3. UROGEN WELL D-ONE KIT CONTENTS
|
|
|
- Nathan Wilkinson
- 5 years ago
- Views:
Transcription
1 UROGEN WELL D-ONE System for the presumptive identification and antimicrobial susceptibility of urogenital mycoplasmas, Neisseria spp., Streptococcus B, Staphylococcus aureus, Enterococcus spp., Escherichia coli, Gardnerella vaginalis, Trichomonas vaginalis, and Candida spp. 1. INTRODUCTION Mycoplasma hominis and Ureaplasma urealyticum / parvum are the species of the class Mollicutes isolated more frequently from the urogenital tract and considered opportunistic pathogens. It has demonstrated a direct relationship between the isolation of these microorganisms and certain diseases such as bacterial vaginosis (BV), pelvic inflammatory disease, infections during pregnancy, preterm labor and neonatal infections (1-2) Rapid diagnosis available today may involve the use of expensive tests not available in all hospitals and not useful for determining antimicrobial susceptibility, essential for treatments implementations, epidemiological studies, etc. In the light of this, a system that allows rapidly growing, in addition to antimicrobial susceptibility test in just hours, without additional equipment, can be an useful tool in the hands of the microbiologist and clinician. In many cases, the positivity of urogenital mycoplasma is associated with the presence of other microorganisms that cause genitourinary infections, such as Neisseria spp., Streptococcus agalactiae (group B), Staphlyococcus aureus, Enterococcus spp., Escherichia coli, Gardnerella vaginalis, Trichomonas vaginalis, and Candida spp. The simultaneous identification of these agents can guide the clinical diagnosis and the procedure to follow with the patient. (1)(7)(8) 2. PRINCIPLE System composed of a polypropylene plate containing 32 conical wells for better viewing of the colorimetric reactions that occur as result of the growth of microorganisms in specially formulated media for selective culture of: Mycoplasma hominis, Mycoplasma spp., Ureaplasma urealyticum/parvum. The Kit allows the presumptive identification of Neisseria spp., Streptococcus agalactiae (group B), Staphlyococcus aureus, Enterococcus spp., Escherichia coli, Gardnerella vaginalis, Trichomonas vaginalis and Candida spp. The system allows to perform, directly from the content of the wells, the sowing in selective media, the microscopic observation, the serological and molecular tests, etc. (6)(11)(12) 3. UROGEN WELL D-ONE KIT CONTENTS (REF. MS01281) 10 Identification panels UROGEN WELL D-ONE (REF. MS01281) 10 x 10 ml Sterile physiological Saline Solution (REF. MS01304) 10 Sterile Swabs 4.REAGENTS REQUI BUT NOT PROVIDED Myco D-One Transport Medium 10 x 2 ml Liquid Sterile Paraffin Culture supplemented media plate, ready to use, for Mycoplasma spp. General Laboratory equipment (REF. MS01314) (REF. MS01316)
2 5. PANEL COMPOSITION Well 1: Selective Medium for the isolation of Mycoplasma hominis Well 2: Selective Medium for the isolation of Mycoplasma hominis Well 3: Selective Medium for presumptive identification of Mycoplasma spp. Well 4: Selective Medium for presumptive identification of Mycoplasma spp. Well 5: Selective Medium for growing of CCU 10 4 Ureaplasma urealyticum/parvum Well 6: Selective Medium for growing of CCU 10 5 Ureaplasma urealyticum/parvum Well 7: Selective Medium for presumptive identification of Ureaplasma urealyticum/parvum Well 8: Enriched Culture Medium for growth control Well 9: Culture medium containing Levofloxacin 1 µg/ml Well 10: Culture medium containing Josamycin 2 µg/ml Well 11: Culture medium containing Moxifloxacin 0,25 µg/ml Well 12: Culture medium containing Doxycycline 4 µg/ml Well 13: Culture medium containing Levofloxacin 2 µg/ml Well 14: Culture medium containing Josamycin 2 µg/ml Well 15: Culture medium containing Moxifloxacin 2 µg/ml Well 16: Culture medium containing Doxycycline 4 µg/ml Well 17: Culture medium containing Levofloxacin 2 µg/ml Well 18: Culture medium containing Josamycin 8 µg/ml Well 19: Culture medium containing Moxifloxacin 0,5 µg/ml Well 20: Culture medium containing Doxycycline 8 µg/ml Well 21: Culture medium containing Levofloxacin 4 µg/ml Well 22: Culture medium containing Josamycin 8 µg/ml Well 23: Culture medium containing Moxifloxacin 4µg/mL Well 24: Culture medium containing Doxycycline 8 µg/ml Well 25: Selective Medium for presumptive identification of Neisseria spp. Well 26: Selective Medium for presumptive identification of Streptococcus agalactiae Well 27: Selective Medium for presumptive identification of Staphlyococcus aureus Well 28 Selective Medium for presumptive identification of Enterococcus spp. Well 29: Selective Medium for presumptive identification of di Escherichia coli Well 30: Selective Medium for presumptive identification of di Candida spp. Well 31: Selective Medium for presumptive identification of Gardnerella vaginalis Well 32: Selective Medium for presumptive identification of Trichomonas vaginalis 6. TEST PROCEDURE 6.1 SAMPLE COLLECTION Sample: endocervical exudate, vaginal exudate, urethral exudate, urine, seminal fluid For the best performance of this system the sample should be taken aseptically according to the methodology implemented in each hospital center and before starting antimicrobial treatment. The sample should be processed immediately after collection. Do not refrigerate or freeze. Protect from light and sun.
3 The results obtained by UROGEN WELL D-ONE should be compared with complementary methods of analysis, established in each laboratory. 6.2 PROCEDURE Sample: Endocervical exudate, vaginal exudate, urethral exudate: - Take the sample according to the procedures established in each laboratory - Resuspend the swab with the sample in sterile saline solution supplied in the kit, left in saline solution for 3-5 minutes, stirring and pressing the swab against the tube walls until obtain an homogeneous suspension. - Add 150 µl of the obtained suspension in each well of the system panel - Add two drops of sterile paraffin in the wells from 1 to Incubate at 36±1 C for hours. Urine-Seminal Liquid - Take the sample according to the procedures established in each laboratory - Reconstitute 200 µl (4 drops) of the obtained sample in sterile saline solution supplied in the kit - Add 150 µl (3 drops) of final suspension obtained in each well of the system - Add two drops of sterile paraffin in the wells from 1 to Incubate at 36 ± 1 C for hours. Incubation It is recommended to incubate the plate for 24/48 hours. It is recommended to incubate up to 6 days, for the identification of M. spp. Presumptive identification of microorganisms included in the System Ureaplasma spp is positive after hours. Some "Fastidious" strains of Ureaplasma spp. can be positive after 36 hours. The presumptive identification of Ureaplasma urealyticum/parvum is associated to the positivity of well 5 and 7. The positivity of well 6 depends on if the concentration (CCU/mL) of Ureaplasma urealyticum/parvum is Strains belonging to the family of Ureaplasma spp. are not able to growth in the well 8. IDENTIFICATION OF Ureaplasma urealyticum/parvum: Ureaplasma urealyticum/parvum CCU ID ID U.urealyticum/parvum GC Ureaplasma urealyticum/parvum CCU ID ID U.urealyticum/parvum GC Mycoplasma hominis, is positive after 24/48 hours. Mycoplasma spp., can be positive after 3 days. The incubation should be extended up to 6 days for "fastidious" species. An incubation longer than 6 days, can be realized at the discretion of the microbiologist.
4 IDENTIFICATION OF M.hominis: ID ID ID ID M.hominis M.spp IDENTIFICATION OF M.spp.: ID ID ID ID M.hominis M.spp Some strains of M.spp., change the colour of the identification wells in different time. The positivity of at least one well of identification in M.spp area, after 3 days of incubation is an indicator of positivity. The antimicrobial susceptibility wells area are intended for M. hominis and Ureaplasma urealyticum/parvum. The species of M.spp do not cause the colour changing in this area. The presence of a transparent yellow color in ID wells for M.spp., should be confirmed by complementary techniques to detect mycoplasmas. In order to perform this confirmation, it is necessary to take carefully the content of the well. In the case that a turbidity is observed in the well 25 Neisseria spp., it is recommended to perform an oxidase test; the confirmation of the Neisseria spp. is realized sowing the well content in a selective medium. It is recommended to perform the test of Sodium Hippurate on the content of well 26 Streptococcus agalactiae. Just strains of S. agalactiae non haemolytic can take up to 48 hours for the colorimetric reaction in the well, or cause any kind of turbidity without colour changing. The Sodium Hippurate test in disk or tube, as well as the latex test of the well content accelerate the microbial identification. It is recommended to perform from the well 27 Staphylococcus aureus a confirmatory coagulase tube test or a latex test, despite of the 99,9% of strains of S.aureus are positive to the medium contained in the system. The positivity of the well 28 is associated to the presence of Enterococcus spp. The medium included in the presumptive identification well of E.coli, detect E.coli inactive. In the case of well 30 and 32, it is recommended a microscopic observation (40 X) of culture media containing in the identification well. Observe the morphological features of Candida spp. and Trichomonas vaginalis. In the case of the well 31, a red colour is an indicator of bacterial growth. It is recommended a microscopic observation ( 100 X) to notice the morphological features of Gardnerella vaginalis. Incubations longer then hours may be carried out based on the consideration of the microbiologist, for a period of 5 days, but the formulations of this kit allow the growth of the most common urogenital mycoplasma (Mycoplasma hominis and Ureaplasma urealyticum / parvum ) in hours (5-6). In the case of incubations longer than 48 hours, it may occur a volume decrease of well 30 and 31, that are indicated for a possible microscopic observation of the microbial structures. The reading of results, despite of this, is not compromised. Nevertheless, to perform the microscopic observation, it can be added a drop of physiological saline solution in order to resuspend the content before preparing the slide. The colorimetric reaction in the well does not interfere with the observation of the microbial structure. It is recommended to use of traditional methods established for the conventional identification of microorganisms that can be detected with this test (5).
5 7. RESULTS INTERPRETATION The results are evidenced by color changes that occur in wells according to reactions due to chemical or chromogenic components contained in specific formulations for each microorganism (See The Reaction Table). Additional microscopic observation is recommended as described in this IFU. Culture of samples is recommended in selective media mentioned in section 4 (Materials required but not provided) in the case of positive samples that have to be confirmed. To perform this procedure, the samples can be stored in the transport medium referred in paragraph 4 or it can be realized a culture from the Wells ID System. 8. WARNING AND PRECAUTIONS 1. For professional and in vitro diagnostic use only, not to be used by the general public. 2. The samples have to be treated as potentially infectious and the test must be carried out only by trained personnel handling clinical and / or microbiological laboratory techniques. 3. Do not open the sealed pouch unless ready to perform the assay. 4. Do not use expired devices. 5. Do not use components from any other type of test kit as a substitute for the components in this kit. 6. Wear protective clothing and disposable gloves while handling the kit reagents and clinical specimens. Wash hands thoroughly after performing the test. 7. Do not smoke, drink or eat in areas where specimens or kit reagents are being handled. 8. Dispose of all specimens and materials used to perform the test as bio-hazardous waste 9.LIMITATIONS Samples collected after antimicrobial treatment may provide false - negative results (4-5).. The samples such as vaginal or endocervical exudate, with ph values due to infrequent particular infectious processes can cause nonspecific reactions with positive colorations in the identification and antimicrobial wells for urogenital mycoplasmas. ( 7-8 ) A sample with a high concentration of color changing units of urogenital mycoplasmas can lead to widespread positivity of identification panel, in such cases it is recommended to dilute the sample (7-8-9 ). The use of the special Myco D-One Transport Medium for sample is recommended for the preservation of samples and / or transportation. In this way samples can be stored at 4 ⁰C for 72 hours or frozen at -70 ⁰ C for 8 weeks. A collection or an improper storage may cause sample microbiological contamination or incompatible agents a with the proper functioning of this kit. The presence of microorganisms responsible of urease enzyme, causes a reaction in the well 8. The presence of a high concentration of Gram negative bacteria, Streptococcus spp., Enterococcus spp., S.aureus, can causes a colour changing in mycoplasmas area, in this cases a turbidity is observed in wells, in addition to the colour changing and the positivity of the relative identification wells. The possible presence of Pseudomonas spp. or multi resistant microorganisms cause nonspecific reactions in the system. Read this instructions for use carefully, in order to avoid mistakes 10.QUALITY CONTROL Each batch of UROGEN Well D-ONE is submitted to a rigorous quality control with the following bacterial reference strains. The control strains are used for both positive reactions from different wells to check the proper functioning of the media formulations of the different wells before any nonspecific reactions.
6 non fastidious Mycoplasmas Strain ATCC Mycoplasma hominis Mycoplasma penetrans Ureaplasma urealyticum For growth control Ureaplasma urealyticum To detect the antimicrobial susceptibility Ureaplasma parvum "Fastidious Mycoplasmas o Slow growing Mycoplasmas Strain ATCC Mycoplasma genitalium Mycoplasma fermentans Mycoplasma hyorhinis * *The inclusion of strain Mycoplasma hyorhinis (cultivar alpha) was performed to test the ability of the formulation to promote the growth of difficult species ( M. genitalium and M. fermentans) in traditional culture media because the dynamics of growth and nutritional requirements of these species are very similar. Other tested strains Candida albicans ATCC Candida tropicalis ATCC Candida krusei ATCC Gardnerella vaginalis ATCC Trichomonas vaginalis ATCC Escherichia coli ATCC Neisseria gonorrhoeae ATCC Streptococcus agalactiae ATCC Staphylococcus aureus ATCC Streptococcus faecalis ATCC The strains used for Quality Control have been carefully selected according to the recommendations published for the growth and susceptibility to antimicrobial agents (5-10). Each laboratory should establish its own internal quality controls. 11. STORAGE Store at 2-8 C in its original package. Do not store near heat sources and avoid extreme temperature variations. Under these conditions the product is valid until the expiration date shown on label of primary and secondary box. Do not use after this date. Discard if there are signs of deterioration. 12. BIBLIOGRAPHY 1. Waites KB, Schelonka RL, Xiao L, Grigsby PL, Novy MJ. Congenital and opportunistic infections: Ureaplasma species and Mycoplasma hominis. Semin Fetal Neonatal Med. 2009;13(4): Kallapur SG, Kramer BW, Jobe AH. Ureaplasma and BPD Semin Perinatol. 2013;37(2): WHO Interim Guidelines 2007: Infection prevention and control of epidemic- and pandemic-prone acute respiratory diseases in health care. WHO/CDS/EPR/ Begoña JCC, Meseguer MAP, Oliver APP, Puig de la Bellacasa J Diagnóstico microbiológico de las infecciones bacterianas del tracto respiratorio inferior. Procedimientos en Microbiología Clínica. SEIM Ed. Cercenado E y Cantón R 5. Acosta BB, Gema MC, Matas GL. Diagnóstico microbiológico de las infecciones por Mycoplasma spp. y Ureaplasma spp Ed. Cercenado E y Cantón R. SEIM PNT-M-03. Procesamiento microbiológico de
7 las muestras genitourinarias y de otras procedencias para cultivo de Ureaplasma spp. y Mycoplasma hominis 6. Validazione del Sistema UROGEN WELL D-ONE in campioni da Infezioni da Mycoplasma spp. ed altri patogeni urogenitali. Comparazione con metodi tradizionali. UROGEN WELL D-ONE Technical File, C.P.M. sas, Krauss-Silva L, Almada-Horta A, Alves MB, Camacho KG, Moreira ME, Braga A. Basic vaginal ph, bacterial vaginosis and aerobic vaginitis: prevalence in early pregnancy and risk of spontaneous preterm delivery, a prospective study in a low socioeconomic and ultiethnic South American population. BMC Pregnancy Childbirth ;14:107. doi: / Hemalatha R, Ramalaxmi BA, Swetha E, Balakrishna N, Mastromarino P. Evaluation of vaginal ph for detection of bacterial vaginosis Indian J Med Res ;138(3): Granato PA, FAAM D. Vaginitis: Clinical and Laboratory Aspects for Diagnosis. Clin Microb New. 2010: 32(15): Clinical and Laboratory Standard Institute (CLSI), M43 -A, Methods for Antimicrobial Susceptibility Testing for Human Mycoplasmas; Approved Guideline, First Edition (2011) 11. Favalli C, Favaro M, Diagnosis of Mycoplasma and Ureaplasma infection by a New Accellerated Micro- Method. ECCMID, Favalli C, Favaro M, Comparative study of a new system MYCO WELL D-ONE with molecular methods. Università di Tor Vergata, 2014
8 COLORIMETRIC REACTIONS POSITIVE NEGATIVE Well 1: Selective Medium for the isolation of Mycoplasma hominis Well 2: Selective Medium for the isolation of Mycoplasma hominis Well 3: Selective medium for the presumptive identification of Mycoplasma spp. Well 4: Selective Medium for the presumptive identification of Mycoplasma spp. Well 5: Selective Medium for the growth of CCU 10 4 Ureaplasma urealyticum/parvum Well6: Selective Medium for the growth of CCU 10 5 Ureaplasma urealyticum/parvum Well 7: Selective Medium for the presumptive identification of Ureaplasma urealyticum/parvum Well 8: Culture Medium to test the presence, in the sample, of microorganism responsible for urease enzyme that do not belong to the family of Ureaplasma Urealyticum (Strain s of Ureaplasma spp., are not able to grow in the well 8) Well 9: Culture medium containing Levofloxacin 1 µg/ml ANTIMICROBIAL SUSCEPTIBILITY TEST RESISTANT SENSIBILE Well 10: Culture medium containing Josamycin 2 µg/ml Well 11: Culture medium containing Moxifloxacin 0,25 µg/ml. Well 12: Culture medium containing Doxycycline 4 µg/ml Well 13: Culture medium containing Levofloxacin 2 µg/ml Well 14: Culture medium containing Josamycin 2 µg/ml Well 15: Culture medium containing Moxifloxacin 2 µg/ml. Well 16: Culture medium containing Doxycycline 4 µg/ml Well 17: Culture medium containing Levofloxacin 2 µg/ml Well 18: Culture medium containing Josamycin 8 µg/ml Well 19: Culture medium containing Moxifloxacin 0,5 µg/ml Well 20: Culture medium containing Doxycycline 8 µg/ml Well 21: Culture medium containing Levofloxacin 4 µg/ml Well 22: Culture medium containing Josamycin 8 µg/ml Well 23: Culture medium containing Moxifloxacin 4 µg/ml Well 24: Culture medium containing Doxycycline 8 µg/ml POSITIVE NEGATIVE Well 25: Selective Medium for the presumptive identification of Neisseria spp. CLOUDY Well 26: Selective Medium for the identification of Streptococcus agalactiae GREEN LIGHT Well 27: Selective Medium for the presumptive identification of Staphlyococcus aureus DARK PURPLE LIGHT Well 28: Selective Medium for the presumptive identification of Enterococcus spp. BLACK LIGHT Well 29: Selective Medium for the presumptive identification of Escherichia coli GREEN WHITE Well 30: Selective Medium for the presumptive identification of Candida spp. GREEN NO COLOUR /CLOUDY WHITE Well 31: Selective Medium for the presumptive identification of Gardnerella vaginalis NO COLOUR Well 32: Selective Medium for the presumptive identification of Trichomonas vaginalis LIGHT BLUE LIGHT BLUE
9 Index of Symbols Reference code LOT number Expiration date Temperature limitation/store between For in vitro diagnostic use Do not reuse Fragile, handle with care Consult Instructions for Use Manufacturer Contains sufficient for <n> tests Conform to the Directive 98/79/EC on in vitro diagnostic medical device C.P.M. sas Via degli Olmetti, (Zona Ind.le) Formello, (ROMA), Italia Tel. +39/ / Fax. +39/ cpm@cpmsas.it
10
MYCO WELL D-ONE Rev /11/2015
 MYCO WELL D-ONE System for the presumptive identification and antimicrobial susceptibility test of urogenital mycoplasmas, Gardnerella vaginalis, Trichomonas vaginalis, Candida albicans and Candida spp.
MYCO WELL D-ONE System for the presumptive identification and antimicrobial susceptibility test of urogenital mycoplasmas, Gardnerella vaginalis, Trichomonas vaginalis, Candida albicans and Candida spp.
ENG MYCO WELL D- ONE REV. 1.UN 29/09/2016 REF. MS01283 REF. MS01321 (COMPLETE KIT)
 ENG MYCO WELL D- ONE MYCO WELL D-ONE System for the presumptive identification and antimicrobial susceptibility test of urogenital mycoplasmas, Gardnerella vaginalis, Trichomonas vaginalis, Candida albicans
ENG MYCO WELL D- ONE MYCO WELL D-ONE System for the presumptive identification and antimicrobial susceptibility test of urogenital mycoplasmas, Gardnerella vaginalis, Trichomonas vaginalis, Candida albicans
A.F. GENITAL SYSTEM. ITEMS NECESSARY BUT NOT INCLUDED IN THE KIT A.F. GENITAL SYSTEM Reagent (ref ) Mycoplasma Transport Broth (ref.
 A.F. GENITAL SYSTEM ENGLISH System for detection, count and susceptibility test of pathogenic urogenital microorganisms DESCRIPTION A.F. GENITAL SYSTEM is a 24-well system containing desiccated biochemical
A.F. GENITAL SYSTEM ENGLISH System for detection, count and susceptibility test of pathogenic urogenital microorganisms DESCRIPTION A.F. GENITAL SYSTEM is a 24-well system containing desiccated biochemical
Chlamydia Rapid Screen Test (RAP-2858) RUO in the USA. Revised 28 Jul 2006
 INDICATION For the rapid detection of Chlamydia Trachomatis antigens in swab specimens. For in vitro diagnostic use only, except in the United States where it is intended for Research Use Only. SUMMARY
INDICATION For the rapid detection of Chlamydia Trachomatis antigens in swab specimens. For in vitro diagnostic use only, except in the United States where it is intended for Research Use Only. SUMMARY
Chlamydia trachomatis (CHLa)Test Kit
 Chlamydia trachomatis (CHLa)Test Kit Instructions For Use Format: Cassette Specimen: Urethral/Genital Swab Catalog Number: VEL-001-CHLa * Please read the instructions carefully before use INTENDED USE
Chlamydia trachomatis (CHLa)Test Kit Instructions For Use Format: Cassette Specimen: Urethral/Genital Swab Catalog Number: VEL-001-CHLa * Please read the instructions carefully before use INTENDED USE
β CARBA Test Rapid detection of carbapenemase-producing Enterobacteriaceae strains Contents 1. INTENDED USE
 β CARBA Test 25 68260 Rapid detection of carbapenemase-producing Enterobacteriaceae strains 881159 2015/05 Contents 1. INTENDED USE 2. SUMMARY AND EXPLANATION OF THE TEST 3. PRINCIPLE OF THE PROCEDURE
β CARBA Test 25 68260 Rapid detection of carbapenemase-producing Enterobacteriaceae strains 881159 2015/05 Contents 1. INTENDED USE 2. SUMMARY AND EXPLANATION OF THE TEST 3. PRINCIPLE OF THE PROCEDURE
Strep-a-Test Twister Test
 Strep-a-Test Twister Test Code: 24524 A rapid test for the qualitative detection of Strep A antigen in throat swab specimens. For professional in vitro diagnostic use only. INTENDED USE The Strep A Twist
Strep-a-Test Twister Test Code: 24524 A rapid test for the qualitative detection of Strep A antigen in throat swab specimens. For professional in vitro diagnostic use only. INTENDED USE The Strep A Twist
Rapid-VIDITEST. Strep A Card. One step Strep A Card for the detection of Group A Streptococcal antigen from throat swabs or culture.
 Rapid-VIDITEST Strep A Card One step Strep A Card for the detection of Group A Streptococcal antigen from throat swabs or culture. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, Vestec,
Rapid-VIDITEST Strep A Card One step Strep A Card for the detection of Group A Streptococcal antigen from throat swabs or culture. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, Vestec,
Human Influenza A (Swine Flu) Rapid test
 Human Influenza A (Swine Flu) Rapid test Cat.No: DTSXY-Z9 Lot. No. (See product label) Size 20T Intended use The Influenza A (Swine Flu) test is a rapid chromatographic immunoassay for the qualitative
Human Influenza A (Swine Flu) Rapid test Cat.No: DTSXY-Z9 Lot. No. (See product label) Size 20T Intended use The Influenza A (Swine Flu) test is a rapid chromatographic immunoassay for the qualitative
Hepatitis C Virus (HCV) Antibody Test
 Hepatitis C Virus (HCV) Antibody Test Instructions For Use Format: Cassette For: Catalog Number: VEL-001-HCV Specimen: Serum/Plasma * Please read the instructions carefully before use INTENDED USE Hepatitis
Hepatitis C Virus (HCV) Antibody Test Instructions For Use Format: Cassette For: Catalog Number: VEL-001-HCV Specimen: Serum/Plasma * Please read the instructions carefully before use INTENDED USE Hepatitis
SAMPLE PROCEDURE , 07/11
 SAMPLE PROCEDURE This Sample Procedure is not intended as a substitute for your facility s Procedure Manual or reagent labeling, but rather as a model for your use in customizing for your laboratory s
SAMPLE PROCEDURE This Sample Procedure is not intended as a substitute for your facility s Procedure Manual or reagent labeling, but rather as a model for your use in customizing for your laboratory s
Rapid-VIDITEST. Strep A Blister. One step Strep A Blister for the detection of Group A Streptococcal antigen from throat swabs or culture.
 Rapid-VIDITEST Strep A Blister One step Strep A Blister for the detection of Group A Streptococcal antigen from throat swabs or culture. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II
Rapid-VIDITEST Strep A Blister One step Strep A Blister for the detection of Group A Streptococcal antigen from throat swabs or culture. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II
Rapid-VIDITEST. Influenza A+B
 Rapid-VIDITEST Influenza A+B (One step Influenza A+B blister Test for the detection of Influenza type A and type B from nasal swabs, nasal wash or nasal aspirate specimens). Instruction manual Producer:
Rapid-VIDITEST Influenza A+B (One step Influenza A+B blister Test for the detection of Influenza type A and type B from nasal swabs, nasal wash or nasal aspirate specimens). Instruction manual Producer:
FAECAL WELL D-ONE. System for the presumptive identification and antibiotic susceptibility of pathogenic microorganisms of the intestinal tract.
 FAECAL WELL D-ONE System for the presumptive identification and antibiotic susceptibility of pathogenic microorganisms of the intestinal tract. 1. INTRODUCTION Intestinal and gastrointestinal infections
FAECAL WELL D-ONE System for the presumptive identification and antibiotic susceptibility of pathogenic microorganisms of the intestinal tract. 1. INTRODUCTION Intestinal and gastrointestinal infections
Rapid-VIDITEST Shigella dysenteriae
 Rapid-VIDITEST Shigella dysenteriae One step Shigella dysenteriae test for the qualitative detection of Shigella dysenteriae in faeces. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365,
Rapid-VIDITEST Shigella dysenteriae One step Shigella dysenteriae test for the qualitative detection of Shigella dysenteriae in faeces. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365,
Microbiology products. Liofilchem Chromatic
 ET Microbiology products M IC R O PL A N www.liofilchem.net Liofilchem Chromatic c h ro m o g e n i c c u l t u re m e d i a LIOFILCHEM s.r.l. Via Scozia, Zona Industriale 64026 Roseto degli Abruzzi (Te)
ET Microbiology products M IC R O PL A N www.liofilchem.net Liofilchem Chromatic c h ro m o g e n i c c u l t u re m e d i a LIOFILCHEM s.r.l. Via Scozia, Zona Industriale 64026 Roseto degli Abruzzi (Te)
Murex Chlamydia Verification Kit
 1F83-01 E C06DK05GB Murex Chlamydia Verification Kit DK05 For the confirmatory testing of clinical samples reactive in Murex Chlamydia (1F82-01) Note Changes Highlighted 2005 Abbott / Printed in the UK
1F83-01 E C06DK05GB Murex Chlamydia Verification Kit DK05 For the confirmatory testing of clinical samples reactive in Murex Chlamydia (1F82-01) Note Changes Highlighted 2005 Abbott / Printed in the UK
Treponema Pallidum (TP) Antibody Test
 Treponema Pallidum (TP) Antibody Test Instructions For Use Format: Cassette For: Catalog Number: VEL-001-TP Specimen: Serum/Plasma * Please read the instructions carefully before use INTENDED USE Velotest
Treponema Pallidum (TP) Antibody Test Instructions For Use Format: Cassette For: Catalog Number: VEL-001-TP Specimen: Serum/Plasma * Please read the instructions carefully before use INTENDED USE Velotest
RealLine Mycoplasma genitalium Str-Format
 Instructions for use ASSAY KIT FOR THE QUALITATIVE DETECTION OF MYCOPLASMA GENITALIUM DNA BY REAL-TIME PCR METHOD In vitro Diagnostics () VBD4396 96 Tests valid from December 2018 Rev06_1218_EN Page 1
Instructions for use ASSAY KIT FOR THE QUALITATIVE DETECTION OF MYCOPLASMA GENITALIUM DNA BY REAL-TIME PCR METHOD In vitro Diagnostics () VBD4396 96 Tests valid from December 2018 Rev06_1218_EN Page 1
Rapid-VIDITEST. Helicobacter pylori. One step Helicobacter pylori Blister test. Instruction manual
 Rapid-VIDITEST Helicobacter pylori One step Helicobacter pylori Blister test. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, 252 50 Vestec, Czech Republic, Tel.: +420 261 090 565,
Rapid-VIDITEST Helicobacter pylori One step Helicobacter pylori Blister test. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, 252 50 Vestec, Czech Republic, Tel.: +420 261 090 565,
Rapid-VIDITEST Swine Flu
 Rapid-VIDITEST Swine Flu One Step Influenza type A Antigen Card test. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, 252 50 Vestec, Czech Republic, Tel.: +420 261 090 565, www.vidia.cz
Rapid-VIDITEST Swine Flu One Step Influenza type A Antigen Card test. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, 252 50 Vestec, Czech Republic, Tel.: +420 261 090 565, www.vidia.cz
Evaluation of the feasibility of the VACUETTE Urine CCM tube for microbial testing of urine samples
 Evaluation of the feasibility of the VACUETTE Urine CCM tube for microbial testing of urine samples Background The VACUETTE Urine CCM tube is for the collection, transport and storage of urine samples
Evaluation of the feasibility of the VACUETTE Urine CCM tube for microbial testing of urine samples Background The VACUETTE Urine CCM tube is for the collection, transport and storage of urine samples
Rapid-VIDITEST C. difficile Ag (GDH) Card/Blister
 Li StarFish S.r.l. Via Cavour, 35-20063 Cernusco S/N (MI), Italy Tel. +39-02-92150794 - Fax. +39-02-92157285 info@listarfish.it -www.listarfish.it Rapid-VIDITEST C. difficile Ag (GDH) Card/Blister One
Li StarFish S.r.l. Via Cavour, 35-20063 Cernusco S/N (MI), Italy Tel. +39-02-92150794 - Fax. +39-02-92157285 info@listarfish.it -www.listarfish.it Rapid-VIDITEST C. difficile Ag (GDH) Card/Blister One
Rapid-VIDITEST. Influenza A
 Rapid-VIDITEST Influenza A (One step Influenza A Card test for the detection of Influenza type A antigen from human nasopharyngeal specimens (swab, nasopharyngeal wash and aspirate). Instruction manual
Rapid-VIDITEST Influenza A (One step Influenza A Card test for the detection of Influenza type A antigen from human nasopharyngeal specimens (swab, nasopharyngeal wash and aspirate). Instruction manual
Rapid-VIDITEST Enterovirus
 Rapid-VIDITEST Enterovirus A rapid one step Enterovirus Card test for the qualitative detection of Enterovirus antigens in human feces. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365,
Rapid-VIDITEST Enterovirus A rapid one step Enterovirus Card test for the qualitative detection of Enterovirus antigens in human feces. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365,
H. pylori Antigen ELISA Kit
 H. pylori Antigen ELISA Kit Catalog Number KA3142 96 assays Version: 04 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
H. pylori Antigen ELISA Kit Catalog Number KA3142 96 assays Version: 04 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
Rapid-VIDITEST. Rota-Adeno Blister. One Step Rotavirus and Adenovirus Antigen Blister test. Instruction manual
 Rapid-VIDITEST Rota-Adeno Blister One Step Rotavirus and Adenovirus Antigen Blister test. Instruction manual INTENDED USE: The Rapid-VIDITEST Rota-Adeno Blister test is a rapid chromatographic immunoassay
Rapid-VIDITEST Rota-Adeno Blister One Step Rotavirus and Adenovirus Antigen Blister test. Instruction manual INTENDED USE: The Rapid-VIDITEST Rota-Adeno Blister test is a rapid chromatographic immunoassay
Instructions for use. TSH rat ELISA. Please use only the valid version of the Instructions for Use provided with the kit AR E-8600
 Instructions for use TSH rat ELISA AR E-8600 TSH rat ELISA 1. INTRODUCTION 1.1 Intended use The TSH rat ELISA is an enzyme immunoassay for the quantitative measurement of TSH in rat serum. For research
Instructions for use TSH rat ELISA AR E-8600 TSH rat ELISA 1. INTRODUCTION 1.1 Intended use The TSH rat ELISA is an enzyme immunoassay for the quantitative measurement of TSH in rat serum. For research
Rotavirus Test Kit. Instructions For Use. Format: Cassette Specimen: Fecal Extract Catalog Number: VEL-001-ROTA
 Rotavirus Test Kit Instructions For Use Format: Cassette Specimen: Fecal Extract Catalog Number: VEL-001-ROTA * Please read the instructions carefully before use INTENDED USE Velotest Rotavirus Test is
Rotavirus Test Kit Instructions For Use Format: Cassette Specimen: Fecal Extract Catalog Number: VEL-001-ROTA * Please read the instructions carefully before use INTENDED USE Velotest Rotavirus Test is
HBeAg and HBeAg Ab ELISA Kit
 HBeAg and HBeAg Ab ELISA Kit Catalog Number KA0290 96 assays Version: 17 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Principle of the Assay... 3
HBeAg and HBeAg Ab ELISA Kit Catalog Number KA0290 96 assays Version: 17 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Principle of the Assay... 3
PLATELIA CANDIDA Ab PLUS Negative and Positive Controls. Quality Control Human Sera
 PLATELIA CANDIDA Ab PLUS Negative and Positive Controls 48 62787 Quality Control Human Sera CONTENTS 1. INTENDED USE...3 2. INDICATIONS FOR USE...3 3. REAGENTS...3 4. WARNING FOR USERS...4 5. PRECAUTIONS
PLATELIA CANDIDA Ab PLUS Negative and Positive Controls 48 62787 Quality Control Human Sera CONTENTS 1. INTENDED USE...3 2. INDICATIONS FOR USE...3 3. REAGENTS...3 4. WARNING FOR USERS...4 5. PRECAUTIONS
IV2-113E Use by. Invitron Glargine ELISA Kit REF LOT IVD. Definitions. English. For in-vitro diagnostic use. Instructions for use.
 Definitions Instructions for use REF Catalogue number IV2-113E Use by English Invitron Glargine ELISA Kit For in-vitro diagnostic use Σ 96 LOT IVD Lot/Batch Code Storage temperature limitations In vitro
Definitions Instructions for use REF Catalogue number IV2-113E Use by English Invitron Glargine ELISA Kit For in-vitro diagnostic use Σ 96 LOT IVD Lot/Batch Code Storage temperature limitations In vitro
Rapid-VIDITEST FOB Blister
 Rapid-VIDITEST FOB Blister One Step Fecal Occult Blood Blister test. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, Vestec, 252 42 Jesenice, Czech Republic, Tel.: +420 261 090 565,
Rapid-VIDITEST FOB Blister One Step Fecal Occult Blood Blister test. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, Vestec, 252 42 Jesenice, Czech Republic, Tel.: +420 261 090 565,
Staphylococci. Gram stain: gram positive cocci arranged in clusters.
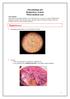 Microbiology lab Respiratory system Third medical year Lab contents: Gram positive bacteria (Staphylococcus and Streptococcus spp), two types of filamentous fungi (Aspergillus and Penicillium spp), and
Microbiology lab Respiratory system Third medical year Lab contents: Gram positive bacteria (Staphylococcus and Streptococcus spp), two types of filamentous fungi (Aspergillus and Penicillium spp), and
FAECAL WELL D-ONE. System for the presumptive identification and antibiotic susceptibility of pathogenic microorganisms of the intestinal tract.
 System for the presumptive identification and antibiotic susceptibility of pathogenic microorganisms of the intestinal tract. 1. INTRODUCTION Intestinal and gastrointestinal infections are a fairly common
System for the presumptive identification and antibiotic susceptibility of pathogenic microorganisms of the intestinal tract. 1. INTRODUCTION Intestinal and gastrointestinal infections are a fairly common
SAMPLE PROCEDURE , 07/11
 SAMPLE PROCEDURE This Sample Procedure is not intended as a substitute for your facility s Procedure Manual or reagent labeling, but rather as a model for your use in customizing for your laboratory s
SAMPLE PROCEDURE This Sample Procedure is not intended as a substitute for your facility s Procedure Manual or reagent labeling, but rather as a model for your use in customizing for your laboratory s
AN IMMUNOASSAY TEST FOR THE QUALITATIVE DETECTION OF STREP A ANTIGEN IN THROAT SWAB SPECIMENS
 CLARITY Strep A Dipsticks FOR LABORATORY AND PROFESSIONAL USE AN IMMUNOASSAY TEST FOR THE QUALITATIVE DETECTION OF STREP A ANTIGEN IN THROAT SWAB SPECIMENS CLIA COMPLEXITY: Waived CLARITY Strep A Dipsticks:
CLARITY Strep A Dipsticks FOR LABORATORY AND PROFESSIONAL USE AN IMMUNOASSAY TEST FOR THE QUALITATIVE DETECTION OF STREP A ANTIGEN IN THROAT SWAB SPECIMENS CLIA COMPLEXITY: Waived CLARITY Strep A Dipsticks:
CDIA TM Rubella IgG/IgM Rapid Test Kit
 CDIA TM Rubella IgG/IgM Rapid Test Kit Cat.No: DTSJZ024 Lot. No. (See product label) Intended Use The CDIA TM Rubella IgG/IgM Rapid Test Kit is a rapid chromatographic immunoassay for the qualitative detection
CDIA TM Rubella IgG/IgM Rapid Test Kit Cat.No: DTSJZ024 Lot. No. (See product label) Intended Use The CDIA TM Rubella IgG/IgM Rapid Test Kit is a rapid chromatographic immunoassay for the qualitative detection
H.Pylori IgM Cat # 1504Z
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC.
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
H.Pylori IgG
 DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
H. pylori IgM. Cat # H. pylori IgM ELISA. ELISA: Enzyme Linked Immunosorbent Assay. ELISA - Indirect; Antigen Coated Plate
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com H. pylori
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com H. pylori
H.Pylori IgG Cat # 1503Z
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
320 MBIO Microbial Diagnosis. Aljawharah F. Alabbad Noorah A. Alkubaisi 2017
 320 MBIO Microbial Diagnosis Aljawharah F. Alabbad Noorah A. Alkubaisi 2017 Pathogens of the Urinary tract The urinary system is composed of organs that regulate the chemical composition and volume of
320 MBIO Microbial Diagnosis Aljawharah F. Alabbad Noorah A. Alkubaisi 2017 Pathogens of the Urinary tract The urinary system is composed of organs that regulate the chemical composition and volume of
HAV IgG/IgM Rapid Test
 HAV IgG/IgM Rapid Test Cat. No.:DTS649 Pkg.Size: Intended use CD HAV IgG/IgM Rapid Test is a solid phase immunochromatographic assay for the rapid, qualitative and differential detection of IgG and IgM
HAV IgG/IgM Rapid Test Cat. No.:DTS649 Pkg.Size: Intended use CD HAV IgG/IgM Rapid Test is a solid phase immunochromatographic assay for the rapid, qualitative and differential detection of IgG and IgM
Malaria Pf/pan antigen Rapid Test
 Malaria Pf/pan antigen Rapid Test Cat. No.:IVDTS003 Pkg.Size:10T/50T Intended use The Malaria Pf/pan antigen Rapid Test is a self-performing, qualitative, sandwich immunoassay, utilizing whole blood for
Malaria Pf/pan antigen Rapid Test Cat. No.:IVDTS003 Pkg.Size:10T/50T Intended use The Malaria Pf/pan antigen Rapid Test is a self-performing, qualitative, sandwich immunoassay, utilizing whole blood for
A rapid test for the qualitative detection of Malaria pf and pv antigen in human blood sample
 A rapid test for the qualitative detection of Malaria pf and pv antigen in human blood sample For in vitro use only Intended Use For the rapid qualitative determination of Malaria P. falciparum specific
A rapid test for the qualitative detection of Malaria pf and pv antigen in human blood sample For in vitro use only Intended Use For the rapid qualitative determination of Malaria P. falciparum specific
CLIA Complexity: WAIVED
 CLIA Complexity: WAIVED INTENDED USE The is intended for the rapid, qualitative detection of Group A Streptococcal antigen from throat swabs or confirmation of presumptive Group A Streptococcal colonies
CLIA Complexity: WAIVED INTENDED USE The is intended for the rapid, qualitative detection of Group A Streptococcal antigen from throat swabs or confirmation of presumptive Group A Streptococcal colonies
Procine sphingomyelin ELISA Kit
 Procine sphingomyelin ELISA Kit For the quantitative in vitro determination of Procine sphingomyelin concentrations in serum - plasma - celiac fluid - tissue homogenate - body fluid FOR LABORATORY RESEARCH
Procine sphingomyelin ELISA Kit For the quantitative in vitro determination of Procine sphingomyelin concentrations in serum - plasma - celiac fluid - tissue homogenate - body fluid FOR LABORATORY RESEARCH
H.pylori IgA Cat #
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Comparison of Two Laboratory Techniques for Detecting Mycoplasmas in Genital Specimens. Osama Mohammed Saed Abdul-Wahab, BSc, MSc, PhD*
 Bahrain Medical Bulletin, Vol. 32, No. 4, December 200 Comparison of Two Laboratory Techniques for Detecting Mycoplasmas in Genital Specimens Osama Mohammed Saed Abdul-Wahab, BSc, MSc, PhD* Objective:
Bahrain Medical Bulletin, Vol. 32, No. 4, December 200 Comparison of Two Laboratory Techniques for Detecting Mycoplasmas in Genital Specimens Osama Mohammed Saed Abdul-Wahab, BSc, MSc, PhD* Objective:
Instruction Manual Test kit for 20 tests individually pouched Including Positive Control REF 41115
 QuickStripe + Positive control A rapid test device for the qualitative detection of Chlamydia trachomatis antigen in female cervical swab, male urethral swab and male urine specimens. Instruction Manual
QuickStripe + Positive control A rapid test device for the qualitative detection of Chlamydia trachomatis antigen in female cervical swab, male urethral swab and male urine specimens. Instruction Manual
See external label 2 C-8 C 96 tests Chemiluminescence. CMV IgM. Cat # Diluted samples, controls & calibrator 100 µl 30 minutes
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Malaria (Pan-LDH) W/B
 CORTEZ DIAGNOSTICS, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 USA Tel: (818) 591-3030 Fax: (818) 591-8383 E-mail: onestep@rapidtest.com Web site: www.rapidtest.com See external label
CORTEZ DIAGNOSTICS, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 USA Tel: (818) 591-3030 Fax: (818) 591-8383 E-mail: onestep@rapidtest.com Web site: www.rapidtest.com See external label
Rapid-VIDITEST. Rota-Adeno. One Step Rotavirus and Adenovirus Antigen Blister test. Instruction manual
 Rapid-VIDITEST Rota-Adeno One Step Rotavirus and Adenovirus Antigen Blister test. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, Vestec, 252 42 Jesenice, Czech Republic, Tel.: +420
Rapid-VIDITEST Rota-Adeno One Step Rotavirus and Adenovirus Antigen Blister test. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, Vestec, 252 42 Jesenice, Czech Republic, Tel.: +420
See external label 2 C-8 C Σ=96 tests Cat # 1505Z. MICROWELL ELISA H.Pylori IgA Cat # 1505Z
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
CMV FEP ALA For nucleic acid amplification of CMV DNA and Fluorescence detection with End Point analysis (FEP) on Aladin (Sacace)
 REF TV7-00FRT VER 3.0.06 CMV FEP ALA For nucleic acid amplification of CMV DNA and Fluorescence detection with End Point analysis (FEP) on Aladin (Sacace) Key to symbols used REF List Number Store at 2-8
REF TV7-00FRT VER 3.0.06 CMV FEP ALA For nucleic acid amplification of CMV DNA and Fluorescence detection with End Point analysis (FEP) on Aladin (Sacace) Key to symbols used REF List Number Store at 2-8
WHO Prequalification of Diagnostics Programme PUBLIC REPORT
 WHO Prequalification of Diagnostics Programme PUBLIC REPORT Product: ABON HIV 1/2/O Tri-Line Human Immunodeficiency Virus Rapid Test Device Number: PQDx 0141-051-00 Abstract ABON HIV 1/2/O Tri-Line Human
WHO Prequalification of Diagnostics Programme PUBLIC REPORT Product: ABON HIV 1/2/O Tri-Line Human Immunodeficiency Virus Rapid Test Device Number: PQDx 0141-051-00 Abstract ABON HIV 1/2/O Tri-Line Human
SAMPLE PROCEDURE 907-9, 06/11
 SAMPLE PROCEDURE This Sample Procedure is not intended as a substitute for your facility s Procedure Manual or reagent labeling, but rather as a model for your use in customizing for your laboratory s
SAMPLE PROCEDURE This Sample Procedure is not intended as a substitute for your facility s Procedure Manual or reagent labeling, but rather as a model for your use in customizing for your laboratory s
CYTOMEGALOVIRUS (CMV) IgM ELISA Kit Protocol
 CYTOMEGALOVIRUS (CMV) IgM ELISA Kit Protocol (Cat. No.:EK-310-91) 330 Beach Road, Burlingame CA Tel: 650-558-8898 Fax: 650-558-1686 E-Mail: info@phoenixpeptide.com www.phoenixpeptide.com INTENDED USE The
CYTOMEGALOVIRUS (CMV) IgM ELISA Kit Protocol (Cat. No.:EK-310-91) 330 Beach Road, Burlingame CA Tel: 650-558-8898 Fax: 650-558-1686 E-Mail: info@phoenixpeptide.com www.phoenixpeptide.com INTENDED USE The
See external label 96 tests HSV 2 IgA. Cat #
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Rapid-VIDITEST Calprotectin
 Rapid-VIDITEST Calprotectin One Step Calprotectin Card Test. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, 252 50 Vestec, Czech Republic, Tel.: +420 261 090 565, www.vidia.cz INTENDED
Rapid-VIDITEST Calprotectin One Step Calprotectin Card Test. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, 252 50 Vestec, Czech Republic, Tel.: +420 261 090 565, www.vidia.cz INTENDED
Rubella Latex Agglutination Test
 Rubella Latex Agglutination Test Cat. No.:DLAT1088 Pkg.Size:30T Intended use The Rubella Latex Agglutination Test is a rapid latex particle agglutination test for the qualitative and semi-quantitative
Rubella Latex Agglutination Test Cat. No.:DLAT1088 Pkg.Size:30T Intended use The Rubella Latex Agglutination Test is a rapid latex particle agglutination test for the qualitative and semi-quantitative
(For In Vitro Diagnostic Use)
 Read this insert carefully before performing the assay and keep for future reference. The reliability of assay procedure other than those described in this package insert cannot be guaranteed. 207727 (For
Read this insert carefully before performing the assay and keep for future reference. The reliability of assay procedure other than those described in this package insert cannot be guaranteed. 207727 (For
Cryptococcal Antigen Latex Agglutination Test
 Cryptococcal Antigen Latex Agglutination Test Cat. No.:DLAT1109 Pkg.Size:100T Intended use The Cryptococcal Antigen Latex Agglutination Test is a simple and rapid latex agglutination test for the qualitative
Cryptococcal Antigen Latex Agglutination Test Cat. No.:DLAT1109 Pkg.Size:100T Intended use The Cryptococcal Antigen Latex Agglutination Test is a simple and rapid latex agglutination test for the qualitative
Toxoplasma gondii IgM (Toxo IgM)
 DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
Herpes Simplex Virus 2 IgM HSV 2 IgM
 DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and 116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and 116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
INSTANT-VIEW H. pylori Rapid Test CLIA WAIVED Test for Whole Blood QUALITATIVE IN-VITRO DIAGNOSTIC TEST FOR EXTERNAL USE ONLY
 INTENDED USE INSTANT-VIEW H. pylori Rapid Test CLIA WAIVED Test for Whole Blood QUALITATIVE IN-VITRO DIAGNOSTIC TEST FOR EXTERNAL USE ONLY INSTANT-VIEW H. pylori Rapid Test is a rapid qualitative immunoassay
INTENDED USE INSTANT-VIEW H. pylori Rapid Test CLIA WAIVED Test for Whole Blood QUALITATIVE IN-VITRO DIAGNOSTIC TEST FOR EXTERNAL USE ONLY INSTANT-VIEW H. pylori Rapid Test is a rapid qualitative immunoassay
Sputum DNA Collection, Preservation and Isolation Kit 50 Individual Devices
 3430 Schmon Parkway Thorold, ON, Canada L2V 4Y6 Phone: 866-667-4362 (905) 227-8848 Fax: (905) 227-1061 Email: techsupport@norgenbiotek.com Sputum DNA Collection, Preservation and Isolation Kit 50 Individual
3430 Schmon Parkway Thorold, ON, Canada L2V 4Y6 Phone: 866-667-4362 (905) 227-8848 Fax: (905) 227-1061 Email: techsupport@norgenbiotek.com Sputum DNA Collection, Preservation and Isolation Kit 50 Individual
MALDI Sepsityper Kit
 Instructions for Use MALDI Sepsityper Kit Kit for identification of microorganisms from positive blood cultures using the MALDI Biotyper system CARE products are designed to support our worldwide customers
Instructions for Use MALDI Sepsityper Kit Kit for identification of microorganisms from positive blood cultures using the MALDI Biotyper system CARE products are designed to support our worldwide customers
Human Cytomegalovirus IgM ELISA Kit
 Human Cytomegalovirus IgM Catalog No: IRAPKT2012 ELISA Kit Lot No: SAMPLE INTENDED USE The CMV IgM ELISA is intended for use in the detection of IgM antibodies to Cytomegalovirus (CMV) infection in human
Human Cytomegalovirus IgM Catalog No: IRAPKT2012 ELISA Kit Lot No: SAMPLE INTENDED USE The CMV IgM ELISA is intended for use in the detection of IgM antibodies to Cytomegalovirus (CMV) infection in human
H. pylori IgM CLIA kit
 H. pylori IgM CLIA kit Cat. No.:DEEL0251 Pkg.Size:96 tests Intended use Helicobacter pylori IgM Chemiluminescence ELISA is intended for use in evaluating the serologic status to H. pylori infection in
H. pylori IgM CLIA kit Cat. No.:DEEL0251 Pkg.Size:96 tests Intended use Helicobacter pylori IgM Chemiluminescence ELISA is intended for use in evaluating the serologic status to H. pylori infection in
Rubella virus IgG ELISA Kit
 Rubella virus IgG ELISA Kit Catalog Number KA0223 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
Rubella virus IgG ELISA Kit Catalog Number KA0223 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
Infectious Mononucleosis IM Cassette Test RAPU04A830
 Infectious Mononucleosis IM Cassette Test RAPU04A830 DIAsource ImmunoAssays S.A. - Rue de l'industrie, 8 - B-1400 Nivelles - Belgium : 090714/1 en DIAsource IM (Mononucleosis) Test Cassette for whole blood,
Infectious Mononucleosis IM Cassette Test RAPU04A830 DIAsource ImmunoAssays S.A. - Rue de l'industrie, 8 - B-1400 Nivelles - Belgium : 090714/1 en DIAsource IM (Mononucleosis) Test Cassette for whole blood,
Rapid-VIDITEST. Astrovirus Card
 Rapid-VIDITEST Astrovirus Card One step Astrovirus Card Test. Instruction manual INTENDED USE: The Rapid-VIDITEST Astrovirus Card is a one step coloured chromatographic immunoassay for the qualitative
Rapid-VIDITEST Astrovirus Card One step Astrovirus Card Test. Instruction manual INTENDED USE: The Rapid-VIDITEST Astrovirus Card is a one step coloured chromatographic immunoassay for the qualitative
Toxoplasma gondii IgM ELISA Kit
 Toxoplasma gondii IgM ELISA Kit Catalog Number KA0226 96 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Toxoplasma gondii IgM ELISA Kit Catalog Number KA0226 96 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Human Cytomegalovirus Virus (CMV) IgG ELISA Kit
 Human Cytomegalovirus Virus Catalog No: IRAPKT1410 (CMV) IgG ELISA Kit Lot No: SAMPLE INTENDED USE The CMV IgG ELISA is intended for use in evaluating a patient s serologic status to cytomegalovirus (CMV)
Human Cytomegalovirus Virus Catalog No: IRAPKT1410 (CMV) IgG ELISA Kit Lot No: SAMPLE INTENDED USE The CMV IgG ELISA is intended for use in evaluating a patient s serologic status to cytomegalovirus (CMV)
Ultra-sensitive Human TSH ELISA Kit. MyBioSource.com
 Ultra-sensitive Human TSH ELISA Kit For the quantitative determination of human thyroid stimulating hormone (TSH) concentrations in serum Catalog Number: MBS590037 96 tests FOR LABORATORY RESEARCH USE
Ultra-sensitive Human TSH ELISA Kit For the quantitative determination of human thyroid stimulating hormone (TSH) concentrations in serum Catalog Number: MBS590037 96 tests FOR LABORATORY RESEARCH USE
USO PROFESSIONALE PROFESSIONAL USE
 Gima S.p.A. - Via Marconi, 1-20060 Gessate (MI) Italy Italia: tel. 199 400 401 - fax 199 400 403 Export: tel. +39 02 953854209/221/225 fax +39 02 95380056 gima@gimaitaly.com - export@gimaitaly.com www.gimaitaly.com
Gima S.p.A. - Via Marconi, 1-20060 Gessate (MI) Italy Italia: tel. 199 400 401 - fax 199 400 403 Export: tel. +39 02 953854209/221/225 fax +39 02 95380056 gima@gimaitaly.com - export@gimaitaly.com www.gimaitaly.com
ABIOpure TM Viral (version 2.0)
 ABIOpure TM Viral (version 2.0) DNA/RNA Extraction Handbook Cat No: M561VT50 FOR RESEARCH USE ONLY Table of Contents Contents Page Kit Components 3 Precautions 3 Stability & Storage 4 General Description
ABIOpure TM Viral (version 2.0) DNA/RNA Extraction Handbook Cat No: M561VT50 FOR RESEARCH USE ONLY Table of Contents Contents Page Kit Components 3 Precautions 3 Stability & Storage 4 General Description
Pneumocystis Carinii Real Time PCR Kit. For In Vitro Diagnostic Use Only User Manual
 Revision No.: ZJ0003 Issue Date: Aug 7 th, 2008 Pneumocystis Carinii Real Time PCR Kit Cat. No.: QD-0082-02 For use with ABI Prism 7000/7300/7500/7900; Smart CyclerII; icycler iq 4/iQ 5; Rotor Gene 2000/3000;
Revision No.: ZJ0003 Issue Date: Aug 7 th, 2008 Pneumocystis Carinii Real Time PCR Kit Cat. No.: QD-0082-02 For use with ABI Prism 7000/7300/7500/7900; Smart CyclerII; icycler iq 4/iQ 5; Rotor Gene 2000/3000;
PreventID TSH. Additional information Manual Medical information
 Additional information Manual Medical information Manual (for professional use) PreventID TSH (KST72116) The PreventID TSH is a rapid test for the determination of TSH in capillary blood, serum or whole
Additional information Manual Medical information Manual (for professional use) PreventID TSH (KST72116) The PreventID TSH is a rapid test for the determination of TSH in capillary blood, serum or whole
Influenza A IgG ELISA
 Influenza A IgG ELISA For the qualitative determination of IgG-class antibodies against Influenza A virus in human serum or plasma (citrate, heparin). For Research Use Only. Not For Use In Diagnostic Procedures.
Influenza A IgG ELISA For the qualitative determination of IgG-class antibodies against Influenza A virus in human serum or plasma (citrate, heparin). For Research Use Only. Not For Use In Diagnostic Procedures.
Hepatitis A virus IgM ELISA Kit
 Hepatitis A virus IgM ELISA Kit Catalog Number KA0285 96 assays Version: 04 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Hepatitis A virus IgM ELISA Kit Catalog Number KA0285 96 assays Version: 04 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
See external label 2 C-8 C = C-REACTIVE PROTEIN (CRP) LATEX SLIDE TEST
 CORTEZ DIAGNOSTICS, INC. 21250 Califa Street, Suite 102 and 116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
CORTEZ DIAGNOSTICS, INC. 21250 Califa Street, Suite 102 and 116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
MycoBlue TM Mycoplasma Detector
 MycoBlue TM Mycoplasma Detector Catalog # D101 Version 5.1 Vazyme biotech co., ltd. Introduction MycoBlue TM Mycoplasma Detector is a rapid detection of mycoplasma kit for cell culture. Its core technology
MycoBlue TM Mycoplasma Detector Catalog # D101 Version 5.1 Vazyme biotech co., ltd. Introduction MycoBlue TM Mycoplasma Detector is a rapid detection of mycoplasma kit for cell culture. Its core technology
Mouse C-Peptide ELISA Kit
 Mouse C-Peptide ELISA Kit Cat.No: DEIA4507 Lot. No. (See product label) Size 96T Intended Use The Mouse C-Peptide ELISA kit is for the quantitative determination of c-peptide in mouse serum, plasma, and
Mouse C-Peptide ELISA Kit Cat.No: DEIA4507 Lot. No. (See product label) Size 96T Intended Use The Mouse C-Peptide ELISA kit is for the quantitative determination of c-peptide in mouse serum, plasma, and
Campylobacter Antigen ELISA Kit
 Campylobacter Antigen ELISA Kit Catalog Number KA3204 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Campylobacter Antigen ELISA Kit Catalog Number KA3204 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
QUICK REFERENCE INSTRUCTIONS. THYROCHEK TSH Cassette
 QUICK REFERENCE INSTRUCTIONS THYROCHEK TSH Cassette A certificate of CLIA waiver is required to perform the testing in a waived setting. If the laboratory does not have a Certificate of Waiver, the Application
QUICK REFERENCE INSTRUCTIONS THYROCHEK TSH Cassette A certificate of CLIA waiver is required to perform the testing in a waived setting. If the laboratory does not have a Certificate of Waiver, the Application
See external label 2 C-8 C 96 tests CHEMILUMINESCENCE. CMV IgG. Cat # Step (20-25 C Room temp.) Volume
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Neonatal Pneumonia-2 assay
 Neonatal Pneumonia-2 assay For the detection of Ureaplasma species (Ureaplasma parvum and Ureaplasma urealyticum), Chlamydia trachomatis and Mycoplasma hominis using the BD MAX TM system. Instructions
Neonatal Pneumonia-2 assay For the detection of Ureaplasma species (Ureaplasma parvum and Ureaplasma urealyticum), Chlamydia trachomatis and Mycoplasma hominis using the BD MAX TM system. Instructions
VGKC-Autoantibody Assay RIA
 Instructions for Use VGKC-Autoantibody Assay RIA 125I-Radio Immuno Assay for the Quantitative Determination of Antibodies to the Voltage-Gated Potassium Channel (VGKC) in Serum I V D REF RA115/25 25 2
Instructions for Use VGKC-Autoantibody Assay RIA 125I-Radio Immuno Assay for the Quantitative Determination of Antibodies to the Voltage-Gated Potassium Channel (VGKC) in Serum I V D REF RA115/25 25 2
Chlamydia Trachomatis IgA
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
cobas PCR Media Uni Swab Sample Kit
 For in vitro diagnostic use cobas PCR Media Uni Swab Sample Kit P/N: 07958030190 Table of contents Intended use... 3 Reagents and materials... 4 cobas PCR Media Uni Swab Sample Kit... 4 Precautions and
For in vitro diagnostic use cobas PCR Media Uni Swab Sample Kit P/N: 07958030190 Table of contents Intended use... 3 Reagents and materials... 4 cobas PCR Media Uni Swab Sample Kit... 4 Precautions and
PNEUMOCOCCUS VALENT LATEX KIT
 PNEUMOCOCCUS 7-10-13-VALENT LATEX KIT Latex particles coated with pneumococcal antiserum raised in rabbits for in vitro diagnostic use Application The Pneumococcus 7-10-13-valent Latex Kit is a ready to
PNEUMOCOCCUS 7-10-13-VALENT LATEX KIT Latex particles coated with pneumococcal antiserum raised in rabbits for in vitro diagnostic use Application The Pneumococcus 7-10-13-valent Latex Kit is a ready to
MALARIA P. FALCIPARUM / P. VIVAX
 MALARIA P. FALCIPARUM / P. VIVAX 1. EXPLANATION OF THE TEST: Malaria is a serious, sometimes fatal, parasitic disease characterized by fever, chills, and anemia and is caused by a parasite that is transmitted
MALARIA P. FALCIPARUM / P. VIVAX 1. EXPLANATION OF THE TEST: Malaria is a serious, sometimes fatal, parasitic disease characterized by fever, chills, and anemia and is caused by a parasite that is transmitted
Carnitine / Acylcarnitines Dried Blood Spots LC-MS/MS Analysis Kit User Manual
 Page 1 / 14 Carnitine / Acylcarnitines Dried Blood Spots LC-MS/MS Analysis Kit User Manual ZV-3051-0200-15 200 Page 2 / 14 Table of Contents 1. INTENDED USE... 3 2. SUMMARY AND EXPLANATION... 3 3. TEST
Page 1 / 14 Carnitine / Acylcarnitines Dried Blood Spots LC-MS/MS Analysis Kit User Manual ZV-3051-0200-15 200 Page 2 / 14 Table of Contents 1. INTENDED USE... 3 2. SUMMARY AND EXPLANATION... 3 3. TEST
Instructions for use. TSH rat ELISA. Please use only the valid version of the Instructions for Use provided with the kit AR E-8600
 Instructions for use TSH rat ELISA AR E8600 TSH rat ELISA INTRODUCTION INTENDED USE The TSH rat ELISA is an enzyme immunoassay for the quantitative measurement of TSH in rat serum. For research use only.
Instructions for use TSH rat ELISA AR E8600 TSH rat ELISA INTRODUCTION INTENDED USE The TSH rat ELISA is an enzyme immunoassay for the quantitative measurement of TSH in rat serum. For research use only.
I.C.E. Embryo Vitrification Kit
 I.C.E. Embryo Vitrification Kit Vitrification Media V1, V2, V3 ICE Embryo Vitrification Instructions For Use Testing and Cautions Innvative Cryo Enterprises LLC 317 Springfield Road Linden, New Jersey
I.C.E. Embryo Vitrification Kit Vitrification Media V1, V2, V3 ICE Embryo Vitrification Instructions For Use Testing and Cautions Innvative Cryo Enterprises LLC 317 Springfield Road Linden, New Jersey
Cotinine (Mouse/Rat) ELISA Kit
 Cotinine (Mouse/Rat) ELISA Kit Catalog Number KA2264 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Cotinine (Mouse/Rat) ELISA Kit Catalog Number KA2264 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
