keywords: Infertility Oligo-anovulation GONAL-f - Page 2 of 24 -
3 Enriched hyperglycosylated recombinant human follicle stimulating hormone (r-hfsh) Study Design Study Type: Interventional Primary Purpose: Treatment Study Phase: Phase 2 Intervention Model: Parallel Assignment Number of Arms: 5 Masking: Single Blind (Outcomes Assessor) Allocation: Randomized Endpoint Classification: Safety/Efficacy Study Enrollment: 71 [Actual] Arms and Interventions Arms Experimental: Assigned Interventions Drug: Enriched 10 microgram (mcg) Single injection of Enriched (hyperglycosylated recombinant human follicle stimulating hormone [r-hfsh]), 10 mcg will be administered subcutaneously on Stimulation Day 1 (S1). Duration of treatment cycle will be up to adequate Other Names: Hyperglycosylated r-hfsh Drug: Follitropin alfa 75 international unit (IU) Follitropin alfa (Gonal-f ) 75 IU will be administered subcutaneously once daily from S1 up to Stimulation Day 14 (S14) based upon ovarian response, until recombinant human chorionic gonadotropin (r-hcg) administration day. Subjects who will be receiving Enriched 10, 20, 30 and 40 mcg will also receive daily dose of follitropin alfa 75 IU subcutaneously from Stimulation Day 7 (S7) up to S14. Duration of treatment cycle will be up to adequate follicular response received or maximum of 14 days. Other Names: Gonal-f Follicle stimulating hormone (FSH) - Page 3 of 24 -
4 Arms Experimental: Experimental: Assigned Interventions Drug: Recombinant human chorionic gonadotropin (r-hcg) Recombinant human chorionic gonadotropin (r-hcg) will be administered as a single dose of 250 mcg subcutaneously, when follicular response is adequate (that is, less than or equal to [=<] 3 follicles with a mean diameter of greater than or equal to [>=] 14 millimeter [mm], and one or two of these follicles with a diameter of >= 17 mm). Drug: Single injection of Enriched (hyperglycosylated r-hfsh), 20 mcg will be administered subcutaneously on S1. Duration of treatment cycle will be up to adequate follicular response received or maximum of 14 days. Other Names: Hyperglycosylated r-hfsh Drug: Follitropin alfa 75 international unit (IU) Follitropin alfa (Gonal-f ) 75 IU will be administered subcutaneously once daily from S1 up to Stimulation Day 14 (S14) based upon ovarian response, until recombinant human chorionic gonadotropin (r-hcg) administration day. Subjects who will be receiving Enriched 10, 20, 30 and 40 mcg will also receive daily dose of follitropin alfa 75 IU subcutaneously from Stimulation Day 7 (S7) up to S14. Duration of treatment cycle will be up to adequate follicular response received or maximum of 14 days. Other Names: Gonal-f Follicle stimulating hormone (FSH) Drug: Recombinant human chorionic gonadotropin (r-hcg) Recombinant human chorionic gonadotropin (r-hcg) will be administered as a single dose of 250 mcg subcutaneously, when follicular response is adequate (that is, less than or equal to [=<] 3 follicles with a mean diameter of greater than or equal to [>=] 14 millimeter [mm], and one or two of these follicles with a diameter of >= 17 mm). Drug: Single injection of Enriched (hyperglycosylated r-hfsh), 30 mcg will be administered subcutaneously on S1. Duration of treatment cycle will be up to adequate follicular response received or maximum of 14 days. Other Names: Hyperglycosylated r-hfsh Drug: Follitropin alfa 75 international unit (IU) - Page 4 of 24 -
5 Arms Experimental: Assigned Interventions Follitropin alfa (Gonal-f ) 75 IU will be administered subcutaneously once daily from S1 up to Stimulation Day 14 (S14) based upon ovarian response, until recombinant human chorionic gonadotropin (r-hcg) administration day. Subjects who will be receiving Enriched 10, 20, 30 and 40 mcg will also receive daily dose of follitropin alfa 75 IU subcutaneously from Stimulation Day 7 (S7) up to S14. Duration of treatment cycle will be up to adequate follicular response received or maximum of 14 days. Other Names: Gonal-f Follicle stimulating hormone (FSH) Drug: Recombinant human chorionic gonadotropin (r-hcg) Recombinant human chorionic gonadotropin (r-hcg) will be administered as a single dose of 250 mcg subcutaneously, when follicular response is adequate (that is, less than or equal to [=<] 3 follicles with a mean diameter of greater than or equal to [>=] 14 millimeter [mm], and one or two of these follicles with a diameter of >= 17 mm). Drug: Single injection of Enriched (hyperglycosylated r-hfsh), 40 mcg will be administered subcutaneously on S1. Duration of treatment cycle will be up to adequate follicular response received or maximum of 14 days. Other Names: Hyperglycosylated r-hfsh Drug: Follitropin alfa 75 international unit (IU) Follitropin alfa (Gonal-f ) 75 IU will be administered subcutaneously once daily from S1 up to Stimulation Day 14 (S14) based upon ovarian response, until recombinant human chorionic gonadotropin (r-hcg) administration day. Subjects who will be receiving Enriched 10, 20, 30 and 40 mcg will also receive daily dose of follitropin alfa 75 IU subcutaneously from Stimulation Day 7 (S7) up to S14. Duration of treatment cycle will be up to adequate follicular response received or maximum of 14 days. Other Names: Gonal-f Follicle stimulating hormone (FSH) Drug: Recombinant human chorionic gonadotropin (r-hcg) Recombinant human chorionic gonadotropin (r-hcg) will be administered as a single dose of 250 mcg subcutaneously, when follicular response is adequate (that is, less than or equal to [=<] 3 follicles with a mean - Page 5 of 24 -
6 Arms Active Comparator: Follitropin alfa 75 IU Assigned Interventions diameter of greater than or equal to [>=] 14 millimeter [mm], and one or two of these follicles with a diameter of >= 17 mm). Drug: Follitropin alfa 75 international unit (IU) Follitropin alfa (Gonal-f ) 75 IU will be administered subcutaneously once daily from S1 up to Stimulation Day 14 (S14) based upon ovarian response, until recombinant human chorionic gonadotropin (r-hcg) administration day. Subjects who will be receiving Enriched 10, 20, 30 and 40 mcg will also receive daily dose of follitropin alfa 75 IU subcutaneously from Stimulation Day 7 (S7) up to S14. Duration of treatment cycle will be up to adequate follicular response received or maximum of 14 days. Other Names: Gonal-f Follicle stimulating hormone (FSH) Drug: Recombinant human chorionic gonadotropin (r-hcg) Recombinant human chorionic gonadotropin (r-hcg) will be administered as a single dose of 250 mcg subcutaneously, when follicular response is adequate (that is, less than or equal to [=<] 3 follicles with a mean diameter of greater than or equal to [>=] 14 millimeter [mm], and one or two of these follicles with a diameter of >= 17 mm). Outcome Measures [See Results Section.] Eligibility Minimum Age: 18 Years Maximum Age: 36 Years Gender: Female Accepts Healthy Volunteers?: No Criteria: Inclusion Criteria: Oligo-anovulation defined by a menstrual period of 35 days to 6 months Spontaneous menses or a positive response to progestin within the prior 6 months or response to clomiphene citrate evidenced by ovulation within the prior 6 months Age between 18 and 36 years, inclusive, at time of informed consent signature Body mass index (BMI) 18 to 30 kilogram per square meter (kg/m^2), inclusive - Page 6 of 24 -
7 No clinically significant abnormalities in serum thyroid stimulating hormone (TSH), dehydroepiandrosterone (DHEA-S), prolactin, and FSH levels in the early follicular phase. Subjects low TSH level who receive replacement therapy could be enrolled at the discretion of the investigator if local laboratory results (thyroxine [T4]) demonstrated satisfactory thyroid function. Subjects receiving stable dose of dopamine agonists could be enrolled at the discretion of the investigator if local laboratory results demonstrated adequate control of prolactin levels No clinically significant abnormalities in fasting glucose and fasting insulin levels Normal uterine cavity and presence of at least one ovary with ipsilateral patent fallopian tube, as determined by means of hysterosalpingography, laparoscopy, hysteroscopy or combination of these procedures within the prior 3 years Papanicolaou (PAP) smear test without clinically significant abnormalities within 6 months prior to the first screening procedure. If PAP smear is not done, it must be performed as part of screening procedures Negative pregnancy test prior to randomization Male partner with semen analysis demonstrating adequacy for insemination via intercourse and/or intrauterine insemination (IUI) within 6 months prior to the first screening procedure. If semen analysis is not done, it must be performed as part of screening procedures Willing and able to comply with all protocol procedures, including pregnancy and neonatal follow-up Voluntary provision of written informed consent, prior to any trial-related procedure that is not part of normal medical care, with the understanding that the subject could withdraw consent at any time without prejudice to her future medical care, including willingness to provide follow-up information on babies born as part of this trial Exclusion Criteria: History of >=2 consecutive gonadotrophin stimulation cycles that did not lead to ovulation History of clomiphene citrate stimulation cycles of which none lead to ovulation Prior excessive response to gonadotrophin stimulation, defined as the development of at least 4 mature follicles (greater than [>]17 mm) or cancellation of the OI cycle due to excessive follicular response after treatment with FSH at a dose of less than 75 IU/day Previous severe ovarian hyperstimulation syndrome (OHSS) Administration of any gonadotrophin, clomiphene citrate, gonadotrophin releasing hormone (GnRH) analogue, tamoxifen or aromatase inhibitors within the prior 30 days Laparoscopic ovarian drilling and/or ovarian cauterization within the prior 6 months Any contraindication to pregnancy and/or to carrying pregnancy to term A clinical pregnancy that ended in a miscarriage within the prior 3 months History of >=3 consecutive miscarriages, due to any cause Abnormal gynecological bleeding of undetermined origin Clinically significant abnormal findings of the uterine cavity evident on a transvaginal pelvic ultrasound performed during screening Presence of endometriosis, Grade III - IV or requiring treatment Ovarian cyst with a mean diameter of >25 mm on the day of randomization History or suspicion of ovarian, uterine or mammary cancer Adrenal congenital hyperplasia, partial or complete enzymatic block Use of metformin or other insulin sensitizing agents related to infertility within the prior 2 months Known allergy or hypersensitivity to human gonadotrophin preparations or to compounds that are structurally similar to any of the other medications administered during the trial Any contra-indication to gonadotrophin therapy Known or suspected infection with human immunodeficiency virus (HIV), Hepatitis B or C in the trial subject or her male partner - Page 7 of 24 -
8 Any active substance abuse or history of drug, medication or alcohol abuse within 5 years Smoker consuming more than 5 cigarettes per day Serum testosterone (central laboratory) that is suggestive of ovarian tumor Previously randomized in this trial or participation in another investigational drug clinical trial within the prior 3 months Any medical condition, which in the judgment of the investigator may interfere with the absorption, distribution, metabolism or excretion of r-hfsh Clinically significant concurrent disease (including diabetes mellitus and autoimmune diseases) that would compromise subject safety or interfere with the study assessments or clinically significant abnormal laboratory finding Contacts/Locations Study Officials: Antonio Pellicer, Professor Dr Study Principal Investigator IVI Valencia Locations: Switzerland Merck Serono S.A. Geneva, Switzerland, 1202 References Citations: Links: Study Data/Documents: - Page 8 of 24 -
9 Study Results Participant Flow Reporting Groups Single injection of Enriched (hyperglycosylated recombinant human follicle stimulating hormone [r-hfsh]), 10 microgram (mcg) administered subcutaneously on Stimulation day 1 (S1) followed by a daily dose of follitropin alfa 75 international unit (IU) subcutaneously starting from Stimulation Day 7 (S7) up to Stimulation Day 14 (S14) based upon ovarian response, until recombinant human chorionic gonadotropin (rhcg) administration day. When follicular response was adequate (that is, less than or equal to [=<] 3 follicles with a mean diameter of greater than or equal to [>=] 14 millimeter [mm], and one or two of these follicles with a diameter of >= 17 mm), ovulation was triggered by single 250 mcg subcutaneous r-hcg injection. Duration of treatment cycle was up to adequate Single injection of Enriched (hyperglycosylated r-hfsh), 20 mcg administered subcutaneously on Single injection of Enriched (hyperglycosylated r-hfsh), 30 mcg administered subcutaneously on Single injection of Enriched (hyperglycosylated r-hfsh), 40 mcg administered subcutaneously on Follitropin alfa (Gonal-f ) 75 IU administered subcutaneously once daily from S1 up to S14 based upon ovarian response, until r-hcg administration day. When follicular response was adequate (that is, =< 3 follicles with a mean diameter of >=14 mm, and one or two of these follicles with a diameter of >=17 mm), ovulation was triggered by single 250 mcg subcutaneous r-hcg injection. Duration of treatment cycle was up to adequate - Page 9 of 24 -
10 Overall Study Started Completed Not Completed Protocol deviation Lack of ovarian response Ovarian hyperstimulation syndrome risk Risk of multiple pregnancy Unspecified Follitropin Alfa 75 IU Baseline Characteristics Reporting Groups Single injection of Enriched (hyperglycosylated recombinant human follicle stimulating hormone [r-hfsh]), 10 microgram (mcg) administered subcutaneously on Stimulation day 1 (S1) followed by a daily dose of follitropin alfa 75 international unit (IU) subcutaneously starting from Stimulation Day 7 (S7) up to Stimulation Day 14 (S14) based upon ovarian response, until recombinant human chorionic gonadotropin (rhcg) administration day. When follicular response was adequate (that is, less than or equal to [=<] 3 follicles with a mean diameter of greater than or equal to [>=] 14 millimeter [mm], and one or two of these follicles with a diameter of >= 17 mm), ovulation was triggered by single 250 mcg subcutaneous r-hcg injection. Duration of treatment cycle was up to adequate Single injection of Enriched (hyperglycosylated r-hfsh), 20 mcg administered subcutaneously on Single injection of Enriched (hyperglycosylated r-hfsh), 30 mcg administered subcutaneously on - Page 10 of 24 -
11 Single injection of Enriched (hyperglycosylated r-hfsh), 40 mcg administered subcutaneously on Follitropin alfa (Gonal-f ) 75 IU administered subcutaneously once daily from S1 up to S14 based upon ovarian response, until r-hcg administration day. When follicular response was adequate (that is, =< 3 follicles with a mean diameter of >=14 mm, and one or two of these follicles with a diameter of >=17 mm), ovulation was triggered by single 250 mcg subcutaneous r-hcg injection. Duration of treatment cycle was up to adequate Baseline Measures Follitropin Alfa 75 IU Total Number of Participants Age, Continuous [units: years] Median (Full Range) 29.5 (23 to 36) 31.5 (23 to 36) 28.0 (26 to 35) 30.0 (23 to 36) 30.0 (23 to 34) 30 (23 to 36) Gender, Male/Female [units: participants] Female Male Outcome Measures 1. Primary Outcome Measure: Measure Title Measure Time Frame Safety Issue? Percentage of Participants With Ovulation Ovulation was defined as a mid-luteal phase progesterone (P4) level >= 30 nanomole per liter (nmol/l) (10 nanogram per milliliter [ng/ml]). In the absence of a positive progesterone response, clinical pregnancy was also considered as evidence of ovulation. Mid-luteal phase progesterone assessed 5-10 days or clinical pregnancy days after recombinant human chorionic gonadotropin (r-hcg) administration day (end of stimulation cycle [approximately 14 days]) No - Page 11 of 24 -
12 Analysis Population Per Protocol (PP) population included all the randomized participants who were without a medically relevant protocol deviation. 'N' (number of participants analyzed) signifies participants who were evaluable for this measure. Reporting Groups Single injection of Enriched (hyperglycosylated recombinant human follicle stimulating hormone [r-hfsh]), 10 microgram (mcg) administered subcutaneously on Stimulation day 1 (S1) followed by a daily dose of follitropin alfa 75 international unit (IU) subcutaneously starting from Stimulation Day 7 (S7) up to Stimulation Day 14 (S14) based upon ovarian response, until recombinant human chorionic gonadotropin (rhcg) administration day. When follicular response was adequate (that is, less than or equal to [=<] 3 follicles with a mean diameter of greater than or equal to [>=] 14 millimeter [mm], and one or two of these follicles with a diameter of >= 17 mm), ovulation was triggered by single 250 mcg subcutaneous r-hcg injection. Duration of treatment cycle was up to adequate Single injection of Enriched (hyperglycosylated r-hfsh), 20 mcg administered subcutaneously on Single injection of Enriched (hyperglycosylated r-hfsh), 30 mcg administered subcutaneously on Single injection of Enriched (hyperglycosylated r-hfsh), 40 mcg administered subcutaneously on Follitropin alfa (Gonal-f ) 75 IU administered subcutaneously once daily from S1 up to S14 based upon ovarian response, until r-hcg administration day. When follicular response was adequate (that is, =< 3 follicles with a mean diameter of >=14 mm, and one or two of these follicles with a diameter of >=17 mm), ovulation was triggered by single 250 mcg subcutaneous r-hcg injection. Duration of treatment cycle was up to adequate - Page 12 of 24 -
13 Measured Values Follitropin Alfa 75 IU Number of Participants Analyzed Percentage of Participants With Ovulation [units: Percentage of participants] Secondary Outcome Measure: Measure Title Measure Time Frame Safety Issue? Percentage of Participants With Clinical Pregnancy Clinical pregnancy was defined as the presence of one or more fetal sacs with fetal heart activity on the Day post r-hcg ultrasound examination. Day post r-hcg administration day (end of stimulation cycle [approximately 14 days]) No Analysis Population PP population included all the randomized participants who were without a medically relevant protocol deviation. 'N' (number of participants analyzed) signifies participants who were evaluable for this measure. Reporting Groups Single injection of Enriched (hyperglycosylated recombinant human follicle stimulating hormone [r-hfsh]), 10 microgram (mcg) administered subcutaneously on Stimulation day 1 (S1) followed by a daily dose of follitropin alfa 75 international unit (IU) subcutaneously starting from Stimulation Day 7 (S7) up to Stimulation Day 14 (S14) based upon ovarian response, until recombinant human chorionic gonadotropin (rhcg) administration day. When follicular response was adequate (that is, less than or equal to [=<] 3 follicles with a mean diameter of greater than or equal to [>=] 14 millimeter [mm], and one or two of these follicles with a diameter of >= 17 mm), ovulation was triggered by single 250 mcg subcutaneous r-hcg injection. Duration of treatment cycle was up to adequate Single injection of Enriched (hyperglycosylated r-hfsh), 20 mcg administered subcutaneously on - Page 13 of 24 -
14 Single injection of Enriched (hyperglycosylated r-hfsh), 30 mcg administered subcutaneously on Single injection of Enriched (hyperglycosylated r-hfsh), 40 mcg administered subcutaneously on Follitropin alfa (Gonal-f ) 75 IU administered subcutaneously once daily from S1 up to S14 based upon ovarian response, until r-hcg administration day. When follicular response was adequate (that is, =< 3 follicles with a mean diameter of >=14 mm, and one or two of these follicles with a diameter of >=17 mm), ovulation was triggered by single 250 mcg subcutaneous r-hcg injection. Duration of treatment cycle was up to adequate Measured Values Number of Participants Analyzed Percentage of Participants With Clinical Pregnancy [units: Percentage of participants] Follitropin Alfa 75 IU Secondary Outcome Measure: Measure Title Measure Time Frame Safety Issue? Duration of Ovarian Stimulation Ovarian stimulation included from first dose of study drug on S1 until day on which r-hcg was administered (r-hcg day). Stimulation Day 1 (S1) up to r-hcg administration day (end of stimulation cycle [approximately 14 days]) No Analysis Population PP population included all the randomized participants who were without a medically relevant protocol deviation. 'N' (number of participants analyzed) signifies participants who were evaluable for this measure. - Page 14 of 24 -
15 Reporting Groups Single injection of Enriched (hyperglycosylated recombinant human follicle stimulating hormone [r-hfsh]), 10 microgram (mcg) administered subcutaneously on Stimulation day 1 (S1) followed by a daily dose of follitropin alfa 75 international unit (IU) subcutaneously starting from Stimulation Day 7 (S7) up to Stimulation Day 14 (S14) based upon ovarian response, until recombinant human chorionic gonadotropin (rhcg) administration day. When follicular response was adequate (that is, less than or equal to [=<] 3 follicles with a mean diameter of greater than or equal to [>=] 14 millimeter [mm], and one or two of these follicles with a diameter of >= 17 mm), ovulation was triggered by single 250 mcg subcutaneous r-hcg injection. Duration of treatment cycle was up to adequate Single injection of Enriched (hyperglycosylated r-hfsh), 20 mcg administered subcutaneously on Single injection of Enriched (hyperglycosylated r-hfsh), 30 mcg administered subcutaneously on Single injection of Enriched (hyperglycosylated r-hfsh), 40 mcg administered subcutaneously on Follitropin alfa (Gonal-f ) 75 IU administered subcutaneously once daily from S1 up to S14 based upon ovarian response, until r-hcg administration day. When follicular response was adequate (that is, =< 3 follicles with a mean diameter of >=14 mm, and one or two of these follicles with a diameter of >=17 mm), ovulation was triggered by single 250 mcg subcutaneous r-hcg injection. Duration of treatment cycle was up to adequate Measured Values Number of Participants Analyzed Follitropin Alfa 75 IU Duration of Ovarian Stimulation 10.0 (5.3) 11.8 (3.4) 14.0 (1.4) 10.8 (3.7) 10.6 (3.2) - Page 15 of 24 -
16 Follitropin Alfa 75 IU [units: Days] Mean (Standard Deviation) 4. Secondary Outcome Measure: Measure Title Measure Time Frame Safety Issue? Duration of Supplemental Follitropin Alfa Treatment Stimulation Day 7 (S7) up to r-hcg administration day (end of stimulation cycle [approximately 14 days]) No Analysis Population PP population included all the randomized participants who were without a medically relevant protocol deviation. 'N' (number of participants analyzed) signifies participants who were evaluable for this measure. Reporting Groups Single injection of Enriched (hyperglycosylated recombinant human follicle stimulating hormone [r-hfsh]), 10 microgram (mcg) administered subcutaneously on Stimulation day 1 (S1) followed by a daily dose of follitropin alfa 75 international unit (IU) subcutaneously starting from Stimulation Day 7 (S7) up to Stimulation Day 14 (S14) based upon ovarian response, until recombinant human chorionic gonadotropin (rhcg) administration day. When follicular response was adequate (that is, less than or equal to [=<] 3 follicles with a mean diameter of greater than or equal to [>=] 14 millimeter [mm], and one or two of these follicles with a diameter of >= 17 mm), ovulation was triggered by single 250 mcg subcutaneous r-hcg injection. Duration of treatment cycle was up to adequate Single injection of Enriched (hyperglycosylated r-hfsh), 20 mcg administered subcutaneously on Single injection of Enriched (hyperglycosylated r-hfsh), 30 mcg administered subcutaneously on - Page 16 of 24 -
17 Single injection of Enriched (hyperglycosylated r-hfsh), 40 mcg administered subcutaneously on Follitropin alfa (Gonal-f ) 75 IU administered subcutaneously once daily from S1 up to S14 based upon ovarian response, until r-hcg administration day. When follicular response was adequate (that is, =< 3 follicles with a mean diameter of >=14 mm, and one or two of these follicles with a diameter of >=17 mm), ovulation was triggered by single 250 mcg subcutaneous r-hcg injection. Duration of treatment cycle was up to adequate Measured Values Number of Participants Analyzed Duration of Supplemental Follitropin Alfa Treatment [units: Days] Mean (Standard Deviation) Follitropin Alfa 75 IU 6.6 (1.4) 6.6 (1.4) 7.7 (1.5) 5.6 (2.2) 5.5 (2.7) 5. Secondary Outcome Measure: Measure Title Measure Time Frame Safety Issue? Cumulative Dose of Supplemental Follitropin Alfa Administered Stimulation Day 7 (S7) up to r-hcg administration day (end of stimulation cycle [approximately 14 days]) No Analysis Population PP population included all the randomized participants who were without a medically relevant protocol deviation. 'N' (number of participants analyzed) signifies participants who were evaluable for this measure. - Page 17 of 24 -
18 Reporting Groups Single injection of Enriched (hyperglycosylated recombinant human follicle stimulating hormone [r-hfsh]), 10 microgram (mcg) administered subcutaneously on Stimulation day 1 (S1) followed by a daily dose of follitropin alfa 75 international unit (IU) subcutaneously starting from Stimulation Day 7 (S7) up to Stimulation Day 14 (S14) based upon ovarian response, until recombinant human chorionic gonadotropin (rhcg) administration day. When follicular response was adequate (that is, less than or equal to [=<] 3 follicles with a mean diameter of greater than or equal to [>=] 14 millimeter [mm], and one or two of these follicles with a diameter of >= 17 mm), ovulation was triggered by single 250 mcg subcutaneous r-hcg injection. Duration of treatment cycle was up to adequate Single injection of Enriched (hyperglycosylated r-hfsh), 20 mcg administered subcutaneously on Single injection of Enriched (hyperglycosylated r-hfsh), 30 mcg administered subcutaneously on Single injection of Enriched (hyperglycosylated r-hfsh), 40 mcg administered subcutaneously on Follitropin alfa (Gonal-f ) 75 IU administered subcutaneously once daily from S1 up to S14 based upon ovarian response, until r-hcg administration day. When follicular response was adequate (that is, =< 3 follicles with a mean diameter of >=14 mm, and one or two of these follicles with a diameter of >=17 mm), ovulation was triggered by single 250 mcg subcutaneous r-hcg injection. Duration of treatment cycle was up to adequate Measured Values Number of Participants Analyzed Cumulative Dose of Supplemental Follitropin Alfa Administered Follitropin Alfa 75 IU (122.5) (108.3) (134.2) (167.9) (226.5) - Page 18 of 24 -
19 Follitropin Alfa 75 IU [units: IU] Mean (Standard Deviation) 6. Secondary Outcome Measure: Measure Title Measure Time Frame Safety Issue? Number of Follicles With Mean Diameter Less Than (<) 11 Millimeter (mm) and Greater Than or Equal to (>=) 11 mm Stimulation Day 5 (S5), S7 and r-hcg administration day (end of stimulation cycle [approximately 14 days]) No Analysis Population PP population included all the randomized participants who were without a medically relevant protocol deviation. 'N' (number of participants analyzed) signifies participants who were evaluable for this measure. 'n' signifies number of participants who were evaluable for specified categories at different time points. Reporting Groups Single injection of Enriched (hyperglycosylated recombinant human follicle stimulating hormone [r-hfsh]), 10 microgram (mcg) administered subcutaneously on Stimulation day 1 (S1) followed by a daily dose of follitropin alfa 75 international unit (IU) subcutaneously starting from Stimulation Day 7 (S7) up to Stimulation Day 14 (S14) based upon ovarian response, until recombinant human chorionic gonadotropin (rhcg) administration day. When follicular response was adequate (that is, less than or equal to [=<] 3 follicles with a mean diameter of greater than or equal to [>=] 14 millimeter [mm], and one or two of these follicles with a diameter of >= 17 mm), ovulation was triggered by single 250 mcg subcutaneous r-hcg injection. Duration of treatment cycle was up to adequate Single injection of Enriched (hyperglycosylated r-hfsh), 20 mcg administered subcutaneously on - Page 19 of 24 -
20 Single injection of Enriched (hyperglycosylated r-hfsh), 30 mcg administered subcutaneously on Single injection of Enriched (hyperglycosylated r-hfsh), 40 mcg administered subcutaneously on Follitropin alfa (Gonal-f ) 75 IU administered subcutaneously once daily from S1 up to S14 based upon ovarian response, until r-hcg administration day. When follicular response was adequate (that is, =< 3 follicles with a mean diameter of >=14 mm, and one or two of these follicles with a diameter of >=17 mm), ovulation was triggered by single 250 mcg subcutaneous r-hcg injection. Duration of treatment cycle was up to adequate Measured Values Number of Participants Analyzed Number of Follicles With Mean Diameter Less Than (<) 11 Millimeter (mm) and Greater Than or Equal to (>=) 11 mm [units: Follicles] Mean (Standard Deviation) Follitropin Alfa 75 IU < 11 mm on S5 (n=12,12,12,12,13) 23.6 (13.8) 28.7 (29.4) 23.2 (6.7) 20.9 (16.7) 21.3 (13.7) < 11 mm on S7 (n=12,12,13,12,12) 22.8 (12.6) 27.2 (30.9) 21.0 (8.9) 21.3 (17.3) 22.0 (16.7) < 11 mm on r-hcg Day (n=5,6,7,6,7) 15.6 (7.3) 11.7 (9.2) 18.4 (12.1) 18.7 (15.0) 14.9 (12.4) >= 11 mm on S5 (n=12,8,7,11,8) 1.5 (2.2) 0.0 (0.0) 1.6 (1.9) 2.8 (3.1) 0.8 (0.9) >= 11 mm on S7 (n=11,9,7,11,11) 3.4 (6.2) 0.2 (0.7) 2.6 (3.5) 2.8 (2.4) 1.8 (2.3) >= 11 mm on r-hcg Day (n=5,6,8,6,8) 2.6 (0.9) 2.5 (1.9) 2.5 (1.3) 1.8 (0.8) 2.9 (1.8) - Page 20 of 24 -
21 Reported Adverse Events Time Frame Additional Reporting Groups Stimulation Day 1 (S1) up to Day post r-hcg administration day (end of stimulation cycle [approximately 14 days]) An adverse event (AE) was defined as any untoward medical occurrence in the form of signs, symptoms, abnormal laboratory findings, or diseases that emerges or worsens relative to baseline during a clinical study with an investigational medicinal product (IMP), regardless of causal relationship and even if no IMP has been administered. Single injection of Enriched (hyperglycosylated recombinant human follicle stimulating hormone [r-hfsh]), 10 microgram (mcg) administered subcutaneously on Stimulation day 1 (S1) followed by a daily dose of follitropin alfa 75 international unit (IU) subcutaneously starting from Stimulation Day 7 (S7) up to Stimulation Day 14 (S14) based upon ovarian response, until recombinant human chorionic gonadotropin (rhcg) administration day. When follicular response was adequate (that is, less than or equal to [=<] 3 follicles with a mean diameter of greater than or equal to [>=] 14 millimeter [mm], and one or two of these follicles with a diameter of >= 17 mm), ovulation was triggered by single 250 mcg subcutaneous r-hcg injection. Duration of treatment cycle was up to adequate Single injection of Enriched (hyperglycosylated r-hfsh), 20 mcg administered subcutaneously on Single injection of Enriched (hyperglycosylated r-hfsh), 30 mcg administered subcutaneously on Single injection of Enriched (hyperglycosylated r-hfsh), 40 mcg administered subcutaneously on - Page 21 of 24 -
22 Follitropin alfa (Gonal-f ) 75 IU administered subcutaneously once daily from S1 up to S14 based upon ovarian response, until r-hcg administration day. When follicular response was adequate (that is, =< 3 follicles with a mean diameter of >=14 mm, and one or two of these follicles with a diameter of >=17 mm), ovulation was triggered by single 250 mcg subcutaneous r-hcg injection. Duration of treatment cycle was up to adequate Serious Adverse Events Affected/At Risk (%) Affected/At Risk (%) Affected/At Risk (%) Affected/At Risk (%) Affected/At Risk (%) Total 0/14 (0%) 0/14 (0%) 0/15 (0%) 0/13 (0%) 0/15 (0%) Other Adverse Events Frequency Threshold Above Which Other Adverse Events are Reported: 0% Affected/At Risk (%) Affected/At Risk (%) Affected/At Risk (%) Affected/At Risk (%) Affected/At Risk (%) Total 7/14 (50%) 3/14 (21.43%) 6/15 (40%) 4/13 (30.77%) 7/15 (46.67%) Eye disorders Eye pain A * 0/14 (0%) 0/14 (0%) 0/15 (0%) 1/13 (7.69%) 0/15 (0%) Gastrointestinal disorders Abdominal distension A * 1/14 (7.14%) 1/14 (7.14%) 0/15 (0%) 1/13 (7.69%) 0/15 (0%) Abdominal pain A * 1/14 (7.14%) 1/14 (7.14%) 0/15 (0%) 1/13 (7.69%) 2/15 (13.33%) Abdominal pain upper A * 0/14 (0%) 0/14 (0%) 0/15 (0%) 1/13 (7.69%) 0/15 (0%) Abdominal tenderness A * 1/14 (7.14%) 0/14 (0%) 0/15 (0%) 0/13 (0%) 0/15 (0%) Diarrhoea A * 0/14 (0%) 0/14 (0%) 0/15 (0%) 0/13 (0%) 1/15 (6.67%) Gastrointestinal pain A * 0/14 (0%) 0/14 (0%) 0/15 (0%) 1/13 (7.69%) 0/15 (0%) Nausea A * 1/14 (7.14%) 1/14 (7.14%) 0/15 (0%) 0/13 (0%) 0/15 (0%) Stomach discomfort A * 0/14 (0%) 0/14 (0%) 0/15 (0%) 1/13 (7.69%) 0/15 (0%) - Page 22 of 24 -
23 Affected/At Risk (%) Affected/At Risk (%) Affected/At Risk (%) Affected/At Risk (%) Affected/At Risk (%) General disorders Fatigue A * 1/14 (7.14%) 1/14 (7.14%) 0/15 (0%) 0/13 (0%) 0/15 (0%) Infections and infestations Gastroenteritis A * 0/14 (0%) 0/14 (0%) 0/15 (0%) 0/13 (0%) 1/15 (6.67%) Nasopharyngitis A * 0/14 (0%) 1/14 (7.14%) 0/15 (0%) 1/13 (7.69%) 1/15 (6.67%) Tonsillitis A * 0/14 (0%) 0/14 (0%) 0/15 (0%) 0/13 (0%) 1/15 (6.67%) Upper respiratory tract infection A * 0/14 (0%) 0/14 (0%) 1/15 (6.67%) 0/13 (0%) 0/15 (0%) Urinary tract infection A * 1/14 (7.14%) 0/14 (0%) 0/15 (0%) 0/13 (0%) 0/15 (0%) Injury, poisoning and procedural complications Procedural pain A * 0/14 (0%) 1/14 (7.14%) 0/15 (0%) 0/13 (0%) 0/15 (0%) Musculoskeletal and connective tissue disorders Pain in extremity A * 1/14 (7.14%) 0/14 (0%) 0/15 (0%) 0/13 (0%) 0/15 (0%) Nervous system disorders Dizziness A * 1/14 (7.14%) 0/14 (0%) 0/15 (0%) 1/13 (7.69%) 0/15 (0%) Headache A * 2/14 (14.29%) 1/14 (7.14%) 3/15 (20%) 3/13 (23.08%) 2/15 (13.33%) Migraine A * 0/14 (0%) 0/14 (0%) 1/15 (6.67%) 0/13 (0%) 0/15 (0%) Syncope vasovagal A * 0/14 (0%) 0/14 (0%) 1/15 (6.67%) 0/13 (0%) 0/15 (0%) Reproductive system and breast disorders Adnexa uteri pain A * 1/14 (7.14%) 0/14 (0%) 1/15 (6.67%) 0/13 (0%) 0/15 (0%) Breast pain A * 0/14 (0%) 1/14 (7.14%) 0/15 (0%) 0/13 (0%) 0/15 (0%) Breast tenderness A * 0/14 (0%) 1/14 (7.14%) 1/15 (6.67%) 0/13 (0%) 0/15 (0%) Genital haemorrhage A * 1/14 (7.14%) 0/14 (0%) 0/15 (0%) 0/13 (0%) 0/15 (0%) - Page 23 of 24 -
24 Affected/At Risk (%) Affected/At Risk (%) Affected/At Risk (%) Affected/At Risk (%) Affected/At Risk (%) Ovarian hyperstimulation syndrome A * 0/14 (0%) 0/14 (0%) 0/15 (0%) 1/13 (7.69%) 0/15 (0%) Vaginal discharge A * 0/14 (0%) 1/14 (7.14%) 0/15 (0%) 0/13 (0%) 0/15 (0%) Vaginal pain A * 0/14 (0%) 0/14 (0%) 0/15 (0%) 0/13 (0%) 2/15 (13.33%) Vulvovaginal pruritus A * 1/14 (7.14%) 0/14 (0%) 0/15 (0%) 0/13 (0%) 0/15 (0%) Respiratory, thoracic and mediastinal disorders Skin and subcutaneous tissue disorders Rhinorrhoea A * 0/14 (0%) 0/14 (0%) 0/15 (0%) 0/13 (0%) 1/15 (6.67%) * Indicates events were collected by non-systematic methods. A Term from vocabulary, MedDRA (Unspecified) Urticaria A * 0/14 (0%) 0/14 (0%) 1/15 (6.67%) 0/13 (0%) 0/15 (0%) Limitations and Caveats The study was terminated after Merck Serono had taken the decision not to pursue the development of enriched in ovulation induction More Information Certain Agreements: Principal Investigators are NOT employed by the organization sponsoring the study. There IS an agreement between the Principal Investigator and the Sponsor (or its agents) that restricts the PI's rights to discuss or publish trial results after the trial is completed. Results Point of Contact: Name/Official Title: Merck KGaA Communication Center Organization: Merck Serono, a division of Merck KGaA Phone: service@merckgroup.com U.S. National Library of Medicine U.S. National Institutes of Health U.S. Department of Health & Human Services - Page 24 of 24 -
ClinicalTrials.gov Protocol Registration and Results System (PRS) Receipt Release Date: 01/20/2014. ClinicalTrials.gov ID: NCT
 ClinicalTrials.gov Protocol Registration and Results System (PRS) Receipt Release Date: 01/20/2014 ClinicalTrials.gov ID: NCT00829244 Study Identification Unique Protocol ID: 28613 Brief Title: CONSORT
ClinicalTrials.gov Protocol Registration and Results System (PRS) Receipt Release Date: 01/20/2014 ClinicalTrials.gov ID: NCT00829244 Study Identification Unique Protocol ID: 28613 Brief Title: CONSORT
ClinicalTrials.gov Protocol Registration and Results System (PRS) Receipt Release Date: 01/19/2016. ClinicalTrials.gov ID: NCT
 ClinicalTrials.gov Protocol Registration and Results System (PRS) Receipt Release Date: 01/19/2016 ClinicalTrials.gov ID: NCT00595413 Study Identification Unique Protocol ID: 27905 Brief Title: Atacicept
ClinicalTrials.gov Protocol Registration and Results System (PRS) Receipt Release Date: 01/19/2016 ClinicalTrials.gov ID: NCT00595413 Study Identification Unique Protocol ID: 27905 Brief Title: Atacicept
ClinicalTrials.gov Protocol Registration and Results System (PRS) Receipt Release Date: 10/11/2013. ClinicalTrials.gov ID: NCT
 ClinicalTrials.gov Protocol Registration and Results System (PRS) Receipt Release Date: 10/11/2013 ClinicalTrials.gov ID: NCT00168454 Study Identification Unique Protocol ID: 191622-077 Brief Title: A
ClinicalTrials.gov Protocol Registration and Results System (PRS) Receipt Release Date: 10/11/2013 ClinicalTrials.gov ID: NCT00168454 Study Identification Unique Protocol ID: 191622-077 Brief Title: A
ClinicalTrials.gov Protocol Registration and Results System (PRS) Receipt Release Date: June 25, ClinicalTrials.gov ID: NCT
 ClinicalTrials.gov Protocol Registration and Results System (PRS) Receipt Release Date: June 25, 2014 ClinicalTrials.gov ID: NCT00372385 Study Identification Unique Protocol ID: VX05-950-104EU Brief Title:
ClinicalTrials.gov Protocol Registration and Results System (PRS) Receipt Release Date: June 25, 2014 ClinicalTrials.gov ID: NCT00372385 Study Identification Unique Protocol ID: VX05-950-104EU Brief Title:
ClinicalTrials.gov Protocol and Results Registration System (PRS) Receipt Release Date: 09/30/2015. ClinicalTrials.gov ID: NCT
 ClinicalTrials.gov Protocol and Results Registration System (PRS) Receipt Release Date: 09/30/2015 ClinicalTrials.gov ID: NCT01378962 Study Identification Unique Protocol ID: ML25514 Brief Title: A Study
ClinicalTrials.gov Protocol and Results Registration System (PRS) Receipt Release Date: 09/30/2015 ClinicalTrials.gov ID: NCT01378962 Study Identification Unique Protocol ID: ML25514 Brief Title: A Study
Infertility: failure to conceive within one year of unprotected regular sexual intercourse. Primary secondary
 Subfertility Infertility: failure to conceive within one year of unprotected regular sexual intercourse. Primary secondary Infertility affects about 15 % of couples. age of the female. Other factors that
Subfertility Infertility: failure to conceive within one year of unprotected regular sexual intercourse. Primary secondary Infertility affects about 15 % of couples. age of the female. Other factors that
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION. 22 September 2010
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 22 September 2010 100 µg/0.5 ml, solution for injection B/1 prefilled syringe + 1 needle (CIP code: 374 590-1) 150
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 22 September 2010 100 µg/0.5 ml, solution for injection B/1 prefilled syringe + 1 needle (CIP code: 374 590-1) 150
Core Safety Profile. Pharmaceutical form(s)/strength: Lyophilised powder for injection / 75 IU. Date of FAR:
 Core Safety Profile Active substance: Urofollitropin Pharmaceutical form(s)/strength: Lyophilised powder for injection / 75 IU P - RMS: UK/H/PSUR/0059/001 Date of FAR: 04.12.2009 4.2 Posology and method
Core Safety Profile Active substance: Urofollitropin Pharmaceutical form(s)/strength: Lyophilised powder for injection / 75 IU P - RMS: UK/H/PSUR/0059/001 Date of FAR: 04.12.2009 4.2 Posology and method
Previous Study Return to List Next Study
 A service of the U.S. National Institutes of Health Trial record 1 of 1 for: 42603ATT3013 Previous Study Return to List Next Study A Study to Evaluate Effectiveness and Safety of Prolonged Release OROS
A service of the U.S. National Institutes of Health Trial record 1 of 1 for: 42603ATT3013 Previous Study Return to List Next Study A Study to Evaluate Effectiveness and Safety of Prolonged Release OROS
GONAL-f PEN Recombinant human follicle stimulating hormone (follitropin alfa [rch])
![GONAL-f PEN Recombinant human follicle stimulating hormone (follitropin alfa [rch]) GONAL-f PEN Recombinant human follicle stimulating hormone (follitropin alfa [rch])](/thumbs/77/75508754.jpg) GONAL-f PEN Recombinant human follicle stimulating hormone (follitropin alfa [rch]) Consumer Medicine Information What is in this leaflet This leaflet answers some common questions about GONALf. It does
GONAL-f PEN Recombinant human follicle stimulating hormone (follitropin alfa [rch]) Consumer Medicine Information What is in this leaflet This leaflet answers some common questions about GONALf. It does
Efficacy Study of Zoledronic Acid and Teriparatide Combination Therapy in Women With Osteoporosis
 A service of the U.S. National Institutes of Health Trial record 1 of 1 for: CZOL446H2409 Previous Study Return to List Next Study Efficacy Study of Zoledronic Acid and Combination Therapy in Women With
A service of the U.S. National Institutes of Health Trial record 1 of 1 for: CZOL446H2409 Previous Study Return to List Next Study Efficacy Study of Zoledronic Acid and Combination Therapy in Women With
GONAL-f PEN Recombinant human follicle stimulating hormone (follitropin alfa [rch])
![GONAL-f PEN Recombinant human follicle stimulating hormone (follitropin alfa [rch]) GONAL-f PEN Recombinant human follicle stimulating hormone (follitropin alfa [rch])](/thumbs/88/115281160.jpg) PEN Recombinant human follicle stimulating hormone (follitropin alfa [rch]) Consumer Medicine Information What is in this leaflet This leaflet answers some common questions about It does not contain all
PEN Recombinant human follicle stimulating hormone (follitropin alfa [rch]) Consumer Medicine Information What is in this leaflet This leaflet answers some common questions about It does not contain all
GONAL-f (follitropin alfa [rch]) solution for injection
![GONAL-f (follitropin alfa [rch]) solution for injection GONAL-f (follitropin alfa [rch]) solution for injection](/thumbs/94/120349310.jpg) (follitropin alfa [rch]) solution for injection Consumer Medicine Information What is in this leaflet This leaflet answers some common questions about. It does not contain all the available information.
(follitropin alfa [rch]) solution for injection Consumer Medicine Information What is in this leaflet This leaflet answers some common questions about. It does not contain all the available information.
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION. 25 June 2008
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 25 June 2008 PERGOVERIS 150 IU/75 IU, powder and solvent for solution for injection B/1 glass vial - one 1 ml vial
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 25 June 2008 PERGOVERIS 150 IU/75 IU, powder and solvent for solution for injection B/1 glass vial - one 1 ml vial
GONAL-F THE COMPLEX PROCESS OF FERTILITY HORMONE DEFICIENCIES CAN LEAD TO INFERTILITY PROBLEMS WHAT IS GONAL-F? HCP FACT SHEET
 HCP FACT SHEET GONAL-F GONAL-f (recombinant follitropin alfa) is prescribed to supplement or replace naturally occurring folliclestimulating hormone (FSH), an essential hormone to treat infertility in
HCP FACT SHEET GONAL-F GONAL-f (recombinant follitropin alfa) is prescribed to supplement or replace naturally occurring folliclestimulating hormone (FSH), an essential hormone to treat infertility in
Infertility DR. RAHUL BEVARA
 Infertility DR. RAHUL BEVARA Definitions Infertility is defined as the inability to conceive after one year of unprotected coitus. Affects 10-15% of couples Primary Infertility, that is inability to conceive
Infertility DR. RAHUL BEVARA Definitions Infertility is defined as the inability to conceive after one year of unprotected coitus. Affects 10-15% of couples Primary Infertility, that is inability to conceive
Package leaflet: Information for the user
 Package leaflet: Information for the user Bemfola 75 IU/0.125 ml solution for injection in a pre-filled pen Bemfola 150 IU/0.25 ml solution for injection in a pre-filled pen Bemfola 225 IU/0.375 ml solution
Package leaflet: Information for the user Bemfola 75 IU/0.125 ml solution for injection in a pre-filled pen Bemfola 150 IU/0.25 ml solution for injection in a pre-filled pen Bemfola 225 IU/0.375 ml solution
Efficacy and Safety of Diclofenac Potassium 25 mg Tablet Taken Three Times Daily in Subjects With Acute Joint Pain
 Page 1 of 8 A service of the U.S. National Institutes of Health Try our beta test site Trial record 1 of 1 for: 853-P-401 Previous Study Return to List Next Study Efficacy and Safety of Taken Three Times
Page 1 of 8 A service of the U.S. National Institutes of Health Try our beta test site Trial record 1 of 1 for: 853-P-401 Previous Study Return to List Next Study Efficacy and Safety of Taken Three Times
FEMALE PATIENT HISTORY
 ew Hope. ew Life. ew Beginnings. A Division of MID-ATLATIC WOME S CARE, PLC FEMALE PATIET HISTORY PLEASE OTE: Infertility patients please complete ALL sections. All other patients, complete section 1.,
ew Hope. ew Life. ew Beginnings. A Division of MID-ATLATIC WOME S CARE, PLC FEMALE PATIET HISTORY PLEASE OTE: Infertility patients please complete ALL sections. All other patients, complete section 1.,
 www.iffs-reproduction.org @IntFertilitySoc Int@FedFertilitySoc Conflict of interest none Outline Causes of ovulatory dysfunction Assessment of women with ovulatory dysfunction Management First line Second
www.iffs-reproduction.org @IntFertilitySoc Int@FedFertilitySoc Conflict of interest none Outline Causes of ovulatory dysfunction Assessment of women with ovulatory dysfunction Management First line Second
ClinicalTrials.gov Protocol Registration and Results System (PRS) Receipt Release Date: 08/01/2013. ClinicalTrials.gov ID: NCT
 ClinicalTrials.gov Protocol Registration and Results System (PRS) Receipt Release Date: 08/01/2013 ClinicalTrials.gov ID: NCT00933686 Study Identification Unique Protocol ID: 27560 Brief Title: Growth
ClinicalTrials.gov Protocol Registration and Results System (PRS) Receipt Release Date: 08/01/2013 ClinicalTrials.gov ID: NCT00933686 Study Identification Unique Protocol ID: 27560 Brief Title: Growth
Infertility. Review and Update Clifford C. Hayslip MD Intrauterine Inseminations
 Infertility Review and Update Clifford C. Hayslip MD Intrauterine Inseminations Beneficial effects of IUI not consistently documented in studies No deleterious effects on fertility 3-4 cycles of IUI should
Infertility Review and Update Clifford C. Hayslip MD Intrauterine Inseminations Beneficial effects of IUI not consistently documented in studies No deleterious effects on fertility 3-4 cycles of IUI should
Polycystic Ovary Syndrome HEATHER BURKS, MD OU PHYSICIANS REPRODUCTIVE MEDICINE SEPTEMBER 21, 2018
 Polycystic Ovary Syndrome HEATHER BURKS, MD OU PHYSICIANS REPRODUCTIVE MEDICINE SEPTEMBER 21, 2018 Learning Objectives At the conclusion of this lecture, learners should: 1) Know the various diagnostic
Polycystic Ovary Syndrome HEATHER BURKS, MD OU PHYSICIANS REPRODUCTIVE MEDICINE SEPTEMBER 21, 2018 Learning Objectives At the conclusion of this lecture, learners should: 1) Know the various diagnostic
Package leaflet: Information for the user. GONAL-f 450 IU/0.75 ml (33 micrograms/0.75 ml) solution for injection in a pre-filled pen Follitropin alfa
 Package leaflet: Information for the user GONAL-f 450 IU/0.75 ml (33 micrograms/0.75 ml) solution for injection in a pre-filled pen Follitropin alfa Read all of this leaflet carefully before you start
Package leaflet: Information for the user GONAL-f 450 IU/0.75 ml (33 micrograms/0.75 ml) solution for injection in a pre-filled pen Follitropin alfa Read all of this leaflet carefully before you start
Original Article. Fauzia HaqNawaz 1*, Saadia Virk 2, Tasleem Qadir 3, Saadia Imam 3, Javed Rizvi 2
 Original Article Comparison of Letrozole and Clomiphene Citrate Efficacy along with Gonadotrophins in Controlled Ovarian Hyperstimulation for Intrauterine Insemination Cycles Fauzia HaqNawaz 1*, Saadia
Original Article Comparison of Letrozole and Clomiphene Citrate Efficacy along with Gonadotrophins in Controlled Ovarian Hyperstimulation for Intrauterine Insemination Cycles Fauzia HaqNawaz 1*, Saadia
In Vitro Fertilization in Clomiphene-Resistant Women with Polycystic Ovary Syndrome
 Original Article Effect of Laparoscopic Ovarian Drilling on Outcomes of In Vitro Fertilization in Clomiphene-Resistant Women with Polycystic Ovary Syndrome Maryam Eftekhar, M.D. 1, Razieh Deghani Firoozabadi,
Original Article Effect of Laparoscopic Ovarian Drilling on Outcomes of In Vitro Fertilization in Clomiphene-Resistant Women with Polycystic Ovary Syndrome Maryam Eftekhar, M.D. 1, Razieh Deghani Firoozabadi,
Orgalutran 0.25 mg/0.5 ml solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION
 1 1. NAME OF THE MEDICINAL PRODUCT 0.25 mg/0.5 ml solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each pre-filled syringe contains 0.25 mg of ganirelix (INN) in 0.5 mg aqueous solution.
1 1. NAME OF THE MEDICINAL PRODUCT 0.25 mg/0.5 ml solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each pre-filled syringe contains 0.25 mg of ganirelix (INN) in 0.5 mg aqueous solution.
Polycystic Ovary Syndrome
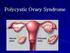 Polycystic Ovary Syndrome Definition: the diagnostic criteria Evidence of hyperandrogenism, biochemical &/or clinical (hirsutism, acne & male pattern baldness). Ovulatory dysfunction; amenorrhoea; oligomenorrhoea
Polycystic Ovary Syndrome Definition: the diagnostic criteria Evidence of hyperandrogenism, biochemical &/or clinical (hirsutism, acne & male pattern baldness). Ovulatory dysfunction; amenorrhoea; oligomenorrhoea
Follitropin alfa (rch)/lutropin alfa (rch) Powder for Injection. This leaflet answers some common questions about PERGOVERIS.
 PERGOVERIS 150 IU / 75 IU Follitropin alfa (rch)/lutropin alfa (rch) Powder for Injection Consumer Medicine Information What is in this leaflet This leaflet answers some common questions about PERGOVERIS.
PERGOVERIS 150 IU / 75 IU Follitropin alfa (rch)/lutropin alfa (rch) Powder for Injection Consumer Medicine Information What is in this leaflet This leaflet answers some common questions about PERGOVERIS.
Link between effectiveness and cost data The costing was undertaken prospectively on the same patient sample that provided the effectiveness data.
 Recombinant versus highly-purified, urinary follicle-stimulating hormone (r-fsh vs. HPuFSH) in ovulation induction: a prospective, randomized study with cost-minimization analysis Revelli A, Poso F, Gennarelli
Recombinant versus highly-purified, urinary follicle-stimulating hormone (r-fsh vs. HPuFSH) in ovulation induction: a prospective, randomized study with cost-minimization analysis Revelli A, Poso F, Gennarelli
NEW PATIENT DATA SHEET Please complete as best you can. It is not necessary to have all information before speaking with a doctor. PATIENT INFORMATION
 NEW PATIENT DATA SHEET Please complete as best you can. It is not necessary to have all information before speaking with a doctor. PATIENT INFORMATION PATIENT NAME DOB AGE PARTNER NAME DOB AGE STREET CITY
NEW PATIENT DATA SHEET Please complete as best you can. It is not necessary to have all information before speaking with a doctor. PATIENT INFORMATION PATIENT NAME DOB AGE PARTNER NAME DOB AGE STREET CITY
Cigna Drug and Biologic Coverage Policy
 Cigna Drug and Biologic Coverage Policy Subject Infertility Injectables Table of Contents Coverage Policy... 1 General Background...16 Coding/Billing Information...20 References...20 Effective Date...
Cigna Drug and Biologic Coverage Policy Subject Infertility Injectables Table of Contents Coverage Policy... 1 General Background...16 Coding/Billing Information...20 References...20 Effective Date...
Neil Goodman, MD, FACE
 Initial Workup of Infertile Couple: Female Neil Goodman, MD, FACE Professor of Medicine Voluntary Faculty University of Miami Miller School of Medicine Scope of Infertility in the United States Affects
Initial Workup of Infertile Couple: Female Neil Goodman, MD, FACE Professor of Medicine Voluntary Faculty University of Miami Miller School of Medicine Scope of Infertility in the United States Affects
CONSENT FORM FOR TREATMENT WITH OVULATION INDUCTION MEDICATIONS AND INTRAUTERINE INSEMINATIONS
 CONSENT FORM FOR TREATMENT WITH OVULATION INDUCTION MEDICATIONS AND INTRAUTERINE INSEMINATIONS INSTRUCTIONS: This consent form provides a description of the treatment that you are undertaking. Read the
CONSENT FORM FOR TREATMENT WITH OVULATION INDUCTION MEDICATIONS AND INTRAUTERINE INSEMINATIONS INSTRUCTIONS: This consent form provides a description of the treatment that you are undertaking. Read the
A service of the U.S. National Institutes of Health Now Available: Final Rule for FDAAA 801 and NIH Policy on Clinical Trial Reporting
 A service of the U.S. National Institutes of Health w Available: Final Rule for FDAAA 81 and NIH Policy on Clinical Trial Reporting Trial record 1 of 1 for: FENHYDPAI414 Previous Study Return to List Next
A service of the U.S. National Institutes of Health w Available: Final Rule for FDAAA 81 and NIH Policy on Clinical Trial Reporting Trial record 1 of 1 for: FENHYDPAI414 Previous Study Return to List Next
New Patient Medical History
 New Patient Medical History MR #: Initial Appointment Date: / / Name: Birth Date: / / Address: City: State: Zip: Best Phone # to reach you: ( ) Second contact #: ( ) Email Address: Occupation: Marital
New Patient Medical History MR #: Initial Appointment Date: / / Name: Birth Date: / / Address: City: State: Zip: Best Phone # to reach you: ( ) Second contact #: ( ) Email Address: Occupation: Marital
Trial record 1 of 1 for: 075-A-301 Previous Study Return to List Next Study
 Efficacy and Safety of a Sore Throat Lozenge Containing Lidocaine and Cetylpyridinium Chloride in Patients With Sore Throat Due to a Comm... Page 1 of 10 A service of the U.S. National Institutes of Health
Efficacy and Safety of a Sore Throat Lozenge Containing Lidocaine and Cetylpyridinium Chloride in Patients With Sore Throat Due to a Comm... Page 1 of 10 A service of the U.S. National Institutes of Health
1. NAME OF THE MEDICINAL PRODUCT. Gonapeptyl 0.1 mg/1 ml solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION
 1. NAME OF THE MEDICINAL PRODUCT Gonapeptyl 0.1 mg/1 ml solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each pre-filled syringe of 1 ml solution for injection contains 100 micrograms
1. NAME OF THE MEDICINAL PRODUCT Gonapeptyl 0.1 mg/1 ml solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each pre-filled syringe of 1 ml solution for injection contains 100 micrograms
Introduction to Intrauterine Insemination (IUI) Service
 Introduction to Intrauterine Insemination (IUI) Service Assisted Reproductive Technology Unit The Chinese University of Hong Kong Prince of Wales Hospital 2017 Treatment Procedures IUI involves six main
Introduction to Intrauterine Insemination (IUI) Service Assisted Reproductive Technology Unit The Chinese University of Hong Kong Prince of Wales Hospital 2017 Treatment Procedures IUI involves six main
Assisted Reproduction. By Dr. Afraa Mahjoob Al-Naddawi
 Assisted Reproduction By Dr. Afraa Mahjoob Al-Naddawi Learning Objectives: By the end of this lecture, you will be able to: 1) Define assisted reproductive techniques (ART). 2) List indications for various
Assisted Reproduction By Dr. Afraa Mahjoob Al-Naddawi Learning Objectives: By the end of this lecture, you will be able to: 1) Define assisted reproductive techniques (ART). 2) List indications for various
Common protocols in intra-uterine insemination cycles
 Common protocols in intra-uterine insemination cycles Doç. Dr. Candan İltemir Duvan Turgut Özal Üniversitesi Tıp Fakültesi Kadın Hastalıkları ve Doğum AD Ovulation induction with intra-uterine insemination
Common protocols in intra-uterine insemination cycles Doç. Dr. Candan İltemir Duvan Turgut Özal Üniversitesi Tıp Fakültesi Kadın Hastalıkları ve Doğum AD Ovulation induction with intra-uterine insemination
PRESCRIBING INFORMATION UK AND IRELAND Please refer to the Summary of Product Characteristics for further information
 PRESCRIBING INFORMATION UK AND IRELAND Please refer to the Summary of Product Characteristics for further information Cetrotide 0.25 mg powder and solvent for solution for injection cetrorelix acetate.
PRESCRIBING INFORMATION UK AND IRELAND Please refer to the Summary of Product Characteristics for further information Cetrotide 0.25 mg powder and solvent for solution for injection cetrorelix acetate.
Search for studies: ClinicalTrials.gov Identifier: NCT
 ClinicalTrials.gov A service of the U.S. National Institutes of Health Search for studies: Example. "Heart attack" AND "Los Angeles" Advanced Search Help Studies by Topic Glossary Find Studies About Clinical
ClinicalTrials.gov A service of the U.S. National Institutes of Health Search for studies: Example. "Heart attack" AND "Los Angeles" Advanced Search Help Studies by Topic Glossary Find Studies About Clinical
Intra uterine insemination (IUI) Information for Patients and Partners
 Intra uterine insemination (IUI) Information for Patients and Partners What is this leaflet about and who is it for? This leaflet is produced to inform couples undergoing IUI (intrauterine insemination)
Intra uterine insemination (IUI) Information for Patients and Partners What is this leaflet about and who is it for? This leaflet is produced to inform couples undergoing IUI (intrauterine insemination)
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 This medicinal product is subject to additional monitoring. This will allow quick identification of new safety information. Healthcare professionals are asked
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 This medicinal product is subject to additional monitoring. This will allow quick identification of new safety information. Healthcare professionals are asked
In Vitro Fertilization What to expect
 Patient Education In Vitro Fertilization What to expect This handout describes how to prepare for and what to expect when you have in vitro fertilization. It provides written information about this process,
Patient Education In Vitro Fertilization What to expect This handout describes how to prepare for and what to expect when you have in vitro fertilization. It provides written information about this process,
GPVTS TEACHING APRIL 2016 FERTILITY
 GPVTS TEACHING APRIL 2016 FERTILITY Djavid Alleemudder - Consultant Obstetrics & Gynaecology DEFINITIONS What is the definition of infertility? Failure to conceive after 12 months despite regular, unprotected
GPVTS TEACHING APRIL 2016 FERTILITY Djavid Alleemudder - Consultant Obstetrics & Gynaecology DEFINITIONS What is the definition of infertility? Failure to conceive after 12 months despite regular, unprotected
INFERTILITY CAUSES. Basic evaluation of the female
 INFERTILITY Infertility is the inability to conceive after 12 months of unprotected intercourse. There are multiple causes of infertility and a systematic way to evaluate the condition. Let s look at some
INFERTILITY Infertility is the inability to conceive after 12 months of unprotected intercourse. There are multiple causes of infertility and a systematic way to evaluate the condition. Let s look at some
Please fill out the following information and have it returned to our office prior to your consultation.
 Please fill out the following information and have it returned to our office prior to your consultation. Patient s Name Partner s Name Address: City: State: Zip: Phone (day#): ( ) (eve#) ( ) (cell) ( )
Please fill out the following information and have it returned to our office prior to your consultation. Patient s Name Partner s Name Address: City: State: Zip: Phone (day#): ( ) (eve#) ( ) (cell) ( )
Injection: Gonal-f RFF Redi-ject 900 International Units per 1.5 ml in prefilled, multiple dose disposable
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use Gonal-f RFF Redi-ject safely and effectively. See full prescribing information for Gonal-f RFF Redi-ject
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use Gonal-f RFF Redi-ject safely and effectively. See full prescribing information for Gonal-f RFF Redi-ject
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Fertavid 50 IU/0.5 ml solution for injection Fertavid 75 IU/0.5 ml solution for injection Fertavid 100 IU/0.5 ml solution for
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Fertavid 50 IU/0.5 ml solution for injection Fertavid 75 IU/0.5 ml solution for injection Fertavid 100 IU/0.5 ml solution for
Infertility History Form
 Date form completed: Infertility History Form Patient s name: _ Age: Date of Birth: Occupation: Partner s name: Age: Date of Birth: Occupation: Prior marriage: Yes No # Prior marriage: Yes No # Attempted
Date form completed: Infertility History Form Patient s name: _ Age: Date of Birth: Occupation: Partner s name: Age: Date of Birth: Occupation: Prior marriage: Yes No # Prior marriage: Yes No # Attempted
Relation between the Number and Size of Follicles in Ovulation Induction and the Rate of Pregnancy
 Relation between the Number and Size of Follicles in Ovulation Induction and the Rate of Pregnancy Aseel Mosa Jabber M.SC.G.O. The department of Obstetrics and Gynecology, Faculty of Medicine Thi-qar university
Relation between the Number and Size of Follicles in Ovulation Induction and the Rate of Pregnancy Aseel Mosa Jabber M.SC.G.O. The department of Obstetrics and Gynecology, Faculty of Medicine Thi-qar university
PUREGON follitropin beta[rch]
![PUREGON follitropin beta[rch] PUREGON follitropin beta[rch]](/thumbs/86/94247981.jpg) PUREGON follitropin beta[rch] Consumer Medicine Information What is in this leaflet This leaflet answers some common questions about PUREGON. It does not contain all the available information. It does
PUREGON follitropin beta[rch] Consumer Medicine Information What is in this leaflet This leaflet answers some common questions about PUREGON. It does not contain all the available information. It does
Principles of Ovarian Stimulation
 Principles of Ovarian Stimulation Dr Genia Rozen Gynaecologist and Fertility Specialist Royal Women s Hospital and Melbourne IVF Learning objectives Why ovarian stimulation Recap physiology Ovarian cycle
Principles of Ovarian Stimulation Dr Genia Rozen Gynaecologist and Fertility Specialist Royal Women s Hospital and Melbourne IVF Learning objectives Why ovarian stimulation Recap physiology Ovarian cycle
PUREGON follitropin beta[rch]
![PUREGON follitropin beta[rch] PUREGON follitropin beta[rch]](/thumbs/94/120349294.jpg) PUREGON follitropin beta[rch] Consumer Medicine Information What is in this leaflet This leaflet answers some common questions about PUREGON. It does not contain all the available information. It does
PUREGON follitropin beta[rch] Consumer Medicine Information What is in this leaflet This leaflet answers some common questions about PUREGON. It does not contain all the available information. It does
Patient Past Medical History
 Patient Past Medical History A. Identifying Data Date this form when completed Your name Partner's name Age Birth date Height Weight Length of marriage (or relationship) How long have you been trying unsuccessfully
Patient Past Medical History A. Identifying Data Date this form when completed Your name Partner's name Age Birth date Height Weight Length of marriage (or relationship) How long have you been trying unsuccessfully
Type of intervention Treatment. Economic study type Cost-effectiveness analysis.
 Recombinant versus urinary follicle-stimulating hormone in intrauterine insemination cycles: a prospective, randomized analysis of cost effectiveness Gerli S, Casini M L, Unfer V, Costabile L, Bini V,
Recombinant versus urinary follicle-stimulating hormone in intrauterine insemination cycles: a prospective, randomized analysis of cost effectiveness Gerli S, Casini M L, Unfer V, Costabile L, Bini V,
AUSTRALIAN PRODUCT INFORMATION ORGALUTRAN (ganirelix acetate) Solution for Injection
 AUSTRALIAN PRODUCT INFORMATION ORGALUTRAN (ganirelix acetate) Solution for Injection 1 NAME OF THE MEDICINE Ganirelix acetate 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Orgalutran 250 µg ganirelix (as
AUSTRALIAN PRODUCT INFORMATION ORGALUTRAN (ganirelix acetate) Solution for Injection 1 NAME OF THE MEDICINE Ganirelix acetate 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Orgalutran 250 µg ganirelix (as
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT GONAL-f 75 IU (5.5 micrograms) powder and solvent for solution for injection. 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT GONAL-f 75 IU (5.5 micrograms) powder and solvent for solution for injection. 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each
In Vitro Fertilization
 Patient Education In Vitro Fertilization About the treatment This handout describes how to prepare for and what to expect when you have in vitro fertilization. It provides written information about this
Patient Education In Vitro Fertilization About the treatment This handout describes how to prepare for and what to expect when you have in vitro fertilization. It provides written information about this
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 This medicinal product is subject to additional monitoring. This will allow quick identification of new safety information. Healthcare professionals are asked
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 This medicinal product is subject to additional monitoring. This will allow quick identification of new safety information. Healthcare professionals are asked
Dosage and Administration, Recommended Dosing for Induction of Spermatogenesis in Men (2.4) 6/2010
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use Follistim AQ safely and effectively. See full prescribing information for Follistim AQ. Follistim
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use Follistim AQ safely and effectively. See full prescribing information for Follistim AQ. Follistim
Infertility F REQUENTLY A SKED Q UESTIONS. Q: Is infertility a common problem?
 Infertility (female factors). In another one third of cases, infertility is due to the man (male factors). The remaining cases are caused by a mixture of male and female factors or by unknown factors.
Infertility (female factors). In another one third of cases, infertility is due to the man (male factors). The remaining cases are caused by a mixture of male and female factors or by unknown factors.
Gonadotrophin treatment in patients with Polycystic Ovary Syndrome
 Int. J. Adv. Res. Biol. Sci. (218). 5(4): 95-99 International Journal of Advanced Research in Biological Sciences ISSN: 2348-869 www.ijarbs.com DOI: 1.22192/ijarbs Coden: IJARQG(USA) Volume 5, Issue 4-218
Int. J. Adv. Res. Biol. Sci. (218). 5(4): 95-99 International Journal of Advanced Research in Biological Sciences ISSN: 2348-869 www.ijarbs.com DOI: 1.22192/ijarbs Coden: IJARQG(USA) Volume 5, Issue 4-218
hmg-ibsa Final Report, 06 June 2014
 1 TITLE PAGE Safety and efficacy study comparing a new hmg formulation (hmg-ibsa) to a reference product (Menopur ) in patients undergoing ovarian stimulation for in vitro fertilisation (IVF). Study No:
1 TITLE PAGE Safety and efficacy study comparing a new hmg formulation (hmg-ibsa) to a reference product (Menopur ) in patients undergoing ovarian stimulation for in vitro fertilisation (IVF). Study No:
Fertility Specialty Care
 Fertility Specialty Care PATIENT INFORMATION: Last Name First Name & Initial Address City State Zip Home Phone ( ) Cell Phone ( ) Date of Birth Social Security Number Marital Status: Married Single Ethnicity:
Fertility Specialty Care PATIENT INFORMATION: Last Name First Name & Initial Address City State Zip Home Phone ( ) Cell Phone ( ) Date of Birth Social Security Number Marital Status: Married Single Ethnicity:
Full Novartis CTRD Results Template
 Full Novartis CTRD Results Template Sponsor Novartis Generic Drug Name vildagliptin Therapeutic Area of Trial Type 2 diabetes Approved Indication Type 2 diabetes Protocol Number CLAF237A23137E1 Title A
Full Novartis CTRD Results Template Sponsor Novartis Generic Drug Name vildagliptin Therapeutic Area of Trial Type 2 diabetes Approved Indication Type 2 diabetes Protocol Number CLAF237A23137E1 Title A
Prescribing Information GONAL F
 Prescribing Information GONAL F 1. NAME OF THE MEDICINAL PRODUCT GONAL-f 300 IU/0.5 ml (22 micrograms/0.5 ml) solution for injection in a pre-filled pen. GONAL-f 450 IU/0.75 ml (33 micrograms/0.75 ml)
Prescribing Information GONAL F 1. NAME OF THE MEDICINAL PRODUCT GONAL-f 300 IU/0.5 ml (22 micrograms/0.5 ml) solution for injection in a pre-filled pen. GONAL-f 450 IU/0.75 ml (33 micrograms/0.75 ml)
Pergoveris 150IU/75IU
 Prescribing Information Pergoveris 150IU/75IU 1. NAME OF THE MEDICINAL PRODUCT Pergoveris 150 IU/75 IU powder and solvent for solution for injection. 2. QUALITATIVE AND QUANTITATIVE COMPOSITION One vial
Prescribing Information Pergoveris 150IU/75IU 1. NAME OF THE MEDICINAL PRODUCT Pergoveris 150 IU/75 IU powder and solvent for solution for injection. 2. QUALITATIVE AND QUANTITATIVE COMPOSITION One vial
Fertility Treatment: Do not be Distracted
 Fertility Treatment: Do not be Distracted Fertility Treatment: do not be distracted by worthless recommendation Fertility Treatment: Do not be Distracted When contemplating options for fertility treatment
Fertility Treatment: Do not be Distracted Fertility Treatment: do not be distracted by worthless recommendation Fertility Treatment: Do not be Distracted When contemplating options for fertility treatment
Women's Health, Naturally Fertility Questionnaire
 Women's Health, Naturally Fertility Questionnaire Name : Age: Date of Birth: Tel. #-Day: - - Evening: -- - Partner's Name: Partner's date of birth: GYNECOLOGICAL HISTORY How old were you when you had your
Women's Health, Naturally Fertility Questionnaire Name : Age: Date of Birth: Tel. #-Day: - - Evening: -- - Partner's Name: Partner's date of birth: GYNECOLOGICAL HISTORY How old were you when you had your
Minimal stimulation protocol for use with intrauterine insemination in the treatment of infertility Dhaliwal L K, Sialy R K, Gopalan S, Majumdar S
 Minimal stimulation protocol for use with intrauterine insemination in the treatment of infertility Dhaliwal L K, Sialy R K, Gopalan S, Majumdar S Record Status This is a critical abstract of an economic
Minimal stimulation protocol for use with intrauterine insemination in the treatment of infertility Dhaliwal L K, Sialy R K, Gopalan S, Majumdar S Record Status This is a critical abstract of an economic
NEW ZEALAND DATA SHEET
 NEW ZEALAND DATA SHEET 1 ORGALUTRAN 250 µg/0.5 ml solution for injection 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Ganirelix (as the acetate salt) 0.5 mg/ml. ORGALUTRAN contains the synthetic decapeptide
NEW ZEALAND DATA SHEET 1 ORGALUTRAN 250 µg/0.5 ml solution for injection 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Ganirelix (as the acetate salt) 0.5 mg/ml. ORGALUTRAN contains the synthetic decapeptide
Infertility treatment
 In the name of God Infertility treatment Treatment options The optimal treatment is one that provide an acceptable success rate, has minimal risk and is costeffective. The treatment options are: 1- Ovulation
In the name of God Infertility treatment Treatment options The optimal treatment is one that provide an acceptable success rate, has minimal risk and is costeffective. The treatment options are: 1- Ovulation
Drug Therapy Guidelines
 Drug Therapy Guidelines Applicable Injectable Fertility Medications: Bravelle, Cetrotide, Follistim AQ, Ganirelix, Gonal-F, human chorionic gonadotropin, leuprolide, Menopur, Novarel, Ovidrel, Pregnyl,
Drug Therapy Guidelines Applicable Injectable Fertility Medications: Bravelle, Cetrotide, Follistim AQ, Ganirelix, Gonal-F, human chorionic gonadotropin, leuprolide, Menopur, Novarel, Ovidrel, Pregnyl,
Drug Therapy Guidelines
 Drug Therapy Guidelines Applicable Medical Benefit Effective: 8/15/18 Pharmacy- Formulary 1 x Next Review: 6/18 Pharmacy- Formulary 2 x Date of Origin: 7/00 Injectable Fertility Medications: Bravelle,
Drug Therapy Guidelines Applicable Medical Benefit Effective: 8/15/18 Pharmacy- Formulary 1 x Next Review: 6/18 Pharmacy- Formulary 2 x Date of Origin: 7/00 Injectable Fertility Medications: Bravelle,
Package leaflet: Information for the user
 Package leaflet: Information for the user Puregon 150 IU/0.18 ml solution for injection Puregon 300 IU/0.36 ml solution for injection Puregon 600 IU/0.72 ml solution for injection Puregon 900 IU/1.08 ml
Package leaflet: Information for the user Puregon 150 IU/0.18 ml solution for injection Puregon 300 IU/0.36 ml solution for injection Puregon 600 IU/0.72 ml solution for injection Puregon 900 IU/1.08 ml
Trial record 1 of 1 for: Previous Study Return to List Next Study
 1 von 6 14.01.2014 09:11 A service of the U.S. National Institutes of Health Trial record 1 of 1 for: 2012-001834-33 Previous Study Return to List Next Study Study of Cabozantinib (XL184) Versus Prednisone
1 von 6 14.01.2014 09:11 A service of the U.S. National Institutes of Health Trial record 1 of 1 for: 2012-001834-33 Previous Study Return to List Next Study Study of Cabozantinib (XL184) Versus Prednisone
WOMEN & INFANTS HOSPITAL Providence, RI CONSENT FOR IN VITRO FERTILIZATION USING A GESTATIONAL CARRIER (PATIENT/INTENDED PARENTS) 1.
 *40675* 40675 MR-838 (9-2017) WOMEN & INFANTS HOSPITAL Providence, RI 02905 CONSENT FOR IN VITRO FERTILIZATION USING A GESTATIONAL CARRIER (PATIENT/INTENDED PARENTS) 1. I, and (Print Patient s name) (Print
*40675* 40675 MR-838 (9-2017) WOMEN & INFANTS HOSPITAL Providence, RI 02905 CONSENT FOR IN VITRO FERTILIZATION USING A GESTATIONAL CARRIER (PATIENT/INTENDED PARENTS) 1. I, and (Print Patient s name) (Print
Approach to ovulation induction and superovulation in women with a history of infertility. Anatte E. Karmon, MD
 Approach to ovulation induction and superovulation in women with a history of infertility Anatte E. Karmon, MD Disclosures- Anatte Karmon, MD No financial relationships to disclose 2 Objectives At the
Approach to ovulation induction and superovulation in women with a history of infertility Anatte E. Karmon, MD Disclosures- Anatte Karmon, MD No financial relationships to disclose 2 Objectives At the
5/5/2010. Infertility FINANCIAL DISCLOSURE. Infertility Definition. Objectives. Normal Human Fertility. Normal Menstrual Cycle
 Infertility FINANCIAL DISCLOSURE I HAVE NO FINANCIAL INTEREST IN ANY OF THE PRODUCTS MENTIONED IN MY PRESENTATION Bryan K. Rone, M.D. University of Kentucky Obstetrics and Gynecology I AM RECEIVING COMPENSATION
Infertility FINANCIAL DISCLOSURE I HAVE NO FINANCIAL INTEREST IN ANY OF THE PRODUCTS MENTIONED IN MY PRESENTATION Bryan K. Rone, M.D. University of Kentucky Obstetrics and Gynecology I AM RECEIVING COMPENSATION
Clinical Study Synopsis
 Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
PRODUCT INFORMATION. Pharmacotherapeutic group: anti-gonadotrophin releasing hormone; ATC code: H01CC01.
 Orgalutran PRODUCT INFORMATION (i) NAME OF THE MEDICINE Orgalutran 250 mg/0.5 ml solution for injection Orgalutran contains the synthetic decapeptide ganirelix (INN) as its acetate salt, with high antagonistic
Orgalutran PRODUCT INFORMATION (i) NAME OF THE MEDICINE Orgalutran 250 mg/0.5 ml solution for injection Orgalutran contains the synthetic decapeptide ganirelix (INN) as its acetate salt, with high antagonistic
Information Booklet. Exploring the causes of infertility and treatment options.
 Information Booklet Exploring the causes of infertility and treatment options www.ptafertility.co.za info@ptafertility.co.za +27 12 998 8854 Faith is taking the first step even if you don t see the whole
Information Booklet Exploring the causes of infertility and treatment options www.ptafertility.co.za info@ptafertility.co.za +27 12 998 8854 Faith is taking the first step even if you don t see the whole
NEW ZEALAND DATA SHEET. LUVERIS 75 IU Powder for Injection. Lutropin alfa (rch)
 NEW ZEALAND DATA SHEET LUVERIS 75 IU Powder for Injection Lutropin alfa (rch) NAME OF THE MEDICINE LUVERIS contains lutropin alfa (rch). # Recombinant-hLH (r-hlh) is a human gonadotropin hormone, composed
NEW ZEALAND DATA SHEET LUVERIS 75 IU Powder for Injection Lutropin alfa (rch) NAME OF THE MEDICINE LUVERIS contains lutropin alfa (rch). # Recombinant-hLH (r-hlh) is a human gonadotropin hormone, composed
Virginia Center for Reproductive Medicine
 Virginia Center for Reproductive Medicine New Patient Questionnaire Date: Patient Name: Date of Birth: / / Age: Social Security #: Address: Phone: (H) ( ) (W) ( ) Cell Phone: ( ) Pharmacy: ( ) Partner
Virginia Center for Reproductive Medicine New Patient Questionnaire Date: Patient Name: Date of Birth: / / Age: Social Security #: Address: Phone: (H) ( ) (W) ( ) Cell Phone: ( ) Pharmacy: ( ) Partner
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 This medicinal product is subject to additional monitoring. This will allow quick identification of new safety information. Healthcare professionals are asked
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 This medicinal product is subject to additional monitoring. This will allow quick identification of new safety information. Healthcare professionals are asked
UW MEDICINE PATIENT EDUCATION. In Vitro Fertilization How to prepare and what to expect DRAFT
 UW MEDICINE PATIENT EDUCATION In Vitro Fertilization How to prepare and what to expect This handout tells how to prepare for and what to expect when you go through a cycle of in vitro fertilization. It
UW MEDICINE PATIENT EDUCATION In Vitro Fertilization How to prepare and what to expect This handout tells how to prepare for and what to expect when you go through a cycle of in vitro fertilization. It
ClinicalTrials.gov "Basic Results" Data Element Definitions (DRAFT)
 ClinicalTrials.gov "Basic Results" Data Element Definitions (DRAFT) January 9, 2009 * Required by ClinicalTrials.gov [*] Conditionally required by ClinicalTrials.gov (FDAAA) May be required to comply with
ClinicalTrials.gov "Basic Results" Data Element Definitions (DRAFT) January 9, 2009 * Required by ClinicalTrials.gov [*] Conditionally required by ClinicalTrials.gov (FDAAA) May be required to comply with
Patient Information: Patient Name: Date of Birth:
 Weill Cornell Medicine Ronald O. Perelman and Claudia Cohen Center for Reproductive Medicine Informed Consent for Oocyte (Egg) Cryopreservation Patient Information: Part 1: I have requested to be treated
Weill Cornell Medicine Ronald O. Perelman and Claudia Cohen Center for Reproductive Medicine Informed Consent for Oocyte (Egg) Cryopreservation Patient Information: Part 1: I have requested to be treated
Infertility. Thomas Lloyd and Samera Dean
 Infertility Thomas Lloyd and Samera Dean Infertility Definition Causes Referral criteria Assisted reproductive techniques Complications Ethics What is infertility? Woman Reproductive age Has not conceived
Infertility Thomas Lloyd and Samera Dean Infertility Definition Causes Referral criteria Assisted reproductive techniques Complications Ethics What is infertility? Woman Reproductive age Has not conceived
NAME OF THE MEDICINE. MENOPUR (human menopausal gonadotrophin), powder and solvent for solution for injection
 NAME OF THE MEDICINE MENOPUR (human menopausal gonadotrophin), powder and solvent for solution for injection MENOPUR 600 IU (600 IU/mL after reconstitution): Each vial with powder contains highly purified
NAME OF THE MEDICINE MENOPUR (human menopausal gonadotrophin), powder and solvent for solution for injection MENOPUR 600 IU (600 IU/mL after reconstitution): Each vial with powder contains highly purified
Understanding Infertility, Evaluations, and Treatment Options
 Understanding Infertility, Evaluations, and Treatment Options Arlene J. Morales, M.D., F.A.C.O.G. Fertility Specialists Medical Group, Inc. What We Will Cover Introduction What is infertility? Briefly
Understanding Infertility, Evaluations, and Treatment Options Arlene J. Morales, M.D., F.A.C.O.G. Fertility Specialists Medical Group, Inc. What We Will Cover Introduction What is infertility? Briefly
Bemfola. What is in this leaflet. What is Bemfola used for. Before you are given. Bemfola
 Recombinant human follicle stimulating hormone (follitropin alfa [rch]) Consumer Medicine Information What is in this leaflet This leaflet answers some common questions about It does not contain all the
Recombinant human follicle stimulating hormone (follitropin alfa [rch]) Consumer Medicine Information What is in this leaflet This leaflet answers some common questions about It does not contain all the
Sponsor Novartis. Generic Drug Name Vildagliptin/Metformin. Therapeutic Area of Trial Type 2 diabetes. Approved Indication Type 2 diabetes
 Clinical Trial Results Database Page 1 Sponsor Novartis Generic Drug Name Vildagliptin/Metformin Therapeutic Area of Trial Type 2 diabetes Approved Indication Type 2 diabetes Study Number CLMF237A2309
Clinical Trial Results Database Page 1 Sponsor Novartis Generic Drug Name Vildagliptin/Metformin Therapeutic Area of Trial Type 2 diabetes Approved Indication Type 2 diabetes Study Number CLMF237A2309
Core Safety Profile Triptorelin 1-month/3-month formulation ATC-code: L02A E 04 March 2010
 Core Safety Profile Triptorelin 1-month/3-month formulation ATC-code: L02A E 04 March 2010 4.3 Contraindications Hypersensitivity to GnRH, its analogues or any other component of the medicinal product
Core Safety Profile Triptorelin 1-month/3-month formulation ATC-code: L02A E 04 March 2010 4.3 Contraindications Hypersensitivity to GnRH, its analogues or any other component of the medicinal product
WOMEN & INFANTS HOSPITAL Providence, RI CONSENT FOR IVF WITH EMBRYO TRANSFER
 *40639* 40639 WOMEN & INFANTS HOSPITAL Providence, RI 02905 CONSENT FOR IVF WITH EMBRYO TRANSFER I have requested treatment by the physicians and (Print Patient s name) staff of the Women & Infants Fertility
*40639* 40639 WOMEN & INFANTS HOSPITAL Providence, RI 02905 CONSENT FOR IVF WITH EMBRYO TRANSFER I have requested treatment by the physicians and (Print Patient s name) staff of the Women & Infants Fertility
IMPORTANT: PLEASE READ
 IMPORTANT: PLEASE READ PART III: CONSUMER INFORMATION PUREGON (follitropin beta) This leaflet is part III of a three-part Product Monograph published when PUREGON was approved for sale in Canada and is
IMPORTANT: PLEASE READ PART III: CONSUMER INFORMATION PUREGON (follitropin beta) This leaflet is part III of a three-part Product Monograph published when PUREGON was approved for sale in Canada and is
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Pergoveris 150 IU/75 IU powder and solvent for solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION One vial
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Pergoveris 150 IU/75 IU powder and solvent for solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION One vial
