Serono Inc., Rockland, Massachusetts, and Serono International, Geneva, Switzerland
|
|
|
- Elvin McBride
- 6 years ago
- Views:
Transcription
1 FERTILITY AND STERILITY VOL. 82, NO. 6, DECEMBER 2004 Copyright 2004 American Society for Reproductive Medicine Published by Elsevier Inc. Printed on acid-free paper in U.S.A. A phase I study of the pharmacokinetics, pharmacodynamics, and safety of singleand multiple-dose anastrozole in healthy, premenopausal female volunteers Donald R. Tredway, M.D., a Mauro Buraglio, M.D., b George Hemsey, M.B.A., a and Geoff Denton, B.S. b Serono Inc., Rockland, Massachusetts, and Serono International, Geneva, Switzerland Objective: To evaluate the pharmacokinetic, pharmacodynamic, and safety profiles of the aromatase inhibitor anastrozole in healthy, premenopausal women. Design: Phase I, single-center study. Setting: Infertility clinic. Patient(s): Twenty-six women with regular ovulatory cycles: 20 received either a single dose of 5 mg, 10 mg, 15 mg, or 20 mg anastrozole, or remained untreated; 6 received five daily doses of 10 mg or 15 mg anastrozole. Intervention(s): Anastrozole was administered on cycle day 2 for the single-dose groups and on days 2 6 for the multiple-dose groups. Ultrasound follicular development and endometrial biopsies were performed. Safety was determined from adverse event reports and laboratory parameters. Main Outcome Measure(s): Pharmacokinetics, pharmacodynamics, and safety. Result(s): The pharmacokinetics of anastrozole were linear, predictable, and consistent with previously published data in healthy volunteers. In the single-dose groups, E 2 levels reached their nadir 3 6 hours after administration, decreasing by an average of 39% from baseline. Follicle-stimulating hormone levels rose by 13%, 52%, 49%, and 75% in the 5-mg, 10-mg, 15-mg, and 20-mg groups, respectively, at approximately 24 hours after dosing. Most subjects recruited just one mature follicle, with no apparent effect on endometrial maturation. No safety concerns were noted. Conclusion(s): Anastrozole was well tolerated and suppressed E 2 levels, with a resultant increase in FSH. (Fertil Steril 2004;82: by American Society for Reproductive Medicine.) Key Words: Aromatase inhibitors, anastrozole, ovulation, infertility Received December 18, 2003; revised and accepted April 22, Reprint requests: Donald R. Tredway, M.D., Serono Inc., 1 Technology Place, Rockland, MA (FAX: (781) ; donald.tredway@serono. com). a Reproductive Health and Global Product Development Unit, Serono Inc., Rockland, Massachusetts. b Human Pharmacology Group, Serono International, Geneva, Switzerland /04/$30.00 doi: /j.fertnstert Anovulation is thought to be the cause of up to 20% of human infertility (1). The causes of anovulation fall into three main categories (1, 2), of which World Health Organization classification group II (hypothalamic pituitary failure) is the largest. First-line fertility treatment in World Health Organization group II anovulatory women typically involves an antiestrogen agent, such as clomiphene citrate (CC). This is because increasing estrogen (E) levels during the follicular phase of the menstrual cycle negatively regulates the production of FSH. Thus, use of an E antagonist should result in increased levels of FSH. Although ovulation is restored in approximately 72% of CC-treated patients, only approximately 35% of CC treatments result in pregnancy (3). This difference between ovulation and pregnancy rates is thought to be due to antiestrogen side effects of CC, including reduced endometrial development (believed to be due to depletion of endometrial E receptors [4]) and vaginal dryness (due to changes in cervical mucus). Furthermore, CC has a long half-life (several weeks [5]) and is associated with an increased incidence of ovarian cysts and multiple pregnancies (6). Clomiphene citrate might also decrease uterine blood flow during the early luteal and peri-implantation stages (5). Recently, inhibitors of aromatase, the key enzyme responsible for the production of E, have been used to stimulate ovarian follicles in 1587
2 CC-resistant patients (4). This novel approach seeks to mimic the effects of CC by releasing the hypothalamic pituitary axis from the negative feedback effects of E, without the adverse effects of E receptor depletion and impaired endometrial development. Third-generation nonsteroidal aromatase inhibitors, such as anastrozole (Arimidex; AstraZeneca, Waltham, MA) and letrozole (Femara; Novartis, East Hanover, NJ), have a short circulating half-life (approximately 40 hours), are well tolerated, relatively inexpensive, and can be administered orally (5). Anastrozole at a dose of 1 mg/day inhibits E levels by 97% but has no effect on E receptors or antiestrogenic side effects (5). Studies in animals have provided several lines of evidence to support the use of aromatase inhibitors for ovulation induction in anovulatory women. Primates treated with an aromatase inhibitor have been found to have decreased levels of serum E and increased levels of serum androstenedione, with no effect on either the number of follicles developed or their size (7, 8). Furthermore, treatment during the follicular phase resulted in the development of multiple mature follicles, which could be induced to ovulate with hcg (8). On the basis of these investigations, Shetty et al. (8) proposed that letrozole used clinically in the treatment of breast cancer could be used to obtain multiple follicles for IVF. In addition, studies in mice have shown that anastrozole does not damage chromosome spindle formation and, once fertilized, preimplantation embryo development was not different from the control (9). Also, animal toxicology studies indicate that anastrozole, in contrast to letrozole and CC, is nonteratogenic (10). Preliminary clinical studies of aromatase inhibitors for ovulation induction suggest that they might offer an effective and well-tolerated alternative to CC (4, 11), as well as a superior uterine environment for implantation over CC (12). In addition, local accumulation of androgens in the ovary is believed to increase follicular sensitivity to FSH (4). Anastrozole, which is a potent and highly selective nonsteroidal aromatase inhibitor, is currently used at a dose of 1 mg/day in the treatment of advanced breast cancer in postmenopausal women. This study was performed to evaluate the safety, pharmacokinetics, and pharmacodynamics of higher-dose anastrozole when administered in single- and multiple-dose regimens to healthy, premenopausal female volunteers. MATERIALS AND METHODS This was a phase I, single-center study of the effects of single and multiple doses of anastrozole in premenopausal women, conducted between December 12, 2001 and July 8, The trial was open-label, with the exception of the ultrasound operatives. This study was approved by the institutional review board at the LCG Laboratories Clinics Group Bioscience, Bourn Hall Clinic at Bourn, Cambridge, United Kingdom. The study was supported by Serono Inc. (Rockland, MA), and the authors are employees of Serono. The study sample size was not based on statistical considerations but was considered adequate to allow estimation of the pharmacokinetic/pharmacodynamic profile and confirmation of the published profile for anastrozole in postmenopausal women (13, 14). All subjects were healthy, premenopausal women (aged years, inclusive) with regular ovulatory cycles (25 35 days), a body mass index 30 kg/m 2, both ovaries present, normal vital signs, normal cervical cytology, and who were to use barrier contraception during the study period. Exclusion criteria included clinically significant systemic disease; known infection with HIV or hepatitis B or C viruses; a positive result for recreational drug use (including alcohol); clinically significant abnormal laboratory results; and any medical condition that might interfere with the absorption, distribution, metabolism, or excretion of the study drug. In addition, the use of androgens, danazol, progesterone (P), a gonadotropin-releasing hormone agonist, or oral contraceptives during or within 3 months of study entry; intrauterine contraceptive device; and parenteral depot contraception in the previous 6 months were criteria for exclusion. Any volunteers with abnormal, undiagnosed gynecologic bleeding and known current substance abuse, including an average use of 5 cigarettes per day, were denied study entry. Anastrozole (Arimidex) tablets for oral administration were used in this study. Each tablet contained 1 mg anastrozole, a nonsteroidal aromatase inhibitor, along with excipients. Oral treatment administration occurred on cycle day 2 for the single-dose groups and on cycle days 2 6 for the multiple-dose groups. All doses were given in the morning. Pharmacokinetic parameters were assessed by serial measurements of anastrozole plasma concentrations before dosing and at 0.5, 1, 1.5, 2, 3, 4, 6, 8, 12, 16, 24, and 48 hours after single, oral dosing. In the multiple-dose group, measurements were taken before dosing and at uniform daily intervals from 1 day after the initial daily dose until 2 days after the last dose. Anastrozole plasma concentration vs. time curves were produced to estimate the area under the curve AUC 0 last, AUC 0, maximum concentration (C max ), time of C max (t max ), and half-life (t 1/2 ). Pharmacodynamics were assessed from serial measurements of androstenedione, E 2, FSH, LH, and T before the study, before dosing, and at 3, 6, and 12 hours after dosing on days 3, 4, 6, 8, and 10 of the cycle, and then daily until ovulation was confirmed in the single-dose groups. In the multiple-dose groups, measurements were taken before the study, before dosing, daily from day 3 to day 8 of the cycle, and then daily from day 10 until ovulation. Progesterone and 17 -hydroxyprogesterone levels were monitored daily from 1588 Treadway et al. Anastrozole in premenopausal women Vol. 82, No. 6, December 2004
3 FIGURE 1 Anastrozole mean plasma concentration vs. time plot in healthy premenopausal women after a single oral dose (n 4 per group). (A) Normal scale; (B) semilog scale. Error bars have been omitted for clarity. day 10 until ovulation and at the midluteal stage. Ovulation was demonstrated by a midluteal (cycle days 18 35, depending on the cycle length) P level of 4 ng/ml. Ultrasound examination of the ovaries and uterus was performed before the study, before dosing, on days 6, 8, and 10 of the cycle, and then daily until ovulation, at the midluteal stage, and after the study in both groups. In addition, the multiple-dose groups were also examined on day 4. Response to anastrozole treatment was monitored by evaluating both ovaries, including the length in three planes, the number of follicles 11 mm present on the largest section through the ovary, the size of any follicles 11 mm, the number of mature follicles ( 14 mm), and the size of any ovarian cysts during the treatment cycle or subsequent cycle. The size of the uterus (length, height, and width), its appearance, and endometrial thickness (from endometrial biopsies) were also recorded. Ovulation, as demonstrated by a midluteal (days 21 23) P level of 4 ng/ml, was recorded as part of the screening process. Safety was determined through the monitoring of adverse events, vital signs, and routine blood analyses, as previously stated. Safety assessments included incidence and severity of adverse events and ovarian hyperstimulation syndrome related symptoms; tolerance of anastrozole administration; clinically significant changes in routine laboratory parameters; and changes in vital signs. Endometrial biopsies were performed during the luteal phase for evaluation of endometrial maturation and synchrony and the incidence and severity of ovarian cysts in subsequent menstrual cycles. RESULTS A total of 39 subjects were booked for prestudy screening. Of these, 26 were enrolled into the clinical phase. The distribution of ages, weights, and heights was similar between dose groups. The first 20 subjects were randomized to receive a single dose of 5 mg, 10 mg, 15 mg, or 20 mg anastrozole, or kept as untreated controls (4 in each group). Subjects and were assigned to receive five daily doses of 10 mg and 15 mg anastrozole, respectively. One subject withdrew from the study on the day of dosing but was monitored for safety. The prestudy screening values for the volunteers were obtained on days 2 4 of the previous menstrual cycle. These baseline values (mean SD) were as follows: FSH: IU/L; LH: IU/L; E 2 : pmol/l; P: nmol/l; androstendione: nmol/l; T: ng/dl. The pharmacokinetic profile of anastrozole was linear, predictable, and consistent with previously published data on healthy male and postmenopausal female volunteers (13), with relatively low intersubject variability. After single oral doses of 5 20 mg, anastrozole was well and rapidly absorbed, with C max achieved within 2 hours (Fig. 1). The sampling schedule of this study did not allow a complete assessment of t 1/2 and AUC 0. However, the interim parameters, AUC 0 last and C max, confirm the pharmacokinetic linearity for doses up to 20 mg. Furthermore, C min fluctuations after repeat doses in a volunteer subgroup (10 mg and 15 mg u.i.d. [once daily]) were consistent with the reported half-life of hours (Fig. 2) and not suggestive of unexpected accumulation. Suppression of E 2 levels was rapid in all anastrozoletreated patients, although there was a high level of interindividual variability. For the single-dose group, E 2 levels reached their nadir approximately 3 6 hours after administration and decreased by an average of 39% from baseline. Mean reductions of 21%, 46%, 55%, and 37% were seen in FERTILITY & STERILITY 1589
4 FIGURE 2 Dose-normalized C min plasma concentrations (red circles) vs. time profiles for anastrozole after multiple-dose (u.i.d) administration of 10 mg and 15 mg in healthy female subjects. (A) Normal scale; (B) semilog scale. Mean standard deviation (n 3 per group). the 5-mg, 10-mg, 15-mg, and 20-mg-dose groups, respectively (Fig. 3). Levels for the control group were essentially unchanged. Estradiol typically returned to pretreatment levels within 24 hours. The multiple-dose groups saw similar rapid declines, and the levels typically returned to baseline by day 2 and day 3 of treatment. Follicle-stimulating hormone levels also varied between subjects but rose in the anastrozole-treated patients, peaking approximately 24 hours after treatment (Fig. 4). At 24 hours, FSH increased from baseline in an apparent dose-dependent relationship by 13%, 52%, 49%, and 75% in the 5-mg, 10-mg, 15-mg, and 20-mg-dose groups, respectively, and returned to FIGURE 3 Mean serum levels of E 2 after single oral doses of anastrozole, 5 20 mg. CD cycle day Treadway et al. Anastrozole in premenopausal women Vol. 82, No. 6, December 2004
5 FIGURE 4 Mean serum levels of FSH after single oral doses of anastrozole, 5 20 mg. CD cycle day. baseline within 3 4 days. Levels were essentially unchanged from baseline in the control group. In the multiple-dose treatment groups (10 mg and 15 mg), FSH levels increased from baseline by an average of 39% from before dosing to day 4 (after first dosing), as compared with 9% in the control group. Levels returned to baseline by the last day of treatment (Fig. 5). No treatment-related effects were seen on the levels of LH, androstenedione, P, 17 -hydroxyprogesterone, or T. In each group, the number of mature follicles recruited was low, with most subjects recruiting a single follicle. It is interesting to note that the group with the highest mean number of follicles was the group with highest exposure to the study drug (15 mg/day for 5 days). However, individual data show this difference to be due largely to the fact that one patient in this group developed three mature follicles. FIGURE 5 Mean serum levels of FSH after multiple oral doses of anastrozole, 10 mg and 15 mg. CD cycle day. FERTILITY & STERILITY 1591
6 TABLE 1 Endometrial biopsy assessments. Endometrial biopsy results In phase (n) Out of phase (n) Out of phase (%) Control 2/4 2/4 50 Anastrozole Single dose 10/15 5/15 33 Multiple dose 5/6 1/6 17 Total 15/21 6/21 29 Endometrial maturation in the midluteal phase was not different among the groups. Histologic evaluation of the endometrium revealed that all patients (controls and treatment groups) ovulated, as verified by secretory endometrium. Compared with the control group, there was no apparent effect on maturation of the endometrium during the luteal phase for all single and multiple doses of anastrozole tested. Biopsies were dated with regard to day of luteal phase. Table 1 summarizes which biopsies were in phase with the actual date of the cycle. In the control group, 2 of 4 biopsies were considered not in phase. In the single-dose groups, 5 of 15 biopsies were not in phase (2 in the 5-mg group, 2 in the 15-mg group, and 1 in the 20-mg group). Among the multiple-dose groups, 1 of 6 were not in phase (10-mg group). In total in the anastrozole treatment groups, 6 of 21 were not in phase. With regard to general histologic findings, no subjects had any evidence of endometritis, hyperplasia, or neoplasia. One subject in the control group had fragments of benign endometrial polyp on biopsy. A total of 96 adverse events were recorded, of which 60 (63%) were considered to be possibly (58) or probably (2, hot flushes and mood swings) related to the study drug. The majority of adverse events (71%) were mild; the rest were moderate. Of the 28 moderate adverse events, 18 were considered to be possibly related to the study drug. There were no severe, serious, or other significant adverse events. One subject withdrew from the study while experiencing adverse events, and one had an adverse event that was unresolved by the time of study completion. The most frequently reported adverse events were headache (24 episodes in 13 subjects), disturbance of menstrual function (8 occasions by 6 subjects), and various gastrointestinal symptoms. There was no evidence of a dose relationship, and these adverse events were also reported in control subjects. Most events were mild and transient in nature. Psychological events (depression, excessive tiredness, mood swings, emotional episodes, and poor concentration) were reported only in anastrozole-treated subjects (7 events in 5 subjects), although no dose relationship was evident. One subject withdrew while experiencing depression and excessive tiredness. There were no clinically significant changes in vital signs or hematologic or biochemical markers. On ultrasonography, residual ovarian cysts 20 mm in the subsequent menstrual cycle were noted in 3 of the 22 subjects treated. Two subjects (one who had received a 5-mg single dose and the other a 15-mg single dose) were noted to have ovarian cysts at the follow-up examination. However, these were resolved at the repeat ultrasound and did not require any further follow-up. One additional subject, who had received a 10-mg multiple dose, was found to have a cystic area near the left ovary. This was first noted on study day 6 and persisted throughout the remainder of the study. Although this was considered as possibly related to treatment, the subject had a history of unexplained infertility, which could, potentially, have been associated with such a lesion. She was referred to her family practitioner for further investigation. No clinically significant abnormalities or changes were noted in other subjects. DISCUSSION The pharmacokinetics of anastrozole in this study were linear, predictable, and consistent with previously published data from healthy volunteers (13, 14). In the single-dose groups, anastrozole was well and rapidly absorbed, with C max occurring within 2 hours. In the multiple-dose groups, the C min vs. time profiles were consistent with linear kinetics and with a compound with a plasma t 1/2 30 hours. The experiment duration was insufficient to definitively evaluate t 1/2 and AUC 0 ; however, the interim parameters AUC 0 last and C max, and C min and T max were all considered sufficient to meet the study objective and to confirm the published profile for anastrozole in postmenopausal women (13, 14). The probable pharmacodynamic mechanism of action is evident upon consideration of the E decrease, with the resulting response of an increase in FSH. Compared with the control group, a rapid decline in E levels occurred at 3 6 hours and persisted for approximately 24 hours with the single-dose regimen and hours in the multiple-dose regimen. This E decline resulted in an FSH increase at 24 hours, persisting for 3 4 days in the single-dose regimen and 4 5 days in the multiple-dose regimen. Surprisingly, no change was noted in LH levels, but this could be owing to the low number of volunteers involved in this study. As the development of this product continues, it will be interesting to compare this E/gonadotropin profile of the normal volunteer to that noted in anovulatory patients. With regard to follicular development, the concern at the beginning of this study was the safety issue of multifollicular development, because it is a common occurrence in female patients with aromatase deficiency (15). With both the single- and multiple-dose regimens, monofollicular develop Treadway et al. Anastrozole in premenopausal women Vol. 82, No. 6, December 2004
7 ment occurred in the healthy volunteers of this study. However, in the interpretation of the follicle data, it should be noted that the subjects recruited into this study differ from the intended target population, inasmuch as the subjects in this study were known to have normal menstrual function before enrollment. The effect of the study drug on endocrine function and follicular development in subjects with anovulatory cycles might differ from that demonstrated in this study and would be worthy of further investigation. Residual ovarian cysts 20 mm in the subsequent menstrual cycle were noted in 3 of the 22 subjects treated, but 1 probably represents a parovarian cyst. Depending on the determination of this paraovarian cyst, the incidence of residual cysts was 9% 13.6%. This rate is less than that reported recently for ultrasound determination of ovarian cysts in subsequent cycles after CC treatment (21.5%) (16) and of clinical ovarian enlargement (13.6%) noted in the original Merrill registration file (17). As noted previously, the antiestrogen effects of CC result in reduced endometrial development. This study noted no delay in endometrial maturation in comparisons of both the single- and the multiple-dose groups with the control group. It is important to note that the endometrial sampling was not done in two consecutive menstrual cycles, which is the accepted practice for the diagnosis of luteal-phase defect, as determined by endometrial biopsy (18). Taking this reservation into account, the potential benefit of anastrozole with regard to the endometrial delay reported with CC is substantiated in this study (5). Anastrozole was well tolerated during this study. The pattern of adverse events observed was broadly similar to that expected on the basis of the known safety profile of the drug. Any adverse events were generally mild, transient, and well tolerated by subjects. There were no serious or other significant adverse events and no clinically significant findings in any of the other safety parameters. Anastrozole was thus considered to be safe in this study when administered in single doses of up to 20 mg and also in repeated doses of up to 15 mg/day for 5 days. In summary, anastrozole was well tolerated at the doses tested. The results demonstrated the expected effects of aromatase inhibitors, which are suppression of endogenous E 2 levels, with a resultant increase in FSH levels. The duration of action was consistent with the half-life of the drug and does not seem to affect overall menstrual function. Clinical effects included monofollicular development, minimal residual cyst formation in the subsequent cycle, and no adverse effects on endometrial maturation. Although the data are promising, the clinical effect on anovulatory cycles cannot be assessed from these limited data. Acknowledgments: The authors thank the Bourn Hall research staff, Bourn, United Kingdom. References 1. Speroff L, Glass RH, Kase NG. Clinical gynecologic endocrinology and infertility. 3rd ed. Baltimore: William and Wilkins, Insler V, Lunenfeld B. Infertility: male and female. New York: Churchill Livingstone, Isaacs JD. Ovulation induction: clomiphene citrate and menotropins. In: Cowan BD, Seifer DB, Cowan R, eds. Clinical reproductive medicine. Philadelphia: Lippincott-Raven, 1997: Mitwally MFM, Casper RF. Use of an aromatase inhibitor for induction of ovulation in patients with an inadequate response to clomiphene citrate. Fertil Steril 2001;75: Mitwally MFM, Casper RF. Aromatase inhibitors for the treatment of infertility. Expert Opin Investig Drugs 2003;12: Gysler M, March CM, Mishell DR Jr, Bailey EJ. A decade s experience with an individualized clomiphene treatment regimen including its effect on the postcoital test. Fertil Steril 1982;37: Zelinski-Wooten MB, Hess DL, Baughman WL, Molskness TA, Wolf DP, Stouffer RL. Administration of an aromatase inhibitor during the late follicular phase of gonadotropin-treated cycles in rhesus monkeys: effects on follicle development, oocyte maturation, and subsequent luteal function. J Clin Endocrinol Metab 1993;76: Shetty G, Krishnamurthy H, Krishnamurthy HN, Bhatnagar S, Moudgal RN. Effect of estrogen deprivation on the reproductive physiology of male and female primates. J Steroid Biochem Mol Biol 1997; 61: Hu Y, Cortvrindt R, Smitz J. Effects of aromatase inhibition on in vitro follicle and oocyte development analyzed by early preantral mouse follicle culture. Mol Reprod Dev 2002;61: Physicians desk reference. 57th ed. Montvale, New Jersey: Medical Economics, Fisher SA, Reid RL, Van Vugt DA, Casper RF. A randomized doubleblind comparison of the effects of clomiphene citrate and the aromatase inhibitor letrozole on ovulatory function in normal women. Fertil Steril 2002;78: Sammour A, Biljan MM, Tan SL, Tulandi T. Prospective randomized trial comparing the effects of letrozole (LE) and clomiphene citrate (CC) on follicular development, endometrial thickness and pregnancy rate in patients undergoing super-ovulation prior to intrauterine insemination (IUI) [abstract]. Fertil Steril 2001;76(Suppl 1):S Plourde PV, Dyroff M, Dukes M. Arimdex: a potent and selective fourth-generation aromatase inhibitor. Breast Cancer Res Treat 1994; 30: Boeddinghaus IM, Dowsett M. Comparative clinical pharmacology and pharmacokinetic interactions of aromatase inhibitors. J Steroid Biochem Mol Biol 2001;79: Bulun SE. Aromatase deficiency and estrogen resistance: from molecular genetics to clinic. Semin Reprod Med 2000;18: Frattarelli JL. Incidence of baseline ovarian cysts in clomiphene citrate ovulation induction cycles [abstract]. Fertil Steril 2001;76(Suppl 1): S Clomid (clomiphene citrate tablets USP), Hoechst Marion Roussel. In: Physicians desk reference. 57th ed. Montvale, New Jersey: Medical Economics, 2003: Wentz AC. Luteal phase inadequacy. In: Sciarra JJ, ed. Gynecology and obstetrics, vol 56. Philadelphia: Lippincott-Raven, 1991:1 10. FERTILITY & STERILITY 1593
Original Article. Fauzia HaqNawaz 1*, Saadia Virk 2, Tasleem Qadir 3, Saadia Imam 3, Javed Rizvi 2
 Original Article Comparison of Letrozole and Clomiphene Citrate Efficacy along with Gonadotrophins in Controlled Ovarian Hyperstimulation for Intrauterine Insemination Cycles Fauzia HaqNawaz 1*, Saadia
Original Article Comparison of Letrozole and Clomiphene Citrate Efficacy along with Gonadotrophins in Controlled Ovarian Hyperstimulation for Intrauterine Insemination Cycles Fauzia HaqNawaz 1*, Saadia
Letrozole versus Clomiphene Citrate in Patients with Anovulatory Infertility
 South Asian Letrozole Federation versus of Clomiphene Obstetrics Citrate and Gynecology, in Patients with January-April Anovulatory Infertility 2009;1(1):19-23 ORIGINAL STUDIES Letrozole versus Clomiphene
South Asian Letrozole Federation versus of Clomiphene Obstetrics Citrate and Gynecology, in Patients with January-April Anovulatory Infertility 2009;1(1):19-23 ORIGINAL STUDIES Letrozole versus Clomiphene
Reproductive Health and Pituitary Disease
 Reproductive Health and Pituitary Disease Janet F. McLaren, MD Assistant Professor Division of Reproductive Endocrinology and Infertility Department of Obstetrics and Gynecology jmclaren@uabmc.edu Objectives
Reproductive Health and Pituitary Disease Janet F. McLaren, MD Assistant Professor Division of Reproductive Endocrinology and Infertility Department of Obstetrics and Gynecology jmclaren@uabmc.edu Objectives
Approach to ovulation induction and superovulation in women with a history of infertility. Anatte E. Karmon, MD
 Approach to ovulation induction and superovulation in women with a history of infertility Anatte E. Karmon, MD Disclosures- Anatte Karmon, MD No financial relationships to disclose 2 Objectives At the
Approach to ovulation induction and superovulation in women with a history of infertility Anatte E. Karmon, MD Disclosures- Anatte Karmon, MD No financial relationships to disclose 2 Objectives At the
Common protocols in intra-uterine insemination cycles
 Common protocols in intra-uterine insemination cycles Doç. Dr. Candan İltemir Duvan Turgut Özal Üniversitesi Tıp Fakültesi Kadın Hastalıkları ve Doğum AD Ovulation induction with intra-uterine insemination
Common protocols in intra-uterine insemination cycles Doç. Dr. Candan İltemir Duvan Turgut Özal Üniversitesi Tıp Fakültesi Kadın Hastalıkları ve Doğum AD Ovulation induction with intra-uterine insemination
Comparison of the success rate of letrozole and clomiphene citrate. in women undergoing intrauterine insemination
 Received: 10.2.2006 Accepted: 8.11.2006 Original Article Comparison of the success rate of letrozole and clomiphene citrate in women undergoing intrauterine insemination Robab Davar*, Maryam Asgharnia
Received: 10.2.2006 Accepted: 8.11.2006 Original Article Comparison of the success rate of letrozole and clomiphene citrate in women undergoing intrauterine insemination Robab Davar*, Maryam Asgharnia
Understanding Infertility, Evaluations, and Treatment Options
 Understanding Infertility, Evaluations, and Treatment Options Arlene J. Morales, M.D., F.A.C.O.G. Fertility Specialists Medical Group, Inc. What We Will Cover Introduction What is infertility? Briefly
Understanding Infertility, Evaluations, and Treatment Options Arlene J. Morales, M.D., F.A.C.O.G. Fertility Specialists Medical Group, Inc. What We Will Cover Introduction What is infertility? Briefly
Mohamed F. M. Mitwally, M.D., and Robert F. Casper, M.D.
 FERTILITY AND STERILITY VOL. 75, NO. 2, FEBRUARY 2001 Copyright 2001 American Society for Reproductive Medicine Published by Elsevier Science Inc. Printed on acid-free paper in U.S.A. OVULATION INDUCTION
FERTILITY AND STERILITY VOL. 75, NO. 2, FEBRUARY 2001 Copyright 2001 American Society for Reproductive Medicine Published by Elsevier Science Inc. Printed on acid-free paper in U.S.A. OVULATION INDUCTION
Hormonal Control of Human Reproduction
 Hormonal Control of Human Reproduction Bởi: OpenStaxCollege The human male and female reproductive cycles are controlled by the interaction of hormones from the hypothalamus and anterior pituitary with
Hormonal Control of Human Reproduction Bởi: OpenStaxCollege The human male and female reproductive cycles are controlled by the interaction of hormones from the hypothalamus and anterior pituitary with
Neil Goodman, MD, FACE
 Initial Workup of Infertile Couple: Female Neil Goodman, MD, FACE Professor of Medicine Voluntary Faculty University of Miami Miller School of Medicine Scope of Infertility in the United States Affects
Initial Workup of Infertile Couple: Female Neil Goodman, MD, FACE Professor of Medicine Voluntary Faculty University of Miami Miller School of Medicine Scope of Infertility in the United States Affects
Comparison of tamoxifen and clomiphene citrate for induction of ovulation in cases with thin endometrium
 Original Article Comparison of tamoxifen and clomiphene citrate for induction of ovulation in cases with thin endometrium Department of Obstetrics and Gynecology, Faculty of Medicine, Suez Canal University
Original Article Comparison of tamoxifen and clomiphene citrate for induction of ovulation in cases with thin endometrium Department of Obstetrics and Gynecology, Faculty of Medicine, Suez Canal University
Article Letrozole versus human menopausal gonadotrophin in women undergoing intrauterine insemination
 RBMOnline - Vol 13. No 2. 2006 208-212 Reproductive BioMedicine Online; www.rbmonline.com/article/2334 on web 30 May 2006 Article Letrozole versus human menopausal gonadotrophin in women undergoing intrauterine
RBMOnline - Vol 13. No 2. 2006 208-212 Reproductive BioMedicine Online; www.rbmonline.com/article/2334 on web 30 May 2006 Article Letrozole versus human menopausal gonadotrophin in women undergoing intrauterine
Infertility treatment
 In the name of God Infertility treatment Treatment options The optimal treatment is one that provide an acceptable success rate, has minimal risk and is costeffective. The treatment options are: 1- Ovulation
In the name of God Infertility treatment Treatment options The optimal treatment is one that provide an acceptable success rate, has minimal risk and is costeffective. The treatment options are: 1- Ovulation
Polycystic Ovary Syndrome HEATHER BURKS, MD OU PHYSICIANS REPRODUCTIVE MEDICINE SEPTEMBER 21, 2018
 Polycystic Ovary Syndrome HEATHER BURKS, MD OU PHYSICIANS REPRODUCTIVE MEDICINE SEPTEMBER 21, 2018 Learning Objectives At the conclusion of this lecture, learners should: 1) Know the various diagnostic
Polycystic Ovary Syndrome HEATHER BURKS, MD OU PHYSICIANS REPRODUCTIVE MEDICINE SEPTEMBER 21, 2018 Learning Objectives At the conclusion of this lecture, learners should: 1) Know the various diagnostic
Principles of Ovarian Stimulation
 Principles of Ovarian Stimulation Dr Genia Rozen Gynaecologist and Fertility Specialist Royal Women s Hospital and Melbourne IVF Learning objectives Why ovarian stimulation Recap physiology Ovarian cycle
Principles of Ovarian Stimulation Dr Genia Rozen Gynaecologist and Fertility Specialist Royal Women s Hospital and Melbourne IVF Learning objectives Why ovarian stimulation Recap physiology Ovarian cycle
Infertility. Review and Update Clifford C. Hayslip MD Intrauterine Inseminations
 Infertility Review and Update Clifford C. Hayslip MD Intrauterine Inseminations Beneficial effects of IUI not consistently documented in studies No deleterious effects on fertility 3-4 cycles of IUI should
Infertility Review and Update Clifford C. Hayslip MD Intrauterine Inseminations Beneficial effects of IUI not consistently documented in studies No deleterious effects on fertility 3-4 cycles of IUI should
Fertility Treatment: Do not be Distracted
 Fertility Treatment: Do not be Distracted Fertility Treatment: do not be distracted by worthless recommendation Fertility Treatment: Do not be Distracted When contemplating options for fertility treatment
Fertility Treatment: Do not be Distracted Fertility Treatment: do not be distracted by worthless recommendation Fertility Treatment: Do not be Distracted When contemplating options for fertility treatment
A Tale of Three Hormones: hcg, Progesterone and AMH
 A Tale of Three Hormones: hcg, Progesterone and AMH Download the Ferring AR ipad/iphone app from the Apple Store: http://bit.ly/1okk74m Human Ovarian Steroidogenesis and Gonadotrophin Stimulation Johan
A Tale of Three Hormones: hcg, Progesterone and AMH Download the Ferring AR ipad/iphone app from the Apple Store: http://bit.ly/1okk74m Human Ovarian Steroidogenesis and Gonadotrophin Stimulation Johan
Investigation: The Human Menstrual Cycle Research Question: How do hormones control the menstrual cycle?
 Investigation: The Human Menstrual Cycle Research Question: How do hormones control the menstrual cycle? Introduction: The menstrual cycle (changes within the uterus) is an approximately 28-day cycle that
Investigation: The Human Menstrual Cycle Research Question: How do hormones control the menstrual cycle? Introduction: The menstrual cycle (changes within the uterus) is an approximately 28-day cycle that
CASE 41. What is the pathophysiologic cause of her amenorrhea? Which cells in the ovary secrete estrogen?
 CASE 41 A 19-year-old woman presents to her gynecologist with complaints of not having had a period for 6 months. She reports having normal periods since menarche at age 12. She denies sexual activity,
CASE 41 A 19-year-old woman presents to her gynecologist with complaints of not having had a period for 6 months. She reports having normal periods since menarche at age 12. She denies sexual activity,
Achieving Pregnancy: Obesity and Infertility. Jordan Vaughan, MSN, APN, WHNP-BC Women s Health Nurse Practitioner Nashville Fertility Center
 Achieving Pregnancy: Obesity and Infertility Jordan Vaughan, MSN, APN, WHNP-BC Women s Health Nurse Practitioner Nashville Fertility Center Disclosures Speakers Bureau EMD Serono Board of Directors Nurse
Achieving Pregnancy: Obesity and Infertility Jordan Vaughan, MSN, APN, WHNP-BC Women s Health Nurse Practitioner Nashville Fertility Center Disclosures Speakers Bureau EMD Serono Board of Directors Nurse
Sonographic determination of a possible adverse effect of domiphene citrate on endometrial growth
 Human Reproduction vol.5 no.6 pp.670-674, 1990 Sonographic determination of a possible adverse effect of domiphene citrate on endometrial growth Yael Gonen 1 and Robert F.Casper Division of Reproductive
Human Reproduction vol.5 no.6 pp.670-674, 1990 Sonographic determination of a possible adverse effect of domiphene citrate on endometrial growth Yael Gonen 1 and Robert F.Casper Division of Reproductive
PERIMENOPAUSE. Objectives. Disclosure. The Perimenopause Perimenopause Menopause. Definitions of Menopausal Transition: STRAW.
 PERIMENOPAUSE Patricia J. Sulak, MD Founder, Living WELL Aware LLC Author, Should I Fire My Doctor? Author, Living WELL Aware: Eleven Essential Elements to Health and Happiness Endowed Professor Texas
PERIMENOPAUSE Patricia J. Sulak, MD Founder, Living WELL Aware LLC Author, Should I Fire My Doctor? Author, Living WELL Aware: Eleven Essential Elements to Health and Happiness Endowed Professor Texas
Richard S. Legro, M.D., Penn State College of Medicine, Dept of Ob/Gyn, Hershey, PA, USA
 What have we learned from Multi-Clinical Trials in PCOS: Focus on Infertility Richard S. Legro, M.D., Penn State College of Medicine, Dept of Ob/Gyn, Hershey, PA, USA Disclosures Consultant: Euroscreen,
What have we learned from Multi-Clinical Trials in PCOS: Focus on Infertility Richard S. Legro, M.D., Penn State College of Medicine, Dept of Ob/Gyn, Hershey, PA, USA Disclosures Consultant: Euroscreen,
Prediction of ovulation with the use of oral and vaginal electrical measurements during treatment with clomiphene citrate*
 FERTILITY AND STERILITY Copyright 1987 The American Fertility Society Printed in U.8A. Prediction of ovulation with the use of oral and vaginal electrical measurements during treatment with clomiphene
FERTILITY AND STERILITY Copyright 1987 The American Fertility Society Printed in U.8A. Prediction of ovulation with the use of oral and vaginal electrical measurements during treatment with clomiphene
Journal of American Science 2013;9(12) Mohamed Elkadi, Amr Elhelaly, Ahmed Ibrahim, Shereen Abdelaziz
 Clomiphene Citrate Alone or Followed by Human Chorionic Gonadotropin In Induction of Ovulation. Mohamed Elkadi, Amr Elhelaly, Ahmed Ibrahim, Shereen Abdelaziz Department of Obstetrics and Gynecology Ain
Clomiphene Citrate Alone or Followed by Human Chorionic Gonadotropin In Induction of Ovulation. Mohamed Elkadi, Amr Elhelaly, Ahmed Ibrahim, Shereen Abdelaziz Department of Obstetrics and Gynecology Ain
Special Features. REVIEW: Aromatase Inhibitors for Ovulation Induction. Página 1 de 23. Abbreviations:
 Página 1 de 23 Use of this content is subject to the Terms and Conditions of the MD Consult web site. Journal of Clinical Endocrinology and Metabolism Volume 91 Number 3 March 2006 Copyright 2006 The Endocrine
Página 1 de 23 Use of this content is subject to the Terms and Conditions of the MD Consult web site. Journal of Clinical Endocrinology and Metabolism Volume 91 Number 3 March 2006 Copyright 2006 The Endocrine
Core Safety Profile. Pharmaceutical form(s)/strength: Lyophilised powder for injection / 75 IU. Date of FAR:
 Core Safety Profile Active substance: Urofollitropin Pharmaceutical form(s)/strength: Lyophilised powder for injection / 75 IU P - RMS: UK/H/PSUR/0059/001 Date of FAR: 04.12.2009 4.2 Posology and method
Core Safety Profile Active substance: Urofollitropin Pharmaceutical form(s)/strength: Lyophilised powder for injection / 75 IU P - RMS: UK/H/PSUR/0059/001 Date of FAR: 04.12.2009 4.2 Posology and method
LUTEAL PHASE SUPPORT. Doç. Dr. Nafiye Yılmaz. Zekai Tahir Burak Kadın Sağlığı Eğitim Araştırma Hastanesi
 LUTEAL PHASE SUPPORT Doç. Dr. Nafiye Yılmaz Zekai Tahir Burak Kadın Sağlığı Eğitim Araştırma Hastanesi TAJEV, 2014 1 ART & success *Live birth rate 2 Optimal luteal phase Etiology of luteal phase deficiency
LUTEAL PHASE SUPPORT Doç. Dr. Nafiye Yılmaz Zekai Tahir Burak Kadın Sağlığı Eğitim Araştırma Hastanesi TAJEV, 2014 1 ART & success *Live birth rate 2 Optimal luteal phase Etiology of luteal phase deficiency
Infertility DR. RAHUL BEVARA
 Infertility DR. RAHUL BEVARA Definitions Infertility is defined as the inability to conceive after one year of unprotected coitus. Affects 10-15% of couples Primary Infertility, that is inability to conceive
Infertility DR. RAHUL BEVARA Definitions Infertility is defined as the inability to conceive after one year of unprotected coitus. Affects 10-15% of couples Primary Infertility, that is inability to conceive
Clinical Study Synopsis
 Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Stage 4 - Ovarian Cancer Symptoms
 WELCOME Stage 4 - Ovarian Cancer Symptoms University of Baghdad College of Nursing Department of Basic Medical Sciences Overview of Anatomy and Physioloy II Second Year Students Asaad Ismail Ahmad,
WELCOME Stage 4 - Ovarian Cancer Symptoms University of Baghdad College of Nursing Department of Basic Medical Sciences Overview of Anatomy and Physioloy II Second Year Students Asaad Ismail Ahmad,
Article Aromatase inhibitors in ovarian stimulation for IVF/ICSI: a pilot study
 RBMOnline - Vol 13. No 2. 2006 166-172 Reproductive BioMedicine Online; www.rbmonline.com/article/2261 on web 19 May 2006 Article Aromatase inhibitors in ovarian stimulation for IVF/ICSI: a pilot study
RBMOnline - Vol 13. No 2. 2006 166-172 Reproductive BioMedicine Online; www.rbmonline.com/article/2261 on web 19 May 2006 Article Aromatase inhibitors in ovarian stimulation for IVF/ICSI: a pilot study
Risk factors for spontaneous abortion in menotropintreated
 FERTILITY AND STERILITY Copyright ~ 1987 The American Fertility Society Vol. 48, No. 4, October 1987 Printed in U.S.A. Risk factors for spontaneous abortion in menotropintreated women Michael Bohrer, M.D.*
FERTILITY AND STERILITY Copyright ~ 1987 The American Fertility Society Vol. 48, No. 4, October 1987 Printed in U.S.A. Risk factors for spontaneous abortion in menotropintreated women Michael Bohrer, M.D.*
Female Reproductive System. Lesson 10
 Female Reproductive System Lesson 10 Learning Goals 1. What are the five hormones involved in the female reproductive system? 2. Understand the four phases of the menstrual cycle. Human Reproductive System
Female Reproductive System Lesson 10 Learning Goals 1. What are the five hormones involved in the female reproductive system? 2. Understand the four phases of the menstrual cycle. Human Reproductive System
ART Drugs. Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.30.02 Subject: ART Drugs Page: 1 of 7 Last Review Date: September 15, 2017 ART Drugs Description Bravelle
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.30.02 Subject: ART Drugs Page: 1 of 7 Last Review Date: September 15, 2017 ART Drugs Description Bravelle
Infertility: failure to conceive within one year of unprotected regular sexual intercourse. Primary secondary
 Subfertility Infertility: failure to conceive within one year of unprotected regular sexual intercourse. Primary secondary Infertility affects about 15 % of couples. age of the female. Other factors that
Subfertility Infertility: failure to conceive within one year of unprotected regular sexual intercourse. Primary secondary Infertility affects about 15 % of couples. age of the female. Other factors that
Infertility: A Generalist s Perspective
 Infertility: A Generalist s Perspective Learning Objectives Fertility and Lifestyle: Patient education Describe the basic infertility workup Basic treatment strategies unexplained Heather Huddleston, MD
Infertility: A Generalist s Perspective Learning Objectives Fertility and Lifestyle: Patient education Describe the basic infertility workup Basic treatment strategies unexplained Heather Huddleston, MD
Do aromatase inhibitors have a place in IVF?
 Do aromatase inhibitors have a place in IVF? Roy Homburg Maccabi Medical Services and Barzilai Medical Centre, Ashkelon, Israel Antalya, September, 2009 Human Follicle Growth Primordial follicle 1 layer
Do aromatase inhibitors have a place in IVF? Roy Homburg Maccabi Medical Services and Barzilai Medical Centre, Ashkelon, Israel Antalya, September, 2009 Human Follicle Growth Primordial follicle 1 layer
Hormonal profile and endometrial morphology in letrozole-controlled ovarian hyperstimulation in ovulatory infertile patients
 OVULATION INDUCTION Hormonal profile and endometrial morphology in letrozole-controlled ovarian hyperstimulation in ovulatory infertile patients Armando Cortínez, M.D., a,b Iara De Carvalho, M.D., a David
OVULATION INDUCTION Hormonal profile and endometrial morphology in letrozole-controlled ovarian hyperstimulation in ovulatory infertile patients Armando Cortínez, M.D., a,b Iara De Carvalho, M.D., a David
Vaginal micronized progesterone versus intramuscular progesterone for luteal support in women undergoing in vitro fertilization embryo transfer
 Vaginal micronized progesterone versus intramuscular progesterone for luteal support in women undergoing in vitro fertilization embryo transfer Mohamed F. Mitwally, M.D., H.C.L.D., a Michael P. Diamond,
Vaginal micronized progesterone versus intramuscular progesterone for luteal support in women undergoing in vitro fertilization embryo transfer Mohamed F. Mitwally, M.D., H.C.L.D., a Michael P. Diamond,
Assisted Reproduction. By Dr. Afraa Mahjoob Al-Naddawi
 Assisted Reproduction By Dr. Afraa Mahjoob Al-Naddawi Learning Objectives: By the end of this lecture, you will be able to: 1) Define assisted reproductive techniques (ART). 2) List indications for various
Assisted Reproduction By Dr. Afraa Mahjoob Al-Naddawi Learning Objectives: By the end of this lecture, you will be able to: 1) Define assisted reproductive techniques (ART). 2) List indications for various
Orgalutran 0.25 mg/0.5 ml solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION
 1 1. NAME OF THE MEDICINAL PRODUCT 0.25 mg/0.5 ml solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each pre-filled syringe contains 0.25 mg of ganirelix (INN) in 0.5 mg aqueous solution.
1 1. NAME OF THE MEDICINAL PRODUCT 0.25 mg/0.5 ml solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each pre-filled syringe contains 0.25 mg of ganirelix (INN) in 0.5 mg aqueous solution.
Module 3. Infertility: Protocols and Patient Management
 Module 3 Infertility: Protocols and Patient Management Ann Scalia, BSN, RN, CNOR Manager Clinical Education Specialists Mary Vietzke, BSN, RN Senior Clinical Educational Specialist Walgreens Faculty Ann
Module 3 Infertility: Protocols and Patient Management Ann Scalia, BSN, RN, CNOR Manager Clinical Education Specialists Mary Vietzke, BSN, RN Senior Clinical Educational Specialist Walgreens Faculty Ann
LOW RESPONDERS. Poor Ovarian Response, Por
 LOW RESPONDERS Poor Ovarian Response, Por Patients with a low number of retrieved oocytes despite adequate ovarian stimulation during fertility treatment. Diagnosis Female About Low responders In patients
LOW RESPONDERS Poor Ovarian Response, Por Patients with a low number of retrieved oocytes despite adequate ovarian stimulation during fertility treatment. Diagnosis Female About Low responders In patients
Gonadotrophin treatment in patients with Polycystic Ovary Syndrome
 Int. J. Adv. Res. Biol. Sci. (218). 5(4): 95-99 International Journal of Advanced Research in Biological Sciences ISSN: 2348-869 www.ijarbs.com DOI: 1.22192/ijarbs Coden: IJARQG(USA) Volume 5, Issue 4-218
Int. J. Adv. Res. Biol. Sci. (218). 5(4): 95-99 International Journal of Advanced Research in Biological Sciences ISSN: 2348-869 www.ijarbs.com DOI: 1.22192/ijarbs Coden: IJARQG(USA) Volume 5, Issue 4-218
Frequency of menses. Duration of menses 3 days to 7 days. Flow/amount of menses Average blood loss with menstruation is 60-80cc.
 Frequency of menses 24 days (0.5%) to 35 days (0.9%) Age 25, 40% are between 25 and 28 days Age 25-35, 60% are between 25 and 28 days Teens and women over 40 s cycles may be longer apart Duration of menses
Frequency of menses 24 days (0.5%) to 35 days (0.9%) Age 25, 40% are between 25 and 28 days Age 25-35, 60% are between 25 and 28 days Teens and women over 40 s cycles may be longer apart Duration of menses
5/5/2010. Infertility FINANCIAL DISCLOSURE. Infertility Definition. Objectives. Normal Human Fertility. Normal Menstrual Cycle
 Infertility FINANCIAL DISCLOSURE I HAVE NO FINANCIAL INTEREST IN ANY OF THE PRODUCTS MENTIONED IN MY PRESENTATION Bryan K. Rone, M.D. University of Kentucky Obstetrics and Gynecology I AM RECEIVING COMPENSATION
Infertility FINANCIAL DISCLOSURE I HAVE NO FINANCIAL INTEREST IN ANY OF THE PRODUCTS MENTIONED IN MY PRESENTATION Bryan K. Rone, M.D. University of Kentucky Obstetrics and Gynecology I AM RECEIVING COMPENSATION
Index. Note: Page numbers of article titles are in boldface type.
 Index Note: Page numbers of article titles are in boldface type. A Abdominal myomectomy in leiomyoma management, 77 Abnormal uterine bleeding (AUB) described, 103 105 normal menstrual bleeding vs., 104
Index Note: Page numbers of article titles are in boldface type. A Abdominal myomectomy in leiomyoma management, 77 Abnormal uterine bleeding (AUB) described, 103 105 normal menstrual bleeding vs., 104
Comparative Evaluation of Sequential Regimes of Gonadotropins with Clomiphene Citrate and Letrozole for Ovulation Induction
 CODEN (USA)-IJPRUR, e-issn: 2348-6465 International Journal of Pharma Research and Health Sciences Available online at www.pharmahealthsciences.net Original Article Comparative Evaluation of Sequential
CODEN (USA)-IJPRUR, e-issn: 2348-6465 International Journal of Pharma Research and Health Sciences Available online at www.pharmahealthsciences.net Original Article Comparative Evaluation of Sequential
Relation between the Number and Size of Follicles in Ovulation Induction and the Rate of Pregnancy
 Relation between the Number and Size of Follicles in Ovulation Induction and the Rate of Pregnancy Aseel Mosa Jabber M.SC.G.O. The department of Obstetrics and Gynecology, Faculty of Medicine Thi-qar university
Relation between the Number and Size of Follicles in Ovulation Induction and the Rate of Pregnancy Aseel Mosa Jabber M.SC.G.O. The department of Obstetrics and Gynecology, Faculty of Medicine Thi-qar university
Reproductive System. Testes. Accessory reproductive organs. gametogenesis hormones. Reproductive tract & Glands
 Reproductive System Testes gametogenesis hormones Accessory reproductive organs Reproductive tract & Glands transport gametes provide nourishment for gametes Hormonal regulation in men Hypothalamus - puberty
Reproductive System Testes gametogenesis hormones Accessory reproductive organs Reproductive tract & Glands transport gametes provide nourishment for gametes Hormonal regulation in men Hypothalamus - puberty
Comparision of Letrozole with Timed Intercourse Versus Clomiphene Citrate with Intrauterine Insemination in Patients with Unexplained Infertility
 The Journal of Reproductive Medicine Comparision of Letrozole with Timed Intercourse Versus Clomiphene Citrate with Intrauterine Insemination in Patients with Unexplained Infertility Jorge Rodriguez-Purata,
The Journal of Reproductive Medicine Comparision of Letrozole with Timed Intercourse Versus Clomiphene Citrate with Intrauterine Insemination in Patients with Unexplained Infertility Jorge Rodriguez-Purata,
Aromatase inhibition improves ovarian response to follicle-stimulating hormone in poor responders
 FERTILITY AND STERILITY VOL. 77, NO. 4, APRIL 2002 Copyright 2002 American Society for Reproductive Medicine Published by Elsevier Science Inc. Printed on acid-free paper in U.S.A. Aromatase inhibition
FERTILITY AND STERILITY VOL. 77, NO. 4, APRIL 2002 Copyright 2002 American Society for Reproductive Medicine Published by Elsevier Science Inc. Printed on acid-free paper in U.S.A. Aromatase inhibition
Article Prediction of pituitary down-regulation by evaluation of endometrial thickness in an IVF programme
 RBMOnline - Vol 8. No 5. 2004 595-599 Reproductive BioMedicine Online; www.rbmonline.com/article/1065 on web 17 March 2004 Article Prediction of pituitary down-regulation by evaluation of endometrial thickness
RBMOnline - Vol 8. No 5. 2004 595-599 Reproductive BioMedicine Online; www.rbmonline.com/article/1065 on web 17 March 2004 Article Prediction of pituitary down-regulation by evaluation of endometrial thickness
Estrogens and progestogens
 Estrogens and progestogens Estradiol and Progesterone hormones produced by the gonads are necessary for: conception embryonic maturation development of primary and secondary sexual characteristics at puberty.
Estrogens and progestogens Estradiol and Progesterone hormones produced by the gonads are necessary for: conception embryonic maturation development of primary and secondary sexual characteristics at puberty.
ART Drugs. Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.08.02 Subject: ART Drugs Page: 1 of 7 Last Review Date: December 3, 2015 ART Drugs Description Bravelle
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.08.02 Subject: ART Drugs Page: 1 of 7 Last Review Date: December 3, 2015 ART Drugs Description Bravelle
The Pharmacology of PCOS
 The Pharmacology of PCOS G. Wright Bates, Jr., M.D. Director Reproductive Endocrinology & Infertility University of Alabama at Birmingham Objectives Review the diagnosis of PCOS Highlight lifestyle modifications
The Pharmacology of PCOS G. Wright Bates, Jr., M.D. Director Reproductive Endocrinology & Infertility University of Alabama at Birmingham Objectives Review the diagnosis of PCOS Highlight lifestyle modifications
Female Reproductive Physiology. Dr Raelia Lew CREI, FRANZCOG, PhD, MMed, MBBS Fertility Specialist, Melbourne IVF
 Female Reproductive Physiology Dr Raelia Lew CREI, FRANZCOG, PhD, MMed, MBBS Fertility Specialist, Melbourne IVF REFERENCE Lew, R, Natural History of ovarian function including assessment of ovarian reserve
Female Reproductive Physiology Dr Raelia Lew CREI, FRANZCOG, PhD, MMed, MBBS Fertility Specialist, Melbourne IVF REFERENCE Lew, R, Natural History of ovarian function including assessment of ovarian reserve
Uterus & Ovary 2015; 2: e904. doi: /uo.904; 2015 by Ahmed M Maged, et al.
 RESEARCH ARTICLE Phytoestrogens as an alternative to estradiol in reversing the antiestrogenic effect of clomid on endometrium in ovulation induction in cases of polycystic ovarian syndrome (PCOS) Ahmed
RESEARCH ARTICLE Phytoestrogens as an alternative to estradiol in reversing the antiestrogenic effect of clomid on endometrium in ovulation induction in cases of polycystic ovarian syndrome (PCOS) Ahmed
In Vitro Fertilization in Clomiphene-Resistant Women with Polycystic Ovary Syndrome
 Original Article Effect of Laparoscopic Ovarian Drilling on Outcomes of In Vitro Fertilization in Clomiphene-Resistant Women with Polycystic Ovary Syndrome Maryam Eftekhar, M.D. 1, Razieh Deghani Firoozabadi,
Original Article Effect of Laparoscopic Ovarian Drilling on Outcomes of In Vitro Fertilization in Clomiphene-Resistant Women with Polycystic Ovary Syndrome Maryam Eftekhar, M.D. 1, Razieh Deghani Firoozabadi,
Biology of fertility control. Higher Human Biology
 Biology of fertility control Higher Human Biology Learning Intention Compare fertile periods in females and males What is infertility? Infertility is the inability of a sexually active, non-contracepting
Biology of fertility control Higher Human Biology Learning Intention Compare fertile periods in females and males What is infertility? Infertility is the inability of a sexually active, non-contracepting
Setting The setting was secondary care. The economic study was carried out in Turkey.
 Letrozole versus human menopausal gonadotrophin in women undergoing intrauterine insemination Baysoy A, Serdaroglu H, Jamal H, Karatekeli E, Ozornek H, Attar E Record Status This is a critical abstract
Letrozole versus human menopausal gonadotrophin in women undergoing intrauterine insemination Baysoy A, Serdaroglu H, Jamal H, Karatekeli E, Ozornek H, Attar E Record Status This is a critical abstract
Aims of this talk. Evaluation & investigation. Basic treatments/options including ovulation induction & Intra uterine Insemination
 Basic treatments/options including ovulation induction & Intra uterine Insemination Karen Woodcock Clinical Nurse Specialist/ Nurse Manager Fertility & Assisted Conception Unit Countess of Chester NHS
Basic treatments/options including ovulation induction & Intra uterine Insemination Karen Woodcock Clinical Nurse Specialist/ Nurse Manager Fertility & Assisted Conception Unit Countess of Chester NHS
Article Effect of cetrorelix dose on premature LH surge during ovarian stimulation
 RBMOnline - Vol 16. No 6. 2008 772-777 Reproductive BioMedicine Online; www.rbmonline.com/article/3181 on web 18 April 2008 Article Effect of cetrorelix dose on premature LH surge during ovarian stimulation
RBMOnline - Vol 16. No 6. 2008 772-777 Reproductive BioMedicine Online; www.rbmonline.com/article/3181 on web 18 April 2008 Article Effect of cetrorelix dose on premature LH surge during ovarian stimulation
Indian Journal of Basic and Applied Medical Research; September 2015: Vol.-4, Issue- 4, P
 Original article: To study post intrauterine insemination conception rate among infertile women with polyp and women with normal uterine endometrium cavity 1Dr. Archana Meena, 2 Dr. Renu Meena, 3 Dr. Kusum
Original article: To study post intrauterine insemination conception rate among infertile women with polyp and women with normal uterine endometrium cavity 1Dr. Archana Meena, 2 Dr. Renu Meena, 3 Dr. Kusum
Which is the Best Protocol of Ovarian Stimulation Prior to Artificial Insemination by Donor
 Journal of Reproduction & Contraception doi: 10.7669j.issn.1001-7844.2014.01.0041 2014 Mar.; 25(1):41-48 E-mail: randc_journal@163.com Which is the Best Protocol of Ovarian Stimulation Prior to Artificial
Journal of Reproduction & Contraception doi: 10.7669j.issn.1001-7844.2014.01.0041 2014 Mar.; 25(1):41-48 E-mail: randc_journal@163.com Which is the Best Protocol of Ovarian Stimulation Prior to Artificial
Progesterone and clinical outcomes
 Synchronization of Slowly Developing Embryos Restores Implantation Success Richard T. Scott, Jr, MD, HCLD Clinical and Scientific Director, Reproductive Medicine Associates of New Jersey Professor and
Synchronization of Slowly Developing Embryos Restores Implantation Success Richard T. Scott, Jr, MD, HCLD Clinical and Scientific Director, Reproductive Medicine Associates of New Jersey Professor and
HCG (human chorionic gonadotropin); Novarel Pregnyl (chorionic gonadotropin); Ovidrel (choriogonadotropin alfa)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.08.09 Subject: HCG Page: 1 of 5 Last Review Date: June 19, 2015 HCG Powder, Novarel, Pregnyl, Ovidrel
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.08.09 Subject: HCG Page: 1 of 5 Last Review Date: June 19, 2015 HCG Powder, Novarel, Pregnyl, Ovidrel
MODULE 1: OVULATION INDUCTION IN NORMAL RESPONDERS, POOR RESPONDERS, AND HYPERRESPONDERS
 MODULE 1: OVULATION INDUCTION IN NORMAL RESPONDERS, POOR RESPONDERS, AND HYPERRESPONDERS Contents OVULATION INDUCTION IN NORMAL RESPONDERS, POOR RESPONDERS, AND HYPERRESPONDERS Ovulation Induction.....................................................................
MODULE 1: OVULATION INDUCTION IN NORMAL RESPONDERS, POOR RESPONDERS, AND HYPERRESPONDERS Contents OVULATION INDUCTION IN NORMAL RESPONDERS, POOR RESPONDERS, AND HYPERRESPONDERS Ovulation Induction.....................................................................
I. ART PROCEDURES. A. In Vitro Fertilization (IVF)
 DFW Fertility Associates ASSISTED REPRODUCTIVE TECHNOLOGY (ART) Welcome to DFW Fertility Associates/ Presbyterian-Harris Methodist Hospital ARTS program. This document provides an overview of treatment
DFW Fertility Associates ASSISTED REPRODUCTIVE TECHNOLOGY (ART) Welcome to DFW Fertility Associates/ Presbyterian-Harris Methodist Hospital ARTS program. This document provides an overview of treatment
Department of Reproduction & Infertility, Mirza Kouchak Khan Women s Hospital, Tehran University of Medical Sciences, Tehran, Iran
 Global Journal of Health Science; Vol. 8, No. 4; 2016 ISSN 1916-9736 E-ISSN 1916-9744 Published by Canadian Center of Science and Education Effects of Letrozole-HMG and Clomiphene-HMG on Incidence of Luteinized
Global Journal of Health Science; Vol. 8, No. 4; 2016 ISSN 1916-9736 E-ISSN 1916-9744 Published by Canadian Center of Science and Education Effects of Letrozole-HMG and Clomiphene-HMG on Incidence of Luteinized
MANAGEMENT OF REFRACTORY ENDOMETRIOSIS
 (339) MANAGEMENT OF REFRACTORY ENDOMETRIOSIS Serdar Bulun, MD JJ Sciarra Professor and Chair Department of Ob/Gyn Northwestern University ENDOMETRIOSIS OCs Teenager: severe dysmenorrhea often starting
(339) MANAGEMENT OF REFRACTORY ENDOMETRIOSIS Serdar Bulun, MD JJ Sciarra Professor and Chair Department of Ob/Gyn Northwestern University ENDOMETRIOSIS OCs Teenager: severe dysmenorrhea often starting
Does triggering ovulation by 5000 IU of uhcg affect ICSI outcome? *
 Middle East Fertility Society Journal Vol. 11, No. 2, 2006 Copyright Middle East Fertility Society Does triggering ovulation by 5000 IU of uhcg affect ICSI outcome? * Amany A.M. Shaltout, M.D. Mohamed
Middle East Fertility Society Journal Vol. 11, No. 2, 2006 Copyright Middle East Fertility Society Does triggering ovulation by 5000 IU of uhcg affect ICSI outcome? * Amany A.M. Shaltout, M.D. Mohamed
1. During the follicular phase of the ovarian cycle, the hypothalamus releases GnRH.
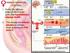 1. During the follicular phase of the ovarian cycle, the hypothalamus releases GnRH. 2. This causes the anterior pituitary to secrete small quantities of FSH and LH. 3. At this time, the follicles in the
1. During the follicular phase of the ovarian cycle, the hypothalamus releases GnRH. 2. This causes the anterior pituitary to secrete small quantities of FSH and LH. 3. At this time, the follicles in the
2017 United HealthCare Services, Inc.
 UnitedHealthcare Pharmacy Clinical Pharmacy Programs Program Number 2017 P 1143-4 Program Prior Authorization/Notification Medication Menopur (menotropins) * P&T Approval Date 8/2014, 5/2015, 5/2016, 5/2017
UnitedHealthcare Pharmacy Clinical Pharmacy Programs Program Number 2017 P 1143-4 Program Prior Authorization/Notification Medication Menopur (menotropins) * P&T Approval Date 8/2014, 5/2015, 5/2016, 5/2017
The Egyptian Journal of Hospital Medicine (July 2018) Vol. 72 (4), Page
 The Egyptian Journal of Hospital Medicine (July 2018) Vol. 72 (4), Page 4382-4388 A Randomized Trial of Combined Letrozole with Metformin Versus Clomiphene Citrate with Metformin in Polycystic Ovary (PCOS)
The Egyptian Journal of Hospital Medicine (July 2018) Vol. 72 (4), Page 4382-4388 A Randomized Trial of Combined Letrozole with Metformin Versus Clomiphene Citrate with Metformin in Polycystic Ovary (PCOS)
IVM in PCOS patients. Introduction (1) Introduction (2) Michael Grynberg René Frydman
 IVM in PCOS patients Michael Grynberg René Frydman Department of Obstetrics and Gynecology A. Beclere Hospital, Clamart, France Maribor, Slovenia, 27-28 February 2009 Introduction (1) IVM could be a major
IVM in PCOS patients Michael Grynberg René Frydman Department of Obstetrics and Gynecology A. Beclere Hospital, Clamart, France Maribor, Slovenia, 27-28 February 2009 Introduction (1) IVM could be a major
Dysfunctional Uterine Bleeding (DUB) OB/GYN Hospital of Fudan University Weiwei Feng, MD,Ph D Tel:
 Dysfunctional Uterine Bleeding (DUB) OB/GYN Hospital of Fudan University Weiwei Feng, MD,Ph D Email: wfeng7347@aliyun.com Tel: 13918551061 2014-8-20 Contents DUB: definition, mechanism of normal menses,
Dysfunctional Uterine Bleeding (DUB) OB/GYN Hospital of Fudan University Weiwei Feng, MD,Ph D Email: wfeng7347@aliyun.com Tel: 13918551061 2014-8-20 Contents DUB: definition, mechanism of normal menses,
Reproductive FSH. Analyte Information
 Reproductive FSH Analyte Information 1 Follicle-stimulating hormone Introduction Follicle-stimulating hormone (FSH, also known as follitropin) is a glycoprotein hormone secreted by the anterior pituitary
Reproductive FSH Analyte Information 1 Follicle-stimulating hormone Introduction Follicle-stimulating hormone (FSH, also known as follitropin) is a glycoprotein hormone secreted by the anterior pituitary
Does previous response to clomifene citrate influence the selection of gonadotropin dosage given in subsequent superovulation treatment cycles?
 J Assist Reprod Genet (26) 23:427 431 DOI 1.17/s1815-6-965-x ASSISTED REPRODUCTION Does previous response to clomifene citrate influence the selection of gonadotropin dosage given in subsequent superovulation
J Assist Reprod Genet (26) 23:427 431 DOI 1.17/s1815-6-965-x ASSISTED REPRODUCTION Does previous response to clomifene citrate influence the selection of gonadotropin dosage given in subsequent superovulation
PCOS and Your Fertility. Jim Toner, MD, PhD Atlanta Center for Reproductive Medicine
 PCOS and Your Fertility Jim Toner, MD, PhD Atlanta Center for Reproductive Medicine PCOS Consequences Androgen excess Acne, oily skin Unwanted hair, male pattern baldness Insulin excess Diabetes, heart
PCOS and Your Fertility Jim Toner, MD, PhD Atlanta Center for Reproductive Medicine PCOS Consequences Androgen excess Acne, oily skin Unwanted hair, male pattern baldness Insulin excess Diabetes, heart
INFERTILITY: DIAGNOSIS, WORKUP AND MANAGEMENT FOR THE COMMUNITY PHYSICIAN
 INFERTILITY: DIAGNOSIS, WORKUP AND MANAGEMENT FOR THE COMMUNITY PHYSICIAN Caitlin Dunne, MD, FRCSC Clinical Assistant Professor Division of Reproductive Endocrinology and Infertility Department of Obstetrics
INFERTILITY: DIAGNOSIS, WORKUP AND MANAGEMENT FOR THE COMMUNITY PHYSICIAN Caitlin Dunne, MD, FRCSC Clinical Assistant Professor Division of Reproductive Endocrinology and Infertility Department of Obstetrics
8605 SW Creekside Place Beaverton, OR Phone: Fax: Samples Collected. Samples Received 06/21/2017
 TEST RESULTS Ordering Provider: Getuwell Clinic Patient Name: Patient Phone Number: 555 555 5555 Gender Female DOB 6/9/1978 (39 yrs) Menses Status Pre-Menopausal 8605 SW Creekside Place Beaverton, OR 97008
TEST RESULTS Ordering Provider: Getuwell Clinic Patient Name: Patient Phone Number: 555 555 5555 Gender Female DOB 6/9/1978 (39 yrs) Menses Status Pre-Menopausal 8605 SW Creekside Place Beaverton, OR 97008
Objectives 06/21/18 STILL A PLACE FOR PILLS DON T IVF EVERYTHING. Clomiphene citrate and Letrozole. Infertility Case Studies. Unexplained Infertility
 STILL A PLACE FOR PILLS DON T IVF EVERYTHING Jeff Roberts M.D. Co-Director, Pacific Centre for Reproductive Medicine Objectives 1 2 3 4 5 Clomiphene citrate and Letrozole Infertility Case Studies Unexplained
STILL A PLACE FOR PILLS DON T IVF EVERYTHING Jeff Roberts M.D. Co-Director, Pacific Centre for Reproductive Medicine Objectives 1 2 3 4 5 Clomiphene citrate and Letrozole Infertility Case Studies Unexplained
Endocrinology of the Female Reproductive Axis
 Endocrinology of the Female Reproductive Axis girlontheriver.com Geralyn Lambert-Messerlian, PhD, FACB Professor Women and Infants Hospital Alpert Medical School at Brown University Women & Infants BROWN
Endocrinology of the Female Reproductive Axis girlontheriver.com Geralyn Lambert-Messerlian, PhD, FACB Professor Women and Infants Hospital Alpert Medical School at Brown University Women & Infants BROWN
Superovulation with human menopausal gonadotropins is associated with endometrial gland-stroma dyssynchrony*
 aes FERTILITY AND STERILITY Vol. 61, No.4, April 1994 Copyright ee) 1994 The American Fertility Society Printed on acid-free paper in U. S. A. r I Superovulation with human menopausal gonadotropins is
aes FERTILITY AND STERILITY Vol. 61, No.4, April 1994 Copyright ee) 1994 The American Fertility Society Printed on acid-free paper in U. S. A. r I Superovulation with human menopausal gonadotropins is
Luteal phase rescue after GnRHa triggering Progesterone and Estradiol
 Luteal phase rescue after GnRHa triggering Progesterone and Estradiol L. Engmann University of Connecticut Disclaimer Fertility Speaker Bureau Merck Pharmaceuticals Introduction GnRH agonist is effective
Luteal phase rescue after GnRHa triggering Progesterone and Estradiol L. Engmann University of Connecticut Disclaimer Fertility Speaker Bureau Merck Pharmaceuticals Introduction GnRH agonist is effective
Milder is better? Advantages and disadvantages of "mild" ovarian stimulation for human in vitro fertilization
 Milder is better? Advantages and disadvantages of "mild" ovarian stimulation for human in vitro fertilization Revelli et al. Reproductive Biology and Endocrinology 2011, 9:25 Presenter: R2 孫怡虹 Background
Milder is better? Advantages and disadvantages of "mild" ovarian stimulation for human in vitro fertilization Revelli et al. Reproductive Biology and Endocrinology 2011, 9:25 Presenter: R2 孫怡虹 Background
Is it the seed or the soil? Arthur Leader, MD, FRCSC
 The Physiological Limits of Ovarian Stimulation Is it the seed or the soil? Arthur Leader, MD, FRCSC Objectives 1. To consider how ovarian stimulation protocols work in IVF 2. To review the key events
The Physiological Limits of Ovarian Stimulation Is it the seed or the soil? Arthur Leader, MD, FRCSC Objectives 1. To consider how ovarian stimulation protocols work in IVF 2. To review the key events
Page 196. Corresponding Author: Swati Garg, Volume 3 Issue - 4, Page No
 ISSN- O: 2458-868X, ISSN P: 2458 8687 Index Copernicus Value: 49. 23 SJIF Impact Factor: 4.271 International Journal of Medical Science and Innovative Research (IJMSIR) IJMSIR : A Medical Publication Hub
ISSN- O: 2458-868X, ISSN P: 2458 8687 Index Copernicus Value: 49. 23 SJIF Impact Factor: 4.271 International Journal of Medical Science and Innovative Research (IJMSIR) IJMSIR : A Medical Publication Hub
Cigna Drug and Biologic Coverage Policy
 Cigna Drug and Biologic Coverage Policy Subject Infertility Injectables Table of Contents Coverage Policy... 1 General Background...16 Coding/Billing Information...20 References...20 Effective Date...
Cigna Drug and Biologic Coverage Policy Subject Infertility Injectables Table of Contents Coverage Policy... 1 General Background...16 Coding/Billing Information...20 References...20 Effective Date...
Managing Anovulatory Infertility
 Managing Anovulatory Infertility Patience, controversy, options, and surveillance Edward D. Tarnawa, M.D. Reproductive Endocrinology & Infertility THEGOS Annual Meeting, 2017 I have no conflicts of interest
Managing Anovulatory Infertility Patience, controversy, options, and surveillance Edward D. Tarnawa, M.D. Reproductive Endocrinology & Infertility THEGOS Annual Meeting, 2017 I have no conflicts of interest
clinical outcome and hormone profiles before and after laparoscopic electroincision of the ovaries in women with polycystic ovary syndrome
 & clinical outcome and hormone profiles before and after laparoscopic electroincision of the ovaries in women with polycystic ovary syndrome Zulfo Godinjak¹*, Ranka Javorić² 1 Gynecology and Obstetrics
& clinical outcome and hormone profiles before and after laparoscopic electroincision of the ovaries in women with polycystic ovary syndrome Zulfo Godinjak¹*, Ranka Javorić² 1 Gynecology and Obstetrics
10.7 The Reproductive Hormones
 10.7 The Reproductive Hormones December 10, 2013. Website survey?? QUESTION: Who is more complicated: men or women? The Female Reproductive System ovaries: produce gametes (eggs) produce estrogen (steroid
10.7 The Reproductive Hormones December 10, 2013. Website survey?? QUESTION: Who is more complicated: men or women? The Female Reproductive System ovaries: produce gametes (eggs) produce estrogen (steroid
9.4 Regulating the Reproductive System
 9.4 Regulating the Reproductive System The Reproductive System to unite a single reproductive cell from a female with a single reproductive cell from a male Both male and female reproductive systems include
9.4 Regulating the Reproductive System The Reproductive System to unite a single reproductive cell from a female with a single reproductive cell from a male Both male and female reproductive systems include
Best practices of ASRM and ESHRE
 Best practices of ASRM and ESHRE Late submission Cortina d Ampezzo, Italy 1-3 March 2012 A joint meeting between the American Society for Reproductive Medicine and the European Society of Human Reproduction
Best practices of ASRM and ESHRE Late submission Cortina d Ampezzo, Italy 1-3 March 2012 A joint meeting between the American Society for Reproductive Medicine and the European Society of Human Reproduction
SAMPLE REPORT. Order Number: PATIENT. Age: 40 Sex: F MRN:
 Patient: Age: 40 Sex: F MRN: SAMPLE PATIENT Order Number: Completed: Received: Collected: SAMPLE REPORT Progesterone ng/ml 0.34 0.95 21.00 DHEA-S mcg/dl Testosterone ng/ml 48 35 0.10 0.54 0.80 430 Sex
Patient: Age: 40 Sex: F MRN: SAMPLE PATIENT Order Number: Completed: Received: Collected: SAMPLE REPORT Progesterone ng/ml 0.34 0.95 21.00 DHEA-S mcg/dl Testosterone ng/ml 48 35 0.10 0.54 0.80 430 Sex
Reproductive Progesterone
 Reproductive Progesterone Analyte Information 1 Progesterone Introduction Progesterone is a natural gestagen belonging to the C 21 steroid group. It is also known as P4 (or pregn-4-ene-3,20-dione, or 4-pregnene-3,20-dione).
Reproductive Progesterone Analyte Information 1 Progesterone Introduction Progesterone is a natural gestagen belonging to the C 21 steroid group. It is also known as P4 (or pregn-4-ene-3,20-dione, or 4-pregnene-3,20-dione).
